User login
Drug-free inactive disease a feasible goal in JIA
Recent recommendations from an international task force advocating that have been backed up by research that finds the goal is a feasible one.
Published online in the Annals of the Rheumatic Diseases, the investigator-initiated, multicenter, randomized BEST for Kids study found that, regardless of initial specific treatments, after 24 months of treatment to target aimed at drug-free inactive disease, 71% of recent-onset patients with JIA had inactive disease (at a median onset of 9 months), and 39% were drug free.
The investigators, led by Petra Hissink Muller, MD, department of pediatrics, Leiden (the Netherlands) University Medical Center said previous studies of JIA supported the “window of opportunity” hypothesis in which disease optimally responds to treatment.
“In JIA, continuous treatment-to-target therapy in a tight control setting, with treatment adjustments based on frequent evaluations of disease activity, has not yet been studied. Recent recommendations agree that treatment to target should be implemented in daily practice,” they wrote.
The aim of the BEST for Kids study was to investigate which of three frequently used treatment-to-target strategies was most effective and safe.
Overall, 94 children (67% girls) with a median age of 9.1 years with new-onset (oligoarticular, juvenile psoriatic arthritis, or rheumatoid factor–negative polyarticular) JIA, without previous disease-modifying antirheumatic drug (DMARD) therapy and symptom duration of less than 18 months were enrolled in the study.
Median symptom duration was 7.5 months and median duration between diagnosis and inclusion in the study was 6 weeks. The investigators noted that two patients left the study during follow-up because of a revised diagnosis.
All study participants were randomized into three strategy arms:
- Initial treatment with conventional synthetic DMARD monotherapy (methotrexate [MTX] or sulfasalazine if preferred by treating physician), n = 32.
- Initial treatment with MTX and 6 weeks of tapered prednisolone (‘bridging therapy), n = 32).
- Initial treatment with MTX and etanercept, n = 30.
In case of inactive disease for at least 3 (oligoarticular disease) or 6 (polyarticular disease) consecutive months, DMARDs were tapered and stopped in all three arms. For combination therapy, etanercept was tapered to once every 2 weeks, followed by a 50% dose reduction, then halted. The dose of MTX or sulfasalazine was reduced by 25% per week to zero.
In case of a disease flare, the last discontinued drug and/or the last effective dose was reintroduced. The protocol did not allow prednisolone to be restarted, whereas etanercept could be restarted but not discontinued for a second time.
The primary outcome of the trial was time to inactive disease and time to flare (defined as the time between first moment of drug-free inactive disease (DFID) and the first arthritis flare as judged by the treating physician) after tapering and stopping all DMARD therapy. Secondary outcomes were adapted ACR Pediatric (ACR Pedi) 30/50/70/90 scores, functional ability, and adverse events.
Results showed that after 24 months inactive disease was achieved by more than 70% of the patients (71%, arm 1; 70%, arm 2; and 72%, arm 3).
Furthermore, drug-free inactive disease (DFID) was achieved by 59% of the cohort (54 of 92 patients; 45% of arm 1, 31% of arm 2, and 41% of arm 3), although the authors noted that early flares did occur in 14 patients and were successfully retreated, resulting in 39% of the patients in DFID at the 2-year study endpoint.
In the treatment of JIA,“we showed that tapering and discontinuation of treatment is a realistic goal. On the other hand, treatment to target resulted in a relatively high use of [biologic] DMARDs, greater than 50% of patients in all arms,” the investigators wrote.
Median time to inactive disease was 9 months in all arms (P = .30), and time to flare was also not significantly different among the groups (overall 3 months [3.0-6.8]; P = .7). The investigators noted that while overall flare rates (26%) were lower than the 37%-60% mentioned in previous cohorts, this finding could be a result of the limited follow-up time.
After 3 months of treatment, more patients who started with MTX and etanercept (arm 3) had achieved rapid improvement as determined by ACR Pedi 70 scores. However, the investigators noted that, because of treatment adjustments in cases of active disease, which were needed more often in arms 1 and 2 than in arm 3, ACR Pedi improvement scores were met in similar percentages of patients over time across the arms.
Adverse events were similar across treatment arms and were generally mild, involving mostly gastrointestinal complaints, upper respiratory tract and other infections, and general malaise.
Overall, the investigators concluded that DFID was a “feasible goal” in the treatment of children with JIA.
Limitations of the study included breaches of treatment protocol by physicians in patients across the three arms.“The physicians did not follow protocol for various reasons, mainly reluctance to intensify therapy based on shared decision making,” they wrote.
Another limitation was the small sample size, which the investigators said could obscure differences between groups that in a larger population might have become clear.
“Long-term follow-up of the BEST for Kids cohort, including radiology results, is initiated to investigate possible lasting positive results of treatment to target in JIA,” they said.
SOURCE: Hissink Muller P et al. Ann Rheum Dis. Oct 12. doi: 10.1136/annrheumdis-2018-213902.
Recent recommendations from an international task force advocating that have been backed up by research that finds the goal is a feasible one.
Published online in the Annals of the Rheumatic Diseases, the investigator-initiated, multicenter, randomized BEST for Kids study found that, regardless of initial specific treatments, after 24 months of treatment to target aimed at drug-free inactive disease, 71% of recent-onset patients with JIA had inactive disease (at a median onset of 9 months), and 39% were drug free.
The investigators, led by Petra Hissink Muller, MD, department of pediatrics, Leiden (the Netherlands) University Medical Center said previous studies of JIA supported the “window of opportunity” hypothesis in which disease optimally responds to treatment.
“In JIA, continuous treatment-to-target therapy in a tight control setting, with treatment adjustments based on frequent evaluations of disease activity, has not yet been studied. Recent recommendations agree that treatment to target should be implemented in daily practice,” they wrote.
The aim of the BEST for Kids study was to investigate which of three frequently used treatment-to-target strategies was most effective and safe.
Overall, 94 children (67% girls) with a median age of 9.1 years with new-onset (oligoarticular, juvenile psoriatic arthritis, or rheumatoid factor–negative polyarticular) JIA, without previous disease-modifying antirheumatic drug (DMARD) therapy and symptom duration of less than 18 months were enrolled in the study.
Median symptom duration was 7.5 months and median duration between diagnosis and inclusion in the study was 6 weeks. The investigators noted that two patients left the study during follow-up because of a revised diagnosis.
All study participants were randomized into three strategy arms:
- Initial treatment with conventional synthetic DMARD monotherapy (methotrexate [MTX] or sulfasalazine if preferred by treating physician), n = 32.
- Initial treatment with MTX and 6 weeks of tapered prednisolone (‘bridging therapy), n = 32).
- Initial treatment with MTX and etanercept, n = 30.
In case of inactive disease for at least 3 (oligoarticular disease) or 6 (polyarticular disease) consecutive months, DMARDs were tapered and stopped in all three arms. For combination therapy, etanercept was tapered to once every 2 weeks, followed by a 50% dose reduction, then halted. The dose of MTX or sulfasalazine was reduced by 25% per week to zero.
In case of a disease flare, the last discontinued drug and/or the last effective dose was reintroduced. The protocol did not allow prednisolone to be restarted, whereas etanercept could be restarted but not discontinued for a second time.
The primary outcome of the trial was time to inactive disease and time to flare (defined as the time between first moment of drug-free inactive disease (DFID) and the first arthritis flare as judged by the treating physician) after tapering and stopping all DMARD therapy. Secondary outcomes were adapted ACR Pediatric (ACR Pedi) 30/50/70/90 scores, functional ability, and adverse events.
Results showed that after 24 months inactive disease was achieved by more than 70% of the patients (71%, arm 1; 70%, arm 2; and 72%, arm 3).
Furthermore, drug-free inactive disease (DFID) was achieved by 59% of the cohort (54 of 92 patients; 45% of arm 1, 31% of arm 2, and 41% of arm 3), although the authors noted that early flares did occur in 14 patients and were successfully retreated, resulting in 39% of the patients in DFID at the 2-year study endpoint.
In the treatment of JIA,“we showed that tapering and discontinuation of treatment is a realistic goal. On the other hand, treatment to target resulted in a relatively high use of [biologic] DMARDs, greater than 50% of patients in all arms,” the investigators wrote.
Median time to inactive disease was 9 months in all arms (P = .30), and time to flare was also not significantly different among the groups (overall 3 months [3.0-6.8]; P = .7). The investigators noted that while overall flare rates (26%) were lower than the 37%-60% mentioned in previous cohorts, this finding could be a result of the limited follow-up time.
After 3 months of treatment, more patients who started with MTX and etanercept (arm 3) had achieved rapid improvement as determined by ACR Pedi 70 scores. However, the investigators noted that, because of treatment adjustments in cases of active disease, which were needed more often in arms 1 and 2 than in arm 3, ACR Pedi improvement scores were met in similar percentages of patients over time across the arms.
Adverse events were similar across treatment arms and were generally mild, involving mostly gastrointestinal complaints, upper respiratory tract and other infections, and general malaise.
Overall, the investigators concluded that DFID was a “feasible goal” in the treatment of children with JIA.
Limitations of the study included breaches of treatment protocol by physicians in patients across the three arms.“The physicians did not follow protocol for various reasons, mainly reluctance to intensify therapy based on shared decision making,” they wrote.
Another limitation was the small sample size, which the investigators said could obscure differences between groups that in a larger population might have become clear.
“Long-term follow-up of the BEST for Kids cohort, including radiology results, is initiated to investigate possible lasting positive results of treatment to target in JIA,” they said.
SOURCE: Hissink Muller P et al. Ann Rheum Dis. Oct 12. doi: 10.1136/annrheumdis-2018-213902.
Recent recommendations from an international task force advocating that have been backed up by research that finds the goal is a feasible one.
Published online in the Annals of the Rheumatic Diseases, the investigator-initiated, multicenter, randomized BEST for Kids study found that, regardless of initial specific treatments, after 24 months of treatment to target aimed at drug-free inactive disease, 71% of recent-onset patients with JIA had inactive disease (at a median onset of 9 months), and 39% were drug free.
The investigators, led by Petra Hissink Muller, MD, department of pediatrics, Leiden (the Netherlands) University Medical Center said previous studies of JIA supported the “window of opportunity” hypothesis in which disease optimally responds to treatment.
“In JIA, continuous treatment-to-target therapy in a tight control setting, with treatment adjustments based on frequent evaluations of disease activity, has not yet been studied. Recent recommendations agree that treatment to target should be implemented in daily practice,” they wrote.
The aim of the BEST for Kids study was to investigate which of three frequently used treatment-to-target strategies was most effective and safe.
Overall, 94 children (67% girls) with a median age of 9.1 years with new-onset (oligoarticular, juvenile psoriatic arthritis, or rheumatoid factor–negative polyarticular) JIA, without previous disease-modifying antirheumatic drug (DMARD) therapy and symptom duration of less than 18 months were enrolled in the study.
Median symptom duration was 7.5 months and median duration between diagnosis and inclusion in the study was 6 weeks. The investigators noted that two patients left the study during follow-up because of a revised diagnosis.
All study participants were randomized into three strategy arms:
- Initial treatment with conventional synthetic DMARD monotherapy (methotrexate [MTX] or sulfasalazine if preferred by treating physician), n = 32.
- Initial treatment with MTX and 6 weeks of tapered prednisolone (‘bridging therapy), n = 32).
- Initial treatment with MTX and etanercept, n = 30.
