User login
Treating cryptococcal meningitis in patients with HIV
One-week treatment with amphotericin B deoxycholate (AmBd)– and flucytosine (5FC)–based therapy, followed by fluconazole (FLU) on days 8 through 14, is probably superior to other regimens for treatment of HIV-associated cryptococcal meningitis, according to the authors of a review of the available literature in the Cochrane Database of Systemic Reviews.
The review is an update of one previous previously published in 2011. The authors found 13 eligible studies that enrolled 2,426 participants and compared 21 interventions. They performed a network meta-analysis using multivariate meta-regression, modeled treatment differences (RR and 95% confidence interval), and determined treatment rankings for 2-week and 10-week mortality outcomes using surface under the cumulative ranking curve, which represents the probability that a treatment will present the best outcome with no uncertainty and was used to develop a hierarchy of treatments for HIV-associated cryptococcal meningitis.
In addition, certainty of the evidence was assessed using the GRADE approach, according to Mark W. Tenforde, MD, of the University of Washington School of Public Health, Seattle, and his colleagues.
They found “reduced 10-week mortality with shortened [AmBd and 5FC] induction therapy, compared to the current gold standard of 2 weeks of AmBd and 5FC, based on moderate-certainty evidence.” They also found no mortality benefit of combination 2 weeks AmBd and FLU, compared with AmBd alone.
“In resource-limited settings, 1-week AmBd- and 5FC-based therapy is probably superior to other regimens for treatment of HIV-associated cryptococcal meningitis,” they wrote. “An all-oral regimen of 2 weeks 5FC and FLU may be an alternative in settings where AmBd is unavailable or intravenous therapy cannot be safely administered.” These results indicated the need to expand access to 5FC in resource-limited settings in which HIV-associated cryptococcal meningitis is most common.
They also reported finding no mortality benefit of 2 weeks of combination AmBd and FLU, compared with AmBd alone.
“Given the absence of data from studies in children, and limited data from high-income countries, our findings provide limited guidance for treatment in these patients and settings,” Dr. Tenforde and his colleagues stated.
The authors reported that they had no relevant conflicts of interest.
SOURCE: Tenforde MW et al. Treatment for HIV-associated cryptococcal meningitis. Cochrane Database Syst Rev. 2018 Jul 25;7:CD005647. doi: 10.1002/14651858.CD005647.
One-week treatment with amphotericin B deoxycholate (AmBd)– and flucytosine (5FC)–based therapy, followed by fluconazole (FLU) on days 8 through 14, is probably superior to other regimens for treatment of HIV-associated cryptococcal meningitis, according to the authors of a review of the available literature in the Cochrane Database of Systemic Reviews.
The review is an update of one previous previously published in 2011. The authors found 13 eligible studies that enrolled 2,426 participants and compared 21 interventions. They performed a network meta-analysis using multivariate meta-regression, modeled treatment differences (RR and 95% confidence interval), and determined treatment rankings for 2-week and 10-week mortality outcomes using surface under the cumulative ranking curve, which represents the probability that a treatment will present the best outcome with no uncertainty and was used to develop a hierarchy of treatments for HIV-associated cryptococcal meningitis.
In addition, certainty of the evidence was assessed using the GRADE approach, according to Mark W. Tenforde, MD, of the University of Washington School of Public Health, Seattle, and his colleagues.
They found “reduced 10-week mortality with shortened [AmBd and 5FC] induction therapy, compared to the current gold standard of 2 weeks of AmBd and 5FC, based on moderate-certainty evidence.” They also found no mortality benefit of combination 2 weeks AmBd and FLU, compared with AmBd alone.
“In resource-limited settings, 1-week AmBd- and 5FC-based therapy is probably superior to other regimens for treatment of HIV-associated cryptococcal meningitis,” they wrote. “An all-oral regimen of 2 weeks 5FC and FLU may be an alternative in settings where AmBd is unavailable or intravenous therapy cannot be safely administered.” These results indicated the need to expand access to 5FC in resource-limited settings in which HIV-associated cryptococcal meningitis is most common.
They also reported finding no mortality benefit of 2 weeks of combination AmBd and FLU, compared with AmBd alone.
“Given the absence of data from studies in children, and limited data from high-income countries, our findings provide limited guidance for treatment in these patients and settings,” Dr. Tenforde and his colleagues stated.
The authors reported that they had no relevant conflicts of interest.
SOURCE: Tenforde MW et al. Treatment for HIV-associated cryptococcal meningitis. Cochrane Database Syst Rev. 2018 Jul 25;7:CD005647. doi: 10.1002/14651858.CD005647.
One-week treatment with amphotericin B deoxycholate (AmBd)– and flucytosine (5FC)–based therapy, followed by fluconazole (FLU) on days 8 through 14, is probably superior to other regimens for treatment of HIV-associated cryptococcal meningitis, according to the authors of a review of the available literature in the Cochrane Database of Systemic Reviews.
The review is an update of one previous previously published in 2011. The authors found 13 eligible studies that enrolled 2,426 participants and compared 21 interventions. They performed a network meta-analysis using multivariate meta-regression, modeled treatment differences (RR and 95% confidence interval), and determined treatment rankings for 2-week and 10-week mortality outcomes using surface under the cumulative ranking curve, which represents the probability that a treatment will present the best outcome with no uncertainty and was used to develop a hierarchy of treatments for HIV-associated cryptococcal meningitis.
In addition, certainty of the evidence was assessed using the GRADE approach, according to Mark W. Tenforde, MD, of the University of Washington School of Public Health, Seattle, and his colleagues.
They found “reduced 10-week mortality with shortened [AmBd and 5FC] induction therapy, compared to the current gold standard of 2 weeks of AmBd and 5FC, based on moderate-certainty evidence.” They also found no mortality benefit of combination 2 weeks AmBd and FLU, compared with AmBd alone.
“In resource-limited settings, 1-week AmBd- and 5FC-based therapy is probably superior to other regimens for treatment of HIV-associated cryptococcal meningitis,” they wrote. “An all-oral regimen of 2 weeks 5FC and FLU may be an alternative in settings where AmBd is unavailable or intravenous therapy cannot be safely administered.” These results indicated the need to expand access to 5FC in resource-limited settings in which HIV-associated cryptococcal meningitis is most common.
They also reported finding no mortality benefit of 2 weeks of combination AmBd and FLU, compared with AmBd alone.
“Given the absence of data from studies in children, and limited data from high-income countries, our findings provide limited guidance for treatment in these patients and settings,” Dr. Tenforde and his colleagues stated.
The authors reported that they had no relevant conflicts of interest.
SOURCE: Tenforde MW et al. Treatment for HIV-associated cryptococcal meningitis. Cochrane Database Syst Rev. 2018 Jul 25;7:CD005647. doi: 10.1002/14651858.CD005647.
FROM COCHRANE DATABASE OF SYSTEMIC REVIEWS
Key clinical point: Shorter drug treatment beat the gold standard for HIV-associated cryptococcal meningitis according to a literature review.
Major finding:
Study details: Updated review of articles, registries, and clinical trials during Jan. 1, 1980–July 9, 2018.
Disclosures: The authors reported that they had no relevant conflicts of interest.
Source: Tenforde MW et al. Treatment for HIV-associated cryptococcal meningitis. Cochrane Database Syst Rev. 2018 Jul 25;7:CD005647. doi: 10.1002/14651858.CD005647.
Relapsing-remitting MS best treated within 6 months of onset
BERLIN – according to real-world data from the Big Multiple Sclerosis Data Network.
Receiving disease-modifying treatments (DMTs) within 6 months of diagnosis was associated with a 28% reduction in the risk of reaching an Expanded Disability Status Scale score of 3.0 or more for the first time at 12 months versus receiving treatment after 6 months (hazard ratio, 0.72; 95% confidence interval, 0.59-0.90; P = .003).
Results were not significant, looking at all the other periods tested at 6-month intervals from 1 year up to 5 years after diagnosis. HRs (95% CIs) comparing a first DMT given at 1 year, 1.5 years, 2 years, 2.5 years, 3 years, 3.5 years, 4 years, 4.5 years, and 5 years were a respective 0.90 (0.78-1.03), 0.89 (0.79-1.01), 0.99 (0.88-1.11), 0.95 (0.85-1.06), 1.01 (0.90-1.12), 0.97 (0.86-1.09), 1.09 (0.96-1.22), 1.11 (0.98-1.25), and 1.06 (0.93-1.20).
“To date, these data represent the largest RRMS cohort with the longest follow-up ever analyzed to determine the long-term effectiveness of the early start of DMTs,” said Pietro Iaffaldano, MD, at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis.
“This study also provides evidence that data sharing from MS registries and databases is feasible,” noted Dr. Iaffaldano, who is assistant professor of neurology at the University of Bari (Italy). Such an approach can provide enough statistical power to detect the impact of treatment on disability outcomes in the long term, he suggested.
For the study, a cohort of 11,934 patients was obtained by screening more than 149,636 patients from five large registries and databases of MS patients – the Italian MS Registry, the Swedish MS Registry, the Danish MS Registry, OFSEP (Observatoire Français de al Sclérose en Plaques), and MSBase. Patients were included in the current analysis if they had at least 10 years of follow-up, had at least three EDSS evaluations, and at least one DMT prescription.
“It is well known that randomized, controlled trials support the early start of treatment in MS, but open-label extensions of the same trials reported inconsistent results about the long-term benefit on disability accumulation,” Dr. Iaffaldano explained. Further, recent observational studies have suggested that initiating DMTs early might not only delay the accumulation of disability but perhaps also death.
The aim of the research was thus to look at what effect the time interval from disease onset to the first administration of a DMT might have on long-term disability accumulation, as measured by the EDSS, in patients with RRMS.
The population of patients studied was mostly (71%) female, with a median age of 27 years at disease onset. The number of relapses prior to starting a DMT was two and the baseline EDSS was 2.0. In almost all (98.9%) cases, DMT was used as first-line treatment (second line in 1.1% of cases). The median follow-up was 13.2 years and cumulative DMT exposure was 10.5 years.
The work was supported by Biogen International on the basis of a sponsored research agreement with the Big Multiple Sclerosis Data Network. Dr. Iaffaldano has served on scientific advisory boards for and received funding for travel and/or speaker honoraria from Biogen and other companies that market DMTs for MS. Several study authors are employees of Biogen, and other study authors also reported financial ties to Biogen and other pharmaceutical companies.
SOURCE: Iaffaldano P et al. Mult Scler. 2018;24(Suppl 2):71-2, Abstract 204.
BERLIN – according to real-world data from the Big Multiple Sclerosis Data Network.
Receiving disease-modifying treatments (DMTs) within 6 months of diagnosis was associated with a 28% reduction in the risk of reaching an Expanded Disability Status Scale score of 3.0 or more for the first time at 12 months versus receiving treatment after 6 months (hazard ratio, 0.72; 95% confidence interval, 0.59-0.90; P = .003).
Results were not significant, looking at all the other periods tested at 6-month intervals from 1 year up to 5 years after diagnosis. HRs (95% CIs) comparing a first DMT given at 1 year, 1.5 years, 2 years, 2.5 years, 3 years, 3.5 years, 4 years, 4.5 years, and 5 years were a respective 0.90 (0.78-1.03), 0.89 (0.79-1.01), 0.99 (0.88-1.11), 0.95 (0.85-1.06), 1.01 (0.90-1.12), 0.97 (0.86-1.09), 1.09 (0.96-1.22), 1.11 (0.98-1.25), and 1.06 (0.93-1.20).
“To date, these data represent the largest RRMS cohort with the longest follow-up ever analyzed to determine the long-term effectiveness of the early start of DMTs,” said Pietro Iaffaldano, MD, at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis.
“This study also provides evidence that data sharing from MS registries and databases is feasible,” noted Dr. Iaffaldano, who is assistant professor of neurology at the University of Bari (Italy). Such an approach can provide enough statistical power to detect the impact of treatment on disability outcomes in the long term, he suggested.
For the study, a cohort of 11,934 patients was obtained by screening more than 149,636 patients from five large registries and databases of MS patients – the Italian MS Registry, the Swedish MS Registry, the Danish MS Registry, OFSEP (Observatoire Français de al Sclérose en Plaques), and MSBase. Patients were included in the current analysis if they had at least 10 years of follow-up, had at least three EDSS evaluations, and at least one DMT prescription.
“It is well known that randomized, controlled trials support the early start of treatment in MS, but open-label extensions of the same trials reported inconsistent results about the long-term benefit on disability accumulation,” Dr. Iaffaldano explained. Further, recent observational studies have suggested that initiating DMTs early might not only delay the accumulation of disability but perhaps also death.
The aim of the research was thus to look at what effect the time interval from disease onset to the first administration of a DMT might have on long-term disability accumulation, as measured by the EDSS, in patients with RRMS.
The population of patients studied was mostly (71%) female, with a median age of 27 years at disease onset. The number of relapses prior to starting a DMT was two and the baseline EDSS was 2.0. In almost all (98.9%) cases, DMT was used as first-line treatment (second line in 1.1% of cases). The median follow-up was 13.2 years and cumulative DMT exposure was 10.5 years.
The work was supported by Biogen International on the basis of a sponsored research agreement with the Big Multiple Sclerosis Data Network. Dr. Iaffaldano has served on scientific advisory boards for and received funding for travel and/or speaker honoraria from Biogen and other companies that market DMTs for MS. Several study authors are employees of Biogen, and other study authors also reported financial ties to Biogen and other pharmaceutical companies.
SOURCE: Iaffaldano P et al. Mult Scler. 2018;24(Suppl 2):71-2, Abstract 204.
BERLIN – according to real-world data from the Big Multiple Sclerosis Data Network.
Receiving disease-modifying treatments (DMTs) within 6 months of diagnosis was associated with a 28% reduction in the risk of reaching an Expanded Disability Status Scale score of 3.0 or more for the first time at 12 months versus receiving treatment after 6 months (hazard ratio, 0.72; 95% confidence interval, 0.59-0.90; P = .003).
Results were not significant, looking at all the other periods tested at 6-month intervals from 1 year up to 5 years after diagnosis. HRs (95% CIs) comparing a first DMT given at 1 year, 1.5 years, 2 years, 2.5 years, 3 years, 3.5 years, 4 years, 4.5 years, and 5 years were a respective 0.90 (0.78-1.03), 0.89 (0.79-1.01), 0.99 (0.88-1.11), 0.95 (0.85-1.06), 1.01 (0.90-1.12), 0.97 (0.86-1.09), 1.09 (0.96-1.22), 1.11 (0.98-1.25), and 1.06 (0.93-1.20).
“To date, these data represent the largest RRMS cohort with the longest follow-up ever analyzed to determine the long-term effectiveness of the early start of DMTs,” said Pietro Iaffaldano, MD, at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis.
“This study also provides evidence that data sharing from MS registries and databases is feasible,” noted Dr. Iaffaldano, who is assistant professor of neurology at the University of Bari (Italy). Such an approach can provide enough statistical power to detect the impact of treatment on disability outcomes in the long term, he suggested.
For the study, a cohort of 11,934 patients was obtained by screening more than 149,636 patients from five large registries and databases of MS patients – the Italian MS Registry, the Swedish MS Registry, the Danish MS Registry, OFSEP (Observatoire Français de al Sclérose en Plaques), and MSBase. Patients were included in the current analysis if they had at least 10 years of follow-up, had at least three EDSS evaluations, and at least one DMT prescription.
“It is well known that randomized, controlled trials support the early start of treatment in MS, but open-label extensions of the same trials reported inconsistent results about the long-term benefit on disability accumulation,” Dr. Iaffaldano explained. Further, recent observational studies have suggested that initiating DMTs early might not only delay the accumulation of disability but perhaps also death.
The aim of the research was thus to look at what effect the time interval from disease onset to the first administration of a DMT might have on long-term disability accumulation, as measured by the EDSS, in patients with RRMS.
The population of patients studied was mostly (71%) female, with a median age of 27 years at disease onset. The number of relapses prior to starting a DMT was two and the baseline EDSS was 2.0. In almost all (98.9%) cases, DMT was used as first-line treatment (second line in 1.1% of cases). The median follow-up was 13.2 years and cumulative DMT exposure was 10.5 years.
The work was supported by Biogen International on the basis of a sponsored research agreement with the Big Multiple Sclerosis Data Network. Dr. Iaffaldano has served on scientific advisory boards for and received funding for travel and/or speaker honoraria from Biogen and other companies that market DMTs for MS. Several study authors are employees of Biogen, and other study authors also reported financial ties to Biogen and other pharmaceutical companies.
SOURCE: Iaffaldano P et al. Mult Scler. 2018;24(Suppl 2):71-2, Abstract 204.
REPORTING FROM ECTRIMS 2018
Key clinical point: Less disease progression occurs if disease-modifying treatments (DMTs) are given early in relapsing-remitting multiple sclerosis (RRMS).
Major finding: DMTs within 6 months vs. later decreased the risk of confirmed first disability progression at 12 months by 28% (P = .003).
Study details: 11,934 patients with RRMS with at least 10 years’ follow-up, three or more Expanded Disability Status Scale evaluations, and at least one DMT prescription.
Disclosures: The work was supported by Biogen International on the basis of a sponsored research agreement with the Big Multiple Sclerosis Data Network. Dr. Iaffaldano has served on scientific advisory boards for and received funding for travel and/or speaker honoraria from Biogen and other companies that market DMTs for MS. Several study authors are employees of Biogen, and other study authors also reported financial ties to Biogen and other pharmaceutical companies.
Source: Iaffaldano P et al. Mult Scler. 2018;24(Suppl 2):71-2, Abstract 204.
Sex workers: High rates of HIV, low rates of treatment
The HIV pandemic among sex workers remains underaddressed and underresourced, with “glaring gaps” in comprehensive measures of HIV prevalence and incidence, and in prevention and treatment, according to the results of an updated literature review published in the Lancet.
Kate Shannon, PhD, director of the gender and sexual health initiative at the University of British Columbia, Vancouver, and her colleagues updated a 2013 literature search for the Lancet series on HIV and sex workers to include reports and manuscripts published from Jan. 1, 2006, to Sept. 6, 2017.
They found that In particular, 4 years after their previous Lancet series on HIV and sex workers, this updated analysis showed that the global HIV burden among female sex workers was still similar to the previously determined 11.8% and “unacceptably high” at 10.4%, (95% confidence interval, 9.5-11.5).
Although there has been some improvement in the assessment of HIV in transgender women since the previous analysis, according to Dr. Shannon and her colleagues, small sample sizes and conflation of transgender women and men who have sex with men (MSM) continue to limit the volume of transgender-specific HIV data, particularly in Africa.
Access to HIV prevention and treatment also remains a considerable problem for sex workers, according to the authors. In particular, “qualitative data in sub-Saharan Africa suggest that profound structural barriers of stigma and discrimination impede progress in the HIV care continuum,” with studies confirming that “successful HIV treatment trajectories are impeded by violence and displacement” because of policing, they wrote.
They pointed out that things may well become worse, with evidence-based progress on full decriminalization grounded in health and human rights – which was a key recommendation in their earlier Lancet Series – having stalled in all but South Africa. In fact, they reported that several countries had even rolled back rights further for sex workers.
“HIV prevention and treatment tools are available but, without comprehensive HIV epidemiology, a lack of denominators and failure to address structural determinants (including decriminalisation of sex work) means that progress in achieving health and rights for all sex workers will fall short,” the researchers concluded.
The authors reported that they had no competing interests.
SOURCE: Shannon K et al. Lancet. 2018 Aug 25;392:698-710.
The HIV pandemic among sex workers remains underaddressed and underresourced, with “glaring gaps” in comprehensive measures of HIV prevalence and incidence, and in prevention and treatment, according to the results of an updated literature review published in the Lancet.
Kate Shannon, PhD, director of the gender and sexual health initiative at the University of British Columbia, Vancouver, and her colleagues updated a 2013 literature search for the Lancet series on HIV and sex workers to include reports and manuscripts published from Jan. 1, 2006, to Sept. 6, 2017.
They found that In particular, 4 years after their previous Lancet series on HIV and sex workers, this updated analysis showed that the global HIV burden among female sex workers was still similar to the previously determined 11.8% and “unacceptably high” at 10.4%, (95% confidence interval, 9.5-11.5).
Although there has been some improvement in the assessment of HIV in transgender women since the previous analysis, according to Dr. Shannon and her colleagues, small sample sizes and conflation of transgender women and men who have sex with men (MSM) continue to limit the volume of transgender-specific HIV data, particularly in Africa.
Access to HIV prevention and treatment also remains a considerable problem for sex workers, according to the authors. In particular, “qualitative data in sub-Saharan Africa suggest that profound structural barriers of stigma and discrimination impede progress in the HIV care continuum,” with studies confirming that “successful HIV treatment trajectories are impeded by violence and displacement” because of policing, they wrote.
They pointed out that things may well become worse, with evidence-based progress on full decriminalization grounded in health and human rights – which was a key recommendation in their earlier Lancet Series – having stalled in all but South Africa. In fact, they reported that several countries had even rolled back rights further for sex workers.
“HIV prevention and treatment tools are available but, without comprehensive HIV epidemiology, a lack of denominators and failure to address structural determinants (including decriminalisation of sex work) means that progress in achieving health and rights for all sex workers will fall short,” the researchers concluded.
The authors reported that they had no competing interests.
SOURCE: Shannon K et al. Lancet. 2018 Aug 25;392:698-710.
The HIV pandemic among sex workers remains underaddressed and underresourced, with “glaring gaps” in comprehensive measures of HIV prevalence and incidence, and in prevention and treatment, according to the results of an updated literature review published in the Lancet.
Kate Shannon, PhD, director of the gender and sexual health initiative at the University of British Columbia, Vancouver, and her colleagues updated a 2013 literature search for the Lancet series on HIV and sex workers to include reports and manuscripts published from Jan. 1, 2006, to Sept. 6, 2017.
They found that In particular, 4 years after their previous Lancet series on HIV and sex workers, this updated analysis showed that the global HIV burden among female sex workers was still similar to the previously determined 11.8% and “unacceptably high” at 10.4%, (95% confidence interval, 9.5-11.5).
Although there has been some improvement in the assessment of HIV in transgender women since the previous analysis, according to Dr. Shannon and her colleagues, small sample sizes and conflation of transgender women and men who have sex with men (MSM) continue to limit the volume of transgender-specific HIV data, particularly in Africa.
Access to HIV prevention and treatment also remains a considerable problem for sex workers, according to the authors. In particular, “qualitative data in sub-Saharan Africa suggest that profound structural barriers of stigma and discrimination impede progress in the HIV care continuum,” with studies confirming that “successful HIV treatment trajectories are impeded by violence and displacement” because of policing, they wrote.
They pointed out that things may well become worse, with evidence-based progress on full decriminalization grounded in health and human rights – which was a key recommendation in their earlier Lancet Series – having stalled in all but South Africa. In fact, they reported that several countries had even rolled back rights further for sex workers.
“HIV prevention and treatment tools are available but, without comprehensive HIV epidemiology, a lack of denominators and failure to address structural determinants (including decriminalisation of sex work) means that progress in achieving health and rights for all sex workers will fall short,” the researchers concluded.
The authors reported that they had no competing interests.
SOURCE: Shannon K et al. Lancet. 2018 Aug 25;392:698-710.
FROM THE LANCET
Key clinical point: The HIV pandemic among sex workers remains underaddressed and underresourced, with “glaring gaps” in assessment, treatment.
Major finding: The global HIV burden among female sex workers shows that HIV prevalence was “unacceptably high” at 10.4%.
Study details: Researchers updated a 2013 literature review with reports published from Jan. 1, 2006, to Sept. 6, 2017.
Disclosures: The authors reported that they had no competing interests.
Source: Shannon K et al. Lancet. 2018 Aug 25;392:698-710.
First Episode: Dr. Lorenzo Norris
At MDedge, we know that medicine can be a bit of an awakening at every step of your career. So, we launched the Postcall Podcast as a way to share your stories: what you love about medicine and what you love outside of your career. This podcast is meant to be a place for you to find your truth.
In the first edition, MDedge producer and host Nick Andrews sits down with Lorenzo Norris, MD. Dr. Norris is the host of the MDedge Psychcast as well as the editor-in-chief of MDedge Psychiatry and Dean at the George Washington University School of Medicine and Health Sciences.
Subscribe to the Postcall Podcast:
Apple Podcasts
Google Podcasts
At MDedge, we know that medicine can be a bit of an awakening at every step of your career. So, we launched the Postcall Podcast as a way to share your stories: what you love about medicine and what you love outside of your career. This podcast is meant to be a place for you to find your truth.
In the first edition, MDedge producer and host Nick Andrews sits down with Lorenzo Norris, MD. Dr. Norris is the host of the MDedge Psychcast as well as the editor-in-chief of MDedge Psychiatry and Dean at the George Washington University School of Medicine and Health Sciences.
Subscribe to the Postcall Podcast:
Apple Podcasts
Google Podcasts
At MDedge, we know that medicine can be a bit of an awakening at every step of your career. So, we launched the Postcall Podcast as a way to share your stories: what you love about medicine and what you love outside of your career. This podcast is meant to be a place for you to find your truth.
In the first edition, MDedge producer and host Nick Andrews sits down with Lorenzo Norris, MD. Dr. Norris is the host of the MDedge Psychcast as well as the editor-in-chief of MDedge Psychiatry and Dean at the George Washington University School of Medicine and Health Sciences.
Subscribe to the Postcall Podcast:
Apple Podcasts
Google Podcasts
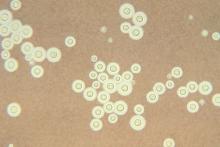
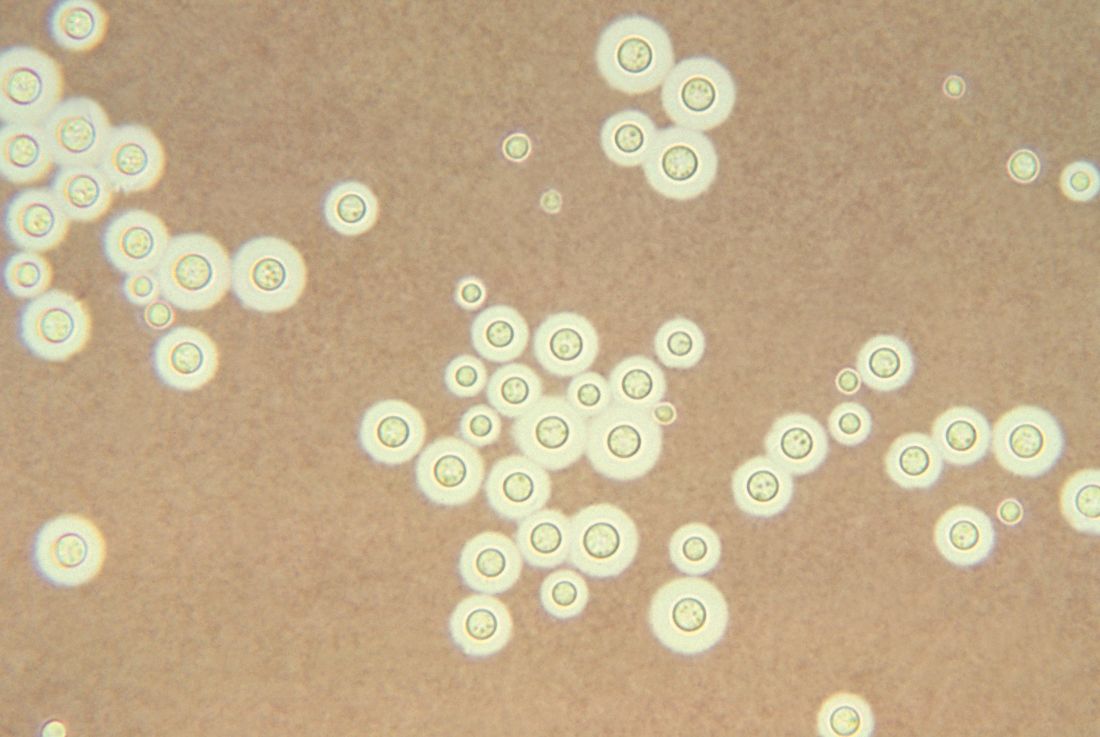
The challenges of diagnosing CMML
DUBROVNIK, CROATIA—Diagnosing chronic myelomonocytic leukemia (CMML) remains a challenge in 2018, according to a presentation at Leukemia and Lymphoma: Europe and the USA, Linking Knowledge and Practice.
Even with updated World Health Organization (WHO) criteria, karyotyping, and genetic analyses, it can be difficult to distinguish CMML from other conditions, according to Nadira Duraković, MD, PhD, of the University Hospital Zagreb in Croatia.
However, Dr. Duraković said there are characteristics that differentiate CMML from myelodysplastic syndromes (MDS), myeloproliferative neoplasms (MPNs), and atypical chronic myeloid leukemia (CML).
Furthermore, studies have suggested that monocyte subset distribution analysis can be useful for diagnosing CMML.
Dr. Duraković began her presentation with an overview of the 2016 WHO classification of CMML (Blood 2016 127:2391-2405).
