User login
What’s growing on your stethoscope? (And what you can do about it)
Background Studies have shown that rubbing alcohol pads on stethoscope diaphragms can reduce bacterial colonization, but alcohol pads are used infrequently used and not always available.
Methods We conducted a prospective, single-blinded study to investigate whether simultaneously scrubbing hands and stethoscope head with alcohol-based hand foam would significantly reduce bacterial counts on the stethoscope. Using their own stethoscope, participants imprinted the stethoscope head onto a chocolate agar plate, then used alcohol-based hand foam to cleanse their hands while simultaneously rubbing the stethoscope head. Once the stethoscope heads were dry, the participants imprinted their stethoscope heads onto a second plate. After 48 hours’ incubation, we determined the bacterial counts for the prewash and post-wash plates, and compared the 2.
Results We analyzed a total of 184 cultures (from 92 stethoscopes). Both the mean (28 prewash vs 3 post-wash, P=.001) and median (11 prewash vs 1 post-wash, P=.001) colony counts were significantly greater before being cleansed. Three methicillin-resistant Staphylococcus aureus (MRSA) colonies were identified in the prewash period; all were destroyed by the foam. The estimated number of hand washes needed to prevent 1 MRSA colony is 31 (95% confidence interval [CI], 18-89).
Conclusion Simultaneously using hand foam to clean hands and stethoscope heads reduces bacterial counts on stethoscopes. Further research is needed to determine whether this intervention can reduce morbidity and mortality associated with bacterial infection.
More than 160 years after a Hungarian physician introduced a protocol of strict handwashing and instrument sterilization to hospital wards,1 many clinicians still don’t wash their hands regularly or properly sterilize their medical equipment.2,3 The lack of stringent infection control, both in inpatient and office settings, is exacerbated by the rise in antibiotic-resistant bacteria. Methicillin-resistant Staphylococcus aureus (MRSA), in particular, including community-acquired MRSA, accounts for infections ranging from severe skin lesions to sepsis, and an estimated 18,650 deaths annually.4,5
Waterless hand cleansers, such as alcohol-based foams and gels, improve handwashing compliance.6-8 These products are effective in reducing both bacterial and viral agents, are convenient to use, and may even be good for caregivers’ skin.9 But would they work on stethoscopes? Our study was designed to find out.
An often-neglected source of bacteria
Infection can spread from patient to patient, not only on hands, but also via fomites such as ventilators, computer keyboards, pagers, and stethoscopes.10-14 Antimicrobial stethoscope covers, including those impregnated with silver ions, do not decrease bacterial colonization; evidence suggests that their use may actually increase it.15 Studies indicate that rubbing alcohol pads on stethoscope diaphragms can reduce bacterial colonization, and it has been suggested that cleansing of stethoscopes daily may be as effective as more frequent cleaning.16 Unfortunately, many clinicians do not clean their stethoscopes on a regular basis.17 In addition, alcohol pads are not always available, and using them requires an extra step and produces waste.
An earlier study by a member of our research team (A.S., unpublished data, 2007) indicated that rubbing stethoscopes exposed to nonpathogenic Staphylococcus epidermidis with alcohol-based hand foam was comparable to using alcohol wipes in reducing bacterial counts. The primary objective of this study was to determine whether clinicians can simultaneously reduce bacteria on stethoscope heads and clean their hands with alcohol-based foam.
Methods
This study was a prospective, single-blinded, “before-and-after” trial—a design in which each participant served as his or her own control and used foam that was already available on site. The study was conducted at 1 community-based hospital and 1 satellite family health center; the study was approved by the hospital Institutional Review Board. A grant from St. Margaret’s Foundation covered the cost of the agar plates.
We began by asking the attending physicians, faculty, nurses, residents, and medical students who attended a grand rounds program to participate; we visited the satellite health facility to recruit participants, as well. We started with 93 participants, but 1 stethoscope was damaged during the study, so we ended up with 92 participants and 184 cultures.
Interventions
In the prewash, or “before” portion of the study, all participants imprinted the head of their stethoscope onto a chocolate agar plate. The clinicians then used a 62.5% ethyl alcohol-based foam to cleanse their hands, simultaneously rubbing the stethoscope head between their hands. After a brief drying time, the clinicians imprinted their stethoscope head onto a separate agar plate (the post-wash, or “after” component).
We did not tell participants how to wash their hands or for how long. We simply told them to cleanse their hands as they normally would and to rub the foam onto the stethoscope head, as well.
Randomization and measurement
Prior to data collection, randomly assigned ID numbers were recorded on the bottom of 200 agar plates, which were then placed in a box. One member of our research team gave each clinician 2 plates. Participants imprinted their stethoscope head onto the first plate and handed it to another investigator, who recorded the prewash ID numbers. Participants then performed the handwashing and stethoscope rub and repeated the imprinting procedure with the second plate. This time, the investigator recorded the professional role of each participant (eg, resident, attending, nurse, faculty) as well as the post-wash ID numbers.
After 48 hours at 35°C incubation, the plates were arranged in numerical order. A member of the research team then counted the number and identified the type of bacterial colonies on each plate and recorded the findings on a data sheet by ID number.
Validation
In order to validate the bacterial counts, the supervisor of the hospital laboratory—who had 20 years’ experience in examining cultures and served as the gold standard—independently examined a random sample of plates. We agreed in advance that any count that deviated by more than 7 (approximately half the effect the study was powered to detect) from the gold standard would require another investigator to intervene. This proved unnecessary as no such deviation was found.
Coagulase studies were performed on all plates with bacterial isolates, and gram staining was performed on selected plates, along with identification of gram-negative stains, using the Microscan (Siemens, New York, NY). An “honest broker”—the only person authorized to match the plates with the stethoscopes’ ID numbers—then matched the prewash and post-wash data by stethoscope and type of health care provider. Another investigator analyzed the final data sheet for accuracy.
Power and sample size
A pilot study was performed to obtain estimates of the average and variance of the bacterial counts in a control group of stethoscopes and to determine whether the act of imprinting the stethoscope itself would significantly reduce the colony counts. The results established that there was no statistical change in either the summary statistics or the distribution of the bacterial counts over the course of multiple imprinting.
Estimates obtained from the pilot study indicated that 58 stethoscopes would be sufficient to yield 80% power (alpha=0.05, 2-tailed) for detecting an average difference of 15 colony counts between the prewash and post-wash samples. Seventy-eight stethoscopes would increase the power to 90%. We ultimately tested 92 stethoscopes.
Statistical analysis
Descriptive statistical measures were calculated to examine the bacterial counts. Linear regression analysis was used to compute the correlation in the validation data. This before-and-after design results in “paired data,” and both parametric and nonparametric statistical tests were used. We used a paired t-test to test the mean difference in bacterial counts between the pre- and post-wash samples, and a random effects model to estimate the individual components of variance. The difference in the median bacterial counts was tested using the signed rank test. We used various diagnostic measures to examine the assumptions of the statistical tests; and means, medians, 95% confidence intervals (CIs), and P-values (using P<.05 as statistical significance) to report the results. The Bonferroni multiple comparisons procedure was used to determine whether the bacterial counts were statistically different among subgroups of health care providers. All statistical analyses were performed using SAS (Cary, NC) software.
Results
A total of 184 culture plates showing before and after samples for 92 stethoscopes were analyzed. The provider breakdown of the sample consisted of nurses (39%), residents (30%), attending physicians (15%), faculty (13%), and medical students (3%). Thirty-five (approximately 1 in 6) of the 184 plates were randomly sampled for validation. There was a high degree of reliability between the investigator’s bacterial counts and the bacterial counts of the gold standard (r=+0.98, P<.001).
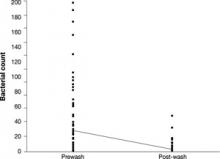
Bacterial counts. The distribution of the bacterial colony counts skewed right in both the prewash (0-198) and post-wash (0-48) samples. The FIGURE shows the skewed distributions in the actual bacterial counts for the 92 pairs of plates before and after hand and stethoscope washing. In the prewash sample, the mean bacterial count was 28.4 (95% CI, 20.2-36.6), vs a post-wash mean of 3.2 (95% CI, 1.8-4.6; P<.001). This resulted in an estimated difference in mean bacterial counts of 25.2 (95% CI, 17.2-33.3). The difference in the medians was also significant, with a prewash median of 11.5 and a post-wash median of 1.0 (P<.001). The difference between the pre- and post-wash periods remained significant even after using various transformations to normalize the data. Random effects modeling showed that very little (<5%) of the total variation was related to the type of health care provider.
Types of bacteria. The TABLE gives the breakdown and frequency of the various types of bacteria that we identified on the stethoscopes. Many were of low pathogenic potential, such as coagulase-negative staph species, which would not cause disease in healthy individuals. However, in hospitalized or immunocompromised patients, they could well induce illness. There were also several clearly pathogenic bacterial isolates, including 3 MRSA colonies (each on a different stethoscope), as well as Pseudomonas and Klebsiella. All of these isolates were killed by scrubbing with foam.
Considering only the MRSA colonies, the number needed to treat is 31 (95% CI, 18-89), indicating that for approximately every 31 hand- and stethoscope-washings with the alcohol-based foam, 1 MRSA colony could potentially be eliminated from a stethoscope head.
FIGURE
Bacterial counts: Prewash and post-wash
The line connects the mean values.
TABLE
What we found on the stethoscopes
| BACTERIA | TOTAL NUMBER OF ISOLATES |
|---|---|
| Coagulase-negative Staphylococcus | 100 |
| Bacillus | 51 |
| Micrococcus | 24 |
| Nonfermenting gram-negative bacteria | 17 |
| MRSA | 3 |
| Coagulase-positive Staphylococcus (non-MRSA) | 2 |
| Lactobacillus | 2 |
| Pseudomonas | 2 |
| Acinetobacter | 1 |
| Enterobacter | 1 |
| Klebsiella | 1 |
| Streptococcus | 1 |
| Zygomycetes | 1 |
| MRSA, methicillin-resistant Staphylococcus aureus. | |
Discussion
The findings of this study suggest that the use of alcohol-based hand foam to simultaneously sterilize the hands and a stethoscope head significantly reduces the number of bacterial colonies, including MRSA. The quantifiable risk of clinical infection with MRSA in patients through brief contact with a contaminated fomite such as a stethoscope is unknown. However, the transmission of the bacteria itself from contaminated surfaces and hands through brief contacts has been well established.11,12
A new standard for cleaning stethoscopes?
Swiping stethoscopes with alcohol pads is currently the gold standard for cleaning these instruments, but physicians do not consistently use alcohol pads for this purpose. Moreover, the pads must be purchased and available for use, require an extra step, and produce waste that must be disposed of—and clinicians still have to cleanse their hands, often using alcohol-based hand foam. Using the foam to cleanse the stethoscope while cleaning hands requires no added cost or additional time, and may reduce or prevent serious nosocomial and community-based infections.
Limitations of the study
One limitation of this study was the lack of control of the washing procedure. But because our goal was to see how the technique fared in actual use among all participants, uniform technique was not required. Knowing they were in a study may have altered the way the participants washed their hands and stethoscopes. If this were true, however, we would expect a much larger proportion of the total variation to be due to differences among clinicians than the 5% that was found.
This technique does not eliminate all bacteria—for instance, sporulating organisms such as Clostridium difficile are not killed by alcohol products.18 Yet friction alone has been found to reduce the number of these pathogens (A.S., unpublished data, 2007).
This study utilized alcohol-based hand foam because it was available at the study institution, so we cannot make any claims for nonalcohol-based products. It does appear, however, that alcohol-based foam may not be susceptible to bacterial resistance, as had previously been found in triclosan-containing products.19
It is not known whether the alcohol-based foam will damage stethoscope diaphragms. Previous studies have suggested that alcohol pads do cause damage to the rubber components of stethoscopes,16 but the foam studied here, like most similar products, contains emollients that may or may not have a protective effect. Another study would be necessary to fully assess this question.
While it is impossible to destroy all bacteria or eliminate all infections by simultaneous hand and stethoscope cleansing, many infections could potentially be prevented with this simple component of a comprehensive infection control program. Alcohol-based hand foam is already in use for hand cleansing between patients in many inpatient and outpatient settings, and this procedure requires no added cost and no additional time. Further research is necessary to determine whether the reduction of bacterial growth also corresponds to a reduction in clinically related disease. The results of this study provide evidence that hand foam, when used to simultaneously sterilize the hands and stethoscope, can significantly reduce the number of bacterial colonies on stethoscopes.
CORRESPONDENCE
Maryellen A. Schroeder, MD, MPH, UPMC St. Margaret, 815 Freeport Road, Pittsburgh, PA 15201; schroederma@upmc.edu
1. Semmelweis IP. Etiology, Concept and Prophylaxis of Childbed Fever. Trans. K C. Carter. Madison: University of Wisconsin; 1983.
2. Chandra PN, Milind K. Lapses in measures recommended for preventing hospital-acquired infection. J Hosp Infect. 2001;47:218-222.
3. Cohen HA, Amir J, Matalon A, et al. Stethoscopes and otoscopes—a potential vector of infection? Fam Pract. 1997;14:446-449.
4. Klevens RM, Morrison MA, Nadle J, et al. Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA. 2007;298:1763-1771.
5. Zeller JL. MRSA Infections. JAMA. 2007;298:1733.-
6. Langley JM. Commentary: waterless hand hygiene: if there’s a will, there’s a way. Pediatr Infect Dis J. 2002;21:496-497.
7. Harbarth S, Didier P, Grady L, et al. Interventional study to evaluate the impact of an alcohol-based hand gel in improving hand hygiene compliance. Pediatr Infect Dis J. 2002;21:489-495.
8. Seal LA, Rizer RL, Maas-Irslinger R. A unique water optional health care personnel handwash provides antimicrobial persistence and residual effects while decreasing the need for additional products. Am J Infect Control. 2005;33:207-216.
9. Larson EL, Aiello AE, Bastyr J, et al. Assessment of two hand hygiene regimens for intensive care unit personnel. Crit Care Med. 2001;29:944-951.
10. Devine J, Cooke RP, Wright EP. Is methicillin-resistant Staphylococcus aureus (MRSA) contamination of ward-based computer terminals a surrogate marker for nosocomial MRSA transmission and handwashing compliance? J Hosp Infect. 2001;48:72-75.
11. Maluf ME, Maldonado AF, Bercial ME, et al. Stethoscope: a friend or an enemy? Sao Paulo Med J. 2002;120:13-15.
12. Oie S, Hosokawa I, Kamiya A. Contamination of room door handles by methicillin-sensitive/methicillin-resistant Staphylococcus aureus. J Hosp Infect. 2002;51:140-143.
13. Zachary KC, Bayne PS, Morrison VJ, et al. Contamination of gowns, gloves, and stethoscopes with vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 2001;22:560-564.
14. Singh D, Kaur H, Gardner WG, et al. Bacterial contamination of hospital pagers. Infect Control Hosp Epidemiol. 2002;23:274-276.
15. Wood MW, Lund RC, Stevenson KB. Bacterial contamination of stethoscopes with antimicrobial diaphragm covers. Am J Infect Control. 2007;35:263-266.
16. Parmar RC, Valvi CC, Sira P, et al. A prospective, randomized, double-blind study of comparative efficacy of immediate versus daily cleaning of stethoscope using 66% ethyl alcohol. Indian J Med Sci. 2004;58:423-430.
17. Hill C, King T, Day R. A strategy to reduce MRSA colonization of stethoscopes. J Hosp Infect. 2006;62:122-123.
18. Weber DJ, Sickbert-Bennett E, Gergen MF, et al. Efficacy of selected hand hygiene agents used to remove Bacillus atrophaeus (a surrogate of Bacillus anthracis) from contaminated hands. JAMA. 2003;289:1274-1277.
19. Levy SB. Antibacterial household products: cause for concern. Emerging Infect Dis. 2001;7(3 suppl):512-515.
Background Studies have shown that rubbing alcohol pads on stethoscope diaphragms can reduce bacterial colonization, but alcohol pads are used infrequently used and not always available.
Methods We conducted a prospective, single-blinded study to investigate whether simultaneously scrubbing hands and stethoscope head with alcohol-based hand foam would significantly reduce bacterial counts on the stethoscope. Using their own stethoscope, participants imprinted the stethoscope head onto a chocolate agar plate, then used alcohol-based hand foam to cleanse their hands while simultaneously rubbing the stethoscope head. Once the stethoscope heads were dry, the participants imprinted their stethoscope heads onto a second plate. After 48 hours’ incubation, we determined the bacterial counts for the prewash and post-wash plates, and compared the 2.
Results We analyzed a total of 184 cultures (from 92 stethoscopes). Both the mean (28 prewash vs 3 post-wash, P=.001) and median (11 prewash vs 1 post-wash, P=.001) colony counts were significantly greater before being cleansed. Three methicillin-resistant Staphylococcus aureus (MRSA) colonies were identified in the prewash period; all were destroyed by the foam. The estimated number of hand washes needed to prevent 1 MRSA colony is 31 (95% confidence interval [CI], 18-89).
Conclusion Simultaneously using hand foam to clean hands and stethoscope heads reduces bacterial counts on stethoscopes. Further research is needed to determine whether this intervention can reduce morbidity and mortality associated with bacterial infection.
More than 160 years after a Hungarian physician introduced a protocol of strict handwashing and instrument sterilization to hospital wards,1 many clinicians still don’t wash their hands regularly or properly sterilize their medical equipment.2,3 The lack of stringent infection control, both in inpatient and office settings, is exacerbated by the rise in antibiotic-resistant bacteria. Methicillin-resistant Staphylococcus aureus (MRSA), in particular, including community-acquired MRSA, accounts for infections ranging from severe skin lesions to sepsis, and an estimated 18,650 deaths annually.4,5
Waterless hand cleansers, such as alcohol-based foams and gels, improve handwashing compliance.6-8 These products are effective in reducing both bacterial and viral agents, are convenient to use, and may even be good for caregivers’ skin.9 But would they work on stethoscopes? Our study was designed to find out.
An often-neglected source of bacteria
Infection can spread from patient to patient, not only on hands, but also via fomites such as ventilators, computer keyboards, pagers, and stethoscopes.10-14 Antimicrobial stethoscope covers, including those impregnated with silver ions, do not decrease bacterial colonization; evidence suggests that their use may actually increase it.15 Studies indicate that rubbing alcohol pads on stethoscope diaphragms can reduce bacterial colonization, and it has been suggested that cleansing of stethoscopes daily may be as effective as more frequent cleaning.16 Unfortunately, many clinicians do not clean their stethoscopes on a regular basis.17 In addition, alcohol pads are not always available, and using them requires an extra step and produces waste.
An earlier study by a member of our research team (A.S., unpublished data, 2007) indicated that rubbing stethoscopes exposed to nonpathogenic Staphylococcus epidermidis with alcohol-based hand foam was comparable to using alcohol wipes in reducing bacterial counts. The primary objective of this study was to determine whether clinicians can simultaneously reduce bacteria on stethoscope heads and clean their hands with alcohol-based foam.
Methods
This study was a prospective, single-blinded, “before-and-after” trial—a design in which each participant served as his or her own control and used foam that was already available on site. The study was conducted at 1 community-based hospital and 1 satellite family health center; the study was approved by the hospital Institutional Review Board. A grant from St. Margaret’s Foundation covered the cost of the agar plates.
We began by asking the attending physicians, faculty, nurses, residents, and medical students who attended a grand rounds program to participate; we visited the satellite health facility to recruit participants, as well. We started with 93 participants, but 1 stethoscope was damaged during the study, so we ended up with 92 participants and 184 cultures.
Interventions
In the prewash, or “before” portion of the study, all participants imprinted the head of their stethoscope onto a chocolate agar plate. The clinicians then used a 62.5% ethyl alcohol-based foam to cleanse their hands, simultaneously rubbing the stethoscope head between their hands. After a brief drying time, the clinicians imprinted their stethoscope head onto a separate agar plate (the post-wash, or “after” component).
We did not tell participants how to wash their hands or for how long. We simply told them to cleanse their hands as they normally would and to rub the foam onto the stethoscope head, as well.
Randomization and measurement
Prior to data collection, randomly assigned ID numbers were recorded on the bottom of 200 agar plates, which were then placed in a box. One member of our research team gave each clinician 2 plates. Participants imprinted their stethoscope head onto the first plate and handed it to another investigator, who recorded the prewash ID numbers. Participants then performed the handwashing and stethoscope rub and repeated the imprinting procedure with the second plate. This time, the investigator recorded the professional role of each participant (eg, resident, attending, nurse, faculty) as well as the post-wash ID numbers.
After 48 hours at 35°C incubation, the plates were arranged in numerical order. A member of the research team then counted the number and identified the type of bacterial colonies on each plate and recorded the findings on a data sheet by ID number.
Validation
In order to validate the bacterial counts, the supervisor of the hospital laboratory—who had 20 years’ experience in examining cultures and served as the gold standard—independently examined a random sample of plates. We agreed in advance that any count that deviated by more than 7 (approximately half the effect the study was powered to detect) from the gold standard would require another investigator to intervene. This proved unnecessary as no such deviation was found.
Coagulase studies were performed on all plates with bacterial isolates, and gram staining was performed on selected plates, along with identification of gram-negative stains, using the Microscan (Siemens, New York, NY). An “honest broker”—the only person authorized to match the plates with the stethoscopes’ ID numbers—then matched the prewash and post-wash data by stethoscope and type of health care provider. Another investigator analyzed the final data sheet for accuracy.
Power and sample size
A pilot study was performed to obtain estimates of the average and variance of the bacterial counts in a control group of stethoscopes and to determine whether the act of imprinting the stethoscope itself would significantly reduce the colony counts. The results established that there was no statistical change in either the summary statistics or the distribution of the bacterial counts over the course of multiple imprinting.
Estimates obtained from the pilot study indicated that 58 stethoscopes would be sufficient to yield 80% power (alpha=0.05, 2-tailed) for detecting an average difference of 15 colony counts between the prewash and post-wash samples. Seventy-eight stethoscopes would increase the power to 90%. We ultimately tested 92 stethoscopes.
Statistical analysis
Descriptive statistical measures were calculated to examine the bacterial counts. Linear regression analysis was used to compute the correlation in the validation data. This before-and-after design results in “paired data,” and both parametric and nonparametric statistical tests were used. We used a paired t-test to test the mean difference in bacterial counts between the pre- and post-wash samples, and a random effects model to estimate the individual components of variance. The difference in the median bacterial counts was tested using the signed rank test. We used various diagnostic measures to examine the assumptions of the statistical tests; and means, medians, 95% confidence intervals (CIs), and P-values (using P<.05 as statistical significance) to report the results. The Bonferroni multiple comparisons procedure was used to determine whether the bacterial counts were statistically different among subgroups of health care providers. All statistical analyses were performed using SAS (Cary, NC) software.
Results
A total of 184 culture plates showing before and after samples for 92 stethoscopes were analyzed. The provider breakdown of the sample consisted of nurses (39%), residents (30%), attending physicians (15%), faculty (13%), and medical students (3%). Thirty-five (approximately 1 in 6) of the 184 plates were randomly sampled for validation. There was a high degree of reliability between the investigator’s bacterial counts and the bacterial counts of the gold standard (r=+0.98, P<.001).
Bacterial counts. The distribution of the bacterial colony counts skewed right in both the prewash (0-198) and post-wash (0-48) samples. The FIGURE shows the skewed distributions in the actual bacterial counts for the 92 pairs of plates before and after hand and stethoscope washing. In the prewash sample, the mean bacterial count was 28.4 (95% CI, 20.2-36.6), vs a post-wash mean of 3.2 (95% CI, 1.8-4.6; P<.001). This resulted in an estimated difference in mean bacterial counts of 25.2 (95% CI, 17.2-33.3). The difference in the medians was also significant, with a prewash median of 11.5 and a post-wash median of 1.0 (P<.001). The difference between the pre- and post-wash periods remained significant even after using various transformations to normalize the data. Random effects modeling showed that very little (<5%) of the total variation was related to the type of health care provider.
Types of bacteria. The TABLE gives the breakdown and frequency of the various types of bacteria that we identified on the stethoscopes. Many were of low pathogenic potential, such as coagulase-negative staph species, which would not cause disease in healthy individuals. However, in hospitalized or immunocompromised patients, they could well induce illness. There were also several clearly pathogenic bacterial isolates, including 3 MRSA colonies (each on a different stethoscope), as well as Pseudomonas and Klebsiella. All of these isolates were killed by scrubbing with foam.
Considering only the MRSA colonies, the number needed to treat is 31 (95% CI, 18-89), indicating that for approximately every 31 hand- and stethoscope-washings with the alcohol-based foam, 1 MRSA colony could potentially be eliminated from a stethoscope head.
FIGURE
Bacterial counts: Prewash and post-wash
The line connects the mean values.
