User login
In the Literature: Research You Need to Know
Clinical question: Can one or both antiplatelet agents be held in a patient requiring surgery who is on dual antiplatelet therapy to manage a drug-eluting stent?
Background: Data suggest that cessation of antiplatelet therapy within one year of placement of a drug-eluting stent, even for brief time periods, is associated with rapid development of stent thrombosis (ST). However, continuation of antiplatelet agents increases perioperative risk related to hemostasis. No published data is available regarding how long one or both antiplatelet agents can be stopped to allow for safer surgery.
Study design: Case review.
Setting: Cases identified through literature search.
Synopsis: In patients with drug-eluting stents, stopping aspirin presents more risk than stopping thienopyridines. Patients who stopped both antiplatelet agents had ST at seven days (median time to event). If a patient stopped thienopyridines but not aspirin, mean time to ST was 122 days. If a patient stopped thienopyridines then subsequently stopped aspirin, the median time to ST was seven days. This suggests a potential seven-day window when both agents might be stopped, and a longer window when thienopyridines might be stopped if aspirin is continued.
The study only looked at ST cases, not the incidence of ST in all patients undergoing surgery for drug-eluting stents while on dual-antiplatelet therapy, so these results only describe the small fraction of patients who actually have surgery when antiplatelet agents are stopped.
The study design, analyzing existing cases without controls, is limited by the infrequency of the event, resulting in huge sample sizes needed to detect a significant result. The study also describes a median time to ST, which does not provide a true window of protection for the entire duration of the window.
Bottom line: The risks of bleeding complications need to be weighed against the risk of stent thrombosis in individual patients. This study also suggests that the risk of thrombosis is lower if a patient is continued on aspirin while thienopyridines are held.
Citation Eisenberg MJ, Richard PR, Libersan D, Filion KB. Safety of short-term discontinuation of antiplatelet therapy in patients with drug-eluting stents. Circulation. 2009;119:1634-1642.
Reviewed for TH eWire by Erin A. Egan, MD, JD, Ethan Cumbler, MD, Jodie Grout, RN, MS, ANP-C, Jeannette Guerrasio, MD, Nichole Zehnder, MD, and Judy Zerzan, MD, MPH, Hospital Medicine Group, University of Colorado Denver.
Clinical question: Can one or both antiplatelet agents be held in a patient requiring surgery who is on dual antiplatelet therapy to manage a drug-eluting stent?
Background: Data suggest that cessation of antiplatelet therapy within one year of placement of a drug-eluting stent, even for brief time periods, is associated with rapid development of stent thrombosis (ST). However, continuation of antiplatelet agents increases perioperative risk related to hemostasis. No published data is available regarding how long one or both antiplatelet agents can be stopped to allow for safer surgery.
Study design: Case review.
Setting: Cases identified through literature search.
Synopsis: In patients with drug-eluting stents, stopping aspirin presents more risk than stopping thienopyridines. Patients who stopped both antiplatelet agents had ST at seven days (median time to event). If a patient stopped thienopyridines but not aspirin, mean time to ST was 122 days. If a patient stopped thienopyridines then subsequently stopped aspirin, the median time to ST was seven days. This suggests a potential seven-day window when both agents might be stopped, and a longer window when thienopyridines might be stopped if aspirin is continued.
The study only looked at ST cases, not the incidence of ST in all patients undergoing surgery for drug-eluting stents while on dual-antiplatelet therapy, so these results only describe the small fraction of patients who actually have surgery when antiplatelet agents are stopped.
The study design, analyzing existing cases without controls, is limited by the infrequency of the event, resulting in huge sample sizes needed to detect a significant result. The study also describes a median time to ST, which does not provide a true window of protection for the entire duration of the window.
Bottom line: The risks of bleeding complications need to be weighed against the risk of stent thrombosis in individual patients. This study also suggests that the risk of thrombosis is lower if a patient is continued on aspirin while thienopyridines are held.
Citation Eisenberg MJ, Richard PR, Libersan D, Filion KB. Safety of short-term discontinuation of antiplatelet therapy in patients with drug-eluting stents. Circulation. 2009;119:1634-1642.
Reviewed for TH eWire by Erin A. Egan, MD, JD, Ethan Cumbler, MD, Jodie Grout, RN, MS, ANP-C, Jeannette Guerrasio, MD, Nichole Zehnder, MD, and Judy Zerzan, MD, MPH, Hospital Medicine Group, University of Colorado Denver.
Clinical question: Can one or both antiplatelet agents be held in a patient requiring surgery who is on dual antiplatelet therapy to manage a drug-eluting stent?
Background: Data suggest that cessation of antiplatelet therapy within one year of placement of a drug-eluting stent, even for brief time periods, is associated with rapid development of stent thrombosis (ST). However, continuation of antiplatelet agents increases perioperative risk related to hemostasis. No published data is available regarding how long one or both antiplatelet agents can be stopped to allow for safer surgery.
Study design: Case review.
Setting: Cases identified through literature search.
Synopsis: In patients with drug-eluting stents, stopping aspirin presents more risk than stopping thienopyridines. Patients who stopped both antiplatelet agents had ST at seven days (median time to event). If a patient stopped thienopyridines but not aspirin, mean time to ST was 122 days. If a patient stopped thienopyridines then subsequently stopped aspirin, the median time to ST was seven days. This suggests a potential seven-day window when both agents might be stopped, and a longer window when thienopyridines might be stopped if aspirin is continued.
The study only looked at ST cases, not the incidence of ST in all patients undergoing surgery for drug-eluting stents while on dual-antiplatelet therapy, so these results only describe the small fraction of patients who actually have surgery when antiplatelet agents are stopped.
The study design, analyzing existing cases without controls, is limited by the infrequency of the event, resulting in huge sample sizes needed to detect a significant result. The study also describes a median time to ST, which does not provide a true window of protection for the entire duration of the window.
Bottom line: The risks of bleeding complications need to be weighed against the risk of stent thrombosis in individual patients. This study also suggests that the risk of thrombosis is lower if a patient is continued on aspirin while thienopyridines are held.
Citation Eisenberg MJ, Richard PR, Libersan D, Filion KB. Safety of short-term discontinuation of antiplatelet therapy in patients with drug-eluting stents. Circulation. 2009;119:1634-1642.
Reviewed for TH eWire by Erin A. Egan, MD, JD, Ethan Cumbler, MD, Jodie Grout, RN, MS, ANP-C, Jeannette Guerrasio, MD, Nichole Zehnder, MD, and Judy Zerzan, MD, MPH, Hospital Medicine Group, University of Colorado Denver.
A Sickle Cell Primer
Sickle cell anemia is a prototypical single gene deletion disorder familiar to medical students everywhere. Most physicians recall that it is a devastating disorder that starts with a single substitution of valine for glutamic acid in the beta-globin gene of the hemoglobin molecule, rendering the hemoglobin molecule unstable in its de-oxygenated state. This leads to polymerization of hemoglobin molecules within the red cell, deformation of the cell membrane, and sickling. Sickling, in turn, causes blood vessel damage, vaso-occlusion, and various other physiologic effects, which lead to both micro- and macro-vascular protean complications.1
Despite extensive basic research in sickle cell disease (SCD), clinical research into the optimal management of this complex disease has lagged behind. Many adult hospitalists may be unfamiliar with the care of adult SCD patients because most of these patients are cared for in academic centers by hematologists.
In this article we review the common complications of sickle cell anemia in adult patients, the management of associated conditions, and the evidence base for treatment guidelines.
Overview
Sickle cell anemia is not just one disorder, but is, rather, a collection of related disorders involving mutations of the hemoglobin molecule. (These disorders are categorized in Table 1, p. 40) In the United States, SCD primarily affects black Americans, with about 9% of this population having sickle trait.2 One in 600 black Americans has sickle cell anemia, also known as hemoglobin SS disease, and there are an estimated 72,000 patients with SCD in the United States.2
In the early 1970s, the average life expectancy for patients with SCD was estimated to be 14.3 years.3 At that time, more than half of SCD patients died before age five, primarily from infectious complications such as pneumococcal sepsis. Twenty years later, the Cooperative Study of SCD documented a much better life expectancy: an average of 42 for males and 48 for females with homozygous S disease (the set of patients with two copies of the defective hemoglobin S mutation).
On average, patients with hemoglobin SCD routinely survive into their 60s. Many factors, including earlier diagnosis of SCD through universal screening programs, better vaccines and vaccination rates, prophylactic antibiotics in infancy and young childhood, and aggressive treatment of fever in infants, as well as other advances in care, have contributed to this progress. A longer life expectancy in this population has heralded the extension of common complications into adulthood, however, along with the emergence of other adult-specific complications of SCD. (See Table 2, p. 40.) In order to best treat adults with SCD, hospitalists should be aware of these complications, as well as of the advances that have been achieved.
Extension of Pediatric Complications
Perhaps the most common manifestation of SCD in children and adults is the painful crisis. This manifests in infants as a painful swelling in the digits of the hands and feet known as dactylitis. In older children and adults, pain occurs more often in the long bones of the arms and legs and in the sternum, vertebrae, and pelvic bones. Risk factors for painful crises include higher hematocrit values and higher sickle fractions, typically greater than 30%.4 Frequent painful crises are a marker of disease severity and an independent risk factor for death in SCD.
Hospitalists who regularly admit SCD patients are familiar with a subset of “frequent flyers” who experience recurrent painful crises. Remember that this is a small fraction of the total SCD population. In fact, 40% of SCD patients don’t suffer any painful crises requiring medical attention in a given year, and only 1% face more than six such events.5 Pain management in SCD is perhaps beyond the scope of this article, and there are no widely accepted guidelines. A few expert reviews are available for guidance, however.6,7
Treatment Options for Painful Crises
The most beneficial new treatment for painful crisis in the past two decades has been the use of hydroxyurea. After early studies showed promise, a phase III clinical trial in adults with SCD who suffered frequent painful crises (i.e., more than four per year) and who were given doses of up to 35 mg/kg/day of hydroxyurea showed a dramatic decrease in painful episodes. These patients also endured fewer episodes of acute chest syndrome, with a relative risk of 0.44, and experienced a reduced need for transfusions, with a relative risk of 0.64. This trial was stopped early (at 21 months average follow-up) due to the striking findings.8 Subsequent longer-term trials have not shown any of the feared theoretical complications of hydroxyurea, including cytopenias (when the production of one or more blood cell types ceases or is greatly reduced) or secondary malignancies.9 (See Table 3, p. 40.)
Childhood Complications
Some SCD complications are more prominent or more severe in early childhood than in adulthood. Two examples of this phenomenon are infectious complications and splenic sequestration. Pediatricians and parents of children with SCD live in fear of febrile illness because overwhelming infections such as pneumococcal sepsis remain the primary reason for death in children with SCD. This was especially true before Hemophilus influenza type b (Hib) and pneumococcal conjugate vaccines were introduced and before penicillin prophylaxis from birth through age five became universal.
Though functionally asplenic, adult patients are at a relatively low risk for overwhelming infections because their immune systems have matured enough to allow type-specific antibody production to polysaccharide antigens.7 Still, adults with SCD should be aggressively evaluated for infectious etiology of any febrile illness and routinely administered empiric antibiotics to cover strep species.
Splenic sequestration occurs when sickled cells are caught up in the spleen; this causes massive hemolysis, splenic enlargement, and cardiovascular compromise, and it is most common in SCD patients younger than five. Thankfully, this is rare in adults with SS and SB0Thal; however, the notable exception to this rule is in patients with hemoglobin SC disease or SB+Thal, where complete splenic infarction does not routinely occur.
Acute chest syndrome (ACS) is perhaps the most feared complication in both children and adults with SCD—and with good reason. It is the second most common cause for hospital admission in SCD patients, and it is an independent risk factor for death in SCD.7,10 Occurring more often in children, ACS is typically more severe in adults, in whom ACS can progress rapidly to an acute respiratory distress syndrome (ARDS)-like picture, with mortality reaching 5%-9%.11
ACS involves a classic triad of fever, chest pain, and new pulmonary infiltrates on chest X-ray, but patients are invariably hypoxic and dyspneic as well. The etiology of ACS varies. In one series, 54% of patients had an identified infectious pathogen, most commonly chlamydia or mycoplasma. Bronchoalveolar lavage (BAL) showed lipid-laden macrophages in about 16%, suggesting fat embolism after bony infarcts. The remainder of patients were presumed to have primary pulmonary infarctions.11 The treatment of ACS involves broad-spectrum antibiotics, especially atypical coverage, along with transfusion (simple versus exchange), supplemental oxygen, pain control, and judicious use of IV fluids.
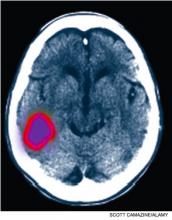
The second most feared complication in SCD is stroke, which can be devastating in both children and young adults. Eleven percent of patients younger than 20 have ischemic strokes, with a risk of stroke that is 200 times higher than that of age-matched peers.9,12 The peak incidence of ischemic stroke occurs before 10 and after 30. These patients are also susceptible to hemorrhagic strokes, the incidence of which peaks in the third decade, for reasons that are unclear.13
Management of acute ischemic stroke in patients with SCD is similar to that used with general patients: antiplatelet therapy, careful attention to normo-glycemia and normovolemia, and maintenance of cerebral perfusion pressure. In addition, SCD patients should receive emergent transfusion to reduce the sickle fraction to less than 30%. Patients with prior transient ischemic attack (TIA) or stroke should be on a chronic monthly transfusion regimen. Also, based on the results of the 1997 STOP I trial (Stroke Prevention Trial in Sickle Cell Anemia), children with SCD should be screened with transcranial Doppler for high velocity flow (>200 cm/second) in the internal jugular and vertebral arteries.14 Children with high velocity flow who were treated with preventative transfusion regimens had a 90% absolute risk reduction in the incidence of first stroke.
In 2001-2006, a follow-up trial (STOP 2) revealed that among patients receiving at least 30 months of chronic transfusion to prevent a first stroke 39% of those randomized to discontinue transfusion had a reversion to high risk Doppler or suffered a stroke within an average of 4.5 months. This has led many to believe that prophylactic transfusion should be a lifelong treatment.15
Complications Seen Primarily in Adults
As the SCD population in this country has grown older, some previously uncommon complications have become more prominent. One predominantly adult complication of SCD is avascular necrosis of the femoral and humeral heads. Though clearly recognized in younger children in whom the incidence is estimated at about 3%, the major burden of femoral osteonecrosis is seen in adults older than 35, in whom prevalence reaches 50%.16 Necrosis of the humeral head can affect nearly 20% of SCD adults as well.17
It is important to recognize this complication as a new or different type of pain, separate from the vaso-occlusive pain usually experienced by SCD patients because it benefits from different therapies. Diagnosis by plain radiography is possible in the late stages, when evidence of remodeling, cystic changes, and sclerosis can be seen, but MRI has become the gold standard, with an estimated diagnostic accuracy of 90%.16 Conservative treatments include NSAIDs and steroid joint injections, but many afflicted patients may need orthopedic referral for joint replacement.
While acute chest syndrome remains a primary cause of mortality in SCD, adults with SCD are also at high risk for the chronic effects of pulmonary arterial hypertension (PAH). Thought to be uncommon in children, PAH affects up to one-third of adult SCD patients.18 Suspicion of this condition can be based on worsened fatigue, new resting hypoxemia, or increased painful crises, but experts advocate universal screening using transthoracic echocardiography. Patients with a tricuspid regurgitant jet velocity of 2.5 m/sec meet diagnostic criteria, and it is notable that the relative risk of death is 7.4 compared with SCD patients without PAH. This correlates with only moderate elevation of pulmonary pressures, suggesting that SCD patients tolerate PAH less well than other populations. Treatment options advocated include hydroxyurea, chronic transfusions, oxygen, pulmonary vasodilators such as prostacyclin and bosentan, and phosphodiesterase inhibitors such as sildenafil.
As SCD patients age, kidney disease is seen with increasing frequency, and three primary mechanisms are recognized. First, ischemic damage in the tubules causes tubular necrosis, which leads to hematuria.19 This condition can range from microscopic to severe gross hematuria, threatening urinary obstruction. Second, damage in the collecting duct impairs the body’s ability to concentrate urine, a condition that is called hyposthenuria. This condition makes SCD patients susceptible to dehydration, especially during physical exertion or in hot weather.
Interestingly, these first two mechanisms of kidney damage are also seen in patients with sickle cell trait. Finally, and most importantly, medullary interstitial fibrosis damages the glomerulus. Clinically, this is the most important mechanism because it leads to nephritic syndrome and chronic kidney disease as well as end-stage renal disease (ESRD).19
Priapism is the persistent, painful erection of the penis in post-pubertal SCD males not associated with sexual desire or relieved by orgasm; this condition is typically defined as lasting more than four to six hours.20 It is particularly common in younger males, with a yearly incidence of 6%-27%; the incidence of priapism in adults approaches 42%. In addition to being extremely painful, prolonged or repeated episodes can lead to impotence. Unfortunately, there are few well-studied treatment options; expert opinion suggests supportive care, including IV fluids, oxygen, ice, elevation, narcotic pain control, and even red cell transfusion in selected cases. Medications used include oral terbutaline, pseudoephedrine, and even stilboestrol, as well as injected phenylephrine, epinephrine, methylene blue, and tenecteplase (TNK-tPA). Surgical procedures attempted have included dorsal penile nerve block, cavernous aspiration, and the Winter procedure, which involves creating a vascular shunt from the corpora cavernosa to the glans penis. Urology consultation is often required for severe cases.
