User login
Target self-medication of mood and anxiety symptoms
SAN FRANCISCO – Drinking to alleviate mood or anxiety symptoms is responsible for 12%-16% of cases of new-onset alcohol use disorder in affected individuals, Jitender Sareen, MD, said at the annual conference of the Anxiety and Depression Association of America.
Similarly, the use of prescription or nonprescription drugs to self-medicate mood or anxiety symptoms accounts for 20% of new-onset drug use disorders in this population, added Dr. Sareen, professor and head of the department of psychiatry at the University of Manitoba in Winnipeg.
He was a coauthor of two landmark longitudinal epidemiologic studies that support the concept of self-medication as a direct causal mechanism that explains a phenomenon often observed in clinical practice: namely, the high rate of comorbid mood or anxiety disorders accompanied by an alcohol or substance use disorder.
“The clinical implication is that questions about self-medication with alcohol or drugs should be included in the assessment of patients with anxiety and mood symptoms, because self-medication is a marker of higher likelihood of psychopathology. And psychologic therapies like cognitive-behavioral therapy and dialectical behavior therapy could prevent onset of substance use disorders by teaching patients emotion regulation skills to manage their mood and anxiety symptoms without self-medication,” Dr. Sareen said.
The first longitudinal study of the role of self-medication in the development of comorbid anxiety and substance use disorders included 34,653 nationally representative adults who completed both the initial face-to-face National Epidemiologic Survey on Alcohol and Related Conditions in 2001-2002 and a follow-up survey conducted 3 years later.
During the 3-year follow-up period, 9.7% of subjects developed a new-onset anxiety disorder, 5.9% of participants newly met DSM-IV diagnostic criteria for alcohol use disorder, and 2% developed a new-onset drug use disorder.
Among subjects who met the criteria for an anxiety disorder at baseline and at that time also reported self-medication with alcohol, 12.6% developed an incident alcohol use disorder during follow-up. Among those who self-medicated with drugs, 10.4% developed a drug use disorder.
In contrast, only 4.7% of subjects with a baseline anxiety disorder who did not self-medicate with alcohol at baseline developed an incident alcohol use disorder. And an incident drug use disorder occurred in 1.7% of patients with a baseline anxiety disorder who did not self-medicate with drugs.
Among patients with a baseline alcohol or other substance use disorder, self-medication with alcohol was associated with an adjusted 2.13-fold increased likelihood of developing social phobia during 3 years of follow-up, while self-medication with other drugs was independently associated with a 3.27-fold increased likelihood of subsequently developing social phobia.
In a multivariate logistic regression analysis, Dr. Sareen and his coinvestigators determined that self-medication with alcohol by patients with an anxiety disorder at baseline was associated with a 2.63-fold increased risk of incident alcohol use disorder during follow-up. Self-medication with drugs in patients with a baseline anxiety disorder was associated with a 4.99-fold risk of a new-onset substance use disorder during the 3 years of follow-up (Arch Gen Psychiatry. 2011;68[8]:800-7).
In a subsequent analysis of the same prospectively studied population, Dr. Sareen and his colleagues focused specifically on drinking to self-medicate mood symptoms. They found that self-medication with alcohol was associated with an adjusted 3.1-fold increased likelihood of new-onset alcohol dependence during the 3-year follow-up, as well as with a 3.45-fold increased risk of persistence of alcohol dependence. Roughly 12% of all cases of incident alcohol dependence arising during follow-up of patients with baseline mood symptoms were attributed to self-medication with alcohol. The increased risk of new-onset alcohol dependence was observed not only in subjects who met DSM-IV criteria for an affective disorder, but in those with subthreshold mood symptoms as well (JAMA Psychiatry. 2013 Jul;70[7]:718-26).
Again, this points to drinking as a behavior employed to self-medicate mood symptoms as a potential target for preventive interventions aimed at reducing the occurrence of alcohol dependence. As yet, however, no formal studies have been done to confirm the effectiveness of this strategy, the psychiatrist continued.
Dr. Sareen was not involved in the third iteration of the National Epidemiologic Survey on Alcohol and Related Conditions, in which a different group of 36,309 nationally representative adults was interviewed during 2011-2013 to assess the impact of the DSM-5 criteria for alcohol use disorder. Using DSM-5, 13.9% of the population met criteria for an alcohol use disorder during the past 12 months, and the lifetime prevalence of alcohol use disorder was 29.1%. Fewer than one in five subjects with a lifetime DSM-5 alcohol use disorder had ever been treated.
In the first national survey, which used DSM-IV criteria, the 12-month and lifetime prevalences of alcohol abuse and/or dependence were 8.5% and 30.3%, respectively.
DSM-5 alcohol use disorder was highly comorbid. Both lifetime and 12-month alcohol use disorder were associated with significantly increased likelihood of other substance use disorders, major depression, bipolar I disorder, borderline personality disorder, and antisocial personality disorder.
These data indicate “an urgent need to educate the public and policy makers about alcohol use disorder and its treatment alternatives, to destigmatize the disorder, and to encourage those who cannot reduce their alcohol consumption on their own, despite substantial harm to themselves and others, to seek treatment,” the investigators wrote (JAMA Psychiatry. 2015 Aug;72[8]:757-66).
The surveys were supported by the National Institute on Alcohol Abuse and Alcoholism. Dr. Sareen reported having no financial conflicts of interest.
SAN FRANCISCO – Drinking to alleviate mood or anxiety symptoms is responsible for 12%-16% of cases of new-onset alcohol use disorder in affected individuals, Jitender Sareen, MD, said at the annual conference of the Anxiety and Depression Association of America.
Similarly, the use of prescription or nonprescription drugs to self-medicate mood or anxiety symptoms accounts for 20% of new-onset drug use disorders in this population, added Dr. Sareen, professor and head of the department of psychiatry at the University of Manitoba in Winnipeg.
He was a coauthor of two landmark longitudinal epidemiologic studies that support the concept of self-medication as a direct causal mechanism that explains a phenomenon often observed in clinical practice: namely, the high rate of comorbid mood or anxiety disorders accompanied by an alcohol or substance use disorder.
“The clinical implication is that questions about self-medication with alcohol or drugs should be included in the assessment of patients with anxiety and mood symptoms, because self-medication is a marker of higher likelihood of psychopathology. And psychologic therapies like cognitive-behavioral therapy and dialectical behavior therapy could prevent onset of substance use disorders by teaching patients emotion regulation skills to manage their mood and anxiety symptoms without self-medication,” Dr. Sareen said.
The first longitudinal study of the role of self-medication in the development of comorbid anxiety and substance use disorders included 34,653 nationally representative adults who completed both the initial face-to-face National Epidemiologic Survey on Alcohol and Related Conditions in 2001-2002 and a follow-up survey conducted 3 years later.
During the 3-year follow-up period, 9.7% of subjects developed a new-onset anxiety disorder, 5.9% of participants newly met DSM-IV diagnostic criteria for alcohol use disorder, and 2% developed a new-onset drug use disorder.
Among subjects who met the criteria for an anxiety disorder at baseline and at that time also reported self-medication with alcohol, 12.6% developed an incident alcohol use disorder during follow-up. Among those who self-medicated with drugs, 10.4% developed a drug use disorder.
In contrast, only 4.7% of subjects with a baseline anxiety disorder who did not self-medicate with alcohol at baseline developed an incident alcohol use disorder. And an incident drug use disorder occurred in 1.7% of patients with a baseline anxiety disorder who did not self-medicate with drugs.
Among patients with a baseline alcohol or other substance use disorder, self-medication with alcohol was associated with an adjusted 2.13-fold increased likelihood of developing social phobia during 3 years of follow-up, while self-medication with other drugs was independently associated with a 3.27-fold increased likelihood of subsequently developing social phobia.
In a multivariate logistic regression analysis, Dr. Sareen and his coinvestigators determined that self-medication with alcohol by patients with an anxiety disorder at baseline was associated with a 2.63-fold increased risk of incident alcohol use disorder during follow-up. Self-medication with drugs in patients with a baseline anxiety disorder was associated with a 4.99-fold risk of a new-onset substance use disorder during the 3 years of follow-up (Arch Gen Psychiatry. 2011;68[8]:800-7).
In a subsequent analysis of the same prospectively studied population, Dr. Sareen and his colleagues focused specifically on drinking to self-medicate mood symptoms. They found that self-medication with alcohol was associated with an adjusted 3.1-fold increased likelihood of new-onset alcohol dependence during the 3-year follow-up, as well as with a 3.45-fold increased risk of persistence of alcohol dependence. Roughly 12% of all cases of incident alcohol dependence arising during follow-up of patients with baseline mood symptoms were attributed to self-medication with alcohol. The increased risk of new-onset alcohol dependence was observed not only in subjects who met DSM-IV criteria for an affective disorder, but in those with subthreshold mood symptoms as well (JAMA Psychiatry. 2013 Jul;70[7]:718-26).
Again, this points to drinking as a behavior employed to self-medicate mood symptoms as a potential target for preventive interventions aimed at reducing the occurrence of alcohol dependence. As yet, however, no formal studies have been done to confirm the effectiveness of this strategy, the psychiatrist continued.
Dr. Sareen was not involved in the third iteration of the National Epidemiologic Survey on Alcohol and Related Conditions, in which a different group of 36,309 nationally representative adults was interviewed during 2011-2013 to assess the impact of the DSM-5 criteria for alcohol use disorder. Using DSM-5, 13.9% of the population met criteria for an alcohol use disorder during the past 12 months, and the lifetime prevalence of alcohol use disorder was 29.1%. Fewer than one in five subjects with a lifetime DSM-5 alcohol use disorder had ever been treated.
In the first national survey, which used DSM-IV criteria, the 12-month and lifetime prevalences of alcohol abuse and/or dependence were 8.5% and 30.3%, respectively.
DSM-5 alcohol use disorder was highly comorbid. Both lifetime and 12-month alcohol use disorder were associated with significantly increased likelihood of other substance use disorders, major depression, bipolar I disorder, borderline personality disorder, and antisocial personality disorder.
These data indicate “an urgent need to educate the public and policy makers about alcohol use disorder and its treatment alternatives, to destigmatize the disorder, and to encourage those who cannot reduce their alcohol consumption on their own, despite substantial harm to themselves and others, to seek treatment,” the investigators wrote (JAMA Psychiatry. 2015 Aug;72[8]:757-66).
The surveys were supported by the National Institute on Alcohol Abuse and Alcoholism. Dr. Sareen reported having no financial conflicts of interest.
SAN FRANCISCO – Drinking to alleviate mood or anxiety symptoms is responsible for 12%-16% of cases of new-onset alcohol use disorder in affected individuals, Jitender Sareen, MD, said at the annual conference of the Anxiety and Depression Association of America.
Similarly, the use of prescription or nonprescription drugs to self-medicate mood or anxiety symptoms accounts for 20% of new-onset drug use disorders in this population, added Dr. Sareen, professor and head of the department of psychiatry at the University of Manitoba in Winnipeg.
He was a coauthor of two landmark longitudinal epidemiologic studies that support the concept of self-medication as a direct causal mechanism that explains a phenomenon often observed in clinical practice: namely, the high rate of comorbid mood or anxiety disorders accompanied by an alcohol or substance use disorder.
“The clinical implication is that questions about self-medication with alcohol or drugs should be included in the assessment of patients with anxiety and mood symptoms, because self-medication is a marker of higher likelihood of psychopathology. And psychologic therapies like cognitive-behavioral therapy and dialectical behavior therapy could prevent onset of substance use disorders by teaching patients emotion regulation skills to manage their mood and anxiety symptoms without self-medication,” Dr. Sareen said.
The first longitudinal study of the role of self-medication in the development of comorbid anxiety and substance use disorders included 34,653 nationally representative adults who completed both the initial face-to-face National Epidemiologic Survey on Alcohol and Related Conditions in 2001-2002 and a follow-up survey conducted 3 years later.
During the 3-year follow-up period, 9.7% of subjects developed a new-onset anxiety disorder, 5.9% of participants newly met DSM-IV diagnostic criteria for alcohol use disorder, and 2% developed a new-onset drug use disorder.
Among subjects who met the criteria for an anxiety disorder at baseline and at that time also reported self-medication with alcohol, 12.6% developed an incident alcohol use disorder during follow-up. Among those who self-medicated with drugs, 10.4% developed a drug use disorder.
In contrast, only 4.7% of subjects with a baseline anxiety disorder who did not self-medicate with alcohol at baseline developed an incident alcohol use disorder. And an incident drug use disorder occurred in 1.7% of patients with a baseline anxiety disorder who did not self-medicate with drugs.
Among patients with a baseline alcohol or other substance use disorder, self-medication with alcohol was associated with an adjusted 2.13-fold increased likelihood of developing social phobia during 3 years of follow-up, while self-medication with other drugs was independently associated with a 3.27-fold increased likelihood of subsequently developing social phobia.
In a multivariate logistic regression analysis, Dr. Sareen and his coinvestigators determined that self-medication with alcohol by patients with an anxiety disorder at baseline was associated with a 2.63-fold increased risk of incident alcohol use disorder during follow-up. Self-medication with drugs in patients with a baseline anxiety disorder was associated with a 4.99-fold risk of a new-onset substance use disorder during the 3 years of follow-up (Arch Gen Psychiatry. 2011;68[8]:800-7).
In a subsequent analysis of the same prospectively studied population, Dr. Sareen and his colleagues focused specifically on drinking to self-medicate mood symptoms. They found that self-medication with alcohol was associated with an adjusted 3.1-fold increased likelihood of new-onset alcohol dependence during the 3-year follow-up, as well as with a 3.45-fold increased risk of persistence of alcohol dependence. Roughly 12% of all cases of incident alcohol dependence arising during follow-up of patients with baseline mood symptoms were attributed to self-medication with alcohol. The increased risk of new-onset alcohol dependence was observed not only in subjects who met DSM-IV criteria for an affective disorder, but in those with subthreshold mood symptoms as well (JAMA Psychiatry. 2013 Jul;70[7]:718-26).
