User login
The Official Newspaper of the American Association for Thoracic Surgery
Poor response to statins predicts growth in plaque
For about one in five patients with known atherosclerotic coronary artery disease, standard-dose therapy with statins did not result in significant lowering of LDL cholesterol.
Furthermore, the results of this large pooled data sample showed that for statin hyporesponders, statin therapy did not prevent progression of intravascular plaque volume as measured by grayscale intravascular ultrasound.
Patients exhibit a wide range of response to standard statin dosing, and the effect of minimal LDL-C lowering on atherosclerotic disease progression had not previously been determined, according to Dr. Yu Kataoka of the University of Adelaide, Australia, and his colleagues (Arterioscler. Thromb. Vasc. Biol. 2015 [doi:10.1161/ATVBAHA.114.304477]).
Investigators pooled data from seven clinical trials that examined 647 total patients with angiographically confirmed CAD who were initiated on statins and followed by serial intravascular ultrasound. The present study analyzed baseline characteristics, serial lipid profile, and atheroma burden for the group.
In all, 130 patients of the 647 (20%) had minimal LDL-C lowering with statin therapy, showing nonsignificant lowering or even an increase in LDL-C levels during the study period. This group of hyporesponders differed in being slightly younger, more obese, less likely to have hypertension and dyslipidemia, and less likely to be receiving beta-blockers than were the statin responders. Other patient characteristics were similar between the two groups. A variety of agents were used, including atorvastatin, rosuvastatin, simvastatin, and pravastatin. Concurrent administration of other antiatherosclerotic agents was permitted and was similar between the groups. Atheroma burden at baseline was also similar between the two groups.
Measuring serial changes in atheroma burden showed a significant difference between statin responders and hyporesponders. The adjusted change in atheroma volume was –0.21% for the responders, compared with +0.83% for the hyporesponders (P = .006). Lumen volume decreased 11.64 mm3 for the responders, while the reduction was 16.54 mm3 for the hyporesponders (P = .006). Of those who responded to lipid therapy with LDL-C lowering, 29.8% had substantial atheroma regression, while 25.9% had substantial plaque progression; among hyporesponders, however, just 13.8% experienced significant plaque regression, while 37.7% had significant atheroma progression, both significant differences.
Dr. Kataoka and his colleagues emphasized that the factors contributing to poor statin response are not well understood. They noted that for this study, the pooled trials all showed adherence rates over 90%, eliminating patient compliance as a variable. Rigorous statistical techniques were used to control for comorbidities and coadministered medications. There are known genetic polymorphisms and phenotypic variations in statin metabolism, though these were not reported here. Although the results were not statistically significant, C-reactive protein levels were higher for the hyporesponse group, suggesting that another factor may be individual response to the anti-inflammatory effect that is among the known pleiotropic effects of this drug class.
In an interview, lead author Stephen Nicholls noted that many clinicians are still reluctant to treat to full effect. Citing the concept of “clinical inertia,” Dr. Nicholls pointed out that “Even when statins are prescribed, they are often at lower doses than ideal. That translated to more plaque growth, which leads directly to more heart attacks and more revascularization procedures.”
Study limitations included the potential residual confounding effects of pooling data from seven discrete clinical trials, though mixed modeling techniques attempted to correct for this effect. The present study also reported atheroma burden, but not actual clinical events. The study authors noted, however, that they had previously reported a direct relationship between atheroma progression and the occurrence of cardiovascular events.
Dr. Nicholls has received speaking honoraria and research support from many pharmaceutical companies, and from Infraredx. Dr. Steven E. Nissen of the Cleveland Clinic was a coinvestigator and has received research support from and is a consultant/adviser to numerous pharmaceutical companies; all honoraria or consulting fees go directly to charity so that he receives neither income nor a tax deduction. The other authors report no conflicts.
For about one in five patients with known atherosclerotic coronary artery disease, standard-dose therapy with statins did not result in significant lowering of LDL cholesterol.
Furthermore, the results of this large pooled data sample showed that for statin hyporesponders, statin therapy did not prevent progression of intravascular plaque volume as measured by grayscale intravascular ultrasound.
Patients exhibit a wide range of response to standard statin dosing, and the effect of minimal LDL-C lowering on atherosclerotic disease progression had not previously been determined, according to Dr. Yu Kataoka of the University of Adelaide, Australia, and his colleagues (Arterioscler. Thromb. Vasc. Biol. 2015 [doi:10.1161/ATVBAHA.114.304477]).
Investigators pooled data from seven clinical trials that examined 647 total patients with angiographically confirmed CAD who were initiated on statins and followed by serial intravascular ultrasound. The present study analyzed baseline characteristics, serial lipid profile, and atheroma burden for the group.
In all, 130 patients of the 647 (20%) had minimal LDL-C lowering with statin therapy, showing nonsignificant lowering or even an increase in LDL-C levels during the study period. This group of hyporesponders differed in being slightly younger, more obese, less likely to have hypertension and dyslipidemia, and less likely to be receiving beta-blockers than were the statin responders. Other patient characteristics were similar between the two groups. A variety of agents were used, including atorvastatin, rosuvastatin, simvastatin, and pravastatin. Concurrent administration of other antiatherosclerotic agents was permitted and was similar between the groups. Atheroma burden at baseline was also similar between the two groups.
Measuring serial changes in atheroma burden showed a significant difference between statin responders and hyporesponders. The adjusted change in atheroma volume was –0.21% for the responders, compared with +0.83% for the hyporesponders (P = .006). Lumen volume decreased 11.64 mm3 for the responders, while the reduction was 16.54 mm3 for the hyporesponders (P = .006). Of those who responded to lipid therapy with LDL-C lowering, 29.8% had substantial atheroma regression, while 25.9% had substantial plaque progression; among hyporesponders, however, just 13.8% experienced significant plaque regression, while 37.7% had significant atheroma progression, both significant differences.
Dr. Kataoka and his colleagues emphasized that the factors contributing to poor statin response are not well understood. They noted that for this study, the pooled trials all showed adherence rates over 90%, eliminating patient compliance as a variable. Rigorous statistical techniques were used to control for comorbidities and coadministered medications. There are known genetic polymorphisms and phenotypic variations in statin metabolism, though these were not reported here. Although the results were not statistically significant, C-reactive protein levels were higher for the hyporesponse group, suggesting that another factor may be individual response to the anti-inflammatory effect that is among the known pleiotropic effects of this drug class.
In an interview, lead author Stephen Nicholls noted that many clinicians are still reluctant to treat to full effect. Citing the concept of “clinical inertia,” Dr. Nicholls pointed out that “Even when statins are prescribed, they are often at lower doses than ideal. That translated to more plaque growth, which leads directly to more heart attacks and more revascularization procedures.”
Study limitations included the potential residual confounding effects of pooling data from seven discrete clinical trials, though mixed modeling techniques attempted to correct for this effect. The present study also reported atheroma burden, but not actual clinical events. The study authors noted, however, that they had previously reported a direct relationship between atheroma progression and the occurrence of cardiovascular events.
Dr. Nicholls has received speaking honoraria and research support from many pharmaceutical companies, and from Infraredx. Dr. Steven E. Nissen of the Cleveland Clinic was a coinvestigator and has received research support from and is a consultant/adviser to numerous pharmaceutical companies; all honoraria or consulting fees go directly to charity so that he receives neither income nor a tax deduction. The other authors report no conflicts.
For about one in five patients with known atherosclerotic coronary artery disease, standard-dose therapy with statins did not result in significant lowering of LDL cholesterol.
Furthermore, the results of this large pooled data sample showed that for statin hyporesponders, statin therapy did not prevent progression of intravascular plaque volume as measured by grayscale intravascular ultrasound.
Patients exhibit a wide range of response to standard statin dosing, and the effect of minimal LDL-C lowering on atherosclerotic disease progression had not previously been determined, according to Dr. Yu Kataoka of the University of Adelaide, Australia, and his colleagues (Arterioscler. Thromb. Vasc. Biol. 2015 [doi:10.1161/ATVBAHA.114.304477]).
Investigators pooled data from seven clinical trials that examined 647 total patients with angiographically confirmed CAD who were initiated on statins and followed by serial intravascular ultrasound. The present study analyzed baseline characteristics, serial lipid profile, and atheroma burden for the group.
In all, 130 patients of the 647 (20%) had minimal LDL-C lowering with statin therapy, showing nonsignificant lowering or even an increase in LDL-C levels during the study period. This group of hyporesponders differed in being slightly younger, more obese, less likely to have hypertension and dyslipidemia, and less likely to be receiving beta-blockers than were the statin responders. Other patient characteristics were similar between the two groups. A variety of agents were used, including atorvastatin, rosuvastatin, simvastatin, and pravastatin. Concurrent administration of other antiatherosclerotic agents was permitted and was similar between the groups. Atheroma burden at baseline was also similar between the two groups.
Measuring serial changes in atheroma burden showed a significant difference between statin responders and hyporesponders. The adjusted change in atheroma volume was –0.21% for the responders, compared with +0.83% for the hyporesponders (P = .006). Lumen volume decreased 11.64 mm3 for the responders, while the reduction was 16.54 mm3 for the hyporesponders (P = .006). Of those who responded to lipid therapy with LDL-C lowering, 29.8% had substantial atheroma regression, while 25.9% had substantial plaque progression; among hyporesponders, however, just 13.8% experienced significant plaque regression, while 37.7% had significant atheroma progression, both significant differences.
Dr. Kataoka and his colleagues emphasized that the factors contributing to poor statin response are not well understood. They noted that for this study, the pooled trials all showed adherence rates over 90%, eliminating patient compliance as a variable. Rigorous statistical techniques were used to control for comorbidities and coadministered medications. There are known genetic polymorphisms and phenotypic variations in statin metabolism, though these were not reported here. Although the results were not statistically significant, C-reactive protein levels were higher for the hyporesponse group, suggesting that another factor may be individual response to the anti-inflammatory effect that is among the known pleiotropic effects of this drug class.
In an interview, lead author Stephen Nicholls noted that many clinicians are still reluctant to treat to full effect. Citing the concept of “clinical inertia,” Dr. Nicholls pointed out that “Even when statins are prescribed, they are often at lower doses than ideal. That translated to more plaque growth, which leads directly to more heart attacks and more revascularization procedures.”
Study limitations included the potential residual confounding effects of pooling data from seven discrete clinical trials, though mixed modeling techniques attempted to correct for this effect. The present study also reported atheroma burden, but not actual clinical events. The study authors noted, however, that they had previously reported a direct relationship between atheroma progression and the occurrence of cardiovascular events.
Dr. Nicholls has received speaking honoraria and research support from many pharmaceutical companies, and from Infraredx. Dr. Steven E. Nissen of the Cleveland Clinic was a coinvestigator and has received research support from and is a consultant/adviser to numerous pharmaceutical companies; all honoraria or consulting fees go directly to charity so that he receives neither income nor a tax deduction. The other authors report no conflicts.
FROM ARTERIOSCLEROSIS, THROMBOSIS, AND VASCULAR BIOLOGY
Key clinical point: Patients on statins who had minimal LDL-C lowering also showed increased atheroma progression.
Major finding: Of 647 patients with CAD, 20% were hyporesponders to statin therapy and experienced greater progression of atheroma volume than statin responders (adjusted +0.83% vs. –0.21%, P = .006).
Data source: Pooled data from seven clinical trials, yielding 647 patients with angiographically confirmed CAD who were initiated on standard lipid dosing and followed by baseline and serial grayscale intravascular ultrasounds.
Disclosures: Dr. Nicholls has received speaking honoraria and research support from many pharmaceutical companies, and from Infraredx. Dr. Steven E. Nissen of the Cleveland Clinic was a coinvestigator and has received research support from and is a consultant/adviser to numerous pharmaceutical companies; all honoraria or consulting fees go directly to charity so that he receives neither income nor a tax deduction. The other authors report no conflicts.
CMS extends 2014 Medicare meaningful use attestation deadline
Physicians and other eligible professionals will now have until March 20 to attest to meaningful use of electronic health records for the 2014 reporting year and avoid a Medicare penalty for 2016.
