User login
CABG best for diabetes patients with CKD – or is it?
ROME – The use of coronary artery bypass graft surgery for revascularization in patients with multivessel CAD and comorbid diabetes plus chronic kidney disease was associated with a significantly lower risk of major cardiovascular and cerebrovascular events than was PCI with first-generation drug-eluting stents in a new secondary analysis from the landmark FREEDOM trial.
“The reason for this presentation is that even though chronic kidney disease is common in patients with diabetes, until now there has not been a large study of the efficacy and safety of coronary revascularization with drug-eluting stents versus CABG in this population in a randomized trial cohort,” explained Usman Baber, MD, who reported the results at the annual congress of the European Society of Cardiology.
FREEDOM (Future Revascularization Evaluation in Patients with Diabetes Mellitus: Optimal Management of Multivessel Disease) randomized 1,900 diabetic patients with multivessel CAD to PCI or CABG. As previously reported, CABG proved superior to PCI, with a significantly lower rate of the composite primary endpoint composed of all-cause mortality, MI, or stroke (N Engl J Med. 2012 Dec 20;367[25]:2375-84).
Dr. Baber presented a post hoc analysis of the 451 FREEDOM participants with baseline comorbid chronic kidney disease (CKD). Their mean SYNTAX score was 27, and their mean baseline estimated glomerular filtration rate was 44 mL/min per 1.73 m2, indicative of mild to moderate CKD.
“Only 28 patients in the FREEDOM trial had an estimated GFR below 30, therefore we can’t make any inferences about revascularization in that setting, which I think is a completely different population,” he noted.
The 5-year rate of major adverse cardiovascular and cerebrovascular events in patients with CKD was 26% in the CABG group, an absolute 9.4% less than the 35.6% rate in subjects randomized to PCI.
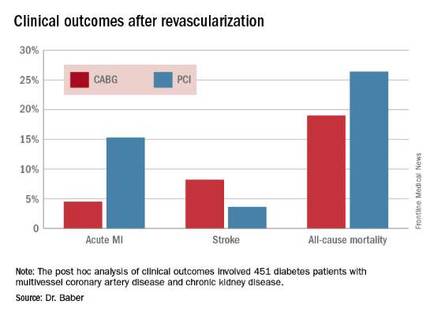
Roughly one-quarter of FREEDOM participants had CKD. They fared significantly worse than did those without CKD. The 5-year incidence of major adverse cardiovascular and cerebrovascular events was 30.8% in patients with CKD and 20.1% in patients without renal impairment. In a multivariate analysis adjusted for age, gender, hypertension, peripheral vascular disease, and other potential confounders, the risk of all-cause mortality was twofold higher in the CKD group. Their risk of cardiac death was increased 1.8-fold, and they were at 1.9-fold increased risk for stroke. Interestingly, however, the acute MI risk did not differ between patients with or without CKD, Dr. Baber observed.
Drilling deeper into the data, the cardiologist reported that CABG was associated with significantly lower rates of MI and a nonsignificant trend for fewer deaths, but with a significantly higher stroke rate than PCI.
One audience member rose to complain that this information won’t be helpful in counseling his diabetic patients with CKD and multivessel CAD because the choices look so grim: a higher risk of MI with percutaneous therapy, and a greater risk of stroke with surgery.
Dr. Baber replied by pointing out that the 10.8% absolute reduction in the risk of MI with CABG compared with PCI was more than twice as large as the absolute 4.6% increase in stroke risk with surgery.
“Most people would say that a heart attack is an inconvenience, and a stroke is a life-changing experience for them and their family,” said session cochair Kim A. Williams, MD, professor of medicine and chairman of cardiology at Rush University Medical Center in Chicago.
At that, Dr. Baber backtracked a bit, observing that since this was a post hoc analysis, the FREEDOM findings in patients with CKD must be viewed as hypothesis-generating rather than definitive. And, of course, contemporary second-generation drug-eluting stents have a better risk/benefit profile than do those used in FREEDOM.
“The number needed to treat/number needed to harm ratio for CABG and PCI probably ends up being roughly equal. The pertinence of an analysis like this is if you look at real-world registry-based data, you find a therapeutic nihilism that’s highly prevalent in CKD patients, where many patients who might benefit are not provided with revascularization therapy. It’s clear that we as clinicians – either because we don’t know there is a benefit or we are too concerned about potential harm – deprive patients of a treatment that might be beneficial. This analysis makes clinicians who might be concerned feel somewhat comforted that there is not unacceptable harm and that there is benefit,” Dr. Baber said.
Follow-up of FREEDOM participants continues and will be the subject of future reports, he added.
The FREEDOM trial was sponsored by the National Heart, Lung and Blood Institute. Dr. Baber reported having no financial conflicts of interest.
ROME – The use of coronary artery bypass graft surgery for revascularization in patients with multivessel CAD and comorbid diabetes plus chronic kidney disease was associated with a significantly lower risk of major cardiovascular and cerebrovascular events than was PCI with first-generation drug-eluting stents in a new secondary analysis from the landmark FREEDOM trial.
“The reason for this presentation is that even though chronic kidney disease is common in patients with diabetes, until now there has not been a large study of the efficacy and safety of coronary revascularization with drug-eluting stents versus CABG in this population in a randomized trial cohort,” explained Usman Baber, MD, who reported the results at the annual congress of the European Society of Cardiology.
FREEDOM (Future Revascularization Evaluation in Patients with Diabetes Mellitus: Optimal Management of Multivessel Disease) randomized 1,900 diabetic patients with multivessel CAD to PCI or CABG. As previously reported, CABG proved superior to PCI, with a significantly lower rate of the composite primary endpoint composed of all-cause mortality, MI, or stroke (N Engl J Med. 2012 Dec 20;367[25]:2375-84).
Dr. Baber presented a post hoc analysis of the 451 FREEDOM participants with baseline comorbid chronic kidney disease (CKD). Their mean SYNTAX score was 27, and their mean baseline estimated glomerular filtration rate was 44 mL/min per 1.73 m2, indicative of mild to moderate CKD.
“Only 28 patients in the FREEDOM trial had an estimated GFR below 30, therefore we can’t make any inferences about revascularization in that setting, which I think is a completely different population,” he noted.
The 5-year rate of major adverse cardiovascular and cerebrovascular events in patients with CKD was 26% in the CABG group, an absolute 9.4% less than the 35.6% rate in subjects randomized to PCI.

Roughly one-quarter of FREEDOM participants had CKD. They fared significantly worse than did those without CKD. The 5-year incidence of major adverse cardiovascular and cerebrovascular events was 30.8% in patients with CKD and 20.1% in patients without renal impairment. In a multivariate analysis adjusted for age, gender, hypertension, peripheral vascular disease, and other potential confounders, the risk of all-cause mortality was twofold higher in the CKD group. Their risk of cardiac death was increased 1.8-fold, and they were at 1.9-fold increased risk for stroke. Interestingly, however, the acute MI risk did not differ between patients with or without CKD, Dr. Baber observed.
Drilling deeper into the data, the cardiologist reported that CABG was associated with significantly lower rates of MI and a nonsignificant trend for fewer deaths, but with a significantly higher stroke rate than PCI.
One audience member rose to complain that this information won’t be helpful in counseling his diabetic patients with CKD and multivessel CAD because the choices look so grim: a higher risk of MI with percutaneous therapy, and a greater risk of stroke with surgery.
Dr. Baber replied by pointing out that the 10.8% absolute reduction in the risk of MI with CABG compared with PCI was more than twice as large as the absolute 4.6% increase in stroke risk with surgery.
“Most people would say that a heart attack is an inconvenience, and a stroke is a life-changing experience for them and their family,” said session cochair Kim A. Williams, MD, professor of medicine and chairman of cardiology at Rush University Medical Center in Chicago.
At that, Dr. Baber backtracked a bit, observing that since this was a post hoc analysis, the FREEDOM findings in patients with CKD must be viewed as hypothesis-generating rather than definitive. And, of course, contemporary second-generation drug-eluting stents have a better risk/benefit profile than do those used in FREEDOM.
“The number needed to treat/number needed to harm ratio for CABG and PCI probably ends up being roughly equal. The pertinence of an analysis like this is if you look at real-world registry-based data, you find a therapeutic nihilism that’s highly prevalent in CKD patients, where many patients who might benefit are not provided with revascularization therapy. It’s clear that we as clinicians – either because we don’t know there is a benefit or we are too concerned about potential harm – deprive patients of a treatment that might be beneficial. This analysis makes clinicians who might be concerned feel somewhat comforted that there is not unacceptable harm and that there is benefit,” Dr. Baber said.
Follow-up of FREEDOM participants continues and will be the subject of future reports, he added.
The FREEDOM trial was sponsored by the National Heart, Lung and Blood Institute. Dr. Baber reported having no financial conflicts of interest.
ROME – The use of coronary artery bypass graft surgery for revascularization in patients with multivessel CAD and comorbid diabetes plus chronic kidney disease was associated with a significantly lower risk of major cardiovascular and cerebrovascular events than was PCI with first-generation drug-eluting stents in a new secondary analysis from the landmark FREEDOM trial.
“The reason for this presentation is that even though chronic kidney disease is common in patients with diabetes, until now there has not been a large study of the efficacy and safety of coronary revascularization with drug-eluting stents versus CABG in this population in a randomized trial cohort,” explained Usman Baber, MD, who reported the results at the annual congress of the European Society of Cardiology.
FREEDOM (Future Revascularization Evaluation in Patients with Diabetes Mellitus: Optimal Management of Multivessel Disease) randomized 1,900 diabetic patients with multivessel CAD to PCI or CABG. As previously reported, CABG proved superior to PCI, with a significantly lower rate of the composite primary endpoint composed of all-cause mortality, MI, or stroke (N Engl J Med. 2012 Dec 20;367[25]:2375-84).
Dr. Baber presented a post hoc analysis of the 451 FREEDOM participants with baseline comorbid chronic kidney disease (CKD). Their mean SYNTAX score was 27, and their mean baseline estimated glomerular filtration rate was 44 mL/min per 1.73 m2, indicative of mild to moderate CKD.
“Only 28 patients in the FREEDOM trial had an estimated GFR below 30, therefore we can’t make any inferences about revascularization in that setting, which I think is a completely different population,” he noted.
The 5-year rate of major adverse cardiovascular and cerebrovascular events in patients with CKD was 26% in the CABG group, an absolute 9.4% less than the 35.6% rate in subjects randomized to PCI.

Roughly one-quarter of FREEDOM participants had CKD. They fared significantly worse than did those without CKD. The 5-year incidence of major adverse cardiovascular and cerebrovascular events was 30.8% in patients with CKD and 20.1% in patients without renal impairment. In a multivariate analysis adjusted for age, gender, hypertension, peripheral vascular disease, and other potential confounders, the risk of all-cause mortality was twofold higher in the CKD group. Their risk of cardiac death was increased 1.8-fold, and they were at 1.9-fold increased risk for stroke. Interestingly, however, the acute MI risk did not differ between patients with or without CKD, Dr. Baber observed.
Drilling deeper into the data, the cardiologist reported that CABG was associated with significantly lower rates of MI and a nonsignificant trend for fewer deaths, but with a significantly higher stroke rate than PCI.
One audience member rose to complain that this information won’t be helpful in counseling his diabetic patients with CKD and multivessel CAD because the choices look so grim: a higher risk of MI with percutaneous therapy, and a greater risk of stroke with surgery.
Dr. Baber replied by pointing out that the 10.8% absolute reduction in the risk of MI with CABG compared with PCI was more than twice as large as the absolute 4.6% increase in stroke risk with surgery.
