User login
How Should Hypertensive Emergencies Be Managed?
Case
A 57-year-old male with hypertension and end-stage renal disease is brought into the ED by his family for evaluation of headache, nausea, blurry vision, and confusion. Blood pressure is 235/130 mmHg. He is somnolent but arousable and oriented only to person; the remainder of his neurologic exam is nonfocal. A fundoscopic exam shows retinal hemorrhages, exudates, and papilledema. How should this patient be managed?
Overview
Hypertension (HTN) is a medical problem that affects an estimated 1 in 3 adults in the U.S. and more than 1 billion people worldwide. The Joint National Committee (JNC) 7 Report defines hypertensive emergency as severe hypertension with evidence of impending or progressive end-organ dysfunction.1 Systolic blood pressure (SBP) in these settings often is >180 mm Hg with diastolic blood pressure (DBP) >120 mm Hg. The JNC 7 Report defines hypertensive urgency as severe HTN without acute end-organ dysfunction. Whereas hypertensive urgencies can be treated with oral antihypertensive agents with close outpatient follow-up, hypertensive emergencies require immediate BP reduction to halt the progression of end-organ damage.
Severe HTN causes shear stress and endothelial injury, leading to activation of the coagulation cascade, fibrinoid necrosis, and tissue ischemia.2 Due to adaptive vascular changes, pre-existing hypertension lowers the probability of a hypertensive emergency developing at a particular BP. The rate of BP rise, rather than the absolute level, determines most end-organ damage.3 In previously normotensive patients, end-organ damage can occur at BPs >160/100 mm Hg; however, organ dysfunction is uncommon in chronically hypertensive individuals, unless BP >220/120 mm Hg.
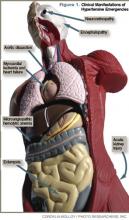
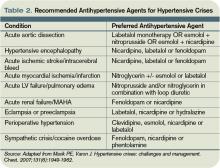
Clinical manifestations of hypertensive emergency depend on the target organs involved (see Figure 1, right). When a patient presents with severe hypertension, a focused evaluation should attempt to identify the presence of end-organ damage. If present, these patients should be admitted to an ICU for close monitoring, and administration of parenteral antihypertensive agents should be started. (Online Exclusive: View a chart of “Parenteral Antihypertensive Agents Used in Hypertensive Emergencies”)
Review of the Data
General principles: The initial therapeutic goal in most hypertensive emergencies is to reduce the mean arterial pressure (MAP) by no more than 25% within the first hour. Precipitous or excessive decreases in BP might worsen renal, cerebral, or coronary ischemia. Due to pressure natriuresis, patients with primary malignant hypertension might be volume-depleted. Restoration of intravascular volume with intravenous (IV) saline can prevent precipitous falls in BP when antihypertensive agents are started.
After the patient stabilizes, the BP can be lowered about 10% per hour to 160/100-110 mm Hg. A gradual reduction to the patient’s baseline BP is targeted over the ensuing 24 to 48 hours. Once there is stable BP control and end-organ damage has ceased, patients can be transitioned to oral therapy.
No large clinical trials have investigated optimal drug therapy in patients with hypertensive emergencies. The choice of pharmacologic agent should be individualized based on drug properties, patient comorbidities, and the end-organ(s) involved.
Selected pharmacologic agents: Sodium nitroprusside (SNP) is a short-acting, potent arterial and venous dilator that has been used extensively in the treatment of hypertensive emergencies. Despite its familiarity, there are several important limitations to its use. SNP can increase intracranial pressure (ICP), worsen myocardial ischemia through coronary steal, and is associated with cyanide and/or thiocyanate toxicity. Although used broadly across many types of hypertensive emergencies, SNP should be considered a first-line agent in acute left ventricular (LV) failure and, when combined with beta-blockers, in acute aortic dissection.
Labetalol is an alpha-1 and nonselective beta-blocker that reduces systemic vascular resistance while preserving cerebral, renal, and coronary blood flow. It is considered a first-line agent in most hypertensive emergencies, with the exception of acute LV failure.
Esmolol is a short-acting, selective beta-blocker that decreases heart rate, myocardial contractility, and cardiac output.
Nicardipine is a second-generation dihydropyridine calcium channel blocker. Although it has a longer duration of action, excess hypotension has not been seen in clinical trials comparing it with SNP.4 Nicardipine is used safely in such hypertensive emergencies as hypertensive encephalopathy, cerebral vascular accidents, and postoperatively.
Fenoldopam creates vasodilation by acting on peripheral dopamine type 1 receptors. It improves creatinine clearance and urine output, and is most useful in acute kidney injury.5 It is a well-tolerated and highly effective agent for use in most hypertensive crises, although is expensive and has limited hard outcome data.
Nitroglycerin is a potent venodilator that is used as an adjunct to other anti-hypertensives in the treatment of acute coronary syndromes and acute pulmonary edema.
Immediate-release nifedipine and clonidine are not recommended; they are long-acting and poorly titratable, with unpredictable hypotensive effects.
Hydralazine may be used in LV failure and in pregnancy.
Specific emergencies: Aortic dissection is the most rapidly fatal complication of severe HTN. Untreated, approximately 80% of patients with acute type-A dissections die within two weeks.6 In this specific setting, SBP should be decreased as rapidly as possible to <110 mm Hg in order to halt propagation of the dissection prior to surgery. Therapy should aim to reduce the shear stress on the aortic wall by decreasing both BP and heart rate. This can be accomplished with a combination of esmolol and SNP. Nicardipine and fenoldopam are effective alternatives to SNP. Labetalol is a good single-agent option, provided adequate heart rate suppression is achieved.
LV failure and acute pulmonary edema are associated with high systemic vascular resistance and activation of the Renin Angiotensin Aldosterone (RAAS) system. First-line therapy should consist of arterial vasodilators (e.g., SNP, nicardipine, fenoldopam) in combination with a loop diuretic. Nitroglycerin can be used as an adjunct to reduce LV preload.
In hypertensive encephalopathy, blood pressure exceeds the cerebral autoregulatory threshold, leading to breakthrough vasodilation and the development of cerebral edema. Characteristic symptoms include the insidious onset of headache, nausea, vomiting, and nonlocalizing neurologic signs (e.g., lethargy, confusion, seizures). It is important to exclude stroke, as treatment strategies differ. SNP is used widely in the treatment of hypertensive encephalopathy; it may increase ICP and should be used with caution. Nicardipine and labetalol are effective alternatives with favorable cerebral hemodynamic profiles.
Malignant HTN is characterized by neuroretinopathy: cotton wool spots, flame hemorrhages, and papilledema. Encephalopathy and other evidence of end-organ dysfunction might not be present, although renal disease is common. Preferred drugs are SNP and labetalol, although fenoldopam has been used successfully.
Appropriate BP management following acute ischemic stroke remains controversial. Elevated BP often is a protective physiologic response to maintain cerebral perfusion. The American Heart Association (AHA) recommends initiating IV antihypertensive therapy for thrombolysis candidates when SBP >185 or DBP >110 mm Hg. For those who are not thrombolysis candidates, the recommended threshold for initiating IV antihypertensives is SBP >220 or DBP >120 mm Hg.7 The goal is to lower the BP by 15% to 25% within the first 24 hours. These goals are less aggressive than in patients with hypertensive encephalopathy without stroke.
Spontaneous intracerebral hemorrhage causes a rise in ICP with reflex systemic hypertension. Although a correlation between HTN and hematoma expansion exists, there is no evidence that shows lowering BP is protective. Two clinical trials are evaluating the effects of BP reduction to specified target levels.8 Pending those results, the AHA recommends BP reduction for patients with SBP >200 or MAP >150 mm Hg, or for patients with SBP >180 or MAP >130 mm Hg and evidence of elevated ICP.7 In both ischemic and hemorrhagic stroke, nicardipine and labetalol are appropriate first-line agents.
Most sympathetic crises are related to the recreational use of sympathomimetic drugs, pheochromocytoma, abrupt antihypertensive withdrawal, or concurrent ingestion of monoamine-oxidase inhibitors and tyramine-containing foods. Selective beta-blockers can increase BP and worsen HTN through unopposed alpha effects.
Although labetalol traditionally has been considered the ideal agent—due to its alpha and beta antagonism—studies have failed to support its use in this clinical setting.9 Phentolamine, nicardipine, and fenoldopam are reasonable selections.
Hypertension is common in the early postoperative period following cardiothoracic, vascular, head and neck, and neurosurgical procedures. No consensus exists regarding the treatment of noncardiac surgery patients, but treatment is recommended for BP >140/90 or MAP >105 mmHg in cardiac surgery patients. Nicardipine, clevidipine, and esmolol are proven agents. All three have been shown more effective than SNP in maintaining target BP, and each is associated with less BP variability.10
In patients with pregnancy-induced hypertension, initial therapy for preeclampsia includes magnesium sulfate for seizure prophylaxis and BP control until delivery of the fetus can be safely undertaken. The FDA does not recommend any specific antihypertensive agents; however, ACE inhibitors and SNP are contraindicated. Although hydralazine is used extensively in this setting, a meta-analysis showed increased risk of maternal hypotension, Cesarean section, placental abruptions, and low Apgar scores.11 Labetalol and nicardipine appear to be safe and effective in pregnant hypertensive patients.
Back to the Case
This case represents a classic presentation of malignant hypertension with hypertensive encephalopathy, which is reversible with timely and appropriate management. The patient’s MAP is approximately 165 mmHg, well above the upper threshold of cerebral vascular autoregulation in most patients with chronic hypertension. A brain MRI should be obtained to definitively rule out stroke, as management goals would be considerably different.
If the scan is negative, treatment should be initiated immediately with a goal of reducing the MAP by no more than 25% within the first hour. Nicardipine or labetalol would be appropriate therapeutic choices, administered in an ICU with close hemodynamic monitoring.
Given the patient’s end-stage renal disease and evidence of intracranial hypertension, SNP would be a suboptimal choice. Over hours two through six, BP could be lowered gradually to 160/100, then to his baseline BP over the ensuing 24 to 48 hours, monitoring closely for signs of neurologic deterioration. Once BP is stable and there is no evidence of worsening end-organ damage, he can be safely transitioned to oral agents.
Bottom Line
The therapeutic goal in hypertensive emergencies is to immediately and safely lower BP to halt end-organ damage. Drug selection should be individualized. TH
Dr. Shanahan is a hospitalist and assistant professor at the Denver VA Medical Center. Dr. Linas is professor of medicine in the division of renal diseases and hypertension at the University of Colorado Denver School of Medicine. Dr. Anderson is associate professor and chief of the hospital medicine section at the Denver VA Medical Center.
References
- Lenfant C, Chobanian AV, Jones DW, Roccella EJ. Seventh report of the Joint National Committee on the Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7): resetting the hypertension sails. Hypertension. 2003;41(6):1178-1179.
- Ault MJ, Ellrodt AG. Pathophysiological events leading to the end-organ effects of acute hypertension. Am J Emerg Med. 1985;3(6 Suppl):10-15.
- Vaughan CJ, Delanty N. Hypertensive emergencies. Lancet. 2000;356(9227):411-417.
- Neutel JM, Smith DH, Wallin D, et al. A comparison of intravenous nicardipine and sodium nitroprusside in the immediate treatment of severe hypertension. Am J Hypertens. 1994;7(7 Pt 1):623-628.
- Shusterman NH, Elliott WJ, White WB. Fenoldopam, but not nitroprusside, improves renal function in severely hypertensive patients with impaired renal function. Am J Med. 1993;95(2):161-168.
- Khan IA, Nair CK. Clinical, diagnostic, and management perspectives of aortic dissection. Chest. 2002;122(1):311-328.
- Adams HP Jr., del Zoppo G, Alberts MJ, et al. Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Circulation. 2007;115(20):e478-534.
- Qureshi AI. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH): rationale and design. Neurocrit Care. 2007;6(1):56-66.
- Marik PE, Varon J. Hypertensive crises: challenges and management. Chest. 2007;131(6):1949-1962.
- Aronson S, Dyke CM, Stierer KA, et al. The ECLIPSE trials: comparative studies of clevidipine to nitroglycerin, sodium nitroprusside, and nicardipine for acute hypertension treatment in cardiac surgery patients. Anesth Analg. 2008;107(4):1110-1121.
- Magee LA, Cham C, Waterman EJ, Ohlsson A, von Dadelszen P. Hydralazine for treatment of severe hypertension in pregnancy: meta-analysis. BMJ. 2003;327(7421):955-960.
- Aggarwal M, Khan IA. Hypertensive crisis: hypertensive emergencies and urgencies. Cardiol Clin. 2006; 24(1):135-146.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 1. Am J Health Syst Pharm. 2009;66(15):1343-1352.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 2. Am J Health Syst Pharm. 2009;66(16):1448-1457.
- Varon J. Treatment of acute severe hypertension: current and newer agents. Drugs. 2008;68(3):283-297.
Case
A 57-year-old male with hypertension and end-stage renal disease is brought into the ED by his family for evaluation of headache, nausea, blurry vision, and confusion. Blood pressure is 235/130 mmHg. He is somnolent but arousable and oriented only to person; the remainder of his neurologic exam is nonfocal. A fundoscopic exam shows retinal hemorrhages, exudates, and papilledema. How should this patient be managed?
Overview
Hypertension (HTN) is a medical problem that affects an estimated 1 in 3 adults in the U.S. and more than 1 billion people worldwide. The Joint National Committee (JNC) 7 Report defines hypertensive emergency as severe hypertension with evidence of impending or progressive end-organ dysfunction.1 Systolic blood pressure (SBP) in these settings often is >180 mm Hg with diastolic blood pressure (DBP) >120 mm Hg. The JNC 7 Report defines hypertensive urgency as severe HTN without acute end-organ dysfunction. Whereas hypertensive urgencies can be treated with oral antihypertensive agents with close outpatient follow-up, hypertensive emergencies require immediate BP reduction to halt the progression of end-organ damage.
Severe HTN causes shear stress and endothelial injury, leading to activation of the coagulation cascade, fibrinoid necrosis, and tissue ischemia.2 Due to adaptive vascular changes, pre-existing hypertension lowers the probability of a hypertensive emergency developing at a particular BP. The rate of BP rise, rather than the absolute level, determines most end-organ damage.3 In previously normotensive patients, end-organ damage can occur at BPs >160/100 mm Hg; however, organ dysfunction is uncommon in chronically hypertensive individuals, unless BP >220/120 mm Hg.
Clinical manifestations of hypertensive emergency depend on the target organs involved (see Figure 1, right). When a patient presents with severe hypertension, a focused evaluation should attempt to identify the presence of end-organ damage. If present, these patients should be admitted to an ICU for close monitoring, and administration of parenteral antihypertensive agents should be started. (Online Exclusive: View a chart of “Parenteral Antihypertensive Agents Used in Hypertensive Emergencies”)
Review of the Data
General principles: The initial therapeutic goal in most hypertensive emergencies is to reduce the mean arterial pressure (MAP) by no more than 25% within the first hour. Precipitous or excessive decreases in BP might worsen renal, cerebral, or coronary ischemia. Due to pressure natriuresis, patients with primary malignant hypertension might be volume-depleted. Restoration of intravascular volume with intravenous (IV) saline can prevent precipitous falls in BP when antihypertensive agents are started.
After the patient stabilizes, the BP can be lowered about 10% per hour to 160/100-110 mm Hg. A gradual reduction to the patient’s baseline BP is targeted over the ensuing 24 to 48 hours. Once there is stable BP control and end-organ damage has ceased, patients can be transitioned to oral therapy.
No large clinical trials have investigated optimal drug therapy in patients with hypertensive emergencies. The choice of pharmacologic agent should be individualized based on drug properties, patient comorbidities, and the end-organ(s) involved.
Selected pharmacologic agents: Sodium nitroprusside (SNP) is a short-acting, potent arterial and venous dilator that has been used extensively in the treatment of hypertensive emergencies. Despite its familiarity, there are several important limitations to its use. SNP can increase intracranial pressure (ICP), worsen myocardial ischemia through coronary steal, and is associated with cyanide and/or thiocyanate toxicity. Although used broadly across many types of hypertensive emergencies, SNP should be considered a first-line agent in acute left ventricular (LV) failure and, when combined with beta-blockers, in acute aortic dissection.
Labetalol is an alpha-1 and nonselective beta-blocker that reduces systemic vascular resistance while preserving cerebral, renal, and coronary blood flow. It is considered a first-line agent in most hypertensive emergencies, with the exception of acute LV failure.
Esmolol is a short-acting, selective beta-blocker that decreases heart rate, myocardial contractility, and cardiac output.
Nicardipine is a second-generation dihydropyridine calcium channel blocker. Although it has a longer duration of action, excess hypotension has not been seen in clinical trials comparing it with SNP.4 Nicardipine is used safely in such hypertensive emergencies as hypertensive encephalopathy, cerebral vascular accidents, and postoperatively.
Fenoldopam creates vasodilation by acting on peripheral dopamine type 1 receptors. It improves creatinine clearance and urine output, and is most useful in acute kidney injury.5 It is a well-tolerated and highly effective agent for use in most hypertensive crises, although is expensive and has limited hard outcome data.
Nitroglycerin is a potent venodilator that is used as an adjunct to other anti-hypertensives in the treatment of acute coronary syndromes and acute pulmonary edema.
Immediate-release nifedipine and clonidine are not recommended; they are long-acting and poorly titratable, with unpredictable hypotensive effects.
Hydralazine may be used in LV failure and in pregnancy.
Specific emergencies: Aortic dissection is the most rapidly fatal complication of severe HTN. Untreated, approximately 80% of patients with acute type-A dissections die within two weeks.6 In this specific setting, SBP should be decreased as rapidly as possible to <110 mm Hg in order to halt propagation of the dissection prior to surgery. Therapy should aim to reduce the shear stress on the aortic wall by decreasing both BP and heart rate. This can be accomplished with a combination of esmolol and SNP. Nicardipine and fenoldopam are effective alternatives to SNP. Labetalol is a good single-agent option, provided adequate heart rate suppression is achieved.
LV failure and acute pulmonary edema are associated with high systemic vascular resistance and activation of the Renin Angiotensin Aldosterone (RAAS) system. First-line therapy should consist of arterial vasodilators (e.g., SNP, nicardipine, fenoldopam) in combination with a loop diuretic. Nitroglycerin can be used as an adjunct to reduce LV preload.
In hypertensive encephalopathy, blood pressure exceeds the cerebral autoregulatory threshold, leading to breakthrough vasodilation and the development of cerebral edema. Characteristic symptoms include the insidious onset of headache, nausea, vomiting, and nonlocalizing neurologic signs (e.g., lethargy, confusion, seizures). It is important to exclude stroke, as treatment strategies differ. SNP is used widely in the treatment of hypertensive encephalopathy; it may increase ICP and should be used with caution. Nicardipine and labetalol are effective alternatives with favorable cerebral hemodynamic profiles.
Malignant HTN is characterized by neuroretinopathy: cotton wool spots, flame hemorrhages, and papilledema. Encephalopathy and other evidence of end-organ dysfunction might not be present, although renal disease is common. Preferred drugs are SNP and labetalol, although fenoldopam has been used successfully.
Appropriate BP management following acute ischemic stroke remains controversial. Elevated BP often is a protective physiologic response to maintain cerebral perfusion. The American Heart Association (AHA) recommends initiating IV antihypertensive therapy for thrombolysis candidates when SBP >185 or DBP >110 mm Hg. For those who are not thrombolysis candidates, the recommended threshold for initiating IV antihypertensives is SBP >220 or DBP >120 mm Hg.7 The goal is to lower the BP by 15% to 25% within the first 24 hours. These goals are less aggressive than in patients with hypertensive encephalopathy without stroke.
Spontaneous intracerebral hemorrhage causes a rise in ICP with reflex systemic hypertension. Although a correlation between HTN and hematoma expansion exists, there is no evidence that shows lowering BP is protective. Two clinical trials are evaluating the effects of BP reduction to specified target levels.8 Pending those results, the AHA recommends BP reduction for patients with SBP >200 or MAP >150 mm Hg, or for patients with SBP >180 or MAP >130 mm Hg and evidence of elevated ICP.7 In both ischemic and hemorrhagic stroke, nicardipine and labetalol are appropriate first-line agents.
Most sympathetic crises are related to the recreational use of sympathomimetic drugs, pheochromocytoma, abrupt antihypertensive withdrawal, or concurrent ingestion of monoamine-oxidase inhibitors and tyramine-containing foods. Selective beta-blockers can increase BP and worsen HTN through unopposed alpha effects.
Although labetalol traditionally has been considered the ideal agent—due to its alpha and beta antagonism—studies have failed to support its use in this clinical setting.9 Phentolamine, nicardipine, and fenoldopam are reasonable selections.
Hypertension is common in the early postoperative period following cardiothoracic, vascular, head and neck, and neurosurgical procedures. No consensus exists regarding the treatment of noncardiac surgery patients, but treatment is recommended for BP >140/90 or MAP >105 mmHg in cardiac surgery patients. Nicardipine, clevidipine, and esmolol are proven agents. All three have been shown more effective than SNP in maintaining target BP, and each is associated with less BP variability.10
In patients with pregnancy-induced hypertension, initial therapy for preeclampsia includes magnesium sulfate for seizure prophylaxis and BP control until delivery of the fetus can be safely undertaken. The FDA does not recommend any specific antihypertensive agents; however, ACE inhibitors and SNP are contraindicated. Although hydralazine is used extensively in this setting, a meta-analysis showed increased risk of maternal hypotension, Cesarean section, placental abruptions, and low Apgar scores.11 Labetalol and nicardipine appear to be safe and effective in pregnant hypertensive patients.
Back to the Case
This case represents a classic presentation of malignant hypertension with hypertensive encephalopathy, which is reversible with timely and appropriate management. The patient’s MAP is approximately 165 mmHg, well above the upper threshold of cerebral vascular autoregulation in most patients with chronic hypertension. A brain MRI should be obtained to definitively rule out stroke, as management goals would be considerably different.
If the scan is negative, treatment should be initiated immediately with a goal of reducing the MAP by no more than 25% within the first hour. Nicardipine or labetalol would be appropriate therapeutic choices, administered in an ICU with close hemodynamic monitoring.
Given the patient’s end-stage renal disease and evidence of intracranial hypertension, SNP would be a suboptimal choice. Over hours two through six, BP could be lowered gradually to 160/100, then to his baseline BP over the ensuing 24 to 48 hours, monitoring closely for signs of neurologic deterioration. Once BP is stable and there is no evidence of worsening end-organ damage, he can be safely transitioned to oral agents.
Bottom Line
The therapeutic goal in hypertensive emergencies is to immediately and safely lower BP to halt end-organ damage. Drug selection should be individualized. TH
Dr. Shanahan is a hospitalist and assistant professor at the Denver VA Medical Center. Dr. Linas is professor of medicine in the division of renal diseases and hypertension at the University of Colorado Denver School of Medicine. Dr. Anderson is associate professor and chief of the hospital medicine section at the Denver VA Medical Center.
References
- Lenfant C, Chobanian AV, Jones DW, Roccella EJ. Seventh report of the Joint National Committee on the Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7): resetting the hypertension sails. Hypertension. 2003;41(6):1178-1179.
- Ault MJ, Ellrodt AG. Pathophysiological events leading to the end-organ effects of acute hypertension. Am J Emerg Med. 1985;3(6 Suppl):10-15.
- Vaughan CJ, Delanty N. Hypertensive emergencies. Lancet. 2000;356(9227):411-417.
- Neutel JM, Smith DH, Wallin D, et al. A comparison of intravenous nicardipine and sodium nitroprusside in the immediate treatment of severe hypertension. Am J Hypertens. 1994;7(7 Pt 1):623-628.
- Shusterman NH, Elliott WJ, White WB. Fenoldopam, but not nitroprusside, improves renal function in severely hypertensive patients with impaired renal function. Am J Med. 1993;95(2):161-168.
- Khan IA, Nair CK. Clinical, diagnostic, and management perspectives of aortic dissection. Chest. 2002;122(1):311-328.
- Adams HP Jr., del Zoppo G, Alberts MJ, et al. Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Circulation. 2007;115(20):e478-534.
- Qureshi AI. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH): rationale and design. Neurocrit Care. 2007;6(1):56-66.
- Marik PE, Varon J. Hypertensive crises: challenges and management. Chest. 2007;131(6):1949-1962.
- Aronson S, Dyke CM, Stierer KA, et al. The ECLIPSE trials: comparative studies of clevidipine to nitroglycerin, sodium nitroprusside, and nicardipine for acute hypertension treatment in cardiac surgery patients. Anesth Analg. 2008;107(4):1110-1121.
- Magee LA, Cham C, Waterman EJ, Ohlsson A, von Dadelszen P. Hydralazine for treatment of severe hypertension in pregnancy: meta-analysis. BMJ. 2003;327(7421):955-960.
- Aggarwal M, Khan IA. Hypertensive crisis: hypertensive emergencies and urgencies. Cardiol Clin. 2006; 24(1):135-146.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 1. Am J Health Syst Pharm. 2009;66(15):1343-1352.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 2. Am J Health Syst Pharm. 2009;66(16):1448-1457.
- Varon J. Treatment of acute severe hypertension: current and newer agents. Drugs. 2008;68(3):283-297.
Case
A 57-year-old male with hypertension and end-stage renal disease is brought into the ED by his family for evaluation of headache, nausea, blurry vision, and confusion. Blood pressure is 235/130 mmHg. He is somnolent but arousable and oriented only to person; the remainder of his neurologic exam is nonfocal. A fundoscopic exam shows retinal hemorrhages, exudates, and papilledema. How should this patient be managed?
Overview
Hypertension (HTN) is a medical problem that affects an estimated 1 in 3 adults in the U.S. and more than 1 billion people worldwide. The Joint National Committee (JNC) 7 Report defines hypertensive emergency as severe hypertension with evidence of impending or progressive end-organ dysfunction.1 Systolic blood pressure (SBP) in these settings often is >180 mm Hg with diastolic blood pressure (DBP) >120 mm Hg. The JNC 7 Report defines hypertensive urgency as severe HTN without acute end-organ dysfunction. Whereas hypertensive urgencies can be treated with oral antihypertensive agents with close outpatient follow-up, hypertensive emergencies require immediate BP reduction to halt the progression of end-organ damage.
Severe HTN causes shear stress and endothelial injury, leading to activation of the coagulation cascade, fibrinoid necrosis, and tissue ischemia.2 Due to adaptive vascular changes, pre-existing hypertension lowers the probability of a hypertensive emergency developing at a particular BP. The rate of BP rise, rather than the absolute level, determines most end-organ damage.3 In previously normotensive patients, end-organ damage can occur at BPs >160/100 mm Hg; however, organ dysfunction is uncommon in chronically hypertensive individuals, unless BP >220/120 mm Hg.
Clinical manifestations of hypertensive emergency depend on the target organs involved (see Figure 1, right). When a patient presents with severe hypertension, a focused evaluation should attempt to identify the presence of end-organ damage. If present, these patients should be admitted to an ICU for close monitoring, and administration of parenteral antihypertensive agents should be started. (Online Exclusive: View a chart of “Parenteral Antihypertensive Agents Used in Hypertensive Emergencies”)
Review of the Data
General principles: The initial therapeutic goal in most hypertensive emergencies is to reduce the mean arterial pressure (MAP) by no more than 25% within the first hour. Precipitous or excessive decreases in BP might worsen renal, cerebral, or coronary ischemia. Due to pressure natriuresis, patients with primary malignant hypertension might be volume-depleted. Restoration of intravascular volume with intravenous (IV) saline can prevent precipitous falls in BP when antihypertensive agents are started.
After the patient stabilizes, the BP can be lowered about 10% per hour to 160/100-110 mm Hg. A gradual reduction to the patient’s baseline BP is targeted over the ensuing 24 to 48 hours. Once there is stable BP control and end-organ damage has ceased, patients can be transitioned to oral therapy.
No large clinical trials have investigated optimal drug therapy in patients with hypertensive emergencies. The choice of pharmacologic agent should be individualized based on drug properties, patient comorbidities, and the end-organ(s) involved.
Selected pharmacologic agents: Sodium nitroprusside (SNP) is a short-acting, potent arterial and venous dilator that has been used extensively in the treatment of hypertensive emergencies. Despite its familiarity, there are several important limitations to its use. SNP can increase intracranial pressure (ICP), worsen myocardial ischemia through coronary steal, and is associated with cyanide and/or thiocyanate toxicity. Although used broadly across many types of hypertensive emergencies, SNP should be considered a first-line agent in acute left ventricular (LV) failure and, when combined with beta-blockers, in acute aortic dissection.
Labetalol is an alpha-1 and nonselective beta-blocker that reduces systemic vascular resistance while preserving cerebral, renal, and coronary blood flow. It is considered a first-line agent in most hypertensive emergencies, with the exception of acute LV failure.
Esmolol is a short-acting, selective beta-blocker that decreases heart rate, myocardial contractility, and cardiac output.
Nicardipine is a second-generation dihydropyridine calcium channel blocker. Although it has a longer duration of action, excess hypotension has not been seen in clinical trials comparing it with SNP.4 Nicardipine is used safely in such hypertensive emergencies as hypertensive encephalopathy, cerebral vascular accidents, and postoperatively.
Fenoldopam creates vasodilation by acting on peripheral dopamine type 1 receptors. It improves creatinine clearance and urine output, and is most useful in acute kidney injury.5 It is a well-tolerated and highly effective agent for use in most hypertensive crises, although is expensive and has limited hard outcome data.
Nitroglycerin is a potent venodilator that is used as an adjunct to other anti-hypertensives in the treatment of acute coronary syndromes and acute pulmonary edema.
Immediate-release nifedipine and clonidine are not recommended; they are long-acting and poorly titratable, with unpredictable hypotensive effects.
Hydralazine may be used in LV failure and in pregnancy.
Specific emergencies: Aortic dissection is the most rapidly fatal complication of severe HTN. Untreated, approximately 80% of patients with acute type-A dissections die within two weeks.6 In this specific setting, SBP should be decreased as rapidly as possible to <110 mm Hg in order to halt propagation of the dissection prior to surgery. Therapy should aim to reduce the shear stress on the aortic wall by decreasing both BP and heart rate. This can be accomplished with a combination of esmolol and SNP. Nicardipine and fenoldopam are effective alternatives to SNP. Labetalol is a good single-agent option, provided adequate heart rate suppression is achieved.
LV failure and acute pulmonary edema are associated with high systemic vascular resistance and activation of the Renin Angiotensin Aldosterone (RAAS) system. First-line therapy should consist of arterial vasodilators (e.g., SNP, nicardipine, fenoldopam) in combination with a loop diuretic. Nitroglycerin can be used as an adjunct to reduce LV preload.
In hypertensive encephalopathy, blood pressure exceeds the cerebral autoregulatory threshold, leading to breakthrough vasodilation and the development of cerebral edema. Characteristic symptoms include the insidious onset of headache, nausea, vomiting, and nonlocalizing neurologic signs (e.g., lethargy, confusion, seizures). It is important to exclude stroke, as treatment strategies differ. SNP is used widely in the treatment of hypertensive encephalopathy; it may increase ICP and should be used with caution. Nicardipine and labetalol are effective alternatives with favorable cerebral hemodynamic profiles.
Malignant HTN is characterized by neuroretinopathy: cotton wool spots, flame hemorrhages, and papilledema. Encephalopathy and other evidence of end-organ dysfunction might not be present, although renal disease is common. Preferred drugs are SNP and labetalol, although fenoldopam has been used successfully.
Appropriate BP management following acute ischemic stroke remains controversial. Elevated BP often is a protective physiologic response to maintain cerebral perfusion. The American Heart Association (AHA) recommends initiating IV antihypertensive therapy for thrombolysis candidates when SBP >185 or DBP >110 mm Hg. For those who are not thrombolysis candidates, the recommended threshold for initiating IV antihypertensives is SBP >220 or DBP >120 mm Hg.7 The goal is to lower the BP by 15% to 25% within the first 24 hours. These goals are less aggressive than in patients with hypertensive encephalopathy without stroke.
Spontaneous intracerebral hemorrhage causes a rise in ICP with reflex systemic hypertension. Although a correlation between HTN and hematoma expansion exists, there is no evidence that shows lowering BP is protective. Two clinical trials are evaluating the effects of BP reduction to specified target levels.8 Pending those results, the AHA recommends BP reduction for patients with SBP >200 or MAP >150 mm Hg, or for patients with SBP >180 or MAP >130 mm Hg and evidence of elevated ICP.7 In both ischemic and hemorrhagic stroke, nicardipine and labetalol are appropriate first-line agents.
Most sympathetic crises are related to the recreational use of sympathomimetic drugs, pheochromocytoma, abrupt antihypertensive withdrawal, or concurrent ingestion of monoamine-oxidase inhibitors and tyramine-containing foods. Selective beta-blockers can increase BP and worsen HTN through unopposed alpha effects.
Although labetalol traditionally has been considered the ideal agent—due to its alpha and beta antagonism—studies have failed to support its use in this clinical setting.9 Phentolamine, nicardipine, and fenoldopam are reasonable selections.
Hypertension is common in the early postoperative period following cardiothoracic, vascular, head and neck, and neurosurgical procedures. No consensus exists regarding the treatment of noncardiac surgery patients, but treatment is recommended for BP >140/90 or MAP >105 mmHg in cardiac surgery patients. Nicardipine, clevidipine, and esmolol are proven agents. All three have been shown more effective than SNP in maintaining target BP, and each is associated with less BP variability.10
In patients with pregnancy-induced hypertension, initial therapy for preeclampsia includes magnesium sulfate for seizure prophylaxis and BP control until delivery of the fetus can be safely undertaken. The FDA does not recommend any specific antihypertensive agents; however, ACE inhibitors and SNP are contraindicated. Although hydralazine is used extensively in this setting, a meta-analysis showed increased risk of maternal hypotension, Cesarean section, placental abruptions, and low Apgar scores.11 Labetalol and nicardipine appear to be safe and effective in pregnant hypertensive patients.
Back to the Case
This case represents a classic presentation of malignant hypertension with hypertensive encephalopathy, which is reversible with timely and appropriate management. The patient’s MAP is approximately 165 mmHg, well above the upper threshold of cerebral vascular autoregulation in most patients with chronic hypertension. A brain MRI should be obtained to definitively rule out stroke, as management goals would be considerably different.
If the scan is negative, treatment should be initiated immediately with a goal of reducing the MAP by no more than 25% within the first hour. Nicardipine or labetalol would be appropriate therapeutic choices, administered in an ICU with close hemodynamic monitoring.
Given the patient’s end-stage renal disease and evidence of intracranial hypertension, SNP would be a suboptimal choice. Over hours two through six, BP could be lowered gradually to 160/100, then to his baseline BP over the ensuing 24 to 48 hours, monitoring closely for signs of neurologic deterioration. Once BP is stable and there is no evidence of worsening end-organ damage, he can be safely transitioned to oral agents.
Bottom Line
The therapeutic goal in hypertensive emergencies is to immediately and safely lower BP to halt end-organ damage. Drug selection should be individualized. TH
Dr. Shanahan is a hospitalist and assistant professor at the Denver VA Medical Center. Dr. Linas is professor of medicine in the division of renal diseases and hypertension at the University of Colorado Denver School of Medicine. Dr. Anderson is associate professor and chief of the hospital medicine section at the Denver VA Medical Center.
References
- Lenfant C, Chobanian AV, Jones DW, Roccella EJ. Seventh report of the Joint National Committee on the Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7): resetting the hypertension sails. Hypertension. 2003;41(6):1178-1179.
- Ault MJ, Ellrodt AG. Pathophysiological events leading to the end-organ effects of acute hypertension. Am J Emerg Med. 1985;3(6 Suppl):10-15.
- Vaughan CJ, Delanty N. Hypertensive emergencies. Lancet. 2000;356(9227):411-417.
- Neutel JM, Smith DH, Wallin D, et al. A comparison of intravenous nicardipine and sodium nitroprusside in the immediate treatment of severe hypertension. Am J Hypertens. 1994;7(7 Pt 1):623-628.
- Shusterman NH, Elliott WJ, White WB. Fenoldopam, but not nitroprusside, improves renal function in severely hypertensive patients with impaired renal function. Am J Med. 1993;95(2):161-168.
- Khan IA, Nair CK. Clinical, diagnostic, and management perspectives of aortic dissection. Chest. 2002;122(1):311-328.
- Adams HP Jr., del Zoppo G, Alberts MJ, et al. Guidelines for the early management of adults with ischemic stroke: a guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology affirms the value of this guideline as an educational tool for neurologists. Circulation. 2007;115(20):e478-534.
- Qureshi AI. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH): rationale and design. Neurocrit Care. 2007;6(1):56-66.
- Marik PE, Varon J. Hypertensive crises: challenges and management. Chest. 2007;131(6):1949-1962.
- Aronson S, Dyke CM, Stierer KA, et al. The ECLIPSE trials: comparative studies of clevidipine to nitroglycerin, sodium nitroprusside, and nicardipine for acute hypertension treatment in cardiac surgery patients. Anesth Analg. 2008;107(4):1110-1121.
- Magee LA, Cham C, Waterman EJ, Ohlsson A, von Dadelszen P. Hydralazine for treatment of severe hypertension in pregnancy: meta-analysis. BMJ. 2003;327(7421):955-960.
- Aggarwal M, Khan IA. Hypertensive crisis: hypertensive emergencies and urgencies. Cardiol Clin. 2006; 24(1):135-146.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 1. Am J Health Syst Pharm. 2009;66(15):1343-1352.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 2. Am J Health Syst Pharm. 2009;66(16):1448-1457.
- Varon J. Treatment of acute severe hypertension: current and newer agents. Drugs. 2008;68(3):283-297.
Change You Should Believe In
Christina Payne, MD, is a third-year resident at Emory University Hospital in Atlanta who will begin her first hospitalist job, with Emory in September. In spite of her dearth of practical experience, she already has experience researching one of the most vexing problems confronting HM: how to improve transitions of care.
Dr. Payne has been studying the benefits of a structured electronic tool that generates a standardized sign-out list of a hospital team’s full census at the time of shift change, compared with the usual, highly variable sign-out practices of medical residents. At a poster presentation at Internal Medicine 2010 in April in Toronto, Dr. Payne and colleagues reported that residents using the tool were twice as confident at performing handoffs, had lower rates of perceived near-miss events, and were happier.1
“Hospitalists everywhere are starting to realize the importance of trying to reduce opportunities for human error that occur during care transitions,” Dr. Payne says. “The biggest thing I learned from this research is the importance of standardizing the handoff process [with information communicated consistently].
“It is essential to keep communication lines open,” Dr. Payne adds. “No tool can replace the importance of communication between doctors and the need to sit down and talk. The ideal signout happens in a quiet room where the two of you can talk about active patients and achieve rapport. But, realistically, how often does that happen?”
Standardization is one of a handful of strategies hospitalists, researchers, and policymakers are using to tackle transitions—both in-hospital handoffs and post-discharge transitions—with outpatient care. Some hospitalists are using practice simulations and training strategies; others have implemented medication reconciliation checks at every discharge, checklists and other communication strategies, team-based quality-improvement (QI) initiatives, and new technologies to enhance and streamline communication. Some interventions follow the patient from the hospital to the community physician with a phone call, follow-up clinic, or other contact; others aim to empower the patient to be a better self-advocate. But for hospitalists, the challenge is to communicate the right amount of transfer information to the right receiver at the right time.
No matter the technique, the goal is the same: Improve the handoff and discharge process in a way that promotes efficiency and patient safety. And hospitalists are at the forefront of the changing landscape of care transitions.
Under the Microscope
Care transitions of all kinds are under the magnifying glass of national healthcare reform, with growing recognition of the need to make care safer and reduce the preventable, costly hospital readmissions caused by incomplete handoffs. Care transitions for hospitalists include internal handoffs, both at daily shift changes and at service changes when an outgoing provider is leaving after a period of consecutive daily shifts. These typically involve a sign-out process and face-to-face encounter, with some kind of written backup. One teaching institution reported that such handoffs take place 4,000 times per day in the hospital, or 1.6 million times per year.2

—Arpana Vidyarthi, MD, University of California at San Francisco
Geographical transitions can be from one floor or department to another, or out the hospital door to another facility or home. Transitions typically involve a discharge process and a written discharge summary. Care transitions also include hospital admissions, which put the hospitalist in the role of handoff receiver rather than initiator, plus a variety of other transitions involving nurses, physician extenders, and other practitioners.
Each transition is a major decision point in the course of a patient’s hospitalization; each transition also presents a time of heightened vulnerability (e.g., potential communication breakdowns, medication errors, patient anxiety or confusion, etc.). In fact, according to a Transitions of Care Consensus Policy Statement published in 2009 by SHM and five other medical societies, handoffs are ubiquitous in HM, with significant patient safety and quality deficiencies in handoffs existing in the current system.3
Poor communication at the time of handoff has been implicated in near-misses and adverse events in a variety of healthcare contexts, including 70% of hospital sentinel events studied by The Joint Commission, which named standardized handoffs (with an opportunity for interactive communication) as a National Patient Safety Goal in 2006.4 The federal government is studying care transitions, supporting demonstration projects for Medicare enrollees, and including readmission rates in national hospital report card data.
“Transitions of care and handoffs are a huge focus right now because of the increased fragmentation of care in the United States. Hospitalists are in charge of a greater percentage of hospitalized patients, which means more coordination of care is needed,” says Vineet Arora, MD, MA, FHM, assistant professor of medicine and associate director of the internal-medicine residency at the University of Chicago, and chair of the SHM task force on handoffs.
Inadequate communication and poor care transitions can undermine hospitalists’ best care-planning efforts, erode patients’ and families’ confidence and satisfaction with hospital care, and leave primary-care physicians (PCPs) feeling unsatisfied with the relationship. As many as 1 in 5 Medicare beneficiary hospitalizations result in a readmission within 30 days, and while not all of these are preventable, far too many are.5 Another prospective cohort study found that 1 in 5 patients discharged from the hospital to the home experienced an adverse event within three weeks of discharge.6 Complex comorbidities, advanced age, unknown PCP, and limited healthcare literacy present hospitalists with extremely difficult transitions.
