User login
EPO improves cognitive impairment in depression
VIENNA – Recombinant human erythropoietin appears to be a cognitive enhancer in patients with unipolar or bipolar depression accompanied by cognitive impairment, Kamilla W. Miskowiak, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
She presented a pooled analysis of her two pilot randomized, placebo-controlled clinical trials totaling 79 patients with treatment-resistant unipolar or partly remitted bipolar depression associated with baseline cognitive impairment. She found that 8 weeks of once-weekly intravenous infusions of high-dose human recombinant erythropoetin (Epogen), also known as epoetin alfa (EPO), resulted in clinically meaningful improvement in cognition 1 week post treatment, as measured on neuropsychological tests assessing the speed of complex cognitive processing, verbal memory, attention span, concentration, working memory, and strategic planning ability. Saline infusions had no effect.
Moreover, follow-up testing conducted 6 weeks after the final treatment session showed continued full maintenance of the initial cognitive gains, which averaged an 11% improvement over baseline. The full duration of effect will be determined in the next round of studies, which will feature 12 weeks of EPO and 6 months of off-treatment follow-up, added Dr. Miskowiak, a senior research psychologist at the University of Copenhagen.
In an interview, she emphasized that the mechanism of benefit doesn’t appear to be related to EPO’s signature ability to boost red blood cell (RBC) production and raise hemoglobin levels. After all, she noted, RBCs were back to normal within 2 weeks after treatment ended, yet the objective cognitive improvements continued. Instead, the mechanism involves EPO’s capacity to enhance brain neuroplasticity, an effect that was demonstrated first in animal studies and subsequently in Dr. Miskowiak’s clinical trials.
“We did MRI functional and structural brain scans at baseline and after completion of treatment, and we saw that EPO increased left hippocampal volume. In fact, it was the structural change in hippocampal volume in response to EPO that was the single strongest predictor of cognitive improvement in multivariate analyses adjusted for age, illness chronicity, depressive symptom severity, gender, and other factors,” according to Dr. Miskowiak.
Physicians who use EPO to treat anemia might blanch at the doses of EPO administered to improve cognitive impairments in patients with affective disorders. Instead of giving 50-100 U/kg of body weight several times per week, as is common in treating anemia, the patients in her studies typically received 40,000 U/wk.
“We used much, much higher doses than are given to affect the bone marrow in patients with anemia, because only about 1% of the IV dose passes across the blood/brain barrier,” she explained.
Because EPO, even in conventional doses, is associated with an increased risk of blood clots, patients were ineligible for the cognition-improvement trials if they had a history of venous thromboembolism, coronary disease, or cancer. Moreover, their RBC and platelet activity levels were monitored on a weekly basis. No untoward effects were seen, Dr. Miskowiak continued.
“Patients really liked coming in for this therapy. Many of them were quite chronic, with years of illness, yet adherence was excellent,” she said.
Early in the developmental pipeline are an intranasal variant of EPO and a carbamylated EPO designed to cross the blood/brain barrier efficiently in order to achieve direct brain neuroplasticity-stimulating effects with minimal impact on RBC production.
Dr. Miskowiak stressed that EPO as a treatment for cognitive dysfunction associated with affective disorders is not ready for prime time use in clinical practice. The next round of randomized, placebo-controlled clinical trials, due to start early in 2017, will attempt to replicate her pilot study findings while pinning down the optimal dosing regimen. The new trials will feature longer off-treatment follow-up as well as quality-of-life measures and an economic analysis that includes assessment of the ability to return to work.
“This therapy may be attractive from an economic perspective,” according to the psychologist. “I don’t see this as first-line therapy in clinical practice, though,” she added. “It will be reserved for the more severely cognitively impaired patients.”
Eduard Vieta, MD, professor and chair of the department of psychiatry and psychology at the University of Barcelona, commented that cognitive impairment is now recognized as a core aspect of affective disorders. There is a huge unmet need for treatments that address this disease aspect. The drugs now available for treatment of bipolar disorder and unipolar depression don’t improve the associated cognitive impairments. Indeed, up to 40% of patients in remission from major depressive disorder and 70% in remission from bipolar disorder continue to experience neurocognitive symptoms.
Dr. Miskowiak’s work was funded by the Danish Ministry of Science, Innovation, and Higher Education. She reported having no relevant financial conflicts of interest.
VIENNA – Recombinant human erythropoietin appears to be a cognitive enhancer in patients with unipolar or bipolar depression accompanied by cognitive impairment, Kamilla W. Miskowiak, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
She presented a pooled analysis of her two pilot randomized, placebo-controlled clinical trials totaling 79 patients with treatment-resistant unipolar or partly remitted bipolar depression associated with baseline cognitive impairment. She found that 8 weeks of once-weekly intravenous infusions of high-dose human recombinant erythropoetin (Epogen), also known as epoetin alfa (EPO), resulted in clinically meaningful improvement in cognition 1 week post treatment, as measured on neuropsychological tests assessing the speed of complex cognitive processing, verbal memory, attention span, concentration, working memory, and strategic planning ability. Saline infusions had no effect.
Moreover, follow-up testing conducted 6 weeks after the final treatment session showed continued full maintenance of the initial cognitive gains, which averaged an 11% improvement over baseline. The full duration of effect will be determined in the next round of studies, which will feature 12 weeks of EPO and 6 months of off-treatment follow-up, added Dr. Miskowiak, a senior research psychologist at the University of Copenhagen.
In an interview, she emphasized that the mechanism of benefit doesn’t appear to be related to EPO’s signature ability to boost red blood cell (RBC) production and raise hemoglobin levels. After all, she noted, RBCs were back to normal within 2 weeks after treatment ended, yet the objective cognitive improvements continued. Instead, the mechanism involves EPO’s capacity to enhance brain neuroplasticity, an effect that was demonstrated first in animal studies and subsequently in Dr. Miskowiak’s clinical trials.
“We did MRI functional and structural brain scans at baseline and after completion of treatment, and we saw that EPO increased left hippocampal volume. In fact, it was the structural change in hippocampal volume in response to EPO that was the single strongest predictor of cognitive improvement in multivariate analyses adjusted for age, illness chronicity, depressive symptom severity, gender, and other factors,” according to Dr. Miskowiak.
Physicians who use EPO to treat anemia might blanch at the doses of EPO administered to improve cognitive impairments in patients with affective disorders. Instead of giving 50-100 U/kg of body weight several times per week, as is common in treating anemia, the patients in her studies typically received 40,000 U/wk.
“We used much, much higher doses than are given to affect the bone marrow in patients with anemia, because only about 1% of the IV dose passes across the blood/brain barrier,” she explained.
Because EPO, even in conventional doses, is associated with an increased risk of blood clots, patients were ineligible for the cognition-improvement trials if they had a history of venous thromboembolism, coronary disease, or cancer. Moreover, their RBC and platelet activity levels were monitored on a weekly basis. No untoward effects were seen, Dr. Miskowiak continued.
“Patients really liked coming in for this therapy. Many of them were quite chronic, with years of illness, yet adherence was excellent,” she said.
Early in the developmental pipeline are an intranasal variant of EPO and a carbamylated EPO designed to cross the blood/brain barrier efficiently in order to achieve direct brain neuroplasticity-stimulating effects with minimal impact on RBC production.
Dr. Miskowiak stressed that EPO as a treatment for cognitive dysfunction associated with affective disorders is not ready for prime time use in clinical practice. The next round of randomized, placebo-controlled clinical trials, due to start early in 2017, will attempt to replicate her pilot study findings while pinning down the optimal dosing regimen. The new trials will feature longer off-treatment follow-up as well as quality-of-life measures and an economic analysis that includes assessment of the ability to return to work.
“This therapy may be attractive from an economic perspective,” according to the psychologist. “I don’t see this as first-line therapy in clinical practice, though,” she added. “It will be reserved for the more severely cognitively impaired patients.”
Eduard Vieta, MD, professor and chair of the department of psychiatry and psychology at the University of Barcelona, commented that cognitive impairment is now recognized as a core aspect of affective disorders. There is a huge unmet need for treatments that address this disease aspect. The drugs now available for treatment of bipolar disorder and unipolar depression don’t improve the associated cognitive impairments. Indeed, up to 40% of patients in remission from major depressive disorder and 70% in remission from bipolar disorder continue to experience neurocognitive symptoms.
Dr. Miskowiak’s work was funded by the Danish Ministry of Science, Innovation, and Higher Education. She reported having no relevant financial conflicts of interest.
VIENNA – Recombinant human erythropoietin appears to be a cognitive enhancer in patients with unipolar or bipolar depression accompanied by cognitive impairment, Kamilla W. Miskowiak, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
She presented a pooled analysis of her two pilot randomized, placebo-controlled clinical trials totaling 79 patients with treatment-resistant unipolar or partly remitted bipolar depression associated with baseline cognitive impairment. She found that 8 weeks of once-weekly intravenous infusions of high-dose human recombinant erythropoetin (Epogen), also known as epoetin alfa (EPO), resulted in clinically meaningful improvement in cognition 1 week post treatment, as measured on neuropsychological tests assessing the speed of complex cognitive processing, verbal memory, attention span, concentration, working memory, and strategic planning ability. Saline infusions had no effect.
Moreover, follow-up testing conducted 6 weeks after the final treatment session showed continued full maintenance of the initial cognitive gains, which averaged an 11% improvement over baseline. The full duration of effect will be determined in the next round of studies, which will feature 12 weeks of EPO and 6 months of off-treatment follow-up, added Dr. Miskowiak, a senior research psychologist at the University of Copenhagen.
In an interview, she emphasized that the mechanism of benefit doesn’t appear to be related to EPO’s signature ability to boost red blood cell (RBC) production and raise hemoglobin levels. After all, she noted, RBCs were back to normal within 2 weeks after treatment ended, yet the objective cognitive improvements continued. Instead, the mechanism involves EPO’s capacity to enhance brain neuroplasticity, an effect that was demonstrated first in animal studies and subsequently in Dr. Miskowiak’s clinical trials.
“We did MRI functional and structural brain scans at baseline and after completion of treatment, and we saw that EPO increased left hippocampal volume. In fact, it was the structural change in hippocampal volume in response to EPO that was the single strongest predictor of cognitive improvement in multivariate analyses adjusted for age, illness chronicity, depressive symptom severity, gender, and other factors,” according to Dr. Miskowiak.
Physicians who use EPO to treat anemia might blanch at the doses of EPO administered to improve cognitive impairments in patients with affective disorders. Instead of giving 50-100 U/kg of body weight several times per week, as is common in treating anemia, the patients in her studies typically received 40,000 U/wk.
“We used much, much higher doses than are given to affect the bone marrow in patients with anemia, because only about 1% of the IV dose passes across the blood/brain barrier,” she explained.
Because EPO, even in conventional doses, is associated with an increased risk of blood clots, patients were ineligible for the cognition-improvement trials if they had a history of venous thromboembolism, coronary disease, or cancer. Moreover, their RBC and platelet activity levels were monitored on a weekly basis. No untoward effects were seen, Dr. Miskowiak continued.
“Patients really liked coming in for this therapy. Many of them were quite chronic, with years of illness, yet adherence was excellent,” she said.
Early in the developmental pipeline are an intranasal variant of EPO and a carbamylated EPO designed to cross the blood/brain barrier efficiently in order to achieve direct brain neuroplasticity-stimulating effects with minimal impact on RBC production.
Dr. Miskowiak stressed that EPO as a treatment for cognitive dysfunction associated with affective disorders is not ready for prime time use in clinical practice. The next round of randomized, placebo-controlled clinical trials, due to start early in 2017, will attempt to replicate her pilot study findings while pinning down the optimal dosing regimen. The new trials will feature longer off-treatment follow-up as well as quality-of-life measures and an economic analysis that includes assessment of the ability to return to work.
“This therapy may be attractive from an economic perspective,” according to the psychologist. “I don’t see this as first-line therapy in clinical practice, though,” she added. “It will be reserved for the more severely cognitively impaired patients.”
Eduard Vieta, MD, professor and chair of the department of psychiatry and psychology at the University of Barcelona, commented that cognitive impairment is now recognized as a core aspect of affective disorders. There is a huge unmet need for treatments that address this disease aspect. The drugs now available for treatment of bipolar disorder and unipolar depression don’t improve the associated cognitive impairments. Indeed, up to 40% of patients in remission from major depressive disorder and 70% in remission from bipolar disorder continue to experience neurocognitive symptoms.
Dr. Miskowiak’s work was funded by the Danish Ministry of Science, Innovation, and Higher Education. She reported having no relevant financial conflicts of interest.
AT THE ECNP CONGRESS
Key clinical point: Recombinant human erythropoietin enhances brain neuroplasticity and achieves measurable improvements in cognitive impairment associated with unipolar or bipolar depression.
Major finding: Patients with cognitive impairment related to unipolar or bipolar depression responded to a series of infusions of recombinant human erythropoietin with a significant improvement in neuropsychological test scores that was maintained unabated 6 weeks after their final infusion.
Data source: A retrospective analysis of pooled data from two randomized, placebo-controlled trials totaling 79 patients with unipolar or bipolar depression.
Disclosures: This work was funded by the Danish Ministry of Science, Innovation, and Higher Education. The presenter reported having no relevant financial conflicts of interest.
Optimal medical therapy doesn’t affect DAPT efficacy
ROME – Continued dual-antiplatelet therapy beyond 12 months after coronary stenting is neither helped nor hindered by concomitant background optimal medical therapy, according to a secondary analysis of the landmark DAPT trial.
Numerous studies have shown that only 46%-66% of patients with stable ischemic heart disease are adherent to guideline-directed optimal medical therapy (OMT) with statins, beta-blockers, and ACE inhibitors or angiotensin receptor blockers. Yet until this new analysis from DAPT (the Dual Antiplatelet Therapy Study), the impact of OMT on the treatment effect of prolonged DAPT was unstudied, Charles D. Resor, MD, said at the annual congress of the European Society of Cardiology.
It was plausible that the treatment benefit of continued DAPT beyond 12 months after coronary stenting might be stunted by concomitant OMT, or alternatively that OMT and DAPT might exert synergistic effects in reducing the risk of ischemic events. As it turned out, neither of these possibilities turned out to be the case, according to Dr. Resor of Brigham and Women’s Hospital, Boston.
“I think OMT is underutilized, but while we would like physicians to emphasize OMT use in more cases, the decision to use DAPT beyond 12 months should be made irrespective of OMT use,” he said.
“That’s an important message for clinicians,” commented session cochair Keith A.A. Fox, MD, of the University of Edinburgh.
The landmark DAPT trial established that there is a significant benefit of continued dual-antiplatelet therapy beyond 12 months from coronary stenting in patients with stable ischemic heart disease (N Engl J Med. 2014 Dec 4;371:2155-66). Dr. Resor presented a secondary analysis of clinical outcomes in 11,643 study participants stratified by their OMT status as well as by whether they were randomized to dual-antiplatelet therapy or aspirin plus placebo.
Sixty-three percent of subjects were on OMT at enrollment. Adherence to OMT was high: At 30 months post stenting, 62% of participants remained on OMT.
Between 12 and 30 months after percutaneous coronary intervention, continued dual-antiplatelet therapy in patients also on OMT was associated with a 2.1% incidence of acute MI, a 36% reduction in risk, compared with the 3.3% rate in subjects on placebo plus OMT. In patients not on OMT, the MI rate was 2.2% in those on dual-antiplatelet therapy versus 5.2% with placebo, for a 59% relative risk reduction in the active treatment arm.
The rate of the composite endpoint of major adverse cardiovascular and cerebrovascular events in patients on dual-antiplatelet therapy and OMT was 4.2%, compared with a 5.0% rate in patients on placebo and OMT.
Moderate or severe bleeding, as measured by the Global Use of Strategies to Open Occluded Arteries (GUSTO) criteria occurred in 2.2% of patients on dual-antiplatelet therapy and OMT, compared with 1.0% in those on placebo plus OMT. In patients not on OMT, the bleeding rate was 2.8% in the dual-antiplatelet therapy group and 2.2% with placebo, a nonsignificant difference.
Overall, patients on OMT had significantly lower rates of acute MI than those not on OMT (2.7%, compared with 3.7%), as well as fewer major adverse cardiovascular and cerebrovascular events (4.6% versus 5.7%), and less moderate or severe bleeding (1.6%, compared with 2.5% in patients not on OMT). However, rates of stroke, stent thrombosis, and death didn’t differ between patients on OMT and those who were not.
“While the associations between OMT use and lower rates of MI and MACCE [major adverse cardiovascular and cerebrovascular events] were expected, the association with lower rates of moderate or severe bleeding was not; we suspect the lower bleeding risk is mostly due to some residual confounding,” according to Dr. Resor.
He reported having no financial conflicts of interest regarding the new secondary analysis of DAPT, which was sponsored by the Harvard Clinical Research Institute.
Simultaneously with Dr. Resor’s presentation, the new DAPT study analysis was published online (Circulation. 2016 Aug 30. doi: 10.1161/CIRCULATIONAHA.116.024531.)
ROME – Continued dual-antiplatelet therapy beyond 12 months after coronary stenting is neither helped nor hindered by concomitant background optimal medical therapy, according to a secondary analysis of the landmark DAPT trial.
Numerous studies have shown that only 46%-66% of patients with stable ischemic heart disease are adherent to guideline-directed optimal medical therapy (OMT) with statins, beta-blockers, and ACE inhibitors or angiotensin receptor blockers. Yet until this new analysis from DAPT (the Dual Antiplatelet Therapy Study), the impact of OMT on the treatment effect of prolonged DAPT was unstudied, Charles D. Resor, MD, said at the annual congress of the European Society of Cardiology.
It was plausible that the treatment benefit of continued DAPT beyond 12 months after coronary stenting might be stunted by concomitant OMT, or alternatively that OMT and DAPT might exert synergistic effects in reducing the risk of ischemic events. As it turned out, neither of these possibilities turned out to be the case, according to Dr. Resor of Brigham and Women’s Hospital, Boston.
“I think OMT is underutilized, but while we would like physicians to emphasize OMT use in more cases, the decision to use DAPT beyond 12 months should be made irrespective of OMT use,” he said.
