User login
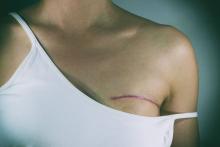
Lumpectomy plus reconstruction outperforms mastectomy plus reconstruction
LAS VEGAS – Lumpectomy plus oncoplastic surgery has a total cost that is comparable to lumpectomy alone and that is less than mastectomy plus reconstructive surgery, according to an analysis of nearly 40,000 women.
While the complication rate was slightly higher than lumpectomy alone, the difference disappeared in the last year of data, possibly because surgical techniques had improved. The study looked at MarketScan Commercial Claims and Encounters Database data from 2000 to 2011 and confirms findings from an earlier study that looked at data from the MD Anderson Cancer Center (Ann Surg Oncol. 2016 Oct;23[10]:3190-8).
Lumpectomy plus oncoplastic surgery is increasingly being performed, but it still only represented about 2% of surgeries in women with invasive epithelial breast cancer, although this percentage had grown to 8.4% by 2011 and is likely higher now, according to Dr. Hwang, an associate professor of breast surgical oncology and surgical oncology at the University of Texas MD Anderson Cancer Center, Houston.
Candidates for lumpectomy plus oncoplastic surgery include those with large volume disease, multifocal/centric disease, poorly located tumors, or macromastia. “If oncoplastic reconstruction was not available, these patients would oftentimes be undergoing total mastectomy with reconstruction,” Dr. Hwang said.
The study included records from 39,518 women who underwent breast surgery with or without reconstruction. It excluded patients who received postmastectomy radiation or neoadjuvant chemotherapy.
A total of 40% underwent lumpectomy plus whole breast irradiation (BCT), 2% received BCT plus oncoplastic reconstruction (BCT+R), 30% had total mastectomy (TM), and 29% had total mastectomy plus reconstruction (TM+R).
After adjusting for age, race, comorbidity, chemotherapy, axillary surgery, and nodal positivity, the complication rate was lowest in the TM group (25%; relative risk compared with BCT+R, 0.71; 95% confidence interval, 0.64-0.78; P less than .001), followed by BCT (29%; RR, 0.80; 95% CI, 0.72-0.88; P less than .001), BCT+R (37%), and TM+R (54%; RR, 1.49; 95% CI, 1.35-1.64; P less than .001).
The rate of complications in BCT+R fell over time, so that, by 2011, it was only slightly higher than those of BCT alone (31.5% vs 29.4%). That trend is “probably just additional experience with the technique,” Dr. Hwang said.
Total costs for TM+R was $89,187, compared with $48,767 for TM, $66,217 for BCT, and $69,781 for BCT+R. The cost difference between BCT and TM+R was about $23,000.
The findings are encouraging, said Judy Boughey, MD, professor of surgery at the Mayo Clinic, Rochester, Minn., who moderated the session. However, she called for caution in interpreting the results because the population of interest made up just 2% of the overall sample. “I think you’re going to have the widest variability of confidence intervals around that group,” Dr. Boughey said. “It’s eye-opening, but I would just view it with caution.”
Dr. Hwang and Dr. Boughey reported having no financial disclosures.
LAS VEGAS – Lumpectomy plus oncoplastic surgery has a total cost that is comparable to lumpectomy alone and that is less than mastectomy plus reconstructive surgery, according to an analysis of nearly 40,000 women.
While the complication rate was slightly higher than lumpectomy alone, the difference disappeared in the last year of data, possibly because surgical techniques had improved. The study looked at MarketScan Commercial Claims and Encounters Database data from 2000 to 2011 and confirms findings from an earlier study that looked at data from the MD Anderson Cancer Center (Ann Surg Oncol. 2016 Oct;23[10]:3190-8).
Lumpectomy plus oncoplastic surgery is increasingly being performed, but it still only represented about 2% of surgeries in women with invasive epithelial breast cancer, although this percentage had grown to 8.4% by 2011 and is likely higher now, according to Dr. Hwang, an associate professor of breast surgical oncology and surgical oncology at the University of Texas MD Anderson Cancer Center, Houston.
Candidates for lumpectomy plus oncoplastic surgery include those with large volume disease, multifocal/centric disease, poorly located tumors, or macromastia. “If oncoplastic reconstruction was not available, these patients would oftentimes be undergoing total mastectomy with reconstruction,” Dr. Hwang said.
The study included records from 39,518 women who underwent breast surgery with or without reconstruction. It excluded patients who received postmastectomy radiation or neoadjuvant chemotherapy.
A total of 40% underwent lumpectomy plus whole breast irradiation (BCT), 2% received BCT plus oncoplastic reconstruction (BCT+R), 30% had total mastectomy (TM), and 29% had total mastectomy plus reconstruction (TM+R).
After adjusting for age, race, comorbidity, chemotherapy, axillary surgery, and nodal positivity, the complication rate was lowest in the TM group (25%; relative risk compared with BCT+R, 0.71; 95% confidence interval, 0.64-0.78; P less than .001), followed by BCT (29%; RR, 0.80; 95% CI, 0.72-0.88; P less than .001), BCT+R (37%), and TM+R (54%; RR, 1.49; 95% CI, 1.35-1.64; P less than .001).
The rate of complications in BCT+R fell over time, so that, by 2011, it was only slightly higher than those of BCT alone (31.5% vs 29.4%). That trend is “probably just additional experience with the technique,” Dr. Hwang said.
Total costs for TM+R was $89,187, compared with $48,767 for TM, $66,217 for BCT, and $69,781 for BCT+R. The cost difference between BCT and TM+R was about $23,000.
The findings are encouraging, said Judy Boughey, MD, professor of surgery at the Mayo Clinic, Rochester, Minn., who moderated the session. However, she called for caution in interpreting the results because the population of interest made up just 2% of the overall sample. “I think you’re going to have the widest variability of confidence intervals around that group,” Dr. Boughey said. “It’s eye-opening, but I would just view it with caution.”
Dr. Hwang and Dr. Boughey reported having no financial disclosures.
LAS VEGAS – Lumpectomy plus oncoplastic surgery has a total cost that is comparable to lumpectomy alone and that is less than mastectomy plus reconstructive surgery, according to an analysis of nearly 40,000 women.
While the complication rate was slightly higher than lumpectomy alone, the difference disappeared in the last year of data, possibly because surgical techniques had improved. The study looked at MarketScan Commercial Claims and Encounters Database data from 2000 to 2011 and confirms findings from an earlier study that looked at data from the MD Anderson Cancer Center (Ann Surg Oncol. 2016 Oct;23[10]:3190-8).
Lumpectomy plus oncoplastic surgery is increasingly being performed, but it still only represented about 2% of surgeries in women with invasive epithelial breast cancer, although this percentage had grown to 8.4% by 2011 and is likely higher now, according to Dr. Hwang, an associate professor of breast surgical oncology and surgical oncology at the University of Texas MD Anderson Cancer Center, Houston.
Candidates for lumpectomy plus oncoplastic surgery include those with large volume disease, multifocal/centric disease, poorly located tumors, or macromastia. “If oncoplastic reconstruction was not available, these patients would oftentimes be undergoing total mastectomy with reconstruction,” Dr. Hwang said.
The study included records from 39,518 women who underwent breast surgery with or without reconstruction. It excluded patients who received postmastectomy radiation or neoadjuvant chemotherapy.
A total of 40% underwent lumpectomy plus whole breast irradiation (BCT), 2% received BCT plus oncoplastic reconstruction (BCT+R), 30% had total mastectomy (TM), and 29% had total mastectomy plus reconstruction (TM+R).
After adjusting for age, race, comorbidity, chemotherapy, axillary surgery, and nodal positivity, the complication rate was lowest in the TM group (25%; relative risk compared with BCT+R, 0.71; 95% confidence interval, 0.64-0.78; P less than .001), followed by BCT (29%; RR, 0.80; 95% CI, 0.72-0.88; P less than .001), BCT+R (37%), and TM+R (54%; RR, 1.49; 95% CI, 1.35-1.64; P less than .001).
The rate of complications in BCT+R fell over time, so that, by 2011, it was only slightly higher than those of BCT alone (31.5% vs 29.4%). That trend is “probably just additional experience with the technique,” Dr. Hwang said.
Total costs for TM+R was $89,187, compared with $48,767 for TM, $66,217 for BCT, and $69,781 for BCT+R. The cost difference between BCT and TM+R was about $23,000.
The findings are encouraging, said Judy Boughey, MD, professor of surgery at the Mayo Clinic, Rochester, Minn., who moderated the session. However, she called for caution in interpreting the results because the population of interest made up just 2% of the overall sample. “I think you’re going to have the widest variability of confidence intervals around that group,” Dr. Boughey said. “It’s eye-opening, but I would just view it with caution.”
Dr. Hwang and Dr. Boughey reported having no financial disclosures.
At ASBS 2017
Key clinical point:
Major finding: Mastectomy with reconstruction had a 49% higher complication rate and $20,000 higher cost, compared with lumpectomy plus reconstruction.
Data source: Nationwide retrospective analysis of 39,518 women.
Disclosures: Dr. Hwang and Dr. Boughey reported having no financial disclosures.
ASCO updates NSCLC guidelines for adjuvant therapy
Adjuvant cisplatin-based chemotherapy is recommended for routine use in non–small cell lung cancer patients with stage IIA, IIB, or IIIA disease who have undergone complete surgical resections, according to updated guidelines from the American Society of Clinical Oncology.
In patients with stage IA disease, the guidelines recommend against cisplatin-based chemotherapy. The new guidelines are based on a systematic review current to January 2016 and an American Society for Radiation Oncology guideline and systematic review, which ASCO had already endorsed, which formed the basis for recommendations on adjuvant radiation therapy, Mark G. Kris, MD, and panel authors said (J Clin Oncol. 2017 April 26. doi: 10.1200/JCO.2017.72.4401).
The guidelines, available online, also recommend against routine cisplatin-based chemotherapy in patients with stage IB disease, but they suggest an evaluation of these patients to explore the pros and cons of adjuvant chemotherapy.
With respect to radiation, the authors recommend against it for patients with resected stage I or stage II disease, and they recommend against it for routine use in patients with stage IIIA. However, in patients with IIIA N2 disease, patients should be evaluated postoperatively, in consultation with a medical oncologist, for the potential risks and benefits of adjuvant radiation.
The authors also provide some recommendations for communicating with patients regarding treatment decisions. There are few studies examining this question, so the recommendations are based on low-quality evidence.
Non–small cell lung cancer patients may have complex social, psychological, and medical issues, and discussions should be carried out with this in mind. Pain, impaired breathing, and fatigue are common after surgery, and smoking cessation can lead to short-term stress as a result of nicotine withdrawal. Older patients may have a range of comorbidities.
Studies show that patients are most satisfied if they feel that physicians allow them to share in the decision making, and if they are given sufficient time to choose. To that end, the authors recommend a session dedicated to discussion of adjuvant therapy.
Communicating risk of death is challenging for many reasons. Patients should be asked how they would like to hear about their risks: some prefer general terms, while others may opt for numbers, charts, or graphs. The guidelines include a risk chart that can help patients understand the potential benefits and risks of chemotherapy.