Several forms of eye disease occur in SCD patients. They can be broadly grouped into non-proliferative and proliferative diseases. In the former group, ocular trauma should be recognized as a visual emergency because patients are at risk for developing hyphema, occlusion of the trabeculae in the anterior chamber, and acute glaucoma. SCD patients of all ages are also at risk for acute retinal artery occlusion. Older SCD patients are at greatest risk, however, for a proliferative retinopathy similar to that seen in diabetes.21
Though red blood cell transfusions are helpful in many SCD patients, repeated transfusions can result in infections, immunologic consequences, and iron overload. (See Table 4, p. 41.) Infections that may result include parvovirus B19, HIV, human T-lymphotropic viruses (HTLV) I and II, and viral hepatitides. Immunologic consequences include alloimmunization, which occurs in up to 50% of SCD patients, and potentially fatal acute hemolytic reactions.22 Finally, patients who require frequent transfusions develop iron overload, which in turn causes fatigue, cardiomyopathy, diabetes mellitus, and cirrhosis. This necessitates iron chelators such as deferoxamine, which are disappointingly difficult to administer, requiring either IV or subcutaneous infusion over 12-24 hours daily. One bright note is the recent approval of deferasirox, an iron chelator that is taken orally once daily.
Conclusion
SCD is a complex genetic disease that affects multiple organs. Advances in medical care have increased longevity for SCD patients, and there are currently more adults living with the disease than ever before. Though patients often receive their care in academic centers, hospitalists may encounter them in routine practice. It is useful to have an understanding of the complications of SCD commonly seen in adults and to review the evidence base for care. TH
References
- Platt OS. Preventing stroke in sickle cell anemia. N Engl J Med. 2005 Dec 29;353(26): 2743-2745.
- Quinn CT, Rogers ZR, Buchanan GR. Survival of children with sickle cell disease. Blood. 2004 Jun 1;103(11):4023-4027.
- Platt OS, Brambilla DJ, Rosse WF, et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. [See comment.] N Engl J Med. 1994 Oct 13;331(15):1022-1023.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease: rates and risk factors. N Engl J Med. 1991 Jul 4;325(1):11-16.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease. rates and risk factors. [See comment.] N Engl J Med. 1991;325(24):1747-1748.
- Ballas SK. Pain management of sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):785-802.
- National Institutes of Health. The Management of Sickle Cell Disease. 4th ed. Washington, DC: National Heart, Lung, and Blood Institute; 2002. NIH publication No. 02-2117.
- Charache S, Terrin ML, Moore RD, et al. Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia. [See comment.] N Engl J Med. 1995 May 18;332(20):1317-1322.
- Okpala IE. New therapies for sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):975-987, ix. Review.
- Johnson CS. The acute chest syndrome. Hematol Oncol Clin North Am. 2005;19(5):857-879, vi-vii.
- Vichinsky EP, Neumayr LD, Earles AN, et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med. 2000 Sep 14;342(25):1855-65.
- Platt OS. Preventing stroke in sickle cell anemia. N Engl J Med. 2005 Dec 29;353(26): 2743-2745.
- Ohene-Frempong K, Weiner SJ, Sleeper LA, et al. Cerebrovascular accidents in sickle cell disease: rates and risk factors. Blood. 1998 Jan;91(1):288-294.
- Adams RJ, McKie VC, Hsu L, et al. Prevention of a first stroke by transfusions in children with sickle cell anemia and abnormal results on transcranial Doppler ultrasonography. [See comment.] N Engl J Med. 1998 Jul;339(20):1477-1478.
- Adams RJ, Brambilla D. Optimizing Primary Stroke Prevention in Sickle Cell Anemia (STOP 2) Trial Investigators. Discontinuing prophylactic transfusions used to prevent stroke in sickle cell disease. [See comment.] N Engl J Med. 2005;353(26):2743-2745.
- Aguilar C, Vichinsky E, Neumayr L. Bone and joint disease in sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):929-941, viii. Review.
- Milner PF, Kraus AP, Sebes JI, et al. Osteonecrosis of the humeral head in sickle cell disease. Clin Orthop Relat Res. 1993;289:136-143.
- Castro O, Gladwin MT. Pulmonary hypertension in sickle cell disease: mechanisms, diagnosis, and management. Hematol Oncol Clin North Am. 2005;19(5):881-896, vii.
- Saborio P, Scheinman JI. Sickle cell nephropathy. J Am Soc Nephrol. 1999 Jan;10(1):187-192. Review.
- Vilke GM, Harrigan RA, Ufberg JW, et al. Emergency evaluation and treatment of priapism. J Emerg Med. 2004 Apr;26(3):325-329. Review.
- Charache S. Eye disease in sickling disorders. Hematol Oncol Clin North Am. 1996;10(6): 1357-1362.
- Wanko SO, Telen MJ. Transfusion management in sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):803-826, v-vi.
- Ohene-Frempong K. Indications for red cell transfusion in sickle cell disease. Semin Hematol. 2001 Jan;38(1 Suppl 1):5-13.
- Vichinsky EP, Haberkern CM, Neumayr L, et al. A comparison of conservative and aggressive transfusion regimens in the perioperative management of sickle cell disease. The Preoperative Transfusion in Sickle Cell Disease Study Group. [See comment.] N Engl J Med. 1995 Jul 27;333(4):206-213.
- Piga A, Galanello R, Cappellini, MD, et al. Phase II study of oral chelator ICL670 in thalassaemia patients with transfusional iron overload: efficacy, safety, pharmacokinetics (PK) and pharmacodynamics (PD) after 6 months of therapy. Blood. 2002;100:5a (abstract).
Sickle cell anemia is a prototypical single gene deletion disorder familiar to medical students everywhere. Most physicians recall that it is a devastating disorder that starts with a single substitution of valine for glutamic acid in the beta-globin gene of the hemoglobin molecule, rendering the hemoglobin molecule unstable in its de-oxygenated state. This leads to polymerization of hemoglobin molecules within the red cell, deformation of the cell membrane, and sickling. Sickling, in turn, causes blood vessel damage, vaso-occlusion, and various other physiologic effects, which lead to both micro- and macro-vascular protean complications.1
Despite extensive basic research in sickle cell disease (SCD), clinical research into the optimal management of this complex disease has lagged behind. Many adult hospitalists may be unfamiliar with the care of adult SCD patients because most of these patients are cared for in academic centers by hematologists.
In this article we review the common complications of sickle cell anemia in adult patients, the management of associated conditions, and the evidence base for treatment guidelines.
Overview
Sickle cell anemia is not just one disorder, but is, rather, a collection of related disorders involving mutations of the hemoglobin molecule. (These disorders are categorized in Table 1, p. 40) In the United States, SCD primarily affects black Americans, with about 9% of this population having sickle trait.2 One in 600 black Americans has sickle cell anemia, also known as hemoglobin SS disease, and there are an estimated 72,000 patients with SCD in the United States.2
In the early 1970s, the average life expectancy for patients with SCD was estimated to be 14.3 years.3 At that time, more than half of SCD patients died before age five, primarily from infectious complications such as pneumococcal sepsis. Twenty years later, the Cooperative Study of SCD documented a much better life expectancy: an average of 42 for males and 48 for females with homozygous S disease (the set of patients with two copies of the defective hemoglobin S mutation).
On average, patients with hemoglobin SCD routinely survive into their 60s. Many factors, including earlier diagnosis of SCD through universal screening programs, better vaccines and vaccination rates, prophylactic antibiotics in infancy and young childhood, and aggressive treatment of fever in infants, as well as other advances in care, have contributed to this progress. A longer life expectancy in this population has heralded the extension of common complications into adulthood, however, along with the emergence of other adult-specific complications of SCD. (See Table 2, p. 40.) In order to best treat adults with SCD, hospitalists should be aware of these complications, as well as of the advances that have been achieved.
Extension of Pediatric Complications
Perhaps the most common manifestation of SCD in children and adults is the painful crisis. This manifests in infants as a painful swelling in the digits of the hands and feet known as dactylitis. In older children and adults, pain occurs more often in the long bones of the arms and legs and in the sternum, vertebrae, and pelvic bones. Risk factors for painful crises include higher hematocrit values and higher sickle fractions, typically greater than 30%.4 Frequent painful crises are a marker of disease severity and an independent risk factor for death in SCD.
Hospitalists who regularly admit SCD patients are familiar with a subset of “frequent flyers” who experience recurrent painful crises. Remember that this is a small fraction of the total SCD population. In fact, 40% of SCD patients don’t suffer any painful crises requiring medical attention in a given year, and only 1% face more than six such events.5 Pain management in SCD is perhaps beyond the scope of this article, and there are no widely accepted guidelines. A few expert reviews are available for guidance, however.6,7
Treatment Options for Painful Crises
The most beneficial new treatment for painful crisis in the past two decades has been the use of hydroxyurea. After early studies showed promise, a phase III clinical trial in adults with SCD who suffered frequent painful crises (i.e., more than four per year) and who were given doses of up to 35 mg/kg/day of hydroxyurea showed a dramatic decrease in painful episodes. These patients also endured fewer episodes of acute chest syndrome, with a relative risk of 0.44, and experienced a reduced need for transfusions, with a relative risk of 0.64. This trial was stopped early (at 21 months average follow-up) due to the striking findings.8 Subsequent longer-term trials have not shown any of the feared theoretical complications of hydroxyurea, including cytopenias (when the production of one or more blood cell types ceases or is greatly reduced) or secondary malignancies.9 (See Table 3, p. 40.)
Childhood Complications
Some SCD complications are more prominent or more severe in early childhood than in adulthood. Two examples of this phenomenon are infectious complications and splenic sequestration. Pediatricians and parents of children with SCD live in fear of febrile illness because overwhelming infections such as pneumococcal sepsis remain the primary reason for death in children with SCD. This was especially true before Hemophilus influenza type b (Hib) and pneumococcal conjugate vaccines were introduced and before penicillin prophylaxis from birth through age five became universal.
Though functionally asplenic, adult patients are at a relatively low risk for overwhelming infections because their immune systems have matured enough to allow type-specific antibody production to polysaccharide antigens.7 Still, adults with SCD should be aggressively evaluated for infectious etiology of any febrile illness and routinely administered empiric antibiotics to cover strep species.
Splenic sequestration occurs when sickled cells are caught up in the spleen; this causes massive hemolysis, splenic enlargement, and cardiovascular compromise, and it is most common in SCD patients younger than five. Thankfully, this is rare in adults with SS and SB0Thal; however, the notable exception to this rule is in patients with hemoglobin SC disease or SB+Thal, where complete splenic infarction does not routinely occur.
Acute chest syndrome (ACS) is perhaps the most feared complication in both children and adults with SCD—and with good reason. It is the second most common cause for hospital admission in SCD patients, and it is an independent risk factor for death in SCD.7,10 Occurring more often in children, ACS is typically more severe in adults, in whom ACS can progress rapidly to an acute respiratory distress syndrome (ARDS)-like picture, with mortality reaching 5%-9%.11
ACS involves a classic triad of fever, chest pain, and new pulmonary infiltrates on chest X-ray, but patients are invariably hypoxic and dyspneic as well. The etiology of ACS varies. In one series, 54% of patients had an identified infectious pathogen, most commonly chlamydia or mycoplasma. Bronchoalveolar lavage (BAL) showed lipid-laden macrophages in about 16%, suggesting fat embolism after bony infarcts. The remainder of patients were presumed to have primary pulmonary infarctions.11 The treatment of ACS involves broad-spectrum antibiotics, especially atypical coverage, along with transfusion (simple versus exchange), supplemental oxygen, pain control, and judicious use of IV fluids.
The second most feared complication in SCD is stroke, which can be devastating in both children and young adults. Eleven percent of patients younger than 20 have ischemic strokes, with a risk of stroke that is 200 times higher than that of age-matched peers.9,12 The peak incidence of ischemic stroke occurs before 10 and after 30. These patients are also susceptible to hemorrhagic strokes, the incidence of which peaks in the third decade, for reasons that are unclear.13
Management of acute ischemic stroke in patients with SCD is similar to that used with general patients: antiplatelet therapy, careful attention to normo-glycemia and normovolemia, and maintenance of cerebral perfusion pressure. In addition, SCD patients should receive emergent transfusion to reduce the sickle fraction to less than 30%. Patients with prior transient ischemic attack (TIA) or stroke should be on a chronic monthly transfusion regimen. Also, based on the results of the 1997 STOP I trial (Stroke Prevention Trial in Sickle Cell Anemia), children with SCD should be screened with transcranial Doppler for high velocity flow (>200 cm/second) in the internal jugular and vertebral arteries.14 Children with high velocity flow who were treated with preventative transfusion regimens had a 90% absolute risk reduction in the incidence of first stroke.
In 2001-2006, a follow-up trial (STOP 2) revealed that among patients receiving at least 30 months of chronic transfusion to prevent a first stroke 39% of those randomized to discontinue transfusion had a reversion to high risk Doppler or suffered a stroke within an average of 4.5 months. This has led many to believe that prophylactic transfusion should be a lifelong treatment.15
Complications Seen Primarily in Adults
As the SCD population in this country has grown older, some previously uncommon complications have become more prominent. One predominantly adult complication of SCD is avascular necrosis of the femoral and humeral heads. Though clearly recognized in younger children in whom the incidence is estimated at about 3%, the major burden of femoral osteonecrosis is seen in adults older than 35, in whom prevalence reaches 50%.16 Necrosis of the humeral head can affect nearly 20% of SCD adults as well.17
It is important to recognize this complication as a new or different type of pain, separate from the vaso-occlusive pain usually experienced by SCD patients because it benefits from different therapies. Diagnosis by plain radiography is possible in the late stages, when evidence of remodeling, cystic changes, and sclerosis can be seen, but MRI has become the gold standard, with an estimated diagnostic accuracy of 90%.16 Conservative treatments include NSAIDs and steroid joint injections, but many afflicted patients may need orthopedic referral for joint replacement.
While acute chest syndrome remains a primary cause of mortality in SCD, adults with SCD are also at high risk for the chronic effects of pulmonary arterial hypertension (PAH). Thought to be uncommon in children, PAH affects up to one-third of adult SCD patients.18 Suspicion of this condition can be based on worsened fatigue, new resting hypoxemia, or increased painful crises, but experts advocate universal screening using transthoracic echocardiography. Patients with a tricuspid regurgitant jet velocity of 2.5 m/sec meet diagnostic criteria, and it is notable that the relative risk of death is 7.4 compared with SCD patients without PAH. This correlates with only moderate elevation of pulmonary pressures, suggesting that SCD patients tolerate PAH less well than other populations. Treatment options advocated include hydroxyurea, chronic transfusions, oxygen, pulmonary vasodilators such as prostacyclin and bosentan, and phosphodiesterase inhibitors such as sildenafil.
As SCD patients age, kidney disease is seen with increasing frequency, and three primary mechanisms are recognized. First, ischemic damage in the tubules causes tubular necrosis, which leads to hematuria.19 This condition can range from microscopic to severe gross hematuria, threatening urinary obstruction. Second, damage in the collecting duct impairs the body’s ability to concentrate urine, a condition that is called hyposthenuria. This condition makes SCD patients susceptible to dehydration, especially during physical exertion or in hot weather.
Interestingly, these first two mechanisms of kidney damage are also seen in patients with sickle cell trait. Finally, and most importantly, medullary interstitial fibrosis damages the glomerulus. Clinically, this is the most important mechanism because it leads to nephritic syndrome and chronic kidney disease as well as end-stage renal disease (ESRD).19
Priapism is the persistent, painful erection of the penis in post-pubertal SCD males not associated with sexual desire or relieved by orgasm; this condition is typically defined as lasting more than four to six hours.20 It is particularly common in younger males, with a yearly incidence of 6%-27%; the incidence of priapism in adults approaches 42%. In addition to being extremely painful, prolonged or repeated episodes can lead to impotence. Unfortunately, there are few well-studied treatment options; expert opinion suggests supportive care, including IV fluids, oxygen, ice, elevation, narcotic pain control, and even red cell transfusion in selected cases. Medications used include oral terbutaline, pseudoephedrine, and even stilboestrol, as well as injected phenylephrine, epinephrine, methylene blue, and tenecteplase (TNK-tPA). Surgical procedures attempted have included dorsal penile nerve block, cavernous aspiration, and the Winter procedure, which involves creating a vascular shunt from the corpora cavernosa to the glans penis. Urology consultation is often required for severe cases.
Several forms of eye disease occur in SCD patients. They can be broadly grouped into non-proliferative and proliferative diseases. In the former group, ocular trauma should be recognized as a visual emergency because patients are at risk for developing hyphema, occlusion of the trabeculae in the anterior chamber, and acute glaucoma. SCD patients of all ages are also at risk for acute retinal artery occlusion. Older SCD patients are at greatest risk, however, for a proliferative retinopathy similar to that seen in diabetes.21
Though red blood cell transfusions are helpful in many SCD patients, repeated transfusions can result in infections, immunologic consequences, and iron overload. (See Table 4, p. 41.) Infections that may result include parvovirus B19, HIV, human T-lymphotropic viruses (HTLV) I and II, and viral hepatitides. Immunologic consequences include alloimmunization, which occurs in up to 50% of SCD patients, and potentially fatal acute hemolytic reactions.22 Finally, patients who require frequent transfusions develop iron overload, which in turn causes fatigue, cardiomyopathy, diabetes mellitus, and cirrhosis. This necessitates iron chelators such as deferoxamine, which are disappointingly difficult to administer, requiring either IV or subcutaneous infusion over 12-24 hours daily. One bright note is the recent approval of deferasirox, an iron chelator that is taken orally once daily.
