User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Pleconaril shows promise for neonatal enteroviral sepsis
PHILADELPHIA – Pleconaril, an oral capsid binder with activity against enterovirus infections, shows promise for the treatment of neonates with enterovirus sepsis, according to findings from a randomized, double-blind, placebo-controlled trial.
Time to culture-negativity for all types of collected specimens combined – including those from the oropharynx, rectum, urine, and serum – was faster in 43 patients treated with 5 mg/kg of pleconaril every 8 hours for 7 days than in18 patients who received placebo (median of 4.0 vs. 7.0 days). The difference approached statistical significance.
Fewer subjects in the treatment group remained polymerase chain reaction-positive at last oropharynx sample (83% and 23% were positive on day 1 and at a median of day 14 in the treatment group, vs. 100% and 58%, respectively, in the placebo group), Dr. Mark Abzug of the University of Colorado, Aurora, reported at an annual scientific meeting on infectious diseases.
Mortality in an intention-to-treat analysis was 23% in the treatment group, compared with 44% in the placebo group, and mortality in those with confirmed enterovirus infection was 23% and 42% in the treatment and placebo groups, respectively, Dr. Abzug said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Patients were neonates with suspected enterovirus sepsis with illness onset in the first 15 days of life. They were randomized 2:1 to receive active treatment or placebo for 7 days. Specimens were obtained serially for 14 days for viral culture and PCR, pharmacokinetic analysis, and safety evaluations, and clinical assessments were performed over 24 months.
Enterovirus was confirmed by culture or PCR in 43 of the 61 patients, including 31 in the treatment group and 12 in the placebo group. The two groups were similar with respect to baseline characteristics, including median age at illness onset (4.5 and 5.0 days, respectively) and time until enrollment (6 to 7 days into the course of their illness).
Of note, pleconaril concentrations exceeded the 90% inhibitory concentration (IC90) level after the first day of treatment in the treatment group, but 41% of subjects did not achieve this target until after day 4, suggesting the need for a loading dose.
Neonatal enterovirus infections are associated with high morbidity and mortality, and therapy is mainly supportive. While intravenous immunoglobulin is thought to confer potential benefit, it remains unproven, and no specific antiviral therapies are currently available, Dr. Abzug said.
Pleconaril has been shown previously to have activity against most enteroviruses and many rhinoviruses. It is well tolerated: In the current study, treatment-related adverse effects occurred in 1 subject in the treatment group, and in 3 in the placebo group.
While the unexpectedly low yield of viral cultures in this study precluded the demonstration of a difference between the groups with regard to the primary endpoint of day 5 oropharyngeal culture positivity, which was 25% on day 1 and 0% on day 5 in the treatment group, vs. 30% on day 1 and 0% on day 5 in the placebo group, the shorter times to culture and PCR negativity and the suggestion of greater survival in the treatment group are encouraging, Dr. Abzug said.
These indicators of biological and clinical efficacy warrant further investigation of pleconaril for neonatal enteroviral disease, he concluded.The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Abzug reported having no disclosures.
PHILADELPHIA – Pleconaril, an oral capsid binder with activity against enterovirus infections, shows promise for the treatment of neonates with enterovirus sepsis, according to findings from a randomized, double-blind, placebo-controlled trial.
Time to culture-negativity for all types of collected specimens combined – including those from the oropharynx, rectum, urine, and serum – was faster in 43 patients treated with 5 mg/kg of pleconaril every 8 hours for 7 days than in18 patients who received placebo (median of 4.0 vs. 7.0 days). The difference approached statistical significance.
Fewer subjects in the treatment group remained polymerase chain reaction-positive at last oropharynx sample (83% and 23% were positive on day 1 and at a median of day 14 in the treatment group, vs. 100% and 58%, respectively, in the placebo group), Dr. Mark Abzug of the University of Colorado, Aurora, reported at an annual scientific meeting on infectious diseases.
Mortality in an intention-to-treat analysis was 23% in the treatment group, compared with 44% in the placebo group, and mortality in those with confirmed enterovirus infection was 23% and 42% in the treatment and placebo groups, respectively, Dr. Abzug said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Patients were neonates with suspected enterovirus sepsis with illness onset in the first 15 days of life. They were randomized 2:1 to receive active treatment or placebo for 7 days. Specimens were obtained serially for 14 days for viral culture and PCR, pharmacokinetic analysis, and safety evaluations, and clinical assessments were performed over 24 months.
Enterovirus was confirmed by culture or PCR in 43 of the 61 patients, including 31 in the treatment group and 12 in the placebo group. The two groups were similar with respect to baseline characteristics, including median age at illness onset (4.5 and 5.0 days, respectively) and time until enrollment (6 to 7 days into the course of their illness).
Of note, pleconaril concentrations exceeded the 90% inhibitory concentration (IC90) level after the first day of treatment in the treatment group, but 41% of subjects did not achieve this target until after day 4, suggesting the need for a loading dose.
Neonatal enterovirus infections are associated with high morbidity and mortality, and therapy is mainly supportive. While intravenous immunoglobulin is thought to confer potential benefit, it remains unproven, and no specific antiviral therapies are currently available, Dr. Abzug said.
Pleconaril has been shown previously to have activity against most enteroviruses and many rhinoviruses. It is well tolerated: In the current study, treatment-related adverse effects occurred in 1 subject in the treatment group, and in 3 in the placebo group.
While the unexpectedly low yield of viral cultures in this study precluded the demonstration of a difference between the groups with regard to the primary endpoint of day 5 oropharyngeal culture positivity, which was 25% on day 1 and 0% on day 5 in the treatment group, vs. 30% on day 1 and 0% on day 5 in the placebo group, the shorter times to culture and PCR negativity and the suggestion of greater survival in the treatment group are encouraging, Dr. Abzug said.
These indicators of biological and clinical efficacy warrant further investigation of pleconaril for neonatal enteroviral disease, he concluded.The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Abzug reported having no disclosures.
PHILADELPHIA – Pleconaril, an oral capsid binder with activity against enterovirus infections, shows promise for the treatment of neonates with enterovirus sepsis, according to findings from a randomized, double-blind, placebo-controlled trial.
Time to culture-negativity for all types of collected specimens combined – including those from the oropharynx, rectum, urine, and serum – was faster in 43 patients treated with 5 mg/kg of pleconaril every 8 hours for 7 days than in18 patients who received placebo (median of 4.0 vs. 7.0 days). The difference approached statistical significance.
Fewer subjects in the treatment group remained polymerase chain reaction-positive at last oropharynx sample (83% and 23% were positive on day 1 and at a median of day 14 in the treatment group, vs. 100% and 58%, respectively, in the placebo group), Dr. Mark Abzug of the University of Colorado, Aurora, reported at an annual scientific meeting on infectious diseases.
Mortality in an intention-to-treat analysis was 23% in the treatment group, compared with 44% in the placebo group, and mortality in those with confirmed enterovirus infection was 23% and 42% in the treatment and placebo groups, respectively, Dr. Abzug said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Patients were neonates with suspected enterovirus sepsis with illness onset in the first 15 days of life. They were randomized 2:1 to receive active treatment or placebo for 7 days. Specimens were obtained serially for 14 days for viral culture and PCR, pharmacokinetic analysis, and safety evaluations, and clinical assessments were performed over 24 months.
Enterovirus was confirmed by culture or PCR in 43 of the 61 patients, including 31 in the treatment group and 12 in the placebo group. The two groups were similar with respect to baseline characteristics, including median age at illness onset (4.5 and 5.0 days, respectively) and time until enrollment (6 to 7 days into the course of their illness).
Of note, pleconaril concentrations exceeded the 90% inhibitory concentration (IC90) level after the first day of treatment in the treatment group, but 41% of subjects did not achieve this target until after day 4, suggesting the need for a loading dose.
Neonatal enterovirus infections are associated with high morbidity and mortality, and therapy is mainly supportive. While intravenous immunoglobulin is thought to confer potential benefit, it remains unproven, and no specific antiviral therapies are currently available, Dr. Abzug said.
Pleconaril has been shown previously to have activity against most enteroviruses and many rhinoviruses. It is well tolerated: In the current study, treatment-related adverse effects occurred in 1 subject in the treatment group, and in 3 in the placebo group.
While the unexpectedly low yield of viral cultures in this study precluded the demonstration of a difference between the groups with regard to the primary endpoint of day 5 oropharyngeal culture positivity, which was 25% on day 1 and 0% on day 5 in the treatment group, vs. 30% on day 1 and 0% on day 5 in the placebo group, the shorter times to culture and PCR negativity and the suggestion of greater survival in the treatment group are encouraging, Dr. Abzug said.
These indicators of biological and clinical efficacy warrant further investigation of pleconaril for neonatal enteroviral disease, he concluded.The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Abzug reported having no disclosures.
AT ID Week 2014
Key clinical point: Pleconaril showed some signs of efficacy for enteroviral sepsis in neonates in a randomized controlled trial.
Major finding: Mortality was 23% in the treatment group, compared with 44% in the placebo group.
Data source: A randomized placebo-controlled trial of 61 neonates.
Disclosures: The study was funded by the National Institute for Allergy and Infectious Diseases. Dr. Abzug reported having no disclosures.
Antibiotic stewardship program reduced pediatric LOS, readmission rate
PHILADELPHIA – Adherence to the recommendations of an antibiotic stewardship program committee decreased 30-day readmissions without increasing length of stay at a freestanding children’s hospital.
Between March 3, 2008, and March 3, 2013, the antibiotic stewardship program (ASP) committee at Children’s Mercy Hospital–Kansas City, Mo., recommended discontinuing a prescribed antibiotic, changing the antibiotic, or reducing the antibiotic dose in 1,240 patients. When the recommendations were followed, no 30-day readmissions occurred, and median length of stay was 79 hours. When they were not followed, readmissions within 30 days occurred in 3% and 4% of otherwise healthy surgical and medical patients and in patients with chronic complex medical conditions, respectively, and median length of stay was 82 hours, Dr. Jason G. Newland reported at an annual scientific meeting on infectious diseases.
The study is the first to demonstrate that a stewardship program designed to control antibiotic prescription and treatment in the hospital setting has beneficial effects on children’s health, Dr. Newland, medical director of patient safety and systems reliability at Children’s Mercy Hospitals and Clinics and the University of Missouri–-Kansas City said during a press briefing at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
“We obviously were excited to see that we didn’t impact length of stay on a negative basis,” he said, adding that the 0% readmission rates in cases in which the physician agreed with the antibiotic stewardship program (ASP) recommendation was a particularly encouraging finding.
The ASP committee at Children’s Mercy Hospital, like most such programs, is led by epidemiologists and infectious disease physicians who are tasked with promoting the appropriate use of antibiotics in an effort to prevent the development of antibiotic resistance.
In the vast majority of cases included in the current study, the ASP committee’s recommendation was to discontinue the antibiotic, as it was deemed unnecessary, Dr. Newland noted.
ASP interventions occurred most often in patients receiving ceftriaxone/cefotaxime (62%), vancomycin (11%), and meropenem (5%). The most common diagnoses were pneumonia (22%), urinary tract infections (19%), and rule out sepsis (9%).
The findings reinforce the importance of viewing ASP committees as extensions of patient-safety initiatives, “as their presence really exemplifies the idea of do no harm,” said Dr. Pranita D. Tamma of Johns Hopkins Children’s Center, Baltimore, who moderated the press briefing.
Dr. Newland and his colleagues demonstrated the value of ASP committees for reducing the likelihood of further negative sequelae to which a child could be exposed as a result of inappropriate antibiotic use, such as the increased risk of health care associated infections that might come with readmission, having to take further medications, and undergo other interventions, and the cost and other burdens to families and the health care system as a whole, she said.
Dr. Newland reported receiving grant funding from Pfizer.
PHILADELPHIA – Adherence to the recommendations of an antibiotic stewardship program committee decreased 30-day readmissions without increasing length of stay at a freestanding children’s hospital.
