User login
Royal family affliction or not, porphyria is treatable
European royal families may be enormously rich, but being a blueblood doesn’t always mean your blood is pristine. Queen Victoria’s DNA is famously believed to have silently bequeathed hemophilia to many of her descendants, including a great-grandson whose severe illness played a tragic role in spurring the Russian Revolution.
And that’s not all.
There’s plenty of skepticism about this theory, which seeks to explain the “madness” of King George III. But one thing is clear. If porphyria does indeed haunt the imperial bloodline that stretches to a new generation – the late Queen Elizabeth II’s great-grandchildren – any royal who’s afflicted going forward is likely to benefit mightily from modern treatment. While this disease may require lifelong vigilance, experts said in interviews that porphyria can often be controlled.
“If patients know they have the diagnosis, and they do the right things and avoid alcohol and risky drugs, most people will have few acute attacks,” said gastroenterologist Herbert Lloyd Bonkovsky, MD, of Wake Forest University, Winston-Salem, N.C., a leading porphyria specialist.
Heme infusions can also be helpful, he added, and the revolutionary new drug givosiran is available for those who suffer recurrent attacks. And “if all else fails, a successful liver transplant is curative” – as long as the transplanted liver doesn’t have porphyria, as happened in at least one case.
But, Dr. Bonkovsky cautioned, the diagnosis is often missed, in some cases for 15 years or more.
Diagnosing porphyria: Awareness and tests are crucial
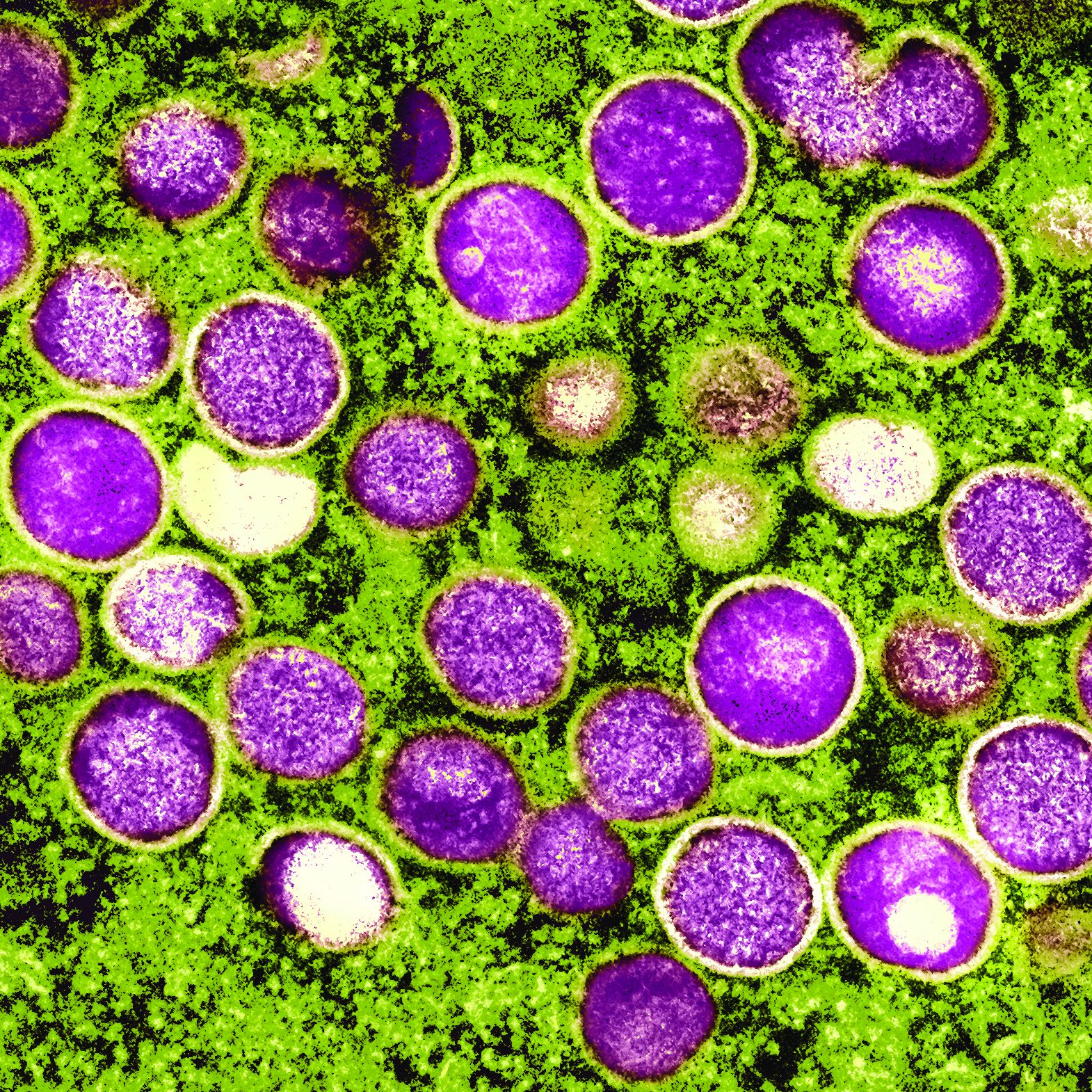
Porphyria is caused when porphyins – essential components of hemoglobin – build up in the body, disrupting systems such as the nerves, skin, and gut. The urine can turn purplish, hence the condition’s name. (Porphyrus is the Greek word for purple.)
According to hematologist Danielle Nance, MD, of Banner MD Anderson Cancer Center in Gilbert, Ariz., acute intermittent porphyria “should be suspected in persons who have recurrent severe attacks of abdominal pain requiring strong pain medication to control symptoms, and there is no obvious physical cause.”
In such cases, practitioners should send out blood and urine for porphobilinogen (PBG) and delta-aminolevulinic acid (dALA or Delta-ALA) testing, Dr. Nance said. “These are almost always elevated, even between attacks, in persons with diagnoses of acute intermittent porphyria. Other types of porphyria, such as erythropoietic porphyria, may require additional testing. Genetic testing should be offered when a patient is suspected of having porphyria, as this can speed the diagnosis.”
The typical patient is a woman from age 18 to 55, often a young woman with recurrent abdominal pain that may occur during the second half of the menstrual cycle, Wake Forest’s Dr. Bonkovsky said. Constipation is common.
“She keeps coming to the clinic or emergency department, and no one knows what’s going on. Eventually, she tends to undergo an appendectomy, often a cholecystectomy, or sometimes gynecologic procedures without cure of the disease. Only after this long and arduous road of misdiagnosis does someone think it’s porphyria and do the correct tests.”
Dr. Bonkovsky led a 2014 study of 108 subjects (81% female) with acute porphyrias and found that the average time to a correct diagnosis was a whopping 15 years. Pain in the abdomen was the most common symptom (74%), followed by nausea/vomiting (73%), weakness (63%), and constipation (60%).
While underdiagnosis is common, porphyrias can also be overdiagnosed. According to Dr. Bonkovsky, a mild increase in urinary porphyrins is often misdiagnosed as porphyria when it may be a sign of liver disease or alcohol use, instead.
Hematologist Kleber Y. Fertrin, MD, PhD, of the University of Washington, Seattle, emphasized the importance of ordering the correct tests. “Urinary porphyrins are often inappropriately ordered because of their name. They are not diagnostic for acute hepatic porphyrias and may be nonspecifically elevated. It is paramount to get the labs from a reliable lab test experienced at performing diagnostic testing for porphyrias and make sure the urine sample needed is correctly obtained and preserved.”
New drug can control attacks, but it’s costly
Treatment should begin right away if porphyria is suspected, without waiting for confirmation, Dr. Nance said. “There are porphyria experts in the U.S. and in many other countries who can help practitioners interpret symptoms and testing results if there is uncertainty or if treatment options are unclear.”
Patients are counseled to avoid attack triggers and eat healthy diets, she said. “Persons with porphyria are encouraged to have a plan for home management of an attack and when to come to the hospital for symptoms that don’t respond to home therapy,” she said.
Dr. Fertrin said “the typical treatment for an acute porphyric attack is the use of intravenous glucose, as well as intravenous hemin infusions, along with pain medications and fluids as needed. Some patients may need medication for high blood pressure. For patients with recurrent attacks, prophylactic doses of hemin can be used.”
Another option is givosiran (Givlaari), the biggest recent advance in porphyria treatment. It was approved by the Food and Drug Administration for acute hepatic porphyria in 2019. “It’s an inhibitory mRNA that shuts down heme synthesis by downregulating aminolevulinic acid synthase,” said hematologist Thomas DeLoughery, MD, of Oregon Health & Science University, Portland.
A 2020 study coauthored by Dr. Bonkovsky found that the drug reduced the mean annualized attack rate in acute intermittent porphyria by 74%, compared with placebo (P < .001). “Givosiran led to lower levels of urinary ALA and porphobilinogen, fewer days of hemin use, and better daily scores for pain than placebo,” the study authors write. “Key adverse events that were observed more frequently in the givosiran group were elevations in serum aminotransferase levels, changes in serum creatinine levels and the estimated glomerular filtration rate, and injection-site reactions.”
Dr. Bonkovsky said the drug is appropriate in patients whose disease is not controlled by lifestyle interventions regarding alcohol, smoking, and substance use. However, while givosiran is helpful, “it’s not a cure,” he said. “Probably about 40% of patients have found the side effects such as nausea, fatigue, headaches, and liver injury to be too adverse and have not decided to take it in the long term.”
In addition, the drug costs about $500,000 a year, and insurers may balk at the expense. Still, specialists say it can be a good option. “Before givosiran, the standard therapy was hematin, which decreased the activity of the ALA synthase,” Dr. DeLoughery said. “This is a nasty drug that can inflame the blood vessels and is not all that effective.”
The royal family may be able to breathe easy
There are other forms of porphyria besides the acute type. Cutaneous porphryias can cause blistering from sunlight exposure, and treatment is avoidance of such triggers, Dr. Fertin said. “For a rare form called erythropoietic protoporphyria, there is a subcutaneous implant with a medication called afamelanotide that increases tolerance to sunlight.”
There’s another type: variegate porphyria, which UpToDate says is “characterized by cutaneous blistering and/or acute neurovisceral attacks.” In 1966, a mother-and-son psychiatrist team theorized that Britain’s King George III suffered from this type of porphyria.
However, researchers have sharply criticized the porphyria theory with regard to that king, and a recent theory now suggests that he actually had bipolar disorder. Dr. Fertin is among the skeptics, noting the high prevalence of porphyria in affected families.
“Since King George III would now have a living number of relatives of about 900 people, we would expect around 180 cases of variegate porphyria to be connected to him, and those are nowhere to be found,” he said. “The idea that it could cause progressive psychosis and dementia is also a misconception. We do not find that porphyria is the underlying cause of psychiatric conditions on their own. Many atypical symptoms of the king were attributed to variegate porphyria, and the few more typical symptoms he reportedly had, of abdominal pain and urine discoloration, can be more easily explained by common diseases, such as kidney stones or gallstones. Finally, attempts at obtaining genetic or biochemical evidence of porphyria from relatives of King George III have been negative or inconclusive.”
That’s good news for the royals, who have many other concerns these days.
Dr. Nance discloses research study funding from Alnylam (maker of Givlaari) and payments to participate in educational activities for patients and practitioners. Dr. Bonkovsky discloses relationships with drugmakers who have developed products to treat porphyria including Alnylam, Mitsubishi Tanabe, Recordati, and Disc. Dr. Fertin is subinvestigator in a clinical trial for a novel treatment for erythropoietic protoporphyria sponsored by Mitsubishi Tanabe. Dr. DeLoughery reports no disclosures.
European royal families may be enormously rich, but being a blueblood doesn’t always mean your blood is pristine. Queen Victoria’s DNA is famously believed to have silently bequeathed hemophilia to many of her descendants, including a great-grandson whose severe illness played a tragic role in spurring the Russian Revolution.
And that’s not all.
There’s plenty of skepticism about this theory, which seeks to explain the “madness” of King George III. But one thing is clear. If porphyria does indeed haunt the imperial bloodline that stretches to a new generation – the late Queen Elizabeth II’s great-grandchildren – any royal who’s afflicted going forward is likely to benefit mightily from modern treatment. While this disease may require lifelong vigilance, experts said in interviews that porphyria can often be controlled.
“If patients know they have the diagnosis, and they do the right things and avoid alcohol and risky drugs, most people will have few acute attacks,” said gastroenterologist Herbert Lloyd Bonkovsky, MD, of Wake Forest University, Winston-Salem, N.C., a leading porphyria specialist.
Heme infusions can also be helpful, he added, and the revolutionary new drug givosiran is available for those who suffer recurrent attacks. And “if all else fails, a successful liver transplant is curative” – as long as the transplanted liver doesn’t have porphyria, as happened in at least one case.
But, Dr. Bonkovsky cautioned, the diagnosis is often missed, in some cases for 15 years or more.
Diagnosing porphyria: Awareness and tests are crucial
Porphyria is caused when porphyins – essential components of hemoglobin – build up in the body, disrupting systems such as the nerves, skin, and gut. The urine can turn purplish, hence the condition’s name. (Porphyrus is the Greek word for purple.)
According to hematologist Danielle Nance, MD, of Banner MD Anderson Cancer Center in Gilbert, Ariz., acute intermittent porphyria “should be suspected in persons who have recurrent severe attacks of abdominal pain requiring strong pain medication to control symptoms, and there is no obvious physical cause.”
In such cases, practitioners should send out blood and urine for porphobilinogen (PBG) and delta-aminolevulinic acid (dALA or Delta-ALA) testing, Dr. Nance said. “These are almost always elevated, even between attacks, in persons with diagnoses of acute intermittent porphyria. Other types of porphyria, such as erythropoietic porphyria, may require additional testing. Genetic testing should be offered when a patient is suspected of having porphyria, as this can speed the diagnosis.”
The typical patient is a woman from age 18 to 55, often a young woman with recurrent abdominal pain that may occur during the second half of the menstrual cycle, Wake Forest’s Dr. Bonkovsky said. Constipation is common.
“She keeps coming to the clinic or emergency department, and no one knows what’s going on. Eventually, she tends to undergo an appendectomy, often a cholecystectomy, or sometimes gynecologic procedures without cure of the disease. Only after this long and arduous road of misdiagnosis does someone think it’s porphyria and do the correct tests.”
Dr. Bonkovsky led a 2014 study of 108 subjects (81% female) with acute porphyrias and found that the average time to a correct diagnosis was a whopping 15 years. Pain in the abdomen was the most common symptom (74%), followed by nausea/vomiting (73%), weakness (63%), and constipation (60%).
While underdiagnosis is common, porphyrias can also be overdiagnosed. According to Dr. Bonkovsky, a mild increase in urinary porphyrins is often misdiagnosed as porphyria when it may be a sign of liver disease or alcohol use, instead.
