User login
What are the adverse effects of prolonged opioid use in patients with chronic pain?
CONSTIPATION, NAUSEA, AND DYSPEPSIA are the most common long-term adverse effects of chronic opioid use (strength of recommendation [SOR]: B, systematic review of low-quality studies). Men may experience depression, fatigue, and sexual dysfunction (SOR: B, 2 observational studies). Prolonged use of opioids also may increase sensitivity to pain (SOR: C, review of case reports and case series). (This review does not address drug seeking or drug escalating.)
Patients on long-term methadone are at risk for cardiac arrhythmias caused by prolonged QT intervals and torsades de pointes (SOR: C, case reports).
Patients taking buprenorphine for opioid dependence may experience acute hepatitis (SOR: C, 1 case report).
Evidence summary
Chronic pain is usually defined as pain persisting longer than 3 months. Evidence of the efficacy of opioids for noncancer pain has led to increased opioid prescribing over the past 20 years and with it, growing concern about adverse effects from long-term use.1
Nausea, constipation, dyspepsia lead side-effects parade
A Cochrane systematic review of 26 studies (25 observational studies and 1 randomized controlled trial [RCT]) of adults who had taken opioids for noncancer pain for at least 6 months assessed the adverse effects of long-term opioid therapy.2 Although the authors couldn’t quantify the incidence of adverse effects because of inconsistent reporting and definition of effects, they stated that the most common complications were nausea, constipation, and dyspepsia. The review found that 22.9% of patients (95% confidence interval [CI], 15.3-32.8) discontinued oral opioids because of adverse effects.
A cross-sectional observational study evaluated self-reported adverse effects in 889 patients who received opioid therapy for noncancer pain lasting at least 3 months.3 Forty percent of patients reported constipation and 18% sexual dysfunction. Patients taking opioids daily experienced more constipation than patients taking the drugs intermittently (39% vs 24%; number needed to harm [NNH]=7; P<.05).
Sexual dysfunction, fatigue, depression aren’t far behind
A case-control study of 20 male cancer survivors with neuropathic pain who took 200 mg of morphine-equivalent daily for a year found that 90% of patients in the opioid group experienced hypogonadism with symptoms of sexual dysfunction, fatigue, and depression, compared with 40% of the 20 controls (NNH=2; 95% CI, 1-5).4
A case-controlled observational study of 54 men with noncancer pain who took opioids for 1 year found that 39 of 45 men who had normal erectile function before opioid therapy reported severe erectile dysfunction while taking the drugs.5 Levels of testosterone and estradiol were significantly lower (P<.0001) in the men taking opioids than the 27 opioid-free controls.
Potentially fatal arrhythmias are a risk for some patients
From 1969 to 2002, 59 cases of QT prolongation or torsades de pointes in methadone users, 5 (8.5%) of them fatal, were reported to the US Food and Drug Administration’s Medwatch Database.6 The mean daily methadone dose was 410 mg (median dose 345 mg, range 29-1680 mg). Length of therapy was not reported. In 44 (75%) of reported cases, patients had other known risks for QT prolongation or torsades de pointes, including female sex, interacting medications, potassium or magnesium abnormalities, and structural heart disease.
Buprenorphine may cause acute hepatitis
No apparent long-term hepatic adverse effects are associated with chronic opioid use. However, a 2004 case series described acute cytolytic hepatitis in 7 patients taking buprenorphine, all with hepatitis C and a history of intravenous drug abuse.7 Acute symptoms resolved quickly in all cases, and only 3 patients required a reduction in buprenorphine dosage.
Prolonged use may increase sensitivity to pain
Case reports and case series have found that prolonged use of opioids causes increased sensitivity to pain in some patients, which is difficult to differentiate from opioid tolerance.8
Recommendations
The American Pain Society (APS) recommends anticipating, identifying, and treating opioid-related adverse effects such as constipation or nausea.1 APS advises against using opioid antagonists to prevent or treat bowel dysfunction, and encourages older patients or patients with an increased risk of developing constipation to start a bowel regimen. Patients with complaints suggesting hypogonadism should be tested for hormonal deficiencies.
The Center for Substance Abuse and Treatment recommends obtaining a cardiac history and an electrocardiogram (EKG) on all patients before starting methadone and repeating the EKG at 30 days and annually thereafter to evaluate for QT prolongation.9 Prescribers should also warn patients of the risk of methadone-induced arrhythmias and be aware of interacting medications that prolong the QT interval or reduce methadone elimination.
1. Chou R, Fanciullo GJ, Adler JA, et al. Clinical guidelines for the use of chronic opioid therapy in chronic non-cancer pain. J Pain. 2009;10:113-130.
2. Noble M, Treadwell JR, Tregear SJ, et al. Long-term opioid management for chronic noncancer pain. Cochrane Database Syst Rev. 2010;(1):CD006605.-
3. Brown RT, Zuelsdorff M, Fleming M. Adverse effects and cognitive function among primary care patients taking opioids for chronic nonmalignant pain. J Opioid Manag. 2006;2:137-146.
4. Rajagopal A, Vassilopoulou-Sellin R, Palmer JL, et al. Symptomatic hypogonadism in male survivors of cancer with chronic exposure to opioids. Cancer. 2004;100:851-858.
5. Daniell HW. Hypogonadism in men consuming sustained-action oral opioids. J Pain. 2002;3:377-384.
6. Pearson EC, Woosley RL. QT prolongation and torsades de pointes among methadone users: reports to the FDA spontaneous reporting system. Pharmacoepidemiol Drug Saf. 2005;14:747-753.
7. Hervé S, Riachi G, Noblet C, et al. Acute hepatitis due to buprenorphine administration. Eur J Gastroenterol Hepatol. 2004;16:1033-1037.
8. Ballantyne JC, Mao J. Opioid therapy for chronic pain. N Engl J Med. 2003;20:1943-1953.
9. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
CONSTIPATION, NAUSEA, AND DYSPEPSIA are the most common long-term adverse effects of chronic opioid use (strength of recommendation [SOR]: B, systematic review of low-quality studies). Men may experience depression, fatigue, and sexual dysfunction (SOR: B, 2 observational studies). Prolonged use of opioids also may increase sensitivity to pain (SOR: C, review of case reports and case series). (This review does not address drug seeking or drug escalating.)
Patients on long-term methadone are at risk for cardiac arrhythmias caused by prolonged QT intervals and torsades de pointes (SOR: C, case reports).
Patients taking buprenorphine for opioid dependence may experience acute hepatitis (SOR: C, 1 case report).
Evidence summary
Chronic pain is usually defined as pain persisting longer than 3 months. Evidence of the efficacy of opioids for noncancer pain has led to increased opioid prescribing over the past 20 years and with it, growing concern about adverse effects from long-term use.1
Nausea, constipation, dyspepsia lead side-effects parade
A Cochrane systematic review of 26 studies (25 observational studies and 1 randomized controlled trial [RCT]) of adults who had taken opioids for noncancer pain for at least 6 months assessed the adverse effects of long-term opioid therapy.2 Although the authors couldn’t quantify the incidence of adverse effects because of inconsistent reporting and definition of effects, they stated that the most common complications were nausea, constipation, and dyspepsia. The review found that 22.9% of patients (95% confidence interval [CI], 15.3-32.8) discontinued oral opioids because of adverse effects.
A cross-sectional observational study evaluated self-reported adverse effects in 889 patients who received opioid therapy for noncancer pain lasting at least 3 months.3 Forty percent of patients reported constipation and 18% sexual dysfunction. Patients taking opioids daily experienced more constipation than patients taking the drugs intermittently (39% vs 24%; number needed to harm [NNH]=7; P<.05).
Sexual dysfunction, fatigue, depression aren’t far behind
A case-control study of 20 male cancer survivors with neuropathic pain who took 200 mg of morphine-equivalent daily for a year found that 90% of patients in the opioid group experienced hypogonadism with symptoms of sexual dysfunction, fatigue, and depression, compared with 40% of the 20 controls (NNH=2; 95% CI, 1-5).4
A case-controlled observational study of 54 men with noncancer pain who took opioids for 1 year found that 39 of 45 men who had normal erectile function before opioid therapy reported severe erectile dysfunction while taking the drugs.5 Levels of testosterone and estradiol were significantly lower (P<.0001) in the men taking opioids than the 27 opioid-free controls.
Potentially fatal arrhythmias are a risk for some patients
From 1969 to 2002, 59 cases of QT prolongation or torsades de pointes in methadone users, 5 (8.5%) of them fatal, were reported to the US Food and Drug Administration’s Medwatch Database.6 The mean daily methadone dose was 410 mg (median dose 345 mg, range 29-1680 mg). Length of therapy was not reported. In 44 (75%) of reported cases, patients had other known risks for QT prolongation or torsades de pointes, including female sex, interacting medications, potassium or magnesium abnormalities, and structural heart disease.
Buprenorphine may cause acute hepatitis
No apparent long-term hepatic adverse effects are associated with chronic opioid use. However, a 2004 case series described acute cytolytic hepatitis in 7 patients taking buprenorphine, all with hepatitis C and a history of intravenous drug abuse.7 Acute symptoms resolved quickly in all cases, and only 3 patients required a reduction in buprenorphine dosage.
Prolonged use may increase sensitivity to pain
Case reports and case series have found that prolonged use of opioids causes increased sensitivity to pain in some patients, which is difficult to differentiate from opioid tolerance.8
Recommendations
The American Pain Society (APS) recommends anticipating, identifying, and treating opioid-related adverse effects such as constipation or nausea.1 APS advises against using opioid antagonists to prevent or treat bowel dysfunction, and encourages older patients or patients with an increased risk of developing constipation to start a bowel regimen. Patients with complaints suggesting hypogonadism should be tested for hormonal deficiencies.
The Center for Substance Abuse and Treatment recommends obtaining a cardiac history and an electrocardiogram (EKG) on all patients before starting methadone and repeating the EKG at 30 days and annually thereafter to evaluate for QT prolongation.9 Prescribers should also warn patients of the risk of methadone-induced arrhythmias and be aware of interacting medications that prolong the QT interval or reduce methadone elimination.
CONSTIPATION, NAUSEA, AND DYSPEPSIA are the most common long-term adverse effects of chronic opioid use (strength of recommendation [SOR]: B, systematic review of low-quality studies). Men may experience depression, fatigue, and sexual dysfunction (SOR: B, 2 observational studies). Prolonged use of opioids also may increase sensitivity to pain (SOR: C, review of case reports and case series). (This review does not address drug seeking or drug escalating.)
Patients on long-term methadone are at risk for cardiac arrhythmias caused by prolonged QT intervals and torsades de pointes (SOR: C, case reports).
Patients taking buprenorphine for opioid dependence may experience acute hepatitis (SOR: C, 1 case report).
Evidence summary
Chronic pain is usually defined as pain persisting longer than 3 months. Evidence of the efficacy of opioids for noncancer pain has led to increased opioid prescribing over the past 20 years and with it, growing concern about adverse effects from long-term use.1
Nausea, constipation, dyspepsia lead side-effects parade
A Cochrane systematic review of 26 studies (25 observational studies and 1 randomized controlled trial [RCT]) of adults who had taken opioids for noncancer pain for at least 6 months assessed the adverse effects of long-term opioid therapy.2 Although the authors couldn’t quantify the incidence of adverse effects because of inconsistent reporting and definition of effects, they stated that the most common complications were nausea, constipation, and dyspepsia. The review found that 22.9% of patients (95% confidence interval [CI], 15.3-32.8) discontinued oral opioids because of adverse effects.
A cross-sectional observational study evaluated self-reported adverse effects in 889 patients who received opioid therapy for noncancer pain lasting at least 3 months.3 Forty percent of patients reported constipation and 18% sexual dysfunction. Patients taking opioids daily experienced more constipation than patients taking the drugs intermittently (39% vs 24%; number needed to harm [NNH]=7; P<.05).
Sexual dysfunction, fatigue, depression aren’t far behind
A case-control study of 20 male cancer survivors with neuropathic pain who took 200 mg of morphine-equivalent daily for a year found that 90% of patients in the opioid group experienced hypogonadism with symptoms of sexual dysfunction, fatigue, and depression, compared with 40% of the 20 controls (NNH=2; 95% CI, 1-5).4
A case-controlled observational study of 54 men with noncancer pain who took opioids for 1 year found that 39 of 45 men who had normal erectile function before opioid therapy reported severe erectile dysfunction while taking the drugs.5 Levels of testosterone and estradiol were significantly lower (P<.0001) in the men taking opioids than the 27 opioid-free controls.
Potentially fatal arrhythmias are a risk for some patients
From 1969 to 2002, 59 cases of QT prolongation or torsades de pointes in methadone users, 5 (8.5%) of them fatal, were reported to the US Food and Drug Administration’s Medwatch Database.6 The mean daily methadone dose was 410 mg (median dose 345 mg, range 29-1680 mg). Length of therapy was not reported. In 44 (75%) of reported cases, patients had other known risks for QT prolongation or torsades de pointes, including female sex, interacting medications, potassium or magnesium abnormalities, and structural heart disease.
Buprenorphine may cause acute hepatitis
No apparent long-term hepatic adverse effects are associated with chronic opioid use. However, a 2004 case series described acute cytolytic hepatitis in 7 patients taking buprenorphine, all with hepatitis C and a history of intravenous drug abuse.7 Acute symptoms resolved quickly in all cases, and only 3 patients required a reduction in buprenorphine dosage.
Prolonged use may increase sensitivity to pain
Case reports and case series have found that prolonged use of opioids causes increased sensitivity to pain in some patients, which is difficult to differentiate from opioid tolerance.8
Recommendations
The American Pain Society (APS) recommends anticipating, identifying, and treating opioid-related adverse effects such as constipation or nausea.1 APS advises against using opioid antagonists to prevent or treat bowel dysfunction, and encourages older patients or patients with an increased risk of developing constipation to start a bowel regimen. Patients with complaints suggesting hypogonadism should be tested for hormonal deficiencies.