Between March 3, 2008, and March 3, 2013, the antibiotic stewardship program (ASP) committee at Children’s Mercy Hospital–Kansas City, Mo., recommended discontinuing a prescribed antibiotic, changing the antibiotic, or reducing the antibiotic dose in 1,240 patients. When the recommendations were followed, no 30-day readmissions occurred, and median length of stay was 79 hours. When they were not followed, readmissions within 30 days occurred in 3% and 4% of otherwise healthy surgical and medical patients and in patients with chronic complex medical conditions, respectively, and median length of stay was 82 hours, Dr. Jason G. Newland reported at an annual scientific meeting on infectious diseases.
The study is the first to demonstrate that a stewardship program designed to control antibiotic prescription and treatment in the hospital setting has beneficial effects on children’s health, Dr. Newland, medical director of patient safety and systems reliability at Children’s Mercy Hospitals and Clinics and the University of Missouri–-Kansas City said during a press briefing at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
“We obviously were excited to see that we didn’t impact length of stay on a negative basis,” he said, adding that the 0% readmission rates in cases in which the physician agreed with the antibiotic stewardship program (ASP) recommendation was a particularly encouraging finding.
The ASP committee at Children’s Mercy Hospital, like most such programs, is led by epidemiologists and infectious disease physicians who are tasked with promoting the appropriate use of antibiotics in an effort to prevent the development of antibiotic resistance.
In the vast majority of cases included in the current study, the ASP committee’s recommendation was to discontinue the antibiotic, as it was deemed unnecessary, Dr. Newland noted.
ASP interventions occurred most often in patients receiving ceftriaxone/cefotaxime (62%), vancomycin (11%), and meropenem (5%). The most common diagnoses were pneumonia (22%), urinary tract infections (19%), and rule out sepsis (9%).
The findings reinforce the importance of viewing ASP committees as extensions of patient-safety initiatives, “as their presence really exemplifies the idea of do no harm,” said Dr. Pranita D. Tamma of Johns Hopkins Children’s Center, Baltimore, who moderated the press briefing.
Dr. Newland and his colleagues demonstrated the value of ASP committees for reducing the likelihood of further negative sequelae to which a child could be exposed as a result of inappropriate antibiotic use, such as the increased risk of health care associated infections that might come with readmission, having to take further medications, and undergo other interventions, and the cost and other burdens to families and the health care system as a whole, she said.
Dr. Newland reported receiving grant funding from Pfizer.
PHILADELPHIA – Adherence to the recommendations of an antibiotic stewardship program committee decreased 30-day readmissions without increasing length of stay at a freestanding children’s hospital.
Between March 3, 2008, and March 3, 2013, the antibiotic stewardship program (ASP) committee at Children’s Mercy Hospital–Kansas City, Mo., recommended discontinuing a prescribed antibiotic, changing the antibiotic, or reducing the antibiotic dose in 1,240 patients. When the recommendations were followed, no 30-day readmissions occurred, and median length of stay was 79 hours. When they were not followed, readmissions within 30 days occurred in 3% and 4% of otherwise healthy surgical and medical patients and in patients with chronic complex medical conditions, respectively, and median length of stay was 82 hours, Dr. Jason G. Newland reported at an annual scientific meeting on infectious diseases.
The study is the first to demonstrate that a stewardship program designed to control antibiotic prescription and treatment in the hospital setting has beneficial effects on children’s health, Dr. Newland, medical director of patient safety and systems reliability at Children’s Mercy Hospitals and Clinics and the University of Missouri–-Kansas City said during a press briefing at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
“We obviously were excited to see that we didn’t impact length of stay on a negative basis,” he said, adding that the 0% readmission rates in cases in which the physician agreed with the antibiotic stewardship program (ASP) recommendation was a particularly encouraging finding.
The ASP committee at Children’s Mercy Hospital, like most such programs, is led by epidemiologists and infectious disease physicians who are tasked with promoting the appropriate use of antibiotics in an effort to prevent the development of antibiotic resistance.
In the vast majority of cases included in the current study, the ASP committee’s recommendation was to discontinue the antibiotic, as it was deemed unnecessary, Dr. Newland noted.
ASP interventions occurred most often in patients receiving ceftriaxone/cefotaxime (62%), vancomycin (11%), and meropenem (5%). The most common diagnoses were pneumonia (22%), urinary tract infections (19%), and rule out sepsis (9%).
The findings reinforce the importance of viewing ASP committees as extensions of patient-safety initiatives, “as their presence really exemplifies the idea of do no harm,” said Dr. Pranita D. Tamma of Johns Hopkins Children’s Center, Baltimore, who moderated the press briefing.
Dr. Newland and his colleagues demonstrated the value of ASP committees for reducing the likelihood of further negative sequelae to which a child could be exposed as a result of inappropriate antibiotic use, such as the increased risk of health care associated infections that might come with readmission, having to take further medications, and undergo other interventions, and the cost and other burdens to families and the health care system as a whole, she said.
Dr. Newland reported receiving grant funding from Pfizer.
Key clinical point: An antibiotic stewardship program benefited hospitalized children’s health outcomes.
Major finding: At 30 days, readmissions were 0% and 3%-4%, and median length of stay was 79 vs. 82 hours; when recommendations were vs. were not followed.
Data source: Observational study of outcomes following intervention in 1,240 of more than 7,000 patients.
Disclosures: Dr. Newland reported receiving grant funding from Pfizer.
Later transplant for renal failure in lupus nephritis may raise graft failure risk
Delaying kidney transplantation to allow for quiescence of systemic lupus erythematosus–related immune activity in patients with lupus nephritis and end-stage renal disease does not appear to improve graft outcomes, according to an analysis of national surveillance data.
Of 4,743 transplant recipients with lupus nephritis and end-stage renal disease (LN-ESRD), 1,239 experienced graft failure. Overall, wait times of 3-12 months and 12-24 months were associated with 25% and 37% increased risk of graft failure, respectively, compared with wait times of less than 3 months, after adjustment for age, race, insurance, hemoglobin, and donor type.
A similar pattern was seen in white patients, except that wait times of more than 36 months in white patients were associated with a near doubling of graft failure risk (hazard ratio, 1.98), Laura C. Plantinga of Emory University, Atlanta, and her colleagues reported (Arthritis Care Res. 2014 Sept. 23 [doi:10.1002/acr.22482]).
Among black patients, longer wait times were not associated with graft failure in the adjusted analysis, and, in fact, there was a nonstatistically significant suggestion of a protective effect for wait time of 2 years or more. This finding may reflect unexplained differences in disease pathology between white and black LN-ESRD patients, the investigators said, adding that there was no increased risk of graft failure in black patients who were transplanted early.
“Our results suggest U.S. recommendations for transplantation in LN-ESRD may not align with evidence from the target population,” they said, noting that the results should be considered hypotheses-generating because of the limitations of the study and that additional study is needed to examine the potential confounding effects of clinically recognized SLE activity on the associations observed in this study.
Some of the investigators were supported through grants from the National Institutes of Health.
Delaying kidney transplantation to allow for quiescence of systemic lupus erythematosus–related immune activity in patients with lupus nephritis and end-stage renal disease does not appear to improve graft outcomes, according to an analysis of national surveillance data.
Of 4,743 transplant recipients with lupus nephritis and end-stage renal disease (LN-ESRD), 1,239 experienced graft failure. Overall, wait times of 3-12 months and 12-24 months were associated with 25% and 37% increased risk of graft failure, respectively, compared with wait times of less than 3 months, after adjustment for age, race, insurance, hemoglobin, and donor type.
A similar pattern was seen in white patients, except that wait times of more than 36 months in white patients were associated with a near doubling of graft failure risk (hazard ratio, 1.98), Laura C. Plantinga of Emory University, Atlanta, and her colleagues reported (Arthritis Care Res. 2014 Sept. 23 [doi:10.1002/acr.22482]).
Among black patients, longer wait times were not associated with graft failure in the adjusted analysis, and, in fact, there was a nonstatistically significant suggestion of a protective effect for wait time of 2 years or more. This finding may reflect unexplained differences in disease pathology between white and black LN-ESRD patients, the investigators said, adding that there was no increased risk of graft failure in black patients who were transplanted early.
“Our results suggest U.S. recommendations for transplantation in LN-ESRD may not align with evidence from the target population,” they said, noting that the results should be considered hypotheses-generating because of the limitations of the study and that additional study is needed to examine the potential confounding effects of clinically recognized SLE activity on the associations observed in this study.
Some of the investigators were supported through grants from the National Institutes of Health.
Delaying kidney transplantation to allow for quiescence of systemic lupus erythematosus–related immune activity in patients with lupus nephritis and end-stage renal disease does not appear to improve graft outcomes, according to an analysis of national surveillance data.
Of 4,743 transplant recipients with lupus nephritis and end-stage renal disease (LN-ESRD), 1,239 experienced graft failure. Overall, wait times of 3-12 months and 12-24 months were associated with 25% and 37% increased risk of graft failure, respectively, compared with wait times of less than 3 months, after adjustment for age, race, insurance, hemoglobin, and donor type.
A similar pattern was seen in white patients, except that wait times of more than 36 months in white patients were associated with a near doubling of graft failure risk (hazard ratio, 1.98), Laura C. Plantinga of Emory University, Atlanta, and her colleagues reported (Arthritis Care Res. 2014 Sept. 23 [doi:10.1002/acr.22482]).
Among black patients, longer wait times were not associated with graft failure in the adjusted analysis, and, in fact, there was a nonstatistically significant suggestion of a protective effect for wait time of 2 years or more. This finding may reflect unexplained differences in disease pathology between white and black LN-ESRD patients, the investigators said, adding that there was no increased risk of graft failure in black patients who were transplanted early.
“Our results suggest U.S. recommendations for transplantation in LN-ESRD may not align with evidence from the target population,” they said, noting that the results should be considered hypotheses-generating because of the limitations of the study and that additional study is needed to examine the potential confounding effects of clinically recognized SLE activity on the associations observed in this study.
Some of the investigators were supported through grants from the National Institutes of Health.
Key clinical point: Delaying transplantation in LN-ESRD patients may do more harm than good, although future studies should determine if longer wait times remain associated with increased risk of graft failure, independent of clinically recognized SLE activity.
Major finding: Overall risk of graft failure was increased by 25% and 37% with wait times of 3-12 months and 12-24 months, respectively (vs. less than 3 months).
Data source: National ESRD surveillance data (U.S. Renal Data System) for 4,743 LN-ESRD transplant recipients.
Disclosures: Some of the investigators were supported through grants from the National Institutes of Health.
Watch for PML after switching from natalizumab to fingolimod
BOSTON– Cases of progressive multifocal leukoencephalopathy in multiple sclerosis patients who switch from natalizumab to fingolimod treatment are more likely a result of the natalizumab exposure than the fingolimod exposure, according to an analysis of cases in the Novartis safety reporting database for fingolimod.
About 15%-20% of fingolimod-treated patients have prior exposure to natalizumab, and of 11 patients from the safety reporting database who have developed progressive multifocal leukoencephalopathy (PML) to date, all had prior natalizumab exposure, according to Dr. Norman Putzki.
The 11 patients with PML had been treated with natalizumab for a median of 4 years, Dr. Putzki, an employee of Novartis Pharma AG, Basel, Switzerland, said at the joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The onset of PML, based on a retrospective review of MRI scans, was prior to the first fingolimod dose in 5 of the 11 patients, and between 3 weeks and 6 months after the first dose in 4 of the patients. Onset was unknown in the remaining 2 patients.
One additional case of possible PML was reported in a patient without prior natalizumab exposure. The patient had MRI features and the presence of AQP4 antibodies consistent with a diagnosis of neuromyelitis optica spectrum disorder, and clinical features that were consistent with a PML diagnosis, but the timing of onset in this patient could not be determined, Dr. Putzki said.