Hematologist Kleber Y. Fertrin, MD, PhD, of the University of Washington, Seattle, emphasized the importance of ordering the correct tests. “Urinary porphyrins are often inappropriately ordered because of their name. They are not diagnostic for acute hepatic porphyrias and may be nonspecifically elevated. It is paramount to get the labs from a reliable lab test experienced at performing diagnostic testing for porphyrias and make sure the urine sample needed is correctly obtained and preserved.”
New drug can control attacks, but it’s costly
Treatment should begin right away if porphyria is suspected, without waiting for confirmation, Dr. Nance said. “There are porphyria experts in the U.S. and in many other countries who can help practitioners interpret symptoms and testing results if there is uncertainty or if treatment options are unclear.”
Patients are counseled to avoid attack triggers and eat healthy diets, she said. “Persons with porphyria are encouraged to have a plan for home management of an attack and when to come to the hospital for symptoms that don’t respond to home therapy,” she said.
Dr. Fertrin said “the typical treatment for an acute porphyric attack is the use of intravenous glucose, as well as intravenous hemin infusions, along with pain medications and fluids as needed. Some patients may need medication for high blood pressure. For patients with recurrent attacks, prophylactic doses of hemin can be used.”
Another option is givosiran (Givlaari), the biggest recent advance in porphyria treatment. It was approved by the Food and Drug Administration for acute hepatic porphyria in 2019. “It’s an inhibitory mRNA that shuts down heme synthesis by downregulating aminolevulinic acid synthase,” said hematologist Thomas DeLoughery, MD, of Oregon Health & Science University, Portland.
A 2020 study coauthored by Dr. Bonkovsky found that the drug reduced the mean annualized attack rate in acute intermittent porphyria by 74%, compared with placebo (P < .001). “Givosiran led to lower levels of urinary ALA and porphobilinogen, fewer days of hemin use, and better daily scores for pain than placebo,” the study authors write. “Key adverse events that were observed more frequently in the givosiran group were elevations in serum aminotransferase levels, changes in serum creatinine levels and the estimated glomerular filtration rate, and injection-site reactions.”
Dr. Bonkovsky said the drug is appropriate in patients whose disease is not controlled by lifestyle interventions regarding alcohol, smoking, and substance use. However, while givosiran is helpful, “it’s not a cure,” he said. “Probably about 40% of patients have found the side effects such as nausea, fatigue, headaches, and liver injury to be too adverse and have not decided to take it in the long term.”
In addition, the drug costs about $500,000 a year, and insurers may balk at the expense. Still, specialists say it can be a good option. “Before givosiran, the standard therapy was hematin, which decreased the activity of the ALA synthase,” Dr. DeLoughery said. “This is a nasty drug that can inflame the blood vessels and is not all that effective.”
The royal family may be able to breathe easy
There are other forms of porphyria besides the acute type. Cutaneous porphryias can cause blistering from sunlight exposure, and treatment is avoidance of such triggers, Dr. Fertin said. “For a rare form called erythropoietic protoporphyria, there is a subcutaneous implant with a medication called afamelanotide that increases tolerance to sunlight.”
There’s another type: variegate porphyria, which UpToDate says is “characterized by cutaneous blistering and/or acute neurovisceral attacks.” In 1966, a mother-and-son psychiatrist team theorized that Britain’s King George III suffered from this type of porphyria.
However, researchers have sharply criticized the porphyria theory with regard to that king, and a recent theory now suggests that he actually had bipolar disorder. Dr. Fertin is among the skeptics, noting the high prevalence of porphyria in affected families.
“Since King George III would now have a living number of relatives of about 900 people, we would expect around 180 cases of variegate porphyria to be connected to him, and those are nowhere to be found,” he said. “The idea that it could cause progressive psychosis and dementia is also a misconception. We do not find that porphyria is the underlying cause of psychiatric conditions on their own. Many atypical symptoms of the king were attributed to variegate porphyria, and the few more typical symptoms he reportedly had, of abdominal pain and urine discoloration, can be more easily explained by common diseases, such as kidney stones or gallstones. Finally, attempts at obtaining genetic or biochemical evidence of porphyria from relatives of King George III have been negative or inconclusive.”
That’s good news for the royals, who have many other concerns these days.
Dr. Nance discloses research study funding from Alnylam (maker of Givlaari) and payments to participate in educational activities for patients and practitioners. Dr. Bonkovsky discloses relationships with drugmakers who have developed products to treat porphyria including Alnylam, Mitsubishi Tanabe, Recordati, and Disc. Dr. Fertin is subinvestigator in a clinical trial for a novel treatment for erythropoietic protoporphyria sponsored by Mitsubishi Tanabe. Dr. DeLoughery reports no disclosures.
European royal families may be enormously rich, but being a blueblood doesn’t always mean your blood is pristine. Queen Victoria’s DNA is famously believed to have silently bequeathed hemophilia to many of her descendants, including a great-grandson whose severe illness played a tragic role in spurring the Russian Revolution.
And that’s not all.
There’s plenty of skepticism about this theory, which seeks to explain the “madness” of King George III. But one thing is clear. If porphyria does indeed haunt the imperial bloodline that stretches to a new generation – the late Queen Elizabeth II’s great-grandchildren – any royal who’s afflicted going forward is likely to benefit mightily from modern treatment. While this disease may require lifelong vigilance, experts said in interviews that porphyria can often be controlled.
“If patients know they have the diagnosis, and they do the right things and avoid alcohol and risky drugs, most people will have few acute attacks,” said gastroenterologist Herbert Lloyd Bonkovsky, MD, of Wake Forest University, Winston-Salem, N.C., a leading porphyria specialist.
Heme infusions can also be helpful, he added, and the revolutionary new drug givosiran is available for those who suffer recurrent attacks. And “if all else fails, a successful liver transplant is curative” – as long as the transplanted liver doesn’t have porphyria, as happened in at least one case.
But, Dr. Bonkovsky cautioned, the diagnosis is often missed, in some cases for 15 years or more.
Diagnosing porphyria: Awareness and tests are crucial
Porphyria is caused when porphyins – essential components of hemoglobin – build up in the body, disrupting systems such as the nerves, skin, and gut. The urine can turn purplish, hence the condition’s name. (Porphyrus is the Greek word for purple.)
According to hematologist Danielle Nance, MD, of Banner MD Anderson Cancer Center in Gilbert, Ariz., acute intermittent porphyria “should be suspected in persons who have recurrent severe attacks of abdominal pain requiring strong pain medication to control symptoms, and there is no obvious physical cause.”
In such cases, practitioners should send out blood and urine for porphobilinogen (PBG) and delta-aminolevulinic acid (dALA or Delta-ALA) testing, Dr. Nance said. “These are almost always elevated, even between attacks, in persons with diagnoses of acute intermittent porphyria. Other types of porphyria, such as erythropoietic porphyria, may require additional testing. Genetic testing should be offered when a patient is suspected of having porphyria, as this can speed the diagnosis.”
The typical patient is a woman from age 18 to 55, often a young woman with recurrent abdominal pain that may occur during the second half of the menstrual cycle, Wake Forest’s Dr. Bonkovsky said. Constipation is common.
“She keeps coming to the clinic or emergency department, and no one knows what’s going on. Eventually, she tends to undergo an appendectomy, often a cholecystectomy, or sometimes gynecologic procedures without cure of the disease. Only after this long and arduous road of misdiagnosis does someone think it’s porphyria and do the correct tests.”
Dr. Bonkovsky led a 2014 study of 108 subjects (81% female) with acute porphyrias and found that the average time to a correct diagnosis was a whopping 15 years. Pain in the abdomen was the most common symptom (74%), followed by nausea/vomiting (73%), weakness (63%), and constipation (60%).
While underdiagnosis is common, porphyrias can also be overdiagnosed. According to Dr. Bonkovsky, a mild increase in urinary porphyrins is often misdiagnosed as porphyria when it may be a sign of liver disease or alcohol use, instead.
Hematologist Kleber Y. Fertrin, MD, PhD, of the University of Washington, Seattle, emphasized the importance of ordering the correct tests. “Urinary porphyrins are often inappropriately ordered because of their name. They are not diagnostic for acute hepatic porphyrias and may be nonspecifically elevated. It is paramount to get the labs from a reliable lab test experienced at performing diagnostic testing for porphyrias and make sure the urine sample needed is correctly obtained and preserved.”
New drug can control attacks, but it’s costly
Treatment should begin right away if porphyria is suspected, without waiting for confirmation, Dr. Nance said. “There are porphyria experts in the U.S. and in many other countries who can help practitioners interpret symptoms and testing results if there is uncertainty or if treatment options are unclear.”
Patients are counseled to avoid attack triggers and eat healthy diets, she said. “Persons with porphyria are encouraged to have a plan for home management of an attack and when to come to the hospital for symptoms that don’t respond to home therapy,” she said.
Dr. Fertrin said “the typical treatment for an acute porphyric attack is the use of intravenous glucose, as well as intravenous hemin infusions, along with pain medications and fluids as needed. Some patients may need medication for high blood pressure. For patients with recurrent attacks, prophylactic doses of hemin can be used.”
Another option is givosiran (Givlaari), the biggest recent advance in porphyria treatment. It was approved by the Food and Drug Administration for acute hepatic porphyria in 2019. “It’s an inhibitory mRNA that shuts down heme synthesis by downregulating aminolevulinic acid synthase,” said hematologist Thomas DeLoughery, MD, of Oregon Health & Science University, Portland.
A 2020 study coauthored by Dr. Bonkovsky found that the drug reduced the mean annualized attack rate in acute intermittent porphyria by 74%, compared with placebo (P < .001). “Givosiran led to lower levels of urinary ALA and porphobilinogen, fewer days of hemin use, and better daily scores for pain than placebo,” the study authors write. “Key adverse events that were observed more frequently in the givosiran group were elevations in serum aminotransferase levels, changes in serum creatinine levels and the estimated glomerular filtration rate, and injection-site reactions.”
Dr. Bonkovsky said the drug is appropriate in patients whose disease is not controlled by lifestyle interventions regarding alcohol, smoking, and substance use. However, while givosiran is helpful, “it’s not a cure,” he said. “Probably about 40% of patients have found the side effects such as nausea, fatigue, headaches, and liver injury to be too adverse and have not decided to take it in the long term.”
In addition, the drug costs about $500,000 a year, and insurers may balk at the expense. Still, specialists say it can be a good option. “Before givosiran, the standard therapy was hematin, which decreased the activity of the ALA synthase,” Dr. DeLoughery said. “This is a nasty drug that can inflame the blood vessels and is not all that effective.”
The royal family may be able to breathe easy
There are other forms of porphyria besides the acute type. Cutaneous porphryias can cause blistering from sunlight exposure, and treatment is avoidance of such triggers, Dr. Fertin said. “For a rare form called erythropoietic protoporphyria, there is a subcutaneous implant with a medication called afamelanotide that increases tolerance to sunlight.”
There’s another type: variegate porphyria, which UpToDate says is “characterized by cutaneous blistering and/or acute neurovisceral attacks.” In 1966, a mother-and-son psychiatrist team theorized that Britain’s King George III suffered from this type of porphyria.
However, researchers have sharply criticized the porphyria theory with regard to that king, and a recent theory now suggests that he actually had bipolar disorder. Dr. Fertin is among the skeptics, noting the high prevalence of porphyria in affected families.
“Since King George III would now have a living number of relatives of about 900 people, we would expect around 180 cases of variegate porphyria to be connected to him, and those are nowhere to be found,” he said. “The idea that it could cause progressive psychosis and dementia is also a misconception. We do not find that porphyria is the underlying cause of psychiatric conditions on their own. Many atypical symptoms of the king were attributed to variegate porphyria, and the few more typical symptoms he reportedly had, of abdominal pain and urine discoloration, can be more easily explained by common diseases, such as kidney stones or gallstones. Finally, attempts at obtaining genetic or biochemical evidence of porphyria from relatives of King George III have been negative or inconclusive.”
That’s good news for the royals, who have many other concerns these days.
Dr. Nance discloses research study funding from Alnylam (maker of Givlaari) and payments to participate in educational activities for patients and practitioners. Dr. Bonkovsky discloses relationships with drugmakers who have developed products to treat porphyria including Alnylam, Mitsubishi Tanabe, Recordati, and Disc. Dr. Fertin is subinvestigator in a clinical trial for a novel treatment for erythropoietic protoporphyria sponsored by Mitsubishi Tanabe. Dr. DeLoughery reports no disclosures.
Biden’s Cancer Moonshot turns its focus to early-detection blood tests
There’s big buzz about the hot prospects for blood tests designed to detect multiple kinds of cancer. President Biden highlighted them in a speech about the Cancer Moonshot program on Sept. 12, just a day after study results touted an experimental test’s ability to detect dozens of kinds of cancer. Meanwhile, the federal government is heralding an upcoming trial that will eventually enroll as many as 225,000 subjects.
There are plenty of reasons to be cautious, however. And if these tests become standard, the oncology field will need to figure out how to navigate a thicket of new challenges.
“Our friends in internal medicine and primary care will be looking to us for guidance. We need to make sure that we’re coming at this without too much optimism before we really have the data,” said Jyoti D. Patel, MD, medical director of thoracic oncology and assistant director for clinical research at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago.
Dr. Patel is a member of the communications workgroup of the Multicancer Early Detection Consortium, a nonprofit, public-private organization that’s providing insight and guidance into the development of screening tests. The consortium published a position paper earlier this year.
According to Dr. Patel, early cancer screening today can detect only five types of cancer: prostate, breast, lung, cervical, and colon. The Cancer Moonshot program has prioritized research into greatly expanding this number. President Biden referred to this goal in his Sept. 12 speech: “Imagine a simple blood test during an annual physical that could detect cancer early, where the chances of a cure are best.”
Biden said the National Cancer Institute is launching a major trial as part of the Cancer Moonshot program. The Vanguard Study on Multi-Cancer Detection plans to enlist 25,000 healthy women and men between 45 and 70 years old in 2024, then later enroll as many as 225,000 people.
Meanwhile, researchers reported on Sept. 11 that the Galleri multicancer detection blood test found positive cancer signals in 1.4% of 6,621 healthy subjects, and cancer was ultimately confirmed in 38% of those in that group. Nineteen solid tumors and 17 hematologic cancers were diagnosed; 26 of these were cancer types that don’t have routine screening available.
The Galleri test is widely available in the United States, although the $950 cost is not covered by insurance.
While the data is exciting, the high false-positive rate is worrisome, Dr. Patel said. “Are there ways that we can further define that by cancer-risk assessment or by having better captures in our technology that reflect RNA methylation or epigenetic changes that may lead to susceptibility to cancers?”
Additional research is essential
Ernest Hawk, MD, vice president and division head of cancer prevention and population sciences at the University of Texas MD Anderson Cancer Center, Houston, said it’s “absolutely essential” that research into screening tests clearly demonstrates improved patient outcomes over time.
“We need to have much longer follow-up of all participants – whether the screening results are positive or negative – and mitigate the potential risks of such testing,” said Dr. Hawk, who’s worked with the Multicancer Early Detection Consortium.