According to the WHO, patients have CMML if:
- They have persistent peripheral blood monocytosis (1×109/L) with monocytes accounting for 10% of the white blood cell count
- They do not meet WHO criteria for BCR-ABL1-positive CML, primary myelofibrosis, polycythemia vera, or essential thrombocythemia
- There is no evidence of PCM1-JAK2 or PDGFRA, PDGFRB, or FGFR1 rearrangement
- They have fewer than 20% blasts in the blood and bone marrow
- They have dysplasia in one or more myeloid lineages
- If myelodysplasia is absent or minimal, an acquired clonal cytogenetic or molecular genetic abnormality must be present.
Alternatively, if patients have monocytosis that has persisted for at least 3 months, and all other causes of monocytosis have been excluded, “you can say that your patient has CMML,” Dr. Duraković said.
Other causes of monocytosis include infections, malignancies, medications, inflammatory conditions, and other conditions such as pregnancy.
However, Dr. Duraković pointed out that the cause of monocytosis cannot always be determined, and, in some cases, CMML patients may not meet the WHO criteria.
“[T]here are cases where there just aren’t enough monocytes to fulfill the WHO criteria,” Dr. Duraković said. “You can have a patient with peripheral blood cytopenia and monocytosis who does not have 1,000 monocytes. Patients can have progressive dysplasia, can have splenomegaly, be really sick, but fail to meet WHO criteria.”
Distinguishing CMML from other conditions
“Differentiating CMML from myelodysplastic syndromes can be tough,” Dr. Duraković said. “There are dysplastic features that are present in CMML . . . but, in CMML, they are more subtle, and they are more difficult to appreciate than in myelodysplastic syndromes.”
The ratio of myeloid to erythroid cells is elevated in CMML, and patients may have atypical monocytes (paramyeloid cells) that are unique to CMML.
Dr. Duraković noted that megakaryocyte dysplasia in CMML can be characterized by “myeloproliferative megakaryocytes,” which are large cells that cluster and have hyperlobulated nuclei, or “MDS megakaryocytes,” which are small, solitary cells with hypolobulated nuclei.
She went on to explain that “MPN phenotype” CMML is characterized by leukocytosis, monocytosis, hepatomegaly, splenomegaly, and clinical features of myeloproliferation (fatigue, night sweats, bone pain, weight loss, etc.).
Thirty percent of cases are associated with splenomegaly, and 30% of patients can have an increase in bone marrow reticulin fibrosis.
Dr. Duraković also noted that a prior MPN diagnosis excludes CMML. The presence of common MPN mutations, such as JAK2, CALR, or MPL, suggests a patient has an MPN with monocytosis rather than CMML.
Patients who have unclassified MPNs or MDS, rather than CMML, either do not have 1,000 monocytes or the monocytes do not represent more than 10% of the differential, Dr. Duraković said.
She also noted that it can be difficult to differentiate CMML from atypical CML.
“Atypical CML is characterized by profound dysgranulopoiesis, absence of the BCR-ABL1 fusion gene, and neutrophilia,” Dr. Duraković explained. “Those patients [commonly] have monocytosis, but, here, that 10% rule is valuable because their monocytes comprise less than 10% of the entire white blood cell count.”
Karyotyping, genotyping, and immunophenotyping
“There is no disease-defining karyotype abnormality [in CMML],” Dr. Duraković noted.
She said 30% of patients have abnormal karyotype, and the most common abnormality is trisomy 8. Unlike in patients with MDS, del(5q) and monosomal karyotypes are infrequent in patients with CMML.
Similarly, there are no “disease-defining” mutations or genetic changes in CMML, although CMML is genetically distinct from MDS, Dr. Duraković said.
For instance, SRSF2 encodes a component of the spliceosome that is mutated in almost half of CMML patients and less than 10% of MDS patients. Likewise, ASLX1 and TET2 are “much more frequently involved” in CMML than in MDS, Dr. Duraković said.
In a 2012 study of 275 CMML patients, researchers found that 93% of patients had at least one somatic mutation in nine recurrently mutated genes—SRFS2, ASXL1, CBL, EZH2, JAK2V617F, KRAS, NRAS, RUNX1, and TET2 (Blood 2012 120:3080-3088).
However, Dr. Duraković noted that these mutations are found in other disorders as well, so this information may not be helpful in differentiating CMML from other disorders.
A 2015 study revealed a technique that does appear useful for identifying CMML—monocyte subset distribution analysis (Blood 2015 125(23): 3618–3626).
For this analysis, monocytes are divided into the following categories:
- Classical/MO1 (CD14bright/CD16−)
- Intermediate/MO2 (CD14bright/CD16+)
- Non-classical/MO3 (CD14dim/CD16+).
The researchers found that CMML patients had an increase in the fraction of classical monocytes (with a cutoff value of 94.0%), as compared to healthy control subjects, patients with another hematologic disorder, and patients with reactive monocytosis.
A 2018 study confirmed that monocyte subset distribution analysis could differentiate CMML from other hematologic disorders, with the exception of atypical CML (Am J Clin Pathol 2018 150(4):293-302).
This study also suggested that a decreased percentage of non-classical monocytes was more sensitive than an increased percentage of classical monocytes.
Despite the differences between these studies, “monocyte subset distribution analysis is showing promise as a method of identifying hard-to-identify CMML patients with ease and affordability,” Dr. Duraković said.
She added that the technique can be implemented in clinical practice using the HematoflowTM solution (Cytometry B Clin Cytom 2018 94(5):658-661).
Dr. Duraković did not report any conflicts of interest.
DUBROVNIK, CROATIA—Diagnosing chronic myelomonocytic leukemia (CMML) remains a challenge in 2018, according to a presentation at Leukemia and Lymphoma: Europe and the USA, Linking Knowledge and Practice.
Even with updated World Health Organization (WHO) criteria, karyotyping, and genetic analyses, it can be difficult to distinguish CMML from other conditions, according to Nadira Duraković, MD, PhD, of the University Hospital Zagreb in Croatia.
However, Dr. Duraković said there are characteristics that differentiate CMML from myelodysplastic syndromes (MDS), myeloproliferative neoplasms (MPNs), and atypical chronic myeloid leukemia (CML).
Furthermore, studies have suggested that monocyte subset distribution analysis can be useful for diagnosing CMML.
Dr. Duraković began her presentation with an overview of the 2016 WHO classification of CMML (Blood 2016 127:2391-2405).
According to the WHO, patients have CMML if:
- They have persistent peripheral blood monocytosis (1×109/L) with monocytes accounting for 10% of the white blood cell count
- They do not meet WHO criteria for BCR-ABL1-positive CML, primary myelofibrosis, polycythemia vera, or essential thrombocythemia
- There is no evidence of PCM1-JAK2 or PDGFRA, PDGFRB, or FGFR1 rearrangement
- They have fewer than 20% blasts in the blood and bone marrow
- They have dysplasia in one or more myeloid lineages
- If myelodysplasia is absent or minimal, an acquired clonal cytogenetic or molecular genetic abnormality must be present.
Alternatively, if patients have monocytosis that has persisted for at least 3 months, and all other causes of monocytosis have been excluded, “you can say that your patient has CMML,” Dr. Duraković said.
Other causes of monocytosis include infections, malignancies, medications, inflammatory conditions, and other conditions such as pregnancy.
However, Dr. Duraković pointed out that the cause of monocytosis cannot always be determined, and, in some cases, CMML patients may not meet the WHO criteria.
“[T]here are cases where there just aren’t enough monocytes to fulfill the WHO criteria,” Dr. Duraković said. “You can have a patient with peripheral blood cytopenia and monocytosis who does not have 1,000 monocytes. Patients can have progressive dysplasia, can have splenomegaly, be really sick, but fail to meet WHO criteria.”
Distinguishing CMML from other conditions
“Differentiating CMML from myelodysplastic syndromes can be tough,” Dr. Duraković said. “There are dysplastic features that are present in CMML . . . but, in CMML, they are more subtle, and they are more difficult to appreciate than in myelodysplastic syndromes.”
The ratio of myeloid to erythroid cells is elevated in CMML, and patients may have atypical monocytes (paramyeloid cells) that are unique to CMML.
Dr. Duraković noted that megakaryocyte dysplasia in CMML can be characterized by “myeloproliferative megakaryocytes,” which are large cells that cluster and have hyperlobulated nuclei, or “MDS megakaryocytes,” which are small, solitary cells with hypolobulated nuclei.
She went on to explain that “MPN phenotype” CMML is characterized by leukocytosis, monocytosis, hepatomegaly, splenomegaly, and clinical features of myeloproliferation (fatigue, night sweats, bone pain, weight loss, etc.).
Thirty percent of cases are associated with splenomegaly, and 30% of patients can have an increase in bone marrow reticulin fibrosis.
Dr. Duraković also noted that a prior MPN diagnosis excludes CMML. The presence of common MPN mutations, such as JAK2, CALR, or MPL, suggests a patient has an MPN with monocytosis rather than CMML.
Patients who have unclassified MPNs or MDS, rather than CMML, either do not have 1,000 monocytes or the monocytes do not represent more than 10% of the differential, Dr. Duraković said.
She also noted that it can be difficult to differentiate CMML from atypical CML.
“Atypical CML is characterized by profound dysgranulopoiesis, absence of the BCR-ABL1 fusion gene, and neutrophilia,” Dr. Duraković explained. “Those patients [commonly] have monocytosis, but, here, that 10% rule is valuable because their monocytes comprise less than 10% of the entire white blood cell count.”
Karyotyping, genotyping, and immunophenotyping
“There is no disease-defining karyotype abnormality [in CMML],” Dr. Duraković noted.
She said 30% of patients have abnormal karyotype, and the most common abnormality is trisomy 8. Unlike in patients with MDS, del(5q) and monosomal karyotypes are infrequent in patients with CMML.
Similarly, there are no “disease-defining” mutations or genetic changes in CMML, although CMML is genetically distinct from MDS, Dr. Duraković said.
For instance, SRSF2 encodes a component of the spliceosome that is mutated in almost half of CMML patients and less than 10% of MDS patients. Likewise, ASLX1 and TET2 are “much more frequently involved” in CMML than in MDS, Dr. Duraković said.
In a 2012 study of 275 CMML patients, researchers found that 93% of patients had at least one somatic mutation in nine recurrently mutated genes—SRFS2, ASXL1, CBL, EZH2, JAK2V617F, KRAS, NRAS, RUNX1, and TET2 (Blood 2012 120:3080-3088).
However, Dr. Duraković noted that these mutations are found in other disorders as well, so this information may not be helpful in differentiating CMML from other disorders.
A 2015 study revealed a technique that does appear useful for identifying CMML—monocyte subset distribution analysis (Blood 2015 125(23): 3618–3626).
For this analysis, monocytes are divided into the following categories:
- Classical/MO1 (CD14bright/CD16−)
- Intermediate/MO2 (CD14bright/CD16+)
- Non-classical/MO3 (CD14dim/CD16+).
The researchers found that CMML patients had an increase in the fraction of classical monocytes (with a cutoff value of 94.0%), as compared to healthy control subjects, patients with another hematologic disorder, and patients with reactive monocytosis.
A 2018 study confirmed that monocyte subset distribution analysis could differentiate CMML from other hematologic disorders, with the exception of atypical CML (Am J Clin Pathol 2018 150(4):293-302).
This study also suggested that a decreased percentage of non-classical monocytes was more sensitive than an increased percentage of classical monocytes.
Despite the differences between these studies, “monocyte subset distribution analysis is showing promise as a method of identifying hard-to-identify CMML patients with ease and affordability,” Dr. Duraković said.
She added that the technique can be implemented in clinical practice using the HematoflowTM solution (Cytometry B Clin Cytom 2018 94(5):658-661).
Dr. Duraković did not report any conflicts of interest.
DUBROVNIK, CROATIA—Diagnosing chronic myelomonocytic leukemia (CMML) remains a challenge in 2018, according to a presentation at Leukemia and Lymphoma: Europe and the USA, Linking Knowledge and Practice.
Even with updated World Health Organization (WHO) criteria, karyotyping, and genetic analyses, it can be difficult to distinguish CMML from other conditions, according to Nadira Duraković, MD, PhD, of the University Hospital Zagreb in Croatia.
However, Dr. Duraković said there are characteristics that differentiate CMML from myelodysplastic syndromes (MDS), myeloproliferative neoplasms (MPNs), and atypical chronic myeloid leukemia (CML).
Furthermore, studies have suggested that monocyte subset distribution analysis can be useful for diagnosing CMML.
Dr. Duraković began her presentation with an overview of the 2016 WHO classification of CMML (Blood 2016 127:2391-2405).
According to the WHO, patients have CMML if:
- They have persistent peripheral blood monocytosis (1×109/L) with monocytes accounting for 10% of the white blood cell count
- They do not meet WHO criteria for BCR-ABL1-positive CML, primary myelofibrosis, polycythemia vera, or essential thrombocythemia
- There is no evidence of PCM1-JAK2 or PDGFRA, PDGFRB, or FGFR1 rearrangement
- They have fewer than 20% blasts in the blood and bone marrow
- They have dysplasia in one or more myeloid lineages
- If myelodysplasia is absent or minimal, an acquired clonal cytogenetic or molecular genetic abnormality must be present.
Alternatively, if patients have monocytosis that has persisted for at least 3 months, and all other causes of monocytosis have been excluded, “you can say that your patient has CMML,” Dr. Duraković said.
Other causes of monocytosis include infections, malignancies, medications, inflammatory conditions, and other conditions such as pregnancy.
However, Dr. Duraković pointed out that the cause of monocytosis cannot always be determined, and, in some cases, CMML patients may not meet the WHO criteria.
“[T]here are cases where there just aren’t enough monocytes to fulfill the WHO criteria,” Dr. Duraković said. “You can have a patient with peripheral blood cytopenia and monocytosis who does not have 1,000 monocytes. Patients can have progressive dysplasia, can have splenomegaly, be really sick, but fail to meet WHO criteria.”
Distinguishing CMML from other conditions
“Differentiating CMML from myelodysplastic syndromes can be tough,” Dr. Duraković said. “There are dysplastic features that are present in CMML . . . but, in CMML, they are more subtle, and they are more difficult to appreciate than in myelodysplastic syndromes.”
The ratio of myeloid to erythroid cells is elevated in CMML, and patients may have atypical monocytes (paramyeloid cells) that are unique to CMML.
Dr. Duraković noted that megakaryocyte dysplasia in CMML can be characterized by “myeloproliferative megakaryocytes,” which are large cells that cluster and have hyperlobulated nuclei, or “MDS megakaryocytes,” which are small, solitary cells with hypolobulated nuclei.
She went on to explain that “MPN phenotype” CMML is characterized by leukocytosis, monocytosis, hepatomegaly, splenomegaly, and clinical features of myeloproliferation (fatigue, night sweats, bone pain, weight loss, etc.).
Thirty percent of cases are associated with splenomegaly, and 30% of patients can have an increase in bone marrow reticulin fibrosis.
Dr. Duraković also noted that a prior MPN diagnosis excludes CMML. The presence of common MPN mutations, such as JAK2, CALR, or MPL, suggests a patient has an MPN with monocytosis rather than CMML.
Patients who have unclassified MPNs or MDS, rather than CMML, either do not have 1,000 monocytes or the monocytes do not represent more than 10% of the differential, Dr. Duraković said.
She also noted that it can be difficult to differentiate CMML from atypical CML.
“Atypical CML is characterized by profound dysgranulopoiesis, absence of the BCR-ABL1 fusion gene, and neutrophilia,” Dr. Duraković explained. “Those patients [commonly] have monocytosis, but, here, that 10% rule is valuable because their monocytes comprise less than 10% of the entire white blood cell count.”
Karyotyping, genotyping, and immunophenotyping
“There is no disease-defining karyotype abnormality [in CMML],” Dr. Duraković noted.
She said 30% of patients have abnormal karyotype, and the most common abnormality is trisomy 8. Unlike in patients with MDS, del(5q) and monosomal karyotypes are infrequent in patients with CMML.
Similarly, there are no “disease-defining” mutations or genetic changes in CMML, although CMML is genetically distinct from MDS, Dr. Duraković said.
For instance, SRSF2 encodes a component of the spliceosome that is mutated in almost half of CMML patients and less than 10% of MDS patients. Likewise, ASLX1 and TET2 are “much more frequently involved” in CMML than in MDS, Dr. Duraković said.
In a 2012 study of 275 CMML patients, researchers found that 93% of patients had at least one somatic mutation in nine recurrently mutated genes—SRFS2, ASXL1, CBL, EZH2, JAK2V617F, KRAS, NRAS, RUNX1, and TET2 (Blood 2012 120:3080-3088).
However, Dr. Duraković noted that these mutations are found in other disorders as well, so this information may not be helpful in differentiating CMML from other disorders.
A 2015 study revealed a technique that does appear useful for identifying CMML—monocyte subset distribution analysis (Blood 2015 125(23): 3618–3626).
For this analysis, monocytes are divided into the following categories:
- Classical/MO1 (CD14bright/CD16−)
- Intermediate/MO2 (CD14bright/CD16+)
- Non-classical/MO3 (CD14dim/CD16+).
The researchers found that CMML patients had an increase in the fraction of classical monocytes (with a cutoff value of 94.0%), as compared to healthy control subjects, patients with another hematologic disorder, and patients with reactive monocytosis.
A 2018 study confirmed that monocyte subset distribution analysis could differentiate CMML from other hematologic disorders, with the exception of atypical CML (Am J Clin Pathol 2018 150(4):293-302).
This study also suggested that a decreased percentage of non-classical monocytes was more sensitive than an increased percentage of classical monocytes.
Despite the differences between these studies, “monocyte subset distribution analysis is showing promise as a method of identifying hard-to-identify CMML patients with ease and affordability,” Dr. Duraković said.
She added that the technique can be implemented in clinical practice using the HematoflowTM solution (Cytometry B Clin Cytom 2018 94(5):658-661).
Dr. Duraković did not report any conflicts of interest.
Crizanlizumab appears effective across subgroups
Crizanlizumab can reduce vaso-occlusive crises (VOCs) across subgroups of patients with sickle cell disease (SCD), according to a post-hoc analysis of the phase 2 SUSTAIN trial.
Researchers found crizanlizumab was more effective than placebo at delaying time to first VOC and eliminating crises in patients who had numerous previous crises, exhibited the HbSS genotype, or were taking concomitant hydroxyurea.
Abdullah Kutlar, MD, of the Medical College of Georgia in Augusta, and his colleagues reported these findings in the American Journal of Hematology.
The phase 2 SUSTAIN trial previously showed that crizanlizumab—a humanized, anti–P-selectin monoclonal antibody—reduced the frequency of VOCs by 45% and delayed time to first crisis by about 3 months.
Additionally, a subgroup analysis showed there was a lower frequency of VOCs with crizanlizumab at 5 mg/kg, compared with placebo, regardless of the number of prior VOCs, concomitant hydroxyurea use, or the SCD genotype.
The present post-hoc analysis took a deeper look at these observations across the same subgroups. Specifically, the investigators assessed elimination of VOCs, time to first crisis, and adverse events in 132 patients.
Crizanlizumab eliminated VOCs about seven times more frequently than did placebo in patients who had a high frequency of VOCs before the study (5 to 10 VOCs in the year prior)—28.0% and 4.2%, respectively.
Crizanlizumab eliminated VOCs about twice as often as placebo in patients with the HbSS genotype—31.9% and 17.0%, respectively—and in patients who were using concomitant hydroxyurea—33.3% and 17.5%, respectively.
Further analysis showed that crizanlizumab delayed time to first VOC across all subgroups.
In patients with the HbSS genotype, the time to first VOC was 4.07 months with crizanlizumab and 1.12 months with placebo.
In patients with a higher frequency of previous VOCs, the time to first on-study VOC was 2.43 months with crizanlizumab and 1.03 months with placebo.
In patients taking hydroxyurea, the time to first VOC was 2.43 months with crizanlizumab and 1.45 months with placebo.
Safety was comparable across subgroups.
This study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
Crizanlizumab can reduce vaso-occlusive crises (VOCs) across subgroups of patients with sickle cell disease (SCD), according to a post-hoc analysis of the phase 2 SUSTAIN trial.
Researchers found crizanlizumab was more effective than placebo at delaying time to first VOC and eliminating crises in patients who had numerous previous crises, exhibited the HbSS genotype, or were taking concomitant hydroxyurea.
Abdullah Kutlar, MD, of the Medical College of Georgia in Augusta, and his colleagues reported these findings in the American Journal of Hematology.
The phase 2 SUSTAIN trial previously showed that crizanlizumab—a humanized, anti–P-selectin monoclonal antibody—reduced the frequency of VOCs by 45% and delayed time to first crisis by about 3 months.
Additionally, a subgroup analysis showed there was a lower frequency of VOCs with crizanlizumab at 5 mg/kg, compared with placebo, regardless of the number of prior VOCs, concomitant hydroxyurea use, or the SCD genotype.
The present post-hoc analysis took a deeper look at these observations across the same subgroups. Specifically, the investigators assessed elimination of VOCs, time to first crisis, and adverse events in 132 patients.
Crizanlizumab eliminated VOCs about seven times more frequently than did placebo in patients who had a high frequency of VOCs before the study (5 to 10 VOCs in the year prior)—28.0% and 4.2%, respectively.
Crizanlizumab eliminated VOCs about twice as often as placebo in patients with the HbSS genotype—31.9% and 17.0%, respectively—and in patients who were using concomitant hydroxyurea—33.3% and 17.5%, respectively.
Further analysis showed that crizanlizumab delayed time to first VOC across all subgroups.
In patients with the HbSS genotype, the time to first VOC was 4.07 months with crizanlizumab and 1.12 months with placebo.
In patients with a higher frequency of previous VOCs, the time to first on-study VOC was 2.43 months with crizanlizumab and 1.03 months with placebo.
In patients taking hydroxyurea, the time to first VOC was 2.43 months with crizanlizumab and 1.45 months with placebo.
Safety was comparable across subgroups.
This study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
Crizanlizumab can reduce vaso-occlusive crises (VOCs) across subgroups of patients with sickle cell disease (SCD), according to a post-hoc analysis of the phase 2 SUSTAIN trial.
Researchers found crizanlizumab was more effective than placebo at delaying time to first VOC and eliminating crises in patients who had numerous previous crises, exhibited the HbSS genotype, or were taking concomitant hydroxyurea.
Abdullah Kutlar, MD, of the Medical College of Georgia in Augusta, and his colleagues reported these findings in the American Journal of Hematology.
The phase 2 SUSTAIN trial previously showed that crizanlizumab—a humanized, anti–P-selectin monoclonal antibody—reduced the frequency of VOCs by 45% and delayed time to first crisis by about 3 months.
Additionally, a subgroup analysis showed there was a lower frequency of VOCs with crizanlizumab at 5 mg/kg, compared with placebo, regardless of the number of prior VOCs, concomitant hydroxyurea use, or the SCD genotype.
The present post-hoc analysis took a deeper look at these observations across the same subgroups. Specifically, the investigators assessed elimination of VOCs, time to first crisis, and adverse events in 132 patients.
Crizanlizumab eliminated VOCs about seven times more frequently than did placebo in patients who had a high frequency of VOCs before the study (5 to 10 VOCs in the year prior)—28.0% and 4.2%, respectively.
Crizanlizumab eliminated VOCs about twice as often as placebo in patients with the HbSS genotype—31.9% and 17.0%, respectively—and in patients who were using concomitant hydroxyurea—33.3% and 17.5%, respectively.
Further analysis showed that crizanlizumab delayed time to first VOC across all subgroups.
In patients with the HbSS genotype, the time to first VOC was 4.07 months with crizanlizumab and 1.12 months with placebo.
In patients with a higher frequency of previous VOCs, the time to first on-study VOC was 2.43 months with crizanlizumab and 1.03 months with placebo.
In patients taking hydroxyurea, the time to first VOC was 2.43 months with crizanlizumab and 1.45 months with placebo.
Safety was comparable across subgroups.
This study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
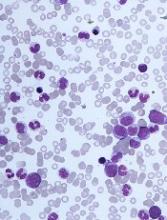
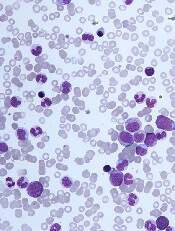
Bacteremic sepsis in ALL tied to neurocognitive dysfunction
Bacteremic sepsis during acute lymphoblastic leukemia (ALL) treatment may contribute to neurocognitive dysfunction later in life, results of a cohort study suggest.
Pediatric ALL survivors who had sepsis while on treatment performed worse on measures of intelligence, attention, executive function, and processing speed than survivors with no sepsis history, according to study results.
Links between sepsis and impaired neurocognitive function found in this study have “practice-changing implications” for cancer survivors, investigators reported in JAMA Pediatrics.
“Prevention of infection, early recognition and appropriate management of sepsis, and preemptive neurocognitive interventions should be prioritized, because these might prevent or ameliorate neurologic damage,” said Joshua Wolf, MBBS, of St. Jude Children’s Research Hospital, Memphis, and the coauthors of the report.
The study included 212 children who, at a median age of 5 years, had received risk-adapted chemotherapy for ALL with no hematopoietic cell transplant or cranial irradiation.
Sixteen of the patients (7.5%) had a history of bacteremic sepsis during ALL therapy, according to retrospectively obtained data.
As a part of the study, all patients participated in neurocognitive testing, which was done at a median of 7.7 years after diagnosis.
Patients with a history of bacteremic sepsis performed poorly on multiple measures of neurocognitive function, as compared with all other patients, according to results of analyses that were adjusted for multiple potentially confounding factors, such as age, race, and leukemia risk category.
Although not all neurocognitive measures were significantly different between groups, survivors with a sepsis history performed worse on evaluations of spatial planning (difference, 0.78; 95% CI, 0.57-1.00), verbal fluency (0.38; 95% CI, 0.14-0.62), and attention (0.63; 95% CI, 0.30-0.95), among other measures.
This is believed to be the first published study looking at potential links between sepsis during ALL treatment and long-term neurocognitive dysfunction, investigators said. However, similar observations have been made in other patient populations, they added.
Exactly how sepsis might lead to neurocognitive deficits remains unclear.
“In the population of children with cancer, these mechanisms might be augmented by increased blood-brain barrier permeability to neurotoxic chemotherapy drugs,” the investigators said in their report.
Further study is needed to look at potential brain injury mechanisms and to validate the current findings in other ALL patient cohorts, they concluded.
The study was supported by the National Institute of Mental Health, the National Cancer Institute, and the American Lebanese Syrian Associated Charities. The researchers reported having no conflicts of interest.
Bacteremic sepsis during acute lymphoblastic leukemia (ALL) treatment may contribute to neurocognitive dysfunction later in life, results of a cohort study suggest.
Pediatric ALL survivors who had sepsis while on treatment performed worse on measures of intelligence, attention, executive function, and processing speed than survivors with no sepsis history, according to study results.
Links between sepsis and impaired neurocognitive function found in this study have “practice-changing implications” for cancer survivors, investigators reported in JAMA Pediatrics.
“Prevention of infection, early recognition and appropriate management of sepsis, and preemptive neurocognitive interventions should be prioritized, because these might prevent or ameliorate neurologic damage,” said Joshua Wolf, MBBS, of St. Jude Children’s Research Hospital, Memphis, and the coauthors of the report.
The study included 212 children who, at a median age of 5 years, had received risk-adapted chemotherapy for ALL with no hematopoietic cell transplant or cranial irradiation.
Sixteen of the patients (7.5%) had a history of bacteremic sepsis during ALL therapy, according to retrospectively obtained data.
As a part of the study, all patients participated in neurocognitive testing, which was done at a median of 7.7 years after diagnosis.
Patients with a history of bacteremic sepsis performed poorly on multiple measures of neurocognitive function, as compared with all other patients, according to results of analyses that were adjusted for multiple potentially confounding factors, such as age, race, and leukemia risk category.
Although not all neurocognitive measures were significantly different between groups, survivors with a sepsis history performed worse on evaluations of spatial planning (difference, 0.78; 95% CI, 0.57-1.00), verbal fluency (0.38; 95% CI, 0.14-0.62), and attention (0.63; 95% CI, 0.30-0.95), among other measures.
This is believed to be the first published study looking at potential links between sepsis during ALL treatment and long-term neurocognitive dysfunction, investigators said. However, similar observations have been made in other patient populations, they added.
Exactly how sepsis might lead to neurocognitive deficits remains unclear.
“In the population of children with cancer, these mechanisms might be augmented by increased blood-brain barrier permeability to neurotoxic chemotherapy drugs,” the investigators said in their report.
Further study is needed to look at potential brain injury mechanisms and to validate the current findings in other ALL patient cohorts, they concluded.
The study was supported by the National Institute of Mental Health, the National Cancer Institute, and the American Lebanese Syrian Associated Charities. The researchers reported having no conflicts of interest.
Bacteremic sepsis during acute lymphoblastic leukemia (ALL) treatment may contribute to neurocognitive dysfunction later in life, results of a cohort study suggest.
Pediatric ALL survivors who had sepsis while on treatment performed worse on measures of intelligence, attention, executive function, and processing speed than survivors with no sepsis history, according to study results.
Links between sepsis and impaired neurocognitive function found in this study have “practice-changing implications” for cancer survivors, investigators reported in JAMA Pediatrics.