With more than 135,800 total patient-years of exposure, including during clinical trials and in the postmarketing period, no evidence of an association between fingolimod and PML has been found, he said.
Treatment with natalizumab, however, is an identified predisposing factor for PML development, and the risk is known to persist after treatment discontinuation. In fact, it is recommended that patients be monitored for any new signs or symptoms suggestive of PML for at least 6 months following discontinuation of natalizumab, he said, noting that this is particularly true for patients with John Cunningham virus (JCV) seropositivity, prior exposure to immunosuppressants, or long-term natalizumab exposure, as these patients may be at increased risk of developing PML.
With exposure to natalizumab for 25-48 months, the rate of PML in JCV-positive patients is 3/1,000 patients in those with no prior exposure to immunosuppressant therapy, and 13/1,000 in those with prior exposure to immunosuppressant therapy, whereas with exposure for 49-72 months, the respective rates are 7/1,000 and 9/1,000, he said.
Although definitive conclusions about PML risk and exposure to natalizumab and fingolimod cannot be made based on the current analysis, an excess risk due to an overlapping pharmacodynamic effect when switching from natalizumab to fingolimod was not established, and a risk of PML cannot be confidently attributed to fingolimod, Dr. Putzki said.
A baseline neurological examination and MRI scan prior to initiating fingolimod treatment may help – at least retrospectively – in attributing PML causality to natalizumab vs. fingolimod, he added.
“Vigilance for PML is warranted following natalizumab discontinuation irrespective of consecutive treatment approaches,” he concluded.
BOSTON– Cases of progressive multifocal leukoencephalopathy in multiple sclerosis patients who switch from natalizumab to fingolimod treatment are more likely a result of the natalizumab exposure than the fingolimod exposure, according to an analysis of cases in the Novartis safety reporting database for fingolimod.
About 15%-20% of fingolimod-treated patients have prior exposure to natalizumab, and of 11 patients from the safety reporting database who have developed progressive multifocal leukoencephalopathy (PML) to date, all had prior natalizumab exposure, according to Dr. Norman Putzki.
The 11 patients with PML had been treated with natalizumab for a median of 4 years, Dr. Putzki, an employee of Novartis Pharma AG, Basel, Switzerland, said at the joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The onset of PML, based on a retrospective review of MRI scans, was prior to the first fingolimod dose in 5 of the 11 patients, and between 3 weeks and 6 months after the first dose in 4 of the patients. Onset was unknown in the remaining 2 patients.
One additional case of possible PML was reported in a patient without prior natalizumab exposure. The patient had MRI features and the presence of AQP4 antibodies consistent with a diagnosis of neuromyelitis optica spectrum disorder, and clinical features that were consistent with a PML diagnosis, but the timing of onset in this patient could not be determined, Dr. Putzki said.
With more than 135,800 total patient-years of exposure, including during clinical trials and in the postmarketing period, no evidence of an association between fingolimod and PML has been found, he said.
Treatment with natalizumab, however, is an identified predisposing factor for PML development, and the risk is known to persist after treatment discontinuation. In fact, it is recommended that patients be monitored for any new signs or symptoms suggestive of PML for at least 6 months following discontinuation of natalizumab, he said, noting that this is particularly true for patients with John Cunningham virus (JCV) seropositivity, prior exposure to immunosuppressants, or long-term natalizumab exposure, as these patients may be at increased risk of developing PML.
With exposure to natalizumab for 25-48 months, the rate of PML in JCV-positive patients is 3/1,000 patients in those with no prior exposure to immunosuppressant therapy, and 13/1,000 in those with prior exposure to immunosuppressant therapy, whereas with exposure for 49-72 months, the respective rates are 7/1,000 and 9/1,000, he said.
Although definitive conclusions about PML risk and exposure to natalizumab and fingolimod cannot be made based on the current analysis, an excess risk due to an overlapping pharmacodynamic effect when switching from natalizumab to fingolimod was not established, and a risk of PML cannot be confidently attributed to fingolimod, Dr. Putzki said.
A baseline neurological examination and MRI scan prior to initiating fingolimod treatment may help – at least retrospectively – in attributing PML causality to natalizumab vs. fingolimod, he added.
“Vigilance for PML is warranted following natalizumab discontinuation irrespective of consecutive treatment approaches,” he concluded.
BOSTON– Cases of progressive multifocal leukoencephalopathy in multiple sclerosis patients who switch from natalizumab to fingolimod treatment are more likely a result of the natalizumab exposure than the fingolimod exposure, according to an analysis of cases in the Novartis safety reporting database for fingolimod.
About 15%-20% of fingolimod-treated patients have prior exposure to natalizumab, and of 11 patients from the safety reporting database who have developed progressive multifocal leukoencephalopathy (PML) to date, all had prior natalizumab exposure, according to Dr. Norman Putzki.
The 11 patients with PML had been treated with natalizumab for a median of 4 years, Dr. Putzki, an employee of Novartis Pharma AG, Basel, Switzerland, said at the joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The onset of PML, based on a retrospective review of MRI scans, was prior to the first fingolimod dose in 5 of the 11 patients, and between 3 weeks and 6 months after the first dose in 4 of the patients. Onset was unknown in the remaining 2 patients.
One additional case of possible PML was reported in a patient without prior natalizumab exposure. The patient had MRI features and the presence of AQP4 antibodies consistent with a diagnosis of neuromyelitis optica spectrum disorder, and clinical features that were consistent with a PML diagnosis, but the timing of onset in this patient could not be determined, Dr. Putzki said.
With more than 135,800 total patient-years of exposure, including during clinical trials and in the postmarketing period, no evidence of an association between fingolimod and PML has been found, he said.
Treatment with natalizumab, however, is an identified predisposing factor for PML development, and the risk is known to persist after treatment discontinuation. In fact, it is recommended that patients be monitored for any new signs or symptoms suggestive of PML for at least 6 months following discontinuation of natalizumab, he said, noting that this is particularly true for patients with John Cunningham virus (JCV) seropositivity, prior exposure to immunosuppressants, or long-term natalizumab exposure, as these patients may be at increased risk of developing PML.
With exposure to natalizumab for 25-48 months, the rate of PML in JCV-positive patients is 3/1,000 patients in those with no prior exposure to immunosuppressant therapy, and 13/1,000 in those with prior exposure to immunosuppressant therapy, whereas with exposure for 49-72 months, the respective rates are 7/1,000 and 9/1,000, he said.
Although definitive conclusions about PML risk and exposure to natalizumab and fingolimod cannot be made based on the current analysis, an excess risk due to an overlapping pharmacodynamic effect when switching from natalizumab to fingolimod was not established, and a risk of PML cannot be confidently attributed to fingolimod, Dr. Putzki said.
A baseline neurological examination and MRI scan prior to initiating fingolimod treatment may help – at least retrospectively – in attributing PML causality to natalizumab vs. fingolimod, he added.
“Vigilance for PML is warranted following natalizumab discontinuation irrespective of consecutive treatment approaches,” he concluded.
Key clinical point: Natalizumab-related PML risk may persist after switch to fingolimod.
Major finding: All PML cases were in patients with prior natalizumab exposure.
Data source: An analysis of 11 PML cases from the Novartis safety reporting database for fingolimod.
Disclosures: Dr. Putzki is employed by Novartis.
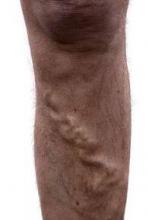
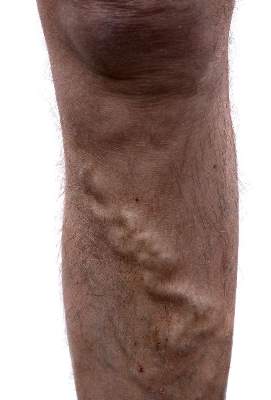
Various varicose vein treatments show similar efficacy
Ultrasound-guided foam sclerotherapy, endovenous laser ablation, and surgery for the treatment of varicose veins were generally similar with respect to quality of life measures and clinical efficacy and safety in a randomized trial involving 785 patients.
At 6 months, disease-specific quality of life was slightly worse after treatment with foam than after surgery (effect size, –1.74; P = .006). A post hoc analysis showed slightly better generic quality of life after laser ablation than after foam sclerotherapy (effect size, 1.54; P = .048).
In addition, a lower rate of procedural complications occurred with laser ablation (1%, compared with 6% and 7% for foam sclerotherapy and surgery, respectively), and a lower rate of completely successful ablation of great saphenous veins at 6 weeks occurred after foam treatment (55%, compared with 84% and 83% for the surgery and laser groups, respectively).
Dr. Julie Brittenden of the University of Aberdeen, Scotland, and her colleagues reported the findings online Sept. 25 in the New England Journal of Medicine (N. Engl. J. Med. 2014 Sept. 25 [doi: 10/1056/NEJMoa1400781]).
Subjects in the Comparison of Laser, Surgery, and Foam Sclerotherapy (CLASS) trial were adults with primary varicose veins who were treated at 11 centers in the United Kingdom between November 2008 and October 2012. They were evaluated at 6 weeks and 6 months after treatment.
The baseline scores and overall improvements in quality of life seen in the CLASS trial were comparable to those in smaller European randomized trials, which supports the generalizability of the findings, the investigators said.
This study was supported by a grant from the Health Technology Assessment Programme of the U.K. National Institute for Health Research. Dr. Brittenden reported having no other disclosures.
Ultrasound-guided foam sclerotherapy, endovenous laser ablation, and surgery for the treatment of varicose veins were generally similar with respect to quality of life measures and clinical efficacy and safety in a randomized trial involving 785 patients.
At 6 months, disease-specific quality of life was slightly worse after treatment with foam than after surgery (effect size, –1.74; P = .006). A post hoc analysis showed slightly better generic quality of life after laser ablation than after foam sclerotherapy (effect size, 1.54; P = .048).
In addition, a lower rate of procedural complications occurred with laser ablation (1%, compared with 6% and 7% for foam sclerotherapy and surgery, respectively), and a lower rate of completely successful ablation of great saphenous veins at 6 weeks occurred after foam treatment (55%, compared with 84% and 83% for the surgery and laser groups, respectively).
Dr. Julie Brittenden of the University of Aberdeen, Scotland, and her colleagues reported the findings online Sept. 25 in the New England Journal of Medicine (N. Engl. J. Med. 2014 Sept. 25 [doi: 10/1056/NEJMoa1400781]).
Subjects in the Comparison of Laser, Surgery, and Foam Sclerotherapy (CLASS) trial were adults with primary varicose veins who were treated at 11 centers in the United Kingdom between November 2008 and October 2012. They were evaluated at 6 weeks and 6 months after treatment.
The baseline scores and overall improvements in quality of life seen in the CLASS trial were comparable to those in smaller European randomized trials, which supports the generalizability of the findings, the investigators said.
This study was supported by a grant from the Health Technology Assessment Programme of the U.K. National Institute for Health Research. Dr. Brittenden reported having no other disclosures.
Ultrasound-guided foam sclerotherapy, endovenous laser ablation, and surgery for the treatment of varicose veins were generally similar with respect to quality of life measures and clinical efficacy and safety in a randomized trial involving 785 patients.
At 6 months, disease-specific quality of life was slightly worse after treatment with foam than after surgery (effect size, –1.74; P = .006). A post hoc analysis showed slightly better generic quality of life after laser ablation than after foam sclerotherapy (effect size, 1.54; P = .048).
In addition, a lower rate of procedural complications occurred with laser ablation (1%, compared with 6% and 7% for foam sclerotherapy and surgery, respectively), and a lower rate of completely successful ablation of great saphenous veins at 6 weeks occurred after foam treatment (55%, compared with 84% and 83% for the surgery and laser groups, respectively).
Dr. Julie Brittenden of the University of Aberdeen, Scotland, and her colleagues reported the findings online Sept. 25 in the New England Journal of Medicine (N. Engl. J. Med. 2014 Sept. 25 [doi: 10/1056/NEJMoa1400781]).