On another front, Northwestern University’s Dr. Patel highlighted that while easy-to-access cancer screening could create tremendous opportunities to treat early cancer and shrink disparities in care, it may produce “an onslaught of patients with early-stage disease. Do we have the workforce to help us?” Also, she said, “if we find a patient with early-stage disease, how are we going to risk-stratify their follow-up and adjuvant therapy? Are there ways to prognosticate with more granularity than we do now?”
What’s next? “Multicancer early-detection tests could truly revolutionize cancer care if they work as we hope they will, but only time, extensive participation in research, and hard work will prove whether that is true or not,” said MD Anderson’s Dr. Hawk. “I anticipate that we’ll have reasonable answers within the next decade, given the pace of existing company-sponsored research and NCI’s planned involvement in testing various technologies available.”
For her part, Dr. Patel said oncologists should be aware that multicancer screening tests are available and be ready to address questions about them. “Think about how you can advise patients in the absence of data,” she said.
Dr. Patel and Dr. Hawk have no relevant disclosures.
There’s big buzz about the hot prospects for blood tests designed to detect multiple kinds of cancer. President Biden highlighted them in a speech about the Cancer Moonshot program on Sept. 12, just a day after study results touted an experimental test’s ability to detect dozens of kinds of cancer. Meanwhile, the federal government is heralding an upcoming trial that will eventually enroll as many as 225,000 subjects.
There are plenty of reasons to be cautious, however. And if these tests become standard, the oncology field will need to figure out how to navigate a thicket of new challenges.
“Our friends in internal medicine and primary care will be looking to us for guidance. We need to make sure that we’re coming at this without too much optimism before we really have the data,” said Jyoti D. Patel, MD, medical director of thoracic oncology and assistant director for clinical research at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago.
Dr. Patel is a member of the communications workgroup of the Multicancer Early Detection Consortium, a nonprofit, public-private organization that’s providing insight and guidance into the development of screening tests. The consortium published a position paper earlier this year.
According to Dr. Patel, early cancer screening today can detect only five types of cancer: prostate, breast, lung, cervical, and colon. The Cancer Moonshot program has prioritized research into greatly expanding this number. President Biden referred to this goal in his Sept. 12 speech: “Imagine a simple blood test during an annual physical that could detect cancer early, where the chances of a cure are best.”
Biden said the National Cancer Institute is launching a major trial as part of the Cancer Moonshot program. The Vanguard Study on Multi-Cancer Detection plans to enlist 25,000 healthy women and men between 45 and 70 years old in 2024, then later enroll as many as 225,000 people.
Meanwhile, researchers reported on Sept. 11 that the Galleri multicancer detection blood test found positive cancer signals in 1.4% of 6,621 healthy subjects, and cancer was ultimately confirmed in 38% of those in that group. Nineteen solid tumors and 17 hematologic cancers were diagnosed; 26 of these were cancer types that don’t have routine screening available.
The Galleri test is widely available in the United States, although the $950 cost is not covered by insurance.
While the data is exciting, the high false-positive rate is worrisome, Dr. Patel said. “Are there ways that we can further define that by cancer-risk assessment or by having better captures in our technology that reflect RNA methylation or epigenetic changes that may lead to susceptibility to cancers?”
Additional research is essential
Ernest Hawk, MD, vice president and division head of cancer prevention and population sciences at the University of Texas MD Anderson Cancer Center, Houston, said it’s “absolutely essential” that research into screening tests clearly demonstrates improved patient outcomes over time.
“We need to have much longer follow-up of all participants – whether the screening results are positive or negative – and mitigate the potential risks of such testing,” said Dr. Hawk, who’s worked with the Multicancer Early Detection Consortium.
On another front, Northwestern University’s Dr. Patel highlighted that while easy-to-access cancer screening could create tremendous opportunities to treat early cancer and shrink disparities in care, it may produce “an onslaught of patients with early-stage disease. Do we have the workforce to help us?” Also, she said, “if we find a patient with early-stage disease, how are we going to risk-stratify their follow-up and adjuvant therapy? Are there ways to prognosticate with more granularity than we do now?”
What’s next? “Multicancer early-detection tests could truly revolutionize cancer care if they work as we hope they will, but only time, extensive participation in research, and hard work will prove whether that is true or not,” said MD Anderson’s Dr. Hawk. “I anticipate that we’ll have reasonable answers within the next decade, given the pace of existing company-sponsored research and NCI’s planned involvement in testing various technologies available.”
For her part, Dr. Patel said oncologists should be aware that multicancer screening tests are available and be ready to address questions about them. “Think about how you can advise patients in the absence of data,” she said.
Dr. Patel and Dr. Hawk have no relevant disclosures.
There’s big buzz about the hot prospects for blood tests designed to detect multiple kinds of cancer. President Biden highlighted them in a speech about the Cancer Moonshot program on Sept. 12, just a day after study results touted an experimental test’s ability to detect dozens of kinds of cancer. Meanwhile, the federal government is heralding an upcoming trial that will eventually enroll as many as 225,000 subjects.
There are plenty of reasons to be cautious, however. And if these tests become standard, the oncology field will need to figure out how to navigate a thicket of new challenges.
“Our friends in internal medicine and primary care will be looking to us for guidance. We need to make sure that we’re coming at this without too much optimism before we really have the data,” said Jyoti D. Patel, MD, medical director of thoracic oncology and assistant director for clinical research at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University, Chicago.
Dr. Patel is a member of the communications workgroup of the Multicancer Early Detection Consortium, a nonprofit, public-private organization that’s providing insight and guidance into the development of screening tests. The consortium published a position paper earlier this year.
According to Dr. Patel, early cancer screening today can detect only five types of cancer: prostate, breast, lung, cervical, and colon. The Cancer Moonshot program has prioritized research into greatly expanding this number. President Biden referred to this goal in his Sept. 12 speech: “Imagine a simple blood test during an annual physical that could detect cancer early, where the chances of a cure are best.”
Biden said the National Cancer Institute is launching a major trial as part of the Cancer Moonshot program. The Vanguard Study on Multi-Cancer Detection plans to enlist 25,000 healthy women and men between 45 and 70 years old in 2024, then later enroll as many as 225,000 people.
Meanwhile, researchers reported on Sept. 11 that the Galleri multicancer detection blood test found positive cancer signals in 1.4% of 6,621 healthy subjects, and cancer was ultimately confirmed in 38% of those in that group. Nineteen solid tumors and 17 hematologic cancers were diagnosed; 26 of these were cancer types that don’t have routine screening available.
The Galleri test is widely available in the United States, although the $950 cost is not covered by insurance.
While the data is exciting, the high false-positive rate is worrisome, Dr. Patel said. “Are there ways that we can further define that by cancer-risk assessment or by having better captures in our technology that reflect RNA methylation or epigenetic changes that may lead to susceptibility to cancers?”
Additional research is essential
Ernest Hawk, MD, vice president and division head of cancer prevention and population sciences at the University of Texas MD Anderson Cancer Center, Houston, said it’s “absolutely essential” that research into screening tests clearly demonstrates improved patient outcomes over time.
“We need to have much longer follow-up of all participants – whether the screening results are positive or negative – and mitigate the potential risks of such testing,” said Dr. Hawk, who’s worked with the Multicancer Early Detection Consortium.
On another front, Northwestern University’s Dr. Patel highlighted that while easy-to-access cancer screening could create tremendous opportunities to treat early cancer and shrink disparities in care, it may produce “an onslaught of patients with early-stage disease. Do we have the workforce to help us?” Also, she said, “if we find a patient with early-stage disease, how are we going to risk-stratify their follow-up and adjuvant therapy? Are there ways to prognosticate with more granularity than we do now?”
What’s next? “Multicancer early-detection tests could truly revolutionize cancer care if they work as we hope they will, but only time, extensive participation in research, and hard work will prove whether that is true or not,” said MD Anderson’s Dr. Hawk. “I anticipate that we’ll have reasonable answers within the next decade, given the pace of existing company-sponsored research and NCI’s planned involvement in testing various technologies available.”
For her part, Dr. Patel said oncologists should be aware that multicancer screening tests are available and be ready to address questions about them. “Think about how you can advise patients in the absence of data,” she said.
Dr. Patel and Dr. Hawk have no relevant disclosures.
VA Launches Virtual Tumor Board
SAN DIEGO – The US Department of Veterans Affairs (VA) TeleOncology program has rolled out a virtual tumor board that brings medical professionals together to offer insight and guidance about challenging hematology cases. Over the past 6 months the board has held 10 sessions and reviewed about 20 cases. A small survey found that participants think the meetings are beneficial.
“Virtual tumor boards help to connect experts across the country to leverage the expertise within the VA,” he-matologist/oncologist Thomas Rodgers, MD, of the Duke Cancer Institute and Durham Veterans Affairs Medical Center, told Federal Practitioner in an interview. He is the lead author of a poster about the program that was pre-sented here at the annual meeting of the Association of VA Hematology/Oncology (AVAHO).
As Dr. Rodgers noted, tumor boards are already in place at some VA centers. However, “they are not available at every VA and often are not set up to cover every cancer type.”
The VA National TeleOncology program created the virtual tumor board program as part of its mission to ex-tend hematology/oncology services across the system. “Cancer care has become increasingly complex. Beyond ad-vancing therapeutics, patient care often involves multiple specialties and medical disciplines,” Dr. Rodgers said. “A tumor board offers a forum for these specialists to communicate with each other in real time, not only to help estab-lish the correct diagnosis and stage of cancer but also to form a consensus on the most fitting treatment option. Think of it as getting all of the people involved in a person’s care in the same room.”
Currently, he said, the virtual tumor boards cover patients with malignant hematology diagnoses such as leuke-mia, multiple myeloma, and lymphomas. “We welcome submissions. If a provider is interested in submitting a case, they can email us and will be provided with a short intake form. Once submitted, we will collect necessary imaging and pathology for review. The provider will then present the patient case on the day of the tumor board.”
Typically, more than 30 medical professionals participate in the virtual tumor boards, Dr. Rodgers said, repre-senting medical oncology/hematology, pathology, radiology, palliative care, pharmacy, social work, and die-tary/nutrition.
According to the poster presented at AVAHO, 9 participants responded to a survey after 4 tumor board sessions. All found the boards to be beneficial or somewhat beneficial, and 55% reported that they were “highly applicable” to their practice.
Pathologist Claudio A. Mosse, MD, PhD, of Vanderbilt University Medical Center and VA Tennessee Valley Healthcare System, praised the virtual tumor board program. “It’s been incredibly useful from my end as a pathologist as it shows me which diagnoses are most challenging for my colleagues,” Dr. Mosse said in an inter-view. “Reviewing and then presenting these challenging cases forces me to go into the published literature to come to a unitary diagnosis based on the patient history, radiology, various laboratory tests, and the biopsy I was asked to review.”
He added that “as a pathologist, I learn so much from the hematologists as they discuss the possible therapeutic options, and that strengthens my ability as a pathologist because I have to understand how one diagnosis versus an-other affects their therapeutic decision tree.”
What’s next for the virtual tumor board program? The next step is to expand to solid tumors, said VA Pittsburgh Healthcare System hematologist/oncologist Vida Almario Passero, MD, MBA, chief medical officer of National TeleOncology, in an interview.
No disclosures were reported.
SAN DIEGO – The US Department of Veterans Affairs (VA) TeleOncology program has rolled out a virtual tumor board that brings medical professionals together to offer insight and guidance about challenging hematology cases. Over the past 6 months the board has held 10 sessions and reviewed about 20 cases. A small survey found that participants think the meetings are beneficial.
“Virtual tumor boards help to connect experts across the country to leverage the expertise within the VA,” he-matologist/oncologist Thomas Rodgers, MD, of the Duke Cancer Institute and Durham Veterans Affairs Medical Center, told Federal Practitioner in an interview. He is the lead author of a poster about the program that was pre-sented here at the annual meeting of the Association of VA Hematology/Oncology (AVAHO).
As Dr. Rodgers noted, tumor boards are already in place at some VA centers. However, “they are not available at every VA and often are not set up to cover every cancer type.”
The VA National TeleOncology program created the virtual tumor board program as part of its mission to ex-tend hematology/oncology services across the system. “Cancer care has become increasingly complex. Beyond ad-vancing therapeutics, patient care often involves multiple specialties and medical disciplines,” Dr. Rodgers said. “A tumor board offers a forum for these specialists to communicate with each other in real time, not only to help estab-lish the correct diagnosis and stage of cancer but also to form a consensus on the most fitting treatment option. Think of it as getting all of the people involved in a person’s care in the same room.”
Currently, he said, the virtual tumor boards cover patients with malignant hematology diagnoses such as leuke-mia, multiple myeloma, and lymphomas. “We welcome submissions. If a provider is interested in submitting a case, they can email us and will be provided with a short intake form. Once submitted, we will collect necessary imaging and pathology for review. The provider will then present the patient case on the day of the tumor board.”
Typically, more than 30 medical professionals participate in the virtual tumor boards, Dr. Rodgers said, repre-senting medical oncology/hematology, pathology, radiology, palliative care, pharmacy, social work, and die-tary/nutrition.
According to the poster presented at AVAHO, 9 participants responded to a survey after 4 tumor board sessions. All found the boards to be beneficial or somewhat beneficial, and 55% reported that they were “highly applicable” to their practice.
Pathologist Claudio A. Mosse, MD, PhD, of Vanderbilt University Medical Center and VA Tennessee Valley Healthcare System, praised the virtual tumor board program. “It’s been incredibly useful from my end as a pathologist as it shows me which diagnoses are most challenging for my colleagues,” Dr. Mosse said in an inter-view. “Reviewing and then presenting these challenging cases forces me to go into the published literature to come to a unitary diagnosis based on the patient history, radiology, various laboratory tests, and the biopsy I was asked to review.”
He added that “as a pathologist, I learn so much from the hematologists as they discuss the possible therapeutic options, and that strengthens my ability as a pathologist because I have to understand how one diagnosis versus an-other affects their therapeutic decision tree.”
What’s next for the virtual tumor board program? The next step is to expand to solid tumors, said VA Pittsburgh Healthcare System hematologist/oncologist Vida Almario Passero, MD, MBA, chief medical officer of National TeleOncology, in an interview.
No disclosures were reported.
SAN DIEGO – The US Department of Veterans Affairs (VA) TeleOncology program has rolled out a virtual tumor board that brings medical professionals together to offer insight and guidance about challenging hematology cases. Over the past 6 months the board has held 10 sessions and reviewed about 20 cases. A small survey found that participants think the meetings are beneficial.
“Virtual tumor boards help to connect experts across the country to leverage the expertise within the VA,” he-matologist/oncologist Thomas Rodgers, MD, of the Duke Cancer Institute and Durham Veterans Affairs Medical Center, told Federal Practitioner in an interview. He is the lead author of a poster about the program that was pre-sented here at the annual meeting of the Association of VA Hematology/Oncology (AVAHO).