The study authors report financial relationships with numerous pharmaceutical companies.
Adjuvant cisplatin-based chemotherapy is recommended for routine use in non–small cell lung cancer patients with stage IIA, IIB, or IIIA disease who have undergone complete surgical resections, according to updated guidelines from the American Society of Clinical Oncology.
In patients with stage IA disease, the guidelines recommend against cisplatin-based chemotherapy. The new guidelines are based on a systematic review current to January 2016 and an American Society for Radiation Oncology guideline and systematic review, which ASCO had already endorsed, which formed the basis for recommendations on adjuvant radiation therapy, Mark G. Kris, MD, and panel authors said (J Clin Oncol. 2017 April 26. doi: 10.1200/JCO.2017.72.4401).
The guidelines, available online, also recommend against routine cisplatin-based chemotherapy in patients with stage IB disease, but they suggest an evaluation of these patients to explore the pros and cons of adjuvant chemotherapy.
With respect to radiation, the authors recommend against it for patients with resected stage I or stage II disease, and they recommend against it for routine use in patients with stage IIIA. However, in patients with IIIA N2 disease, patients should be evaluated postoperatively, in consultation with a medical oncologist, for the potential risks and benefits of adjuvant radiation.
The authors also provide some recommendations for communicating with patients regarding treatment decisions. There are few studies examining this question, so the recommendations are based on low-quality evidence.
Non–small cell lung cancer patients may have complex social, psychological, and medical issues, and discussions should be carried out with this in mind. Pain, impaired breathing, and fatigue are common after surgery, and smoking cessation can lead to short-term stress as a result of nicotine withdrawal. Older patients may have a range of comorbidities.
Studies show that patients are most satisfied if they feel that physicians allow them to share in the decision making, and if they are given sufficient time to choose. To that end, the authors recommend a session dedicated to discussion of adjuvant therapy.
Communicating risk of death is challenging for many reasons. Patients should be asked how they would like to hear about their risks: some prefer general terms, while others may opt for numbers, charts, or graphs. The guidelines include a risk chart that can help patients understand the potential benefits and risks of chemotherapy.
The study authors report financial relationships with numerous pharmaceutical companies.
Adjuvant cisplatin-based chemotherapy is recommended for routine use in non–small cell lung cancer patients with stage IIA, IIB, or IIIA disease who have undergone complete surgical resections, according to updated guidelines from the American Society of Clinical Oncology.
In patients with stage IA disease, the guidelines recommend against cisplatin-based chemotherapy. The new guidelines are based on a systematic review current to January 2016 and an American Society for Radiation Oncology guideline and systematic review, which ASCO had already endorsed, which formed the basis for recommendations on adjuvant radiation therapy, Mark G. Kris, MD, and panel authors said (J Clin Oncol. 2017 April 26. doi: 10.1200/JCO.2017.72.4401).
The guidelines, available online, also recommend against routine cisplatin-based chemotherapy in patients with stage IB disease, but they suggest an evaluation of these patients to explore the pros and cons of adjuvant chemotherapy.
With respect to radiation, the authors recommend against it for patients with resected stage I or stage II disease, and they recommend against it for routine use in patients with stage IIIA. However, in patients with IIIA N2 disease, patients should be evaluated postoperatively, in consultation with a medical oncologist, for the potential risks and benefits of adjuvant radiation.
The authors also provide some recommendations for communicating with patients regarding treatment decisions. There are few studies examining this question, so the recommendations are based on low-quality evidence.
Non–small cell lung cancer patients may have complex social, psychological, and medical issues, and discussions should be carried out with this in mind. Pain, impaired breathing, and fatigue are common after surgery, and smoking cessation can lead to short-term stress as a result of nicotine withdrawal. Older patients may have a range of comorbidities.
Studies show that patients are most satisfied if they feel that physicians allow them to share in the decision making, and if they are given sufficient time to choose. To that end, the authors recommend a session dedicated to discussion of adjuvant therapy.
Communicating risk of death is challenging for many reasons. Patients should be asked how they would like to hear about their risks: some prefer general terms, while others may opt for numbers, charts, or graphs. The guidelines include a risk chart that can help patients understand the potential benefits and risks of chemotherapy.
The study authors report financial relationships with numerous pharmaceutical companies.
Pharmacist-run PrEP clinic proves effective, profitable
SEATTLE – A pharmacist-run HIV pre-exposure prophylaxis (PrEP) clinic had a retention rate of 75% and achieved a financial return within the first year of operation.
The Seattle-area project allowed individuals to leave their first appointment with medication in hand, and it drew in many men who had no primary care provider.
The approach is a departure from typical PrEP assignment, in which a patient must navigate a provider, lab testing, a pharmacist, and the need for prior authorization, explained Elyse Tung-Wisner, PharmD, director of clinical services at Kelley-Ross Pharmacy, Seattle. That process can take a few days to a few weeks.
“For years, pharmacists have done glucose testing. A finger stick for an HIV test is a similar model. We applied the same protocol to PrEP,” said Dr. Tung-Wisner, who presented an analysis of the program in a poster session at the Conference on Retroviruses & Opportunistic Infections in partnership with the International Antiviral Society.
In the program, patients can come in, receive counseling, undergo all tests, and the pharmacist can work through all the prior authorizations and patient-assistance programs. “Oftentimes, a patient can leave with medication in hand within an hour,” said Dr. Tung-Wisner.
“As far as we know, we’re the first in the country to have a pharmacist-run HIV PrEP clinic in a community pharmacy setting,” she added.
She and her colleagues presented data on the effectiveness and financial viability of what they term One-Step PrEP. In the program, pharmacists take a medical and sexual history of each patient, perform a risk assessment and laboratory testing, provide education, and prescribe and dispense PrEP if appropriate. The pharmacist also performs all guideline-recommended follow-up care.
From March 2015 through March 2016, 373 patients inquired about PrEP services. A total of 251 patients were evaluated in person and 245 (98%) went on to begin PrEP. Among those who started PrEP, 88% identified as men who have sex with men (mean age, 34 years; range, 18-64).
The program had a 75% retention rate over the first year, with one HIV seroconversion.
The initial start-up costs were recouped at 9 months, suggesting that the program quickly became financially sustainable.
It also reached a highly vulnerable, underserved population. The average men who have sex with men index score was 20, which indicates that the patients were high risk, Dr. Tung-Wisner noted.
And just 23% of the patients who were evaluated in person had a primary care provider. “That suggests we were accessing a patient population that has not already established care anywhere else,” she explained.
The program was run by Kelley-Ross Pharmacye. Dr. Tung-Wisner is an employee of the pharmacy.
SEATTLE – A pharmacist-run HIV pre-exposure prophylaxis (PrEP) clinic had a retention rate of 75% and achieved a financial return within the first year of operation.
The Seattle-area project allowed individuals to leave their first appointment with medication in hand, and it drew in many men who had no primary care provider.
The approach is a departure from typical PrEP assignment, in which a patient must navigate a provider, lab testing, a pharmacist, and the need for prior authorization, explained Elyse Tung-Wisner, PharmD, director of clinical services at Kelley-Ross Pharmacy, Seattle. That process can take a few days to a few weeks.
“For years, pharmacists have done glucose testing. A finger stick for an HIV test is a similar model. We applied the same protocol to PrEP,” said Dr. Tung-Wisner, who presented an analysis of the program in a poster session at the Conference on Retroviruses & Opportunistic Infections in partnership with the International Antiviral Society.
In the program, patients can come in, receive counseling, undergo all tests, and the pharmacist can work through all the prior authorizations and patient-assistance programs. “Oftentimes, a patient can leave with medication in hand within an hour,” said Dr. Tung-Wisner.
“As far as we know, we’re the first in the country to have a pharmacist-run HIV PrEP clinic in a community pharmacy setting,” she added.
She and her colleagues presented data on the effectiveness and financial viability of what they term One-Step PrEP. In the program, pharmacists take a medical and sexual history of each patient, perform a risk assessment and laboratory testing, provide education, and prescribe and dispense PrEP if appropriate. The pharmacist also performs all guideline-recommended follow-up care.
From March 2015 through March 2016, 373 patients inquired about PrEP services. A total of 251 patients were evaluated in person and 245 (98%) went on to begin PrEP. Among those who started PrEP, 88% identified as men who have sex with men (mean age, 34 years; range, 18-64).
The program had a 75% retention rate over the first year, with one HIV seroconversion.
The initial start-up costs were recouped at 9 months, suggesting that the program quickly became financially sustainable.
It also reached a highly vulnerable, underserved population. The average men who have sex with men index score was 20, which indicates that the patients were high risk, Dr. Tung-Wisner noted.
And just 23% of the patients who were evaluated in person had a primary care provider. “That suggests we were accessing a patient population that has not already established care anywhere else,” she explained.
The program was run by Kelley-Ross Pharmacye. Dr. Tung-Wisner is an employee of the pharmacy.
SEATTLE – A pharmacist-run HIV pre-exposure prophylaxis (PrEP) clinic had a retention rate of 75% and achieved a financial return within the first year of operation.
The Seattle-area project allowed individuals to leave their first appointment with medication in hand, and it drew in many men who had no primary care provider.
The approach is a departure from typical PrEP assignment, in which a patient must navigate a provider, lab testing, a pharmacist, and the need for prior authorization, explained Elyse Tung-Wisner, PharmD, director of clinical services at Kelley-Ross Pharmacy, Seattle. That process can take a few days to a few weeks.
“For years, pharmacists have done glucose testing. A finger stick for an HIV test is a similar model. We applied the same protocol to PrEP,” said Dr. Tung-Wisner, who presented an analysis of the program in a poster session at the Conference on Retroviruses & Opportunistic Infections in partnership with the International Antiviral Society.
In the program, patients can come in, receive counseling, undergo all tests, and the pharmacist can work through all the prior authorizations and patient-assistance programs. “Oftentimes, a patient can leave with medication in hand within an hour,” said Dr. Tung-Wisner.
“As far as we know, we’re the first in the country to have a pharmacist-run HIV PrEP clinic in a community pharmacy setting,” she added.
She and her colleagues presented data on the effectiveness and financial viability of what they term One-Step PrEP. In the program, pharmacists take a medical and sexual history of each patient, perform a risk assessment and laboratory testing, provide education, and prescribe and dispense PrEP if appropriate. The pharmacist also performs all guideline-recommended follow-up care.
From March 2015 through March 2016, 373 patients inquired about PrEP services. A total of 251 patients were evaluated in person and 245 (98%) went on to begin PrEP. Among those who started PrEP, 88% identified as men who have sex with men (mean age, 34 years; range, 18-64).
The program had a 75% retention rate over the first year, with one HIV seroconversion.
The initial start-up costs were recouped at 9 months, suggesting that the program quickly became financially sustainable.
It also reached a highly vulnerable, underserved population. The average men who have sex with men index score was 20, which indicates that the patients were high risk, Dr. Tung-Wisner noted.
And just 23% of the patients who were evaluated in person had a primary care provider. “That suggests we were accessing a patient population that has not already established care anywhere else,” she explained.
The program was run by Kelley-Ross Pharmacye. Dr. Tung-Wisner is an employee of the pharmacy.
