User login
Altha J. Stewart, MD, on the state of psychiatry
For this Psychiatry Leaders’ Perspectives, Awais Aftab, MD, interviewed Altha J. Stewart, MD. Dr. Stewart is Senior Associate Dean for Community Health Engagement at the University of Tennessee Health Science Center (UTHSC)–Memphis. She also serves as Chief of the Division of Social and Community Psychiatry and Director, Center for Health in Justice Involved Youth at UTHSC, where she manages community-based programs serving children impacted by trauma and mental illness and their families. In 2018, she was elected President of the American Psychiatric Association, the first African American individual elected in the 175-year history of the organization.
Dr. Aftab: Structural racism in academic and organized psychiatry is an issue that is close to your heart. What is your perspective on the current state of structural racism in American psychiatry, and what do you think we can do about it?
Dr. Stewart: That’s a good question to start with because I think the conversations that we need to have in academia in general and in academic psychiatry specifically really do frame the current issues that we are facing, whether we’re talking about eliminating health disparities or achieving mental health equity. Historically, from the very beginning these discussions have been structured in a racist manner. The early days of American psychiatry were very clearly directed towards maintaining a system that excluded large segments of the population of the time, since a particularly violent form of chattel slavery was being practiced in this country.
The mental health care system was primarily designed for the landowning white men of some standing in society, and so there was never any intent to do much in the way of providing quality humane service to people who were not part of that group. What we have today is a system that was designed for a racist societal structure, that was intended to perpetuate certain behaviors, policies, and practices that had at their core a racist framework. We have to acknowledge and start from this beginning point. This is not to blame anyone currently alive. These are larger structural problems. Before we can begin setting up strategic plans and other actions, we have to go back and acknowledge how we got here. We have to accept the responsibility for being here, and then we have to allow the conversations that need to happen to happen in a safe way, without further alienating people, or maligning and demeaning people who are for the most part well-intentioned but perhaps operating on automatic pilot in a system that is structurally racist.
Dr. Aftab: Do you think that the conversations that need to happen are taking place?
Dr. Stewart: Yes, I think they are beginning to happen. I do a fair number of talks and grand rounds, and what I discover when I meet with different academic departments and different groups is that most places now have a diversity committee, or the residents and students have assigned themselves as diversity leaders. They are really pushing to have these conversations, to insert these conversations into the training and education curricula. The structures in power are so deeply entrenched that many people, particularly younger people, are easily frustrated by the lack of forward motion. One of the things that seasoned leaders in psychiatry have to do is to help everyone understand that the movement forward might be glacial in the beginning, but any movement forward is good when it comes to this. The psychiatrists of my generation talked about cultural competence in psychiatry, but generations of today talk about structural competence. These are similar concepts, except that cultural competency worked within the traditional model, while structural competency recognizes that the system itself needs to change. I find this development very encouraging.
Dr. Aftab: What do you see as some of the strengths of our profession?
Continue to: Dr. Stewart
Dr. Stewart: I am a hopeful optimist when it comes to psychiatry. I have dedicated my professional life to psychiatry and specifically to community psychiatry. Throughout the time that I have practiced psychiatry, I have been encouraged that what we do as a medical specialty really does improve the quality of life for the people we serve. Situationally right now, we’re in a unique position because the COVID pandemic has laid open and then laid bare the whole issue of how we deal with psychological distress, whether it’s diagnosed mental illness or a natural, normal response to a catastrophic event. We are the experts in this. This is our sweet spot, our wheelhouse, whatever analogy you prefer. This is the moment where we assert our expertise as the leaders—not as service add-ons, not as followers, not as adjuncts, but as the leaders.
I am so impressed with the next generation of psychiatrists. They have a wonderful blend of pride and privilege at what they have been able to accomplish to get to the point where they are doctors and psychiatrists, but they have aligned that with a strong core sense of social justice, and they are moved by their responsibility to the people in the society around them.
Another strength of our profession is what we consider to be the “art” of psychiatry. That is, the way we marry the relational aspects of psychiatry with the biological, technical, and digital aspects to arrive at a happy collaboration that benefits people. It is our great skill to engage people, to interact with them therapeutically, to recognize and acknowledge the nonverbal cues. This skill will be even more important in the age of online mental health services. I’m an “old-school” therapist. I like that face-to-face interaction. I think it’s important to preserve that aspect of our practice, even as we move towards online services.
Dr. Aftab: Are there ways in which the status quo in psychiatry falls short of the ideal? What are our areas of relative weakness?
Dr. Stewart: I don’t think we can afford to remain in status quo, because we need to constantly think and rethink, evaluate and re-evaluate, assess things in the light of new information. Particularly if we’re talking about people who rely on public funding to get even the bare minimum services, status quo doesn’t cut it. It’s not good enough. I had a teacher during my residency, a child psychiatrist, who used to say, “Good, better, best. Never let it rest, until your good is better and your better is best.” Something about that has stuck with me. As my career progressed, I heard variations of it, including one from former Surgeon General of the United States David Satcher, who was not a psychiatrist, but pulled together the group that published the first Surgeon General’s report on mental health, followed by the Surgeon General’s report on mental health, culture, race, and ethnicity. He had the penetrating insight that risk factors are not to be accepted as predictive factors due to protective factors. If I am at risk for mental illness or a chronic medical condition based on my race or ethnicity or socioeconomic status or employment status, this does not mean that I am destined to experience that illness. In fact, we are not doing our job if we accept these outcomes as inevitable and make no attempt to change them. So, for me, if we accept the status quo, we give up on the message of “Good, better, best. Never let it rest, until your good is better and your better is best.”
Continue to: Dr. Aftab
Dr. Aftab: What is your perception of the threats that psychiatry faces or is likely to face in the future?
Dr. Stewart: Well, this is going to sound harsh, and I do hope that the readers do not feel that I intend it to be harsh. We get in our own way. I work in the public sector, for example, and the reality is that there aren’t enough psychiatrists to provide all the necessary psychiatric services for the people who need them. So many mental health clinics and practices employ other mental health professionals, whether they are psychologists or nurse practitioners or physician assistants with special training in mental health to provide those services. To have a blanket concern about anyone who is not an MD practicing in what is considered “our area” just begs the question that if we can’t do it and we don’t have enough psychiatrists to do it, should people just not get mental health treatment? Is that the solution? I don’t think so. I don’t think that’s what people want, either, but because of the energy that gets aroused around these issues, we lose sight of that end goal. I think the answer is that we must take leadership for ensuring that our colleagues are well-trained, maybe not as well-trained as physicians, but well-trained enough to provide good care working under our supervision.
Dr. Aftab: What do you envision for the future of psychiatry? What sort of opportunities lie ahead for us?
Dr. Stewart: I think we are moving naturally into the space of integrated or collaborative care. I think we’re going to have to acknowledge that going forward, the path to being a good psychiatrist means that we will also be consultants. Not just the consultation-liaison kind of consultant that we typically think of, but a consultant to the rest of medicine around shaping programs, addressing how we treat comorbid illness, looking at ways to minimize the morbidity and mortality associated with some of the chronic medical and mental diseases. We’re moving naturally in that direction. For some people, that must be frightening. All throughout medicine people are witnessing change, and we need to adapt. I would hope that the specialty that is designed to help others deal with change will figure out how to use those skills to help themselves deal with the changes that are coming!
For this Psychiatry Leaders’ Perspectives, Awais Aftab, MD, interviewed Altha J. Stewart, MD. Dr. Stewart is Senior Associate Dean for Community Health Engagement at the University of Tennessee Health Science Center (UTHSC)–Memphis. She also serves as Chief of the Division of Social and Community Psychiatry and Director, Center for Health in Justice Involved Youth at UTHSC, where she manages community-based programs serving children impacted by trauma and mental illness and their families. In 2018, she was elected President of the American Psychiatric Association, the first African American individual elected in the 175-year history of the organization.
Dr. Aftab: Structural racism in academic and organized psychiatry is an issue that is close to your heart. What is your perspective on the current state of structural racism in American psychiatry, and what do you think we can do about it?
Dr. Stewart: That’s a good question to start with because I think the conversations that we need to have in academia in general and in academic psychiatry specifically really do frame the current issues that we are facing, whether we’re talking about eliminating health disparities or achieving mental health equity. Historically, from the very beginning these discussions have been structured in a racist manner. The early days of American psychiatry were very clearly directed towards maintaining a system that excluded large segments of the population of the time, since a particularly violent form of chattel slavery was being practiced in this country.
The mental health care system was primarily designed for the landowning white men of some standing in society, and so there was never any intent to do much in the way of providing quality humane service to people who were not part of that group. What we have today is a system that was designed for a racist societal structure, that was intended to perpetuate certain behaviors, policies, and practices that had at their core a racist framework. We have to acknowledge and start from this beginning point. This is not to blame anyone currently alive. These are larger structural problems. Before we can begin setting up strategic plans and other actions, we have to go back and acknowledge how we got here. We have to accept the responsibility for being here, and then we have to allow the conversations that need to happen to happen in a safe way, without further alienating people, or maligning and demeaning people who are for the most part well-intentioned but perhaps operating on automatic pilot in a system that is structurally racist.
Dr. Aftab: Do you think that the conversations that need to happen are taking place?
Dr. Stewart: Yes, I think they are beginning to happen. I do a fair number of talks and grand rounds, and what I discover when I meet with different academic departments and different groups is that most places now have a diversity committee, or the residents and students have assigned themselves as diversity leaders. They are really pushing to have these conversations, to insert these conversations into the training and education curricula. The structures in power are so deeply entrenched that many people, particularly younger people, are easily frustrated by the lack of forward motion. One of the things that seasoned leaders in psychiatry have to do is to help everyone understand that the movement forward might be glacial in the beginning, but any movement forward is good when it comes to this. The psychiatrists of my generation talked about cultural competence in psychiatry, but generations of today talk about structural competence. These are similar concepts, except that cultural competency worked within the traditional model, while structural competency recognizes that the system itself needs to change. I find this development very encouraging.
Dr. Aftab: What do you see as some of the strengths of our profession?
Continue to: Dr. Stewart
Dr. Stewart: I am a hopeful optimist when it comes to psychiatry. I have dedicated my professional life to psychiatry and specifically to community psychiatry. Throughout the time that I have practiced psychiatry, I have been encouraged that what we do as a medical specialty really does improve the quality of life for the people we serve. Situationally right now, we’re in a unique position because the COVID pandemic has laid open and then laid bare the whole issue of how we deal with psychological distress, whether it’s diagnosed mental illness or a natural, normal response to a catastrophic event. We are the experts in this. This is our sweet spot, our wheelhouse, whatever analogy you prefer. This is the moment where we assert our expertise as the leaders—not as service add-ons, not as followers, not as adjuncts, but as the leaders.
I am so impressed with the next generation of psychiatrists. They have a wonderful blend of pride and privilege at what they have been able to accomplish to get to the point where they are doctors and psychiatrists, but they have aligned that with a strong core sense of social justice, and they are moved by their responsibility to the people in the society around them.
Another strength of our profession is what we consider to be the “art” of psychiatry. That is, the way we marry the relational aspects of psychiatry with the biological, technical, and digital aspects to arrive at a happy collaboration that benefits people. It is our great skill to engage people, to interact with them therapeutically, to recognize and acknowledge the nonverbal cues. This skill will be even more important in the age of online mental health services. I’m an “old-school” therapist. I like that face-to-face interaction. I think it’s important to preserve that aspect of our practice, even as we move towards online services.
Dr. Aftab: Are there ways in which the status quo in psychiatry falls short of the ideal? What are our areas of relative weakness?
Dr. Stewart: I don’t think we can afford to remain in status quo, because we need to constantly think and rethink, evaluate and re-evaluate, assess things in the light of new information. Particularly if we’re talking about people who rely on public funding to get even the bare minimum services, status quo doesn’t cut it. It’s not good enough. I had a teacher during my residency, a child psychiatrist, who used to say, “Good, better, best. Never let it rest, until your good is better and your better is best.” Something about that has stuck with me. As my career progressed, I heard variations of it, including one from former Surgeon General of the United States David Satcher, who was not a psychiatrist, but pulled together the group that published the first Surgeon General’s report on mental health, followed by the Surgeon General’s report on mental health, culture, race, and ethnicity. He had the penetrating insight that risk factors are not to be accepted as predictive factors due to protective factors. If I am at risk for mental illness or a chronic medical condition based on my race or ethnicity or socioeconomic status or employment status, this does not mean that I am destined to experience that illness. In fact, we are not doing our job if we accept these outcomes as inevitable and make no attempt to change them. So, for me, if we accept the status quo, we give up on the message of “Good, better, best. Never let it rest, until your good is better and your better is best.”
Continue to: Dr. Aftab
Dr. Aftab: What is your perception of the threats that psychiatry faces or is likely to face in the future?
Dr. Stewart: Well, this is going to sound harsh, and I do hope that the readers do not feel that I intend it to be harsh. We get in our own way. I work in the public sector, for example, and the reality is that there aren’t enough psychiatrists to provide all the necessary psychiatric services for the people who need them. So many mental health clinics and practices employ other mental health professionals, whether they are psychologists or nurse practitioners or physician assistants with special training in mental health to provide those services. To have a blanket concern about anyone who is not an MD practicing in what is considered “our area” just begs the question that if we can’t do it and we don’t have enough psychiatrists to do it, should people just not get mental health treatment? Is that the solution? I don’t think so. I don’t think that’s what people want, either, but because of the energy that gets aroused around these issues, we lose sight of that end goal. I think the answer is that we must take leadership for ensuring that our colleagues are well-trained, maybe not as well-trained as physicians, but well-trained enough to provide good care working under our supervision.
Dr. Aftab: What do you envision for the future of psychiatry? What sort of opportunities lie ahead for us?
Dr. Stewart: I think we are moving naturally into the space of integrated or collaborative care. I think we’re going to have to acknowledge that going forward, the path to being a good psychiatrist means that we will also be consultants. Not just the consultation-liaison kind of consultant that we typically think of, but a consultant to the rest of medicine around shaping programs, addressing how we treat comorbid illness, looking at ways to minimize the morbidity and mortality associated with some of the chronic medical and mental diseases. We’re moving naturally in that direction. For some people, that must be frightening. All throughout medicine people are witnessing change, and we need to adapt. I would hope that the specialty that is designed to help others deal with change will figure out how to use those skills to help themselves deal with the changes that are coming!
For this Psychiatry Leaders’ Perspectives, Awais Aftab, MD, interviewed Altha J. Stewart, MD. Dr. Stewart is Senior Associate Dean for Community Health Engagement at the University of Tennessee Health Science Center (UTHSC)–Memphis. She also serves as Chief of the Division of Social and Community Psychiatry and Director, Center for Health in Justice Involved Youth at UTHSC, where she manages community-based programs serving children impacted by trauma and mental illness and their families. In 2018, she was elected President of the American Psychiatric Association, the first African American individual elected in the 175-year history of the organization.
Dr. Aftab: Structural racism in academic and organized psychiatry is an issue that is close to your heart. What is your perspective on the current state of structural racism in American psychiatry, and what do you think we can do about it?
Dr. Stewart: That’s a good question to start with because I think the conversations that we need to have in academia in general and in academic psychiatry specifically really do frame the current issues that we are facing, whether we’re talking about eliminating health disparities or achieving mental health equity. Historically, from the very beginning these discussions have been structured in a racist manner. The early days of American psychiatry were very clearly directed towards maintaining a system that excluded large segments of the population of the time, since a particularly violent form of chattel slavery was being practiced in this country.
The mental health care system was primarily designed for the landowning white men of some standing in society, and so there was never any intent to do much in the way of providing quality humane service to people who were not part of that group. What we have today is a system that was designed for a racist societal structure, that was intended to perpetuate certain behaviors, policies, and practices that had at their core a racist framework. We have to acknowledge and start from this beginning point. This is not to blame anyone currently alive. These are larger structural problems. Before we can begin setting up strategic plans and other actions, we have to go back and acknowledge how we got here. We have to accept the responsibility for being here, and then we have to allow the conversations that need to happen to happen in a safe way, without further alienating people, or maligning and demeaning people who are for the most part well-intentioned but perhaps operating on automatic pilot in a system that is structurally racist.
Dr. Aftab: Do you think that the conversations that need to happen are taking place?
Dr. Stewart: Yes, I think they are beginning to happen. I do a fair number of talks and grand rounds, and what I discover when I meet with different academic departments and different groups is that most places now have a diversity committee, or the residents and students have assigned themselves as diversity leaders. They are really pushing to have these conversations, to insert these conversations into the training and education curricula. The structures in power are so deeply entrenched that many people, particularly younger people, are easily frustrated by the lack of forward motion. One of the things that seasoned leaders in psychiatry have to do is to help everyone understand that the movement forward might be glacial in the beginning, but any movement forward is good when it comes to this. The psychiatrists of my generation talked about cultural competence in psychiatry, but generations of today talk about structural competence. These are similar concepts, except that cultural competency worked within the traditional model, while structural competency recognizes that the system itself needs to change. I find this development very encouraging.
Dr. Aftab: What do you see as some of the strengths of our profession?
Continue to: Dr. Stewart
Dr. Stewart: I am a hopeful optimist when it comes to psychiatry. I have dedicated my professional life to psychiatry and specifically to community psychiatry. Throughout the time that I have practiced psychiatry, I have been encouraged that what we do as a medical specialty really does improve the quality of life for the people we serve. Situationally right now, we’re in a unique position because the COVID pandemic has laid open and then laid bare the whole issue of how we deal with psychological distress, whether it’s diagnosed mental illness or a natural, normal response to a catastrophic event. We are the experts in this. This is our sweet spot, our wheelhouse, whatever analogy you prefer. This is the moment where we assert our expertise as the leaders—not as service add-ons, not as followers, not as adjuncts, but as the leaders.
I am so impressed with the next generation of psychiatrists. They have a wonderful blend of pride and privilege at what they have been able to accomplish to get to the point where they are doctors and psychiatrists, but they have aligned that with a strong core sense of social justice, and they are moved by their responsibility to the people in the society around them.
Another strength of our profession is what we consider to be the “art” of psychiatry. That is, the way we marry the relational aspects of psychiatry with the biological, technical, and digital aspects to arrive at a happy collaboration that benefits people. It is our great skill to engage people, to interact with them therapeutically, to recognize and acknowledge the nonverbal cues. This skill will be even more important in the age of online mental health services. I’m an “old-school” therapist. I like that face-to-face interaction. I think it’s important to preserve that aspect of our practice, even as we move towards online services.
Dr. Aftab: Are there ways in which the status quo in psychiatry falls short of the ideal? What are our areas of relative weakness?
Dr. Stewart: I don’t think we can afford to remain in status quo, because we need to constantly think and rethink, evaluate and re-evaluate, assess things in the light of new information. Particularly if we’re talking about people who rely on public funding to get even the bare minimum services, status quo doesn’t cut it. It’s not good enough. I had a teacher during my residency, a child psychiatrist, who used to say, “Good, better, best. Never let it rest, until your good is better and your better is best.” Something about that has stuck with me. As my career progressed, I heard variations of it, including one from former Surgeon General of the United States David Satcher, who was not a psychiatrist, but pulled together the group that published the first Surgeon General’s report on mental health, followed by the Surgeon General’s report on mental health, culture, race, and ethnicity. He had the penetrating insight that risk factors are not to be accepted as predictive factors due to protective factors. If I am at risk for mental illness or a chronic medical condition based on my race or ethnicity or socioeconomic status or employment status, this does not mean that I am destined to experience that illness. In fact, we are not doing our job if we accept these outcomes as inevitable and make no attempt to change them. So, for me, if we accept the status quo, we give up on the message of “Good, better, best. Never let it rest, until your good is better and your better is best.”
Continue to: Dr. Aftab
Dr. Aftab: What is your perception of the threats that psychiatry faces or is likely to face in the future?
Dr. Stewart: Well, this is going to sound harsh, and I do hope that the readers do not feel that I intend it to be harsh. We get in our own way. I work in the public sector, for example, and the reality is that there aren’t enough psychiatrists to provide all the necessary psychiatric services for the people who need them. So many mental health clinics and practices employ other mental health professionals, whether they are psychologists or nurse practitioners or physician assistants with special training in mental health to provide those services. To have a blanket concern about anyone who is not an MD practicing in what is considered “our area” just begs the question that if we can’t do it and we don’t have enough psychiatrists to do it, should people just not get mental health treatment? Is that the solution? I don’t think so. I don’t think that’s what people want, either, but because of the energy that gets aroused around these issues, we lose sight of that end goal. I think the answer is that we must take leadership for ensuring that our colleagues are well-trained, maybe not as well-trained as physicians, but well-trained enough to provide good care working under our supervision.
Dr. Aftab: What do you envision for the future of psychiatry? What sort of opportunities lie ahead for us?
Dr. Stewart: I think we are moving naturally into the space of integrated or collaborative care. I think we’re going to have to acknowledge that going forward, the path to being a good psychiatrist means that we will also be consultants. Not just the consultation-liaison kind of consultant that we typically think of, but a consultant to the rest of medicine around shaping programs, addressing how we treat comorbid illness, looking at ways to minimize the morbidity and mortality associated with some of the chronic medical and mental diseases. We’re moving naturally in that direction. For some people, that must be frightening. All throughout medicine people are witnessing change, and we need to adapt. I would hope that the specialty that is designed to help others deal with change will figure out how to use those skills to help themselves deal with the changes that are coming!
What’s lost, what’s saved
DDW is now history. While rejoicing that DDW happened (as opposed to when it couldn’t in 2020), the virtual format precluded all those hallway conversations, meetings with mentors and small group (after hour) discussions. This year, AGA saved substantial monies in travel costs. Of note, at Michigan Medicine, we track the miles patients did not have to travel because of our conversion to virtual care (currently about 30% of all ambulatory visits). To date, our “virtual first” protocol has saved over 24 million patient travel-miles since February 2020 (average of 62 miles per patient visit).
The pandemic forced rapid adoption of virtual care and alternative care delivery models. As patients adapted to telehealth, businesses saw opportunities. Health systems have begun to downsize their brick-and-mortar footprints for both clinical and office space. Hospital at Home models are developing as viable alternatives to inpatient care using a hybrid system of on-site nurses and remote physician supervision.
Digital health start-ups are developing rapidly, and equity funding for digital health companies has reached an all-time high of $26.5 billion in 2020. Multiple companies went public through traditional initial public offerings or special purpose acquisition companies. Sameer Berry, MD, recently collected an inventory of major GI digital health companies counted at least 16 with more appearing each month. These companies focus on management of a single condition (for example IBS or Celiac) or full-service virtual GI care that includes “at-risk” financial contracts
I am delighted to announce that Megan Adams, MD, JD, MSc, has been chosen to be the fourth editor in chief of GI & Hepatology News. She and her team will transition into editorial control during Fall 2021. I have known Megan since meeting her at an AGA young faculty function almost 10 years ago. She is extremely talented and knowledgeable about gastroenterology from a variety of viewpoints. She has recruited a strong and dedicated editorial board.
I have enjoyed the last 5 years leading the current board as we have brought breaking news to the GI community. I wish to publicly thank our editorial board and the Frontline staff who monthly publish AGA’s official newspaper.