As Dr. Rodgers noted, tumor boards are already in place at some VA centers. However, “they are not available at every VA and often are not set up to cover every cancer type.”
The VA National TeleOncology program created the virtual tumor board program as part of its mission to ex-tend hematology/oncology services across the system. “Cancer care has become increasingly complex. Beyond ad-vancing therapeutics, patient care often involves multiple specialties and medical disciplines,” Dr. Rodgers said. “A tumor board offers a forum for these specialists to communicate with each other in real time, not only to help estab-lish the correct diagnosis and stage of cancer but also to form a consensus on the most fitting treatment option. Think of it as getting all of the people involved in a person’s care in the same room.”
Currently, he said, the virtual tumor boards cover patients with malignant hematology diagnoses such as leuke-mia, multiple myeloma, and lymphomas. “We welcome submissions. If a provider is interested in submitting a case, they can email us and will be provided with a short intake form. Once submitted, we will collect necessary imaging and pathology for review. The provider will then present the patient case on the day of the tumor board.”
Typically, more than 30 medical professionals participate in the virtual tumor boards, Dr. Rodgers said, repre-senting medical oncology/hematology, pathology, radiology, palliative care, pharmacy, social work, and die-tary/nutrition.
According to the poster presented at AVAHO, 9 participants responded to a survey after 4 tumor board sessions. All found the boards to be beneficial or somewhat beneficial, and 55% reported that they were “highly applicable” to their practice.
Pathologist Claudio A. Mosse, MD, PhD, of Vanderbilt University Medical Center and VA Tennessee Valley Healthcare System, praised the virtual tumor board program. “It’s been incredibly useful from my end as a pathologist as it shows me which diagnoses are most challenging for my colleagues,” Dr. Mosse said in an inter-view. “Reviewing and then presenting these challenging cases forces me to go into the published literature to come to a unitary diagnosis based on the patient history, radiology, various laboratory tests, and the biopsy I was asked to review.”
He added that “as a pathologist, I learn so much from the hematologists as they discuss the possible therapeutic options, and that strengthens my ability as a pathologist because I have to understand how one diagnosis versus an-other affects their therapeutic decision tree.”
What’s next for the virtual tumor board program? The next step is to expand to solid tumors, said VA Pittsburgh Healthcare System hematologist/oncologist Vida Almario Passero, MD, MBA, chief medical officer of National TeleOncology, in an interview.
No disclosures were reported.
New Research Supports a Changing Approach to Peripheral Artery Disease
SAN DIEGO–A cardiologist/vascular medicine specialist urged hematologist and oncologists within the US Department of Veterans Affairs system to think beyond the guidelines–at least until they’re updated–when they consider how to treat peripheral artery disease (PAD).
The 2016 American College of Cardiology/American Heart Association guidelines for PAD care are due for an update and don’t reflect recent positive research into the role that the blood thinner rivaroxaban can play in certain patients, said Geoffrey Barnes, MD, MSc, of the University of Michigan Health System, in a presentation here at the annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Recent research has “really got us excited about the potential for this drug in this particular patient population,” Barnes said, although he cautioned that it’s most appropriate for patients at highest risk of PAD.
Research has found that patients with PAD are more likely to develop cancer, apparently because of common risk factors, and there’s discussion about whether they should undergo special screening. Cancer treatment may also boost the risk of PAD, according to a 2021 US study that tracked 248 patients with both breast cancer and PAD. “Of all patients, 48% were on statins and 54% were on antiplatelet therapies,” the study found, although the 2016 guidelines recommend both (statins for all patients with PAD, antiplatelets for those with symptoms).
In his presentation, Barnes noted that the 2016 guidelines specifically recommend aspirin (75-325 mg daily) or clopidogrel (75 mg) in patients with symptomatic PAD. Treatment is especially important, he said, because the risk of cardiovascular mortality in PAD is high. A 2020 study found that 9.1% of 13,885 patients died over a median 30-month follow-up.
The good news about treatment Brand said, came in a 2020 industry-funded study of patients with PAD who had undergone revascularization. Various outcomes such as amputation, heart attack, and death from cardiovascular causes—the primary efficacy outcome—were less common in subjects who took 2.5 mg twice daily of rivaroxaban plus aspirin or placebo plus aspirin (hazard ratio, 0.85, 95% CI, 0.76-0.96; P = .009).
So who should go on rivaroxaban? As Brand noted, a 2019 study found that patients with no high-risk features didn’t benefit much in terms of risk of vascular events, but those with high-risk features did. In higher-risk patients, the study found, “rivaroxaban and aspirin prevented 33 serious vascular events, whereas in lower-risk patients, rivaroxaban and aspirin treatment led to the avoidance of 10 events per 1,000 patients treated for 30 months.”
Per the study, patients at higher risk are those with heart failure, at least 2 vascular beds affected, renal insufficiency, or diabetes.
Brand supports the use of rivaroxaban in these patients. However, he cautioned colleagues not to switch out the drug with apixaban, another blood thinner. “These are not interchangeable,” he said. “You do need to stick with rivaroxaban. And you do need to remember that you’re going to use 2.5 milligrams twice a day—very different than many of the other ways we are using rivaroxaban.”
Brand discloses consulting fees (Pfizer/Bristol-Myers Squib, Janssen, Acelis Connected Health, Boston Scientific, Abbott Vascular), grant funding (Boston Scientific) and board of directors service (Anticoagulation Forum).
SAN DIEGO–A cardiologist/vascular medicine specialist urged hematologist and oncologists within the US Department of Veterans Affairs system to think beyond the guidelines–at least until they’re updated–when they consider how to treat peripheral artery disease (PAD).
The 2016 American College of Cardiology/American Heart Association guidelines for PAD care are due for an update and don’t reflect recent positive research into the role that the blood thinner rivaroxaban can play in certain patients, said Geoffrey Barnes, MD, MSc, of the University of Michigan Health System, in a presentation here at the annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Recent research has “really got us excited about the potential for this drug in this particular patient population,” Barnes said, although he cautioned that it’s most appropriate for patients at highest risk of PAD.
Research has found that patients with PAD are more likely to develop cancer, apparently because of common risk factors, and there’s discussion about whether they should undergo special screening. Cancer treatment may also boost the risk of PAD, according to a 2021 US study that tracked 248 patients with both breast cancer and PAD. “Of all patients, 48% were on statins and 54% were on antiplatelet therapies,” the study found, although the 2016 guidelines recommend both (statins for all patients with PAD, antiplatelets for those with symptoms).
In his presentation, Barnes noted that the 2016 guidelines specifically recommend aspirin (75-325 mg daily) or clopidogrel (75 mg) in patients with symptomatic PAD. Treatment is especially important, he said, because the risk of cardiovascular mortality in PAD is high. A 2020 study found that 9.1% of 13,885 patients died over a median 30-month follow-up.
The good news about treatment Brand said, came in a 2020 industry-funded study of patients with PAD who had undergone revascularization. Various outcomes such as amputation, heart attack, and death from cardiovascular causes—the primary efficacy outcome—were less common in subjects who took 2.5 mg twice daily of rivaroxaban plus aspirin or placebo plus aspirin (hazard ratio, 0.85, 95% CI, 0.76-0.96; P = .009).
So who should go on rivaroxaban? As Brand noted, a 2019 study found that patients with no high-risk features didn’t benefit much in terms of risk of vascular events, but those with high-risk features did. In higher-risk patients, the study found, “rivaroxaban and aspirin prevented 33 serious vascular events, whereas in lower-risk patients, rivaroxaban and aspirin treatment led to the avoidance of 10 events per 1,000 patients treated for 30 months.”
Per the study, patients at higher risk are those with heart failure, at least 2 vascular beds affected, renal insufficiency, or diabetes.
Brand supports the use of rivaroxaban in these patients. However, he cautioned colleagues not to switch out the drug with apixaban, another blood thinner. “These are not interchangeable,” he said. “You do need to stick with rivaroxaban. And you do need to remember that you’re going to use 2.5 milligrams twice a day—very different than many of the other ways we are using rivaroxaban.”
Brand discloses consulting fees (Pfizer/Bristol-Myers Squib, Janssen, Acelis Connected Health, Boston Scientific, Abbott Vascular), grant funding (Boston Scientific) and board of directors service (Anticoagulation Forum).
SAN DIEGO–A cardiologist/vascular medicine specialist urged hematologist and oncologists within the US Department of Veterans Affairs system to think beyond the guidelines–at least until they’re updated–when they consider how to treat peripheral artery disease (PAD).
The 2016 American College of Cardiology/American Heart Association guidelines for PAD care are due for an update and don’t reflect recent positive research into the role that the blood thinner rivaroxaban can play in certain patients, said Geoffrey Barnes, MD, MSc, of the University of Michigan Health System, in a presentation here at the annual meeting of the Association of VA Hematology/Oncology (AVAHO).
Recent research has “really got us excited about the potential for this drug in this particular patient population,” Barnes said, although he cautioned that it’s most appropriate for patients at highest risk of PAD.
Research has found that patients with PAD are more likely to develop cancer, apparently because of common risk factors, and there’s discussion about whether they should undergo special screening. Cancer treatment may also boost the risk of PAD, according to a 2021 US study that tracked 248 patients with both breast cancer and PAD. “Of all patients, 48% were on statins and 54% were on antiplatelet therapies,” the study found, although the 2016 guidelines recommend both (statins for all patients with PAD, antiplatelets for those with symptoms).
In his presentation, Barnes noted that the 2016 guidelines specifically recommend aspirin (75-325 mg daily) or clopidogrel (75 mg) in patients with symptomatic PAD. Treatment is especially important, he said, because the risk of cardiovascular mortality in PAD is high. A 2020 study found that 9.1% of 13,885 patients died over a median 30-month follow-up.
The good news about treatment Brand said, came in a 2020 industry-funded study of patients with PAD who had undergone revascularization. Various outcomes such as amputation, heart attack, and death from cardiovascular causes—the primary efficacy outcome—were less common in subjects who took 2.5 mg twice daily of rivaroxaban plus aspirin or placebo plus aspirin (hazard ratio, 0.85, 95% CI, 0.76-0.96; P = .009).
So who should go on rivaroxaban? As Brand noted, a 2019 study found that patients with no high-risk features didn’t benefit much in terms of risk of vascular events, but those with high-risk features did. In higher-risk patients, the study found, “rivaroxaban and aspirin prevented 33 serious vascular events, whereas in lower-risk patients, rivaroxaban and aspirin treatment led to the avoidance of 10 events per 1,000 patients treated for 30 months.”
Per the study, patients at higher risk are those with heart failure, at least 2 vascular beds affected, renal insufficiency, or diabetes.
Brand supports the use of rivaroxaban in these patients. However, he cautioned colleagues not to switch out the drug with apixaban, another blood thinner. “These are not interchangeable,” he said. “You do need to stick with rivaroxaban. And you do need to remember that you’re going to use 2.5 milligrams twice a day—very different than many of the other ways we are using rivaroxaban.”
Brand discloses consulting fees (Pfizer/Bristol-Myers Squib, Janssen, Acelis Connected Health, Boston Scientific, Abbott Vascular), grant funding (Boston Scientific) and board of directors service (Anticoagulation Forum).
Time to pull back on postsurgery radiation in breast cancer?
A new study suggests that oncologists can safely pull back on standard locoregional radiotherapy (RT) in select patients with cT1-2N1 breast cancer who are treated with primary chemotherapy prior to surgery. The key is to divide patients by risk level and treat them according to the study’s guidelines, the researchers reported.
lead study author Sabine de Wild, MD, a PhD student at Maastricht (the Netherlands) University Medical Center, said in an interview.
The study, published in The Lancet Oncology, was intended to provide insight into which breast cancer patients need adjuvant locoregional radiotherapy following postchemotherapy surgery, coauthor Liesbeth Boersma, MD, PhD, a radiation oncologist at Maastricht University Medical Center, said in an interview. “It is not yet known which of these patients would benefit from adjuvant locoregional radiotherapy and to what extent the response of the tumor to the chemotherapy should be taken into account.”
For the study, believed to be the first prospective analysis tackling this topic, researchers tracked 838 patients in The Netherlands who were treated for cT1-2N1 breast cancer with primary chemotherapy and surgery of the breast and axilla from 2011-2015. Tumors were less than 5 cm and metastases were one to three axillary nodes.
The subjects were divided into groups based on risk of locoregional recurrence, and each group underwent different therapies.
- Low-risk group: no metastases were present in the nodes (n = 291). “We omitted regional radiotherapy, and we omitted RT of the chest wall in case of a mastectomy. After breast conserving surgery, regular RT of the breast was recommended,” Dr. de Wild said.
- Intermediate-risk group, one to three metastases were still present (n = 370). “We omitted regional radiotherapy, but irradiated the chest wall or breast,” she said.
- High-risk group, three metastases were present (n = 177). “We did not de-escalate, and all patients were treated with locoregional RT,” she said.
According to the study, “the 5-year locoregional recurrence rate in all patients was 2.2% (95% confidence interval, 1.4-3.4). The 5-year locoregional recurrence rate was 2.1% (95% CI, 0.9-4.3) in the low-risk group, 2.2% (95% CI, 1.0-4.1) in the intermediate-risk group, and 2.3% (95% CI, 0.8-5.5) in the high-risk group.”
In 26% of cases, patients received more radiotherapy than the study guidelines suggested. “Remarkably,” the researchers wrote, “this did not seem to affect locoregional recurrence rate, recurrence-free interval, and overall survival in a statistically significant or clinically relevant way.”
As for limitations, the authors noted that, “in each risk group, the actual sample size treated according to the study guideline was smaller than required based on the power calculation. Nevertheless, when performing the analyses in the subset of patients treated according to the study guideline, the upper limit of 95% CI of 5-year locoregional recurrence rate did not exceed 7.8%.”
The study authors wrote that, “in the future, the results of this study might lead to more frequent omission of locoregional radiotherapy, which could result in lower morbidity and a better quality of life for patients with breast cancer who are receiving primary chemotherapy.”
However, Dr. de Wild said randomized trials are necessary “to investigate how treatment can be individualized further, i.e., by taking into account specific tumor characteristics.” Also, most patients in the study underwent axillary lymph node dissection, “while patients in daily practice may instead undergo targeted axillary dissection. Future studies are needed to determine if less radiotherapy is also safe in patients in whom axillary lymph node dissection is omitted.”
The study was funded by the Dutch Cancer Society. One coauthor reported a pending patent plus grants from AstraZeneca, Eurocept Plaza, Roche, Genentech, Gilead Sciences, Tesaro, Novartis, Dutch Cancer Society, ZonMw, and A Sister’s Hope; as well as consulting fees and other financial support from a variety of pharmaceutical companies. The other authors had no disclosures.
A new study suggests that oncologists can safely pull back on standard locoregional radiotherapy (RT) in select patients with cT1-2N1 breast cancer who are treated with primary chemotherapy prior to surgery. The key is to divide patients by risk level and treat them according to the study’s guidelines, the researchers reported.
lead study author Sabine de Wild, MD, a PhD student at Maastricht (the Netherlands) University Medical Center, said in an interview.