“Most people would say that a heart attack is an inconvenience, and a stroke is a life-changing experience for them and their family,” said session cochair Kim A. Williams, MD, professor of medicine and chairman of cardiology at Rush University Medical Center in Chicago.
At that, Dr. Baber backtracked a bit, observing that since this was a post hoc analysis, the FREEDOM findings in patients with CKD must be viewed as hypothesis-generating rather than definitive. And, of course, contemporary second-generation drug-eluting stents have a better risk/benefit profile than do those used in FREEDOM.
“The number needed to treat/number needed to harm ratio for CABG and PCI probably ends up being roughly equal. The pertinence of an analysis like this is if you look at real-world registry-based data, you find a therapeutic nihilism that’s highly prevalent in CKD patients, where many patients who might benefit are not provided with revascularization therapy. It’s clear that we as clinicians – either because we don’t know there is a benefit or we are too concerned about potential harm – deprive patients of a treatment that might be beneficial. This analysis makes clinicians who might be concerned feel somewhat comforted that there is not unacceptable harm and that there is benefit,” Dr. Baber said.
Follow-up of FREEDOM participants continues and will be the subject of future reports, he added.
The FREEDOM trial was sponsored by the National Heart, Lung and Blood Institute. Dr. Baber reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2016
Key clinical point: Coronary artery bypass graft surgery resulted in fewer myocardial infarctions but more strokes than did percutaneous coronary intervention at 5 years of follow-up in diabetic patients with multivessel coronary artery disease and chronic kidney disease.
Major finding: The cumulative MI rates in patients randomized to CABG versus PCI were 4.5% and 15.3%, respectively, while the stroke rates were 8.2% versus 3.6%.
Data source: A post hoc analysis of clinical outcomes in 451 diabetic patients with multivessel CAD and chronic kidney disease who were randomized to CABG or PCI in the prospective multicenter FREEDOM trial.
Disclosures: The FREEDOM trial was sponsored by the National Heart, Lung, and Blood Institute. The presenter reported having no financial conflicts of interest.
Research yields fresh insights into ketamine for depression
VIENNA – The year 2016 has brought answers to two key questions regarding the off-label use of intravenous ketamine in patients with treatment-resistant depression: What’s the optimal dosing schedule? And what’s the likely mechanism of benefit?
Ketamine has generated enormous interest among psychiatrists and patients because the response is so dramatic, with marked improvement seen within hours in a much higher proportion of patients than respond to conventional antidepressants, which target the serotonergic system. But the benefits are not long lasting, and psychiatrists have wondered how often the treatment should be repeated. That question has been answered in a multicenter, double-blind U.S. randomized trial, Eduard Vieta, MD, PhD, noted at the annual congress of the European College of Neuropsychopharmacology.
Investigators randomized 67 patients with treatment-resistant depression to ketamine at 0.5 mg/kg of body weight at either two or three times per week, or to placebo. The mean reduction on the Montgomery-Åsberg Depression Rating Scale at day 15 was 18.4 points with twice-weekly therapy and similar at 17.7 with thrice-weekly therapy, both significantly better than with placebo (Am J Psychiatry. 2016 Aug 1;173[8]:816-26).
“It turns out that two and three times per week were equally effective, so obviously twice per week is enough,” said Dr. Vieta, professor of psychiatry and head of the bipolar disorders program at the University of Barcelona.
Ketamine’s approved indication is as an anesthetic agent. Its long-term safety as an antidepressant remains an open question. The drug has undesirable psychotropic side effects, including dissociation, but related compounds without those issues are speeding through the developmental pipeline. The Food and Drug Administration has granted Janssen Pharmaceuticals “fast track” and “breakthrough therapy” status for intranasal esketamine, the S(+) enantiomer of ketamine, which is now in phase III clinical trials for treatment-resistant depression as well as for depression with suicidal thoughts. The FDA reserves these designations for potential therapies addressing a major unmet need. Allergan has received the same designations from the FDA for its drug rapastinel, which also is now in phase III clinical trials.
“Ketamine is clearly not something to use as first-line therapy. I think there is a problem in certain places: I know in the U.S. there are now plenty of ketamine clinics administering the drug to first comers. That doesn’t make sense to me. But ketamine does open an important new avenue,” he said.
Dr. Vieta asserted that the future of new drug development for mood disorders lies in the glutamatergic system. However, a recent study by investigators at the National Institute of Mental Health – who pioneered the use of ketamine as an antidepressant – and colleagues at the University of Maryland, Baltimore, casts doubt upon the conventional wisdom that ketamine’s mechanism of benefit as an antidepressant involves N-methyl-d-aspartate receptor (NMDA) antagonism.
Instead, they reported, the antidepressant effect is actually exerted by a ketamine metabolite known as HNK, or (2S,6S;2R,6R)-hydroxynorketamine. And HNK’s antidepressant effect is not related to NMDA receptors, but is instead tied to activation of alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors. And in mice, at least, HNK lacks the unwelcome psychotomimetic side effects of ketamine (Nature. 2016 May 4;533[7604]:481-6).
“This is a very nice paper and very important. This opens up a new avenue in drug development, looking at agents that act on AMPA receptors to provide rapid relief of depressive symptoms in unipolar depression but probably also in bipolar depression,” said Dr. Vieta.
He reported receiving research grants from numerous pharmaceutical companies having an interest in treatments for mood disorders.
VIENNA – The year 2016 has brought answers to two key questions regarding the off-label use of intravenous ketamine in patients with treatment-resistant depression: What’s the optimal dosing schedule? And what’s the likely mechanism of benefit?
Ketamine has generated enormous interest among psychiatrists and patients because the response is so dramatic, with marked improvement seen within hours in a much higher proportion of patients than respond to conventional antidepressants, which target the serotonergic system. But the benefits are not long lasting, and psychiatrists have wondered how often the treatment should be repeated. That question has been answered in a multicenter, double-blind U.S. randomized trial, Eduard Vieta, MD, PhD, noted at the annual congress of the European College of Neuropsychopharmacology.
Investigators randomized 67 patients with treatment-resistant depression to ketamine at 0.5 mg/kg of body weight at either two or three times per week, or to placebo. The mean reduction on the Montgomery-Åsberg Depression Rating Scale at day 15 was 18.4 points with twice-weekly therapy and similar at 17.7 with thrice-weekly therapy, both significantly better than with placebo (Am J Psychiatry. 2016 Aug 1;173[8]:816-26).
“It turns out that two and three times per week were equally effective, so obviously twice per week is enough,” said Dr. Vieta, professor of psychiatry and head of the bipolar disorders program at the University of Barcelona.
Ketamine’s approved indication is as an anesthetic agent. Its long-term safety as an antidepressant remains an open question. The drug has undesirable psychotropic side effects, including dissociation, but related compounds without those issues are speeding through the developmental pipeline. The Food and Drug Administration has granted Janssen Pharmaceuticals “fast track” and “breakthrough therapy” status for intranasal esketamine, the S(+) enantiomer of ketamine, which is now in phase III clinical trials for treatment-resistant depression as well as for depression with suicidal thoughts. The FDA reserves these designations for potential therapies addressing a major unmet need. Allergan has received the same designations from the FDA for its drug rapastinel, which also is now in phase III clinical trials.
“Ketamine is clearly not something to use as first-line therapy. I think there is a problem in certain places: I know in the U.S. there are now plenty of ketamine clinics administering the drug to first comers. That doesn’t make sense to me. But ketamine does open an important new avenue,” he said.
Dr. Vieta asserted that the future of new drug development for mood disorders lies in the glutamatergic system. However, a recent study by investigators at the National Institute of Mental Health – who pioneered the use of ketamine as an antidepressant – and colleagues at the University of Maryland, Baltimore, casts doubt upon the conventional wisdom that ketamine’s mechanism of benefit as an antidepressant involves N-methyl-d-aspartate receptor (NMDA) antagonism.
Instead, they reported, the antidepressant effect is actually exerted by a ketamine metabolite known as HNK, or (2S,6S;2R,6R)-hydroxynorketamine. And HNK’s antidepressant effect is not related to NMDA receptors, but is instead tied to activation of alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors. And in mice, at least, HNK lacks the unwelcome psychotomimetic side effects of ketamine (Nature. 2016 May 4;533[7604]:481-6).
“This is a very nice paper and very important. This opens up a new avenue in drug development, looking at agents that act on AMPA receptors to provide rapid relief of depressive symptoms in unipolar depression but probably also in bipolar depression,” said Dr. Vieta.
He reported receiving research grants from numerous pharmaceutical companies having an interest in treatments for mood disorders.
VIENNA – The year 2016 has brought answers to two key questions regarding the off-label use of intravenous ketamine in patients with treatment-resistant depression: What’s the optimal dosing schedule? And what’s the likely mechanism of benefit?
Ketamine has generated enormous interest among psychiatrists and patients because the response is so dramatic, with marked improvement seen within hours in a much higher proportion of patients than respond to conventional antidepressants, which target the serotonergic system. But the benefits are not long lasting, and psychiatrists have wondered how often the treatment should be repeated. That question has been answered in a multicenter, double-blind U.S. randomized trial, Eduard Vieta, MD, PhD, noted at the annual congress of the European College of Neuropsychopharmacology.
Investigators randomized 67 patients with treatment-resistant depression to ketamine at 0.5 mg/kg of body weight at either two or three times per week, or to placebo. The mean reduction on the Montgomery-Åsberg Depression Rating Scale at day 15 was 18.4 points with twice-weekly therapy and similar at 17.7 with thrice-weekly therapy, both significantly better than with placebo (Am J Psychiatry. 2016 Aug 1;173[8]:816-26).
“It turns out that two and three times per week were equally effective, so obviously twice per week is enough,” said Dr. Vieta, professor of psychiatry and head of the bipolar disorders program at the University of Barcelona.
Ketamine’s approved indication is as an anesthetic agent. Its long-term safety as an antidepressant remains an open question. The drug has undesirable psychotropic side effects, including dissociation, but related compounds without those issues are speeding through the developmental pipeline. The Food and Drug Administration has granted Janssen Pharmaceuticals “fast track” and “breakthrough therapy” status for intranasal esketamine, the S(+) enantiomer of ketamine, which is now in phase III clinical trials for treatment-resistant depression as well as for depression with suicidal thoughts. The FDA reserves these designations for potential therapies addressing a major unmet need. Allergan has received the same designations from the FDA for its drug rapastinel, which also is now in phase III clinical trials.
“Ketamine is clearly not something to use as first-line therapy. I think there is a problem in certain places: I know in the U.S. there are now plenty of ketamine clinics administering the drug to first comers. That doesn’t make sense to me. But ketamine does open an important new avenue,” he said.
Dr. Vieta asserted that the future of new drug development for mood disorders lies in the glutamatergic system. However, a recent study by investigators at the National Institute of Mental Health – who pioneered the use of ketamine as an antidepressant – and colleagues at the University of Maryland, Baltimore, casts doubt upon the conventional wisdom that ketamine’s mechanism of benefit as an antidepressant involves N-methyl-d-aspartate receptor (NMDA) antagonism.
Instead, they reported, the antidepressant effect is actually exerted by a ketamine metabolite known as HNK, or (2S,6S;2R,6R)-hydroxynorketamine. And HNK’s antidepressant effect is not related to NMDA receptors, but is instead tied to activation of alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) receptors. And in mice, at least, HNK lacks the unwelcome psychotomimetic side effects of ketamine (Nature. 2016 May 4;533[7604]:481-6).
“This is a very nice paper and very important. This opens up a new avenue in drug development, looking at agents that act on AMPA receptors to provide rapid relief of depressive symptoms in unipolar depression but probably also in bipolar depression,” said Dr. Vieta.