The Center for Substance Abuse and Treatment recommends obtaining a cardiac history and an electrocardiogram (EKG) on all patients before starting methadone and repeating the EKG at 30 days and annually thereafter to evaluate for QT prolongation.9 Prescribers should also warn patients of the risk of methadone-induced arrhythmias and be aware of interacting medications that prolong the QT interval or reduce methadone elimination.
1. Chou R, Fanciullo GJ, Adler JA, et al. Clinical guidelines for the use of chronic opioid therapy in chronic non-cancer pain. J Pain. 2009;10:113-130.
2. Noble M, Treadwell JR, Tregear SJ, et al. Long-term opioid management for chronic noncancer pain. Cochrane Database Syst Rev. 2010;(1):CD006605.-
3. Brown RT, Zuelsdorff M, Fleming M. Adverse effects and cognitive function among primary care patients taking opioids for chronic nonmalignant pain. J Opioid Manag. 2006;2:137-146.
4. Rajagopal A, Vassilopoulou-Sellin R, Palmer JL, et al. Symptomatic hypogonadism in male survivors of cancer with chronic exposure to opioids. Cancer. 2004;100:851-858.
5. Daniell HW. Hypogonadism in men consuming sustained-action oral opioids. J Pain. 2002;3:377-384.
6. Pearson EC, Woosley RL. QT prolongation and torsades de pointes among methadone users: reports to the FDA spontaneous reporting system. Pharmacoepidemiol Drug Saf. 2005;14:747-753.
7. Hervé S, Riachi G, Noblet C, et al. Acute hepatitis due to buprenorphine administration. Eur J Gastroenterol Hepatol. 2004;16:1033-1037.
8. Ballantyne JC, Mao J. Opioid therapy for chronic pain. N Engl J Med. 2003;20:1943-1953.
9. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
1. Chou R, Fanciullo GJ, Adler JA, et al. Clinical guidelines for the use of chronic opioid therapy in chronic non-cancer pain. J Pain. 2009;10:113-130.
2. Noble M, Treadwell JR, Tregear SJ, et al. Long-term opioid management for chronic noncancer pain. Cochrane Database Syst Rev. 2010;(1):CD006605.-
3. Brown RT, Zuelsdorff M, Fleming M. Adverse effects and cognitive function among primary care patients taking opioids for chronic nonmalignant pain. J Opioid Manag. 2006;2:137-146.
4. Rajagopal A, Vassilopoulou-Sellin R, Palmer JL, et al. Symptomatic hypogonadism in male survivors of cancer with chronic exposure to opioids. Cancer. 2004;100:851-858.
5. Daniell HW. Hypogonadism in men consuming sustained-action oral opioids. J Pain. 2002;3:377-384.
6. Pearson EC, Woosley RL. QT prolongation and torsades de pointes among methadone users: reports to the FDA spontaneous reporting system. Pharmacoepidemiol Drug Saf. 2005;14:747-753.
7. Hervé S, Riachi G, Noblet C, et al. Acute hepatitis due to buprenorphine administration. Eur J Gastroenterol Hepatol. 2004;16:1033-1037.
8. Ballantyne JC, Mao J. Opioid therapy for chronic pain. N Engl J Med. 2003;20:1943-1953.
9. Krantz MJ, Martin J, Stimmel B, et al. QTc interval screening in methadone treatment. Ann Intern Med. 2009;150:387-395.
Evidence-based answers from the Family Physicians Inquiries Network
PPI therapy: When to worry about fracture risk
• For most patients with chronic heartburn and regurgitation, step-down therapy to the lowest effective dose of proton pump inhibitors (PPIs) or treatment with a histamine-2 receptor antagonist (H2RA) is a reasonable, cost-effective approach. A
• Advise elderly patients who require long-term, high-dose PPI therapy to increase their dietary and/or supplemental calcium intake. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE 1 Damian F,* a 39-year-old construction worker who takes omeprazole for chronic gastroesophageal reflux disease (GERD), comes in to request a refill. He’s had several accidents in recent years—he fell off a ladder on one occasion, and went down a flight of stairs on another—but none that resulted in significant trauma. Damian admits that he could better control his GERD symptoms by avoiding spicy and fatty foods, limiting alcohol consumption, and quitting smoking, but takes omeprazole nearly every day instead.
CASE 2 Estella G,* a 71-year-old retiree, has been on continuous proton pump inhibitor (PPI) therapy for chronic GERD and erosive esophagitis for nearly 20 years. The patient is a frail woman (body mass index=19.8 kg/m2) and a former smoker (1½ packs a day), both of which increase her risk of osteoporosis. But she has never had a dual energy x-ray absorptiometry (DEXA) scan.
*These cases are based on real patients in my practice, but their names and details have been changed to protect their identity.
Proton pump inhibitors (PPIs) are one of the most commonly used prescription drug categories in the United States,1 but they have been associated with an increase in fracture risk. A US Food and Drug Administration (FDA) safety update issued in March 2011 noted that there is little problem with the lower doses and shorter duration for which over-the-counter PPIs are intended, but patients who take higher-dose prescription PPIs or take prescription PPIs for more than a year may be at greater risk.2
If Damian and Estella were your patients, would you continue to prescribe PPI therapy or offer them alternatives? How should you treat other patients with chronic upper gastrointestinal (GI) distress? The evidence review that follows can help you answer those questions.
How high is the risk? Evidence is mixed (or lacking)
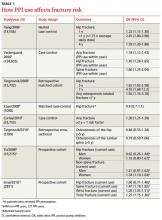
Several retrospective studies have demonstrated a modest increased risk for hip, spine, and wrist fractures in men and women taking PPIs, with the highest risk in patients who have taken higher than standard doses for >4 years.3-6 Concomitant risk factors (alcohol abuse, cigarette smoking, diabetes, and neurologic or renal disease) may increase fracture risk.6 But other retrospective studies, as well as prospective studies, have found no significant increase in fracture risk in patients taking PPIs,7-9 even after 5 years of therapy.7 However, some studies that failed to find an increased risk of osteoporosis with PPI use had a small number of subjects,8,9 resulting in a wide range in confidence intervals.
These findings, based on 6 retrospective case-control, cohort, and cross-sectional studies and 2 prospective cohort studies, are summarized in TABLE 1. No prospective randomized, blinded, controlled trials have examined the potential increased fracture risk associated with PPI use.
Do PPIs interfere with calcium metabolism?
Here, too, the findings are mixed. PPIs are known to inhibit the production and secretion of intragastric hydrochloric acid, which mediates small intestinal absorption of calcium,10 but evidence is conflicting about the role of intragastric hydrochloric acid in calcium absorption. Osteoclasts also have proton pumps, and some researchers have suggested that PPIs have the potential to limit the activity of these proton pumps, leading to reduced bone resorption.11
To date, the only studies that have examined the impact of PPIs on intestinal calcium absorption were limited by the health status of the participants—all either had renal failure and were on hemodialysis or had hypo- or achlorhydria, chronic conditions known to adversely affect calcium metabolism.12 Long-term randomized, double-blinded, placebo-controlled trials are needed to determine whether PPIs adversely affect intestinal calcium absorption and result in bone resorption abnormalities and increased fracture risk.
A closer look at the data
The varying responses associated with PPI dose and duration and the possibility that acid inhibition may decrease calcium absorption support a causal association between PPI use and fracture risk. But the low magnitude of the proposed association (most odds ratios <2) and the lack of data assessing potentially confounding factors limit evidence of causality.3,5,6,9 One key limitation of the earlier studies is that they were not designed to define the specific mechanism underlying the association between PPI therapy and fracture risk.
Older studies suggest a causal relationship
Two case-control studies3,4 found a causal association between PPI use and fracture risk, but one of them failed to identify either a dose-response or a duration-response effect.4 And neither study was designed to define underlying mechanisms to explain the potential association between fracture risk and PPI therapy.
A retrospective matched cohort study5 found an increase in the overall risk of fracture among patients with ≥7 years of PPI therapy and an in-creased risk of hip fracture with ≥5 years of therapy, but short-term risk of fracture was not found to be significant. The results of this study suggest that the risk of osteoporotic fracture increases with duration of exposure to PPI therapy, but not in a dose-dependent fashion.
Newer data are less worrisome
The results of a retrospective cross-sectional trial, published last year, are more reassuring. The researchers determined via univariate analysis that PPI use was associated with a lower risk of osteoporosis, both at the lumbar spine (for all levels of PPI use) and the hip (in patients who had taken more than 1500 standard PPI doses over the previous 5 years).7
This finding—that increasing intensity (both longer duration and higher dosage) of PPI exposure is not associated with an increased risk of osteoporosis—contrasts with results of the authors’ earlier study.5 This may be because they monitored annualized changes in BMD and were able to detect significant changes in other medications participants were taking that might affect bone loss or gain. That allowed them to validate their findings regarding a lack of true association between bone loss and PPI use, the authors reported.
A matched, nested case-control trial8 determined that the use of PPIs does not increase the risk of hip fracture in patients without associated major risk factors (ie, alcohol dependence, underlying neurologic disease, accidental falls, and senility). The researchers suggested that the difference between their findings and those of an earlier nested case-control study3 could mean that the increased risk of hip fracture found in the older study occurred only among PPI users with definable risk factors for hip fracture.
Recent results from the Women’s Health Initiative (WHI) suggest that in postmenopausal women, PPI use is not associated with hip fractures. The WHI did, however, find a modest association between PPI use and clinical spine, forearm, or wrist fracture, as well as total fractures.13 Compared with previous trials, this large cohort study had a large number of fracture events and assessed confounding factors that had not been addressed, including calcium intake. It also was the first trial to assess associations between BMD and fracture risk relative to PPI dosing. Although no specific conclusion was reported, the researchers did not find evidence of dose dependence.
A reasonable approach to PPI use
A consensus statement from the FDA2 and the authors of 2 meta-analyses14,15 recommend that PPIs be used only for appropriate indications—GERD, peptic ulcer disease, dyspepsia, and treatment of Helicobacter pylori—and not in higher doses or for longer periods than are necessary to achieve the desired results.
Whenever possible, implement step-down therapy to the lowest effective dose or prescribe an H2RA rather than a PPI. Both are cost-effective ways to treat most patients with upper GI symptoms.2 It is important, too, to advise elderly patients who require long-term, high-dose PPI therapy to increase their dietary and/or supplemental calcium intake, to recommend DEXA scans for individuals at risk for osteoporosis, and to counsel patients who suffer from GI distress to avoid foods that are known to exacerbate symptoms (TABLE 2).16
TABLE 2
GERD and diet: Foods that worsen symptoms16
| Alcohol |
| Caffeine-containing beverages |
| Citrus fruits |
| Chocolate |
| Fried and fatty foods |
| Garlic and onions |
| Mint flavorings |
| Spicy foods |
| Tomato-based foods (eg, chili, pizza, spaghetti sauce, salsa) |
CASE 1 Damian
You talk to Damian about the association between prolonged PPI therapy and fracture risk and stress the need for dietary changes and lifestyle modifications, particularly smoking cessation. On a return visit several months later, he reports that he has stopped smoking and cut way back on alcohol consumption, and eats fast food less frequently. As a result, he no longer requires chronic use of PPI therapy, and now takes omeprazole only when he has symptoms of GERD—usually, after indulging in fried or fatty foods.
CASE 2 Estella
Estella has severe GERD and erosive esophagitis and will probably need lifelong PPI therapy to adequately control her symptoms. After a detailed discussion of potential risks vs benefits of PPIs, she agrees to a DEXA scan to evaluate for osteoporosis. Her test results show osteopenia in the lumbar spine and femoral neck, but no evidence of osteoporosis. You advise her to increase her consumption of calcium and to undergo DEXA scanning in another 2 years.
CORRESPONDENCE
Joel J. Heidelbaugh, MD, FAAFP, FACG, Ypsilanti Health Center, 200 Arnet Suite 200, Ypsilanti, MI 48198; jheidel@med.umich.edu
1. Bartholow M. Top 200 prescription drugs of 2009. May 11, 2010. Pharmacy Times. Available at: http://www.pharmacytimes. http://www.pharmacytimes.com/publications/issue/2010/May2010/RxFocusTopDrugs-0510. Accessed April 8, 2011.
2. US Food and Drug Administration. FDA Drug Safety Communication: Possible increased risk of fractures of the hip, wrist, and spine with the use of proton pump inhibitors. May 25, 2010; March 23, 2011 update. Available at: http://www.fda.gov/Drugs/DrugSafety/postmarketdrugsafetyInformationforpatientsandproviders/ucm213206.htm#SafetyAnnouncement. Accessed March 24, 2011.
3. Yang YX, Lewis JD, Epstein S, et al. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296:2947-2953.
4. Vestergaard P, Rejnmark L, Mosekilde L. Proton pump inhibitors, histamine h(2) receptor antagonists, and other antacid medications and the risk of fracture. Calcif Tissue Int. 2006;79:76-83.
5. Targownik LE, Lix LM, Metge CJ. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ. 2008;179:319-326.
6. Corley DA, Kubo A, Zhao W, et al. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology. 2010;139:93-101.
7. Targownik LE, Lix LM, Leung S, et al. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology. 2010;138:896-904.
8. Kaye JA, Jick H. Proton pump inhibitor use and risk of hip fractures in patients without major risk factors. Pharmacotherapy. 2008;28:951-959.
9. Yu EW, Blackwell T, Ensrud KE, et al. Acid-suppressive medications and risk of bone loss and fracture in older adults. Calcif Tissue Int. 2008;83:251-259.
10. Bo-Linn GW, Davis GR, Buddrus DJ, et al. An evaluation of the importance of gastric acid secretion in the absorption of dietary calcium. J Clin Invest. 1984;73:640-647.
11. Farina C, Gagliardi S. Selective inhibition of osteoclast vacuolar H+-ATPase. Curr Pharm Des. 2002;8:2033-2048.
12. Insogna KL. The effect of proton pump-inhibiting drugs on mineral metabolism. Am J Gastroenterol. 2009;104(suppl 2):S2-S4.
13. Gray SL, LaCroix AZ, Larson L, et al. Proton pump inhibitor use, hip fracture, and change in bone mineral density in postmenopausal women. Arch Intern Med. 2010;170:765-771.