TABLE
What we found on the stethoscopes
| BACTERIA | TOTAL NUMBER OF ISOLATES |
|---|---|
| Coagulase-negative Staphylococcus | 100 |
| Bacillus | 51 |
| Micrococcus | 24 |
| Nonfermenting gram-negative bacteria | 17 |
| MRSA | 3 |
| Coagulase-positive Staphylococcus (non-MRSA) | 2 |
| Lactobacillus | 2 |
| Pseudomonas | 2 |
| Acinetobacter | 1 |
| Enterobacter | 1 |
| Klebsiella | 1 |
| Streptococcus | 1 |
| Zygomycetes | 1 |
| MRSA, methicillin-resistant Staphylococcus aureus. | |
Discussion
The findings of this study suggest that the use of alcohol-based hand foam to simultaneously sterilize the hands and a stethoscope head significantly reduces the number of bacterial colonies, including MRSA. The quantifiable risk of clinical infection with MRSA in patients through brief contact with a contaminated fomite such as a stethoscope is unknown. However, the transmission of the bacteria itself from contaminated surfaces and hands through brief contacts has been well established.11,12
A new standard for cleaning stethoscopes?
Swiping stethoscopes with alcohol pads is currently the gold standard for cleaning these instruments, but physicians do not consistently use alcohol pads for this purpose. Moreover, the pads must be purchased and available for use, require an extra step, and produce waste that must be disposed of—and clinicians still have to cleanse their hands, often using alcohol-based hand foam. Using the foam to cleanse the stethoscope while cleaning hands requires no added cost or additional time, and may reduce or prevent serious nosocomial and community-based infections.
Limitations of the study
One limitation of this study was the lack of control of the washing procedure. But because our goal was to see how the technique fared in actual use among all participants, uniform technique was not required. Knowing they were in a study may have altered the way the participants washed their hands and stethoscopes. If this were true, however, we would expect a much larger proportion of the total variation to be due to differences among clinicians than the 5% that was found.
This technique does not eliminate all bacteria—for instance, sporulating organisms such as Clostridium difficile are not killed by alcohol products.18 Yet friction alone has been found to reduce the number of these pathogens (A.S., unpublished data, 2007).
This study utilized alcohol-based hand foam because it was available at the study institution, so we cannot make any claims for nonalcohol-based products. It does appear, however, that alcohol-based foam may not be susceptible to bacterial resistance, as had previously been found in triclosan-containing products.19
It is not known whether the alcohol-based foam will damage stethoscope diaphragms. Previous studies have suggested that alcohol pads do cause damage to the rubber components of stethoscopes,16 but the foam studied here, like most similar products, contains emollients that may or may not have a protective effect. Another study would be necessary to fully assess this question.
While it is impossible to destroy all bacteria or eliminate all infections by simultaneous hand and stethoscope cleansing, many infections could potentially be prevented with this simple component of a comprehensive infection control program. Alcohol-based hand foam is already in use for hand cleansing between patients in many inpatient and outpatient settings, and this procedure requires no added cost and no additional time. Further research is necessary to determine whether the reduction of bacterial growth also corresponds to a reduction in clinically related disease. The results of this study provide evidence that hand foam, when used to simultaneously sterilize the hands and stethoscope, can significantly reduce the number of bacterial colonies on stethoscopes.
CORRESPONDENCE
Maryellen A. Schroeder, MD, MPH, UPMC St. Margaret, 815 Freeport Road, Pittsburgh, PA 15201; schroederma@upmc.edu
Background Studies have shown that rubbing alcohol pads on stethoscope diaphragms can reduce bacterial colonization, but alcohol pads are used infrequently used and not always available.
Methods We conducted a prospective, single-blinded study to investigate whether simultaneously scrubbing hands and stethoscope head with alcohol-based hand foam would significantly reduce bacterial counts on the stethoscope. Using their own stethoscope, participants imprinted the stethoscope head onto a chocolate agar plate, then used alcohol-based hand foam to cleanse their hands while simultaneously rubbing the stethoscope head. Once the stethoscope heads were dry, the participants imprinted their stethoscope heads onto a second plate. After 48 hours’ incubation, we determined the bacterial counts for the prewash and post-wash plates, and compared the 2.
Results We analyzed a total of 184 cultures (from 92 stethoscopes). Both the mean (28 prewash vs 3 post-wash, P=.001) and median (11 prewash vs 1 post-wash, P=.001) colony counts were significantly greater before being cleansed. Three methicillin-resistant Staphylococcus aureus (MRSA) colonies were identified in the prewash period; all were destroyed by the foam. The estimated number of hand washes needed to prevent 1 MRSA colony is 31 (95% confidence interval [CI], 18-89).
Conclusion Simultaneously using hand foam to clean hands and stethoscope heads reduces bacterial counts on stethoscopes. Further research is needed to determine whether this intervention can reduce morbidity and mortality associated with bacterial infection.
More than 160 years after a Hungarian physician introduced a protocol of strict handwashing and instrument sterilization to hospital wards,1 many clinicians still don’t wash their hands regularly or properly sterilize their medical equipment.2,3 The lack of stringent infection control, both in inpatient and office settings, is exacerbated by the rise in antibiotic-resistant bacteria. Methicillin-resistant Staphylococcus aureus (MRSA), in particular, including community-acquired MRSA, accounts for infections ranging from severe skin lesions to sepsis, and an estimated 18,650 deaths annually.4,5
Waterless hand cleansers, such as alcohol-based foams and gels, improve handwashing compliance.6-8 These products are effective in reducing both bacterial and viral agents, are convenient to use, and may even be good for caregivers’ skin.9 But would they work on stethoscopes? Our study was designed to find out.
An often-neglected source of bacteria
Infection can spread from patient to patient, not only on hands, but also via fomites such as ventilators, computer keyboards, pagers, and stethoscopes.10-14 Antimicrobial stethoscope covers, including those impregnated with silver ions, do not decrease bacterial colonization; evidence suggests that their use may actually increase it.15 Studies indicate that rubbing alcohol pads on stethoscope diaphragms can reduce bacterial colonization, and it has been suggested that cleansing of stethoscopes daily may be as effective as more frequent cleaning.16 Unfortunately, many clinicians do not clean their stethoscopes on a regular basis.17 In addition, alcohol pads are not always available, and using them requires an extra step and produces waste.
An earlier study by a member of our research team (A.S., unpublished data, 2007) indicated that rubbing stethoscopes exposed to nonpathogenic Staphylococcus epidermidis with alcohol-based hand foam was comparable to using alcohol wipes in reducing bacterial counts. The primary objective of this study was to determine whether clinicians can simultaneously reduce bacteria on stethoscope heads and clean their hands with alcohol-based foam.
Methods
This study was a prospective, single-blinded, “before-and-after” trial—a design in which each participant served as his or her own control and used foam that was already available on site. The study was conducted at 1 community-based hospital and 1 satellite family health center; the study was approved by the hospital Institutional Review Board. A grant from St. Margaret’s Foundation covered the cost of the agar plates.
We began by asking the attending physicians, faculty, nurses, residents, and medical students who attended a grand rounds program to participate; we visited the satellite health facility to recruit participants, as well. We started with 93 participants, but 1 stethoscope was damaged during the study, so we ended up with 92 participants and 184 cultures.
Interventions
In the prewash, or “before” portion of the study, all participants imprinted the head of their stethoscope onto a chocolate agar plate. The clinicians then used a 62.5% ethyl alcohol-based foam to cleanse their hands, simultaneously rubbing the stethoscope head between their hands. After a brief drying time, the clinicians imprinted their stethoscope head onto a separate agar plate (the post-wash, or “after” component).
We did not tell participants how to wash their hands or for how long. We simply told them to cleanse their hands as they normally would and to rub the foam onto the stethoscope head, as well.
Randomization and measurement
Prior to data collection, randomly assigned ID numbers were recorded on the bottom of 200 agar plates, which were then placed in a box. One member of our research team gave each clinician 2 plates. Participants imprinted their stethoscope head onto the first plate and handed it to another investigator, who recorded the prewash ID numbers. Participants then performed the handwashing and stethoscope rub and repeated the imprinting procedure with the second plate. This time, the investigator recorded the professional role of each participant (eg, resident, attending, nurse, faculty) as well as the post-wash ID numbers.
After 48 hours at 35°C incubation, the plates were arranged in numerical order. A member of the research team then counted the number and identified the type of bacterial colonies on each plate and recorded the findings on a data sheet by ID number.
Validation
In order to validate the bacterial counts, the supervisor of the hospital laboratory—who had 20 years’ experience in examining cultures and served as the gold standard—independently examined a random sample of plates. We agreed in advance that any count that deviated by more than 7 (approximately half the effect the study was powered to detect) from the gold standard would require another investigator to intervene. This proved unnecessary as no such deviation was found.
Coagulase studies were performed on all plates with bacterial isolates, and gram staining was performed on selected plates, along with identification of gram-negative stains, using the Microscan (Siemens, New York, NY). An “honest broker”—the only person authorized to match the plates with the stethoscopes’ ID numbers—then matched the prewash and post-wash data by stethoscope and type of health care provider. Another investigator analyzed the final data sheet for accuracy.
Power and sample size
A pilot study was performed to obtain estimates of the average and variance of the bacterial counts in a control group of stethoscopes and to determine whether the act of imprinting the stethoscope itself would significantly reduce the colony counts. The results established that there was no statistical change in either the summary statistics or the distribution of the bacterial counts over the course of multiple imprinting.
Estimates obtained from the pilot study indicated that 58 stethoscopes would be sufficient to yield 80% power (alpha=0.05, 2-tailed) for detecting an average difference of 15 colony counts between the prewash and post-wash samples. Seventy-eight stethoscopes would increase the power to 90%. We ultimately tested 92 stethoscopes.
Statistical analysis
Descriptive statistical measures were calculated to examine the bacterial counts. Linear regression analysis was used to compute the correlation in the validation data. This before-and-after design results in “paired data,” and both parametric and nonparametric statistical tests were used. We used a paired t-test to test the mean difference in bacterial counts between the pre- and post-wash samples, and a random effects model to estimate the individual components of variance. The difference in the median bacterial counts was tested using the signed rank test. We used various diagnostic measures to examine the assumptions of the statistical tests; and means, medians, 95% confidence intervals (CIs), and P-values (using P<.05 as statistical significance) to report the results. The Bonferroni multiple comparisons procedure was used to determine whether the bacterial counts were statistically different among subgroups of health care providers. All statistical analyses were performed using SAS (Cary, NC) software.
Results
A total of 184 culture plates showing before and after samples for 92 stethoscopes were analyzed. The provider breakdown of the sample consisted of nurses (39%), residents (30%), attending physicians (15%), faculty (13%), and medical students (3%). Thirty-five (approximately 1 in 6) of the 184 plates were randomly sampled for validation. There was a high degree of reliability between the investigator’s bacterial counts and the bacterial counts of the gold standard (r=+0.98, P<.001).
Bacterial counts. The distribution of the bacterial colony counts skewed right in both the prewash (0-198) and post-wash (0-48) samples. The FIGURE shows the skewed distributions in the actual bacterial counts for the 92 pairs of plates before and after hand and stethoscope washing. In the prewash sample, the mean bacterial count was 28.4 (95% CI, 20.2-36.6), vs a post-wash mean of 3.2 (95% CI, 1.8-4.6; P<.001). This resulted in an estimated difference in mean bacterial counts of 25.2 (95% CI, 17.2-33.3). The difference in the medians was also significant, with a prewash median of 11.5 and a post-wash median of 1.0 (P<.001). The difference between the pre- and post-wash periods remained significant even after using various transformations to normalize the data. Random effects modeling showed that very little (<5%) of the total variation was related to the type of health care provider.
Types of bacteria. The TABLE gives the breakdown and frequency of the various types of bacteria that we identified on the stethoscopes. Many were of low pathogenic potential, such as coagulase-negative staph species, which would not cause disease in healthy individuals. However, in hospitalized or immunocompromised patients, they could well induce illness. There were also several clearly pathogenic bacterial isolates, including 3 MRSA colonies (each on a different stethoscope), as well as Pseudomonas and Klebsiella. All of these isolates were killed by scrubbing with foam.
Considering only the MRSA colonies, the number needed to treat is 31 (95% CI, 18-89), indicating that for approximately every 31 hand- and stethoscope-washings with the alcohol-based foam, 1 MRSA colony could potentially be eliminated from a stethoscope head.
FIGURE
Bacterial counts: Prewash and post-wash
The line connects the mean values.
TABLE
What we found on the stethoscopes
| BACTERIA | TOTAL NUMBER OF ISOLATES |
|---|---|
| Coagulase-negative Staphylococcus | 100 |
| Bacillus | 51 |
| Micrococcus | 24 |
| Nonfermenting gram-negative bacteria | 17 |
| MRSA | 3 |
| Coagulase-positive Staphylococcus (non-MRSA) | 2 |
| Lactobacillus | 2 |
| Pseudomonas | 2 |
| Acinetobacter | 1 |
| Enterobacter | 1 |
| Klebsiella | 1 |
| Streptococcus | 1 |
| Zygomycetes | 1 |
| MRSA, methicillin-resistant Staphylococcus aureus. | |
Discussion
The findings of this study suggest that the use of alcohol-based hand foam to simultaneously sterilize the hands and a stethoscope head significantly reduces the number of bacterial colonies, including MRSA. The quantifiable risk of clinical infection with MRSA in patients through brief contact with a contaminated fomite such as a stethoscope is unknown. However, the transmission of the bacteria itself from contaminated surfaces and hands through brief contacts has been well established.11,12
A new standard for cleaning stethoscopes?
Swiping stethoscopes with alcohol pads is currently the gold standard for cleaning these instruments, but physicians do not consistently use alcohol pads for this purpose. Moreover, the pads must be purchased and available for use, require an extra step, and produce waste that must be disposed of—and clinicians still have to cleanse their hands, often using alcohol-based hand foam. Using the foam to cleanse the stethoscope while cleaning hands requires no added cost or additional time, and may reduce or prevent serious nosocomial and community-based infections.
Limitations of the study
One limitation of this study was the lack of control of the washing procedure. But because our goal was to see how the technique fared in actual use among all participants, uniform technique was not required. Knowing they were in a study may have altered the way the participants washed their hands and stethoscopes. If this were true, however, we would expect a much larger proportion of the total variation to be due to differences among clinicians than the 5% that was found.
This technique does not eliminate all bacteria—for instance, sporulating organisms such as Clostridium difficile are not killed by alcohol products.18 Yet friction alone has been found to reduce the number of these pathogens (A.S., unpublished data, 2007).
This study utilized alcohol-based hand foam because it was available at the study institution, so we cannot make any claims for nonalcohol-based products. It does appear, however, that alcohol-based foam may not be susceptible to bacterial resistance, as had previously been found in triclosan-containing products.19
It is not known whether the alcohol-based foam will damage stethoscope diaphragms. Previous studies have suggested that alcohol pads do cause damage to the rubber components of stethoscopes,16 but the foam studied here, like most similar products, contains emollients that may or may not have a protective effect. Another study would be necessary to fully assess this question.
While it is impossible to destroy all bacteria or eliminate all infections by simultaneous hand and stethoscope cleansing, many infections could potentially be prevented with this simple component of a comprehensive infection control program. Alcohol-based hand foam is already in use for hand cleansing between patients in many inpatient and outpatient settings, and this procedure requires no added cost and no additional time. Further research is necessary to determine whether the reduction of bacterial growth also corresponds to a reduction in clinically related disease. The results of this study provide evidence that hand foam, when used to simultaneously sterilize the hands and stethoscope, can significantly reduce the number of bacterial colonies on stethoscopes.
CORRESPONDENCE
Maryellen A. Schroeder, MD, MPH, UPMC St. Margaret, 815 Freeport Road, Pittsburgh, PA 15201; schroederma@upmc.edu
1. Semmelweis IP. Etiology, Concept and Prophylaxis of Childbed Fever. Trans. K C. Carter. Madison: University of Wisconsin; 1983.
2. Chandra PN, Milind K. Lapses in measures recommended for preventing hospital-acquired infection. J Hosp Infect. 2001;47:218-222.
3. Cohen HA, Amir J, Matalon A, et al. Stethoscopes and otoscopes—a potential vector of infection? Fam Pract. 1997;14:446-449.
4. Klevens RM, Morrison MA, Nadle J, et al. Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA. 2007;298:1763-1771.
5. Zeller JL. MRSA Infections. JAMA. 2007;298:1733.-
6. Langley JM. Commentary: waterless hand hygiene: if there’s a will, there’s a way. Pediatr Infect Dis J. 2002;21:496-497.
7. Harbarth S, Didier P, Grady L, et al. Interventional study to evaluate the impact of an alcohol-based hand gel in improving hand hygiene compliance. Pediatr Infect Dis J. 2002;21:489-495.
8. Seal LA, Rizer RL, Maas-Irslinger R. A unique water optional health care personnel handwash provides antimicrobial persistence and residual effects while decreasing the need for additional products. Am J Infect Control. 2005;33:207-216.
9. Larson EL, Aiello AE, Bastyr J, et al. Assessment of two hand hygiene regimens for intensive care unit personnel. Crit Care Med. 2001;29:944-951.
10. Devine J, Cooke RP, Wright EP. Is methicillin-resistant Staphylococcus aureus (MRSA) contamination of ward-based computer terminals a surrogate marker for nosocomial MRSA transmission and handwashing compliance? J Hosp Infect. 2001;48:72-75.
11. Maluf ME, Maldonado AF, Bercial ME, et al. Stethoscope: a friend or an enemy? Sao Paulo Med J. 2002;120:13-15.
12. Oie S, Hosokawa I, Kamiya A. Contamination of room door handles by methicillin-sensitive/methicillin-resistant Staphylococcus aureus. J Hosp Infect. 2002;51:140-143.
13. Zachary KC, Bayne PS, Morrison VJ, et al. Contamination of gowns, gloves, and stethoscopes with vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 2001;22:560-564.
14. Singh D, Kaur H, Gardner WG, et al. Bacterial contamination of hospital pagers. Infect Control Hosp Epidemiol. 2002;23:274-276.
15. Wood MW, Lund RC, Stevenson KB. Bacterial contamination of stethoscopes with antimicrobial diaphragm covers. Am J Infect Control. 2007;35:263-266.
16. Parmar RC, Valvi CC, Sira P, et al. A prospective, randomized, double-blind study of comparative efficacy of immediate versus daily cleaning of stethoscope using 66% ethyl alcohol. Indian J Med Sci. 2004;58:423-430.
17. Hill C, King T, Day R. A strategy to reduce MRSA colonization of stethoscopes. J Hosp Infect. 2006;62:122-123.
18. Weber DJ, Sickbert-Bennett E, Gergen MF, et al. Efficacy of selected hand hygiene agents used to remove Bacillus atrophaeus (a surrogate of Bacillus anthracis) from contaminated hands. JAMA. 2003;289:1274-1277.
19. Levy SB. Antibacterial household products: cause for concern. Emerging Infect Dis. 2001;7(3 suppl):512-515.
1. Semmelweis IP. Etiology, Concept and Prophylaxis of Childbed Fever. Trans. K C. Carter. Madison: University of Wisconsin; 1983.
2. Chandra PN, Milind K. Lapses in measures recommended for preventing hospital-acquired infection. J Hosp Infect. 2001;47:218-222.
3. Cohen HA, Amir J, Matalon A, et al. Stethoscopes and otoscopes—a potential vector of infection? Fam Pract. 1997;14:446-449.
4. Klevens RM, Morrison MA, Nadle J, et al. Active Bacterial Core surveillance (ABCs) MRSA Investigators. Invasive methicillin-resistant Staphylococcus aureus infections in the United States. JAMA. 2007;298:1763-1771.
5. Zeller JL. MRSA Infections. JAMA. 2007;298:1733.-
6. Langley JM. Commentary: waterless hand hygiene: if there’s a will, there’s a way. Pediatr Infect Dis J. 2002;21:496-497.
7. Harbarth S, Didier P, Grady L, et al. Interventional study to evaluate the impact of an alcohol-based hand gel in improving hand hygiene compliance. Pediatr Infect Dis J. 2002;21:489-495.
8. Seal LA, Rizer RL, Maas-Irslinger R. A unique water optional health care personnel handwash provides antimicrobial persistence and residual effects while decreasing the need for additional products. Am J Infect Control. 2005;33:207-216.
9. Larson EL, Aiello AE, Bastyr J, et al. Assessment of two hand hygiene regimens for intensive care unit personnel. Crit Care Med. 2001;29:944-951.
10. Devine J, Cooke RP, Wright EP. Is methicillin-resistant Staphylococcus aureus (MRSA) contamination of ward-based computer terminals a surrogate marker for nosocomial MRSA transmission and handwashing compliance? J Hosp Infect. 2001;48:72-75.
11. Maluf ME, Maldonado AF, Bercial ME, et al. Stethoscope: a friend or an enemy? Sao Paulo Med J. 2002;120:13-15.
12. Oie S, Hosokawa I, Kamiya A. Contamination of room door handles by methicillin-sensitive/methicillin-resistant Staphylococcus aureus. J Hosp Infect. 2002;51:140-143.
13. Zachary KC, Bayne PS, Morrison VJ, et al. Contamination of gowns, gloves, and stethoscopes with vancomycin-resistant enterococci. Infect Control Hosp Epidemiol. 2001;22:560-564.
14. Singh D, Kaur H, Gardner WG, et al. Bacterial contamination of hospital pagers. Infect Control Hosp Epidemiol. 2002;23:274-276.
15. Wood MW, Lund RC, Stevenson KB. Bacterial contamination of stethoscopes with antimicrobial diaphragm covers. Am J Infect Control. 2007;35:263-266.
16. Parmar RC, Valvi CC, Sira P, et al. A prospective, randomized, double-blind study of comparative efficacy of immediate versus daily cleaning of stethoscope using 66% ethyl alcohol. Indian J Med Sci. 2004;58:423-430.
17. Hill C, King T, Day R. A strategy to reduce MRSA colonization of stethoscopes. J Hosp Infect. 2006;62:122-123.
18. Weber DJ, Sickbert-Bennett E, Gergen MF, et al. Efficacy of selected hand hygiene agents used to remove Bacillus atrophaeus (a surrogate of Bacillus anthracis) from contaminated hands. JAMA. 2003;289:1274-1277.
19. Levy SB. Antibacterial household products: cause for concern. Emerging Infect Dis. 2001;7(3 suppl):512-515.
Sex with former patients: OK after retirement?
Dear Dr. Mossman,
A psychiatrist retires from practice and goes into some other line of work—perhaps managing a restaurant. He then has an “affair” with a former patient whom he had not treated for several years. Could the retired psychiatrist’s conduct be the basis of a successful lawsuit?—Submitted by “Dr. D”
Evidence tells us that the retired psychiatrist’s behavior likely could do emotional harm to his former patient. If the former patient suffers some injury, a successful suit could follow—if not on grounds of malpractice, then on other grounds. In this article we’ll see why by looking at:
- rates of doctor-patient sex
- potential harm from doctor-patient sex
- ethical bans on sex with former patients
- possible legal actions.
Sex with patients: Rates and risk
Doctors and patients often develop erotic thoughts about each other.1,2 But as Sigmund Freud noted almost a century ago, an actual love relationship between a doctor and a psychotherapy patient can cause a “complete defeat for the treatment” and destroy the patient’s chance for recovery.3
More than 5 decades later, surveys of medical professionals supplemented Freud’s observations with data about the frequency and impact of doctor-patient sex. In a 1973 survey, 11% of physicians said they had erotic contact with patients, and 5% reported intercourse.4 In a 1986 survey of psychiatrists, 3% of women and 7% of men acknowledged having sexual contact with patients.5 In a 1992 study of 10,000 nonpsychiatric physicians, 9% of respondents reported having sex with patients.6 Actual rates of doctor-patient sex probably are much higher than reported because physicians may be reluctant to admit to having erotic contact with patients, even in anonymous surveys.7 The typical therapist-patient sex scenario involves a male doctor and an adult female patient, but same-sex encounters and sexual contact with minors occur, too.8
Sex between a therapist and a patient is likely to cause emotional injury. For example, a 1991 study found that 90% of psychotherapy patients who had sexual involvement with a prior therapist had been harmed by the experience.9 Books, articles, and Web sites offer vivid individual accounts of harm patients have suffered ( Table ). Doctors who have sex with patients could face public opprobrium, civil lawsuits, actions against their medical licenses, and prosecution in states that make sex with psychiatric patients a criminal offense.10
Table
How sexual relationships can harm patients
| Type of harm | Explanation |
|---|---|
| Ambivalence | Psychological paralysis regarding whether to protect or take action against the abusive therapist |
| Cognitive dysfunction | Impaired memory and concentration, intrusive thoughts, flashbacks |
| Emotional lability | Unpredictable emotional responses, abrupt changes in mood, severe disruption of the patient’s typical way of feeling |
| Emptiness, isolation | Lost sense of self, feeling cut off from others |
| Guilt | Irrational self-blame for causing the sexual contact |
| Impaired trust | Fear of being taken advantage of, used, or abused in future therapy |
| Suicide | 14% of patients who had sex with a therapist attempt suicide; approximately 1% commit suicide |
| Role confusion | Treatment sessions and the therapeutic relationship serve the therapist’s needs rather than the patient’s; this perception may generalize to later therapies and other relationships |
| Sexual confusion | Examples include disgust with sexual feelings, uncertainty about sexual orientation, belief that self-worth comes from gratifying others’ sexual desires |
| Confusion about anger | Rage at self, self-loathing, need to suppress angry feelings, mistaken beliefs that others are angry at you |
| Source: Adapted from reference 8 | |
What about former patients?