Again, this points to drinking as a behavior employed to self-medicate mood symptoms as a potential target for preventive interventions aimed at reducing the occurrence of alcohol dependence. As yet, however, no formal studies have been done to confirm the effectiveness of this strategy, the psychiatrist continued.
Dr. Sareen was not involved in the third iteration of the National Epidemiologic Survey on Alcohol and Related Conditions, in which a different group of 36,309 nationally representative adults was interviewed during 2011-2013 to assess the impact of the DSM-5 criteria for alcohol use disorder. Using DSM-5, 13.9% of the population met criteria for an alcohol use disorder during the past 12 months, and the lifetime prevalence of alcohol use disorder was 29.1%. Fewer than one in five subjects with a lifetime DSM-5 alcohol use disorder had ever been treated.
In the first national survey, which used DSM-IV criteria, the 12-month and lifetime prevalences of alcohol abuse and/or dependence were 8.5% and 30.3%, respectively.
DSM-5 alcohol use disorder was highly comorbid. Both lifetime and 12-month alcohol use disorder were associated with significantly increased likelihood of other substance use disorders, major depression, bipolar I disorder, borderline personality disorder, and antisocial personality disorder.
These data indicate “an urgent need to educate the public and policy makers about alcohol use disorder and its treatment alternatives, to destigmatize the disorder, and to encourage those who cannot reduce their alcohol consumption on their own, despite substantial harm to themselves and others, to seek treatment,” the investigators wrote (JAMA Psychiatry. 2015 Aug;72[8]:757-66).
The surveys were supported by the National Institute on Alcohol Abuse and Alcoholism. Dr. Sareen reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM THE ANXIETY AND DEPRESSION CONFERENCE 2017
What drives readmissions within 90 days after MI hospitalization
WASHINGTON – In a large population of Medicare patients hospitalized for acute MI, four factors stood out as predictors of increased likelihood of readmission within 90 days, Aaron D. Kugelmass, MD, reported at the annual meeting of the American College of Cardiology.
These four predictors of 90-day readmission were end-stage renal disease at the time of the initial admission for MI, which in a logistic regression model was independently associated with an 88% relative increase in readmission risk; no percutaneous coronary intervention (PCI) during the index hospitalization, which carried a 64% increase in risk; type 1 diabetes, with a 57% increased readmission rate; and heart failure at the initial hospitalization, with an associated 34% greater risk, according to Dr. Kugelmass, chief of cardiology and medical director of the heart and vascular center at Baystate Medical Center in Springfield, Mass.
“This is going to be a learning curve for everyone,” he said.
“The best way to deal with this change is to understand the factors driving costs and morbidity and mortality,” Dr. Kugelmass said, in explaining why he conducted a retrospective study of readmissions within 90 days in a population of 143,286 Medicare beneficiaries hospitalized for acute MI in 2014. The study focus was on readmissions because they add so much to total cost of care for a 90-day episode.
Twenty-eight percent of patients were readmitted at least once within 90 days of discharge following their acute MI. The Medicare bundled payment plan divides MI patients into two separate groups: those who undergo PCI during their initial hospitalization and those who receive medical management only. Thirty-one percent of the readmitted patients in Dr. Kugelmass’s study had undergone PCI during their index hospitalization, while the other 69% were managed medically.
Heart failure was the No. 1 reason for readmission within 90 days in patients who had PCI during the index hospitalization. It was the primary reason for 17.6% of readmissions. Next came recurrent angina or chest pain, which accounted for 6.6% of readmissions; chronic obstructive pulmonary disease or pneumonia, 6.3%; and GI bleeding with hemorrhage, which was the primary reason for 6.0% of readmissions. Together these four causes accounted for more than 36% of all readmissions in the PCI group.
“The GI bleeding data were really interesting,” the cardiologist said. “There’s a lot of talk now about reducing the duration of dual-antiplatelet therapy [DAPT] after PCI. This is an administrative data set that’s quite large, and it shows that GI bleeding in a post-PCI group early in the duration of DAPT is in fact a significant cause of readmission and poses significant hazard.”
Among patients who were medically managed during their index hospitalization, the top four reasons for readmission were heart failure, accounting for 20.6% of readmissions; cardiac surgery, 13.5%; sepsis, 7.8%; and chronic obstructive pulmonary disease/pneumonia, 6.3%. GI bleeding wasn’t a significant cause of readmission in this group.
“I think what we need to do next is dive deeper into the medically managed group. There is a cohort in there that’s incredibly sick and are likely to drive costs and be prone to readmission. And there’s another component of the medically managed group that had to be fairly healthy because they were able to undergo coronary artery bypass surgery within 90 days,” Dr. Kugelmass said.
He reported having no financial conflicts regarding his study.
WASHINGTON – In a large population of Medicare patients hospitalized for acute MI, four factors stood out as predictors of increased likelihood of readmission within 90 days, Aaron D. Kugelmass, MD, reported at the annual meeting of the American College of Cardiology.
These four predictors of 90-day readmission were end-stage renal disease at the time of the initial admission for MI, which in a logistic regression model was independently associated with an 88% relative increase in readmission risk; no percutaneous coronary intervention (PCI) during the index hospitalization, which carried a 64% increase in risk; type 1 diabetes, with a 57% increased readmission rate; and heart failure at the initial hospitalization, with an associated 34% greater risk, according to Dr. Kugelmass, chief of cardiology and medical director of the heart and vascular center at Baystate Medical Center in Springfield, Mass.
“This is going to be a learning curve for everyone,” he said.
“The best way to deal with this change is to understand the factors driving costs and morbidity and mortality,” Dr. Kugelmass said, in explaining why he conducted a retrospective study of readmissions within 90 days in a population of 143,286 Medicare beneficiaries hospitalized for acute MI in 2014. The study focus was on readmissions because they add so much to total cost of care for a 90-day episode.
Twenty-eight percent of patients were readmitted at least once within 90 days of discharge following their acute MI. The Medicare bundled payment plan divides MI patients into two separate groups: those who undergo PCI during their initial hospitalization and those who receive medical management only. Thirty-one percent of the readmitted patients in Dr. Kugelmass’s study had undergone PCI during their index hospitalization, while the other 69% were managed medically.
Heart failure was the No. 1 reason for readmission within 90 days in patients who had PCI during the index hospitalization. It was the primary reason for 17.6% of readmissions. Next came recurrent angina or chest pain, which accounted for 6.6% of readmissions; chronic obstructive pulmonary disease or pneumonia, 6.3%; and GI bleeding with hemorrhage, which was the primary reason for 6.0% of readmissions. Together these four causes accounted for more than 36% of all readmissions in the PCI group.
“The GI bleeding data were really interesting,” the cardiologist said. “There’s a lot of talk now about reducing the duration of dual-antiplatelet therapy [DAPT] after PCI. This is an administrative data set that’s quite large, and it shows that GI bleeding in a post-PCI group early in the duration of DAPT is in fact a significant cause of readmission and poses significant hazard.”
Among patients who were medically managed during their index hospitalization, the top four reasons for readmission were heart failure, accounting for 20.6% of readmissions; cardiac surgery, 13.5%; sepsis, 7.8%; and chronic obstructive pulmonary disease/pneumonia, 6.3%. GI bleeding wasn’t a significant cause of readmission in this group.
“I think what we need to do next is dive deeper into the medically managed group. There is a cohort in there that’s incredibly sick and are likely to drive costs and be prone to readmission. And there’s another component of the medically managed group that had to be fairly healthy because they were able to undergo coronary artery bypass surgery within 90 days,” Dr. Kugelmass said.
He reported having no financial conflicts regarding his study.
WASHINGTON – In a large population of Medicare patients hospitalized for acute MI, four factors stood out as predictors of increased likelihood of readmission within 90 days, Aaron D. Kugelmass, MD, reported at the annual meeting of the American College of Cardiology.
These four predictors of 90-day readmission were end-stage renal disease at the time of the initial admission for MI, which in a logistic regression model was independently associated with an 88% relative increase in readmission risk; no percutaneous coronary intervention (PCI) during the index hospitalization, which carried a 64% increase in risk; type 1 diabetes, with a 57% increased readmission rate; and heart failure at the initial hospitalization, with an associated 34% greater risk, according to Dr. Kugelmass, chief of cardiology and medical director of the heart and vascular center at Baystate Medical Center in Springfield, Mass.
“This is going to be a learning curve for everyone,” he said.
“The best way to deal with this change is to understand the factors driving costs and morbidity and mortality,” Dr. Kugelmass said, in explaining why he conducted a retrospective study of readmissions within 90 days in a population of 143,286 Medicare beneficiaries hospitalized for acute MI in 2014. The study focus was on readmissions because they add so much to total cost of care for a 90-day episode.
Twenty-eight percent of patients were readmitted at least once within 90 days of discharge following their acute MI. The Medicare bundled payment plan divides MI patients into two separate groups: those who undergo PCI during their initial hospitalization and those who receive medical management only. Thirty-one percent of the readmitted patients in Dr. Kugelmass’s study had undergone PCI during their index hospitalization, while the other 69% were managed medically.
Heart failure was the No. 1 reason for readmission within 90 days in patients who had PCI during the index hospitalization. It was the primary reason for 17.6% of readmissions. Next came recurrent angina or chest pain, which accounted for 6.6% of readmissions; chronic obstructive pulmonary disease or pneumonia, 6.3%; and GI bleeding with hemorrhage, which was the primary reason for 6.0% of readmissions. Together these four causes accounted for more than 36% of all readmissions in the PCI group.
“The GI bleeding data were really interesting,” the cardiologist said. “There’s a lot of talk now about reducing the duration of dual-antiplatelet therapy [DAPT] after PCI. This is an administrative data set that’s quite large, and it shows that GI bleeding in a post-PCI group early in the duration of DAPT is in fact a significant cause of readmission and poses significant hazard.”
Among patients who were medically managed during their index hospitalization, the top four reasons for readmission were heart failure, accounting for 20.6% of readmissions; cardiac surgery, 13.5%; sepsis, 7.8%; and chronic obstructive pulmonary disease/pneumonia, 6.3%. GI bleeding wasn’t a significant cause of readmission in this group.
“I think what we need to do next is dive deeper into the medically managed group. There is a cohort in there that’s incredibly sick and are likely to drive costs and be prone to readmission. And there’s another component of the medically managed group that had to be fairly healthy because they were able to undergo coronary artery bypass surgery within 90 days,” Dr. Kugelmass said.
He reported having no financial conflicts regarding his study.
Key clinical point:
Major finding: Twenty-eight percent of Medicare patients hospitalized for acute MI were readmitted within 90 days.
Data source: A retrospective study of readmissions within 90 days among more than 143,000 Medicare beneficiaries hospitalized for acute MI in 2014.
Disclosures: The study presenter reported having no financial conflicts.
Corticosteroids may shorten flares of pediatric acute-onset neuropsychiatric syndrome
SAN FRANCISCO – Oral corticosteroids appear to be beneficial in treating flares of pediatric acute-onset neuropsychiatric syndrome, or PANS, according to Margo Thienemann, MD.
“Corticosteroids shorten the duration of flares, and if you treat patients early in their first episode, their overall course seems to be better,” said Dr. Thienemann, a child psychiatrist at Stanford (Calif.) Children’s Hospital.
She is part of a multidisciplinary Stanford PANS clinic, together with a pediatric immunologist, a pediatric rheumatologist, two nurse practitioners, a child psychologist, and a social worker, all devoted to the study and treatment of the debilitating condition.
Dr. Thienemann presented the findings of a retrospective, observational study of 98 patients at the PANS clinic who collectively had 403 disease flares. Eighty-five of the flares were treated with 102 courses of oral steroids, either in a 4- to 5-day burst or longer-duration regimens of up to 8 weeks. Dosing was weight based and averaged roughly 60 mg/day. Treatment response was assessed within 14 days after initiating short-burst therapy or at the end of a longer course.
When a child’s first episode of PANS was treated with oral steroids, the episode lasted for an average of 10.3 weeks; if untreated, the average duration was 16.5 weeks, Dr. Thienemann reported at the annual conference of the Anxiety and Depression Association of America.
Improvement of neuropsychiatric symptoms began on average 3.6 days into a course of oral steroids. That improvement lasted an average of 43.9 days before the next escalation of symptoms.
Longer treatment was better: Each additional day of steroid therapy was associated with a 2.56-day increase in the duration of improvement of neuropsychiatric symptoms in a logistic regression analysis adjusted for age, sex, weeks since onset of current PANS illness, use of cognitive-behavioral therapy during flares, antibiotic therapy, and the number of psychiatric medications a patient was on.
On the other hand, each day of delay in initiating oral corticosteroids was associated with an adjusted 0.18-week longer flare duration.
No improvement in PANS symptoms occurred in patients who developed an infection within 14 days after initiating corticosteroids. Among 31 such patients, 11 had no response to steroids, and only 6 were complete responders. In contrast, among a matched group of 31 patients without infection, there was 1 nonresponder, and there were 12 complete responders.
The Stanford group now is using intravenous corticosteroids as well to treat PANS. Although the group is still collecting data and isn’t yet ready to report results, Dr. Thienemann said intravenous therapy looks very promising.
“We’re seeing a more dramatic response with IV steroids, and with [fewer] side effects,” she said. “With oral steroids, patients become more labile for a day or two, and everything gets worse for that time before things start getting better.”
PANS is a strikingly abrupt-onset disorder. It is defined by dramatic onset of obsessive-compulsive disorder over the course of less than 72 hours and/or severe eating restriction, with at least two coinciding, debilitating neuropsychiatric symptoms. These PANS-defining symptoms may include anxiety, mood dysregulation, irritability or aggression, behavioral regression, cognitive deterioration, sensorimotor abnormalities, and/or somatic symptoms.
The average age of onset of PANS is 7-9 years. The course is typically relapsing/remitting.
“The symptoms are largely psychiatric. We see huge separation anxiety. And aggression – biting, hitting, and kicking in sweet kids who suddenly go crazy,” Dr. Thienemann said in an interview. “They regress behaviorally, have foggy brain, can’t process information, and they have frequent urination and bed-wetting, even if they never did that before. And their handwriting deteriorates.