John I. Allen, MD, MBA, AGAF
Editor in Chief
DDW is now history. While rejoicing that DDW happened (as opposed to when it couldn’t in 2020), the virtual format precluded all those hallway conversations, meetings with mentors and small group (after hour) discussions. This year, AGA saved substantial monies in travel costs. Of note, at Michigan Medicine, we track the miles patients did not have to travel because of our conversion to virtual care (currently about 30% of all ambulatory visits). To date, our “virtual first” protocol has saved over 24 million patient travel-miles since February 2020 (average of 62 miles per patient visit).
The pandemic forced rapid adoption of virtual care and alternative care delivery models. As patients adapted to telehealth, businesses saw opportunities. Health systems have begun to downsize their brick-and-mortar footprints for both clinical and office space. Hospital at Home models are developing as viable alternatives to inpatient care using a hybrid system of on-site nurses and remote physician supervision.
Digital health start-ups are developing rapidly, and equity funding for digital health companies has reached an all-time high of $26.5 billion in 2020. Multiple companies went public through traditional initial public offerings or special purpose acquisition companies. Sameer Berry, MD, recently collected an inventory of major GI digital health companies counted at least 16 with more appearing each month. These companies focus on management of a single condition (for example IBS or Celiac) or full-service virtual GI care that includes “at-risk” financial contracts
I am delighted to announce that Megan Adams, MD, JD, MSc, has been chosen to be the fourth editor in chief of GI & Hepatology News. She and her team will transition into editorial control during Fall 2021. I have known Megan since meeting her at an AGA young faculty function almost 10 years ago. She is extremely talented and knowledgeable about gastroenterology from a variety of viewpoints. She has recruited a strong and dedicated editorial board.
I have enjoyed the last 5 years leading the current board as we have brought breaking news to the GI community. I wish to publicly thank our editorial board and the Frontline staff who monthly publish AGA’s official newspaper.
John I. Allen, MD, MBA, AGAF
Editor in Chief
DDW is now history. While rejoicing that DDW happened (as opposed to when it couldn’t in 2020), the virtual format precluded all those hallway conversations, meetings with mentors and small group (after hour) discussions. This year, AGA saved substantial monies in travel costs. Of note, at Michigan Medicine, we track the miles patients did not have to travel because of our conversion to virtual care (currently about 30% of all ambulatory visits). To date, our “virtual first” protocol has saved over 24 million patient travel-miles since February 2020 (average of 62 miles per patient visit).
The pandemic forced rapid adoption of virtual care and alternative care delivery models. As patients adapted to telehealth, businesses saw opportunities. Health systems have begun to downsize their brick-and-mortar footprints for both clinical and office space. Hospital at Home models are developing as viable alternatives to inpatient care using a hybrid system of on-site nurses and remote physician supervision.
Digital health start-ups are developing rapidly, and equity funding for digital health companies has reached an all-time high of $26.5 billion in 2020. Multiple companies went public through traditional initial public offerings or special purpose acquisition companies. Sameer Berry, MD, recently collected an inventory of major GI digital health companies counted at least 16 with more appearing each month. These companies focus on management of a single condition (for example IBS or Celiac) or full-service virtual GI care that includes “at-risk” financial contracts
I am delighted to announce that Megan Adams, MD, JD, MSc, has been chosen to be the fourth editor in chief of GI & Hepatology News. She and her team will transition into editorial control during Fall 2021. I have known Megan since meeting her at an AGA young faculty function almost 10 years ago. She is extremely talented and knowledgeable about gastroenterology from a variety of viewpoints. She has recruited a strong and dedicated editorial board.
I have enjoyed the last 5 years leading the current board as we have brought breaking news to the GI community. I wish to publicly thank our editorial board and the Frontline staff who monthly publish AGA’s official newspaper.
John I. Allen, MD, MBA, AGAF
Editor in Chief
Harassment of health care workers: A survey
During the course of my residency training, I have experienced and witnessed patients and visitors harassing health care workers (HCWs) by cursing or directing racial slurs at them, making sexist comments, or threatening their lives. What should be the correct response to this harassment? To say nothing may avoid conflict, but the silence perpetuates such abuse. To speak up may provoke aggression or even a physical assault. Further, does our response change if it is not the patient but someone who is accompanying them who exhibits this behavior?
I conducted a survey of psychiatry HCWs at our institution to evaluate the prevalence of and factors associated with such harassment.
An all-too-common problem
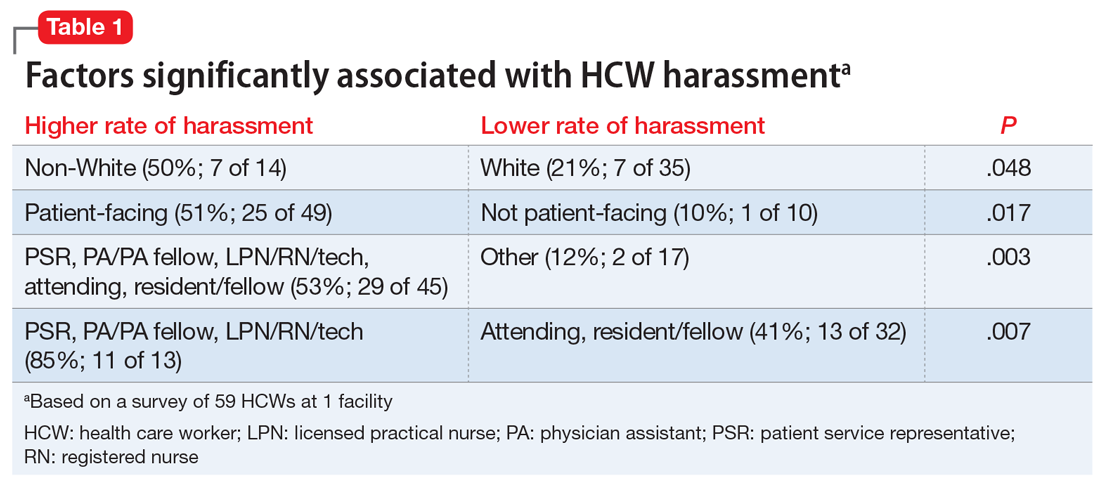
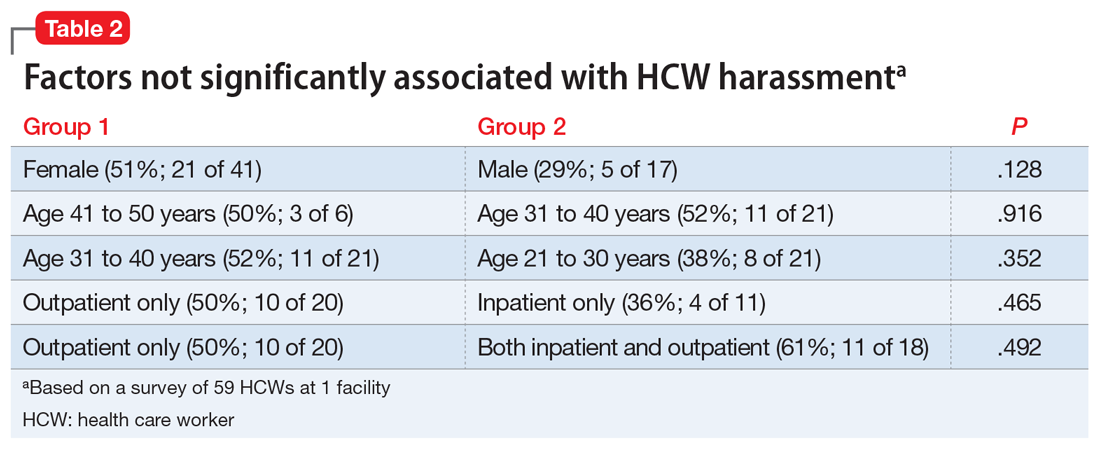
In a December 2020 internal survey at the University of Missouri Department of Psychiatry, 59 of 158 HCWs responded, and 26 (44%) reported experiencing or witnessing on-the-job harassment or abuse. Factors that were statistically significantly associated with experiencing or witnessing on-the-job harassment or abuse included being non-White, working in a patient-facing position, and being a nonphysician patient-facing HCW (Table 1). Factors that were not significantly associated with experiencing or witnessing on-the-job harassment or abuse included clinical setting, HCW age, and HCW gender (Table 2).
In addition to comments from patients and visitors, respondents stated that the harassment or abuse also included:
- physically threatening behavior and assault
- reporting a HCW for HIPAA (Health Insurance Portability and Accountability Act) violations after the HCW declined to provide an early refill of a controlled substance
- being accused of being a bad person for declining to prescribe a specific medication
- insults about not being intelligent enough to be on the treatment team
- comments from colleagues.
At the most basic level of response, the emergency department (ED) remains under the Emergency Medical Treatment and Labor Act (EMTALA) obligation to see, screen, and stabilize any patient, and if psychiatry is consulted in the ED, we should similarly provide this standard of care. Beyond this, we can create behavioral plans for when a relevant diagnosis exists or does not exist, and patients and/or visitors can be terminated from their stay at the location/service/health care system. Whether or not a patient is receiving psychiatric care and/or treatment is irrelevant to the responses to harassment we might consider.
During the incident itself, we are empowered to remove ourselves from the patient encounter. Historically, HCWs have had strong opinions on the next steps, either deciding, “Yes, I am a professional and I will not be bullied,” or “No, I am a professional and I don’t need to deal with this.” Just as we prioritize our patients’ dignities, we should also respect our own and our colleagues’ dignities.
How harassment is handled at our facility
HCWs are commonly unsure whether to “call out” abusive comments during the encounter itself or afterwards. In our hospital, HCWs are encouraged to independently choose to immediately respond, immediately report to a supervisor or hospital security, or defer and report to leadership afterwards via the Patient Safety Network (PSN). The PSN is our hospital’s reporting system for medical errors, near misses, and abuse, neglect, and workplace violence. Relevant examples of abuse, neglect, and workplace violence include:
- Threats. Expression of intent to cause harm, including verbal or written threats and threatening body language
- Physical assault. Attacks ranging from slapping and beating to rape, the use of weapons, or homicide
- Sexual assault. Any type of sexual contact or behavior that occurs without the explicit consent of the recipient, such as forced sexual intercourse, forcible sodomy, child molestation, incest, fondling, and attempted rape.
Continue to: Once complete...
Once complete, the PSN report is sent to Risk Management and other relevant groups, such as a 5-person team of security investigators, who are trained in trauma-informed interviewing and re-directive techniques. This team can immediately speak to the patient face-to-face in the inpatient setting or follow-up via phone in the outpatient setting.
The PSN report may result in the creation of a behavior plan for the patient that outlines the behaviors of concern, staff interventions, and consequences for persistent violations. The behavior plan is saved in the patient’s medical chart, and an alert pops up every time the chart is opened. The behavior plan is reviewed once annually for revision or deletion, as appropriate.
Lessons from our facility’s policy
In our health care system, our primary response to HCW harassment is to create a patient behavior plan that lays out specific expectations, care parameters, and consequences (up to terminating a patient from the entire health care system, except for EMTALA-level care). Clinicians are encouraged to report harassment to hospital administration, and a team of security investigators discusses expectations with the patient and/or visitors to prevent further abuse. We believe that describing our policies may be helpful to other health care systems and HCWs who confront this widespread issue.
During the course of my residency training, I have experienced and witnessed patients and visitors harassing health care workers (HCWs) by cursing or directing racial slurs at them, making sexist comments, or threatening their lives. What should be the correct response to this harassment? To say nothing may avoid conflict, but the silence perpetuates such abuse. To speak up may provoke aggression or even a physical assault. Further, does our response change if it is not the patient but someone who is accompanying them who exhibits this behavior?

I conducted a survey of psychiatry HCWs at our institution to evaluate the prevalence of and factors associated with such harassment.
An all-too-common problem
In a December 2020 internal survey at the University of Missouri Department of Psychiatry, 59 of 158 HCWs responded, and 26 (44%) reported experiencing or witnessing on-the-job harassment or abuse. Factors that were statistically significantly associated with experiencing or witnessing on-the-job harassment or abuse included being non-White, working in a patient-facing position, and being a nonphysician patient-facing HCW (Table 1). Factors that were not significantly associated with experiencing or witnessing on-the-job harassment or abuse included clinical setting, HCW age, and HCW gender (Table 2).

In addition to comments from patients and visitors, respondents stated that the harassment or abuse also included:
- physically threatening behavior and assault
- reporting a HCW for HIPAA (Health Insurance Portability and Accountability Act) violations after the HCW declined to provide an early refill of a controlled substance
- being accused of being a bad person for declining to prescribe a specific medication
- insults about not being intelligent enough to be on the treatment team
- comments from colleagues.
At the most basic level of response, the emergency department (ED) remains under the Emergency Medical Treatment and Labor Act (EMTALA) obligation to see, screen, and stabilize any patient, and if psychiatry is consulted in the ED, we should similarly provide this standard of care. Beyond this, we can create behavioral plans for when a relevant diagnosis exists or does not exist, and patients and/or visitors can be terminated from their stay at the location/service/health care system. Whether or not a patient is receiving psychiatric care and/or treatment is irrelevant to the responses to harassment we might consider.
During the incident itself, we are empowered to remove ourselves from the patient encounter. Historically, HCWs have had strong opinions on the next steps, either deciding, “Yes, I am a professional and I will not be bullied,” or “No, I am a professional and I don’t need to deal with this.” Just as we prioritize our patients’ dignities, we should also respect our own and our colleagues’ dignities.
How harassment is handled at our facility
HCWs are commonly unsure whether to “call out” abusive comments during the encounter itself or afterwards. In our hospital, HCWs are encouraged to independently choose to immediately respond, immediately report to a supervisor or hospital security, or defer and report to leadership afterwards via the Patient Safety Network (PSN). The PSN is our hospital’s reporting system for medical errors, near misses, and abuse, neglect, and workplace violence. Relevant examples of abuse, neglect, and workplace violence include:
- Threats. Expression of intent to cause harm, including verbal or written threats and threatening body language
- Physical assault. Attacks ranging from slapping and beating to rape, the use of weapons, or homicide
- Sexual assault. Any type of sexual contact or behavior that occurs without the explicit consent of the recipient, such as forced sexual intercourse, forcible sodomy, child molestation, incest, fondling, and attempted rape.
Continue to: Once complete...
Once complete, the PSN report is sent to Risk Management and other relevant groups, such as a 5-person team of security investigators, who are trained in trauma-informed interviewing and re-directive techniques. This team can immediately speak to the patient face-to-face in the inpatient setting or follow-up via phone in the outpatient setting.
The PSN report may result in the creation of a behavior plan for the patient that outlines the behaviors of concern, staff interventions, and consequences for persistent violations. The behavior plan is saved in the patient’s medical chart, and an alert pops up every time the chart is opened. The behavior plan is reviewed once annually for revision or deletion, as appropriate.
Lessons from our facility’s policy
In our health care system, our primary response to HCW harassment is to create a patient behavior plan that lays out specific expectations, care parameters, and consequences (up to terminating a patient from the entire health care system, except for EMTALA-level care). Clinicians are encouraged to report harassment to hospital administration, and a team of security investigators discusses expectations with the patient and/or visitors to prevent further abuse. We believe that describing our policies may be helpful to other health care systems and HCWs who confront this widespread issue.
During the course of my residency training, I have experienced and witnessed patients and visitors harassing health care workers (HCWs) by cursing or directing racial slurs at them, making sexist comments, or threatening their lives. What should be the correct response to this harassment? To say nothing may avoid conflict, but the silence perpetuates such abuse. To speak up may provoke aggression or even a physical assault. Further, does our response change if it is not the patient but someone who is accompanying them who exhibits this behavior?

I conducted a survey of psychiatry HCWs at our institution to evaluate the prevalence of and factors associated with such harassment.
An all-too-common problem
In a December 2020 internal survey at the University of Missouri Department of Psychiatry, 59 of 158 HCWs responded, and 26 (44%) reported experiencing or witnessing on-the-job harassment or abuse. Factors that were statistically significantly associated with experiencing or witnessing on-the-job harassment or abuse included being non-White, working in a patient-facing position, and being a nonphysician patient-facing HCW (Table 1). Factors that were not significantly associated with experiencing or witnessing on-the-job harassment or abuse included clinical setting, HCW age, and HCW gender (Table 2).

In addition to comments from patients and visitors, respondents stated that the harassment or abuse also included:
- physically threatening behavior and assault
- reporting a HCW for HIPAA (Health Insurance Portability and Accountability Act) violations after the HCW declined to provide an early refill of a controlled substance
- being accused of being a bad person for declining to prescribe a specific medication
- insults about not being intelligent enough to be on the treatment team
- comments from colleagues.
At the most basic level of response, the emergency department (ED) remains under the Emergency Medical Treatment and Labor Act (EMTALA) obligation to see, screen, and stabilize any patient, and if psychiatry is consulted in the ED, we should similarly provide this standard of care. Beyond this, we can create behavioral plans for when a relevant diagnosis exists or does not exist, and patients and/or visitors can be terminated from their stay at the location/service/health care system. Whether or not a patient is receiving psychiatric care and/or treatment is irrelevant to the responses to harassment we might consider.
During the incident itself, we are empowered to remove ourselves from the patient encounter. Historically, HCWs have had strong opinions on the next steps, either deciding, “Yes, I am a professional and I will not be bullied,” or “No, I am a professional and I don’t need to deal with this.” Just as we prioritize our patients’ dignities, we should also respect our own and our colleagues’ dignities.
How harassment is handled at our facility
HCWs are commonly unsure whether to “call out” abusive comments during the encounter itself or afterwards. In our hospital, HCWs are encouraged to independently choose to immediately respond, immediately report to a supervisor or hospital security, or defer and report to leadership afterwards via the Patient Safety Network (PSN). The PSN is our hospital’s reporting system for medical errors, near misses, and abuse, neglect, and workplace violence. Relevant examples of abuse, neglect, and workplace violence include:
- Threats. Expression of intent to cause harm, including verbal or written threats and threatening body language
- Physical assault. Attacks ranging from slapping and beating to rape, the use of weapons, or homicide
- Sexual assault. Any type of sexual contact or behavior that occurs without the explicit consent of the recipient, such as forced sexual intercourse, forcible sodomy, child molestation, incest, fondling, and attempted rape.
Continue to: Once complete...
Once complete, the PSN report is sent to Risk Management and other relevant groups, such as a 5-person team of security investigators, who are trained in trauma-informed interviewing and re-directive techniques. This team can immediately speak to the patient face-to-face in the inpatient setting or follow-up via phone in the outpatient setting.
The PSN report may result in the creation of a behavior plan for the patient that outlines the behaviors of concern, staff interventions, and consequences for persistent violations. The behavior plan is saved in the patient’s medical chart, and an alert pops up every time the chart is opened. The behavior plan is reviewed once annually for revision or deletion, as appropriate.
Lessons from our facility’s policy
In our health care system, our primary response to HCW harassment is to create a patient behavior plan that lays out specific expectations, care parameters, and consequences (up to terminating a patient from the entire health care system, except for EMTALA-level care). Clinicians are encouraged to report harassment to hospital administration, and a team of security investigators discusses expectations with the patient and/or visitors to prevent further abuse. We believe that describing our policies may be helpful to other health care systems and HCWs who confront this widespread issue.
Private practice: The basics for psychiatry trainees
Many psychiatry trainees consider private practice as a career option or form of supplemental income. In my experience, however, residency training may provide limited introduction to the general steps involved in starting a practice. In this article, I briefly summarize what I learned while exploring the private practice option as a psychiatry resident.
A good specialty for private practice
Trainees in the earlier stages of their education should be aware that the first step toward private practice may actually occur during medical school, when they are considering which specialty to pursue. If a student is particularly interested in solo private practice, they may want to select a specialty with the potential for less overhead in an independent setting. Psychiatry typically has lower overhead costs than some other specialties. This gap widens even further with the increased popularity and acceptance of telepsychiatry.
Budgeting and finance
Once you decide to pursue private practice, you will want to consider whether you prefer solo practice or group practice, and part-time or full-time. If working for yourself, you will need to understand business planning and budgeting, including how to project revenue and expenses. When first starting in solo practice—especially if you are not taking over a previously established practice—it is useful to have secondary sources of income. This can be a part-time clinical position, working with on-demand health care companies, contracting, consulting, etc. Many new physicians begin with a full-time position and decide to initiate their private practice on a part-time basis. This approach provides a level of financial security that you otherwise would not have. However, a full-time position requires full-time energy, hours, and attention, and it can be challenging to balance full-time and part-time work. Whichever approach you decide to take, it can be most helpful to simply keep an open mind and always consider looking further into any new opportunity that interests you.
Insurance and licensing
You don’t have to wait to establish your own practice to purchase malpractice insurance. Shop around for the best rates and the coverage that most comprehensively fits your needs. If your training program allows “moonlighting,” you might need your own insurance to work at sites other than your training hospital. Many residents begin to apply for independent state licensure at the same time they begin pursuing moonlighting opportunities. It may be helpful not to wait until the last minute to do this, because the process has quite a few steps and can take a while. If your state requires letters of reference, think about which of your supervisors you can ask for one. If you plan to work in a state other than that of your training location, it may be helpful to simultaneously apply for your medical license in that state, because you will already be going through the process. Certain states offer reciprocity regarding medical licenses. The Interstate Medical Licensure Compact offers an expedited pathway to licensure for qualified physicians who want to practice in multiple states.1
Marketing your practice
Potential sources for building a panel of patients include referral networks, insurance panels, professional organizations, social media, networking, directories, and word of mouth. If you plan to accept health insurance, the directories provided by insurance panels will allow potential patients to find you when searching for practitioners who accept their plan. Professional organizations offer similar directories, and some private companies also provide directories, either for free or for a fee.
Use technology to your advantage
The exciting thing about starting a private practice today is that the technology available to support a small practice has drastically improved. Many software applications can help with scheduling and billing, which minimizes the need for office staff and enables you to be more productive. These programs typically are available via an online subscription that gives you access to an electronic medical record and other features for a monthly fee. Many of these programs provide add-ons such as a website for your practice and integrated telehealth services. While these programs typically perform many of the same functions, each has a different setup and varying workflows. An online search can facilitate a side-by-side comparison of the software programs that most closely meet your needs.
Seek out mentors and consultants
Finally, try to find a private practice mentor, and reach out to as many people as possible who have worked in any type of private practice setting. A mentor can alert you to factors you might not otherwise have considered. It also may be helpful to establish some form of supervision; such opportunities can be found through professional societies and other groups for private practice clinicians. In these groups, you also can ask other clinicians to recommend private practice and practice management consultants.
Stepping into the unknown can be an intimidating experience; however, you will not know what you are capable of until you try. Fortunately, psychiatry offers the flexibility to create a hybrid career that allows you to follow your passion and maintain your level of comfort. The American Psychiatric Association offers members additional information in the practice management resources section of its website.2
1. Interstate Medical Licensure Compact. Information for physicians. 2020. Accessed March 8, 2021. https://www.imlcc.org/information-for-physicians
2. American Psychiatric Association. Online practice handbook. 2021. Accessed March 21, 2021. https://www.psychiatry.org/psychiatrists/practice/practice-management/starting-a-practice/online-practice-handbook
Many psychiatry trainees consider private practice as a career option or form of supplemental income. In my experience, however, residency training may provide limited introduction to the general steps involved in starting a practice. In this article, I briefly summarize what I learned while exploring the private practice option as a psychiatry resident.