“Prevention of infection, early recognition and appropriate management of sepsis, and preemptive neurocognitive interventions should be prioritized, because these might prevent or ameliorate neurologic damage,” said Joshua Wolf, MBBS, of St. Jude Children’s Research Hospital, Memphis, and the coauthors of the report.
The study included 212 children who, at a median age of 5 years, had received risk-adapted chemotherapy for ALL with no hematopoietic cell transplant or cranial irradiation.
Sixteen of the patients (7.5%) had a history of bacteremic sepsis during ALL therapy, according to retrospectively obtained data.
As a part of the study, all patients participated in neurocognitive testing, which was done at a median of 7.7 years after diagnosis.
Patients with a history of bacteremic sepsis performed poorly on multiple measures of neurocognitive function, as compared with all other patients, according to results of analyses that were adjusted for multiple potentially confounding factors, such as age, race, and leukemia risk category.
Although not all neurocognitive measures were significantly different between groups, survivors with a sepsis history performed worse on evaluations of spatial planning (difference, 0.78; 95% CI, 0.57-1.00), verbal fluency (0.38; 95% CI, 0.14-0.62), and attention (0.63; 95% CI, 0.30-0.95), among other measures.
This is believed to be the first published study looking at potential links between sepsis during ALL treatment and long-term neurocognitive dysfunction, investigators said. However, similar observations have been made in other patient populations, they added.
Exactly how sepsis might lead to neurocognitive deficits remains unclear.
“In the population of children with cancer, these mechanisms might be augmented by increased blood-brain barrier permeability to neurotoxic chemotherapy drugs,” the investigators said in their report.
Further study is needed to look at potential brain injury mechanisms and to validate the current findings in other ALL patient cohorts, they concluded.
The study was supported by the National Institute of Mental Health, the National Cancer Institute, and the American Lebanese Syrian Associated Charities. The researchers reported having no conflicts of interest.
NSAIDs’ short-term safety, IVUS beats angio, and more
, NSAIDs show cardiovascular and renal safety in the short term, a registry analysis supports use of direct oral anticoagulants over warfarin, and IVUS-guided stent placement bests angiography-led placement.
Subscribe to Cardiocast wherever you get your podcasts.
Amazon
Alexa
Apple Podcasts
Google Podcasts
, NSAIDs show cardiovascular and renal safety in the short term, a registry analysis supports use of direct oral anticoagulants over warfarin, and IVUS-guided stent placement bests angiography-led placement.
Subscribe to Cardiocast wherever you get your podcasts.
Amazon
Alexa
Apple Podcasts
Google Podcasts
, NSAIDs show cardiovascular and renal safety in the short term, a registry analysis supports use of direct oral anticoagulants over warfarin, and IVUS-guided stent placement bests angiography-led placement.
Subscribe to Cardiocast wherever you get your podcasts.
Amazon
Alexa
Apple Podcasts
Google Podcasts
Patient Preferences in Office-Based Orthopedic Care: A Prospective Evaluation
ABSTRACT
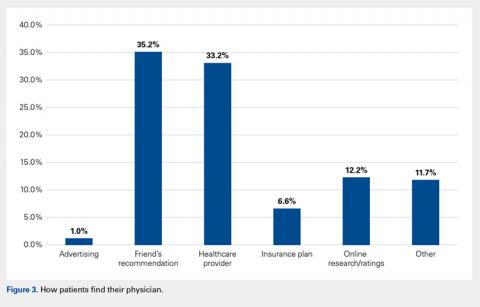
Patient satisfaction has become a topic of interest within orthopedics as the landscape of provider reimbursement continues to evolve to reward value of care. Online internet physician rating sites are becoming increasingly popular ways for patients to subjectively express their provider experience. Understanding what patients value during their episode of care is important in the modern healthcare environment. The purpose of this study is to determine what preferences, if any, patients have when selecting their physician and how they experience care in an outpatient orthopedic setting. A prospective survey was electronically administered to 212 patients in an adult reconstruction clinic. One hundred ninety-six patients (92.5%) completed the survey. Demographic questions regarding age, sex, ethnicity, and prior adult reconstruction surgical history were obtained. When patients were asked how much time they would like the doctor to spend with them on a routine visit, the most common answer was 10 to 15 minutes (41.3%), with only 10.2% patients desiring >20 minutes. The majority of patients (83.1%) believe ≥30 minutes is too long to wait to see their surgeon. Less than half of patients (41.8%) stated that they would feel as though they were receiving below average care if seen only by a nurse practitioner or physician’s assistant at a postoperative visit. Patients reported no significant age, gender, or ethnicity preferences for their physician. Recommendations from friends or other physicians was the most common (66.4%) way for patients to find their physicians, while 12.2% utilized online rating sites during their search. Optimizing patient experiences in the office may include keeping wait times to <30 minutes and educating patients on the roles of physician extenders. More work needs to be done to further elucidate variables influencing the subjective patient experience with their orthopedic care.
Continue to: Patient satisfaction...
Patient satisfaction has become an important focus in the rapidly changing healthcare environment due to the significant impact it has on healthcare delivery, healthcare economics, assessment of the quality of care, development of patient-care models, and quality improvement initiatives.1-4 Historically, the quality of care was measured by objective metrics such as complication rates, range-of-motion, and the provider’s expert opinion on the outcome. While those metrics are still impactful variables when defining a successful outcome, the medical community is now increasingly recognizing the importance of patients’ perspectives when defining successful treatments. Patient satisfaction is now highly regarded by clinicians and the government when considering outcomes and is even being incorporated into determining the value of care. Under the Affordable Care Act, patients assumed a more active role in clinical decision-making as well as in creating quality and efficiency initiatives.5,6 By 2017, 2% of the United States government’s Medicare payments will be redistributed among hospitals and physicians based on their quality and efficiency metrics, which are largely determined by patients’ evaluations of care.7 As a result, there has been significant interest in identifying variables influencing patient satisfaction and subjective outcomes.8,9
Patient satisfaction is related to both the outcomes of care and the process of care. As first described by Donabedian,10patients may be satisfied with the successful outcome of their care, but dissatisfied with how they received their care. The process of care is complex and considers many aspects of healthcare delivery, including time, cost, healthcare provider interactions, and burdens faced. While patient satisfaction with outcomes and process of care are heavily related, they should be regarded separately. It is essential that providers understand what variables are important to patients with regards to how they experience healthcare and choose their provider, especially surrounding elective procedures such as hip and knee arthroplasty.11,12
Within orthopedic surgery, patient satisfaction scores are beginning to be incorporated as part of the standard-of-care quality metrics obtained along with patient-reported outcome measures (PROMs) at defined time points postoperatively. Furthermore, PROMs and patient satisfaction data are becoming an increasingly important component of medical decision-making.13-16 Several authors have reported that increased patient satisfaction is correlated with increased compliance, improved treatment outcomes across numerous medical settings, including orthopedics, decreased risk of litigation, and higher patient ratings of the quality of care.17,18 Various factors, including meeting of expectations, staff politeness, the communication skills of the surgeon, and waiting times, have been suggested to influence eventual patient satisfaction within the surgical literature.19-21 However, within orthopedic surgery there is a paucity of investigations evaluating how patients determine preferences and satisfaction with the process of care.
The purpose of this study is to determine what preferences, if any, patients have when selecting their physician and how they experience care in an outpatient orthopedic setting. The authors hypothesize that the majority of patients find their physicians through online rating sites or recommendations from family and friends. The authors believe that patients expect to be seen in <30 minutes and will be unsatisfied overall with the amount of time that they spend with their physician.
Continue to: METHODS...
METHODS
The senior author (BRL) and a research team created a 15-question survey to evaluate patient preferences regarding the demographic characteristics (eg, age, gender, ethnicity) of their physician, wait times in a waiting room, time spent with the physician, care received from physician extenders (eg, nurse practitioners, physician assistants), and how they learned of their physician (Appendix). An a priori power analysis was conducted to determine that approximately 200 patients were needed for inclusion.11,22 Following Institutional Review Board approval (ORA 15051104), the survey was administered to 212 patients in a single-surgeon, adult reconstruction clinic. The survey was digitally administered on a touch-screen tablet using an electronic independent third party survey center (SurveyMonkey Inc) devoid of any identifying data. The survey was offered to all patients >21 years of age who were English-speaking and in the common area as patients waiting to be seen, from June 2015 to March 2016. A research assistant approached patients in the waiting room and asked if they would like to participate in a short survey regarding what factors influence the patient-physician relationship from the patient’s perspective.
Appendix 1
- Do you wish to partake in this 3-minute survey?
- Have you had a prior knee or hip replacement?
- What is your age?
- 30-40 years
- 40-50 years
- 50-60 years
- 60-70 years
- 70-80 years
- 80+ years
- What is your gender?
- Which of the following best represents your racial or ethnic heritage?
- African American
- How much time would you like the doctor to spend talking to you on a routine visit?
- 0-5 minutes
- 5-10 minutes
- 10-15 minutes
- 15-20 minutes
- 20-30 minutes
- >30 minutes
- How long is too long to wait to see the doctor?
- 10 minutes
- 20 minutes
- 30 minutes
- 40 minutes
- 50 minutes
- An hour or more
- If you were to only see a physician’s assistant or nurse practitioner at your follow-up visit and not the doctor, would you feel like you were getting below average care?
- Overall I am satisfied with the time the doctor spends with me.
- If you were to need a major surgery, would you want the physician to tell you what he or she would do if they were in your shoes?
- Would you prefer your doctor to be the same race/ethnicity as you?
- No
- No Preference
- Would you feel more comfortable with a male as opposed to a female orthopedic surgeon?
- Would you feel more comfortable with a female as opposed to a male orthopedic surgeon?
- What age would you like your physician to be?
- 25-35 years old
- 35-45 years old
- 45-55 years old
- 55-65 years old
- 65 years and older
- No preference
- How do you usually find your physician?
- Friends’ recommendations
- Healthcare provider’s recommendations
- Insurance plans
- Online research/ratings
- Other
Descriptive statistics were used to analyze subject demographics and survey responses. Chi-square analyses and multinomial logistic regressions were utilized to compare responses. All statistical analyses were conducted using SPSS version 24.0 software (SPSS Inc). Statistical significance was set at P < 0.05.
RESULTS
Of the 212 patients who were invited to participate, 196 patients (92.4%) agreed and completed the survey. Demographic and surgical history information can be found in Table 1. The majority of patients were female (62%) and above the age of 50 years (92.4%). Almost half (48.5%) of patients had a prior hip or knee replacement.
Table 1. Survey Respondent Demographics
| Number | Percent |
Age Range | ||
30-40 years | 4 | 2.0% |
40-50 years | 11 | 5.6% |
50-60 years | 47 | 24.0% |
60-70 years | 84 | 42.9% |
70-80 years | 41 | 20.9% |
>80 years | 9 | 4.6% |
Gender | ||
Male | 74 | 37.8% |
Female | 122 | 62.2% |
Ethnicity | ||
African American | 39 | 19.9% |
Asian | 3 | 1.5% |
Caucasian | 140 | 71.4% |
Hispanic | 10 | 5.1% |
Other | 4 | 2.0% |
Prior knee or hip replacement | ||
Yes | 95 | 48.5% |
No | 55 | 28.1% |
No Response | 46 | 23.5% |
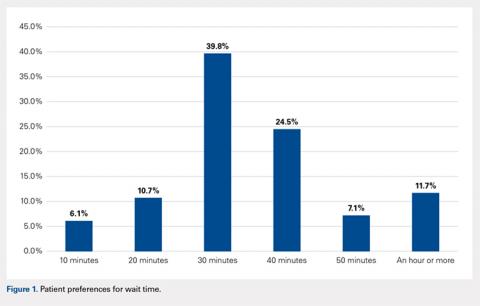
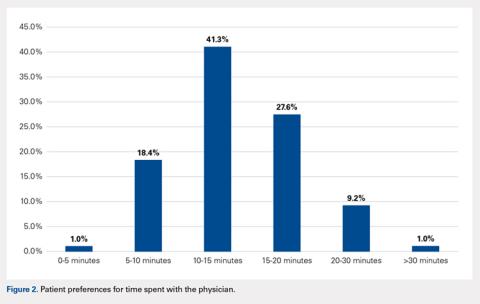
When asked how long is too long to wait to see the doctor, 30 minutes (39.8%) was most commonly selected, followed by 40 minutes (24.5%) (Figure 1). When asked how much time patients would like the doctor to spend with them during an office visit, the majority (68.9%) selected either 10 to 15 minutes (41.3%) or 15 to 20 minutes (27.6%) (Figure 2). The majority of patients (92.3%) were satisfied with the amount of time the doctor spent with them. In addition, 94.9% of respondents would want their doctor to tell them what they would do if they were in the patient’s shoes when making decisions regarding their medical care (Table 2). Less than half of respondents (41.8%) believe that seeing a physician extender (eg, nurse practitioner or physician assistant) at a postoperative visit would result in a lower quality of care (Table 2).
Table 2. Responses to Survey Questions
If you were to only see a physician's assistant or nurse practitioner at your follow-up visit and not the doctor, would you feel like you were getting below average care? | ||
Answer choices | Number | Percent |
No | 114 | 58.2% |
Yes | 82 | 41.8% |
If you were to need a major surgery would you want the physician to tell you what he or she would do if they were in your shoes? | ||
Answer choices | Number | Percent |
No | 10 | 5.1% |
Yes | 186 | 94.9% |
Would you prefer your doctor to be the same race/ethnicity as you? | ||
Answer choices | Number | Percent |
No | 29 | 14.8% |
Yes | 3 | 1.5% |
No Preference | 164 | 83.7% |
When asked if patients preferred a doctor of the same race/ethnicity, the vast majority (83.7%) had no preference (Table 2). There was no significant difference found between male and female respondents when asked if they would feel more comfortable with a male as opposed to a female orthopedic surgeon (P = .118) and vice versa (P = .604) (Table 3). Most patients preferred a physician between the ages of 45 and 55 years (39.3%), followed by 35 and 45 years (23.0%); however, this preference was not statistically significant (Table 4). Older patients were more likely to prefer younger physicians (odds ratio, 4.612 for 25-35 years of age; odds ratio, 1.328 for 35-45 years of age). Only 12.2% of patients reported online research/rating sites as the main resource utilized when selecting their physician (Figure 3). The majority (68.4%) reported that recommendations from either friends (35.2%) or healthcare providers (33.2%) were the main avenues through which they found their physicians.
Table 3. Overall Responses to Questions Regarding Male and Female Orthopedic Surgeonsa
Would you feel more comfortable with a male as opposed to a female orthopedic surgeon? | |||||
Answer choices | Number | Percent | Female responses | Male responses | P value |
No | 164 | 83.7% | 106 (86.9%) | 58 (78.4%) | 0.118 |
Yes | 32 | 16.3% | 16 (13.1%) | 16 (21.6%) |
|
Would you feel more comfortable with a female as opposed to a male orthopedic surgeon? | |||||
Answer choices | Number | Percent | Female responses | Male responses | P value |
No | 186 | 94.9% | 115 (94.3%) | 71 (95.9%) | 0.604 |
Yes | 10 | 5.1% | 7 (5.7%) | 3 (4.1%) |
|
aResponses were broken down by gender and compared utilizing a 2 x 2 chi-square analysis to test for significant differences in respondents’ gender preferences for their orthopedic surgeon.
Table 4. Patient Preferences Regarding Physician Age
What age would you like your physician to be? |
| 95% Confidence Interval | ||||
Answer Choices | Number or Responses | Percent | P value | Exp(β) | Lower Bound | Upper Bound |
25-35 years | 1 | 0.5% | 0.217 | 4.612 | 0.407 | 52.283 |
35-45 years | 45 | 23.0% | 0.161 | 1.328 | 0.893 | 1.975 |
45-55 years | 77 | 39.3% | 0.159 | 1.276 | 0.909 | 1.791 |
55-65 years | 9 | 4.6% | 0.483 | 1.302 | 0.624 | 2.717 |
≥65 years | 2 | 1.0% | 0.272 | 0.491 | 0.138 | 1.748 |
No preferencea | 62 | 31.6% | Reference | |||
aNo preference was used as the reference category for the answer choices, while the age bracket “>80 years” was used as the reference for the age of respondent variable.
Continue to: DISCUSSION...
DISCUSSION
The results of this study demonstrate that patients have several expectations and preferences with regards to the care they receive from physicians in the office. Patients prefer to wait <30 minutes before seeing their provider and desire only 10 to 20 minutes with their doctor. Patients do not have specific preferences with regards to the gender or ethnicity of their physician but would prefer a physician in the middle of their career, aged 45 to 55 years. Ultimately, patients do believe that seeing a physician at a postoperative visit is important, as just under half of patients thought that seeing a physician extender alone at a postoperative visit resulted in a lower quality of care.
While these results were obtained in a population specifically seeking the care of an orthopedic adult reconstruction surgeon, the results demonstrate that patients do not necessarily desire an unreasonable amount of time with their doctor. Patients simply want to be seen in a timely fashion and receive the full undivided attention of their doctor for approximately 20 minutes. Similarly, Patterson and colleagues22 found, in their series of 182 patients who presented to an orthopedic surgeon, that there was a significant correlation between time spent with the surgeon and overall patient satisfaction. Interestingly, the authors reported that patient satisfaction was not correlated with education level, sex, marital status, whether the patients were evaluated by a resident physician before seeing the attending surgeon, self-reported mental status, tobacco usage, the type of clinic visit, or the waiting time to see the surgeon (average, about 40 minutes for this cohort).22 Similarly, Teunis and colleagues23 reported an average 32-minute wait time in 81 patients presenting for care at an orthopedic hand clinic and demonstrated that a longer wait time was associated with decreased patient satisfaction. These results corroborate the findings of this study that a short wait time is important to patients when evaluating the process of care. Additionally, patients do not have unreasonable expectations with regards to the amount of time they would like to spend with the physician. A physician who has a clinic for 9 hours a day would thus be able to see 54 patients and still spend at least 10 minutes with each patient. The quality of the physician-patient interaction is likely more important than the actual amount of time spent; however, based on this study, patients do have certain expectations about how much time physicians should spend with them.
There were no significant sex, age, or ethnicity preferences in our specific patient cohort. However, a sizable percentage of respondents, 41.8%, believed that they were receiving inferior care if they only saw a physician extender at a routine follow-up visit. Many orthopedic surgeons rely on the care provided by physician extenders to enable them to see additional patients. Physician extenders are well trained to provide high-quality care, including at routine postoperative visits. The results of this study, that many patients believe physician extenders provide lower-quality care, may be a result of inadequate patient education regarding the extensive training and education physician extenders undergo. Physician extenders are qualified, licensed healthcare professionals who are playing increasingly important roles within orthopedics and medicine as a whole. As the demand for orthopedic surgeons to see more patients increases, so does the role of physician extenders. Future research is warranted into educating the public regarding the importance of these healthcare providers and the adequacy of their training.
While many practices now routinely obtain patient satisfaction scores, another modality through which patients can express their satisfaction and experiences with healthcare providers is through online internet physician rating sites (IPRS). These sites have exploded in number and popularity in recent years and, according to some studies, have a very real effect on provider selection.24 Interestingly, a low percentage of patients in this study utilized IPRS reviews to find their doctors. In a recent prospective survey study of 1000 consecutive patients presenting for care at the Mayo Clinic, Burkle and Keegan24 reported that 27% of patients would choose not to see a physician based on a negative IPRS review. Interestingly, only 1.0% of patients reported finding their doctor through advertising. Numerous authors have recently addressed advertising in orthopedic surgery, specifically direct-to-consumer marking, including the influence of physician self-promotion on patients.25,26 Specifically, Halawi and Barsoum26 discussed how direct-to-consumer marketing is commonly disseminated to the public through television and print advertisements, which are modalities more commonly utilized by older generations. However, many advertising agencies are moving toward internet-based advertising, especially through orthopedic group and individual surgeon websites for self-promoting advertisement, as approximately 75% of Americans use the internet for health-related information.25,27 The fact that many patients in this study did not utilize IPRS reviews or advertising (much of which is electronic) may be a result of the older, less internet-centric demographic that is often seen in an adult reconstruction clinic. Future research is warranted to determine what demographic of patients value IPRS reviews and how those reviews influence physician selection and the patient experience.
There are several limitations to this study. First, the majority of the surveyed population was Caucasian, and our results may not be equally reflective of diverse ethnic backgrounds. Second, the cohort size, while based on previous studies conducted in a similar fashion, may be underpowered to detect significant differences for 1 or more of these questions. In addition, having a question regarding the patient’s medical background or experiences may have provided further insight as to why patients selected the answers that they did. Furthermore, questions regarding the patient’s education level, religious background, and income brackets may have provided further context in which to evaluate their responses. These questions were omitted in an effort to keep the questionnaire at a length that would maximize enrollment and prevent survey fatigue. Future research is warranted to determine what patient-specific, injury/symptom-specific, and treatment-specific variables influence the subjective patient experience.
CONCLUSION
The vast majority of patients desire only 10 to 20 minutes with their doctor and are highly satisfied with the amount of time their surgeon spends with them. Patients reported no significant gender- or ethnicity-based preferences for their doctor. The majority of patients believe that a wait time exceeding 30 minutes is too long. A greater effort needs to be made to educate patients and the public about the significant and effective roles nurse practitioners and physician assistants can play within the healthcare system. While this cohort did not report notable utilization of IPRS reviews, it remains essential to understand what factors influence patients’ subjective experiences with their providers to ensure that patients achieve their desired outcomes, and report as such on these websites as they continue to gain popularity. Diminishing clinic wait times and understanding patient preferences may lead to a greater percentage of “satisfied” patients. While the majority of focus has been and will likely continue to be on improving patients’ satisfaction with their outcomes, more work needs to be done focusing specifically on the process through which outcomes are achieved.
1. Kocher MS, Steadman JR, Briggs K, Zurakowski D, Sterett WI, Hawkins RJ. Determinants of patient satisfaction with outcome after anterior cruciate ligament reconstruction. J Bone Joint Surg Am. 2002;84–A(9):1560-1572.
2. Carr-Hill RA. The measurement of patient satisfaction. J Public Health Med. 1992;14(3):236-249.
3. Ross CK, Steward CA, Sinacore JM. A comparative study of seven measures of patient satisfaction. Med Care. 1995;33(4):392-406. doi:10.1097/00005650-199504000-00006.
4. Strasser S, Aharony L, Greenberger D. The patient satisfaction process: moving toward a comprehensive model. Med Care Rev. 1993;50(2):219-248. doi:10.1177/107755879305000205.
5. Bozic KJ. Orthopaedic healthcare worldwide: shared medical decision making in orthopaedics. Clin Orthop Relat Res. 2013;471(5):1412-1414. doi:10.1007/s11999-013-2838-5.
6. Youm J, Chenok KE, Belkora J, Chiu V, Bozic KJ. The emerging case for shared decision making in orthopaedics. Instr Course Lect. 2013;62:587-594. doi:10.2106/00004623-201210170-00011.
7. Blumenthal D, Abrams M, Nuzum R. The affordable CARE Act at 5 years. N Engl J Med. 2015;373(16):1580. doi:10.1056/NEJMc1510015.
8. Shirley ED, Sanders JO. Patient satisfaction: implications and predictors of success. J Bone Joint Surg Am. 2013;95(10):e69. doi:10.2106/JBJS.L.01048.
9. Morris BJ, Jahangir AA, Sethi MK. Patient satisfaction: an emerging health policy issue. AAOS Now Web site. http://www.aaos.org/AAOSNow/2013/Jun/advocacy/advocacy5/?ssopc=1. Published June 2013. Accessed November 19, 2016.
10. Donabedian A. The quality of care. How can it be assessed? JAMA. 1988;260(12):1743-1748. doi:10.1001/jama.260.12.1743.
11. Bozic KJ, Kaufman D, Chan VC, Caminiti S, Lewis C. Factors that influence provider selection for elective total joint arthroplasty. Clin Orthop Relat Res. 2013;471(6):1865-1872. doi:10.1007/s11999-012-2640-9.
12. Davies AR, Ware JE Jr. Involving consumers in quality of care assessment. Health Aff (Millwood). 1988;7(1):33-48.
13. Black N, Burke L, Forrest CB, et al. Patient-reported outcomes: pathways to better health, better services, and better societies. Qual Life Res. 2016;25(5):1103-1112. doi:10.1007/s11136-015-1168-3.
14. Gilbert A, Sebag-Montefiore D, Davidson S, Velikova G. Use of patient-reported outcomes to measure symptoms and health related quality of life in the clinic. Gynecol Oncol. 2015;136(3):429-439. doi:10.1016/j.ygyno.2014.11.071.
15. Van Der Wees PJ, Nijhuis-Van Der Sanden MW, Ayanian JZ, Black N, Westert GP, Schneider EC. Integrating the use of patient-reported outcomes for both clinical practice and performance measurement: views of experts from 3 countries. Milbank Q. 2014;92(4):754-775. doi:10.1111/1468-0009.12091.
16. Franklin PD, Lewallen D, Bozic K, Hallstrom B, Jiranek W, Ayers DC. Implementation of patient-reported outcome measures in U.S. Total joint replacement registries: rationale, status, and plans. J Bone Joint Surg Am. 2014;96(Suppl 1):104-109. doi:10.2106/JBJS.N.00328.
17. Williams B. Patient satisfaction: a valid concept? Soc Sci Med. 1994;38(4):509-516. doi:10.1016/0277-9536(94)90247-X.
18. Hickson GB, Clayton EW, Entman SS, et al. Obstetricians' prior malpractice experience and patients' satisfaction with care. JAMA. 1994;272(20):1583-1587. doi:10.1001/jama.1994.03520200039032.
19. Larsson BW, Larsson G, Chantereau MW, von Holstein KS. International comparisons of patients' views on quality of care. Int J Health Care Qual Assur Inc Leadersh Health Serv. 2005;18(1):62-73. doi:10.1108/09526860510576974.
20. McLafferty RB, Williams RG, Lambert AD, Dunnington GL. Surgeon communication behaviors that lead patients to not recommend the surgeon to family members or friends: analysis and impact. Surgery. 2006;140(4):616-624. doi:https://doi.org/10.1016/j.surg.2006.06.021.
21. Mira JJ, Tomás O, Virtudes-Pérez M, Nebot C, Rodríguez-Marín J. Predictors of patient satisfaction in surgery. Surgery. 2009;145(5):536-541. doi:10.1016/j.surg.2009.01.012.
22. Patterson BM, Eskildsen SM, Clement RC, et al. Patient satisfaction is associated with time with provider but not clinic wait time among orthopedic patients. Orthopedics. 2017;40(1):43-48. doi:10.3928/01477447-20161013-05.
23. Teunis T, Thornton ER, Jayakumar P, Ring D. Time seeing a hand surgeon is not associated With patient satisfaction. Clin Orthop Relat Res. 2015;473(7):2362-2368. doi:10.1007/s11999-014-4090-z.
24. Burkle CM, Keegan MT. Popularity of internet physician rating sites and their apparent influence on patients' choices of physicians. BMC Health Serv Res. 2015;15:416. doi:10.1186/s12913-015-1099-2.
25. Mohney S, Lee DJ, Elfar JC. The effect of orthopedic advertising and self-promotion on a naive population. Am J Orthop. 2016;45(4):E227-E232.
26. Halawi MJ, Barsoum WK. Direct-to-consumer marketing: implications for patient care and orthopedic education. Am J Orthop. 2016;45(6):E335-E336.
27. Mostaghimi A, Crotty BH, Landon BE. The availability and nature of physician information on the internet. J Gen Intern Med. 2010;25(11):1152-1156. doi:10.1007/s11606-010-1425-7.
ABSTRACT
Patient satisfaction has become a topic of interest within orthopedics as the landscape of provider reimbursement continues to evolve to reward value of care. Online internet physician rating sites are becoming increasingly popular ways for patients to subjectively express their provider experience. Understanding what patients value during their episode of care is important in the modern healthcare environment. The purpose of this study is to determine what preferences, if any, patients have when selecting their physician and how they experience care in an outpatient orthopedic setting. A prospective survey was electronically administered to 212 patients in an adult reconstruction clinic. One hundred ninety-six patients (92.5%) completed the survey. Demographic questions regarding age, sex, ethnicity, and prior adult reconstruction surgical history were obtained. When patients were asked how much time they would like the doctor to spend with them on a routine visit, the most common answer was 10 to 15 minutes (41.3%), with only 10.2% patients desiring >20 minutes. The majority of patients (83.1%) believe ≥30 minutes is too long to wait to see their surgeon. Less than half of patients (41.8%) stated that they would feel as though they were receiving below average care if seen only by a nurse practitioner or physician’s assistant at a postoperative visit. Patients reported no significant age, gender, or ethnicity preferences for their physician. Recommendations from friends or other physicians was the most common (66.4%) way for patients to find their physicians, while 12.2% utilized online rating sites during their search. Optimizing patient experiences in the office may include keeping wait times to <30 minutes and educating patients on the roles of physician extenders. More work needs to be done to further elucidate variables influencing the subjective patient experience with their orthopedic care.
Continue to: Patient satisfaction...