“That’s an important message for clinicians,” commented session cochair Keith A.A. Fox, MD, of the University of Edinburgh.
The landmark DAPT trial established that there is a significant benefit of continued dual-antiplatelet therapy beyond 12 months from coronary stenting in patients with stable ischemic heart disease (N Engl J Med. 2014 Dec 4;371:2155-66). Dr. Resor presented a secondary analysis of clinical outcomes in 11,643 study participants stratified by their OMT status as well as by whether they were randomized to dual-antiplatelet therapy or aspirin plus placebo.
Sixty-three percent of subjects were on OMT at enrollment. Adherence to OMT was high: At 30 months post stenting, 62% of participants remained on OMT.
Between 12 and 30 months after percutaneous coronary intervention, continued dual-antiplatelet therapy in patients also on OMT was associated with a 2.1% incidence of acute MI, a 36% reduction in risk, compared with the 3.3% rate in subjects on placebo plus OMT. In patients not on OMT, the MI rate was 2.2% in those on dual-antiplatelet therapy versus 5.2% with placebo, for a 59% relative risk reduction in the active treatment arm.
The rate of the composite endpoint of major adverse cardiovascular and cerebrovascular events in patients on dual-antiplatelet therapy and OMT was 4.2%, compared with a 5.0% rate in patients on placebo and OMT.
Moderate or severe bleeding, as measured by the Global Use of Strategies to Open Occluded Arteries (GUSTO) criteria occurred in 2.2% of patients on dual-antiplatelet therapy and OMT, compared with 1.0% in those on placebo plus OMT. In patients not on OMT, the bleeding rate was 2.8% in the dual-antiplatelet therapy group and 2.2% with placebo, a nonsignificant difference.
Overall, patients on OMT had significantly lower rates of acute MI than those not on OMT (2.7%, compared with 3.7%), as well as fewer major adverse cardiovascular and cerebrovascular events (4.6% versus 5.7%), and less moderate or severe bleeding (1.6%, compared with 2.5% in patients not on OMT). However, rates of stroke, stent thrombosis, and death didn’t differ between patients on OMT and those who were not.
“While the associations between OMT use and lower rates of MI and MACCE [major adverse cardiovascular and cerebrovascular events] were expected, the association with lower rates of moderate or severe bleeding was not; we suspect the lower bleeding risk is mostly due to some residual confounding,” according to Dr. Resor.
He reported having no financial conflicts of interest regarding the new secondary analysis of DAPT, which was sponsored by the Harvard Clinical Research Institute.
Simultaneously with Dr. Resor’s presentation, the new DAPT study analysis was published online (Circulation. 2016 Aug 30. doi: 10.1161/CIRCULATIONAHA.116.024531.)
ROME – Continued dual-antiplatelet therapy beyond 12 months after coronary stenting is neither helped nor hindered by concomitant background optimal medical therapy, according to a secondary analysis of the landmark DAPT trial.
Numerous studies have shown that only 46%-66% of patients with stable ischemic heart disease are adherent to guideline-directed optimal medical therapy (OMT) with statins, beta-blockers, and ACE inhibitors or angiotensin receptor blockers. Yet until this new analysis from DAPT (the Dual Antiplatelet Therapy Study), the impact of OMT on the treatment effect of prolonged DAPT was unstudied, Charles D. Resor, MD, said at the annual congress of the European Society of Cardiology.
It was plausible that the treatment benefit of continued DAPT beyond 12 months after coronary stenting might be stunted by concomitant OMT, or alternatively that OMT and DAPT might exert synergistic effects in reducing the risk of ischemic events. As it turned out, neither of these possibilities turned out to be the case, according to Dr. Resor of Brigham and Women’s Hospital, Boston.
“I think OMT is underutilized, but while we would like physicians to emphasize OMT use in more cases, the decision to use DAPT beyond 12 months should be made irrespective of OMT use,” he said.
“That’s an important message for clinicians,” commented session cochair Keith A.A. Fox, MD, of the University of Edinburgh.
The landmark DAPT trial established that there is a significant benefit of continued dual-antiplatelet therapy beyond 12 months from coronary stenting in patients with stable ischemic heart disease (N Engl J Med. 2014 Dec 4;371:2155-66). Dr. Resor presented a secondary analysis of clinical outcomes in 11,643 study participants stratified by their OMT status as well as by whether they were randomized to dual-antiplatelet therapy or aspirin plus placebo.
Sixty-three percent of subjects were on OMT at enrollment. Adherence to OMT was high: At 30 months post stenting, 62% of participants remained on OMT.
Between 12 and 30 months after percutaneous coronary intervention, continued dual-antiplatelet therapy in patients also on OMT was associated with a 2.1% incidence of acute MI, a 36% reduction in risk, compared with the 3.3% rate in subjects on placebo plus OMT. In patients not on OMT, the MI rate was 2.2% in those on dual-antiplatelet therapy versus 5.2% with placebo, for a 59% relative risk reduction in the active treatment arm.
The rate of the composite endpoint of major adverse cardiovascular and cerebrovascular events in patients on dual-antiplatelet therapy and OMT was 4.2%, compared with a 5.0% rate in patients on placebo and OMT.
Moderate or severe bleeding, as measured by the Global Use of Strategies to Open Occluded Arteries (GUSTO) criteria occurred in 2.2% of patients on dual-antiplatelet therapy and OMT, compared with 1.0% in those on placebo plus OMT. In patients not on OMT, the bleeding rate was 2.8% in the dual-antiplatelet therapy group and 2.2% with placebo, a nonsignificant difference.
Overall, patients on OMT had significantly lower rates of acute MI than those not on OMT (2.7%, compared with 3.7%), as well as fewer major adverse cardiovascular and cerebrovascular events (4.6% versus 5.7%), and less moderate or severe bleeding (1.6%, compared with 2.5% in patients not on OMT). However, rates of stroke, stent thrombosis, and death didn’t differ between patients on OMT and those who were not.
“While the associations between OMT use and lower rates of MI and MACCE [major adverse cardiovascular and cerebrovascular events] were expected, the association with lower rates of moderate or severe bleeding was not; we suspect the lower bleeding risk is mostly due to some residual confounding,” according to Dr. Resor.
He reported having no financial conflicts of interest regarding the new secondary analysis of DAPT, which was sponsored by the Harvard Clinical Research Institute.
Simultaneously with Dr. Resor’s presentation, the new DAPT study analysis was published online (Circulation. 2016 Aug 30. doi: 10.1161/CIRCULATIONAHA.116.024531.)
AT THE ESC CONGRESS 2016
Key clinical point: The decision to use DAPT beyond 12 months should be made irrespective of optimal medical therapy use.
Major finding: Between 12 and 30 months after coronary stenting, continued dual-antiplatelet therapy was associated with a 2.1% rate of acute MI in patients on optimal medical therapy and a 2.2% rate in those not on optimal medical therapy.
Data source: A secondary analysis of 11,648 participants in the double-blind, randomized Dual Antiplatelet Therapy Study who were followed prospectively for the period of 12-30 months post PCI.
Disclosures: The study presenter reported having no financial conflicts regarding the new secondary analysis of DAPT, which was sponsored by the Harvard Clinical Research Institute.
Decision rule identifies unprovoked VTE patients who can halt anticoagulation
ROME – Half of all women who experience a first unprovoked venous thromboembolism (VTE) can safely be spared lifelong anticoagulation through application of the newly validated HERDOO2 decision rule, Marc A. Rodger, MD, reported at the annual congress of the European Society of Cardiology.
“We’ve validated that a simple, memorable decision rule on anticoagulation applied at the clinically relevant time point works. And it is the only clinical decision rule that has now been prospectively validated,” said Dr. Rodger, professor of medicine, chief and chair of the division of hematology, and head of the thrombosis program at the University of Ottawa.
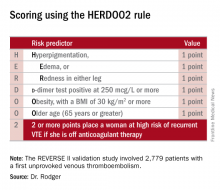
He presented the results of the validation study, known as the REVERSE II study, which included 2,779 patients with a first unprovoked VTE at 44 centers in seven countries. The full name of the decision rule is “Men Continue and HERDOO2,” a name that says it all: the rule posits that all men as well as those women with a HERDOO2 (Hyperpigmentation, Edema, Redness, d-dimer, Obesity, Older age, 2 or more points) score of at least 2 out of a possible 4 points need to stay on anticoagulation indefinitely because their risk of a recurrent VTE off-therapy clearly exceeds that of a bleeding event on-therapy. In contrast, women with a HERDOO2 score of 0 or 1 can safely stop anticoagulation after the standard 3-6 months of acute short-term therapy.
“Sorry, gentlemen, but we could find no low-risk group of men. They were all high risk,” he said. “But 50% of women with unprovoked vein blood clots can be spared the burdens, costs, and risks of lifelong blood thinners.”
Dr. Rodger and coinvestigators began work on developing a multivariate clinical decision rule in 2001. They examined 69 risk predictors, eventually winnowing down to a manageable four potent risk predictors identified by the acronym HERDOO2.
The derivation study was published 8 years ago (CMAJ. 2008;Aug 26;179[5]:417-26). It showed that women with a HERDOO2 score of 2 or more as well as all men had roughly a 14% rate of recurrent VTE in the first year after stopping anticoagulation, while women with a score of 0 or 1 had about a 1.6% risk. The International Society on Thrombosis and Haemostasis suggests that it’s safe to discontinue anticoagulants if the risk of recurrent thrombosis at 1 year off-therapy is less than 5%, given the significant risk of serious bleeding on-therapy and the fact that a serious bleed event is two to three times more likely than a VTE to be fatal.
Dr. Rodger and coinvestigators recognized that a clinical decision rule needs to be externally validated before it’s ready for prime-time use in clinical practice. Thus, they conducted the REVERSE II study, in which the decision rule was applied after the 2,799 participants had been on anticoagulation for 5-12 months. All had a first proximal deep vein thrombosis and/or a segmental or greater pulmonary embolism. Patients were still on anticoagulation at the time the rule was applied, which is why the cut point for a positive d-dimer test in HERDOO2 is 250 mcg/L, half of the threshold value for a positive test in patients not on anticoagulation.
They identified 631 women as low risk, with a HERDOO2 score of 0 or 1. They and their physicians were instructed to stop anticoagulation at that time. The 2,148 high-risk subjects – that is, all of the men and the high-risk women – were advised to remain on anticoagulation. The primary study endpoint was the rate of recurrent VTE in the 12 months following testing and patient guidance. The lost-to-follow-up rate was 2.2%.
The recurrent VTE rate was 3% in the 591 low-risk women who discontinued anticoagulants and zero in 31 others who elected to stay on medication. In the high-risk group identified by the HERDOO2 rule, the recurrent VTE rate at 12 months was 8.1% in the 323 who opted to discontinue anticoagulants and just 1.6% in 1,802 who continued on therapy as advised, a finding that underscores the effectiveness of selectively applied long-term anticoagulation therapy, he continued.
The recurrent VTE rate among the 291 women with a HERDOO2 score of 0 or 1 who were on exogenous estrogen was 1.4%, while in high-risk women taking estrogen the rate was more than doubled at 3.1%. But in women aged 50-64 identified by the HERDOO2 rule as being low risk, the actual recurrent VTE rate was 5.7%, a finding that raised a red flag for the investigators.
“There may be an evolution of the HERDOO2 decision rule to a lower age cut point. But that’s something that requires further study in postmenopausal women,” according to Dr. Rodger.
The investigators defined a first unprovoked VTE as one occurring in the absence during the previous 90 days of major surgery, a fracture or cast, more than 3 days of immobilization, or malignancy within the last 5 years.
Venous thromboembolism is the second most common cardiovascular disorder and the third most common cause of cardiovascular death. Unprovoked VTEs account for half of all VTEs. Their management has been a controversial subject. Both the American College of Chest Physicians and the European Society of Cardiology recommend continuing anticoagulation indefinitely in patients who aren’t at high bleeding risk.
“But this is a relatively weak 2B recommendation because of the tightly balanced competing risks of recurrent thrombosis off anticoagulation and major bleeding on anticoagulation,” Dr. Rodger said. He added that he considers REVERSE II to be practice changing, and predicted that once the results are published the guidelines will be revised.
Discussant Giancarlo Agnelli, MD, was a tough critic who gave fair warning.
“I am friends with many of the authors of this paper, and in this country we are usually gentle with enemies and nasty with friends,” declared Dr. Agnelli, professor of internal medicine and director of internal and cardiovascular medicine and the stroke unit at the University of Perugia, Italy.
He didn’t find the REVERSE II study or the HERDOO2 rule persuasive. On the plus side, he said, the HERDOO2 rule has now been validated, unlike the proposed DASH and Vienna rules. And it was tested in a diverse multinational patient population. But the fact that the HERDOO2 rule is only applicable in women is a major limitation. And REVERSE II was not a randomized trial, Dr. Agnelli noted.
Moreover, 1 year of follow-up seems insufficient, he continued. He cited a French multicenter trial in which patients with a first unprovoked VTE received 6 months of anticoagulants and were then randomized to another 18 months of anticoagulation or placebo. During that 18 months, the group on anticoagulants had a significantly lower rate of the composite endpoint comprised of recurrent VTE or major bleeding, but once that period was over they experienced catchup. By the time the study ended at 42 months, the two study arms didn’t differ significantly in the composite endpoint (JAMA. 2015 Jul 7;314[1]:31-40).
More broadly, Dr. Agnelli also questioned the need for an anticoagulation discontinuation rule in the contemporary era of new oral anticoagulants (NOACs). He was lead investigator in the AMPLIFY study, a major randomized trial of fixed-dose apixaban (Eliquis) versus conventional therapy with subcutaneous enoxaparin (Lovenox) bridging to warfarin in 5,395 patients with acute VTE. The NOAC was associated with a 69% reduction in the relative risk of bleeding and was noninferior to standard therapy in the risk of recurrent VTE (N Engl J Med. 2013 Aug 29;369[9]:799-808).
“Why should we think about withholding anticoagulation in some patients when we now have such a safe approach?” he asked.
Dr. Rodger reported receiving research grants from the French government as well as from Biomerieux, which funded the REVERSE II study. Dr. Agnelli reported having no financial conflicts.
ROME – Half of all women who experience a first unprovoked venous thromboembolism (VTE) can safely be spared lifelong anticoagulation through application of the newly validated HERDOO2 decision rule, Marc A. Rodger, MD, reported at the annual congress of the European Society of Cardiology.
“We’ve validated that a simple, memorable decision rule on anticoagulation applied at the clinically relevant time point works. And it is the only clinical decision rule that has now been prospectively validated,” said Dr. Rodger, professor of medicine, chief and chair of the division of hematology, and head of the thrombosis program at the University of Ottawa.
He presented the results of the validation study, known as the REVERSE II study, which included 2,779 patients with a first unprovoked VTE at 44 centers in seven countries. The full name of the decision rule is “Men Continue and HERDOO2,” a name that says it all: the rule posits that all men as well as those women with a HERDOO2 (Hyperpigmentation, Edema, Redness, d-dimer, Obesity, Older age, 2 or more points) score of at least 2 out of a possible 4 points need to stay on anticoagulation indefinitely because their risk of a recurrent VTE off-therapy clearly exceeds that of a bleeding event on-therapy. In contrast, women with a HERDOO2 score of 0 or 1 can safely stop anticoagulation after the standard 3-6 months of acute short-term therapy.
“Sorry, gentlemen, but we could find no low-risk group of men. They were all high risk,” he said. “But 50% of women with unprovoked vein blood clots can be spared the burdens, costs, and risks of lifelong blood thinners.”
Dr. Rodger and coinvestigators began work on developing a multivariate clinical decision rule in 2001. They examined 69 risk predictors, eventually winnowing down to a manageable four potent risk predictors identified by the acronym HERDOO2.
The derivation study was published 8 years ago (CMAJ. 2008;Aug 26;179[5]:417-26). It showed that women with a HERDOO2 score of 2 or more as well as all men had roughly a 14% rate of recurrent VTE in the first year after stopping anticoagulation, while women with a score of 0 or 1 had about a 1.6% risk. The International Society on Thrombosis and Haemostasis suggests that it’s safe to discontinue anticoagulants if the risk of recurrent thrombosis at 1 year off-therapy is less than 5%, given the significant risk of serious bleeding on-therapy and the fact that a serious bleed event is two to three times more likely than a VTE to be fatal.
Dr. Rodger and coinvestigators recognized that a clinical decision rule needs to be externally validated before it’s ready for prime-time use in clinical practice. Thus, they conducted the REVERSE II study, in which the decision rule was applied after the 2,799 participants had been on anticoagulation for 5-12 months. All had a first proximal deep vein thrombosis and/or a segmental or greater pulmonary embolism. Patients were still on anticoagulation at the time the rule was applied, which is why the cut point for a positive d-dimer test in HERDOO2 is 250 mcg/L, half of the threshold value for a positive test in patients not on anticoagulation.
They identified 631 women as low risk, with a HERDOO2 score of 0 or 1. They and their physicians were instructed to stop anticoagulation at that time. The 2,148 high-risk subjects – that is, all of the men and the high-risk women – were advised to remain on anticoagulation. The primary study endpoint was the rate of recurrent VTE in the 12 months following testing and patient guidance. The lost-to-follow-up rate was 2.2%.
The recurrent VTE rate was 3% in the 591 low-risk women who discontinued anticoagulants and zero in 31 others who elected to stay on medication. In the high-risk group identified by the HERDOO2 rule, the recurrent VTE rate at 12 months was 8.1% in the 323 who opted to discontinue anticoagulants and just 1.6% in 1,802 who continued on therapy as advised, a finding that underscores the effectiveness of selectively applied long-term anticoagulation therapy, he continued.
The recurrent VTE rate among the 291 women with a HERDOO2 score of 0 or 1 who were on exogenous estrogen was 1.4%, while in high-risk women taking estrogen the rate was more than doubled at 3.1%. But in women aged 50-64 identified by the HERDOO2 rule as being low risk, the actual recurrent VTE rate was 5.7%, a finding that raised a red flag for the investigators.
“There may be an evolution of the HERDOO2 decision rule to a lower age cut point. But that’s something that requires further study in postmenopausal women,” according to Dr. Rodger.
