User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Low IgG1/high IgG4 ratios seen in pregnancy may alter flu vaccine response
PHILADELPHIA – Low IgG1/high IgG4 ratios appear to be more common in pregnant women and may be associated with a diminished response to influenza vaccination, according to Dr. Elizabeth P. Schlaudecker.
“Basically, we know that at the maternal-fetal interface, there are lots of cytokine changes going on and lots of immunologic changes going on, but does this immune milieu of pregnancy actually influence systemic response? Obviously we think it does, but does it really affect the pregnant woman’s response to immunization? In turn, does it really influence the magnitude and character of the antibody response to flu vaccine?” Dr. Schlaudecker, of Cincinnati Children’s Hospital Medical Center, said at an annual scientific meeting on infectious diseases.
These questions prompted her research, and the answers are important because pregnant women don’t do well during flu seasons, she said, noting that “this was especially brought to light during the H1N1 pandemic,” when pregnant women had higher rates of hospital admission, more medical encounters with confirmed or suspected influenza, and greater severity of disease during late pregnancy than nonpregnant women and others.
These effects, which also occur during regular flu seasons, are more pronounced during the second and third trimesters, she said.
Because of this, influenza vaccine is recommended universally in pregnancy with the goal of preventing infection in both mothers and infants.
To determine whether the effects of pregnancy that worsen the outcome of influenza infection also suppress the response to flu vaccine, Dr. Schlaudecker and her colleagues reviewed reports about the immunogenicity of influenza vaccine in pregnancy.
Most studies and reports show that vaccinated pregnant women are likely to have seroprotective responses, but few have compared pregnant and nonpregnant women, so she recruited 70 pregnant women and 65 nonpregnant women, aged 18-39 years, and compared sera before and 28 days after influenza immunization during the 2011-2012 and 2012-2013 flu seasons.
Hemagglutination inhibition (HAI), as expected, was reduced during pregnancy. The pregnant women, who were in either their second or third trimester, had significantly lower HAI titers for anti-influenza H3N2 (154.55 vs. 242.51), and the differences approached significance for both H1N1 (129.96 vs. 181.84) and B antigens (24.91 v. 35.20). She reported these findings at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
“So I took this information and realized that women may have a decreased antibody response when they are pregnant, and hypothesized that during this time of pregnancy, there are cytokines associated with this maternal-fetal interface that suppress the IgG1 and IgG3 response and promote an IgG4 response to influenza vaccination, particularly during the second and third trimesters, when production of these cytokines should be highest,” she said.
This matters, because the four subclasses of IgG have functional differences. The effector functions of the IgG subclasses are usually opsonization and complement activation, and activation of inflammatory cells through Fc-gamma receptors. IgG1 and IgG3 are more effective than IgG2 or IgG4 in binding stimulatory Fc-gamma receptors and activating complement, she explained.
“IgG4 is also functionally monovalent, so it doesn’t aggregate antigens very well, and it makes it less protective against viruses,” she said, explaining that in normal, nonpregnant women, IgG1 and IgG3 are the predominant responders to viral infection, and they are most likely to be involved in flu virus protection.
“So the question is, although we know [down-regulation of IgG1 and IgG3, and up-regulation of IgG4,] is taking place around the placenta, are these cytokine effects actually circulating systemically enough to affect the flu vaccine?” she asked.
In the study participants, there was a general trend for most pregnant women to have responses that had high IgG4s and low IgG1s, which is not protective, and for nonpregnant women to have high IgG1s and low IgG4s.
The difference between the groups in this regard was not statistically significant, but there were significantly more pregnant women than nonpregnant women with high IgG4 and low IgG1 (10% vs. 0%), and there were significantly fewer pregnant women than nonpregnant women with low IgG4 and high IgG1 (3% vs. 15%), she said.
Both anti-H1N1 HAI and IgG1 titers were significantly lower in pregnant vs. nonpregnant women, but for anti-H1N1 IgG4 titers, the levels were much higher in pregnant vs. nonpregnant women, she said.
“These correlated with each other, suggesting that when you have a high HAI titer, you have a high IgG1 response, which goes along with IgG1 being the predominant IgG isotype and the one most associated with protecting against viral disease,” she said.
“A subset of these women had this very high IgG4 and low IgG1 response, and this suggests a Th2/Treg influence. This unique isotype profile was not found in any nonpregnant women,” she said, adding that very few pregnant women in their second or third trimester make the high IgG1/low IgG4 that should provide a good response to flu vaccine.
Although Dr. Schlaudecker acknowledged that 80%-90% of the pregnant women in the study had protective HAI titers after immunization, she said the findings have important implications.
“I’m concerned that these low IgG1/high IgG4 ratios seen in pregnant women might actually be giving poor protection against flu infection, which brings us back to the pediatric patients. If pregnant women are not protected well, we are not protecting the babies as well. This suggests that we might need to reconsider approaches to timing of flu vaccine or actually the particular vaccines that we give to pregnant women, and it also shows that pregnancy likely effects systemic responses to things like flu vaccine and other vaccines,” she said.
Dr. Schlaudecker reported having no disclosures.
PHILADELPHIA – Low IgG1/high IgG4 ratios appear to be more common in pregnant women and may be associated with a diminished response to influenza vaccination, according to Dr. Elizabeth P. Schlaudecker.
“Basically, we know that at the maternal-fetal interface, there are lots of cytokine changes going on and lots of immunologic changes going on, but does this immune milieu of pregnancy actually influence systemic response? Obviously we think it does, but does it really affect the pregnant woman’s response to immunization? In turn, does it really influence the magnitude and character of the antibody response to flu vaccine?” Dr. Schlaudecker, of Cincinnati Children’s Hospital Medical Center, said at an annual scientific meeting on infectious diseases.
These questions prompted her research, and the answers are important because pregnant women don’t do well during flu seasons, she said, noting that “this was especially brought to light during the H1N1 pandemic,” when pregnant women had higher rates of hospital admission, more medical encounters with confirmed or suspected influenza, and greater severity of disease during late pregnancy than nonpregnant women and others.
These effects, which also occur during regular flu seasons, are more pronounced during the second and third trimesters, she said.
Because of this, influenza vaccine is recommended universally in pregnancy with the goal of preventing infection in both mothers and infants.
To determine whether the effects of pregnancy that worsen the outcome of influenza infection also suppress the response to flu vaccine, Dr. Schlaudecker and her colleagues reviewed reports about the immunogenicity of influenza vaccine in pregnancy.
Most studies and reports show that vaccinated pregnant women are likely to have seroprotective responses, but few have compared pregnant and nonpregnant women, so she recruited 70 pregnant women and 65 nonpregnant women, aged 18-39 years, and compared sera before and 28 days after influenza immunization during the 2011-2012 and 2012-2013 flu seasons.
Hemagglutination inhibition (HAI), as expected, was reduced during pregnancy. The pregnant women, who were in either their second or third trimester, had significantly lower HAI titers for anti-influenza H3N2 (154.55 vs. 242.51), and the differences approached significance for both H1N1 (129.96 vs. 181.84) and B antigens (24.91 v. 35.20). She reported these findings at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
“So I took this information and realized that women may have a decreased antibody response when they are pregnant, and hypothesized that during this time of pregnancy, there are cytokines associated with this maternal-fetal interface that suppress the IgG1 and IgG3 response and promote an IgG4 response to influenza vaccination, particularly during the second and third trimesters, when production of these cytokines should be highest,” she said.
This matters, because the four subclasses of IgG have functional differences. The effector functions of the IgG subclasses are usually opsonization and complement activation, and activation of inflammatory cells through Fc-gamma receptors. IgG1 and IgG3 are more effective than IgG2 or IgG4 in binding stimulatory Fc-gamma receptors and activating complement, she explained.
“IgG4 is also functionally monovalent, so it doesn’t aggregate antigens very well, and it makes it less protective against viruses,” she said, explaining that in normal, nonpregnant women, IgG1 and IgG3 are the predominant responders to viral infection, and they are most likely to be involved in flu virus protection.
“So the question is, although we know [down-regulation of IgG1 and IgG3, and up-regulation of IgG4,] is taking place around the placenta, are these cytokine effects actually circulating systemically enough to affect the flu vaccine?” she asked.
In the study participants, there was a general trend for most pregnant women to have responses that had high IgG4s and low IgG1s, which is not protective, and for nonpregnant women to have high IgG1s and low IgG4s.
The difference between the groups in this regard was not statistically significant, but there were significantly more pregnant women than nonpregnant women with high IgG4 and low IgG1 (10% vs. 0%), and there were significantly fewer pregnant women than nonpregnant women with low IgG4 and high IgG1 (3% vs. 15%), she said.
Both anti-H1N1 HAI and IgG1 titers were significantly lower in pregnant vs. nonpregnant women, but for anti-H1N1 IgG4 titers, the levels were much higher in pregnant vs. nonpregnant women, she said.
“These correlated with each other, suggesting that when you have a high HAI titer, you have a high IgG1 response, which goes along with IgG1 being the predominant IgG isotype and the one most associated with protecting against viral disease,” she said.
“A subset of these women had this very high IgG4 and low IgG1 response, and this suggests a Th2/Treg influence. This unique isotype profile was not found in any nonpregnant women,” she said, adding that very few pregnant women in their second or third trimester make the high IgG1/low IgG4 that should provide a good response to flu vaccine.
Although Dr. Schlaudecker acknowledged that 80%-90% of the pregnant women in the study had protective HAI titers after immunization, she said the findings have important implications.
“I’m concerned that these low IgG1/high IgG4 ratios seen in pregnant women might actually be giving poor protection against flu infection, which brings us back to the pediatric patients. If pregnant women are not protected well, we are not protecting the babies as well. This suggests that we might need to reconsider approaches to timing of flu vaccine or actually the particular vaccines that we give to pregnant women, and it also shows that pregnancy likely effects systemic responses to things like flu vaccine and other vaccines,” she said.
Dr. Schlaudecker reported having no disclosures.
PHILADELPHIA – Low IgG1/high IgG4 ratios appear to be more common in pregnant women and may be associated with a diminished response to influenza vaccination, according to Dr. Elizabeth P. Schlaudecker.
“Basically, we know that at the maternal-fetal interface, there are lots of cytokine changes going on and lots of immunologic changes going on, but does this immune milieu of pregnancy actually influence systemic response? Obviously we think it does, but does it really affect the pregnant woman’s response to immunization? In turn, does it really influence the magnitude and character of the antibody response to flu vaccine?” Dr. Schlaudecker, of Cincinnati Children’s Hospital Medical Center, said at an annual scientific meeting on infectious diseases.
These questions prompted her research, and the answers are important because pregnant women don’t do well during flu seasons, she said, noting that “this was especially brought to light during the H1N1 pandemic,” when pregnant women had higher rates of hospital admission, more medical encounters with confirmed or suspected influenza, and greater severity of disease during late pregnancy than nonpregnant women and others.
These effects, which also occur during regular flu seasons, are more pronounced during the second and third trimesters, she said.
Because of this, influenza vaccine is recommended universally in pregnancy with the goal of preventing infection in both mothers and infants.
To determine whether the effects of pregnancy that worsen the outcome of influenza infection also suppress the response to flu vaccine, Dr. Schlaudecker and her colleagues reviewed reports about the immunogenicity of influenza vaccine in pregnancy.
Most studies and reports show that vaccinated pregnant women are likely to have seroprotective responses, but few have compared pregnant and nonpregnant women, so she recruited 70 pregnant women and 65 nonpregnant women, aged 18-39 years, and compared sera before and 28 days after influenza immunization during the 2011-2012 and 2012-2013 flu seasons.
Hemagglutination inhibition (HAI), as expected, was reduced during pregnancy. The pregnant women, who were in either their second or third trimester, had significantly lower HAI titers for anti-influenza H3N2 (154.55 vs. 242.51), and the differences approached significance for both H1N1 (129.96 vs. 181.84) and B antigens (24.91 v. 35.20). She reported these findings at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
“So I took this information and realized that women may have a decreased antibody response when they are pregnant, and hypothesized that during this time of pregnancy, there are cytokines associated with this maternal-fetal interface that suppress the IgG1 and IgG3 response and promote an IgG4 response to influenza vaccination, particularly during the second and third trimesters, when production of these cytokines should be highest,” she said.
This matters, because the four subclasses of IgG have functional differences. The effector functions of the IgG subclasses are usually opsonization and complement activation, and activation of inflammatory cells through Fc-gamma receptors. IgG1 and IgG3 are more effective than IgG2 or IgG4 in binding stimulatory Fc-gamma receptors and activating complement, she explained.
“IgG4 is also functionally monovalent, so it doesn’t aggregate antigens very well, and it makes it less protective against viruses,” she said, explaining that in normal, nonpregnant women, IgG1 and IgG3 are the predominant responders to viral infection, and they are most likely to be involved in flu virus protection.
“So the question is, although we know [down-regulation of IgG1 and IgG3, and up-regulation of IgG4,] is taking place around the placenta, are these cytokine effects actually circulating systemically enough to affect the flu vaccine?” she asked.
In the study participants, there was a general trend for most pregnant women to have responses that had high IgG4s and low IgG1s, which is not protective, and for nonpregnant women to have high IgG1s and low IgG4s.
The difference between the groups in this regard was not statistically significant, but there were significantly more pregnant women than nonpregnant women with high IgG4 and low IgG1 (10% vs. 0%), and there were significantly fewer pregnant women than nonpregnant women with low IgG4 and high IgG1 (3% vs. 15%), she said.