14. Laine L. Proton pump inhibitors and bone fractures? Am J Gastroenterol. 2009;104(suppl 2):S21-S26.
15. Heidelbaugh JJ, Goldberg KL, Inadomi JM. Overutilization of proton pump inhibitors: a review of cost-effectiveness and risk. Am J Gastroenterol.;2009;104(suppl 2):S27-S32.
16. National Digestive Diseases Information Clearinghouse. Heartburn, gastroesophageal reflux (GER), and gastroesophageal reflux disease (GERD). Available at: http://digestive.niddk.nih.gov/ddiseases/pubs/gerd. Accessed April 18, 2011.
• For most patients with chronic heartburn and regurgitation, step-down therapy to the lowest effective dose of proton pump inhibitors (PPIs) or treatment with a histamine-2 receptor antagonist (H2RA) is a reasonable, cost-effective approach. A
• Advise elderly patients who require long-term, high-dose PPI therapy to increase their dietary and/or supplemental calcium intake. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE 1 Damian F,* a 39-year-old construction worker who takes omeprazole for chronic gastroesophageal reflux disease (GERD), comes in to request a refill. He’s had several accidents in recent years—he fell off a ladder on one occasion, and went down a flight of stairs on another—but none that resulted in significant trauma. Damian admits that he could better control his GERD symptoms by avoiding spicy and fatty foods, limiting alcohol consumption, and quitting smoking, but takes omeprazole nearly every day instead.
CASE 2 Estella G,* a 71-year-old retiree, has been on continuous proton pump inhibitor (PPI) therapy for chronic GERD and erosive esophagitis for nearly 20 years. The patient is a frail woman (body mass index=19.8 kg/m2) and a former smoker (1½ packs a day), both of which increase her risk of osteoporosis. But she has never had a dual energy x-ray absorptiometry (DEXA) scan.
*These cases are based on real patients in my practice, but their names and details have been changed to protect their identity.
Proton pump inhibitors (PPIs) are one of the most commonly used prescription drug categories in the United States,1 but they have been associated with an increase in fracture risk. A US Food and Drug Administration (FDA) safety update issued in March 2011 noted that there is little problem with the lower doses and shorter duration for which over-the-counter PPIs are intended, but patients who take higher-dose prescription PPIs or take prescription PPIs for more than a year may be at greater risk.2
If Damian and Estella were your patients, would you continue to prescribe PPI therapy or offer them alternatives? How should you treat other patients with chronic upper gastrointestinal (GI) distress? The evidence review that follows can help you answer those questions.
How high is the risk? Evidence is mixed (or lacking)
Several retrospective studies have demonstrated a modest increased risk for hip, spine, and wrist fractures in men and women taking PPIs, with the highest risk in patients who have taken higher than standard doses for >4 years.3-6 Concomitant risk factors (alcohol abuse, cigarette smoking, diabetes, and neurologic or renal disease) may increase fracture risk.6 But other retrospective studies, as well as prospective studies, have found no significant increase in fracture risk in patients taking PPIs,7-9 even after 5 years of therapy.7 However, some studies that failed to find an increased risk of osteoporosis with PPI use had a small number of subjects,8,9 resulting in a wide range in confidence intervals.
These findings, based on 6 retrospective case-control, cohort, and cross-sectional studies and 2 prospective cohort studies, are summarized in TABLE 1. No prospective randomized, blinded, controlled trials have examined the potential increased fracture risk associated with PPI use.
Do PPIs interfere with calcium metabolism?
Here, too, the findings are mixed. PPIs are known to inhibit the production and secretion of intragastric hydrochloric acid, which mediates small intestinal absorption of calcium,10 but evidence is conflicting about the role of intragastric hydrochloric acid in calcium absorption. Osteoclasts also have proton pumps, and some researchers have suggested that PPIs have the potential to limit the activity of these proton pumps, leading to reduced bone resorption.11
To date, the only studies that have examined the impact of PPIs on intestinal calcium absorption were limited by the health status of the participants—all either had renal failure and were on hemodialysis or had hypo- or achlorhydria, chronic conditions known to adversely affect calcium metabolism.12 Long-term randomized, double-blinded, placebo-controlled trials are needed to determine whether PPIs adversely affect intestinal calcium absorption and result in bone resorption abnormalities and increased fracture risk.
A closer look at the data
The varying responses associated with PPI dose and duration and the possibility that acid inhibition may decrease calcium absorption support a causal association between PPI use and fracture risk. But the low magnitude of the proposed association (most odds ratios <2) and the lack of data assessing potentially confounding factors limit evidence of causality.3,5,6,9 One key limitation of the earlier studies is that they were not designed to define the specific mechanism underlying the association between PPI therapy and fracture risk.
Older studies suggest a causal relationship
Two case-control studies3,4 found a causal association between PPI use and fracture risk, but one of them failed to identify either a dose-response or a duration-response effect.4 And neither study was designed to define underlying mechanisms to explain the potential association between fracture risk and PPI therapy.
A retrospective matched cohort study5 found an increase in the overall risk of fracture among patients with ≥7 years of PPI therapy and an in-creased risk of hip fracture with ≥5 years of therapy, but short-term risk of fracture was not found to be significant. The results of this study suggest that the risk of osteoporotic fracture increases with duration of exposure to PPI therapy, but not in a dose-dependent fashion.
Newer data are less worrisome
The results of a retrospective cross-sectional trial, published last year, are more reassuring. The researchers determined via univariate analysis that PPI use was associated with a lower risk of osteoporosis, both at the lumbar spine (for all levels of PPI use) and the hip (in patients who had taken more than 1500 standard PPI doses over the previous 5 years).7
This finding—that increasing intensity (both longer duration and higher dosage) of PPI exposure is not associated with an increased risk of osteoporosis—contrasts with results of the authors’ earlier study.5 This may be because they monitored annualized changes in BMD and were able to detect significant changes in other medications participants were taking that might affect bone loss or gain. That allowed them to validate their findings regarding a lack of true association between bone loss and PPI use, the authors reported.
A matched, nested case-control trial8 determined that the use of PPIs does not increase the risk of hip fracture in patients without associated major risk factors (ie, alcohol dependence, underlying neurologic disease, accidental falls, and senility). The researchers suggested that the difference between their findings and those of an earlier nested case-control study3 could mean that the increased risk of hip fracture found in the older study occurred only among PPI users with definable risk factors for hip fracture.
Recent results from the Women’s Health Initiative (WHI) suggest that in postmenopausal women, PPI use is not associated with hip fractures. The WHI did, however, find a modest association between PPI use and clinical spine, forearm, or wrist fracture, as well as total fractures.13 Compared with previous trials, this large cohort study had a large number of fracture events and assessed confounding factors that had not been addressed, including calcium intake. It also was the first trial to assess associations between BMD and fracture risk relative to PPI dosing. Although no specific conclusion was reported, the researchers did not find evidence of dose dependence.
A reasonable approach to PPI use
A consensus statement from the FDA2 and the authors of 2 meta-analyses14,15 recommend that PPIs be used only for appropriate indications—GERD, peptic ulcer disease, dyspepsia, and treatment of Helicobacter pylori—and not in higher doses or for longer periods than are necessary to achieve the desired results.
Whenever possible, implement step-down therapy to the lowest effective dose or prescribe an H2RA rather than a PPI. Both are cost-effective ways to treat most patients with upper GI symptoms.2 It is important, too, to advise elderly patients who require long-term, high-dose PPI therapy to increase their dietary and/or supplemental calcium intake, to recommend DEXA scans for individuals at risk for osteoporosis, and to counsel patients who suffer from GI distress to avoid foods that are known to exacerbate symptoms (TABLE 2).16
TABLE 2
GERD and diet: Foods that worsen symptoms16
| Alcohol |
| Caffeine-containing beverages |
| Citrus fruits |
| Chocolate |
| Fried and fatty foods |
| Garlic and onions |
| Mint flavorings |
| Spicy foods |
| Tomato-based foods (eg, chili, pizza, spaghetti sauce, salsa) |
CASE 1 Damian
You talk to Damian about the association between prolonged PPI therapy and fracture risk and stress the need for dietary changes and lifestyle modifications, particularly smoking cessation. On a return visit several months later, he reports that he has stopped smoking and cut way back on alcohol consumption, and eats fast food less frequently. As a result, he no longer requires chronic use of PPI therapy, and now takes omeprazole only when he has symptoms of GERD—usually, after indulging in fried or fatty foods.
CASE 2 Estella
Estella has severe GERD and erosive esophagitis and will probably need lifelong PPI therapy to adequately control her symptoms. After a detailed discussion of potential risks vs benefits of PPIs, she agrees to a DEXA scan to evaluate for osteoporosis. Her test results show osteopenia in the lumbar spine and femoral neck, but no evidence of osteoporosis. You advise her to increase her consumption of calcium and to undergo DEXA scanning in another 2 years.
CORRESPONDENCE
Joel J. Heidelbaugh, MD, FAAFP, FACG, Ypsilanti Health Center, 200 Arnet Suite 200, Ypsilanti, MI 48198; jheidel@med.umich.edu
• For most patients with chronic heartburn and regurgitation, step-down therapy to the lowest effective dose of proton pump inhibitors (PPIs) or treatment with a histamine-2 receptor antagonist (H2RA) is a reasonable, cost-effective approach. A
• Advise elderly patients who require long-term, high-dose PPI therapy to increase their dietary and/or supplemental calcium intake. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE 1 Damian F,* a 39-year-old construction worker who takes omeprazole for chronic gastroesophageal reflux disease (GERD), comes in to request a refill. He’s had several accidents in recent years—he fell off a ladder on one occasion, and went down a flight of stairs on another—but none that resulted in significant trauma. Damian admits that he could better control his GERD symptoms by avoiding spicy and fatty foods, limiting alcohol consumption, and quitting smoking, but takes omeprazole nearly every day instead.
CASE 2 Estella G,* a 71-year-old retiree, has been on continuous proton pump inhibitor (PPI) therapy for chronic GERD and erosive esophagitis for nearly 20 years. The patient is a frail woman (body mass index=19.8 kg/m2) and a former smoker (1½ packs a day), both of which increase her risk of osteoporosis. But she has never had a dual energy x-ray absorptiometry (DEXA) scan.
*These cases are based on real patients in my practice, but their names and details have been changed to protect their identity.
Proton pump inhibitors (PPIs) are one of the most commonly used prescription drug categories in the United States,1 but they have been associated with an increase in fracture risk. A US Food and Drug Administration (FDA) safety update issued in March 2011 noted that there is little problem with the lower doses and shorter duration for which over-the-counter PPIs are intended, but patients who take higher-dose prescription PPIs or take prescription PPIs for more than a year may be at greater risk.2
If Damian and Estella were your patients, would you continue to prescribe PPI therapy or offer them alternatives? How should you treat other patients with chronic upper gastrointestinal (GI) distress? The evidence review that follows can help you answer those questions.
How high is the risk? Evidence is mixed (or lacking)
Several retrospective studies have demonstrated a modest increased risk for hip, spine, and wrist fractures in men and women taking PPIs, with the highest risk in patients who have taken higher than standard doses for >4 years.3-6 Concomitant risk factors (alcohol abuse, cigarette smoking, diabetes, and neurologic or renal disease) may increase fracture risk.6 But other retrospective studies, as well as prospective studies, have found no significant increase in fracture risk in patients taking PPIs,7-9 even after 5 years of therapy.7 However, some studies that failed to find an increased risk of osteoporosis with PPI use had a small number of subjects,8,9 resulting in a wide range in confidence intervals.
These findings, based on 6 retrospective case-control, cohort, and cross-sectional studies and 2 prospective cohort studies, are summarized in TABLE 1. No prospective randomized, blinded, controlled trials have examined the potential increased fracture risk associated with PPI use.
Do PPIs interfere with calcium metabolism?
Here, too, the findings are mixed. PPIs are known to inhibit the production and secretion of intragastric hydrochloric acid, which mediates small intestinal absorption of calcium,10 but evidence is conflicting about the role of intragastric hydrochloric acid in calcium absorption. Osteoclasts also have proton pumps, and some researchers have suggested that PPIs have the potential to limit the activity of these proton pumps, leading to reduced bone resorption.11
To date, the only studies that have examined the impact of PPIs on intestinal calcium absorption were limited by the health status of the participants—all either had renal failure and were on hemodialysis or had hypo- or achlorhydria, chronic conditions known to adversely affect calcium metabolism.12 Long-term randomized, double-blinded, placebo-controlled trials are needed to determine whether PPIs adversely affect intestinal calcium absorption and result in bone resorption abnormalities and increased fracture risk.
A closer look at the data
The varying responses associated with PPI dose and duration and the possibility that acid inhibition may decrease calcium absorption support a causal association between PPI use and fracture risk. But the low magnitude of the proposed association (most odds ratios <2) and the lack of data assessing potentially confounding factors limit evidence of causality.3,5,6,9 One key limitation of the earlier studies is that they were not designed to define the specific mechanism underlying the association between PPI therapy and fracture risk.
Older studies suggest a causal relationship
Two case-control studies3,4 found a causal association between PPI use and fracture risk, but one of them failed to identify either a dose-response or a duration-response effect.4 And neither study was designed to define underlying mechanisms to explain the potential association between fracture risk and PPI therapy.
A retrospective matched cohort study5 found an increase in the overall risk of fracture among patients with ≥7 years of PPI therapy and an in-creased risk of hip fracture with ≥5 years of therapy, but short-term risk of fracture was not found to be significant. The results of this study suggest that the risk of osteoporotic fracture increases with duration of exposure to PPI therapy, but not in a dose-dependent fashion.
Newer data are less worrisome
The results of a retrospective cross-sectional trial, published last year, are more reassuring. The researchers determined via univariate analysis that PPI use was associated with a lower risk of osteoporosis, both at the lumbar spine (for all levels of PPI use) and the hip (in patients who had taken more than 1500 standard PPI doses over the previous 5 years).7
This finding—that increasing intensity (both longer duration and higher dosage) of PPI exposure is not associated with an increased risk of osteoporosis—contrasts with results of the authors’ earlier study.5 This may be because they monitored annualized changes in BMD and were able to detect significant changes in other medications participants were taking that might affect bone loss or gain. That allowed them to validate their findings regarding a lack of true association between bone loss and PPI use, the authors reported.