Patient safety and cost control are the linchpins to national efforts to improve transitions of care. Dr. Arora recently coauthored an original research paper, which will be published in the Journal of Hospital Medicine in September, showing older hospitalized patients are twice as likely to report problems after discharge if their PCPs were not aware they were hospitalized.
“With escalating healthcare costs, people are looking at ways to save money and reduce redundant care,” Dr. Arora explains, pointing out, as an example, repeated tests resulting from inadequate communication between healthcare providers.
The System Must Change
“All of the effort we put into saving someone’s life—the years of experience, training, medical school, and residency—all of it comes to bear on that hospitalized patient. And it can all be unraveled at the time of discharge if it’s not handled properly,” says Arpana Vidyarthi, MD, a hospitalist and director of quality at the University of California at San Francisco.
Dr. Vidyarthi views in-hospital and discharge transitions as integrally related. “The analysis is similar, even if different techniques may be needed,” she says, adding that, fundamentally, it involves having a system that allows people—or forces them—to do the “right thing.”
That’s why achieving effective care transitions will require more than just a standardized tool or process, Dr. Vidyarthi says. “This is about understanding the ways people communicate and finding ways to train them to communicate better,” she says. “The problem we have is not a lack of information, but how to communicate what, to whom, and when.”
What’s really needed, Dr. Vidyarthi says, is a hospital’s commitment to more effective transitions and its hospitalists’ leadership in driving a comprehensive, multidisciplinary, team- and evidence-based QI process. The new process should be a QI-based solution to a hospital’s care-transitions issues. “Before you can standardize your process, you need to understand it,” she says. “This is a complex problem, and it needs a multifaceted solution. But this lies squarely within the hospitalist arena. We’re part of everything that happens in the hospital.

—Anuj Dalal, MD, Brigham and Women’s Hospital, Boston
Hospital administrators are looking to HM to solve transition and readmission problems now, says Tina Budnitz, MPH, BOOST Project Director (Better Outcomes for Older Adults through Safe Transitions). She expects the scrutiny from the C-suite, legislators, and watchdog groups to increase as the spotlight continues to shine on the healthcare system.
“Any hospitalist can act as a leader in their institution,” Budnitz says. “Be a change agent, pull a group together, and start asking questions: Do we have safe care-transitions practices and processes in place? Just by asking the right question, you can be a catalyst for the system.”
Budnitz also emphasizes the importance of teamwork in the hospital setting. “How can I help my teammates? What am I communicating to the nurses on rounds?” she says. “Can you initiate dialogue with your outpatient medical groups: ‘These faxes we’re sending you—is that information getting to you in ways and times that are helpful? And, by the way, when your patient is admitted, this information would really help me.’ ”
Innovative Strategies
One of the most important initiatives responding to concerns about care transitions is Project BOOST (www.hos pitalmedicine.org/BOOST), a comprehensive toolkit for improving a hospital’s transitions of care. The project aims to build a national consensus for best practices in transitions; collaborate with representatives from the Agency for Healthcare Research and Quality (AHRQ), the Centers for Medicare and Medicaid Services (CMS), and the Joint Commission; and develop a national resource library, Budnitz says.
“Project BOOST not only puts forth best practices for admitting patients, planning for discharge, and then doing the discharge, it also helps show facilities how to change their systems, with resources and tools for analyzing and re-engineering the system,” she says. “Sites get one-to-one assistance from a mentor.”
Six hospitals signed on to the pilot program in 2008; 24 more joined last year. In January, SHM announced a collaborative with the University of Michigan and Blue Cross Blue Shield of Michigan for 15 Michigan hospitals to receive training and mentorship starting in May. And last month, SHM and the California HealthCare Foundation announced a Project BOOST initiative for 20 of the health system’s hospitals (see “California Dreamin’”, p. 6). Other free resources offered on the BOOST Web portal include clinical, data collection, and project management tools. SHM also has a DVD that explains how to use the “teachback” method to improve communication with patients.
Jennifer Myers, MD, FHM, assistant professor of clinical medicine and patient-safety officer at the Hospital of the University of Pennsylvania in Philadelphia, is a Project BOOST participant who spearheaded a process change to improve the quality of her facility’s discharge summary, along with accompanying resident education.7 The discharge summary recently was integrated with the hospital’s electronic health record (EHR) system.
“We’ve gone from dictating the discharge summary to an electronic version completed by the hospitalist, with prompts for key components of the summary, which allows us to create summaries more efficiently—ideally on the day of discharge, but usually within 48 hours,” Dr. Myers says. “We previously researched whether teaching made a difference in the quality of discharges; we found that it did. So we look forward to standardizing our teaching approach around this important topic for all residents.”
Another care-transitions innovation receiving a lot of attention from the government and the private sector is Project RED (Re-Engineered Discharge), led by Brian Jack, MD, vice chair of the department of family medicine at Boston Medical Center. The Project RED research group develops and tests strategies to improve the hospital discharge process to promote patient safety and reduce rehospitalization rates.
“We used re-engineering tools borrowed from other fields, brought together experts from all over the hospital, divided up the whole discharge process, and identified key principles,” Dr. Jack explains. The resulting discharge strategy is reflected in an 11-item checklist of discrete, mutually reinforcing components, which have been shown to reduce rehospitalization rates by 32% while raising patient satisfaction.8 It includes comprehensive discharge and after-hospital plans, a nurse discharge advocate, and a medication reconciliation phone call to the patient. A virtual “patient advocate,” a computerized avatar named Louise, is now being tested. If successful, it will allow patients to interact with a touch-screen teacher of the after-care plan who has time to work at the patient’s pace.
Technology and Transitions
Informatics can be a key player in facilitating care transitions, says Anuj Dalal, MD, a hospitalist and instructor in medicine at Brigham and Women’s Hospital in Boston. He is using one of his hospital’s technological strengths—a well-established, firewall-protected e-mail system—to help improve the discharge process.
“We decided to try to improve awareness of test results pending at the time of discharge,” Dr. Dalal explains. “We created an intervention that automatically triggers an e-mail with the finalized test results to the responsible providers. The intervention creates a loop of communication between the inpatient attending and the PCP. What we hope to show in our research over the next year or two is whether the intervention actually increases awareness of test results by providers.”
One thing to remember is that “all kinds of things can go wrong with care transitions,” no matter the size of the institution, the experience of the staff, or technological limitations, says Vineet Chopra, MD, FACP, a hospitalist at the University of Michigan Health System in Ann Arbor. “The problems of transitions vary from place to place, day to day, time of day, shift changes; and let’s not forget physician extenders and the other members of the healthcare team,” he says. “The more complicated the team, the more complicated the information needing to be handed off becomes.”
Before he joined the group at the university, Dr. Chopra worked at a community hospital, St. Joseph’s Mercy Hospital in Hot Springs, Ark. “It’s hard to come up with a one-size-fits-all solution when there are so many variables,” he says. At the community hospital, “we mandated that the hospitalist call the PCP at the time of discharge. At the academic medical center, we share an EHR with the PCPs and can reach them electronically. We are required to have the discharge summary in the computer before the patient leaves the hospital, and we mandate that hospitalists are reachable by e-mail or phone when they are off.
“I’m not a believer in throwing more technology at problems and just adding more layers of information tools,” Dr. Chopra adds. “Hospitalists who used to carry stethoscopes now also have a clipboard, phone, pager, PDA, and nine different signouts in their pockets. What we want to do is make their life easier. Here, we are looking at technology as a means to do that.”
Dr. Chopra and hospitalist colleague Prasanth Gosineni, MD, have been working with an Ann Arbor tech company called Synaptin to develop a lightweight, mobile client application designed to work on smartphones. Still in pilot testing, it would allow for task-oriented and priority-based messaging in real time and the systematic transfer of important information for the next hospitalist shift.
“You need to be able to share information in a systematic way, but that’s only half of the answer. The other half is the ability to ask specific questions,” Dr. Chopra says. “Technology doesn’t take away from the face-to-face encounter that needs to happen. Nothing will replace face time, but part of the solution is to provide data efficiently and in a way that is easily accessible.”
Dr. Chopra admits that EHR presents both positives and negatives to improved transitions and patient care, “depending on how well it works and what smart features it offers,” he says, “but also recognizing that EHR and other technologies have also taken us farther away from face-to-face exchanges. Some would say that’s part of the problem.”
Handoffs, discharges, and other transitions are ubiquitous in HM—and fraught with the potential for costly and harmful errors. The ideal of an interactive, face-to-face handoff simply is not available for many care transitions. However, hospitalists are challenged to find solutions that will work in their hospitals, with their teams, and their types of patients. Patients and policymakers expect nothing less. TH
Larry Beresford is a freelance writer based in Oakland, Calif.
References
- Payne C, Stein J, Dressler D. Implementation of a structured electronic tool to improve patient handoffs and resident satisfaction. Poster abstract: Internal Medicine 2010, April 21-24, 2010, Toronto.
- Vidyarthi AR. Triple Handoff. AHRQ WebM&M website. Available at: webmm.ahrq.gov/case.aspx? caseID=134. Published May 2006. Accessed May 29, 2010.
- Snow V, Beck D, Budnitz T, et al. Transitions of Care Consensus Policy Statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, and Society for Academic Emergency Medicine. J Hosp Med. 2009;4(6):364-370.
- 2006 National Patient Safety Goals. The Joint Commission website. Available at: www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/06_npsgs.htm. Accessed June 8, 2010.
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009; 2:360:1418-1428.
- Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW. The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161-167.
- Myers JS, Jaipaul CK, Kogan JR, Krekun S, Bellini LM, Shea JA. Are discharge summaries teachable? The effects of a discharge summary curriculum on the quality of discharge summaries in an internal medicine residency program. Acad Med. 2006; 81(10):S5-S8.
- Jack BW, Chetty VK, Anthony D, et al. A reengineered hospital discharge program to decrease rehospitalization: a randomized trial. Ann Intern Med. 2009;150(3):178-187.
- Arora VM, Manjarrez E, Dressler DD, Basaviah P, Halasyamani L, Kripalani S. Hospitalist handoffs: a systematic review and task force recommendations. J Hosp Med. 2009;4(7): 433-440.
- Halasyamani L, Kripalani S, Coleman E, et al. Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists. J Hosp Med. 2006;1(6):354-360.
- Schnipper JL, Kirwin JL, Cotugno MC, et al. Role of pharmacist counseling in preventing adverse drug events after hospitalization. Arch Int Med. 2006;166(5):565-571.
- Dudas V, Bookwalter T, Kerr KM, Pantilat SZ. The impact of follow-up telephone calls to patients after hospitalization. Am J Med. 2001;111(9B): 26S-30S.
Christina Payne, MD, is a third-year resident at Emory University Hospital in Atlanta who will begin her first hospitalist job, with Emory in September. In spite of her dearth of practical experience, she already has experience researching one of the most vexing problems confronting HM: how to improve transitions of care.
Dr. Payne has been studying the benefits of a structured electronic tool that generates a standardized sign-out list of a hospital team’s full census at the time of shift change, compared with the usual, highly variable sign-out practices of medical residents. At a poster presentation at Internal Medicine 2010 in April in Toronto, Dr. Payne and colleagues reported that residents using the tool were twice as confident at performing handoffs, had lower rates of perceived near-miss events, and were happier.1
“Hospitalists everywhere are starting to realize the importance of trying to reduce opportunities for human error that occur during care transitions,” Dr. Payne says. “The biggest thing I learned from this research is the importance of standardizing the handoff process [with information communicated consistently].
“It is essential to keep communication lines open,” Dr. Payne adds. “No tool can replace the importance of communication between doctors and the need to sit down and talk. The ideal signout happens in a quiet room where the two of you can talk about active patients and achieve rapport. But, realistically, how often does that happen?”
Standardization is one of a handful of strategies hospitalists, researchers, and policymakers are using to tackle transitions—both in-hospital handoffs and post-discharge transitions—with outpatient care. Some hospitalists are using practice simulations and training strategies; others have implemented medication reconciliation checks at every discharge, checklists and other communication strategies, team-based quality-improvement (QI) initiatives, and new technologies to enhance and streamline communication. Some interventions follow the patient from the hospital to the community physician with a phone call, follow-up clinic, or other contact; others aim to empower the patient to be a better self-advocate. But for hospitalists, the challenge is to communicate the right amount of transfer information to the right receiver at the right time.
No matter the technique, the goal is the same: Improve the handoff and discharge process in a way that promotes efficiency and patient safety. And hospitalists are at the forefront of the changing landscape of care transitions.
Under the Microscope
Care transitions of all kinds are under the magnifying glass of national healthcare reform, with growing recognition of the need to make care safer and reduce the preventable, costly hospital readmissions caused by incomplete handoffs. Care transitions for hospitalists include internal handoffs, both at daily shift changes and at service changes when an outgoing provider is leaving after a period of consecutive daily shifts. These typically involve a sign-out process and face-to-face encounter, with some kind of written backup. One teaching institution reported that such handoffs take place 4,000 times per day in the hospital, or 1.6 million times per year.2

—Arpana Vidyarthi, MD, University of California at San Francisco
Geographical transitions can be from one floor or department to another, or out the hospital door to another facility or home. Transitions typically involve a discharge process and a written discharge summary. Care transitions also include hospital admissions, which put the hospitalist in the role of handoff receiver rather than initiator, plus a variety of other transitions involving nurses, physician extenders, and other practitioners.
Each transition is a major decision point in the course of a patient’s hospitalization; each transition also presents a time of heightened vulnerability (e.g., potential communication breakdowns, medication errors, patient anxiety or confusion, etc.). In fact, according to a Transitions of Care Consensus Policy Statement published in 2009 by SHM and five other medical societies, handoffs are ubiquitous in HM, with significant patient safety and quality deficiencies in handoffs existing in the current system.3
Poor communication at the time of handoff has been implicated in near-misses and adverse events in a variety of healthcare contexts, including 70% of hospital sentinel events studied by The Joint Commission, which named standardized handoffs (with an opportunity for interactive communication) as a National Patient Safety Goal in 2006.4 The federal government is studying care transitions, supporting demonstration projects for Medicare enrollees, and including readmission rates in national hospital report card data.
“Transitions of care and handoffs are a huge focus right now because of the increased fragmentation of care in the United States. Hospitalists are in charge of a greater percentage of hospitalized patients, which means more coordination of care is needed,” says Vineet Arora, MD, MA, FHM, assistant professor of medicine and associate director of the internal-medicine residency at the University of Chicago, and chair of the SHM task force on handoffs.
Inadequate communication and poor care transitions can undermine hospitalists’ best care-planning efforts, erode patients’ and families’ confidence and satisfaction with hospital care, and leave primary-care physicians (PCPs) feeling unsatisfied with the relationship. As many as 1 in 5 Medicare beneficiary hospitalizations result in a readmission within 30 days, and while not all of these are preventable, far too many are.5 Another prospective cohort study found that 1 in 5 patients discharged from the hospital to the home experienced an adverse event within three weeks of discharge.6 Complex comorbidities, advanced age, unknown PCP, and limited healthcare literacy present hospitalists with extremely difficult transitions.
Patient safety and cost control are the linchpins to national efforts to improve transitions of care. Dr. Arora recently coauthored an original research paper, which will be published in the Journal of Hospital Medicine in September, showing older hospitalized patients are twice as likely to report problems after discharge if their PCPs were not aware they were hospitalized.
“With escalating healthcare costs, people are looking at ways to save money and reduce redundant care,” Dr. Arora explains, pointing out, as an example, repeated tests resulting from inadequate communication between healthcare providers.
The System Must Change
“All of the effort we put into saving someone’s life—the years of experience, training, medical school, and residency—all of it comes to bear on that hospitalized patient. And it can all be unraveled at the time of discharge if it’s not handled properly,” says Arpana Vidyarthi, MD, a hospitalist and director of quality at the University of California at San Francisco.
Dr. Vidyarthi views in-hospital and discharge transitions as integrally related. “The analysis is similar, even if different techniques may be needed,” she says, adding that, fundamentally, it involves having a system that allows people—or forces them—to do the “right thing.”
That’s why achieving effective care transitions will require more than just a standardized tool or process, Dr. Vidyarthi says. “This is about understanding the ways people communicate and finding ways to train them to communicate better,” she says. “The problem we have is not a lack of information, but how to communicate what, to whom, and when.”
What’s really needed, Dr. Vidyarthi says, is a hospital’s commitment to more effective transitions and its hospitalists’ leadership in driving a comprehensive, multidisciplinary, team- and evidence-based QI process. The new process should be a QI-based solution to a hospital’s care-transitions issues. “Before you can standardize your process, you need to understand it,” she says. “This is a complex problem, and it needs a multifaceted solution. But this lies squarely within the hospitalist arena. We’re part of everything that happens in the hospital.

—Anuj Dalal, MD, Brigham and Women’s Hospital, Boston
Hospital administrators are looking to HM to solve transition and readmission problems now, says Tina Budnitz, MPH, BOOST Project Director (Better Outcomes for Older Adults through Safe Transitions). She expects the scrutiny from the C-suite, legislators, and watchdog groups to increase as the spotlight continues to shine on the healthcare system.
“Any hospitalist can act as a leader in their institution,” Budnitz says. “Be a change agent, pull a group together, and start asking questions: Do we have safe care-transitions practices and processes in place? Just by asking the right question, you can be a catalyst for the system.”
Budnitz also emphasizes the importance of teamwork in the hospital setting. “How can I help my teammates? What am I communicating to the nurses on rounds?” she says. “Can you initiate dialogue with your outpatient medical groups: ‘These faxes we’re sending you—is that information getting to you in ways and times that are helpful? And, by the way, when your patient is admitted, this information would really help me.’ ”
Innovative Strategies
One of the most important initiatives responding to concerns about care transitions is Project BOOST (www.hos pitalmedicine.org/BOOST), a comprehensive toolkit for improving a hospital’s transitions of care. The project aims to build a national consensus for best practices in transitions; collaborate with representatives from the Agency for Healthcare Research and Quality (AHRQ), the Centers for Medicare and Medicaid Services (CMS), and the Joint Commission; and develop a national resource library, Budnitz says.
“Project BOOST not only puts forth best practices for admitting patients, planning for discharge, and then doing the discharge, it also helps show facilities how to change their systems, with resources and tools for analyzing and re-engineering the system,” she says. “Sites get one-to-one assistance from a mentor.”
Six hospitals signed on to the pilot program in 2008; 24 more joined last year. In January, SHM announced a collaborative with the University of Michigan and Blue Cross Blue Shield of Michigan for 15 Michigan hospitals to receive training and mentorship starting in May. And last month, SHM and the California HealthCare Foundation announced a Project BOOST initiative for 20 of the health system’s hospitals (see “California Dreamin’”, p. 6). Other free resources offered on the BOOST Web portal include clinical, data collection, and project management tools. SHM also has a DVD that explains how to use the “teachback” method to improve communication with patients.
Jennifer Myers, MD, FHM, assistant professor of clinical medicine and patient-safety officer at the Hospital of the University of Pennsylvania in Philadelphia, is a Project BOOST participant who spearheaded a process change to improve the quality of her facility’s discharge summary, along with accompanying resident education.7 The discharge summary recently was integrated with the hospital’s electronic health record (EHR) system.
“We’ve gone from dictating the discharge summary to an electronic version completed by the hospitalist, with prompts for key components of the summary, which allows us to create summaries more efficiently—ideally on the day of discharge, but usually within 48 hours,” Dr. Myers says. “We previously researched whether teaching made a difference in the quality of discharges; we found that it did. So we look forward to standardizing our teaching approach around this important topic for all residents.”
Another care-transitions innovation receiving a lot of attention from the government and the private sector is Project RED (Re-Engineered Discharge), led by Brian Jack, MD, vice chair of the department of family medicine at Boston Medical Center. The Project RED research group develops and tests strategies to improve the hospital discharge process to promote patient safety and reduce rehospitalization rates.
“We used re-engineering tools borrowed from other fields, brought together experts from all over the hospital, divided up the whole discharge process, and identified key principles,” Dr. Jack explains. The resulting discharge strategy is reflected in an 11-item checklist of discrete, mutually reinforcing components, which have been shown to reduce rehospitalization rates by 32% while raising patient satisfaction.8 It includes comprehensive discharge and after-hospital plans, a nurse discharge advocate, and a medication reconciliation phone call to the patient. A virtual “patient advocate,” a computerized avatar named Louise, is now being tested. If successful, it will allow patients to interact with a touch-screen teacher of the after-care plan who has time to work at the patient’s pace.
Technology and Transitions
Informatics can be a key player in facilitating care transitions, says Anuj Dalal, MD, a hospitalist and instructor in medicine at Brigham and Women’s Hospital in Boston. He is using one of his hospital’s technological strengths—a well-established, firewall-protected e-mail system—to help improve the discharge process.
“We decided to try to improve awareness of test results pending at the time of discharge,” Dr. Dalal explains. “We created an intervention that automatically triggers an e-mail with the finalized test results to the responsible providers. The intervention creates a loop of communication between the inpatient attending and the PCP. What we hope to show in our research over the next year or two is whether the intervention actually increases awareness of test results by providers.”
One thing to remember is that “all kinds of things can go wrong with care transitions,” no matter the size of the institution, the experience of the staff, or technological limitations, says Vineet Chopra, MD, FACP, a hospitalist at the University of Michigan Health System in Ann Arbor. “The problems of transitions vary from place to place, day to day, time of day, shift changes; and let’s not forget physician extenders and the other members of the healthcare team,” he says. “The more complicated the team, the more complicated the information needing to be handed off becomes.”
Before he joined the group at the university, Dr. Chopra worked at a community hospital, St. Joseph’s Mercy Hospital in Hot Springs, Ark. “It’s hard to come up with a one-size-fits-all solution when there are so many variables,” he says. At the community hospital, “we mandated that the hospitalist call the PCP at the time of discharge. At the academic medical center, we share an EHR with the PCPs and can reach them electronically. We are required to have the discharge summary in the computer before the patient leaves the hospital, and we mandate that hospitalists are reachable by e-mail or phone when they are off.
“I’m not a believer in throwing more technology at problems and just adding more layers of information tools,” Dr. Chopra adds. “Hospitalists who used to carry stethoscopes now also have a clipboard, phone, pager, PDA, and nine different signouts in their pockets. What we want to do is make their life easier. Here, we are looking at technology as a means to do that.”
Dr. Chopra and hospitalist colleague Prasanth Gosineni, MD, have been working with an Ann Arbor tech company called Synaptin to develop a lightweight, mobile client application designed to work on smartphones. Still in pilot testing, it would allow for task-oriented and priority-based messaging in real time and the systematic transfer of important information for the next hospitalist shift.
“You need to be able to share information in a systematic way, but that’s only half of the answer. The other half is the ability to ask specific questions,” Dr. Chopra says. “Technology doesn’t take away from the face-to-face encounter that needs to happen. Nothing will replace face time, but part of the solution is to provide data efficiently and in a way that is easily accessible.”
Dr. Chopra admits that EHR presents both positives and negatives to improved transitions and patient care, “depending on how well it works and what smart features it offers,” he says, “but also recognizing that EHR and other technologies have also taken us farther away from face-to-face exchanges. Some would say that’s part of the problem.”
Handoffs, discharges, and other transitions are ubiquitous in HM—and fraught with the potential for costly and harmful errors. The ideal of an interactive, face-to-face handoff simply is not available for many care transitions. However, hospitalists are challenged to find solutions that will work in their hospitals, with their teams, and their types of patients. Patients and policymakers expect nothing less. TH
Larry Beresford is a freelance writer based in Oakland, Calif.
References
- Payne C, Stein J, Dressler D. Implementation of a structured electronic tool to improve patient handoffs and resident satisfaction. Poster abstract: Internal Medicine 2010, April 21-24, 2010, Toronto.
- Vidyarthi AR. Triple Handoff. AHRQ WebM&M website. Available at: webmm.ahrq.gov/case.aspx? caseID=134. Published May 2006. Accessed May 29, 2010.
- Snow V, Beck D, Budnitz T, et al. Transitions of Care Consensus Policy Statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, and Society for Academic Emergency Medicine. J Hosp Med. 2009;4(6):364-370.
- 2006 National Patient Safety Goals. The Joint Commission website. Available at: www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/06_npsgs.htm. Accessed June 8, 2010.
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009; 2:360:1418-1428.
- Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW. The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161-167.
- Myers JS, Jaipaul CK, Kogan JR, Krekun S, Bellini LM, Shea JA. Are discharge summaries teachable? The effects of a discharge summary curriculum on the quality of discharge summaries in an internal medicine residency program. Acad Med. 2006; 81(10):S5-S8.
- Jack BW, Chetty VK, Anthony D, et al. A reengineered hospital discharge program to decrease rehospitalization: a randomized trial. Ann Intern Med. 2009;150(3):178-187.
- Arora VM, Manjarrez E, Dressler DD, Basaviah P, Halasyamani L, Kripalani S. Hospitalist handoffs: a systematic review and task force recommendations. J Hosp Med. 2009;4(7): 433-440.
- Halasyamani L, Kripalani S, Coleman E, et al. Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists. J Hosp Med. 2006;1(6):354-360.
- Schnipper JL, Kirwin JL, Cotugno MC, et al. Role of pharmacist counseling in preventing adverse drug events after hospitalization. Arch Int Med. 2006;166(5):565-571.
- Dudas V, Bookwalter T, Kerr KM, Pantilat SZ. The impact of follow-up telephone calls to patients after hospitalization. Am J Med. 2001;111(9B): 26S-30S.
Christina Payne, MD, is a third-year resident at Emory University Hospital in Atlanta who will begin her first hospitalist job, with Emory in September. In spite of her dearth of practical experience, she already has experience researching one of the most vexing problems confronting HM: how to improve transitions of care.
Dr. Payne has been studying the benefits of a structured electronic tool that generates a standardized sign-out list of a hospital team’s full census at the time of shift change, compared with the usual, highly variable sign-out practices of medical residents. At a poster presentation at Internal Medicine 2010 in April in Toronto, Dr. Payne and colleagues reported that residents using the tool were twice as confident at performing handoffs, had lower rates of perceived near-miss events, and were happier.1
“Hospitalists everywhere are starting to realize the importance of trying to reduce opportunities for human error that occur during care transitions,” Dr. Payne says. “The biggest thing I learned from this research is the importance of standardizing the handoff process [with information communicated consistently].
“It is essential to keep communication lines open,” Dr. Payne adds. “No tool can replace the importance of communication between doctors and the need to sit down and talk. The ideal signout happens in a quiet room where the two of you can talk about active patients and achieve rapport. But, realistically, how often does that happen?”
Standardization is one of a handful of strategies hospitalists, researchers, and policymakers are using to tackle transitions—both in-hospital handoffs and post-discharge transitions—with outpatient care. Some hospitalists are using practice simulations and training strategies; others have implemented medication reconciliation checks at every discharge, checklists and other communication strategies, team-based quality-improvement (QI) initiatives, and new technologies to enhance and streamline communication. Some interventions follow the patient from the hospital to the community physician with a phone call, follow-up clinic, or other contact; others aim to empower the patient to be a better self-advocate. But for hospitalists, the challenge is to communicate the right amount of transfer information to the right receiver at the right time.
No matter the technique, the goal is the same: Improve the handoff and discharge process in a way that promotes efficiency and patient safety. And hospitalists are at the forefront of the changing landscape of care transitions.
Under the Microscope
Care transitions of all kinds are under the magnifying glass of national healthcare reform, with growing recognition of the need to make care safer and reduce the preventable, costly hospital readmissions caused by incomplete handoffs. Care transitions for hospitalists include internal handoffs, both at daily shift changes and at service changes when an outgoing provider is leaving after a period of consecutive daily shifts. These typically involve a sign-out process and face-to-face encounter, with some kind of written backup. One teaching institution reported that such handoffs take place 4,000 times per day in the hospital, or 1.6 million times per year.2

—Arpana Vidyarthi, MD, University of California at San Francisco
Geographical transitions can be from one floor or department to another, or out the hospital door to another facility or home. Transitions typically involve a discharge process and a written discharge summary. Care transitions also include hospital admissions, which put the hospitalist in the role of handoff receiver rather than initiator, plus a variety of other transitions involving nurses, physician extenders, and other practitioners.
Each transition is a major decision point in the course of a patient’s hospitalization; each transition also presents a time of heightened vulnerability (e.g., potential communication breakdowns, medication errors, patient anxiety or confusion, etc.). In fact, according to a Transitions of Care Consensus Policy Statement published in 2009 by SHM and five other medical societies, handoffs are ubiquitous in HM, with significant patient safety and quality deficiencies in handoffs existing in the current system.3
Poor communication at the time of handoff has been implicated in near-misses and adverse events in a variety of healthcare contexts, including 70% of hospital sentinel events studied by The Joint Commission, which named standardized handoffs (with an opportunity for interactive communication) as a National Patient Safety Goal in 2006.4 The federal government is studying care transitions, supporting demonstration projects for Medicare enrollees, and including readmission rates in national hospital report card data.
“Transitions of care and handoffs are a huge focus right now because of the increased fragmentation of care in the United States. Hospitalists are in charge of a greater percentage of hospitalized patients, which means more coordination of care is needed,” says Vineet Arora, MD, MA, FHM, assistant professor of medicine and associate director of the internal-medicine residency at the University of Chicago, and chair of the SHM task force on handoffs.
Inadequate communication and poor care transitions can undermine hospitalists’ best care-planning efforts, erode patients’ and families’ confidence and satisfaction with hospital care, and leave primary-care physicians (PCPs) feeling unsatisfied with the relationship. As many as 1 in 5 Medicare beneficiary hospitalizations result in a readmission within 30 days, and while not all of these are preventable, far too many are.5 Another prospective cohort study found that 1 in 5 patients discharged from the hospital to the home experienced an adverse event within three weeks of discharge.6 Complex comorbidities, advanced age, unknown PCP, and limited healthcare literacy present hospitalists with extremely difficult transitions.
Patient safety and cost control are the linchpins to national efforts to improve transitions of care. Dr. Arora recently coauthored an original research paper, which will be published in the Journal of Hospital Medicine in September, showing older hospitalized patients are twice as likely to report problems after discharge if their PCPs were not aware they were hospitalized.
“With escalating healthcare costs, people are looking at ways to save money and reduce redundant care,” Dr. Arora explains, pointing out, as an example, repeated tests resulting from inadequate communication between healthcare providers.
The System Must Change
“All of the effort we put into saving someone’s life—the years of experience, training, medical school, and residency—all of it comes to bear on that hospitalized patient. And it can all be unraveled at the time of discharge if it’s not handled properly,” says Arpana Vidyarthi, MD, a hospitalist and director of quality at the University of California at San Francisco.
Dr. Vidyarthi views in-hospital and discharge transitions as integrally related. “The analysis is similar, even if different techniques may be needed,” she says, adding that, fundamentally, it involves having a system that allows people—or forces them—to do the “right thing.”
That’s why achieving effective care transitions will require more than just a standardized tool or process, Dr. Vidyarthi says. “This is about understanding the ways people communicate and finding ways to train them to communicate better,” she says. “The problem we have is not a lack of information, but how to communicate what, to whom, and when.”
What’s really needed, Dr. Vidyarthi says, is a hospital’s commitment to more effective transitions and its hospitalists’ leadership in driving a comprehensive, multidisciplinary, team- and evidence-based QI process. The new process should be a QI-based solution to a hospital’s care-transitions issues. “Before you can standardize your process, you need to understand it,” she says. “This is a complex problem, and it needs a multifaceted solution. But this lies squarely within the hospitalist arena. We’re part of everything that happens in the hospital.

—Anuj Dalal, MD, Brigham and Women’s Hospital, Boston
Hospital administrators are looking to HM to solve transition and readmission problems now, says Tina Budnitz, MPH, BOOST Project Director (Better Outcomes for Older Adults through Safe Transitions). She expects the scrutiny from the C-suite, legislators, and watchdog groups to increase as the spotlight continues to shine on the healthcare system.
“Any hospitalist can act as a leader in their institution,” Budnitz says. “Be a change agent, pull a group together, and start asking questions: Do we have safe care-transitions practices and processes in place? Just by asking the right question, you can be a catalyst for the system.”
Budnitz also emphasizes the importance of teamwork in the hospital setting. “How can I help my teammates? What am I communicating to the nurses on rounds?” she says. “Can you initiate dialogue with your outpatient medical groups: ‘These faxes we’re sending you—is that information getting to you in ways and times that are helpful? And, by the way, when your patient is admitted, this information would really help me.’ ”
Innovative Strategies
One of the most important initiatives responding to concerns about care transitions is Project BOOST (www.hos pitalmedicine.org/BOOST), a comprehensive toolkit for improving a hospital’s transitions of care. The project aims to build a national consensus for best practices in transitions; collaborate with representatives from the Agency for Healthcare Research and Quality (AHRQ), the Centers for Medicare and Medicaid Services (CMS), and the Joint Commission; and develop a national resource library, Budnitz says.
“Project BOOST not only puts forth best practices for admitting patients, planning for discharge, and then doing the discharge, it also helps show facilities how to change their systems, with resources and tools for analyzing and re-engineering the system,” she says. “Sites get one-to-one assistance from a mentor.”
Six hospitals signed on to the pilot program in 2008; 24 more joined last year. In January, SHM announced a collaborative with the University of Michigan and Blue Cross Blue Shield of Michigan for 15 Michigan hospitals to receive training and mentorship starting in May. And last month, SHM and the California HealthCare Foundation announced a Project BOOST initiative for 20 of the health system’s hospitals (see “California Dreamin’”, p. 6). Other free resources offered on the BOOST Web portal include clinical, data collection, and project management tools. SHM also has a DVD that explains how to use the “teachback” method to improve communication with patients.
Jennifer Myers, MD, FHM, assistant professor of clinical medicine and patient-safety officer at the Hospital of the University of Pennsylvania in Philadelphia, is a Project BOOST participant who spearheaded a process change to improve the quality of her facility’s discharge summary, along with accompanying resident education.7 The discharge summary recently was integrated with the hospital’s electronic health record (EHR) system.
“We’ve gone from dictating the discharge summary to an electronic version completed by the hospitalist, with prompts for key components of the summary, which allows us to create summaries more efficiently—ideally on the day of discharge, but usually within 48 hours,” Dr. Myers says. “We previously researched whether teaching made a difference in the quality of discharges; we found that it did. So we look forward to standardizing our teaching approach around this important topic for all residents.”
Another care-transitions innovation receiving a lot of attention from the government and the private sector is Project RED (Re-Engineered Discharge), led by Brian Jack, MD, vice chair of the department of family medicine at Boston Medical Center. The Project RED research group develops and tests strategies to improve the hospital discharge process to promote patient safety and reduce rehospitalization rates.
“We used re-engineering tools borrowed from other fields, brought together experts from all over the hospital, divided up the whole discharge process, and identified key principles,” Dr. Jack explains. The resulting discharge strategy is reflected in an 11-item checklist of discrete, mutually reinforcing components, which have been shown to reduce rehospitalization rates by 32% while raising patient satisfaction.8 It includes comprehensive discharge and after-hospital plans, a nurse discharge advocate, and a medication reconciliation phone call to the patient. A virtual “patient advocate,” a computerized avatar named Louise, is now being tested. If successful, it will allow patients to interact with a touch-screen teacher of the after-care plan who has time to work at the patient’s pace.
Technology and Transitions
Informatics can be a key player in facilitating care transitions, says Anuj Dalal, MD, a hospitalist and instructor in medicine at Brigham and Women’s Hospital in Boston. He is using one of his hospital’s technological strengths—a well-established, firewall-protected e-mail system—to help improve the discharge process.
“We decided to try to improve awareness of test results pending at the time of discharge,” Dr. Dalal explains. “We created an intervention that automatically triggers an e-mail with the finalized test results to the responsible providers. The intervention creates a loop of communication between the inpatient attending and the PCP. What we hope to show in our research over the next year or two is whether the intervention actually increases awareness of test results by providers.”
One thing to remember is that “all kinds of things can go wrong with care transitions,” no matter the size of the institution, the experience of the staff, or technological limitations, says Vineet Chopra, MD, FACP, a hospitalist at the University of Michigan Health System in Ann Arbor. “The problems of transitions vary from place to place, day to day, time of day, shift changes; and let’s not forget physician extenders and the other members of the healthcare team,” he says. “The more complicated the team, the more complicated the information needing to be handed off becomes.”
Before he joined the group at the university, Dr. Chopra worked at a community hospital, St. Joseph’s Mercy Hospital in Hot Springs, Ark. “It’s hard to come up with a one-size-fits-all solution when there are so many variables,” he says. At the community hospital, “we mandated that the hospitalist call the PCP at the time of discharge. At the academic medical center, we share an EHR with the PCPs and can reach them electronically. We are required to have the discharge summary in the computer before the patient leaves the hospital, and we mandate that hospitalists are reachable by e-mail or phone when they are off.
“I’m not a believer in throwing more technology at problems and just adding more layers of information tools,” Dr. Chopra adds. “Hospitalists who used to carry stethoscopes now also have a clipboard, phone, pager, PDA, and nine different signouts in their pockets. What we want to do is make their life easier. Here, we are looking at technology as a means to do that.”
Dr. Chopra and hospitalist colleague Prasanth Gosineni, MD, have been working with an Ann Arbor tech company called Synaptin to develop a lightweight, mobile client application designed to work on smartphones. Still in pilot testing, it would allow for task-oriented and priority-based messaging in real time and the systematic transfer of important information for the next hospitalist shift.
“You need to be able to share information in a systematic way, but that’s only half of the answer. The other half is the ability to ask specific questions,” Dr. Chopra says. “Technology doesn’t take away from the face-to-face encounter that needs to happen. Nothing will replace face time, but part of the solution is to provide data efficiently and in a way that is easily accessible.”
Dr. Chopra admits that EHR presents both positives and negatives to improved transitions and patient care, “depending on how well it works and what smart features it offers,” he says, “but also recognizing that EHR and other technologies have also taken us farther away from face-to-face exchanges. Some would say that’s part of the problem.”
Handoffs, discharges, and other transitions are ubiquitous in HM—and fraught with the potential for costly and harmful errors. The ideal of an interactive, face-to-face handoff simply is not available for many care transitions. However, hospitalists are challenged to find solutions that will work in their hospitals, with their teams, and their types of patients. Patients and policymakers expect nothing less. TH
Larry Beresford is a freelance writer based in Oakland, Calif.
References
- Payne C, Stein J, Dressler D. Implementation of a structured electronic tool to improve patient handoffs and resident satisfaction. Poster abstract: Internal Medicine 2010, April 21-24, 2010, Toronto.
- Vidyarthi AR. Triple Handoff. AHRQ WebM&M website. Available at: webmm.ahrq.gov/case.aspx? caseID=134. Published May 2006. Accessed May 29, 2010.
- Snow V, Beck D, Budnitz T, et al. Transitions of Care Consensus Policy Statement: American College of Physicians, Society of General Internal Medicine, Society of Hospital Medicine, American Geriatrics Society, American College of Emergency Physicians, and Society for Academic Emergency Medicine. J Hosp Med. 2009;4(6):364-370.
- 2006 National Patient Safety Goals. The Joint Commission website. Available at: www.jointcommission.org/PatientSafety/NationalPatientSafetyGoals/06_npsgs.htm. Accessed June 8, 2010.
- Jencks SF, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009; 2:360:1418-1428.
- Forster AJ, Murff HJ, Peterson JF, Gandhi TK, Bates DW. The incidence and severity of adverse events affecting patients after discharge from the hospital. Ann Intern Med. 2003;138(3):161-167.
- Myers JS, Jaipaul CK, Kogan JR, Krekun S, Bellini LM, Shea JA. Are discharge summaries teachable? The effects of a discharge summary curriculum on the quality of discharge summaries in an internal medicine residency program. Acad Med. 2006; 81(10):S5-S8.
- Jack BW, Chetty VK, Anthony D, et al. A reengineered hospital discharge program to decrease rehospitalization: a randomized trial. Ann Intern Med. 2009;150(3):178-187.
- Arora VM, Manjarrez E, Dressler DD, Basaviah P, Halasyamani L, Kripalani S. Hospitalist handoffs: a systematic review and task force recommendations. J Hosp Med. 2009;4(7): 433-440.
- Halasyamani L, Kripalani S, Coleman E, et al. Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists. J Hosp Med. 2006;1(6):354-360.
- Schnipper JL, Kirwin JL, Cotugno MC, et al. Role of pharmacist counseling in preventing adverse drug events after hospitalization. Arch Int Med. 2006;166(5):565-571.
- Dudas V, Bookwalter T, Kerr KM, Pantilat SZ. The impact of follow-up telephone calls to patients after hospitalization. Am J Med. 2001;111(9B): 26S-30S.
Market Watch
New Drugs, Indications, Dosage Forms, and Approval Recommendations
- Acetaminophen intravenous (Ofirmev) received a complete response letter in February from the FDA related to facility manufacturing deficiencies. The FDA did not cite any safety or efficacy issues and is not requiring any additional studies to be done prior to approval.1 The third-party manufacturer has submitted its response to the FDA and is ready to resubmit their new drug application (NDA) for this agent. It is being investigated to treat fever and pain in adults and children.2
- Ciprofloxacin dry powder inhaler (DPI) has received orphan drug status from the FDA for treating pulmonary infections in cystic fibrosis (CF) patients.3,4 It is in clinical trials to determine if it can improve pulmonary function in CF patients with Pseudomonas aeruginosa infections.
- Carglumic acid (Carbaglu) has been approved by the FDA to treat the metabolic disorder N-acetylglutamate synthetase (NAGS) deficiency.5 NAGS deficiency is an extremely rare genetic disorder that presents shortly after birth. It results in hyperammonemia, and can be fatal if not rapidly detected and managed. Carglumic acid treats the hyperammonemia within three days, with a lowering of the ammonia level within 24 hours. In clinical trials, a small number of patients (n=23) received the drug from six months to 21 years; the majority of patients were able to maintain normal ammonia levels long-term with continued treatment. It is recommended that carglumic acid only be administered by physicians who have experience dealing with metabolic disorders. The starting dose is between 100 mg/kg/day and 250 mg/kg/day for treatment of acute hyperammonemia. Using other agents to lower the ammonia level during acute episodes is recommended. Dosing should be based on the ammonia level and the patient’s symptoms.