Both anti-H1N1 HAI and IgG1 titers were significantly lower in pregnant vs. nonpregnant women, but for anti-H1N1 IgG4 titers, the levels were much higher in pregnant vs. nonpregnant women, she said.
“These correlated with each other, suggesting that when you have a high HAI titer, you have a high IgG1 response, which goes along with IgG1 being the predominant IgG isotype and the one most associated with protecting against viral disease,” she said.
“A subset of these women had this very high IgG4 and low IgG1 response, and this suggests a Th2/Treg influence. This unique isotype profile was not found in any nonpregnant women,” she said, adding that very few pregnant women in their second or third trimester make the high IgG1/low IgG4 that should provide a good response to flu vaccine.
Although Dr. Schlaudecker acknowledged that 80%-90% of the pregnant women in the study had protective HAI titers after immunization, she said the findings have important implications.
“I’m concerned that these low IgG1/high IgG4 ratios seen in pregnant women might actually be giving poor protection against flu infection, which brings us back to the pediatric patients. If pregnant women are not protected well, we are not protecting the babies as well. This suggests that we might need to reconsider approaches to timing of flu vaccine or actually the particular vaccines that we give to pregnant women, and it also shows that pregnancy likely effects systemic responses to things like flu vaccine and other vaccines,” she said.
Dr. Schlaudecker reported having no disclosures.
Key clinical point: Changes during pregnancy may diminish the effects of influenza vaccine, requiring a new approach to vaccination in this population.
Major finding: Significantly more pregnant women than nonpregnant women had high IgG4 and low IgG1 (10% vs. 0%), and significantly fewer pregnant women than nonpregnant women had low IgG4 and high IgG1 (3% vs. 15%).
Data source: An observational study of 70 pregnant and 65 nonpregnant women.
Disclosures: Dr. Schlaudecker reported having no disclosures.
Comborbidities likely explain opioid + sleep apnea mortality risk
AUSTIN, TEX. – Any association between opioid use and death in patients with sleep apnea cannot be explained by sleep apnea alone, according to a retrospective analysis of data from the prospective observational DREAM study.
In 1,867 patients with moderate or severe sleep apnea who were on an opioid medication, no association was seen between opioid use and severity of sleep-disordered breathing, even with increasing doses, Dr. Husham Sharifi reported at the annual meeting of the American College of Chest Physicians.
Further, when opioid use was analyzed as an unadjusted variable, it was associated with an increase in mortality (odds ratio, 1.53), but this effect was attenuated by adjustment for sleep apnea (OR, 1.52), and was further attenuated – to the point where it was no longer statistically significant – by adjustment for both sleep apnea and Charlson Comorbidity Index (OR, 1.37), said Dr. Sharifi, who was an attending physician at Brigham and Women’s Hospital, Boston, at the time he completed this research. He is now a fellow at Stanford (Calif.) University.
Sleep apnea remained an independent predictor of mortality even after adjustment for opioid use and Charlson Comorbidity Index, he said.
The DREAM study, which stands for Determining Risk of Vascular Events by Apnea Monitoring, includes a well-defined cohort of patients at three Veterans Administration sleep centers who were referred for overnight polysomnography for suspected sleep-disordered breathing between Jan. 1, 2000, and Dec. 31, 2004. All patients had an Apnea-Hypopnea Index score greater than 15, indicating moderate to severe sleep apnea, and all were on opioid medication. The patients were followed for between 3 and 10 years, with follow-up ending Dec. 31, 2010.
Opioid use has increased dramatically over the past 20 years – by more than 700%, Dr. Sharifi said.
While there does not appear to be a significant impact of opioid use on daytime respiration, there are some limited data suggesting that it may be associated with sleep-disordered breathing, he said.
The current findings, though limited by a number of factors including the observational nature of the study and possible selection bias as the DREAM cohort is a referral population, suggest that the relationship between opioid use and sleep apnea death is likely explained not by sleep apnea, but by an increased prevalence of known risk factors for morbidity and mortality in patients who take opioids and have sleep apnea, he concluded.
Dr. Sharifi reported having no disclosures.
AUSTIN, TEX. – Any association between opioid use and death in patients with sleep apnea cannot be explained by sleep apnea alone, according to a retrospective analysis of data from the prospective observational DREAM study.
In 1,867 patients with moderate or severe sleep apnea who were on an opioid medication, no association was seen between opioid use and severity of sleep-disordered breathing, even with increasing doses, Dr. Husham Sharifi reported at the annual meeting of the American College of Chest Physicians.
Further, when opioid use was analyzed as an unadjusted variable, it was associated with an increase in mortality (odds ratio, 1.53), but this effect was attenuated by adjustment for sleep apnea (OR, 1.52), and was further attenuated – to the point where it was no longer statistically significant – by adjustment for both sleep apnea and Charlson Comorbidity Index (OR, 1.37), said Dr. Sharifi, who was an attending physician at Brigham and Women’s Hospital, Boston, at the time he completed this research. He is now a fellow at Stanford (Calif.) University.
Sleep apnea remained an independent predictor of mortality even after adjustment for opioid use and Charlson Comorbidity Index, he said.
The DREAM study, which stands for Determining Risk of Vascular Events by Apnea Monitoring, includes a well-defined cohort of patients at three Veterans Administration sleep centers who were referred for overnight polysomnography for suspected sleep-disordered breathing between Jan. 1, 2000, and Dec. 31, 2004. All patients had an Apnea-Hypopnea Index score greater than 15, indicating moderate to severe sleep apnea, and all were on opioid medication. The patients were followed for between 3 and 10 years, with follow-up ending Dec. 31, 2010.
Opioid use has increased dramatically over the past 20 years – by more than 700%, Dr. Sharifi said.
While there does not appear to be a significant impact of opioid use on daytime respiration, there are some limited data suggesting that it may be associated with sleep-disordered breathing, he said.
The current findings, though limited by a number of factors including the observational nature of the study and possible selection bias as the DREAM cohort is a referral population, suggest that the relationship between opioid use and sleep apnea death is likely explained not by sleep apnea, but by an increased prevalence of known risk factors for morbidity and mortality in patients who take opioids and have sleep apnea, he concluded.
Dr. Sharifi reported having no disclosures.
AUSTIN, TEX. – Any association between opioid use and death in patients with sleep apnea cannot be explained by sleep apnea alone, according to a retrospective analysis of data from the prospective observational DREAM study.
In 1,867 patients with moderate or severe sleep apnea who were on an opioid medication, no association was seen between opioid use and severity of sleep-disordered breathing, even with increasing doses, Dr. Husham Sharifi reported at the annual meeting of the American College of Chest Physicians.
Further, when opioid use was analyzed as an unadjusted variable, it was associated with an increase in mortality (odds ratio, 1.53), but this effect was attenuated by adjustment for sleep apnea (OR, 1.52), and was further attenuated – to the point where it was no longer statistically significant – by adjustment for both sleep apnea and Charlson Comorbidity Index (OR, 1.37), said Dr. Sharifi, who was an attending physician at Brigham and Women’s Hospital, Boston, at the time he completed this research. He is now a fellow at Stanford (Calif.) University.
Sleep apnea remained an independent predictor of mortality even after adjustment for opioid use and Charlson Comorbidity Index, he said.
The DREAM study, which stands for Determining Risk of Vascular Events by Apnea Monitoring, includes a well-defined cohort of patients at three Veterans Administration sleep centers who were referred for overnight polysomnography for suspected sleep-disordered breathing between Jan. 1, 2000, and Dec. 31, 2004. All patients had an Apnea-Hypopnea Index score greater than 15, indicating moderate to severe sleep apnea, and all were on opioid medication. The patients were followed for between 3 and 10 years, with follow-up ending Dec. 31, 2010.
Opioid use has increased dramatically over the past 20 years – by more than 700%, Dr. Sharifi said.
While there does not appear to be a significant impact of opioid use on daytime respiration, there are some limited data suggesting that it may be associated with sleep-disordered breathing, he said.
The current findings, though limited by a number of factors including the observational nature of the study and possible selection bias as the DREAM cohort is a referral population, suggest that the relationship between opioid use and sleep apnea death is likely explained not by sleep apnea, but by an increased prevalence of known risk factors for morbidity and mortality in patients who take opioids and have sleep apnea, he concluded.
Dr. Sharifi reported having no disclosures.
Key clinical point: Comorbid conditions may explain the relationship between opioid use, sleep apnea, and death.
Major finding: Opioid use was not significantly associated with mortality after adjustment for sleep apnea and comorbidities (odds ratio, 1.37).
Data source: A retrospective analysis of data for 1,867 patients from the prospective observational DREAM cohort.
Disclosures: Dr. Sharifi reported having no disclosures.
Pneumonia-related readmissions plummeted under QI initiative
AUSTIN, TEX. – A multidisciplinary intervention, including implementation of a diagnostic scoring system and daily interdepartmental meetings to review cases, significantly decreased readmission rates for patients at a tertiary care center who were discharged with a diagnosis of pneumonia.
From November 2012 to January 2013 – a 3-month period after implementation of the quality improvement (QI) initiative – the all-cause readmission rates among 227 patients discharged with a diagnosis of pneumonia declined by 7.5 percentage points, compared with the all-cause readmission rates among 236 patients discharged during the same period in the prior year, before implementation of the initiative (from 20.7% to 13.2%), Dr. Hussein Hussein, a fellow at the University of Oklahoma Health Sciences Center, Oklahoma City, reported at the annual meeting of the American College of Chest Physicians.
A similar reduction was seen for pneumonia-related readmissions, which declined from 10.5% to 3% during the same period, said Dr. Hussein, who was with Yale New Haven Hospital, New Haven, Conn., at the time the research was conducted.
Further, after implementation of the scoring system – a modified Clinical Pulmonary Infection Score (MCPIS) calculated based on patient temperature, white blood cell count, sputum cultures, oxygen requirements, and radiographic appearance, which was administered at admission and again at 32 hours – the accuracy of pneumonia diagnoses appeared to improve; the mean MCPIS scores among patients with a discharge diagnosis of pneumonia increased significantly after implementation (from 4 to 6); the proportion of patients considered unlikely to have pneumonia decreased from 42.6% to 3.6%; the proportion considered to probably have pneumonia decreased from 31.9% to 17.9%; and the number deemed likely to have pneumonia increased from 25.5% to 78.6%.
All of the changes were statistically significant, and the improved accuracy of diagnosis was likely a result of provider education that led to increased use of sputum cultures, Dr. Hussein noted.
Pneumonia is the second most common discharge diagnosis among Medicare beneficiaries, and nearly 20% of these patients are readmitted within 30 days at a cost exceeding $17 billion annually.
“This is why the Centers for Medicare & Medicaid Services is penalizing poor-performing hospitals with high rates of readmission,” he said.
The MCPIS was implemented in 2012 as a quality improvement tool. Based on the scores, patients were categorized as unlikely to have pneumonia (score of 3 or less), probably having pneumonia (score of 4-5), and likely to have pneumonia (score of 6 or greater). The daily meetings during which patients admitted with pneumonia were reviewed, involved participation of physicians from different medical divisions and representatives from nursing, social work, and continuing care.
The primary goal of these rounds was to ensure timely follow-up after discharge, Dr. Hussein explained, adding that if the diagnosis was felt to be incorrect, the case was discussed with the team that was caring for the patient.
To assess the effects of this intervention, he and his colleagues conducted a retrospective chart review of all patients discharged with a diagnosis of pneumonia during each of the two assessment periods.
“Our multidisciplinary intervention resulted in a significant decrease in readmission rates in patients discharged with a diagnosis of pneumonia, as well as improvement in the accuracy of diagnosis,” he said.
Dr. Hussein reported having no disclosures.
AUSTIN, TEX. – A multidisciplinary intervention, including implementation of a diagnostic scoring system and daily interdepartmental meetings to review cases, significantly decreased readmission rates for patients at a tertiary care center who were discharged with a diagnosis of pneumonia.
From November 2012 to January 2013 – a 3-month period after implementation of the quality improvement (QI) initiative – the all-cause readmission rates among 227 patients discharged with a diagnosis of pneumonia declined by 7.5 percentage points, compared with the all-cause readmission rates among 236 patients discharged during the same period in the prior year, before implementation of the initiative (from 20.7% to 13.2%), Dr. Hussein Hussein, a fellow at the University of Oklahoma Health Sciences Center, Oklahoma City, reported at the annual meeting of the American College of Chest Physicians.
A similar reduction was seen for pneumonia-related readmissions, which declined from 10.5% to 3% during the same period, said Dr. Hussein, who was with Yale New Haven Hospital, New Haven, Conn., at the time the research was conducted.
Further, after implementation of the scoring system – a modified Clinical Pulmonary Infection Score (MCPIS) calculated based on patient temperature, white blood cell count, sputum cultures, oxygen requirements, and radiographic appearance, which was administered at admission and again at 32 hours – the accuracy of pneumonia diagnoses appeared to improve; the mean MCPIS scores among patients with a discharge diagnosis of pneumonia increased significantly after implementation (from 4 to 6); the proportion of patients considered unlikely to have pneumonia decreased from 42.6% to 3.6%; the proportion considered to probably have pneumonia decreased from 31.9% to 17.9%; and the number deemed likely to have pneumonia increased from 25.5% to 78.6%.
All of the changes were statistically significant, and the improved accuracy of diagnosis was likely a result of provider education that led to increased use of sputum cultures, Dr. Hussein noted.