The Centers for Medicare & Medicaid Services announced it was extending the deadline from Feb. 28. Eligible providers must attest to meaningful use every year to receive bonus payments under the EHR Incentive Program. Those who fail to attest by March 20 for the 2014 reporting year will see a 2% reduction in Medicare payments in 2016.
During this extension period, providers can make a one-time switch between the Medicare and Medicaid EHR Incentive Programs.
Physicians and other eligible professionals will now have until March 20 to attest to meaningful use of electronic health records for the 2014 reporting year and avoid a Medicare penalty for 2016.
The Centers for Medicare & Medicaid Services announced it was extending the deadline from Feb. 28. Eligible providers must attest to meaningful use every year to receive bonus payments under the EHR Incentive Program. Those who fail to attest by March 20 for the 2014 reporting year will see a 2% reduction in Medicare payments in 2016.
During this extension period, providers can make a one-time switch between the Medicare and Medicaid EHR Incentive Programs.
Physicians and other eligible professionals will now have until March 20 to attest to meaningful use of electronic health records for the 2014 reporting year and avoid a Medicare penalty for 2016.
The Centers for Medicare & Medicaid Services announced it was extending the deadline from Feb. 28. Eligible providers must attest to meaningful use every year to receive bonus payments under the EHR Incentive Program. Those who fail to attest by March 20 for the 2014 reporting year will see a 2% reduction in Medicare payments in 2016.
During this extension period, providers can make a one-time switch between the Medicare and Medicaid EHR Incentive Programs.
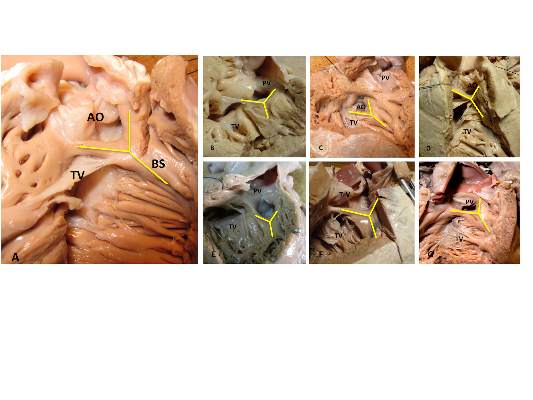
Anatomy of VSD in outflow tract defects indicates a continuum and has surgical relevance
The outlet ventricular septal defect is a cornerstone of the outflow tract defects and exists on a continuum that is anatomically different from the isolated central perimembranous VSD, according to the results of an observational study of 277 preserved heart specimens with isolated outlet ventricular septal defect without subpulmonary stenosis.
“In all of the specimens studied, the VSD always opened in the outlet of the right ventricle, cradled between the two limbs of the septal band, irrespective of the presence or absence of a fibrous continuity between the aortic and tricuspid valves, and the presence of an outlet septum,” according to the report published in the March issue of the Journal of Thoracic and Cardiovascular Surgery by Dr. Meriem Mostefa-Kara of the Paris Descartes University and her colleagues.
The 277 specimens comprised 19 with isolated ventricular septal defect; 71 with tetralogy of Fallot (TOF); 51 with TOF with pulmonary atresia (PA); 54 with common arterial trunk (CAT); 65 with double-outlet right ventricle (DORV), with subaortic, doubly committed, or subpulmonary ventricular septal defect; and 17 with interrupted aortic arch (IAA) type B (doi:10.1016/j.jtcvs.2014.11.087).
Previous studies have shown that all malalignment defects include a VSD because of the malalignment and the absence of fusion between the outlet septum and the rest of the ventricular septum, and all authors agree that this VSD is cradled between the two limbs of the septal band, according to the researchers.
They found such an outlet VSD in all of the heart specimens studied, Dr. Mostefa-Kara and her colleagues added. In addition, they found that its anatomic variants were distributed differently according to the defect involved. This was especially true when focusing of the posteroinferior rim and particularly on the aortic-tricuspid fibrous continuity. In addition, this continuity occurred with different frequency among the various outflow tract defects studied.
They found the highest rate of continuity in isolated outlet VSD, then decreasing progressively from TOF to TOF-PA, then DORV, becoming “exceedingly rare” in CAT and absent in IAA type B.
The researchers also analyzed 26 hearts with isolated central perimembranous VSD from their anatomic collection and compared these with the outlet VSD hearts. All 26 of these VSDs were located behind the septal leaflet of the tricuspid valve, under the posteroinferior limb of the septal band, and NOT between the two limbs of the septal band as was the case with the outlet VSDs.
This led them to state that there was a “blatant anatomical difference between the these two types of VSDs,” and pointed out the risk of confusion. “The presence of a fibrous continuity at the posteroinferior rim of the VSD is important for the surgeon, because it makes the conduction axis vulnerable during surgery and therefore must be described specifically in the preoperative assessment of the defect,” they warned.
“This anatomic approach places the outlet VSD as a cornerstone of the outflow tract defects, anatomically different from the isolated central perimembranous VSD. This may help us to better understand the anatomy of the VSDs and to clarify their classification and terminology,” Dr. Mostefa-Kara and her colleagues concluded.
The study was sponsored by the French Society of Cardiology. The authors reported having no relevant disclosures.
The Paris researchers’ study is important for several reasons, according to the invited editorial commentary by Dr. Robert H. Anderson (doi:10.1016/j,jtcvs.2014.12.003). “First, it shows that careful examination of archives of autopsied hearts can still provide new information. Second, to provide all the information required to achieve safe and secure surgical closures of channels between the ventricles, they emphasize that knowledge is required how the defect opens toward the right ventricle and regarding the boundaries around which the surgeon will place a patch to restore septal integrity. The location of the defect relative to the right ventricle is geography. The details of the margins of the channel requiring closure represent its geometry. In earlier years, investigators tended to use either the geography or the geometry to provide their definitions, or else they accorded priority to one of these features. Both features are surgically important.” In addition, “as the Parisian investigators stress, it is not sufficient simply to state that a defect is perimembranous. We should now be distinguishing between perimembranous defects opening centrally, those that open to the outlet of the right ventricle between the limbs of the septal band, and those that can open to the right ventricular inlet. Another important feature of their research is the presence or absence of septal malalignment.”
Dr. Anderson is a professorial fellow at the Institute of Genetic Medicine, Newcastle University, Newcastle-upon-Tyne, England.
The Paris researchers’ study is important for several reasons, according to the invited editorial commentary by Dr. Robert H. Anderson (doi:10.1016/j,jtcvs.2014.12.003). “First, it shows that careful examination of archives of autopsied hearts can still provide new information. Second, to provide all the information required to achieve safe and secure surgical closures of channels between the ventricles, they emphasize that knowledge is required how the defect opens toward the right ventricle and regarding the boundaries around which the surgeon will place a patch to restore septal integrity. The location of the defect relative to the right ventricle is geography. The details of the margins of the channel requiring closure represent its geometry. In earlier years, investigators tended to use either the geography or the geometry to provide their definitions, or else they accorded priority to one of these features. Both features are surgically important.” In addition, “as the Parisian investigators stress, it is not sufficient simply to state that a defect is perimembranous. We should now be distinguishing between perimembranous defects opening centrally, those that open to the outlet of the right ventricle between the limbs of the septal band, and those that can open to the right ventricular inlet. Another important feature of their research is the presence or absence of septal malalignment.”
Dr. Anderson is a professorial fellow at the Institute of Genetic Medicine, Newcastle University, Newcastle-upon-Tyne, England.
The Paris researchers’ study is important for several reasons, according to the invited editorial commentary by Dr. Robert H. Anderson (doi:10.1016/j,jtcvs.2014.12.003). “First, it shows that careful examination of archives of autopsied hearts can still provide new information. Second, to provide all the information required to achieve safe and secure surgical closures of channels between the ventricles, they emphasize that knowledge is required how the defect opens toward the right ventricle and regarding the boundaries around which the surgeon will place a patch to restore septal integrity. The location of the defect relative to the right ventricle is geography. The details of the margins of the channel requiring closure represent its geometry. In earlier years, investigators tended to use either the geography or the geometry to provide their definitions, or else they accorded priority to one of these features. Both features are surgically important.” In addition, “as the Parisian investigators stress, it is not sufficient simply to state that a defect is perimembranous. We should now be distinguishing between perimembranous defects opening centrally, those that open to the outlet of the right ventricle between the limbs of the septal band, and those that can open to the right ventricular inlet. Another important feature of their research is the presence or absence of septal malalignment.”
Dr. Anderson is a professorial fellow at the Institute of Genetic Medicine, Newcastle University, Newcastle-upon-Tyne, England.
The outlet ventricular septal defect is a cornerstone of the outflow tract defects and exists on a continuum that is anatomically different from the isolated central perimembranous VSD, according to the results of an observational study of 277 preserved heart specimens with isolated outlet ventricular septal defect without subpulmonary stenosis.
“In all of the specimens studied, the VSD always opened in the outlet of the right ventricle, cradled between the two limbs of the septal band, irrespective of the presence or absence of a fibrous continuity between the aortic and tricuspid valves, and the presence of an outlet septum,” according to the report published in the March issue of the Journal of Thoracic and Cardiovascular Surgery by Dr. Meriem Mostefa-Kara of the Paris Descartes University and her colleagues.
The 277 specimens comprised 19 with isolated ventricular septal defect; 71 with tetralogy of Fallot (TOF); 51 with TOF with pulmonary atresia (PA); 54 with common arterial trunk (CAT); 65 with double-outlet right ventricle (DORV), with subaortic, doubly committed, or subpulmonary ventricular septal defect; and 17 with interrupted aortic arch (IAA) type B (doi:10.1016/j.jtcvs.2014.11.087).
Previous studies have shown that all malalignment defects include a VSD because of the malalignment and the absence of fusion between the outlet septum and the rest of the ventricular septum, and all authors agree that this VSD is cradled between the two limbs of the septal band, according to the researchers.
They found such an outlet VSD in all of the heart specimens studied, Dr. Mostefa-Kara and her colleagues added. In addition, they found that its anatomic variants were distributed differently according to the defect involved. This was especially true when focusing of the posteroinferior rim and particularly on the aortic-tricuspid fibrous continuity. In addition, this continuity occurred with different frequency among the various outflow tract defects studied.
They found the highest rate of continuity in isolated outlet VSD, then decreasing progressively from TOF to TOF-PA, then DORV, becoming “exceedingly rare” in CAT and absent in IAA type B.
The researchers also analyzed 26 hearts with isolated central perimembranous VSD from their anatomic collection and compared these with the outlet VSD hearts. All 26 of these VSDs were located behind the septal leaflet of the tricuspid valve, under the posteroinferior limb of the septal band, and NOT between the two limbs of the septal band as was the case with the outlet VSDs.
This led them to state that there was a “blatant anatomical difference between the these two types of VSDs,” and pointed out the risk of confusion. “The presence of a fibrous continuity at the posteroinferior rim of the VSD is important for the surgeon, because it makes the conduction axis vulnerable during surgery and therefore must be described specifically in the preoperative assessment of the defect,” they warned.
“This anatomic approach places the outlet VSD as a cornerstone of the outflow tract defects, anatomically different from the isolated central perimembranous VSD. This may help us to better understand the anatomy of the VSDs and to clarify their classification and terminology,” Dr. Mostefa-Kara and her colleagues concluded.
The study was sponsored by the French Society of Cardiology. The authors reported having no relevant disclosures.
The outlet ventricular septal defect is a cornerstone of the outflow tract defects and exists on a continuum that is anatomically different from the isolated central perimembranous VSD, according to the results of an observational study of 277 preserved heart specimens with isolated outlet ventricular septal defect without subpulmonary stenosis.
“In all of the specimens studied, the VSD always opened in the outlet of the right ventricle, cradled between the two limbs of the septal band, irrespective of the presence or absence of a fibrous continuity between the aortic and tricuspid valves, and the presence of an outlet septum,” according to the report published in the March issue of the Journal of Thoracic and Cardiovascular Surgery by Dr. Meriem Mostefa-Kara of the Paris Descartes University and her colleagues.