The study, published in The Lancet Oncology, was intended to provide insight into which breast cancer patients need adjuvant locoregional radiotherapy following postchemotherapy surgery, coauthor Liesbeth Boersma, MD, PhD, a radiation oncologist at Maastricht University Medical Center, said in an interview. “It is not yet known which of these patients would benefit from adjuvant locoregional radiotherapy and to what extent the response of the tumor to the chemotherapy should be taken into account.”
For the study, believed to be the first prospective analysis tackling this topic, researchers tracked 838 patients in The Netherlands who were treated for cT1-2N1 breast cancer with primary chemotherapy and surgery of the breast and axilla from 2011-2015. Tumors were less than 5 cm and metastases were one to three axillary nodes.
The subjects were divided into groups based on risk of locoregional recurrence, and each group underwent different therapies.
- Low-risk group: no metastases were present in the nodes (n = 291). “We omitted regional radiotherapy, and we omitted RT of the chest wall in case of a mastectomy. After breast conserving surgery, regular RT of the breast was recommended,” Dr. de Wild said.
- Intermediate-risk group, one to three metastases were still present (n = 370). “We omitted regional radiotherapy, but irradiated the chest wall or breast,” she said.
- High-risk group, three metastases were present (n = 177). “We did not de-escalate, and all patients were treated with locoregional RT,” she said.
According to the study, “the 5-year locoregional recurrence rate in all patients was 2.2% (95% confidence interval, 1.4-3.4). The 5-year locoregional recurrence rate was 2.1% (95% CI, 0.9-4.3) in the low-risk group, 2.2% (95% CI, 1.0-4.1) in the intermediate-risk group, and 2.3% (95% CI, 0.8-5.5) in the high-risk group.”
In 26% of cases, patients received more radiotherapy than the study guidelines suggested. “Remarkably,” the researchers wrote, “this did not seem to affect locoregional recurrence rate, recurrence-free interval, and overall survival in a statistically significant or clinically relevant way.”
As for limitations, the authors noted that, “in each risk group, the actual sample size treated according to the study guideline was smaller than required based on the power calculation. Nevertheless, when performing the analyses in the subset of patients treated according to the study guideline, the upper limit of 95% CI of 5-year locoregional recurrence rate did not exceed 7.8%.”
The study authors wrote that, “in the future, the results of this study might lead to more frequent omission of locoregional radiotherapy, which could result in lower morbidity and a better quality of life for patients with breast cancer who are receiving primary chemotherapy.”
However, Dr. de Wild said randomized trials are necessary “to investigate how treatment can be individualized further, i.e., by taking into account specific tumor characteristics.” Also, most patients in the study underwent axillary lymph node dissection, “while patients in daily practice may instead undergo targeted axillary dissection. Future studies are needed to determine if less radiotherapy is also safe in patients in whom axillary lymph node dissection is omitted.”
The study was funded by the Dutch Cancer Society. One coauthor reported a pending patent plus grants from AstraZeneca, Eurocept Plaza, Roche, Genentech, Gilead Sciences, Tesaro, Novartis, Dutch Cancer Society, ZonMw, and A Sister’s Hope; as well as consulting fees and other financial support from a variety of pharmaceutical companies. The other authors had no disclosures.
A new study suggests that oncologists can safely pull back on standard locoregional radiotherapy (RT) in select patients with cT1-2N1 breast cancer who are treated with primary chemotherapy prior to surgery. The key is to divide patients by risk level and treat them according to the study’s guidelines, the researchers reported.
lead study author Sabine de Wild, MD, a PhD student at Maastricht (the Netherlands) University Medical Center, said in an interview.
The study, published in The Lancet Oncology, was intended to provide insight into which breast cancer patients need adjuvant locoregional radiotherapy following postchemotherapy surgery, coauthor Liesbeth Boersma, MD, PhD, a radiation oncologist at Maastricht University Medical Center, said in an interview. “It is not yet known which of these patients would benefit from adjuvant locoregional radiotherapy and to what extent the response of the tumor to the chemotherapy should be taken into account.”
For the study, believed to be the first prospective analysis tackling this topic, researchers tracked 838 patients in The Netherlands who were treated for cT1-2N1 breast cancer with primary chemotherapy and surgery of the breast and axilla from 2011-2015. Tumors were less than 5 cm and metastases were one to three axillary nodes.
The subjects were divided into groups based on risk of locoregional recurrence, and each group underwent different therapies.
- Low-risk group: no metastases were present in the nodes (n = 291). “We omitted regional radiotherapy, and we omitted RT of the chest wall in case of a mastectomy. After breast conserving surgery, regular RT of the breast was recommended,” Dr. de Wild said.
- Intermediate-risk group, one to three metastases were still present (n = 370). “We omitted regional radiotherapy, but irradiated the chest wall or breast,” she said.
- High-risk group, three metastases were present (n = 177). “We did not de-escalate, and all patients were treated with locoregional RT,” she said.
According to the study, “the 5-year locoregional recurrence rate in all patients was 2.2% (95% confidence interval, 1.4-3.4). The 5-year locoregional recurrence rate was 2.1% (95% CI, 0.9-4.3) in the low-risk group, 2.2% (95% CI, 1.0-4.1) in the intermediate-risk group, and 2.3% (95% CI, 0.8-5.5) in the high-risk group.”
In 26% of cases, patients received more radiotherapy than the study guidelines suggested. “Remarkably,” the researchers wrote, “this did not seem to affect locoregional recurrence rate, recurrence-free interval, and overall survival in a statistically significant or clinically relevant way.”
As for limitations, the authors noted that, “in each risk group, the actual sample size treated according to the study guideline was smaller than required based on the power calculation. Nevertheless, when performing the analyses in the subset of patients treated according to the study guideline, the upper limit of 95% CI of 5-year locoregional recurrence rate did not exceed 7.8%.”
The study authors wrote that, “in the future, the results of this study might lead to more frequent omission of locoregional radiotherapy, which could result in lower morbidity and a better quality of life for patients with breast cancer who are receiving primary chemotherapy.”
However, Dr. de Wild said randomized trials are necessary “to investigate how treatment can be individualized further, i.e., by taking into account specific tumor characteristics.” Also, most patients in the study underwent axillary lymph node dissection, “while patients in daily practice may instead undergo targeted axillary dissection. Future studies are needed to determine if less radiotherapy is also safe in patients in whom axillary lymph node dissection is omitted.”
The study was funded by the Dutch Cancer Society. One coauthor reported a pending patent plus grants from AstraZeneca, Eurocept Plaza, Roche, Genentech, Gilead Sciences, Tesaro, Novartis, Dutch Cancer Society, ZonMw, and A Sister’s Hope; as well as consulting fees and other financial support from a variety of pharmaceutical companies. The other authors had no disclosures.
FROM THE LANCET ONCOLOGY
Oncologists often misinterpret posttreatment HNSCC scan details
Patient outcomes could be threatened because of misinterpretation by oncologic surgeons of free-form posttreatment radiological reports in head and neck squamous cell carcinoma (HNSCC), a new study finds.
“Clinician perception of patient response from the post-RT [radiation treament] PET/CT free-form report is unreliable and does not consistently reflect the radiologist’s intended meaning, which was strongly associated with survival,” researchers wrote in a study published Aug. 18 in JAMA Otolaryngology-Head & Neck Surgery. They found “minimal agreement between clinicians’ consensus perspective on the patient’s response status derived from free-form imaging reports and the criterion standard response category assigned by a nuclear medicine specialist after PET/CT image review.”
According to radiation oncologist Ryan T. Hughes, MD, and colleagues at Wake Forest University, Winston-Salem, N.C., it’s common for patients with HNSCC to get PET, CT, or PET/CT imaging following treatment in order to assess how patients responded. Accurate communication about the results is essential to determining next steps, they write.
However, they write, “to our knowledge there is no universally accepted standardized method for communicating results,” such as whether there’s been a complete or partial response. Discrepancies between a radiological posttreatment report and an oncologist’s perception of the findings “may contribute to unnecessary patient care complexities, including elevated patient anxiety, unnecessary follow-up testing/procedures, and failure to recognize and adequately treat residual, recurrent, or progressive disease,” the researchers write.
For the new study, the authors tracked 171 patients (26.3% women, median age 61 years, ethnicity not provided), mainly (87%) with stage III-IV disease. Most (89%) received concurrent chemotherapy, and 30% received radiotherapy following operations.
Four oncologists reviewed free-form radiologic reports and determined whether the patient had a complete, indeterminate or partial response, or progressive disease. “Next, the group conferred to assign a consensus clinician MDS [modified Deauville score] and associated response category to assess the percentage of agreement with the criterion standard nuclear medicine physician MDS response derived from PET/CT image review.”
The researchers found that “interrater reliability of clinician-perceived post-RT PET/CT response was moderate [k = 0.680; 95% confidence interval, 0.638-0.721], and there was minimal reliability and low rate of agreement between clinician perception and radiologist-intended PET/CT response [63.7%; k = 0.365; 95% CI, 0.251-0.478).”
The clinicians were more likely to perceive patients as having an indeterminate response (28.1%), compared with the radiologists (9.3%). “There were 16 instances of significant discordance: 7 patients for whom the clinician perception MDS was 1 to 2 and nuclear medicine MDS 3 to 4, and 9 patients for whom the clinician perception MDS was 3 to 4 and nuclear medicine MDS 1 to 2.”
Due to statistical limitations, the researchers were unable to link the MDS scores to prognoses. The researchers suggest it’s time to further standardize the assessment of posttreatment responses to therapy. They add that “the decision to use a standardized interpretation and reporting system rather than free-form reporting is more important than the specific system selected.”
As for next steps, the researchers report that “prospective studies of post-RT PET/CT standardized reporting among patients with HNSCC are warranted, and a prospective implementation study of this workflow is planned at our institution.”
The study was funded by the National Center for Advancing Translational Sciences and National Institutes of Health. The authors had no disclosures.
Patient outcomes could be threatened because of misinterpretation by oncologic surgeons of free-form posttreatment radiological reports in head and neck squamous cell carcinoma (HNSCC), a new study finds.
“Clinician perception of patient response from the post-RT [radiation treament] PET/CT free-form report is unreliable and does not consistently reflect the radiologist’s intended meaning, which was strongly associated with survival,” researchers wrote in a study published Aug. 18 in JAMA Otolaryngology-Head & Neck Surgery. They found “minimal agreement between clinicians’ consensus perspective on the patient’s response status derived from free-form imaging reports and the criterion standard response category assigned by a nuclear medicine specialist after PET/CT image review.”
According to radiation oncologist Ryan T. Hughes, MD, and colleagues at Wake Forest University, Winston-Salem, N.C., it’s common for patients with HNSCC to get PET, CT, or PET/CT imaging following treatment in order to assess how patients responded. Accurate communication about the results is essential to determining next steps, they write.
However, they write, “to our knowledge there is no universally accepted standardized method for communicating results,” such as whether there’s been a complete or partial response. Discrepancies between a radiological posttreatment report and an oncologist’s perception of the findings “may contribute to unnecessary patient care complexities, including elevated patient anxiety, unnecessary follow-up testing/procedures, and failure to recognize and adequately treat residual, recurrent, or progressive disease,” the researchers write.
For the new study, the authors tracked 171 patients (26.3% women, median age 61 years, ethnicity not provided), mainly (87%) with stage III-IV disease. Most (89%) received concurrent chemotherapy, and 30% received radiotherapy following operations.
Four oncologists reviewed free-form radiologic reports and determined whether the patient had a complete, indeterminate or partial response, or progressive disease. “Next, the group conferred to assign a consensus clinician MDS [modified Deauville score] and associated response category to assess the percentage of agreement with the criterion standard nuclear medicine physician MDS response derived from PET/CT image review.”
The researchers found that “interrater reliability of clinician-perceived post-RT PET/CT response was moderate [k = 0.680; 95% confidence interval, 0.638-0.721], and there was minimal reliability and low rate of agreement between clinician perception and radiologist-intended PET/CT response [63.7%; k = 0.365; 95% CI, 0.251-0.478).”
The clinicians were more likely to perceive patients as having an indeterminate response (28.1%), compared with the radiologists (9.3%). “There were 16 instances of significant discordance: 7 patients for whom the clinician perception MDS was 1 to 2 and nuclear medicine MDS 3 to 4, and 9 patients for whom the clinician perception MDS was 3 to 4 and nuclear medicine MDS 1 to 2.”
Due to statistical limitations, the researchers were unable to link the MDS scores to prognoses. The researchers suggest it’s time to further standardize the assessment of posttreatment responses to therapy. They add that “the decision to use a standardized interpretation and reporting system rather than free-form reporting is more important than the specific system selected.”
As for next steps, the researchers report that “prospective studies of post-RT PET/CT standardized reporting among patients with HNSCC are warranted, and a prospective implementation study of this workflow is planned at our institution.”
The study was funded by the National Center for Advancing Translational Sciences and National Institutes of Health. The authors had no disclosures.
Patient outcomes could be threatened because of misinterpretation by oncologic surgeons of free-form posttreatment radiological reports in head and neck squamous cell carcinoma (HNSCC), a new study finds.
“Clinician perception of patient response from the post-RT [radiation treament] PET/CT free-form report is unreliable and does not consistently reflect the radiologist’s intended meaning, which was strongly associated with survival,” researchers wrote in a study published Aug. 18 in JAMA Otolaryngology-Head & Neck Surgery. They found “minimal agreement between clinicians’ consensus perspective on the patient’s response status derived from free-form imaging reports and the criterion standard response category assigned by a nuclear medicine specialist after PET/CT image review.”
According to radiation oncologist Ryan T. Hughes, MD, and colleagues at Wake Forest University, Winston-Salem, N.C., it’s common for patients with HNSCC to get PET, CT, or PET/CT imaging following treatment in order to assess how patients responded. Accurate communication about the results is essential to determining next steps, they write.
However, they write, “to our knowledge there is no universally accepted standardized method for communicating results,” such as whether there’s been a complete or partial response. Discrepancies between a radiological posttreatment report and an oncologist’s perception of the findings “may contribute to unnecessary patient care complexities, including elevated patient anxiety, unnecessary follow-up testing/procedures, and failure to recognize and adequately treat residual, recurrent, or progressive disease,” the researchers write.
For the new study, the authors tracked 171 patients (26.3% women, median age 61 years, ethnicity not provided), mainly (87%) with stage III-IV disease. Most (89%) received concurrent chemotherapy, and 30% received radiotherapy following operations.
Four oncologists reviewed free-form radiologic reports and determined whether the patient had a complete, indeterminate or partial response, or progressive disease. “Next, the group conferred to assign a consensus clinician MDS [modified Deauville score] and associated response category to assess the percentage of agreement with the criterion standard nuclear medicine physician MDS response derived from PET/CT image review.”
