User login
The Appropriate Patient Census
What's the appropriate number of patients that an FTE hospitalist should see in one day? More than half of those surveyed on the-hospitalist.org believe they should see between 11 and 15 patients. According to two members of Team Hospitalist, 10 to 20 patients per day is a reasonable guideline.
"On average, 15 to 18 patients per day is a pretty easy-to-manage number," says Rachel George, MD, MBA, FHM, CPE, chief operating officer for Cogent HMG's west and north-central regions. But daily patient census depends on several factors, such as the types of patients admitted, the length of the doctor's shift, and the level of support from other staff on duty, she explains.
Readers were given one of five choices to respond with: "10 or fewer patients," "11-15," "16-20," "21-25," and "more than 25." Of the 421 responses, 51% felt that the average full-time hospitalist should see from 11 to 15 patients per day, followed by 35% who say they'd prefer to see 16 to 20 patients. Six percent voted for "10 or fewer." Only 4% of respondents said 20 or more patients a day was optimum.
"Honestly, I try not to get fixated on numbers," says Ken Simone, DO, SFHM, founder and president of Hospitalist and Practice Solutions in Veazie, Maine. As a consultant, he says that rather than trying to expect physicians to attend to a standard census, HM groups should focus on acuity of illness and quality of care, and let patient needs dictate the staff required. Dr. Simone also recalled working with some groups who have delegated one or more staff members to handle admitting and screening, so that hospitalists can concentrate on the patients already in beds.
What's the appropriate number of patients that an FTE hospitalist should see in one day? More than half of those surveyed on the-hospitalist.org believe they should see between 11 and 15 patients. According to two members of Team Hospitalist, 10 to 20 patients per day is a reasonable guideline.
"On average, 15 to 18 patients per day is a pretty easy-to-manage number," says Rachel George, MD, MBA, FHM, CPE, chief operating officer for Cogent HMG's west and north-central regions. But daily patient census depends on several factors, such as the types of patients admitted, the length of the doctor's shift, and the level of support from other staff on duty, she explains.
Readers were given one of five choices to respond with: "10 or fewer patients," "11-15," "16-20," "21-25," and "more than 25." Of the 421 responses, 51% felt that the average full-time hospitalist should see from 11 to 15 patients per day, followed by 35% who say they'd prefer to see 16 to 20 patients. Six percent voted for "10 or fewer." Only 4% of respondents said 20 or more patients a day was optimum.
"Honestly, I try not to get fixated on numbers," says Ken Simone, DO, SFHM, founder and president of Hospitalist and Practice Solutions in Veazie, Maine. As a consultant, he says that rather than trying to expect physicians to attend to a standard census, HM groups should focus on acuity of illness and quality of care, and let patient needs dictate the staff required. Dr. Simone also recalled working with some groups who have delegated one or more staff members to handle admitting and screening, so that hospitalists can concentrate on the patients already in beds.
What's the appropriate number of patients that an FTE hospitalist should see in one day? More than half of those surveyed on the-hospitalist.org believe they should see between 11 and 15 patients. According to two members of Team Hospitalist, 10 to 20 patients per day is a reasonable guideline.
"On average, 15 to 18 patients per day is a pretty easy-to-manage number," says Rachel George, MD, MBA, FHM, CPE, chief operating officer for Cogent HMG's west and north-central regions. But daily patient census depends on several factors, such as the types of patients admitted, the length of the doctor's shift, and the level of support from other staff on duty, she explains.
Readers were given one of five choices to respond with: "10 or fewer patients," "11-15," "16-20," "21-25," and "more than 25." Of the 421 responses, 51% felt that the average full-time hospitalist should see from 11 to 15 patients per day, followed by 35% who say they'd prefer to see 16 to 20 patients. Six percent voted for "10 or fewer." Only 4% of respondents said 20 or more patients a day was optimum.
"Honestly, I try not to get fixated on numbers," says Ken Simone, DO, SFHM, founder and president of Hospitalist and Practice Solutions in Veazie, Maine. As a consultant, he says that rather than trying to expect physicians to attend to a standard census, HM groups should focus on acuity of illness and quality of care, and let patient needs dictate the staff required. Dr. Simone also recalled working with some groups who have delegated one or more staff members to handle admitting and screening, so that hospitalists can concentrate on the patients already in beds.
By the Numbers: $4,000
According to a new study in American Economic Journal: Applied Economics by MIT economist Joseph Doyle, a $4,000 increase in per-patient hospital expenditures equates to a 1.4% decrease in mortality rates. Doyle studied 37,000 hospitalized patients in Florida who entered through the ED from 1996 to 2003. However, he focused on those visiting from other states in order to identify variation resulting from the level of care itself, not the prior health of the patients. The greater expense—and benefits—of care in the higher-cost hospital appeared to come from the broader application of ICU tools and greater complement of medical personnel, he notes.
“There are smart ways to spend money and ineffective ways to spend money,” he says, “and we’re still trying to figure out which are which, as much as possible.”
According to a new study in American Economic Journal: Applied Economics by MIT economist Joseph Doyle, a $4,000 increase in per-patient hospital expenditures equates to a 1.4% decrease in mortality rates. Doyle studied 37,000 hospitalized patients in Florida who entered through the ED from 1996 to 2003. However, he focused on those visiting from other states in order to identify variation resulting from the level of care itself, not the prior health of the patients. The greater expense—and benefits—of care in the higher-cost hospital appeared to come from the broader application of ICU tools and greater complement of medical personnel, he notes.
“There are smart ways to spend money and ineffective ways to spend money,” he says, “and we’re still trying to figure out which are which, as much as possible.”
According to a new study in American Economic Journal: Applied Economics by MIT economist Joseph Doyle, a $4,000 increase in per-patient hospital expenditures equates to a 1.4% decrease in mortality rates. Doyle studied 37,000 hospitalized patients in Florida who entered through the ED from 1996 to 2003. However, he focused on those visiting from other states in order to identify variation resulting from the level of care itself, not the prior health of the patients. The greater expense—and benefits—of care in the higher-cost hospital appeared to come from the broader application of ICU tools and greater complement of medical personnel, he notes.
“There are smart ways to spend money and ineffective ways to spend money,” he says, “and we’re still trying to figure out which are which, as much as possible.”
Transferring “Boarders” Could Save Millions
A pilot project that transferred “boarded” patients from one hospital’s ED to an inpatient bed at another nearby hospital in the same health system suggests that the concept could save hospitals millions.
“Improvement in Emergency Department Treatment Capacity: A Health System Integration Approach” was the subject of an oral presentation at HM11 in Dallas. Lead researcher Diego Martinez-Vasquez, MD, MPH, FACP, CPE, medical director for clinical resource utilization at Franklin Square Hospital Center in Baltimore, conducted the research in the University of Maryland medical system.
In the project, 265 patients who consented were transferred 1.3 miles to a sister hospital. Without “boarders”—admitted patients held in the ED—Dr. Martinez-Vasquez’s team found that the referring hospital could have regained enough capacity for an additional 2.9 patients per day. The project also showed median net revenue of $520,000 for the referring hospital and $1.9 million for the accepting hospital.
“The hospitalist group at the receiving hospital was the instrument that facilitated this program,” Dr. Martinez-Vasquez says. “But really what kept my attention was that healthcare systems sometimes don’t use resources effectively. So when I looked at the problem that this particular hospital was having regarding increased boarding time and decreased treatment capacity, I said, ‘Well, one way to resolve this issue is to develop a process that connects two system hospitals and their bed resources.’ ”
A pilot project that transferred “boarded” patients from one hospital’s ED to an inpatient bed at another nearby hospital in the same health system suggests that the concept could save hospitals millions.
“Improvement in Emergency Department Treatment Capacity: A Health System Integration Approach” was the subject of an oral presentation at HM11 in Dallas. Lead researcher Diego Martinez-Vasquez, MD, MPH, FACP, CPE, medical director for clinical resource utilization at Franklin Square Hospital Center in Baltimore, conducted the research in the University of Maryland medical system.
In the project, 265 patients who consented were transferred 1.3 miles to a sister hospital. Without “boarders”—admitted patients held in the ED—Dr. Martinez-Vasquez’s team found that the referring hospital could have regained enough capacity for an additional 2.9 patients per day. The project also showed median net revenue of $520,000 for the referring hospital and $1.9 million for the accepting hospital.
“The hospitalist group at the receiving hospital was the instrument that facilitated this program,” Dr. Martinez-Vasquez says. “But really what kept my attention was that healthcare systems sometimes don’t use resources effectively. So when I looked at the problem that this particular hospital was having regarding increased boarding time and decreased treatment capacity, I said, ‘Well, one way to resolve this issue is to develop a process that connects two system hospitals and their bed resources.’ ”
A pilot project that transferred “boarded” patients from one hospital’s ED to an inpatient bed at another nearby hospital in the same health system suggests that the concept could save hospitals millions.
“Improvement in Emergency Department Treatment Capacity: A Health System Integration Approach” was the subject of an oral presentation at HM11 in Dallas. Lead researcher Diego Martinez-Vasquez, MD, MPH, FACP, CPE, medical director for clinical resource utilization at Franklin Square Hospital Center in Baltimore, conducted the research in the University of Maryland medical system.
In the project, 265 patients who consented were transferred 1.3 miles to a sister hospital. Without “boarders”—admitted patients held in the ED—Dr. Martinez-Vasquez’s team found that the referring hospital could have regained enough capacity for an additional 2.9 patients per day. The project also showed median net revenue of $520,000 for the referring hospital and $1.9 million for the accepting hospital.
“The hospitalist group at the receiving hospital was the instrument that facilitated this program,” Dr. Martinez-Vasquez says. “But really what kept my attention was that healthcare systems sometimes don’t use resources effectively. So when I looked at the problem that this particular hospital was having regarding increased boarding time and decreased treatment capacity, I said, ‘Well, one way to resolve this issue is to develop a process that connects two system hospitals and their bed resources.’ ”
High-Performing Hospitals Invest in QI Infrastructure
A new study evaluating outcomes for hospitals participating in the American Heart Association’s Get with the Guidelines program found no correlation between high performance on adhering to measures and care standards for acute myocardial infarction and for heart failure despite overlap between the sets of care processes (J Am Coll Cardio. 2011;58:637-644).
A total of 400,000 heart patients were studied, and 283 participating hospitals were stratified into thirds based on their adherence to core quality measures for each disease, with the upper third labeled superior in performance. Lead author Tracy Wang, MD, MHS, MSc, of the Duke Clinical Research Institute in Durham, N.C., and colleagues found that superior performance for only one of the two diseases led to such end-result outcomes as in-hospital mortality that were no better than for hospitals that were not high performers for either condition. But hospitals with superior performance for both conditions had lower in-hospital mortality rates.
“Perhaps quality is more than just following checklists,” Dr. Wang says. “There’s something special about these high-performing hospitals across the board, with better QI, perhaps a little more investment in infrastructure for quality.”
This result, Dr. Wang says, should give ammunition for hospitalists and other physicians to go to their hospital administrators to request more investment in quality improvement overall, not just for specific conditions.
A new study evaluating outcomes for hospitals participating in the American Heart Association’s Get with the Guidelines program found no correlation between high performance on adhering to measures and care standards for acute myocardial infarction and for heart failure despite overlap between the sets of care processes (J Am Coll Cardio. 2011;58:637-644).
A total of 400,000 heart patients were studied, and 283 participating hospitals were stratified into thirds based on their adherence to core quality measures for each disease, with the upper third labeled superior in performance. Lead author Tracy Wang, MD, MHS, MSc, of the Duke Clinical Research Institute in Durham, N.C., and colleagues found that superior performance for only one of the two diseases led to such end-result outcomes as in-hospital mortality that were no better than for hospitals that were not high performers for either condition. But hospitals with superior performance for both conditions had lower in-hospital mortality rates.
“Perhaps quality is more than just following checklists,” Dr. Wang says. “There’s something special about these high-performing hospitals across the board, with better QI, perhaps a little more investment in infrastructure for quality.”
This result, Dr. Wang says, should give ammunition for hospitalists and other physicians to go to their hospital administrators to request more investment in quality improvement overall, not just for specific conditions.
A new study evaluating outcomes for hospitals participating in the American Heart Association’s Get with the Guidelines program found no correlation between high performance on adhering to measures and care standards for acute myocardial infarction and for heart failure despite overlap between the sets of care processes (J Am Coll Cardio. 2011;58:637-644).
A total of 400,000 heart patients were studied, and 283 participating hospitals were stratified into thirds based on their adherence to core quality measures for each disease, with the upper third labeled superior in performance. Lead author Tracy Wang, MD, MHS, MSc, of the Duke Clinical Research Institute in Durham, N.C., and colleagues found that superior performance for only one of the two diseases led to such end-result outcomes as in-hospital mortality that were no better than for hospitals that were not high performers for either condition. But hospitals with superior performance for both conditions had lower in-hospital mortality rates.
“Perhaps quality is more than just following checklists,” Dr. Wang says. “There’s something special about these high-performing hospitals across the board, with better QI, perhaps a little more investment in infrastructure for quality.”
This result, Dr. Wang says, should give ammunition for hospitalists and other physicians to go to their hospital administrators to request more investment in quality improvement overall, not just for specific conditions.
Joint Commission Launches Certification for Hospital Palliative Care
A new Joint Commission program offering advanced certification for hospital-based palliative-care services is accepting applications and conducting daylong surveys through the end of this month. As with the Joint Commission’s reviews of other specialty services (e.g. primary stroke centers), certification is narrower in scope, with service-specific evaluation of care and outcomes, than a full accreditation survey—which is an organizationwide evaluation of core processes and functions.
Advanced certification in palliative care is voluntary for the steadily growing number of acute-care hospitals offering palliative-care services (1,568, according to the latest count by the American Hospital Association), but the hospital seeking it must be accredited by the Joint Commission.1 Certification is intended for formal, defined, inpatient palliative care, whether dedicated units or consultation services, with the ability to direct clinical management of patients.