Subjects in the Comparison of Laser, Surgery, and Foam Sclerotherapy (CLASS) trial were adults with primary varicose veins who were treated at 11 centers in the United Kingdom between November 2008 and October 2012. They were evaluated at 6 weeks and 6 months after treatment.
The baseline scores and overall improvements in quality of life seen in the CLASS trial were comparable to those in smaller European randomized trials, which supports the generalizability of the findings, the investigators said.
This study was supported by a grant from the Health Technology Assessment Programme of the U.K. National Institute for Health Research. Dr. Brittenden reported having no other disclosures.
Key clinical point: Quality of life outcomes don’t differ substantially between varicose vein treatments.
Major finding: The rate of complications was lower with laser ablation (1%) than with foam sclerotherapy and surgery (6% and 7%, respectively).
Data source: The randomized CLASS trial involving 785 subjects.
Disclosures: This study was supported by a grant from the Health Technology Assessment Programme of the National Institute for Health Research. Dr. Brittenden reported having no other disclosures.
Nurses’ health studies show no link between diet, multiple sclerosis
BOSTON – Diet does not appear to contribute to the development of multiple sclerosis, according to findings from more than 185,000 prospectively followed women from the Nurses’ Health Study and the Nurses’ Health Study II.
None of three dietary indices measured at baseline – the Alternative Healthy Eating Index-2010 (AHEI-2010), the Alternate Mediterranean Diet index (aMED), and the Dietary Approaches to Stop Hypertension index (DASH) – were significantly associated with the risk of developing multiple sclerosis in the two longitudinal cohort studies, Dr. Dalia Rotstein reported at the a joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
After adjustment for known confounders of multiple sclerosis (MS) risk, including age, latitude of residence at age 15 years, body mass index at age 18 years, pack-years of smoking, total energy intake, supplemental vitamin D intake, and ethnicity, the pooled relative risk of MS for the highest vs. the lowest quintile of scores for each index was 0.89 for the AHEI-2010, 1.10 for aMED, and 1.30 for DASH, said Dr. Rotstein of Brigham and Women’s Hospital, Boston.
A principal-components analysis identified two general dietary patterns among the subjects. One was a “Western” dietary pattern with high intake of red and processed meats, refined grains, and sweets, and the other was a “prudent” dietary pattern with high intake of vegetables, fruit, legumes, fish, poultry, and whole grains. Neither was associated with MS risk (relative risk for the highest vs. lowest quintile of scores, 0.71 and 1.09, respectively).
The results were similar when looking at mean cumulative dietary scores and when looking at the two cohorts separately, she said.
An exception in the analysis using mean cumulative dietary score was for the Western dietary pattern, which was shown to have a significant inverse association with MS risk (relative risk, 0.66). “This just met statistical significance, and we believe that this result is an artifact,” Dr. Rotstein said.
Study participants completed semiquantitative food frequency questionnaires every 4 years, beginning in 1984 for the Nurses’ Health Study (NHS) and in 1991 for NHS II. The MS cases were documented as of 2004 for NHS (130 cases) and as of 2009 for NHS II (350 cases).
With the exception of studies on vitamin D intake, prior studies have yielded null or inconsistent results with respect to the role of diet in MS development, and most have been limited by a focus on individual dietary elements and by small sample size.
The current study is the first large, prospective, population-based study to investigate the relationship, and it shows no evidence of an association between overall dietary quality and MS, she said.
The study is limited by the inherent subjectivity in dietary scores and by a basis in population norms rather than ideal consumption patterns. Also, the study is conceived around recommendations for cardiovascular health, but other dietary patterns may be more relevant for immunological health, she said, noting that additional study is needed to determine if dietary quality and individual dietary elements in the early years play a role in MS development.
Dr. Rotstein reported having no disclosures.
BOSTON – Diet does not appear to contribute to the development of multiple sclerosis, according to findings from more than 185,000 prospectively followed women from the Nurses’ Health Study and the Nurses’ Health Study II.
None of three dietary indices measured at baseline – the Alternative Healthy Eating Index-2010 (AHEI-2010), the Alternate Mediterranean Diet index (aMED), and the Dietary Approaches to Stop Hypertension index (DASH) – were significantly associated with the risk of developing multiple sclerosis in the two longitudinal cohort studies, Dr. Dalia Rotstein reported at the a joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
After adjustment for known confounders of multiple sclerosis (MS) risk, including age, latitude of residence at age 15 years, body mass index at age 18 years, pack-years of smoking, total energy intake, supplemental vitamin D intake, and ethnicity, the pooled relative risk of MS for the highest vs. the lowest quintile of scores for each index was 0.89 for the AHEI-2010, 1.10 for aMED, and 1.30 for DASH, said Dr. Rotstein of Brigham and Women’s Hospital, Boston.
A principal-components analysis identified two general dietary patterns among the subjects. One was a “Western” dietary pattern with high intake of red and processed meats, refined grains, and sweets, and the other was a “prudent” dietary pattern with high intake of vegetables, fruit, legumes, fish, poultry, and whole grains. Neither was associated with MS risk (relative risk for the highest vs. lowest quintile of scores, 0.71 and 1.09, respectively).
The results were similar when looking at mean cumulative dietary scores and when looking at the two cohorts separately, she said.
An exception in the analysis using mean cumulative dietary score was for the Western dietary pattern, which was shown to have a significant inverse association with MS risk (relative risk, 0.66). “This just met statistical significance, and we believe that this result is an artifact,” Dr. Rotstein said.
Study participants completed semiquantitative food frequency questionnaires every 4 years, beginning in 1984 for the Nurses’ Health Study (NHS) and in 1991 for NHS II. The MS cases were documented as of 2004 for NHS (130 cases) and as of 2009 for NHS II (350 cases).
With the exception of studies on vitamin D intake, prior studies have yielded null or inconsistent results with respect to the role of diet in MS development, and most have been limited by a focus on individual dietary elements and by small sample size.
The current study is the first large, prospective, population-based study to investigate the relationship, and it shows no evidence of an association between overall dietary quality and MS, she said.
The study is limited by the inherent subjectivity in dietary scores and by a basis in population norms rather than ideal consumption patterns. Also, the study is conceived around recommendations for cardiovascular health, but other dietary patterns may be more relevant for immunological health, she said, noting that additional study is needed to determine if dietary quality and individual dietary elements in the early years play a role in MS development.
Dr. Rotstein reported having no disclosures.
BOSTON – Diet does not appear to contribute to the development of multiple sclerosis, according to findings from more than 185,000 prospectively followed women from the Nurses’ Health Study and the Nurses’ Health Study II.
None of three dietary indices measured at baseline – the Alternative Healthy Eating Index-2010 (AHEI-2010), the Alternate Mediterranean Diet index (aMED), and the Dietary Approaches to Stop Hypertension index (DASH) – were significantly associated with the risk of developing multiple sclerosis in the two longitudinal cohort studies, Dr. Dalia Rotstein reported at the a joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
After adjustment for known confounders of multiple sclerosis (MS) risk, including age, latitude of residence at age 15 years, body mass index at age 18 years, pack-years of smoking, total energy intake, supplemental vitamin D intake, and ethnicity, the pooled relative risk of MS for the highest vs. the lowest quintile of scores for each index was 0.89 for the AHEI-2010, 1.10 for aMED, and 1.30 for DASH, said Dr. Rotstein of Brigham and Women’s Hospital, Boston.
A principal-components analysis identified two general dietary patterns among the subjects. One was a “Western” dietary pattern with high intake of red and processed meats, refined grains, and sweets, and the other was a “prudent” dietary pattern with high intake of vegetables, fruit, legumes, fish, poultry, and whole grains. Neither was associated with MS risk (relative risk for the highest vs. lowest quintile of scores, 0.71 and 1.09, respectively).
The results were similar when looking at mean cumulative dietary scores and when looking at the two cohorts separately, she said.
An exception in the analysis using mean cumulative dietary score was for the Western dietary pattern, which was shown to have a significant inverse association with MS risk (relative risk, 0.66). “This just met statistical significance, and we believe that this result is an artifact,” Dr. Rotstein said.
Study participants completed semiquantitative food frequency questionnaires every 4 years, beginning in 1984 for the Nurses’ Health Study (NHS) and in 1991 for NHS II. The MS cases were documented as of 2004 for NHS (130 cases) and as of 2009 for NHS II (350 cases).
With the exception of studies on vitamin D intake, prior studies have yielded null or inconsistent results with respect to the role of diet in MS development, and most have been limited by a focus on individual dietary elements and by small sample size.
The current study is the first large, prospective, population-based study to investigate the relationship, and it shows no evidence of an association between overall dietary quality and MS, she said.
The study is limited by the inherent subjectivity in dietary scores and by a basis in population norms rather than ideal consumption patterns. Also, the study is conceived around recommendations for cardiovascular health, but other dietary patterns may be more relevant for immunological health, she said, noting that additional study is needed to determine if dietary quality and individual dietary elements in the early years play a role in MS development.
Dr. Rotstein reported having no disclosures.
AT MSBOSTON 2014
Key clinical point: Evidence suggests that dietary quality does not influence risk for MS.
Major finding: The top vs. bottom quintile of scores for a “Western” diet and a “prudent” diet were not associated with relative risk values that reached statistical significance (0.71 and 1.09, respectively).
Data source: The Nurses’ Health Study and the Nurses’ Health Study II, involving more than 185,000 subjects.
Disclosures: Dr. Rotstein reported having no disclosures.
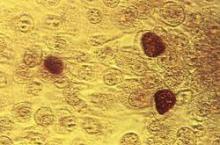
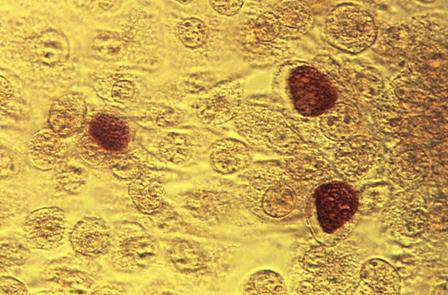
USPSTF releases updated recommendations for STI counseling, screening
Intensive behavioral counseling reduces risky sexual behaviors and the likelihood of sexually transmitted infections, and is recommended for all sexually active adolescents and adults who are at increased risk for acquiring or transmitting STIs, according to the U.S. Preventive Services Task Force.
In a separate but related recommendation statement, the USPSTF called for screening for chlamydia and gonorrhea in sexually active females aged 24 years or younger, and in older women at increased risk for infection.
Both recommendation statements were published online Sept. 23 in Annals of Internal Medicine.
The counseling recommendation updates a 2008 USPSTF recommendation, and is based on a review of the evidence for behavioral counseling in the primary care setting. The task force found adequate evidence in support of behavioral counseling, and concluded with moderate certainty that such counseling results in a moderate net benefit, reported Dr. Michael L. LeFevre, the task force chair and professor and associate chair of family medicine at the University of Missouri at Columbia.
The updated recommendation reaffirms the value of high-intensity behavioral counseling for those at increased risk of STIs and acknowledges that some interventions of lesser intensity are also effective.
Clinicians should be aware of populations at particularly high risk for STIs, according to the task force.
“African Americans have the highest STI prevalence of any racial/ethnic group, and STI prevalence is higher in American Indians, Alaska Natives, and Latinos than in white persons,” according to the recommendation statement (Ann. Internal. Med. 2014 Sept. 23 [doi:10.7326/M14-1965]).
Other high-risk groups include men who have sex with men, those with low incomes living in urban settings, current or former inmates, military recruits, those who exchange sex for money or drugs, those with mental illness or disability, those who use or have used drugs, those with a history of sexual abuse, and patients at public STI clinics.
Counseling interventions ranging in intensity from 30 minutes to 2 or more hours of contact time are beneficial, the task force said, noting that the evidence indicates that the benefits of counseling increase in tandem with the level of intensity.