“We think it’s probably basal ganglia inflammation,” she explained. “The same way a patient might immunologically attack his joints or heart after strep infection, we think it’s brain inflammation resulting from an abnormal immune response to infection.”
This postulated etiology is supported both by PET brain imaging studies and several animal models of PANS, she added.
If the symptoms are associated with a group A streptococcal infection, the disorder is called Pediatric Autoimmune Neuropsychiatric Disorder Associated With Streptococcal Infections, or PANDAS, which was first described in 1999 and predates PANS as a defined entity.
Based on the encouraging Stanford experience, a formal double-blind, placebo-controlled, randomized trial of corticosteroid therapy in patients with PANS is warranted, Dr. Thienemann said.
Awareness of PANS as a real entity is “getting better” among general pediatricians, according to the child psychiatrist.
“I think more and more it’s no longer a question about whether this exists,” she said. “Now, it’s a matter of disseminating treatment guidelines.”
The PANDAS Physicians Network has already released diagnostic guidelines. Preliminary treatment guidelines have been developed and will soon be published separately in immunology, infectious diseases, and psychiatry/behavioral medicine journals.
Dr. Thienemann reported having no financial conflicts regarding her study, which was supported by Stanford University.
SAN FRANCISCO – Oral corticosteroids appear to be beneficial in treating flares of pediatric acute-onset neuropsychiatric syndrome, or PANS, according to Margo Thienemann, MD.
“Corticosteroids shorten the duration of flares, and if you treat patients early in their first episode, their overall course seems to be better,” said Dr. Thienemann, a child psychiatrist at Stanford (Calif.) Children’s Hospital.
She is part of a multidisciplinary Stanford PANS clinic, together with a pediatric immunologist, a pediatric rheumatologist, two nurse practitioners, a child psychologist, and a social worker, all devoted to the study and treatment of the debilitating condition.
Dr. Thienemann presented the findings of a retrospective, observational study of 98 patients at the PANS clinic who collectively had 403 disease flares. Eighty-five of the flares were treated with 102 courses of oral steroids, either in a 4- to 5-day burst or longer-duration regimens of up to 8 weeks. Dosing was weight based and averaged roughly 60 mg/day. Treatment response was assessed within 14 days after initiating short-burst therapy or at the end of a longer course.
When a child’s first episode of PANS was treated with oral steroids, the episode lasted for an average of 10.3 weeks; if untreated, the average duration was 16.5 weeks, Dr. Thienemann reported at the annual conference of the Anxiety and Depression Association of America.
Improvement of neuropsychiatric symptoms began on average 3.6 days into a course of oral steroids. That improvement lasted an average of 43.9 days before the next escalation of symptoms.
Longer treatment was better: Each additional day of steroid therapy was associated with a 2.56-day increase in the duration of improvement of neuropsychiatric symptoms in a logistic regression analysis adjusted for age, sex, weeks since onset of current PANS illness, use of cognitive-behavioral therapy during flares, antibiotic therapy, and the number of psychiatric medications a patient was on.
On the other hand, each day of delay in initiating oral corticosteroids was associated with an adjusted 0.18-week longer flare duration.
No improvement in PANS symptoms occurred in patients who developed an infection within 14 days after initiating corticosteroids. Among 31 such patients, 11 had no response to steroids, and only 6 were complete responders. In contrast, among a matched group of 31 patients without infection, there was 1 nonresponder, and there were 12 complete responders.
The Stanford group now is using intravenous corticosteroids as well to treat PANS. Although the group is still collecting data and isn’t yet ready to report results, Dr. Thienemann said intravenous therapy looks very promising.
“We’re seeing a more dramatic response with IV steroids, and with [fewer] side effects,” she said. “With oral steroids, patients become more labile for a day or two, and everything gets worse for that time before things start getting better.”
PANS is a strikingly abrupt-onset disorder. It is defined by dramatic onset of obsessive-compulsive disorder over the course of less than 72 hours and/or severe eating restriction, with at least two coinciding, debilitating neuropsychiatric symptoms. These PANS-defining symptoms may include anxiety, mood dysregulation, irritability or aggression, behavioral regression, cognitive deterioration, sensorimotor abnormalities, and/or somatic symptoms.
The average age of onset of PANS is 7-9 years. The course is typically relapsing/remitting.
“The symptoms are largely psychiatric. We see huge separation anxiety. And aggression – biting, hitting, and kicking in sweet kids who suddenly go crazy,” Dr. Thienemann said in an interview. “They regress behaviorally, have foggy brain, can’t process information, and they have frequent urination and bed-wetting, even if they never did that before. And their handwriting deteriorates.
“We think it’s probably basal ganglia inflammation,” she explained. “The same way a patient might immunologically attack his joints or heart after strep infection, we think it’s brain inflammation resulting from an abnormal immune response to infection.”
This postulated etiology is supported both by PET brain imaging studies and several animal models of PANS, she added.
If the symptoms are associated with a group A streptococcal infection, the disorder is called Pediatric Autoimmune Neuropsychiatric Disorder Associated With Streptococcal Infections, or PANDAS, which was first described in 1999 and predates PANS as a defined entity.
Based on the encouraging Stanford experience, a formal double-blind, placebo-controlled, randomized trial of corticosteroid therapy in patients with PANS is warranted, Dr. Thienemann said.
Awareness of PANS as a real entity is “getting better” among general pediatricians, according to the child psychiatrist.
“I think more and more it’s no longer a question about whether this exists,” she said. “Now, it’s a matter of disseminating treatment guidelines.”
The PANDAS Physicians Network has already released diagnostic guidelines. Preliminary treatment guidelines have been developed and will soon be published separately in immunology, infectious diseases, and psychiatry/behavioral medicine journals.
Dr. Thienemann reported having no financial conflicts regarding her study, which was supported by Stanford University.
SAN FRANCISCO – Oral corticosteroids appear to be beneficial in treating flares of pediatric acute-onset neuropsychiatric syndrome, or PANS, according to Margo Thienemann, MD.
“Corticosteroids shorten the duration of flares, and if you treat patients early in their first episode, their overall course seems to be better,” said Dr. Thienemann, a child psychiatrist at Stanford (Calif.) Children’s Hospital.
She is part of a multidisciplinary Stanford PANS clinic, together with a pediatric immunologist, a pediatric rheumatologist, two nurse practitioners, a child psychologist, and a social worker, all devoted to the study and treatment of the debilitating condition.
Dr. Thienemann presented the findings of a retrospective, observational study of 98 patients at the PANS clinic who collectively had 403 disease flares. Eighty-five of the flares were treated with 102 courses of oral steroids, either in a 4- to 5-day burst or longer-duration regimens of up to 8 weeks. Dosing was weight based and averaged roughly 60 mg/day. Treatment response was assessed within 14 days after initiating short-burst therapy or at the end of a longer course.
When a child’s first episode of PANS was treated with oral steroids, the episode lasted for an average of 10.3 weeks; if untreated, the average duration was 16.5 weeks, Dr. Thienemann reported at the annual conference of the Anxiety and Depression Association of America.
Improvement of neuropsychiatric symptoms began on average 3.6 days into a course of oral steroids. That improvement lasted an average of 43.9 days before the next escalation of symptoms.
Longer treatment was better: Each additional day of steroid therapy was associated with a 2.56-day increase in the duration of improvement of neuropsychiatric symptoms in a logistic regression analysis adjusted for age, sex, weeks since onset of current PANS illness, use of cognitive-behavioral therapy during flares, antibiotic therapy, and the number of psychiatric medications a patient was on.
On the other hand, each day of delay in initiating oral corticosteroids was associated with an adjusted 0.18-week longer flare duration.
No improvement in PANS symptoms occurred in patients who developed an infection within 14 days after initiating corticosteroids. Among 31 such patients, 11 had no response to steroids, and only 6 were complete responders. In contrast, among a matched group of 31 patients without infection, there was 1 nonresponder, and there were 12 complete responders.
The Stanford group now is using intravenous corticosteroids as well to treat PANS. Although the group is still collecting data and isn’t yet ready to report results, Dr. Thienemann said intravenous therapy looks very promising.
“We’re seeing a more dramatic response with IV steroids, and with [fewer] side effects,” she said. “With oral steroids, patients become more labile for a day or two, and everything gets worse for that time before things start getting better.”
PANS is a strikingly abrupt-onset disorder. It is defined by dramatic onset of obsessive-compulsive disorder over the course of less than 72 hours and/or severe eating restriction, with at least two coinciding, debilitating neuropsychiatric symptoms. These PANS-defining symptoms may include anxiety, mood dysregulation, irritability or aggression, behavioral regression, cognitive deterioration, sensorimotor abnormalities, and/or somatic symptoms.
The average age of onset of PANS is 7-9 years. The course is typically relapsing/remitting.
“The symptoms are largely psychiatric. We see huge separation anxiety. And aggression – biting, hitting, and kicking in sweet kids who suddenly go crazy,” Dr. Thienemann said in an interview. “They regress behaviorally, have foggy brain, can’t process information, and they have frequent urination and bed-wetting, even if they never did that before. And their handwriting deteriorates.
“We think it’s probably basal ganglia inflammation,” she explained. “The same way a patient might immunologically attack his joints or heart after strep infection, we think it’s brain inflammation resulting from an abnormal immune response to infection.”
This postulated etiology is supported both by PET brain imaging studies and several animal models of PANS, she added.
If the symptoms are associated with a group A streptococcal infection, the disorder is called Pediatric Autoimmune Neuropsychiatric Disorder Associated With Streptococcal Infections, or PANDAS, which was first described in 1999 and predates PANS as a defined entity.
Based on the encouraging Stanford experience, a formal double-blind, placebo-controlled, randomized trial of corticosteroid therapy in patients with PANS is warranted, Dr. Thienemann said.
Awareness of PANS as a real entity is “getting better” among general pediatricians, according to the child psychiatrist.
“I think more and more it’s no longer a question about whether this exists,” she said. “Now, it’s a matter of disseminating treatment guidelines.”
The PANDAS Physicians Network has already released diagnostic guidelines. Preliminary treatment guidelines have been developed and will soon be published separately in immunology, infectious diseases, and psychiatry/behavioral medicine journals.
Dr. Thienemann reported having no financial conflicts regarding her study, which was supported by Stanford University.
AT ANXIETY AND DEPRESSION CONFERENCE 2017
Key clinical point:
Major finding: Each additional day of oral corticosteroid therapy for a PANS flare was independently associated with a 2.56-day longer duration of improved neuropsychiatric symptoms.
Data source: A retrospective observational study of 98 patients with PANS who had 403 disease flares, 85 of which were treated using oral corticosteroids.
Disclosures: Dr. Thienemann reported having no financial conflicts regarding her study, which was supported by Stanford University.
CRT-D beneficial in mild HF with ejection fraction above 30%
WASHINGTON – Patients with mild heart failure symptoms, left bundle branch block, and a left ventricular ejection fraction of 31% to 44% who received cardiac resynchronization therapy with a built-in defibrillator experienced a significant reduction in all-cause mortality, compared with those randomized to an implantable cardioverter-defibrillator alone during 7 years of follow-up.
These results from a new MADIT-CRT (Multicenter Automatic Defibrillator Implantation Trial With Cardiac Resynchronization Therapy) long-term follow-up substudy “suggest that patients with a relatively preserved ejection fraction greater than 30% benefit from CRT-D [cardiac resynchronization therapy defibrillator] and could potentially be considered for this therapy,” said Katherine Vermilye, MD, at the annual meeting of the American College of Cardiology.
In a subsequent publication, the MADIT-CRT investigators reported that, with extension of follow-up to 7 years, CRT-D also provided a significant benefit in terms of all-cause mortality in addition to the reduced rate of heart failure events (N Engl J Med. 2014 May 1;370[18]:1694-701).
However, even though an LVEF of 30% or less was a requirement for participation in MADIT-CRT, it turned out that, when the initial screening echocardiograms were eventually analyzed in a central core laboratory, one-third of study participants actually had a baseline LVEF of 31% to 44%, with the majority of excessive values being in the 31%-35% range.
Dr. Vermilye, of the University of Rochester in New York, presented a post hoc analysis of long-term outcomes in the subgroup having a baseline LVEF greater than 30%. They totaled 450 of 1,224 MADIT-CRT participants with left bundle branch block. They were significantly older and more likely to be female than the 824 subjects with an LVEF of 30% or less. They also had a shorter QRS duration – an average of 160 ms, versus 165 ms in patients with an LVEF of 30% or lower – and a smaller baseline left ventricular end systolic volume of 151 mL, compared with 196 mL in patients with a lower LVEF.
In a multivariate Cox regression analysis adjusted for potential confounders, CRT-D in patients with a baseline LVEF greater than 30% was associated with a 54% reduction in the risk of all-cause mortality at 7 years of follow-up, compared with receipt of an ICD-only device and with a smaller yet significant 31% reduction in risk in those with an LVEF of 30% or less. Worsening heart failure events were reduced by 64% in patients with a baseline LVEF greater than 30% who received CRT-D, compared with ICD-only, and by 54% in those with a lower baseline LVEF.
The reduction in all-cause mortality seen with CRT-D was confined to patients who were high responders to CRT as defined echocardiographically by at least a 35% change in left ventricular end systolic volume 1 year post implantation. They had an 85% reduction in the risk of death during 7 years of follow-up with CRT-D if their baseline LVEF was greater than 30% and a 58% relative risk reduction if their LVEF was 30% or less.
In contrast, CRT-D brought a significantly reduced risk of heart failure events regardless of whether a patient was a low or high responder, although the magnitude of benefit was greater in the high responders. Among patients with a baseline LVEF greater than 30%, CRT-D low responders had a 52% reduction in risk of heart failure events, compared with ICD recipients, while CRT-D high responders had an 81% relative risk reduction. Similarly, in patients with a baseline LVEF of 30% or less, CRT-D low responders had 48% reduction in heart failure events and high responders had a 79% risk reduction, compared with the ICD-only group.
