User login
The Blog Rounds
2008-09 apparently was a good time to find a job in HM. The Wall Street Journal’s Health Blog cites a new report from Merritt Hawkins & Associates (download PDF) that shows from April 2008 to March 2009, 85% of searches offered signing bonuses averaging $24,850. That’s in contrast to 46% of searches in 2005-2006, when the average bonus was $14,030.
The report also included average salaries for HM, which was the third-most-requested search assignment following family medicine and general internal medicine. The average salary for HM during that time period, excluding benefits or productivity bonuses, was $201,000, according to the report. It represents a 14.8% increase since 2005-2006, when the annual average annual salary for a hospitalist was $175,000, according to the report.
Fun & Games
On his blog, Running a Hospital, Paul Levy, president and CEO of Beth Israel Deaconess Medical Center in Boston, offers physicians a game to help them better understand the value of standardized medicine. Interestingly, the game involves drawing a pig. For instructions, visit http://runningahospital.blogspot.com/2009/06/pig-part-1.html.
Work-Life Balance
In her Well blog, New York Times writer Tara Parker Pope cites a recent article from Times columnist Pauline Chen, MD, in which Dr. Chen recalls the toll her intense medical training took on her temperament and personal relationships.
Doctors responded to the post with a variety of viewpoints. Here’s what one old-timer had to say:
“I am from the era of 120-hour weeks, every second or third night on call. No ‘cap’ on the numbers of patients we admitted or carried on our service. I remember still being in the hospital at 10 p.m. after a night on call (40 hours straight). I learned how to manage the sickest patients through the entire course of their hospitalization. My residency and fellowship after medical school was seven years in duration, and I loved it. The ONLY regret I have after all these years is reading the NYT comments and blogs about all the doctor-haters and disgruntled patients who think that medicine is an easy path to riches.”
2008-09 apparently was a good time to find a job in HM. The Wall Street Journal’s Health Blog cites a new report from Merritt Hawkins & Associates (download PDF) that shows from April 2008 to March 2009, 85% of searches offered signing bonuses averaging $24,850. That’s in contrast to 46% of searches in 2005-2006, when the average bonus was $14,030.
The report also included average salaries for HM, which was the third-most-requested search assignment following family medicine and general internal medicine. The average salary for HM during that time period, excluding benefits or productivity bonuses, was $201,000, according to the report. It represents a 14.8% increase since 2005-2006, when the annual average annual salary for a hospitalist was $175,000, according to the report.
Fun & Games
On his blog, Running a Hospital, Paul Levy, president and CEO of Beth Israel Deaconess Medical Center in Boston, offers physicians a game to help them better understand the value of standardized medicine. Interestingly, the game involves drawing a pig. For instructions, visit http://runningahospital.blogspot.com/2009/06/pig-part-1.html.
Work-Life Balance
In her Well blog, New York Times writer Tara Parker Pope cites a recent article from Times columnist Pauline Chen, MD, in which Dr. Chen recalls the toll her intense medical training took on her temperament and personal relationships.
Doctors responded to the post with a variety of viewpoints. Here’s what one old-timer had to say:
“I am from the era of 120-hour weeks, every second or third night on call. No ‘cap’ on the numbers of patients we admitted or carried on our service. I remember still being in the hospital at 10 p.m. after a night on call (40 hours straight). I learned how to manage the sickest patients through the entire course of their hospitalization. My residency and fellowship after medical school was seven years in duration, and I loved it. The ONLY regret I have after all these years is reading the NYT comments and blogs about all the doctor-haters and disgruntled patients who think that medicine is an easy path to riches.”
2008-09 apparently was a good time to find a job in HM. The Wall Street Journal’s Health Blog cites a new report from Merritt Hawkins & Associates (download PDF) that shows from April 2008 to March 2009, 85% of searches offered signing bonuses averaging $24,850. That’s in contrast to 46% of searches in 2005-2006, when the average bonus was $14,030.
The report also included average salaries for HM, which was the third-most-requested search assignment following family medicine and general internal medicine. The average salary for HM during that time period, excluding benefits or productivity bonuses, was $201,000, according to the report. It represents a 14.8% increase since 2005-2006, when the annual average annual salary for a hospitalist was $175,000, according to the report.
Fun & Games
On his blog, Running a Hospital, Paul Levy, president and CEO of Beth Israel Deaconess Medical Center in Boston, offers physicians a game to help them better understand the value of standardized medicine. Interestingly, the game involves drawing a pig. For instructions, visit http://runningahospital.blogspot.com/2009/06/pig-part-1.html.
Work-Life Balance
In her Well blog, New York Times writer Tara Parker Pope cites a recent article from Times columnist Pauline Chen, MD, in which Dr. Chen recalls the toll her intense medical training took on her temperament and personal relationships.
Doctors responded to the post with a variety of viewpoints. Here’s what one old-timer had to say:
“I am from the era of 120-hour weeks, every second or third night on call. No ‘cap’ on the numbers of patients we admitted or carried on our service. I remember still being in the hospital at 10 p.m. after a night on call (40 hours straight). I learned how to manage the sickest patients through the entire course of their hospitalization. My residency and fellowship after medical school was seven years in duration, and I loved it. The ONLY regret I have after all these years is reading the NYT comments and blogs about all the doctor-haters and disgruntled patients who think that medicine is an easy path to riches.”
Ofatumumab
Dr. William Wierda discusses ofatumumab, an investigational monoclonal antibody that targets B cells and has been studied in the treatment of chronic lymphocytic leukemia. Mitchel Zoler of the Global Medical News Network (GMNN) reports from the annual congress of the European Hematology Association in Berlin.
Dr. William Wierda discusses ofatumumab, an investigational monoclonal antibody that targets B cells and has been studied in the treatment of chronic lymphocytic leukemia. Mitchel Zoler of the Global Medical News Network (GMNN) reports from the annual congress of the European Hematology Association in Berlin.
Dr. William Wierda discusses ofatumumab, an investigational monoclonal antibody that targets B cells and has been studied in the treatment of chronic lymphocytic leukemia. Mitchel Zoler of the Global Medical News Network (GMNN) reports from the annual congress of the European Hematology Association in Berlin.
A Hospitalist Is Born
The hospitalist business model has been adopted nationwide to help stem the tide of obstetrician/gynecologists who forgo delivering babies at hospitals—or providing emergency obstetric care—because of skyrocketing malpractice costs and a frustration with on-call schedules. Like the familiar HM model, a new class of OB-GYN has cropped up to work in medical centers.
Called laborists or OB hospitalists, this breed resembles hospital-based physicians: They work out of their respective institutions but don’t have private-practice patients. Hospitals like the arrangement because they have trained delivery staff in-house, and private-practice physicians don't mind giving up hospital calls because they can earn more cycling patients through their office.
The OB Hospitalist Group of Greenville, S.C., already has placed 60 OB hospitalists in six states, including Texas, California, and Florida. Group president Chris Swain, MD, hopes to place 100 by year's end, but only if he can find enough qualified candidates to manage both deliveries and OB emergencies.
"If you're going to be in a hospital 24 hours a day, you want to be able to handle all the emergencies that come in," Dr. Swain says. “You can't take time to look it up. You need to know it."
Dr. Swain's group pays for that knowledge. The average OB-GYN makes about $280,000 per year; Dr. Swain's OB hospitalists earn roughly $300,000 a year. In exchange for the higher salaries and defined work schedules, OB Hospitalist Group requires board certification, monitoring courses, and continued training. "We want our doctors to be the experts in emergency obstetrics care," Dr. Swain says. "We pay our doctors more; we expect more out of them."
The hospitalist business model has been adopted nationwide to help stem the tide of obstetrician/gynecologists who forgo delivering babies at hospitals—or providing emergency obstetric care—because of skyrocketing malpractice costs and a frustration with on-call schedules. Like the familiar HM model, a new class of OB-GYN has cropped up to work in medical centers.
Called laborists or OB hospitalists, this breed resembles hospital-based physicians: They work out of their respective institutions but don’t have private-practice patients. Hospitals like the arrangement because they have trained delivery staff in-house, and private-practice physicians don't mind giving up hospital calls because they can earn more cycling patients through their office.
The OB Hospitalist Group of Greenville, S.C., already has placed 60 OB hospitalists in six states, including Texas, California, and Florida. Group president Chris Swain, MD, hopes to place 100 by year's end, but only if he can find enough qualified candidates to manage both deliveries and OB emergencies.
"If you're going to be in a hospital 24 hours a day, you want to be able to handle all the emergencies that come in," Dr. Swain says. “You can't take time to look it up. You need to know it."
Dr. Swain's group pays for that knowledge. The average OB-GYN makes about $280,000 per year; Dr. Swain's OB hospitalists earn roughly $300,000 a year. In exchange for the higher salaries and defined work schedules, OB Hospitalist Group requires board certification, monitoring courses, and continued training. "We want our doctors to be the experts in emergency obstetrics care," Dr. Swain says. "We pay our doctors more; we expect more out of them."
The hospitalist business model has been adopted nationwide to help stem the tide of obstetrician/gynecologists who forgo delivering babies at hospitals—or providing emergency obstetric care—because of skyrocketing malpractice costs and a frustration with on-call schedules. Like the familiar HM model, a new class of OB-GYN has cropped up to work in medical centers.
Called laborists or OB hospitalists, this breed resembles hospital-based physicians: They work out of their respective institutions but don’t have private-practice patients. Hospitals like the arrangement because they have trained delivery staff in-house, and private-practice physicians don't mind giving up hospital calls because they can earn more cycling patients through their office.
The OB Hospitalist Group of Greenville, S.C., already has placed 60 OB hospitalists in six states, including Texas, California, and Florida. Group president Chris Swain, MD, hopes to place 100 by year's end, but only if he can find enough qualified candidates to manage both deliveries and OB emergencies.
"If you're going to be in a hospital 24 hours a day, you want to be able to handle all the emergencies that come in," Dr. Swain says. “You can't take time to look it up. You need to know it."
Dr. Swain's group pays for that knowledge. The average OB-GYN makes about $280,000 per year; Dr. Swain's OB hospitalists earn roughly $300,000 a year. In exchange for the higher salaries and defined work schedules, OB Hospitalist Group requires board certification, monitoring courses, and continued training. "We want our doctors to be the experts in emergency obstetrics care," Dr. Swain says. "We pay our doctors more; we expect more out of them."
In the Literature: The Latest Research You Need to Know
Clinical question: Does intensive glucose control reduce mortality at 90 days in adult ICU patients?
Background: The American Diabetic Association currently recommends tight glucose control for patients admitted to an ICU, despite conflicting evidence in the literature about the benefits of this practice.
Study design: Randomized controlled trial.
Setting: Medical and surgical ICUs at 42 hospitals in Australia, Canada, and New Zealand.
Synopsis: More than 6,000 medical and surgical ICU patients were randomly assigned to receive either intensive (target blood sugar range of 81 mg/dL to 108 mg/dL) or conventional (target blood sugar of <180 mg/dL) glucose control. Eligible patients were expected to stay at least three days in the ICU. Mortality at 90 days for the intensive treatment group was 27.5% versus 24.9% in the conventional treatment group, with an absolute difference in mortality of 2.6% resulting in a number needed to harm of 38.5. This difference in mortality remained significant whether the patients were in a SICU or MICU, diabetic or nondiabetic, with or without sepsis, and with APACHE II scores of above or below 25. The excess deaths were attributed to cardiovascular causes, but more investigation is needed. This study demonstrates that there is no additional benefit and that there may be harm in pursuing aggressive glucose control in ICU patients.
Bottom line: When compared with conventional practice in adult ICU patients, intensive glucose control resulted in an increase in 90-day mortality.
Citation: NICE-SUGAR Study Investigators, Finfer S, Chittock DR, et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283-1297.
Clinical question: Does intensive glucose control reduce mortality at 90 days in adult ICU patients?
Background: The American Diabetic Association currently recommends tight glucose control for patients admitted to an ICU, despite conflicting evidence in the literature about the benefits of this practice.
Study design: Randomized controlled trial.
Setting: Medical and surgical ICUs at 42 hospitals in Australia, Canada, and New Zealand.
Synopsis: More than 6,000 medical and surgical ICU patients were randomly assigned to receive either intensive (target blood sugar range of 81 mg/dL to 108 mg/dL) or conventional (target blood sugar of <180 mg/dL) glucose control. Eligible patients were expected to stay at least three days in the ICU. Mortality at 90 days for the intensive treatment group was 27.5% versus 24.9% in the conventional treatment group, with an absolute difference in mortality of 2.6% resulting in a number needed to harm of 38.5. This difference in mortality remained significant whether the patients were in a SICU or MICU, diabetic or nondiabetic, with or without sepsis, and with APACHE II scores of above or below 25. The excess deaths were attributed to cardiovascular causes, but more investigation is needed. This study demonstrates that there is no additional benefit and that there may be harm in pursuing aggressive glucose control in ICU patients.
Bottom line: When compared with conventional practice in adult ICU patients, intensive glucose control resulted in an increase in 90-day mortality.
Citation: NICE-SUGAR Study Investigators, Finfer S, Chittock DR, et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283-1297.
Clinical question: Does intensive glucose control reduce mortality at 90 days in adult ICU patients?
Background: The American Diabetic Association currently recommends tight glucose control for patients admitted to an ICU, despite conflicting evidence in the literature about the benefits of this practice.
Study design: Randomized controlled trial.
Setting: Medical and surgical ICUs at 42 hospitals in Australia, Canada, and New Zealand.
Synopsis: More than 6,000 medical and surgical ICU patients were randomly assigned to receive either intensive (target blood sugar range of 81 mg/dL to 108 mg/dL) or conventional (target blood sugar of <180 mg/dL) glucose control. Eligible patients were expected to stay at least three days in the ICU. Mortality at 90 days for the intensive treatment group was 27.5% versus 24.9% in the conventional treatment group, with an absolute difference in mortality of 2.6% resulting in a number needed to harm of 38.5. This difference in mortality remained significant whether the patients were in a SICU or MICU, diabetic or nondiabetic, with or without sepsis, and with APACHE II scores of above or below 25. The excess deaths were attributed to cardiovascular causes, but more investigation is needed. This study demonstrates that there is no additional benefit and that there may be harm in pursuing aggressive glucose control in ICU patients.
Bottom line: When compared with conventional practice in adult ICU patients, intensive glucose control resulted in an increase in 90-day mortality.
Citation: NICE-SUGAR Study Investigators, Finfer S, Chittock DR, et al. Intensive versus conventional glucose control in critically ill patients. N Engl J Med. 2009;360(13):1283-1297.
Team Approach to Patient Care
Hospitalists find themselves working with nonphysician providers (NPP) more and more as institutions spread workloads and cut costs. The work arrangement can be panaceas when they work well, barely palatable when they don’t.
Several sessions at HM09 in Chicago explored the best practices of HM groups that use nurse practitioners (NPs) and physician assistants (PAs). A particularly popular session examined case studies that showed two main trends: NPP programs run into trouble when the people involved view their positions as competition; programs succeed when there is buy-in from all stakeholders.
“There are things DOs and MDs do better than NPs and PAs, and there are things NPs and PAs do better than DOs and MDs,” says Mitch Wilson, MD, FHM, corporate medical director for Eagle Hospital Physicians in Atlanta. "It’s all about understanding the cumulative skill set.”
Dr. Wilson and Tracy Cardin, ACNP, at the University of Chicago Medical Center, used care stories as teaching tools for hospitals looking to implement new programs. The first cautionary tale failed because an “old school” culture left a new NPP so isolated from physicians and nursing staff that she quit. Two other cases showed NPPs integrate more easily because of mutual respect, defined responsibilities, and an alignment of expectations between both hospitalists and NPPs.
Mac McCormick, MD, vice president of clinical services at Eagle Hospital Physicians in Atlanta, offers the following suggestions to best utilize NPPs:
•Conduct an analysis of your practice environment, bylaws, staff experience, and pre-existing attitudes to identify potential barriers and optimal opportunities in hiring NPPs;
•Avoid pigeonholing NPPs into such narrow roles as completing discharge summaries or collecting data. Tasks that tend not to utilize their skills set might lead to professional dissatisfaction and likely aren't the most cost-efficient use of resources; and
•Approach things in a team model. Shared visits are one way to accomplish this. Keep communication lines open to make sure accurate and timely information is available to all.
Hospitalists find themselves working with nonphysician providers (NPP) more and more as institutions spread workloads and cut costs. The work arrangement can be panaceas when they work well, barely palatable when they don’t.
Several sessions at HM09 in Chicago explored the best practices of HM groups that use nurse practitioners (NPs) and physician assistants (PAs). A particularly popular session examined case studies that showed two main trends: NPP programs run into trouble when the people involved view their positions as competition; programs succeed when there is buy-in from all stakeholders.
“There are things DOs and MDs do better than NPs and PAs, and there are things NPs and PAs do better than DOs and MDs,” says Mitch Wilson, MD, FHM, corporate medical director for Eagle Hospital Physicians in Atlanta. "It’s all about understanding the cumulative skill set.”
Dr. Wilson and Tracy Cardin, ACNP, at the University of Chicago Medical Center, used care stories as teaching tools for hospitals looking to implement new programs. The first cautionary tale failed because an “old school” culture left a new NPP so isolated from physicians and nursing staff that she quit. Two other cases showed NPPs integrate more easily because of mutual respect, defined responsibilities, and an alignment of expectations between both hospitalists and NPPs.
Mac McCormick, MD, vice president of clinical services at Eagle Hospital Physicians in Atlanta, offers the following suggestions to best utilize NPPs:
•Conduct an analysis of your practice environment, bylaws, staff experience, and pre-existing attitudes to identify potential barriers and optimal opportunities in hiring NPPs;
•Avoid pigeonholing NPPs into such narrow roles as completing discharge summaries or collecting data. Tasks that tend not to utilize their skills set might lead to professional dissatisfaction and likely aren't the most cost-efficient use of resources; and
•Approach things in a team model. Shared visits are one way to accomplish this. Keep communication lines open to make sure accurate and timely information is available to all.
Hospitalists find themselves working with nonphysician providers (NPP) more and more as institutions spread workloads and cut costs. The work arrangement can be panaceas when they work well, barely palatable when they don’t.
Several sessions at HM09 in Chicago explored the best practices of HM groups that use nurse practitioners (NPs) and physician assistants (PAs). A particularly popular session examined case studies that showed two main trends: NPP programs run into trouble when the people involved view their positions as competition; programs succeed when there is buy-in from all stakeholders.
“There are things DOs and MDs do better than NPs and PAs, and there are things NPs and PAs do better than DOs and MDs,” says Mitch Wilson, MD, FHM, corporate medical director for Eagle Hospital Physicians in Atlanta. "It’s all about understanding the cumulative skill set.”
Dr. Wilson and Tracy Cardin, ACNP, at the University of Chicago Medical Center, used care stories as teaching tools for hospitals looking to implement new programs. The first cautionary tale failed because an “old school” culture left a new NPP so isolated from physicians and nursing staff that she quit. Two other cases showed NPPs integrate more easily because of mutual respect, defined responsibilities, and an alignment of expectations between both hospitalists and NPPs.
Mac McCormick, MD, vice president of clinical services at Eagle Hospital Physicians in Atlanta, offers the following suggestions to best utilize NPPs:
•Conduct an analysis of your practice environment, bylaws, staff experience, and pre-existing attitudes to identify potential barriers and optimal opportunities in hiring NPPs;
•Avoid pigeonholing NPPs into such narrow roles as completing discharge summaries or collecting data. Tasks that tend not to utilize their skills set might lead to professional dissatisfaction and likely aren't the most cost-efficient use of resources; and
•Approach things in a team model. Shared visits are one way to accomplish this. Keep communication lines open to make sure accurate and timely information is available to all.
Tackle Medical School Debt
Overwhelmed by medical school debt? You're not alone. A 2008 American Medical Association survey showed that the average 2007 medical school graduate was left with a $139,000 debt burden and a powerful incentive to avoid primary care.
But according to Renee Zerehi, the American College of Physicians' manager of health policy, increasing numbers of students choose HM because of flexible scheduling and opportunities to reduce their debt. Zerehi and Bijo Chacko, MD, FHM, a member of SHM's Young Physicians Committee and hospitalist program medical director for Preferred Health Partners in New York City, offer these strategies for debt reduction.
•Understand your debt portfolio: Talk to a financial consultant to assess debt, your family situation, and lifestyle issues. "A strong, keen understanding of how debt impacts your budget is essential," Dr. Chacko says. Medical school loans often come with different interest rates and grace periods, so try to pay off the high-interest loans immediately, he explains.
•Consolidate your debt: Loans from different lenders with different balances, interest rates, and due dates may best be handled by a federal consolidation loan. The AMA explains it all in
"The Ins and Outs of Student Loan Consolidation."
•Student loan forgiveness: A number of hospitalist programs offer loan repayment programs. The National Health Service Corps, the Health Professions Scholarship Program, and state loan repayment programs offer loan forgiveness for physicians practicing in underserved areas. Visit the AAMC Web site for a comprehensive list.
•NIH Faculty Loan Forgiveness: For academic hospitalists doing research, the National Institutes of Health (NIH) offers up to $35,000 a year for loan repayment and tax reimbursement for each year of service.
For more information, visit SHM's Young Physician microsite.
Overwhelmed by medical school debt? You're not alone. A 2008 American Medical Association survey showed that the average 2007 medical school graduate was left with a $139,000 debt burden and a powerful incentive to avoid primary care.
But according to Renee Zerehi, the American College of Physicians' manager of health policy, increasing numbers of students choose HM because of flexible scheduling and opportunities to reduce their debt. Zerehi and Bijo Chacko, MD, FHM, a member of SHM's Young Physicians Committee and hospitalist program medical director for Preferred Health Partners in New York City, offer these strategies for debt reduction.
•Understand your debt portfolio: Talk to a financial consultant to assess debt, your family situation, and lifestyle issues. "A strong, keen understanding of how debt impacts your budget is essential," Dr. Chacko says. Medical school loans often come with different interest rates and grace periods, so try to pay off the high-interest loans immediately, he explains.
•Consolidate your debt: Loans from different lenders with different balances, interest rates, and due dates may best be handled by a federal consolidation loan. The AMA explains it all in
"The Ins and Outs of Student Loan Consolidation."
•Student loan forgiveness: A number of hospitalist programs offer loan repayment programs. The National Health Service Corps, the Health Professions Scholarship Program, and state loan repayment programs offer loan forgiveness for physicians practicing in underserved areas. Visit the AAMC Web site for a comprehensive list.
•NIH Faculty Loan Forgiveness: For academic hospitalists doing research, the National Institutes of Health (NIH) offers up to $35,000 a year for loan repayment and tax reimbursement for each year of service.
For more information, visit SHM's Young Physician microsite.
Overwhelmed by medical school debt? You're not alone. A 2008 American Medical Association survey showed that the average 2007 medical school graduate was left with a $139,000 debt burden and a powerful incentive to avoid primary care.
But according to Renee Zerehi, the American College of Physicians' manager of health policy, increasing numbers of students choose HM because of flexible scheduling and opportunities to reduce their debt. Zerehi and Bijo Chacko, MD, FHM, a member of SHM's Young Physicians Committee and hospitalist program medical director for Preferred Health Partners in New York City, offer these strategies for debt reduction.
•Understand your debt portfolio: Talk to a financial consultant to assess debt, your family situation, and lifestyle issues. "A strong, keen understanding of how debt impacts your budget is essential," Dr. Chacko says. Medical school loans often come with different interest rates and grace periods, so try to pay off the high-interest loans immediately, he explains.
•Consolidate your debt: Loans from different lenders with different balances, interest rates, and due dates may best be handled by a federal consolidation loan. The AMA explains it all in
"The Ins and Outs of Student Loan Consolidation."
•Student loan forgiveness: A number of hospitalist programs offer loan repayment programs. The National Health Service Corps, the Health Professions Scholarship Program, and state loan repayment programs offer loan forgiveness for physicians practicing in underserved areas. Visit the AAMC Web site for a comprehensive list.
•NIH Faculty Loan Forgiveness: For academic hospitalists doing research, the National Institutes of Health (NIH) offers up to $35,000 a year for loan repayment and tax reimbursement for each year of service.
For more information, visit SHM's Young Physician microsite.
Quality Care for COPD, Secondary Stroke Prevention, Treat Classic CTA
Factors Influencing the Treatment of COPD
Lindenauer PK, Pekow P, Gao S, et al. Quality of care for patients hospitalized for acute exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 2006;144:894-903.
Background
Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death in the United States, resulting in more than $18 billion in annual health costs. Acute exacerbations of COPD can lead to respiratory compromise and are one of the 10 leading causes of hospitalization in the United States.
Hospitalists currently have evidence-based guidelines available that recommend therapies for patients with acute exacerbations of COPD. This study was designed to evaluate the practice patterns in the United States and to evaluate the quality of care provided to hospitalized patients based on comparisons with these published guidelines. The authors did not report any conflicts of interest, and this work was performed without external grant support.
Methods
Using administrative data from the 360 hospitals that participate in Perspective, a database developed for measuring healthcare quality and utilization, the authors performed a retrospective cohort study. Patients hospitalized for a primary diagnosis of acute exacerbation of COPD were chosen. Patients with pneumonia were specifically excluded. The outcomes of interest included adherence to the diagnostic and therapeutic recommendations of the joint American College of Physicians and American College of Chest Physicians evidence-based COPD guideline, published in 2001.