The researchers found that “interrater reliability of clinician-perceived post-RT PET/CT response was moderate [k = 0.680; 95% confidence interval, 0.638-0.721], and there was minimal reliability and low rate of agreement between clinician perception and radiologist-intended PET/CT response [63.7%; k = 0.365; 95% CI, 0.251-0.478).”
The clinicians were more likely to perceive patients as having an indeterminate response (28.1%), compared with the radiologists (9.3%). “There were 16 instances of significant discordance: 7 patients for whom the clinician perception MDS was 1 to 2 and nuclear medicine MDS 3 to 4, and 9 patients for whom the clinician perception MDS was 3 to 4 and nuclear medicine MDS 1 to 2.”
Due to statistical limitations, the researchers were unable to link the MDS scores to prognoses. The researchers suggest it’s time to further standardize the assessment of posttreatment responses to therapy. They add that “the decision to use a standardized interpretation and reporting system rather than free-form reporting is more important than the specific system selected.”
As for next steps, the researchers report that “prospective studies of post-RT PET/CT standardized reporting among patients with HNSCC are warranted, and a prospective implementation study of this workflow is planned at our institution.”
The study was funded by the National Center for Advancing Translational Sciences and National Institutes of Health. The authors had no disclosures.
FROM JAMA OTOLARYNGOLOGY–HEAD & NECK SURGERY
Thyroid autoimmunity linked to cancer, but screening not advised
A new study provides more evidence that people with thyroid autoimmunity are more likely than are others to develop papillary thyroid cancer (odds ratio [OR] = 1.90, 95% confidence interval [CI], 1.33-2.70), although the overall risk remains very low.
Researchers aren't recommending routine screening in all patients with thyroid autoimmunity, but they're calling for more research into whether it's a good idea in severe cases. "This is the one circumstance where screening for subclinical disease could make sense," said Donald McLeod, MPH, PhD, an epidemiologist at Royal Brisbane & Women's Hospital in Australia and lead author of the study, published in the Journal of Clinical Oncology. "However, more research is needed because our study is the first to show this result, and we need to prove that screening would make a difference to the prognosis of these patients."
According to Dr. McLeod, "doctors and patients have been wondering about the connection between thyroid autoimmunity and thyroid cancer for many years. In fact, the first report was in 1955. While the association was plausible, all previous studies had potential for biases that could have influenced the results."
For example, he said, multiple studies didn't control for confounders, while others didn't account for the possibility that cancer could have triggered an immune response. "Other case-control studies could have been affected by selection bias, where a diagnosis of thyroid autoimmunity leads to thyroid cancer identification and entry into the study," he said. "Finally, medical surveillance of people diagnosed with thyroid autoimmunity could lead to overdiagnosis, where small, subclinical cancers are diagnosed in those patients but not identified in people who are not under medical follow-up."
For the new retrospective case-control study, researchers compared 451 active-duty members of the U.S. military who developed papillary thyroid cancer from the period of 1996-2014 to matched controls (61% of all subjects were men and the mean age was 36). Those with cancer had their serum collected 3-5 years and 7-10 years before the date of diagnosis - the index date for all subjects. Some of those considered to have thyroid autoimmunity had conditions such as Graves' disease and Hashimoto's thyroiditis.
"Eighty-five percent of cases (379 of 451) had a thyroid-related diagnosis recorded ... before their index date, compared with 5% of controls," the researchers reported. "Most cases (80%) had classical papillary thyroid cancer, with the rest having the follicular variant of papillary thyroid cancer."
After adjustment to account for various confounders, those who were positive for thyroid peroxidase antibodies 7-10 years prior to the index date were more likely to have developed thyroid cancer (OR = 1.90, 95% CI, 1.33-2.70). "The results could not be fully explained by diagnosis of thyroid autoimmunity," the researchers reported, "although when autoimmunity had been identified, thyroid cancers were diagnosed at a very early stage."
Two groups - those with the highest thyroid antibody levels and women - faced the greatest risk, Dr. McLeod said. The results regarding women were the most surprising in the study, he said. "This is the first time this has been found. We think this result needs to be confirmed. If true, it could explain why women have a three-times-higher risk of thyroid cancer than men."
The overall incidence of thyroid cancer in the U.S. was estimated at 13.49 per 100,000 person-years in 2018, with women (76% of cases) and Whites (81%) accounting for the majority. Rates have nearly doubled since 2000. The authors of a 2022 report that disclosed these numbers suggest the rise is due to overdiagnosis of small tumors.
It's not clear why thyroid autoimmunity and thyroid cancer may be linked. "Chronic inflammation from thyroid autoimmunity could cause thyroid cancer, as chronic inflammation in other organs precedes cancers at those sites," Dr. McLeod said. "Alternatively, thyroid autoimmunity could appear to be associated with thyroid cancer because of biases inherent in previous studies, including previous diagnosis of autoimmunity. Thyroid cancer could also induce an immune response, which mimics thyroid autoimmunity and could bias assessment."
As for screening of patients with thyroid autoimmunity, "the main danger is that you will commonly identify small thyroid cancers that would never become clinically apparent," he said. "This leads to unnecessary treatments that can cause complications and give people a cancer label, which can also cause harm. Diagnosis and treatment guidelines recommend against screening the general population for this reason."
Many of those with thyroid autoimmunity developed small cancers, he said, most likely "detected from ultrasound being performed because autoimmune thyroid disease was known. If all patients with thyroid autoimmunity were screened for thyroid cancer, the likelihood is that many people's cancers would be overdiagnosed."
The study was funded by the Walton Family Foundation. Dr. McLeod reports no disclosures. Some of the authors report various relationships with industry.
A new study provides more evidence that people with thyroid autoimmunity are more likely than are others to develop papillary thyroid cancer (odds ratio [OR] = 1.90, 95% confidence interval [CI], 1.33-2.70), although the overall risk remains very low.
Researchers aren't recommending routine screening in all patients with thyroid autoimmunity, but they're calling for more research into whether it's a good idea in severe cases. "This is the one circumstance where screening for subclinical disease could make sense," said Donald McLeod, MPH, PhD, an epidemiologist at Royal Brisbane & Women's Hospital in Australia and lead author of the study, published in the Journal of Clinical Oncology. "However, more research is needed because our study is the first to show this result, and we need to prove that screening would make a difference to the prognosis of these patients."
According to Dr. McLeod, "doctors and patients have been wondering about the connection between thyroid autoimmunity and thyroid cancer for many years. In fact, the first report was in 1955. While the association was plausible, all previous studies had potential for biases that could have influenced the results."
For example, he said, multiple studies didn't control for confounders, while others didn't account for the possibility that cancer could have triggered an immune response. "Other case-control studies could have been affected by selection bias, where a diagnosis of thyroid autoimmunity leads to thyroid cancer identification and entry into the study," he said. "Finally, medical surveillance of people diagnosed with thyroid autoimmunity could lead to overdiagnosis, where small, subclinical cancers are diagnosed in those patients but not identified in people who are not under medical follow-up."
For the new retrospective case-control study, researchers compared 451 active-duty members of the U.S. military who developed papillary thyroid cancer from the period of 1996-2014 to matched controls (61% of all subjects were men and the mean age was 36). Those with cancer had their serum collected 3-5 years and 7-10 years before the date of diagnosis - the index date for all subjects. Some of those considered to have thyroid autoimmunity had conditions such as Graves' disease and Hashimoto's thyroiditis.
"Eighty-five percent of cases (379 of 451) had a thyroid-related diagnosis recorded ... before their index date, compared with 5% of controls," the researchers reported. "Most cases (80%) had classical papillary thyroid cancer, with the rest having the follicular variant of papillary thyroid cancer."
After adjustment to account for various confounders, those who were positive for thyroid peroxidase antibodies 7-10 years prior to the index date were more likely to have developed thyroid cancer (OR = 1.90, 95% CI, 1.33-2.70). "The results could not be fully explained by diagnosis of thyroid autoimmunity," the researchers reported, "although when autoimmunity had been identified, thyroid cancers were diagnosed at a very early stage."
Two groups - those with the highest thyroid antibody levels and women - faced the greatest risk, Dr. McLeod said. The results regarding women were the most surprising in the study, he said. "This is the first time this has been found. We think this result needs to be confirmed. If true, it could explain why women have a three-times-higher risk of thyroid cancer than men."
The overall incidence of thyroid cancer in the U.S. was estimated at 13.49 per 100,000 person-years in 2018, with women (76% of cases) and Whites (81%) accounting for the majority. Rates have nearly doubled since 2000. The authors of a 2022 report that disclosed these numbers suggest the rise is due to overdiagnosis of small tumors.
It's not clear why thyroid autoimmunity and thyroid cancer may be linked. "Chronic inflammation from thyroid autoimmunity could cause thyroid cancer, as chronic inflammation in other organs precedes cancers at those sites," Dr. McLeod said. "Alternatively, thyroid autoimmunity could appear to be associated with thyroid cancer because of biases inherent in previous studies, including previous diagnosis of autoimmunity. Thyroid cancer could also induce an immune response, which mimics thyroid autoimmunity and could bias assessment."
As for screening of patients with thyroid autoimmunity, "the main danger is that you will commonly identify small thyroid cancers that would never become clinically apparent," he said. "This leads to unnecessary treatments that can cause complications and give people a cancer label, which can also cause harm. Diagnosis and treatment guidelines recommend against screening the general population for this reason."
Many of those with thyroid autoimmunity developed small cancers, he said, most likely "detected from ultrasound being performed because autoimmune thyroid disease was known. If all patients with thyroid autoimmunity were screened for thyroid cancer, the likelihood is that many people's cancers would be overdiagnosed."
The study was funded by the Walton Family Foundation. Dr. McLeod reports no disclosures. Some of the authors report various relationships with industry.
A new study provides more evidence that people with thyroid autoimmunity are more likely than are others to develop papillary thyroid cancer (odds ratio [OR] = 1.90, 95% confidence interval [CI], 1.33-2.70), although the overall risk remains very low.
Researchers aren't recommending routine screening in all patients with thyroid autoimmunity, but they're calling for more research into whether it's a good idea in severe cases. "This is the one circumstance where screening for subclinical disease could make sense," said Donald McLeod, MPH, PhD, an epidemiologist at Royal Brisbane & Women's Hospital in Australia and lead author of the study, published in the Journal of Clinical Oncology. "However, more research is needed because our study is the first to show this result, and we need to prove that screening would make a difference to the prognosis of these patients."
According to Dr. McLeod, "doctors and patients have been wondering about the connection between thyroid autoimmunity and thyroid cancer for many years. In fact, the first report was in 1955. While the association was plausible, all previous studies had potential for biases that could have influenced the results."
For example, he said, multiple studies didn't control for confounders, while others didn't account for the possibility that cancer could have triggered an immune response. "Other case-control studies could have been affected by selection bias, where a diagnosis of thyroid autoimmunity leads to thyroid cancer identification and entry into the study," he said. "Finally, medical surveillance of people diagnosed with thyroid autoimmunity could lead to overdiagnosis, where small, subclinical cancers are diagnosed in those patients but not identified in people who are not under medical follow-up."
For the new retrospective case-control study, researchers compared 451 active-duty members of the U.S. military who developed papillary thyroid cancer from the period of 1996-2014 to matched controls (61% of all subjects were men and the mean age was 36). Those with cancer had their serum collected 3-5 years and 7-10 years before the date of diagnosis - the index date for all subjects. Some of those considered to have thyroid autoimmunity had conditions such as Graves' disease and Hashimoto's thyroiditis.
"Eighty-five percent of cases (379 of 451) had a thyroid-related diagnosis recorded ... before their index date, compared with 5% of controls," the researchers reported. "Most cases (80%) had classical papillary thyroid cancer, with the rest having the follicular variant of papillary thyroid cancer."
After adjustment to account for various confounders, those who were positive for thyroid peroxidase antibodies 7-10 years prior to the index date were more likely to have developed thyroid cancer (OR = 1.90, 95% CI, 1.33-2.70). "The results could not be fully explained by diagnosis of thyroid autoimmunity," the researchers reported, "although when autoimmunity had been identified, thyroid cancers were diagnosed at a very early stage."
Two groups - those with the highest thyroid antibody levels and women - faced the greatest risk, Dr. McLeod said. The results regarding women were the most surprising in the study, he said. "This is the first time this has been found. We think this result needs to be confirmed. If true, it could explain why women have a three-times-higher risk of thyroid cancer than men."
The overall incidence of thyroid cancer in the U.S. was estimated at 13.49 per 100,000 person-years in 2018, with women (76% of cases) and Whites (81%) accounting for the majority. Rates have nearly doubled since 2000. The authors of a 2022 report that disclosed these numbers suggest the rise is due to overdiagnosis of small tumors.
It's not clear why thyroid autoimmunity and thyroid cancer may be linked. "Chronic inflammation from thyroid autoimmunity could cause thyroid cancer, as chronic inflammation in other organs precedes cancers at those sites," Dr. McLeod said. "Alternatively, thyroid autoimmunity could appear to be associated with thyroid cancer because of biases inherent in previous studies, including previous diagnosis of autoimmunity. Thyroid cancer could also induce an immune response, which mimics thyroid autoimmunity and could bias assessment."
As for screening of patients with thyroid autoimmunity, "the main danger is that you will commonly identify small thyroid cancers that would never become clinically apparent," he said. "This leads to unnecessary treatments that can cause complications and give people a cancer label, which can also cause harm. Diagnosis and treatment guidelines recommend against screening the general population for this reason."
Many of those with thyroid autoimmunity developed small cancers, he said, most likely "detected from ultrasound being performed because autoimmune thyroid disease was known. If all patients with thyroid autoimmunity were screened for thyroid cancer, the likelihood is that many people's cancers would be overdiagnosed."
The study was funded by the Walton Family Foundation. Dr. McLeod reports no disclosures. Some of the authors report various relationships with industry.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
‘Molecular map’ of CLL yields fresh genetic insights
Released in a report in Nature Genetics, the map has doubled the number of genetic traits linked the disease from around 100 to 202, lead author Binyamin A. Knisbacher, PhD, a postdoctoral fellow at the Broad Institute of MIT and Harvard Medical Schoo, Boston, said in an interview.
“It also delineated the molecular landscape of the two immunoglobulin gene (IGHV) subtypes, refined CLL subtyping, and built richer genetic prognostic models,” he said.
According to Dr. Knisbacher, CLL “has been at the forefront of genomic discovery,” and research has shown that there’s a wide variety of somatic mutations that drive CLL initiation across the patient population. However, as many as 10% of cases don’t appear to be driven by any known genetic variation, he said, and there’s a need to identify more subtypes and “build richer prognostic models of patient survival” based on genetics and multiomics such as genomics, transcriptomics, and epigenomics.
For the new study, researchers analyzed RNA and DNA from 1,095 patients with CLL and 54 patients with monoclonal B cell lymphocytosis and built what they say is the largest CLL dataset in existence. It’s twice the size of previous datasets, Dr. Knisbacher said.
