User login
High-Value, Cost-Conscious Evaluation for PCOS: Which Tests Should Be Routinely Ordered in Acne Patients?
The adult female patient presenting with severe acne vulgaris may raise special diagnostic concerns, including consideration of an underlying hormonal disorder. Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in women of reproductive age with an estimated prevalence as high as 12%.1 Many women with undiagnosed PCOS may be referred to dermatologists for evaluation of its cutaneous manifestations of hyperandrogenism including acne, hirsutism, and androgenic alopecia.2 Given the prevalence of PCOS and its long-term health implications, dermatologists can play an important role in the initial evaluation of these patients. Acne and androgenic alopecia, however, are quite common, and in the absence of red flags such as menstrual irregularities, virilization, visual field deficits, or signs of Cushing syndrome,3 clinicians must decide when to pursue limited versus comprehensive evaluation.
Despite being common in patients with PCOS, a recent study suggests that acne is an unreliable marker of biochemical hyperandrogenism, and specific features of acne (ie, lesion counts, lesional types, distribution) cannot reliably discriminate women who meet PCOS diagnostic criteria from those who do not.4 Similarly, the study found that androgenic alopecia was not associated with biochemical hyperandrogenism and was no more common in women with PCOS than women of similar age in a high-risk population. Unlike acne and androgenic alopecia, however, the study identified hirsutism, especially truncal hirsutism, as a reliable indicator of hyperandrogenemia and PCOS. Hirsutism also is associated with metabolic sequelae of PCOS. These findings suggest that hirsutism, but not acne or androgenic alopecia, in a female of reproductive age warrants a workup for PCOS.4 This report is consistent with a recommendation from the Androgen Excess and Polycystic Ovary Syndrome Society (AE-PCOS) to pursue a diagnostic evaluation in any woman presenting with hirsutism.5 Acanthosis nigricans also was found to be a reliable indicator of hyperandrogenemia, PCOS, and associated metabolic derangement. Thus, although recent evidence indicates that acne as an isolated cutaneous finding does not warrant further diagnostic evaluation, acne in the setting of hirsutism, acanthosis nigricans, menstrual irregularities, or additional specific signs of endocrine dysregulation should prompt focused workup.4
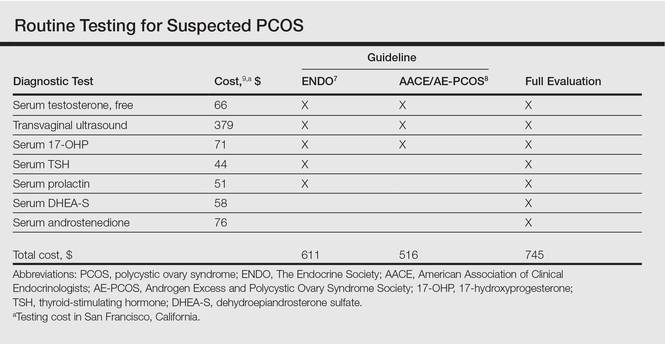
Multiple clinical practice guidelines for the evaluation of hirsutism and PCOS based on literature review and expert opinion have been proposed5-8; however, these guidelines vary in recommendations for routine diagnostic steps to exclude mimickers of PCOS such as prolactinoma/pituitary adenoma and congenital adrenal hyperplasia (CAH)(Table). In 2009, an AE-PCOS task force suggested that routine testing of thyroid function and serum prolactin in the absence of additional clinical signs may not be necessary based on the low prevalence of thyroid disorders and hyperprolactinemia in patients presenting with hyperandrogenism.6 In 2013, the Endocrine Society’s (ENDO) clinical guideline, however, recommended routine measurement of serum thyroid-stimulating hormone (TSH) to exclude thyroid disease and serum prolactin to exclude hyperprolactinemia in all women before making a diagnosis of PCOS.7 In 2015, the AE-PCOS collaborated with the American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology to publish an updated guideline for best practices, which was consistent with the prior AE-PCOS recommendation in 2009 for routine screening including to test 17-hydroxyprogesterone to exclude nonclassical CAH.8
Importantly, these recommendations for routine testing for mimickers of PCOS are based on the rare prevalence of these etiologies in multiple studies of women presenting for hyperandrogenism. One study included 873 women presenting to an academic reproductive endocrine clinic for evaluation of symptoms potentially related to androgen excess. In addition to cutaneous manifestations of hirsutism, acne, and alopecia, the study also included women presenting with oligomenorrhea/amenorrhea, ovulatory dysfunction, and even virilization.10 A second study included 950 women presenting to academic endocrine departments with hirsutism, acne, or androgenic alopecia.11 Both studies defined hirsutism as having a modified Ferriman-Gallwey score of 6 or greater. Both studies also only measured serum prolactin or TSH when clinically indicated (ie, patients with ovulatory dysfunction).10,11
The diagnostic yield of tests for mimickers of PCOS was exceedingly low in both studies. For example, of the patients evaluated, only 0.4% to 0.7% had thyroid dysfunction, 0% to 0.3% had hyperprolactinemia, 0.2% had androgen-secreting neoplasms, 2.1% to 4.3% had nonclassical CAH, 0.7% had CAH, and 3.8% had HAIR-AN (hyperandrogenism, insulin resistance, and acanthosis nigricans) syndrome.10,11 Because patients in both studies were only tested for hyperprolactinemia and thyroid dysfunction when clinically indicated, it is probable that routine screening without clinical indication would result in even lower yields.
Given the increasing importance of high-value, cost-conscious care,12 clinicians must consider the costs associated with testing in the face of low pretest probability. Although some studies have examined the cost-effectiveness of fertility treatments in PCOS,13,14 no studies have examined the cost-effectiveness of diagnostic strategies for PCOS. Cost-effectiveness studies are emerging to provide important guidance on high-value, cost-conscious diagnostic evaluation and monitoring15 and are much needed in dermatology.16,17
In the case of PCOS, the costs of some diagnostic tests are relatively low. For example, based on estimates from Healthcare Bluebook,9 serum TSH and prolactin tests in San Francisco, California, are $44 and $51, respectively. However, the cumulative costs for even the most stringent routine workup for PCOS recommended in the AACE/AE-PCOS guideline consisting of a free testosterone measurement, 17-hydroxyprogesterone, and transvaginal ultrasound would still cost a total of $516. Additional TSH and prolactin tests recommended by ENDO would increase the cost of PCOS testing by approximately 18%. Routine testing for additional serum androgens—dehydroepiandrosterone sulfate (DHEA-S) and androstenedione—would further increase this amount by an additional $134 to a total cost of $745. The ENDO guideline only recommends DHEA-S testing to assist in the diagnosis of an androgen-secreting tumor when signs of virilization are present, while the AACE/AE-PCOS guideline discourages routine testing for DHEA-S and androstenedione based on the low frequency of cases in which these androgens are elevated in isolation.7,8
Although the selection of tests influences total cost, the setting of tests (ie, hospitals, physician offices, independent test settings) also can contribute to wide variations in cost. For example, Healthcare Bluebook’s estimates for transvaginal ultrasound in Chicago, Illinois, range from $236 to more than $740.9 When the separate physician visit fees are included, the total cost of a routine diagnostic evaluation of a patient with acne or hirsutism concerning for PCOS is not trivial.
Large national clinical registries and formal cost-effectiveness analyses are necessary to shed light on this issue, but it is clear that clinicians should rely on their clinical judgment when ordering laboratory tests in the evaluation for PCOS given the apparent low yield of routine screening for PCOS mimickers in the absence of clinical indications. For example, a TSH would not be warranted in a patient without evidence of thyroid dysfunction (ie, weight gain, fatigue, constipation, menstrual irregularities). Similarly, clinicians should routinely consider the principle of high-value care: whether the results of a test will change management of the patient. For example, a woman with amenorrhea and severe acne who already meets diagnostic criteria for PCOS would benefit from a combined oral contraceptive for both acne and endometrial protection. An ovarian ultrasound may not be needed to confirm the diagnosis unless there is suspicion for an ovarian condition other than PCOS causing the symptoms.
Finally, clinicians should discuss testing options and involve patients in decisions around testing. Although PCOS treatments generally target individual symptoms rather than the syndrome as a whole, confirmation of a PCOS diagnosis importantly informs women of their risk for cardiovascular and metabolic disease. The ENDO recommends screening for impaired glucose tolerance, type 2 diabetes mellitus, obesity, family history of early cardiovascular disease, tobacco use, hypertension, dyslipidemia, and obstructive sleep apnea in all women with PCOS, including nonobese patients.7 Ongoing efforts to gain and understand evidence to support high-value, cost-conscious care should be prioritized and kept in balance with shared decision-making in individual patients suspected of having PCOS.
- March WA, Moore VM, Willson KJ, et al. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010;25:544-551.
- Sivayoganathan D, Maruthini D, Glanville JM, et al. Full investigation of patients with polycystic ovary syndrome (PCOS) presenting to four different clinical specialties reveals significant differences and undiagnosed morbidity. Hum Fertil. 2011;14:261-265.
- Schmidt TH, Shinkai K. Evidence-based approach to cutaneous hyperandrogenism in women. J Am Acad Dermatol. 2015;73:672-690.
- Schmidt TH, Khanijow K, Cedars MI, et al. Cutaneous findings and systemic associations in women with polycystic ovary syndrome. JAMA Dermatol. 2015;152:391-398.
- Escobar-Morreale HF, Carmina E, Dewailly D, et al. Epidemiology, diagnosis and management of hirsutism: a consensus statement by the Androgen Excess and Polycystic Ovary Syndrome Society. Hum Reprod Update. 2012;18:146-170.
- Azziz R, Carmina E, Dewailly D, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91:456-488.
- Legro RS, Arslanian SA, Ehermann DA, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline [published online October 22, 2013]. J Clin Endocrinol Metab. 2013;98:4565-4592.
- Goodman NF, Cobin RH, Futterweit W, et al. American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society disease state clinical review: guide to the best practices in the evaluation and treatment of polycystic ovary syndrome—part 1. Endocr Pract. 2015;21:1291-1300.
- Healthcare Bluebook. https://healthcarebluebook.com. Accessed June 13, 2016.
- Azziz R, Sanchez LA, Knochenhauer ES, et al. Androgen excess in women: experience with over 1000 consecutive patients. J Clin Endocrinol Metab. 2004;89:453-462.
- Carmina E, Rosato F, Jannì A, et al. Relative prevalence of different androgen excess disorders in 950 women referred because of clinical hyperandrogenism [published online November 1, 2005]. J Clin Endocrinol Metab. 2006;91:2-6.
- Owens DK, Qaseem A, Chou R, et al. High-value, cost-conscious health care: concepts for clinicians to evaluate the benefits, harms, and costs of medical interventions. Ann Intern Med. 2011;154:174-180.
- Nahuis MJ, Oude Lohuis E, Kose N, et al. Long-term follow-up of laparoscopic electrocautery of the ovaries versus ovulation induction with recombinant FSH in clomiphene citrate-resistant women with polycystic ovary syndrome: an economic evaluation. Hum Reprod Oxf Engl. 2012;27:3577-3582.
- Moolenaar LM, Nahuis MJ, Hompes PG, et al. Cost-effectiveness of treatment strategies in women with PCOS who do not conceive after six cycles of clomiphene citrate. Reprod Biomed Online. 2014;28:606-613.
- Chogle A, Saps M. Yield and cost of performing screening tests for constipation in children. Can J Gastroenterol. 2013;27:E35-E38.
- Lee YH, Scharnitz TP, Muscat J, et al. Laboratory monitoring during isotretinoin therapy for acne: a systematic review and meta-analysis. JAMA Dermatol. 2016;152:35-44.
- Shinkai K, McMichael A, Linos E. Isotretinoin laboratory test monitoring—a call to decrease testing in an era of high-value, cost-conscious care. JAMA Dermatol. 2016;152:17-19.
The adult female patient presenting with severe acne vulgaris may raise special diagnostic concerns, including consideration of an underlying hormonal disorder. Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in women of reproductive age with an estimated prevalence as high as 12%.1 Many women with undiagnosed PCOS may be referred to dermatologists for evaluation of its cutaneous manifestations of hyperandrogenism including acne, hirsutism, and androgenic alopecia.2 Given the prevalence of PCOS and its long-term health implications, dermatologists can play an important role in the initial evaluation of these patients. Acne and androgenic alopecia, however, are quite common, and in the absence of red flags such as menstrual irregularities, virilization, visual field deficits, or signs of Cushing syndrome,3 clinicians must decide when to pursue limited versus comprehensive evaluation.
Despite being common in patients with PCOS, a recent study suggests that acne is an unreliable marker of biochemical hyperandrogenism, and specific features of acne (ie, lesion counts, lesional types, distribution) cannot reliably discriminate women who meet PCOS diagnostic criteria from those who do not.4 Similarly, the study found that androgenic alopecia was not associated with biochemical hyperandrogenism and was no more common in women with PCOS than women of similar age in a high-risk population. Unlike acne and androgenic alopecia, however, the study identified hirsutism, especially truncal hirsutism, as a reliable indicator of hyperandrogenemia and PCOS. Hirsutism also is associated with metabolic sequelae of PCOS. These findings suggest that hirsutism, but not acne or androgenic alopecia, in a female of reproductive age warrants a workup for PCOS.4 This report is consistent with a recommendation from the Androgen Excess and Polycystic Ovary Syndrome Society (AE-PCOS) to pursue a diagnostic evaluation in any woman presenting with hirsutism.5 Acanthosis nigricans also was found to be a reliable indicator of hyperandrogenemia, PCOS, and associated metabolic derangement. Thus, although recent evidence indicates that acne as an isolated cutaneous finding does not warrant further diagnostic evaluation, acne in the setting of hirsutism, acanthosis nigricans, menstrual irregularities, or additional specific signs of endocrine dysregulation should prompt focused workup.4
Multiple clinical practice guidelines for the evaluation of hirsutism and PCOS based on literature review and expert opinion have been proposed5-8; however, these guidelines vary in recommendations for routine diagnostic steps to exclude mimickers of PCOS such as prolactinoma/pituitary adenoma and congenital adrenal hyperplasia (CAH)(Table). In 2009, an AE-PCOS task force suggested that routine testing of thyroid function and serum prolactin in the absence of additional clinical signs may not be necessary based on the low prevalence of thyroid disorders and hyperprolactinemia in patients presenting with hyperandrogenism.6 In 2013, the Endocrine Society’s (ENDO) clinical guideline, however, recommended routine measurement of serum thyroid-stimulating hormone (TSH) to exclude thyroid disease and serum prolactin to exclude hyperprolactinemia in all women before making a diagnosis of PCOS.7 In 2015, the AE-PCOS collaborated with the American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology to publish an updated guideline for best practices, which was consistent with the prior AE-PCOS recommendation in 2009 for routine screening including to test 17-hydroxyprogesterone to exclude nonclassical CAH.8

Importantly, these recommendations for routine testing for mimickers of PCOS are based on the rare prevalence of these etiologies in multiple studies of women presenting for hyperandrogenism. One study included 873 women presenting to an academic reproductive endocrine clinic for evaluation of symptoms potentially related to androgen excess. In addition to cutaneous manifestations of hirsutism, acne, and alopecia, the study also included women presenting with oligomenorrhea/amenorrhea, ovulatory dysfunction, and even virilization.10 A second study included 950 women presenting to academic endocrine departments with hirsutism, acne, or androgenic alopecia.11 Both studies defined hirsutism as having a modified Ferriman-Gallwey score of 6 or greater. Both studies also only measured serum prolactin or TSH when clinically indicated (ie, patients with ovulatory dysfunction).10,11
The diagnostic yield of tests for mimickers of PCOS was exceedingly low in both studies. For example, of the patients evaluated, only 0.4% to 0.7% had thyroid dysfunction, 0% to 0.3% had hyperprolactinemia, 0.2% had androgen-secreting neoplasms, 2.1% to 4.3% had nonclassical CAH, 0.7% had CAH, and 3.8% had HAIR-AN (hyperandrogenism, insulin resistance, and acanthosis nigricans) syndrome.10,11 Because patients in both studies were only tested for hyperprolactinemia and thyroid dysfunction when clinically indicated, it is probable that routine screening without clinical indication would result in even lower yields.
