User login
The Official Newspaper of the American Association for Thoracic Surgery
Maintenance of certification took center stage at AMA Congress of Delegates
CHICAGO – The American Medical Association should continue to work with the American Board of Medical Specialties to address physicians’ concerns about Maintenance of Certification – that was the consensus at the annual meeting of the AMA House of Delegates.
The AMA’s delegates defeated a resolution that asked the organization to put a moratorium on MOC until it was proven to improve the quality of care and patient outcomes. However, they did agree to a new policy that directs the AMA to:
• Explore with independent entities the feasibility of conducting a study to evaluate the effect MOC requirements and Maintenance of Licensure principles have on workforce, practice costs, patient outcomes, patient safety, and patient access.
• Work with the American Board of Medical Specialties and its 24 member boards to collect data on why physicians choose to maintain or discontinue their board certification.
• Work with the ABMS and the Federation of State Medical Boards to study whether MOC and the principles of Maintenance of Licensure are important factors to physicians when deciding whether to retire and whether they have a direct effect on workforce.
• Oppose making MOC mandatory as a condition of medical licensure, and encourage physicians to strive constantly to improve their care of patients by the means they find most effective.
The new policy applies to both the ABMS MOC process and the Osteopathic Continuous Certification (OCC) process.
Physicians have increasingly voiced their concerns about MOC. Dr. Paul Teirstein, chief of cardiology and director of interventional cardiology for Scripps Clinic in La Jolla, Calif., launched a petition drive to overhaul the American Board of Internal Medicine’s MOC process. The petition has more than 17,000 signatures.
The ABIM says that it is listening to physicians and is making changes in the process, but also recently said that more than 150,000 physicians had participated in its MOC process – making the May 1 deadline to be listed on the ABIM website as having met the MOC criteria.
But anger is still bubbling up, and was expressed at the AMA’s meeting.
"Practicing physicians on the front lines are increasingly burdened, hassled, and confused by the onerous and expensive process of Maintenance of Certification and Maintenance of Licensure," said Dr. James A. Goodyear, a delegate from Pennsylvania.
Dr. Goodyear introduced the resolution to seek a moratorium on the MOC.
But Dr. Darlyne Menscer, a member of the AMA Council on Medical Education, told the delegates that such a moratorium would put a wedge in the close working relationship the AMA has had with the ABMS. "This is more prescriptive than we can commit to as a council, although we definitely do hear the concerns of the House," added Dr. Menscer.
The AMA has been discussing the concerns about MOC with the ABMS, most recently holding a meeting in Chicago in early June.
Dr. Joshua Cohen, a delegate from the American Academy of Neurology, and a member of the AMA Foundation’s Board of Directors, who attended that meeting, also argued against a moratorium. "It would make it impossible for the AMA to improve the process going forward," said Dr. Cohen.
Dr. Chuck Wilson, a pediatrician and delegate from the North Carolina delegation, also opposed any major change in direction for the AMA. He noted that if the AMA was seen as opposed to MOC, it might not be viewed well. "We all want it to be less onerous," said Dr. Wilson. But, he noted, "the Council on Medical Education is working in that direction. Let’s give them a chance to be successful."
In a statement after the HOD meeting, the AMA said that it "continues to ensure the MOC process does not disrupt physician practice or reduce the capacity of the overall physician workforce." Concerns about MOC "center around the need for relevance to the daily practice of physicians and the better integration into physician practices to optimally support learning and improvement."
On Twitter @aliciaault
CHICAGO – The American Medical Association should continue to work with the American Board of Medical Specialties to address physicians’ concerns about Maintenance of Certification – that was the consensus at the annual meeting of the AMA House of Delegates.
The AMA’s delegates defeated a resolution that asked the organization to put a moratorium on MOC until it was proven to improve the quality of care and patient outcomes. However, they did agree to a new policy that directs the AMA to:
• Explore with independent entities the feasibility of conducting a study to evaluate the effect MOC requirements and Maintenance of Licensure principles have on workforce, practice costs, patient outcomes, patient safety, and patient access.
• Work with the American Board of Medical Specialties and its 24 member boards to collect data on why physicians choose to maintain or discontinue their board certification.
• Work with the ABMS and the Federation of State Medical Boards to study whether MOC and the principles of Maintenance of Licensure are important factors to physicians when deciding whether to retire and whether they have a direct effect on workforce.
• Oppose making MOC mandatory as a condition of medical licensure, and encourage physicians to strive constantly to improve their care of patients by the means they find most effective.
The new policy applies to both the ABMS MOC process and the Osteopathic Continuous Certification (OCC) process.
Physicians have increasingly voiced their concerns about MOC. Dr. Paul Teirstein, chief of cardiology and director of interventional cardiology for Scripps Clinic in La Jolla, Calif., launched a petition drive to overhaul the American Board of Internal Medicine’s MOC process. The petition has more than 17,000 signatures.
The ABIM says that it is listening to physicians and is making changes in the process, but also recently said that more than 150,000 physicians had participated in its MOC process – making the May 1 deadline to be listed on the ABIM website as having met the MOC criteria.
But anger is still bubbling up, and was expressed at the AMA’s meeting.
"Practicing physicians on the front lines are increasingly burdened, hassled, and confused by the onerous and expensive process of Maintenance of Certification and Maintenance of Licensure," said Dr. James A. Goodyear, a delegate from Pennsylvania.
Dr. Goodyear introduced the resolution to seek a moratorium on the MOC.
But Dr. Darlyne Menscer, a member of the AMA Council on Medical Education, told the delegates that such a moratorium would put a wedge in the close working relationship the AMA has had with the ABMS. "This is more prescriptive than we can commit to as a council, although we definitely do hear the concerns of the House," added Dr. Menscer.
The AMA has been discussing the concerns about MOC with the ABMS, most recently holding a meeting in Chicago in early June.
Dr. Joshua Cohen, a delegate from the American Academy of Neurology, and a member of the AMA Foundation’s Board of Directors, who attended that meeting, also argued against a moratorium. "It would make it impossible for the AMA to improve the process going forward," said Dr. Cohen.
Dr. Chuck Wilson, a pediatrician and delegate from the North Carolina delegation, also opposed any major change in direction for the AMA. He noted that if the AMA was seen as opposed to MOC, it might not be viewed well. "We all want it to be less onerous," said Dr. Wilson. But, he noted, "the Council on Medical Education is working in that direction. Let’s give them a chance to be successful."
In a statement after the HOD meeting, the AMA said that it "continues to ensure the MOC process does not disrupt physician practice or reduce the capacity of the overall physician workforce." Concerns about MOC "center around the need for relevance to the daily practice of physicians and the better integration into physician practices to optimally support learning and improvement."
On Twitter @aliciaault
CHICAGO – The American Medical Association should continue to work with the American Board of Medical Specialties to address physicians’ concerns about Maintenance of Certification – that was the consensus at the annual meeting of the AMA House of Delegates.
The AMA’s delegates defeated a resolution that asked the organization to put a moratorium on MOC until it was proven to improve the quality of care and patient outcomes. However, they did agree to a new policy that directs the AMA to:
• Explore with independent entities the feasibility of conducting a study to evaluate the effect MOC requirements and Maintenance of Licensure principles have on workforce, practice costs, patient outcomes, patient safety, and patient access.
• Work with the American Board of Medical Specialties and its 24 member boards to collect data on why physicians choose to maintain or discontinue their board certification.
• Work with the ABMS and the Federation of State Medical Boards to study whether MOC and the principles of Maintenance of Licensure are important factors to physicians when deciding whether to retire and whether they have a direct effect on workforce.
• Oppose making MOC mandatory as a condition of medical licensure, and encourage physicians to strive constantly to improve their care of patients by the means they find most effective.
The new policy applies to both the ABMS MOC process and the Osteopathic Continuous Certification (OCC) process.
Physicians have increasingly voiced their concerns about MOC. Dr. Paul Teirstein, chief of cardiology and director of interventional cardiology for Scripps Clinic in La Jolla, Calif., launched a petition drive to overhaul the American Board of Internal Medicine’s MOC process. The petition has more than 17,000 signatures.
The ABIM says that it is listening to physicians and is making changes in the process, but also recently said that more than 150,000 physicians had participated in its MOC process – making the May 1 deadline to be listed on the ABIM website as having met the MOC criteria.
But anger is still bubbling up, and was expressed at the AMA’s meeting.
"Practicing physicians on the front lines are increasingly burdened, hassled, and confused by the onerous and expensive process of Maintenance of Certification and Maintenance of Licensure," said Dr. James A. Goodyear, a delegate from Pennsylvania.
Dr. Goodyear introduced the resolution to seek a moratorium on the MOC.
But Dr. Darlyne Menscer, a member of the AMA Council on Medical Education, told the delegates that such a moratorium would put a wedge in the close working relationship the AMA has had with the ABMS. "This is more prescriptive than we can commit to as a council, although we definitely do hear the concerns of the House," added Dr. Menscer.
The AMA has been discussing the concerns about MOC with the ABMS, most recently holding a meeting in Chicago in early June.
Dr. Joshua Cohen, a delegate from the American Academy of Neurology, and a member of the AMA Foundation’s Board of Directors, who attended that meeting, also argued against a moratorium. "It would make it impossible for the AMA to improve the process going forward," said Dr. Cohen.
Dr. Chuck Wilson, a pediatrician and delegate from the North Carolina delegation, also opposed any major change in direction for the AMA. He noted that if the AMA was seen as opposed to MOC, it might not be viewed well. "We all want it to be less onerous," said Dr. Wilson. But, he noted, "the Council on Medical Education is working in that direction. Let’s give them a chance to be successful."
In a statement after the HOD meeting, the AMA said that it "continues to ensure the MOC process does not disrupt physician practice or reduce the capacity of the overall physician workforce." Concerns about MOC "center around the need for relevance to the daily practice of physicians and the better integration into physician practices to optimally support learning and improvement."
On Twitter @aliciaault
AT THE AMA HOD MEETING
Residents, postop complications linked
BOSTON – Resident participation in emergency general surgery cases was independently associated with a host of complications – pulmonary embolism, surgical site infections, and unplanned reoperation in a secondary analysis of the American College of Surgeons prospective National Surgical Quality Improvement database.
Adequate exposure of residents to emergency general surgery is crucial for surgical training, but academic operating teams should be mindful of this association, Dr. George Kasotakis said at the annual meeting of the American Surgical Association.
Three papers have shown resident participation modestly increases complications in elective surgery, but their impact is not well understood for emergency general surgery, where patient physiology is typically deranged and timely surgery is imperative.
Dr. Kasotakis and his colleagues at Boston University identified 141,010 patients who underwent emergency general surgery procedures in the 2005-2010 American College of Surgeons prospective National Surgical Quality Improvement database. Because of the nonrandom assignment of more complex cases to resident participation, patients were matched 1:1 based on age; gender; use of alcohol, tobacco, and steroids; inpatient status; obesity; diabetes; renal failure; cardiopulmonary disease; and expected probability for morbidity and mortality. Regression models were fitted for each outcome and adjusted for the same risk factors and operative time.
The most common procedures were appendectomy (40%), exploratory laparotomy (8.75%), bowel resection (9.2%), cholecystectomy (6%), and lysis of adhesions (6%).
Thirty-day mortality was similar with and without residents (3.25% vs. 2.96%; P = .082), but hospital length of stay was longer by about a half a day with residents (4.97 days vs. 4.59 days; P = .019), said Dr. Kasotakis, an acute care surgeon and intensivist.
Resident participation added about 20 minutes to operative (75 minutes vs. 59 minutes; P less than .001) and anesthesia (122 minutes vs. 100 minutes; P less than .001) times.
Intraoperative transfusions were more common with residents (3.43% vs. 2.55%; P less than .001), perhaps because of longer operating room times, and, as a result, fewer postoperative transfusions were needed (1.12% vs. 1.28% P = .031), he said. Unplanned reoperations, however, were more common with residents, as well (4.22% vs. 3.80%; P = .002).
Postsurgical superficial wound infections (3.5% vs. 2.78%; P less than .001) and organ space surgical site infections (2.27% vs. 1.77%; P less than .001) were more common in the resident group, while wound dehiscence was not (0.63% vs. 0.69%; P = .266), Dr. Kasotakis noted.
Pulmonary complications were significantly more common in the resident group including postoperative pneumonia (1.85% vs. 1.67%; P = .04), reintubation (1.64% vs. 1.15%; P less than .001), and mechanical ventilation for more than 48 hours (2.87% vs. 2.06%; P less than .001).
The same was true for deep vein thrombosis (DVT) (0.80% vs. 0.62%; P = .002) and pulmonary embolism (PE) (0.43% vs. 0.28%; P less than .001).
Urinary tract infections (UTI) were higher with resident participation (1.45% vs. 1.14%; P less than .001), as was sepsis (2.42% vs. 2.13%; P = .005), likely because of the increase in surgical infections, Dr. Kasotakis said.
Thankfully, significant cardiac complications and septic shock were not more common with residents, he said.
Adjusted analyses
After adjustment for operative duration, case complexity and pre-existing comorbidities, residents did not increase length of stay (odds ratio, 0.07; P = .242) or septic events (OR, 1.07; P = .155), but their participation was still independently associated with about 20% more superficial surgical site infections (odds ratio, 1.23; P less than .001), organ space infections (OR, 1.21; P less than .001), UTIs (OR, 1.23; P = .001), and intraoperative transfusions (OR, 1.20; P = .001), he said.
Also, about 8% more patients required a return trip to the operating room when residents participated (OR, 1.08; P = .041).
"These outcomes can perhaps be attributed to their underdeveloped surgical skills," Dr. Kasotakis said.
The incidence of DVT and PE were also higher by about 25% (OR, 1.25; P = .011) and 40% (OR, 1.42; P = .005), respectively, perhaps because of delayed DVT prophylaxis initiation because of concerns of hemostasis or missed doses due to additional return trips to the emergency department, he suggested.
Interestingly, reintubation and prolonged mechanical ventilation rates were increased by about 40% (OR, 1.38; OR, 1.43; both P less than .001), perhaps because of prolonged operative times or greater resuscitation requirements, he added.
Dr. Kasotakis was quick to point out that this was a secondary analysis of a data set not originally intended to assess the effect of trainee participation, that no information was available on the degree of resident involvement during surgery or in perioperative care, and that participating institutions were skewed toward tertiary centers, which typically receive more complex cases.
"Staff surgeons should supervise as needed and minimize unnecessary [emergency department] time. And residents, for their part, should be well prepared for emergency procedures through simulation training and aim to maximize their operating room efficiency," he suggested.
The results sparked a flurry of rebuttals led off by discussant Dr. Julie Ann Sosa, Duke University, Durham, N.C.,who said they conflict with other analyses showing little to no impact from residents in elective cases.
"If not interpreted with care, policy makers, payers, and the public could construe that surgical care at academic health centers is compromised by trainees, which could have unfortunate ramifications for everyone in the room as well as the trainees and the patients," she said.
Dr. Sosa expressed concern about drawing causal inferences from an observational study in the setting of possible selection bias and said attempts to match for case complexity using CPT codes do not necessarily account for say, "the difference between a routine appendectomy that takes 15-30 minutes and a complex one that takes 3 hours for a perforation."