The 277 specimens comprised 19 with isolated ventricular septal defect; 71 with tetralogy of Fallot (TOF); 51 with TOF with pulmonary atresia (PA); 54 with common arterial trunk (CAT); 65 with double-outlet right ventricle (DORV), with subaortic, doubly committed, or subpulmonary ventricular septal defect; and 17 with interrupted aortic arch (IAA) type B (doi:10.1016/j.jtcvs.2014.11.087).
Previous studies have shown that all malalignment defects include a VSD because of the malalignment and the absence of fusion between the outlet septum and the rest of the ventricular septum, and all authors agree that this VSD is cradled between the two limbs of the septal band, according to the researchers.
They found such an outlet VSD in all of the heart specimens studied, Dr. Mostefa-Kara and her colleagues added. In addition, they found that its anatomic variants were distributed differently according to the defect involved. This was especially true when focusing of the posteroinferior rim and particularly on the aortic-tricuspid fibrous continuity. In addition, this continuity occurred with different frequency among the various outflow tract defects studied.
They found the highest rate of continuity in isolated outlet VSD, then decreasing progressively from TOF to TOF-PA, then DORV, becoming “exceedingly rare” in CAT and absent in IAA type B.
The researchers also analyzed 26 hearts with isolated central perimembranous VSD from their anatomic collection and compared these with the outlet VSD hearts. All 26 of these VSDs were located behind the septal leaflet of the tricuspid valve, under the posteroinferior limb of the septal band, and NOT between the two limbs of the septal band as was the case with the outlet VSDs.
This led them to state that there was a “blatant anatomical difference between the these two types of VSDs,” and pointed out the risk of confusion. “The presence of a fibrous continuity at the posteroinferior rim of the VSD is important for the surgeon, because it makes the conduction axis vulnerable during surgery and therefore must be described specifically in the preoperative assessment of the defect,” they warned.
“This anatomic approach places the outlet VSD as a cornerstone of the outflow tract defects, anatomically different from the isolated central perimembranous VSD. This may help us to better understand the anatomy of the VSDs and to clarify their classification and terminology,” Dr. Mostefa-Kara and her colleagues concluded.
The study was sponsored by the French Society of Cardiology. The authors reported having no relevant disclosures.
FROM THE JOURNAL OF THORACIC AND CARDIOVASCULAR SURGERY
Key clinical point: The presence of a fibrous continuity at the postinferior rim of the VSD is important for the surgeon because it makes the conduction axis vulnerable during surgery and therefore must be described specifically in the preoperative assessment.
Major finding: The outlet VSD is a cornerstone of the outflow tract defects and exists on a continuum that is anatomically different from the isolated central perimembranous VSD.
Data source: The researchers examined 277 preserved heart specimens with isolated outlet ventricular septal defect.
Disclosures: The study was sponsored by the French Society of Cardiology. The authors reported having no relevant disclosures.
Asymptomatic PAD treatment - New clinical guidelines add to LE debate
The Society for Vascular Surgery has just released its long-awaited, “SVS practice guidelines for atherosclerotic occlusive disease of the lower extremities: Management of asymptomatic disease and claudication,” and they are available online.
View the guidelines at this link.
The guidelines represent more than four years of collaborative effort by a team of physicians, led by co-chairmen Drs. Michael S. Conte and Frank B. Pomposelli.
The Society for Vascular Surgery has just released its long-awaited, “SVS practice guidelines for atherosclerotic occlusive disease of the lower extremities: Management of asymptomatic disease and claudication,” and they are available online.
View the guidelines at this link.
The guidelines represent more than four years of collaborative effort by a team of physicians, led by co-chairmen Drs. Michael S. Conte and Frank B. Pomposelli.
The Society for Vascular Surgery has just released its long-awaited, “SVS practice guidelines for atherosclerotic occlusive disease of the lower extremities: Management of asymptomatic disease and claudication,” and they are available online.
View the guidelines at this link.
The guidelines represent more than four years of collaborative effort by a team of physicians, led by co-chairmen Drs. Michael S. Conte and Frank B. Pomposelli.
Registration & Housing Now Open for AATS Week 2015
AATS Week 2015 includes two terrific events: The Mitral Conclave (April 23-24, 2015, New York City) and AATS 95th Annual Meeting (April 25-29, 2015, Seattle WA).
Register for both and receive a $100 discount off the Mitral Conclave fee!
Registration and housing reservations are now open. Information can be found at: http://aats.org/aatsweek.
Three ways to register:
** Online at www.aats.org. Registration and hotel reservations can be made at the same time.
** Call the AATS/Experient
Customer Service Desk:
US: (800) 424-5249
International: (847) 996-5829
** Mail/fax Registration form is available upon request from LK Coughlin, lkcoughlin@aats.org. Fill out and mail to: AATS/Experient P.O. Box 4088 Frederick, MD 21705 with check and credit card information. Or fax to: 301-694-5124 (fax requires credit card information).
AATS Week 2015 includes two terrific events: The Mitral Conclave (April 23-24, 2015, New York City) and AATS 95th Annual Meeting (April 25-29, 2015, Seattle WA).
Register for both and receive a $100 discount off the Mitral Conclave fee!
Registration and housing reservations are now open. Information can be found at: http://aats.org/aatsweek.
Three ways to register:
** Online at www.aats.org. Registration and hotel reservations can be made at the same time.
** Call the AATS/Experient
Customer Service Desk:
US: (800) 424-5249
International: (847) 996-5829
** Mail/fax Registration form is available upon request from LK Coughlin, lkcoughlin@aats.org. Fill out and mail to: AATS/Experient P.O. Box 4088 Frederick, MD 21705 with check and credit card information. Or fax to: 301-694-5124 (fax requires credit card information).
AATS Week 2015 includes two terrific events: The Mitral Conclave (April 23-24, 2015, New York City) and AATS 95th Annual Meeting (April 25-29, 2015, Seattle WA).
Register for both and receive a $100 discount off the Mitral Conclave fee!
Registration and housing reservations are now open. Information can be found at: http://aats.org/aatsweek.
Three ways to register:
** Online at www.aats.org. Registration and hotel reservations can be made at the same time.
** Call the AATS/Experient
Customer Service Desk:
US: (800) 424-5249
International: (847) 996-5829
** Mail/fax Registration form is available upon request from LK Coughlin, lkcoughlin@aats.org. Fill out and mail to: AATS/Experient P.O. Box 4088 Frederick, MD 21705 with check and credit card information. Or fax to: 301-694-5124 (fax requires credit card information).
Device reduces coronary sinus, relieves refractory angina
A new endoluminal device that reduces the coronary sinus significantly improved refractory angina in patients with advanced coronary artery disease who weren’t candidates for revascularization in a small phase II clinical trial. The results were published online Feb. 5 in the New England Journal of Medicine.
The international, randomized, double-blind study, sponsored by Neovasc, maker of the Reducer device, involved 104 patients enrolled who had Canadian Cardiovascular Society (CCS) class III or IV angina despite maximal medical therapy, evidence of reversible myocardial ischemia, and a left ventricular ejection fraction over 25%. These participants (mean age 68 years; range, 35-87 years) underwent right heart catheterization with angiography of the coronary sinus to determine whether the anatomy was suitable for implantation. Then 52 underwent implantation (the treatment group) and 52 underwent a sham procedure (the control group), and all were followed for up to 3 years, said Dr. Stefan Verheye of Antwerp (Belgium) Cardiovascular Center and his associates.
The stainless steel device is balloon expandable and shaped like an hourglass. It creates a focal narrowing that increases pressure in the coronary sinus, which is thought to recruit coronary collateral blood flow, redistributing blood from the less-ischemic epicardium to the ischemic endocardium. The primary efficacy end point of the trial – the number of patients whose angina improved by two or more CCS classes within 6 months – was achieved by more than twice as many patients in the treatment group (18, or 35%) as in the control group (8, or 15%).
A total of 37 patients (71%) who received active treatment showed an improvement of at least one CCS class, compared with 22 (42%) of those who received the sham treatment. Quality of life, as measured by Seattle Angina Questionnaire score, improved by 17.6 out of a possible 100 points for active treatment, compared with 7.6 points for sham treatment. And exercise duration improved by 1 minute (13%) for active treatment, compared with 4 seconds (1%) for sham treatment, Dr. Verheye and his associates said (N. Engl. J. Med. 2015 Feb. 5 [doi:10.1056/NEJMoa1402556]). “Our study was not statistically powered to detect an improvement in ischemia by means of objective measures such as stress testing or wall-motion index. A larger trial would be necessary to show such a benefit,” they noted.
There were 76 adverse events in patients who received the implant and 93 in the control group; the numbers of serious adverse events in this high-risk cohort were 10 and 24, respectively. This included one periprocedural MI that occurred in a patient receiving active treatment. Among 36 of the patients who received the implant and later underwent CT angiography, none showed any evidence of device migration or occlusion, the investigators added.
This study was supported by Neovasc, manufacturer of the Reducer device. Dr. Verheye reported having no financial disclosures. His associates reported ties to Neovasc, Abbott, Boston Scientific, Novo Nordisk, Eli Lilly, and Zealand Pharma.

|
Dr. Bernard Gersh |
Even though this trial was of high quality and well performed, the total number of patients in both study groups who had a positive outcome is only 26, which is not sufficient for a reliable estimation of modest treatment effects.
The small sample size necessarily limits confidence about this new device’s effectiveness. But if these findings are confirmed in future studies, coronary-sinus–reducing therapy may become a welcome and necessary addition to our options to improve quality of life for patients with refractory angina.
Christopher B. Granger, M.D., is at Duke Clinical Research Institute, Durham N.C. He reported numerous ties to industry sources. Bernard J. Gersh, M.D., D.Phil., is in the department of cardiovascular diseases at the Mayo Clinic, Rochester Minn. He reported having no financial disclosures. Dr. Granger and Dr. Gersh made these remarks in an editorial accompanying Dr. Verheye’s report (N. Engl. J. Med. 2015 Feb. 5 [doi:10.1056/NEJMe1413941]).

|
Dr. Bernard Gersh |
Even though this trial was of high quality and well performed, the total number of patients in both study groups who had a positive outcome is only 26, which is not sufficient for a reliable estimation of modest treatment effects.
The small sample size necessarily limits confidence about this new device’s effectiveness. But if these findings are confirmed in future studies, coronary-sinus–reducing therapy may become a welcome and necessary addition to our options to improve quality of life for patients with refractory angina.
Christopher B. Granger, M.D., is at Duke Clinical Research Institute, Durham N.C. He reported numerous ties to industry sources. Bernard J. Gersh, M.D., D.Phil., is in the department of cardiovascular diseases at the Mayo Clinic, Rochester Minn. He reported having no financial disclosures. Dr. Granger and Dr. Gersh made these remarks in an editorial accompanying Dr. Verheye’s report (N. Engl. J. Med. 2015 Feb. 5 [doi:10.1056/NEJMe1413941]).

|
Dr. Bernard Gersh |
Even though this trial was of high quality and well performed, the total number of patients in both study groups who had a positive outcome is only 26, which is not sufficient for a reliable estimation of modest treatment effects.
The small sample size necessarily limits confidence about this new device’s effectiveness. But if these findings are confirmed in future studies, coronary-sinus–reducing therapy may become a welcome and necessary addition to our options to improve quality of life for patients with refractory angina.
Christopher B. Granger, M.D., is at Duke Clinical Research Institute, Durham N.C. He reported numerous ties to industry sources. Bernard J. Gersh, M.D., D.Phil., is in the department of cardiovascular diseases at the Mayo Clinic, Rochester Minn. He reported having no financial disclosures. Dr. Granger and Dr. Gersh made these remarks in an editorial accompanying Dr. Verheye’s report (N. Engl. J. Med. 2015 Feb. 5 [doi:10.1056/NEJMe1413941]).
A new endoluminal device that reduces the coronary sinus significantly improved refractory angina in patients with advanced coronary artery disease who weren’t candidates for revascularization in a small phase II clinical trial. The results were published online Feb. 5 in the New England Journal of Medicine.