Given the increasing importance of high-value, cost-conscious care,12 clinicians must consider the costs associated with testing in the face of low pretest probability. Although some studies have examined the cost-effectiveness of fertility treatments in PCOS,13,14 no studies have examined the cost-effectiveness of diagnostic strategies for PCOS. Cost-effectiveness studies are emerging to provide important guidance on high-value, cost-conscious diagnostic evaluation and monitoring15 and are much needed in dermatology.16,17
In the case of PCOS, the costs of some diagnostic tests are relatively low. For example, based on estimates from Healthcare Bluebook,9 serum TSH and prolactin tests in San Francisco, California, are $44 and $51, respectively. However, the cumulative costs for even the most stringent routine workup for PCOS recommended in the AACE/AE-PCOS guideline consisting of a free testosterone measurement, 17-hydroxyprogesterone, and transvaginal ultrasound would still cost a total of $516. Additional TSH and prolactin tests recommended by ENDO would increase the cost of PCOS testing by approximately 18%. Routine testing for additional serum androgens—dehydroepiandrosterone sulfate (DHEA-S) and androstenedione—would further increase this amount by an additional $134 to a total cost of $745. The ENDO guideline only recommends DHEA-S testing to assist in the diagnosis of an androgen-secreting tumor when signs of virilization are present, while the AACE/AE-PCOS guideline discourages routine testing for DHEA-S and androstenedione based on the low frequency of cases in which these androgens are elevated in isolation.7,8
Although the selection of tests influences total cost, the setting of tests (ie, hospitals, physician offices, independent test settings) also can contribute to wide variations in cost. For example, Healthcare Bluebook’s estimates for transvaginal ultrasound in Chicago, Illinois, range from $236 to more than $740.9 When the separate physician visit fees are included, the total cost of a routine diagnostic evaluation of a patient with acne or hirsutism concerning for PCOS is not trivial.
Large national clinical registries and formal cost-effectiveness analyses are necessary to shed light on this issue, but it is clear that clinicians should rely on their clinical judgment when ordering laboratory tests in the evaluation for PCOS given the apparent low yield of routine screening for PCOS mimickers in the absence of clinical indications. For example, a TSH would not be warranted in a patient without evidence of thyroid dysfunction (ie, weight gain, fatigue, constipation, menstrual irregularities). Similarly, clinicians should routinely consider the principle of high-value care: whether the results of a test will change management of the patient. For example, a woman with amenorrhea and severe acne who already meets diagnostic criteria for PCOS would benefit from a combined oral contraceptive for both acne and endometrial protection. An ovarian ultrasound may not be needed to confirm the diagnosis unless there is suspicion for an ovarian condition other than PCOS causing the symptoms.
Finally, clinicians should discuss testing options and involve patients in decisions around testing. Although PCOS treatments generally target individual symptoms rather than the syndrome as a whole, confirmation of a PCOS diagnosis importantly informs women of their risk for cardiovascular and metabolic disease. The ENDO recommends screening for impaired glucose tolerance, type 2 diabetes mellitus, obesity, family history of early cardiovascular disease, tobacco use, hypertension, dyslipidemia, and obstructive sleep apnea in all women with PCOS, including nonobese patients.7 Ongoing efforts to gain and understand evidence to support high-value, cost-conscious care should be prioritized and kept in balance with shared decision-making in individual patients suspected of having PCOS.
The adult female patient presenting with severe acne vulgaris may raise special diagnostic concerns, including consideration of an underlying hormonal disorder. Polycystic ovary syndrome (PCOS) is the most common endocrine disorder in women of reproductive age with an estimated prevalence as high as 12%.1 Many women with undiagnosed PCOS may be referred to dermatologists for evaluation of its cutaneous manifestations of hyperandrogenism including acne, hirsutism, and androgenic alopecia.2 Given the prevalence of PCOS and its long-term health implications, dermatologists can play an important role in the initial evaluation of these patients. Acne and androgenic alopecia, however, are quite common, and in the absence of red flags such as menstrual irregularities, virilization, visual field deficits, or signs of Cushing syndrome,3 clinicians must decide when to pursue limited versus comprehensive evaluation.
Despite being common in patients with PCOS, a recent study suggests that acne is an unreliable marker of biochemical hyperandrogenism, and specific features of acne (ie, lesion counts, lesional types, distribution) cannot reliably discriminate women who meet PCOS diagnostic criteria from those who do not.4 Similarly, the study found that androgenic alopecia was not associated with biochemical hyperandrogenism and was no more common in women with PCOS than women of similar age in a high-risk population. Unlike acne and androgenic alopecia, however, the study identified hirsutism, especially truncal hirsutism, as a reliable indicator of hyperandrogenemia and PCOS. Hirsutism also is associated with metabolic sequelae of PCOS. These findings suggest that hirsutism, but not acne or androgenic alopecia, in a female of reproductive age warrants a workup for PCOS.4 This report is consistent with a recommendation from the Androgen Excess and Polycystic Ovary Syndrome Society (AE-PCOS) to pursue a diagnostic evaluation in any woman presenting with hirsutism.5 Acanthosis nigricans also was found to be a reliable indicator of hyperandrogenemia, PCOS, and associated metabolic derangement. Thus, although recent evidence indicates that acne as an isolated cutaneous finding does not warrant further diagnostic evaluation, acne in the setting of hirsutism, acanthosis nigricans, menstrual irregularities, or additional specific signs of endocrine dysregulation should prompt focused workup.4
Multiple clinical practice guidelines for the evaluation of hirsutism and PCOS based on literature review and expert opinion have been proposed5-8; however, these guidelines vary in recommendations for routine diagnostic steps to exclude mimickers of PCOS such as prolactinoma/pituitary adenoma and congenital adrenal hyperplasia (CAH)(Table). In 2009, an AE-PCOS task force suggested that routine testing of thyroid function and serum prolactin in the absence of additional clinical signs may not be necessary based on the low prevalence of thyroid disorders and hyperprolactinemia in patients presenting with hyperandrogenism.6 In 2013, the Endocrine Society’s (ENDO) clinical guideline, however, recommended routine measurement of serum thyroid-stimulating hormone (TSH) to exclude thyroid disease and serum prolactin to exclude hyperprolactinemia in all women before making a diagnosis of PCOS.7 In 2015, the AE-PCOS collaborated with the American Association of Clinical Endocrinologists (AACE) and American College of Endocrinology to publish an updated guideline for best practices, which was consistent with the prior AE-PCOS recommendation in 2009 for routine screening including to test 17-hydroxyprogesterone to exclude nonclassical CAH.8

Importantly, these recommendations for routine testing for mimickers of PCOS are based on the rare prevalence of these etiologies in multiple studies of women presenting for hyperandrogenism. One study included 873 women presenting to an academic reproductive endocrine clinic for evaluation of symptoms potentially related to androgen excess. In addition to cutaneous manifestations of hirsutism, acne, and alopecia, the study also included women presenting with oligomenorrhea/amenorrhea, ovulatory dysfunction, and even virilization.10 A second study included 950 women presenting to academic endocrine departments with hirsutism, acne, or androgenic alopecia.11 Both studies defined hirsutism as having a modified Ferriman-Gallwey score of 6 or greater. Both studies also only measured serum prolactin or TSH when clinically indicated (ie, patients with ovulatory dysfunction).10,11
The diagnostic yield of tests for mimickers of PCOS was exceedingly low in both studies. For example, of the patients evaluated, only 0.4% to 0.7% had thyroid dysfunction, 0% to 0.3% had hyperprolactinemia, 0.2% had androgen-secreting neoplasms, 2.1% to 4.3% had nonclassical CAH, 0.7% had CAH, and 3.8% had HAIR-AN (hyperandrogenism, insulin resistance, and acanthosis nigricans) syndrome.10,11 Because patients in both studies were only tested for hyperprolactinemia and thyroid dysfunction when clinically indicated, it is probable that routine screening without clinical indication would result in even lower yields.
Given the increasing importance of high-value, cost-conscious care,12 clinicians must consider the costs associated with testing in the face of low pretest probability. Although some studies have examined the cost-effectiveness of fertility treatments in PCOS,13,14 no studies have examined the cost-effectiveness of diagnostic strategies for PCOS. Cost-effectiveness studies are emerging to provide important guidance on high-value, cost-conscious diagnostic evaluation and monitoring15 and are much needed in dermatology.16,17
In the case of PCOS, the costs of some diagnostic tests are relatively low. For example, based on estimates from Healthcare Bluebook,9 serum TSH and prolactin tests in San Francisco, California, are $44 and $51, respectively. However, the cumulative costs for even the most stringent routine workup for PCOS recommended in the AACE/AE-PCOS guideline consisting of a free testosterone measurement, 17-hydroxyprogesterone, and transvaginal ultrasound would still cost a total of $516. Additional TSH and prolactin tests recommended by ENDO would increase the cost of PCOS testing by approximately 18%. Routine testing for additional serum androgens—dehydroepiandrosterone sulfate (DHEA-S) and androstenedione—would further increase this amount by an additional $134 to a total cost of $745. The ENDO guideline only recommends DHEA-S testing to assist in the diagnosis of an androgen-secreting tumor when signs of virilization are present, while the AACE/AE-PCOS guideline discourages routine testing for DHEA-S and androstenedione based on the low frequency of cases in which these androgens are elevated in isolation.7,8
Although the selection of tests influences total cost, the setting of tests (ie, hospitals, physician offices, independent test settings) also can contribute to wide variations in cost. For example, Healthcare Bluebook’s estimates for transvaginal ultrasound in Chicago, Illinois, range from $236 to more than $740.9 When the separate physician visit fees are included, the total cost of a routine diagnostic evaluation of a patient with acne or hirsutism concerning for PCOS is not trivial.
Large national clinical registries and formal cost-effectiveness analyses are necessary to shed light on this issue, but it is clear that clinicians should rely on their clinical judgment when ordering laboratory tests in the evaluation for PCOS given the apparent low yield of routine screening for PCOS mimickers in the absence of clinical indications. For example, a TSH would not be warranted in a patient without evidence of thyroid dysfunction (ie, weight gain, fatigue, constipation, menstrual irregularities). Similarly, clinicians should routinely consider the principle of high-value care: whether the results of a test will change management of the patient. For example, a woman with amenorrhea and severe acne who already meets diagnostic criteria for PCOS would benefit from a combined oral contraceptive for both acne and endometrial protection. An ovarian ultrasound may not be needed to confirm the diagnosis unless there is suspicion for an ovarian condition other than PCOS causing the symptoms.
Finally, clinicians should discuss testing options and involve patients in decisions around testing. Although PCOS treatments generally target individual symptoms rather than the syndrome as a whole, confirmation of a PCOS diagnosis importantly informs women of their risk for cardiovascular and metabolic disease. The ENDO recommends screening for impaired glucose tolerance, type 2 diabetes mellitus, obesity, family history of early cardiovascular disease, tobacco use, hypertension, dyslipidemia, and obstructive sleep apnea in all women with PCOS, including nonobese patients.7 Ongoing efforts to gain and understand evidence to support high-value, cost-conscious care should be prioritized and kept in balance with shared decision-making in individual patients suspected of having PCOS.
- March WA, Moore VM, Willson KJ, et al. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010;25:544-551.
- Sivayoganathan D, Maruthini D, Glanville JM, et al. Full investigation of patients with polycystic ovary syndrome (PCOS) presenting to four different clinical specialties reveals significant differences and undiagnosed morbidity. Hum Fertil. 2011;14:261-265.
- Schmidt TH, Shinkai K. Evidence-based approach to cutaneous hyperandrogenism in women. J Am Acad Dermatol. 2015;73:672-690.
- Schmidt TH, Khanijow K, Cedars MI, et al. Cutaneous findings and systemic associations in women with polycystic ovary syndrome. JAMA Dermatol. 2015;152:391-398.
- Escobar-Morreale HF, Carmina E, Dewailly D, et al. Epidemiology, diagnosis and management of hirsutism: a consensus statement by the Androgen Excess and Polycystic Ovary Syndrome Society. Hum Reprod Update. 2012;18:146-170.
- Azziz R, Carmina E, Dewailly D, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91:456-488.
- Legro RS, Arslanian SA, Ehermann DA, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline [published online October 22, 2013]. J Clin Endocrinol Metab. 2013;98:4565-4592.
- Goodman NF, Cobin RH, Futterweit W, et al. American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society disease state clinical review: guide to the best practices in the evaluation and treatment of polycystic ovary syndrome—part 1. Endocr Pract. 2015;21:1291-1300.
- Healthcare Bluebook. https://healthcarebluebook.com. Accessed June 13, 2016.
- Azziz R, Sanchez LA, Knochenhauer ES, et al. Androgen excess in women: experience with over 1000 consecutive patients. J Clin Endocrinol Metab. 2004;89:453-462.
- Carmina E, Rosato F, Jannì A, et al. Relative prevalence of different androgen excess disorders in 950 women referred because of clinical hyperandrogenism [published online November 1, 2005]. J Clin Endocrinol Metab. 2006;91:2-6.
- Owens DK, Qaseem A, Chou R, et al. High-value, cost-conscious health care: concepts for clinicians to evaluate the benefits, harms, and costs of medical interventions. Ann Intern Med. 2011;154:174-180.
- Nahuis MJ, Oude Lohuis E, Kose N, et al. Long-term follow-up of laparoscopic electrocautery of the ovaries versus ovulation induction with recombinant FSH in clomiphene citrate-resistant women with polycystic ovary syndrome: an economic evaluation. Hum Reprod Oxf Engl. 2012;27:3577-3582.
- Moolenaar LM, Nahuis MJ, Hompes PG, et al. Cost-effectiveness of treatment strategies in women with PCOS who do not conceive after six cycles of clomiphene citrate. Reprod Biomed Online. 2014;28:606-613.
- Chogle A, Saps M. Yield and cost of performing screening tests for constipation in children. Can J Gastroenterol. 2013;27:E35-E38.
- Lee YH, Scharnitz TP, Muscat J, et al. Laboratory monitoring during isotretinoin therapy for acne: a systematic review and meta-analysis. JAMA Dermatol. 2016;152:35-44.
- Shinkai K, McMichael A, Linos E. Isotretinoin laboratory test monitoring—a call to decrease testing in an era of high-value, cost-conscious care. JAMA Dermatol. 2016;152:17-19.