Pneumonia is the second most common discharge diagnosis among Medicare beneficiaries, and nearly 20% of these patients are readmitted within 30 days at a cost exceeding $17 billion annually.
“This is why the Centers for Medicare & Medicaid Services is penalizing poor-performing hospitals with high rates of readmission,” he said.
The MCPIS was implemented in 2012 as a quality improvement tool. Based on the scores, patients were categorized as unlikely to have pneumonia (score of 3 or less), probably having pneumonia (score of 4-5), and likely to have pneumonia (score of 6 or greater). The daily meetings during which patients admitted with pneumonia were reviewed, involved participation of physicians from different medical divisions and representatives from nursing, social work, and continuing care.
The primary goal of these rounds was to ensure timely follow-up after discharge, Dr. Hussein explained, adding that if the diagnosis was felt to be incorrect, the case was discussed with the team that was caring for the patient.
To assess the effects of this intervention, he and his colleagues conducted a retrospective chart review of all patients discharged with a diagnosis of pneumonia during each of the two assessment periods.
“Our multidisciplinary intervention resulted in a significant decrease in readmission rates in patients discharged with a diagnosis of pneumonia, as well as improvement in the accuracy of diagnosis,” he said.
Dr. Hussein reported having no disclosures.
AUSTIN, TEX. – A multidisciplinary intervention, including implementation of a diagnostic scoring system and daily interdepartmental meetings to review cases, significantly decreased readmission rates for patients at a tertiary care center who were discharged with a diagnosis of pneumonia.
From November 2012 to January 2013 – a 3-month period after implementation of the quality improvement (QI) initiative – the all-cause readmission rates among 227 patients discharged with a diagnosis of pneumonia declined by 7.5 percentage points, compared with the all-cause readmission rates among 236 patients discharged during the same period in the prior year, before implementation of the initiative (from 20.7% to 13.2%), Dr. Hussein Hussein, a fellow at the University of Oklahoma Health Sciences Center, Oklahoma City, reported at the annual meeting of the American College of Chest Physicians.
A similar reduction was seen for pneumonia-related readmissions, which declined from 10.5% to 3% during the same period, said Dr. Hussein, who was with Yale New Haven Hospital, New Haven, Conn., at the time the research was conducted.
Further, after implementation of the scoring system – a modified Clinical Pulmonary Infection Score (MCPIS) calculated based on patient temperature, white blood cell count, sputum cultures, oxygen requirements, and radiographic appearance, which was administered at admission and again at 32 hours – the accuracy of pneumonia diagnoses appeared to improve; the mean MCPIS scores among patients with a discharge diagnosis of pneumonia increased significantly after implementation (from 4 to 6); the proportion of patients considered unlikely to have pneumonia decreased from 42.6% to 3.6%; the proportion considered to probably have pneumonia decreased from 31.9% to 17.9%; and the number deemed likely to have pneumonia increased from 25.5% to 78.6%.
All of the changes were statistically significant, and the improved accuracy of diagnosis was likely a result of provider education that led to increased use of sputum cultures, Dr. Hussein noted.
Pneumonia is the second most common discharge diagnosis among Medicare beneficiaries, and nearly 20% of these patients are readmitted within 30 days at a cost exceeding $17 billion annually.
“This is why the Centers for Medicare & Medicaid Services is penalizing poor-performing hospitals with high rates of readmission,” he said.
The MCPIS was implemented in 2012 as a quality improvement tool. Based on the scores, patients were categorized as unlikely to have pneumonia (score of 3 or less), probably having pneumonia (score of 4-5), and likely to have pneumonia (score of 6 or greater). The daily meetings during which patients admitted with pneumonia were reviewed, involved participation of physicians from different medical divisions and representatives from nursing, social work, and continuing care.
The primary goal of these rounds was to ensure timely follow-up after discharge, Dr. Hussein explained, adding that if the diagnosis was felt to be incorrect, the case was discussed with the team that was caring for the patient.
To assess the effects of this intervention, he and his colleagues conducted a retrospective chart review of all patients discharged with a diagnosis of pneumonia during each of the two assessment periods.
“Our multidisciplinary intervention resulted in a significant decrease in readmission rates in patients discharged with a diagnosis of pneumonia, as well as improvement in the accuracy of diagnosis,” he said.
Dr. Hussein reported having no disclosures.
Key clinical point: A scoring system and multidisciplinary effort improved pneumonia patient readmission rates and diagnostic accuracy.
Major finding: All-cause and pneumonia-related readmission rates declined from 20.7% to 13.2%, and from 10.5% to 3%, respectively.
Data source: A retrospective review of the charts of 463 patients.
Disclosures: Dr. Hussein reported having no disclosures.
LAMA/LABA combo tops fluticasone/salmeterol in moderate/severe COPD
AUSTIN, TEX.– Once daily combination treatment with umeclidinium and vilanterol was more effective than twice-daily combination treatment with fluticasone and salmeterol in patients with moderate to severe chronic obstructive pulmonary disease in two 12-week double-blind, parallel-group double-dummy studies.
In the two multicenter studies, 706 and 697 patients, respectively, were randomized to receive either 62.5 mcg of the long-acting muscarinic antagonist (LAMA) umeclidinium and 25 mcg of the long-acting beta-2 agonist (LABA) vilanterol – a recently approved combination bronchodilator maintenance treatment for COPD – or a combination of 250 mcg of the inhaled corticosteroid (ICS) fluticasone and 50 mcg of the LABA salmeterol, which is also indicated as a maintenance therapy for COPD.
The patients in the LAMA/LABA groups, who were treated once daily for 12 weeks, had significantly greater improvements on all lung function measures, compared with those in the ICS/LABA groups, who were treated twice daily, Dr. James F. Donohue of the University of North Carolina at Chapel Hill reported at the annual meeting of the American College of Chest Physicians.
In the first study, the improvement from baseline to day 84 (the primary study endpoint) in 0- to 24-hour weighted mean forced expiratory volume in 1 second (FEV1) was 165 mL for the LAMA/LABA group, compared with 91 mL in the ICS/LABA group. In the second study, the improvement in the two groups was 213 mL and 112 mL.
The LAMA/LABA combination also improved trough FEV1 on day 85 by 82 mL and 98 mL more than did the ICS/LABA combination in the two studies, respectively.
Both combinations provided clinically meaningful improvements in dyspnea and quality of life scores, Dr. Donohue said.
Adverse events occurred during treatment in a similar proportion of patients in both treatment groups in both studies: 26% and 27% in the LAMA/LABA and ICS/LABA patients in the first study, and 30% and 31%, respectively, in the second study. The most common adverse events were headache and nasopharyngitis.
In the first study, serious adverse events occurred in 2% of the LAMA/LABA patients and 3% of ICS/LABA patients, and in 3% and 4% of patients in the second study.
One death occurred in the ICS/LABA group in the first study, but it was not considered study related. Five deaths occurred in the second study, including two in the LAMA/LABA patients and three in the ICS/LABA patients. One of the deaths in the ICS/LABA group was because of pneumonia and was reported as drug related by the investigator.
No new safety signals were detected in these studies, Dr. Donohue said.
Patients in both studies had FEV1 between 30% and 70%, and had not experienced a COPD exacerbation within the previous year. The LAMA/LABA therapy was delivered via Ellipta inhaler, and the ICS/LABA therapy was delivered via Diskus inhaler.
The Food and Drug Administration approved umeclidinium/vilanterol combination therapy (Anoro Ellipta) in December 2013, the first LAMA/LABA therapy approved in the United States. Dr. Donohue and his colleagues conducted the pivotal regulatory trial of the drug combination, which was published in July 2013 (Respir. Med. 2013;107:1538-46).
The current trials represent an effort to determine where the treatment fits into the armamentarium for treating patients with COPD, he said.
The “really robust findings as befits two bronchodilators” suggest umeclidinium/vilanterol combination therapy is an effective treatment option that provides greater lung function than fluticasone/salmeterol for moderate to severe COPD in patients with infrequent exacerbations, Dr. Donohue said.
GSK, which developed the umeclidinium/vilanterol combination product with Theravance, funded the studies. Dr. Donohue reported receiving consultant fees and/or serving on an advisory committee for Almirall, AstraZeneca, Boehringer Ingelheim, Dey, Elevation Pharmaceutical, Forest Laboratories, GlaxoSmithKline, Novartis, Pearl Pharmaceuticals, Pfizer, and Sunovion. He has also served as a member of drug safety monitoring boards for the National Institutes of Health, Novartis, Otsuda, Pearl, and Teva.
AUSTIN, TEX.– Once daily combination treatment with umeclidinium and vilanterol was more effective than twice-daily combination treatment with fluticasone and salmeterol in patients with moderate to severe chronic obstructive pulmonary disease in two 12-week double-blind, parallel-group double-dummy studies.
In the two multicenter studies, 706 and 697 patients, respectively, were randomized to receive either 62.5 mcg of the long-acting muscarinic antagonist (LAMA) umeclidinium and 25 mcg of the long-acting beta-2 agonist (LABA) vilanterol – a recently approved combination bronchodilator maintenance treatment for COPD – or a combination of 250 mcg of the inhaled corticosteroid (ICS) fluticasone and 50 mcg of the LABA salmeterol, which is also indicated as a maintenance therapy for COPD.
The patients in the LAMA/LABA groups, who were treated once daily for 12 weeks, had significantly greater improvements on all lung function measures, compared with those in the ICS/LABA groups, who were treated twice daily, Dr. James F. Donohue of the University of North Carolina at Chapel Hill reported at the annual meeting of the American College of Chest Physicians.
In the first study, the improvement from baseline to day 84 (the primary study endpoint) in 0- to 24-hour weighted mean forced expiratory volume in 1 second (FEV1) was 165 mL for the LAMA/LABA group, compared with 91 mL in the ICS/LABA group. In the second study, the improvement in the two groups was 213 mL and 112 mL.
The LAMA/LABA combination also improved trough FEV1 on day 85 by 82 mL and 98 mL more than did the ICS/LABA combination in the two studies, respectively.
Both combinations provided clinically meaningful improvements in dyspnea and quality of life scores, Dr. Donohue said.
Adverse events occurred during treatment in a similar proportion of patients in both treatment groups in both studies: 26% and 27% in the LAMA/LABA and ICS/LABA patients in the first study, and 30% and 31%, respectively, in the second study. The most common adverse events were headache and nasopharyngitis.
In the first study, serious adverse events occurred in 2% of the LAMA/LABA patients and 3% of ICS/LABA patients, and in 3% and 4% of patients in the second study.
One death occurred in the ICS/LABA group in the first study, but it was not considered study related. Five deaths occurred in the second study, including two in the LAMA/LABA patients and three in the ICS/LABA patients. One of the deaths in the ICS/LABA group was because of pneumonia and was reported as drug related by the investigator.
No new safety signals were detected in these studies, Dr. Donohue said.
Patients in both studies had FEV1 between 30% and 70%, and had not experienced a COPD exacerbation within the previous year. The LAMA/LABA therapy was delivered via Ellipta inhaler, and the ICS/LABA therapy was delivered via Diskus inhaler.
The Food and Drug Administration approved umeclidinium/vilanterol combination therapy (Anoro Ellipta) in December 2013, the first LAMA/LABA therapy approved in the United States. Dr. Donohue and his colleagues conducted the pivotal regulatory trial of the drug combination, which was published in July 2013 (Respir. Med. 2013;107:1538-46).
The current trials represent an effort to determine where the treatment fits into the armamentarium for treating patients with COPD, he said.
The “really robust findings as befits two bronchodilators” suggest umeclidinium/vilanterol combination therapy is an effective treatment option that provides greater lung function than fluticasone/salmeterol for moderate to severe COPD in patients with infrequent exacerbations, Dr. Donohue said.
GSK, which developed the umeclidinium/vilanterol combination product with Theravance, funded the studies. Dr. Donohue reported receiving consultant fees and/or serving on an advisory committee for Almirall, AstraZeneca, Boehringer Ingelheim, Dey, Elevation Pharmaceutical, Forest Laboratories, GlaxoSmithKline, Novartis, Pearl Pharmaceuticals, Pfizer, and Sunovion. He has also served as a member of drug safety monitoring boards for the National Institutes of Health, Novartis, Otsuda, Pearl, and Teva.
AUSTIN, TEX.– Once daily combination treatment with umeclidinium and vilanterol was more effective than twice-daily combination treatment with fluticasone and salmeterol in patients with moderate to severe chronic obstructive pulmonary disease in two 12-week double-blind, parallel-group double-dummy studies.
In the two multicenter studies, 706 and 697 patients, respectively, were randomized to receive either 62.5 mcg of the long-acting muscarinic antagonist (LAMA) umeclidinium and 25 mcg of the long-acting beta-2 agonist (LABA) vilanterol – a recently approved combination bronchodilator maintenance treatment for COPD – or a combination of 250 mcg of the inhaled corticosteroid (ICS) fluticasone and 50 mcg of the LABA salmeterol, which is also indicated as a maintenance therapy for COPD.
The patients in the LAMA/LABA groups, who were treated once daily for 12 weeks, had significantly greater improvements on all lung function measures, compared with those in the ICS/LABA groups, who were treated twice daily, Dr. James F. Donohue of the University of North Carolina at Chapel Hill reported at the annual meeting of the American College of Chest Physicians.
In the first study, the improvement from baseline to day 84 (the primary study endpoint) in 0- to 24-hour weighted mean forced expiratory volume in 1 second (FEV1) was 165 mL for the LAMA/LABA group, compared with 91 mL in the ICS/LABA group. In the second study, the improvement in the two groups was 213 mL and 112 mL.