Interventions can be delivered by primary care clinicians or by trained behavioral counselors, and the most successful approaches provide basic information about STIs and STI transmission, include risk assessment, and provide training in condom use, communication about safe sex, problem solving, and goal setting.
Interventions can include face-to-face counseling, videos, written materials, and telephone support.
The recommendation statement is based on a commissioned systematic review of the literature, which included 31 trials that reported STI outcomes and/or sexual behavioral outcomes in adolescents or adults (Ann. Internal Med. 2014 Sept. 23 [doi:10.7326/M14-0475]).
The chlamydia and gonorrhea screening recommendation statement updates a 2007 chlamydia recommendation and expands the 2005 gonorrhea recommendation to include all sexually active females aged 24 and younger. The updated statement is based on evidence that screening can accurately detect chlamydia and gonorrhea, and reduces complications of both infections with a moderate magnitude of benefit.
The review found insufficient evidence to assess the balance of benefits and harms of screening in men (Ann. Internal Med. 2014 Sept. 22 [doi:10.7326/M14-1981).
The highest infection rates occur in women aged 20-24 years, followed by those aged 15-19 years, the task force said.
“Chlamydia trachomatis and Neisseria gonorrhoeae infections should be diagnosed via nucleic acid amplification tests because their sensitivity and specificity are high and they are approved by the Food and Drug Administration for use on urogenital sites, including male and female urine, as well as clinician-collected endocervical, vaginal, and male urethral specimens,” according to the statement.
Evidence with respect to screening intervals is lacking, and a reasonable approach would be to screen patients whose sexual history reveals new or persistent risk factors since the last negative test results, the USPSTF said, adding that posttest counseling is an integral part of managing patients who are diagnosed with an STI.
The chlamydia and gonorrhea screening recommendation also is based on a commissioned systematic review of recent literature, including 10 fair-quality studies on diagnostic accuracy, 1 good-quality randomized controlled trial of sexually active young women, and large cohort studies of screening in pregnant women at increased risk for infection.
An estimated 20 million new cases of STIs occur each year in the United States, and half of these occur in adolescents and young adults aged 15-24 years. Chlamydia and gonorrhea are the most commonly reported STIs in the United States, with more than 1.4 million cases of chlamydia and more than 330,000 cases of gonorrhea reported in 2012, according to the USPSTF, which stated that these new recommendations are important because many STIs are asymptomatic and can result in serious sequelae such as pelvic inflammatory disease, infertility, and cancer.
Dr. LeFevre reported having no disclosures.
Intensive behavioral counseling reduces risky sexual behaviors and the likelihood of sexually transmitted infections, and is recommended for all sexually active adolescents and adults who are at increased risk for acquiring or transmitting STIs, according to the U.S. Preventive Services Task Force.
In a separate but related recommendation statement, the USPSTF called for screening for chlamydia and gonorrhea in sexually active females aged 24 years or younger, and in older women at increased risk for infection.
Both recommendation statements were published online Sept. 23 in Annals of Internal Medicine.
The counseling recommendation updates a 2008 USPSTF recommendation, and is based on a review of the evidence for behavioral counseling in the primary care setting. The task force found adequate evidence in support of behavioral counseling, and concluded with moderate certainty that such counseling results in a moderate net benefit, reported Dr. Michael L. LeFevre, the task force chair and professor and associate chair of family medicine at the University of Missouri at Columbia.
The updated recommendation reaffirms the value of high-intensity behavioral counseling for those at increased risk of STIs and acknowledges that some interventions of lesser intensity are also effective.
Clinicians should be aware of populations at particularly high risk for STIs, according to the task force.
“African Americans have the highest STI prevalence of any racial/ethnic group, and STI prevalence is higher in American Indians, Alaska Natives, and Latinos than in white persons,” according to the recommendation statement (Ann. Internal. Med. 2014 Sept. 23 [doi:10.7326/M14-1965]).
Other high-risk groups include men who have sex with men, those with low incomes living in urban settings, current or former inmates, military recruits, those who exchange sex for money or drugs, those with mental illness or disability, those who use or have used drugs, those with a history of sexual abuse, and patients at public STI clinics.
Counseling interventions ranging in intensity from 30 minutes to 2 or more hours of contact time are beneficial, the task force said, noting that the evidence indicates that the benefits of counseling increase in tandem with the level of intensity.
Interventions can be delivered by primary care clinicians or by trained behavioral counselors, and the most successful approaches provide basic information about STIs and STI transmission, include risk assessment, and provide training in condom use, communication about safe sex, problem solving, and goal setting.
Interventions can include face-to-face counseling, videos, written materials, and telephone support.
The recommendation statement is based on a commissioned systematic review of the literature, which included 31 trials that reported STI outcomes and/or sexual behavioral outcomes in adolescents or adults (Ann. Internal Med. 2014 Sept. 23 [doi:10.7326/M14-0475]).
The chlamydia and gonorrhea screening recommendation statement updates a 2007 chlamydia recommendation and expands the 2005 gonorrhea recommendation to include all sexually active females aged 24 and younger. The updated statement is based on evidence that screening can accurately detect chlamydia and gonorrhea, and reduces complications of both infections with a moderate magnitude of benefit.
The review found insufficient evidence to assess the balance of benefits and harms of screening in men (Ann. Internal Med. 2014 Sept. 22 [doi:10.7326/M14-1981).
The highest infection rates occur in women aged 20-24 years, followed by those aged 15-19 years, the task force said.
“Chlamydia trachomatis and Neisseria gonorrhoeae infections should be diagnosed via nucleic acid amplification tests because their sensitivity and specificity are high and they are approved by the Food and Drug Administration for use on urogenital sites, including male and female urine, as well as clinician-collected endocervical, vaginal, and male urethral specimens,” according to the statement.
Evidence with respect to screening intervals is lacking, and a reasonable approach would be to screen patients whose sexual history reveals new or persistent risk factors since the last negative test results, the USPSTF said, adding that posttest counseling is an integral part of managing patients who are diagnosed with an STI.
The chlamydia and gonorrhea screening recommendation also is based on a commissioned systematic review of recent literature, including 10 fair-quality studies on diagnostic accuracy, 1 good-quality randomized controlled trial of sexually active young women, and large cohort studies of screening in pregnant women at increased risk for infection.
An estimated 20 million new cases of STIs occur each year in the United States, and half of these occur in adolescents and young adults aged 15-24 years. Chlamydia and gonorrhea are the most commonly reported STIs in the United States, with more than 1.4 million cases of chlamydia and more than 330,000 cases of gonorrhea reported in 2012, according to the USPSTF, which stated that these new recommendations are important because many STIs are asymptomatic and can result in serious sequelae such as pelvic inflammatory disease, infertility, and cancer.
Dr. LeFevre reported having no disclosures.
Intensive behavioral counseling reduces risky sexual behaviors and the likelihood of sexually transmitted infections, and is recommended for all sexually active adolescents and adults who are at increased risk for acquiring or transmitting STIs, according to the U.S. Preventive Services Task Force.
In a separate but related recommendation statement, the USPSTF called for screening for chlamydia and gonorrhea in sexually active females aged 24 years or younger, and in older women at increased risk for infection.
Both recommendation statements were published online Sept. 23 in Annals of Internal Medicine.
The counseling recommendation updates a 2008 USPSTF recommendation, and is based on a review of the evidence for behavioral counseling in the primary care setting. The task force found adequate evidence in support of behavioral counseling, and concluded with moderate certainty that such counseling results in a moderate net benefit, reported Dr. Michael L. LeFevre, the task force chair and professor and associate chair of family medicine at the University of Missouri at Columbia.
The updated recommendation reaffirms the value of high-intensity behavioral counseling for those at increased risk of STIs and acknowledges that some interventions of lesser intensity are also effective.
Clinicians should be aware of populations at particularly high risk for STIs, according to the task force.
“African Americans have the highest STI prevalence of any racial/ethnic group, and STI prevalence is higher in American Indians, Alaska Natives, and Latinos than in white persons,” according to the recommendation statement (Ann. Internal. Med. 2014 Sept. 23 [doi:10.7326/M14-1965]).
Other high-risk groups include men who have sex with men, those with low incomes living in urban settings, current or former inmates, military recruits, those who exchange sex for money or drugs, those with mental illness or disability, those who use or have used drugs, those with a history of sexual abuse, and patients at public STI clinics.
Counseling interventions ranging in intensity from 30 minutes to 2 or more hours of contact time are beneficial, the task force said, noting that the evidence indicates that the benefits of counseling increase in tandem with the level of intensity.
Interventions can be delivered by primary care clinicians or by trained behavioral counselors, and the most successful approaches provide basic information about STIs and STI transmission, include risk assessment, and provide training in condom use, communication about safe sex, problem solving, and goal setting.
Interventions can include face-to-face counseling, videos, written materials, and telephone support.
The recommendation statement is based on a commissioned systematic review of the literature, which included 31 trials that reported STI outcomes and/or sexual behavioral outcomes in adolescents or adults (Ann. Internal Med. 2014 Sept. 23 [doi:10.7326/M14-0475]).
The chlamydia and gonorrhea screening recommendation statement updates a 2007 chlamydia recommendation and expands the 2005 gonorrhea recommendation to include all sexually active females aged 24 and younger. The updated statement is based on evidence that screening can accurately detect chlamydia and gonorrhea, and reduces complications of both infections with a moderate magnitude of benefit.
The review found insufficient evidence to assess the balance of benefits and harms of screening in men (Ann. Internal Med. 2014 Sept. 22 [doi:10.7326/M14-1981).
The highest infection rates occur in women aged 20-24 years, followed by those aged 15-19 years, the task force said.
“Chlamydia trachomatis and Neisseria gonorrhoeae infections should be diagnosed via nucleic acid amplification tests because their sensitivity and specificity are high and they are approved by the Food and Drug Administration for use on urogenital sites, including male and female urine, as well as clinician-collected endocervical, vaginal, and male urethral specimens,” according to the statement.
Evidence with respect to screening intervals is lacking, and a reasonable approach would be to screen patients whose sexual history reveals new or persistent risk factors since the last negative test results, the USPSTF said, adding that posttest counseling is an integral part of managing patients who are diagnosed with an STI.
The chlamydia and gonorrhea screening recommendation also is based on a commissioned systematic review of recent literature, including 10 fair-quality studies on diagnostic accuracy, 1 good-quality randomized controlled trial of sexually active young women, and large cohort studies of screening in pregnant women at increased risk for infection.
An estimated 20 million new cases of STIs occur each year in the United States, and half of these occur in adolescents and young adults aged 15-24 years. Chlamydia and gonorrhea are the most commonly reported STIs in the United States, with more than 1.4 million cases of chlamydia and more than 330,000 cases of gonorrhea reported in 2012, according to the USPSTF, which stated that these new recommendations are important because many STIs are asymptomatic and can result in serious sequelae such as pelvic inflammatory disease, infertility, and cancer.
Dr. LeFevre reported having no disclosures.
FROM ANNALS OF INTERNAL MEDICINE
Trial probes mechanism of THC-CBD improvement of multiple sclerosis spasticity
BOSTON – A tetrahydrocannabinol and cannabidiol (THC-CBD) oromucosal spray significantly improved spasticity symptoms in a randomized, placebo-controlled study of 43 multiple sclerosis patients.
The spray, which goes by the trade name Sativex, has been shown to reduce symptoms associated with spasticity in MS, and is approved in several European countries and commercially licensed in many others, including the United States, where it received fast-track status from the Food and Drug Administration earlier this year. But little has been known about the correlates of these effects on objective measures of spasticity or corticospinal excitability, Dr. Letizia Leocani said at the joint meeting of the Americas and European Committees for Treatment and Research in Multiple Sclerosis.
The findings of the study suggest, however, that mechanisms other than corticospinal excitability and the monosynaptic component of the stretch reflex may play a role in spasticity, Dr. Leocani reported.