- CK-2017357 has received orphan drug status for treating amyotrophic lateral sclerosis (ALS), or Lou Gehrig’s disease.6
- Desirudin injection (Iprivask), a direct thrombin inhibitor, has been approved by the FDA for the prevention of DVT.7 In clinical trials, it was superior to enoxaparin and unfractionated heparin for preventing proximal DVT and prevention of major venous thromboembolic events following elective hip replacement surgery. Desirudin is administered as a fixed subcutaneous dose. It does not cause thrombocytopenia, is relatively short-acting, and is easy to monitor. Some of the adverse reactions in clinical trials were thrombosis, hypotension, lower-extremity edema, fever, decreased hemoglobin level, and hematuria.8 Also known as Revasc, this medication has been available in Europe for more than 10 years.
- Doxepin tablets (Silenor) have been approved by the FDA for the treatment of short-term and chronic insomnia distinguished by difficulty with sleep maintenance in adults and elderly patients.9 Sleep maintenance includes difficulty staying asleep, waking up too much or too early, and not being able to fall back asleep. In clinical trials, adverse reactions were similar to placebo, there was a low-therapy discontinuation rate, and no evidence of amnesia, tolerance, or complex sleep behaviors such as sleep eating or sleep driving.10 It will be available in 3-mg and 6-mg tablets. It is not designated as a controlled substance.
- GVAX pancreas vaccine has received orphan drug status as a potential treatment for pancreatic cancer.11 It also is being investigated for other cancers, including those of the breast and for leukemias.
- Ritonavir (Norvir) has been approved by the FDA in a new formulation, which is heat-stable and can be stored at room temperature rather than in the refrigerator.12 The rate of drug absorption with the new formulation is different but does not require a dosage change.
- Somatropin [rDNA origin] prefilled injection pen (Norditropin FlexPro) has been approved by the FDA to treat adults and children with growth hormone disorders.13 The pen has an audible click and does not require any reconstitution or cartridge loading. After initial use, the pen can be left at room temperature for up to three weeks without worry of drug degradation.
Pipeline
- Agalsidase (Replagal) has received fast-track status from the FDA for treating Fabry disease.14 It is an enzyme replacement therapy.
- Exenatide LAR (Bydureon), the once-weekly version of exenatide (Byetta), is in final discussions at the FDA. The FDA has asked for additional information related to the product label, risk mitigation, and manufacturing, which the manufacturer is addressing.15
- Insulin powder for inhalation, ultra-rapid-acting (Afrezza), has been reviewed by the FDA, and the agency has requested additional information related to safety and labeling.16
- A combination therapy of saxagliptin/metformin has been submitted to the FDA as a once-daily treatment of Type 2 diabetes mellitus as an adjunct to diet in adults who cannot adequately control their diabetes on metformin monotherapy, or in treatment-naïve patients.17
- Vilanterol/fluticasone is a combination of the inhaled corticosteroid fluticasone and the long-acting beta-agonist (LABA) vilanterol, which is currently in Phase 3 clinical trials for treating asthma.18 The trial will compare the combination’s efficacy and safety to fluticasone/salmeterol (Advair). TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Cadence pharmaceuticals receives complete response letter from FDA for intravenous acetaminophen NDA. Cadence Pharmaceuticals website. Available at: http://cadx.client.shareholder.com/releasedetail.cfm?ReleaseID=444303. Accessed March 23, 2010.
- Cadence pharmaceuticals reports fourth quarter and full year 2009 financial results. Cadence Pharmaceuticals website. Available at: http://files.shareholder.com/downloads/CADX/874963043x0x359109/1cf00f72-0872-4d4e-b27d-bde1c03d625a/CADX_News_2010_3_15_General_Releases.pdf. Accessed March 23, 2010.
- Bayer lung infection drug gets orphan status. Forbes website. Available at: http://www.forbes.com/feeds/ap/2010/03/11/business-energy-us-bayer-healthcare-orphan-drug_7427368.html. Accessed March 23, 2010.
- FDA grants orphan status for Bayer’s ciprofloxacin for cystic fibrosis. The Pharma Letter website. Available at: http://www.thepharmaletter.com/file/4e4fb33313eddf122cdb730e3ea71840/fda-grants-orphan-status-for-bayers-ciprofloxacin-for-cystic-fibrosis.html. Accessed March 23, 2010.
- Burgess S. FDA approves drug to treat condition that causes elevated ammonia levels. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm205150.htm. Accessed March 23, 2010.
- UPDATE 1: Cytokinetics’ Lou Gehrig’s drug gets orphan status. Reuters website. Available at: http://www.reuters.com/assets/print?aid=USSGE6290J820100310. Accessed March 23, 2010.
- Iprivask available for prevention of deep vein thrombosis. Monthly Prescribing Reference website. Available at: http://www.empr.com/iprivask-available-for-prevention-of-deep-vein-thrombosis/article/164779/. Accessed March 23, 2010.
- First direct thrombin inhibitor for DVT prevention now available from Canyon Pharmaceuticals. Canyon Pharmaceuticals website. Available at: http://www.canyonpharma.com/newsexpand.aspx?id=7. Accessed March 23, 2010.
- FDA approves Silenor. Drugs.com website. Available at: http://www.drugs.com/newdrugs/somaxon-announces-fda-approval-silenor-doxepin-insomnia-2070.html?printable=1. Accessed March 23, 2010.
- UPDATE 1: Somaxon gets FDA nod for insomnia drug, shares soar. Reuters website. Available at: http://www.reuters.com/assets/print?aid=USSGE62H0KI20100318. Accessed March 21, 2010.
- BioSante announces FDA orphan drug designation for GVAX pancreatic cancer vaccine. The New York Times website. Available at: http://markets.on.nytimes.com/research/stocks/news/press_release.asp?docTag=201003150755BIZWIRE_USPRX____BW5298&feedID=600&press_symbol=64917. Accessed March 23, 2010.
- Abbott receives U.S. FDA approval for heat-stable Norvir (ritonavir) tablets. Abbott website. Available at: http://www.abbott.com/global/url/pressRelease/en_US/60.5:5/Press_Release_0820.htm. Accessed March 23, 2010.
- Novo Nordisk receives FDA approval for Norditropin FlexPro for growth hormone treatment. Novo Nordisk website. Available at: http://press.novonordisk-us.com/index.php?s=43&item=239. Accessed March 23, 2010.
- Dane L. Shire receives FDA fast-track designation for Replagal. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=97AF48EBA7054EF0857F055877E82B2C&logRowId=352323. Accessed March 23, 2010.
- Amylin shares up on FDA response for diabetes drug. Forbes website. Available at: http://www.forbes.com/feeds/ap/2010/03/15/business-health-care-us-amylin-mover_7435579.html. Accessed March 23, 2010.
- Russell J. MannKind drug fails to win FDA approval. Los Angeles Business Journal website. Available at: http://labusinessjournal.com/article.asp?aid=4998235.0597195.1894204.8500584.7421642.713. Accessed March 23, 2010.
- U.S. Food and Drug Administration accepts NDA for once-daily fixed dose combination of Onglyza (saxagliptin) and extended-release metformin for the treatment of type 2 diabetes mellitus in adults. AstraZeneca website. Available at: http://www.astrazeneca-us.com/about-astrazeneca-us/newsroom/product/8804120?itemId=8804120#. Accessed March 23, 2010.
- Dennis M. GlaxoSmithKline begins late-stage clinical programme for asthma drug Relovair. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E256469FBD8F4A2F80C5DD3E844CC1E1&logRowId=356423. Accessed March 23, 2010.
New Drugs, Indications, Dosage Forms, and Approval Recommendations
- Acetaminophen intravenous (Ofirmev) received a complete response letter in February from the FDA related to facility manufacturing deficiencies. The FDA did not cite any safety or efficacy issues and is not requiring any additional studies to be done prior to approval.1 The third-party manufacturer has submitted its response to the FDA and is ready to resubmit their new drug application (NDA) for this agent. It is being investigated to treat fever and pain in adults and children.2
- Ciprofloxacin dry powder inhaler (DPI) has received orphan drug status from the FDA for treating pulmonary infections in cystic fibrosis (CF) patients.3,4 It is in clinical trials to determine if it can improve pulmonary function in CF patients with Pseudomonas aeruginosa infections.
- Carglumic acid (Carbaglu) has been approved by the FDA to treat the metabolic disorder N-acetylglutamate synthetase (NAGS) deficiency.5 NAGS deficiency is an extremely rare genetic disorder that presents shortly after birth. It results in hyperammonemia, and can be fatal if not rapidly detected and managed. Carglumic acid treats the hyperammonemia within three days, with a lowering of the ammonia level within 24 hours. In clinical trials, a small number of patients (n=23) received the drug from six months to 21 years; the majority of patients were able to maintain normal ammonia levels long-term with continued treatment. It is recommended that carglumic acid only be administered by physicians who have experience dealing with metabolic disorders. The starting dose is between 100 mg/kg/day and 250 mg/kg/day for treatment of acute hyperammonemia. Using other agents to lower the ammonia level during acute episodes is recommended. Dosing should be based on the ammonia level and the patient’s symptoms.
- CK-2017357 has received orphan drug status for treating amyotrophic lateral sclerosis (ALS), or Lou Gehrig’s disease.6
- Desirudin injection (Iprivask), a direct thrombin inhibitor, has been approved by the FDA for the prevention of DVT.7 In clinical trials, it was superior to enoxaparin and unfractionated heparin for preventing proximal DVT and prevention of major venous thromboembolic events following elective hip replacement surgery. Desirudin is administered as a fixed subcutaneous dose. It does not cause thrombocytopenia, is relatively short-acting, and is easy to monitor. Some of the adverse reactions in clinical trials were thrombosis, hypotension, lower-extremity edema, fever, decreased hemoglobin level, and hematuria.8 Also known as Revasc, this medication has been available in Europe for more than 10 years.
- Doxepin tablets (Silenor) have been approved by the FDA for the treatment of short-term and chronic insomnia distinguished by difficulty with sleep maintenance in adults and elderly patients.9 Sleep maintenance includes difficulty staying asleep, waking up too much or too early, and not being able to fall back asleep. In clinical trials, adverse reactions were similar to placebo, there was a low-therapy discontinuation rate, and no evidence of amnesia, tolerance, or complex sleep behaviors such as sleep eating or sleep driving.10 It will be available in 3-mg and 6-mg tablets. It is not designated as a controlled substance.
- GVAX pancreas vaccine has received orphan drug status as a potential treatment for pancreatic cancer.11 It also is being investigated for other cancers, including those of the breast and for leukemias.
- Ritonavir (Norvir) has been approved by the FDA in a new formulation, which is heat-stable and can be stored at room temperature rather than in the refrigerator.12 The rate of drug absorption with the new formulation is different but does not require a dosage change.
- Somatropin [rDNA origin] prefilled injection pen (Norditropin FlexPro) has been approved by the FDA to treat adults and children with growth hormone disorders.13 The pen has an audible click and does not require any reconstitution or cartridge loading. After initial use, the pen can be left at room temperature for up to three weeks without worry of drug degradation.
Pipeline
- Agalsidase (Replagal) has received fast-track status from the FDA for treating Fabry disease.14 It is an enzyme replacement therapy.
- Exenatide LAR (Bydureon), the once-weekly version of exenatide (Byetta), is in final discussions at the FDA. The FDA has asked for additional information related to the product label, risk mitigation, and manufacturing, which the manufacturer is addressing.15
- Insulin powder for inhalation, ultra-rapid-acting (Afrezza), has been reviewed by the FDA, and the agency has requested additional information related to safety and labeling.16
- A combination therapy of saxagliptin/metformin has been submitted to the FDA as a once-daily treatment of Type 2 diabetes mellitus as an adjunct to diet in adults who cannot adequately control their diabetes on metformin monotherapy, or in treatment-naïve patients.17
- Vilanterol/fluticasone is a combination of the inhaled corticosteroid fluticasone and the long-acting beta-agonist (LABA) vilanterol, which is currently in Phase 3 clinical trials for treating asthma.18 The trial will compare the combination’s efficacy and safety to fluticasone/salmeterol (Advair). TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Cadence pharmaceuticals receives complete response letter from FDA for intravenous acetaminophen NDA. Cadence Pharmaceuticals website. Available at: http://cadx.client.shareholder.com/releasedetail.cfm?ReleaseID=444303. Accessed March 23, 2010.
- Cadence pharmaceuticals reports fourth quarter and full year 2009 financial results. Cadence Pharmaceuticals website. Available at: http://files.shareholder.com/downloads/CADX/874963043x0x359109/1cf00f72-0872-4d4e-b27d-bde1c03d625a/CADX_News_2010_3_15_General_Releases.pdf. Accessed March 23, 2010.
- Bayer lung infection drug gets orphan status. Forbes website. Available at: http://www.forbes.com/feeds/ap/2010/03/11/business-energy-us-bayer-healthcare-orphan-drug_7427368.html. Accessed March 23, 2010.
- FDA grants orphan status for Bayer’s ciprofloxacin for cystic fibrosis. The Pharma Letter website. Available at: http://www.thepharmaletter.com/file/4e4fb33313eddf122cdb730e3ea71840/fda-grants-orphan-status-for-bayers-ciprofloxacin-for-cystic-fibrosis.html. Accessed March 23, 2010.
- Burgess S. FDA approves drug to treat condition that causes elevated ammonia levels. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm205150.htm. Accessed March 23, 2010.
- UPDATE 1: Cytokinetics’ Lou Gehrig’s drug gets orphan status. Reuters website. Available at: http://www.reuters.com/assets/print?aid=USSGE6290J820100310. Accessed March 23, 2010.
- Iprivask available for prevention of deep vein thrombosis. Monthly Prescribing Reference website. Available at: http://www.empr.com/iprivask-available-for-prevention-of-deep-vein-thrombosis/article/164779/. Accessed March 23, 2010.
- First direct thrombin inhibitor for DVT prevention now available from Canyon Pharmaceuticals. Canyon Pharmaceuticals website. Available at: http://www.canyonpharma.com/newsexpand.aspx?id=7. Accessed March 23, 2010.
- FDA approves Silenor. Drugs.com website. Available at: http://www.drugs.com/newdrugs/somaxon-announces-fda-approval-silenor-doxepin-insomnia-2070.html?printable=1. Accessed March 23, 2010.
- UPDATE 1: Somaxon gets FDA nod for insomnia drug, shares soar. Reuters website. Available at: http://www.reuters.com/assets/print?aid=USSGE62H0KI20100318. Accessed March 21, 2010.
- BioSante announces FDA orphan drug designation for GVAX pancreatic cancer vaccine. The New York Times website. Available at: http://markets.on.nytimes.com/research/stocks/news/press_release.asp?docTag=201003150755BIZWIRE_USPRX____BW5298&feedID=600&press_symbol=64917. Accessed March 23, 2010.
- Abbott receives U.S. FDA approval for heat-stable Norvir (ritonavir) tablets. Abbott website. Available at: http://www.abbott.com/global/url/pressRelease/en_US/60.5:5/Press_Release_0820.htm. Accessed March 23, 2010.
- Novo Nordisk receives FDA approval for Norditropin FlexPro for growth hormone treatment. Novo Nordisk website. Available at: http://press.novonordisk-us.com/index.php?s=43&item=239. Accessed March 23, 2010.
- Dane L. Shire receives FDA fast-track designation for Replagal. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=97AF48EBA7054EF0857F055877E82B2C&logRowId=352323. Accessed March 23, 2010.
- Amylin shares up on FDA response for diabetes drug. Forbes website. Available at: http://www.forbes.com/feeds/ap/2010/03/15/business-health-care-us-amylin-mover_7435579.html. Accessed March 23, 2010.
- Russell J. MannKind drug fails to win FDA approval. Los Angeles Business Journal website. Available at: http://labusinessjournal.com/article.asp?aid=4998235.0597195.1894204.8500584.7421642.713. Accessed March 23, 2010.
- U.S. Food and Drug Administration accepts NDA for once-daily fixed dose combination of Onglyza (saxagliptin) and extended-release metformin for the treatment of type 2 diabetes mellitus in adults. AstraZeneca website. Available at: http://www.astrazeneca-us.com/about-astrazeneca-us/newsroom/product/8804120?itemId=8804120#. Accessed March 23, 2010.
- Dennis M. GlaxoSmithKline begins late-stage clinical programme for asthma drug Relovair. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E256469FBD8F4A2F80C5DD3E844CC1E1&logRowId=356423. Accessed March 23, 2010.
New Drugs, Indications, Dosage Forms, and Approval Recommendations
- Acetaminophen intravenous (Ofirmev) received a complete response letter in February from the FDA related to facility manufacturing deficiencies. The FDA did not cite any safety or efficacy issues and is not requiring any additional studies to be done prior to approval.1 The third-party manufacturer has submitted its response to the FDA and is ready to resubmit their new drug application (NDA) for this agent. It is being investigated to treat fever and pain in adults and children.2
- Ciprofloxacin dry powder inhaler (DPI) has received orphan drug status from the FDA for treating pulmonary infections in cystic fibrosis (CF) patients.3,4 It is in clinical trials to determine if it can improve pulmonary function in CF patients with Pseudomonas aeruginosa infections.
- Carglumic acid (Carbaglu) has been approved by the FDA to treat the metabolic disorder N-acetylglutamate synthetase (NAGS) deficiency.5 NAGS deficiency is an extremely rare genetic disorder that presents shortly after birth. It results in hyperammonemia, and can be fatal if not rapidly detected and managed. Carglumic acid treats the hyperammonemia within three days, with a lowering of the ammonia level within 24 hours. In clinical trials, a small number of patients (n=23) received the drug from six months to 21 years; the majority of patients were able to maintain normal ammonia levels long-term with continued treatment. It is recommended that carglumic acid only be administered by physicians who have experience dealing with metabolic disorders. The starting dose is between 100 mg/kg/day and 250 mg/kg/day for treatment of acute hyperammonemia. Using other agents to lower the ammonia level during acute episodes is recommended. Dosing should be based on the ammonia level and the patient’s symptoms.
- CK-2017357 has received orphan drug status for treating amyotrophic lateral sclerosis (ALS), or Lou Gehrig’s disease.6
- Desirudin injection (Iprivask), a direct thrombin inhibitor, has been approved by the FDA for the prevention of DVT.7 In clinical trials, it was superior to enoxaparin and unfractionated heparin for preventing proximal DVT and prevention of major venous thromboembolic events following elective hip replacement surgery. Desirudin is administered as a fixed subcutaneous dose. It does not cause thrombocytopenia, is relatively short-acting, and is easy to monitor. Some of the adverse reactions in clinical trials were thrombosis, hypotension, lower-extremity edema, fever, decreased hemoglobin level, and hematuria.8 Also known as Revasc, this medication has been available in Europe for more than 10 years.
- Doxepin tablets (Silenor) have been approved by the FDA for the treatment of short-term and chronic insomnia distinguished by difficulty with sleep maintenance in adults and elderly patients.9 Sleep maintenance includes difficulty staying asleep, waking up too much or too early, and not being able to fall back asleep. In clinical trials, adverse reactions were similar to placebo, there was a low-therapy discontinuation rate, and no evidence of amnesia, tolerance, or complex sleep behaviors such as sleep eating or sleep driving.10 It will be available in 3-mg and 6-mg tablets. It is not designated as a controlled substance.
- GVAX pancreas vaccine has received orphan drug status as a potential treatment for pancreatic cancer.11 It also is being investigated for other cancers, including those of the breast and for leukemias.
- Ritonavir (Norvir) has been approved by the FDA in a new formulation, which is heat-stable and can be stored at room temperature rather than in the refrigerator.12 The rate of drug absorption with the new formulation is different but does not require a dosage change.
- Somatropin [rDNA origin] prefilled injection pen (Norditropin FlexPro) has been approved by the FDA to treat adults and children with growth hormone disorders.13 The pen has an audible click and does not require any reconstitution or cartridge loading. After initial use, the pen can be left at room temperature for up to three weeks without worry of drug degradation.
Pipeline
- Agalsidase (Replagal) has received fast-track status from the FDA for treating Fabry disease.14 It is an enzyme replacement therapy.
- Exenatide LAR (Bydureon), the once-weekly version of exenatide (Byetta), is in final discussions at the FDA. The FDA has asked for additional information related to the product label, risk mitigation, and manufacturing, which the manufacturer is addressing.15
- Insulin powder for inhalation, ultra-rapid-acting (Afrezza), has been reviewed by the FDA, and the agency has requested additional information related to safety and labeling.16
- A combination therapy of saxagliptin/metformin has been submitted to the FDA as a once-daily treatment of Type 2 diabetes mellitus as an adjunct to diet in adults who cannot adequately control their diabetes on metformin monotherapy, or in treatment-naïve patients.17
- Vilanterol/fluticasone is a combination of the inhaled corticosteroid fluticasone and the long-acting beta-agonist (LABA) vilanterol, which is currently in Phase 3 clinical trials for treating asthma.18 The trial will compare the combination’s efficacy and safety to fluticasone/salmeterol (Advair). TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Cadence pharmaceuticals receives complete response letter from FDA for intravenous acetaminophen NDA. Cadence Pharmaceuticals website. Available at: http://cadx.client.shareholder.com/releasedetail.cfm?ReleaseID=444303. Accessed March 23, 2010.
- Cadence pharmaceuticals reports fourth quarter and full year 2009 financial results. Cadence Pharmaceuticals website. Available at: http://files.shareholder.com/downloads/CADX/874963043x0x359109/1cf00f72-0872-4d4e-b27d-bde1c03d625a/CADX_News_2010_3_15_General_Releases.pdf. Accessed March 23, 2010.
- Bayer lung infection drug gets orphan status. Forbes website. Available at: http://www.forbes.com/feeds/ap/2010/03/11/business-energy-us-bayer-healthcare-orphan-drug_7427368.html. Accessed March 23, 2010.
- FDA grants orphan status for Bayer’s ciprofloxacin for cystic fibrosis. The Pharma Letter website. Available at: http://www.thepharmaletter.com/file/4e4fb33313eddf122cdb730e3ea71840/fda-grants-orphan-status-for-bayers-ciprofloxacin-for-cystic-fibrosis.html. Accessed March 23, 2010.
- Burgess S. FDA approves drug to treat condition that causes elevated ammonia levels. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm205150.htm. Accessed March 23, 2010.
- UPDATE 1: Cytokinetics’ Lou Gehrig’s drug gets orphan status. Reuters website. Available at: http://www.reuters.com/assets/print?aid=USSGE6290J820100310. Accessed March 23, 2010.
- Iprivask available for prevention of deep vein thrombosis. Monthly Prescribing Reference website. Available at: http://www.empr.com/iprivask-available-for-prevention-of-deep-vein-thrombosis/article/164779/. Accessed March 23, 2010.
- First direct thrombin inhibitor for DVT prevention now available from Canyon Pharmaceuticals. Canyon Pharmaceuticals website. Available at: http://www.canyonpharma.com/newsexpand.aspx?id=7. Accessed March 23, 2010.
- FDA approves Silenor. Drugs.com website. Available at: http://www.drugs.com/newdrugs/somaxon-announces-fda-approval-silenor-doxepin-insomnia-2070.html?printable=1. Accessed March 23, 2010.
- UPDATE 1: Somaxon gets FDA nod for insomnia drug, shares soar. Reuters website. Available at: http://www.reuters.com/assets/print?aid=USSGE62H0KI20100318. Accessed March 21, 2010.
- BioSante announces FDA orphan drug designation for GVAX pancreatic cancer vaccine. The New York Times website. Available at: http://markets.on.nytimes.com/research/stocks/news/press_release.asp?docTag=201003150755BIZWIRE_USPRX____BW5298&feedID=600&press_symbol=64917. Accessed March 23, 2010.
- Abbott receives U.S. FDA approval for heat-stable Norvir (ritonavir) tablets. Abbott website. Available at: http://www.abbott.com/global/url/pressRelease/en_US/60.5:5/Press_Release_0820.htm. Accessed March 23, 2010.
- Novo Nordisk receives FDA approval for Norditropin FlexPro for growth hormone treatment. Novo Nordisk website. Available at: http://press.novonordisk-us.com/index.php?s=43&item=239. Accessed March 23, 2010.
- Dane L. Shire receives FDA fast-track designation for Replagal. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=97AF48EBA7054EF0857F055877E82B2C&logRowId=352323. Accessed March 23, 2010.
- Amylin shares up on FDA response for diabetes drug. Forbes website. Available at: http://www.forbes.com/feeds/ap/2010/03/15/business-health-care-us-amylin-mover_7435579.html. Accessed March 23, 2010.
- Russell J. MannKind drug fails to win FDA approval. Los Angeles Business Journal website. Available at: http://labusinessjournal.com/article.asp?aid=4998235.0597195.1894204.8500584.7421642.713. Accessed March 23, 2010.
- U.S. Food and Drug Administration accepts NDA for once-daily fixed dose combination of Onglyza (saxagliptin) and extended-release metformin for the treatment of type 2 diabetes mellitus in adults. AstraZeneca website. Available at: http://www.astrazeneca-us.com/about-astrazeneca-us/newsroom/product/8804120?itemId=8804120#. Accessed March 23, 2010.
- Dennis M. GlaxoSmithKline begins late-stage clinical programme for asthma drug Relovair. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E256469FBD8F4A2F80C5DD3E844CC1E1&logRowId=356423. Accessed March 23, 2010.
In the Literature
In This Edition
Literature at a Glance
A guide to this month’s studies
- Effect of restrictive antibiotic policies on dosing timeliness
- Desired consultation format and content
- Risk of cancer associated with CT imaging
- Bleeding, mortality with aspirin after peptic ulcer bleed
- Diagnosis of lung cancer after pneumonia
- Outcomes associated with hyponatremia
- Patient awareness, interest in inpatient medication list
- Monoclonal antibodies in C. difficile
Restrictive Antimicrobial Policy Delays Administration
Clinical question: Does the approval process for restricted on-formulary antimicrobials cause a significant delay in their administration?
Background: Widespread and often unwarranted, antimicrobial use in the hospital lends itself to the development of microbial resistance and increases overall costs. To curb such practices, many hospitals require subspecialty approval prior to dispensing select broad-spectrum antimicrobials. Though shown to improve outcomes, the impact of the approval process on the timeliness of antimicrobial administration remains to be seen.
Study design: Retrospective cohort study.
Setting: Tertiary-care university hospital.
Synopsis: The study included 3,251 inpatients with computerized orders for a “stat” first dose of any of 24 pre-selected, parenteral antimicrobials. Time lag (more than one hour, and more than two hours) to nursing documentation of drug administration was separately analyzed for restricted and unrestricted antimicrobials.
Delay of more than one hour was significantly higher for restricted antimicrobials with an odds ratio of 1.49 (95% CI; 1.23-1.82), while the odds ratio for a delay of more than two hours was 1.78 (95% CI, 1.39-2.21). Also, for restricted antimicrobials, the percentage of orders delayed for more than one hour was significantly different between daytime and nighttime (when the first dose was exempt from pre-approval) orders: 46.1% versus 38.8% (P<0.001). For unrestricted drugs, delay was uniform irrespective of time of day (36.4% of daytime and 36.6% of nighttime orders were delayed more than one hour). The effect of delay in drug administration on patient outcomes was not evaluated.
Though the approval process aims in part to affect resistance patterns and overall costs, this research highlights the need to minimize the delay in administration and probably skip the approval for the first dose in critically ill patients.
Bottom line: Antibiotic approval processes can delay their administration in hospitalized patients, but the effect of this delay on patient outcomes is not yet known.
Citation: Winters BD, Thiemann DR, Brotman DJ. Impact of a restrictive antimicrobial policy on the process and timing of antimicrobial administration. J Hosp Med. 2010;5(1):E41-45.
Physicians Uphold Tenets of Effective Consultation while Highlighting Some Newer Viewpoints
Clinical question: What key features of a consultation are most desirable for physicians?
Background: With new changes in healthcare delivery, the standardization offered by the electronic health record (EHR) system will undoubtedly be confronted by the heterogeneity of clinical consultations. Determination of the various characteristics considered essential for a consultation can help standardize the processes and improve the quality of communication.
Study design: Opinion surveys with a 16-question, Web-based questionnaire about inpatient consultations.
Setting: Four Minnesota teaching hospitals affiliated with the University of Minnesota.
Synopsis: This study surveyed 651 physicians, mostly from general medicine and pediatrics (30% in-training; 54% were more than five years out of training). The response rate to the survey was 50% (323). Responses were analyzed separately for physicians predominantly requesting consultations (requesters) and those predominantly providing them (consultants).
Regarding the consultation request, the majority of consultants preferred a precise consult question (94%), contact information of the ordering provider (68%), and the urgency of consultation (66%), with telephonic communication for emergent consults (75%). Responses were similar regardless of practice site, specialty, or experience.
Regarding the consultation, more requesters desired verbal communication over written advice alone: Sixty-six percent preferred to have the rationale of the recommendations explained. They also preferred a separate recommendations section (48%) with bulleted suggestions (69%) at the top or bottom of the note (74%). Emphasis was placed on specificity of drug names, dose, and duration of therapy (80%), along with alternative options (76%). Most requesters desired a clear “signoff” note when appropriate, with a follow-up plan (74%) or scheduled appointments (44%).
Bottom line: For consultations, the majority of physicians prefer an explanation of medical decision-making, a crisp recommendation section, and specific directions for follow-up.
Citation: Boulware DR, Dekarske AS, Filice GA. Physician preferences for elements of effective consultations. J Gen Intern Med. 2010;25(1):25-30.
CT Scanning Could Be Related to a Future Risk of Cancer at a Population Level
Clinical question: Does the accelerated use of CT scans increase the future risk for radiation-related cancer?
Background: Computed tomography (CT) has come through as a powerful diagnostic and interventional imaging modality at the cost of higher radiation exposures. The potential cancer risk is minimal at an individual level; however, CT technology is used in more than 70 million scans annually. This volume can translate into a significant number of future cancers in the population.
Study design: Indirect risk modeling based on CT scan frequencies and radiation risk models.
Synopsis: Annual frequencies of CT scans (age- and sex-specific) were extracted from insurance claims. The study included 57 million scans, of which 30% were performed in adults 35 to 54 years old. The majority of scans were in females (60%).
Age-specific cancer risk for each CT scan type was estimated through published radiation risk models and national surveys. The projected number of incident cancers per 10,000 scans was highest for chest or abdominal CT angiography (CTA) and whole-body CT. Incidence was higher for females.
The CT scan frequencies were combined with the cancer risk, and it was estimated that approximately 29,000 (95% UL, 15,000-45,000) future cancers could be related to the exposure from CT scans. Uncertainty limits (UL), an estimation of the total error of measurement, accounted for statistical and subjective uncertainties. The risk was dependent on the radiation dose (chest CTA) and frequency of use (abdomen/pelvis followed by chest and head). The most common cancers were lung, colon, and leukemia.
Two-thirds of the projected cancers were in females and attributable to the higher frequency of scans in women coupled with their dual risk of breast and lung cancer with chest radiation. The results provide potential study targets for risk-reduction efforts.
Bottom line: CTA of the chest, abdomen, or pelvis could be related to risk of future cancers, especially in middle-aged females.
Citation: Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
Early Resumption of Low-Dose Aspirin after Peptic Ulcer Bleeding Might Be Beneficial
Clinical question: Is it safe to restart aspirin after acute gastrointestinal (GI) bleeding in patients with cardiovascular or cerebrovascular disease?
Background: The increasing cardiovascular burden in the aging population has indirectly increased aspirin-related peptic ulcer bleeding. Proton-pump inhibitors (PPI) have shown promise in reducing recurrent GI bleeding in non-aspirin-related cases. It is unclear if this protective effect applies to patients on aspirin and, if so, if aspirin resumption after endoscopic treatment is safe.
Study design: Parallel, randomized, placebo-controlled, noninferiority trial.
Setting: Single tertiary endoscopy center in Hong Kong.
Synopsis: One hundred fifty-six patients with aspirin-related peptic ulcer bleeding were selected for the study. After successful endoscopic treatment and 72 hours on pantoprazole infusion, the patients were started on oral pantoprazole for the duration of the study (eight weeks). Patients were equally randomized to receive low-dose aspirin (80 mg/d) or placebo. Primary outcome was recurrent bleeding within 30 days. Secondary outcomes included eight-week all-cause mortality, cause-specific mortality, and recurrence of cardiovascular events.
The aspirin group had a 50% higher risk of recurrent bleeding within 30 days compared with placebo (10.3% vs. 5.4%). However, for the secondary endpoints, aspirin had lower all-cause mortality (1.3% vs. 12.9%), which was not related to increased GI bleeding. On the other hand, discontinuation of aspirin and use of PPI in the placebo group did not prevent mortality related to GI complications.
The small numbers restrict interpretation of the mortality rates but offer support to the fact that the cardioprotective effects of aspirin outweigh its potential for GI bleeding. It is to be noted that these results cannot be extrapolated to higher doses of aspirin.
Bottom line: Early resumption of aspirin after successful treatment of peptic ulcer bleeding might increase the risk of rebleeding but potentially decreases overall mortality.
Citation: Sung JJ, Lau JY, Ching JY, et al. Continuation of low-dose aspirin therapy in peptic ulcer bleeding: a randomized trial. Ann Intern Med. 2010;152(1):1-9.
A Substantial Number of Elderly Patients with Pneumonia Have Pulmonary Malignancy
Clinical question: What is the incidence of, and risk factors for, diagnosis of lung cancer after discharge for pneumonia?
Background: Pneumonia-related admissions in elderly individuals have increased by nearly 20% during the past two decades. Based on the risk profile of this age group, many physicians recommend follow-up chest imaging after pneumonia to ensure resolution and exclude underlying malignancy. However, this practice is not backed by substantial evidence.
Study design: Retrospective cohort study from administrative databases of the VA system.
Setting: Veteran Affairs (VA) Health Care System.
Synopsis: More than 40,000 patients (older than 65, 98.1% male) hospitalized for pneumonia were included in the study. These patients had no pneumonia in the preceding year and did not carry a diagnosis of lung cancer. During the follow-up period of up to five years, a significant proportion (9.2%) of these patients were diagnosed with pulmonary malignancy.
Pertinent factors associated with increased risk of diagnosis included active tobacco use, COPD, and prior nonpulmonary malignancy. Interestingly, stroke, diabetes, dementia, and heart failure were associated with a lower risk of diagnosis, likely due to early mortality from these diseases prior to diagnosis of lung cancer.
Mean time to diagnosis was 297 days, with just 27% diagnosed within 30 days. On mortality analysis, 12.9% (n=5270) of the patients died within 30 days and 20.7% (n=8451) within 90 days. Thus, a period of surveillance of 30 to 90 days following pneumonia, especially in patients with risk factors, could be beneficial.
This study was limited due to the shortcomings of database analyses. Also, the predominantly male, elderly, veteran population restricts extrapolation to the general population.
Bottom line: Patients with risk factors for lung cancer might benefit from surveillance chest imaging after hospitalization for pneumonia to rule out an underlying malignancy.
Citation: Mortensen EM, Copeland LA, Pugh MJ, et al. Diagnosis of pulmonary malignancy after hospitalization for pneumonia. Am J Med. 2010:123(1):66-71.
Hospital-Associated Hyponatremia of Any Severity Adversely Impacts Mortality and Financial Metrics
Clinical question: Does hyponatremia during a hospitalization prophesize a worse outcome?
Study design: Retrospective cohort study from 2002-2007.
Setting: Urban academic medical center.
Synopsis: This study included 53,236 adults based on the presence of admission or subsequent hyponatremia (defined as [Na+] <138 mEq/L). The patients were classified as community-acquired (CAH=37.9%), hospital-aggravated (5.7%), or hospital-acquired hyponatremia (HAH=38.2%).
Across all subgroups, all types of hyponatremia were independently associated with worse primary outcomes, including an increase in hospital mortality (CAH 52%, HAH 66%), prolongation of hospital stay, and discharge to a facility. Also, for the same [Na+], HAH had significantly increased mortality compared with CAH. Though the elderly were more prone to develop hyponatremia, patients younger than 65 had worse outcomes.
The severity of hyponatremia prognosticated adverse outcomes. The liberal definition of hyponatremia, as opposed to the current standard of <135 mEq/L, explains the large numbers in prevalence. However, even mild hyponatremia (133 mEq/L to 137) was linked to poor outcomes (adjusted OR 1.34; CI 1.18-1.51).
The study weaknesses include the use of administrative codes to identify comorbidities, less applicability to outpatient setting, and lack of evaluation of outcomes postdischarge. However, the robust numbers do establish inpatient hyponatremia as a marker of worse outcomes.
Bottom line: Inpatient hyponatremia of any severity is a marker of increased mortality and excessive financial burden.
Citation: Wald R, Jaber BL, Price LL, Upadhyay A, Madias NE. Impact of hospital-associated hyponatremia on selected outcomes. Arch Intern Med. 2010;170(3):294-302.
Patients Lack Awareness and Prefer to Be Updated Regarding Their Inpatient Medications
Clinical question: Is patient knowledge of their medications deficient, and does this reflect a lack of desire to be involved in the medication reconciliation process?
Background: Medication errors remain a significant healthcare problem due to their potential to increase morbidity. For medication administration errors, apart from the dispensing pharmacist and the nurses, patients could be the final checkpoint to ensure medication safety. However, their awareness and enthusiasm to participate has not been adequately assessed in the literature.
Study design: A cross-sectional study using individual surveys to assess awareness and attitudes regarding inpatient medications.
Setting: Single tertiary-care academic teaching hospital.
Synopsis: Fifty cognitively intact adult patients were consented for the study. Of these, 54% provided an accurate recollection of their outpatient medications. When they were surveyed regarding inpatient medications, 96% omitted at least one medication, with the average of 6.8 medication omissions. This was noted to correlate with age >65 years. Also, 44% erroneously presumed they were on a medication while they were in the hospital, even though they weren’t.
When attitudes were surveyed, most of the patients would have preferred to get an inpatient medication list (78%) with the goal of improving their satisfaction (81%) and reducing errors (94%). Also, no association was found between patients’ errors of omission and their reported desire to be involved in the medication safety process.
This small study was limited to cognitively intact patients only. Also, the relatively younger age might cause an overestimation of patient interest in participation. However, the results highlight key medication reconciliation issues. Although patient involvement is desirable, a systematic program of educating them about their medications would be required to make their feedback effective and useful.
Bottom line: Healthy patients might be unaware of their exact hospital medications but prefer to be kept in the loop.
Citation: Cumbler E, Wald H, Kutner J. Lack of patient knowledge regarding hospital medications. J Hosp Med. 2010;5(2):83-86.
Monoclonal Antibodies against Clostridium difficile Toxins Prevent Recurrence
Clinical question: Are human monoclonal antibodies against C. difficile toxin A (CDA1) and B (CDB1) effective in preventing recurrence of C. diff infection (CDI)?
Background: Widespread use of antibiotics, coupled with the emergence of the hypervirulent (B1/NAP1/027) strain of C. diff, has altered the epidemiology of CDI. Even with effective treatment regimens, there is an escalation in severity, treatment failures, and recurrences. Antibodies against the C. diff toxins are being evaluated as the next frontier in treatment of CDI.
Study design: Phase 2 randomized, double-blind, placebo-controlled trial.
Setting: Thirty study centers in Canada and the U.S.
Synopsis: Two hundred patients with laboratory documented CDI on standard therapy with either metronidazole or vancomycin were randomized to receive a single IV infusion of combined monoclonal antibodies against CDA1 and CDB1 (n=101) or a normal saline placebo infusion (n=99). Patients were followed for 84 days with daily stool counts and intermittent blood samples for immunogenicity analysis.
The primary endpoint of recurrence of laboratory-proven C. diff diarrhea was significantly lower in the monoclonal antibody group (7% vs. 25% in placebo. 95% CI, 7-29; P <0.001). In a subgroup analysis of the epidemic BI/NAP1/027 strain, this favorable association persisted (8% vs. 32%). Recurrence in the antibody group was seen more in elderly patients hospitalized with a higher severity of underlying disease.
Secondary endpoints relating to the initial episode of CDI including treatment failure, severity of diarrhea, and duration to resolution were not significantly different between the two groups. Fewer accounts of serious adverse events were documented in the antibody group (18 patients vs. 28 patients in placebo, P=0.09), and immunogenicity was not detected in any patient.
Bottom line: Monoclonal antibody infusion against C. diff toxins reduces recurrence of infection, even with a hypervirulent (B1/NAP1/027) strain, without any significant adverse events.
Citation: Lowy I, Molrine DC, Leav BA, et al. Treatment with monoclonal antibodies against Clostridium difficile toxins. N Engl J Med. 2010; 362(3):197-205. TH
In This Edition
Literature at a Glance
A guide to this month’s studies
- Effect of restrictive antibiotic policies on dosing timeliness
- Desired consultation format and content
- Risk of cancer associated with CT imaging
- Bleeding, mortality with aspirin after peptic ulcer bleed
- Diagnosis of lung cancer after pneumonia
- Outcomes associated with hyponatremia
- Patient awareness, interest in inpatient medication list
- Monoclonal antibodies in C. difficile
Restrictive Antimicrobial Policy Delays Administration
Clinical question: Does the approval process for restricted on-formulary antimicrobials cause a significant delay in their administration?
Background: Widespread and often unwarranted, antimicrobial use in the hospital lends itself to the development of microbial resistance and increases overall costs. To curb such practices, many hospitals require subspecialty approval prior to dispensing select broad-spectrum antimicrobials. Though shown to improve outcomes, the impact of the approval process on the timeliness of antimicrobial administration remains to be seen.
Study design: Retrospective cohort study.
Setting: Tertiary-care university hospital.
Synopsis: The study included 3,251 inpatients with computerized orders for a “stat” first dose of any of 24 pre-selected, parenteral antimicrobials. Time lag (more than one hour, and more than two hours) to nursing documentation of drug administration was separately analyzed for restricted and unrestricted antimicrobials.