Sex between providers and current patients is opposed by all major healthcare organizations, including the American Psychiatric Association (APA),11 American Medical Association,12 and American Psychological Association.13 The last 2 groups strongly discourage sex with former patients, but the APA’s ethics code states that such activity is always unethical.
The APA’s position reflects 2 general truths of psychiatric practice:
- Psychiatric patients often return for care years after initial treatment has ended. “Former patients” are really “possible future patients.” Improper relationships with former patients disrupt the doctor’s obligation to remain available for future care.
- Even if a patient never returns to treatment, intense feelings about a doctor can last for years. A psychiatrist who engages in sex with a former patient may evoke and manipulate feelings “left over” from therapy.
Psychiatrists therefore “have only one kind of relationship with a patient—that is, a doctor-patient relationship.”14 Moreover, as Simon and Shuman observe, “[N]o patient [is] strong enough, no pause is long enough, and no love is true enough to justify compromising this boundary.”15
Legal actions
If a physician no longer practices medicine, can any of his activities—including with a former patient—be malpractice? In fact, sex between practicing doctors and current patients might not always be malpractice. If a psychiatrist gains sexual access to a patient by saying that the sex will be therapeutic, the psychiatrist has perpetrated fraud and this intentional action might not be covered by malpractice insurance.16
In several cases involving nonpsychiatric physicians,17 courts have held that consensual doctor-patient sex is not malpractice, though it might represent some other form of wrongdoing. The argument is that sex with a patient is an intentional act that is never a professional service, whereas malpractice by definition arises unintentionally from negligence while rendering professional services. Other courts, however, have held that doctor-patient sex can be malpractice because it breaches the physician’s fiduciary relationship and can constitute an abuse of power.18
After retirement, physicians still have responsibilities to former patients: to protect records, to respect confidentiality, and to release information upon proper requests. Some fiduciary duties to patients survive the conclusion of treatment, and behavior that breaches those responsibilities can bring legal action.
Psychiatrists should realize that many former patients remain vulnerable because of feelings “left over” from therapy. Therefore, potential civil actions against a retired psychiatrist might include:
A suit for intentional infliction of emotional distress. This tort action requires proving more than mere insults or indignities; it occurs only when someone “by extreme and outrageous conduct intentionally or recklessly causes severe emotional distress to another.”19 Initiating sex with a former patient is strongly disapproved and meets the legal criterion of having a high probability of causing mental distress.20
A suit for negligent infliction of emotional distress. Modern law permits recourse for negligently inflicted emotional distress when harm occurs in “the course of specified categories of activities, undertakings, or relationships in which the negligent conduct is especially likely to cause emotional disturbance.”21
Suits for exploitation. Some jurisdictions allow suits against therapists who have sex with former patients, irrespective of therapists’ license status. For example, Minnesota allows lawsuits for “sexual exploitation” if the former patient’s capacity to consent was impaired by emotional dependence on the psychotherapist.22
Actions by licensing boards. Many retired practitioners maintain their medical licenses. Retired-but-still-licensed psychiatrists can be subject to professional disciplinary actions.
- Submit your malpractice-related questions to Dr. Mossman at douglas.mossman@dowdenhealth.com.
- Include your name, address, and practice location. If your question is chosen for publication, your name can be withheld by request.
- All readers who submit questions will be included in quarterly drawings for a $50 gift certificate for Professional Risk Management Services, Inc’s online marketplace of risk management publications and resources (www.prms.com).
1. Pope KS, Keith-Spiegel P, Tabachnick BG. Sexual attraction to clients. The human therapist and the (sometimes) inhuman training system. Am Psychol. 1986;4:147-158.
2. Golden GA, Brennan M. Managing erotic feelings in the physician-patient relationship. CMAJ. 1995;153:1241-1245.
3. Freud S. Observations on transference-love. In: Strachey J, ed. Complete psychological works of Sigmund Freud, standard edition, vol 12. London, UK: Hogarth Press; 1958:157-173.
4. Kardener SH, Fuller M, Mensh IN. A survey of physicians’ attitudes and practice regarding erotic and non-erotic contact with patients. Am J Psychiatry. 1973;130:1077-1081.
5. Gartrell N, Herman J, Olarte S, et al. Psychiatrist-patient sexual contact: results of a national survey, 1: prevalence. Am J Psychiatry. 1986;143:1126-1131.
6. Gartrell N, Milliken N, Goodson WH, et al. Physician-patient sexual contact—prevalence and problems. West J Med. 1992;157:139-143.
7. Roman B, Kay J. Residency education on the prevention of physician-patient sexual misconduct. Acad Psychiatry. 1997;21:26-34.
8. Pope KS. Sex between therapists and clients. In: Worrell J, ed. Encyclopedia of women and gender: sex similarities and differences and the impact of society on gender. New York, NY: Academic Press; 2001;955-962.
9. Pope KS, Vetter VA. Prior therapist-patient sexual involvement among patients seen by psychologists. Psychotherapy. 1991;28:429-438.
10. Simon RI. Clinical psychiatry and the law, 2nd edition. Arlington, VA: American Psychiatric Publishing, Inc.; 2003.
11. American Psychiatric Association. The principles of medical ethics with annotations especially applicable to psychiatry. Available at: http://www.psych.org/MainMenu/PsychiatricPractice/Ethics/
ResourcesStandards/PrinciplesofMedicalEthics.aspx. Accessed May 4, 2009.
12. American Medical Association Council on Ethical and Judicial Affairs. Sexual misconduct in the practice of medicine. JAMA. 1991;266:2741-2745.
13. American Psychological Association. Ethical principles of psychologists and code of conduct. Available at: http://www.apa.org/ethics/code2002.html. Accessed May 4, 2009.
14. Gruenberg PB. Boundary violations. In: Ethics primer of the American Psychiatric Association. Washington, DC: American Psychiatric Association; 2001;1-9.
15. Simon RI, Shuman DW. Clinical manual of psychiatry and the law. Arlington, VA: American Psychiatric Publishing, Inc.; 2007.
16. Sadock BJ, Sadock VA. Kaplan and Sadock’s synopsis of psychiatry, 10th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2007.
17. Clemente v Roth, 171 Fed. Appx. 999 (4th Cir. Md. 2006).
18. Hoopes v Hammargren, 102 Nev. 425, 725 P.2d 238 (1986).
19. Restatement (Third) of Torts: Liability for Physical Harm, ch 8, §45 (2007 draft).
20. Prosser WL, Keeton WP, Dobbs DB, et al. Prosser and Keeton on torts, 5th ed. St. Paul, MN: West Publishing Co.; 1984.
21. Restatement (Third) of Torts: Liability for Physical Harm, ch 8, §46 (2007 draft).
22. Minnesota Statutes §148A (2008).
Dear Dr. Mossman,
A psychiatrist retires from practice and goes into some other line of work—perhaps managing a restaurant. He then has an “affair” with a former patient whom he had not treated for several years. Could the retired psychiatrist’s conduct be the basis of a successful lawsuit?—Submitted by “Dr. D”
Evidence tells us that the retired psychiatrist’s behavior likely could do emotional harm to his former patient. If the former patient suffers some injury, a successful suit could follow—if not on grounds of malpractice, then on other grounds. In this article we’ll see why by looking at:
- rates of doctor-patient sex
- potential harm from doctor-patient sex
- ethical bans on sex with former patients
- possible legal actions.
Sex with patients: Rates and risk
Doctors and patients often develop erotic thoughts about each other.1,2 But as Sigmund Freud noted almost a century ago, an actual love relationship between a doctor and a psychotherapy patient can cause a “complete defeat for the treatment” and destroy the patient’s chance for recovery.3
More than 5 decades later, surveys of medical professionals supplemented Freud’s observations with data about the frequency and impact of doctor-patient sex. In a 1973 survey, 11% of physicians said they had erotic contact with patients, and 5% reported intercourse.4 In a 1986 survey of psychiatrists, 3% of women and 7% of men acknowledged having sexual contact with patients.5 In a 1992 study of 10,000 nonpsychiatric physicians, 9% of respondents reported having sex with patients.6 Actual rates of doctor-patient sex probably are much higher than reported because physicians may be reluctant to admit to having erotic contact with patients, even in anonymous surveys.7 The typical therapist-patient sex scenario involves a male doctor and an adult female patient, but same-sex encounters and sexual contact with minors occur, too.8
Sex between a therapist and a patient is likely to cause emotional injury. For example, a 1991 study found that 90% of psychotherapy patients who had sexual involvement with a prior therapist had been harmed by the experience.9 Books, articles, and Web sites offer vivid individual accounts of harm patients have suffered ( Table ). Doctors who have sex with patients could face public opprobrium, civil lawsuits, actions against their medical licenses, and prosecution in states that make sex with psychiatric patients a criminal offense.10
Table
How sexual relationships can harm patients
| Type of harm | Explanation |
|---|---|
| Ambivalence | Psychological paralysis regarding whether to protect or take action against the abusive therapist |
| Cognitive dysfunction | Impaired memory and concentration, intrusive thoughts, flashbacks |
| Emotional lability | Unpredictable emotional responses, abrupt changes in mood, severe disruption of the patient’s typical way of feeling |
| Emptiness, isolation | Lost sense of self, feeling cut off from others |
| Guilt | Irrational self-blame for causing the sexual contact |
| Impaired trust | Fear of being taken advantage of, used, or abused in future therapy |
| Suicide | 14% of patients who had sex with a therapist attempt suicide; approximately 1% commit suicide |
| Role confusion | Treatment sessions and the therapeutic relationship serve the therapist’s needs rather than the patient’s; this perception may generalize to later therapies and other relationships |
| Sexual confusion | Examples include disgust with sexual feelings, uncertainty about sexual orientation, belief that self-worth comes from gratifying others’ sexual desires |
| Confusion about anger | Rage at self, self-loathing, need to suppress angry feelings, mistaken beliefs that others are angry at you |
| Source: Adapted from reference 8 | |
What about former patients?
Sex between providers and current patients is opposed by all major healthcare organizations, including the American Psychiatric Association (APA),11 American Medical Association,12 and American Psychological Association.13 The last 2 groups strongly discourage sex with former patients, but the APA’s ethics code states that such activity is always unethical.
The APA’s position reflects 2 general truths of psychiatric practice:
- Psychiatric patients often return for care years after initial treatment has ended. “Former patients” are really “possible future patients.” Improper relationships with former patients disrupt the doctor’s obligation to remain available for future care.
- Even if a patient never returns to treatment, intense feelings about a doctor can last for years. A psychiatrist who engages in sex with a former patient may evoke and manipulate feelings “left over” from therapy.
Psychiatrists therefore “have only one kind of relationship with a patient—that is, a doctor-patient relationship.”14 Moreover, as Simon and Shuman observe, “[N]o patient [is] strong enough, no pause is long enough, and no love is true enough to justify compromising this boundary.”15
Legal actions
If a physician no longer practices medicine, can any of his activities—including with a former patient—be malpractice? In fact, sex between practicing doctors and current patients might not always be malpractice. If a psychiatrist gains sexual access to a patient by saying that the sex will be therapeutic, the psychiatrist has perpetrated fraud and this intentional action might not be covered by malpractice insurance.16
In several cases involving nonpsychiatric physicians,17 courts have held that consensual doctor-patient sex is not malpractice, though it might represent some other form of wrongdoing. The argument is that sex with a patient is an intentional act that is never a professional service, whereas malpractice by definition arises unintentionally from negligence while rendering professional services. Other courts, however, have held that doctor-patient sex can be malpractice because it breaches the physician’s fiduciary relationship and can constitute an abuse of power.18
After retirement, physicians still have responsibilities to former patients: to protect records, to respect confidentiality, and to release information upon proper requests. Some fiduciary duties to patients survive the conclusion of treatment, and behavior that breaches those responsibilities can bring legal action.
Psychiatrists should realize that many former patients remain vulnerable because of feelings “left over” from therapy. Therefore, potential civil actions against a retired psychiatrist might include:
A suit for intentional infliction of emotional distress. This tort action requires proving more than mere insults or indignities; it occurs only when someone “by extreme and outrageous conduct intentionally or recklessly causes severe emotional distress to another.”19 Initiating sex with a former patient is strongly disapproved and meets the legal criterion of having a high probability of causing mental distress.20
A suit for negligent infliction of emotional distress. Modern law permits recourse for negligently inflicted emotional distress when harm occurs in “the course of specified categories of activities, undertakings, or relationships in which the negligent conduct is especially likely to cause emotional disturbance.”21
Suits for exploitation. Some jurisdictions allow suits against therapists who have sex with former patients, irrespective of therapists’ license status. For example, Minnesota allows lawsuits for “sexual exploitation” if the former patient’s capacity to consent was impaired by emotional dependence on the psychotherapist.22
Actions by licensing boards. Many retired practitioners maintain their medical licenses. Retired-but-still-licensed psychiatrists can be subject to professional disciplinary actions.
- Submit your malpractice-related questions to Dr. Mossman at douglas.mossman@dowdenhealth.com.
- Include your name, address, and practice location. If your question is chosen for publication, your name can be withheld by request.
- All readers who submit questions will be included in quarterly drawings for a $50 gift certificate for Professional Risk Management Services, Inc’s online marketplace of risk management publications and resources (www.prms.com).
Dear Dr. Mossman,
A psychiatrist retires from practice and goes into some other line of work—perhaps managing a restaurant. He then has an “affair” with a former patient whom he had not treated for several years. Could the retired psychiatrist’s conduct be the basis of a successful lawsuit?—Submitted by “Dr. D”
Evidence tells us that the retired psychiatrist’s behavior likely could do emotional harm to his former patient. If the former patient suffers some injury, a successful suit could follow—if not on grounds of malpractice, then on other grounds. In this article we’ll see why by looking at:
- rates of doctor-patient sex
- potential harm from doctor-patient sex
- ethical bans on sex with former patients
- possible legal actions.
Sex with patients: Rates and risk
Doctors and patients often develop erotic thoughts about each other.1,2 But as Sigmund Freud noted almost a century ago, an actual love relationship between a doctor and a psychotherapy patient can cause a “complete defeat for the treatment” and destroy the patient’s chance for recovery.3
More than 5 decades later, surveys of medical professionals supplemented Freud’s observations with data about the frequency and impact of doctor-patient sex. In a 1973 survey, 11% of physicians said they had erotic contact with patients, and 5% reported intercourse.4 In a 1986 survey of psychiatrists, 3% of women and 7% of men acknowledged having sexual contact with patients.5 In a 1992 study of 10,000 nonpsychiatric physicians, 9% of respondents reported having sex with patients.6 Actual rates of doctor-patient sex probably are much higher than reported because physicians may be reluctant to admit to having erotic contact with patients, even in anonymous surveys.7 The typical therapist-patient sex scenario involves a male doctor and an adult female patient, but same-sex encounters and sexual contact with minors occur, too.8
Sex between a therapist and a patient is likely to cause emotional injury. For example, a 1991 study found that 90% of psychotherapy patients who had sexual involvement with a prior therapist had been harmed by the experience.9 Books, articles, and Web sites offer vivid individual accounts of harm patients have suffered ( Table ). Doctors who have sex with patients could face public opprobrium, civil lawsuits, actions against their medical licenses, and prosecution in states that make sex with psychiatric patients a criminal offense.10
Table
How sexual relationships can harm patients
| Type of harm | Explanation |
|---|---|
| Ambivalence | Psychological paralysis regarding whether to protect or take action against the abusive therapist |
| Cognitive dysfunction | Impaired memory and concentration, intrusive thoughts, flashbacks |
| Emotional lability | Unpredictable emotional responses, abrupt changes in mood, severe disruption of the patient’s typical way of feeling |
| Emptiness, isolation | Lost sense of self, feeling cut off from others |
| Guilt | Irrational self-blame for causing the sexual contact |
| Impaired trust | Fear of being taken advantage of, used, or abused in future therapy |
| Suicide | 14% of patients who had sex with a therapist attempt suicide; approximately 1% commit suicide |
| Role confusion | Treatment sessions and the therapeutic relationship serve the therapist’s needs rather than the patient’s; this perception may generalize to later therapies and other relationships |
| Sexual confusion | Examples include disgust with sexual feelings, uncertainty about sexual orientation, belief that self-worth comes from gratifying others’ sexual desires |
| Confusion about anger | Rage at self, self-loathing, need to suppress angry feelings, mistaken beliefs that others are angry at you |
| Source: Adapted from reference 8 | |
What about former patients?
Sex between providers and current patients is opposed by all major healthcare organizations, including the American Psychiatric Association (APA),11 American Medical Association,12 and American Psychological Association.13 The last 2 groups strongly discourage sex with former patients, but the APA’s ethics code states that such activity is always unethical.
The APA’s position reflects 2 general truths of psychiatric practice:
- Psychiatric patients often return for care years after initial treatment has ended. “Former patients” are really “possible future patients.” Improper relationships with former patients disrupt the doctor’s obligation to remain available for future care.
- Even if a patient never returns to treatment, intense feelings about a doctor can last for years. A psychiatrist who engages in sex with a former patient may evoke and manipulate feelings “left over” from therapy.
Psychiatrists therefore “have only one kind of relationship with a patient—that is, a doctor-patient relationship.”14 Moreover, as Simon and Shuman observe, “[N]o patient [is] strong enough, no pause is long enough, and no love is true enough to justify compromising this boundary.”15
Legal actions
If a physician no longer practices medicine, can any of his activities—including with a former patient—be malpractice? In fact, sex between practicing doctors and current patients might not always be malpractice. If a psychiatrist gains sexual access to a patient by saying that the sex will be therapeutic, the psychiatrist has perpetrated fraud and this intentional action might not be covered by malpractice insurance.16
In several cases involving nonpsychiatric physicians,17 courts have held that consensual doctor-patient sex is not malpractice, though it might represent some other form of wrongdoing. The argument is that sex with a patient is an intentional act that is never a professional service, whereas malpractice by definition arises unintentionally from negligence while rendering professional services. Other courts, however, have held that doctor-patient sex can be malpractice because it breaches the physician’s fiduciary relationship and can constitute an abuse of power.18
After retirement, physicians still have responsibilities to former patients: to protect records, to respect confidentiality, and to release information upon proper requests. Some fiduciary duties to patients survive the conclusion of treatment, and behavior that breaches those responsibilities can bring legal action.
Psychiatrists should realize that many former patients remain vulnerable because of feelings “left over” from therapy. Therefore, potential civil actions against a retired psychiatrist might include:
A suit for intentional infliction of emotional distress. This tort action requires proving more than mere insults or indignities; it occurs only when someone “by extreme and outrageous conduct intentionally or recklessly causes severe emotional distress to another.”19 Initiating sex with a former patient is strongly disapproved and meets the legal criterion of having a high probability of causing mental distress.20
A suit for negligent infliction of emotional distress. Modern law permits recourse for negligently inflicted emotional distress when harm occurs in “the course of specified categories of activities, undertakings, or relationships in which the negligent conduct is especially likely to cause emotional disturbance.”21
Suits for exploitation. Some jurisdictions allow suits against therapists who have sex with former patients, irrespective of therapists’ license status. For example, Minnesota allows lawsuits for “sexual exploitation” if the former patient’s capacity to consent was impaired by emotional dependence on the psychotherapist.22
Actions by licensing boards. Many retired practitioners maintain their medical licenses. Retired-but-still-licensed psychiatrists can be subject to professional disciplinary actions.
- Submit your malpractice-related questions to Dr. Mossman at douglas.mossman@dowdenhealth.com.
- Include your name, address, and practice location. If your question is chosen for publication, your name can be withheld by request.
- All readers who submit questions will be included in quarterly drawings for a $50 gift certificate for Professional Risk Management Services, Inc’s online marketplace of risk management publications and resources (www.prms.com).
1. Pope KS, Keith-Spiegel P, Tabachnick BG. Sexual attraction to clients. The human therapist and the (sometimes) inhuman training system. Am Psychol. 1986;4:147-158.
2. Golden GA, Brennan M. Managing erotic feelings in the physician-patient relationship. CMAJ. 1995;153:1241-1245.
3. Freud S. Observations on transference-love. In: Strachey J, ed. Complete psychological works of Sigmund Freud, standard edition, vol 12. London, UK: Hogarth Press; 1958:157-173.
4. Kardener SH, Fuller M, Mensh IN. A survey of physicians’ attitudes and practice regarding erotic and non-erotic contact with patients. Am J Psychiatry. 1973;130:1077-1081.
5. Gartrell N, Herman J, Olarte S, et al. Psychiatrist-patient sexual contact: results of a national survey, 1: prevalence. Am J Psychiatry. 1986;143:1126-1131.
6. Gartrell N, Milliken N, Goodson WH, et al. Physician-patient sexual contact—prevalence and problems. West J Med. 1992;157:139-143.
7. Roman B, Kay J. Residency education on the prevention of physician-patient sexual misconduct. Acad Psychiatry. 1997;21:26-34.
8. Pope KS. Sex between therapists and clients. In: Worrell J, ed. Encyclopedia of women and gender: sex similarities and differences and the impact of society on gender. New York, NY: Academic Press; 2001;955-962.
9. Pope KS, Vetter VA. Prior therapist-patient sexual involvement among patients seen by psychologists. Psychotherapy. 1991;28:429-438.
10. Simon RI. Clinical psychiatry and the law, 2nd edition. Arlington, VA: American Psychiatric Publishing, Inc.; 2003.
11. American Psychiatric Association. The principles of medical ethics with annotations especially applicable to psychiatry. Available at: http://www.psych.org/MainMenu/PsychiatricPractice/Ethics/
ResourcesStandards/PrinciplesofMedicalEthics.aspx. Accessed May 4, 2009.
12. American Medical Association Council on Ethical and Judicial Affairs. Sexual misconduct in the practice of medicine. JAMA. 1991;266:2741-2745.
13. American Psychological Association. Ethical principles of psychologists and code of conduct. Available at: http://www.apa.org/ethics/code2002.html. Accessed May 4, 2009.
14. Gruenberg PB. Boundary violations. In: Ethics primer of the American Psychiatric Association. Washington, DC: American Psychiatric Association; 2001;1-9.
15. Simon RI, Shuman DW. Clinical manual of psychiatry and the law. Arlington, VA: American Psychiatric Publishing, Inc.; 2007.
16. Sadock BJ, Sadock VA. Kaplan and Sadock’s synopsis of psychiatry, 10th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2007.
17. Clemente v Roth, 171 Fed. Appx. 999 (4th Cir. Md. 2006).
18. Hoopes v Hammargren, 102 Nev. 425, 725 P.2d 238 (1986).
19. Restatement (Third) of Torts: Liability for Physical Harm, ch 8, §45 (2007 draft).
20. Prosser WL, Keeton WP, Dobbs DB, et al. Prosser and Keeton on torts, 5th ed. St. Paul, MN: West Publishing Co.; 1984.
21. Restatement (Third) of Torts: Liability for Physical Harm, ch 8, §46 (2007 draft).
22. Minnesota Statutes §148A (2008).
1. Pope KS, Keith-Spiegel P, Tabachnick BG. Sexual attraction to clients. The human therapist and the (sometimes) inhuman training system. Am Psychol. 1986;4:147-158.
2. Golden GA, Brennan M. Managing erotic feelings in the physician-patient relationship. CMAJ. 1995;153:1241-1245.
3. Freud S. Observations on transference-love. In: Strachey J, ed. Complete psychological works of Sigmund Freud, standard edition, vol 12. London, UK: Hogarth Press; 1958:157-173.
4. Kardener SH, Fuller M, Mensh IN. A survey of physicians’ attitudes and practice regarding erotic and non-erotic contact with patients. Am J Psychiatry. 1973;130:1077-1081.
5. Gartrell N, Herman J, Olarte S, et al. Psychiatrist-patient sexual contact: results of a national survey, 1: prevalence. Am J Psychiatry. 1986;143:1126-1131.
6. Gartrell N, Milliken N, Goodson WH, et al. Physician-patient sexual contact—prevalence and problems. West J Med. 1992;157:139-143.
7. Roman B, Kay J. Residency education on the prevention of physician-patient sexual misconduct. Acad Psychiatry. 1997;21:26-34.
8. Pope KS. Sex between therapists and clients. In: Worrell J, ed. Encyclopedia of women and gender: sex similarities and differences and the impact of society on gender. New York, NY: Academic Press; 2001;955-962.
9. Pope KS, Vetter VA. Prior therapist-patient sexual involvement among patients seen by psychologists. Psychotherapy. 1991;28:429-438.
10. Simon RI. Clinical psychiatry and the law, 2nd edition. Arlington, VA: American Psychiatric Publishing, Inc.; 2003.
11. American Psychiatric Association. The principles of medical ethics with annotations especially applicable to psychiatry. Available at: http://www.psych.org/MainMenu/PsychiatricPractice/Ethics/
ResourcesStandards/PrinciplesofMedicalEthics.aspx. Accessed May 4, 2009.
12. American Medical Association Council on Ethical and Judicial Affairs. Sexual misconduct in the practice of medicine. JAMA. 1991;266:2741-2745.