He reported receiving research grants from numerous pharmaceutical companies having an interest in treatments for mood disorders.
EXPERT ANALYSIS FROM THE ECNP CONGRESS
Male libido problems successfully treated with bright light
VIENNA – Daily exposure to high-intensity light early in the morning resulted in significantly improved sexual satisfaction scores and a boost in testosterone levels in men with reduced libido or erection difficulty in a randomized, placebo-controlled clinical trial, Andrea Fagiolini, MD., said at the annual meeting of the American Thyroid Association.
“We found a very strong difference in sexual satisfaction and also in blood testosterone levels with intense light therapy. Patients seemed to respond to it really well, and our placebo group didn’t respond at all,” according to Dr. Fagiolini, professor and chairman of the department of psychiatry at the University of Siena (Italy).
“It’s still early to make a general recommendation. This was a small study. We need a larger trial with more patients. But if these results are confirmed, it would be very helpful because as of now we don’t have many tools to treat sexual dysfunction. So if light therapy works, I think it may be a better option than medications like Viagra [sildenafil] or similar drugs, which have side effects,” he said in an interview.
The trial included 38 men seen at the university urology clinic with a diagnosis of primary hypoactive sexual desire or sexual arousal disorder. Half were randomized to daily use of a white fluorescent light box for 30 minutes in the morning, preferably between 7 and 8 a.m. The light box, equipped with a UV filter, was the same as that used in treating seasonal affective disorder. Light therapy for seasonal affective disorder is an established therapy in the psychiatric world, one that has proved as effective as antidepressant therapy for that condition. It is “absolutely safe and tolerable,” except in patients with eye conditions or on photosensitizing medications, the psychiatrist noted.
The control group was on the same treatment schedule, but with light boxes that had been modified through the use of a neutral-density gel filter to provide a dose of just 100 lux instead of the 10,000 lux at a distance of 1 m from the light source to the cornea generated by the active therapy light box.
The study endpoints were change from baseline through 2 weeks of therapy on a self-administered rating of sexual satisfaction on a scale of 1-10, as well as testosterone levels.
From a baseline mean self-rated sexual satisfaction score of about 2, the group exposed to daily bright light for 2 weeks had a more than threefold increase to a score of 6.3, while the controls showed no significant change.
In addition, testosterone levels in the bright light group increased from 2.1 ng/mL to 3.6 ng/mL after 2 weeks, while remaining unchanged in controls.
“Our idea for this study came from the observation that sexual satisfaction is known to increase during the spring and summer, compared to fall and winter. We thought that may have something to do with the natural daylight,” Dr. Fagiolini said.
The most likely mechanism of benefit springs from the established finding that bright light stimulates the pituitary gland to produce leutinizing hormone. This results in increased levels of testosterone and inhibition of melatonin production by the pineal gland. Decreased plasma melatonin is thought to lead to reduced serum prolactin – and high prolactin has been linked to sexual dysfunction, he explained.
The trial was conducted in the fall and winter months to avoid confounding by more natural daylight during the other seasons. But if the study results are confirmed, this therapy might well be used year-round, the psychiatrist said.
He reported having no financial conflicts of interest regarding the study, which was conducted without external funding.
VIENNA – Daily exposure to high-intensity light early in the morning resulted in significantly improved sexual satisfaction scores and a boost in testosterone levels in men with reduced libido or erection difficulty in a randomized, placebo-controlled clinical trial, Andrea Fagiolini, MD., said at the annual meeting of the American Thyroid Association.
“We found a very strong difference in sexual satisfaction and also in blood testosterone levels with intense light therapy. Patients seemed to respond to it really well, and our placebo group didn’t respond at all,” according to Dr. Fagiolini, professor and chairman of the department of psychiatry at the University of Siena (Italy).
“It’s still early to make a general recommendation. This was a small study. We need a larger trial with more patients. But if these results are confirmed, it would be very helpful because as of now we don’t have many tools to treat sexual dysfunction. So if light therapy works, I think it may be a better option than medications like Viagra [sildenafil] or similar drugs, which have side effects,” he said in an interview.
The trial included 38 men seen at the university urology clinic with a diagnosis of primary hypoactive sexual desire or sexual arousal disorder. Half were randomized to daily use of a white fluorescent light box for 30 minutes in the morning, preferably between 7 and 8 a.m. The light box, equipped with a UV filter, was the same as that used in treating seasonal affective disorder. Light therapy for seasonal affective disorder is an established therapy in the psychiatric world, one that has proved as effective as antidepressant therapy for that condition. It is “absolutely safe and tolerable,” except in patients with eye conditions or on photosensitizing medications, the psychiatrist noted.
The control group was on the same treatment schedule, but with light boxes that had been modified through the use of a neutral-density gel filter to provide a dose of just 100 lux instead of the 10,000 lux at a distance of 1 m from the light source to the cornea generated by the active therapy light box.
The study endpoints were change from baseline through 2 weeks of therapy on a self-administered rating of sexual satisfaction on a scale of 1-10, as well as testosterone levels.
From a baseline mean self-rated sexual satisfaction score of about 2, the group exposed to daily bright light for 2 weeks had a more than threefold increase to a score of 6.3, while the controls showed no significant change.
In addition, testosterone levels in the bright light group increased from 2.1 ng/mL to 3.6 ng/mL after 2 weeks, while remaining unchanged in controls.
“Our idea for this study came from the observation that sexual satisfaction is known to increase during the spring and summer, compared to fall and winter. We thought that may have something to do with the natural daylight,” Dr. Fagiolini said.
The most likely mechanism of benefit springs from the established finding that bright light stimulates the pituitary gland to produce leutinizing hormone. This results in increased levels of testosterone and inhibition of melatonin production by the pineal gland. Decreased plasma melatonin is thought to lead to reduced serum prolactin – and high prolactin has been linked to sexual dysfunction, he explained.
The trial was conducted in the fall and winter months to avoid confounding by more natural daylight during the other seasons. But if the study results are confirmed, this therapy might well be used year-round, the psychiatrist said.
He reported having no financial conflicts of interest regarding the study, which was conducted without external funding.
VIENNA – Daily exposure to high-intensity light early in the morning resulted in significantly improved sexual satisfaction scores and a boost in testosterone levels in men with reduced libido or erection difficulty in a randomized, placebo-controlled clinical trial, Andrea Fagiolini, MD., said at the annual meeting of the American Thyroid Association.
“We found a very strong difference in sexual satisfaction and also in blood testosterone levels with intense light therapy. Patients seemed to respond to it really well, and our placebo group didn’t respond at all,” according to Dr. Fagiolini, professor and chairman of the department of psychiatry at the University of Siena (Italy).
“It’s still early to make a general recommendation. This was a small study. We need a larger trial with more patients. But if these results are confirmed, it would be very helpful because as of now we don’t have many tools to treat sexual dysfunction. So if light therapy works, I think it may be a better option than medications like Viagra [sildenafil] or similar drugs, which have side effects,” he said in an interview.
The trial included 38 men seen at the university urology clinic with a diagnosis of primary hypoactive sexual desire or sexual arousal disorder. Half were randomized to daily use of a white fluorescent light box for 30 minutes in the morning, preferably between 7 and 8 a.m. The light box, equipped with a UV filter, was the same as that used in treating seasonal affective disorder. Light therapy for seasonal affective disorder is an established therapy in the psychiatric world, one that has proved as effective as antidepressant therapy for that condition. It is “absolutely safe and tolerable,” except in patients with eye conditions or on photosensitizing medications, the psychiatrist noted.
The control group was on the same treatment schedule, but with light boxes that had been modified through the use of a neutral-density gel filter to provide a dose of just 100 lux instead of the 10,000 lux at a distance of 1 m from the light source to the cornea generated by the active therapy light box.
The study endpoints were change from baseline through 2 weeks of therapy on a self-administered rating of sexual satisfaction on a scale of 1-10, as well as testosterone levels.
From a baseline mean self-rated sexual satisfaction score of about 2, the group exposed to daily bright light for 2 weeks had a more than threefold increase to a score of 6.3, while the controls showed no significant change.
In addition, testosterone levels in the bright light group increased from 2.1 ng/mL to 3.6 ng/mL after 2 weeks, while remaining unchanged in controls.
“Our idea for this study came from the observation that sexual satisfaction is known to increase during the spring and summer, compared to fall and winter. We thought that may have something to do with the natural daylight,” Dr. Fagiolini said.
The most likely mechanism of benefit springs from the established finding that bright light stimulates the pituitary gland to produce leutinizing hormone. This results in increased levels of testosterone and inhibition of melatonin production by the pineal gland. Decreased plasma melatonin is thought to lead to reduced serum prolactin – and high prolactin has been linked to sexual dysfunction, he explained.
The trial was conducted in the fall and winter months to avoid confounding by more natural daylight during the other seasons. But if the study results are confirmed, this therapy might well be used year-round, the psychiatrist said.
He reported having no financial conflicts of interest regarding the study, which was conducted without external funding.
AT THE ECNP CONGRESS
Key clinical point: Daily exposure to intense white light, an established treatment for seasonal affective disorder, also may improve male sexual dysfunction.
Major finding: After 2 weeks of daily bright light therapy, men with low libido or erection difficulty showed an improvement in self-rated sexual satisfaction from 2.1 to 6.3 on a 1-10 scale.
Data source: A randomized trial that included 38 men diagnosed with hypoactive sexual desire disorder or sexual arousal disorder who received 2 weeks of high-intensity light therapy for 30 minutes per morning or a sham placebo involving exposure to far less intense light.
Disclosures: The presenter reported having no financial conflicts of interest regarding the study, which was conducted without external funding.
Psychiatric risks quantified in siblings of mental disorder patients
VIENNA – The brothers and sisters of patients hospitalized for schizophrenia, bipolar disorder, or unipolar depression are themselves at strikingly high risk of subsequently developing not only the same disorder as their sibling, but other forms of major mental illness as well, Mark Weiser, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
He presented the results of the first comprehensive national population-based study to examine in this fashion the extent to which heritability contributes to schizophrenia and affective disorders. This nested case-control study included all siblings of 6,111 Israeli patients hospitalized for schizophrenia, schizoaffective disorder, bipolar disorder, or unipolar depression. The siblings’ rates of and reasons for subsequent psychiatric hospitalization were compared with those of 74,988 age- and gender-matched Israeli controls. All admission and discharge diagnoses were made by board-certified psychiatrists.
Siblings of individuals with schizophrenia were at 9.4-fold increased risk of subsequent hospitalization for schizophrenia, 8.5-fold relative risk for schizoaffective disorder, and 7.7-fold increased risk for bipolar disorder, compared with controls.
Moreover, siblings of patients with bipolar disorder were not only at 8.4-fold increased risk of subsequent hospitalization for that disease, they also were at 4.2-fold greater risk than controls for schizophrenia and 7.6-fold increased risk for hospitalization for other psychiatric disorders, a grab bag category that included anxiety disorders, dissociative disorder, post-traumatic stress disorder, eating disorders, pervasive developmental disorders, and personality disorders, according to Dr. Weiser, professor of psychiatry at Tel Aviv University.
Siblings of patients hospitalized for unipolar depression were at 6.2-fold relative risk of subsequent hospitalization for schizophrenia and 9.7-fold increased risk of hospitalization of other psychiatric disorders. “The bottom line of our study is it’s not a one gene/one disorder model. There’s not a gene for schizophrenia and a different gene for bipolar disorder. There are probably a bunch of different genes that increase the risk for schizophrenia but also increase risk for bipolar disorder, and the other way around,” the psychiatrist explained in an interview.