In case of inactive disease for at least 3 (oligoarticular disease) or 6 (polyarticular disease) consecutive months, DMARDs were tapered and stopped in all three arms. For combination therapy, etanercept was tapered to once every 2 weeks, followed by a 50% dose reduction, then halted. The dose of MTX or sulfasalazine was reduced by 25% per week to zero.
In case of a disease flare, the last discontinued drug and/or the last effective dose was reintroduced. The protocol did not allow prednisolone to be restarted, whereas etanercept could be restarted but not discontinued for a second time.
The primary outcome of the trial was time to inactive disease and time to flare (defined as the time between first moment of drug-free inactive disease (DFID) and the first arthritis flare as judged by the treating physician) after tapering and stopping all DMARD therapy. Secondary outcomes were adapted ACR Pediatric (ACR Pedi) 30/50/70/90 scores, functional ability, and adverse events.
Results showed that after 24 months inactive disease was achieved by more than 70% of the patients (71%, arm 1; 70%, arm 2; and 72%, arm 3).
Furthermore, drug-free inactive disease (DFID) was achieved by 59% of the cohort (54 of 92 patients; 45% of arm 1, 31% of arm 2, and 41% of arm 3), although the authors noted that early flares did occur in 14 patients and were successfully retreated, resulting in 39% of the patients in DFID at the 2-year study endpoint.
In the treatment of JIA,“we showed that tapering and discontinuation of treatment is a realistic goal. On the other hand, treatment to target resulted in a relatively high use of [biologic] DMARDs, greater than 50% of patients in all arms,” the investigators wrote.
Median time to inactive disease was 9 months in all arms (P = .30), and time to flare was also not significantly different among the groups (overall 3 months [3.0-6.8]; P = .7). The investigators noted that while overall flare rates (26%) were lower than the 37%-60% mentioned in previous cohorts, this finding could be a result of the limited follow-up time.
After 3 months of treatment, more patients who started with MTX and etanercept (arm 3) had achieved rapid improvement as determined by ACR Pedi 70 scores. However, the investigators noted that, because of treatment adjustments in cases of active disease, which were needed more often in arms 1 and 2 than in arm 3, ACR Pedi improvement scores were met in similar percentages of patients over time across the arms.
Adverse events were similar across treatment arms and were generally mild, involving mostly gastrointestinal complaints, upper respiratory tract and other infections, and general malaise.
Overall, the investigators concluded that DFID was a “feasible goal” in the treatment of children with JIA.
Limitations of the study included breaches of treatment protocol by physicians in patients across the three arms.“The physicians did not follow protocol for various reasons, mainly reluctance to intensify therapy based on shared decision making,” they wrote.
Another limitation was the small sample size, which the investigators said could obscure differences between groups that in a larger population might have become clear.
“Long-term follow-up of the BEST for Kids cohort, including radiology results, is initiated to investigate possible lasting positive results of treatment to target in JIA,” they said.
SOURCE: Hissink Muller P et al. Ann Rheum Dis. Oct 12. doi: 10.1136/annrheumdis-2018-213902.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: Drug-free inactive disease is a feasible goal in the treatment of children with JIA.
Major finding: After 24 months of treatment to target aimed at achieving drug-free inactive disease, 71% of recent-onset patients with JIA had inactive disease (median onset 9 months), and 39% were drug free.
Study details: A randomized, single-blinded study that assessed three treatment strategies in 94 patients with JIA who were DMARD naive.
Disclosures: The study was investigator initiated but received financial support from Pfizer. No competing interests were declared by the authors.
Source: Hissink Muller P et al. Ann Rheum Dis. 2018 Oct 12. doi: 10.1136/annrheumdis-2018-213902.
Breaking the glass ceiling: Women in pulmonary medicine face both barriers and opportunities
SAN ANTONIO – Women in medicine have made great strides in cracking the glass ceiling, but it’s not shattered yet, said Stephanie M. Levine, MD, FCCP, the incoming president of CHEST.
At a session on women in medicine at the annual meeting of the American College of Chest Physicians, Dr. Levine discussed the challenges of breaking through the metaphorical invisible barrier. The “glass ceiling” refers to multiple ways in which women lack equality with men in medicine: leadership roles, positions and titles, progress in academic medicine, gaps in salaries and compensation, and overall gender parity in specialties.
For example, according to data from the American Association of Medical Colleges for 2017-2018, women comprise 50% of medical school graduates but only 34% of the physician workforce and 22% of leadership roles. Women are 13% less likely to be promoted to professor. They receive salaries an average 21% lower than those of their male peers, said Dr. Levine, professor of medicine and director of the pulmonary/critical care fellowship program at the University of Texas, San Antonio.
Disparities exist particularly within specialties and subspecialties, she said. Women make 85% of what men earn in primary care but, in the specialties, only 75% of what men earn. Among active fellow trainees in the areas of medicine most represented by CHEST, one-third (32%) of critical care physicians and less than a third (29%) of pulmonary physicians are female.
Why the lag in specialty parity?
The reasons for these disparities are complex, Dr. Levine argued, but the problem is not insurmountable. They begin, in a sense, with the problem itself: When there are fewer mentors, role models, sponsors, and leaders, and less overall representation of women in the first place, it is harder for women to advance.
One male audience member, for example, asked how his department could recruit more women, because most turned down interviews despite the fact that more women than men were being invited. “How many women are in your leadership?” Dr. Levine asked. He acknowledged that there were none – and therein lies the likely problem. Applicants are looking for female representation in leadership.
Gender bias and discrimination certainly play a role among peers, leadership, and even patients. Patients referring to female physicians by their first names and asking questions such as “Are you my nurse?” are subtle but cutting examples of the ways in which they reveal implicit bias and reinforce gender stereotypes, Dr. Levine said to weary nods of agreement among the attendees.
Implicit, unconscious bias is also built into the culture of a place and the way things have always been done. Lack of equity in salary, space to work, support, and promotion all compound one another. Work-life integration challenges often do not favor women. Studies have shown that in the hiring process, CVs with female names do not receive as much attention as do CVs with male names, Dr. Levine noted.
Some of the challenges lie with the way women themselves do or do not advocate for themselves. Research has long shown that women do not negotiate as well – or at all – compared with men. Women tend to be less aggressive in seeking higher compensation and leadership roles, possibly because of existing implicit bias against female assertiveness in general.
The catch-22 is that being more assertive or direct can lead others to interpret a woman as being rude or brusk, as one audience member noted when she described how colleagues perceive her simple, direct tone as seeming “upset.”
Conscious bias remains alive and well: The stereotypes that women are caretakers and men are take-charge dominators persist and can reinforce gender disparities in leadership roles.
Women also must make calculations and trade-offs between their academic promotion clocks and their biologic clocks, Dr. Levine explained.
“The 30s are great for both academic and biologic productivity,” she told attendees. The typical age for a person’s first National Institutes of Health Research Project grant (R01) is in the early 40s.
How to improve gender equality
Women bring diverse skills and perspectives to the table, Dr. Levine explained. Women tend to have stronger collaborative skills and greater compassion and empathy, for example. They tend to be less hierarchal and better at mentoring and empowerment, she said.
There are many ways to poke more cracks in the ceiling, starting with diversity and inclusion officers who make it a priority to focus on parity. Formal programs can educate staff and colleagues about implicit bias so that they might more easily recognize it when it kicks in, and training for gatekeepers can lead to more proportional hiring of women at every level.
Institutions should review their policies – salary inequities, diversity in promotion, processes for selecting leaders – and set formal interventional goals that are then evaluated in honest, documented annual reviews.
Some of these policies should address work-life balance as well: Offering part-time and flexible work options during early child-rearing years helps not only mothers, but also fathers who are now taking a more active role in parenting. Slowing or prorating the promotion clock can help those building families, and shifting meetings away from times such as 7:00 a.m. and 6:00 p.m. allow mothers and fathers alike to get their kids to and from school and attend children’s events.
Sponsorship of women is an important strategy in breaking the glass ceiling, Dr. Levine said. Sponsors can support women with untapped leadership potential and do the necessary networking and introductions that help make that advance happen. And it must be done by sponsors with power and influence, including men, Dr. Levine said.
Men can play important roles in promoting gender parity by suggesting women for key roles, leadership positions, and committees and also notifying women of upcoming opportunities, such as editorial board spots and other hot jobs. For women who aspire to be leaders, men can seek to convey leadership skills that may be needed to chair committees and other groups. Search committees need to expand beyond looking for “token women,” she said.
Dr. Levine illustrated her address with her own story. She described how many of these strategies had helped her career and how many male supervisors, mentors, and colleagues helped her, including introducing her to other male leaders who then offered her opportunities to contribute to the American College of Chest Physicians. She ran for CHEST president twice before being elected on her third run in September. She is the fifth woman to lead CHEST.
“Don’t give up,” she encouraged women in the audience, telling them to advocate for themselves and to encourage, mentor, and sponsor their female fellows and junior faculty.
“This will result in closing the gaps and will help women achieve leadership roles and competitive salaries as well as work-life integration,” Dr. Levine said.
SAN ANTONIO – Women in medicine have made great strides in cracking the glass ceiling, but it’s not shattered yet, said Stephanie M. Levine, MD, FCCP, the incoming president of CHEST.
At a session on women in medicine at the annual meeting of the American College of Chest Physicians, Dr. Levine discussed the challenges of breaking through the metaphorical invisible barrier. The “glass ceiling” refers to multiple ways in which women lack equality with men in medicine: leadership roles, positions and titles, progress in academic medicine, gaps in salaries and compensation, and overall gender parity in specialties.
For example, according to data from the American Association of Medical Colleges for 2017-2018, women comprise 50% of medical school graduates but only 34% of the physician workforce and 22% of leadership roles. Women are 13% less likely to be promoted to professor. They receive salaries an average 21% lower than those of their male peers, said Dr. Levine, professor of medicine and director of the pulmonary/critical care fellowship program at the University of Texas, San Antonio.
Disparities exist particularly within specialties and subspecialties, she said. Women make 85% of what men earn in primary care but, in the specialties, only 75% of what men earn. Among active fellow trainees in the areas of medicine most represented by CHEST, one-third (32%) of critical care physicians and less than a third (29%) of pulmonary physicians are female.
Why the lag in specialty parity?
The reasons for these disparities are complex, Dr. Levine argued, but the problem is not insurmountable. They begin, in a sense, with the problem itself: When there are fewer mentors, role models, sponsors, and leaders, and less overall representation of women in the first place, it is harder for women to advance.
One male audience member, for example, asked how his department could recruit more women, because most turned down interviews despite the fact that more women than men were being invited. “How many women are in your leadership?” Dr. Levine asked. He acknowledged that there were none – and therein lies the likely problem. Applicants are looking for female representation in leadership.
Gender bias and discrimination certainly play a role among peers, leadership, and even patients. Patients referring to female physicians by their first names and asking questions such as “Are you my nurse?” are subtle but cutting examples of the ways in which they reveal implicit bias and reinforce gender stereotypes, Dr. Levine said to weary nods of agreement among the attendees.
Implicit, unconscious bias is also built into the culture of a place and the way things have always been done. Lack of equity in salary, space to work, support, and promotion all compound one another. Work-life integration challenges often do not favor women. Studies have shown that in the hiring process, CVs with female names do not receive as much attention as do CVs with male names, Dr. Levine noted.
Some of the challenges lie with the way women themselves do or do not advocate for themselves. Research has long shown that women do not negotiate as well – or at all – compared with men. Women tend to be less aggressive in seeking higher compensation and leadership roles, possibly because of existing implicit bias against female assertiveness in general.