Some attendees questioned why the authors didn’t match the institutions in the analysis and chose to ascribe all of the outcomes to residents, with a round of applause following the suggestion that the paper should be titled "Academic centers increase emergency surgery complications." Other attendees questioned whether the poor outcomes reflect resident training and supervision.
Dr. George Velmahos, Massachusetts General Hospital, Boston, questioned whether hospitals have a medical/legal responsibility to inform patients that a resident is in the operating room and may impact outcomes.
Dr. Kasotakis said that institutions may want to add a clause to consent paperwork stating that residents and trainees will be participating.
The complete manuscript of this study and its presentation at the American Surgical Association’s 134th Annual Meeting is anticipated to be published in the Annals of Surgery, pending editorial review.
Dr. Kasotakis reported no conflicts.

|
| Dr. Sapan S. Desai |
While it is tempting to value the very high sample sizes reported in studies that utilize administrative database sources such as NSQIP, it is with great caution that we should interpret the results of these studies. Databases are a valuable source of determining incidence, costs, and even some correlations among various factors. Correlations, however, do not imply causation. For instance, one could say that there is a high correlation between the number of taxis on the street after 3 a.m. and the number of crimes, but that does not mean that taxi drivers transform into criminals during the hour of the wolf.
Databases are a blunt tool for identifying trends in data over large samples and over many years. However, as noted in the study presented by Dr. Kasotakis, the NSQIP does not track the extent of resident participation in operative cases. In fact, the exact variable that is tracked is entitled "ATTEND" in position 16 of the NSQIP and is as follows: 1) Attending & Resident in OR, 2) Attending Alone, and 3) Attending Not Present, but Available. It does not characterize the amount of the case completed by the resident, nor does it state the level of training of the resident. It does not mention whether medical students, visitors, nurse trainees, PAs, new scrub techs, or other personnel are present. In fact, there is no way to know who did what during the case, which is a requirement in order to be able to reliably and accurately determine that the resident is the culprit for longer case times and more intraoperative transfusions. This specific variable has insufficient granularity: the conclusion that "[adding] a clause to consent paperwork stating that residents and trainees will be participating in the case" due to the "underdeveloped surgical skills" of trainees, among other factors, is invalid.
As Dr. Sosa indicated, it is dangerous to imply that these correlations somehow imply causation, as this may lead to adverse impacts on training and negatively impact patient care far more than a possible 20 minute increase in the duration of an operation. The sole utility of this paper is that it is hypothesis generating; only a properly designed prospective study can truly study the positive and negative impacts on patient care due to resident education in the operating room. The question is, do we really want to know the answer to this question and will it truly affect what we do now?
Sapan S. Desai, M.D., is the resident medical editor for Vascular Specialist.

|
| Dr. Sapan S. Desai |
While it is tempting to value the very high sample sizes reported in studies that utilize administrative database sources such as NSQIP, it is with great caution that we should interpret the results of these studies. Databases are a valuable source of determining incidence, costs, and even some correlations among various factors. Correlations, however, do not imply causation. For instance, one could say that there is a high correlation between the number of taxis on the street after 3 a.m. and the number of crimes, but that does not mean that taxi drivers transform into criminals during the hour of the wolf.
Databases are a blunt tool for identifying trends in data over large samples and over many years. However, as noted in the study presented by Dr. Kasotakis, the NSQIP does not track the extent of resident participation in operative cases. In fact, the exact variable that is tracked is entitled "ATTEND" in position 16 of the NSQIP and is as follows: 1) Attending & Resident in OR, 2) Attending Alone, and 3) Attending Not Present, but Available. It does not characterize the amount of the case completed by the resident, nor does it state the level of training of the resident. It does not mention whether medical students, visitors, nurse trainees, PAs, new scrub techs, or other personnel are present. In fact, there is no way to know who did what during the case, which is a requirement in order to be able to reliably and accurately determine that the resident is the culprit for longer case times and more intraoperative transfusions. This specific variable has insufficient granularity: the conclusion that "[adding] a clause to consent paperwork stating that residents and trainees will be participating in the case" due to the "underdeveloped surgical skills" of trainees, among other factors, is invalid.
As Dr. Sosa indicated, it is dangerous to imply that these correlations somehow imply causation, as this may lead to adverse impacts on training and negatively impact patient care far more than a possible 20 minute increase in the duration of an operation. The sole utility of this paper is that it is hypothesis generating; only a properly designed prospective study can truly study the positive and negative impacts on patient care due to resident education in the operating room. The question is, do we really want to know the answer to this question and will it truly affect what we do now?
Sapan S. Desai, M.D., is the resident medical editor for Vascular Specialist.

|
| Dr. Sapan S. Desai |
While it is tempting to value the very high sample sizes reported in studies that utilize administrative database sources such as NSQIP, it is with great caution that we should interpret the results of these studies. Databases are a valuable source of determining incidence, costs, and even some correlations among various factors. Correlations, however, do not imply causation. For instance, one could say that there is a high correlation between the number of taxis on the street after 3 a.m. and the number of crimes, but that does not mean that taxi drivers transform into criminals during the hour of the wolf.
Databases are a blunt tool for identifying trends in data over large samples and over many years. However, as noted in the study presented by Dr. Kasotakis, the NSQIP does not track the extent of resident participation in operative cases. In fact, the exact variable that is tracked is entitled "ATTEND" in position 16 of the NSQIP and is as follows: 1) Attending & Resident in OR, 2) Attending Alone, and 3) Attending Not Present, but Available. It does not characterize the amount of the case completed by the resident, nor does it state the level of training of the resident. It does not mention whether medical students, visitors, nurse trainees, PAs, new scrub techs, or other personnel are present. In fact, there is no way to know who did what during the case, which is a requirement in order to be able to reliably and accurately determine that the resident is the culprit for longer case times and more intraoperative transfusions. This specific variable has insufficient granularity: the conclusion that "[adding] a clause to consent paperwork stating that residents and trainees will be participating in the case" due to the "underdeveloped surgical skills" of trainees, among other factors, is invalid.
As Dr. Sosa indicated, it is dangerous to imply that these correlations somehow imply causation, as this may lead to adverse impacts on training and negatively impact patient care far more than a possible 20 minute increase in the duration of an operation. The sole utility of this paper is that it is hypothesis generating; only a properly designed prospective study can truly study the positive and negative impacts on patient care due to resident education in the operating room. The question is, do we really want to know the answer to this question and will it truly affect what we do now?
Sapan S. Desai, M.D., is the resident medical editor for Vascular Specialist.
BOSTON – Resident participation in emergency general surgery cases was independently associated with a host of complications – pulmonary embolism, surgical site infections, and unplanned reoperation in a secondary analysis of the American College of Surgeons prospective National Surgical Quality Improvement database.
Adequate exposure of residents to emergency general surgery is crucial for surgical training, but academic operating teams should be mindful of this association, Dr. George Kasotakis said at the annual meeting of the American Surgical Association.
Three papers have shown resident participation modestly increases complications in elective surgery, but their impact is not well understood for emergency general surgery, where patient physiology is typically deranged and timely surgery is imperative.
Dr. Kasotakis and his colleagues at Boston University identified 141,010 patients who underwent emergency general surgery procedures in the 2005-2010 American College of Surgeons prospective National Surgical Quality Improvement database. Because of the nonrandom assignment of more complex cases to resident participation, patients were matched 1:1 based on age; gender; use of alcohol, tobacco, and steroids; inpatient status; obesity; diabetes; renal failure; cardiopulmonary disease; and expected probability for morbidity and mortality. Regression models were fitted for each outcome and adjusted for the same risk factors and operative time.
The most common procedures were appendectomy (40%), exploratory laparotomy (8.75%), bowel resection (9.2%), cholecystectomy (6%), and lysis of adhesions (6%).
Thirty-day mortality was similar with and without residents (3.25% vs. 2.96%; P = .082), but hospital length of stay was longer by about a half a day with residents (4.97 days vs. 4.59 days; P = .019), said Dr. Kasotakis, an acute care surgeon and intensivist.
Resident participation added about 20 minutes to operative (75 minutes vs. 59 minutes; P less than .001) and anesthesia (122 minutes vs. 100 minutes; P less than .001) times.
Intraoperative transfusions were more common with residents (3.43% vs. 2.55%; P less than .001), perhaps because of longer operating room times, and, as a result, fewer postoperative transfusions were needed (1.12% vs. 1.28% P = .031), he said. Unplanned reoperations, however, were more common with residents, as well (4.22% vs. 3.80%; P = .002).
Postsurgical superficial wound infections (3.5% vs. 2.78%; P less than .001) and organ space surgical site infections (2.27% vs. 1.77%; P less than .001) were more common in the resident group, while wound dehiscence was not (0.63% vs. 0.69%; P = .266), Dr. Kasotakis noted.
Pulmonary complications were significantly more common in the resident group including postoperative pneumonia (1.85% vs. 1.67%; P = .04), reintubation (1.64% vs. 1.15%; P less than .001), and mechanical ventilation for more than 48 hours (2.87% vs. 2.06%; P less than .001).
The same was true for deep vein thrombosis (DVT) (0.80% vs. 0.62%; P = .002) and pulmonary embolism (PE) (0.43% vs. 0.28%; P less than .001).
Urinary tract infections (UTI) were higher with resident participation (1.45% vs. 1.14%; P less than .001), as was sepsis (2.42% vs. 2.13%; P = .005), likely because of the increase in surgical infections, Dr. Kasotakis said.
Thankfully, significant cardiac complications and septic shock were not more common with residents, he said.
Adjusted analyses
After adjustment for operative duration, case complexity and pre-existing comorbidities, residents did not increase length of stay (odds ratio, 0.07; P = .242) or septic events (OR, 1.07; P = .155), but their participation was still independently associated with about 20% more superficial surgical site infections (odds ratio, 1.23; P less than .001), organ space infections (OR, 1.21; P less than .001), UTIs (OR, 1.23; P = .001), and intraoperative transfusions (OR, 1.20; P = .001), he said.
Also, about 8% more patients required a return trip to the operating room when residents participated (OR, 1.08; P = .041).
"These outcomes can perhaps be attributed to their underdeveloped surgical skills," Dr. Kasotakis said.
The incidence of DVT and PE were also higher by about 25% (OR, 1.25; P = .011) and 40% (OR, 1.42; P = .005), respectively, perhaps because of delayed DVT prophylaxis initiation because of concerns of hemostasis or missed doses due to additional return trips to the emergency department, he suggested.
Interestingly, reintubation and prolonged mechanical ventilation rates were increased by about 40% (OR, 1.38; OR, 1.43; both P less than .001), perhaps because of prolonged operative times or greater resuscitation requirements, he added.
Dr. Kasotakis was quick to point out that this was a secondary analysis of a data set not originally intended to assess the effect of trainee participation, that no information was available on the degree of resident involvement during surgery or in perioperative care, and that participating institutions were skewed toward tertiary centers, which typically receive more complex cases.
"Staff surgeons should supervise as needed and minimize unnecessary [emergency department] time. And residents, for their part, should be well prepared for emergency procedures through simulation training and aim to maximize their operating room efficiency," he suggested.
The results sparked a flurry of rebuttals led off by discussant Dr. Julie Ann Sosa, Duke University, Durham, N.C.,who said they conflict with other analyses showing little to no impact from residents in elective cases.
"If not interpreted with care, policy makers, payers, and the public could construe that surgical care at academic health centers is compromised by trainees, which could have unfortunate ramifications for everyone in the room as well as the trainees and the patients," she said.
Dr. Sosa expressed concern about drawing causal inferences from an observational study in the setting of possible selection bias and said attempts to match for case complexity using CPT codes do not necessarily account for say, "the difference between a routine appendectomy that takes 15-30 minutes and a complex one that takes 3 hours for a perforation."
Some attendees questioned why the authors didn’t match the institutions in the analysis and chose to ascribe all of the outcomes to residents, with a round of applause following the suggestion that the paper should be titled "Academic centers increase emergency surgery complications." Other attendees questioned whether the poor outcomes reflect resident training and supervision.
Dr. George Velmahos, Massachusetts General Hospital, Boston, questioned whether hospitals have a medical/legal responsibility to inform patients that a resident is in the operating room and may impact outcomes.
Dr. Kasotakis said that institutions may want to add a clause to consent paperwork stating that residents and trainees will be participating.
The complete manuscript of this study and its presentation at the American Surgical Association’s 134th Annual Meeting is anticipated to be published in the Annals of Surgery, pending editorial review.
Dr. Kasotakis reported no conflicts.
BOSTON – Resident participation in emergency general surgery cases was independently associated with a host of complications – pulmonary embolism, surgical site infections, and unplanned reoperation in a secondary analysis of the American College of Surgeons prospective National Surgical Quality Improvement database.
Adequate exposure of residents to emergency general surgery is crucial for surgical training, but academic operating teams should be mindful of this association, Dr. George Kasotakis said at the annual meeting of the American Surgical Association.
Three papers have shown resident participation modestly increases complications in elective surgery, but their impact is not well understood for emergency general surgery, where patient physiology is typically deranged and timely surgery is imperative.
Dr. Kasotakis and his colleagues at Boston University identified 141,010 patients who underwent emergency general surgery procedures in the 2005-2010 American College of Surgeons prospective National Surgical Quality Improvement database. Because of the nonrandom assignment of more complex cases to resident participation, patients were matched 1:1 based on age; gender; use of alcohol, tobacco, and steroids; inpatient status; obesity; diabetes; renal failure; cardiopulmonary disease; and expected probability for morbidity and mortality. Regression models were fitted for each outcome and adjusted for the same risk factors and operative time.
The most common procedures were appendectomy (40%), exploratory laparotomy (8.75%), bowel resection (9.2%), cholecystectomy (6%), and lysis of adhesions (6%).
Thirty-day mortality was similar with and without residents (3.25% vs. 2.96%; P = .082), but hospital length of stay was longer by about a half a day with residents (4.97 days vs. 4.59 days; P = .019), said Dr. Kasotakis, an acute care surgeon and intensivist.
Resident participation added about 20 minutes to operative (75 minutes vs. 59 minutes; P less than .001) and anesthesia (122 minutes vs. 100 minutes; P less than .001) times.
Intraoperative transfusions were more common with residents (3.43% vs. 2.55%; P less than .001), perhaps because of longer operating room times, and, as a result, fewer postoperative transfusions were needed (1.12% vs. 1.28% P = .031), he said. Unplanned reoperations, however, were more common with residents, as well (4.22% vs. 3.80%; P = .002).
Postsurgical superficial wound infections (3.5% vs. 2.78%; P less than .001) and organ space surgical site infections (2.27% vs. 1.77%; P less than .001) were more common in the resident group, while wound dehiscence was not (0.63% vs. 0.69%; P = .266), Dr. Kasotakis noted.
Pulmonary complications were significantly more common in the resident group including postoperative pneumonia (1.85% vs. 1.67%; P = .04), reintubation (1.64% vs. 1.15%; P less than .001), and mechanical ventilation for more than 48 hours (2.87% vs. 2.06%; P less than .001).
The same was true for deep vein thrombosis (DVT) (0.80% vs. 0.62%; P = .002) and pulmonary embolism (PE) (0.43% vs. 0.28%; P less than .001).
Urinary tract infections (UTI) were higher with resident participation (1.45% vs. 1.14%; P less than .001), as was sepsis (2.42% vs. 2.13%; P = .005), likely because of the increase in surgical infections, Dr. Kasotakis said.