Delay of more than one hour was significantly higher for restricted antimicrobials with an odds ratio of 1.49 (95% CI; 1.23-1.82), while the odds ratio for a delay of more than two hours was 1.78 (95% CI, 1.39-2.21). Also, for restricted antimicrobials, the percentage of orders delayed for more than one hour was significantly different between daytime and nighttime (when the first dose was exempt from pre-approval) orders: 46.1% versus 38.8% (P<0.001). For unrestricted drugs, delay was uniform irrespective of time of day (36.4% of daytime and 36.6% of nighttime orders were delayed more than one hour). The effect of delay in drug administration on patient outcomes was not evaluated.
Though the approval process aims in part to affect resistance patterns and overall costs, this research highlights the need to minimize the delay in administration and probably skip the approval for the first dose in critically ill patients.
Bottom line: Antibiotic approval processes can delay their administration in hospitalized patients, but the effect of this delay on patient outcomes is not yet known.
Citation: Winters BD, Thiemann DR, Brotman DJ. Impact of a restrictive antimicrobial policy on the process and timing of antimicrobial administration. J Hosp Med. 2010;5(1):E41-45.
Physicians Uphold Tenets of Effective Consultation while Highlighting Some Newer Viewpoints
Clinical question: What key features of a consultation are most desirable for physicians?
Background: With new changes in healthcare delivery, the standardization offered by the electronic health record (EHR) system will undoubtedly be confronted by the heterogeneity of clinical consultations. Determination of the various characteristics considered essential for a consultation can help standardize the processes and improve the quality of communication.
Study design: Opinion surveys with a 16-question, Web-based questionnaire about inpatient consultations.
Setting: Four Minnesota teaching hospitals affiliated with the University of Minnesota.
Synopsis: This study surveyed 651 physicians, mostly from general medicine and pediatrics (30% in-training; 54% were more than five years out of training). The response rate to the survey was 50% (323). Responses were analyzed separately for physicians predominantly requesting consultations (requesters) and those predominantly providing them (consultants).
Regarding the consultation request, the majority of consultants preferred a precise consult question (94%), contact information of the ordering provider (68%), and the urgency of consultation (66%), with telephonic communication for emergent consults (75%). Responses were similar regardless of practice site, specialty, or experience.
Regarding the consultation, more requesters desired verbal communication over written advice alone: Sixty-six percent preferred to have the rationale of the recommendations explained. They also preferred a separate recommendations section (48%) with bulleted suggestions (69%) at the top or bottom of the note (74%). Emphasis was placed on specificity of drug names, dose, and duration of therapy (80%), along with alternative options (76%). Most requesters desired a clear “signoff” note when appropriate, with a follow-up plan (74%) or scheduled appointments (44%).
Bottom line: For consultations, the majority of physicians prefer an explanation of medical decision-making, a crisp recommendation section, and specific directions for follow-up.
Citation: Boulware DR, Dekarske AS, Filice GA. Physician preferences for elements of effective consultations. J Gen Intern Med. 2010;25(1):25-30.
CT Scanning Could Be Related to a Future Risk of Cancer at a Population Level
Clinical question: Does the accelerated use of CT scans increase the future risk for radiation-related cancer?
Background: Computed tomography (CT) has come through as a powerful diagnostic and interventional imaging modality at the cost of higher radiation exposures. The potential cancer risk is minimal at an individual level; however, CT technology is used in more than 70 million scans annually. This volume can translate into a significant number of future cancers in the population.
Study design: Indirect risk modeling based on CT scan frequencies and radiation risk models.
Synopsis: Annual frequencies of CT scans (age- and sex-specific) were extracted from insurance claims. The study included 57 million scans, of which 30% were performed in adults 35 to 54 years old. The majority of scans were in females (60%).
Age-specific cancer risk for each CT scan type was estimated through published radiation risk models and national surveys. The projected number of incident cancers per 10,000 scans was highest for chest or abdominal CT angiography (CTA) and whole-body CT. Incidence was higher for females.
The CT scan frequencies were combined with the cancer risk, and it was estimated that approximately 29,000 (95% UL, 15,000-45,000) future cancers could be related to the exposure from CT scans. Uncertainty limits (UL), an estimation of the total error of measurement, accounted for statistical and subjective uncertainties. The risk was dependent on the radiation dose (chest CTA) and frequency of use (abdomen/pelvis followed by chest and head). The most common cancers were lung, colon, and leukemia.
Two-thirds of the projected cancers were in females and attributable to the higher frequency of scans in women coupled with their dual risk of breast and lung cancer with chest radiation. The results provide potential study targets for risk-reduction efforts.
Bottom line: CTA of the chest, abdomen, or pelvis could be related to risk of future cancers, especially in middle-aged females.
Citation: Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
Early Resumption of Low-Dose Aspirin after Peptic Ulcer Bleeding Might Be Beneficial
Clinical question: Is it safe to restart aspirin after acute gastrointestinal (GI) bleeding in patients with cardiovascular or cerebrovascular disease?
Background: The increasing cardiovascular burden in the aging population has indirectly increased aspirin-related peptic ulcer bleeding. Proton-pump inhibitors (PPI) have shown promise in reducing recurrent GI bleeding in non-aspirin-related cases. It is unclear if this protective effect applies to patients on aspirin and, if so, if aspirin resumption after endoscopic treatment is safe.
Study design: Parallel, randomized, placebo-controlled, noninferiority trial.
Setting: Single tertiary endoscopy center in Hong Kong.
Synopsis: One hundred fifty-six patients with aspirin-related peptic ulcer bleeding were selected for the study. After successful endoscopic treatment and 72 hours on pantoprazole infusion, the patients were started on oral pantoprazole for the duration of the study (eight weeks). Patients were equally randomized to receive low-dose aspirin (80 mg/d) or placebo. Primary outcome was recurrent bleeding within 30 days. Secondary outcomes included eight-week all-cause mortality, cause-specific mortality, and recurrence of cardiovascular events.
The aspirin group had a 50% higher risk of recurrent bleeding within 30 days compared with placebo (10.3% vs. 5.4%). However, for the secondary endpoints, aspirin had lower all-cause mortality (1.3% vs. 12.9%), which was not related to increased GI bleeding. On the other hand, discontinuation of aspirin and use of PPI in the placebo group did not prevent mortality related to GI complications.
The small numbers restrict interpretation of the mortality rates but offer support to the fact that the cardioprotective effects of aspirin outweigh its potential for GI bleeding. It is to be noted that these results cannot be extrapolated to higher doses of aspirin.
Bottom line: Early resumption of aspirin after successful treatment of peptic ulcer bleeding might increase the risk of rebleeding but potentially decreases overall mortality.
Citation: Sung JJ, Lau JY, Ching JY, et al. Continuation of low-dose aspirin therapy in peptic ulcer bleeding: a randomized trial. Ann Intern Med. 2010;152(1):1-9.
A Substantial Number of Elderly Patients with Pneumonia Have Pulmonary Malignancy
Clinical question: What is the incidence of, and risk factors for, diagnosis of lung cancer after discharge for pneumonia?
Background: Pneumonia-related admissions in elderly individuals have increased by nearly 20% during the past two decades. Based on the risk profile of this age group, many physicians recommend follow-up chest imaging after pneumonia to ensure resolution and exclude underlying malignancy. However, this practice is not backed by substantial evidence.
Study design: Retrospective cohort study from administrative databases of the VA system.
Setting: Veteran Affairs (VA) Health Care System.
Synopsis: More than 40,000 patients (older than 65, 98.1% male) hospitalized for pneumonia were included in the study. These patients had no pneumonia in the preceding year and did not carry a diagnosis of lung cancer. During the follow-up period of up to five years, a significant proportion (9.2%) of these patients were diagnosed with pulmonary malignancy.
Pertinent factors associated with increased risk of diagnosis included active tobacco use, COPD, and prior nonpulmonary malignancy. Interestingly, stroke, diabetes, dementia, and heart failure were associated with a lower risk of diagnosis, likely due to early mortality from these diseases prior to diagnosis of lung cancer.
Mean time to diagnosis was 297 days, with just 27% diagnosed within 30 days. On mortality analysis, 12.9% (n=5270) of the patients died within 30 days and 20.7% (n=8451) within 90 days. Thus, a period of surveillance of 30 to 90 days following pneumonia, especially in patients with risk factors, could be beneficial.
This study was limited due to the shortcomings of database analyses. Also, the predominantly male, elderly, veteran population restricts extrapolation to the general population.
Bottom line: Patients with risk factors for lung cancer might benefit from surveillance chest imaging after hospitalization for pneumonia to rule out an underlying malignancy.
Citation: Mortensen EM, Copeland LA, Pugh MJ, et al. Diagnosis of pulmonary malignancy after hospitalization for pneumonia. Am J Med. 2010:123(1):66-71.
Hospital-Associated Hyponatremia of Any Severity Adversely Impacts Mortality and Financial Metrics
Clinical question: Does hyponatremia during a hospitalization prophesize a worse outcome?
Study design: Retrospective cohort study from 2002-2007.
Setting: Urban academic medical center.
Synopsis: This study included 53,236 adults based on the presence of admission or subsequent hyponatremia (defined as [Na+] <138 mEq/L). The patients were classified as community-acquired (CAH=37.9%), hospital-aggravated (5.7%), or hospital-acquired hyponatremia (HAH=38.2%).
Across all subgroups, all types of hyponatremia were independently associated with worse primary outcomes, including an increase in hospital mortality (CAH 52%, HAH 66%), prolongation of hospital stay, and discharge to a facility. Also, for the same [Na+], HAH had significantly increased mortality compared with CAH. Though the elderly were more prone to develop hyponatremia, patients younger than 65 had worse outcomes.
The severity of hyponatremia prognosticated adverse outcomes. The liberal definition of hyponatremia, as opposed to the current standard of <135 mEq/L, explains the large numbers in prevalence. However, even mild hyponatremia (133 mEq/L to 137) was linked to poor outcomes (adjusted OR 1.34; CI 1.18-1.51).
The study weaknesses include the use of administrative codes to identify comorbidities, less applicability to outpatient setting, and lack of evaluation of outcomes postdischarge. However, the robust numbers do establish inpatient hyponatremia as a marker of worse outcomes.
Bottom line: Inpatient hyponatremia of any severity is a marker of increased mortality and excessive financial burden.
Citation: Wald R, Jaber BL, Price LL, Upadhyay A, Madias NE. Impact of hospital-associated hyponatremia on selected outcomes. Arch Intern Med. 2010;170(3):294-302.
Patients Lack Awareness and Prefer to Be Updated Regarding Their Inpatient Medications
Clinical question: Is patient knowledge of their medications deficient, and does this reflect a lack of desire to be involved in the medication reconciliation process?
Background: Medication errors remain a significant healthcare problem due to their potential to increase morbidity. For medication administration errors, apart from the dispensing pharmacist and the nurses, patients could be the final checkpoint to ensure medication safety. However, their awareness and enthusiasm to participate has not been adequately assessed in the literature.
Study design: A cross-sectional study using individual surveys to assess awareness and attitudes regarding inpatient medications.
Setting: Single tertiary-care academic teaching hospital.
Synopsis: Fifty cognitively intact adult patients were consented for the study. Of these, 54% provided an accurate recollection of their outpatient medications. When they were surveyed regarding inpatient medications, 96% omitted at least one medication, with the average of 6.8 medication omissions. This was noted to correlate with age >65 years. Also, 44% erroneously presumed they were on a medication while they were in the hospital, even though they weren’t.
When attitudes were surveyed, most of the patients would have preferred to get an inpatient medication list (78%) with the goal of improving their satisfaction (81%) and reducing errors (94%). Also, no association was found between patients’ errors of omission and their reported desire to be involved in the medication safety process.
This small study was limited to cognitively intact patients only. Also, the relatively younger age might cause an overestimation of patient interest in participation. However, the results highlight key medication reconciliation issues. Although patient involvement is desirable, a systematic program of educating them about their medications would be required to make their feedback effective and useful.
Bottom line: Healthy patients might be unaware of their exact hospital medications but prefer to be kept in the loop.
Citation: Cumbler E, Wald H, Kutner J. Lack of patient knowledge regarding hospital medications. J Hosp Med. 2010;5(2):83-86.
Monoclonal Antibodies against Clostridium difficile Toxins Prevent Recurrence
Clinical question: Are human monoclonal antibodies against C. difficile toxin A (CDA1) and B (CDB1) effective in preventing recurrence of C. diff infection (CDI)?
Background: Widespread use of antibiotics, coupled with the emergence of the hypervirulent (B1/NAP1/027) strain of C. diff, has altered the epidemiology of CDI. Even with effective treatment regimens, there is an escalation in severity, treatment failures, and recurrences. Antibodies against the C. diff toxins are being evaluated as the next frontier in treatment of CDI.
Study design: Phase 2 randomized, double-blind, placebo-controlled trial.
Setting: Thirty study centers in Canada and the U.S.
Synopsis: Two hundred patients with laboratory documented CDI on standard therapy with either metronidazole or vancomycin were randomized to receive a single IV infusion of combined monoclonal antibodies against CDA1 and CDB1 (n=101) or a normal saline placebo infusion (n=99). Patients were followed for 84 days with daily stool counts and intermittent blood samples for immunogenicity analysis.
The primary endpoint of recurrence of laboratory-proven C. diff diarrhea was significantly lower in the monoclonal antibody group (7% vs. 25% in placebo. 95% CI, 7-29; P <0.001). In a subgroup analysis of the epidemic BI/NAP1/027 strain, this favorable association persisted (8% vs. 32%). Recurrence in the antibody group was seen more in elderly patients hospitalized with a higher severity of underlying disease.
Secondary endpoints relating to the initial episode of CDI including treatment failure, severity of diarrhea, and duration to resolution were not significantly different between the two groups. Fewer accounts of serious adverse events were documented in the antibody group (18 patients vs. 28 patients in placebo, P=0.09), and immunogenicity was not detected in any patient.
Bottom line: Monoclonal antibody infusion against C. diff toxins reduces recurrence of infection, even with a hypervirulent (B1/NAP1/027) strain, without any significant adverse events.
Citation: Lowy I, Molrine DC, Leav BA, et al. Treatment with monoclonal antibodies against Clostridium difficile toxins. N Engl J Med. 2010; 362(3):197-205. TH
In This Edition
Literature at a Glance
A guide to this month’s studies
- Effect of restrictive antibiotic policies on dosing timeliness
- Desired consultation format and content
- Risk of cancer associated with CT imaging
- Bleeding, mortality with aspirin after peptic ulcer bleed
- Diagnosis of lung cancer after pneumonia
- Outcomes associated with hyponatremia
- Patient awareness, interest in inpatient medication list
- Monoclonal antibodies in C. difficile
Restrictive Antimicrobial Policy Delays Administration
Clinical question: Does the approval process for restricted on-formulary antimicrobials cause a significant delay in their administration?
Background: Widespread and often unwarranted, antimicrobial use in the hospital lends itself to the development of microbial resistance and increases overall costs. To curb such practices, many hospitals require subspecialty approval prior to dispensing select broad-spectrum antimicrobials. Though shown to improve outcomes, the impact of the approval process on the timeliness of antimicrobial administration remains to be seen.
Study design: Retrospective cohort study.
Setting: Tertiary-care university hospital.
Synopsis: The study included 3,251 inpatients with computerized orders for a “stat” first dose of any of 24 pre-selected, parenteral antimicrobials. Time lag (more than one hour, and more than two hours) to nursing documentation of drug administration was separately analyzed for restricted and unrestricted antimicrobials.
Delay of more than one hour was significantly higher for restricted antimicrobials with an odds ratio of 1.49 (95% CI; 1.23-1.82), while the odds ratio for a delay of more than two hours was 1.78 (95% CI, 1.39-2.21). Also, for restricted antimicrobials, the percentage of orders delayed for more than one hour was significantly different between daytime and nighttime (when the first dose was exempt from pre-approval) orders: 46.1% versus 38.8% (P<0.001). For unrestricted drugs, delay was uniform irrespective of time of day (36.4% of daytime and 36.6% of nighttime orders were delayed more than one hour). The effect of delay in drug administration on patient outcomes was not evaluated.
Though the approval process aims in part to affect resistance patterns and overall costs, this research highlights the need to minimize the delay in administration and probably skip the approval for the first dose in critically ill patients.
Bottom line: Antibiotic approval processes can delay their administration in hospitalized patients, but the effect of this delay on patient outcomes is not yet known.
Citation: Winters BD, Thiemann DR, Brotman DJ. Impact of a restrictive antimicrobial policy on the process and timing of antimicrobial administration. J Hosp Med. 2010;5(1):E41-45.
Physicians Uphold Tenets of Effective Consultation while Highlighting Some Newer Viewpoints
Clinical question: What key features of a consultation are most desirable for physicians?
Background: With new changes in healthcare delivery, the standardization offered by the electronic health record (EHR) system will undoubtedly be confronted by the heterogeneity of clinical consultations. Determination of the various characteristics considered essential for a consultation can help standardize the processes and improve the quality of communication.
Study design: Opinion surveys with a 16-question, Web-based questionnaire about inpatient consultations.
Setting: Four Minnesota teaching hospitals affiliated with the University of Minnesota.
Synopsis: This study surveyed 651 physicians, mostly from general medicine and pediatrics (30% in-training; 54% were more than five years out of training). The response rate to the survey was 50% (323). Responses were analyzed separately for physicians predominantly requesting consultations (requesters) and those predominantly providing them (consultants).
Regarding the consultation request, the majority of consultants preferred a precise consult question (94%), contact information of the ordering provider (68%), and the urgency of consultation (66%), with telephonic communication for emergent consults (75%). Responses were similar regardless of practice site, specialty, or experience.
Regarding the consultation, more requesters desired verbal communication over written advice alone: Sixty-six percent preferred to have the rationale of the recommendations explained. They also preferred a separate recommendations section (48%) with bulleted suggestions (69%) at the top or bottom of the note (74%). Emphasis was placed on specificity of drug names, dose, and duration of therapy (80%), along with alternative options (76%). Most requesters desired a clear “signoff” note when appropriate, with a follow-up plan (74%) or scheduled appointments (44%).
Bottom line: For consultations, the majority of physicians prefer an explanation of medical decision-making, a crisp recommendation section, and specific directions for follow-up.
Citation: Boulware DR, Dekarske AS, Filice GA. Physician preferences for elements of effective consultations. J Gen Intern Med. 2010;25(1):25-30.
CT Scanning Could Be Related to a Future Risk of Cancer at a Population Level
Clinical question: Does the accelerated use of CT scans increase the future risk for radiation-related cancer?
Background: Computed tomography (CT) has come through as a powerful diagnostic and interventional imaging modality at the cost of higher radiation exposures. The potential cancer risk is minimal at an individual level; however, CT technology is used in more than 70 million scans annually. This volume can translate into a significant number of future cancers in the population.
Study design: Indirect risk modeling based on CT scan frequencies and radiation risk models.
Synopsis: Annual frequencies of CT scans (age- and sex-specific) were extracted from insurance claims. The study included 57 million scans, of which 30% were performed in adults 35 to 54 years old. The majority of scans were in females (60%).
Age-specific cancer risk for each CT scan type was estimated through published radiation risk models and national surveys. The projected number of incident cancers per 10,000 scans was highest for chest or abdominal CT angiography (CTA) and whole-body CT. Incidence was higher for females.
The CT scan frequencies were combined with the cancer risk, and it was estimated that approximately 29,000 (95% UL, 15,000-45,000) future cancers could be related to the exposure from CT scans. Uncertainty limits (UL), an estimation of the total error of measurement, accounted for statistical and subjective uncertainties. The risk was dependent on the radiation dose (chest CTA) and frequency of use (abdomen/pelvis followed by chest and head). The most common cancers were lung, colon, and leukemia.
Two-thirds of the projected cancers were in females and attributable to the higher frequency of scans in women coupled with their dual risk of breast and lung cancer with chest radiation. The results provide potential study targets for risk-reduction efforts.
Bottom line: CTA of the chest, abdomen, or pelvis could be related to risk of future cancers, especially in middle-aged females.
Citation: Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169(22):2071-2077.
Early Resumption of Low-Dose Aspirin after Peptic Ulcer Bleeding Might Be Beneficial
Clinical question: Is it safe to restart aspirin after acute gastrointestinal (GI) bleeding in patients with cardiovascular or cerebrovascular disease?
Background: The increasing cardiovascular burden in the aging population has indirectly increased aspirin-related peptic ulcer bleeding. Proton-pump inhibitors (PPI) have shown promise in reducing recurrent GI bleeding in non-aspirin-related cases. It is unclear if this protective effect applies to patients on aspirin and, if so, if aspirin resumption after endoscopic treatment is safe.
Study design: Parallel, randomized, placebo-controlled, noninferiority trial.
Setting: Single tertiary endoscopy center in Hong Kong.
Synopsis: One hundred fifty-six patients with aspirin-related peptic ulcer bleeding were selected for the study. After successful endoscopic treatment and 72 hours on pantoprazole infusion, the patients were started on oral pantoprazole for the duration of the study (eight weeks). Patients were equally randomized to receive low-dose aspirin (80 mg/d) or placebo. Primary outcome was recurrent bleeding within 30 days. Secondary outcomes included eight-week all-cause mortality, cause-specific mortality, and recurrence of cardiovascular events.
The aspirin group had a 50% higher risk of recurrent bleeding within 30 days compared with placebo (10.3% vs. 5.4%). However, for the secondary endpoints, aspirin had lower all-cause mortality (1.3% vs. 12.9%), which was not related to increased GI bleeding. On the other hand, discontinuation of aspirin and use of PPI in the placebo group did not prevent mortality related to GI complications.
The small numbers restrict interpretation of the mortality rates but offer support to the fact that the cardioprotective effects of aspirin outweigh its potential for GI bleeding. It is to be noted that these results cannot be extrapolated to higher doses of aspirin.
Bottom line: Early resumption of aspirin after successful treatment of peptic ulcer bleeding might increase the risk of rebleeding but potentially decreases overall mortality.
Citation: Sung JJ, Lau JY, Ching JY, et al. Continuation of low-dose aspirin therapy in peptic ulcer bleeding: a randomized trial. Ann Intern Med. 2010;152(1):1-9.
A Substantial Number of Elderly Patients with Pneumonia Have Pulmonary Malignancy
Clinical question: What is the incidence of, and risk factors for, diagnosis of lung cancer after discharge for pneumonia?
Background: Pneumonia-related admissions in elderly individuals have increased by nearly 20% during the past two decades. Based on the risk profile of this age group, many physicians recommend follow-up chest imaging after pneumonia to ensure resolution and exclude underlying malignancy. However, this practice is not backed by substantial evidence.
Study design: Retrospective cohort study from administrative databases of the VA system.
Setting: Veteran Affairs (VA) Health Care System.
Synopsis: More than 40,000 patients (older than 65, 98.1% male) hospitalized for pneumonia were included in the study. These patients had no pneumonia in the preceding year and did not carry a diagnosis of lung cancer. During the follow-up period of up to five years, a significant proportion (9.2%) of these patients were diagnosed with pulmonary malignancy.
Pertinent factors associated with increased risk of diagnosis included active tobacco use, COPD, and prior nonpulmonary malignancy. Interestingly, stroke, diabetes, dementia, and heart failure were associated with a lower risk of diagnosis, likely due to early mortality from these diseases prior to diagnosis of lung cancer.
Mean time to diagnosis was 297 days, with just 27% diagnosed within 30 days. On mortality analysis, 12.9% (n=5270) of the patients died within 30 days and 20.7% (n=8451) within 90 days. Thus, a period of surveillance of 30 to 90 days following pneumonia, especially in patients with risk factors, could be beneficial.
This study was limited due to the shortcomings of database analyses. Also, the predominantly male, elderly, veteran population restricts extrapolation to the general population.
Bottom line: Patients with risk factors for lung cancer might benefit from surveillance chest imaging after hospitalization for pneumonia to rule out an underlying malignancy.
Citation: Mortensen EM, Copeland LA, Pugh MJ, et al. Diagnosis of pulmonary malignancy after hospitalization for pneumonia. Am J Med. 2010:123(1):66-71.
Hospital-Associated Hyponatremia of Any Severity Adversely Impacts Mortality and Financial Metrics
Clinical question: Does hyponatremia during a hospitalization prophesize a worse outcome?
Study design: Retrospective cohort study from 2002-2007.
Setting: Urban academic medical center.
Synopsis: This study included 53,236 adults based on the presence of admission or subsequent hyponatremia (defined as [Na+] <138 mEq/L). The patients were classified as community-acquired (CAH=37.9%), hospital-aggravated (5.7%), or hospital-acquired hyponatremia (HAH=38.2%).
Across all subgroups, all types of hyponatremia were independently associated with worse primary outcomes, including an increase in hospital mortality (CAH 52%, HAH 66%), prolongation of hospital stay, and discharge to a facility. Also, for the same [Na+], HAH had significantly increased mortality compared with CAH. Though the elderly were more prone to develop hyponatremia, patients younger than 65 had worse outcomes.
The severity of hyponatremia prognosticated adverse outcomes. The liberal definition of hyponatremia, as opposed to the current standard of <135 mEq/L, explains the large numbers in prevalence. However, even mild hyponatremia (133 mEq/L to 137) was linked to poor outcomes (adjusted OR 1.34; CI 1.18-1.51).
The study weaknesses include the use of administrative codes to identify comorbidities, less applicability to outpatient setting, and lack of evaluation of outcomes postdischarge. However, the robust numbers do establish inpatient hyponatremia as a marker of worse outcomes.
Bottom line: Inpatient hyponatremia of any severity is a marker of increased mortality and excessive financial burden.
Citation: Wald R, Jaber BL, Price LL, Upadhyay A, Madias NE. Impact of hospital-associated hyponatremia on selected outcomes. Arch Intern Med. 2010;170(3):294-302.
Patients Lack Awareness and Prefer to Be Updated Regarding Their Inpatient Medications
Clinical question: Is patient knowledge of their medications deficient, and does this reflect a lack of desire to be involved in the medication reconciliation process?
Background: Medication errors remain a significant healthcare problem due to their potential to increase morbidity. For medication administration errors, apart from the dispensing pharmacist and the nurses, patients could be the final checkpoint to ensure medication safety. However, their awareness and enthusiasm to participate has not been adequately assessed in the literature.
Study design: A cross-sectional study using individual surveys to assess awareness and attitudes regarding inpatient medications.
Setting: Single tertiary-care academic teaching hospital.
Synopsis: Fifty cognitively intact adult patients were consented for the study. Of these, 54% provided an accurate recollection of their outpatient medications. When they were surveyed regarding inpatient medications, 96% omitted at least one medication, with the average of 6.8 medication omissions. This was noted to correlate with age >65 years. Also, 44% erroneously presumed they were on a medication while they were in the hospital, even though they weren’t.
When attitudes were surveyed, most of the patients would have preferred to get an inpatient medication list (78%) with the goal of improving their satisfaction (81%) and reducing errors (94%). Also, no association was found between patients’ errors of omission and their reported desire to be involved in the medication safety process.
This small study was limited to cognitively intact patients only. Also, the relatively younger age might cause an overestimation of patient interest in participation. However, the results highlight key medication reconciliation issues. Although patient involvement is desirable, a systematic program of educating them about their medications would be required to make their feedback effective and useful.
Bottom line: Healthy patients might be unaware of their exact hospital medications but prefer to be kept in the loop.
Citation: Cumbler E, Wald H, Kutner J. Lack of patient knowledge regarding hospital medications. J Hosp Med. 2010;5(2):83-86.
Monoclonal Antibodies against Clostridium difficile Toxins Prevent Recurrence
Clinical question: Are human monoclonal antibodies against C. difficile toxin A (CDA1) and B (CDB1) effective in preventing recurrence of C. diff infection (CDI)?
Background: Widespread use of antibiotics, coupled with the emergence of the hypervirulent (B1/NAP1/027) strain of C. diff, has altered the epidemiology of CDI. Even with effective treatment regimens, there is an escalation in severity, treatment failures, and recurrences. Antibodies against the C. diff toxins are being evaluated as the next frontier in treatment of CDI.
Study design: Phase 2 randomized, double-blind, placebo-controlled trial.
Setting: Thirty study centers in Canada and the U.S.
Synopsis: Two hundred patients with laboratory documented CDI on standard therapy with either metronidazole or vancomycin were randomized to receive a single IV infusion of combined monoclonal antibodies against CDA1 and CDB1 (n=101) or a normal saline placebo infusion (n=99). Patients were followed for 84 days with daily stool counts and intermittent blood samples for immunogenicity analysis.
The primary endpoint of recurrence of laboratory-proven C. diff diarrhea was significantly lower in the monoclonal antibody group (7% vs. 25% in placebo. 95% CI, 7-29; P <0.001). In a subgroup analysis of the epidemic BI/NAP1/027 strain, this favorable association persisted (8% vs. 32%). Recurrence in the antibody group was seen more in elderly patients hospitalized with a higher severity of underlying disease.
Secondary endpoints relating to the initial episode of CDI including treatment failure, severity of diarrhea, and duration to resolution were not significantly different between the two groups. Fewer accounts of serious adverse events were documented in the antibody group (18 patients vs. 28 patients in placebo, P=0.09), and immunogenicity was not detected in any patient.
Bottom line: Monoclonal antibody infusion against C. diff toxins reduces recurrence of infection, even with a hypervirulent (B1/NAP1/027) strain, without any significant adverse events.
Citation: Lowy I, Molrine DC, Leav BA, et al. Treatment with monoclonal antibodies against Clostridium difficile toxins. N Engl J Med. 2010; 362(3):197-205. TH
Observation Care
Many conditions once treated during an “inpatient” hospital stay are currently treated during an “observation” stay (OBS). Although the care remains the same, physician billing is different and requires close attention to admission details for effective charge capture.
Let’s take a look at a typical OBS scenario. A 65-year-old female with longstanding diabetes presents to the ED at 10 p.m. with palpitations, lightheadedness, mild disorientation, and elevated blood sugar. The hospitalist admits the patient to observation, treats her for dehydration, and discharges her the next day. Before billing, the hospitalist should consider the following factors.
Physician of Record
The attending of record writes the orders to admit the patient to observation; indicates the reason for the stay; outlines the plan of care; and manages the patient during the stay. The attending reports the initial patient encounter with the most appropriate initial observation-care code, as reflected by the documentation:1
- 99218: Initial observation care, requiring both a detailed or comprehensive history and exam, and straightforward/low-complexity medical decision-making. Usually, the problem(s) is of low severity.
- 99219: Initial observation care, requiring both a comprehensive history and exam, and moderate-complexity medical decision-making. Usually, the problem(s) is of moderate severity.
- 99220: Initial observation care, requiring both a comprehensive history and exam, and high-complexity medical decision-making. Usually, the problem(s) is of high severity.
While other physicians (e.g., specialists) might be involved in the patient’s care, only the attending physician reports codes 99218-99220. Specialists typically are called to an OBS case for their opinion or advice but do not function as the attending of record. Billing for the specialist (consultation) service depends upon the payor.
For a non-Medicare patient who pays for consultation codes, the specialist reports an outpatient consultation code (99241-99245) for the appropriately documented service. Conversely, Medicare no longer recognizes consultation codes, and specialists must report either a new patient visit code (99201-99205) or established patient visit code (99212-99215) for Medicare beneficiaries.
Selection of the new or established patient codes follows the “three-year rule”: A “new patient” has not received any face-to-face services (e.g., visit or procedure) in any location from any physician within the same group and same specialty within the past three years.2 There could be occasion when a hospitalist is not the attending of record but is asked to provide their opinion, and must report one of the “non-OBS” codes.
The attending of record is permitted to report a discharge service as long as this service occurs on a calendar day different from the admission service (as in the listed scenario). The attending documents the face-to-face discharge service and any pertinent clinical details, and reports 99217 (observation-care discharge-day management).
Length of Stay
Observation-care services typically do not exceed 24 hours and two calendar days. Observation care for more than 48 hours without inpatient admission is not considered medically necessary but might be payable after medical review. Should the OBS stay span more than two calendar days (as might be the case with “downgraded” hospitalizations), hospitalists should report established patient visit codes (99212-99215) for the calendar day(s) between the admission service (99218-99220) and the discharge service (99217).3 The physician must provide and document a face-to-face encounter on each date of service for which a claim was submitted.
A more likely occurrence is the admission and discharge from OBS on the same calendar date. The attending of record reports the code that corresponds to the patient’s length of stay (LOS). If the total LOS is less than eight hours, the attending only reports standard OBS codes (99218-99220). The hospitalist does not separately report the OBS discharge service (99217), even though the documentation must reflect the attending discharge order and corresponding discharge plan. If the total duration of the patient’s stay lasts more than eight hours and does not overlap two calendar days, the attending reports the same-day admit/discharge codes:1
- 99234: Observation or inpatient care, same date admission and discharge, requiring both a detailed or comprehensive history and exam, and straightforward or low-complexity medical decision-making. Usually the presenting problem(s) is of low severity.
- 99235: Observation or inpatient care, same date admission and discharge, requiring a comprehensive history and exam, and moderate-complexity medical decision-making. Usually the presenting problem(s) is of moderate severity.
- 99236: Observation or inpatient care, same date admission and discharge, requiring a comprehensive history and exam, and high-complexity medical decision-making. Usually the presenting problem(s) is of high severity.
OBS discharge service (99217) is not separately reported with 99234-99236 because these codes are valued to include the discharge component (e.g., the comprehensive service, 99236 [4.26 wRVU, $211], is equivalent to its components, 99220 [2.99 wRVU, $148] and 99217 [1.28 wRVU, $68]). The attending must document the total duration of the stay, as well as the face-to-face service and the corresponding details of each service component (i.e., both an admission and discharge note).3TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center in Philadelphia. She is also on the faculty of SHM’s inpatient coding course.
References
- Abraham M, Beebe M, Dalton J, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2010:11-16.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.7A. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.8C. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.8D. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 1, Section 50.3. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c01.pdf. Accessed May 12, 2010.
Many conditions once treated during an “inpatient” hospital stay are currently treated during an “observation” stay (OBS). Although the care remains the same, physician billing is different and requires close attention to admission details for effective charge capture.
Let’s take a look at a typical OBS scenario. A 65-year-old female with longstanding diabetes presents to the ED at 10 p.m. with palpitations, lightheadedness, mild disorientation, and elevated blood sugar. The hospitalist admits the patient to observation, treats her for dehydration, and discharges her the next day. Before billing, the hospitalist should consider the following factors.
Physician of Record
The attending of record writes the orders to admit the patient to observation; indicates the reason for the stay; outlines the plan of care; and manages the patient during the stay. The attending reports the initial patient encounter with the most appropriate initial observation-care code, as reflected by the documentation:1
- 99218: Initial observation care, requiring both a detailed or comprehensive history and exam, and straightforward/low-complexity medical decision-making. Usually, the problem(s) is of low severity.
- 99219: Initial observation care, requiring both a comprehensive history and exam, and moderate-complexity medical decision-making. Usually, the problem(s) is of moderate severity.
- 99220: Initial observation care, requiring both a comprehensive history and exam, and high-complexity medical decision-making. Usually, the problem(s) is of high severity.
While other physicians (e.g., specialists) might be involved in the patient’s care, only the attending physician reports codes 99218-99220. Specialists typically are called to an OBS case for their opinion or advice but do not function as the attending of record. Billing for the specialist (consultation) service depends upon the payor.
For a non-Medicare patient who pays for consultation codes, the specialist reports an outpatient consultation code (99241-99245) for the appropriately documented service. Conversely, Medicare no longer recognizes consultation codes, and specialists must report either a new patient visit code (99201-99205) or established patient visit code (99212-99215) for Medicare beneficiaries.
Selection of the new or established patient codes follows the “three-year rule”: A “new patient” has not received any face-to-face services (e.g., visit or procedure) in any location from any physician within the same group and same specialty within the past three years.2 There could be occasion when a hospitalist is not the attending of record but is asked to provide their opinion, and must report one of the “non-OBS” codes.
The attending of record is permitted to report a discharge service as long as this service occurs on a calendar day different from the admission service (as in the listed scenario). The attending documents the face-to-face discharge service and any pertinent clinical details, and reports 99217 (observation-care discharge-day management).
Length of Stay
Observation-care services typically do not exceed 24 hours and two calendar days. Observation care for more than 48 hours without inpatient admission is not considered medically necessary but might be payable after medical review. Should the OBS stay span more than two calendar days (as might be the case with “downgraded” hospitalizations), hospitalists should report established patient visit codes (99212-99215) for the calendar day(s) between the admission service (99218-99220) and the discharge service (99217).3 The physician must provide and document a face-to-face encounter on each date of service for which a claim was submitted.
A more likely occurrence is the admission and discharge from OBS on the same calendar date. The attending of record reports the code that corresponds to the patient’s length of stay (LOS). If the total LOS is less than eight hours, the attending only reports standard OBS codes (99218-99220). The hospitalist does not separately report the OBS discharge service (99217), even though the documentation must reflect the attending discharge order and corresponding discharge plan. If the total duration of the patient’s stay lasts more than eight hours and does not overlap two calendar days, the attending reports the same-day admit/discharge codes:1
- 99234: Observation or inpatient care, same date admission and discharge, requiring both a detailed or comprehensive history and exam, and straightforward or low-complexity medical decision-making. Usually the presenting problem(s) is of low severity.
- 99235: Observation or inpatient care, same date admission and discharge, requiring a comprehensive history and exam, and moderate-complexity medical decision-making. Usually the presenting problem(s) is of moderate severity.
- 99236: Observation or inpatient care, same date admission and discharge, requiring a comprehensive history and exam, and high-complexity medical decision-making. Usually the presenting problem(s) is of high severity.
OBS discharge service (99217) is not separately reported with 99234-99236 because these codes are valued to include the discharge component (e.g., the comprehensive service, 99236 [4.26 wRVU, $211], is equivalent to its components, 99220 [2.99 wRVU, $148] and 99217 [1.28 wRVU, $68]). The attending must document the total duration of the stay, as well as the face-to-face service and the corresponding details of each service component (i.e., both an admission and discharge note).3TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center in Philadelphia. She is also on the faculty of SHM’s inpatient coding course.
References
- Abraham M, Beebe M, Dalton J, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2010:11-16.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.7A. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.8C. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.8D. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 1, Section 50.3. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c01.pdf. Accessed May 12, 2010.
Many conditions once treated during an “inpatient” hospital stay are currently treated during an “observation” stay (OBS). Although the care remains the same, physician billing is different and requires close attention to admission details for effective charge capture.
Let’s take a look at a typical OBS scenario. A 65-year-old female with longstanding diabetes presents to the ED at 10 p.m. with palpitations, lightheadedness, mild disorientation, and elevated blood sugar. The hospitalist admits the patient to observation, treats her for dehydration, and discharges her the next day. Before billing, the hospitalist should consider the following factors.
Physician of Record
The attending of record writes the orders to admit the patient to observation; indicates the reason for the stay; outlines the plan of care; and manages the patient during the stay. The attending reports the initial patient encounter with the most appropriate initial observation-care code, as reflected by the documentation:1
- 99218: Initial observation care, requiring both a detailed or comprehensive history and exam, and straightforward/low-complexity medical decision-making. Usually, the problem(s) is of low severity.
- 99219: Initial observation care, requiring both a comprehensive history and exam, and moderate-complexity medical decision-making. Usually, the problem(s) is of moderate severity.
- 99220: Initial observation care, requiring both a comprehensive history and exam, and high-complexity medical decision-making. Usually, the problem(s) is of high severity.
While other physicians (e.g., specialists) might be involved in the patient’s care, only the attending physician reports codes 99218-99220. Specialists typically are called to an OBS case for their opinion or advice but do not function as the attending of record. Billing for the specialist (consultation) service depends upon the payor.
For a non-Medicare patient who pays for consultation codes, the specialist reports an outpatient consultation code (99241-99245) for the appropriately documented service. Conversely, Medicare no longer recognizes consultation codes, and specialists must report either a new patient visit code (99201-99205) or established patient visit code (99212-99215) for Medicare beneficiaries.
Selection of the new or established patient codes follows the “three-year rule”: A “new patient” has not received any face-to-face services (e.g., visit or procedure) in any location from any physician within the same group and same specialty within the past three years.2 There could be occasion when a hospitalist is not the attending of record but is asked to provide their opinion, and must report one of the “non-OBS” codes.
The attending of record is permitted to report a discharge service as long as this service occurs on a calendar day different from the admission service (as in the listed scenario). The attending documents the face-to-face discharge service and any pertinent clinical details, and reports 99217 (observation-care discharge-day management).
Length of Stay
Observation-care services typically do not exceed 24 hours and two calendar days. Observation care for more than 48 hours without inpatient admission is not considered medically necessary but might be payable after medical review. Should the OBS stay span more than two calendar days (as might be the case with “downgraded” hospitalizations), hospitalists should report established patient visit codes (99212-99215) for the calendar day(s) between the admission service (99218-99220) and the discharge service (99217).3 The physician must provide and document a face-to-face encounter on each date of service for which a claim was submitted.
A more likely occurrence is the admission and discharge from OBS on the same calendar date. The attending of record reports the code that corresponds to the patient’s length of stay (LOS). If the total LOS is less than eight hours, the attending only reports standard OBS codes (99218-99220). The hospitalist does not separately report the OBS discharge service (99217), even though the documentation must reflect the attending discharge order and corresponding discharge plan. If the total duration of the patient’s stay lasts more than eight hours and does not overlap two calendar days, the attending reports the same-day admit/discharge codes:1
- 99234: Observation or inpatient care, same date admission and discharge, requiring both a detailed or comprehensive history and exam, and straightforward or low-complexity medical decision-making. Usually the presenting problem(s) is of low severity.
- 99235: Observation or inpatient care, same date admission and discharge, requiring a comprehensive history and exam, and moderate-complexity medical decision-making. Usually the presenting problem(s) is of moderate severity.
- 99236: Observation or inpatient care, same date admission and discharge, requiring a comprehensive history and exam, and high-complexity medical decision-making. Usually the presenting problem(s) is of high severity.