The LAMA/LABA combination also improved trough FEV1 on day 85 by 82 mL and 98 mL more than did the ICS/LABA combination in the two studies, respectively.
Both combinations provided clinically meaningful improvements in dyspnea and quality of life scores, Dr. Donohue said.
Adverse events occurred during treatment in a similar proportion of patients in both treatment groups in both studies: 26% and 27% in the LAMA/LABA and ICS/LABA patients in the first study, and 30% and 31%, respectively, in the second study. The most common adverse events were headache and nasopharyngitis.
In the first study, serious adverse events occurred in 2% of the LAMA/LABA patients and 3% of ICS/LABA patients, and in 3% and 4% of patients in the second study.
One death occurred in the ICS/LABA group in the first study, but it was not considered study related. Five deaths occurred in the second study, including two in the LAMA/LABA patients and three in the ICS/LABA patients. One of the deaths in the ICS/LABA group was because of pneumonia and was reported as drug related by the investigator.
No new safety signals were detected in these studies, Dr. Donohue said.
Patients in both studies had FEV1 between 30% and 70%, and had not experienced a COPD exacerbation within the previous year. The LAMA/LABA therapy was delivered via Ellipta inhaler, and the ICS/LABA therapy was delivered via Diskus inhaler.
The Food and Drug Administration approved umeclidinium/vilanterol combination therapy (Anoro Ellipta) in December 2013, the first LAMA/LABA therapy approved in the United States. Dr. Donohue and his colleagues conducted the pivotal regulatory trial of the drug combination, which was published in July 2013 (Respir. Med. 2013;107:1538-46).
The current trials represent an effort to determine where the treatment fits into the armamentarium for treating patients with COPD, he said.
The “really robust findings as befits two bronchodilators” suggest umeclidinium/vilanterol combination therapy is an effective treatment option that provides greater lung function than fluticasone/salmeterol for moderate to severe COPD in patients with infrequent exacerbations, Dr. Donohue said.
GSK, which developed the umeclidinium/vilanterol combination product with Theravance, funded the studies. Dr. Donohue reported receiving consultant fees and/or serving on an advisory committee for Almirall, AstraZeneca, Boehringer Ingelheim, Dey, Elevation Pharmaceutical, Forest Laboratories, GlaxoSmithKline, Novartis, Pearl Pharmaceuticals, Pfizer, and Sunovion. He has also served as a member of drug safety monitoring boards for the National Institutes of Health, Novartis, Otsuda, Pearl, and Teva.
AT CHEST 2014
Key clinical point: Umeclidinium/vilanterol is an effective treatment option for moderate to severe COPD patients with infrequent exacerbations.
Major finding: The improvement in 0- to 24-hour weighted mean FEV1 on day 84 with LAMA/LABA vs. ICS/LABA was 165 mL vs. 91 mL and 213 mL vs. 112 mL in two randomized trials, respectively.
Data source: Two randomized double-blind, parallel-group double-dummy studies involving 706 and 697 patients.
Disclosures: GSK, which developed the umeclidinium/vilanterol combination product with Theravance, funded the studies. Dr. Donohue reported receiving consultant fees and/or serving on an advisory committee for Almirall, AstraZeneca, Boehringer Ingelheim, Dey, Elevation Pharmaceutical, Forest Laboratories, GlaxoSmithKline, Novartis, Pearl Pharmaceuticals, Pfizer, and Sunovion. He has also served as a member of drug safety monitoring boards for the National Institutes of Health, Novartis, Otsuda, Pearl, and Teva.
Post-PCV13 findings prompt continued surveillance
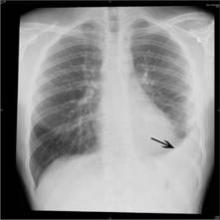
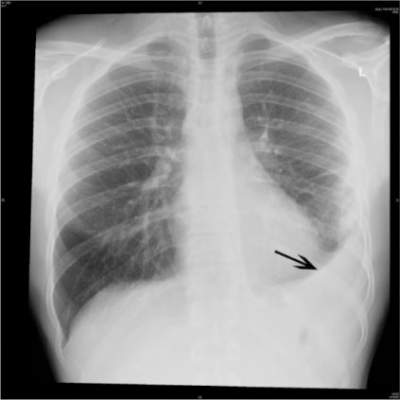
PHILADELPHIA – Invasive pneumococcal infections declined by 42% in 2011, compared with 2007-2009, the years after the introduction of the 13-valent pneumococcal conjugate vaccine, according to an analysis of data from eight U.S. children’s hospitals.
Infections continued to decline – by 48% overall – 2-3 years after full implementation of the vaccine. Declines were seen in the rates of bacteremia, pneumonia, and mastoiditis, but the rates of pneumococcal meningitis in the latter years remained unchanged, with an increasing proportion of cases occurring among children with invasive disease.
The most common 13-valent pneumococcal conjugate vaccine (PCV13) serotypes causing invasive pneumococcal infections in 2011 were 19A and 7F. The declines in invasive infections in 2012 and 2013 were mainly because of declines in serotype 19A and 7F isolates, which decreased by 58% and 54%, respectively, but serotype 3 isolates remained at pre-PCV13 levels, Dr. Sheldon L. Kaplan of Baylor College of Medicine and Texas Children’s Hospital, in Houston, reported at an annual scientific meeting on infectious diseases.
“We had 29 children with invasive pneumococcal disease caused by a PCV13 serotype [in 2012-2013]. All but one were a 19A or serotype 3,” he said at thecombined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Nine children with serotype 19A isolates, and 4 with serotype 3 isolates had received at least two doses of PCV13 prior to developing infections. In 2013, 3 of 12 children with 19A isolates had received four PCV13 doses prior to developing serotype 19A infections.
Only three serotype 1-, 7-, and 19F-related invasive infections occurred during the study period, and no serotype 5–related infections occurred.
In 2012-2013, non-PCV13 serotypes – mainly 33F and 22F – accounted for 71% of isolates, Dr. Kaplan said.
Of note, 45% of children at the eight hospitals that are part of the U.S. Pediatric Multicenter Pneumococcal Surveillance Study Group – which has been conducting pneumococcal surveillance since 1993 – had an underlying condition during 2011-2013, compared with 37% in 2007-2009. The difference was statistically significant, Dr. Kaplan said, noting that malignancies were the most common type of underlying condition, followed by central nervous system disorders, genetic disorder, cardiovascular disorders, and renal disorders.
Non-PCV13 serotypes accounted for 78% of isolates from children with underlying conditions, he said.
Mortality was 0.8% (5 of 626) in 2007-2009, and 3.7% (13 of 352) in 2011-2013. Of the 13 children who died in 2011-2013, 10 had a non-PCV13 serotype, and 9 had an underlying condition, Dr. Kaplan said.
Almost all (99%) of non–central nervous system infections in 2011-2013 were susceptible to penicillin and ceftriaxone.
“Continued surveillance is warranted, especially to monitor non-PCV13 serotypes associated with invasive pneumococcal disease,” he said.
As for pneumococcal meningitis, rates decreased significantly after the introduction of PCV7, but the proportion among children with invasive pneumococcal disease increased by 67% over time, accounting for 12% of cases during 2007-2009, compared with 20% of cases during 2011-2013, according to Dr. Liset Olarte, also of Baylor College of Medicine and Texas Children’s Hospital.
The proportion decreased by 21% among children younger than 24 months, but increased significantly in those aged 24 to 59 months, she noted.
Clinical data showed that in 2007-2009, 19A, 7F, and 3 were the most common serotypes associated with pneumococcal meningitis in the eight hospitals in the surveillance study group. In 2011-13, 19A remained the most common serotype, followed by non-PCV13 serotypes 35B and 22F. Overall, non-PCV13 serotypes causing pneumococcal meningitis increased in both number of cases (24 to 49) and proportion of cases (46% to 73%) after PCV13 introduction.
“In our study, only the change in serotype 7F was statistically significant,” said Dr. Olarte.
The proportion of isolates that were non-susceptible to penicillin did not change significantly from 2007-2009 to 2011-2013 (26% and 25%, respectively), but the proportion nonsusceptible to ceftriaxone, and thus requiring vancomycin for treatment, decreased significantly – by 77% – from 13% to 3%.
All of the isolates with a ceftriaxone minimum inhibitory concentration of 1 mcg/mL or greater and penicillin minimum inhibitory concentration of 2 mcg/mL or greater were serotype 19A, Dr. Olarte said.
Mortality did not change significantly during the study period, but there was a marked, significant increase in hemiparesis cases, from 1% to 12%, after introduction of PCV13. However, this increase was not associated with any particular serotype.
Additionally, subdural empyema on neuroimaging increased from 1% to 16%, but was not associated with any particular serotype and was not associated with the cases of hemiparesis.
Overall, 52% of survivors had some sort of neurologic sequelae at the time of discharge, but there were no differences in terms of specific sequelae between the two groups.
The rates of morbidity and mortality for pneumococcal meningitis remain substantial and have not changed significantly since introduction of PCV13, Dr. Olarte said.
However, if the number of cephalosporin nonsusceptible pneumococci isolated from children with meningitis continues to decrease, modification of empiric antibiotic selection for suspected bacterial meningitis might be considered in the future, therefore ongoing surveillance is warranted, she said.
The studies were partially supported by Pfizer. The authors reported having no other disclosures.
PHILADELPHIA – Invasive pneumococcal infections declined by 42% in 2011, compared with 2007-2009, the years after the introduction of the 13-valent pneumococcal conjugate vaccine, according to an analysis of data from eight U.S. children’s hospitals.
Infections continued to decline – by 48% overall – 2-3 years after full implementation of the vaccine. Declines were seen in the rates of bacteremia, pneumonia, and mastoiditis, but the rates of pneumococcal meningitis in the latter years remained unchanged, with an increasing proportion of cases occurring among children with invasive disease.
The most common 13-valent pneumococcal conjugate vaccine (PCV13) serotypes causing invasive pneumococcal infections in 2011 were 19A and 7F. The declines in invasive infections in 2012 and 2013 were mainly because of declines in serotype 19A and 7F isolates, which decreased by 58% and 54%, respectively, but serotype 3 isolates remained at pre-PCV13 levels, Dr. Sheldon L. Kaplan of Baylor College of Medicine and Texas Children’s Hospital, in Houston, reported at an annual scientific meeting on infectious diseases.
“We had 29 children with invasive pneumococcal disease caused by a PCV13 serotype [in 2012-2013]. All but one were a 19A or serotype 3,” he said at thecombined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Nine children with serotype 19A isolates, and 4 with serotype 3 isolates had received at least two doses of PCV13 prior to developing infections. In 2013, 3 of 12 children with 19A isolates had received four PCV13 doses prior to developing serotype 19A infections.
Only three serotype 1-, 7-, and 19F-related invasive infections occurred during the study period, and no serotype 5–related infections occurred.
In 2012-2013, non-PCV13 serotypes – mainly 33F and 22F – accounted for 71% of isolates, Dr. Kaplan said.
Of note, 45% of children at the eight hospitals that are part of the U.S. Pediatric Multicenter Pneumococcal Surveillance Study Group – which has been conducting pneumococcal surveillance since 1993 – had an underlying condition during 2011-2013, compared with 37% in 2007-2009. The difference was statistically significant, Dr. Kaplan said, noting that malignancies were the most common type of underlying condition, followed by central nervous system disorders, genetic disorder, cardiovascular disorders, and renal disorders.
Non-PCV13 serotypes accounted for 78% of isolates from children with underlying conditions, he said.
Mortality was 0.8% (5 of 626) in 2007-2009, and 3.7% (13 of 352) in 2011-2013. Of the 13 children who died in 2011-2013, 10 had a non-PCV13 serotype, and 9 had an underlying condition, Dr. Kaplan said.
Almost all (99%) of non–central nervous system infections in 2011-2013 were susceptible to penicillin and ceftriaxone.
“Continued surveillance is warranted, especially to monitor non-PCV13 serotypes associated with invasive pneumococcal disease,” he said.
As for pneumococcal meningitis, rates decreased significantly after the introduction of PCV7, but the proportion among children with invasive pneumococcal disease increased by 67% over time, accounting for 12% of cases during 2007-2009, compared with 20% of cases during 2011-2013, according to Dr. Liset Olarte, also of Baylor College of Medicine and Texas Children’s Hospital.
The proportion decreased by 21% among children younger than 24 months, but increased significantly in those aged 24 to 59 months, she noted.
Clinical data showed that in 2007-2009, 19A, 7F, and 3 were the most common serotypes associated with pneumococcal meningitis in the eight hospitals in the surveillance study group. In 2011-13, 19A remained the most common serotype, followed by non-PCV13 serotypes 35B and 22F. Overall, non-PCV13 serotypes causing pneumococcal meningitis increased in both number of cases (24 to 49) and proportion of cases (46% to 73%) after PCV13 introduction.
“In our study, only the change in serotype 7F was statistically significant,” said Dr. Olarte.
The proportion of isolates that were non-susceptible to penicillin did not change significantly from 2007-2009 to 2011-2013 (26% and 25%, respectively), but the proportion nonsusceptible to ceftriaxone, and thus requiring vancomycin for treatment, decreased significantly – by 77% – from 13% to 3%.
All of the isolates with a ceftriaxone minimum inhibitory concentration of 1 mcg/mL or greater and penicillin minimum inhibitory concentration of 2 mcg/mL or greater were serotype 19A, Dr. Olarte said.