The treatment effect, as measured by the modified Ashworth Scale (MAS), was significantly greater in those who were randomized to receive active treatment, compared with those who received placebo (change in MAS score, –1.51 vs. 0.16, respectively). Patients in the treatment group improved by about 18%; those in the placebo group improved by less than 7%, said Dr. Leocani, clinical group leader of the experimental neurophysiology unit of San Raffaele Hospital, Milan.
A 20% or better response on the MAS occurred in 44% of patients on active treatment, compared with about 15% of those who received placebo.
Patients in the study were adults with progressive MS and clinical evidence of spasticity. They were randomized in double-blind fashion to a 2-week titration period plus 2 weeks of stable dose treatment with either Sativex or a placebo formulation. A second double-blind, crossover cycle followed after another 2-week washout.
The MAS and several other clinical neurophysiological measures – including spasticity and pain numeric rating scales (NRS) - were obtained before and after each cycle. MAS improvement and improvement in NRS spasticity were significantly correlated (r = 0.38), but other neurophysiological measures did not differ in the treatment and placebo groups, and were not correlated with clinical parameters, with the exception of a trend between percent change in the MAS and in the bilateral soleus ratio of maximum H reflex to maximum M response.
While the current findings confirm the clinical benefit of treatment for spasticity, there was a lack of corresponding changes on measures of corticospinal excitability and on the monosynaptic component of the stretch reflex; these findings point to a role for – and a need for exploration of – other spinal and supraspinal mechanisms in spasticity physiopathology, she concluded.
Dr. Leocani reported having no relevant disclosures.
BOSTON – A tetrahydrocannabinol and cannabidiol (THC-CBD) oromucosal spray significantly improved spasticity symptoms in a randomized, placebo-controlled study of 43 multiple sclerosis patients.
The spray, which goes by the trade name Sativex, has been shown to reduce symptoms associated with spasticity in MS, and is approved in several European countries and commercially licensed in many others, including the United States, where it received fast-track status from the Food and Drug Administration earlier this year. But little has been known about the correlates of these effects on objective measures of spasticity or corticospinal excitability, Dr. Letizia Leocani said at the joint meeting of the Americas and European Committees for Treatment and Research in Multiple Sclerosis.
The findings of the study suggest, however, that mechanisms other than corticospinal excitability and the monosynaptic component of the stretch reflex may play a role in spasticity, Dr. Leocani reported.
The treatment effect, as measured by the modified Ashworth Scale (MAS), was significantly greater in those who were randomized to receive active treatment, compared with those who received placebo (change in MAS score, –1.51 vs. 0.16, respectively). Patients in the treatment group improved by about 18%; those in the placebo group improved by less than 7%, said Dr. Leocani, clinical group leader of the experimental neurophysiology unit of San Raffaele Hospital, Milan.
A 20% or better response on the MAS occurred in 44% of patients on active treatment, compared with about 15% of those who received placebo.
Patients in the study were adults with progressive MS and clinical evidence of spasticity. They were randomized in double-blind fashion to a 2-week titration period plus 2 weeks of stable dose treatment with either Sativex or a placebo formulation. A second double-blind, crossover cycle followed after another 2-week washout.
The MAS and several other clinical neurophysiological measures – including spasticity and pain numeric rating scales (NRS) - were obtained before and after each cycle. MAS improvement and improvement in NRS spasticity were significantly correlated (r = 0.38), but other neurophysiological measures did not differ in the treatment and placebo groups, and were not correlated with clinical parameters, with the exception of a trend between percent change in the MAS and in the bilateral soleus ratio of maximum H reflex to maximum M response.
While the current findings confirm the clinical benefit of treatment for spasticity, there was a lack of corresponding changes on measures of corticospinal excitability and on the monosynaptic component of the stretch reflex; these findings point to a role for – and a need for exploration of – other spinal and supraspinal mechanisms in spasticity physiopathology, she concluded.
Dr. Leocani reported having no relevant disclosures.
BOSTON – A tetrahydrocannabinol and cannabidiol (THC-CBD) oromucosal spray significantly improved spasticity symptoms in a randomized, placebo-controlled study of 43 multiple sclerosis patients.
The spray, which goes by the trade name Sativex, has been shown to reduce symptoms associated with spasticity in MS, and is approved in several European countries and commercially licensed in many others, including the United States, where it received fast-track status from the Food and Drug Administration earlier this year. But little has been known about the correlates of these effects on objective measures of spasticity or corticospinal excitability, Dr. Letizia Leocani said at the joint meeting of the Americas and European Committees for Treatment and Research in Multiple Sclerosis.
The findings of the study suggest, however, that mechanisms other than corticospinal excitability and the monosynaptic component of the stretch reflex may play a role in spasticity, Dr. Leocani reported.
The treatment effect, as measured by the modified Ashworth Scale (MAS), was significantly greater in those who were randomized to receive active treatment, compared with those who received placebo (change in MAS score, –1.51 vs. 0.16, respectively). Patients in the treatment group improved by about 18%; those in the placebo group improved by less than 7%, said Dr. Leocani, clinical group leader of the experimental neurophysiology unit of San Raffaele Hospital, Milan.
A 20% or better response on the MAS occurred in 44% of patients on active treatment, compared with about 15% of those who received placebo.
Patients in the study were adults with progressive MS and clinical evidence of spasticity. They were randomized in double-blind fashion to a 2-week titration period plus 2 weeks of stable dose treatment with either Sativex or a placebo formulation. A second double-blind, crossover cycle followed after another 2-week washout.
The MAS and several other clinical neurophysiological measures – including spasticity and pain numeric rating scales (NRS) - were obtained before and after each cycle. MAS improvement and improvement in NRS spasticity were significantly correlated (r = 0.38), but other neurophysiological measures did not differ in the treatment and placebo groups, and were not correlated with clinical parameters, with the exception of a trend between percent change in the MAS and in the bilateral soleus ratio of maximum H reflex to maximum M response.
While the current findings confirm the clinical benefit of treatment for spasticity, there was a lack of corresponding changes on measures of corticospinal excitability and on the monosynaptic component of the stretch reflex; these findings point to a role for – and a need for exploration of – other spinal and supraspinal mechanisms in spasticity physiopathology, she concluded.
Dr. Leocani reported having no relevant disclosures.
AT MSBOSTON 2014
Key clinical point: Sativex reduces symptoms of MS spasticity but does not induce corresponding changes on measures of corticospinal excitability and on the monosynaptic component of the stretch reflex.
Major finding: A 20% or better response on the MAS occurred in 44% of patients on active treatment, compared with about 15% of those who received placebo.
Data source: A randomized, placebo-controlled study of 43 patients.
Disclosures: Dr. Leocani reported having no relevant disclosures.
Stepwise approach improves diagnostic accuracy in polymyalgia rheumatica
LAS VEGAS – Up to 30% of patients diagnosed with polymyalgia rheumatica actually have a different disease, according to Dr. Eric L. Matteson.
The proximal pain and stiffness syndrome, which is a commonly accepted phenotype of polymyalgia rheumatica (PMR), also occurs in many other rheumatologic and inflammatory illnesses, Dr. Matteson said at the annual Perspectives in Rheumatic Diseases. Laboratory tests are nonspecific, and there is significant overlap – in terms of pathobiology – with other forms of inflammatory diseases associated with synovitis and vasculitis. Further, multiple conditions can coexist – including osteoarthritis, rotator cuff lesions, secondary capsulitis, and other periarticular lesions – and several conditions can mimic PMR.
Dr. Matteson of the Mayo Clinic College of Medicine, Rochester, Minn., described one 73-year-old patient with “discitis” who was initially thought to have PMR. She was admitted to the hospital with acute stiffness in the shoulder and acute onset backache. In keeping with a PMR diagnosis, her C-reactive protein (CRP) level was 113 mg/L, and she responded to 20 mg of prednisolone.
Another case involved a 63-year-old man with metastatic cancer. He presented with typical symptoms of PMR, a CRP level of 150 mg/L, and an alkaline phosphatase level of 1.5 times the upper limit of normal. He did not respond to 15 mg/day of prednisolone but felt better on 30 mg/day, with alkaline phosphatase rising to three times the upper limit of normal.
A third case involved a patient with antineutrophil cytoplasmic autoantibody vasculitis who was initially thought to have PMR. The patient presented with constitutional symptoms and was started on 15 mg/day of oral prednisone. Hemoptysis occurred 4 weeks later.
In an effort to improve diagnosis, the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) developed consensus candidate classification criteria. An expert panel that included Dr. Matteson had recognized that opinions differ as to the definition of PMR and its treatment, and that classification criteria are needed. In pursuit of this goal, the panel identified seven core criteria that 70% of survey respondents agreed upon, including age of 50 years or older, duration of at least 2 weeks, bilateral shoulder and/or pelvic girdle aching, morning stiffness duration of more than 45 minutes, elevated erythrocyte sedimentation rate (ESR), elevated CRP level, and rapid corticosteroid response with greater than 75% global response within 1 week to 15-20 mg of prednisolone or prednisone daily.
In addition, more than 70% of survey respondents agreed on the importance of assessing shoulder pain and limitation and/or hip motion. Agreement was low for peripheral signs such as carpal tunnel syndrome, tenosynovitis, and peripheral arthritis (J. Rheumatol. 2008;35:270-7).
Dr. Matteson suggested a stepwise approach to diagnosis, which includes evaluating for inclusion criteria, evaluating for exclusion criteria, prescribing steroids and evaluating patient response, and confirming the diagnosis at follow-up.
Inclusion criteria include age of 50 years or older, bilateral shoulder or pelvic girdle aching, morning stiffness for at least 45 minutes, symptom duration of at least 2 weeks, and acute phase response with an ESR greater than 30 mm/hr or a CRP level greater than 10 mg/L. All of these features must be present, he said at the conference held by Global Academy for Medical Education.
The panel also found that typical findings of synovitis in the shoulder and hip regions can assist in the classification of PMR.
Core exclusion criteria include active infection, neoplasia, and giant cell arteritis. Other criteria that reduce the likelihood that PMR is present include rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, and inflammatory myopathy.
Drug-induced myalgia, pain syndromes, endocrine disorders, and Parkinson’s disease are mimics of PMR and must be excluded.
Corticosteroid response should be carefully assessed in those thought to have PMR, and the response should be rapid, complete, and sustained. Especially high doses increase the probability of diagnostic error, and nonresponse is an indication for reevaluation of the diagnosis and reassessment of the disease, Dr. Matteson said.
A scoring algorithm that incorporates ultrasound findings is also useful in the evaluation of a patient with suspected PMR. In patients aged 50 years or older with bilateral shoulder aching and abnormal ESR/CRP levels, points are awarded as follows: 2 points for morning stiffness for more than 45 minutes, 1 point for hip pain or limited range of motion, 2 points for normal rheumatoid factor or anticitrullinated peptide antibodies (ACPAs), 1 point for the absence of other joint pain. Further, 1 point is awarded if ultrasound identifies subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis in at least one shoulder. Also, 1 point is awarded if ultrasound shows subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis in both shoulders.
A score of 5 using this algorithm has 71% sensitivity and 70% specificity for discriminating all comparison subjects from PMR. The specificity is higher (85%) for discriminating shoulder conditions from PMR, and lower (65%) for discriminating rheumatoid arthritis from PMR, Dr. Matteson noted.
Clues that a patient does not have PMR include younger age, chronic onset, male gender, severe constitutional symptoms, lack of shoulder involvement, peripheral arthritis, rheumatoid factor–positive/ACPA status, normal acute phase reactants (CRP, ESR) levels, peripheral arthritis, foot and ankle involvement, and poor response to low-dose corticosteroids. Also, keep in mind that the closest mimic of PMR is late-onset spondyloarthropathy, he said.