The core palliative-care team includes “licensed independent practitioners” (typically physicians), registered nurses, chaplains, and social workers.2 The service should follow palliative-care guidelines and evidence-based practice, and it must collect quality data on four performance measures—two of them clinical—and use these data to improve performance.
According to Michelle Sacco, the Joint Commission’s executive director for palliative care, evidence-based practice includes ensuring appropriate transitions to other community resources, such as hospices. She thinks the program is perfect for hospitalists, as HM increasingly is participating in palliative care in their hospitals. “This is also an opportunity to change the mindset that palliative care is for the end-stage only,” Sacco says.
Two-year certification costs $9,655, including the onsite review. For more information, visit the Joint Commission website (www.jointcommission.org/certification) or the Center to Advance Palliative Care’s site (www.capc.org).
References
- Palliative care in hospitals continues rapid growth for 10th straight year, according to latest analysis. Center to Advance Palliative Care website. Available at: www.capc.org/news-and-events/releases/07-14-11. Accessed Aug. 30, 2011.
- The National Consensus Project’s Clinical Practice Guidelines for Quality Palliative Care. The National Consensus Project website. Available at: www.nationalconsensusproject.org/. Accessed Aug. 31, 2011.
A new Joint Commission program offering advanced certification for hospital-based palliative-care services is accepting applications and conducting daylong surveys through the end of this month. As with the Joint Commission’s reviews of other specialty services (e.g. primary stroke centers), certification is narrower in scope, with service-specific evaluation of care and outcomes, than a full accreditation survey—which is an organizationwide evaluation of core processes and functions.
Advanced certification in palliative care is voluntary for the steadily growing number of acute-care hospitals offering palliative-care services (1,568, according to the latest count by the American Hospital Association), but the hospital seeking it must be accredited by the Joint Commission.1 Certification is intended for formal, defined, inpatient palliative care, whether dedicated units or consultation services, with the ability to direct clinical management of patients.
The core palliative-care team includes “licensed independent practitioners” (typically physicians), registered nurses, chaplains, and social workers.2 The service should follow palliative-care guidelines and evidence-based practice, and it must collect quality data on four performance measures—two of them clinical—and use these data to improve performance.
According to Michelle Sacco, the Joint Commission’s executive director for palliative care, evidence-based practice includes ensuring appropriate transitions to other community resources, such as hospices. She thinks the program is perfect for hospitalists, as HM increasingly is participating in palliative care in their hospitals. “This is also an opportunity to change the mindset that palliative care is for the end-stage only,” Sacco says.
Two-year certification costs $9,655, including the onsite review. For more information, visit the Joint Commission website (www.jointcommission.org/certification) or the Center to Advance Palliative Care’s site (www.capc.org).
References
- Palliative care in hospitals continues rapid growth for 10th straight year, according to latest analysis. Center to Advance Palliative Care website. Available at: www.capc.org/news-and-events/releases/07-14-11. Accessed Aug. 30, 2011.
- The National Consensus Project’s Clinical Practice Guidelines for Quality Palliative Care. The National Consensus Project website. Available at: www.nationalconsensusproject.org/. Accessed Aug. 31, 2011.
A new Joint Commission program offering advanced certification for hospital-based palliative-care services is accepting applications and conducting daylong surveys through the end of this month. As with the Joint Commission’s reviews of other specialty services (e.g. primary stroke centers), certification is narrower in scope, with service-specific evaluation of care and outcomes, than a full accreditation survey—which is an organizationwide evaluation of core processes and functions.
Advanced certification in palliative care is voluntary for the steadily growing number of acute-care hospitals offering palliative-care services (1,568, according to the latest count by the American Hospital Association), but the hospital seeking it must be accredited by the Joint Commission.1 Certification is intended for formal, defined, inpatient palliative care, whether dedicated units or consultation services, with the ability to direct clinical management of patients.
The core palliative-care team includes “licensed independent practitioners” (typically physicians), registered nurses, chaplains, and social workers.2 The service should follow palliative-care guidelines and evidence-based practice, and it must collect quality data on four performance measures—two of them clinical—and use these data to improve performance.
According to Michelle Sacco, the Joint Commission’s executive director for palliative care, evidence-based practice includes ensuring appropriate transitions to other community resources, such as hospices. She thinks the program is perfect for hospitalists, as HM increasingly is participating in palliative care in their hospitals. “This is also an opportunity to change the mindset that palliative care is for the end-stage only,” Sacco says.
Two-year certification costs $9,655, including the onsite review. For more information, visit the Joint Commission website (www.jointcommission.org/certification) or the Center to Advance Palliative Care’s site (www.capc.org).
References
- Palliative care in hospitals continues rapid growth for 10th straight year, according to latest analysis. Center to Advance Palliative Care website. Available at: www.capc.org/news-and-events/releases/07-14-11. Accessed Aug. 30, 2011.
- The National Consensus Project’s Clinical Practice Guidelines for Quality Palliative Care. The National Consensus Project website. Available at: www.nationalconsensusproject.org/. Accessed Aug. 31, 2011.
PET Scans Key to Less Radiation for Hodgkin's Lymphoma
MIAMI BEACH – Patients with Hodgkin’s lymphoma may be spared additional radiotherapy following chemotherapy if they have a negative positron-emission tomography result, investigators from the German Hodgkin Study Group reported.
The negative predictive value for FDG (18fluorodeoxyglucose)–PET at 1 year was 94%, said Dr. Rolf P. Mueller of the University of Cologne (Germany). Among patients who had residual tumors measuring 2.5 cm or greater in diameter following chemotherapy, only 4% of those who were negative for residual disease on FDG-PET scans relapsed or required additional radiotherapy, compared with 11% of FDG-PET–positive patients.
"Thus, only those advanced-stage Hodgkin lymphoma patients with residual disease who are PET-positive patients might need additional radiotherapy," Dr. Mueller said at the annual meeting of the American Society of Radiation Oncology (ASTRO).
The investigators also found a significant difference in time-to-progression favoring PET-negative patients (P =.008) with Hodgkin’s lymphoma, also known as Hodgkin’s disease.
The percentage of patients who received radiation in this clinical trial, designated GHSG (German Hodgkin Study Group) HD-15, was 11%, compared with 70% of patients in the group’s GHSG-9 trial, Mueller noted. GHSG-15 studied the role of FDG-PET for evaluating residual disease and relapse risk among patients with advanced-stage Hodgkin’s lymphoma who had undergone six to eight cycles of chemotherapy with the BEACOPP regimen (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) (J. Clin. Oncol. 2003;21:1734-9).
Early results were published in 2008 (Blood 2008;112: 3989-94). In the current report, Mueller presented data on a larger cohort.
All patients with a partial response or better and a residual mass measuring 2.5 cm or greater received FDG-PET scans. Of the 728 patients with residual disease following BEACOPP, 540 (74.2%) were PET negative, and 188 were PET positive. Mueller presented data on 701 patients who had at least 1 year of follow-up.
At 1 year, 96% (522) of PET-negative patients had neither progression nor relapse, compared with 11% of those who were PET positive. Of the PET-negative patients, 23 experienced disease progression (eight in the residual mass, six with new disease outside of the mass, and nine with progression/relapse in both areas). An additional eight PET-negative patients required additional radiotherapy.
The study was funded by the member centers of the GSHG. Dr. Mueller had no conflict of interest disclosures.
MIAMI BEACH – Patients with Hodgkin’s lymphoma may be spared additional radiotherapy following chemotherapy if they have a negative positron-emission tomography result, investigators from the German Hodgkin Study Group reported.
The negative predictive value for FDG (18fluorodeoxyglucose)–PET at 1 year was 94%, said Dr. Rolf P. Mueller of the University of Cologne (Germany). Among patients who had residual tumors measuring 2.5 cm or greater in diameter following chemotherapy, only 4% of those who were negative for residual disease on FDG-PET scans relapsed or required additional radiotherapy, compared with 11% of FDG-PET–positive patients.
"Thus, only those advanced-stage Hodgkin lymphoma patients with residual disease who are PET-positive patients might need additional radiotherapy," Dr. Mueller said at the annual meeting of the American Society of Radiation Oncology (ASTRO).
The investigators also found a significant difference in time-to-progression favoring PET-negative patients (P =.008) with Hodgkin’s lymphoma, also known as Hodgkin’s disease.
The percentage of patients who received radiation in this clinical trial, designated GHSG (German Hodgkin Study Group) HD-15, was 11%, compared with 70% of patients in the group’s GHSG-9 trial, Mueller noted. GHSG-15 studied the role of FDG-PET for evaluating residual disease and relapse risk among patients with advanced-stage Hodgkin’s lymphoma who had undergone six to eight cycles of chemotherapy with the BEACOPP regimen (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) (J. Clin. Oncol. 2003;21:1734-9).
Early results were published in 2008 (Blood 2008;112: 3989-94). In the current report, Mueller presented data on a larger cohort.
All patients with a partial response or better and a residual mass measuring 2.5 cm or greater received FDG-PET scans. Of the 728 patients with residual disease following BEACOPP, 540 (74.2%) were PET negative, and 188 were PET positive. Mueller presented data on 701 patients who had at least 1 year of follow-up.
At 1 year, 96% (522) of PET-negative patients had neither progression nor relapse, compared with 11% of those who were PET positive. Of the PET-negative patients, 23 experienced disease progression (eight in the residual mass, six with new disease outside of the mass, and nine with progression/relapse in both areas). An additional eight PET-negative patients required additional radiotherapy.
The study was funded by the member centers of the GSHG. Dr. Mueller had no conflict of interest disclosures.
MIAMI BEACH – Patients with Hodgkin’s lymphoma may be spared additional radiotherapy following chemotherapy if they have a negative positron-emission tomography result, investigators from the German Hodgkin Study Group reported.
The negative predictive value for FDG (18fluorodeoxyglucose)–PET at 1 year was 94%, said Dr. Rolf P. Mueller of the University of Cologne (Germany). Among patients who had residual tumors measuring 2.5 cm or greater in diameter following chemotherapy, only 4% of those who were negative for residual disease on FDG-PET scans relapsed or required additional radiotherapy, compared with 11% of FDG-PET–positive patients.
"Thus, only those advanced-stage Hodgkin lymphoma patients with residual disease who are PET-positive patients might need additional radiotherapy," Dr. Mueller said at the annual meeting of the American Society of Radiation Oncology (ASTRO).
The investigators also found a significant difference in time-to-progression favoring PET-negative patients (P =.008) with Hodgkin’s lymphoma, also known as Hodgkin’s disease.
The percentage of patients who received radiation in this clinical trial, designated GHSG (German Hodgkin Study Group) HD-15, was 11%, compared with 70% of patients in the group’s GHSG-9 trial, Mueller noted. GHSG-15 studied the role of FDG-PET for evaluating residual disease and relapse risk among patients with advanced-stage Hodgkin’s lymphoma who had undergone six to eight cycles of chemotherapy with the BEACOPP regimen (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) (J. Clin. Oncol. 2003;21:1734-9).
Early results were published in 2008 (Blood 2008;112: 3989-94). In the current report, Mueller presented data on a larger cohort.
All patients with a partial response or better and a residual mass measuring 2.5 cm or greater received FDG-PET scans. Of the 728 patients with residual disease following BEACOPP, 540 (74.2%) were PET negative, and 188 were PET positive. Mueller presented data on 701 patients who had at least 1 year of follow-up.
At 1 year, 96% (522) of PET-negative patients had neither progression nor relapse, compared with 11% of those who were PET positive. Of the PET-negative patients, 23 experienced disease progression (eight in the residual mass, six with new disease outside of the mass, and nine with progression/relapse in both areas). An additional eight PET-negative patients required additional radiotherapy.
The study was funded by the member centers of the GSHG. Dr. Mueller had no conflict of interest disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR RADIATION ONCOLOGY
Major Finding: FDG-PET scans following chemotherapy in patients with advanced-stage Hodgkin’s lymphoma have a negative predictive value of 94%.
Data Source: The prospective GHSG HD-15 trial involving 701 patients.
Disclosures: The study was funded by the GSHG. Dr. Mueller had no conflict of interest disclosures.
Academic Hospitalists Gear Up for Learning
The challenges of academic HM are different from other sectors of the specialty. Academic hospitalists, division chiefs, and administrators at academic teaching hospitals contend with the pressure of receiving grants, presenting at grand rounds, and reserving time for research and educational projects.
While it can be overwhelming, especially for academic hospitalists early in their careers, the Academic Hospitalist Academy helps untangle those challenges and turn them into long-term professional opportunities. Hosted jointly by SHM, the Society of General Internal Medicine (SGIM), and the Association of Chiefs and Leaders of General Internal Medicine (ACLGIM), the academy is a three-day course dedicated to education, scholarship, and professional success for academic hospitalists.
In addition to helping them become better hospitalists, Academic Hospitalist Academy uses didactic sessions, small-group exercises, and other interactive techniques to help academic hospitalists become better teachers, create and publish scholarly work, and get first in line for promotions.
Now in its third year, Academic Hospitalist Academy is consistently met with rave reviews from attendees. According to evaluations from the 2010 academy, attendees unanimously felt the course was worth their time and money; 99% said they would recommend it to a colleague.
The challenges of academic HM are different from other sectors of the specialty. Academic hospitalists, division chiefs, and administrators at academic teaching hospitals contend with the pressure of receiving grants, presenting at grand rounds, and reserving time for research and educational projects.
While it can be overwhelming, especially for academic hospitalists early in their careers, the Academic Hospitalist Academy helps untangle those challenges and turn them into long-term professional opportunities. Hosted jointly by SHM, the Society of General Internal Medicine (SGIM), and the Association of Chiefs and Leaders of General Internal Medicine (ACLGIM), the academy is a three-day course dedicated to education, scholarship, and professional success for academic hospitalists.