A matched, nested case-control trial8 determined that the use of PPIs does not increase the risk of hip fracture in patients without associated major risk factors (ie, alcohol dependence, underlying neurologic disease, accidental falls, and senility). The researchers suggested that the difference between their findings and those of an earlier nested case-control study3 could mean that the increased risk of hip fracture found in the older study occurred only among PPI users with definable risk factors for hip fracture.
Recent results from the Women’s Health Initiative (WHI) suggest that in postmenopausal women, PPI use is not associated with hip fractures. The WHI did, however, find a modest association between PPI use and clinical spine, forearm, or wrist fracture, as well as total fractures.13 Compared with previous trials, this large cohort study had a large number of fracture events and assessed confounding factors that had not been addressed, including calcium intake. It also was the first trial to assess associations between BMD and fracture risk relative to PPI dosing. Although no specific conclusion was reported, the researchers did not find evidence of dose dependence.
A reasonable approach to PPI use
A consensus statement from the FDA2 and the authors of 2 meta-analyses14,15 recommend that PPIs be used only for appropriate indications—GERD, peptic ulcer disease, dyspepsia, and treatment of Helicobacter pylori—and not in higher doses or for longer periods than are necessary to achieve the desired results.
Whenever possible, implement step-down therapy to the lowest effective dose or prescribe an H2RA rather than a PPI. Both are cost-effective ways to treat most patients with upper GI symptoms.2 It is important, too, to advise elderly patients who require long-term, high-dose PPI therapy to increase their dietary and/or supplemental calcium intake, to recommend DEXA scans for individuals at risk for osteoporosis, and to counsel patients who suffer from GI distress to avoid foods that are known to exacerbate symptoms (TABLE 2).16
TABLE 2
GERD and diet: Foods that worsen symptoms16
| Alcohol |
| Caffeine-containing beverages |
| Citrus fruits |
| Chocolate |
| Fried and fatty foods |
| Garlic and onions |
| Mint flavorings |
| Spicy foods |
| Tomato-based foods (eg, chili, pizza, spaghetti sauce, salsa) |
CASE 1 Damian
You talk to Damian about the association between prolonged PPI therapy and fracture risk and stress the need for dietary changes and lifestyle modifications, particularly smoking cessation. On a return visit several months later, he reports that he has stopped smoking and cut way back on alcohol consumption, and eats fast food less frequently. As a result, he no longer requires chronic use of PPI therapy, and now takes omeprazole only when he has symptoms of GERD—usually, after indulging in fried or fatty foods.
CASE 2 Estella
Estella has severe GERD and erosive esophagitis and will probably need lifelong PPI therapy to adequately control her symptoms. After a detailed discussion of potential risks vs benefits of PPIs, she agrees to a DEXA scan to evaluate for osteoporosis. Her test results show osteopenia in the lumbar spine and femoral neck, but no evidence of osteoporosis. You advise her to increase her consumption of calcium and to undergo DEXA scanning in another 2 years.
CORRESPONDENCE
Joel J. Heidelbaugh, MD, FAAFP, FACG, Ypsilanti Health Center, 200 Arnet Suite 200, Ypsilanti, MI 48198; jheidel@med.umich.edu
1. Bartholow M. Top 200 prescription drugs of 2009. May 11, 2010. Pharmacy Times. Available at: http://www.pharmacytimes. http://www.pharmacytimes.com/publications/issue/2010/May2010/RxFocusTopDrugs-0510. Accessed April 8, 2011.
2. US Food and Drug Administration. FDA Drug Safety Communication: Possible increased risk of fractures of the hip, wrist, and spine with the use of proton pump inhibitors. May 25, 2010; March 23, 2011 update. Available at: http://www.fda.gov/Drugs/DrugSafety/postmarketdrugsafetyInformationforpatientsandproviders/ucm213206.htm#SafetyAnnouncement. Accessed March 24, 2011.
3. Yang YX, Lewis JD, Epstein S, et al. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296:2947-2953.
4. Vestergaard P, Rejnmark L, Mosekilde L. Proton pump inhibitors, histamine h(2) receptor antagonists, and other antacid medications and the risk of fracture. Calcif Tissue Int. 2006;79:76-83.
5. Targownik LE, Lix LM, Metge CJ. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ. 2008;179:319-326.
6. Corley DA, Kubo A, Zhao W, et al. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology. 2010;139:93-101.
7. Targownik LE, Lix LM, Leung S, et al. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology. 2010;138:896-904.
8. Kaye JA, Jick H. Proton pump inhibitor use and risk of hip fractures in patients without major risk factors. Pharmacotherapy. 2008;28:951-959.
9. Yu EW, Blackwell T, Ensrud KE, et al. Acid-suppressive medications and risk of bone loss and fracture in older adults. Calcif Tissue Int. 2008;83:251-259.
10. Bo-Linn GW, Davis GR, Buddrus DJ, et al. An evaluation of the importance of gastric acid secretion in the absorption of dietary calcium. J Clin Invest. 1984;73:640-647.
11. Farina C, Gagliardi S. Selective inhibition of osteoclast vacuolar H+-ATPase. Curr Pharm Des. 2002;8:2033-2048.
12. Insogna KL. The effect of proton pump-inhibiting drugs on mineral metabolism. Am J Gastroenterol. 2009;104(suppl 2):S2-S4.
13. Gray SL, LaCroix AZ, Larson L, et al. Proton pump inhibitor use, hip fracture, and change in bone mineral density in postmenopausal women. Arch Intern Med. 2010;170:765-771.
14. Laine L. Proton pump inhibitors and bone fractures? Am J Gastroenterol. 2009;104(suppl 2):S21-S26.
15. Heidelbaugh JJ, Goldberg KL, Inadomi JM. Overutilization of proton pump inhibitors: a review of cost-effectiveness and risk. Am J Gastroenterol.;2009;104(suppl 2):S27-S32.
16. National Digestive Diseases Information Clearinghouse. Heartburn, gastroesophageal reflux (GER), and gastroesophageal reflux disease (GERD). Available at: http://digestive.niddk.nih.gov/ddiseases/pubs/gerd. Accessed April 18, 2011.
1. Bartholow M. Top 200 prescription drugs of 2009. May 11, 2010. Pharmacy Times. Available at: http://www.pharmacytimes. http://www.pharmacytimes.com/publications/issue/2010/May2010/RxFocusTopDrugs-0510. Accessed April 8, 2011.
2. US Food and Drug Administration. FDA Drug Safety Communication: Possible increased risk of fractures of the hip, wrist, and spine with the use of proton pump inhibitors. May 25, 2010; March 23, 2011 update. Available at: http://www.fda.gov/Drugs/DrugSafety/postmarketdrugsafetyInformationforpatientsandproviders/ucm213206.htm#SafetyAnnouncement. Accessed March 24, 2011.
3. Yang YX, Lewis JD, Epstein S, et al. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296:2947-2953.
4. Vestergaard P, Rejnmark L, Mosekilde L. Proton pump inhibitors, histamine h(2) receptor antagonists, and other antacid medications and the risk of fracture. Calcif Tissue Int. 2006;79:76-83.
5. Targownik LE, Lix LM, Metge CJ. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ. 2008;179:319-326.
6. Corley DA, Kubo A, Zhao W, et al. Proton pump inhibitors and histamine-2 receptor antagonists are associated with hip fractures among at-risk patients. Gastroenterology. 2010;139:93-101.
7. Targownik LE, Lix LM, Leung S, et al. Proton-pump inhibitor use is not associated with osteoporosis or accelerated bone mineral density loss. Gastroenterology. 2010;138:896-904.
8. Kaye JA, Jick H. Proton pump inhibitor use and risk of hip fractures in patients without major risk factors. Pharmacotherapy. 2008;28:951-959.
9. Yu EW, Blackwell T, Ensrud KE, et al. Acid-suppressive medications and risk of bone loss and fracture in older adults. Calcif Tissue Int. 2008;83:251-259.
10. Bo-Linn GW, Davis GR, Buddrus DJ, et al. An evaluation of the importance of gastric acid secretion in the absorption of dietary calcium. J Clin Invest. 1984;73:640-647.
11. Farina C, Gagliardi S. Selective inhibition of osteoclast vacuolar H+-ATPase. Curr Pharm Des. 2002;8:2033-2048.
12. Insogna KL. The effect of proton pump-inhibiting drugs on mineral metabolism. Am J Gastroenterol. 2009;104(suppl 2):S2-S4.
13. Gray SL, LaCroix AZ, Larson L, et al. Proton pump inhibitor use, hip fracture, and change in bone mineral density in postmenopausal women. Arch Intern Med. 2010;170:765-771.
14. Laine L. Proton pump inhibitors and bone fractures? Am J Gastroenterol. 2009;104(suppl 2):S21-S26.
15. Heidelbaugh JJ, Goldberg KL, Inadomi JM. Overutilization of proton pump inhibitors: a review of cost-effectiveness and risk. Am J Gastroenterol.;2009;104(suppl 2):S27-S32.
16. National Digestive Diseases Information Clearinghouse. Heartburn, gastroesophageal reflux (GER), and gastroesophageal reflux disease (GERD). Available at: http://digestive.niddk.nih.gov/ddiseases/pubs/gerd. Accessed April 18, 2011.
Coronary Revascularization In Ischemic Heart Disease
Coronary revascularization using bypass grafting with arterial or venous conduits has been with us since 1968 when Dr. Rene Favaloro performed the first saphenous venous graft for the treatment of angina pectoris (J. Thorac. Cardiovasc. Surg. 1969;58:178-85). Although it is clear that coronary artery bypass grafting (CABG) has been effective in decreasing symptomatic angina, with few exceptions there has been little to support its benefit in prolonging life. One of those exceptions was identified in a subgroup of the initial Coronary Artery Surgery Study carried out in the 1980s and sponsored by the National Heart, Lung, and Blood Institute (N. Engl. J. Med. 1985;312:1665-71). Of the 780 patients with chronic stable angina randomized to medicine only or CABG, there was a significant decrease in both angina and mortality in a subgroup of 160 patients with ejection fractions below 50%, primarily in patients with triple-vessel disease.
Since that report in 1985, there have been no clinical mortality trials examining the clinical benefit of CABG surgery in patients with ischemic heart failure. A randomized trial to evaluate the benefit of surgical ventricular reconstruction plus CABG, compared with CABG alone, failed to observe any benefit (N. Engl. J. Med. 2009:360;1705-17).
The suggestion that CABG could improve ventricular function is based on the observations by Dr. Shahbudin Rahimtoola in the 1980s in studies showing improved function in patients before and after CABG (Am. Heart J. 1989;117:211-21). He proposed the concept that areas of “hibernating myocardium” exist in the ischemic ventricle that can be revived by restoring its blood supply by CABG. But to a large degree, patients with ischemic heart failure have not been a prime target for CABG, and attempts to show clinical benefit in symptomatic improvement in heart failure has not been explored.
The recent report of the Surgical Treatment for Ischemic Heart Failure (STICH) trial has provided important information supporting the mortality and morbidity benefit of revascularization in patients with symptomatic ischemic heart failure (N. Engl. J. Med. 2011;364:1607-16). This study, also supported by NHLBI, was carried out in 26 countries throughout the world. In the 1,212 patients randomized to standard medical therapy alone, compared with medical therapy plus CABG, there was no significant benefit observed in the CABG patients in all-cause mortality, but there was a 19% decrease in cardiovascular mortality (P = .05) over a 3-year mean follow-up, and a 26% decrease in all-cause mortality and cardiovascular hospitalization (P less than .001). When patients who received CABG either by random assignment or because they were crossed over to surgery (620) were compared with those patients who remained on medical therapy (592), the effects of surgery were even more impressive, with a 30% decrease in all-cause mortality (P less than .001). The patients included in STICH were severely symptomatic, almost all with significant angina and 37% in NYHA HF class III/IV with a mean ejection fraction of 27%. Surgery carried an early up-front mortality risk of approximately 4%, which took about 2 years to overcome.
One interesting additional aspect of STICH was the viability study carried out in a subset of 601 patients using either dobutamine echocardiograms or SPECT stress testing. Although patients who demonstrated viability had a better outcome, viability did not define those patients who would benefit by CABG (N. Engl. J. Med. 2011;364:1617-25).
The “backstory” of the STICH trial was the failure of the U.S. cardiothoracic surgery centers to participate in it in a significant way. A total of 26 countries were required to achieve the 2,136 patients enrolled in the total STICH trial, and only 307 patients (14%) were American. The failure of the academic and large clinical centers to grasp the importance of this trial, and their reluctance to participate, was unfortunate.
The results of STICH indicate that the addition of CABG to patients already receiving optimal medical therapy provides a significant mortality and morbidity benefit. Unfortunately, viability studies do not provide helpful information in regard to the optimal selection of patients for CABG in ischemic heart failure. That decision appears to depend upon the availability of acceptable target vessels. But the data do support CABG, performed with an acceptable risk in experienced hands, as providing long-term benefits for heart failure patients.
Revascularization provides an additional mode of therapy for the treatment of patients with symptomatic ischemic heart failure, which could become a potential therapeutic target for percutaneous intervention in patients with the appropriate anatomy.
Coronary revascularization using bypass grafting with arterial or venous conduits has been with us since 1968 when Dr. Rene Favaloro performed the first saphenous venous graft for the treatment of angina pectoris (J. Thorac. Cardiovasc. Surg. 1969;58:178-85). Although it is clear that coronary artery bypass grafting (CABG) has been effective in decreasing symptomatic angina, with few exceptions there has been little to support its benefit in prolonging life. One of those exceptions was identified in a subgroup of the initial Coronary Artery Surgery Study carried out in the 1980s and sponsored by the National Heart, Lung, and Blood Institute (N. Engl. J. Med. 1985;312:1665-71). Of the 780 patients with chronic stable angina randomized to medicine only or CABG, there was a significant decrease in both angina and mortality in a subgroup of 160 patients with ejection fractions below 50%, primarily in patients with triple-vessel disease.