The investigators defined a first unprovoked VTE as one occurring in the absence during the previous 90 days of major surgery, a fracture or cast, more than 3 days of immobilization, or malignancy within the last 5 years.
Venous thromboembolism is the second most common cardiovascular disorder and the third most common cause of cardiovascular death. Unprovoked VTEs account for half of all VTEs. Their management has been a controversial subject. Both the American College of Chest Physicians and the European Society of Cardiology recommend continuing anticoagulation indefinitely in patients who aren’t at high bleeding risk.
“But this is a relatively weak 2B recommendation because of the tightly balanced competing risks of recurrent thrombosis off anticoagulation and major bleeding on anticoagulation,” Dr. Rodger said. He added that he considers REVERSE II to be practice changing, and predicted that once the results are published the guidelines will be revised.
Discussant Giancarlo Agnelli, MD, was a tough critic who gave fair warning.
“I am friends with many of the authors of this paper, and in this country we are usually gentle with enemies and nasty with friends,” declared Dr. Agnelli, professor of internal medicine and director of internal and cardiovascular medicine and the stroke unit at the University of Perugia, Italy.
He didn’t find the REVERSE II study or the HERDOO2 rule persuasive. On the plus side, he said, the HERDOO2 rule has now been validated, unlike the proposed DASH and Vienna rules. And it was tested in a diverse multinational patient population. But the fact that the HERDOO2 rule is only applicable in women is a major limitation. And REVERSE II was not a randomized trial, Dr. Agnelli noted.
Moreover, 1 year of follow-up seems insufficient, he continued. He cited a French multicenter trial in which patients with a first unprovoked VTE received 6 months of anticoagulants and were then randomized to another 18 months of anticoagulation or placebo. During that 18 months, the group on anticoagulants had a significantly lower rate of the composite endpoint comprised of recurrent VTE or major bleeding, but once that period was over they experienced catchup. By the time the study ended at 42 months, the two study arms didn’t differ significantly in the composite endpoint (JAMA. 2015 Jul 7;314[1]:31-40).
More broadly, Dr. Agnelli also questioned the need for an anticoagulation discontinuation rule in the contemporary era of new oral anticoagulants (NOACs). He was lead investigator in the AMPLIFY study, a major randomized trial of fixed-dose apixaban (Eliquis) versus conventional therapy with subcutaneous enoxaparin (Lovenox) bridging to warfarin in 5,395 patients with acute VTE. The NOAC was associated with a 69% reduction in the relative risk of bleeding and was noninferior to standard therapy in the risk of recurrent VTE (N Engl J Med. 2013 Aug 29;369[9]:799-808).
“Why should we think about withholding anticoagulation in some patients when we now have such a safe approach?” he asked.
Dr. Rodger reported receiving research grants from the French government as well as from Biomerieux, which funded the REVERSE II study. Dr. Agnelli reported having no financial conflicts.
ROME – Half of all women who experience a first unprovoked venous thromboembolism (VTE) can safely be spared lifelong anticoagulation through application of the newly validated HERDOO2 decision rule, Marc A. Rodger, MD, reported at the annual congress of the European Society of Cardiology.
“We’ve validated that a simple, memorable decision rule on anticoagulation applied at the clinically relevant time point works. And it is the only clinical decision rule that has now been prospectively validated,” said Dr. Rodger, professor of medicine, chief and chair of the division of hematology, and head of the thrombosis program at the University of Ottawa.
He presented the results of the validation study, known as the REVERSE II study, which included 2,779 patients with a first unprovoked VTE at 44 centers in seven countries. The full name of the decision rule is “Men Continue and HERDOO2,” a name that says it all: the rule posits that all men as well as those women with a HERDOO2 (Hyperpigmentation, Edema, Redness, d-dimer, Obesity, Older age, 2 or more points) score of at least 2 out of a possible 4 points need to stay on anticoagulation indefinitely because their risk of a recurrent VTE off-therapy clearly exceeds that of a bleeding event on-therapy. In contrast, women with a HERDOO2 score of 0 or 1 can safely stop anticoagulation after the standard 3-6 months of acute short-term therapy.
“Sorry, gentlemen, but we could find no low-risk group of men. They were all high risk,” he said. “But 50% of women with unprovoked vein blood clots can be spared the burdens, costs, and risks of lifelong blood thinners.”
Dr. Rodger and coinvestigators began work on developing a multivariate clinical decision rule in 2001. They examined 69 risk predictors, eventually winnowing down to a manageable four potent risk predictors identified by the acronym HERDOO2.
The derivation study was published 8 years ago (CMAJ. 2008;Aug 26;179[5]:417-26). It showed that women with a HERDOO2 score of 2 or more as well as all men had roughly a 14% rate of recurrent VTE in the first year after stopping anticoagulation, while women with a score of 0 or 1 had about a 1.6% risk. The International Society on Thrombosis and Haemostasis suggests that it’s safe to discontinue anticoagulants if the risk of recurrent thrombosis at 1 year off-therapy is less than 5%, given the significant risk of serious bleeding on-therapy and the fact that a serious bleed event is two to three times more likely than a VTE to be fatal.
Dr. Rodger and coinvestigators recognized that a clinical decision rule needs to be externally validated before it’s ready for prime-time use in clinical practice. Thus, they conducted the REVERSE II study, in which the decision rule was applied after the 2,799 participants had been on anticoagulation for 5-12 months. All had a first proximal deep vein thrombosis and/or a segmental or greater pulmonary embolism. Patients were still on anticoagulation at the time the rule was applied, which is why the cut point for a positive d-dimer test in HERDOO2 is 250 mcg/L, half of the threshold value for a positive test in patients not on anticoagulation.
They identified 631 women as low risk, with a HERDOO2 score of 0 or 1. They and their physicians were instructed to stop anticoagulation at that time. The 2,148 high-risk subjects – that is, all of the men and the high-risk women – were advised to remain on anticoagulation. The primary study endpoint was the rate of recurrent VTE in the 12 months following testing and patient guidance. The lost-to-follow-up rate was 2.2%.
The recurrent VTE rate was 3% in the 591 low-risk women who discontinued anticoagulants and zero in 31 others who elected to stay on medication. In the high-risk group identified by the HERDOO2 rule, the recurrent VTE rate at 12 months was 8.1% in the 323 who opted to discontinue anticoagulants and just 1.6% in 1,802 who continued on therapy as advised, a finding that underscores the effectiveness of selectively applied long-term anticoagulation therapy, he continued.
The recurrent VTE rate among the 291 women with a HERDOO2 score of 0 or 1 who were on exogenous estrogen was 1.4%, while in high-risk women taking estrogen the rate was more than doubled at 3.1%. But in women aged 50-64 identified by the HERDOO2 rule as being low risk, the actual recurrent VTE rate was 5.7%, a finding that raised a red flag for the investigators.
“There may be an evolution of the HERDOO2 decision rule to a lower age cut point. But that’s something that requires further study in postmenopausal women,” according to Dr. Rodger.
The investigators defined a first unprovoked VTE as one occurring in the absence during the previous 90 days of major surgery, a fracture or cast, more than 3 days of immobilization, or malignancy within the last 5 years.
Venous thromboembolism is the second most common cardiovascular disorder and the third most common cause of cardiovascular death. Unprovoked VTEs account for half of all VTEs. Their management has been a controversial subject. Both the American College of Chest Physicians and the European Society of Cardiology recommend continuing anticoagulation indefinitely in patients who aren’t at high bleeding risk.
“But this is a relatively weak 2B recommendation because of the tightly balanced competing risks of recurrent thrombosis off anticoagulation and major bleeding on anticoagulation,” Dr. Rodger said. He added that he considers REVERSE II to be practice changing, and predicted that once the results are published the guidelines will be revised.
Discussant Giancarlo Agnelli, MD, was a tough critic who gave fair warning.
“I am friends with many of the authors of this paper, and in this country we are usually gentle with enemies and nasty with friends,” declared Dr. Agnelli, professor of internal medicine and director of internal and cardiovascular medicine and the stroke unit at the University of Perugia, Italy.
He didn’t find the REVERSE II study or the HERDOO2 rule persuasive. On the plus side, he said, the HERDOO2 rule has now been validated, unlike the proposed DASH and Vienna rules. And it was tested in a diverse multinational patient population. But the fact that the HERDOO2 rule is only applicable in women is a major limitation. And REVERSE II was not a randomized trial, Dr. Agnelli noted.
Moreover, 1 year of follow-up seems insufficient, he continued. He cited a French multicenter trial in which patients with a first unprovoked VTE received 6 months of anticoagulants and were then randomized to another 18 months of anticoagulation or placebo. During that 18 months, the group on anticoagulants had a significantly lower rate of the composite endpoint comprised of recurrent VTE or major bleeding, but once that period was over they experienced catchup. By the time the study ended at 42 months, the two study arms didn’t differ significantly in the composite endpoint (JAMA. 2015 Jul 7;314[1]:31-40).
More broadly, Dr. Agnelli also questioned the need for an anticoagulation discontinuation rule in the contemporary era of new oral anticoagulants (NOACs). He was lead investigator in the AMPLIFY study, a major randomized trial of fixed-dose apixaban (Eliquis) versus conventional therapy with subcutaneous enoxaparin (Lovenox) bridging to warfarin in 5,395 patients with acute VTE. The NOAC was associated with a 69% reduction in the relative risk of bleeding and was noninferior to standard therapy in the risk of recurrent VTE (N Engl J Med. 2013 Aug 29;369[9]:799-808).
“Why should we think about withholding anticoagulation in some patients when we now have such a safe approach?” he asked.
Dr. Rodger reported receiving research grants from the French government as well as from Biomerieux, which funded the REVERSE II study. Dr. Agnelli reported having no financial conflicts.
AT THE ESC CONGRESS 2016
Key clinical point: Half of women who have a first unprovoked venous thromboembolism can safely be spared lifelong anticoagulation through application of the newly validated HERDOO2 decision rule.
Major finding: Women with a first unprovoked venous thromboembolism identified as being at low risk of recurrence on the basis of the HERDOO2 decision rule had a 3% recurrence rate in the year after stopping anticoagulation therapy, while those identified as high risk had an 8.1% recurrence rate if they discontinued anticoagulants.
Data source: This was a prospective, multinational, observational study involving 2,779 patients with a first unprovoked venous thromboembolism.
Disclosures: The presenter reported receiving research grants from the French government as well as from Biomerieux, which funded the REVERSE II study.
First-generation DES looking good at 10 years
ROME – There’s good news for the millions of patients living with a first-generation metallic drug-eluting stent for coronary revascularization implanted in years past: The devices perform reassuringly well a full decade after implantation, Lorenz Räber, MD, reported at the annual congress of the European Society of Cardiology.
That’s the key message of the SIRTAX VERY LATE study, the only randomized trial of first-generation drug-eluting stents (DES) that didn’t turn out the lights at a maximum of 5 years of follow-up. In fact, at the 10-year mark, SIRTAX VERY LATE shows that regardless of whether the first-generation DES was paclitaxel- or sirolimus-eluting, the risk of major adverse cardiac events due to device failure was substantially lower in the second half-decade than in the first 5 years after deployment, according to Dr. Räber of Bern (Switzerland) University Hospital.
More specifically, the cumulative risk of ischemia-driven target lesion revascularization was 14.6% at 5 years and 17.7% at 10 years, while the 5- and 10-year cumulative risks of definite stent thrombosis were 4.5% and 5.6%, respectively.
The annual risk of ischemia-driven target lesion revascularization dropped by 64%, from 1.8%/year during years 1-5 to 0.7% during years 6-10. Similarly, the annual risk of definite stent thrombosis fell from 0.67%/year to 0.23%/year after year 5, a 69% relative risk reduction. And importantly, these attenuations in risk occurred independent of age.
“The lower risk of late clinical events suggests stabilization of delayed arterial healing over time after first-generation DES implantation, with reduced chronic inflammation and neoatherosclerosis,” he said.
This is reassuring in light of the stormy history of the first-generation DES. Three years after the devices came on the U.S. market, the so-called ESC firestorm erupted. At the 2006 ESC congress, investigators presented meta-analyses suggesting the devices carried a possible late increased thrombotic risk beyond the then-recommended 3-6 months of prescribed dual-antiplatelet therapy. The use of these devices declined sharply in response, even though a Food and Drug Administration advisory panel charged with looking at the totality of evidence concluded that concerns about thrombosis didn’t outweigh the benefits of the first-generation DES over bare-metal stents.
The reductions in very late stent thrombosis and ischemia-driven target lesion revascularization beyond 5 years seen in the SIRTAX VERY LATE trial occurred despite the fact that only 15% of patients were on dual-antiplatelet therapy throughout the first 5 years and 11% were on dual-antiplatelet therapy afterwards, Dr. Räber noted.
“Our findings may have implications for secondary prevention after PCI with a first-generation DES, including the need for long-term antiplatelet therapy,” the cardiologist added.
The previously reported 5-year results of SIRTAX (the Sirolimus-Eluting Versus Paclitaxel-Eluting Stents for Coronary Revascularization trial) showed a steady increase over time in late lumen loss and an ongoing risk of very late stent thrombosis (Circulation. 2011 Jun 21;123(24):2819-28). Much the same was seen at the 5-year mark in the other major trials of first-generation DES, including RAVEL, SIRIUS, and TAXUS.
However, all those studies ended at 5 years, leaving unanswered the key question of what happens later. Cardiologists have wondered if the first-generation DES they put in their patients years ago were associated with a continued steady climb in the risk of device-related adverse events, or if the risk plateaued or even dropped off. The SIRTAX VERY LATE study was conducted in order to provide answers.
SIRTAX included 1,012 Swiss patients randomized to coronary revascularization using a first-generation sirolimus- or paclitaxel-eluting stent in 2003-2004. Roughly half had stable coronary artery disease and half presented with acute coronary syndromes. The 10-year follow-up conducted in the SIRTAX VERY LATE study captured 895 (88%) of the original 1,012 subjects.
The cumulative incidence of major cardiac adverse events – a composite of cardiac death, MI, and ischemia-driven target lesion revascularization – was 20.8% at 5 years and 33.8% at 10 years. The rate was similar between years 1-5 and 6-10.
The cumulative all-cause mortality rate was 10.4% at 5 years and 24.2% at 10 years. The rate was 2.0%/year during years 1-5 and accelerated significantly to 3.1%/year in years 6-10. However, this increase appears to be largely due to the background impact of advancing age rather than to any effect of having a first-generation DES. The 5- and 10-year all-cause mortality rates in the age- and sex-matched general Swiss population are similar to those seen in SIRTAX VERY LATE, at 9.6% and 22.1%, respectively, the cardiologist observed.
The cumulative incidence of MI in the study population was 7% at 5 years and 9.7% at 10 years. Between years 1-5 the rate was 0.9%/year, dropping to 0.6%/year during years 6-10.
One of the useful potential purposes for the new SIRTAX VERY LATE follow-up data beyond 5 years is that the results could serve as a benchmark in evaluating the long-term safety and efficacy of the much newer drug-eluting fully bioresorbable vascular scaffolds, since the potential benefits of these new devices may not appear until relatively late, after the devices themselves have disappeared. SIRTAX VERY LATE sets the bar for stent-related adverse events 5 years or more after device implantation at an annual risk of less than 0.3%/year for stent thrombosis and less than 1%/year for ischemia-driven target lesion revascularization.
Session co-chair Hector Bueno, MD, drew attention to the fact that no significant differences in clinical outcomes were seen at either 5 or 10 years between the sirolimus- and paclitaxel-eluting stent recipients. That’s noteworthy because more than a decade ago when the primary endpoint of SIRTAX was reported, much was made of the finding that the 9-month rate of major adverse cardiac events was significantly lower in the sirolimus-eluting stent group (N Engl J Med. 2005 Aug 18;353[7]:653-62). Over time, any outcome differences between the two devices were erased, observed Dr. Bueno of Complutense University of Madrid.
The SIRTAX VERY LATE study was funded by grants from Bern University Hospital. Dr. Räber reported having no relevant financial interests.
Simultaneously with his presentation in Rome at ESC 2016, the study results were published online (Eur Heart J. 2016 Aug 30. doi: 10.1093/eurheartj/ehw343).
ROME – There’s good news for the millions of patients living with a first-generation metallic drug-eluting stent for coronary revascularization implanted in years past: The devices perform reassuringly well a full decade after implantation, Lorenz Räber, MD, reported at the annual congress of the European Society of Cardiology.
That’s the key message of the SIRTAX VERY LATE study, the only randomized trial of first-generation drug-eluting stents (DES) that didn’t turn out the lights at a maximum of 5 years of follow-up. In fact, at the 10-year mark, SIRTAX VERY LATE shows that regardless of whether the first-generation DES was paclitaxel- or sirolimus-eluting, the risk of major adverse cardiac events due to device failure was substantially lower in the second half-decade than in the first 5 years after deployment, according to Dr. Räber of Bern (Switzerland) University Hospital.
More specifically, the cumulative risk of ischemia-driven target lesion revascularization was 14.6% at 5 years and 17.7% at 10 years, while the 5- and 10-year cumulative risks of definite stent thrombosis were 4.5% and 5.6%, respectively.
The annual risk of ischemia-driven target lesion revascularization dropped by 64%, from 1.8%/year during years 1-5 to 0.7% during years 6-10. Similarly, the annual risk of definite stent thrombosis fell from 0.67%/year to 0.23%/year after year 5, a 69% relative risk reduction. And importantly, these attenuations in risk occurred independent of age.
“The lower risk of late clinical events suggests stabilization of delayed arterial healing over time after first-generation DES implantation, with reduced chronic inflammation and neoatherosclerosis,” he said.
This is reassuring in light of the stormy history of the first-generation DES. Three years after the devices came on the U.S. market, the so-called ESC firestorm erupted. At the 2006 ESC congress, investigators presented meta-analyses suggesting the devices carried a possible late increased thrombotic risk beyond the then-recommended 3-6 months of prescribed dual-antiplatelet therapy. The use of these devices declined sharply in response, even though a Food and Drug Administration advisory panel charged with looking at the totality of evidence concluded that concerns about thrombosis didn’t outweigh the benefits of the first-generation DES over bare-metal stents.