Results
Of the 69,820 patients included in the analysis, 33% received “ideal care,” defined as all of the recommended care and none of the non-beneficial interventions. Specific results included varied utilization of recommended care: 95% had chest radiography, 91% received supplemental oxygen, 97% had bronchodilators, 85% were given systemic steroids, and 85% received antibiotics.
Overall, 45% of patients received at least one non-beneficial intervention specified in the guidelines: 24% were treated with methylxanthines, 14% underwent sputum testing, 12% had acute spirometry, 6% received chest physiotherapy, and 2% were given mucolytics.
Older patients and women were more likely to receive ideal care as defined, but hospitals with a higher annual volume of COPD cases were not associated with improved performance in this analysis.
Conclusions
Given a widely accepted evidence-based practice guideline as a benchmark, significant variation exists across hospitals in the quality of care for acute exacerbations of COPD. Opportunities exist to improve the quality of care, in particular by increasing the use of systemic corticosteroids and antibiotic therapy and reducing the utilization of many diagnostic and therapeutic interventions that are not only not recommended but are also potentially harmful.
Commentary
COPD management in the acute inpatient setting is on the horizon as a focus of policymakers, and this study suggests that significant opportunities exist for inpatient physicians to reduce variation in practice and utilize an evidence-based approach to the treatment of acute exacerbations of COPD. This study is limited by its use of administrative data, its inability to use clinical data to best determine appropriate care processes for individual patients, and its retrospective design.
As we move toward external quality metrics for the care of patients with acute exacerbations of COPD, further prospective studies evaluating clinical outcomes of interest, including mortality and readmission rates, are needed to determine the effects of adherence to ideal or recommended care for acute exacerbations of COPD.1-3
References
- Snow V, Lascher S, Mottur-Pilson C, et al. Evidence base for management of acute exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 2001 Apr 3;134(7):595-599.
- American Thoracic Society. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease (COPD) and asthma. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, November 1986. Am Rev Respir Dis. 1987 Jul;136(1):225-244.
- Agency for Healthcare Research and Quality. Management of Acute Exacerbations of Chronic Obstructive Pulmonary Disease. Rockville, Md.: Agency for Healthcare Research and Quality; 2000.
The Role of Dipyridamole in the Secondary Prevention of Stroke
ESPRIT Study Group. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPRIT): randomized controlled trial. Lancet. 2006 May 20;367(9623):1665-1673.
Background
To date, studies have resulted in inconsistent results in trials of aspirin versus aspirin in combination with dipyridamole for secondary prevention of ischemic stroke. Four early, smaller studies have yielded non-significant results, in contrast to the statistically significant relative risk reduction seen with the addition of dipyridamole to aspirin in the European Stroke Prevention Study 2 (ESPS-2).1-2
Methods
The European/Australian Stroke Prevention in Reversible Ischaemia Trial (ESPRIT) study group conducted a prospective randomized controlled trial of 2,763 patients with transient ischemic attacks or minor ischemic stroke of presumed arterial origin who received aspirin (30-325 mg daily) with or without dipyridamole (200 mg twice daily) as secondary prevention. The primary outcome for this study was a composite of death from vascular causes, nonfatal stroke, nonfatal myocardial infarction, or major bleeding complication. Mean follow-up of patients enrolled was 3.5 years.
Results
In an intention-to-treat analysis, the primary combined endpoint occurred in 16% (216) of the patients on aspirin alone (median aspirin dose was 75 mg in both groups) compared with 13% (173) of the patients on aspirin plus dipyridamole. This result was statistically significant, with an absolute risk reduction of 1% per year. As noted in other trials, patients on dipyridamole discontinued their study medication more frequently than patients on aspirin alone, mostly due to headache.
Conclusions
The results of this trial, taken in the context of previously published data, support the combination of aspirin plus dipyridamole over aspirin alone for the secondary prevention of ischemic stroke of presumed arterial origin. Addition of these data to the previous meta-analysis of trials resulted in a statistically significant risk ratio for the composite endpoint of 0.82 (95% confidence interval, 0.74-0.91).1
Commentary
Ischemic stroke and transient ischemic attacks remain a challenge to effectively manage medically and are appropriately greatly feared health complications for many patients, resulting in significant morbidity and mortality. Prior studies of secondary prevention with aspirin therapy have demonstrated only a modest reduction in vascular complications in these patients.3-4
The results of this trial are consistent with data from the Second European Stroke Prevention Study, and in combination, these data confirm that the addition of dipyridamole for patients who can tolerate it offers significant benefit.2 The magnitude of the effect results in a number needed to treat of 100 patients for one year to prevent one vascular death, stroke, or myocardial infarction. Given the clinical significance of these outcomes, many patients may prefer a trial on combination therapy.
References
- Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002 Jan 12;324(7329):71-86.
- Diener HC, Cunha L, Forbes C, et al. European Stroke Prevention Study. Dipyridamole and acetylsalicylic acid in the secondary prevention of stroke. J Neurol Sci. 1996;143:1-13.
- Warlow C. Secondary prevention of stroke. Lancet. 1992;339:724-727.
- Algra A, van Gijn J. Cumulative meta-analysis of aspirin efficacy after cerebral ischaemia of arterial origin. J Neurol Neurosurg Psychiatry. 1999 Feb;66(2):255.
The Effectiveness of CTA in Diagnosing Acute Pulmonary Embolism
Stein PD, Fowler SE, Goodman LR, et al. Multidetector computed tomography for acute pulmonary embolism. N Engl J Med. 2006 Jun 1;354(22):2317-2327.
Background
The Prospective Investigation of Pulmonary Embolism Diagnosis II (PIOPED II) trial was designed to answer questions about the accuracy of contrast-enhanced multidetector computed tomographic angiography (CTA). Recent studies of the use of single-row or multidetector CTA alone have suggested a low incidence of pulmonary embolism in follow-up of untreated patients with normal findings on CTA.
The specific goals of this study were to determine the ability of multidetector CTA to rule out or detect pulmonary embolism, and to evaluate whether the addition of computed tomographic venography (CTV) improves the diagnostic accuracy of CTA.
Methods
Using a technique similar to PIOPED I, the investigators performed a prospective, multi-center trial using a composite reference standard to confirm the diagnosis of pulmonary embolism. Once again, for ethical reasons, the use of pulmonary artery digital-subtraction angiography was limited to patients whose diagnosis could neither be confirmed nor ruled out by less invasive tests. In contrast to PIOPED I, a clinical scoring system was used to assess the clinical probability of pulmonary embolism. Central readings were performed on all imaging studies except for venous ultrasonography.
Results
Of the 7,284 patients screened for the study, 3,262 were eligible, and 1,090 were enrolled. Of those, 824 patients received a completed CTA study and a reference standard for analysis. In 51 patients, the quality of the CTA was not suitable for interpretation, and these patients were excluded from the subsequent analysis. Pulmonary embolism was diagnosed in192 patients.
CTA was found to have a sensitivity of 83% and a specificity of 96%, yielding a likelihood ratio for a positive multidetector CTA test of 19.6 (95% confidence interval, 13.3 to 29.0), while the likelihood ratio for a negative test was 0.18 (95% confidence interval, 0.13 to 0.24). The quality of results on CTA-CTV was not adequate for interpretation in 87 patients; when these patients were excluded from analysis, the sensitivity was 90% with a specificity of 95%, yielding likelihood ratios of 16.5 (95% confidence interval, 11.6 to 23.5) for a positive test and 0.11 (95% confidence interval, 0.07 to 0.16) for a negative test.
Conclusions
Multidetector CTA and CTA-CTV perform well when the results of these tests are concordant with pre-test clinical probabilities of pulmonary embolism. CTA-CTV offers slightly increased sensitivity compared with CTA alone, with no significant difference in specificity. If the results of CTA or CTA-CTV are inconsistent with the clinical probability of pulmonary embolism, additional diagnostic testing is indicated.
Commentary
CTA has been used widely, and in many centers has largely replaced other diagnostic tests for pulmonary embolism. This well-done study incorporated recent advances in technology with multidetector CTA-CTV, along with a clinical prediction rule to better estimate pre-test probabilities of pulmonary embolism.2 It is important to recognize that 266 of the 1,090 patients enrolled were not included in the calculations of sensitivity and specificity for CTA-CTV because they did not have interpretable test results.
Although the specificity of both CTA and the CTA-CTV combination were high, the sensitivity was not sufficient to identify all cases of pulmonary embolism. This result contrasts to the recent outcomes studies of CTA, in which low rates of venous thromboembolism were seen in follow-up of patients with negative multidetector CTA.3,4 Although multidetector CTA has a higher sensitivity than single-slice technology, this test may still miss small subsegmental thrombi that might be detected using other diagnostic tests (ventilation-perfusion scintigraphy and/or pulmonary digital-subtraction angiography).
An important take-home message from this study is to recognize once again the importance of utilizing established clinical prediction rules for venous thrombosis and pulmonary embolism (such as the Wells clinical model).2 As with the majority of diagnostic tests at our disposal, when our clinical judgment is in contrast with test results, as in the case of a high likelihood of a potentially fatal disease like pulmonary embolism with a normal CTA result, additional diagnostic testing is necessary.
References
- The PIOPED Investigators. Value of the ventilation/perfusion scan in acute pulmonary embolism: results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990;263:2753-2759.
- Wells PS, Anderson DR, Rodger M, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and d-dimer. Ann Intern Med. 2001 Jul 17;135(2):98-107.
- Perrier A, Roy PM, Sanchez O, et al. Multidetector-row computed tomography in suspected pulmonary embolism. N Engl J Med. 2005 Apr;352(17):1760-1768.
- van Belle A, Buller HR, Huisman MV, et al. Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D-dimer testing, and computed tomography. JAMA. 2006 Jan 11;295(2):172-179.
Classic Article:
PIOPED Investigators
The PIOPED Investigators. Value of ventilation/perfusion scan in acute pulmonary embolism: results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990;263:2753-2759.
Background
The risk of untreated pulmonary embolism requires either the diagnosis or the exclusion of this diagnosis when clinical suspicion exists. The reference test for pulmonary embolism, standard pulmonary angiography, is invasive and expensive, and carries with it a measurable procedural risk.
Non-invasive diagnostic tests, including ventilation/perfusion (V/Q) scintigraphy, have been used to detect perfusion defects consistent with pulmonary embolism, though the performance characteristics of this diagnostic test were not well known prior to 1990. This study was designed to evaluate the sensitivity and specificities of ventilation/perfusion lung scans for pulmonary embolism in the acute setting.
Methods
This prospective, multi-center study evaluated V/Q scintigraphy on a random sample of 931 patients. A composite reference standard was used because only 755 patients underwent scintigraphy and pulmonary angiography. Clinical follow-up and subsequent diagnostic testing were employed in untreated patients with low clinical probabilities of pulmonary embolism who did not undergo angiography. Clinical assessment of the probability of pulmonary embolism was determined on the basis of the clinician’s judgment, without systematic prediction rules.
Results
Almost all patients with pulmonary embolism had abnormal ventilation/perfusion lung scans of high, intermediate, or low probability. Unfortunately, most patients without pulmonary embolism also had abnormal studies, limiting the utility of this test. Clinical follow-up and angiography revealed that pulmonary embolism occurred among 12% of patients with low-probability scans.
Conclusions
V/Q scintigraphy is useful in establishing or excluding the diagnosis of pulmonary embolism in only a minority of patients, where clinical suspicion of pulmonary embolism is concordant with the diagnostic test findings. The likelihood of pulmonary embolism in patients with a high pre-test probability of pulmonary embolism and a high probability scan is 95%, while in low probability patients with a low probability or normal scan the probability is 4% or 2%, respectively.
Commentary
This original PIOPED study established the diagnostic characteristics of V/Q scintigraphy and demonstrated, for the first time, evidence of the role of clinical assessment and prior probability in a diagnostic strategy for pulmonary embolism. Although subsequent studies have significantly advanced our knowledge of clinical prediction and diagnostic strategies in venous thromboembolism, the first PIOPED study continues to serve as an example of a high-quality, multi-center diagnostic test study utilizing a composite reference standard in a difficult-to-study disease. Unfortunately, the results of this study demonstrated that V/Q scintigraphy performs well for only a minority of patients. The majority of patients (72%) had clinical probabilities of pulmonary embolism and ventilation/perfusion scan results, which yielded post-test probabilities of 15-86%, leaving, in many cases, enough remaining diagnostic uncertainty to warrant additional testing.—TO TH
Factors Influencing the Treatment of COPD
Lindenauer PK, Pekow P, Gao S, et al. Quality of care for patients hospitalized for acute exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 2006;144:894-903.
Background
Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death in the United States, resulting in more than $18 billion in annual health costs. Acute exacerbations of COPD can lead to respiratory compromise and are one of the 10 leading causes of hospitalization in the United States.
Hospitalists currently have evidence-based guidelines available that recommend therapies for patients with acute exacerbations of COPD. This study was designed to evaluate the practice patterns in the United States and to evaluate the quality of care provided to hospitalized patients based on comparisons with these published guidelines. The authors did not report any conflicts of interest, and this work was performed without external grant support.
Methods
Using administrative data from the 360 hospitals that participate in Perspective, a database developed for measuring healthcare quality and utilization, the authors performed a retrospective cohort study. Patients hospitalized for a primary diagnosis of acute exacerbation of COPD were chosen. Patients with pneumonia were specifically excluded. The outcomes of interest included adherence to the diagnostic and therapeutic recommendations of the joint American College of Physicians and American College of Chest Physicians evidence-based COPD guideline, published in 2001.
Results
Of the 69,820 patients included in the analysis, 33% received “ideal care,” defined as all of the recommended care and none of the non-beneficial interventions. Specific results included varied utilization of recommended care: 95% had chest radiography, 91% received supplemental oxygen, 97% had bronchodilators, 85% were given systemic steroids, and 85% received antibiotics.
Overall, 45% of patients received at least one non-beneficial intervention specified in the guidelines: 24% were treated with methylxanthines, 14% underwent sputum testing, 12% had acute spirometry, 6% received chest physiotherapy, and 2% were given mucolytics.
Older patients and women were more likely to receive ideal care as defined, but hospitals with a higher annual volume of COPD cases were not associated with improved performance in this analysis.
Conclusions
Given a widely accepted evidence-based practice guideline as a benchmark, significant variation exists across hospitals in the quality of care for acute exacerbations of COPD. Opportunities exist to improve the quality of care, in particular by increasing the use of systemic corticosteroids and antibiotic therapy and reducing the utilization of many diagnostic and therapeutic interventions that are not only not recommended but are also potentially harmful.
Commentary
COPD management in the acute inpatient setting is on the horizon as a focus of policymakers, and this study suggests that significant opportunities exist for inpatient physicians to reduce variation in practice and utilize an evidence-based approach to the treatment of acute exacerbations of COPD. This study is limited by its use of administrative data, its inability to use clinical data to best determine appropriate care processes for individual patients, and its retrospective design.
As we move toward external quality metrics for the care of patients with acute exacerbations of COPD, further prospective studies evaluating clinical outcomes of interest, including mortality and readmission rates, are needed to determine the effects of adherence to ideal or recommended care for acute exacerbations of COPD.1-3
References
- Snow V, Lascher S, Mottur-Pilson C, et al. Evidence base for management of acute exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 2001 Apr 3;134(7):595-599.
- American Thoracic Society. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease (COPD) and asthma. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, November 1986. Am Rev Respir Dis. 1987 Jul;136(1):225-244.
- Agency for Healthcare Research and Quality. Management of Acute Exacerbations of Chronic Obstructive Pulmonary Disease. Rockville, Md.: Agency for Healthcare Research and Quality; 2000.
The Role of Dipyridamole in the Secondary Prevention of Stroke
ESPRIT Study Group. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPRIT): randomized controlled trial. Lancet. 2006 May 20;367(9623):1665-1673.
Background
To date, studies have resulted in inconsistent results in trials of aspirin versus aspirin in combination with dipyridamole for secondary prevention of ischemic stroke. Four early, smaller studies have yielded non-significant results, in contrast to the statistically significant relative risk reduction seen with the addition of dipyridamole to aspirin in the European Stroke Prevention Study 2 (ESPS-2).1-2
Methods
The European/Australian Stroke Prevention in Reversible Ischaemia Trial (ESPRIT) study group conducted a prospective randomized controlled trial of 2,763 patients with transient ischemic attacks or minor ischemic stroke of presumed arterial origin who received aspirin (30-325 mg daily) with or without dipyridamole (200 mg twice daily) as secondary prevention. The primary outcome for this study was a composite of death from vascular causes, nonfatal stroke, nonfatal myocardial infarction, or major bleeding complication. Mean follow-up of patients enrolled was 3.5 years.
Results
In an intention-to-treat analysis, the primary combined endpoint occurred in 16% (216) of the patients on aspirin alone (median aspirin dose was 75 mg in both groups) compared with 13% (173) of the patients on aspirin plus dipyridamole. This result was statistically significant, with an absolute risk reduction of 1% per year. As noted in other trials, patients on dipyridamole discontinued their study medication more frequently than patients on aspirin alone, mostly due to headache.
Conclusions
The results of this trial, taken in the context of previously published data, support the combination of aspirin plus dipyridamole over aspirin alone for the secondary prevention of ischemic stroke of presumed arterial origin. Addition of these data to the previous meta-analysis of trials resulted in a statistically significant risk ratio for the composite endpoint of 0.82 (95% confidence interval, 0.74-0.91).1
Commentary
Ischemic stroke and transient ischemic attacks remain a challenge to effectively manage medically and are appropriately greatly feared health complications for many patients, resulting in significant morbidity and mortality. Prior studies of secondary prevention with aspirin therapy have demonstrated only a modest reduction in vascular complications in these patients.3-4
The results of this trial are consistent with data from the Second European Stroke Prevention Study, and in combination, these data confirm that the addition of dipyridamole for patients who can tolerate it offers significant benefit.2 The magnitude of the effect results in a number needed to treat of 100 patients for one year to prevent one vascular death, stroke, or myocardial infarction. Given the clinical significance of these outcomes, many patients may prefer a trial on combination therapy.
References
- Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002 Jan 12;324(7329):71-86.
- Diener HC, Cunha L, Forbes C, et al. European Stroke Prevention Study. Dipyridamole and acetylsalicylic acid in the secondary prevention of stroke. J Neurol Sci. 1996;143:1-13.
- Warlow C. Secondary prevention of stroke. Lancet. 1992;339:724-727.
- Algra A, van Gijn J. Cumulative meta-analysis of aspirin efficacy after cerebral ischaemia of arterial origin. J Neurol Neurosurg Psychiatry. 1999 Feb;66(2):255.
The Effectiveness of CTA in Diagnosing Acute Pulmonary Embolism
Stein PD, Fowler SE, Goodman LR, et al. Multidetector computed tomography for acute pulmonary embolism. N Engl J Med. 2006 Jun 1;354(22):2317-2327.
Background
The Prospective Investigation of Pulmonary Embolism Diagnosis II (PIOPED II) trial was designed to answer questions about the accuracy of contrast-enhanced multidetector computed tomographic angiography (CTA). Recent studies of the use of single-row or multidetector CTA alone have suggested a low incidence of pulmonary embolism in follow-up of untreated patients with normal findings on CTA.
The specific goals of this study were to determine the ability of multidetector CTA to rule out or detect pulmonary embolism, and to evaluate whether the addition of computed tomographic venography (CTV) improves the diagnostic accuracy of CTA.
Methods
Using a technique similar to PIOPED I, the investigators performed a prospective, multi-center trial using a composite reference standard to confirm the diagnosis of pulmonary embolism. Once again, for ethical reasons, the use of pulmonary artery digital-subtraction angiography was limited to patients whose diagnosis could neither be confirmed nor ruled out by less invasive tests. In contrast to PIOPED I, a clinical scoring system was used to assess the clinical probability of pulmonary embolism. Central readings were performed on all imaging studies except for venous ultrasonography.
Results
Of the 7,284 patients screened for the study, 3,262 were eligible, and 1,090 were enrolled. Of those, 824 patients received a completed CTA study and a reference standard for analysis. In 51 patients, the quality of the CTA was not suitable for interpretation, and these patients were excluded from the subsequent analysis. Pulmonary embolism was diagnosed in192 patients.
CTA was found to have a sensitivity of 83% and a specificity of 96%, yielding a likelihood ratio for a positive multidetector CTA test of 19.6 (95% confidence interval, 13.3 to 29.0), while the likelihood ratio for a negative test was 0.18 (95% confidence interval, 0.13 to 0.24). The quality of results on CTA-CTV was not adequate for interpretation in 87 patients; when these patients were excluded from analysis, the sensitivity was 90% with a specificity of 95%, yielding likelihood ratios of 16.5 (95% confidence interval, 11.6 to 23.5) for a positive test and 0.11 (95% confidence interval, 0.07 to 0.16) for a negative test.
Conclusions
Multidetector CTA and CTA-CTV perform well when the results of these tests are concordant with pre-test clinical probabilities of pulmonary embolism. CTA-CTV offers slightly increased sensitivity compared with CTA alone, with no significant difference in specificity. If the results of CTA or CTA-CTV are inconsistent with the clinical probability of pulmonary embolism, additional diagnostic testing is indicated.
Commentary
CTA has been used widely, and in many centers has largely replaced other diagnostic tests for pulmonary embolism. This well-done study incorporated recent advances in technology with multidetector CTA-CTV, along with a clinical prediction rule to better estimate pre-test probabilities of pulmonary embolism.2 It is important to recognize that 266 of the 1,090 patients enrolled were not included in the calculations of sensitivity and specificity for CTA-CTV because they did not have interpretable test results.
Although the specificity of both CTA and the CTA-CTV combination were high, the sensitivity was not sufficient to identify all cases of pulmonary embolism. This result contrasts to the recent outcomes studies of CTA, in which low rates of venous thromboembolism were seen in follow-up of patients with negative multidetector CTA.3,4 Although multidetector CTA has a higher sensitivity than single-slice technology, this test may still miss small subsegmental thrombi that might be detected using other diagnostic tests (ventilation-perfusion scintigraphy and/or pulmonary digital-subtraction angiography).
An important take-home message from this study is to recognize once again the importance of utilizing established clinical prediction rules for venous thrombosis and pulmonary embolism (such as the Wells clinical model).2 As with the majority of diagnostic tests at our disposal, when our clinical judgment is in contrast with test results, as in the case of a high likelihood of a potentially fatal disease like pulmonary embolism with a normal CTA result, additional diagnostic testing is necessary.
References
- The PIOPED Investigators. Value of the ventilation/perfusion scan in acute pulmonary embolism: results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990;263:2753-2759.
- Wells PS, Anderson DR, Rodger M, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and d-dimer. Ann Intern Med. 2001 Jul 17;135(2):98-107.
- Perrier A, Roy PM, Sanchez O, et al. Multidetector-row computed tomography in suspected pulmonary embolism. N Engl J Med. 2005 Apr;352(17):1760-1768.
- van Belle A, Buller HR, Huisman MV, et al. Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D-dimer testing, and computed tomography. JAMA. 2006 Jan 11;295(2):172-179.
Classic Article:
PIOPED Investigators
The PIOPED Investigators. Value of ventilation/perfusion scan in acute pulmonary embolism: results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990;263:2753-2759.
Background
The risk of untreated pulmonary embolism requires either the diagnosis or the exclusion of this diagnosis when clinical suspicion exists. The reference test for pulmonary embolism, standard pulmonary angiography, is invasive and expensive, and carries with it a measurable procedural risk.
Non-invasive diagnostic tests, including ventilation/perfusion (V/Q) scintigraphy, have been used to detect perfusion defects consistent with pulmonary embolism, though the performance characteristics of this diagnostic test were not well known prior to 1990. This study was designed to evaluate the sensitivity and specificities of ventilation/perfusion lung scans for pulmonary embolism in the acute setting.
Methods
This prospective, multi-center study evaluated V/Q scintigraphy on a random sample of 931 patients. A composite reference standard was used because only 755 patients underwent scintigraphy and pulmonary angiography. Clinical follow-up and subsequent diagnostic testing were employed in untreated patients with low clinical probabilities of pulmonary embolism who did not undergo angiography. Clinical assessment of the probability of pulmonary embolism was determined on the basis of the clinician’s judgment, without systematic prediction rules.
Results
Almost all patients with pulmonary embolism had abnormal ventilation/perfusion lung scans of high, intermediate, or low probability. Unfortunately, most patients without pulmonary embolism also had abnormal studies, limiting the utility of this test. Clinical follow-up and angiography revealed that pulmonary embolism occurred among 12% of patients with low-probability scans.
Conclusions
V/Q scintigraphy is useful in establishing or excluding the diagnosis of pulmonary embolism in only a minority of patients, where clinical suspicion of pulmonary embolism is concordant with the diagnostic test findings. The likelihood of pulmonary embolism in patients with a high pre-test probability of pulmonary embolism and a high probability scan is 95%, while in low probability patients with a low probability or normal scan the probability is 4% or 2%, respectively.