In addition to helping them become better hospitalists, Academic Hospitalist Academy uses didactic sessions, small-group exercises, and other interactive techniques to help academic hospitalists become better teachers, create and publish scholarly work, and get first in line for promotions.
Now in its third year, Academic Hospitalist Academy is consistently met with rave reviews from attendees. According to evaluations from the 2010 academy, attendees unanimously felt the course was worth their time and money; 99% said they would recommend it to a colleague.
The challenges of academic HM are different from other sectors of the specialty. Academic hospitalists, division chiefs, and administrators at academic teaching hospitals contend with the pressure of receiving grants, presenting at grand rounds, and reserving time for research and educational projects.
While it can be overwhelming, especially for academic hospitalists early in their careers, the Academic Hospitalist Academy helps untangle those challenges and turn them into long-term professional opportunities. Hosted jointly by SHM, the Society of General Internal Medicine (SGIM), and the Association of Chiefs and Leaders of General Internal Medicine (ACLGIM), the academy is a three-day course dedicated to education, scholarship, and professional success for academic hospitalists.
In addition to helping them become better hospitalists, Academic Hospitalist Academy uses didactic sessions, small-group exercises, and other interactive techniques to help academic hospitalists become better teachers, create and publish scholarly work, and get first in line for promotions.
Now in its third year, Academic Hospitalist Academy is consistently met with rave reviews from attendees. According to evaluations from the 2010 academy, attendees unanimously felt the course was worth their time and money; 99% said they would recommend it to a colleague.
HM12 Research and Award Submissions Deadline Nears
Hospitalists interested in promoting their research still have time to submit applications for SHM’s Research, Innovation, and Clinical Vignettes (RIV) competition. RIV abstracts will be presented at HM12 in San Diego.
SHM also is accepting nominations for its annual awards program, which honors hospitalists who demonstrate excellence in clinical work, teaching, scholarly research, and service to the specialty.
Applications for both programs can be obtained at www.hospitalmedicine.org. The deadline for SHM’s annual award submissions is Nov. 1; applications for RIV abstracts will be accepted until Dec. 1.
Both sets of awards will be presented live on stage at HM12.
The annual awards often are a precursor to even more prestige within the specialty. In 2005, SHM’s immediate past president Jeff Wiese, MD, SFHM, FACP won the “Excellence in Teaching” award, SHM president Joseph Ming-Wah Li, MD, SFHM, won for “Outstanding Service in Hospital Medicine,” and SHM president-elect Shaun Frost, MD, won for “Clinical Excellence.”
Hospitalists interested in promoting their research still have time to submit applications for SHM’s Research, Innovation, and Clinical Vignettes (RIV) competition. RIV abstracts will be presented at HM12 in San Diego.
SHM also is accepting nominations for its annual awards program, which honors hospitalists who demonstrate excellence in clinical work, teaching, scholarly research, and service to the specialty.
Applications for both programs can be obtained at www.hospitalmedicine.org. The deadline for SHM’s annual award submissions is Nov. 1; applications for RIV abstracts will be accepted until Dec. 1.
Both sets of awards will be presented live on stage at HM12.
The annual awards often are a precursor to even more prestige within the specialty. In 2005, SHM’s immediate past president Jeff Wiese, MD, SFHM, FACP won the “Excellence in Teaching” award, SHM president Joseph Ming-Wah Li, MD, SFHM, won for “Outstanding Service in Hospital Medicine,” and SHM president-elect Shaun Frost, MD, won for “Clinical Excellence.”
Hospitalists interested in promoting their research still have time to submit applications for SHM’s Research, Innovation, and Clinical Vignettes (RIV) competition. RIV abstracts will be presented at HM12 in San Diego.
SHM also is accepting nominations for its annual awards program, which honors hospitalists who demonstrate excellence in clinical work, teaching, scholarly research, and service to the specialty.
Applications for both programs can be obtained at www.hospitalmedicine.org. The deadline for SHM’s annual award submissions is Nov. 1; applications for RIV abstracts will be accepted until Dec. 1.
Both sets of awards will be presented live on stage at HM12.
The annual awards often are a precursor to even more prestige within the specialty. In 2005, SHM’s immediate past president Jeff Wiese, MD, SFHM, FACP won the “Excellence in Teaching” award, SHM president Joseph Ming-Wah Li, MD, SFHM, won for “Outstanding Service in Hospital Medicine,” and SHM president-elect Shaun Frost, MD, won for “Clinical Excellence.”
How Hospitalists Can Team with Nursing to Improve Patient Care
Establishing mutual respect and trust between hospitalists and nurses is an important part of ensuring patient safety, whether you’re on your first job or your 20th, says Angela Beck, RN, director of critical-care services at Nebraska Medical Center in Omaha.
“Nurses are important coordinators of care,” she says. “Recognizing and valuing nurses for that is truly the most important thing for the patient, and can also help hospitalists build relationships.”
Key Partners
Forming a collaborative relationship with the nursing service might depend on where you start. At Northwestern Memorial Hospital in Chicago, the nursing service enjoys a “close and collaborative relationship” with hospitalists, according to Kristin Ramsey, RN, MSN, MPPM, NE-BC, associate chief nurse and executive director of operations. New hospitalists are oriented to the care-delivery models on the inpatient care units. In addition, hospitalists are acculturated into the hospital’s coleadership model.
“We have partnered with our hospitalists to create a model in which the physician and nurse leader collaboratively lead the development of multidisciplinary, subspecialty teams to ensure quality outcomes,” Ramsey says. “The model is so successful with the hospitalists that we are now extending it to other areas in the organization.”
Round Sharing
Absent a formalized training protocol for partnering with nursing, hospitalists still can learn a great deal by listening to and communicating with the nursing staff, says Connie Ogden, RN, MSN, NEA-BC, executive director of adult acute services at Nebraska Medical Center. “Nurses are there around the clock caring for patients and may have a different insight” about patients’ evolving conditions, she says.
Care for the patient improves if everyone is on the same page, Ogden adds. That’s why it makes sense, she says, to include nurses during rounds. Beck agrees: “If nurses aren’t there to hear how the plan of care comes about, there is no reason to believe they can effectively describe it once the physician turns around and walks away to see another patient.”
In critical-care units, according to Beck, nurses can function as a bridge between patients and physicians. For example, they can help patients define and express their goals. Some of these goals can be incremental, she notes, such as “I really want to get out of bed this afternoon,” or “I really want my family here to listen to this message.”
Different Role, Same Goal
As director of adult acute services, Ogden often receives complaints from physicians about calls they receive from nurses. Often, these calls emanate from a concern for the patient (e.g. a 2 a.m. call for a Tylenol order to address a headache) or from the requirement that nurses follow policy and clarify orders. If hospitalists understand the back story of the call, their perception of its purpose can change.
Although there have been strides toward better nurse-physician collaboration, “we still have a lot of opportunities for improvement,” Beck asserts.
Establishing mutual respect and trust is not an overnight accomplishment. As Ogden explains, physicians and nurses have different roles, but they share the same goal: quality outcomes in patient care.
Gretchen Henkel is a freelance writer based in southern California.
Best Ways to Improve Hospitalist-Nursing Collaboration
“A good portion of nurses are relationship builders,” says Beck, director of critical-care services at Nebraska Medical Center. She urges hospitalists on a new job to just “be physically present, in the beginning, on inpatient units” whenever possible. “Acting like you care is really important, and nurses will respond to that,” she says. “You can create an environment in which nurses’ feedback is valued.”
She also recommends, especially for new hospitalists, Dr. Peter J. Pronovost’s three-part talk “The Science of Safety,” delivered to incoming residents at Johns Hopkins University Medical Center in Baltimore, where Dr. Provonost is medical director of the quality and safety research group.—GH
Establishing mutual respect and trust between hospitalists and nurses is an important part of ensuring patient safety, whether you’re on your first job or your 20th, says Angela Beck, RN, director of critical-care services at Nebraska Medical Center in Omaha.
“Nurses are important coordinators of care,” she says. “Recognizing and valuing nurses for that is truly the most important thing for the patient, and can also help hospitalists build relationships.”
Key Partners
Forming a collaborative relationship with the nursing service might depend on where you start. At Northwestern Memorial Hospital in Chicago, the nursing service enjoys a “close and collaborative relationship” with hospitalists, according to Kristin Ramsey, RN, MSN, MPPM, NE-BC, associate chief nurse and executive director of operations. New hospitalists are oriented to the care-delivery models on the inpatient care units. In addition, hospitalists are acculturated into the hospital’s coleadership model.
“We have partnered with our hospitalists to create a model in which the physician and nurse leader collaboratively lead the development of multidisciplinary, subspecialty teams to ensure quality outcomes,” Ramsey says. “The model is so successful with the hospitalists that we are now extending it to other areas in the organization.”
Round Sharing
Absent a formalized training protocol for partnering with nursing, hospitalists still can learn a great deal by listening to and communicating with the nursing staff, says Connie Ogden, RN, MSN, NEA-BC, executive director of adult acute services at Nebraska Medical Center. “Nurses are there around the clock caring for patients and may have a different insight” about patients’ evolving conditions, she says.
Care for the patient improves if everyone is on the same page, Ogden adds. That’s why it makes sense, she says, to include nurses during rounds. Beck agrees: “If nurses aren’t there to hear how the plan of care comes about, there is no reason to believe they can effectively describe it once the physician turns around and walks away to see another patient.”
In critical-care units, according to Beck, nurses can function as a bridge between patients and physicians. For example, they can help patients define and express their goals. Some of these goals can be incremental, she notes, such as “I really want to get out of bed this afternoon,” or “I really want my family here to listen to this message.”
Different Role, Same Goal
As director of adult acute services, Ogden often receives complaints from physicians about calls they receive from nurses. Often, these calls emanate from a concern for the patient (e.g. a 2 a.m. call for a Tylenol order to address a headache) or from the requirement that nurses follow policy and clarify orders. If hospitalists understand the back story of the call, their perception of its purpose can change.
Although there have been strides toward better nurse-physician collaboration, “we still have a lot of opportunities for improvement,” Beck asserts.
Establishing mutual respect and trust is not an overnight accomplishment. As Ogden explains, physicians and nurses have different roles, but they share the same goal: quality outcomes in patient care.
Gretchen Henkel is a freelance writer based in southern California.
Best Ways to Improve Hospitalist-Nursing Collaboration
“A good portion of nurses are relationship builders,” says Beck, director of critical-care services at Nebraska Medical Center. She urges hospitalists on a new job to just “be physically present, in the beginning, on inpatient units” whenever possible. “Acting like you care is really important, and nurses will respond to that,” she says. “You can create an environment in which nurses’ feedback is valued.”
She also recommends, especially for new hospitalists, Dr. Peter J. Pronovost’s three-part talk “The Science of Safety,” delivered to incoming residents at Johns Hopkins University Medical Center in Baltimore, where Dr. Provonost is medical director of the quality and safety research group.—GH
Establishing mutual respect and trust between hospitalists and nurses is an important part of ensuring patient safety, whether you’re on your first job or your 20th, says Angela Beck, RN, director of critical-care services at Nebraska Medical Center in Omaha.
“Nurses are important coordinators of care,” she says. “Recognizing and valuing nurses for that is truly the most important thing for the patient, and can also help hospitalists build relationships.”
Key Partners
Forming a collaborative relationship with the nursing service might depend on where you start. At Northwestern Memorial Hospital in Chicago, the nursing service enjoys a “close and collaborative relationship” with hospitalists, according to Kristin Ramsey, RN, MSN, MPPM, NE-BC, associate chief nurse and executive director of operations. New hospitalists are oriented to the care-delivery models on the inpatient care units. In addition, hospitalists are acculturated into the hospital’s coleadership model.
“We have partnered with our hospitalists to create a model in which the physician and nurse leader collaboratively lead the development of multidisciplinary, subspecialty teams to ensure quality outcomes,” Ramsey says. “The model is so successful with the hospitalists that we are now extending it to other areas in the organization.”
Round Sharing
Absent a formalized training protocol for partnering with nursing, hospitalists still can learn a great deal by listening to and communicating with the nursing staff, says Connie Ogden, RN, MSN, NEA-BC, executive director of adult acute services at Nebraska Medical Center. “Nurses are there around the clock caring for patients and may have a different insight” about patients’ evolving conditions, she says.
Care for the patient improves if everyone is on the same page, Ogden adds. That’s why it makes sense, she says, to include nurses during rounds. Beck agrees: “If nurses aren’t there to hear how the plan of care comes about, there is no reason to believe they can effectively describe it once the physician turns around and walks away to see another patient.”
In critical-care units, according to Beck, nurses can function as a bridge between patients and physicians. For example, they can help patients define and express their goals. Some of these goals can be incremental, she notes, such as “I really want to get out of bed this afternoon,” or “I really want my family here to listen to this message.”
Different Role, Same Goal
As director of adult acute services, Ogden often receives complaints from physicians about calls they receive from nurses. Often, these calls emanate from a concern for the patient (e.g. a 2 a.m. call for a Tylenol order to address a headache) or from the requirement that nurses follow policy and clarify orders. If hospitalists understand the back story of the call, their perception of its purpose can change.
Although there have been strides toward better nurse-physician collaboration, “we still have a lot of opportunities for improvement,” Beck asserts.
Establishing mutual respect and trust is not an overnight accomplishment. As Ogden explains, physicians and nurses have different roles, but they share the same goal: quality outcomes in patient care.
Gretchen Henkel is a freelance writer based in southern California.
Best Ways to Improve Hospitalist-Nursing Collaboration
“A good portion of nurses are relationship builders,” says Beck, director of critical-care services at Nebraska Medical Center. She urges hospitalists on a new job to just “be physically present, in the beginning, on inpatient units” whenever possible. “Acting like you care is really important, and nurses will respond to that,” she says. “You can create an environment in which nurses’ feedback is valued.”