The reductions in very late stent thrombosis and ischemia-driven target lesion revascularization beyond 5 years seen in the SIRTAX VERY LATE trial occurred despite the fact that only 15% of patients were on dual-antiplatelet therapy throughout the first 5 years and 11% were on dual-antiplatelet therapy afterwards, Dr. Räber noted.
“Our findings may have implications for secondary prevention after PCI with a first-generation DES, including the need for long-term antiplatelet therapy,” the cardiologist added.
The previously reported 5-year results of SIRTAX (the Sirolimus-Eluting Versus Paclitaxel-Eluting Stents for Coronary Revascularization trial) showed a steady increase over time in late lumen loss and an ongoing risk of very late stent thrombosis (Circulation. 2011 Jun 21;123(24):2819-28). Much the same was seen at the 5-year mark in the other major trials of first-generation DES, including RAVEL, SIRIUS, and TAXUS.
However, all those studies ended at 5 years, leaving unanswered the key question of what happens later. Cardiologists have wondered if the first-generation DES they put in their patients years ago were associated with a continued steady climb in the risk of device-related adverse events, or if the risk plateaued or even dropped off. The SIRTAX VERY LATE study was conducted in order to provide answers.
SIRTAX included 1,012 Swiss patients randomized to coronary revascularization using a first-generation sirolimus- or paclitaxel-eluting stent in 2003-2004. Roughly half had stable coronary artery disease and half presented with acute coronary syndromes. The 10-year follow-up conducted in the SIRTAX VERY LATE study captured 895 (88%) of the original 1,012 subjects.
The cumulative incidence of major cardiac adverse events – a composite of cardiac death, MI, and ischemia-driven target lesion revascularization – was 20.8% at 5 years and 33.8% at 10 years. The rate was similar between years 1-5 and 6-10.
The cumulative all-cause mortality rate was 10.4% at 5 years and 24.2% at 10 years. The rate was 2.0%/year during years 1-5 and accelerated significantly to 3.1%/year in years 6-10. However, this increase appears to be largely due to the background impact of advancing age rather than to any effect of having a first-generation DES. The 5- and 10-year all-cause mortality rates in the age- and sex-matched general Swiss population are similar to those seen in SIRTAX VERY LATE, at 9.6% and 22.1%, respectively, the cardiologist observed.
The cumulative incidence of MI in the study population was 7% at 5 years and 9.7% at 10 years. Between years 1-5 the rate was 0.9%/year, dropping to 0.6%/year during years 6-10.
One of the useful potential purposes for the new SIRTAX VERY LATE follow-up data beyond 5 years is that the results could serve as a benchmark in evaluating the long-term safety and efficacy of the much newer drug-eluting fully bioresorbable vascular scaffolds, since the potential benefits of these new devices may not appear until relatively late, after the devices themselves have disappeared. SIRTAX VERY LATE sets the bar for stent-related adverse events 5 years or more after device implantation at an annual risk of less than 0.3%/year for stent thrombosis and less than 1%/year for ischemia-driven target lesion revascularization.
Session co-chair Hector Bueno, MD, drew attention to the fact that no significant differences in clinical outcomes were seen at either 5 or 10 years between the sirolimus- and paclitaxel-eluting stent recipients. That’s noteworthy because more than a decade ago when the primary endpoint of SIRTAX was reported, much was made of the finding that the 9-month rate of major adverse cardiac events was significantly lower in the sirolimus-eluting stent group (N Engl J Med. 2005 Aug 18;353[7]:653-62). Over time, any outcome differences between the two devices were erased, observed Dr. Bueno of Complutense University of Madrid.
The SIRTAX VERY LATE study was funded by grants from Bern University Hospital. Dr. Räber reported having no relevant financial interests.
Simultaneously with his presentation in Rome at ESC 2016, the study results were published online (Eur Heart J. 2016 Aug 30. doi: 10.1093/eurheartj/ehw343).
ROME – There’s good news for the millions of patients living with a first-generation metallic drug-eluting stent for coronary revascularization implanted in years past: The devices perform reassuringly well a full decade after implantation, Lorenz Räber, MD, reported at the annual congress of the European Society of Cardiology.
That’s the key message of the SIRTAX VERY LATE study, the only randomized trial of first-generation drug-eluting stents (DES) that didn’t turn out the lights at a maximum of 5 years of follow-up. In fact, at the 10-year mark, SIRTAX VERY LATE shows that regardless of whether the first-generation DES was paclitaxel- or sirolimus-eluting, the risk of major adverse cardiac events due to device failure was substantially lower in the second half-decade than in the first 5 years after deployment, according to Dr. Räber of Bern (Switzerland) University Hospital.
More specifically, the cumulative risk of ischemia-driven target lesion revascularization was 14.6% at 5 years and 17.7% at 10 years, while the 5- and 10-year cumulative risks of definite stent thrombosis were 4.5% and 5.6%, respectively.
The annual risk of ischemia-driven target lesion revascularization dropped by 64%, from 1.8%/year during years 1-5 to 0.7% during years 6-10. Similarly, the annual risk of definite stent thrombosis fell from 0.67%/year to 0.23%/year after year 5, a 69% relative risk reduction. And importantly, these attenuations in risk occurred independent of age.
“The lower risk of late clinical events suggests stabilization of delayed arterial healing over time after first-generation DES implantation, with reduced chronic inflammation and neoatherosclerosis,” he said.
This is reassuring in light of the stormy history of the first-generation DES. Three years after the devices came on the U.S. market, the so-called ESC firestorm erupted. At the 2006 ESC congress, investigators presented meta-analyses suggesting the devices carried a possible late increased thrombotic risk beyond the then-recommended 3-6 months of prescribed dual-antiplatelet therapy. The use of these devices declined sharply in response, even though a Food and Drug Administration advisory panel charged with looking at the totality of evidence concluded that concerns about thrombosis didn’t outweigh the benefits of the first-generation DES over bare-metal stents.
The reductions in very late stent thrombosis and ischemia-driven target lesion revascularization beyond 5 years seen in the SIRTAX VERY LATE trial occurred despite the fact that only 15% of patients were on dual-antiplatelet therapy throughout the first 5 years and 11% were on dual-antiplatelet therapy afterwards, Dr. Räber noted.
“Our findings may have implications for secondary prevention after PCI with a first-generation DES, including the need for long-term antiplatelet therapy,” the cardiologist added.
The previously reported 5-year results of SIRTAX (the Sirolimus-Eluting Versus Paclitaxel-Eluting Stents for Coronary Revascularization trial) showed a steady increase over time in late lumen loss and an ongoing risk of very late stent thrombosis (Circulation. 2011 Jun 21;123(24):2819-28). Much the same was seen at the 5-year mark in the other major trials of first-generation DES, including RAVEL, SIRIUS, and TAXUS.
However, all those studies ended at 5 years, leaving unanswered the key question of what happens later. Cardiologists have wondered if the first-generation DES they put in their patients years ago were associated with a continued steady climb in the risk of device-related adverse events, or if the risk plateaued or even dropped off. The SIRTAX VERY LATE study was conducted in order to provide answers.
SIRTAX included 1,012 Swiss patients randomized to coronary revascularization using a first-generation sirolimus- or paclitaxel-eluting stent in 2003-2004. Roughly half had stable coronary artery disease and half presented with acute coronary syndromes. The 10-year follow-up conducted in the SIRTAX VERY LATE study captured 895 (88%) of the original 1,012 subjects.
The cumulative incidence of major cardiac adverse events – a composite of cardiac death, MI, and ischemia-driven target lesion revascularization – was 20.8% at 5 years and 33.8% at 10 years. The rate was similar between years 1-5 and 6-10.
The cumulative all-cause mortality rate was 10.4% at 5 years and 24.2% at 10 years. The rate was 2.0%/year during years 1-5 and accelerated significantly to 3.1%/year in years 6-10. However, this increase appears to be largely due to the background impact of advancing age rather than to any effect of having a first-generation DES. The 5- and 10-year all-cause mortality rates in the age- and sex-matched general Swiss population are similar to those seen in SIRTAX VERY LATE, at 9.6% and 22.1%, respectively, the cardiologist observed.
The cumulative incidence of MI in the study population was 7% at 5 years and 9.7% at 10 years. Between years 1-5 the rate was 0.9%/year, dropping to 0.6%/year during years 6-10.
One of the useful potential purposes for the new SIRTAX VERY LATE follow-up data beyond 5 years is that the results could serve as a benchmark in evaluating the long-term safety and efficacy of the much newer drug-eluting fully bioresorbable vascular scaffolds, since the potential benefits of these new devices may not appear until relatively late, after the devices themselves have disappeared. SIRTAX VERY LATE sets the bar for stent-related adverse events 5 years or more after device implantation at an annual risk of less than 0.3%/year for stent thrombosis and less than 1%/year for ischemia-driven target lesion revascularization.
Session co-chair Hector Bueno, MD, drew attention to the fact that no significant differences in clinical outcomes were seen at either 5 or 10 years between the sirolimus- and paclitaxel-eluting stent recipients. That’s noteworthy because more than a decade ago when the primary endpoint of SIRTAX was reported, much was made of the finding that the 9-month rate of major adverse cardiac events was significantly lower in the sirolimus-eluting stent group (N Engl J Med. 2005 Aug 18;353[7]:653-62). Over time, any outcome differences between the two devices were erased, observed Dr. Bueno of Complutense University of Madrid.
The SIRTAX VERY LATE study was funded by grants from Bern University Hospital. Dr. Räber reported having no relevant financial interests.
Simultaneously with his presentation in Rome at ESC 2016, the study results were published online (Eur Heart J. 2016 Aug 30. doi: 10.1093/eurheartj/ehw343).
AT THE ESC CONGRESS 2016
Key clinical point: The annual risks of ischemia-driven target lesion revascularization and stent thrombosis significantly decreased starting 5 years after implantation of a first-generation sirolimus- or paclitaxel-eluting stent.
Major finding: The annual risk of ischemia-driven target lesion revascularization was 1.8%/year between 1 and 5 years after implantation of a first-generation drug-eluting stent, but only 0.7%/year during years 6-10.
Data source: This was a unique extended 10-year follow-up of 88% of the original 1,012 participants in a randomized, assessor-blinded Swiss trial of coronary revascularization using a first-generation sirolimus- or paclitaxel-eluting stent.
Disclosures: The SIRTAX VERY LATE study was funded by grants from Bern University Hospital. The presenter reported having no relevant financial interests.
ANTARCTIC results chill enthusiasm for platelet monitoring
ROME – Measuring platelet function in order to tailor antiplatelet therapy in elderly patients undergoing percutaneous coronary intervention for acute coronary syndromes did not improve their clinical outcomes in the randomized ANTARCTIC trial, Gilles Montalescot, MD, reported at the annual congress of the European Society of Cardiology.
“We found absolutely no benefit for this strategy of adjustment of antiplatelet therapy based upon platelet function testing. The study was completely neutral on all types of endpoints, ischemic as well as bleeding,” said Dr. Montalescot, professor of cardiology at the University of Paris VI and director of the cardiac care unit at Pitié-Salpêtrière Hospital.
This was a disappointing result in what was the largest-ever randomized clinical trial involving PCI in elderly patients, he said. This was a high-risk population, not only by virtue of everyone being over age 75 years, but because they all presented with ACS. Indeed, one-third of ANTARCTIC participants underwent primary PCI for ST-segment elevation myocardial infarction.
ANTARCTIC (Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged Over 75 Years to Reduce the Composite of Bleeding, Stent Thrombosis, and Ischemic Complications) was carried out as a follow-up to the earlier ARCTIC randomized trial, also conducted by Dr. Montalescot and his coinvestigators. Like ANTARCTIC, ARCTIC, too, showed no clinical benefit for platelet function testing in order to adjust antiplatelet therapy (N Engl J Med. 2012;367:2100-9). At the time, ARCTIC’s critics argued that this individualized strategy didn’t achieve the expected improved outcomes because the trial was conducted in low-risk, stable patients undergoing elective scheduled PCI. In contrast, if there was ever a high-risk population in which platelet function testing and tailored antiplatelet therapy should work, it was in the very high-risk ANTARCTIC population, he said.
ANTARCTIC included 877 elderly patients undergoing urgent PCI for ACS who were placed on low-dose aspirin and randomized to standard antiplatelet therapy with prasugrel (Effient) at 5 mg/day, the European approved dose for long-term maintenance therapy in elderly patients, or to tailored antiplatelet therapy.
Patients in the tailored therapy arm received prasugrel at 5 mg/day for the first 14 days, then underwent platelet function testing with the VerifyNow P2Y12 system. If they demonstrated high on-drug platelet activity, defined as at least 208 P2Y12 reaction units (PRU), their prasugrel was bumped up to 10 mg/day. If their PRU measurement was in what is considered the optimal range for quelling ischemia without promoting bleeding – that is, less than 208 but more than 85 PRU – they remained on prasugrel at 5 mg/day. And if they scored less than 85 PRU, exposing them to excess bleeding risk due to high suppression of platelets, they were switched to clopidogrel (Plavix) at 75 mg/day, a less potent antiplatelet regimen.
Two weeks after their first platelet function measurement, participants in the tailored therapy arm returned for a second round of platelet activity testing, with their antiplatelet regimen once again being adjusted on the basis of the results.
The primary study endpoint was net clinical benefit over a 12-month follow-up period. This was defined as the composite of cardiovascular death, MI, stroke, urgent revascularization, stent thrombosis, and Bleeding Academic Research Consortium (BARC) types 2, 3, or 5. This composite endpoint occurred in 27.6% of the platelet monitoring group and a near-identical 27.8% of conventionally managed patients.
Of note, 42% of patients in the actively monitored group were within the target platelet inhibition range when tested 14 days into the study. At study’s end, 55% of patients remained on prasugrel at 5 mg/day, 39% were on clopidogrel at 75 mg/day, and less than 4% were on prasugrel at 10 mg/day. Thus, most patients who underwent a dose adjustment on the basis of their VerifyNow results were downgraded to a less-potent antiplatelet regimen. Very few required enhanced platelet suppression in the form of 10 mg/day of prasugrel.
“Platelet function monitoring is difficult to use. Patients have to come back twice to be monitored. It’s costly. It’s time consuming. And platelet function monitoring clearly does not help,” the cardiologist said.
The ANTARCTIC results will likely lead to a revision of the American and European guidelines, which currently give a class IIb/level of evidence C recommendation for platelet function testing in high-risk situations.
“There is a huge literature showing that platelet reactivity affects clinical outcomes,” Dr. Montalescot continued. “One hypothesis now is that platelet reactivity may be only a marker of risk; you can modify it, but that has no impact on patient outcomes. We may be in the same situation here as with HDL cholesterol, for example.”
Discussant Steen Dalby Kristensen, MD, noted that ANTARCTIC is just the latest in a slew of negative randomized clinical trials of individualized antiplatelet therapy for coronary artery disease. In addition to ARCTIC, others include GRAVITAS, TRIGGER PCI, and ASCET. One study, the German/Austrian TROPICAL ACS trial, remains ongoing.
“It really is an intriguing concept that many of us have been fascinated by for years: to identify the sweet spot where, by measuring platelet aggregation and maybe changing the therapy, we can find just the right balance between bleeding and ischemia. The ANTARCTIC results are quite disappointing for platelet-monitoring enthusiasts. Is the whole concept wrong?” said Dr. Kristensen, professor of cardiology and head of the cardiovascular research center at Aarhus (Denmark) University Hospital.
“I think even more disappointing for me than the lack of impact on ischemic events was the bleeding. I would have anticipated that maybe bleeding could be avoided by adjusting the dose, but this was not the case,” he added.
But Stephan Gielen, MD, saw a silver lining in the negative results for ANTARCTIC.
“From my perspective as a clinical interventionalist, I’m happy that you ended up in the way you did. Putting things positively, this study confirms the safety of dual-platelet inhibition with prasugrel at the reduced dose of 5 mg in an elderly population. There is no need to go to the trouble of monitoring platelet function even in this elderly population, which I think for clinical practice is a good message,” said Dr. Gielen of Detmold (Germany) Hospital, who cochaired a press conference where Dr. Montalescot presented the ANTARCTIC results.
Simultaneously with Dr. Montalescot’s presentation at ESC 2016 in Rome, the ANTARCTIC results were published online (Lancet. 2016 Aug 26. doi: 10.1016/S0140-6736(16)31323-X).
ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
ROME – Measuring platelet function in order to tailor antiplatelet therapy in elderly patients undergoing percutaneous coronary intervention for acute coronary syndromes did not improve their clinical outcomes in the randomized ANTARCTIC trial, Gilles Montalescot, MD, reported at the annual congress of the European Society of Cardiology.
“We found absolutely no benefit for this strategy of adjustment of antiplatelet therapy based upon platelet function testing. The study was completely neutral on all types of endpoints, ischemic as well as bleeding,” said Dr. Montalescot, professor of cardiology at the University of Paris VI and director of the cardiac care unit at Pitié-Salpêtrière Hospital.
This was a disappointing result in what was the largest-ever randomized clinical trial involving PCI in elderly patients, he said. This was a high-risk population, not only by virtue of everyone being over age 75 years, but because they all presented with ACS. Indeed, one-third of ANTARCTIC participants underwent primary PCI for ST-segment elevation myocardial infarction.
ANTARCTIC (Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged Over 75 Years to Reduce the Composite of Bleeding, Stent Thrombosis, and Ischemic Complications) was carried out as a follow-up to the earlier ARCTIC randomized trial, also conducted by Dr. Montalescot and his coinvestigators. Like ANTARCTIC, ARCTIC, too, showed no clinical benefit for platelet function testing in order to adjust antiplatelet therapy (N Engl J Med. 2012;367:2100-9). At the time, ARCTIC’s critics argued that this individualized strategy didn’t achieve the expected improved outcomes because the trial was conducted in low-risk, stable patients undergoing elective scheduled PCI. In contrast, if there was ever a high-risk population in which platelet function testing and tailored antiplatelet therapy should work, it was in the very high-risk ANTARCTIC population, he said.