- March WA, Moore VM, Willson KJ, et al. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010;25:544-551.
- Sivayoganathan D, Maruthini D, Glanville JM, et al. Full investigation of patients with polycystic ovary syndrome (PCOS) presenting to four different clinical specialties reveals significant differences and undiagnosed morbidity. Hum Fertil. 2011;14:261-265.
- Schmidt TH, Shinkai K. Evidence-based approach to cutaneous hyperandrogenism in women. J Am Acad Dermatol. 2015;73:672-690.
- Schmidt TH, Khanijow K, Cedars MI, et al. Cutaneous findings and systemic associations in women with polycystic ovary syndrome. JAMA Dermatol. 2015;152:391-398.
- Escobar-Morreale HF, Carmina E, Dewailly D, et al. Epidemiology, diagnosis and management of hirsutism: a consensus statement by the Androgen Excess and Polycystic Ovary Syndrome Society. Hum Reprod Update. 2012;18:146-170.
- Azziz R, Carmina E, Dewailly D, et al. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91:456-488.
- Legro RS, Arslanian SA, Ehermann DA, et al. Diagnosis and treatment of polycystic ovary syndrome: an Endocrine Society clinical practice guideline [published online October 22, 2013]. J Clin Endocrinol Metab. 2013;98:4565-4592.
- Goodman NF, Cobin RH, Futterweit W, et al. American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and PCOS Society disease state clinical review: guide to the best practices in the evaluation and treatment of polycystic ovary syndrome—part 1. Endocr Pract. 2015;21:1291-1300.
- Healthcare Bluebook. https://healthcarebluebook.com. Accessed June 13, 2016.
- Azziz R, Sanchez LA, Knochenhauer ES, et al. Androgen excess in women: experience with over 1000 consecutive patients. J Clin Endocrinol Metab. 2004;89:453-462.
- Carmina E, Rosato F, Jannì A, et al. Relative prevalence of different androgen excess disorders in 950 women referred because of clinical hyperandrogenism [published online November 1, 2005]. J Clin Endocrinol Metab. 2006;91:2-6.
- Owens DK, Qaseem A, Chou R, et al. High-value, cost-conscious health care: concepts for clinicians to evaluate the benefits, harms, and costs of medical interventions. Ann Intern Med. 2011;154:174-180.
- Nahuis MJ, Oude Lohuis E, Kose N, et al. Long-term follow-up of laparoscopic electrocautery of the ovaries versus ovulation induction with recombinant FSH in clomiphene citrate-resistant women with polycystic ovary syndrome: an economic evaluation. Hum Reprod Oxf Engl. 2012;27:3577-3582.
- Moolenaar LM, Nahuis MJ, Hompes PG, et al. Cost-effectiveness of treatment strategies in women with PCOS who do not conceive after six cycles of clomiphene citrate. Reprod Biomed Online. 2014;28:606-613.
- Chogle A, Saps M. Yield and cost of performing screening tests for constipation in children. Can J Gastroenterol. 2013;27:E35-E38.
- Lee YH, Scharnitz TP, Muscat J, et al. Laboratory monitoring during isotretinoin therapy for acne: a systematic review and meta-analysis. JAMA Dermatol. 2016;152:35-44.
- Shinkai K, McMichael A, Linos E. Isotretinoin laboratory test monitoring—a call to decrease testing in an era of high-value, cost-conscious care. JAMA Dermatol. 2016;152:17-19.
VA, DoD, and Zika Funding Bills Held Up By Partisan Wrangling
A bill providing $82.5 billion in discretionary funding for 2017 military and Veterans Affairs appropriations and Zika funding has stalled in the Senate. The bill would provide funding for veterans’ benefits and programs and to house, train, and equip military personnel; provide housing and services to military families; and help maintain base infrastructure.
Voting on the bill fell along party lines and failed to advance when it could not muster 60 votes. No Democrats voted for the bill, and 2 Republicans also voted against the bill, which contained an unrelated provision that would have eliminated a rule that bars the flying of Confederate flags at veterans’ cemeteries. Democrats also complained about a provision that waived Clean Water Act rules to allow the increased spraying of pesticides in waterways, even though Zika-carrying Aedes aegypti mosquitoes do not breed in rivers and in other moving waterways. Another provision would have prohibited funding from going to contraceptive services, like Planned Parenthood, for women in Zika-affected areas.
Senate Democrats also expressed concern that the House version of the bill reduced VA funding by $500 million. President Obama has promised to veto the bill unless the additional provisions were removed.
The bill provided $52.8 billion for VA medical services, including $9 billion for mental health care services; $7.3 billion in services for homeless veterans; $1.5 billion for hepatitis C treatment; $284 million for traumatic brain injury treatment; $250 million in rural health initiatives; and $173 million for suicide prevention. Another $260 million in the bill is earmarked for the modernization of the VA electronic health record system.
In addition, the bill contained appropriations of $7.9 billion for military construction. Specifically, the bill included the President’s requested $304 million for construction and alterations of new or existing military medical facilities.
Since February, Democrats have been pushing for emergency funding for Zika, with President Obama asking Congress for $1.9 billion in the fight. The Senate passed legislation in May to provide the $1.1 billion in funding.
The new bill’s failure to pass comes at a season of urgency for combatting against the Zika in southern states, which are expected to be hit the hardest. Florida has reported 223 Zika cases, including 40 pregnant women thus far. The most recent case of an infant born with microcephaly in the U.S. was confirmed in Florida the same day the bill was blocked.
"If you don't think the Zika crisis is an emergency, just wait," warned Senator Bill Nelson, D-Fla. "These numbers are just going to increase...We need to stop playing these political games."
Due to the vote, advances on projects from the National Institute of Allergy and Infectious Diseases (NIAID), such as working on 3 potential vaccines against Zika and overseeing a study of pregnant woman infected, could be slowed.
“If we don’t get new money, we won’t be able to do things at a pace that is necessary and appropriate to the urgency of this threat,” said Dr. Anthony S. Fauci, director of the NIAID.
The bill that was blocked was the third attempt to come to an agreement among Senate Republicans and Democrats on funding for Zika, and with Senate taking a brief recess for the Fourth of July holiday, it leaves little time left to solve the problem.
A bill providing $82.5 billion in discretionary funding for 2017 military and Veterans Affairs appropriations and Zika funding has stalled in the Senate. The bill would provide funding for veterans’ benefits and programs and to house, train, and equip military personnel; provide housing and services to military families; and help maintain base infrastructure.
Voting on the bill fell along party lines and failed to advance when it could not muster 60 votes. No Democrats voted for the bill, and 2 Republicans also voted against the bill, which contained an unrelated provision that would have eliminated a rule that bars the flying of Confederate flags at veterans’ cemeteries. Democrats also complained about a provision that waived Clean Water Act rules to allow the increased spraying of pesticides in waterways, even though Zika-carrying Aedes aegypti mosquitoes do not breed in rivers and in other moving waterways. Another provision would have prohibited funding from going to contraceptive services, like Planned Parenthood, for women in Zika-affected areas.
Senate Democrats also expressed concern that the House version of the bill reduced VA funding by $500 million. President Obama has promised to veto the bill unless the additional provisions were removed.
The bill provided $52.8 billion for VA medical services, including $9 billion for mental health care services; $7.3 billion in services for homeless veterans; $1.5 billion for hepatitis C treatment; $284 million for traumatic brain injury treatment; $250 million in rural health initiatives; and $173 million for suicide prevention. Another $260 million in the bill is earmarked for the modernization of the VA electronic health record system.
In addition, the bill contained appropriations of $7.9 billion for military construction. Specifically, the bill included the President’s requested $304 million for construction and alterations of new or existing military medical facilities.
Since February, Democrats have been pushing for emergency funding for Zika, with President Obama asking Congress for $1.9 billion in the fight. The Senate passed legislation in May to provide the $1.1 billion in funding.
The new bill’s failure to pass comes at a season of urgency for combatting against the Zika in southern states, which are expected to be hit the hardest. Florida has reported 223 Zika cases, including 40 pregnant women thus far. The most recent case of an infant born with microcephaly in the U.S. was confirmed in Florida the same day the bill was blocked.
"If you don't think the Zika crisis is an emergency, just wait," warned Senator Bill Nelson, D-Fla. "These numbers are just going to increase...We need to stop playing these political games."
Due to the vote, advances on projects from the National Institute of Allergy and Infectious Diseases (NIAID), such as working on 3 potential vaccines against Zika and overseeing a study of pregnant woman infected, could be slowed.
“If we don’t get new money, we won’t be able to do things at a pace that is necessary and appropriate to the urgency of this threat,” said Dr. Anthony S. Fauci, director of the NIAID.
The bill that was blocked was the third attempt to come to an agreement among Senate Republicans and Democrats on funding for Zika, and with Senate taking a brief recess for the Fourth of July holiday, it leaves little time left to solve the problem.
A bill providing $82.5 billion in discretionary funding for 2017 military and Veterans Affairs appropriations and Zika funding has stalled in the Senate. The bill would provide funding for veterans’ benefits and programs and to house, train, and equip military personnel; provide housing and services to military families; and help maintain base infrastructure.
Voting on the bill fell along party lines and failed to advance when it could not muster 60 votes. No Democrats voted for the bill, and 2 Republicans also voted against the bill, which contained an unrelated provision that would have eliminated a rule that bars the flying of Confederate flags at veterans’ cemeteries. Democrats also complained about a provision that waived Clean Water Act rules to allow the increased spraying of pesticides in waterways, even though Zika-carrying Aedes aegypti mosquitoes do not breed in rivers and in other moving waterways. Another provision would have prohibited funding from going to contraceptive services, like Planned Parenthood, for women in Zika-affected areas.
Senate Democrats also expressed concern that the House version of the bill reduced VA funding by $500 million. President Obama has promised to veto the bill unless the additional provisions were removed.
The bill provided $52.8 billion for VA medical services, including $9 billion for mental health care services; $7.3 billion in services for homeless veterans; $1.5 billion for hepatitis C treatment; $284 million for traumatic brain injury treatment; $250 million in rural health initiatives; and $173 million for suicide prevention. Another $260 million in the bill is earmarked for the modernization of the VA electronic health record system.
In addition, the bill contained appropriations of $7.9 billion for military construction. Specifically, the bill included the President’s requested $304 million for construction and alterations of new or existing military medical facilities.
Since February, Democrats have been pushing for emergency funding for Zika, with President Obama asking Congress for $1.9 billion in the fight. The Senate passed legislation in May to provide the $1.1 billion in funding.
The new bill’s failure to pass comes at a season of urgency for combatting against the Zika in southern states, which are expected to be hit the hardest. Florida has reported 223 Zika cases, including 40 pregnant women thus far. The most recent case of an infant born with microcephaly in the U.S. was confirmed in Florida the same day the bill was blocked.
"If you don't think the Zika crisis is an emergency, just wait," warned Senator Bill Nelson, D-Fla. "These numbers are just going to increase...We need to stop playing these political games."
Due to the vote, advances on projects from the National Institute of Allergy and Infectious Diseases (NIAID), such as working on 3 potential vaccines against Zika and overseeing a study of pregnant woman infected, could be slowed.
“If we don’t get new money, we won’t be able to do things at a pace that is necessary and appropriate to the urgency of this threat,” said Dr. Anthony S. Fauci, director of the NIAID.
The bill that was blocked was the third attempt to come to an agreement among Senate Republicans and Democrats on funding for Zika, and with Senate taking a brief recess for the Fourth of July holiday, it leaves little time left to solve the problem.
Can Stem Cells Halt Progression of MS?
NATIONAL HARBOR, MD—High-dose immunosuppressive therapy and autologous hematopoietic cell transplantation induces a high rate of remission among patients with highly active relapsing-remitting multiple sclerosis (MS), according to data presented at the 2016 CMSC Annual Meeting. Furthermore, that remission is sustained for five years without maintenance therapy. "We saw about 70% long-term, disease-free survival at five years," said Richard A. Nash, MD, a physician at Colorado Blood Cancer Institute in Denver and a HALT-MS investigator.
Richard A. Nash, MD
“What we studied was high-dose therapy, followed by transplant, for treatment of patients with poor-prognosis MS,” said Dr. Nash on behalf of his study collaborators. Their hypothesis was that intensive immunosuppressive therapy followed by transplant would arrest MS disease activity. Their phase II multicenter study had a prospective, open-label, single-arm design. There were three centers involved—Baylor College of Medicine in Houston, the Fred Hutchinson Cancer Research Center in Seattle, and the Ohio State University MS Center in Columbus.
The aim of the study was to determine the five-year durability of disease stabilization in patients with MS after high-dose therapy and autologous transplantation. An interim analysis at three years was published in JAMA Neurology.
The primary end point was event-free survival after transplant in the five-year period of follow-up. The end point included relapse, defined as neurologic signs or symptoms lasting more than 48 hours; MRI abnormalities at more than 12 months after transplant; progression in disability after six months post-transplant (as measured by Expanded Disability Status Scale or EDSS); and mortality. The end point was similar to no evidence of disease activity (NEDA), said Dr. Nash.
Patients who were eligible for the study were age 18 to 60, met McDonald criteria, and had had MS for less than 15 years. Patients had relapsing-remitting MS with cumulative disability or progressive-relapsing MS. EDSS score had to be between 3.0 and 5.5. Patients had to have T2 abnormalities consistent with MS and two or more relapses within 18 months on therapy, with an EDSS increase of more than 0.5. “I don’t think we had anyone in the study that met the second criterion, which was relapse on therapy with an EDSS increase of more than 1.0 and one separate event with gadolinium-enhancing lesions on MRI,” Dr. Nash said. A panel of two neurologists and one transplant physician reviewed the patients.
There were 25 patients in the HALT-MS trial. Median age at mobilization was 37 (range, 26 to 52). The study cohort was mostly female (17 female, 8 male). Baseline EDSS score was 4.5 (range, 3.0 to 5.5). Median disease duration was 4.9 years (range, seven months to 12 years). Therapies that patients had failed prior to study entry included interferon beta-1a (22 patients), interferon beta-1b (one patient), glatiramer acetate (18 patients), mitoxantrone (eight patients), natalizumab (six patients), and other therapies (11 patients).
James D. Bowen, MD, of the Swedish Neuroscience Institute in Seattle and a HALT-MS investigator, described the study intervention. “The protocol starts with mobilization of stem cells. Patients receive prednisone during this time so that the CSF is not vulnerable to MS attack. They then undergo leukapheresis to collect stem cells, which are further concentrated with a CD34 selection procedure that allows us to purify them.” Most patients with cancer require four or five pheresis sessions, Dr. Bowen noted, but patients with MS generally have healthy bone marrow, so most of the participants in HALT-MS only required two sessions. A few required three pheresis sessions.
“For our transplant protocol, we used BEAM plus ATG, which is a five-drug cocktail—BCNU, etoposide, Ara C, melphalan, and rabbit antithymocyte globulin,” said Dr. Bowen. “Immediately following that, transplant of the stem cells was done. After that, they got granulocyte-colony stimulating factor (G-CSF) to rev up the bone marrow, and we cover that with prednisone at days seven to 21 to avoid MS attacks precipitated by graft syndrome.”