Key clinical point: A pharmacist-run HIV pre-exposure prophylaxis (PrEP) clinic had a retention rate of 75% and achieved a financial return within the first year of operation.
Major finding: The retention rate was 75%, and only 23% of patients had a primary care physician.
Data source: A retrospective analysis of a program that served 251 patients.
Disclosures: The program was run by Kelley-Ross Pharmacy in Seattle. Dr. Tung-Wisner is an employee of the pharmacy.
Postsurgical O2 may cut AHI events
Postoperative oxygen therapy in patients with previously undetected obstructive sleep apnea (OSA) led to a reduction in apnea-hypopnea index (AHI) events per hour with no increase in apnea-hypopnea event duration.
The results suggest that postoperative oxygen could be useful in patients with OSA who refuse continuous positive airway pressure (CPAP) therapy, those with newly diagnosed OSA, and those with suspected OSA.
The researchers set out to determine if postoperative oxygen therapy could improve oxygenation in patients with previously undiagnosed OSA, reasoning that the intervention could reduce adverse events.
The study, published in CHEST (2017 March;151[3]:597-611), provided generally good news, but with a caveat: “Essentially we are saying, yes, if you give supplemental oxygen, you improve oxygenation of the patient. But overall we have to be careful because a significant number of patients have significant carbon dioxide retention when receiving supplemental oxygen. So we have to monitor patients – not just oxygen, but we may have to monitor carbon dioxide levels, too,” said lead study author Frances Chung, MBBS, professor of anesthesiology at the University of Toronto and Toronto Western Hospital.
The researchers randomized 123 patients with an AHI of at least five events per hour to postoperative oxygen (3 L/min for 3 nights via nasal prongs) or no postoperative oxygen.
On the third night, the oxygen group had a higher average oxygen saturation than controls (95.2% plus or minus 3.2% vs. 91.4% plus or minus 3.5%; P less than .0001) and a lower oxygen desaturation index (median, 2.3 events per hour vs. median, 18.5; P less than .0001).
A lower number of AHI events per hour occurred in the oxygen group (median, 8.0) than in the control group (median, 15.6; P = .016).
On average, the longest apnea-hypopnea event (median, 33.8 seconds) was shorter for a patient on oxygen, compared with a patient who did not receive oxygen (median, 49.6 seconds; P = .002).
But one finding surprised the researchers and led to some concern: Across both groups, 11.4% of patients experienced substantial CO2 retention. Specifically, for at least 10% of 1 of the nights, these patients had a partial pressure of CO2 of at least 55 mm Hg, according to measurements taken with a transcutaneous CO2 monitor. Of the 14 patients who experienced this event, 13 were receiving oxygen.
Dr. Chung said the results argue strongly for postsurgical oxygen in patients with OSA, who are known to be at increased risk for complications. “We are not doing something about it, and we should be doing something. Because one death from a complication is too many,” she said.
The study was funded by the University Health Network Foundation, Toronto, and the University of Toronto. Dr. Chung reported receiving research grant support from Ontario Ministry of Health Innovation Grant, University Health Network Foundation, ResMed Foundation, Acacia, and Medtronic.
Postoperative oxygen therapy in patients with previously undetected obstructive sleep apnea (OSA) led to a reduction in apnea-hypopnea index (AHI) events per hour with no increase in apnea-hypopnea event duration.
The results suggest that postoperative oxygen could be useful in patients with OSA who refuse continuous positive airway pressure (CPAP) therapy, those with newly diagnosed OSA, and those with suspected OSA.
The researchers set out to determine if postoperative oxygen therapy could improve oxygenation in patients with previously undiagnosed OSA, reasoning that the intervention could reduce adverse events.
The study, published in CHEST (2017 March;151[3]:597-611), provided generally good news, but with a caveat: “Essentially we are saying, yes, if you give supplemental oxygen, you improve oxygenation of the patient. But overall we have to be careful because a significant number of patients have significant carbon dioxide retention when receiving supplemental oxygen. So we have to monitor patients – not just oxygen, but we may have to monitor carbon dioxide levels, too,” said lead study author Frances Chung, MBBS, professor of anesthesiology at the University of Toronto and Toronto Western Hospital.
The researchers randomized 123 patients with an AHI of at least five events per hour to postoperative oxygen (3 L/min for 3 nights via nasal prongs) or no postoperative oxygen.
On the third night, the oxygen group had a higher average oxygen saturation than controls (95.2% plus or minus 3.2% vs. 91.4% plus or minus 3.5%; P less than .0001) and a lower oxygen desaturation index (median, 2.3 events per hour vs. median, 18.5; P less than .0001).
A lower number of AHI events per hour occurred in the oxygen group (median, 8.0) than in the control group (median, 15.6; P = .016).
On average, the longest apnea-hypopnea event (median, 33.8 seconds) was shorter for a patient on oxygen, compared with a patient who did not receive oxygen (median, 49.6 seconds; P = .002).
But one finding surprised the researchers and led to some concern: Across both groups, 11.4% of patients experienced substantial CO2 retention. Specifically, for at least 10% of 1 of the nights, these patients had a partial pressure of CO2 of at least 55 mm Hg, according to measurements taken with a transcutaneous CO2 monitor. Of the 14 patients who experienced this event, 13 were receiving oxygen.
Dr. Chung said the results argue strongly for postsurgical oxygen in patients with OSA, who are known to be at increased risk for complications. “We are not doing something about it, and we should be doing something. Because one death from a complication is too many,” she said.
The study was funded by the University Health Network Foundation, Toronto, and the University of Toronto. Dr. Chung reported receiving research grant support from Ontario Ministry of Health Innovation Grant, University Health Network Foundation, ResMed Foundation, Acacia, and Medtronic.
Postoperative oxygen therapy in patients with previously undetected obstructive sleep apnea (OSA) led to a reduction in apnea-hypopnea index (AHI) events per hour with no increase in apnea-hypopnea event duration.
The results suggest that postoperative oxygen could be useful in patients with OSA who refuse continuous positive airway pressure (CPAP) therapy, those with newly diagnosed OSA, and those with suspected OSA.
The researchers set out to determine if postoperative oxygen therapy could improve oxygenation in patients with previously undiagnosed OSA, reasoning that the intervention could reduce adverse events.
The study, published in CHEST (2017 March;151[3]:597-611), provided generally good news, but with a caveat: “Essentially we are saying, yes, if you give supplemental oxygen, you improve oxygenation of the patient. But overall we have to be careful because a significant number of patients have significant carbon dioxide retention when receiving supplemental oxygen. So we have to monitor patients – not just oxygen, but we may have to monitor carbon dioxide levels, too,” said lead study author Frances Chung, MBBS, professor of anesthesiology at the University of Toronto and Toronto Western Hospital.
The researchers randomized 123 patients with an AHI of at least five events per hour to postoperative oxygen (3 L/min for 3 nights via nasal prongs) or no postoperative oxygen.
On the third night, the oxygen group had a higher average oxygen saturation than controls (95.2% plus or minus 3.2% vs. 91.4% plus or minus 3.5%; P less than .0001) and a lower oxygen desaturation index (median, 2.3 events per hour vs. median, 18.5; P less than .0001).
A lower number of AHI events per hour occurred in the oxygen group (median, 8.0) than in the control group (median, 15.6; P = .016).
On average, the longest apnea-hypopnea event (median, 33.8 seconds) was shorter for a patient on oxygen, compared with a patient who did not receive oxygen (median, 49.6 seconds; P = .002).
But one finding surprised the researchers and led to some concern: Across both groups, 11.4% of patients experienced substantial CO2 retention. Specifically, for at least 10% of 1 of the nights, these patients had a partial pressure of CO2 of at least 55 mm Hg, according to measurements taken with a transcutaneous CO2 monitor. Of the 14 patients who experienced this event, 13 were receiving oxygen.
Dr. Chung said the results argue strongly for postsurgical oxygen in patients with OSA, who are known to be at increased risk for complications. “We are not doing something about it, and we should be doing something. Because one death from a complication is too many,” she said.
The study was funded by the University Health Network Foundation, Toronto, and the University of Toronto. Dr. Chung reported receiving research grant support from Ontario Ministry of Health Innovation Grant, University Health Network Foundation, ResMed Foundation, Acacia, and Medtronic.
FROM CHEST
Key clinical point: Postsurgical oxygen may reduce complications in patients with obstructive sleep apnea.
Major finding: For patients receiving oxygen, the longest apnea-hypopnea event duration was 33.8 seconds versus 49.6 for controls.
Data source: A randomized, placebo-controlled study of 123 patients.
Disclosures: The study was funded by the University Health Network Foundation, Toronto, and the University of Toronto. Dr. Chung reported receiving research grant support from various entities.
BUN increase tracks with upper GI bleeding outcomes
In patients with acute nonvariceal upper GI bleeding (UGIB), increased blood urea nitrogen (BUN) levels at 24 hours were associated with worse outcomes. The marker, already proven useful in acute pancreatitis, could help physicians determine a patient’s prognosis.
Existing measures of UGIB risk are effective, but only about 30% of physicians ever calculate risk scores when evaluating UGIB patients, perhaps because they require measurements at multiple time points. “We personally think the reason for this is the busyness of clinical practices, especially the acute nature of upper GI bleeding. It’s often hard to step back to calculate a score that has multiple variables,” said study author Navin Kumar, MD, a fellow in gastroenterology at Brigham and Women’s Hospital, Boston.
The study was published in Gastrointestinal Endoscopy (2017. doi: 10.1016/j.gie.2017.03.1533).
Like acute pancreatitis, upper GI bleeding requires resuscitation management, which suggested that BUN levels might be a useful marker in this condition as well. To find out, the researchers analyzed data from 357 patients who were treated at the Brigham and Women’s Hospital emergency department and ultimately hospitalized for UGIB during 2004-2014.
The researchers analyzed BUN levels measured at admission and at the time closest to 24 hours after hospitalization, which ranged from 6 hours to 48 hours.
Thirty-seven patients (10%) experienced an increase in BUN level, while all the rest had levels that stayed steady or decreased. Those patients with BUN increases had a lower mean Glasgow-Blatchford score (7.8 vs. 9.6; P =.010), but there was no difference in AIMS65 scores.
Patients with BUN increases had greater odds of the composite outcome, which included inpatient death from any cause, inpatient rebleeding, a need for surgical or radiologic intervention, and/or a need for endoscopic reintervention during hospitalization (22% vs. 9%; P =.014). Inpatient mortality was higher in the increased BUN group (8% vs. 1%; P =.004).
Overall, BUN increase at 24 hours was associated with an odds ratio of 2.75 for the composite outcome (95% confidence interval, 1.13-6.70; P = .026).
A potential limitation to using the BUN is that it could just be catching patients with underlying renal disease. But when researchers adjusted for this, the odds ratio for increased BUN remained significant (OR, 3.00; P =.021).
“The nice part of the study is that it’s so easy to interpret and apply in a clinical setting. You just need two data points: BUN at presentation and at 24 hours. If the BUN level has risen, you need to have a higher degree of suspicion for the prognosis of those patients,” said Dr. Kumar.