Mortality did not change significantly during the study period, but there was a marked, significant increase in hemiparesis cases, from 1% to 12%, after introduction of PCV13. However, this increase was not associated with any particular serotype.
Additionally, subdural empyema on neuroimaging increased from 1% to 16%, but was not associated with any particular serotype and was not associated with the cases of hemiparesis.
Overall, 52% of survivors had some sort of neurologic sequelae at the time of discharge, but there were no differences in terms of specific sequelae between the two groups.
The rates of morbidity and mortality for pneumococcal meningitis remain substantial and have not changed significantly since introduction of PCV13, Dr. Olarte said.
However, if the number of cephalosporin nonsusceptible pneumococci isolated from children with meningitis continues to decrease, modification of empiric antibiotic selection for suspected bacterial meningitis might be considered in the future, therefore ongoing surveillance is warranted, she said.
The studies were partially supported by Pfizer. The authors reported having no other disclosures.
PHILADELPHIA – Invasive pneumococcal infections declined by 42% in 2011, compared with 2007-2009, the years after the introduction of the 13-valent pneumococcal conjugate vaccine, according to an analysis of data from eight U.S. children’s hospitals.
Infections continued to decline – by 48% overall – 2-3 years after full implementation of the vaccine. Declines were seen in the rates of bacteremia, pneumonia, and mastoiditis, but the rates of pneumococcal meningitis in the latter years remained unchanged, with an increasing proportion of cases occurring among children with invasive disease.
The most common 13-valent pneumococcal conjugate vaccine (PCV13) serotypes causing invasive pneumococcal infections in 2011 were 19A and 7F. The declines in invasive infections in 2012 and 2013 were mainly because of declines in serotype 19A and 7F isolates, which decreased by 58% and 54%, respectively, but serotype 3 isolates remained at pre-PCV13 levels, Dr. Sheldon L. Kaplan of Baylor College of Medicine and Texas Children’s Hospital, in Houston, reported at an annual scientific meeting on infectious diseases.
“We had 29 children with invasive pneumococcal disease caused by a PCV13 serotype [in 2012-2013]. All but one were a 19A or serotype 3,” he said at thecombined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Nine children with serotype 19A isolates, and 4 with serotype 3 isolates had received at least two doses of PCV13 prior to developing infections. In 2013, 3 of 12 children with 19A isolates had received four PCV13 doses prior to developing serotype 19A infections.
Only three serotype 1-, 7-, and 19F-related invasive infections occurred during the study period, and no serotype 5–related infections occurred.
In 2012-2013, non-PCV13 serotypes – mainly 33F and 22F – accounted for 71% of isolates, Dr. Kaplan said.
Of note, 45% of children at the eight hospitals that are part of the U.S. Pediatric Multicenter Pneumococcal Surveillance Study Group – which has been conducting pneumococcal surveillance since 1993 – had an underlying condition during 2011-2013, compared with 37% in 2007-2009. The difference was statistically significant, Dr. Kaplan said, noting that malignancies were the most common type of underlying condition, followed by central nervous system disorders, genetic disorder, cardiovascular disorders, and renal disorders.
Non-PCV13 serotypes accounted for 78% of isolates from children with underlying conditions, he said.
Mortality was 0.8% (5 of 626) in 2007-2009, and 3.7% (13 of 352) in 2011-2013. Of the 13 children who died in 2011-2013, 10 had a non-PCV13 serotype, and 9 had an underlying condition, Dr. Kaplan said.
Almost all (99%) of non–central nervous system infections in 2011-2013 were susceptible to penicillin and ceftriaxone.
“Continued surveillance is warranted, especially to monitor non-PCV13 serotypes associated with invasive pneumococcal disease,” he said.
As for pneumococcal meningitis, rates decreased significantly after the introduction of PCV7, but the proportion among children with invasive pneumococcal disease increased by 67% over time, accounting for 12% of cases during 2007-2009, compared with 20% of cases during 2011-2013, according to Dr. Liset Olarte, also of Baylor College of Medicine and Texas Children’s Hospital.
The proportion decreased by 21% among children younger than 24 months, but increased significantly in those aged 24 to 59 months, she noted.
Clinical data showed that in 2007-2009, 19A, 7F, and 3 were the most common serotypes associated with pneumococcal meningitis in the eight hospitals in the surveillance study group. In 2011-13, 19A remained the most common serotype, followed by non-PCV13 serotypes 35B and 22F. Overall, non-PCV13 serotypes causing pneumococcal meningitis increased in both number of cases (24 to 49) and proportion of cases (46% to 73%) after PCV13 introduction.
“In our study, only the change in serotype 7F was statistically significant,” said Dr. Olarte.
The proportion of isolates that were non-susceptible to penicillin did not change significantly from 2007-2009 to 2011-2013 (26% and 25%, respectively), but the proportion nonsusceptible to ceftriaxone, and thus requiring vancomycin for treatment, decreased significantly – by 77% – from 13% to 3%.
All of the isolates with a ceftriaxone minimum inhibitory concentration of 1 mcg/mL or greater and penicillin minimum inhibitory concentration of 2 mcg/mL or greater were serotype 19A, Dr. Olarte said.
Mortality did not change significantly during the study period, but there was a marked, significant increase in hemiparesis cases, from 1% to 12%, after introduction of PCV13. However, this increase was not associated with any particular serotype.
Additionally, subdural empyema on neuroimaging increased from 1% to 16%, but was not associated with any particular serotype and was not associated with the cases of hemiparesis.
Overall, 52% of survivors had some sort of neurologic sequelae at the time of discharge, but there were no differences in terms of specific sequelae between the two groups.
The rates of morbidity and mortality for pneumococcal meningitis remain substantial and have not changed significantly since introduction of PCV13, Dr. Olarte said.
However, if the number of cephalosporin nonsusceptible pneumococci isolated from children with meningitis continues to decrease, modification of empiric antibiotic selection for suspected bacterial meningitis might be considered in the future, therefore ongoing surveillance is warranted, she said.
The studies were partially supported by Pfizer. The authors reported having no other disclosures.
Key clinical point: PCV13 has had a beneficial effect on the rates of invasive pneumococcal infection, but continued surveillance is warranted.
Major finding: Invasive pneumococcal infections have declined by 48% since introduction of PCV13, but rates of pneumococcal meningitis remain unchanged.
Data source: Analyses of data from the eight children’s hospitals in the U.S. Pediatric Multicenter Pneumococcal Surveillance Study Group.
Disclosures: The studies were partially supported by Pfizer. The authors reported having no other disclosures.
Pediatric ID hospitalizations down after vaccine introductions
PHILADELPHIA– Infectious diseases played a primary role in 16% of pediatric hospitalizations between 1997 and 2009, and remain a leading cause of pediatric hospitalizations and health care expenditures, but the proportion of hospitalizations associated with an infectious disease declined by 18% during the study period, according to an analysis of the Kids’ Inpatient Database.
Periods of decline were temporally associated with the introduction of pneumococcal and rotavirus vaccines, Chris Stockmann of the University of Utah Health Sciences Center, Salt Lake City, reported at an annual scientific meeting on infectious diseases.
Infectious diseases were second only to newborn deliveries as a cause of pediatric hospitalizations during the study period, Mr. Stockmann said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The burden was highest among children under age 5 years, who accounted for about 75% of all infectious disease hospitalizations; those under age 3 months accounted for 15%.
Eight diagnoses accounted for more than 80% of the hospitalizations. The most common were pneumonia and bronchiolitis, followed by urinary tract infections, other upper respiratory tract infections, intestinal infections, other viral infections, sepsis, and meningitis. Respiratory infections accounted for about two-thirds of all hospitalizations, he said.
Pneumonia was the leading cause of hospitalizations across all age groups. The second leading cause was bronchiolitis in those under age 5 years, and UTI in those aged 5 years and older.
“A smattering of other conditions, including intestinal infections, viral infections, and meningitis, also make an appearance in the top 4,” he said.
Over time, pneumonia, bronchiolitis, and other upper respiratory tract infections decreased by 15% to 30%, and the intestinal infection rate was nearly halved.
The decrease in pneumonia hospitalizations coincided with the introduction of the 7-valent pneumococcal conjugate vaccine in 2000, and the introduction of the rotavirus vaccine in 2007. The introduction of these vaccines also was associated with a reduction in intestinal infection hospitalizations, he said.
During the study period, the overall charge for all infectious disease hospitalizations was $4.3 billion per year nationally. At the patient level, the mean charge was just under $15,000, and the mean length of stay was just over 3 days, Mr. Stockmann said.
Total hospital charges were driven by pneumonia and bronchiolitis, but at the individual patient level, sepsis and meningitis were the most expensive conditions, he noted.
The data for this analysis were derived from the National Inpatient Survey’s Kids’ Inpatient Database (KID), owned and operated by the Agency for Healthcare Research and Quality, and maintained by the Healthcare Cost and Utilization Project. Currently, 3,500 hospitals in 44 states contribute data to KID, which is released every 3 years; data for this study were available for 1997, 2000, 2003, 2006, and 2009 and were weighted with a complex survey design to more than 36 million discharges nationwide.
The findings underscore the need for additional efforts to reduce the burden of pediatric respiratory tract infections, Mr. Stockmann said. “We would posit that respiratory viral vaccines may play a key role in that,” he said.
Mr. Stockmann reported having no disclosures.
PHILADELPHIA– Infectious diseases played a primary role in 16% of pediatric hospitalizations between 1997 and 2009, and remain a leading cause of pediatric hospitalizations and health care expenditures, but the proportion of hospitalizations associated with an infectious disease declined by 18% during the study period, according to an analysis of the Kids’ Inpatient Database.
Periods of decline were temporally associated with the introduction of pneumococcal and rotavirus vaccines, Chris Stockmann of the University of Utah Health Sciences Center, Salt Lake City, reported at an annual scientific meeting on infectious diseases.
Infectious diseases were second only to newborn deliveries as a cause of pediatric hospitalizations during the study period, Mr. Stockmann said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The burden was highest among children under age 5 years, who accounted for about 75% of all infectious disease hospitalizations; those under age 3 months accounted for 15%.
Eight diagnoses accounted for more than 80% of the hospitalizations. The most common were pneumonia and bronchiolitis, followed by urinary tract infections, other upper respiratory tract infections, intestinal infections, other viral infections, sepsis, and meningitis. Respiratory infections accounted for about two-thirds of all hospitalizations, he said.
Pneumonia was the leading cause of hospitalizations across all age groups. The second leading cause was bronchiolitis in those under age 5 years, and UTI in those aged 5 years and older.
“A smattering of other conditions, including intestinal infections, viral infections, and meningitis, also make an appearance in the top 4,” he said.
Over time, pneumonia, bronchiolitis, and other upper respiratory tract infections decreased by 15% to 30%, and the intestinal infection rate was nearly halved.
The decrease in pneumonia hospitalizations coincided with the introduction of the 7-valent pneumococcal conjugate vaccine in 2000, and the introduction of the rotavirus vaccine in 2007. The introduction of these vaccines also was associated with a reduction in intestinal infection hospitalizations, he said.
During the study period, the overall charge for all infectious disease hospitalizations was $4.3 billion per year nationally. At the patient level, the mean charge was just under $15,000, and the mean length of stay was just over 3 days, Mr. Stockmann said.
Total hospital charges were driven by pneumonia and bronchiolitis, but at the individual patient level, sepsis and meningitis were the most expensive conditions, he noted.
The data for this analysis were derived from the National Inpatient Survey’s Kids’ Inpatient Database (KID), owned and operated by the Agency for Healthcare Research and Quality, and maintained by the Healthcare Cost and Utilization Project. Currently, 3,500 hospitals in 44 states contribute data to KID, which is released every 3 years; data for this study were available for 1997, 2000, 2003, 2006, and 2009 and were weighted with a complex survey design to more than 36 million discharges nationwide.
The findings underscore the need for additional efforts to reduce the burden of pediatric respiratory tract infections, Mr. Stockmann said. “We would posit that respiratory viral vaccines may play a key role in that,” he said.
Mr. Stockmann reported having no disclosures.
PHILADELPHIA– Infectious diseases played a primary role in 16% of pediatric hospitalizations between 1997 and 2009, and remain a leading cause of pediatric hospitalizations and health care expenditures, but the proportion of hospitalizations associated with an infectious disease declined by 18% during the study period, according to an analysis of the Kids’ Inpatient Database.
Periods of decline were temporally associated with the introduction of pneumococcal and rotavirus vaccines, Chris Stockmann of the University of Utah Health Sciences Center, Salt Lake City, reported at an annual scientific meeting on infectious diseases.
Infectious diseases were second only to newborn deliveries as a cause of pediatric hospitalizations during the study period, Mr. Stockmann said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The burden was highest among children under age 5 years, who accounted for about 75% of all infectious disease hospitalizations; those under age 3 months accounted for 15%.
Eight diagnoses accounted for more than 80% of the hospitalizations. The most common were pneumonia and bronchiolitis, followed by urinary tract infections, other upper respiratory tract infections, intestinal infections, other viral infections, sepsis, and meningitis. Respiratory infections accounted for about two-thirds of all hospitalizations, he said.
Pneumonia was the leading cause of hospitalizations across all age groups. The second leading cause was bronchiolitis in those under age 5 years, and UTI in those aged 5 years and older.
“A smattering of other conditions, including intestinal infections, viral infections, and meningitis, also make an appearance in the top 4,” he said.