“Clinically it’s well known from the literature that if I have schizophrenia, there’s an increased chance that my brother will have it as well, so when my brother comes in having trouble, you obviously suggest that he might be developing schizophrenia. What these data imply is that if the brother of a schizophrenia patient comes in seeking help, it might not be schizophrenia, because he’s also at increased risk for bipolar disorder. So your index of suspicion should be much broader, not only for the one specific illness but for the whole idea of psychopathology in general. It’s a challenge. It demands for clinicians to be more broad-minded and to understand that these genes we’re looking for in large studies are not specific for one particular illness,” Dr. Weiser said.
This study was made possible because Israel, like Denmark, maintains multiple comprehensive national registries in which health care researchers are able to tap into and connect.
“A study like this can’t be done in the United States,” he said. “No how, no way.”
Dr. Weiser reported having no financial conflicts of interest regarding this study, which was conducted without external funding.
VIENNA – The brothers and sisters of patients hospitalized for schizophrenia, bipolar disorder, or unipolar depression are themselves at strikingly high risk of subsequently developing not only the same disorder as their sibling, but other forms of major mental illness as well, Mark Weiser, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
He presented the results of the first comprehensive national population-based study to examine in this fashion the extent to which heritability contributes to schizophrenia and affective disorders. This nested case-control study included all siblings of 6,111 Israeli patients hospitalized for schizophrenia, schizoaffective disorder, bipolar disorder, or unipolar depression. The siblings’ rates of and reasons for subsequent psychiatric hospitalization were compared with those of 74,988 age- and gender-matched Israeli controls. All admission and discharge diagnoses were made by board-certified psychiatrists.
Siblings of individuals with schizophrenia were at 9.4-fold increased risk of subsequent hospitalization for schizophrenia, 8.5-fold relative risk for schizoaffective disorder, and 7.7-fold increased risk for bipolar disorder, compared with controls.
Moreover, siblings of patients with bipolar disorder were not only at 8.4-fold increased risk of subsequent hospitalization for that disease, they also were at 4.2-fold greater risk than controls for schizophrenia and 7.6-fold increased risk for hospitalization for other psychiatric disorders, a grab bag category that included anxiety disorders, dissociative disorder, post-traumatic stress disorder, eating disorders, pervasive developmental disorders, and personality disorders, according to Dr. Weiser, professor of psychiatry at Tel Aviv University.
Siblings of patients hospitalized for unipolar depression were at 6.2-fold relative risk of subsequent hospitalization for schizophrenia and 9.7-fold increased risk of hospitalization of other psychiatric disorders. “The bottom line of our study is it’s not a one gene/one disorder model. There’s not a gene for schizophrenia and a different gene for bipolar disorder. There are probably a bunch of different genes that increase the risk for schizophrenia but also increase risk for bipolar disorder, and the other way around,” the psychiatrist explained in an interview.
“Clinically it’s well known from the literature that if I have schizophrenia, there’s an increased chance that my brother will have it as well, so when my brother comes in having trouble, you obviously suggest that he might be developing schizophrenia. What these data imply is that if the brother of a schizophrenia patient comes in seeking help, it might not be schizophrenia, because he’s also at increased risk for bipolar disorder. So your index of suspicion should be much broader, not only for the one specific illness but for the whole idea of psychopathology in general. It’s a challenge. It demands for clinicians to be more broad-minded and to understand that these genes we’re looking for in large studies are not specific for one particular illness,” Dr. Weiser said.
This study was made possible because Israel, like Denmark, maintains multiple comprehensive national registries in which health care researchers are able to tap into and connect.
“A study like this can’t be done in the United States,” he said. “No how, no way.”
Dr. Weiser reported having no financial conflicts of interest regarding this study, which was conducted without external funding.
VIENNA – The brothers and sisters of patients hospitalized for schizophrenia, bipolar disorder, or unipolar depression are themselves at strikingly high risk of subsequently developing not only the same disorder as their sibling, but other forms of major mental illness as well, Mark Weiser, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
He presented the results of the first comprehensive national population-based study to examine in this fashion the extent to which heritability contributes to schizophrenia and affective disorders. This nested case-control study included all siblings of 6,111 Israeli patients hospitalized for schizophrenia, schizoaffective disorder, bipolar disorder, or unipolar depression. The siblings’ rates of and reasons for subsequent psychiatric hospitalization were compared with those of 74,988 age- and gender-matched Israeli controls. All admission and discharge diagnoses were made by board-certified psychiatrists.
Siblings of individuals with schizophrenia were at 9.4-fold increased risk of subsequent hospitalization for schizophrenia, 8.5-fold relative risk for schizoaffective disorder, and 7.7-fold increased risk for bipolar disorder, compared with controls.
Moreover, siblings of patients with bipolar disorder were not only at 8.4-fold increased risk of subsequent hospitalization for that disease, they also were at 4.2-fold greater risk than controls for schizophrenia and 7.6-fold increased risk for hospitalization for other psychiatric disorders, a grab bag category that included anxiety disorders, dissociative disorder, post-traumatic stress disorder, eating disorders, pervasive developmental disorders, and personality disorders, according to Dr. Weiser, professor of psychiatry at Tel Aviv University.
Siblings of patients hospitalized for unipolar depression were at 6.2-fold relative risk of subsequent hospitalization for schizophrenia and 9.7-fold increased risk of hospitalization of other psychiatric disorders. “The bottom line of our study is it’s not a one gene/one disorder model. There’s not a gene for schizophrenia and a different gene for bipolar disorder. There are probably a bunch of different genes that increase the risk for schizophrenia but also increase risk for bipolar disorder, and the other way around,” the psychiatrist explained in an interview.
“Clinically it’s well known from the literature that if I have schizophrenia, there’s an increased chance that my brother will have it as well, so when my brother comes in having trouble, you obviously suggest that he might be developing schizophrenia. What these data imply is that if the brother of a schizophrenia patient comes in seeking help, it might not be schizophrenia, because he’s also at increased risk for bipolar disorder. So your index of suspicion should be much broader, not only for the one specific illness but for the whole idea of psychopathology in general. It’s a challenge. It demands for clinicians to be more broad-minded and to understand that these genes we’re looking for in large studies are not specific for one particular illness,” Dr. Weiser said.
This study was made possible because Israel, like Denmark, maintains multiple comprehensive national registries in which health care researchers are able to tap into and connect.
“A study like this can’t be done in the United States,” he said. “No how, no way.”
Dr. Weiser reported having no financial conflicts of interest regarding this study, which was conducted without external funding.
AT THE ECNP CONGRESS
Key clinical point: Siblings of patients with schizophrenia or bipolar disorder are at sharply increased risk of subsequent hospitalization for a range of psychiatric disorders, not just what their sibling has.
Major finding: Siblings of schizophrenia patients were not only at 9.4-fold increased risk of subsequent hospitalization for schizophrenia in a national study, but also at 7.7-fold greater risk for bipolar disorder.
Data source: This nested case-control study compared psychiatric hospitalization rates for all siblings of 6,111 Israeli patients with schizophrenia, bipolar disorder, or unipolar depression to nearly 75,000 matched controls.
Disclosures: The study was conducted without external funding. The presenter reported having no financial conflicts of interest.
Prospects brighten for an HIV vaccine
DURBAN, SOUTH AFRICA – A new optimism regarding the possibility of creating a safe and effective HIV preventive vaccine was very much in evidence at the 21st International AIDS Conference.
“The HIV vaccine field is open for business,” an exuberant Larry Corey, MD, declared in a plenary address highlighting recent major progress in HIV vaccine development.
Three extremely important HIV vaccine efficacy clinical trials testing diverse promising strategies are now either in progress or soon to start, noted Dr. Corey, professor of laboratory medicine and medicine at the University of Washington and emeritus director of the Fred Hutchinson Cancer Research Center in Seattle.
“We are finally moving the needle forward with human efficacy trials that are commensurate with the need for developing an HIV vaccine,” Dr. Corey said.
He emphasized that HIV is “still the world’s most pressing global health issue,” with more than 45,000 new infections occurring annually in the United States and more than 2 million annually worldwide. And while numerous nonvaccine prevention methods have been developed, they share a major limitation: Their extended effectiveness requires continuous adherence.
“With asymptomatic acquisition, prolonged subclinical infection, and sexual transmission, getting to an AIDS-free generation will require a vaccine,” Dr. Corey predicted.
After years of discouragingly negative HIV vaccine studies, researchers finally turned a corner in 2009 with the reported results of the U.S. Military HIV Research Program–led RV144 trial, commonly known as the Thai Trial, Anthony S. Fauci, MD, recalled in an interview.
The trial, which randomized more than 16,000 young adult Thais, showed a modest 31% efficacy at 3.5 years, but a more substantial and encouraging 60% efficacy at 12 months (N Engl J Med. 2009 Dec 3;361[23]:2209-20). More importantly, the Thai trial opened up a whole new avenue to HIV vaccine development.
“We thought the Thai vaccine would induce neutralizing antibodies, but it didn’t. Instead, it induced nonneutralizing antibodies against a component of the V1V2 loop region of the HIV envelope, which was associated with protection. So the good news about that study was that even though that vaccine wasn’t efficacious enough to make it a usable vaccine, it gave us something we could improve upon by using the same platform and enhancing the response to provide greater depth, breadth, and durability,” explained Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases (NIAID).
The improved vaccine consists of a canarypox-based vaccine called ALVAC-HIV and a bivalent gp120 protein subunit vaccine with MF59, a different adjuvant from that used in the Thai trial, in an effort to achieve a more robust immune response. Also, the four-injection series studied in the Thai trial is now bolstered by a booster injection at the 12-month mark. This vaccine has been altered to be specific to HIV clade C, the predominant HIV subtype in southern Africa, where the bulk of new HIV infections occur.
At the AIDS 2016 conference, Linda-Gail Bekker, MD, presented the primary immunogenicity results from the HIV Vaccine Trials Network (HVTN) 100 trial, a phase 1/2, double-blind, placebo-controlled study of the improved version of the Thai trial vaccine, known as the Clade C ALVAC-(vCP2438) and bivalent subtype C gp120/MF59 vaccine, in 252 HIV-uninfected South African adults.
The ALVAC/protein vaccine achieved cellular and humoral immune responses that exceeded all four predetermined criteria as correlates of protection. As a result, a pivotal phase III, randomized, double-blind, placebo-controlled vaccine efficacy trial known as HVTN 702 got the green light to begin in November 2016 in 5,400 HIV-negative adults in South Africa. Participants will be assessed at 24 and 36 months of follow-up, announced Dr. Bekker, cochair of HVTN 702, International AIDS Society president-elect, and deputy director of the Desmond Tutu HIV Center in Cape Town, South Africa.
As a principal investigator in the NIAID-supported HIV Vaccine Trials Network, Dr. Corey has been deeply involved in the development of this vaccine. He also is chair of the ongoing HVTN 703 and 704 phase IIb trials, testing an entirely different vaccine approach. The hypothesis being tested in HVTN 703 and 704 is that a passively infused monoclonal antibody can protect against HIV infection in 2,400 men who have sex with men and transgender men in North and South America, as well as in 1,500 women in sub-Saharan Africa. Both studies began in spring 2016.
“Every card-carrying virologist feels this should work,” according to Dr. Corey.
The rationale for having two study populations is that investigators suspect the effects of the antibody may vary depending upon whether the route of HIV acquisition is rectal or vaginal, he explained.
The monoclonal antibody contains VRC01, which effectively blocks viral binding to CD4 cells. Study participants will receive an intravenous infusion of VRC01 at 10 or 30 mg/kg or placebo every 2 months. If the results are positive and a second-generation product and delivery system can be developed, antibody-mediated prevention could also have a major potential role in interrupting maternal to child transmission of HIV resulting from intrapartum exposure or breastfeeding.