Twenty-four patients went on to transplant. One of the patients had a pulmonary embolus and was not deemed a good candidate for transplantation. The rate of event-free survival, the primary end point, was 69.2%. Seven patients met the primary end point: two by progression of their disease, three by relapse of their disease, and two patients met end points much later in the follow-up, by changes in their MRI at 3.5 to 4.0 years.
The rate of five-year relapse-free survival was 86.9%. Two patients had changes on MRI. The two patients met primary end points and had their disease activity about 3.5 years after transplant. “There was an early event as well, at one year, but the patient who had MRI changes at one year had already met a primary end point by relapsing at about six months,” Dr. Nash said. The rate of disease progression-free survival was 91%.
Regarding change from baseline in T1 and T2 lesion volume, “we had very low incidence, except for two patients who at about 3.5 to 4.0 years after transplant became positive,” Dr. Nash reported. No other patients had development of significant lesions. The T2 lesion volume actually decreased over time and remained decreased through the five years of follow-up. Starting at about three years after transplantation, the researchers noted stability of brain volume.
The researchers recorded three deaths that were considered to be unrelated to the transplant. Two deaths were thought to be possibly related to progression of MS, and one death was in a patient who had evidence of asthma prior to transplant. “That patient was seen by pulmonary medicine, and it was thought that the patient was reasonable to go on to transplant, but the patient had persistent problems after transplant and died at about four years after the transplant,” Dr. Nash said.
The transplant itself appeared to be well tolerated. “There were few serious complications other than what we might expect from a transplant,” Dr. Nash said. It was highly effective for inducing sustained remission for highly active patients with relapsing-remitting MS through five years, and these patients have not received any maintenance therapy since the transplant. MRI lesion volume was reduced, and the brain volume stabilized at years three through five.
The HALT-MS study was sponsored by the National Institute of Allergy & Infectious Diseases (NIAID) and run by the Immune Tolerance Network.
—Glenn S. Williams
Suggested Reading
Bowen JD, Kraft GH, Wundes A, et al. Autologous hematopoietic cell transplantation following high-dose immunosuppressive therapy for advanced multiple sclerosis: long-term results. Bone Marrow Transplant. 2012;47(7):946-951.
Nash RA, Bowen JD, McSweeney PA, et al. High-dose immunosuppressive therapy and autologous peripheral blood stem cell transplantation for severe multiple sclerosis. Blood. 2003;102(7):2364-2372.
Nash RA, Hutton GJ, Racke MK, et al. High-dose immunosuppressive therapy and autologous hematopoietic cell transplantation for relapsing-remitting multiple sclerosis (HALT-MS): a 3-year interim report. JAMA Neurol. 2015;72(2):159-169.
NATIONAL HARBOR, MD—High-dose immunosuppressive therapy and autologous hematopoietic cell transplantation induces a high rate of remission among patients with highly active relapsing-remitting multiple sclerosis (MS), according to data presented at the 2016 CMSC Annual Meeting. Furthermore, that remission is sustained for five years without maintenance therapy. "We saw about 70% long-term, disease-free survival at five years," said Richard A. Nash, MD, a physician at Colorado Blood Cancer Institute in Denver and a HALT-MS investigator.
Richard A. Nash, MD
“What we studied was high-dose therapy, followed by transplant, for treatment of patients with poor-prognosis MS,” said Dr. Nash on behalf of his study collaborators. Their hypothesis was that intensive immunosuppressive therapy followed by transplant would arrest MS disease activity. Their phase II multicenter study had a prospective, open-label, single-arm design. There were three centers involved—Baylor College of Medicine in Houston, the Fred Hutchinson Cancer Research Center in Seattle, and the Ohio State University MS Center in Columbus.
The aim of the study was to determine the five-year durability of disease stabilization in patients with MS after high-dose therapy and autologous transplantation. An interim analysis at three years was published in JAMA Neurology.
The primary end point was event-free survival after transplant in the five-year period of follow-up. The end point included relapse, defined as neurologic signs or symptoms lasting more than 48 hours; MRI abnormalities at more than 12 months after transplant; progression in disability after six months post-transplant (as measured by Expanded Disability Status Scale or EDSS); and mortality. The end point was similar to no evidence of disease activity (NEDA), said Dr. Nash.
Patients who were eligible for the study were age 18 to 60, met McDonald criteria, and had had MS for less than 15 years. Patients had relapsing-remitting MS with cumulative disability or progressive-relapsing MS. EDSS score had to be between 3.0 and 5.5. Patients had to have T2 abnormalities consistent with MS and two or more relapses within 18 months on therapy, with an EDSS increase of more than 0.5. “I don’t think we had anyone in the study that met the second criterion, which was relapse on therapy with an EDSS increase of more than 1.0 and one separate event with gadolinium-enhancing lesions on MRI,” Dr. Nash said. A panel of two neurologists and one transplant physician reviewed the patients.
There were 25 patients in the HALT-MS trial. Median age at mobilization was 37 (range, 26 to 52). The study cohort was mostly female (17 female, 8 male). Baseline EDSS score was 4.5 (range, 3.0 to 5.5). Median disease duration was 4.9 years (range, seven months to 12 years). Therapies that patients had failed prior to study entry included interferon beta-1a (22 patients), interferon beta-1b (one patient), glatiramer acetate (18 patients), mitoxantrone (eight patients), natalizumab (six patients), and other therapies (11 patients).
James D. Bowen, MD, of the Swedish Neuroscience Institute in Seattle and a HALT-MS investigator, described the study intervention. “The protocol starts with mobilization of stem cells. Patients receive prednisone during this time so that the CSF is not vulnerable to MS attack. They then undergo leukapheresis to collect stem cells, which are further concentrated with a CD34 selection procedure that allows us to purify them.” Most patients with cancer require four or five pheresis sessions, Dr. Bowen noted, but patients with MS generally have healthy bone marrow, so most of the participants in HALT-MS only required two sessions. A few required three pheresis sessions.
“For our transplant protocol, we used BEAM plus ATG, which is a five-drug cocktail—BCNU, etoposide, Ara C, melphalan, and rabbit antithymocyte globulin,” said Dr. Bowen. “Immediately following that, transplant of the stem cells was done. After that, they got granulocyte-colony stimulating factor (G-CSF) to rev up the bone marrow, and we cover that with prednisone at days seven to 21 to avoid MS attacks precipitated by graft syndrome.”
Twenty-four patients went on to transplant. One of the patients had a pulmonary embolus and was not deemed a good candidate for transplantation. The rate of event-free survival, the primary end point, was 69.2%. Seven patients met the primary end point: two by progression of their disease, three by relapse of their disease, and two patients met end points much later in the follow-up, by changes in their MRI at 3.5 to 4.0 years.
The rate of five-year relapse-free survival was 86.9%. Two patients had changes on MRI. The two patients met primary end points and had their disease activity about 3.5 years after transplant. “There was an early event as well, at one year, but the patient who had MRI changes at one year had already met a primary end point by relapsing at about six months,” Dr. Nash said. The rate of disease progression-free survival was 91%.
Regarding change from baseline in T1 and T2 lesion volume, “we had very low incidence, except for two patients who at about 3.5 to 4.0 years after transplant became positive,” Dr. Nash reported. No other patients had development of significant lesions. The T2 lesion volume actually decreased over time and remained decreased through the five years of follow-up. Starting at about three years after transplantation, the researchers noted stability of brain volume.
The researchers recorded three deaths that were considered to be unrelated to the transplant. Two deaths were thought to be possibly related to progression of MS, and one death was in a patient who had evidence of asthma prior to transplant. “That patient was seen by pulmonary medicine, and it was thought that the patient was reasonable to go on to transplant, but the patient had persistent problems after transplant and died at about four years after the transplant,” Dr. Nash said.
The transplant itself appeared to be well tolerated. “There were few serious complications other than what we might expect from a transplant,” Dr. Nash said. It was highly effective for inducing sustained remission for highly active patients with relapsing-remitting MS through five years, and these patients have not received any maintenance therapy since the transplant. MRI lesion volume was reduced, and the brain volume stabilized at years three through five.
The HALT-MS study was sponsored by the National Institute of Allergy & Infectious Diseases (NIAID) and run by the Immune Tolerance Network.
—Glenn S. Williams
NATIONAL HARBOR, MD—High-dose immunosuppressive therapy and autologous hematopoietic cell transplantation induces a high rate of remission among patients with highly active relapsing-remitting multiple sclerosis (MS), according to data presented at the 2016 CMSC Annual Meeting. Furthermore, that remission is sustained for five years without maintenance therapy. "We saw about 70% long-term, disease-free survival at five years," said Richard A. Nash, MD, a physician at Colorado Blood Cancer Institute in Denver and a HALT-MS investigator.
Richard A. Nash, MD
“What we studied was high-dose therapy, followed by transplant, for treatment of patients with poor-prognosis MS,” said Dr. Nash on behalf of his study collaborators. Their hypothesis was that intensive immunosuppressive therapy followed by transplant would arrest MS disease activity. Their phase II multicenter study had a prospective, open-label, single-arm design. There were three centers involved—Baylor College of Medicine in Houston, the Fred Hutchinson Cancer Research Center in Seattle, and the Ohio State University MS Center in Columbus.
The aim of the study was to determine the five-year durability of disease stabilization in patients with MS after high-dose therapy and autologous transplantation. An interim analysis at three years was published in JAMA Neurology.
The primary end point was event-free survival after transplant in the five-year period of follow-up. The end point included relapse, defined as neurologic signs or symptoms lasting more than 48 hours; MRI abnormalities at more than 12 months after transplant; progression in disability after six months post-transplant (as measured by Expanded Disability Status Scale or EDSS); and mortality. The end point was similar to no evidence of disease activity (NEDA), said Dr. Nash.
Patients who were eligible for the study were age 18 to 60, met McDonald criteria, and had had MS for less than 15 years. Patients had relapsing-remitting MS with cumulative disability or progressive-relapsing MS. EDSS score had to be between 3.0 and 5.5. Patients had to have T2 abnormalities consistent with MS and two or more relapses within 18 months on therapy, with an EDSS increase of more than 0.5. “I don’t think we had anyone in the study that met the second criterion, which was relapse on therapy with an EDSS increase of more than 1.0 and one separate event with gadolinium-enhancing lesions on MRI,” Dr. Nash said. A panel of two neurologists and one transplant physician reviewed the patients.
There were 25 patients in the HALT-MS trial. Median age at mobilization was 37 (range, 26 to 52). The study cohort was mostly female (17 female, 8 male). Baseline EDSS score was 4.5 (range, 3.0 to 5.5). Median disease duration was 4.9 years (range, seven months to 12 years). Therapies that patients had failed prior to study entry included interferon beta-1a (22 patients), interferon beta-1b (one patient), glatiramer acetate (18 patients), mitoxantrone (eight patients), natalizumab (six patients), and other therapies (11 patients).
James D. Bowen, MD, of the Swedish Neuroscience Institute in Seattle and a HALT-MS investigator, described the study intervention. “The protocol starts with mobilization of stem cells. Patients receive prednisone during this time so that the CSF is not vulnerable to MS attack. They then undergo leukapheresis to collect stem cells, which are further concentrated with a CD34 selection procedure that allows us to purify them.” Most patients with cancer require four or five pheresis sessions, Dr. Bowen noted, but patients with MS generally have healthy bone marrow, so most of the participants in HALT-MS only required two sessions. A few required three pheresis sessions.
“For our transplant protocol, we used BEAM plus ATG, which is a five-drug cocktail—BCNU, etoposide, Ara C, melphalan, and rabbit antithymocyte globulin,” said Dr. Bowen. “Immediately following that, transplant of the stem cells was done. After that, they got granulocyte-colony stimulating factor (G-CSF) to rev up the bone marrow, and we cover that with prednisone at days seven to 21 to avoid MS attacks precipitated by graft syndrome.”
Twenty-four patients went on to transplant. One of the patients had a pulmonary embolus and was not deemed a good candidate for transplantation. The rate of event-free survival, the primary end point, was 69.2%. Seven patients met the primary end point: two by progression of their disease, three by relapse of their disease, and two patients met end points much later in the follow-up, by changes in their MRI at 3.5 to 4.0 years.
The rate of five-year relapse-free survival was 86.9%. Two patients had changes on MRI. The two patients met primary end points and had their disease activity about 3.5 years after transplant. “There was an early event as well, at one year, but the patient who had MRI changes at one year had already met a primary end point by relapsing at about six months,” Dr. Nash said. The rate of disease progression-free survival was 91%.
Regarding change from baseline in T1 and T2 lesion volume, “we had very low incidence, except for two patients who at about 3.5 to 4.0 years after transplant became positive,” Dr. Nash reported. No other patients had development of significant lesions. The T2 lesion volume actually decreased over time and remained decreased through the five years of follow-up. Starting at about three years after transplantation, the researchers noted stability of brain volume.
The researchers recorded three deaths that were considered to be unrelated to the transplant. Two deaths were thought to be possibly related to progression of MS, and one death was in a patient who had evidence of asthma prior to transplant. “That patient was seen by pulmonary medicine, and it was thought that the patient was reasonable to go on to transplant, but the patient had persistent problems after transplant and died at about four years after the transplant,” Dr. Nash said.
The transplant itself appeared to be well tolerated. “There were few serious complications other than what we might expect from a transplant,” Dr. Nash said. It was highly effective for inducing sustained remission for highly active patients with relapsing-remitting MS through five years, and these patients have not received any maintenance therapy since the transplant. MRI lesion volume was reduced, and the brain volume stabilized at years three through five.
The HALT-MS study was sponsored by the National Institute of Allergy & Infectious Diseases (NIAID) and run by the Immune Tolerance Network.
—Glenn S. Williams
Suggested Reading
Bowen JD, Kraft GH, Wundes A, et al. Autologous hematopoietic cell transplantation following high-dose immunosuppressive therapy for advanced multiple sclerosis: long-term results. Bone Marrow Transplant. 2012;47(7):946-951.
Nash RA, Bowen JD, McSweeney PA, et al. High-dose immunosuppressive therapy and autologous peripheral blood stem cell transplantation for severe multiple sclerosis. Blood. 2003;102(7):2364-2372.
Nash RA, Hutton GJ, Racke MK, et al. High-dose immunosuppressive therapy and autologous hematopoietic cell transplantation for relapsing-remitting multiple sclerosis (HALT-MS): a 3-year interim report. JAMA Neurol. 2015;72(2):159-169.
Suggested Reading
Bowen JD, Kraft GH, Wundes A, et al. Autologous hematopoietic cell transplantation following high-dose immunosuppressive therapy for advanced multiple sclerosis: long-term results. Bone Marrow Transplant. 2012;47(7):946-951.
Nash RA, Bowen JD, McSweeney PA, et al. High-dose immunosuppressive therapy and autologous peripheral blood stem cell transplantation for severe multiple sclerosis. Blood. 2003;102(7):2364-2372.
Nash RA, Hutton GJ, Racke MK, et al. High-dose immunosuppressive therapy and autologous hematopoietic cell transplantation for relapsing-remitting multiple sclerosis (HALT-MS): a 3-year interim report. JAMA Neurol. 2015;72(2):159-169.