Commentary
This original PIOPED study established the diagnostic characteristics of V/Q scintigraphy and demonstrated, for the first time, evidence of the role of clinical assessment and prior probability in a diagnostic strategy for pulmonary embolism. Although subsequent studies have significantly advanced our knowledge of clinical prediction and diagnostic strategies in venous thromboembolism, the first PIOPED study continues to serve as an example of a high-quality, multi-center diagnostic test study utilizing a composite reference standard in a difficult-to-study disease. Unfortunately, the results of this study demonstrated that V/Q scintigraphy performs well for only a minority of patients. The majority of patients (72%) had clinical probabilities of pulmonary embolism and ventilation/perfusion scan results, which yielded post-test probabilities of 15-86%, leaving, in many cases, enough remaining diagnostic uncertainty to warrant additional testing.—TO TH
Factors Influencing the Treatment of COPD
Lindenauer PK, Pekow P, Gao S, et al. Quality of care for patients hospitalized for acute exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 2006;144:894-903.
Background
Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death in the United States, resulting in more than $18 billion in annual health costs. Acute exacerbations of COPD can lead to respiratory compromise and are one of the 10 leading causes of hospitalization in the United States.
Hospitalists currently have evidence-based guidelines available that recommend therapies for patients with acute exacerbations of COPD. This study was designed to evaluate the practice patterns in the United States and to evaluate the quality of care provided to hospitalized patients based on comparisons with these published guidelines. The authors did not report any conflicts of interest, and this work was performed without external grant support.
Methods
Using administrative data from the 360 hospitals that participate in Perspective, a database developed for measuring healthcare quality and utilization, the authors performed a retrospective cohort study. Patients hospitalized for a primary diagnosis of acute exacerbation of COPD were chosen. Patients with pneumonia were specifically excluded. The outcomes of interest included adherence to the diagnostic and therapeutic recommendations of the joint American College of Physicians and American College of Chest Physicians evidence-based COPD guideline, published in 2001.
Results
Of the 69,820 patients included in the analysis, 33% received “ideal care,” defined as all of the recommended care and none of the non-beneficial interventions. Specific results included varied utilization of recommended care: 95% had chest radiography, 91% received supplemental oxygen, 97% had bronchodilators, 85% were given systemic steroids, and 85% received antibiotics.
Overall, 45% of patients received at least one non-beneficial intervention specified in the guidelines: 24% were treated with methylxanthines, 14% underwent sputum testing, 12% had acute spirometry, 6% received chest physiotherapy, and 2% were given mucolytics.
Older patients and women were more likely to receive ideal care as defined, but hospitals with a higher annual volume of COPD cases were not associated with improved performance in this analysis.
Conclusions
Given a widely accepted evidence-based practice guideline as a benchmark, significant variation exists across hospitals in the quality of care for acute exacerbations of COPD. Opportunities exist to improve the quality of care, in particular by increasing the use of systemic corticosteroids and antibiotic therapy and reducing the utilization of many diagnostic and therapeutic interventions that are not only not recommended but are also potentially harmful.
Commentary
COPD management in the acute inpatient setting is on the horizon as a focus of policymakers, and this study suggests that significant opportunities exist for inpatient physicians to reduce variation in practice and utilize an evidence-based approach to the treatment of acute exacerbations of COPD. This study is limited by its use of administrative data, its inability to use clinical data to best determine appropriate care processes for individual patients, and its retrospective design.
As we move toward external quality metrics for the care of patients with acute exacerbations of COPD, further prospective studies evaluating clinical outcomes of interest, including mortality and readmission rates, are needed to determine the effects of adherence to ideal or recommended care for acute exacerbations of COPD.1-3
References
- Snow V, Lascher S, Mottur-Pilson C, et al. Evidence base for management of acute exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 2001 Apr 3;134(7):595-599.
- American Thoracic Society. Standards for the diagnosis and care of patients with chronic obstructive pulmonary disease (COPD) and asthma. This official statement of the American Thoracic Society was adopted by the ATS Board of Directors, November 1986. Am Rev Respir Dis. 1987 Jul;136(1):225-244.
- Agency for Healthcare Research and Quality. Management of Acute Exacerbations of Chronic Obstructive Pulmonary Disease. Rockville, Md.: Agency for Healthcare Research and Quality; 2000.
The Role of Dipyridamole in the Secondary Prevention of Stroke
ESPRIT Study Group. Aspirin plus dipyridamole versus aspirin alone after cerebral ischaemia of arterial origin (ESPRIT): randomized controlled trial. Lancet. 2006 May 20;367(9623):1665-1673.
Background
To date, studies have resulted in inconsistent results in trials of aspirin versus aspirin in combination with dipyridamole for secondary prevention of ischemic stroke. Four early, smaller studies have yielded non-significant results, in contrast to the statistically significant relative risk reduction seen with the addition of dipyridamole to aspirin in the European Stroke Prevention Study 2 (ESPS-2).1-2
Methods
The European/Australian Stroke Prevention in Reversible Ischaemia Trial (ESPRIT) study group conducted a prospective randomized controlled trial of 2,763 patients with transient ischemic attacks or minor ischemic stroke of presumed arterial origin who received aspirin (30-325 mg daily) with or without dipyridamole (200 mg twice daily) as secondary prevention. The primary outcome for this study was a composite of death from vascular causes, nonfatal stroke, nonfatal myocardial infarction, or major bleeding complication. Mean follow-up of patients enrolled was 3.5 years.
Results
In an intention-to-treat analysis, the primary combined endpoint occurred in 16% (216) of the patients on aspirin alone (median aspirin dose was 75 mg in both groups) compared with 13% (173) of the patients on aspirin plus dipyridamole. This result was statistically significant, with an absolute risk reduction of 1% per year. As noted in other trials, patients on dipyridamole discontinued their study medication more frequently than patients on aspirin alone, mostly due to headache.
Conclusions
The results of this trial, taken in the context of previously published data, support the combination of aspirin plus dipyridamole over aspirin alone for the secondary prevention of ischemic stroke of presumed arterial origin. Addition of these data to the previous meta-analysis of trials resulted in a statistically significant risk ratio for the composite endpoint of 0.82 (95% confidence interval, 0.74-0.91).1
Commentary
Ischemic stroke and transient ischemic attacks remain a challenge to effectively manage medically and are appropriately greatly feared health complications for many patients, resulting in significant morbidity and mortality. Prior studies of secondary prevention with aspirin therapy have demonstrated only a modest reduction in vascular complications in these patients.3-4
The results of this trial are consistent with data from the Second European Stroke Prevention Study, and in combination, these data confirm that the addition of dipyridamole for patients who can tolerate it offers significant benefit.2 The magnitude of the effect results in a number needed to treat of 100 patients for one year to prevent one vascular death, stroke, or myocardial infarction. Given the clinical significance of these outcomes, many patients may prefer a trial on combination therapy.
References
- Antithrombotic Trialists’ Collaboration. Collaborative meta-analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients. BMJ. 2002 Jan 12;324(7329):71-86.
- Diener HC, Cunha L, Forbes C, et al. European Stroke Prevention Study. Dipyridamole and acetylsalicylic acid in the secondary prevention of stroke. J Neurol Sci. 1996;143:1-13.
- Warlow C. Secondary prevention of stroke. Lancet. 1992;339:724-727.
- Algra A, van Gijn J. Cumulative meta-analysis of aspirin efficacy after cerebral ischaemia of arterial origin. J Neurol Neurosurg Psychiatry. 1999 Feb;66(2):255.
The Effectiveness of CTA in Diagnosing Acute Pulmonary Embolism
Stein PD, Fowler SE, Goodman LR, et al. Multidetector computed tomography for acute pulmonary embolism. N Engl J Med. 2006 Jun 1;354(22):2317-2327.
Background
The Prospective Investigation of Pulmonary Embolism Diagnosis II (PIOPED II) trial was designed to answer questions about the accuracy of contrast-enhanced multidetector computed tomographic angiography (CTA). Recent studies of the use of single-row or multidetector CTA alone have suggested a low incidence of pulmonary embolism in follow-up of untreated patients with normal findings on CTA.
The specific goals of this study were to determine the ability of multidetector CTA to rule out or detect pulmonary embolism, and to evaluate whether the addition of computed tomographic venography (CTV) improves the diagnostic accuracy of CTA.
Methods
Using a technique similar to PIOPED I, the investigators performed a prospective, multi-center trial using a composite reference standard to confirm the diagnosis of pulmonary embolism. Once again, for ethical reasons, the use of pulmonary artery digital-subtraction angiography was limited to patients whose diagnosis could neither be confirmed nor ruled out by less invasive tests. In contrast to PIOPED I, a clinical scoring system was used to assess the clinical probability of pulmonary embolism. Central readings were performed on all imaging studies except for venous ultrasonography.
Results
Of the 7,284 patients screened for the study, 3,262 were eligible, and 1,090 were enrolled. Of those, 824 patients received a completed CTA study and a reference standard for analysis. In 51 patients, the quality of the CTA was not suitable for interpretation, and these patients were excluded from the subsequent analysis. Pulmonary embolism was diagnosed in192 patients.
CTA was found to have a sensitivity of 83% and a specificity of 96%, yielding a likelihood ratio for a positive multidetector CTA test of 19.6 (95% confidence interval, 13.3 to 29.0), while the likelihood ratio for a negative test was 0.18 (95% confidence interval, 0.13 to 0.24). The quality of results on CTA-CTV was not adequate for interpretation in 87 patients; when these patients were excluded from analysis, the sensitivity was 90% with a specificity of 95%, yielding likelihood ratios of 16.5 (95% confidence interval, 11.6 to 23.5) for a positive test and 0.11 (95% confidence interval, 0.07 to 0.16) for a negative test.
Conclusions
Multidetector CTA and CTA-CTV perform well when the results of these tests are concordant with pre-test clinical probabilities of pulmonary embolism. CTA-CTV offers slightly increased sensitivity compared with CTA alone, with no significant difference in specificity. If the results of CTA or CTA-CTV are inconsistent with the clinical probability of pulmonary embolism, additional diagnostic testing is indicated.
Commentary
CTA has been used widely, and in many centers has largely replaced other diagnostic tests for pulmonary embolism. This well-done study incorporated recent advances in technology with multidetector CTA-CTV, along with a clinical prediction rule to better estimate pre-test probabilities of pulmonary embolism.2 It is important to recognize that 266 of the 1,090 patients enrolled were not included in the calculations of sensitivity and specificity for CTA-CTV because they did not have interpretable test results.
Although the specificity of both CTA and the CTA-CTV combination were high, the sensitivity was not sufficient to identify all cases of pulmonary embolism. This result contrasts to the recent outcomes studies of CTA, in which low rates of venous thromboembolism were seen in follow-up of patients with negative multidetector CTA.3,4 Although multidetector CTA has a higher sensitivity than single-slice technology, this test may still miss small subsegmental thrombi that might be detected using other diagnostic tests (ventilation-perfusion scintigraphy and/or pulmonary digital-subtraction angiography).
An important take-home message from this study is to recognize once again the importance of utilizing established clinical prediction rules for venous thrombosis and pulmonary embolism (such as the Wells clinical model).2 As with the majority of diagnostic tests at our disposal, when our clinical judgment is in contrast with test results, as in the case of a high likelihood of a potentially fatal disease like pulmonary embolism with a normal CTA result, additional diagnostic testing is necessary.
References
- The PIOPED Investigators. Value of the ventilation/perfusion scan in acute pulmonary embolism: results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990;263:2753-2759.
- Wells PS, Anderson DR, Rodger M, et al. Excluding pulmonary embolism at the bedside without diagnostic imaging: management of patients with suspected pulmonary embolism presenting to the emergency department by using a simple clinical model and d-dimer. Ann Intern Med. 2001 Jul 17;135(2):98-107.
- Perrier A, Roy PM, Sanchez O, et al. Multidetector-row computed tomography in suspected pulmonary embolism. N Engl J Med. 2005 Apr;352(17):1760-1768.
- van Belle A, Buller HR, Huisman MV, et al. Effectiveness of managing suspected pulmonary embolism using an algorithm combining clinical probability, D-dimer testing, and computed tomography. JAMA. 2006 Jan 11;295(2):172-179.
Classic Article:
PIOPED Investigators
The PIOPED Investigators. Value of ventilation/perfusion scan in acute pulmonary embolism: results of the prospective investigation of pulmonary embolism diagnosis (PIOPED). JAMA. 1990;263:2753-2759.
Background
The risk of untreated pulmonary embolism requires either the diagnosis or the exclusion of this diagnosis when clinical suspicion exists. The reference test for pulmonary embolism, standard pulmonary angiography, is invasive and expensive, and carries with it a measurable procedural risk.
Non-invasive diagnostic tests, including ventilation/perfusion (V/Q) scintigraphy, have been used to detect perfusion defects consistent with pulmonary embolism, though the performance characteristics of this diagnostic test were not well known prior to 1990. This study was designed to evaluate the sensitivity and specificities of ventilation/perfusion lung scans for pulmonary embolism in the acute setting.
Methods
This prospective, multi-center study evaluated V/Q scintigraphy on a random sample of 931 patients. A composite reference standard was used because only 755 patients underwent scintigraphy and pulmonary angiography. Clinical follow-up and subsequent diagnostic testing were employed in untreated patients with low clinical probabilities of pulmonary embolism who did not undergo angiography. Clinical assessment of the probability of pulmonary embolism was determined on the basis of the clinician’s judgment, without systematic prediction rules.
Results
Almost all patients with pulmonary embolism had abnormal ventilation/perfusion lung scans of high, intermediate, or low probability. Unfortunately, most patients without pulmonary embolism also had abnormal studies, limiting the utility of this test. Clinical follow-up and angiography revealed that pulmonary embolism occurred among 12% of patients with low-probability scans.
Conclusions
V/Q scintigraphy is useful in establishing or excluding the diagnosis of pulmonary embolism in only a minority of patients, where clinical suspicion of pulmonary embolism is concordant with the diagnostic test findings. The likelihood of pulmonary embolism in patients with a high pre-test probability of pulmonary embolism and a high probability scan is 95%, while in low probability patients with a low probability or normal scan the probability is 4% or 2%, respectively.
Commentary
This original PIOPED study established the diagnostic characteristics of V/Q scintigraphy and demonstrated, for the first time, evidence of the role of clinical assessment and prior probability in a diagnostic strategy for pulmonary embolism. Although subsequent studies have significantly advanced our knowledge of clinical prediction and diagnostic strategies in venous thromboembolism, the first PIOPED study continues to serve as an example of a high-quality, multi-center diagnostic test study utilizing a composite reference standard in a difficult-to-study disease. Unfortunately, the results of this study demonstrated that V/Q scintigraphy performs well for only a minority of patients. The majority of patients (72%) had clinical probabilities of pulmonary embolism and ventilation/perfusion scan results, which yielded post-test probabilities of 15-86%, leaving, in many cases, enough remaining diagnostic uncertainty to warrant additional testing.—TO TH
New Tools of the Trade
Andrew Fishmann, MD, FCCP, FACP, listened for more than an hour as an award-winning hospitalist talked about strategies to improve communication between physicians and administrators. Brian Bossard, MD, FACP, FHM, gleaned lessons from case studies of how two HM groups (HMGs) navigated the adolescent years. And dozens more hospitalists sat still for the bulk of an hour as they were given a nuts-and-bolts tutorial of HM finances.
Welcome to SHM’s new practice management track, a three-day series of training sessions that debuted at HM09 and focuses on the inner workings of HMGs—from startup to coding and billing to expansion. “There’s always a quality track, there’s always a clinical track, and there’s always a leadership track,” says Kimberly Dickinson, vice president of operations for Cogent Healthcare in Brentwood, Tenn., and an SHM Career Satisfaction Task Force member. “This was trying to bring together some of the nonclinical aspects.”
The experiment drew positive reviews, if rooms crowded with physicians—some lined up along the walls—are any indication. Dr. Fishmann, a Cogent co-founder who is practicing as a hospitalist with California Lung Associates in Los Angeles, says the courses give younger hospitalists different perspectives. “It’s taking doctors a little out of the hospital and putting them in the boardroom,” he says. “You don’t have a choice. … You need to be involved and have more input.”
The practice management courses are structured to give a broad overview of nearly every facet of opening and operating an HMG. One popular course, which ran nearly 15 minutes long because of participant questions, focused on managing HMG growth. Another session spent more than an hour taking physicians through comanagement issues that arise during collaborations with surgeons.
Real-Life Experiences
Most of the courses were led by familiar SHM leaders. But several attendees said they enjoyed sessions that were led by rank-and-file hospitalists and administrators who live the front-line struggles every day.
The “Case Studies in Managing Program Growth” course featured detailed explanations of the growing pains of HM programs in Michigan and Massachusetts. In the former case, Carole Montgomery, MD, talked about how the Michigan Medical PC group she helped start in western Michigan struggled to formalize certain procedures when it signed its first contract with a hospital in 2002. The group doubled its hospitalist roster and instantly went from an informal HM practice in which everyone knew each other and had relatively similar opinions to a business in which it was unclear who would make major decisions.
In response, Dr. Montgomery’s group crafted a mission statement, created a hospitalist executive committee to make routine operational decisions, and changed how it negotiated contracts with hospitals. Soon after, the group fired its first physician. Last year, the group instituted “internal governance guidelines” to make management decisions clearer. Dr. Montgomery says each of the developments taught her that solving major issues takes patience and a willingness to continually adapt. “I thought I was done each time,” she says. “Now I realize it’s an interactive process.”
Results-Oriented
Peter Short, MD, FAAP, CPE, medical director of Northeast Hospital Corp., shared his recent struggles to hire one hospitalist and expand night coverage. Hospitalists in Dr. Short’s service, who practice at Beverly Hospital in Beverly, Mass., resisted the change at first, not wanting to add additional night-shift responsibility. Dr. Short also spoke about financial concerns to the group and the hospital. After explaining the pros and cons of hiring a sixth rounder, the hospitalists embraced the idea. So far, hospital administrators have had zero complaints, as the first three months of data show a reduction in length of stay by 0.3 days and an average cost decrease of roughly $500 per case.
Dr. Short also explained an unexpected byproduct of the hiring: Night work proved so popular that the hospitalists have demanded that they receive a specified number of overnight shifts a year. “They went from not wanting it to fighting for it,” Dr. Short says proudly. “There is no one-size-fits-all for hospital programs.”
Dr. Bossard, who founded Inpatient Physician Associates in Lincoln, Neb., says the types of detail-oriented presentations made by Drs. Montgomery and Short are useful if physicians learn lessons they can take home and adapt to their practice. “If it’s a lot of smoke without an action item or a bullet point we can latch on to,” he says, “it’s not as helpful.”
Richard Quinn is a freelance writer based in New Jersey.
Andrew Fishmann, MD, FCCP, FACP, listened for more than an hour as an award-winning hospitalist talked about strategies to improve communication between physicians and administrators. Brian Bossard, MD, FACP, FHM, gleaned lessons from case studies of how two HM groups (HMGs) navigated the adolescent years. And dozens more hospitalists sat still for the bulk of an hour as they were given a nuts-and-bolts tutorial of HM finances.
Welcome to SHM’s new practice management track, a three-day series of training sessions that debuted at HM09 and focuses on the inner workings of HMGs—from startup to coding and billing to expansion. “There’s always a quality track, there’s always a clinical track, and there’s always a leadership track,” says Kimberly Dickinson, vice president of operations for Cogent Healthcare in Brentwood, Tenn., and an SHM Career Satisfaction Task Force member. “This was trying to bring together some of the nonclinical aspects.”
The experiment drew positive reviews, if rooms crowded with physicians—some lined up along the walls—are any indication. Dr. Fishmann, a Cogent co-founder who is practicing as a hospitalist with California Lung Associates in Los Angeles, says the courses give younger hospitalists different perspectives. “It’s taking doctors a little out of the hospital and putting them in the boardroom,” he says. “You don’t have a choice. … You need to be involved and have more input.”
The practice management courses are structured to give a broad overview of nearly every facet of opening and operating an HMG. One popular course, which ran nearly 15 minutes long because of participant questions, focused on managing HMG growth. Another session spent more than an hour taking physicians through comanagement issues that arise during collaborations with surgeons.
Real-Life Experiences
Most of the courses were led by familiar SHM leaders. But several attendees said they enjoyed sessions that were led by rank-and-file hospitalists and administrators who live the front-line struggles every day.
The “Case Studies in Managing Program Growth” course featured detailed explanations of the growing pains of HM programs in Michigan and Massachusetts. In the former case, Carole Montgomery, MD, talked about how the Michigan Medical PC group she helped start in western Michigan struggled to formalize certain procedures when it signed its first contract with a hospital in 2002. The group doubled its hospitalist roster and instantly went from an informal HM practice in which everyone knew each other and had relatively similar opinions to a business in which it was unclear who would make major decisions.
In response, Dr. Montgomery’s group crafted a mission statement, created a hospitalist executive committee to make routine operational decisions, and changed how it negotiated contracts with hospitals. Soon after, the group fired its first physician. Last year, the group instituted “internal governance guidelines” to make management decisions clearer. Dr. Montgomery says each of the developments taught her that solving major issues takes patience and a willingness to continually adapt. “I thought I was done each time,” she says. “Now I realize it’s an interactive process.”
Results-Oriented
Peter Short, MD, FAAP, CPE, medical director of Northeast Hospital Corp., shared his recent struggles to hire one hospitalist and expand night coverage. Hospitalists in Dr. Short’s service, who practice at Beverly Hospital in Beverly, Mass., resisted the change at first, not wanting to add additional night-shift responsibility. Dr. Short also spoke about financial concerns to the group and the hospital. After explaining the pros and cons of hiring a sixth rounder, the hospitalists embraced the idea. So far, hospital administrators have had zero complaints, as the first three months of data show a reduction in length of stay by 0.3 days and an average cost decrease of roughly $500 per case.
Dr. Short also explained an unexpected byproduct of the hiring: Night work proved so popular that the hospitalists have demanded that they receive a specified number of overnight shifts a year. “They went from not wanting it to fighting for it,” Dr. Short says proudly. “There is no one-size-fits-all for hospital programs.”
Dr. Bossard, who founded Inpatient Physician Associates in Lincoln, Neb., says the types of detail-oriented presentations made by Drs. Montgomery and Short are useful if physicians learn lessons they can take home and adapt to their practice. “If it’s a lot of smoke without an action item or a bullet point we can latch on to,” he says, “it’s not as helpful.”
Richard Quinn is a freelance writer based in New Jersey.
Andrew Fishmann, MD, FCCP, FACP, listened for more than an hour as an award-winning hospitalist talked about strategies to improve communication between physicians and administrators. Brian Bossard, MD, FACP, FHM, gleaned lessons from case studies of how two HM groups (HMGs) navigated the adolescent years. And dozens more hospitalists sat still for the bulk of an hour as they were given a nuts-and-bolts tutorial of HM finances.
Welcome to SHM’s new practice management track, a three-day series of training sessions that debuted at HM09 and focuses on the inner workings of HMGs—from startup to coding and billing to expansion. “There’s always a quality track, there’s always a clinical track, and there’s always a leadership track,” says Kimberly Dickinson, vice president of operations for Cogent Healthcare in Brentwood, Tenn., and an SHM Career Satisfaction Task Force member. “This was trying to bring together some of the nonclinical aspects.”
The experiment drew positive reviews, if rooms crowded with physicians—some lined up along the walls—are any indication. Dr. Fishmann, a Cogent co-founder who is practicing as a hospitalist with California Lung Associates in Los Angeles, says the courses give younger hospitalists different perspectives. “It’s taking doctors a little out of the hospital and putting them in the boardroom,” he says. “You don’t have a choice. … You need to be involved and have more input.”
The practice management courses are structured to give a broad overview of nearly every facet of opening and operating an HMG. One popular course, which ran nearly 15 minutes long because of participant questions, focused on managing HMG growth. Another session spent more than an hour taking physicians through comanagement issues that arise during collaborations with surgeons.
Real-Life Experiences
Most of the courses were led by familiar SHM leaders. But several attendees said they enjoyed sessions that were led by rank-and-file hospitalists and administrators who live the front-line struggles every day.
The “Case Studies in Managing Program Growth” course featured detailed explanations of the growing pains of HM programs in Michigan and Massachusetts. In the former case, Carole Montgomery, MD, talked about how the Michigan Medical PC group she helped start in western Michigan struggled to formalize certain procedures when it signed its first contract with a hospital in 2002. The group doubled its hospitalist roster and instantly went from an informal HM practice in which everyone knew each other and had relatively similar opinions to a business in which it was unclear who would make major decisions.
In response, Dr. Montgomery’s group crafted a mission statement, created a hospitalist executive committee to make routine operational decisions, and changed how it negotiated contracts with hospitals. Soon after, the group fired its first physician. Last year, the group instituted “internal governance guidelines” to make management decisions clearer. Dr. Montgomery says each of the developments taught her that solving major issues takes patience and a willingness to continually adapt. “I thought I was done each time,” she says. “Now I realize it’s an interactive process.”
Results-Oriented
Peter Short, MD, FAAP, CPE, medical director of Northeast Hospital Corp., shared his recent struggles to hire one hospitalist and expand night coverage. Hospitalists in Dr. Short’s service, who practice at Beverly Hospital in Beverly, Mass., resisted the change at first, not wanting to add additional night-shift responsibility. Dr. Short also spoke about financial concerns to the group and the hospital. After explaining the pros and cons of hiring a sixth rounder, the hospitalists embraced the idea. So far, hospital administrators have had zero complaints, as the first three months of data show a reduction in length of stay by 0.3 days and an average cost decrease of roughly $500 per case.