The downside to BUN is that it doesn’t provide information for the first 24 hours. For that reason, BUN shouldn’t replace measures like the Glasgow-Blatchford score and the AIMS65 score. “But it’s very helpful to use this change in BUN score to get a sense of where the patient is trending. If it’s rising, there’s a higher risk of worse outcomes, and this could influence decisions about whether the patient should be in the ICU or the medical ward,” said Dr. Kumar.
In patients with acute nonvariceal upper GI bleeding (UGIB), increased blood urea nitrogen (BUN) levels at 24 hours were associated with worse outcomes. The marker, already proven useful in acute pancreatitis, could help physicians determine a patient’s prognosis.
Existing measures of UGIB risk are effective, but only about 30% of physicians ever calculate risk scores when evaluating UGIB patients, perhaps because they require measurements at multiple time points. “We personally think the reason for this is the busyness of clinical practices, especially the acute nature of upper GI bleeding. It’s often hard to step back to calculate a score that has multiple variables,” said study author Navin Kumar, MD, a fellow in gastroenterology at Brigham and Women’s Hospital, Boston.
The study was published in Gastrointestinal Endoscopy (2017. doi: 10.1016/j.gie.2017.03.1533).
Like acute pancreatitis, upper GI bleeding requires resuscitation management, which suggested that BUN levels might be a useful marker in this condition as well. To find out, the researchers analyzed data from 357 patients who were treated at the Brigham and Women’s Hospital emergency department and ultimately hospitalized for UGIB during 2004-2014.
The researchers analyzed BUN levels measured at admission and at the time closest to 24 hours after hospitalization, which ranged from 6 hours to 48 hours.
Thirty-seven patients (10%) experienced an increase in BUN level, while all the rest had levels that stayed steady or decreased. Those patients with BUN increases had a lower mean Glasgow-Blatchford score (7.8 vs. 9.6; P =.010), but there was no difference in AIMS65 scores.
Patients with BUN increases had greater odds of the composite outcome, which included inpatient death from any cause, inpatient rebleeding, a need for surgical or radiologic intervention, and/or a need for endoscopic reintervention during hospitalization (22% vs. 9%; P =.014). Inpatient mortality was higher in the increased BUN group (8% vs. 1%; P =.004).
Overall, BUN increase at 24 hours was associated with an odds ratio of 2.75 for the composite outcome (95% confidence interval, 1.13-6.70; P = .026).
A potential limitation to using the BUN is that it could just be catching patients with underlying renal disease. But when researchers adjusted for this, the odds ratio for increased BUN remained significant (OR, 3.00; P =.021).
“The nice part of the study is that it’s so easy to interpret and apply in a clinical setting. You just need two data points: BUN at presentation and at 24 hours. If the BUN level has risen, you need to have a higher degree of suspicion for the prognosis of those patients,” said Dr. Kumar.
The downside to BUN is that it doesn’t provide information for the first 24 hours. For that reason, BUN shouldn’t replace measures like the Glasgow-Blatchford score and the AIMS65 score. “But it’s very helpful to use this change in BUN score to get a sense of where the patient is trending. If it’s rising, there’s a higher risk of worse outcomes, and this could influence decisions about whether the patient should be in the ICU or the medical ward,” said Dr. Kumar.
In patients with acute nonvariceal upper GI bleeding (UGIB), increased blood urea nitrogen (BUN) levels at 24 hours were associated with worse outcomes. The marker, already proven useful in acute pancreatitis, could help physicians determine a patient’s prognosis.
Existing measures of UGIB risk are effective, but only about 30% of physicians ever calculate risk scores when evaluating UGIB patients, perhaps because they require measurements at multiple time points. “We personally think the reason for this is the busyness of clinical practices, especially the acute nature of upper GI bleeding. It’s often hard to step back to calculate a score that has multiple variables,” said study author Navin Kumar, MD, a fellow in gastroenterology at Brigham and Women’s Hospital, Boston.
The study was published in Gastrointestinal Endoscopy (2017. doi: 10.1016/j.gie.2017.03.1533).
Like acute pancreatitis, upper GI bleeding requires resuscitation management, which suggested that BUN levels might be a useful marker in this condition as well. To find out, the researchers analyzed data from 357 patients who were treated at the Brigham and Women’s Hospital emergency department and ultimately hospitalized for UGIB during 2004-2014.
The researchers analyzed BUN levels measured at admission and at the time closest to 24 hours after hospitalization, which ranged from 6 hours to 48 hours.
Thirty-seven patients (10%) experienced an increase in BUN level, while all the rest had levels that stayed steady or decreased. Those patients with BUN increases had a lower mean Glasgow-Blatchford score (7.8 vs. 9.6; P =.010), but there was no difference in AIMS65 scores.
Patients with BUN increases had greater odds of the composite outcome, which included inpatient death from any cause, inpatient rebleeding, a need for surgical or radiologic intervention, and/or a need for endoscopic reintervention during hospitalization (22% vs. 9%; P =.014). Inpatient mortality was higher in the increased BUN group (8% vs. 1%; P =.004).
Overall, BUN increase at 24 hours was associated with an odds ratio of 2.75 for the composite outcome (95% confidence interval, 1.13-6.70; P = .026).
A potential limitation to using the BUN is that it could just be catching patients with underlying renal disease. But when researchers adjusted for this, the odds ratio for increased BUN remained significant (OR, 3.00; P =.021).
“The nice part of the study is that it’s so easy to interpret and apply in a clinical setting. You just need two data points: BUN at presentation and at 24 hours. If the BUN level has risen, you need to have a higher degree of suspicion for the prognosis of those patients,” said Dr. Kumar.
The downside to BUN is that it doesn’t provide information for the first 24 hours. For that reason, BUN shouldn’t replace measures like the Glasgow-Blatchford score and the AIMS65 score. “But it’s very helpful to use this change in BUN score to get a sense of where the patient is trending. If it’s rising, there’s a higher risk of worse outcomes, and this could influence decisions about whether the patient should be in the ICU or the medical ward,” said Dr. Kumar.
FROM GASTROINTESTINAL ENDOSCOPY
Key clinical point: BUN could be a useful prognostic marker.
Major finding: BUN increase indicated a threefold increased risk of poor outcomes.
Data source: Retrospective analysis of 357 patients at a single center.
Disclosures: The study did not receive external funding. Dr. Kumar reported having no financial disclosures.
A viral inducer of celiac disease?
A viral infection may be the culprit behind celiac disease, which is caused by an autoimmune response to dietary gluten. The findings are based on an engineered reovirus, which is normally benign. The researchers believe that a reovirus may disrupt intestinal immune homeostasis in susceptible individuals as a result of infection during childhood.
According to in vitro and mouse studies carried out by the researchers, one strain of reovirus suppresses peripheral regulatory T-cell conversion and promotes T helper 1 immune response at sites that normally induce tolerance to dietary antigens. The work appeared in the April issue of Science (2017;356:44-50).
The researchers decided to investigate reoviruses. They often infect humans, commonly in early childhood when gluten usually is first introduced. They also infect humans and mice similarly, allowing a more straightforward comparison between human and mouse studies than would be possible in other virus types.
The researchers created an engineered virus made from two reovirus strains, T1L and T3D, which naturally reassort in human hosts. T1L infects the intestine, while T3D does not. The new strain, T3D-RV, retains most of the characteristics of T3D but can also infect the intestine.
The researchers then conducted mouse studies and showed that both T1L and T3D-RV affect immune responses to dietary antigens at the inductive and effector sites of oral tolerance. However, the original T1L strain caused more changes in gene transcription, both in the number of genes and the intensity of transcription level. This suggested that T1L might uniquely alter immunogenic responses to dietary antigens.
A further test in mice showed that T1L also prompted a proinflammatory response in dendritic cells that take up ovalbumin, but T3D-RV did not. Furthermore, T1L interfered with induction of peripheral tolerance to oral ovalbumin, and T3D-RV did not.
With this data in hand, the researchers turned to human subjects. They compared 73 healthy controls to 160 patients with celiac disease who were on a gluten-free diet. Celiac disease patients had higher mean antireovirus antibody titers, though the result fell short of statistical significance (P = .06), and subjects with celiac disease were over-represented among subjects who had antireovirus titers above the median value.
“You can have two viruses of the same family infecting the intestine in the same way, inducing protective immunity, and being cleared, but only one sets the stage for disease. Finally, using these two viruses allows [us] to dissociate protective immunity from immunopathology. Only the virus that has the capacity to enter the site where dietary proteins are seen by the immune system can trigger disease,” said Bana Jabri, MD, PhD, professor of medicine at the University of Chicago.
Reovirus is unlikely to be the only, otherwise harmless, virus that could prompt wayward immune responses. The research points the way to the identification of viruses linked to celiac disease and other autoimmune diseases and could inform vaccine strategies to prevent such conditions.
The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
A viral infection may be the culprit behind celiac disease, which is caused by an autoimmune response to dietary gluten. The findings are based on an engineered reovirus, which is normally benign. The researchers believe that a reovirus may disrupt intestinal immune homeostasis in susceptible individuals as a result of infection during childhood.
According to in vitro and mouse studies carried out by the researchers, one strain of reovirus suppresses peripheral regulatory T-cell conversion and promotes T helper 1 immune response at sites that normally induce tolerance to dietary antigens. The work appeared in the April issue of Science (2017;356:44-50).
The researchers decided to investigate reoviruses. They often infect humans, commonly in early childhood when gluten usually is first introduced. They also infect humans and mice similarly, allowing a more straightforward comparison between human and mouse studies than would be possible in other virus types.
The researchers created an engineered virus made from two reovirus strains, T1L and T3D, which naturally reassort in human hosts. T1L infects the intestine, while T3D does not. The new strain, T3D-RV, retains most of the characteristics of T3D but can also infect the intestine.
The researchers then conducted mouse studies and showed that both T1L and T3D-RV affect immune responses to dietary antigens at the inductive and effector sites of oral tolerance. However, the original T1L strain caused more changes in gene transcription, both in the number of genes and the intensity of transcription level. This suggested that T1L might uniquely alter immunogenic responses to dietary antigens.
A further test in mice showed that T1L also prompted a proinflammatory response in dendritic cells that take up ovalbumin, but T3D-RV did not. Furthermore, T1L interfered with induction of peripheral tolerance to oral ovalbumin, and T3D-RV did not.
With this data in hand, the researchers turned to human subjects. They compared 73 healthy controls to 160 patients with celiac disease who were on a gluten-free diet. Celiac disease patients had higher mean antireovirus antibody titers, though the result fell short of statistical significance (P = .06), and subjects with celiac disease were over-represented among subjects who had antireovirus titers above the median value.
“You can have two viruses of the same family infecting the intestine in the same way, inducing protective immunity, and being cleared, but only one sets the stage for disease. Finally, using these two viruses allows [us] to dissociate protective immunity from immunopathology. Only the virus that has the capacity to enter the site where dietary proteins are seen by the immune system can trigger disease,” said Bana Jabri, MD, PhD, professor of medicine at the University of Chicago.
Reovirus is unlikely to be the only, otherwise harmless, virus that could prompt wayward immune responses. The research points the way to the identification of viruses linked to celiac disease and other autoimmune diseases and could inform vaccine strategies to prevent such conditions.