Patient satisfaction has become an important focus in the rapidly changing healthcare environment due to the significant impact it has on healthcare delivery, healthcare economics, assessment of the quality of care, development of patient-care models, and quality improvement initiatives.1-4 Historically, the quality of care was measured by objective metrics such as complication rates, range-of-motion, and the provider’s expert opinion on the outcome. While those metrics are still impactful variables when defining a successful outcome, the medical community is now increasingly recognizing the importance of patients’ perspectives when defining successful treatments. Patient satisfaction is now highly regarded by clinicians and the government when considering outcomes and is even being incorporated into determining the value of care. Under the Affordable Care Act, patients assumed a more active role in clinical decision-making as well as in creating quality and efficiency initiatives.5,6 By 2017, 2% of the United States government’s Medicare payments will be redistributed among hospitals and physicians based on their quality and efficiency metrics, which are largely determined by patients’ evaluations of care.7 As a result, there has been significant interest in identifying variables influencing patient satisfaction and subjective outcomes.8,9
Patient satisfaction is related to both the outcomes of care and the process of care. As first described by Donabedian,10patients may be satisfied with the successful outcome of their care, but dissatisfied with how they received their care. The process of care is complex and considers many aspects of healthcare delivery, including time, cost, healthcare provider interactions, and burdens faced. While patient satisfaction with outcomes and process of care are heavily related, they should be regarded separately. It is essential that providers understand what variables are important to patients with regards to how they experience healthcare and choose their provider, especially surrounding elective procedures such as hip and knee arthroplasty.11,12
Within orthopedic surgery, patient satisfaction scores are beginning to be incorporated as part of the standard-of-care quality metrics obtained along with patient-reported outcome measures (PROMs) at defined time points postoperatively. Furthermore, PROMs and patient satisfaction data are becoming an increasingly important component of medical decision-making.13-16 Several authors have reported that increased patient satisfaction is correlated with increased compliance, improved treatment outcomes across numerous medical settings, including orthopedics, decreased risk of litigation, and higher patient ratings of the quality of care.17,18 Various factors, including meeting of expectations, staff politeness, the communication skills of the surgeon, and waiting times, have been suggested to influence eventual patient satisfaction within the surgical literature.19-21 However, within orthopedic surgery there is a paucity of investigations evaluating how patients determine preferences and satisfaction with the process of care.
The purpose of this study is to determine what preferences, if any, patients have when selecting their physician and how they experience care in an outpatient orthopedic setting. The authors hypothesize that the majority of patients find their physicians through online rating sites or recommendations from family and friends. The authors believe that patients expect to be seen in <30 minutes and will be unsatisfied overall with the amount of time that they spend with their physician.
Continue to: METHODS...
METHODS
The senior author (BRL) and a research team created a 15-question survey to evaluate patient preferences regarding the demographic characteristics (eg, age, gender, ethnicity) of their physician, wait times in a waiting room, time spent with the physician, care received from physician extenders (eg, nurse practitioners, physician assistants), and how they learned of their physician (Appendix). An a priori power analysis was conducted to determine that approximately 200 patients were needed for inclusion.11,22 Following Institutional Review Board approval (ORA 15051104), the survey was administered to 212 patients in a single-surgeon, adult reconstruction clinic. The survey was digitally administered on a touch-screen tablet using an electronic independent third party survey center (SurveyMonkey Inc) devoid of any identifying data. The survey was offered to all patients >21 years of age who were English-speaking and in the common area as patients waiting to be seen, from June 2015 to March 2016. A research assistant approached patients in the waiting room and asked if they would like to participate in a short survey regarding what factors influence the patient-physician relationship from the patient’s perspective.
Appendix 1
- Do you wish to partake in this 3-minute survey?
- Have you had a prior knee or hip replacement?
- What is your age?
- 30-40 years
- 40-50 years
- 50-60 years
- 60-70 years
- 70-80 years
- 80+ years
- What is your gender?
- Which of the following best represents your racial or ethnic heritage?
- African American
- How much time would you like the doctor to spend talking to you on a routine visit?
- 0-5 minutes
- 5-10 minutes
- 10-15 minutes
- 15-20 minutes
- 20-30 minutes
- >30 minutes
- How long is too long to wait to see the doctor?
- 10 minutes
- 20 minutes
- 30 minutes
- 40 minutes
- 50 minutes
- An hour or more
- If you were to only see a physician’s assistant or nurse practitioner at your follow-up visit and not the doctor, would you feel like you were getting below average care?
- Overall I am satisfied with the time the doctor spends with me.
- If you were to need a major surgery, would you want the physician to tell you what he or she would do if they were in your shoes?
- Would you prefer your doctor to be the same race/ethnicity as you?
- No
- No Preference
- Would you feel more comfortable with a male as opposed to a female orthopedic surgeon?
- Would you feel more comfortable with a female as opposed to a male orthopedic surgeon?
- What age would you like your physician to be?
- 25-35 years old
- 35-45 years old
- 45-55 years old
- 55-65 years old
- 65 years and older
- No preference
- How do you usually find your physician?
- Friends’ recommendations
- Healthcare provider’s recommendations
- Insurance plans
- Online research/ratings
- Other
Descriptive statistics were used to analyze subject demographics and survey responses. Chi-square analyses and multinomial logistic regressions were utilized to compare responses. All statistical analyses were conducted using SPSS version 24.0 software (SPSS Inc). Statistical significance was set at P < 0.05.
RESULTS
Of the 212 patients who were invited to participate, 196 patients (92.4%) agreed and completed the survey. Demographic and surgical history information can be found in Table 1. The majority of patients were female (62%) and above the age of 50 years (92.4%). Almost half (48.5%) of patients had a prior hip or knee replacement.
Table 1. Survey Respondent Demographics
| Number | Percent |
Age Range | ||
30-40 years | 4 | 2.0% |
40-50 years | 11 | 5.6% |
50-60 years | 47 | 24.0% |
60-70 years | 84 | 42.9% |
70-80 years | 41 | 20.9% |
>80 years | 9 | 4.6% |
Gender | ||
Male | 74 | 37.8% |
Female | 122 | 62.2% |
Ethnicity | ||
African American | 39 | 19.9% |
Asian | 3 | 1.5% |
Caucasian | 140 | 71.4% |
Hispanic | 10 | 5.1% |
Other | 4 | 2.0% |
Prior knee or hip replacement | ||
Yes | 95 | 48.5% |
No | 55 | 28.1% |
No Response | 46 | 23.5% |
When asked how long is too long to wait to see the doctor, 30 minutes (39.8%) was most commonly selected, followed by 40 minutes (24.5%) (Figure 1). When asked how much time patients would like the doctor to spend with them during an office visit, the majority (68.9%) selected either 10 to 15 minutes (41.3%) or 15 to 20 minutes (27.6%) (Figure 2). The majority of patients (92.3%) were satisfied with the amount of time the doctor spent with them. In addition, 94.9% of respondents would want their doctor to tell them what they would do if they were in the patient’s shoes when making decisions regarding their medical care (Table 2). Less than half of respondents (41.8%) believe that seeing a physician extender (eg, nurse practitioner or physician assistant) at a postoperative visit would result in a lower quality of care (Table 2).
Table 2. Responses to Survey Questions
If you were to only see a physician's assistant or nurse practitioner at your follow-up visit and not the doctor, would you feel like you were getting below average care? | ||
Answer choices | Number | Percent |
No | 114 | 58.2% |
Yes | 82 | 41.8% |
If you were to need a major surgery would you want the physician to tell you what he or she would do if they were in your shoes? | ||
Answer choices | Number | Percent |
No | 10 | 5.1% |
Yes | 186 | 94.9% |
Would you prefer your doctor to be the same race/ethnicity as you? | ||
Answer choices | Number | Percent |
No | 29 | 14.8% |
Yes | 3 | 1.5% |
No Preference | 164 | 83.7% |
When asked if patients preferred a doctor of the same race/ethnicity, the vast majority (83.7%) had no preference (Table 2). There was no significant difference found between male and female respondents when asked if they would feel more comfortable with a male as opposed to a female orthopedic surgeon (P = .118) and vice versa (P = .604) (Table 3). Most patients preferred a physician between the ages of 45 and 55 years (39.3%), followed by 35 and 45 years (23.0%); however, this preference was not statistically significant (Table 4). Older patients were more likely to prefer younger physicians (odds ratio, 4.612 for 25-35 years of age; odds ratio, 1.328 for 35-45 years of age). Only 12.2% of patients reported online research/rating sites as the main resource utilized when selecting their physician (Figure 3). The majority (68.4%) reported that recommendations from either friends (35.2%) or healthcare providers (33.2%) were the main avenues through which they found their physicians.
Table 3. Overall Responses to Questions Regarding Male and Female Orthopedic Surgeonsa
Would you feel more comfortable with a male as opposed to a female orthopedic surgeon? | |||||
Answer choices | Number | Percent | Female responses | Male responses | P value |
No | 164 | 83.7% | 106 (86.9%) | 58 (78.4%) | 0.118 |
Yes | 32 | 16.3% | 16 (13.1%) | 16 (21.6%) |
|
Would you feel more comfortable with a female as opposed to a male orthopedic surgeon? | |||||
Answer choices | Number | Percent | Female responses | Male responses | P value |
No | 186 | 94.9% | 115 (94.3%) | 71 (95.9%) | 0.604 |
Yes | 10 | 5.1% | 7 (5.7%) | 3 (4.1%) |
|
aResponses were broken down by gender and compared utilizing a 2 x 2 chi-square analysis to test for significant differences in respondents’ gender preferences for their orthopedic surgeon.
Table 4. Patient Preferences Regarding Physician Age
What age would you like your physician to be? |
| 95% Confidence Interval | ||||
Answer Choices | Number or Responses | Percent | P value | Exp(β) | Lower Bound | Upper Bound |
25-35 years | 1 | 0.5% | 0.217 | 4.612 | 0.407 | 52.283 |
35-45 years | 45 | 23.0% | 0.161 | 1.328 | 0.893 | 1.975 |
45-55 years | 77 | 39.3% | 0.159 | 1.276 | 0.909 | 1.791 |
55-65 years | 9 | 4.6% | 0.483 | 1.302 | 0.624 | 2.717 |
≥65 years | 2 | 1.0% | 0.272 | 0.491 | 0.138 | 1.748 |
No preferencea | 62 | 31.6% | Reference | |||
aNo preference was used as the reference category for the answer choices, while the age bracket “>80 years” was used as the reference for the age of respondent variable.
Continue to: DISCUSSION...
DISCUSSION
The results of this study demonstrate that patients have several expectations and preferences with regards to the care they receive from physicians in the office. Patients prefer to wait <30 minutes before seeing their provider and desire only 10 to 20 minutes with their doctor. Patients do not have specific preferences with regards to the gender or ethnicity of their physician but would prefer a physician in the middle of their career, aged 45 to 55 years. Ultimately, patients do believe that seeing a physician at a postoperative visit is important, as just under half of patients thought that seeing a physician extender alone at a postoperative visit resulted in a lower quality of care.
While these results were obtained in a population specifically seeking the care of an orthopedic adult reconstruction surgeon, the results demonstrate that patients do not necessarily desire an unreasonable amount of time with their doctor. Patients simply want to be seen in a timely fashion and receive the full undivided attention of their doctor for approximately 20 minutes. Similarly, Patterson and colleagues22 found, in their series of 182 patients who presented to an orthopedic surgeon, that there was a significant correlation between time spent with the surgeon and overall patient satisfaction. Interestingly, the authors reported that patient satisfaction was not correlated with education level, sex, marital status, whether the patients were evaluated by a resident physician before seeing the attending surgeon, self-reported mental status, tobacco usage, the type of clinic visit, or the waiting time to see the surgeon (average, about 40 minutes for this cohort).22 Similarly, Teunis and colleagues23 reported an average 32-minute wait time in 81 patients presenting for care at an orthopedic hand clinic and demonstrated that a longer wait time was associated with decreased patient satisfaction. These results corroborate the findings of this study that a short wait time is important to patients when evaluating the process of care. Additionally, patients do not have unreasonable expectations with regards to the amount of time they would like to spend with the physician. A physician who has a clinic for 9 hours a day would thus be able to see 54 patients and still spend at least 10 minutes with each patient. The quality of the physician-patient interaction is likely more important than the actual amount of time spent; however, based on this study, patients do have certain expectations about how much time physicians should spend with them.
There were no significant sex, age, or ethnicity preferences in our specific patient cohort. However, a sizable percentage of respondents, 41.8%, believed that they were receiving inferior care if they only saw a physician extender at a routine follow-up visit. Many orthopedic surgeons rely on the care provided by physician extenders to enable them to see additional patients. Physician extenders are well trained to provide high-quality care, including at routine postoperative visits. The results of this study, that many patients believe physician extenders provide lower-quality care, may be a result of inadequate patient education regarding the extensive training and education physician extenders undergo. Physician extenders are qualified, licensed healthcare professionals who are playing increasingly important roles within orthopedics and medicine as a whole. As the demand for orthopedic surgeons to see more patients increases, so does the role of physician extenders. Future research is warranted into educating the public regarding the importance of these healthcare providers and the adequacy of their training.
While many practices now routinely obtain patient satisfaction scores, another modality through which patients can express their satisfaction and experiences with healthcare providers is through online internet physician rating sites (IPRS). These sites have exploded in number and popularity in recent years and, according to some studies, have a very real effect on provider selection.24 Interestingly, a low percentage of patients in this study utilized IPRS reviews to find their doctors. In a recent prospective survey study of 1000 consecutive patients presenting for care at the Mayo Clinic, Burkle and Keegan24 reported that 27% of patients would choose not to see a physician based on a negative IPRS review. Interestingly, only 1.0% of patients reported finding their doctor through advertising. Numerous authors have recently addressed advertising in orthopedic surgery, specifically direct-to-consumer marking, including the influence of physician self-promotion on patients.25,26 Specifically, Halawi and Barsoum26 discussed how direct-to-consumer marketing is commonly disseminated to the public through television and print advertisements, which are modalities more commonly utilized by older generations. However, many advertising agencies are moving toward internet-based advertising, especially through orthopedic group and individual surgeon websites for self-promoting advertisement, as approximately 75% of Americans use the internet for health-related information.25,27 The fact that many patients in this study did not utilize IPRS reviews or advertising (much of which is electronic) may be a result of the older, less internet-centric demographic that is often seen in an adult reconstruction clinic. Future research is warranted to determine what demographic of patients value IPRS reviews and how those reviews influence physician selection and the patient experience.
There are several limitations to this study. First, the majority of the surveyed population was Caucasian, and our results may not be equally reflective of diverse ethnic backgrounds. Second, the cohort size, while based on previous studies conducted in a similar fashion, may be underpowered to detect significant differences for 1 or more of these questions. In addition, having a question regarding the patient’s medical background or experiences may have provided further insight as to why patients selected the answers that they did. Furthermore, questions regarding the patient’s education level, religious background, and income brackets may have provided further context in which to evaluate their responses. These questions were omitted in an effort to keep the questionnaire at a length that would maximize enrollment and prevent survey fatigue. Future research is warranted to determine what patient-specific, injury/symptom-specific, and treatment-specific variables influence the subjective patient experience.
CONCLUSION
The vast majority of patients desire only 10 to 20 minutes with their doctor and are highly satisfied with the amount of time their surgeon spends with them. Patients reported no significant gender- or ethnicity-based preferences for their doctor. The majority of patients believe that a wait time exceeding 30 minutes is too long. A greater effort needs to be made to educate patients and the public about the significant and effective roles nurse practitioners and physician assistants can play within the healthcare system. While this cohort did not report notable utilization of IPRS reviews, it remains essential to understand what factors influence patients’ subjective experiences with their providers to ensure that patients achieve their desired outcomes, and report as such on these websites as they continue to gain popularity. Diminishing clinic wait times and understanding patient preferences may lead to a greater percentage of “satisfied” patients. While the majority of focus has been and will likely continue to be on improving patients’ satisfaction with their outcomes, more work needs to be done focusing specifically on the process through which outcomes are achieved.
ABSTRACT
Patient satisfaction has become a topic of interest within orthopedics as the landscape of provider reimbursement continues to evolve to reward value of care. Online internet physician rating sites are becoming increasingly popular ways for patients to subjectively express their provider experience. Understanding what patients value during their episode of care is important in the modern healthcare environment. The purpose of this study is to determine what preferences, if any, patients have when selecting their physician and how they experience care in an outpatient orthopedic setting. A prospective survey was electronically administered to 212 patients in an adult reconstruction clinic. One hundred ninety-six patients (92.5%) completed the survey. Demographic questions regarding age, sex, ethnicity, and prior adult reconstruction surgical history were obtained. When patients were asked how much time they would like the doctor to spend with them on a routine visit, the most common answer was 10 to 15 minutes (41.3%), with only 10.2% patients desiring >20 minutes. The majority of patients (83.1%) believe ≥30 minutes is too long to wait to see their surgeon. Less than half of patients (41.8%) stated that they would feel as though they were receiving below average care if seen only by a nurse practitioner or physician’s assistant at a postoperative visit. Patients reported no significant age, gender, or ethnicity preferences for their physician. Recommendations from friends or other physicians was the most common (66.4%) way for patients to find their physicians, while 12.2% utilized online rating sites during their search. Optimizing patient experiences in the office may include keeping wait times to <30 minutes and educating patients on the roles of physician extenders. More work needs to be done to further elucidate variables influencing the subjective patient experience with their orthopedic care.
Continue to: Patient satisfaction...
Patient satisfaction has become an important focus in the rapidly changing healthcare environment due to the significant impact it has on healthcare delivery, healthcare economics, assessment of the quality of care, development of patient-care models, and quality improvement initiatives.1-4 Historically, the quality of care was measured by objective metrics such as complication rates, range-of-motion, and the provider’s expert opinion on the outcome. While those metrics are still impactful variables when defining a successful outcome, the medical community is now increasingly recognizing the importance of patients’ perspectives when defining successful treatments. Patient satisfaction is now highly regarded by clinicians and the government when considering outcomes and is even being incorporated into determining the value of care. Under the Affordable Care Act, patients assumed a more active role in clinical decision-making as well as in creating quality and efficiency initiatives.5,6 By 2017, 2% of the United States government’s Medicare payments will be redistributed among hospitals and physicians based on their quality and efficiency metrics, which are largely determined by patients’ evaluations of care.7 As a result, there has been significant interest in identifying variables influencing patient satisfaction and subjective outcomes.8,9
Patient satisfaction is related to both the outcomes of care and the process of care. As first described by Donabedian,10patients may be satisfied with the successful outcome of their care, but dissatisfied with how they received their care. The process of care is complex and considers many aspects of healthcare delivery, including time, cost, healthcare provider interactions, and burdens faced. While patient satisfaction with outcomes and process of care are heavily related, they should be regarded separately. It is essential that providers understand what variables are important to patients with regards to how they experience healthcare and choose their provider, especially surrounding elective procedures such as hip and knee arthroplasty.11,12
Within orthopedic surgery, patient satisfaction scores are beginning to be incorporated as part of the standard-of-care quality metrics obtained along with patient-reported outcome measures (PROMs) at defined time points postoperatively. Furthermore, PROMs and patient satisfaction data are becoming an increasingly important component of medical decision-making.13-16 Several authors have reported that increased patient satisfaction is correlated with increased compliance, improved treatment outcomes across numerous medical settings, including orthopedics, decreased risk of litigation, and higher patient ratings of the quality of care.17,18 Various factors, including meeting of expectations, staff politeness, the communication skills of the surgeon, and waiting times, have been suggested to influence eventual patient satisfaction within the surgical literature.19-21 However, within orthopedic surgery there is a paucity of investigations evaluating how patients determine preferences and satisfaction with the process of care.
The purpose of this study is to determine what preferences, if any, patients have when selecting their physician and how they experience care in an outpatient orthopedic setting. The authors hypothesize that the majority of patients find their physicians through online rating sites or recommendations from family and friends. The authors believe that patients expect to be seen in <30 minutes and will be unsatisfied overall with the amount of time that they spend with their physician.
Continue to: METHODS...
METHODS
The senior author (BRL) and a research team created a 15-question survey to evaluate patient preferences regarding the demographic characteristics (eg, age, gender, ethnicity) of their physician, wait times in a waiting room, time spent with the physician, care received from physician extenders (eg, nurse practitioners, physician assistants), and how they learned of their physician (Appendix). An a priori power analysis was conducted to determine that approximately 200 patients were needed for inclusion.11,22 Following Institutional Review Board approval (ORA 15051104), the survey was administered to 212 patients in a single-surgeon, adult reconstruction clinic. The survey was digitally administered on a touch-screen tablet using an electronic independent third party survey center (SurveyMonkey Inc) devoid of any identifying data. The survey was offered to all patients >21 years of age who were English-speaking and in the common area as patients waiting to be seen, from June 2015 to March 2016. A research assistant approached patients in the waiting room and asked if they would like to participate in a short survey regarding what factors influence the patient-physician relationship from the patient’s perspective.
Appendix 1
- Do you wish to partake in this 3-minute survey?
- Have you had a prior knee or hip replacement?
- What is your age?
- 30-40 years
- 40-50 years
- 50-60 years
- 60-70 years
- 70-80 years
- 80+ years
- What is your gender?
- Which of the following best represents your racial or ethnic heritage?
- African American
- How much time would you like the doctor to spend talking to you on a routine visit?
- 0-5 minutes
- 5-10 minutes
- 10-15 minutes
- 15-20 minutes
- 20-30 minutes
- >30 minutes
- How long is too long to wait to see the doctor?
- 10 minutes
- 20 minutes
- 30 minutes
- 40 minutes
- 50 minutes
- An hour or more
- If you were to only see a physician’s assistant or nurse practitioner at your follow-up visit and not the doctor, would you feel like you were getting below average care?
- Overall I am satisfied with the time the doctor spends with me.
- If you were to need a major surgery, would you want the physician to tell you what he or she would do if they were in your shoes?
- Would you prefer your doctor to be the same race/ethnicity as you?
- No
- No Preference
- Would you feel more comfortable with a male as opposed to a female orthopedic surgeon?
- Would you feel more comfortable with a female as opposed to a male orthopedic surgeon?
- What age would you like your physician to be?
- 25-35 years old
- 35-45 years old
- 45-55 years old
- 55-65 years old
- 65 years and older
- No preference
- How do you usually find your physician?
- Friends’ recommendations
- Healthcare provider’s recommendations
- Insurance plans
- Online research/ratings
- Other
Descriptive statistics were used to analyze subject demographics and survey responses. Chi-square analyses and multinomial logistic regressions were utilized to compare responses. All statistical analyses were conducted using SPSS version 24.0 software (SPSS Inc). Statistical significance was set at P < 0.05.
RESULTS
Of the 212 patients who were invited to participate, 196 patients (92.4%) agreed and completed the survey. Demographic and surgical history information can be found in Table 1. The majority of patients were female (62%) and above the age of 50 years (92.4%). Almost half (48.5%) of patients had a prior hip or knee replacement.
Table 1. Survey Respondent Demographics
| Number | Percent |
Age Range | ||
30-40 years | 4 | 2.0% |
40-50 years | 11 | 5.6% |
50-60 years | 47 | 24.0% |
60-70 years | 84 | 42.9% |
70-80 years | 41 | 20.9% |
>80 years | 9 | 4.6% |
Gender | ||
Male | 74 | 37.8% |
Female | 122 | 62.2% |
Ethnicity | ||
African American | 39 | 19.9% |
Asian | 3 | 1.5% |
Caucasian | 140 | 71.4% |
Hispanic | 10 | 5.1% |
Other | 4 | 2.0% |
Prior knee or hip replacement | ||
Yes | 95 | 48.5% |
No | 55 | 28.1% |
No Response | 46 | 23.5% |
When asked how long is too long to wait to see the doctor, 30 minutes (39.8%) was most commonly selected, followed by 40 minutes (24.5%) (Figure 1). When asked how much time patients would like the doctor to spend with them during an office visit, the majority (68.9%) selected either 10 to 15 minutes (41.3%) or 15 to 20 minutes (27.6%) (Figure 2). The majority of patients (92.3%) were satisfied with the amount of time the doctor spent with them. In addition, 94.9% of respondents would want their doctor to tell them what they would do if they were in the patient’s shoes when making decisions regarding their medical care (Table 2). Less than half of respondents (41.8%) believe that seeing a physician extender (eg, nurse practitioner or physician assistant) at a postoperative visit would result in a lower quality of care (Table 2).
Table 2. Responses to Survey Questions
If you were to only see a physician's assistant or nurse practitioner at your follow-up visit and not the doctor, would you feel like you were getting below average care? | ||
Answer choices | Number | Percent |
No | 114 | 58.2% |
Yes | 82 | 41.8% |
If you were to need a major surgery would you want the physician to tell you what he or she would do if they were in your shoes? | ||
Answer choices | Number | Percent |
No | 10 | 5.1% |
Yes | 186 | 94.9% |
Would you prefer your doctor to be the same race/ethnicity as you? | ||
Answer choices | Number | Percent |
No | 29 | 14.8% |
Yes | 3 | 1.5% |
No Preference | 164 | 83.7% |
When asked if patients preferred a doctor of the same race/ethnicity, the vast majority (83.7%) had no preference (Table 2). There was no significant difference found between male and female respondents when asked if they would feel more comfortable with a male as opposed to a female orthopedic surgeon (P = .118) and vice versa (P = .604) (Table 3). Most patients preferred a physician between the ages of 45 and 55 years (39.3%), followed by 35 and 45 years (23.0%); however, this preference was not statistically significant (Table 4). Older patients were more likely to prefer younger physicians (odds ratio, 4.612 for 25-35 years of age; odds ratio, 1.328 for 35-45 years of age). Only 12.2% of patients reported online research/rating sites as the main resource utilized when selecting their physician (Figure 3). The majority (68.4%) reported that recommendations from either friends (35.2%) or healthcare providers (33.2%) were the main avenues through which they found their physicians.
Table 3. Overall Responses to Questions Regarding Male and Female Orthopedic Surgeonsa
Would you feel more comfortable with a male as opposed to a female orthopedic surgeon? | |||||
Answer choices | Number | Percent | Female responses | Male responses | P value |
No | 164 | 83.7% | 106 (86.9%) | 58 (78.4%) | 0.118 |
Yes | 32 | 16.3% | 16 (13.1%) | 16 (21.6%) |
|
Would you feel more comfortable with a female as opposed to a male orthopedic surgeon? | |||||
Answer choices | Number | Percent | Female responses | Male responses | P value |
No | 186 | 94.9% | 115 (94.3%) | 71 (95.9%) | 0.604 |
Yes | 10 | 5.1% | 7 (5.7%) | 3 (4.1%) |
|
aResponses were broken down by gender and compared utilizing a 2 x 2 chi-square analysis to test for significant differences in respondents’ gender preferences for their orthopedic surgeon.
Table 4. Patient Preferences Regarding Physician Age
What age would you like your physician to be? |
| 95% Confidence Interval | ||||
Answer Choices | Number or Responses | Percent | P value | Exp(β) | Lower Bound | Upper Bound |
25-35 years | 1 | 0.5% | 0.217 | 4.612 | 0.407 | 52.283 |
35-45 years | 45 | 23.0% | 0.161 | 1.328 | 0.893 | 1.975 |
45-55 years | 77 | 39.3% | 0.159 | 1.276 | 0.909 | 1.791 |
55-65 years | 9 | 4.6% | 0.483 | 1.302 | 0.624 | 2.717 |
≥65 years | 2 | 1.0% | 0.272 | 0.491 | 0.138 | 1.748 |
No preferencea | 62 | 31.6% | Reference | |||
aNo preference was used as the reference category for the answer choices, while the age bracket “>80 years” was used as the reference for the age of respondent variable.
Continue to: DISCUSSION...
DISCUSSION
The results of this study demonstrate that patients have several expectations and preferences with regards to the care they receive from physicians in the office. Patients prefer to wait <30 minutes before seeing their provider and desire only 10 to 20 minutes with their doctor. Patients do not have specific preferences with regards to the gender or ethnicity of their physician but would prefer a physician in the middle of their career, aged 45 to 55 years. Ultimately, patients do believe that seeing a physician at a postoperative visit is important, as just under half of patients thought that seeing a physician extender alone at a postoperative visit resulted in a lower quality of care.
While these results were obtained in a population specifically seeking the care of an orthopedic adult reconstruction surgeon, the results demonstrate that patients do not necessarily desire an unreasonable amount of time with their doctor. Patients simply want to be seen in a timely fashion and receive the full undivided attention of their doctor for approximately 20 minutes. Similarly, Patterson and colleagues22 found, in their series of 182 patients who presented to an orthopedic surgeon, that there was a significant correlation between time spent with the surgeon and overall patient satisfaction. Interestingly, the authors reported that patient satisfaction was not correlated with education level, sex, marital status, whether the patients were evaluated by a resident physician before seeing the attending surgeon, self-reported mental status, tobacco usage, the type of clinic visit, or the waiting time to see the surgeon (average, about 40 minutes for this cohort).22 Similarly, Teunis and colleagues23 reported an average 32-minute wait time in 81 patients presenting for care at an orthopedic hand clinic and demonstrated that a longer wait time was associated with decreased patient satisfaction. These results corroborate the findings of this study that a short wait time is important to patients when evaluating the process of care. Additionally, patients do not have unreasonable expectations with regards to the amount of time they would like to spend with the physician. A physician who has a clinic for 9 hours a day would thus be able to see 54 patients and still spend at least 10 minutes with each patient. The quality of the physician-patient interaction is likely more important than the actual amount of time spent; however, based on this study, patients do have certain expectations about how much time physicians should spend with them.