OBS discharge service (99217) is not separately reported with 99234-99236 because these codes are valued to include the discharge component (e.g., the comprehensive service, 99236 [4.26 wRVU, $211], is equivalent to its components, 99220 [2.99 wRVU, $148] and 99217 [1.28 wRVU, $68]). The attending must document the total duration of the stay, as well as the face-to-face service and the corresponding details of each service component (i.e., both an admission and discharge note).3TH
Carol Pohlig is a billing and coding expert with the University of Pennsylvania Medical Center in Philadelphia. She is also on the faculty of SHM’s inpatient coding course.
References
- Abraham M, Beebe M, Dalton J, Evans D, Glenn R. Current Procedural Terminology Professional Edition. Chicago: American Medical Association Press; 2010:11-16.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.7A. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.8C. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 12, Section 30.6.8D. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c12.pdf. Accessed May 11, 2010.
- Medicare Claims Processing Manual: Chapter 1, Section 50.3. Centers for Medicare and Medicaid Services website. Available at: www.cms.hhs.gov/manuals/downloads/clm104c01.pdf. Accessed May 12, 2010.
What Is the Most Cost- Effective Evaluation for a First Syncopal Episode?
Case
A 71-year-old woman is admitted after losing consciousness and falling at home. Her history is significant for hypertension, bilateral internal carotid artery stenoses, chronic kidney disease, and diabetes. Resting vital signs are normal. Cardiac, pulmonary, and neurologic exams are unremarkable, as is an electrocardiogram (ECG). She was noted to have a small scalp laceration. Noncontrast CT of the head demonstrates a small occipital subdural bleed thought to be a result of her fall.
What is the most cost-effective evaluation for this patient admitted with suspected first syncopal episode?
Overview
Syncope is defined as sudden, self-terminating loss of consciousness. The final common pathway of all causes of syncope is global cerebral hypoperfusion—specifically, hypoperfusion of the reticular activating system. The differential diagnosis of syncopal loss of consciousness includes neurally mediated (e.g., vasovagal) syncope, orthostatic hypotension, cardiac arrhythmias, structural heart disease, and cerebrovascular disease.
Among young, otherwise healthy people, neurally mediated syncope, which has a relatively benign prognosis, is by far the most common etiology, while in older patients, primary cardiac causes are more common. Nonsyncopal mechanisms, such as seizure and hypoglycemia, should also be considered in the differential diagnosis of transient loss of consciousness (see Table 1, p. 19).1
Syncope is a common problem, accounting for 1% to 2% of ED visits in the U.S.2 The primary objective for evaluation is identification of individuals at increased risk of death due to associated conditions, especially cardiac conditions such as structural heart disease; myocardial ischemia and infarction (MI); Wolff-Parkinson-White, Brugada, or long QT syndromes; and polymorphic ventricular tachycardia.3 True syncope can be associated with other concerning causes, such as aortic stenosis, aortic dissection, and massive pulmonary embolus, as well as arrhythmias from underlying cardiac disease.4
Review of the Data
History: A detailed history and physical examination reveals the cause in 50% of syncopal episodes. Key factors include the account of third-party observers, although it is important to note that tonic-clonic movements can be associated with the global cerebral hypoperfusion of syncope as well as with seizure.4 History of dyspnea, chest pain, or palpitations argue for a primary cardiac or pulmonary cause.
Among patients who endorse a history of palpitations, the sensation of rapid and regular pulsations in the neck points very strongly toward an AV-nodal re-entrant tachycardia, because the carotid pulse and cannon A wave arrive simultaneously in the neck.5 Postictal confusion and focal neurologic exam findings suggest a neurologic rather than syncopal cause, although there are exceptions, and the relationship between seizure and impaired myocardial perfusion is complex.
One factor shown to be significantly associated with a cardiac cause for episodic syncope is family history of sudden cardiac death.4
Physical exam: A thorough general and neurologic exam is critical in evaluation of a first syncopal episode. Supine and standing blood pressure and heart rate should be measured, waiting at least one minute (and by some protocols up to three minutes) after the patient stands up to record standing vitals. Measurement of sitting vitals is not necessary. Neither postural hypotension nor tachycardia are sensitive for hypovolemia in normal volunteers, but a 30-point increase in heart rate is more than 99% specific; a 20-point drop in systolic blood pressure (SBP) is less so.5 A difference of more than 20 mmHg between systolic arm pressures in an individual with chest pain suggests aortic dissection.
Other findings useful in diagnosis of syncope include signs of aortic stenosis, pulmonary hypertension, and myxoma. Absent aortic component of S2, a late-peaking or prolonged murmur, sustained apical impulse, and delayed carotid upstroke (“pulsus tardus”) strongly support the presence of severe aortic stenosis. Absence of a palpable pulmonic component of S2 argues strongly against significant pulmonary hypertension.
There are few physical exam findings reliably seen in pulmonary embolism, but the presence of tachycardia is reliably seen about 80% of the time.4 Although atrial myxoma is associated with a characteristic “tumor plop” heard in early diastole, the finding is not common.5
Diagnostic studies: In a recent observational study of more than 2,000 adults older than 65 hospitalized after a syncopal episode, cardiac enzymes, electroencephalography, CT scan of the head, and carotid ultrasonography determined the etiology of syncope in less than 1% of cases. Inpatient monitoring on telemetry was helpful about 5% of the time. On the other hand, postural vital signs contributed to the diagnosis more than 20% of the time, at a fraction of the cost of these other diagnostics (see Table 2, above).6
The role of transthoracic echocardiogram (echo) is more controversial. It does seem clear, however, that echo is more likely to be diagnostic in adults older than 60, in the presence of a heart murmur on physical exam or with an abnormal ECG.7 Although it demonstrated an arrhythmia that determined the etiology of syncope in only 3% of cases in this study, ECG is still routinely recommended; it is relatively inexpensive, risk-free, and can reveal abnormalities, including bundle-branch block, prior MI, and nonsustained ventricular tachycardia, which may be associated with cardiovascular comorbidities.4
Although the diagnostic tests above are of limited utility in uncovering the cause of syncopal episodes, they may be helpful when history or physical examination suggests a specific cause. For example, in individuals presenting with syncope who have lateralizing neurologic signs or symptoms or carotid bruits, carotid ultrasound is an appropriate diagnostic tool. In a retrospective analysis of 140 older adults who underwent carotid ultrasonography as part of a syncope evaluation, carotid lesions were identified in just 2% of subjects.8 These lesions were not thought to have been the primary etiology of syncope but did prompt additional evaluation or intervention.
Among older individuals or individuals with known heart disease, longer-term cardiac monitoring might be appropriate. The method of cardiac monitoring should be guided by the frequency of episodes. For events occurring daily, Holter monitoring is appropriate. For events occurring at least monthly, an event monitor is appropriate.
For less frequent events, the clinician can consider an implantable loop recorder (ILR).3 In a study of 167 individuals without a clear cause of syncope after initial evaluation, diagnosis was achieved in 90% of patients after one year of monitoring by ILR.9
Among individuals in whom the etiology remains unclear, tilt-table testing is often considered. This modality remains controversial and is unlikely to establish a diagnosis in individuals with an otherwise normal evaluation.3 Electrophysiologic testing is of similarly low yield in individuals with otherwise normal evaluation and is generally not recommended, except in individuals with known heart disease, including history of MI, congestive heart failure (CHF), and pre-excitation.10
Diagnostic algorithms: Algorithm-driven diagnostic protocols for evaluation of syncope do exist, but they are generally based on expert consensus opinion rather than large-scale studies. There are evidence-based syncope risk scores under development, but definitive validation is forthcoming. Examination of two such protocols is provided here.
The San Francisco Syncope Rule is among the most well-known algorithms, and predicts adverse outcomes at seven days. The study cohort included 684 patients presenting with syncope to an academic ED. Adverse outcomes, including death, myocardial infarction, arrhythmia, pulmonary embolus, stroke, subarachnoid hemorrhage, ED return, or hospitalization at seven days, were identified. History of CHF, hematocrit less than 30%, ECG abnormality, shortness of breath, and SBP less than 90 mmHg at presentation were associated with increased risk of an adverse outcome. If any of these findings is present, a patient is considered at high risk for adverse outcome at one week.2 The rule is simple to use; however, external validation has been controversial.
In another risk-prediction study—a large multicenter study of patients older than 60—age greater than 90 years, male sex, history of arrhythmia, SBP greater than 160 mm Hg, ECG abnormality, and elevated troponin I were used to construct a score for risk stratification.11 Specific ECG abnormalities included nonsinus rhythm, heart rate less than 40 beats per minute, evidence of acute or chronic ischemic heart disease, prolonged QRS or QT, left or right ventricular hypertrophy, left-axis deviation, and bundle-branch block. Notably, in this older cohort, CHF (specifically, systolic dysfunction with ejection fraction less than 40%) was not significantly associated with risk of adverse event at 30 days. Study authors stratified participants into low- (score ≤0), intermediate- (score 1-2), and high-risk groups (score >2), with 30-day risk of an adverse event ranging from 2.5% to 20%.
One caveat to the interpretation of these data is the fact that even in the “low risk” group, risk of adverse event was still 2.5%, a figure that many clinicians might consider intolerably high.11 This risk score has not been externally validated.
Back to the Case
Our patient was admitted to the inpatient medicine service. She was monitored overnight on telemetry without evidence of arrhythmia. Collateral history revealed new use of multiple antihypertensives prescribed by outside providers, including both atenolol and propranolol. Her subdural hematoma was managed conservatively and she remained free of neurologic deficits. On discharge, her hypertension regimen was simplified. She was referred for outpatient stress echocardiogram.
Bottom Line
Detailed history and physical exam, including postural vital signs, should form the backbone of the routine evaluation of syncope. An ECG is a critical—and inexpensive—initial diagnostic test, while inpatient telemetry, although a routine component of inpatient evaluation, is expensive and relatively low-yield. Risk prediction rules might ultimately help guide admission decisions and inpatient workup, but definitive external validation of these rules has yet to be accomplished. TH
Dr. Wander is a resident in the Department of Medicine at the University of Washington School of Medicine in Seattle. Dr. Best is an assistant professor of medicine in the Division of General Internal Medicine at the University of Washington School of Medicine.
References
- Jhanjee R, Can I, Benditt DG. Syncope. Dis Mon. 2009;55(9):532-585.
- Quinn JV, Stiell IG, McDermott DA, Sellers KL, Kohn MA, Wells GA. Derivation of the San Francisco Syncope Rule to predict patients with short-term serious outcomes. Ann Emerg Med. 2004;43(2):224-232.
- Strickberger SA, Benson DW, Biaggioni I, et al. AHA/ACCF Scientific Statement on the evaluation of syncope: from the American Heart Association Councils on Clinical Cardiology, Cardiovascular Nursing, Cardiovascular Disease in the Young, and Stroke, and the Quality of Care and Outcomes Research Interdisciplinary Working Group; and the American College of Cardiology Foundation: in collaboration with the Heart Rhythm Society: endorsed by the American Autonomic Society. Circulation. 2006;113(2):316-327.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 1: Value of history, physical examination, and electrocardiography. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;126(12):989-996.
- McGee S. Evidence-Based Physical Diagnosis. Saunders; 2001.
- Mendu ML, McAvay G, Lampert R, Stoehr J, Tinetti ME. Yield of diagnostic tests in evaluating syncopal episodes in older patients. Arch Intern Med. 2009;169(14):1299-1305.
- Panther R, Mahmood S, Gal R. Echocardiography in the diagnostic evaluation of syncope. J Am Soc Echocardiogr. 1998;11(3):294-298.
- Schnipper JL, Ackerman RH, Krier JB, Honour M. Diagnostic yield and utility of neurovascular ultrasonography in the evaluation of patients with syncope. Mayo Clin Proc. 2005;80(4):480-488.
- Assar MD, Krahn AD, Klein GJ, Yee R, Skanes AC. Optimal duration of monitoring in patients with unexplained syncope. Am J Cardiol. 2003;92(10):1231-1233.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 2: Unexplained syncope. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;127(1):76-86.
- Sun BC, Derose SF, Liang LJ, et al. Predictors of 30-day serious events in older patients with syncope. Ann Emerg Med. 2009;54(6):769-778.e1-5.
Case
A 71-year-old woman is admitted after losing consciousness and falling at home. Her history is significant for hypertension, bilateral internal carotid artery stenoses, chronic kidney disease, and diabetes. Resting vital signs are normal. Cardiac, pulmonary, and neurologic exams are unremarkable, as is an electrocardiogram (ECG). She was noted to have a small scalp laceration. Noncontrast CT of the head demonstrates a small occipital subdural bleed thought to be a result of her fall.
What is the most cost-effective evaluation for this patient admitted with suspected first syncopal episode?
Overview
Syncope is defined as sudden, self-terminating loss of consciousness. The final common pathway of all causes of syncope is global cerebral hypoperfusion—specifically, hypoperfusion of the reticular activating system. The differential diagnosis of syncopal loss of consciousness includes neurally mediated (e.g., vasovagal) syncope, orthostatic hypotension, cardiac arrhythmias, structural heart disease, and cerebrovascular disease.
Among young, otherwise healthy people, neurally mediated syncope, which has a relatively benign prognosis, is by far the most common etiology, while in older patients, primary cardiac causes are more common. Nonsyncopal mechanisms, such as seizure and hypoglycemia, should also be considered in the differential diagnosis of transient loss of consciousness (see Table 1, p. 19).1
Syncope is a common problem, accounting for 1% to 2% of ED visits in the U.S.2 The primary objective for evaluation is identification of individuals at increased risk of death due to associated conditions, especially cardiac conditions such as structural heart disease; myocardial ischemia and infarction (MI); Wolff-Parkinson-White, Brugada, or long QT syndromes; and polymorphic ventricular tachycardia.3 True syncope can be associated with other concerning causes, such as aortic stenosis, aortic dissection, and massive pulmonary embolus, as well as arrhythmias from underlying cardiac disease.4
Review of the Data
History: A detailed history and physical examination reveals the cause in 50% of syncopal episodes. Key factors include the account of third-party observers, although it is important to note that tonic-clonic movements can be associated with the global cerebral hypoperfusion of syncope as well as with seizure.4 History of dyspnea, chest pain, or palpitations argue for a primary cardiac or pulmonary cause.
Among patients who endorse a history of palpitations, the sensation of rapid and regular pulsations in the neck points very strongly toward an AV-nodal re-entrant tachycardia, because the carotid pulse and cannon A wave arrive simultaneously in the neck.5 Postictal confusion and focal neurologic exam findings suggest a neurologic rather than syncopal cause, although there are exceptions, and the relationship between seizure and impaired myocardial perfusion is complex.
One factor shown to be significantly associated with a cardiac cause for episodic syncope is family history of sudden cardiac death.4
Physical exam: A thorough general and neurologic exam is critical in evaluation of a first syncopal episode. Supine and standing blood pressure and heart rate should be measured, waiting at least one minute (and by some protocols up to three minutes) after the patient stands up to record standing vitals. Measurement of sitting vitals is not necessary. Neither postural hypotension nor tachycardia are sensitive for hypovolemia in normal volunteers, but a 30-point increase in heart rate is more than 99% specific; a 20-point drop in systolic blood pressure (SBP) is less so.5 A difference of more than 20 mmHg between systolic arm pressures in an individual with chest pain suggests aortic dissection.
Other findings useful in diagnosis of syncope include signs of aortic stenosis, pulmonary hypertension, and myxoma. Absent aortic component of S2, a late-peaking or prolonged murmur, sustained apical impulse, and delayed carotid upstroke (“pulsus tardus”) strongly support the presence of severe aortic stenosis. Absence of a palpable pulmonic component of S2 argues strongly against significant pulmonary hypertension.
There are few physical exam findings reliably seen in pulmonary embolism, but the presence of tachycardia is reliably seen about 80% of the time.4 Although atrial myxoma is associated with a characteristic “tumor plop” heard in early diastole, the finding is not common.5
Diagnostic studies: In a recent observational study of more than 2,000 adults older than 65 hospitalized after a syncopal episode, cardiac enzymes, electroencephalography, CT scan of the head, and carotid ultrasonography determined the etiology of syncope in less than 1% of cases. Inpatient monitoring on telemetry was helpful about 5% of the time. On the other hand, postural vital signs contributed to the diagnosis more than 20% of the time, at a fraction of the cost of these other diagnostics (see Table 2, above).6
The role of transthoracic echocardiogram (echo) is more controversial. It does seem clear, however, that echo is more likely to be diagnostic in adults older than 60, in the presence of a heart murmur on physical exam or with an abnormal ECG.7 Although it demonstrated an arrhythmia that determined the etiology of syncope in only 3% of cases in this study, ECG is still routinely recommended; it is relatively inexpensive, risk-free, and can reveal abnormalities, including bundle-branch block, prior MI, and nonsustained ventricular tachycardia, which may be associated with cardiovascular comorbidities.4
Although the diagnostic tests above are of limited utility in uncovering the cause of syncopal episodes, they may be helpful when history or physical examination suggests a specific cause. For example, in individuals presenting with syncope who have lateralizing neurologic signs or symptoms or carotid bruits, carotid ultrasound is an appropriate diagnostic tool. In a retrospective analysis of 140 older adults who underwent carotid ultrasonography as part of a syncope evaluation, carotid lesions were identified in just 2% of subjects.8 These lesions were not thought to have been the primary etiology of syncope but did prompt additional evaluation or intervention.
Among older individuals or individuals with known heart disease, longer-term cardiac monitoring might be appropriate. The method of cardiac monitoring should be guided by the frequency of episodes. For events occurring daily, Holter monitoring is appropriate. For events occurring at least monthly, an event monitor is appropriate.
For less frequent events, the clinician can consider an implantable loop recorder (ILR).3 In a study of 167 individuals without a clear cause of syncope after initial evaluation, diagnosis was achieved in 90% of patients after one year of monitoring by ILR.9
Among individuals in whom the etiology remains unclear, tilt-table testing is often considered. This modality remains controversial and is unlikely to establish a diagnosis in individuals with an otherwise normal evaluation.3 Electrophysiologic testing is of similarly low yield in individuals with otherwise normal evaluation and is generally not recommended, except in individuals with known heart disease, including history of MI, congestive heart failure (CHF), and pre-excitation.10
Diagnostic algorithms: Algorithm-driven diagnostic protocols for evaluation of syncope do exist, but they are generally based on expert consensus opinion rather than large-scale studies. There are evidence-based syncope risk scores under development, but definitive validation is forthcoming. Examination of two such protocols is provided here.
The San Francisco Syncope Rule is among the most well-known algorithms, and predicts adverse outcomes at seven days. The study cohort included 684 patients presenting with syncope to an academic ED. Adverse outcomes, including death, myocardial infarction, arrhythmia, pulmonary embolus, stroke, subarachnoid hemorrhage, ED return, or hospitalization at seven days, were identified. History of CHF, hematocrit less than 30%, ECG abnormality, shortness of breath, and SBP less than 90 mmHg at presentation were associated with increased risk of an adverse outcome. If any of these findings is present, a patient is considered at high risk for adverse outcome at one week.2 The rule is simple to use; however, external validation has been controversial.
In another risk-prediction study—a large multicenter study of patients older than 60—age greater than 90 years, male sex, history of arrhythmia, SBP greater than 160 mm Hg, ECG abnormality, and elevated troponin I were used to construct a score for risk stratification.11 Specific ECG abnormalities included nonsinus rhythm, heart rate less than 40 beats per minute, evidence of acute or chronic ischemic heart disease, prolonged QRS or QT, left or right ventricular hypertrophy, left-axis deviation, and bundle-branch block. Notably, in this older cohort, CHF (specifically, systolic dysfunction with ejection fraction less than 40%) was not significantly associated with risk of adverse event at 30 days. Study authors stratified participants into low- (score ≤0), intermediate- (score 1-2), and high-risk groups (score >2), with 30-day risk of an adverse event ranging from 2.5% to 20%.
One caveat to the interpretation of these data is the fact that even in the “low risk” group, risk of adverse event was still 2.5%, a figure that many clinicians might consider intolerably high.11 This risk score has not been externally validated.
Back to the Case
Our patient was admitted to the inpatient medicine service. She was monitored overnight on telemetry without evidence of arrhythmia. Collateral history revealed new use of multiple antihypertensives prescribed by outside providers, including both atenolol and propranolol. Her subdural hematoma was managed conservatively and she remained free of neurologic deficits. On discharge, her hypertension regimen was simplified. She was referred for outpatient stress echocardiogram.
Bottom Line
Detailed history and physical exam, including postural vital signs, should form the backbone of the routine evaluation of syncope. An ECG is a critical—and inexpensive—initial diagnostic test, while inpatient telemetry, although a routine component of inpatient evaluation, is expensive and relatively low-yield. Risk prediction rules might ultimately help guide admission decisions and inpatient workup, but definitive external validation of these rules has yet to be accomplished. TH
Dr. Wander is a resident in the Department of Medicine at the University of Washington School of Medicine in Seattle. Dr. Best is an assistant professor of medicine in the Division of General Internal Medicine at the University of Washington School of Medicine.
References
- Jhanjee R, Can I, Benditt DG. Syncope. Dis Mon. 2009;55(9):532-585.
- Quinn JV, Stiell IG, McDermott DA, Sellers KL, Kohn MA, Wells GA. Derivation of the San Francisco Syncope Rule to predict patients with short-term serious outcomes. Ann Emerg Med. 2004;43(2):224-232.
- Strickberger SA, Benson DW, Biaggioni I, et al. AHA/ACCF Scientific Statement on the evaluation of syncope: from the American Heart Association Councils on Clinical Cardiology, Cardiovascular Nursing, Cardiovascular Disease in the Young, and Stroke, and the Quality of Care and Outcomes Research Interdisciplinary Working Group; and the American College of Cardiology Foundation: in collaboration with the Heart Rhythm Society: endorsed by the American Autonomic Society. Circulation. 2006;113(2):316-327.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 1: Value of history, physical examination, and electrocardiography. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;126(12):989-996.
- McGee S. Evidence-Based Physical Diagnosis. Saunders; 2001.
- Mendu ML, McAvay G, Lampert R, Stoehr J, Tinetti ME. Yield of diagnostic tests in evaluating syncopal episodes in older patients. Arch Intern Med. 2009;169(14):1299-1305.
- Panther R, Mahmood S, Gal R. Echocardiography in the diagnostic evaluation of syncope. J Am Soc Echocardiogr. 1998;11(3):294-298.
- Schnipper JL, Ackerman RH, Krier JB, Honour M. Diagnostic yield and utility of neurovascular ultrasonography in the evaluation of patients with syncope. Mayo Clin Proc. 2005;80(4):480-488.
- Assar MD, Krahn AD, Klein GJ, Yee R, Skanes AC. Optimal duration of monitoring in patients with unexplained syncope. Am J Cardiol. 2003;92(10):1231-1233.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 2: Unexplained syncope. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;127(1):76-86.
- Sun BC, Derose SF, Liang LJ, et al. Predictors of 30-day serious events in older patients with syncope. Ann Emerg Med. 2009;54(6):769-778.e1-5.
Case
A 71-year-old woman is admitted after losing consciousness and falling at home. Her history is significant for hypertension, bilateral internal carotid artery stenoses, chronic kidney disease, and diabetes. Resting vital signs are normal. Cardiac, pulmonary, and neurologic exams are unremarkable, as is an electrocardiogram (ECG). She was noted to have a small scalp laceration. Noncontrast CT of the head demonstrates a small occipital subdural bleed thought to be a result of her fall.
What is the most cost-effective evaluation for this patient admitted with suspected first syncopal episode?
Overview
Syncope is defined as sudden, self-terminating loss of consciousness. The final common pathway of all causes of syncope is global cerebral hypoperfusion—specifically, hypoperfusion of the reticular activating system. The differential diagnosis of syncopal loss of consciousness includes neurally mediated (e.g., vasovagal) syncope, orthostatic hypotension, cardiac arrhythmias, structural heart disease, and cerebrovascular disease.
Among young, otherwise healthy people, neurally mediated syncope, which has a relatively benign prognosis, is by far the most common etiology, while in older patients, primary cardiac causes are more common. Nonsyncopal mechanisms, such as seizure and hypoglycemia, should also be considered in the differential diagnosis of transient loss of consciousness (see Table 1, p. 19).1
Syncope is a common problem, accounting for 1% to 2% of ED visits in the U.S.2 The primary objective for evaluation is identification of individuals at increased risk of death due to associated conditions, especially cardiac conditions such as structural heart disease; myocardial ischemia and infarction (MI); Wolff-Parkinson-White, Brugada, or long QT syndromes; and polymorphic ventricular tachycardia.3 True syncope can be associated with other concerning causes, such as aortic stenosis, aortic dissection, and massive pulmonary embolus, as well as arrhythmias from underlying cardiac disease.4
Review of the Data
History: A detailed history and physical examination reveals the cause in 50% of syncopal episodes. Key factors include the account of third-party observers, although it is important to note that tonic-clonic movements can be associated with the global cerebral hypoperfusion of syncope as well as with seizure.4 History of dyspnea, chest pain, or palpitations argue for a primary cardiac or pulmonary cause.
Among patients who endorse a history of palpitations, the sensation of rapid and regular pulsations in the neck points very strongly toward an AV-nodal re-entrant tachycardia, because the carotid pulse and cannon A wave arrive simultaneously in the neck.5 Postictal confusion and focal neurologic exam findings suggest a neurologic rather than syncopal cause, although there are exceptions, and the relationship between seizure and impaired myocardial perfusion is complex.
One factor shown to be significantly associated with a cardiac cause for episodic syncope is family history of sudden cardiac death.4
Physical exam: A thorough general and neurologic exam is critical in evaluation of a first syncopal episode. Supine and standing blood pressure and heart rate should be measured, waiting at least one minute (and by some protocols up to three minutes) after the patient stands up to record standing vitals. Measurement of sitting vitals is not necessary. Neither postural hypotension nor tachycardia are sensitive for hypovolemia in normal volunteers, but a 30-point increase in heart rate is more than 99% specific; a 20-point drop in systolic blood pressure (SBP) is less so.5 A difference of more than 20 mmHg between systolic arm pressures in an individual with chest pain suggests aortic dissection.
Other findings useful in diagnosis of syncope include signs of aortic stenosis, pulmonary hypertension, and myxoma. Absent aortic component of S2, a late-peaking or prolonged murmur, sustained apical impulse, and delayed carotid upstroke (“pulsus tardus”) strongly support the presence of severe aortic stenosis. Absence of a palpable pulmonic component of S2 argues strongly against significant pulmonary hypertension.
There are few physical exam findings reliably seen in pulmonary embolism, but the presence of tachycardia is reliably seen about 80% of the time.4 Although atrial myxoma is associated with a characteristic “tumor plop” heard in early diastole, the finding is not common.5
Diagnostic studies: In a recent observational study of more than 2,000 adults older than 65 hospitalized after a syncopal episode, cardiac enzymes, electroencephalography, CT scan of the head, and carotid ultrasonography determined the etiology of syncope in less than 1% of cases. Inpatient monitoring on telemetry was helpful about 5% of the time. On the other hand, postural vital signs contributed to the diagnosis more than 20% of the time, at a fraction of the cost of these other diagnostics (see Table 2, above).6
The role of transthoracic echocardiogram (echo) is more controversial. It does seem clear, however, that echo is more likely to be diagnostic in adults older than 60, in the presence of a heart murmur on physical exam or with an abnormal ECG.7 Although it demonstrated an arrhythmia that determined the etiology of syncope in only 3% of cases in this study, ECG is still routinely recommended; it is relatively inexpensive, risk-free, and can reveal abnormalities, including bundle-branch block, prior MI, and nonsustained ventricular tachycardia, which may be associated with cardiovascular comorbidities.4
Although the diagnostic tests above are of limited utility in uncovering the cause of syncopal episodes, they may be helpful when history or physical examination suggests a specific cause. For example, in individuals presenting with syncope who have lateralizing neurologic signs or symptoms or carotid bruits, carotid ultrasound is an appropriate diagnostic tool. In a retrospective analysis of 140 older adults who underwent carotid ultrasonography as part of a syncope evaluation, carotid lesions were identified in just 2% of subjects.8 These lesions were not thought to have been the primary etiology of syncope but did prompt additional evaluation or intervention.
Among older individuals or individuals with known heart disease, longer-term cardiac monitoring might be appropriate. The method of cardiac monitoring should be guided by the frequency of episodes. For events occurring daily, Holter monitoring is appropriate. For events occurring at least monthly, an event monitor is appropriate.
For less frequent events, the clinician can consider an implantable loop recorder (ILR).3 In a study of 167 individuals without a clear cause of syncope after initial evaluation, diagnosis was achieved in 90% of patients after one year of monitoring by ILR.9
Among individuals in whom the etiology remains unclear, tilt-table testing is often considered. This modality remains controversial and is unlikely to establish a diagnosis in individuals with an otherwise normal evaluation.3 Electrophysiologic testing is of similarly low yield in individuals with otherwise normal evaluation and is generally not recommended, except in individuals with known heart disease, including history of MI, congestive heart failure (CHF), and pre-excitation.10
Diagnostic algorithms: Algorithm-driven diagnostic protocols for evaluation of syncope do exist, but they are generally based on expert consensus opinion rather than large-scale studies. There are evidence-based syncope risk scores under development, but definitive validation is forthcoming. Examination of two such protocols is provided here.
The San Francisco Syncope Rule is among the most well-known algorithms, and predicts adverse outcomes at seven days. The study cohort included 684 patients presenting with syncope to an academic ED. Adverse outcomes, including death, myocardial infarction, arrhythmia, pulmonary embolus, stroke, subarachnoid hemorrhage, ED return, or hospitalization at seven days, were identified. History of CHF, hematocrit less than 30%, ECG abnormality, shortness of breath, and SBP less than 90 mmHg at presentation were associated with increased risk of an adverse outcome. If any of these findings is present, a patient is considered at high risk for adverse outcome at one week.2 The rule is simple to use; however, external validation has been controversial.
In another risk-prediction study—a large multicenter study of patients older than 60—age greater than 90 years, male sex, history of arrhythmia, SBP greater than 160 mm Hg, ECG abnormality, and elevated troponin I were used to construct a score for risk stratification.11 Specific ECG abnormalities included nonsinus rhythm, heart rate less than 40 beats per minute, evidence of acute or chronic ischemic heart disease, prolonged QRS or QT, left or right ventricular hypertrophy, left-axis deviation, and bundle-branch block. Notably, in this older cohort, CHF (specifically, systolic dysfunction with ejection fraction less than 40%) was not significantly associated with risk of adverse event at 30 days. Study authors stratified participants into low- (score ≤0), intermediate- (score 1-2), and high-risk groups (score >2), with 30-day risk of an adverse event ranging from 2.5% to 20%.
One caveat to the interpretation of these data is the fact that even in the “low risk” group, risk of adverse event was still 2.5%, a figure that many clinicians might consider intolerably high.11 This risk score has not been externally validated.
Back to the Case
Our patient was admitted to the inpatient medicine service. She was monitored overnight on telemetry without evidence of arrhythmia. Collateral history revealed new use of multiple antihypertensives prescribed by outside providers, including both atenolol and propranolol. Her subdural hematoma was managed conservatively and she remained free of neurologic deficits. On discharge, her hypertension regimen was simplified. She was referred for outpatient stress echocardiogram.
Bottom Line
Detailed history and physical exam, including postural vital signs, should form the backbone of the routine evaluation of syncope. An ECG is a critical—and inexpensive—initial diagnostic test, while inpatient telemetry, although a routine component of inpatient evaluation, is expensive and relatively low-yield. Risk prediction rules might ultimately help guide admission decisions and inpatient workup, but definitive external validation of these rules has yet to be accomplished. TH
Dr. Wander is a resident in the Department of Medicine at the University of Washington School of Medicine in Seattle. Dr. Best is an assistant professor of medicine in the Division of General Internal Medicine at the University of Washington School of Medicine.
References
- Jhanjee R, Can I, Benditt DG. Syncope. Dis Mon. 2009;55(9):532-585.
- Quinn JV, Stiell IG, McDermott DA, Sellers KL, Kohn MA, Wells GA. Derivation of the San Francisco Syncope Rule to predict patients with short-term serious outcomes. Ann Emerg Med. 2004;43(2):224-232.
- Strickberger SA, Benson DW, Biaggioni I, et al. AHA/ACCF Scientific Statement on the evaluation of syncope: from the American Heart Association Councils on Clinical Cardiology, Cardiovascular Nursing, Cardiovascular Disease in the Young, and Stroke, and the Quality of Care and Outcomes Research Interdisciplinary Working Group; and the American College of Cardiology Foundation: in collaboration with the Heart Rhythm Society: endorsed by the American Autonomic Society. Circulation. 2006;113(2):316-327.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 1: Value of history, physical examination, and electrocardiography. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;126(12):989-996.
- McGee S. Evidence-Based Physical Diagnosis. Saunders; 2001.
- Mendu ML, McAvay G, Lampert R, Stoehr J, Tinetti ME. Yield of diagnostic tests in evaluating syncopal episodes in older patients. Arch Intern Med. 2009;169(14):1299-1305.
- Panther R, Mahmood S, Gal R. Echocardiography in the diagnostic evaluation of syncope. J Am Soc Echocardiogr. 1998;11(3):294-298.
- Schnipper JL, Ackerman RH, Krier JB, Honour M. Diagnostic yield and utility of neurovascular ultrasonography in the evaluation of patients with syncope. Mayo Clin Proc. 2005;80(4):480-488.
- Assar MD, Krahn AD, Klein GJ, Yee R, Skanes AC. Optimal duration of monitoring in patients with unexplained syncope. Am J Cardiol. 2003;92(10):1231-1233.
- Linzer M, Yang EH, Estes NA 3rd, Wang P, Vorperian VR, Kapoor WN. Diagnosing syncope. Part 2: Unexplained syncope. Clinical Efficacy Assessment Project of the American College of Physicians. Ann Intern Med. 1997;127(1):76-86.
- Sun BC, Derose SF, Liang LJ, et al. Predictors of 30-day serious events in older patients with syncope. Ann Emerg Med. 2009;54(6):769-778.e1-5.
Rule Proposes Electronic Prescription of Controlled Substances, Doesn’t Scrap Pen-and-Paper Method
Is it true that the Drug Enforcement Administration (DEA) is going to allow doctors to prescribe controlled drugs electronically?
Will I still be able to prescribe on my prescription pads, or is this big government forcing me to use a computer for prescriptions?
J. Hockenstein, DO
Des Moines, Iowa
Dr. Hospitalist responds: On March 31, the DEA published in the Federal Register an interim final rule regarding the “electronic prescription for controlled substances.” (View the entire rule at www.gpoaccess.gov/fr.) The DEA is seeking comment on the proposed rule for the next 60 days. Some of us might remember that the DEA proposed a similar rule for electronic prescribing in June 2008, but that rule did not meet the security requirements already in place at federal healthcare facilities.
Under the current system, providers can create prescriptions electronically, but the prescription has to be printed on paper. The new rule proposes a system of true electronic prescribing; data can be transmitted electronically from the hospital or doctor’s office to the pharmacy without the use of a printer or fax.
This proposed rule does not eliminate the traditional method of paper and pen for prescriptions but allows providers the voluntary option of prescribing controlled substances electronically. This proposed rule also allows pharmacies to receive, dispense, and archive these electronic prescriptions.
For those providers who choose to prescribe electronically, there will be specific requirements to prevent diversion and maintain privacy. Providers must utilize software that meets the rule’s specific requirements. For example, the software system will require a two-step process to authenticate the prescribing provider. These measures might include a password, a token, or the use of biometric identifier (e.g., fingerprint or handprint). For some of us, this might sound space-aged, but such biometric systems are commonplace in other industries. For example, I provided my fingerprint as part of the test center security system when I checked in for my American Board of Internal Medicine (ABIM) recertification examination.
There are several other issues with the proposed rule that one should consider. The new proposal does not affect the existing rule regarding emergency prescriptions. The current law allows physicians to prescribe a Schedule II controlled substance by telephone and the pharmacist to dispense this substance, provided that the amount being dispensed is limited to what is reasonably required during the emergency time period and that the provider provides a hard copy of the prescription to the pharmacist within seven days of the telephone prescription. Under the proposed rule, providers will still be able to prescribe Schedule II substances by telephone under emergency situations but will have the option of providing an electronic copy of the prescription, rather than a paper one, within seven days.
There are other components of the proposed rule that could change your practice. The rule clearly states that an electronic prescription cannot be changed after transmission and that any change to the content of the prescription will render it invalid. This might be important in a handful of situations. For example, if the provider electronically prescribes a brand-name drug, the pharmacist would not be able to make a generic substitution.
Another component of the proposed rule is that it precludes the printing of an electronic prescription, which already has been transmitted and precludes the electronic transmission of a prescription that already has been printed. This situation might arise if the electronic prescription did not transmit due to a computer problem. The provider would not be able to print or fax a copy of the electronic prescription.
The proposed rule has the potential to reduce medical errors, reduce prescription forgeries, and help providers and hospitals integrate their medical records. True electronic prescribing is long overdue. In the future, I envision hospitalists prescribing from their handheld devices.
The key to success, like any computerized system, will be the ability to keep the system running and continuously maintaining and upgrading security measures. For more information regarding electronic prescriptions for controlled substances, visit www.DEAdiversion.usdoj.gov. TH
Is it true that the Drug Enforcement Administration (DEA) is going to allow doctors to prescribe controlled drugs electronically?
Will I still be able to prescribe on my prescription pads, or is this big government forcing me to use a computer for prescriptions?
J. Hockenstein, DO
Des Moines, Iowa
Dr. Hospitalist responds: On March 31, the DEA published in the Federal Register an interim final rule regarding the “electronic prescription for controlled substances.” (View the entire rule at www.gpoaccess.gov/fr.) The DEA is seeking comment on the proposed rule for the next 60 days. Some of us might remember that the DEA proposed a similar rule for electronic prescribing in June 2008, but that rule did not meet the security requirements already in place at federal healthcare facilities.
Under the current system, providers can create prescriptions electronically, but the prescription has to be printed on paper. The new rule proposes a system of true electronic prescribing; data can be transmitted electronically from the hospital or doctor’s office to the pharmacy without the use of a printer or fax.
This proposed rule does not eliminate the traditional method of paper and pen for prescriptions but allows providers the voluntary option of prescribing controlled substances electronically. This proposed rule also allows pharmacies to receive, dispense, and archive these electronic prescriptions.
For those providers who choose to prescribe electronically, there will be specific requirements to prevent diversion and maintain privacy. Providers must utilize software that meets the rule’s specific requirements. For example, the software system will require a two-step process to authenticate the prescribing provider. These measures might include a password, a token, or the use of biometric identifier (e.g., fingerprint or handprint). For some of us, this might sound space-aged, but such biometric systems are commonplace in other industries. For example, I provided my fingerprint as part of the test center security system when I checked in for my American Board of Internal Medicine (ABIM) recertification examination.
There are several other issues with the proposed rule that one should consider. The new proposal does not affect the existing rule regarding emergency prescriptions. The current law allows physicians to prescribe a Schedule II controlled substance by telephone and the pharmacist to dispense this substance, provided that the amount being dispensed is limited to what is reasonably required during the emergency time period and that the provider provides a hard copy of the prescription to the pharmacist within seven days of the telephone prescription. Under the proposed rule, providers will still be able to prescribe Schedule II substances by telephone under emergency situations but will have the option of providing an electronic copy of the prescription, rather than a paper one, within seven days.
There are other components of the proposed rule that could change your practice. The rule clearly states that an electronic prescription cannot be changed after transmission and that any change to the content of the prescription will render it invalid. This might be important in a handful of situations. For example, if the provider electronically prescribes a brand-name drug, the pharmacist would not be able to make a generic substitution.
Another component of the proposed rule is that it precludes the printing of an electronic prescription, which already has been transmitted and precludes the electronic transmission of a prescription that already has been printed. This situation might arise if the electronic prescription did not transmit due to a computer problem. The provider would not be able to print or fax a copy of the electronic prescription.
The proposed rule has the potential to reduce medical errors, reduce prescription forgeries, and help providers and hospitals integrate their medical records. True electronic prescribing is long overdue. In the future, I envision hospitalists prescribing from their handheld devices.
The key to success, like any computerized system, will be the ability to keep the system running and continuously maintaining and upgrading security measures. For more information regarding electronic prescriptions for controlled substances, visit www.DEAdiversion.usdoj.gov. TH
Is it true that the Drug Enforcement Administration (DEA) is going to allow doctors to prescribe controlled drugs electronically?
Will I still be able to prescribe on my prescription pads, or is this big government forcing me to use a computer for prescriptions?
J. Hockenstein, DO
Des Moines, Iowa
Dr. Hospitalist responds: On March 31, the DEA published in the Federal Register an interim final rule regarding the “electronic prescription for controlled substances.” (View the entire rule at www.gpoaccess.gov/fr.) The DEA is seeking comment on the proposed rule for the next 60 days. Some of us might remember that the DEA proposed a similar rule for electronic prescribing in June 2008, but that rule did not meet the security requirements already in place at federal healthcare facilities.
Under the current system, providers can create prescriptions electronically, but the prescription has to be printed on paper. The new rule proposes a system of true electronic prescribing; data can be transmitted electronically from the hospital or doctor’s office to the pharmacy without the use of a printer or fax.
This proposed rule does not eliminate the traditional method of paper and pen for prescriptions but allows providers the voluntary option of prescribing controlled substances electronically. This proposed rule also allows pharmacies to receive, dispense, and archive these electronic prescriptions.
For those providers who choose to prescribe electronically, there will be specific requirements to prevent diversion and maintain privacy. Providers must utilize software that meets the rule’s specific requirements. For example, the software system will require a two-step process to authenticate the prescribing provider. These measures might include a password, a token, or the use of biometric identifier (e.g., fingerprint or handprint). For some of us, this might sound space-aged, but such biometric systems are commonplace in other industries. For example, I provided my fingerprint as part of the test center security system when I checked in for my American Board of Internal Medicine (ABIM) recertification examination.