Conclusion
SCD is a complex genetic disease that affects multiple organs. Advances in medical care have increased longevity for SCD patients, and there are currently more adults living with the disease than ever before. Though patients often receive their care in academic centers, hospitalists may encounter them in routine practice. It is useful to have an understanding of the complications of SCD commonly seen in adults and to review the evidence base for care. TH
References
- Platt OS. Preventing stroke in sickle cell anemia. N Engl J Med. 2005 Dec 29;353(26): 2743-2745.
- Quinn CT, Rogers ZR, Buchanan GR. Survival of children with sickle cell disease. Blood. 2004 Jun 1;103(11):4023-4027.
- Platt OS, Brambilla DJ, Rosse WF, et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. [See comment.] N Engl J Med. 1994 Oct 13;331(15):1022-1023.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease: rates and risk factors. N Engl J Med. 1991 Jul 4;325(1):11-16.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease. rates and risk factors. [See comment.] N Engl J Med. 1991;325(24):1747-1748.
- Ballas SK. Pain management of sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):785-802.
- National Institutes of Health. The Management of Sickle Cell Disease. 4th ed. Washington, DC: National Heart, Lung, and Blood Institute; 2002. NIH publication No. 02-2117.
- Charache S, Terrin ML, Moore RD, et al. Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia. [See comment.] N Engl J Med. 1995 May 18;332(20):1317-1322.
- Okpala IE. New therapies for sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):975-987, ix. Review.
- Johnson CS. The acute chest syndrome. Hematol Oncol Clin North Am. 2005;19(5):857-879, vi-vii.
- Vichinsky EP, Neumayr LD, Earles AN, et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med. 2000 Sep 14;342(25):1855-65.
- Platt OS. Preventing stroke in sickle cell anemia. N Engl J Med. 2005 Dec 29;353(26): 2743-2745.
- Ohene-Frempong K, Weiner SJ, Sleeper LA, et al. Cerebrovascular accidents in sickle cell disease: rates and risk factors. Blood. 1998 Jan;91(1):288-294.
- Adams RJ, McKie VC, Hsu L, et al. Prevention of a first stroke by transfusions in children with sickle cell anemia and abnormal results on transcranial Doppler ultrasonography. [See comment.] N Engl J Med. 1998 Jul;339(20):1477-1478.
- Adams RJ, Brambilla D. Optimizing Primary Stroke Prevention in Sickle Cell Anemia (STOP 2) Trial Investigators. Discontinuing prophylactic transfusions used to prevent stroke in sickle cell disease. [See comment.] N Engl J Med. 2005;353(26):2743-2745.
- Aguilar C, Vichinsky E, Neumayr L. Bone and joint disease in sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):929-941, viii. Review.
- Milner PF, Kraus AP, Sebes JI, et al. Osteonecrosis of the humeral head in sickle cell disease. Clin Orthop Relat Res. 1993;289:136-143.
- Castro O, Gladwin MT. Pulmonary hypertension in sickle cell disease: mechanisms, diagnosis, and management. Hematol Oncol Clin North Am. 2005;19(5):881-896, vii.
- Saborio P, Scheinman JI. Sickle cell nephropathy. J Am Soc Nephrol. 1999 Jan;10(1):187-192. Review.
- Vilke GM, Harrigan RA, Ufberg JW, et al. Emergency evaluation and treatment of priapism. J Emerg Med. 2004 Apr;26(3):325-329. Review.
- Charache S. Eye disease in sickling disorders. Hematol Oncol Clin North Am. 1996;10(6): 1357-1362.
- Wanko SO, Telen MJ. Transfusion management in sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):803-826, v-vi.
- Ohene-Frempong K. Indications for red cell transfusion in sickle cell disease. Semin Hematol. 2001 Jan;38(1 Suppl 1):5-13.
- Vichinsky EP, Haberkern CM, Neumayr L, et al. A comparison of conservative and aggressive transfusion regimens in the perioperative management of sickle cell disease. The Preoperative Transfusion in Sickle Cell Disease Study Group. [See comment.] N Engl J Med. 1995 Jul 27;333(4):206-213.
- Piga A, Galanello R, Cappellini, MD, et al. Phase II study of oral chelator ICL670 in thalassaemia patients with transfusional iron overload: efficacy, safety, pharmacokinetics (PK) and pharmacodynamics (PD) after 6 months of therapy. Blood. 2002;100:5a (abstract).
Sickle cell anemia is a prototypical single gene deletion disorder familiar to medical students everywhere. Most physicians recall that it is a devastating disorder that starts with a single substitution of valine for glutamic acid in the beta-globin gene of the hemoglobin molecule, rendering the hemoglobin molecule unstable in its de-oxygenated state. This leads to polymerization of hemoglobin molecules within the red cell, deformation of the cell membrane, and sickling. Sickling, in turn, causes blood vessel damage, vaso-occlusion, and various other physiologic effects, which lead to both micro- and macro-vascular protean complications.1
Despite extensive basic research in sickle cell disease (SCD), clinical research into the optimal management of this complex disease has lagged behind. Many adult hospitalists may be unfamiliar with the care of adult SCD patients because most of these patients are cared for in academic centers by hematologists.
In this article we review the common complications of sickle cell anemia in adult patients, the management of associated conditions, and the evidence base for treatment guidelines.
Overview
Sickle cell anemia is not just one disorder, but is, rather, a collection of related disorders involving mutations of the hemoglobin molecule. (These disorders are categorized in Table 1, p. 40) In the United States, SCD primarily affects black Americans, with about 9% of this population having sickle trait.2 One in 600 black Americans has sickle cell anemia, also known as hemoglobin SS disease, and there are an estimated 72,000 patients with SCD in the United States.2
In the early 1970s, the average life expectancy for patients with SCD was estimated to be 14.3 years.3 At that time, more than half of SCD patients died before age five, primarily from infectious complications such as pneumococcal sepsis. Twenty years later, the Cooperative Study of SCD documented a much better life expectancy: an average of 42 for males and 48 for females with homozygous S disease (the set of patients with two copies of the defective hemoglobin S mutation).
On average, patients with hemoglobin SCD routinely survive into their 60s. Many factors, including earlier diagnosis of SCD through universal screening programs, better vaccines and vaccination rates, prophylactic antibiotics in infancy and young childhood, and aggressive treatment of fever in infants, as well as other advances in care, have contributed to this progress. A longer life expectancy in this population has heralded the extension of common complications into adulthood, however, along with the emergence of other adult-specific complications of SCD. (See Table 2, p. 40.) In order to best treat adults with SCD, hospitalists should be aware of these complications, as well as of the advances that have been achieved.
Extension of Pediatric Complications
Perhaps the most common manifestation of SCD in children and adults is the painful crisis. This manifests in infants as a painful swelling in the digits of the hands and feet known as dactylitis. In older children and adults, pain occurs more often in the long bones of the arms and legs and in the sternum, vertebrae, and pelvic bones. Risk factors for painful crises include higher hematocrit values and higher sickle fractions, typically greater than 30%.4 Frequent painful crises are a marker of disease severity and an independent risk factor for death in SCD.
Hospitalists who regularly admit SCD patients are familiar with a subset of “frequent flyers” who experience recurrent painful crises. Remember that this is a small fraction of the total SCD population. In fact, 40% of SCD patients don’t suffer any painful crises requiring medical attention in a given year, and only 1% face more than six such events.5 Pain management in SCD is perhaps beyond the scope of this article, and there are no widely accepted guidelines. A few expert reviews are available for guidance, however.6,7
Treatment Options for Painful Crises
The most beneficial new treatment for painful crisis in the past two decades has been the use of hydroxyurea. After early studies showed promise, a phase III clinical trial in adults with SCD who suffered frequent painful crises (i.e., more than four per year) and who were given doses of up to 35 mg/kg/day of hydroxyurea showed a dramatic decrease in painful episodes. These patients also endured fewer episodes of acute chest syndrome, with a relative risk of 0.44, and experienced a reduced need for transfusions, with a relative risk of 0.64. This trial was stopped early (at 21 months average follow-up) due to the striking findings.8 Subsequent longer-term trials have not shown any of the feared theoretical complications of hydroxyurea, including cytopenias (when the production of one or more blood cell types ceases or is greatly reduced) or secondary malignancies.9 (See Table 3, p. 40.)
Childhood Complications
Some SCD complications are more prominent or more severe in early childhood than in adulthood. Two examples of this phenomenon are infectious complications and splenic sequestration. Pediatricians and parents of children with SCD live in fear of febrile illness because overwhelming infections such as pneumococcal sepsis remain the primary reason for death in children with SCD. This was especially true before Hemophilus influenza type b (Hib) and pneumococcal conjugate vaccines were introduced and before penicillin prophylaxis from birth through age five became universal.
Though functionally asplenic, adult patients are at a relatively low risk for overwhelming infections because their immune systems have matured enough to allow type-specific antibody production to polysaccharide antigens.7 Still, adults with SCD should be aggressively evaluated for infectious etiology of any febrile illness and routinely administered empiric antibiotics to cover strep species.
Splenic sequestration occurs when sickled cells are caught up in the spleen; this causes massive hemolysis, splenic enlargement, and cardiovascular compromise, and it is most common in SCD patients younger than five. Thankfully, this is rare in adults with SS and SB0Thal; however, the notable exception to this rule is in patients with hemoglobin SC disease or SB+Thal, where complete splenic infarction does not routinely occur.
Acute chest syndrome (ACS) is perhaps the most feared complication in both children and adults with SCD—and with good reason. It is the second most common cause for hospital admission in SCD patients, and it is an independent risk factor for death in SCD.7,10 Occurring more often in children, ACS is typically more severe in adults, in whom ACS can progress rapidly to an acute respiratory distress syndrome (ARDS)-like picture, with mortality reaching 5%-9%.11
ACS involves a classic triad of fever, chest pain, and new pulmonary infiltrates on chest X-ray, but patients are invariably hypoxic and dyspneic as well. The etiology of ACS varies. In one series, 54% of patients had an identified infectious pathogen, most commonly chlamydia or mycoplasma. Bronchoalveolar lavage (BAL) showed lipid-laden macrophages in about 16%, suggesting fat embolism after bony infarcts. The remainder of patients were presumed to have primary pulmonary infarctions.11 The treatment of ACS involves broad-spectrum antibiotics, especially atypical coverage, along with transfusion (simple versus exchange), supplemental oxygen, pain control, and judicious use of IV fluids.
The second most feared complication in SCD is stroke, which can be devastating in both children and young adults. Eleven percent of patients younger than 20 have ischemic strokes, with a risk of stroke that is 200 times higher than that of age-matched peers.9,12 The peak incidence of ischemic stroke occurs before 10 and after 30. These patients are also susceptible to hemorrhagic strokes, the incidence of which peaks in the third decade, for reasons that are unclear.13
Management of acute ischemic stroke in patients with SCD is similar to that used with general patients: antiplatelet therapy, careful attention to normo-glycemia and normovolemia, and maintenance of cerebral perfusion pressure. In addition, SCD patients should receive emergent transfusion to reduce the sickle fraction to less than 30%. Patients with prior transient ischemic attack (TIA) or stroke should be on a chronic monthly transfusion regimen. Also, based on the results of the 1997 STOP I trial (Stroke Prevention Trial in Sickle Cell Anemia), children with SCD should be screened with transcranial Doppler for high velocity flow (>200 cm/second) in the internal jugular and vertebral arteries.14 Children with high velocity flow who were treated with preventative transfusion regimens had a 90% absolute risk reduction in the incidence of first stroke.
In 2001-2006, a follow-up trial (STOP 2) revealed that among patients receiving at least 30 months of chronic transfusion to prevent a first stroke 39% of those randomized to discontinue transfusion had a reversion to high risk Doppler or suffered a stroke within an average of 4.5 months. This has led many to believe that prophylactic transfusion should be a lifelong treatment.15
Complications Seen Primarily in Adults
As the SCD population in this country has grown older, some previously uncommon complications have become more prominent. One predominantly adult complication of SCD is avascular necrosis of the femoral and humeral heads. Though clearly recognized in younger children in whom the incidence is estimated at about 3%, the major burden of femoral osteonecrosis is seen in adults older than 35, in whom prevalence reaches 50%.16 Necrosis of the humeral head can affect nearly 20% of SCD adults as well.17
It is important to recognize this complication as a new or different type of pain, separate from the vaso-occlusive pain usually experienced by SCD patients because it benefits from different therapies. Diagnosis by plain radiography is possible in the late stages, when evidence of remodeling, cystic changes, and sclerosis can be seen, but MRI has become the gold standard, with an estimated diagnostic accuracy of 90%.16 Conservative treatments include NSAIDs and steroid joint injections, but many afflicted patients may need orthopedic referral for joint replacement.
While acute chest syndrome remains a primary cause of mortality in SCD, adults with SCD are also at high risk for the chronic effects of pulmonary arterial hypertension (PAH). Thought to be uncommon in children, PAH affects up to one-third of adult SCD patients.18 Suspicion of this condition can be based on worsened fatigue, new resting hypoxemia, or increased painful crises, but experts advocate universal screening using transthoracic echocardiography. Patients with a tricuspid regurgitant jet velocity of 2.5 m/sec meet diagnostic criteria, and it is notable that the relative risk of death is 7.4 compared with SCD patients without PAH. This correlates with only moderate elevation of pulmonary pressures, suggesting that SCD patients tolerate PAH less well than other populations. Treatment options advocated include hydroxyurea, chronic transfusions, oxygen, pulmonary vasodilators such as prostacyclin and bosentan, and phosphodiesterase inhibitors such as sildenafil.
As SCD patients age, kidney disease is seen with increasing frequency, and three primary mechanisms are recognized. First, ischemic damage in the tubules causes tubular necrosis, which leads to hematuria.19 This condition can range from microscopic to severe gross hematuria, threatening urinary obstruction. Second, damage in the collecting duct impairs the body’s ability to concentrate urine, a condition that is called hyposthenuria. This condition makes SCD patients susceptible to dehydration, especially during physical exertion or in hot weather.
Interestingly, these first two mechanisms of kidney damage are also seen in patients with sickle cell trait. Finally, and most importantly, medullary interstitial fibrosis damages the glomerulus. Clinically, this is the most important mechanism because it leads to nephritic syndrome and chronic kidney disease as well as end-stage renal disease (ESRD).19
Priapism is the persistent, painful erection of the penis in post-pubertal SCD males not associated with sexual desire or relieved by orgasm; this condition is typically defined as lasting more than four to six hours.20 It is particularly common in younger males, with a yearly incidence of 6%-27%; the incidence of priapism in adults approaches 42%. In addition to being extremely painful, prolonged or repeated episodes can lead to impotence. Unfortunately, there are few well-studied treatment options; expert opinion suggests supportive care, including IV fluids, oxygen, ice, elevation, narcotic pain control, and even red cell transfusion in selected cases. Medications used include oral terbutaline, pseudoephedrine, and even stilboestrol, as well as injected phenylephrine, epinephrine, methylene blue, and tenecteplase (TNK-tPA). Surgical procedures attempted have included dorsal penile nerve block, cavernous aspiration, and the Winter procedure, which involves creating a vascular shunt from the corpora cavernosa to the glans penis. Urology consultation is often required for severe cases.
Several forms of eye disease occur in SCD patients. They can be broadly grouped into non-proliferative and proliferative diseases. In the former group, ocular trauma should be recognized as a visual emergency because patients are at risk for developing hyphema, occlusion of the trabeculae in the anterior chamber, and acute glaucoma. SCD patients of all ages are also at risk for acute retinal artery occlusion. Older SCD patients are at greatest risk, however, for a proliferative retinopathy similar to that seen in diabetes.21
Though red blood cell transfusions are helpful in many SCD patients, repeated transfusions can result in infections, immunologic consequences, and iron overload. (See Table 4, p. 41.) Infections that may result include parvovirus B19, HIV, human T-lymphotropic viruses (HTLV) I and II, and viral hepatitides. Immunologic consequences include alloimmunization, which occurs in up to 50% of SCD patients, and potentially fatal acute hemolytic reactions.22 Finally, patients who require frequent transfusions develop iron overload, which in turn causes fatigue, cardiomyopathy, diabetes mellitus, and cirrhosis. This necessitates iron chelators such as deferoxamine, which are disappointingly difficult to administer, requiring either IV or subcutaneous infusion over 12-24 hours daily. One bright note is the recent approval of deferasirox, an iron chelator that is taken orally once daily.
Conclusion
SCD is a complex genetic disease that affects multiple organs. Advances in medical care have increased longevity for SCD patients, and there are currently more adults living with the disease than ever before. Though patients often receive their care in academic centers, hospitalists may encounter them in routine practice. It is useful to have an understanding of the complications of SCD commonly seen in adults and to review the evidence base for care. TH
References
- Platt OS. Preventing stroke in sickle cell anemia. N Engl J Med. 2005 Dec 29;353(26): 2743-2745.
- Quinn CT, Rogers ZR, Buchanan GR. Survival of children with sickle cell disease. Blood. 2004 Jun 1;103(11):4023-4027.
- Platt OS, Brambilla DJ, Rosse WF, et al. Mortality in sickle cell disease. Life expectancy and risk factors for early death. [See comment.] N Engl J Med. 1994 Oct 13;331(15):1022-1023.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease: rates and risk factors. N Engl J Med. 1991 Jul 4;325(1):11-16.
- Platt OS, Thorington BD, Brambilla DJ, et al. Pain in sickle cell disease. rates and risk factors. [See comment.] N Engl J Med. 1991;325(24):1747-1748.
- Ballas SK. Pain management of sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):785-802.
- National Institutes of Health. The Management of Sickle Cell Disease. 4th ed. Washington, DC: National Heart, Lung, and Blood Institute; 2002. NIH publication No. 02-2117.
- Charache S, Terrin ML, Moore RD, et al. Effect of hydroxyurea on the frequency of painful crises in sickle cell anemia. Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia. [See comment.] N Engl J Med. 1995 May 18;332(20):1317-1322.