The severity of PMR is based on the intensity of pain, stiffness, and disability. Comorbidities should also be assessed, as these may affect the choice of corticosteroid dose. The corticosteroid dose, as well as other therapy, should be individualized based on the severity and comorbidities, and on the patient’s preference.
Patients diagnosed with PMR should be educated about the disease, its treatment and the potential related complications, and about precautions and monitoring requirements, Dr. Matteson said. Range-of-motion exercises for the shoulder and pelvic girdle muscles should be encouraged, and referral to physiotherapy should be made if necessary.
Patients should be monitored for relapse; in a recent study, relapse-free survival at 1 year and 2 years among patients with PMR was 68.8% and 42.4%, respectively, he noted.
Dr. Matteson reported serving as an advisory board member for Janssen; providing editorial services for UpToDate and inPractice; receiving research grants from the ACR, EULAR, Celgene, Biogen Idec, Centocor/Janssen, Genentech, Novartis, Roche, Ardea Biosciences, UCB Group, Sanofi, the Veterans Administration, the National Institutes of Health, Pfizer, and Mesoblast; and owning stock in Exact Sciences.
Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Up to 30% of patients diagnosed with polymyalgia rheumatica actually have a different disease, according to Dr. Eric L. Matteson.
The proximal pain and stiffness syndrome, which is a commonly accepted phenotype of polymyalgia rheumatica (PMR), also occurs in many other rheumatologic and inflammatory illnesses, Dr. Matteson said at the annual Perspectives in Rheumatic Diseases. Laboratory tests are nonspecific, and there is significant overlap – in terms of pathobiology – with other forms of inflammatory diseases associated with synovitis and vasculitis. Further, multiple conditions can coexist – including osteoarthritis, rotator cuff lesions, secondary capsulitis, and other periarticular lesions – and several conditions can mimic PMR.
Dr. Matteson of the Mayo Clinic College of Medicine, Rochester, Minn., described one 73-year-old patient with “discitis” who was initially thought to have PMR. She was admitted to the hospital with acute stiffness in the shoulder and acute onset backache. In keeping with a PMR diagnosis, her C-reactive protein (CRP) level was 113 mg/L, and she responded to 20 mg of prednisolone.
Another case involved a 63-year-old man with metastatic cancer. He presented with typical symptoms of PMR, a CRP level of 150 mg/L, and an alkaline phosphatase level of 1.5 times the upper limit of normal. He did not respond to 15 mg/day of prednisolone but felt better on 30 mg/day, with alkaline phosphatase rising to three times the upper limit of normal.
A third case involved a patient with antineutrophil cytoplasmic autoantibody vasculitis who was initially thought to have PMR. The patient presented with constitutional symptoms and was started on 15 mg/day of oral prednisone. Hemoptysis occurred 4 weeks later.
In an effort to improve diagnosis, the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) developed consensus candidate classification criteria. An expert panel that included Dr. Matteson had recognized that opinions differ as to the definition of PMR and its treatment, and that classification criteria are needed. In pursuit of this goal, the panel identified seven core criteria that 70% of survey respondents agreed upon, including age of 50 years or older, duration of at least 2 weeks, bilateral shoulder and/or pelvic girdle aching, morning stiffness duration of more than 45 minutes, elevated erythrocyte sedimentation rate (ESR), elevated CRP level, and rapid corticosteroid response with greater than 75% global response within 1 week to 15-20 mg of prednisolone or prednisone daily.
In addition, more than 70% of survey respondents agreed on the importance of assessing shoulder pain and limitation and/or hip motion. Agreement was low for peripheral signs such as carpal tunnel syndrome, tenosynovitis, and peripheral arthritis (J. Rheumatol. 2008;35:270-7).
Dr. Matteson suggested a stepwise approach to diagnosis, which includes evaluating for inclusion criteria, evaluating for exclusion criteria, prescribing steroids and evaluating patient response, and confirming the diagnosis at follow-up.
Inclusion criteria include age of 50 years or older, bilateral shoulder or pelvic girdle aching, morning stiffness for at least 45 minutes, symptom duration of at least 2 weeks, and acute phase response with an ESR greater than 30 mm/hr or a CRP level greater than 10 mg/L. All of these features must be present, he said at the conference held by Global Academy for Medical Education.
The panel also found that typical findings of synovitis in the shoulder and hip regions can assist in the classification of PMR.
Core exclusion criteria include active infection, neoplasia, and giant cell arteritis. Other criteria that reduce the likelihood that PMR is present include rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, and inflammatory myopathy.
Drug-induced myalgia, pain syndromes, endocrine disorders, and Parkinson’s disease are mimics of PMR and must be excluded.
Corticosteroid response should be carefully assessed in those thought to have PMR, and the response should be rapid, complete, and sustained. Especially high doses increase the probability of diagnostic error, and nonresponse is an indication for reevaluation of the diagnosis and reassessment of the disease, Dr. Matteson said.
A scoring algorithm that incorporates ultrasound findings is also useful in the evaluation of a patient with suspected PMR. In patients aged 50 years or older with bilateral shoulder aching and abnormal ESR/CRP levels, points are awarded as follows: 2 points for morning stiffness for more than 45 minutes, 1 point for hip pain or limited range of motion, 2 points for normal rheumatoid factor or anticitrullinated peptide antibodies (ACPAs), 1 point for the absence of other joint pain. Further, 1 point is awarded if ultrasound identifies subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis in at least one shoulder. Also, 1 point is awarded if ultrasound shows subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis in both shoulders.
A score of 5 using this algorithm has 71% sensitivity and 70% specificity for discriminating all comparison subjects from PMR. The specificity is higher (85%) for discriminating shoulder conditions from PMR, and lower (65%) for discriminating rheumatoid arthritis from PMR, Dr. Matteson noted.
Clues that a patient does not have PMR include younger age, chronic onset, male gender, severe constitutional symptoms, lack of shoulder involvement, peripheral arthritis, rheumatoid factor–positive/ACPA status, normal acute phase reactants (CRP, ESR) levels, peripheral arthritis, foot and ankle involvement, and poor response to low-dose corticosteroids. Also, keep in mind that the closest mimic of PMR is late-onset spondyloarthropathy, he said.
The severity of PMR is based on the intensity of pain, stiffness, and disability. Comorbidities should also be assessed, as these may affect the choice of corticosteroid dose. The corticosteroid dose, as well as other therapy, should be individualized based on the severity and comorbidities, and on the patient’s preference.
Patients diagnosed with PMR should be educated about the disease, its treatment and the potential related complications, and about precautions and monitoring requirements, Dr. Matteson said. Range-of-motion exercises for the shoulder and pelvic girdle muscles should be encouraged, and referral to physiotherapy should be made if necessary.
Patients should be monitored for relapse; in a recent study, relapse-free survival at 1 year and 2 years among patients with PMR was 68.8% and 42.4%, respectively, he noted.
Dr. Matteson reported serving as an advisory board member for Janssen; providing editorial services for UpToDate and inPractice; receiving research grants from the ACR, EULAR, Celgene, Biogen Idec, Centocor/Janssen, Genentech, Novartis, Roche, Ardea Biosciences, UCB Group, Sanofi, the Veterans Administration, the National Institutes of Health, Pfizer, and Mesoblast; and owning stock in Exact Sciences.
Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
LAS VEGAS – Up to 30% of patients diagnosed with polymyalgia rheumatica actually have a different disease, according to Dr. Eric L. Matteson.
The proximal pain and stiffness syndrome, which is a commonly accepted phenotype of polymyalgia rheumatica (PMR), also occurs in many other rheumatologic and inflammatory illnesses, Dr. Matteson said at the annual Perspectives in Rheumatic Diseases. Laboratory tests are nonspecific, and there is significant overlap – in terms of pathobiology – with other forms of inflammatory diseases associated with synovitis and vasculitis. Further, multiple conditions can coexist – including osteoarthritis, rotator cuff lesions, secondary capsulitis, and other periarticular lesions – and several conditions can mimic PMR.
Dr. Matteson of the Mayo Clinic College of Medicine, Rochester, Minn., described one 73-year-old patient with “discitis” who was initially thought to have PMR. She was admitted to the hospital with acute stiffness in the shoulder and acute onset backache. In keeping with a PMR diagnosis, her C-reactive protein (CRP) level was 113 mg/L, and she responded to 20 mg of prednisolone.
Another case involved a 63-year-old man with metastatic cancer. He presented with typical symptoms of PMR, a CRP level of 150 mg/L, and an alkaline phosphatase level of 1.5 times the upper limit of normal. He did not respond to 15 mg/day of prednisolone but felt better on 30 mg/day, with alkaline phosphatase rising to three times the upper limit of normal.
A third case involved a patient with antineutrophil cytoplasmic autoantibody vasculitis who was initially thought to have PMR. The patient presented with constitutional symptoms and was started on 15 mg/day of oral prednisone. Hemoptysis occurred 4 weeks later.
In an effort to improve diagnosis, the American College of Rheumatology (ACR) and the European League Against Rheumatism (EULAR) developed consensus candidate classification criteria. An expert panel that included Dr. Matteson had recognized that opinions differ as to the definition of PMR and its treatment, and that classification criteria are needed. In pursuit of this goal, the panel identified seven core criteria that 70% of survey respondents agreed upon, including age of 50 years or older, duration of at least 2 weeks, bilateral shoulder and/or pelvic girdle aching, morning stiffness duration of more than 45 minutes, elevated erythrocyte sedimentation rate (ESR), elevated CRP level, and rapid corticosteroid response with greater than 75% global response within 1 week to 15-20 mg of prednisolone or prednisone daily.
In addition, more than 70% of survey respondents agreed on the importance of assessing shoulder pain and limitation and/or hip motion. Agreement was low for peripheral signs such as carpal tunnel syndrome, tenosynovitis, and peripheral arthritis (J. Rheumatol. 2008;35:270-7).
Dr. Matteson suggested a stepwise approach to diagnosis, which includes evaluating for inclusion criteria, evaluating for exclusion criteria, prescribing steroids and evaluating patient response, and confirming the diagnosis at follow-up.
Inclusion criteria include age of 50 years or older, bilateral shoulder or pelvic girdle aching, morning stiffness for at least 45 minutes, symptom duration of at least 2 weeks, and acute phase response with an ESR greater than 30 mm/hr or a CRP level greater than 10 mg/L. All of these features must be present, he said at the conference held by Global Academy for Medical Education.
The panel also found that typical findings of synovitis in the shoulder and hip regions can assist in the classification of PMR.
Core exclusion criteria include active infection, neoplasia, and giant cell arteritis. Other criteria that reduce the likelihood that PMR is present include rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, and inflammatory myopathy.
Drug-induced myalgia, pain syndromes, endocrine disorders, and Parkinson’s disease are mimics of PMR and must be excluded.
Corticosteroid response should be carefully assessed in those thought to have PMR, and the response should be rapid, complete, and sustained. Especially high doses increase the probability of diagnostic error, and nonresponse is an indication for reevaluation of the diagnosis and reassessment of the disease, Dr. Matteson said.
A scoring algorithm that incorporates ultrasound findings is also useful in the evaluation of a patient with suspected PMR. In patients aged 50 years or older with bilateral shoulder aching and abnormal ESR/CRP levels, points are awarded as follows: 2 points for morning stiffness for more than 45 minutes, 1 point for hip pain or limited range of motion, 2 points for normal rheumatoid factor or anticitrullinated peptide antibodies (ACPAs), 1 point for the absence of other joint pain. Further, 1 point is awarded if ultrasound identifies subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis in at least one shoulder. Also, 1 point is awarded if ultrasound shows subdeltoid bursitis, biceps tenosynovitis, or glenohumeral synovitis in both shoulders.
A score of 5 using this algorithm has 71% sensitivity and 70% specificity for discriminating all comparison subjects from PMR. The specificity is higher (85%) for discriminating shoulder conditions from PMR, and lower (65%) for discriminating rheumatoid arthritis from PMR, Dr. Matteson noted.