Because this is a post hoc analysis, these new MADIT-CRT findings require validation in future studies, Dr. Vermilye observed.
MADIT-CRT was supported by Boston Scientific. Dr.. Vermilye reported having no financial conflicts.
WASHINGTON – Patients with mild heart failure symptoms, left bundle branch block, and a left ventricular ejection fraction of 31% to 44% who received cardiac resynchronization therapy with a built-in defibrillator experienced a significant reduction in all-cause mortality, compared with those randomized to an implantable cardioverter-defibrillator alone during 7 years of follow-up.
These results from a new MADIT-CRT (Multicenter Automatic Defibrillator Implantation Trial With Cardiac Resynchronization Therapy) long-term follow-up substudy “suggest that patients with a relatively preserved ejection fraction greater than 30% benefit from CRT-D [cardiac resynchronization therapy defibrillator] and could potentially be considered for this therapy,” said Katherine Vermilye, MD, at the annual meeting of the American College of Cardiology.
In a subsequent publication, the MADIT-CRT investigators reported that, with extension of follow-up to 7 years, CRT-D also provided a significant benefit in terms of all-cause mortality in addition to the reduced rate of heart failure events (N Engl J Med. 2014 May 1;370[18]:1694-701).
However, even though an LVEF of 30% or less was a requirement for participation in MADIT-CRT, it turned out that, when the initial screening echocardiograms were eventually analyzed in a central core laboratory, one-third of study participants actually had a baseline LVEF of 31% to 44%, with the majority of excessive values being in the 31%-35% range.
Dr. Vermilye, of the University of Rochester in New York, presented a post hoc analysis of long-term outcomes in the subgroup having a baseline LVEF greater than 30%. They totaled 450 of 1,224 MADIT-CRT participants with left bundle branch block. They were significantly older and more likely to be female than the 824 subjects with an LVEF of 30% or less. They also had a shorter QRS duration – an average of 160 ms, versus 165 ms in patients with an LVEF of 30% or lower – and a smaller baseline left ventricular end systolic volume of 151 mL, compared with 196 mL in patients with a lower LVEF.
In a multivariate Cox regression analysis adjusted for potential confounders, CRT-D in patients with a baseline LVEF greater than 30% was associated with a 54% reduction in the risk of all-cause mortality at 7 years of follow-up, compared with receipt of an ICD-only device and with a smaller yet significant 31% reduction in risk in those with an LVEF of 30% or less. Worsening heart failure events were reduced by 64% in patients with a baseline LVEF greater than 30% who received CRT-D, compared with ICD-only, and by 54% in those with a lower baseline LVEF.
The reduction in all-cause mortality seen with CRT-D was confined to patients who were high responders to CRT as defined echocardiographically by at least a 35% change in left ventricular end systolic volume 1 year post implantation. They had an 85% reduction in the risk of death during 7 years of follow-up with CRT-D if their baseline LVEF was greater than 30% and a 58% relative risk reduction if their LVEF was 30% or less.
In contrast, CRT-D brought a significantly reduced risk of heart failure events regardless of whether a patient was a low or high responder, although the magnitude of benefit was greater in the high responders. Among patients with a baseline LVEF greater than 30%, CRT-D low responders had a 52% reduction in risk of heart failure events, compared with ICD recipients, while CRT-D high responders had an 81% relative risk reduction. Similarly, in patients with a baseline LVEF of 30% or less, CRT-D low responders had 48% reduction in heart failure events and high responders had a 79% risk reduction, compared with the ICD-only group.
Because this is a post hoc analysis, these new MADIT-CRT findings require validation in future studies, Dr. Vermilye observed.
MADIT-CRT was supported by Boston Scientific. Dr.. Vermilye reported having no financial conflicts.
WASHINGTON – Patients with mild heart failure symptoms, left bundle branch block, and a left ventricular ejection fraction of 31% to 44% who received cardiac resynchronization therapy with a built-in defibrillator experienced a significant reduction in all-cause mortality, compared with those randomized to an implantable cardioverter-defibrillator alone during 7 years of follow-up.
These results from a new MADIT-CRT (Multicenter Automatic Defibrillator Implantation Trial With Cardiac Resynchronization Therapy) long-term follow-up substudy “suggest that patients with a relatively preserved ejection fraction greater than 30% benefit from CRT-D [cardiac resynchronization therapy defibrillator] and could potentially be considered for this therapy,” said Katherine Vermilye, MD, at the annual meeting of the American College of Cardiology.
In a subsequent publication, the MADIT-CRT investigators reported that, with extension of follow-up to 7 years, CRT-D also provided a significant benefit in terms of all-cause mortality in addition to the reduced rate of heart failure events (N Engl J Med. 2014 May 1;370[18]:1694-701).
However, even though an LVEF of 30% or less was a requirement for participation in MADIT-CRT, it turned out that, when the initial screening echocardiograms were eventually analyzed in a central core laboratory, one-third of study participants actually had a baseline LVEF of 31% to 44%, with the majority of excessive values being in the 31%-35% range.
Dr. Vermilye, of the University of Rochester in New York, presented a post hoc analysis of long-term outcomes in the subgroup having a baseline LVEF greater than 30%. They totaled 450 of 1,224 MADIT-CRT participants with left bundle branch block. They were significantly older and more likely to be female than the 824 subjects with an LVEF of 30% or less. They also had a shorter QRS duration – an average of 160 ms, versus 165 ms in patients with an LVEF of 30% or lower – and a smaller baseline left ventricular end systolic volume of 151 mL, compared with 196 mL in patients with a lower LVEF.
In a multivariate Cox regression analysis adjusted for potential confounders, CRT-D in patients with a baseline LVEF greater than 30% was associated with a 54% reduction in the risk of all-cause mortality at 7 years of follow-up, compared with receipt of an ICD-only device and with a smaller yet significant 31% reduction in risk in those with an LVEF of 30% or less. Worsening heart failure events were reduced by 64% in patients with a baseline LVEF greater than 30% who received CRT-D, compared with ICD-only, and by 54% in those with a lower baseline LVEF.
The reduction in all-cause mortality seen with CRT-D was confined to patients who were high responders to CRT as defined echocardiographically by at least a 35% change in left ventricular end systolic volume 1 year post implantation. They had an 85% reduction in the risk of death during 7 years of follow-up with CRT-D if their baseline LVEF was greater than 30% and a 58% relative risk reduction if their LVEF was 30% or less.
In contrast, CRT-D brought a significantly reduced risk of heart failure events regardless of whether a patient was a low or high responder, although the magnitude of benefit was greater in the high responders. Among patients with a baseline LVEF greater than 30%, CRT-D low responders had a 52% reduction in risk of heart failure events, compared with ICD recipients, while CRT-D high responders had an 81% relative risk reduction. Similarly, in patients with a baseline LVEF of 30% or less, CRT-D low responders had 48% reduction in heart failure events and high responders had a 79% risk reduction, compared with the ICD-only group.
Because this is a post hoc analysis, these new MADIT-CRT findings require validation in future studies, Dr. Vermilye observed.
MADIT-CRT was supported by Boston Scientific. Dr.. Vermilye reported having no financial conflicts.
AT ACC 2017
Key clinical point:
Major finding: The risk of all-cause mortality was reduced by 54% with CRT-D as compared with an ICD alone in MADIT-CRT participants with a baseline LVEF greater than 30% and by 31% in those with an LVEF of 30% or lower.
Data source: An analysis of 7-year rates of all-cause mortality and worsening heart failure events in 1,224 MADIT-CRT participants with left bundle branch block, 450 of whom had a baseline LVEF greater than 30%.
Disclosures: The MADIT-CRT study was supported by Boston Scientific. The presenter reported having no financial conflicts.
High readmits after peripheral arterial procedures
WASHINGTON – More than one in six patients who undergo a lower extremity arterial endovascular or surgical procedure are readmitted within 30 days, according to a large national study.
The annual total cost of these early readmissions is high, in excess of $360 million. But because there turned out to be surprisingly little difference in readmission rates between hospitals, 30-day readmissions may not be a rational quality measure on which to base institutional reimbursement or withholding of payment for peripheral arterial interventions, Eric A. Secemsky, MD, said at the annual meeting of the American College of Cardiology.
Forty-seven percent of patients had an endovascular procedure, 42% had surgery, and the remainder had hybrid procedures in which both endovascular and surgical interventions took place during the same admission. Patients with hybrid procedures contributed data to both treatment groups.
In-hospital mortality occurred in 2.5% of patients.
Of the patients who survived to discharge, 21,589, or 17.4%, were readmitted within 30 days. The early readmission rate was higher following endovascular procedures, at 18.7%, than the 16.1% rate in the surgical group. The average cost of a readmission was $15,876. Death during readmission occurred in 4.2% of patients.
The median rate ratio – a measure of the amount of variance in readmission rates between hospitals – was 1.12. That’s a low figure.
“If the median rate ratio is lower, like here, it says there’s not a lot of interhospital variability across the country. So overall this burden seems to be pretty uniform across the institutions included in our analysis,” Dr. Secemsky explained.
This observation drew the attention of session comoderator Naomi M. Hamburg, MD.
“It’s interesting that you didn’t see a lot of heterogeneity across hospitals, because we often think of readmissions as a potentially modifiable quality metric. Do you think it’s modifiable, or is this just the nature of the disease?” asked Dr. Hamburg of Boston Medical Center.
It’s the disease process, Dr. Secemsky replied.
“We were surprised by the lack of hospital variation,” he added. None of the institutional characteristics examined, including teaching hospital status, bed size, and procedural volume, had a significant impact on readmission rates.
But that doesn’t mean there aren’t opportunities to whittle down those readmissions, according to Dr. Secemsky.
He noted that the high readmission rates were driven by procedural complications such as graft or stent failure. Indeed, procedural complications accounted for 29% of all early readmissions. The procedural complication rate was about 20% following endovascular procedures and 39% after surgery. It’s likely that identification and implementation of best practices could trim those high rates. Unfortunately, however, the nationwide database relies upon ICD-9 codes, which don’t provide the granular level of detail required to home in on specific best practices. That will require further studies, according to Dr. Secemsky.
A distant second on the list of causes of early readmission was peripheral atherosclerosis, meaning persistent claudication or rest pain. This accounted for 8.8% of readmissions. Rounding out the top five causes of readmission were sepsis, which was the reason for 6.7% of readmissions; diabetes with complications, at 4.7%; and heart failure, at 4.6%.
The strongest predictors of readmission included having renal disease at baseline, Medicare rather than private insurance, and discharge to a subacute nursing facility or home with home care.
Dr. Hamburg commented that a focus on reducing readmissions for sepsis as well as for skin and soft tissue infections, which accounted for 2.1% of 30-day hospitalizations, could be fruitful.
Dr. Secemsky reported having no financial conflicts regarding his study.
WASHINGTON – More than one in six patients who undergo a lower extremity arterial endovascular or surgical procedure are readmitted within 30 days, according to a large national study.
The annual total cost of these early readmissions is high, in excess of $360 million. But because there turned out to be surprisingly little difference in readmission rates between hospitals, 30-day readmissions may not be a rational quality measure on which to base institutional reimbursement or withholding of payment for peripheral arterial interventions, Eric A. Secemsky, MD, said at the annual meeting of the American College of Cardiology.
Forty-seven percent of patients had an endovascular procedure, 42% had surgery, and the remainder had hybrid procedures in which both endovascular and surgical interventions took place during the same admission. Patients with hybrid procedures contributed data to both treatment groups.
In-hospital mortality occurred in 2.5% of patients.
Of the patients who survived to discharge, 21,589, or 17.4%, were readmitted within 30 days. The early readmission rate was higher following endovascular procedures, at 18.7%, than the 16.1% rate in the surgical group. The average cost of a readmission was $15,876. Death during readmission occurred in 4.2% of patients.
The median rate ratio – a measure of the amount of variance in readmission rates between hospitals – was 1.12. That’s a low figure.
“If the median rate ratio is lower, like here, it says there’s not a lot of interhospital variability across the country. So overall this burden seems to be pretty uniform across the institutions included in our analysis,” Dr. Secemsky explained.
This observation drew the attention of session comoderator Naomi M. Hamburg, MD.
“It’s interesting that you didn’t see a lot of heterogeneity across hospitals, because we often think of readmissions as a potentially modifiable quality metric. Do you think it’s modifiable, or is this just the nature of the disease?” asked Dr. Hamburg of Boston Medical Center.
It’s the disease process, Dr. Secemsky replied.
“We were surprised by the lack of hospital variation,” he added. None of the institutional characteristics examined, including teaching hospital status, bed size, and procedural volume, had a significant impact on readmission rates.
But that doesn’t mean there aren’t opportunities to whittle down those readmissions, according to Dr. Secemsky.
He noted that the high readmission rates were driven by procedural complications such as graft or stent failure. Indeed, procedural complications accounted for 29% of all early readmissions. The procedural complication rate was about 20% following endovascular procedures and 39% after surgery. It’s likely that identification and implementation of best practices could trim those high rates. Unfortunately, however, the nationwide database relies upon ICD-9 codes, which don’t provide the granular level of detail required to home in on specific best practices. That will require further studies, according to Dr. Secemsky.
A distant second on the list of causes of early readmission was peripheral atherosclerosis, meaning persistent claudication or rest pain. This accounted for 8.8% of readmissions. Rounding out the top five causes of readmission were sepsis, which was the reason for 6.7% of readmissions; diabetes with complications, at 4.7%; and heart failure, at 4.6%.
The strongest predictors of readmission included having renal disease at baseline, Medicare rather than private insurance, and discharge to a subacute nursing facility or home with home care.
Dr. Hamburg commented that a focus on reducing readmissions for sepsis as well as for skin and soft tissue infections, which accounted for 2.1% of 30-day hospitalizations, could be fruitful.
Dr. Secemsky reported having no financial conflicts regarding his study.
WASHINGTON – More than one in six patients who undergo a lower extremity arterial endovascular or surgical procedure are readmitted within 30 days, according to a large national study.