A good specialty for private practice
Trainees in the earlier stages of their education should be aware that the first step toward private practice may actually occur during medical school, when they are considering which specialty to pursue. If a student is particularly interested in solo private practice, they may want to select a specialty with the potential for less overhead in an independent setting. Psychiatry typically has lower overhead costs than some other specialties. This gap widens even further with the increased popularity and acceptance of telepsychiatry.
Budgeting and finance
Once you decide to pursue private practice, you will want to consider whether you prefer solo practice or group practice, and part-time or full-time. If working for yourself, you will need to understand business planning and budgeting, including how to project revenue and expenses. When first starting in solo practice—especially if you are not taking over a previously established practice—it is useful to have secondary sources of income. This can be a part-time clinical position, working with on-demand health care companies, contracting, consulting, etc. Many new physicians begin with a full-time position and decide to initiate their private practice on a part-time basis. This approach provides a level of financial security that you otherwise would not have. However, a full-time position requires full-time energy, hours, and attention, and it can be challenging to balance full-time and part-time work. Whichever approach you decide to take, it can be most helpful to simply keep an open mind and always consider looking further into any new opportunity that interests you.
Insurance and licensing
You don’t have to wait to establish your own practice to purchase malpractice insurance. Shop around for the best rates and the coverage that most comprehensively fits your needs. If your training program allows “moonlighting,” you might need your own insurance to work at sites other than your training hospital. Many residents begin to apply for independent state licensure at the same time they begin pursuing moonlighting opportunities. It may be helpful not to wait until the last minute to do this, because the process has quite a few steps and can take a while. If your state requires letters of reference, think about which of your supervisors you can ask for one. If you plan to work in a state other than that of your training location, it may be helpful to simultaneously apply for your medical license in that state, because you will already be going through the process. Certain states offer reciprocity regarding medical licenses. The Interstate Medical Licensure Compact offers an expedited pathway to licensure for qualified physicians who want to practice in multiple states.1
Marketing your practice
Potential sources for building a panel of patients include referral networks, insurance panels, professional organizations, social media, networking, directories, and word of mouth. If you plan to accept health insurance, the directories provided by insurance panels will allow potential patients to find you when searching for practitioners who accept their plan. Professional organizations offer similar directories, and some private companies also provide directories, either for free or for a fee.
Use technology to your advantage
The exciting thing about starting a private practice today is that the technology available to support a small practice has drastically improved. Many software applications can help with scheduling and billing, which minimizes the need for office staff and enables you to be more productive. These programs typically are available via an online subscription that gives you access to an electronic medical record and other features for a monthly fee. Many of these programs provide add-ons such as a website for your practice and integrated telehealth services. While these programs typically perform many of the same functions, each has a different setup and varying workflows. An online search can facilitate a side-by-side comparison of the software programs that most closely meet your needs.
Seek out mentors and consultants
Finally, try to find a private practice mentor, and reach out to as many people as possible who have worked in any type of private practice setting. A mentor can alert you to factors you might not otherwise have considered. It also may be helpful to establish some form of supervision; such opportunities can be found through professional societies and other groups for private practice clinicians. In these groups, you also can ask other clinicians to recommend private practice and practice management consultants.
Stepping into the unknown can be an intimidating experience; however, you will not know what you are capable of until you try. Fortunately, psychiatry offers the flexibility to create a hybrid career that allows you to follow your passion and maintain your level of comfort. The American Psychiatric Association offers members additional information in the practice management resources section of its website.2
Many psychiatry trainees consider private practice as a career option or form of supplemental income. In my experience, however, residency training may provide limited introduction to the general steps involved in starting a practice. In this article, I briefly summarize what I learned while exploring the private practice option as a psychiatry resident.
A good specialty for private practice
Trainees in the earlier stages of their education should be aware that the first step toward private practice may actually occur during medical school, when they are considering which specialty to pursue. If a student is particularly interested in solo private practice, they may want to select a specialty with the potential for less overhead in an independent setting. Psychiatry typically has lower overhead costs than some other specialties. This gap widens even further with the increased popularity and acceptance of telepsychiatry.
Budgeting and finance
Once you decide to pursue private practice, you will want to consider whether you prefer solo practice or group practice, and part-time or full-time. If working for yourself, you will need to understand business planning and budgeting, including how to project revenue and expenses. When first starting in solo practice—especially if you are not taking over a previously established practice—it is useful to have secondary sources of income. This can be a part-time clinical position, working with on-demand health care companies, contracting, consulting, etc. Many new physicians begin with a full-time position and decide to initiate their private practice on a part-time basis. This approach provides a level of financial security that you otherwise would not have. However, a full-time position requires full-time energy, hours, and attention, and it can be challenging to balance full-time and part-time work. Whichever approach you decide to take, it can be most helpful to simply keep an open mind and always consider looking further into any new opportunity that interests you.
Insurance and licensing
You don’t have to wait to establish your own practice to purchase malpractice insurance. Shop around for the best rates and the coverage that most comprehensively fits your needs. If your training program allows “moonlighting,” you might need your own insurance to work at sites other than your training hospital. Many residents begin to apply for independent state licensure at the same time they begin pursuing moonlighting opportunities. It may be helpful not to wait until the last minute to do this, because the process has quite a few steps and can take a while. If your state requires letters of reference, think about which of your supervisors you can ask for one. If you plan to work in a state other than that of your training location, it may be helpful to simultaneously apply for your medical license in that state, because you will already be going through the process. Certain states offer reciprocity regarding medical licenses. The Interstate Medical Licensure Compact offers an expedited pathway to licensure for qualified physicians who want to practice in multiple states.1
Marketing your practice
Potential sources for building a panel of patients include referral networks, insurance panels, professional organizations, social media, networking, directories, and word of mouth. If you plan to accept health insurance, the directories provided by insurance panels will allow potential patients to find you when searching for practitioners who accept their plan. Professional organizations offer similar directories, and some private companies also provide directories, either for free or for a fee.
Use technology to your advantage
The exciting thing about starting a private practice today is that the technology available to support a small practice has drastically improved. Many software applications can help with scheduling and billing, which minimizes the need for office staff and enables you to be more productive. These programs typically are available via an online subscription that gives you access to an electronic medical record and other features for a monthly fee. Many of these programs provide add-ons such as a website for your practice and integrated telehealth services. While these programs typically perform many of the same functions, each has a different setup and varying workflows. An online search can facilitate a side-by-side comparison of the software programs that most closely meet your needs.
Seek out mentors and consultants
Finally, try to find a private practice mentor, and reach out to as many people as possible who have worked in any type of private practice setting. A mentor can alert you to factors you might not otherwise have considered. It also may be helpful to establish some form of supervision; such opportunities can be found through professional societies and other groups for private practice clinicians. In these groups, you also can ask other clinicians to recommend private practice and practice management consultants.
Stepping into the unknown can be an intimidating experience; however, you will not know what you are capable of until you try. Fortunately, psychiatry offers the flexibility to create a hybrid career that allows you to follow your passion and maintain your level of comfort. The American Psychiatric Association offers members additional information in the practice management resources section of its website.2
1. Interstate Medical Licensure Compact. Information for physicians. 2020. Accessed March 8, 2021. https://www.imlcc.org/information-for-physicians
2. American Psychiatric Association. Online practice handbook. 2021. Accessed March 21, 2021. https://www.psychiatry.org/psychiatrists/practice/practice-management/starting-a-practice/online-practice-handbook
1. Interstate Medical Licensure Compact. Information for physicians. 2020. Accessed March 8, 2021. https://www.imlcc.org/information-for-physicians
2. American Psychiatric Association. Online practice handbook. 2021. Accessed March 21, 2021. https://www.psychiatry.org/psychiatrists/practice/practice-management/starting-a-practice/online-practice-handbook
Psychiatry is Neurology: White matter pathology permeates psychiatric disorders
Ask neurologists or psychiatrists to name a white matter (WM) brain disease and they are very likely to say multiple sclerosis (MS), a demyelinating brain disorder caused by immune-mediated destruction of oligodendrocytes, the glial cells that manufacture myelin without which brain communications would come to a standstill.
MS is often associated with mood or psychotic disorders, yet it is regarded as a neurologic illness, not a psychiatric disorder.
Many neurologists and psychiatrists may not be aware that during the past few years, multiple diffusion tensor imaging (DTI) studies have revealed that many psychiatric disorders are associated with WM pathology.1
Most people think that the brain is composed mostly of neurons, but in fact the bulk of brain volume (60%) is comprised of WM and only 40% is gray matter, which includes both neurons and glial cells (astroglia, microglia, and oligodendroglia). WM includes >137,000 km of myelinated fibers, an extensive network that connects all brain regions and integrates its complex, multifaceted functions, culminating in a unified sense of self and agency.
The role of the corpus callosum
Early in my research career, I became interested in the corpus callosum, the largest interhemispheric WM commissure connecting homologous areas across the 2 cerebral hemispheres. It is comprised of 200 million fibers of various diameters. Reasons for my fascination with the corpus callosum were:
The studies of Roger Sperry, the 1981 Nobel Laureate who led the team that was awarded the prize for split-brain research, which involved patients whose corpus callosum was cut to prevent the transfer of intractable epilepsy from 1 hemisphere to the other. Using a tachistoscope that he designed, Sperry discovered that the right and left hemispheres are 2 independent spheres of consciousness (ie, 2 individuals) with different skills.2 Cerebral dominance (laterality) fully integrates the 2 hemispheres via the corpus callosum, with a verbal hemisphere (the left, in 90% of people) dominating the other hemisphere and serving as the “spokesman self.” Thus, we all have 2 persons in our brain completely integrated into 1 “self.”2 This led me to wonder about the effects of an impaired corpus callosum on the “unified self.”
Postmortem and MRI studies conducted by our research group showed a significant difference in the thickness of the corpus callosum in a group of patients with schizophrenia vs healthy controls, which implied abnormal connectivity across the left and right hemispheres.3
Continue to: I then conducted a clinical study
I then conducted a clinical study examining patients with tumors impinging on the corpus callosum, which revealed that they developed psychotic symptoms (delusions and hallucinations).4 This study suggested that disrupting the integrity of the callosal inter-hemispheric fibers can trigger fixed false beliefs and perceptual anomalies.4
A ‘dysconnection’ between hemispheres
I translated those observations about the corpus callosum into a published hypothesis5 in which I proposed that Schneider’s First-Rank Symptoms of schizophrenia of thought insertion, thought withdrawal, and thought broadcasting—as well as delusional experiences of “external control”—may be due to a neurobiologic abnormality in the corpus callosum that disrupts the flow of ongoing bits of information transmitted from the left to the right hemisphere, and vice versa. I proposed in my model that this disruption leads to the verbal left hemisphere of a psychotic patient to describe having thoughts inserted into it from an alien source, failing to recognize that the thoughts it is receiving are being transmitted from the disconnected right hemisphere, which is no longer part of the “self.” Similarly, impulses from the right hemispheric consciousness are now perceived by the patient’s verbal left hemisphere (which talks to the examining physician) as “external control.” Thus, I postulated that an abnormal corpus callosum structure would lead to a “dysconnection” (not “disconnection”) between the 2 hemispheres, and that anomalous dysconnectivity may generate both delusions and hallucinations. 6
Two decades later, my assumptions were vindicated when DTI was invented, enabling the measurement of WM integrity, including the corpus callosum, the largest body of WM in the brain. Table 1 defines the main parameters of WM integrity, anisotropy and diffusivity, which measure water flow inside WM fibers.

During the past 15 years, many studies have confirmed the presence of significant abnormalities in the myelinated fibers of the corpus callosum in schizophrenia, which can be considered a validation of my hypothesis that the corpus callosum becomes a dysfunctional channel of communications between the right and left hemisphere. Subsequently, DTI studies have reported a spectrum of WM pathologies in various other cerebral bundles and not only in schizophrenia, but also in other major psychiatric disorders (Table 27-19).
The pathophysiology of WM pathology in many psychiatric disorders may include neurodevelopmental aberrations (genetic, environmental, or both, which may alter WM structure and/or myelination), neuroinflammation, or oxidative stress (free radicals), which can cause disintegration of the vital myelin sheaths, leading to disruption of brain connectivity.6,7 Researchers now consider the brain’s WM network dysconnectivity as generating a variety of psychiatric symptoms, including psychosis, depression, mania, anxiety, autism, aggression, impulsivity, psychopathy, and cognitive impairments.
It is not surprising that WM repair has become a therapeutic target in psychiatry and neurology. Among the strategies being investigated are inhibiting the Nogo-A signaling pathways20 or modulating the Lingo-1 signaling.21 However, the most well-established myelin repair pathway is prolactin, a neuroprotective hormone with several beneficial effects on the brain (Table 322,23), including the proliferation of oligodendroglia, the main source of myelin (and the number of which declines in schizophrenia). Antipsychotics that increase prolactin have been shown to increase WM volume.24,25 It has even been proposed that a decline in oligodendrocytes and low myelin synthesis may be one of the neurobiologic pathologies in schizophrenia.26 One of the 24 neuroprotective properties of the second-generation antipsychotics (SGAs) is the restoration of WM integrity.27 It’s worth noting that WM pathology has been found to be present at the onset of schizophrenia before treatment, and that SGAs have been reported to correct it.28

Continue to: In conclusion...
In conclusion, psychiatric disorders, usually referred to as “mental illnesses,” are unquestionably neurologic disorders. Similarly, all neurologic disorders are associated with psychiatric manifestations. WM pathology is only 1 of numerous structural brain abnormalities that have been documented across psychiatric disorders, which proves that psychiatry is a clinical neuroscience, just like neurology. I strongly advocate that psychiatry and neurology reunite into a single medical specialty. Both focus on disorders of brain structure and/or function, and these disorders also share much more than WM pathology.29
1. Sagarwala R and Nasrallah HA. White matter pathology is shared across multiple psychiatric brain disorders: Is abnormal diffusivity a transdiagnostic biomarker for psychopathology? Biomarkers in Neuropsychiatry. 2020;2:00010. https://doi.org/10.1016/j.bionps.2019.100010
2. Pearce JMS; FRCP. The “split brain” and Roger Wolcott Sperry (1913-1994). Rev Neurol (Paris). 2019;175(4):217-220.
3. Nasrallah HA, Andreasen NC, Coffman JA, et al. A controlled magnetic resonance imaging study of corpus callosum thickness in schizophrenia. Biol Psychiatry. 1986;21(3):274-282.
4. Nasrallah HA, McChesney CM. Psychopathology of corpus callosum tumors. Biol Psychiatry. 1981;16(7):663-669.
5. Nasrallah HA. The unintegrated right cerebral hemispheric consciousness as alien intruder: a possible mechanism for Schneiderian delusions in schizophrenia. Compr Psychiatry. 1985;26(3):273-282.
6. Friston K, Brown HR, Siemerkus J, et al. The dysconnection hypothesis (2016). Schizophr Res. 2016;176(2-3):83-94.
7. Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res. 2015;161(1):102-112.
8. Benedetti F, Bollettini I. Recent findings on the role of white matter pathology in bipolar disorder. Harv Rev Psychiatry. 2014;22(6):338-341.
9. Zheng H, Bergamino M, Ford BN, et al; Tulsa 1000 Investigators. Replicable association between human cytomegalovirus infection and reduced white matter fractional anisotropy in major depressive disorder. Neuropsychopharmacology. 2021;46(5):928-938.
10. Sagarwala R, Nasrallah HA. A systematic review of diffusion tensor imaging studies in drug-naïve OCD patients before and after pharmacotherapy. Ann Clin Psychiatry. 2020;32(1):42-47.
11. Lee KS, Lee SH. White matter-based structural brain network of anxiety. Adv Exp Med Biol. 2020;1191:61-70.
12. Swanson MR, Hazlett HC. White matter as a monitoring biomarker for neurodevelopmental disorder intervention studies. J Neurodev Disord. 2019;11(1):33.
13. Hampton WH, Hanik IM, Olson IR. Substance abuse and white matter: findings, limitations, and future of diffusion tensor imaging research. Drug Alcohol Depend. 2019;197:288-298.
14. Waller R, Dotterer HL, Murray L, et al. White-matter tract abnormalities and antisocial behavior: a systematic review of diffusion tensor imaging studies across development. Neuroimage Clin. 2017;14:201-215.
15. Wolf RC, Pujara MS, Motzkin JC, et al. Interpersonal traits of psychopathy linked to reduced integrity of the uncinate fasciculus. Hum Brain Mapp. 2015;36(10):4202-4209.
16. Puzzo I, Seunarine K, Sully K, et al. Altered white-matter microstructure in conduct disorder is specifically associated with elevated callous-unemotional traits. J Abnorm Child Psychol. 2018;46(7):1451-1466.
17. Finger EC, Marsh A, Blair KS, et al. Impaired functional but preserved structural connectivity in limbic white matter tracts in youth with conduct disorder or oppositional defiant disorder plus psychopathic traits. Psychiatry Res. 2012;202(3):239-244.
18. Li C, Dong M, Womer FY, et al. Transdiagnostic time-varying dysconnectivity across major psychiatric disorders. Hum Brain Mapp. 2021;42(4):1182-1196.
19. Khanbabaei M, Hughes E, Ellegood J, et al. Precocious myelination in a mouse model of autism. Transl Psychiatry. 2019;9(1):251.
20. Petratos S, Theotokis P, Kim MJ, et al. That’s a wrap! Molecular drivers governing neuronal nogo receptor-dependent myelin plasticity and integrity. Front Cell Neurosci. 2020;14:227
21. Fernandez-Enright F, Andrews JL, Newell KA, et al. Novel implications of Lingo-1 and its signaling partners in schizophrenia. Transl Psychiatry. 2014;4(1):e348. doi: 10.1038/tp.2013.121
22. Bartzokis G, Lu PH, Stewart SB, et al. In vivo evidence of differential impact of typical and atypical antipsychotics on intracortical myelin in adults with schizophrenia. Schizophr Res. 2009;113(2-3):322-331.
23. Bartzokis G, Lu PH, Amar CP, et al. Long acting injection versus oral risperidone in first-episode schizophrenia: differential impact on white matter myelination trajectory. Schizophr Res. 2011 Oct;132(1):35-41
24. Tishler TA, Bartzokis G, Lu PH, et al. Abnormal trajectory of intracortical myelination in schizophrenia implicates white matter in disease pathophysiology and the therapeutic mechanism of action of antipsychotics. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3(5):454-462.
25. Ren Y, Wang H, Xiao L. Improving myelin/oligodendrocyte-related dysfunction: a new mechanism of antipsychotics in the treatment of schizophrenia? Int J Neuropsychopharmacol. 2013;16(3):691-700.
26. Dietz AG, Goldman SA, Nedergaard M. Glial cells in schizophrenia: a unified hypothesis. Lancet Psychiatry. 2020;7(3):272-281.
27. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7
28. Sagarwala R, Nasrallah HA. (In press.) The effect of antipsychotic medications on white matter integrity in first-episode drug naïve patients with psychosis. Asian Journal of Psychiatry.
29. Nasrallah HA. Let’s tear down the silos and reunify psychiatry and neurology. Current Psychiatry. 2013;12(8):9-10.
Ask neurologists or psychiatrists to name a white matter (WM) brain disease and they are very likely to say multiple sclerosis (MS), a demyelinating brain disorder caused by immune-mediated destruction of oligodendrocytes, the glial cells that manufacture myelin without which brain communications would come to a standstill.
MS is often associated with mood or psychotic disorders, yet it is regarded as a neurologic illness, not a psychiatric disorder.
Many neurologists and psychiatrists may not be aware that during the past few years, multiple diffusion tensor imaging (DTI) studies have revealed that many psychiatric disorders are associated with WM pathology.1
Most people think that the brain is composed mostly of neurons, but in fact the bulk of brain volume (60%) is comprised of WM and only 40% is gray matter, which includes both neurons and glial cells (astroglia, microglia, and oligodendroglia). WM includes >137,000 km of myelinated fibers, an extensive network that connects all brain regions and integrates its complex, multifaceted functions, culminating in a unified sense of self and agency.
The role of the corpus callosum
Early in my research career, I became interested in the corpus callosum, the largest interhemispheric WM commissure connecting homologous areas across the 2 cerebral hemispheres. It is comprised of 200 million fibers of various diameters. Reasons for my fascination with the corpus callosum were:
The studies of Roger Sperry, the 1981 Nobel Laureate who led the team that was awarded the prize for split-brain research, which involved patients whose corpus callosum was cut to prevent the transfer of intractable epilepsy from 1 hemisphere to the other. Using a tachistoscope that he designed, Sperry discovered that the right and left hemispheres are 2 independent spheres of consciousness (ie, 2 individuals) with different skills.2 Cerebral dominance (laterality) fully integrates the 2 hemispheres via the corpus callosum, with a verbal hemisphere (the left, in 90% of people) dominating the other hemisphere and serving as the “spokesman self.” Thus, we all have 2 persons in our brain completely integrated into 1 “self.”2 This led me to wonder about the effects of an impaired corpus callosum on the “unified self.”
Postmortem and MRI studies conducted by our research group showed a significant difference in the thickness of the corpus callosum in a group of patients with schizophrenia vs healthy controls, which implied abnormal connectivity across the left and right hemispheres.3
Continue to: I then conducted a clinical study
I then conducted a clinical study examining patients with tumors impinging on the corpus callosum, which revealed that they developed psychotic symptoms (delusions and hallucinations).4 This study suggested that disrupting the integrity of the callosal inter-hemispheric fibers can trigger fixed false beliefs and perceptual anomalies.4
A ‘dysconnection’ between hemispheres
I translated those observations about the corpus callosum into a published hypothesis5 in which I proposed that Schneider’s First-Rank Symptoms of schizophrenia of thought insertion, thought withdrawal, and thought broadcasting—as well as delusional experiences of “external control”—may be due to a neurobiologic abnormality in the corpus callosum that disrupts the flow of ongoing bits of information transmitted from the left to the right hemisphere, and vice versa. I proposed in my model that this disruption leads to the verbal left hemisphere of a psychotic patient to describe having thoughts inserted into it from an alien source, failing to recognize that the thoughts it is receiving are being transmitted from the disconnected right hemisphere, which is no longer part of the “self.” Similarly, impulses from the right hemispheric consciousness are now perceived by the patient’s verbal left hemisphere (which talks to the examining physician) as “external control.” Thus, I postulated that an abnormal corpus callosum structure would lead to a “dysconnection” (not “disconnection”) between the 2 hemispheres, and that anomalous dysconnectivity may generate both delusions and hallucinations. 6
Two decades later, my assumptions were vindicated when DTI was invented, enabling the measurement of WM integrity, including the corpus callosum, the largest body of WM in the brain. Table 1 defines the main parameters of WM integrity, anisotropy and diffusivity, which measure water flow inside WM fibers.