The catch-22 is that being more assertive or direct can lead others to interpret a woman as being rude or brusk, as one audience member noted when she described how colleagues perceive her simple, direct tone as seeming “upset.”
Conscious bias remains alive and well: The stereotypes that women are caretakers and men are take-charge dominators persist and can reinforce gender disparities in leadership roles.
Women also must make calculations and trade-offs between their academic promotion clocks and their biologic clocks, Dr. Levine explained.
“The 30s are great for both academic and biologic productivity,” she told attendees. The typical age for a person’s first National Institutes of Health Research Project grant (R01) is in the early 40s.
How to improve gender equality
Women bring diverse skills and perspectives to the table, Dr. Levine explained. Women tend to have stronger collaborative skills and greater compassion and empathy, for example. They tend to be less hierarchal and better at mentoring and empowerment, she said.
There are many ways to poke more cracks in the ceiling, starting with diversity and inclusion officers who make it a priority to focus on parity. Formal programs can educate staff and colleagues about implicit bias so that they might more easily recognize it when it kicks in, and training for gatekeepers can lead to more proportional hiring of women at every level.
Institutions should review their policies – salary inequities, diversity in promotion, processes for selecting leaders – and set formal interventional goals that are then evaluated in honest, documented annual reviews.
Some of these policies should address work-life balance as well: Offering part-time and flexible work options during early child-rearing years helps not only mothers, but also fathers who are now taking a more active role in parenting. Slowing or prorating the promotion clock can help those building families, and shifting meetings away from times such as 7:00 a.m. and 6:00 p.m. allow mothers and fathers alike to get their kids to and from school and attend children’s events.
Sponsorship of women is an important strategy in breaking the glass ceiling, Dr. Levine said. Sponsors can support women with untapped leadership potential and do the necessary networking and introductions that help make that advance happen. And it must be done by sponsors with power and influence, including men, Dr. Levine said.
Men can play important roles in promoting gender parity by suggesting women for key roles, leadership positions, and committees and also notifying women of upcoming opportunities, such as editorial board spots and other hot jobs. For women who aspire to be leaders, men can seek to convey leadership skills that may be needed to chair committees and other groups. Search committees need to expand beyond looking for “token women,” she said.
Dr. Levine illustrated her address with her own story. She described how many of these strategies had helped her career and how many male supervisors, mentors, and colleagues helped her, including introducing her to other male leaders who then offered her opportunities to contribute to the American College of Chest Physicians. She ran for CHEST president twice before being elected on her third run in September. She is the fifth woman to lead CHEST.
“Don’t give up,” she encouraged women in the audience, telling them to advocate for themselves and to encourage, mentor, and sponsor their female fellows and junior faculty.
“This will result in closing the gaps and will help women achieve leadership roles and competitive salaries as well as work-life integration,” Dr. Levine said.
SAN ANTONIO – Women in medicine have made great strides in cracking the glass ceiling, but it’s not shattered yet, said Stephanie M. Levine, MD, FCCP, the incoming president of CHEST.
At a session on women in medicine at the annual meeting of the American College of Chest Physicians, Dr. Levine discussed the challenges of breaking through the metaphorical invisible barrier. The “glass ceiling” refers to multiple ways in which women lack equality with men in medicine: leadership roles, positions and titles, progress in academic medicine, gaps in salaries and compensation, and overall gender parity in specialties.
For example, according to data from the American Association of Medical Colleges for 2017-2018, women comprise 50% of medical school graduates but only 34% of the physician workforce and 22% of leadership roles. Women are 13% less likely to be promoted to professor. They receive salaries an average 21% lower than those of their male peers, said Dr. Levine, professor of medicine and director of the pulmonary/critical care fellowship program at the University of Texas, San Antonio.
Disparities exist particularly within specialties and subspecialties, she said. Women make 85% of what men earn in primary care but, in the specialties, only 75% of what men earn. Among active fellow trainees in the areas of medicine most represented by CHEST, one-third (32%) of critical care physicians and less than a third (29%) of pulmonary physicians are female.
Why the lag in specialty parity?
The reasons for these disparities are complex, Dr. Levine argued, but the problem is not insurmountable. They begin, in a sense, with the problem itself: When there are fewer mentors, role models, sponsors, and leaders, and less overall representation of women in the first place, it is harder for women to advance.
One male audience member, for example, asked how his department could recruit more women, because most turned down interviews despite the fact that more women than men were being invited. “How many women are in your leadership?” Dr. Levine asked. He acknowledged that there were none – and therein lies the likely problem. Applicants are looking for female representation in leadership.
Gender bias and discrimination certainly play a role among peers, leadership, and even patients. Patients referring to female physicians by their first names and asking questions such as “Are you my nurse?” are subtle but cutting examples of the ways in which they reveal implicit bias and reinforce gender stereotypes, Dr. Levine said to weary nods of agreement among the attendees.
Implicit, unconscious bias is also built into the culture of a place and the way things have always been done. Lack of equity in salary, space to work, support, and promotion all compound one another. Work-life integration challenges often do not favor women. Studies have shown that in the hiring process, CVs with female names do not receive as much attention as do CVs with male names, Dr. Levine noted.
Some of the challenges lie with the way women themselves do or do not advocate for themselves. Research has long shown that women do not negotiate as well – or at all – compared with men. Women tend to be less aggressive in seeking higher compensation and leadership roles, possibly because of existing implicit bias against female assertiveness in general.
The catch-22 is that being more assertive or direct can lead others to interpret a woman as being rude or brusk, as one audience member noted when she described how colleagues perceive her simple, direct tone as seeming “upset.”
Conscious bias remains alive and well: The stereotypes that women are caretakers and men are take-charge dominators persist and can reinforce gender disparities in leadership roles.
Women also must make calculations and trade-offs between their academic promotion clocks and their biologic clocks, Dr. Levine explained.
“The 30s are great for both academic and biologic productivity,” she told attendees. The typical age for a person’s first National Institutes of Health Research Project grant (R01) is in the early 40s.
How to improve gender equality
Women bring diverse skills and perspectives to the table, Dr. Levine explained. Women tend to have stronger collaborative skills and greater compassion and empathy, for example. They tend to be less hierarchal and better at mentoring and empowerment, she said.
There are many ways to poke more cracks in the ceiling, starting with diversity and inclusion officers who make it a priority to focus on parity. Formal programs can educate staff and colleagues about implicit bias so that they might more easily recognize it when it kicks in, and training for gatekeepers can lead to more proportional hiring of women at every level.
Institutions should review their policies – salary inequities, diversity in promotion, processes for selecting leaders – and set formal interventional goals that are then evaluated in honest, documented annual reviews.
Some of these policies should address work-life balance as well: Offering part-time and flexible work options during early child-rearing years helps not only mothers, but also fathers who are now taking a more active role in parenting. Slowing or prorating the promotion clock can help those building families, and shifting meetings away from times such as 7:00 a.m. and 6:00 p.m. allow mothers and fathers alike to get their kids to and from school and attend children’s events.
Sponsorship of women is an important strategy in breaking the glass ceiling, Dr. Levine said. Sponsors can support women with untapped leadership potential and do the necessary networking and introductions that help make that advance happen. And it must be done by sponsors with power and influence, including men, Dr. Levine said.
Men can play important roles in promoting gender parity by suggesting women for key roles, leadership positions, and committees and also notifying women of upcoming opportunities, such as editorial board spots and other hot jobs. For women who aspire to be leaders, men can seek to convey leadership skills that may be needed to chair committees and other groups. Search committees need to expand beyond looking for “token women,” she said.
Dr. Levine illustrated her address with her own story. She described how many of these strategies had helped her career and how many male supervisors, mentors, and colleagues helped her, including introducing her to other male leaders who then offered her opportunities to contribute to the American College of Chest Physicians. She ran for CHEST president twice before being elected on her third run in September. She is the fifth woman to lead CHEST.
“Don’t give up,” she encouraged women in the audience, telling them to advocate for themselves and to encourage, mentor, and sponsor their female fellows and junior faculty.
“This will result in closing the gaps and will help women achieve leadership roles and competitive salaries as well as work-life integration,” Dr. Levine said.
REPORTING FROM CHEST 2018
Top AGA Community patient cases
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community (https://community.gastro.org/discussions) to seek advice from colleagues about therapy and disease management options, best practices and diagnoses.
In case you missed it, here are the most popular clinical discussions shared in the forum recently:
1. Addressing early-onset CRC
With the recommendation by the American Cancer Society to start colorectal cancer screening at 45, Dr. Samir Gupta and Dr. Peter Liang led a hearty discussion on the intended and unintended consequences of widespread implementation of these recommendations.
2. Surveillance colonoscopies in IBD patients
The question “are GIs doing too many surveillance colonoscopies in IBD patients” evolved into a call for more clinical guidance on the topic. IBD experts, AGA President Dr. David Lieberman, and the AGA Guidelines and Clinical Practice Update Committees tackle next steps and recommendations.
3. Patient case: severe colitis in asymptomatic patient
When a 51-year-old patient was seen for a colonoscopy screening, subsequent biopsies revealed severe active chronic colitis with lymphoplasmacytic infiltrate, crypts, and crypt abscesses and no granulomas. Would you treat as ulcerative colitis or wait?
4. Patient case: IBD patient with steroid dependency
A 35-year-old female who was seen for refractory diarrhea and cramps tested positive for perinuclear antineutrophil antibodies cytoplasmic (pANCA). Her symptoms resolved after she received prednisone for an unrelated issue. The physician asks: is a low dose of prednisone “safer” than Remicade?
More clinical cases and discussions are at https://community.gastro.org/discussions.
ginews@gastro.org
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community (https://community.gastro.org/discussions) to seek advice from colleagues about therapy and disease management options, best practices and diagnoses.
In case you missed it, here are the most popular clinical discussions shared in the forum recently:
1. Addressing early-onset CRC
With the recommendation by the American Cancer Society to start colorectal cancer screening at 45, Dr. Samir Gupta and Dr. Peter Liang led a hearty discussion on the intended and unintended consequences of widespread implementation of these recommendations.
2. Surveillance colonoscopies in IBD patients
The question “are GIs doing too many surveillance colonoscopies in IBD patients” evolved into a call for more clinical guidance on the topic. IBD experts, AGA President Dr. David Lieberman, and the AGA Guidelines and Clinical Practice Update Committees tackle next steps and recommendations.
3. Patient case: severe colitis in asymptomatic patient
When a 51-year-old patient was seen for a colonoscopy screening, subsequent biopsies revealed severe active chronic colitis with lymphoplasmacytic infiltrate, crypts, and crypt abscesses and no granulomas. Would you treat as ulcerative colitis or wait?
4. Patient case: IBD patient with steroid dependency
A 35-year-old female who was seen for refractory diarrhea and cramps tested positive for perinuclear antineutrophil antibodies cytoplasmic (pANCA). Her symptoms resolved after she received prednisone for an unrelated issue. The physician asks: is a low dose of prednisone “safer” than Remicade?
More clinical cases and discussions are at https://community.gastro.org/discussions.
ginews@gastro.org
Physicians with difficult patient scenarios regularly bring their questions to the AGA Community (https://community.gastro.org/discussions) to seek advice from colleagues about therapy and disease management options, best practices and diagnoses.
In case you missed it, here are the most popular clinical discussions shared in the forum recently:
1. Addressing early-onset CRC
With the recommendation by the American Cancer Society to start colorectal cancer screening at 45, Dr. Samir Gupta and Dr. Peter Liang led a hearty discussion on the intended and unintended consequences of widespread implementation of these recommendations.