Since that report in 1985, there have been no clinical mortality trials examining the clinical benefit of CABG surgery in patients with ischemic heart failure. A randomized trial to evaluate the benefit of surgical ventricular reconstruction plus CABG, compared with CABG alone, failed to observe any benefit (N. Engl. J. Med. 2009:360;1705-17).
The suggestion that CABG could improve ventricular function is based on the observations by Dr. Shahbudin Rahimtoola in the 1980s in studies showing improved function in patients before and after CABG (Am. Heart J. 1989;117:211-21). He proposed the concept that areas of “hibernating myocardium” exist in the ischemic ventricle that can be revived by restoring its blood supply by CABG. But to a large degree, patients with ischemic heart failure have not been a prime target for CABG, and attempts to show clinical benefit in symptomatic improvement in heart failure has not been explored.
The recent report of the Surgical Treatment for Ischemic Heart Failure (STICH) trial has provided important information supporting the mortality and morbidity benefit of revascularization in patients with symptomatic ischemic heart failure (N. Engl. J. Med. 2011;364:1607-16). This study, also supported by NHLBI, was carried out in 26 countries throughout the world. In the 1,212 patients randomized to standard medical therapy alone, compared with medical therapy plus CABG, there was no significant benefit observed in the CABG patients in all-cause mortality, but there was a 19% decrease in cardiovascular mortality (P = .05) over a 3-year mean follow-up, and a 26% decrease in all-cause mortality and cardiovascular hospitalization (P less than .001). When patients who received CABG either by random assignment or because they were crossed over to surgery (620) were compared with those patients who remained on medical therapy (592), the effects of surgery were even more impressive, with a 30% decrease in all-cause mortality (P less than .001). The patients included in STICH were severely symptomatic, almost all with significant angina and 37% in NYHA HF class III/IV with a mean ejection fraction of 27%. Surgery carried an early up-front mortality risk of approximately 4%, which took about 2 years to overcome.
One interesting additional aspect of STICH was the viability study carried out in a subset of 601 patients using either dobutamine echocardiograms or SPECT stress testing. Although patients who demonstrated viability had a better outcome, viability did not define those patients who would benefit by CABG (N. Engl. J. Med. 2011;364:1617-25).
The “backstory” of the STICH trial was the failure of the U.S. cardiothoracic surgery centers to participate in it in a significant way. A total of 26 countries were required to achieve the 2,136 patients enrolled in the total STICH trial, and only 307 patients (14%) were American. The failure of the academic and large clinical centers to grasp the importance of this trial, and their reluctance to participate, was unfortunate.
The results of STICH indicate that the addition of CABG to patients already receiving optimal medical therapy provides a significant mortality and morbidity benefit. Unfortunately, viability studies do not provide helpful information in regard to the optimal selection of patients for CABG in ischemic heart failure. That decision appears to depend upon the availability of acceptable target vessels. But the data do support CABG, performed with an acceptable risk in experienced hands, as providing long-term benefits for heart failure patients.
Revascularization provides an additional mode of therapy for the treatment of patients with symptomatic ischemic heart failure, which could become a potential therapeutic target for percutaneous intervention in patients with the appropriate anatomy.
Coronary revascularization using bypass grafting with arterial or venous conduits has been with us since 1968 when Dr. Rene Favaloro performed the first saphenous venous graft for the treatment of angina pectoris (J. Thorac. Cardiovasc. Surg. 1969;58:178-85). Although it is clear that coronary artery bypass grafting (CABG) has been effective in decreasing symptomatic angina, with few exceptions there has been little to support its benefit in prolonging life. One of those exceptions was identified in a subgroup of the initial Coronary Artery Surgery Study carried out in the 1980s and sponsored by the National Heart, Lung, and Blood Institute (N. Engl. J. Med. 1985;312:1665-71). Of the 780 patients with chronic stable angina randomized to medicine only or CABG, there was a significant decrease in both angina and mortality in a subgroup of 160 patients with ejection fractions below 50%, primarily in patients with triple-vessel disease.
Since that report in 1985, there have been no clinical mortality trials examining the clinical benefit of CABG surgery in patients with ischemic heart failure. A randomized trial to evaluate the benefit of surgical ventricular reconstruction plus CABG, compared with CABG alone, failed to observe any benefit (N. Engl. J. Med. 2009:360;1705-17).
The suggestion that CABG could improve ventricular function is based on the observations by Dr. Shahbudin Rahimtoola in the 1980s in studies showing improved function in patients before and after CABG (Am. Heart J. 1989;117:211-21). He proposed the concept that areas of “hibernating myocardium” exist in the ischemic ventricle that can be revived by restoring its blood supply by CABG. But to a large degree, patients with ischemic heart failure have not been a prime target for CABG, and attempts to show clinical benefit in symptomatic improvement in heart failure has not been explored.
The recent report of the Surgical Treatment for Ischemic Heart Failure (STICH) trial has provided important information supporting the mortality and morbidity benefit of revascularization in patients with symptomatic ischemic heart failure (N. Engl. J. Med. 2011;364:1607-16). This study, also supported by NHLBI, was carried out in 26 countries throughout the world. In the 1,212 patients randomized to standard medical therapy alone, compared with medical therapy plus CABG, there was no significant benefit observed in the CABG patients in all-cause mortality, but there was a 19% decrease in cardiovascular mortality (P = .05) over a 3-year mean follow-up, and a 26% decrease in all-cause mortality and cardiovascular hospitalization (P less than .001). When patients who received CABG either by random assignment or because they were crossed over to surgery (620) were compared with those patients who remained on medical therapy (592), the effects of surgery were even more impressive, with a 30% decrease in all-cause mortality (P less than .001). The patients included in STICH were severely symptomatic, almost all with significant angina and 37% in NYHA HF class III/IV with a mean ejection fraction of 27%. Surgery carried an early up-front mortality risk of approximately 4%, which took about 2 years to overcome.
One interesting additional aspect of STICH was the viability study carried out in a subset of 601 patients using either dobutamine echocardiograms or SPECT stress testing. Although patients who demonstrated viability had a better outcome, viability did not define those patients who would benefit by CABG (N. Engl. J. Med. 2011;364:1617-25).
The “backstory” of the STICH trial was the failure of the U.S. cardiothoracic surgery centers to participate in it in a significant way. A total of 26 countries were required to achieve the 2,136 patients enrolled in the total STICH trial, and only 307 patients (14%) were American. The failure of the academic and large clinical centers to grasp the importance of this trial, and their reluctance to participate, was unfortunate.
The results of STICH indicate that the addition of CABG to patients already receiving optimal medical therapy provides a significant mortality and morbidity benefit. Unfortunately, viability studies do not provide helpful information in regard to the optimal selection of patients for CABG in ischemic heart failure. That decision appears to depend upon the availability of acceptable target vessels. But the data do support CABG, performed with an acceptable risk in experienced hands, as providing long-term benefits for heart failure patients.
Revascularization provides an additional mode of therapy for the treatment of patients with symptomatic ischemic heart failure, which could become a potential therapeutic target for percutaneous intervention in patients with the appropriate anatomy.
CASE LETTER Hydroxyurea-induced palmar-plantar erythrodysesthesia in an adult with sickle cell disease
Ramandeep K. Bambrah, MD, Fauzia Rana, MD, and Dat C. Pham, MD
Department of Medicine, University of Florida College of Medicine, Jacksonville, FL
Sickle cell disease consists of hemolytic anemia and episodes of vaso-occlusion, which are caused by sickling of red blood cells (RBCs) precipitated by deoxygenation. The change in shape of the RBCs is what causes vascular occlusion, leading to acute sickle crisis.
Hydroxyurea has been approved by the US Food and Drug Administration for the treatment of adult patients with clinically severe disease. Hydroxyurea blocks DNA synthesis via enzymatic inhibition of ribonucleotide reductase.1 Administration of hydroxyurea is associated with an increase in HbF (fetal hemoglobin) levels,2 thereby reducing the severity of vaso-occlusive crises, acute pain, acute chest syndrome, transfusion requirements, and hospitalizations. 3 As a cytotoxic, cell cycle–specific agent, hydroxyurea is associated with several adverse reactions, namely bone marrow suppression.4 Other side effects associated with hydroxyurea use include gastrointestinal upset, mild dermatologic reactions, alopecia, and leg ulcers.
The case study presented here focuses on an adult with sickle cell disease who complained of painful discoloration and edema of his hands and feet with blisters about 1 week after starting hydroxyurea therapy.
Case presentation
A 50-year-old black man with a history of atrial fibrillation, avascular necrosis of the shoulder, and sickle cell disease had frequent hospitalizations for acute crises requiring multiple packed RBC transfusions. He had no known history of leukemia/ lymphoma or exposure to other cytotoxic drugs. The patient was placed on hydroxyurea to reduce the frequency of sickle cell crises. Once hydroxyurea was begun, the patient noticed a decrease in acute pain and improvement in his general well-being.
Approximately 1 week after initiation of hydroxyurea therapy, the patient started noticing darkening of his hands. Two weeks later, hyperpigmentation of the palmar creases was noted. Three weeks after hydroxyurea was started, the patient had edema and blisters of his hands and feet with associated desquamation. Symptoms were severe and painful and interfered with activities of daily living (Figure 1). The patient’s fingers were so swollen that he was unable to make a fist. His feet were so edematous that it was difficult to walk. After these signs of hand-foot syndrome developed, his hematologist discontinued hydroxyurea, and the edema and pain gradually improved. Three months after hydroxyurea discontinuation, the edema and erythema had resolved, but the patient still had some hyperpigmentation.
Discussion
Palmar-plantar erythrodysesthesia, also known as hand-foot syndrome or acral erythema, is a known adverse reaction of several antineoplastic medications, although few published articles mention hand-foot syndrome as a common adverse effect of hydroxyurea therapy.3–5 The incidence of hand-foot syndrome in patients being treated with 5-fluorouracil, capecitabine (Xeloda), or liposomal doxorubicin (Doxil) ranges from 7% to 63%.6 It is a known adverse event of hydroxyurea therapy, although its exact frequency has not been established.
In a study examining mucocutaneous changes in 158 patients with chronic myeloid leukemia (CML) and long-term hydroxyurea therapy, 21 had severe changes and acral erythema.7 In these CML patients acral persistent erythema involved palmar, plantar, and facial areas, with less frequent involvement of the scrotum. 7 Symptoms were described as a burning sensation associated with redness, scaling, and fissuring.7 Acral erythema was noted to disappear gradually after discontinuation of hydroxyurea.7 Handfoot syndrome is known to occur in patients with CML being treated with hydroxyurea, but our case report describes a patient with hand-foot syndrome associated with hydroxyurea therapy and sickle cell disease.
Most cases of hand-foot syndrome improve, if not completely resolve, after cessation of the offending agent. Our patient did not require treatment specifically for hand-foot syndrome, as termination of hydroxyurea resulted in resolution of edema and pain.
Among the most common therapeutic agents for hand-foot syndrome is pyridoxine (vitamin B6). Pyridoxine has not been known to prevent the development of hand-foot syndrome, but observations suggest better symptom control with this vitamin, although randomized controlled studies are needed to further support this use.6 Another agent to consider for hand-foot syndrome is topical 99% dimethyl sulfoxide (DMSO). Case descriptions have reported improvement with DMSO in soft-tissue damage and edema in patients being treated with liposomal doxorubicin.8
In conclusion, hand-foot syndrome is a potentially reversible condition that is a common complication of certain chemotherapeutic drugs. However, it is not frequently reported with the use of hydroxyurea in adult patients with sickle cell disease.
Disclosures
The authors have nothing to disclose.
References
1. Navarra P, Preziosi P. Hydroxyurea: new insights on an old drug. Crit Rev Oncol Hematol 1999;29:249–255.
2. Steinberg MH, Lu ZH, Barton FB, Terrin ML, Charache S, Dover GJ. Fetal hemoglobin in sickle cell anemia: determinants of response to hydroxyurea. Multicenter Study of Hydroxyurea. Blood 1997;89:1078–1088.
3. Kovacic P. Hydroxyurea (therapeutics and mechanism): metabolism, carbamoyl nitroso, nitroxyl, radicals, cell signaling, and clinical applications. Med Hypotheses 2011;76:24–31.
4. Singh H, Dulhani N, Kumar BN, Singh P, Tiwari P. Effective control of sickle cell disease with hydroxyurea therapy. Indian J Pharmacol 2010;42:32–35.
5. Steinberg MH, McCarthy WF, Castro O, et al; Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia and MSH Patients’ Follow-Up. The risks and benefits of long-term use of hydroxyurea in sickle cell anemia: a 17.5 year follow-up. Am J Hematol 2010;85:403–408.
6. Clark AS, Vahdat LT. Chemotherapy-induced palmar-plantar erythrodysesthesia syndrome: etiology and emerging therapies. Support Cancer Ther 2004;1:213–218.
7. Vassallo C, Passamonti F, Merante S, et al. Muco-cutaneous changes during long-term therapy with hydroxyurea in chronic myeloid leukaemia. Clin Exp Dermatol 2001;26:141–148.
8. Lopez AM, Wallace L, Dorr RT, Koff M, Hersh EM, Alberts DS. Topical DMSO treatment for pegylated liposomal doxorubicin-induced palmar-plantar erythrodysesthesia. Cancer Chemother Pharmacol 1999;44:303–306.
Ramandeep K. Bambrah, MD, Fauzia Rana, MD, and Dat C. Pham, MD
Department of Medicine, University of Florida College of Medicine, Jacksonville, FL
Sickle cell disease consists of hemolytic anemia and episodes of vaso-occlusion, which are caused by sickling of red blood cells (RBCs) precipitated by deoxygenation. The change in shape of the RBCs is what causes vascular occlusion, leading to acute sickle crisis.
Hydroxyurea has been approved by the US Food and Drug Administration for the treatment of adult patients with clinically severe disease. Hydroxyurea blocks DNA synthesis via enzymatic inhibition of ribonucleotide reductase.1 Administration of hydroxyurea is associated with an increase in HbF (fetal hemoglobin) levels,2 thereby reducing the severity of vaso-occlusive crises, acute pain, acute chest syndrome, transfusion requirements, and hospitalizations. 3 As a cytotoxic, cell cycle–specific agent, hydroxyurea is associated with several adverse reactions, namely bone marrow suppression.4 Other side effects associated with hydroxyurea use include gastrointestinal upset, mild dermatologic reactions, alopecia, and leg ulcers.