The annual total cost of these early readmissions is high, in excess of $360 million. But because there turned out to be surprisingly little difference in readmission rates between hospitals, 30-day readmissions may not be a rational quality measure on which to base institutional reimbursement or withholding of payment for peripheral arterial interventions, Eric A. Secemsky, MD, said at the annual meeting of the American College of Cardiology.
Forty-seven percent of patients had an endovascular procedure, 42% had surgery, and the remainder had hybrid procedures in which both endovascular and surgical interventions took place during the same admission. Patients with hybrid procedures contributed data to both treatment groups.
In-hospital mortality occurred in 2.5% of patients.
Of the patients who survived to discharge, 21,589, or 17.4%, were readmitted within 30 days. The early readmission rate was higher following endovascular procedures, at 18.7%, than the 16.1% rate in the surgical group. The average cost of a readmission was $15,876. Death during readmission occurred in 4.2% of patients.
The median rate ratio – a measure of the amount of variance in readmission rates between hospitals – was 1.12. That’s a low figure.
“If the median rate ratio is lower, like here, it says there’s not a lot of interhospital variability across the country. So overall this burden seems to be pretty uniform across the institutions included in our analysis,” Dr. Secemsky explained.
This observation drew the attention of session comoderator Naomi M. Hamburg, MD.
“It’s interesting that you didn’t see a lot of heterogeneity across hospitals, because we often think of readmissions as a potentially modifiable quality metric. Do you think it’s modifiable, or is this just the nature of the disease?” asked Dr. Hamburg of Boston Medical Center.
It’s the disease process, Dr. Secemsky replied.
“We were surprised by the lack of hospital variation,” he added. None of the institutional characteristics examined, including teaching hospital status, bed size, and procedural volume, had a significant impact on readmission rates.
But that doesn’t mean there aren’t opportunities to whittle down those readmissions, according to Dr. Secemsky.
He noted that the high readmission rates were driven by procedural complications such as graft or stent failure. Indeed, procedural complications accounted for 29% of all early readmissions. The procedural complication rate was about 20% following endovascular procedures and 39% after surgery. It’s likely that identification and implementation of best practices could trim those high rates. Unfortunately, however, the nationwide database relies upon ICD-9 codes, which don’t provide the granular level of detail required to home in on specific best practices. That will require further studies, according to Dr. Secemsky.
A distant second on the list of causes of early readmission was peripheral atherosclerosis, meaning persistent claudication or rest pain. This accounted for 8.8% of readmissions. Rounding out the top five causes of readmission were sepsis, which was the reason for 6.7% of readmissions; diabetes with complications, at 4.7%; and heart failure, at 4.6%.
The strongest predictors of readmission included having renal disease at baseline, Medicare rather than private insurance, and discharge to a subacute nursing facility or home with home care.
Dr. Hamburg commented that a focus on reducing readmissions for sepsis as well as for skin and soft tissue infections, which accounted for 2.1% of 30-day hospitalizations, could be fruitful.
Dr. Secemsky reported having no financial conflicts regarding his study.
AT ACC 2017
Key clinical point:
Major finding: Readmission within 30 days after a peripheral arterial procedure occurred nationally in 17.4% of patients, with little between-hospital variation in rates.
Data source: A retrospective analysis of nearly 124,000 hospital admissions for lower extremity arterial endovascular or surgical procedures.
Disclosures: The study presenter reported having no financial conflicts of interest.
Refining SLE cardiovascular risk estimation
WASHINGTON – Red blood cell distribution width provides a novel tool for cardiovascular risk stratification in patients with systemic lupus erythematosus (SLE), Chang H. Kim, MD, reported at the annual meeting of the American College of Cardiology.
In a retrospective cohort study of nearly 70,000 patients with SLE, the 10-year rate of major adverse cardiovascular events (MACE) rose stepwise according to quintile of red cell distribution width (RDW) from 5.3% in patients with an RDW of 13.2% or less to 38.6% in those with an RDW of 15.8% or greater, according to Dr. Kim of Case Western Reserve University in Cleveland.
He utilized the Explorys database to determine the 10-year cumulative incidence of MACE – defined as acute MI, heart failure, or cerebrovascular accident – during 2007-2016 in 69,920 patients with SLE and 14,825,240 controls. Explorys is an 8-year-old Cleveland-based company that maintains a health care database incorporating 26 health care systems across the United States with nearly 50 million patients. It is part of IBM Watson Health.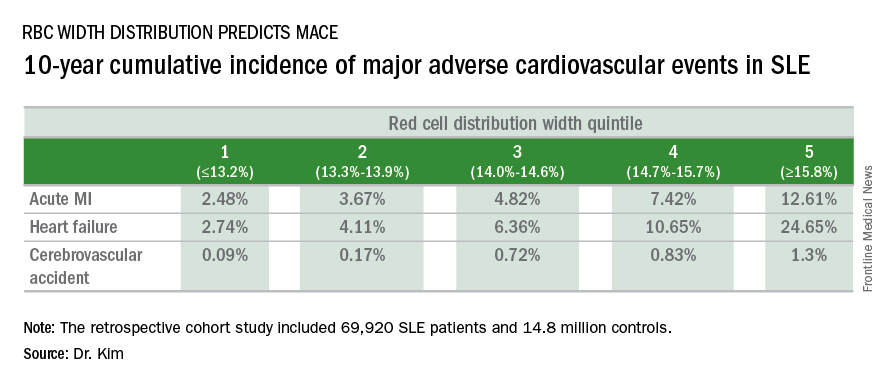
The MACE rate in patients with SLE displayed a graded increase in association with RDW quintile as measured in a routine CBC. (See table.) MACE rates were significantly higher in male than female SLE patients, but the graded relationship between RDW quintile and 10-year incidence of MACE persisted after adjustment for gender and the presence of anemia.
A graded association between RDW quintile and MACE also was noted in the control group of nearly 15 million individuals, but the absolute incidence of MACE in the non-SLE controls was far lower.
Dr. Kim reported having no financial conflicts regarding this unfunded study.
WASHINGTON – Red blood cell distribution width provides a novel tool for cardiovascular risk stratification in patients with systemic lupus erythematosus (SLE), Chang H. Kim, MD, reported at the annual meeting of the American College of Cardiology.
In a retrospective cohort study of nearly 70,000 patients with SLE, the 10-year rate of major adverse cardiovascular events (MACE) rose stepwise according to quintile of red cell distribution width (RDW) from 5.3% in patients with an RDW of 13.2% or less to 38.6% in those with an RDW of 15.8% or greater, according to Dr. Kim of Case Western Reserve University in Cleveland.
He utilized the Explorys database to determine the 10-year cumulative incidence of MACE – defined as acute MI, heart failure, or cerebrovascular accident – during 2007-2016 in 69,920 patients with SLE and 14,825,240 controls. Explorys is an 8-year-old Cleveland-based company that maintains a health care database incorporating 26 health care systems across the United States with nearly 50 million patients. It is part of IBM Watson Health.
The MACE rate in patients with SLE displayed a graded increase in association with RDW quintile as measured in a routine CBC. (See table.) MACE rates were significantly higher in male than female SLE patients, but the graded relationship between RDW quintile and 10-year incidence of MACE persisted after adjustment for gender and the presence of anemia.
A graded association between RDW quintile and MACE also was noted in the control group of nearly 15 million individuals, but the absolute incidence of MACE in the non-SLE controls was far lower.
Dr. Kim reported having no financial conflicts regarding this unfunded study.
WASHINGTON – Red blood cell distribution width provides a novel tool for cardiovascular risk stratification in patients with systemic lupus erythematosus (SLE), Chang H. Kim, MD, reported at the annual meeting of the American College of Cardiology.
In a retrospective cohort study of nearly 70,000 patients with SLE, the 10-year rate of major adverse cardiovascular events (MACE) rose stepwise according to quintile of red cell distribution width (RDW) from 5.3% in patients with an RDW of 13.2% or less to 38.6% in those with an RDW of 15.8% or greater, according to Dr. Kim of Case Western Reserve University in Cleveland.
He utilized the Explorys database to determine the 10-year cumulative incidence of MACE – defined as acute MI, heart failure, or cerebrovascular accident – during 2007-2016 in 69,920 patients with SLE and 14,825,240 controls. Explorys is an 8-year-old Cleveland-based company that maintains a health care database incorporating 26 health care systems across the United States with nearly 50 million patients. It is part of IBM Watson Health.
The MACE rate in patients with SLE displayed a graded increase in association with RDW quintile as measured in a routine CBC. (See table.) MACE rates were significantly higher in male than female SLE patients, but the graded relationship between RDW quintile and 10-year incidence of MACE persisted after adjustment for gender and the presence of anemia.
A graded association between RDW quintile and MACE also was noted in the control group of nearly 15 million individuals, but the absolute incidence of MACE in the non-SLE controls was far lower.
Dr. Kim reported having no financial conflicts regarding this unfunded study.
AT ACC 2017
Key clinical point:
Major finding: Systemic lupus erythematosus patients in the top quintile of RBC distribution width had a 10-year incidence of major adverse cardiovascular events of 39%.
Data source: This retrospective cohort study included nearly 70,000 SLE patients and 14.8 million controls.
Disclosures: The presenter reported no financial conflicts with regard to this unfunded study.
High early stroke risk for adult congenital heart disease
WASHINGTON – Adults with congenital heart disease are at fourfold greater risk of experiencing an ischemic stroke by age 60 than is the general population, Mette Glavind reported at the annual meeting of the American College of Cardiology.
She presented a population-based study that included all 14,710 Danish adults with congenital heart disease (ACHD) diagnosed in 1963-1994. Taking advantage of Denmark’s comprehensive system of linked national registries, she and her coinvestigators created a control group consisting of 144,735 age- and birth year–matched individuals from the general population.
During follow-up, a total of 2,868 Danes included in the study had an ischemic stroke. The cumulative incidence of ischemic stroke in the ACHD cohort was 0.8% by age 30 and 8.2% by age 60, compared with 0.09% and 2.9%, respectively, in controls, according to Ms. Glavind, a medical student at Aarhus (Denmark) University.
The median age at diagnosis of stroke was 52 years in the ACHD group and 69 years in controls. The risk of early ischemic stroke – defined as stroke at age 18-60 – was increased by 3.97-fold in the ACHD group, compared with controls. The risk of stroke after age 60 was increased by 1.68-fold.
Stroke was more likely to prove fatal in the ACHD group. Their 30-day stroke mortality rate was 10%, compared with 9.6% in controls. This corresponded to an adjusted 44% increased risk of stroke mortality, which was statistically significant.
The severity of congenital heart disease modified the stroke risk. Patients with mild or moderate ACHD had a 3.25-fold increased risk of early stroke, compared with controls, while those with severe or univentricular ACHD were at 5.97-fold greater risk.
For purposes of this study, mild ACHD was defined as a biventricular defect that was not repaired surgically or percutaneously. Moderate ACHD was considered to have biventricular pathophysiology with surgical or percutaneous intervention. The severe ACHD category was reserved for cases involving complex biventricular abnormalities.
By these definitions, 41% of patients had mild ACHD, 21% moderate, 22% severe, and 1% univentricular ACHD; the rest of the patients were unclassified.
This study was supported by Aarhus University and Cincinnati Children’s Hospital. Ms. Glavind reported having no financial conflicts.
WASHINGTON – Adults with congenital heart disease are at fourfold greater risk of experiencing an ischemic stroke by age 60 than is the general population, Mette Glavind reported at the annual meeting of the American College of Cardiology.
She presented a population-based study that included all 14,710 Danish adults with congenital heart disease (ACHD) diagnosed in 1963-1994. Taking advantage of Denmark’s comprehensive system of linked national registries, she and her coinvestigators created a control group consisting of 144,735 age- and birth year–matched individuals from the general population.
During follow-up, a total of 2,868 Danes included in the study had an ischemic stroke. The cumulative incidence of ischemic stroke in the ACHD cohort was 0.8% by age 30 and 8.2% by age 60, compared with 0.09% and 2.9%, respectively, in controls, according to Ms. Glavind, a medical student at Aarhus (Denmark) University.
The median age at diagnosis of stroke was 52 years in the ACHD group and 69 years in controls. The risk of early ischemic stroke – defined as stroke at age 18-60 – was increased by 3.97-fold in the ACHD group, compared with controls. The risk of stroke after age 60 was increased by 1.68-fold.
Stroke was more likely to prove fatal in the ACHD group. Their 30-day stroke mortality rate was 10%, compared with 9.6% in controls. This corresponded to an adjusted 44% increased risk of stroke mortality, which was statistically significant.
The severity of congenital heart disease modified the stroke risk. Patients with mild or moderate ACHD had a 3.25-fold increased risk of early stroke, compared with controls, while those with severe or univentricular ACHD were at 5.97-fold greater risk.
For purposes of this study, mild ACHD was defined as a biventricular defect that was not repaired surgically or percutaneously. Moderate ACHD was considered to have biventricular pathophysiology with surgical or percutaneous intervention. The severe ACHD category was reserved for cases involving complex biventricular abnormalities.
By these definitions, 41% of patients had mild ACHD, 21% moderate, 22% severe, and 1% univentricular ACHD; the rest of the patients were unclassified.
This study was supported by Aarhus University and Cincinnati Children’s Hospital. Ms. Glavind reported having no financial conflicts.
WASHINGTON – Adults with congenital heart disease are at fourfold greater risk of experiencing an ischemic stroke by age 60 than is the general population, Mette Glavind reported at the annual meeting of the American College of Cardiology.
She presented a population-based study that included all 14,710 Danish adults with congenital heart disease (ACHD) diagnosed in 1963-1994. Taking advantage of Denmark’s comprehensive system of linked national registries, she and her coinvestigators created a control group consisting of 144,735 age- and birth year–matched individuals from the general population.
During follow-up, a total of 2,868 Danes included in the study had an ischemic stroke. The cumulative incidence of ischemic stroke in the ACHD cohort was 0.8% by age 30 and 8.2% by age 60, compared with 0.09% and 2.9%, respectively, in controls, according to Ms. Glavind, a medical student at Aarhus (Denmark) University.