Dr. Corey also highlighted a third strategy of HIV vaccine development, one at an earlier stage. Investigators at Johnson & Johnson, in collaboration with the NIAID, HVTN, and other partners, are pursuing a multi-clade approach, one designed to protect against all clades of HIV found around the world. This strategy entails first giving an adenovirus serotype 26–vectored vaccine to prime the immune system, following up with administration of several boosters containing mosaic inserts to increase the response. This vaccine is in phase I studies with no results yet.
Dr. Fauci is not sure which if any of these three approaches will yield a safe and effective vaccine for HIV prevention.
“It’s important to realize that this is a very difficult scientific challenge,” he said. “The body does not readily make an adequate immune response against HIV, unlike virtually any other viral infection. Even the serious ones that cause a degree of morbidity and mortality – smallpox, measles, rubella, polio – ultimately the body does make a good immune response and allows us to clear the virus and leaves us with protection against subsequent exposure to the same virus. We don’t have that advantage with HIV. So it’s going to be difficult to get a safe and effective HIV vaccine, but I think the scientific challenge is worth going after and there’s a reasonable chance we might get there.”
Dr. Corey, Dr. Fauci, and Dr. Bekker reported having no financial conflicts of interest.
DURBAN, SOUTH AFRICA – A new optimism regarding the possibility of creating a safe and effective HIV preventive vaccine was very much in evidence at the 21st International AIDS Conference.
“The HIV vaccine field is open for business,” an exuberant Larry Corey, MD, declared in a plenary address highlighting recent major progress in HIV vaccine development.
Three extremely important HIV vaccine efficacy clinical trials testing diverse promising strategies are now either in progress or soon to start, noted Dr. Corey, professor of laboratory medicine and medicine at the University of Washington and emeritus director of the Fred Hutchinson Cancer Research Center in Seattle.
“We are finally moving the needle forward with human efficacy trials that are commensurate with the need for developing an HIV vaccine,” Dr. Corey said.
He emphasized that HIV is “still the world’s most pressing global health issue,” with more than 45,000 new infections occurring annually in the United States and more than 2 million annually worldwide. And while numerous nonvaccine prevention methods have been developed, they share a major limitation: Their extended effectiveness requires continuous adherence.
“With asymptomatic acquisition, prolonged subclinical infection, and sexual transmission, getting to an AIDS-free generation will require a vaccine,” Dr. Corey predicted.
After years of discouragingly negative HIV vaccine studies, researchers finally turned a corner in 2009 with the reported results of the U.S. Military HIV Research Program–led RV144 trial, commonly known as the Thai Trial, Anthony S. Fauci, MD, recalled in an interview.
The trial, which randomized more than 16,000 young adult Thais, showed a modest 31% efficacy at 3.5 years, but a more substantial and encouraging 60% efficacy at 12 months (N Engl J Med. 2009 Dec 3;361[23]:2209-20). More importantly, the Thai trial opened up a whole new avenue to HIV vaccine development.
“We thought the Thai vaccine would induce neutralizing antibodies, but it didn’t. Instead, it induced nonneutralizing antibodies against a component of the V1V2 loop region of the HIV envelope, which was associated with protection. So the good news about that study was that even though that vaccine wasn’t efficacious enough to make it a usable vaccine, it gave us something we could improve upon by using the same platform and enhancing the response to provide greater depth, breadth, and durability,” explained Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases (NIAID).
The improved vaccine consists of a canarypox-based vaccine called ALVAC-HIV and a bivalent gp120 protein subunit vaccine with MF59, a different adjuvant from that used in the Thai trial, in an effort to achieve a more robust immune response. Also, the four-injection series studied in the Thai trial is now bolstered by a booster injection at the 12-month mark. This vaccine has been altered to be specific to HIV clade C, the predominant HIV subtype in southern Africa, where the bulk of new HIV infections occur.
At the AIDS 2016 conference, Linda-Gail Bekker, MD, presented the primary immunogenicity results from the HIV Vaccine Trials Network (HVTN) 100 trial, a phase 1/2, double-blind, placebo-controlled study of the improved version of the Thai trial vaccine, known as the Clade C ALVAC-(vCP2438) and bivalent subtype C gp120/MF59 vaccine, in 252 HIV-uninfected South African adults.
The ALVAC/protein vaccine achieved cellular and humoral immune responses that exceeded all four predetermined criteria as correlates of protection. As a result, a pivotal phase III, randomized, double-blind, placebo-controlled vaccine efficacy trial known as HVTN 702 got the green light to begin in November 2016 in 5,400 HIV-negative adults in South Africa. Participants will be assessed at 24 and 36 months of follow-up, announced Dr. Bekker, cochair of HVTN 702, International AIDS Society president-elect, and deputy director of the Desmond Tutu HIV Center in Cape Town, South Africa.
As a principal investigator in the NIAID-supported HIV Vaccine Trials Network, Dr. Corey has been deeply involved in the development of this vaccine. He also is chair of the ongoing HVTN 703 and 704 phase IIb trials, testing an entirely different vaccine approach. The hypothesis being tested in HVTN 703 and 704 is that a passively infused monoclonal antibody can protect against HIV infection in 2,400 men who have sex with men and transgender men in North and South America, as well as in 1,500 women in sub-Saharan Africa. Both studies began in spring 2016.
“Every card-carrying virologist feels this should work,” according to Dr. Corey.
The rationale for having two study populations is that investigators suspect the effects of the antibody may vary depending upon whether the route of HIV acquisition is rectal or vaginal, he explained.
The monoclonal antibody contains VRC01, which effectively blocks viral binding to CD4 cells. Study participants will receive an intravenous infusion of VRC01 at 10 or 30 mg/kg or placebo every 2 months. If the results are positive and a second-generation product and delivery system can be developed, antibody-mediated prevention could also have a major potential role in interrupting maternal to child transmission of HIV resulting from intrapartum exposure or breastfeeding.
Dr. Corey also highlighted a third strategy of HIV vaccine development, one at an earlier stage. Investigators at Johnson & Johnson, in collaboration with the NIAID, HVTN, and other partners, are pursuing a multi-clade approach, one designed to protect against all clades of HIV found around the world. This strategy entails first giving an adenovirus serotype 26–vectored vaccine to prime the immune system, following up with administration of several boosters containing mosaic inserts to increase the response. This vaccine is in phase I studies with no results yet.
Dr. Fauci is not sure which if any of these three approaches will yield a safe and effective vaccine for HIV prevention.
“It’s important to realize that this is a very difficult scientific challenge,” he said. “The body does not readily make an adequate immune response against HIV, unlike virtually any other viral infection. Even the serious ones that cause a degree of morbidity and mortality – smallpox, measles, rubella, polio – ultimately the body does make a good immune response and allows us to clear the virus and leaves us with protection against subsequent exposure to the same virus. We don’t have that advantage with HIV. So it’s going to be difficult to get a safe and effective HIV vaccine, but I think the scientific challenge is worth going after and there’s a reasonable chance we might get there.”
Dr. Corey, Dr. Fauci, and Dr. Bekker reported having no financial conflicts of interest.
DURBAN, SOUTH AFRICA – A new optimism regarding the possibility of creating a safe and effective HIV preventive vaccine was very much in evidence at the 21st International AIDS Conference.
“The HIV vaccine field is open for business,” an exuberant Larry Corey, MD, declared in a plenary address highlighting recent major progress in HIV vaccine development.
Three extremely important HIV vaccine efficacy clinical trials testing diverse promising strategies are now either in progress or soon to start, noted Dr. Corey, professor of laboratory medicine and medicine at the University of Washington and emeritus director of the Fred Hutchinson Cancer Research Center in Seattle.
“We are finally moving the needle forward with human efficacy trials that are commensurate with the need for developing an HIV vaccine,” Dr. Corey said.
He emphasized that HIV is “still the world’s most pressing global health issue,” with more than 45,000 new infections occurring annually in the United States and more than 2 million annually worldwide. And while numerous nonvaccine prevention methods have been developed, they share a major limitation: Their extended effectiveness requires continuous adherence.
“With asymptomatic acquisition, prolonged subclinical infection, and sexual transmission, getting to an AIDS-free generation will require a vaccine,” Dr. Corey predicted.
After years of discouragingly negative HIV vaccine studies, researchers finally turned a corner in 2009 with the reported results of the U.S. Military HIV Research Program–led RV144 trial, commonly known as the Thai Trial, Anthony S. Fauci, MD, recalled in an interview.
The trial, which randomized more than 16,000 young adult Thais, showed a modest 31% efficacy at 3.5 years, but a more substantial and encouraging 60% efficacy at 12 months (N Engl J Med. 2009 Dec 3;361[23]:2209-20). More importantly, the Thai trial opened up a whole new avenue to HIV vaccine development.
“We thought the Thai vaccine would induce neutralizing antibodies, but it didn’t. Instead, it induced nonneutralizing antibodies against a component of the V1V2 loop region of the HIV envelope, which was associated with protection. So the good news about that study was that even though that vaccine wasn’t efficacious enough to make it a usable vaccine, it gave us something we could improve upon by using the same platform and enhancing the response to provide greater depth, breadth, and durability,” explained Dr. Fauci, director of the National Institute of Allergy and Infectious Diseases (NIAID).
The improved vaccine consists of a canarypox-based vaccine called ALVAC-HIV and a bivalent gp120 protein subunit vaccine with MF59, a different adjuvant from that used in the Thai trial, in an effort to achieve a more robust immune response. Also, the four-injection series studied in the Thai trial is now bolstered by a booster injection at the 12-month mark. This vaccine has been altered to be specific to HIV clade C, the predominant HIV subtype in southern Africa, where the bulk of new HIV infections occur.
At the AIDS 2016 conference, Linda-Gail Bekker, MD, presented the primary immunogenicity results from the HIV Vaccine Trials Network (HVTN) 100 trial, a phase 1/2, double-blind, placebo-controlled study of the improved version of the Thai trial vaccine, known as the Clade C ALVAC-(vCP2438) and bivalent subtype C gp120/MF59 vaccine, in 252 HIV-uninfected South African adults.
The ALVAC/protein vaccine achieved cellular and humoral immune responses that exceeded all four predetermined criteria as correlates of protection. As a result, a pivotal phase III, randomized, double-blind, placebo-controlled vaccine efficacy trial known as HVTN 702 got the green light to begin in November 2016 in 5,400 HIV-negative adults in South Africa. Participants will be assessed at 24 and 36 months of follow-up, announced Dr. Bekker, cochair of HVTN 702, International AIDS Society president-elect, and deputy director of the Desmond Tutu HIV Center in Cape Town, South Africa.
As a principal investigator in the NIAID-supported HIV Vaccine Trials Network, Dr. Corey has been deeply involved in the development of this vaccine. He also is chair of the ongoing HVTN 703 and 704 phase IIb trials, testing an entirely different vaccine approach. The hypothesis being tested in HVTN 703 and 704 is that a passively infused monoclonal antibody can protect against HIV infection in 2,400 men who have sex with men and transgender men in North and South America, as well as in 1,500 women in sub-Saharan Africa. Both studies began in spring 2016.
“Every card-carrying virologist feels this should work,” according to Dr. Corey.
The rationale for having two study populations is that investigators suspect the effects of the antibody may vary depending upon whether the route of HIV acquisition is rectal or vaginal, he explained.
The monoclonal antibody contains VRC01, which effectively blocks viral binding to CD4 cells. Study participants will receive an intravenous infusion of VRC01 at 10 or 30 mg/kg or placebo every 2 months. If the results are positive and a second-generation product and delivery system can be developed, antibody-mediated prevention could also have a major potential role in interrupting maternal to child transmission of HIV resulting from intrapartum exposure or breastfeeding.