Dr. Short also explained an unexpected byproduct of the hiring: Night work proved so popular that the hospitalists have demanded that they receive a specified number of overnight shifts a year. “They went from not wanting it to fighting for it,” Dr. Short says proudly. “There is no one-size-fits-all for hospital programs.”
Dr. Bossard, who founded Inpatient Physician Associates in Lincoln, Neb., says the types of detail-oriented presentations made by Drs. Montgomery and Short are useful if physicians learn lessons they can take home and adapt to their practice. “If it’s a lot of smoke without an action item or a bullet point we can latch on to,” he says, “it’s not as helpful.”
Richard Quinn is a freelance writer based in New Jersey.
Reporting Hospital Quality
Acknowledging striking deficiencies in the quality and safety of healthcare, the Institute of Medicine, policy makers, and payors have called for transformation of the US healthcare system.1 Public reporting of hospital performance is one key strategy for accelerating improvement2 and may improve quality in several ways. First, feedback about performance relative to peers may stimulate quality improvement activities by appealing to professionalism. Second, the desire to preserve one's reputation by not appearing on a list of poor performers may be a powerful incentive. Finally, patients and referring providers could use reports to select high‐quality hospitals, thereby shifting care from low‐quality to high‐quality hospitals and stimulating quality improvement efforts to maintain or enhance market share.
Almost 20 years after New York and Pennsylvania began reporting cardiac surgery outcomes,3 the evidence that public reporting improves healthcare quality is equivocal.4 Moreover, stakeholders have embraced public reporting to differing degrees. Public reporting does lead to greater engagement in quality improvement activities,58 and additional financial incentives provide modest incremental benefits.9 Purchasers, too, are starting to pay attention.10 In New York State, payors appear to contract more with high‐quality surgeons and avoid poorly performing outliers.11 Some payors are creating tiered systems, assigning higher patient copayments for hospitals with poor quality metrics. These new systems have not been rigorously studied and should raise concern among hospitals.12
In contrast to hospitals and payors, patients have been slow to embrace public reporting. In a survey of coronary artery bypass graft (CABG) patients in Pennsylvania, only 2% said that public reporting of mortality rates affected their decision making.13 Eight years later, only 11% of patients sought information about hospitals before deciding on elective major surgery,14 although a majority of patients in both studies expressed interest in the information. It is not clear whether recent proliferation of information on the internet will change patient behavior, but to date public reporting appears not to effect market share.5, 15, 16
Barriers to patients' use of public reporting include difficulty accessing the information, lack of trust, information that is not salient, and data that are difficult to interpret.17 In the absence of consensus on what or how to report, a growing number of organizations, including state and federal government, accrediting bodies, private foundations, and for‐profit companies report a variety of measures relating to structure, processes, and outcomes. Although these sites purport to target consumers, they sometimes offer conflicting information18 and are not easily interpreted by lay readers.19
To realize the benefits of public reporting, and minimize the unintended consequences, rating systems must report salient information in a way that is comprehensible to patients and trusted by the doctors who advise them. At the same time, they should be fair to hospitals and offer useful data for quality improvement. We offer 10 recommendations for improving the public reporting of healthcare quality information: 5 describing what to report and 5 detailing how it should be reported (Figure 1). We also examine 3 leading performance reporting programs to see how well they implement these recommendations.

Recommendations to Make Data Salient for Patients
1. Prioritize Elective Procedures
Hospital quality is not uniform across conditions.2 For data to be salient, then, it should be disease‐specific and focus on common elective procedures, for which consumer choice is possible. Table 1 compares 3 popular reporting services. Hospital Compare, produced by the Center for Medicare Services (CMS, US Department of Health and Human Services, Washington, DC), provides process of care measures for 4 conditions, 3 of which are not elective. The fourth, surgical infection prevention, contains 5 measures3 related to perioperative antibiotics and 2 related to thromboembolism prophylaxisfor all surgical cases. Recently, more conditions have been added, but reports are limited to the number of cases and mean Medicare charge. By year 2011, however, Hospital Compare will offer many new measures, including rates of central line infection, ventilator‐associated pneumonia, and surgical site infection. HealthGrades, a private company, offers comparative mortality rates on over 30 diagnoses, of which 15 can be considered elective, at least some of the time. Only the Leapfrog group, an industry consortium, focuses exclusively on elective procedures, offering volume measures on 7 and outcome measures on 2.
| Rule | Hospital Compare | HealthGrades | Leapfrog | |||
|---|---|---|---|---|---|---|
| ||||||
| 1. Prioritize elective procedures | Yes | 22/28 at least partially elective | Yes | 15/31 at least partially elective | Yes | 7/8 elective |
| 2. Include quality of life and outcome data, if possible | Yes | Mortality for AMI and CHF | Yes | Mortality or complications* | Yes | Outcomes for CABG, PCI, and AVR |
| 3. Include standardized patient satisfaction and service measures | Yes | HCAHPS | No | No | ||
| 4. Offer composite measures that are weighted and evidence‐based | No | No | Specialty excellence award, not evidence‐based | No | ||
| 5. Costs comparisons should include patient prices | Yes | Average Medicare payment | Yes | Charges, health plan and Medicare costs available for a fee | No | |
| 6. Adjust outcomes for severity and risk | Yes | Methodology published on website | Yes | Methodology not public | Yes | Various methodologies published or referenced on website |
| 7. Identify differences not due to chance | Yes | Compares mortality to national mean | Yes | Compares mortality or complications to mean | Yes | Compares mortality to national mean |
| 8. Standardize reporting periods | October 2005 to September 2006 | 2004‐2006 | 12‐24 months, ending 12/31/07 or 6/30/08 | |||
| 9. Avoid use of nonvalidated administrative data | Yes | None used | No | Uses PSIs for safety rating | Yes | None used |
| 10. Utilization rates should be evidence‐based | No | Surgical case volume of Medicare patients | No | Includes Caesarian‐section rates | Yes | Some case volume rates are evidence‐based |
2. Include Quality of Life and Outcome Data
Outcomes are more valuable to patients than process measures, but the risk adjustment needed to compare outcomes requires considerable effort. So far, public reporting of risk‐adjusted outcomes has been limited almost exclusively to mortality. Yet a patient contemplating knee replacement surgery would find no meaningful difference in mortalitythere were only 510 deaths nationally in year 200620but might be interested in whether patients return to full mobility after surgery, and all patients should compare rates of nosocomial infections. For some low‐risk procedures, HealthGrades Inc. (Golden, CO) includes a composite measure of major complications, including complication of an orthopedic implant, stroke, cardiac arrest, excessive bleeding, and some types of infection; CMS will soon add rates of infection and readmission.
3. Include Measures of Patient Experience, Such as Satisfaction and Service Measures
Beyond outcomes, patients want to know about the experience of others.21 Satisfaction surveys should be standardized and made disease‐specific, since patients' experiences may differ between the cardiology suite and the delivery unit. Questions could address the attentiveness of the nursing staff, how well privacy was respected, how easy it was to deal with insurance issues, whether patients were promptly informed of test results, and whether the care team answered questions fully. Medicare has begun reporting patient satisfaction using the Hospital Consumer Assessment of Healthcare Providers (HCAHPS) survey on Hospital Compare, but the data are not disease‐specific and audit a very small number of patients from each institution. Other services are unlikely to perform their own surveys, as multiple surveys would prove burdensome. Social networking sites that allow patients to post their own personal reviews of hospitals and doctors offer an additional if less reliable dimension to traditional public reporting. Such sites are already transforming the market for other industries, such as travel.22
4. Offer Composite Measures That Are Weighted and Evidence‐Based
Interpreting multiple measures, some of which are more important than others, and some of which have better evidence than others, is difficult for health care providers and may be impossible for patients. Is it more important to get aspirin on arrival or at discharge? Also, how does a patient weigh a 1% difference in the number of heart attack patients who get aspirin on arrival against a 14% difference in those who are offered smoking cessation? Because patients may be overwhelmed by data,23 public reports should include evidence‐based, weighted measures of overall care for a given condition, with higher weights attached to those process measures most likely to have clinical benefit, and careful attention to visual representations that convey relative differences.19, 23 More sophisticated measures should be developed to guard against overuse. For example, while hospitals should be rewarded for providing vaccination, they should be penalized for vaccinating the same patient twice.
None of the services we examined provides weighted outcomes. Leapfrog (The Leapfrog Group, Washington, DC) offers a composite snapshot containing 9 pie charts, divided into 4 leaps. The 6 pies representing high‐risk procedures are of equal size, even though 2 of these, esophagectomy and pancreatic resection represent very rare surgeries, even at major medical centers. From a visual perspective, however, these are equivalent to having computerized physician order entry and full‐time intensive care unit staffing, which affect thousands more patients. Similarly, in determining pay‐for‐performance measures, CMS created a composite based on the total number of opportunities of all interventions, weighting all measures equally. Because no validated weighting measures exist, future research will be necessary to achieve this goal. Also, none of the evidence‐based measures contained safeguards against overtreatment.
5. Cost Comparisons Should Include Patient Prices
In an era of patient copayments and deductibles, consumers are increasingly aware of costs. For patients with very high deductible plans or no health insurance, hospital fees are a common cause of bankruptcy.24 Several public reporting agencies, including Hospital Compare and HealthGrades have incorporated Medicare costs into their reported measures, but these have little connection to what patients actually pay. Health sites aimed at consumers should publish the average patient copayment.
Recommendations to Ensure That Data Reflects Hospital Quality
6. Adjust Outcomes for Severity and Risk
Not all bypass operations are the same and not all patients are at equal risk. More difficult operations (eg, CABG for a patient with a previous bypass) will have more complications; similarly, patients with serious comorbidities will experience worse outcomes. Since hospitals which specialize in a procedure will attract complicated cases and higher risk patients, it is important to adjust outcomes to account for these differences. Otherwise, hospitals and surgeons may be discouraged from taking difficult cases. Outside of cardiac surgery, most risk adjustment systems use administrative claims data but vary dramatically in the numbers of variables considered and the underlying proprietary models, which are often criticized as being black boxes that yield discordant results.25 Thus, a hospital's mortality may appear below expected by 1 system and above expected by another. Instead, risk adjustment systems should include clinical data abstracted from patient records using standardized data definitions. Although costly to collect, clinical data offer more predictive information than do administrative data. For example, for heart failure patients undergoing CABG, the ejection fraction predicts mortality better than many stable comorbid diagnoses. A single transparent risk‐adjustment system should be recognized as the industry standard. The American College of Surgeons' standardized risk‐adjusted outcome reporting for the National Surgical Quality Improvement Program (NSQIP) is a good example of such an effort.
7. Identify Differences Not Due to Chance
As a result of random variation, during any period, some hospitals will appear better than average and others worse. Statistical tests should be employed to identify hospitals that differ from the mean, and to allow consumers to compare 2 hospitals directly, with appropriate caveats when the hospitals serve very different patient populations. Medicare's mortality rating system for myocardial infarction identifies only 17 hospitals in the nation as better than average and 7 as worse, out of 4,500 institutions. HealthGrades compares hospitals' actual mortality or complication rates to their predicted rates based on disease‐specific logistic regression models and reports whether the hospital is statistically better or worse than predicted. Hospitals are not compared directly to one another. Given the rarity of mortality in most procedures, other outcome measures will be necessary to distinguish among hospitals.26
8. Standardize Reporting Periods
In a world of continuous quality improvement, public reporting should represent a hospital's recent performance, but reporting periods also need to be long enough to provide a stable estimate of infrequent events, especially at low‐volume institutions. In contrast, the lag time between the end of the reporting period and public availability should be kept to a minimum. We found that reporting periods varied from 1 to 3 years, and did not always cover the same years for all conditions, even on the same website. Some data were 3 years old. Patients will have a hard time making decisions on data that is 1 year old, and hospitals will have little incentive to make improvements that will not be acknowledged for years.
9. Avoid Use of Nonvalidated Administrative Data
Administrative data collected for billing purposes, unlike most clinical data, are already in electronic format, and can inexpensively produce quality rankings using validated models.27 In contrast, screening tools, such as the Agency for Healthcare Research and Quality's patient safety indicators (PSIs), were designed to identify potential quality problems, such as postoperative deep vein thrombosis, for internal quality improvement. Cases identified by the PSI software require additional chart review,28, 29 and should not be used as quality indicators. Even so, HealthGrades reports PSIs and some insurers use them in pay‐for‐performance initiatives. Improvements in PSIs, including present‐on‐admission coding, may increase accuracy,30 but these measures need to be validated before they can be adopted for public reporting.
10. Utilization Rates Should Be Evidence‐Based
Although utilization rates for most procedures vary as much as 2‐fold by state or institution, there is little evidence for a best rate. Nevertheless, HealthGrades reports utilization rates for several obstetrical procedures. At present, there are no standards for these, and it is possible that utilization could be too low in some places. Further research is needed; until then, utilization should not purport to measure quality.
Discussion
The growing commitment to making hospital performance data public could transform the quality and safety of care in the US, introducing competition on quality and price and fostering informed consumer choice. To date, the promise of public reporting remains only partially fulfilled. Few hospitals have done more than comply with regulatory mandates and payer incentives, and consumers have failed to respond. To capture the full benefits of public reporting, we have made 10 recommendations to benefit patients and better engage hospitals. We suggest that reporting be patient‐centered, with an emphasis on making the data useful, meaningful, important, interpretable, and relevant. At the same time, hospitals, which are being judged on their performance, should have a level playing field, with measures that are timely, consistent, severity‐adjusted, evidence‐based, and which foster good clinical care. Of the 3 services we examined, Hospital Compare came closest to meeting these recommendations.
Although this blueprint for public reporting is easy to draft, it is challenging to implement. In particular, some of our suggestions, such as the one regarding risk adjustment, may not currently be feasible, because the complexity and cost of collecting clinical data, even in the era of electronic medical records, may be prohibitive. Until such data are readily available, it may be preferable to report nothing at all, rather than report data that are misleading. In the rush to make hospitals accountable, enthusiasm has often outstripped science,31 and several measures have had to be revised for unintended consequences.32
Any initiative to improve public reporting should have the buy‐in of all stakeholders, but particularly hospitals, which stand to benefit in several ways. By receiving regular feedback, they can focus on improving care, becoming better organizations. These improvements may be rewarded through direct compensation (pay‐for‐performance), decreased costs from complications, or increased market share. Hospitals will be more engaged if the data reflect actual quality, are adequately adjusted for severity, and acknowledge the role of chance. Otherwise, they will merely comply, or worse, look for opportunities to game the system. To succeed, public reporting needs to involve hospitals in establishing standards for reporting and validation, as well as auditing procedures to prevent fraud.33 The Hospital Quality Alliance (HQA, Washington, DC), a first step in this direction, at present has few measures. NSQIP (American College of Surgeons, Chicago, IL) is perhaps a better example of hospitals cooperating to set measurement standards to promote best‐practices. Public release of NSQIP data might accelerate progress. Alternatively, the National Quality Forum (NQF, Washington, DC) could expand its role from endorsing quality measures to include standardizing the way these measures are used in public reporting.
Still, if you build it, will they come? To date, public reporting has not been embraced by the public, despite its stated interest in the information. Several explanations could be offered. First, we may be presenting the wrong data. Process measures and mortality rates are important but represent abstract concepts for most patients. Surveys tell us that patients value most the experiences of other patients.14, 21 They want to know whether their pain will be controlled, whether the doctor will listen to them, whether the nurse will come when they call. The recent advent of the HCAHPS survey (AHRQ, Washington, DC) is another positive step. Stratifying the results by diagnosis and adding a few diagnosis‐specific questions would make HCAHPS even more valuable. Second, the data may not be readily available. Although most public reporting is done on the web, older patients who are deciding about hospitals may not have Internet access. Some reports are still proprietary, and cost could present another obstacle. Finally, even if freely‐available and patient‐centered, the results may not be interpretable by physicians, let alone patients.34
If public reporting is to succeed, it will require measures that better reflect patients' concerns. In order to collect the massive amounts of data required and present them in a timely fashion, better electronic record systems will be necessary. But these are no panacea; others have noted that the Department of Veterans Affairs, a leader in electronic records, still invests considerable time and money to review charts for NSQIP.35 Given the value that Americans place on transparency in other facets of their lives, it is clear that public reporting is here to stay. While much progress has been made over the past 5 years, additional research is needed to better measure quality from the patient's perspective, and to determine how this information can be used to help guide decision‐making, and to reward hospitals for offering the highest‐quality care.
Acknowledgements
The authors thank Kenneth Flax for his help with an earlier version of this manuscript.
- Committee on Quality of Health Care in America IoM.Crossing the Quality Chasm: A New Health System for the 21st Century.Washington, DC:National Academy Press;2001.
- ,,,.Care in U.S. hospitals: the Hospital Quality Alliance program.N Engl J Med.2005;353(3):265–274.
- .Achieving and sustaining improved quality: lessons from New York state and cardiac surgery.Health Aff. 20022002;21(4):40–51.
- ,,,,.Systematic review: the evidence that publishing patient care performance data improves quality of care.Ann Intern Med.2008;148(2):111–123.
- ,,.Hospital performance reports: impact on quality, market share, and reputation.Health Aff (Millwood).2005;24(4):1150–1160.
- ,,.Does publicizing hospital performance stimulate quality improvement efforts?Health Aff (Millwood).2003;22(2):84–94.
- ,,,,.Improving the outcomes of coronary artery bypass surgery in New York State.JAMA.1994;271(10):761–766.
- ,,.Declines in hospital mortality associated with a regional initiative to measure hospital performance.Am J Med Qual.1997;12(2):103–112.
- ,,, et al.Public reporting and pay for performance in hospital quality improvement.N Engl J Med.2007;356(5):486–496.
- ,,,,,.Do quality report cards play a role in HMOs' contracting practices? Evidence from New York State.Health Serv Res.2000;35(1 Pt 2):319–332.
- ,,,.Quality of cardiac surgeons and managed care contracting practices.Health Serv Res.2002;37(5):1129–1144.
- ,,,,,.Using performance data to identify preferred hospitals.Health Serv Res.2007;42(6 Pt 1):2109–2119; discussion 2294–2323.
- ,.Use of public performance reports: a survey of patients undergoing cardiac surgery.JAMA.1998;279(20):1638–1642.
- ,,.How do elderly patients decide where to go for major surgery? Telephone interview survey.BMJ.2005;331(7520):821.
- ,,,,,.The effect of publicly reporting hospital performance on market share and risk‐adjusted mortality at high‐mortality hospitals.Med Care.2003;41(6):729–740.
- ,.The predictive accuracy of the New York State coronary artery bypass surgery report‐card system.Health Aff (Millwood).2006;25(3):844–855.
- ,.Publicly disclosed information about the quality of health care: response of the US public.Qual Saf Health Care.2001;10(2):96–103.
- ,,,,.Choosing the best hospital: the limitations of public reporting of hospital quality.Health Aff (Millwood).2008;27(6):1680–1687.
- ,.Will quality report cards help consumers?Health Aff (Millwood).1997;16(3):218–228.
- Agency for Healthcare Research and Quality. HCUPnet, Healthcare Cost and Utilization Project. Available at: http://hcupnet.ahrq.gov. Accessed January 2009.
- ,,.Recovering from cardiac surgery: what patients want you to know.Am J Crit Care.2002;11(4):333–343.
- Trip Advisor. Available at: http://www.tripadvisor.com. Accessed January 2009.
- ,,,,.Less is more in presenting quality information to consumers.Med Care Res Rev.2007;64(2):169–190.
- ,,,.MarketWatch: illness and injury as contributors to bankruptcy.Health Aff (Millwood)2005;(Suppl Web Exclusives):W5‐63–W5‐73.
- .The Lake Wobegon effect: when all the patients are sicker.Am J Med Qual.2006;21(6):365–366.
- ,,.Surgical mortality as an indicator of hospital quality: the problem with small sample size.JAMA.2004;292(7):847–851.
- ,,, et al.An administrative claims model suitable for profiling hospital performance based on 30‐day mortality rates among patients with heart failure.Circulation.2006;113(13):1693–1701.
- ,,,.Can administrative data be used to compare postoperative complication rates across hospitals?Med Care.2002;40(10):856–867.
- ,,,,.Impact of diagnosis‐timing indicators on measures of safety, comorbidity, and case mix groupings from administrative data sources.Med Care.2007;45(8):781–788.
- ,,,,.Do the AHRQ patient safety indicators flag conditions that are present at the time of hospital admission?Med Care.2008;46(5):516–522.
- ,,.The tension between needing to improve care and knowing how to do it.N Engl J Med.2007;357(6):608–613.
- ,,,.Public reporting of antibiotic timing in patients with pneumonia: lessons from a flawed performance measure.Ann Intern Med.2008;149(1):29–32.
- ,,.The GAAP in quality measurement and reporting.JAMA.2007;298(15):1800–1802.
- ,,,.Consumer competencies and the use of comparative quality information: it isn't just about literacy.Med Care Res Rev.2007;64(4):379–394.
- .Performance measurement in search of a path.N Engl J Med.2007;356(9):951–953.
Acknowledging striking deficiencies in the quality and safety of healthcare, the Institute of Medicine, policy makers, and payors have called for transformation of the US healthcare system.1 Public reporting of hospital performance is one key strategy for accelerating improvement2 and may improve quality in several ways. First, feedback about performance relative to peers may stimulate quality improvement activities by appealing to professionalism. Second, the desire to preserve one's reputation by not appearing on a list of poor performers may be a powerful incentive. Finally, patients and referring providers could use reports to select high‐quality hospitals, thereby shifting care from low‐quality to high‐quality hospitals and stimulating quality improvement efforts to maintain or enhance market share.
Almost 20 years after New York and Pennsylvania began reporting cardiac surgery outcomes,3 the evidence that public reporting improves healthcare quality is equivocal.4 Moreover, stakeholders have embraced public reporting to differing degrees. Public reporting does lead to greater engagement in quality improvement activities,58 and additional financial incentives provide modest incremental benefits.9 Purchasers, too, are starting to pay attention.10 In New York State, payors appear to contract more with high‐quality surgeons and avoid poorly performing outliers.11 Some payors are creating tiered systems, assigning higher patient copayments for hospitals with poor quality metrics. These new systems have not been rigorously studied and should raise concern among hospitals.12
In contrast to hospitals and payors, patients have been slow to embrace public reporting. In a survey of coronary artery bypass graft (CABG) patients in Pennsylvania, only 2% said that public reporting of mortality rates affected their decision making.13 Eight years later, only 11% of patients sought information about hospitals before deciding on elective major surgery,14 although a majority of patients in both studies expressed interest in the information. It is not clear whether recent proliferation of information on the internet will change patient behavior, but to date public reporting appears not to effect market share.5, 15, 16
Barriers to patients' use of public reporting include difficulty accessing the information, lack of trust, information that is not salient, and data that are difficult to interpret.17 In the absence of consensus on what or how to report, a growing number of organizations, including state and federal government, accrediting bodies, private foundations, and for‐profit companies report a variety of measures relating to structure, processes, and outcomes. Although these sites purport to target consumers, they sometimes offer conflicting information18 and are not easily interpreted by lay readers.19
To realize the benefits of public reporting, and minimize the unintended consequences, rating systems must report salient information in a way that is comprehensible to patients and trusted by the doctors who advise them. At the same time, they should be fair to hospitals and offer useful data for quality improvement. We offer 10 recommendations for improving the public reporting of healthcare quality information: 5 describing what to report and 5 detailing how it should be reported (Figure 1). We also examine 3 leading performance reporting programs to see how well they implement these recommendations.

Recommendations to Make Data Salient for Patients
1. Prioritize Elective Procedures
Hospital quality is not uniform across conditions.2 For data to be salient, then, it should be disease‐specific and focus on common elective procedures, for which consumer choice is possible. Table 1 compares 3 popular reporting services. Hospital Compare, produced by the Center for Medicare Services (CMS, US Department of Health and Human Services, Washington, DC), provides process of care measures for 4 conditions, 3 of which are not elective. The fourth, surgical infection prevention, contains 5 measures3 related to perioperative antibiotics and 2 related to thromboembolism prophylaxisfor all surgical cases. Recently, more conditions have been added, but reports are limited to the number of cases and mean Medicare charge. By year 2011, however, Hospital Compare will offer many new measures, including rates of central line infection, ventilator‐associated pneumonia, and surgical site infection. HealthGrades, a private company, offers comparative mortality rates on over 30 diagnoses, of which 15 can be considered elective, at least some of the time. Only the Leapfrog group, an industry consortium, focuses exclusively on elective procedures, offering volume measures on 7 and outcome measures on 2.
| Rule | Hospital Compare | HealthGrades | Leapfrog | |||
|---|---|---|---|---|---|---|
| ||||||
| 1. Prioritize elective procedures | Yes | 22/28 at least partially elective | Yes | 15/31 at least partially elective | Yes | 7/8 elective |
| 2. Include quality of life and outcome data, if possible | Yes | Mortality for AMI and CHF | Yes | Mortality or complications* | Yes | Outcomes for CABG, PCI, and AVR |
| 3. Include standardized patient satisfaction and service measures | Yes | HCAHPS | No | No | ||
| 4. Offer composite measures that are weighted and evidence‐based | No | No | Specialty excellence award, not evidence‐based | No | ||
| 5. Costs comparisons should include patient prices | Yes | Average Medicare payment | Yes | Charges, health plan and Medicare costs available for a fee | No | |
| 6. Adjust outcomes for severity and risk | Yes | Methodology published on website | Yes | Methodology not public | Yes | Various methodologies published or referenced on website |
| 7. Identify differences not due to chance | Yes | Compares mortality to national mean | Yes | Compares mortality or complications to mean | Yes | Compares mortality to national mean |
| 8. Standardize reporting periods | October 2005 to September 2006 | 2004‐2006 | 12‐24 months, ending 12/31/07 or 6/30/08 | |||
| 9. Avoid use of nonvalidated administrative data | Yes | None used | No | Uses PSIs for safety rating | Yes | None used |
| 10. Utilization rates should be evidence‐based | No | Surgical case volume of Medicare patients | No | Includes Caesarian‐section rates | Yes | Some case volume rates are evidence‐based |
2. Include Quality of Life and Outcome Data
Outcomes are more valuable to patients than process measures, but the risk adjustment needed to compare outcomes requires considerable effort. So far, public reporting of risk‐adjusted outcomes has been limited almost exclusively to mortality. Yet a patient contemplating knee replacement surgery would find no meaningful difference in mortalitythere were only 510 deaths nationally in year 200620but might be interested in whether patients return to full mobility after surgery, and all patients should compare rates of nosocomial infections. For some low‐risk procedures, HealthGrades Inc. (Golden, CO) includes a composite measure of major complications, including complication of an orthopedic implant, stroke, cardiac arrest, excessive bleeding, and some types of infection; CMS will soon add rates of infection and readmission.