There were no significant sex, age, or ethnicity preferences in our specific patient cohort. However, a sizable percentage of respondents, 41.8%, believed that they were receiving inferior care if they only saw a physician extender at a routine follow-up visit. Many orthopedic surgeons rely on the care provided by physician extenders to enable them to see additional patients. Physician extenders are well trained to provide high-quality care, including at routine postoperative visits. The results of this study, that many patients believe physician extenders provide lower-quality care, may be a result of inadequate patient education regarding the extensive training and education physician extenders undergo. Physician extenders are qualified, licensed healthcare professionals who are playing increasingly important roles within orthopedics and medicine as a whole. As the demand for orthopedic surgeons to see more patients increases, so does the role of physician extenders. Future research is warranted into educating the public regarding the importance of these healthcare providers and the adequacy of their training.
While many practices now routinely obtain patient satisfaction scores, another modality through which patients can express their satisfaction and experiences with healthcare providers is through online internet physician rating sites (IPRS). These sites have exploded in number and popularity in recent years and, according to some studies, have a very real effect on provider selection.24 Interestingly, a low percentage of patients in this study utilized IPRS reviews to find their doctors. In a recent prospective survey study of 1000 consecutive patients presenting for care at the Mayo Clinic, Burkle and Keegan24 reported that 27% of patients would choose not to see a physician based on a negative IPRS review. Interestingly, only 1.0% of patients reported finding their doctor through advertising. Numerous authors have recently addressed advertising in orthopedic surgery, specifically direct-to-consumer marking, including the influence of physician self-promotion on patients.25,26 Specifically, Halawi and Barsoum26 discussed how direct-to-consumer marketing is commonly disseminated to the public through television and print advertisements, which are modalities more commonly utilized by older generations. However, many advertising agencies are moving toward internet-based advertising, especially through orthopedic group and individual surgeon websites for self-promoting advertisement, as approximately 75% of Americans use the internet for health-related information.25,27 The fact that many patients in this study did not utilize IPRS reviews or advertising (much of which is electronic) may be a result of the older, less internet-centric demographic that is often seen in an adult reconstruction clinic. Future research is warranted to determine what demographic of patients value IPRS reviews and how those reviews influence physician selection and the patient experience.
There are several limitations to this study. First, the majority of the surveyed population was Caucasian, and our results may not be equally reflective of diverse ethnic backgrounds. Second, the cohort size, while based on previous studies conducted in a similar fashion, may be underpowered to detect significant differences for 1 or more of these questions. In addition, having a question regarding the patient’s medical background or experiences may have provided further insight as to why patients selected the answers that they did. Furthermore, questions regarding the patient’s education level, religious background, and income brackets may have provided further context in which to evaluate their responses. These questions were omitted in an effort to keep the questionnaire at a length that would maximize enrollment and prevent survey fatigue. Future research is warranted to determine what patient-specific, injury/symptom-specific, and treatment-specific variables influence the subjective patient experience.
CONCLUSION
The vast majority of patients desire only 10 to 20 minutes with their doctor and are highly satisfied with the amount of time their surgeon spends with them. Patients reported no significant gender- or ethnicity-based preferences for their doctor. The majority of patients believe that a wait time exceeding 30 minutes is too long. A greater effort needs to be made to educate patients and the public about the significant and effective roles nurse practitioners and physician assistants can play within the healthcare system. While this cohort did not report notable utilization of IPRS reviews, it remains essential to understand what factors influence patients’ subjective experiences with their providers to ensure that patients achieve their desired outcomes, and report as such on these websites as they continue to gain popularity. Diminishing clinic wait times and understanding patient preferences may lead to a greater percentage of “satisfied” patients. While the majority of focus has been and will likely continue to be on improving patients’ satisfaction with their outcomes, more work needs to be done focusing specifically on the process through which outcomes are achieved.
1. Kocher MS, Steadman JR, Briggs K, Zurakowski D, Sterett WI, Hawkins RJ. Determinants of patient satisfaction with outcome after anterior cruciate ligament reconstruction. J Bone Joint Surg Am. 2002;84–A(9):1560-1572.
2. Carr-Hill RA. The measurement of patient satisfaction. J Public Health Med. 1992;14(3):236-249.
3. Ross CK, Steward CA, Sinacore JM. A comparative study of seven measures of patient satisfaction. Med Care. 1995;33(4):392-406. doi:10.1097/00005650-199504000-00006.
4. Strasser S, Aharony L, Greenberger D. The patient satisfaction process: moving toward a comprehensive model. Med Care Rev. 1993;50(2):219-248. doi:10.1177/107755879305000205.
5. Bozic KJ. Orthopaedic healthcare worldwide: shared medical decision making in orthopaedics. Clin Orthop Relat Res. 2013;471(5):1412-1414. doi:10.1007/s11999-013-2838-5.
6. Youm J, Chenok KE, Belkora J, Chiu V, Bozic KJ. The emerging case for shared decision making in orthopaedics. Instr Course Lect. 2013;62:587-594. doi:10.2106/00004623-201210170-00011.
7. Blumenthal D, Abrams M, Nuzum R. The affordable CARE Act at 5 years. N Engl J Med. 2015;373(16):1580. doi:10.1056/NEJMc1510015.
8. Shirley ED, Sanders JO. Patient satisfaction: implications and predictors of success. J Bone Joint Surg Am. 2013;95(10):e69. doi:10.2106/JBJS.L.01048.
9. Morris BJ, Jahangir AA, Sethi MK. Patient satisfaction: an emerging health policy issue. AAOS Now Web site. http://www.aaos.org/AAOSNow/2013/Jun/advocacy/advocacy5/?ssopc=1. Published June 2013. Accessed November 19, 2016.
10. Donabedian A. The quality of care. How can it be assessed? JAMA. 1988;260(12):1743-1748. doi:10.1001/jama.260.12.1743.
11. Bozic KJ, Kaufman D, Chan VC, Caminiti S, Lewis C. Factors that influence provider selection for elective total joint arthroplasty. Clin Orthop Relat Res. 2013;471(6):1865-1872. doi:10.1007/s11999-012-2640-9.
12. Davies AR, Ware JE Jr. Involving consumers in quality of care assessment. Health Aff (Millwood). 1988;7(1):33-48.
13. Black N, Burke L, Forrest CB, et al. Patient-reported outcomes: pathways to better health, better services, and better societies. Qual Life Res. 2016;25(5):1103-1112. doi:10.1007/s11136-015-1168-3.
14. Gilbert A, Sebag-Montefiore D, Davidson S, Velikova G. Use of patient-reported outcomes to measure symptoms and health related quality of life in the clinic. Gynecol Oncol. 2015;136(3):429-439. doi:10.1016/j.ygyno.2014.11.071.
15. Van Der Wees PJ, Nijhuis-Van Der Sanden MW, Ayanian JZ, Black N, Westert GP, Schneider EC. Integrating the use of patient-reported outcomes for both clinical practice and performance measurement: views of experts from 3 countries. Milbank Q. 2014;92(4):754-775. doi:10.1111/1468-0009.12091.
16. Franklin PD, Lewallen D, Bozic K, Hallstrom B, Jiranek W, Ayers DC. Implementation of patient-reported outcome measures in U.S. Total joint replacement registries: rationale, status, and plans. J Bone Joint Surg Am. 2014;96(Suppl 1):104-109. doi:10.2106/JBJS.N.00328.
17. Williams B. Patient satisfaction: a valid concept? Soc Sci Med. 1994;38(4):509-516. doi:10.1016/0277-9536(94)90247-X.
18. Hickson GB, Clayton EW, Entman SS, et al. Obstetricians' prior malpractice experience and patients' satisfaction with care. JAMA. 1994;272(20):1583-1587. doi:10.1001/jama.1994.03520200039032.
19. Larsson BW, Larsson G, Chantereau MW, von Holstein KS. International comparisons of patients' views on quality of care. Int J Health Care Qual Assur Inc Leadersh Health Serv. 2005;18(1):62-73. doi:10.1108/09526860510576974.
20. McLafferty RB, Williams RG, Lambert AD, Dunnington GL. Surgeon communication behaviors that lead patients to not recommend the surgeon to family members or friends: analysis and impact. Surgery. 2006;140(4):616-624. doi:https://doi.org/10.1016/j.surg.2006.06.021.
21. Mira JJ, Tomás O, Virtudes-Pérez M, Nebot C, Rodríguez-Marín J. Predictors of patient satisfaction in surgery. Surgery. 2009;145(5):536-541. doi:10.1016/j.surg.2009.01.012.
22. Patterson BM, Eskildsen SM, Clement RC, et al. Patient satisfaction is associated with time with provider but not clinic wait time among orthopedic patients. Orthopedics. 2017;40(1):43-48. doi:10.3928/01477447-20161013-05.
23. Teunis T, Thornton ER, Jayakumar P, Ring D. Time seeing a hand surgeon is not associated With patient satisfaction. Clin Orthop Relat Res. 2015;473(7):2362-2368. doi:10.1007/s11999-014-4090-z.
24. Burkle CM, Keegan MT. Popularity of internet physician rating sites and their apparent influence on patients' choices of physicians. BMC Health Serv Res. 2015;15:416. doi:10.1186/s12913-015-1099-2.
25. Mohney S, Lee DJ, Elfar JC. The effect of orthopedic advertising and self-promotion on a naive population. Am J Orthop. 2016;45(4):E227-E232.
26. Halawi MJ, Barsoum WK. Direct-to-consumer marketing: implications for patient care and orthopedic education. Am J Orthop. 2016;45(6):E335-E336.
27. Mostaghimi A, Crotty BH, Landon BE. The availability and nature of physician information on the internet. J Gen Intern Med. 2010;25(11):1152-1156. doi:10.1007/s11606-010-1425-7.
1. Kocher MS, Steadman JR, Briggs K, Zurakowski D, Sterett WI, Hawkins RJ. Determinants of patient satisfaction with outcome after anterior cruciate ligament reconstruction. J Bone Joint Surg Am. 2002;84–A(9):1560-1572.
2. Carr-Hill RA. The measurement of patient satisfaction. J Public Health Med. 1992;14(3):236-249.
3. Ross CK, Steward CA, Sinacore JM. A comparative study of seven measures of patient satisfaction. Med Care. 1995;33(4):392-406. doi:10.1097/00005650-199504000-00006.
4. Strasser S, Aharony L, Greenberger D. The patient satisfaction process: moving toward a comprehensive model. Med Care Rev. 1993;50(2):219-248. doi:10.1177/107755879305000205.
5. Bozic KJ. Orthopaedic healthcare worldwide: shared medical decision making in orthopaedics. Clin Orthop Relat Res. 2013;471(5):1412-1414. doi:10.1007/s11999-013-2838-5.
6. Youm J, Chenok KE, Belkora J, Chiu V, Bozic KJ. The emerging case for shared decision making in orthopaedics. Instr Course Lect. 2013;62:587-594. doi:10.2106/00004623-201210170-00011.
7. Blumenthal D, Abrams M, Nuzum R. The affordable CARE Act at 5 years. N Engl J Med. 2015;373(16):1580. doi:10.1056/NEJMc1510015.
8. Shirley ED, Sanders JO. Patient satisfaction: implications and predictors of success. J Bone Joint Surg Am. 2013;95(10):e69. doi:10.2106/JBJS.L.01048.
9. Morris BJ, Jahangir AA, Sethi MK. Patient satisfaction: an emerging health policy issue. AAOS Now Web site. http://www.aaos.org/AAOSNow/2013/Jun/advocacy/advocacy5/?ssopc=1. Published June 2013. Accessed November 19, 2016.
10. Donabedian A. The quality of care. How can it be assessed? JAMA. 1988;260(12):1743-1748. doi:10.1001/jama.260.12.1743.
11. Bozic KJ, Kaufman D, Chan VC, Caminiti S, Lewis C. Factors that influence provider selection for elective total joint arthroplasty. Clin Orthop Relat Res. 2013;471(6):1865-1872. doi:10.1007/s11999-012-2640-9.
12. Davies AR, Ware JE Jr. Involving consumers in quality of care assessment. Health Aff (Millwood). 1988;7(1):33-48.
13. Black N, Burke L, Forrest CB, et al. Patient-reported outcomes: pathways to better health, better services, and better societies. Qual Life Res. 2016;25(5):1103-1112. doi:10.1007/s11136-015-1168-3.
14. Gilbert A, Sebag-Montefiore D, Davidson S, Velikova G. Use of patient-reported outcomes to measure symptoms and health related quality of life in the clinic. Gynecol Oncol. 2015;136(3):429-439. doi:10.1016/j.ygyno.2014.11.071.
15. Van Der Wees PJ, Nijhuis-Van Der Sanden MW, Ayanian JZ, Black N, Westert GP, Schneider EC. Integrating the use of patient-reported outcomes for both clinical practice and performance measurement: views of experts from 3 countries. Milbank Q. 2014;92(4):754-775. doi:10.1111/1468-0009.12091.
16. Franklin PD, Lewallen D, Bozic K, Hallstrom B, Jiranek W, Ayers DC. Implementation of patient-reported outcome measures in U.S. Total joint replacement registries: rationale, status, and plans. J Bone Joint Surg Am. 2014;96(Suppl 1):104-109. doi:10.2106/JBJS.N.00328.
17. Williams B. Patient satisfaction: a valid concept? Soc Sci Med. 1994;38(4):509-516. doi:10.1016/0277-9536(94)90247-X.
18. Hickson GB, Clayton EW, Entman SS, et al. Obstetricians' prior malpractice experience and patients' satisfaction with care. JAMA. 1994;272(20):1583-1587. doi:10.1001/jama.1994.03520200039032.
19. Larsson BW, Larsson G, Chantereau MW, von Holstein KS. International comparisons of patients' views on quality of care. Int J Health Care Qual Assur Inc Leadersh Health Serv. 2005;18(1):62-73. doi:10.1108/09526860510576974.
20. McLafferty RB, Williams RG, Lambert AD, Dunnington GL. Surgeon communication behaviors that lead patients to not recommend the surgeon to family members or friends: analysis and impact. Surgery. 2006;140(4):616-624. doi:https://doi.org/10.1016/j.surg.2006.06.021.
21. Mira JJ, Tomás O, Virtudes-Pérez M, Nebot C, Rodríguez-Marín J. Predictors of patient satisfaction in surgery. Surgery. 2009;145(5):536-541. doi:10.1016/j.surg.2009.01.012.
22. Patterson BM, Eskildsen SM, Clement RC, et al. Patient satisfaction is associated with time with provider but not clinic wait time among orthopedic patients. Orthopedics. 2017;40(1):43-48. doi:10.3928/01477447-20161013-05.
23. Teunis T, Thornton ER, Jayakumar P, Ring D. Time seeing a hand surgeon is not associated With patient satisfaction. Clin Orthop Relat Res. 2015;473(7):2362-2368. doi:10.1007/s11999-014-4090-z.
24. Burkle CM, Keegan MT. Popularity of internet physician rating sites and their apparent influence on patients' choices of physicians. BMC Health Serv Res. 2015;15:416. doi:10.1186/s12913-015-1099-2.
25. Mohney S, Lee DJ, Elfar JC. The effect of orthopedic advertising and self-promotion on a naive population. Am J Orthop. 2016;45(4):E227-E232.
26. Halawi MJ, Barsoum WK. Direct-to-consumer marketing: implications for patient care and orthopedic education. Am J Orthop. 2016;45(6):E335-E336.
27. Mostaghimi A, Crotty BH, Landon BE. The availability and nature of physician information on the internet. J Gen Intern Med. 2010;25(11):1152-1156. doi:10.1007/s11606-010-1425-7.
TAKE-HOME POINTS
- The vast majority of patients desire only 10 to 20 minutes with their doctor and are highly satisfied with the amount of time their surgeon spends with them.
- Patients reported no significant gender- or ethnicity-based preferences for their doctor.
- The majority of patients believe that a wait time exceeding 30 minutes is too long.
- Nearly 42% of respondents felt they would be receiving below average medical care if seen only by a nurse practitioner or physician’s assistant at a postoperative appointment.
- Recommendations from friends is the most common way patients find their physicians.
Hip and Core Muscle Injuries in Soccer
ABSTRACT
Soccer is the most popular sport in the world and has the fourth highest number of sports injuries. Hip and groin injuries account for 14% of soccer injuries and can be difficult to recognize and treat as they often require a high level of suspicion and advanced imaging. Groin pain can be separated into 3 categories: (1) defined clinical entities for groin pain (adductor-related, iliopsoas-related, inguinal-related [sports hernias/athletic pubalgia], and pubic-related groin pain), (2) hip-related groin pain (hip morphologic abnormalities, labral tears, and chondral injuries), and (3) other causes of groin pain. Conservative approaches are typically the first line of treatment, but operative intervention has been reported to result in higher rates of return to sport in athletes with hip-related and inguinal-related groin pain injuries. In patients with concurrent hip-related and inguinal-related groin pain, the failure to recognize the relationship and treat both conditions may result in lower rates of return to sport. Preseason screening programs can identify high-risk athletes, who may benefit from a targeted prevention program. Further study on exercise therapy, early surgical intervention, and potential biologic intervention are needed to determine the most effective methods of preventing groin injuries in athletes.
Continue to: Each year, the global audience for soccer grows...
Each year, the global audience for soccer grows. Soccer has long surpassed all other sports as the most popular sport in the world, reaching 3.2 billion viewers during the 2014 World Cup.1 In the latest Fédération Internationale de Football Association (FIFA) Big Count survey, the organization estimated that 265 million people are actively involved in soccer, accounting for approximately 4% of the world’s population.2 Moreover, the number of people playing soccer increased by 9.5% within 6 years after the previous Big Count Survey.2 In the United States, soccer accounts for the fourth most common cause of sports injuries next to basketball, exercise, and football with approximately 228,000 injuries per year.3 The total cost of treatment related to worldwide soccer injuries tops $30 billion.4 The most common body parts injured are the thigh (25%), knee (18%), and hip and/or groin (14%).5
Hip and groin injuries in soccer players can be separated into 3 main categories based on the Doha Agreement:6 (1) defined clinical entities for groin pain, (2) hip-related groin pain, and (3) other causes of groin pain in athletes. Defined clinical entities include adductor-related, iliopsoas-related, inguinal-related (sports hernia/athletic pubalgia), and pubic-related groin pain; while hip-related groin pain includes hip morphologic abnormalities, labral tears, and chondral injuries. Included in other causes of groin pain are injuries not clinically defined. The Doha Agreement has acknowledged that not all causes of groin pain fit into the classification system including injuries of the rectus femoris, but they will be included under defined clinical entities for groin pain in this review. While they are not a cause of groin pain, proximal hamstring and gluteal and piriformis injuries are important causes of posterior and lateral hip pain in soccer players and will also be covered in the first section of this review.
DEFINED CLINICAL ENTITIES FOR GROIN PAIN IN SOCCER ATHLETES
ADDUCTOR-RELATED GROIN PAIN
Acute groin pain in soccer players is most commonly caused by muscle strain.7 Of the muscle strains, 66% involve the adductor longus, 25% the iliopsoas, and 23% the rectus femoris.7 The Doha Agreement defines adductor-related groin pain as adductor tenderness and pain on resisted adduction.6 Adductor longus strains in soccer players are typically noncontact injuries (62.5%) and most commonly the result of kicking (40%).7-9 Many athletes will remember a pop at the time of the original injury.9 The combination of history and physical examination is usually sufficient for diagnosis; however, magnetic resonance imaging (MRI) may be helpful in complicated situations with a reported 86% sensitivity and 89% specificity.10 The average playing time lost is 2 weeks.5 Management includes rest, anti-inflammatory medication, physical therapy with core strengthening, and avoidance of aggressive stretching. While partial and distal avulsions can heal with conservative measures, proximal osseous and retracted avulsions of the adductor longus can be treated surgically.11
Continue to: ILIOPSOAS-RELATED GROIN PAIN...
ILIOPSOAS-RELATED GROIN PAIN
Iliopsoas strains account for 25% of acute groin strains and typically result from an impact that causes eccentric overload while kicking the ball.7,12 Iliopsoas-related groin pain is defined by the Doha Agreement as groin pain that is reproducible with resisted hip flexion or hip flexor stretch.6 Iliopsoas strains respond well to conservative treatment such as rest, anti-inflammatory medication, and physical therapy. Rarely do these athletes become surgical candidates in the acute setting. Chronic cases of iliopsoas pathology occasionally require an arthroscopic intervention.
INGUINAL-RELATED GROIN PAIN
Inguinal-related groin pain is one of the most misleading diagnoses in sports because of its poorly defined and under-researched nature. The varying nomenclature of this entity illustrates the heterogeneity and includes sports hernia,9,13-15 athletic pubalgia,16 core muscle injury,17 athletic hernia,18 Gilmore’s groin,15 osteitis pubis,19 sportsman’s hernia,20,21 sportsmen’s groin,22 symphysis syndrome,23 and inguinal disruption.24 It is important to realize that in inguinal-related groin pain, regardless of the nomenclature, there is no true hernia present. The Doha Agreement has defined inguinal-related groin pain as “pain in the location of the inguinal region with associated tenderness of the inguinal canal,” which “is more likely if the pain is aggravated with resistance testing of the abdominal muscles or on Valsalva/cough/sneeze.”6 The condition is a painful soft tissue injury in the groin or inguinal area, involving a constellation of various anatomic areas including the abdominal musculature, sacroiliac joint, neural structures, pubic symphysis, adductors, and hip joint. This may account for up to 50% of chronic groin pain.25,26
One important theory in the development of inguinal-related groin pain is its relationship with femoroacetabular impingement (FAI). Cadaver studies demonstrate that cam deformities cause a 35% increase in motion at the pubic symphysis altering the biomechanics of the adductors and abdominal musculature and, with repetitive stress, may lead to tearing or attenuation of the transversalis fascia, rectus abdominis, internal obliques, and/or external obliques.12,27,28 Another prevailing theory of this is that the increased pubic stress causes weakness in the posterior portion of the inguinal canal, which then stretches and entraps the genitofemoral, ilioinguinal, lateral femoral cutaneous, or obturator nerves, ultimately causing pain.28,29
Physical examination findings include pain over the conjoined tendon, pubic tubercle/symphysis (present in 22% of patients), adductor origin (36%), and inguinal ring.25,30 Pain with resisted sit-ups is present in 46% of patients and pain with coughing/Valsalva is present in 10%.25,30,31 Selective injections can be a critical part of the evaluation to differentiate inguinal-related groin pain from FAI, osteitis pubis, and adductor strains while helping to determine the appropriate treatment.25,32 The role of advanced imaging is unclear as the clinical entity is still uncertain and the standard imaging findings have not been definitively established.33 However, several studies have reported MRI findings suggestive of inguinal-related groin pain. One of the more common MRI findings is the “secondary cleft sign,” which requires injecting a dye into the pubic symphysis.34 Several studies have shown that the radiographic dye extravasates preferentially into the side where the groin symptoms exist and are thought to be secondary to micro-tearing at the common attachment of the musculotendinous structures to the anterior pubis.34,35 However, it should be noted that the lack of imaging findings does not exclude the possibility of inguinal-related groin pathology.
Initial treatment consists of rest, anti-inflammatory medication, injections, and physical therapy with core strengthening.25 A study by Paajanen and colleagues36 suggested that early surgical intervention may be preferred over conservative management in a randomized trial comparing physical therapy, injections, anti-inflammatory medication, and rest vs an extraperitoneal laparoscopic mesh repair behind the pubic symphysis. In the conservative group, 20% of athletes returned to sport at 1 month, 27% at 3 months, and 50% at 12 months.36 In comparison, the surgical group had 67% return to sport at 1 month, 90% at 3 months, and 97% at 12 months.36 If surgical management is chosen, there are a variety of surgical options including laparoscopy, open or mini-open repairs of the abdominal musculature/fascia or pelvic floor with and without mesh, neurolysis, and adductor release. Muschawek and Berger37described a series of 129 patients that had an open-suture repair of the posterior wall of the inguinal canal with 67% of professional athletes returning to sport within 2 weeks and 83.7% of athletes returning to sport overall. The rates of return to play are consistently 80% to 100% without demonstrated superiority of one technique over another up to this point.30
Continue to: PUBIC-RELATED GROIN PAIN...
PUBIC-RELATED GROIN PAIN
Pubic-related groin pain is defined as tenderness to palpation over the pubic symphysis and adjacent bone.6 Osteitis pubis is a chronic overuse injury characterized by localized pain to the pubic symphysis and is believed to be caused by repetitive microtrauma from a dynamic rotation of the sacroiliac joint with suggested imbalances between the rectus abdominis and the adductor musculature.12,38 In soccer players, the condition may be related to the constant torsional stresses of kicking, running, or twisting.12 If performed, radiographs often show lytic areas of the pubic symphysis, widening of the symphysis, sclerosis, and cystic changes, while bone marrow edema may be present on MRI.38Management consists of rest, anti-inflammatory medication, and corticosteroid injections with gentle stretching once asymptomatic.12,39
RECTUS FEMORIS INJURIES
The most common injury to the rectus femoris is a strain as a result of an eccentric overload while a soccer player is hit trying to extend his or her leg to kick a ball.12 In pediatric soccer athletes, an avulsion of the anterior inferior iliac spine from the direct head of the rectus femoris is the second most common avulsion injury.40 Radiographs are diagnostic and can help determine treatment. Most avulsions are minimally displaced and can be treated conservatively, but surgical intervention should be considered for an avulsion >2 cm.12
PROXIMAL HAMSTRING INJURIES
Proximal hamstring injuries are important causes of acute posterior hip pain and are caused by an eccentric overload in hip flexion and knee extension.25 In soccer players, the typical mechanism is that the planted leg slipping on the playing turf creates a sudden violent flexion of the hip with the knee in an extended position. While relatively uncommon, when a significant avulsion occurs in a professional athlete, surgical intervention is often necessary. In general, these injuries may involve partial or full avulsions off the ischial tuberosity or separation of the bony apophysis in pediatric athletes. A physical examination in the acute setting typically demonstrates massive posterior thigh ecchymosis, a palpable defect, and/or weakness with knee flexion. Imaging is helpful to confirm the diagnosis and evaluate for surgical repair. Radiographs may show a bony avulsion, which is more commonly seen in pediatric apophyseal avulsions. MRI can be used to differentiate a complete tear (involving all 3 tendons) vs a partial tear and evaluate for retraction of the tendon distally. Complete and partial tears of 2 tendons with retraction of >2 cm should be surgically repaired.25 Partial tears without tendon retraction may be treated conservatively with rest, anti-inflammatory medication, and physical therapy and then followed later by a hamstring prevention program.25 We have found that biologic augmentation with platelet-rich plasma can help accelerate healing in partial thickness injuries; however, the evidence is conflicting.
GLUTEAL INJURIES
Chronic overuse injuries of the gluteal musculature are common causes of lateral hip pain. Abductor overuse caused by weakness in the gluteus medius with a normal tensor fascia lata can cause pain with sitting and side-lying.25Overuse of the gluteal muscles with muscular imbalances along with increased tension on the iliotibial band can lead to greater trochanteric pain syndrome.25 A physical examination may demonstrate tenderness over the greater trochanter bursa and positive flexion, abduction, and external rotation testing.25 Abductor overuse syndrome and greater trochanteric pain syndrome are best treated with anti-inflammatory medication and physical therapy to balance the core/pelvic musculature.41
PIRIFORMIS INJURIES
Piriformis syndrome is a compressive neuropathy of the sciatic nerve. The mechanism of injury in the athlete is through a minor trauma to the buttock or pelvis.25,42,43 Presenting symptoms include pain with sitting and internal rotation of the hip.12 Zeren and colleagues42 published the only study that includes 2 cases of bilateral piriformis syndrome in professional soccer players. The diagnosis was confirmed with electromyography that was negative at rest and positive when measured after running.42 The athletes exhausted conservative treatment with physical therapy, anti-inflammatory medications, injections, and rest and were treated with surgical decompression.42 Both players returned to professional soccer after 6 months and played for an average of 7 years.42
Continue to: HIP-RELATED GROIN PAIN IN SOCCER ATHLETES...