She also recommends, especially for new hospitalists, Dr. Peter J. Pronovost’s three-part talk “The Science of Safety,” delivered to incoming residents at Johns Hopkins University Medical Center in Baltimore, where Dr. Provonost is medical director of the quality and safety research group.—GH
Cost‐Related Medication Underuse
The affordability of prescription medications continues to be one of the most pressing public health issues in the United States. Many patients reduce their prescribed doses to make medications last longer or do not fill prescriptions because of cost.1 Cost‐related medication underuse affects patients with and without drug insurance coverage,2 and is likely to become even more problematic as employers scale back on drug benefits3 and drug prices continue to increase.4 The landmark Patient Protection and Affordability Act passed in March 2010 does little to address this issue.5
Existing estimates of cost‐related medication underuse come largely from surveys of ambulatory patients. For example, using data from the Medicare Current Beneficiary Survey, Maden et al. estimated that 11% to 15% of patients reduced medication use in the past year because of cost.6 Tseng and colleagues found very similar rates of cost‐related underuse in managed care beneficiaries with diabetes.7
Hospitalized patients, who have a high burden of disease and tend to use more medications than their ambulatory counterparts, may be particularly vulnerable to cost‐related underuse but, thus far, have been subject to little investigation. New medications, which are frequently prescribed at the time of discharge, may exacerbate these issues further and contribute to preventable readmissions. Accordingly, we surveyed a cohort of medical inpatients at a large academic medical center to estimate the prevalence and predictors of cost‐related medication underuse for hospitalized managed care patients, and to identify strategies that patients perceive as helpful to make medications more affordable.
METHODS
Study Sample
We identified consecutive patients newly admitted to the general medicine, cardiology, or oncology services at Brigham and Women's Hospital from November 2008 to December 2009. For our survey, we included only those patients who received medical benefits through 1 of 3 large insurers with whom our hospital has pay‐for‐performance contracts. Annually, there are approximately 4000 patients covered by these insurers admitted to the 3 clinical services we evaluated, We focused on patients who had a primary care physician at one of the hospital's outpatient practices because of the existence of an automated infrastructure to identify these managed care beneficiaries of these insurers who are newly hospitalized, and because patients covered by commercial insurance plans likely represent a conservative lower‐bound of cost‐related medication underuse among hospitalized patients.
Patients were surveyed on the first non‐holiday weekday after admission. We excluded patients who had been discharged prior to the daily admission list being generated, or who, on a previous admission, had completed our survey or declined to be surveyed. We also excluded several patients who were not beneficiaries of the target insurers and were erroneously included on the managed care admission roster.
Potentially eligible patients were approached on the hospital ward by 1 of 3 study care coordinators (2 nurses and 1 pharmacist) and were asked if they were willing to participate in a research project about medication use that involved a short verbally delivered in‐person (inpatient) survey, a brief postdischarge telephone call, and a review of their electronic health record. The Institutional Review Board of Brigham and Women's Hospital approved this study.
Inpatient Survey
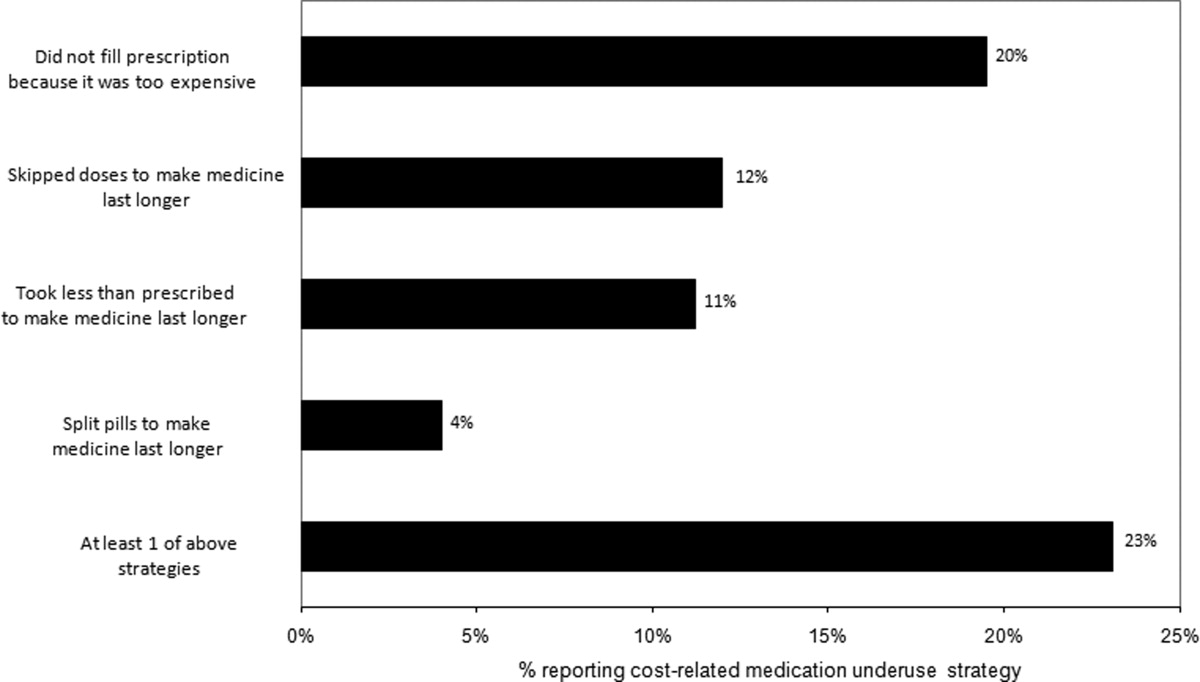
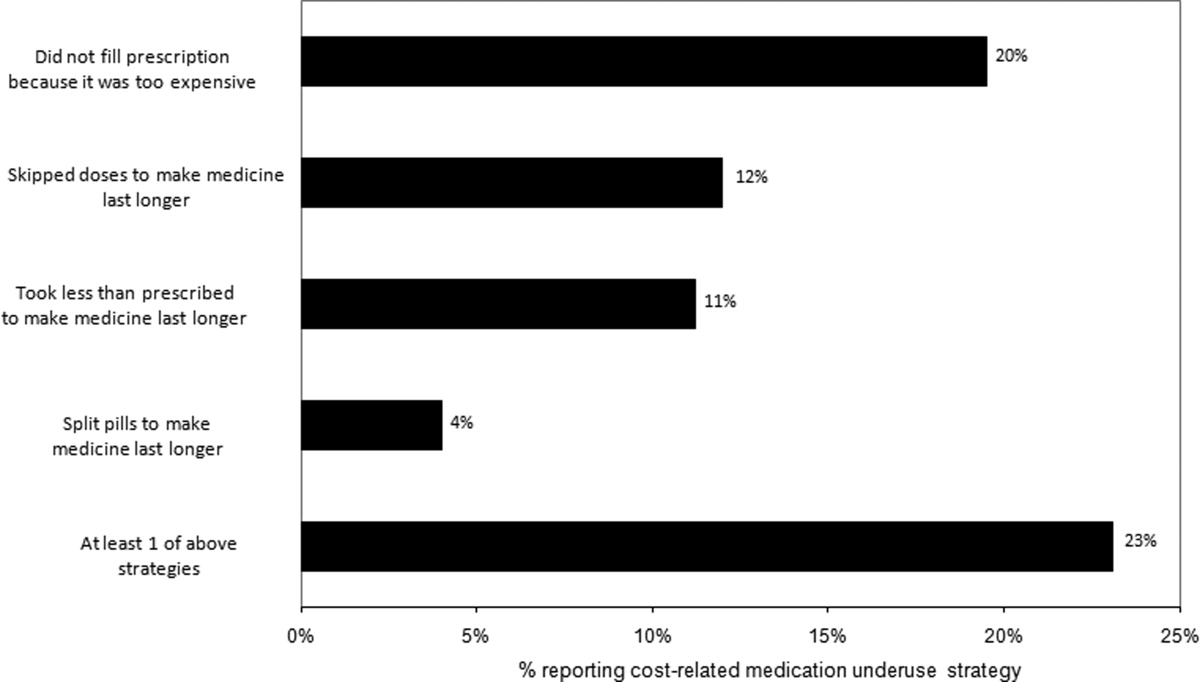
Our survey instrument was developed iteratively and pilot‐tested to improve face validity. Questions about cost‐related underuse were based on validated measures.8, 9 Specifically, we asked whether in the past year patients had: (1) not filled a prescription because it was too expensive, (2) skipped doses to make medicines last longer, (3) took less medicine than prescribed to make the medicine last longer, or (4) split pills to make the medication last longer.
Questions about strategies to improve medication affordability assessed whether patients thought it would be helpful to: (1) discuss medication affordability with healthcare workers (inpatient doctors, outpatient doctors, nurses, pharmacists, or social workers); (2) have their medications reviewed by a nurse or pharmacist; (3) receive information about lower cost but equally effective medication options, or about programs that provide medications at reduced costs; and/or (4) have their copayments/coinsurance lowered. Possible responses to all of these questions were binary, ie, yes or no.
In addition, patients were asked about the nature of their drug insurance coverage, the prescription medications that they currently use, whether they know their copayment levels (for generic and brand‐name medications), and, if so, what these amounts were, their annual household income, and their self‐identified race. Information on patient age, gender, and the primary reason for hospitalization was obtained from the electronic health record. This source was also used to verify the accuracy of the self‐reported preadmission medication list. When there were discrepancies between preadmission medications reported by patients and those recorded in their chart, the later was used because our hospital reconciles and records all medications at the time of hospital admission for all patients.
Postdischarge Survey
Within 3 days of discharge, patients were contacted by telephone and asked about new medications they were prescribed on discharge, if any. The discharge summary was used to verify the accuracy of the information provided by patients. The interviewers clarified any apparent discrepancies between the 2 sources of information with the patient. Patients who had been prescribed a new medication were asked whether or not they had filled their prescription. For patients who had, we asked whether: (1) they knew how much they would have to pay prior to going to the pharmacy, (2) they had discussed less expensive options with their pharmacist, and (3) they had discussed medication costs with their inpatient or outpatient physicians.
Data Analysis
We used descriptive statistics to summarize the characteristics of our respondents and our overall survey results. We generated univariate and multivariable logistic regression models to identify whether prehospitalization cost‐related medication underuse was influenced by patient age, gender, income, race, and the number of medications patients used on a regular basis. For the purpose of these analyses, we classified patients as reporting cost‐related underuse if they responded yes to any of the 4 strategies described above (ie, not filling medications, skipping doses, taking less medication, or splitting pills to make medicines last longer). Patients whose incomes were above the median level in our cohort were categorized as being of high‐income. Our multivariable model had a c‐statistic of 0.75, suggesting good discriminative ability.
RESULTS
During the study period, 483 potentially‐eligible patients were admitted to the general medicine, cardiology, and oncology services. We excluded 167 because they had been discharged prior to being identified, had been surveyed or already declined participation on a prior admission, or were not managed care enrollees (see Appendix A). Of the remaining 316 subjects, 130 participated in the inpatient survey (response rate = 41%); 93 (75%) of these patients were reached by telephone after hospital discharge and completed the postdischarge survey. The baseline characteristics of our respondents are presented in Table 1. Patients had a mean age of 52 years, were 50% male and two‐thirds of white race, represented a range of household incomes, and almost all had employer‐sponsored prescription coverage. Prior to admission, patients took an average of 5 prescription medications and paid an average copayment of $10.80 and $21.60 for each generic and brand‐name prescription, respectively.
| Characteristic | N = 130 |
|---|---|
| |
| Age, mean years (SD) | 52 (11.2) |
| Male, % | 65 (50.0) |
| Race/ethnicity,* n (%) | |
| Caucasian/white | 84 (67.2) |
| Black/African American | 20 (16.0) |
| Latino/Hispanic | 13 (10.4) |
| Asian | 3 (2.4) |
| American Indian or Alaska Native | 1 (0.8) |
| Other | 4 (3.2) |
| Annual household income,* n (%) | |
| <$30,000 | 15 (12.8) |
| $30,000‐$75,000 | 49 (41.9) |
| >$75,000 | 53 (45.3) |
| Insurance coverage for outpatient prescription drugs,* n (%) | |
| Employer or spouse's employer | 123 (96.0) |
| Independent | 5 (3.9) |
| Medication copayments,* mean $ (SD) | |
| Brand‐name medications | 21.6 (14.2) |
| Generic medications | 10.8 (6.0) |
| No. of medications prior to admission, mean (SD) | 5.5 (4.3) |
| Category of discharge diagnosis, n (%) | |
| Cardiovascular | 40 (30.8) |
| Gastrointestinal | 23 (17.7) |
| Pulmonary | 23 (17.7) |
| Infectious | 13 (10.0) |
| Oncology | 5 (3.8) |
| Renal | 6 (4.6) |
| Psychiatric | 3 (2.3) |
| Hematologic | 4 (3.1) |
| Neurologic | 5 (3.8) |
| Musculoskeletal | 5 (3.8) |
| Respiratory | 2 (1.5) |
| Endocrine | 1 (0.8) |
Cost‐Related Medication Underuse
Thirty (23%) of the survey respondents reported at least 1 cost‐related medication underuse strategy in the year prior to their hospital admission (Figure 1), most commonly not filling a prescription at all because of cost (n = 26; 20%). Rates of cost‐related underuse were highest for patients of black race, low income, and women (Figure 2).