Individual Patient Characteristics Should Guide Choice of Device for Epilepsy Seizure Control
VANCOUVER—For patients with refractory epilepsy who are not candidates for resective surgery, neuromodulatory devices can be helpful. But which device should be considered for which patient? “Individualize the treatment,” said Christianne Heck, MD, at the 68th Annual Meeting of the American Academy of Neurology. With open- and closed-loop treatments available, factors such as compliance and patient lifestyle are some of the considerations physicians should keep in mind.
Of the approximately 50 million people worldwide with epilepsy, more than one-third have drug-resistant epilepsy (DRE). “DRE is defined as the failure of two or more appropriate antiepileptic drugs to achieve complete seizure control,” said Dr. Heck, Medical Director of the University of Southern California Comprehensive Epilepsy Program at the Keck Medical Center in Los Angeles. “It is associated with major depression, an increased risk in injury and death, cognitive and attention deficits, lower quality of life, and increased healthcare costs.”
Even as new medications have become available, drug resistance rates have remained the same. In addition, adverse effects are quite common and interfere with daily living. “They’re the primary cause of treatment failure in 13% to 40% of patients, depending on the drug,” Dr. Heck noted. “Devices for epilepsy have demonstrated quality-of-life improvements and improvements in mood scores, as well as sustained effectiveness over time.”
Surgery is appropriate for about half of patients with DRE who undergo a presurgical workup in an epilepsy center. The other 50%, however, either have an epileptic zone that is not well defined or an epileptic zone that localizes to eloquent cortex that cannot be resected.
Vagus Nerve Stimulation
Open-loop devices provide automated continuous or nearly continuous stimulation to the area of focus. They control excess electrical activity in the brain by administering regular electrical impulses to reduce the frequency and severity of seizures. Among the open-loop systems is vagus nerve stimulation (VNS), which was approved by the FDA as adjunctive therapy in reducing the frequency of seizures in adults and in adolescents older than 12 with refractory partial onset seizures.
The VNS device is implanted under the skin in the left chest. A horizontal incision in the left neck allows for an electrode from the stimulator to be wrapped around the left vagus nerve. In the Vagus Nerve Stimulation Study Group E01-E05 trials, researchers observed a 43% responder rate—defined as a 50% or greater reduction in seizure frequency—among VNS users at three years.
While VNS is an open-loop system, patients who have auras and can tell when a seizure might occur can pass a special magnet near the implanted device to trigger stimulation outside of the programmed intervals on demand, Dr. Heck said. “However, some patients say they have no way to know when to use the magnet. I try to make it clear to them that magnet activation is a luxury, not a requirement.”
A closed-loop VNS device, AspireSR (Cyberonics), was approved by the FDA in 2015. Closed-loop devices provide stimulation only in response to a trigger. For AspireSR, elevated heart rate triggers stimulation. “The parameters for the change in heart rate can be adjusted based upon the individual’s cardiac changes,” said Dr. Heck. “But it also works in conjunction with its normal cycling, as well as magnet modes.”
One concern about using heart rate as a biomarker for epileptic seizures is that the definition of ictal tachycardia varies widely in the medical literature—from 100 bpm to 140 bpm to a 10-bpm increase. “It’s difficult at this point to determine whether heart rate is the ideal biomarker,” Dr. Heck noted.
The automated nature of VNS therapy lessens concerns about patient compliance, and the treatment has minimal side effects, Dr. Heck said. Patients may, however, experience hoarseness or other voice changes during stimulation—a consideration for those whose voice is an important part of their job, including singers. In addition, patients may become slightly out of breath during stimulation, which could be a concern for athletes. Furthermore, the device is costly and requires surgical implantation, yet there is no way to predict before surgery whether the patient’s seizures will be responsive to neurostimulation therapy.
Deep Brain Stimulation
Deep brain stimulation (DBS) for epilepsy targets the anterior nucleus of the thalamus. DBS for the treatment of epilepsy has not yet been approved in the US. During DBS surgery, two electrodes are placed deep inside the brain through small holes made in the skull. MRI or another imaging technique helps ensure the electrodes are placed correctly. The electrodes are connected to wires that run under the skin behind the ear and down the neck to a neurostimulator that is implanted under the skin in the upper chest, below the collarbone. As with VNS, a patient with auras can activate the device in addition to receiving automated treatment.
Dr. Heck cited a recent trial of DBS that included 110 patients with epilepsy who had experienced at least six seizures per month and failed at least three antiepileptic drugs. During long-term follow-up, the responder rate was 43% at one year and 68% at five years. Sixteen percent of the patients were seizure-free for at least six months, and scores on the Liverpool Seizure Severity Scale improved over baseline. There was an adverse event rate of 34%.
Noninvasive Stimulation
Another open-loop device is the external trigeminal nerve stimulator (eTNS), which is noninvasive and easy to use, Dr. Heck said. “It’s preset with a certain stimulation algorithm and therefore doesn’t require programming. The patient simply adjusts the current to [his or her] comfort level.”
The eTNS unit includes an electrode that is applied to the forehead. Patients can wear the device at night or under a cap during the day, Dr. Heck said. eTNS is approved for prescription use in the EU, Canada, and Australia as adjunctive therapy for epilepsy in patients ages 9 and older.
In a small, 18-week randomized active control trial of 50 patients with DRE who experienced two or more partial onset seizures per month, researchers observed a 40% improvement in the treatment group versus a 16% improvement in the control group. “Although we don’t have any data on this so far, eTNS may be a convenient way to test whether neuromodulation works for patients before they invest in an implantable device that is considerably more expensive,” Dr. Heck said.
Responsive Neurostimulation
Responsive neurostimulation (RNS) devices are placed under the scalp and within the skull. One or two electrodes connected to the neurostimulator are placed at the seizure focus site.
“At this point, I think that the RNS System [NeuroPace] is the gold standard for responsive closed-loop epilepsy therapy. It responds specifically to cortical activity that represents seizure,” Dr. Heck said. “The cranial implant is not visible, which is good for patients who care about scarring. The small stimulation pulses [are] delivered through the leads directly to the brain and those pulses add up to less than six minutes of electrical stimulation per day on average.” The RNS System received premarket approval by the FDA in late 2013 to treat medically refractory partial epilepsy.
Patients do not know when they are receiving stimulation. RNS “provides an alternative for localization-related DRE when surgery is not an option,” she added. RNS also may be appropriate for patients who have multiple seizure onset zones because more than one electrode can be used to cover various sites.
A disadvantage of using RNS is the shorter battery life, compared with VNS (three to four years vs four to 10 years, respectively). In addition, it is a complex device to use. “I began to work with this device in 2006 and I still would not claim to be an absolute expert yet. I think it’s an exciting kind of technology, but the actual parameters for therapy still need to be worked out,” Dr. Heck said. Because of the need for frequent follow-up visits to program the device, RNS may be a difficult treatment option for patients who must travel long distances.
The RNS System Pivotal trial evaluated responder rates at two years. Patients using RNS demonstrated gradual improvement, with median seizure reductions of approximately 53%, compared with baseline. There were no persistent clinically significant neurologic sequelae.
—Adriene Marshall
Suggested Reading
DeGiorgio CM, Soss J, Cook IA, et al. Randomized controlled trial of trigeminal nerve stimulation for drug-resistant epilepsy. Neurology. 2013;80(9):786-791.
Heck CN, King-Stephens D, Massey AD, et al. Two-year seizure reduction in adults with medically intractable partial onset epilepsy treated with responsive neurostimulation: final results of the RNS System Pivotal trial. Epilepsia. 2014;55(3):432-441.
Morris GL 3rd, Mueller WM. Long-term treatment with vagus nerve stimulation in patients with refractory epilepsy. The Vagus Nerve Stimulation Study Group E01-E05. Neurology. 1999;53(8):1731-1735.
Salanova V, Witt T, Worth R, et al; SANTE Study Group. Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology. 2015;84(10):1017-1025.
VANCOUVER—For patients with refractory epilepsy who are not candidates for resective surgery, neuromodulatory devices can be helpful. But which device should be considered for which patient? “Individualize the treatment,” said Christianne Heck, MD, at the 68th Annual Meeting of the American Academy of Neurology. With open- and closed-loop treatments available, factors such as compliance and patient lifestyle are some of the considerations physicians should keep in mind.
Of the approximately 50 million people worldwide with epilepsy, more than one-third have drug-resistant epilepsy (DRE). “DRE is defined as the failure of two or more appropriate antiepileptic drugs to achieve complete seizure control,” said Dr. Heck, Medical Director of the University of Southern California Comprehensive Epilepsy Program at the Keck Medical Center in Los Angeles. “It is associated with major depression, an increased risk in injury and death, cognitive and attention deficits, lower quality of life, and increased healthcare costs.”
Even as new medications have become available, drug resistance rates have remained the same. In addition, adverse effects are quite common and interfere with daily living. “They’re the primary cause of treatment failure in 13% to 40% of patients, depending on the drug,” Dr. Heck noted. “Devices for epilepsy have demonstrated quality-of-life improvements and improvements in mood scores, as well as sustained effectiveness over time.”
Surgery is appropriate for about half of patients with DRE who undergo a presurgical workup in an epilepsy center. The other 50%, however, either have an epileptic zone that is not well defined or an epileptic zone that localizes to eloquent cortex that cannot be resected.
Vagus Nerve Stimulation
Open-loop devices provide automated continuous or nearly continuous stimulation to the area of focus. They control excess electrical activity in the brain by administering regular electrical impulses to reduce the frequency and severity of seizures. Among the open-loop systems is vagus nerve stimulation (VNS), which was approved by the FDA as adjunctive therapy in reducing the frequency of seizures in adults and in adolescents older than 12 with refractory partial onset seizures.
The VNS device is implanted under the skin in the left chest. A horizontal incision in the left neck allows for an electrode from the stimulator to be wrapped around the left vagus nerve. In the Vagus Nerve Stimulation Study Group E01-E05 trials, researchers observed a 43% responder rate—defined as a 50% or greater reduction in seizure frequency—among VNS users at three years.
While VNS is an open-loop system, patients who have auras and can tell when a seizure might occur can pass a special magnet near the implanted device to trigger stimulation outside of the programmed intervals on demand, Dr. Heck said. “However, some patients say they have no way to know when to use the magnet. I try to make it clear to them that magnet activation is a luxury, not a requirement.”
A closed-loop VNS device, AspireSR (Cyberonics), was approved by the FDA in 2015. Closed-loop devices provide stimulation only in response to a trigger. For AspireSR, elevated heart rate triggers stimulation. “The parameters for the change in heart rate can be adjusted based upon the individual’s cardiac changes,” said Dr. Heck. “But it also works in conjunction with its normal cycling, as well as magnet modes.”
One concern about using heart rate as a biomarker for epileptic seizures is that the definition of ictal tachycardia varies widely in the medical literature—from 100 bpm to 140 bpm to a 10-bpm increase. “It’s difficult at this point to determine whether heart rate is the ideal biomarker,” Dr. Heck noted.
The automated nature of VNS therapy lessens concerns about patient compliance, and the treatment has minimal side effects, Dr. Heck said. Patients may, however, experience hoarseness or other voice changes during stimulation—a consideration for those whose voice is an important part of their job, including singers. In addition, patients may become slightly out of breath during stimulation, which could be a concern for athletes. Furthermore, the device is costly and requires surgical implantation, yet there is no way to predict before surgery whether the patient’s seizures will be responsive to neurostimulation therapy.
Deep Brain Stimulation
Deep brain stimulation (DBS) for epilepsy targets the anterior nucleus of the thalamus. DBS for the treatment of epilepsy has not yet been approved in the US. During DBS surgery, two electrodes are placed deep inside the brain through small holes made in the skull. MRI or another imaging technique helps ensure the electrodes are placed correctly. The electrodes are connected to wires that run under the skin behind the ear and down the neck to a neurostimulator that is implanted under the skin in the upper chest, below the collarbone. As with VNS, a patient with auras can activate the device in addition to receiving automated treatment.
Dr. Heck cited a recent trial of DBS that included 110 patients with epilepsy who had experienced at least six seizures per month and failed at least three antiepileptic drugs. During long-term follow-up, the responder rate was 43% at one year and 68% at five years. Sixteen percent of the patients were seizure-free for at least six months, and scores on the Liverpool Seizure Severity Scale improved over baseline. There was an adverse event rate of 34%.
Noninvasive Stimulation
Another open-loop device is the external trigeminal nerve stimulator (eTNS), which is noninvasive and easy to use, Dr. Heck said. “It’s preset with a certain stimulation algorithm and therefore doesn’t require programming. The patient simply adjusts the current to [his or her] comfort level.”
The eTNS unit includes an electrode that is applied to the forehead. Patients can wear the device at night or under a cap during the day, Dr. Heck said. eTNS is approved for prescription use in the EU, Canada, and Australia as adjunctive therapy for epilepsy in patients ages 9 and older.
In a small, 18-week randomized active control trial of 50 patients with DRE who experienced two or more partial onset seizures per month, researchers observed a 40% improvement in the treatment group versus a 16% improvement in the control group. “Although we don’t have any data on this so far, eTNS may be a convenient way to test whether neuromodulation works for patients before they invest in an implantable device that is considerably more expensive,” Dr. Heck said.
Responsive Neurostimulation
Responsive neurostimulation (RNS) devices are placed under the scalp and within the skull. One or two electrodes connected to the neurostimulator are placed at the seizure focus site.
“At this point, I think that the RNS System [NeuroPace] is the gold standard for responsive closed-loop epilepsy therapy. It responds specifically to cortical activity that represents seizure,” Dr. Heck said. “The cranial implant is not visible, which is good for patients who care about scarring. The small stimulation pulses [are] delivered through the leads directly to the brain and those pulses add up to less than six minutes of electrical stimulation per day on average.” The RNS System received premarket approval by the FDA in late 2013 to treat medically refractory partial epilepsy.
Patients do not know when they are receiving stimulation. RNS “provides an alternative for localization-related DRE when surgery is not an option,” she added. RNS also may be appropriate for patients who have multiple seizure onset zones because more than one electrode can be used to cover various sites.
A disadvantage of using RNS is the shorter battery life, compared with VNS (three to four years vs four to 10 years, respectively). In addition, it is a complex device to use. “I began to work with this device in 2006 and I still would not claim to be an absolute expert yet. I think it’s an exciting kind of technology, but the actual parameters for therapy still need to be worked out,” Dr. Heck said. Because of the need for frequent follow-up visits to program the device, RNS may be a difficult treatment option for patients who must travel long distances.
The RNS System Pivotal trial evaluated responder rates at two years. Patients using RNS demonstrated gradual improvement, with median seizure reductions of approximately 53%, compared with baseline. There were no persistent clinically significant neurologic sequelae.
—Adriene Marshall
VANCOUVER—For patients with refractory epilepsy who are not candidates for resective surgery, neuromodulatory devices can be helpful. But which device should be considered for which patient? “Individualize the treatment,” said Christianne Heck, MD, at the 68th Annual Meeting of the American Academy of Neurology. With open- and closed-loop treatments available, factors such as compliance and patient lifestyle are some of the considerations physicians should keep in mind.