3. Include Measures of Patient Experience, Such as Satisfaction and Service Measures
Beyond outcomes, patients want to know about the experience of others.21 Satisfaction surveys should be standardized and made disease‐specific, since patients' experiences may differ between the cardiology suite and the delivery unit. Questions could address the attentiveness of the nursing staff, how well privacy was respected, how easy it was to deal with insurance issues, whether patients were promptly informed of test results, and whether the care team answered questions fully. Medicare has begun reporting patient satisfaction using the Hospital Consumer Assessment of Healthcare Providers (HCAHPS) survey on Hospital Compare, but the data are not disease‐specific and audit a very small number of patients from each institution. Other services are unlikely to perform their own surveys, as multiple surveys would prove burdensome. Social networking sites that allow patients to post their own personal reviews of hospitals and doctors offer an additional if less reliable dimension to traditional public reporting. Such sites are already transforming the market for other industries, such as travel.22
4. Offer Composite Measures That Are Weighted and Evidence‐Based
Interpreting multiple measures, some of which are more important than others, and some of which have better evidence than others, is difficult for health care providers and may be impossible for patients. Is it more important to get aspirin on arrival or at discharge? Also, how does a patient weigh a 1% difference in the number of heart attack patients who get aspirin on arrival against a 14% difference in those who are offered smoking cessation? Because patients may be overwhelmed by data,23 public reports should include evidence‐based, weighted measures of overall care for a given condition, with higher weights attached to those process measures most likely to have clinical benefit, and careful attention to visual representations that convey relative differences.19, 23 More sophisticated measures should be developed to guard against overuse. For example, while hospitals should be rewarded for providing vaccination, they should be penalized for vaccinating the same patient twice.
None of the services we examined provides weighted outcomes. Leapfrog (The Leapfrog Group, Washington, DC) offers a composite snapshot containing 9 pie charts, divided into 4 leaps. The 6 pies representing high‐risk procedures are of equal size, even though 2 of these, esophagectomy and pancreatic resection represent very rare surgeries, even at major medical centers. From a visual perspective, however, these are equivalent to having computerized physician order entry and full‐time intensive care unit staffing, which affect thousands more patients. Similarly, in determining pay‐for‐performance measures, CMS created a composite based on the total number of opportunities of all interventions, weighting all measures equally. Because no validated weighting measures exist, future research will be necessary to achieve this goal. Also, none of the evidence‐based measures contained safeguards against overtreatment.
5. Cost Comparisons Should Include Patient Prices
In an era of patient copayments and deductibles, consumers are increasingly aware of costs. For patients with very high deductible plans or no health insurance, hospital fees are a common cause of bankruptcy.24 Several public reporting agencies, including Hospital Compare and HealthGrades have incorporated Medicare costs into their reported measures, but these have little connection to what patients actually pay. Health sites aimed at consumers should publish the average patient copayment.
Recommendations to Ensure That Data Reflects Hospital Quality
6. Adjust Outcomes for Severity and Risk
Not all bypass operations are the same and not all patients are at equal risk. More difficult operations (eg, CABG for a patient with a previous bypass) will have more complications; similarly, patients with serious comorbidities will experience worse outcomes. Since hospitals which specialize in a procedure will attract complicated cases and higher risk patients, it is important to adjust outcomes to account for these differences. Otherwise, hospitals and surgeons may be discouraged from taking difficult cases. Outside of cardiac surgery, most risk adjustment systems use administrative claims data but vary dramatically in the numbers of variables considered and the underlying proprietary models, which are often criticized as being black boxes that yield discordant results.25 Thus, a hospital's mortality may appear below expected by 1 system and above expected by another. Instead, risk adjustment systems should include clinical data abstracted from patient records using standardized data definitions. Although costly to collect, clinical data offer more predictive information than do administrative data. For example, for heart failure patients undergoing CABG, the ejection fraction predicts mortality better than many stable comorbid diagnoses. A single transparent risk‐adjustment system should be recognized as the industry standard. The American College of Surgeons' standardized risk‐adjusted outcome reporting for the National Surgical Quality Improvement Program (NSQIP) is a good example of such an effort.
7. Identify Differences Not Due to Chance
As a result of random variation, during any period, some hospitals will appear better than average and others worse. Statistical tests should be employed to identify hospitals that differ from the mean, and to allow consumers to compare 2 hospitals directly, with appropriate caveats when the hospitals serve very different patient populations. Medicare's mortality rating system for myocardial infarction identifies only 17 hospitals in the nation as better than average and 7 as worse, out of 4,500 institutions. HealthGrades compares hospitals' actual mortality or complication rates to their predicted rates based on disease‐specific logistic regression models and reports whether the hospital is statistically better or worse than predicted. Hospitals are not compared directly to one another. Given the rarity of mortality in most procedures, other outcome measures will be necessary to distinguish among hospitals.26
8. Standardize Reporting Periods
In a world of continuous quality improvement, public reporting should represent a hospital's recent performance, but reporting periods also need to be long enough to provide a stable estimate of infrequent events, especially at low‐volume institutions. In contrast, the lag time between the end of the reporting period and public availability should be kept to a minimum. We found that reporting periods varied from 1 to 3 years, and did not always cover the same years for all conditions, even on the same website. Some data were 3 years old. Patients will have a hard time making decisions on data that is 1 year old, and hospitals will have little incentive to make improvements that will not be acknowledged for years.
9. Avoid Use of Nonvalidated Administrative Data
Administrative data collected for billing purposes, unlike most clinical data, are already in electronic format, and can inexpensively produce quality rankings using validated models.27 In contrast, screening tools, such as the Agency for Healthcare Research and Quality's patient safety indicators (PSIs), were designed to identify potential quality problems, such as postoperative deep vein thrombosis, for internal quality improvement. Cases identified by the PSI software require additional chart review,28, 29 and should not be used as quality indicators. Even so, HealthGrades reports PSIs and some insurers use them in pay‐for‐performance initiatives. Improvements in PSIs, including present‐on‐admission coding, may increase accuracy,30 but these measures need to be validated before they can be adopted for public reporting.
10. Utilization Rates Should Be Evidence‐Based
Although utilization rates for most procedures vary as much as 2‐fold by state or institution, there is little evidence for a best rate. Nevertheless, HealthGrades reports utilization rates for several obstetrical procedures. At present, there are no standards for these, and it is possible that utilization could be too low in some places. Further research is needed; until then, utilization should not purport to measure quality.
Discussion
The growing commitment to making hospital performance data public could transform the quality and safety of care in the US, introducing competition on quality and price and fostering informed consumer choice. To date, the promise of public reporting remains only partially fulfilled. Few hospitals have done more than comply with regulatory mandates and payer incentives, and consumers have failed to respond. To capture the full benefits of public reporting, we have made 10 recommendations to benefit patients and better engage hospitals. We suggest that reporting be patient‐centered, with an emphasis on making the data useful, meaningful, important, interpretable, and relevant. At the same time, hospitals, which are being judged on their performance, should have a level playing field, with measures that are timely, consistent, severity‐adjusted, evidence‐based, and which foster good clinical care. Of the 3 services we examined, Hospital Compare came closest to meeting these recommendations.
Although this blueprint for public reporting is easy to draft, it is challenging to implement. In particular, some of our suggestions, such as the one regarding risk adjustment, may not currently be feasible, because the complexity and cost of collecting clinical data, even in the era of electronic medical records, may be prohibitive. Until such data are readily available, it may be preferable to report nothing at all, rather than report data that are misleading. In the rush to make hospitals accountable, enthusiasm has often outstripped science,31 and several measures have had to be revised for unintended consequences.32
Any initiative to improve public reporting should have the buy‐in of all stakeholders, but particularly hospitals, which stand to benefit in several ways. By receiving regular feedback, they can focus on improving care, becoming better organizations. These improvements may be rewarded through direct compensation (pay‐for‐performance), decreased costs from complications, or increased market share. Hospitals will be more engaged if the data reflect actual quality, are adequately adjusted for severity, and acknowledge the role of chance. Otherwise, they will merely comply, or worse, look for opportunities to game the system. To succeed, public reporting needs to involve hospitals in establishing standards for reporting and validation, as well as auditing procedures to prevent fraud.33 The Hospital Quality Alliance (HQA, Washington, DC), a first step in this direction, at present has few measures. NSQIP (American College of Surgeons, Chicago, IL) is perhaps a better example of hospitals cooperating to set measurement standards to promote best‐practices. Public release of NSQIP data might accelerate progress. Alternatively, the National Quality Forum (NQF, Washington, DC) could expand its role from endorsing quality measures to include standardizing the way these measures are used in public reporting.
Still, if you build it, will they come? To date, public reporting has not been embraced by the public, despite its stated interest in the information. Several explanations could be offered. First, we may be presenting the wrong data. Process measures and mortality rates are important but represent abstract concepts for most patients. Surveys tell us that patients value most the experiences of other patients.14, 21 They want to know whether their pain will be controlled, whether the doctor will listen to them, whether the nurse will come when they call. The recent advent of the HCAHPS survey (AHRQ, Washington, DC) is another positive step. Stratifying the results by diagnosis and adding a few diagnosis‐specific questions would make HCAHPS even more valuable. Second, the data may not be readily available. Although most public reporting is done on the web, older patients who are deciding about hospitals may not have Internet access. Some reports are still proprietary, and cost could present another obstacle. Finally, even if freely‐available and patient‐centered, the results may not be interpretable by physicians, let alone patients.34
If public reporting is to succeed, it will require measures that better reflect patients' concerns. In order to collect the massive amounts of data required and present them in a timely fashion, better electronic record systems will be necessary. But these are no panacea; others have noted that the Department of Veterans Affairs, a leader in electronic records, still invests considerable time and money to review charts for NSQIP.35 Given the value that Americans place on transparency in other facets of their lives, it is clear that public reporting is here to stay. While much progress has been made over the past 5 years, additional research is needed to better measure quality from the patient's perspective, and to determine how this information can be used to help guide decision‐making, and to reward hospitals for offering the highest‐quality care.
Acknowledgements
The authors thank Kenneth Flax for his help with an earlier version of this manuscript.
Acknowledging striking deficiencies in the quality and safety of healthcare, the Institute of Medicine, policy makers, and payors have called for transformation of the US healthcare system.1 Public reporting of hospital performance is one key strategy for accelerating improvement2 and may improve quality in several ways. First, feedback about performance relative to peers may stimulate quality improvement activities by appealing to professionalism. Second, the desire to preserve one's reputation by not appearing on a list of poor performers may be a powerful incentive. Finally, patients and referring providers could use reports to select high‐quality hospitals, thereby shifting care from low‐quality to high‐quality hospitals and stimulating quality improvement efforts to maintain or enhance market share.
Almost 20 years after New York and Pennsylvania began reporting cardiac surgery outcomes,3 the evidence that public reporting improves healthcare quality is equivocal.4 Moreover, stakeholders have embraced public reporting to differing degrees. Public reporting does lead to greater engagement in quality improvement activities,58 and additional financial incentives provide modest incremental benefits.9 Purchasers, too, are starting to pay attention.10 In New York State, payors appear to contract more with high‐quality surgeons and avoid poorly performing outliers.11 Some payors are creating tiered systems, assigning higher patient copayments for hospitals with poor quality metrics. These new systems have not been rigorously studied and should raise concern among hospitals.12
In contrast to hospitals and payors, patients have been slow to embrace public reporting. In a survey of coronary artery bypass graft (CABG) patients in Pennsylvania, only 2% said that public reporting of mortality rates affected their decision making.13 Eight years later, only 11% of patients sought information about hospitals before deciding on elective major surgery,14 although a majority of patients in both studies expressed interest in the information. It is not clear whether recent proliferation of information on the internet will change patient behavior, but to date public reporting appears not to effect market share.5, 15, 16
Barriers to patients' use of public reporting include difficulty accessing the information, lack of trust, information that is not salient, and data that are difficult to interpret.17 In the absence of consensus on what or how to report, a growing number of organizations, including state and federal government, accrediting bodies, private foundations, and for‐profit companies report a variety of measures relating to structure, processes, and outcomes. Although these sites purport to target consumers, they sometimes offer conflicting information18 and are not easily interpreted by lay readers.19
To realize the benefits of public reporting, and minimize the unintended consequences, rating systems must report salient information in a way that is comprehensible to patients and trusted by the doctors who advise them. At the same time, they should be fair to hospitals and offer useful data for quality improvement. We offer 10 recommendations for improving the public reporting of healthcare quality information: 5 describing what to report and 5 detailing how it should be reported (Figure 1). We also examine 3 leading performance reporting programs to see how well they implement these recommendations.

Recommendations to Make Data Salient for Patients
1. Prioritize Elective Procedures
Hospital quality is not uniform across conditions.2 For data to be salient, then, it should be disease‐specific and focus on common elective procedures, for which consumer choice is possible. Table 1 compares 3 popular reporting services. Hospital Compare, produced by the Center for Medicare Services (CMS, US Department of Health and Human Services, Washington, DC), provides process of care measures for 4 conditions, 3 of which are not elective. The fourth, surgical infection prevention, contains 5 measures3 related to perioperative antibiotics and 2 related to thromboembolism prophylaxisfor all surgical cases. Recently, more conditions have been added, but reports are limited to the number of cases and mean Medicare charge. By year 2011, however, Hospital Compare will offer many new measures, including rates of central line infection, ventilator‐associated pneumonia, and surgical site infection. HealthGrades, a private company, offers comparative mortality rates on over 30 diagnoses, of which 15 can be considered elective, at least some of the time. Only the Leapfrog group, an industry consortium, focuses exclusively on elective procedures, offering volume measures on 7 and outcome measures on 2.
| Rule | Hospital Compare | HealthGrades | Leapfrog | |||
|---|---|---|---|---|---|---|
| ||||||
| 1. Prioritize elective procedures | Yes | 22/28 at least partially elective | Yes | 15/31 at least partially elective | Yes | 7/8 elective |
| 2. Include quality of life and outcome data, if possible | Yes | Mortality for AMI and CHF | Yes | Mortality or complications* | Yes | Outcomes for CABG, PCI, and AVR |
| 3. Include standardized patient satisfaction and service measures | Yes | HCAHPS | No | No | ||
| 4. Offer composite measures that are weighted and evidence‐based | No | No | Specialty excellence award, not evidence‐based | No | ||
| 5. Costs comparisons should include patient prices | Yes | Average Medicare payment | Yes | Charges, health plan and Medicare costs available for a fee | No | |
| 6. Adjust outcomes for severity and risk | Yes | Methodology published on website | Yes | Methodology not public | Yes | Various methodologies published or referenced on website |
| 7. Identify differences not due to chance | Yes | Compares mortality to national mean | Yes | Compares mortality or complications to mean | Yes | Compares mortality to national mean |
| 8. Standardize reporting periods | October 2005 to September 2006 | 2004‐2006 | 12‐24 months, ending 12/31/07 or 6/30/08 | |||
| 9. Avoid use of nonvalidated administrative data | Yes | None used | No | Uses PSIs for safety rating | Yes | None used |
| 10. Utilization rates should be evidence‐based | No | Surgical case volume of Medicare patients | No | Includes Caesarian‐section rates | Yes | Some case volume rates are evidence‐based |
2. Include Quality of Life and Outcome Data
Outcomes are more valuable to patients than process measures, but the risk adjustment needed to compare outcomes requires considerable effort. So far, public reporting of risk‐adjusted outcomes has been limited almost exclusively to mortality. Yet a patient contemplating knee replacement surgery would find no meaningful difference in mortalitythere were only 510 deaths nationally in year 200620but might be interested in whether patients return to full mobility after surgery, and all patients should compare rates of nosocomial infections. For some low‐risk procedures, HealthGrades Inc. (Golden, CO) includes a composite measure of major complications, including complication of an orthopedic implant, stroke, cardiac arrest, excessive bleeding, and some types of infection; CMS will soon add rates of infection and readmission.
3. Include Measures of Patient Experience, Such as Satisfaction and Service Measures
Beyond outcomes, patients want to know about the experience of others.21 Satisfaction surveys should be standardized and made disease‐specific, since patients' experiences may differ between the cardiology suite and the delivery unit. Questions could address the attentiveness of the nursing staff, how well privacy was respected, how easy it was to deal with insurance issues, whether patients were promptly informed of test results, and whether the care team answered questions fully. Medicare has begun reporting patient satisfaction using the Hospital Consumer Assessment of Healthcare Providers (HCAHPS) survey on Hospital Compare, but the data are not disease‐specific and audit a very small number of patients from each institution. Other services are unlikely to perform their own surveys, as multiple surveys would prove burdensome. Social networking sites that allow patients to post their own personal reviews of hospitals and doctors offer an additional if less reliable dimension to traditional public reporting. Such sites are already transforming the market for other industries, such as travel.22
4. Offer Composite Measures That Are Weighted and Evidence‐Based
Interpreting multiple measures, some of which are more important than others, and some of which have better evidence than others, is difficult for health care providers and may be impossible for patients. Is it more important to get aspirin on arrival or at discharge? Also, how does a patient weigh a 1% difference in the number of heart attack patients who get aspirin on arrival against a 14% difference in those who are offered smoking cessation? Because patients may be overwhelmed by data,23 public reports should include evidence‐based, weighted measures of overall care for a given condition, with higher weights attached to those process measures most likely to have clinical benefit, and careful attention to visual representations that convey relative differences.19, 23 More sophisticated measures should be developed to guard against overuse. For example, while hospitals should be rewarded for providing vaccination, they should be penalized for vaccinating the same patient twice.
None of the services we examined provides weighted outcomes. Leapfrog (The Leapfrog Group, Washington, DC) offers a composite snapshot containing 9 pie charts, divided into 4 leaps. The 6 pies representing high‐risk procedures are of equal size, even though 2 of these, esophagectomy and pancreatic resection represent very rare surgeries, even at major medical centers. From a visual perspective, however, these are equivalent to having computerized physician order entry and full‐time intensive care unit staffing, which affect thousands more patients. Similarly, in determining pay‐for‐performance measures, CMS created a composite based on the total number of opportunities of all interventions, weighting all measures equally. Because no validated weighting measures exist, future research will be necessary to achieve this goal. Also, none of the evidence‐based measures contained safeguards against overtreatment.
5. Cost Comparisons Should Include Patient Prices
In an era of patient copayments and deductibles, consumers are increasingly aware of costs. For patients with very high deductible plans or no health insurance, hospital fees are a common cause of bankruptcy.24 Several public reporting agencies, including Hospital Compare and HealthGrades have incorporated Medicare costs into their reported measures, but these have little connection to what patients actually pay. Health sites aimed at consumers should publish the average patient copayment.
Recommendations to Ensure That Data Reflects Hospital Quality
6. Adjust Outcomes for Severity and Risk
Not all bypass operations are the same and not all patients are at equal risk. More difficult operations (eg, CABG for a patient with a previous bypass) will have more complications; similarly, patients with serious comorbidities will experience worse outcomes. Since hospitals which specialize in a procedure will attract complicated cases and higher risk patients, it is important to adjust outcomes to account for these differences. Otherwise, hospitals and surgeons may be discouraged from taking difficult cases. Outside of cardiac surgery, most risk adjustment systems use administrative claims data but vary dramatically in the numbers of variables considered and the underlying proprietary models, which are often criticized as being black boxes that yield discordant results.25 Thus, a hospital's mortality may appear below expected by 1 system and above expected by another. Instead, risk adjustment systems should include clinical data abstracted from patient records using standardized data definitions. Although costly to collect, clinical data offer more predictive information than do administrative data. For example, for heart failure patients undergoing CABG, the ejection fraction predicts mortality better than many stable comorbid diagnoses. A single transparent risk‐adjustment system should be recognized as the industry standard. The American College of Surgeons' standardized risk‐adjusted outcome reporting for the National Surgical Quality Improvement Program (NSQIP) is a good example of such an effort.
7. Identify Differences Not Due to Chance
As a result of random variation, during any period, some hospitals will appear better than average and others worse. Statistical tests should be employed to identify hospitals that differ from the mean, and to allow consumers to compare 2 hospitals directly, with appropriate caveats when the hospitals serve very different patient populations. Medicare's mortality rating system for myocardial infarction identifies only 17 hospitals in the nation as better than average and 7 as worse, out of 4,500 institutions. HealthGrades compares hospitals' actual mortality or complication rates to their predicted rates based on disease‐specific logistic regression models and reports whether the hospital is statistically better or worse than predicted. Hospitals are not compared directly to one another. Given the rarity of mortality in most procedures, other outcome measures will be necessary to distinguish among hospitals.26
8. Standardize Reporting Periods
In a world of continuous quality improvement, public reporting should represent a hospital's recent performance, but reporting periods also need to be long enough to provide a stable estimate of infrequent events, especially at low‐volume institutions. In contrast, the lag time between the end of the reporting period and public availability should be kept to a minimum. We found that reporting periods varied from 1 to 3 years, and did not always cover the same years for all conditions, even on the same website. Some data were 3 years old. Patients will have a hard time making decisions on data that is 1 year old, and hospitals will have little incentive to make improvements that will not be acknowledged for years.
9. Avoid Use of Nonvalidated Administrative Data
Administrative data collected for billing purposes, unlike most clinical data, are already in electronic format, and can inexpensively produce quality rankings using validated models.27 In contrast, screening tools, such as the Agency for Healthcare Research and Quality's patient safety indicators (PSIs), were designed to identify potential quality problems, such as postoperative deep vein thrombosis, for internal quality improvement. Cases identified by the PSI software require additional chart review,28, 29 and should not be used as quality indicators. Even so, HealthGrades reports PSIs and some insurers use them in pay‐for‐performance initiatives. Improvements in PSIs, including present‐on‐admission coding, may increase accuracy,30 but these measures need to be validated before they can be adopted for public reporting.
10. Utilization Rates Should Be Evidence‐Based
Although utilization rates for most procedures vary as much as 2‐fold by state or institution, there is little evidence for a best rate. Nevertheless, HealthGrades reports utilization rates for several obstetrical procedures. At present, there are no standards for these, and it is possible that utilization could be too low in some places. Further research is needed; until then, utilization should not purport to measure quality.
Discussion
The growing commitment to making hospital performance data public could transform the quality and safety of care in the US, introducing competition on quality and price and fostering informed consumer choice. To date, the promise of public reporting remains only partially fulfilled. Few hospitals have done more than comply with regulatory mandates and payer incentives, and consumers have failed to respond. To capture the full benefits of public reporting, we have made 10 recommendations to benefit patients and better engage hospitals. We suggest that reporting be patient‐centered, with an emphasis on making the data useful, meaningful, important, interpretable, and relevant. At the same time, hospitals, which are being judged on their performance, should have a level playing field, with measures that are timely, consistent, severity‐adjusted, evidence‐based, and which foster good clinical care. Of the 3 services we examined, Hospital Compare came closest to meeting these recommendations.
Although this blueprint for public reporting is easy to draft, it is challenging to implement. In particular, some of our suggestions, such as the one regarding risk adjustment, may not currently be feasible, because the complexity and cost of collecting clinical data, even in the era of electronic medical records, may be prohibitive. Until such data are readily available, it may be preferable to report nothing at all, rather than report data that are misleading. In the rush to make hospitals accountable, enthusiasm has often outstripped science,31 and several measures have had to be revised for unintended consequences.32
Any initiative to improve public reporting should have the buy‐in of all stakeholders, but particularly hospitals, which stand to benefit in several ways. By receiving regular feedback, they can focus on improving care, becoming better organizations. These improvements may be rewarded through direct compensation (pay‐for‐performance), decreased costs from complications, or increased market share. Hospitals will be more engaged if the data reflect actual quality, are adequately adjusted for severity, and acknowledge the role of chance. Otherwise, they will merely comply, or worse, look for opportunities to game the system. To succeed, public reporting needs to involve hospitals in establishing standards for reporting and validation, as well as auditing procedures to prevent fraud.33 The Hospital Quality Alliance (HQA, Washington, DC), a first step in this direction, at present has few measures. NSQIP (American College of Surgeons, Chicago, IL) is perhaps a better example of hospitals cooperating to set measurement standards to promote best‐practices. Public release of NSQIP data might accelerate progress. Alternatively, the National Quality Forum (NQF, Washington, DC) could expand its role from endorsing quality measures to include standardizing the way these measures are used in public reporting.