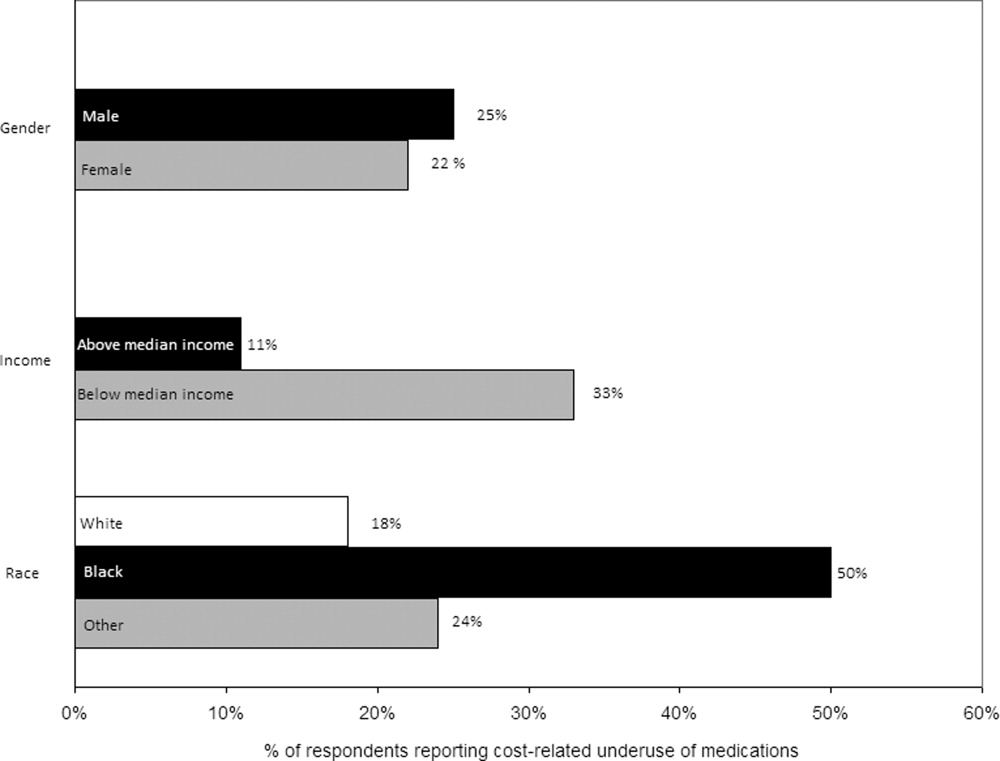
In unadjusted analyses, black respondents had 4.60 (95% confidence interval [CI], 1.63 to 13.0) times the odds of reporting cost‐related underuse than non‐Hispanic white respondents (Table 2). The association of black race and cost‐related underuse appears to be confounded, in part, by income (adjusted odds ratio for black race was 4.16; 95% CI, 1.34 to 12.86) and the number of medications patients used on a regular basis (adjusted odds ratio for black race was 4.14; 95% CI, 1.44 to 11.96). After controlling for these variables, as well as age and gender, the relationship between race and cost‐related underuse remained statistically significant (adjusted odds ratio 3.39; 95% CI, 1.05 to 11.02) (Table 2).
| Predictor | Unadjusted Odds Ratio (95% CI) | Adjusted Odds Ratio (95% CI) |
|---|---|---|
| ||
| Age (per additional year) | 0.98 (0.941.02) | 0.97 (0.931.01) |
| Male (vs female) | 0.84 (0.371.90) | 1.03 (0.432.48) |
| Race (vs white race) | ||
| Black | 4.60 (1.6313.0) | 3.39 (1.0511.02) |
| Other | 1.10 (0.363.37) | 0.77 (0.202.99) |
| No. of medications (per additional medication) | 1.10 (1.001.20) | 1.10 (1.001.22) |
| High income (vs low income) | 0.62 (0.271.42) | 0.71 (0.242.07) |
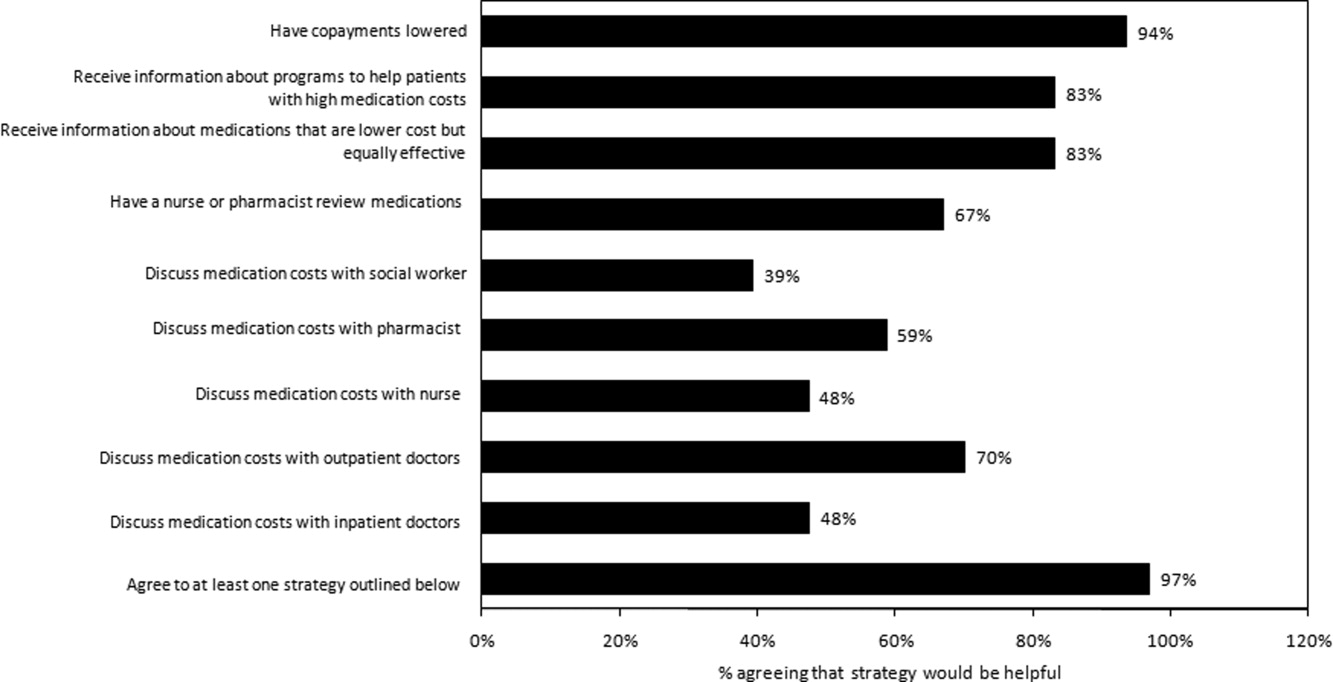
Strategies to Help Make Medications More Affordable
Virtually all respondents (n = 123; 95%) endorsed at least one of the proposed strategies to make medications more affordable (Figure 3). A majority felt that lowering cost sharing (94%), or receiving information about lower‐cost medication options (83%) or programs to subsidize medication costs (83%) would be helpful. Approximately 70% of patients stated that speaking to their outpatient physicians might be helpful, although only 14% reported actually speaking with their primary care provider about medication costs in the past year. Results were mixed for other strategies, including speaking with their inpatient physicians.
Postdischarge Medication Use
Seventy‐six (82%) respondents to the outpatient survey were prescribed a new medication at the time of hospital discharge, and virtually all (95%) had filled prescriptions for these medications by the time of the follow‐up survey. Patients paid an average of $27.63 (standard deviation $39.24) in out‐of‐pocket costs for these medications. Few (16%) patients knew how much they would have to pay before they had gone to the pharmacy to fill their prescription (see Appendix B). Even fewer patients asked, or were spoken to by their pharmacist, about less expensive medication options (7%), and almost none had spoken to their inpatient (4%) or outpatient providers (2%) about the cost of their newly prescribed drugs.
DISCUSSION
Almost a quarter of the medical inpatients we surveyed had not filled a medication because of cost, or had skipped doses, reduced dosages, or split pills to make their medicines last longer in the prior year. This amount is larger than that found in many prior studies, conducted in outpatient settings, in which 11% to 19% of patients report cost‐related underuse.68, 10, 11 Our results are particularly striking considering that our study cohort consisted exclusively of patients with commercial health insurance, the vast majority of whom also had employer‐sponsored drug coverage. Cost‐related medication underuse may be even more prevalent among hospitalized patients with less generous benefits, including the uninsured and perhaps even beneficiaries of Medicare Part D.
Reductions in medication use because of cost were particularly high among black patients, whose odds of reporting cost‐related underuse were more than 3 times higher than that of patients of non‐Hispanic white race. Race‐related differences in cost‐related underuse have been observed in outpatient studies,68, 12 and may be an important contributor to racial disparities in evidence‐based medication use.1315 These differences may, in part, reflect racial variations in socioeconomic status; lower income patients, who are more likely to be from a racial or ethnic minority, are more sensitive to cost sharing than higher income individuals.16 Consistent with this, the relationship between race and cost‐related underuse in our study was smaller but still highly significant in multivariable models that adjusted for income.
Not surprisingly, the underuse of effective prescription medications is associated with adverse clinical and economic consequences.17 Heisler et al. found that patients who had restricted medications because of cost were 76% more likely to report a decline in their health status than those who had not.18 The health effects of cost‐related underuse are likely to be particularly significant for hospitalized patients, given their high burden of disease and the frequency with which they are prescribed medications at discharge to treat the condition that led to their initial hospitalization. Thus, targeting efforts to address cost‐related underuse patients who are hospitalized may be an efficient method of improving patient health and reducing preventable readmissions. This is consistent with efforts that address, in the inpatient setting, other health issues that are commonly encountered in the ambulatory arena, such as immunizations and smoking cessation.19
Our survey respondents endorsed numerous strategies as being potentially helpful. Predictably, support for lowering copayments was extremely high. While this may not be practical or even desirable for some medications, lowering copayments for highly effective medications, such as statins and antihypertensives, in the context of value‐based insurance design, is an increasingly adopted strategy that has the potential to simultaneously improve clinical outcomes and reduce overall health spending.20, 21
While the majority of patients felt that talking to their outpatient physicians or pharmacists about medication costs might be helpful, the effectiveness of this strategy is unclear. Consistent with prior results,22, 23 the vast majority of the patients we surveyed had not discussed medication costs prior to their admission or after filling newly prescribed medications. Further, although physicians could help reduce drug expenditures in a variety of ways, including the increased ordering of generic drugs,24 many physicians are uncomfortable talking to their patients about costs,25 have limited knowledge about their patients' out‐of‐pocket expenditures, feel that addressing this issue is not their responsibility,26 or do not have resources, such as electronic formulary information, that could facilitate these discussions in an efficient manner.
An alternative strategy may be to provide patients with better education about medication costs. Virtually none of the patients we surveyed knew how much they would pay for their new prescriptions before visiting the pharmacy. These findings are similar to those observed in the outpatient setting,27 and suggest an opportunity to provide patients with information about the cost of their newly and previously prescribed drugs, and to facilitate discussions between patients and inpatient providers about predischarge prescribing decisions, in the same spirit as other predischarge patient education.28 Of course, issues related to transitions of care between the hospital and community setting, and coordination between inpatient and outpatient providers, must be adequately addressed for this strategy to be effective.
Our study has several notable limitations. It had a relatively small sample size and low response rate. Respondents may have differed systematically from non‐respondents, and we were unable to compare the characteristics of both populations. Further, we studied commercially insured inpatients on internal medicine services at an academic medical center, and thus our results may not be generalizable to patients hospitalized in other settings, or with different types of insurance coverage, including the uninsured. The primary outcome of our study was to determine self‐reported cost‐related underuse. While we used validated measures,8 it is possible that patients who reported reducing their medication use in response to cost may not have actually done so. We did not collect information on education or health literacy, nor did we have access to detailed information about our respondents' pharmacy benefit design structures; these important factors may have confounded our analyses, and/or may have been mediators of our observed results, and should be evaluated further in future studies. We did not have adequate statistical power to evaluate whether patients using specific classes of medications were particularly prone to cost‐related underuse.
Despite these limitations, our study is the first, to our knowledge, to evaluate the impact of medication costs on use in a cohort of hospitalized individuals. The high levels of cost‐related underuse that we observed is concerning. Our results support calls for the further development of interventions to address high medication costs and for the consideration of novel approaches to assist patients around the time of hospital discharge.
APPENDICES
APPENDIX A. Survey response flow diagram.
APPENDIX B. Behaviors to address the cost of medications prescribed at hospital discharge.
- USA Today/Kaiser Family Foundation/Harvard School of Public Health.The Public on Prescription Drugs and Pharmaceutical Companies.2008. Available at: http://www.kff.org/kaiserpolls/pomr030408pkg.cfm. Accessed September 5, 2008.
- ,,, et al.Pharmacy benefits and the use of drugs by the chronically ill.JAMA.2004;291(19):2344–2350.
- Kaiser Family Foundation and Health Research and Educational Trust.Employer Health Benefits Annual Survey,2009.year="2009"2009. Available at: http://ehbs.kff.org/pdf/2009/7936.pdf. Accessed May 5,year="2010"2010.
- Kaiser Family Foundation.Prescription Drug Trends.2007. Available at: http://www.kff.org/rxdrugs/upload/3057_06.pdf. Accessed December 5,year="2007"2007.
- The Patient Protection and Affordable Care Act, H.R. 3590, Section 2713 (c).Washington, DC:111 Congress;2010.
- ,,, et al.Cost‐related medication nonadherence and spending on basic needs following implementation of Medicare Part D.JAMA.2008;299(16):1922–1928.
- ,,, et al.Race/ethnicity and economic differences in cost‐related medication underuse among insured adults with diabetes: the Translating Research Into Action for Diabetes Study.Diabetes Care.2008;31(2):261–266.
- ,,, et al.Cost‐related medication nonadherence among elderly and disabled Medicare beneficiaries: a national survey 1 year before the Medicare drug benefit.Arch Intern Med.2006;166(17):1829–1835.