There are several other issues with the proposed rule that one should consider. The new proposal does not affect the existing rule regarding emergency prescriptions. The current law allows physicians to prescribe a Schedule II controlled substance by telephone and the pharmacist to dispense this substance, provided that the amount being dispensed is limited to what is reasonably required during the emergency time period and that the provider provides a hard copy of the prescription to the pharmacist within seven days of the telephone prescription. Under the proposed rule, providers will still be able to prescribe Schedule II substances by telephone under emergency situations but will have the option of providing an electronic copy of the prescription, rather than a paper one, within seven days.
There are other components of the proposed rule that could change your practice. The rule clearly states that an electronic prescription cannot be changed after transmission and that any change to the content of the prescription will render it invalid. This might be important in a handful of situations. For example, if the provider electronically prescribes a brand-name drug, the pharmacist would not be able to make a generic substitution.
Another component of the proposed rule is that it precludes the printing of an electronic prescription, which already has been transmitted and precludes the electronic transmission of a prescription that already has been printed. This situation might arise if the electronic prescription did not transmit due to a computer problem. The provider would not be able to print or fax a copy of the electronic prescription.
The proposed rule has the potential to reduce medical errors, reduce prescription forgeries, and help providers and hospitals integrate their medical records. True electronic prescribing is long overdue. In the future, I envision hospitalists prescribing from their handheld devices.
The key to success, like any computerized system, will be the ability to keep the system running and continuously maintaining and upgrading security measures. For more information regarding electronic prescriptions for controlled substances, visit www.DEAdiversion.usdoj.gov. TH
Care Critical
There is a brewing crisis in critical-care medicine, a crisis that already is impacting HM. It would be easier for hospitalists to look the other way and say, “This is not our problem.” Make no mistake: It is our problem. There are not enough intensive-care physicians to go around.
In my generation, most physicians who trained in pulmonary and critical-care medicine thought they would do some office care, some bronchoscopies, and some critical care for the first several years of their professional lives. Few imagined that, in their 60s, their practice would still include rescuing patients from death at 2 a.m. Couple the intensity of care with such a demanding lifestyle, and it is hardly surprising that many critical-care doctors seek less-stressful practices (e.g., sleep medicine) or retirement.
New physicians who see the work-life imbalance are shying away from critical care in significant enough numbers, yet the demand for these doctors is growing. Whether that shift is due to the attraction of careers in HM, emergency medicine, or other IM specialties, or the perceived negatives to being a full-time intensivist, is beside the point. The medical workforce needs more critical-care physicians. And we are not going to be able to meet this need by assuming we will be able to recruit and train more surgeons, anesthesiologists, or pulmonary physicians in traditional critical-care pathways.
The situation is further complicated by workforce shortages on the rest of the ICU healthcare team. Many senior ICU nurses are reaching the end of their careers, and the pipeline to replace them is anything but robust. And all this comes at a time when the acuity of hospitalized patients is increasing and the demands for ICU expertise is at its height.
It is no surprise that hospitalists are called upon to fill the critical-care gap. SHM surveys show that 92% of hospitalists include ICU care in their practice. While hospitalists clearly do not have the training or skills to replace the intensivist, we clearly are witnessing a “scope creep.” Hospitalists are being asked to stretch their skills to fill the void in critical care.
Competence Question
The response to this is manifest in many ways. For example, steadily increasing numbers of hospitalists attend SHM’s annual critical-care precourses, and our procedures courses invariably sell out. These brief courses are important to hospitalists and their patients. Yet a day (or even two) of focused training for hospitalists will not raise their skill set to replace or even augment critical-care-trained physicians at their hospitals. The patients will keep coming and continue to need the expertise for their most-acute-care needs. Something must be done.
There are pockets of experiments on filling the increasing critical-care gap. Emory University’s Center for Critical Care in Atlanta will soon launch an experimental, HM-critical-care training program that will attempt to develop and verify critical-care competencies in just one postgraduate year after IM residency. A complementary approach could include a hospitalist-focused track within the three years of IM residency to include less outpatient medicine and more intensive-care training. This could be part of a broader restructuring of internal and family medicine residencies, which recognize the career paths (and needs) of their residents as some enter hospital-focused practice (e.g., as hospitalists, cardiologists, intensivists) and some concentrate more on the patient outside the hospital (e.g., primary care, endocrinology, rheumatology).
As training evolves, there will be practical issues of credentialing. Currently, a general-IM-trained hospitalist is required to complete a two-year fellowship in critical care to be a specialist. Interestingly, if one’s residency training is in surgery, anesthesia, or rheumatology, that physician requires only one additional year in critical care to become eligible for added/special qualifications in critical-care medicine. What, precisely, is the carryover of the longer residency experience that makes it possible to attain competency in critical care in half the training time?
There also is the risk that we will create a workforce that includes the intensivist-lite—someone who does not have complete, recognized training in critical care but has more than the typical hospitalist, and is perceived as “better than having no intensivist.” Is this in the best interest of our patients or our discipline?
Supply Solutions
There are other approaches to the workforce challenge beyond asking hospitalists to step away from practice for an additional year or two of training. As in other aspects of the hospital workforce, it is time to examine alternative deployment of the entire healthcare team. If intensivists and hospitalists are in limited supply, we need to revisit their roles and further look for opportunities to engage acute-care nurse practitioners, physician assistants, RNs, and others on the healthcare team to meet the expanding needs of our patients and our hospitals.
Another strategy would include regionalization of healthcare in population centers with multiple hospitals. In Orange County, Calif., where I live, there are 33 hospitals for 3 million people, each with some form of an ICU. Is it time to set standards of coverage and expertise so that 10 to 15 hospitals can provide a fully staffed ICU, and the other hospitals refer their most-acute patients rather than be stretched to staff their ICUs at a time of workforce shortages? Is it time to do what we did with trauma centers and set various levels of care so that not every hospital can or should be doing the most intense and costly level of care?
SHM, HM, and, most importantly, hospitalists find themselves in the midst of this growing problem. We must be part of the solution.
It is important to recognize the significant variability in the intensivist skill sets that individual hospitalists and HM groups currently possess; the variance creates hurdles in being able to step up and fill the critical-care gap. I’ve heard about hospitalists pausing their practices to obtain additional training in critical care. Hospitals across America are (or soon will be) scrambling to integrate their hospitalists and intensivists to maximize coverage and expertise. SHM has noticed an increased demand on hospitalists to increase knowledge and skills so they can extend the local coverage of critically ill patients.
It is time for SHM to clearly understand how this sea change is affecting you professionally and personally, because you practice on the frontlines of our nation’s hospitals.
It is time for SHM to engage our colleagues in the ICU—critical-care physicians and their professional societies—to understand their perspective and initiatives on this growing crisis.
It is time for SHM to engage medical educators in residency and fellowship training to explore potential changes in the curriculum—changes aimed at young physicians proceeding through their training that yield hospital-based physicians better prepared to enter the hospital environment of the 21st century.
And SHM may need to engage the boards and other credentialing bodies to look for flexibility that will reflect today’s realities, attract the best-trained physicians to care for the most-acutely-ill patients, and protect our patients by demanding expertise and training at the most appropriate levels. TH
Dr. Wellikson is CEO of SHM.
There is a brewing crisis in critical-care medicine, a crisis that already is impacting HM. It would be easier for hospitalists to look the other way and say, “This is not our problem.” Make no mistake: It is our problem. There are not enough intensive-care physicians to go around.
In my generation, most physicians who trained in pulmonary and critical-care medicine thought they would do some office care, some bronchoscopies, and some critical care for the first several years of their professional lives. Few imagined that, in their 60s, their practice would still include rescuing patients from death at 2 a.m. Couple the intensity of care with such a demanding lifestyle, and it is hardly surprising that many critical-care doctors seek less-stressful practices (e.g., sleep medicine) or retirement.
New physicians who see the work-life imbalance are shying away from critical care in significant enough numbers, yet the demand for these doctors is growing. Whether that shift is due to the attraction of careers in HM, emergency medicine, or other IM specialties, or the perceived negatives to being a full-time intensivist, is beside the point. The medical workforce needs more critical-care physicians. And we are not going to be able to meet this need by assuming we will be able to recruit and train more surgeons, anesthesiologists, or pulmonary physicians in traditional critical-care pathways.
The situation is further complicated by workforce shortages on the rest of the ICU healthcare team. Many senior ICU nurses are reaching the end of their careers, and the pipeline to replace them is anything but robust. And all this comes at a time when the acuity of hospitalized patients is increasing and the demands for ICU expertise is at its height.
It is no surprise that hospitalists are called upon to fill the critical-care gap. SHM surveys show that 92% of hospitalists include ICU care in their practice. While hospitalists clearly do not have the training or skills to replace the intensivist, we clearly are witnessing a “scope creep.” Hospitalists are being asked to stretch their skills to fill the void in critical care.
Competence Question
The response to this is manifest in many ways. For example, steadily increasing numbers of hospitalists attend SHM’s annual critical-care precourses, and our procedures courses invariably sell out. These brief courses are important to hospitalists and their patients. Yet a day (or even two) of focused training for hospitalists will not raise their skill set to replace or even augment critical-care-trained physicians at their hospitals. The patients will keep coming and continue to need the expertise for their most-acute-care needs. Something must be done.
There are pockets of experiments on filling the increasing critical-care gap. Emory University’s Center for Critical Care in Atlanta will soon launch an experimental, HM-critical-care training program that will attempt to develop and verify critical-care competencies in just one postgraduate year after IM residency. A complementary approach could include a hospitalist-focused track within the three years of IM residency to include less outpatient medicine and more intensive-care training. This could be part of a broader restructuring of internal and family medicine residencies, which recognize the career paths (and needs) of their residents as some enter hospital-focused practice (e.g., as hospitalists, cardiologists, intensivists) and some concentrate more on the patient outside the hospital (e.g., primary care, endocrinology, rheumatology).
As training evolves, there will be practical issues of credentialing. Currently, a general-IM-trained hospitalist is required to complete a two-year fellowship in critical care to be a specialist. Interestingly, if one’s residency training is in surgery, anesthesia, or rheumatology, that physician requires only one additional year in critical care to become eligible for added/special qualifications in critical-care medicine. What, precisely, is the carryover of the longer residency experience that makes it possible to attain competency in critical care in half the training time?
There also is the risk that we will create a workforce that includes the intensivist-lite—someone who does not have complete, recognized training in critical care but has more than the typical hospitalist, and is perceived as “better than having no intensivist.” Is this in the best interest of our patients or our discipline?
Supply Solutions
There are other approaches to the workforce challenge beyond asking hospitalists to step away from practice for an additional year or two of training. As in other aspects of the hospital workforce, it is time to examine alternative deployment of the entire healthcare team. If intensivists and hospitalists are in limited supply, we need to revisit their roles and further look for opportunities to engage acute-care nurse practitioners, physician assistants, RNs, and others on the healthcare team to meet the expanding needs of our patients and our hospitals.
Another strategy would include regionalization of healthcare in population centers with multiple hospitals. In Orange County, Calif., where I live, there are 33 hospitals for 3 million people, each with some form of an ICU. Is it time to set standards of coverage and expertise so that 10 to 15 hospitals can provide a fully staffed ICU, and the other hospitals refer their most-acute patients rather than be stretched to staff their ICUs at a time of workforce shortages? Is it time to do what we did with trauma centers and set various levels of care so that not every hospital can or should be doing the most intense and costly level of care?
SHM, HM, and, most importantly, hospitalists find themselves in the midst of this growing problem. We must be part of the solution.
It is important to recognize the significant variability in the intensivist skill sets that individual hospitalists and HM groups currently possess; the variance creates hurdles in being able to step up and fill the critical-care gap. I’ve heard about hospitalists pausing their practices to obtain additional training in critical care. Hospitals across America are (or soon will be) scrambling to integrate their hospitalists and intensivists to maximize coverage and expertise. SHM has noticed an increased demand on hospitalists to increase knowledge and skills so they can extend the local coverage of critically ill patients.
It is time for SHM to clearly understand how this sea change is affecting you professionally and personally, because you practice on the frontlines of our nation’s hospitals.
It is time for SHM to engage our colleagues in the ICU—critical-care physicians and their professional societies—to understand their perspective and initiatives on this growing crisis.
It is time for SHM to engage medical educators in residency and fellowship training to explore potential changes in the curriculum—changes aimed at young physicians proceeding through their training that yield hospital-based physicians better prepared to enter the hospital environment of the 21st century.
And SHM may need to engage the boards and other credentialing bodies to look for flexibility that will reflect today’s realities, attract the best-trained physicians to care for the most-acutely-ill patients, and protect our patients by demanding expertise and training at the most appropriate levels. TH
Dr. Wellikson is CEO of SHM.
There is a brewing crisis in critical-care medicine, a crisis that already is impacting HM. It would be easier for hospitalists to look the other way and say, “This is not our problem.” Make no mistake: It is our problem. There are not enough intensive-care physicians to go around.
In my generation, most physicians who trained in pulmonary and critical-care medicine thought they would do some office care, some bronchoscopies, and some critical care for the first several years of their professional lives. Few imagined that, in their 60s, their practice would still include rescuing patients from death at 2 a.m. Couple the intensity of care with such a demanding lifestyle, and it is hardly surprising that many critical-care doctors seek less-stressful practices (e.g., sleep medicine) or retirement.
New physicians who see the work-life imbalance are shying away from critical care in significant enough numbers, yet the demand for these doctors is growing. Whether that shift is due to the attraction of careers in HM, emergency medicine, or other IM specialties, or the perceived negatives to being a full-time intensivist, is beside the point. The medical workforce needs more critical-care physicians. And we are not going to be able to meet this need by assuming we will be able to recruit and train more surgeons, anesthesiologists, or pulmonary physicians in traditional critical-care pathways.
The situation is further complicated by workforce shortages on the rest of the ICU healthcare team. Many senior ICU nurses are reaching the end of their careers, and the pipeline to replace them is anything but robust. And all this comes at a time when the acuity of hospitalized patients is increasing and the demands for ICU expertise is at its height.
It is no surprise that hospitalists are called upon to fill the critical-care gap. SHM surveys show that 92% of hospitalists include ICU care in their practice. While hospitalists clearly do not have the training or skills to replace the intensivist, we clearly are witnessing a “scope creep.” Hospitalists are being asked to stretch their skills to fill the void in critical care.
Competence Question
The response to this is manifest in many ways. For example, steadily increasing numbers of hospitalists attend SHM’s annual critical-care precourses, and our procedures courses invariably sell out. These brief courses are important to hospitalists and their patients. Yet a day (or even two) of focused training for hospitalists will not raise their skill set to replace or even augment critical-care-trained physicians at their hospitals. The patients will keep coming and continue to need the expertise for their most-acute-care needs. Something must be done.
There are pockets of experiments on filling the increasing critical-care gap. Emory University’s Center for Critical Care in Atlanta will soon launch an experimental, HM-critical-care training program that will attempt to develop and verify critical-care competencies in just one postgraduate year after IM residency. A complementary approach could include a hospitalist-focused track within the three years of IM residency to include less outpatient medicine and more intensive-care training. This could be part of a broader restructuring of internal and family medicine residencies, which recognize the career paths (and needs) of their residents as some enter hospital-focused practice (e.g., as hospitalists, cardiologists, intensivists) and some concentrate more on the patient outside the hospital (e.g., primary care, endocrinology, rheumatology).
As training evolves, there will be practical issues of credentialing. Currently, a general-IM-trained hospitalist is required to complete a two-year fellowship in critical care to be a specialist. Interestingly, if one’s residency training is in surgery, anesthesia, or rheumatology, that physician requires only one additional year in critical care to become eligible for added/special qualifications in critical-care medicine. What, precisely, is the carryover of the longer residency experience that makes it possible to attain competency in critical care in half the training time?
There also is the risk that we will create a workforce that includes the intensivist-lite—someone who does not have complete, recognized training in critical care but has more than the typical hospitalist, and is perceived as “better than having no intensivist.” Is this in the best interest of our patients or our discipline?
Supply Solutions
There are other approaches to the workforce challenge beyond asking hospitalists to step away from practice for an additional year or two of training. As in other aspects of the hospital workforce, it is time to examine alternative deployment of the entire healthcare team. If intensivists and hospitalists are in limited supply, we need to revisit their roles and further look for opportunities to engage acute-care nurse practitioners, physician assistants, RNs, and others on the healthcare team to meet the expanding needs of our patients and our hospitals.
Another strategy would include regionalization of healthcare in population centers with multiple hospitals. In Orange County, Calif., where I live, there are 33 hospitals for 3 million people, each with some form of an ICU. Is it time to set standards of coverage and expertise so that 10 to 15 hospitals can provide a fully staffed ICU, and the other hospitals refer their most-acute patients rather than be stretched to staff their ICUs at a time of workforce shortages? Is it time to do what we did with trauma centers and set various levels of care so that not every hospital can or should be doing the most intense and costly level of care?
SHM, HM, and, most importantly, hospitalists find themselves in the midst of this growing problem. We must be part of the solution.
It is important to recognize the significant variability in the intensivist skill sets that individual hospitalists and HM groups currently possess; the variance creates hurdles in being able to step up and fill the critical-care gap. I’ve heard about hospitalists pausing their practices to obtain additional training in critical care. Hospitals across America are (or soon will be) scrambling to integrate their hospitalists and intensivists to maximize coverage and expertise. SHM has noticed an increased demand on hospitalists to increase knowledge and skills so they can extend the local coverage of critically ill patients.
It is time for SHM to clearly understand how this sea change is affecting you professionally and personally, because you practice on the frontlines of our nation’s hospitals.
It is time for SHM to engage our colleagues in the ICU—critical-care physicians and their professional societies—to understand their perspective and initiatives on this growing crisis.
It is time for SHM to engage medical educators in residency and fellowship training to explore potential changes in the curriculum—changes aimed at young physicians proceeding through their training that yield hospital-based physicians better prepared to enter the hospital environment of the 21st century.
And SHM may need to engage the boards and other credentialing bodies to look for flexibility that will reflect today’s realities, attract the best-trained physicians to care for the most-acutely-ill patients, and protect our patients by demanding expertise and training at the most appropriate levels. TH
Dr. Wellikson is CEO of SHM.
How should Parkinson’s disease be managed perioperatively?
Case
A 67-year-old female with moderately advanced Parkinson’s disease (PD) had a mechanical fall in her home, which resulted in a humeral fracture. The fall occurred in the morning before she was able to take her medications and was related to her difficulty in initiating movements.
On her current regimen, her PD symptoms are controlled. She is able to perform daily living activities independently and ambulates without assistance. She also performs more complex tasks (e.g., cooking and managing her finances). She has not exhibited any symptoms consistent with dementia. She occasionally experiences dyspnea on exertion and dysphagia, but she has not been evaluated for these complaints. She takes carbidopa/levodopa (CD/LD) 25 mg/100 mg four times a day, amantadine 100 mg twice daily, and ropinirole 3 mg three times a day.
She is scheduled for open reduction internal fixation of her fracture; the orthopedic surgeon has requested a perioperative risk assessment and recommendations concerning her medications. How should PD be managed perioperatively?
Overview
Advances in surgical and anesthetic techniques, in combination with an aging population, have contributed to an increasing number of geriatric patients undergoing surgery. As many as 50% of Americans older than 65 will undergo a surgical procedure; hospitalists will comanage many of these patients in the perioperative period.1
Although cardiopulmonary disorders receive a great deal of attention with regard to perioperative risk assessment, other comorbid conditions also contribute to perioperative risk—namely, disorders specific to the elderly population. Parkinson’s disease is one such condition that deserves attention.
PD is a progressive, neurodegenerative condition associated with loss of dopaminergic neurons and the presence of Lewy bodies within the substantia nigra and other areas of the brain and peripheral autonomic nervous system.2 Cardinal clinical features include rigidity, bradykinesia, and resting tremor. A supportive feature is a consistent response to levodopa. Postural instability, cognitive impairment, and autonomic dysfunction usually occur later in the disease.3,4
As the population ages, Parkinson’s disease is becoming more prevalent, affecting approximately 1% of individuals older than 60.5 These patients pose a specific challenge to the hospitalist, not only because the multiorgan system manifestations of PD can raise surgical risk, but also due to the direct effects of dopaminergic medications used to treat PD, lack of a parenteral route for these medications in NPO patients, and the risks associated with abrupt withdrawal of these medications.
Although surgical risk in PD patients has received intermittent attention in surgical, anesthesia, and neurology literature, there is no broad consensus statement or treatment guideline for the perioperative approach.
Literature Review
A retrospective cohort of 51 PD patients undergoing various types of surgery revealed that PD patients have a longer hospital stay than matched cohorts.6 Pepper et al studied a cohort of 234 PD patients in the Veterans Administration population who were undergoing a variety of surgeries.7 They found that patients with PD had a longer acute hospital stay and had higher in-hospital mortality.7 The multisystem manifestations of PD might account for this global increase in perioperative risk.
The following are reviews of organ-system manifestations of PD and their relevance to the perioperative period.
Motor: The motor symptoms of PD place patients at increased risk for falls and might impair their ability to participate in rehabilitation. Mueller et al demonstrated that there was a significantly increased risk of postoperative falls and a higher need for inpatient rehabilitation due to motor difficulties in the PD cohort.6
Pulmonary: PD patients have increased risk of abnormal pulmonary function secondary to rigidity and akinesia. Increased airway resistance and decreased lung elastic recoil lead to obstructive lung disease.8,9 Rigidity of voluntary chest wall and upper airway muscles leads to a restrictive lung disease pattern.8,10 Furthermore, respiratory dyskinesia is a common side effect of levodopa, which can result in restrictive and dyskinetic ventilation.11 As a consequence of disordered respiratory mechanics (especially in combination with disordered swallow mechanics), PD patients are at increased risk of lower respiratory infections. In fact, pneumonia remains the leading cause of mortality among PD patients.11
Not surprisingly, several cohorts have suggested that PD patients undergoing surgical procedures are at higher risk for atelectasis, pneumonia, and postoperative respiratory failure.7,12 Postoperative VTE rates are not statistically different between PD patients and matched cohorts.
Gastrointestinal: Abnor-malities in muscles of the mouth, pharynx, and esophagus account for the dysphagia commonly noted in PD.13 Barium swallow tests are abnormal in 80% or more of PD patients.14 Dysphagia can lead to aspiration, as well as inadequate oral intake, resulting in pneumonia and malnutrition, respectively.15 Dysfunction of the myenteric plexus (evidenced by Lewy Body deposition) accounts for gastrointestinal dysmotility manifested as gastroparesis, ileus, and slow colonic transit, which results in constipation.16
PD patients in the postoperative period are at risk for swallowing difficulties, which increases the risk of aspiration and might delay initiation of oral medications. Gastroparesis threatens appropriate delivery of oral medications for adequate absorption. In addition, postoperative ileus and constipation can pose challenges.
Cardiovascular: Such cardiac sympathetic abnormalities as orthostatic hypotension, postprandial or exercise-induced hypotension, impaired heart rate variability, and dysrhythmias are common in PD.17 Pepper et al found a trend toward increased risk of hypotension and acute myocardial infarction (MI) in PD patients undergoing surgery.7
Genitourinary: Urinary complaints (e.g., nocturia, frequency, urgency, and urge incontinence) are common in PD patients.18 These clinical complaints correspond to involuntary detrusor contractions (detrusor hyperreflexia).19 Pepper et al found an increased risk of postoperative urinary tract infection in PD patients.7
Cognitive: A recognized feature of advanced PD is cognitive impairment. Studies estimate the prevalence of dementia in cohorts of PD patients is from 28% to 44%. PD with dementia has been associated with shortened survival, impaired quality of life, and increased caregiver distress.20 Pepper et al noted a trend toward increased incidence of postoperative delirium in their cohort of 234 PD patients undergoing surgery.7
Medication: Management of anti-Parkinsonian medications in the perioperative period poses unique challenges. These medications’ prodopaminergic effects can lead to hemodynamic compromise and are potentially arrhythmogenic. At the same time, abrupt withdrawal of these medications can lead to a potentially lethal condition called Parkinsonism-hyperpyrexia syndrome (PHS), which is clinically similar to neuroleptic malignant syndrome.21 PHS is characterized by very high fever, extreme muscle rigidity, autonomic instability, altered consciousness, and multiple severe systemic complications (e.g., acute renal failure, disseminated intravascular coagulation, autonomic failure, aspiration pneumonia, and infections). PHS occurs in up to 4% of PD patients; mortality is reported to be from 4% for treated to 20% for untreated episodes.22-24
As many as 30% of patients who survive a PHS episode have worsening of their PD symptoms and never return to their pre-PHS baseline. PHS prevention in hospitalized patients by uninterrupted administration of PD medications should be the goal. Early recognition and aggressive treatment is key to successful recovery.
Furthermore, even brief interruption of medications can lead to decompensation of Parkinsonian symptoms, which not only delays recovery from surgery, but also increases the risk for multisystem complications as discussed above.25 Traditional anti-Parkinsonian medications can only be delivered orally, presenting significant challenges for NPO patients, especially those undergoing enteric surgery requiring bowel rest.
Case reports describe various approaches to medication management in the perioperative period, but no single consensus statement (or treatment guideline) exists. The most common clinical scenarios are:
- Patient undergoes short surgery and is able to take oral medication immediately after the procedure (e.g., orthopedic, eye);
- Patient undergoes more lengthy surgery and will be able to take enteric medications perioperatively (e.g., longer orthopedic surgeries, genitourinary); and
- Patient undergoes lengthy procedure in which they will be required to adhere to bowel rest (e.g., bowel resection).
Depending on the category, the approach to medication management might differ.
Furuya et al describe a 70-year-old male with PD who had previously experienced perioperative complications associated with his medication management, including postoperative rigidity, dysphagia, and difficulty maintaining respiratory secretions.26 These symptoms were reversed with intravenous levodopa. However, the patient experienced hypotension and premature ventricular contractions as side effects of this therapy. This patient was scheduled to undergo hepatic lobectomy. Given his previous complications, Furuya et al provided enteral administration of CD/LD via nasogastric tube every two hours during the surgery, with placement of a duodenostomy for postoperative administration of CD/LD. The patient maintained hemodynamic stability throughout the perioperative period and emerged from anesthesia smoothly with no muscle rigidity or postoperative complications.26
Fujii et al described three cases of PD patients undergoing gastrointestinal surgery. They suggested that the dose of medication required to control symptoms should be minimized before surgery to avoid withdrawal symptoms. They also described the use of intravenous levodopa immediately, postoperatively, while the patient was unable to tolerate enteric medications, and suggested the use of prokinetics to prevent ileus and maximize drug absorption.27
Gálvez-Jiménez et al discuss the limitations of intravenous levodopa, including hemodynamic compromise, need for escalating doses, frequent adjustments to maintain effect, and large amount of fluids required. They propose the use of subcutaneous apomorphine, which is a potent D1/D2 dopamine agonist in conjunction with rectal domeperidone, a D2 antagonist with poor blood-brain-barrier penetration, to counteract the peripheral dopaminergic side effects. The main limitation to this approach is that domeperidone is not available in the U.S.28
Intravenous antihistamines and anticholinergics are readily available. However, they show limited efficacy in halting Parkinsonian symptoms and carry multiple side effects (e.g., confusion, delay in gastrointestinal recovery, and urinary retention).28
Various anesthesiologists agree with administering anti-PD medications immediately, preoperatively, and restarting medications as soon as possible postoperatively. However, they do not provide uniform treatment guidelines regarding specific medication management.29,30
Deep-brain-stimulation (DBS) management: DBS is an effective treatment for advanced PD. There are more than 60,000 patients around the world who have DBS for various conditions, mostly for PD. Therefore, it is increasingly likely that hospitalists will encounter hospitalized patients with advanced PD who are treated both pharmacologically and with DBS. It is important to recognize that stimulation, just like PD medications, cannot be stopped suddenly. If there is any concern of the DBS system malfunctioning (i.e., fracture of the hardware during a fall), the neurologist or neurosurgeon managing the DBS should be contacted immediately. Certain diagnostic tests (MRI) and treatment procedures (diathermy) are contraindicated, and if done inappropriately, can result in permanent brain damage.31,32,33
During surgeries requiring blood-vessel cauterization, DBS should be temporarily turned off. This can be done with the patient’s handheld device or, preferably, by a trained technician usually available through 24/7 technical support services provided by the manufacturer.
Summary of recommendations: There are no clear treatment guidelines regarding the optimal perioperative management of PD patients. The following measures are based on available data and are extensions of routine perioperative management; however, there is no evidence to demonstrate their efficacy in decreasing complication rates among patients with PD:
- A thorough preoperative history and physical examination should include Parkinsonian signs and symptoms, precise medication regimen with doses and timing of intake, effects of medication withdrawal or missed doses, type of surgery planned, and comorbid conditions;
- Depending on symptoms mentioned in the history, consider further testing for dysphagia (preoperative swallow evaluation) and dyspnea (preoperative pulmonary function tests);
- The major goal of medication management in the perioperative period is to continue administration of dopamine replacement therapy as close to the outpatient regimen as possible.
- Titrate down dose of anti-Parkinsonian medications to lowest possible dose prior to surgery if prolonged NPO status is anticipated;
- Ensure medications are administered immediately prior to surgery;
- For short, nonenteric procedures, resume outpatient medication doses and timing of administration as soon as possible postoperatively. For longer, nonenteric surgeries, consider placement of nasogastric tube for medication delivery during procedure and immediately postoperatively;
- If the major limitation of oral medication intake is dysphagia, the use of orally disintegrated formulation CD/LD (parcopa) is helpful;
- For longer enteric surgeries in which patient must be on bowel rest, recommend consultation with neurologist specialized in movement disorders to guide use of intravenous or subcutaneous agents. Transdermal delivery systems of the dopamine agonist rotigotine are in the process of being approved in the U.S. market and might be helpful for this purpose;
- Initiation of tube feeding, when co-administered with levodopa, might result in sudden changes in medication absorption and potential worsening of PD symptoms; feeding should be started slowly and preferably at night when the body’s dopamine requirements are lower;
- Consider use of promotility agents;
- If apomorphine or intravenous LD are not available, consider trial of intravenous anticholinergics or antihistamines, carefully observing for potential cognitive and behavioral side effects;
- Avoid such dopamine antagonists as droperidol, haloperidol, risperidone, metaclopramide, prochlorperazine, or promethazine, as these medications can worsen Parkinsonian symptoms; and
- If patient was on MAO-B inhibitors (selegiline, rasagiline) before surgery, be aware of multiple potential interactions with various medications that are commonly used in perioperative period, including anesthesia agents and certain analgesics, specifically meperidine. These interactions include serotonin syndrome, and can be life-threatening.34
- Psychiatric considerations: delirium precautions;
- Motor considerations: early PT/OT, early referral to inpatient rehabilitation; fall precautions;
- Pulmonary considerations: institute aggressive incentive spirometry, postural drainage, management of respiratory secretions, and breathing exercises; VTE prophylaxis;
- Gastrointestinal considerations: aspiration precautions and prompt speech therapy to evaluate for aspiration and to teach appropriate swallow techniques (chin tuck); institute aggressive bowel regimen; maximize fluids, electrolytes, and avoid narcotics to prevent precipitating or exacerbating ileus;
- Cardiovascular considerations: monitor orthostatic vital signs; fall precautions to avoid syncopal falls; and
- Genitourinary considerations: early urinary catheter removal; vigilance in monitoring for urinary tract infection.
Back to the Case
The patient underwent repair of her fracture, was extubated, and recovered from general anesthesia without incident. She was evaluated in the postanesthesia care unit, at which time she had a slight tremor and mild rigidity. She was immediately given a dose of her CD/LD, and her evening doses of amantadine and ropinirole were resumed. The patient had no significant flare of her Parkinsonian symptoms and did not exhibit any evidence of PHS.
A postoperative consultation was placed for speech therapy, physical therapy, and occupational therapy. She was given low-molecular-weight heparin for VTE prophylaxis and asked to use incentive spirometry. On postoperative day one, she complained of urinary frequency. A urinalysis was consistent with possible infection. She was discharged home on her previous medication regimen, in addition to antibiotics for cystitis.
If the procedure had not been emergent, the patient might have benefited from a preoperative swallow evaluation, given her dysphagia. Consultation with a speech therapist would have ensured that the patient was educated regarding aspiration precautions. Although this patient did not have difficulty with extubation or experience postoperative respiratory failure, abnormal preoperative pulmonary function tests might have prompted the anesthesiologists to consider alternative low-risk techniques (e.g., a local nerve block).
Bottom Line
Perioperative management of patients with Parkinson’s disease requires knowledge of the multisystem disease characteristics that raise perioperative risk and the effects of the medications used to treat PD. To date, no clear treatment guidelines exist for the optimal perioperative management of PD patients.
However, vigilance in detecting possible complications and instituting attentive perioperative care can aid a hospitalist consultant in improving overall care for these patients. TH
Dr. Patel is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Denver VA Medical Center and instructor of medicine at the University of Colorado Denver. Dr. Anderson is a hospitalist at the Denver VA Medical Center and assistant professor of medicine at the University of Colorado Denver. Dr. Klepitskaya is a neurologist and assistant professor of neurology at the University of Colorado Denver.
References
- Ersan T. Perioperative management of the geriatric patient. Web MD website. Available at: http://emedicine.medscape.com/article/285433-overview. Accessed May 10, 2010.
- Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211.
- Ward CD, Gibb WR. Research diagnostic criteria for Parkinson’s disease. Adv Neurol. 1990;53:245-249.
- Stern MB, Koller WC. Parkinsonian Syndromes. New York: Informa Healthcare Publishing; 1993.
- Nutt JG, Wooten GF. Clinical practice. Diagnosis and initial management of Parkinson’s disease. N Engl J Med. 2005;353(10):1021-1027.
- Mueller MC, Jüptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394(3):511-515.
- Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47(8):967-972.
- Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci. 1998;5(2):131-135.
- Neu HC, Connolly JJ Jr., Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Ann Rev Respir Dis. 1967;95(1):33-47.
- Chokroverty S. The spectrum of ventilatory disturbances in movement disorders. In: Chokroverty S, ed. Movement Disorders. Great Neck, New York: PMA Publishing Corp.; 1990:365-392.
- Shill H, Stacy M. Respiratory complications of Parkinson’s disease. Semin Respir Crit Care Med. 2002;23(3):261-265.
- Easdown LJ, Tessler KJ, Minuk J. Upper airway involvement in Parkinson’s disease resulting in postoperative respiratory failure. Can J Anaesth. 1995;42(4):344-347.
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2(2):107-116.
- Castell JA, Johnston BT, Colcher A, Li Q, Gideon RM, Castell DO. Manometric abnormalities of the oesophagus in patients with Parkinson’s disease. Neurogastroenterol Motil. 2001;13(4):361-364.
- Marcason W. What are the primary nutritional issues for a patient with Parkinson’s disease? J Am Diet Assoc. 2009;109(7):1316.
- Djaldetti R, Lev N, Melamed E. Lesions outside the CNS in Parkinson’s disease. Mov Disord. 2009;24(6):793-800.
- Orimo S, Takahashi A, Uchihara T, Mori F, Kakita A, Wakabayashi K, Takahashi H. Degeneration of cardiac sympathetic nerve begins in the early disease process of Parkinson’s disease. Brain Pathol. 2007;17(1):24-30.
- Lemack GE, Dewey RB Jr., Roehrborn CG, O’Suilleabhain PE, Zimmern PE. Questionnaire-based assessment of bladder dysfunction in patients with mild to moderate Parkinson’s disease. Urology. 2000;56(2):250-254.
- Singer C. Urinary dysfunction in Parkinson’s disease. Clin Neurosci. 1998;5(2):78-86.
- Leverenz JB, Quinn JF, Zabetian C, Zhang J, Montine KS, Montine TJ. Cognitive impairment and dementia in patients with Parkinson disease. Curr Top Med Chem. 2009;9(10):903-912.
- Factor SA, Santiago A. Parkinson-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht S, Fahn S, eds. Movement Disorder Emergencies: Diagnosis and Treatment. New York: Humana Press; 2005:29-40.
- Reed AP, Han DG: Intraoperative exacerbation of Parkinson’s disease. Anesth Analg. 1992;75(5):850-853.
- Harada T, Mitsuoka K, Kumagai R, et al. Clinical features of malignant syndrome in Parkinson’s disease and related neurological disorders. Parkinsonism Relat Disord. 2003;9(Suppl 1):S15-23.
- Onofrj M, Thomas A. Acute akinesia in Parkinson disease. Neurology. 2005;64(7):1162-1169.
- Serrano-Dueñas M. Neuroleptic malignant syndrome-like, or—dopaminergic malignant syndrome—due to levodopa therapy withdrawal. Clinical features in 11 patients. Parkinsonism Relat Disord. 2003;9(3):175-178.
- Furuya R, Hirai A, Andoh T, Kudoh I, Okumura F. Successful perioperative management of a patient with Parkinson’s disease by enteral levodopa administration under propofol anesthesia. Anesthesiology. 1998;89(1):261-263.
- Fujii T, Nakabayashi T, Hashimoto S, Kuwano H. Successful perioperative management of patients with Parkinson’s disease following gastrointestinal surgery: report of three cases. Surg Today. 2009;39(9):807-810.
- Gálvez-Jiménez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22(2):367-377.
- Kalenka A, Schwarz A. Anasthesia and Parkinson’s disease: how to manage with new therapies? Curr Opin Anesthesiol. 2009;22(3):419-424.
- Nicholson G, Pereira AC, Hall GM. Parkinson’s disease and anesthesia. Br J Anaesthesia. 2002;89(6):904-916.
- Rezai AR, Baker KB, Tkach JA, et al. Is magnetic resonance imaging safe for patients with neurostimulation systems used for deep brain stimulation? Neurosurgery. 2005:57(5):1056-1062.
- Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery. 2005:57(5):E1063.
- Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology. 2001;56 (10):1384-1386.
- Gordon PH, Frucht SJ. Neuroleptic malignant syndrome in advanced Parkinson’s disease. Mov Disord. 2001;16(5):960-962.
Case
A 67-year-old female with moderately advanced Parkinson’s disease (PD) had a mechanical fall in her home, which resulted in a humeral fracture. The fall occurred in the morning before she was able to take her medications and was related to her difficulty in initiating movements.
On her current regimen, her PD symptoms are controlled. She is able to perform daily living activities independently and ambulates without assistance. She also performs more complex tasks (e.g., cooking and managing her finances). She has not exhibited any symptoms consistent with dementia. She occasionally experiences dyspnea on exertion and dysphagia, but she has not been evaluated for these complaints. She takes carbidopa/levodopa (CD/LD) 25 mg/100 mg four times a day, amantadine 100 mg twice daily, and ropinirole 3 mg three times a day.
She is scheduled for open reduction internal fixation of her fracture; the orthopedic surgeon has requested a perioperative risk assessment and recommendations concerning her medications. How should PD be managed perioperatively?
Overview
Advances in surgical and anesthetic techniques, in combination with an aging population, have contributed to an increasing number of geriatric patients undergoing surgery. As many as 50% of Americans older than 65 will undergo a surgical procedure; hospitalists will comanage many of these patients in the perioperative period.1
Although cardiopulmonary disorders receive a great deal of attention with regard to perioperative risk assessment, other comorbid conditions also contribute to perioperative risk—namely, disorders specific to the elderly population. Parkinson’s disease is one such condition that deserves attention.
PD is a progressive, neurodegenerative condition associated with loss of dopaminergic neurons and the presence of Lewy bodies within the substantia nigra and other areas of the brain and peripheral autonomic nervous system.2 Cardinal clinical features include rigidity, bradykinesia, and resting tremor. A supportive feature is a consistent response to levodopa. Postural instability, cognitive impairment, and autonomic dysfunction usually occur later in the disease.3,4
As the population ages, Parkinson’s disease is becoming more prevalent, affecting approximately 1% of individuals older than 60.5 These patients pose a specific challenge to the hospitalist, not only because the multiorgan system manifestations of PD can raise surgical risk, but also due to the direct effects of dopaminergic medications used to treat PD, lack of a parenteral route for these medications in NPO patients, and the risks associated with abrupt withdrawal of these medications.
Although surgical risk in PD patients has received intermittent attention in surgical, anesthesia, and neurology literature, there is no broad consensus statement or treatment guideline for the perioperative approach.
Literature Review
A retrospective cohort of 51 PD patients undergoing various types of surgery revealed that PD patients have a longer hospital stay than matched cohorts.6 Pepper et al studied a cohort of 234 PD patients in the Veterans Administration population who were undergoing a variety of surgeries.7 They found that patients with PD had a longer acute hospital stay and had higher in-hospital mortality.7 The multisystem manifestations of PD might account for this global increase in perioperative risk.
The following are reviews of organ-system manifestations of PD and their relevance to the perioperative period.
Motor: The motor symptoms of PD place patients at increased risk for falls and might impair their ability to participate in rehabilitation. Mueller et al demonstrated that there was a significantly increased risk of postoperative falls and a higher need for inpatient rehabilitation due to motor difficulties in the PD cohort.6
Pulmonary: PD patients have increased risk of abnormal pulmonary function secondary to rigidity and akinesia. Increased airway resistance and decreased lung elastic recoil lead to obstructive lung disease.8,9 Rigidity of voluntary chest wall and upper airway muscles leads to a restrictive lung disease pattern.8,10 Furthermore, respiratory dyskinesia is a common side effect of levodopa, which can result in restrictive and dyskinetic ventilation.11 As a consequence of disordered respiratory mechanics (especially in combination with disordered swallow mechanics), PD patients are at increased risk of lower respiratory infections. In fact, pneumonia remains the leading cause of mortality among PD patients.11
Not surprisingly, several cohorts have suggested that PD patients undergoing surgical procedures are at higher risk for atelectasis, pneumonia, and postoperative respiratory failure.7,12 Postoperative VTE rates are not statistically different between PD patients and matched cohorts.