ANTARCTIC included 877 elderly patients undergoing urgent PCI for ACS who were placed on low-dose aspirin and randomized to standard antiplatelet therapy with prasugrel (Effient) at 5 mg/day, the European approved dose for long-term maintenance therapy in elderly patients, or to tailored antiplatelet therapy.
Patients in the tailored therapy arm received prasugrel at 5 mg/day for the first 14 days, then underwent platelet function testing with the VerifyNow P2Y12 system. If they demonstrated high on-drug platelet activity, defined as at least 208 P2Y12 reaction units (PRU), their prasugrel was bumped up to 10 mg/day. If their PRU measurement was in what is considered the optimal range for quelling ischemia without promoting bleeding – that is, less than 208 but more than 85 PRU – they remained on prasugrel at 5 mg/day. And if they scored less than 85 PRU, exposing them to excess bleeding risk due to high suppression of platelets, they were switched to clopidogrel (Plavix) at 75 mg/day, a less potent antiplatelet regimen.
Two weeks after their first platelet function measurement, participants in the tailored therapy arm returned for a second round of platelet activity testing, with their antiplatelet regimen once again being adjusted on the basis of the results.
The primary study endpoint was net clinical benefit over a 12-month follow-up period. This was defined as the composite of cardiovascular death, MI, stroke, urgent revascularization, stent thrombosis, and Bleeding Academic Research Consortium (BARC) types 2, 3, or 5. This composite endpoint occurred in 27.6% of the platelet monitoring group and a near-identical 27.8% of conventionally managed patients.
Of note, 42% of patients in the actively monitored group were within the target platelet inhibition range when tested 14 days into the study. At study’s end, 55% of patients remained on prasugrel at 5 mg/day, 39% were on clopidogrel at 75 mg/day, and less than 4% were on prasugrel at 10 mg/day. Thus, most patients who underwent a dose adjustment on the basis of their VerifyNow results were downgraded to a less-potent antiplatelet regimen. Very few required enhanced platelet suppression in the form of 10 mg/day of prasugrel.
“Platelet function monitoring is difficult to use. Patients have to come back twice to be monitored. It’s costly. It’s time consuming. And platelet function monitoring clearly does not help,” the cardiologist said.
The ANTARCTIC results will likely lead to a revision of the American and European guidelines, which currently give a class IIb/level of evidence C recommendation for platelet function testing in high-risk situations.
“There is a huge literature showing that platelet reactivity affects clinical outcomes,” Dr. Montalescot continued. “One hypothesis now is that platelet reactivity may be only a marker of risk; you can modify it, but that has no impact on patient outcomes. We may be in the same situation here as with HDL cholesterol, for example.”
Discussant Steen Dalby Kristensen, MD, noted that ANTARCTIC is just the latest in a slew of negative randomized clinical trials of individualized antiplatelet therapy for coronary artery disease. In addition to ARCTIC, others include GRAVITAS, TRIGGER PCI, and ASCET. One study, the German/Austrian TROPICAL ACS trial, remains ongoing.
“It really is an intriguing concept that many of us have been fascinated by for years: to identify the sweet spot where, by measuring platelet aggregation and maybe changing the therapy, we can find just the right balance between bleeding and ischemia. The ANTARCTIC results are quite disappointing for platelet-monitoring enthusiasts. Is the whole concept wrong?” said Dr. Kristensen, professor of cardiology and head of the cardiovascular research center at Aarhus (Denmark) University Hospital.
“I think even more disappointing for me than the lack of impact on ischemic events was the bleeding. I would have anticipated that maybe bleeding could be avoided by adjusting the dose, but this was not the case,” he added.
But Stephan Gielen, MD, saw a silver lining in the negative results for ANTARCTIC.
“From my perspective as a clinical interventionalist, I’m happy that you ended up in the way you did. Putting things positively, this study confirms the safety of dual-platelet inhibition with prasugrel at the reduced dose of 5 mg in an elderly population. There is no need to go to the trouble of monitoring platelet function even in this elderly population, which I think for clinical practice is a good message,” said Dr. Gielen of Detmold (Germany) Hospital, who cochaired a press conference where Dr. Montalescot presented the ANTARCTIC results.
Simultaneously with Dr. Montalescot’s presentation at ESC 2016 in Rome, the ANTARCTIC results were published online (Lancet. 2016 Aug 26. doi: 10.1016/S0140-6736(16)31323-X).
ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
ROME – Measuring platelet function in order to tailor antiplatelet therapy in elderly patients undergoing percutaneous coronary intervention for acute coronary syndromes did not improve their clinical outcomes in the randomized ANTARCTIC trial, Gilles Montalescot, MD, reported at the annual congress of the European Society of Cardiology.
“We found absolutely no benefit for this strategy of adjustment of antiplatelet therapy based upon platelet function testing. The study was completely neutral on all types of endpoints, ischemic as well as bleeding,” said Dr. Montalescot, professor of cardiology at the University of Paris VI and director of the cardiac care unit at Pitié-Salpêtrière Hospital.
This was a disappointing result in what was the largest-ever randomized clinical trial involving PCI in elderly patients, he said. This was a high-risk population, not only by virtue of everyone being over age 75 years, but because they all presented with ACS. Indeed, one-third of ANTARCTIC participants underwent primary PCI for ST-segment elevation myocardial infarction.
ANTARCTIC (Assessment of a Normal Versus Tailored Dose of Prasugrel After Stenting in Patients Aged Over 75 Years to Reduce the Composite of Bleeding, Stent Thrombosis, and Ischemic Complications) was carried out as a follow-up to the earlier ARCTIC randomized trial, also conducted by Dr. Montalescot and his coinvestigators. Like ANTARCTIC, ARCTIC, too, showed no clinical benefit for platelet function testing in order to adjust antiplatelet therapy (N Engl J Med. 2012;367:2100-9). At the time, ARCTIC’s critics argued that this individualized strategy didn’t achieve the expected improved outcomes because the trial was conducted in low-risk, stable patients undergoing elective scheduled PCI. In contrast, if there was ever a high-risk population in which platelet function testing and tailored antiplatelet therapy should work, it was in the very high-risk ANTARCTIC population, he said.
ANTARCTIC included 877 elderly patients undergoing urgent PCI for ACS who were placed on low-dose aspirin and randomized to standard antiplatelet therapy with prasugrel (Effient) at 5 mg/day, the European approved dose for long-term maintenance therapy in elderly patients, or to tailored antiplatelet therapy.
Patients in the tailored therapy arm received prasugrel at 5 mg/day for the first 14 days, then underwent platelet function testing with the VerifyNow P2Y12 system. If they demonstrated high on-drug platelet activity, defined as at least 208 P2Y12 reaction units (PRU), their prasugrel was bumped up to 10 mg/day. If their PRU measurement was in what is considered the optimal range for quelling ischemia without promoting bleeding – that is, less than 208 but more than 85 PRU – they remained on prasugrel at 5 mg/day. And if they scored less than 85 PRU, exposing them to excess bleeding risk due to high suppression of platelets, they were switched to clopidogrel (Plavix) at 75 mg/day, a less potent antiplatelet regimen.
Two weeks after their first platelet function measurement, participants in the tailored therapy arm returned for a second round of platelet activity testing, with their antiplatelet regimen once again being adjusted on the basis of the results.
The primary study endpoint was net clinical benefit over a 12-month follow-up period. This was defined as the composite of cardiovascular death, MI, stroke, urgent revascularization, stent thrombosis, and Bleeding Academic Research Consortium (BARC) types 2, 3, or 5. This composite endpoint occurred in 27.6% of the platelet monitoring group and a near-identical 27.8% of conventionally managed patients.
Of note, 42% of patients in the actively monitored group were within the target platelet inhibition range when tested 14 days into the study. At study’s end, 55% of patients remained on prasugrel at 5 mg/day, 39% were on clopidogrel at 75 mg/day, and less than 4% were on prasugrel at 10 mg/day. Thus, most patients who underwent a dose adjustment on the basis of their VerifyNow results were downgraded to a less-potent antiplatelet regimen. Very few required enhanced platelet suppression in the form of 10 mg/day of prasugrel.
“Platelet function monitoring is difficult to use. Patients have to come back twice to be monitored. It’s costly. It’s time consuming. And platelet function monitoring clearly does not help,” the cardiologist said.
The ANTARCTIC results will likely lead to a revision of the American and European guidelines, which currently give a class IIb/level of evidence C recommendation for platelet function testing in high-risk situations.
“There is a huge literature showing that platelet reactivity affects clinical outcomes,” Dr. Montalescot continued. “One hypothesis now is that platelet reactivity may be only a marker of risk; you can modify it, but that has no impact on patient outcomes. We may be in the same situation here as with HDL cholesterol, for example.”
Discussant Steen Dalby Kristensen, MD, noted that ANTARCTIC is just the latest in a slew of negative randomized clinical trials of individualized antiplatelet therapy for coronary artery disease. In addition to ARCTIC, others include GRAVITAS, TRIGGER PCI, and ASCET. One study, the German/Austrian TROPICAL ACS trial, remains ongoing.
“It really is an intriguing concept that many of us have been fascinated by for years: to identify the sweet spot where, by measuring platelet aggregation and maybe changing the therapy, we can find just the right balance between bleeding and ischemia. The ANTARCTIC results are quite disappointing for platelet-monitoring enthusiasts. Is the whole concept wrong?” said Dr. Kristensen, professor of cardiology and head of the cardiovascular research center at Aarhus (Denmark) University Hospital.
“I think even more disappointing for me than the lack of impact on ischemic events was the bleeding. I would have anticipated that maybe bleeding could be avoided by adjusting the dose, but this was not the case,” he added.
But Stephan Gielen, MD, saw a silver lining in the negative results for ANTARCTIC.
“From my perspective as a clinical interventionalist, I’m happy that you ended up in the way you did. Putting things positively, this study confirms the safety of dual-platelet inhibition with prasugrel at the reduced dose of 5 mg in an elderly population. There is no need to go to the trouble of monitoring platelet function even in this elderly population, which I think for clinical practice is a good message,” said Dr. Gielen of Detmold (Germany) Hospital, who cochaired a press conference where Dr. Montalescot presented the ANTARCTIC results.
Simultaneously with Dr. Montalescot’s presentation at ESC 2016 in Rome, the ANTARCTIC results were published online (Lancet. 2016 Aug 26. doi: 10.1016/S0140-6736(16)31323-X).
ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
AT THE ESC CONGRESS 2016
Key clinical point: Researchers have just about given up on the notion that monitoring platelet function in order to individualize antiplatelet therapy in patients undergoing PCI provides any clinical benefit.
Major finding: Individualized antiplatelet therapy based upon serial measurements of platelet function did not result in improved outcomes in elderly patients undergoing PCI for acute coronary syndrome (27.6% in the platelet monitoring group and 27.8% in conventionally managed patients).
Data source: ANTARCTIC was an open-label, blinded-endpoint randomized trial of tailored versus standard antiplatelet therapy in 877 elderly patients undergoing PCI for ACS.
Disclosures: ANTARCTIC was funded by Eli Lilly, Daiichi Sankyo, Stentys, Accriva Diagnostics, Medtronic, and the French Foundation for Heart Research. The presenter reported receiving research grants from and/or serving as a consultant to those organizations and numerous others.
Ticagrelor slashes first stroke risk after MI
ROME – Adding ticagrelor at 60 mg twice daily in patients on low-dose aspirin due to a prior MI reduced their risk of a first stroke by 25% in a secondary analysis of the landmark PEGASUS-TIMI 54 trial, Marc P. Bonaca, MD, reported at the annual congress of the European Society of Cardiology.
PEGASUS-TIMI 54 was a randomized, double-blind, placebo-controlled clinical trial conducted in more than 21,000 stable patients on low-dose aspirin with a history of an acute MI 1-3 years earlier. The significant reduction in secondary cardiovascular events seen in this study during a median 33 months of follow-up (N Engl J Med. 2015 May 7;372[19]:1791-800) led to approval of ticagrelor (Brilinta) at 60 mg twice daily for long-term secondary prevention.
But while PEGASUS-TIMI 54 was a secondary prevention study in terms of cardiovascular events, it was actually a primary prevention study in terms of stroke, since patients with a history of stroke weren’t eligible for enrollment. And in this trial, recipients of ticagrelor at 50 mg twice daily experienced a 25% reduction in the risk of stroke relative to placebo, from 1.94% at 3 years to 1.47%. This benefit was driven by fewer ischemic strokes, with no increase in hemorrhagic strokes seen with ticagrelor. And therein lies a clinical take home point: “When evaluating the overall benefits and risks of long-term ticagrelor in patients with prior MI, stroke reduction should also be considered,” according to Dr. Bonaca of Brigham and Women’s Hospital, Boston.
All strokes were adjudicated and subclassified by a blinded central committee. A total of 213 stroke events occurred during follow-up: 81% ischemic, 7% hemorrhagic, 4% ischemic with hemorrhagic conversion, and 8% unknown; 18% of the strokes were fatal. Another 15% resulted in moderate or severe disability at 30 days. All PEGASUS-TIMI 54 participants were on aspirin and more than 90% were on statin therapy.
The strokes that occurred in patients on ticagrelor were generally less severe than in controls. The risk of having a modified Rankin score of 3-6, which encompasses outcomes ranging from moderate disability to death, was reduced by 43% in stroke patients on ticagrelor relative to those on placebo, the cardiologist continued.
To ensure that the stroke benefit with ticagrelor seen in PEGASUS-TIMI 54 wasn’t a fluke, Dr. Bonaca and his coinvestigators performed a meta-analysis of four placebo-controlled randomized trials of more intensive versus less intensive antiplatelet therapy in nearly 45,000 participants with coronary disease in the CHARISMA, DAPT, PEGASUS-TIMI 54, and TRA 2*P-TIMI 50 trials. A total of 532 strokes occurred in this enlarged analysis. More intensive antiplatelet therapy – typically adding a second drug to low-dose aspirin – resulted in a 34% reduction in ischemic stroke, compared with low-dose aspirin and placebo.
Excluding from the meta-analysis the large subgroup of patients in TRA 2*P-TIMI 50 who were on triple-drug antiplatelet therapy, investigators were left with 32,348 participants in the four trials who were randomized to dual-antiplatelet therapy or monotherapy with aspirin. In this population, there was no increase in the risk of hemorrhagic stroke associated with more intensive antiplatelet therapy, according to Dr. Bonaca.
Session co-chair Keith A.A. Fox, MD, of the University of Edinburgh, noted that various studies have shown monotherapy with aspirin or another antiplatelet agent reduces stroke risk by about 15%, and now PEGASUS-TIMI 54 shows that ticagrelor plus aspirin decreases stroke risk by 25%. He posed a direct question: “How much is too much?”
“More and more antiplatelet therapy begets more bleeding, so I think that more than two agents may be approaching too much, although it really depends on what agents you’re using and in what dosages,” Dr. Bonaca replied.
He reported serving as a consultant to AstraZeneca, Merck, and Bayer.
Simultaneous with Dr. Bonaca’s presentation at ESC 2016 in Rome, the new report from PEGASUS-TIMI 54 including the four-trial meta-analysis was published online (Circulation. 2016 Aug 30. doi: circulationaha.116.024637).
ROME – Adding ticagrelor at 60 mg twice daily in patients on low-dose aspirin due to a prior MI reduced their risk of a first stroke by 25% in a secondary analysis of the landmark PEGASUS-TIMI 54 trial, Marc P. Bonaca, MD, reported at the annual congress of the European Society of Cardiology.
PEGASUS-TIMI 54 was a randomized, double-blind, placebo-controlled clinical trial conducted in more than 21,000 stable patients on low-dose aspirin with a history of an acute MI 1-3 years earlier. The significant reduction in secondary cardiovascular events seen in this study during a median 33 months of follow-up (N Engl J Med. 2015 May 7;372[19]:1791-800) led to approval of ticagrelor (Brilinta) at 60 mg twice daily for long-term secondary prevention.
But while PEGASUS-TIMI 54 was a secondary prevention study in terms of cardiovascular events, it was actually a primary prevention study in terms of stroke, since patients with a history of stroke weren’t eligible for enrollment. And in this trial, recipients of ticagrelor at 50 mg twice daily experienced a 25% reduction in the risk of stroke relative to placebo, from 1.94% at 3 years to 1.47%. This benefit was driven by fewer ischemic strokes, with no increase in hemorrhagic strokes seen with ticagrelor. And therein lies a clinical take home point: “When evaluating the overall benefits and risks of long-term ticagrelor in patients with prior MI, stroke reduction should also be considered,” according to Dr. Bonaca of Brigham and Women’s Hospital, Boston.
All strokes were adjudicated and subclassified by a blinded central committee. A total of 213 stroke events occurred during follow-up: 81% ischemic, 7% hemorrhagic, 4% ischemic with hemorrhagic conversion, and 8% unknown; 18% of the strokes were fatal. Another 15% resulted in moderate or severe disability at 30 days. All PEGASUS-TIMI 54 participants were on aspirin and more than 90% were on statin therapy.
The strokes that occurred in patients on ticagrelor were generally less severe than in controls. The risk of having a modified Rankin score of 3-6, which encompasses outcomes ranging from moderate disability to death, was reduced by 43% in stroke patients on ticagrelor relative to those on placebo, the cardiologist continued.
To ensure that the stroke benefit with ticagrelor seen in PEGASUS-TIMI 54 wasn’t a fluke, Dr. Bonaca and his coinvestigators performed a meta-analysis of four placebo-controlled randomized trials of more intensive versus less intensive antiplatelet therapy in nearly 45,000 participants with coronary disease in the CHARISMA, DAPT, PEGASUS-TIMI 54, and TRA 2*P-TIMI 50 trials. A total of 532 strokes occurred in this enlarged analysis. More intensive antiplatelet therapy – typically adding a second drug to low-dose aspirin – resulted in a 34% reduction in ischemic stroke, compared with low-dose aspirin and placebo.
Excluding from the meta-analysis the large subgroup of patients in TRA 2*P-TIMI 50 who were on triple-drug antiplatelet therapy, investigators were left with 32,348 participants in the four trials who were randomized to dual-antiplatelet therapy or monotherapy with aspirin. In this population, there was no increase in the risk of hemorrhagic stroke associated with more intensive antiplatelet therapy, according to Dr. Bonaca.