Over time, pneumonia, bronchiolitis, and other upper respiratory tract infections decreased by 15% to 30%, and the intestinal infection rate was nearly halved.
The decrease in pneumonia hospitalizations coincided with the introduction of the 7-valent pneumococcal conjugate vaccine in 2000, and the introduction of the rotavirus vaccine in 2007. The introduction of these vaccines also was associated with a reduction in intestinal infection hospitalizations, he said.
During the study period, the overall charge for all infectious disease hospitalizations was $4.3 billion per year nationally. At the patient level, the mean charge was just under $15,000, and the mean length of stay was just over 3 days, Mr. Stockmann said.
Total hospital charges were driven by pneumonia and bronchiolitis, but at the individual patient level, sepsis and meningitis were the most expensive conditions, he noted.
The data for this analysis were derived from the National Inpatient Survey’s Kids’ Inpatient Database (KID), owned and operated by the Agency for Healthcare Research and Quality, and maintained by the Healthcare Cost and Utilization Project. Currently, 3,500 hospitals in 44 states contribute data to KID, which is released every 3 years; data for this study were available for 1997, 2000, 2003, 2006, and 2009 and were weighted with a complex survey design to more than 36 million discharges nationwide.
The findings underscore the need for additional efforts to reduce the burden of pediatric respiratory tract infections, Mr. Stockmann said. “We would posit that respiratory viral vaccines may play a key role in that,” he said.
Mr. Stockmann reported having no disclosures.
Key clinical point: Pneumococcal and rotavirus vaccine introductions have been associated with decreases in the rate of pediatric infectious disease hospitalizations.
Major finding: Pediatric infectious disease hospitalizations declined by 18% between 1997 and 2009.
Data source: An analysis of the Kids’ Inpatient Database weighted to 36.3 million discharges.
Disclosures: Mr. Stockmann reported having no disclosures.
Treating HPV vaccine as ‘routine’ ups vaccination rate
PHILADELPHIA– A “routine recommendation” approach was the most effective but least used strategy for promoting same-day human papillomavirus vaccination in adolescent boys and girls, according to an industry-funded study of more than 150 well-visit interactions.
Of 204 well visits with 20 pediatricians, 167 (82%) involved discussion of adolescent vaccination, and 152 (75%) specifically mentioned HPV vaccination.
Vaccine recommendation approaches used in the recorded visits were categorized into three styles: “routine recommendation,” which was used in 11% of visits, yielding a 94% same-day vaccination rate; “brief mention,” which was used in 17% of visits, yielding a 27% same-day vaccination rate; and “detailed discussion,” which was used in 72% of visits, yielding a 38% same-day vaccination rate, Dr. Liana R. Clark reported at an annual scientific meeting on infectious diseases.
More specifically, the routine recommendation approach was used during 15% of visits with 32 girls aged 11-12 years, 7% of visits with 42 boys aged 11-12 years, and 10% of visits with 78 boys aged 13-18 years, yielding same-day vaccination rates of 80%, 100%, and 100%, respectively.
The brief intervention approach was used in 19%, 29%, and 13% of the visits, yielding same-day vaccination rates of 17%, 8%, and 20%, respectively. And the detailed discussion approach was used in 66%, 64%, and 77% of the visits, yielding same-day vaccination rates of 19%, 33%, and 47%, respectively, Dr. Clark of Merck Vaccines, West Point, Pa., said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The visits recorded for this study took place between January 2013 and June 2013, and involved girls aged 11-12 years and boys aged 11-18 years. Physician participants were members of a panel for a dialogue research company. While the physicians knew their interactions were being recorded, they were not aware that HPV vaccination approach was being evaluated.
The vaccination recommendation–style categories were based on an analysis of the visit recordings. The “routine recommendation” style involved a matter-of-fact recommendation made the same way recommendations for other adolescent vaccines were made. The “brief mention” style involved mention of the vaccine without a recommendation or direction. And the “detailed discussion” style involved a discussion with a parent in greater detail than was provided for other adolescent vaccines.
The findings of this study – the first qualitative project to assess how physicians discuss and recommend HPV vaccination to patients and caregivers – are important, because HPV vaccination rates in the United States remain relatively low, Dr. Clark explained.
Of note, physicians report significant parental barriers to acceptance of routine HPV vaccine recommendations, but parents report a high likelihood of accepting the recommendations of the provider, she added.
Evaluation of the dialogue between physicians and patients in this study suggests that physician recommendations to patients are not consistent with vaccination recommendations from the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices, Dr. Clark said, and many physicians are uncomfortable discussing HPV vaccination.
In addition, some physicians recommended vaccination at age 14-15 years and/or tied vaccination recommendations to sexual activity, rather than recommending vaccination at age 11-12 years. Many physicians also left the decision up to the caregiver and failed to make a strong recommendation, Dr. Clark said. Furthermore, many physicians told patients and parents that vaccination did not need to happen that day, allowing for a missed vaccination opportunity.
Though limited by the small pediatrician sample size and the fact that the participating physicians were part of a paid panel and thus may not be representative of the broader pediatric community, the findings suggest that pediatricians may benefit from training to improve their HPV vaccination recommendations, Dr. Clark said.
Dr. Clark is an employee of Merck Vaccines.
PHILADELPHIA– A “routine recommendation” approach was the most effective but least used strategy for promoting same-day human papillomavirus vaccination in adolescent boys and girls, according to an industry-funded study of more than 150 well-visit interactions.
Of 204 well visits with 20 pediatricians, 167 (82%) involved discussion of adolescent vaccination, and 152 (75%) specifically mentioned HPV vaccination.
Vaccine recommendation approaches used in the recorded visits were categorized into three styles: “routine recommendation,” which was used in 11% of visits, yielding a 94% same-day vaccination rate; “brief mention,” which was used in 17% of visits, yielding a 27% same-day vaccination rate; and “detailed discussion,” which was used in 72% of visits, yielding a 38% same-day vaccination rate, Dr. Liana R. Clark reported at an annual scientific meeting on infectious diseases.
More specifically, the routine recommendation approach was used during 15% of visits with 32 girls aged 11-12 years, 7% of visits with 42 boys aged 11-12 years, and 10% of visits with 78 boys aged 13-18 years, yielding same-day vaccination rates of 80%, 100%, and 100%, respectively.
The brief intervention approach was used in 19%, 29%, and 13% of the visits, yielding same-day vaccination rates of 17%, 8%, and 20%, respectively. And the detailed discussion approach was used in 66%, 64%, and 77% of the visits, yielding same-day vaccination rates of 19%, 33%, and 47%, respectively, Dr. Clark of Merck Vaccines, West Point, Pa., said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The visits recorded for this study took place between January 2013 and June 2013, and involved girls aged 11-12 years and boys aged 11-18 years. Physician participants were members of a panel for a dialogue research company. While the physicians knew their interactions were being recorded, they were not aware that HPV vaccination approach was being evaluated.
The vaccination recommendation–style categories were based on an analysis of the visit recordings. The “routine recommendation” style involved a matter-of-fact recommendation made the same way recommendations for other adolescent vaccines were made. The “brief mention” style involved mention of the vaccine without a recommendation or direction. And the “detailed discussion” style involved a discussion with a parent in greater detail than was provided for other adolescent vaccines.
The findings of this study – the first qualitative project to assess how physicians discuss and recommend HPV vaccination to patients and caregivers – are important, because HPV vaccination rates in the United States remain relatively low, Dr. Clark explained.
Of note, physicians report significant parental barriers to acceptance of routine HPV vaccine recommendations, but parents report a high likelihood of accepting the recommendations of the provider, she added.
Evaluation of the dialogue between physicians and patients in this study suggests that physician recommendations to patients are not consistent with vaccination recommendations from the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices, Dr. Clark said, and many physicians are uncomfortable discussing HPV vaccination.
In addition, some physicians recommended vaccination at age 14-15 years and/or tied vaccination recommendations to sexual activity, rather than recommending vaccination at age 11-12 years. Many physicians also left the decision up to the caregiver and failed to make a strong recommendation, Dr. Clark said. Furthermore, many physicians told patients and parents that vaccination did not need to happen that day, allowing for a missed vaccination opportunity.
Though limited by the small pediatrician sample size and the fact that the participating physicians were part of a paid panel and thus may not be representative of the broader pediatric community, the findings suggest that pediatricians may benefit from training to improve their HPV vaccination recommendations, Dr. Clark said.
Dr. Clark is an employee of Merck Vaccines.
PHILADELPHIA– A “routine recommendation” approach was the most effective but least used strategy for promoting same-day human papillomavirus vaccination in adolescent boys and girls, according to an industry-funded study of more than 150 well-visit interactions.
Of 204 well visits with 20 pediatricians, 167 (82%) involved discussion of adolescent vaccination, and 152 (75%) specifically mentioned HPV vaccination.
Vaccine recommendation approaches used in the recorded visits were categorized into three styles: “routine recommendation,” which was used in 11% of visits, yielding a 94% same-day vaccination rate; “brief mention,” which was used in 17% of visits, yielding a 27% same-day vaccination rate; and “detailed discussion,” which was used in 72% of visits, yielding a 38% same-day vaccination rate, Dr. Liana R. Clark reported at an annual scientific meeting on infectious diseases.
More specifically, the routine recommendation approach was used during 15% of visits with 32 girls aged 11-12 years, 7% of visits with 42 boys aged 11-12 years, and 10% of visits with 78 boys aged 13-18 years, yielding same-day vaccination rates of 80%, 100%, and 100%, respectively.
The brief intervention approach was used in 19%, 29%, and 13% of the visits, yielding same-day vaccination rates of 17%, 8%, and 20%, respectively. And the detailed discussion approach was used in 66%, 64%, and 77% of the visits, yielding same-day vaccination rates of 19%, 33%, and 47%, respectively, Dr. Clark of Merck Vaccines, West Point, Pa., said at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The visits recorded for this study took place between January 2013 and June 2013, and involved girls aged 11-12 years and boys aged 11-18 years. Physician participants were members of a panel for a dialogue research company. While the physicians knew their interactions were being recorded, they were not aware that HPV vaccination approach was being evaluated.
The vaccination recommendation–style categories were based on an analysis of the visit recordings. The “routine recommendation” style involved a matter-of-fact recommendation made the same way recommendations for other adolescent vaccines were made. The “brief mention” style involved mention of the vaccine without a recommendation or direction. And the “detailed discussion” style involved a discussion with a parent in greater detail than was provided for other adolescent vaccines.
The findings of this study – the first qualitative project to assess how physicians discuss and recommend HPV vaccination to patients and caregivers – are important, because HPV vaccination rates in the United States remain relatively low, Dr. Clark explained.
Of note, physicians report significant parental barriers to acceptance of routine HPV vaccine recommendations, but parents report a high likelihood of accepting the recommendations of the provider, she added.
Evaluation of the dialogue between physicians and patients in this study suggests that physician recommendations to patients are not consistent with vaccination recommendations from the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices, Dr. Clark said, and many physicians are uncomfortable discussing HPV vaccination.
In addition, some physicians recommended vaccination at age 14-15 years and/or tied vaccination recommendations to sexual activity, rather than recommending vaccination at age 11-12 years. Many physicians also left the decision up to the caregiver and failed to make a strong recommendation, Dr. Clark said. Furthermore, many physicians told patients and parents that vaccination did not need to happen that day, allowing for a missed vaccination opportunity.
Though limited by the small pediatrician sample size and the fact that the participating physicians were part of a paid panel and thus may not be representative of the broader pediatric community, the findings suggest that pediatricians may benefit from training to improve their HPV vaccination recommendations, Dr. Clark said.
Dr. Clark is an employee of Merck Vaccines.
AT IDWEEK 2014
Key clinical point: Treating HPV vaccination as a routine part of pediatric care may increase vaccination rates.
Major finding: A “routine recommendation” approach to HPV vaccine was used by only 11% of physicians, but yielded a 94% same-day vaccination rate.
Data source: A qualitative study of 204 patient visits with 20 pediatricians.
Disclosures: Dr. Clark is an employee of Merck Vaccines.
Lower frequency glatiramer acetate regimen appears safer
BOSTON– Delivering subcutaneous glatiramer acetate as a 40-mg/mL dose three times weekly rather than 20 mg/mL daily halved the annualized rate of injection-related adverse events in a randomized, open-label, phase IIIb trial of patients with relapsing-remitting multiple sclerosis.
The reduction in the annualized rate of moderate and severe injection-related adverse events in those with lower-frequency dosing was even greater, at about 60%, in the trial, called GLACIER (Glatiramer Acetate Low Frequency Safety and Patient Experience), Dr. Jerry S. Wolinsky reported at the joint meeting of the European and Americas committees for Treatment and Research in Multiple Sclerosis.
“As we all know, long-term, frequent injections are required for the first-line therapies for relapsing forms of multiple sclerosis, and this form of drug delivery presents a challenge for patients. Modification of the treatment regimen, such as using alternative dosages and lower-frequency administration schedules for these drugs with long-term proven efficacy, has the potential to improve the patient experience by reducing, perhaps, the number of adverse experiences that they have, improving tolerability, and in particular, potentially increasing adherence to treatments, especially in the longer-term,” Dr. Wolinsky said, explaining the basis for the GLACIER trial.