13. American Psychological Association. Ethical principles of psychologists and code of conduct. Available at: http://www.apa.org/ethics/code2002.html. Accessed May 4, 2009.
14. Gruenberg PB. Boundary violations. In: Ethics primer of the American Psychiatric Association. Washington, DC: American Psychiatric Association; 2001;1-9.
15. Simon RI, Shuman DW. Clinical manual of psychiatry and the law. Arlington, VA: American Psychiatric Publishing, Inc.; 2007.
16. Sadock BJ, Sadock VA. Kaplan and Sadock’s synopsis of psychiatry, 10th ed. Baltimore, MD: Lippincott Williams & Wilkins; 2007.
17. Clemente v Roth, 171 Fed. Appx. 999 (4th Cir. Md. 2006).
18. Hoopes v Hammargren, 102 Nev. 425, 725 P.2d 238 (1986).
19. Restatement (Third) of Torts: Liability for Physical Harm, ch 8, §45 (2007 draft).
20. Prosser WL, Keeton WP, Dobbs DB, et al. Prosser and Keeton on torts, 5th ed. St. Paul, MN: West Publishing Co.; 1984.
21. Restatement (Third) of Torts: Liability for Physical Harm, ch 8, §46 (2007 draft).
22. Minnesota Statutes §148A (2008).
The Child With a Suspicious Cough
A thorough differential diagnosis, primarily based on history and physical examination, is essential when a child presents with a suspicious cough. Certain imaging modalities are also useful for diagnosis.
Identification of an underlying cause is crucial. When doing your history and physical exam, look for something that does not fit a routine presentation. For example, a cough in the presence of a constitutional change, such as weight loss, can indicate a more serious problem. In addition, a cough with a relatively sudden onset or one associated with labored breathing can be worrisome. Also, a choking episode followed by sudden cough, for example, can indicate the presence of a foreign body.
Asthma is the most common cause of chronic cough in the pediatric population, but also consider less common etiologies such as tracheoesophageal fistula, cystic fibrosis (CF), and bronchopulmonary dysplasia. Failure to thrive, clubbing, cardiac signs, and persistent stridor suggest alternative diagnoses.
Patient age offers some guidance in your differential diagnosis. In a neonate (younger than 28 days), persistent cough might suggest an infection or a congenital anomaly such as compression of the esophagus and trachea by a vascular ring. Infectious etiologies include rhinovirus, adenovirus, respiratory syncytial virus, and pertussis.
In preschool children, think upper or lower respiratory tract infection, rhinitis, postnasal drip syndrome, gastroesophageal reflux, an irritant source (such as passive smoking or air pollution), and, of course, asthma.
Among school-age children and adolescents, consider the same possibilities, but add inhalant or other substance abuse to your list of possible irritant causes. In addition, these older children can develop psychogenic or “habit” cough, one that is absent during sleep, distraction, or periods of concentration. Vocal cord dysfunction, also known as laryngeal wheeze, is another possibility in this group.
General pediatricians commonly treat children with a cough that lasts 5-10 days in the context of an upper respiratory tract illness, such as a cold. If a child still coughs incessantly after other cold symptoms have resolved, I would be concerned. This is not necessarily a call to refer the patient to a specialist, but this scenario is a call to do further diagnostic evaluation.
If the child already is diagnosed with asthma and develops a cough, determine whether the patient is taking the appropriate medication and/or is compliant with therapy. Also, ask about the child's environment, particularly the presence of passive smoking, dust, and pets.
In terms of allergy testing, I recommend a radioallergosorbent allergen-specific IgE antibody assay. This is indicated if a child has other lateral symptoms, such as eczema, and/or during peak times for seasonal allergies.
It is helpful when pediatricians do spirometry for a child with a suspicious cough. Nationwide, about 20%-25% of general pediatricians do pulmonary function testing. Pediatric pulmonologists like me would like to see more pediatricians perform these tests. Sinus x-rays also can be helpful, and are within the purview of the general pediatrician. Some might consider this an unnecessary test, however, or one for which you need a high index of suspicion before ordering.
A test that is generally unnecessary is a sweat test for cystic fibrosis. A lot of pediatricians get this test, and I would not tell them not to because often the child with CF has other symptoms that are more diagnostic.
A thorough differential diagnosis, primarily based on history and physical examination, is essential when a child presents with a suspicious cough. Certain imaging modalities are also useful for diagnosis.
Identification of an underlying cause is crucial. When doing your history and physical exam, look for something that does not fit a routine presentation. For example, a cough in the presence of a constitutional change, such as weight loss, can indicate a more serious problem. In addition, a cough with a relatively sudden onset or one associated with labored breathing can be worrisome. Also, a choking episode followed by sudden cough, for example, can indicate the presence of a foreign body.
Asthma is the most common cause of chronic cough in the pediatric population, but also consider less common etiologies such as tracheoesophageal fistula, cystic fibrosis (CF), and bronchopulmonary dysplasia. Failure to thrive, clubbing, cardiac signs, and persistent stridor suggest alternative diagnoses.
Patient age offers some guidance in your differential diagnosis. In a neonate (younger than 28 days), persistent cough might suggest an infection or a congenital anomaly such as compression of the esophagus and trachea by a vascular ring. Infectious etiologies include rhinovirus, adenovirus, respiratory syncytial virus, and pertussis.
In preschool children, think upper or lower respiratory tract infection, rhinitis, postnasal drip syndrome, gastroesophageal reflux, an irritant source (such as passive smoking or air pollution), and, of course, asthma.
Among school-age children and adolescents, consider the same possibilities, but add inhalant or other substance abuse to your list of possible irritant causes. In addition, these older children can develop psychogenic or “habit” cough, one that is absent during sleep, distraction, or periods of concentration. Vocal cord dysfunction, also known as laryngeal wheeze, is another possibility in this group.
General pediatricians commonly treat children with a cough that lasts 5-10 days in the context of an upper respiratory tract illness, such as a cold. If a child still coughs incessantly after other cold symptoms have resolved, I would be concerned. This is not necessarily a call to refer the patient to a specialist, but this scenario is a call to do further diagnostic evaluation.
If the child already is diagnosed with asthma and develops a cough, determine whether the patient is taking the appropriate medication and/or is compliant with therapy. Also, ask about the child's environment, particularly the presence of passive smoking, dust, and pets.
In terms of allergy testing, I recommend a radioallergosorbent allergen-specific IgE antibody assay. This is indicated if a child has other lateral symptoms, such as eczema, and/or during peak times for seasonal allergies.
It is helpful when pediatricians do spirometry for a child with a suspicious cough. Nationwide, about 20%-25% of general pediatricians do pulmonary function testing. Pediatric pulmonologists like me would like to see more pediatricians perform these tests. Sinus x-rays also can be helpful, and are within the purview of the general pediatrician. Some might consider this an unnecessary test, however, or one for which you need a high index of suspicion before ordering.
A test that is generally unnecessary is a sweat test for cystic fibrosis. A lot of pediatricians get this test, and I would not tell them not to because often the child with CF has other symptoms that are more diagnostic.
A thorough differential diagnosis, primarily based on history and physical examination, is essential when a child presents with a suspicious cough. Certain imaging modalities are also useful for diagnosis.
Identification of an underlying cause is crucial. When doing your history and physical exam, look for something that does not fit a routine presentation. For example, a cough in the presence of a constitutional change, such as weight loss, can indicate a more serious problem. In addition, a cough with a relatively sudden onset or one associated with labored breathing can be worrisome. Also, a choking episode followed by sudden cough, for example, can indicate the presence of a foreign body.
Asthma is the most common cause of chronic cough in the pediatric population, but also consider less common etiologies such as tracheoesophageal fistula, cystic fibrosis (CF), and bronchopulmonary dysplasia. Failure to thrive, clubbing, cardiac signs, and persistent stridor suggest alternative diagnoses.
Patient age offers some guidance in your differential diagnosis. In a neonate (younger than 28 days), persistent cough might suggest an infection or a congenital anomaly such as compression of the esophagus and trachea by a vascular ring. Infectious etiologies include rhinovirus, adenovirus, respiratory syncytial virus, and pertussis.
In preschool children, think upper or lower respiratory tract infection, rhinitis, postnasal drip syndrome, gastroesophageal reflux, an irritant source (such as passive smoking or air pollution), and, of course, asthma.
Among school-age children and adolescents, consider the same possibilities, but add inhalant or other substance abuse to your list of possible irritant causes. In addition, these older children can develop psychogenic or “habit” cough, one that is absent during sleep, distraction, or periods of concentration. Vocal cord dysfunction, also known as laryngeal wheeze, is another possibility in this group.
General pediatricians commonly treat children with a cough that lasts 5-10 days in the context of an upper respiratory tract illness, such as a cold. If a child still coughs incessantly after other cold symptoms have resolved, I would be concerned. This is not necessarily a call to refer the patient to a specialist, but this scenario is a call to do further diagnostic evaluation.
If the child already is diagnosed with asthma and develops a cough, determine whether the patient is taking the appropriate medication and/or is compliant with therapy. Also, ask about the child's environment, particularly the presence of passive smoking, dust, and pets.
In terms of allergy testing, I recommend a radioallergosorbent allergen-specific IgE antibody assay. This is indicated if a child has other lateral symptoms, such as eczema, and/or during peak times for seasonal allergies.
It is helpful when pediatricians do spirometry for a child with a suspicious cough. Nationwide, about 20%-25% of general pediatricians do pulmonary function testing. Pediatric pulmonologists like me would like to see more pediatricians perform these tests. Sinus x-rays also can be helpful, and are within the purview of the general pediatrician. Some might consider this an unnecessary test, however, or one for which you need a high index of suspicion before ordering.
A test that is generally unnecessary is a sweat test for cystic fibrosis. A lot of pediatricians get this test, and I would not tell them not to because often the child with CF has other symptoms that are more diagnostic.
Regulated Relationship?
As the American Medical Association (AMA) develops a toolkit to help physicians implement contracts that govern the professional relationship between physicians and non-physician providers (NPPs), one SHM committee member says the trade group should keep an open mind on the pros and cons of such agreements.
The AMA House of Delegates last month tasked its staff to develop the toolkit after several delegates expressed concerns that without the so-called “practice agreements,” NPPs and physicians do not have clear boundaries on the scope of practice responsibilities, according to American Medical News, the society’s newspaper.
Lorraine Britting, MS, NP-C, a member of SHM’s Non-Physician Provider Committee, says the agreements can be a framework for certain practices but might be seen as burdensome to HM groups that have worked for years without the contracts in place.
“In the states that already have practice agreements mandated, you put these regulations in place and they work well. That being said, if new requirements are too restrictive, there is certainly potential for there to be some conflict between the physicians and the nurse practitioners,” says Britting, lead nurse practitioner in the department of cardiology medicine at the Cardiovascular Institute at Beth Israel Deaconess Medical Center in Boston.
Britting notes that many HM groups already have practice agreements in place and that she has never worked without one because Massachusetts requires them. She says professional relationships between doctors and NPPs could be helped by rules on who is responsible for what—but only if those agreements are developed with input from all stakeholders.
“It’s hard to make a blanket statement. Someone who has 10 years’ experience working in hospital medicine versus somebody who just graduated ... their needs are going to be very different,” Britting says. “It has to be individually tailored.”
As the American Medical Association (AMA) develops a toolkit to help physicians implement contracts that govern the professional relationship between physicians and non-physician providers (NPPs), one SHM committee member says the trade group should keep an open mind on the pros and cons of such agreements.
The AMA House of Delegates last month tasked its staff to develop the toolkit after several delegates expressed concerns that without the so-called “practice agreements,” NPPs and physicians do not have clear boundaries on the scope of practice responsibilities, according to American Medical News, the society’s newspaper.
Lorraine Britting, MS, NP-C, a member of SHM’s Non-Physician Provider Committee, says the agreements can be a framework for certain practices but might be seen as burdensome to HM groups that have worked for years without the contracts in place.
“In the states that already have practice agreements mandated, you put these regulations in place and they work well. That being said, if new requirements are too restrictive, there is certainly potential for there to be some conflict between the physicians and the nurse practitioners,” says Britting, lead nurse practitioner in the department of cardiology medicine at the Cardiovascular Institute at Beth Israel Deaconess Medical Center in Boston.
Britting notes that many HM groups already have practice agreements in place and that she has never worked without one because Massachusetts requires them. She says professional relationships between doctors and NPPs could be helped by rules on who is responsible for what—but only if those agreements are developed with input from all stakeholders.
“It’s hard to make a blanket statement. Someone who has 10 years’ experience working in hospital medicine versus somebody who just graduated ... their needs are going to be very different,” Britting says. “It has to be individually tailored.”
As the American Medical Association (AMA) develops a toolkit to help physicians implement contracts that govern the professional relationship between physicians and non-physician providers (NPPs), one SHM committee member says the trade group should keep an open mind on the pros and cons of such agreements.
The AMA House of Delegates last month tasked its staff to develop the toolkit after several delegates expressed concerns that without the so-called “practice agreements,” NPPs and physicians do not have clear boundaries on the scope of practice responsibilities, according to American Medical News, the society’s newspaper.
Lorraine Britting, MS, NP-C, a member of SHM’s Non-Physician Provider Committee, says the agreements can be a framework for certain practices but might be seen as burdensome to HM groups that have worked for years without the contracts in place.
“In the states that already have practice agreements mandated, you put these regulations in place and they work well. That being said, if new requirements are too restrictive, there is certainly potential for there to be some conflict between the physicians and the nurse practitioners,” says Britting, lead nurse practitioner in the department of cardiology medicine at the Cardiovascular Institute at Beth Israel Deaconess Medical Center in Boston.
Britting notes that many HM groups already have practice agreements in place and that she has never worked without one because Massachusetts requires them. She says professional relationships between doctors and NPPs could be helped by rules on who is responsible for what—but only if those agreements are developed with input from all stakeholders.
“It’s hard to make a blanket statement. Someone who has 10 years’ experience working in hospital medicine versus somebody who just graduated ... their needs are going to be very different,” Britting says. “It has to be individually tailored.”
Development Phase
More than 350 pediatric hospitalists convened in Tampa, Fla., last week for Pediatric Hospital Medicine (PHM) 2009, tri-sponsored by SHM, the Academic Pediatric Association (APA), and the American Academy of Pediatrics (AAP). More impressive than the continued growth of the field, however, was the palpable theme of development and maturation.
The theme was immediately evident as Patrick Conway, MD, MSc, took the stage to give the keynote address. Dr. Conway, a card-carrying pediatric hospitalist, has built upon his beginnings in health services research and a White House fellowship to become the chief medical officer in the Department of Health and Human Services (HHS) Office of the Secretary/Assistant Secretary for Planning and Evaluation. He also is the executive director of the Federal Coordinating Council for Comparative Effectiveness Research.
After providing an insider’s view of HHS, comparative effectiveness research, and healthcare reform and policy, he challenged pediatric hospitalists to demonstrate their value to the healthcare system.
Synergistically, this year’s meeting also provided an opportunity for the PHM Roundtable, a strategic planning session of pediatric hospitalist leaders, to fully share its vision for transforming the delivery of hospital care to children. As a manifestation of this vision, collaborative discussions and workgroup plans coalesced amid the enlightening mix of clinical, practice management, academic, and quality and patient safety workshops.
Growth and development are central concepts in pediatrics, and PHM 2009 highlighted the field’s energetic steps towards maturation.
Dr. Shen is pediatric editor of The Hospitalist and medical director of hospital medicine at Dell Children’s Medical Center in Austin, Texas
More than 350 pediatric hospitalists convened in Tampa, Fla., last week for Pediatric Hospital Medicine (PHM) 2009, tri-sponsored by SHM, the Academic Pediatric Association (APA), and the American Academy of Pediatrics (AAP). More impressive than the continued growth of the field, however, was the palpable theme of development and maturation.
The theme was immediately evident as Patrick Conway, MD, MSc, took the stage to give the keynote address. Dr. Conway, a card-carrying pediatric hospitalist, has built upon his beginnings in health services research and a White House fellowship to become the chief medical officer in the Department of Health and Human Services (HHS) Office of the Secretary/Assistant Secretary for Planning and Evaluation. He also is the executive director of the Federal Coordinating Council for Comparative Effectiveness Research.
After providing an insider’s view of HHS, comparative effectiveness research, and healthcare reform and policy, he challenged pediatric hospitalists to demonstrate their value to the healthcare system.
Synergistically, this year’s meeting also provided an opportunity for the PHM Roundtable, a strategic planning session of pediatric hospitalist leaders, to fully share its vision for transforming the delivery of hospital care to children. As a manifestation of this vision, collaborative discussions and workgroup plans coalesced amid the enlightening mix of clinical, practice management, academic, and quality and patient safety workshops.
Growth and development are central concepts in pediatrics, and PHM 2009 highlighted the field’s energetic steps towards maturation.
Dr. Shen is pediatric editor of The Hospitalist and medical director of hospital medicine at Dell Children’s Medical Center in Austin, Texas
More than 350 pediatric hospitalists convened in Tampa, Fla., last week for Pediatric Hospital Medicine (PHM) 2009, tri-sponsored by SHM, the Academic Pediatric Association (APA), and the American Academy of Pediatrics (AAP). More impressive than the continued growth of the field, however, was the palpable theme of development and maturation.
The theme was immediately evident as Patrick Conway, MD, MSc, took the stage to give the keynote address. Dr. Conway, a card-carrying pediatric hospitalist, has built upon his beginnings in health services research and a White House fellowship to become the chief medical officer in the Department of Health and Human Services (HHS) Office of the Secretary/Assistant Secretary for Planning and Evaluation. He also is the executive director of the Federal Coordinating Council for Comparative Effectiveness Research.
After providing an insider’s view of HHS, comparative effectiveness research, and healthcare reform and policy, he challenged pediatric hospitalists to demonstrate their value to the healthcare system.
Synergistically, this year’s meeting also provided an opportunity for the PHM Roundtable, a strategic planning session of pediatric hospitalist leaders, to fully share its vision for transforming the delivery of hospital care to children. As a manifestation of this vision, collaborative discussions and workgroup plans coalesced amid the enlightening mix of clinical, practice management, academic, and quality and patient safety workshops.
Growth and development are central concepts in pediatrics, and PHM 2009 highlighted the field’s energetic steps towards maturation.
Dr. Shen is pediatric editor of The Hospitalist and medical director of hospital medicine at Dell Children’s Medical Center in Austin, Texas
Docs recommend transdermal patch for HRT
Boston—Postmenopausal women on hormone replacement therapy (HRT) are known to be at increased risk of venous thromboembolism (VTE). But whether the route of administration influences the risk was not known until now.
Investigators at Kings College Hospital NHS Foundation Trust in London discovered that VTE risk is increased in women using oral HRT but not in those using a transdermal patch. Catherine N. Bagot, MD, reported these results at the 22nd Congress of the International Society on Thrombosis and Haemostasis (ISTH).
The investigators have thus far recruited 155 women to this ongoing study, 98 on HRT and 57 controls not on HRT. Fifty-four women were on oral HRT and 44 were using the transdermal patch.
Dr Bagot and colleagues used thrombin generation as a marker of thrombotic risk. They found that women taking HRT had significantly higher peak thrombin generation than controls (P=0.0019). They performed a subgroup analysis and the difference was only detectable in women using oral HRT (P<0.0001) and not in women using the transdermal route (P=0.7).
Investigators verified the data further and confirmed that peak thrombin generation was significantly higher in women on oral compared to transdermal HRT (P<0.0001).
The presence of progestogen or testosterone did not have any impact on the results.
These findings indicate that postmenopausal women taking oral HRT are at greater risk for VTE than those using transdermal administration. Dr Bagot suggested that transdermal administration may be safe in women who have had previous VTEs.
The research team also investigated the relationship between estradiol and peak thrombin generation.
They analyzed blood samples of 132 women. Eighty-six women were on HRT, 42 oral and 44 transdermal. The remaining women not on HRT served as controls.
Investigators excluded women whose HRT formulation contained equine estrogens or who had less than 100 pMol/L estradiol levels.
They found that estradiol levels were significantly higher in women using either formulation of HRT than controls. However, the levels were not significantly different between the two HRT groups.
Investigators also found peak thrombin generation to be significantly higher in women on HRT than controls, and this correlated with estradiol concentrations. Women taking oral HRT had a significantly higher peak thrombin generation compared to women using the patch. Investigators observed a correlation between estradiol levels and peak thrombin generation only in women using oral HRT.
Dr Bagot indicated that a limitation of this second study was that there was no way of knowing whether the estradiol levels were at the peak or trough. Nevertheless, this study confirmed further a causal link between oral estrogens, hypercoagulability, and an increased risk of VTE.
The investigators again recommended the transdermal route over oral HRT administration in postmenopausal women to achieve the lowest thrombotic risk. ![]()
Boston—Postmenopausal women on hormone replacement therapy (HRT) are known to be at increased risk of venous thromboembolism (VTE). But whether the route of administration influences the risk was not known until now.
Investigators at Kings College Hospital NHS Foundation Trust in London discovered that VTE risk is increased in women using oral HRT but not in those using a transdermal patch. Catherine N. Bagot, MD, reported these results at the 22nd Congress of the International Society on Thrombosis and Haemostasis (ISTH).
The investigators have thus far recruited 155 women to this ongoing study, 98 on HRT and 57 controls not on HRT. Fifty-four women were on oral HRT and 44 were using the transdermal patch.
Dr Bagot and colleagues used thrombin generation as a marker of thrombotic risk. They found that women taking HRT had significantly higher peak thrombin generation than controls (P=0.0019). They performed a subgroup analysis and the difference was only detectable in women using oral HRT (P<0.0001) and not in women using the transdermal route (P=0.7).
Investigators verified the data further and confirmed that peak thrombin generation was significantly higher in women on oral compared to transdermal HRT (P<0.0001).
The presence of progestogen or testosterone did not have any impact on the results.
These findings indicate that postmenopausal women taking oral HRT are at greater risk for VTE than those using transdermal administration. Dr Bagot suggested that transdermal administration may be safe in women who have had previous VTEs.
The research team also investigated the relationship between estradiol and peak thrombin generation.
They analyzed blood samples of 132 women. Eighty-six women were on HRT, 42 oral and 44 transdermal. The remaining women not on HRT served as controls.
Investigators excluded women whose HRT formulation contained equine estrogens or who had less than 100 pMol/L estradiol levels.
They found that estradiol levels were significantly higher in women using either formulation of HRT than controls. However, the levels were not significantly different between the two HRT groups.
Investigators also found peak thrombin generation to be significantly higher in women on HRT than controls, and this correlated with estradiol concentrations. Women taking oral HRT had a significantly higher peak thrombin generation compared to women using the patch. Investigators observed a correlation between estradiol levels and peak thrombin generation only in women using oral HRT.
Dr Bagot indicated that a limitation of this second study was that there was no way of knowing whether the estradiol levels were at the peak or trough. Nevertheless, this study confirmed further a causal link between oral estrogens, hypercoagulability, and an increased risk of VTE.
The investigators again recommended the transdermal route over oral HRT administration in postmenopausal women to achieve the lowest thrombotic risk. ![]()
Boston—Postmenopausal women on hormone replacement therapy (HRT) are known to be at increased risk of venous thromboembolism (VTE). But whether the route of administration influences the risk was not known until now.
Investigators at Kings College Hospital NHS Foundation Trust in London discovered that VTE risk is increased in women using oral HRT but not in those using a transdermal patch. Catherine N. Bagot, MD, reported these results at the 22nd Congress of the International Society on Thrombosis and Haemostasis (ISTH).
The investigators have thus far recruited 155 women to this ongoing study, 98 on HRT and 57 controls not on HRT. Fifty-four women were on oral HRT and 44 were using the transdermal patch.
Dr Bagot and colleagues used thrombin generation as a marker of thrombotic risk. They found that women taking HRT had significantly higher peak thrombin generation than controls (P=0.0019). They performed a subgroup analysis and the difference was only detectable in women using oral HRT (P<0.0001) and not in women using the transdermal route (P=0.7).
Investigators verified the data further and confirmed that peak thrombin generation was significantly higher in women on oral compared to transdermal HRT (P<0.0001).
The presence of progestogen or testosterone did not have any impact on the results.
These findings indicate that postmenopausal women taking oral HRT are at greater risk for VTE than those using transdermal administration. Dr Bagot suggested that transdermal administration may be safe in women who have had previous VTEs.
The research team also investigated the relationship between estradiol and peak thrombin generation.
They analyzed blood samples of 132 women. Eighty-six women were on HRT, 42 oral and 44 transdermal. The remaining women not on HRT served as controls.
Investigators excluded women whose HRT formulation contained equine estrogens or who had less than 100 pMol/L estradiol levels.
They found that estradiol levels were significantly higher in women using either formulation of HRT than controls. However, the levels were not significantly different between the two HRT groups.
Investigators also found peak thrombin generation to be significantly higher in women on HRT than controls, and this correlated with estradiol concentrations. Women taking oral HRT had a significantly higher peak thrombin generation compared to women using the patch. Investigators observed a correlation between estradiol levels and peak thrombin generation only in women using oral HRT.