- Okpala IE. New therapies for sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):975-987, ix. Review.
- Johnson CS. The acute chest syndrome. Hematol Oncol Clin North Am. 2005;19(5):857-879, vi-vii.
- Vichinsky EP, Neumayr LD, Earles AN, et al. Causes and outcomes of the acute chest syndrome in sickle cell disease. National Acute Chest Syndrome Study Group. N Engl J Med. 2000 Sep 14;342(25):1855-65.
- Platt OS. Preventing stroke in sickle cell anemia. N Engl J Med. 2005 Dec 29;353(26): 2743-2745.
- Ohene-Frempong K, Weiner SJ, Sleeper LA, et al. Cerebrovascular accidents in sickle cell disease: rates and risk factors. Blood. 1998 Jan;91(1):288-294.
- Adams RJ, McKie VC, Hsu L, et al. Prevention of a first stroke by transfusions in children with sickle cell anemia and abnormal results on transcranial Doppler ultrasonography. [See comment.] N Engl J Med. 1998 Jul;339(20):1477-1478.
- Adams RJ, Brambilla D. Optimizing Primary Stroke Prevention in Sickle Cell Anemia (STOP 2) Trial Investigators. Discontinuing prophylactic transfusions used to prevent stroke in sickle cell disease. [See comment.] N Engl J Med. 2005;353(26):2743-2745.
- Aguilar C, Vichinsky E, Neumayr L. Bone and joint disease in sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):929-941, viii. Review.
- Milner PF, Kraus AP, Sebes JI, et al. Osteonecrosis of the humeral head in sickle cell disease. Clin Orthop Relat Res. 1993;289:136-143.
- Castro O, Gladwin MT. Pulmonary hypertension in sickle cell disease: mechanisms, diagnosis, and management. Hematol Oncol Clin North Am. 2005;19(5):881-896, vii.
- Saborio P, Scheinman JI. Sickle cell nephropathy. J Am Soc Nephrol. 1999 Jan;10(1):187-192. Review.
- Vilke GM, Harrigan RA, Ufberg JW, et al. Emergency evaluation and treatment of priapism. J Emerg Med. 2004 Apr;26(3):325-329. Review.
- Charache S. Eye disease in sickling disorders. Hematol Oncol Clin North Am. 1996;10(6): 1357-1362.
- Wanko SO, Telen MJ. Transfusion management in sickle cell disease. Hematol Oncol Clin North Am. 2005 Oct;19(5):803-826, v-vi.
- Ohene-Frempong K. Indications for red cell transfusion in sickle cell disease. Semin Hematol. 2001 Jan;38(1 Suppl 1):5-13.
- Vichinsky EP, Haberkern CM, Neumayr L, et al. A comparison of conservative and aggressive transfusion regimens in the perioperative management of sickle cell disease. The Preoperative Transfusion in Sickle Cell Disease Study Group. [See comment.] N Engl J Med. 1995 Jul 27;333(4):206-213.
- Piga A, Galanello R, Cappellini, MD, et al. Phase II study of oral chelator ICL670 in thalassaemia patients with transfusional iron overload: efficacy, safety, pharmacokinetics (PK) and pharmacodynamics (PD) after 6 months of therapy. Blood. 2002;100:5a (abstract).
Initial Impact
The Journal of Hospital Medicine’s debut impact factor (IF) ranks it in the top 20% of its cohort—no small achievement for a peer-reviewed medical journal in its fourth year of publication.
JHM’s 2008 IF is 3.163, a stronger-than-expected showing that Editor-in-Chief Mark V. Williams, MD, FACP, FHM, hopes will translate into increased submissions.
JHM ranks No. 21 out of 107 journals in the Medicine, General, and Internal subject category. Impact factors are an industry metric used as a rough average of citations received by peer-reviewed journals. For comparison's sake, the IF for the Journal of General Internal Medicine is 2.72; the IF for the Annals of Internal Medicine is 17.457; and the IF for the New England Journal of Medicine is 50.017.
“In context of other journals of similar editorial scope … an [IF score] does indicate something about its influence,” says James Testa, senior director of editorial development and publisher relations for Thomson Reuters, which calculates the score.
Dr. Williams is a little more effusive. The IF, he says, “tells us we need to keep doing what we’re doing. ... There are journals that are 20 years old that don’t have impact factors as high as we do.” Dr. Williams is professor and chief of the Division of Hospital Medicine at Northwestern University Feinberg School of Medicine in Chicago. “Hopefully, this will lead to academicians across the world being interested in submitting their scholarly work.”
JHM's top-cited source is Core Competencies in Hospital Medicine: A Framework for Curriculum Development, a supplement published with Volume 1, Issue 1. The supplement's clinical content is an essential tool for practicing hospitalists, providing guidance in the areas of clinical conditions, procedures, healthcare systems, development and methodologies.
"SHM is very proud of our journal, JHM, and getting such a sterling impact factor is a further acknowledgement of JHM’s credibility, reach, and world-class content,” says Larry Wellikson, MD, FHM, CEO of SHM.
The Journal of Hospital Medicine’s debut impact factor (IF) ranks it in the top 20% of its cohort—no small achievement for a peer-reviewed medical journal in its fourth year of publication.
JHM’s 2008 IF is 3.163, a stronger-than-expected showing that Editor-in-Chief Mark V. Williams, MD, FACP, FHM, hopes will translate into increased submissions.
JHM ranks No. 21 out of 107 journals in the Medicine, General, and Internal subject category. Impact factors are an industry metric used as a rough average of citations received by peer-reviewed journals. For comparison's sake, the IF for the Journal of General Internal Medicine is 2.72; the IF for the Annals of Internal Medicine is 17.457; and the IF for the New England Journal of Medicine is 50.017.
“In context of other journals of similar editorial scope … an [IF score] does indicate something about its influence,” says James Testa, senior director of editorial development and publisher relations for Thomson Reuters, which calculates the score.
Dr. Williams is a little more effusive. The IF, he says, “tells us we need to keep doing what we’re doing. ... There are journals that are 20 years old that don’t have impact factors as high as we do.” Dr. Williams is professor and chief of the Division of Hospital Medicine at Northwestern University Feinberg School of Medicine in Chicago. “Hopefully, this will lead to academicians across the world being interested in submitting their scholarly work.”
JHM's top-cited source is Core Competencies in Hospital Medicine: A Framework for Curriculum Development, a supplement published with Volume 1, Issue 1. The supplement's clinical content is an essential tool for practicing hospitalists, providing guidance in the areas of clinical conditions, procedures, healthcare systems, development and methodologies.
"SHM is very proud of our journal, JHM, and getting such a sterling impact factor is a further acknowledgement of JHM’s credibility, reach, and world-class content,” says Larry Wellikson, MD, FHM, CEO of SHM.
The Journal of Hospital Medicine’s debut impact factor (IF) ranks it in the top 20% of its cohort—no small achievement for a peer-reviewed medical journal in its fourth year of publication.
JHM’s 2008 IF is 3.163, a stronger-than-expected showing that Editor-in-Chief Mark V. Williams, MD, FACP, FHM, hopes will translate into increased submissions.
JHM ranks No. 21 out of 107 journals in the Medicine, General, and Internal subject category. Impact factors are an industry metric used as a rough average of citations received by peer-reviewed journals. For comparison's sake, the IF for the Journal of General Internal Medicine is 2.72; the IF for the Annals of Internal Medicine is 17.457; and the IF for the New England Journal of Medicine is 50.017.
“In context of other journals of similar editorial scope … an [IF score] does indicate something about its influence,” says James Testa, senior director of editorial development and publisher relations for Thomson Reuters, which calculates the score.
Dr. Williams is a little more effusive. The IF, he says, “tells us we need to keep doing what we’re doing. ... There are journals that are 20 years old that don’t have impact factors as high as we do.” Dr. Williams is professor and chief of the Division of Hospital Medicine at Northwestern University Feinberg School of Medicine in Chicago. “Hopefully, this will lead to academicians across the world being interested in submitting their scholarly work.”
JHM's top-cited source is Core Competencies in Hospital Medicine: A Framework for Curriculum Development, a supplement published with Volume 1, Issue 1. The supplement's clinical content is an essential tool for practicing hospitalists, providing guidance in the areas of clinical conditions, procedures, healthcare systems, development and methodologies.
"SHM is very proud of our journal, JHM, and getting such a sterling impact factor is a further acknowledgement of JHM’s credibility, reach, and world-class content,” says Larry Wellikson, MD, FHM, CEO of SHM.
Pharma Promises Price Reductions
An $80 billion deal to help reduce out-of-pocket drug costs for Medicare beneficiaries has elicited mixed reactions on what it might mean for patients, as well as calls for hospitalists to remain vigilant about prescription drug expenses.
Under a pledge negotiated with the White House and congressional Democrats, the pharmaceutical industry has promised a 50% discount for name-brand drugs to beneficiaries stuck in the notorious gap of the Medicare Part D prescription drug plan, commonly called the “doughnut hole.” In 2009, the gap in coverage kicks in after $2,700 in total drug costs and persists until $6,154 in total costs, by which point patients have spent as much as $4,350 of their own money for prescription drugs.
President Obama says the gap “has been placing a crushing burden on many older Americans who live on fixed incomes and can’t afford thousands of dollars in out-of-pocket expenses.” The AARP hails the “unique solution” as a “major step forward,” though other industry observers have taken a more circumspect stance and said they want to see tougher cost-control measures in writing.
“You don’t want to look an $80 billion gift horse in the mouth, but there’s some halitosis in this mouth,” says Bill Vaughan, a health policy analyst for Consumers Union in Washington, D.C. “It’s not as pure and altruistic as it seems at first blush, and people need to keep pushing for generics because these [brand-name drugs] are grossly overpriced.”
The deal includes several caveats: The pledge doesn’t address the cost of brand-name drugs before or after the coverage gap, and the doughnut-hole price reduction would go into effect only if Congress enacts healthcare reform legislation.
William D. Atchley Jr., MD, FACP, FHM, says hospitalists need to know what’s available in the hospital pharmacy and maintain an open line of communication with their patients in terms of their access and ability to pay for prescriptions.
“You need to understand patients’ economic status. You need to know if they get their medications from Walmart or the VA hospital pharmacy,” says Dr. Atchley, chief of the division of hospital medicine for Sentara Medical Group in Norfolk, Va., and a member of SHM’s Public Policy Committee. “Cost is an issue to our Medicare patients, and it’s important to collaborate with them to make sure they can afford the drug. If they can’t, you need to work with them to find another affordable drug that will provide the same benefit.”
An $80 billion deal to help reduce out-of-pocket drug costs for Medicare beneficiaries has elicited mixed reactions on what it might mean for patients, as well as calls for hospitalists to remain vigilant about prescription drug expenses.
Under a pledge negotiated with the White House and congressional Democrats, the pharmaceutical industry has promised a 50% discount for name-brand drugs to beneficiaries stuck in the notorious gap of the Medicare Part D prescription drug plan, commonly called the “doughnut hole.” In 2009, the gap in coverage kicks in after $2,700 in total drug costs and persists until $6,154 in total costs, by which point patients have spent as much as $4,350 of their own money for prescription drugs.
President Obama says the gap “has been placing a crushing burden on many older Americans who live on fixed incomes and can’t afford thousands of dollars in out-of-pocket expenses.” The AARP hails the “unique solution” as a “major step forward,” though other industry observers have taken a more circumspect stance and said they want to see tougher cost-control measures in writing.
“You don’t want to look an $80 billion gift horse in the mouth, but there’s some halitosis in this mouth,” says Bill Vaughan, a health policy analyst for Consumers Union in Washington, D.C. “It’s not as pure and altruistic as it seems at first blush, and people need to keep pushing for generics because these [brand-name drugs] are grossly overpriced.”
The deal includes several caveats: The pledge doesn’t address the cost of brand-name drugs before or after the coverage gap, and the doughnut-hole price reduction would go into effect only if Congress enacts healthcare reform legislation.
William D. Atchley Jr., MD, FACP, FHM, says hospitalists need to know what’s available in the hospital pharmacy and maintain an open line of communication with their patients in terms of their access and ability to pay for prescriptions.
“You need to understand patients’ economic status. You need to know if they get their medications from Walmart or the VA hospital pharmacy,” says Dr. Atchley, chief of the division of hospital medicine for Sentara Medical Group in Norfolk, Va., and a member of SHM’s Public Policy Committee. “Cost is an issue to our Medicare patients, and it’s important to collaborate with them to make sure they can afford the drug. If they can’t, you need to work with them to find another affordable drug that will provide the same benefit.”
An $80 billion deal to help reduce out-of-pocket drug costs for Medicare beneficiaries has elicited mixed reactions on what it might mean for patients, as well as calls for hospitalists to remain vigilant about prescription drug expenses.
Under a pledge negotiated with the White House and congressional Democrats, the pharmaceutical industry has promised a 50% discount for name-brand drugs to beneficiaries stuck in the notorious gap of the Medicare Part D prescription drug plan, commonly called the “doughnut hole.” In 2009, the gap in coverage kicks in after $2,700 in total drug costs and persists until $6,154 in total costs, by which point patients have spent as much as $4,350 of their own money for prescription drugs.
President Obama says the gap “has been placing a crushing burden on many older Americans who live on fixed incomes and can’t afford thousands of dollars in out-of-pocket expenses.” The AARP hails the “unique solution” as a “major step forward,” though other industry observers have taken a more circumspect stance and said they want to see tougher cost-control measures in writing.
“You don’t want to look an $80 billion gift horse in the mouth, but there’s some halitosis in this mouth,” says Bill Vaughan, a health policy analyst for Consumers Union in Washington, D.C. “It’s not as pure and altruistic as it seems at first blush, and people need to keep pushing for generics because these [brand-name drugs] are grossly overpriced.”
The deal includes several caveats: The pledge doesn’t address the cost of brand-name drugs before or after the coverage gap, and the doughnut-hole price reduction would go into effect only if Congress enacts healthcare reform legislation.
William D. Atchley Jr., MD, FACP, FHM, says hospitalists need to know what’s available in the hospital pharmacy and maintain an open line of communication with their patients in terms of their access and ability to pay for prescriptions.
“You need to understand patients’ economic status. You need to know if they get their medications from Walmart or the VA hospital pharmacy,” says Dr. Atchley, chief of the division of hospital medicine for Sentara Medical Group in Norfolk, Va., and a member of SHM’s Public Policy Committee. “Cost is an issue to our Medicare patients, and it’s important to collaborate with them to make sure they can afford the drug. If they can’t, you need to work with them to find another affordable drug that will provide the same benefit.”
Bouncebacks
Mr. D is an 80-year-old gentleman who is treated in the hospital for congestive heart failure (CHF) with a question of pneumonia. He is deemed de-conditioned secondary to his medical illness and is sent to an extended care facility. He returns from the extended care facility 18 hours later with fever and shortness of breath. The emergency department (ED) attending speaks to the primary care attending who took care of him during his previous admission; the primary care physician now wants the patient on the hospitalist service.
Does this scenario sound familiar? We have all dealt with such requests during our hospitalist careers and have wondered what the potential repercussions might be.
There is a danger—always present—that the hospitalist service will be used as a receptacle for undesirable patients. The word undesirable is used loosely here to include complicated patients, patients who keep returning to the hospital with recurrent problems, patients with no insurance or poor insurance coverage, and, of course, problematic patients with problematic families.
Complicated Patients, Complicated Consequences
In the scenario described above, the patient comes back with the same diagnoses but now winds up on the hospitalist service. From an ethical perspective alone this seems objectionable. What about the legal ramifications of such a situation? Two different admissions with the same diagnoses occur within a short time frame, but different physician groups are involved in caring for the patient. Additionally, such scenarios often cause patient dissatisfaction and even hostility. Surely it doesn’t make the patient happy to hear that the primary care physician no longer wants the patient on her service.
The hospitalist service usually deals with more complicated patients than the average physician. Often, the primary physicians, who have more constraints on their time, want hospitalists to take care of the more complicated patients. This becomes a problem when a patient who was on a physician’s service is readmitted. Naturally, the primary doctors are frustrated with the recurrent admissions. A case could be made to admit to the hospitalist service when the readmitting diagnosis is different from the previous discharge diagnosis, but when the discharge and readmission diagnoses are the same, the jury is hung.
Loss of Revenue
To many physicians’ minds, a readmission occurs less than 24 hours after the previous discharge; to others, however, a readmission means something else. For example, Medicaid, considers a readmission one that occurs within fewer than seven days of the previous admission. This situation brings about an automatic readmission review, and the readmission is denied if it is perceived that the patient discharge was not appropriate or was premature.
In the case of Mr. D, the hospitalists might end up getting a denial—and suffering a loss of revenue—for a readmission that had nothing to do with them. Hospitalists are seldom cognizant of such repercussions because we are programmed to perform patient care without contemplating the financial implications.
The Joint Commission on Accreditation of Healthcare Organizations (JCAHO) collects data for their core measures program. A part of this effort also involves reporting the readmission rate within 14 days—both for the same diagnosis and for a different one.
Looking again at the case of Mr. D, the core measures would have included estimation of ejection fraction, use of angiotensin-converting enzyme inhibitors, and appropriateness and timing of antibiotics in pneumonia. The hospitalists, who had no control of these indices on the first admission, might have been penalized if these particular measures were not carried out and their omission contributed to the patient’s readmission.
Resident Overflow
Another potential point of contention is the practice of admitting patients to the hospitalist service once the resident teaching service is capped. This is institution-specific. In cases where the entire resident overflow is admitted to the hospitalist service, patients seldom go back to the teaching service because ED doctors label them as belonging to the hospitalist service. Many of these patients either have no insurance (or have Medicaid); in addition, they often have multiple health problems, and noncompliance runs rife. Because unscheduled readmissions are viewed negatively under current guidelines, patients who are handed off in this manner can cause resulting penalties for the hospitalists who end up serving them.