The case study presented here focuses on an adult with sickle cell disease who complained of painful discoloration and edema of his hands and feet with blisters about 1 week after starting hydroxyurea therapy.
Case presentation
A 50-year-old black man with a history of atrial fibrillation, avascular necrosis of the shoulder, and sickle cell disease had frequent hospitalizations for acute crises requiring multiple packed RBC transfusions. He had no known history of leukemia/ lymphoma or exposure to other cytotoxic drugs. The patient was placed on hydroxyurea to reduce the frequency of sickle cell crises. Once hydroxyurea was begun, the patient noticed a decrease in acute pain and improvement in his general well-being.
Approximately 1 week after initiation of hydroxyurea therapy, the patient started noticing darkening of his hands. Two weeks later, hyperpigmentation of the palmar creases was noted. Three weeks after hydroxyurea was started, the patient had edema and blisters of his hands and feet with associated desquamation. Symptoms were severe and painful and interfered with activities of daily living (Figure 1). The patient’s fingers were so swollen that he was unable to make a fist. His feet were so edematous that it was difficult to walk. After these signs of hand-foot syndrome developed, his hematologist discontinued hydroxyurea, and the edema and pain gradually improved. Three months after hydroxyurea discontinuation, the edema and erythema had resolved, but the patient still had some hyperpigmentation.
Discussion
Palmar-plantar erythrodysesthesia, also known as hand-foot syndrome or acral erythema, is a known adverse reaction of several antineoplastic medications, although few published articles mention hand-foot syndrome as a common adverse effect of hydroxyurea therapy.3–5 The incidence of hand-foot syndrome in patients being treated with 5-fluorouracil, capecitabine (Xeloda), or liposomal doxorubicin (Doxil) ranges from 7% to 63%.6 It is a known adverse event of hydroxyurea therapy, although its exact frequency has not been established.
In a study examining mucocutaneous changes in 158 patients with chronic myeloid leukemia (CML) and long-term hydroxyurea therapy, 21 had severe changes and acral erythema.7 In these CML patients acral persistent erythema involved palmar, plantar, and facial areas, with less frequent involvement of the scrotum. 7 Symptoms were described as a burning sensation associated with redness, scaling, and fissuring.7 Acral erythema was noted to disappear gradually after discontinuation of hydroxyurea.7 Handfoot syndrome is known to occur in patients with CML being treated with hydroxyurea, but our case report describes a patient with hand-foot syndrome associated with hydroxyurea therapy and sickle cell disease.
Most cases of hand-foot syndrome improve, if not completely resolve, after cessation of the offending agent. Our patient did not require treatment specifically for hand-foot syndrome, as termination of hydroxyurea resulted in resolution of edema and pain.
Among the most common therapeutic agents for hand-foot syndrome is pyridoxine (vitamin B6). Pyridoxine has not been known to prevent the development of hand-foot syndrome, but observations suggest better symptom control with this vitamin, although randomized controlled studies are needed to further support this use.6 Another agent to consider for hand-foot syndrome is topical 99% dimethyl sulfoxide (DMSO). Case descriptions have reported improvement with DMSO in soft-tissue damage and edema in patients being treated with liposomal doxorubicin.8
In conclusion, hand-foot syndrome is a potentially reversible condition that is a common complication of certain chemotherapeutic drugs. However, it is not frequently reported with the use of hydroxyurea in adult patients with sickle cell disease.
Disclosures
The authors have nothing to disclose.
References
1. Navarra P, Preziosi P. Hydroxyurea: new insights on an old drug. Crit Rev Oncol Hematol 1999;29:249–255.
2. Steinberg MH, Lu ZH, Barton FB, Terrin ML, Charache S, Dover GJ. Fetal hemoglobin in sickle cell anemia: determinants of response to hydroxyurea. Multicenter Study of Hydroxyurea. Blood 1997;89:1078–1088.
3. Kovacic P. Hydroxyurea (therapeutics and mechanism): metabolism, carbamoyl nitroso, nitroxyl, radicals, cell signaling, and clinical applications. Med Hypotheses 2011;76:24–31.
4. Singh H, Dulhani N, Kumar BN, Singh P, Tiwari P. Effective control of sickle cell disease with hydroxyurea therapy. Indian J Pharmacol 2010;42:32–35.
5. Steinberg MH, McCarthy WF, Castro O, et al; Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia and MSH Patients’ Follow-Up. The risks and benefits of long-term use of hydroxyurea in sickle cell anemia: a 17.5 year follow-up. Am J Hematol 2010;85:403–408.
6. Clark AS, Vahdat LT. Chemotherapy-induced palmar-plantar erythrodysesthesia syndrome: etiology and emerging therapies. Support Cancer Ther 2004;1:213–218.
7. Vassallo C, Passamonti F, Merante S, et al. Muco-cutaneous changes during long-term therapy with hydroxyurea in chronic myeloid leukaemia. Clin Exp Dermatol 2001;26:141–148.
8. Lopez AM, Wallace L, Dorr RT, Koff M, Hersh EM, Alberts DS. Topical DMSO treatment for pegylated liposomal doxorubicin-induced palmar-plantar erythrodysesthesia. Cancer Chemother Pharmacol 1999;44:303–306.
Ramandeep K. Bambrah, MD, Fauzia Rana, MD, and Dat C. Pham, MD
Department of Medicine, University of Florida College of Medicine, Jacksonville, FL
Sickle cell disease consists of hemolytic anemia and episodes of vaso-occlusion, which are caused by sickling of red blood cells (RBCs) precipitated by deoxygenation. The change in shape of the RBCs is what causes vascular occlusion, leading to acute sickle crisis.
Hydroxyurea has been approved by the US Food and Drug Administration for the treatment of adult patients with clinically severe disease. Hydroxyurea blocks DNA synthesis via enzymatic inhibition of ribonucleotide reductase.1 Administration of hydroxyurea is associated with an increase in HbF (fetal hemoglobin) levels,2 thereby reducing the severity of vaso-occlusive crises, acute pain, acute chest syndrome, transfusion requirements, and hospitalizations. 3 As a cytotoxic, cell cycle–specific agent, hydroxyurea is associated with several adverse reactions, namely bone marrow suppression.4 Other side effects associated with hydroxyurea use include gastrointestinal upset, mild dermatologic reactions, alopecia, and leg ulcers.
The case study presented here focuses on an adult with sickle cell disease who complained of painful discoloration and edema of his hands and feet with blisters about 1 week after starting hydroxyurea therapy.
Case presentation
A 50-year-old black man with a history of atrial fibrillation, avascular necrosis of the shoulder, and sickle cell disease had frequent hospitalizations for acute crises requiring multiple packed RBC transfusions. He had no known history of leukemia/ lymphoma or exposure to other cytotoxic drugs. The patient was placed on hydroxyurea to reduce the frequency of sickle cell crises. Once hydroxyurea was begun, the patient noticed a decrease in acute pain and improvement in his general well-being.
Approximately 1 week after initiation of hydroxyurea therapy, the patient started noticing darkening of his hands. Two weeks later, hyperpigmentation of the palmar creases was noted. Three weeks after hydroxyurea was started, the patient had edema and blisters of his hands and feet with associated desquamation. Symptoms were severe and painful and interfered with activities of daily living (Figure 1). The patient’s fingers were so swollen that he was unable to make a fist. His feet were so edematous that it was difficult to walk. After these signs of hand-foot syndrome developed, his hematologist discontinued hydroxyurea, and the edema and pain gradually improved. Three months after hydroxyurea discontinuation, the edema and erythema had resolved, but the patient still had some hyperpigmentation.
Discussion
Palmar-plantar erythrodysesthesia, also known as hand-foot syndrome or acral erythema, is a known adverse reaction of several antineoplastic medications, although few published articles mention hand-foot syndrome as a common adverse effect of hydroxyurea therapy.3–5 The incidence of hand-foot syndrome in patients being treated with 5-fluorouracil, capecitabine (Xeloda), or liposomal doxorubicin (Doxil) ranges from 7% to 63%.6 It is a known adverse event of hydroxyurea therapy, although its exact frequency has not been established.
In a study examining mucocutaneous changes in 158 patients with chronic myeloid leukemia (CML) and long-term hydroxyurea therapy, 21 had severe changes and acral erythema.7 In these CML patients acral persistent erythema involved palmar, plantar, and facial areas, with less frequent involvement of the scrotum. 7 Symptoms were described as a burning sensation associated with redness, scaling, and fissuring.7 Acral erythema was noted to disappear gradually after discontinuation of hydroxyurea.7 Handfoot syndrome is known to occur in patients with CML being treated with hydroxyurea, but our case report describes a patient with hand-foot syndrome associated with hydroxyurea therapy and sickle cell disease.
Most cases of hand-foot syndrome improve, if not completely resolve, after cessation of the offending agent. Our patient did not require treatment specifically for hand-foot syndrome, as termination of hydroxyurea resulted in resolution of edema and pain.
Among the most common therapeutic agents for hand-foot syndrome is pyridoxine (vitamin B6). Pyridoxine has not been known to prevent the development of hand-foot syndrome, but observations suggest better symptom control with this vitamin, although randomized controlled studies are needed to further support this use.6 Another agent to consider for hand-foot syndrome is topical 99% dimethyl sulfoxide (DMSO). Case descriptions have reported improvement with DMSO in soft-tissue damage and edema in patients being treated with liposomal doxorubicin.8
In conclusion, hand-foot syndrome is a potentially reversible condition that is a common complication of certain chemotherapeutic drugs. However, it is not frequently reported with the use of hydroxyurea in adult patients with sickle cell disease.
Disclosures
The authors have nothing to disclose.
References
1. Navarra P, Preziosi P. Hydroxyurea: new insights on an old drug. Crit Rev Oncol Hematol 1999;29:249–255.
2. Steinberg MH, Lu ZH, Barton FB, Terrin ML, Charache S, Dover GJ. Fetal hemoglobin in sickle cell anemia: determinants of response to hydroxyurea. Multicenter Study of Hydroxyurea. Blood 1997;89:1078–1088.
3. Kovacic P. Hydroxyurea (therapeutics and mechanism): metabolism, carbamoyl nitroso, nitroxyl, radicals, cell signaling, and clinical applications. Med Hypotheses 2011;76:24–31.
4. Singh H, Dulhani N, Kumar BN, Singh P, Tiwari P. Effective control of sickle cell disease with hydroxyurea therapy. Indian J Pharmacol 2010;42:32–35.
5. Steinberg MH, McCarthy WF, Castro O, et al; Investigators of the Multicenter Study of Hydroxyurea in Sickle Cell Anemia and MSH Patients’ Follow-Up. The risks and benefits of long-term use of hydroxyurea in sickle cell anemia: a 17.5 year follow-up. Am J Hematol 2010;85:403–408.
6. Clark AS, Vahdat LT. Chemotherapy-induced palmar-plantar erythrodysesthesia syndrome: etiology and emerging therapies. Support Cancer Ther 2004;1:213–218.
7. Vassallo C, Passamonti F, Merante S, et al. Muco-cutaneous changes during long-term therapy with hydroxyurea in chronic myeloid leukaemia. Clin Exp Dermatol 2001;26:141–148.
8. Lopez AM, Wallace L, Dorr RT, Koff M, Hersh EM, Alberts DS. Topical DMSO treatment for pegylated liposomal doxorubicin-induced palmar-plantar erythrodysesthesia. Cancer Chemother Pharmacol 1999;44:303–306.
Helping Your Patient With a Bee Sting Allergy
Although diagnosis of a bee sting allergy is often straightforward, it's important to go through the history. Ask when the children were stung, what type of reaction they had, and how soon after the sting they experienced symptoms.
A large local reaction can be impressive in size, but it may not be as serious as the child who presents with systemic symptoms such as hives or difficulty breathing.
Immediately direct a child experiencing acute anaphylaxis to emergency care. Acute effects will be seen right away, generally within 15-30 minutes. The parents of a child with a known sensitivity to bee stings, in particular, will know to head to the emergency department right away, especially after self-administration of epinephrine by an autoinjector.
It is more likely that a patient will come to you with a less severe reaction or for advice on how to manage a potential allergy. In general, local reactions are no larger than 10 cm, and you can treat the area with ice or cold compresses in your office. Typical local reactions are a little bump, a local hive, or an indurated area of swelling that is warm or hot.
Take photos of the allergic reaction. This can be very helpful if you later refer the child to a specialist. It helps us immediately see the size and location of the reaction.
Check to see if the stinger is still in place when a flustered child (or parent) comes in right after a bee sting. Although most people remove it immediately, some patients come in with the stinger still in the skin. You want to scrape or brush across the skin with a credit card or coin to remove the stinger. The removal technique is important because honey bees can leave both their stinger and venom sac behind as a last defense. If you just try to pull out the stinger, unintentional squeezing of the venom sac can mean more venom gets injected into the allergic child.
Consider referral to a pediatric allergy specialist if a child has a history of adverse or severe reactions to bee stings. The risk of future severe reactions, including anaphylaxis, will be elevated in a patient who has already spent any time in the emergency department, for example. When you refer, include a list of any local or systemic symptoms and any medications the child is taking.
Each subsequent exposure to bee venom increases the risk of a more severe reaction. One question I always get is: “I've been stung 15 times before. How come this time I developed an anaphylactic reaction?” I explain that a person needs to be stung only once before the body can develop an allergy, and any exposure after that may trigger a serious or life-threatening reaction.
You can perform allergy testing in your primary care office, but the question is what to do with the results. Such testing prior to referral does not tend to help us a lot. We often perform a more comprehensive evaluation. For example, as a general rule I order IgE protein-specific tests for the five common flying insect venoms, because most children cannot tell if a wasp, hornet, or bee stung them.