During the past 15 years, many studies have confirmed the presence of significant abnormalities in the myelinated fibers of the corpus callosum in schizophrenia, which can be considered a validation of my hypothesis that the corpus callosum becomes a dysfunctional channel of communications between the right and left hemisphere. Subsequently, DTI studies have reported a spectrum of WM pathologies in various other cerebral bundles and not only in schizophrenia, but also in other major psychiatric disorders (Table 27-19).
The pathophysiology of WM pathology in many psychiatric disorders may include neurodevelopmental aberrations (genetic, environmental, or both, which may alter WM structure and/or myelination), neuroinflammation, or oxidative stress (free radicals), which can cause disintegration of the vital myelin sheaths, leading to disruption of brain connectivity.6,7 Researchers now consider the brain’s WM network dysconnectivity as generating a variety of psychiatric symptoms, including psychosis, depression, mania, anxiety, autism, aggression, impulsivity, psychopathy, and cognitive impairments.
It is not surprising that WM repair has become a therapeutic target in psychiatry and neurology. Among the strategies being investigated are inhibiting the Nogo-A signaling pathways20 or modulating the Lingo-1 signaling.21 However, the most well-established myelin repair pathway is prolactin, a neuroprotective hormone with several beneficial effects on the brain (Table 322,23), including the proliferation of oligodendroglia, the main source of myelin (and the number of which declines in schizophrenia). Antipsychotics that increase prolactin have been shown to increase WM volume.24,25 It has even been proposed that a decline in oligodendrocytes and low myelin synthesis may be one of the neurobiologic pathologies in schizophrenia.26 One of the 24 neuroprotective properties of the second-generation antipsychotics (SGAs) is the restoration of WM integrity.27 It’s worth noting that WM pathology has been found to be present at the onset of schizophrenia before treatment, and that SGAs have been reported to correct it.28

Continue to: In conclusion...
In conclusion, psychiatric disorders, usually referred to as “mental illnesses,” are unquestionably neurologic disorders. Similarly, all neurologic disorders are associated with psychiatric manifestations. WM pathology is only 1 of numerous structural brain abnormalities that have been documented across psychiatric disorders, which proves that psychiatry is a clinical neuroscience, just like neurology. I strongly advocate that psychiatry and neurology reunite into a single medical specialty. Both focus on disorders of brain structure and/or function, and these disorders also share much more than WM pathology.29
Ask neurologists or psychiatrists to name a white matter (WM) brain disease and they are very likely to say multiple sclerosis (MS), a demyelinating brain disorder caused by immune-mediated destruction of oligodendrocytes, the glial cells that manufacture myelin without which brain communications would come to a standstill.
MS is often associated with mood or psychotic disorders, yet it is regarded as a neurologic illness, not a psychiatric disorder.
Many neurologists and psychiatrists may not be aware that during the past few years, multiple diffusion tensor imaging (DTI) studies have revealed that many psychiatric disorders are associated with WM pathology.1
Most people think that the brain is composed mostly of neurons, but in fact the bulk of brain volume (60%) is comprised of WM and only 40% is gray matter, which includes both neurons and glial cells (astroglia, microglia, and oligodendroglia). WM includes >137,000 km of myelinated fibers, an extensive network that connects all brain regions and integrates its complex, multifaceted functions, culminating in a unified sense of self and agency.
The role of the corpus callosum
Early in my research career, I became interested in the corpus callosum, the largest interhemispheric WM commissure connecting homologous areas across the 2 cerebral hemispheres. It is comprised of 200 million fibers of various diameters. Reasons for my fascination with the corpus callosum were:
The studies of Roger Sperry, the 1981 Nobel Laureate who led the team that was awarded the prize for split-brain research, which involved patients whose corpus callosum was cut to prevent the transfer of intractable epilepsy from 1 hemisphere to the other. Using a tachistoscope that he designed, Sperry discovered that the right and left hemispheres are 2 independent spheres of consciousness (ie, 2 individuals) with different skills.2 Cerebral dominance (laterality) fully integrates the 2 hemispheres via the corpus callosum, with a verbal hemisphere (the left, in 90% of people) dominating the other hemisphere and serving as the “spokesman self.” Thus, we all have 2 persons in our brain completely integrated into 1 “self.”2 This led me to wonder about the effects of an impaired corpus callosum on the “unified self.”
Postmortem and MRI studies conducted by our research group showed a significant difference in the thickness of the corpus callosum in a group of patients with schizophrenia vs healthy controls, which implied abnormal connectivity across the left and right hemispheres.3
Continue to: I then conducted a clinical study
I then conducted a clinical study examining patients with tumors impinging on the corpus callosum, which revealed that they developed psychotic symptoms (delusions and hallucinations).4 This study suggested that disrupting the integrity of the callosal inter-hemispheric fibers can trigger fixed false beliefs and perceptual anomalies.4
A ‘dysconnection’ between hemispheres
I translated those observations about the corpus callosum into a published hypothesis5 in which I proposed that Schneider’s First-Rank Symptoms of schizophrenia of thought insertion, thought withdrawal, and thought broadcasting—as well as delusional experiences of “external control”—may be due to a neurobiologic abnormality in the corpus callosum that disrupts the flow of ongoing bits of information transmitted from the left to the right hemisphere, and vice versa. I proposed in my model that this disruption leads to the verbal left hemisphere of a psychotic patient to describe having thoughts inserted into it from an alien source, failing to recognize that the thoughts it is receiving are being transmitted from the disconnected right hemisphere, which is no longer part of the “self.” Similarly, impulses from the right hemispheric consciousness are now perceived by the patient’s verbal left hemisphere (which talks to the examining physician) as “external control.” Thus, I postulated that an abnormal corpus callosum structure would lead to a “dysconnection” (not “disconnection”) between the 2 hemispheres, and that anomalous dysconnectivity may generate both delusions and hallucinations. 6
Two decades later, my assumptions were vindicated when DTI was invented, enabling the measurement of WM integrity, including the corpus callosum, the largest body of WM in the brain. Table 1 defines the main parameters of WM integrity, anisotropy and diffusivity, which measure water flow inside WM fibers.

During the past 15 years, many studies have confirmed the presence of significant abnormalities in the myelinated fibers of the corpus callosum in schizophrenia, which can be considered a validation of my hypothesis that the corpus callosum becomes a dysfunctional channel of communications between the right and left hemisphere. Subsequently, DTI studies have reported a spectrum of WM pathologies in various other cerebral bundles and not only in schizophrenia, but also in other major psychiatric disorders (Table 27-19).
The pathophysiology of WM pathology in many psychiatric disorders may include neurodevelopmental aberrations (genetic, environmental, or both, which may alter WM structure and/or myelination), neuroinflammation, or oxidative stress (free radicals), which can cause disintegration of the vital myelin sheaths, leading to disruption of brain connectivity.6,7 Researchers now consider the brain’s WM network dysconnectivity as generating a variety of psychiatric symptoms, including psychosis, depression, mania, anxiety, autism, aggression, impulsivity, psychopathy, and cognitive impairments.
It is not surprising that WM repair has become a therapeutic target in psychiatry and neurology. Among the strategies being investigated are inhibiting the Nogo-A signaling pathways20 or modulating the Lingo-1 signaling.21 However, the most well-established myelin repair pathway is prolactin, a neuroprotective hormone with several beneficial effects on the brain (Table 322,23), including the proliferation of oligodendroglia, the main source of myelin (and the number of which declines in schizophrenia). Antipsychotics that increase prolactin have been shown to increase WM volume.24,25 It has even been proposed that a decline in oligodendrocytes and low myelin synthesis may be one of the neurobiologic pathologies in schizophrenia.26 One of the 24 neuroprotective properties of the second-generation antipsychotics (SGAs) is the restoration of WM integrity.27 It’s worth noting that WM pathology has been found to be present at the onset of schizophrenia before treatment, and that SGAs have been reported to correct it.28

Continue to: In conclusion...
In conclusion, psychiatric disorders, usually referred to as “mental illnesses,” are unquestionably neurologic disorders. Similarly, all neurologic disorders are associated with psychiatric manifestations. WM pathology is only 1 of numerous structural brain abnormalities that have been documented across psychiatric disorders, which proves that psychiatry is a clinical neuroscience, just like neurology. I strongly advocate that psychiatry and neurology reunite into a single medical specialty. Both focus on disorders of brain structure and/or function, and these disorders also share much more than WM pathology.29
1. Sagarwala R and Nasrallah HA. White matter pathology is shared across multiple psychiatric brain disorders: Is abnormal diffusivity a transdiagnostic biomarker for psychopathology? Biomarkers in Neuropsychiatry. 2020;2:00010. https://doi.org/10.1016/j.bionps.2019.100010
2. Pearce JMS; FRCP. The “split brain” and Roger Wolcott Sperry (1913-1994). Rev Neurol (Paris). 2019;175(4):217-220.
3. Nasrallah HA, Andreasen NC, Coffman JA, et al. A controlled magnetic resonance imaging study of corpus callosum thickness in schizophrenia. Biol Psychiatry. 1986;21(3):274-282.
4. Nasrallah HA, McChesney CM. Psychopathology of corpus callosum tumors. Biol Psychiatry. 1981;16(7):663-669.
5. Nasrallah HA. The unintegrated right cerebral hemispheric consciousness as alien intruder: a possible mechanism for Schneiderian delusions in schizophrenia. Compr Psychiatry. 1985;26(3):273-282.
6. Friston K, Brown HR, Siemerkus J, et al. The dysconnection hypothesis (2016). Schizophr Res. 2016;176(2-3):83-94.
7. Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res. 2015;161(1):102-112.
8. Benedetti F, Bollettini I. Recent findings on the role of white matter pathology in bipolar disorder. Harv Rev Psychiatry. 2014;22(6):338-341.
9. Zheng H, Bergamino M, Ford BN, et al; Tulsa 1000 Investigators. Replicable association between human cytomegalovirus infection and reduced white matter fractional anisotropy in major depressive disorder. Neuropsychopharmacology. 2021;46(5):928-938.
10. Sagarwala R, Nasrallah HA. A systematic review of diffusion tensor imaging studies in drug-naïve OCD patients before and after pharmacotherapy. Ann Clin Psychiatry. 2020;32(1):42-47.
11. Lee KS, Lee SH. White matter-based structural brain network of anxiety. Adv Exp Med Biol. 2020;1191:61-70.
12. Swanson MR, Hazlett HC. White matter as a monitoring biomarker for neurodevelopmental disorder intervention studies. J Neurodev Disord. 2019;11(1):33.
13. Hampton WH, Hanik IM, Olson IR. Substance abuse and white matter: findings, limitations, and future of diffusion tensor imaging research. Drug Alcohol Depend. 2019;197:288-298.
14. Waller R, Dotterer HL, Murray L, et al. White-matter tract abnormalities and antisocial behavior: a systematic review of diffusion tensor imaging studies across development. Neuroimage Clin. 2017;14:201-215.
15. Wolf RC, Pujara MS, Motzkin JC, et al. Interpersonal traits of psychopathy linked to reduced integrity of the uncinate fasciculus. Hum Brain Mapp. 2015;36(10):4202-4209.
16. Puzzo I, Seunarine K, Sully K, et al. Altered white-matter microstructure in conduct disorder is specifically associated with elevated callous-unemotional traits. J Abnorm Child Psychol. 2018;46(7):1451-1466.
17. Finger EC, Marsh A, Blair KS, et al. Impaired functional but preserved structural connectivity in limbic white matter tracts in youth with conduct disorder or oppositional defiant disorder plus psychopathic traits. Psychiatry Res. 2012;202(3):239-244.
18. Li C, Dong M, Womer FY, et al. Transdiagnostic time-varying dysconnectivity across major psychiatric disorders. Hum Brain Mapp. 2021;42(4):1182-1196.
19. Khanbabaei M, Hughes E, Ellegood J, et al. Precocious myelination in a mouse model of autism. Transl Psychiatry. 2019;9(1):251.
20. Petratos S, Theotokis P, Kim MJ, et al. That’s a wrap! Molecular drivers governing neuronal nogo receptor-dependent myelin plasticity and integrity. Front Cell Neurosci. 2020;14:227
21. Fernandez-Enright F, Andrews JL, Newell KA, et al. Novel implications of Lingo-1 and its signaling partners in schizophrenia. Transl Psychiatry. 2014;4(1):e348. doi: 10.1038/tp.2013.121
22. Bartzokis G, Lu PH, Stewart SB, et al. In vivo evidence of differential impact of typical and atypical antipsychotics on intracortical myelin in adults with schizophrenia. Schizophr Res. 2009;113(2-3):322-331.
23. Bartzokis G, Lu PH, Amar CP, et al. Long acting injection versus oral risperidone in first-episode schizophrenia: differential impact on white matter myelination trajectory. Schizophr Res. 2011 Oct;132(1):35-41
24. Tishler TA, Bartzokis G, Lu PH, et al. Abnormal trajectory of intracortical myelination in schizophrenia implicates white matter in disease pathophysiology and the therapeutic mechanism of action of antipsychotics. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3(5):454-462.
25. Ren Y, Wang H, Xiao L. Improving myelin/oligodendrocyte-related dysfunction: a new mechanism of antipsychotics in the treatment of schizophrenia? Int J Neuropsychopharmacol. 2013;16(3):691-700.
26. Dietz AG, Goldman SA, Nedergaard M. Glial cells in schizophrenia: a unified hypothesis. Lancet Psychiatry. 2020;7(3):272-281.
27. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7
28. Sagarwala R, Nasrallah HA. (In press.) The effect of antipsychotic medications on white matter integrity in first-episode drug naïve patients with psychosis. Asian Journal of Psychiatry.
29. Nasrallah HA. Let’s tear down the silos and reunify psychiatry and neurology. Current Psychiatry. 2013;12(8):9-10.
1. Sagarwala R and Nasrallah HA. White matter pathology is shared across multiple psychiatric brain disorders: Is abnormal diffusivity a transdiagnostic biomarker for psychopathology? Biomarkers in Neuropsychiatry. 2020;2:00010. https://doi.org/10.1016/j.bionps.2019.100010
2. Pearce JMS; FRCP. The “split brain” and Roger Wolcott Sperry (1913-1994). Rev Neurol (Paris). 2019;175(4):217-220.
3. Nasrallah HA, Andreasen NC, Coffman JA, et al. A controlled magnetic resonance imaging study of corpus callosum thickness in schizophrenia. Biol Psychiatry. 1986;21(3):274-282.
4. Nasrallah HA, McChesney CM. Psychopathology of corpus callosum tumors. Biol Psychiatry. 1981;16(7):663-669.
5. Nasrallah HA. The unintegrated right cerebral hemispheric consciousness as alien intruder: a possible mechanism for Schneiderian delusions in schizophrenia. Compr Psychiatry. 1985;26(3):273-282.
6. Friston K, Brown HR, Siemerkus J, et al. The dysconnection hypothesis (2016). Schizophr Res. 2016;176(2-3):83-94.
7. Najjar S, Pearlman DM. Neuroinflammation and white matter pathology in schizophrenia: systematic review. Schizophr Res. 2015;161(1):102-112.
8. Benedetti F, Bollettini I. Recent findings on the role of white matter pathology in bipolar disorder. Harv Rev Psychiatry. 2014;22(6):338-341.
9. Zheng H, Bergamino M, Ford BN, et al; Tulsa 1000 Investigators. Replicable association between human cytomegalovirus infection and reduced white matter fractional anisotropy in major depressive disorder. Neuropsychopharmacology. 2021;46(5):928-938.
10. Sagarwala R, Nasrallah HA. A systematic review of diffusion tensor imaging studies in drug-naïve OCD patients before and after pharmacotherapy. Ann Clin Psychiatry. 2020;32(1):42-47.
11. Lee KS, Lee SH. White matter-based structural brain network of anxiety. Adv Exp Med Biol. 2020;1191:61-70.
12. Swanson MR, Hazlett HC. White matter as a monitoring biomarker for neurodevelopmental disorder intervention studies. J Neurodev Disord. 2019;11(1):33.
13. Hampton WH, Hanik IM, Olson IR. Substance abuse and white matter: findings, limitations, and future of diffusion tensor imaging research. Drug Alcohol Depend. 2019;197:288-298.
14. Waller R, Dotterer HL, Murray L, et al. White-matter tract abnormalities and antisocial behavior: a systematic review of diffusion tensor imaging studies across development. Neuroimage Clin. 2017;14:201-215.
15. Wolf RC, Pujara MS, Motzkin JC, et al. Interpersonal traits of psychopathy linked to reduced integrity of the uncinate fasciculus. Hum Brain Mapp. 2015;36(10):4202-4209.
16. Puzzo I, Seunarine K, Sully K, et al. Altered white-matter microstructure in conduct disorder is specifically associated with elevated callous-unemotional traits. J Abnorm Child Psychol. 2018;46(7):1451-1466.
17. Finger EC, Marsh A, Blair KS, et al. Impaired functional but preserved structural connectivity in limbic white matter tracts in youth with conduct disorder or oppositional defiant disorder plus psychopathic traits. Psychiatry Res. 2012;202(3):239-244.
18. Li C, Dong M, Womer FY, et al. Transdiagnostic time-varying dysconnectivity across major psychiatric disorders. Hum Brain Mapp. 2021;42(4):1182-1196.
19. Khanbabaei M, Hughes E, Ellegood J, et al. Precocious myelination in a mouse model of autism. Transl Psychiatry. 2019;9(1):251.
20. Petratos S, Theotokis P, Kim MJ, et al. That’s a wrap! Molecular drivers governing neuronal nogo receptor-dependent myelin plasticity and integrity. Front Cell Neurosci. 2020;14:227
21. Fernandez-Enright F, Andrews JL, Newell KA, et al. Novel implications of Lingo-1 and its signaling partners in schizophrenia. Transl Psychiatry. 2014;4(1):e348. doi: 10.1038/tp.2013.121
22. Bartzokis G, Lu PH, Stewart SB, et al. In vivo evidence of differential impact of typical and atypical antipsychotics on intracortical myelin in adults with schizophrenia. Schizophr Res. 2009;113(2-3):322-331.
23. Bartzokis G, Lu PH, Amar CP, et al. Long acting injection versus oral risperidone in first-episode schizophrenia: differential impact on white matter myelination trajectory. Schizophr Res. 2011 Oct;132(1):35-41
24. Tishler TA, Bartzokis G, Lu PH, et al. Abnormal trajectory of intracortical myelination in schizophrenia implicates white matter in disease pathophysiology and the therapeutic mechanism of action of antipsychotics. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3(5):454-462.
25. Ren Y, Wang H, Xiao L. Improving myelin/oligodendrocyte-related dysfunction: a new mechanism of antipsychotics in the treatment of schizophrenia? Int J Neuropsychopharmacol. 2013;16(3):691-700.
26. Dietz AG, Goldman SA, Nedergaard M. Glial cells in schizophrenia: a unified hypothesis. Lancet Psychiatry. 2020;7(3):272-281.
27. Chen AT, Nasrallah HA. Neuroprotective effects of the second generation antipsychotics. Schizophr Res. 2019;208:1-7
28. Sagarwala R, Nasrallah HA. (In press.) The effect of antipsychotic medications on white matter integrity in first-episode drug naïve patients with psychosis. Asian Journal of Psychiatry.
29. Nasrallah HA. Let’s tear down the silos and reunify psychiatry and neurology. Current Psychiatry. 2013;12(8):9-10.
How to help vaccinated patients navigate FOGO (fear of going out)
Remember FOMO (fear of missing out)? The pandemic cured most of us of that! In its place, many are suffering from a new syndrome that has been coined “FOGO” (fear of going out). As the COVID-19 vaccines roll out, restrictions lessen, and cases decline, we face new challenges. The pandemic showed us that “we are all in it together.” Now our patients, family, friends – and even we, ourselves – may face similar anxieties as we transition back.
Our brains love routines. They save energy as we transverse the same pathway with ease. We created new patterns in the first 30 days of quarantine, and we spent more than a year engraining them. Many people are feeling even more anxiety as restrictions are lifting and expectations are rising. Those with preexisting anxiety disorders may have an even more difficult time resuming routine activities.
Since the virus is still among us, we need to maintain caution, so some degree of FOGO is wise. But when we limit our activities too much, we create a whole new host of issues. The pandemic gave us all a taste of the agoraphobic lifestyle. It is difficult to know where exactly to draw the line right now between healthy anxiety and anxiety that becomes the disease for ourselves, our families and friends – and our patients.
Recommendations for FOGO
- Talk to your families, friends, and patients about what activities you recommend, which they might resume and which they should continue to avoid. People should make plans to optimize their physical and mental health while continuing to protect themselves from COVID-19. If anxiety is becoming the main problem, psychotherapy or medication may be necessary to treat their symptoms.
- Continue to encourage those with FOGO to practice techniques to be calm. Suggest that they take deep breaths with long exhales. This breathing pattern activates the parasympathetic nervous system and will help them feel calmer. We have all been under chronic stress, and our sympathetic nervous system has been in overdrive. We need to be calm to make the best decisions so our frontal lobe can be in charge rather than our primitive, fear-based brain that has been running the show for more a year. Encourage calming activities, such as yoga, meditation, warm baths, spending time in nature, hugging a pet, and more.
- Advise sufferers to start slowly. They should resume activities where they feel the safest. Walking outside with a friend is a good way to start. We now know that transmission is remarkably low or nonexistent if both parties are vaccinated. Exercise is a great way to combat many psychological issues, including FOGO.
- FOGO sufferers should build confidence gradually. Recommend taking one day at a time and trying to find ways to enjoy new ventures out. Soon, our brains will adapt to the new routines and the days of COVID-19 will recede from our thoughts.
- Respect whatever feelings emerge. The closer we and our patients were to trauma, the more challenging it may be to recover. If you or your patients suffered from COVID-19 or had a close family member or friend who did, be prepared to reemerge more slowly. Don’t feel pressured by what others are doing. Go at your own pace. Only you can decide what is the right way to move forward in these times.
- Look for signs of substance overuse or misuse. FOGO sufferers may turn to drugs or alcohol to mask their anxiety. This is a common pothole and should be avoided. Be alert for this problem and discuss it with patients, friends, or family members who may be making unhealthy choices.
Time is a great healer, and remind others that “this too shall pass.” FOGO will give rise to another yet-to-be named syndrome. We seem to be moving in a very positive direction at a remarkable pace. As Alexander Pope so wisely wrote, “Hope springs eternal.” Better times are ahead.
Dr. Ritvo, who has almost 30 years’ experience in psychiatry, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa. Momosa Publishing, 2018). Dr. Ritvo has no disclosures.
Remember FOMO (fear of missing out)? The pandemic cured most of us of that! In its place, many are suffering from a new syndrome that has been coined “FOGO” (fear of going out). As the COVID-19 vaccines roll out, restrictions lessen, and cases decline, we face new challenges. The pandemic showed us that “we are all in it together.” Now our patients, family, friends – and even we, ourselves – may face similar anxieties as we transition back.
Our brains love routines. They save energy as we transverse the same pathway with ease. We created new patterns in the first 30 days of quarantine, and we spent more than a year engraining them. Many people are feeling even more anxiety as restrictions are lifting and expectations are rising. Those with preexisting anxiety disorders may have an even more difficult time resuming routine activities.