2. Surveillance colonoscopies in IBD patients
The question “are GIs doing too many surveillance colonoscopies in IBD patients” evolved into a call for more clinical guidance on the topic. IBD experts, AGA President Dr. David Lieberman, and the AGA Guidelines and Clinical Practice Update Committees tackle next steps and recommendations.
3. Patient case: severe colitis in asymptomatic patient
When a 51-year-old patient was seen for a colonoscopy screening, subsequent biopsies revealed severe active chronic colitis with lymphoplasmacytic infiltrate, crypts, and crypt abscesses and no granulomas. Would you treat as ulcerative colitis or wait?
4. Patient case: IBD patient with steroid dependency
A 35-year-old female who was seen for refractory diarrhea and cramps tested positive for perinuclear antineutrophil antibodies cytoplasmic (pANCA). Her symptoms resolved after she received prednisone for an unrelated issue. The physician asks: is a low dose of prednisone “safer” than Remicade?
More clinical cases and discussions are at https://community.gastro.org/discussions.
ginews@gastro.org
AGA advocates on Capitol Hill
Thank you to members who met with their congressperson and who participated in Virtual Advocacy Day.
Advocates met with House and Senate offices to push for passage of the Removing Barriers to Colorectal Cancer Screening Act, legislation that waives the coinsurance for screening colonoscopies that become therapeutic and has broad, bipartisan and bicameral support. They made the argument that paying for prevention services saves the government money in the long term by preventing treatment costs on cancer treatment.
H.R. 2077, the Restoring Patient’s Voice Act, addresses step therapy protocols that threaten the physician-patient relationship and delay timely treatment to care. Support for the legislation is growing and our advocates were able to relay experiences they have encountered with their patients’ care being delayed and also the administrative burden this policy places on practices.
Katherine Clark, D-Mass., a member of the House Labor-HHS Appropriations Subcommittee, met with our advocates and let them know that the House-Senate conferees agreed to the $2 billion increase in NIH funding in the final bill. Rep. Clark is a strong supporter of NIH and called it the “pillar of our economy.” AGA members encouraged their legislators to support the final Labor-HHS package that includes this $2 billion increase, which amounts to a 5.5% increase. The Senate recently approved the final agreement on Labor-HHS for fiscal year 2019 and we call on the House to follow suit.
AGA appreciates all those advocates who took time out of their busy schedules to advocate on behalf of their colleagues and patients. We also appreciate those who took time to participate in Virtual Advocacy Day. Remember, if we don’t advocate for GI, no one will.
To learn more about how you can get involved visit www.gastro.org/advocacy.
Thank you to members who met with their congressperson and who participated in Virtual Advocacy Day.
Advocates met with House and Senate offices to push for passage of the Removing Barriers to Colorectal Cancer Screening Act, legislation that waives the coinsurance for screening colonoscopies that become therapeutic and has broad, bipartisan and bicameral support. They made the argument that paying for prevention services saves the government money in the long term by preventing treatment costs on cancer treatment.
H.R. 2077, the Restoring Patient’s Voice Act, addresses step therapy protocols that threaten the physician-patient relationship and delay timely treatment to care. Support for the legislation is growing and our advocates were able to relay experiences they have encountered with their patients’ care being delayed and also the administrative burden this policy places on practices.
Katherine Clark, D-Mass., a member of the House Labor-HHS Appropriations Subcommittee, met with our advocates and let them know that the House-Senate conferees agreed to the $2 billion increase in NIH funding in the final bill. Rep. Clark is a strong supporter of NIH and called it the “pillar of our economy.” AGA members encouraged their legislators to support the final Labor-HHS package that includes this $2 billion increase, which amounts to a 5.5% increase. The Senate recently approved the final agreement on Labor-HHS for fiscal year 2019 and we call on the House to follow suit.
AGA appreciates all those advocates who took time out of their busy schedules to advocate on behalf of their colleagues and patients. We also appreciate those who took time to participate in Virtual Advocacy Day. Remember, if we don’t advocate for GI, no one will.
To learn more about how you can get involved visit www.gastro.org/advocacy.
Thank you to members who met with their congressperson and who participated in Virtual Advocacy Day.
Advocates met with House and Senate offices to push for passage of the Removing Barriers to Colorectal Cancer Screening Act, legislation that waives the coinsurance for screening colonoscopies that become therapeutic and has broad, bipartisan and bicameral support. They made the argument that paying for prevention services saves the government money in the long term by preventing treatment costs on cancer treatment.
H.R. 2077, the Restoring Patient’s Voice Act, addresses step therapy protocols that threaten the physician-patient relationship and delay timely treatment to care. Support for the legislation is growing and our advocates were able to relay experiences they have encountered with their patients’ care being delayed and also the administrative burden this policy places on practices.
Katherine Clark, D-Mass., a member of the House Labor-HHS Appropriations Subcommittee, met with our advocates and let them know that the House-Senate conferees agreed to the $2 billion increase in NIH funding in the final bill. Rep. Clark is a strong supporter of NIH and called it the “pillar of our economy.” AGA members encouraged their legislators to support the final Labor-HHS package that includes this $2 billion increase, which amounts to a 5.5% increase. The Senate recently approved the final agreement on Labor-HHS for fiscal year 2019 and we call on the House to follow suit.
AGA appreciates all those advocates who took time out of their busy schedules to advocate on behalf of their colleagues and patients. We also appreciate those who took time to participate in Virtual Advocacy Day. Remember, if we don’t advocate for GI, no one will.
To learn more about how you can get involved visit www.gastro.org/advocacy.
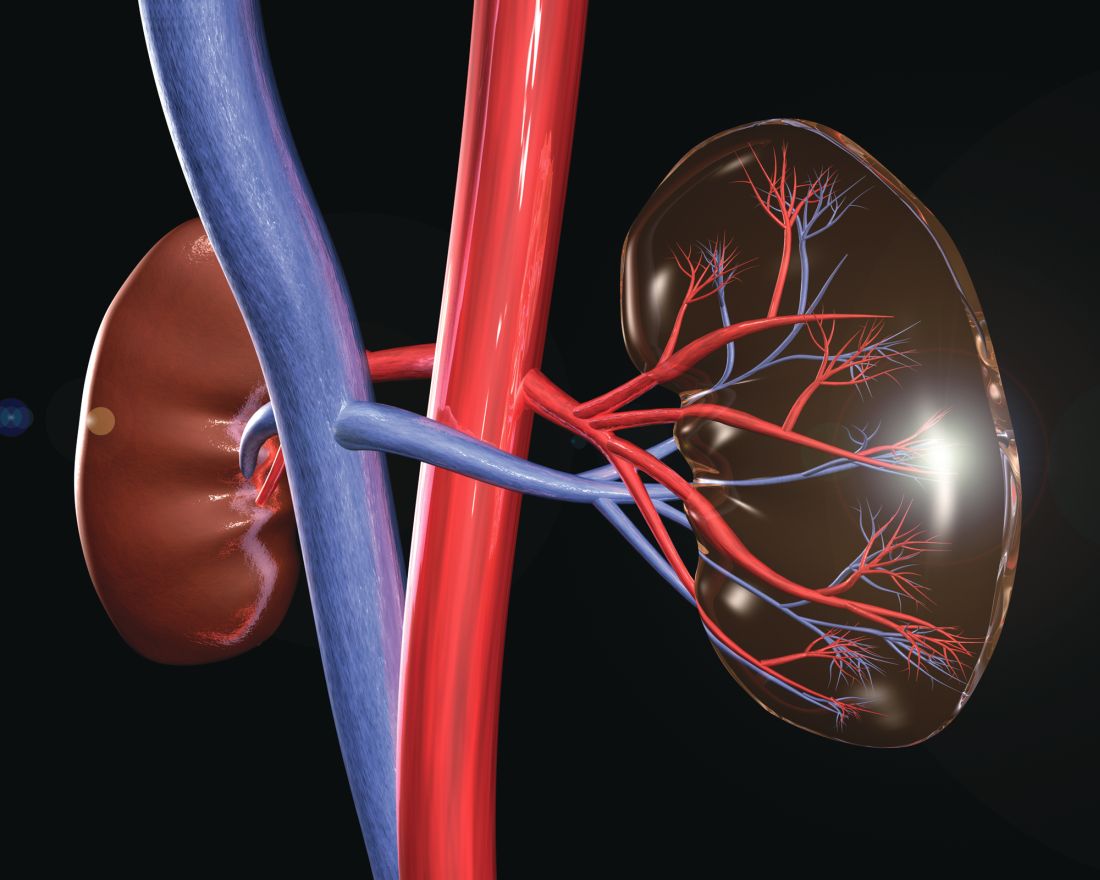
HCV, HBV, and HIV associated with autoimmune kidney diseases
Chronic viral infection can be associated with a variety of autoimmune kidney diseases, according to a review published in Rheumatic Disease Clinics.
In particular, hepatitis C virus (HCV) infection can cause several kidney disorders. These include cryoglobulinemic glomerulonephritis, membranous nephropathy, fibrillary glomerulopathy, immunotactoid glomerulopathy, and IgA nephropathy, wrote Joshua D. Long and his colleagues at Massachusetts General Hospital, Boston.
Similarly, hepatitis B virus (HBV) infection was found to be associated with both membranous nephropathy and polyarteritis nodosa, and human immunodeficiency virus (HIV) infection can cause HIV-associated nephropathy and HIV-associated immune complex diseases, which affect the kidneys.
In their detailed review, the authors discussed the various causal mechanisms and clinical presentations of each of these various autoimmune kidney diseases caused by HCV, HBV, and HIV, along with current treatment modalities.
“Control of the kidney disease relies primarily on treatment of viremia with antiviral agents; however, immunosuppression also may be needed in severe cases,” said the reviewers. However, “more clinical trials are needed to determine first-line therapies for patients who develop autoimmune kidney diseases in the context of chronic viral infections and to define when adjunctive immunosuppressive therapy is warranted,” they concluded.
One of the authors reported grant support and acting as a consultant for various pharmaceutical companies.
mlesney@mdedge.com
SOURCE: Long JD et al. Rheum Dis Clin N Am 2018;44:675-98.
Chronic viral infection can be associated with a variety of autoimmune kidney diseases, according to a review published in Rheumatic Disease Clinics.
In particular, hepatitis C virus (HCV) infection can cause several kidney disorders. These include cryoglobulinemic glomerulonephritis, membranous nephropathy, fibrillary glomerulopathy, immunotactoid glomerulopathy, and IgA nephropathy, wrote Joshua D. Long and his colleagues at Massachusetts General Hospital, Boston.
Similarly, hepatitis B virus (HBV) infection was found to be associated with both membranous nephropathy and polyarteritis nodosa, and human immunodeficiency virus (HIV) infection can cause HIV-associated nephropathy and HIV-associated immune complex diseases, which affect the kidneys.
In their detailed review, the authors discussed the various causal mechanisms and clinical presentations of each of these various autoimmune kidney diseases caused by HCV, HBV, and HIV, along with current treatment modalities.
“Control of the kidney disease relies primarily on treatment of viremia with antiviral agents; however, immunosuppression also may be needed in severe cases,” said the reviewers. However, “more clinical trials are needed to determine first-line therapies for patients who develop autoimmune kidney diseases in the context of chronic viral infections and to define when adjunctive immunosuppressive therapy is warranted,” they concluded.