Session co-chair Keith A.A. Fox, MD, of the University of Edinburgh, noted that various studies have shown monotherapy with aspirin or another antiplatelet agent reduces stroke risk by about 15%, and now PEGASUS-TIMI 54 shows that ticagrelor plus aspirin decreases stroke risk by 25%. He posed a direct question: “How much is too much?”
“More and more antiplatelet therapy begets more bleeding, so I think that more than two agents may be approaching too much, although it really depends on what agents you’re using and in what dosages,” Dr. Bonaca replied.
He reported serving as a consultant to AstraZeneca, Merck, and Bayer.
Simultaneous with Dr. Bonaca’s presentation at ESC 2016 in Rome, the new report from PEGASUS-TIMI 54 including the four-trial meta-analysis was published online (Circulation. 2016 Aug 30. doi: circulationaha.116.024637).
ROME – Adding ticagrelor at 60 mg twice daily in patients on low-dose aspirin due to a prior MI reduced their risk of a first stroke by 25% in a secondary analysis of the landmark PEGASUS-TIMI 54 trial, Marc P. Bonaca, MD, reported at the annual congress of the European Society of Cardiology.
PEGASUS-TIMI 54 was a randomized, double-blind, placebo-controlled clinical trial conducted in more than 21,000 stable patients on low-dose aspirin with a history of an acute MI 1-3 years earlier. The significant reduction in secondary cardiovascular events seen in this study during a median 33 months of follow-up (N Engl J Med. 2015 May 7;372[19]:1791-800) led to approval of ticagrelor (Brilinta) at 60 mg twice daily for long-term secondary prevention.
But while PEGASUS-TIMI 54 was a secondary prevention study in terms of cardiovascular events, it was actually a primary prevention study in terms of stroke, since patients with a history of stroke weren’t eligible for enrollment. And in this trial, recipients of ticagrelor at 50 mg twice daily experienced a 25% reduction in the risk of stroke relative to placebo, from 1.94% at 3 years to 1.47%. This benefit was driven by fewer ischemic strokes, with no increase in hemorrhagic strokes seen with ticagrelor. And therein lies a clinical take home point: “When evaluating the overall benefits and risks of long-term ticagrelor in patients with prior MI, stroke reduction should also be considered,” according to Dr. Bonaca of Brigham and Women’s Hospital, Boston.
All strokes were adjudicated and subclassified by a blinded central committee. A total of 213 stroke events occurred during follow-up: 81% ischemic, 7% hemorrhagic, 4% ischemic with hemorrhagic conversion, and 8% unknown; 18% of the strokes were fatal. Another 15% resulted in moderate or severe disability at 30 days. All PEGASUS-TIMI 54 participants were on aspirin and more than 90% were on statin therapy.
The strokes that occurred in patients on ticagrelor were generally less severe than in controls. The risk of having a modified Rankin score of 3-6, which encompasses outcomes ranging from moderate disability to death, was reduced by 43% in stroke patients on ticagrelor relative to those on placebo, the cardiologist continued.
To ensure that the stroke benefit with ticagrelor seen in PEGASUS-TIMI 54 wasn’t a fluke, Dr. Bonaca and his coinvestigators performed a meta-analysis of four placebo-controlled randomized trials of more intensive versus less intensive antiplatelet therapy in nearly 45,000 participants with coronary disease in the CHARISMA, DAPT, PEGASUS-TIMI 54, and TRA 2*P-TIMI 50 trials. A total of 532 strokes occurred in this enlarged analysis. More intensive antiplatelet therapy – typically adding a second drug to low-dose aspirin – resulted in a 34% reduction in ischemic stroke, compared with low-dose aspirin and placebo.
Excluding from the meta-analysis the large subgroup of patients in TRA 2*P-TIMI 50 who were on triple-drug antiplatelet therapy, investigators were left with 32,348 participants in the four trials who were randomized to dual-antiplatelet therapy or monotherapy with aspirin. In this population, there was no increase in the risk of hemorrhagic stroke associated with more intensive antiplatelet therapy, according to Dr. Bonaca.
Session co-chair Keith A.A. Fox, MD, of the University of Edinburgh, noted that various studies have shown monotherapy with aspirin or another antiplatelet agent reduces stroke risk by about 15%, and now PEGASUS-TIMI 54 shows that ticagrelor plus aspirin decreases stroke risk by 25%. He posed a direct question: “How much is too much?”
“More and more antiplatelet therapy begets more bleeding, so I think that more than two agents may be approaching too much, although it really depends on what agents you’re using and in what dosages,” Dr. Bonaca replied.
He reported serving as a consultant to AstraZeneca, Merck, and Bayer.
Simultaneous with Dr. Bonaca’s presentation at ESC 2016 in Rome, the new report from PEGASUS-TIMI 54 including the four-trial meta-analysis was published online (Circulation. 2016 Aug 30. doi: circulationaha.116.024637).
AT THE ESC CONGRESS 2016
Key clinical point: Ticagrelor reduced the risk of a first stroke by 25% in patients with a prior MI.
Major finding: Ticagrelor, at the approved dose of 60 mg twice daily for long-term secondary cardiovascular prevention, reduced the risk of a first stroke by 25% in patients with a prior MI.
Data source: This secondary analysis of a randomized, double-blind, placebo-controlled trial included 14,112 stable patients with a prior MI 1-3 years earlier who were randomized to ticagrelor at 60 mg twice daily or placebo and followed prospectively for a median of 33 months.
Disclosures: PEGASUS-TIMI 54 was supported by AstraZeneca. The presenter of the updated analysis reported serving as a consultant to AstraZeneca, Merck, and Bayer.
September 2016: Click for Credit
Here are 5 articles in the August issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Women With BRCA1 Mutations at Higher Risk for Endometrial Cancers
To take the posttest, go to: http://bit.ly/2t6SPIY
Expires June 30, 2017
VITALSKey clinical point: Clinicians may wish to discuss the option of hysterectomy at the time of salpingo-oophorectomy in women with deleterious BRCA1 mutations.
Major finding: Among women with BRCA1 but not BRCA2 mutations there was increased risk for serous/serous-like endometrial carcinomas.
Data source: Prospective multicenter follow-up study of 1,083 women with BRCA mutations who underwent salpingo-oophorectomy without hysterectomy.
Disclosures: The study was supported by grants from the Department of Defense, National Institutes of Health, and public and private foundations. Coauthor Robert Soslow, MD, disclosed consulting for EMD Serono. No others reported conflicts of interest. The editorialists reported no conflicts of interest related to the study.
2. Cochrane Review: Topical Steroid—Vitamin D Combo Best for Scalp Psoriasis
To take the posttest, go to: http://bit.ly/2sIyLNI
Expires July 14, 2017
VITALSKey clinical point: The combination of a topical steroid and topical vitamin D is marginally better but with a similar safety profile to steroids alone as a treatment for psoriasis on the scalp.
Major finding: The combination of a topical steroid and vitamin D showed a small but statistically significant advantage over steroids alone, and a greater advantage over vitamin D alone.
Data source: A systematic review of 59 randomized controlled studies in 11,561 patients.
Disclosures: The study was supported by the Universidade Federal de São Paulo, Brazil; the Universidade Federal do Rio Grande do Norte, Brazil; and the National Institute for Health Research, United Kingdom. Six authors and one clinical referee declared speakers' fees, research grants, and funding from the pharmaceutical industry. One author had no conflicts of interest to disclose.
3. Study Finds Emergence of Azithromycin-resistant Gonorrhea
To take the posttest, go to: http://bit.ly/2u1nMmb
Expires July 16, 2017
VITALSKey clinical point:Resistance to azithromycin is emerging among patients diagnosed with gonorrhea.
Major finding: Among patients with gonorrhea, resistance to azithromycin increased from 0.6% in 2013 to 2.5% in 2014, predominantly in the Midwest.
Data source: An analysis of 5,093 Neisseria gonorrhoeae isolates from 27 clinics as part of the CDC's Gonococcal Isolate Surveillance Project.
Disclosures: The researchers had no financial disclosures.
4. Statins Improve Ovarian Cancer Survival
To take the posttest, go to: http://bit.ly/2t6swCF
Expires June 16, 2017
VITALSKey clinical point: The risk of all-cause mortality in ovarian cancer patients on statin therapy was reduced by one-third.
Major finding: Mean survival in a large cohort of women with stage III ovarian cancer was 5.8 months longer among those on statin therapy.
Data source: A retrospective study of 1,510 women diagnosed with epithelial ovarian cancer during 2007-2009.
Disclosures: Dr. Vogel reported having no financial conflicts regarding this study, conducted without commercial support.
5. Common Surgeries Linked to Chronic Opioid Use Among Opioid-naive Patients
To take the posttest, go to: http://bit.ly/2ub9fFg
Expires June 18, 2017
VITALSKey clinical point: Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially among those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories.
Major finding: After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; and total hip replacement and simple mastectomy almost threefold.
Data source: Insurance claims of more than 18 million people.
Disclosures: The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
Here are 5 articles in the August issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Women With BRCA1 Mutations at Higher Risk for Endometrial Cancers
To take the posttest, go to: http://bit.ly/2t6SPIY
Expires June 30, 2017
VITALSKey clinical point: Clinicians may wish to discuss the option of hysterectomy at the time of salpingo-oophorectomy in women with deleterious BRCA1 mutations.
Major finding: Among women with BRCA1 but not BRCA2 mutations there was increased risk for serous/serous-like endometrial carcinomas.
Data source: Prospective multicenter follow-up study of 1,083 women with BRCA mutations who underwent salpingo-oophorectomy without hysterectomy.
Disclosures: The study was supported by grants from the Department of Defense, National Institutes of Health, and public and private foundations. Coauthor Robert Soslow, MD, disclosed consulting for EMD Serono. No others reported conflicts of interest. The editorialists reported no conflicts of interest related to the study.
2. Cochrane Review: Topical Steroid—Vitamin D Combo Best for Scalp Psoriasis
To take the posttest, go to: http://bit.ly/2sIyLNI
Expires July 14, 2017
VITALSKey clinical point: The combination of a topical steroid and topical vitamin D is marginally better but with a similar safety profile to steroids alone as a treatment for psoriasis on the scalp.
Major finding: The combination of a topical steroid and vitamin D showed a small but statistically significant advantage over steroids alone, and a greater advantage over vitamin D alone.
Data source: A systematic review of 59 randomized controlled studies in 11,561 patients.
Disclosures: The study was supported by the Universidade Federal de São Paulo, Brazil; the Universidade Federal do Rio Grande do Norte, Brazil; and the National Institute for Health Research, United Kingdom. Six authors and one clinical referee declared speakers' fees, research grants, and funding from the pharmaceutical industry. One author had no conflicts of interest to disclose.
3. Study Finds Emergence of Azithromycin-resistant Gonorrhea
To take the posttest, go to: http://bit.ly/2u1nMmb
Expires July 16, 2017
VITALSKey clinical point:Resistance to azithromycin is emerging among patients diagnosed with gonorrhea.
Major finding: Among patients with gonorrhea, resistance to azithromycin increased from 0.6% in 2013 to 2.5% in 2014, predominantly in the Midwest.
Data source: An analysis of 5,093 Neisseria gonorrhoeae isolates from 27 clinics as part of the CDC's Gonococcal Isolate Surveillance Project.
Disclosures: The researchers had no financial disclosures.
4. Statins Improve Ovarian Cancer Survival
To take the posttest, go to: http://bit.ly/2t6swCF
Expires June 16, 2017
VITALSKey clinical point: The risk of all-cause mortality in ovarian cancer patients on statin therapy was reduced by one-third.
Major finding: Mean survival in a large cohort of women with stage III ovarian cancer was 5.8 months longer among those on statin therapy.
Data source: A retrospective study of 1,510 women diagnosed with epithelial ovarian cancer during 2007-2009.
Disclosures: Dr. Vogel reported having no financial conflicts regarding this study, conducted without commercial support.
5. Common Surgeries Linked to Chronic Opioid Use Among Opioid-naive Patients
To take the posttest, go to: http://bit.ly/2ub9fFg
Expires June 18, 2017
VITALSKey clinical point: Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially among those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories.
Major finding: After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; and total hip replacement and simple mastectomy almost threefold.
Data source: Insurance claims of more than 18 million people.
Disclosures: The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
Here are 5 articles in the August issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Women With BRCA1 Mutations at Higher Risk for Endometrial Cancers
To take the posttest, go to: http://bit.ly/2t6SPIY
Expires June 30, 2017
VITALSKey clinical point: Clinicians may wish to discuss the option of hysterectomy at the time of salpingo-oophorectomy in women with deleterious BRCA1 mutations.
Major finding: Among women with BRCA1 but not BRCA2 mutations there was increased risk for serous/serous-like endometrial carcinomas.
Data source: Prospective multicenter follow-up study of 1,083 women with BRCA mutations who underwent salpingo-oophorectomy without hysterectomy.
Disclosures: The study was supported by grants from the Department of Defense, National Institutes of Health, and public and private foundations. Coauthor Robert Soslow, MD, disclosed consulting for EMD Serono. No others reported conflicts of interest. The editorialists reported no conflicts of interest related to the study.
2. Cochrane Review: Topical Steroid—Vitamin D Combo Best for Scalp Psoriasis
To take the posttest, go to: http://bit.ly/2sIyLNI
Expires July 14, 2017
VITALSKey clinical point: The combination of a topical steroid and topical vitamin D is marginally better but with a similar safety profile to steroids alone as a treatment for psoriasis on the scalp.
Major finding: The combination of a topical steroid and vitamin D showed a small but statistically significant advantage over steroids alone, and a greater advantage over vitamin D alone.
Data source: A systematic review of 59 randomized controlled studies in 11,561 patients.
Disclosures: The study was supported by the Universidade Federal de São Paulo, Brazil; the Universidade Federal do Rio Grande do Norte, Brazil; and the National Institute for Health Research, United Kingdom. Six authors and one clinical referee declared speakers' fees, research grants, and funding from the pharmaceutical industry. One author had no conflicts of interest to disclose.
3. Study Finds Emergence of Azithromycin-resistant Gonorrhea
To take the posttest, go to: http://bit.ly/2u1nMmb
Expires July 16, 2017
VITALSKey clinical point:Resistance to azithromycin is emerging among patients diagnosed with gonorrhea.
Major finding: Among patients with gonorrhea, resistance to azithromycin increased from 0.6% in 2013 to 2.5% in 2014, predominantly in the Midwest.
Data source: An analysis of 5,093 Neisseria gonorrhoeae isolates from 27 clinics as part of the CDC's Gonococcal Isolate Surveillance Project.
Disclosures: The researchers had no financial disclosures.
4. Statins Improve Ovarian Cancer Survival
To take the posttest, go to: http://bit.ly/2t6swCF
Expires June 16, 2017
VITALSKey clinical point: The risk of all-cause mortality in ovarian cancer patients on statin therapy was reduced by one-third.
Major finding: Mean survival in a large cohort of women with stage III ovarian cancer was 5.8 months longer among those on statin therapy.
Data source: A retrospective study of 1,510 women diagnosed with epithelial ovarian cancer during 2007-2009.
Disclosures: Dr. Vogel reported having no financial conflicts regarding this study, conducted without commercial support.
5. Common Surgeries Linked to Chronic Opioid Use Among Opioid-naive Patients
To take the posttest, go to: http://bit.ly/2ub9fFg
Expires June 18, 2017
VITALSKey clinical point: Common surgeries increase the risk of chronic opioid use in opioid-naive adults, especially among those using antidepressants or benzodiazepines before their operations, and those with substance abuse histories.
Major finding: After adjustment for potential confounders, knee replacement increased the risk fivefold; open cholecystectomy almost fourfold; and total hip replacement and simple mastectomy almost threefold.
Data source: Insurance claims of more than 18 million people.
Disclosures: The authors had no disclosures. The work was funded in part by the Foundation for Anesthesia Education and Research and the Anesthesia Quality Institute. Claims data came from MarketScan (Truven Health Analytics).
ENSURE-AF trial supports edoxaban for electrical cardioversion
ROME – Results of the largest-ever randomized clinical trial of anticoagulation for electrical cardioversion of patients with nonvalvular atrial fibrillation demonstrate that edoxaban is a safe, effective, and convenient alternative to the standard strategy of enoxaparin as a bridge to warfarin.
The ENSURE-AF trial was a phase IIIb study involving 2,199 patients with atrial fibrillation who underwent electrical cardioversion at 239 sites in the United States and 19 European countries. The key finding: The edoxaban-treated group had rates of thromboembolism and major bleeding at 28-30 days follow-up similar to those of the enoxaparin/warfarin-treated controls.
And edoxaban offered a major practical advantage: Because “edoxaban kicks in within 2 hours, you can do the procedure just 2 hours after initiation of therapy in a patient with a reassuring transesophageal echocardiographic exam, which is definitely not possible with warfarin,” Andreas Goette, MD, observed at the annual congress of the European Society of Cardiology.
Roughly half of participants were treated at centers that don’t routinely use a transesophageal echo-guided management strategy and therefore delayed cardioversion until patients were anticoagulated for at least 3 weeks. The safety and efficacy outcomes were similar regardless of whether or not transesophageal echocardiography (TEE) guidance was used, according to Dr. Goette of St. Vincenz Hospital in Paderborn, Germany.
Edoxaban (Savaysa) was prescribed at 60 mg once daily except in patients weighing 60 kg or less or having a creatinine clearance rate of 15-50 mL/min, in which case they received 30 mg once daily. In the control arm, enoxaparin (Lovenox) was used until warfarin achieved an International Normalized Ratio of 2.0-3.0. Patients in the enoxaparin/warfarin arm spent a mean of 71% of their treatment time within the target INR range.
The primary efficacy outcome was the 28-day composite of stroke or other systemic embolic events, MI, or cardiovascular mortality. The rate was 0.5% in the edoxaban arm and 1.0% in the enoxaparin/warfarin group. In patients whose management strategy was TEE-guided, the rate was 0.3% in the edoxaban group and 0.8% with enoxaparin/warfarin. In non-TEE-guided patients, the rates were 0.6% and 1.2% with edoxaban and warfarin, respectively.