HIP-RELATED GROIN PAIN IN SOCCER ATHLETES
Hip-related groin pain has garnered more attention in the last several years after being a previously underdiagnosed entity. One study found that practitioners treated groin pain in athletes for 7 months on average before recognizing that the pathology was intra-articular.44 FAI, labral tears, and chondral injuries are the major intra-articular pathologies that cause groin pain in athletes and ultimately impaired performance.45,46
FEMOROACETABULAR IMPINGEMENT
FAI is caused by pincer-type, cam-type, or combined-type deformities. Pincer lesions are defined as an increased acetabular overhang, while cam lesions are described as an increased bone at the femoral head/neck junction. These deformities in isolation or in combination cause decreased hip motion and increased contact pressures between the anterolateral acetabulum and femoral head-neck junction, which may ultimately lead to labral tears, chondral lesions, and osteoarthritis.47 During hip flexion, cam deformities impact the anterolateral acetabulum, preferentially causing articular cartilage damage, while sparing the labrum.25 Conversely, pincer deformities cause repetitive microtrauma to the labrum, crushing it between the acetabular rim and femoral neck with secondary damage to the articular cartilage.25 Over time, the damage to the labrum and articular cartilage may lead to premature osteoarthritis, which occurs at a much younger age in the athletic population.48
We know from previous studies that soccer athletes have a high prevalence of morphologic abnormalities of the hip, most commonly FAI. Gerhardt and colleagues49 documented the prevalence of hip morphologic abnormalities in elite soccer players and found abnormalities in 72% of men and over 50% of women. It should be noted that this series looked at asymptomatic athletes; however, it has been shown that hip dysmorphia is a risk factor for hip and groin injuries and may provide an opportunity for injury prevention strategies.50
Physical examination findings in FAI include decreased hip internal rotation and pain with provocative testing. Wyss and colleagues51 measured hip internal rotation in athletes with and without FAI. They found that the athletes with FAI have an average of 4° of internal rotation compared with that of the non-FAI athletes with 28°.51 A worsening internal rotation deficit has been linked to increasing severity of the deformity and when <20° was correlated with joint damage.51 Provocative testing has a high sensitivity with a recent meta-analysis demonstrating the most sensitive tests to be the anterior impingement test (flexion-adduction-internal rotation) with 94% to 99% sensitivity and the flexion-internal rotation test with 96% sensitivity.52 While provocative tests are sensitive, there is no current consensus on physical examination findings that are specific in the diagnosis of FAI.6 Diagnosis is made with both positive physical examination and radiographic morphologic findings (alpha angle >55°).33 Advanced imaging with an MRI arthrogram can be helpful in diagnosing underlying injuries such as labral tears in athletes presenting with compatible symptoms.
Symptomatic patients are typically treated surgically through either open or arthroscopic procedures, which have favorable and comparable functional results, biomechanics, and return to sport.53 In soccer players, return to sport at the professional level after arthroscopic surgery was found to be 96%.54 Players returned to sport on average 9.2 months postoperatively and played an average of 70 games after surgery.54
Continue to: LABRAL TEARS...
LABRAL TEARS
Labral tears present with groin pain, limited hip range of motion, and symptoms of catching, locking, and instability.25Causes of labral tears include trauma, FAI, hip dysplasia, capsular laxity, and degeneration.55 Labral tears rarely occur in isolation and have a high association (87%) with morphologic abnormalities of the hip, most commonly FAI and occasionally dysplasia.56,57 Physical examination findings include positive anterior impingement tests (flexion-adduction-internal rotation) in athletes with anterior labral tears and, less commonly, positive flexion, abduction, and external rotation tests for athletes with lateral and posterolateral labral tears.57 Radiographic imaging is used to evaluate for concurrent morphologic abnormalities of the hip, and MRI arthrogram is used to confirm the diagnosis of a labral tear with a sensitivity of 76% to 91%.58 Initial treatment consists of conservative treatment, which includes rest, anti-inflammatory medication, activity modification, and physical therapy. In patient refractory to conservative treatment, arthroscopic surgery is effective with high rates of return to sport.59 It is important to note that when treating labral tears surgically, any morphologic abnormality needs to be addressed to prevent recurrence of the tear.
CHONDRAL INJURIES
Focal chondral lesions in the hip are commonly found in athletes with FAI and labral tears during arthroscopic evaluation.60 Full-thickness defects and unstable flaps in weight-bearing areas are indications for surgical intervention with microfracture.60 There are no studies examining the efficacy of microfracture in isolation; however, Locks and colleagues54 have demonstrated a 96% return to professional soccer after an arthroscopic treatment for FAI and found that severe chondral damage with microfracture did not lengthen the return to sport.
RELATIONSHIP BETWEEN INGUINAL-RELATED GROIN PAIN AND FEMOROACETABULAR IMPINGEMENT
The altered biomechanics and restricted range of motion in athletes with FAI cause an increase in compensatory motion at the pelvis and lumbosacral areas, which may contribute to the development of inguinal-related groin pain, bursitis, adductor, and gluteal dysfunction.25 In athletes with concurrent intra-articular hip pathology and inguinal-related groin pain, treating 1 condition in isolation will result in poor results. Larson and colleagues61 found that when only inguinal-related groin pain or FAI were addressed, return to sport was only 25% and 50%, respectively, while concurrent surgical treatment resulted in a return to sport of 89%.
DISCUSSION AND FUTURE DIRECTIONS
Groin injuries in soccer players can cause significant decreases in athletic performance, result in lost playing time, and may ultimately need a surgical intervention. Efforts are underway to determine the role and efficacy of identifying high-risk athletes that may benefit from targeted prevention strategies. Wyles and colleagues48 identified adolescent athletes with hip internal rotation of <10° and found at 5-year follow-up that 95% had abnormal MRI findings compared with 54% in the age-matched control group. Wollin and colleagues62 developed an in-season screening protocol using adductor strength reductions of 15%, adductor/abductor strength ratio <0.9, and hip and groin outcome scores <75 as indicators of at-risk individuals. By employing preseason and in-season screening protocols, we can identify high-risk athletes for further workup and close follow-up throughout the season. Pelvic radiographs in these high-risk athletes may help us determine the presence of abnormalities in hip morphology, which would place an athlete into a high-risk group where prevention strategies could then be employed. There are no data available to determine the most effective prevention strategy at this time. However, levels II and III evidence exists indicating that exercise programs may reduce the incidence of groin injuries.63 Additional strategies, like limiting adolescent playing time similar to strategies employed in baseball pitches with pitch counts, could potentially reduce the potential for injury. Further studies on preseason screening and in-season monitoring protocols, targeted exercise therapy, early surgical intervention, and potential biologic intervention are needed to determine the most effective methods of preventing groin injuries in athletes.
1. Kantar Media. 2014 FIFA World Cup Brazil television audience report. https://resources.fifa.com/mm/document/affederation/tv/02/74/55/57/2014f...(draft5)(issuedate14.12.15)_neutral.pdf. Accessed March 20, 2018.
2. Fédération Internationale de Football Association. FIFA Big Count. http://www.fifa.com/mm/document/fifafacts/bcoffsurv/emaga_9384_10704.pdf. Published July 2007. Accessed March 20, 2018.
3. United States Consumer Product Safety Commission. Neiss data highlights - 2015. https://www.cpsc.gov/s3fs-public/2015 Neiss data highlights.pdf. Accessed March 20, 2018.
4. Hassabi M, Mohammad-Javad Mortazavi S, Giti MR, Hassabi M, Mansournia MA, Shapouran S. Injury profile of a professional soccer team in the premier league of Iran. Asian J Sports Med. 2010;1(4):201-208.
5. Ekstrand J, Hagglund M, Walden M. Injury incidence and injury patterns in professional football: the UEFA injury study. Br J Sports Med. 2011;45(7):553-558.
6. Weir A, Brukner P, Delahunt E, et al. Doha agreement meeting on terminology and definitions in groin pain in athletes. Br J Sports Med. 2015;49(12):768-774.
7. Serner A, Tol JL, Jomaah N, et al. Diagnosis of acute groin injuries: a prospective study of 110 athletes. Am J Sports Med. 2015;43(8):1857-1864. doi:10.1177/0363546515585123.
8. Eckard TG, Padua DA, Dompier TP, Dalton SL, Thorborg K, Kerr ZY. Epidemiology of hip flexor and hip adductor strains in national collegiate athletic association athletes, 2009/2010-2014/2015. Am J Sports Med. 2017;45(12):2713-2722. doi:10.1177/0363546517716179.
9. Hopkins JN, Brown W, Lee CA. Sports hernia: definition, evaluation, and treatment. JBJS Rev. 2017;5(9):e6. doi:10.2106/JBJS.RVW.17.00022.
10. Omar IM, Zoga AC, Kavanagh EC, et al. Athletic pubalgia and "sports hernia": optimal MR imaging technique and findings. Radiographics. 2008;28(5):1415-1438. doi:10.1148/rg.285075217.
11. Vogt S, Ansah P, Imhoff AB. Complete osseous avulsion of the adductor longus muscle: acute repair with three Wberwire suture anchors. Arch Orthop Trauma Surg. 2007;127:613-615. doi:10.1007/s00402-007-0328-5.
12. Anderson K, Strickland SM, Warren R. Hip and groin injuries in athletes. Am J Sports Med. 2001;29(4):521-533. doi:10.1177/03635465010290042501.
13. Choi HR, Elattar O, Dills VD, Busconi B. Return to play after sports hernia surgery. Clin Sports Med. 2016;35(4):621-636. doi:10.1016/j.csm.2016.05.007.
14. Garvey JF, Hazard H. Sports hernia or groin disruption injury? Chronic athletic groin pain: a retrospective study of 100 patients with long-term follow-up. Hernia. 2014;18(6):815-823. doi:10.1007/s10029-013-1161-0.
15. Gilmore J. Groin pain in the soccer athlete: fact, fiction, and treatment. Clin Sports Med. 1998;17(4):787-793, vii. doi:10.1016/S0278-5919(05)70119-8.
16. Cohen B, Kleinhenz D, Schiller J, Tabaddor R. Understanding athletic pubalgia: a review. R I Med J. 2016;99(10):31-35.
17. Ross JR, Stone RM, Larson CM. Core muscle injury/sports hernia/athletic pubalgia, and femoroacetabular impingement. Sports Med Arthrosc Rev. 2015;23(4):213-220. doi:10.1097/JSA.0000000000000083.
18. Swan KG Jr, Wolcott M. The athletic hernia: a systematic review. Clin Orthop Relat Res. 2007;455:78-87. doi:10.1097/BLO.0b013e31802eb3ea.
19. Matikainen M, Hermunen H, Paajanen H. Athletic pubalgia in females: predictive value of MRI in outcomes of endoscopic surgery. Orthop J Sports Med. 2017;5(8):2325967117720171. doi:10.1177/2325967117720171.
20. Garvey JF, Read JW, Turner A. Sportsman hernia: what can we do? Hernia. 2010;14(1):17-25. doi:10.1007/s10029-009-0611-1.
21. Paksoy M, Sekmen U. Sportsman hernia; the review of current diagnosis and treatment modalities. Ulusal Cerrahi Derg. 2016;32(2):122-129. doi:10.5152/UCD.2015.3132.
22. Pokorny H, Resinger C, Fischer I, et al. Fast early recovery after transabdominal preperitoneal repair in athletes with sportsman's groin: a prospective clinical cohort study. J Laparoendosc Adv Surg Tech A. 2017;27(3):272-276. doi:10.1089/lap.2016.0188.
23. Biedert RM, Warnke K, Meyer S. Symphysis syndrome in athletes: surgical treatment for chronic lower abdominal, groin, and adductor pain in athletes. Clin J Sport Med. 2003;13(5):278-284.
24. Sheen AJ, Stephenson BM, Lloyd DM, et al. 'Treatment of the sportsman's groin': British Hernia Society's 2014 position statement based on the Manchester Consensus Conference. Br J Sports Med. 2014;48(14):1079-1087.
25. Miller M, Thompson S. DeLee & Drez's Orthopaedic Sports Medicine. 4th ed. Philadelphia, PA: Elsevier/Saunders; 2015.
26. Lovell G. The diagnosis of chronic groin pain in athletes: a review of 189 cases. J Sci Med Sport. 1995;27:76-79.
27. Dimitrakopoulou A, Schilders E. Sportsman's hernia? An ambiguous term. J Hip Preserv Surg. 2016;3(1):16-22. doi:10.1093/jhps/hnv083.
28. Strosberg DS, Ellis TJ, Renton DB. The role of femoroacetabular impingement in core muscle injury/athletic pubalgia: diagnosis and management. Front Surg. 2016;3:6. doi:10.3389/fsurg.2016.00006.
29. Muschaweck U, Berger LM. Sportsmen's groin-diagnostic approach and treatment with the minimal repair technique: a single-center uncontrolled clinical review. Sports Health. 2010;2(3):216-221. doi:10.1177/1941738110367623.
30. Larson CM. Sports hernia/athletic pubalgia: evaluation and management. Sports Health. 2014;6(2):139-144. doi:10.1177/1941738114523557.
31. Meyers WC, Foley DP, Garrett WE, Lohnes JH, Mandlebaum BR. Management of severe lower abdominal or inguinal pain in high-performance athletes. Am J Sports Med. 2000; 28(1):2-8. doi:10.1177/03635465000280011501.
32. Gerhardt MB, Mandelbaum BR, Hutchinson WB. Ancillary modalities in the treatment of athletic groin Pain: Local Anesthetics, Corticosteroids, and Orthobiologics. In: Diduch DR, Brunt LM, eds. Sports Hernia and Athletic Pubalgia: Diagnosis and Treatment. Boston, MA: Springer US; 2014:183-187.
33. Notzli HP, Wyss TF, Stoecklin CH, et al. The contour of the femoral head-neck junction as a predictor for the risk of anterior impingement. J Bone Joint Surg Br. 2002;84(4):556-560.
34. Brennan D, O’Connell MJ, Ryan M, et al. Secondary cleft sign as a marker of injury in athletes with groin pain: MR image appearance and interpretation. Radiology. 2005;235(1):162-167. doi:10.1148/radiol.2351040045.
35. Byrne CA, Bowden DJ, Alkhayat A, Kavanagh EC, Eustace SJ. Sports-related groin pain secondary to symphysis pubis disorders: correlation between MRI findings and outcome after fluoroscopy-guided injection of steroid and local anesthetic. Am J Roentgenol. 2017;209(2):380-388. doi:10.2214/AJR.16.17578.
36. Paajanen H, Brinck T, Hermunen H, Airo I. Laparoscopic surgery for chronic groin pain in athletes is more effective than nonoperative treatment: a randomized clinical trial with magnetic resonance imaging of 60 patients with sportsman's hernia (athletic pubalgia). Surgery. 2011;150(1):99-107. doi:10.1016/j.surg.2011.02.016.
37. Muschaweck U, Berger L. Minimal repair technique of sportsmen's groin: an innovative open-suture repair to treat chronic inguinal pain. Hernia. 2010;14(1):27-33. doi:10.1007/s10029-009-0614-y.
38. Lynch TS, Bedi A, Larson CM. Athletic hip injuries. J Am Acad Orthop Surg. 2017;25(4):269-279. doi:10.5435/JAAOS-D-16-00171.
39. Holt MA, Keene JS, Graf BK, Helwig DC. Treatment of osteitis pubis in athletes. Results of corticosteroid injections. Am J Sports Med. 1995;23(5):601-606.doi:10.1177/036354659502300515.
40. Rossi F, Dragoni S. Acute avulsion fractures of the pelvis in adolescent competitive athletes: prevalence, location and sports distribution of 203 cases collected. Skeletal Radiol. 2001;30(3):127-131. doi: 10.1007/s002560000319.
41. Grumet RC, Frank RM, Slabaugh MA, Virkus WW, Bush-Joseph CA, Nho SJ. Lateral hip pain in an athletic population: differential diagnosis and treatment options. Sports Health. 2010;2(3):191-196. doi:10.1177/1941738110366829.
42. Zeren B, Canbek U, Oztekin HH, Imerci A, Akgun U. Bilateral piriformis syndrome in two elite soccer players: report of two cases. Orthop Traumatol Surg Res. 2015;101(8):987-990. doi:10.1016/j.otsr.2015.07.022.
43. Keskula DR, Tamburello M. Conservative management of piriformis syndrome. J Athl Train. 1992;27(2):102-110.
44. Byrd JW, Jones KS. Hip arthroscopy in athletes. Clin Sports Med. 2001;20(4):749-761.
45. Nepple JJ, Goljan P, Briggs KK, Garvey SE, Ryan M, Philippon MJ. Hip strength deficits in patients with symptomatic femoroacetabular impingement and labral tears. Arthroscopy.2015;31(11):2106-2111.
46. Mullins K, Hanlon M, Carton P. Differences in athletic performance between sportsmen with symptomatic femoroacetabular impingement and healthy controls. Clin J Sport Med.2018;28(4):370-376. doi:10.1097/JSM.0000000000000460.
47. Ganz R, Parvizi J, Beck M, Leunig M, Notzli H, Siebenrock KA. Femoroacetabular impingement: a cause for osteoarthritis of the hip. Clin Orthop Relat Res. 2003;417:112-120. doi:10.1097/01.blo.0000096804.78689.c2.
48. Wyles CC, Norambuena GA, Howe BM, et al. Cam deformities and limited hip range of motion are associated with early osteoarthritic changes in adolescent athletes: a prospective matched cohort study. Am J Sports Med. 2017;45(13):3036-3043. doi:10.1177/0363546517719460 .
49. Gerhardt MB, Romero AA, Silvers HJ, Harris DJ, Watanabe D, Mandelbaum BR. The prevalence of radiographic hip abnormalities in elite soccer players. Am J Sports Med. 2012;40(3):584-588. doi:10.1177/0363546511432711.
50. Larson CM, Ross JR, Kuhn AW, et al. Radiographic hip anatomy correlates with range of motion and symptoms in national hockey league players. Am J Sports Med. 2017;45(7):1633-1639. doi:10.1177/0363546517692542.
51. Wyss TF, Clark JM, Weishaupt D, Notzli HP. Correlation between internal rotation and bony anatomy in the hip. Clin Orthop Relat Res. 2007;460:152-158. doi:10.1097/BLO.0b013e3180399430.
52. Reiman MP, Goode AP, Cook CE, Holmich P, Thorborg K. Diagnostic accuracy of clinical tests for the diagnosis of hip femoroacetabular impingement/labral tear: a systematic review with meta-analysis. Br J Sports Med. 2015;49:811. doi:10.1136/bjsports-2014-094302.
53. Papalia R, Del Buono A, Franceschi F, Marinozzi A, Maffulli N, Denaro V. Femoroacetabular impingement syndrome management: arthroscopy or open surgery? Int Orthop. 2012;36(5):903-914. doi:10.1007/s00264-011-1443-z.
54. Locks R, Utsunomiya H, Briggs KK, McNamara S, Chahla J, Philippon MJ. Return to play after hip arthroscopic surgery for femoroacetabular impingement in professional soccer players. Am J Sports Med. 2018;46(2):273-279. doi:10.1177/0363546517738741.
55. Kelly BT, Weiland DE, Schenker ML, Philippon MJ. Arthroscopic labral repair in the hip: surgical technique and review of the literature. Arthroscopy. 2005;21(12):1496-1504. doi:10.1016/j.arthro.2005.08.013.
56. Wenger DE, Kendell KR, Miner MR, Trousdale RT. Acetabular labral tears rarely occur in the absence of bony abnormalities. Clin Orthop Relat Res. 2004;426:145-150. doi:10.1097/01.blo.0000136903.01368.20.
57. Domb BG, Hartigan DE, Perets I. Decision making for labral treatment in the hip: repair versus débridement versus reconstruction. J Am Acad Orthop Surg. 2017;25(3):e53-e62. doi:10.5435/JAAOS-D-16-00144.
58. Frank JS, Gambacorta PL, Eisner EA. Hip pathology in the adolescent athlete. J Am Acad Orthop Surg. 2013;21(11):665-674. doi:10.5435/JAAOS-21-11-665.
59. Singh PJ, O'Donnell JM. The outcome of hip arthroscopy in Australian football league players: a review of 27 hips. Arthroscopy. 2010;26(6):743-749. doi:10.1016/j.arthro.2009.10.010.
60. Crawford K, Philippon MJ, Sekiya JK, Rodkey WG, Steadman JR. Microfracture of the hip in athletes. Clin Sports Med. 2006;25(2):327-335. doi:10.1016/j.csm.2005.12.004.
61. Larson CM, Pierce BR, Giveans MR. Treatment of athletes with symptomatic intra-articular hip pathology and athletic pubalgia/sports hernia: a case series. Arthroscopy.2011;27(6):768-775. doi:10.1016/j.arthro.2011.01.018.
62. Wollin M, Thorborg K, Welvaert M, Pizzari T. In-season monitoring of hip and groin strength, health and function in elite youth soccer: implementing an early detection and management strategy over two consecutive seasons. J Sci Med Sport. 2018;21(10):988. doi:10.1016/j.jsams.2018.03.004.
63. Charlton PC, Drew MK, Mentiplay BF, Grimaldi A, Clark RA. Exercise interventions for the prevention and treatment of groin pain and injury in athletes: a critical and systematic review. Sports Med. 2017;47:2011. doi:10.1007/s40279-017-0742-y.
ABSTRACT
Soccer is the most popular sport in the world and has the fourth highest number of sports injuries. Hip and groin injuries account for 14% of soccer injuries and can be difficult to recognize and treat as they often require a high level of suspicion and advanced imaging. Groin pain can be separated into 3 categories: (1) defined clinical entities for groin pain (adductor-related, iliopsoas-related, inguinal-related [sports hernias/athletic pubalgia], and pubic-related groin pain), (2) hip-related groin pain (hip morphologic abnormalities, labral tears, and chondral injuries), and (3) other causes of groin pain. Conservative approaches are typically the first line of treatment, but operative intervention has been reported to result in higher rates of return to sport in athletes with hip-related and inguinal-related groin pain injuries. In patients with concurrent hip-related and inguinal-related groin pain, the failure to recognize the relationship and treat both conditions may result in lower rates of return to sport. Preseason screening programs can identify high-risk athletes, who may benefit from a targeted prevention program. Further study on exercise therapy, early surgical intervention, and potential biologic intervention are needed to determine the most effective methods of preventing groin injuries in athletes.
Continue to: Each year, the global audience for soccer grows...
Each year, the global audience for soccer grows. Soccer has long surpassed all other sports as the most popular sport in the world, reaching 3.2 billion viewers during the 2014 World Cup.1 In the latest Fédération Internationale de Football Association (FIFA) Big Count survey, the organization estimated that 265 million people are actively involved in soccer, accounting for approximately 4% of the world’s population.2 Moreover, the number of people playing soccer increased by 9.5% within 6 years after the previous Big Count Survey.2 In the United States, soccer accounts for the fourth most common cause of sports injuries next to basketball, exercise, and football with approximately 228,000 injuries per year.3 The total cost of treatment related to worldwide soccer injuries tops $30 billion.4 The most common body parts injured are the thigh (25%), knee (18%), and hip and/or groin (14%).5
Hip and groin injuries in soccer players can be separated into 3 main categories based on the Doha Agreement:6 (1) defined clinical entities for groin pain, (2) hip-related groin pain, and (3) other causes of groin pain in athletes. Defined clinical entities include adductor-related, iliopsoas-related, inguinal-related (sports hernia/athletic pubalgia), and pubic-related groin pain; while hip-related groin pain includes hip morphologic abnormalities, labral tears, and chondral injuries. Included in other causes of groin pain are injuries not clinically defined. The Doha Agreement has acknowledged that not all causes of groin pain fit into the classification system including injuries of the rectus femoris, but they will be included under defined clinical entities for groin pain in this review. While they are not a cause of groin pain, proximal hamstring and gluteal and piriformis injuries are important causes of posterior and lateral hip pain in soccer players and will also be covered in the first section of this review.
DEFINED CLINICAL ENTITIES FOR GROIN PAIN IN SOCCER ATHLETES
ADDUCTOR-RELATED GROIN PAIN
Acute groin pain in soccer players is most commonly caused by muscle strain.7 Of the muscle strains, 66% involve the adductor longus, 25% the iliopsoas, and 23% the rectus femoris.7 The Doha Agreement defines adductor-related groin pain as adductor tenderness and pain on resisted adduction.6 Adductor longus strains in soccer players are typically noncontact injuries (62.5%) and most commonly the result of kicking (40%).7-9 Many athletes will remember a pop at the time of the original injury.9 The combination of history and physical examination is usually sufficient for diagnosis; however, magnetic resonance imaging (MRI) may be helpful in complicated situations with a reported 86% sensitivity and 89% specificity.10 The average playing time lost is 2 weeks.5 Management includes rest, anti-inflammatory medication, physical therapy with core strengthening, and avoidance of aggressive stretching. While partial and distal avulsions can heal with conservative measures, proximal osseous and retracted avulsions of the adductor longus can be treated surgically.11
Continue to: ILIOPSOAS-RELATED GROIN PAIN...
ILIOPSOAS-RELATED GROIN PAIN
Iliopsoas strains account for 25% of acute groin strains and typically result from an impact that causes eccentric overload while kicking the ball.7,12 Iliopsoas-related groin pain is defined by the Doha Agreement as groin pain that is reproducible with resisted hip flexion or hip flexor stretch.6 Iliopsoas strains respond well to conservative treatment such as rest, anti-inflammatory medication, and physical therapy. Rarely do these athletes become surgical candidates in the acute setting. Chronic cases of iliopsoas pathology occasionally require an arthroscopic intervention.
INGUINAL-RELATED GROIN PAIN
Inguinal-related groin pain is one of the most misleading diagnoses in sports because of its poorly defined and under-researched nature. The varying nomenclature of this entity illustrates the heterogeneity and includes sports hernia,9,13-15 athletic pubalgia,16 core muscle injury,17 athletic hernia,18 Gilmore’s groin,15 osteitis pubis,19 sportsman’s hernia,20,21 sportsmen’s groin,22 symphysis syndrome,23 and inguinal disruption.24 It is important to realize that in inguinal-related groin pain, regardless of the nomenclature, there is no true hernia present. The Doha Agreement has defined inguinal-related groin pain as “pain in the location of the inguinal region with associated tenderness of the inguinal canal,” which “is more likely if the pain is aggravated with resistance testing of the abdominal muscles or on Valsalva/cough/sneeze.”6 The condition is a painful soft tissue injury in the groin or inguinal area, involving a constellation of various anatomic areas including the abdominal musculature, sacroiliac joint, neural structures, pubic symphysis, adductors, and hip joint. This may account for up to 50% of chronic groin pain.25,26
One important theory in the development of inguinal-related groin pain is its relationship with femoroacetabular impingement (FAI). Cadaver studies demonstrate that cam deformities cause a 35% increase in motion at the pubic symphysis altering the biomechanics of the adductors and abdominal musculature and, with repetitive stress, may lead to tearing or attenuation of the transversalis fascia, rectus abdominis, internal obliques, and/or external obliques.12,27,28 Another prevailing theory of this is that the increased pubic stress causes weakness in the posterior portion of the inguinal canal, which then stretches and entraps the genitofemoral, ilioinguinal, lateral femoral cutaneous, or obturator nerves, ultimately causing pain.28,29
Physical examination findings include pain over the conjoined tendon, pubic tubercle/symphysis (present in 22% of patients), adductor origin (36%), and inguinal ring.25,30 Pain with resisted sit-ups is present in 46% of patients and pain with coughing/Valsalva is present in 10%.25,30,31 Selective injections can be a critical part of the evaluation to differentiate inguinal-related groin pain from FAI, osteitis pubis, and adductor strains while helping to determine the appropriate treatment.25,32 The role of advanced imaging is unclear as the clinical entity is still uncertain and the standard imaging findings have not been definitively established.33 However, several studies have reported MRI findings suggestive of inguinal-related groin pain. One of the more common MRI findings is the “secondary cleft sign,” which requires injecting a dye into the pubic symphysis.34 Several studies have shown that the radiographic dye extravasates preferentially into the side where the groin symptoms exist and are thought to be secondary to micro-tearing at the common attachment of the musculotendinous structures to the anterior pubis.34,35 However, it should be noted that the lack of imaging findings does not exclude the possibility of inguinal-related groin pathology.
Initial treatment consists of rest, anti-inflammatory medication, injections, and physical therapy with core strengthening.25 A study by Paajanen and colleagues36 suggested that early surgical intervention may be preferred over conservative management in a randomized trial comparing physical therapy, injections, anti-inflammatory medication, and rest vs an extraperitoneal laparoscopic mesh repair behind the pubic symphysis. In the conservative group, 20% of athletes returned to sport at 1 month, 27% at 3 months, and 50% at 12 months.36 In comparison, the surgical group had 67% return to sport at 1 month, 90% at 3 months, and 97% at 12 months.36 If surgical management is chosen, there are a variety of surgical options including laparoscopy, open or mini-open repairs of the abdominal musculature/fascia or pelvic floor with and without mesh, neurolysis, and adductor release. Muschawek and Berger37described a series of 129 patients that had an open-suture repair of the posterior wall of the inguinal canal with 67% of professional athletes returning to sport within 2 weeks and 83.7% of athletes returning to sport overall. The rates of return to play are consistently 80% to 100% without demonstrated superiority of one technique over another up to this point.30
Continue to: PUBIC-RELATED GROIN PAIN...