- ,,, et al.Prescription drug coverage and seniors: findings from a 2003 national survey.Health Aff (Millwood). Jan‐Jun 2005;Suppl Web Exclusives: W5‐152‐W155‐166.
- ,,.Cost‐related medication underuse among chronically ill adults: the treatments people forgo, how often, and who is at risk.Am J Public Health.2004;94(10):1782–1787.
- ,,.Problems paying out‐of‐pocket medication costs among older adults with diabetes.Diabetes Care.2004;27(2):384–391.
- ,,.Race/ethnicity and nonadherence to prescription medications among seniors: results of a national study.J Gen Intern Med.2007;22(11):1572–1578.
- ,,,,,.Long‐term persistence in use of statin therapy in elderly patients.JAMA.2002;288(4):455–461.
- ,,, et al.Predictors of adherence with antihypertensive and lipid‐lowering therapy.Arch Intern Med.2005;165(10):1147–1152.
- ,,,.Racial disparities in the quality of medication use in older adults: baseline findings from a longitudinal study.J Gen Intern Med.2010;25(3)228–234.
- ,,,,,.Effects of increased patient cost sharing on socioeconomic disparities in health care.J Gen Intern Med.2008;23(8):1131–1136.
- .Relationship between high cost sharing and adverse outcomes: a truism that's tough to prove.Am J Manag Care.2010;16(4):287–289.
- ,,,,,.The health effects of restricting prescription medication use because of cost.Med Care.2004;42(7):626–634.
- ,.Smoking cessation initiated during hospital stay for patients with coronary artery disease: a randomized controlled trial.Can Med Assoc J.2009;180(13):1297–1303.
- .Copayment levels and medication adherence: less is more.Circulation.2009;119(3):365–367.
- ,,,,.Cost‐effectiveness of providing full drug coverage to increase medication adherence in post‐myocardial infarction Medicare beneficiaries.Circulation.2008;117(10):1261–1268.
- ,,.Cost‐related medication underuse: do patients with chronic illnesses tell their doctors?Arch Intern Med.2004;164(16):1749–1755.
- ,,.Patient‐physician communication about out‐of‐pocket costs.JAMA.2003;290(7):953–958.
- ,,,,.Patients' perceptions of generic medications.Health Aff (Millwood).2009;28(2):546–556.
- ,,.Physician strategies to reduce patients' out‐of‐pocket prescription costs.Arch Intern Med.2005;165(6):633–636.
- ,,, et al.Physicians' perceived knowledge of and responsibility for managing patients' out‐of‐pocket costs for prescription drugs.Ann Pharmacother.2006;40(9):1534–1540.
- ,,, et al.The effect of pharmacy benefit design on patient‐physician communication about costs.J Gen Intern Med.2006;21(4):334–339.
- ,,,.Discharge education improves clinical outcomes in patients with chronic heart failure.Circulation.2005;111(2):179–185.
The affordability of prescription medications continues to be one of the most pressing public health issues in the United States. Many patients reduce their prescribed doses to make medications last longer or do not fill prescriptions because of cost.1 Cost‐related medication underuse affects patients with and without drug insurance coverage,2 and is likely to become even more problematic as employers scale back on drug benefits3 and drug prices continue to increase.4 The landmark Patient Protection and Affordability Act passed in March 2010 does little to address this issue.5
Existing estimates of cost‐related medication underuse come largely from surveys of ambulatory patients. For example, using data from the Medicare Current Beneficiary Survey, Maden et al. estimated that 11% to 15% of patients reduced medication use in the past year because of cost.6 Tseng and colleagues found very similar rates of cost‐related underuse in managed care beneficiaries with diabetes.7
Hospitalized patients, who have a high burden of disease and tend to use more medications than their ambulatory counterparts, may be particularly vulnerable to cost‐related underuse but, thus far, have been subject to little investigation. New medications, which are frequently prescribed at the time of discharge, may exacerbate these issues further and contribute to preventable readmissions. Accordingly, we surveyed a cohort of medical inpatients at a large academic medical center to estimate the prevalence and predictors of cost‐related medication underuse for hospitalized managed care patients, and to identify strategies that patients perceive as helpful to make medications more affordable.
METHODS
Study Sample
We identified consecutive patients newly admitted to the general medicine, cardiology, or oncology services at Brigham and Women's Hospital from November 2008 to December 2009. For our survey, we included only those patients who received medical benefits through 1 of 3 large insurers with whom our hospital has pay‐for‐performance contracts. Annually, there are approximately 4000 patients covered by these insurers admitted to the 3 clinical services we evaluated, We focused on patients who had a primary care physician at one of the hospital's outpatient practices because of the existence of an automated infrastructure to identify these managed care beneficiaries of these insurers who are newly hospitalized, and because patients covered by commercial insurance plans likely represent a conservative lower‐bound of cost‐related medication underuse among hospitalized patients.
Patients were surveyed on the first non‐holiday weekday after admission. We excluded patients who had been discharged prior to the daily admission list being generated, or who, on a previous admission, had completed our survey or declined to be surveyed. We also excluded several patients who were not beneficiaries of the target insurers and were erroneously included on the managed care admission roster.
Potentially eligible patients were approached on the hospital ward by 1 of 3 study care coordinators (2 nurses and 1 pharmacist) and were asked if they were willing to participate in a research project about medication use that involved a short verbally delivered in‐person (inpatient) survey, a brief postdischarge telephone call, and a review of their electronic health record. The Institutional Review Board of Brigham and Women's Hospital approved this study.
Inpatient Survey
Our survey instrument was developed iteratively and pilot‐tested to improve face validity. Questions about cost‐related underuse were based on validated measures.8, 9 Specifically, we asked whether in the past year patients had: (1) not filled a prescription because it was too expensive, (2) skipped doses to make medicines last longer, (3) took less medicine than prescribed to make the medicine last longer, or (4) split pills to make the medication last longer.
Questions about strategies to improve medication affordability assessed whether patients thought it would be helpful to: (1) discuss medication affordability with healthcare workers (inpatient doctors, outpatient doctors, nurses, pharmacists, or social workers); (2) have their medications reviewed by a nurse or pharmacist; (3) receive information about lower cost but equally effective medication options, or about programs that provide medications at reduced costs; and/or (4) have their copayments/coinsurance lowered. Possible responses to all of these questions were binary, ie, yes or no.
In addition, patients were asked about the nature of their drug insurance coverage, the prescription medications that they currently use, whether they know their copayment levels (for generic and brand‐name medications), and, if so, what these amounts were, their annual household income, and their self‐identified race. Information on patient age, gender, and the primary reason for hospitalization was obtained from the electronic health record. This source was also used to verify the accuracy of the self‐reported preadmission medication list. When there were discrepancies between preadmission medications reported by patients and those recorded in their chart, the later was used because our hospital reconciles and records all medications at the time of hospital admission for all patients.
Postdischarge Survey
Within 3 days of discharge, patients were contacted by telephone and asked about new medications they were prescribed on discharge, if any. The discharge summary was used to verify the accuracy of the information provided by patients. The interviewers clarified any apparent discrepancies between the 2 sources of information with the patient. Patients who had been prescribed a new medication were asked whether or not they had filled their prescription. For patients who had, we asked whether: (1) they knew how much they would have to pay prior to going to the pharmacy, (2) they had discussed less expensive options with their pharmacist, and (3) they had discussed medication costs with their inpatient or outpatient physicians.
Data Analysis
We used descriptive statistics to summarize the characteristics of our respondents and our overall survey results. We generated univariate and multivariable logistic regression models to identify whether prehospitalization cost‐related medication underuse was influenced by patient age, gender, income, race, and the number of medications patients used on a regular basis. For the purpose of these analyses, we classified patients as reporting cost‐related underuse if they responded yes to any of the 4 strategies described above (ie, not filling medications, skipping doses, taking less medication, or splitting pills to make medicines last longer). Patients whose incomes were above the median level in our cohort were categorized as being of high‐income. Our multivariable model had a c‐statistic of 0.75, suggesting good discriminative ability.
RESULTS
During the study period, 483 potentially‐eligible patients were admitted to the general medicine, cardiology, and oncology services. We excluded 167 because they had been discharged prior to being identified, had been surveyed or already declined participation on a prior admission, or were not managed care enrollees (see Appendix A). Of the remaining 316 subjects, 130 participated in the inpatient survey (response rate = 41%); 93 (75%) of these patients were reached by telephone after hospital discharge and completed the postdischarge survey. The baseline characteristics of our respondents are presented in Table 1. Patients had a mean age of 52 years, were 50% male and two‐thirds of white race, represented a range of household incomes, and almost all had employer‐sponsored prescription coverage. Prior to admission, patients took an average of 5 prescription medications and paid an average copayment of $10.80 and $21.60 for each generic and brand‐name prescription, respectively.
| Characteristic | N = 130 |
|---|---|
| |
| Age, mean years (SD) | 52 (11.2) |
| Male, % | 65 (50.0) |
| Race/ethnicity,* n (%) | |
| Caucasian/white | 84 (67.2) |
| Black/African American | 20 (16.0) |
| Latino/Hispanic | 13 (10.4) |
| Asian | 3 (2.4) |
| American Indian or Alaska Native | 1 (0.8) |
| Other | 4 (3.2) |
| Annual household income,* n (%) | |
| <$30,000 | 15 (12.8) |
| $30,000‐$75,000 | 49 (41.9) |
| >$75,000 | 53 (45.3) |
| Insurance coverage for outpatient prescription drugs,* n (%) | |
| Employer or spouse's employer | 123 (96.0) |
| Independent | 5 (3.9) |
| Medication copayments,* mean $ (SD) | |
| Brand‐name medications | 21.6 (14.2) |
| Generic medications | 10.8 (6.0) |
| No. of medications prior to admission, mean (SD) | 5.5 (4.3) |
| Category of discharge diagnosis, n (%) | |
| Cardiovascular | 40 (30.8) |
| Gastrointestinal | 23 (17.7) |
| Pulmonary | 23 (17.7) |
| Infectious | 13 (10.0) |
| Oncology | 5 (3.8) |
| Renal | 6 (4.6) |
| Psychiatric | 3 (2.3) |
| Hematologic | 4 (3.1) |
| Neurologic | 5 (3.8) |
| Musculoskeletal | 5 (3.8) |
| Respiratory | 2 (1.5) |
| Endocrine | 1 (0.8) |
Cost‐Related Medication Underuse
Thirty (23%) of the survey respondents reported at least 1 cost‐related medication underuse strategy in the year prior to their hospital admission (Figure 1), most commonly not filling a prescription at all because of cost (n = 26; 20%). Rates of cost‐related underuse were highest for patients of black race, low income, and women (Figure 2).


In unadjusted analyses, black respondents had 4.60 (95% confidence interval [CI], 1.63 to 13.0) times the odds of reporting cost‐related underuse than non‐Hispanic white respondents (Table 2). The association of black race and cost‐related underuse appears to be confounded, in part, by income (adjusted odds ratio for black race was 4.16; 95% CI, 1.34 to 12.86) and the number of medications patients used on a regular basis (adjusted odds ratio for black race was 4.14; 95% CI, 1.44 to 11.96). After controlling for these variables, as well as age and gender, the relationship between race and cost‐related underuse remained statistically significant (adjusted odds ratio 3.39; 95% CI, 1.05 to 11.02) (Table 2).
| Predictor | Unadjusted Odds Ratio (95% CI) | Adjusted Odds Ratio (95% CI) |
|---|---|---|
| ||
| Age (per additional year) | 0.98 (0.941.02) | 0.97 (0.931.01) |
| Male (vs female) | 0.84 (0.371.90) | 1.03 (0.432.48) |
| Race (vs white race) | ||
| Black | 4.60 (1.6313.0) | 3.39 (1.0511.02) |
| Other | 1.10 (0.363.37) | 0.77 (0.202.99) |
| No. of medications (per additional medication) | 1.10 (1.001.20) | 1.10 (1.001.22) |
| High income (vs low income) | 0.62 (0.271.42) | 0.71 (0.242.07) |
Strategies to Help Make Medications More Affordable
Virtually all respondents (n = 123; 95%) endorsed at least one of the proposed strategies to make medications more affordable (Figure 3). A majority felt that lowering cost sharing (94%), or receiving information about lower‐cost medication options (83%) or programs to subsidize medication costs (83%) would be helpful. Approximately 70% of patients stated that speaking to their outpatient physicians might be helpful, although only 14% reported actually speaking with their primary care provider about medication costs in the past year. Results were mixed for other strategies, including speaking with their inpatient physicians.

Postdischarge Medication Use
Seventy‐six (82%) respondents to the outpatient survey were prescribed a new medication at the time of hospital discharge, and virtually all (95%) had filled prescriptions for these medications by the time of the follow‐up survey. Patients paid an average of $27.63 (standard deviation $39.24) in out‐of‐pocket costs for these medications. Few (16%) patients knew how much they would have to pay before they had gone to the pharmacy to fill their prescription (see Appendix B). Even fewer patients asked, or were spoken to by their pharmacist, about less expensive medication options (7%), and almost none had spoken to their inpatient (4%) or outpatient providers (2%) about the cost of their newly prescribed drugs.
DISCUSSION
Almost a quarter of the medical inpatients we surveyed had not filled a medication because of cost, or had skipped doses, reduced dosages, or split pills to make their medicines last longer in the prior year. This amount is larger than that found in many prior studies, conducted in outpatient settings, in which 11% to 19% of patients report cost‐related underuse.68, 10, 11 Our results are particularly striking considering that our study cohort consisted exclusively of patients with commercial health insurance, the vast majority of whom also had employer‐sponsored drug coverage. Cost‐related medication underuse may be even more prevalent among hospitalized patients with less generous benefits, including the uninsured and perhaps even beneficiaries of Medicare Part D.