Clues that a patient does not have PMR include younger age, chronic onset, male gender, severe constitutional symptoms, lack of shoulder involvement, peripheral arthritis, rheumatoid factor–positive/ACPA status, normal acute phase reactants (CRP, ESR) levels, peripheral arthritis, foot and ankle involvement, and poor response to low-dose corticosteroids. Also, keep in mind that the closest mimic of PMR is late-onset spondyloarthropathy, he said.
The severity of PMR is based on the intensity of pain, stiffness, and disability. Comorbidities should also be assessed, as these may affect the choice of corticosteroid dose. The corticosteroid dose, as well as other therapy, should be individualized based on the severity and comorbidities, and on the patient’s preference.
Patients diagnosed with PMR should be educated about the disease, its treatment and the potential related complications, and about precautions and monitoring requirements, Dr. Matteson said. Range-of-motion exercises for the shoulder and pelvic girdle muscles should be encouraged, and referral to physiotherapy should be made if necessary.
Patients should be monitored for relapse; in a recent study, relapse-free survival at 1 year and 2 years among patients with PMR was 68.8% and 42.4%, respectively, he noted.
Dr. Matteson reported serving as an advisory board member for Janssen; providing editorial services for UpToDate and inPractice; receiving research grants from the ACR, EULAR, Celgene, Biogen Idec, Centocor/Janssen, Genentech, Novartis, Roche, Ardea Biosciences, UCB Group, Sanofi, the Veterans Administration, the National Institutes of Health, Pfizer, and Mesoblast; and owning stock in Exact Sciences.
Global Academy for Medical Education and this news organization are owned by Frontline Medical Communications.
EXPERT ANALYSIS FROM THE ANNUAL PERSPECTIVES IN RHEUMATIC DISEASES
Interferon beta-1a, glatiramer acetate prove cost effective for long-term MS treatment
BOSTON - Treatment with interferon beta-1a or glatiramer acetate, if used long term, has the potential to be cost effective and to alter the natural history of relapsing-remitting multiple sclerosis, according to findings from a 6-year observational study.
In 4,137 patients with relapsing-remitting MS who were followed for a median of 6 years, the observed increase in the Expanded Disability Status Scale (EDSS) scores was lower than the increase seen in a natural history cohort that served as a comparator group (relative rates, 0.76 using a continuous Markov model and 0.61 using a multilevel model). The observed progression for utility – a quality of life measure derived from the EDSS scores, and the primary outcome of the study – was consistent with this finding (relative rates of 0.58 and 0.56 according to the two models, respectively), Dr. Jacqueline Palace reported at the joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The outcome – essentially a reduction of about 42% on the primary outcome measure, compared with the natural history cohort – was better than the 38% reduction that was predicted, said Dr. Palace, leader of the multiple sclerosis and neuromyelitis group at Oxford University Hospitals Trust in England.
A range of sensitivity analyses that examined the potential for various biases consistently demonstrated better outcomes in the treated patients, compared with the natural history cohort, Dr. Palace noted.
If sustained over 20 years, the magnitude of the treatment effect observed in this study would be consistent with a predefined cost-effectiveness target of about $58,400 per quality-adjusted life-year, she said.
Patients included in this study were adults with a mean age of 38 years and a mean age at disease onset of 31 years. Most (76%) were women. Disease duration was a mean of 7.7 years at baseline, and the patients had a mean of three relapses over the prior 2 years. Baseline EDSS score was 3.06.
“In 2002, the National Institute of Clinical Excellence [NICE], which is the U.K. body that sets the guidelines that decide what should be available under the National Health Service, concluded that beta-interferon and glatiramer acetate were not cost effective during the short-term, and thus it recommended that it couldn’t be available under the NHS at that stage,” Dr. Palace explained.
The NICE did notice, however, that if the drug effect was maintained over the long term it might be cost effective and invited the Department of Health to “try and find a way to make the drugs available in a cost-effective manner,” she said.
“This was the birth of the Risk-Sharing Scheme, and it is a way of providing disease-modifying therapies to patients cost effectively,” she said.
At the start of the scheme, the drug prices were reduced in line with a cost-effective requirement using the NICE model, which equated to about $58,400/quality-adjusted life-year..
“We were allowed to reach this target over a 20-year lifespan, and the idea was to monitor a clinical cohort of patients, measuring their EDSS scores,” she said.
The natural history dataset was used as a “virtual placebo,” and a price adjustment would be required if the outcomes of the treated patients were less than predicted to be on target for cost effectiveness.
The findings of this study – the largest observational study to measure the effect and cost-effectiveness of interferon beta-1a and glatiramer acetate – provide evidence that the treatment alters the natural history of relapsing-remitting MS in the real life setting, she concluded.
Dr. Palace reported receiving support for scientific meetings and honorariums for advisory work from Teva, which makes glatiramer acetate, and Bayer Schering, which makes interferon beta-1a, as well as other companies that market drugs for MS. She’s also received unrestricted grants from Bayer Schering and other companies that market drugs for MS.
BOSTON - Treatment with interferon beta-1a or glatiramer acetate, if used long term, has the potential to be cost effective and to alter the natural history of relapsing-remitting multiple sclerosis, according to findings from a 6-year observational study.
In 4,137 patients with relapsing-remitting MS who were followed for a median of 6 years, the observed increase in the Expanded Disability Status Scale (EDSS) scores was lower than the increase seen in a natural history cohort that served as a comparator group (relative rates, 0.76 using a continuous Markov model and 0.61 using a multilevel model). The observed progression for utility – a quality of life measure derived from the EDSS scores, and the primary outcome of the study – was consistent with this finding (relative rates of 0.58 and 0.56 according to the two models, respectively), Dr. Jacqueline Palace reported at the joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The outcome – essentially a reduction of about 42% on the primary outcome measure, compared with the natural history cohort – was better than the 38% reduction that was predicted, said Dr. Palace, leader of the multiple sclerosis and neuromyelitis group at Oxford University Hospitals Trust in England.
A range of sensitivity analyses that examined the potential for various biases consistently demonstrated better outcomes in the treated patients, compared with the natural history cohort, Dr. Palace noted.
If sustained over 20 years, the magnitude of the treatment effect observed in this study would be consistent with a predefined cost-effectiveness target of about $58,400 per quality-adjusted life-year, she said.
Patients included in this study were adults with a mean age of 38 years and a mean age at disease onset of 31 years. Most (76%) were women. Disease duration was a mean of 7.7 years at baseline, and the patients had a mean of three relapses over the prior 2 years. Baseline EDSS score was 3.06.
“In 2002, the National Institute of Clinical Excellence [NICE], which is the U.K. body that sets the guidelines that decide what should be available under the National Health Service, concluded that beta-interferon and glatiramer acetate were not cost effective during the short-term, and thus it recommended that it couldn’t be available under the NHS at that stage,” Dr. Palace explained.
The NICE did notice, however, that if the drug effect was maintained over the long term it might be cost effective and invited the Department of Health to “try and find a way to make the drugs available in a cost-effective manner,” she said.
“This was the birth of the Risk-Sharing Scheme, and it is a way of providing disease-modifying therapies to patients cost effectively,” she said.
At the start of the scheme, the drug prices were reduced in line with a cost-effective requirement using the NICE model, which equated to about $58,400/quality-adjusted life-year..
“We were allowed to reach this target over a 20-year lifespan, and the idea was to monitor a clinical cohort of patients, measuring their EDSS scores,” she said.
The natural history dataset was used as a “virtual placebo,” and a price adjustment would be required if the outcomes of the treated patients were less than predicted to be on target for cost effectiveness.
The findings of this study – the largest observational study to measure the effect and cost-effectiveness of interferon beta-1a and glatiramer acetate – provide evidence that the treatment alters the natural history of relapsing-remitting MS in the real life setting, she concluded.
Dr. Palace reported receiving support for scientific meetings and honorariums for advisory work from Teva, which makes glatiramer acetate, and Bayer Schering, which makes interferon beta-1a, as well as other companies that market drugs for MS. She’s also received unrestricted grants from Bayer Schering and other companies that market drugs for MS.
BOSTON - Treatment with interferon beta-1a or glatiramer acetate, if used long term, has the potential to be cost effective and to alter the natural history of relapsing-remitting multiple sclerosis, according to findings from a 6-year observational study.
In 4,137 patients with relapsing-remitting MS who were followed for a median of 6 years, the observed increase in the Expanded Disability Status Scale (EDSS) scores was lower than the increase seen in a natural history cohort that served as a comparator group (relative rates, 0.76 using a continuous Markov model and 0.61 using a multilevel model). The observed progression for utility – a quality of life measure derived from the EDSS scores, and the primary outcome of the study – was consistent with this finding (relative rates of 0.58 and 0.56 according to the two models, respectively), Dr. Jacqueline Palace reported at the joint meeting of the European and Americas Committees for Treatment and Research in Multiple Sclerosis.
The outcome – essentially a reduction of about 42% on the primary outcome measure, compared with the natural history cohort – was better than the 38% reduction that was predicted, said Dr. Palace, leader of the multiple sclerosis and neuromyelitis group at Oxford University Hospitals Trust in England.
A range of sensitivity analyses that examined the potential for various biases consistently demonstrated better outcomes in the treated patients, compared with the natural history cohort, Dr. Palace noted.
If sustained over 20 years, the magnitude of the treatment effect observed in this study would be consistent with a predefined cost-effectiveness target of about $58,400 per quality-adjusted life-year, she said.
Patients included in this study were adults with a mean age of 38 years and a mean age at disease onset of 31 years. Most (76%) were women. Disease duration was a mean of 7.7 years at baseline, and the patients had a mean of three relapses over the prior 2 years. Baseline EDSS score was 3.06.
“In 2002, the National Institute of Clinical Excellence [NICE], which is the U.K. body that sets the guidelines that decide what should be available under the National Health Service, concluded that beta-interferon and glatiramer acetate were not cost effective during the short-term, and thus it recommended that it couldn’t be available under the NHS at that stage,” Dr. Palace explained.
The NICE did notice, however, that if the drug effect was maintained over the long term it might be cost effective and invited the Department of Health to “try and find a way to make the drugs available in a cost-effective manner,” she said.
“This was the birth of the Risk-Sharing Scheme, and it is a way of providing disease-modifying therapies to patients cost effectively,” she said.
At the start of the scheme, the drug prices were reduced in line with a cost-effective requirement using the NICE model, which equated to about $58,400/quality-adjusted life-year..
“We were allowed to reach this target over a 20-year lifespan, and the idea was to monitor a clinical cohort of patients, measuring their EDSS scores,” she said.
The natural history dataset was used as a “virtual placebo,” and a price adjustment would be required if the outcomes of the treated patients were less than predicted to be on target for cost effectiveness.
The findings of this study – the largest observational study to measure the effect and cost-effectiveness of interferon beta-1a and glatiramer acetate – provide evidence that the treatment alters the natural history of relapsing-remitting MS in the real life setting, she concluded.
Dr. Palace reported receiving support for scientific meetings and honorariums for advisory work from Teva, which makes glatiramer acetate, and Bayer Schering, which makes interferon beta-1a, as well as other companies that market drugs for MS. She’s also received unrestricted grants from Bayer Schering and other companies that market drugs for MS.
AT MSBOSTON 2014
Key clinical point: Treatment with either glatiramer acetate or interferon beta-1a alters the natural history of relapsing-remitting MS in the real-life setting and appears to be cost effective in the long term.
Major finding: The observed treatment effect, if sustained long-term, would meet the predefined cost-effectiveness target of about $58,400/quality-adjusted life-year.
Data source: An observational study of 4,137 MS patients.
Disclosures: Dr. Palace reported receiving support for scientific meetings and honorariums for advisory work from Teva, which makes glatiramer acetate, and Bayer Schering, which makes interferon beta-1a, as well as other companies that market drugs for MS. She’s also received unrestricted grants from Bayer Schering and other companies that market drugs for MS.