PUBIC-RELATED GROIN PAIN
Pubic-related groin pain is defined as tenderness to palpation over the pubic symphysis and adjacent bone.6 Osteitis pubis is a chronic overuse injury characterized by localized pain to the pubic symphysis and is believed to be caused by repetitive microtrauma from a dynamic rotation of the sacroiliac joint with suggested imbalances between the rectus abdominis and the adductor musculature.12,38 In soccer players, the condition may be related to the constant torsional stresses of kicking, running, or twisting.12 If performed, radiographs often show lytic areas of the pubic symphysis, widening of the symphysis, sclerosis, and cystic changes, while bone marrow edema may be present on MRI.38Management consists of rest, anti-inflammatory medication, and corticosteroid injections with gentle stretching once asymptomatic.12,39
RECTUS FEMORIS INJURIES
The most common injury to the rectus femoris is a strain as a result of an eccentric overload while a soccer player is hit trying to extend his or her leg to kick a ball.12 In pediatric soccer athletes, an avulsion of the anterior inferior iliac spine from the direct head of the rectus femoris is the second most common avulsion injury.40 Radiographs are diagnostic and can help determine treatment. Most avulsions are minimally displaced and can be treated conservatively, but surgical intervention should be considered for an avulsion >2 cm.12
PROXIMAL HAMSTRING INJURIES
Proximal hamstring injuries are important causes of acute posterior hip pain and are caused by an eccentric overload in hip flexion and knee extension.25 In soccer players, the typical mechanism is that the planted leg slipping on the playing turf creates a sudden violent flexion of the hip with the knee in an extended position. While relatively uncommon, when a significant avulsion occurs in a professional athlete, surgical intervention is often necessary. In general, these injuries may involve partial or full avulsions off the ischial tuberosity or separation of the bony apophysis in pediatric athletes. A physical examination in the acute setting typically demonstrates massive posterior thigh ecchymosis, a palpable defect, and/or weakness with knee flexion. Imaging is helpful to confirm the diagnosis and evaluate for surgical repair. Radiographs may show a bony avulsion, which is more commonly seen in pediatric apophyseal avulsions. MRI can be used to differentiate a complete tear (involving all 3 tendons) vs a partial tear and evaluate for retraction of the tendon distally. Complete and partial tears of 2 tendons with retraction of >2 cm should be surgically repaired.25 Partial tears without tendon retraction may be treated conservatively with rest, anti-inflammatory medication, and physical therapy and then followed later by a hamstring prevention program.25 We have found that biologic augmentation with platelet-rich plasma can help accelerate healing in partial thickness injuries; however, the evidence is conflicting.
GLUTEAL INJURIES
Chronic overuse injuries of the gluteal musculature are common causes of lateral hip pain. Abductor overuse caused by weakness in the gluteus medius with a normal tensor fascia lata can cause pain with sitting and side-lying.25Overuse of the gluteal muscles with muscular imbalances along with increased tension on the iliotibial band can lead to greater trochanteric pain syndrome.25 A physical examination may demonstrate tenderness over the greater trochanter bursa and positive flexion, abduction, and external rotation testing.25 Abductor overuse syndrome and greater trochanteric pain syndrome are best treated with anti-inflammatory medication and physical therapy to balance the core/pelvic musculature.41
PIRIFORMIS INJURIES
Piriformis syndrome is a compressive neuropathy of the sciatic nerve. The mechanism of injury in the athlete is through a minor trauma to the buttock or pelvis.25,42,43 Presenting symptoms include pain with sitting and internal rotation of the hip.12 Zeren and colleagues42 published the only study that includes 2 cases of bilateral piriformis syndrome in professional soccer players. The diagnosis was confirmed with electromyography that was negative at rest and positive when measured after running.42 The athletes exhausted conservative treatment with physical therapy, anti-inflammatory medications, injections, and rest and were treated with surgical decompression.42 Both players returned to professional soccer after 6 months and played for an average of 7 years.42
Continue to: HIP-RELATED GROIN PAIN IN SOCCER ATHLETES...
HIP-RELATED GROIN PAIN IN SOCCER ATHLETES
Hip-related groin pain has garnered more attention in the last several years after being a previously underdiagnosed entity. One study found that practitioners treated groin pain in athletes for 7 months on average before recognizing that the pathology was intra-articular.44 FAI, labral tears, and chondral injuries are the major intra-articular pathologies that cause groin pain in athletes and ultimately impaired performance.45,46
FEMOROACETABULAR IMPINGEMENT
FAI is caused by pincer-type, cam-type, or combined-type deformities. Pincer lesions are defined as an increased acetabular overhang, while cam lesions are described as an increased bone at the femoral head/neck junction. These deformities in isolation or in combination cause decreased hip motion and increased contact pressures between the anterolateral acetabulum and femoral head-neck junction, which may ultimately lead to labral tears, chondral lesions, and osteoarthritis.47 During hip flexion, cam deformities impact the anterolateral acetabulum, preferentially causing articular cartilage damage, while sparing the labrum.25 Conversely, pincer deformities cause repetitive microtrauma to the labrum, crushing it between the acetabular rim and femoral neck with secondary damage to the articular cartilage.25 Over time, the damage to the labrum and articular cartilage may lead to premature osteoarthritis, which occurs at a much younger age in the athletic population.48
We know from previous studies that soccer athletes have a high prevalence of morphologic abnormalities of the hip, most commonly FAI. Gerhardt and colleagues49 documented the prevalence of hip morphologic abnormalities in elite soccer players and found abnormalities in 72% of men and over 50% of women. It should be noted that this series looked at asymptomatic athletes; however, it has been shown that hip dysmorphia is a risk factor for hip and groin injuries and may provide an opportunity for injury prevention strategies.50
Physical examination findings in FAI include decreased hip internal rotation and pain with provocative testing. Wyss and colleagues51 measured hip internal rotation in athletes with and without FAI. They found that the athletes with FAI have an average of 4° of internal rotation compared with that of the non-FAI athletes with 28°.51 A worsening internal rotation deficit has been linked to increasing severity of the deformity and when <20° was correlated with joint damage.51 Provocative testing has a high sensitivity with a recent meta-analysis demonstrating the most sensitive tests to be the anterior impingement test (flexion-adduction-internal rotation) with 94% to 99% sensitivity and the flexion-internal rotation test with 96% sensitivity.52 While provocative tests are sensitive, there is no current consensus on physical examination findings that are specific in the diagnosis of FAI.6 Diagnosis is made with both positive physical examination and radiographic morphologic findings (alpha angle >55°).33 Advanced imaging with an MRI arthrogram can be helpful in diagnosing underlying injuries such as labral tears in athletes presenting with compatible symptoms.
Symptomatic patients are typically treated surgically through either open or arthroscopic procedures, which have favorable and comparable functional results, biomechanics, and return to sport.53 In soccer players, return to sport at the professional level after arthroscopic surgery was found to be 96%.54 Players returned to sport on average 9.2 months postoperatively and played an average of 70 games after surgery.54
Continue to: LABRAL TEARS...
LABRAL TEARS
Labral tears present with groin pain, limited hip range of motion, and symptoms of catching, locking, and instability.25Causes of labral tears include trauma, FAI, hip dysplasia, capsular laxity, and degeneration.55 Labral tears rarely occur in isolation and have a high association (87%) with morphologic abnormalities of the hip, most commonly FAI and occasionally dysplasia.56,57 Physical examination findings include positive anterior impingement tests (flexion-adduction-internal rotation) in athletes with anterior labral tears and, less commonly, positive flexion, abduction, and external rotation tests for athletes with lateral and posterolateral labral tears.57 Radiographic imaging is used to evaluate for concurrent morphologic abnormalities of the hip, and MRI arthrogram is used to confirm the diagnosis of a labral tear with a sensitivity of 76% to 91%.58 Initial treatment consists of conservative treatment, which includes rest, anti-inflammatory medication, activity modification, and physical therapy. In patient refractory to conservative treatment, arthroscopic surgery is effective with high rates of return to sport.59 It is important to note that when treating labral tears surgically, any morphologic abnormality needs to be addressed to prevent recurrence of the tear.
CHONDRAL INJURIES
Focal chondral lesions in the hip are commonly found in athletes with FAI and labral tears during arthroscopic evaluation.60 Full-thickness defects and unstable flaps in weight-bearing areas are indications for surgical intervention with microfracture.60 There are no studies examining the efficacy of microfracture in isolation; however, Locks and colleagues54 have demonstrated a 96% return to professional soccer after an arthroscopic treatment for FAI and found that severe chondral damage with microfracture did not lengthen the return to sport.
RELATIONSHIP BETWEEN INGUINAL-RELATED GROIN PAIN AND FEMOROACETABULAR IMPINGEMENT
The altered biomechanics and restricted range of motion in athletes with FAI cause an increase in compensatory motion at the pelvis and lumbosacral areas, which may contribute to the development of inguinal-related groin pain, bursitis, adductor, and gluteal dysfunction.25 In athletes with concurrent intra-articular hip pathology and inguinal-related groin pain, treating 1 condition in isolation will result in poor results. Larson and colleagues61 found that when only inguinal-related groin pain or FAI were addressed, return to sport was only 25% and 50%, respectively, while concurrent surgical treatment resulted in a return to sport of 89%.
DISCUSSION AND FUTURE DIRECTIONS
Groin injuries in soccer players can cause significant decreases in athletic performance, result in lost playing time, and may ultimately need a surgical intervention. Efforts are underway to determine the role and efficacy of identifying high-risk athletes that may benefit from targeted prevention strategies. Wyles and colleagues48 identified adolescent athletes with hip internal rotation of <10° and found at 5-year follow-up that 95% had abnormal MRI findings compared with 54% in the age-matched control group. Wollin and colleagues62 developed an in-season screening protocol using adductor strength reductions of 15%, adductor/abductor strength ratio <0.9, and hip and groin outcome scores <75 as indicators of at-risk individuals. By employing preseason and in-season screening protocols, we can identify high-risk athletes for further workup and close follow-up throughout the season. Pelvic radiographs in these high-risk athletes may help us determine the presence of abnormalities in hip morphology, which would place an athlete into a high-risk group where prevention strategies could then be employed. There are no data available to determine the most effective prevention strategy at this time. However, levels II and III evidence exists indicating that exercise programs may reduce the incidence of groin injuries.63 Additional strategies, like limiting adolescent playing time similar to strategies employed in baseball pitches with pitch counts, could potentially reduce the potential for injury. Further studies on preseason screening and in-season monitoring protocols, targeted exercise therapy, early surgical intervention, and potential biologic intervention are needed to determine the most effective methods of preventing groin injuries in athletes.
ABSTRACT
Soccer is the most popular sport in the world and has the fourth highest number of sports injuries. Hip and groin injuries account for 14% of soccer injuries and can be difficult to recognize and treat as they often require a high level of suspicion and advanced imaging. Groin pain can be separated into 3 categories: (1) defined clinical entities for groin pain (adductor-related, iliopsoas-related, inguinal-related [sports hernias/athletic pubalgia], and pubic-related groin pain), (2) hip-related groin pain (hip morphologic abnormalities, labral tears, and chondral injuries), and (3) other causes of groin pain. Conservative approaches are typically the first line of treatment, but operative intervention has been reported to result in higher rates of return to sport in athletes with hip-related and inguinal-related groin pain injuries. In patients with concurrent hip-related and inguinal-related groin pain, the failure to recognize the relationship and treat both conditions may result in lower rates of return to sport. Preseason screening programs can identify high-risk athletes, who may benefit from a targeted prevention program. Further study on exercise therapy, early surgical intervention, and potential biologic intervention are needed to determine the most effective methods of preventing groin injuries in athletes.
Continue to: Each year, the global audience for soccer grows...
Each year, the global audience for soccer grows. Soccer has long surpassed all other sports as the most popular sport in the world, reaching 3.2 billion viewers during the 2014 World Cup.1 In the latest Fédération Internationale de Football Association (FIFA) Big Count survey, the organization estimated that 265 million people are actively involved in soccer, accounting for approximately 4% of the world’s population.2 Moreover, the number of people playing soccer increased by 9.5% within 6 years after the previous Big Count Survey.2 In the United States, soccer accounts for the fourth most common cause of sports injuries next to basketball, exercise, and football with approximately 228,000 injuries per year.3 The total cost of treatment related to worldwide soccer injuries tops $30 billion.4 The most common body parts injured are the thigh (25%), knee (18%), and hip and/or groin (14%).5
Hip and groin injuries in soccer players can be separated into 3 main categories based on the Doha Agreement:6 (1) defined clinical entities for groin pain, (2) hip-related groin pain, and (3) other causes of groin pain in athletes. Defined clinical entities include adductor-related, iliopsoas-related, inguinal-related (sports hernia/athletic pubalgia), and pubic-related groin pain; while hip-related groin pain includes hip morphologic abnormalities, labral tears, and chondral injuries. Included in other causes of groin pain are injuries not clinically defined. The Doha Agreement has acknowledged that not all causes of groin pain fit into the classification system including injuries of the rectus femoris, but they will be included under defined clinical entities for groin pain in this review. While they are not a cause of groin pain, proximal hamstring and gluteal and piriformis injuries are important causes of posterior and lateral hip pain in soccer players and will also be covered in the first section of this review.
DEFINED CLINICAL ENTITIES FOR GROIN PAIN IN SOCCER ATHLETES
ADDUCTOR-RELATED GROIN PAIN
Acute groin pain in soccer players is most commonly caused by muscle strain.7 Of the muscle strains, 66% involve the adductor longus, 25% the iliopsoas, and 23% the rectus femoris.7 The Doha Agreement defines adductor-related groin pain as adductor tenderness and pain on resisted adduction.6 Adductor longus strains in soccer players are typically noncontact injuries (62.5%) and most commonly the result of kicking (40%).7-9 Many athletes will remember a pop at the time of the original injury.9 The combination of history and physical examination is usually sufficient for diagnosis; however, magnetic resonance imaging (MRI) may be helpful in complicated situations with a reported 86% sensitivity and 89% specificity.10 The average playing time lost is 2 weeks.5 Management includes rest, anti-inflammatory medication, physical therapy with core strengthening, and avoidance of aggressive stretching. While partial and distal avulsions can heal with conservative measures, proximal osseous and retracted avulsions of the adductor longus can be treated surgically.11
Continue to: ILIOPSOAS-RELATED GROIN PAIN...
ILIOPSOAS-RELATED GROIN PAIN
Iliopsoas strains account for 25% of acute groin strains and typically result from an impact that causes eccentric overload while kicking the ball.7,12 Iliopsoas-related groin pain is defined by the Doha Agreement as groin pain that is reproducible with resisted hip flexion or hip flexor stretch.6 Iliopsoas strains respond well to conservative treatment such as rest, anti-inflammatory medication, and physical therapy. Rarely do these athletes become surgical candidates in the acute setting. Chronic cases of iliopsoas pathology occasionally require an arthroscopic intervention.
INGUINAL-RELATED GROIN PAIN
Inguinal-related groin pain is one of the most misleading diagnoses in sports because of its poorly defined and under-researched nature. The varying nomenclature of this entity illustrates the heterogeneity and includes sports hernia,9,13-15 athletic pubalgia,16 core muscle injury,17 athletic hernia,18 Gilmore’s groin,15 osteitis pubis,19 sportsman’s hernia,20,21 sportsmen’s groin,22 symphysis syndrome,23 and inguinal disruption.24 It is important to realize that in inguinal-related groin pain, regardless of the nomenclature, there is no true hernia present. The Doha Agreement has defined inguinal-related groin pain as “pain in the location of the inguinal region with associated tenderness of the inguinal canal,” which “is more likely if the pain is aggravated with resistance testing of the abdominal muscles or on Valsalva/cough/sneeze.”6 The condition is a painful soft tissue injury in the groin or inguinal area, involving a constellation of various anatomic areas including the abdominal musculature, sacroiliac joint, neural structures, pubic symphysis, adductors, and hip joint. This may account for up to 50% of chronic groin pain.25,26
One important theory in the development of inguinal-related groin pain is its relationship with femoroacetabular impingement (FAI). Cadaver studies demonstrate that cam deformities cause a 35% increase in motion at the pubic symphysis altering the biomechanics of the adductors and abdominal musculature and, with repetitive stress, may lead to tearing or attenuation of the transversalis fascia, rectus abdominis, internal obliques, and/or external obliques.12,27,28 Another prevailing theory of this is that the increased pubic stress causes weakness in the posterior portion of the inguinal canal, which then stretches and entraps the genitofemoral, ilioinguinal, lateral femoral cutaneous, or obturator nerves, ultimately causing pain.28,29
Physical examination findings include pain over the conjoined tendon, pubic tubercle/symphysis (present in 22% of patients), adductor origin (36%), and inguinal ring.25,30 Pain with resisted sit-ups is present in 46% of patients and pain with coughing/Valsalva is present in 10%.25,30,31 Selective injections can be a critical part of the evaluation to differentiate inguinal-related groin pain from FAI, osteitis pubis, and adductor strains while helping to determine the appropriate treatment.25,32 The role of advanced imaging is unclear as the clinical entity is still uncertain and the standard imaging findings have not been definitively established.33 However, several studies have reported MRI findings suggestive of inguinal-related groin pain. One of the more common MRI findings is the “secondary cleft sign,” which requires injecting a dye into the pubic symphysis.34 Several studies have shown that the radiographic dye extravasates preferentially into the side where the groin symptoms exist and are thought to be secondary to micro-tearing at the common attachment of the musculotendinous structures to the anterior pubis.34,35 However, it should be noted that the lack of imaging findings does not exclude the possibility of inguinal-related groin pathology.
Initial treatment consists of rest, anti-inflammatory medication, injections, and physical therapy with core strengthening.25 A study by Paajanen and colleagues36 suggested that early surgical intervention may be preferred over conservative management in a randomized trial comparing physical therapy, injections, anti-inflammatory medication, and rest vs an extraperitoneal laparoscopic mesh repair behind the pubic symphysis. In the conservative group, 20% of athletes returned to sport at 1 month, 27% at 3 months, and 50% at 12 months.36 In comparison, the surgical group had 67% return to sport at 1 month, 90% at 3 months, and 97% at 12 months.36 If surgical management is chosen, there are a variety of surgical options including laparoscopy, open or mini-open repairs of the abdominal musculature/fascia or pelvic floor with and without mesh, neurolysis, and adductor release. Muschawek and Berger37described a series of 129 patients that had an open-suture repair of the posterior wall of the inguinal canal with 67% of professional athletes returning to sport within 2 weeks and 83.7% of athletes returning to sport overall. The rates of return to play are consistently 80% to 100% without demonstrated superiority of one technique over another up to this point.30
Continue to: PUBIC-RELATED GROIN PAIN...
PUBIC-RELATED GROIN PAIN
Pubic-related groin pain is defined as tenderness to palpation over the pubic symphysis and adjacent bone.6 Osteitis pubis is a chronic overuse injury characterized by localized pain to the pubic symphysis and is believed to be caused by repetitive microtrauma from a dynamic rotation of the sacroiliac joint with suggested imbalances between the rectus abdominis and the adductor musculature.12,38 In soccer players, the condition may be related to the constant torsional stresses of kicking, running, or twisting.12 If performed, radiographs often show lytic areas of the pubic symphysis, widening of the symphysis, sclerosis, and cystic changes, while bone marrow edema may be present on MRI.38Management consists of rest, anti-inflammatory medication, and corticosteroid injections with gentle stretching once asymptomatic.12,39
RECTUS FEMORIS INJURIES
The most common injury to the rectus femoris is a strain as a result of an eccentric overload while a soccer player is hit trying to extend his or her leg to kick a ball.12 In pediatric soccer athletes, an avulsion of the anterior inferior iliac spine from the direct head of the rectus femoris is the second most common avulsion injury.40 Radiographs are diagnostic and can help determine treatment. Most avulsions are minimally displaced and can be treated conservatively, but surgical intervention should be considered for an avulsion >2 cm.12
PROXIMAL HAMSTRING INJURIES
Proximal hamstring injuries are important causes of acute posterior hip pain and are caused by an eccentric overload in hip flexion and knee extension.25 In soccer players, the typical mechanism is that the planted leg slipping on the playing turf creates a sudden violent flexion of the hip with the knee in an extended position. While relatively uncommon, when a significant avulsion occurs in a professional athlete, surgical intervention is often necessary. In general, these injuries may involve partial or full avulsions off the ischial tuberosity or separation of the bony apophysis in pediatric athletes. A physical examination in the acute setting typically demonstrates massive posterior thigh ecchymosis, a palpable defect, and/or weakness with knee flexion. Imaging is helpful to confirm the diagnosis and evaluate for surgical repair. Radiographs may show a bony avulsion, which is more commonly seen in pediatric apophyseal avulsions. MRI can be used to differentiate a complete tear (involving all 3 tendons) vs a partial tear and evaluate for retraction of the tendon distally. Complete and partial tears of 2 tendons with retraction of >2 cm should be surgically repaired.25 Partial tears without tendon retraction may be treated conservatively with rest, anti-inflammatory medication, and physical therapy and then followed later by a hamstring prevention program.25 We have found that biologic augmentation with platelet-rich plasma can help accelerate healing in partial thickness injuries; however, the evidence is conflicting.
GLUTEAL INJURIES
Chronic overuse injuries of the gluteal musculature are common causes of lateral hip pain. Abductor overuse caused by weakness in the gluteus medius with a normal tensor fascia lata can cause pain with sitting and side-lying.25Overuse of the gluteal muscles with muscular imbalances along with increased tension on the iliotibial band can lead to greater trochanteric pain syndrome.25 A physical examination may demonstrate tenderness over the greater trochanter bursa and positive flexion, abduction, and external rotation testing.25 Abductor overuse syndrome and greater trochanteric pain syndrome are best treated with anti-inflammatory medication and physical therapy to balance the core/pelvic musculature.41
PIRIFORMIS INJURIES
Piriformis syndrome is a compressive neuropathy of the sciatic nerve. The mechanism of injury in the athlete is through a minor trauma to the buttock or pelvis.25,42,43 Presenting symptoms include pain with sitting and internal rotation of the hip.12 Zeren and colleagues42 published the only study that includes 2 cases of bilateral piriformis syndrome in professional soccer players. The diagnosis was confirmed with electromyography that was negative at rest and positive when measured after running.42 The athletes exhausted conservative treatment with physical therapy, anti-inflammatory medications, injections, and rest and were treated with surgical decompression.42 Both players returned to professional soccer after 6 months and played for an average of 7 years.42
Continue to: HIP-RELATED GROIN PAIN IN SOCCER ATHLETES...
HIP-RELATED GROIN PAIN IN SOCCER ATHLETES
Hip-related groin pain has garnered more attention in the last several years after being a previously underdiagnosed entity. One study found that practitioners treated groin pain in athletes for 7 months on average before recognizing that the pathology was intra-articular.44 FAI, labral tears, and chondral injuries are the major intra-articular pathologies that cause groin pain in athletes and ultimately impaired performance.45,46
FEMOROACETABULAR IMPINGEMENT
FAI is caused by pincer-type, cam-type, or combined-type deformities. Pincer lesions are defined as an increased acetabular overhang, while cam lesions are described as an increased bone at the femoral head/neck junction. These deformities in isolation or in combination cause decreased hip motion and increased contact pressures between the anterolateral acetabulum and femoral head-neck junction, which may ultimately lead to labral tears, chondral lesions, and osteoarthritis.47 During hip flexion, cam deformities impact the anterolateral acetabulum, preferentially causing articular cartilage damage, while sparing the labrum.25 Conversely, pincer deformities cause repetitive microtrauma to the labrum, crushing it between the acetabular rim and femoral neck with secondary damage to the articular cartilage.25 Over time, the damage to the labrum and articular cartilage may lead to premature osteoarthritis, which occurs at a much younger age in the athletic population.48
We know from previous studies that soccer athletes have a high prevalence of morphologic abnormalities of the hip, most commonly FAI. Gerhardt and colleagues49 documented the prevalence of hip morphologic abnormalities in elite soccer players and found abnormalities in 72% of men and over 50% of women. It should be noted that this series looked at asymptomatic athletes; however, it has been shown that hip dysmorphia is a risk factor for hip and groin injuries and may provide an opportunity for injury prevention strategies.50
Physical examination findings in FAI include decreased hip internal rotation and pain with provocative testing. Wyss and colleagues51 measured hip internal rotation in athletes with and without FAI. They found that the athletes with FAI have an average of 4° of internal rotation compared with that of the non-FAI athletes with 28°.51 A worsening internal rotation deficit has been linked to increasing severity of the deformity and when <20° was correlated with joint damage.51 Provocative testing has a high sensitivity with a recent meta-analysis demonstrating the most sensitive tests to be the anterior impingement test (flexion-adduction-internal rotation) with 94% to 99% sensitivity and the flexion-internal rotation test with 96% sensitivity.52 While provocative tests are sensitive, there is no current consensus on physical examination findings that are specific in the diagnosis of FAI.6 Diagnosis is made with both positive physical examination and radiographic morphologic findings (alpha angle >55°).33 Advanced imaging with an MRI arthrogram can be helpful in diagnosing underlying injuries such as labral tears in athletes presenting with compatible symptoms.
Symptomatic patients are typically treated surgically through either open or arthroscopic procedures, which have favorable and comparable functional results, biomechanics, and return to sport.53 In soccer players, return to sport at the professional level after arthroscopic surgery was found to be 96%.54 Players returned to sport on average 9.2 months postoperatively and played an average of 70 games after surgery.54
Continue to: LABRAL TEARS...
LABRAL TEARS
Labral tears present with groin pain, limited hip range of motion, and symptoms of catching, locking, and instability.25Causes of labral tears include trauma, FAI, hip dysplasia, capsular laxity, and degeneration.55 Labral tears rarely occur in isolation and have a high association (87%) with morphologic abnormalities of the hip, most commonly FAI and occasionally dysplasia.56,57 Physical examination findings include positive anterior impingement tests (flexion-adduction-internal rotation) in athletes with anterior labral tears and, less commonly, positive flexion, abduction, and external rotation tests for athletes with lateral and posterolateral labral tears.57 Radiographic imaging is used to evaluate for concurrent morphologic abnormalities of the hip, and MRI arthrogram is used to confirm the diagnosis of a labral tear with a sensitivity of 76% to 91%.58 Initial treatment consists of conservative treatment, which includes rest, anti-inflammatory medication, activity modification, and physical therapy. In patient refractory to conservative treatment, arthroscopic surgery is effective with high rates of return to sport.59 It is important to note that when treating labral tears surgically, any morphologic abnormality needs to be addressed to prevent recurrence of the tear.
CHONDRAL INJURIES
Focal chondral lesions in the hip are commonly found in athletes with FAI and labral tears during arthroscopic evaluation.60 Full-thickness defects and unstable flaps in weight-bearing areas are indications for surgical intervention with microfracture.60 There are no studies examining the efficacy of microfracture in isolation; however, Locks and colleagues54 have demonstrated a 96% return to professional soccer after an arthroscopic treatment for FAI and found that severe chondral damage with microfracture did not lengthen the return to sport.
RELATIONSHIP BETWEEN INGUINAL-RELATED GROIN PAIN AND FEMOROACETABULAR IMPINGEMENT
The altered biomechanics and restricted range of motion in athletes with FAI cause an increase in compensatory motion at the pelvis and lumbosacral areas, which may contribute to the development of inguinal-related groin pain, bursitis, adductor, and gluteal dysfunction.25 In athletes with concurrent intra-articular hip pathology and inguinal-related groin pain, treating 1 condition in isolation will result in poor results. Larson and colleagues61 found that when only inguinal-related groin pain or FAI were addressed, return to sport was only 25% and 50%, respectively, while concurrent surgical treatment resulted in a return to sport of 89%.
DISCUSSION AND FUTURE DIRECTIONS
Groin injuries in soccer players can cause significant decreases in athletic performance, result in lost playing time, and may ultimately need a surgical intervention. Efforts are underway to determine the role and efficacy of identifying high-risk athletes that may benefit from targeted prevention strategies. Wyles and colleagues48 identified adolescent athletes with hip internal rotation of <10° and found at 5-year follow-up that 95% had abnormal MRI findings compared with 54% in the age-matched control group. Wollin and colleagues62 developed an in-season screening protocol using adductor strength reductions of 15%, adductor/abductor strength ratio <0.9, and hip and groin outcome scores <75 as indicators of at-risk individuals. By employing preseason and in-season screening protocols, we can identify high-risk athletes for further workup and close follow-up throughout the season. Pelvic radiographs in these high-risk athletes may help us determine the presence of abnormalities in hip morphology, which would place an athlete into a high-risk group where prevention strategies could then be employed. There are no data available to determine the most effective prevention strategy at this time. However, levels II and III evidence exists indicating that exercise programs may reduce the incidence of groin injuries.63 Additional strategies, like limiting adolescent playing time similar to strategies employed in baseball pitches with pitch counts, could potentially reduce the potential for injury. Further studies on preseason screening and in-season monitoring protocols, targeted exercise therapy, early surgical intervention, and potential biologic intervention are needed to determine the most effective methods of preventing groin injuries in athletes.
1. Kantar Media. 2014 FIFA World Cup Brazil television audience report. https://resources.fifa.com/mm/document/affederation/tv/02/74/55/57/2014f...(draft5)(issuedate14.12.15)_neutral.pdf. Accessed March 20, 2018.
2. Fédération Internationale de Football Association. FIFA Big Count. http://www.fifa.com/mm/document/fifafacts/bcoffsurv/emaga_9384_10704.pdf. Published July 2007. Accessed March 20, 2018.
3. United States Consumer Product Safety Commission. Neiss data highlights - 2015. https://www.cpsc.gov/s3fs-public/2015 Neiss data highlights.pdf. Accessed March 20, 2018.
4. Hassabi M, Mohammad-Javad Mortazavi S, Giti MR, Hassabi M, Mansournia MA, Shapouran S. Injury profile of a professional soccer team in the premier league of Iran. Asian J Sports Med. 2010;1(4):201-208.
5. Ekstrand J, Hagglund M, Walden M. Injury incidence and injury patterns in professional football: the UEFA injury study. Br J Sports Med. 2011;45(7):553-558.