“We found that RNA expression data was extremely informative for characterizing CLL,” Dr. Knisbacher said. “The RNA expression subtypes refined the ‘classic’ two IGHV subtypes. It is well documented that patients with U-CLL (IGHV-unmutated CLL) have substantially worse clinical outcome in comparison to M-CLL patients (IGHV-mutated CLLs). We found that M-CLLs that have RNA expression profiles similar to U-CLLs have worse survival than M-CLLs with a typical expression profile. Failure-free survival was 50% shorter – 5.3 versus 10.7 years median failure-free survival.”
In addition, he said, “U-CLLs with expression similar to M-CLLs had better survival than U-CLLs with an RNA expression profile typical to U-CLLs.”
The researchers have made their molecular map publicly available at https://cllmap.org/. Researchers can use it “to discover more about each subtype of CLL, and these future studies can help to improve clinical prognosis for the benefit of the patient,” Dr. Knisbacher said.
The study authors added that “this molecular foundation may allow for better prediction of response to therapy or provide the basis for rational combination of novel agents.”
Lee Greenberger, PhD, chief science officer of the Leukemia & Lymphoma Society, said in an interview that the study “provides foundational data further subtyping CLL patients and outcomes. It identifies new targets for therapy or diagnostic predictions in the future. This type of foundational work has proven invaluable in the development of new medicines for cancer in general.”
While there are many medications that have improved therapeutic outcomes in CLL, he added, “cures – or life-long disease control –remain elusive for many patients. Therefore, new molecular insights are needed that could personalize therapies or even lead to entirely new therapies.”
In addition, he said, although prevention of CLL still remains elusive, “it is conceivable that some of the mutations found in this paper occur early in the CLL trajectory, perhaps even before the disease is presented clinically.”
The study was funded by the National Institutes of Health and the Broad/IBM Cancer Resistance Research Project. Dr. Knisbacher and several other authors disclose that they are inventors on a patent related to CLL. Several authors report various relationships with industry. Dr. Greenberger has no disclosures.
Released in a report in Nature Genetics, the map has doubled the number of genetic traits linked the disease from around 100 to 202, lead author Binyamin A. Knisbacher, PhD, a postdoctoral fellow at the Broad Institute of MIT and Harvard Medical Schoo, Boston, said in an interview.
“It also delineated the molecular landscape of the two immunoglobulin gene (IGHV) subtypes, refined CLL subtyping, and built richer genetic prognostic models,” he said.
According to Dr. Knisbacher, CLL “has been at the forefront of genomic discovery,” and research has shown that there’s a wide variety of somatic mutations that drive CLL initiation across the patient population. However, as many as 10% of cases don’t appear to be driven by any known genetic variation, he said, and there’s a need to identify more subtypes and “build richer prognostic models of patient survival” based on genetics and multiomics such as genomics, transcriptomics, and epigenomics.
For the new study, researchers analyzed RNA and DNA from 1,095 patients with CLL and 54 patients with monoclonal B cell lymphocytosis and built what they say is the largest CLL dataset in existence. It’s twice the size of previous datasets, Dr. Knisbacher said.
“We found that RNA expression data was extremely informative for characterizing CLL,” Dr. Knisbacher said. “The RNA expression subtypes refined the ‘classic’ two IGHV subtypes. It is well documented that patients with U-CLL (IGHV-unmutated CLL) have substantially worse clinical outcome in comparison to M-CLL patients (IGHV-mutated CLLs). We found that M-CLLs that have RNA expression profiles similar to U-CLLs have worse survival than M-CLLs with a typical expression profile. Failure-free survival was 50% shorter – 5.3 versus 10.7 years median failure-free survival.”
In addition, he said, “U-CLLs with expression similar to M-CLLs had better survival than U-CLLs with an RNA expression profile typical to U-CLLs.”
The researchers have made their molecular map publicly available at https://cllmap.org/. Researchers can use it “to discover more about each subtype of CLL, and these future studies can help to improve clinical prognosis for the benefit of the patient,” Dr. Knisbacher said.
The study authors added that “this molecular foundation may allow for better prediction of response to therapy or provide the basis for rational combination of novel agents.”
Lee Greenberger, PhD, chief science officer of the Leukemia & Lymphoma Society, said in an interview that the study “provides foundational data further subtyping CLL patients and outcomes. It identifies new targets for therapy or diagnostic predictions in the future. This type of foundational work has proven invaluable in the development of new medicines for cancer in general.”
While there are many medications that have improved therapeutic outcomes in CLL, he added, “cures – or life-long disease control –remain elusive for many patients. Therefore, new molecular insights are needed that could personalize therapies or even lead to entirely new therapies.”
In addition, he said, although prevention of CLL still remains elusive, “it is conceivable that some of the mutations found in this paper occur early in the CLL trajectory, perhaps even before the disease is presented clinically.”
The study was funded by the National Institutes of Health and the Broad/IBM Cancer Resistance Research Project. Dr. Knisbacher and several other authors disclose that they are inventors on a patent related to CLL. Several authors report various relationships with industry. Dr. Greenberger has no disclosures.
Released in a report in Nature Genetics, the map has doubled the number of genetic traits linked the disease from around 100 to 202, lead author Binyamin A. Knisbacher, PhD, a postdoctoral fellow at the Broad Institute of MIT and Harvard Medical Schoo, Boston, said in an interview.
“It also delineated the molecular landscape of the two immunoglobulin gene (IGHV) subtypes, refined CLL subtyping, and built richer genetic prognostic models,” he said.
According to Dr. Knisbacher, CLL “has been at the forefront of genomic discovery,” and research has shown that there’s a wide variety of somatic mutations that drive CLL initiation across the patient population. However, as many as 10% of cases don’t appear to be driven by any known genetic variation, he said, and there’s a need to identify more subtypes and “build richer prognostic models of patient survival” based on genetics and multiomics such as genomics, transcriptomics, and epigenomics.
For the new study, researchers analyzed RNA and DNA from 1,095 patients with CLL and 54 patients with monoclonal B cell lymphocytosis and built what they say is the largest CLL dataset in existence. It’s twice the size of previous datasets, Dr. Knisbacher said.
“We found that RNA expression data was extremely informative for characterizing CLL,” Dr. Knisbacher said. “The RNA expression subtypes refined the ‘classic’ two IGHV subtypes. It is well documented that patients with U-CLL (IGHV-unmutated CLL) have substantially worse clinical outcome in comparison to M-CLL patients (IGHV-mutated CLLs). We found that M-CLLs that have RNA expression profiles similar to U-CLLs have worse survival than M-CLLs with a typical expression profile. Failure-free survival was 50% shorter – 5.3 versus 10.7 years median failure-free survival.”
In addition, he said, “U-CLLs with expression similar to M-CLLs had better survival than U-CLLs with an RNA expression profile typical to U-CLLs.”
The researchers have made their molecular map publicly available at https://cllmap.org/. Researchers can use it “to discover more about each subtype of CLL, and these future studies can help to improve clinical prognosis for the benefit of the patient,” Dr. Knisbacher said.
The study authors added that “this molecular foundation may allow for better prediction of response to therapy or provide the basis for rational combination of novel agents.”
Lee Greenberger, PhD, chief science officer of the Leukemia & Lymphoma Society, said in an interview that the study “provides foundational data further subtyping CLL patients and outcomes. It identifies new targets for therapy or diagnostic predictions in the future. This type of foundational work has proven invaluable in the development of new medicines for cancer in general.”
While there are many medications that have improved therapeutic outcomes in CLL, he added, “cures – or life-long disease control –remain elusive for many patients. Therefore, new molecular insights are needed that could personalize therapies or even lead to entirely new therapies.”
In addition, he said, although prevention of CLL still remains elusive, “it is conceivable that some of the mutations found in this paper occur early in the CLL trajectory, perhaps even before the disease is presented clinically.”
The study was funded by the National Institutes of Health and the Broad/IBM Cancer Resistance Research Project. Dr. Knisbacher and several other authors disclose that they are inventors on a patent related to CLL. Several authors report various relationships with industry. Dr. Greenberger has no disclosures.
FROM NATURE GENETICS
Young children with leukemia are outliving teens
“Outcomes are improving. However, additional efforts, support, and resources are needed to further improve short- and long-term survival for acute leukemia survivors. Targeted efforts focused on populations that face greater disparities in their survival are needed to move the needle faster,” Michael Roth, MD, codirector of the Adolescent and Young Adult Oncology Program at the University of Texas M.D. Anderson Cancer Center, said in an interview.
In one study, released in The Lancet Child & Adolescent Health, an international team of researchers tracked survival outcomes from various types of leukemia in 61 nations. The study focused on the years 2000-2014 and followed patients aged 0-24.
“Age-standardized 5-year net survival in children, adolescents, and young adults for all leukemias combined during 2010-14 varied widely, ranging from 46% in Mexico to more than 85% in Canada, Cyprus, Belgium, Denmark, Finland, and Australia,” the researchers wrote. “Throughout 2000-14, survival from all leukemias combined remained consistently higher for children than adolescents and young adults, and minimal improvement was seen for adolescents and young adults in most countries.”
The U.S. data came from 41 states that cover 86% of the nation’s population, lead author Naomi Ssenyonga, a research fellow at London School of Hygiene & Tropical Medicine, said in an interview.
The 5-year survival rate for acute lymphoid leukemia (ALL) rose from 80% during 2000-2004 to 86% during 2010-2014. Survival in patients with acute myeloid leukemia (AML) was lower than for other subtypes: 66% in 2010-2014 vs. 57% in 2000-2004.
In regard to all leukemias, “we noted a steady increase in the U.S. of 6 percentage points in 5-year survival, up from 77% for patients diagnosed during 2000-2004 to 83% for those diagnosed during 2010-2014,” Ms. Ssenyonga said. “The gains were largely driven by the improvements seen among children.”
Why haven’t adolescents and young adults gained as much ground in survival?
“They often have unique clinical needs,” Ms. Ssenyonga said. “Over the past few years, adolescents and young adults with leukemia in some parts of the world, including the U.S., have increasingly been treated under pediatric protocols. This has led to higher survival. However, this approach has not been adopted consistently, and survival for adolescents and young adults with leukemia is still generally lower than survival for children.”
Gwen Nichols, MD, chief medical officer of the Leukemia & Lymphoma Society, agreed that pediatric treatment protocols hold promise as treatments for young adults. However, “because we arbitrarily set an age cutoff for being an adult, many of these patients are treated by an adult [nonpediatric] hematologist/oncologist, and some patients in the 20-39 age group do not receive the more intensive treatment regimens given to children,” she said in an interview.
In another study, published in Cancer Epidemiology, Biomarkers, & Prevention, M.D. Anderson Cancer Center’s Dr. Roth and colleagues tracked 1,938 patients with ALL and 2,350 with AML who were diagnosed at ages 15-39 from 1980 to 2009. All lived at least 5 years after diagnosis. In both groups, about 58% were White, and most of the rest were Hispanic. The median age of diagnosis for ALL was 23 (range: 15-39) and 28 years for AML (range: 15-39).
“For ALL, 10-year survival for those diagnosed in the 1980s, 1990s, and 2000s was 83%, 88%, and 88%, respectively,” the researchers reported. “Ten-year survival for AML was 82%, 90%, and 90% for those diagnosed in the 1980s, 1990s, and 2000s, respectively.”
“Early mortality within 10 years of diagnosis was mostly secondary to leukemia progressing or recurring. We believe that later mortality is secondary to the development of late side effects from their cancer treatment,” Dr. Roth said.
He noted that many adolescents and young adults with ALL or AML receive stem-cell transplants. “This treatment approach is effective. However, it is associated with short- and long-term toxicity that impacts patients’ health for many years after treatment.”
Indeed, up to 80% of acute leukemia survivors have significant health complications after therapy, said the Leukemia & Lymphoma Society’s Dr. Nichols, who wasn’t surprised by the findings. According to the society, “even when treatments are effective, more than 70% of childhood cancer survivors have a chronic health condition and 42% have a severe, disabling or life-threatening condition 30 years after diagnosis.”
“It would be interesting to understand the male predominance better,” she added, noting that the study found that male patients had worse long-term survival than females (survival time ratio: 0.61, 95% confidence interval, 0.45-0.82). “While it is tempting to suggest it is due to difference in cardiac disease, I am not aware of data to support why there is this survival difference.”
What’s next? “In ALL, we now have a number of new modalities to treat high-risk and relapsed disease such as antibodies and CAR-T,” Dr. Nichols said. “We anticipate that 5-year survival can improve utilizing these modalities due to getting more patients into remission, hopefully while reducing chemotherapeutic toxicity.”
Dr. Nichol’s also highlighted the society’s new genomic-led Pediatric Acute Leukemia (PedAL) Master Clinical Trial, which began enrolling children with acute leukemia in the United States and Canada this year, in an effort to transform medicine’s traditional high-level chemotherapy strategy to their care. The project was launched in collaboration with the National Cancer Institute, Children’s Oncology Group, and the European Pediatric Acute Leukemia Foundation.
As part of the screening process, the biology of each child’s cancer will be identified, and families will be encouraged to enroll them in appropriate targeted therapy trials.
“Until we are able to decrease the toxicity of leukemia regimens, we won’t see a dramatic shift in late effects and thus in morbidity and mortality,” Dr. Nichols said. “The trial is an effort to test newer, less toxic regimens to begin to change that cycle.”
The 5-year survival study was funded by Children with Cancer UK, Institut National du Cancer, La Ligue Contre le Cancer, Centers for Disease Control and Prevention, Swiss Re, Swiss Cancer Research foundation, Swiss Cancer League, Rossy Family Foundation, National Cancer Institute, and the American Cancer Society. One author reports a grant from Macmillan Cancer Support, consultancy fees from Pfizer, and unsolicited small gifts from Moondance Cancer Initiative for philanthropic work. The other authors report no disclosures.
The long-term survival study was funded by the National Cancer Institute, the Archer Foundation and LyondellBasell Industries. Dr. Roth reports no disclosures; other authors report various disclosures. Dr. Nichols reports no disclosures.
“Outcomes are improving. However, additional efforts, support, and resources are needed to further improve short- and long-term survival for acute leukemia survivors. Targeted efforts focused on populations that face greater disparities in their survival are needed to move the needle faster,” Michael Roth, MD, codirector of the Adolescent and Young Adult Oncology Program at the University of Texas M.D. Anderson Cancer Center, said in an interview.
In one study, released in The Lancet Child & Adolescent Health, an international team of researchers tracked survival outcomes from various types of leukemia in 61 nations. The study focused on the years 2000-2014 and followed patients aged 0-24.
“Age-standardized 5-year net survival in children, adolescents, and young adults for all leukemias combined during 2010-14 varied widely, ranging from 46% in Mexico to more than 85% in Canada, Cyprus, Belgium, Denmark, Finland, and Australia,” the researchers wrote. “Throughout 2000-14, survival from all leukemias combined remained consistently higher for children than adolescents and young adults, and minimal improvement was seen for adolescents and young adults in most countries.”