Dr Bagot indicated that a limitation of this second study was that there was no way of knowing whether the estradiol levels were at the peak or trough. Nevertheless, this study confirmed further a causal link between oral estrogens, hypercoagulability, and an increased risk of VTE.
The investigators again recommended the transdermal route over oral HRT administration in postmenopausal women to achieve the lowest thrombotic risk. ![]()
Caution Urged in Interpreting Glargine Cancer Risk
A series of studies reported last month by a European health organization has questioned whether the use of insulin glargine, known commercially as Lantus, inflates a patient’s risk of cancer. But according to one source, hospitalists with a large census of diabetic or hypoglycemic patients shouldn’t pull their patients off the treatment just yet.
Four different population-based studies were reported on the Web site for Diabetologia, the journal of the European Association of the Study of Diabetes. A German study of 127,000 patients in an insurance database found that for every 100 patients taking Lantus, there was one more person diagnosed with cancer when compared with 100 patients taking similar doses of human insulin. The risk increased with the dosage, the study reported.
But the American Diabetes Association quickly released a statement calling the studies “conflicting and confusing.”
Hospitalist Jeffrey Schnipper, MD, MPH, FHM, who estimates one-third of his patients are either diabetic or hypoglycemic, agrees, saying that observational studies make it hard to draw any conclusions.
“I tell patients we don’t know enough,” says Dr. Schnipper, director of clinical research and associate physician in the Division of General Medicine at Brigham and Women’s Hospital Hospitalist Service in Boston. “Right now, there’s no good strong evidence that Lantus is worse than any other alternative.”
Dr. Schnipper cautioned fellow hospitalists to not overreact to the reports, noting that without a randomized trial to follow through on the hypotheses raised, there is no resolution to the confounding-by-indication bias that can plague observational studies.
“There’s an adage in medicine that you never want to be the first person or the last person to use a drug,” Dr. Schnipper says. “I would say you should never be the first person or the last person to stop using a drug.”
A series of studies reported last month by a European health organization has questioned whether the use of insulin glargine, known commercially as Lantus, inflates a patient’s risk of cancer. But according to one source, hospitalists with a large census of diabetic or hypoglycemic patients shouldn’t pull their patients off the treatment just yet.
Four different population-based studies were reported on the Web site for Diabetologia, the journal of the European Association of the Study of Diabetes. A German study of 127,000 patients in an insurance database found that for every 100 patients taking Lantus, there was one more person diagnosed with cancer when compared with 100 patients taking similar doses of human insulin. The risk increased with the dosage, the study reported.
But the American Diabetes Association quickly released a statement calling the studies “conflicting and confusing.”
Hospitalist Jeffrey Schnipper, MD, MPH, FHM, who estimates one-third of his patients are either diabetic or hypoglycemic, agrees, saying that observational studies make it hard to draw any conclusions.
“I tell patients we don’t know enough,” says Dr. Schnipper, director of clinical research and associate physician in the Division of General Medicine at Brigham and Women’s Hospital Hospitalist Service in Boston. “Right now, there’s no good strong evidence that Lantus is worse than any other alternative.”
Dr. Schnipper cautioned fellow hospitalists to not overreact to the reports, noting that without a randomized trial to follow through on the hypotheses raised, there is no resolution to the confounding-by-indication bias that can plague observational studies.
“There’s an adage in medicine that you never want to be the first person or the last person to use a drug,” Dr. Schnipper says. “I would say you should never be the first person or the last person to stop using a drug.”
A series of studies reported last month by a European health organization has questioned whether the use of insulin glargine, known commercially as Lantus, inflates a patient’s risk of cancer. But according to one source, hospitalists with a large census of diabetic or hypoglycemic patients shouldn’t pull their patients off the treatment just yet.
Four different population-based studies were reported on the Web site for Diabetologia, the journal of the European Association of the Study of Diabetes. A German study of 127,000 patients in an insurance database found that for every 100 patients taking Lantus, there was one more person diagnosed with cancer when compared with 100 patients taking similar doses of human insulin. The risk increased with the dosage, the study reported.
But the American Diabetes Association quickly released a statement calling the studies “conflicting and confusing.”
Hospitalist Jeffrey Schnipper, MD, MPH, FHM, who estimates one-third of his patients are either diabetic or hypoglycemic, agrees, saying that observational studies make it hard to draw any conclusions.
“I tell patients we don’t know enough,” says Dr. Schnipper, director of clinical research and associate physician in the Division of General Medicine at Brigham and Women’s Hospital Hospitalist Service in Boston. “Right now, there’s no good strong evidence that Lantus is worse than any other alternative.”
Dr. Schnipper cautioned fellow hospitalists to not overreact to the reports, noting that without a randomized trial to follow through on the hypotheses raised, there is no resolution to the confounding-by-indication bias that can plague observational studies.
“There’s an adage in medicine that you never want to be the first person or the last person to use a drug,” Dr. Schnipper says. “I would say you should never be the first person or the last person to stop using a drug.”
JHM Names Thomas Baudendistel, MD, New CME Editor
Some physicians have an interest in teaching; some are really good at it, and some make a career out of it. For Thomas Baudendistel, MD, teaching comes second nature and, as one of his former colleagues says, is a definition of who he is.
It’s those qualities, his experience in both academic and community hospital settings, and a passion for innovation that Dr. Baudendistel, the internal-medicine residency program director for Kaiser Permanente in Oakland, Calif., hopes to infuse as CME editor of the Journal of Hospital Medicine (JHM). He was appointed to the new position in June; the first issue containing article-level CME, the answers to which will be submitted online, is scheduled to appear in October.
“Tom is a superlative educator. He has defined himself that way. He has a passion for it and a talent for it,” says Brian J. Harte, MD, FHM, chair of the department of hospital medicine at The Cleveland Clinic and a deputy editor of JHM.
Dr. Harte, who first met Dr. Baudendistel in 1996 during his residency at the University of California at San Francisco, says his former mentor “can take a submission, drill down to the most important teaching point, and challenge the readership.”
Dr. Baudendistel, who admits teaching is why he “gets out of bed in the morning,” says he wants to take advantage of the young, tech-savvy nature of most HM physicians. “JHM has been an innovative journal. I see the CME piece as being equally innovative,” he says. “I’d like to move [CME] past the pencil-and-paper phase.”
Some physicians have an interest in teaching; some are really good at it, and some make a career out of it. For Thomas Baudendistel, MD, teaching comes second nature and, as one of his former colleagues says, is a definition of who he is.
It’s those qualities, his experience in both academic and community hospital settings, and a passion for innovation that Dr. Baudendistel, the internal-medicine residency program director for Kaiser Permanente in Oakland, Calif., hopes to infuse as CME editor of the Journal of Hospital Medicine (JHM). He was appointed to the new position in June; the first issue containing article-level CME, the answers to which will be submitted online, is scheduled to appear in October.
“Tom is a superlative educator. He has defined himself that way. He has a passion for it and a talent for it,” says Brian J. Harte, MD, FHM, chair of the department of hospital medicine at The Cleveland Clinic and a deputy editor of JHM.
Dr. Harte, who first met Dr. Baudendistel in 1996 during his residency at the University of California at San Francisco, says his former mentor “can take a submission, drill down to the most important teaching point, and challenge the readership.”
Dr. Baudendistel, who admits teaching is why he “gets out of bed in the morning,” says he wants to take advantage of the young, tech-savvy nature of most HM physicians. “JHM has been an innovative journal. I see the CME piece as being equally innovative,” he says. “I’d like to move [CME] past the pencil-and-paper phase.”
Some physicians have an interest in teaching; some are really good at it, and some make a career out of it. For Thomas Baudendistel, MD, teaching comes second nature and, as one of his former colleagues says, is a definition of who he is.
It’s those qualities, his experience in both academic and community hospital settings, and a passion for innovation that Dr. Baudendistel, the internal-medicine residency program director for Kaiser Permanente in Oakland, Calif., hopes to infuse as CME editor of the Journal of Hospital Medicine (JHM). He was appointed to the new position in June; the first issue containing article-level CME, the answers to which will be submitted online, is scheduled to appear in October.
“Tom is a superlative educator. He has defined himself that way. He has a passion for it and a talent for it,” says Brian J. Harte, MD, FHM, chair of the department of hospital medicine at The Cleveland Clinic and a deputy editor of JHM.
Dr. Harte, who first met Dr. Baudendistel in 1996 during his residency at the University of California at San Francisco, says his former mentor “can take a submission, drill down to the most important teaching point, and challenge the readership.”
Dr. Baudendistel, who admits teaching is why he “gets out of bed in the morning,” says he wants to take advantage of the young, tech-savvy nature of most HM physicians. “JHM has been an innovative journal. I see the CME piece as being equally innovative,” he says. “I’d like to move [CME] past the pencil-and-paper phase.”
Perceptions of Hospital Discharge Software
During the transition from inpatient to outpatient care, patients are vulnerable to adverse events.1 Poor communication between hospital personnel and either the patient or the outpatient primary care physician has been associated with preventable or ameliorable adverse events after discharge.1 Systematic reviews confirm that discharge communication is often delayed, inaccurate, or ineffective.2, 3
Discharge communication failures may occur if hospital processes rely on dictated discharge summaries.2 For several reasons, discharge summaries are inadequate for communication. Most patients complete their initial posthospital clinic visit before their primary care physician receives the discharge summary.4 For many patients, the discharge summary is unavailable for all posthospital visits.4 Discharge summaries often fail as communication because they are not generated or transmitted.4
Recommendations to improve discharge communication include the use of health information technology.2, 5 The benefits of computer‐generated discharge summaries include decreases in delivery time for discharge communications.2 The benefits of computerized physician order entry (CPOE) include reduction of medical errors.6 These theoretical benefits create a rationale for clinical trials to measure improvements after discharge software applications with CPOE.5
In an effort to improve discharge communication and clinically relevant outcomes, we performed a cluster‐randomized trial to assess the value of a discharge software application of CPOE. The clustered design followed recommendations from a systematic review of discharge interventions.3 We applied our research intervention at the physician level and measured outcomes at the patient level. Our objective was to assess the benefit of discharge software with CPOE vs. usual care when used to discharge patients at high risk for repeat admission. In a previous work, we reported that discharge software did not reduce rates of hospital readmission, emergency department visits, or postdischarge adverse events due to medical management.7 In the present article, we compare secondary outcomes after the research intervention: perceptions of the discharge from the perspectives of patients, primary care physicians, and hospital physicians.
Methods
The trial design was a cluster randomized, controlled trial. The setting was the postdischarge environment following index hospitalization at a 730‐bed, tertiary care, teaching hospital in central Illinois. The Peoria Institutional Review Board approved the protocol for human research.
Participants
We enrolled consenting hospital physicians and their patients between November 2004 and January 2007. The hospital physician defined the cluster. Patients discharged by the physician comprised the cluster. The eligibility criteria for hospital physicians required internal medicine resident or attending physicians with assignments to inpatient duties for at least 2 months during the 27‐month enrollment period. After achieving informed consent from physicians, research personnel screened all consecutive, adult inpatients who were discharged to home. Patient inclusion required a probability of repeat admission (Pra) equal to or greater than 0.40.8, 9 The purpose of the inclusion criterion was to enrich the sample with patients likely to benefit from interventions to improve discharge processes. Furthermore, hospital readmission was the primary endpoint of the study, as reported separately.7 The Pra came from a predictive model with scores for age, gender, prior hospitalizations, prior doctor visits, self‐rated health status, presence of informal caregiver in the home, and comorbid coronary heart disease and diabetes mellitus. Research coordinators calculated the Pra within 2 days before discharge from the index hospitalization.
Exclusion Criteria
We excluded patients previously enrolled in the study, candidates for hospice, and patients unable to participate in outcome ascertainment. Cognitive impairment was a conditional exclusion criterion for patients. We defined cognitive impairment as a score less than 9 on the 10‐point clock test.10 Patients with cognitive impairment participated only with consent from their legally authorized representative. We enrolled patients with cognitive impairment only if a proxy spent at least 3 hours daily with the patient and the proxy agreed to answer postdischarge interviews. If a patient's outpatient primary care physician treated the patient during the index hospitalization, then there was no perceived barrier in physician‐to‐physician communication and we excluded the patient.
Intervention
The research intervention was discharge software with CPOE. Detailed description of the software appeared previously.5 In summary, the CPOE software application facilitated communication at the time of hospital discharge to patients, retail pharmacists, and community physicians. The application had basic levels of clinical decision support, required fields, pick lists, standard drug doses, alerts, reminders, and online reference information. The software addressed deficiencies in the usual care discharge process reported globally and reviewed previously.5 For example, 1 deficiency occurred when inpatient physicians failed to warn outpatient physicians about diagnostic tests with results pending at discharge.11 Another deficiency was discharge medication error.12 The software prompted the discharging physician to enter pending tests, order tests after discharge, and perform medication reconciliation. On the day of discharge, hospital physicians used the software to automatically generate discharge documents and reconcile prescriptions for the patient, primary care physician, retail pharmacist, and the ward nurse. The discharge letter went to the outpatient practitioner via facsimile transmission plus a duplicate via U.S. mail.
The control intervention was the usual care, handwritten discharge process commonly used by hospitalists.2 Hospital physicians and ward nurses completed handwritten discharge forms on the day of discharge. The forms contained blanks for discharge diagnoses, discharge medications, medication instructions, postdischarge activities and restrictions, postdischarge diet, postdischarge diagnostic and therapeutic interventions, and appointments. Patients received handwritten copies of the forms, 1 page of which also included medication instructions and prescriptions. In a previous publication, we provided details about the usual care discharge process as well as the standard care available to all study patients regardless of intervention.5
Randomization
The hospital physician who completed the discharge process was the unit of randomization. Random allocation was to discharge software or usual care discharge process, with a randomization ratio of 1:1 and block size of 2. We concealed allocation with the following process. An investigator who was not involved with hospital physician recruitment generated the randomization sequence with a computerized random number generator. The randomization list was maintained in a secure location. Another investigator who was unaware of the next random assignment performed the hospital physician recruitment and informed consent. After confirming eligibility and obtaining informed consent from physicians, the blinded investigator requested the next random assignment from the custodian of the randomization list. Hospital physicians subsequently used their randomly assigned process when discharging their patients who enrolled in the study. After random allocation, it was not possible to conceal the test or control intervention from physicians or their patients.
Hospital physicians underwent training on the software or usual care discharge process; the details appeared previously.7 Physicians assigned to usual care did not receive training on the discharge software and were blocked from using the software. Patients were passive recipients of the research intervention performed by their discharging physician. Patients received the research intervention on the day of discharge of the index hospitalization.
Baseline Assessment
During the index hospitalization, trained data abstractors recorded baseline patient demographic data plus variables to calculate the Pra for probability for repeat admission. We recorded the availability of an informal caregiver in response to the question, Is there a friend, relative, or neighbor who would take care of you for a few days, if necessary? Data came from the patient or proxy for physical functioning, mental health,13 heart failure, and number of previous emergency department visits. Other data came from hospital records for chronic obstructive pulmonary disease, number of discharge medications, and length of stay for the index hospitalization.
Outcome Assessment
We assessed the patient's perception of the discharge with 2 validated survey instruments. One week after discharge, research personnel performed telephone interviews with patients or proxies. While following a script, interviewers instructed patients to avoid mentioning the discharge process. Interviewers read items from the B‐PREPARED questionnaire.14, 15 and the Satisfaction with Information About Medicines Scale (SIMS).16 The B‐PREPARED scale assessed 3 principal components of patient preparedness for discharge: self‐care information for medications and activities, equipment and services, and confidence. The scale demonstrated internal consistency, construct validity, and predictive validity. High scale values reflected high perceptions of discharge preparedness from the patient perspective.15 SIMS measured patient satisfaction with information about discharge medications. Validation studies revealed SIMS had internal consistency, test‐retest reliability, and criterion‐related validity.16 Interviewers recorded responses to calculate a total SIMS score. Patients with high total SIMS scores had high satisfaction. While assessing B‐PREPARED and SIMS, interviewers were blind to intervention assignment. We evaluated the adequacy of blinding by asking interviewers to guess the patient's intervention assignment.
We measured the quality of hospital discharge from the outpatient physician perspective. During the index hospitalization, patients designated an outpatient primary care practitioner to receive discharge reports and results of diagnostic tests. Ten days after discharge, research personnel mailed the Physician‐PREPARED questionnaire to the designated community practitioner.17 The sum of item responses comprised the Modified Physician‐PREPARED scale and demonstrated internal consistency and construct validity. The principal components of the Modified Physician‐PREPARED were timeliness of communication and adequacy of discharge plan/transmission. High scale values reflected high perceptions of discharge quality.17 Outpatient practitioners gave implied consent when they completed and returned questionnaires. We requested 1 questionnaire for each enrolled patient, so the outcome assessment was at the patient level. The assessment was not blinded because primary care physicians received the output of discharge software or usual care discharge.
We assessed the satisfaction of hospital physicians who used the discharge software and the usual care. After hospital physicians participated in the trial for 6 months, they rated their assigned discharge process on Likert scales. The first question was, On a scale of 1 to 10, indicate your satisfaction with your portion of the discharge process. The scale anchors were 1 for very dissatisfied and 10 for very satisfied. The second question was, On a scale of 1 to 10, indicate the effort to complete your portion of the discharge process. For the second question, the scale anchors were 1 for very difficult and 10 for very easy. It was not possible to mask the hospital physicians after they received their intervention assignment. Consequently, their outcome assessment was not blinded.
Statistical Methods
The cluster number and size were selected to maintain test significance level, 1‐sided alpha less than 0.05, and power greater than 80%. We previously published the assumptions and rationale for 35 hospital physician clusters per intervention and 9 patients per cluster.7 We did not perform separate sample size estimates for the secondary outcomes reported herein.
The statistical analyses employed SPSS PC (Version 15.0.1; SPSS, Inc., Chicago, IL). Statistical procedures for baseline variables were descriptive and included means and standard deviations (SDs) for interval variables and percentages for categorical variables. For all analyses, we employed the principle of intention‐to‐treat. We assumed patient or physician exposure to the intervention randomly assigned to the discharging physician. Analyses employed standard tests for normal distribution, homogeneity of variance, and linearity of relationships between independent and dependent variables. If assumptions failed, then we stratified variables or performed transformations. We accepted P < 0.05 as significant.
We tested hypotheses for patient‐level outcomes with generalized estimating equations (GEEs) that corrected for clustering by hospital physician. We employed GEEs because they provide unbiased estimates of standard errors for parameters even with incorrect specification of the intracluster dependence structure.18 Each patient‐level outcome was the dependent variable in a separate GEE. The intervention variable for each GEE was discharge software vs. usual care, handwritten discharge. The statistic of interest was the coefficient for the intervention variable. The null hypothesis was no difference between discharge software and usual care. The statistical definition of the null hypothesis was an intervention variable coefficient with a 95% confidence interval (CI) that included 0.
For analyses that were unaffected by the cluster assumption, we performed standard tests. The hypothesis for hospital physicians was significantly higher satisfaction for discharge software users and the inferential procedure was the t test. When we assessed the success of the study blinding, we assumed no association between true intervention allocation and guesses by outcome assessors. We used chi‐square for assessment of the blinding.
Results
We screened 127 physicians who were general internal medicine hospital physicians. Seventy physicians consented and received random allocation to discharge software or usual care. We excluded 57 physicians for reasons shown in the trial flow diagram (Figure 1). We approached 6884 patients during their index hospitalization. After excluding 6253 ineligible patients, we enrolled 631 willing patients (Supplementary Appendix). As depicted in Figure 1, the most common reason for ineligibility occurred for patients with Pra score <0.40 (2168/6253 exclusions; 34.7%). We followed 631 patients who received the discharge intervention (Figure 1). There was no differential dropout between the interventions. Protocol deviations were rare, 0.5% (3/631). Three patients erroneously received usual care discharge from physicians assigned to discharge software. All 631 patients were included in the intention‐to‐treat analysis. The baseline characteristics of the randomly assigned hospital physicians and their patients are in Table 1. Most of the hospital physicians were residents in the first year of postgraduate training.
| Discharge Software | Usual Care | |
|---|---|---|
| ||
| Hospital physician characteristics, n (%) | n = 35 | n = 35 |
| Postgraduate year 1 | 18 (51.4) | 23 (65.7) |
| Postgraduate years 2‐4 | 10 (28.6) | 7 (20.0) |
| Attending physician | 7 (20.0) | 5 (14.3) |
| Patient characteristics, n (%) | n = 316 | n = 315 |
| Gender, male | 136 (43.0) | 147 (46.7) |
| Age, years | ||
| 18‐44 | 68 (21.5) | 95 (30.2) |
| 45‐54 | 79 (25.0) | 76 (24.1) |
| 55‐64 | 86 (27.2) | 74 (23.5) |
| 65‐98 | 83 (26.3) | 70 (22.2) |
| Self‐rated health status | ||
| Poor | 82 (25.9) | 108 (34.3) |
| Fair | 169 (53.5) | 147 (46.7) |
| Good | 54 (17.1) | 46 (14.6) |
| Very good | 10 (3.2) | 11 (3.5) |
| Excellent | 1 (0.3) | 3 (1.0) |
| Diabetes mellitus | 172 (54.4) | 177 (56.2) |
| Chronic obstructive pulmonary disease | ||
| None | 259 (82.0) | 257 (81.6) |
| Without oral steroid or home oxygen | 28 (8.9) | 26 (8.3) |
| With chronic oral steroid | 10 (3.2) | 8 (2.5) |
| With home oxygen oral steroid | 19 (6.0) | 24 (7.6) |
| Coronary heart disease | 133 (42.1) | 120 (38.1) |
| Heart failure | 80 (25.3) | 67 (21.3) |
| Physical Functioning from SF‐36 | ||
| Lowest third | 128 (40.5) | 121 (38.4) |
| Upper two‐thirds | 188 (59.5) | 194 (61.6) |
| Mental Health from SF‐36 | ||
| Lowest one‐third | 113 (35.8) | 117 (37.1)* |
| Upper two‐thirds | 203 (64.2) | 197 (62.5)* |
| Emergency department visits during 6 months before index admission | ||
| 0 or 1 | 194 (61.4) | 168 (53.3) |
| 2 or more | 122 (38.6) | 147 (46.7) |
| Mean (SD) | ||
| Number of discharge medications | 10.5 (4.8) | 9.9 (5.1) |
| Index hospital length of stay, days | 3.9 (3.5) | 3.5 (3.5) |
| Pra | 0.486 (0.072) | 0.495 (0.076) |
We assessed the patient's perception of discharge preparedness. One week after discharge, research personnel interviewed 92.4% (292/316) of patients in the discharge software group and 92.4% (291/315) in the usual care group. The mean (SD) B‐PREPARED scores for discharge preparedness were 17.7 (4.1) in the discharge software group and 17.2 (4.0) in the usual care group. In the generalized estimating equation that accounted for potential clustering within hospital physicians, the parameter estimate for the intervention variable coefficient was small but significant (P = 0.042; Table 2). Patients in the discharge software group had slightly better perceptions of their discharge preparedness.
| Outcome Variable | Discharge Software [mean (SD)] | Usual Care [mean (SD)] | Parameter Estimate Without Cluster Correction (95% CI) | P Value | Parameter Estimate with Cluster Correction (95% CI) | P Value |
|---|---|---|---|---|---|---|
| ||||||
| Patient perception of discharge preparedness (B‐PREPARED) | 17.7 (4.1) | 17.2 (4.0) | 0.147* (0.006‐0.288) | 0.040 | 0.147* (0.005‐0.289) | 0.042 |
| Patient satisfaction with medication information score (SIMS) | 12.3 (4.8) | 12.1 (4.6) | 0.212 (0.978‐0.554) | 0.587 | 0.212 (0.937‐0.513) | 0.567 |
| Outpatient physician perception (Modified Physician‐PREPARED) | 17.2 (3.8) | 16.5 (3.9) | 0.133 (0.012‐0.254) | 0.031 | 0.133 (0.015‐0.251) | 0.027 |
Another outcome was the patient's satisfaction with information about discharge medications (Table 2). One week after discharge, mean (SD) SIMS scores for satisfaction were 12.3 (4.8) in the discharge software group and 12.1 (4.6) in the usual care group. The generalized estimating equation revealed an insignificant coefficient for the intervention variable (P = 0.567; Table 2).
We assessed the outpatient physician perception of the discharge with a questionnaire sent 10 days after discharge. We received 496 out of 631 questionnaires (78.6%) from outpatient practitioners and the median response time was 19 days after the date of discharge. The practitioner specialty was internal medicine for 38.9% (193/496), family medicine for 27.2% (135/496), medicine‐pediatrics for 27.0% (134/496), advance practice nurse for 4.4% (22/496), other physician specialties for 2.0% (10/496), and physician assistant for 0.4% (2/496). We excluded 18 questionnaires from analysis because outpatient practitioners failed to answer 2 or more items in the Modified Physician‐PREPARED scale. When we compared baseline characteristics for patients who had complete questionnaires vs. patients with nonrespondent or excluded questionnaires, we found no significant differences (data available upon request). Among the discharge software group, 72.2% (228/316) of patients had complete questionnaires from their outpatient physicians. The response rate with complete questionnaires was 79.4% (250/315) of patients assigned to usual care. On the Modified Physician‐PREPARED scale, the mean (SD) scores were 17.2 (3.8) for the discharge software group and 16.5 (3.9) for the usual care group. The parameter estimate from the generalized estimating equation was significant (P = 0.027; Table 2). Outpatient physicians had slightly better perception of discharge quality for patients assigned to discharge software.