Gastrointestinal: Abnor-malities in muscles of the mouth, pharynx, and esophagus account for the dysphagia commonly noted in PD.13 Barium swallow tests are abnormal in 80% or more of PD patients.14 Dysphagia can lead to aspiration, as well as inadequate oral intake, resulting in pneumonia and malnutrition, respectively.15 Dysfunction of the myenteric plexus (evidenced by Lewy Body deposition) accounts for gastrointestinal dysmotility manifested as gastroparesis, ileus, and slow colonic transit, which results in constipation.16
PD patients in the postoperative period are at risk for swallowing difficulties, which increases the risk of aspiration and might delay initiation of oral medications. Gastroparesis threatens appropriate delivery of oral medications for adequate absorption. In addition, postoperative ileus and constipation can pose challenges.
Cardiovascular: Such cardiac sympathetic abnormalities as orthostatic hypotension, postprandial or exercise-induced hypotension, impaired heart rate variability, and dysrhythmias are common in PD.17 Pepper et al found a trend toward increased risk of hypotension and acute myocardial infarction (MI) in PD patients undergoing surgery.7
Genitourinary: Urinary complaints (e.g., nocturia, frequency, urgency, and urge incontinence) are common in PD patients.18 These clinical complaints correspond to involuntary detrusor contractions (detrusor hyperreflexia).19 Pepper et al found an increased risk of postoperative urinary tract infection in PD patients.7
Cognitive: A recognized feature of advanced PD is cognitive impairment. Studies estimate the prevalence of dementia in cohorts of PD patients is from 28% to 44%. PD with dementia has been associated with shortened survival, impaired quality of life, and increased caregiver distress.20 Pepper et al noted a trend toward increased incidence of postoperative delirium in their cohort of 234 PD patients undergoing surgery.7
Medication: Management of anti-Parkinsonian medications in the perioperative period poses unique challenges. These medications’ prodopaminergic effects can lead to hemodynamic compromise and are potentially arrhythmogenic. At the same time, abrupt withdrawal of these medications can lead to a potentially lethal condition called Parkinsonism-hyperpyrexia syndrome (PHS), which is clinically similar to neuroleptic malignant syndrome.21 PHS is characterized by very high fever, extreme muscle rigidity, autonomic instability, altered consciousness, and multiple severe systemic complications (e.g., acute renal failure, disseminated intravascular coagulation, autonomic failure, aspiration pneumonia, and infections). PHS occurs in up to 4% of PD patients; mortality is reported to be from 4% for treated to 20% for untreated episodes.22-24
As many as 30% of patients who survive a PHS episode have worsening of their PD symptoms and never return to their pre-PHS baseline. PHS prevention in hospitalized patients by uninterrupted administration of PD medications should be the goal. Early recognition and aggressive treatment is key to successful recovery.
Furthermore, even brief interruption of medications can lead to decompensation of Parkinsonian symptoms, which not only delays recovery from surgery, but also increases the risk for multisystem complications as discussed above.25 Traditional anti-Parkinsonian medications can only be delivered orally, presenting significant challenges for NPO patients, especially those undergoing enteric surgery requiring bowel rest.
Case reports describe various approaches to medication management in the perioperative period, but no single consensus statement (or treatment guideline) exists. The most common clinical scenarios are:
- Patient undergoes short surgery and is able to take oral medication immediately after the procedure (e.g., orthopedic, eye);
- Patient undergoes more lengthy surgery and will be able to take enteric medications perioperatively (e.g., longer orthopedic surgeries, genitourinary); and
- Patient undergoes lengthy procedure in which they will be required to adhere to bowel rest (e.g., bowel resection).
Depending on the category, the approach to medication management might differ.
Furuya et al describe a 70-year-old male with PD who had previously experienced perioperative complications associated with his medication management, including postoperative rigidity, dysphagia, and difficulty maintaining respiratory secretions.26 These symptoms were reversed with intravenous levodopa. However, the patient experienced hypotension and premature ventricular contractions as side effects of this therapy. This patient was scheduled to undergo hepatic lobectomy. Given his previous complications, Furuya et al provided enteral administration of CD/LD via nasogastric tube every two hours during the surgery, with placement of a duodenostomy for postoperative administration of CD/LD. The patient maintained hemodynamic stability throughout the perioperative period and emerged from anesthesia smoothly with no muscle rigidity or postoperative complications.26
Fujii et al described three cases of PD patients undergoing gastrointestinal surgery. They suggested that the dose of medication required to control symptoms should be minimized before surgery to avoid withdrawal symptoms. They also described the use of intravenous levodopa immediately, postoperatively, while the patient was unable to tolerate enteric medications, and suggested the use of prokinetics to prevent ileus and maximize drug absorption.27
Gálvez-Jiménez et al discuss the limitations of intravenous levodopa, including hemodynamic compromise, need for escalating doses, frequent adjustments to maintain effect, and large amount of fluids required. They propose the use of subcutaneous apomorphine, which is a potent D1/D2 dopamine agonist in conjunction with rectal domeperidone, a D2 antagonist with poor blood-brain-barrier penetration, to counteract the peripheral dopaminergic side effects. The main limitation to this approach is that domeperidone is not available in the U.S.28
Intravenous antihistamines and anticholinergics are readily available. However, they show limited efficacy in halting Parkinsonian symptoms and carry multiple side effects (e.g., confusion, delay in gastrointestinal recovery, and urinary retention).28
Various anesthesiologists agree with administering anti-PD medications immediately, preoperatively, and restarting medications as soon as possible postoperatively. However, they do not provide uniform treatment guidelines regarding specific medication management.29,30
Deep-brain-stimulation (DBS) management: DBS is an effective treatment for advanced PD. There are more than 60,000 patients around the world who have DBS for various conditions, mostly for PD. Therefore, it is increasingly likely that hospitalists will encounter hospitalized patients with advanced PD who are treated both pharmacologically and with DBS. It is important to recognize that stimulation, just like PD medications, cannot be stopped suddenly. If there is any concern of the DBS system malfunctioning (i.e., fracture of the hardware during a fall), the neurologist or neurosurgeon managing the DBS should be contacted immediately. Certain diagnostic tests (MRI) and treatment procedures (diathermy) are contraindicated, and if done inappropriately, can result in permanent brain damage.31,32,33
During surgeries requiring blood-vessel cauterization, DBS should be temporarily turned off. This can be done with the patient’s handheld device or, preferably, by a trained technician usually available through 24/7 technical support services provided by the manufacturer.
Summary of recommendations: There are no clear treatment guidelines regarding the optimal perioperative management of PD patients. The following measures are based on available data and are extensions of routine perioperative management; however, there is no evidence to demonstrate their efficacy in decreasing complication rates among patients with PD:
- A thorough preoperative history and physical examination should include Parkinsonian signs and symptoms, precise medication regimen with doses and timing of intake, effects of medication withdrawal or missed doses, type of surgery planned, and comorbid conditions;
- Depending on symptoms mentioned in the history, consider further testing for dysphagia (preoperative swallow evaluation) and dyspnea (preoperative pulmonary function tests);
- The major goal of medication management in the perioperative period is to continue administration of dopamine replacement therapy as close to the outpatient regimen as possible.
- Titrate down dose of anti-Parkinsonian medications to lowest possible dose prior to surgery if prolonged NPO status is anticipated;
- Ensure medications are administered immediately prior to surgery;
- For short, nonenteric procedures, resume outpatient medication doses and timing of administration as soon as possible postoperatively. For longer, nonenteric surgeries, consider placement of nasogastric tube for medication delivery during procedure and immediately postoperatively;
- If the major limitation of oral medication intake is dysphagia, the use of orally disintegrated formulation CD/LD (parcopa) is helpful;
- For longer enteric surgeries in which patient must be on bowel rest, recommend consultation with neurologist specialized in movement disorders to guide use of intravenous or subcutaneous agents. Transdermal delivery systems of the dopamine agonist rotigotine are in the process of being approved in the U.S. market and might be helpful for this purpose;
- Initiation of tube feeding, when co-administered with levodopa, might result in sudden changes in medication absorption and potential worsening of PD symptoms; feeding should be started slowly and preferably at night when the body’s dopamine requirements are lower;
- Consider use of promotility agents;
- If apomorphine or intravenous LD are not available, consider trial of intravenous anticholinergics or antihistamines, carefully observing for potential cognitive and behavioral side effects;
- Avoid such dopamine antagonists as droperidol, haloperidol, risperidone, metaclopramide, prochlorperazine, or promethazine, as these medications can worsen Parkinsonian symptoms; and
- If patient was on MAO-B inhibitors (selegiline, rasagiline) before surgery, be aware of multiple potential interactions with various medications that are commonly used in perioperative period, including anesthesia agents and certain analgesics, specifically meperidine. These interactions include serotonin syndrome, and can be life-threatening.34
- Psychiatric considerations: delirium precautions;
- Motor considerations: early PT/OT, early referral to inpatient rehabilitation; fall precautions;
- Pulmonary considerations: institute aggressive incentive spirometry, postural drainage, management of respiratory secretions, and breathing exercises; VTE prophylaxis;
- Gastrointestinal considerations: aspiration precautions and prompt speech therapy to evaluate for aspiration and to teach appropriate swallow techniques (chin tuck); institute aggressive bowel regimen; maximize fluids, electrolytes, and avoid narcotics to prevent precipitating or exacerbating ileus;
- Cardiovascular considerations: monitor orthostatic vital signs; fall precautions to avoid syncopal falls; and
- Genitourinary considerations: early urinary catheter removal; vigilance in monitoring for urinary tract infection.
Back to the Case
The patient underwent repair of her fracture, was extubated, and recovered from general anesthesia without incident. She was evaluated in the postanesthesia care unit, at which time she had a slight tremor and mild rigidity. She was immediately given a dose of her CD/LD, and her evening doses of amantadine and ropinirole were resumed. The patient had no significant flare of her Parkinsonian symptoms and did not exhibit any evidence of PHS.
A postoperative consultation was placed for speech therapy, physical therapy, and occupational therapy. She was given low-molecular-weight heparin for VTE prophylaxis and asked to use incentive spirometry. On postoperative day one, she complained of urinary frequency. A urinalysis was consistent with possible infection. She was discharged home on her previous medication regimen, in addition to antibiotics for cystitis.
If the procedure had not been emergent, the patient might have benefited from a preoperative swallow evaluation, given her dysphagia. Consultation with a speech therapist would have ensured that the patient was educated regarding aspiration precautions. Although this patient did not have difficulty with extubation or experience postoperative respiratory failure, abnormal preoperative pulmonary function tests might have prompted the anesthesiologists to consider alternative low-risk techniques (e.g., a local nerve block).
Bottom Line
Perioperative management of patients with Parkinson’s disease requires knowledge of the multisystem disease characteristics that raise perioperative risk and the effects of the medications used to treat PD. To date, no clear treatment guidelines exist for the optimal perioperative management of PD patients.
However, vigilance in detecting possible complications and instituting attentive perioperative care can aid a hospitalist consultant in improving overall care for these patients. TH
Dr. Patel is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Denver VA Medical Center and instructor of medicine at the University of Colorado Denver. Dr. Anderson is a hospitalist at the Denver VA Medical Center and assistant professor of medicine at the University of Colorado Denver. Dr. Klepitskaya is a neurologist and assistant professor of neurology at the University of Colorado Denver.
References
- Ersan T. Perioperative management of the geriatric patient. Web MD website. Available at: http://emedicine.medscape.com/article/285433-overview. Accessed May 10, 2010.
- Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211.
- Ward CD, Gibb WR. Research diagnostic criteria for Parkinson’s disease. Adv Neurol. 1990;53:245-249.
- Stern MB, Koller WC. Parkinsonian Syndromes. New York: Informa Healthcare Publishing; 1993.
- Nutt JG, Wooten GF. Clinical practice. Diagnosis and initial management of Parkinson’s disease. N Engl J Med. 2005;353(10):1021-1027.
- Mueller MC, Jüptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394(3):511-515.
- Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47(8):967-972.
- Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci. 1998;5(2):131-135.
- Neu HC, Connolly JJ Jr., Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Ann Rev Respir Dis. 1967;95(1):33-47.
- Chokroverty S. The spectrum of ventilatory disturbances in movement disorders. In: Chokroverty S, ed. Movement Disorders. Great Neck, New York: PMA Publishing Corp.; 1990:365-392.
- Shill H, Stacy M. Respiratory complications of Parkinson’s disease. Semin Respir Crit Care Med. 2002;23(3):261-265.
- Easdown LJ, Tessler KJ, Minuk J. Upper airway involvement in Parkinson’s disease resulting in postoperative respiratory failure. Can J Anaesth. 1995;42(4):344-347.
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2(2):107-116.
- Castell JA, Johnston BT, Colcher A, Li Q, Gideon RM, Castell DO. Manometric abnormalities of the oesophagus in patients with Parkinson’s disease. Neurogastroenterol Motil. 2001;13(4):361-364.
- Marcason W. What are the primary nutritional issues for a patient with Parkinson’s disease? J Am Diet Assoc. 2009;109(7):1316.
- Djaldetti R, Lev N, Melamed E. Lesions outside the CNS in Parkinson’s disease. Mov Disord. 2009;24(6):793-800.
- Orimo S, Takahashi A, Uchihara T, Mori F, Kakita A, Wakabayashi K, Takahashi H. Degeneration of cardiac sympathetic nerve begins in the early disease process of Parkinson’s disease. Brain Pathol. 2007;17(1):24-30.
- Lemack GE, Dewey RB Jr., Roehrborn CG, O’Suilleabhain PE, Zimmern PE. Questionnaire-based assessment of bladder dysfunction in patients with mild to moderate Parkinson’s disease. Urology. 2000;56(2):250-254.
- Singer C. Urinary dysfunction in Parkinson’s disease. Clin Neurosci. 1998;5(2):78-86.
- Leverenz JB, Quinn JF, Zabetian C, Zhang J, Montine KS, Montine TJ. Cognitive impairment and dementia in patients with Parkinson disease. Curr Top Med Chem. 2009;9(10):903-912.
- Factor SA, Santiago A. Parkinson-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht S, Fahn S, eds. Movement Disorder Emergencies: Diagnosis and Treatment. New York: Humana Press; 2005:29-40.
- Reed AP, Han DG: Intraoperative exacerbation of Parkinson’s disease. Anesth Analg. 1992;75(5):850-853.
- Harada T, Mitsuoka K, Kumagai R, et al. Clinical features of malignant syndrome in Parkinson’s disease and related neurological disorders. Parkinsonism Relat Disord. 2003;9(Suppl 1):S15-23.
- Onofrj M, Thomas A. Acute akinesia in Parkinson disease. Neurology. 2005;64(7):1162-1169.
- Serrano-Dueñas M. Neuroleptic malignant syndrome-like, or—dopaminergic malignant syndrome—due to levodopa therapy withdrawal. Clinical features in 11 patients. Parkinsonism Relat Disord. 2003;9(3):175-178.
- Furuya R, Hirai A, Andoh T, Kudoh I, Okumura F. Successful perioperative management of a patient with Parkinson’s disease by enteral levodopa administration under propofol anesthesia. Anesthesiology. 1998;89(1):261-263.
- Fujii T, Nakabayashi T, Hashimoto S, Kuwano H. Successful perioperative management of patients with Parkinson’s disease following gastrointestinal surgery: report of three cases. Surg Today. 2009;39(9):807-810.
- Gálvez-Jiménez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22(2):367-377.
- Kalenka A, Schwarz A. Anasthesia and Parkinson’s disease: how to manage with new therapies? Curr Opin Anesthesiol. 2009;22(3):419-424.
- Nicholson G, Pereira AC, Hall GM. Parkinson’s disease and anesthesia. Br J Anaesthesia. 2002;89(6):904-916.
- Rezai AR, Baker KB, Tkach JA, et al. Is magnetic resonance imaging safe for patients with neurostimulation systems used for deep brain stimulation? Neurosurgery. 2005:57(5):1056-1062.
- Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery. 2005:57(5):E1063.
- Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology. 2001;56 (10):1384-1386.
- Gordon PH, Frucht SJ. Neuroleptic malignant syndrome in advanced Parkinson’s disease. Mov Disord. 2001;16(5):960-962.
Case
A 67-year-old female with moderately advanced Parkinson’s disease (PD) had a mechanical fall in her home, which resulted in a humeral fracture. The fall occurred in the morning before she was able to take her medications and was related to her difficulty in initiating movements.
On her current regimen, her PD symptoms are controlled. She is able to perform daily living activities independently and ambulates without assistance. She also performs more complex tasks (e.g., cooking and managing her finances). She has not exhibited any symptoms consistent with dementia. She occasionally experiences dyspnea on exertion and dysphagia, but she has not been evaluated for these complaints. She takes carbidopa/levodopa (CD/LD) 25 mg/100 mg four times a day, amantadine 100 mg twice daily, and ropinirole 3 mg three times a day.
She is scheduled for open reduction internal fixation of her fracture; the orthopedic surgeon has requested a perioperative risk assessment and recommendations concerning her medications. How should PD be managed perioperatively?
Overview
Advances in surgical and anesthetic techniques, in combination with an aging population, have contributed to an increasing number of geriatric patients undergoing surgery. As many as 50% of Americans older than 65 will undergo a surgical procedure; hospitalists will comanage many of these patients in the perioperative period.1
Although cardiopulmonary disorders receive a great deal of attention with regard to perioperative risk assessment, other comorbid conditions also contribute to perioperative risk—namely, disorders specific to the elderly population. Parkinson’s disease is one such condition that deserves attention.
PD is a progressive, neurodegenerative condition associated with loss of dopaminergic neurons and the presence of Lewy bodies within the substantia nigra and other areas of the brain and peripheral autonomic nervous system.2 Cardinal clinical features include rigidity, bradykinesia, and resting tremor. A supportive feature is a consistent response to levodopa. Postural instability, cognitive impairment, and autonomic dysfunction usually occur later in the disease.3,4
As the population ages, Parkinson’s disease is becoming more prevalent, affecting approximately 1% of individuals older than 60.5 These patients pose a specific challenge to the hospitalist, not only because the multiorgan system manifestations of PD can raise surgical risk, but also due to the direct effects of dopaminergic medications used to treat PD, lack of a parenteral route for these medications in NPO patients, and the risks associated with abrupt withdrawal of these medications.
Although surgical risk in PD patients has received intermittent attention in surgical, anesthesia, and neurology literature, there is no broad consensus statement or treatment guideline for the perioperative approach.
Literature Review
A retrospective cohort of 51 PD patients undergoing various types of surgery revealed that PD patients have a longer hospital stay than matched cohorts.6 Pepper et al studied a cohort of 234 PD patients in the Veterans Administration population who were undergoing a variety of surgeries.7 They found that patients with PD had a longer acute hospital stay and had higher in-hospital mortality.7 The multisystem manifestations of PD might account for this global increase in perioperative risk.
The following are reviews of organ-system manifestations of PD and their relevance to the perioperative period.
Motor: The motor symptoms of PD place patients at increased risk for falls and might impair their ability to participate in rehabilitation. Mueller et al demonstrated that there was a significantly increased risk of postoperative falls and a higher need for inpatient rehabilitation due to motor difficulties in the PD cohort.6
Pulmonary: PD patients have increased risk of abnormal pulmonary function secondary to rigidity and akinesia. Increased airway resistance and decreased lung elastic recoil lead to obstructive lung disease.8,9 Rigidity of voluntary chest wall and upper airway muscles leads to a restrictive lung disease pattern.8,10 Furthermore, respiratory dyskinesia is a common side effect of levodopa, which can result in restrictive and dyskinetic ventilation.11 As a consequence of disordered respiratory mechanics (especially in combination with disordered swallow mechanics), PD patients are at increased risk of lower respiratory infections. In fact, pneumonia remains the leading cause of mortality among PD patients.11
Not surprisingly, several cohorts have suggested that PD patients undergoing surgical procedures are at higher risk for atelectasis, pneumonia, and postoperative respiratory failure.7,12 Postoperative VTE rates are not statistically different between PD patients and matched cohorts.
Gastrointestinal: Abnor-malities in muscles of the mouth, pharynx, and esophagus account for the dysphagia commonly noted in PD.13 Barium swallow tests are abnormal in 80% or more of PD patients.14 Dysphagia can lead to aspiration, as well as inadequate oral intake, resulting in pneumonia and malnutrition, respectively.15 Dysfunction of the myenteric plexus (evidenced by Lewy Body deposition) accounts for gastrointestinal dysmotility manifested as gastroparesis, ileus, and slow colonic transit, which results in constipation.16
PD patients in the postoperative period are at risk for swallowing difficulties, which increases the risk of aspiration and might delay initiation of oral medications. Gastroparesis threatens appropriate delivery of oral medications for adequate absorption. In addition, postoperative ileus and constipation can pose challenges.
Cardiovascular: Such cardiac sympathetic abnormalities as orthostatic hypotension, postprandial or exercise-induced hypotension, impaired heart rate variability, and dysrhythmias are common in PD.17 Pepper et al found a trend toward increased risk of hypotension and acute myocardial infarction (MI) in PD patients undergoing surgery.7
Genitourinary: Urinary complaints (e.g., nocturia, frequency, urgency, and urge incontinence) are common in PD patients.18 These clinical complaints correspond to involuntary detrusor contractions (detrusor hyperreflexia).19 Pepper et al found an increased risk of postoperative urinary tract infection in PD patients.7
Cognitive: A recognized feature of advanced PD is cognitive impairment. Studies estimate the prevalence of dementia in cohorts of PD patients is from 28% to 44%. PD with dementia has been associated with shortened survival, impaired quality of life, and increased caregiver distress.20 Pepper et al noted a trend toward increased incidence of postoperative delirium in their cohort of 234 PD patients undergoing surgery.7
Medication: Management of anti-Parkinsonian medications in the perioperative period poses unique challenges. These medications’ prodopaminergic effects can lead to hemodynamic compromise and are potentially arrhythmogenic. At the same time, abrupt withdrawal of these medications can lead to a potentially lethal condition called Parkinsonism-hyperpyrexia syndrome (PHS), which is clinically similar to neuroleptic malignant syndrome.21 PHS is characterized by very high fever, extreme muscle rigidity, autonomic instability, altered consciousness, and multiple severe systemic complications (e.g., acute renal failure, disseminated intravascular coagulation, autonomic failure, aspiration pneumonia, and infections). PHS occurs in up to 4% of PD patients; mortality is reported to be from 4% for treated to 20% for untreated episodes.22-24
As many as 30% of patients who survive a PHS episode have worsening of their PD symptoms and never return to their pre-PHS baseline. PHS prevention in hospitalized patients by uninterrupted administration of PD medications should be the goal. Early recognition and aggressive treatment is key to successful recovery.
Furthermore, even brief interruption of medications can lead to decompensation of Parkinsonian symptoms, which not only delays recovery from surgery, but also increases the risk for multisystem complications as discussed above.25 Traditional anti-Parkinsonian medications can only be delivered orally, presenting significant challenges for NPO patients, especially those undergoing enteric surgery requiring bowel rest.
Case reports describe various approaches to medication management in the perioperative period, but no single consensus statement (or treatment guideline) exists. The most common clinical scenarios are:
- Patient undergoes short surgery and is able to take oral medication immediately after the procedure (e.g., orthopedic, eye);
- Patient undergoes more lengthy surgery and will be able to take enteric medications perioperatively (e.g., longer orthopedic surgeries, genitourinary); and
- Patient undergoes lengthy procedure in which they will be required to adhere to bowel rest (e.g., bowel resection).
Depending on the category, the approach to medication management might differ.
Furuya et al describe a 70-year-old male with PD who had previously experienced perioperative complications associated with his medication management, including postoperative rigidity, dysphagia, and difficulty maintaining respiratory secretions.26 These symptoms were reversed with intravenous levodopa. However, the patient experienced hypotension and premature ventricular contractions as side effects of this therapy. This patient was scheduled to undergo hepatic lobectomy. Given his previous complications, Furuya et al provided enteral administration of CD/LD via nasogastric tube every two hours during the surgery, with placement of a duodenostomy for postoperative administration of CD/LD. The patient maintained hemodynamic stability throughout the perioperative period and emerged from anesthesia smoothly with no muscle rigidity or postoperative complications.26
Fujii et al described three cases of PD patients undergoing gastrointestinal surgery. They suggested that the dose of medication required to control symptoms should be minimized before surgery to avoid withdrawal symptoms. They also described the use of intravenous levodopa immediately, postoperatively, while the patient was unable to tolerate enteric medications, and suggested the use of prokinetics to prevent ileus and maximize drug absorption.27
Gálvez-Jiménez et al discuss the limitations of intravenous levodopa, including hemodynamic compromise, need for escalating doses, frequent adjustments to maintain effect, and large amount of fluids required. They propose the use of subcutaneous apomorphine, which is a potent D1/D2 dopamine agonist in conjunction with rectal domeperidone, a D2 antagonist with poor blood-brain-barrier penetration, to counteract the peripheral dopaminergic side effects. The main limitation to this approach is that domeperidone is not available in the U.S.28
Intravenous antihistamines and anticholinergics are readily available. However, they show limited efficacy in halting Parkinsonian symptoms and carry multiple side effects (e.g., confusion, delay in gastrointestinal recovery, and urinary retention).28
Various anesthesiologists agree with administering anti-PD medications immediately, preoperatively, and restarting medications as soon as possible postoperatively. However, they do not provide uniform treatment guidelines regarding specific medication management.29,30
Deep-brain-stimulation (DBS) management: DBS is an effective treatment for advanced PD. There are more than 60,000 patients around the world who have DBS for various conditions, mostly for PD. Therefore, it is increasingly likely that hospitalists will encounter hospitalized patients with advanced PD who are treated both pharmacologically and with DBS. It is important to recognize that stimulation, just like PD medications, cannot be stopped suddenly. If there is any concern of the DBS system malfunctioning (i.e., fracture of the hardware during a fall), the neurologist or neurosurgeon managing the DBS should be contacted immediately. Certain diagnostic tests (MRI) and treatment procedures (diathermy) are contraindicated, and if done inappropriately, can result in permanent brain damage.31,32,33
During surgeries requiring blood-vessel cauterization, DBS should be temporarily turned off. This can be done with the patient’s handheld device or, preferably, by a trained technician usually available through 24/7 technical support services provided by the manufacturer.
Summary of recommendations: There are no clear treatment guidelines regarding the optimal perioperative management of PD patients. The following measures are based on available data and are extensions of routine perioperative management; however, there is no evidence to demonstrate their efficacy in decreasing complication rates among patients with PD:
- A thorough preoperative history and physical examination should include Parkinsonian signs and symptoms, precise medication regimen with doses and timing of intake, effects of medication withdrawal or missed doses, type of surgery planned, and comorbid conditions;
- Depending on symptoms mentioned in the history, consider further testing for dysphagia (preoperative swallow evaluation) and dyspnea (preoperative pulmonary function tests);
- The major goal of medication management in the perioperative period is to continue administration of dopamine replacement therapy as close to the outpatient regimen as possible.
- Titrate down dose of anti-Parkinsonian medications to lowest possible dose prior to surgery if prolonged NPO status is anticipated;
- Ensure medications are administered immediately prior to surgery;
- For short, nonenteric procedures, resume outpatient medication doses and timing of administration as soon as possible postoperatively. For longer, nonenteric surgeries, consider placement of nasogastric tube for medication delivery during procedure and immediately postoperatively;
- If the major limitation of oral medication intake is dysphagia, the use of orally disintegrated formulation CD/LD (parcopa) is helpful;
- For longer enteric surgeries in which patient must be on bowel rest, recommend consultation with neurologist specialized in movement disorders to guide use of intravenous or subcutaneous agents. Transdermal delivery systems of the dopamine agonist rotigotine are in the process of being approved in the U.S. market and might be helpful for this purpose;
- Initiation of tube feeding, when co-administered with levodopa, might result in sudden changes in medication absorption and potential worsening of PD symptoms; feeding should be started slowly and preferably at night when the body’s dopamine requirements are lower;
- Consider use of promotility agents;
- If apomorphine or intravenous LD are not available, consider trial of intravenous anticholinergics or antihistamines, carefully observing for potential cognitive and behavioral side effects;
- Avoid such dopamine antagonists as droperidol, haloperidol, risperidone, metaclopramide, prochlorperazine, or promethazine, as these medications can worsen Parkinsonian symptoms; and
- If patient was on MAO-B inhibitors (selegiline, rasagiline) before surgery, be aware of multiple potential interactions with various medications that are commonly used in perioperative period, including anesthesia agents and certain analgesics, specifically meperidine. These interactions include serotonin syndrome, and can be life-threatening.34
- Psychiatric considerations: delirium precautions;
- Motor considerations: early PT/OT, early referral to inpatient rehabilitation; fall precautions;
- Pulmonary considerations: institute aggressive incentive spirometry, postural drainage, management of respiratory secretions, and breathing exercises; VTE prophylaxis;
- Gastrointestinal considerations: aspiration precautions and prompt speech therapy to evaluate for aspiration and to teach appropriate swallow techniques (chin tuck); institute aggressive bowel regimen; maximize fluids, electrolytes, and avoid narcotics to prevent precipitating or exacerbating ileus;
- Cardiovascular considerations: monitor orthostatic vital signs; fall precautions to avoid syncopal falls; and
- Genitourinary considerations: early urinary catheter removal; vigilance in monitoring for urinary tract infection.
Back to the Case
The patient underwent repair of her fracture, was extubated, and recovered from general anesthesia without incident. She was evaluated in the postanesthesia care unit, at which time she had a slight tremor and mild rigidity. She was immediately given a dose of her CD/LD, and her evening doses of amantadine and ropinirole were resumed. The patient had no significant flare of her Parkinsonian symptoms and did not exhibit any evidence of PHS.
A postoperative consultation was placed for speech therapy, physical therapy, and occupational therapy. She was given low-molecular-weight heparin for VTE prophylaxis and asked to use incentive spirometry. On postoperative day one, she complained of urinary frequency. A urinalysis was consistent with possible infection. She was discharged home on her previous medication regimen, in addition to antibiotics for cystitis.
If the procedure had not been emergent, the patient might have benefited from a preoperative swallow evaluation, given her dysphagia. Consultation with a speech therapist would have ensured that the patient was educated regarding aspiration precautions. Although this patient did not have difficulty with extubation or experience postoperative respiratory failure, abnormal preoperative pulmonary function tests might have prompted the anesthesiologists to consider alternative low-risk techniques (e.g., a local nerve block).
Bottom Line
Perioperative management of patients with Parkinson’s disease requires knowledge of the multisystem disease characteristics that raise perioperative risk and the effects of the medications used to treat PD. To date, no clear treatment guidelines exist for the optimal perioperative management of PD patients.
However, vigilance in detecting possible complications and instituting attentive perioperative care can aid a hospitalist consultant in improving overall care for these patients. TH
Dr. Patel is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Denver VA Medical Center and instructor of medicine at the University of Colorado Denver. Dr. Anderson is a hospitalist at the Denver VA Medical Center and assistant professor of medicine at the University of Colorado Denver. Dr. Klepitskaya is a neurologist and assistant professor of neurology at the University of Colorado Denver.
References
- Ersan T. Perioperative management of the geriatric patient. Web MD website. Available at: http://emedicine.medscape.com/article/285433-overview. Accessed May 10, 2010.
- Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211.
- Ward CD, Gibb WR. Research diagnostic criteria for Parkinson’s disease. Adv Neurol. 1990;53:245-249.
- Stern MB, Koller WC. Parkinsonian Syndromes. New York: Informa Healthcare Publishing; 1993.
- Nutt JG, Wooten GF. Clinical practice. Diagnosis and initial management of Parkinson’s disease. N Engl J Med. 2005;353(10):1021-1027.
- Mueller MC, Jüptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394(3):511-515.
- Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47(8):967-972.
- Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci. 1998;5(2):131-135.
- Neu HC, Connolly JJ Jr., Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Ann Rev Respir Dis. 1967;95(1):33-47.
- Chokroverty S. The spectrum of ventilatory disturbances in movement disorders. In: Chokroverty S, ed. Movement Disorders. Great Neck, New York: PMA Publishing Corp.; 1990:365-392.
- Shill H, Stacy M. Respiratory complications of Parkinson’s disease. Semin Respir Crit Care Med. 2002;23(3):261-265.
- Easdown LJ, Tessler KJ, Minuk J. Upper airway involvement in Parkinson’s disease resulting in postoperative respiratory failure. Can J Anaesth. 1995;42(4):344-347.
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2(2):107-116.
- Castell JA, Johnston BT, Colcher A, Li Q, Gideon RM, Castell DO. Manometric abnormalities of the oesophagus in patients with Parkinson’s disease. Neurogastroenterol Motil. 2001;13(4):361-364.
- Marcason W. What are the primary nutritional issues for a patient with Parkinson’s disease? J Am Diet Assoc. 2009;109(7):1316.
- Djaldetti R, Lev N, Melamed E. Lesions outside the CNS in Parkinson’s disease. Mov Disord. 2009;24(6):793-800.
- Orimo S, Takahashi A, Uchihara T, Mori F, Kakita A, Wakabayashi K, Takahashi H. Degeneration of cardiac sympathetic nerve begins in the early disease process of Parkinson’s disease. Brain Pathol. 2007;17(1):24-30.
- Lemack GE, Dewey RB Jr., Roehrborn CG, O’Suilleabhain PE, Zimmern PE. Questionnaire-based assessment of bladder dysfunction in patients with mild to moderate Parkinson’s disease. Urology. 2000;56(2):250-254.
- Singer C. Urinary dysfunction in Parkinson’s disease. Clin Neurosci. 1998;5(2):78-86.
- Leverenz JB, Quinn JF, Zabetian C, Zhang J, Montine KS, Montine TJ. Cognitive impairment and dementia in patients with Parkinson disease. Curr Top Med Chem. 2009;9(10):903-912.
- Factor SA, Santiago A. Parkinson-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht S, Fahn S, eds. Movement Disorder Emergencies: Diagnosis and Treatment. New York: Humana Press; 2005:29-40.
- Reed AP, Han DG: Intraoperative exacerbation of Parkinson’s disease. Anesth Analg. 1992;75(5):850-853.
- Harada T, Mitsuoka K, Kumagai R, et al. Clinical features of malignant syndrome in Parkinson’s disease and related neurological disorders. Parkinsonism Relat Disord. 2003;9(Suppl 1):S15-23.
- Onofrj M, Thomas A. Acute akinesia in Parkinson disease. Neurology. 2005;64(7):1162-1169.
- Serrano-Dueñas M. Neuroleptic malignant syndrome-like, or—dopaminergic malignant syndrome—due to levodopa therapy withdrawal. Clinical features in 11 patients. Parkinsonism Relat Disord. 2003;9(3):175-178.
- Furuya R, Hirai A, Andoh T, Kudoh I, Okumura F. Successful perioperative management of a patient with Parkinson’s disease by enteral levodopa administration under propofol anesthesia. Anesthesiology. 1998;89(1):261-263.
- Fujii T, Nakabayashi T, Hashimoto S, Kuwano H. Successful perioperative management of patients with Parkinson’s disease following gastrointestinal surgery: report of three cases. Surg Today. 2009;39(9):807-810.
- Gálvez-Jiménez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22(2):367-377.
- Kalenka A, Schwarz A. Anasthesia and Parkinson’s disease: how to manage with new therapies? Curr Opin Anesthesiol. 2009;22(3):419-424.
- Nicholson G, Pereira AC, Hall GM. Parkinson’s disease and anesthesia. Br J Anaesthesia. 2002;89(6):904-916.
- Rezai AR, Baker KB, Tkach JA, et al. Is magnetic resonance imaging safe for patients with neurostimulation systems used for deep brain stimulation? Neurosurgery. 2005:57(5):1056-1062.
- Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery. 2005:57(5):E1063.
- Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology. 2001;56 (10):1384-1386.
- Gordon PH, Frucht SJ. Neuroleptic malignant syndrome in advanced Parkinson’s disease. Mov Disord. 2001;16(5):960-962.
In the Literature: HM-Related Research You Need to Know
In This Edition:
- Catheter ablation for atrial fibrillation
- Noncontrast CT for appendicitis diagnosis
- Effectiveness of whole-leg ultrasound for DVT diagnosis
- Physician localization and nurse-physician communication
- PPI versus H2 blocker in patients with aspirin-related peptic ulcer disease
- Intensive insulin therapy in steroid-induced hyperglycemia associated with septic shock
- S. aureus decolonization and surgical-site infections
- ABCD2 score and risk of subsequent stroke
Catheter Ablation of Paroxysmal AF Might Be More Effective than Antiarrhythmic Therapy
Clinical question: In patients with paroxysmal atrial fibrillation (AF) unresponsive to initial antiarrhythmic therapy, what is the efficacy of catheter ablation compared with continued attempts at rhythm control?
Background: Although antiarrhythmic drug therapy (ADT) is generally first-line AF therapy, AF recurrence is high and treatment is associated with adverse effects. Catheter ablation is an alternative treatment. Recent studies comparing antiarrhythmic drugs and catheter ablation have involved small populations and have shown mixed results.
Study design: Prospective, multicenter, unblinded, randomized trial.
Setting: Nineteen hospitals—15 in the U.S.—with considerable experience in AF ablation.
Synopsis: This trial compared catheter ablation (n=106) vs. ADT (n=61) for symptomatic, paroxysmal AF refractory to at least one antiarrhythmic drug. Patients in the ablation arm were allowed up to three procedures within an 80-day period; patients in the ADT arm were treated with a previously unused class I or class III antiarrhythmic. After nine months, 34% of ablated patients had failed treatment compared with 84% of patients receiving ADT (HR 0.30 [95% CI, 0.19-0.47]).
While the results are encouraging, some limitations should be noted. Attempts to generalize the results of this study might be limited, as the hospitals had considerable experience in AF ablation, the patient population was relatively young (mean age=56 years), and patients with significant left ventrical dysfunction and persistent AF were excluded.
Furthermore, the long-term effectiveness of ablation was not evaluated, and the study did not assess such outcomes as mortality, stroke, or AF progression. The effectiveness of specific ablation techniques could not be determined because a variety of approaches was employed.
Bottom line: Patients with symptomatic paroxysmal AF without advanced heart failure might benefit from catheter ablation at experienced institutions.
Citation: Wilber DJ, Pappone C, Neuzil P, et al. Comparison of antiarrhythmic drug therapy and radiofrequency catheter ablation in patients with paroxysmal atrial fibrillation: a randomized controlled trial. JAMA. 2010;303(4):333-340.
Noncontrast CT Can Be Used to Evaluate Suspected Appendicitis
Clinical question: What is the diagnostic accuracy of noncontrast helical CT in the evaluation of suspected acute appendicitis in the ED?
Background: In the workup of acute appendicitis, various CT protocols are used, including combinations of oral, intravenous, and rectal contrast, as well as noncontrast protocols. Noncontrast CT is less time-consuming and avoids risk of allergic reaction or contrast-induced nephropathy. The diagnostic accuracy of noncontrast CT, however, is controversial.
Study design: Systematic review/meta-analysis.
Setting: Seven studies evaluating acute appendicitis with noncontrast CT in ED settings.
Synopsis: This is the first systematic review of noncontrast CT (helical/multislice) in adults with suspected appendicitis. The authors pooled seven studies (1,060 patients) comparing noncontrast CT with a reference standard of final diagnosis at surgery or followup at a minimum of two weeks. The review yielded the following pooled estimates: sensitivity 93%, specificity 96%, positive likelihood ratio 24, and negative likelihood ratio 0.08. Overall, the diagnostic accuracy of noncontrast CT was high.
A few limitations should be noted. In the original studies, results were reported inconsistently. In one study, 24% of scans were inconclusive, with an associated likelihood ratio of approximately 1. Future studies must address this important clinical question of how to treat patients with inconclusive scans. Another question is to what degree the accuracy reported in this study reflects the expertise of the institutions, the majority of which were university-affiliated. According to one study author, “a certain level of experience is required for skillful interpretation” of noncontrast CT.
Bottom line: Noncontrast CT has a high sensitivity and specificity for acute appendicitis, and should be considered an alternative to contrast CT, particularly in patients with contraindications to contrast or those at risk of contrast-induced nephropathy.
Citation: Hlibczuk V, Dattaro JA, Jin Z, Falzon L, Brown MD. Diagnostic accuracy of noncontrast computed tomography for appendicitis in adults: a systematic review. Ann Emerg Med. 2010;55(1):51-59.e1.
Whole-Leg Compression Ultrasound for DVT Has a Low Failure Rate in Outpatients
Clinical question: How useful is compression ultrasound (CUS) for diagnosing distal DVT?
Background: CUS can reliably evaluate proximal DVT, but its accuracy for distal DVT is controversial. Because of the risk of extension of distal DVT (up to 25%), guidelines recommend that some patients undergo serial proximal CUS after an initial negative result. As an alternative, recent studies have evaluated one-time, whole-leg CUS.
Study design: Systematic review and meta-analysis.
Setting: Review of randomized controlled trials and prospective cohort studies.
Synopsis: The study pooled data from seven studies and more than 4,700 patients with suspected DVT for whom anticoagulation was withheld after a single, negative, whole-leg CUS. At the three-month followup, the combined symptomatic VTE event rate was 0.57%, and the authors concluded that withholding anticoagulation was associated with a low VTE risk.
Although encouraging, this study had several limitations. First, whole-leg CUS is not widely performed or standardized, and the CUS technique varied slightly across the studies. Second, any attempt to generalize the results of this study might be limited, as the proportion of pregnant patients and those with malignancy was low. Furthermore, only one of the seven studies included inpatients that might be at higher VTE risk. Third, pre-test probability was assessed for only a subset of patients, limiting assessment of VTE by risk level. For example, the overall finding of the study—an event rate of 0.57%—appears low but is difficult to apply clinically when subset analyses for high-risk patients yielded a VTE rate of 2.5% with wide confidence intervals ranging from 0% to 7%.
Bottom line: Whole-leg CUS might be a practical alternative to serial proximal CUS, but more data incorporating pre-test probabilities and involving more inpatients are needed. Hospitalists should be cautious in applying pooled summary estimates.
Citation: Johnson SA, Stevens SM, Woller SC, et al. Risk of deep vein thrombosis following a single negative whole-leg compression ultrasound: a systematic review and meta-analysis. JAMA. 2010;303(5):438-445.