The U.S. data came from 41 states that cover 86% of the nation’s population, lead author Naomi Ssenyonga, a research fellow at London School of Hygiene & Tropical Medicine, said in an interview.
The 5-year survival rate for acute lymphoid leukemia (ALL) rose from 80% during 2000-2004 to 86% during 2010-2014. Survival in patients with acute myeloid leukemia (AML) was lower than for other subtypes: 66% in 2010-2014 vs. 57% in 2000-2004.
In regard to all leukemias, “we noted a steady increase in the U.S. of 6 percentage points in 5-year survival, up from 77% for patients diagnosed during 2000-2004 to 83% for those diagnosed during 2010-2014,” Ms. Ssenyonga said. “The gains were largely driven by the improvements seen among children.”
Why haven’t adolescents and young adults gained as much ground in survival?
“They often have unique clinical needs,” Ms. Ssenyonga said. “Over the past few years, adolescents and young adults with leukemia in some parts of the world, including the U.S., have increasingly been treated under pediatric protocols. This has led to higher survival. However, this approach has not been adopted consistently, and survival for adolescents and young adults with leukemia is still generally lower than survival for children.”
Gwen Nichols, MD, chief medical officer of the Leukemia & Lymphoma Society, agreed that pediatric treatment protocols hold promise as treatments for young adults. However, “because we arbitrarily set an age cutoff for being an adult, many of these patients are treated by an adult [nonpediatric] hematologist/oncologist, and some patients in the 20-39 age group do not receive the more intensive treatment regimens given to children,” she said in an interview.
In another study, published in Cancer Epidemiology, Biomarkers, & Prevention, M.D. Anderson Cancer Center’s Dr. Roth and colleagues tracked 1,938 patients with ALL and 2,350 with AML who were diagnosed at ages 15-39 from 1980 to 2009. All lived at least 5 years after diagnosis. In both groups, about 58% were White, and most of the rest were Hispanic. The median age of diagnosis for ALL was 23 (range: 15-39) and 28 years for AML (range: 15-39).
“For ALL, 10-year survival for those diagnosed in the 1980s, 1990s, and 2000s was 83%, 88%, and 88%, respectively,” the researchers reported. “Ten-year survival for AML was 82%, 90%, and 90% for those diagnosed in the 1980s, 1990s, and 2000s, respectively.”
“Early mortality within 10 years of diagnosis was mostly secondary to leukemia progressing or recurring. We believe that later mortality is secondary to the development of late side effects from their cancer treatment,” Dr. Roth said.
He noted that many adolescents and young adults with ALL or AML receive stem-cell transplants. “This treatment approach is effective. However, it is associated with short- and long-term toxicity that impacts patients’ health for many years after treatment.”
Indeed, up to 80% of acute leukemia survivors have significant health complications after therapy, said the Leukemia & Lymphoma Society’s Dr. Nichols, who wasn’t surprised by the findings. According to the society, “even when treatments are effective, more than 70% of childhood cancer survivors have a chronic health condition and 42% have a severe, disabling or life-threatening condition 30 years after diagnosis.”
“It would be interesting to understand the male predominance better,” she added, noting that the study found that male patients had worse long-term survival than females (survival time ratio: 0.61, 95% confidence interval, 0.45-0.82). “While it is tempting to suggest it is due to difference in cardiac disease, I am not aware of data to support why there is this survival difference.”
What’s next? “In ALL, we now have a number of new modalities to treat high-risk and relapsed disease such as antibodies and CAR-T,” Dr. Nichols said. “We anticipate that 5-year survival can improve utilizing these modalities due to getting more patients into remission, hopefully while reducing chemotherapeutic toxicity.”
Dr. Nichol’s also highlighted the society’s new genomic-led Pediatric Acute Leukemia (PedAL) Master Clinical Trial, which began enrolling children with acute leukemia in the United States and Canada this year, in an effort to transform medicine’s traditional high-level chemotherapy strategy to their care. The project was launched in collaboration with the National Cancer Institute, Children’s Oncology Group, and the European Pediatric Acute Leukemia Foundation.
As part of the screening process, the biology of each child’s cancer will be identified, and families will be encouraged to enroll them in appropriate targeted therapy trials.
“Until we are able to decrease the toxicity of leukemia regimens, we won’t see a dramatic shift in late effects and thus in morbidity and mortality,” Dr. Nichols said. “The trial is an effort to test newer, less toxic regimens to begin to change that cycle.”
The 5-year survival study was funded by Children with Cancer UK, Institut National du Cancer, La Ligue Contre le Cancer, Centers for Disease Control and Prevention, Swiss Re, Swiss Cancer Research foundation, Swiss Cancer League, Rossy Family Foundation, National Cancer Institute, and the American Cancer Society. One author reports a grant from Macmillan Cancer Support, consultancy fees from Pfizer, and unsolicited small gifts from Moondance Cancer Initiative for philanthropic work. The other authors report no disclosures.
The long-term survival study was funded by the National Cancer Institute, the Archer Foundation and LyondellBasell Industries. Dr. Roth reports no disclosures; other authors report various disclosures. Dr. Nichols reports no disclosures.
“Outcomes are improving. However, additional efforts, support, and resources are needed to further improve short- and long-term survival for acute leukemia survivors. Targeted efforts focused on populations that face greater disparities in their survival are needed to move the needle faster,” Michael Roth, MD, codirector of the Adolescent and Young Adult Oncology Program at the University of Texas M.D. Anderson Cancer Center, said in an interview.
In one study, released in The Lancet Child & Adolescent Health, an international team of researchers tracked survival outcomes from various types of leukemia in 61 nations. The study focused on the years 2000-2014 and followed patients aged 0-24.
“Age-standardized 5-year net survival in children, adolescents, and young adults for all leukemias combined during 2010-14 varied widely, ranging from 46% in Mexico to more than 85% in Canada, Cyprus, Belgium, Denmark, Finland, and Australia,” the researchers wrote. “Throughout 2000-14, survival from all leukemias combined remained consistently higher for children than adolescents and young adults, and minimal improvement was seen for adolescents and young adults in most countries.”
The U.S. data came from 41 states that cover 86% of the nation’s population, lead author Naomi Ssenyonga, a research fellow at London School of Hygiene & Tropical Medicine, said in an interview.
The 5-year survival rate for acute lymphoid leukemia (ALL) rose from 80% during 2000-2004 to 86% during 2010-2014. Survival in patients with acute myeloid leukemia (AML) was lower than for other subtypes: 66% in 2010-2014 vs. 57% in 2000-2004.
In regard to all leukemias, “we noted a steady increase in the U.S. of 6 percentage points in 5-year survival, up from 77% for patients diagnosed during 2000-2004 to 83% for those diagnosed during 2010-2014,” Ms. Ssenyonga said. “The gains were largely driven by the improvements seen among children.”
Why haven’t adolescents and young adults gained as much ground in survival?
“They often have unique clinical needs,” Ms. Ssenyonga said. “Over the past few years, adolescents and young adults with leukemia in some parts of the world, including the U.S., have increasingly been treated under pediatric protocols. This has led to higher survival. However, this approach has not been adopted consistently, and survival for adolescents and young adults with leukemia is still generally lower than survival for children.”
Gwen Nichols, MD, chief medical officer of the Leukemia & Lymphoma Society, agreed that pediatric treatment protocols hold promise as treatments for young adults. However, “because we arbitrarily set an age cutoff for being an adult, many of these patients are treated by an adult [nonpediatric] hematologist/oncologist, and some patients in the 20-39 age group do not receive the more intensive treatment regimens given to children,” she said in an interview.
In another study, published in Cancer Epidemiology, Biomarkers, & Prevention, M.D. Anderson Cancer Center’s Dr. Roth and colleagues tracked 1,938 patients with ALL and 2,350 with AML who were diagnosed at ages 15-39 from 1980 to 2009. All lived at least 5 years after diagnosis. In both groups, about 58% were White, and most of the rest were Hispanic. The median age of diagnosis for ALL was 23 (range: 15-39) and 28 years for AML (range: 15-39).
“For ALL, 10-year survival for those diagnosed in the 1980s, 1990s, and 2000s was 83%, 88%, and 88%, respectively,” the researchers reported. “Ten-year survival for AML was 82%, 90%, and 90% for those diagnosed in the 1980s, 1990s, and 2000s, respectively.”
“Early mortality within 10 years of diagnosis was mostly secondary to leukemia progressing or recurring. We believe that later mortality is secondary to the development of late side effects from their cancer treatment,” Dr. Roth said.
He noted that many adolescents and young adults with ALL or AML receive stem-cell transplants. “This treatment approach is effective. However, it is associated with short- and long-term toxicity that impacts patients’ health for many years after treatment.”
Indeed, up to 80% of acute leukemia survivors have significant health complications after therapy, said the Leukemia & Lymphoma Society’s Dr. Nichols, who wasn’t surprised by the findings. According to the society, “even when treatments are effective, more than 70% of childhood cancer survivors have a chronic health condition and 42% have a severe, disabling or life-threatening condition 30 years after diagnosis.”
“It would be interesting to understand the male predominance better,” she added, noting that the study found that male patients had worse long-term survival than females (survival time ratio: 0.61, 95% confidence interval, 0.45-0.82). “While it is tempting to suggest it is due to difference in cardiac disease, I am not aware of data to support why there is this survival difference.”
What’s next? “In ALL, we now have a number of new modalities to treat high-risk and relapsed disease such as antibodies and CAR-T,” Dr. Nichols said. “We anticipate that 5-year survival can improve utilizing these modalities due to getting more patients into remission, hopefully while reducing chemotherapeutic toxicity.”
Dr. Nichol’s also highlighted the society’s new genomic-led Pediatric Acute Leukemia (PedAL) Master Clinical Trial, which began enrolling children with acute leukemia in the United States and Canada this year, in an effort to transform medicine’s traditional high-level chemotherapy strategy to their care. The project was launched in collaboration with the National Cancer Institute, Children’s Oncology Group, and the European Pediatric Acute Leukemia Foundation.
As part of the screening process, the biology of each child’s cancer will be identified, and families will be encouraged to enroll them in appropriate targeted therapy trials.
“Until we are able to decrease the toxicity of leukemia regimens, we won’t see a dramatic shift in late effects and thus in morbidity and mortality,” Dr. Nichols said. “The trial is an effort to test newer, less toxic regimens to begin to change that cycle.”
The 5-year survival study was funded by Children with Cancer UK, Institut National du Cancer, La Ligue Contre le Cancer, Centers for Disease Control and Prevention, Swiss Re, Swiss Cancer Research foundation, Swiss Cancer League, Rossy Family Foundation, National Cancer Institute, and the American Cancer Society. One author reports a grant from Macmillan Cancer Support, consultancy fees from Pfizer, and unsolicited small gifts from Moondance Cancer Initiative for philanthropic work. The other authors report no disclosures.
The long-term survival study was funded by the National Cancer Institute, the Archer Foundation and LyondellBasell Industries. Dr. Roth reports no disclosures; other authors report various disclosures. Dr. Nichols reports no disclosures.
New international dermatology registry tracks monkeypox cases
The American Academy of Dermatology and the International League of Dermatological Societies (ILDS) have created a new registry that now accepts reports from health care providers worldwide about monkeypox cases and monkeypox vaccine reactions.
Patient data such as names and dates of birth will not be collected.
“As with our joint COVID-19 registry, we will be doing real-time data analysis during the outbreak,” dermatologist Esther Freeman, MD, PhD, director of MGH Global Health Dermatology at Massachusetts General Hospital, Boston, and a member of the AAD’s monkeypox task force, said in an interview. “We will to try to feed information back to our front line in terms of clinical characteristics of cases, morphology, and any unexpected findings.”
According to Dr. Freeman, the principal investigator for the COVID-19 registry, this registry has allowed the quick gathering of information about dermatologic findings of COVID-19 from over 53 countries. “We have published over 15 papers, and we share data with outside investigators wishing to do their own analysis of registry-related data,” she said. “Our most-cited paper on COVID vaccine skin reactions has been cited almost 500 times since 2021. It has been used to educate the public on vaccine side effects and to combat vaccine hesitancy.”
The monkeypox registry “doesn’t belong to any one group or person,” Dr. Freeman said. “The idea with rapid data analysis is to be able to give back to the dermatologic community what is hard for us to see with any single case: Patterns and new findings that can be helpful to share with dermatologists and other physicians worldwide, all working together to stop an outbreak.”
The American Academy of Dermatology and the International League of Dermatological Societies (ILDS) have created a new registry that now accepts reports from health care providers worldwide about monkeypox cases and monkeypox vaccine reactions.
Patient data such as names and dates of birth will not be collected.
“As with our joint COVID-19 registry, we will be doing real-time data analysis during the outbreak,” dermatologist Esther Freeman, MD, PhD, director of MGH Global Health Dermatology at Massachusetts General Hospital, Boston, and a member of the AAD’s monkeypox task force, said in an interview. “We will to try to feed information back to our front line in terms of clinical characteristics of cases, morphology, and any unexpected findings.”
According to Dr. Freeman, the principal investigator for the COVID-19 registry, this registry has allowed the quick gathering of information about dermatologic findings of COVID-19 from over 53 countries. “We have published over 15 papers, and we share data with outside investigators wishing to do their own analysis of registry-related data,” she said. “Our most-cited paper on COVID vaccine skin reactions has been cited almost 500 times since 2021. It has been used to educate the public on vaccine side effects and to combat vaccine hesitancy.”
The monkeypox registry “doesn’t belong to any one group or person,” Dr. Freeman said. “The idea with rapid data analysis is to be able to give back to the dermatologic community what is hard for us to see with any single case: Patterns and new findings that can be helpful to share with dermatologists and other physicians worldwide, all working together to stop an outbreak.”
The American Academy of Dermatology and the International League of Dermatological Societies (ILDS) have created a new registry that now accepts reports from health care providers worldwide about monkeypox cases and monkeypox vaccine reactions.
Patient data such as names and dates of birth will not be collected.
“As with our joint COVID-19 registry, we will be doing real-time data analysis during the outbreak,” dermatologist Esther Freeman, MD, PhD, director of MGH Global Health Dermatology at Massachusetts General Hospital, Boston, and a member of the AAD’s monkeypox task force, said in an interview. “We will to try to feed information back to our front line in terms of clinical characteristics of cases, morphology, and any unexpected findings.”
According to Dr. Freeman, the principal investigator for the COVID-19 registry, this registry has allowed the quick gathering of information about dermatologic findings of COVID-19 from over 53 countries. “We have published over 15 papers, and we share data with outside investigators wishing to do their own analysis of registry-related data,” she said. “Our most-cited paper on COVID vaccine skin reactions has been cited almost 500 times since 2021. It has been used to educate the public on vaccine side effects and to combat vaccine hesitancy.”
The monkeypox registry “doesn’t belong to any one group or person,” Dr. Freeman said. “The idea with rapid data analysis is to be able to give back to the dermatologic community what is hard for us to see with any single case: Patterns and new findings that can be helpful to share with dermatologists and other physicians worldwide, all working together to stop an outbreak.”