Thankfully, significant cardiac complications and septic shock were not more common with residents, he said.
Adjusted analyses
After adjustment for operative duration, case complexity and pre-existing comorbidities, residents did not increase length of stay (odds ratio, 0.07; P = .242) or septic events (OR, 1.07; P = .155), but their participation was still independently associated with about 20% more superficial surgical site infections (odds ratio, 1.23; P less than .001), organ space infections (OR, 1.21; P less than .001), UTIs (OR, 1.23; P = .001), and intraoperative transfusions (OR, 1.20; P = .001), he said.
Also, about 8% more patients required a return trip to the operating room when residents participated (OR, 1.08; P = .041).
"These outcomes can perhaps be attributed to their underdeveloped surgical skills," Dr. Kasotakis said.
The incidence of DVT and PE were also higher by about 25% (OR, 1.25; P = .011) and 40% (OR, 1.42; P = .005), respectively, perhaps because of delayed DVT prophylaxis initiation because of concerns of hemostasis or missed doses due to additional return trips to the emergency department, he suggested.
Interestingly, reintubation and prolonged mechanical ventilation rates were increased by about 40% (OR, 1.38; OR, 1.43; both P less than .001), perhaps because of prolonged operative times or greater resuscitation requirements, he added.
Dr. Kasotakis was quick to point out that this was a secondary analysis of a data set not originally intended to assess the effect of trainee participation, that no information was available on the degree of resident involvement during surgery or in perioperative care, and that participating institutions were skewed toward tertiary centers, which typically receive more complex cases.
"Staff surgeons should supervise as needed and minimize unnecessary [emergency department] time. And residents, for their part, should be well prepared for emergency procedures through simulation training and aim to maximize their operating room efficiency," he suggested.
The results sparked a flurry of rebuttals led off by discussant Dr. Julie Ann Sosa, Duke University, Durham, N.C.,who said they conflict with other analyses showing little to no impact from residents in elective cases.
"If not interpreted with care, policy makers, payers, and the public could construe that surgical care at academic health centers is compromised by trainees, which could have unfortunate ramifications for everyone in the room as well as the trainees and the patients," she said.
Dr. Sosa expressed concern about drawing causal inferences from an observational study in the setting of possible selection bias and said attempts to match for case complexity using CPT codes do not necessarily account for say, "the difference between a routine appendectomy that takes 15-30 minutes and a complex one that takes 3 hours for a perforation."
Some attendees questioned why the authors didn’t match the institutions in the analysis and chose to ascribe all of the outcomes to residents, with a round of applause following the suggestion that the paper should be titled "Academic centers increase emergency surgery complications." Other attendees questioned whether the poor outcomes reflect resident training and supervision.
Dr. George Velmahos, Massachusetts General Hospital, Boston, questioned whether hospitals have a medical/legal responsibility to inform patients that a resident is in the operating room and may impact outcomes.
Dr. Kasotakis said that institutions may want to add a clause to consent paperwork stating that residents and trainees will be participating.
The complete manuscript of this study and its presentation at the American Surgical Association’s 134th Annual Meeting is anticipated to be published in the Annals of Surgery, pending editorial review.
Dr. Kasotakis reported no conflicts.
Major finding: Rates were significantly higher with resident participation than without for DVT (0.80% vs. 0.62%; P = .002) and PE (0.43% vs. 0.28%; P less than .001).
Data source: A secondary analysis of 141,010 emergency general surgery cases in the prospective ACS NSQIP database.
Disclosures: Dr. Kasotakis and his coauthors reported no conflicting interests.
Endofibrinolysis a 'game changer' in acute PE
WASHINGTON – Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive or submassive pulmonary embolism significantly improves right ventricular function, reduces pulmonary hypertension and angiographic evidence of obstruction, and lessens the risk of fibrinolysis-associated intracranial hemorrhage, according to a prospective multicenter clinical trial.
"By minimizing the risk of intracranial bleeding, ultrasound-facilitated, catheter-directed, low-dose fibrinolysis represents a potential game changer in the treatment of high-risk pulmonary embolism patients," Dr. Gregory Piazza said in presenting the results of the SEATTLE II study at the annual meeting of the American College of Cardiology.
Full-dose systemic fibrinolysis has long been the go-to advanced therapy for high-risk pulmonary embolism (PE), but physicians are leery of the associated 2%-3% risk of catastrophic intracranial hemorrhage, noted Dr. Piazza of Brigham and Women’s Hospital and Harvard University, Boston.
SEATTLE II was a single-arm, 21-site, prospective study in which 150 patients with high-risk PE underwent treatment using the commercially available EKOS EkoSonic Endovascular System.
Twenty-one percent of patients had massive PE, defined as presentation with syncope, cardiogenic shock, resuscitated cardiac arrest, or persistent hypotension. The remaining 79% had submassive PE, with normal blood pressure but evidence of right ventricular dysfunction. All participants had to have a right ventricular/left ventricular ratio (RV/LV) of 0.9 or greater on the same chest CT scan used in diagnosing the PE. This CT documentation of RV dysfunction has been associated in a meta-analysis of patients with submassive PE with a 7.4-fold increased risk of death from PE, compared with normotensive PE patients with normal RV function (J. Thromb. Haemost. 2013;11:1823-32).
The primary endpoint was change in RV/LV on chest CT from baseline to 48 hours after initiation of fibrinolysis. This ratio improved from 1.55 to 1.13, for a statistically and clinically significant 27% reduction. A similar-size improvement was seen in pulmonary artery systolic pressure – a secondary efficacy endpoint – which decreased from 51.4 mm Hg before treatment to 37.5 mm Hg post procedure and 36.9 mm Hg at 48 hours. Both efficacy endpoints improved to a similar extent regardless of whether patients had massive or submassive PE.
The mean Modified Miller Pulmonary Artery Angiographic Obstruction Score improved by 30%, from 22.5 pretreatment to 15.8 at 48 hours.
Three in-hospital deaths occurred. One was due to massive PE that occurred before the fibrinolysis procedure could be completed. The others were not directly attributable to PE: One involved overwhelming sepsis and the other was due to progressive respiratory failure. Major bleeding occurred in 11% of patients; however, 16 of the 17 events were classified as GUSTO moderate bleeds, with only a single GUSTO severe hemorrhage.
There were no intracranial hemorrhages.
The fibrinolytic agent used in SEATTLE II was tissue plasminogen activator, delivered at 1 mg/hr for a total dose of 24 mg. Patients with unilateral PE received a single device and 24 hours of infusion time. The 86% of patients who had bilateral disease got two devices and 12 hours of therapy.
The proprietary EKOS system consists of two catheters: an outer infusion catheter with side holes that elute the fibrinolytic agent, and an inner-core catheter with ultrasound transducers placed at regular intervals. These transducers produce low-intensity ultrasound that serves two purposes. Through a process called acoustic streaming, the low-intensity ultrasound helps push the fibrinolytic agent closer to the thrombus. Plus, the ultrasound energy causes the clot fibrin to reconfigure from a tight lattice to a more porous structure that promotes deeper penetration of the fibrinolytic, Dr. Piazza explained.
Dr. Piazza said the next step in defining the EKOS system’s role in clinical practice will be to study briefer infusion times as a means of achieving faster patient improvement with reduced use of hospital resources.
The EKOS system has been approved in the United States since 2005 for treatment of blood clots in the arms and legs. In Europe it gained an additional indication for treatment of massive and submassive PE in 2011.
The SEATTLE II study was sponsored by EKOS Corp. Dr. Piazza reported receiving a research grant from the company.
WASHINGTON – Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive or submassive pulmonary embolism significantly improves right ventricular function, reduces pulmonary hypertension and angiographic evidence of obstruction, and lessens the risk of fibrinolysis-associated intracranial hemorrhage, according to a prospective multicenter clinical trial.
"By minimizing the risk of intracranial bleeding, ultrasound-facilitated, catheter-directed, low-dose fibrinolysis represents a potential game changer in the treatment of high-risk pulmonary embolism patients," Dr. Gregory Piazza said in presenting the results of the SEATTLE II study at the annual meeting of the American College of Cardiology.
Full-dose systemic fibrinolysis has long been the go-to advanced therapy for high-risk pulmonary embolism (PE), but physicians are leery of the associated 2%-3% risk of catastrophic intracranial hemorrhage, noted Dr. Piazza of Brigham and Women’s Hospital and Harvard University, Boston.
SEATTLE II was a single-arm, 21-site, prospective study in which 150 patients with high-risk PE underwent treatment using the commercially available EKOS EkoSonic Endovascular System.
Twenty-one percent of patients had massive PE, defined as presentation with syncope, cardiogenic shock, resuscitated cardiac arrest, or persistent hypotension. The remaining 79% had submassive PE, with normal blood pressure but evidence of right ventricular dysfunction. All participants had to have a right ventricular/left ventricular ratio (RV/LV) of 0.9 or greater on the same chest CT scan used in diagnosing the PE. This CT documentation of RV dysfunction has been associated in a meta-analysis of patients with submassive PE with a 7.4-fold increased risk of death from PE, compared with normotensive PE patients with normal RV function (J. Thromb. Haemost. 2013;11:1823-32).
The primary endpoint was change in RV/LV on chest CT from baseline to 48 hours after initiation of fibrinolysis. This ratio improved from 1.55 to 1.13, for a statistically and clinically significant 27% reduction. A similar-size improvement was seen in pulmonary artery systolic pressure – a secondary efficacy endpoint – which decreased from 51.4 mm Hg before treatment to 37.5 mm Hg post procedure and 36.9 mm Hg at 48 hours. Both efficacy endpoints improved to a similar extent regardless of whether patients had massive or submassive PE.
The mean Modified Miller Pulmonary Artery Angiographic Obstruction Score improved by 30%, from 22.5 pretreatment to 15.8 at 48 hours.
Three in-hospital deaths occurred. One was due to massive PE that occurred before the fibrinolysis procedure could be completed. The others were not directly attributable to PE: One involved overwhelming sepsis and the other was due to progressive respiratory failure. Major bleeding occurred in 11% of patients; however, 16 of the 17 events were classified as GUSTO moderate bleeds, with only a single GUSTO severe hemorrhage.
There were no intracranial hemorrhages.
The fibrinolytic agent used in SEATTLE II was tissue plasminogen activator, delivered at 1 mg/hr for a total dose of 24 mg. Patients with unilateral PE received a single device and 24 hours of infusion time. The 86% of patients who had bilateral disease got two devices and 12 hours of therapy.
The proprietary EKOS system consists of two catheters: an outer infusion catheter with side holes that elute the fibrinolytic agent, and an inner-core catheter with ultrasound transducers placed at regular intervals. These transducers produce low-intensity ultrasound that serves two purposes. Through a process called acoustic streaming, the low-intensity ultrasound helps push the fibrinolytic agent closer to the thrombus. Plus, the ultrasound energy causes the clot fibrin to reconfigure from a tight lattice to a more porous structure that promotes deeper penetration of the fibrinolytic, Dr. Piazza explained.
Dr. Piazza said the next step in defining the EKOS system’s role in clinical practice will be to study briefer infusion times as a means of achieving faster patient improvement with reduced use of hospital resources.
The EKOS system has been approved in the United States since 2005 for treatment of blood clots in the arms and legs. In Europe it gained an additional indication for treatment of massive and submassive PE in 2011.
The SEATTLE II study was sponsored by EKOS Corp. Dr. Piazza reported receiving a research grant from the company.
WASHINGTON – Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive or submassive pulmonary embolism significantly improves right ventricular function, reduces pulmonary hypertension and angiographic evidence of obstruction, and lessens the risk of fibrinolysis-associated intracranial hemorrhage, according to a prospective multicenter clinical trial.
"By minimizing the risk of intracranial bleeding, ultrasound-facilitated, catheter-directed, low-dose fibrinolysis represents a potential game changer in the treatment of high-risk pulmonary embolism patients," Dr. Gregory Piazza said in presenting the results of the SEATTLE II study at the annual meeting of the American College of Cardiology.
Full-dose systemic fibrinolysis has long been the go-to advanced therapy for high-risk pulmonary embolism (PE), but physicians are leery of the associated 2%-3% risk of catastrophic intracranial hemorrhage, noted Dr. Piazza of Brigham and Women’s Hospital and Harvard University, Boston.
SEATTLE II was a single-arm, 21-site, prospective study in which 150 patients with high-risk PE underwent treatment using the commercially available EKOS EkoSonic Endovascular System.
Twenty-one percent of patients had massive PE, defined as presentation with syncope, cardiogenic shock, resuscitated cardiac arrest, or persistent hypotension. The remaining 79% had submassive PE, with normal blood pressure but evidence of right ventricular dysfunction. All participants had to have a right ventricular/left ventricular ratio (RV/LV) of 0.9 or greater on the same chest CT scan used in diagnosing the PE. This CT documentation of RV dysfunction has been associated in a meta-analysis of patients with submassive PE with a 7.4-fold increased risk of death from PE, compared with normotensive PE patients with normal RV function (J. Thromb. Haemost. 2013;11:1823-32).
The primary endpoint was change in RV/LV on chest CT from baseline to 48 hours after initiation of fibrinolysis. This ratio improved from 1.55 to 1.13, for a statistically and clinically significant 27% reduction. A similar-size improvement was seen in pulmonary artery systolic pressure – a secondary efficacy endpoint – which decreased from 51.4 mm Hg before treatment to 37.5 mm Hg post procedure and 36.9 mm Hg at 48 hours. Both efficacy endpoints improved to a similar extent regardless of whether patients had massive or submassive PE.
The mean Modified Miller Pulmonary Artery Angiographic Obstruction Score improved by 30%, from 22.5 pretreatment to 15.8 at 48 hours.
Three in-hospital deaths occurred. One was due to massive PE that occurred before the fibrinolysis procedure could be completed. The others were not directly attributable to PE: One involved overwhelming sepsis and the other was due to progressive respiratory failure. Major bleeding occurred in 11% of patients; however, 16 of the 17 events were classified as GUSTO moderate bleeds, with only a single GUSTO severe hemorrhage.
There were no intracranial hemorrhages.
The fibrinolytic agent used in SEATTLE II was tissue plasminogen activator, delivered at 1 mg/hr for a total dose of 24 mg. Patients with unilateral PE received a single device and 24 hours of infusion time. The 86% of patients who had bilateral disease got two devices and 12 hours of therapy.
The proprietary EKOS system consists of two catheters: an outer infusion catheter with side holes that elute the fibrinolytic agent, and an inner-core catheter with ultrasound transducers placed at regular intervals. These transducers produce low-intensity ultrasound that serves two purposes. Through a process called acoustic streaming, the low-intensity ultrasound helps push the fibrinolytic agent closer to the thrombus. Plus, the ultrasound energy causes the clot fibrin to reconfigure from a tight lattice to a more porous structure that promotes deeper penetration of the fibrinolytic, Dr. Piazza explained.
Dr. Piazza said the next step in defining the EKOS system’s role in clinical practice will be to study briefer infusion times as a means of achieving faster patient improvement with reduced use of hospital resources.
The EKOS system has been approved in the United States since 2005 for treatment of blood clots in the arms and legs. In Europe it gained an additional indication for treatment of massive and submassive PE in 2011.