One of the authors reported grant support and acting as a consultant for various pharmaceutical companies.
mlesney@mdedge.com
SOURCE: Long JD et al. Rheum Dis Clin N Am 2018;44:675-98.
Chronic viral infection can be associated with a variety of autoimmune kidney diseases, according to a review published in Rheumatic Disease Clinics.
In particular, hepatitis C virus (HCV) infection can cause several kidney disorders. These include cryoglobulinemic glomerulonephritis, membranous nephropathy, fibrillary glomerulopathy, immunotactoid glomerulopathy, and IgA nephropathy, wrote Joshua D. Long and his colleagues at Massachusetts General Hospital, Boston.
Similarly, hepatitis B virus (HBV) infection was found to be associated with both membranous nephropathy and polyarteritis nodosa, and human immunodeficiency virus (HIV) infection can cause HIV-associated nephropathy and HIV-associated immune complex diseases, which affect the kidneys.
In their detailed review, the authors discussed the various causal mechanisms and clinical presentations of each of these various autoimmune kidney diseases caused by HCV, HBV, and HIV, along with current treatment modalities.
“Control of the kidney disease relies primarily on treatment of viremia with antiviral agents; however, immunosuppression also may be needed in severe cases,” said the reviewers. However, “more clinical trials are needed to determine first-line therapies for patients who develop autoimmune kidney diseases in the context of chronic viral infections and to define when adjunctive immunosuppressive therapy is warranted,” they concluded.
One of the authors reported grant support and acting as a consultant for various pharmaceutical companies.
mlesney@mdedge.com
SOURCE: Long JD et al. Rheum Dis Clin N Am 2018;44:675-98.
FROM RHEUMATIC DISEASE CLINICS
Hospital medicine and palliative care: Wearing both hats
Dr. Barbara Egan leads SHM’s Palliative Care Work Group
Editor’s note: Each month, the Society of Hospitalist Medicine puts the spotlight on some of our most active members who are making substantial contributions to hospital medicine. Visit www.hospitalmedicine.org for more information on how you can lend your expertise to help improve the care of hospitalized patients.
This month, The Hospitalist spotlights Barbara Egan, MD, FACP, SFHM, chief of the hospital medicine service in the department of medicine at Memorial Sloan Kettering Cancer Center in New York. Barbara has been a member of SHM since 2005, is dual certified in hospital medicine and palliative care, and is the chair of SHM’s Palliative Care Work Group.
When did you first hear about SHM, and why did you decide to become a member?
I first learned about SHM when I was an internal medicine resident at Brigham and Women’s Hospital, Boston, in the early 2000s. BWH had an extremely strong hospitalist group; the staff I worked with served as powerful role models for me and inspired my interest in becoming a hospitalist. One of my attendings suggested that I join SHM, which I did right after I graduated from residency. I attended my first SHM Annual Conference in 2005. By then, I was working as a hospitalist at Memorial Sloan Kettering Cancer Center. SHM and the field of hospital medicine have exploded since my career first began, and I am happy to have grown alongside them. Similarly, our hospital medicine group here at MSKCC has dramatically grown, from 1 hospitalist (me) to more than 30!
How did you get involved with SHM’s Palliative Care Work Group, and what has the work group accomplished since you joined?
I was honored to be invited to join SHM’s Palliative Care Work Group in 2017 by Wendy Anderson, MD, a colleague and now a friend from University of California, San Francisco. Wendy is a visionary leader who practices and researches at the intersection of palliative care and hospital medicine. She and I met during 2015, when we were both invited to join a collaboration between SHM and the Hastings Center in Garrison, N.Y., which was aimed at improving hospitalists’ ability to provide outstanding care to hospitalized patients with life-limiting illnesses. That collaboration resulted in the Improving Communication about Serious Illness–Implementation Guide, a compilation of resources and best practices.
Wendy was chairing the SHM Palliative Care Work Group and invited me to join, which I did with great enthusiasm. This group consists of several passionate and brilliant hospitalists whose practices, in a variety of ways, involve both hospital medicine and palliative medicine. I was so honored when Wendy passed the baton to me last spring and invited me to chair the Work Group. I am lucky to have the opportunity to collaborate with this group of dynamic individuals, and we are well supported by an outstanding SHM staff member, Nick Marzano.
Are there any new projects that the work group is currently focusing on?
The primary focus of SHM’s Palliative Care Work Group is educational. That is, we aim to assess and help meet the educational needs of hospitalists, thereby helping to empower them to be outstanding providers of primary palliative care to seriously ill, hospitalized patients. To that end, we were very proud to orchestrate a palliative care mini-track for the first time at HM18. To our group’s delight, the attendance and reviews of that track were great. Thus, we were invited to further expand the palliative care offerings at HM19. We are busy planning for HM19: a full-day pre-course in palliative medicine; several podium presentations which will touch on ethical challenges, symptom management, prognostication, and other important topics; and a workshop in communication skills.
What led to your dual certification and how do your two specialties overlap?
I am board certified in internal medicine with Focused Practice in Hospital Medicine by virtue of my clinical training and my primary clinical practice as a hospitalist. As a hospitalist in a cancer center, I spend most of my time caring for patients with late- and end-stage malignancy. As such, early in my career, I had to develop a broad base of palliative medical skills, such as pain and symptom management and communication skills. I find this work extremely rewarding, albeit emotionally taxing. I have learned to redefine what clinical “success” looks like – my patients often have unfixable medical problems, but I can always strive to improve their lives in some way, even if that means helping to provide them with a painless, dignified death as opposed to curing them.
When the American Board of Medical Specialties established a board certification in Hospice and Palliative Medicine, there briefly existed a pathway to be “grandfathered” in, i.e., to qualify for board certification through an examination and clinical experience, as opposed to a fellowship. I jumped at the chance to formalize my palliative care skills and experience, and I attained board certification in 2012. This allowed me to further diversify my clinical practice here at MSKCC.
Hospital medicine is still my first love, and I spend most of my time practicing as a hospitalist on our solid tumor services. But now I also spend several weeks each year attending as a consultant on our inpatient supportive care service. In that role, I am able to collaborate with a fantastic multidisciplinary team consisting of MDs, NPs, a chaplain, a pharmacist, a social worker, and integrative medicine practitioners. I also love the opportunity to teach and mentor our palliative medicine fellows.
To me, the opportunity to marry hospital medicine and palliative medicine in my career was a natural fit. Hospitalists, particularly those caring exclusively for cancer patients like I do, need to provide excellent palliative care to our patients every day. The opportunity to further my training and to obtain board certification was a golden one, and I love being able to wear both hats here at MSKCC.
Ms. Steele is a marketing communications specialist at the Society of Hospital Medicine.
Dr. Barbara Egan leads SHM’s Palliative Care Work Group
Dr. Barbara Egan leads SHM’s Palliative Care Work Group
Editor’s note: Each month, the Society of Hospitalist Medicine puts the spotlight on some of our most active members who are making substantial contributions to hospital medicine. Visit www.hospitalmedicine.org for more information on how you can lend your expertise to help improve the care of hospitalized patients.
This month, The Hospitalist spotlights Barbara Egan, MD, FACP, SFHM, chief of the hospital medicine service in the department of medicine at Memorial Sloan Kettering Cancer Center in New York. Barbara has been a member of SHM since 2005, is dual certified in hospital medicine and palliative care, and is the chair of SHM’s Palliative Care Work Group.
When did you first hear about SHM, and why did you decide to become a member?
I first learned about SHM when I was an internal medicine resident at Brigham and Women’s Hospital, Boston, in the early 2000s. BWH had an extremely strong hospitalist group; the staff I worked with served as powerful role models for me and inspired my interest in becoming a hospitalist. One of my attendings suggested that I join SHM, which I did right after I graduated from residency. I attended my first SHM Annual Conference in 2005. By then, I was working as a hospitalist at Memorial Sloan Kettering Cancer Center. SHM and the field of hospital medicine have exploded since my career first began, and I am happy to have grown alongside them. Similarly, our hospital medicine group here at MSKCC has dramatically grown, from 1 hospitalist (me) to more than 30!
How did you get involved with SHM’s Palliative Care Work Group, and what has the work group accomplished since you joined?
I was honored to be invited to join SHM’s Palliative Care Work Group in 2017 by Wendy Anderson, MD, a colleague and now a friend from University of California, San Francisco. Wendy is a visionary leader who practices and researches at the intersection of palliative care and hospital medicine. She and I met during 2015, when we were both invited to join a collaboration between SHM and the Hastings Center in Garrison, N.Y., which was aimed at improving hospitalists’ ability to provide outstanding care to hospitalized patients with life-limiting illnesses. That collaboration resulted in the Improving Communication about Serious Illness–Implementation Guide, a compilation of resources and best practices.
Wendy was chairing the SHM Palliative Care Work Group and invited me to join, which I did with great enthusiasm. This group consists of several passionate and brilliant hospitalists whose practices, in a variety of ways, involve both hospital medicine and palliative medicine. I was so honored when Wendy passed the baton to me last spring and invited me to chair the Work Group. I am lucky to have the opportunity to collaborate with this group of dynamic individuals, and we are well supported by an outstanding SHM staff member, Nick Marzano.
Are there any new projects that the work group is currently focusing on?
The primary focus of SHM’s Palliative Care Work Group is educational. That is, we aim to assess and help meet the educational needs of hospitalists, thereby helping to empower them to be outstanding providers of primary palliative care to seriously ill, hospitalized patients. To that end, we were very proud to orchestrate a palliative care mini-track for the first time at HM18. To our group’s delight, the attendance and reviews of that track were great. Thus, we were invited to further expand the palliative care offerings at HM19. We are busy planning for HM19: a full-day pre-course in palliative medicine; several podium presentations which will touch on ethical challenges, symptom management, prognostication, and other important topics; and a workshop in communication skills.
What led to your dual certification and how do your two specialties overlap?
I am board certified in internal medicine with Focused Practice in Hospital Medicine by virtue of my clinical training and my primary clinical practice as a hospitalist. As a hospitalist in a cancer center, I spend most of my time caring for patients with late- and end-stage malignancy. As such, early in my career, I had to develop a broad base of palliative medical skills, such as pain and symptom management and communication skills. I find this work extremely rewarding, albeit emotionally taxing. I have learned to redefine what clinical “success” looks like – my patients often have unfixable medical problems, but I can always strive to improve their lives in some way, even if that means helping to provide them with a painless, dignified death as opposed to curing them.
When the American Board of Medical Specialties established a board certification in Hospice and Palliative Medicine, there briefly existed a pathway to be “grandfathered” in, i.e., to qualify for board certification through an examination and clinical experience, as opposed to a fellowship. I jumped at the chance to formalize my palliative care skills and experience, and I attained board certification in 2012. This allowed me to further diversify my clinical practice here at MSKCC.
Hospital medicine is still my first love, and I spend most of my time practicing as a hospitalist on our solid tumor services. But now I also spend several weeks each year attending as a consultant on our inpatient supportive care service. In that role, I am able to collaborate with a fantastic multidisciplinary team consisting of MDs, NPs, a chaplain, a pharmacist, a social worker, and integrative medicine practitioners. I also love the opportunity to teach and mentor our palliative medicine fellows.
To me, the opportunity to marry hospital medicine and palliative medicine in my career was a natural fit. Hospitalists, particularly those caring exclusively for cancer patients like I do, need to provide excellent palliative care to our patients every day. The opportunity to further my training and to obtain board certification was a golden one, and I love being able to wear both hats here at MSKCC.
Ms. Steele is a marketing communications specialist at the Society of Hospital Medicine.