Since the virus is still among us, we need to maintain caution, so some degree of FOGO is wise. But when we limit our activities too much, we create a whole new host of issues. The pandemic gave us all a taste of the agoraphobic lifestyle. It is difficult to know where exactly to draw the line right now between healthy anxiety and anxiety that becomes the disease for ourselves, our families and friends – and our patients.
Recommendations for FOGO
- Talk to your families, friends, and patients about what activities you recommend, which they might resume and which they should continue to avoid. People should make plans to optimize their physical and mental health while continuing to protect themselves from COVID-19. If anxiety is becoming the main problem, psychotherapy or medication may be necessary to treat their symptoms.
- Continue to encourage those with FOGO to practice techniques to be calm. Suggest that they take deep breaths with long exhales. This breathing pattern activates the parasympathetic nervous system and will help them feel calmer. We have all been under chronic stress, and our sympathetic nervous system has been in overdrive. We need to be calm to make the best decisions so our frontal lobe can be in charge rather than our primitive, fear-based brain that has been running the show for more a year. Encourage calming activities, such as yoga, meditation, warm baths, spending time in nature, hugging a pet, and more.
- Advise sufferers to start slowly. They should resume activities where they feel the safest. Walking outside with a friend is a good way to start. We now know that transmission is remarkably low or nonexistent if both parties are vaccinated. Exercise is a great way to combat many psychological issues, including FOGO.
- FOGO sufferers should build confidence gradually. Recommend taking one day at a time and trying to find ways to enjoy new ventures out. Soon, our brains will adapt to the new routines and the days of COVID-19 will recede from our thoughts.
- Respect whatever feelings emerge. The closer we and our patients were to trauma, the more challenging it may be to recover. If you or your patients suffered from COVID-19 or had a close family member or friend who did, be prepared to reemerge more slowly. Don’t feel pressured by what others are doing. Go at your own pace. Only you can decide what is the right way to move forward in these times.
- Look for signs of substance overuse or misuse. FOGO sufferers may turn to drugs or alcohol to mask their anxiety. This is a common pothole and should be avoided. Be alert for this problem and discuss it with patients, friends, or family members who may be making unhealthy choices.
Time is a great healer, and remind others that “this too shall pass.” FOGO will give rise to another yet-to-be named syndrome. We seem to be moving in a very positive direction at a remarkable pace. As Alexander Pope so wisely wrote, “Hope springs eternal.” Better times are ahead.
Dr. Ritvo, who has almost 30 years’ experience in psychiatry, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa. Momosa Publishing, 2018). Dr. Ritvo has no disclosures.
Remember FOMO (fear of missing out)? The pandemic cured most of us of that! In its place, many are suffering from a new syndrome that has been coined “FOGO” (fear of going out). As the COVID-19 vaccines roll out, restrictions lessen, and cases decline, we face new challenges. The pandemic showed us that “we are all in it together.” Now our patients, family, friends – and even we, ourselves – may face similar anxieties as we transition back.
Our brains love routines. They save energy as we transverse the same pathway with ease. We created new patterns in the first 30 days of quarantine, and we spent more than a year engraining them. Many people are feeling even more anxiety as restrictions are lifting and expectations are rising. Those with preexisting anxiety disorders may have an even more difficult time resuming routine activities.
Since the virus is still among us, we need to maintain caution, so some degree of FOGO is wise. But when we limit our activities too much, we create a whole new host of issues. The pandemic gave us all a taste of the agoraphobic lifestyle. It is difficult to know where exactly to draw the line right now between healthy anxiety and anxiety that becomes the disease for ourselves, our families and friends – and our patients.
Recommendations for FOGO
- Talk to your families, friends, and patients about what activities you recommend, which they might resume and which they should continue to avoid. People should make plans to optimize their physical and mental health while continuing to protect themselves from COVID-19. If anxiety is becoming the main problem, psychotherapy or medication may be necessary to treat their symptoms.
- Continue to encourage those with FOGO to practice techniques to be calm. Suggest that they take deep breaths with long exhales. This breathing pattern activates the parasympathetic nervous system and will help them feel calmer. We have all been under chronic stress, and our sympathetic nervous system has been in overdrive. We need to be calm to make the best decisions so our frontal lobe can be in charge rather than our primitive, fear-based brain that has been running the show for more a year. Encourage calming activities, such as yoga, meditation, warm baths, spending time in nature, hugging a pet, and more.
- Advise sufferers to start slowly. They should resume activities where they feel the safest. Walking outside with a friend is a good way to start. We now know that transmission is remarkably low or nonexistent if both parties are vaccinated. Exercise is a great way to combat many psychological issues, including FOGO.
- FOGO sufferers should build confidence gradually. Recommend taking one day at a time and trying to find ways to enjoy new ventures out. Soon, our brains will adapt to the new routines and the days of COVID-19 will recede from our thoughts.
- Respect whatever feelings emerge. The closer we and our patients were to trauma, the more challenging it may be to recover. If you or your patients suffered from COVID-19 or had a close family member or friend who did, be prepared to reemerge more slowly. Don’t feel pressured by what others are doing. Go at your own pace. Only you can decide what is the right way to move forward in these times.
- Look for signs of substance overuse or misuse. FOGO sufferers may turn to drugs or alcohol to mask their anxiety. This is a common pothole and should be avoided. Be alert for this problem and discuss it with patients, friends, or family members who may be making unhealthy choices.
Time is a great healer, and remind others that “this too shall pass.” FOGO will give rise to another yet-to-be named syndrome. We seem to be moving in a very positive direction at a remarkable pace. As Alexander Pope so wisely wrote, “Hope springs eternal.” Better times are ahead.
Dr. Ritvo, who has almost 30 years’ experience in psychiatry, practices in Miami Beach, Fla. She is the author of “Bekindr – The Transformative Power of Kindness” (Hellertown, Pa. Momosa Publishing, 2018). Dr. Ritvo has no disclosures.
Novel drug approvals of 2020
In 2020, the Food and Drug Administration approved 53 new drugs for humans. One of these agents, Annovera (segesterone and ethinyl estradiol), is a vaginal ring to prevent pregnancy and is not relevant in this article. A second drug, Asparlas (calaspargase pegol), indicated to treat acute lymphoblastic leukemia, has not yet been released by its manufacturer. Orgovyx (relugolix) is used for prostate cancer and Lampit (nifurtimox) is drug used in children – neither of these two agents will be covered. The remaining 49 are covered below. The agents with molecular weights less than 1,000 probably cross the placenta in the first half of pregnancy, but nearly all, regardless of MW, will cross in the second half of pregnancy.
No human pregnancy data for these agents has been found, but there are five drugs included in pregnancy registries. It will take some time before the outcomes of these drugs are published. The routine absence of pregnancy data for most drugs was pointed out in an article that I coauthored, “Should pregnant women be included in phase 4 clinical drug trials?”. The article makes a strong argument for including some pregnant women in these trials.
Anti-infectives
Artesunate (384)
The drug appears low risk when used in the second and third trimesters. There is inadequate information regarding its use in the first trimester, so the safest course for the embryo appears to be avoiding its use during this period. A single intravenous dose given to rats early in gestation resulted in embryolethality.
Ebanga (ansuvimab) (147,000)
Studies on its use in pregnant animals have not been conducted.
Inmazeb (atoltivimab, maftivimab, odesivimab) (144,000-146,000)
Inmazeb is a combination of the three agents. Studies on its use in pregnant animals have not been conducted.
Veklury (remdesivir) (603)
Veklury is indicated for the treatment of pregnant women hospitalized with COVID-19 who are at risk for serious morbidity and mortality. The drug should be used during pregnancy only if the potential benefit justifies the potential risk for the mother and the fetus.
Antineoplastics
Ayvakit (avapritinib) (499)
The drug may cause fetal harm. The drug was teratogenic in animals.
Blenrep (belantamab mafodotin-blmf) (152,000)
A B-cell maturation antigen, it is indicated for the treatment of multiple myeloma. No human or animal pregnancy data have been located.
Danyelza (naxitamab-gqgk) (144,000)
This agent is used for the treatment of neuroblastoma. Based on its mechanism of action it may cause fetal harm if used in pregnancy.
Gavreto (pralsetinib) (534)
Gavreto is indicated for the treatment of small cell lung cancer. It may cause embryo-fetal harm if used in pregnancy.
Inqovi (cedazuridine + decitabine) (268,228)
The drug combination can cause fetal harm in human pregnancy. It is toxic in pregnant animals.
Margenza (margetuximab-cmkb) (149,000)
Although there are no data on the use of this drug in human pregnancy, the findings in animals and mechanism of action suggest that it will cause fetal harm.
Monjuvi (tafasitamab-cxix) (150,000)
This drug is a cytolytic antibody that is indicated in combination with lenalidomide. The combination may cause fetal harm.
Pemazyre (pemigatinib) (488)
It is indicated for the treatment of cholangiocarcinoma. In an animal study, the drug caused fetal defects, fetal growth retardation, and embryo-fetal death at maternal exposures lower than the human exposure.
Qinlock (ripretinib) (510)
This drug is used for the treatment of patients with advanced gastrointestinal stromal tumor. The drug was teratogenic in pregnant animals.
Retevmo (selpercatinib) (526)
This is a kinase inhibitor used for the treatment of small cell lung cancer. The drug is teratogenic in animals.
Sarclisa (isatuximab-irfc) (148,000)This drug is used in combination with pomalidomide and dexamethasone. The combination would probably cause major toxicity in an embryo or fetus.
Tabrecta (capmatinib) (412 – free base)Capmatinib is a kinase inhibitor used for the treatment of metastatic non–small cell lung cancer. It is teratogenic in animals.
Tazverik (tazemetostat) (654)Tazemetostat is indicated for the treatment of epithelioid sarcoma and follicular lymphoma, The drug is teratogenic in animals.
Trodelvy (sacituzumab govitecan-hziy) (1,602)This agent is used for the treatment of breast cancer. The drug has not been tested in pregnant animals. However, according to the manufacturer, there is a high possibility of human teratogenicity if it is given to a pregnant woman.
Tukysa (tucatinib) (481)
Tukysa is a tyrosine kinase inhibitor that is used in combination with trastuzumab and capecitabine for the treatment of breast cancer. The drug is teratogenic in animals.
Zeposia (ozanimod) (441)
Zeposia is indicated for the treatment of multiple sclerosis. The drug takes about 3 months to eliminate from the body. The drug is teratogenic in animals.
Zepzelca (lurbinectedin) (785)
This agent is used for the treatment of metastatic small cell lung cancer. The drug is teratogenic in animals.
Antiemetics
Barhemsys (amisulpride) (369)
This agent is Indicated to prevent nausea and vomiting. Animal data suggest low risk of embryo/fetal birth defects.
Antimigraine
Nurtec (rimegepant) (611)
Nurtec is indicated for acute treatment of migraine. Development toxicity was not observed in animals given doses similar to those used in humans.
Vyepti (eptinezumab-jjmr) (143,000)
A humanized monoclonal antibody that is given every 3 months to prevent migraine. There was no embryo-fetal harm in animals given the drug.
CNS
Byfavo (remimazolam) (493 – free base)
This drug is indicated for procedural sedation in adults undergoing procedures lasting 30 minutes or less. No defects were observed in animals.
Diagnostics
Cerianna (fluoroestradiol F 18) (289)
It is indicated for use with PET for characterization of estrogen receptor status in patients with ER-positive breast cancer. It has the potential to cause fetal harm depending on the fetal stage of development and the magnitude of radiation dose. There are no data on its use in pregnant women or animals.
Detectnet (copper CU-64 dotatate) (1,497)
All radiopharmaceuticals have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of the radiation dose. There are no pregnancy data in humans or animals
Miscellaneous
Dojolvi (triheptanoin) (429)
This agent is indicated as a source of calories and fatty acids for the treatment of pediatric and adult patients with molecularly confirmed long-chain fatty acid oxidation disorders. Advise patients that there is a pregnancy safety study that collects pregnancy outcome data in women taking Dojolvi during pregnancy. Pregnant patients can enroll in the study by calling 1-888-756-8657.
Enspryng (satralizumab-mwge) (143,000)
It is indicated for the treatment of neuromyelitis optica spectrum disorder in adult patients who are anti–aquaporin-4 (AQP4) antibody positive. No information is available on the risks, if any, in pregnancy. No adverse effects on maternal or fetal development were observed in pregnant monkeys and their offspring.
Evrysdi (risdiplam) (401)
This is a prescription medicine used to treat spinal muscular atrophy in adults and children aged 2 months and older. In pregnant animals the drug caused adverse effects on fetal development.
Gemtesa (vibegron) (445)
Gemtesa is used in adults to treat the symptoms of overactive bladder. The drug had no adverse effects on pregnant animals.
Imcivree (setmelanotide) (1,117)
This drug is indicated for chronic weight management in adult and pediatric patients aged 6 years and older with obesity because of proopiomelanocortin, proprotein convertase subtilisin/kexin type 1, or leptin receptor deficiency. The drug was not embryo toxic in animals.
Isturisa (osilodrostat) (325)
Isturisa is a cortisol synthesis inhibitor indicated for the treatment of adult patients with Cushing’s disease. No adverse fetal effects were observed in pregnant animals.
Klisyri (tirbanibulin) (431)
Tirbanibulin ointment is a microtubule inhibitor that is used to treat actinic keratosis. Information on its effects in pregnancy is not available.
Koselugo (selumetinib) (556)
This is a kinase inhibitor indicated for the treatment of pediatric patients aged 2 years and older. The drug is toxic in pregnant animals but its effects in human pregnancy are not known.
Nexletol (bempedoic acid) (344)
Nexletol is indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or established atherosclerotic cardiovascular disease who require additional lowering of LDL cholesterol. The drug was not teratogenic in animals. Discontinue Nexletol when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus.
Olinvyk (oliceridine) (503)
Olinvyk injection is indicated in adults for the management of acute pain severe enough to require an intravenous opioid analgesic. Prolonged use of Olinvyk during pregnancy can result in neonatal opioid withdrawal syndrome. The drug was not teratogenic in animals.
Ongentys (opicapone) (413)
Ongentys is indicated as adjunctive treatment to levodopa/carbidopa in patients with Parkinson’s disease experiencing “off” episodes. The drug was teratogenic in rabbits but not in rats.
Orladeyo (berotralstat) (635)
This drug is a plasma kallikrein inhibitor indicated for prophylaxis to prevent attacks of hereditary angioedema. It was not teratogenic in animals.
Oxlumo (lumasiran) (17,286)
Oxlumo is a HAO1-directed small interfering ribonucleic acid indicated for the treatment of primary hyperoxaluria type 1 to lower urinary oxalate levels. No adverse effects on pregnancy or embryo-fetal development related to the drug were observed in animals.
Pizensy (lactitol) (344)
Lactitol is minimally absorbed systemically following oral administration. It is unknown whether maternal use will result in fetal exposure to the drug. No effects on embryo-fetal development were observed in animals at doses much higher than the maximum recommended human dosage.
Rukobia (fostemsavir) (705; 584 for free acid)
This drug is an HIV-1–directed attachment inhibitor, in combination with other antiretrovirals. There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to the drug during pregnancy. Health care providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry at 1-800-258-4263.
Sogroya (somapacitan-beco) (23,305)
This is a human growth hormone analog indicated for replacement of endogenous growth hormone in adults with growth hormone deficiency. The drug was not teratogenic in animals.
Tepezza (teprotumumab-trbw) (148,000)
Drug is indicated for the treatment of thyroid eye disease. The drug was teratogenic in cynomolgus monkeys. The manufacturer states that because of the risk, the drug should not be used in pregnancy.
Tauvid (flortaucipir F-18) (262)
This drug is indicated for use with PET imaging of the brain to evaluate for Alzheimer’s disease. It is a radioactive drug and should not be used in pregnant women.
Uplizna (inebilizumab-cdon) (149,000)
Uplizna is indicated for the treatment of neuromyelitis optica spectrum disorder in adult patients who are anti-AQP4 antibody positive. It is a humanized IgG1 monoclonal antibody and immunoglobulins are known to cross the placental barrier. Based on animal data, the drug can cause fetal harm because of B-cell lymphopenia and reduce antibody response in offspring exposed to the drug. Women of childbearing potential should use contraception while receiving Uplizna and for 6 months after the last dose.
Winlevi (clascoterone) (403)
This cream is an androgen receptor inhibitor that is indicated for the topical treatment of acne vulgaris in patients aged 12 years and older. Subcutaneous use in animals was associated with fetal defects.
Xeglyze (abametapir) (1,840)
Xeglyze is indicated for the topical treatment of head lice infestation in patients aged 6 months and older. The drug was not teratogenic in animals.
Zokinvy (lonafarnib) (639)
Zokinvy is indicated in patients 12 months or older to reduce the risk of mortality in several conditions. Animal studies have found embryo-fetal harm.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at obnews@mdedge.com.
In 2020, the Food and Drug Administration approved 53 new drugs for humans. One of these agents, Annovera (segesterone and ethinyl estradiol), is a vaginal ring to prevent pregnancy and is not relevant in this article. A second drug, Asparlas (calaspargase pegol), indicated to treat acute lymphoblastic leukemia, has not yet been released by its manufacturer. Orgovyx (relugolix) is used for prostate cancer and Lampit (nifurtimox) is drug used in children – neither of these two agents will be covered. The remaining 49 are covered below. The agents with molecular weights less than 1,000 probably cross the placenta in the first half of pregnancy, but nearly all, regardless of MW, will cross in the second half of pregnancy.
No human pregnancy data for these agents has been found, but there are five drugs included in pregnancy registries. It will take some time before the outcomes of these drugs are published. The routine absence of pregnancy data for most drugs was pointed out in an article that I coauthored, “Should pregnant women be included in phase 4 clinical drug trials?”. The article makes a strong argument for including some pregnant women in these trials.
Anti-infectives
Artesunate (384)
The drug appears low risk when used in the second and third trimesters. There is inadequate information regarding its use in the first trimester, so the safest course for the embryo appears to be avoiding its use during this period. A single intravenous dose given to rats early in gestation resulted in embryolethality.
Ebanga (ansuvimab) (147,000)
Studies on its use in pregnant animals have not been conducted.
Inmazeb (atoltivimab, maftivimab, odesivimab) (144,000-146,000)
Inmazeb is a combination of the three agents. Studies on its use in pregnant animals have not been conducted.
Veklury (remdesivir) (603)
Veklury is indicated for the treatment of pregnant women hospitalized with COVID-19 who are at risk for serious morbidity and mortality. The drug should be used during pregnancy only if the potential benefit justifies the potential risk for the mother and the fetus.
Antineoplastics
Ayvakit (avapritinib) (499)
The drug may cause fetal harm. The drug was teratogenic in animals.
Blenrep (belantamab mafodotin-blmf) (152,000)
A B-cell maturation antigen, it is indicated for the treatment of multiple myeloma. No human or animal pregnancy data have been located.
Danyelza (naxitamab-gqgk) (144,000)
This agent is used for the treatment of neuroblastoma. Based on its mechanism of action it may cause fetal harm if used in pregnancy.
Gavreto (pralsetinib) (534)
Gavreto is indicated for the treatment of small cell lung cancer. It may cause embryo-fetal harm if used in pregnancy.
Inqovi (cedazuridine + decitabine) (268,228)
The drug combination can cause fetal harm in human pregnancy. It is toxic in pregnant animals.
Margenza (margetuximab-cmkb) (149,000)
Although there are no data on the use of this drug in human pregnancy, the findings in animals and mechanism of action suggest that it will cause fetal harm.
Monjuvi (tafasitamab-cxix) (150,000)
This drug is a cytolytic antibody that is indicated in combination with lenalidomide. The combination may cause fetal harm.
Pemazyre (pemigatinib) (488)
It is indicated for the treatment of cholangiocarcinoma. In an animal study, the drug caused fetal defects, fetal growth retardation, and embryo-fetal death at maternal exposures lower than the human exposure.
Qinlock (ripretinib) (510)
This drug is used for the treatment of patients with advanced gastrointestinal stromal tumor. The drug was teratogenic in pregnant animals.
Retevmo (selpercatinib) (526)
This is a kinase inhibitor used for the treatment of small cell lung cancer. The drug is teratogenic in animals.
Sarclisa (isatuximab-irfc) (148,000)This drug is used in combination with pomalidomide and dexamethasone. The combination would probably cause major toxicity in an embryo or fetus.
Tabrecta (capmatinib) (412 – free base)Capmatinib is a kinase inhibitor used for the treatment of metastatic non–small cell lung cancer. It is teratogenic in animals.
Tazverik (tazemetostat) (654)Tazemetostat is indicated for the treatment of epithelioid sarcoma and follicular lymphoma, The drug is teratogenic in animals.
Trodelvy (sacituzumab govitecan-hziy) (1,602)This agent is used for the treatment of breast cancer. The drug has not been tested in pregnant animals. However, according to the manufacturer, there is a high possibility of human teratogenicity if it is given to a pregnant woman.
Tukysa (tucatinib) (481)
Tukysa is a tyrosine kinase inhibitor that is used in combination with trastuzumab and capecitabine for the treatment of breast cancer. The drug is teratogenic in animals.
Zeposia (ozanimod) (441)
Zeposia is indicated for the treatment of multiple sclerosis. The drug takes about 3 months to eliminate from the body. The drug is teratogenic in animals.
Zepzelca (lurbinectedin) (785)
This agent is used for the treatment of metastatic small cell lung cancer. The drug is teratogenic in animals.
Antiemetics
Barhemsys (amisulpride) (369)
This agent is Indicated to prevent nausea and vomiting. Animal data suggest low risk of embryo/fetal birth defects.
Antimigraine
Nurtec (rimegepant) (611)
Nurtec is indicated for acute treatment of migraine. Development toxicity was not observed in animals given doses similar to those used in humans.
Vyepti (eptinezumab-jjmr) (143,000)
A humanized monoclonal antibody that is given every 3 months to prevent migraine. There was no embryo-fetal harm in animals given the drug.
CNS
Byfavo (remimazolam) (493 – free base)
This drug is indicated for procedural sedation in adults undergoing procedures lasting 30 minutes or less. No defects were observed in animals.
Diagnostics
Cerianna (fluoroestradiol F 18) (289)
It is indicated for use with PET for characterization of estrogen receptor status in patients with ER-positive breast cancer. It has the potential to cause fetal harm depending on the fetal stage of development and the magnitude of radiation dose. There are no data on its use in pregnant women or animals.
Detectnet (copper CU-64 dotatate) (1,497)
All radiopharmaceuticals have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of the radiation dose. There are no pregnancy data in humans or animals
Miscellaneous
Dojolvi (triheptanoin) (429)
This agent is indicated as a source of calories and fatty acids for the treatment of pediatric and adult patients with molecularly confirmed long-chain fatty acid oxidation disorders. Advise patients that there is a pregnancy safety study that collects pregnancy outcome data in women taking Dojolvi during pregnancy. Pregnant patients can enroll in the study by calling 1-888-756-8657.
Enspryng (satralizumab-mwge) (143,000)
It is indicated for the treatment of neuromyelitis optica spectrum disorder in adult patients who are anti–aquaporin-4 (AQP4) antibody positive. No information is available on the risks, if any, in pregnancy. No adverse effects on maternal or fetal development were observed in pregnant monkeys and their offspring.