The SEATTLE II study was sponsored by EKOS Corp. Dr. Piazza reported receiving a research grant from the company.
Risks of two types of polymer stents comparable
Biodegradable biolimus-eluting stents are as safe and effective as durable everolimus-eluting stents at 2 years’ follow-up, with no significant differences seen in rates of target lesion revascularization, mortality, or myocardial infarction.
The findings come from NEXT (NOBORI Biolimus-Eluting vs. XIENCE/PROMUS Everolimus-Eluting Stent Trial), a 3-year randomized trial led by Dr. Masahiro Natsuaki of Saiseikai Fukuoka (Japan) General Hospital. They were presented at the annual meeting of the American College of Cardiology and published in JAMA (doi:10.1001/jama.2014.3584).
NEXT compared the biodegradable polymer biolimus-eluting stent (BP-BES) to the durable polymer everolimus-eluting stent (DP-EES), measuring target lesion revascularization and whether BP-BES carries a risk for excess mortality or MI, compared with DP-EES, as shorter studies and meta-analyses have suggested.
Dr. Natsuaki and colleagues randomized 3,235 patients from nearly 100 treatment centers to BP-BES (n = 1,617) or DP-EES (n = 1,618), with 98% of all patients completing follow-up. Mortality and MI were comparable for both stents (7.8% for BP-BES vs. 7.7% for DP-EES; noninferiority, P = .003), and the need for target lesion revascularization was also comparable for both stents (6.2% vs. 6%; noninferiority, P less than .001). The researchers noted that "2 years is not long enough to confirm the long-term safety of BP-BES, and the study was underpowered for the interim analysis. Follow-up at 3 years will be important."
NEXT was sponsored by Terumo Japan, maker of the biodegradable stents. Two investigators disclosed they are advisers for Terumo Japan and Abbott Vascular Japan, maker of the durable polymer stents used.
Biodegradable biolimus-eluting stents are as safe and effective as durable everolimus-eluting stents at 2 years’ follow-up, with no significant differences seen in rates of target lesion revascularization, mortality, or myocardial infarction.
The findings come from NEXT (NOBORI Biolimus-Eluting vs. XIENCE/PROMUS Everolimus-Eluting Stent Trial), a 3-year randomized trial led by Dr. Masahiro Natsuaki of Saiseikai Fukuoka (Japan) General Hospital. They were presented at the annual meeting of the American College of Cardiology and published in JAMA (doi:10.1001/jama.2014.3584).
NEXT compared the biodegradable polymer biolimus-eluting stent (BP-BES) to the durable polymer everolimus-eluting stent (DP-EES), measuring target lesion revascularization and whether BP-BES carries a risk for excess mortality or MI, compared with DP-EES, as shorter studies and meta-analyses have suggested.
Dr. Natsuaki and colleagues randomized 3,235 patients from nearly 100 treatment centers to BP-BES (n = 1,617) or DP-EES (n = 1,618), with 98% of all patients completing follow-up. Mortality and MI were comparable for both stents (7.8% for BP-BES vs. 7.7% for DP-EES; noninferiority, P = .003), and the need for target lesion revascularization was also comparable for both stents (6.2% vs. 6%; noninferiority, P less than .001). The researchers noted that "2 years is not long enough to confirm the long-term safety of BP-BES, and the study was underpowered for the interim analysis. Follow-up at 3 years will be important."
NEXT was sponsored by Terumo Japan, maker of the biodegradable stents. Two investigators disclosed they are advisers for Terumo Japan and Abbott Vascular Japan, maker of the durable polymer stents used.
Biodegradable biolimus-eluting stents are as safe and effective as durable everolimus-eluting stents at 2 years’ follow-up, with no significant differences seen in rates of target lesion revascularization, mortality, or myocardial infarction.
The findings come from NEXT (NOBORI Biolimus-Eluting vs. XIENCE/PROMUS Everolimus-Eluting Stent Trial), a 3-year randomized trial led by Dr. Masahiro Natsuaki of Saiseikai Fukuoka (Japan) General Hospital. They were presented at the annual meeting of the American College of Cardiology and published in JAMA (doi:10.1001/jama.2014.3584).
NEXT compared the biodegradable polymer biolimus-eluting stent (BP-BES) to the durable polymer everolimus-eluting stent (DP-EES), measuring target lesion revascularization and whether BP-BES carries a risk for excess mortality or MI, compared with DP-EES, as shorter studies and meta-analyses have suggested.
Dr. Natsuaki and colleagues randomized 3,235 patients from nearly 100 treatment centers to BP-BES (n = 1,617) or DP-EES (n = 1,618), with 98% of all patients completing follow-up. Mortality and MI were comparable for both stents (7.8% for BP-BES vs. 7.7% for DP-EES; noninferiority, P = .003), and the need for target lesion revascularization was also comparable for both stents (6.2% vs. 6%; noninferiority, P less than .001). The researchers noted that "2 years is not long enough to confirm the long-term safety of BP-BES, and the study was underpowered for the interim analysis. Follow-up at 3 years will be important."
NEXT was sponsored by Terumo Japan, maker of the biodegradable stents. Two investigators disclosed they are advisers for Terumo Japan and Abbott Vascular Japan, maker of the durable polymer stents used.
Major finding: Mortality and myocardial infarction were comparable for BP-BES (7.8%) vs. DP-EES (7.7%); noninferiority, P = .003), as was the need for target lesion revascularization (6.2% vs. 6%; noninferiority, P less than .001).
Data source: The NOBORI Biolimus-Eluting vs. XIENCE/PROMUS Everolimus-Eluting Stent Trial, a randomized trial of 3,235 patients.
Disclosures: NEXT was sponsored by Terumo Japan, the maker of BP-BES. Two investigators disclosed that they serve as advisers for Terumo Japan and Abbott Vascular Japan, maker of DP-EES.
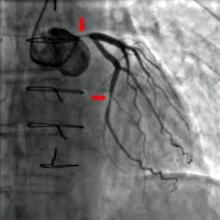
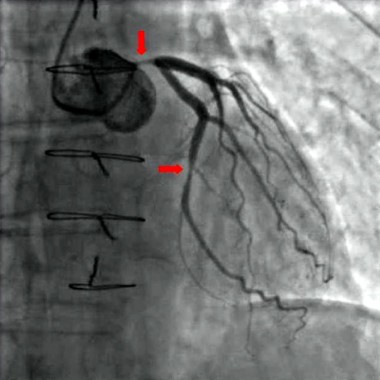
Angiography timing not a risk for acute kidney injury
Acute kidney injury is a serious adverse effect of cardiac surgery, and contrast-induced nephropathy from coronary angiography has been suggested as a potentially important component. However, the results of a retrospective study of more than 2,500 patients showed that acute kidney injury is significantly higher only in those patients who have combined cardiac surgery within 24 hours of catheterization.
The study by Dr. Giovanni Mariscalco of the Varese (Italy) University Hospital, and his colleagues assessed all consecutive patients undergoing cardiac surgery at the hospital between Jan. 1, 2005, and Dec. 31, 2011. The operations performed were isolated coronary artery bypass grafting (CABG), valve surgery with or without concomitant CABG, and proximal aortic procedures. After exclusion of patients who did not undergo cardiopulmonary bypass, a known major cause of acute kidney injury (AKI), and those who died during the procedure, a total of 2,504 patients remained. These patients had a mean age of 68.4 years; 67.3% were men, according to the report.
The primary endpoint was the effect of timing between cardiac catheterization and surgery on the development of AKI. Postoperative AKI was defined by the consensus RIFLE criteria (risk, injury, failure, loss of function, and end-stage renal disease), using the maximal change in serum creatinine and the estimated glomerular filtration rate during the first 7 days after surgery, compared with baseline.
The overall incidence of AKI after surgery was 9.2% (230/2,504 patients). A breakdown by procedure showed that AKI occurred in 7.7% of isolated CABG patients, 12.2% of isolated valve patients, 9.5% of combined-procedure patients, and 9.5% of the proximal aorta surgery patients (Int. J. Cardiol. 2014;173:46-54).
As has been seen in previous studies, AKI was associated with patient-specific pre- and perioperative variables, including increased patient age, added comorbidities, longer cardiopulmonary bypass (CPB) times, higher rates of combined procedures, and the use of intra-aortic balloon pumps.
Unadjusted analysis of the total cohort showed AKI was significantly associated with contrast exposure within 1 day of surgery. However, in multivariable analysis, the time interval between catheterization and surgery was not an independent predictor of postoperative AKI for the total cohort. In subgroup analysis, only the combined valve and CABG group of patients showed an independent association of contrast exposure within 1 day before surgery and AKI in both the prematched (odds ratio, 2.69; P = .004) and the postmatched (OR, 3.68; P = .014) groups.
"Avoiding surgery within 1 day after contrast exposure should be recommended for patients undergoing valve surgery with concomitant CABG only. For other types of cardiac operations, delaying cardiac surgery after contrast exposure seems not to be justified," the researchers concluded.
The study was supported by the Fondazione Cesare Bartorelli. The authors reported having no disclosures.
mlesney@frontlinemedcom.com
Acute kidney injury is a serious adverse effect of cardiac surgery, and contrast-induced nephropathy from coronary angiography has been suggested as a potentially important component. However, the results of a retrospective study of more than 2,500 patients showed that acute kidney injury is significantly higher only in those patients who have combined cardiac surgery within 24 hours of catheterization.
The study by Dr. Giovanni Mariscalco of the Varese (Italy) University Hospital, and his colleagues assessed all consecutive patients undergoing cardiac surgery at the hospital between Jan. 1, 2005, and Dec. 31, 2011. The operations performed were isolated coronary artery bypass grafting (CABG), valve surgery with or without concomitant CABG, and proximal aortic procedures. After exclusion of patients who did not undergo cardiopulmonary bypass, a known major cause of acute kidney injury (AKI), and those who died during the procedure, a total of 2,504 patients remained. These patients had a mean age of 68.4 years; 67.3% were men, according to the report.
The primary endpoint was the effect of timing between cardiac catheterization and surgery on the development of AKI. Postoperative AKI was defined by the consensus RIFLE criteria (risk, injury, failure, loss of function, and end-stage renal disease), using the maximal change in serum creatinine and the estimated glomerular filtration rate during the first 7 days after surgery, compared with baseline.
The overall incidence of AKI after surgery was 9.2% (230/2,504 patients). A breakdown by procedure showed that AKI occurred in 7.7% of isolated CABG patients, 12.2% of isolated valve patients, 9.5% of combined-procedure patients, and 9.5% of the proximal aorta surgery patients (Int. J. Cardiol. 2014;173:46-54).
As has been seen in previous studies, AKI was associated with patient-specific pre- and perioperative variables, including increased patient age, added comorbidities, longer cardiopulmonary bypass (CPB) times, higher rates of combined procedures, and the use of intra-aortic balloon pumps.
Unadjusted analysis of the total cohort showed AKI was significantly associated with contrast exposure within 1 day of surgery. However, in multivariable analysis, the time interval between catheterization and surgery was not an independent predictor of postoperative AKI for the total cohort. In subgroup analysis, only the combined valve and CABG group of patients showed an independent association of contrast exposure within 1 day before surgery and AKI in both the prematched (odds ratio, 2.69; P = .004) and the postmatched (OR, 3.68; P = .014) groups.
"Avoiding surgery within 1 day after contrast exposure should be recommended for patients undergoing valve surgery with concomitant CABG only. For other types of cardiac operations, delaying cardiac surgery after contrast exposure seems not to be justified," the researchers concluded.
The study was supported by the Fondazione Cesare Bartorelli. The authors reported having no disclosures.
mlesney@frontlinemedcom.com
Acute kidney injury is a serious adverse effect of cardiac surgery, and contrast-induced nephropathy from coronary angiography has been suggested as a potentially important component. However, the results of a retrospective study of more than 2,500 patients showed that acute kidney injury is significantly higher only in those patients who have combined cardiac surgery within 24 hours of catheterization.
The study by Dr. Giovanni Mariscalco of the Varese (Italy) University Hospital, and his colleagues assessed all consecutive patients undergoing cardiac surgery at the hospital between Jan. 1, 2005, and Dec. 31, 2011. The operations performed were isolated coronary artery bypass grafting (CABG), valve surgery with or without concomitant CABG, and proximal aortic procedures. After exclusion of patients who did not undergo cardiopulmonary bypass, a known major cause of acute kidney injury (AKI), and those who died during the procedure, a total of 2,504 patients remained. These patients had a mean age of 68.4 years; 67.3% were men, according to the report.
The primary endpoint was the effect of timing between cardiac catheterization and surgery on the development of AKI. Postoperative AKI was defined by the consensus RIFLE criteria (risk, injury, failure, loss of function, and end-stage renal disease), using the maximal change in serum creatinine and the estimated glomerular filtration rate during the first 7 days after surgery, compared with baseline.
The overall incidence of AKI after surgery was 9.2% (230/2,504 patients). A breakdown by procedure showed that AKI occurred in 7.7% of isolated CABG patients, 12.2% of isolated valve patients, 9.5% of combined-procedure patients, and 9.5% of the proximal aorta surgery patients (Int. J. Cardiol. 2014;173:46-54).
As has been seen in previous studies, AKI was associated with patient-specific pre- and perioperative variables, including increased patient age, added comorbidities, longer cardiopulmonary bypass (CPB) times, higher rates of combined procedures, and the use of intra-aortic balloon pumps.
Unadjusted analysis of the total cohort showed AKI was significantly associated with contrast exposure within 1 day of surgery. However, in multivariable analysis, the time interval between catheterization and surgery was not an independent predictor of postoperative AKI for the total cohort. In subgroup analysis, only the combined valve and CABG group of patients showed an independent association of contrast exposure within 1 day before surgery and AKI in both the prematched (odds ratio, 2.69; P = .004) and the postmatched (OR, 3.68; P = .014) groups.
"Avoiding surgery within 1 day after contrast exposure should be recommended for patients undergoing valve surgery with concomitant CABG only. For other types of cardiac operations, delaying cardiac surgery after contrast exposure seems not to be justified," the researchers concluded.
The study was supported by the Fondazione Cesare Bartorelli. The authors reported having no disclosures.
mlesney@frontlinemedcom.com
Major finding: Only combined surgery plus contrast within 1 day before surgery was significantly associated with AKI in the prematched (OR, 2.69) and postmatched (OR, 3.68) groups.
Data source: A single-institute, retrospective study of 2,504 cardiac surgery patients.
Disclosures: The study was supported by the Fondazione Cesare Bartorelli. The authors reported having no financial disclosures.
Mentorship: An essential key to growth and success
In 2011, an article titled "Building a Successful Career: Advice From Leaders in Thoracic Surgery" was published in Thoracic Surgical Clinics of North America.1 In developing this paper, Dr. Sean Grondin states, "I have been fortunate to have had mentors who have guided me through my surgical training and early years of practice," and he continues, "I realize how much I have learned from and been influenced by the experience and guidance of other surgeons."
Based on his experiences, Dr. Grondin selected a group of 20 outstanding leaders in thoracic surgery, and asked them to each write a short summary of what they deemed to be key elements for developing a successful thoracic surgical career. The inspiration for the paper was an appreciation for mentorship and a desire to further disseminate the influences of the greats in our field; it is both revealing and remarkably appropriate that, in their responses, the majority of the recognized leaders discussed the importance of cultivating strong mentorship relationships, with the word "mentor" mentioned 34 times in the article. The importance of having strong mentors to guide oneself in career development is highlighted over and over again.