Of the approximately 50 million people worldwide with epilepsy, more than one-third have drug-resistant epilepsy (DRE). “DRE is defined as the failure of two or more appropriate antiepileptic drugs to achieve complete seizure control,” said Dr. Heck, Medical Director of the University of Southern California Comprehensive Epilepsy Program at the Keck Medical Center in Los Angeles. “It is associated with major depression, an increased risk in injury and death, cognitive and attention deficits, lower quality of life, and increased healthcare costs.”
Even as new medications have become available, drug resistance rates have remained the same. In addition, adverse effects are quite common and interfere with daily living. “They’re the primary cause of treatment failure in 13% to 40% of patients, depending on the drug,” Dr. Heck noted. “Devices for epilepsy have demonstrated quality-of-life improvements and improvements in mood scores, as well as sustained effectiveness over time.”
Surgery is appropriate for about half of patients with DRE who undergo a presurgical workup in an epilepsy center. The other 50%, however, either have an epileptic zone that is not well defined or an epileptic zone that localizes to eloquent cortex that cannot be resected.
Vagus Nerve Stimulation
Open-loop devices provide automated continuous or nearly continuous stimulation to the area of focus. They control excess electrical activity in the brain by administering regular electrical impulses to reduce the frequency and severity of seizures. Among the open-loop systems is vagus nerve stimulation (VNS), which was approved by the FDA as adjunctive therapy in reducing the frequency of seizures in adults and in adolescents older than 12 with refractory partial onset seizures.
The VNS device is implanted under the skin in the left chest. A horizontal incision in the left neck allows for an electrode from the stimulator to be wrapped around the left vagus nerve. In the Vagus Nerve Stimulation Study Group E01-E05 trials, researchers observed a 43% responder rate—defined as a 50% or greater reduction in seizure frequency—among VNS users at three years.
While VNS is an open-loop system, patients who have auras and can tell when a seizure might occur can pass a special magnet near the implanted device to trigger stimulation outside of the programmed intervals on demand, Dr. Heck said. “However, some patients say they have no way to know when to use the magnet. I try to make it clear to them that magnet activation is a luxury, not a requirement.”
A closed-loop VNS device, AspireSR (Cyberonics), was approved by the FDA in 2015. Closed-loop devices provide stimulation only in response to a trigger. For AspireSR, elevated heart rate triggers stimulation. “The parameters for the change in heart rate can be adjusted based upon the individual’s cardiac changes,” said Dr. Heck. “But it also works in conjunction with its normal cycling, as well as magnet modes.”
One concern about using heart rate as a biomarker for epileptic seizures is that the definition of ictal tachycardia varies widely in the medical literature—from 100 bpm to 140 bpm to a 10-bpm increase. “It’s difficult at this point to determine whether heart rate is the ideal biomarker,” Dr. Heck noted.
The automated nature of VNS therapy lessens concerns about patient compliance, and the treatment has minimal side effects, Dr. Heck said. Patients may, however, experience hoarseness or other voice changes during stimulation—a consideration for those whose voice is an important part of their job, including singers. In addition, patients may become slightly out of breath during stimulation, which could be a concern for athletes. Furthermore, the device is costly and requires surgical implantation, yet there is no way to predict before surgery whether the patient’s seizures will be responsive to neurostimulation therapy.
Deep Brain Stimulation
Deep brain stimulation (DBS) for epilepsy targets the anterior nucleus of the thalamus. DBS for the treatment of epilepsy has not yet been approved in the US. During DBS surgery, two electrodes are placed deep inside the brain through small holes made in the skull. MRI or another imaging technique helps ensure the electrodes are placed correctly. The electrodes are connected to wires that run under the skin behind the ear and down the neck to a neurostimulator that is implanted under the skin in the upper chest, below the collarbone. As with VNS, a patient with auras can activate the device in addition to receiving automated treatment.
Dr. Heck cited a recent trial of DBS that included 110 patients with epilepsy who had experienced at least six seizures per month and failed at least three antiepileptic drugs. During long-term follow-up, the responder rate was 43% at one year and 68% at five years. Sixteen percent of the patients were seizure-free for at least six months, and scores on the Liverpool Seizure Severity Scale improved over baseline. There was an adverse event rate of 34%.
Noninvasive Stimulation
Another open-loop device is the external trigeminal nerve stimulator (eTNS), which is noninvasive and easy to use, Dr. Heck said. “It’s preset with a certain stimulation algorithm and therefore doesn’t require programming. The patient simply adjusts the current to [his or her] comfort level.”
The eTNS unit includes an electrode that is applied to the forehead. Patients can wear the device at night or under a cap during the day, Dr. Heck said. eTNS is approved for prescription use in the EU, Canada, and Australia as adjunctive therapy for epilepsy in patients ages 9 and older.
In a small, 18-week randomized active control trial of 50 patients with DRE who experienced two or more partial onset seizures per month, researchers observed a 40% improvement in the treatment group versus a 16% improvement in the control group. “Although we don’t have any data on this so far, eTNS may be a convenient way to test whether neuromodulation works for patients before they invest in an implantable device that is considerably more expensive,” Dr. Heck said.
Responsive Neurostimulation
Responsive neurostimulation (RNS) devices are placed under the scalp and within the skull. One or two electrodes connected to the neurostimulator are placed at the seizure focus site.
“At this point, I think that the RNS System [NeuroPace] is the gold standard for responsive closed-loop epilepsy therapy. It responds specifically to cortical activity that represents seizure,” Dr. Heck said. “The cranial implant is not visible, which is good for patients who care about scarring. The small stimulation pulses [are] delivered through the leads directly to the brain and those pulses add up to less than six minutes of electrical stimulation per day on average.” The RNS System received premarket approval by the FDA in late 2013 to treat medically refractory partial epilepsy.
Patients do not know when they are receiving stimulation. RNS “provides an alternative for localization-related DRE when surgery is not an option,” she added. RNS also may be appropriate for patients who have multiple seizure onset zones because more than one electrode can be used to cover various sites.
A disadvantage of using RNS is the shorter battery life, compared with VNS (three to four years vs four to 10 years, respectively). In addition, it is a complex device to use. “I began to work with this device in 2006 and I still would not claim to be an absolute expert yet. I think it’s an exciting kind of technology, but the actual parameters for therapy still need to be worked out,” Dr. Heck said. Because of the need for frequent follow-up visits to program the device, RNS may be a difficult treatment option for patients who must travel long distances.
The RNS System Pivotal trial evaluated responder rates at two years. Patients using RNS demonstrated gradual improvement, with median seizure reductions of approximately 53%, compared with baseline. There were no persistent clinically significant neurologic sequelae.
—Adriene Marshall
Suggested Reading
DeGiorgio CM, Soss J, Cook IA, et al. Randomized controlled trial of trigeminal nerve stimulation for drug-resistant epilepsy. Neurology. 2013;80(9):786-791.
Heck CN, King-Stephens D, Massey AD, et al. Two-year seizure reduction in adults with medically intractable partial onset epilepsy treated with responsive neurostimulation: final results of the RNS System Pivotal trial. Epilepsia. 2014;55(3):432-441.
Morris GL 3rd, Mueller WM. Long-term treatment with vagus nerve stimulation in patients with refractory epilepsy. The Vagus Nerve Stimulation Study Group E01-E05. Neurology. 1999;53(8):1731-1735.
Salanova V, Witt T, Worth R, et al; SANTE Study Group. Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology. 2015;84(10):1017-1025.
Suggested Reading
DeGiorgio CM, Soss J, Cook IA, et al. Randomized controlled trial of trigeminal nerve stimulation for drug-resistant epilepsy. Neurology. 2013;80(9):786-791.
Heck CN, King-Stephens D, Massey AD, et al. Two-year seizure reduction in adults with medically intractable partial onset epilepsy treated with responsive neurostimulation: final results of the RNS System Pivotal trial. Epilepsia. 2014;55(3):432-441.
Morris GL 3rd, Mueller WM. Long-term treatment with vagus nerve stimulation in patients with refractory epilepsy. The Vagus Nerve Stimulation Study Group E01-E05. Neurology. 1999;53(8):1731-1735.
Salanova V, Witt T, Worth R, et al; SANTE Study Group. Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology. 2015;84(10):1017-1025.
Screen teens for suicide risk, AAP advises
Screening teens for suicide risk is among the recommendations in a new clinical report from the American Academy of Pediatrics Committee on Adolescence.
Suicide is now the second-leading cause of death for teens aged 15-19 years, having surpassed homicide since 2007, when the last report on adolescent suicide was published by the AAP (Pediatrics. 2016 June 27 doi: 10.1542/peds.2016-1420). Excessive Internet use and cyberbullying are considered to be risk factors for suicide.
In addition to screening for suicide risk, the AAP advises screening for mood disorders, and for substance abuse and dependence. “Ask about emotional difficulties and use of drugs and alcohol, identify lack of developmental progress, and estimate level of distress, impairment of functioning, and level of danger to self and others,” the committee members wrote. The report offers several examples of questions to use for screening during the visit or interview.
“Self-report scales may be helpful, but they do not substitute for pediatricians asking teens directly (and sensitively) about mood disorders, substance use, suicidal and self-harmful thoughts, behaviors, stress, and distress,” Benjamin Shain, MD, PhD, the report’s author and a member of the AAP Committee on Adolescence, said in an interview. “Since ‘black box’ warning labels were added to antidepressants in 2004, new, population-based research has indicated that the benefits of these medications outweigh the risks for many patients. Physicians should not shy away from prescribing these medications when they are clinically indicated.”
Risk factors
A significant risk factor for suicide is involvement in bullying, whether the teen is the victim, the bully, or both, with the latter – bully victims – at the highest risk. The increased risk for suicidal thoughts or attempts extends to involvement with cyberbullying, in addition to physical, social, and verbal bullying, according to the committee members.
Results of a meta-analysis indicate girls who are victims or perpetrators of bullying are at higher risk for suicidal thoughts or attempts regardless of how common or rare the bullying is, whereas boys had an increased risk only when the bullying was frequent. Bullying as early as age 8 years was associated with attempted and completed suicides in adolescence, according to one study.
In a 2013 survey of U.S. high school students, 24% of girls and 16% of boys reported being bullied on school property in the past year. Cyberbullying occurred among 21% of the girls and 9% of the boys.
Teens reporting video game or Internet use for at least 5 hours a day were at higher risk for suicidal thoughts, suicide attempts, and depression, the committee noted. An additional wrinkle of Internet use is that teens may read about others’ suicides, which increases their own risk, or they may seek out pro-suicide websites.
“Suicide-related searches were found to be associated with completed suicides among young adults,” the report noted.
The good news is that major search engines prioritize institutional sites and mental health support sites in response to searches for “suicide,” Dr. Shain said.
“Despite the negatives, there is evidence that the Internet and electronic media, in general, provide a large amount of support, including keeping teens connected to their friends, family, teachers, and others, and also providing entertainment and valuable or interesting information,” Dr. Shain noted. “The Internet may be particularly useful for distressed teens.”
“More than 90% of adolescent suicide victims met criteria for a psychiatric disorder before their death,” the report noted. “Immediate risk factors include agitation, intoxication, and a recent stressful life event.”
Take action to help at-risk teens
The committee advises taking the following steps to assist at-risk teens:
In routine history taking, ask questions about mood disorders, use of drugs and alcohol, suicidal thoughts, bullying, sexual orientation, and other risk factors associated with suicide throughout adolescence.
Develop working relationships with emergency departments and colleagues in child and adolescent psychiatry, clinical psychology, and other mental health professions to optimally evaluate and manage the care of adolescents who are at risk for suicide.
Become familiar with local, state, and national resources that are concerned with treatment of psychopathology and suicide prevention in youth, including local hospitals with psychiatric units, mental health agencies, family and children’s services, crisis hotlines, and crisis intervention centers.
Consider additional training and ongoing education in diagnosing and managing adolescent mood disorders, especially if you practice in an underserved area.
During routine evaluations and where consistent with state law, ask whether firearms are kept in the home and discuss with parents the increased risk of adolescent suicide with the presence of firearms. Specifically for adolescents at risk for suicide, advise parents to remove guns and ammunition from the house and secure supplies of prescription and over-the-counter medications.
Learn about the benefits and risks of antidepressant medications, and how to monitor depressed patients. Educate the family regarding the following warning signs that warrant contacting you: new or more frequent thoughts of wanting to die; self-destructive behavior; signs of increased anxiety/panic, agitation, aggressiveness, impulsivity, insomnia or irritability; new or more involuntary restlessness (akathisia), such as pacing or fidgeting; extreme degree of elation or energy; fast, driven speech; or new onset of unrealistic plans or goals.
Risk factors for suicide
Fixed risk factors
• Family history of suicide or suicide attempts.
• History of adoption.
• Male gender.
• Parental mental health problems.
• Lesbian, gay, bisexual, or questioning sexual orientation.
• Transgender identification.
• A history of physical or sexual abuse.
• Previous suicide attempt.
Social/environmental risk factors
• Bullying.
• Impaired parent-child relationship.
• Living outside of the home (homeless or in a corrections facility or group home).
• Difficulties in school.
• Neither working nor attending school.
• Social isolation.
• Presence of stressful life events, such as legal or romantic difficulties or an argument with a parent.
• An unsupported social environment for LGBTQ adolescents.
Personal mental health problems
• Sleep disturbances.
• Depression.
• Bipolar disorder.
• Substance intoxication and substance use disorders.
• Psychosis.
• Posttraumatic stress disorder.
• Panic attacks.
• A history of aggression.
• Impulsivity.
• Severe anger.
• Pathologic Internet use.
Source: Pediatrics. 2016 June 27 doi: 10.1542/peds.2016-1420.
Screening teens for suicide risk is among the recommendations in a new clinical report from the American Academy of Pediatrics Committee on Adolescence.
Suicide is now the second-leading cause of death for teens aged 15-19 years, having surpassed homicide since 2007, when the last report on adolescent suicide was published by the AAP (Pediatrics. 2016 June 27 doi: 10.1542/peds.2016-1420). Excessive Internet use and cyberbullying are considered to be risk factors for suicide.
In addition to screening for suicide risk, the AAP advises screening for mood disorders, and for substance abuse and dependence. “Ask about emotional difficulties and use of drugs and alcohol, identify lack of developmental progress, and estimate level of distress, impairment of functioning, and level of danger to self and others,” the committee members wrote. The report offers several examples of questions to use for screening during the visit or interview.
“Self-report scales may be helpful, but they do not substitute for pediatricians asking teens directly (and sensitively) about mood disorders, substance use, suicidal and self-harmful thoughts, behaviors, stress, and distress,” Benjamin Shain, MD, PhD, the report’s author and a member of the AAP Committee on Adolescence, said in an interview. “Since ‘black box’ warning labels were added to antidepressants in 2004, new, population-based research has indicated that the benefits of these medications outweigh the risks for many patients. Physicians should not shy away from prescribing these medications when they are clinically indicated.”
Risk factors
A significant risk factor for suicide is involvement in bullying, whether the teen is the victim, the bully, or both, with the latter – bully victims – at the highest risk. The increased risk for suicidal thoughts or attempts extends to involvement with cyberbullying, in addition to physical, social, and verbal bullying, according to the committee members.
Results of a meta-analysis indicate girls who are victims or perpetrators of bullying are at higher risk for suicidal thoughts or attempts regardless of how common or rare the bullying is, whereas boys had an increased risk only when the bullying was frequent. Bullying as early as age 8 years was associated with attempted and completed suicides in adolescence, according to one study.