Dr. Corey also highlighted a third strategy of HIV vaccine development, one at an earlier stage. Investigators at Johnson & Johnson, in collaboration with the NIAID, HVTN, and other partners, are pursuing a multi-clade approach, one designed to protect against all clades of HIV found around the world. This strategy entails first giving an adenovirus serotype 26–vectored vaccine to prime the immune system, following up with administration of several boosters containing mosaic inserts to increase the response. This vaccine is in phase I studies with no results yet.
Dr. Fauci is not sure which if any of these three approaches will yield a safe and effective vaccine for HIV prevention.
“It’s important to realize that this is a very difficult scientific challenge,” he said. “The body does not readily make an adequate immune response against HIV, unlike virtually any other viral infection. Even the serious ones that cause a degree of morbidity and mortality – smallpox, measles, rubella, polio – ultimately the body does make a good immune response and allows us to clear the virus and leaves us with protection against subsequent exposure to the same virus. We don’t have that advantage with HIV. So it’s going to be difficult to get a safe and effective HIV vaccine, but I think the scientific challenge is worth going after and there’s a reasonable chance we might get there.”
Dr. Corey, Dr. Fauci, and Dr. Bekker reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM AIDS 2016
ARIA trial: Triumeq proves a winner in HIV-infected women
DURBAN, SOUTH AFRICA – A novel single-tablet, triple-antiretroviral combination regimen outperformed the well-established combination of ritonavir-boosted atazanavir plus tenofovir/emtricitabine in the ARIA trial, a phase IIIb study conducted in treatment-naive women with HIV infection.
The fixed-dose once-daily combination of dolutegravir 50 mg/abacavir 600 mg/lamivudine 300 mg, marketed in the United States as Triumeq since its approval in 2014, showed superior efficacy and a more favorable safety profile than did ritonavir-boosted atazanavir (Reyataz) plus tenofovir/emtricitabine (Truvada), Catherine Orrell, MD, reported at the 21st International AIDS Conference.
ARIA was a 48-week open-label randomized trial involving 495 treatment-naive HIV-infected women in the United States and 11 other countries. The primary endpoint – a plasma HIV RNA viral load below 50 copies/mL after 48 weeks of treatment – was achieved in 82% of patients on dolutegravir/abacavir/lamivudine (DTG/ABC/3TC), compared with 71% on ritonavir-boosted atazanavir plus tenofovir/emtricitabine (ATV+RTV+FTC/TDF).
Of note, the difference in efficacy was even more pronounced among women with a baseline viral load in excess of 100,000 copies/mL: an 80% success rate in attaining a viral load of less than 50 copies/mL at 48 weeks in the DTG/ABC/3TC group, compared with 64% in the comparator arm. Women with this baseline massive viral load comprised 28% of study participants, added Dr. Orrell of the University of Cape Town, South Africa.
To be eligible for the study, women had to be negative for an HLA-B*5701 genetic screen for allergic hypersensitivity to abacavir.
A particularly attractive feature of DTG/ABC/3TC is that dolutegravir, an unboosted integrin strand transfer inhibitor, provides a high barrier to development of drug resistance. Indeed, no subjects in the dolutegravir arm developed treatment-emergent primary integrin strand transfer inhibitor or abacavir/lamivudine resistance mutations, according to Dr. Orrell.
The superior efficacy of the single-tablet regimen was driven in part by fewer discontinuations due to adverse events: a 4% rate versus 7% in the comparator arm. Another key factor was the substantially lower virologic failure rate in DTG/ABC/3TC-treated women: 6% versus 14% in the comparator arm.
The single-tablet regimen also had a better safety profile. The combined rate of the most common drug-related adverse events – nausea, diarrhea, headache, and jaundice – was 22% in the DTG/ABC/3TC group compared with 38% with ATV+RTV+FTC/TDF.
The incidence of treatment-emergent psychiatric events – insomnia, anxiety, depression, or suicidal ideation – was roughly 14% in each treatment arm. That’s an important finding because some other studies have found an increase in psychiatric events in patients receiving integrin strand transfer inhibitors.
“The overall results, I think, are important for the field,” Kimberly Smith, MD, said at a press conference highlighting the ARIA trial.
“Women are often underrepresented in HIV clinical trials even though they bear much of the burden of the HIV epidemic,” added Dr. Smith, vice president for global medical strategy and head of research and development at ViiV Healthcare in Research Triangle Park, N.C.
“This fixed-dose combination is a winner,” said Salim Abdool Karim, MD, director of the Center for the AIDS Program of Research in South Africa, Durban, who chaired the press conference.
Dr. Orrell received a research grant from ViiV Healthcare, which sponsored the ARIA study.
DURBAN, SOUTH AFRICA – A novel single-tablet, triple-antiretroviral combination regimen outperformed the well-established combination of ritonavir-boosted atazanavir plus tenofovir/emtricitabine in the ARIA trial, a phase IIIb study conducted in treatment-naive women with HIV infection.
The fixed-dose once-daily combination of dolutegravir 50 mg/abacavir 600 mg/lamivudine 300 mg, marketed in the United States as Triumeq since its approval in 2014, showed superior efficacy and a more favorable safety profile than did ritonavir-boosted atazanavir (Reyataz) plus tenofovir/emtricitabine (Truvada), Catherine Orrell, MD, reported at the 21st International AIDS Conference.
ARIA was a 48-week open-label randomized trial involving 495 treatment-naive HIV-infected women in the United States and 11 other countries. The primary endpoint – a plasma HIV RNA viral load below 50 copies/mL after 48 weeks of treatment – was achieved in 82% of patients on dolutegravir/abacavir/lamivudine (DTG/ABC/3TC), compared with 71% on ritonavir-boosted atazanavir plus tenofovir/emtricitabine (ATV+RTV+FTC/TDF).
Of note, the difference in efficacy was even more pronounced among women with a baseline viral load in excess of 100,000 copies/mL: an 80% success rate in attaining a viral load of less than 50 copies/mL at 48 weeks in the DTG/ABC/3TC group, compared with 64% in the comparator arm. Women with this baseline massive viral load comprised 28% of study participants, added Dr. Orrell of the University of Cape Town, South Africa.
To be eligible for the study, women had to be negative for an HLA-B*5701 genetic screen for allergic hypersensitivity to abacavir.
A particularly attractive feature of DTG/ABC/3TC is that dolutegravir, an unboosted integrin strand transfer inhibitor, provides a high barrier to development of drug resistance. Indeed, no subjects in the dolutegravir arm developed treatment-emergent primary integrin strand transfer inhibitor or abacavir/lamivudine resistance mutations, according to Dr. Orrell.
The superior efficacy of the single-tablet regimen was driven in part by fewer discontinuations due to adverse events: a 4% rate versus 7% in the comparator arm. Another key factor was the substantially lower virologic failure rate in DTG/ABC/3TC-treated women: 6% versus 14% in the comparator arm.
The single-tablet regimen also had a better safety profile. The combined rate of the most common drug-related adverse events – nausea, diarrhea, headache, and jaundice – was 22% in the DTG/ABC/3TC group compared with 38% with ATV+RTV+FTC/TDF.
The incidence of treatment-emergent psychiatric events – insomnia, anxiety, depression, or suicidal ideation – was roughly 14% in each treatment arm. That’s an important finding because some other studies have found an increase in psychiatric events in patients receiving integrin strand transfer inhibitors.
“The overall results, I think, are important for the field,” Kimberly Smith, MD, said at a press conference highlighting the ARIA trial.
“Women are often underrepresented in HIV clinical trials even though they bear much of the burden of the HIV epidemic,” added Dr. Smith, vice president for global medical strategy and head of research and development at ViiV Healthcare in Research Triangle Park, N.C.
“This fixed-dose combination is a winner,” said Salim Abdool Karim, MD, director of the Center for the AIDS Program of Research in South Africa, Durban, who chaired the press conference.
Dr. Orrell received a research grant from ViiV Healthcare, which sponsored the ARIA study.
DURBAN, SOUTH AFRICA – A novel single-tablet, triple-antiretroviral combination regimen outperformed the well-established combination of ritonavir-boosted atazanavir plus tenofovir/emtricitabine in the ARIA trial, a phase IIIb study conducted in treatment-naive women with HIV infection.
The fixed-dose once-daily combination of dolutegravir 50 mg/abacavir 600 mg/lamivudine 300 mg, marketed in the United States as Triumeq since its approval in 2014, showed superior efficacy and a more favorable safety profile than did ritonavir-boosted atazanavir (Reyataz) plus tenofovir/emtricitabine (Truvada), Catherine Orrell, MD, reported at the 21st International AIDS Conference.
ARIA was a 48-week open-label randomized trial involving 495 treatment-naive HIV-infected women in the United States and 11 other countries. The primary endpoint – a plasma HIV RNA viral load below 50 copies/mL after 48 weeks of treatment – was achieved in 82% of patients on dolutegravir/abacavir/lamivudine (DTG/ABC/3TC), compared with 71% on ritonavir-boosted atazanavir plus tenofovir/emtricitabine (ATV+RTV+FTC/TDF).
Of note, the difference in efficacy was even more pronounced among women with a baseline viral load in excess of 100,000 copies/mL: an 80% success rate in attaining a viral load of less than 50 copies/mL at 48 weeks in the DTG/ABC/3TC group, compared with 64% in the comparator arm. Women with this baseline massive viral load comprised 28% of study participants, added Dr. Orrell of the University of Cape Town, South Africa.
To be eligible for the study, women had to be negative for an HLA-B*5701 genetic screen for allergic hypersensitivity to abacavir.
A particularly attractive feature of DTG/ABC/3TC is that dolutegravir, an unboosted integrin strand transfer inhibitor, provides a high barrier to development of drug resistance. Indeed, no subjects in the dolutegravir arm developed treatment-emergent primary integrin strand transfer inhibitor or abacavir/lamivudine resistance mutations, according to Dr. Orrell.
The superior efficacy of the single-tablet regimen was driven in part by fewer discontinuations due to adverse events: a 4% rate versus 7% in the comparator arm. Another key factor was the substantially lower virologic failure rate in DTG/ABC/3TC-treated women: 6% versus 14% in the comparator arm.
The single-tablet regimen also had a better safety profile. The combined rate of the most common drug-related adverse events – nausea, diarrhea, headache, and jaundice – was 22% in the DTG/ABC/3TC group compared with 38% with ATV+RTV+FTC/TDF.
The incidence of treatment-emergent psychiatric events – insomnia, anxiety, depression, or suicidal ideation – was roughly 14% in each treatment arm. That’s an important finding because some other studies have found an increase in psychiatric events in patients receiving integrin strand transfer inhibitors.
“The overall results, I think, are important for the field,” Kimberly Smith, MD, said at a press conference highlighting the ARIA trial.
“Women are often underrepresented in HIV clinical trials even though they bear much of the burden of the HIV epidemic,” added Dr. Smith, vice president for global medical strategy and head of research and development at ViiV Healthcare in Research Triangle Park, N.C.
“This fixed-dose combination is a winner,” said Salim Abdool Karim, MD, director of the Center for the AIDS Program of Research in South Africa, Durban, who chaired the press conference.
Dr. Orrell received a research grant from ViiV Healthcare, which sponsored the ARIA study.
AT AIDS 2016
Key clinical point: The once-daily fixed-dose combination of dolutegravir/abacavir/lamivudine is significantly more effective and has a better safety profile than does ritonavir-boosted atazanavir plus tenofovir/emtricitabine in treatment-naive women with HIV infection.
Major finding: Eighty-two percent of women assigned to dolutegravir/abacavir/lamivudine had a plasma HIV RNA viral load below 50 copies/mL after 48 weeks of treatment, compared with 71% on ritonavir-boosted atazanavir plus tenofovir/emtricitabine.