Reductions in medication use because of cost were particularly high among black patients, whose odds of reporting cost‐related underuse were more than 3 times higher than that of patients of non‐Hispanic white race. Race‐related differences in cost‐related underuse have been observed in outpatient studies,68, 12 and may be an important contributor to racial disparities in evidence‐based medication use.1315 These differences may, in part, reflect racial variations in socioeconomic status; lower income patients, who are more likely to be from a racial or ethnic minority, are more sensitive to cost sharing than higher income individuals.16 Consistent with this, the relationship between race and cost‐related underuse in our study was smaller but still highly significant in multivariable models that adjusted for income.
Not surprisingly, the underuse of effective prescription medications is associated with adverse clinical and economic consequences.17 Heisler et al. found that patients who had restricted medications because of cost were 76% more likely to report a decline in their health status than those who had not.18 The health effects of cost‐related underuse are likely to be particularly significant for hospitalized patients, given their high burden of disease and the frequency with which they are prescribed medications at discharge to treat the condition that led to their initial hospitalization. Thus, targeting efforts to address cost‐related underuse patients who are hospitalized may be an efficient method of improving patient health and reducing preventable readmissions. This is consistent with efforts that address, in the inpatient setting, other health issues that are commonly encountered in the ambulatory arena, such as immunizations and smoking cessation.19
Our survey respondents endorsed numerous strategies as being potentially helpful. Predictably, support for lowering copayments was extremely high. While this may not be practical or even desirable for some medications, lowering copayments for highly effective medications, such as statins and antihypertensives, in the context of value‐based insurance design, is an increasingly adopted strategy that has the potential to simultaneously improve clinical outcomes and reduce overall health spending.20, 21
While the majority of patients felt that talking to their outpatient physicians or pharmacists about medication costs might be helpful, the effectiveness of this strategy is unclear. Consistent with prior results,22, 23 the vast majority of the patients we surveyed had not discussed medication costs prior to their admission or after filling newly prescribed medications. Further, although physicians could help reduce drug expenditures in a variety of ways, including the increased ordering of generic drugs,24 many physicians are uncomfortable talking to their patients about costs,25 have limited knowledge about their patients' out‐of‐pocket expenditures, feel that addressing this issue is not their responsibility,26 or do not have resources, such as electronic formulary information, that could facilitate these discussions in an efficient manner.
An alternative strategy may be to provide patients with better education about medication costs. Virtually none of the patients we surveyed knew how much they would pay for their new prescriptions before visiting the pharmacy. These findings are similar to those observed in the outpatient setting,27 and suggest an opportunity to provide patients with information about the cost of their newly and previously prescribed drugs, and to facilitate discussions between patients and inpatient providers about predischarge prescribing decisions, in the same spirit as other predischarge patient education.28 Of course, issues related to transitions of care between the hospital and community setting, and coordination between inpatient and outpatient providers, must be adequately addressed for this strategy to be effective.
Our study has several notable limitations. It had a relatively small sample size and low response rate. Respondents may have differed systematically from non‐respondents, and we were unable to compare the characteristics of both populations. Further, we studied commercially insured inpatients on internal medicine services at an academic medical center, and thus our results may not be generalizable to patients hospitalized in other settings, or with different types of insurance coverage, including the uninsured. The primary outcome of our study was to determine self‐reported cost‐related underuse. While we used validated measures,8 it is possible that patients who reported reducing their medication use in response to cost may not have actually done so. We did not collect information on education or health literacy, nor did we have access to detailed information about our respondents' pharmacy benefit design structures; these important factors may have confounded our analyses, and/or may have been mediators of our observed results, and should be evaluated further in future studies. We did not have adequate statistical power to evaluate whether patients using specific classes of medications were particularly prone to cost‐related underuse.
Despite these limitations, our study is the first, to our knowledge, to evaluate the impact of medication costs on use in a cohort of hospitalized individuals. The high levels of cost‐related underuse that we observed is concerning. Our results support calls for the further development of interventions to address high medication costs and for the consideration of novel approaches to assist patients around the time of hospital discharge.
APPENDICES
APPENDIX A. Survey response flow diagram.
APPENDIX B. Behaviors to address the cost of medications prescribed at hospital discharge.
The affordability of prescription medications continues to be one of the most pressing public health issues in the United States. Many patients reduce their prescribed doses to make medications last longer or do not fill prescriptions because of cost.1 Cost‐related medication underuse affects patients with and without drug insurance coverage,2 and is likely to become even more problematic as employers scale back on drug benefits3 and drug prices continue to increase.4 The landmark Patient Protection and Affordability Act passed in March 2010 does little to address this issue.5
Existing estimates of cost‐related medication underuse come largely from surveys of ambulatory patients. For example, using data from the Medicare Current Beneficiary Survey, Maden et al. estimated that 11% to 15% of patients reduced medication use in the past year because of cost.6 Tseng and colleagues found very similar rates of cost‐related underuse in managed care beneficiaries with diabetes.7
Hospitalized patients, who have a high burden of disease and tend to use more medications than their ambulatory counterparts, may be particularly vulnerable to cost‐related underuse but, thus far, have been subject to little investigation. New medications, which are frequently prescribed at the time of discharge, may exacerbate these issues further and contribute to preventable readmissions. Accordingly, we surveyed a cohort of medical inpatients at a large academic medical center to estimate the prevalence and predictors of cost‐related medication underuse for hospitalized managed care patients, and to identify strategies that patients perceive as helpful to make medications more affordable.
METHODS
Study Sample
We identified consecutive patients newly admitted to the general medicine, cardiology, or oncology services at Brigham and Women's Hospital from November 2008 to December 2009. For our survey, we included only those patients who received medical benefits through 1 of 3 large insurers with whom our hospital has pay‐for‐performance contracts. Annually, there are approximately 4000 patients covered by these insurers admitted to the 3 clinical services we evaluated, We focused on patients who had a primary care physician at one of the hospital's outpatient practices because of the existence of an automated infrastructure to identify these managed care beneficiaries of these insurers who are newly hospitalized, and because patients covered by commercial insurance plans likely represent a conservative lower‐bound of cost‐related medication underuse among hospitalized patients.
Patients were surveyed on the first non‐holiday weekday after admission. We excluded patients who had been discharged prior to the daily admission list being generated, or who, on a previous admission, had completed our survey or declined to be surveyed. We also excluded several patients who were not beneficiaries of the target insurers and were erroneously included on the managed care admission roster.
Potentially eligible patients were approached on the hospital ward by 1 of 3 study care coordinators (2 nurses and 1 pharmacist) and were asked if they were willing to participate in a research project about medication use that involved a short verbally delivered in‐person (inpatient) survey, a brief postdischarge telephone call, and a review of their electronic health record. The Institutional Review Board of Brigham and Women's Hospital approved this study.
Inpatient Survey
Our survey instrument was developed iteratively and pilot‐tested to improve face validity. Questions about cost‐related underuse were based on validated measures.8, 9 Specifically, we asked whether in the past year patients had: (1) not filled a prescription because it was too expensive, (2) skipped doses to make medicines last longer, (3) took less medicine than prescribed to make the medicine last longer, or (4) split pills to make the medication last longer.
Questions about strategies to improve medication affordability assessed whether patients thought it would be helpful to: (1) discuss medication affordability with healthcare workers (inpatient doctors, outpatient doctors, nurses, pharmacists, or social workers); (2) have their medications reviewed by a nurse or pharmacist; (3) receive information about lower cost but equally effective medication options, or about programs that provide medications at reduced costs; and/or (4) have their copayments/coinsurance lowered. Possible responses to all of these questions were binary, ie, yes or no.
In addition, patients were asked about the nature of their drug insurance coverage, the prescription medications that they currently use, whether they know their copayment levels (for generic and brand‐name medications), and, if so, what these amounts were, their annual household income, and their self‐identified race. Information on patient age, gender, and the primary reason for hospitalization was obtained from the electronic health record. This source was also used to verify the accuracy of the self‐reported preadmission medication list. When there were discrepancies between preadmission medications reported by patients and those recorded in their chart, the later was used because our hospital reconciles and records all medications at the time of hospital admission for all patients.
Postdischarge Survey
Within 3 days of discharge, patients were contacted by telephone and asked about new medications they were prescribed on discharge, if any. The discharge summary was used to verify the accuracy of the information provided by patients. The interviewers clarified any apparent discrepancies between the 2 sources of information with the patient. Patients who had been prescribed a new medication were asked whether or not they had filled their prescription. For patients who had, we asked whether: (1) they knew how much they would have to pay prior to going to the pharmacy, (2) they had discussed less expensive options with their pharmacist, and (3) they had discussed medication costs with their inpatient or outpatient physicians.
Data Analysis
We used descriptive statistics to summarize the characteristics of our respondents and our overall survey results. We generated univariate and multivariable logistic regression models to identify whether prehospitalization cost‐related medication underuse was influenced by patient age, gender, income, race, and the number of medications patients used on a regular basis. For the purpose of these analyses, we classified patients as reporting cost‐related underuse if they responded yes to any of the 4 strategies described above (ie, not filling medications, skipping doses, taking less medication, or splitting pills to make medicines last longer). Patients whose incomes were above the median level in our cohort were categorized as being of high‐income. Our multivariable model had a c‐statistic of 0.75, suggesting good discriminative ability.
RESULTS
During the study period, 483 potentially‐eligible patients were admitted to the general medicine, cardiology, and oncology services. We excluded 167 because they had been discharged prior to being identified, had been surveyed or already declined participation on a prior admission, or were not managed care enrollees (see Appendix A). Of the remaining 316 subjects, 130 participated in the inpatient survey (response rate = 41%); 93 (75%) of these patients were reached by telephone after hospital discharge and completed the postdischarge survey. The baseline characteristics of our respondents are presented in Table 1. Patients had a mean age of 52 years, were 50% male and two‐thirds of white race, represented a range of household incomes, and almost all had employer‐sponsored prescription coverage. Prior to admission, patients took an average of 5 prescription medications and paid an average copayment of $10.80 and $21.60 for each generic and brand‐name prescription, respectively.
| Characteristic | N = 130 |
|---|---|
| |
| Age, mean years (SD) | 52 (11.2) |
| Male, % | 65 (50.0) |
| Race/ethnicity,* n (%) | |
| Caucasian/white | 84 (67.2) |
| Black/African American | 20 (16.0) |
| Latino/Hispanic | 13 (10.4) |
| Asian | 3 (2.4) |
| American Indian or Alaska Native | 1 (0.8) |
| Other | 4 (3.2) |
| Annual household income,* n (%) | |
| <$30,000 | 15 (12.8) |
| $30,000‐$75,000 | 49 (41.9) |
| >$75,000 | 53 (45.3) |
| Insurance coverage for outpatient prescription drugs,* n (%) | |
| Employer or spouse's employer | 123 (96.0) |
| Independent | 5 (3.9) |
| Medication copayments,* mean $ (SD) | |
| Brand‐name medications | 21.6 (14.2) |
| Generic medications | 10.8 (6.0) |
| No. of medications prior to admission, mean (SD) | 5.5 (4.3) |
| Category of discharge diagnosis, n (%) | |
| Cardiovascular | 40 (30.8) |
| Gastrointestinal | 23 (17.7) |
| Pulmonary | 23 (17.7) |
| Infectious | 13 (10.0) |
| Oncology | 5 (3.8) |
| Renal | 6 (4.6) |
| Psychiatric | 3 (2.3) |
| Hematologic | 4 (3.1) |
| Neurologic | 5 (3.8) |
| Musculoskeletal | 5 (3.8) |
| Respiratory | 2 (1.5) |
| Endocrine | 1 (0.8) |
Cost‐Related Medication Underuse
Thirty (23%) of the survey respondents reported at least 1 cost‐related medication underuse strategy in the year prior to their hospital admission (Figure 1), most commonly not filling a prescription at all because of cost (n = 26; 20%). Rates of cost‐related underuse were highest for patients of black race, low income, and women (Figure 2).


In unadjusted analyses, black respondents had 4.60 (95% confidence interval [CI], 1.63 to 13.0) times the odds of reporting cost‐related underuse than non‐Hispanic white respondents (Table 2). The association of black race and cost‐related underuse appears to be confounded, in part, by income (adjusted odds ratio for black race was 4.16; 95% CI, 1.34 to 12.86) and the number of medications patients used on a regular basis (adjusted odds ratio for black race was 4.14; 95% CI, 1.44 to 11.96). After controlling for these variables, as well as age and gender, the relationship between race and cost‐related underuse remained statistically significant (adjusted odds ratio 3.39; 95% CI, 1.05 to 11.02) (Table 2).
| Predictor | Unadjusted Odds Ratio (95% CI) | Adjusted Odds Ratio (95% CI) |
|---|---|---|
| ||
| Age (per additional year) | 0.98 (0.941.02) | 0.97 (0.931.01) |
| Male (vs female) | 0.84 (0.371.90) | 1.03 (0.432.48) |
| Race (vs white race) | ||
| Black | 4.60 (1.6313.0) | 3.39 (1.0511.02) |
| Other | 1.10 (0.363.37) | 0.77 (0.202.99) |
| No. of medications (per additional medication) | 1.10 (1.001.20) | 1.10 (1.001.22) |
| High income (vs low income) | 0.62 (0.271.42) | 0.71 (0.242.07) |
Strategies to Help Make Medications More Affordable
Virtually all respondents (n = 123; 95%) endorsed at least one of the proposed strategies to make medications more affordable (Figure 3). A majority felt that lowering cost sharing (94%), or receiving information about lower‐cost medication options (83%) or programs to subsidize medication costs (83%) would be helpful. Approximately 70% of patients stated that speaking to their outpatient physicians might be helpful, although only 14% reported actually speaking with their primary care provider about medication costs in the past year. Results were mixed for other strategies, including speaking with their inpatient physicians.