In a 2013 survey of U.S. high school students, 24% of girls and 16% of boys reported being bullied on school property in the past year. Cyberbullying occurred among 21% of the girls and 9% of the boys.
Teens reporting video game or Internet use for at least 5 hours a day were at higher risk for suicidal thoughts, suicide attempts, and depression, the committee noted. An additional wrinkle of Internet use is that teens may read about others’ suicides, which increases their own risk, or they may seek out pro-suicide websites.
“Suicide-related searches were found to be associated with completed suicides among young adults,” the report noted.
The good news is that major search engines prioritize institutional sites and mental health support sites in response to searches for “suicide,” Dr. Shain said.
“Despite the negatives, there is evidence that the Internet and electronic media, in general, provide a large amount of support, including keeping teens connected to their friends, family, teachers, and others, and also providing entertainment and valuable or interesting information,” Dr. Shain noted. “The Internet may be particularly useful for distressed teens.”
“More than 90% of adolescent suicide victims met criteria for a psychiatric disorder before their death,” the report noted. “Immediate risk factors include agitation, intoxication, and a recent stressful life event.”
Take action to help at-risk teens
The committee advises taking the following steps to assist at-risk teens:
In routine history taking, ask questions about mood disorders, use of drugs and alcohol, suicidal thoughts, bullying, sexual orientation, and other risk factors associated with suicide throughout adolescence.
Develop working relationships with emergency departments and colleagues in child and adolescent psychiatry, clinical psychology, and other mental health professions to optimally evaluate and manage the care of adolescents who are at risk for suicide.
Become familiar with local, state, and national resources that are concerned with treatment of psychopathology and suicide prevention in youth, including local hospitals with psychiatric units, mental health agencies, family and children’s services, crisis hotlines, and crisis intervention centers.
Consider additional training and ongoing education in diagnosing and managing adolescent mood disorders, especially if you practice in an underserved area.
During routine evaluations and where consistent with state law, ask whether firearms are kept in the home and discuss with parents the increased risk of adolescent suicide with the presence of firearms. Specifically for adolescents at risk for suicide, advise parents to remove guns and ammunition from the house and secure supplies of prescription and over-the-counter medications.
Learn about the benefits and risks of antidepressant medications, and how to monitor depressed patients. Educate the family regarding the following warning signs that warrant contacting you: new or more frequent thoughts of wanting to die; self-destructive behavior; signs of increased anxiety/panic, agitation, aggressiveness, impulsivity, insomnia or irritability; new or more involuntary restlessness (akathisia), such as pacing or fidgeting; extreme degree of elation or energy; fast, driven speech; or new onset of unrealistic plans or goals.
Risk factors for suicide
Fixed risk factors
• Family history of suicide or suicide attempts.
• History of adoption.
• Male gender.
• Parental mental health problems.
• Lesbian, gay, bisexual, or questioning sexual orientation.
• Transgender identification.
• A history of physical or sexual abuse.
• Previous suicide attempt.
Social/environmental risk factors
• Bullying.
• Impaired parent-child relationship.
• Living outside of the home (homeless or in a corrections facility or group home).
• Difficulties in school.
• Neither working nor attending school.
• Social isolation.
• Presence of stressful life events, such as legal or romantic difficulties or an argument with a parent.
• An unsupported social environment for LGBTQ adolescents.
Personal mental health problems
• Sleep disturbances.
• Depression.
• Bipolar disorder.
• Substance intoxication and substance use disorders.
• Psychosis.
• Posttraumatic stress disorder.
• Panic attacks.
• A history of aggression.
• Impulsivity.
• Severe anger.
• Pathologic Internet use.
Source: Pediatrics. 2016 June 27 doi: 10.1542/peds.2016-1420.
Screening teens for suicide risk is among the recommendations in a new clinical report from the American Academy of Pediatrics Committee on Adolescence.
Suicide is now the second-leading cause of death for teens aged 15-19 years, having surpassed homicide since 2007, when the last report on adolescent suicide was published by the AAP (Pediatrics. 2016 June 27 doi: 10.1542/peds.2016-1420). Excessive Internet use and cyberbullying are considered to be risk factors for suicide.
In addition to screening for suicide risk, the AAP advises screening for mood disorders, and for substance abuse and dependence. “Ask about emotional difficulties and use of drugs and alcohol, identify lack of developmental progress, and estimate level of distress, impairment of functioning, and level of danger to self and others,” the committee members wrote. The report offers several examples of questions to use for screening during the visit or interview.
“Self-report scales may be helpful, but they do not substitute for pediatricians asking teens directly (and sensitively) about mood disorders, substance use, suicidal and self-harmful thoughts, behaviors, stress, and distress,” Benjamin Shain, MD, PhD, the report’s author and a member of the AAP Committee on Adolescence, said in an interview. “Since ‘black box’ warning labels were added to antidepressants in 2004, new, population-based research has indicated that the benefits of these medications outweigh the risks for many patients. Physicians should not shy away from prescribing these medications when they are clinically indicated.”
Risk factors
A significant risk factor for suicide is involvement in bullying, whether the teen is the victim, the bully, or both, with the latter – bully victims – at the highest risk. The increased risk for suicidal thoughts or attempts extends to involvement with cyberbullying, in addition to physical, social, and verbal bullying, according to the committee members.
Results of a meta-analysis indicate girls who are victims or perpetrators of bullying are at higher risk for suicidal thoughts or attempts regardless of how common or rare the bullying is, whereas boys had an increased risk only when the bullying was frequent. Bullying as early as age 8 years was associated with attempted and completed suicides in adolescence, according to one study.
In a 2013 survey of U.S. high school students, 24% of girls and 16% of boys reported being bullied on school property in the past year. Cyberbullying occurred among 21% of the girls and 9% of the boys.
Teens reporting video game or Internet use for at least 5 hours a day were at higher risk for suicidal thoughts, suicide attempts, and depression, the committee noted. An additional wrinkle of Internet use is that teens may read about others’ suicides, which increases their own risk, or they may seek out pro-suicide websites.
“Suicide-related searches were found to be associated with completed suicides among young adults,” the report noted.
The good news is that major search engines prioritize institutional sites and mental health support sites in response to searches for “suicide,” Dr. Shain said.
“Despite the negatives, there is evidence that the Internet and electronic media, in general, provide a large amount of support, including keeping teens connected to their friends, family, teachers, and others, and also providing entertainment and valuable or interesting information,” Dr. Shain noted. “The Internet may be particularly useful for distressed teens.”
“More than 90% of adolescent suicide victims met criteria for a psychiatric disorder before their death,” the report noted. “Immediate risk factors include agitation, intoxication, and a recent stressful life event.”
Take action to help at-risk teens
The committee advises taking the following steps to assist at-risk teens:
In routine history taking, ask questions about mood disorders, use of drugs and alcohol, suicidal thoughts, bullying, sexual orientation, and other risk factors associated with suicide throughout adolescence.
Develop working relationships with emergency departments and colleagues in child and adolescent psychiatry, clinical psychology, and other mental health professions to optimally evaluate and manage the care of adolescents who are at risk for suicide.
Become familiar with local, state, and national resources that are concerned with treatment of psychopathology and suicide prevention in youth, including local hospitals with psychiatric units, mental health agencies, family and children’s services, crisis hotlines, and crisis intervention centers.
Consider additional training and ongoing education in diagnosing and managing adolescent mood disorders, especially if you practice in an underserved area.
During routine evaluations and where consistent with state law, ask whether firearms are kept in the home and discuss with parents the increased risk of adolescent suicide with the presence of firearms. Specifically for adolescents at risk for suicide, advise parents to remove guns and ammunition from the house and secure supplies of prescription and over-the-counter medications.
Learn about the benefits and risks of antidepressant medications, and how to monitor depressed patients. Educate the family regarding the following warning signs that warrant contacting you: new or more frequent thoughts of wanting to die; self-destructive behavior; signs of increased anxiety/panic, agitation, aggressiveness, impulsivity, insomnia or irritability; new or more involuntary restlessness (akathisia), such as pacing or fidgeting; extreme degree of elation or energy; fast, driven speech; or new onset of unrealistic plans or goals.
Risk factors for suicide
Fixed risk factors
• Family history of suicide or suicide attempts.
• History of adoption.
• Male gender.
• Parental mental health problems.
• Lesbian, gay, bisexual, or questioning sexual orientation.
• Transgender identification.
• A history of physical or sexual abuse.
• Previous suicide attempt.
Social/environmental risk factors
• Bullying.
• Impaired parent-child relationship.
• Living outside of the home (homeless or in a corrections facility or group home).
• Difficulties in school.
• Neither working nor attending school.
• Social isolation.
• Presence of stressful life events, such as legal or romantic difficulties or an argument with a parent.
• An unsupported social environment for LGBTQ adolescents.
Personal mental health problems
• Sleep disturbances.
• Depression.
• Bipolar disorder.
• Substance intoxication and substance use disorders.
• Psychosis.
• Posttraumatic stress disorder.
• Panic attacks.
• A history of aggression.
• Impulsivity.
• Severe anger.
• Pathologic Internet use.
Source: Pediatrics. 2016 June 27 doi: 10.1542/peds.2016-1420.
FROM PEDIATRICS
Rediscovering clozapine
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Novel vaccine scores better hepatitis B seroprotection in type 2 diabetes
NEW ORLEANS – An investigational hepatitis B vaccine known as Heplisav-B provided significantly better seroprotection among adults with type 2 diabetes than did a currently licensed vaccine, based on results from a randomized phase III trial.
A total of 321 patients received three doses of the Food and Drug Administration–approved vaccine Engerix-B at weeks 0, 4, and 24, while 640 patients received two doses of the investigational vaccine Heplisav-B at weeks 0 and 4 and a placebo injection at week 24. In both groups of patients, two-thirds of subjects had diabetes for 5 or more years, Randall N. Hyer, MD, reported in a poster at the annual scientific sessions of the American Diabetes Association.
At 28 weeks, 90% of subjects in the Heplisav-B group and 65% in the Engerix-B group achieved seroprotection, defined as anti–hepatitis B titers of 10 mIU/mL or greater. The difference reached statistical significance. Among study participants aged 60-70 years, 85.8% of subjects in the Heplisav-B group achieved seroprotection, compared with 58.5% in the Engerix-B group. Among study participants with a body mass index of 30 kg/m2, 89.5% of subjects in the Heplisav-B group achieved seroprotection, compared with 61.4% in the Engerix-B group, reported Dr. Hyer, who is vice president of medical affairs for Dynavax, the maker of Heplisav-B.
The safety profile, including the incidence of immune-mediated adverse events, was similar in both treatment groups. The most frequently reported local reaction was injection-site pain, while the most common systemic reactions were fatigue, headache, and malaise.
Dynavax submitted a biologics license application to the FDA in March 2016, and a decision on whether to approve Heplisav-B is expected by mid-December. “The Centers for Disease Control and Prevention does recommend that all adults with diabetes aged 19-59 be vaccinated against HBV [hepatitis B virus] as soon as feasible after diagnosis. For folks 60 and above, it’s at the discretion of the physician. People with type 2 diabetes are twice as likely to get HBV,” Dr. Hyer said in an interview.
Dynavax funded the study. Dr. Hyer is an employee of the company.
NEW ORLEANS – An investigational hepatitis B vaccine known as Heplisav-B provided significantly better seroprotection among adults with type 2 diabetes than did a currently licensed vaccine, based on results from a randomized phase III trial.
A total of 321 patients received three doses of the Food and Drug Administration–approved vaccine Engerix-B at weeks 0, 4, and 24, while 640 patients received two doses of the investigational vaccine Heplisav-B at weeks 0 and 4 and a placebo injection at week 24. In both groups of patients, two-thirds of subjects had diabetes for 5 or more years, Randall N. Hyer, MD, reported in a poster at the annual scientific sessions of the American Diabetes Association.
At 28 weeks, 90% of subjects in the Heplisav-B group and 65% in the Engerix-B group achieved seroprotection, defined as anti–hepatitis B titers of 10 mIU/mL or greater. The difference reached statistical significance. Among study participants aged 60-70 years, 85.8% of subjects in the Heplisav-B group achieved seroprotection, compared with 58.5% in the Engerix-B group. Among study participants with a body mass index of 30 kg/m2, 89.5% of subjects in the Heplisav-B group achieved seroprotection, compared with 61.4% in the Engerix-B group, reported Dr. Hyer, who is vice president of medical affairs for Dynavax, the maker of Heplisav-B.
The safety profile, including the incidence of immune-mediated adverse events, was similar in both treatment groups. The most frequently reported local reaction was injection-site pain, while the most common systemic reactions were fatigue, headache, and malaise.
Dynavax submitted a biologics license application to the FDA in March 2016, and a decision on whether to approve Heplisav-B is expected by mid-December. “The Centers for Disease Control and Prevention does recommend that all adults with diabetes aged 19-59 be vaccinated against HBV [hepatitis B virus] as soon as feasible after diagnosis. For folks 60 and above, it’s at the discretion of the physician. People with type 2 diabetes are twice as likely to get HBV,” Dr. Hyer said in an interview.
Dynavax funded the study. Dr. Hyer is an employee of the company.
NEW ORLEANS – An investigational hepatitis B vaccine known as Heplisav-B provided significantly better seroprotection among adults with type 2 diabetes than did a currently licensed vaccine, based on results from a randomized phase III trial.
A total of 321 patients received three doses of the Food and Drug Administration–approved vaccine Engerix-B at weeks 0, 4, and 24, while 640 patients received two doses of the investigational vaccine Heplisav-B at weeks 0 and 4 and a placebo injection at week 24. In both groups of patients, two-thirds of subjects had diabetes for 5 or more years, Randall N. Hyer, MD, reported in a poster at the annual scientific sessions of the American Diabetes Association.
At 28 weeks, 90% of subjects in the Heplisav-B group and 65% in the Engerix-B group achieved seroprotection, defined as anti–hepatitis B titers of 10 mIU/mL or greater. The difference reached statistical significance. Among study participants aged 60-70 years, 85.8% of subjects in the Heplisav-B group achieved seroprotection, compared with 58.5% in the Engerix-B group. Among study participants with a body mass index of 30 kg/m2, 89.5% of subjects in the Heplisav-B group achieved seroprotection, compared with 61.4% in the Engerix-B group, reported Dr. Hyer, who is vice president of medical affairs for Dynavax, the maker of Heplisav-B.
The safety profile, including the incidence of immune-mediated adverse events, was similar in both treatment groups. The most frequently reported local reaction was injection-site pain, while the most common systemic reactions were fatigue, headache, and malaise.
Dynavax submitted a biologics license application to the FDA in March 2016, and a decision on whether to approve Heplisav-B is expected by mid-December. “The Centers for Disease Control and Prevention does recommend that all adults with diabetes aged 19-59 be vaccinated against HBV [hepatitis B virus] as soon as feasible after diagnosis. For folks 60 and above, it’s at the discretion of the physician. People with type 2 diabetes are twice as likely to get HBV,” Dr. Hyer said in an interview.
Dynavax funded the study. Dr. Hyer is an employee of the company.