In the questionnaire sent to outpatient practitioners, we requested additional information about discharge communication. When asked about timeliness, outpatient physicians perceived no faster communication with the discharge software (Table 3). We asked about the media for discharge information exchange. It was uncommon for community physicians to receive discharge information via electronic mail (Table 3). Outpatient physicians acknowledged receipt of a minority of facsimile transmissions with no significant difference between discharge software vs. usual care (Table 3). Investigators documented facsimile transmission of the output from the discharge software to outpatient practitioners. Transmission occurred on the first business day after discharge. Despite the documentation of all facsimile transmissions, only 23.4% of patients assigned to discharge software had community practitioners who acknowledged receipt.
| Discharge Software (n = 316) [n (%)] | Usual Care (n = 315) [n (%)] | |
|---|---|---|
| ||
| Question: How soon after discharge did you receive any information (in any form) relating to this patient's hospital admission and discharge plans? | ||
| Within 1‐2 days | 72 (22.8) | 55 (17.5) |
| Within 1 week | 105 (33.2) | 125 (39.7) |
| Longer than 1 week | 36 (11.4) | 41 (13.0) |
| Not received | 20 (6.3) | 26 (8.3) |
| Other | 4 (1.3) | 7 (2.2) |
| Question: How did you receive discharge health status information? (Check all that apply)* | ||
| Written/typed letter | 106 (33.5) | 89 (28.3) |
| Telephone call | 82 (25.9) | 67 (21.3) |
| Fax (facsimile transmission) | 74 (23.4) | 90 (28.6) |
| Electronic mail | 8 (2.5) | 23 (7.3) |
| Other | 15 (4.7) | 15 (4.8) |
In exploratory analyses, we evaluated the effect of hospital physician level of training. We wondered if discharging physician experience or seniority affected perceptions of patients or primary care physicians. We entered level of training as a covariate in generalized estimating equations. When patient perception of discharge preparedness (B‐PREPARED) was the dependent variable, then physician level of training had a nonsignificant coefficient (P > 0.219). Likewise, physician level of training was nonsignificant in models of patient satisfaction with medication information, SIMS (P > 0.068), and outpatient physician perception, Modified Physician‐PREPARED (P > 0.177). We concluded that physician level of training had no influence on the patient‐level outcomes assessed in our study.
We compared the satisfaction of hospital physicians who used the discharge software and the usual care discharge. The proportions of hospital physicians who returned questionnaires were 85.7% (30/35) in the discharge software group and 97% (34/35) in the usual care group. After using their assigned discharge process for at least 6 months, discharge software users had mean (SD) satisfaction 7.4 (1.4) vs. 7.9 (1.4) for usual care physicians (difference = 0.5; 95% CI = 0.2‐1.3; P = 0.129). The effort for discharge software users was more difficult than the effort for usual care (mean [SD] effort = 6.5 [1.9] vs. 7.9 [2.1], respectively). The mean difference in effort was significant (difference = 1.4; 95% CI = 0.3‐2.4; P = 0.011). We reviewed free‐text comments on hospital physician questionnaires. The common theme was software users spent more time to complete discharges. We did not perform time‐motion assessments so we cannot confirm or refute these impressions. Even though hospital physicians found the discharge software significantly more difficult, they did not report a significant decrease in their satisfaction between the 2 discharge interventions.
The cluster design of our trial assumed variance in outcomes measured at the patient level. We predicted some variance attributable to clustering by hospital physician. After the trial, we calculated the intracluster correlation coefficients for B‐PREPARED, SIMS, and Modified Physician‐PREPARED. For all of these outcome variables, the intracluster correlation coefficients were negligible. We also evaluated generalized estimating equations with and without correction for hospital physician cluster. We confirmed the negligible cluster effect on CIs for intervention coefficients (Table 2).
We evaluated the adequacy of the blind for outcome assessors who interviewed patients for B‐PREPARED and SIMS. The guesses of outcomes assessors were unrelated to true intervention assignment (P = 0.253). We interpreted the blind as adequate.
Discussion
We performed a cluster‐randomized clinical trial to measure the effects of discharge software vs. usual care discharge. The discharge software incorporated the ASTM (American Society for Testing and Material) Continuity of Care Record (CCR) standards.19 The CCR is a patient health summary standard with widespread support from medical and specialty organizations. The rationale for the CCR was the need for continuity of care from 1 provider or practitioner to any other practitioner. Our discharge software had the same rationale as the CCR and included a subset of the clinical content specified by the CCR. Like the CCR, our discharge software produced concise reports, and emphasized a brief, postdischarge, care plan. Since we included clinical data elements recommended by the CCR, we hypothesized our discharge software would produce clinically relevant improvements.
Our discharge software also implemented elements of high‐quality discharge planning and communication endorsed by the Society of Hospital Medicine.20 For example, the discharge software produced a legible, typed, discharge plan for the patient or caregiver with medication instructions, follow‐up tests, studies, and appointments. The discharge software generated a discharge summary for the outpatient primary care physician and other clinicians who provided postdischarge care. The summary included discharge diagnoses, key findings and test results, follow‐up appointments, pending diagnostic tests, documentation of patient education, reconciled medication list, and contact information for the hospital physician. The discharge software compiled data for purposes of benchmarking, measurement, and continuous quality improvement. We thought our implementation of discharge software would lead to improved outcomes.
Despite our deployment of recommended strategies, we detected only small increases in patient perceptions of discharge preparedness. We do not know if small changes in B‐PREPARED values were clinically important. We found no improvement in patient satisfaction with medication information. Our results are consistent with systematic reviews that revealed limited benefit of interventions other than discharge planning with postdischarge support.21 Since our discharge software was added to robust discharge planning and support, we possibly had limited ability to detect benefit unless the intervention had a large effect size.
Our discharge software caused a small increase in positive perception reported by outpatient physicians. Small changes in the Modified Physician‐PREPARED had uncertain clinical relevance. Potential delays imposed by our distribution method may have contributed to our findings. Output from our discharge software went to community physicians via facsimile transmission with backup copies via standard U.S. mail. Our distribution system responded to several realities. Most community physicians in our area had no access to interoperable electronic medical records or secured e‐mail. In addition, electronic transmittal of prescriptions was not commonplace. Our discharge intervention did not control the flow of information inside the offices of outpatient physicians. We did not know if our facsimile transmissions joined piles of unread laboratory and imaging reports on the desks of busy primary care physicians. Despite the limited technology available to community physicians, they perceived communication generated by the software to be an improvement over the handwritten process. Our results support previous studies in which physicians preferred computer‐generated discharge summaries and summaries in standardized formats.2224
One of the limitations of our trial design was the unmasked intervention. Hospital physicians assigned to usual care might have improved their handwritten and verbal discharge communication after observation of their colleagues assigned to discharge software. This phenomenon is encountered in unmasked trials and is called contamination. We attempted to minimize contamination when we blocked usual care physicians from access to the discharge software. However, we could not eliminate cross‐talk among unmasked hospital physicians who worked together in close proximity during 27 months of patient enrollment. Some contamination was inevitable. When contamination occurred, there was bias toward the null (increased type II error).
Another limitation was the large proportion of hospital physicians in the first year of postgraduate training. There was a potential for variance from multilevel clusters with patient‐level outcomes clustered within first‐year hospital physicians who were clustered within teams supervised by senior resident or attending physicians. Our results argued against hierarchical clusters because intracluster correlation coefficients were negligible. Furthermore, our exploratory analysis suggested physician training level had no influence on patient outcomes measured in our study. We speculate the highly structured discharge process for both usual care and software minimized variance attributable to physician training level.
The research intervention in our trial was a stand‐alone software application. The discharge software did not integrate with the hospital electronic medical record. Consequently, hospital physician users had to reenter patient demographic data and prescription data that already existed in the electronic record. Data reentry probably caused hospital physicians to attribute greater effort to the discharge software.
In our study, hospital physicians incorporated discharge software with CPOE into their clinical workflow without deterioration in their satisfaction. Our experience may inform the decisions of hospital personnel who design health information systems. When designing discharge functions, developers should consider medication reconciliation and the standards of the CCR.19 Modules within the discharge software would likely be more efficient with prepopulated data from the electronic record. Then users could shift their work from data entry to data verification and possibly mitigate their perceived effort. Software developers may wish to explore options for data transmission to community physicians: secure e‐mail, automated fax servers, and direct digital file transfer. Future studies should test the acceptability of discharge functions incorporated within electronic health records with robust clinical decision support.
Our results apply to a population of adults of all ages with high risk for readmission. The results may not generalize to children, surgical patients, or people with low risk for readmission. All of the patients in our study were discharged to home. The exclusion of other discharge destinations helped us to enroll a homogenous cohort. However, the exclusion criteria did not allow us to generalize our results to patients discharged to nursing homes, inpatient rehabilitation units, or other acute care facilities. We designed the intervention to apply to the hospitalist model, in which responsibility for patient care transitions to a different physician after discharge. The results of our study do not apply when the inpatient and outpatient physician are the same. Since we enrolled general internal medicine hospital physicians, our results may not generalize to care provided by other specialists.
Conclusions
A discharge software application with CPOE improved perceptions of the hospital discharge process for patients and their outpatient physicians. When compared to the handwritten discharge process, the improvements were small in magnitude. Hospital physicians who used the discharge software reported more effort but otherwise no decrement in their satisfaction with the discharge process.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- ,,,,,.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- ,,,.Discharge planning from hospital to home.Cochrane Database Syst Rev.2004;(1):CD000313.
- ,,.Dissemination of discharge summaries. Not reaching follow‐up physicians.Can Fam Physician.2002;48:737–742.
- ,,.Software design to facilitate information transfer at hospital discharge.Inform Prim Care.2006;14:109–119.
- ,.Computer physician order entry: benefits, costs, and issues.Ann Intern Med.2003;139:31–39.
- ,,, et al.Patient readmissions, emergency visits, and adverse events after software‐assisted discharge from hospital: cluster randomized trial.J Hosp Med.2009; DOI: 10.1002/jhm.459. PMID: 19479782.
- ,,.Predictive validity of a questionnaire that identifies older persons at risk for hospital admission.J Am Geriatr Soc.1995;43:374–377.
- ,.Prediction of early readmission in medical inpatients using the Probability of Repeated Admission instrument.Nurs Res.2008;57:406–415.
- ,.The ten point clock test: a quick screen and grading method for cognitive impairment in medical and surgical patients.Int J Psychiatry Med.1994;24:229–244.
- ,,, et al.Patient safety concerns arising from test results that return after hospital discharge.Ann Intern Med.2005;143:121–128.
- ,,, et al.Role of pharmacist counseling in preventing adverse drug events after hospitalization.Arch Intern Med.2006;166:565–571.
- .SF‐36 health survey update.Spine.2000;25:3130–3139.
- ,.The development, validity and application of a new instrument to assess the quality of discharge planning activities from the community perspective.Int J Qual Health Care.2001;13:109–116.
- ,,.Brief scale measuring patient preparedness for hospital discharge to home: psychometric properties.J Hosp Med.2008;3:446–454.
- ,,.The Satisfaction with Information about Medicines Scale (SIMS): a new measurement tool for audit and research.Qual Health Care.2001;10:135–140.
- ,,.Discharge planning scale: community physicians' perspective.J Hosp Med.2008;3:455–464.
- ,.An introduction to generalized estimating equations and an application to assess selectivity effects in a longitudinal study on very old individuals.J Educ Behav Stat.2004;29:421–437. Available at: http://jeb.sagepub.com/cgi/content/abstract/29/4/421. Accessed June 2009.
- ASTM. E2369‐05 Standard Specification for Continuity of Care Record (CCR). Available at: http://www.astm.org/Standards/E2369.htm. Accessed June2009.
- ,,, et al.Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- ,,.Interventions aimed at reducing problems in adult patients discharged from hospital to home: a systematic meta‐review.BMC Health Serv Res.2007;7:47.
- ,,,,,.Evaluation of a computer‐generated discharge summary for patients with acute coronary syndromes.Br J Gen Pract.1998;48:1163–1164.
- ,,,.Standardized or narrative discharge summaries. Which do family physicians prefer?Can Fam Physician.1998;44:62–69.
- ,,,.Dictated versus database‐generated discharge summaries: a randomized clinical trial.CMAJ.1999;160:319–326.
During the transition from inpatient to outpatient care, patients are vulnerable to adverse events.1 Poor communication between hospital personnel and either the patient or the outpatient primary care physician has been associated with preventable or ameliorable adverse events after discharge.1 Systematic reviews confirm that discharge communication is often delayed, inaccurate, or ineffective.2, 3
Discharge communication failures may occur if hospital processes rely on dictated discharge summaries.2 For several reasons, discharge summaries are inadequate for communication. Most patients complete their initial posthospital clinic visit before their primary care physician receives the discharge summary.4 For many patients, the discharge summary is unavailable for all posthospital visits.4 Discharge summaries often fail as communication because they are not generated or transmitted.4
Recommendations to improve discharge communication include the use of health information technology.2, 5 The benefits of computer‐generated discharge summaries include decreases in delivery time for discharge communications.2 The benefits of computerized physician order entry (CPOE) include reduction of medical errors.6 These theoretical benefits create a rationale for clinical trials to measure improvements after discharge software applications with CPOE.5
In an effort to improve discharge communication and clinically relevant outcomes, we performed a cluster‐randomized trial to assess the value of a discharge software application of CPOE. The clustered design followed recommendations from a systematic review of discharge interventions.3 We applied our research intervention at the physician level and measured outcomes at the patient level. Our objective was to assess the benefit of discharge software with CPOE vs. usual care when used to discharge patients at high risk for repeat admission. In a previous work, we reported that discharge software did not reduce rates of hospital readmission, emergency department visits, or postdischarge adverse events due to medical management.7 In the present article, we compare secondary outcomes after the research intervention: perceptions of the discharge from the perspectives of patients, primary care physicians, and hospital physicians.
Methods
The trial design was a cluster randomized, controlled trial. The setting was the postdischarge environment following index hospitalization at a 730‐bed, tertiary care, teaching hospital in central Illinois. The Peoria Institutional Review Board approved the protocol for human research.
Participants
We enrolled consenting hospital physicians and their patients between November 2004 and January 2007. The hospital physician defined the cluster. Patients discharged by the physician comprised the cluster. The eligibility criteria for hospital physicians required internal medicine resident or attending physicians with assignments to inpatient duties for at least 2 months during the 27‐month enrollment period. After achieving informed consent from physicians, research personnel screened all consecutive, adult inpatients who were discharged to home. Patient inclusion required a probability of repeat admission (Pra) equal to or greater than 0.40.8, 9 The purpose of the inclusion criterion was to enrich the sample with patients likely to benefit from interventions to improve discharge processes. Furthermore, hospital readmission was the primary endpoint of the study, as reported separately.7 The Pra came from a predictive model with scores for age, gender, prior hospitalizations, prior doctor visits, self‐rated health status, presence of informal caregiver in the home, and comorbid coronary heart disease and diabetes mellitus. Research coordinators calculated the Pra within 2 days before discharge from the index hospitalization.
Exclusion Criteria
We excluded patients previously enrolled in the study, candidates for hospice, and patients unable to participate in outcome ascertainment. Cognitive impairment was a conditional exclusion criterion for patients. We defined cognitive impairment as a score less than 9 on the 10‐point clock test.10 Patients with cognitive impairment participated only with consent from their legally authorized representative. We enrolled patients with cognitive impairment only if a proxy spent at least 3 hours daily with the patient and the proxy agreed to answer postdischarge interviews. If a patient's outpatient primary care physician treated the patient during the index hospitalization, then there was no perceived barrier in physician‐to‐physician communication and we excluded the patient.
Intervention
The research intervention was discharge software with CPOE. Detailed description of the software appeared previously.5 In summary, the CPOE software application facilitated communication at the time of hospital discharge to patients, retail pharmacists, and community physicians. The application had basic levels of clinical decision support, required fields, pick lists, standard drug doses, alerts, reminders, and online reference information. The software addressed deficiencies in the usual care discharge process reported globally and reviewed previously.5 For example, 1 deficiency occurred when inpatient physicians failed to warn outpatient physicians about diagnostic tests with results pending at discharge.11 Another deficiency was discharge medication error.12 The software prompted the discharging physician to enter pending tests, order tests after discharge, and perform medication reconciliation. On the day of discharge, hospital physicians used the software to automatically generate discharge documents and reconcile prescriptions for the patient, primary care physician, retail pharmacist, and the ward nurse. The discharge letter went to the outpatient practitioner via facsimile transmission plus a duplicate via U.S. mail.
The control intervention was the usual care, handwritten discharge process commonly used by hospitalists.2 Hospital physicians and ward nurses completed handwritten discharge forms on the day of discharge. The forms contained blanks for discharge diagnoses, discharge medications, medication instructions, postdischarge activities and restrictions, postdischarge diet, postdischarge diagnostic and therapeutic interventions, and appointments. Patients received handwritten copies of the forms, 1 page of which also included medication instructions and prescriptions. In a previous publication, we provided details about the usual care discharge process as well as the standard care available to all study patients regardless of intervention.5
Randomization
The hospital physician who completed the discharge process was the unit of randomization. Random allocation was to discharge software or usual care discharge process, with a randomization ratio of 1:1 and block size of 2. We concealed allocation with the following process. An investigator who was not involved with hospital physician recruitment generated the randomization sequence with a computerized random number generator. The randomization list was maintained in a secure location. Another investigator who was unaware of the next random assignment performed the hospital physician recruitment and informed consent. After confirming eligibility and obtaining informed consent from physicians, the blinded investigator requested the next random assignment from the custodian of the randomization list. Hospital physicians subsequently used their randomly assigned process when discharging their patients who enrolled in the study. After random allocation, it was not possible to conceal the test or control intervention from physicians or their patients.
Hospital physicians underwent training on the software or usual care discharge process; the details appeared previously.7 Physicians assigned to usual care did not receive training on the discharge software and were blocked from using the software. Patients were passive recipients of the research intervention performed by their discharging physician. Patients received the research intervention on the day of discharge of the index hospitalization.
Baseline Assessment
During the index hospitalization, trained data abstractors recorded baseline patient demographic data plus variables to calculate the Pra for probability for repeat admission. We recorded the availability of an informal caregiver in response to the question, Is there a friend, relative, or neighbor who would take care of you for a few days, if necessary? Data came from the patient or proxy for physical functioning, mental health,13 heart failure, and number of previous emergency department visits. Other data came from hospital records for chronic obstructive pulmonary disease, number of discharge medications, and length of stay for the index hospitalization.
Outcome Assessment
We assessed the patient's perception of the discharge with 2 validated survey instruments. One week after discharge, research personnel performed telephone interviews with patients or proxies. While following a script, interviewers instructed patients to avoid mentioning the discharge process. Interviewers read items from the B‐PREPARED questionnaire.14, 15 and the Satisfaction with Information About Medicines Scale (SIMS).16 The B‐PREPARED scale assessed 3 principal components of patient preparedness for discharge: self‐care information for medications and activities, equipment and services, and confidence. The scale demonstrated internal consistency, construct validity, and predictive validity. High scale values reflected high perceptions of discharge preparedness from the patient perspective.15 SIMS measured patient satisfaction with information about discharge medications. Validation studies revealed SIMS had internal consistency, test‐retest reliability, and criterion‐related validity.16 Interviewers recorded responses to calculate a total SIMS score. Patients with high total SIMS scores had high satisfaction. While assessing B‐PREPARED and SIMS, interviewers were blind to intervention assignment. We evaluated the adequacy of blinding by asking interviewers to guess the patient's intervention assignment.
We measured the quality of hospital discharge from the outpatient physician perspective. During the index hospitalization, patients designated an outpatient primary care practitioner to receive discharge reports and results of diagnostic tests. Ten days after discharge, research personnel mailed the Physician‐PREPARED questionnaire to the designated community practitioner.17 The sum of item responses comprised the Modified Physician‐PREPARED scale and demonstrated internal consistency and construct validity. The principal components of the Modified Physician‐PREPARED were timeliness of communication and adequacy of discharge plan/transmission. High scale values reflected high perceptions of discharge quality.17 Outpatient practitioners gave implied consent when they completed and returned questionnaires. We requested 1 questionnaire for each enrolled patient, so the outcome assessment was at the patient level. The assessment was not blinded because primary care physicians received the output of discharge software or usual care discharge.
We assessed the satisfaction of hospital physicians who used the discharge software and the usual care. After hospital physicians participated in the trial for 6 months, they rated their assigned discharge process on Likert scales. The first question was, On a scale of 1 to 10, indicate your satisfaction with your portion of the discharge process. The scale anchors were 1 for very dissatisfied and 10 for very satisfied. The second question was, On a scale of 1 to 10, indicate the effort to complete your portion of the discharge process. For the second question, the scale anchors were 1 for very difficult and 10 for very easy. It was not possible to mask the hospital physicians after they received their intervention assignment. Consequently, their outcome assessment was not blinded.
Statistical Methods
The cluster number and size were selected to maintain test significance level, 1‐sided alpha less than 0.05, and power greater than 80%. We previously published the assumptions and rationale for 35 hospital physician clusters per intervention and 9 patients per cluster.7 We did not perform separate sample size estimates for the secondary outcomes reported herein.
The statistical analyses employed SPSS PC (Version 15.0.1; SPSS, Inc., Chicago, IL). Statistical procedures for baseline variables were descriptive and included means and standard deviations (SDs) for interval variables and percentages for categorical variables. For all analyses, we employed the principle of intention‐to‐treat. We assumed patient or physician exposure to the intervention randomly assigned to the discharging physician. Analyses employed standard tests for normal distribution, homogeneity of variance, and linearity of relationships between independent and dependent variables. If assumptions failed, then we stratified variables or performed transformations. We accepted P < 0.05 as significant.
We tested hypotheses for patient‐level outcomes with generalized estimating equations (GEEs) that corrected for clustering by hospital physician. We employed GEEs because they provide unbiased estimates of standard errors for parameters even with incorrect specification of the intracluster dependence structure.18 Each patient‐level outcome was the dependent variable in a separate GEE. The intervention variable for each GEE was discharge software vs. usual care, handwritten discharge. The statistic of interest was the coefficient for the intervention variable. The null hypothesis was no difference between discharge software and usual care. The statistical definition of the null hypothesis was an intervention variable coefficient with a 95% confidence interval (CI) that included 0.
For analyses that were unaffected by the cluster assumption, we performed standard tests. The hypothesis for hospital physicians was significantly higher satisfaction for discharge software users and the inferential procedure was the t test. When we assessed the success of the study blinding, we assumed no association between true intervention allocation and guesses by outcome assessors. We used chi‐square for assessment of the blinding.
Results
We screened 127 physicians who were general internal medicine hospital physicians. Seventy physicians consented and received random allocation to discharge software or usual care. We excluded 57 physicians for reasons shown in the trial flow diagram (Figure 1). We approached 6884 patients during their index hospitalization. After excluding 6253 ineligible patients, we enrolled 631 willing patients (Supplementary Appendix). As depicted in Figure 1, the most common reason for ineligibility occurred for patients with Pra score <0.40 (2168/6253 exclusions; 34.7%). We followed 631 patients who received the discharge intervention (Figure 1). There was no differential dropout between the interventions. Protocol deviations were rare, 0.5% (3/631). Three patients erroneously received usual care discharge from physicians assigned to discharge software. All 631 patients were included in the intention‐to‐treat analysis. The baseline characteristics of the randomly assigned hospital physicians and their patients are in Table 1. Most of the hospital physicians were residents in the first year of postgraduate training.