In his comments, Dr. Douglas Mathisen states, "Residency training usually introduces you to the most important mentors in your life. They will be the ones who nurture you, educate you, and point you in the right direction. They are likely to bring out the desire in you to become an academic surgeon and educator."
Advice for success from Dr. Valerie Rusch included eight key points, one of which focused on mentorship: "Peer review and senior advice are frequently helpful, and most senior academic physicians are delighted to provide this. Mentors may be surgeons but are also often found in other specialties or even nonclinical settings." And, from the mentors’ perspective, Dr. F.G. Pearson remarks, "In a residency training program, the opportunity to act as a meaningful mentor is a gift and a rewarding opportunity."
For a cardiothoracic surgeon’s individual success, it is clear that having (and utilizing) the right mentors can make a significant impact. Further, when one considers the success of a group of individuals, it is equally important that mentorship relationships are present and strong. In the summer of 2011, the Senior Tour (a group of retired cardiothoracic surgeons committed to education and service for their profession) was asked by the Joint Council on Thoracic Surgery Education to visit 10 thoracic surgical training programs that were considered above average according to a poll taken of thoracic surgical residents in the fall of 2010 by the Thoracic Surgery Residents Association.2
As they summarized their findings, they noted that the top programs possessed many common features leading to superior training in cardiac and general thoracic surgery. The authors specifically noted seven key factors, and, among them, was "a significant emphasis on mentorship, with the program director playing the primary roles and with voluntary relationships between residents and other faculty and with mentorship also including involvement in job finding."
If mentorship is so important to success in this career path, exactly what opportunities exist for trainees seeking mentorship? A Google search for ["mentor" AND "thoracic surgery"] yields a number of results, with ultimate direction leading to essentially two organizations: the AATS and Women in Thoracic Surgery (WTS).
From the AATS website, there is a list of names and contact information for mentors who have been selected or self-nominated for prospective candidates for thoracic surgery residency. These mentors are available to offer guidance, recommendations, and research opportunities, as well as clinical rotations in cardiothoracic surgery (http://aats.org/TSR/contactsMentors.cgi).
A number of opportunities for mentorship are also available through WTS. There is a formal mentoring program, aiming to pair women interested in cardiothoracic surgical careers with established WTS members. Dr. Shanda Blackmon, the Mentoring Chair for the WTS, reflects on the program, stating, "Many surgeons think that mentorship has to be a formal relationship at the same institution. What we have learned at WTS is that mentorship relationships can be equally effective even when they are not formal and the mentor is not from the same institution." She continues, reporting that, "in the survey we conducted through WTS, we discovered that mentorship was one of the most important factors to young surgeons aspiring to start a career in thoracic surgery." It is for this reason that WTS places such emphasis on helping trainees find and sustain mentorship opportunities.
WTS specifically offers scholarships to the Society of Thoracic Surgeons meeting, the Scanlan/WTS Traveling Mentorship, the Carolyn Reed Award, a list of mentors online, and resident mentors. (Further details of all of these programs can be found at www.wtsnet.org.) WTS provides a number of opportunities for female trainees to connect with female mentors. But is it absolutely necessary to find mentors who are "like you"?
Dr. Jennifer Lawton, president of WTS, acknowledges that everyone needs mentoring, but urges that "mentoring of women and minorities is particularly important in order to take advantage of the vast array of benefits to be gained by ethnic, racial, and gender diversity in the workplace."
She continues: "If we don’t see others successful in a chosen field, we do not perceive the goal attainable for ourselves. I have seen this many times in cardiothoracic surgery and women. Women who are not exposed to female mentors have expressed concern that practice in the field is not possible for women (especially those who aspire to have a family)." And, further, she explains that "we are typically more comfortable in mentoring situations with others like ourselves (similar challenges, needs, concerns). Surveys demonstrate that medical students often choose fields of interest because of mentors. For these reasons, it is important for individuals to find mentors who are similar to themselves."
Additional formal mentoring programs exist, and include the Brooks Scholarship through the Southern Thoracic Surgical Association (STSA), the Society of Thoracic Surgeons Looking to the Future scholarship program, the AATS Member for a Day program, and more.
Exactly what should one hope to get out of a mentorship relationship? The truth is, it really depends on the stage of career and the specific needs of the mentee. Mentorship needs change over time, and even well-established mentoring relationships will evolve and adjust along with participants in the interaction.
Sarah Schubert is a fourth-year medical student at Penn State, who has matched into the I-6 program at the University of Virginia and is the current recipient of the Scanlan/WTS scholarship. This award provides trainees with an opportunity to gain exposure to women cardiothoracic surgeon mentors by visiting a WTS member for an elective period. Ms. Schubert reports that she applied for the scholarship because she "wanted to work with and learn from a highly successful female cardiac surgeon in hopes of initiating and developing a professional relationship with a woman in the field."
As a medical student, she has fairly basic mentorship needs. She tells us that "right now, I need mentors who can serve as good role models – in the OR, with patients, with colleagues and trainees, and in their personal lives outside the hospital. As I prepare to enter residency, I anticipate that I will need to further develop my patient care skills and operative techniques, as well as skills to manage time and obligations. Knowing and working with people who have already figured out how to juggle those different obligations will be immensely helpful." Other medical students may be looking for advice on interviewing and ranking residency programs, and, like Ms. Schubert, may be interested in tips on how to get a great start in internship.
For individuals deep in the throes of cardiothoracic training, the needs are clearly different. Dr. Ryan Shelstad, a first-year fellow at the University of Colorado, states: "In my mind, the primary objective of clinical fellowship is acquisition of technical skills and clinical decision making in CT surgery. Thus, mentorship at this stage follows this objective." He continues, "CT fellowship is a relatively short time to achieve this goal as well as position oneself to successfully transition to a faculty position or private practice, which is the second objective—and area for mentoring."
Dr. Shelstad acknowledges and explains the transition in his mentorship needs, "I think earlier in training, mentorship focused on career choice, research, and transition to a fellowship. Much was focused on getting to the next step. While that is still important, now is the time to focus on the clinical aspects of CT surgery that I will use the rest of my career." And, certainly, the specific mentoring needs will continue to change with time.
Dr. Lawton, who holds the rank of professor at Washington University, St. Louis, states that, "after reaching full professor, surgeons need mentoring and sponsoring to reach goals such as dean positions, leadership positions, national organization officer positions, etc. We also all continually need mosaic mentoring that evolves over time (someone to mentor for research career aspects, managing work-life balance issues, family issues, teaching issues, financial issues, etc.). As we progress in our careers, we have the opportunity to also be mentored in other areas by younger generations."
Wait, did she just say that progressing surgeons can be mentored by younger generations? Yes, she did. And not only can seasoned surgeons be mentored by younger surgeons, they also stand to gain considerably from their own mentees. Dr. Lawton further states, "There is always reciprocal gain in a mentoring relationship for the mentor. There is no better joy than to see a mentee succeed and be fulfilled and happy in her chosen field. For some, this provides a rich legacy of trainees and mentees to provide continued enrichment, excitement, and a source of tremendous pride in later years." In the previously mentioned article by Grondin, Dr. Gail Darling states, "Academic careers are usually measured in papers published, grants awarded, invited lectures, and academic standing. Equally important are the students we teach and motivate toward careers in surgery, residents and fellows we have taught and mentored who will provide care to patients and who will go out and teach new generations of surgeons who will in turn provide care."
Once mentorship bonds have been established, it is critical to cultivate these relationships and to gain the most possible from these interactions. When asked how he has achieved these ends, Dr. Shelstad replies, "You and your mentors have the same goal: mutual success. Work hard for your mentors, and they will work hard for you. I would encourage explicit discussion of goals and expectations to ensure you and a potential mentor are on the same page. Mentorship is essential to success in cardiothoracic surgery."
Without a doubt, participation in mentorship relationships is a key element of success at every stage of the career and development of a cardiothoracic surgeon. Outstanding programs already exist to support modern trainees, and further efforts will certainly be met with great enthusiasm. Also apparent is the need for ongoing, career-long mentorship – and the paucity of formal programs to support it. This need may be a great opportunity for the establishment of future programming.
On a personal note, when reflecting on mentorship, I think of a statement made by Benjamin Franklin: "Tell me and I forget, teach me and I may remember, involve me and I learn." To those incredible individuals who have taken the time, effort, and interest to truly involve me, I have immense gratitude. I will aim for the rest of my career to make it worth your while, with hopes to bring you pride and inspire others as you have inspired me. To all who have taken time to truly involve any student, resident, or junior colleague – you have motivated us and given strength to the ongoing legacy of our amazing field.
Citations
1. Grondin SC. Building a Successful Career: Advice From Leaders in Thoracic Surgery. Thorac. Surg. Clin. 2011;21(3):395-415.
2. J. Thorac. Cardiovasc. Surg. 2014;147(1):15-17.
In 2011, an article titled "Building a Successful Career: Advice From Leaders in Thoracic Surgery" was published in Thoracic Surgical Clinics of North America.1 In developing this paper, Dr. Sean Grondin states, "I have been fortunate to have had mentors who have guided me through my surgical training and early years of practice," and he continues, "I realize how much I have learned from and been influenced by the experience and guidance of other surgeons."
Based on his experiences, Dr. Grondin selected a group of 20 outstanding leaders in thoracic surgery, and asked them to each write a short summary of what they deemed to be key elements for developing a successful thoracic surgical career. The inspiration for the paper was an appreciation for mentorship and a desire to further disseminate the influences of the greats in our field; it is both revealing and remarkably appropriate that, in their responses, the majority of the recognized leaders discussed the importance of cultivating strong mentorship relationships, with the word "mentor" mentioned 34 times in the article. The importance of having strong mentors to guide oneself in career development is highlighted over and over again.
In his comments, Dr. Douglas Mathisen states, "Residency training usually introduces you to the most important mentors in your life. They will be the ones who nurture you, educate you, and point you in the right direction. They are likely to bring out the desire in you to become an academic surgeon and educator."
Advice for success from Dr. Valerie Rusch included eight key points, one of which focused on mentorship: "Peer review and senior advice are frequently helpful, and most senior academic physicians are delighted to provide this. Mentors may be surgeons but are also often found in other specialties or even nonclinical settings." And, from the mentors’ perspective, Dr. F.G. Pearson remarks, "In a residency training program, the opportunity to act as a meaningful mentor is a gift and a rewarding opportunity."
For a cardiothoracic surgeon’s individual success, it is clear that having (and utilizing) the right mentors can make a significant impact. Further, when one considers the success of a group of individuals, it is equally important that mentorship relationships are present and strong. In the summer of 2011, the Senior Tour (a group of retired cardiothoracic surgeons committed to education and service for their profession) was asked by the Joint Council on Thoracic Surgery Education to visit 10 thoracic surgical training programs that were considered above average according to a poll taken of thoracic surgical residents in the fall of 2010 by the Thoracic Surgery Residents Association.2
As they summarized their findings, they noted that the top programs possessed many common features leading to superior training in cardiac and general thoracic surgery. The authors specifically noted seven key factors, and, among them, was "a significant emphasis on mentorship, with the program director playing the primary roles and with voluntary relationships between residents and other faculty and with mentorship also including involvement in job finding."
If mentorship is so important to success in this career path, exactly what opportunities exist for trainees seeking mentorship? A Google search for ["mentor" AND "thoracic surgery"] yields a number of results, with ultimate direction leading to essentially two organizations: the AATS and Women in Thoracic Surgery (WTS).
From the AATS website, there is a list of names and contact information for mentors who have been selected or self-nominated for prospective candidates for thoracic surgery residency. These mentors are available to offer guidance, recommendations, and research opportunities, as well as clinical rotations in cardiothoracic surgery (http://aats.org/TSR/contactsMentors.cgi).
A number of opportunities for mentorship are also available through WTS. There is a formal mentoring program, aiming to pair women interested in cardiothoracic surgical careers with established WTS members. Dr. Shanda Blackmon, the Mentoring Chair for the WTS, reflects on the program, stating, "Many surgeons think that mentorship has to be a formal relationship at the same institution. What we have learned at WTS is that mentorship relationships can be equally effective even when they are not formal and the mentor is not from the same institution." She continues, reporting that, "in the survey we conducted through WTS, we discovered that mentorship was one of the most important factors to young surgeons aspiring to start a career in thoracic surgery." It is for this reason that WTS places such emphasis on helping trainees find and sustain mentorship opportunities.
WTS specifically offers scholarships to the Society of Thoracic Surgeons meeting, the Scanlan/WTS Traveling Mentorship, the Carolyn Reed Award, a list of mentors online, and resident mentors. (Further details of all of these programs can be found at www.wtsnet.org.) WTS provides a number of opportunities for female trainees to connect with female mentors. But is it absolutely necessary to find mentors who are "like you"?
Dr. Jennifer Lawton, president of WTS, acknowledges that everyone needs mentoring, but urges that "mentoring of women and minorities is particularly important in order to take advantage of the vast array of benefits to be gained by ethnic, racial, and gender diversity in the workplace."
She continues: "If we don’t see others successful in a chosen field, we do not perceive the goal attainable for ourselves. I have seen this many times in cardiothoracic surgery and women. Women who are not exposed to female mentors have expressed concern that practice in the field is not possible for women (especially those who aspire to have a family)." And, further, she explains that "we are typically more comfortable in mentoring situations with others like ourselves (similar challenges, needs, concerns). Surveys demonstrate that medical students often choose fields of interest because of mentors. For these reasons, it is important for individuals to find mentors who are similar to themselves."
Additional formal mentoring programs exist, and include the Brooks Scholarship through the Southern Thoracic Surgical Association (STSA), the Society of Thoracic Surgeons Looking to the Future scholarship program, the AATS Member for a Day program, and more.
Exactly what should one hope to get out of a mentorship relationship? The truth is, it really depends on the stage of career and the specific needs of the mentee. Mentorship needs change over time, and even well-established mentoring relationships will evolve and adjust along with participants in the interaction.
Sarah Schubert is a fourth-year medical student at Penn State, who has matched into the I-6 program at the University of Virginia and is the current recipient of the Scanlan/WTS scholarship. This award provides trainees with an opportunity to gain exposure to women cardiothoracic surgeon mentors by visiting a WTS member for an elective period. Ms. Schubert reports that she applied for the scholarship because she "wanted to work with and learn from a highly successful female cardiac surgeon in hopes of initiating and developing a professional relationship with a woman in the field."
As a medical student, she has fairly basic mentorship needs. She tells us that "right now, I need mentors who can serve as good role models – in the OR, with patients, with colleagues and trainees, and in their personal lives outside the hospital. As I prepare to enter residency, I anticipate that I will need to further develop my patient care skills and operative techniques, as well as skills to manage time and obligations. Knowing and working with people who have already figured out how to juggle those different obligations will be immensely helpful." Other medical students may be looking for advice on interviewing and ranking residency programs, and, like Ms. Schubert, may be interested in tips on how to get a great start in internship.
For individuals deep in the throes of cardiothoracic training, the needs are clearly different. Dr. Ryan Shelstad, a first-year fellow at the University of Colorado, states: "In my mind, the primary objective of clinical fellowship is acquisition of technical skills and clinical decision making in CT surgery. Thus, mentorship at this stage follows this objective." He continues, "CT fellowship is a relatively short time to achieve this goal as well as position oneself to successfully transition to a faculty position or private practice, which is the second objective—and area for mentoring."