Data source: A phase IIIb open-label randomized trial of 495 HIV-infected women.
Disclosures: The presenter received a research grant from ViiV Healthcare, which sponsored the study.
GAD tied to twice the risk of cancer mortality in men
VIENNA – Generalized anxiety disorder in men over age 40 was independently associated with a greater than twofold increased risk of death due to cancer during 15 years of follow-up, compared with men without the psychiatric disorder, in a large longitudinal population-based study, Olivia Remes reported at the annual congress of the European College of Neuropsychopharmacology.
In contrast, women with generalized anxiety disorder (GAD) were at no increased risk for cancer mortality, according to Ms. Remes, a PhD candidate in the department of public health and primary care at the University of Cambridge, England.
“I think an important message of this study is that anxiety is not just a personality trait,” she said in an interview. “In some cases, it may be more than worries and stress; it can represent a possible marker for future ill physical health that general practitioners and psychiatrists ought to be aware of.”
This creates a dilemma, because individuals with GAD typically are high users of health care resources. They frequently present in physicians’ offices and emergency departments with a wide range of physical complaints, including stomach pain, breathing difficulty, fatigue, nausea, or peripheral numbness.
Ms. Remes presented an analysis of 7,139 men and 8,799 women over age 40 at enrollment in the European Prospective Investigation of Cancer–Norfolk Study from the period of 1996-2000. At entry, the patients underwent a physical examination and completed numerous health-related questionnaires. Among these was the Health and Life Experiences Questionnaire (HLEQ), the responses to which enabled investigators to identify 1.8% of the men and 2.4% of women as meeting DSM-IV diagnostic criteria for GAD.
During 15 years of follow-up, 796 men and 648 women died of cancer. In multivariate analysis, men with GAD were at a 2.14-fold increased risk of death due to cancer. This analysis was extensively adjusted for potential confounders, including age, marital status, education level, socioeconomic status, major chronic physical illnesses, smoking, alcohol intake, major depressive disorder, body mass index, physical activity level, and the use of antidepressants or antihypnotic agents.
This was the first large, long-term study to examine the relationship between GAD and cancer mortality. Prior studies yielded conflicting results but were limited by small sample size and/or short follow-up, Ms. Remes said.
Even with more than 1,400 cancer deaths in the study, the sample size is too small to say whether GAD in men is preferentially associated with death due to any specific type of cancer.
Severe chronic anxiety has been linked to increased systemic inflammation, an observation that points to a possible mechanism by which GAD might increase affected individuals’ risk of fatal cancer as well as cardiovascular disease, chronic lung disease, and other conditions where inflammation figures prominently. But that doesn’t explain the gender difference in cancer mortality risk, the source of which remains speculative. It’s not the result of men being more likely to be smokers or to abuse alcohol, since those carcinogenic lifestyle factors were controlled for in the multivariate analysis, she observed.
One hypothetical possibility is that men with GAD who develop a malignancy might be more likely to delay seeking medical attention for it than might men without the anxiety disorder. Ms. Remes and her coinvestigators plan to explore this possibility in a roundabout way by analyzing the Norfolk study database looking for differences between men with and without GAD in the time from cancer diagnosis to death.
To eliminate the possibility that GAD in men might be triggered by an occult and ultimately fatal cancer, the investigators reanalyzed the Norfolk study data after excluding deaths that occurred during the first 5 years of follow-up. The results remained unchanged, according to Ms. Remes.
She reported having no financial conflicts regarding this study, which was sponsored by the U.K. Medical Research Council and Cancer Research UK.
VIENNA – Generalized anxiety disorder in men over age 40 was independently associated with a greater than twofold increased risk of death due to cancer during 15 years of follow-up, compared with men without the psychiatric disorder, in a large longitudinal population-based study, Olivia Remes reported at the annual congress of the European College of Neuropsychopharmacology.
In contrast, women with generalized anxiety disorder (GAD) were at no increased risk for cancer mortality, according to Ms. Remes, a PhD candidate in the department of public health and primary care at the University of Cambridge, England.
“I think an important message of this study is that anxiety is not just a personality trait,” she said in an interview. “In some cases, it may be more than worries and stress; it can represent a possible marker for future ill physical health that general practitioners and psychiatrists ought to be aware of.”
This creates a dilemma, because individuals with GAD typically are high users of health care resources. They frequently present in physicians’ offices and emergency departments with a wide range of physical complaints, including stomach pain, breathing difficulty, fatigue, nausea, or peripheral numbness.
Ms. Remes presented an analysis of 7,139 men and 8,799 women over age 40 at enrollment in the European Prospective Investigation of Cancer–Norfolk Study from the period of 1996-2000. At entry, the patients underwent a physical examination and completed numerous health-related questionnaires. Among these was the Health and Life Experiences Questionnaire (HLEQ), the responses to which enabled investigators to identify 1.8% of the men and 2.4% of women as meeting DSM-IV diagnostic criteria for GAD.
During 15 years of follow-up, 796 men and 648 women died of cancer. In multivariate analysis, men with GAD were at a 2.14-fold increased risk of death due to cancer. This analysis was extensively adjusted for potential confounders, including age, marital status, education level, socioeconomic status, major chronic physical illnesses, smoking, alcohol intake, major depressive disorder, body mass index, physical activity level, and the use of antidepressants or antihypnotic agents.
This was the first large, long-term study to examine the relationship between GAD and cancer mortality. Prior studies yielded conflicting results but were limited by small sample size and/or short follow-up, Ms. Remes said.
Even with more than 1,400 cancer deaths in the study, the sample size is too small to say whether GAD in men is preferentially associated with death due to any specific type of cancer.
Severe chronic anxiety has been linked to increased systemic inflammation, an observation that points to a possible mechanism by which GAD might increase affected individuals’ risk of fatal cancer as well as cardiovascular disease, chronic lung disease, and other conditions where inflammation figures prominently. But that doesn’t explain the gender difference in cancer mortality risk, the source of which remains speculative. It’s not the result of men being more likely to be smokers or to abuse alcohol, since those carcinogenic lifestyle factors were controlled for in the multivariate analysis, she observed.
One hypothetical possibility is that men with GAD who develop a malignancy might be more likely to delay seeking medical attention for it than might men without the anxiety disorder. Ms. Remes and her coinvestigators plan to explore this possibility in a roundabout way by analyzing the Norfolk study database looking for differences between men with and without GAD in the time from cancer diagnosis to death.
To eliminate the possibility that GAD in men might be triggered by an occult and ultimately fatal cancer, the investigators reanalyzed the Norfolk study data after excluding deaths that occurred during the first 5 years of follow-up. The results remained unchanged, according to Ms. Remes.
She reported having no financial conflicts regarding this study, which was sponsored by the U.K. Medical Research Council and Cancer Research UK.
VIENNA – Generalized anxiety disorder in men over age 40 was independently associated with a greater than twofold increased risk of death due to cancer during 15 years of follow-up, compared with men without the psychiatric disorder, in a large longitudinal population-based study, Olivia Remes reported at the annual congress of the European College of Neuropsychopharmacology.
In contrast, women with generalized anxiety disorder (GAD) were at no increased risk for cancer mortality, according to Ms. Remes, a PhD candidate in the department of public health and primary care at the University of Cambridge, England.
“I think an important message of this study is that anxiety is not just a personality trait,” she said in an interview. “In some cases, it may be more than worries and stress; it can represent a possible marker for future ill physical health that general practitioners and psychiatrists ought to be aware of.”
This creates a dilemma, because individuals with GAD typically are high users of health care resources. They frequently present in physicians’ offices and emergency departments with a wide range of physical complaints, including stomach pain, breathing difficulty, fatigue, nausea, or peripheral numbness.
Ms. Remes presented an analysis of 7,139 men and 8,799 women over age 40 at enrollment in the European Prospective Investigation of Cancer–Norfolk Study from the period of 1996-2000. At entry, the patients underwent a physical examination and completed numerous health-related questionnaires. Among these was the Health and Life Experiences Questionnaire (HLEQ), the responses to which enabled investigators to identify 1.8% of the men and 2.4% of women as meeting DSM-IV diagnostic criteria for GAD.
During 15 years of follow-up, 796 men and 648 women died of cancer. In multivariate analysis, men with GAD were at a 2.14-fold increased risk of death due to cancer. This analysis was extensively adjusted for potential confounders, including age, marital status, education level, socioeconomic status, major chronic physical illnesses, smoking, alcohol intake, major depressive disorder, body mass index, physical activity level, and the use of antidepressants or antihypnotic agents.
This was the first large, long-term study to examine the relationship between GAD and cancer mortality. Prior studies yielded conflicting results but were limited by small sample size and/or short follow-up, Ms. Remes said.
Even with more than 1,400 cancer deaths in the study, the sample size is too small to say whether GAD in men is preferentially associated with death due to any specific type of cancer.
Severe chronic anxiety has been linked to increased systemic inflammation, an observation that points to a possible mechanism by which GAD might increase affected individuals’ risk of fatal cancer as well as cardiovascular disease, chronic lung disease, and other conditions where inflammation figures prominently. But that doesn’t explain the gender difference in cancer mortality risk, the source of which remains speculative. It’s not the result of men being more likely to be smokers or to abuse alcohol, since those carcinogenic lifestyle factors were controlled for in the multivariate analysis, she observed.
One hypothetical possibility is that men with GAD who develop a malignancy might be more likely to delay seeking medical attention for it than might men without the anxiety disorder. Ms. Remes and her coinvestigators plan to explore this possibility in a roundabout way by analyzing the Norfolk study database looking for differences between men with and without GAD in the time from cancer diagnosis to death.
To eliminate the possibility that GAD in men might be triggered by an occult and ultimately fatal cancer, the investigators reanalyzed the Norfolk study data after excluding deaths that occurred during the first 5 years of follow-up. The results remained unchanged, according to Ms. Remes.
She reported having no financial conflicts regarding this study, which was sponsored by the U.K. Medical Research Council and Cancer Research UK.
AT THE ECNP CONGRESS
Key clinical point: Men with generalized anxiety disorder are more than twice as likely to die from cancer, compared with men without the disorder.
Major finding: Men over age 40 with generalized anxiety disorder were 2.14 times more likely to die of cancer within 15 years than were those without the disorder.
Data source: A longitudinal, population-based study of nearly 16,000 Norfolk, England–area adults aged 40 or older who were followed for 15 years.
Disclosures: The U.K. Medical Research Council and Cancer Research UK sponsored the study. The presenter reported having no financial conflicts of interest.
OCD undertreatment: Is Internet-based CBT the answer?
VIENNA – Christian Rück, MD, believes he has seen the future of treatment for obsessive-compulsive disorder, and it’s Internet-based cognitive-behavioral therapy.
The numbers tell the tale. Obsessive-compulsive disorder (OCD) affects 2% of the general population. It’s an often disabling condition marked by shame and stigma. Major practice guidelines recommend cognitive-behavioral therapy (CBT) as the evidence-based first-line nonpharmacologic therapy. Yet only 5%-10% of OCD patients ever receive conventional face-to-face CBT because of the severe shortage and geographic maldistribution of therapists trained in its use, the psychiatrist observed at the annual congress of the European College of Neuropsychopharmacology.
Thus, a huge unmet need exists for treatment access. And persuasive evidence now exists that Internet-based CBT (I-CBT) for OCD can be provided effectively, safely, and in an extremely cost-effective fashion. Indeed, it takes up very little therapist time – an average of 6-10 minutes per patient per week devoted to reading and answering participants’ emails. And since the therapist’s main role in this treatment approach is simply to encourage patient engagement in the structured online program, I-CBT readily lends itself to use by primary care physicians and other nonspecialists, according to Dr. Rück, a psychiatrist at the Karolinska Institute in Stockholm.