The median age at diagnosis of stroke was 52 years in the ACHD group and 69 years in controls. The risk of early ischemic stroke – defined as stroke at age 18-60 – was increased by 3.97-fold in the ACHD group, compared with controls. The risk of stroke after age 60 was increased by 1.68-fold.
Stroke was more likely to prove fatal in the ACHD group. Their 30-day stroke mortality rate was 10%, compared with 9.6% in controls. This corresponded to an adjusted 44% increased risk of stroke mortality, which was statistically significant.
The severity of congenital heart disease modified the stroke risk. Patients with mild or moderate ACHD had a 3.25-fold increased risk of early stroke, compared with controls, while those with severe or univentricular ACHD were at 5.97-fold greater risk.
For purposes of this study, mild ACHD was defined as a biventricular defect that was not repaired surgically or percutaneously. Moderate ACHD was considered to have biventricular pathophysiology with surgical or percutaneous intervention. The severe ACHD category was reserved for cases involving complex biventricular abnormalities.
By these definitions, 41% of patients had mild ACHD, 21% moderate, 22% severe, and 1% univentricular ACHD; the rest of the patients were unclassified.
This study was supported by Aarhus University and Cincinnati Children’s Hospital. Ms. Glavind reported having no financial conflicts.
AT ACC 2017
Key clinical point:
Major finding: Danish adults with complex biventricular congenital heart disease were sixfold more likely to have an ischemic stroke by age 60 years, compared with the general population.
Data source: A population-based registry study that included all Danish adults with congenital heart disease diagnosed in 1963-1994 and nearly 145,000 age- and birth year–matched controls drawn from the general Danish population.
Disclosures: This study was supported by Aarhus University and Cincinnati Children’s Hospital. The presenter reported having no financial conflicts.
Statins in HIV-positive patients are a missed opportunity
WASHINGTON – Only one in four U.S. adults with known HIV infection is on statin therapy, even though premature coronary heart disease has become a leading cause of morbidity and mortality for HIV-positive individuals in an era of greatly extended life expectancy due to highly active antiretroviral therapy, Robert S. Rosenson, MD, reported at the annual meeting of the American College of Cardiology.
Not only are statins greatly underutilized in HIV-positive patients, but in a large national observational study, 8% of those who were on a statin were on one that was contraindicated because of concomitant use of antiretroviral therapy or other agents posing a risk for major drug interactions, added Dr. Rosenson, professor of medicine and director of cardiometabolic disorders at Icahn School of Medicine at Mount Sinai, New York.
He presented a retrospective cohort study of 23,119 HIV-positive U.S. adults with commercial health insurance in 2014. The data source was the MarketScan database.
During the study year, 5,931 HIV-positive adults (26%) were taking a statin. In a multivariate regression analysis, factors associated with increased likelihood of statin therapy were age 50 years or older, male gender, known coronary heart disease, hypertension, and diabetes.
A total of 491 patients were taking a contraindicated statin or a statin at too high a dose to be safe, given the other medications they were on.
The protease inhibitors, nonnucleoside reverse transcriptase inhibitors, and some statins are metabolized by the cytochrome P450 isoenzyme CYP3A4. As a consequence, areas under the curve for simvastatin and lovastatin skyrocket when those statins are given with some protease inhibitors, placing patients at increased risk for myopathy, including rhabdomyolysis.
As a consequence, simvastatin and lovastatin are widely considered contraindicated in HIV-positive patients.
Infectious Diseases Society of America guidelines recommend pravastatin or low-dose atorvastatin at 10 mg/day as first-line therapy for lipid-lowering and cardiovascular risk reduction in HIV-positive patients, with fluvastatin a reasonable alternative.
Dr. Rosenson noted that in his study, 62% of the HIV-infected patients on a contraindicated statin were taking a protease inhibitor, 29% were on gemfibrozil (Lopid), and the remaining handful were on other agents posing significant risk of drug interactions, including azole antifungals, calcium channel blockers, or cobicistat (Tybost).
The statin study was jointly funded by Amgen, Mount Sinai, and the University of Alabama. Dr. Rosenson reported serving as a consultant to Amgen, Eli Lilly, Regeneron, and Sanofi.
WASHINGTON – Only one in four U.S. adults with known HIV infection is on statin therapy, even though premature coronary heart disease has become a leading cause of morbidity and mortality for HIV-positive individuals in an era of greatly extended life expectancy due to highly active antiretroviral therapy, Robert S. Rosenson, MD, reported at the annual meeting of the American College of Cardiology.
Not only are statins greatly underutilized in HIV-positive patients, but in a large national observational study, 8% of those who were on a statin were on one that was contraindicated because of concomitant use of antiretroviral therapy or other agents posing a risk for major drug interactions, added Dr. Rosenson, professor of medicine and director of cardiometabolic disorders at Icahn School of Medicine at Mount Sinai, New York.
He presented a retrospective cohort study of 23,119 HIV-positive U.S. adults with commercial health insurance in 2014. The data source was the MarketScan database.
During the study year, 5,931 HIV-positive adults (26%) were taking a statin. In a multivariate regression analysis, factors associated with increased likelihood of statin therapy were age 50 years or older, male gender, known coronary heart disease, hypertension, and diabetes.
A total of 491 patients were taking a contraindicated statin or a statin at too high a dose to be safe, given the other medications they were on.
The protease inhibitors, nonnucleoside reverse transcriptase inhibitors, and some statins are metabolized by the cytochrome P450 isoenzyme CYP3A4. As a consequence, areas under the curve for simvastatin and lovastatin skyrocket when those statins are given with some protease inhibitors, placing patients at increased risk for myopathy, including rhabdomyolysis.
As a consequence, simvastatin and lovastatin are widely considered contraindicated in HIV-positive patients.
Infectious Diseases Society of America guidelines recommend pravastatin or low-dose atorvastatin at 10 mg/day as first-line therapy for lipid-lowering and cardiovascular risk reduction in HIV-positive patients, with fluvastatin a reasonable alternative.
Dr. Rosenson noted that in his study, 62% of the HIV-infected patients on a contraindicated statin were taking a protease inhibitor, 29% were on gemfibrozil (Lopid), and the remaining handful were on other agents posing significant risk of drug interactions, including azole antifungals, calcium channel blockers, or cobicistat (Tybost).
The statin study was jointly funded by Amgen, Mount Sinai, and the University of Alabama. Dr. Rosenson reported serving as a consultant to Amgen, Eli Lilly, Regeneron, and Sanofi.
WASHINGTON – Only one in four U.S. adults with known HIV infection is on statin therapy, even though premature coronary heart disease has become a leading cause of morbidity and mortality for HIV-positive individuals in an era of greatly extended life expectancy due to highly active antiretroviral therapy, Robert S. Rosenson, MD, reported at the annual meeting of the American College of Cardiology.
Not only are statins greatly underutilized in HIV-positive patients, but in a large national observational study, 8% of those who were on a statin were on one that was contraindicated because of concomitant use of antiretroviral therapy or other agents posing a risk for major drug interactions, added Dr. Rosenson, professor of medicine and director of cardiometabolic disorders at Icahn School of Medicine at Mount Sinai, New York.
He presented a retrospective cohort study of 23,119 HIV-positive U.S. adults with commercial health insurance in 2014. The data source was the MarketScan database.
During the study year, 5,931 HIV-positive adults (26%) were taking a statin. In a multivariate regression analysis, factors associated with increased likelihood of statin therapy were age 50 years or older, male gender, known coronary heart disease, hypertension, and diabetes.
A total of 491 patients were taking a contraindicated statin or a statin at too high a dose to be safe, given the other medications they were on.
The protease inhibitors, nonnucleoside reverse transcriptase inhibitors, and some statins are metabolized by the cytochrome P450 isoenzyme CYP3A4. As a consequence, areas under the curve for simvastatin and lovastatin skyrocket when those statins are given with some protease inhibitors, placing patients at increased risk for myopathy, including rhabdomyolysis.
As a consequence, simvastatin and lovastatin are widely considered contraindicated in HIV-positive patients.
Infectious Diseases Society of America guidelines recommend pravastatin or low-dose atorvastatin at 10 mg/day as first-line therapy for lipid-lowering and cardiovascular risk reduction in HIV-positive patients, with fluvastatin a reasonable alternative.
Dr. Rosenson noted that in his study, 62% of the HIV-infected patients on a contraindicated statin were taking a protease inhibitor, 29% were on gemfibrozil (Lopid), and the remaining handful were on other agents posing significant risk of drug interactions, including azole antifungals, calcium channel blockers, or cobicistat (Tybost).
The statin study was jointly funded by Amgen, Mount Sinai, and the University of Alabama. Dr. Rosenson reported serving as a consultant to Amgen, Eli Lilly, Regeneron, and Sanofi.
AT ACC 2017
Key clinical point:
Major finding: Lipid-lowering in HIV-positive adults can be tricky: Eight percent of those who were on statin therapy were taking a statin contraindicated in HIV-infected individuals.
Data source: A retrospective study of more than 23,000 HIV-positive U.S. adults with commercial health insurance in 2014.
Disclosures: The statin study was jointly funded by Amgen and two U.S. medical schools. Dr. Rosenson reported serving as a consultant to Amgen, Eli Lilly, Regeneron, and Sanofi.
Antianginal agents underutilized after MI
WASHINGTON – Non–beta-blocker antianginal drugs are seriously underprescribed in patients with angina following percutaneous coronary intervention (PCI) for a myocardial infarction (MI), according to a large national study, Alexander C. Fanaroff, MD, said at the annual meeting of the American College of Cardiology.
Clinical practice in this regard appears to be largely out of touch with current evidence-based guidelines regarding angina management in patients with stable ischemic heart disease. Those ACC/American Heart Association guidelines recommend a stepwise approach beginning with a beta-blocker and sublingual nitroglycerin, adding a calcium channel blocker if the beta-blocker isn’t tolerated or effective, further adding a long-acting nitrate if symptoms persist, incorporating ranolazine (Ranexa) as needed, and finally turning to revascularization for symptomatic relief if multidrug therapy proves inadequate (J Am Coll Cardiol. 2012 Dec 18;60[24]:e44-e164).
“When you ask patients and physicians separately about the prevalence of angina, they tend to disagree, with physicians consistently thinking their patients have less angina than patients report themselves. Together, with our data, this indicates that strategies to improve clinician recognition and treatment of angina symptoms are needed,” he asserted.
TRANSLATE-ACS was a large, longitudinal, observational study of patients who underwent PCI at 233 U.S. hospitals during 2010-2012. Dr. Fanaroff focused on 12-month follow-up data on the 10,870 patients whose PCI was for treatment of an acute MI. In telephone interviews 6 weeks postdischarge which incorporated the validated Seattle Angina Questionnaire, 3,190 of these individuals (31%) with stable ischemic heart disease reported angina symptoms. Of those, 79% said their symptoms occurred at least monthly, 18% weekly, and 3% daily. Yet, only 1 in 5 patients with angina 6 weeks postdischarge were on a non–beta-blocker antianginal medication: 16% were on monotherapy with a calcium channel blocker or long-acting nitrate, 3% were on dual therapy, and less than 1% were on three non–beta-blockers for their angina.
The prevalence of angina dropped over time. At 6 months postdischarge, 40% of the original cohort of patients who had angina at 6 weeks still reported angina. At 12 months, it was 33%. Meanwhile, the use of non–beta-blocker antianginal agents crept up modestly over time, from 20% in patients with angina at 6 weeks postdischarge, to 22% in those with angina at 6 months, and 23% at 12 months.
On the other hand, 33% of patients with angina at 6 weeks had persistent angina through 12 months. Remarkably, 69% of these individuals never received a non–beta-blocker antianginal drug during all that time, including 61% of those who experienced daily or weekly angina.
The use of non–beta-blocker antianginal drugs was more frequent in patients with more severe angina: 25% of patients with daily or weekly angina at 6 weeks were on such medication, as were 34% at 6 months and 38% at 12 months, compared with 16%, 18%, and 22% of those with monthly angina. Still, most of these patients were on only a single non–beta-blocker antianginal agent.
Of patients with angina at 6 weeks, 12% subsequently underwent repeat PCI at some point during the first year postdischarge. Of these individuals, 19% were on a single non–beta-blocker antianginal drug at the time of their procedure, 6% were on two such drugs, and only 1% were on three.
Roughly 10% of patients with multivessel coronary disease who were identified angiographically at the time of their acute MI did not undergo multivessel PCI at that time. This was not predictive of angina 6 weeks post discharge, Dr. Fanaroff continued.
One audience member asked Dr. Fanaroff how he knows that the patients with self-reported angina didn’t actually have noncardiac chest pain, in which case their physicians’ decision not to prescribe second- and third-tier antianginal drugs would be appropriate.
Dr. Fanaroff responded that it’s possible, and even likely, that some patients with self-reported angina had a noncardiac source of their chest pain. However, they would be in the minority. The fact is that multiple studies have shown that patients who report experiencing angina after PCI for acute MI have about a 1.5-times increased risk of readmission and repeat revascularization, a 2-times increased risk of developing new depressive symptoms, and diminished quality of life scores.
The TRANSLATE-ACS study was sponsored by Eli Lilly. Dr. Fanaroff reported having no financial conflicts.
WASHINGTON – Non–beta-blocker antianginal drugs are seriously underprescribed in patients with angina following percutaneous coronary intervention (PCI) for a myocardial infarction (MI), according to a large national study, Alexander C. Fanaroff, MD, said at the annual meeting of the American College of Cardiology.
Clinical practice in this regard appears to be largely out of touch with current evidence-based guidelines regarding angina management in patients with stable ischemic heart disease. Those ACC/American Heart Association guidelines recommend a stepwise approach beginning with a beta-blocker and sublingual nitroglycerin, adding a calcium channel blocker if the beta-blocker isn’t tolerated or effective, further adding a long-acting nitrate if symptoms persist, incorporating ranolazine (Ranexa) as needed, and finally turning to revascularization for symptomatic relief if multidrug therapy proves inadequate (J Am Coll Cardiol. 2012 Dec 18;60[24]:e44-e164).