The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
A viral infection may be the culprit behind celiac disease, which is caused by an autoimmune response to dietary gluten. The findings are based on an engineered reovirus, which is normally benign. The researchers believe that a reovirus may disrupt intestinal immune homeostasis in susceptible individuals as a result of infection during childhood.
According to in vitro and mouse studies carried out by the researchers, one strain of reovirus suppresses peripheral regulatory T-cell conversion and promotes T helper 1 immune response at sites that normally induce tolerance to dietary antigens. The work appeared in the April issue of Science (2017;356:44-50).
The researchers decided to investigate reoviruses. They often infect humans, commonly in early childhood when gluten usually is first introduced. They also infect humans and mice similarly, allowing a more straightforward comparison between human and mouse studies than would be possible in other virus types.
The researchers created an engineered virus made from two reovirus strains, T1L and T3D, which naturally reassort in human hosts. T1L infects the intestine, while T3D does not. The new strain, T3D-RV, retains most of the characteristics of T3D but can also infect the intestine.
The researchers then conducted mouse studies and showed that both T1L and T3D-RV affect immune responses to dietary antigens at the inductive and effector sites of oral tolerance. However, the original T1L strain caused more changes in gene transcription, both in the number of genes and the intensity of transcription level. This suggested that T1L might uniquely alter immunogenic responses to dietary antigens.
A further test in mice showed that T1L also prompted a proinflammatory response in dendritic cells that take up ovalbumin, but T3D-RV did not. Furthermore, T1L interfered with induction of peripheral tolerance to oral ovalbumin, and T3D-RV did not.
With this data in hand, the researchers turned to human subjects. They compared 73 healthy controls to 160 patients with celiac disease who were on a gluten-free diet. Celiac disease patients had higher mean antireovirus antibody titers, though the result fell short of statistical significance (P = .06), and subjects with celiac disease were over-represented among subjects who had antireovirus titers above the median value.
“You can have two viruses of the same family infecting the intestine in the same way, inducing protective immunity, and being cleared, but only one sets the stage for disease. Finally, using these two viruses allows [us] to dissociate protective immunity from immunopathology. Only the virus that has the capacity to enter the site where dietary proteins are seen by the immune system can trigger disease,” said Bana Jabri, MD, PhD, professor of medicine at the University of Chicago.
Reovirus is unlikely to be the only, otherwise harmless, virus that could prompt wayward immune responses. The research points the way to the identification of viruses linked to celiac disease and other autoimmune diseases and could inform vaccine strategies to prevent such conditions.
The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
FROM SCIENCE
Key clinical point: Celiac disease patients have high reovirus antibody titers.
Major finding: Researchers detail mechanistic pathway that could explain a viral link.
Data source: In vitro, human, and mouse observational studies.
Disclosures: The study received funding from the National Institutes of Health and the University of Chicago. No conflict of interest information was disclosed in the article.
Choice of protease inhibitor may impact CVD risk in HIV+ patients
SEATTLE – A study of newer generation protease inhibitors (PIs) suggests that ritonavir-boosted atazanavir (ATV/r) has a better cardiovascular safety profile than ritonavir-boosted darunavir (DRV/r). After adjustment for age and other factors, patients taking DRV/r had a higher incidence rate ratio of cardiovascular disease during a 5-year follow-up compared to pre-exposure, while no such increase was seen in patients taking ATV/r.
Older protease inhibitors had been shown to be associated with increased CVD risk, but the newer generation of drugs had not been similarly examined. A previous analysis of the D:A:D (Data collection on Adverse events of Anti-HIV Drugs) study had shown no effect of ATV/r with cardiovascular risk, but the follow-up time was short.
The data suggest that ATV/r may be the best choice for patients at heightened risk of cardiovascular disease. “This is just the results from the first five years, and we need to reassess at some point, but according to what we know now, it does really look like a quite real effect,” Dr Ryom added.
The researchers examined longer-term results from 35,711 (47.8% white, 73.6% men, median age 44) participants in the D:A:D study, who were followed beginning January 1, 2009 through the earliest CVD diagnosis, last visit plus 6 months, or February 1, 2016.
After adjustment for baseline variables potentially on the causal pathway between PI use and cardiovascular disease, the researchers found that, compared to baseline pre-exposure levels, patients taking ATV/r had a 5-year incidence rate ratio (IRR) of CVD of 1.03 (95% confidence interval 0.90-1.18), while those taking DRV/r had an IRR 1.59 (95% CI, 1.33-1.91).
Time-updated adjustment analyses for factors potentially on the causal pathway made no meaningful difference in the calculation of incidence risk ratios. “This suggests that the association we say for boosted darunavir is not moderated by dyslipidemia, which is interesting and in contrast to what we saw in first-generation protease inhibitors,” Dr Ryom said during her talk.
Adjusting for bilirubin levels had no impact on the associations.
There was no data on drug dose, and due to the observational nature of the study, the researchers could not prove a causal association between CVD risk and use of either drug, but the results were convincing enough for Dr Ryom to consider a drug that appears friendlier to the cardiovascular system, particularly in high risk patients, though she also noted that there is evidence that ATV/r could lead to kidney stones and chronic kidney disease. “So it’s very difficult to balance the different risks. You really need to tailor your choice according to the patient that is in front of you,” Dr Ryom said.
The study was funded by The Oversight Committee for the Evaluation of Metabolic Complications of HAART, which included both academic and industry sources, and the Danish National Research Foundation. Dr Ryom reported having no financial disclosures.
SEATTLE – A study of newer generation protease inhibitors (PIs) suggests that ritonavir-boosted atazanavir (ATV/r) has a better cardiovascular safety profile than ritonavir-boosted darunavir (DRV/r). After adjustment for age and other factors, patients taking DRV/r had a higher incidence rate ratio of cardiovascular disease during a 5-year follow-up compared to pre-exposure, while no such increase was seen in patients taking ATV/r.
Older protease inhibitors had been shown to be associated with increased CVD risk, but the newer generation of drugs had not been similarly examined. A previous analysis of the D:A:D (Data collection on Adverse events of Anti-HIV Drugs) study had shown no effect of ATV/r with cardiovascular risk, but the follow-up time was short.
The data suggest that ATV/r may be the best choice for patients at heightened risk of cardiovascular disease. “This is just the results from the first five years, and we need to reassess at some point, but according to what we know now, it does really look like a quite real effect,” Dr Ryom added.
The researchers examined longer-term results from 35,711 (47.8% white, 73.6% men, median age 44) participants in the D:A:D study, who were followed beginning January 1, 2009 through the earliest CVD diagnosis, last visit plus 6 months, or February 1, 2016.
After adjustment for baseline variables potentially on the causal pathway between PI use and cardiovascular disease, the researchers found that, compared to baseline pre-exposure levels, patients taking ATV/r had a 5-year incidence rate ratio (IRR) of CVD of 1.03 (95% confidence interval 0.90-1.18), while those taking DRV/r had an IRR 1.59 (95% CI, 1.33-1.91).
Time-updated adjustment analyses for factors potentially on the causal pathway made no meaningful difference in the calculation of incidence risk ratios. “This suggests that the association we say for boosted darunavir is not moderated by dyslipidemia, which is interesting and in contrast to what we saw in first-generation protease inhibitors,” Dr Ryom said during her talk.
Adjusting for bilirubin levels had no impact on the associations.
There was no data on drug dose, and due to the observational nature of the study, the researchers could not prove a causal association between CVD risk and use of either drug, but the results were convincing enough for Dr Ryom to consider a drug that appears friendlier to the cardiovascular system, particularly in high risk patients, though she also noted that there is evidence that ATV/r could lead to kidney stones and chronic kidney disease. “So it’s very difficult to balance the different risks. You really need to tailor your choice according to the patient that is in front of you,” Dr Ryom said.
The study was funded by The Oversight Committee for the Evaluation of Metabolic Complications of HAART, which included both academic and industry sources, and the Danish National Research Foundation. Dr Ryom reported having no financial disclosures.
SEATTLE – A study of newer generation protease inhibitors (PIs) suggests that ritonavir-boosted atazanavir (ATV/r) has a better cardiovascular safety profile than ritonavir-boosted darunavir (DRV/r). After adjustment for age and other factors, patients taking DRV/r had a higher incidence rate ratio of cardiovascular disease during a 5-year follow-up compared to pre-exposure, while no such increase was seen in patients taking ATV/r.
Older protease inhibitors had been shown to be associated with increased CVD risk, but the newer generation of drugs had not been similarly examined. A previous analysis of the D:A:D (Data collection on Adverse events of Anti-HIV Drugs) study had shown no effect of ATV/r with cardiovascular risk, but the follow-up time was short.
The data suggest that ATV/r may be the best choice for patients at heightened risk of cardiovascular disease. “This is just the results from the first five years, and we need to reassess at some point, but according to what we know now, it does really look like a quite real effect,” Dr Ryom added.
The researchers examined longer-term results from 35,711 (47.8% white, 73.6% men, median age 44) participants in the D:A:D study, who were followed beginning January 1, 2009 through the earliest CVD diagnosis, last visit plus 6 months, or February 1, 2016.
After adjustment for baseline variables potentially on the causal pathway between PI use and cardiovascular disease, the researchers found that, compared to baseline pre-exposure levels, patients taking ATV/r had a 5-year incidence rate ratio (IRR) of CVD of 1.03 (95% confidence interval 0.90-1.18), while those taking DRV/r had an IRR 1.59 (95% CI, 1.33-1.91).
Time-updated adjustment analyses for factors potentially on the causal pathway made no meaningful difference in the calculation of incidence risk ratios. “This suggests that the association we say for boosted darunavir is not moderated by dyslipidemia, which is interesting and in contrast to what we saw in first-generation protease inhibitors,” Dr Ryom said during her talk.
Adjusting for bilirubin levels had no impact on the associations.
There was no data on drug dose, and due to the observational nature of the study, the researchers could not prove a causal association between CVD risk and use of either drug, but the results were convincing enough for Dr Ryom to consider a drug that appears friendlier to the cardiovascular system, particularly in high risk patients, though she also noted that there is evidence that ATV/r could lead to kidney stones and chronic kidney disease. “So it’s very difficult to balance the different risks. You really need to tailor your choice according to the patient that is in front of you,” Dr Ryom said.
The study was funded by The Oversight Committee for the Evaluation of Metabolic Complications of HAART, which included both academic and industry sources, and the Danish National Research Foundation. Dr Ryom reported having no financial disclosures.
AT CROI 2017
Key clinical point: Atazanavir may be safer for HIV-positive patients at high CVD risk.
Major finding: Patients taking darunavir had a 59% higher incidence of cardiovascular disease than those taking atazanavir.
Data source: Retrospective analysis of 35,711 patients.
Disclosures: The study was funded by The Oversight Committee for the Evaluation of Metabolic Complications of HAART, which included both academic and industry sources, and the Danish National Research Foundation. Dr Ryom reported having no financial disclosures.
Rate of heroin use in U.S. soars, especially among white individuals
Rates of heroin use and heroin use disorder rose dramatically between 2001-2002 and 2012-2013, and the trend was greatest among the white population. The rise among white individuals could be tied to the opioid epidemic, because nonmedical opioid also rose disproportionately in that group, according to research published online March 29.