| Discharge Software | Usual Care | |
|---|---|---|
| ||
| Hospital physician characteristics, n (%) | n = 35 | n = 35 |
| Postgraduate year 1 | 18 (51.4) | 23 (65.7) |
| Postgraduate years 2‐4 | 10 (28.6) | 7 (20.0) |
| Attending physician | 7 (20.0) | 5 (14.3) |
| Patient characteristics, n (%) | n = 316 | n = 315 |
| Gender, male | 136 (43.0) | 147 (46.7) |
| Age, years | ||
| 18‐44 | 68 (21.5) | 95 (30.2) |
| 45‐54 | 79 (25.0) | 76 (24.1) |
| 55‐64 | 86 (27.2) | 74 (23.5) |
| 65‐98 | 83 (26.3) | 70 (22.2) |
| Self‐rated health status | ||
| Poor | 82 (25.9) | 108 (34.3) |
| Fair | 169 (53.5) | 147 (46.7) |
| Good | 54 (17.1) | 46 (14.6) |
| Very good | 10 (3.2) | 11 (3.5) |
| Excellent | 1 (0.3) | 3 (1.0) |
| Diabetes mellitus | 172 (54.4) | 177 (56.2) |
| Chronic obstructive pulmonary disease | ||
| None | 259 (82.0) | 257 (81.6) |
| Without oral steroid or home oxygen | 28 (8.9) | 26 (8.3) |
| With chronic oral steroid | 10 (3.2) | 8 (2.5) |
| With home oxygen oral steroid | 19 (6.0) | 24 (7.6) |
| Coronary heart disease | 133 (42.1) | 120 (38.1) |
| Heart failure | 80 (25.3) | 67 (21.3) |
| Physical Functioning from SF‐36 | ||
| Lowest third | 128 (40.5) | 121 (38.4) |
| Upper two‐thirds | 188 (59.5) | 194 (61.6) |
| Mental Health from SF‐36 | ||
| Lowest one‐third | 113 (35.8) | 117 (37.1)* |
| Upper two‐thirds | 203 (64.2) | 197 (62.5)* |
| Emergency department visits during 6 months before index admission | ||
| 0 or 1 | 194 (61.4) | 168 (53.3) |
| 2 or more | 122 (38.6) | 147 (46.7) |
| Mean (SD) | ||
| Number of discharge medications | 10.5 (4.8) | 9.9 (5.1) |
| Index hospital length of stay, days | 3.9 (3.5) | 3.5 (3.5) |
| Pra | 0.486 (0.072) | 0.495 (0.076) |
We assessed the patient's perception of discharge preparedness. One week after discharge, research personnel interviewed 92.4% (292/316) of patients in the discharge software group and 92.4% (291/315) in the usual care group. The mean (SD) B‐PREPARED scores for discharge preparedness were 17.7 (4.1) in the discharge software group and 17.2 (4.0) in the usual care group. In the generalized estimating equation that accounted for potential clustering within hospital physicians, the parameter estimate for the intervention variable coefficient was small but significant (P = 0.042; Table 2). Patients in the discharge software group had slightly better perceptions of their discharge preparedness.
| Outcome Variable | Discharge Software [mean (SD)] | Usual Care [mean (SD)] | Parameter Estimate Without Cluster Correction (95% CI) | P Value | Parameter Estimate with Cluster Correction (95% CI) | P Value |
|---|---|---|---|---|---|---|
| ||||||
| Patient perception of discharge preparedness (B‐PREPARED) | 17.7 (4.1) | 17.2 (4.0) | 0.147* (0.006‐0.288) | 0.040 | 0.147* (0.005‐0.289) | 0.042 |
| Patient satisfaction with medication information score (SIMS) | 12.3 (4.8) | 12.1 (4.6) | 0.212 (0.978‐0.554) | 0.587 | 0.212 (0.937‐0.513) | 0.567 |
| Outpatient physician perception (Modified Physician‐PREPARED) | 17.2 (3.8) | 16.5 (3.9) | 0.133 (0.012‐0.254) | 0.031 | 0.133 (0.015‐0.251) | 0.027 |
Another outcome was the patient's satisfaction with information about discharge medications (Table 2). One week after discharge, mean (SD) SIMS scores for satisfaction were 12.3 (4.8) in the discharge software group and 12.1 (4.6) in the usual care group. The generalized estimating equation revealed an insignificant coefficient for the intervention variable (P = 0.567; Table 2).
We assessed the outpatient physician perception of the discharge with a questionnaire sent 10 days after discharge. We received 496 out of 631 questionnaires (78.6%) from outpatient practitioners and the median response time was 19 days after the date of discharge. The practitioner specialty was internal medicine for 38.9% (193/496), family medicine for 27.2% (135/496), medicine‐pediatrics for 27.0% (134/496), advance practice nurse for 4.4% (22/496), other physician specialties for 2.0% (10/496), and physician assistant for 0.4% (2/496). We excluded 18 questionnaires from analysis because outpatient practitioners failed to answer 2 or more items in the Modified Physician‐PREPARED scale. When we compared baseline characteristics for patients who had complete questionnaires vs. patients with nonrespondent or excluded questionnaires, we found no significant differences (data available upon request). Among the discharge software group, 72.2% (228/316) of patients had complete questionnaires from their outpatient physicians. The response rate with complete questionnaires was 79.4% (250/315) of patients assigned to usual care. On the Modified Physician‐PREPARED scale, the mean (SD) scores were 17.2 (3.8) for the discharge software group and 16.5 (3.9) for the usual care group. The parameter estimate from the generalized estimating equation was significant (P = 0.027; Table 2). Outpatient physicians had slightly better perception of discharge quality for patients assigned to discharge software.
In the questionnaire sent to outpatient practitioners, we requested additional information about discharge communication. When asked about timeliness, outpatient physicians perceived no faster communication with the discharge software (Table 3). We asked about the media for discharge information exchange. It was uncommon for community physicians to receive discharge information via electronic mail (Table 3). Outpatient physicians acknowledged receipt of a minority of facsimile transmissions with no significant difference between discharge software vs. usual care (Table 3). Investigators documented facsimile transmission of the output from the discharge software to outpatient practitioners. Transmission occurred on the first business day after discharge. Despite the documentation of all facsimile transmissions, only 23.4% of patients assigned to discharge software had community practitioners who acknowledged receipt.
| Discharge Software (n = 316) [n (%)] | Usual Care (n = 315) [n (%)] | |
|---|---|---|
| ||
| Question: How soon after discharge did you receive any information (in any form) relating to this patient's hospital admission and discharge plans? | ||
| Within 1‐2 days | 72 (22.8) | 55 (17.5) |
| Within 1 week | 105 (33.2) | 125 (39.7) |
| Longer than 1 week | 36 (11.4) | 41 (13.0) |
| Not received | 20 (6.3) | 26 (8.3) |
| Other | 4 (1.3) | 7 (2.2) |
| Question: How did you receive discharge health status information? (Check all that apply)* | ||
| Written/typed letter | 106 (33.5) | 89 (28.3) |
| Telephone call | 82 (25.9) | 67 (21.3) |
| Fax (facsimile transmission) | 74 (23.4) | 90 (28.6) |
| Electronic mail | 8 (2.5) | 23 (7.3) |
| Other | 15 (4.7) | 15 (4.8) |
In exploratory analyses, we evaluated the effect of hospital physician level of training. We wondered if discharging physician experience or seniority affected perceptions of patients or primary care physicians. We entered level of training as a covariate in generalized estimating equations. When patient perception of discharge preparedness (B‐PREPARED) was the dependent variable, then physician level of training had a nonsignificant coefficient (P > 0.219). Likewise, physician level of training was nonsignificant in models of patient satisfaction with medication information, SIMS (P > 0.068), and outpatient physician perception, Modified Physician‐PREPARED (P > 0.177). We concluded that physician level of training had no influence on the patient‐level outcomes assessed in our study.
We compared the satisfaction of hospital physicians who used the discharge software and the usual care discharge. The proportions of hospital physicians who returned questionnaires were 85.7% (30/35) in the discharge software group and 97% (34/35) in the usual care group. After using their assigned discharge process for at least 6 months, discharge software users had mean (SD) satisfaction 7.4 (1.4) vs. 7.9 (1.4) for usual care physicians (difference = 0.5; 95% CI = 0.2‐1.3; P = 0.129). The effort for discharge software users was more difficult than the effort for usual care (mean [SD] effort = 6.5 [1.9] vs. 7.9 [2.1], respectively). The mean difference in effort was significant (difference = 1.4; 95% CI = 0.3‐2.4; P = 0.011). We reviewed free‐text comments on hospital physician questionnaires. The common theme was software users spent more time to complete discharges. We did not perform time‐motion assessments so we cannot confirm or refute these impressions. Even though hospital physicians found the discharge software significantly more difficult, they did not report a significant decrease in their satisfaction between the 2 discharge interventions.
The cluster design of our trial assumed variance in outcomes measured at the patient level. We predicted some variance attributable to clustering by hospital physician. After the trial, we calculated the intracluster correlation coefficients for B‐PREPARED, SIMS, and Modified Physician‐PREPARED. For all of these outcome variables, the intracluster correlation coefficients were negligible. We also evaluated generalized estimating equations with and without correction for hospital physician cluster. We confirmed the negligible cluster effect on CIs for intervention coefficients (Table 2).
We evaluated the adequacy of the blind for outcome assessors who interviewed patients for B‐PREPARED and SIMS. The guesses of outcomes assessors were unrelated to true intervention assignment (P = 0.253). We interpreted the blind as adequate.
Discussion
We performed a cluster‐randomized clinical trial to measure the effects of discharge software vs. usual care discharge. The discharge software incorporated the ASTM (American Society for Testing and Material) Continuity of Care Record (CCR) standards.19 The CCR is a patient health summary standard with widespread support from medical and specialty organizations. The rationale for the CCR was the need for continuity of care from 1 provider or practitioner to any other practitioner. Our discharge software had the same rationale as the CCR and included a subset of the clinical content specified by the CCR. Like the CCR, our discharge software produced concise reports, and emphasized a brief, postdischarge, care plan. Since we included clinical data elements recommended by the CCR, we hypothesized our discharge software would produce clinically relevant improvements.
Our discharge software also implemented elements of high‐quality discharge planning and communication endorsed by the Society of Hospital Medicine.20 For example, the discharge software produced a legible, typed, discharge plan for the patient or caregiver with medication instructions, follow‐up tests, studies, and appointments. The discharge software generated a discharge summary for the outpatient primary care physician and other clinicians who provided postdischarge care. The summary included discharge diagnoses, key findings and test results, follow‐up appointments, pending diagnostic tests, documentation of patient education, reconciled medication list, and contact information for the hospital physician. The discharge software compiled data for purposes of benchmarking, measurement, and continuous quality improvement. We thought our implementation of discharge software would lead to improved outcomes.
Despite our deployment of recommended strategies, we detected only small increases in patient perceptions of discharge preparedness. We do not know if small changes in B‐PREPARED values were clinically important. We found no improvement in patient satisfaction with medication information. Our results are consistent with systematic reviews that revealed limited benefit of interventions other than discharge planning with postdischarge support.21 Since our discharge software was added to robust discharge planning and support, we possibly had limited ability to detect benefit unless the intervention had a large effect size.
Our discharge software caused a small increase in positive perception reported by outpatient physicians. Small changes in the Modified Physician‐PREPARED had uncertain clinical relevance. Potential delays imposed by our distribution method may have contributed to our findings. Output from our discharge software went to community physicians via facsimile transmission with backup copies via standard U.S. mail. Our distribution system responded to several realities. Most community physicians in our area had no access to interoperable electronic medical records or secured e‐mail. In addition, electronic transmittal of prescriptions was not commonplace. Our discharge intervention did not control the flow of information inside the offices of outpatient physicians. We did not know if our facsimile transmissions joined piles of unread laboratory and imaging reports on the desks of busy primary care physicians. Despite the limited technology available to community physicians, they perceived communication generated by the software to be an improvement over the handwritten process. Our results support previous studies in which physicians preferred computer‐generated discharge summaries and summaries in standardized formats.2224
One of the limitations of our trial design was the unmasked intervention. Hospital physicians assigned to usual care might have improved their handwritten and verbal discharge communication after observation of their colleagues assigned to discharge software. This phenomenon is encountered in unmasked trials and is called contamination. We attempted to minimize contamination when we blocked usual care physicians from access to the discharge software. However, we could not eliminate cross‐talk among unmasked hospital physicians who worked together in close proximity during 27 months of patient enrollment. Some contamination was inevitable. When contamination occurred, there was bias toward the null (increased type II error).
Another limitation was the large proportion of hospital physicians in the first year of postgraduate training. There was a potential for variance from multilevel clusters with patient‐level outcomes clustered within first‐year hospital physicians who were clustered within teams supervised by senior resident or attending physicians. Our results argued against hierarchical clusters because intracluster correlation coefficients were negligible. Furthermore, our exploratory analysis suggested physician training level had no influence on patient outcomes measured in our study. We speculate the highly structured discharge process for both usual care and software minimized variance attributable to physician training level.
The research intervention in our trial was a stand‐alone software application. The discharge software did not integrate with the hospital electronic medical record. Consequently, hospital physician users had to reenter patient demographic data and prescription data that already existed in the electronic record. Data reentry probably caused hospital physicians to attribute greater effort to the discharge software.
In our study, hospital physicians incorporated discharge software with CPOE into their clinical workflow without deterioration in their satisfaction. Our experience may inform the decisions of hospital personnel who design health information systems. When designing discharge functions, developers should consider medication reconciliation and the standards of the CCR.19 Modules within the discharge software would likely be more efficient with prepopulated data from the electronic record. Then users could shift their work from data entry to data verification and possibly mitigate their perceived effort. Software developers may wish to explore options for data transmission to community physicians: secure e‐mail, automated fax servers, and direct digital file transfer. Future studies should test the acceptability of discharge functions incorporated within electronic health records with robust clinical decision support.
Our results apply to a population of adults of all ages with high risk for readmission. The results may not generalize to children, surgical patients, or people with low risk for readmission. All of the patients in our study were discharged to home. The exclusion of other discharge destinations helped us to enroll a homogenous cohort. However, the exclusion criteria did not allow us to generalize our results to patients discharged to nursing homes, inpatient rehabilitation units, or other acute care facilities. We designed the intervention to apply to the hospitalist model, in which responsibility for patient care transitions to a different physician after discharge. The results of our study do not apply when the inpatient and outpatient physician are the same. Since we enrolled general internal medicine hospital physicians, our results may not generalize to care provided by other specialists.
Conclusions
A discharge software application with CPOE improved perceptions of the hospital discharge process for patients and their outpatient physicians. When compared to the handwritten discharge process, the improvements were small in magnitude. Hospital physicians who used the discharge software reported more effort but otherwise no decrement in their satisfaction with the discharge process.
During the transition from inpatient to outpatient care, patients are vulnerable to adverse events.1 Poor communication between hospital personnel and either the patient or the outpatient primary care physician has been associated with preventable or ameliorable adverse events after discharge.1 Systematic reviews confirm that discharge communication is often delayed, inaccurate, or ineffective.2, 3
Discharge communication failures may occur if hospital processes rely on dictated discharge summaries.2 For several reasons, discharge summaries are inadequate for communication. Most patients complete their initial posthospital clinic visit before their primary care physician receives the discharge summary.4 For many patients, the discharge summary is unavailable for all posthospital visits.4 Discharge summaries often fail as communication because they are not generated or transmitted.4
Recommendations to improve discharge communication include the use of health information technology.2, 5 The benefits of computer‐generated discharge summaries include decreases in delivery time for discharge communications.2 The benefits of computerized physician order entry (CPOE) include reduction of medical errors.6 These theoretical benefits create a rationale for clinical trials to measure improvements after discharge software applications with CPOE.5
In an effort to improve discharge communication and clinically relevant outcomes, we performed a cluster‐randomized trial to assess the value of a discharge software application of CPOE. The clustered design followed recommendations from a systematic review of discharge interventions.3 We applied our research intervention at the physician level and measured outcomes at the patient level. Our objective was to assess the benefit of discharge software with CPOE vs. usual care when used to discharge patients at high risk for repeat admission. In a previous work, we reported that discharge software did not reduce rates of hospital readmission, emergency department visits, or postdischarge adverse events due to medical management.7 In the present article, we compare secondary outcomes after the research intervention: perceptions of the discharge from the perspectives of patients, primary care physicians, and hospital physicians.
Methods
The trial design was a cluster randomized, controlled trial. The setting was the postdischarge environment following index hospitalization at a 730‐bed, tertiary care, teaching hospital in central Illinois. The Peoria Institutional Review Board approved the protocol for human research.
Participants
We enrolled consenting hospital physicians and their patients between November 2004 and January 2007. The hospital physician defined the cluster. Patients discharged by the physician comprised the cluster. The eligibility criteria for hospital physicians required internal medicine resident or attending physicians with assignments to inpatient duties for at least 2 months during the 27‐month enrollment period. After achieving informed consent from physicians, research personnel screened all consecutive, adult inpatients who were discharged to home. Patient inclusion required a probability of repeat admission (Pra) equal to or greater than 0.40.8, 9 The purpose of the inclusion criterion was to enrich the sample with patients likely to benefit from interventions to improve discharge processes. Furthermore, hospital readmission was the primary endpoint of the study, as reported separately.7 The Pra came from a predictive model with scores for age, gender, prior hospitalizations, prior doctor visits, self‐rated health status, presence of informal caregiver in the home, and comorbid coronary heart disease and diabetes mellitus. Research coordinators calculated the Pra within 2 days before discharge from the index hospitalization.
Exclusion Criteria
We excluded patients previously enrolled in the study, candidates for hospice, and patients unable to participate in outcome ascertainment. Cognitive impairment was a conditional exclusion criterion for patients. We defined cognitive impairment as a score less than 9 on the 10‐point clock test.10 Patients with cognitive impairment participated only with consent from their legally authorized representative. We enrolled patients with cognitive impairment only if a proxy spent at least 3 hours daily with the patient and the proxy agreed to answer postdischarge interviews. If a patient's outpatient primary care physician treated the patient during the index hospitalization, then there was no perceived barrier in physician‐to‐physician communication and we excluded the patient.
Intervention
The research intervention was discharge software with CPOE. Detailed description of the software appeared previously.5 In summary, the CPOE software application facilitated communication at the time of hospital discharge to patients, retail pharmacists, and community physicians. The application had basic levels of clinical decision support, required fields, pick lists, standard drug doses, alerts, reminders, and online reference information. The software addressed deficiencies in the usual care discharge process reported globally and reviewed previously.5 For example, 1 deficiency occurred when inpatient physicians failed to warn outpatient physicians about diagnostic tests with results pending at discharge.11 Another deficiency was discharge medication error.12 The software prompted the discharging physician to enter pending tests, order tests after discharge, and perform medication reconciliation. On the day of discharge, hospital physicians used the software to automatically generate discharge documents and reconcile prescriptions for the patient, primary care physician, retail pharmacist, and the ward nurse. The discharge letter went to the outpatient practitioner via facsimile transmission plus a duplicate via U.S. mail.
The control intervention was the usual care, handwritten discharge process commonly used by hospitalists.2 Hospital physicians and ward nurses completed handwritten discharge forms on the day of discharge. The forms contained blanks for discharge diagnoses, discharge medications, medication instructions, postdischarge activities and restrictions, postdischarge diet, postdischarge diagnostic and therapeutic interventions, and appointments. Patients received handwritten copies of the forms, 1 page of which also included medication instructions and prescriptions. In a previous publication, we provided details about the usual care discharge process as well as the standard care available to all study patients regardless of intervention.5
Randomization
The hospital physician who completed the discharge process was the unit of randomization. Random allocation was to discharge software or usual care discharge process, with a randomization ratio of 1:1 and block size of 2. We concealed allocation with the following process. An investigator who was not involved with hospital physician recruitment generated the randomization sequence with a computerized random number generator. The randomization list was maintained in a secure location. Another investigator who was unaware of the next random assignment performed the hospital physician recruitment and informed consent. After confirming eligibility and obtaining informed consent from physicians, the blinded investigator requested the next random assignment from the custodian of the randomization list. Hospital physicians subsequently used their randomly assigned process when discharging their patients who enrolled in the study. After random allocation, it was not possible to conceal the test or control intervention from physicians or their patients.
Hospital physicians underwent training on the software or usual care discharge process; the details appeared previously.7 Physicians assigned to usual care did not receive training on the discharge software and were blocked from using the software. Patients were passive recipients of the research intervention performed by their discharging physician. Patients received the research intervention on the day of discharge of the index hospitalization.
Baseline Assessment
During the index hospitalization, trained data abstractors recorded baseline patient demographic data plus variables to calculate the Pra for probability for repeat admission. We recorded the availability of an informal caregiver in response to the question, Is there a friend, relative, or neighbor who would take care of you for a few days, if necessary? Data came from the patient or proxy for physical functioning, mental health,13 heart failure, and number of previous emergency department visits. Other data came from hospital records for chronic obstructive pulmonary disease, number of discharge medications, and length of stay for the index hospitalization.
Outcome Assessment
We assessed the patient's perception of the discharge with 2 validated survey instruments. One week after discharge, research personnel performed telephone interviews with patients or proxies. While following a script, interviewers instructed patients to avoid mentioning the discharge process. Interviewers read items from the B‐PREPARED questionnaire.14, 15 and the Satisfaction with Information About Medicines Scale (SIMS).16 The B‐PREPARED scale assessed 3 principal components of patient preparedness for discharge: self‐care information for medications and activities, equipment and services, and confidence. The scale demonstrated internal consistency, construct validity, and predictive validity. High scale values reflected high perceptions of discharge preparedness from the patient perspective.15 SIMS measured patient satisfaction with information about discharge medications. Validation studies revealed SIMS had internal consistency, test‐retest reliability, and criterion‐related validity.16 Interviewers recorded responses to calculate a total SIMS score. Patients with high total SIMS scores had high satisfaction. While assessing B‐PREPARED and SIMS, interviewers were blind to intervention assignment. We evaluated the adequacy of blinding by asking interviewers to guess the patient's intervention assignment.
We measured the quality of hospital discharge from the outpatient physician perspective. During the index hospitalization, patients designated an outpatient primary care practitioner to receive discharge reports and results of diagnostic tests. Ten days after discharge, research personnel mailed the Physician‐PREPARED questionnaire to the designated community practitioner.17 The sum of item responses comprised the Modified Physician‐PREPARED scale and demonstrated internal consistency and construct validity. The principal components of the Modified Physician‐PREPARED were timeliness of communication and adequacy of discharge plan/transmission. High scale values reflected high perceptions of discharge quality.17 Outpatient practitioners gave implied consent when they completed and returned questionnaires. We requested 1 questionnaire for each enrolled patient, so the outcome assessment was at the patient level. The assessment was not blinded because primary care physicians received the output of discharge software or usual care discharge.
We assessed the satisfaction of hospital physicians who used the discharge software and the usual care. After hospital physicians participated in the trial for 6 months, they rated their assigned discharge process on Likert scales. The first question was, On a scale of 1 to 10, indicate your satisfaction with your portion of the discharge process. The scale anchors were 1 for very dissatisfied and 10 for very satisfied. The second question was, On a scale of 1 to 10, indicate the effort to complete your portion of the discharge process. For the second question, the scale anchors were 1 for very difficult and 10 for very easy. It was not possible to mask the hospital physicians after they received their intervention assignment. Consequently, their outcome assessment was not blinded.
Statistical Methods
The cluster number and size were selected to maintain test significance level, 1‐sided alpha less than 0.05, and power greater than 80%. We previously published the assumptions and rationale for 35 hospital physician clusters per intervention and 9 patients per cluster.7 We did not perform separate sample size estimates for the secondary outcomes reported herein.
The statistical analyses employed SPSS PC (Version 15.0.1; SPSS, Inc., Chicago, IL). Statistical procedures for baseline variables were descriptive and included means and standard deviations (SDs) for interval variables and percentages for categorical variables. For all analyses, we employed the principle of intention‐to‐treat. We assumed patient or physician exposure to the intervention randomly assigned to the discharging physician. Analyses employed standard tests for normal distribution, homogeneity of variance, and linearity of relationships between independent and dependent variables. If assumptions failed, then we stratified variables or performed transformations. We accepted P < 0.05 as significant.
We tested hypotheses for patient‐level outcomes with generalized estimating equations (GEEs) that corrected for clustering by hospital physician. We employed GEEs because they provide unbiased estimates of standard errors for parameters even with incorrect specification of the intracluster dependence structure.18 Each patient‐level outcome was the dependent variable in a separate GEE. The intervention variable for each GEE was discharge software vs. usual care, handwritten discharge. The statistic of interest was the coefficient for the intervention variable. The null hypothesis was no difference between discharge software and usual care. The statistical definition of the null hypothesis was an intervention variable coefficient with a 95% confidence interval (CI) that included 0.
For analyses that were unaffected by the cluster assumption, we performed standard tests. The hypothesis for hospital physicians was significantly higher satisfaction for discharge software users and the inferential procedure was the t test. When we assessed the success of the study blinding, we assumed no association between true intervention allocation and guesses by outcome assessors. We used chi‐square for assessment of the blinding.
Results
We screened 127 physicians who were general internal medicine hospital physicians. Seventy physicians consented and received random allocation to discharge software or usual care. We excluded 57 physicians for reasons shown in the trial flow diagram (Figure 1). We approached 6884 patients during their index hospitalization. After excluding 6253 ineligible patients, we enrolled 631 willing patients (Supplementary Appendix). As depicted in Figure 1, the most common reason for ineligibility occurred for patients with Pra score <0.40 (2168/6253 exclusions; 34.7%). We followed 631 patients who received the discharge intervention (Figure 1). There was no differential dropout between the interventions. Protocol deviations were rare, 0.5% (3/631). Three patients erroneously received usual care discharge from physicians assigned to discharge software. All 631 patients were included in the intention‐to‐treat analysis. The baseline characteristics of the randomly assigned hospital physicians and their patients are in Table 1. Most of the hospital physicians were residents in the first year of postgraduate training.