Editor’s note: Each month, the Society of Hospitalist Medicine puts the spotlight on some of our most active members who are making substantial contributions to hospital medicine. Visit www.hospitalmedicine.org for more information on how you can lend your expertise to help improve the care of hospitalized patients.
This month, The Hospitalist spotlights Barbara Egan, MD, FACP, SFHM, chief of the hospital medicine service in the department of medicine at Memorial Sloan Kettering Cancer Center in New York. Barbara has been a member of SHM since 2005, is dual certified in hospital medicine and palliative care, and is the chair of SHM’s Palliative Care Work Group.
When did you first hear about SHM, and why did you decide to become a member?
I first learned about SHM when I was an internal medicine resident at Brigham and Women’s Hospital, Boston, in the early 2000s. BWH had an extremely strong hospitalist group; the staff I worked with served as powerful role models for me and inspired my interest in becoming a hospitalist. One of my attendings suggested that I join SHM, which I did right after I graduated from residency. I attended my first SHM Annual Conference in 2005. By then, I was working as a hospitalist at Memorial Sloan Kettering Cancer Center. SHM and the field of hospital medicine have exploded since my career first began, and I am happy to have grown alongside them. Similarly, our hospital medicine group here at MSKCC has dramatically grown, from 1 hospitalist (me) to more than 30!
How did you get involved with SHM’s Palliative Care Work Group, and what has the work group accomplished since you joined?
I was honored to be invited to join SHM’s Palliative Care Work Group in 2017 by Wendy Anderson, MD, a colleague and now a friend from University of California, San Francisco. Wendy is a visionary leader who practices and researches at the intersection of palliative care and hospital medicine. She and I met during 2015, when we were both invited to join a collaboration between SHM and the Hastings Center in Garrison, N.Y., which was aimed at improving hospitalists’ ability to provide outstanding care to hospitalized patients with life-limiting illnesses. That collaboration resulted in the Improving Communication about Serious Illness–Implementation Guide, a compilation of resources and best practices.
Wendy was chairing the SHM Palliative Care Work Group and invited me to join, which I did with great enthusiasm. This group consists of several passionate and brilliant hospitalists whose practices, in a variety of ways, involve both hospital medicine and palliative medicine. I was so honored when Wendy passed the baton to me last spring and invited me to chair the Work Group. I am lucky to have the opportunity to collaborate with this group of dynamic individuals, and we are well supported by an outstanding SHM staff member, Nick Marzano.
Are there any new projects that the work group is currently focusing on?
The primary focus of SHM’s Palliative Care Work Group is educational. That is, we aim to assess and help meet the educational needs of hospitalists, thereby helping to empower them to be outstanding providers of primary palliative care to seriously ill, hospitalized patients. To that end, we were very proud to orchestrate a palliative care mini-track for the first time at HM18. To our group’s delight, the attendance and reviews of that track were great. Thus, we were invited to further expand the palliative care offerings at HM19. We are busy planning for HM19: a full-day pre-course in palliative medicine; several podium presentations which will touch on ethical challenges, symptom management, prognostication, and other important topics; and a workshop in communication skills.
What led to your dual certification and how do your two specialties overlap?
I am board certified in internal medicine with Focused Practice in Hospital Medicine by virtue of my clinical training and my primary clinical practice as a hospitalist. As a hospitalist in a cancer center, I spend most of my time caring for patients with late- and end-stage malignancy. As such, early in my career, I had to develop a broad base of palliative medical skills, such as pain and symptom management and communication skills. I find this work extremely rewarding, albeit emotionally taxing. I have learned to redefine what clinical “success” looks like – my patients often have unfixable medical problems, but I can always strive to improve their lives in some way, even if that means helping to provide them with a painless, dignified death as opposed to curing them.
When the American Board of Medical Specialties established a board certification in Hospice and Palliative Medicine, there briefly existed a pathway to be “grandfathered” in, i.e., to qualify for board certification through an examination and clinical experience, as opposed to a fellowship. I jumped at the chance to formalize my palliative care skills and experience, and I attained board certification in 2012. This allowed me to further diversify my clinical practice here at MSKCC.
Hospital medicine is still my first love, and I spend most of my time practicing as a hospitalist on our solid tumor services. But now I also spend several weeks each year attending as a consultant on our inpatient supportive care service. In that role, I am able to collaborate with a fantastic multidisciplinary team consisting of MDs, NPs, a chaplain, a pharmacist, a social worker, and integrative medicine practitioners. I also love the opportunity to teach and mentor our palliative medicine fellows.
To me, the opportunity to marry hospital medicine and palliative medicine in my career was a natural fit. Hospitalists, particularly those caring exclusively for cancer patients like I do, need to provide excellent palliative care to our patients every day. The opportunity to further my training and to obtain board certification was a golden one, and I love being able to wear both hats here at MSKCC.
Ms. Steele is a marketing communications specialist at the Society of Hospital Medicine.
TKIs and immunotherapy hold promise for alveolar soft part sarcoma
Alveolar soft part sarcoma (ASPS) has often proven to be resistant to conventional doxorubicin-based chemotherapy, but tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICIs) may provide new treatment strategies for this rare type of sarcoma, according to a literature review.
A rare, translocation-driven sarcoma of the soft tissues, ASPS often affects young adults and is characterized by indolent behavior and early metastasis. Despite its resistance to chemotherapy, studies indicate that survival is often prolonged in patients with metastatic disease. The literature has shown 5-year survival rates at about 60%, and this percentage has remained fairly consistent for the past 3 decades.
Luca Paoluzzi, MD, of New York University, and Robert G. Maki, MD, PhD, of Hofstra University, Hempstead, N.Y., reviewed the literature from 1952 to March 2018, in order to gain a better understanding of ASPS and the opportunities “for the translation of such knowledge into clinical practice,” they wrote in JAMA.
From a therapeutic standpoint, ASPS is characterized by sensitivity to vascular endothelial growth factor receptor–predominant TKIs, compared with other soft tissue sarcomas (STS), and recent data have emphasized that it is responsive to new immunotherapy regimens including ICIs. Pazopanib is currently the only agent that has received regulatory approval for use in STS refractory to other treatments and it appears to have consistent activity in metastatic ASPS. Management of ASPS generally also involves surgical resection and/or systemic treatment for metastatic disease. Conventional agents such as anthracycline-based chemotherapy have demonstrated a poor response rate lower than 10%, and while a complete resection may be curative, metastases are common and can occur years after resection of the primary tumor.
Conversely, ICIs “represent a promising area of drug development in ASPS; the data to date are limited but encouraging,” wrote Dr. Paoluzzi and Dr. Maki.
They pointed to one study that included 50 patients with sarcoma with 14 different subtypes of STS who were enrolled in immunotherapy trials conducted at the University of Texas MD Anderson Cancer Center, Houston. There were two pretreated patients with ASPS (two to four prior lines) in the cohort who received antiprogrammed death-ligand 1–based therapy, and achieved a partial response bordering on a complete response that lasted 8 and 12 months. An additional two patients achieved stable disease.
Another paper, presented at the 2017 Connective Tissue Oncology Society annual meeting, presented preliminary data from a phase 2 study that showed four of nine evaluable patients with ASPS treated with the TKI axitinib, combined with pembrolizumab, achieved a partial response. Three others had stable disease.
“Pathway-driven basket trials facilitate the enrollment of patients with such uncommon cancers and should provide valuable information regarding a second type of immune responsiveness to ICIs, one that is not a function of high tumor mutational burden,” the authors concluded.
No outside funding sources were reported. Dr. Maki reported receiving consultant fees from numerous sources and research support to New York University from Immune Design, Immunocore, Eli Lilly, Presage Biosciences, TRACON Pharmaceuticals, SARC, Regeneron, and Genentech. No other conflicts were reported.
SOURCE: doi: 10.1001/jamaoncol.2018.4490.
Alveolar soft part sarcoma (ASPS) has often proven to be resistant to conventional doxorubicin-based chemotherapy, but tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICIs) may provide new treatment strategies for this rare type of sarcoma, according to a literature review.
A rare, translocation-driven sarcoma of the soft tissues, ASPS often affects young adults and is characterized by indolent behavior and early metastasis. Despite its resistance to chemotherapy, studies indicate that survival is often prolonged in patients with metastatic disease. The literature has shown 5-year survival rates at about 60%, and this percentage has remained fairly consistent for the past 3 decades.
Luca Paoluzzi, MD, of New York University, and Robert G. Maki, MD, PhD, of Hofstra University, Hempstead, N.Y., reviewed the literature from 1952 to March 2018, in order to gain a better understanding of ASPS and the opportunities “for the translation of such knowledge into clinical practice,” they wrote in JAMA.
From a therapeutic standpoint, ASPS is characterized by sensitivity to vascular endothelial growth factor receptor–predominant TKIs, compared with other soft tissue sarcomas (STS), and recent data have emphasized that it is responsive to new immunotherapy regimens including ICIs. Pazopanib is currently the only agent that has received regulatory approval for use in STS refractory to other treatments and it appears to have consistent activity in metastatic ASPS. Management of ASPS generally also involves surgical resection and/or systemic treatment for metastatic disease. Conventional agents such as anthracycline-based chemotherapy have demonstrated a poor response rate lower than 10%, and while a complete resection may be curative, metastases are common and can occur years after resection of the primary tumor.
Conversely, ICIs “represent a promising area of drug development in ASPS; the data to date are limited but encouraging,” wrote Dr. Paoluzzi and Dr. Maki.
They pointed to one study that included 50 patients with sarcoma with 14 different subtypes of STS who were enrolled in immunotherapy trials conducted at the University of Texas MD Anderson Cancer Center, Houston. There were two pretreated patients with ASPS (two to four prior lines) in the cohort who received antiprogrammed death-ligand 1–based therapy, and achieved a partial response bordering on a complete response that lasted 8 and 12 months. An additional two patients achieved stable disease.
Another paper, presented at the 2017 Connective Tissue Oncology Society annual meeting, presented preliminary data from a phase 2 study that showed four of nine evaluable patients with ASPS treated with the TKI axitinib, combined with pembrolizumab, achieved a partial response. Three others had stable disease.
“Pathway-driven basket trials facilitate the enrollment of patients with such uncommon cancers and should provide valuable information regarding a second type of immune responsiveness to ICIs, one that is not a function of high tumor mutational burden,” the authors concluded.
No outside funding sources were reported. Dr. Maki reported receiving consultant fees from numerous sources and research support to New York University from Immune Design, Immunocore, Eli Lilly, Presage Biosciences, TRACON Pharmaceuticals, SARC, Regeneron, and Genentech. No other conflicts were reported.
SOURCE: doi: 10.1001/jamaoncol.2018.4490.
Alveolar soft part sarcoma (ASPS) has often proven to be resistant to conventional doxorubicin-based chemotherapy, but tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICIs) may provide new treatment strategies for this rare type of sarcoma, according to a literature review.
A rare, translocation-driven sarcoma of the soft tissues, ASPS often affects young adults and is characterized by indolent behavior and early metastasis. Despite its resistance to chemotherapy, studies indicate that survival is often prolonged in patients with metastatic disease. The literature has shown 5-year survival rates at about 60%, and this percentage has remained fairly consistent for the past 3 decades.
Luca Paoluzzi, MD, of New York University, and Robert G. Maki, MD, PhD, of Hofstra University, Hempstead, N.Y., reviewed the literature from 1952 to March 2018, in order to gain a better understanding of ASPS and the opportunities “for the translation of such knowledge into clinical practice,” they wrote in JAMA.