Extended Care Readmissions
Patients with established primary care physicians often go to extended-care facilities where there is another physician of record. At readmission, the new attending is recorded as the patient’s physician. The prior primary attending might have wanted to follow the patient during the readmission. Unfortunately, the ED physician will typically call the newly assigned attending because that is the name that appears on the transfer note. If, at this time, the new attending decides to admit the patient to the hospitalist service, a misunderstanding may ensue. The original primary care attending may view this as an attempt on the part of the hospitalist service to appropriate patients, though the decision to admit to the hospitalist service is seldom made by the hospitalist.
The pitfalls of these practices are accentuated when the readmission occurs within a relatively short time frame. Another downside may arise if the new attending, who knows little of the patient’s history, orders another extensive inpatient workup. This example highlights a potential, and avoidable, cause of spiraling healthcare costs.
The Hospitalist’s Role
We practice in a milieu of increasing scrutiny. Pay for performance is gaining momentum and acceptance. Two years from now, non-compliance with specific indicators, such as readmission rates, will be met with financial penalties. Hospitals complain of decreasing reimbursements. Unscheduled readmissions to the hospital continue to be a source of lost revenue and patient dissatisfaction.
The hospitalist plays a central role in the management of the patient from a medical standpoint. Rules of admission to the hospitalist service vary widely amongst different institutions. Often, depending on patient load and available staffing, these rules are in flux even within institutions. Procedures run the gamut from the so-called “closed system,” in which only specific physicians can admit patients to the hospitalist service, to the “open system,” in which everyone is welcome, on a voluntary basis, to admit to the hospitalist service. The potential pitfalls of the open system will become more and more apparent in the years to come, and many of us will be forced to rethink our models of healthcare delivery. TH
Dr. Chabria is a hospitalist at Waterbury Hospital, Conn., and a clinical instructor at Yale University School of Medicine, New Haven, Conn.
Mr. D is an 80-year-old gentleman who is treated in the hospital for congestive heart failure (CHF) with a question of pneumonia. He is deemed de-conditioned secondary to his medical illness and is sent to an extended care facility. He returns from the extended care facility 18 hours later with fever and shortness of breath. The emergency department (ED) attending speaks to the primary care attending who took care of him during his previous admission; the primary care physician now wants the patient on the hospitalist service.
Does this scenario sound familiar? We have all dealt with such requests during our hospitalist careers and have wondered what the potential repercussions might be.
There is a danger—always present—that the hospitalist service will be used as a receptacle for undesirable patients. The word undesirable is used loosely here to include complicated patients, patients who keep returning to the hospital with recurrent problems, patients with no insurance or poor insurance coverage, and, of course, problematic patients with problematic families.
Complicated Patients, Complicated Consequences
In the scenario described above, the patient comes back with the same diagnoses but now winds up on the hospitalist service. From an ethical perspective alone this seems objectionable. What about the legal ramifications of such a situation? Two different admissions with the same diagnoses occur within a short time frame, but different physician groups are involved in caring for the patient. Additionally, such scenarios often cause patient dissatisfaction and even hostility. Surely it doesn’t make the patient happy to hear that the primary care physician no longer wants the patient on her service.
The hospitalist service usually deals with more complicated patients than the average physician. Often, the primary physicians, who have more constraints on their time, want hospitalists to take care of the more complicated patients. This becomes a problem when a patient who was on a physician’s service is readmitted. Naturally, the primary doctors are frustrated with the recurrent admissions. A case could be made to admit to the hospitalist service when the readmitting diagnosis is different from the previous discharge diagnosis, but when the discharge and readmission diagnoses are the same, the jury is hung.
Loss of Revenue
To many physicians’ minds, a readmission occurs less than 24 hours after the previous discharge; to others, however, a readmission means something else. For example, Medicaid, considers a readmission one that occurs within fewer than seven days of the previous admission. This situation brings about an automatic readmission review, and the readmission is denied if it is perceived that the patient discharge was not appropriate or was premature.
In the case of Mr. D, the hospitalists might end up getting a denial—and suffering a loss of revenue—for a readmission that had nothing to do with them. Hospitalists are seldom cognizant of such repercussions because we are programmed to perform patient care without contemplating the financial implications.
The Joint Commission on Accreditation of Healthcare Organizations (JCAHO) collects data for their core measures program. A part of this effort also involves reporting the readmission rate within 14 days—both for the same diagnosis and for a different one.
Looking again at the case of Mr. D, the core measures would have included estimation of ejection fraction, use of angiotensin-converting enzyme inhibitors, and appropriateness and timing of antibiotics in pneumonia. The hospitalists, who had no control of these indices on the first admission, might have been penalized if these particular measures were not carried out and their omission contributed to the patient’s readmission.
Resident Overflow
Another potential point of contention is the practice of admitting patients to the hospitalist service once the resident teaching service is capped. This is institution-specific. In cases where the entire resident overflow is admitted to the hospitalist service, patients seldom go back to the teaching service because ED doctors label them as belonging to the hospitalist service. Many of these patients either have no insurance (or have Medicaid); in addition, they often have multiple health problems, and noncompliance runs rife. Because unscheduled readmissions are viewed negatively under current guidelines, patients who are handed off in this manner can cause resulting penalties for the hospitalists who end up serving them.
Extended Care Readmissions
Patients with established primary care physicians often go to extended-care facilities where there is another physician of record. At readmission, the new attending is recorded as the patient’s physician. The prior primary attending might have wanted to follow the patient during the readmission. Unfortunately, the ED physician will typically call the newly assigned attending because that is the name that appears on the transfer note. If, at this time, the new attending decides to admit the patient to the hospitalist service, a misunderstanding may ensue. The original primary care attending may view this as an attempt on the part of the hospitalist service to appropriate patients, though the decision to admit to the hospitalist service is seldom made by the hospitalist.
The pitfalls of these practices are accentuated when the readmission occurs within a relatively short time frame. Another downside may arise if the new attending, who knows little of the patient’s history, orders another extensive inpatient workup. This example highlights a potential, and avoidable, cause of spiraling healthcare costs.
The Hospitalist’s Role
We practice in a milieu of increasing scrutiny. Pay for performance is gaining momentum and acceptance. Two years from now, non-compliance with specific indicators, such as readmission rates, will be met with financial penalties. Hospitals complain of decreasing reimbursements. Unscheduled readmissions to the hospital continue to be a source of lost revenue and patient dissatisfaction.
The hospitalist plays a central role in the management of the patient from a medical standpoint. Rules of admission to the hospitalist service vary widely amongst different institutions. Often, depending on patient load and available staffing, these rules are in flux even within institutions. Procedures run the gamut from the so-called “closed system,” in which only specific physicians can admit patients to the hospitalist service, to the “open system,” in which everyone is welcome, on a voluntary basis, to admit to the hospitalist service. The potential pitfalls of the open system will become more and more apparent in the years to come, and many of us will be forced to rethink our models of healthcare delivery. TH
Dr. Chabria is a hospitalist at Waterbury Hospital, Conn., and a clinical instructor at Yale University School of Medicine, New Haven, Conn.
Mr. D is an 80-year-old gentleman who is treated in the hospital for congestive heart failure (CHF) with a question of pneumonia. He is deemed de-conditioned secondary to his medical illness and is sent to an extended care facility. He returns from the extended care facility 18 hours later with fever and shortness of breath. The emergency department (ED) attending speaks to the primary care attending who took care of him during his previous admission; the primary care physician now wants the patient on the hospitalist service.
Does this scenario sound familiar? We have all dealt with such requests during our hospitalist careers and have wondered what the potential repercussions might be.
There is a danger—always present—that the hospitalist service will be used as a receptacle for undesirable patients. The word undesirable is used loosely here to include complicated patients, patients who keep returning to the hospital with recurrent problems, patients with no insurance or poor insurance coverage, and, of course, problematic patients with problematic families.
Complicated Patients, Complicated Consequences
In the scenario described above, the patient comes back with the same diagnoses but now winds up on the hospitalist service. From an ethical perspective alone this seems objectionable. What about the legal ramifications of such a situation? Two different admissions with the same diagnoses occur within a short time frame, but different physician groups are involved in caring for the patient. Additionally, such scenarios often cause patient dissatisfaction and even hostility. Surely it doesn’t make the patient happy to hear that the primary care physician no longer wants the patient on her service.
The hospitalist service usually deals with more complicated patients than the average physician. Often, the primary physicians, who have more constraints on their time, want hospitalists to take care of the more complicated patients. This becomes a problem when a patient who was on a physician’s service is readmitted. Naturally, the primary doctors are frustrated with the recurrent admissions. A case could be made to admit to the hospitalist service when the readmitting diagnosis is different from the previous discharge diagnosis, but when the discharge and readmission diagnoses are the same, the jury is hung.
Loss of Revenue
To many physicians’ minds, a readmission occurs less than 24 hours after the previous discharge; to others, however, a readmission means something else. For example, Medicaid, considers a readmission one that occurs within fewer than seven days of the previous admission. This situation brings about an automatic readmission review, and the readmission is denied if it is perceived that the patient discharge was not appropriate or was premature.
In the case of Mr. D, the hospitalists might end up getting a denial—and suffering a loss of revenue—for a readmission that had nothing to do with them. Hospitalists are seldom cognizant of such repercussions because we are programmed to perform patient care without contemplating the financial implications.
The Joint Commission on Accreditation of Healthcare Organizations (JCAHO) collects data for their core measures program. A part of this effort also involves reporting the readmission rate within 14 days—both for the same diagnosis and for a different one.
Looking again at the case of Mr. D, the core measures would have included estimation of ejection fraction, use of angiotensin-converting enzyme inhibitors, and appropriateness and timing of antibiotics in pneumonia. The hospitalists, who had no control of these indices on the first admission, might have been penalized if these particular measures were not carried out and their omission contributed to the patient’s readmission.
Resident Overflow
Another potential point of contention is the practice of admitting patients to the hospitalist service once the resident teaching service is capped. This is institution-specific. In cases where the entire resident overflow is admitted to the hospitalist service, patients seldom go back to the teaching service because ED doctors label them as belonging to the hospitalist service. Many of these patients either have no insurance (or have Medicaid); in addition, they often have multiple health problems, and noncompliance runs rife. Because unscheduled readmissions are viewed negatively under current guidelines, patients who are handed off in this manner can cause resulting penalties for the hospitalists who end up serving them.
Extended Care Readmissions
Patients with established primary care physicians often go to extended-care facilities where there is another physician of record. At readmission, the new attending is recorded as the patient’s physician. The prior primary attending might have wanted to follow the patient during the readmission. Unfortunately, the ED physician will typically call the newly assigned attending because that is the name that appears on the transfer note. If, at this time, the new attending decides to admit the patient to the hospitalist service, a misunderstanding may ensue. The original primary care attending may view this as an attempt on the part of the hospitalist service to appropriate patients, though the decision to admit to the hospitalist service is seldom made by the hospitalist.
The pitfalls of these practices are accentuated when the readmission occurs within a relatively short time frame. Another downside may arise if the new attending, who knows little of the patient’s history, orders another extensive inpatient workup. This example highlights a potential, and avoidable, cause of spiraling healthcare costs.
The Hospitalist’s Role
We practice in a milieu of increasing scrutiny. Pay for performance is gaining momentum and acceptance. Two years from now, non-compliance with specific indicators, such as readmission rates, will be met with financial penalties. Hospitals complain of decreasing reimbursements. Unscheduled readmissions to the hospital continue to be a source of lost revenue and patient dissatisfaction.
The hospitalist plays a central role in the management of the patient from a medical standpoint. Rules of admission to the hospitalist service vary widely amongst different institutions. Often, depending on patient load and available staffing, these rules are in flux even within institutions. Procedures run the gamut from the so-called “closed system,” in which only specific physicians can admit patients to the hospitalist service, to the “open system,” in which everyone is welcome, on a voluntary basis, to admit to the hospitalist service. The potential pitfalls of the open system will become more and more apparent in the years to come, and many of us will be forced to rethink our models of healthcare delivery. TH
Dr. Chabria is a hospitalist at Waterbury Hospital, Conn., and a clinical instructor at Yale University School of Medicine, New Haven, Conn.
Relational Growth
A new survey conducted by SHM and the American Medical Association’s (AMA) Organized Medical Staff Section finds that while more than 90% of hospitalists feel an HM presence improves the quality of hospital care, less than half of primary-care physicians (PCPs) feel the same way. On the bright side, the percentage of PCPs with favorable views of HM is climbing.
“There seems to be a general trend toward improvement of how primary-care physicians view hospitalists,” says Chad Whelan, MD, FHM, associate professor of medicine and director of the division of hospital medicine at Loyola University Chicago Stritch School of Medicine and chair of SHM’s Career Satisfaction Task Force. “But there are still very different views. ... It’s a narrower gap, but it’s still a gap.”
The data come from a recent survey of 874 hospitalists and 497 PCPs. The "Survey on the Growing Hospitalist Trend" is a follow-up to a similar study conducted two years ago to gauge the effects of HM on the primary-care model, and vice versa. The study also looks to define perceptions of the hospitalist model on the care of shared patients.
One key issue the survey examined was viewpoints on how well hospitalists and PCPs communicate with each other. Upon admission, the survey shows, half of hospitalists feel they communicate effectively with PCPs, but only 25% of PCPs feel the same way about hospitalists. On discharge, the disparity grows, with 70% of hospitalists saying they feel they communicate well with PCPs; however, only 35% of PCPs agree.
Still, 46% of PCPs agree or strongly agree that hospitalists have improved the overall quality of hospital care. That number is up from 40% two years ago.
“Everyone can learn from this,” says James DeNuccio, director of the AMA’s Organized Medical Staff Services and Physicians in Practice. “If the hospitalists and PCPs both can learn something from this, that helps them adjust their practice. In the end, patients will benefit.”
A new survey conducted by SHM and the American Medical Association’s (AMA) Organized Medical Staff Section finds that while more than 90% of hospitalists feel an HM presence improves the quality of hospital care, less than half of primary-care physicians (PCPs) feel the same way. On the bright side, the percentage of PCPs with favorable views of HM is climbing.
“There seems to be a general trend toward improvement of how primary-care physicians view hospitalists,” says Chad Whelan, MD, FHM, associate professor of medicine and director of the division of hospital medicine at Loyola University Chicago Stritch School of Medicine and chair of SHM’s Career Satisfaction Task Force. “But there are still very different views. ... It’s a narrower gap, but it’s still a gap.”
The data come from a recent survey of 874 hospitalists and 497 PCPs. The "Survey on the Growing Hospitalist Trend" is a follow-up to a similar study conducted two years ago to gauge the effects of HM on the primary-care model, and vice versa. The study also looks to define perceptions of the hospitalist model on the care of shared patients.
One key issue the survey examined was viewpoints on how well hospitalists and PCPs communicate with each other. Upon admission, the survey shows, half of hospitalists feel they communicate effectively with PCPs, but only 25% of PCPs feel the same way about hospitalists. On discharge, the disparity grows, with 70% of hospitalists saying they feel they communicate well with PCPs; however, only 35% of PCPs agree.
Still, 46% of PCPs agree or strongly agree that hospitalists have improved the overall quality of hospital care. That number is up from 40% two years ago.
“Everyone can learn from this,” says James DeNuccio, director of the AMA’s Organized Medical Staff Services and Physicians in Practice. “If the hospitalists and PCPs both can learn something from this, that helps them adjust their practice. In the end, patients will benefit.”
A new survey conducted by SHM and the American Medical Association’s (AMA) Organized Medical Staff Section finds that while more than 90% of hospitalists feel an HM presence improves the quality of hospital care, less than half of primary-care physicians (PCPs) feel the same way. On the bright side, the percentage of PCPs with favorable views of HM is climbing.
“There seems to be a general trend toward improvement of how primary-care physicians view hospitalists,” says Chad Whelan, MD, FHM, associate professor of medicine and director of the division of hospital medicine at Loyola University Chicago Stritch School of Medicine and chair of SHM’s Career Satisfaction Task Force. “But there are still very different views. ... It’s a narrower gap, but it’s still a gap.”
The data come from a recent survey of 874 hospitalists and 497 PCPs. The "Survey on the Growing Hospitalist Trend" is a follow-up to a similar study conducted two years ago to gauge the effects of HM on the primary-care model, and vice versa. The study also looks to define perceptions of the hospitalist model on the care of shared patients.
One key issue the survey examined was viewpoints on how well hospitalists and PCPs communicate with each other. Upon admission, the survey shows, half of hospitalists feel they communicate effectively with PCPs, but only 25% of PCPs feel the same way about hospitalists. On discharge, the disparity grows, with 70% of hospitalists saying they feel they communicate well with PCPs; however, only 35% of PCPs agree.
Still, 46% of PCPs agree or strongly agree that hospitalists have improved the overall quality of hospital care. That number is up from 40% two years ago.
“Everyone can learn from this,” says James DeNuccio, director of the AMA’s Organized Medical Staff Services and Physicians in Practice. “If the hospitalists and PCPs both can learn something from this, that helps them adjust their practice. In the end, patients will benefit.”
In the Literature: Research You Need to Know
Clinical question: Do the benefits of statin use in secondary stroke prevention apply equally to younger patients as they do elderly patients?
Background: The 2003 SPARCL trial assigned stroke patients with LDL of 100 mg/dL to 190 mg/dL to treatment with 80 mg atorvastatin or placebo. Over median of 4.9 years of followup, 13.1% of patients in the placebo group had a recurrent stroke, compared with 11.2% of patients in the atorvastatin group, resulting in a significant, 2.2% absolute reduction of stroke risk.