Glatiramer acetate 20 mg/mL administered by daily subcutaneous injection is a well-characterized treatment with a good long-term safety profile. It has been used for more than 20 years with more than 2 million patient-years of overall exposures, and it has established efficacy for the treatment of relapsing-remitting MS (RRMS). In January, the Food and Drug Administration approved a supplemental New Drug Application for the 40-mg/mL three times weekly regimen after it was shown to have an efficacy and safety profile similar to that of the 20-mg/mL regimen.
The GLACIER findings suggest that the glatiramer acetate 40-mg/mL three times per week dosing regimen is, for some patients, a favorable treatment option, particularly in those who want to be on glatiramer acetate, but who prefer fewer injections and more convenient dosing, concluded Dr. Wolinsky, director of the multiple sclerosis research group at the University of Texas Health Science Center at Houston.
The annualized rate of injection-related adverse events (IRAEs) in 108 adult patients with RRMS who were randomized to receive the thrice weekly injections was 35.3, compared with 70.4 in 101 patients who received daily injections (relative risk, 0.50). The annualized rate of moderate and severe IRAEs was 0.88 vs. 2.2 in the groups, respectively (relative risk, 0.40), he said.
The most common injection site reactions were pain, erythema, mass, swelling, and pruritus, and all of these were substantially reduced with lower-frequency dosing, compared with daily dosing. For example, the annualized rate of pain events was 24.8 vs. 55.3 with lower- vs. higher-frequency dosing, and the annualized rate of erythema events was 21.4 vs. 43.5.
GLACIER trial patients were aged 18 years or older (mean of 51 years) with a diagnosis of RRMS and an Expanded Disability Status Scale score of 0-5.5. Most (82%) were women, and 94% were white.
All were treated continuously with the 20-mg/mL daily dosing regimen for at least 6 months and were stable on that regimen for at least 2 months prior to screening for the GLACIER trial.
At least 75% treatment compliance was achieved by 99% of the GLACIER trial patients, and only 10 withdrew before completing the 4-month treatment phase, including 3 in the daily dosing group, and 7 in the lower-frequency dosing group. Only one patient withdrew because of an adverse event, Dr. Wolinsky said.
The GLACIER trial was funded by Teva Pharmaceutical Industries. Dr. Wolinsky disclosed ties to Teva and other companies marketing MS drugs.
BOSTON– Delivering subcutaneous glatiramer acetate as a 40-mg/mL dose three times weekly rather than 20 mg/mL daily halved the annualized rate of injection-related adverse events in a randomized, open-label, phase IIIb trial of patients with relapsing-remitting multiple sclerosis.
The reduction in the annualized rate of moderate and severe injection-related adverse events in those with lower-frequency dosing was even greater, at about 60%, in the trial, called GLACIER (Glatiramer Acetate Low Frequency Safety and Patient Experience), Dr. Jerry S. Wolinsky reported at the joint meeting of the European and Americas committees for Treatment and Research in Multiple Sclerosis.
“As we all know, long-term, frequent injections are required for the first-line therapies for relapsing forms of multiple sclerosis, and this form of drug delivery presents a challenge for patients. Modification of the treatment regimen, such as using alternative dosages and lower-frequency administration schedules for these drugs with long-term proven efficacy, has the potential to improve the patient experience by reducing, perhaps, the number of adverse experiences that they have, improving tolerability, and in particular, potentially increasing adherence to treatments, especially in the longer-term,” Dr. Wolinsky said, explaining the basis for the GLACIER trial.
Glatiramer acetate 20 mg/mL administered by daily subcutaneous injection is a well-characterized treatment with a good long-term safety profile. It has been used for more than 20 years with more than 2 million patient-years of overall exposures, and it has established efficacy for the treatment of relapsing-remitting MS (RRMS). In January, the Food and Drug Administration approved a supplemental New Drug Application for the 40-mg/mL three times weekly regimen after it was shown to have an efficacy and safety profile similar to that of the 20-mg/mL regimen.
The GLACIER findings suggest that the glatiramer acetate 40-mg/mL three times per week dosing regimen is, for some patients, a favorable treatment option, particularly in those who want to be on glatiramer acetate, but who prefer fewer injections and more convenient dosing, concluded Dr. Wolinsky, director of the multiple sclerosis research group at the University of Texas Health Science Center at Houston.
The annualized rate of injection-related adverse events (IRAEs) in 108 adult patients with RRMS who were randomized to receive the thrice weekly injections was 35.3, compared with 70.4 in 101 patients who received daily injections (relative risk, 0.50). The annualized rate of moderate and severe IRAEs was 0.88 vs. 2.2 in the groups, respectively (relative risk, 0.40), he said.
The most common injection site reactions were pain, erythema, mass, swelling, and pruritus, and all of these were substantially reduced with lower-frequency dosing, compared with daily dosing. For example, the annualized rate of pain events was 24.8 vs. 55.3 with lower- vs. higher-frequency dosing, and the annualized rate of erythema events was 21.4 vs. 43.5.
GLACIER trial patients were aged 18 years or older (mean of 51 years) with a diagnosis of RRMS and an Expanded Disability Status Scale score of 0-5.5. Most (82%) were women, and 94% were white.
All were treated continuously with the 20-mg/mL daily dosing regimen for at least 6 months and were stable on that regimen for at least 2 months prior to screening for the GLACIER trial.
At least 75% treatment compliance was achieved by 99% of the GLACIER trial patients, and only 10 withdrew before completing the 4-month treatment phase, including 3 in the daily dosing group, and 7 in the lower-frequency dosing group. Only one patient withdrew because of an adverse event, Dr. Wolinsky said.
The GLACIER trial was funded by Teva Pharmaceutical Industries. Dr. Wolinsky disclosed ties to Teva and other companies marketing MS drugs.
BOSTON– Delivering subcutaneous glatiramer acetate as a 40-mg/mL dose three times weekly rather than 20 mg/mL daily halved the annualized rate of injection-related adverse events in a randomized, open-label, phase IIIb trial of patients with relapsing-remitting multiple sclerosis.
The reduction in the annualized rate of moderate and severe injection-related adverse events in those with lower-frequency dosing was even greater, at about 60%, in the trial, called GLACIER (Glatiramer Acetate Low Frequency Safety and Patient Experience), Dr. Jerry S. Wolinsky reported at the joint meeting of the European and Americas committees for Treatment and Research in Multiple Sclerosis.
“As we all know, long-term, frequent injections are required for the first-line therapies for relapsing forms of multiple sclerosis, and this form of drug delivery presents a challenge for patients. Modification of the treatment regimen, such as using alternative dosages and lower-frequency administration schedules for these drugs with long-term proven efficacy, has the potential to improve the patient experience by reducing, perhaps, the number of adverse experiences that they have, improving tolerability, and in particular, potentially increasing adherence to treatments, especially in the longer-term,” Dr. Wolinsky said, explaining the basis for the GLACIER trial.
Glatiramer acetate 20 mg/mL administered by daily subcutaneous injection is a well-characterized treatment with a good long-term safety profile. It has been used for more than 20 years with more than 2 million patient-years of overall exposures, and it has established efficacy for the treatment of relapsing-remitting MS (RRMS). In January, the Food and Drug Administration approved a supplemental New Drug Application for the 40-mg/mL three times weekly regimen after it was shown to have an efficacy and safety profile similar to that of the 20-mg/mL regimen.
The GLACIER findings suggest that the glatiramer acetate 40-mg/mL three times per week dosing regimen is, for some patients, a favorable treatment option, particularly in those who want to be on glatiramer acetate, but who prefer fewer injections and more convenient dosing, concluded Dr. Wolinsky, director of the multiple sclerosis research group at the University of Texas Health Science Center at Houston.
The annualized rate of injection-related adverse events (IRAEs) in 108 adult patients with RRMS who were randomized to receive the thrice weekly injections was 35.3, compared with 70.4 in 101 patients who received daily injections (relative risk, 0.50). The annualized rate of moderate and severe IRAEs was 0.88 vs. 2.2 in the groups, respectively (relative risk, 0.40), he said.
The most common injection site reactions were pain, erythema, mass, swelling, and pruritus, and all of these were substantially reduced with lower-frequency dosing, compared with daily dosing. For example, the annualized rate of pain events was 24.8 vs. 55.3 with lower- vs. higher-frequency dosing, and the annualized rate of erythema events was 21.4 vs. 43.5.
GLACIER trial patients were aged 18 years or older (mean of 51 years) with a diagnosis of RRMS and an Expanded Disability Status Scale score of 0-5.5. Most (82%) were women, and 94% were white.
All were treated continuously with the 20-mg/mL daily dosing regimen for at least 6 months and were stable on that regimen for at least 2 months prior to screening for the GLACIER trial.
At least 75% treatment compliance was achieved by 99% of the GLACIER trial patients, and only 10 withdrew before completing the 4-month treatment phase, including 3 in the daily dosing group, and 7 in the lower-frequency dosing group. Only one patient withdrew because of an adverse event, Dr. Wolinsky said.
The GLACIER trial was funded by Teva Pharmaceutical Industries. Dr. Wolinsky disclosed ties to Teva and other companies marketing MS drugs.
AT MSBOSTON 2014
Key clinical point: The glatiramer acetate 40-mg/mL three times per week dosing regimen may be a good option for patients who prefer fewer injections and more convenient dosing.
Major finding: The relative risk of IRAEs for lower- vs. higher-frequency dosing was 0.50.
Data source: The randomized, open-label GLACIER trial involving 209 patients.
Disclosures: Dr. Wolinsky disclosed ties to Teva Pharmaceutical Industries, which funded the trial, and other companies marketing MS drugs.
Contact sports raise risk of MRSA carriage by college athletes
PHILADELPHIA – College athletes who play contact sports are more likely than are those who play noncontact sports to carry methicillin-resistant Staphylococcus aureus, putting them at higher risk for infection and for infecting others, according to findings from a 2-year observational study.
During the course of the study, MRSA colonization ranged from 8% to 31% in 224 students who played contact sports, including football, basketball, soccer, and lacrosse, compared with 0% to 23% in 153 students who participated in noncontact or limited-contact sports, including baseball, cross country, and golf. MRSA colonization in the general population ranges from 5% to 10%, Natalia Jimenez-Truque, Ph.D., of Vanderbilt University Medical Center, Nashville, Tenn., reported at an annual scientific meeting on infectious diseases.
Overall, contact sports athletes in this study had more than twice the risk of being colonized with MRSA (odds ratio 2.3), and tended to carry S. aureus for longer period of time (OR 3.0 for intermittent carriage, 2.39 for persistent carriage), compared with noncontact sports athletes. Also, contact sports athletes who were not carriers at baseline acquired S. aureus more quickly (hazard ratio 1.6), Dr. Jimenez-Truque said during a press conference at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The findings have implications for advising young athletes about the importance of good hygiene practices, including covering any open wounds, washing hands frequently, showering as soon as possible after all practices and games, laundering gym clothes, bags, and towels promptly, and not sharing razors and other personal hygiene items, she said.
Athletes are known to have a higher risk of infection with S. aureus than the general population, but most studies have not assessed staphylococcal carriage or compared risk in contact and noncontact sports participants, Dr. Jimenez-Truque said. The lack of available information about the natural history of carriage across sports limits strategies for preventing infection in athletes, she noted.
For the current study – the first to observe both male and female college athletes who were not part of a MRSA outbreak – Dr. Jimenez-Truque and her colleagues followed varsity collegiate athletes from August 2008 to April 2010. Nasal and oropharyngeal swabs were collected at enrollment and monthly thereafter.
The increased rates of MRSA carriage and acquisition in contact sports athletes in this study may be associated with increased likelihood of having cuts, scrapes, turf burns, and skin-to-skin contact with other athletes, further underscoring the need for good hygiene, she said.
MRSA is a leading cause of both invasive and skin and soft tissue infections. Athletes are commonly affected, and up to 70% of those who develop infection require hospitalization and intravenous antibiotic treatment.
However, despite the “really significant number” of contact sports athletes who carry MRSA, the number who become infected is small, Dr. Jimenez-Truque said.
“What that tells us, we think, is that colonization with specific strains of staph in specific people at specific times is needed for infections to occur, and therefore for outbreaks to occur. When we put it in that sort of framework, we begin to realize that we need to understand the epidemiology of specific strains of S. aureus, we need to understand the periods of time when athletes are at highest risk – that’s probably when they’re newly acquiring strains that they’ve never seen before – and we need to take into account things that we can do that may be unrelated to staph, but may affect their risk for infection,” said lead author C. Buddy Creech, also of Vanderbilt University Medical Center.
For example, during the course of the current study, a cluster of infections “perfectly coincided” with the development of pandemic H1N1 influenza in a number of athletes, Dr. Creech said, adding that even though the two conditions are unrelated, this relationship between S. aureus and influenza shouldn’t be diminished.
“So even though it’s unrelated, all of the things we do to protect collegiate and professional athletes, like influenza vaccination and general health maintenance ... go a long way, and may have widespread implications,” said Dr. Creech.
He noted however, that although strategies of covering compromised skin and wounds are important for preventing the spread of disease, they are comparable to “putting our finger in the dike while we wait for other preventive strategies like vaccination or other more powerful strategies.”
Dr. Creech and Dr. Jimenez-Truque reported having no disclosures.
PHILADELPHIA – College athletes who play contact sports are more likely than are those who play noncontact sports to carry methicillin-resistant Staphylococcus aureus, putting them at higher risk for infection and for infecting others, according to findings from a 2-year observational study.