Localization of Inpatient Physicians on Hospital Units Increases Provider Communication Frequency
Clinical question: Does localization of inpatient physicians on hospital units improve communication with nursing staff?
Background: While nurses are typically localized on a hospital unit, inpatient physicians often care for patients on multiple units. This lack of regionalization makes it difficult for physicians and nurses to discuss care plans directly. No prior research has evaluated the effect of physician localization on nurse-physician communication.
Study design: Cross-sectional, pre- and postintervention study.
Setting: Tertiary-care teaching hospital, general medical service.
Synopsis: The study was a cross-sectional survey of nurses and physicians prelocalization (n=342 patients) and postlocalization (n=294 patients) of physicians on hospital units. Localization was associated with increased frequency of communication; however, it did not improve the consistency of nurse-physician agreement on the care plan. Nurse-physician agreement was improved on two aspects of the care plan—planned tests and anticipated length of stay—but not on primary diagnosis, planned procedures, medication changes, or consultations.
Limitations of the study were that it was conducted at a single teaching hospital, communication patterns might have changed during the year between pre- and postlocalization, and physicians were not completely localized to specific units (73% localization).
Despite the limitations, this study is the first to evaluate staff localization and communication on a general medical service. The findings suggest that localization is a first step toward interdisciplinary communication. It also shows that quality and content of communication require further assessment.
Future studies must assess the impact of communication on the quality of patient care.
Bottom line: Physician localization improved the frequency of nurse-physician dialogue but did not consistently facilitate a shared understanding of the care plan. Although not assessed in this study, the implication is that the quality of communication between providers needs improvement.
Citation: O’Leary KJ, Wayne DB, Landler MP, et al. J Gen Int Med. 2009;24(11):1223-1227.
Pantoprazole Better than Double-Dose Famotidine in Secondary Prevention of Aspirin-Related Injury
Clinical question: Is a twice-daily H2-receptor antagonist (H2RA) or a once-daily proton pump inhibitor (PPI) better in patients who must continue low-dose-aspirin therapy despite aspirin-related peptic ulcer disease?
Background: Some patients with aspirin-related peptic ulcer disease require continued aspirin therapy. It often is assumed that PPIs are superior to H2RAs in secondary prevention of low-dose aspirin-related injury, although no randomized trials have specifically addressed this question.
Study design: A prospective, double-blind, randomized controlled trial.
Setting: A university hospital in Hong Kong.
Synopsis: In this trial, 160 patients with aspirin-related peptic ulcers/erosions were randomized to 48 weeks of oral famotidine (40 mg twice daily) or pantoprazole (20 mg daily) after mucosal healing and eradication of H pylori. During this time, all patients continued to receive aspirin (80 mg daily).
The rates of recurrent dyspeptic or bleeding ulcers/erosions within 48 weeks were 20% with famotidine versus 0% with pantoprazole. The rates of gastrointestinal (GI) bleeding alone were 7.7% versus 0%, respectively.
Of note, none of the five patients with GI bleeding had significant dyspepsia, which is consistent with prior reports that NSAID-induced injury might be silent.
Overall, high-dose famotidine was inferior to pantoprazole in the prevention of recurrent aspirin-related injury. These findings support the ACCF/ACG/AHA 2008 recommendation of PPIs over double-dose H2RAs in this clinical setting. It is not known, however, whether the PPI strategy extends to average GI-risk patients taking aspirin.
Furthermore, although not demonstrated in this study, it is possible that the lower rates of dyspepsia with pantoprazole might facilitate adherence to prolonged aspirin therapy.
Bottom line: High-dose famotidine was inferior to pantoprazole in the prevention of recurrent low-dose-aspirin-related injury.
Citation: Ng FH, Wong SY, Lam KF, et al. Famotidine is inferior to pantoprazole in preventing recurrence of aspirin-related peptic ulcers or erosions. Gastroenterology. 2010;138
(1):82-88.
Intensive Insulin Therapy Is Not Beneficial in Corticosteroid-Induced Hyperglycemia Associated with Septic Shock
Clinical question: In septic shock treated with hydrocortisone, does intensive insulin therapy reduce in-hospital mortality compared with conventional glucose management?
Background: Corticosteroids might benefit patients with septic shock, but they also can exacerbate illness-induced hyperglycemia. It is hypothesized that normalization of blood glucose with intensive insulin might improve outcomes in these patients.
Study design: A multicenter, 2x2 factorial, randomized controlled trial.
Setting: Eleven ICUs in France.
Synopsis: In this 2x2 factorial comparison, 509 patients with septic shock treated with hydrocortisone were randomized to IV insulin, conventional insulin, fludrocortisone plus IV insulin, and fludrocortisone plus conventional insulin. The primary objective was to assess intensive IV versus conventional insulin, and a secondary objective was to assess the benefit of adding fludrocortisone to hydrocortisone therapy.
Overall, analysis showed no difference in in-hospital mortality in either of the two comparisons.
These null findings should be interpreted considering the following study limitations. In the intensive-insulin groups, there was a failure to reach target glucose levels of 80 mg/dL to 110 mg/dL (mean achieved levels 120 mg/dL to 130 mg/dL and higher). These levels overlapped to some degree with the control group, which targeted glucose levels <150 mg/dL and achieved mean levels of 140 mg/dL to 150 mg/dL.
The lack of substantial difference in glucose levels might have contributed to the null findings. Furthermore, the absolute risk reduction in the original sample-size calculations was overestimated. The result was an underpowered study, which also might have contributed to the null findings.
Bottom line: In septic shock treated with hydrocortisone, the optimal blood-glucose level and insulin strategy are unknown.
Citation: Annane D, Cariou A, Maxime V, et al. Corticosteroid treatment and intensive insulin therapy for septic shock in adults: a randomized controlled trial. JAMA. 2010;303(4):341-348.
Mupirocin-Chlorhexidine Decolonization of Nasal S. Aureus Carriers Reduces Surgical-Site Infections
Clinical question: Can rapid screening and decolonization of nasal carriers of Staphylococcus aureus on admission reduce surgical-site S. aureus infections?
Background: More than 80% of healthcare-associated (HCA) S. aureus infections are endogenous in origin. High-level nasal carriers have a three- to sixfold increased risk of infection with this organism compared with noncarriers and low-level carriers. Decolonization of nasal and extranasal S. aureus on admission might reduce this risk of infection.
Study design: Randomized, double-blinded, placebo-controlled, multicenter trial.
Setting: Three university hospitals and two general hospitals in the Netherlands.
Synopsis: In this study, 918 mostly surgical patients with nasal S. aureus identified preoperatively by PCR tests were randomized to decolonization versus placebo. The five-day decolonization protocol involved mupirocin nasal ointment and chlorhexidine soap baths. Decolonization reduced length of stay by nearly two days. Through six weeks postdischarge, the cumulative incidence of S. aureus infection was 3.4% after decolonization versus 7.7% with placebo (RR 0.42; 95% CI, 0.23-0.75). Among the sites of infection, deep surgical sites had the greatest risk reduction (RR 0.21; 95% CI, 0.07-0.62).
The results of this study are encouraging, but a few limitations should be noted. The decolonization protocol lasted five days, which might make implementation less practical. Also, the relative contributions of mupirocin and chlorhexidine are unclear.
S. aureus is important, but it represents a minority of surgical-site infections; the effect of the protocol on other organisms is unknown. Lastly, MRSA is not prevalent in the Netherlands and no carriers were identified in the study. Although the protocol was designed to eradicate MRSA, such carriers might have different carriage patterns requiring throat swabs in addition to nasal swabs.
Bottom line: Preoperative detection of S. aureus nasal carriage and nasal and extranasal decolonization significantly reduced endogenous S. aureus infection and length of stay. Decolonization might be most beneficial for carriers at increased risk of deep infection, such as those undergoing cardiac surgery.
Citation: Bode LG, Kluytmans JA, Wertheim HF, et al. Preventing surgical-site infections in nasal carriers of Staphylococcus aureus. N Engl J Med. 2010;362(1):9-17.
ABCD2 Is a Poor Predictor of Early Ischemic Stroke after Transient Ischemic Attack
Clinical question: How well does the ABCD2 score predict stroke risk within seven days of transient ischemic attack (TIA)?
Background: After TIA diagnosis, the seven-day risk of ischemic stroke is approximately 5%. Identifying these high-risk patients might facilitate ED decision-making. The ABCD2 score, a prediction tool for stroke after TIA, might be useful but has not been prospectively validated in a large, broad-patient population.
Study design: Prospective validation study, convenience sample.
Setting: Sixteen hospitals in North Carolina.
Synopsis: The ABCD2 score (range 0-7 points) predicts stroke risk after TIA. The investigators evaluated the accuracy of ABCD2 in predicting seven-day ischemic stroke risk in a convenience sample of 1,667 TIA patients. Strokes were categorized as disabling or nondisabling.
Overall, the score was poorly predictive of all ischemic stroke (c stat 0.59) and moderately predictive of the subset of disabling ischemic stroke (c stat 0.71). The ABCD2 had the most discriminatory power when used to identify patients at low risk of disabling stroke (0-3 points); for these patients, the negative likelihood ratio (LR) was 0.16 (0.04-0.64).
The study is the largest published external-validation study of the ABCD2 score, but it had significant limitations that should be considered. There was potential sampling bias because of nonconsecutive sampling, and unaccounted patients with TIA were discharged from the ED. Furthermore, ABCD2 scores were incalculable for 35% of patients, although the authors report that imputed data did not change the findings significantly.
Bottom line: The ABCD2 score does not sufficiently predict the seven-day risk of ischemic stroke after TIA. Further validation studies are needed.
Citation: Asimos AW, Johnson AM, Rosamond WD, et al. A multicenter evaluation of the ABCD2 score’s accuracy for predicting early ischemic stroke in admitted patients with transient ischemic attack. Ann Emerg Med. 2010;55(2):201-210.e5. TH
Dr. Kim is a hospitalist at Brigham and Women’s Hospital in Boston, and an instructor at Harvard Medical School.
In This Edition:
- Catheter ablation for atrial fibrillation
- Noncontrast CT for appendicitis diagnosis
- Effectiveness of whole-leg ultrasound for DVT diagnosis
- Physician localization and nurse-physician communication
- PPI versus H2 blocker in patients with aspirin-related peptic ulcer disease
- Intensive insulin therapy in steroid-induced hyperglycemia associated with septic shock
- S. aureus decolonization and surgical-site infections
- ABCD2 score and risk of subsequent stroke
Catheter Ablation of Paroxysmal AF Might Be More Effective than Antiarrhythmic Therapy
Clinical question: In patients with paroxysmal atrial fibrillation (AF) unresponsive to initial antiarrhythmic therapy, what is the efficacy of catheter ablation compared with continued attempts at rhythm control?
Background: Although antiarrhythmic drug therapy (ADT) is generally first-line AF therapy, AF recurrence is high and treatment is associated with adverse effects. Catheter ablation is an alternative treatment. Recent studies comparing antiarrhythmic drugs and catheter ablation have involved small populations and have shown mixed results.
Study design: Prospective, multicenter, unblinded, randomized trial.
Setting: Nineteen hospitals—15 in the U.S.—with considerable experience in AF ablation.
Synopsis: This trial compared catheter ablation (n=106) vs. ADT (n=61) for symptomatic, paroxysmal AF refractory to at least one antiarrhythmic drug. Patients in the ablation arm were allowed up to three procedures within an 80-day period; patients in the ADT arm were treated with a previously unused class I or class III antiarrhythmic. After nine months, 34% of ablated patients had failed treatment compared with 84% of patients receiving ADT (HR 0.30 [95% CI, 0.19-0.47]).
While the results are encouraging, some limitations should be noted. Attempts to generalize the results of this study might be limited, as the hospitals had considerable experience in AF ablation, the patient population was relatively young (mean age=56 years), and patients with significant left ventrical dysfunction and persistent AF were excluded.
Furthermore, the long-term effectiveness of ablation was not evaluated, and the study did not assess such outcomes as mortality, stroke, or AF progression. The effectiveness of specific ablation techniques could not be determined because a variety of approaches was employed.
Bottom line: Patients with symptomatic paroxysmal AF without advanced heart failure might benefit from catheter ablation at experienced institutions.
Citation: Wilber DJ, Pappone C, Neuzil P, et al. Comparison of antiarrhythmic drug therapy and radiofrequency catheter ablation in patients with paroxysmal atrial fibrillation: a randomized controlled trial. JAMA. 2010;303(4):333-340.
Noncontrast CT Can Be Used to Evaluate Suspected Appendicitis
Clinical question: What is the diagnostic accuracy of noncontrast helical CT in the evaluation of suspected acute appendicitis in the ED?
Background: In the workup of acute appendicitis, various CT protocols are used, including combinations of oral, intravenous, and rectal contrast, as well as noncontrast protocols. Noncontrast CT is less time-consuming and avoids risk of allergic reaction or contrast-induced nephropathy. The diagnostic accuracy of noncontrast CT, however, is controversial.
Study design: Systematic review/meta-analysis.
Setting: Seven studies evaluating acute appendicitis with noncontrast CT in ED settings.
Synopsis: This is the first systematic review of noncontrast CT (helical/multislice) in adults with suspected appendicitis. The authors pooled seven studies (1,060 patients) comparing noncontrast CT with a reference standard of final diagnosis at surgery or followup at a minimum of two weeks. The review yielded the following pooled estimates: sensitivity 93%, specificity 96%, positive likelihood ratio 24, and negative likelihood ratio 0.08. Overall, the diagnostic accuracy of noncontrast CT was high.
A few limitations should be noted. In the original studies, results were reported inconsistently. In one study, 24% of scans were inconclusive, with an associated likelihood ratio of approximately 1. Future studies must address this important clinical question of how to treat patients with inconclusive scans. Another question is to what degree the accuracy reported in this study reflects the expertise of the institutions, the majority of which were university-affiliated. According to one study author, “a certain level of experience is required for skillful interpretation” of noncontrast CT.
Bottom line: Noncontrast CT has a high sensitivity and specificity for acute appendicitis, and should be considered an alternative to contrast CT, particularly in patients with contraindications to contrast or those at risk of contrast-induced nephropathy.
Citation: Hlibczuk V, Dattaro JA, Jin Z, Falzon L, Brown MD. Diagnostic accuracy of noncontrast computed tomography for appendicitis in adults: a systematic review. Ann Emerg Med. 2010;55(1):51-59.e1.
Whole-Leg Compression Ultrasound for DVT Has a Low Failure Rate in Outpatients
Clinical question: How useful is compression ultrasound (CUS) for diagnosing distal DVT?
Background: CUS can reliably evaluate proximal DVT, but its accuracy for distal DVT is controversial. Because of the risk of extension of distal DVT (up to 25%), guidelines recommend that some patients undergo serial proximal CUS after an initial negative result. As an alternative, recent studies have evaluated one-time, whole-leg CUS.
Study design: Systematic review and meta-analysis.
Setting: Review of randomized controlled trials and prospective cohort studies.
Synopsis: The study pooled data from seven studies and more than 4,700 patients with suspected DVT for whom anticoagulation was withheld after a single, negative, whole-leg CUS. At the three-month followup, the combined symptomatic VTE event rate was 0.57%, and the authors concluded that withholding anticoagulation was associated with a low VTE risk.
Although encouraging, this study had several limitations. First, whole-leg CUS is not widely performed or standardized, and the CUS technique varied slightly across the studies. Second, any attempt to generalize the results of this study might be limited, as the proportion of pregnant patients and those with malignancy was low. Furthermore, only one of the seven studies included inpatients that might be at higher VTE risk. Third, pre-test probability was assessed for only a subset of patients, limiting assessment of VTE by risk level. For example, the overall finding of the study—an event rate of 0.57%—appears low but is difficult to apply clinically when subset analyses for high-risk patients yielded a VTE rate of 2.5% with wide confidence intervals ranging from 0% to 7%.
Bottom line: Whole-leg CUS might be a practical alternative to serial proximal CUS, but more data incorporating pre-test probabilities and involving more inpatients are needed. Hospitalists should be cautious in applying pooled summary estimates.
Citation: Johnson SA, Stevens SM, Woller SC, et al. Risk of deep vein thrombosis following a single negative whole-leg compression ultrasound: a systematic review and meta-analysis. JAMA. 2010;303(5):438-445.
Localization of Inpatient Physicians on Hospital Units Increases Provider Communication Frequency
Clinical question: Does localization of inpatient physicians on hospital units improve communication with nursing staff?
Background: While nurses are typically localized on a hospital unit, inpatient physicians often care for patients on multiple units. This lack of regionalization makes it difficult for physicians and nurses to discuss care plans directly. No prior research has evaluated the effect of physician localization on nurse-physician communication.
Study design: Cross-sectional, pre- and postintervention study.
Setting: Tertiary-care teaching hospital, general medical service.
Synopsis: The study was a cross-sectional survey of nurses and physicians prelocalization (n=342 patients) and postlocalization (n=294 patients) of physicians on hospital units. Localization was associated with increased frequency of communication; however, it did not improve the consistency of nurse-physician agreement on the care plan. Nurse-physician agreement was improved on two aspects of the care plan—planned tests and anticipated length of stay—but not on primary diagnosis, planned procedures, medication changes, or consultations.
Limitations of the study were that it was conducted at a single teaching hospital, communication patterns might have changed during the year between pre- and postlocalization, and physicians were not completely localized to specific units (73% localization).
Despite the limitations, this study is the first to evaluate staff localization and communication on a general medical service. The findings suggest that localization is a first step toward interdisciplinary communication. It also shows that quality and content of communication require further assessment.
Future studies must assess the impact of communication on the quality of patient care.
Bottom line: Physician localization improved the frequency of nurse-physician dialogue but did not consistently facilitate a shared understanding of the care plan. Although not assessed in this study, the implication is that the quality of communication between providers needs improvement.
Citation: O’Leary KJ, Wayne DB, Landler MP, et al. J Gen Int Med. 2009;24(11):1223-1227.
Pantoprazole Better than Double-Dose Famotidine in Secondary Prevention of Aspirin-Related Injury
Clinical question: Is a twice-daily H2-receptor antagonist (H2RA) or a once-daily proton pump inhibitor (PPI) better in patients who must continue low-dose-aspirin therapy despite aspirin-related peptic ulcer disease?
Background: Some patients with aspirin-related peptic ulcer disease require continued aspirin therapy. It often is assumed that PPIs are superior to H2RAs in secondary prevention of low-dose aspirin-related injury, although no randomized trials have specifically addressed this question.
Study design: A prospective, double-blind, randomized controlled trial.
Setting: A university hospital in Hong Kong.
Synopsis: In this trial, 160 patients with aspirin-related peptic ulcers/erosions were randomized to 48 weeks of oral famotidine (40 mg twice daily) or pantoprazole (20 mg daily) after mucosal healing and eradication of H pylori. During this time, all patients continued to receive aspirin (80 mg daily).
The rates of recurrent dyspeptic or bleeding ulcers/erosions within 48 weeks were 20% with famotidine versus 0% with pantoprazole. The rates of gastrointestinal (GI) bleeding alone were 7.7% versus 0%, respectively.
Of note, none of the five patients with GI bleeding had significant dyspepsia, which is consistent with prior reports that NSAID-induced injury might be silent.
Overall, high-dose famotidine was inferior to pantoprazole in the prevention of recurrent aspirin-related injury. These findings support the ACCF/ACG/AHA 2008 recommendation of PPIs over double-dose H2RAs in this clinical setting. It is not known, however, whether the PPI strategy extends to average GI-risk patients taking aspirin.
Furthermore, although not demonstrated in this study, it is possible that the lower rates of dyspepsia with pantoprazole might facilitate adherence to prolonged aspirin therapy.
Bottom line: High-dose famotidine was inferior to pantoprazole in the prevention of recurrent low-dose-aspirin-related injury.
Citation: Ng FH, Wong SY, Lam KF, et al. Famotidine is inferior to pantoprazole in preventing recurrence of aspirin-related peptic ulcers or erosions. Gastroenterology. 2010;138
(1):82-88.
Intensive Insulin Therapy Is Not Beneficial in Corticosteroid-Induced Hyperglycemia Associated with Septic Shock
Clinical question: In septic shock treated with hydrocortisone, does intensive insulin therapy reduce in-hospital mortality compared with conventional glucose management?
Background: Corticosteroids might benefit patients with septic shock, but they also can exacerbate illness-induced hyperglycemia. It is hypothesized that normalization of blood glucose with intensive insulin might improve outcomes in these patients.
Study design: A multicenter, 2x2 factorial, randomized controlled trial.
Setting: Eleven ICUs in France.
Synopsis: In this 2x2 factorial comparison, 509 patients with septic shock treated with hydrocortisone were randomized to IV insulin, conventional insulin, fludrocortisone plus IV insulin, and fludrocortisone plus conventional insulin. The primary objective was to assess intensive IV versus conventional insulin, and a secondary objective was to assess the benefit of adding fludrocortisone to hydrocortisone therapy.
Overall, analysis showed no difference in in-hospital mortality in either of the two comparisons.
These null findings should be interpreted considering the following study limitations. In the intensive-insulin groups, there was a failure to reach target glucose levels of 80 mg/dL to 110 mg/dL (mean achieved levels 120 mg/dL to 130 mg/dL and higher). These levels overlapped to some degree with the control group, which targeted glucose levels <150 mg/dL and achieved mean levels of 140 mg/dL to 150 mg/dL.
The lack of substantial difference in glucose levels might have contributed to the null findings. Furthermore, the absolute risk reduction in the original sample-size calculations was overestimated. The result was an underpowered study, which also might have contributed to the null findings.
Bottom line: In septic shock treated with hydrocortisone, the optimal blood-glucose level and insulin strategy are unknown.
Citation: Annane D, Cariou A, Maxime V, et al. Corticosteroid treatment and intensive insulin therapy for septic shock in adults: a randomized controlled trial. JAMA. 2010;303(4):341-348.
Mupirocin-Chlorhexidine Decolonization of Nasal S. Aureus Carriers Reduces Surgical-Site Infections
Clinical question: Can rapid screening and decolonization of nasal carriers of Staphylococcus aureus on admission reduce surgical-site S. aureus infections?
Background: More than 80% of healthcare-associated (HCA) S. aureus infections are endogenous in origin. High-level nasal carriers have a three- to sixfold increased risk of infection with this organism compared with noncarriers and low-level carriers. Decolonization of nasal and extranasal S. aureus on admission might reduce this risk of infection.
Study design: Randomized, double-blinded, placebo-controlled, multicenter trial.
Setting: Three university hospitals and two general hospitals in the Netherlands.
Synopsis: In this study, 918 mostly surgical patients with nasal S. aureus identified preoperatively by PCR tests were randomized to decolonization versus placebo. The five-day decolonization protocol involved mupirocin nasal ointment and chlorhexidine soap baths. Decolonization reduced length of stay by nearly two days. Through six weeks postdischarge, the cumulative incidence of S. aureus infection was 3.4% after decolonization versus 7.7% with placebo (RR 0.42; 95% CI, 0.23-0.75). Among the sites of infection, deep surgical sites had the greatest risk reduction (RR 0.21; 95% CI, 0.07-0.62).
The results of this study are encouraging, but a few limitations should be noted. The decolonization protocol lasted five days, which might make implementation less practical. Also, the relative contributions of mupirocin and chlorhexidine are unclear.
S. aureus is important, but it represents a minority of surgical-site infections; the effect of the protocol on other organisms is unknown. Lastly, MRSA is not prevalent in the Netherlands and no carriers were identified in the study. Although the protocol was designed to eradicate MRSA, such carriers might have different carriage patterns requiring throat swabs in addition to nasal swabs.
Bottom line: Preoperative detection of S. aureus nasal carriage and nasal and extranasal decolonization significantly reduced endogenous S. aureus infection and length of stay. Decolonization might be most beneficial for carriers at increased risk of deep infection, such as those undergoing cardiac surgery.
Citation: Bode LG, Kluytmans JA, Wertheim HF, et al. Preventing surgical-site infections in nasal carriers of Staphylococcus aureus. N Engl J Med. 2010;362(1):9-17.
ABCD2 Is a Poor Predictor of Early Ischemic Stroke after Transient Ischemic Attack
Clinical question: How well does the ABCD2 score predict stroke risk within seven days of transient ischemic attack (TIA)?
Background: After TIA diagnosis, the seven-day risk of ischemic stroke is approximately 5%. Identifying these high-risk patients might facilitate ED decision-making. The ABCD2 score, a prediction tool for stroke after TIA, might be useful but has not been prospectively validated in a large, broad-patient population.
Study design: Prospective validation study, convenience sample.
Setting: Sixteen hospitals in North Carolina.
Synopsis: The ABCD2 score (range 0-7 points) predicts stroke risk after TIA. The investigators evaluated the accuracy of ABCD2 in predicting seven-day ischemic stroke risk in a convenience sample of 1,667 TIA patients. Strokes were categorized as disabling or nondisabling.
Overall, the score was poorly predictive of all ischemic stroke (c stat 0.59) and moderately predictive of the subset of disabling ischemic stroke (c stat 0.71). The ABCD2 had the most discriminatory power when used to identify patients at low risk of disabling stroke (0-3 points); for these patients, the negative likelihood ratio (LR) was 0.16 (0.04-0.64).
The study is the largest published external-validation study of the ABCD2 score, but it had significant limitations that should be considered. There was potential sampling bias because of nonconsecutive sampling, and unaccounted patients with TIA were discharged from the ED. Furthermore, ABCD2 scores were incalculable for 35% of patients, although the authors report that imputed data did not change the findings significantly.
Bottom line: The ABCD2 score does not sufficiently predict the seven-day risk of ischemic stroke after TIA. Further validation studies are needed.
Citation: Asimos AW, Johnson AM, Rosamond WD, et al. A multicenter evaluation of the ABCD2 score’s accuracy for predicting early ischemic stroke in admitted patients with transient ischemic attack. Ann Emerg Med. 2010;55(2):201-210.e5. TH
Dr. Kim is a hospitalist at Brigham and Women’s Hospital in Boston, and an instructor at Harvard Medical School.
In This Edition:
- Catheter ablation for atrial fibrillation
- Noncontrast CT for appendicitis diagnosis
- Effectiveness of whole-leg ultrasound for DVT diagnosis
- Physician localization and nurse-physician communication
- PPI versus H2 blocker in patients with aspirin-related peptic ulcer disease
- Intensive insulin therapy in steroid-induced hyperglycemia associated with septic shock
- S. aureus decolonization and surgical-site infections
- ABCD2 score and risk of subsequent stroke
Catheter Ablation of Paroxysmal AF Might Be More Effective than Antiarrhythmic Therapy
Clinical question: In patients with paroxysmal atrial fibrillation (AF) unresponsive to initial antiarrhythmic therapy, what is the efficacy of catheter ablation compared with continued attempts at rhythm control?
Background: Although antiarrhythmic drug therapy (ADT) is generally first-line AF therapy, AF recurrence is high and treatment is associated with adverse effects. Catheter ablation is an alternative treatment. Recent studies comparing antiarrhythmic drugs and catheter ablation have involved small populations and have shown mixed results.
Study design: Prospective, multicenter, unblinded, randomized trial.
Setting: Nineteen hospitals—15 in the U.S.—with considerable experience in AF ablation.
Synopsis: This trial compared catheter ablation (n=106) vs. ADT (n=61) for symptomatic, paroxysmal AF refractory to at least one antiarrhythmic drug. Patients in the ablation arm were allowed up to three procedures within an 80-day period; patients in the ADT arm were treated with a previously unused class I or class III antiarrhythmic. After nine months, 34% of ablated patients had failed treatment compared with 84% of patients receiving ADT (HR 0.30 [95% CI, 0.19-0.47]).
While the results are encouraging, some limitations should be noted. Attempts to generalize the results of this study might be limited, as the hospitals had considerable experience in AF ablation, the patient population was relatively young (mean age=56 years), and patients with significant left ventrical dysfunction and persistent AF were excluded.
Furthermore, the long-term effectiveness of ablation was not evaluated, and the study did not assess such outcomes as mortality, stroke, or AF progression. The effectiveness of specific ablation techniques could not be determined because a variety of approaches was employed.
Bottom line: Patients with symptomatic paroxysmal AF without advanced heart failure might benefit from catheter ablation at experienced institutions.
Citation: Wilber DJ, Pappone C, Neuzil P, et al. Comparison of antiarrhythmic drug therapy and radiofrequency catheter ablation in patients with paroxysmal atrial fibrillation: a randomized controlled trial. JAMA. 2010;303(4):333-340.
Noncontrast CT Can Be Used to Evaluate Suspected Appendicitis
Clinical question: What is the diagnostic accuracy of noncontrast helical CT in the evaluation of suspected acute appendicitis in the ED?
Background: In the workup of acute appendicitis, various CT protocols are used, including combinations of oral, intravenous, and rectal contrast, as well as noncontrast protocols. Noncontrast CT is less time-consuming and avoids risk of allergic reaction or contrast-induced nephropathy. The diagnostic accuracy of noncontrast CT, however, is controversial.
Study design: Systematic review/meta-analysis.
Setting: Seven studies evaluating acute appendicitis with noncontrast CT in ED settings.
Synopsis: This is the first systematic review of noncontrast CT (helical/multislice) in adults with suspected appendicitis. The authors pooled seven studies (1,060 patients) comparing noncontrast CT with a reference standard of final diagnosis at surgery or followup at a minimum of two weeks. The review yielded the following pooled estimates: sensitivity 93%, specificity 96%, positive likelihood ratio 24, and negative likelihood ratio 0.08. Overall, the diagnostic accuracy of noncontrast CT was high.
A few limitations should be noted. In the original studies, results were reported inconsistently. In one study, 24% of scans were inconclusive, with an associated likelihood ratio of approximately 1. Future studies must address this important clinical question of how to treat patients with inconclusive scans. Another question is to what degree the accuracy reported in this study reflects the expertise of the institutions, the majority of which were university-affiliated. According to one study author, “a certain level of experience is required for skillful interpretation” of noncontrast CT.
Bottom line: Noncontrast CT has a high sensitivity and specificity for acute appendicitis, and should be considered an alternative to contrast CT, particularly in patients with contraindications to contrast or those at risk of contrast-induced nephropathy.
Citation: Hlibczuk V, Dattaro JA, Jin Z, Falzon L, Brown MD. Diagnostic accuracy of noncontrast computed tomography for appendicitis in adults: a systematic review. Ann Emerg Med. 2010;55(1):51-59.e1.
Whole-Leg Compression Ultrasound for DVT Has a Low Failure Rate in Outpatients
Clinical question: How useful is compression ultrasound (CUS) for diagnosing distal DVT?
Background: CUS can reliably evaluate proximal DVT, but its accuracy for distal DVT is controversial. Because of the risk of extension of distal DVT (up to 25%), guidelines recommend that some patients undergo serial proximal CUS after an initial negative result. As an alternative, recent studies have evaluated one-time, whole-leg CUS.
Study design: Systematic review and meta-analysis.
Setting: Review of randomized controlled trials and prospective cohort studies.
Synopsis: The study pooled data from seven studies and more than 4,700 patients with suspected DVT for whom anticoagulation was withheld after a single, negative, whole-leg CUS. At the three-month followup, the combined symptomatic VTE event rate was 0.57%, and the authors concluded that withholding anticoagulation was associated with a low VTE risk.
Although encouraging, this study had several limitations. First, whole-leg CUS is not widely performed or standardized, and the CUS technique varied slightly across the studies. Second, any attempt to generalize the results of this study might be limited, as the proportion of pregnant patients and those with malignancy was low. Furthermore, only one of the seven studies included inpatients that might be at higher VTE risk. Third, pre-test probability was assessed for only a subset of patients, limiting assessment of VTE by risk level. For example, the overall finding of the study—an event rate of 0.57%—appears low but is difficult to apply clinically when subset analyses for high-risk patients yielded a VTE rate of 2.5% with wide confidence intervals ranging from 0% to 7%.
Bottom line: Whole-leg CUS might be a practical alternative to serial proximal CUS, but more data incorporating pre-test probabilities and involving more inpatients are needed. Hospitalists should be cautious in applying pooled summary estimates.
Citation: Johnson SA, Stevens SM, Woller SC, et al. Risk of deep vein thrombosis following a single negative whole-leg compression ultrasound: a systematic review and meta-analysis. JAMA. 2010;303(5):438-445.
Localization of Inpatient Physicians on Hospital Units Increases Provider Communication Frequency
Clinical question: Does localization of inpatient physicians on hospital units improve communication with nursing staff?
Background: While nurses are typically localized on a hospital unit, inpatient physicians often care for patients on multiple units. This lack of regionalization makes it difficult for physicians and nurses to discuss care plans directly. No prior research has evaluated the effect of physician localization on nurse-physician communication.
Study design: Cross-sectional, pre- and postintervention study.
Setting: Tertiary-care teaching hospital, general medical service.
Synopsis: The study was a cross-sectional survey of nurses and physicians prelocalization (n=342 patients) and postlocalization (n=294 patients) of physicians on hospital units. Localization was associated with increased frequency of communication; however, it did not improve the consistency of nurse-physician agreement on the care plan. Nurse-physician agreement was improved on two aspects of the care plan—planned tests and anticipated length of stay—but not on primary diagnosis, planned procedures, medication changes, or consultations.
Limitations of the study were that it was conducted at a single teaching hospital, communication patterns might have changed during the year between pre- and postlocalization, and physicians were not completely localized to specific units (73% localization).
Despite the limitations, this study is the first to evaluate staff localization and communication on a general medical service. The findings suggest that localization is a first step toward interdisciplinary communication. It also shows that quality and content of communication require further assessment.
Future studies must assess the impact of communication on the quality of patient care.
Bottom line: Physician localization improved the frequency of nurse-physician dialogue but did not consistently facilitate a shared understanding of the care plan. Although not assessed in this study, the implication is that the quality of communication between providers needs improvement.
Citation: O’Leary KJ, Wayne DB, Landler MP, et al. J Gen Int Med. 2009;24(11):1223-1227.
Pantoprazole Better than Double-Dose Famotidine in Secondary Prevention of Aspirin-Related Injury
Clinical question: Is a twice-daily H2-receptor antagonist (H2RA) or a once-daily proton pump inhibitor (PPI) better in patients who must continue low-dose-aspirin therapy despite aspirin-related peptic ulcer disease?
Background: Some patients with aspirin-related peptic ulcer disease require continued aspirin therapy. It often is assumed that PPIs are superior to H2RAs in secondary prevention of low-dose aspirin-related injury, although no randomized trials have specifically addressed this question.
Study design: A prospective, double-blind, randomized controlled trial.
Setting: A university hospital in Hong Kong.
Synopsis: In this trial, 160 patients with aspirin-related peptic ulcers/erosions were randomized to 48 weeks of oral famotidine (40 mg twice daily) or pantoprazole (20 mg daily) after mucosal healing and eradication of H pylori. During this time, all patients continued to receive aspirin (80 mg daily).
The rates of recurrent dyspeptic or bleeding ulcers/erosions within 48 weeks were 20% with famotidine versus 0% with pantoprazole. The rates of gastrointestinal (GI) bleeding alone were 7.7% versus 0%, respectively.
Of note, none of the five patients with GI bleeding had significant dyspepsia, which is consistent with prior reports that NSAID-induced injury might be silent.
Overall, high-dose famotidine was inferior to pantoprazole in the prevention of recurrent aspirin-related injury. These findings support the ACCF/ACG/AHA 2008 recommendation of PPIs over double-dose H2RAs in this clinical setting. It is not known, however, whether the PPI strategy extends to average GI-risk patients taking aspirin.
Furthermore, although not demonstrated in this study, it is possible that the lower rates of dyspepsia with pantoprazole might facilitate adherence to prolonged aspirin therapy.
Bottom line: High-dose famotidine was inferior to pantoprazole in the prevention of recurrent low-dose-aspirin-related injury.
Citation: Ng FH, Wong SY, Lam KF, et al. Famotidine is inferior to pantoprazole in preventing recurrence of aspirin-related peptic ulcers or erosions. Gastroenterology. 2010;138
(1):82-88.
Intensive Insulin Therapy Is Not Beneficial in Corticosteroid-Induced Hyperglycemia Associated with Septic Shock
Clinical question: In septic shock treated with hydrocortisone, does intensive insulin therapy reduce in-hospital mortality compared with conventional glucose management?
Background: Corticosteroids might benefit patients with septic shock, but they also can exacerbate illness-induced hyperglycemia. It is hypothesized that normalization of blood glucose with intensive insulin might improve outcomes in these patients.
Study design: A multicenter, 2x2 factorial, randomized controlled trial.
Setting: Eleven ICUs in France.
Synopsis: In this 2x2 factorial comparison, 509 patients with septic shock treated with hydrocortisone were randomized to IV insulin, conventional insulin, fludrocortisone plus IV insulin, and fludrocortisone plus conventional insulin. The primary objective was to assess intensive IV versus conventional insulin, and a secondary objective was to assess the benefit of adding fludrocortisone to hydrocortisone therapy.
Overall, analysis showed no difference in in-hospital mortality in either of the two comparisons.
These null findings should be interpreted considering the following study limitations. In the intensive-insulin groups, there was a failure to reach target glucose levels of 80 mg/dL to 110 mg/dL (mean achieved levels 120 mg/dL to 130 mg/dL and higher). These levels overlapped to some degree with the control group, which targeted glucose levels <150 mg/dL and achieved mean levels of 140 mg/dL to 150 mg/dL.
The lack of substantial difference in glucose levels might have contributed to the null findings. Furthermore, the absolute risk reduction in the original sample-size calculations was overestimated. The result was an underpowered study, which also might have contributed to the null findings.
Bottom line: In septic shock treated with hydrocortisone, the optimal blood-glucose level and insulin strategy are unknown.
Citation: Annane D, Cariou A, Maxime V, et al. Corticosteroid treatment and intensive insulin therapy for septic shock in adults: a randomized controlled trial. JAMA. 2010;303(4):341-348.
Mupirocin-Chlorhexidine Decolonization of Nasal S. Aureus Carriers Reduces Surgical-Site Infections
Clinical question: Can rapid screening and decolonization of nasal carriers of Staphylococcus aureus on admission reduce surgical-site S. aureus infections?
Background: More than 80% of healthcare-associated (HCA) S. aureus infections are endogenous in origin. High-level nasal carriers have a three- to sixfold increased risk of infection with this organism compared with noncarriers and low-level carriers. Decolonization of nasal and extranasal S. aureus on admission might reduce this risk of infection.
Study design: Randomized, double-blinded, placebo-controlled, multicenter trial.
Setting: Three university hospitals and two general hospitals in the Netherlands.
Synopsis: In this study, 918 mostly surgical patients with nasal S. aureus identified preoperatively by PCR tests were randomized to decolonization versus placebo. The five-day decolonization protocol involved mupirocin nasal ointment and chlorhexidine soap baths. Decolonization reduced length of stay by nearly two days. Through six weeks postdischarge, the cumulative incidence of S. aureus infection was 3.4% after decolonization versus 7.7% with placebo (RR 0.42; 95% CI, 0.23-0.75). Among the sites of infection, deep surgical sites had the greatest risk reduction (RR 0.21; 95% CI, 0.07-0.62).
The results of this study are encouraging, but a few limitations should be noted. The decolonization protocol lasted five days, which might make implementation less practical. Also, the relative contributions of mupirocin and chlorhexidine are unclear.
S. aureus is important, but it represents a minority of surgical-site infections; the effect of the protocol on other organisms is unknown. Lastly, MRSA is not prevalent in the Netherlands and no carriers were identified in the study. Although the protocol was designed to eradicate MRSA, such carriers might have different carriage patterns requiring throat swabs in addition to nasal swabs.
Bottom line: Preoperative detection of S. aureus nasal carriage and nasal and extranasal decolonization significantly reduced endogenous S. aureus infection and length of stay. Decolonization might be most beneficial for carriers at increased risk of deep infection, such as those undergoing cardiac surgery.
Citation: Bode LG, Kluytmans JA, Wertheim HF, et al. Preventing surgical-site infections in nasal carriers of Staphylococcus aureus. N Engl J Med. 2010;362(1):9-17.
ABCD2 Is a Poor Predictor of Early Ischemic Stroke after Transient Ischemic Attack
Clinical question: How well does the ABCD2 score predict stroke risk within seven days of transient ischemic attack (TIA)?
Background: After TIA diagnosis, the seven-day risk of ischemic stroke is approximately 5%. Identifying these high-risk patients might facilitate ED decision-making. The ABCD2 score, a prediction tool for stroke after TIA, might be useful but has not been prospectively validated in a large, broad-patient population.
Study design: Prospective validation study, convenience sample.
Setting: Sixteen hospitals in North Carolina.
Synopsis: The ABCD2 score (range 0-7 points) predicts stroke risk after TIA. The investigators evaluated the accuracy of ABCD2 in predicting seven-day ischemic stroke risk in a convenience sample of 1,667 TIA patients. Strokes were categorized as disabling or nondisabling.
Overall, the score was poorly predictive of all ischemic stroke (c stat 0.59) and moderately predictive of the subset of disabling ischemic stroke (c stat 0.71). The ABCD2 had the most discriminatory power when used to identify patients at low risk of disabling stroke (0-3 points); for these patients, the negative likelihood ratio (LR) was 0.16 (0.04-0.64).
The study is the largest published external-validation study of the ABCD2 score, but it had significant limitations that should be considered. There was potential sampling bias because of nonconsecutive sampling, and unaccounted patients with TIA were discharged from the ED. Furthermore, ABCD2 scores were incalculable for 35% of patients, although the authors report that imputed data did not change the findings significantly.
Bottom line: The ABCD2 score does not sufficiently predict the seven-day risk of ischemic stroke after TIA. Further validation studies are needed.
Citation: Asimos AW, Johnson AM, Rosamond WD, et al. A multicenter evaluation of the ABCD2 score’s accuracy for predicting early ischemic stroke in admitted patients with transient ischemic attack. Ann Emerg Med. 2010;55(2):201-210.e5. TH
Dr. Kim is a hospitalist at Brigham and Women’s Hospital in Boston, and an instructor at Harvard Medical School.