Study design: Secondary analysis of a randomized, double-blind, placebo-controlled trial.
Setting: Multicenter study.
Synopsis: Using data from the original SPARCL trial, 2,482 patients in the younger cohort (<65)—a mean age of 54 years—were compared with 2,249 patients in the elderly group (>65) with a mean age of 72.4 years. There was comparable reduction of LDL cholesterol in both groups and no significant differences in adverse drug events.
The magnitude of reduction in fatal or nonfatal second strokes was greater for younger patients (RRR 26%, P=0.02) than for older patients (RRR 10%, P=0.33). However, in the elderly cohort, secondary endpoints including stroke or TIA (HR 0.79, P=0.01), major coronary events (HR 0.68, P=0.035), or any coronary heart event (HR 0.61, P=0.0006) were significantly reduced by statin use. Mortality was not reduced in either group.
The authors found a lack of heterogeneity of benefit in treatment-age interaction analysis and concluded that the results support use of statins in the elderly after stroke. This study is limited by its post-hoc analysis, which might have been underpowered to detect a difference in the primary endpoint when separated into the age groups.
Bottom line: Elderly patients derive less stroke-reduction benefit from high-dose, high-potency statins compared with younger patients in the post-stroke period. Even so, statins continue to derive statistically significant reductions in other cerebrovascular and cardiac events.
Citation: Chaturvedi S, Zivin J, Breazna A, et al. Effect of atorvastatin in elderly patients with a recent stroke or transient ischemic attack. Neurology. 2009;72:688-694.
Reviewed for TH eWire by Erin A. Egan, MD, JD, Ethan Cumbler, MD, Jodie Grout, RN, MS, ANP-C, Jeannette Guerrasio, MD, Nichole Zehnder, MD, and Judy Zerzan, MD, MPH, Hospital Medicine Group, University of Colorado Denver
Clinical question: Do the benefits of statin use in secondary stroke prevention apply equally to younger patients as they do elderly patients?
Background: The 2003 SPARCL trial assigned stroke patients with LDL of 100 mg/dL to 190 mg/dL to treatment with 80 mg atorvastatin or placebo. Over median of 4.9 years of followup, 13.1% of patients in the placebo group had a recurrent stroke, compared with 11.2% of patients in the atorvastatin group, resulting in a significant, 2.2% absolute reduction of stroke risk.
Study design: Secondary analysis of a randomized, double-blind, placebo-controlled trial.
Setting: Multicenter study.
Synopsis: Using data from the original SPARCL trial, 2,482 patients in the younger cohort (<65)—a mean age of 54 years—were compared with 2,249 patients in the elderly group (>65) with a mean age of 72.4 years. There was comparable reduction of LDL cholesterol in both groups and no significant differences in adverse drug events.
The magnitude of reduction in fatal or nonfatal second strokes was greater for younger patients (RRR 26%, P=0.02) than for older patients (RRR 10%, P=0.33). However, in the elderly cohort, secondary endpoints including stroke or TIA (HR 0.79, P=0.01), major coronary events (HR 0.68, P=0.035), or any coronary heart event (HR 0.61, P=0.0006) were significantly reduced by statin use. Mortality was not reduced in either group.
The authors found a lack of heterogeneity of benefit in treatment-age interaction analysis and concluded that the results support use of statins in the elderly after stroke. This study is limited by its post-hoc analysis, which might have been underpowered to detect a difference in the primary endpoint when separated into the age groups.
Bottom line: Elderly patients derive less stroke-reduction benefit from high-dose, high-potency statins compared with younger patients in the post-stroke period. Even so, statins continue to derive statistically significant reductions in other cerebrovascular and cardiac events.
Citation: Chaturvedi S, Zivin J, Breazna A, et al. Effect of atorvastatin in elderly patients with a recent stroke or transient ischemic attack. Neurology. 2009;72:688-694.
Reviewed for TH eWire by Erin A. Egan, MD, JD, Ethan Cumbler, MD, Jodie Grout, RN, MS, ANP-C, Jeannette Guerrasio, MD, Nichole Zehnder, MD, and Judy Zerzan, MD, MPH, Hospital Medicine Group, University of Colorado Denver
Clinical question: Do the benefits of statin use in secondary stroke prevention apply equally to younger patients as they do elderly patients?
Background: The 2003 SPARCL trial assigned stroke patients with LDL of 100 mg/dL to 190 mg/dL to treatment with 80 mg atorvastatin or placebo. Over median of 4.9 years of followup, 13.1% of patients in the placebo group had a recurrent stroke, compared with 11.2% of patients in the atorvastatin group, resulting in a significant, 2.2% absolute reduction of stroke risk.
Study design: Secondary analysis of a randomized, double-blind, placebo-controlled trial.
Setting: Multicenter study.
Synopsis: Using data from the original SPARCL trial, 2,482 patients in the younger cohort (<65)—a mean age of 54 years—were compared with 2,249 patients in the elderly group (>65) with a mean age of 72.4 years. There was comparable reduction of LDL cholesterol in both groups and no significant differences in adverse drug events.
The magnitude of reduction in fatal or nonfatal second strokes was greater for younger patients (RRR 26%, P=0.02) than for older patients (RRR 10%, P=0.33). However, in the elderly cohort, secondary endpoints including stroke or TIA (HR 0.79, P=0.01), major coronary events (HR 0.68, P=0.035), or any coronary heart event (HR 0.61, P=0.0006) were significantly reduced by statin use. Mortality was not reduced in either group.
The authors found a lack of heterogeneity of benefit in treatment-age interaction analysis and concluded that the results support use of statins in the elderly after stroke. This study is limited by its post-hoc analysis, which might have been underpowered to detect a difference in the primary endpoint when separated into the age groups.
Bottom line: Elderly patients derive less stroke-reduction benefit from high-dose, high-potency statins compared with younger patients in the post-stroke period. Even so, statins continue to derive statistically significant reductions in other cerebrovascular and cardiac events.
Citation: Chaturvedi S, Zivin J, Breazna A, et al. Effect of atorvastatin in elderly patients with a recent stroke or transient ischemic attack. Neurology. 2009;72:688-694.
Reviewed for TH eWire by Erin A. Egan, MD, JD, Ethan Cumbler, MD, Jodie Grout, RN, MS, ANP-C, Jeannette Guerrasio, MD, Nichole Zehnder, MD, and Judy Zerzan, MD, MPH, Hospital Medicine Group, University of Colorado Denver
Internal Peer Review
As far as I can tell, few hospitalist groups conduct any sort of formal peer review. Most seem to rely on the hospital’s medical staff peer review to encourage quality of care and address shortcomings; the review is often coupled with a salary incentive paid for good performance on certain quality metrics. While these reviews are of some value, I think they are pretty blunt instruments. Every hospitalist practice should think about developing a more robust system of peer review for their group.
Review Categories
Assessment of each provider’s individual performance, whether they are an MD, nurse practitioner, or physician assistant, can be divided into three general categories. The first is the traditional “human resources” category of performance, which includes whether the person gets along well with others in the practice as well as other hospital staff, patients, and families. Does the person arrive at work when scheduled, manage time effectively, and work efficiently? Do nurses and other hospital staff have compliments or complaints about this doctor?
The second category of performance can encompass the hospitalist’s business and operational contributions to the practice. Do they document, code, and bill visits correctly? Do they attend and participate in meetings and serve on one or more hospital committees?
The third category assesses measurable quality of care. This could include an assessment of mortality, readmission rate, performance on such quality metrics as core measures, and performance on selected initiatives (e.g., appropriate VTE prophylaxis). Aggregate data for these measures can be difficult to attribute to a single hospitalist, so this may require a review of individual charts instead of relying on reports generated by the hospital’s data systems.
A number of metrics might apply to more than one of the three categories. For example, documenting accurate medicine reconciliation can be thought of as both a quality issue (good for patients) and a business issue (e.g., your hospital might provide a financial reward to your group for good performance). Ensuring the referring physician is “CC’d” on all dictated reports is both a quality and business issue. It really doesn’t matter which category you put these in.
The categories I have listed, and the sample items in each, are intended as examples. You should think about the unique attributes of your practice and its current priorities in order to develop the best internal peer review system for your group. You probably will want to change metrics periodically. For example, you may choose to focus on VTE prophylaxis for now, but at some point it may make sense to replace it with a new metric, such as glycemic control.
Manage the Review
There is no single right approach to conducting your own peer review. Just make sure that the process is fair and meaningful for all involved. The process probably will be more valuable if most of the data on each hospitalist can be reviewed by the whole group, or at least by a designated peer review committee. The main exceptions to such transparency are issues in the first human resources category. If a nurse or another hospitalist has specific criticisms of one hospitalist, it is best not to share that information with the whole group. But it should be fine for everyone in the group to know who is best and worst at things like documenting and coding visits or ordering VTE prophylaxis when needed. Beyond these general principles, the specific process your group uses for peer review can take many forms.
It may make sense to form a peer review committee that performs all the reviews on everyone in the group, including the members of the committee itself. Each member of the committee should have a specified term, such as one or two years. It might not make sense for some groups, especially ones with less than 10 hospitalists, to have a formal committee. In that case, every member of the group could serve as a reviewer for all other doctors except themselves.
The group should hold formal peer review sessions quarterly or semi-annually. The group for which I serve as medical director reviews about one-fourth of the doctors at a roughly two-hour meeting each quarter. Prior to each meeting, we conduct a survey (see Figure 1) using a free Web-based tool to collect opinions about the doctors under review. We use SurveyMonkey.com, though there are many other options. The tool makes it easy to send reminders to get everyone to complete the survey and to collect and analyze the results. At the beginning of the meeting, the medical director of the practice reviews the results with the doctor being surveyed; they are not shared with others.
Most of the meeting time is spent assessing 10 charts for the doctor under review. Using the billing system, we select patients the doctor saw for many consecutive days. We want to avoid pulling charts at random only to find that the doctor only made one visit and there isn’t much to review. We assess a number of measures:
- Was VTE prophylaxis addressed appropriately?
- Was the referring doctor CC’d in the dictated reports?
- Did the doctor choose the appropriate CPT code for each visit?
- Was there a good plan for transition of care at discharge?
The doctor is provided a summary of all the findings of the peer review session, and a copy is kept on file. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson/Flores Associates, a national hospitalist practice management consulting firm. He also is part of the faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
As far as I can tell, few hospitalist groups conduct any sort of formal peer review. Most seem to rely on the hospital’s medical staff peer review to encourage quality of care and address shortcomings; the review is often coupled with a salary incentive paid for good performance on certain quality metrics. While these reviews are of some value, I think they are pretty blunt instruments. Every hospitalist practice should think about developing a more robust system of peer review for their group.
Review Categories
Assessment of each provider’s individual performance, whether they are an MD, nurse practitioner, or physician assistant, can be divided into three general categories. The first is the traditional “human resources” category of performance, which includes whether the person gets along well with others in the practice as well as other hospital staff, patients, and families. Does the person arrive at work when scheduled, manage time effectively, and work efficiently? Do nurses and other hospital staff have compliments or complaints about this doctor?
The second category of performance can encompass the hospitalist’s business and operational contributions to the practice. Do they document, code, and bill visits correctly? Do they attend and participate in meetings and serve on one or more hospital committees?
The third category assesses measurable quality of care. This could include an assessment of mortality, readmission rate, performance on such quality metrics as core measures, and performance on selected initiatives (e.g., appropriate VTE prophylaxis). Aggregate data for these measures can be difficult to attribute to a single hospitalist, so this may require a review of individual charts instead of relying on reports generated by the hospital’s data systems.
A number of metrics might apply to more than one of the three categories. For example, documenting accurate medicine reconciliation can be thought of as both a quality issue (good for patients) and a business issue (e.g., your hospital might provide a financial reward to your group for good performance). Ensuring the referring physician is “CC’d” on all dictated reports is both a quality and business issue. It really doesn’t matter which category you put these in.
The categories I have listed, and the sample items in each, are intended as examples. You should think about the unique attributes of your practice and its current priorities in order to develop the best internal peer review system for your group. You probably will want to change metrics periodically. For example, you may choose to focus on VTE prophylaxis for now, but at some point it may make sense to replace it with a new metric, such as glycemic control.
Manage the Review
There is no single right approach to conducting your own peer review. Just make sure that the process is fair and meaningful for all involved. The process probably will be more valuable if most of the data on each hospitalist can be reviewed by the whole group, or at least by a designated peer review committee. The main exceptions to such transparency are issues in the first human resources category. If a nurse or another hospitalist has specific criticisms of one hospitalist, it is best not to share that information with the whole group. But it should be fine for everyone in the group to know who is best and worst at things like documenting and coding visits or ordering VTE prophylaxis when needed. Beyond these general principles, the specific process your group uses for peer review can take many forms.
It may make sense to form a peer review committee that performs all the reviews on everyone in the group, including the members of the committee itself. Each member of the committee should have a specified term, such as one or two years. It might not make sense for some groups, especially ones with less than 10 hospitalists, to have a formal committee. In that case, every member of the group could serve as a reviewer for all other doctors except themselves.
The group should hold formal peer review sessions quarterly or semi-annually. The group for which I serve as medical director reviews about one-fourth of the doctors at a roughly two-hour meeting each quarter. Prior to each meeting, we conduct a survey (see Figure 1) using a free Web-based tool to collect opinions about the doctors under review. We use SurveyMonkey.com, though there are many other options. The tool makes it easy to send reminders to get everyone to complete the survey and to collect and analyze the results. At the beginning of the meeting, the medical director of the practice reviews the results with the doctor being surveyed; they are not shared with others.
Most of the meeting time is spent assessing 10 charts for the doctor under review. Using the billing system, we select patients the doctor saw for many consecutive days. We want to avoid pulling charts at random only to find that the doctor only made one visit and there isn’t much to review. We assess a number of measures:
- Was VTE prophylaxis addressed appropriately?
- Was the referring doctor CC’d in the dictated reports?
- Did the doctor choose the appropriate CPT code for each visit?
- Was there a good plan for transition of care at discharge?
The doctor is provided a summary of all the findings of the peer review session, and a copy is kept on file. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson/Flores Associates, a national hospitalist practice management consulting firm. He also is part of the faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
As far as I can tell, few hospitalist groups conduct any sort of formal peer review. Most seem to rely on the hospital’s medical staff peer review to encourage quality of care and address shortcomings; the review is often coupled with a salary incentive paid for good performance on certain quality metrics. While these reviews are of some value, I think they are pretty blunt instruments. Every hospitalist practice should think about developing a more robust system of peer review for their group.
Review Categories
Assessment of each provider’s individual performance, whether they are an MD, nurse practitioner, or physician assistant, can be divided into three general categories. The first is the traditional “human resources” category of performance, which includes whether the person gets along well with others in the practice as well as other hospital staff, patients, and families. Does the person arrive at work when scheduled, manage time effectively, and work efficiently? Do nurses and other hospital staff have compliments or complaints about this doctor?
The second category of performance can encompass the hospitalist’s business and operational contributions to the practice. Do they document, code, and bill visits correctly? Do they attend and participate in meetings and serve on one or more hospital committees?
The third category assesses measurable quality of care. This could include an assessment of mortality, readmission rate, performance on such quality metrics as core measures, and performance on selected initiatives (e.g., appropriate VTE prophylaxis). Aggregate data for these measures can be difficult to attribute to a single hospitalist, so this may require a review of individual charts instead of relying on reports generated by the hospital’s data systems.
A number of metrics might apply to more than one of the three categories. For example, documenting accurate medicine reconciliation can be thought of as both a quality issue (good for patients) and a business issue (e.g., your hospital might provide a financial reward to your group for good performance). Ensuring the referring physician is “CC’d” on all dictated reports is both a quality and business issue. It really doesn’t matter which category you put these in.
The categories I have listed, and the sample items in each, are intended as examples. You should think about the unique attributes of your practice and its current priorities in order to develop the best internal peer review system for your group. You probably will want to change metrics periodically. For example, you may choose to focus on VTE prophylaxis for now, but at some point it may make sense to replace it with a new metric, such as glycemic control.
Manage the Review
There is no single right approach to conducting your own peer review. Just make sure that the process is fair and meaningful for all involved. The process probably will be more valuable if most of the data on each hospitalist can be reviewed by the whole group, or at least by a designated peer review committee. The main exceptions to such transparency are issues in the first human resources category. If a nurse or another hospitalist has specific criticisms of one hospitalist, it is best not to share that information with the whole group. But it should be fine for everyone in the group to know who is best and worst at things like documenting and coding visits or ordering VTE prophylaxis when needed. Beyond these general principles, the specific process your group uses for peer review can take many forms.
It may make sense to form a peer review committee that performs all the reviews on everyone in the group, including the members of the committee itself. Each member of the committee should have a specified term, such as one or two years. It might not make sense for some groups, especially ones with less than 10 hospitalists, to have a formal committee. In that case, every member of the group could serve as a reviewer for all other doctors except themselves.
The group should hold formal peer review sessions quarterly or semi-annually. The group for which I serve as medical director reviews about one-fourth of the doctors at a roughly two-hour meeting each quarter. Prior to each meeting, we conduct a survey (see Figure 1) using a free Web-based tool to collect opinions about the doctors under review. We use SurveyMonkey.com, though there are many other options. The tool makes it easy to send reminders to get everyone to complete the survey and to collect and analyze the results. At the beginning of the meeting, the medical director of the practice reviews the results with the doctor being surveyed; they are not shared with others.
Most of the meeting time is spent assessing 10 charts for the doctor under review. Using the billing system, we select patients the doctor saw for many consecutive days. We want to avoid pulling charts at random only to find that the doctor only made one visit and there isn’t much to review. We assess a number of measures:
- Was VTE prophylaxis addressed appropriately?