During the course of the study, MRSA colonization ranged from 8% to 31% in 224 students who played contact sports, including football, basketball, soccer, and lacrosse, compared with 0% to 23% in 153 students who participated in noncontact or limited-contact sports, including baseball, cross country, and golf. MRSA colonization in the general population ranges from 5% to 10%, Natalia Jimenez-Truque, Ph.D., of Vanderbilt University Medical Center, Nashville, Tenn., reported at an annual scientific meeting on infectious diseases.
Overall, contact sports athletes in this study had more than twice the risk of being colonized with MRSA (odds ratio 2.3), and tended to carry S. aureus for longer period of time (OR 3.0 for intermittent carriage, 2.39 for persistent carriage), compared with noncontact sports athletes. Also, contact sports athletes who were not carriers at baseline acquired S. aureus more quickly (hazard ratio 1.6), Dr. Jimenez-Truque said during a press conference at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The findings have implications for advising young athletes about the importance of good hygiene practices, including covering any open wounds, washing hands frequently, showering as soon as possible after all practices and games, laundering gym clothes, bags, and towels promptly, and not sharing razors and other personal hygiene items, she said.
Athletes are known to have a higher risk of infection with S. aureus than the general population, but most studies have not assessed staphylococcal carriage or compared risk in contact and noncontact sports participants, Dr. Jimenez-Truque said. The lack of available information about the natural history of carriage across sports limits strategies for preventing infection in athletes, she noted.
For the current study – the first to observe both male and female college athletes who were not part of a MRSA outbreak – Dr. Jimenez-Truque and her colleagues followed varsity collegiate athletes from August 2008 to April 2010. Nasal and oropharyngeal swabs were collected at enrollment and monthly thereafter.
The increased rates of MRSA carriage and acquisition in contact sports athletes in this study may be associated with increased likelihood of having cuts, scrapes, turf burns, and skin-to-skin contact with other athletes, further underscoring the need for good hygiene, she said.
MRSA is a leading cause of both invasive and skin and soft tissue infections. Athletes are commonly affected, and up to 70% of those who develop infection require hospitalization and intravenous antibiotic treatment.
However, despite the “really significant number” of contact sports athletes who carry MRSA, the number who become infected is small, Dr. Jimenez-Truque said.
“What that tells us, we think, is that colonization with specific strains of staph in specific people at specific times is needed for infections to occur, and therefore for outbreaks to occur. When we put it in that sort of framework, we begin to realize that we need to understand the epidemiology of specific strains of S. aureus, we need to understand the periods of time when athletes are at highest risk – that’s probably when they’re newly acquiring strains that they’ve never seen before – and we need to take into account things that we can do that may be unrelated to staph, but may affect their risk for infection,” said lead author C. Buddy Creech, also of Vanderbilt University Medical Center.
For example, during the course of the current study, a cluster of infections “perfectly coincided” with the development of pandemic H1N1 influenza in a number of athletes, Dr. Creech said, adding that even though the two conditions are unrelated, this relationship between S. aureus and influenza shouldn’t be diminished.
“So even though it’s unrelated, all of the things we do to protect collegiate and professional athletes, like influenza vaccination and general health maintenance ... go a long way, and may have widespread implications,” said Dr. Creech.
He noted however, that although strategies of covering compromised skin and wounds are important for preventing the spread of disease, they are comparable to “putting our finger in the dike while we wait for other preventive strategies like vaccination or other more powerful strategies.”
Dr. Creech and Dr. Jimenez-Truque reported having no disclosures.
PHILADELPHIA – College athletes who play contact sports are more likely than are those who play noncontact sports to carry methicillin-resistant Staphylococcus aureus, putting them at higher risk for infection and for infecting others, according to findings from a 2-year observational study.
During the course of the study, MRSA colonization ranged from 8% to 31% in 224 students who played contact sports, including football, basketball, soccer, and lacrosse, compared with 0% to 23% in 153 students who participated in noncontact or limited-contact sports, including baseball, cross country, and golf. MRSA colonization in the general population ranges from 5% to 10%, Natalia Jimenez-Truque, Ph.D., of Vanderbilt University Medical Center, Nashville, Tenn., reported at an annual scientific meeting on infectious diseases.
Overall, contact sports athletes in this study had more than twice the risk of being colonized with MRSA (odds ratio 2.3), and tended to carry S. aureus for longer period of time (OR 3.0 for intermittent carriage, 2.39 for persistent carriage), compared with noncontact sports athletes. Also, contact sports athletes who were not carriers at baseline acquired S. aureus more quickly (hazard ratio 1.6), Dr. Jimenez-Truque said during a press conference at the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The findings have implications for advising young athletes about the importance of good hygiene practices, including covering any open wounds, washing hands frequently, showering as soon as possible after all practices and games, laundering gym clothes, bags, and towels promptly, and not sharing razors and other personal hygiene items, she said.
Athletes are known to have a higher risk of infection with S. aureus than the general population, but most studies have not assessed staphylococcal carriage or compared risk in contact and noncontact sports participants, Dr. Jimenez-Truque said. The lack of available information about the natural history of carriage across sports limits strategies for preventing infection in athletes, she noted.
For the current study – the first to observe both male and female college athletes who were not part of a MRSA outbreak – Dr. Jimenez-Truque and her colleagues followed varsity collegiate athletes from August 2008 to April 2010. Nasal and oropharyngeal swabs were collected at enrollment and monthly thereafter.
The increased rates of MRSA carriage and acquisition in contact sports athletes in this study may be associated with increased likelihood of having cuts, scrapes, turf burns, and skin-to-skin contact with other athletes, further underscoring the need for good hygiene, she said.
MRSA is a leading cause of both invasive and skin and soft tissue infections. Athletes are commonly affected, and up to 70% of those who develop infection require hospitalization and intravenous antibiotic treatment.
However, despite the “really significant number” of contact sports athletes who carry MRSA, the number who become infected is small, Dr. Jimenez-Truque said.
“What that tells us, we think, is that colonization with specific strains of staph in specific people at specific times is needed for infections to occur, and therefore for outbreaks to occur. When we put it in that sort of framework, we begin to realize that we need to understand the epidemiology of specific strains of S. aureus, we need to understand the periods of time when athletes are at highest risk – that’s probably when they’re newly acquiring strains that they’ve never seen before – and we need to take into account things that we can do that may be unrelated to staph, but may affect their risk for infection,” said lead author C. Buddy Creech, also of Vanderbilt University Medical Center.
For example, during the course of the current study, a cluster of infections “perfectly coincided” with the development of pandemic H1N1 influenza in a number of athletes, Dr. Creech said, adding that even though the two conditions are unrelated, this relationship between S. aureus and influenza shouldn’t be diminished.
“So even though it’s unrelated, all of the things we do to protect collegiate and professional athletes, like influenza vaccination and general health maintenance ... go a long way, and may have widespread implications,” said Dr. Creech.
He noted however, that although strategies of covering compromised skin and wounds are important for preventing the spread of disease, they are comparable to “putting our finger in the dike while we wait for other preventive strategies like vaccination or other more powerful strategies.”
Dr. Creech and Dr. Jimenez-Truque reported having no disclosures.
Key clinical point: Contact sports athletes should be advised about MRSA and good hygiene practices to reduce infection risk.
Major finding: MRSA colonization ranged from 8% to 31% in contact sport athletes, and 0% to 23% in noncontact athletes.
Data source: An observational study of 377 college athletes.
Disclosures: Dr. Creech and Dr. Jimenez-Truque reported having no disclosures.
Fidaxomicin appears safe, effective for CDAD in children
PHILADELPHIA – Fidaxomicin, an approved treatment for Clostridium difficile-associated diarrhea in adults, appears safe and effective in children, according to findings from an open-label study.
The overall clinical response rate was 92% in 38 children aged 11 months through 17 years who were treated with 32 mg/kg/day for 10 days. Although 74% of patients had at least one adverse event, most of the events were mild (45%) or moderate (21%) in severity and were the type of events that would be expected in children with moderate to severe underlying illness, Pam Sears, Ph.D. of Cubist Pharmaceuticals, San Diego, reported at an annual scientific meeting on infectious diseases.
The children included in this first pediatric study of fidaxomicin had diarrhea and tested positive for C. difficile toxin. Most had relatively complex medical histories; 24% had neoplasms and 79% had gastrointestinal disorders. They received either the commercial tablet formulation of fidaxomicin or an oral suspension.
Plasma concentrations were generally low, with a mean across the strata ranging from 8.87-16.6 ng/mL and metabolite OP-1118 levels that ranged from 27.5-130 ng/mL at 1-2 hours following the first dose. No age-related trends in concentrations were seen.
Fecal concentrations of fidaxomicin averaged 3,230 mcg/g with a trend toward higher concentrations in the youngest age group. This could be because samples from the youngest group were collected from diapers and could have become dehydrated, Dr. Sears explained the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Of the children in the study with a clinical response, 29% experienced a relapse, but all but one of these children had a history of recurrent C. difficile-related diarrhea.
Fidaxomicin is a first-in class narrow spectrum macrocyclic antibiotic drug approved in the United States and several other countries for the treatment of C. difficile-associated diarrhea in adults.
These findings demonstrate that the pharmacokinetic profile in children is similar to that in adults – with low plasma levels and high fecal concentrations, and suggest that clinical response in children is also excellent, Dr. Sears said.
This study was funded by Cubist Pharmaceuticals. Dr. Sears is an employee of Cubist Pharmaceuticals.
PHILADELPHIA – Fidaxomicin, an approved treatment for Clostridium difficile-associated diarrhea in adults, appears safe and effective in children, according to findings from an open-label study.
The overall clinical response rate was 92% in 38 children aged 11 months through 17 years who were treated with 32 mg/kg/day for 10 days. Although 74% of patients had at least one adverse event, most of the events were mild (45%) or moderate (21%) in severity and were the type of events that would be expected in children with moderate to severe underlying illness, Pam Sears, Ph.D. of Cubist Pharmaceuticals, San Diego, reported at an annual scientific meeting on infectious diseases.
The children included in this first pediatric study of fidaxomicin had diarrhea and tested positive for C. difficile toxin. Most had relatively complex medical histories; 24% had neoplasms and 79% had gastrointestinal disorders. They received either the commercial tablet formulation of fidaxomicin or an oral suspension.
Plasma concentrations were generally low, with a mean across the strata ranging from 8.87-16.6 ng/mL and metabolite OP-1118 levels that ranged from 27.5-130 ng/mL at 1-2 hours following the first dose. No age-related trends in concentrations were seen.
Fecal concentrations of fidaxomicin averaged 3,230 mcg/g with a trend toward higher concentrations in the youngest age group. This could be because samples from the youngest group were collected from diapers and could have become dehydrated, Dr. Sears explained the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Of the children in the study with a clinical response, 29% experienced a relapse, but all but one of these children had a history of recurrent C. difficile-related diarrhea.
Fidaxomicin is a first-in class narrow spectrum macrocyclic antibiotic drug approved in the United States and several other countries for the treatment of C. difficile-associated diarrhea in adults.
These findings demonstrate that the pharmacokinetic profile in children is similar to that in adults – with low plasma levels and high fecal concentrations, and suggest that clinical response in children is also excellent, Dr. Sears said.
This study was funded by Cubist Pharmaceuticals. Dr. Sears is an employee of Cubist Pharmaceuticals.
PHILADELPHIA – Fidaxomicin, an approved treatment for Clostridium difficile-associated diarrhea in adults, appears safe and effective in children, according to findings from an open-label study.
The overall clinical response rate was 92% in 38 children aged 11 months through 17 years who were treated with 32 mg/kg/day for 10 days. Although 74% of patients had at least one adverse event, most of the events were mild (45%) or moderate (21%) in severity and were the type of events that would be expected in children with moderate to severe underlying illness, Pam Sears, Ph.D. of Cubist Pharmaceuticals, San Diego, reported at an annual scientific meeting on infectious diseases.
The children included in this first pediatric study of fidaxomicin had diarrhea and tested positive for C. difficile toxin. Most had relatively complex medical histories; 24% had neoplasms and 79% had gastrointestinal disorders. They received either the commercial tablet formulation of fidaxomicin or an oral suspension.
Plasma concentrations were generally low, with a mean across the strata ranging from 8.87-16.6 ng/mL and metabolite OP-1118 levels that ranged from 27.5-130 ng/mL at 1-2 hours following the first dose. No age-related trends in concentrations were seen.
Fecal concentrations of fidaxomicin averaged 3,230 mcg/g with a trend toward higher concentrations in the youngest age group. This could be because samples from the youngest group were collected from diapers and could have become dehydrated, Dr. Sears explained the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Of the children in the study with a clinical response, 29% experienced a relapse, but all but one of these children had a history of recurrent C. difficile-related diarrhea.
Fidaxomicin is a first-in class narrow spectrum macrocyclic antibiotic drug approved in the United States and several other countries for the treatment of C. difficile-associated diarrhea in adults.
These findings demonstrate that the pharmacokinetic profile in children is similar to that in adults – with low plasma levels and high fecal concentrations, and suggest that clinical response in children is also excellent, Dr. Sears said.
This study was funded by Cubist Pharmaceuticals. Dr. Sears is an employee of Cubist Pharmaceuticals.
Key clinical point: Fidaxomicin appear to have excellent clinical efficacy in children with C. difficile-associated diarrhea and a pharmacokinetic profile similar to that seen in adults.
Major finding: The overall clinical response rate was 92%.
Data source: An open-label study in 38 children.
Disclosures: This study was funded by Cubist Pharmaceuticals. Dr. Sears is an employee of Cubist Pharmaceuticals.