6. Weir A, Brukner P, Delahunt E, et al. Doha agreement meeting on terminology and definitions in groin pain in athletes. Br J Sports Med. 2015;49(12):768-774.
7. Serner A, Tol JL, Jomaah N, et al. Diagnosis of acute groin injuries: a prospective study of 110 athletes. Am J Sports Med. 2015;43(8):1857-1864. doi:10.1177/0363546515585123.
8. Eckard TG, Padua DA, Dompier TP, Dalton SL, Thorborg K, Kerr ZY. Epidemiology of hip flexor and hip adductor strains in national collegiate athletic association athletes, 2009/2010-2014/2015. Am J Sports Med. 2017;45(12):2713-2722. doi:10.1177/0363546517716179.
9. Hopkins JN, Brown W, Lee CA. Sports hernia: definition, evaluation, and treatment. JBJS Rev. 2017;5(9):e6. doi:10.2106/JBJS.RVW.17.00022.
10. Omar IM, Zoga AC, Kavanagh EC, et al. Athletic pubalgia and "sports hernia": optimal MR imaging technique and findings. Radiographics. 2008;28(5):1415-1438. doi:10.1148/rg.285075217.
11. Vogt S, Ansah P, Imhoff AB. Complete osseous avulsion of the adductor longus muscle: acute repair with three Wberwire suture anchors. Arch Orthop Trauma Surg. 2007;127:613-615. doi:10.1007/s00402-007-0328-5.
12. Anderson K, Strickland SM, Warren R. Hip and groin injuries in athletes. Am J Sports Med. 2001;29(4):521-533. doi:10.1177/03635465010290042501.
13. Choi HR, Elattar O, Dills VD, Busconi B. Return to play after sports hernia surgery. Clin Sports Med. 2016;35(4):621-636. doi:10.1016/j.csm.2016.05.007.
14. Garvey JF, Hazard H. Sports hernia or groin disruption injury? Chronic athletic groin pain: a retrospective study of 100 patients with long-term follow-up. Hernia. 2014;18(6):815-823. doi:10.1007/s10029-013-1161-0.
15. Gilmore J. Groin pain in the soccer athlete: fact, fiction, and treatment. Clin Sports Med. 1998;17(4):787-793, vii. doi:10.1016/S0278-5919(05)70119-8.
16. Cohen B, Kleinhenz D, Schiller J, Tabaddor R. Understanding athletic pubalgia: a review. R I Med J. 2016;99(10):31-35.
17. Ross JR, Stone RM, Larson CM. Core muscle injury/sports hernia/athletic pubalgia, and femoroacetabular impingement. Sports Med Arthrosc Rev. 2015;23(4):213-220. doi:10.1097/JSA.0000000000000083.
18. Swan KG Jr, Wolcott M. The athletic hernia: a systematic review. Clin Orthop Relat Res. 2007;455:78-87. doi:10.1097/BLO.0b013e31802eb3ea.
19. Matikainen M, Hermunen H, Paajanen H. Athletic pubalgia in females: predictive value of MRI in outcomes of endoscopic surgery. Orthop J Sports Med. 2017;5(8):2325967117720171. doi:10.1177/2325967117720171.
20. Garvey JF, Read JW, Turner A. Sportsman hernia: what can we do? Hernia. 2010;14(1):17-25. doi:10.1007/s10029-009-0611-1.
21. Paksoy M, Sekmen U. Sportsman hernia; the review of current diagnosis and treatment modalities. Ulusal Cerrahi Derg. 2016;32(2):122-129. doi:10.5152/UCD.2015.3132.
22. Pokorny H, Resinger C, Fischer I, et al. Fast early recovery after transabdominal preperitoneal repair in athletes with sportsman's groin: a prospective clinical cohort study. J Laparoendosc Adv Surg Tech A. 2017;27(3):272-276. doi:10.1089/lap.2016.0188.
23. Biedert RM, Warnke K, Meyer S. Symphysis syndrome in athletes: surgical treatment for chronic lower abdominal, groin, and adductor pain in athletes. Clin J Sport Med. 2003;13(5):278-284.
24. Sheen AJ, Stephenson BM, Lloyd DM, et al. 'Treatment of the sportsman's groin': British Hernia Society's 2014 position statement based on the Manchester Consensus Conference. Br J Sports Med. 2014;48(14):1079-1087.
25. Miller M, Thompson S. DeLee & Drez's Orthopaedic Sports Medicine. 4th ed. Philadelphia, PA: Elsevier/Saunders; 2015.
26. Lovell G. The diagnosis of chronic groin pain in athletes: a review of 189 cases. J Sci Med Sport. 1995;27:76-79.
27. Dimitrakopoulou A, Schilders E. Sportsman's hernia? An ambiguous term. J Hip Preserv Surg. 2016;3(1):16-22. doi:10.1093/jhps/hnv083.
28. Strosberg DS, Ellis TJ, Renton DB. The role of femoroacetabular impingement in core muscle injury/athletic pubalgia: diagnosis and management. Front Surg. 2016;3:6. doi:10.3389/fsurg.2016.00006.
29. Muschaweck U, Berger LM. Sportsmen's groin-diagnostic approach and treatment with the minimal repair technique: a single-center uncontrolled clinical review. Sports Health. 2010;2(3):216-221. doi:10.1177/1941738110367623.
30. Larson CM. Sports hernia/athletic pubalgia: evaluation and management. Sports Health. 2014;6(2):139-144. doi:10.1177/1941738114523557.
31. Meyers WC, Foley DP, Garrett WE, Lohnes JH, Mandlebaum BR. Management of severe lower abdominal or inguinal pain in high-performance athletes. Am J Sports Med. 2000; 28(1):2-8. doi:10.1177/03635465000280011501.
32. Gerhardt MB, Mandelbaum BR, Hutchinson WB. Ancillary modalities in the treatment of athletic groin Pain: Local Anesthetics, Corticosteroids, and Orthobiologics. In: Diduch DR, Brunt LM, eds. Sports Hernia and Athletic Pubalgia: Diagnosis and Treatment. Boston, MA: Springer US; 2014:183-187.
33. Notzli HP, Wyss TF, Stoecklin CH, et al. The contour of the femoral head-neck junction as a predictor for the risk of anterior impingement. J Bone Joint Surg Br. 2002;84(4):556-560.
34. Brennan D, O’Connell MJ, Ryan M, et al. Secondary cleft sign as a marker of injury in athletes with groin pain: MR image appearance and interpretation. Radiology. 2005;235(1):162-167. doi:10.1148/radiol.2351040045.
35. Byrne CA, Bowden DJ, Alkhayat A, Kavanagh EC, Eustace SJ. Sports-related groin pain secondary to symphysis pubis disorders: correlation between MRI findings and outcome after fluoroscopy-guided injection of steroid and local anesthetic. Am J Roentgenol. 2017;209(2):380-388. doi:10.2214/AJR.16.17578.
36. Paajanen H, Brinck T, Hermunen H, Airo I. Laparoscopic surgery for chronic groin pain in athletes is more effective than nonoperative treatment: a randomized clinical trial with magnetic resonance imaging of 60 patients with sportsman's hernia (athletic pubalgia). Surgery. 2011;150(1):99-107. doi:10.1016/j.surg.2011.02.016.
37. Muschaweck U, Berger L. Minimal repair technique of sportsmen's groin: an innovative open-suture repair to treat chronic inguinal pain. Hernia. 2010;14(1):27-33. doi:10.1007/s10029-009-0614-y.
38. Lynch TS, Bedi A, Larson CM. Athletic hip injuries. J Am Acad Orthop Surg. 2017;25(4):269-279. doi:10.5435/JAAOS-D-16-00171.
39. Holt MA, Keene JS, Graf BK, Helwig DC. Treatment of osteitis pubis in athletes. Results of corticosteroid injections. Am J Sports Med. 1995;23(5):601-606.doi:10.1177/036354659502300515.
40. Rossi F, Dragoni S. Acute avulsion fractures of the pelvis in adolescent competitive athletes: prevalence, location and sports distribution of 203 cases collected. Skeletal Radiol. 2001;30(3):127-131. doi: 10.1007/s002560000319.
41. Grumet RC, Frank RM, Slabaugh MA, Virkus WW, Bush-Joseph CA, Nho SJ. Lateral hip pain in an athletic population: differential diagnosis and treatment options. Sports Health. 2010;2(3):191-196. doi:10.1177/1941738110366829.
42. Zeren B, Canbek U, Oztekin HH, Imerci A, Akgun U. Bilateral piriformis syndrome in two elite soccer players: report of two cases. Orthop Traumatol Surg Res. 2015;101(8):987-990. doi:10.1016/j.otsr.2015.07.022.
43. Keskula DR, Tamburello M. Conservative management of piriformis syndrome. J Athl Train. 1992;27(2):102-110.
44. Byrd JW, Jones KS. Hip arthroscopy in athletes. Clin Sports Med. 2001;20(4):749-761.
45. Nepple JJ, Goljan P, Briggs KK, Garvey SE, Ryan M, Philippon MJ. Hip strength deficits in patients with symptomatic femoroacetabular impingement and labral tears. Arthroscopy.2015;31(11):2106-2111.
46. Mullins K, Hanlon M, Carton P. Differences in athletic performance between sportsmen with symptomatic femoroacetabular impingement and healthy controls. Clin J Sport Med.2018;28(4):370-376. doi:10.1097/JSM.0000000000000460.
47. Ganz R, Parvizi J, Beck M, Leunig M, Notzli H, Siebenrock KA. Femoroacetabular impingement: a cause for osteoarthritis of the hip. Clin Orthop Relat Res. 2003;417:112-120. doi:10.1097/01.blo.0000096804.78689.c2.
48. Wyles CC, Norambuena GA, Howe BM, et al. Cam deformities and limited hip range of motion are associated with early osteoarthritic changes in adolescent athletes: a prospective matched cohort study. Am J Sports Med. 2017;45(13):3036-3043. doi:10.1177/0363546517719460 .
49. Gerhardt MB, Romero AA, Silvers HJ, Harris DJ, Watanabe D, Mandelbaum BR. The prevalence of radiographic hip abnormalities in elite soccer players. Am J Sports Med. 2012;40(3):584-588. doi:10.1177/0363546511432711.
50. Larson CM, Ross JR, Kuhn AW, et al. Radiographic hip anatomy correlates with range of motion and symptoms in national hockey league players. Am J Sports Med. 2017;45(7):1633-1639. doi:10.1177/0363546517692542.
51. Wyss TF, Clark JM, Weishaupt D, Notzli HP. Correlation between internal rotation and bony anatomy in the hip. Clin Orthop Relat Res. 2007;460:152-158. doi:10.1097/BLO.0b013e3180399430.
52. Reiman MP, Goode AP, Cook CE, Holmich P, Thorborg K. Diagnostic accuracy of clinical tests for the diagnosis of hip femoroacetabular impingement/labral tear: a systematic review with meta-analysis. Br J Sports Med. 2015;49:811. doi:10.1136/bjsports-2014-094302.
53. Papalia R, Del Buono A, Franceschi F, Marinozzi A, Maffulli N, Denaro V. Femoroacetabular impingement syndrome management: arthroscopy or open surgery? Int Orthop. 2012;36(5):903-914. doi:10.1007/s00264-011-1443-z.
54. Locks R, Utsunomiya H, Briggs KK, McNamara S, Chahla J, Philippon MJ. Return to play after hip arthroscopic surgery for femoroacetabular impingement in professional soccer players. Am J Sports Med. 2018;46(2):273-279. doi:10.1177/0363546517738741.
55. Kelly BT, Weiland DE, Schenker ML, Philippon MJ. Arthroscopic labral repair in the hip: surgical technique and review of the literature. Arthroscopy. 2005;21(12):1496-1504. doi:10.1016/j.arthro.2005.08.013.
56. Wenger DE, Kendell KR, Miner MR, Trousdale RT. Acetabular labral tears rarely occur in the absence of bony abnormalities. Clin Orthop Relat Res. 2004;426:145-150. doi:10.1097/01.blo.0000136903.01368.20.
57. Domb BG, Hartigan DE, Perets I. Decision making for labral treatment in the hip: repair versus débridement versus reconstruction. J Am Acad Orthop Surg. 2017;25(3):e53-e62. doi:10.5435/JAAOS-D-16-00144.
58. Frank JS, Gambacorta PL, Eisner EA. Hip pathology in the adolescent athlete. J Am Acad Orthop Surg. 2013;21(11):665-674. doi:10.5435/JAAOS-21-11-665.
59. Singh PJ, O'Donnell JM. The outcome of hip arthroscopy in Australian football league players: a review of 27 hips. Arthroscopy. 2010;26(6):743-749. doi:10.1016/j.arthro.2009.10.010.
60. Crawford K, Philippon MJ, Sekiya JK, Rodkey WG, Steadman JR. Microfracture of the hip in athletes. Clin Sports Med. 2006;25(2):327-335. doi:10.1016/j.csm.2005.12.004.
61. Larson CM, Pierce BR, Giveans MR. Treatment of athletes with symptomatic intra-articular hip pathology and athletic pubalgia/sports hernia: a case series. Arthroscopy.2011;27(6):768-775. doi:10.1016/j.arthro.2011.01.018.
62. Wollin M, Thorborg K, Welvaert M, Pizzari T. In-season monitoring of hip and groin strength, health and function in elite youth soccer: implementing an early detection and management strategy over two consecutive seasons. J Sci Med Sport. 2018;21(10):988. doi:10.1016/j.jsams.2018.03.004.
63. Charlton PC, Drew MK, Mentiplay BF, Grimaldi A, Clark RA. Exercise interventions for the prevention and treatment of groin pain and injury in athletes: a critical and systematic review. Sports Med. 2017;47:2011. doi:10.1007/s40279-017-0742-y.
1. Kantar Media. 2014 FIFA World Cup Brazil television audience report. https://resources.fifa.com/mm/document/affederation/tv/02/74/55/57/2014f...(draft5)(issuedate14.12.15)_neutral.pdf. Accessed March 20, 2018.
2. Fédération Internationale de Football Association. FIFA Big Count. http://www.fifa.com/mm/document/fifafacts/bcoffsurv/emaga_9384_10704.pdf. Published July 2007. Accessed March 20, 2018.
3. United States Consumer Product Safety Commission. Neiss data highlights - 2015. https://www.cpsc.gov/s3fs-public/2015 Neiss data highlights.pdf. Accessed March 20, 2018.
4. Hassabi M, Mohammad-Javad Mortazavi S, Giti MR, Hassabi M, Mansournia MA, Shapouran S. Injury profile of a professional soccer team in the premier league of Iran. Asian J Sports Med. 2010;1(4):201-208.
5. Ekstrand J, Hagglund M, Walden M. Injury incidence and injury patterns in professional football: the UEFA injury study. Br J Sports Med. 2011;45(7):553-558.
6. Weir A, Brukner P, Delahunt E, et al. Doha agreement meeting on terminology and definitions in groin pain in athletes. Br J Sports Med. 2015;49(12):768-774.
7. Serner A, Tol JL, Jomaah N, et al. Diagnosis of acute groin injuries: a prospective study of 110 athletes. Am J Sports Med. 2015;43(8):1857-1864. doi:10.1177/0363546515585123.
8. Eckard TG, Padua DA, Dompier TP, Dalton SL, Thorborg K, Kerr ZY. Epidemiology of hip flexor and hip adductor strains in national collegiate athletic association athletes, 2009/2010-2014/2015. Am J Sports Med. 2017;45(12):2713-2722. doi:10.1177/0363546517716179.
9. Hopkins JN, Brown W, Lee CA. Sports hernia: definition, evaluation, and treatment. JBJS Rev. 2017;5(9):e6. doi:10.2106/JBJS.RVW.17.00022.
10. Omar IM, Zoga AC, Kavanagh EC, et al. Athletic pubalgia and "sports hernia": optimal MR imaging technique and findings. Radiographics. 2008;28(5):1415-1438. doi:10.1148/rg.285075217.
11. Vogt S, Ansah P, Imhoff AB. Complete osseous avulsion of the adductor longus muscle: acute repair with three Wberwire suture anchors. Arch Orthop Trauma Surg. 2007;127:613-615. doi:10.1007/s00402-007-0328-5.
12. Anderson K, Strickland SM, Warren R. Hip and groin injuries in athletes. Am J Sports Med. 2001;29(4):521-533. doi:10.1177/03635465010290042501.
13. Choi HR, Elattar O, Dills VD, Busconi B. Return to play after sports hernia surgery. Clin Sports Med. 2016;35(4):621-636. doi:10.1016/j.csm.2016.05.007.
14. Garvey JF, Hazard H. Sports hernia or groin disruption injury? Chronic athletic groin pain: a retrospective study of 100 patients with long-term follow-up. Hernia. 2014;18(6):815-823. doi:10.1007/s10029-013-1161-0.
15. Gilmore J. Groin pain in the soccer athlete: fact, fiction, and treatment. Clin Sports Med. 1998;17(4):787-793, vii. doi:10.1016/S0278-5919(05)70119-8.
16. Cohen B, Kleinhenz D, Schiller J, Tabaddor R. Understanding athletic pubalgia: a review. R I Med J. 2016;99(10):31-35.
17. Ross JR, Stone RM, Larson CM. Core muscle injury/sports hernia/athletic pubalgia, and femoroacetabular impingement. Sports Med Arthrosc Rev. 2015;23(4):213-220. doi:10.1097/JSA.0000000000000083.
18. Swan KG Jr, Wolcott M. The athletic hernia: a systematic review. Clin Orthop Relat Res. 2007;455:78-87. doi:10.1097/BLO.0b013e31802eb3ea.
19. Matikainen M, Hermunen H, Paajanen H. Athletic pubalgia in females: predictive value of MRI in outcomes of endoscopic surgery. Orthop J Sports Med. 2017;5(8):2325967117720171. doi:10.1177/2325967117720171.
20. Garvey JF, Read JW, Turner A. Sportsman hernia: what can we do? Hernia. 2010;14(1):17-25. doi:10.1007/s10029-009-0611-1.
21. Paksoy M, Sekmen U. Sportsman hernia; the review of current diagnosis and treatment modalities. Ulusal Cerrahi Derg. 2016;32(2):122-129. doi:10.5152/UCD.2015.3132.
22. Pokorny H, Resinger C, Fischer I, et al. Fast early recovery after transabdominal preperitoneal repair in athletes with sportsman's groin: a prospective clinical cohort study. J Laparoendosc Adv Surg Tech A. 2017;27(3):272-276. doi:10.1089/lap.2016.0188.
23. Biedert RM, Warnke K, Meyer S. Symphysis syndrome in athletes: surgical treatment for chronic lower abdominal, groin, and adductor pain in athletes. Clin J Sport Med. 2003;13(5):278-284.
24. Sheen AJ, Stephenson BM, Lloyd DM, et al. 'Treatment of the sportsman's groin': British Hernia Society's 2014 position statement based on the Manchester Consensus Conference. Br J Sports Med. 2014;48(14):1079-1087.
25. Miller M, Thompson S. DeLee & Drez's Orthopaedic Sports Medicine. 4th ed. Philadelphia, PA: Elsevier/Saunders; 2015.
26. Lovell G. The diagnosis of chronic groin pain in athletes: a review of 189 cases. J Sci Med Sport. 1995;27:76-79.
27. Dimitrakopoulou A, Schilders E. Sportsman's hernia? An ambiguous term. J Hip Preserv Surg. 2016;3(1):16-22. doi:10.1093/jhps/hnv083.
28. Strosberg DS, Ellis TJ, Renton DB. The role of femoroacetabular impingement in core muscle injury/athletic pubalgia: diagnosis and management. Front Surg. 2016;3:6. doi:10.3389/fsurg.2016.00006.
29. Muschaweck U, Berger LM. Sportsmen's groin-diagnostic approach and treatment with the minimal repair technique: a single-center uncontrolled clinical review. Sports Health. 2010;2(3):216-221. doi:10.1177/1941738110367623.
30. Larson CM. Sports hernia/athletic pubalgia: evaluation and management. Sports Health. 2014;6(2):139-144. doi:10.1177/1941738114523557.
31. Meyers WC, Foley DP, Garrett WE, Lohnes JH, Mandlebaum BR. Management of severe lower abdominal or inguinal pain in high-performance athletes. Am J Sports Med. 2000; 28(1):2-8. doi:10.1177/03635465000280011501.
32. Gerhardt MB, Mandelbaum BR, Hutchinson WB. Ancillary modalities in the treatment of athletic groin Pain: Local Anesthetics, Corticosteroids, and Orthobiologics. In: Diduch DR, Brunt LM, eds. Sports Hernia and Athletic Pubalgia: Diagnosis and Treatment. Boston, MA: Springer US; 2014:183-187.
33. Notzli HP, Wyss TF, Stoecklin CH, et al. The contour of the femoral head-neck junction as a predictor for the risk of anterior impingement. J Bone Joint Surg Br. 2002;84(4):556-560.
34. Brennan D, O’Connell MJ, Ryan M, et al. Secondary cleft sign as a marker of injury in athletes with groin pain: MR image appearance and interpretation. Radiology. 2005;235(1):162-167. doi:10.1148/radiol.2351040045.
35. Byrne CA, Bowden DJ, Alkhayat A, Kavanagh EC, Eustace SJ. Sports-related groin pain secondary to symphysis pubis disorders: correlation between MRI findings and outcome after fluoroscopy-guided injection of steroid and local anesthetic. Am J Roentgenol. 2017;209(2):380-388. doi:10.2214/AJR.16.17578.
36. Paajanen H, Brinck T, Hermunen H, Airo I. Laparoscopic surgery for chronic groin pain in athletes is more effective than nonoperative treatment: a randomized clinical trial with magnetic resonance imaging of 60 patients with sportsman's hernia (athletic pubalgia). Surgery. 2011;150(1):99-107. doi:10.1016/j.surg.2011.02.016.
37. Muschaweck U, Berger L. Minimal repair technique of sportsmen's groin: an innovative open-suture repair to treat chronic inguinal pain. Hernia. 2010;14(1):27-33. doi:10.1007/s10029-009-0614-y.
38. Lynch TS, Bedi A, Larson CM. Athletic hip injuries. J Am Acad Orthop Surg. 2017;25(4):269-279. doi:10.5435/JAAOS-D-16-00171.
39. Holt MA, Keene JS, Graf BK, Helwig DC. Treatment of osteitis pubis in athletes. Results of corticosteroid injections. Am J Sports Med. 1995;23(5):601-606.doi:10.1177/036354659502300515.
40. Rossi F, Dragoni S. Acute avulsion fractures of the pelvis in adolescent competitive athletes: prevalence, location and sports distribution of 203 cases collected. Skeletal Radiol. 2001;30(3):127-131. doi: 10.1007/s002560000319.
41. Grumet RC, Frank RM, Slabaugh MA, Virkus WW, Bush-Joseph CA, Nho SJ. Lateral hip pain in an athletic population: differential diagnosis and treatment options. Sports Health. 2010;2(3):191-196. doi:10.1177/1941738110366829.
42. Zeren B, Canbek U, Oztekin HH, Imerci A, Akgun U. Bilateral piriformis syndrome in two elite soccer players: report of two cases. Orthop Traumatol Surg Res. 2015;101(8):987-990. doi:10.1016/j.otsr.2015.07.022.
43. Keskula DR, Tamburello M. Conservative management of piriformis syndrome. J Athl Train. 1992;27(2):102-110.
44. Byrd JW, Jones KS. Hip arthroscopy in athletes. Clin Sports Med. 2001;20(4):749-761.
45. Nepple JJ, Goljan P, Briggs KK, Garvey SE, Ryan M, Philippon MJ. Hip strength deficits in patients with symptomatic femoroacetabular impingement and labral tears. Arthroscopy.2015;31(11):2106-2111.
46. Mullins K, Hanlon M, Carton P. Differences in athletic performance between sportsmen with symptomatic femoroacetabular impingement and healthy controls. Clin J Sport Med.2018;28(4):370-376. doi:10.1097/JSM.0000000000000460.
47. Ganz R, Parvizi J, Beck M, Leunig M, Notzli H, Siebenrock KA. Femoroacetabular impingement: a cause for osteoarthritis of the hip. Clin Orthop Relat Res. 2003;417:112-120. doi:10.1097/01.blo.0000096804.78689.c2.
48. Wyles CC, Norambuena GA, Howe BM, et al. Cam deformities and limited hip range of motion are associated with early osteoarthritic changes in adolescent athletes: a prospective matched cohort study. Am J Sports Med. 2017;45(13):3036-3043. doi:10.1177/0363546517719460 .
49. Gerhardt MB, Romero AA, Silvers HJ, Harris DJ, Watanabe D, Mandelbaum BR. The prevalence of radiographic hip abnormalities in elite soccer players. Am J Sports Med. 2012;40(3):584-588. doi:10.1177/0363546511432711.
50. Larson CM, Ross JR, Kuhn AW, et al. Radiographic hip anatomy correlates with range of motion and symptoms in national hockey league players. Am J Sports Med. 2017;45(7):1633-1639. doi:10.1177/0363546517692542.
51. Wyss TF, Clark JM, Weishaupt D, Notzli HP. Correlation between internal rotation and bony anatomy in the hip. Clin Orthop Relat Res. 2007;460:152-158. doi:10.1097/BLO.0b013e3180399430.
52. Reiman MP, Goode AP, Cook CE, Holmich P, Thorborg K. Diagnostic accuracy of clinical tests for the diagnosis of hip femoroacetabular impingement/labral tear: a systematic review with meta-analysis. Br J Sports Med. 2015;49:811. doi:10.1136/bjsports-2014-094302.
53. Papalia R, Del Buono A, Franceschi F, Marinozzi A, Maffulli N, Denaro V. Femoroacetabular impingement syndrome management: arthroscopy or open surgery? Int Orthop. 2012;36(5):903-914. doi:10.1007/s00264-011-1443-z.
54. Locks R, Utsunomiya H, Briggs KK, McNamara S, Chahla J, Philippon MJ. Return to play after hip arthroscopic surgery for femoroacetabular impingement in professional soccer players. Am J Sports Med. 2018;46(2):273-279. doi:10.1177/0363546517738741.
55. Kelly BT, Weiland DE, Schenker ML, Philippon MJ. Arthroscopic labral repair in the hip: surgical technique and review of the literature. Arthroscopy. 2005;21(12):1496-1504. doi:10.1016/j.arthro.2005.08.013.
56. Wenger DE, Kendell KR, Miner MR, Trousdale RT. Acetabular labral tears rarely occur in the absence of bony abnormalities. Clin Orthop Relat Res. 2004;426:145-150. doi:10.1097/01.blo.0000136903.01368.20.
57. Domb BG, Hartigan DE, Perets I. Decision making for labral treatment in the hip: repair versus débridement versus reconstruction. J Am Acad Orthop Surg. 2017;25(3):e53-e62. doi:10.5435/JAAOS-D-16-00144.
58. Frank JS, Gambacorta PL, Eisner EA. Hip pathology in the adolescent athlete. J Am Acad Orthop Surg. 2013;21(11):665-674. doi:10.5435/JAAOS-21-11-665.
59. Singh PJ, O'Donnell JM. The outcome of hip arthroscopy in Australian football league players: a review of 27 hips. Arthroscopy. 2010;26(6):743-749. doi:10.1016/j.arthro.2009.10.010.
60. Crawford K, Philippon MJ, Sekiya JK, Rodkey WG, Steadman JR. Microfracture of the hip in athletes. Clin Sports Med. 2006;25(2):327-335. doi:10.1016/j.csm.2005.12.004.
61. Larson CM, Pierce BR, Giveans MR. Treatment of athletes with symptomatic intra-articular hip pathology and athletic pubalgia/sports hernia: a case series. Arthroscopy.2011;27(6):768-775. doi:10.1016/j.arthro.2011.01.018.
62. Wollin M, Thorborg K, Welvaert M, Pizzari T. In-season monitoring of hip and groin strength, health and function in elite youth soccer: implementing an early detection and management strategy over two consecutive seasons. J Sci Med Sport. 2018;21(10):988. doi:10.1016/j.jsams.2018.03.004.
63. Charlton PC, Drew MK, Mentiplay BF, Grimaldi A, Clark RA. Exercise interventions for the prevention and treatment of groin pain and injury in athletes: a critical and systematic review. Sports Med. 2017;47:2011. doi:10.1007/s40279-017-0742-y.
TAKE-HOME POINTS
- Groin injuries in soccer players can cause significant decreases in athletic performance, result in lost playing time, and may ultimately need surgical intervention.
- Groin pain can be separated into 3 categories: (1) defined clinical entities for groin pain (adductor-related, iliopsoas-related, inguinal-related [sports hernias/athletic pubalgia], and pubic-related groin pain), (2) hip-related groin pain (hip morphologic abnormalities, labral tears, and chondral injuries), and (3) other causes of groin pain.
- Acute groin pain in soccer players is most commonly caused by muscle strain involving the adductor longus, the iliopsoas or the rectus femoris.
- Inguinal-related groin pain is a common cause of chronic groin pain and typically is the most challenging to treat with a complex pathophysiology and a high association with femoroacetabular impingement.
- Hip-related groin pain (femoroacetabular impingement, labral tears, and chondral injuries) usually respond well to surgical intervention and has high rates of return to sport.