| Discharge Software | Usual Care | |
|---|---|---|
| ||
| Hospital physician characteristics, n (%) | n = 35 | n = 35 |
| Postgraduate year 1 | 18 (51.4) | 23 (65.7) |
| Postgraduate years 2‐4 | 10 (28.6) | 7 (20.0) |
| Attending physician | 7 (20.0) | 5 (14.3) |
| Patient characteristics, n (%) | n = 316 | n = 315 |
| Gender, male | 136 (43.0) | 147 (46.7) |
| Age, years | ||
| 18‐44 | 68 (21.5) | 95 (30.2) |
| 45‐54 | 79 (25.0) | 76 (24.1) |
| 55‐64 | 86 (27.2) | 74 (23.5) |
| 65‐98 | 83 (26.3) | 70 (22.2) |
| Self‐rated health status | ||
| Poor | 82 (25.9) | 108 (34.3) |
| Fair | 169 (53.5) | 147 (46.7) |
| Good | 54 (17.1) | 46 (14.6) |
| Very good | 10 (3.2) | 11 (3.5) |
| Excellent | 1 (0.3) | 3 (1.0) |
| Diabetes mellitus | 172 (54.4) | 177 (56.2) |
| Chronic obstructive pulmonary disease | ||
| None | 259 (82.0) | 257 (81.6) |
| Without oral steroid or home oxygen | 28 (8.9) | 26 (8.3) |
| With chronic oral steroid | 10 (3.2) | 8 (2.5) |
| With home oxygen oral steroid | 19 (6.0) | 24 (7.6) |
| Coronary heart disease | 133 (42.1) | 120 (38.1) |
| Heart failure | 80 (25.3) | 67 (21.3) |
| Physical Functioning from SF‐36 | ||
| Lowest third | 128 (40.5) | 121 (38.4) |
| Upper two‐thirds | 188 (59.5) | 194 (61.6) |
| Mental Health from SF‐36 | ||
| Lowest one‐third | 113 (35.8) | 117 (37.1)* |
| Upper two‐thirds | 203 (64.2) | 197 (62.5)* |
| Emergency department visits during 6 months before index admission | ||
| 0 or 1 | 194 (61.4) | 168 (53.3) |
| 2 or more | 122 (38.6) | 147 (46.7) |
| Mean (SD) | ||
| Number of discharge medications | 10.5 (4.8) | 9.9 (5.1) |
| Index hospital length of stay, days | 3.9 (3.5) | 3.5 (3.5) |
| Pra | 0.486 (0.072) | 0.495 (0.076) |
We assessed the patient's perception of discharge preparedness. One week after discharge, research personnel interviewed 92.4% (292/316) of patients in the discharge software group and 92.4% (291/315) in the usual care group. The mean (SD) B‐PREPARED scores for discharge preparedness were 17.7 (4.1) in the discharge software group and 17.2 (4.0) in the usual care group. In the generalized estimating equation that accounted for potential clustering within hospital physicians, the parameter estimate for the intervention variable coefficient was small but significant (P = 0.042; Table 2). Patients in the discharge software group had slightly better perceptions of their discharge preparedness.
| Outcome Variable | Discharge Software [mean (SD)] | Usual Care [mean (SD)] | Parameter Estimate Without Cluster Correction (95% CI) | P Value | Parameter Estimate with Cluster Correction (95% CI) | P Value |
|---|---|---|---|---|---|---|
| ||||||
| Patient perception of discharge preparedness (B‐PREPARED) | 17.7 (4.1) | 17.2 (4.0) | 0.147* (0.006‐0.288) | 0.040 | 0.147* (0.005‐0.289) | 0.042 |
| Patient satisfaction with medication information score (SIMS) | 12.3 (4.8) | 12.1 (4.6) | 0.212 (0.978‐0.554) | 0.587 | 0.212 (0.937‐0.513) | 0.567 |
| Outpatient physician perception (Modified Physician‐PREPARED) | 17.2 (3.8) | 16.5 (3.9) | 0.133 (0.012‐0.254) | 0.031 | 0.133 (0.015‐0.251) | 0.027 |
Another outcome was the patient's satisfaction with information about discharge medications (Table 2). One week after discharge, mean (SD) SIMS scores for satisfaction were 12.3 (4.8) in the discharge software group and 12.1 (4.6) in the usual care group. The generalized estimating equation revealed an insignificant coefficient for the intervention variable (P = 0.567; Table 2).
We assessed the outpatient physician perception of the discharge with a questionnaire sent 10 days after discharge. We received 496 out of 631 questionnaires (78.6%) from outpatient practitioners and the median response time was 19 days after the date of discharge. The practitioner specialty was internal medicine for 38.9% (193/496), family medicine for 27.2% (135/496), medicine‐pediatrics for 27.0% (134/496), advance practice nurse for 4.4% (22/496), other physician specialties for 2.0% (10/496), and physician assistant for 0.4% (2/496). We excluded 18 questionnaires from analysis because outpatient practitioners failed to answer 2 or more items in the Modified Physician‐PREPARED scale. When we compared baseline characteristics for patients who had complete questionnaires vs. patients with nonrespondent or excluded questionnaires, we found no significant differences (data available upon request). Among the discharge software group, 72.2% (228/316) of patients had complete questionnaires from their outpatient physicians. The response rate with complete questionnaires was 79.4% (250/315) of patients assigned to usual care. On the Modified Physician‐PREPARED scale, the mean (SD) scores were 17.2 (3.8) for the discharge software group and 16.5 (3.9) for the usual care group. The parameter estimate from the generalized estimating equation was significant (P = 0.027; Table 2). Outpatient physicians had slightly better perception of discharge quality for patients assigned to discharge software.
In the questionnaire sent to outpatient practitioners, we requested additional information about discharge communication. When asked about timeliness, outpatient physicians perceived no faster communication with the discharge software (Table 3). We asked about the media for discharge information exchange. It was uncommon for community physicians to receive discharge information via electronic mail (Table 3). Outpatient physicians acknowledged receipt of a minority of facsimile transmissions with no significant difference between discharge software vs. usual care (Table 3). Investigators documented facsimile transmission of the output from the discharge software to outpatient practitioners. Transmission occurred on the first business day after discharge. Despite the documentation of all facsimile transmissions, only 23.4% of patients assigned to discharge software had community practitioners who acknowledged receipt.
| Discharge Software (n = 316) [n (%)] | Usual Care (n = 315) [n (%)] | |
|---|---|---|
| ||
| Question: How soon after discharge did you receive any information (in any form) relating to this patient's hospital admission and discharge plans? | ||
| Within 1‐2 days | 72 (22.8) | 55 (17.5) |
| Within 1 week | 105 (33.2) | 125 (39.7) |
| Longer than 1 week | 36 (11.4) | 41 (13.0) |
| Not received | 20 (6.3) | 26 (8.3) |
| Other | 4 (1.3) | 7 (2.2) |
| Question: How did you receive discharge health status information? (Check all that apply)* | ||
| Written/typed letter | 106 (33.5) | 89 (28.3) |
| Telephone call | 82 (25.9) | 67 (21.3) |
| Fax (facsimile transmission) | 74 (23.4) | 90 (28.6) |
| Electronic mail | 8 (2.5) | 23 (7.3) |
| Other | 15 (4.7) | 15 (4.8) |
In exploratory analyses, we evaluated the effect of hospital physician level of training. We wondered if discharging physician experience or seniority affected perceptions of patients or primary care physicians. We entered level of training as a covariate in generalized estimating equations. When patient perception of discharge preparedness (B‐PREPARED) was the dependent variable, then physician level of training had a nonsignificant coefficient (P > 0.219). Likewise, physician level of training was nonsignificant in models of patient satisfaction with medication information, SIMS (P > 0.068), and outpatient physician perception, Modified Physician‐PREPARED (P > 0.177). We concluded that physician level of training had no influence on the patient‐level outcomes assessed in our study.
We compared the satisfaction of hospital physicians who used the discharge software and the usual care discharge. The proportions of hospital physicians who returned questionnaires were 85.7% (30/35) in the discharge software group and 97% (34/35) in the usual care group. After using their assigned discharge process for at least 6 months, discharge software users had mean (SD) satisfaction 7.4 (1.4) vs. 7.9 (1.4) for usual care physicians (difference = 0.5; 95% CI = 0.2‐1.3; P = 0.129). The effort for discharge software users was more difficult than the effort for usual care (mean [SD] effort = 6.5 [1.9] vs. 7.9 [2.1], respectively). The mean difference in effort was significant (difference = 1.4; 95% CI = 0.3‐2.4; P = 0.011). We reviewed free‐text comments on hospital physician questionnaires. The common theme was software users spent more time to complete discharges. We did not perform time‐motion assessments so we cannot confirm or refute these impressions. Even though hospital physicians found the discharge software significantly more difficult, they did not report a significant decrease in their satisfaction between the 2 discharge interventions.
The cluster design of our trial assumed variance in outcomes measured at the patient level. We predicted some variance attributable to clustering by hospital physician. After the trial, we calculated the intracluster correlation coefficients for B‐PREPARED, SIMS, and Modified Physician‐PREPARED. For all of these outcome variables, the intracluster correlation coefficients were negligible. We also evaluated generalized estimating equations with and without correction for hospital physician cluster. We confirmed the negligible cluster effect on CIs for intervention coefficients (Table 2).
We evaluated the adequacy of the blind for outcome assessors who interviewed patients for B‐PREPARED and SIMS. The guesses of outcomes assessors were unrelated to true intervention assignment (P = 0.253). We interpreted the blind as adequate.
Discussion
We performed a cluster‐randomized clinical trial to measure the effects of discharge software vs. usual care discharge. The discharge software incorporated the ASTM (American Society for Testing and Material) Continuity of Care Record (CCR) standards.19 The CCR is a patient health summary standard with widespread support from medical and specialty organizations. The rationale for the CCR was the need for continuity of care from 1 provider or practitioner to any other practitioner. Our discharge software had the same rationale as the CCR and included a subset of the clinical content specified by the CCR. Like the CCR, our discharge software produced concise reports, and emphasized a brief, postdischarge, care plan. Since we included clinical data elements recommended by the CCR, we hypothesized our discharge software would produce clinically relevant improvements.
Our discharge software also implemented elements of high‐quality discharge planning and communication endorsed by the Society of Hospital Medicine.20 For example, the discharge software produced a legible, typed, discharge plan for the patient or caregiver with medication instructions, follow‐up tests, studies, and appointments. The discharge software generated a discharge summary for the outpatient primary care physician and other clinicians who provided postdischarge care. The summary included discharge diagnoses, key findings and test results, follow‐up appointments, pending diagnostic tests, documentation of patient education, reconciled medication list, and contact information for the hospital physician. The discharge software compiled data for purposes of benchmarking, measurement, and continuous quality improvement. We thought our implementation of discharge software would lead to improved outcomes.
Despite our deployment of recommended strategies, we detected only small increases in patient perceptions of discharge preparedness. We do not know if small changes in B‐PREPARED values were clinically important. We found no improvement in patient satisfaction with medication information. Our results are consistent with systematic reviews that revealed limited benefit of interventions other than discharge planning with postdischarge support.21 Since our discharge software was added to robust discharge planning and support, we possibly had limited ability to detect benefit unless the intervention had a large effect size.
Our discharge software caused a small increase in positive perception reported by outpatient physicians. Small changes in the Modified Physician‐PREPARED had uncertain clinical relevance. Potential delays imposed by our distribution method may have contributed to our findings. Output from our discharge software went to community physicians via facsimile transmission with backup copies via standard U.S. mail. Our distribution system responded to several realities. Most community physicians in our area had no access to interoperable electronic medical records or secured e‐mail. In addition, electronic transmittal of prescriptions was not commonplace. Our discharge intervention did not control the flow of information inside the offices of outpatient physicians. We did not know if our facsimile transmissions joined piles of unread laboratory and imaging reports on the desks of busy primary care physicians. Despite the limited technology available to community physicians, they perceived communication generated by the software to be an improvement over the handwritten process. Our results support previous studies in which physicians preferred computer‐generated discharge summaries and summaries in standardized formats.2224
One of the limitations of our trial design was the unmasked intervention. Hospital physicians assigned to usual care might have improved their handwritten and verbal discharge communication after observation of their colleagues assigned to discharge software. This phenomenon is encountered in unmasked trials and is called contamination. We attempted to minimize contamination when we blocked usual care physicians from access to the discharge software. However, we could not eliminate cross‐talk among unmasked hospital physicians who worked together in close proximity during 27 months of patient enrollment. Some contamination was inevitable. When contamination occurred, there was bias toward the null (increased type II error).
Another limitation was the large proportion of hospital physicians in the first year of postgraduate training. There was a potential for variance from multilevel clusters with patient‐level outcomes clustered within first‐year hospital physicians who were clustered within teams supervised by senior resident or attending physicians. Our results argued against hierarchical clusters because intracluster correlation coefficients were negligible. Furthermore, our exploratory analysis suggested physician training level had no influence on patient outcomes measured in our study. We speculate the highly structured discharge process for both usual care and software minimized variance attributable to physician training level.
The research intervention in our trial was a stand‐alone software application. The discharge software did not integrate with the hospital electronic medical record. Consequently, hospital physician users had to reenter patient demographic data and prescription data that already existed in the electronic record. Data reentry probably caused hospital physicians to attribute greater effort to the discharge software.
In our study, hospital physicians incorporated discharge software with CPOE into their clinical workflow without deterioration in their satisfaction. Our experience may inform the decisions of hospital personnel who design health information systems. When designing discharge functions, developers should consider medication reconciliation and the standards of the CCR.19 Modules within the discharge software would likely be more efficient with prepopulated data from the electronic record. Then users could shift their work from data entry to data verification and possibly mitigate their perceived effort. Software developers may wish to explore options for data transmission to community physicians: secure e‐mail, automated fax servers, and direct digital file transfer. Future studies should test the acceptability of discharge functions incorporated within electronic health records with robust clinical decision support.
Our results apply to a population of adults of all ages with high risk for readmission. The results may not generalize to children, surgical patients, or people with low risk for readmission. All of the patients in our study were discharged to home. The exclusion of other discharge destinations helped us to enroll a homogenous cohort. However, the exclusion criteria did not allow us to generalize our results to patients discharged to nursing homes, inpatient rehabilitation units, or other acute care facilities. We designed the intervention to apply to the hospitalist model, in which responsibility for patient care transitions to a different physician after discharge. The results of our study do not apply when the inpatient and outpatient physician are the same. Since we enrolled general internal medicine hospital physicians, our results may not generalize to care provided by other specialists.
Conclusions
A discharge software application with CPOE improved perceptions of the hospital discharge process for patients and their outpatient physicians. When compared to the handwritten discharge process, the improvements were small in magnitude. Hospital physicians who used the discharge software reported more effort but otherwise no decrement in their satisfaction with the discharge process.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- ,,,,,.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- ,,,.Discharge planning from hospital to home.Cochrane Database Syst Rev.2004;(1):CD000313.
- ,,.Dissemination of discharge summaries. Not reaching follow‐up physicians.Can Fam Physician.2002;48:737–742.
- ,,.Software design to facilitate information transfer at hospital discharge.Inform Prim Care.2006;14:109–119.
- ,.Computer physician order entry: benefits, costs, and issues.Ann Intern Med.2003;139:31–39.
- ,,, et al.Patient readmissions, emergency visits, and adverse events after software‐assisted discharge from hospital: cluster randomized trial.J Hosp Med.2009; DOI: 10.1002/jhm.459. PMID: 19479782.
- ,,.Predictive validity of a questionnaire that identifies older persons at risk for hospital admission.J Am Geriatr Soc.1995;43:374–377.
- ,.Prediction of early readmission in medical inpatients using the Probability of Repeated Admission instrument.Nurs Res.2008;57:406–415.
- ,.The ten point clock test: a quick screen and grading method for cognitive impairment in medical and surgical patients.Int J Psychiatry Med.1994;24:229–244.
- ,,, et al.Patient safety concerns arising from test results that return after hospital discharge.Ann Intern Med.2005;143:121–128.
- ,,, et al.Role of pharmacist counseling in preventing adverse drug events after hospitalization.Arch Intern Med.2006;166:565–571.
- .SF‐36 health survey update.Spine.2000;25:3130–3139.
- ,.The development, validity and application of a new instrument to assess the quality of discharge planning activities from the community perspective.Int J Qual Health Care.2001;13:109–116.
- ,,.Brief scale measuring patient preparedness for hospital discharge to home: psychometric properties.J Hosp Med.2008;3:446–454.
- ,,.The Satisfaction with Information about Medicines Scale (SIMS): a new measurement tool for audit and research.Qual Health Care.2001;10:135–140.
- ,,.Discharge planning scale: community physicians' perspective.J Hosp Med.2008;3:455–464.
- ,.An introduction to generalized estimating equations and an application to assess selectivity effects in a longitudinal study on very old individuals.J Educ Behav Stat.2004;29:421–437. Available at: http://jeb.sagepub.com/cgi/content/abstract/29/4/421. Accessed June 2009.
- ASTM. E2369‐05 Standard Specification for Continuity of Care Record (CCR). Available at: http://www.astm.org/Standards/E2369.htm. Accessed June2009.
- ,,, et al.Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- ,,.Interventions aimed at reducing problems in adult patients discharged from hospital to home: a systematic meta‐review.BMC Health Serv Res.2007;7:47.
- ,,,,,.Evaluation of a computer‐generated discharge summary for patients with acute coronary syndromes.Br J Gen Pract.1998;48:1163–1164.
- ,,,.Standardized or narrative discharge summaries. Which do family physicians prefer?Can Fam Physician.1998;44:62–69.
- ,,,.Dictated versus database‐generated discharge summaries: a randomized clinical trial.CMAJ.1999;160:319–326.
- ,,,,.The incidence and severity of adverse events affecting patients after discharge from the hospital.Ann Intern Med.2003;138:161–167.
- ,,,,,.Deficits in communication and information transfer between hospital‐based and primary care physicians: implications for patient safety and continuity of care.JAMA.2007;297:831–841.
- ,,,.Discharge planning from hospital to home.Cochrane Database Syst Rev.2004;(1):CD000313.
- ,,.Dissemination of discharge summaries. Not reaching follow‐up physicians.Can Fam Physician.2002;48:737–742.
- ,,.Software design to facilitate information transfer at hospital discharge.Inform Prim Care.2006;14:109–119.
- ,.Computer physician order entry: benefits, costs, and issues.Ann Intern Med.2003;139:31–39.
- ,,, et al.Patient readmissions, emergency visits, and adverse events after software‐assisted discharge from hospital: cluster randomized trial.J Hosp Med.2009; DOI: 10.1002/jhm.459. PMID: 19479782.
- ,,.Predictive validity of a questionnaire that identifies older persons at risk for hospital admission.J Am Geriatr Soc.1995;43:374–377.
- ,.Prediction of early readmission in medical inpatients using the Probability of Repeated Admission instrument.Nurs Res.2008;57:406–415.
- ,.The ten point clock test: a quick screen and grading method for cognitive impairment in medical and surgical patients.Int J Psychiatry Med.1994;24:229–244.
- ,,, et al.Patient safety concerns arising from test results that return after hospital discharge.Ann Intern Med.2005;143:121–128.
- ,,, et al.Role of pharmacist counseling in preventing adverse drug events after hospitalization.Arch Intern Med.2006;166:565–571.
- .SF‐36 health survey update.Spine.2000;25:3130–3139.
- ,.The development, validity and application of a new instrument to assess the quality of discharge planning activities from the community perspective.Int J Qual Health Care.2001;13:109–116.
- ,,.Brief scale measuring patient preparedness for hospital discharge to home: psychometric properties.J Hosp Med.2008;3:446–454.
- ,,.The Satisfaction with Information about Medicines Scale (SIMS): a new measurement tool for audit and research.Qual Health Care.2001;10:135–140.
- ,,.Discharge planning scale: community physicians' perspective.J Hosp Med.2008;3:455–464.
- ,.An introduction to generalized estimating equations and an application to assess selectivity effects in a longitudinal study on very old individuals.J Educ Behav Stat.2004;29:421–437. Available at: http://jeb.sagepub.com/cgi/content/abstract/29/4/421. Accessed June 2009.
- ASTM. E2369‐05 Standard Specification for Continuity of Care Record (CCR). Available at: http://www.astm.org/Standards/E2369.htm. Accessed June2009.
- ,,, et al.Transition of care for hospitalized elderly patients—development of a discharge checklist for hospitalists.J Hosp Med.2006;1:354–360.
- ,,.Interventions aimed at reducing problems in adult patients discharged from hospital to home: a systematic meta‐review.BMC Health Serv Res.2007;7:47.
- ,,,,,.Evaluation of a computer‐generated discharge summary for patients with acute coronary syndromes.Br J Gen Pract.1998;48:1163–1164.
- ,,,.Standardized or narrative discharge summaries. Which do family physicians prefer?Can Fam Physician.1998;44:62–69.
- ,,,.Dictated versus database‐generated discharge summaries: a randomized clinical trial.CMAJ.1999;160:319–326.
Copyright © 2009 Society of Hospital Medicine
H1N1 Pandemic Should Increase Adult Vaccination Awareness
The H1N1 pandemic has contributed to more than 200 U.S. deaths and the Center for Disease Control reports more than 37,000 probable or confirmed cases nationwide, which highlights the need for increased education for adult immunizations.
The H1N1 vaccination is expected to be available this fall and has attracted the attention of inpatient and outpatient physicians alike, says hospitalist and infectious-disease specialist John J. Ross, MD, of Brigham and Women's Hospital in Boston. Dr. Ross expects a spike in adult vaccinations similar to the increase witnessed after the Sept. 11 attacks. "You saw an increase in vaccinations because of increased panic," he says. "I expect that to be the same this fall with the introduction of the swine flu shot.”
The American College of Physicians (ACP) and the Infectious Diseases Society of America (IDSA) also express the need for heightened awareness. The medical groups recently issued a joint statement calling for a "renewed and stronger" emphasis on adult vaccinations. The statement, also supported by SHM, emphasizes an increase in patient education and documentation. It suggests hospital workers review and update their own immunizations, with particular attention to annual influenza immunizations.
Though most vaccinations are administered in the outpatient setting, hospitalists are required to update their patients’ vaccination status. "While hospitalists are doing a good job of handling pneumonia and influenza vaccinations, they could do a better job of handling some of the less common disease vaccinations,” says Ross.
Dr. Ross says hospitalists should remain knowledgeable in all of the immunization categories, a complete list of which is available at the CDC Web site. "The more knowledgeable the hospitalist is on vaccinations, the more they can educate their patients," he says.
The H1N1 pandemic has contributed to more than 200 U.S. deaths and the Center for Disease Control reports more than 37,000 probable or confirmed cases nationwide, which highlights the need for increased education for adult immunizations.
The H1N1 vaccination is expected to be available this fall and has attracted the attention of inpatient and outpatient physicians alike, says hospitalist and infectious-disease specialist John J. Ross, MD, of Brigham and Women's Hospital in Boston. Dr. Ross expects a spike in adult vaccinations similar to the increase witnessed after the Sept. 11 attacks. "You saw an increase in vaccinations because of increased panic," he says. "I expect that to be the same this fall with the introduction of the swine flu shot.”
The American College of Physicians (ACP) and the Infectious Diseases Society of America (IDSA) also express the need for heightened awareness. The medical groups recently issued a joint statement calling for a "renewed and stronger" emphasis on adult vaccinations. The statement, also supported by SHM, emphasizes an increase in patient education and documentation. It suggests hospital workers review and update their own immunizations, with particular attention to annual influenza immunizations.
Though most vaccinations are administered in the outpatient setting, hospitalists are required to update their patients’ vaccination status. "While hospitalists are doing a good job of handling pneumonia and influenza vaccinations, they could do a better job of handling some of the less common disease vaccinations,” says Ross.
Dr. Ross says hospitalists should remain knowledgeable in all of the immunization categories, a complete list of which is available at the CDC Web site. "The more knowledgeable the hospitalist is on vaccinations, the more they can educate their patients," he says.
The H1N1 pandemic has contributed to more than 200 U.S. deaths and the Center for Disease Control reports more than 37,000 probable or confirmed cases nationwide, which highlights the need for increased education for adult immunizations.
The H1N1 vaccination is expected to be available this fall and has attracted the attention of inpatient and outpatient physicians alike, says hospitalist and infectious-disease specialist John J. Ross, MD, of Brigham and Women's Hospital in Boston. Dr. Ross expects a spike in adult vaccinations similar to the increase witnessed after the Sept. 11 attacks. "You saw an increase in vaccinations because of increased panic," he says. "I expect that to be the same this fall with the introduction of the swine flu shot.”
The American College of Physicians (ACP) and the Infectious Diseases Society of America (IDSA) also express the need for heightened awareness. The medical groups recently issued a joint statement calling for a "renewed and stronger" emphasis on adult vaccinations. The statement, also supported by SHM, emphasizes an increase in patient education and documentation. It suggests hospital workers review and update their own immunizations, with particular attention to annual influenza immunizations.
Though most vaccinations are administered in the outpatient setting, hospitalists are required to update their patients’ vaccination status. "While hospitalists are doing a good job of handling pneumonia and influenza vaccinations, they could do a better job of handling some of the less common disease vaccinations,” says Ross.
Dr. Ross says hospitalists should remain knowledgeable in all of the immunization categories, a complete list of which is available at the CDC Web site. "The more knowledgeable the hospitalist is on vaccinations, the more they can educate their patients," he says.