From a therapeutic standpoint, ASPS is characterized by sensitivity to vascular endothelial growth factor receptor–predominant TKIs, compared with other soft tissue sarcomas (STS), and recent data have emphasized that it is responsive to new immunotherapy regimens including ICIs. Pazopanib is currently the only agent that has received regulatory approval for use in STS refractory to other treatments and it appears to have consistent activity in metastatic ASPS. Management of ASPS generally also involves surgical resection and/or systemic treatment for metastatic disease. Conventional agents such as anthracycline-based chemotherapy have demonstrated a poor response rate lower than 10%, and while a complete resection may be curative, metastases are common and can occur years after resection of the primary tumor.
Conversely, ICIs “represent a promising area of drug development in ASPS; the data to date are limited but encouraging,” wrote Dr. Paoluzzi and Dr. Maki.
They pointed to one study that included 50 patients with sarcoma with 14 different subtypes of STS who were enrolled in immunotherapy trials conducted at the University of Texas MD Anderson Cancer Center, Houston. There were two pretreated patients with ASPS (two to four prior lines) in the cohort who received antiprogrammed death-ligand 1–based therapy, and achieved a partial response bordering on a complete response that lasted 8 and 12 months. An additional two patients achieved stable disease.
Another paper, presented at the 2017 Connective Tissue Oncology Society annual meeting, presented preliminary data from a phase 2 study that showed four of nine evaluable patients with ASPS treated with the TKI axitinib, combined with pembrolizumab, achieved a partial response. Three others had stable disease.
“Pathway-driven basket trials facilitate the enrollment of patients with such uncommon cancers and should provide valuable information regarding a second type of immune responsiveness to ICIs, one that is not a function of high tumor mutational burden,” the authors concluded.
No outside funding sources were reported. Dr. Maki reported receiving consultant fees from numerous sources and research support to New York University from Immune Design, Immunocore, Eli Lilly, Presage Biosciences, TRACON Pharmaceuticals, SARC, Regeneron, and Genentech. No other conflicts were reported.
SOURCE: doi: 10.1001/jamaoncol.2018.4490.
FROM JAMA
Key clinical point: Alveolar soft part sarcoma has often proven to be resistant to conventional doxorubicin-based chemotherapy, tyrosine kinase inhibitors and immune checkpoint inhibitors may provide new treatment strategies.
Major finding: In one study of sarcoma patients enrolled in immunotherapy trials, two pretreated patients with alveolar soft part sarcoma (two to four prior lines) who received antiprogrammed death-ligand 1–based therapy achieved partial responses, bordering on a complete response, that lasted 8 and 12 months.
Study details: A review of literature concerning treatment for alveolar soft part sarcoma.
Disclosures: No outside funding sources were reported. Dr. Maki reported receiving consultant fees from numerous sources and research support to New York University from Immune Design, Immunocore, Eli Lilly, Presage Biosciences, TRACON Pharmaceuticals, SARC, Regeneron, and Genentech. No other conflicts were reported.
Source:
Age, insurance type tied to delay in pediatric febrile UTI treatment
even after adjustment for confounders, according to a report in the Journal of Pediatrics.
Stephanie W. Hum and Nader Shaikh, MD, MPH, of the University of Pittsburgh, drew from data provided by two studies, Randomized Intervention for Children With Vesicoureteral Reflux and Careful Urinary Tract Infection Evaluation. Specifically, they extracted data regarding patients’ age, sex, history of UTIs, ethnicity, race, insurance, household size, and duration of fever before initiation of antimicrobial therapy, as well as primary caregivers’ level of education and income. Some factors were analyzed because of associations seen in adult studies, and others because of concerns about access to care. In this analysis, the researchers defined “treatment delay” as the number of hours between onset of fever and initiation of antimicrobial treatment and, after exclusion of afebrile children and those with missing data, included 660 patients.
In univariate analysis, both older age and commercial insurance were found to be significantly associated with delays in treatment. Compared with time to treatment seen with younger children, treatment was delayed by an average of 26.2 hours in children aged 12 months and older (P less than .001). Patients with commercial insurance were treated a mean of 12.6 hours later than were those with noncommercial insurance (P = .002). These associations remained significant even after adjustment in a multivariable regression model for sex, history of UTIs, ethnicity, race, primary caregivers’ level of education, insurance, and income level.
The finding regarding age is consistent with a previous study, and Ms. Hum and Dr. Shaikh suggested it may reflect parents experiencing reduced urgency regarding febrile illnesses among older children. However, the researchers also noted that greater rates of renal scarring are seen in older children, so “it seems important to educate physicians, parents, and triage nurses about the importance of early evaluation of children with fever,” even those older than 12 months.
The finding regarding insurance status, however, is contrary to what studies in adult populations have found, as well as those in pediatric EDs. The researchers suggested that perhaps parents with noncommercial insurance are more likely to take their children to EDs, where testing can be done on-site 24 hours a day, rather than to private clinics, which often have to send out testing to off-site laboratories.
One of the strengths of the study is its relatively large sample size, they said. Among its weaknesses is that treatment delays were self-reported by parents and might be inaccurate and that information regarding location of initial evaluation was not gathered and could not be examined with other factors.
This study was supported by a T35 training grant from the National Institute of Diabetes and Digestive and Kidney Diseases, sponsored by Tom R. Kleyman, MD, chief of the renal-electrolyte division at the University of Pittsburgh. The authors declared no conflicts of interest.
SOURCE: Hum SW et al. J Pediatr. 2018 Oct 16. doi: 10.1016/j.jpeds.2018.09.029.
This article was updated 10/24/18.
even after adjustment for confounders, according to a report in the Journal of Pediatrics.
Stephanie W. Hum and Nader Shaikh, MD, MPH, of the University of Pittsburgh, drew from data provided by two studies, Randomized Intervention for Children With Vesicoureteral Reflux and Careful Urinary Tract Infection Evaluation. Specifically, they extracted data regarding patients’ age, sex, history of UTIs, ethnicity, race, insurance, household size, and duration of fever before initiation of antimicrobial therapy, as well as primary caregivers’ level of education and income. Some factors were analyzed because of associations seen in adult studies, and others because of concerns about access to care. In this analysis, the researchers defined “treatment delay” as the number of hours between onset of fever and initiation of antimicrobial treatment and, after exclusion of afebrile children and those with missing data, included 660 patients.
In univariate analysis, both older age and commercial insurance were found to be significantly associated with delays in treatment. Compared with time to treatment seen with younger children, treatment was delayed by an average of 26.2 hours in children aged 12 months and older (P less than .001). Patients with commercial insurance were treated a mean of 12.6 hours later than were those with noncommercial insurance (P = .002). These associations remained significant even after adjustment in a multivariable regression model for sex, history of UTIs, ethnicity, race, primary caregivers’ level of education, insurance, and income level.
The finding regarding age is consistent with a previous study, and Ms. Hum and Dr. Shaikh suggested it may reflect parents experiencing reduced urgency regarding febrile illnesses among older children. However, the researchers also noted that greater rates of renal scarring are seen in older children, so “it seems important to educate physicians, parents, and triage nurses about the importance of early evaluation of children with fever,” even those older than 12 months.
The finding regarding insurance status, however, is contrary to what studies in adult populations have found, as well as those in pediatric EDs. The researchers suggested that perhaps parents with noncommercial insurance are more likely to take their children to EDs, where testing can be done on-site 24 hours a day, rather than to private clinics, which often have to send out testing to off-site laboratories.
One of the strengths of the study is its relatively large sample size, they said. Among its weaknesses is that treatment delays were self-reported by parents and might be inaccurate and that information regarding location of initial evaluation was not gathered and could not be examined with other factors.
This study was supported by a T35 training grant from the National Institute of Diabetes and Digestive and Kidney Diseases, sponsored by Tom R. Kleyman, MD, chief of the renal-electrolyte division at the University of Pittsburgh. The authors declared no conflicts of interest.
SOURCE: Hum SW et al. J Pediatr. 2018 Oct 16. doi: 10.1016/j.jpeds.2018.09.029.
This article was updated 10/24/18.
even after adjustment for confounders, according to a report in the Journal of Pediatrics.
Stephanie W. Hum and Nader Shaikh, MD, MPH, of the University of Pittsburgh, drew from data provided by two studies, Randomized Intervention for Children With Vesicoureteral Reflux and Careful Urinary Tract Infection Evaluation. Specifically, they extracted data regarding patients’ age, sex, history of UTIs, ethnicity, race, insurance, household size, and duration of fever before initiation of antimicrobial therapy, as well as primary caregivers’ level of education and income. Some factors were analyzed because of associations seen in adult studies, and others because of concerns about access to care. In this analysis, the researchers defined “treatment delay” as the number of hours between onset of fever and initiation of antimicrobial treatment and, after exclusion of afebrile children and those with missing data, included 660 patients.
In univariate analysis, both older age and commercial insurance were found to be significantly associated with delays in treatment. Compared with time to treatment seen with younger children, treatment was delayed by an average of 26.2 hours in children aged 12 months and older (P less than .001). Patients with commercial insurance were treated a mean of 12.6 hours later than were those with noncommercial insurance (P = .002). These associations remained significant even after adjustment in a multivariable regression model for sex, history of UTIs, ethnicity, race, primary caregivers’ level of education, insurance, and income level.
The finding regarding age is consistent with a previous study, and Ms. Hum and Dr. Shaikh suggested it may reflect parents experiencing reduced urgency regarding febrile illnesses among older children. However, the researchers also noted that greater rates of renal scarring are seen in older children, so “it seems important to educate physicians, parents, and triage nurses about the importance of early evaluation of children with fever,” even those older than 12 months.
The finding regarding insurance status, however, is contrary to what studies in adult populations have found, as well as those in pediatric EDs. The researchers suggested that perhaps parents with noncommercial insurance are more likely to take their children to EDs, where testing can be done on-site 24 hours a day, rather than to private clinics, which often have to send out testing to off-site laboratories.
One of the strengths of the study is its relatively large sample size, they said. Among its weaknesses is that treatment delays were self-reported by parents and might be inaccurate and that information regarding location of initial evaluation was not gathered and could not be examined with other factors.
This study was supported by a T35 training grant from the National Institute of Diabetes and Digestive and Kidney Diseases, sponsored by Tom R. Kleyman, MD, chief of the renal-electrolyte division at the University of Pittsburgh. The authors declared no conflicts of interest.
SOURCE: Hum SW et al. J Pediatr. 2018 Oct 16. doi: 10.1016/j.jpeds.2018.09.029.
This article was updated 10/24/18.
FROM THE JOURNAL OF PEDIATRICS
USPSTF advocates screening reproductive age women for intimate partner violence
Intimate partner violence (IPV), defined to include sexual violence, physical violence, and stalking, occurs in approximately 36% of women and 33% of men in the United States, the Task Force members said.
The screening of women of reproductive age is a B recommendation. However, evidence remains insufficient to recommend routine screening for IPV for men, and screening for abuse in elders and vulnerable adult populations received an I statement based on insufficient evidence to assess the balance of risks and benefits, wrote Susan J. Curry, PhD, of the University of Iowa, Iowa City, and her colleagues. Vulnerable adults were defined as those who are not able to protect themselves because of age, disability, or both. The recommendations were published in JAMA.
The Task Force reviewed the available evidence and determined that screening tools can help identify intimate partner violence in women of reproductive age, and that support services can reduce the risk of physical, sexual, and psychological abuse in these women.