The good news is that if an individual meets criteria and is treated with immunotherapy or allergy shots, he or she has a success rate of about 98%. Even so, I recommend that a child with a history of bee sting adverse reactions carry an autoinjectable epinephrine device and practice bee avoidance measures.
You can teach children how to stay away from bees. Tell them not to play in or around woods, for example. Make sure they know not to provoke or aggravate any bees they encounter, and that bees are attracted by bright-colored clothing, perfume, and cologne. I tell patients to avoid drinking cans of soda outdoors. Bees attracted to the sweet soda will fly into these cans and, unfortunately, it is not uncommon for people to be very surprised and get stung in the mouth, on the tongue, or on the lips this way.
Although diagnosis of a bee sting allergy is often straightforward, it's important to go through the history. Ask when the children were stung, what type of reaction they had, and how soon after the sting they experienced symptoms.
A large local reaction can be impressive in size, but it may not be as serious as the child who presents with systemic symptoms such as hives or difficulty breathing.
Immediately direct a child experiencing acute anaphylaxis to emergency care. Acute effects will be seen right away, generally within 15-30 minutes. The parents of a child with a known sensitivity to bee stings, in particular, will know to head to the emergency department right away, especially after self-administration of epinephrine by an autoinjector.
It is more likely that a patient will come to you with a less severe reaction or for advice on how to manage a potential allergy. In general, local reactions are no larger than 10 cm, and you can treat the area with ice or cold compresses in your office. Typical local reactions are a little bump, a local hive, or an indurated area of swelling that is warm or hot.
Take photos of the allergic reaction. This can be very helpful if you later refer the child to a specialist. It helps us immediately see the size and location of the reaction.
Check to see if the stinger is still in place when a flustered child (or parent) comes in right after a bee sting. Although most people remove it immediately, some patients come in with the stinger still in the skin. You want to scrape or brush across the skin with a credit card or coin to remove the stinger. The removal technique is important because honey bees can leave both their stinger and venom sac behind as a last defense. If you just try to pull out the stinger, unintentional squeezing of the venom sac can mean more venom gets injected into the allergic child.
Consider referral to a pediatric allergy specialist if a child has a history of adverse or severe reactions to bee stings. The risk of future severe reactions, including anaphylaxis, will be elevated in a patient who has already spent any time in the emergency department, for example. When you refer, include a list of any local or systemic symptoms and any medications the child is taking.
Each subsequent exposure to bee venom increases the risk of a more severe reaction. One question I always get is: “I've been stung 15 times before. How come this time I developed an anaphylactic reaction?” I explain that a person needs to be stung only once before the body can develop an allergy, and any exposure after that may trigger a serious or life-threatening reaction.
You can perform allergy testing in your primary care office, but the question is what to do with the results. Such testing prior to referral does not tend to help us a lot. We often perform a more comprehensive evaluation. For example, as a general rule I order IgE protein-specific tests for the five common flying insect venoms, because most children cannot tell if a wasp, hornet, or bee stung them.
The good news is that if an individual meets criteria and is treated with immunotherapy or allergy shots, he or she has a success rate of about 98%. Even so, I recommend that a child with a history of bee sting adverse reactions carry an autoinjectable epinephrine device and practice bee avoidance measures.
You can teach children how to stay away from bees. Tell them not to play in or around woods, for example. Make sure they know not to provoke or aggravate any bees they encounter, and that bees are attracted by bright-colored clothing, perfume, and cologne. I tell patients to avoid drinking cans of soda outdoors. Bees attracted to the sweet soda will fly into these cans and, unfortunately, it is not uncommon for people to be very surprised and get stung in the mouth, on the tongue, or on the lips this way.
Although diagnosis of a bee sting allergy is often straightforward, it's important to go through the history. Ask when the children were stung, what type of reaction they had, and how soon after the sting they experienced symptoms.
A large local reaction can be impressive in size, but it may not be as serious as the child who presents with systemic symptoms such as hives or difficulty breathing.
Immediately direct a child experiencing acute anaphylaxis to emergency care. Acute effects will be seen right away, generally within 15-30 minutes. The parents of a child with a known sensitivity to bee stings, in particular, will know to head to the emergency department right away, especially after self-administration of epinephrine by an autoinjector.
It is more likely that a patient will come to you with a less severe reaction or for advice on how to manage a potential allergy. In general, local reactions are no larger than 10 cm, and you can treat the area with ice or cold compresses in your office. Typical local reactions are a little bump, a local hive, or an indurated area of swelling that is warm or hot.
Take photos of the allergic reaction. This can be very helpful if you later refer the child to a specialist. It helps us immediately see the size and location of the reaction.
Check to see if the stinger is still in place when a flustered child (or parent) comes in right after a bee sting. Although most people remove it immediately, some patients come in with the stinger still in the skin. You want to scrape or brush across the skin with a credit card or coin to remove the stinger. The removal technique is important because honey bees can leave both their stinger and venom sac behind as a last defense. If you just try to pull out the stinger, unintentional squeezing of the venom sac can mean more venom gets injected into the allergic child.
Consider referral to a pediatric allergy specialist if a child has a history of adverse or severe reactions to bee stings. The risk of future severe reactions, including anaphylaxis, will be elevated in a patient who has already spent any time in the emergency department, for example. When you refer, include a list of any local or systemic symptoms and any medications the child is taking.
Each subsequent exposure to bee venom increases the risk of a more severe reaction. One question I always get is: “I've been stung 15 times before. How come this time I developed an anaphylactic reaction?” I explain that a person needs to be stung only once before the body can develop an allergy, and any exposure after that may trigger a serious or life-threatening reaction.
You can perform allergy testing in your primary care office, but the question is what to do with the results. Such testing prior to referral does not tend to help us a lot. We often perform a more comprehensive evaluation. For example, as a general rule I order IgE protein-specific tests for the five common flying insect venoms, because most children cannot tell if a wasp, hornet, or bee stung them.
The good news is that if an individual meets criteria and is treated with immunotherapy or allergy shots, he or she has a success rate of about 98%. Even so, I recommend that a child with a history of bee sting adverse reactions carry an autoinjectable epinephrine device and practice bee avoidance measures.
You can teach children how to stay away from bees. Tell them not to play in or around woods, for example. Make sure they know not to provoke or aggravate any bees they encounter, and that bees are attracted by bright-colored clothing, perfume, and cologne. I tell patients to avoid drinking cans of soda outdoors. Bees attracted to the sweet soda will fly into these cans and, unfortunately, it is not uncommon for people to be very surprised and get stung in the mouth, on the tongue, or on the lips this way.
Measuring VTE Risk
A risk-stratification tool is a first step for hospitalists and others trying to identify patients with sufficient VTE risk to warrant pharmacological prophylaxis, according to a new Journal of Hospital Medicine report.
The authors of the retrospective cohort study noted that while both the American College of Chest Physicians (ACCP) and the Joint Commission mandate inpatients can be assessed for VTE risk, there are no validated risk-stratification tools. So a team of researchers from Baystate Medical Center in Springfield, Mass., and Tufts University School of Medicine in Boston reviewed patients with a primary diagnosis of pneumonia, heart failure, chronic obstructive pulmonary disease (COPD), stroke, and urinary tract infection. Length of stay had to be greater than three days.
The authors reported the strongest risk factors were inherited thrombophilia (OR 4.00), length of stay equal to or greater than six days (OR 3.22), inflammatory bowel disease (OR 3.11), central venous catheter (OR 1.87), and cancer. But more research needs to be done to determine exactly what risk levels should trigger the use of prophylaxis, says lead author Michael Rothberg, MD, MPH, associate professor of medicine at Tufts and Baystate's interim chief of the Division of General Medicine.
"I would hope people would use the model as a way to measure a patient's risk," says Dr. Rothberg, who also serves as Baystate's director of scholarly activities in the Internal Medicine Training Program. "The problem is, we don't know the threshold."
Dr. Rothberg and his colleagues are currently preparing a grant application to take the next step in the research, which would be an attempt to define just what risk levels in patients should trigger pharmacological prophylaxis. While such treatments, including the use of heparin, have relatively low risks for patients, "the costs are real," Dr. Rothberg says.
In the meantime, he says, the take-home message from his team's preliminary work is that without accepted risk thresholds in place, physicians should determine prophylaxis use on a patient-by-patient basis.
"There's a need for a more nuanced approach," he says. "It's a not a one-size-fits-all."
A risk-stratification tool is a first step for hospitalists and others trying to identify patients with sufficient VTE risk to warrant pharmacological prophylaxis, according to a new Journal of Hospital Medicine report.
The authors of the retrospective cohort study noted that while both the American College of Chest Physicians (ACCP) and the Joint Commission mandate inpatients can be assessed for VTE risk, there are no validated risk-stratification tools. So a team of researchers from Baystate Medical Center in Springfield, Mass., and Tufts University School of Medicine in Boston reviewed patients with a primary diagnosis of pneumonia, heart failure, chronic obstructive pulmonary disease (COPD), stroke, and urinary tract infection. Length of stay had to be greater than three days.
The authors reported the strongest risk factors were inherited thrombophilia (OR 4.00), length of stay equal to or greater than six days (OR 3.22), inflammatory bowel disease (OR 3.11), central venous catheter (OR 1.87), and cancer. But more research needs to be done to determine exactly what risk levels should trigger the use of prophylaxis, says lead author Michael Rothberg, MD, MPH, associate professor of medicine at Tufts and Baystate's interim chief of the Division of General Medicine.
"I would hope people would use the model as a way to measure a patient's risk," says Dr. Rothberg, who also serves as Baystate's director of scholarly activities in the Internal Medicine Training Program. "The problem is, we don't know the threshold."
Dr. Rothberg and his colleagues are currently preparing a grant application to take the next step in the research, which would be an attempt to define just what risk levels in patients should trigger pharmacological prophylaxis. While such treatments, including the use of heparin, have relatively low risks for patients, "the costs are real," Dr. Rothberg says.
In the meantime, he says, the take-home message from his team's preliminary work is that without accepted risk thresholds in place, physicians should determine prophylaxis use on a patient-by-patient basis.
"There's a need for a more nuanced approach," he says. "It's a not a one-size-fits-all."
A risk-stratification tool is a first step for hospitalists and others trying to identify patients with sufficient VTE risk to warrant pharmacological prophylaxis, according to a new Journal of Hospital Medicine report.
The authors of the retrospective cohort study noted that while both the American College of Chest Physicians (ACCP) and the Joint Commission mandate inpatients can be assessed for VTE risk, there are no validated risk-stratification tools. So a team of researchers from Baystate Medical Center in Springfield, Mass., and Tufts University School of Medicine in Boston reviewed patients with a primary diagnosis of pneumonia, heart failure, chronic obstructive pulmonary disease (COPD), stroke, and urinary tract infection. Length of stay had to be greater than three days.
The authors reported the strongest risk factors were inherited thrombophilia (OR 4.00), length of stay equal to or greater than six days (OR 3.22), inflammatory bowel disease (OR 3.11), central venous catheter (OR 1.87), and cancer. But more research needs to be done to determine exactly what risk levels should trigger the use of prophylaxis, says lead author Michael Rothberg, MD, MPH, associate professor of medicine at Tufts and Baystate's interim chief of the Division of General Medicine.
"I would hope people would use the model as a way to measure a patient's risk," says Dr. Rothberg, who also serves as Baystate's director of scholarly activities in the Internal Medicine Training Program. "The problem is, we don't know the threshold."
Dr. Rothberg and his colleagues are currently preparing a grant application to take the next step in the research, which would be an attempt to define just what risk levels in patients should trigger pharmacological prophylaxis. While such treatments, including the use of heparin, have relatively low risks for patients, "the costs are real," Dr. Rothberg says.
In the meantime, he says, the take-home message from his team's preliminary work is that without accepted risk thresholds in place, physicians should determine prophylaxis use on a patient-by-patient basis.
"There's a need for a more nuanced approach," he says. "It's a not a one-size-fits-all."
SHM Board Adds First Family-Medicine-Trained Member
Hospitalist Robert Harrington Jr., MD, SFHM, chief medical officer for Alpharetta, Ga.-based Locum Leaders, a national physician recruiting and staffing company for hospitalists and other specialties, was recently elected to an at-large seat on SHM's Board of Directors. Dr. Harrington, the first family-medicine-trained hospitalist to serve on the board, says he brings a particular focus on the community-based hospitalist's perspective and will work to make SHM better for all HM providers.
"I regularly interact with community hospitals and physicians. I have also practiced in community settings," he says. "Being an advocate for community-based physicians, I'm a big believer in the 'big tent' philosophy of SHM's recent discussions about its future."
Those discussions about SHM's future have highlighted the variety of "hyphenated" and specialized hospitalists that have emerged in recent years. Dr. Harrington says that SHM should nurture specialist HM groups. "My goal on the board is to continue to push SHM to make it a home for all members of the patient care team," he says. "I continue to believe that hospital medicine is truly a team sport, with quality care delivered by inpatient care teams.”
Dr. Harrington chairs SHM's Family Medicine Task Force and recently was honored as a Senior Fellow in Hospital Medicine. He attended medical school at Temple University in Philadelphia and completed his residency at the Medical Center of Delaware in Wilmington, followed by a stint in the U.S. Air Force Medical Corps. He will commence his three-year term on the board at HM11 in Grapevine, Texas, in May.
Hospitalist Robert Harrington Jr., MD, SFHM, chief medical officer for Alpharetta, Ga.-based Locum Leaders, a national physician recruiting and staffing company for hospitalists and other specialties, was recently elected to an at-large seat on SHM's Board of Directors. Dr. Harrington, the first family-medicine-trained hospitalist to serve on the board, says he brings a particular focus on the community-based hospitalist's perspective and will work to make SHM better for all HM providers.
"I regularly interact with community hospitals and physicians. I have also practiced in community settings," he says. "Being an advocate for community-based physicians, I'm a big believer in the 'big tent' philosophy of SHM's recent discussions about its future."
Those discussions about SHM's future have highlighted the variety of "hyphenated" and specialized hospitalists that have emerged in recent years. Dr. Harrington says that SHM should nurture specialist HM groups. "My goal on the board is to continue to push SHM to make it a home for all members of the patient care team," he says. "I continue to believe that hospital medicine is truly a team sport, with quality care delivered by inpatient care teams.”