The international, randomized, double-blind study, sponsored by Neovasc, maker of the Reducer device, involved 104 patients enrolled who had Canadian Cardiovascular Society (CCS) class III or IV angina despite maximal medical therapy, evidence of reversible myocardial ischemia, and a left ventricular ejection fraction over 25%. These participants (mean age 68 years; range, 35-87 years) underwent right heart catheterization with angiography of the coronary sinus to determine whether the anatomy was suitable for implantation. Then 52 underwent implantation (the treatment group) and 52 underwent a sham procedure (the control group), and all were followed for up to 3 years, said Dr. Stefan Verheye of Antwerp (Belgium) Cardiovascular Center and his associates.
The stainless steel device is balloon expandable and shaped like an hourglass. It creates a focal narrowing that increases pressure in the coronary sinus, which is thought to recruit coronary collateral blood flow, redistributing blood from the less-ischemic epicardium to the ischemic endocardium. The primary efficacy end point of the trial – the number of patients whose angina improved by two or more CCS classes within 6 months – was achieved by more than twice as many patients in the treatment group (18, or 35%) as in the control group (8, or 15%).
A total of 37 patients (71%) who received active treatment showed an improvement of at least one CCS class, compared with 22 (42%) of those who received the sham treatment. Quality of life, as measured by Seattle Angina Questionnaire score, improved by 17.6 out of a possible 100 points for active treatment, compared with 7.6 points for sham treatment. And exercise duration improved by 1 minute (13%) for active treatment, compared with 4 seconds (1%) for sham treatment, Dr. Verheye and his associates said (N. Engl. J. Med. 2015 Feb. 5 [doi:10.1056/NEJMoa1402556]). “Our study was not statistically powered to detect an improvement in ischemia by means of objective measures such as stress testing or wall-motion index. A larger trial would be necessary to show such a benefit,” they noted.
There were 76 adverse events in patients who received the implant and 93 in the control group; the numbers of serious adverse events in this high-risk cohort were 10 and 24, respectively. This included one periprocedural MI that occurred in a patient receiving active treatment. Among 36 of the patients who received the implant and later underwent CT angiography, none showed any evidence of device migration or occlusion, the investigators added.
This study was supported by Neovasc, manufacturer of the Reducer device. Dr. Verheye reported having no financial disclosures. His associates reported ties to Neovasc, Abbott, Boston Scientific, Novo Nordisk, Eli Lilly, and Zealand Pharma.
A new endoluminal device that reduces the coronary sinus significantly improved refractory angina in patients with advanced coronary artery disease who weren’t candidates for revascularization in a small phase II clinical trial. The results were published online Feb. 5 in the New England Journal of Medicine.
The international, randomized, double-blind study, sponsored by Neovasc, maker of the Reducer device, involved 104 patients enrolled who had Canadian Cardiovascular Society (CCS) class III or IV angina despite maximal medical therapy, evidence of reversible myocardial ischemia, and a left ventricular ejection fraction over 25%. These participants (mean age 68 years; range, 35-87 years) underwent right heart catheterization with angiography of the coronary sinus to determine whether the anatomy was suitable for implantation. Then 52 underwent implantation (the treatment group) and 52 underwent a sham procedure (the control group), and all were followed for up to 3 years, said Dr. Stefan Verheye of Antwerp (Belgium) Cardiovascular Center and his associates.
The stainless steel device is balloon expandable and shaped like an hourglass. It creates a focal narrowing that increases pressure in the coronary sinus, which is thought to recruit coronary collateral blood flow, redistributing blood from the less-ischemic epicardium to the ischemic endocardium. The primary efficacy end point of the trial – the number of patients whose angina improved by two or more CCS classes within 6 months – was achieved by more than twice as many patients in the treatment group (18, or 35%) as in the control group (8, or 15%).
A total of 37 patients (71%) who received active treatment showed an improvement of at least one CCS class, compared with 22 (42%) of those who received the sham treatment. Quality of life, as measured by Seattle Angina Questionnaire score, improved by 17.6 out of a possible 100 points for active treatment, compared with 7.6 points for sham treatment. And exercise duration improved by 1 minute (13%) for active treatment, compared with 4 seconds (1%) for sham treatment, Dr. Verheye and his associates said (N. Engl. J. Med. 2015 Feb. 5 [doi:10.1056/NEJMoa1402556]). “Our study was not statistically powered to detect an improvement in ischemia by means of objective measures such as stress testing or wall-motion index. A larger trial would be necessary to show such a benefit,” they noted.
There were 76 adverse events in patients who received the implant and 93 in the control group; the numbers of serious adverse events in this high-risk cohort were 10 and 24, respectively. This included one periprocedural MI that occurred in a patient receiving active treatment. Among 36 of the patients who received the implant and later underwent CT angiography, none showed any evidence of device migration or occlusion, the investigators added.
This study was supported by Neovasc, manufacturer of the Reducer device. Dr. Verheye reported having no financial disclosures. His associates reported ties to Neovasc, Abbott, Boston Scientific, Novo Nordisk, Eli Lilly, and Zealand Pharma.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: A new endoluminal device inserted into the coronary sinus creates focal pressure that relieves refractory angina in patients who can’t undergo revascularization.
Major finding: The number of patients whose angina improved by two or more CCS classes within 6 months was achieved by more than twice as many patients in the treatment group (18, or 35%) than in the control group (8, or 15%).
Data source: A small, multicenter, randomized, sham-controlled phase II clinical trial involving 104 patients with refractory, severe angina who were followed for 3 years.
Disclosures: This study was supported by Neovasc, manufacturer of the Reducer device. Dr. Verheye reported having no financial disclosures; his associates reported ties to Neovasc, Abbott, Boston Scientific, Novo Nordisk, Eli Lilly, and Zealand Pharma.
The private-academic surgeon salary gap: Would you pick academia if you stood to lose $1.3 million?
LAKE BUENA VISTA, FLA. – Academic surgeons earn an average of 10% or $1.3 million less in gross income across their lifetime than surgeons in private practice, an analysis shows.
Some surgical specialties fare better than others, with academic neurosurgeons having the largest reduction in gross income at $4.2 million (-24.2%), while academic pediatric surgeons earn $238,376 more (1.53%) than their private practice counterparts. They were the only ones to do so.
Several academic surgical specialties did not make the 10% average including trauma surgeons whose lifetime earnings were down 12% or $2.4 million, vascular surgeons at 13.8% or $1.7 million, and surgical oncologists at 12.2% or $1.3 million.
“The concern that we have is that the academic surgeons are where the education of the future lies,” lead study author Dr. Joseph Martin Lopez said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma (EAST).
Every year a new class of surgeons is faced with the question of academic practice or private practice, but they are also struggling with increasing student loan debt and longer training as more surgical residents elect to enter fellowship rather than general practice. This growing financial liability coupled with declining physician reimbursement could rapidly shift physician practices and thus threaten the fiscal viability of certain surgical fields or academic surgical careers.
“The more financially irresponsible you make it to become an academic surgeon, the more we put at risk our current mode of training,” Dr. Lopez of Wake Forest University in Winston-Salem, N.C., said.
To account for additional factors outside gross income, the investigators ran the numbers using a second analysis, a net present value calculation, however, and came up with roughly the same salary gap to contend with.
Net present value (NPV) calculations are commonly used in business to calculate the profitability of an investment and also have been used in the medical field to gauge return on investment for various careers. The NPV calculation accounts for positive and negative cash flows over the entire length of a career, using in this case, a 5% discount rate and adjusting for inflation, Dr. Lopez explained.
Both the lifetime gross income and 5% NPV calculation used data from the Medical Group Management Association’s 2012 physician salary report, the 2012 Association of American Medical Colleges physician salary report, and the AAMC database for residency and fellow salary.
The NPV assumed a career length of 37-39 years, based on a retirement age of 65 years for all specialties. Positive cash flows included annual salary less federal income tax. Negative cash flows included the average principal for student loans, according to the AAMC, and interest at 5%, the average for the three largest student loan lenders in 2014, he said. Student loan repayment was calculated for a fixed-rate loan to be paid over 25 years beginning after residency or any required fellowship.
The average reduction in 5% NPV across surgical specialties for an academic surgeon versus a privately employed surgeon was 12.8% or $246,499, Dr. Lopez said.
Once again, academic neurosurgeons had the largest reduction in 5% NPV at 25.5% or a loss of $619,681, followed closely by trauma surgeons (23% or $381,179) and surgical oncologists (16.3% or $256,373). Academic pediatric surgeons had the smallest reduction in 5% NPV at 4.2% or $88,827.
During a discussion of the provocative poster, attendees questioned whether it was fair to say that private surgeons make more money without acknowledging the risk they face, compared with surgeons employed in an academic setting.
Dr. Lopez countered that increasingly, even private surgeons are no longer truly private surgeons.
“More and more surgical groups are being bought up by hospitals, and even the private surgical groups are being bought up by hospitals, which does stabilize your income to some extent,” he said. “We all still have RVU goals to meet and RVU incentives that make it so you can get paid a little more, but it’s something that’s a consideration. It is a risk-reward to be a private surgeon. Depending on how your contract is structured or how your group decides to pay the partners, it may be that if you don’t take very much call or take that many cases, you’ll end up on the short end of the stick.”
Dr. Ben L. Zarzaur, a general surgeon at Indiana University in Indianapolis who comoderated the poster discussion, pointed out that market pressures unaccounted for in the model can dramatically influence a surgeon’s salary over a lifetime.
Dr. Lopez agreed, citing how the increasing number of stent placements by cardiologists, for example, has impacted the bottom line of cardiothoracic surgeons. The NPV calculation was specifically used, however, because it gets at market forces such as inflation and return on investment, not addressed by gross income figures alone.
Finally, Dr. Zarzaur turned and asked the relatively young crowd what they would do if offered $600,000 a year, but had to work 110 hours a week or could get $250,000 and work only 40 hours a week. Most responded that they’d choose the former to repay their student loans and then switch to the lower-paying position. Responders made much of job satisfaction, work-life balance, and the ability of surgeons in academic practice to take time away from clinical work to conduct research, their ready access to continuing medical education, and their ability to educate the next generation of surgeons.
“Any time we see this academic-private disparity, you have to think about these secondary gains,” Dr. Zarzaur said. “This is really interesting work. It gets into why we choose what we do, why we’d take $600,000, work 110 hours a week, and get our rear ends kicked. The flip side is, if I saw this, why would you ever go into academics? But people still choose to do it. I’m in academics so there’s a bias, but we choose to do it anyway up to a point. I don’t know where that point is, but up to a point we do.”
LAKE BUENA VISTA, FLA. – Academic surgeons earn an average of 10% or $1.3 million less in gross income across their lifetime than surgeons in private practice, an analysis shows.
Some surgical specialties fare better than others, with academic neurosurgeons having the largest reduction in gross income at $4.2 million (-24.2%), while academic pediatric surgeons earn $238,376 more (1.53%) than their private practice counterparts. They were the only ones to do so.
Several academic surgical specialties did not make the 10% average including trauma surgeons whose lifetime earnings were down 12% or $2.4 million, vascular surgeons at 13.8% or $1.7 million, and surgical oncologists at 12.2% or $1.3 million.
“The concern that we have is that the academic surgeons are where the education of the future lies,” lead study author Dr. Joseph Martin Lopez said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma (EAST).
Every year a new class of surgeons is faced with the question of academic practice or private practice, but they are also struggling with increasing student loan debt and longer training as more surgical residents elect to enter fellowship rather than general practice. This growing financial liability coupled with declining physician reimbursement could rapidly shift physician practices and thus threaten the fiscal viability of certain surgical fields or academic surgical careers.
“The more financially irresponsible you make it to become an academic surgeon, the more we put at risk our current mode of training,” Dr. Lopez of Wake Forest University in Winston-Salem, N.C., said.
To account for additional factors outside gross income, the investigators ran the numbers using a second analysis, a net present value calculation, however, and came up with roughly the same salary gap to contend with.