- Was the referring doctor CC’d in the dictated reports?
- Did the doctor choose the appropriate CPT code for each visit?
- Was there a good plan for transition of care at discharge?
The doctor is provided a summary of all the findings of the peer review session, and a copy is kept on file. TH
Dr. Nelson has been a practicing hospitalist since 1988 and is co-founder and past president of SHM. He is a principal in Nelson/Flores Associates, a national hospitalist practice management consulting firm. He also is part of the faculty for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. This column represents his views and is not intended to reflect an official position of SHM.
Clarion Call
A specialty without a disease: It was a major hangup for the field of hospital medicine in the early days. We’d hurdled many of the traditional barriers to specialty status—research fellowships, textbooks, an active society, a growing body of research, and thousands of practitioners focusing their practice on the hospital setting. However, despite several examples of site-defined specialties like emergency and critical-care medicine, cynics clung to the time-honored need for a specialty to own an organ, or at least a disease.
Early SHM efforts made a strong push to make VTE our disease—both its treatment and, more importantly, its prevention. It was a laudable effort, one that has saved many thousands of lives and limbs. It made sense for hospitalists to tackle VTE; it’s an incapacitating disease that affects many, is largely preventable, and had no strong inpatient advocate. While hematologists were the obvious “owner” of this disease, they were neither available nor able to redesign the inpatient systems of care necessary to thwart this illness. Hospitalists, invested in this issue by consequence of direct care of many at-risk patients and through commitment to improving hospital systems, came to own VTE prevention.
The Next Frontier: Stroke
It is time hospitalists apply this experience to the management of an even more incapacitating, largely preventable disease looking for an inpatient steward: stroke. A stroke occurs in the U.S. every 40 seconds, with disabling sequelae that often are avoidable with rapid detection and treatment. This is especially true when we are given the clarion call of a transient ischemic attack (TIA). The problem is that most American hospitals are not kingpins of efficiency—the key ingredient of the processes necessary to improve stroke outcomes. Furthermore, while our neurologic colleagues are the obvious group to lead the deployment of stroke QI programs, there just aren’t enough of them to do so.
Hospitalists provide a significant amount of neurologic care. One study notes that TIA and stroke were among the most commonly cared-for diseases by hospitalists.1 This places us in a prime position to take the lead and own this disease in collaboration with our neurology colleagues. Just as with reliable VTE prevention, the key to effective stroke care requires effective systems engineering in conjunction with disease-specific expertise.
Rapid Care for Acute Medical Crisis
EDs are equipped with stroke pathways to efficiently evaluate, triage, scan, and intervene for patients who present with stroke symptoms. While most hospitals have a long way to go to perfect these systems, some hospitals—mostly in large, urban centers—have achieved the appropriate level of ED efficiency. These hospitals are recognized by The Joint Commission accreditation as stroke centers. As a result, patients who present to these hospitals with stroke symptoms often receive thrombolytics—a disease- and life-altering therapy—within the appropriate, but very limited, time window for benefit.
But what happens if that same patient develops those same signs and symptoms in the hospital? Will they get the same level of efficient evaluation, triage, scanning, and intervention that occurs in the ED? This is more than an academic question. Fifteen percent of all strokes are heralded by transient neurologic deficits, so many of the estimated 300,000 annual TIA patients are admitted to the hospital. What’s the reason for the admission? To facilitate the diagnostic workup, monitor for stroke symptoms and apply timely interventions should this occur.
But are we equipped to provide this kind of timely care in the hospital?
Case Study
Let’s take a 70-year-old diabetic male who presents with 45 minutes of aphasia and right-side arm weakness that resolve prior to presentation to the ED. If the patient is hypertensive on admission, the ABCD tool would suggest that he has a risk of stroke that approximates 20% in 90 days.2 Importantly, nearly half of that risk is in the first 48 hours. In other words, he has about a 10% risk of having a stroke in the next two days. Thus, we rightly admit him for monitoring in order to react quickly to any new signs and symptoms.
The problem is that most hospitals don’t have a system to efficiently manage patients who develop new stroke symptoms in the hospital. Does your hospital have an inpatient stroke pathway? That is, for a patient who has stroke onset while already in the hospital:
- Are the nurses aware of stroke signs and symptoms?
- Do nurses have a phone number to call to initiate an evaluation?
- Is there a team with stroke expertise immediately available to respond to those calls?
- Is there a priority path to get the patient promptly transported to the CT scanner for brain imaging with immediate radiology interpretation?
- How fast can thrombolytics be delivered, and is there an inpatient neurologist available 24/7 to assist?
The goal is 25 minutes from first recognition of symptoms to CT scan, and 60 minutes to complete evaluation and commencement of treatment. What percentage of your inpatient units could meet that goal? Could they do it any day of the week, at any time of the day or night?
ED systems had to be developed and implemented to ensure that appropriate candidates receive thrombolytic therapy in a timely manner. If those systems are not in place outside your hospital’s ED, then inpatient stroke cases are likely to miss the window of opportunity for thrombolytics. Ironically, they might have been better off going home and coming to the ED with any new symptoms. As a hospitalist, that is a sobering thought.
Hospitalist Ownership
It takes more than hospitalists being on-site to improve stroke outcomes. Processes need to be sharpened, roles defined, and outcomes monitored and acted upon to further sharpen the process. All of this plays to the strengths of hospitalists and should be undertaken with the vigor afforded to VTE prevention. This will take recognition that there’s a problem with the system, a dedication of resources, and a commitment to relentlessly work to improve and streamline the processes of stroke care.
In other words, it takes ownership. TH
Dr. Glasheen is associate professor of medicine and director of the hospital medicine group and hospitalist training program at the University of Colorado Denver. Ethan Cumbler, MD, contributed to this article. Dr. Cumbler is assistant professor of medicine at UC Denver and a member of the university’s Hospital Stroke Council.
This column represents the opinions of the author and is not intended to reflect an official position of SHM.
References
- Glasheen JJ, Epstein KR, Siegal E, Kutner J, Prochazka AV. The spectrum of community-based hospitalist practice: a call to tailor internal medicine residency training. Arch Intern Med. 2007;167(7):727-728.
- Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369(9558):283-292.
A specialty without a disease: It was a major hangup for the field of hospital medicine in the early days. We’d hurdled many of the traditional barriers to specialty status—research fellowships, textbooks, an active society, a growing body of research, and thousands of practitioners focusing their practice on the hospital setting. However, despite several examples of site-defined specialties like emergency and critical-care medicine, cynics clung to the time-honored need for a specialty to own an organ, or at least a disease.
Early SHM efforts made a strong push to make VTE our disease—both its treatment and, more importantly, its prevention. It was a laudable effort, one that has saved many thousands of lives and limbs. It made sense for hospitalists to tackle VTE; it’s an incapacitating disease that affects many, is largely preventable, and had no strong inpatient advocate. While hematologists were the obvious “owner” of this disease, they were neither available nor able to redesign the inpatient systems of care necessary to thwart this illness. Hospitalists, invested in this issue by consequence of direct care of many at-risk patients and through commitment to improving hospital systems, came to own VTE prevention.
The Next Frontier: Stroke
It is time hospitalists apply this experience to the management of an even more incapacitating, largely preventable disease looking for an inpatient steward: stroke. A stroke occurs in the U.S. every 40 seconds, with disabling sequelae that often are avoidable with rapid detection and treatment. This is especially true when we are given the clarion call of a transient ischemic attack (TIA). The problem is that most American hospitals are not kingpins of efficiency—the key ingredient of the processes necessary to improve stroke outcomes. Furthermore, while our neurologic colleagues are the obvious group to lead the deployment of stroke QI programs, there just aren’t enough of them to do so.
Hospitalists provide a significant amount of neurologic care. One study notes that TIA and stroke were among the most commonly cared-for diseases by hospitalists.1 This places us in a prime position to take the lead and own this disease in collaboration with our neurology colleagues. Just as with reliable VTE prevention, the key to effective stroke care requires effective systems engineering in conjunction with disease-specific expertise.
Rapid Care for Acute Medical Crisis
EDs are equipped with stroke pathways to efficiently evaluate, triage, scan, and intervene for patients who present with stroke symptoms. While most hospitals have a long way to go to perfect these systems, some hospitals—mostly in large, urban centers—have achieved the appropriate level of ED efficiency. These hospitals are recognized by The Joint Commission accreditation as stroke centers. As a result, patients who present to these hospitals with stroke symptoms often receive thrombolytics—a disease- and life-altering therapy—within the appropriate, but very limited, time window for benefit.
But what happens if that same patient develops those same signs and symptoms in the hospital? Will they get the same level of efficient evaluation, triage, scanning, and intervention that occurs in the ED? This is more than an academic question. Fifteen percent of all strokes are heralded by transient neurologic deficits, so many of the estimated 300,000 annual TIA patients are admitted to the hospital. What’s the reason for the admission? To facilitate the diagnostic workup, monitor for stroke symptoms and apply timely interventions should this occur.
But are we equipped to provide this kind of timely care in the hospital?
Case Study
Let’s take a 70-year-old diabetic male who presents with 45 minutes of aphasia and right-side arm weakness that resolve prior to presentation to the ED. If the patient is hypertensive on admission, the ABCD tool would suggest that he has a risk of stroke that approximates 20% in 90 days.2 Importantly, nearly half of that risk is in the first 48 hours. In other words, he has about a 10% risk of having a stroke in the next two days. Thus, we rightly admit him for monitoring in order to react quickly to any new signs and symptoms.
The problem is that most hospitals don’t have a system to efficiently manage patients who develop new stroke symptoms in the hospital. Does your hospital have an inpatient stroke pathway? That is, for a patient who has stroke onset while already in the hospital:
- Are the nurses aware of stroke signs and symptoms?
- Do nurses have a phone number to call to initiate an evaluation?
- Is there a team with stroke expertise immediately available to respond to those calls?
- Is there a priority path to get the patient promptly transported to the CT scanner for brain imaging with immediate radiology interpretation?
- How fast can thrombolytics be delivered, and is there an inpatient neurologist available 24/7 to assist?
The goal is 25 minutes from first recognition of symptoms to CT scan, and 60 minutes to complete evaluation and commencement of treatment. What percentage of your inpatient units could meet that goal? Could they do it any day of the week, at any time of the day or night?
ED systems had to be developed and implemented to ensure that appropriate candidates receive thrombolytic therapy in a timely manner. If those systems are not in place outside your hospital’s ED, then inpatient stroke cases are likely to miss the window of opportunity for thrombolytics. Ironically, they might have been better off going home and coming to the ED with any new symptoms. As a hospitalist, that is a sobering thought.
Hospitalist Ownership
It takes more than hospitalists being on-site to improve stroke outcomes. Processes need to be sharpened, roles defined, and outcomes monitored and acted upon to further sharpen the process. All of this plays to the strengths of hospitalists and should be undertaken with the vigor afforded to VTE prevention. This will take recognition that there’s a problem with the system, a dedication of resources, and a commitment to relentlessly work to improve and streamline the processes of stroke care.
In other words, it takes ownership. TH
Dr. Glasheen is associate professor of medicine and director of the hospital medicine group and hospitalist training program at the University of Colorado Denver. Ethan Cumbler, MD, contributed to this article. Dr. Cumbler is assistant professor of medicine at UC Denver and a member of the university’s Hospital Stroke Council.
This column represents the opinions of the author and is not intended to reflect an official position of SHM.
References
- Glasheen JJ, Epstein KR, Siegal E, Kutner J, Prochazka AV. The spectrum of community-based hospitalist practice: a call to tailor internal medicine residency training. Arch Intern Med. 2007;167(7):727-728.
- Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369(9558):283-292.
A specialty without a disease: It was a major hangup for the field of hospital medicine in the early days. We’d hurdled many of the traditional barriers to specialty status—research fellowships, textbooks, an active society, a growing body of research, and thousands of practitioners focusing their practice on the hospital setting. However, despite several examples of site-defined specialties like emergency and critical-care medicine, cynics clung to the time-honored need for a specialty to own an organ, or at least a disease.
Early SHM efforts made a strong push to make VTE our disease—both its treatment and, more importantly, its prevention. It was a laudable effort, one that has saved many thousands of lives and limbs. It made sense for hospitalists to tackle VTE; it’s an incapacitating disease that affects many, is largely preventable, and had no strong inpatient advocate. While hematologists were the obvious “owner” of this disease, they were neither available nor able to redesign the inpatient systems of care necessary to thwart this illness. Hospitalists, invested in this issue by consequence of direct care of many at-risk patients and through commitment to improving hospital systems, came to own VTE prevention.
The Next Frontier: Stroke
It is time hospitalists apply this experience to the management of an even more incapacitating, largely preventable disease looking for an inpatient steward: stroke. A stroke occurs in the U.S. every 40 seconds, with disabling sequelae that often are avoidable with rapid detection and treatment. This is especially true when we are given the clarion call of a transient ischemic attack (TIA). The problem is that most American hospitals are not kingpins of efficiency—the key ingredient of the processes necessary to improve stroke outcomes. Furthermore, while our neurologic colleagues are the obvious group to lead the deployment of stroke QI programs, there just aren’t enough of them to do so.
Hospitalists provide a significant amount of neurologic care. One study notes that TIA and stroke were among the most commonly cared-for diseases by hospitalists.1 This places us in a prime position to take the lead and own this disease in collaboration with our neurology colleagues. Just as with reliable VTE prevention, the key to effective stroke care requires effective systems engineering in conjunction with disease-specific expertise.
Rapid Care for Acute Medical Crisis
EDs are equipped with stroke pathways to efficiently evaluate, triage, scan, and intervene for patients who present with stroke symptoms. While most hospitals have a long way to go to perfect these systems, some hospitals—mostly in large, urban centers—have achieved the appropriate level of ED efficiency. These hospitals are recognized by The Joint Commission accreditation as stroke centers. As a result, patients who present to these hospitals with stroke symptoms often receive thrombolytics—a disease- and life-altering therapy—within the appropriate, but very limited, time window for benefit.
But what happens if that same patient develops those same signs and symptoms in the hospital? Will they get the same level of efficient evaluation, triage, scanning, and intervention that occurs in the ED? This is more than an academic question. Fifteen percent of all strokes are heralded by transient neurologic deficits, so many of the estimated 300,000 annual TIA patients are admitted to the hospital. What’s the reason for the admission? To facilitate the diagnostic workup, monitor for stroke symptoms and apply timely interventions should this occur.
But are we equipped to provide this kind of timely care in the hospital?
Case Study
Let’s take a 70-year-old diabetic male who presents with 45 minutes of aphasia and right-side arm weakness that resolve prior to presentation to the ED. If the patient is hypertensive on admission, the ABCD tool would suggest that he has a risk of stroke that approximates 20% in 90 days.2 Importantly, nearly half of that risk is in the first 48 hours. In other words, he has about a 10% risk of having a stroke in the next two days. Thus, we rightly admit him for monitoring in order to react quickly to any new signs and symptoms.
The problem is that most hospitals don’t have a system to efficiently manage patients who develop new stroke symptoms in the hospital. Does your hospital have an inpatient stroke pathway? That is, for a patient who has stroke onset while already in the hospital:
- Are the nurses aware of stroke signs and symptoms?
- Do nurses have a phone number to call to initiate an evaluation?
- Is there a team with stroke expertise immediately available to respond to those calls?
- Is there a priority path to get the patient promptly transported to the CT scanner for brain imaging with immediate radiology interpretation?
- How fast can thrombolytics be delivered, and is there an inpatient neurologist available 24/7 to assist?
The goal is 25 minutes from first recognition of symptoms to CT scan, and 60 minutes to complete evaluation and commencement of treatment. What percentage of your inpatient units could meet that goal? Could they do it any day of the week, at any time of the day or night?
ED systems had to be developed and implemented to ensure that appropriate candidates receive thrombolytic therapy in a timely manner. If those systems are not in place outside your hospital’s ED, then inpatient stroke cases are likely to miss the window of opportunity for thrombolytics. Ironically, they might have been better off going home and coming to the ED with any new symptoms. As a hospitalist, that is a sobering thought.
Hospitalist Ownership
It takes more than hospitalists being on-site to improve stroke outcomes. Processes need to be sharpened, roles defined, and outcomes monitored and acted upon to further sharpen the process. All of this plays to the strengths of hospitalists and should be undertaken with the vigor afforded to VTE prevention. This will take recognition that there’s a problem with the system, a dedication of resources, and a commitment to relentlessly work to improve and streamline the processes of stroke care.
In other words, it takes ownership. TH
Dr. Glasheen is associate professor of medicine and director of the hospital medicine group and hospitalist training program at the University of Colorado Denver. Ethan Cumbler, MD, contributed to this article. Dr. Cumbler is assistant professor of medicine at UC Denver and a member of the university’s Hospital Stroke Council.
This column represents the opinions of the author and is not intended to reflect an official position of SHM.
References
- Glasheen JJ, Epstein KR, Siegal E, Kutner J, Prochazka AV. The spectrum of community-based hospitalist practice: a call to tailor internal medicine residency training. Arch Intern Med. 2007;167(7):727-728.
- Johnston SC, Rothwell PM, Nguyen-Huynh MN, et al. Validation and refinement of scores to predict very early stroke risk after transient ischaemic attack. Lancet. 2007;369(9558):283-292.
Bundling Bedlam
Even if you receive your salary as an employee of your hospital or hospitalist group, you should keep a close eye on discussions taking place in Washington about reshaping the way hospital care is paid for. It seems that every 10 to 20 years, a seismic tremor starts on Capitol Hill and fundamentally shakes up the way healthcare is funded. It starts with Medicare, then quickly is adopted by private insurers; it not only changes the distribution of dollars, but the new incentives also drive the way medicine is practiced.