Still, if you build it, will they come? To date, public reporting has not been embraced by the public, despite its stated interest in the information. Several explanations could be offered. First, we may be presenting the wrong data. Process measures and mortality rates are important but represent abstract concepts for most patients. Surveys tell us that patients value most the experiences of other patients.14, 21 They want to know whether their pain will be controlled, whether the doctor will listen to them, whether the nurse will come when they call. The recent advent of the HCAHPS survey (AHRQ, Washington, DC) is another positive step. Stratifying the results by diagnosis and adding a few diagnosis‐specific questions would make HCAHPS even more valuable. Second, the data may not be readily available. Although most public reporting is done on the web, older patients who are deciding about hospitals may not have Internet access. Some reports are still proprietary, and cost could present another obstacle. Finally, even if freely‐available and patient‐centered, the results may not be interpretable by physicians, let alone patients.34
If public reporting is to succeed, it will require measures that better reflect patients' concerns. In order to collect the massive amounts of data required and present them in a timely fashion, better electronic record systems will be necessary. But these are no panacea; others have noted that the Department of Veterans Affairs, a leader in electronic records, still invests considerable time and money to review charts for NSQIP.35 Given the value that Americans place on transparency in other facets of their lives, it is clear that public reporting is here to stay. While much progress has been made over the past 5 years, additional research is needed to better measure quality from the patient's perspective, and to determine how this information can be used to help guide decision‐making, and to reward hospitals for offering the highest‐quality care.
Acknowledgements
The authors thank Kenneth Flax for his help with an earlier version of this manuscript.
- Committee on Quality of Health Care in America IoM.Crossing the Quality Chasm: A New Health System for the 21st Century.Washington, DC:National Academy Press;2001.
- ,,,.Care in U.S. hospitals: the Hospital Quality Alliance program.N Engl J Med.2005;353(3):265–274.
- .Achieving and sustaining improved quality: lessons from New York state and cardiac surgery.Health Aff. 20022002;21(4):40–51.
- ,,,,.Systematic review: the evidence that publishing patient care performance data improves quality of care.Ann Intern Med.2008;148(2):111–123.
- ,,.Hospital performance reports: impact on quality, market share, and reputation.Health Aff (Millwood).2005;24(4):1150–1160.
- ,,.Does publicizing hospital performance stimulate quality improvement efforts?Health Aff (Millwood).2003;22(2):84–94.
- ,,,,.Improving the outcomes of coronary artery bypass surgery in New York State.JAMA.1994;271(10):761–766.
- ,,.Declines in hospital mortality associated with a regional initiative to measure hospital performance.Am J Med Qual.1997;12(2):103–112.
- ,,, et al.Public reporting and pay for performance in hospital quality improvement.N Engl J Med.2007;356(5):486–496.
- ,,,,,.Do quality report cards play a role in HMOs' contracting practices? Evidence from New York State.Health Serv Res.2000;35(1 Pt 2):319–332.
- ,,,.Quality of cardiac surgeons and managed care contracting practices.Health Serv Res.2002;37(5):1129–1144.
- ,,,,,.Using performance data to identify preferred hospitals.Health Serv Res.2007;42(6 Pt 1):2109–2119; discussion 2294–2323.
- ,.Use of public performance reports: a survey of patients undergoing cardiac surgery.JAMA.1998;279(20):1638–1642.
- ,,.How do elderly patients decide where to go for major surgery? Telephone interview survey.BMJ.2005;331(7520):821.
- ,,,,,.The effect of publicly reporting hospital performance on market share and risk‐adjusted mortality at high‐mortality hospitals.Med Care.2003;41(6):729–740.
- ,.The predictive accuracy of the New York State coronary artery bypass surgery report‐card system.Health Aff (Millwood).2006;25(3):844–855.
- ,.Publicly disclosed information about the quality of health care: response of the US public.Qual Saf Health Care.2001;10(2):96–103.
- ,,,,.Choosing the best hospital: the limitations of public reporting of hospital quality.Health Aff (Millwood).2008;27(6):1680–1687.
- ,.Will quality report cards help consumers?Health Aff (Millwood).1997;16(3):218–228.
- Agency for Healthcare Research and Quality. HCUPnet, Healthcare Cost and Utilization Project. Available at: http://hcupnet.ahrq.gov. Accessed January 2009.
- ,,.Recovering from cardiac surgery: what patients want you to know.Am J Crit Care.2002;11(4):333–343.
- Trip Advisor. Available at: http://www.tripadvisor.com. Accessed January 2009.
- ,,,,.Less is more in presenting quality information to consumers.Med Care Res Rev.2007;64(2):169–190.
- ,,,.MarketWatch: illness and injury as contributors to bankruptcy.Health Aff (Millwood)2005;(Suppl Web Exclusives):W5‐63–W5‐73.
- .The Lake Wobegon effect: when all the patients are sicker.Am J Med Qual.2006;21(6):365–366.
- ,,.Surgical mortality as an indicator of hospital quality: the problem with small sample size.JAMA.2004;292(7):847–851.
- ,,, et al.An administrative claims model suitable for profiling hospital performance based on 30‐day mortality rates among patients with heart failure.Circulation.2006;113(13):1693–1701.
- ,,,.Can administrative data be used to compare postoperative complication rates across hospitals?Med Care.2002;40(10):856–867.
- ,,,,.Impact of diagnosis‐timing indicators on measures of safety, comorbidity, and case mix groupings from administrative data sources.Med Care.2007;45(8):781–788.
- ,,,,.Do the AHRQ patient safety indicators flag conditions that are present at the time of hospital admission?Med Care.2008;46(5):516–522.
- ,,.The tension between needing to improve care and knowing how to do it.N Engl J Med.2007;357(6):608–613.
- ,,,.Public reporting of antibiotic timing in patients with pneumonia: lessons from a flawed performance measure.Ann Intern Med.2008;149(1):29–32.
- ,,.The GAAP in quality measurement and reporting.JAMA.2007;298(15):1800–1802.
- ,,,.Consumer competencies and the use of comparative quality information: it isn't just about literacy.Med Care Res Rev.2007;64(4):379–394.
- .Performance measurement in search of a path.N Engl J Med.2007;356(9):951–953.
- Committee on Quality of Health Care in America IoM.Crossing the Quality Chasm: A New Health System for the 21st Century.Washington, DC:National Academy Press;2001.
- ,,,.Care in U.S. hospitals: the Hospital Quality Alliance program.N Engl J Med.2005;353(3):265–274.
- .Achieving and sustaining improved quality: lessons from New York state and cardiac surgery.Health Aff. 20022002;21(4):40–51.
- ,,,,.Systematic review: the evidence that publishing patient care performance data improves quality of care.Ann Intern Med.2008;148(2):111–123.
- ,,.Hospital performance reports: impact on quality, market share, and reputation.Health Aff (Millwood).2005;24(4):1150–1160.
- ,,.Does publicizing hospital performance stimulate quality improvement efforts?Health Aff (Millwood).2003;22(2):84–94.
- ,,,,.Improving the outcomes of coronary artery bypass surgery in New York State.JAMA.1994;271(10):761–766.
- ,,.Declines in hospital mortality associated with a regional initiative to measure hospital performance.Am J Med Qual.1997;12(2):103–112.
- ,,, et al.Public reporting and pay for performance in hospital quality improvement.N Engl J Med.2007;356(5):486–496.
- ,,,,,.Do quality report cards play a role in HMOs' contracting practices? Evidence from New York State.Health Serv Res.2000;35(1 Pt 2):319–332.
- ,,,.Quality of cardiac surgeons and managed care contracting practices.Health Serv Res.2002;37(5):1129–1144.
- ,,,,,.Using performance data to identify preferred hospitals.Health Serv Res.2007;42(6 Pt 1):2109–2119; discussion 2294–2323.
- ,.Use of public performance reports: a survey of patients undergoing cardiac surgery.JAMA.1998;279(20):1638–1642.
- ,,.How do elderly patients decide where to go for major surgery? Telephone interview survey.BMJ.2005;331(7520):821.
- ,,,,,.The effect of publicly reporting hospital performance on market share and risk‐adjusted mortality at high‐mortality hospitals.Med Care.2003;41(6):729–740.
- ,.The predictive accuracy of the New York State coronary artery bypass surgery report‐card system.Health Aff (Millwood).2006;25(3):844–855.
- ,.Publicly disclosed information about the quality of health care: response of the US public.Qual Saf Health Care.2001;10(2):96–103.
- ,,,,.Choosing the best hospital: the limitations of public reporting of hospital quality.Health Aff (Millwood).2008;27(6):1680–1687.
- ,.Will quality report cards help consumers?Health Aff (Millwood).1997;16(3):218–228.
- Agency for Healthcare Research and Quality. HCUPnet, Healthcare Cost and Utilization Project. Available at: http://hcupnet.ahrq.gov. Accessed January 2009.
- ,,.Recovering from cardiac surgery: what patients want you to know.Am J Crit Care.2002;11(4):333–343.
- Trip Advisor. Available at: http://www.tripadvisor.com. Accessed January 2009.
- ,,,,.Less is more in presenting quality information to consumers.Med Care Res Rev.2007;64(2):169–190.
- ,,,.MarketWatch: illness and injury as contributors to bankruptcy.Health Aff (Millwood)2005;(Suppl Web Exclusives):W5‐63–W5‐73.
- .The Lake Wobegon effect: when all the patients are sicker.Am J Med Qual.2006;21(6):365–366.
- ,,.Surgical mortality as an indicator of hospital quality: the problem with small sample size.JAMA.2004;292(7):847–851.
- ,,, et al.An administrative claims model suitable for profiling hospital performance based on 30‐day mortality rates among patients with heart failure.Circulation.2006;113(13):1693–1701.
- ,,,.Can administrative data be used to compare postoperative complication rates across hospitals?Med Care.2002;40(10):856–867.
- ,,,,.Impact of diagnosis‐timing indicators on measures of safety, comorbidity, and case mix groupings from administrative data sources.Med Care.2007;45(8):781–788.
- ,,,,.Do the AHRQ patient safety indicators flag conditions that are present at the time of hospital admission?Med Care.2008;46(5):516–522.
- ,,.The tension between needing to improve care and knowing how to do it.N Engl J Med.2007;357(6):608–613.
- ,,,.Public reporting of antibiotic timing in patients with pneumonia: lessons from a flawed performance measure.Ann Intern Med.2008;149(1):29–32.
- ,,.The GAAP in quality measurement and reporting.JAMA.2007;298(15):1800–1802.
- ,,,.Consumer competencies and the use of comparative quality information: it isn't just about literacy.Med Care Res Rev.2007;64(4):379–394.
- .Performance measurement in search of a path.N Engl J Med.2007;356(9):951–953.
Copyright © 2009 Society of Hospital Medicine
Acute Vertebral Fracture
An 89‐year‐old female presents to the Emergency Department with lower back pain for the past 5 days. The patient has a past medical history of polymyalgia rheumatica and hypothyroidism. Her medications include prednisone 10 mg daily and levothyroxine 50 g daily. Aside from tenderness over the third lumbar vertebra, her physical exam is unremarkable. An x‐ray shows a fracture of the third lumbar vertebra. Basic laboratory studies, including calcium and creatinine are normal.
The Clinical Problem and Impact
Vertebral fractures (often termed vertebral compression fractures) affect approximately 25% of all postmenopausal women.1, 2 Only one‐third of vertebral fractures are brought to medical attention.3, 4 In the remaining two‐thirds, patients are either asymptomatic or do not seek medical attention. The lifetime risk of a clinical vertebral fracture is approximately 16% and 5% in white women and men, respectively.1 The risk of vertebral fracture increases with age, lower bone mineral density (BMD), and prior vertebral fracture.1, 5 Women with a preexisting vertebral fracture have a 5‐fold increased risk for a new vertebral fracture relative to those without a history of vertebral fracture.5, 6 Approximately 20% of women who sustain a vertebral fracture will have a new vertebral fracture in the subsequent year.5, 6 Vertebral fractures are frequently overlooked on chest x‐rays and hence there is a need for increased awareness and improved recognition of radiographically‐demonstrated fractures.7 Hospitalists must appreciate that a vertebral fracture is often the first clue to underlying osteoporosis. At least 90% of vertebral and hip fractures are attributable to osteoporosis.5
Typically, the pain related to an acute vertebral fracture improves over 4 to 6 weeks.8, 9 However, pain can persist, resulting in functional impairment, and a decline in the quality of life.1013 Vertebral fractures may lead to kyphosis and reduced lung function. This, in turn, may increase the risk for pneumonia, the most common cause of death in patients with osteoporosis.4 Both clinical and subclinical vertebral fractures are, in fact, independently associated with increased mortality,4, 14, 15 particularly in the period immediately following the event.16 The economic impact of osteoporosis and its related fractures is substantial.1719 In 1995, the annual direct medical cost for the inpatient care of vertebral fractures was estimated to be $575 million.19
Evidence‐Based Approach to the Hospitalized Patient
Evaluation
Vertebral fractures most commonly occur between T7 and L4. Acute vertebral fracture pain is typically sudden in onset and located in the mid to lower back. The pain may occur while performing an ordinary task such as lifting an object or bending over, although in many cases there is no preceding trauma. Physical activity exacerbates the pain and patients' movements may be limited due to pain. Spinal tenderness is usually present. A history of weight loss, prior malignancy, or fever should serve as red flags to the hospitalist and prompt evaluation for underlying malignancy or infection.
For a suspected fracture, frontal and lateral radiographs of the thoracolumbar spine are the initial imaging of choice. Magnetic resonance imaging (MRI) is useful when infection, malignancy, or spinal cord compression is suspected. The presence of neurological deficits should always prompt imaging with MRI or computed tomography (CT).
Patients with vertebral fractures should be evaluated with bone density testing, as osteoporosis is usually the underlying etiology. Because a number of medical conditions commonly contribute to bone loss (Table 1), laboratory testing is indicated for most patients. Although debate exists as to the optimal testing strategy,20 the National Osteoporosis Foundation guidelines recommend the evaluation of blood count, chemistry, and thyroid‐stimulating hormone (TSH) in patients with osteoporosis.21 Because vitamin D deficiency is common in patients who sustain osteoporotic fractures, 25‐hydroxyvitamin D levels should be checked in most patients.2225 Depending on the clinical scenario, additional testing may include the following: serum testosterone, serum intact parathyroid hormone (PTH), 24‐hour calcium excretion, serum protein electrophoresis, urine protein electrophoresis, erythrocyte sedimentation rate, and celiac sprue antibodies.26
| Endocrine disease or metabolic causes | Hypogonadism |
| Cushing's syndrome | |
| Hyperthyroidism | |
| Anorexia nervosa | |
| Hyperparathyroidism | |
| Nutritional conditions | Vitamin D deficiency |
| Calcium deficiency | |
| Malabsorption | |
| Drugs | Glucocorticoids |
| Antiepileptic drugs | |
| Excessive thyroid medication | |
| Long‐term heparin or low molecular weight heparin therapy (eg, >1 month) | |
| Other | Multiple myeloma |
| Rheumatoid arthritis | |
| Organ transplantation |
Management
Short Term
Short‐term management goals include the relief of pain and recovery of mobility. Nonsteroidal antiinflammatory drugs (NSAIDs) and low‐dose opioid medications should be used first in an effort to relieve pain. Beyond its potential effect on bone density, calcitonin (Miacalcin, Fortical) has long been used in acute vertebral fractures for analgesia. A systematic review of 5 randomized controlled trials concluded that calcitonin significantly reduced the pain from acute vertebral fractures.27 Calcitonin improved pain as early as 1 week into treatment and the benefit was persistent at 4 weeks. The analgesic mechanism of action for calcitonin is not entirely clear. Proposed mechanisms include increased ‐endorphin release, an antiinflammatory effect, and a direct effect on specific receptors in the central nervous system.27, 28
Pamidronate (Aredia) also has shown efficacy at reducing acute fracture pain. In a double‐blinded trial, Armigeat et al.29 evaluated pamidronate 30 mg daily for 3 days as compared to placebo in 32 patients. Pain scores were improved at 7 and 30 days with pamidronate. The mechanism of analgesia is unclear. Bisphosphonates are known to inhibit osteoclast activity, but may also work by blocking the effect of inflammatory cytokines.29
Early pain relief is critical in order to encourage physical activity. Bed rest should be avoided, as immobility may increase the risk for pressure ulcers, venous thromboembolism, and pneumonia.3033 Although bracing is frequently used in acute vertebral fracture, the modality has not been formally studied. Although also not well studied in the acute setting, physical therapy has been shown to reduce pain and improve functioning for patients with chronic pain from vertebral fracture,34 and is generally recommended.35
Percutaneous vertebral augmentation procedures include vertebroplasty and kyphoplasty. In vertebroplasty, polymethylmethacrylate cement is injected through a needle under fluoroscopic guidance into the collapsed vertebral body. With kyphoplasty, balloon tamps are used to elevate vertebral endplates prior to injection of cement (Figure 1).36 The proposed mechanism of action for both procedures is stabilization of the fracture by the hardened polymethylmethacrylate cement. These procedures are commonly performed by interventional radiologists without the need for general anesthesia; however, depending on the institution, they may be done by orthopedic surgeons, neurosurgeons, or anesthesiologists. The procedure can be performed as an outpatient, if indicated. Although the volume of these procedures has grown dramatically in recent years,37, 38 the quality of evidence supporting their use is relatively weak.3941 Only 1 randomized controlled trial has been published evaluating the potential benefit of vertebroplasty over conservative management.42 Voormolen et al.42 evaluated patients with vertebral fractures and pain refractive to 6 weeks of optimal medical therapy. Patients were treated with vertebroplasty or continuation of medical therapy. Vertebroplasty significantly improved pain initially, but not after 2 weeks. Like the study by Voormolen et al.,42 most studies evaluating percutaneous vertebral augmentation procedures have been conducted on patients with long‐term pain refractory to medical management. One notable exception is a nonrandomized trial published by Diamond et al.6 In that study, 55 patients were treated with vertebroplasty while 24 were treated conservatively. Pain at 24 hours was significantly improved in patients treated with vertebroplasty. At 6 weeks, however, there was no difference among the 2 groups.
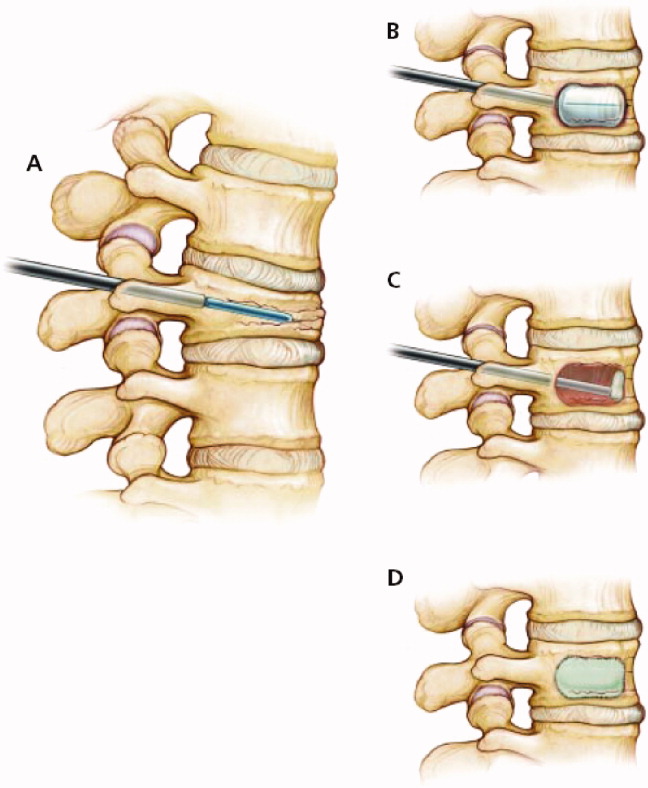
The risk of short‐term complications from vertebral augmentation procedures is difficult to assess in light of the small sample sizes and methodological limitations of existing studies. Cement leakage occurs in 40% to 41% of patients treated with vertebroplasty, as compared with 8% to 9% with kyphoplasty.39, 41 Pulmonary emboli occur in 0.6% and 0.01% of patients treated with vertebroplasty and kyphoplasty, respectively, while neurologic complications occur in 0.6% and 0.03% of patients.41 Concern exists about whether percutaneous vertebral augmentation procedures might increase the risk for subsequent fractures,43, 44 as the incidence of new fractures appears to be elevated in the period immediately following the procedure and approximately two‐thirds of new fractures occur in vertebrae adjacent to the augmented vertebra.39, 41, 44 However, the 20% incidence of new vertebral fractures in the year following vertebral augmentation is similar to the fracture rate seen in patients not treated with osteoporosis therapy.44
Assessment of the impact of vertebral augmentation procedures on the cost of care is limited by the lack of high‐quality clinical studies.45, 46 Randomized controlled trials evaluating the benefit and risk of these procedures compared to conservative management are underway.4749 Pending further evidence, these procedures are best reserved for patients who fail to benefit from other measures to control pain and improve mobility.
Long Term
A comprehensive discussion of the long‐term management of osteoporosis is beyond the scope of this work. However, the inpatient setting presents an opportune time to initiate long‐term medical therapy. Studies show that the majority of patients who sustain osteoporotic fractures do not receive pharmacologic treatment for osteoporosis.5053 Hospitalists have the opportunity to start medications that can reduce the risk for subsequent fracture by nearly 50%.5458 A total calcium intake of 1200 to 1500 mg per day and vitamin D of 400 to 800 IU per day are recommended for all postmenopausal women. Patients who smoke should receive smoking cessation counseling and be considered for pharmacologic treatment for tobacco dependence. All patients should be assessed for fall risk, including a review of medications and assessment of alcohol intake.
Before considering pharmacologic treatment for osteoporosis, secondary causes of low bone mass must be excluded. Bisphosphonates are generally considered first‐line pharmacologic therapy for osteoporosis. Alendronate (Fosamax), risedronate (Actonel), and ibandronate (Boniva) have been shown in randomized trials to increase bone density and reduce the risk of osteoporotic fractures.55, 56, 59 Daily, weekly, and monthly preparations of bisphosphonates now exist. Pill‐induced esophagitis is a potential adverse effect of bisphosphonate therapy, but is extremely rare if proper precautions are taken. Patients should take oral bisphosphonates on an empty stomach, with a full glass of water, sitting upright, and have nothing to eat or drink for at least one half hour. If compliance with oral bisphosphonates is not possible, or esophageal abnormalities preclude oral bisphosphonate use, one may consider the use of intravenous ibandronate or zoledronic acid (Reclast). A 3‐year randomized controlled trial of yearly zoledronic acid improved bone density and reduced the incidence of osteoporotic fractures.60 Bisphosphonates are generally not recommended when creatinine clearance is less than 30 mL/minute. Other pharmacologic options for the treatment of osteoporosis include selective estrogen receptor modulators and anabolic agents. The reader is referred to an excellent review by Rosen61 for additional discussion of these therapies. The American College of Rheumatology clinical guidelines for the management of glucocorticoid induced osteoporosis are also worthy of review.62
Hospitalists are naturally suited to improve the quality of care for patients hospitalized with vertebral fractures. Most patients who currently sustain osteoporotic fractures do not receive appropriate evaluation and treatment. One study used an interdisciplinary team to identify, assess, and begin treatment for appropriate patients hospitalized with osteoporotic fractures.63 The intervention resulted in significantly more patients taking osteoporosis treatment medications 6 months after the incident fracture.
Conclusions
Acute vertebral fracture is a common clinical problem associated with significant morbidity and increased risk of mortality. Treatment of vertebral fracture should include analgesics and physical therapy. Percutaneous augmentation procedures may be considered in patients who fail optimal medical therapy. Because most vertebral fractures are due to osteoporosis and the healthcare system currently fails to appropriately assess and treat most patients who have sustained osteoporotic fractures, hospitalists are in an optimal position to initiate long‐term preventative treatment for these patients.
- .Epidemiology of spinal osteoporosis.Spine.1997;22(24 suppl):2S–11S.
- ,,,,,.Prevalence and incidence of vertebral deformities.Osteoporos Int.1993;3(3):113–119.
- ,,.The epidemiology of vertebral fractures. European Vertebral Osteoporosis Study Group.Bone.1993;14(suppl 1):S89–S97.
- ,,,,,.Vertebral fractures and mortality in older women: a prospective study. Study of Osteoporotic Fractures Research Group.Arch Intern Med.1999;159(11):1215–1220.
- ,,,.Pre‐existing fractures and bone mass predict vertebral fracture incidence in women.Ann Intern Med.1991;114(11):919–923.
- ,,, et al.Risk of new vertebral fracture in the year following a fracture.JAMA.2001;285(3):320–323.
- ,,,,,.Recognition of vertebral fracture in a clinical setting.Osteoporos Int.2000;11(7):577–582.