“When you ask patients and physicians separately about the prevalence of angina, they tend to disagree, with physicians consistently thinking their patients have less angina than patients report themselves. Together, with our data, this indicates that strategies to improve clinician recognition and treatment of angina symptoms are needed,” he asserted.
TRANSLATE-ACS was a large, longitudinal, observational study of patients who underwent PCI at 233 U.S. hospitals during 2010-2012. Dr. Fanaroff focused on 12-month follow-up data on the 10,870 patients whose PCI was for treatment of an acute MI. In telephone interviews 6 weeks postdischarge which incorporated the validated Seattle Angina Questionnaire, 3,190 of these individuals (31%) with stable ischemic heart disease reported angina symptoms. Of those, 79% said their symptoms occurred at least monthly, 18% weekly, and 3% daily. Yet, only 1 in 5 patients with angina 6 weeks postdischarge were on a non–beta-blocker antianginal medication: 16% were on monotherapy with a calcium channel blocker or long-acting nitrate, 3% were on dual therapy, and less than 1% were on three non–beta-blockers for their angina.
The prevalence of angina dropped over time. At 6 months postdischarge, 40% of the original cohort of patients who had angina at 6 weeks still reported angina. At 12 months, it was 33%. Meanwhile, the use of non–beta-blocker antianginal agents crept up modestly over time, from 20% in patients with angina at 6 weeks postdischarge, to 22% in those with angina at 6 months, and 23% at 12 months.
On the other hand, 33% of patients with angina at 6 weeks had persistent angina through 12 months. Remarkably, 69% of these individuals never received a non–beta-blocker antianginal drug during all that time, including 61% of those who experienced daily or weekly angina.
The use of non–beta-blocker antianginal drugs was more frequent in patients with more severe angina: 25% of patients with daily or weekly angina at 6 weeks were on such medication, as were 34% at 6 months and 38% at 12 months, compared with 16%, 18%, and 22% of those with monthly angina. Still, most of these patients were on only a single non–beta-blocker antianginal agent.
Of patients with angina at 6 weeks, 12% subsequently underwent repeat PCI at some point during the first year postdischarge. Of these individuals, 19% were on a single non–beta-blocker antianginal drug at the time of their procedure, 6% were on two such drugs, and only 1% were on three.
Roughly 10% of patients with multivessel coronary disease who were identified angiographically at the time of their acute MI did not undergo multivessel PCI at that time. This was not predictive of angina 6 weeks post discharge, Dr. Fanaroff continued.
One audience member asked Dr. Fanaroff how he knows that the patients with self-reported angina didn’t actually have noncardiac chest pain, in which case their physicians’ decision not to prescribe second- and third-tier antianginal drugs would be appropriate.
Dr. Fanaroff responded that it’s possible, and even likely, that some patients with self-reported angina had a noncardiac source of their chest pain. However, they would be in the minority. The fact is that multiple studies have shown that patients who report experiencing angina after PCI for acute MI have about a 1.5-times increased risk of readmission and repeat revascularization, a 2-times increased risk of developing new depressive symptoms, and diminished quality of life scores.
The TRANSLATE-ACS study was sponsored by Eli Lilly. Dr. Fanaroff reported having no financial conflicts.
WASHINGTON – Non–beta-blocker antianginal drugs are seriously underprescribed in patients with angina following percutaneous coronary intervention (PCI) for a myocardial infarction (MI), according to a large national study, Alexander C. Fanaroff, MD, said at the annual meeting of the American College of Cardiology.
Clinical practice in this regard appears to be largely out of touch with current evidence-based guidelines regarding angina management in patients with stable ischemic heart disease. Those ACC/American Heart Association guidelines recommend a stepwise approach beginning with a beta-blocker and sublingual nitroglycerin, adding a calcium channel blocker if the beta-blocker isn’t tolerated or effective, further adding a long-acting nitrate if symptoms persist, incorporating ranolazine (Ranexa) as needed, and finally turning to revascularization for symptomatic relief if multidrug therapy proves inadequate (J Am Coll Cardiol. 2012 Dec 18;60[24]:e44-e164).
“When you ask patients and physicians separately about the prevalence of angina, they tend to disagree, with physicians consistently thinking their patients have less angina than patients report themselves. Together, with our data, this indicates that strategies to improve clinician recognition and treatment of angina symptoms are needed,” he asserted.
TRANSLATE-ACS was a large, longitudinal, observational study of patients who underwent PCI at 233 U.S. hospitals during 2010-2012. Dr. Fanaroff focused on 12-month follow-up data on the 10,870 patients whose PCI was for treatment of an acute MI. In telephone interviews 6 weeks postdischarge which incorporated the validated Seattle Angina Questionnaire, 3,190 of these individuals (31%) with stable ischemic heart disease reported angina symptoms. Of those, 79% said their symptoms occurred at least monthly, 18% weekly, and 3% daily. Yet, only 1 in 5 patients with angina 6 weeks postdischarge were on a non–beta-blocker antianginal medication: 16% were on monotherapy with a calcium channel blocker or long-acting nitrate, 3% were on dual therapy, and less than 1% were on three non–beta-blockers for their angina.
The prevalence of angina dropped over time. At 6 months postdischarge, 40% of the original cohort of patients who had angina at 6 weeks still reported angina. At 12 months, it was 33%. Meanwhile, the use of non–beta-blocker antianginal agents crept up modestly over time, from 20% in patients with angina at 6 weeks postdischarge, to 22% in those with angina at 6 months, and 23% at 12 months.
On the other hand, 33% of patients with angina at 6 weeks had persistent angina through 12 months. Remarkably, 69% of these individuals never received a non–beta-blocker antianginal drug during all that time, including 61% of those who experienced daily or weekly angina.
The use of non–beta-blocker antianginal drugs was more frequent in patients with more severe angina: 25% of patients with daily or weekly angina at 6 weeks were on such medication, as were 34% at 6 months and 38% at 12 months, compared with 16%, 18%, and 22% of those with monthly angina. Still, most of these patients were on only a single non–beta-blocker antianginal agent.
Of patients with angina at 6 weeks, 12% subsequently underwent repeat PCI at some point during the first year postdischarge. Of these individuals, 19% were on a single non–beta-blocker antianginal drug at the time of their procedure, 6% were on two such drugs, and only 1% were on three.
Roughly 10% of patients with multivessel coronary disease who were identified angiographically at the time of their acute MI did not undergo multivessel PCI at that time. This was not predictive of angina 6 weeks post discharge, Dr. Fanaroff continued.
One audience member asked Dr. Fanaroff how he knows that the patients with self-reported angina didn’t actually have noncardiac chest pain, in which case their physicians’ decision not to prescribe second- and third-tier antianginal drugs would be appropriate.
Dr. Fanaroff responded that it’s possible, and even likely, that some patients with self-reported angina had a noncardiac source of their chest pain. However, they would be in the minority. The fact is that multiple studies have shown that patients who report experiencing angina after PCI for acute MI have about a 1.5-times increased risk of readmission and repeat revascularization, a 2-times increased risk of developing new depressive symptoms, and diminished quality of life scores.
The TRANSLATE-ACS study was sponsored by Eli Lilly. Dr. Fanaroff reported having no financial conflicts.
AT ACC 17
Key clinical point:
Major finding: Only 31% of patients who reported persistent angina for the first 12 months post PCI for acute MI reported taking any non–beta-blocker antianginal drugs during that period.
Data source: An analysis of nearly 11,000 patients who underwent PCI for an acute MI at 233 U.S. hospitals in the longitudinal observational TRANSLATE-ACS study.
Disclosures: The TRANSLATE-ACS study was sponsored by Eli Lilly. The presenter reported having no financial conflicts.
How to get the most out of methotrexate for psoriasis
WAILEA, HAWAII – In an era when affordable health insurance could become increasingly tough to come by, it’s worth emphasizing that methotrexate is the most cost-effective way to manage extensive psoriasis – and the liquid formulation designed for intramuscular injection can be taken orally to reduce the cost even further, according to Craig L. Leonardi, MD.
“The absolute cheapest way to manage the patient who has no insurance and has bad psoriasis is to put him on methotrexate and teach him how to draw the liquid solution up in a syringe, dump it in a cup of juice, and drink it,” Dr. Leonardi said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Research Foundation. “The bioavailability is equivalent to [that of] the tablets, and it’s only about one-tenth the cost.”
He urged his fellow dermatologists not to forget about methotrexate in the current flashy era of highly effective – and very expensive – biologic therapies for psoriasis.
“I was thinking methotrexate was going to go away, but it turns out I rely more on that drug than ever before. It’s very useful, and it’s safe if used correctly,” said Dr. Leonardi, a dermatologist at Saint Louis University and a prominent clinical trialist.
He highlighted numerous clinical scenarios in which methotrexate remains a valuable treatment in patients with moderate to severe psoriasis. He also touched on patient monitoring requirements, adverse events, and common reasons he receives referrals from physicians whose patients seem to be having problems on methotrexate – referrals that, in most cases, could be avoided, he said, if the referring physician had a fuller understanding of the drug.
“Patients are referred to me all the time for methotrexate intolerance,” Dr. Leonardi said. “When you take methotrexate, you get a brief, dramatic spike in transaminase levels that peaks within a day and then drops off. But, when you get lab tests in these patients, you want to look at trough levels. You want to see the best liver function test values for the week.
“That means you have to tell the patient what day to take the drug and what day to get labs drawn,” he continued. “I’m here to tell you that the vast majority of issues that I see where patients have elevated liver function tests involves them getting their testing done in the first 4 days after taking methotrexate. Take the time to ask about this, and I think you’ll be pleasantly surprised.
“If you just make an adjustment and get them on the right schedule, you’ll discover that the patient is tolerating the drug just fine,” Dr. Leonardi added. “We like getting labs on Monday and dosing on Tuesday.”
Methotrexate’s half-life is 3-15 hours. Psoriasis is often controlled at a methotrexate dose of about 15 mg/week.
Methotrexate’s advantages include ease of use. It’s a straightforward matter to start and stop the drug and to make small dose adjustments. Methotrexate comes in a variety of formulations: the 2.5-mg tablets, the 25 mg/mL solution for IM injection, and prefilled autoinjector devices for subcutaneous administration.
These preloaded pens for subcutaneous injection are just as effective as IM therapy, Dr. Leonardi said. Methotrexate is also better absorbed subcutaneously than it is orally. The bioavailability of oral methotrexate plateaus at a dose of about 22 mg/week, while subcutaneously delivered methotrexate does not. So, if a patient’s psoriasis isn’t adequately controlled on higher-dose oral therapy, it’s worth considering a switch to the preloaded pens.
“It’s a very simple injection, very cost-effective, and your patients may get more bang for the buck by doing that,” according to the dermatologist.
Also, nausea, vomiting, diarrhea, and abdominal pain have been shown to be significantly less frequent and intense with subcutaneous methotrexate than with the tablets. So, if a patient is experiencing limiting gastrointestinal issues on methotrexate tablets, a shift to subcutaneous therapy is often the solution.
What kind of efficacy can physicians expect with methotrexate monotherapy?
Dr. Leonardi cited the results of the European randomized, open-label RESTORE-1 trial (Br J Dermatol. 2011 Nov;165[5]:1109-17) as being consistent with his own extensive clinical experience: a week-16 PASI (Psoriasis Area and Severity Index ) 75 response rate of 42% with methotrexate, compared with 78% for infliximab (Remicade).
Of course, some patients can’t receive a biologic agent because of their age, lack of insurance coverage, or medical contraindications.
“I use methotrexate a lot in Medicare patients, where, with Part D, it’s hard to get access to biologics without really good coinsurance. I think all of us who prescribe biologics understand that,” he observed.
In pediatric patients with extensive psoriasis, he turns to methotrexate as first-line systemic therapy. After 3 months, if the young patient hasn’t responded satisfactorily, Dr. Leonardi asks the insurance company for access to a biologic agent and usually gets it.
Methotrexate really shines in combination with a biologic agent. It inhibits formation of antibiologic antibodies, an important cause of loss of effectiveness of monoclonal antibody therapy.
In one study, 28% of patients on adalimumab (Humira) developed antiadalimumab antibodies during the first 3 years of therapy. These patients were more likely to drop out of therapy for lack of effectiveness.
A key finding in this study was that two-thirds of patients who developed antiadalimumab antibodies did so during the first 28 weeks of therapy (JAMA. 2011 Apr 13;305[14]:1460-8). This time line has influenced Dr. Leonardi’s own clinical practice. He routinely keeps patients on methotrexate for their first 28-36 weeks on a biologic, then tapers the methotrexate in favor of biologic monotherapy.
Other benefits and guidelines
Methotrexate is also a boon in managing psoriasis flares in patients on a biologic.
“We’ve all had patients who are doing well on a biologic, they’re cruising along at 140 weeks, then they get strep throat or another infection, and, next thing you know, you have destabilized psoriasis,” Dr. Leonardi noted.
“One thing I’ll do is add methotrexate to the mix, try to get things under control, and, if we do, then we’ll try to taper the methotrexate,” he continued. “That whole episode might take 4-6 months to resolve before the patient might be able to tolerate biologic monotherapy, though. If they can’t at that point, you might consider rotating to another biologic.”
Current American Academy of Dermatology guidelines recommend that women on methotrexate limit their alcohol intake to one drink per day, two drinks for men. British Society for Rheumatology guidelines recommend a ceiling of 32 g to 64 g of ethanol per week.
“We don’t insist on abstinence. There’s no good evidence that it’s needed,” Dr. Leonardi noted. “If you dose this drug on Tuesday, you can be sure that it’s eliminated from the body on Friday, and that’s when I’ll generally green-light patients to socialize over the weekend. If you can make life a little more tolerable for your patients and they’re willing to follow your instructions, I think it’s a better deal all the way around.”