Dr. Harrington chairs SHM's Family Medicine Task Force and recently was honored as a Senior Fellow in Hospital Medicine. He attended medical school at Temple University in Philadelphia and completed his residency at the Medical Center of Delaware in Wilmington, followed by a stint in the U.S. Air Force Medical Corps. He will commence his three-year term on the board at HM11 in Grapevine, Texas, in May.
Hospitalist Robert Harrington Jr., MD, SFHM, chief medical officer for Alpharetta, Ga.-based Locum Leaders, a national physician recruiting and staffing company for hospitalists and other specialties, was recently elected to an at-large seat on SHM's Board of Directors. Dr. Harrington, the first family-medicine-trained hospitalist to serve on the board, says he brings a particular focus on the community-based hospitalist's perspective and will work to make SHM better for all HM providers.
"I regularly interact with community hospitals and physicians. I have also practiced in community settings," he says. "Being an advocate for community-based physicians, I'm a big believer in the 'big tent' philosophy of SHM's recent discussions about its future."
Those discussions about SHM's future have highlighted the variety of "hyphenated" and specialized hospitalists that have emerged in recent years. Dr. Harrington says that SHM should nurture specialist HM groups. "My goal on the board is to continue to push SHM to make it a home for all members of the patient care team," he says. "I continue to believe that hospital medicine is truly a team sport, with quality care delivered by inpatient care teams.”
Dr. Harrington chairs SHM's Family Medicine Task Force and recently was honored as a Senior Fellow in Hospital Medicine. He attended medical school at Temple University in Philadelphia and completed his residency at the Medical Center of Delaware in Wilmington, followed by a stint in the U.S. Air Force Medical Corps. He will commence his three-year term on the board at HM11 in Grapevine, Texas, in May.
Benefits of Using Lipid-Lowering Agents Persist After Trials End
NEW ORLEANS – In major clinical trials of lipid-lowering agents, the mortality benefit derived from medical therapy persists long after the studies end, according to a meta-analysis presented at the annual meeting of the American College of Cardiology.
Furthermore, placebo recipients who cross over to lipid-lowering therapy in the open-label phases of the studies demonstrate survival benefits as well, but never attain the protection achieved by being randomized to active treatment earlier on, according to Dr. William J. Kostis of Massachusetts General Hospital, Boston. "Persons with risk factors for coronary artery disease should be treated early," Dr. Kostis said in an interview. "The sooner you treat, the better."
He and his colleagues searched several major databases to identify randomized trials of lipid-lowering therapies that also contained an analysis of patient outcomes after the randomized portion of the trials had ended and an open-label phase had begun. Active treatment in the trials involved statins, niacin, cholestyramine, or gemfibrozil.
The analysis included eight clinical trials involving 44,255 patients, of whom 8,144 died during follow-up.
The average patient remained on the assigned treatment for approximately 5 years and was on the lipid-lowering agent in the open-label phase for approximately 6 years.
During the randomized phase of the trials, the mean all-cause mortality was significantly lower for the active treatment group (odds ratio, 0.84; P = .0006), as was cardiovascular mortality (0.72; P less than .001). The lower mortality among those initially receiving active therapy persisted during the open-label follow-up phase (OR, 0.90; P = .0035), as did the reduction in cardiovascular mortality (OR, 0.82; P = .0014).
"Being treated with a beneficial medication for a longer period of time is better, possibly because we are arresting pathophysiology at an earlier stage," Dr. Kostis proposed. He added that statins may be reducing the size of infarcts in patients who have myocardial infarctions.
Dr. Patrick Moriarty, a lipid specialist and a professor of medicine at the University of Kansas in Kansas City, agreed. "We need to start lipid-lowering therapy early to get the most benefit," and this includes interventions in children when necessary, he added.
"We treat pediatric patients all the time," he said, "not only those with familial hyperlipidemias but also those with metabolic syndrome. ... The future emphasis will be, ‘the sooner the better.’ "
Dr. Kostis and Dr. Moriarty reported having no relevant conflicts of interest.
NEW ORLEANS – In major clinical trials of lipid-lowering agents, the mortality benefit derived from medical therapy persists long after the studies end, according to a meta-analysis presented at the annual meeting of the American College of Cardiology.
Furthermore, placebo recipients who cross over to lipid-lowering therapy in the open-label phases of the studies demonstrate survival benefits as well, but never attain the protection achieved by being randomized to active treatment earlier on, according to Dr. William J. Kostis of Massachusetts General Hospital, Boston. "Persons with risk factors for coronary artery disease should be treated early," Dr. Kostis said in an interview. "The sooner you treat, the better."
He and his colleagues searched several major databases to identify randomized trials of lipid-lowering therapies that also contained an analysis of patient outcomes after the randomized portion of the trials had ended and an open-label phase had begun. Active treatment in the trials involved statins, niacin, cholestyramine, or gemfibrozil.
The analysis included eight clinical trials involving 44,255 patients, of whom 8,144 died during follow-up.
The average patient remained on the assigned treatment for approximately 5 years and was on the lipid-lowering agent in the open-label phase for approximately 6 years.
During the randomized phase of the trials, the mean all-cause mortality was significantly lower for the active treatment group (odds ratio, 0.84; P = .0006), as was cardiovascular mortality (0.72; P less than .001). The lower mortality among those initially receiving active therapy persisted during the open-label follow-up phase (OR, 0.90; P = .0035), as did the reduction in cardiovascular mortality (OR, 0.82; P = .0014).
"Being treated with a beneficial medication for a longer period of time is better, possibly because we are arresting pathophysiology at an earlier stage," Dr. Kostis proposed. He added that statins may be reducing the size of infarcts in patients who have myocardial infarctions.
Dr. Patrick Moriarty, a lipid specialist and a professor of medicine at the University of Kansas in Kansas City, agreed. "We need to start lipid-lowering therapy early to get the most benefit," and this includes interventions in children when necessary, he added.
"We treat pediatric patients all the time," he said, "not only those with familial hyperlipidemias but also those with metabolic syndrome. ... The future emphasis will be, ‘the sooner the better.’ "
Dr. Kostis and Dr. Moriarty reported having no relevant conflicts of interest.
NEW ORLEANS – In major clinical trials of lipid-lowering agents, the mortality benefit derived from medical therapy persists long after the studies end, according to a meta-analysis presented at the annual meeting of the American College of Cardiology.
Furthermore, placebo recipients who cross over to lipid-lowering therapy in the open-label phases of the studies demonstrate survival benefits as well, but never attain the protection achieved by being randomized to active treatment earlier on, according to Dr. William J. Kostis of Massachusetts General Hospital, Boston. "Persons with risk factors for coronary artery disease should be treated early," Dr. Kostis said in an interview. "The sooner you treat, the better."
He and his colleagues searched several major databases to identify randomized trials of lipid-lowering therapies that also contained an analysis of patient outcomes after the randomized portion of the trials had ended and an open-label phase had begun. Active treatment in the trials involved statins, niacin, cholestyramine, or gemfibrozil.
The analysis included eight clinical trials involving 44,255 patients, of whom 8,144 died during follow-up.
The average patient remained on the assigned treatment for approximately 5 years and was on the lipid-lowering agent in the open-label phase for approximately 6 years.
During the randomized phase of the trials, the mean all-cause mortality was significantly lower for the active treatment group (odds ratio, 0.84; P = .0006), as was cardiovascular mortality (0.72; P less than .001). The lower mortality among those initially receiving active therapy persisted during the open-label follow-up phase (OR, 0.90; P = .0035), as did the reduction in cardiovascular mortality (OR, 0.82; P = .0014).
"Being treated with a beneficial medication for a longer period of time is better, possibly because we are arresting pathophysiology at an earlier stage," Dr. Kostis proposed. He added that statins may be reducing the size of infarcts in patients who have myocardial infarctions.
Dr. Patrick Moriarty, a lipid specialist and a professor of medicine at the University of Kansas in Kansas City, agreed. "We need to start lipid-lowering therapy early to get the most benefit," and this includes interventions in children when necessary, he added.
"We treat pediatric patients all the time," he said, "not only those with familial hyperlipidemias but also those with metabolic syndrome. ... The future emphasis will be, ‘the sooner the better.’ "
Dr. Kostis and Dr. Moriarty reported having no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF CARDIOLOGY
Major Finding: During the open-label phase of randomized trials studied, the lower mortality among those who initially received active therapy persisted (odds ratio, 0.90; P = .0035), as did the reduction in cardiovascular mortality (OR, 0.82; P = .0014).
Data Source: A meta-analysis involving 44,255 patients in eight clinical trials of lipid-lowering therapy. All trials involved an open-label phase after the randomized treatment period ended.
Disclosures: Dr. Kostis and Dr. Moriarty reported having no relevant conflicts of interest.
ONLINE EXCLUSIVE: TKTK
Enter text here
Enter text here
Enter text here
How to Find a Mentor in the Mentorship Gap
Looking for a mentor to help guide your career development? HM presents young physicians with unique challenges and opportunities.
As a relatively young field, HM hasn't yet accrued a depth of senior leaders. Protected time is in shorter supply in academic medicine, as hospitalists bridge the Accreditation Council for Graduate Medical Education (ACGME) duty-hour coverage gap. In community hospitals, hospitalists are expressly hired to provide clinical coverage, and rewards for career advancement might not be included in the hospital's contract with the program. (For more on career advancement, listen to experts talk about why hospitalists make great hospital leaders.)

—Jeff Wiese, MD, SFHM, professor, associate dean for Graduate Medical Education, Tulane University Health Sciences Center, New Orleans
Each is a reason Steven B. Deitelzweig, MD, MMM, FACP, SFHM, vice president of medical affairs and system chairman of the Department of Hospital Medicine at Ochsner Health System in New Orleans, concludes: "If you rely on hospital medicine to teach you hospital medicine, you're only going to get so far in this day and age."
Big Community Ops
Working a week on/week off schedule might be the preferred goal for some hospitalists in the community setting, says Erik DeLue, MD, MBA, SFHM, hospitalist medical director for the Virtua healthcare system, which runs four hospitals in southern New Jersey. But if you find yourself asking, "Is there more out there for me?" community settings offer lots of opportunities to explore.
For example, one PhD physician in Dr. DeLue's group is parlaying her interest in research to the next level by submitting an abstract to the SHM annual meeting's poster competition regarding care coordination and fragmentation rates. Two other hospitalists share a clinical position (each works 0.5 FTE). On their off time, one works in information technology (IT) for Virtua, which pays the other half of his salary; the other does IT work for two hospital systems in New York City.
Click here to read the full story.
Searching for a new opportunity? View hundreds of jobs at SHM's Career Center.
Looking for a mentor to help guide your career development? HM presents young physicians with unique challenges and opportunities.
As a relatively young field, HM hasn't yet accrued a depth of senior leaders. Protected time is in shorter supply in academic medicine, as hospitalists bridge the Accreditation Council for Graduate Medical Education (ACGME) duty-hour coverage gap. In community hospitals, hospitalists are expressly hired to provide clinical coverage, and rewards for career advancement might not be included in the hospital's contract with the program. (For more on career advancement, listen to experts talk about why hospitalists make great hospital leaders.)

—Jeff Wiese, MD, SFHM, professor, associate dean for Graduate Medical Education, Tulane University Health Sciences Center, New Orleans
Each is a reason Steven B. Deitelzweig, MD, MMM, FACP, SFHM, vice president of medical affairs and system chairman of the Department of Hospital Medicine at Ochsner Health System in New Orleans, concludes: "If you rely on hospital medicine to teach you hospital medicine, you're only going to get so far in this day and age."
Big Community Ops
Working a week on/week off schedule might be the preferred goal for some hospitalists in the community setting, says Erik DeLue, MD, MBA, SFHM, hospitalist medical director for the Virtua healthcare system, which runs four hospitals in southern New Jersey. But if you find yourself asking, "Is there more out there for me?" community settings offer lots of opportunities to explore.
For example, one PhD physician in Dr. DeLue's group is parlaying her interest in research to the next level by submitting an abstract to the SHM annual meeting's poster competition regarding care coordination and fragmentation rates. Two other hospitalists share a clinical position (each works 0.5 FTE). On their off time, one works in information technology (IT) for Virtua, which pays the other half of his salary; the other does IT work for two hospital systems in New York City.
Click here to read the full story.
Searching for a new opportunity? View hundreds of jobs at SHM's Career Center.
Looking for a mentor to help guide your career development? HM presents young physicians with unique challenges and opportunities.
As a relatively young field, HM hasn't yet accrued a depth of senior leaders. Protected time is in shorter supply in academic medicine, as hospitalists bridge the Accreditation Council for Graduate Medical Education (ACGME) duty-hour coverage gap. In community hospitals, hospitalists are expressly hired to provide clinical coverage, and rewards for career advancement might not be included in the hospital's contract with the program. (For more on career advancement, listen to experts talk about why hospitalists make great hospital leaders.)

—Jeff Wiese, MD, SFHM, professor, associate dean for Graduate Medical Education, Tulane University Health Sciences Center, New Orleans
Each is a reason Steven B. Deitelzweig, MD, MMM, FACP, SFHM, vice president of medical affairs and system chairman of the Department of Hospital Medicine at Ochsner Health System in New Orleans, concludes: "If you rely on hospital medicine to teach you hospital medicine, you're only going to get so far in this day and age."
Big Community Ops
Working a week on/week off schedule might be the preferred goal for some hospitalists in the community setting, says Erik DeLue, MD, MBA, SFHM, hospitalist medical director for the Virtua healthcare system, which runs four hospitals in southern New Jersey. But if you find yourself asking, "Is there more out there for me?" community settings offer lots of opportunities to explore.
For example, one PhD physician in Dr. DeLue's group is parlaying her interest in research to the next level by submitting an abstract to the SHM annual meeting's poster competition regarding care coordination and fragmentation rates. Two other hospitalists share a clinical position (each works 0.5 FTE). On their off time, one works in information technology (IT) for Virtua, which pays the other half of his salary; the other does IT work for two hospital systems in New York City.
Click here to read the full story.