Although rates were consistently numerically lower in the edoxaban group, the differences did not reach statistical significance, Dr. Goette explained.
The combined rate of major or clinically relevant nonmajor bleeding through 30 days was 1.5% with edoxaban and similar at 1.0% with enoxaparin plus warfarin. Three patients in the edoxaban group experienced a major bleeding event, as did five in the comparator arm.
Because anticoagulation with edoxaban is so convenient and allows cardioversion to safely be performed in short order, the ENSURE-AF investigators are in the process of calculating the potential savings in health care costs obtainable through this strategy, the cardiologist said.
ENSURE-AF provides the first prospective randomized data on the use of edoxaban as an alternative to warfarin for pericardioversion anticoagulation. There has been one other randomized trial of a novel oral anticoagulant (NOAC) in this setting, the 1,504-patient X-VeRT trial (Eur Heart J. 2014 Dec 14;35[47]:3346-55), involving rivaroxaban (Xarelto).
Riccardo Cappato, MD, first author of the X-VeRT publication, served as the designated discussant for ENSURE-AF. He noted that the results of the two trials are “completely superimposable.” Rates of the composite efficacy endpoint were identical at 0.5% for both NOACs versus 1.0% for the vitamin K antagonist arms of X-VeRT and ENSURE-AF. The major bleeding rates also were identical for edoxaban and rivaroxaban in the two studies. Moreover, the major bleeding rates associated with warfarin or other vitamin K antagonists were spot-on the same in the two trials.
“It’s a rather unusual situation for such large numbers of patients,” observed Dr. Cappato of Humanitas Research Institute in Milan.
“These data go very clearly in the same direction. I think a good take-home message here for us today is that both of these novel oral anticoagulants can be safely and efficaciously applied to patients undergoing elective cardioversion of nonvalvular atrial fibrillation,” he added.
In an interview, Mark A. Creager, MD, immediate past president of the American Heart Association, said that many U.S. physicians are switching to NOACs for this purpose.
“We are already using the novel oral anticoagulants to facilitate anticoagulation for patients undergoing cardioversion, so ENSURE-AF provides objective evidence that edoxaban is a reasonable drug,” said Dr. Creager, director of the Dartmouth-Hitchcock Heart and Vascular Center in New Hampshire.
The ENSURE-AF trial was funded by Daiichi Sankyo. Dr. Goette and Dr. Cappato reported receiving research grants from and serving as consultants to that company and other pharmaceutical and medical device manufacturers.
Simultaneously with Dr. Goette’s presentation in Rome, the ENSURE-AF results were published online Aug. 30 in The Lancet.
ROME – Results of the largest-ever randomized clinical trial of anticoagulation for electrical cardioversion of patients with nonvalvular atrial fibrillation demonstrate that edoxaban is a safe, effective, and convenient alternative to the standard strategy of enoxaparin as a bridge to warfarin.
The ENSURE-AF trial was a phase IIIb study involving 2,199 patients with atrial fibrillation who underwent electrical cardioversion at 239 sites in the United States and 19 European countries. The key finding: The edoxaban-treated group had rates of thromboembolism and major bleeding at 28-30 days follow-up similar to those of the enoxaparin/warfarin-treated controls.
And edoxaban offered a major practical advantage: Because “edoxaban kicks in within 2 hours, you can do the procedure just 2 hours after initiation of therapy in a patient with a reassuring transesophageal echocardiographic exam, which is definitely not possible with warfarin,” Andreas Goette, MD, observed at the annual congress of the European Society of Cardiology.
Roughly half of participants were treated at centers that don’t routinely use a transesophageal echo-guided management strategy and therefore delayed cardioversion until patients were anticoagulated for at least 3 weeks. The safety and efficacy outcomes were similar regardless of whether or not transesophageal echocardiography (TEE) guidance was used, according to Dr. Goette of St. Vincenz Hospital in Paderborn, Germany.
Edoxaban (Savaysa) was prescribed at 60 mg once daily except in patients weighing 60 kg or less or having a creatinine clearance rate of 15-50 mL/min, in which case they received 30 mg once daily. In the control arm, enoxaparin (Lovenox) was used until warfarin achieved an International Normalized Ratio of 2.0-3.0. Patients in the enoxaparin/warfarin arm spent a mean of 71% of their treatment time within the target INR range.
The primary efficacy outcome was the 28-day composite of stroke or other systemic embolic events, MI, or cardiovascular mortality. The rate was 0.5% in the edoxaban arm and 1.0% in the enoxaparin/warfarin group. In patients whose management strategy was TEE-guided, the rate was 0.3% in the edoxaban group and 0.8% with enoxaparin/warfarin. In non-TEE-guided patients, the rates were 0.6% and 1.2% with edoxaban and warfarin, respectively.
Although rates were consistently numerically lower in the edoxaban group, the differences did not reach statistical significance, Dr. Goette explained.
The combined rate of major or clinically relevant nonmajor bleeding through 30 days was 1.5% with edoxaban and similar at 1.0% with enoxaparin plus warfarin. Three patients in the edoxaban group experienced a major bleeding event, as did five in the comparator arm.
Because anticoagulation with edoxaban is so convenient and allows cardioversion to safely be performed in short order, the ENSURE-AF investigators are in the process of calculating the potential savings in health care costs obtainable through this strategy, the cardiologist said.
ENSURE-AF provides the first prospective randomized data on the use of edoxaban as an alternative to warfarin for pericardioversion anticoagulation. There has been one other randomized trial of a novel oral anticoagulant (NOAC) in this setting, the 1,504-patient X-VeRT trial (Eur Heart J. 2014 Dec 14;35[47]:3346-55), involving rivaroxaban (Xarelto).
Riccardo Cappato, MD, first author of the X-VeRT publication, served as the designated discussant for ENSURE-AF. He noted that the results of the two trials are “completely superimposable.” Rates of the composite efficacy endpoint were identical at 0.5% for both NOACs versus 1.0% for the vitamin K antagonist arms of X-VeRT and ENSURE-AF. The major bleeding rates also were identical for edoxaban and rivaroxaban in the two studies. Moreover, the major bleeding rates associated with warfarin or other vitamin K antagonists were spot-on the same in the two trials.
“It’s a rather unusual situation for such large numbers of patients,” observed Dr. Cappato of Humanitas Research Institute in Milan.
“These data go very clearly in the same direction. I think a good take-home message here for us today is that both of these novel oral anticoagulants can be safely and efficaciously applied to patients undergoing elective cardioversion of nonvalvular atrial fibrillation,” he added.
In an interview, Mark A. Creager, MD, immediate past president of the American Heart Association, said that many U.S. physicians are switching to NOACs for this purpose.
“We are already using the novel oral anticoagulants to facilitate anticoagulation for patients undergoing cardioversion, so ENSURE-AF provides objective evidence that edoxaban is a reasonable drug,” said Dr. Creager, director of the Dartmouth-Hitchcock Heart and Vascular Center in New Hampshire.
The ENSURE-AF trial was funded by Daiichi Sankyo. Dr. Goette and Dr. Cappato reported receiving research grants from and serving as consultants to that company and other pharmaceutical and medical device manufacturers.
Simultaneously with Dr. Goette’s presentation in Rome, the ENSURE-AF results were published online Aug. 30 in The Lancet.
ROME – Results of the largest-ever randomized clinical trial of anticoagulation for electrical cardioversion of patients with nonvalvular atrial fibrillation demonstrate that edoxaban is a safe, effective, and convenient alternative to the standard strategy of enoxaparin as a bridge to warfarin.
The ENSURE-AF trial was a phase IIIb study involving 2,199 patients with atrial fibrillation who underwent electrical cardioversion at 239 sites in the United States and 19 European countries. The key finding: The edoxaban-treated group had rates of thromboembolism and major bleeding at 28-30 days follow-up similar to those of the enoxaparin/warfarin-treated controls.
And edoxaban offered a major practical advantage: Because “edoxaban kicks in within 2 hours, you can do the procedure just 2 hours after initiation of therapy in a patient with a reassuring transesophageal echocardiographic exam, which is definitely not possible with warfarin,” Andreas Goette, MD, observed at the annual congress of the European Society of Cardiology.
Roughly half of participants were treated at centers that don’t routinely use a transesophageal echo-guided management strategy and therefore delayed cardioversion until patients were anticoagulated for at least 3 weeks. The safety and efficacy outcomes were similar regardless of whether or not transesophageal echocardiography (TEE) guidance was used, according to Dr. Goette of St. Vincenz Hospital in Paderborn, Germany.
Edoxaban (Savaysa) was prescribed at 60 mg once daily except in patients weighing 60 kg or less or having a creatinine clearance rate of 15-50 mL/min, in which case they received 30 mg once daily. In the control arm, enoxaparin (Lovenox) was used until warfarin achieved an International Normalized Ratio of 2.0-3.0. Patients in the enoxaparin/warfarin arm spent a mean of 71% of their treatment time within the target INR range.
The primary efficacy outcome was the 28-day composite of stroke or other systemic embolic events, MI, or cardiovascular mortality. The rate was 0.5% in the edoxaban arm and 1.0% in the enoxaparin/warfarin group. In patients whose management strategy was TEE-guided, the rate was 0.3% in the edoxaban group and 0.8% with enoxaparin/warfarin. In non-TEE-guided patients, the rates were 0.6% and 1.2% with edoxaban and warfarin, respectively.
Although rates were consistently numerically lower in the edoxaban group, the differences did not reach statistical significance, Dr. Goette explained.
The combined rate of major or clinically relevant nonmajor bleeding through 30 days was 1.5% with edoxaban and similar at 1.0% with enoxaparin plus warfarin. Three patients in the edoxaban group experienced a major bleeding event, as did five in the comparator arm.
Because anticoagulation with edoxaban is so convenient and allows cardioversion to safely be performed in short order, the ENSURE-AF investigators are in the process of calculating the potential savings in health care costs obtainable through this strategy, the cardiologist said.
ENSURE-AF provides the first prospective randomized data on the use of edoxaban as an alternative to warfarin for pericardioversion anticoagulation. There has been one other randomized trial of a novel oral anticoagulant (NOAC) in this setting, the 1,504-patient X-VeRT trial (Eur Heart J. 2014 Dec 14;35[47]:3346-55), involving rivaroxaban (Xarelto).
Riccardo Cappato, MD, first author of the X-VeRT publication, served as the designated discussant for ENSURE-AF. He noted that the results of the two trials are “completely superimposable.” Rates of the composite efficacy endpoint were identical at 0.5% for both NOACs versus 1.0% for the vitamin K antagonist arms of X-VeRT and ENSURE-AF. The major bleeding rates also were identical for edoxaban and rivaroxaban in the two studies. Moreover, the major bleeding rates associated with warfarin or other vitamin K antagonists were spot-on the same in the two trials.
“It’s a rather unusual situation for such large numbers of patients,” observed Dr. Cappato of Humanitas Research Institute in Milan.
“These data go very clearly in the same direction. I think a good take-home message here for us today is that both of these novel oral anticoagulants can be safely and efficaciously applied to patients undergoing elective cardioversion of nonvalvular atrial fibrillation,” he added.
In an interview, Mark A. Creager, MD, immediate past president of the American Heart Association, said that many U.S. physicians are switching to NOACs for this purpose.
“We are already using the novel oral anticoagulants to facilitate anticoagulation for patients undergoing cardioversion, so ENSURE-AF provides objective evidence that edoxaban is a reasonable drug,” said Dr. Creager, director of the Dartmouth-Hitchcock Heart and Vascular Center in New Hampshire.
The ENSURE-AF trial was funded by Daiichi Sankyo. Dr. Goette and Dr. Cappato reported receiving research grants from and serving as consultants to that company and other pharmaceutical and medical device manufacturers.
Simultaneously with Dr. Goette’s presentation in Rome, the ENSURE-AF results were published online Aug. 30 in The Lancet.
AT THE ESC CONGRESS 2016
Key clinical point: Edoxaban is a safe, effective, and convenient alternative to warfarin for anticoagulation in patients undergoing electrical cardioversion of atrial fibrillation.
Major finding: The composite endpoint of stroke, other systemic embolic events, MI, or cardiovascular death occurred in 0.5% of patients with atrial fibrillation assigned to edoxaban for pericardioversion anticoagulation and in 1.0% on enoxaparin bridging to warfarin.
Data source: A randomized prospective multinational trial of 2,199 patients scheduled for electrical cardioversion of their nonvalvular atrial fibrillation.
Disclosures: The ENSURE-AF trial was funded by Daiichi Sankyo. The presenter reported receiving research grants from and serving as a consultant to that company as well as other pharmaceutical and medical device manufacturers.
Postpartum HIV treatment reduces key maternal illnesses
DURBAN, SOUTH AFRICA – HIV-infected women who remained on antiretroviral therapy throughout the postpartum period reduced their risk of clinical stage 2 or 3 HIV disease events by 53%, compared with those who stopped treatment postpartum in the PROMISE 1077HS trial, Judith Currier, MD, reported at the 21st International AIDS Conference.
PROMISE (Promoting Maternal and Infant Survival Everywhere) is an ongoing multinational, multicomponent series of clinical trials. PROMISE 1077HS was designed to assess the risks and benefits of continued antiretroviral therapy (ART), compared with stopping therapy among nonbreastfeeding women postpartum, explained Dr. Currier, professor of medicine and chief of infectious diseases at the University of California, Los Angeles.
PROMISE 1077HS included 1,653 HIV-positive women in the United States and seven low- or middle-income countries. All participants were relatively healthy as evidenced by their median CD4+ count of 550 cells/mm3 prior to starting ART in pregnancy. None were planning to breastfeed. Upon delivery, the women were randomized to continue ART – the chief regimen was ritonavir-boosted lopinavir (Kaletra) plus tenofovir/emtricitabine (Truvada) – or stop therapy, restarting only when their CD4+ count fell below 350 cells/mm3.
Participants were prospectively followed for a median of 2.3 years post delivery. At that point, in summer 2015, the results of the landmark START trial were released (N Engl J Med. 2015 Aug 27;373[9]:795-807), paving the way for the current global strategy of ART for life in HIV-infected patients regardless of their CD4+ cell count.
The primary efficacy endpoint in PROMISE 1077HS was a composite of an AIDS event, death, or a serious cardiovascular, renal, or hepatic event. In this relatively healthy population, too few of these events occurred to allow the researchers to draw conclusions (four in the continued-ART group and six in the controls who stopped ART post partum).
But the secondary endpoint of time to World Health Organization (WHO) clinical stage 2 or 3 HIV disease events was a different story. A total of 39 of these events occurred in the continued-ART group, for a rate of 2.02% per year, compared with 80 events and a rate of 4.36% per year in controls. That difference translated into a 53% relative risk reduction. Some key WHO clinical stage 2 and 3 HIV disease events include pulmonary tuberculosis, herpes zoster, oral candidiasis, thrombocytopenia, and bacterial infection.
Fully 23% of patients in the continued-ART group had laboratory-confirmed virologic failure as defined by a viral load of HIV RNA in excess of 1,000 copies/mL after at least 24 weeks of postpartum ART. Additionally, resistance testing indicated two-thirds of affected patients showed no evidence of resistance at the time of virologic failure, meaning their viremia was due to nonadherence to ART.
“This virologic failure rate highlights the importance of the challenge of adherence in this population,” Dr. Currier said. “Interventions to improve adherence as well as studies to examine newer regimens with a high genetic barrier to resistance are needed to ensure maximal long-term benefit from this strategy of continued ART postpartum.”
The PROMISE studies are funded by the National Institutes of Health. Dr. Currier reported having no financial disclosures.
DURBAN, SOUTH AFRICA – HIV-infected women who remained on antiretroviral therapy throughout the postpartum period reduced their risk of clinical stage 2 or 3 HIV disease events by 53%, compared with those who stopped treatment postpartum in the PROMISE 1077HS trial, Judith Currier, MD, reported at the 21st International AIDS Conference.
PROMISE (Promoting Maternal and Infant Survival Everywhere) is an ongoing multinational, multicomponent series of clinical trials. PROMISE 1077HS was designed to assess the risks and benefits of continued antiretroviral therapy (ART), compared with stopping therapy among nonbreastfeeding women postpartum, explained Dr. Currier, professor of medicine and chief of infectious diseases at the University of California, Los Angeles.
PROMISE 1077HS included 1,653 HIV-positive women in the United States and seven low- or middle-income countries. All participants were relatively healthy as evidenced by their median CD4+ count of 550 cells/mm3 prior to starting ART in pregnancy. None were planning to breastfeed. Upon delivery, the women were randomized to continue ART – the chief regimen was ritonavir-boosted lopinavir (Kaletra) plus tenofovir/emtricitabine (Truvada) – or stop therapy, restarting only when their CD4+ count fell below 350 cells/mm3.
Participants were prospectively followed for a median of 2.3 years post delivery. At that point, in summer 2015, the results of the landmark START trial were released (N Engl J Med. 2015 Aug 27;373[9]:795-807), paving the way for the current global strategy of ART for life in HIV-infected patients regardless of their CD4+ cell count.
The primary efficacy endpoint in PROMISE 1077HS was a composite of an AIDS event, death, or a serious cardiovascular, renal, or hepatic event. In this relatively healthy population, too few of these events occurred to allow the researchers to draw conclusions (four in the continued-ART group and six in the controls who stopped ART post partum).

But the secondary endpoint of time to World Health Organization (WHO) clinical stage 2 or 3 HIV disease events was a different story. A total of 39 of these events occurred in the continued-ART group, for a rate of 2.02% per year, compared with 80 events and a rate of 4.36% per year in controls. That difference translated into a 53% relative risk reduction. Some key WHO clinical stage 2 and 3 HIV disease events include pulmonary tuberculosis, herpes zoster, oral candidiasis, thrombocytopenia, and bacterial infection.
Fully 23% of patients in the continued-ART group had laboratory-confirmed virologic failure as defined by a viral load of HIV RNA in excess of 1,000 copies/mL after at least 24 weeks of postpartum ART. Additionally, resistance testing indicated two-thirds of affected patients showed no evidence of resistance at the time of virologic failure, meaning their viremia was due to nonadherence to ART.