Prior to initiating methotrexate, he obtains a CBC with platelet count, liver function tests, serum urea nitrogen, creatinine, and screens for latent tuberculosis. In terms of on-treatment monitoring, he gets a CBC and liver function tests every 4-12 weeks and keeps an eye on renal function, especially in older patients, because methotrexate is eliminated renally.
“The guidelines have relaxed regarding the need for liver biopsies,” Dr. Leonardi said. “Most of us are not getting liver biopsies anymore, as is true of our friends in rheumatology.”
He reported having financial relationships with more than a dozen pharmaceutical companies. The SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – In an era when affordable health insurance could become increasingly tough to come by, it’s worth emphasizing that methotrexate is the most cost-effective way to manage extensive psoriasis – and the liquid formulation designed for intramuscular injection can be taken orally to reduce the cost even further, according to Craig L. Leonardi, MD.
“The absolute cheapest way to manage the patient who has no insurance and has bad psoriasis is to put him on methotrexate and teach him how to draw the liquid solution up in a syringe, dump it in a cup of juice, and drink it,” Dr. Leonardi said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Research Foundation. “The bioavailability is equivalent to [that of] the tablets, and it’s only about one-tenth the cost.”
He urged his fellow dermatologists not to forget about methotrexate in the current flashy era of highly effective – and very expensive – biologic therapies for psoriasis.
“I was thinking methotrexate was going to go away, but it turns out I rely more on that drug than ever before. It’s very useful, and it’s safe if used correctly,” said Dr. Leonardi, a dermatologist at Saint Louis University and a prominent clinical trialist.
He highlighted numerous clinical scenarios in which methotrexate remains a valuable treatment in patients with moderate to severe psoriasis. He also touched on patient monitoring requirements, adverse events, and common reasons he receives referrals from physicians whose patients seem to be having problems on methotrexate – referrals that, in most cases, could be avoided, he said, if the referring physician had a fuller understanding of the drug.
“Patients are referred to me all the time for methotrexate intolerance,” Dr. Leonardi said. “When you take methotrexate, you get a brief, dramatic spike in transaminase levels that peaks within a day and then drops off. But, when you get lab tests in these patients, you want to look at trough levels. You want to see the best liver function test values for the week.
“That means you have to tell the patient what day to take the drug and what day to get labs drawn,” he continued. “I’m here to tell you that the vast majority of issues that I see where patients have elevated liver function tests involves them getting their testing done in the first 4 days after taking methotrexate. Take the time to ask about this, and I think you’ll be pleasantly surprised.
“If you just make an adjustment and get them on the right schedule, you’ll discover that the patient is tolerating the drug just fine,” Dr. Leonardi added. “We like getting labs on Monday and dosing on Tuesday.”
Methotrexate’s half-life is 3-15 hours. Psoriasis is often controlled at a methotrexate dose of about 15 mg/week.
Methotrexate’s advantages include ease of use. It’s a straightforward matter to start and stop the drug and to make small dose adjustments. Methotrexate comes in a variety of formulations: the 2.5-mg tablets, the 25 mg/mL solution for IM injection, and prefilled autoinjector devices for subcutaneous administration.
These preloaded pens for subcutaneous injection are just as effective as IM therapy, Dr. Leonardi said. Methotrexate is also better absorbed subcutaneously than it is orally. The bioavailability of oral methotrexate plateaus at a dose of about 22 mg/week, while subcutaneously delivered methotrexate does not. So, if a patient’s psoriasis isn’t adequately controlled on higher-dose oral therapy, it’s worth considering a switch to the preloaded pens.
“It’s a very simple injection, very cost-effective, and your patients may get more bang for the buck by doing that,” according to the dermatologist.
Also, nausea, vomiting, diarrhea, and abdominal pain have been shown to be significantly less frequent and intense with subcutaneous methotrexate than with the tablets. So, if a patient is experiencing limiting gastrointestinal issues on methotrexate tablets, a shift to subcutaneous therapy is often the solution.
What kind of efficacy can physicians expect with methotrexate monotherapy?
Dr. Leonardi cited the results of the European randomized, open-label RESTORE-1 trial (Br J Dermatol. 2011 Nov;165[5]:1109-17) as being consistent with his own extensive clinical experience: a week-16 PASI (Psoriasis Area and Severity Index ) 75 response rate of 42% with methotrexate, compared with 78% for infliximab (Remicade).
Of course, some patients can’t receive a biologic agent because of their age, lack of insurance coverage, or medical contraindications.
“I use methotrexate a lot in Medicare patients, where, with Part D, it’s hard to get access to biologics without really good coinsurance. I think all of us who prescribe biologics understand that,” he observed.
In pediatric patients with extensive psoriasis, he turns to methotrexate as first-line systemic therapy. After 3 months, if the young patient hasn’t responded satisfactorily, Dr. Leonardi asks the insurance company for access to a biologic agent and usually gets it.
Methotrexate really shines in combination with a biologic agent. It inhibits formation of antibiologic antibodies, an important cause of loss of effectiveness of monoclonal antibody therapy.
In one study, 28% of patients on adalimumab (Humira) developed antiadalimumab antibodies during the first 3 years of therapy. These patients were more likely to drop out of therapy for lack of effectiveness.
A key finding in this study was that two-thirds of patients who developed antiadalimumab antibodies did so during the first 28 weeks of therapy (JAMA. 2011 Apr 13;305[14]:1460-8). This time line has influenced Dr. Leonardi’s own clinical practice. He routinely keeps patients on methotrexate for their first 28-36 weeks on a biologic, then tapers the methotrexate in favor of biologic monotherapy.
Other benefits and guidelines
Methotrexate is also a boon in managing psoriasis flares in patients on a biologic.
“We’ve all had patients who are doing well on a biologic, they’re cruising along at 140 weeks, then they get strep throat or another infection, and, next thing you know, you have destabilized psoriasis,” Dr. Leonardi noted.
“One thing I’ll do is add methotrexate to the mix, try to get things under control, and, if we do, then we’ll try to taper the methotrexate,” he continued. “That whole episode might take 4-6 months to resolve before the patient might be able to tolerate biologic monotherapy, though. If they can’t at that point, you might consider rotating to another biologic.”
Current American Academy of Dermatology guidelines recommend that women on methotrexate limit their alcohol intake to one drink per day, two drinks for men. British Society for Rheumatology guidelines recommend a ceiling of 32 g to 64 g of ethanol per week.
“We don’t insist on abstinence. There’s no good evidence that it’s needed,” Dr. Leonardi noted. “If you dose this drug on Tuesday, you can be sure that it’s eliminated from the body on Friday, and that’s when I’ll generally green-light patients to socialize over the weekend. If you can make life a little more tolerable for your patients and they’re willing to follow your instructions, I think it’s a better deal all the way around.”
Prior to initiating methotrexate, he obtains a CBC with platelet count, liver function tests, serum urea nitrogen, creatinine, and screens for latent tuberculosis. In terms of on-treatment monitoring, he gets a CBC and liver function tests every 4-12 weeks and keeps an eye on renal function, especially in older patients, because methotrexate is eliminated renally.
“The guidelines have relaxed regarding the need for liver biopsies,” Dr. Leonardi said. “Most of us are not getting liver biopsies anymore, as is true of our friends in rheumatology.”
He reported having financial relationships with more than a dozen pharmaceutical companies. The SDEF and this news organization are owned by the same parent company.
WAILEA, HAWAII – In an era when affordable health insurance could become increasingly tough to come by, it’s worth emphasizing that methotrexate is the most cost-effective way to manage extensive psoriasis – and the liquid formulation designed for intramuscular injection can be taken orally to reduce the cost even further, according to Craig L. Leonardi, MD.
“The absolute cheapest way to manage the patient who has no insurance and has bad psoriasis is to put him on methotrexate and teach him how to draw the liquid solution up in a syringe, dump it in a cup of juice, and drink it,” Dr. Leonardi said at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Research Foundation. “The bioavailability is equivalent to [that of] the tablets, and it’s only about one-tenth the cost.”
He urged his fellow dermatologists not to forget about methotrexate in the current flashy era of highly effective – and very expensive – biologic therapies for psoriasis.
“I was thinking methotrexate was going to go away, but it turns out I rely more on that drug than ever before. It’s very useful, and it’s safe if used correctly,” said Dr. Leonardi, a dermatologist at Saint Louis University and a prominent clinical trialist.
He highlighted numerous clinical scenarios in which methotrexate remains a valuable treatment in patients with moderate to severe psoriasis. He also touched on patient monitoring requirements, adverse events, and common reasons he receives referrals from physicians whose patients seem to be having problems on methotrexate – referrals that, in most cases, could be avoided, he said, if the referring physician had a fuller understanding of the drug.
“Patients are referred to me all the time for methotrexate intolerance,” Dr. Leonardi said. “When you take methotrexate, you get a brief, dramatic spike in transaminase levels that peaks within a day and then drops off. But, when you get lab tests in these patients, you want to look at trough levels. You want to see the best liver function test values for the week.
“That means you have to tell the patient what day to take the drug and what day to get labs drawn,” he continued. “I’m here to tell you that the vast majority of issues that I see where patients have elevated liver function tests involves them getting their testing done in the first 4 days after taking methotrexate. Take the time to ask about this, and I think you’ll be pleasantly surprised.
“If you just make an adjustment and get them on the right schedule, you’ll discover that the patient is tolerating the drug just fine,” Dr. Leonardi added. “We like getting labs on Monday and dosing on Tuesday.”
Methotrexate’s half-life is 3-15 hours. Psoriasis is often controlled at a methotrexate dose of about 15 mg/week.
Methotrexate’s advantages include ease of use. It’s a straightforward matter to start and stop the drug and to make small dose adjustments. Methotrexate comes in a variety of formulations: the 2.5-mg tablets, the 25 mg/mL solution for IM injection, and prefilled autoinjector devices for subcutaneous administration.
These preloaded pens for subcutaneous injection are just as effective as IM therapy, Dr. Leonardi said. Methotrexate is also better absorbed subcutaneously than it is orally. The bioavailability of oral methotrexate plateaus at a dose of about 22 mg/week, while subcutaneously delivered methotrexate does not. So, if a patient’s psoriasis isn’t adequately controlled on higher-dose oral therapy, it’s worth considering a switch to the preloaded pens.
“It’s a very simple injection, very cost-effective, and your patients may get more bang for the buck by doing that,” according to the dermatologist.
Also, nausea, vomiting, diarrhea, and abdominal pain have been shown to be significantly less frequent and intense with subcutaneous methotrexate than with the tablets. So, if a patient is experiencing limiting gastrointestinal issues on methotrexate tablets, a shift to subcutaneous therapy is often the solution.
What kind of efficacy can physicians expect with methotrexate monotherapy?
Dr. Leonardi cited the results of the European randomized, open-label RESTORE-1 trial (Br J Dermatol. 2011 Nov;165[5]:1109-17) as being consistent with his own extensive clinical experience: a week-16 PASI (Psoriasis Area and Severity Index ) 75 response rate of 42% with methotrexate, compared with 78% for infliximab (Remicade).
Of course, some patients can’t receive a biologic agent because of their age, lack of insurance coverage, or medical contraindications.
“I use methotrexate a lot in Medicare patients, where, with Part D, it’s hard to get access to biologics without really good coinsurance. I think all of us who prescribe biologics understand that,” he observed.
In pediatric patients with extensive psoriasis, he turns to methotrexate as first-line systemic therapy. After 3 months, if the young patient hasn’t responded satisfactorily, Dr. Leonardi asks the insurance company for access to a biologic agent and usually gets it.
Methotrexate really shines in combination with a biologic agent. It inhibits formation of antibiologic antibodies, an important cause of loss of effectiveness of monoclonal antibody therapy.
In one study, 28% of patients on adalimumab (Humira) developed antiadalimumab antibodies during the first 3 years of therapy. These patients were more likely to drop out of therapy for lack of effectiveness.
A key finding in this study was that two-thirds of patients who developed antiadalimumab antibodies did so during the first 28 weeks of therapy (JAMA. 2011 Apr 13;305[14]:1460-8). This time line has influenced Dr. Leonardi’s own clinical practice. He routinely keeps patients on methotrexate for their first 28-36 weeks on a biologic, then tapers the methotrexate in favor of biologic monotherapy.
Other benefits and guidelines
Methotrexate is also a boon in managing psoriasis flares in patients on a biologic.
“We’ve all had patients who are doing well on a biologic, they’re cruising along at 140 weeks, then they get strep throat or another infection, and, next thing you know, you have destabilized psoriasis,” Dr. Leonardi noted.
“One thing I’ll do is add methotrexate to the mix, try to get things under control, and, if we do, then we’ll try to taper the methotrexate,” he continued. “That whole episode might take 4-6 months to resolve before the patient might be able to tolerate biologic monotherapy, though. If they can’t at that point, you might consider rotating to another biologic.”
Current American Academy of Dermatology guidelines recommend that women on methotrexate limit their alcohol intake to one drink per day, two drinks for men. British Society for Rheumatology guidelines recommend a ceiling of 32 g to 64 g of ethanol per week.
“We don’t insist on abstinence. There’s no good evidence that it’s needed,” Dr. Leonardi noted. “If you dose this drug on Tuesday, you can be sure that it’s eliminated from the body on Friday, and that’s when I’ll generally green-light patients to socialize over the weekend. If you can make life a little more tolerable for your patients and they’re willing to follow your instructions, I think it’s a better deal all the way around.”
Prior to initiating methotrexate, he obtains a CBC with platelet count, liver function tests, serum urea nitrogen, creatinine, and screens for latent tuberculosis. In terms of on-treatment monitoring, he gets a CBC and liver function tests every 4-12 weeks and keeps an eye on renal function, especially in older patients, because methotrexate is eliminated renally.
“The guidelines have relaxed regarding the need for liver biopsies,” Dr. Leonardi said. “Most of us are not getting liver biopsies anymore, as is true of our friends in rheumatology.”
He reported having financial relationships with more than a dozen pharmaceutical companies. The SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE SDEF HAWAII DERMATOLOGY SEMINAR