Postdischarge Medication Use
Seventy‐six (82%) respondents to the outpatient survey were prescribed a new medication at the time of hospital discharge, and virtually all (95%) had filled prescriptions for these medications by the time of the follow‐up survey. Patients paid an average of $27.63 (standard deviation $39.24) in out‐of‐pocket costs for these medications. Few (16%) patients knew how much they would have to pay before they had gone to the pharmacy to fill their prescription (see Appendix B). Even fewer patients asked, or were spoken to by their pharmacist, about less expensive medication options (7%), and almost none had spoken to their inpatient (4%) or outpatient providers (2%) about the cost of their newly prescribed drugs.
DISCUSSION
Almost a quarter of the medical inpatients we surveyed had not filled a medication because of cost, or had skipped doses, reduced dosages, or split pills to make their medicines last longer in the prior year. This amount is larger than that found in many prior studies, conducted in outpatient settings, in which 11% to 19% of patients report cost‐related underuse.68, 10, 11 Our results are particularly striking considering that our study cohort consisted exclusively of patients with commercial health insurance, the vast majority of whom also had employer‐sponsored drug coverage. Cost‐related medication underuse may be even more prevalent among hospitalized patients with less generous benefits, including the uninsured and perhaps even beneficiaries of Medicare Part D.
Reductions in medication use because of cost were particularly high among black patients, whose odds of reporting cost‐related underuse were more than 3 times higher than that of patients of non‐Hispanic white race. Race‐related differences in cost‐related underuse have been observed in outpatient studies,68, 12 and may be an important contributor to racial disparities in evidence‐based medication use.1315 These differences may, in part, reflect racial variations in socioeconomic status; lower income patients, who are more likely to be from a racial or ethnic minority, are more sensitive to cost sharing than higher income individuals.16 Consistent with this, the relationship between race and cost‐related underuse in our study was smaller but still highly significant in multivariable models that adjusted for income.
Not surprisingly, the underuse of effective prescription medications is associated with adverse clinical and economic consequences.17 Heisler et al. found that patients who had restricted medications because of cost were 76% more likely to report a decline in their health status than those who had not.18 The health effects of cost‐related underuse are likely to be particularly significant for hospitalized patients, given their high burden of disease and the frequency with which they are prescribed medications at discharge to treat the condition that led to their initial hospitalization. Thus, targeting efforts to address cost‐related underuse patients who are hospitalized may be an efficient method of improving patient health and reducing preventable readmissions. This is consistent with efforts that address, in the inpatient setting, other health issues that are commonly encountered in the ambulatory arena, such as immunizations and smoking cessation.19
Our survey respondents endorsed numerous strategies as being potentially helpful. Predictably, support for lowering copayments was extremely high. While this may not be practical or even desirable for some medications, lowering copayments for highly effective medications, such as statins and antihypertensives, in the context of value‐based insurance design, is an increasingly adopted strategy that has the potential to simultaneously improve clinical outcomes and reduce overall health spending.20, 21
While the majority of patients felt that talking to their outpatient physicians or pharmacists about medication costs might be helpful, the effectiveness of this strategy is unclear. Consistent with prior results,22, 23 the vast majority of the patients we surveyed had not discussed medication costs prior to their admission or after filling newly prescribed medications. Further, although physicians could help reduce drug expenditures in a variety of ways, including the increased ordering of generic drugs,24 many physicians are uncomfortable talking to their patients about costs,25 have limited knowledge about their patients' out‐of‐pocket expenditures, feel that addressing this issue is not their responsibility,26 or do not have resources, such as electronic formulary information, that could facilitate these discussions in an efficient manner.
An alternative strategy may be to provide patients with better education about medication costs. Virtually none of the patients we surveyed knew how much they would pay for their new prescriptions before visiting the pharmacy. These findings are similar to those observed in the outpatient setting,27 and suggest an opportunity to provide patients with information about the cost of their newly and previously prescribed drugs, and to facilitate discussions between patients and inpatient providers about predischarge prescribing decisions, in the same spirit as other predischarge patient education.28 Of course, issues related to transitions of care between the hospital and community setting, and coordination between inpatient and outpatient providers, must be adequately addressed for this strategy to be effective.
Our study has several notable limitations. It had a relatively small sample size and low response rate. Respondents may have differed systematically from non‐respondents, and we were unable to compare the characteristics of both populations. Further, we studied commercially insured inpatients on internal medicine services at an academic medical center, and thus our results may not be generalizable to patients hospitalized in other settings, or with different types of insurance coverage, including the uninsured. The primary outcome of our study was to determine self‐reported cost‐related underuse. While we used validated measures,8 it is possible that patients who reported reducing their medication use in response to cost may not have actually done so. We did not collect information on education or health literacy, nor did we have access to detailed information about our respondents' pharmacy benefit design structures; these important factors may have confounded our analyses, and/or may have been mediators of our observed results, and should be evaluated further in future studies. We did not have adequate statistical power to evaluate whether patients using specific classes of medications were particularly prone to cost‐related underuse.
Despite these limitations, our study is the first, to our knowledge, to evaluate the impact of medication costs on use in a cohort of hospitalized individuals. The high levels of cost‐related underuse that we observed is concerning. Our results support calls for the further development of interventions to address high medication costs and for the consideration of novel approaches to assist patients around the time of hospital discharge.
APPENDICES
APPENDIX A. Survey response flow diagram.
APPENDIX B. Behaviors to address the cost of medications prescribed at hospital discharge.
- USA Today/Kaiser Family Foundation/Harvard School of Public Health.The Public on Prescription Drugs and Pharmaceutical Companies.2008. Available at: http://www.kff.org/kaiserpolls/pomr030408pkg.cfm. Accessed September 5, 2008.
- ,,, et al.Pharmacy benefits and the use of drugs by the chronically ill.JAMA.2004;291(19):2344–2350.
- Kaiser Family Foundation and Health Research and Educational Trust.Employer Health Benefits Annual Survey,2009.year="2009"2009. Available at: http://ehbs.kff.org/pdf/2009/7936.pdf. Accessed May 5,year="2010"2010.
- Kaiser Family Foundation.Prescription Drug Trends.2007. Available at: http://www.kff.org/rxdrugs/upload/3057_06.pdf. Accessed December 5,year="2007"2007.
- The Patient Protection and Affordable Care Act, H.R. 3590, Section 2713 (c).Washington, DC:111 Congress;2010.
- ,,, et al.Cost‐related medication nonadherence and spending on basic needs following implementation of Medicare Part D.JAMA.2008;299(16):1922–1928.
- ,,, et al.Race/ethnicity and economic differences in cost‐related medication underuse among insured adults with diabetes: the Translating Research Into Action for Diabetes Study.Diabetes Care.2008;31(2):261–266.
- ,,, et al.Cost‐related medication nonadherence among elderly and disabled Medicare beneficiaries: a national survey 1 year before the Medicare drug benefit.Arch Intern Med.2006;166(17):1829–1835.
- ,,, et al.Prescription drug coverage and seniors: findings from a 2003 national survey.Health Aff (Millwood). Jan‐Jun 2005;Suppl Web Exclusives: W5‐152‐W155‐166.
- ,,.Cost‐related medication underuse among chronically ill adults: the treatments people forgo, how often, and who is at risk.Am J Public Health.2004;94(10):1782–1787.
- ,,.Problems paying out‐of‐pocket medication costs among older adults with diabetes.Diabetes Care.2004;27(2):384–391.
- ,,.Race/ethnicity and nonadherence to prescription medications among seniors: results of a national study.J Gen Intern Med.2007;22(11):1572–1578.
- ,,,,,.Long‐term persistence in use of statin therapy in elderly patients.JAMA.2002;288(4):455–461.
- ,,, et al.Predictors of adherence with antihypertensive and lipid‐lowering therapy.Arch Intern Med.2005;165(10):1147–1152.
- ,,,.Racial disparities in the quality of medication use in older adults: baseline findings from a longitudinal study.J Gen Intern Med.2010;25(3)228–234.
- ,,,,,.Effects of increased patient cost sharing on socioeconomic disparities in health care.J Gen Intern Med.2008;23(8):1131–1136.
- .Relationship between high cost sharing and adverse outcomes: a truism that's tough to prove.Am J Manag Care.2010;16(4):287–289.
- ,,,,,.The health effects of restricting prescription medication use because of cost.Med Care.2004;42(7):626–634.
- ,.Smoking cessation initiated during hospital stay for patients with coronary artery disease: a randomized controlled trial.Can Med Assoc J.2009;180(13):1297–1303.
- .Copayment levels and medication adherence: less is more.Circulation.2009;119(3):365–367.
- ,,,,.Cost‐effectiveness of providing full drug coverage to increase medication adherence in post‐myocardial infarction Medicare beneficiaries.Circulation.2008;117(10):1261–1268.
- ,,.Cost‐related medication underuse: do patients with chronic illnesses tell their doctors?Arch Intern Med.2004;164(16):1749–1755.
- ,,.Patient‐physician communication about out‐of‐pocket costs.JAMA.2003;290(7):953–958.
- ,,,,.Patients' perceptions of generic medications.Health Aff (Millwood).2009;28(2):546–556.
- ,,.Physician strategies to reduce patients' out‐of‐pocket prescription costs.Arch Intern Med.2005;165(6):633–636.
- ,,, et al.Physicians' perceived knowledge of and responsibility for managing patients' out‐of‐pocket costs for prescription drugs.Ann Pharmacother.2006;40(9):1534–1540.
- ,,, et al.The effect of pharmacy benefit design on patient‐physician communication about costs.J Gen Intern Med.2006;21(4):334–339.
- ,,,.Discharge education improves clinical outcomes in patients with chronic heart failure.Circulation.2005;111(2):179–185.
- USA Today/Kaiser Family Foundation/Harvard School of Public Health.The Public on Prescription Drugs and Pharmaceutical Companies.2008. Available at: http://www.kff.org/kaiserpolls/pomr030408pkg.cfm. Accessed September 5, 2008.
- ,,, et al.Pharmacy benefits and the use of drugs by the chronically ill.JAMA.2004;291(19):2344–2350.
- Kaiser Family Foundation and Health Research and Educational Trust.Employer Health Benefits Annual Survey,2009.year="2009"2009. Available at: http://ehbs.kff.org/pdf/2009/7936.pdf. Accessed May 5,year="2010"2010.
- Kaiser Family Foundation.Prescription Drug Trends.2007. Available at: http://www.kff.org/rxdrugs/upload/3057_06.pdf. Accessed December 5,year="2007"2007.
- The Patient Protection and Affordable Care Act, H.R. 3590, Section 2713 (c).Washington, DC:111 Congress;2010.
- ,,, et al.Cost‐related medication nonadherence and spending on basic needs following implementation of Medicare Part D.JAMA.2008;299(16):1922–1928.
- ,,, et al.Race/ethnicity and economic differences in cost‐related medication underuse among insured adults with diabetes: the Translating Research Into Action for Diabetes Study.Diabetes Care.2008;31(2):261–266.
- ,,, et al.Cost‐related medication nonadherence among elderly and disabled Medicare beneficiaries: a national survey 1 year before the Medicare drug benefit.Arch Intern Med.2006;166(17):1829–1835.
- ,,, et al.Prescription drug coverage and seniors: findings from a 2003 national survey.Health Aff (Millwood). Jan‐Jun 2005;Suppl Web Exclusives: W5‐152‐W155‐166.
- ,,.Cost‐related medication underuse among chronically ill adults: the treatments people forgo, how often, and who is at risk.Am J Public Health.2004;94(10):1782–1787.
- ,,.Problems paying out‐of‐pocket medication costs among older adults with diabetes.Diabetes Care.2004;27(2):384–391.
- ,,.Race/ethnicity and nonadherence to prescription medications among seniors: results of a national study.J Gen Intern Med.2007;22(11):1572–1578.
- ,,,,,.Long‐term persistence in use of statin therapy in elderly patients.JAMA.2002;288(4):455–461.
- ,,, et al.Predictors of adherence with antihypertensive and lipid‐lowering therapy.Arch Intern Med.2005;165(10):1147–1152.
- ,,,.Racial disparities in the quality of medication use in older adults: baseline findings from a longitudinal study.J Gen Intern Med.2010;25(3)228–234.
- ,,,,,.Effects of increased patient cost sharing on socioeconomic disparities in health care.J Gen Intern Med.2008;23(8):1131–1136.
- .Relationship between high cost sharing and adverse outcomes: a truism that's tough to prove.Am J Manag Care.2010;16(4):287–289.
- ,,,,,.The health effects of restricting prescription medication use because of cost.Med Care.2004;42(7):626–634.
- ,.Smoking cessation initiated during hospital stay for patients with coronary artery disease: a randomized controlled trial.Can Med Assoc J.2009;180(13):1297–1303.
- .Copayment levels and medication adherence: less is more.Circulation.2009;119(3):365–367.
- ,,,,.Cost‐effectiveness of providing full drug coverage to increase medication adherence in post‐myocardial infarction Medicare beneficiaries.Circulation.2008;117(10):1261–1268.
- ,,.Cost‐related medication underuse: do patients with chronic illnesses tell their doctors?Arch Intern Med.2004;164(16):1749–1755.
- ,,.Patient‐physician communication about out‐of‐pocket costs.JAMA.2003;290(7):953–958.
- ,,,,.Patients' perceptions of generic medications.Health Aff (Millwood).2009;28(2):546–556.
- ,,.Physician strategies to reduce patients' out‐of‐pocket prescription costs.Arch Intern Med.2005;165(6):633–636.
- ,,, et al.Physicians' perceived knowledge of and responsibility for managing patients' out‐of‐pocket costs for prescription drugs.Ann Pharmacother.2006;40(9):1534–1540.
- ,,, et al.The effect of pharmacy benefit design on patient‐physician communication about costs.J Gen Intern Med.2006;21(4):334–339.
- ,,,.Discharge education improves clinical outcomes in patients with chronic heart failure.Circulation.2005;111(2):179–185.
Copyright © 2011 Society of Hospital Medicine