Dr. Shelstad acknowledges and explains the transition in his mentorship needs, "I think earlier in training, mentorship focused on career choice, research, and transition to a fellowship. Much was focused on getting to the next step. While that is still important, now is the time to focus on the clinical aspects of CT surgery that I will use the rest of my career." And, certainly, the specific mentoring needs will continue to change with time.
Dr. Lawton, who holds the rank of professor at Washington University, St. Louis, states that, "after reaching full professor, surgeons need mentoring and sponsoring to reach goals such as dean positions, leadership positions, national organization officer positions, etc. We also all continually need mosaic mentoring that evolves over time (someone to mentor for research career aspects, managing work-life balance issues, family issues, teaching issues, financial issues, etc.). As we progress in our careers, we have the opportunity to also be mentored in other areas by younger generations."
Wait, did she just say that progressing surgeons can be mentored by younger generations? Yes, she did. And not only can seasoned surgeons be mentored by younger surgeons, they also stand to gain considerably from their own mentees. Dr. Lawton further states, "There is always reciprocal gain in a mentoring relationship for the mentor. There is no better joy than to see a mentee succeed and be fulfilled and happy in her chosen field. For some, this provides a rich legacy of trainees and mentees to provide continued enrichment, excitement, and a source of tremendous pride in later years." In the previously mentioned article by Grondin, Dr. Gail Darling states, "Academic careers are usually measured in papers published, grants awarded, invited lectures, and academic standing. Equally important are the students we teach and motivate toward careers in surgery, residents and fellows we have taught and mentored who will provide care to patients and who will go out and teach new generations of surgeons who will in turn provide care."
Once mentorship bonds have been established, it is critical to cultivate these relationships and to gain the most possible from these interactions. When asked how he has achieved these ends, Dr. Shelstad replies, "You and your mentors have the same goal: mutual success. Work hard for your mentors, and they will work hard for you. I would encourage explicit discussion of goals and expectations to ensure you and a potential mentor are on the same page. Mentorship is essential to success in cardiothoracic surgery."
Without a doubt, participation in mentorship relationships is a key element of success at every stage of the career and development of a cardiothoracic surgeon. Outstanding programs already exist to support modern trainees, and further efforts will certainly be met with great enthusiasm. Also apparent is the need for ongoing, career-long mentorship – and the paucity of formal programs to support it. This need may be a great opportunity for the establishment of future programming.
On a personal note, when reflecting on mentorship, I think of a statement made by Benjamin Franklin: "Tell me and I forget, teach me and I may remember, involve me and I learn." To those incredible individuals who have taken the time, effort, and interest to truly involve me, I have immense gratitude. I will aim for the rest of my career to make it worth your while, with hopes to bring you pride and inspire others as you have inspired me. To all who have taken time to truly involve any student, resident, or junior colleague – you have motivated us and given strength to the ongoing legacy of our amazing field.
Citations
1. Grondin SC. Building a Successful Career: Advice From Leaders in Thoracic Surgery. Thorac. Surg. Clin. 2011;21(3):395-415.
2. J. Thorac. Cardiovasc. Surg. 2014;147(1):15-17.
In 2011, an article titled "Building a Successful Career: Advice From Leaders in Thoracic Surgery" was published in Thoracic Surgical Clinics of North America.1 In developing this paper, Dr. Sean Grondin states, "I have been fortunate to have had mentors who have guided me through my surgical training and early years of practice," and he continues, "I realize how much I have learned from and been influenced by the experience and guidance of other surgeons."
Based on his experiences, Dr. Grondin selected a group of 20 outstanding leaders in thoracic surgery, and asked them to each write a short summary of what they deemed to be key elements for developing a successful thoracic surgical career. The inspiration for the paper was an appreciation for mentorship and a desire to further disseminate the influences of the greats in our field; it is both revealing and remarkably appropriate that, in their responses, the majority of the recognized leaders discussed the importance of cultivating strong mentorship relationships, with the word "mentor" mentioned 34 times in the article. The importance of having strong mentors to guide oneself in career development is highlighted over and over again.
In his comments, Dr. Douglas Mathisen states, "Residency training usually introduces you to the most important mentors in your life. They will be the ones who nurture you, educate you, and point you in the right direction. They are likely to bring out the desire in you to become an academic surgeon and educator."
Advice for success from Dr. Valerie Rusch included eight key points, one of which focused on mentorship: "Peer review and senior advice are frequently helpful, and most senior academic physicians are delighted to provide this. Mentors may be surgeons but are also often found in other specialties or even nonclinical settings." And, from the mentors’ perspective, Dr. F.G. Pearson remarks, "In a residency training program, the opportunity to act as a meaningful mentor is a gift and a rewarding opportunity."
For a cardiothoracic surgeon’s individual success, it is clear that having (and utilizing) the right mentors can make a significant impact. Further, when one considers the success of a group of individuals, it is equally important that mentorship relationships are present and strong. In the summer of 2011, the Senior Tour (a group of retired cardiothoracic surgeons committed to education and service for their profession) was asked by the Joint Council on Thoracic Surgery Education to visit 10 thoracic surgical training programs that were considered above average according to a poll taken of thoracic surgical residents in the fall of 2010 by the Thoracic Surgery Residents Association.2
As they summarized their findings, they noted that the top programs possessed many common features leading to superior training in cardiac and general thoracic surgery. The authors specifically noted seven key factors, and, among them, was "a significant emphasis on mentorship, with the program director playing the primary roles and with voluntary relationships between residents and other faculty and with mentorship also including involvement in job finding."
If mentorship is so important to success in this career path, exactly what opportunities exist for trainees seeking mentorship? A Google search for ["mentor" AND "thoracic surgery"] yields a number of results, with ultimate direction leading to essentially two organizations: the AATS and Women in Thoracic Surgery (WTS).
From the AATS website, there is a list of names and contact information for mentors who have been selected or self-nominated for prospective candidates for thoracic surgery residency. These mentors are available to offer guidance, recommendations, and research opportunities, as well as clinical rotations in cardiothoracic surgery (http://aats.org/TSR/contactsMentors.cgi).
A number of opportunities for mentorship are also available through WTS. There is a formal mentoring program, aiming to pair women interested in cardiothoracic surgical careers with established WTS members. Dr. Shanda Blackmon, the Mentoring Chair for the WTS, reflects on the program, stating, "Many surgeons think that mentorship has to be a formal relationship at the same institution. What we have learned at WTS is that mentorship relationships can be equally effective even when they are not formal and the mentor is not from the same institution." She continues, reporting that, "in the survey we conducted through WTS, we discovered that mentorship was one of the most important factors to young surgeons aspiring to start a career in thoracic surgery." It is for this reason that WTS places such emphasis on helping trainees find and sustain mentorship opportunities.
WTS specifically offers scholarships to the Society of Thoracic Surgeons meeting, the Scanlan/WTS Traveling Mentorship, the Carolyn Reed Award, a list of mentors online, and resident mentors. (Further details of all of these programs can be found at www.wtsnet.org.) WTS provides a number of opportunities for female trainees to connect with female mentors. But is it absolutely necessary to find mentors who are "like you"?
Dr. Jennifer Lawton, president of WTS, acknowledges that everyone needs mentoring, but urges that "mentoring of women and minorities is particularly important in order to take advantage of the vast array of benefits to be gained by ethnic, racial, and gender diversity in the workplace."
She continues: "If we don’t see others successful in a chosen field, we do not perceive the goal attainable for ourselves. I have seen this many times in cardiothoracic surgery and women. Women who are not exposed to female mentors have expressed concern that practice in the field is not possible for women (especially those who aspire to have a family)." And, further, she explains that "we are typically more comfortable in mentoring situations with others like ourselves (similar challenges, needs, concerns). Surveys demonstrate that medical students often choose fields of interest because of mentors. For these reasons, it is important for individuals to find mentors who are similar to themselves."
Additional formal mentoring programs exist, and include the Brooks Scholarship through the Southern Thoracic Surgical Association (STSA), the Society of Thoracic Surgeons Looking to the Future scholarship program, the AATS Member for a Day program, and more.
Exactly what should one hope to get out of a mentorship relationship? The truth is, it really depends on the stage of career and the specific needs of the mentee. Mentorship needs change over time, and even well-established mentoring relationships will evolve and adjust along with participants in the interaction.
Sarah Schubert is a fourth-year medical student at Penn State, who has matched into the I-6 program at the University of Virginia and is the current recipient of the Scanlan/WTS scholarship. This award provides trainees with an opportunity to gain exposure to women cardiothoracic surgeon mentors by visiting a WTS member for an elective period. Ms. Schubert reports that she applied for the scholarship because she "wanted to work with and learn from a highly successful female cardiac surgeon in hopes of initiating and developing a professional relationship with a woman in the field."
As a medical student, she has fairly basic mentorship needs. She tells us that "right now, I need mentors who can serve as good role models – in the OR, with patients, with colleagues and trainees, and in their personal lives outside the hospital. As I prepare to enter residency, I anticipate that I will need to further develop my patient care skills and operative techniques, as well as skills to manage time and obligations. Knowing and working with people who have already figured out how to juggle those different obligations will be immensely helpful." Other medical students may be looking for advice on interviewing and ranking residency programs, and, like Ms. Schubert, may be interested in tips on how to get a great start in internship.
For individuals deep in the throes of cardiothoracic training, the needs are clearly different. Dr. Ryan Shelstad, a first-year fellow at the University of Colorado, states: "In my mind, the primary objective of clinical fellowship is acquisition of technical skills and clinical decision making in CT surgery. Thus, mentorship at this stage follows this objective." He continues, "CT fellowship is a relatively short time to achieve this goal as well as position oneself to successfully transition to a faculty position or private practice, which is the second objective—and area for mentoring."
Dr. Shelstad acknowledges and explains the transition in his mentorship needs, "I think earlier in training, mentorship focused on career choice, research, and transition to a fellowship. Much was focused on getting to the next step. While that is still important, now is the time to focus on the clinical aspects of CT surgery that I will use the rest of my career." And, certainly, the specific mentoring needs will continue to change with time.
Dr. Lawton, who holds the rank of professor at Washington University, St. Louis, states that, "after reaching full professor, surgeons need mentoring and sponsoring to reach goals such as dean positions, leadership positions, national organization officer positions, etc. We also all continually need mosaic mentoring that evolves over time (someone to mentor for research career aspects, managing work-life balance issues, family issues, teaching issues, financial issues, etc.). As we progress in our careers, we have the opportunity to also be mentored in other areas by younger generations."
Wait, did she just say that progressing surgeons can be mentored by younger generations? Yes, she did. And not only can seasoned surgeons be mentored by younger surgeons, they also stand to gain considerably from their own mentees. Dr. Lawton further states, "There is always reciprocal gain in a mentoring relationship for the mentor. There is no better joy than to see a mentee succeed and be fulfilled and happy in her chosen field. For some, this provides a rich legacy of trainees and mentees to provide continued enrichment, excitement, and a source of tremendous pride in later years." In the previously mentioned article by Grondin, Dr. Gail Darling states, "Academic careers are usually measured in papers published, grants awarded, invited lectures, and academic standing. Equally important are the students we teach and motivate toward careers in surgery, residents and fellows we have taught and mentored who will provide care to patients and who will go out and teach new generations of surgeons who will in turn provide care."
Once mentorship bonds have been established, it is critical to cultivate these relationships and to gain the most possible from these interactions. When asked how he has achieved these ends, Dr. Shelstad replies, "You and your mentors have the same goal: mutual success. Work hard for your mentors, and they will work hard for you. I would encourage explicit discussion of goals and expectations to ensure you and a potential mentor are on the same page. Mentorship is essential to success in cardiothoracic surgery."
Without a doubt, participation in mentorship relationships is a key element of success at every stage of the career and development of a cardiothoracic surgeon. Outstanding programs already exist to support modern trainees, and further efforts will certainly be met with great enthusiasm. Also apparent is the need for ongoing, career-long mentorship – and the paucity of formal programs to support it. This need may be a great opportunity for the establishment of future programming.
On a personal note, when reflecting on mentorship, I think of a statement made by Benjamin Franklin: "Tell me and I forget, teach me and I may remember, involve me and I learn." To those incredible individuals who have taken the time, effort, and interest to truly involve me, I have immense gratitude. I will aim for the rest of my career to make it worth your while, with hopes to bring you pride and inspire others as you have inspired me. To all who have taken time to truly involve any student, resident, or junior colleague – you have motivated us and given strength to the ongoing legacy of our amazing field.
Citations
1. Grondin SC. Building a Successful Career: Advice From Leaders in Thoracic Surgery. Thorac. Surg. Clin. 2011;21(3):395-415.
2. J. Thorac. Cardiovasc. Surg. 2014;147(1):15-17.
Open TAAA repair in octogenarians: Exhibit caution
NEW YORK – Outcomes of thoracoabdominal aortic aneurysm (TAAA) repair in octogenarians vary considerably with the extent of repair. Those who undergo Extent II TAAA repair have significantly higher risks of morbidity and mortality, while Extent I, III, and IV repairs can be performed with relatively good outcomes, according to Dr. Muhammad Aftab, who presented the findings at the 2014 AATSAortic Symposium
"Extensive TAAA repair should be performed with caution in octogenarians," says Dr. Aftab, a Fellow in CT surgery at the Baylor College of Medicine–Texas Heart Institute, Houston. He recommends that a thorough preoperative discussion to assess the risks and benefits with the patient and his family is necessary before proceeding with surgery.
In this retrospective review of patients seen between January 2005 and September 2013, octogenarians with TAAAs (n = 88) were compared with a younger cohort (n = 1,179 patients, aged 70 years). Dr. Aftab found that octogenarians were threefold more likely to present with aneurysm rupture (13.6% vs. 4.6%; P less than .001) but less likely to present with aortic dissections (12.5% vs. 43.9%; P less than .001) than did the younger patients.
The use of adjunctive interventions, such as left heart bypass, cerebrospinal fluid drainage, cold renal perfusion, and visceral perfusion differed significantly among the octogenarians based on the extent of repair and clinical condition (all P less than .001). Because the octogenarians had a greater atherosclerotic burden and higher incidence of renal and mesenteric occlusive disease, they were also more likely to require renal/visceral endarterectomy, stenting, or both (57.9% vs. 33.6%; P less than.001).
Octogenarians had higher rates of operative mortality (26.1% vs. 6.9%), in-hospital deaths (25% vs. 6.4%), 30-day deaths (13.6% vs. 4.8%), and adverse outcomes (36.4% vs. 15.7%; P less than .001) than did the younger group, all significant differences.
The outcomes included significantly higher rates of permanent renal failure, cardiac complications, and pulmonary complications. The octogenarians had longer recovery times, as suggested by longer postoperative ICU and hospital stays. Poor outcomes differed according to the extent of surgery, and seemed to be exacerbated for those who underwent repair of Extent II aneurysms (according to the Crawford Classification, these involve the subclavian artery and extend to the bifurcation of the aorta in the pelvis). For instance, the Extent II group had the highest risk of operative mortality (61.5%) vs. Extent I (31.6%), III (21.4%), and IV (10.7%), a significant difference. The Extent II group also had much higher rates of in-hospital and 30-day death rates. The most common causes of deaths for the Extent II octogenarians were multisystem organ failure and cardiac problems.