The findings come from an analysis of 43,093 people who responded to the 2001-2002 National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), and 36,309 respondents to the 2012-2013 NESARC-III.
In addition, Dr. Martins and her associates found a significant rise in the number of white heroin users who had started nonmedical prescription opioid (NMPO) use before heroin (35.83% to 52.83%; P =.01). In contrast, the percentage of nonwhite individuals who started off with NMPO use dropped from 44.12% to 26.20% (P = .04).
The increase in heroin use was larger among individuals at less than 100% of the poverty level (0.44% to 2.42%; P less than .001), as well as among people with education levels of less than high school (heroin use, 0.41% to 2.01%; P = .03; heroin use disorder, 0.24% to 0.87%; P = .08) and among those with no more than high school education (heroin use, 0.39% to 2.15%; P =.003; heroin use disorder, 0.29% to 1.11%; P = .003). The absolute values of the findings may be inexact, because the methods of the two surveys differed slightly. In addition, the investigators did not include homeless and incarcerated individuals.
Based on their analysis, Dr. Martins and her associates offered strategies aimed at addressing the crisis. “To curb the heroin epidemic, particularly among younger adults, collective prevention and intervention efforts may be most effective,” they wrote. “Promising examples include expansion of access to medication-assisted treatment (including methadone hydrochloride, buprenorphine hydrochloride, or injectable naltrexone hydrochloride), educational programs in schools and community settings, overdose prevention training in concert with comprehensive naloxone hydrochloride distribution programs, and consistent use of prescription drug–monitoring programs that implement best practices by prescribers.”
NESARC and NESARC-III were funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute on Drug Abuse. The authors received funding from several sources, including the National Institute on Drug Abuse, the New York State Psychiatric Institute, and the J. William Fulbright and the Colciencias doctoral scholarships. One of the study authors, Deborah S. Hasin, PhD, was a principal investigator on a study that was funded by InVentiv Health Consulting, which pool funds from nine pharmaceutical companies.
Opioid misuse can be prevented by the medical community with a change in prescribing practices aimed at limiting the supply of prescription opioids, Bertha K. Madras, PhD, wrote in an accompanying editorial (JAMA Psychiatry. 2017 Mar 29. doi: 10.1001/jamapsychiatry.2017.0163). Also, medical training “should include awareness of the risks posed by high opioid doses, immediate-release formulations, use combined with alcohol and/or benzodiazepines, history of overdoses, and other factors,” she wrote.
The United States has more than 14,000 drug treatment programs, but many are staffed with clinicians who are not licensed. One way to foster comprehensive services would be to develop an integrated medical and behavioral treatment system that would be supervised by a physician and substance abuse specialist. “Resources, training, and workforce issues are a concern, but the benefits of integrated health care and behavioral treatment conceivably outweigh the risks of maintaining the status quo,” she wrote.
Dr. Madras is affiliated with the department of psychiatry at Harvard Medical School in Boston, and McLean Hospital in Belmont, Mass. She also serves on the scientific advisory board of RiverMend Health and consults for Guidepoint.
Opioid misuse can be prevented by the medical community with a change in prescribing practices aimed at limiting the supply of prescription opioids, Bertha K. Madras, PhD, wrote in an accompanying editorial (JAMA Psychiatry. 2017 Mar 29. doi: 10.1001/jamapsychiatry.2017.0163). Also, medical training “should include awareness of the risks posed by high opioid doses, immediate-release formulations, use combined with alcohol and/or benzodiazepines, history of overdoses, and other factors,” she wrote.
The United States has more than 14,000 drug treatment programs, but many are staffed with clinicians who are not licensed. One way to foster comprehensive services would be to develop an integrated medical and behavioral treatment system that would be supervised by a physician and substance abuse specialist. “Resources, training, and workforce issues are a concern, but the benefits of integrated health care and behavioral treatment conceivably outweigh the risks of maintaining the status quo,” she wrote.
Dr. Madras is affiliated with the department of psychiatry at Harvard Medical School in Boston, and McLean Hospital in Belmont, Mass. She also serves on the scientific advisory board of RiverMend Health and consults for Guidepoint.
Opioid misuse can be prevented by the medical community with a change in prescribing practices aimed at limiting the supply of prescription opioids, Bertha K. Madras, PhD, wrote in an accompanying editorial (JAMA Psychiatry. 2017 Mar 29. doi: 10.1001/jamapsychiatry.2017.0163). Also, medical training “should include awareness of the risks posed by high opioid doses, immediate-release formulations, use combined with alcohol and/or benzodiazepines, history of overdoses, and other factors,” she wrote.
The United States has more than 14,000 drug treatment programs, but many are staffed with clinicians who are not licensed. One way to foster comprehensive services would be to develop an integrated medical and behavioral treatment system that would be supervised by a physician and substance abuse specialist. “Resources, training, and workforce issues are a concern, but the benefits of integrated health care and behavioral treatment conceivably outweigh the risks of maintaining the status quo,” she wrote.
Dr. Madras is affiliated with the department of psychiatry at Harvard Medical School in Boston, and McLean Hospital in Belmont, Mass. She also serves on the scientific advisory board of RiverMend Health and consults for Guidepoint.
Rates of heroin use and heroin use disorder rose dramatically between 2001-2002 and 2012-2013, and the trend was greatest among the white population. The rise among white individuals could be tied to the opioid epidemic, because nonmedical opioid also rose disproportionately in that group, according to research published online March 29.
The findings come from an analysis of 43,093 people who responded to the 2001-2002 National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), and 36,309 respondents to the 2012-2013 NESARC-III.
In addition, Dr. Martins and her associates found a significant rise in the number of white heroin users who had started nonmedical prescription opioid (NMPO) use before heroin (35.83% to 52.83%; P =.01). In contrast, the percentage of nonwhite individuals who started off with NMPO use dropped from 44.12% to 26.20% (P = .04).
The increase in heroin use was larger among individuals at less than 100% of the poverty level (0.44% to 2.42%; P less than .001), as well as among people with education levels of less than high school (heroin use, 0.41% to 2.01%; P = .03; heroin use disorder, 0.24% to 0.87%; P = .08) and among those with no more than high school education (heroin use, 0.39% to 2.15%; P =.003; heroin use disorder, 0.29% to 1.11%; P = .003). The absolute values of the findings may be inexact, because the methods of the two surveys differed slightly. In addition, the investigators did not include homeless and incarcerated individuals.
Based on their analysis, Dr. Martins and her associates offered strategies aimed at addressing the crisis. “To curb the heroin epidemic, particularly among younger adults, collective prevention and intervention efforts may be most effective,” they wrote. “Promising examples include expansion of access to medication-assisted treatment (including methadone hydrochloride, buprenorphine hydrochloride, or injectable naltrexone hydrochloride), educational programs in schools and community settings, overdose prevention training in concert with comprehensive naloxone hydrochloride distribution programs, and consistent use of prescription drug–monitoring programs that implement best practices by prescribers.”
NESARC and NESARC-III were funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute on Drug Abuse. The authors received funding from several sources, including the National Institute on Drug Abuse, the New York State Psychiatric Institute, and the J. William Fulbright and the Colciencias doctoral scholarships. One of the study authors, Deborah S. Hasin, PhD, was a principal investigator on a study that was funded by InVentiv Health Consulting, which pool funds from nine pharmaceutical companies.
Rates of heroin use and heroin use disorder rose dramatically between 2001-2002 and 2012-2013, and the trend was greatest among the white population. The rise among white individuals could be tied to the opioid epidemic, because nonmedical opioid also rose disproportionately in that group, according to research published online March 29.
The findings come from an analysis of 43,093 people who responded to the 2001-2002 National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), and 36,309 respondents to the 2012-2013 NESARC-III.
In addition, Dr. Martins and her associates found a significant rise in the number of white heroin users who had started nonmedical prescription opioid (NMPO) use before heroin (35.83% to 52.83%; P =.01). In contrast, the percentage of nonwhite individuals who started off with NMPO use dropped from 44.12% to 26.20% (P = .04).
The increase in heroin use was larger among individuals at less than 100% of the poverty level (0.44% to 2.42%; P less than .001), as well as among people with education levels of less than high school (heroin use, 0.41% to 2.01%; P = .03; heroin use disorder, 0.24% to 0.87%; P = .08) and among those with no more than high school education (heroin use, 0.39% to 2.15%; P =.003; heroin use disorder, 0.29% to 1.11%; P = .003). The absolute values of the findings may be inexact, because the methods of the two surveys differed slightly. In addition, the investigators did not include homeless and incarcerated individuals.
Based on their analysis, Dr. Martins and her associates offered strategies aimed at addressing the crisis. “To curb the heroin epidemic, particularly among younger adults, collective prevention and intervention efforts may be most effective,” they wrote. “Promising examples include expansion of access to medication-assisted treatment (including methadone hydrochloride, buprenorphine hydrochloride, or injectable naltrexone hydrochloride), educational programs in schools and community settings, overdose prevention training in concert with comprehensive naloxone hydrochloride distribution programs, and consistent use of prescription drug–monitoring programs that implement best practices by prescribers.”
NESARC and NESARC-III were funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute on Drug Abuse. The authors received funding from several sources, including the National Institute on Drug Abuse, the New York State Psychiatric Institute, and the J. William Fulbright and the Colciencias doctoral scholarships. One of the study authors, Deborah S. Hasin, PhD, was a principal investigator on a study that was funded by InVentiv Health Consulting, which pool funds from nine pharmaceutical companies.
FROM JAMA PSYCHIATRY
Key clinical point: Campaigns are needed to educate the public about harms tied to heroin use, and access should be expanded to populations at risk for both heroin use and heroin use disorder.
Major finding: Rates of lifetime heroin use rose from 0.33% to 1.61%.
Data source: Retrospective analysis of 43,093 respondents to the 2001-2002 National Epidemiologic Survey on Alcohol and Related Conditions (NESARC), and 36,309 respondents to the 2012-2013 NESARC-III.
Disclosures: NESARC and NESARC-III were funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute on Drug Abuse. The authors received funding from several sources, including the National Institute on Drug Abuse, the New York State Psychiatric Institute, and the J. William Fulbright and the Colciencias doctoral scholarships. One of the study authors, Deborah S. Hasin, PhD, was a principal investigator on a study that was funded by InVentiv Health Consulting, which pools funds from nine pharmaceutical companies.
A little rivaroxaban goes a long way
In patients with venous thromboembolism at equipoise for further anticoagulation therapy, treatment with a 10-mg dose of rivaroxaban (Xarelto) had comparable efficacy to a 20-mg dose, with both leading to fewer recurrences than treatment with aspirin. There were no statistically significant differences in clinically relevant nonmajor bleeding between the three groups.
The study’s conclusions are limited to relatively healthy patients such as the ones who were selected for the study.
The findings were presented at the annual meeting of the American College of Cardiology and published simultaneously in the New England Journal of Medicine (N Engl J Med 2017 March 18. doi: 10.1056/NEJMoa1700518).