Evrysdi (risdiplam) (401)
This is a prescription medicine used to treat spinal muscular atrophy in adults and children aged 2 months and older. In pregnant animals the drug caused adverse effects on fetal development.
Gemtesa (vibegron) (445)
Gemtesa is used in adults to treat the symptoms of overactive bladder. The drug had no adverse effects on pregnant animals.
Imcivree (setmelanotide) (1,117)
This drug is indicated for chronic weight management in adult and pediatric patients aged 6 years and older with obesity because of proopiomelanocortin, proprotein convertase subtilisin/kexin type 1, or leptin receptor deficiency. The drug was not embryo toxic in animals.
Isturisa (osilodrostat) (325)
Isturisa is a cortisol synthesis inhibitor indicated for the treatment of adult patients with Cushing’s disease. No adverse fetal effects were observed in pregnant animals.
Klisyri (tirbanibulin) (431)
Tirbanibulin ointment is a microtubule inhibitor that is used to treat actinic keratosis. Information on its effects in pregnancy is not available.
Koselugo (selumetinib) (556)
This is a kinase inhibitor indicated for the treatment of pediatric patients aged 2 years and older. The drug is toxic in pregnant animals but its effects in human pregnancy are not known.
Nexletol (bempedoic acid) (344)
Nexletol is indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or established atherosclerotic cardiovascular disease who require additional lowering of LDL cholesterol. The drug was not teratogenic in animals. Discontinue Nexletol when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus.
Olinvyk (oliceridine) (503)
Olinvyk injection is indicated in adults for the management of acute pain severe enough to require an intravenous opioid analgesic. Prolonged use of Olinvyk during pregnancy can result in neonatal opioid withdrawal syndrome. The drug was not teratogenic in animals.
Ongentys (opicapone) (413)
Ongentys is indicated as adjunctive treatment to levodopa/carbidopa in patients with Parkinson’s disease experiencing “off” episodes. The drug was teratogenic in rabbits but not in rats.
Orladeyo (berotralstat) (635)
This drug is a plasma kallikrein inhibitor indicated for prophylaxis to prevent attacks of hereditary angioedema. It was not teratogenic in animals.
Oxlumo (lumasiran) (17,286)
Oxlumo is a HAO1-directed small interfering ribonucleic acid indicated for the treatment of primary hyperoxaluria type 1 to lower urinary oxalate levels. No adverse effects on pregnancy or embryo-fetal development related to the drug were observed in animals.
Pizensy (lactitol) (344)
Lactitol is minimally absorbed systemically following oral administration. It is unknown whether maternal use will result in fetal exposure to the drug. No effects on embryo-fetal development were observed in animals at doses much higher than the maximum recommended human dosage.
Rukobia (fostemsavir) (705; 584 for free acid)
This drug is an HIV-1–directed attachment inhibitor, in combination with other antiretrovirals. There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to the drug during pregnancy. Health care providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry at 1-800-258-4263.
Sogroya (somapacitan-beco) (23,305)
This is a human growth hormone analog indicated for replacement of endogenous growth hormone in adults with growth hormone deficiency. The drug was not teratogenic in animals.
Tepezza (teprotumumab-trbw) (148,000)
Drug is indicated for the treatment of thyroid eye disease. The drug was teratogenic in cynomolgus monkeys. The manufacturer states that because of the risk, the drug should not be used in pregnancy.
Tauvid (flortaucipir F-18) (262)
This drug is indicated for use with PET imaging of the brain to evaluate for Alzheimer’s disease. It is a radioactive drug and should not be used in pregnant women.
Uplizna (inebilizumab-cdon) (149,000)
Uplizna is indicated for the treatment of neuromyelitis optica spectrum disorder in adult patients who are anti-AQP4 antibody positive. It is a humanized IgG1 monoclonal antibody and immunoglobulins are known to cross the placental barrier. Based on animal data, the drug can cause fetal harm because of B-cell lymphopenia and reduce antibody response in offspring exposed to the drug. Women of childbearing potential should use contraception while receiving Uplizna and for 6 months after the last dose.
Winlevi (clascoterone) (403)
This cream is an androgen receptor inhibitor that is indicated for the topical treatment of acne vulgaris in patients aged 12 years and older. Subcutaneous use in animals was associated with fetal defects.
Xeglyze (abametapir) (1,840)
Xeglyze is indicated for the topical treatment of head lice infestation in patients aged 6 months and older. The drug was not teratogenic in animals.
Zokinvy (lonafarnib) (639)
Zokinvy is indicated in patients 12 months or older to reduce the risk of mortality in several conditions. Animal studies have found embryo-fetal harm.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at obnews@mdedge.com.
In 2020, the Food and Drug Administration approved 53 new drugs for humans. One of these agents, Annovera (segesterone and ethinyl estradiol), is a vaginal ring to prevent pregnancy and is not relevant in this article. A second drug, Asparlas (calaspargase pegol), indicated to treat acute lymphoblastic leukemia, has not yet been released by its manufacturer. Orgovyx (relugolix) is used for prostate cancer and Lampit (nifurtimox) is drug used in children – neither of these two agents will be covered. The remaining 49 are covered below. The agents with molecular weights less than 1,000 probably cross the placenta in the first half of pregnancy, but nearly all, regardless of MW, will cross in the second half of pregnancy.
No human pregnancy data for these agents has been found, but there are five drugs included in pregnancy registries. It will take some time before the outcomes of these drugs are published. The routine absence of pregnancy data for most drugs was pointed out in an article that I coauthored, “Should pregnant women be included in phase 4 clinical drug trials?”. The article makes a strong argument for including some pregnant women in these trials.
Anti-infectives
Artesunate (384)
The drug appears low risk when used in the second and third trimesters. There is inadequate information regarding its use in the first trimester, so the safest course for the embryo appears to be avoiding its use during this period. A single intravenous dose given to rats early in gestation resulted in embryolethality.
Ebanga (ansuvimab) (147,000)
Studies on its use in pregnant animals have not been conducted.
Inmazeb (atoltivimab, maftivimab, odesivimab) (144,000-146,000)
Inmazeb is a combination of the three agents. Studies on its use in pregnant animals have not been conducted.
Veklury (remdesivir) (603)
Veklury is indicated for the treatment of pregnant women hospitalized with COVID-19 who are at risk for serious morbidity and mortality. The drug should be used during pregnancy only if the potential benefit justifies the potential risk for the mother and the fetus.
Antineoplastics
Ayvakit (avapritinib) (499)
The drug may cause fetal harm. The drug was teratogenic in animals.
Blenrep (belantamab mafodotin-blmf) (152,000)
A B-cell maturation antigen, it is indicated for the treatment of multiple myeloma. No human or animal pregnancy data have been located.
Danyelza (naxitamab-gqgk) (144,000)
This agent is used for the treatment of neuroblastoma. Based on its mechanism of action it may cause fetal harm if used in pregnancy.
Gavreto (pralsetinib) (534)
Gavreto is indicated for the treatment of small cell lung cancer. It may cause embryo-fetal harm if used in pregnancy.
Inqovi (cedazuridine + decitabine) (268,228)
The drug combination can cause fetal harm in human pregnancy. It is toxic in pregnant animals.
Margenza (margetuximab-cmkb) (149,000)
Although there are no data on the use of this drug in human pregnancy, the findings in animals and mechanism of action suggest that it will cause fetal harm.
Monjuvi (tafasitamab-cxix) (150,000)
This drug is a cytolytic antibody that is indicated in combination with lenalidomide. The combination may cause fetal harm.
Pemazyre (pemigatinib) (488)
It is indicated for the treatment of cholangiocarcinoma. In an animal study, the drug caused fetal defects, fetal growth retardation, and embryo-fetal death at maternal exposures lower than the human exposure.
Qinlock (ripretinib) (510)
This drug is used for the treatment of patients with advanced gastrointestinal stromal tumor. The drug was teratogenic in pregnant animals.
Retevmo (selpercatinib) (526)
This is a kinase inhibitor used for the treatment of small cell lung cancer. The drug is teratogenic in animals.
Sarclisa (isatuximab-irfc) (148,000)This drug is used in combination with pomalidomide and dexamethasone. The combination would probably cause major toxicity in an embryo or fetus.
Tabrecta (capmatinib) (412 – free base)Capmatinib is a kinase inhibitor used for the treatment of metastatic non–small cell lung cancer. It is teratogenic in animals.
Tazverik (tazemetostat) (654)Tazemetostat is indicated for the treatment of epithelioid sarcoma and follicular lymphoma, The drug is teratogenic in animals.
Trodelvy (sacituzumab govitecan-hziy) (1,602)This agent is used for the treatment of breast cancer. The drug has not been tested in pregnant animals. However, according to the manufacturer, there is a high possibility of human teratogenicity if it is given to a pregnant woman.
Tukysa (tucatinib) (481)
Tukysa is a tyrosine kinase inhibitor that is used in combination with trastuzumab and capecitabine for the treatment of breast cancer. The drug is teratogenic in animals.
Zeposia (ozanimod) (441)
Zeposia is indicated for the treatment of multiple sclerosis. The drug takes about 3 months to eliminate from the body. The drug is teratogenic in animals.
Zepzelca (lurbinectedin) (785)
This agent is used for the treatment of metastatic small cell lung cancer. The drug is teratogenic in animals.
Antiemetics
Barhemsys (amisulpride) (369)
This agent is Indicated to prevent nausea and vomiting. Animal data suggest low risk of embryo/fetal birth defects.
Antimigraine
Nurtec (rimegepant) (611)
Nurtec is indicated for acute treatment of migraine. Development toxicity was not observed in animals given doses similar to those used in humans.
Vyepti (eptinezumab-jjmr) (143,000)
A humanized monoclonal antibody that is given every 3 months to prevent migraine. There was no embryo-fetal harm in animals given the drug.
CNS
Byfavo (remimazolam) (493 – free base)
This drug is indicated for procedural sedation in adults undergoing procedures lasting 30 minutes or less. No defects were observed in animals.
Diagnostics
Cerianna (fluoroestradiol F 18) (289)
It is indicated for use with PET for characterization of estrogen receptor status in patients with ER-positive breast cancer. It has the potential to cause fetal harm depending on the fetal stage of development and the magnitude of radiation dose. There are no data on its use in pregnant women or animals.
Detectnet (copper CU-64 dotatate) (1,497)
All radiopharmaceuticals have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of the radiation dose. There are no pregnancy data in humans or animals
Miscellaneous
Dojolvi (triheptanoin) (429)
This agent is indicated as a source of calories and fatty acids for the treatment of pediatric and adult patients with molecularly confirmed long-chain fatty acid oxidation disorders. Advise patients that there is a pregnancy safety study that collects pregnancy outcome data in women taking Dojolvi during pregnancy. Pregnant patients can enroll in the study by calling 1-888-756-8657.
Enspryng (satralizumab-mwge) (143,000)
It is indicated for the treatment of neuromyelitis optica spectrum disorder in adult patients who are anti–aquaporin-4 (AQP4) antibody positive. No information is available on the risks, if any, in pregnancy. No adverse effects on maternal or fetal development were observed in pregnant monkeys and their offspring.
Evrysdi (risdiplam) (401)
This is a prescription medicine used to treat spinal muscular atrophy in adults and children aged 2 months and older. In pregnant animals the drug caused adverse effects on fetal development.
Gemtesa (vibegron) (445)
Gemtesa is used in adults to treat the symptoms of overactive bladder. The drug had no adverse effects on pregnant animals.
Imcivree (setmelanotide) (1,117)
This drug is indicated for chronic weight management in adult and pediatric patients aged 6 years and older with obesity because of proopiomelanocortin, proprotein convertase subtilisin/kexin type 1, or leptin receptor deficiency. The drug was not embryo toxic in animals.
Isturisa (osilodrostat) (325)
Isturisa is a cortisol synthesis inhibitor indicated for the treatment of adult patients with Cushing’s disease. No adverse fetal effects were observed in pregnant animals.
Klisyri (tirbanibulin) (431)
Tirbanibulin ointment is a microtubule inhibitor that is used to treat actinic keratosis. Information on its effects in pregnancy is not available.
Koselugo (selumetinib) (556)
This is a kinase inhibitor indicated for the treatment of pediatric patients aged 2 years and older. The drug is toxic in pregnant animals but its effects in human pregnancy are not known.
Nexletol (bempedoic acid) (344)
Nexletol is indicated as an adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or established atherosclerotic cardiovascular disease who require additional lowering of LDL cholesterol. The drug was not teratogenic in animals. Discontinue Nexletol when pregnancy is recognized unless the benefits of therapy outweigh the potential risks to the fetus.
Olinvyk (oliceridine) (503)
Olinvyk injection is indicated in adults for the management of acute pain severe enough to require an intravenous opioid analgesic. Prolonged use of Olinvyk during pregnancy can result in neonatal opioid withdrawal syndrome. The drug was not teratogenic in animals.
Ongentys (opicapone) (413)
Ongentys is indicated as adjunctive treatment to levodopa/carbidopa in patients with Parkinson’s disease experiencing “off” episodes. The drug was teratogenic in rabbits but not in rats.
Orladeyo (berotralstat) (635)
This drug is a plasma kallikrein inhibitor indicated for prophylaxis to prevent attacks of hereditary angioedema. It was not teratogenic in animals.
Oxlumo (lumasiran) (17,286)
Oxlumo is a HAO1-directed small interfering ribonucleic acid indicated for the treatment of primary hyperoxaluria type 1 to lower urinary oxalate levels. No adverse effects on pregnancy or embryo-fetal development related to the drug were observed in animals.
Pizensy (lactitol) (344)
Lactitol is minimally absorbed systemically following oral administration. It is unknown whether maternal use will result in fetal exposure to the drug. No effects on embryo-fetal development were observed in animals at doses much higher than the maximum recommended human dosage.
Rukobia (fostemsavir) (705; 584 for free acid)
This drug is an HIV-1–directed attachment inhibitor, in combination with other antiretrovirals. There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to the drug during pregnancy. Health care providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry at 1-800-258-4263.
Sogroya (somapacitan-beco) (23,305)
This is a human growth hormone analog indicated for replacement of endogenous growth hormone in adults with growth hormone deficiency. The drug was not teratogenic in animals.
Tepezza (teprotumumab-trbw) (148,000)
Drug is indicated for the treatment of thyroid eye disease. The drug was teratogenic in cynomolgus monkeys. The manufacturer states that because of the risk, the drug should not be used in pregnancy.
Tauvid (flortaucipir F-18) (262)
This drug is indicated for use with PET imaging of the brain to evaluate for Alzheimer’s disease. It is a radioactive drug and should not be used in pregnant women.
Uplizna (inebilizumab-cdon) (149,000)
Uplizna is indicated for the treatment of neuromyelitis optica spectrum disorder in adult patients who are anti-AQP4 antibody positive. It is a humanized IgG1 monoclonal antibody and immunoglobulins are known to cross the placental barrier. Based on animal data, the drug can cause fetal harm because of B-cell lymphopenia and reduce antibody response in offspring exposed to the drug. Women of childbearing potential should use contraception while receiving Uplizna and for 6 months after the last dose.
Winlevi (clascoterone) (403)
This cream is an androgen receptor inhibitor that is indicated for the topical treatment of acne vulgaris in patients aged 12 years and older. Subcutaneous use in animals was associated with fetal defects.
Xeglyze (abametapir) (1,840)
Xeglyze is indicated for the topical treatment of head lice infestation in patients aged 6 months and older. The drug was not teratogenic in animals.
Zokinvy (lonafarnib) (639)
Zokinvy is indicated in patients 12 months or older to reduce the risk of mortality in several conditions. Animal studies have found embryo-fetal harm.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at obnews@mdedge.com.
Unhealthy drinking may worsen after weight loss surgery
Internal medicine primarily affords us the skill to cope with disorders of chronicity that rarely disappear. For every pneumococcal pneumonia we eradicate, we have multiple patients with HIV who will be treated indefinitely. Diabetes, once a lethal disease, is now a chronic condition for most patients, and even with treatment the trajectory is usually one of progression.
One gratifying exception in my professional lifetime has been the introduction of gastric surgeries that reduce morbidity and seem to extend the life span of those who successfully undergo these procedures. The Roux-en-Y gastric bypass and sleeve gastrectomy have kept thousands of patients in better health for many years, giving them a second chance. For a subset, however, this second chance comes with a stumbling block of substance use – most notably alcohol – that exceeds their preoperative use.
Increased alcohol use after surgery
A group affiliated with the Department of Veterans Affairs (VA) recently reviewed the large central database to identify changes in alcohol consumption among patients who had undergone successful bariatric surgery. The VA regularly administers the Alcohol Use Disorders Identification Test-Consumption (AUDIT-C), a survey validated as a reliable estimate of individual alcohol consumption. It is inserted into the VA electronic health record where it can be readily retrieved. By matching these survey results with individuals who underwent bariatric surgery at the VA and survived at least 8 years post op, the authors were able to follow trends in alcohol consumption, beginning 2 years before surgery through 8 years after.
Using the same database, the authors identified a larger number of nonoperative control patients with slightly less obesity but otherwise matched for several elements of comorbidity, such as hypertension, certain psychiatric disorders, and personal habits, including alcohol consumption.
Alcohol use was categorized as none, minor social use, and “unhealthy” use. Among those with no or minor social use preoperatively, 4% converted to unhealthy use at 3 years and about 5% at 8 years, significantly more than in the nonoperative control group. Those who had gastric bypass had somewhat more conversion than did those who had sleeve gastrectomy, though not significantly so.
Patients with an alcohol concern preoperatively took an interesting course. Consumption declined from 2 years pre op to the year of surgery, suggesting that curtailing its use may have been a surgical precondition. Postoperatively, they returned to unhealthy drinking levels. Those who underwent the sleeve gastrectomy consumed about the same amount of alcohol as did their matched nonoperative controls, but those who underwent bypass increased their baseline unhealthy use beyond that of the controls.
Because total abstinence is often the recommendation for treating alcoholism, the research group assessed how adherent the excessive drinkers were to abstinence. In anticipation of surgery, the rates of abstinence increased until the year of surgery, but by 3 years post op, consumption was often up to unhealthy levels, though no more than that of control participants with preexisting drinking problems.
Smoking and illicit drug use
Although increased alcohol consumption has generated the most studies, some attention has been given to smoking and illicit drug use, which may also increase over time.
One small study looked at composite tobacco, alcohol, and drug use pre- and postoperatively over 2 years, using population data. The authors found a parallel pattern of users voluntarily reducing their substance use in anticipation of surgery but relapsing as the procedure made them more functional and perhaps more independent. Of the substances people resumed, alcohol by far involved the largest increase in use from the preoperative baseline.
These studies, as important as they are, reveal what happened more effectively than they disclose why it happened. The latter requires some clinical experience. Curtailing cigarettes and alcohol use preoperatively may have been done to stay in the good graces of the surgeon. Many patients may have seen this as their path to a second chance that they intended to maintain.
The incentive to proceed to surgical weight loss, which incurs a measure of risk and forces changes in long ingrained eating habits, involves avoiding future morbidity and promoting longevity. Thus, the postoperative behaviors that threaten the long-term goal need to become a component of ongoing follow-up.
The acquisition of adverse behaviors not present preoperatively seems more difficult to sort out, and obligates those of us following these patients to ask about changes in alcohol use and provide resources for them should they need intervention.
Dr. Plotzker is a retired endocrinologist with 40 years of experience treating patients in both private practice and hospital settings.
A version of this article first appeared on Medscape.com.
Internal medicine primarily affords us the skill to cope with disorders of chronicity that rarely disappear. For every pneumococcal pneumonia we eradicate, we have multiple patients with HIV who will be treated indefinitely. Diabetes, once a lethal disease, is now a chronic condition for most patients, and even with treatment the trajectory is usually one of progression.
One gratifying exception in my professional lifetime has been the introduction of gastric surgeries that reduce morbidity and seem to extend the life span of those who successfully undergo these procedures. The Roux-en-Y gastric bypass and sleeve gastrectomy have kept thousands of patients in better health for many years, giving them a second chance. For a subset, however, this second chance comes with a stumbling block of substance use – most notably alcohol – that exceeds their preoperative use.
Increased alcohol use after surgery
A group affiliated with the Department of Veterans Affairs (VA) recently reviewed the large central database to identify changes in alcohol consumption among patients who had undergone successful bariatric surgery. The VA regularly administers the Alcohol Use Disorders Identification Test-Consumption (AUDIT-C), a survey validated as a reliable estimate of individual alcohol consumption. It is inserted into the VA electronic health record where it can be readily retrieved. By matching these survey results with individuals who underwent bariatric surgery at the VA and survived at least 8 years post op, the authors were able to follow trends in alcohol consumption, beginning 2 years before surgery through 8 years after.
Using the same database, the authors identified a larger number of nonoperative control patients with slightly less obesity but otherwise matched for several elements of comorbidity, such as hypertension, certain psychiatric disorders, and personal habits, including alcohol consumption.
Alcohol use was categorized as none, minor social use, and “unhealthy” use. Among those with no or minor social use preoperatively, 4% converted to unhealthy use at 3 years and about 5% at 8 years, significantly more than in the nonoperative control group. Those who had gastric bypass had somewhat more conversion than did those who had sleeve gastrectomy, though not significantly so.
Patients with an alcohol concern preoperatively took an interesting course. Consumption declined from 2 years pre op to the year of surgery, suggesting that curtailing its use may have been a surgical precondition. Postoperatively, they returned to unhealthy drinking levels. Those who underwent the sleeve gastrectomy consumed about the same amount of alcohol as did their matched nonoperative controls, but those who underwent bypass increased their baseline unhealthy use beyond that of the controls.
Because total abstinence is often the recommendation for treating alcoholism, the research group assessed how adherent the excessive drinkers were to abstinence. In anticipation of surgery, the rates of abstinence increased until the year of surgery, but by 3 years post op, consumption was often up to unhealthy levels, though no more than that of control participants with preexisting drinking problems.
Smoking and illicit drug use
Although increased alcohol consumption has generated the most studies, some attention has been given to smoking and illicit drug use, which may also increase over time.
One small study looked at composite tobacco, alcohol, and drug use pre- and postoperatively over 2 years, using population data. The authors found a parallel pattern of users voluntarily reducing their substance use in anticipation of surgery but relapsing as the procedure made them more functional and perhaps more independent. Of the substances people resumed, alcohol by far involved the largest increase in use from the preoperative baseline.
These studies, as important as they are, reveal what happened more effectively than they disclose why it happened. The latter requires some clinical experience. Curtailing cigarettes and alcohol use preoperatively may have been done to stay in the good graces of the surgeon. Many patients may have seen this as their path to a second chance that they intended to maintain.
The incentive to proceed to surgical weight loss, which incurs a measure of risk and forces changes in long ingrained eating habits, involves avoiding future morbidity and promoting longevity. Thus, the postoperative behaviors that threaten the long-term goal need to become a component of ongoing follow-up.
The acquisition of adverse behaviors not present preoperatively seems more difficult to sort out, and obligates those of us following these patients to ask about changes in alcohol use and provide resources for them should they need intervention.
Dr. Plotzker is a retired endocrinologist with 40 years of experience treating patients in both private practice and hospital settings.
A version of this article first appeared on Medscape.com.