- ,,.Management of acute osteoporotic vertebral fractures: a nonrandomized trial comparing percutaneous vertebroplasty with conservative therapy.Am J Med.2003;114(4):257–265.
- .The clinical consequences of vertebral compression fracture.Bone.1992;13(suppl 2):S27–S31.
- ,,,.A case‐control study of quality of life and functional impairment in women with long‐standing vertebral osteoporotic fracture.Osteoporos Int.1999;9(6):508–515.
- ,,, et al.The burden of prevalent fractures on health‐related quality of life in postmenopausal women with osteoporosis: the IMOF study.J Rheumatol.2007;34(7):1551–1560.
- ,,,,.The relationship of health‐related quality of life to prevalent and incident vertebral fractures in postmenopausal women with osteoporosis: results from the Multiple Outcomes of Raloxifene Evaluation Study.Arthritis Rheum2001;44(11):2611–2619.
- ,,, et al.The association of radiographically detected vertebral fractures with back pain and function: a prospective study.Ann Int Med.1998;128:793–800.
- ,,,,.Mortality after all major types of osteoporotic fracture in men and women: an observational study.Lancet. 131999;353(9156):878–882.
- ,,,,.Population‐based study of survival after osteoporotic fractures.Am J Epidemiol.1993;137(9):1001–1005.
- ,,,,.Excess mortality after hospitalisation for vertebral fracture.Osteoporos Int.2004;15(2):108–112.
- ,.The cost of treating osteoporotic fractures in the United Kingdom female population.Osteoporos Int.1998;8(6):611–617.
- ,,, et al.Direct medical costs attributable to osteoporotic fractures.Osteoporos Int.2002;13(4):323–330.
- ,,,.Medical expenditures for the treatment of osteoporotic fractures in the United States in 1995: report from the National Osteoporosis Foundation.J Bone Miner Res.1997;12(1):24–35.
- .Laboratory workup for osteoporosis. Which tests are most cost‐effective?Postgrad Med.2003;114(3):35–38,41–34.
- National Osteoporosis Foundation. Clinician's Guide to Prevention and Treatment of Osteoporosis. Available at: http://www.nof.org/professionals/Clinicians_Guide.htm. Accessed February2009.
- ,,, et al.Prevalence of vitamin D inadequacy among postmenopausal North American women receiving osteoporosis therapy.J Clin Endocr Metab.2005;90:3215–3224.
- ,,,,.Prevalence of vitamin D inadequacy in a minimal trauma fracture population.Curr Med Res Opin.2005;21:1069–1074.
- ,,,,,.Occult vitamin D deficiency in postmenopausal US women with acute hip fracture.JAMA.1999;281:1505–1511.
- ,,,,.Secondary contributors for bone loss in osteoporotic hip fractures.Osteoporos Int.2008;19(7):991–999.
- .Evaluation of the patient with osteoporosis or at risk for osteoporosis. In: Marcus R, Feldman D, Kelsey J, eds.Osteoporosis. Vol.2.San Diego:Academic Press;2001:403–408.
- ,,,,.Calcitonin for treating acute pain of osteoporotic vertebral compression fractures: a systematic review of randomized, controlled trials.Osteoporos Int.2005;16(10):1281–1290.
- .Possible mechanisms of the analgesic action of calcitonin.Bone.2002;30(5 suppl):80S–83S.
- ,,,,.Intravenous pamidronate for pain relief in recent osteoporotic vertebral compression fracture: a randomized double‐blind controlled study.Osteoporos Int.2006;17(11):1659–1665.
- ,,,,.Pressure ulcer risk factors among hospitalized patients with activity limitation.JAMA.1995;273(11):865–870.
- ,.Risk factors for venous thromboembolism.Circulation. 172003;107(23 suppl 1):I9–I16.
- ,,.Infectious diseases and mortality among US nursing home residents.Am J Public Health.1993;83(12):1739–1742.
- ,,,,.Risk factors for pneumonia and other lower respiratory tract infections in elderly residents of long‐term care facilities.Arch Intern Med. 271999;159(17):2058–2064.
- ,,,.Positive effects of physiotherapy on chronic pain and performance in osteoporosis.Osteoporos Int.1998;8(3):215–221.
- ,,, et al.Health professional's guide to rehabilitation of the patient with osteoporosis.Osteoporos Int.2003;14(suppl 2):S1–S22.
- ,,,.Vertebral compression fractures: manage aggressively to prevent sequelae.Cleve Clin J Med.2003;70(2):147–156. Reprinted with permission. Copyright (c) 2003 Cleveland Clinic Foundation. All rights reserved.
- ,,,.Vertebroplasty in the United States: guidance method and provider distribution, 2001–2003.Radiology.2007;243(1):166–170.
- ,,,,.Thoracic and lumbar vertebroplasties performed in US Medicare enrollees, 2001–2005.JAMA.2007;298(15):1760–1762.
- ,,.Balloon kyphoplasty and vertebroplasty for vertebral compression fractures: a comparative systematic review of efficacy and safety.Spine.2006;31(23):2747–2755.
- ,,,,.Efficacy and safety of balloon kyphoplasty in the treatment of vertebral compression fractures: a systematic review.Eur Spine J.2006;15(7):1050–1067.
- ,,,.Vertebroplasty and kyphoplasty: a systematic review of 69 clinical studies.Spine.2006;31(17):1983–2001.
- ,,, et al.Percutaneous vertebroplasty compared with optimal pain medication treatment: short‐term clinical outcome of patients with subacute or chronic painful osteoporotic vertebral compression fractures. The VERTOS study.AJNR Am J Neuroradiol.2007;28(3):555–560.
- ,.Recurrent fracture after vertebral kyphoplasty.Spine J.2006;6(5):488–493.
- ,.Does vertebroplasty cause incident vertebral fractures? A review of available data.AJNR Am J Neuroradiol.2006;27(7):1397–1403.
- Centers for Medicare and Medicaid Services. Agency for Healthcare Research and Quality (AHRQ). Technology Assessment. Percutaneous Kyphoplasty for Vertebral Fractures Caused by Osteoporosis and Malignancy, 2005. Available at: http://www.cms.hhs.gov/mcd/viewtechassess.asp?from2=viewtechassess.asp13(5):550–555.
- .Randomized vertebroplasty trials: current status and challenges.Acad Radiol.2006;13(5):546–549.
- ,,, et al.VERTOS II: percutaneous vertebroplasty versus conservative therapy in patients with painful osteoporotic vertebral compression fractures; rationale, objectives and design of a multicenter randomized controlled trial.Trials.2007;8(1):33.
- ,.Vertebroplasty: a promising but as yet unproven intervention for painful osteoporotic spinal fractures.Med J Aust.2006;185(7):351–352.
- ,,, et al.Low frequency of treatment of osteoporosis among postmenopausal women following a fracture.Arch Intern Med.2003;163(17):2052–2057.
- ,,,,.Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture.Am J Med.2000;109(4):326–328.
- ,,.Missing a therapeutic window of opportunity: an audit of patients attending a tertiary teaching hospital with potentially osteoporotic hip and wrist fractures.J Rheumatol.2001;28(11):2504–2508.
- ,,,,.Underuse of osteoporosis medications in elderly patients with fractures.Am J Med.2003;115(5):398–400.
- ,,, et al.Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group.Lancet.1996;348(9041):1535–1541.
- ,,,,,.Meta‐analyses of therapies for postmenopausal osteoporosis. IX: Summary of meta‐analyses of therapies for postmenopausal osteoporosis.Endocr Rev.2002;23(4):570–578.
- ,,, et al.Summary of meta‐analyses of therapies for postmenopausal osteoporosis and the relationship between bone density and fractures.Endocrinol Metab Clin North Am.2002;31(3):659–679, xii.
- ,,, et al.Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group.JAMA.1999;282(14):1344–1352.
- ,,, et al.Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group.N Engl J Med.2001;344(5):333–340.
- ,,, et al.Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis.J Bone Miner Res.2004;19(8):1241–1249.
- ,,, et al.Once‐yearly zoledronic acid for treatment of postmenopausal osteoporosis.N Engl J Med. 32007;356(18):1809–1822.
- .Clinical practice. Postmenopausal osteoporosis.N Engl J Med.2005;353(6):595–603.
- American College of Rheumatology Ad Hoc Committee on Glucocorticoid‐Induced Osteoporosis.Recommendations for the prevention and treatment of glucocorticoid‐induced osteoporosis: 2001 update. [Review].Arthritis Rheum.2001;44(7):1496–1503.
- ,,, et al.An osteoporosis and fracture intervention program increases the diagnosis and treatment for osteoporosis for patients with minimal trauma fractures.Jt Comm J Qual Patient Saf.2005;31(5):267–274.
An 89‐year‐old female presents to the Emergency Department with lower back pain for the past 5 days. The patient has a past medical history of polymyalgia rheumatica and hypothyroidism. Her medications include prednisone 10 mg daily and levothyroxine 50 g daily. Aside from tenderness over the third lumbar vertebra, her physical exam is unremarkable. An x‐ray shows a fracture of the third lumbar vertebra. Basic laboratory studies, including calcium and creatinine are normal.
The Clinical Problem and Impact
Vertebral fractures (often termed vertebral compression fractures) affect approximately 25% of all postmenopausal women.1, 2 Only one‐third of vertebral fractures are brought to medical attention.3, 4 In the remaining two‐thirds, patients are either asymptomatic or do not seek medical attention. The lifetime risk of a clinical vertebral fracture is approximately 16% and 5% in white women and men, respectively.1 The risk of vertebral fracture increases with age, lower bone mineral density (BMD), and prior vertebral fracture.1, 5 Women with a preexisting vertebral fracture have a 5‐fold increased risk for a new vertebral fracture relative to those without a history of vertebral fracture.5, 6 Approximately 20% of women who sustain a vertebral fracture will have a new vertebral fracture in the subsequent year.5, 6 Vertebral fractures are frequently overlooked on chest x‐rays and hence there is a need for increased awareness and improved recognition of radiographically‐demonstrated fractures.7 Hospitalists must appreciate that a vertebral fracture is often the first clue to underlying osteoporosis. At least 90% of vertebral and hip fractures are attributable to osteoporosis.5
Typically, the pain related to an acute vertebral fracture improves over 4 to 6 weeks.8, 9 However, pain can persist, resulting in functional impairment, and a decline in the quality of life.1013 Vertebral fractures may lead to kyphosis and reduced lung function. This, in turn, may increase the risk for pneumonia, the most common cause of death in patients with osteoporosis.4 Both clinical and subclinical vertebral fractures are, in fact, independently associated with increased mortality,4, 14, 15 particularly in the period immediately following the event.16 The economic impact of osteoporosis and its related fractures is substantial.1719 In 1995, the annual direct medical cost for the inpatient care of vertebral fractures was estimated to be $575 million.19
Evidence‐Based Approach to the Hospitalized Patient
Evaluation
Vertebral fractures most commonly occur between T7 and L4. Acute vertebral fracture pain is typically sudden in onset and located in the mid to lower back. The pain may occur while performing an ordinary task such as lifting an object or bending over, although in many cases there is no preceding trauma. Physical activity exacerbates the pain and patients' movements may be limited due to pain. Spinal tenderness is usually present. A history of weight loss, prior malignancy, or fever should serve as red flags to the hospitalist and prompt evaluation for underlying malignancy or infection.
For a suspected fracture, frontal and lateral radiographs of the thoracolumbar spine are the initial imaging of choice. Magnetic resonance imaging (MRI) is useful when infection, malignancy, or spinal cord compression is suspected. The presence of neurological deficits should always prompt imaging with MRI or computed tomography (CT).
Patients with vertebral fractures should be evaluated with bone density testing, as osteoporosis is usually the underlying etiology. Because a number of medical conditions commonly contribute to bone loss (Table 1), laboratory testing is indicated for most patients. Although debate exists as to the optimal testing strategy,20 the National Osteoporosis Foundation guidelines recommend the evaluation of blood count, chemistry, and thyroid‐stimulating hormone (TSH) in patients with osteoporosis.21 Because vitamin D deficiency is common in patients who sustain osteoporotic fractures, 25‐hydroxyvitamin D levels should be checked in most patients.2225 Depending on the clinical scenario, additional testing may include the following: serum testosterone, serum intact parathyroid hormone (PTH), 24‐hour calcium excretion, serum protein electrophoresis, urine protein electrophoresis, erythrocyte sedimentation rate, and celiac sprue antibodies.26
| Endocrine disease or metabolic causes | Hypogonadism |
| Cushing's syndrome | |
| Hyperthyroidism | |
| Anorexia nervosa | |
| Hyperparathyroidism | |
| Nutritional conditions | Vitamin D deficiency |
| Calcium deficiency | |
| Malabsorption | |
| Drugs | Glucocorticoids |
| Antiepileptic drugs | |
| Excessive thyroid medication | |
| Long‐term heparin or low molecular weight heparin therapy (eg, >1 month) | |
| Other | Multiple myeloma |
| Rheumatoid arthritis | |
| Organ transplantation |
Management
Short Term
Short‐term management goals include the relief of pain and recovery of mobility. Nonsteroidal antiinflammatory drugs (NSAIDs) and low‐dose opioid medications should be used first in an effort to relieve pain. Beyond its potential effect on bone density, calcitonin (Miacalcin, Fortical) has long been used in acute vertebral fractures for analgesia. A systematic review of 5 randomized controlled trials concluded that calcitonin significantly reduced the pain from acute vertebral fractures.27 Calcitonin improved pain as early as 1 week into treatment and the benefit was persistent at 4 weeks. The analgesic mechanism of action for calcitonin is not entirely clear. Proposed mechanisms include increased ‐endorphin release, an antiinflammatory effect, and a direct effect on specific receptors in the central nervous system.27, 28
Pamidronate (Aredia) also has shown efficacy at reducing acute fracture pain. In a double‐blinded trial, Armigeat et al.29 evaluated pamidronate 30 mg daily for 3 days as compared to placebo in 32 patients. Pain scores were improved at 7 and 30 days with pamidronate. The mechanism of analgesia is unclear. Bisphosphonates are known to inhibit osteoclast activity, but may also work by blocking the effect of inflammatory cytokines.29
Early pain relief is critical in order to encourage physical activity. Bed rest should be avoided, as immobility may increase the risk for pressure ulcers, venous thromboembolism, and pneumonia.3033 Although bracing is frequently used in acute vertebral fracture, the modality has not been formally studied. Although also not well studied in the acute setting, physical therapy has been shown to reduce pain and improve functioning for patients with chronic pain from vertebral fracture,34 and is generally recommended.35
Percutaneous vertebral augmentation procedures include vertebroplasty and kyphoplasty. In vertebroplasty, polymethylmethacrylate cement is injected through a needle under fluoroscopic guidance into the collapsed vertebral body. With kyphoplasty, balloon tamps are used to elevate vertebral endplates prior to injection of cement (Figure 1).36 The proposed mechanism of action for both procedures is stabilization of the fracture by the hardened polymethylmethacrylate cement. These procedures are commonly performed by interventional radiologists without the need for general anesthesia; however, depending on the institution, they may be done by orthopedic surgeons, neurosurgeons, or anesthesiologists. The procedure can be performed as an outpatient, if indicated. Although the volume of these procedures has grown dramatically in recent years,37, 38 the quality of evidence supporting their use is relatively weak.3941 Only 1 randomized controlled trial has been published evaluating the potential benefit of vertebroplasty over conservative management.42 Voormolen et al.42 evaluated patients with vertebral fractures and pain refractive to 6 weeks of optimal medical therapy. Patients were treated with vertebroplasty or continuation of medical therapy. Vertebroplasty significantly improved pain initially, but not after 2 weeks. Like the study by Voormolen et al.,42 most studies evaluating percutaneous vertebral augmentation procedures have been conducted on patients with long‐term pain refractory to medical management. One notable exception is a nonrandomized trial published by Diamond et al.6 In that study, 55 patients were treated with vertebroplasty while 24 were treated conservatively. Pain at 24 hours was significantly improved in patients treated with vertebroplasty. At 6 weeks, however, there was no difference among the 2 groups.

The risk of short‐term complications from vertebral augmentation procedures is difficult to assess in light of the small sample sizes and methodological limitations of existing studies. Cement leakage occurs in 40% to 41% of patients treated with vertebroplasty, as compared with 8% to 9% with kyphoplasty.39, 41 Pulmonary emboli occur in 0.6% and 0.01% of patients treated with vertebroplasty and kyphoplasty, respectively, while neurologic complications occur in 0.6% and 0.03% of patients.41 Concern exists about whether percutaneous vertebral augmentation procedures might increase the risk for subsequent fractures,43, 44 as the incidence of new fractures appears to be elevated in the period immediately following the procedure and approximately two‐thirds of new fractures occur in vertebrae adjacent to the augmented vertebra.39, 41, 44 However, the 20% incidence of new vertebral fractures in the year following vertebral augmentation is similar to the fracture rate seen in patients not treated with osteoporosis therapy.44
Assessment of the impact of vertebral augmentation procedures on the cost of care is limited by the lack of high‐quality clinical studies.45, 46 Randomized controlled trials evaluating the benefit and risk of these procedures compared to conservative management are underway.4749 Pending further evidence, these procedures are best reserved for patients who fail to benefit from other measures to control pain and improve mobility.
Long Term
A comprehensive discussion of the long‐term management of osteoporosis is beyond the scope of this work. However, the inpatient setting presents an opportune time to initiate long‐term medical therapy. Studies show that the majority of patients who sustain osteoporotic fractures do not receive pharmacologic treatment for osteoporosis.5053 Hospitalists have the opportunity to start medications that can reduce the risk for subsequent fracture by nearly 50%.5458 A total calcium intake of 1200 to 1500 mg per day and vitamin D of 400 to 800 IU per day are recommended for all postmenopausal women. Patients who smoke should receive smoking cessation counseling and be considered for pharmacologic treatment for tobacco dependence. All patients should be assessed for fall risk, including a review of medications and assessment of alcohol intake.
Before considering pharmacologic treatment for osteoporosis, secondary causes of low bone mass must be excluded. Bisphosphonates are generally considered first‐line pharmacologic therapy for osteoporosis. Alendronate (Fosamax), risedronate (Actonel), and ibandronate (Boniva) have been shown in randomized trials to increase bone density and reduce the risk of osteoporotic fractures.55, 56, 59 Daily, weekly, and monthly preparations of bisphosphonates now exist. Pill‐induced esophagitis is a potential adverse effect of bisphosphonate therapy, but is extremely rare if proper precautions are taken. Patients should take oral bisphosphonates on an empty stomach, with a full glass of water, sitting upright, and have nothing to eat or drink for at least one half hour. If compliance with oral bisphosphonates is not possible, or esophageal abnormalities preclude oral bisphosphonate use, one may consider the use of intravenous ibandronate or zoledronic acid (Reclast). A 3‐year randomized controlled trial of yearly zoledronic acid improved bone density and reduced the incidence of osteoporotic fractures.60 Bisphosphonates are generally not recommended when creatinine clearance is less than 30 mL/minute. Other pharmacologic options for the treatment of osteoporosis include selective estrogen receptor modulators and anabolic agents. The reader is referred to an excellent review by Rosen61 for additional discussion of these therapies. The American College of Rheumatology clinical guidelines for the management of glucocorticoid induced osteoporosis are also worthy of review.62
Hospitalists are naturally suited to improve the quality of care for patients hospitalized with vertebral fractures. Most patients who currently sustain osteoporotic fractures do not receive appropriate evaluation and treatment. One study used an interdisciplinary team to identify, assess, and begin treatment for appropriate patients hospitalized with osteoporotic fractures.63 The intervention resulted in significantly more patients taking osteoporosis treatment medications 6 months after the incident fracture.
Conclusions
Acute vertebral fracture is a common clinical problem associated with significant morbidity and increased risk of mortality. Treatment of vertebral fracture should include analgesics and physical therapy. Percutaneous augmentation procedures may be considered in patients who fail optimal medical therapy. Because most vertebral fractures are due to osteoporosis and the healthcare system currently fails to appropriately assess and treat most patients who have sustained osteoporotic fractures, hospitalists are in an optimal position to initiate long‐term preventative treatment for these patients.
An 89‐year‐old female presents to the Emergency Department with lower back pain for the past 5 days. The patient has a past medical history of polymyalgia rheumatica and hypothyroidism. Her medications include prednisone 10 mg daily and levothyroxine 50 g daily. Aside from tenderness over the third lumbar vertebra, her physical exam is unremarkable. An x‐ray shows a fracture of the third lumbar vertebra. Basic laboratory studies, including calcium and creatinine are normal.
The Clinical Problem and Impact
Vertebral fractures (often termed vertebral compression fractures) affect approximately 25% of all postmenopausal women.1, 2 Only one‐third of vertebral fractures are brought to medical attention.3, 4 In the remaining two‐thirds, patients are either asymptomatic or do not seek medical attention. The lifetime risk of a clinical vertebral fracture is approximately 16% and 5% in white women and men, respectively.1 The risk of vertebral fracture increases with age, lower bone mineral density (BMD), and prior vertebral fracture.1, 5 Women with a preexisting vertebral fracture have a 5‐fold increased risk for a new vertebral fracture relative to those without a history of vertebral fracture.5, 6 Approximately 20% of women who sustain a vertebral fracture will have a new vertebral fracture in the subsequent year.5, 6 Vertebral fractures are frequently overlooked on chest x‐rays and hence there is a need for increased awareness and improved recognition of radiographically‐demonstrated fractures.7 Hospitalists must appreciate that a vertebral fracture is often the first clue to underlying osteoporosis. At least 90% of vertebral and hip fractures are attributable to osteoporosis.5
Typically, the pain related to an acute vertebral fracture improves over 4 to 6 weeks.8, 9 However, pain can persist, resulting in functional impairment, and a decline in the quality of life.1013 Vertebral fractures may lead to kyphosis and reduced lung function. This, in turn, may increase the risk for pneumonia, the most common cause of death in patients with osteoporosis.4 Both clinical and subclinical vertebral fractures are, in fact, independently associated with increased mortality,4, 14, 15 particularly in the period immediately following the event.16 The economic impact of osteoporosis and its related fractures is substantial.1719 In 1995, the annual direct medical cost for the inpatient care of vertebral fractures was estimated to be $575 million.19
Evidence‐Based Approach to the Hospitalized Patient
Evaluation
Vertebral fractures most commonly occur between T7 and L4. Acute vertebral fracture pain is typically sudden in onset and located in the mid to lower back. The pain may occur while performing an ordinary task such as lifting an object or bending over, although in many cases there is no preceding trauma. Physical activity exacerbates the pain and patients' movements may be limited due to pain. Spinal tenderness is usually present. A history of weight loss, prior malignancy, or fever should serve as red flags to the hospitalist and prompt evaluation for underlying malignancy or infection.
For a suspected fracture, frontal and lateral radiographs of the thoracolumbar spine are the initial imaging of choice. Magnetic resonance imaging (MRI) is useful when infection, malignancy, or spinal cord compression is suspected. The presence of neurological deficits should always prompt imaging with MRI or computed tomography (CT).
Patients with vertebral fractures should be evaluated with bone density testing, as osteoporosis is usually the underlying etiology. Because a number of medical conditions commonly contribute to bone loss (Table 1), laboratory testing is indicated for most patients. Although debate exists as to the optimal testing strategy,20 the National Osteoporosis Foundation guidelines recommend the evaluation of blood count, chemistry, and thyroid‐stimulating hormone (TSH) in patients with osteoporosis.21 Because vitamin D deficiency is common in patients who sustain osteoporotic fractures, 25‐hydroxyvitamin D levels should be checked in most patients.2225 Depending on the clinical scenario, additional testing may include the following: serum testosterone, serum intact parathyroid hormone (PTH), 24‐hour calcium excretion, serum protein electrophoresis, urine protein electrophoresis, erythrocyte sedimentation rate, and celiac sprue antibodies.26
| Endocrine disease or metabolic causes | Hypogonadism |
| Cushing's syndrome | |
| Hyperthyroidism | |
| Anorexia nervosa | |
| Hyperparathyroidism | |
| Nutritional conditions | Vitamin D deficiency |
| Calcium deficiency | |
| Malabsorption | |
| Drugs | Glucocorticoids |
| Antiepileptic drugs | |
| Excessive thyroid medication | |
| Long‐term heparin or low molecular weight heparin therapy (eg, >1 month) | |
| Other | Multiple myeloma |
| Rheumatoid arthritis | |
| Organ transplantation |
Management
Short Term
Short‐term management goals include the relief of pain and recovery of mobility. Nonsteroidal antiinflammatory drugs (NSAIDs) and low‐dose opioid medications should be used first in an effort to relieve pain. Beyond its potential effect on bone density, calcitonin (Miacalcin, Fortical) has long been used in acute vertebral fractures for analgesia. A systematic review of 5 randomized controlled trials concluded that calcitonin significantly reduced the pain from acute vertebral fractures.27 Calcitonin improved pain as early as 1 week into treatment and the benefit was persistent at 4 weeks. The analgesic mechanism of action for calcitonin is not entirely clear. Proposed mechanisms include increased ‐endorphin release, an antiinflammatory effect, and a direct effect on specific receptors in the central nervous system.27, 28
Pamidronate (Aredia) also has shown efficacy at reducing acute fracture pain. In a double‐blinded trial, Armigeat et al.29 evaluated pamidronate 30 mg daily for 3 days as compared to placebo in 32 patients. Pain scores were improved at 7 and 30 days with pamidronate. The mechanism of analgesia is unclear. Bisphosphonates are known to inhibit osteoclast activity, but may also work by blocking the effect of inflammatory cytokines.29
Early pain relief is critical in order to encourage physical activity. Bed rest should be avoided, as immobility may increase the risk for pressure ulcers, venous thromboembolism, and pneumonia.3033 Although bracing is frequently used in acute vertebral fracture, the modality has not been formally studied. Although also not well studied in the acute setting, physical therapy has been shown to reduce pain and improve functioning for patients with chronic pain from vertebral fracture,34 and is generally recommended.35
Percutaneous vertebral augmentation procedures include vertebroplasty and kyphoplasty. In vertebroplasty, polymethylmethacrylate cement is injected through a needle under fluoroscopic guidance into the collapsed vertebral body. With kyphoplasty, balloon tamps are used to elevate vertebral endplates prior to injection of cement (Figure 1).36 The proposed mechanism of action for both procedures is stabilization of the fracture by the hardened polymethylmethacrylate cement. These procedures are commonly performed by interventional radiologists without the need for general anesthesia; however, depending on the institution, they may be done by orthopedic surgeons, neurosurgeons, or anesthesiologists. The procedure can be performed as an outpatient, if indicated. Although the volume of these procedures has grown dramatically in recent years,37, 38 the quality of evidence supporting their use is relatively weak.3941 Only 1 randomized controlled trial has been published evaluating the potential benefit of vertebroplasty over conservative management.42 Voormolen et al.42 evaluated patients with vertebral fractures and pain refractive to 6 weeks of optimal medical therapy. Patients were treated with vertebroplasty or continuation of medical therapy. Vertebroplasty significantly improved pain initially, but not after 2 weeks. Like the study by Voormolen et al.,42 most studies evaluating percutaneous vertebral augmentation procedures have been conducted on patients with long‐term pain refractory to medical management. One notable exception is a nonrandomized trial published by Diamond et al.6 In that study, 55 patients were treated with vertebroplasty while 24 were treated conservatively. Pain at 24 hours was significantly improved in patients treated with vertebroplasty. At 6 weeks, however, there was no difference among the 2 groups.