Anticoagulants are the primary treatment for prevention of venous thromboembolism recurrences, but the medications are often stopped after 6-12 months because of concerns about bleeding. To counter that issue, physicians may prescribe lower doses of anticoagulants such as rivaroxaban, or substitute aspirin.
The work follows another recent study of apixaban (Eliquis), which showed that a 2.5-mg twice-daily dose performed the same as did a 5.0-mg twice-daily dose in the prevention of venous thromboembolism recurrence (N Engl J Med. 2013;368[8]:699-708).
“I think the story is kind of the same here,” said David Garcia, MD, professor of hematology at the University of Washington, Seattle, who was one of the principal investigators in the trial.
Patients with unprompted venous thromboembolism are increasingly being offered anticoagulant therapy to prevent recurrences. Those drugs have inherent bleeding risks, but the newer drugs and even warfarin are becoming safer. Even so, “as we embark on that, one has to remember that the risk of anticoagulants is cumulative. It may only carry a risk of 1% per year of major hemorrhage, but if the patient has to take it for 10 or 20 or 30 years, it’s a nontrivial risk of major bleeding over that time,” said Dr. Garcia.
The researchers conducted the Reduced-Dose Rivaroxaban in the Long-Term Prevention of Recurrent Symptomatic Venous Thromboembolism (EINSTEIN CHOICE) trial, in which 3,365 patients from 24 sites were randomized to receive 20 mg rivaroxaban, 10 mg rivaroxaban, or 100 mg aspirin for up to 1 year following an initial 6-12 months of treatment with anticoagulation therapy.
During a median follow-up of 1 year, 4.4% of patients on aspirin experienced a recurrence, compared with 1.5% of patients in the 20-mg rivaroxaban group (hazard ratio versus aspirin, 0.34; 95% confidence interval, 0.20-0.59; P less than .001), and 1.2% in the 10-mg rivaroxaban group (HR versus aspirin, 0.26; 95% CI, 0.14-0.47; P less than .001). There was no statistical significance between the two doses of rivaroxaban.
The rates of fatal thromboembolism were similar, at 0.2% in the 20-mg rivaroxaban group, 0% in the 10-mg group, and 0.2% in the aspirin group.
Major bleeding occurred in 0.5% of patients in the 20-mg rivaroxaban group, in 0.4% of the 10-mg rivaroxaban group, and 0.3% in the aspirin group. Nonmajor, clinically relevant bleeding was also similar between groups, at 2.7% in the 20-mg group, 2.0% in the 10-mg group, and 1.8% in the aspirin group. These differences were not statistically significant.
The study is good news for clinicians as they help patients decide whether to undergo preventive therapy. “Even before the newer agents arrived on the market, we had moved the needle a lot in terms of maximizing the safety of warfarin. I think these drugs take it to yet another level,” said Dr. Garcia.
Bayer Pharmaceuticals funded the study. Dr. Garcia has received honoraria from Bristol-Meyers Squibb, Pfizer, and Boehringer Ingelheim.
“Given the protection from recurrent venous thromboembolism afforded by reduced-dose rivaroxaban, extending treatment beyond 3 months could be considered in patients with provoked venous thromboembolism who are at average risk for bleeding and who are strongly averse to having another episode of venous thromboembolism. In light of the safety profile of low-dose rivaroxaban, the benefit of this strategy does not need to be large in order to justify the extension of therapy.
“This trial suggests that it would be helpful to evaluate the effects of reduced doses of rivaroxaban within 6 months after an episode of venous thromboembolism.
“For patients without cancer, the use of direct oral anticoagulant agents might be considered as first-line treatment for those with acute venous thromboembolism. Full-dose treatment could be continued for a minimum of 3-6 months. In patients in whom there is equipoise with respect to continuing anticoagulant therapy beyond this period, the use of a reduced-intensity direct oral anticoagulant agent might be considered. Clinicians who choose this strategy can be confident of excellent efficacy and low bleeding risk similar to that observed with aspirin or placebo” (N Engl J Med 2017 March 18. doi: 10.1056/NEJMe1701628).
Mark Crowther, MD, is professor of medicine at McMaster University, Hamilton, Ont. Adam Cuker is assistant professor of medicine at the hospital of the University of Pennsylvania, Philadelphia.
“Given the protection from recurrent venous thromboembolism afforded by reduced-dose rivaroxaban, extending treatment beyond 3 months could be considered in patients with provoked venous thromboembolism who are at average risk for bleeding and who are strongly averse to having another episode of venous thromboembolism. In light of the safety profile of low-dose rivaroxaban, the benefit of this strategy does not need to be large in order to justify the extension of therapy.
“This trial suggests that it would be helpful to evaluate the effects of reduced doses of rivaroxaban within 6 months after an episode of venous thromboembolism.
“For patients without cancer, the use of direct oral anticoagulant agents might be considered as first-line treatment for those with acute venous thromboembolism. Full-dose treatment could be continued for a minimum of 3-6 months. In patients in whom there is equipoise with respect to continuing anticoagulant therapy beyond this period, the use of a reduced-intensity direct oral anticoagulant agent might be considered. Clinicians who choose this strategy can be confident of excellent efficacy and low bleeding risk similar to that observed with aspirin or placebo” (N Engl J Med 2017 March 18. doi: 10.1056/NEJMe1701628).
Mark Crowther, MD, is professor of medicine at McMaster University, Hamilton, Ont. Adam Cuker is assistant professor of medicine at the hospital of the University of Pennsylvania, Philadelphia.
“Given the protection from recurrent venous thromboembolism afforded by reduced-dose rivaroxaban, extending treatment beyond 3 months could be considered in patients with provoked venous thromboembolism who are at average risk for bleeding and who are strongly averse to having another episode of venous thromboembolism. In light of the safety profile of low-dose rivaroxaban, the benefit of this strategy does not need to be large in order to justify the extension of therapy.
“This trial suggests that it would be helpful to evaluate the effects of reduced doses of rivaroxaban within 6 months after an episode of venous thromboembolism.
“For patients without cancer, the use of direct oral anticoagulant agents might be considered as first-line treatment for those with acute venous thromboembolism. Full-dose treatment could be continued for a minimum of 3-6 months. In patients in whom there is equipoise with respect to continuing anticoagulant therapy beyond this period, the use of a reduced-intensity direct oral anticoagulant agent might be considered. Clinicians who choose this strategy can be confident of excellent efficacy and low bleeding risk similar to that observed with aspirin or placebo” (N Engl J Med 2017 March 18. doi: 10.1056/NEJMe1701628).
Mark Crowther, MD, is professor of medicine at McMaster University, Hamilton, Ont. Adam Cuker is assistant professor of medicine at the hospital of the University of Pennsylvania, Philadelphia.
In patients with venous thromboembolism at equipoise for further anticoagulation therapy, treatment with a 10-mg dose of rivaroxaban (Xarelto) had comparable efficacy to a 20-mg dose, with both leading to fewer recurrences than treatment with aspirin. There were no statistically significant differences in clinically relevant nonmajor bleeding between the three groups.
The study’s conclusions are limited to relatively healthy patients such as the ones who were selected for the study.
The findings were presented at the annual meeting of the American College of Cardiology and published simultaneously in the New England Journal of Medicine (N Engl J Med 2017 March 18. doi: 10.1056/NEJMoa1700518).
Anticoagulants are the primary treatment for prevention of venous thromboembolism recurrences, but the medications are often stopped after 6-12 months because of concerns about bleeding. To counter that issue, physicians may prescribe lower doses of anticoagulants such as rivaroxaban, or substitute aspirin.
The work follows another recent study of apixaban (Eliquis), which showed that a 2.5-mg twice-daily dose performed the same as did a 5.0-mg twice-daily dose in the prevention of venous thromboembolism recurrence (N Engl J Med. 2013;368[8]:699-708).
“I think the story is kind of the same here,” said David Garcia, MD, professor of hematology at the University of Washington, Seattle, who was one of the principal investigators in the trial.
Patients with unprompted venous thromboembolism are increasingly being offered anticoagulant therapy to prevent recurrences. Those drugs have inherent bleeding risks, but the newer drugs and even warfarin are becoming safer. Even so, “as we embark on that, one has to remember that the risk of anticoagulants is cumulative. It may only carry a risk of 1% per year of major hemorrhage, but if the patient has to take it for 10 or 20 or 30 years, it’s a nontrivial risk of major bleeding over that time,” said Dr. Garcia.
The researchers conducted the Reduced-Dose Rivaroxaban in the Long-Term Prevention of Recurrent Symptomatic Venous Thromboembolism (EINSTEIN CHOICE) trial, in which 3,365 patients from 24 sites were randomized to receive 20 mg rivaroxaban, 10 mg rivaroxaban, or 100 mg aspirin for up to 1 year following an initial 6-12 months of treatment with anticoagulation therapy.
During a median follow-up of 1 year, 4.4% of patients on aspirin experienced a recurrence, compared with 1.5% of patients in the 20-mg rivaroxaban group (hazard ratio versus aspirin, 0.34; 95% confidence interval, 0.20-0.59; P less than .001), and 1.2% in the 10-mg rivaroxaban group (HR versus aspirin, 0.26; 95% CI, 0.14-0.47; P less than .001). There was no statistical significance between the two doses of rivaroxaban.
The rates of fatal thromboembolism were similar, at 0.2% in the 20-mg rivaroxaban group, 0% in the 10-mg group, and 0.2% in the aspirin group.
Major bleeding occurred in 0.5% of patients in the 20-mg rivaroxaban group, in 0.4% of the 10-mg rivaroxaban group, and 0.3% in the aspirin group. Nonmajor, clinically relevant bleeding was also similar between groups, at 2.7% in the 20-mg group, 2.0% in the 10-mg group, and 1.8% in the aspirin group. These differences were not statistically significant.
The study is good news for clinicians as they help patients decide whether to undergo preventive therapy. “Even before the newer agents arrived on the market, we had moved the needle a lot in terms of maximizing the safety of warfarin. I think these drugs take it to yet another level,” said Dr. Garcia.
Bayer Pharmaceuticals funded the study. Dr. Garcia has received honoraria from Bristol-Meyers Squibb, Pfizer, and Boehringer Ingelheim.
In patients with venous thromboembolism at equipoise for further anticoagulation therapy, treatment with a 10-mg dose of rivaroxaban (Xarelto) had comparable efficacy to a 20-mg dose, with both leading to fewer recurrences than treatment with aspirin. There were no statistically significant differences in clinically relevant nonmajor bleeding between the three groups.
The study’s conclusions are limited to relatively healthy patients such as the ones who were selected for the study.
The findings were presented at the annual meeting of the American College of Cardiology and published simultaneously in the New England Journal of Medicine (N Engl J Med 2017 March 18. doi: 10.1056/NEJMoa1700518).
Anticoagulants are the primary treatment for prevention of venous thromboembolism recurrences, but the medications are often stopped after 6-12 months because of concerns about bleeding. To counter that issue, physicians may prescribe lower doses of anticoagulants such as rivaroxaban, or substitute aspirin.
The work follows another recent study of apixaban (Eliquis), which showed that a 2.5-mg twice-daily dose performed the same as did a 5.0-mg twice-daily dose in the prevention of venous thromboembolism recurrence (N Engl J Med. 2013;368[8]:699-708).