Internal medicine primarily affords us the skill to cope with disorders of chronicity that rarely disappear. For every pneumococcal pneumonia we eradicate, we have multiple patients with HIV who will be treated indefinitely. Diabetes, once a lethal disease, is now a chronic condition for most patients, and even with treatment the trajectory is usually one of progression.
One gratifying exception in my professional lifetime has been the introduction of gastric surgeries that reduce morbidity and seem to extend the life span of those who successfully undergo these procedures. The Roux-en-Y gastric bypass and sleeve gastrectomy have kept thousands of patients in better health for many years, giving them a second chance. For a subset, however, this second chance comes with a stumbling block of substance use – most notably alcohol – that exceeds their preoperative use.
Increased alcohol use after surgery
A group affiliated with the Department of Veterans Affairs (VA) recently reviewed the large central database to identify changes in alcohol consumption among patients who had undergone successful bariatric surgery. The VA regularly administers the Alcohol Use Disorders Identification Test-Consumption (AUDIT-C), a survey validated as a reliable estimate of individual alcohol consumption. It is inserted into the VA electronic health record where it can be readily retrieved. By matching these survey results with individuals who underwent bariatric surgery at the VA and survived at least 8 years post op, the authors were able to follow trends in alcohol consumption, beginning 2 years before surgery through 8 years after.
Using the same database, the authors identified a larger number of nonoperative control patients with slightly less obesity but otherwise matched for several elements of comorbidity, such as hypertension, certain psychiatric disorders, and personal habits, including alcohol consumption.
Alcohol use was categorized as none, minor social use, and “unhealthy” use. Among those with no or minor social use preoperatively, 4% converted to unhealthy use at 3 years and about 5% at 8 years, significantly more than in the nonoperative control group. Those who had gastric bypass had somewhat more conversion than did those who had sleeve gastrectomy, though not significantly so.
Patients with an alcohol concern preoperatively took an interesting course. Consumption declined from 2 years pre op to the year of surgery, suggesting that curtailing its use may have been a surgical precondition. Postoperatively, they returned to unhealthy drinking levels. Those who underwent the sleeve gastrectomy consumed about the same amount of alcohol as did their matched nonoperative controls, but those who underwent bypass increased their baseline unhealthy use beyond that of the controls.
Because total abstinence is often the recommendation for treating alcoholism, the research group assessed how adherent the excessive drinkers were to abstinence. In anticipation of surgery, the rates of abstinence increased until the year of surgery, but by 3 years post op, consumption was often up to unhealthy levels, though no more than that of control participants with preexisting drinking problems.
Smoking and illicit drug use
Although increased alcohol consumption has generated the most studies, some attention has been given to smoking and illicit drug use, which may also increase over time.
One small study looked at composite tobacco, alcohol, and drug use pre- and postoperatively over 2 years, using population data. The authors found a parallel pattern of users voluntarily reducing their substance use in anticipation of surgery but relapsing as the procedure made them more functional and perhaps more independent. Of the substances people resumed, alcohol by far involved the largest increase in use from the preoperative baseline.
These studies, as important as they are, reveal what happened more effectively than they disclose why it happened. The latter requires some clinical experience. Curtailing cigarettes and alcohol use preoperatively may have been done to stay in the good graces of the surgeon. Many patients may have seen this as their path to a second chance that they intended to maintain.
The incentive to proceed to surgical weight loss, which incurs a measure of risk and forces changes in long ingrained eating habits, involves avoiding future morbidity and promoting longevity. Thus, the postoperative behaviors that threaten the long-term goal need to become a component of ongoing follow-up.
The acquisition of adverse behaviors not present preoperatively seems more difficult to sort out, and obligates those of us following these patients to ask about changes in alcohol use and provide resources for them should they need intervention.
Dr. Plotzker is a retired endocrinologist with 40 years of experience treating patients in both private practice and hospital settings.
A version of this article first appeared on Medscape.com.
New targeted treatments are major advances for HER2-positive breast cancer
Before 2001, HER2/neu-positive breast cancer (HER2+) was one of the most dreaded diagnoses a woman could face, as treatment was largely ineffective. The discovery of trastuzumab changed that dramatically.
Over the next 20 years, two additional HER2-targeted therapies – lapatinib and trastuzumab emtansine (TDM-1) – earned approval from the Food and Drug Administration for selected patients with early and late HER2+ breast cancer.
Since 2019, four additional HER2-targeted therapies have been approved by the FDA for HER2+ metastatic breast cancer (MBC), changing the treatment paradigm for those patients substantially.
The new agents are especially useful in certain patient populations. The agents offer the promise of improved survival for patients with recurrent metastatic disease and the potential for further reductions in relapse rates in earlier settings.
Trastuzumab deruxtecan
Trastuzumab deruxtecan is an antibody-drug conjugate that links three components: an anti-HER2 monoclonal antibody, a highly potent topoisomerase I inhibitor payload, and a tetrapeptide-based cleavable linker.
Trastuzumab deruxtecan has a high drug-to-antibody ratio. A membrane-permeable payload offers the potential for activity against adjacent HER2-negative cells in heterogeneous tumors. It has a long half-life (6 days).
Trastuzumab deruxtecan received accelerated approval from the FDA in December 2019 to treat patients with HER2+ MBC who have received two or more prior HER2-targeted regimens, based on the results of the DESTINY-Breast 01 trial.
DESTINY-Breast 01 trial
In the phase 2 DESTINY-Breast 01 trial, 184 patients with a median of six previous treatments received trastuzumab deruxtecan (5.4 mg/kg) intravenously every 21 days. There were 24 patients with treated, asymptomatic brain metastases who participated. Patients with untreated or symptomatic brain metastases were excluded.
Overall, a response to therapy was reported in 112 patients (60.9%), with 6.0% complete and 54.9% partial responses. Most of the patients for whom both baseline and postbaseline data were available had a reduction in tumor size.
The median time until response was 1.6 months, an interval that corresponded to the time until the first scheduled imaging. Three patients (1.6%) had progressive disease, and two patients (1.1%) could not be evaluated.
The median duration of follow-up was 11.1 months, and the median response duration was 14.8 months.
The median progression-free survival (PFS) was 16.4 months, and the median overall survival (OS) was not reached. The median PFS in the patients with brain involvement was 18.1 months.
The most common adverse events of grade 3 or higher were a decreased neutrophil count (20.7%), anemia (8.7%), and nausea (7.6%). Most concerning was that trastuzumab deruxtecan was associated with interstitial lung disease in 13.6% of patients.
Tucatinib
Tucatinib is an oral, highly selective HER2 tyrosine kinase inhibitor (TKI). In April 2020, it was approved by the FDA, in combination with trastuzumab and capecitabine, for adult patients with advanced unresectable or metastatic HER2+ breast cancer who have received one or more prior anti-HER2–based regimens for MBC. The approval included patients with brain metastases.
The recommended tucatinib dose is 300 mg orally twice a day in combination with trastuzumab (at the standard dose) and capecitabine (1,000 mg/m2 given orally twice daily on days 1-14) on a 21-day cycle, until disease progression or unacceptable toxicity.
HER2CLIMB trial
The study that led to the approval of tucatinib was the HER2CLIMB trial. The trial enrolled 612 HER2+ MBC patients who had prior treatment with trastuzumab, pertuzumab, and T-DM1. Patients had received a median of 4 (range, 2-17) prior lines of HER2-targeted therapy.
The patients were randomized 2:1 to receive trastuzumab plus capecitabine and either tucatinib or an identical placebo twice daily.
The primary endpoint was PFS, evaluated in the initial 480 randomized patients. The median PFS was 7.8 months in the tucatinib arm and 5.6 months in the control arm (hazard ratio, 0.54; 95% confidence interval, 0.42-0.71; P < .001).
The confirmed overall response rate for patients with measurable disease was 40.6% in the tucatinib arm and 22.8% in the control arm (P = .001). The proportion of patients still in response at 12 months was 33.1% and 12.3%, respectively.
The median OS was 21.9 months in the tucatinib arm and 17.4 months in the placebo arm (HR, 0.66; 95% CI, 0.50-0.88; P = .005). At 24 months, 44.9% and 26.6% of patients, respectively, were still alive.
The most common grade 3 or higher adverse events (in the tucatinib and placebo arms, respectively) were palmar-plantar erythrodysesthesia syndrome (13.1% vs. 9.1%), diarrhea (12.9% vs. 8.6%), elevations in ALT and AST (approximately 5% vs. 0.5% for each), and fatigue (4.7% vs. 4.1%).
Tucatinib in patients with brain involvement
A unique feature of the HER2CLIMB study was that patients with MBC and untreated, symptomatic brain metastases were eligible. Patients with active, untreated central nervous system disease are excluded from virtually all other trials, especially drug-approval trials.
There were 291 patients with brain metastases in HER2CLIMB, 198 (48%) in the tucatinib arm and 93 (46%) in the control arm.
The risk of intracranial progression or death was reduced by 68% in the tucatinib arm (HR, 0.32; 95% CI, 0.22 to 0.48; P < .0001).
The 1-year CNS-PFS rate was 40.2% in the tucatinib arm and 0% in the placebo arm. The median duration of CNS-PFS was 9.9 months and 4.2 months, respectively.
The risk of death was reduced by 42% in the tucatinib arm (HR, 0.58; 95% CI, 0.40-0.85; P = .005). The median OS was 18.1 months and 12.0 months, respectively.
There were more objective responses in the brain with tucatinib (47.3%) than with placebo (20.0%; P = .03). The median duration of response was 6.8 months and 3.0 months, respectively.
Particularly because of its CNS activity and lack of serious, long-term toxicity, tucatinib combination therapy represents an attractive new option for patients with HER2+ MBC.
Neratinib
Neratinib is an irreversible pan-HER TKI that was approved by the FDA in July 2017 for extended adjuvant therapy in patients with early-stage HER2+ breast cancer, following the use of trastuzumab-based therapy.
Long-term results of the ExteNet study led to the approval for use as extended adjuvant therapy.
In February 2020, neratinib was FDA approved in combination with capecitabine for patients with HER2+ MBC after two or more prior anti-HER2–based regimens. The more recent FDA approval was based on results of the NALA trial.
NALA trial
The phase 3 NALA trial included 621 patients with HER2+ MBC who had received at least two prior anti-HER2 based regimens.
Patients were randomized 1:1 to receive neratinib at 240 mg orally once daily on days 1-21 with capecitabine at 750 mg/m2 orally twice daily on days 1-14 or lapatinib at 1,250 mg orally once daily on days 1-21 with capecitabine at 1,000 mg/m2 orally twice daily on days 1-14 for each 21-day cycle. Patients were treated until disease progression or unacceptable toxicity.
The primary endpoints were PFS and OS by blinded, independent, central review.
The median PFS was 5.6 months in the neratinib arm and 5.5 months in the lapatinib arm (HR, 0.76; 95% CI, 0.63-0.93; P = .0059). The PFS rate at 12 months was 28.8% and 14.8%, respectively.
The median OS was 21.0 months in the neratinib arm and 18.7 months in the lapatinib arm (HR, 0.88; 95% CI, 0.72-1.07; P = .2086). The ORR was 32.8% and 26.7%, respectively. The median response duration was 8.5 months and 5.6 months, respectively.
Fewer interventions for CNS disease were required in the neratinib arm than in the lapatinib arm (cumulative incidence, 22.8% vs. 29.2%; P = .043).
The most frequently reported grade 3-4 adverse reactions for the neratinib combination were diarrhea, nausea, vomiting, fatigue, and decreased appetite.
Grade 3 diarrhea occurred in 24.4% of those in the neratinib arm and 12.5% of those in the lapatinib arm. Antidiarrheal medication was used by 98.3% of patients receiving neratinib and 62.1% of patients receiving lapatinib.
Margetuximab-cmkb
Margetuximab is a chimeric Fc-engineered anti-HER2 monoclonal antibody that targets the same epitope as trastuzumab and exerts similar antiproliferative effects.
Compared with trastuzumab, margetuximab has higher affinity for both 158V (high-binding) and 158F (low-binding) alleles of the activating Fc receptor, CD16A. As a result, margetuximab enhances innate immunity, including CD16A-mediated antibody-dependent cellular cytotoxicity, more effectively than trastuzumab. Margetuximab also potentiates adaptive immunity, including enhanced clonality of the T-cell repertoire and induction of HER2-specific T- and B-cell responses.
In December 2020, margetuximab, in combination with chemotherapy, was approved by the FDA for patients with HER2+ MBC after two or more prior anti-HER2 regimens, at least one of which was for metastatic disease. The approved dose is 15 mg/kg IV every 3 weeks.
The study that led to margetuximab’s approval was the phase 3 SOPHIA trial.
SOPHIA trial
SOPHIA was a randomized trial of 536 patients with HER2+ MBC who had received prior treatment with other anti-HER2 therapies, including one to three lines of therapy for MBC.
Patients were randomly assigned 1:1 to receive margetuximab plus chemotherapy or trastuzumab plus chemotherapy. Assignment was stratified by chemotherapy choice (capecitabine, eribulin, gemcitabine, or vinorelbine), the number of previous lines of therapy for MBC, and disease extent.
Co–primary outcome measures were PFS by blinded, independent, central review and OS.
At the second interim analysis, the median PFS was 5.8 months in the margetuximab arm and 4.9 months in the trastuzumab arm (HR, 0.76; 95% CI, 0.59-0.98; P = .033). Results were more impressive in patients with CD16A genotypes containing a 158F allele. In this group, the median PFS was 6.9 months with margetuximab and 5.1 months with trastuzumab (HR, 0.68, 95% CI, 0.52-0.90; P = .005).
At the second interim analysis, the median OS was 21.6 months in the margetuximab arm and 19.8 months in the trastuzumab arm (HR, 0.89; 95% CI, 0.69-1.13; P = .33).
Subgroup data showed no differences in OS between the two arms for any subgroup except HER2+ MBC patients with an IHC score of 2 or higher. This is consistent with the postulated mechanism of action of margetuximab.
The confirmed ORR was 25% in the margetuximab arm and 14% in the trastuzumab arm, with similar durations of response between the study arms.
The most common adverse events in both arms (≥20%), regardless of causality, were fatigue, nausea, diarrhea, and neutropenia. Vomiting was common in the margetuximab arm, and anemia was common in the trastuzumab arm.
Grade 3 or higher adverse events occurred in 53.8% of patients receiving margetuximab and 52.6% of those receiving trastuzumab.
In view of margetuximab’s modest benefits in the SOPHIA trial, the ultimate role for margetuximab in HER2+ MBC may be restricted to patients with the CD16A-158F allele. A neoadjuvant trial is planned in that population.
Take-home messages
There are legitimate arguments regarding whether curing MBC is within reach for certain patient subsets, but there is no argument about whether the outlook for patients with HER2+ MBC has improved dramatically in recent years; it has.
The approval of four unique, new agents for the treatment of women with HER2+ MBC in relapse provides further improvements in outcome for these patients and distinctly different opportunities for tailoring treatment to the special circumstances of each patient (e.g., whether brain metastases are present, desire for oral therapy, comorbidities, experience with prior chemotherapy, etc).
When considered along with the potential for incorporating these drugs in earlier settings in well-designed clinical trials, these new drugs offer great promise to a group of patients who faced a dismal outcome just 2 decades ago.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Before 2001, HER2/neu-positive breast cancer (HER2+) was one of the most dreaded diagnoses a woman could face, as treatment was largely ineffective. The discovery of trastuzumab changed that dramatically.
Over the next 20 years, two additional HER2-targeted therapies – lapatinib and trastuzumab emtansine (TDM-1) – earned approval from the Food and Drug Administration for selected patients with early and late HER2+ breast cancer.
Since 2019, four additional HER2-targeted therapies have been approved by the FDA for HER2+ metastatic breast cancer (MBC), changing the treatment paradigm for those patients substantially.
The new agents are especially useful in certain patient populations. The agents offer the promise of improved survival for patients with recurrent metastatic disease and the potential for further reductions in relapse rates in earlier settings.
Trastuzumab deruxtecan
Trastuzumab deruxtecan is an antibody-drug conjugate that links three components: an anti-HER2 monoclonal antibody, a highly potent topoisomerase I inhibitor payload, and a tetrapeptide-based cleavable linker.
Trastuzumab deruxtecan has a high drug-to-antibody ratio. A membrane-permeable payload offers the potential for activity against adjacent HER2-negative cells in heterogeneous tumors. It has a long half-life (6 days).
Trastuzumab deruxtecan received accelerated approval from the FDA in December 2019 to treat patients with HER2+ MBC who have received two or more prior HER2-targeted regimens, based on the results of the DESTINY-Breast 01 trial.
DESTINY-Breast 01 trial
In the phase 2 DESTINY-Breast 01 trial, 184 patients with a median of six previous treatments received trastuzumab deruxtecan (5.4 mg/kg) intravenously every 21 days. There were 24 patients with treated, asymptomatic brain metastases who participated. Patients with untreated or symptomatic brain metastases were excluded.
Overall, a response to therapy was reported in 112 patients (60.9%), with 6.0% complete and 54.9% partial responses. Most of the patients for whom both baseline and postbaseline data were available had a reduction in tumor size.
The median time until response was 1.6 months, an interval that corresponded to the time until the first scheduled imaging. Three patients (1.6%) had progressive disease, and two patients (1.1%) could not be evaluated.
The median duration of follow-up was 11.1 months, and the median response duration was 14.8 months.
The median progression-free survival (PFS) was 16.4 months, and the median overall survival (OS) was not reached. The median PFS in the patients with brain involvement was 18.1 months.
The most common adverse events of grade 3 or higher were a decreased neutrophil count (20.7%), anemia (8.7%), and nausea (7.6%). Most concerning was that trastuzumab deruxtecan was associated with interstitial lung disease in 13.6% of patients.
Tucatinib
Tucatinib is an oral, highly selective HER2 tyrosine kinase inhibitor (TKI). In April 2020, it was approved by the FDA, in combination with trastuzumab and capecitabine, for adult patients with advanced unresectable or metastatic HER2+ breast cancer who have received one or more prior anti-HER2–based regimens for MBC. The approval included patients with brain metastases.
The recommended tucatinib dose is 300 mg orally twice a day in combination with trastuzumab (at the standard dose) and capecitabine (1,000 mg/m2 given orally twice daily on days 1-14) on a 21-day cycle, until disease progression or unacceptable toxicity.
HER2CLIMB trial
The study that led to the approval of tucatinib was the HER2CLIMB trial. The trial enrolled 612 HER2+ MBC patients who had prior treatment with trastuzumab, pertuzumab, and T-DM1. Patients had received a median of 4 (range, 2-17) prior lines of HER2-targeted therapy.
The patients were randomized 2:1 to receive trastuzumab plus capecitabine and either tucatinib or an identical placebo twice daily.
The primary endpoint was PFS, evaluated in the initial 480 randomized patients. The median PFS was 7.8 months in the tucatinib arm and 5.6 months in the control arm (hazard ratio, 0.54; 95% confidence interval, 0.42-0.71; P < .001).
The confirmed overall response rate for patients with measurable disease was 40.6% in the tucatinib arm and 22.8% in the control arm (P = .001). The proportion of patients still in response at 12 months was 33.1% and 12.3%, respectively.
The median OS was 21.9 months in the tucatinib arm and 17.4 months in the placebo arm (HR, 0.66; 95% CI, 0.50-0.88; P = .005). At 24 months, 44.9% and 26.6% of patients, respectively, were still alive.
The most common grade 3 or higher adverse events (in the tucatinib and placebo arms, respectively) were palmar-plantar erythrodysesthesia syndrome (13.1% vs. 9.1%), diarrhea (12.9% vs. 8.6%), elevations in ALT and AST (approximately 5% vs. 0.5% for each), and fatigue (4.7% vs. 4.1%).
Tucatinib in patients with brain involvement
A unique feature of the HER2CLIMB study was that patients with MBC and untreated, symptomatic brain metastases were eligible. Patients with active, untreated central nervous system disease are excluded from virtually all other trials, especially drug-approval trials.
There were 291 patients with brain metastases in HER2CLIMB, 198 (48%) in the tucatinib arm and 93 (46%) in the control arm.
The risk of intracranial progression or death was reduced by 68% in the tucatinib arm (HR, 0.32; 95% CI, 0.22 to 0.48; P < .0001).
The 1-year CNS-PFS rate was 40.2% in the tucatinib arm and 0% in the placebo arm. The median duration of CNS-PFS was 9.9 months and 4.2 months, respectively.
The risk of death was reduced by 42% in the tucatinib arm (HR, 0.58; 95% CI, 0.40-0.85; P = .005). The median OS was 18.1 months and 12.0 months, respectively.
There were more objective responses in the brain with tucatinib (47.3%) than with placebo (20.0%; P = .03). The median duration of response was 6.8 months and 3.0 months, respectively.
Particularly because of its CNS activity and lack of serious, long-term toxicity, tucatinib combination therapy represents an attractive new option for patients with HER2+ MBC.
Neratinib
Neratinib is an irreversible pan-HER TKI that was approved by the FDA in July 2017 for extended adjuvant therapy in patients with early-stage HER2+ breast cancer, following the use of trastuzumab-based therapy.
Long-term results of the ExteNet study led to the approval for use as extended adjuvant therapy.
In February 2020, neratinib was FDA approved in combination with capecitabine for patients with HER2+ MBC after two or more prior anti-HER2–based regimens. The more recent FDA approval was based on results of the NALA trial.
NALA trial
The phase 3 NALA trial included 621 patients with HER2+ MBC who had received at least two prior anti-HER2 based regimens.
Patients were randomized 1:1 to receive neratinib at 240 mg orally once daily on days 1-21 with capecitabine at 750 mg/m2 orally twice daily on days 1-14 or lapatinib at 1,250 mg orally once daily on days 1-21 with capecitabine at 1,000 mg/m2 orally twice daily on days 1-14 for each 21-day cycle. Patients were treated until disease progression or unacceptable toxicity.
The primary endpoints were PFS and OS by blinded, independent, central review.
The median PFS was 5.6 months in the neratinib arm and 5.5 months in the lapatinib arm (HR, 0.76; 95% CI, 0.63-0.93; P = .0059). The PFS rate at 12 months was 28.8% and 14.8%, respectively.
The median OS was 21.0 months in the neratinib arm and 18.7 months in the lapatinib arm (HR, 0.88; 95% CI, 0.72-1.07; P = .2086). The ORR was 32.8% and 26.7%, respectively. The median response duration was 8.5 months and 5.6 months, respectively.
Fewer interventions for CNS disease were required in the neratinib arm than in the lapatinib arm (cumulative incidence, 22.8% vs. 29.2%; P = .043).
The most frequently reported grade 3-4 adverse reactions for the neratinib combination were diarrhea, nausea, vomiting, fatigue, and decreased appetite.
Grade 3 diarrhea occurred in 24.4% of those in the neratinib arm and 12.5% of those in the lapatinib arm. Antidiarrheal medication was used by 98.3% of patients receiving neratinib and 62.1% of patients receiving lapatinib.
Margetuximab-cmkb
Margetuximab is a chimeric Fc-engineered anti-HER2 monoclonal antibody that targets the same epitope as trastuzumab and exerts similar antiproliferative effects.