Net present value (NPV) calculations are commonly used in business to calculate the profitability of an investment and also have been used in the medical field to gauge return on investment for various careers. The NPV calculation accounts for positive and negative cash flows over the entire length of a career, using in this case, a 5% discount rate and adjusting for inflation, Dr. Lopez explained.
Both the lifetime gross income and 5% NPV calculation used data from the Medical Group Management Association’s 2012 physician salary report, the 2012 Association of American Medical Colleges physician salary report, and the AAMC database for residency and fellow salary.
The NPV assumed a career length of 37-39 years, based on a retirement age of 65 years for all specialties. Positive cash flows included annual salary less federal income tax. Negative cash flows included the average principal for student loans, according to the AAMC, and interest at 5%, the average for the three largest student loan lenders in 2014, he said. Student loan repayment was calculated for a fixed-rate loan to be paid over 25 years beginning after residency or any required fellowship.
The average reduction in 5% NPV across surgical specialties for an academic surgeon versus a privately employed surgeon was 12.8% or $246,499, Dr. Lopez said.
Once again, academic neurosurgeons had the largest reduction in 5% NPV at 25.5% or a loss of $619,681, followed closely by trauma surgeons (23% or $381,179) and surgical oncologists (16.3% or $256,373). Academic pediatric surgeons had the smallest reduction in 5% NPV at 4.2% or $88,827.
During a discussion of the provocative poster, attendees questioned whether it was fair to say that private surgeons make more money without acknowledging the risk they face, compared with surgeons employed in an academic setting.
Dr. Lopez countered that increasingly, even private surgeons are no longer truly private surgeons.
“More and more surgical groups are being bought up by hospitals, and even the private surgical groups are being bought up by hospitals, which does stabilize your income to some extent,” he said. “We all still have RVU goals to meet and RVU incentives that make it so you can get paid a little more, but it’s something that’s a consideration. It is a risk-reward to be a private surgeon. Depending on how your contract is structured or how your group decides to pay the partners, it may be that if you don’t take very much call or take that many cases, you’ll end up on the short end of the stick.”
Dr. Ben L. Zarzaur, a general surgeon at Indiana University in Indianapolis who comoderated the poster discussion, pointed out that market pressures unaccounted for in the model can dramatically influence a surgeon’s salary over a lifetime.
Dr. Lopez agreed, citing how the increasing number of stent placements by cardiologists, for example, has impacted the bottom line of cardiothoracic surgeons. The NPV calculation was specifically used, however, because it gets at market forces such as inflation and return on investment, not addressed by gross income figures alone.
Finally, Dr. Zarzaur turned and asked the relatively young crowd what they would do if offered $600,000 a year, but had to work 110 hours a week or could get $250,000 and work only 40 hours a week. Most responded that they’d choose the former to repay their student loans and then switch to the lower-paying position. Responders made much of job satisfaction, work-life balance, and the ability of surgeons in academic practice to take time away from clinical work to conduct research, their ready access to continuing medical education, and their ability to educate the next generation of surgeons.
“Any time we see this academic-private disparity, you have to think about these secondary gains,” Dr. Zarzaur said. “This is really interesting work. It gets into why we choose what we do, why we’d take $600,000, work 110 hours a week, and get our rear ends kicked. The flip side is, if I saw this, why would you ever go into academics? But people still choose to do it. I’m in academics so there’s a bias, but we choose to do it anyway up to a point. I don’t know where that point is, but up to a point we do.”
LAKE BUENA VISTA, FLA. – Academic surgeons earn an average of 10% or $1.3 million less in gross income across their lifetime than surgeons in private practice, an analysis shows.
Some surgical specialties fare better than others, with academic neurosurgeons having the largest reduction in gross income at $4.2 million (-24.2%), while academic pediatric surgeons earn $238,376 more (1.53%) than their private practice counterparts. They were the only ones to do so.
Several academic surgical specialties did not make the 10% average including trauma surgeons whose lifetime earnings were down 12% or $2.4 million, vascular surgeons at 13.8% or $1.7 million, and surgical oncologists at 12.2% or $1.3 million.
“The concern that we have is that the academic surgeons are where the education of the future lies,” lead study author Dr. Joseph Martin Lopez said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma (EAST).
Every year a new class of surgeons is faced with the question of academic practice or private practice, but they are also struggling with increasing student loan debt and longer training as more surgical residents elect to enter fellowship rather than general practice. This growing financial liability coupled with declining physician reimbursement could rapidly shift physician practices and thus threaten the fiscal viability of certain surgical fields or academic surgical careers.
“The more financially irresponsible you make it to become an academic surgeon, the more we put at risk our current mode of training,” Dr. Lopez of Wake Forest University in Winston-Salem, N.C., said.
To account for additional factors outside gross income, the investigators ran the numbers using a second analysis, a net present value calculation, however, and came up with roughly the same salary gap to contend with.
Net present value (NPV) calculations are commonly used in business to calculate the profitability of an investment and also have been used in the medical field to gauge return on investment for various careers. The NPV calculation accounts for positive and negative cash flows over the entire length of a career, using in this case, a 5% discount rate and adjusting for inflation, Dr. Lopez explained.
Both the lifetime gross income and 5% NPV calculation used data from the Medical Group Management Association’s 2012 physician salary report, the 2012 Association of American Medical Colleges physician salary report, and the AAMC database for residency and fellow salary.
The NPV assumed a career length of 37-39 years, based on a retirement age of 65 years for all specialties. Positive cash flows included annual salary less federal income tax. Negative cash flows included the average principal for student loans, according to the AAMC, and interest at 5%, the average for the three largest student loan lenders in 2014, he said. Student loan repayment was calculated for a fixed-rate loan to be paid over 25 years beginning after residency or any required fellowship.
The average reduction in 5% NPV across surgical specialties for an academic surgeon versus a privately employed surgeon was 12.8% or $246,499, Dr. Lopez said.
Once again, academic neurosurgeons had the largest reduction in 5% NPV at 25.5% or a loss of $619,681, followed closely by trauma surgeons (23% or $381,179) and surgical oncologists (16.3% or $256,373). Academic pediatric surgeons had the smallest reduction in 5% NPV at 4.2% or $88,827.
During a discussion of the provocative poster, attendees questioned whether it was fair to say that private surgeons make more money without acknowledging the risk they face, compared with surgeons employed in an academic setting.
Dr. Lopez countered that increasingly, even private surgeons are no longer truly private surgeons.
“More and more surgical groups are being bought up by hospitals, and even the private surgical groups are being bought up by hospitals, which does stabilize your income to some extent,” he said. “We all still have RVU goals to meet and RVU incentives that make it so you can get paid a little more, but it’s something that’s a consideration. It is a risk-reward to be a private surgeon. Depending on how your contract is structured or how your group decides to pay the partners, it may be that if you don’t take very much call or take that many cases, you’ll end up on the short end of the stick.”
Dr. Ben L. Zarzaur, a general surgeon at Indiana University in Indianapolis who comoderated the poster discussion, pointed out that market pressures unaccounted for in the model can dramatically influence a surgeon’s salary over a lifetime.
Dr. Lopez agreed, citing how the increasing number of stent placements by cardiologists, for example, has impacted the bottom line of cardiothoracic surgeons. The NPV calculation was specifically used, however, because it gets at market forces such as inflation and return on investment, not addressed by gross income figures alone.
Finally, Dr. Zarzaur turned and asked the relatively young crowd what they would do if offered $600,000 a year, but had to work 110 hours a week or could get $250,000 and work only 40 hours a week. Most responded that they’d choose the former to repay their student loans and then switch to the lower-paying position. Responders made much of job satisfaction, work-life balance, and the ability of surgeons in academic practice to take time away from clinical work to conduct research, their ready access to continuing medical education, and their ability to educate the next generation of surgeons.
“Any time we see this academic-private disparity, you have to think about these secondary gains,” Dr. Zarzaur said. “This is really interesting work. It gets into why we choose what we do, why we’d take $600,000, work 110 hours a week, and get our rear ends kicked. The flip side is, if I saw this, why would you ever go into academics? But people still choose to do it. I’m in academics so there’s a bias, but we choose to do it anyway up to a point. I don’t know where that point is, but up to a point we do.”
AT THE EAST SCIENTIFIC ASSEMBLY
Key clinical point: Whether calculated as gross lifetime income or 5% net present value, a salary disparity exists between academic and private practice surgeons.
Major finding: Academic surgeons earn an average of 10% or $1.3 million less in gross lifetime income than surgeons in private practice.
Data source: Salary analysis and net present value calculation.
Disclosures: Dr. Lopez and his coauthors reported having no financial disclosures. Dr. Zarzaur disclosed honorarium from and serving as an advisor for Merck.
Medicare at 50: Hassles lead to doctor opt-outs, new business models
After suffering through reduced reimbursement year after year and encountering government rules that caused her to restrict the way she practiced, Atlanta otolaryngologist Dr. Elaina George was fed up with Medicare and what seemed its endless red tape.
“We found that the time it took to be reimbursed was much longer than the average commercial payer,” Dr. George said in an interview. Because of global period payment rules, “several times, we got paid nothing. [Medicare] stopped me from doing the things I was trained to do. I stopped doing head and neck surgeries because it wasn’t cost effective.”
Instead of complaining or appealing payment decisions, Dr. George made a more drastic move. She dropped Medicare altogether. Nearly a decade later, the solo practitioner continues to opt out of Medicare, obtaining payment through some commercial insurers and direct pay contracts with patients.
“Direct pay is going to be the future, and anybody who can figure out how to work around the (traditional) insurance model is going to save money,” said Dr. George, an advisory council member of Project 21 black leadership network, an initiative of the National Center for Public Policy Research, a conservative think tank and policy institute.
Dr. George is far from alone. She is part of a growing vocal minority that says dropping Medicare is the only remedy to ongoing payment reductions, extended waits for reimbursement, audits, and growing regulations, such as meaningful use. The feasibility of leaving the program however, depends on specialty, geographic location, and patient base, physician leaders say.
Measuring the number of doctors who opt out of Medicare isn’t easy. The U.S. Department of Health & Human Service’s Office of Inspector General (OIG) said in a 2012 letter to the Centers for Medicare & Medicaid Services (CMS) that CMS does not maintain sufficient data regarding physicians who opt out of Medicare, and therefore the OIG could not complete an analysis into reasons doctors choose not to participate.
However, federal data released to the Wall Street Journal in 2013 show that 9,539 physicians who previously accepted Medicare opted out of the program in 2012, up from 3,700 in 2009. The CMS had never before released annual opt-out figures, and the data cannot be found on CMS’ website. A CMS spokeswoman declined comment for this story.
Despite the drop-out figures, government statistics paint a picture of growing physician participation in Medicare. A 2014 CMS report shows that a total of 1,226,728 health providers of all specialties participated in Medicare in 2013, up from 1,089,306 in 2012, according to federal data. (The report noted physicians may have been counted in more than one specialty.) There were 219,536 primary care physicians/suppliers who treated Medicare patients in 2013, up from 215,919 in 2012.
But the stats on physician participation do not tell the whole story, said Dr. Austin King, president of the Texas Medical Association and an Abilene otolaryngologist. While many physicians take Medicare patients, he notes that a large portion do not accept new Medicare patients. In Abilene for example, Dr. King said he knows of no internists who accept new Medicare patients. The dilemma means as more of the population reaches Medicare age, there could be fewer doctors to treat them.
“It seems like the government is almost making it more difficult for physicians to treat Medicare patients,” said Dr. King, who limits the number of Medicare patients he treats. “It’s difficult for many reasons, but what I hear most are complaints about the enormous amount of red tape and bureaucracy associated with Medicare.”
For family physician Dr. Andrew Merritt of Marcellus, N.Y., the decision not to accept new Medicare patients made sense 15 years ago and still does today. Medicare is one of the lowest payers in the Marcellus area, he said, second only to Medicaid.
“The trends were there” back in 2000 when he made the decision, he said. “For us, it was fees and regulations, and the regulations have gotten worse.”