In the 1960s, change began with President Johnson and the development of Medicare and Medicaid. It was the first time specific populations—seniors and the poor—were “entitled” to healthcare coverage. In essence, Johnson created the largest “insurance company” in the country, and it became the tail that wagged the dog.
In the 1970s, President Nixon pushed through support for HMOs, and capitation and managed care spread well beyond the Kaisers of the world. This system incentivized controlling costs, because the total amount was capped, while maintaining an acceptable level of quality. For the first time, doing more did not generate more money.
In the 1980s, diagnosis-related groups (DRGs) changed Medicare payments to hospitals from cost-plus billings to a bundled fee for an episode of care. This motivated hospitals to work with their physicians—sometimes driven by protocols and case managers—to efficiently manage resources and length of stay (LOS). Between capitation, case rates, and DRGs, hospitals have had to refashion themselves to be leaner and more efficient.
Today, with national thought leaders like John Wennberg and Elliot Fisher at Dartmouth and Brent James at Intermountain revealing the many variations in the way healthcare is practiced—and throwing around statistics like “40% of healthcare is wasteful”—it is no wonder that as President Obama and Congress look to add 47 million uninsured persons to the system and try to reduce variation and increase accountabilities, there is every indication that radical changes will be made to the payment system.
One of these newfangled approaches is the bundling of payment for an episode of care to include both the facility charges (e.g., hospital care) and the professional charges (e.g., physician care). Bundling can be a good thing or a worrisome approach, depending on where you sit in this dialogue and how bundling is actually implemented.
Background on Bundling
The motivation of the government—and, by extension, all insurers—is that efforts to control what they pay per unit for a visit, a procedure, or even an entire hospitalization has not curbed costs or led to a satisfactory level of performance. With respect to hospitalized patients, Obama has stated that he wants to eliminate waste by reducing unnecessary readmissions to the tune of $6.8 billion annually. Furthermore, Medicare officials want to look for strategies that either keep people out of the ED post-discharge or at least eliminate Medicare’s need to pay for this care, which they feel is unnecessary and avoidable.
By bundling payment for a specific admission (e.g., decompensated heart failure or pneumonia) and including the facility and professional-care fees, both during hospitalization and for a period of time (e.g., 30 days post-discharge) and providing incentives for best performance, the insurer (i.e., Medicare) can hand off responsibility to the hospitals and the doctors to figure it all out. There is nothing like the accountability of knowing “this is all you are going to get,” or “if you want more, you have to meet these standards,” to motivate professionals to reshape their system to improve their discharge process, engage the outpatient physicians, and do the job right the first time. This can play to HM’s strengths, and SHM already has started developing and implementing change in the discharge process through Project BOOST (Better Outcomes for Older Adults through Safe Transitions, www.hospitalmedicine.org/boost).
Potential Problems
One key concern is not knowing who—or what—will control the dollars once Medicare sets the bundled payment. Right now, hospitals receive the DRG payment and physicians bill for their own professional services. In the future, will all the money flow to the hospital? How will these dollars be distributed? Who determines who will be awarded performance bonuses?
In California and other states with significant managed-care populations and large medical groups, there is real-life experience with setting up efficient physician-hospital organizations (PHOs) to solve these issues. Some take the form of independent physician associations (IPAs), which represent the physicians in PHOs. There is no reason PHOs cannot be developed to administrate these bundled funds, and hospitalists, who are seeing an increasing number of hospitalized patients on medicine and surgery, should be key leaders in such PHO arrangements.
But HM is not a monolith in this discussion. The diversity in how HM groups are organized, their relationship with their hospitals, and how hospitalists or their groups receive funding can, and will, influence the group’s perspective on this issue. Hospitalist groups that are independent from their hospitals, or those that rely on referrals from primary-care physicians (PCPs) or the ED, might be justifiably concerned about all of “their” money having to flow through the hospital. Hospitalists who are employed by a hospital might be concerned that they will need to develop new metrics to justify their salaries and bonuses. HM groups that contract with the hospital might be concerned that a change in the flow of funding from Medicare to the hospital might make their contractual arrangements more difficult.
For those who battle with hospital administration over hospital support of their HM group, they might find bundling alleviates the need for the current use of Part A dollars to support hospitalists, because the new bundling of Part A (current payments for hospital facility charges) and Part B (current payment for physicians’ professional services) can allow for a more professional discussion, based on the value hospitalists bring. The need for subsidies or support could diminish or vanish.
Change Is Coming
No matter your perspective or viewpoint, one reality is coming into focus: This president and this Congress will make sweeping changes, and it appears from our conversations with Sen. Max Baucus (D-Mont.), chair of the powerful Senate Finance Committee (see “Medicine’s Change Agent,” May 2009, p. 18), that bundling and value-based purchasing will be part of healthcare reform.
With this in mind, SHM’s Public Policy Committee is actively engaged in trying to shape bundling in a way that fits emerging changes in the care of hospitalized patients. We want a system that works for the way healthcare will be practiced in the future, not a Band-Aid on the system of the past. This is very important stuff. Hospitalists will be affected by reform because so many of our patients are on Medicare and our compensation is generated by patient care in the hospital.
SHM has created an easy-to-use, Web-based system to send a message to members of Congress through a partnership with Capwiz. Visit www.hospitalmedicine.org/beheard to get started.
While the uncertainty of healthcare reform and, more specifically, payment reform is at times frightening, mainly because it is so sweeping and at this point so undefined, HM has been forged in the cauldron of change and ambiguity. Hospitalists are positioned as well as any health professionals to seize the opportunities that a new system will provide. And SHM will do its part to help shape the new reality and assist our members in creating successful strategies in this new environment. TH
Dr. Wellikson is CEO of SHM.
Even if you receive your salary as an employee of your hospital or hospitalist group, you should keep a close eye on discussions taking place in Washington about reshaping the way hospital care is paid for. It seems that every 10 to 20 years, a seismic tremor starts on Capitol Hill and fundamentally shakes up the way healthcare is funded. It starts with Medicare, then quickly is adopted by private insurers; it not only changes the distribution of dollars, but the new incentives also drive the way medicine is practiced.
In the 1960s, change began with President Johnson and the development of Medicare and Medicaid. It was the first time specific populations—seniors and the poor—were “entitled” to healthcare coverage. In essence, Johnson created the largest “insurance company” in the country, and it became the tail that wagged the dog.
In the 1970s, President Nixon pushed through support for HMOs, and capitation and managed care spread well beyond the Kaisers of the world. This system incentivized controlling costs, because the total amount was capped, while maintaining an acceptable level of quality. For the first time, doing more did not generate more money.
In the 1980s, diagnosis-related groups (DRGs) changed Medicare payments to hospitals from cost-plus billings to a bundled fee for an episode of care. This motivated hospitals to work with their physicians—sometimes driven by protocols and case managers—to efficiently manage resources and length of stay (LOS). Between capitation, case rates, and DRGs, hospitals have had to refashion themselves to be leaner and more efficient.
Today, with national thought leaders like John Wennberg and Elliot Fisher at Dartmouth and Brent James at Intermountain revealing the many variations in the way healthcare is practiced—and throwing around statistics like “40% of healthcare is wasteful”—it is no wonder that as President Obama and Congress look to add 47 million uninsured persons to the system and try to reduce variation and increase accountabilities, there is every indication that radical changes will be made to the payment system.
One of these newfangled approaches is the bundling of payment for an episode of care to include both the facility charges (e.g., hospital care) and the professional charges (e.g., physician care). Bundling can be a good thing or a worrisome approach, depending on where you sit in this dialogue and how bundling is actually implemented.
Background on Bundling
The motivation of the government—and, by extension, all insurers—is that efforts to control what they pay per unit for a visit, a procedure, or even an entire hospitalization has not curbed costs or led to a satisfactory level of performance. With respect to hospitalized patients, Obama has stated that he wants to eliminate waste by reducing unnecessary readmissions to the tune of $6.8 billion annually. Furthermore, Medicare officials want to look for strategies that either keep people out of the ED post-discharge or at least eliminate Medicare’s need to pay for this care, which they feel is unnecessary and avoidable.
By bundling payment for a specific admission (e.g., decompensated heart failure or pneumonia) and including the facility and professional-care fees, both during hospitalization and for a period of time (e.g., 30 days post-discharge) and providing incentives for best performance, the insurer (i.e., Medicare) can hand off responsibility to the hospitals and the doctors to figure it all out. There is nothing like the accountability of knowing “this is all you are going to get,” or “if you want more, you have to meet these standards,” to motivate professionals to reshape their system to improve their discharge process, engage the outpatient physicians, and do the job right the first time. This can play to HM’s strengths, and SHM already has started developing and implementing change in the discharge process through Project BOOST (Better Outcomes for Older Adults through Safe Transitions, www.hospitalmedicine.org/boost).
Potential Problems
One key concern is not knowing who—or what—will control the dollars once Medicare sets the bundled payment. Right now, hospitals receive the DRG payment and physicians bill for their own professional services. In the future, will all the money flow to the hospital? How will these dollars be distributed? Who determines who will be awarded performance bonuses?
In California and other states with significant managed-care populations and large medical groups, there is real-life experience with setting up efficient physician-hospital organizations (PHOs) to solve these issues. Some take the form of independent physician associations (IPAs), which represent the physicians in PHOs. There is no reason PHOs cannot be developed to administrate these bundled funds, and hospitalists, who are seeing an increasing number of hospitalized patients on medicine and surgery, should be key leaders in such PHO arrangements.
But HM is not a monolith in this discussion. The diversity in how HM groups are organized, their relationship with their hospitals, and how hospitalists or their groups receive funding can, and will, influence the group’s perspective on this issue. Hospitalist groups that are independent from their hospitals, or those that rely on referrals from primary-care physicians (PCPs) or the ED, might be justifiably concerned about all of “their” money having to flow through the hospital. Hospitalists who are employed by a hospital might be concerned that they will need to develop new metrics to justify their salaries and bonuses. HM groups that contract with the hospital might be concerned that a change in the flow of funding from Medicare to the hospital might make their contractual arrangements more difficult.
For those who battle with hospital administration over hospital support of their HM group, they might find bundling alleviates the need for the current use of Part A dollars to support hospitalists, because the new bundling of Part A (current payments for hospital facility charges) and Part B (current payment for physicians’ professional services) can allow for a more professional discussion, based on the value hospitalists bring. The need for subsidies or support could diminish or vanish.
Change Is Coming
No matter your perspective or viewpoint, one reality is coming into focus: This president and this Congress will make sweeping changes, and it appears from our conversations with Sen. Max Baucus (D-Mont.), chair of the powerful Senate Finance Committee (see “Medicine’s Change Agent,” May 2009, p. 18), that bundling and value-based purchasing will be part of healthcare reform.
With this in mind, SHM’s Public Policy Committee is actively engaged in trying to shape bundling in a way that fits emerging changes in the care of hospitalized patients. We want a system that works for the way healthcare will be practiced in the future, not a Band-Aid on the system of the past. This is very important stuff. Hospitalists will be affected by reform because so many of our patients are on Medicare and our compensation is generated by patient care in the hospital.
SHM has created an easy-to-use, Web-based system to send a message to members of Congress through a partnership with Capwiz. Visit www.hospitalmedicine.org/beheard to get started.
While the uncertainty of healthcare reform and, more specifically, payment reform is at times frightening, mainly because it is so sweeping and at this point so undefined, HM has been forged in the cauldron of change and ambiguity. Hospitalists are positioned as well as any health professionals to seize the opportunities that a new system will provide. And SHM will do its part to help shape the new reality and assist our members in creating successful strategies in this new environment. TH
Dr. Wellikson is CEO of SHM.
Even if you receive your salary as an employee of your hospital or hospitalist group, you should keep a close eye on discussions taking place in Washington about reshaping the way hospital care is paid for. It seems that every 10 to 20 years, a seismic tremor starts on Capitol Hill and fundamentally shakes up the way healthcare is funded. It starts with Medicare, then quickly is adopted by private insurers; it not only changes the distribution of dollars, but the new incentives also drive the way medicine is practiced.
In the 1960s, change began with President Johnson and the development of Medicare and Medicaid. It was the first time specific populations—seniors and the poor—were “entitled” to healthcare coverage. In essence, Johnson created the largest “insurance company” in the country, and it became the tail that wagged the dog.
In the 1970s, President Nixon pushed through support for HMOs, and capitation and managed care spread well beyond the Kaisers of the world. This system incentivized controlling costs, because the total amount was capped, while maintaining an acceptable level of quality. For the first time, doing more did not generate more money.
In the 1980s, diagnosis-related groups (DRGs) changed Medicare payments to hospitals from cost-plus billings to a bundled fee for an episode of care. This motivated hospitals to work with their physicians—sometimes driven by protocols and case managers—to efficiently manage resources and length of stay (LOS). Between capitation, case rates, and DRGs, hospitals have had to refashion themselves to be leaner and more efficient.
Today, with national thought leaders like John Wennberg and Elliot Fisher at Dartmouth and Brent James at Intermountain revealing the many variations in the way healthcare is practiced—and throwing around statistics like “40% of healthcare is wasteful”—it is no wonder that as President Obama and Congress look to add 47 million uninsured persons to the system and try to reduce variation and increase accountabilities, there is every indication that radical changes will be made to the payment system.
One of these newfangled approaches is the bundling of payment for an episode of care to include both the facility charges (e.g., hospital care) and the professional charges (e.g., physician care). Bundling can be a good thing or a worrisome approach, depending on where you sit in this dialogue and how bundling is actually implemented.
Background on Bundling
The motivation of the government—and, by extension, all insurers—is that efforts to control what they pay per unit for a visit, a procedure, or even an entire hospitalization has not curbed costs or led to a satisfactory level of performance. With respect to hospitalized patients, Obama has stated that he wants to eliminate waste by reducing unnecessary readmissions to the tune of $6.8 billion annually. Furthermore, Medicare officials want to look for strategies that either keep people out of the ED post-discharge or at least eliminate Medicare’s need to pay for this care, which they feel is unnecessary and avoidable.
By bundling payment for a specific admission (e.g., decompensated heart failure or pneumonia) and including the facility and professional-care fees, both during hospitalization and for a period of time (e.g., 30 days post-discharge) and providing incentives for best performance, the insurer (i.e., Medicare) can hand off responsibility to the hospitals and the doctors to figure it all out. There is nothing like the accountability of knowing “this is all you are going to get,” or “if you want more, you have to meet these standards,” to motivate professionals to reshape their system to improve their discharge process, engage the outpatient physicians, and do the job right the first time. This can play to HM’s strengths, and SHM already has started developing and implementing change in the discharge process through Project BOOST (Better Outcomes for Older Adults through Safe Transitions, www.hospitalmedicine.org/boost).
Potential Problems
One key concern is not knowing who—or what—will control the dollars once Medicare sets the bundled payment. Right now, hospitals receive the DRG payment and physicians bill for their own professional services. In the future, will all the money flow to the hospital? How will these dollars be distributed? Who determines who will be awarded performance bonuses?
In California and other states with significant managed-care populations and large medical groups, there is real-life experience with setting up efficient physician-hospital organizations (PHOs) to solve these issues. Some take the form of independent physician associations (IPAs), which represent the physicians in PHOs. There is no reason PHOs cannot be developed to administrate these bundled funds, and hospitalists, who are seeing an increasing number of hospitalized patients on medicine and surgery, should be key leaders in such PHO arrangements.
But HM is not a monolith in this discussion. The diversity in how HM groups are organized, their relationship with their hospitals, and how hospitalists or their groups receive funding can, and will, influence the group’s perspective on this issue. Hospitalist groups that are independent from their hospitals, or those that rely on referrals from primary-care physicians (PCPs) or the ED, might be justifiably concerned about all of “their” money having to flow through the hospital. Hospitalists who are employed by a hospital might be concerned that they will need to develop new metrics to justify their salaries and bonuses. HM groups that contract with the hospital might be concerned that a change in the flow of funding from Medicare to the hospital might make their contractual arrangements more difficult.
For those who battle with hospital administration over hospital support of their HM group, they might find bundling alleviates the need for the current use of Part A dollars to support hospitalists, because the new bundling of Part A (current payments for hospital facility charges) and Part B (current payment for physicians’ professional services) can allow for a more professional discussion, based on the value hospitalists bring. The need for subsidies or support could diminish or vanish.
Change Is Coming
No matter your perspective or viewpoint, one reality is coming into focus: This president and this Congress will make sweeping changes, and it appears from our conversations with Sen. Max Baucus (D-Mont.), chair of the powerful Senate Finance Committee (see “Medicine’s Change Agent,” May 2009, p. 18), that bundling and value-based purchasing will be part of healthcare reform.
With this in mind, SHM’s Public Policy Committee is actively engaged in trying to shape bundling in a way that fits emerging changes in the care of hospitalized patients. We want a system that works for the way healthcare will be practiced in the future, not a Band-Aid on the system of the past. This is very important stuff. Hospitalists will be affected by reform because so many of our patients are on Medicare and our compensation is generated by patient care in the hospital.
SHM has created an easy-to-use, Web-based system to send a message to members of Congress through a partnership with Capwiz. Visit www.hospitalmedicine.org/beheard to get started.
While the uncertainty of healthcare reform and, more specifically, payment reform is at times frightening, mainly because it is so sweeping and at this point so undefined, HM has been forged in the cauldron of change and ambiguity. Hospitalists are positioned as well as any health professionals to seize the opportunities that a new system will provide. And SHM will do its part to help shape the new reality and assist our members in creating successful strategies in this new environment. TH
Dr. Wellikson is CEO of SHM.