AT THE ADA ANNUAL SCIENTIFIC SESSIONS
Key clinical point: Compared with Engerix-B, Heplisav-B protected more subjects with diabetes against hepatitis B.
Major finding: At 28 weeks, 90% of the Heplisav-B group and 65% of the Engerix-B group had anti–hepatitis B titers of 10 mIU/mL or greater.
Data source: A phase III randomized trial in which 321 patients received three doses of Engerix-B at weeks 0, 4, and 24, while 640 patients received two doses of Heplisav-B at weeks 0 and 4 (plus placebo at week 24).
Disclosures: Dynavax funded the study. Dr. Hyer is an employee of the company.
Blisters on toes
A biopsy was performed and direct immunofluorescence came back negative. This, along with the patient’s history of diabetes, led us to diagnose bullosis diabeticorum.
This condition, also known as bullous disease of diabetes, is characterized by abrupt development of noninflammatory bullae on acral areas in patients with diabetes. The skin appears normal except for the bullae. Bullosis diabeticorum occurs in just .5% of patients with diabetes and is twice as common in men as it is in women.
The etiology of bullosis diabeticorum is unknown. The acral location suggests that trauma may be a contributing factor. Although electron microscopy has suggested an abnormality in anchoring fibrils, this cellular change does not fully explain the development of multiple blisters at varying sites. Glycemic control is not thought to play a role.
The distribution of lesions and the presence—or absence—of systemic symptoms goes a long way toward narrowing the differential of blistering diseases. The presence of generalized blistering and systemic symptoms would suggest conditions related to medication exposure, such as Stevens-Johnson syndrome or toxic epidermal necrolysis; infectious etiologies (eg, staphylococcal scalded skin syndrome); autoimmune causes; or underlying malignancy. Generalized blistering in the absence of systemic symptoms would support diagnoses such as bullous impetigo and pemphigoid.
Lesion distribution provides important clues, too. Sun exposure-related causes typically leave lesions on the hands and forearms, not just the toes. A dermatomal distribution would suggest herpes zoster. A linear distribution of blisters argues for contact dermatitis.
A diagnosis of bullosis diabeticorum can be made when biopsy with immunofluorescence excludes other histologically similar entities such as epidermolysis bullosa, noninflammatory bullous pemphigoid, and porphyria cutanea tarda. And while immunofluorescence findings are typically negative, elevated levels of immunoglobulin M and C3 have, on occasion, been reported. Cultures are warranted only if a secondary infection is suspected.
The bullae of this condition spontaneously resolve over several weeks without treatment, but tend to recur. The lesions typically heal without significant scarring, although they may have a darker pigmentation after the first occurrence. Treatment may be warranted if a patient develops a secondary infection.
In our patient’s case, the bullae resolved within 2 weeks without treatment, although mild hyperpigmentation remained.
Adapted from: Mims L, Savage A, Chessman A. Blisters on an elderly woman's toes. J Fam Pract. 2014;63:273-274.
A biopsy was performed and direct immunofluorescence came back negative. This, along with the patient’s history of diabetes, led us to diagnose bullosis diabeticorum.
This condition, also known as bullous disease of diabetes, is characterized by abrupt development of noninflammatory bullae on acral areas in patients with diabetes. The skin appears normal except for the bullae. Bullosis diabeticorum occurs in just .5% of patients with diabetes and is twice as common in men as it is in women.
The etiology of bullosis diabeticorum is unknown. The acral location suggests that trauma may be a contributing factor. Although electron microscopy has suggested an abnormality in anchoring fibrils, this cellular change does not fully explain the development of multiple blisters at varying sites. Glycemic control is not thought to play a role.
The distribution of lesions and the presence—or absence—of systemic symptoms goes a long way toward narrowing the differential of blistering diseases. The presence of generalized blistering and systemic symptoms would suggest conditions related to medication exposure, such as Stevens-Johnson syndrome or toxic epidermal necrolysis; infectious etiologies (eg, staphylococcal scalded skin syndrome); autoimmune causes; or underlying malignancy. Generalized blistering in the absence of systemic symptoms would support diagnoses such as bullous impetigo and pemphigoid.
Lesion distribution provides important clues, too. Sun exposure-related causes typically leave lesions on the hands and forearms, not just the toes. A dermatomal distribution would suggest herpes zoster. A linear distribution of blisters argues for contact dermatitis.
A diagnosis of bullosis diabeticorum can be made when biopsy with immunofluorescence excludes other histologically similar entities such as epidermolysis bullosa, noninflammatory bullous pemphigoid, and porphyria cutanea tarda. And while immunofluorescence findings are typically negative, elevated levels of immunoglobulin M and C3 have, on occasion, been reported. Cultures are warranted only if a secondary infection is suspected.
The bullae of this condition spontaneously resolve over several weeks without treatment, but tend to recur. The lesions typically heal without significant scarring, although they may have a darker pigmentation after the first occurrence. Treatment may be warranted if a patient develops a secondary infection.
In our patient’s case, the bullae resolved within 2 weeks without treatment, although mild hyperpigmentation remained.
Adapted from: Mims L, Savage A, Chessman A. Blisters on an elderly woman's toes. J Fam Pract. 2014;63:273-274.
A biopsy was performed and direct immunofluorescence came back negative. This, along with the patient’s history of diabetes, led us to diagnose bullosis diabeticorum.
This condition, also known as bullous disease of diabetes, is characterized by abrupt development of noninflammatory bullae on acral areas in patients with diabetes. The skin appears normal except for the bullae. Bullosis diabeticorum occurs in just .5% of patients with diabetes and is twice as common in men as it is in women.
The etiology of bullosis diabeticorum is unknown. The acral location suggests that trauma may be a contributing factor. Although electron microscopy has suggested an abnormality in anchoring fibrils, this cellular change does not fully explain the development of multiple blisters at varying sites. Glycemic control is not thought to play a role.
The distribution of lesions and the presence—or absence—of systemic symptoms goes a long way toward narrowing the differential of blistering diseases. The presence of generalized blistering and systemic symptoms would suggest conditions related to medication exposure, such as Stevens-Johnson syndrome or toxic epidermal necrolysis; infectious etiologies (eg, staphylococcal scalded skin syndrome); autoimmune causes; or underlying malignancy. Generalized blistering in the absence of systemic symptoms would support diagnoses such as bullous impetigo and pemphigoid.
Lesion distribution provides important clues, too. Sun exposure-related causes typically leave lesions on the hands and forearms, not just the toes. A dermatomal distribution would suggest herpes zoster. A linear distribution of blisters argues for contact dermatitis.
A diagnosis of bullosis diabeticorum can be made when biopsy with immunofluorescence excludes other histologically similar entities such as epidermolysis bullosa, noninflammatory bullous pemphigoid, and porphyria cutanea tarda. And while immunofluorescence findings are typically negative, elevated levels of immunoglobulin M and C3 have, on occasion, been reported. Cultures are warranted only if a secondary infection is suspected.
The bullae of this condition spontaneously resolve over several weeks without treatment, but tend to recur. The lesions typically heal without significant scarring, although they may have a darker pigmentation after the first occurrence. Treatment may be warranted if a patient develops a secondary infection.
In our patient’s case, the bullae resolved within 2 weeks without treatment, although mild hyperpigmentation remained.
Adapted from: Mims L, Savage A, Chessman A. Blisters on an elderly woman's toes. J Fam Pract. 2014;63:273-274.
Ketamine for treatment-resistant depression
Study: CMV doesn’t lower risk of relapse, death

Small studies have suggested that early cytomegalovirus (CMV) reactivation may protect against leukemia relapse and even death after hematopoietic stem cell transplant.
However, a new study, based on data from about 9500 patients, suggests otherwise.
Results showed no association between CMV reactivation and relapse but suggested CMV reactivation increases the risk of non-relapse mortality.
Researchers reported these findings in Blood.
“The original purpose of the study was to confirm that CMV infection may prevent leukemia relapse, prevent death, and become a major therapeutic tool for improving patient survival rates,” said study author Pierre Teira, MD, of the University of Montreal in Quebec, Canada.
“However, we found the exact opposite. Our results clearly show that . . . the virus not only does not prevent leukemia relapse [it] also remains a major factor associated with the risk of death. Monitoring of CMV after transplantation remains a priority for patients.”
For this study, Dr Teira and his colleagues analyzed data from 9469 patients who received a transplant between 2003 and 2010.
The patients had acute myeloid leukemia (AML, n=5310), acute lymphoblastic leukemia (ALL, n=1883), chronic myeloid leukemia (CML, n=1079), or myelodysplastic syndromes (MDS, n=1197).
The median time to initial CMV reactivation was 41 days (range, 1-362 days).
The researchers found no significant association between CMV reactivation and disease relapse for AML (P=0.60), ALL (P=0.08), CML (P=0.94), or MDS (P=0.58).
However, CMV reactivation was associated with a significantly higher risk of nonrelapse mortality for AML (P<0.0001), ALL (P<0.0001), CML (P=0.0004), and MDS (P=0.0002).
Therefore, CMV reactivation was associated with significantly lower overall survival for AML (P<0.0001), ALL (P<0.0001), CML (P=0.0005), and MDS (P=0.003).
“Deaths due to uncontrolled CMV reactivation are virtually zero in this study, so uncontrolled CMV reactivation is not what reduces survival rates after transplantation,” Dr Teira noted. “The link between this common virus and increased risk of death remains a biological mystery.”
One possible explanation is that CMV decreases the ability of the patient’s immune system to fight against other types of infection. This is supported by the fact that death rates from infections other than CMV are higher in patients infected with CMV or patients whose donors were.
For researchers, the next step is therefore to verify whether the latest generation of anti-CMV treatments can prevent both reactivation of the virus and weakening of the patient’s immune system against other types of infection in the presence of CMV infection.
“CMV has a complex impact on the outcomes for transplant patients, and, each year, more than 30,000 patients around the world receive bone marrow transplants from donors,” Dr Teira said.
“It is therefore essential for future research to better understand the role played by CMV after bone marrow transplantation and improve the chances of success of the transplant. This will help to better choose the right donor for the right patient.” ![]()

Small studies have suggested that early cytomegalovirus (CMV) reactivation may protect against leukemia relapse and even death after hematopoietic stem cell transplant.
However, a new study, based on data from about 9500 patients, suggests otherwise.
Results showed no association between CMV reactivation and relapse but suggested CMV reactivation increases the risk of non-relapse mortality.
Researchers reported these findings in Blood.
“The original purpose of the study was to confirm that CMV infection may prevent leukemia relapse, prevent death, and become a major therapeutic tool for improving patient survival rates,” said study author Pierre Teira, MD, of the University of Montreal in Quebec, Canada.
“However, we found the exact opposite. Our results clearly show that . . . the virus not only does not prevent leukemia relapse [it] also remains a major factor associated with the risk of death. Monitoring of CMV after transplantation remains a priority for patients.”
For this study, Dr Teira and his colleagues analyzed data from 9469 patients who received a transplant between 2003 and 2010.
The patients had acute myeloid leukemia (AML, n=5310), acute lymphoblastic leukemia (ALL, n=1883), chronic myeloid leukemia (CML, n=1079), or myelodysplastic syndromes (MDS, n=1197).
The median time to initial CMV reactivation was 41 days (range, 1-362 days).
The researchers found no significant association between CMV reactivation and disease relapse for AML (P=0.60), ALL (P=0.08), CML (P=0.94), or MDS (P=0.58).
However, CMV reactivation was associated with a significantly higher risk of nonrelapse mortality for AML (P<0.0001), ALL (P<0.0001), CML (P=0.0004), and MDS (P=0.0002).
Therefore, CMV reactivation was associated with significantly lower overall survival for AML (P<0.0001), ALL (P<0.0001), CML (P=0.0005), and MDS (P=0.003).
“Deaths due to uncontrolled CMV reactivation are virtually zero in this study, so uncontrolled CMV reactivation is not what reduces survival rates after transplantation,” Dr Teira noted. “The link between this common virus and increased risk of death remains a biological mystery.”
One possible explanation is that CMV decreases the ability of the patient’s immune system to fight against other types of infection. This is supported by the fact that death rates from infections other than CMV are higher in patients infected with CMV or patients whose donors were.
For researchers, the next step is therefore to verify whether the latest generation of anti-CMV treatments can prevent both reactivation of the virus and weakening of the patient’s immune system against other types of infection in the presence of CMV infection.
“CMV has a complex impact on the outcomes for transplant patients, and, each year, more than 30,000 patients around the world receive bone marrow transplants from donors,” Dr Teira said.
“It is therefore essential for future research to better understand the role played by CMV after bone marrow transplantation and improve the chances of success of the transplant. This will help to better choose the right donor for the right patient.” ![]()

Small studies have suggested that early cytomegalovirus (CMV) reactivation may protect against leukemia relapse and even death after hematopoietic stem cell transplant.
However, a new study, based on data from about 9500 patients, suggests otherwise.
Results showed no association between CMV reactivation and relapse but suggested CMV reactivation increases the risk of non-relapse mortality.
Researchers reported these findings in Blood.
“The original purpose of the study was to confirm that CMV infection may prevent leukemia relapse, prevent death, and become a major therapeutic tool for improving patient survival rates,” said study author Pierre Teira, MD, of the University of Montreal in Quebec, Canada.
“However, we found the exact opposite. Our results clearly show that . . . the virus not only does not prevent leukemia relapse [it] also remains a major factor associated with the risk of death. Monitoring of CMV after transplantation remains a priority for patients.”
For this study, Dr Teira and his colleagues analyzed data from 9469 patients who received a transplant between 2003 and 2010.
The patients had acute myeloid leukemia (AML, n=5310), acute lymphoblastic leukemia (ALL, n=1883), chronic myeloid leukemia (CML, n=1079), or myelodysplastic syndromes (MDS, n=1197).
The median time to initial CMV reactivation was 41 days (range, 1-362 days).
The researchers found no significant association between CMV reactivation and disease relapse for AML (P=0.60), ALL (P=0.08), CML (P=0.94), or MDS (P=0.58).
However, CMV reactivation was associated with a significantly higher risk of nonrelapse mortality for AML (P<0.0001), ALL (P<0.0001), CML (P=0.0004), and MDS (P=0.0002).
Therefore, CMV reactivation was associated with significantly lower overall survival for AML (P<0.0001), ALL (P<0.0001), CML (P=0.0005), and MDS (P=0.003).
“Deaths due to uncontrolled CMV reactivation are virtually zero in this study, so uncontrolled CMV reactivation is not what reduces survival rates after transplantation,” Dr Teira noted. “The link between this common virus and increased risk of death remains a biological mystery.”
One possible explanation is that CMV decreases the ability of the patient’s immune system to fight against other types of infection. This is supported by the fact that death rates from infections other than CMV are higher in patients infected with CMV or patients whose donors were.
For researchers, the next step is therefore to verify whether the latest generation of anti-CMV treatments can prevent both reactivation of the virus and weakening of the patient’s immune system against other types of infection in the presence of CMV infection.
“CMV has a complex impact on the outcomes for transplant patients, and, each year, more than 30,000 patients around the world receive bone marrow transplants from donors,” Dr Teira said.
“It is therefore essential for future research to better understand the role played by CMV after bone marrow transplantation and improve the chances of success of the transplant. This will help to better choose the right donor for the right patient.” ![]()