The risk of short‐term complications from vertebral augmentation procedures is difficult to assess in light of the small sample sizes and methodological limitations of existing studies. Cement leakage occurs in 40% to 41% of patients treated with vertebroplasty, as compared with 8% to 9% with kyphoplasty.39, 41 Pulmonary emboli occur in 0.6% and 0.01% of patients treated with vertebroplasty and kyphoplasty, respectively, while neurologic complications occur in 0.6% and 0.03% of patients.41 Concern exists about whether percutaneous vertebral augmentation procedures might increase the risk for subsequent fractures,43, 44 as the incidence of new fractures appears to be elevated in the period immediately following the procedure and approximately two‐thirds of new fractures occur in vertebrae adjacent to the augmented vertebra.39, 41, 44 However, the 20% incidence of new vertebral fractures in the year following vertebral augmentation is similar to the fracture rate seen in patients not treated with osteoporosis therapy.44
Assessment of the impact of vertebral augmentation procedures on the cost of care is limited by the lack of high‐quality clinical studies.45, 46 Randomized controlled trials evaluating the benefit and risk of these procedures compared to conservative management are underway.4749 Pending further evidence, these procedures are best reserved for patients who fail to benefit from other measures to control pain and improve mobility.
Long Term
A comprehensive discussion of the long‐term management of osteoporosis is beyond the scope of this work. However, the inpatient setting presents an opportune time to initiate long‐term medical therapy. Studies show that the majority of patients who sustain osteoporotic fractures do not receive pharmacologic treatment for osteoporosis.5053 Hospitalists have the opportunity to start medications that can reduce the risk for subsequent fracture by nearly 50%.5458 A total calcium intake of 1200 to 1500 mg per day and vitamin D of 400 to 800 IU per day are recommended for all postmenopausal women. Patients who smoke should receive smoking cessation counseling and be considered for pharmacologic treatment for tobacco dependence. All patients should be assessed for fall risk, including a review of medications and assessment of alcohol intake.
Before considering pharmacologic treatment for osteoporosis, secondary causes of low bone mass must be excluded. Bisphosphonates are generally considered first‐line pharmacologic therapy for osteoporosis. Alendronate (Fosamax), risedronate (Actonel), and ibandronate (Boniva) have been shown in randomized trials to increase bone density and reduce the risk of osteoporotic fractures.55, 56, 59 Daily, weekly, and monthly preparations of bisphosphonates now exist. Pill‐induced esophagitis is a potential adverse effect of bisphosphonate therapy, but is extremely rare if proper precautions are taken. Patients should take oral bisphosphonates on an empty stomach, with a full glass of water, sitting upright, and have nothing to eat or drink for at least one half hour. If compliance with oral bisphosphonates is not possible, or esophageal abnormalities preclude oral bisphosphonate use, one may consider the use of intravenous ibandronate or zoledronic acid (Reclast). A 3‐year randomized controlled trial of yearly zoledronic acid improved bone density and reduced the incidence of osteoporotic fractures.60 Bisphosphonates are generally not recommended when creatinine clearance is less than 30 mL/minute. Other pharmacologic options for the treatment of osteoporosis include selective estrogen receptor modulators and anabolic agents. The reader is referred to an excellent review by Rosen61 for additional discussion of these therapies. The American College of Rheumatology clinical guidelines for the management of glucocorticoid induced osteoporosis are also worthy of review.62
Hospitalists are naturally suited to improve the quality of care for patients hospitalized with vertebral fractures. Most patients who currently sustain osteoporotic fractures do not receive appropriate evaluation and treatment. One study used an interdisciplinary team to identify, assess, and begin treatment for appropriate patients hospitalized with osteoporotic fractures.63 The intervention resulted in significantly more patients taking osteoporosis treatment medications 6 months after the incident fracture.
Conclusions
Acute vertebral fracture is a common clinical problem associated with significant morbidity and increased risk of mortality. Treatment of vertebral fracture should include analgesics and physical therapy. Percutaneous augmentation procedures may be considered in patients who fail optimal medical therapy. Because most vertebral fractures are due to osteoporosis and the healthcare system currently fails to appropriately assess and treat most patients who have sustained osteoporotic fractures, hospitalists are in an optimal position to initiate long‐term preventative treatment for these patients.
- .Epidemiology of spinal osteoporosis.Spine.1997;22(24 suppl):2S–11S.
- ,,,,,.Prevalence and incidence of vertebral deformities.Osteoporos Int.1993;3(3):113–119.
- ,,.The epidemiology of vertebral fractures. European Vertebral Osteoporosis Study Group.Bone.1993;14(suppl 1):S89–S97.
- ,,,,,.Vertebral fractures and mortality in older women: a prospective study. Study of Osteoporotic Fractures Research Group.Arch Intern Med.1999;159(11):1215–1220.
- ,,,.Pre‐existing fractures and bone mass predict vertebral fracture incidence in women.Ann Intern Med.1991;114(11):919–923.
- ,,, et al.Risk of new vertebral fracture in the year following a fracture.JAMA.2001;285(3):320–323.
- ,,,,,.Recognition of vertebral fracture in a clinical setting.Osteoporos Int.2000;11(7):577–582.
- ,,.Management of acute osteoporotic vertebral fractures: a nonrandomized trial comparing percutaneous vertebroplasty with conservative therapy.Am J Med.2003;114(4):257–265.
- .The clinical consequences of vertebral compression fracture.Bone.1992;13(suppl 2):S27–S31.
- ,,,.A case‐control study of quality of life and functional impairment in women with long‐standing vertebral osteoporotic fracture.Osteoporos Int.1999;9(6):508–515.
- ,,, et al.The burden of prevalent fractures on health‐related quality of life in postmenopausal women with osteoporosis: the IMOF study.J Rheumatol.2007;34(7):1551–1560.
- ,,,,.The relationship of health‐related quality of life to prevalent and incident vertebral fractures in postmenopausal women with osteoporosis: results from the Multiple Outcomes of Raloxifene Evaluation Study.Arthritis Rheum2001;44(11):2611–2619.
- ,,, et al.The association of radiographically detected vertebral fractures with back pain and function: a prospective study.Ann Int Med.1998;128:793–800.
- ,,,,.Mortality after all major types of osteoporotic fracture in men and women: an observational study.Lancet. 131999;353(9156):878–882.
- ,,,,.Population‐based study of survival after osteoporotic fractures.Am J Epidemiol.1993;137(9):1001–1005.
- ,,,,.Excess mortality after hospitalisation for vertebral fracture.Osteoporos Int.2004;15(2):108–112.
- ,.The cost of treating osteoporotic fractures in the United Kingdom female population.Osteoporos Int.1998;8(6):611–617.
- ,,, et al.Direct medical costs attributable to osteoporotic fractures.Osteoporos Int.2002;13(4):323–330.
- ,,,.Medical expenditures for the treatment of osteoporotic fractures in the United States in 1995: report from the National Osteoporosis Foundation.J Bone Miner Res.1997;12(1):24–35.
- .Laboratory workup for osteoporosis. Which tests are most cost‐effective?Postgrad Med.2003;114(3):35–38,41–34.
- National Osteoporosis Foundation. Clinician's Guide to Prevention and Treatment of Osteoporosis. Available at: http://www.nof.org/professionals/Clinicians_Guide.htm. Accessed February2009.
- ,,, et al.Prevalence of vitamin D inadequacy among postmenopausal North American women receiving osteoporosis therapy.J Clin Endocr Metab.2005;90:3215–3224.
- ,,,,.Prevalence of vitamin D inadequacy in a minimal trauma fracture population.Curr Med Res Opin.2005;21:1069–1074.
- ,,,,,.Occult vitamin D deficiency in postmenopausal US women with acute hip fracture.JAMA.1999;281:1505–1511.
- ,,,,.Secondary contributors for bone loss in osteoporotic hip fractures.Osteoporos Int.2008;19(7):991–999.
- .Evaluation of the patient with osteoporosis or at risk for osteoporosis. In: Marcus R, Feldman D, Kelsey J, eds.Osteoporosis. Vol.2.San Diego:Academic Press;2001:403–408.
- ,,,,.Calcitonin for treating acute pain of osteoporotic vertebral compression fractures: a systematic review of randomized, controlled trials.Osteoporos Int.2005;16(10):1281–1290.
- .Possible mechanisms of the analgesic action of calcitonin.Bone.2002;30(5 suppl):80S–83S.
- ,,,,.Intravenous pamidronate for pain relief in recent osteoporotic vertebral compression fracture: a randomized double‐blind controlled study.Osteoporos Int.2006;17(11):1659–1665.
- ,,,,.Pressure ulcer risk factors among hospitalized patients with activity limitation.JAMA.1995;273(11):865–870.
- ,.Risk factors for venous thromboembolism.Circulation. 172003;107(23 suppl 1):I9–I16.
- ,,.Infectious diseases and mortality among US nursing home residents.Am J Public Health.1993;83(12):1739–1742.
- ,,,,.Risk factors for pneumonia and other lower respiratory tract infections in elderly residents of long‐term care facilities.Arch Intern Med. 271999;159(17):2058–2064.
- ,,,.Positive effects of physiotherapy on chronic pain and performance in osteoporosis.Osteoporos Int.1998;8(3):215–221.
- ,,, et al.Health professional's guide to rehabilitation of the patient with osteoporosis.Osteoporos Int.2003;14(suppl 2):S1–S22.
- ,,,.Vertebral compression fractures: manage aggressively to prevent sequelae.Cleve Clin J Med.2003;70(2):147–156. Reprinted with permission. Copyright (c) 2003 Cleveland Clinic Foundation. All rights reserved.
- ,,,.Vertebroplasty in the United States: guidance method and provider distribution, 2001–2003.Radiology.2007;243(1):166–170.
- ,,,,.Thoracic and lumbar vertebroplasties performed in US Medicare enrollees, 2001–2005.JAMA.2007;298(15):1760–1762.
- ,,.Balloon kyphoplasty and vertebroplasty for vertebral compression fractures: a comparative systematic review of efficacy and safety.Spine.2006;31(23):2747–2755.
- ,,,,.Efficacy and safety of balloon kyphoplasty in the treatment of vertebral compression fractures: a systematic review.Eur Spine J.2006;15(7):1050–1067.
- ,,,.Vertebroplasty and kyphoplasty: a systematic review of 69 clinical studies.Spine.2006;31(17):1983–2001.
- ,,, et al.Percutaneous vertebroplasty compared with optimal pain medication treatment: short‐term clinical outcome of patients with subacute or chronic painful osteoporotic vertebral compression fractures. The VERTOS study.AJNR Am J Neuroradiol.2007;28(3):555–560.
- ,.Recurrent fracture after vertebral kyphoplasty.Spine J.2006;6(5):488–493.
- ,.Does vertebroplasty cause incident vertebral fractures? A review of available data.AJNR Am J Neuroradiol.2006;27(7):1397–1403.
- Centers for Medicare and Medicaid Services. Agency for Healthcare Research and Quality (AHRQ). Technology Assessment. Percutaneous Kyphoplasty for Vertebral Fractures Caused by Osteoporosis and Malignancy, 2005. Available at: http://www.cms.hhs.gov/mcd/viewtechassess.asp?from2=viewtechassess.asp13(5):550–555.
- .Randomized vertebroplasty trials: current status and challenges.Acad Radiol.2006;13(5):546–549.
- ,,, et al.VERTOS II: percutaneous vertebroplasty versus conservative therapy in patients with painful osteoporotic vertebral compression fractures; rationale, objectives and design of a multicenter randomized controlled trial.Trials.2007;8(1):33.
- ,.Vertebroplasty: a promising but as yet unproven intervention for painful osteoporotic spinal fractures.Med J Aust.2006;185(7):351–352.
- ,,, et al.Low frequency of treatment of osteoporosis among postmenopausal women following a fracture.Arch Intern Med.2003;163(17):2052–2057.
- ,,,,.Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture.Am J Med.2000;109(4):326–328.
- ,,.Missing a therapeutic window of opportunity: an audit of patients attending a tertiary teaching hospital with potentially osteoporotic hip and wrist fractures.J Rheumatol.2001;28(11):2504–2508.
- ,,,,.Underuse of osteoporosis medications in elderly patients with fractures.Am J Med.2003;115(5):398–400.
- ,,, et al.Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group.Lancet.1996;348(9041):1535–1541.
- ,,,,,.Meta‐analyses of therapies for postmenopausal osteoporosis. IX: Summary of meta‐analyses of therapies for postmenopausal osteoporosis.Endocr Rev.2002;23(4):570–578.
- ,,, et al.Summary of meta‐analyses of therapies for postmenopausal osteoporosis and the relationship between bone density and fractures.Endocrinol Metab Clin North Am.2002;31(3):659–679, xii.
- ,,, et al.Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group.JAMA.1999;282(14):1344–1352.
- ,,, et al.Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group.N Engl J Med.2001;344(5):333–340.
- ,,, et al.Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis.J Bone Miner Res.2004;19(8):1241–1249.
- ,,, et al.Once‐yearly zoledronic acid for treatment of postmenopausal osteoporosis.N Engl J Med. 32007;356(18):1809–1822.
- .Clinical practice. Postmenopausal osteoporosis.N Engl J Med.2005;353(6):595–603.
- American College of Rheumatology Ad Hoc Committee on Glucocorticoid‐Induced Osteoporosis.Recommendations for the prevention and treatment of glucocorticoid‐induced osteoporosis: 2001 update. [Review].Arthritis Rheum.2001;44(7):1496–1503.
- ,,, et al.An osteoporosis and fracture intervention program increases the diagnosis and treatment for osteoporosis for patients with minimal trauma fractures.Jt Comm J Qual Patient Saf.2005;31(5):267–274.
- .Epidemiology of spinal osteoporosis.Spine.1997;22(24 suppl):2S–11S.
- ,,,,,.Prevalence and incidence of vertebral deformities.Osteoporos Int.1993;3(3):113–119.
- ,,.The epidemiology of vertebral fractures. European Vertebral Osteoporosis Study Group.Bone.1993;14(suppl 1):S89–S97.
- ,,,,,.Vertebral fractures and mortality in older women: a prospective study. Study of Osteoporotic Fractures Research Group.Arch Intern Med.1999;159(11):1215–1220.
- ,,,.Pre‐existing fractures and bone mass predict vertebral fracture incidence in women.Ann Intern Med.1991;114(11):919–923.
- ,,, et al.Risk of new vertebral fracture in the year following a fracture.JAMA.2001;285(3):320–323.
- ,,,,,.Recognition of vertebral fracture in a clinical setting.Osteoporos Int.2000;11(7):577–582.
- ,,.Management of acute osteoporotic vertebral fractures: a nonrandomized trial comparing percutaneous vertebroplasty with conservative therapy.Am J Med.2003;114(4):257–265.
- .The clinical consequences of vertebral compression fracture.Bone.1992;13(suppl 2):S27–S31.
- ,,,.A case‐control study of quality of life and functional impairment in women with long‐standing vertebral osteoporotic fracture.Osteoporos Int.1999;9(6):508–515.
- ,,, et al.The burden of prevalent fractures on health‐related quality of life in postmenopausal women with osteoporosis: the IMOF study.J Rheumatol.2007;34(7):1551–1560.
- ,,,,.The relationship of health‐related quality of life to prevalent and incident vertebral fractures in postmenopausal women with osteoporosis: results from the Multiple Outcomes of Raloxifene Evaluation Study.Arthritis Rheum2001;44(11):2611–2619.
- ,,, et al.The association of radiographically detected vertebral fractures with back pain and function: a prospective study.Ann Int Med.1998;128:793–800.
- ,,,,.Mortality after all major types of osteoporotic fracture in men and women: an observational study.Lancet. 131999;353(9156):878–882.
- ,,,,.Population‐based study of survival after osteoporotic fractures.Am J Epidemiol.1993;137(9):1001–1005.
- ,,,,.Excess mortality after hospitalisation for vertebral fracture.Osteoporos Int.2004;15(2):108–112.
- ,.The cost of treating osteoporotic fractures in the United Kingdom female population.Osteoporos Int.1998;8(6):611–617.
- ,,, et al.Direct medical costs attributable to osteoporotic fractures.Osteoporos Int.2002;13(4):323–330.
- ,,,.Medical expenditures for the treatment of osteoporotic fractures in the United States in 1995: report from the National Osteoporosis Foundation.J Bone Miner Res.1997;12(1):24–35.
- .Laboratory workup for osteoporosis. Which tests are most cost‐effective?Postgrad Med.2003;114(3):35–38,41–34.
- National Osteoporosis Foundation. Clinician's Guide to Prevention and Treatment of Osteoporosis. Available at: http://www.nof.org/professionals/Clinicians_Guide.htm. Accessed February2009.
- ,,, et al.Prevalence of vitamin D inadequacy among postmenopausal North American women receiving osteoporosis therapy.J Clin Endocr Metab.2005;90:3215–3224.
- ,,,,.Prevalence of vitamin D inadequacy in a minimal trauma fracture population.Curr Med Res Opin.2005;21:1069–1074.
- ,,,,,.Occult vitamin D deficiency in postmenopausal US women with acute hip fracture.JAMA.1999;281:1505–1511.
- ,,,,.Secondary contributors for bone loss in osteoporotic hip fractures.Osteoporos Int.2008;19(7):991–999.
- .Evaluation of the patient with osteoporosis or at risk for osteoporosis. In: Marcus R, Feldman D, Kelsey J, eds.Osteoporosis. Vol.2.San Diego:Academic Press;2001:403–408.
- ,,,,.Calcitonin for treating acute pain of osteoporotic vertebral compression fractures: a systematic review of randomized, controlled trials.Osteoporos Int.2005;16(10):1281–1290.
- .Possible mechanisms of the analgesic action of calcitonin.Bone.2002;30(5 suppl):80S–83S.
- ,,,,.Intravenous pamidronate for pain relief in recent osteoporotic vertebral compression fracture: a randomized double‐blind controlled study.Osteoporos Int.2006;17(11):1659–1665.
- ,,,,.Pressure ulcer risk factors among hospitalized patients with activity limitation.JAMA.1995;273(11):865–870.
- ,.Risk factors for venous thromboembolism.Circulation. 172003;107(23 suppl 1):I9–I16.
- ,,.Infectious diseases and mortality among US nursing home residents.Am J Public Health.1993;83(12):1739–1742.
- ,,,,.Risk factors for pneumonia and other lower respiratory tract infections in elderly residents of long‐term care facilities.Arch Intern Med. 271999;159(17):2058–2064.
- ,,,.Positive effects of physiotherapy on chronic pain and performance in osteoporosis.Osteoporos Int.1998;8(3):215–221.
- ,,, et al.Health professional's guide to rehabilitation of the patient with osteoporosis.Osteoporos Int.2003;14(suppl 2):S1–S22.
- ,,,.Vertebral compression fractures: manage aggressively to prevent sequelae.Cleve Clin J Med.2003;70(2):147–156. Reprinted with permission. Copyright (c) 2003 Cleveland Clinic Foundation. All rights reserved.
- ,,,.Vertebroplasty in the United States: guidance method and provider distribution, 2001–2003.Radiology.2007;243(1):166–170.
- ,,,,.Thoracic and lumbar vertebroplasties performed in US Medicare enrollees, 2001–2005.JAMA.2007;298(15):1760–1762.
- ,,.Balloon kyphoplasty and vertebroplasty for vertebral compression fractures: a comparative systematic review of efficacy and safety.Spine.2006;31(23):2747–2755.
- ,,,,.Efficacy and safety of balloon kyphoplasty in the treatment of vertebral compression fractures: a systematic review.Eur Spine J.2006;15(7):1050–1067.
- ,,,.Vertebroplasty and kyphoplasty: a systematic review of 69 clinical studies.Spine.2006;31(17):1983–2001.
- ,,, et al.Percutaneous vertebroplasty compared with optimal pain medication treatment: short‐term clinical outcome of patients with subacute or chronic painful osteoporotic vertebral compression fractures. The VERTOS study.AJNR Am J Neuroradiol.2007;28(3):555–560.
- ,.Recurrent fracture after vertebral kyphoplasty.Spine J.2006;6(5):488–493.
- ,.Does vertebroplasty cause incident vertebral fractures? A review of available data.AJNR Am J Neuroradiol.2006;27(7):1397–1403.
- Centers for Medicare and Medicaid Services. Agency for Healthcare Research and Quality (AHRQ). Technology Assessment. Percutaneous Kyphoplasty for Vertebral Fractures Caused by Osteoporosis and Malignancy, 2005. Available at: http://www.cms.hhs.gov/mcd/viewtechassess.asp?from2=viewtechassess.asp13(5):550–555.
- .Randomized vertebroplasty trials: current status and challenges.Acad Radiol.2006;13(5):546–549.
- ,,, et al.VERTOS II: percutaneous vertebroplasty versus conservative therapy in patients with painful osteoporotic vertebral compression fractures; rationale, objectives and design of a multicenter randomized controlled trial.Trials.2007;8(1):33.
- ,.Vertebroplasty: a promising but as yet unproven intervention for painful osteoporotic spinal fractures.Med J Aust.2006;185(7):351–352.
- ,,, et al.Low frequency of treatment of osteoporosis among postmenopausal women following a fracture.Arch Intern Med.2003;163(17):2052–2057.
- ,,,,.Failure to diagnose and treat osteoporosis in elderly patients hospitalized with hip fracture.Am J Med.2000;109(4):326–328.
- ,,.Missing a therapeutic window of opportunity: an audit of patients attending a tertiary teaching hospital with potentially osteoporotic hip and wrist fractures.J Rheumatol.2001;28(11):2504–2508.
- ,,,,.Underuse of osteoporosis medications in elderly patients with fractures.Am J Med.2003;115(5):398–400.
- ,,, et al.Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group.Lancet.1996;348(9041):1535–1541.
- ,,,,,.Meta‐analyses of therapies for postmenopausal osteoporosis. IX: Summary of meta‐analyses of therapies for postmenopausal osteoporosis.Endocr Rev.2002;23(4):570–578.
- ,,, et al.Summary of meta‐analyses of therapies for postmenopausal osteoporosis and the relationship between bone density and fractures.Endocrinol Metab Clin North Am.2002;31(3):659–679, xii.
- ,,, et al.Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy With Risedronate Therapy (VERT) Study Group.JAMA.1999;282(14):1344–1352.
- ,,, et al.Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group.N Engl J Med.2001;344(5):333–340.
- ,,, et al.Effects of oral ibandronate administered daily or intermittently on fracture risk in postmenopausal osteoporosis.J Bone Miner Res.2004;19(8):1241–1249.
- ,,, et al.Once‐yearly zoledronic acid for treatment of postmenopausal osteoporosis.N Engl J Med. 32007;356(18):1809–1822.
- .Clinical practice. Postmenopausal osteoporosis.N Engl J Med.2005;353(6):595–603.
- American College of Rheumatology Ad Hoc Committee on Glucocorticoid‐Induced Osteoporosis.Recommendations for the prevention and treatment of glucocorticoid‐induced osteoporosis: 2001 update. [Review].Arthritis Rheum.2001;44(7):1496–1503.
- ,,, et al.An osteoporosis and fracture intervention program increases the diagnosis and treatment for osteoporosis for patients with minimal trauma fractures.Jt Comm J Qual Patient Saf.2005;31(5):267–274.