In the evidence review accompanying the recommendations, Cynthia Feltner, MD, MPH, of the University of North Carolina, Chapel Hill, and her colleagues analyzed data from 30 studies including 14,959 individuals.
They reviewed studies of IPV for adolescents through women in their 40s and identified several screening tools that accurately detected IPV in adult women within the past year: Humiliation, Afraid, Rape, Kick (HARK); Hurt, Insulted, Threaten, Scream (HITS); Extended–Hurt, Insult, Threaten, Scream (E-HITS); Partner Violence Screen (PVS); and Woman Abuse Screening Tool (WAST). The sensitivity of these tools ranged from 65% to 87%, and specificity ranged from 80% to 95%.
Data from three randomized, controlled trials including 3,759 women found no benefit to screening them for IPV over 3-18 months, but no randomized, controlled trials found any harms associated with screening.
In addition, the data showed no evidence that screening reduced IPV or improved quality of life over a 3-18-month period, the researchers noted.
In addition, the Task Force researchers found no reliable screening tools to identify IPV in men or to identify abuse of elders or vulnerable adults in the absence of recognized signs and symptoms of abuse.
The complete recommendations are available online in JAMA, and on the USPSTF website: http://www.uspreventiveservicestaskforce.org.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers had no financial conflicts to disclose.
SOURCES: Curry SJ et al. JAMA. 2018;320(16):1678-87; Feltner C et al. JAMA. 2018;320(16):1688-701.
The USPSTF recommendations to screen women of reproductive age for intimate partner violence (IPV) should be supported and implemented, but that should be just a starting point, Karin V. Rhodes, MD, Melissa E. Dichter, PhD, and Kristofer L. Smith, MD, said in an accompanying editorial.
“Given the long-term and wide-ranging harms of abuse, and increased health care utilization and costs of care related to abuse, health systems should begin to expand screening and to test interventions for abuse beyond the recommendations to include older women, men, and elderly and vulnerable populations,” they said, and called on federal, state, and commercial insurance companies to pay for IPV screening for these groups as well as for women of reproductive age.
The recommendations show a notable lack of advancement since those issued in 2013, the editorialists noted. The evidence remains inconclusive to support expanded screening to elders and vulnerable adults. However, evidence-based research is challenging in these groups because randomized, controlled trials involving abuse patients are rare given the ethical considerations of offering care to all abuse victims.
They offered some ideas for improving and expanding screening and enabling evidence-based research, including the use of patient portals and patient-administered screening, as well as making use of the medical home model and coordinating with community-based services.
“It is critical, however, that screening for abuse be conducted with careful attention to confidentiality, safety, and respect for individual self-determination in documentation and follow-up intervention,” the editorialists wrote.
“Such care in screening processes may facilitate disclosure; and, even if patients do not disclose, screening may still have an educational and therapeutic benefit,” they concluded.
Dr. Rhodes and Dr. Smith are affiliated with Hofstra/Northwell, Manhasset, N.Y. Dr. Dichter is affiliated with the University of Pennsylvania, Philadelphia. They had no financial conflicts to disclose. Their editorial accompanying the report by Curry SJ et al. appeared in JAMA 2018 Oct 23/30;320(16):1645-7.
The USPSTF recommendations to screen women of reproductive age for intimate partner violence (IPV) should be supported and implemented, but that should be just a starting point, Karin V. Rhodes, MD, Melissa E. Dichter, PhD, and Kristofer L. Smith, MD, said in an accompanying editorial.
“Given the long-term and wide-ranging harms of abuse, and increased health care utilization and costs of care related to abuse, health systems should begin to expand screening and to test interventions for abuse beyond the recommendations to include older women, men, and elderly and vulnerable populations,” they said, and called on federal, state, and commercial insurance companies to pay for IPV screening for these groups as well as for women of reproductive age.
The recommendations show a notable lack of advancement since those issued in 2013, the editorialists noted. The evidence remains inconclusive to support expanded screening to elders and vulnerable adults. However, evidence-based research is challenging in these groups because randomized, controlled trials involving abuse patients are rare given the ethical considerations of offering care to all abuse victims.
They offered some ideas for improving and expanding screening and enabling evidence-based research, including the use of patient portals and patient-administered screening, as well as making use of the medical home model and coordinating with community-based services.
“It is critical, however, that screening for abuse be conducted with careful attention to confidentiality, safety, and respect for individual self-determination in documentation and follow-up intervention,” the editorialists wrote.
“Such care in screening processes may facilitate disclosure; and, even if patients do not disclose, screening may still have an educational and therapeutic benefit,” they concluded.
Dr. Rhodes and Dr. Smith are affiliated with Hofstra/Northwell, Manhasset, N.Y. Dr. Dichter is affiliated with the University of Pennsylvania, Philadelphia. They had no financial conflicts to disclose. Their editorial accompanying the report by Curry SJ et al. appeared in JAMA 2018 Oct 23/30;320(16):1645-7.
The USPSTF recommendations to screen women of reproductive age for intimate partner violence (IPV) should be supported and implemented, but that should be just a starting point, Karin V. Rhodes, MD, Melissa E. Dichter, PhD, and Kristofer L. Smith, MD, said in an accompanying editorial.
“Given the long-term and wide-ranging harms of abuse, and increased health care utilization and costs of care related to abuse, health systems should begin to expand screening and to test interventions for abuse beyond the recommendations to include older women, men, and elderly and vulnerable populations,” they said, and called on federal, state, and commercial insurance companies to pay for IPV screening for these groups as well as for women of reproductive age.
The recommendations show a notable lack of advancement since those issued in 2013, the editorialists noted. The evidence remains inconclusive to support expanded screening to elders and vulnerable adults. However, evidence-based research is challenging in these groups because randomized, controlled trials involving abuse patients are rare given the ethical considerations of offering care to all abuse victims.
They offered some ideas for improving and expanding screening and enabling evidence-based research, including the use of patient portals and patient-administered screening, as well as making use of the medical home model and coordinating with community-based services.
“It is critical, however, that screening for abuse be conducted with careful attention to confidentiality, safety, and respect for individual self-determination in documentation and follow-up intervention,” the editorialists wrote.
“Such care in screening processes may facilitate disclosure; and, even if patients do not disclose, screening may still have an educational and therapeutic benefit,” they concluded.
Dr. Rhodes and Dr. Smith are affiliated with Hofstra/Northwell, Manhasset, N.Y. Dr. Dichter is affiliated with the University of Pennsylvania, Philadelphia. They had no financial conflicts to disclose. Their editorial accompanying the report by Curry SJ et al. appeared in JAMA 2018 Oct 23/30;320(16):1645-7.
Intimate partner violence (IPV), defined to include sexual violence, physical violence, and stalking, occurs in approximately 36% of women and 33% of men in the United States, the Task Force members said.
The screening of women of reproductive age is a B recommendation. However, evidence remains insufficient to recommend routine screening for IPV for men, and screening for abuse in elders and vulnerable adult populations received an I statement based on insufficient evidence to assess the balance of risks and benefits, wrote Susan J. Curry, PhD, of the University of Iowa, Iowa City, and her colleagues. Vulnerable adults were defined as those who are not able to protect themselves because of age, disability, or both. The recommendations were published in JAMA.
The Task Force reviewed the available evidence and determined that screening tools can help identify intimate partner violence in women of reproductive age, and that support services can reduce the risk of physical, sexual, and psychological abuse in these women.
In the evidence review accompanying the recommendations, Cynthia Feltner, MD, MPH, of the University of North Carolina, Chapel Hill, and her colleagues analyzed data from 30 studies including 14,959 individuals.
They reviewed studies of IPV for adolescents through women in their 40s and identified several screening tools that accurately detected IPV in adult women within the past year: Humiliation, Afraid, Rape, Kick (HARK); Hurt, Insulted, Threaten, Scream (HITS); Extended–Hurt, Insult, Threaten, Scream (E-HITS); Partner Violence Screen (PVS); and Woman Abuse Screening Tool (WAST). The sensitivity of these tools ranged from 65% to 87%, and specificity ranged from 80% to 95%.
Data from three randomized, controlled trials including 3,759 women found no benefit to screening them for IPV over 3-18 months, but no randomized, controlled trials found any harms associated with screening.
In addition, the data showed no evidence that screening reduced IPV or improved quality of life over a 3-18-month period, the researchers noted.
In addition, the Task Force researchers found no reliable screening tools to identify IPV in men or to identify abuse of elders or vulnerable adults in the absence of recognized signs and symptoms of abuse.
The complete recommendations are available online in JAMA, and on the USPSTF website: http://www.uspreventiveservicestaskforce.org.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers had no financial conflicts to disclose.
SOURCES: Curry SJ et al. JAMA. 2018;320(16):1678-87; Feltner C et al. JAMA. 2018;320(16):1688-701.
Intimate partner violence (IPV), defined to include sexual violence, physical violence, and stalking, occurs in approximately 36% of women and 33% of men in the United States, the Task Force members said.
The screening of women of reproductive age is a B recommendation. However, evidence remains insufficient to recommend routine screening for IPV for men, and screening for abuse in elders and vulnerable adult populations received an I statement based on insufficient evidence to assess the balance of risks and benefits, wrote Susan J. Curry, PhD, of the University of Iowa, Iowa City, and her colleagues. Vulnerable adults were defined as those who are not able to protect themselves because of age, disability, or both. The recommendations were published in JAMA.
The Task Force reviewed the available evidence and determined that screening tools can help identify intimate partner violence in women of reproductive age, and that support services can reduce the risk of physical, sexual, and psychological abuse in these women.
In the evidence review accompanying the recommendations, Cynthia Feltner, MD, MPH, of the University of North Carolina, Chapel Hill, and her colleagues analyzed data from 30 studies including 14,959 individuals.
They reviewed studies of IPV for adolescents through women in their 40s and identified several screening tools that accurately detected IPV in adult women within the past year: Humiliation, Afraid, Rape, Kick (HARK); Hurt, Insulted, Threaten, Scream (HITS); Extended–Hurt, Insult, Threaten, Scream (E-HITS); Partner Violence Screen (PVS); and Woman Abuse Screening Tool (WAST). The sensitivity of these tools ranged from 65% to 87%, and specificity ranged from 80% to 95%.
Data from three randomized, controlled trials including 3,759 women found no benefit to screening them for IPV over 3-18 months, but no randomized, controlled trials found any harms associated with screening.
In addition, the data showed no evidence that screening reduced IPV or improved quality of life over a 3-18-month period, the researchers noted.
In addition, the Task Force researchers found no reliable screening tools to identify IPV in men or to identify abuse of elders or vulnerable adults in the absence of recognized signs and symptoms of abuse.
The complete recommendations are available online in JAMA, and on the USPSTF website: http://www.uspreventiveservicestaskforce.org.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The researchers had no financial conflicts to disclose.
SOURCES: Curry SJ et al. JAMA. 2018;320(16):1678-87; Feltner C et al. JAMA. 2018;320(16):1688-701.
FROM JAMA
Checkpoint inhibitor seems safe and effective for patients with HIV
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
REPORTING FROM ESMO 2018
Key clinical point: Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent.
Major finding: No immune-related adverse events or deaths due to drug toxicity occurred.
Study details: A retrospective analysis of 20 patients with HIV who received nivolumab immunotherapy.
Disclosures: Jean-Philippe Spano, principal investigator, disclosed relationships with Gilead, Roche, BMS, and others.
Source: Gobert et al. ESMO 2018, Abstract 1213P_PR.