The government disputes that more doctors are rejecting new Medicare patients. The percentage of all office-based physicians who report accepting new Medicare patients has not changed significantly between 2005 and 2012, with 87.9% of physicians accepting new Medicare patients in 2005 and 90.7% accepting new patients in 2012, according to a 2013 issue brief from the HHS Office of the Assistant Secretary for Planning and Evaluation. The percentage of doctors accepting new Medicare patients in 2011-2012 is slightly higher than the percentage accepting new private insurance – about 86% of physicians in 2012 accepted new privately insured patients, according to the brief.
“To the extent that there may have been a very small increase in the number of providers ‘opting out,’ that increase has been mitigated by an increase in the share of other physicians who accept new Medicare patients,” according to the issue brief. “Further, the total number of providers participating in and billing Medicare has steadily increased since 2007.”
Dr. Merritt notes while opting out of Medicare might work for some physicians, it’s not practical for all. For instance, in his area, most psychiatrists have opted out of Medicare. “In primary care, it becomes difficult,” he said. “You have to see a lot of people, and it depends on the level of competition.”
Similarly, where a doctor practices impacts Medicare participation, said Dr. Theodore Mazer, a San Diego otolaryngologist and house of delegates speaker at the California Medical Association.
“In my area, Medicare payment is kind of a gold standard,” he said in an interview. “There’s not much that pays better than that. If I can’t pay costs with Medicare, that means I can’t pay costs with any carrier in the area.”
The Association of American Physicians and Surgeons (AAPS) on the other hand, believes with some assessment and planning, most physicians could successfully opt out of Medicare. The AAPS has been hosting a series of workshops and presentations on how to drop Medicare and move to a cash-only practice.
When considering the transition, physicians need to examine their patient base, practice demographics and costs with and without Medicare, said Dr. Lawrence Huntoon, editor-in-chief of the Journal of the Association of American Physicians and Surgeons. He notes a significant amount of professional time and money is often spent on Medicare-related paperwork and administrative burdens.
Dr. Huntoon stopped participating in Medicare in 2004. His practice works with no third-party insurers, including Medicare, and contracts directly with patients for payment.
“I’m very happy with it, and I’ve never regretted it,” he said in an interview. “You just don’t have someone constantly interfering with the care you’re trying to provide.”
On Twitter @legal_med
After suffering through reduced reimbursement year after year and encountering government rules that caused her to restrict the way she practiced, Atlanta otolaryngologist Dr. Elaina George was fed up with Medicare and what seemed its endless red tape.
“We found that the time it took to be reimbursed was much longer than the average commercial payer,” Dr. George said in an interview. Because of global period payment rules, “several times, we got paid nothing. [Medicare] stopped me from doing the things I was trained to do. I stopped doing head and neck surgeries because it wasn’t cost effective.”
Instead of complaining or appealing payment decisions, Dr. George made a more drastic move. She dropped Medicare altogether. Nearly a decade later, the solo practitioner continues to opt out of Medicare, obtaining payment through some commercial insurers and direct pay contracts with patients.
“Direct pay is going to be the future, and anybody who can figure out how to work around the (traditional) insurance model is going to save money,” said Dr. George, an advisory council member of Project 21 black leadership network, an initiative of the National Center for Public Policy Research, a conservative think tank and policy institute.
Dr. George is far from alone. She is part of a growing vocal minority that says dropping Medicare is the only remedy to ongoing payment reductions, extended waits for reimbursement, audits, and growing regulations, such as meaningful use. The feasibility of leaving the program however, depends on specialty, geographic location, and patient base, physician leaders say.
Measuring the number of doctors who opt out of Medicare isn’t easy. The U.S. Department of Health & Human Service’s Office of Inspector General (OIG) said in a 2012 letter to the Centers for Medicare & Medicaid Services (CMS) that CMS does not maintain sufficient data regarding physicians who opt out of Medicare, and therefore the OIG could not complete an analysis into reasons doctors choose not to participate.
However, federal data released to the Wall Street Journal in 2013 show that 9,539 physicians who previously accepted Medicare opted out of the program in 2012, up from 3,700 in 2009. The CMS had never before released annual opt-out figures, and the data cannot be found on CMS’ website. A CMS spokeswoman declined comment for this story.
Despite the drop-out figures, government statistics paint a picture of growing physician participation in Medicare. A 2014 CMS report shows that a total of 1,226,728 health providers of all specialties participated in Medicare in 2013, up from 1,089,306 in 2012, according to federal data. (The report noted physicians may have been counted in more than one specialty.) There were 219,536 primary care physicians/suppliers who treated Medicare patients in 2013, up from 215,919 in 2012.
But the stats on physician participation do not tell the whole story, said Dr. Austin King, president of the Texas Medical Association and an Abilene otolaryngologist. While many physicians take Medicare patients, he notes that a large portion do not accept new Medicare patients. In Abilene for example, Dr. King said he knows of no internists who accept new Medicare patients. The dilemma means as more of the population reaches Medicare age, there could be fewer doctors to treat them.
“It seems like the government is almost making it more difficult for physicians to treat Medicare patients,” said Dr. King, who limits the number of Medicare patients he treats. “It’s difficult for many reasons, but what I hear most are complaints about the enormous amount of red tape and bureaucracy associated with Medicare.”
For family physician Dr. Andrew Merritt of Marcellus, N.Y., the decision not to accept new Medicare patients made sense 15 years ago and still does today. Medicare is one of the lowest payers in the Marcellus area, he said, second only to Medicaid.
“The trends were there” back in 2000 when he made the decision, he said. “For us, it was fees and regulations, and the regulations have gotten worse.”
The government disputes that more doctors are rejecting new Medicare patients. The percentage of all office-based physicians who report accepting new Medicare patients has not changed significantly between 2005 and 2012, with 87.9% of physicians accepting new Medicare patients in 2005 and 90.7% accepting new patients in 2012, according to a 2013 issue brief from the HHS Office of the Assistant Secretary for Planning and Evaluation. The percentage of doctors accepting new Medicare patients in 2011-2012 is slightly higher than the percentage accepting new private insurance – about 86% of physicians in 2012 accepted new privately insured patients, according to the brief.
“To the extent that there may have been a very small increase in the number of providers ‘opting out,’ that increase has been mitigated by an increase in the share of other physicians who accept new Medicare patients,” according to the issue brief. “Further, the total number of providers participating in and billing Medicare has steadily increased since 2007.”
Dr. Merritt notes while opting out of Medicare might work for some physicians, it’s not practical for all. For instance, in his area, most psychiatrists have opted out of Medicare. “In primary care, it becomes difficult,” he said. “You have to see a lot of people, and it depends on the level of competition.”
Similarly, where a doctor practices impacts Medicare participation, said Dr. Theodore Mazer, a San Diego otolaryngologist and house of delegates speaker at the California Medical Association.
“In my area, Medicare payment is kind of a gold standard,” he said in an interview. “There’s not much that pays better than that. If I can’t pay costs with Medicare, that means I can’t pay costs with any carrier in the area.”
The Association of American Physicians and Surgeons (AAPS) on the other hand, believes with some assessment and planning, most physicians could successfully opt out of Medicare. The AAPS has been hosting a series of workshops and presentations on how to drop Medicare and move to a cash-only practice.
When considering the transition, physicians need to examine their patient base, practice demographics and costs with and without Medicare, said Dr. Lawrence Huntoon, editor-in-chief of the Journal of the Association of American Physicians and Surgeons. He notes a significant amount of professional time and money is often spent on Medicare-related paperwork and administrative burdens.
Dr. Huntoon stopped participating in Medicare in 2004. His practice works with no third-party insurers, including Medicare, and contracts directly with patients for payment.
“I’m very happy with it, and I’ve never regretted it,” he said in an interview. “You just don’t have someone constantly interfering with the care you’re trying to provide.”
On Twitter @legal_med
After suffering through reduced reimbursement year after year and encountering government rules that caused her to restrict the way she practiced, Atlanta otolaryngologist Dr. Elaina George was fed up with Medicare and what seemed its endless red tape.
“We found that the time it took to be reimbursed was much longer than the average commercial payer,” Dr. George said in an interview. Because of global period payment rules, “several times, we got paid nothing. [Medicare] stopped me from doing the things I was trained to do. I stopped doing head and neck surgeries because it wasn’t cost effective.”
Instead of complaining or appealing payment decisions, Dr. George made a more drastic move. She dropped Medicare altogether. Nearly a decade later, the solo practitioner continues to opt out of Medicare, obtaining payment through some commercial insurers and direct pay contracts with patients.
“Direct pay is going to be the future, and anybody who can figure out how to work around the (traditional) insurance model is going to save money,” said Dr. George, an advisory council member of Project 21 black leadership network, an initiative of the National Center for Public Policy Research, a conservative think tank and policy institute.
Dr. George is far from alone. She is part of a growing vocal minority that says dropping Medicare is the only remedy to ongoing payment reductions, extended waits for reimbursement, audits, and growing regulations, such as meaningful use. The feasibility of leaving the program however, depends on specialty, geographic location, and patient base, physician leaders say.
Measuring the number of doctors who opt out of Medicare isn’t easy. The U.S. Department of Health & Human Service’s Office of Inspector General (OIG) said in a 2012 letter to the Centers for Medicare & Medicaid Services (CMS) that CMS does not maintain sufficient data regarding physicians who opt out of Medicare, and therefore the OIG could not complete an analysis into reasons doctors choose not to participate.
However, federal data released to the Wall Street Journal in 2013 show that 9,539 physicians who previously accepted Medicare opted out of the program in 2012, up from 3,700 in 2009. The CMS had never before released annual opt-out figures, and the data cannot be found on CMS’ website. A CMS spokeswoman declined comment for this story.
Despite the drop-out figures, government statistics paint a picture of growing physician participation in Medicare. A 2014 CMS report shows that a total of 1,226,728 health providers of all specialties participated in Medicare in 2013, up from 1,089,306 in 2012, according to federal data. (The report noted physicians may have been counted in more than one specialty.) There were 219,536 primary care physicians/suppliers who treated Medicare patients in 2013, up from 215,919 in 2012.
But the stats on physician participation do not tell the whole story, said Dr. Austin King, president of the Texas Medical Association and an Abilene otolaryngologist. While many physicians take Medicare patients, he notes that a large portion do not accept new Medicare patients. In Abilene for example, Dr. King said he knows of no internists who accept new Medicare patients. The dilemma means as more of the population reaches Medicare age, there could be fewer doctors to treat them.
“It seems like the government is almost making it more difficult for physicians to treat Medicare patients,” said Dr. King, who limits the number of Medicare patients he treats. “It’s difficult for many reasons, but what I hear most are complaints about the enormous amount of red tape and bureaucracy associated with Medicare.”
For family physician Dr. Andrew Merritt of Marcellus, N.Y., the decision not to accept new Medicare patients made sense 15 years ago and still does today. Medicare is one of the lowest payers in the Marcellus area, he said, second only to Medicaid.
“The trends were there” back in 2000 when he made the decision, he said. “For us, it was fees and regulations, and the regulations have gotten worse.”
The government disputes that more doctors are rejecting new Medicare patients. The percentage of all office-based physicians who report accepting new Medicare patients has not changed significantly between 2005 and 2012, with 87.9% of physicians accepting new Medicare patients in 2005 and 90.7% accepting new patients in 2012, according to a 2013 issue brief from the HHS Office of the Assistant Secretary for Planning and Evaluation. The percentage of doctors accepting new Medicare patients in 2011-2012 is slightly higher than the percentage accepting new private insurance – about 86% of physicians in 2012 accepted new privately insured patients, according to the brief.
“To the extent that there may have been a very small increase in the number of providers ‘opting out,’ that increase has been mitigated by an increase in the share of other physicians who accept new Medicare patients,” according to the issue brief. “Further, the total number of providers participating in and billing Medicare has steadily increased since 2007.”
Dr. Merritt notes while opting out of Medicare might work for some physicians, it’s not practical for all. For instance, in his area, most psychiatrists have opted out of Medicare. “In primary care, it becomes difficult,” he said. “You have to see a lot of people, and it depends on the level of competition.”