Compared with trastuzumab, margetuximab has higher affinity for both 158V (high-binding) and 158F (low-binding) alleles of the activating Fc receptor, CD16A. As a result, margetuximab enhances innate immunity, including CD16A-mediated antibody-dependent cellular cytotoxicity, more effectively than trastuzumab. Margetuximab also potentiates adaptive immunity, including enhanced clonality of the T-cell repertoire and induction of HER2-specific T- and B-cell responses.
In December 2020, margetuximab, in combination with chemotherapy, was approved by the FDA for patients with HER2+ MBC after two or more prior anti-HER2 regimens, at least one of which was for metastatic disease. The approved dose is 15 mg/kg IV every 3 weeks.
The study that led to margetuximab’s approval was the phase 3 SOPHIA trial.
SOPHIA trial
SOPHIA was a randomized trial of 536 patients with HER2+ MBC who had received prior treatment with other anti-HER2 therapies, including one to three lines of therapy for MBC.
Patients were randomly assigned 1:1 to receive margetuximab plus chemotherapy or trastuzumab plus chemotherapy. Assignment was stratified by chemotherapy choice (capecitabine, eribulin, gemcitabine, or vinorelbine), the number of previous lines of therapy for MBC, and disease extent.
Co–primary outcome measures were PFS by blinded, independent, central review and OS.
At the second interim analysis, the median PFS was 5.8 months in the margetuximab arm and 4.9 months in the trastuzumab arm (HR, 0.76; 95% CI, 0.59-0.98; P = .033). Results were more impressive in patients with CD16A genotypes containing a 158F allele. In this group, the median PFS was 6.9 months with margetuximab and 5.1 months with trastuzumab (HR, 0.68, 95% CI, 0.52-0.90; P = .005).
At the second interim analysis, the median OS was 21.6 months in the margetuximab arm and 19.8 months in the trastuzumab arm (HR, 0.89; 95% CI, 0.69-1.13; P = .33).
Subgroup data showed no differences in OS between the two arms for any subgroup except HER2+ MBC patients with an IHC score of 2 or higher. This is consistent with the postulated mechanism of action of margetuximab.
The confirmed ORR was 25% in the margetuximab arm and 14% in the trastuzumab arm, with similar durations of response between the study arms.
The most common adverse events in both arms (≥20%), regardless of causality, were fatigue, nausea, diarrhea, and neutropenia. Vomiting was common in the margetuximab arm, and anemia was common in the trastuzumab arm.
Grade 3 or higher adverse events occurred in 53.8% of patients receiving margetuximab and 52.6% of those receiving trastuzumab.
In view of margetuximab’s modest benefits in the SOPHIA trial, the ultimate role for margetuximab in HER2+ MBC may be restricted to patients with the CD16A-158F allele. A neoadjuvant trial is planned in that population.
Take-home messages
There are legitimate arguments regarding whether curing MBC is within reach for certain patient subsets, but there is no argument about whether the outlook for patients with HER2+ MBC has improved dramatically in recent years; it has.
The approval of four unique, new agents for the treatment of women with HER2+ MBC in relapse provides further improvements in outcome for these patients and distinctly different opportunities for tailoring treatment to the special circumstances of each patient (e.g., whether brain metastases are present, desire for oral therapy, comorbidities, experience with prior chemotherapy, etc).
When considered along with the potential for incorporating these drugs in earlier settings in well-designed clinical trials, these new drugs offer great promise to a group of patients who faced a dismal outcome just 2 decades ago.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Before 2001, HER2/neu-positive breast cancer (HER2+) was one of the most dreaded diagnoses a woman could face, as treatment was largely ineffective. The discovery of trastuzumab changed that dramatically.
Over the next 20 years, two additional HER2-targeted therapies – lapatinib and trastuzumab emtansine (TDM-1) – earned approval from the Food and Drug Administration for selected patients with early and late HER2+ breast cancer.
Since 2019, four additional HER2-targeted therapies have been approved by the FDA for HER2+ metastatic breast cancer (MBC), changing the treatment paradigm for those patients substantially.
The new agents are especially useful in certain patient populations. The agents offer the promise of improved survival for patients with recurrent metastatic disease and the potential for further reductions in relapse rates in earlier settings.
Trastuzumab deruxtecan
Trastuzumab deruxtecan is an antibody-drug conjugate that links three components: an anti-HER2 monoclonal antibody, a highly potent topoisomerase I inhibitor payload, and a tetrapeptide-based cleavable linker.
Trastuzumab deruxtecan has a high drug-to-antibody ratio. A membrane-permeable payload offers the potential for activity against adjacent HER2-negative cells in heterogeneous tumors. It has a long half-life (6 days).
Trastuzumab deruxtecan received accelerated approval from the FDA in December 2019 to treat patients with HER2+ MBC who have received two or more prior HER2-targeted regimens, based on the results of the DESTINY-Breast 01 trial.
DESTINY-Breast 01 trial
In the phase 2 DESTINY-Breast 01 trial, 184 patients with a median of six previous treatments received trastuzumab deruxtecan (5.4 mg/kg) intravenously every 21 days. There were 24 patients with treated, asymptomatic brain metastases who participated. Patients with untreated or symptomatic brain metastases were excluded.
Overall, a response to therapy was reported in 112 patients (60.9%), with 6.0% complete and 54.9% partial responses. Most of the patients for whom both baseline and postbaseline data were available had a reduction in tumor size.
The median time until response was 1.6 months, an interval that corresponded to the time until the first scheduled imaging. Three patients (1.6%) had progressive disease, and two patients (1.1%) could not be evaluated.
The median duration of follow-up was 11.1 months, and the median response duration was 14.8 months.
The median progression-free survival (PFS) was 16.4 months, and the median overall survival (OS) was not reached. The median PFS in the patients with brain involvement was 18.1 months.
The most common adverse events of grade 3 or higher were a decreased neutrophil count (20.7%), anemia (8.7%), and nausea (7.6%). Most concerning was that trastuzumab deruxtecan was associated with interstitial lung disease in 13.6% of patients.
Tucatinib
Tucatinib is an oral, highly selective HER2 tyrosine kinase inhibitor (TKI). In April 2020, it was approved by the FDA, in combination with trastuzumab and capecitabine, for adult patients with advanced unresectable or metastatic HER2+ breast cancer who have received one or more prior anti-HER2–based regimens for MBC. The approval included patients with brain metastases.
The recommended tucatinib dose is 300 mg orally twice a day in combination with trastuzumab (at the standard dose) and capecitabine (1,000 mg/m2 given orally twice daily on days 1-14) on a 21-day cycle, until disease progression or unacceptable toxicity.
HER2CLIMB trial
The study that led to the approval of tucatinib was the HER2CLIMB trial. The trial enrolled 612 HER2+ MBC patients who had prior treatment with trastuzumab, pertuzumab, and T-DM1. Patients had received a median of 4 (range, 2-17) prior lines of HER2-targeted therapy.
The patients were randomized 2:1 to receive trastuzumab plus capecitabine and either tucatinib or an identical placebo twice daily.
The primary endpoint was PFS, evaluated in the initial 480 randomized patients. The median PFS was 7.8 months in the tucatinib arm and 5.6 months in the control arm (hazard ratio, 0.54; 95% confidence interval, 0.42-0.71; P < .001).
The confirmed overall response rate for patients with measurable disease was 40.6% in the tucatinib arm and 22.8% in the control arm (P = .001). The proportion of patients still in response at 12 months was 33.1% and 12.3%, respectively.
The median OS was 21.9 months in the tucatinib arm and 17.4 months in the placebo arm (HR, 0.66; 95% CI, 0.50-0.88; P = .005). At 24 months, 44.9% and 26.6% of patients, respectively, were still alive.
The most common grade 3 or higher adverse events (in the tucatinib and placebo arms, respectively) were palmar-plantar erythrodysesthesia syndrome (13.1% vs. 9.1%), diarrhea (12.9% vs. 8.6%), elevations in ALT and AST (approximately 5% vs. 0.5% for each), and fatigue (4.7% vs. 4.1%).
Tucatinib in patients with brain involvement
A unique feature of the HER2CLIMB study was that patients with MBC and untreated, symptomatic brain metastases were eligible. Patients with active, untreated central nervous system disease are excluded from virtually all other trials, especially drug-approval trials.
There were 291 patients with brain metastases in HER2CLIMB, 198 (48%) in the tucatinib arm and 93 (46%) in the control arm.
The risk of intracranial progression or death was reduced by 68% in the tucatinib arm (HR, 0.32; 95% CI, 0.22 to 0.48; P < .0001).
The 1-year CNS-PFS rate was 40.2% in the tucatinib arm and 0% in the placebo arm. The median duration of CNS-PFS was 9.9 months and 4.2 months, respectively.
The risk of death was reduced by 42% in the tucatinib arm (HR, 0.58; 95% CI, 0.40-0.85; P = .005). The median OS was 18.1 months and 12.0 months, respectively.
There were more objective responses in the brain with tucatinib (47.3%) than with placebo (20.0%; P = .03). The median duration of response was 6.8 months and 3.0 months, respectively.
Particularly because of its CNS activity and lack of serious, long-term toxicity, tucatinib combination therapy represents an attractive new option for patients with HER2+ MBC.
Neratinib
Neratinib is an irreversible pan-HER TKI that was approved by the FDA in July 2017 for extended adjuvant therapy in patients with early-stage HER2+ breast cancer, following the use of trastuzumab-based therapy.
Long-term results of the ExteNet study led to the approval for use as extended adjuvant therapy.
In February 2020, neratinib was FDA approved in combination with capecitabine for patients with HER2+ MBC after two or more prior anti-HER2–based regimens. The more recent FDA approval was based on results of the NALA trial.
NALA trial
The phase 3 NALA trial included 621 patients with HER2+ MBC who had received at least two prior anti-HER2 based regimens.
Patients were randomized 1:1 to receive neratinib at 240 mg orally once daily on days 1-21 with capecitabine at 750 mg/m2 orally twice daily on days 1-14 or lapatinib at 1,250 mg orally once daily on days 1-21 with capecitabine at 1,000 mg/m2 orally twice daily on days 1-14 for each 21-day cycle. Patients were treated until disease progression or unacceptable toxicity.
The primary endpoints were PFS and OS by blinded, independent, central review.
The median PFS was 5.6 months in the neratinib arm and 5.5 months in the lapatinib arm (HR, 0.76; 95% CI, 0.63-0.93; P = .0059). The PFS rate at 12 months was 28.8% and 14.8%, respectively.
The median OS was 21.0 months in the neratinib arm and 18.7 months in the lapatinib arm (HR, 0.88; 95% CI, 0.72-1.07; P = .2086). The ORR was 32.8% and 26.7%, respectively. The median response duration was 8.5 months and 5.6 months, respectively.
Fewer interventions for CNS disease were required in the neratinib arm than in the lapatinib arm (cumulative incidence, 22.8% vs. 29.2%; P = .043).
The most frequently reported grade 3-4 adverse reactions for the neratinib combination were diarrhea, nausea, vomiting, fatigue, and decreased appetite.
Grade 3 diarrhea occurred in 24.4% of those in the neratinib arm and 12.5% of those in the lapatinib arm. Antidiarrheal medication was used by 98.3% of patients receiving neratinib and 62.1% of patients receiving lapatinib.
Margetuximab-cmkb
Margetuximab is a chimeric Fc-engineered anti-HER2 monoclonal antibody that targets the same epitope as trastuzumab and exerts similar antiproliferative effects.
Compared with trastuzumab, margetuximab has higher affinity for both 158V (high-binding) and 158F (low-binding) alleles of the activating Fc receptor, CD16A. As a result, margetuximab enhances innate immunity, including CD16A-mediated antibody-dependent cellular cytotoxicity, more effectively than trastuzumab. Margetuximab also potentiates adaptive immunity, including enhanced clonality of the T-cell repertoire and induction of HER2-specific T- and B-cell responses.
In December 2020, margetuximab, in combination with chemotherapy, was approved by the FDA for patients with HER2+ MBC after two or more prior anti-HER2 regimens, at least one of which was for metastatic disease. The approved dose is 15 mg/kg IV every 3 weeks.
The study that led to margetuximab’s approval was the phase 3 SOPHIA trial.
SOPHIA trial
SOPHIA was a randomized trial of 536 patients with HER2+ MBC who had received prior treatment with other anti-HER2 therapies, including one to three lines of therapy for MBC.
Patients were randomly assigned 1:1 to receive margetuximab plus chemotherapy or trastuzumab plus chemotherapy. Assignment was stratified by chemotherapy choice (capecitabine, eribulin, gemcitabine, or vinorelbine), the number of previous lines of therapy for MBC, and disease extent.
Co–primary outcome measures were PFS by blinded, independent, central review and OS.
At the second interim analysis, the median PFS was 5.8 months in the margetuximab arm and 4.9 months in the trastuzumab arm (HR, 0.76; 95% CI, 0.59-0.98; P = .033). Results were more impressive in patients with CD16A genotypes containing a 158F allele. In this group, the median PFS was 6.9 months with margetuximab and 5.1 months with trastuzumab (HR, 0.68, 95% CI, 0.52-0.90; P = .005).
At the second interim analysis, the median OS was 21.6 months in the margetuximab arm and 19.8 months in the trastuzumab arm (HR, 0.89; 95% CI, 0.69-1.13; P = .33).
Subgroup data showed no differences in OS between the two arms for any subgroup except HER2+ MBC patients with an IHC score of 2 or higher. This is consistent with the postulated mechanism of action of margetuximab.
The confirmed ORR was 25% in the margetuximab arm and 14% in the trastuzumab arm, with similar durations of response between the study arms.
The most common adverse events in both arms (≥20%), regardless of causality, were fatigue, nausea, diarrhea, and neutropenia. Vomiting was common in the margetuximab arm, and anemia was common in the trastuzumab arm.
Grade 3 or higher adverse events occurred in 53.8% of patients receiving margetuximab and 52.6% of those receiving trastuzumab.
In view of margetuximab’s modest benefits in the SOPHIA trial, the ultimate role for margetuximab in HER2+ MBC may be restricted to patients with the CD16A-158F allele. A neoadjuvant trial is planned in that population.
Take-home messages
There are legitimate arguments regarding whether curing MBC is within reach for certain patient subsets, but there is no argument about whether the outlook for patients with HER2+ MBC has improved dramatically in recent years; it has.
The approval of four unique, new agents for the treatment of women with HER2+ MBC in relapse provides further improvements in outcome for these patients and distinctly different opportunities for tailoring treatment to the special circumstances of each patient (e.g., whether brain metastases are present, desire for oral therapy, comorbidities, experience with prior chemotherapy, etc).
When considered along with the potential for incorporating these drugs in earlier settings in well-designed clinical trials, these new drugs offer great promise to a group of patients who faced a dismal outcome just 2 decades ago.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers, as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Coping with postpandemic school hesitancy
As the protective effect of the vaccines becomes increasingly apparent, a large number of school systems are beginning to return to prepandemic in-school learning. But anecdotal reports from around the country are making it clear that some children or their families are hesitant to return to the old norm of face to face learning (Goldstein D. “Schools Are Open, but Many Families Remain Hesitant to Return.” New York Times. 2021 May 9). The possible explanations for this hesitancy include a broad list that goes well beyond the obvious concern about the child contracting COVID-19.
I hear from my grandchildren that remote learning has for the most part been unpleasant and lacked the rigor of their in-class experiences. But, they admit that they have found that, in some situations, they prefer the environment at home because it is less distracting. They also acknowledge that, while they miss seeing their friends, at times the isolation has allowed them to be more efficient. Of course, their observations must be viewed in light of their personalities and the support provided by their parents. For these motivated teenagers, the bottom line is that they would prefer to be in school.
However, for the children who have always been a bit ambivalent about school either because they were anxious in social situations or because they found the academics too challenging, one can easily understand why they might prefer to remain in a less-intimidating home environment. For them, missing their friends may have little draw because they may not have had any friends. And, the negative feedback and bullying they have received at school is too overwhelming. A teenager for whom the pandemic has offered the out-of-school free time to explore her independence, feel more like an adult, and enjoy the benefits of having a job may be hesitant to return to the restrictions imposed by what she sees as the childishness of in-school learning.
Compounding the problem is the risk avoidance posture of some school systems and the hesitancy of some teachers to return to an environment that they continue to view as unsafe despite the evidence of the effectiveness of the vaccines and the minimal threat of in-school spread. It is going to be interesting to see how school administrators and politicians deal with this level of institutional hesitancy. Some schools may take what might be considered a hard-line approach and eliminate remote learning completely.
Regardless of how swiftly and thoughtfully schools return to in-class learning, a large number of children will eventually be faced with the stark reality of returning to a place in which they had felt painfully uncomfortable in the past. Pediatricians must be prepared to see this current wave of school hesitancy morph into a full-fledged tsunami of school refusals.
Successful management of a family whose child finds school too challenging emotionally has always required a combination of careful attention to the possible medical causes of the child’s complaints, consultation with a mental health practitioner, and thoughtful coordination with educators sensitive to the child’s school-generated distress.
It has never been easy to reassure the family of a child with frequent headaches or belly pain that his symptoms have no physical basis and then gently point out that the stress of school attendance may be a contributing factor. Some families who buy into the association may be fortunate enough to be able to offer their child home schooling as a solution to school refusal. But this strategy often requires that one parent remain home and has the temperament and the skills to teach.
Now that we have all seen that remote learning has the potential to work in a crisis, will some parents begin to demand it for their children with school refusal? Who will pay for it? I think you and I would prefer to see a solution that targeted therapeutic interventions aimed at getting the child back in school. But you and I also know those strategies don’t always work.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
As the protective effect of the vaccines becomes increasingly apparent, a large number of school systems are beginning to return to prepandemic in-school learning. But anecdotal reports from around the country are making it clear that some children or their families are hesitant to return to the old norm of face to face learning (Goldstein D. “Schools Are Open, but Many Families Remain Hesitant to Return.” New York Times. 2021 May 9). The possible explanations for this hesitancy include a broad list that goes well beyond the obvious concern about the child contracting COVID-19.
I hear from my grandchildren that remote learning has for the most part been unpleasant and lacked the rigor of their in-class experiences. But, they admit that they have found that, in some situations, they prefer the environment at home because it is less distracting. They also acknowledge that, while they miss seeing their friends, at times the isolation has allowed them to be more efficient. Of course, their observations must be viewed in light of their personalities and the support provided by their parents. For these motivated teenagers, the bottom line is that they would prefer to be in school.
However, for the children who have always been a bit ambivalent about school either because they were anxious in social situations or because they found the academics too challenging, one can easily understand why they might prefer to remain in a less-intimidating home environment. For them, missing their friends may have little draw because they may not have had any friends. And, the negative feedback and bullying they have received at school is too overwhelming. A teenager for whom the pandemic has offered the out-of-school free time to explore her independence, feel more like an adult, and enjoy the benefits of having a job may be hesitant to return to the restrictions imposed by what she sees as the childishness of in-school learning.
Compounding the problem is the risk avoidance posture of some school systems and the hesitancy of some teachers to return to an environment that they continue to view as unsafe despite the evidence of the effectiveness of the vaccines and the minimal threat of in-school spread. It is going to be interesting to see how school administrators and politicians deal with this level of institutional hesitancy. Some schools may take what might be considered a hard-line approach and eliminate remote learning completely.
Regardless of how swiftly and thoughtfully schools return to in-class learning, a large number of children will eventually be faced with the stark reality of returning to a place in which they had felt painfully uncomfortable in the past. Pediatricians must be prepared to see this current wave of school hesitancy morph into a full-fledged tsunami of school refusals.
Successful management of a family whose child finds school too challenging emotionally has always required a combination of careful attention to the possible medical causes of the child’s complaints, consultation with a mental health practitioner, and thoughtful coordination with educators sensitive to the child’s school-generated distress.
It has never been easy to reassure the family of a child with frequent headaches or belly pain that his symptoms have no physical basis and then gently point out that the stress of school attendance may be a contributing factor. Some families who buy into the association may be fortunate enough to be able to offer their child home schooling as a solution to school refusal. But this strategy often requires that one parent remain home and has the temperament and the skills to teach.
Now that we have all seen that remote learning has the potential to work in a crisis, will some parents begin to demand it for their children with school refusal? Who will pay for it? I think you and I would prefer to see a solution that targeted therapeutic interventions aimed at getting the child back in school. But you and I also know those strategies don’t always work.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
As the protective effect of the vaccines becomes increasingly apparent, a large number of school systems are beginning to return to prepandemic in-school learning. But anecdotal reports from around the country are making it clear that some children or their families are hesitant to return to the old norm of face to face learning (Goldstein D. “Schools Are Open, but Many Families Remain Hesitant to Return.” New York Times. 2021 May 9). The possible explanations for this hesitancy include a broad list that goes well beyond the obvious concern about the child contracting COVID-19.
I hear from my grandchildren that remote learning has for the most part been unpleasant and lacked the rigor of their in-class experiences. But, they admit that they have found that, in some situations, they prefer the environment at home because it is less distracting. They also acknowledge that, while they miss seeing their friends, at times the isolation has allowed them to be more efficient. Of course, their observations must be viewed in light of their personalities and the support provided by their parents. For these motivated teenagers, the bottom line is that they would prefer to be in school.
However, for the children who have always been a bit ambivalent about school either because they were anxious in social situations or because they found the academics too challenging, one can easily understand why they might prefer to remain in a less-intimidating home environment. For them, missing their friends may have little draw because they may not have had any friends. And, the negative feedback and bullying they have received at school is too overwhelming. A teenager for whom the pandemic has offered the out-of-school free time to explore her independence, feel more like an adult, and enjoy the benefits of having a job may be hesitant to return to the restrictions imposed by what she sees as the childishness of in-school learning.
Compounding the problem is the risk avoidance posture of some school systems and the hesitancy of some teachers to return to an environment that they continue to view as unsafe despite the evidence of the effectiveness of the vaccines and the minimal threat of in-school spread. It is going to be interesting to see how school administrators and politicians deal with this level of institutional hesitancy. Some schools may take what might be considered a hard-line approach and eliminate remote learning completely.
Regardless of how swiftly and thoughtfully schools return to in-class learning, a large number of children will eventually be faced with the stark reality of returning to a place in which they had felt painfully uncomfortable in the past. Pediatricians must be prepared to see this current wave of school hesitancy morph into a full-fledged tsunami of school refusals.
Successful management of a family whose child finds school too challenging emotionally has always required a combination of careful attention to the possible medical causes of the child’s complaints, consultation with a mental health practitioner, and thoughtful coordination with educators sensitive to the child’s school-generated distress.
It has never been easy to reassure the family of a child with frequent headaches or belly pain that his symptoms have no physical basis and then gently point out that the stress of school attendance may be a contributing factor. Some families who buy into the association may be fortunate enough to be able to offer their child home schooling as a solution to school refusal. But this strategy often requires that one parent remain home and has the temperament and the skills to teach.
Now that we have all seen that remote learning has the potential to work in a crisis, will some parents begin to demand it for their children with school refusal? Who will pay for it? I think you and I would prefer to see a solution that targeted therapeutic interventions aimed at getting the child back in school. But you and I also know those strategies don’t always work.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.

