Adverse outcomes were also significantly much higher for the Extent II group (76.9%) than for the other groups (42.1%, 28.6%, and 21.4%). Similar patterns were found for permanent paraplegia, renal failure requiring permanent dialysis, stroke, and days spent in the ICU. Almost 85% of those who required Extent II repair needed renal/visceral endarterectomy, stenting, or both as a part of the surgical procedure.
Extent II TAAA repair was an independent predictor of perioperative mortality by multivariate analysis, conferring an 11-fold increased risk of death. Aneurysm rupture and dissection were also identified as predictors of perioperative mortality while only Extent II TAAA and dissection were independent predictors of adverse outcomes.
While these problems were exacerbated in those with Extent II repairs, Extent I, III, and IV TAAA repairs may be performed with relatively low risk, according to Dr. Aftab.
Dr. Aftab had no disclosures.
NEW YORK – Outcomes of thoracoabdominal aortic aneurysm (TAAA) repair in octogenarians vary considerably with the extent of repair. Those who undergo Extent II TAAA repair have significantly higher risks of morbidity and mortality, while Extent I, III, and IV repairs can be performed with relatively good outcomes, according to Dr. Muhammad Aftab, who presented the findings at the 2014 AATSAortic Symposium
"Extensive TAAA repair should be performed with caution in octogenarians," says Dr. Aftab, a Fellow in CT surgery at the Baylor College of Medicine–Texas Heart Institute, Houston. He recommends that a thorough preoperative discussion to assess the risks and benefits with the patient and his family is necessary before proceeding with surgery.
In this retrospective review of patients seen between January 2005 and September 2013, octogenarians with TAAAs (n = 88) were compared with a younger cohort (n = 1,179 patients, aged 70 years). Dr. Aftab found that octogenarians were threefold more likely to present with aneurysm rupture (13.6% vs. 4.6%; P less than .001) but less likely to present with aortic dissections (12.5% vs. 43.9%; P less than .001) than did the younger patients.
The use of adjunctive interventions, such as left heart bypass, cerebrospinal fluid drainage, cold renal perfusion, and visceral perfusion differed significantly among the octogenarians based on the extent of repair and clinical condition (all P less than .001). Because the octogenarians had a greater atherosclerotic burden and higher incidence of renal and mesenteric occlusive disease, they were also more likely to require renal/visceral endarterectomy, stenting, or both (57.9% vs. 33.6%; P less than.001).
Octogenarians had higher rates of operative mortality (26.1% vs. 6.9%), in-hospital deaths (25% vs. 6.4%), 30-day deaths (13.6% vs. 4.8%), and adverse outcomes (36.4% vs. 15.7%; P less than .001) than did the younger group, all significant differences.
The outcomes included significantly higher rates of permanent renal failure, cardiac complications, and pulmonary complications. The octogenarians had longer recovery times, as suggested by longer postoperative ICU and hospital stays. Poor outcomes differed according to the extent of surgery, and seemed to be exacerbated for those who underwent repair of Extent II aneurysms (according to the Crawford Classification, these involve the subclavian artery and extend to the bifurcation of the aorta in the pelvis). For instance, the Extent II group had the highest risk of operative mortality (61.5%) vs. Extent I (31.6%), III (21.4%), and IV (10.7%), a significant difference. The Extent II group also had much higher rates of in-hospital and 30-day death rates. The most common causes of deaths for the Extent II octogenarians were multisystem organ failure and cardiac problems.
Adverse outcomes were also significantly much higher for the Extent II group (76.9%) than for the other groups (42.1%, 28.6%, and 21.4%). Similar patterns were found for permanent paraplegia, renal failure requiring permanent dialysis, stroke, and days spent in the ICU. Almost 85% of those who required Extent II repair needed renal/visceral endarterectomy, stenting, or both as a part of the surgical procedure.
Extent II TAAA repair was an independent predictor of perioperative mortality by multivariate analysis, conferring an 11-fold increased risk of death. Aneurysm rupture and dissection were also identified as predictors of perioperative mortality while only Extent II TAAA and dissection were independent predictors of adverse outcomes.
While these problems were exacerbated in those with Extent II repairs, Extent I, III, and IV TAAA repairs may be performed with relatively low risk, according to Dr. Aftab.
Dr. Aftab had no disclosures.
NEW YORK – Outcomes of thoracoabdominal aortic aneurysm (TAAA) repair in octogenarians vary considerably with the extent of repair. Those who undergo Extent II TAAA repair have significantly higher risks of morbidity and mortality, while Extent I, III, and IV repairs can be performed with relatively good outcomes, according to Dr. Muhammad Aftab, who presented the findings at the 2014 AATSAortic Symposium
"Extensive TAAA repair should be performed with caution in octogenarians," says Dr. Aftab, a Fellow in CT surgery at the Baylor College of Medicine–Texas Heart Institute, Houston. He recommends that a thorough preoperative discussion to assess the risks and benefits with the patient and his family is necessary before proceeding with surgery.
In this retrospective review of patients seen between January 2005 and September 2013, octogenarians with TAAAs (n = 88) were compared with a younger cohort (n = 1,179 patients, aged 70 years). Dr. Aftab found that octogenarians were threefold more likely to present with aneurysm rupture (13.6% vs. 4.6%; P less than .001) but less likely to present with aortic dissections (12.5% vs. 43.9%; P less than .001) than did the younger patients.
The use of adjunctive interventions, such as left heart bypass, cerebrospinal fluid drainage, cold renal perfusion, and visceral perfusion differed significantly among the octogenarians based on the extent of repair and clinical condition (all P less than .001). Because the octogenarians had a greater atherosclerotic burden and higher incidence of renal and mesenteric occlusive disease, they were also more likely to require renal/visceral endarterectomy, stenting, or both (57.9% vs. 33.6%; P less than.001).
Octogenarians had higher rates of operative mortality (26.1% vs. 6.9%), in-hospital deaths (25% vs. 6.4%), 30-day deaths (13.6% vs. 4.8%), and adverse outcomes (36.4% vs. 15.7%; P less than .001) than did the younger group, all significant differences.
The outcomes included significantly higher rates of permanent renal failure, cardiac complications, and pulmonary complications. The octogenarians had longer recovery times, as suggested by longer postoperative ICU and hospital stays. Poor outcomes differed according to the extent of surgery, and seemed to be exacerbated for those who underwent repair of Extent II aneurysms (according to the Crawford Classification, these involve the subclavian artery and extend to the bifurcation of the aorta in the pelvis). For instance, the Extent II group had the highest risk of operative mortality (61.5%) vs. Extent I (31.6%), III (21.4%), and IV (10.7%), a significant difference. The Extent II group also had much higher rates of in-hospital and 30-day death rates. The most common causes of deaths for the Extent II octogenarians were multisystem organ failure and cardiac problems.
Adverse outcomes were also significantly much higher for the Extent II group (76.9%) than for the other groups (42.1%, 28.6%, and 21.4%). Similar patterns were found for permanent paraplegia, renal failure requiring permanent dialysis, stroke, and days spent in the ICU. Almost 85% of those who required Extent II repair needed renal/visceral endarterectomy, stenting, or both as a part of the surgical procedure.
Extent II TAAA repair was an independent predictor of perioperative mortality by multivariate analysis, conferring an 11-fold increased risk of death. Aneurysm rupture and dissection were also identified as predictors of perioperative mortality while only Extent II TAAA and dissection were independent predictors of adverse outcomes.
While these problems were exacerbated in those with Extent II repairs, Extent I, III, and IV TAAA repairs may be performed with relatively low risk, according to Dr. Aftab.
Dr. Aftab had no disclosures.
Key clinical point: Octogenarians with TAAAs present more challenges than younger individuals and their outcomes vary greatly according to the type of aneurysm repair.
Major finding: A study that compared octogenarians with thoracoabdominal aortic aneurysms (TAAAs) to a younger cohort found that octogenarians were more at risk for aneurysm rupture, were more likely to need visceral-branch endarterectomy/stenting, had more adverse postoperative outcomes, and higher rates of operative mortality and longer postoperative ICU and hospital stays. While these problems were exacerbated in those with Extent II repairs, Extent I, III, and IV TAAA repairs can be performed with relatively low risk. Younger patients were more likely than octogenarians to present with aortic dissections.
Data source: Retrospective review.
Disclosures: Dr. Aftab had no relevant disclosures.
Attendings in 'simultaneous' ORs
TORONTO – In academic medical centers, attending cardiothoracic surgeons often perform simultaneous procedures in different operating rooms as a means of increasing training opportunities for surgical fellows and to decrease hospital costs. However, the practice of running simultaneous operating rooms did not appear to affect perioperative timing or negatively affect patient outcomes, according to the results of a single institution review by Dr. Kenan W. Yount.
He and his colleagues at the University of Virginia, Charlottesville, wanted to examine their own data in guiding hospital policy as several major centers have recently proposed implementing a 1:1 ratio of attending surgeon-to-operating room.
In his presentation, Dr. Yount discussed the results of their review study, which categorized 1,377 cardiac and 1,682 general thoracic operations performed from July 2011 to July 2013 by attending, case type, and whether the attending was simultaneously supervising two surgeries. "Our institution adheres to a strict policy of attending surgeon oversight of and involvement in the critical and key portions of all operations," said Dr. Yount.
They compared operative duration, starting and closing times, postoperative complications, and 30-day mortality in each category. They also compared rates postoperative complications, hospital length of stay, and operative mortality in each category.
Interestingly, timing effects varied between the two overall types of surgery. Running two rooms had no effect on room start times, but thoracic rooms finished 16 minutes later than scheduled. Across six surgeons and 15 operation types, however, there were no differences in operative times.
"Furthermore, running two rooms was not associated with any differences in operative duration, morbidity, or mortality in our multivariate regression analyses, and there were no statistically significant differences in observed outcomes in any category," Dr. Yount said.
"In academic cardiothoracic surgical centers that rely on surgical support from fellowship training, the practice of running simultaneous operating rooms can be efficient and does not appear to negatively impact patient outcomes," said Dr. Yount. "In addition, the practice did not significantly increase operative duration or dramatically [affect] operating room starting or closing times," he concluded.
In discussing the implications of these results, he said, "Obviously, there are caveats: Attendings must be intimately involved in operations and scrubbed for every key and critical portion of the operation; also operations being scheduled in separate rooms must be done so with reasonable foresight."
As long as institutions are following these practices, he concluded, "It would appear that lens of current policy efforts is too narrow by focusing on perception. The debate should be refocused by challenging training programs to strengthen attending involvement and ensure the requisite competence of their trainees."
Dr. Yount reported no relevant disclosures.
TORONTO – In academic medical centers, attending cardiothoracic surgeons often perform simultaneous procedures in different operating rooms as a means of increasing training opportunities for surgical fellows and to decrease hospital costs. However, the practice of running simultaneous operating rooms did not appear to affect perioperative timing or negatively affect patient outcomes, according to the results of a single institution review by Dr. Kenan W. Yount.
He and his colleagues at the University of Virginia, Charlottesville, wanted to examine their own data in guiding hospital policy as several major centers have recently proposed implementing a 1:1 ratio of attending surgeon-to-operating room.
In his presentation, Dr. Yount discussed the results of their review study, which categorized 1,377 cardiac and 1,682 general thoracic operations performed from July 2011 to July 2013 by attending, case type, and whether the attending was simultaneously supervising two surgeries. "Our institution adheres to a strict policy of attending surgeon oversight of and involvement in the critical and key portions of all operations," said Dr. Yount.
They compared operative duration, starting and closing times, postoperative complications, and 30-day mortality in each category. They also compared rates postoperative complications, hospital length of stay, and operative mortality in each category.
Interestingly, timing effects varied between the two overall types of surgery. Running two rooms had no effect on room start times, but thoracic rooms finished 16 minutes later than scheduled. Across six surgeons and 15 operation types, however, there were no differences in operative times.
"Furthermore, running two rooms was not associated with any differences in operative duration, morbidity, or mortality in our multivariate regression analyses, and there were no statistically significant differences in observed outcomes in any category," Dr. Yount said.
"In academic cardiothoracic surgical centers that rely on surgical support from fellowship training, the practice of running simultaneous operating rooms can be efficient and does not appear to negatively impact patient outcomes," said Dr. Yount. "In addition, the practice did not significantly increase operative duration or dramatically [affect] operating room starting or closing times," he concluded.
In discussing the implications of these results, he said, "Obviously, there are caveats: Attendings must be intimately involved in operations and scrubbed for every key and critical portion of the operation; also operations being scheduled in separate rooms must be done so with reasonable foresight."
As long as institutions are following these practices, he concluded, "It would appear that lens of current policy efforts is too narrow by focusing on perception. The debate should be refocused by challenging training programs to strengthen attending involvement and ensure the requisite competence of their trainees."
Dr. Yount reported no relevant disclosures.
TORONTO – In academic medical centers, attending cardiothoracic surgeons often perform simultaneous procedures in different operating rooms as a means of increasing training opportunities for surgical fellows and to decrease hospital costs. However, the practice of running simultaneous operating rooms did not appear to affect perioperative timing or negatively affect patient outcomes, according to the results of a single institution review by Dr. Kenan W. Yount.
He and his colleagues at the University of Virginia, Charlottesville, wanted to examine their own data in guiding hospital policy as several major centers have recently proposed implementing a 1:1 ratio of attending surgeon-to-operating room.
In his presentation, Dr. Yount discussed the results of their review study, which categorized 1,377 cardiac and 1,682 general thoracic operations performed from July 2011 to July 2013 by attending, case type, and whether the attending was simultaneously supervising two surgeries. "Our institution adheres to a strict policy of attending surgeon oversight of and involvement in the critical and key portions of all operations," said Dr. Yount.
They compared operative duration, starting and closing times, postoperative complications, and 30-day mortality in each category. They also compared rates postoperative complications, hospital length of stay, and operative mortality in each category.
Interestingly, timing effects varied between the two overall types of surgery. Running two rooms had no effect on room start times, but thoracic rooms finished 16 minutes later than scheduled. Across six surgeons and 15 operation types, however, there were no differences in operative times.
"Furthermore, running two rooms was not associated with any differences in operative duration, morbidity, or mortality in our multivariate regression analyses, and there were no statistically significant differences in observed outcomes in any category," Dr. Yount said.
"In academic cardiothoracic surgical centers that rely on surgical support from fellowship training, the practice of running simultaneous operating rooms can be efficient and does not appear to negatively impact patient outcomes," said Dr. Yount. "In addition, the practice did not significantly increase operative duration or dramatically [affect] operating room starting or closing times," he concluded.
In discussing the implications of these results, he said, "Obviously, there are caveats: Attendings must be intimately involved in operations and scrubbed for every key and critical portion of the operation; also operations being scheduled in separate rooms must be done so with reasonable foresight."
As long as institutions are following these practices, he concluded, "It would appear that lens of current policy efforts is too narrow by focusing on perception. The debate should be refocused by challenging training programs to strengthen attending involvement and ensure the requisite competence of their trainees."
Dr. Yount reported no relevant disclosures.
Major finding: Running two rooms was not associated with any differences in operative duration, morbidity, or mortality in multivariate regression analyses, and there were no statistically significant differences in observed outcomes in any category.