Similarly, where a doctor practices impacts Medicare participation, said Dr. Theodore Mazer, a San Diego otolaryngologist and house of delegates speaker at the California Medical Association.
“In my area, Medicare payment is kind of a gold standard,” he said in an interview. “There’s not much that pays better than that. If I can’t pay costs with Medicare, that means I can’t pay costs with any carrier in the area.”
The Association of American Physicians and Surgeons (AAPS) on the other hand, believes with some assessment and planning, most physicians could successfully opt out of Medicare. The AAPS has been hosting a series of workshops and presentations on how to drop Medicare and move to a cash-only practice.
When considering the transition, physicians need to examine their patient base, practice demographics and costs with and without Medicare, said Dr. Lawrence Huntoon, editor-in-chief of the Journal of the Association of American Physicians and Surgeons. He notes a significant amount of professional time and money is often spent on Medicare-related paperwork and administrative burdens.
Dr. Huntoon stopped participating in Medicare in 2004. His practice works with no third-party insurers, including Medicare, and contracts directly with patients for payment.
“I’m very happy with it, and I’ve never regretted it,” he said in an interview. “You just don’t have someone constantly interfering with the care you’re trying to provide.”
On Twitter @legal_med
Zero coronary calcium means very low 10-year event risk
CHICAGO – Absence of coronary artery calcium upon imaging results in an impressively low cardiovascular event rate over the next 10 years regardless of an individual’s level of standard risk factors, according to prospective data from the MESA study.
In contrast, a coronary artery calcium (CAC) score of 1-10, often described as minimal CAC, nearly doubles the 10-year risk, compared with a baseline CAC score of 0.
Prior to these new 10-year data, many cardiologists considered a CAC score of 1-10 as tantamount to no CAC. Not so, Dr. Parag H. Joshi said at the American Heart Association scientific sessions.
“A CAC of 0 is presumably identifying someone without any atherosclerosis. Just the presence of minimal calcium suggests that atherosclerosis is building up. Our data suggest that among individuals with a CAC of 1-10, current smoking, elevated non-HDL cholesterol, and particularly hypertension should be treated aggressively,” said Dr. Joshi, a clinical fellow in cardiovascular diseases and prevention at Johns Hopkins University, Baltimore.
Prior studies totaling more than 50,000 subjects with a CAC score of 0 have shown very low cardiovascular event rates over 4-5 years of follow-up. However, current cardiovascular risk estimates focus on 10-year risk. This new analysis from MESA (Multi-Ethnic Study of Atherosclerosis) is the first study to provide prospective, 10-year events data, and those data are highly reassuring, he added.
MESA is a prospective, population-based cohort study. This analysis included 6,814 subjects aged 45-84 who were free of clinical cardiovascular disease at baseline, when their CAC score was determined. At that time, 3,415 participants had a CAC score of 0 and 508 had a score of 1-10.
During a median 10.3 years of follow-up, 123 cardiovascular events occurred, roughly one-third of which were nonfatal acute MIs and half of which were nonfatal strokes; the remainder were cardiovascular deaths.
The event rate was 2.9/1,000 person-years in subjects with a CAC of 0 and significantly greater at 5.5/1,000 person-years with a score of 1-10. However, since the cardiovascular risk factor profile of the zero CAC group was generally more favorable, Dr. Joshi and coinvestigators carried out a Cox proportional hazards analysis factoring in demographics, standard cardiovascular risk factors, body mass index, C-reactive protein level, and carotid intima media thickness. The adjusted 10-year event risk in the group with a CAC score of 1-10 was 1.9-fold greater than with a CAC of 0.
The highest 10-year event rate was noted in subjects with at least three of the following four risk factors at baseline: hypertension, current smoking, diabetes, and hyperlipidemia. The rate was 6.5/1,000 person-years in such individuals if they had a CAC of 0 and doubled at 13.1/1,000 person-years with a score of 1-10.
In a multivariate Cox analysis, age, smoking, and hypertension proved to be significant predictors of cardiovascular events in the group with a CAC of 0 as well as in those with a CAC of 1-10. But there was one important difference between the two groups: While the hazard ratio for cardiovascular events associated with hypertension versus no hypertension was 2.1 in subjects with a CAC of 0, the presence of hypertension in individuals with a CAC of 1-10 increased their event risk by 10.2-fold, or nearly five times greater than the risk increase associated with hypertension in persons with a CAC of 0, Dr. Joshi observed.
Non–HDL cholesterol level was predictive of cardiovascular risk in subjects with a CAC of 1-10 but not in those with a score of 0.
When actual event rates were compared with those predicted by the atherosclerotic cardiovascular disease (ASCVD) risk estimator introduced in the 2013 AHA/American College of Cardiology cholesterol guidelines, the event rate in subjects with an ASCVD 10-year risk estimate of 7.5%-15% but a CAC of 0 was just 4.4%.
Audience members noted that CAC scores didn’t do a very good job of stratifying stroke risk in MESA. That’s not surprising, since the score reflects coronary but not carotid artery calcium. But it is a limitation of CAC as a predictive tool, especially in light of the fact that strokes accounted for half of all cardiovascular events in the study.
Asked where he and his coinvestigators plan to go from here, Dr. Joshi said a randomized, controlled trial would be ideal, but to date funding isn’t available. However, the observational data from MESA and other studies suggest such a trial may not even be needed.
“Certainly the guidelines do allow for CAC scoring to be used in clinical decision making,” he noted.
The MESA study is funded by the National Heart, Lung, and Blood Institute. Dr. Joshi reported having no financial conflicts.
CHICAGO – Absence of coronary artery calcium upon imaging results in an impressively low cardiovascular event rate over the next 10 years regardless of an individual’s level of standard risk factors, according to prospective data from the MESA study.
In contrast, a coronary artery calcium (CAC) score of 1-10, often described as minimal CAC, nearly doubles the 10-year risk, compared with a baseline CAC score of 0.
Prior to these new 10-year data, many cardiologists considered a CAC score of 1-10 as tantamount to no CAC. Not so, Dr. Parag H. Joshi said at the American Heart Association scientific sessions.
“A CAC of 0 is presumably identifying someone without any atherosclerosis. Just the presence of minimal calcium suggests that atherosclerosis is building up. Our data suggest that among individuals with a CAC of 1-10, current smoking, elevated non-HDL cholesterol, and particularly hypertension should be treated aggressively,” said Dr. Joshi, a clinical fellow in cardiovascular diseases and prevention at Johns Hopkins University, Baltimore.
Prior studies totaling more than 50,000 subjects with a CAC score of 0 have shown very low cardiovascular event rates over 4-5 years of follow-up. However, current cardiovascular risk estimates focus on 10-year risk. This new analysis from MESA (Multi-Ethnic Study of Atherosclerosis) is the first study to provide prospective, 10-year events data, and those data are highly reassuring, he added.
MESA is a prospective, population-based cohort study. This analysis included 6,814 subjects aged 45-84 who were free of clinical cardiovascular disease at baseline, when their CAC score was determined. At that time, 3,415 participants had a CAC score of 0 and 508 had a score of 1-10.
During a median 10.3 years of follow-up, 123 cardiovascular events occurred, roughly one-third of which were nonfatal acute MIs and half of which were nonfatal strokes; the remainder were cardiovascular deaths.
The event rate was 2.9/1,000 person-years in subjects with a CAC of 0 and significantly greater at 5.5/1,000 person-years with a score of 1-10. However, since the cardiovascular risk factor profile of the zero CAC group was generally more favorable, Dr. Joshi and coinvestigators carried out a Cox proportional hazards analysis factoring in demographics, standard cardiovascular risk factors, body mass index, C-reactive protein level, and carotid intima media thickness. The adjusted 10-year event risk in the group with a CAC score of 1-10 was 1.9-fold greater than with a CAC of 0.
The highest 10-year event rate was noted in subjects with at least three of the following four risk factors at baseline: hypertension, current smoking, diabetes, and hyperlipidemia. The rate was 6.5/1,000 person-years in such individuals if they had a CAC of 0 and doubled at 13.1/1,000 person-years with a score of 1-10.
In a multivariate Cox analysis, age, smoking, and hypertension proved to be significant predictors of cardiovascular events in the group with a CAC of 0 as well as in those with a CAC of 1-10. But there was one important difference between the two groups: While the hazard ratio for cardiovascular events associated with hypertension versus no hypertension was 2.1 in subjects with a CAC of 0, the presence of hypertension in individuals with a CAC of 1-10 increased their event risk by 10.2-fold, or nearly five times greater than the risk increase associated with hypertension in persons with a CAC of 0, Dr. Joshi observed.
Non–HDL cholesterol level was predictive of cardiovascular risk in subjects with a CAC of 1-10 but not in those with a score of 0.
When actual event rates were compared with those predicted by the atherosclerotic cardiovascular disease (ASCVD) risk estimator introduced in the 2013 AHA/American College of Cardiology cholesterol guidelines, the event rate in subjects with an ASCVD 10-year risk estimate of 7.5%-15% but a CAC of 0 was just 4.4%.
Audience members noted that CAC scores didn’t do a very good job of stratifying stroke risk in MESA. That’s not surprising, since the score reflects coronary but not carotid artery calcium. But it is a limitation of CAC as a predictive tool, especially in light of the fact that strokes accounted for half of all cardiovascular events in the study.
Asked where he and his coinvestigators plan to go from here, Dr. Joshi said a randomized, controlled trial would be ideal, but to date funding isn’t available. However, the observational data from MESA and other studies suggest such a trial may not even be needed.
“Certainly the guidelines do allow for CAC scoring to be used in clinical decision making,” he noted.
The MESA study is funded by the National Heart, Lung, and Blood Institute. Dr. Joshi reported having no financial conflicts.
CHICAGO – Absence of coronary artery calcium upon imaging results in an impressively low cardiovascular event rate over the next 10 years regardless of an individual’s level of standard risk factors, according to prospective data from the MESA study.
In contrast, a coronary artery calcium (CAC) score of 1-10, often described as minimal CAC, nearly doubles the 10-year risk, compared with a baseline CAC score of 0.
Prior to these new 10-year data, many cardiologists considered a CAC score of 1-10 as tantamount to no CAC. Not so, Dr. Parag H. Joshi said at the American Heart Association scientific sessions.
“A CAC of 0 is presumably identifying someone without any atherosclerosis. Just the presence of minimal calcium suggests that atherosclerosis is building up. Our data suggest that among individuals with a CAC of 1-10, current smoking, elevated non-HDL cholesterol, and particularly hypertension should be treated aggressively,” said Dr. Joshi, a clinical fellow in cardiovascular diseases and prevention at Johns Hopkins University, Baltimore.
Prior studies totaling more than 50,000 subjects with a CAC score of 0 have shown very low cardiovascular event rates over 4-5 years of follow-up. However, current cardiovascular risk estimates focus on 10-year risk. This new analysis from MESA (Multi-Ethnic Study of Atherosclerosis) is the first study to provide prospective, 10-year events data, and those data are highly reassuring, he added.
MESA is a prospective, population-based cohort study. This analysis included 6,814 subjects aged 45-84 who were free of clinical cardiovascular disease at baseline, when their CAC score was determined. At that time, 3,415 participants had a CAC score of 0 and 508 had a score of 1-10.
During a median 10.3 years of follow-up, 123 cardiovascular events occurred, roughly one-third of which were nonfatal acute MIs and half of which were nonfatal strokes; the remainder were cardiovascular deaths.
The event rate was 2.9/1,000 person-years in subjects with a CAC of 0 and significantly greater at 5.5/1,000 person-years with a score of 1-10. However, since the cardiovascular risk factor profile of the zero CAC group was generally more favorable, Dr. Joshi and coinvestigators carried out a Cox proportional hazards analysis factoring in demographics, standard cardiovascular risk factors, body mass index, C-reactive protein level, and carotid intima media thickness. The adjusted 10-year event risk in the group with a CAC score of 1-10 was 1.9-fold greater than with a CAC of 0.