“I think the story is kind of the same here,” said David Garcia, MD, professor of hematology at the University of Washington, Seattle, who was one of the principal investigators in the trial.
Patients with unprompted venous thromboembolism are increasingly being offered anticoagulant therapy to prevent recurrences. Those drugs have inherent bleeding risks, but the newer drugs and even warfarin are becoming safer. Even so, “as we embark on that, one has to remember that the risk of anticoagulants is cumulative. It may only carry a risk of 1% per year of major hemorrhage, but if the patient has to take it for 10 or 20 or 30 years, it’s a nontrivial risk of major bleeding over that time,” said Dr. Garcia.
The researchers conducted the Reduced-Dose Rivaroxaban in the Long-Term Prevention of Recurrent Symptomatic Venous Thromboembolism (EINSTEIN CHOICE) trial, in which 3,365 patients from 24 sites were randomized to receive 20 mg rivaroxaban, 10 mg rivaroxaban, or 100 mg aspirin for up to 1 year following an initial 6-12 months of treatment with anticoagulation therapy.
During a median follow-up of 1 year, 4.4% of patients on aspirin experienced a recurrence, compared with 1.5% of patients in the 20-mg rivaroxaban group (hazard ratio versus aspirin, 0.34; 95% confidence interval, 0.20-0.59; P less than .001), and 1.2% in the 10-mg rivaroxaban group (HR versus aspirin, 0.26; 95% CI, 0.14-0.47; P less than .001). There was no statistical significance between the two doses of rivaroxaban.
The rates of fatal thromboembolism were similar, at 0.2% in the 20-mg rivaroxaban group, 0% in the 10-mg group, and 0.2% in the aspirin group.
Major bleeding occurred in 0.5% of patients in the 20-mg rivaroxaban group, in 0.4% of the 10-mg rivaroxaban group, and 0.3% in the aspirin group. Nonmajor, clinically relevant bleeding was also similar between groups, at 2.7% in the 20-mg group, 2.0% in the 10-mg group, and 1.8% in the aspirin group. These differences were not statistically significant.
The study is good news for clinicians as they help patients decide whether to undergo preventive therapy. “Even before the newer agents arrived on the market, we had moved the needle a lot in terms of maximizing the safety of warfarin. I think these drugs take it to yet another level,” said Dr. Garcia.
Bayer Pharmaceuticals funded the study. Dr. Garcia has received honoraria from Bristol-Meyers Squibb, Pfizer, and Boehringer Ingelheim.
FROM ACC 17
Key clinical point: In venous thromboembolism prevention, a 10-mg dose matched 20 mg.
Major finding: The recurrence rates were 1.2% at 10 mg versus 4.4% with aspirin.
Data source: Randomized comparison trial of 3,365 patients.
Disclosures: Bayer Pharmaceuticals funded the study. Dr. Garcia has received honoraria from Bristol-Meyers Squibb, Pfizer, and Boehringer Ingelheim.
HCV testing stagnant among baby boomers
Despite the urging of the United States Preventive Services Task Force and other organizations in 2013, the percentage of baby boomers who underwent testing for hepatitis C (HCV) infection had barely changed 2 years later – from 12.3% in 2013 to 13.8% in 2015.
The numbers are particularly troubling because new and improved antiviral drugs offer cures that could forestall liver cancer, cirrhosis, and other potential complications, with shorter regimens and fewer side effects than older regimens.
Other reactions were more forceful. “Kind of pathetic, isn’t it?” said John D. Scott, MD, assistant director of the Hepatitis and Liver Clinic at Harborview Medical Center, and an associate professor of medicine at the University of Washington, Seattle.
The researchers analyzed 2013 and 2015 data from the National Health Interview Survey, which included records for 21,827 baby boomers with HCV testing data.
The slight increase overall of 12.3% to 13.8% was small but also statistically significant (P = .013). Some populations fared better: Compared with the privately insured, those with Medicare plus Medicaid were more likely to have been tested (prevalence ratio, 1.83; 95% confidence interval, 1.32-2.53), as were those only on Medicaid (PR, 1.35; 95% CI, 1.04-1.76), and those with military insurance (PR, 1.62; 95% CI, 1.16-2.26).
The study could be subject to recall bias, since it relied on participants’ self-reports.
The authors speculate that the higher prevalence of testing in those with military insurance may reflect efforts by the Veterans Health Administration to reduce the high prevalence of HCV-associated disease among veterans.
It’s entirely possible to increase testing rates, according to Dr. Scott, who has a grant from the Centers for Disease Control and Prevention to study ways to increase uptake. “Probably the easiest thing to do is just incorporate this information into your electronic medical record and make it part of your alerts and standard preventative practices. Try to automate a lot of this rather than remind a very busy primary care doctor of all the things they have to do,” he said.
For example, one strategy that Seattle’s King County has employed is to automatically notify the testing laboratory if an antibody test is positive. “The lab knows to keep that blood and run a second (nucleic acid) test without the patient having to come back. That has helped to get our confirmatory rates up,” said Dr. Scott.
More broadly, the importance of testing needs to be emphasized, according to Paul J. Thuluvath, MD, medical director at the Institute of Digestive Health and Liver Disease at Mercy Medical Center, Baltimore, and a professor of medicine and surgery at the University of Maryland. “We need everybody to buy into this: the primary care physicians, internists, and gynecologists. If they are not convinced of the importance of this, it’s not going to happen. And I don’t think many primary care physicians and internists are convinced yet,” he said.
AGA Resource
Through the HCV Clinical Service Line, AGA offers tools to help you become more efficient, understand quality standards and improve the process of care for patients. Learn more at http://www.gastro.org/patient-care/conditions-diseases/hepatitis-c
Despite the urging of the United States Preventive Services Task Force and other organizations in 2013, the percentage of baby boomers who underwent testing for hepatitis C (HCV) infection had barely changed 2 years later – from 12.3% in 2013 to 13.8% in 2015.
The numbers are particularly troubling because new and improved antiviral drugs offer cures that could forestall liver cancer, cirrhosis, and other potential complications, with shorter regimens and fewer side effects than older regimens.
Other reactions were more forceful. “Kind of pathetic, isn’t it?” said John D. Scott, MD, assistant director of the Hepatitis and Liver Clinic at Harborview Medical Center, and an associate professor of medicine at the University of Washington, Seattle.
The researchers analyzed 2013 and 2015 data from the National Health Interview Survey, which included records for 21,827 baby boomers with HCV testing data.
The slight increase overall of 12.3% to 13.8% was small but also statistically significant (P = .013). Some populations fared better: Compared with the privately insured, those with Medicare plus Medicaid were more likely to have been tested (prevalence ratio, 1.83; 95% confidence interval, 1.32-2.53), as were those only on Medicaid (PR, 1.35; 95% CI, 1.04-1.76), and those with military insurance (PR, 1.62; 95% CI, 1.16-2.26).
The study could be subject to recall bias, since it relied on participants’ self-reports.
The authors speculate that the higher prevalence of testing in those with military insurance may reflect efforts by the Veterans Health Administration to reduce the high prevalence of HCV-associated disease among veterans.
It’s entirely possible to increase testing rates, according to Dr. Scott, who has a grant from the Centers for Disease Control and Prevention to study ways to increase uptake. “Probably the easiest thing to do is just incorporate this information into your electronic medical record and make it part of your alerts and standard preventative practices. Try to automate a lot of this rather than remind a very busy primary care doctor of all the things they have to do,” he said.
For example, one strategy that Seattle’s King County has employed is to automatically notify the testing laboratory if an antibody test is positive. “The lab knows to keep that blood and run a second (nucleic acid) test without the patient having to come back. That has helped to get our confirmatory rates up,” said Dr. Scott.
More broadly, the importance of testing needs to be emphasized, according to Paul J. Thuluvath, MD, medical director at the Institute of Digestive Health and Liver Disease at Mercy Medical Center, Baltimore, and a professor of medicine and surgery at the University of Maryland. “We need everybody to buy into this: the primary care physicians, internists, and gynecologists. If they are not convinced of the importance of this, it’s not going to happen. And I don’t think many primary care physicians and internists are convinced yet,” he said.
AGA Resource
Through the HCV Clinical Service Line, AGA offers tools to help you become more efficient, understand quality standards and improve the process of care for patients. Learn more at http://www.gastro.org/patient-care/conditions-diseases/hepatitis-c
Despite the urging of the United States Preventive Services Task Force and other organizations in 2013, the percentage of baby boomers who underwent testing for hepatitis C (HCV) infection had barely changed 2 years later – from 12.3% in 2013 to 13.8% in 2015.
The numbers are particularly troubling because new and improved antiviral drugs offer cures that could forestall liver cancer, cirrhosis, and other potential complications, with shorter regimens and fewer side effects than older regimens.
Other reactions were more forceful. “Kind of pathetic, isn’t it?” said John D. Scott, MD, assistant director of the Hepatitis and Liver Clinic at Harborview Medical Center, and an associate professor of medicine at the University of Washington, Seattle.
The researchers analyzed 2013 and 2015 data from the National Health Interview Survey, which included records for 21,827 baby boomers with HCV testing data.
The slight increase overall of 12.3% to 13.8% was small but also statistically significant (P = .013). Some populations fared better: Compared with the privately insured, those with Medicare plus Medicaid were more likely to have been tested (prevalence ratio, 1.83; 95% confidence interval, 1.32-2.53), as were those only on Medicaid (PR, 1.35; 95% CI, 1.04-1.76), and those with military insurance (PR, 1.62; 95% CI, 1.16-2.26).
The study could be subject to recall bias, since it relied on participants’ self-reports.
The authors speculate that the higher prevalence of testing in those with military insurance may reflect efforts by the Veterans Health Administration to reduce the high prevalence of HCV-associated disease among veterans.
It’s entirely possible to increase testing rates, according to Dr. Scott, who has a grant from the Centers for Disease Control and Prevention to study ways to increase uptake. “Probably the easiest thing to do is just incorporate this information into your electronic medical record and make it part of your alerts and standard preventative practices. Try to automate a lot of this rather than remind a very busy primary care doctor of all the things they have to do,” he said.
For example, one strategy that Seattle’s King County has employed is to automatically notify the testing laboratory if an antibody test is positive. “The lab knows to keep that blood and run a second (nucleic acid) test without the patient having to come back. That has helped to get our confirmatory rates up,” said Dr. Scott.
More broadly, the importance of testing needs to be emphasized, according to Paul J. Thuluvath, MD, medical director at the Institute of Digestive Health and Liver Disease at Mercy Medical Center, Baltimore, and a professor of medicine and surgery at the University of Maryland. “We need everybody to buy into this: the primary care physicians, internists, and gynecologists. If they are not convinced of the importance of this, it’s not going to happen. And I don’t think many primary care physicians and internists are convinced yet,” he said.
AGA Resource
Through the HCV Clinical Service Line, AGA offers tools to help you become more efficient, understand quality standards and improve the process of care for patients. Learn more at http://www.gastro.org/patient-care/conditions-diseases/hepatitis-c
FROM AMERICAN JOURNAL OF PREVENTIVE MEDICINE