“This virologic failure rate highlights the importance of the challenge of adherence in this population,” Dr. Currier said. “Interventions to improve adherence as well as studies to examine newer regimens with a high genetic barrier to resistance are needed to ensure maximal long-term benefit from this strategy of continued ART postpartum.”
The PROMISE studies are funded by the National Institutes of Health. Dr. Currier reported having no financial disclosures.
DURBAN, SOUTH AFRICA – HIV-infected women who remained on antiretroviral therapy throughout the postpartum period reduced their risk of clinical stage 2 or 3 HIV disease events by 53%, compared with those who stopped treatment postpartum in the PROMISE 1077HS trial, Judith Currier, MD, reported at the 21st International AIDS Conference.
PROMISE (Promoting Maternal and Infant Survival Everywhere) is an ongoing multinational, multicomponent series of clinical trials. PROMISE 1077HS was designed to assess the risks and benefits of continued antiretroviral therapy (ART), compared with stopping therapy among nonbreastfeeding women postpartum, explained Dr. Currier, professor of medicine and chief of infectious diseases at the University of California, Los Angeles.
PROMISE 1077HS included 1,653 HIV-positive women in the United States and seven low- or middle-income countries. All participants were relatively healthy as evidenced by their median CD4+ count of 550 cells/mm3 prior to starting ART in pregnancy. None were planning to breastfeed. Upon delivery, the women were randomized to continue ART – the chief regimen was ritonavir-boosted lopinavir (Kaletra) plus tenofovir/emtricitabine (Truvada) – or stop therapy, restarting only when their CD4+ count fell below 350 cells/mm3.
Participants were prospectively followed for a median of 2.3 years post delivery. At that point, in summer 2015, the results of the landmark START trial were released (N Engl J Med. 2015 Aug 27;373[9]:795-807), paving the way for the current global strategy of ART for life in HIV-infected patients regardless of their CD4+ cell count.
The primary efficacy endpoint in PROMISE 1077HS was a composite of an AIDS event, death, or a serious cardiovascular, renal, or hepatic event. In this relatively healthy population, too few of these events occurred to allow the researchers to draw conclusions (four in the continued-ART group and six in the controls who stopped ART post partum).

But the secondary endpoint of time to World Health Organization (WHO) clinical stage 2 or 3 HIV disease events was a different story. A total of 39 of these events occurred in the continued-ART group, for a rate of 2.02% per year, compared with 80 events and a rate of 4.36% per year in controls. That difference translated into a 53% relative risk reduction. Some key WHO clinical stage 2 and 3 HIV disease events include pulmonary tuberculosis, herpes zoster, oral candidiasis, thrombocytopenia, and bacterial infection.
Fully 23% of patients in the continued-ART group had laboratory-confirmed virologic failure as defined by a viral load of HIV RNA in excess of 1,000 copies/mL after at least 24 weeks of postpartum ART. Additionally, resistance testing indicated two-thirds of affected patients showed no evidence of resistance at the time of virologic failure, meaning their viremia was due to nonadherence to ART.
“This virologic failure rate highlights the importance of the challenge of adherence in this population,” Dr. Currier said. “Interventions to improve adherence as well as studies to examine newer regimens with a high genetic barrier to resistance are needed to ensure maximal long-term benefit from this strategy of continued ART postpartum.”
The PROMISE studies are funded by the National Institutes of Health. Dr. Currier reported having no financial disclosures.
AT AIDS 2016
Key clinical point: Women with HIV should continue antiretroviral therapy post partum.
Major finding: HIV-infected women who continued antiretroviral therapy post partum experienced 53% fewer WHO clinical stage 2 or 3 HIV disease events than women assigned to stop therapy after delivery.
Data source: The PROMISE 1077HS study included 1,653 HIV-positive, nonbreastfeeding women randomized to either continue or stop antiretroviral therapy post partum.
Disclosures: The PROMISE studies are funded by the National Institutes of Health. Dr. Currier reported having no financial disclosures.
VIDEO: Apheresis shows promise for refractory angina with high Lp(a)
ROME – Weekly lipoprotein apheresis in patients with highly refractory angina accompanied by high plasma lipoprotein(a) without elevated LDL cholesterol led to significantly improved myocardial blood flow in a randomized, blinded, sham-controlled clinical trial, Tina Khan, MD, reported at the annual congress of the European Society of Cardiology.
Participants also experienced clinically meaningful improvements in the secondary endpoints of quality of life, angina symptoms, exercise capacity, and atheroma burden, added Dr. Khan of Imperial College London.
Angina pectoris that is refractory to maximal pharmacologic, percutaneous, and surgical interventions is a major and growing problem. More than 100,000 new cases occur per year in the United States.
“We have a desperate need to develop new therapeutic options,” she said.
Lipoprotein(a), or Lp(a), is a potent independent cardiovascular risk factor. And it figures prominently in refractory angina. Indeed, 60% of the patients with refractory angina screened by Dr. Khan for her clinical trial had an isolated plasma Lp(a) level of 50 mg/dL or more, the threshold at which cardiovascular risk sharply increases. Statins have no effect on Lp(a) levels.
While a couple of observational cohort studies have suggested that reducing elevated Lp(a) in patients with cardiovascular disease is associated with a decrease in major adverse cardiovascular events, until now there have been no randomized controlled trials of apheresis as Lp(a)-lowering therapy in patients with refractory angina, according to Dr. Khan.
She presented a randomized, crossover design study in 20 patients with severe refractory angina and an Lp(a) in excess of 50 mg/dL but no elevation in LDL. They underwent 3 months of blinded weekly extracorporeal lipoprotein apheresis using a dextran sulfate filtration system or sham apheresis, followed by a month-long washout period. Participants were then crossed over to the other study arm to increase the statistical power of this small study.
The primary study endpoint was change in myocardial perfusion reserve as measured by cardiovascular magnetic resonance imaging at baseline and after 3 months of true or sham apheresis. The myocardial perfusion reserve index improved by a net of 0.63 after apheresis from a baseline of 1.45. This effect was strongly driven by a substantial increase in stress myocardial perfusion, with very little change in perfusion at rest.
Significant improvements were also recorded after apheresis in the secondary endpoints of change in carotid atheroma as reflected in total carotid wall volume, improvement on various domains of the Seattle Angina Questionnaire, physical limitations as scored on the SF-36, and exercise capacity as measured on the 6-minute walk test.
Discussant Peter Libby, MD, was effusive in his praise for Dr. Khan’s study.
“This is a wonderful example of how we may be able to offer new hope for patients and families with high Lp(a),” declared Dr. Libby, chief of cardiovascular medicine at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, Boston.
He approved of the methodology and embraced what he called “the intriguing preliminary picture of benefit for lowering Lp(a).”
Most of all, he was pleased that Dr. Khan’s well conducted albeit small study has thrown a spotlight on Lp(a), which he characterized as a greatly underappreciated causal risk factor for a range of cardiovascular diseases.
“Lp(a) stands out like a Manhattan skyscraper as the major driver of calcific aortic stenosis, an epidemic that we see in our aging population,” he observed.
He added that topics worthy of further research with regard to Lp(a)-lowering apheresis as a treatment for refractory angina include the question of whether the mechanism of benefit involves structural changes in atherosclerosis or functional changes. And if it’s the latter, is it a matter of changes in microvascular function, macrovascular function, or a combination of the two?
Apheresis is extremely costly, inconvenient, and invasive, and there are only several dozen apheresis centers in the United States. So the future of Lp(a)-lowering to treat refractory angina, calcific aortic stenosis, and other cardiovascular conditions where elevated Lp(a) is an important player may lie in pharmacotherapy with the PCSK9 inhibitors, Dr. Libby predicted.
He cited “very promising” data showing that evolocumab (Repatha) and alirocumab (Praluent) lower Lp(a) in dose-dependent fashion. Ongoing very large clinical trials with hard clinical endpoints should eventually provide key information regarding the cardiovascular benefits of lowering Lp(a).
“We may be entering an era where we may be able to offer our patients and families – because this is often a familial problem – a non-apheresis approach to controlling what we are learning is a very important causal risk factor for atherosclerosis,” Dr. Libby said.
Patrick M. Moriarty, MD, said in a video interview that he was very intrigued by the results, and “not personally surprised.” They should stimulate interest in cardiologists to start measuring Lp(a) in their patients like those in this study, in whom the disease severity doesn’t match up with the clinical risk factors, said Dr. Moriarty of the University of Kansas, Kansas City.
Dr. Khan’s study was funded by the UK National Institute for Health Research. She reported having no financial conflicts of interest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ROME – Weekly lipoprotein apheresis in patients with highly refractory angina accompanied by high plasma lipoprotein(a) without elevated LDL cholesterol led to significantly improved myocardial blood flow in a randomized, blinded, sham-controlled clinical trial, Tina Khan, MD, reported at the annual congress of the European Society of Cardiology.
Participants also experienced clinically meaningful improvements in the secondary endpoints of quality of life, angina symptoms, exercise capacity, and atheroma burden, added Dr. Khan of Imperial College London.
Angina pectoris that is refractory to maximal pharmacologic, percutaneous, and surgical interventions is a major and growing problem. More than 100,000 new cases occur per year in the United States.
“We have a desperate need to develop new therapeutic options,” she said.
Lipoprotein(a), or Lp(a), is a potent independent cardiovascular risk factor. And it figures prominently in refractory angina. Indeed, 60% of the patients with refractory angina screened by Dr. Khan for her clinical trial had an isolated plasma Lp(a) level of 50 mg/dL or more, the threshold at which cardiovascular risk sharply increases. Statins have no effect on Lp(a) levels.
While a couple of observational cohort studies have suggested that reducing elevated Lp(a) in patients with cardiovascular disease is associated with a decrease in major adverse cardiovascular events, until now there have been no randomized controlled trials of apheresis as Lp(a)-lowering therapy in patients with refractory angina, according to Dr. Khan.
She presented a randomized, crossover design study in 20 patients with severe refractory angina and an Lp(a) in excess of 50 mg/dL but no elevation in LDL. They underwent 3 months of blinded weekly extracorporeal lipoprotein apheresis using a dextran sulfate filtration system or sham apheresis, followed by a month-long washout period. Participants were then crossed over to the other study arm to increase the statistical power of this small study.
The primary study endpoint was change in myocardial perfusion reserve as measured by cardiovascular magnetic resonance imaging at baseline and after 3 months of true or sham apheresis. The myocardial perfusion reserve index improved by a net of 0.63 after apheresis from a baseline of 1.45. This effect was strongly driven by a substantial increase in stress myocardial perfusion, with very little change in perfusion at rest.
Significant improvements were also recorded after apheresis in the secondary endpoints of change in carotid atheroma as reflected in total carotid wall volume, improvement on various domains of the Seattle Angina Questionnaire, physical limitations as scored on the SF-36, and exercise capacity as measured on the 6-minute walk test.
Discussant Peter Libby, MD, was effusive in his praise for Dr. Khan’s study.
“This is a wonderful example of how we may be able to offer new hope for patients and families with high Lp(a),” declared Dr. Libby, chief of cardiovascular medicine at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, Boston.
He approved of the methodology and embraced what he called “the intriguing preliminary picture of benefit for lowering Lp(a).”
Most of all, he was pleased that Dr. Khan’s well conducted albeit small study has thrown a spotlight on Lp(a), which he characterized as a greatly underappreciated causal risk factor for a range of cardiovascular diseases.
“Lp(a) stands out like a Manhattan skyscraper as the major driver of calcific aortic stenosis, an epidemic that we see in our aging population,” he observed.
He added that topics worthy of further research with regard to Lp(a)-lowering apheresis as a treatment for refractory angina include the question of whether the mechanism of benefit involves structural changes in atherosclerosis or functional changes. And if it’s the latter, is it a matter of changes in microvascular function, macrovascular function, or a combination of the two?
Apheresis is extremely costly, inconvenient, and invasive, and there are only several dozen apheresis centers in the United States. So the future of Lp(a)-lowering to treat refractory angina, calcific aortic stenosis, and other cardiovascular conditions where elevated Lp(a) is an important player may lie in pharmacotherapy with the PCSK9 inhibitors, Dr. Libby predicted.
He cited “very promising” data showing that evolocumab (Repatha) and alirocumab (Praluent) lower Lp(a) in dose-dependent fashion. Ongoing very large clinical trials with hard clinical endpoints should eventually provide key information regarding the cardiovascular benefits of lowering Lp(a).
“We may be entering an era where we may be able to offer our patients and families – because this is often a familial problem – a non-apheresis approach to controlling what we are learning is a very important causal risk factor for atherosclerosis,” Dr. Libby said.
Patrick M. Moriarty, MD, said in a video interview that he was very intrigued by the results, and “not personally surprised.” They should stimulate interest in cardiologists to start measuring Lp(a) in their patients like those in this study, in whom the disease severity doesn’t match up with the clinical risk factors, said Dr. Moriarty of the University of Kansas, Kansas City.
Dr. Khan’s study was funded by the UK National Institute for Health Research. She reported having no financial conflicts of interest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ROME – Weekly lipoprotein apheresis in patients with highly refractory angina accompanied by high plasma lipoprotein(a) without elevated LDL cholesterol led to significantly improved myocardial blood flow in a randomized, blinded, sham-controlled clinical trial, Tina Khan, MD, reported at the annual congress of the European Society of Cardiology.
Participants also experienced clinically meaningful improvements in the secondary endpoints of quality of life, angina symptoms, exercise capacity, and atheroma burden, added Dr. Khan of Imperial College London.
Angina pectoris that is refractory to maximal pharmacologic, percutaneous, and surgical interventions is a major and growing problem. More than 100,000 new cases occur per year in the United States.
“We have a desperate need to develop new therapeutic options,” she said.
Lipoprotein(a), or Lp(a), is a potent independent cardiovascular risk factor. And it figures prominently in refractory angina. Indeed, 60% of the patients with refractory angina screened by Dr. Khan for her clinical trial had an isolated plasma Lp(a) level of 50 mg/dL or more, the threshold at which cardiovascular risk sharply increases. Statins have no effect on Lp(a) levels.
While a couple of observational cohort studies have suggested that reducing elevated Lp(a) in patients with cardiovascular disease is associated with a decrease in major adverse cardiovascular events, until now there have been no randomized controlled trials of apheresis as Lp(a)-lowering therapy in patients with refractory angina, according to Dr. Khan.
She presented a randomized, crossover design study in 20 patients with severe refractory angina and an Lp(a) in excess of 50 mg/dL but no elevation in LDL. They underwent 3 months of blinded weekly extracorporeal lipoprotein apheresis using a dextran sulfate filtration system or sham apheresis, followed by a month-long washout period. Participants were then crossed over to the other study arm to increase the statistical power of this small study.
The primary study endpoint was change in myocardial perfusion reserve as measured by cardiovascular magnetic resonance imaging at baseline and after 3 months of true or sham apheresis. The myocardial perfusion reserve index improved by a net of 0.63 after apheresis from a baseline of 1.45. This effect was strongly driven by a substantial increase in stress myocardial perfusion, with very little change in perfusion at rest.
Significant improvements were also recorded after apheresis in the secondary endpoints of change in carotid atheroma as reflected in total carotid wall volume, improvement on various domains of the Seattle Angina Questionnaire, physical limitations as scored on the SF-36, and exercise capacity as measured on the 6-minute walk test.
Discussant Peter Libby, MD, was effusive in his praise for Dr. Khan’s study.
“This is a wonderful example of how we may be able to offer new hope for patients and families with high Lp(a),” declared Dr. Libby, chief of cardiovascular medicine at Brigham and Women’s Hospital and professor of medicine at Harvard Medical School, Boston.
He approved of the methodology and embraced what he called “the intriguing preliminary picture of benefit for lowering Lp(a).”
Most of all, he was pleased that Dr. Khan’s well conducted albeit small study has thrown a spotlight on Lp(a), which he characterized as a greatly underappreciated causal risk factor for a range of cardiovascular diseases.
“Lp(a) stands out like a Manhattan skyscraper as the major driver of calcific aortic stenosis, an epidemic that we see in our aging population,” he observed.
He added that topics worthy of further research with regard to Lp(a)-lowering apheresis as a treatment for refractory angina include the question of whether the mechanism of benefit involves structural changes in atherosclerosis or functional changes. And if it’s the latter, is it a matter of changes in microvascular function, macrovascular function, or a combination of the two?
Apheresis is extremely costly, inconvenient, and invasive, and there are only several dozen apheresis centers in the United States. So the future of Lp(a)-lowering to treat refractory angina, calcific aortic stenosis, and other cardiovascular conditions where elevated Lp(a) is an important player may lie in pharmacotherapy with the PCSK9 inhibitors, Dr. Libby predicted.
He cited “very promising” data showing that evolocumab (Repatha) and alirocumab (Praluent) lower Lp(a) in dose-dependent fashion. Ongoing very large clinical trials with hard clinical endpoints should eventually provide key information regarding the cardiovascular benefits of lowering Lp(a).
“We may be entering an era where we may be able to offer our patients and families – because this is often a familial problem – a non-apheresis approach to controlling what we are learning is a very important causal risk factor for atherosclerosis,” Dr. Libby said.
Patrick M. Moriarty, MD, said in a video interview that he was very intrigued by the results, and “not personally surprised.” They should stimulate interest in cardiologists to start measuring Lp(a) in their patients like those in this study, in whom the disease severity doesn’t match up with the clinical risk factors, said Dr. Moriarty of the University of Kansas, Kansas City.
Dr. Khan’s study was funded by the UK National Institute for Health Research. She reported having no financial conflicts of interest.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ESC CONGRESS 2016
Key clinical point: Lipoprotein apheresis may provide a needed novel treatment for many patients with refractory angina.
Major finding: Myocardial perfusion reserve improved by 43% after apheresis, compared with no significant change after sham apheresis in patients with refractory angina and elevated lipoprotein(a).
Data source: A randomized, blinded, sham-controlled, crossover trial in 20 patients with refractory angina and elevated Lp(a) in the absence of high LDL cholesterol.
Disclosures: The UK National Institute for Health Research funded the study. The presenter reported having no financial conflicts of interest.