The highest 10-year event rate was noted in subjects with at least three of the following four risk factors at baseline: hypertension, current smoking, diabetes, and hyperlipidemia. The rate was 6.5/1,000 person-years in such individuals if they had a CAC of 0 and doubled at 13.1/1,000 person-years with a score of 1-10.
In a multivariate Cox analysis, age, smoking, and hypertension proved to be significant predictors of cardiovascular events in the group with a CAC of 0 as well as in those with a CAC of 1-10. But there was one important difference between the two groups: While the hazard ratio for cardiovascular events associated with hypertension versus no hypertension was 2.1 in subjects with a CAC of 0, the presence of hypertension in individuals with a CAC of 1-10 increased their event risk by 10.2-fold, or nearly five times greater than the risk increase associated with hypertension in persons with a CAC of 0, Dr. Joshi observed.
Non–HDL cholesterol level was predictive of cardiovascular risk in subjects with a CAC of 1-10 but not in those with a score of 0.
When actual event rates were compared with those predicted by the atherosclerotic cardiovascular disease (ASCVD) risk estimator introduced in the 2013 AHA/American College of Cardiology cholesterol guidelines, the event rate in subjects with an ASCVD 10-year risk estimate of 7.5%-15% but a CAC of 0 was just 4.4%.
Audience members noted that CAC scores didn’t do a very good job of stratifying stroke risk in MESA. That’s not surprising, since the score reflects coronary but not carotid artery calcium. But it is a limitation of CAC as a predictive tool, especially in light of the fact that strokes accounted for half of all cardiovascular events in the study.
Asked where he and his coinvestigators plan to go from here, Dr. Joshi said a randomized, controlled trial would be ideal, but to date funding isn’t available. However, the observational data from MESA and other studies suggest such a trial may not even be needed.
“Certainly the guidelines do allow for CAC scoring to be used in clinical decision making,” he noted.
The MESA study is funded by the National Heart, Lung, and Blood Institute. Dr. Joshi reported having no financial conflicts.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: A coronary artery calcium score of 0 appears to trump the 10-year atherosclerotic cardiovascular disease risk estimator introduced in the 2013 AHA/ACC cholesterol guidelines.
Major finding: The actual 10-year cardiovascular event rate in subjects with a coronary artery calcium score of 0 was just 4.4% – below the guideline-recommended threshold for statin therapy– even though their predicted risk using the AHA/ACC risk estimator was 7.5%-15%.
Data source: The Multi-Ethnic Study of Atherosclerosis is a prospective, population-based cohort study. This analysis included 6,814 subjects aged 45-84 who were free of clinical cardiovascular disease at baseline.
Disclosures: The MESA study is funded by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts.
Don't Miss AATS 95th Annual Meeting April 25-29, 2015
Mark your calendar to join cardiothoracic surgery professionals from around the world for a five-day program of state-of-the-art presentations by renowned experts. Attendees will enhance their knowledge and skills in a wide-range of subjects including general and specialized cardiac surgery, emerging technologies, congenital heart disease, critical care and aortic/endovascular.
AATS 95th Annual Meeting
April 25 – 29, 2015
Washington State Convention Center
Seattle, WA
President & Annual Meeting Chair
Pedro J. del Nido
Annual Meeting Co-Chairs
David H. Adams
Yolonda L. Colson
AATS is excited to be hosting its Annual Meeting in Seattle for the first time. This vibrant city combines sophisticated urbanity with the unpretentious natural surroundings of the Pacific Northwest. Nicknamed the “Emerald City” for its lush evergreen forests, Seattle has something for everyone — culture, entertainment, shopping, restaurants and outdoor activities. And, Seattle is the home of great coffee, which can be purchased from carts on every corner.
The meeting site — the recently renovated Washington State Convention Center — is located in the heart of downtown. Within walking distance is Seattle’s famous Space Needle, where visitors can view the city, Cascade Mountains and Mt. Rainer, the waters of Elliott Bay and surrounding forests from the 520-foot observation deck. Other attractions nearby are the Pike Place Market, Pioneer Square, water tours and ferries. Visitors and Seattleites enjoy Seattle’s lively downtown, great shopping, wonderful restaurants, espresso carts on every corner, and thriving community full of live theatre and museu
Attend the Saturday and Sunday Symposia, which include:
Saturday, April 25th
• Adult Cardiac Skills: How I Would Like My Operation Done
• Congenital Heart Disease Skills: Dealing with Challenging Conditions –Pearls and Pitfalls
• General Thoracic Skills: Implementing Innovation: What Future Leaders Need to Know
• Allied Health Personnel Symposium: Advancing the Team Based Care Management Model in Cardiothoracic Surgery
• Therapies for End-Stage Thoracic Organ Failure with an Emphasis on ECMO, MCS and Transplant
Sunday, April 26th
• AATS/STS Adult Cardiac Surgery Symposium: Decision Making in Adult Cardiac Surg
• AATS/STS Congenital Heart Disease Symposium: Unsettled and Unanswered Questions in Congenital Heart Surgery
• AATS/STS General Thoracic Surgery Symposium: The Evolving Role of Thoracic Surgeons
View full Saturday and Sunday programs online: http://aats.org/annualmeeting/Program-Books/2015/Program.cgi
Saturday and Sunday Courses and Symposium
Register for a Saturday course and/or a Sunday symposium, and you will have access to all other courses/symposia taking place that same day. You must select a primary course and will receive one syllabus book for that selection. Additional course syllabi will be available for purchase onsite for $25 each.
Sunday Symposia Legends Luncheon
Take advantage of this unique opportunity to hear from true pioneers in cardiothoracic surgery as they share their experiences during the interactive Legends Luncheons at each postgraduate symposium.
Speakers include:
• Professor Sir Magdi Yacoub — Imperial College, London
• Jean Deslauriers — IUCPQ, Canada
You must be registered for a Sunday postgraduate symposium to attend the Legends Luncheons.
Mark your calendar to join cardiothoracic surgery professionals from around the world for a five-day program of state-of-the-art presentations by renowned experts. Attendees will enhance their knowledge and skills in a wide-range of subjects including general and specialized cardiac surgery, emerging technologies, congenital heart disease, critical care and aortic/endovascular.
AATS 95th Annual Meeting
April 25 – 29, 2015
Washington State Convention Center
Seattle, WA
President & Annual Meeting Chair
Pedro J. del Nido
Annual Meeting Co-Chairs
David H. Adams
Yolonda L. Colson
AATS is excited to be hosting its Annual Meeting in Seattle for the first time. This vibrant city combines sophisticated urbanity with the unpretentious natural surroundings of the Pacific Northwest. Nicknamed the “Emerald City” for its lush evergreen forests, Seattle has something for everyone — culture, entertainment, shopping, restaurants and outdoor activities. And, Seattle is the home of great coffee, which can be purchased from carts on every corner.
The meeting site — the recently renovated Washington State Convention Center — is located in the heart of downtown. Within walking distance is Seattle’s famous Space Needle, where visitors can view the city, Cascade Mountains and Mt. Rainer, the waters of Elliott Bay and surrounding forests from the 520-foot observation deck. Other attractions nearby are the Pike Place Market, Pioneer Square, water tours and ferries. Visitors and Seattleites enjoy Seattle’s lively downtown, great shopping, wonderful restaurants, espresso carts on every corner, and thriving community full of live theatre and museu
Attend the Saturday and Sunday Symposia, which include:
Saturday, April 25th
• Adult Cardiac Skills: How I Would Like My Operation Done
• Congenital Heart Disease Skills: Dealing with Challenging Conditions –Pearls and Pitfalls
• General Thoracic Skills: Implementing Innovation: What Future Leaders Need to Know
• Allied Health Personnel Symposium: Advancing the Team Based Care Management Model in Cardiothoracic Surgery
• Therapies for End-Stage Thoracic Organ Failure with an Emphasis on ECMO, MCS and Transplant
Sunday, April 26th
• AATS/STS Adult Cardiac Surgery Symposium: Decision Making in Adult Cardiac Surg
• AATS/STS Congenital Heart Disease Symposium: Unsettled and Unanswered Questions in Congenital Heart Surgery
• AATS/STS General Thoracic Surgery Symposium: The Evolving Role of Thoracic Surgeons
View full Saturday and Sunday programs online: http://aats.org/annualmeeting/Program-Books/2015/Program.cgi
Saturday and Sunday Courses and Symposium
Register for a Saturday course and/or a Sunday symposium, and you will have access to all other courses/symposia taking place that same day. You must select a primary course and will receive one syllabus book for that selection. Additional course syllabi will be available for purchase onsite for $25 each.
Sunday Symposia Legends Luncheon
Take advantage of this unique opportunity to hear from true pioneers in cardiothoracic surgery as they share their experiences during the interactive Legends Luncheons at each postgraduate symposium.
Speakers include:
• Professor Sir Magdi Yacoub — Imperial College, London
• Jean Deslauriers — IUCPQ, Canada
You must be registered for a Sunday postgraduate symposium to attend the Legends Luncheons.
Mark your calendar to join cardiothoracic surgery professionals from around the world for a five-day program of state-of-the-art presentations by renowned experts. Attendees will enhance their knowledge and skills in a wide-range of subjects including general and specialized cardiac surgery, emerging technologies, congenital heart disease, critical care and aortic/endovascular.
AATS 95th Annual Meeting
April 25 – 29, 2015
Washington State Convention Center
Seattle, WA
President & Annual Meeting Chair
Pedro J. del Nido
Annual Meeting Co-Chairs
David H. Adams
Yolonda L. Colson
AATS is excited to be hosting its Annual Meeting in Seattle for the first time. This vibrant city combines sophisticated urbanity with the unpretentious natural surroundings of the Pacific Northwest. Nicknamed the “Emerald City” for its lush evergreen forests, Seattle has something for everyone — culture, entertainment, shopping, restaurants and outdoor activities. And, Seattle is the home of great coffee, which can be purchased from carts on every corner.
The meeting site — the recently renovated Washington State Convention Center — is located in the heart of downtown. Within walking distance is Seattle’s famous Space Needle, where visitors can view the city, Cascade Mountains and Mt. Rainer, the waters of Elliott Bay and surrounding forests from the 520-foot observation deck. Other attractions nearby are the Pike Place Market, Pioneer Square, water tours and ferries. Visitors and Seattleites enjoy Seattle’s lively downtown, great shopping, wonderful restaurants, espresso carts on every corner, and thriving community full of live theatre and museu
Attend the Saturday and Sunday Symposia, which include:
Saturday, April 25th
• Adult Cardiac Skills: How I Would Like My Operation Done
• Congenital Heart Disease Skills: Dealing with Challenging Conditions –Pearls and Pitfalls
• General Thoracic Skills: Implementing Innovation: What Future Leaders Need to Know
• Allied Health Personnel Symposium: Advancing the Team Based Care Management Model in Cardiothoracic Surgery
• Therapies for End-Stage Thoracic Organ Failure with an Emphasis on ECMO, MCS and Transplant
Sunday, April 26th
• AATS/STS Adult Cardiac Surgery Symposium: Decision Making in Adult Cardiac Surg
• AATS/STS Congenital Heart Disease Symposium: Unsettled and Unanswered Questions in Congenital Heart Surgery
• AATS/STS General Thoracic Surgery Symposium: The Evolving Role of Thoracic Surgeons
View full Saturday and Sunday programs online: http://aats.org/annualmeeting/Program-Books/2015/Program.cgi
Saturday and Sunday Courses and Symposium
Register for a Saturday course and/or a Sunday symposium, and you will have access to all other courses/symposia taking place that same day. You must select a primary course and will receive one syllabus book for that selection. Additional course syllabi will be available for purchase onsite for $25 each.
Sunday Symposia Legends Luncheon
Take advantage of this unique opportunity to hear from true pioneers in cardiothoracic surgery as they share their experiences during the interactive Legends Luncheons at each postgraduate symposium.
Speakers include:
• Professor Sir Magdi Yacoub — Imperial College, London
• Jean Deslauriers — IUCPQ, Canada
You must be registered for a Sunday postgraduate symposium to attend the Legends Luncheons.