Data source: The study reviewed 1,377 cardiac and 1682 general thoracic operations performed from July 2011 to July 2013 by attending, case type, and whether the attending was simultaneously supervising two surgeries.
Disclosures: Dr. Yount had no disclosures.
Delay can reduce TEVAR problems with type B dissections
NEW YORK – Patients with acute type B aortic dissections who are treated with thoracic endovascular aortic repair within 48 hours of symptom onset are more than three times as likely to have a severe complication as are those who are treated between 2 and 6 weeks after initial presentation, according to Dr. Nimesh D. Desai.
"There has been a dramatic increase in survival of patients managed with TEVAR for life-threatening complications of type B dissection versus any other therapy. Recently, [FDA] approval of two devices in the U.S. (Gore cTAG and Medtronic Valiant Captiva TEVAR grafts) has really opened up the field of inquiry into the optimal timing of TEVAR in the nonemergent, non–life-threatening complicated type B dissection patient. In the current study, we analyzed the impact of timing of intervention," Dr. Desai said at the meeting sponsored by the American Association for Thoracic Surgery.
Between 2005 and 2012, 317 people were admitted to the Hospital of the University of Pennsylvania, Philadelphia, for acute (less than 6 weeks) type B dissections, said Dr. Desai. He reviewed the outcomes for 132 patients who had undergone TEVAR, dividing them into three groups according to the timing of the procedure: acute/early (TEVAR within 48 hours of symptom onset), n = 70; acute/delayed (TEVAR 48 hours to 14 days following symptom onset), n = 44; and subacute (TEVAR 2-6 weeks following symptom onset), n = 18. Patients in all three groups were generally between 63 and 65 years old. Most were men who had histories of severe hypertension and smoking. About 10% had a previous stroke.
Those in the acute/early group were more likely to have undergone TEVAR for life-threatening conditions. For instance, 43.28% of the acute/early group had a contained rupture vs. 25% of the acute/delayed and 11.11% of the subacute patients, a significant difference. The acute/early group also had higher rates of frank rupture and clinical malperfusion than did the other groups.
In contrast, 56% of the subacute group underwent TEVAR because they manifested "softer indications" of impending rupture or radiographic malperfusion, which were not considered to be as emergent. Three-quarters of the subacute group had already been discharged home and were readmitted for the procedure, reported Dr. Desai, who is at the University of Pennsylvania.
The overall rate of severe postoperative complications was 39% in the acute/early group, 27% in the acute/delayed group, and 11.1% in the subacute group, a significant difference.
In-house mortality was 8.5% in the acute/early group (nine patients), 4.5% in the acute/delayed group (three), and 0 in the subacute group, a nonsignificant difference. Similar, but nonsignificant trends were found in 30-day mortality (11.7%, 6.8%, and 0%) and stroke (5.6%, 4.6%, and 0%).
An opposite trend toward higher rates of paralysis was observed in the subacute group (7.04%, 4.5%, and 9.5%, respectively, in the three groups).
The incidence of retrograde type A aortic dissection was 8.5% in the acute/early, 6.8% in the acute/delayed, and 4.7% in the subacute groups. "Retrograde type A dissections are an iatrogenic disease caused by stenting type B dissections," said Dr. Desai. "They almost never happen with any other type of stent graft category."
He said that in his experience, the risk increases with excessive oversizing of stents and the dissections tend to occur at the interface between the native aorta and stent graft. In addition, he noted, many of the dissections occur a year or more after the original dissection.
"Delayed intervention appears to lower the risk of complications of TEVAR for aortic dissection in patients who are stable enough to wait. Our practice is to wait 10-14 days for remodeling indications," said Dr. Desai. Type B patients who are initially managed medically should be followed closely, he said, because they are at risk for the onset of new indications that might require TEVAR.
Operator experience and adequate case planning are critical when treating patients in the setting of acute or subacute type A dissections, Dr. Desai said, adding that the development of devices specific for dissections, rather than those designed for aneurysm pathologies, may lead to fewer complications.
Dr. Desai is a primary investigator for Food and Drug Administration TEVAR trials for W.L. Gore and Associates, Medtronic, and Cook Medical.
NEW YORK – Patients with acute type B aortic dissections who are treated with thoracic endovascular aortic repair within 48 hours of symptom onset are more than three times as likely to have a severe complication as are those who are treated between 2 and 6 weeks after initial presentation, according to Dr. Nimesh D. Desai.
"There has been a dramatic increase in survival of patients managed with TEVAR for life-threatening complications of type B dissection versus any other therapy. Recently, [FDA] approval of two devices in the U.S. (Gore cTAG and Medtronic Valiant Captiva TEVAR grafts) has really opened up the field of inquiry into the optimal timing of TEVAR in the nonemergent, non–life-threatening complicated type B dissection patient. In the current study, we analyzed the impact of timing of intervention," Dr. Desai said at the meeting sponsored by the American Association for Thoracic Surgery.
Between 2005 and 2012, 317 people were admitted to the Hospital of the University of Pennsylvania, Philadelphia, for acute (less than 6 weeks) type B dissections, said Dr. Desai. He reviewed the outcomes for 132 patients who had undergone TEVAR, dividing them into three groups according to the timing of the procedure: acute/early (TEVAR within 48 hours of symptom onset), n = 70; acute/delayed (TEVAR 48 hours to 14 days following symptom onset), n = 44; and subacute (TEVAR 2-6 weeks following symptom onset), n = 18. Patients in all three groups were generally between 63 and 65 years old. Most were men who had histories of severe hypertension and smoking. About 10% had a previous stroke.
Those in the acute/early group were more likely to have undergone TEVAR for life-threatening conditions. For instance, 43.28% of the acute/early group had a contained rupture vs. 25% of the acute/delayed and 11.11% of the subacute patients, a significant difference. The acute/early group also had higher rates of frank rupture and clinical malperfusion than did the other groups.
In contrast, 56% of the subacute group underwent TEVAR because they manifested "softer indications" of impending rupture or radiographic malperfusion, which were not considered to be as emergent. Three-quarters of the subacute group had already been discharged home and were readmitted for the procedure, reported Dr. Desai, who is at the University of Pennsylvania.
The overall rate of severe postoperative complications was 39% in the acute/early group, 27% in the acute/delayed group, and 11.1% in the subacute group, a significant difference.
In-house mortality was 8.5% in the acute/early group (nine patients), 4.5% in the acute/delayed group (three), and 0 in the subacute group, a nonsignificant difference. Similar, but nonsignificant trends were found in 30-day mortality (11.7%, 6.8%, and 0%) and stroke (5.6%, 4.6%, and 0%).
An opposite trend toward higher rates of paralysis was observed in the subacute group (7.04%, 4.5%, and 9.5%, respectively, in the three groups).
The incidence of retrograde type A aortic dissection was 8.5% in the acute/early, 6.8% in the acute/delayed, and 4.7% in the subacute groups. "Retrograde type A dissections are an iatrogenic disease caused by stenting type B dissections," said Dr. Desai. "They almost never happen with any other type of stent graft category."
He said that in his experience, the risk increases with excessive oversizing of stents and the dissections tend to occur at the interface between the native aorta and stent graft. In addition, he noted, many of the dissections occur a year or more after the original dissection.
"Delayed intervention appears to lower the risk of complications of TEVAR for aortic dissection in patients who are stable enough to wait. Our practice is to wait 10-14 days for remodeling indications," said Dr. Desai. Type B patients who are initially managed medically should be followed closely, he said, because they are at risk for the onset of new indications that might require TEVAR.
Operator experience and adequate case planning are critical when treating patients in the setting of acute or subacute type A dissections, Dr. Desai said, adding that the development of devices specific for dissections, rather than those designed for aneurysm pathologies, may lead to fewer complications.
Dr. Desai is a primary investigator for Food and Drug Administration TEVAR trials for W.L. Gore and Associates, Medtronic, and Cook Medical.
NEW YORK – Patients with acute type B aortic dissections who are treated with thoracic endovascular aortic repair within 48 hours of symptom onset are more than three times as likely to have a severe complication as are those who are treated between 2 and 6 weeks after initial presentation, according to Dr. Nimesh D. Desai.
"There has been a dramatic increase in survival of patients managed with TEVAR for life-threatening complications of type B dissection versus any other therapy. Recently, [FDA] approval of two devices in the U.S. (Gore cTAG and Medtronic Valiant Captiva TEVAR grafts) has really opened up the field of inquiry into the optimal timing of TEVAR in the nonemergent, non–life-threatening complicated type B dissection patient. In the current study, we analyzed the impact of timing of intervention," Dr. Desai said at the meeting sponsored by the American Association for Thoracic Surgery.
Between 2005 and 2012, 317 people were admitted to the Hospital of the University of Pennsylvania, Philadelphia, for acute (less than 6 weeks) type B dissections, said Dr. Desai. He reviewed the outcomes for 132 patients who had undergone TEVAR, dividing them into three groups according to the timing of the procedure: acute/early (TEVAR within 48 hours of symptom onset), n = 70; acute/delayed (TEVAR 48 hours to 14 days following symptom onset), n = 44; and subacute (TEVAR 2-6 weeks following symptom onset), n = 18. Patients in all three groups were generally between 63 and 65 years old. Most were men who had histories of severe hypertension and smoking. About 10% had a previous stroke.
Those in the acute/early group were more likely to have undergone TEVAR for life-threatening conditions. For instance, 43.28% of the acute/early group had a contained rupture vs. 25% of the acute/delayed and 11.11% of the subacute patients, a significant difference. The acute/early group also had higher rates of frank rupture and clinical malperfusion than did the other groups.
In contrast, 56% of the subacute group underwent TEVAR because they manifested "softer indications" of impending rupture or radiographic malperfusion, which were not considered to be as emergent. Three-quarters of the subacute group had already been discharged home and were readmitted for the procedure, reported Dr. Desai, who is at the University of Pennsylvania.
The overall rate of severe postoperative complications was 39% in the acute/early group, 27% in the acute/delayed group, and 11.1% in the subacute group, a significant difference.
In-house mortality was 8.5% in the acute/early group (nine patients), 4.5% in the acute/delayed group (three), and 0 in the subacute group, a nonsignificant difference. Similar, but nonsignificant trends were found in 30-day mortality (11.7%, 6.8%, and 0%) and stroke (5.6%, 4.6%, and 0%).
An opposite trend toward higher rates of paralysis was observed in the subacute group (7.04%, 4.5%, and 9.5%, respectively, in the three groups).
The incidence of retrograde type A aortic dissection was 8.5% in the acute/early, 6.8% in the acute/delayed, and 4.7% in the subacute groups. "Retrograde type A dissections are an iatrogenic disease caused by stenting type B dissections," said Dr. Desai. "They almost never happen with any other type of stent graft category."
He said that in his experience, the risk increases with excessive oversizing of stents and the dissections tend to occur at the interface between the native aorta and stent graft. In addition, he noted, many of the dissections occur a year or more after the original dissection.
"Delayed intervention appears to lower the risk of complications of TEVAR for aortic dissection in patients who are stable enough to wait. Our practice is to wait 10-14 days for remodeling indications," said Dr. Desai. Type B patients who are initially managed medically should be followed closely, he said, because they are at risk for the onset of new indications that might require TEVAR.
Operator experience and adequate case planning are critical when treating patients in the setting of acute or subacute type A dissections, Dr. Desai said, adding that the development of devices specific for dissections, rather than those designed for aneurysm pathologies, may lead to fewer complications.
Dr. Desai is a primary investigator for Food and Drug Administration TEVAR trials for W.L. Gore and Associates, Medtronic, and Cook Medical.
Key clinical point: Delayed intervention appears to lower the risk of complications of TEVAR for aortic dissection in patients who are stable enough to wait.
Major finding: The overall risk of severe complications was more than threefold higher in patients who underwent TEVAR for acute type B aortic dissections within 48 hours of initial presentation, compared with those whose treatment could be delayed until 2-6 weeks after symptom onset.
Data source: A single-center, retrospective registry analysis of 317 patients.
Disclosures: Dr. Desai said he had no relevant financial disclosures.
Imaging procedure costs higher in the United States
When it comes to diagnostic imaging, the average costs of angiograms, abdominal CTs, and MRIs are higher in the United States than in other industrialized countries, according to the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of an angiogram in the United States last year was $907, about 11% higher than Argentina’s $818, which was the second-highest of the six countries included in all three IFHP comparisons. The lowest cost among the six countries was in the Netherlands, at $174.
For an abdominal CT scan, the average cost in the United States was $896 in 2013, compared with $500 in Australia. Spain had the lowest cost, with an average of $94, the IFHP reported. In the United States, the average price for an MRI in 2013 was $1,145, with the Netherlands second at $461 and Switzerland lowest at $138.
New Zealand, which was not included in the angiogram analysis and therefore left out of the graph below, was actually the second most-expensive country in which to get a CT scan ($731) and an MRI ($1,005).
The IFHP is composed of more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, and some are from the private sector. U.S. averages are calculated from more than 100 million claims in the Truven MarketScan Research databases.
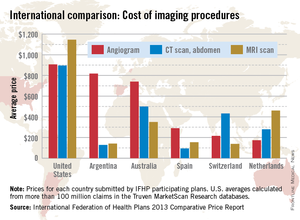
When it comes to diagnostic imaging, the average costs of angiograms, abdominal CTs, and MRIs are higher in the United States than in other industrialized countries, according to the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of an angiogram in the United States last year was $907, about 11% higher than Argentina’s $818, which was the second-highest of the six countries included in all three IFHP comparisons. The lowest cost among the six countries was in the Netherlands, at $174.
For an abdominal CT scan, the average cost in the United States was $896 in 2013, compared with $500 in Australia. Spain had the lowest cost, with an average of $94, the IFHP reported. In the United States, the average price for an MRI in 2013 was $1,145, with the Netherlands second at $461 and Switzerland lowest at $138.
New Zealand, which was not included in the angiogram analysis and therefore left out of the graph below, was actually the second most-expensive country in which to get a CT scan ($731) and an MRI ($1,005).
The IFHP is composed of more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, and some are from the private sector. U.S. averages are calculated from more than 100 million claims in the Truven MarketScan Research databases.

When it comes to diagnostic imaging, the average costs of angiograms, abdominal CTs, and MRIs are higher in the United States than in other industrialized countries, according to the International Federation of Health Plans’ 2013 Comparative Price Report.
The average cost of an angiogram in the United States last year was $907, about 11% higher than Argentina’s $818, which was the second-highest of the six countries included in all three IFHP comparisons. The lowest cost among the six countries was in the Netherlands, at $174.
For an abdominal CT scan, the average cost in the United States was $896 in 2013, compared with $500 in Australia. Spain had the lowest cost, with an average of $94, the IFHP reported. In the United States, the average price for an MRI in 2013 was $1,145, with the Netherlands second at $461 and Switzerland lowest at $138.
New Zealand, which was not included in the angiogram analysis and therefore left out of the graph below, was actually the second most-expensive country in which to get a CT scan ($731) and an MRI ($1,005).
The IFHP is composed of more than 100 member companies in 25 countries. For the survey, the price for each country was submitted by participating member plans. Some prices are drawn from the public sector, and some are from the private sector. U.S. averages are calculated from more than 100 million claims in the Truven MarketScan Research databases.