“The vast majority of OCD patients have access to nothing. To me, I-CBT provides us with a unique opportunity to sustain quality of care but still make it very widely available. It’s sort of like serving a top-notch lobster meal at McDonald’s prices,” he said.
Over the past 15 years, I-CBT programs have been developed and proven effective in more than 150 randomized controlled trials for a wide range of psychiatric and medical conditions, including depression, panic disorder, social phobia, severe health anxiety and other anxiety disorders, erectile dysfunction, atrial fibrillation, fibromyalgia, irritable bowel syndrome, and insomnia.
More recently, Dr. Rück and his coinvestigators have pioneered the development of I-CBT as an evidence-based treatment for OCD. They now are in the process of doing the same for body dysmorphic disorder, a related condition that’s often particularly challenging to treat successfully.
The investigators’ first large randomized controlled trial of I-CBT for OCD included 101 patients with a mean 18-year history of the disorder. They were assigned to 10 weeks of the online therapy or to an online supportive care control arm. Mean scores on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) improved from 21.4 at baseline to 12.9 upon completion of the I-CBT program and 12.6 at follow-up 4 months post treatment. Sixty percent of the I-CBT group demonstrated clinically significant improvement, compared with 6% of controls (Psychol Med. 2012 Oct;42[10]:2193-203).
The benefit proved durable, as demonstrated in an extension study with periodic follow-up out to 24 months post completion of the I-CBT program. The incorporation of an Internet-based booster program in one arm of the follow-up protocol protected against relapses (Psychol Med. 2014 Oct;44[13]2877-87).
Dr. Rück and his coinvestigators also have shown in a 128-patient, double-blind trial that d-cycloserine, a partial N-methyl-d-aspartate agonist that promotes fear extinction, augmented the response to I-CBT for OCD, but only in the subgroup not on concomitant antidepressant therapy. The remission rate was 60% at 3 months follow-up post I-CBT in antidepressant-free patients on d-cycloserine, compared with just 24% in d-cycloserine recipients taking antidepressants during I-CBT. Apparently, antidepressants blunt d-cycloserine’s fear-extinction effect (JAMA Psychiatry. 2015 Jul;72[7]:659-67).
The program content of I-CBT is the same as in conventional manualized CBT, but without the face-to-face contact. The I-CBT program consists of 10-15 weekly chapters or modules. There is homework, worksheets, and titrated exposures to fear-eliciting situations. Progress to the next module is contingent upon completion of the previous one, with the patient being required to successfully answer questions that show mastery of the material.
Most patients rate the program favorably. They like not having to show up in the therapist’s office week after week.
“One of the advantages of I-CBT is you can interact every day. If your exposure goes wrong you don’t have to wait 6 days until your next face-to-face appointment with your therapist; you can actually get treatment right away and in that way increase the speed of progress,” Dr. Rück observed.
Also, if the patient forgets a basic concept, he or she can go back and reread.
“Everything is saved. This makes it very easy to supervise. There’s nothing going on that can’t be seen. Isaac M. Marks, MD, an early OCD researcher at Imperial College London, once said that one of the greatest advantages of I-CBT is that the therapist can’t sleep with the patient,” Dr. Rück recalled.
His research group is now in the midst of a clinical trial designed to answer two key questions: Is I-CBT for OCD as effective as face-to-face CBT? And is the therapist really necessary in order to achieve optimal outcomes in I-CBT?
“It would be a kind of shock to us if all our therapist impact is for nothing, but we’ll find out soon,” he promised.
Dr. Rück and his coinvestigators recently developed a program for therapist-supervised I-CBT for patients with body dysmorphic disorder (BDD). In a single-blind pilot clinical trial involving 94 adults with BDD, the clinical response rate at 6 months of follow-up as defined by at least a 30% reduction from baseline in scores on the BDD–Y-BOCS was 56% in the I-CBT arm and 13% in controls assigned to online supportive care. The number needed to treat was impressively low, at 2.3. And those results were achieved by clinical psychology students having no prior experience with BDD, although they were closely supervised by an experienced clinician (BMJ. 2016 Feb 2;352:i241.
Dr. Rück has been disappointed to find that physicians, psychotherapists, and health care systems have been slow to embrace I-CBT for the conditions where there is solid supportive randomized trial evidence. I-CBT has been implemented within health care systems only in Australia and Stockholm County, the places where most of the research has been conducted.
“The literature has been around for about 15 years now, and we are noticing that the rollout of this is terribly slow. I’m starting to think that the health care system itself is one of the most problematic roadblocks, especially with regard to reimbursement issues. Maybe I-CBT should be offered outside of the health care system. Sometimes I think Google would do this better than a nationalized health care system,” the psychiatrist mused.
He reported having no financial conflicts of interest regarding his presentation.
VIENNA – Christian Rück, MD, believes he has seen the future of treatment for obsessive-compulsive disorder, and it’s Internet-based cognitive-behavioral therapy.
The numbers tell the tale. Obsessive-compulsive disorder (OCD) affects 2% of the general population. It’s an often disabling condition marked by shame and stigma. Major practice guidelines recommend cognitive-behavioral therapy (CBT) as the evidence-based first-line nonpharmacologic therapy. Yet only 5%-10% of OCD patients ever receive conventional face-to-face CBT because of the severe shortage and geographic maldistribution of therapists trained in its use, the psychiatrist observed at the annual congress of the European College of Neuropsychopharmacology.
Thus, a huge unmet need exists for treatment access. And persuasive evidence now exists that Internet-based CBT (I-CBT) for OCD can be provided effectively, safely, and in an extremely cost-effective fashion. Indeed, it takes up very little therapist time – an average of 6-10 minutes per patient per week devoted to reading and answering participants’ emails. And since the therapist’s main role in this treatment approach is simply to encourage patient engagement in the structured online program, I-CBT readily lends itself to use by primary care physicians and other nonspecialists, according to Dr. Rück, a psychiatrist at the Karolinska Institute in Stockholm.
“The vast majority of OCD patients have access to nothing. To me, I-CBT provides us with a unique opportunity to sustain quality of care but still make it very widely available. It’s sort of like serving a top-notch lobster meal at McDonald’s prices,” he said.
Over the past 15 years, I-CBT programs have been developed and proven effective in more than 150 randomized controlled trials for a wide range of psychiatric and medical conditions, including depression, panic disorder, social phobia, severe health anxiety and other anxiety disorders, erectile dysfunction, atrial fibrillation, fibromyalgia, irritable bowel syndrome, and insomnia.
More recently, Dr. Rück and his coinvestigators have pioneered the development of I-CBT as an evidence-based treatment for OCD. They now are in the process of doing the same for body dysmorphic disorder, a related condition that’s often particularly challenging to treat successfully.
The investigators’ first large randomized controlled trial of I-CBT for OCD included 101 patients with a mean 18-year history of the disorder. They were assigned to 10 weeks of the online therapy or to an online supportive care control arm. Mean scores on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) improved from 21.4 at baseline to 12.9 upon completion of the I-CBT program and 12.6 at follow-up 4 months post treatment. Sixty percent of the I-CBT group demonstrated clinically significant improvement, compared with 6% of controls (Psychol Med. 2012 Oct;42[10]:2193-203).
The benefit proved durable, as demonstrated in an extension study with periodic follow-up out to 24 months post completion of the I-CBT program. The incorporation of an Internet-based booster program in one arm of the follow-up protocol protected against relapses (Psychol Med. 2014 Oct;44[13]2877-87).
Dr. Rück and his coinvestigators also have shown in a 128-patient, double-blind trial that d-cycloserine, a partial N-methyl-d-aspartate agonist that promotes fear extinction, augmented the response to I-CBT for OCD, but only in the subgroup not on concomitant antidepressant therapy. The remission rate was 60% at 3 months follow-up post I-CBT in antidepressant-free patients on d-cycloserine, compared with just 24% in d-cycloserine recipients taking antidepressants during I-CBT. Apparently, antidepressants blunt d-cycloserine’s fear-extinction effect (JAMA Psychiatry. 2015 Jul;72[7]:659-67).
The program content of I-CBT is the same as in conventional manualized CBT, but without the face-to-face contact. The I-CBT program consists of 10-15 weekly chapters or modules. There is homework, worksheets, and titrated exposures to fear-eliciting situations. Progress to the next module is contingent upon completion of the previous one, with the patient being required to successfully answer questions that show mastery of the material.
Most patients rate the program favorably. They like not having to show up in the therapist’s office week after week.
“One of the advantages of I-CBT is you can interact every day. If your exposure goes wrong you don’t have to wait 6 days until your next face-to-face appointment with your therapist; you can actually get treatment right away and in that way increase the speed of progress,” Dr. Rück observed.
Also, if the patient forgets a basic concept, he or she can go back and reread.
“Everything is saved. This makes it very easy to supervise. There’s nothing going on that can’t be seen. Isaac M. Marks, MD, an early OCD researcher at Imperial College London, once said that one of the greatest advantages of I-CBT is that the therapist can’t sleep with the patient,” Dr. Rück recalled.
His research group is now in the midst of a clinical trial designed to answer two key questions: Is I-CBT for OCD as effective as face-to-face CBT? And is the therapist really necessary in order to achieve optimal outcomes in I-CBT?
“It would be a kind of shock to us if all our therapist impact is for nothing, but we’ll find out soon,” he promised.
Dr. Rück and his coinvestigators recently developed a program for therapist-supervised I-CBT for patients with body dysmorphic disorder (BDD). In a single-blind pilot clinical trial involving 94 adults with BDD, the clinical response rate at 6 months of follow-up as defined by at least a 30% reduction from baseline in scores on the BDD–Y-BOCS was 56% in the I-CBT arm and 13% in controls assigned to online supportive care. The number needed to treat was impressively low, at 2.3. And those results were achieved by clinical psychology students having no prior experience with BDD, although they were closely supervised by an experienced clinician (BMJ. 2016 Feb 2;352:i241.
Dr. Rück has been disappointed to find that physicians, psychotherapists, and health care systems have been slow to embrace I-CBT for the conditions where there is solid supportive randomized trial evidence. I-CBT has been implemented within health care systems only in Australia and Stockholm County, the places where most of the research has been conducted.
“The literature has been around for about 15 years now, and we are noticing that the rollout of this is terribly slow. I’m starting to think that the health care system itself is one of the most problematic roadblocks, especially with regard to reimbursement issues. Maybe I-CBT should be offered outside of the health care system. Sometimes I think Google would do this better than a nationalized health care system,” the psychiatrist mused.
He reported having no financial conflicts of interest regarding his presentation.
VIENNA – Christian Rück, MD, believes he has seen the future of treatment for obsessive-compulsive disorder, and it’s Internet-based cognitive-behavioral therapy.
The numbers tell the tale. Obsessive-compulsive disorder (OCD) affects 2% of the general population. It’s an often disabling condition marked by shame and stigma. Major practice guidelines recommend cognitive-behavioral therapy (CBT) as the evidence-based first-line nonpharmacologic therapy. Yet only 5%-10% of OCD patients ever receive conventional face-to-face CBT because of the severe shortage and geographic maldistribution of therapists trained in its use, the psychiatrist observed at the annual congress of the European College of Neuropsychopharmacology.
Thus, a huge unmet need exists for treatment access. And persuasive evidence now exists that Internet-based CBT (I-CBT) for OCD can be provided effectively, safely, and in an extremely cost-effective fashion. Indeed, it takes up very little therapist time – an average of 6-10 minutes per patient per week devoted to reading and answering participants’ emails. And since the therapist’s main role in this treatment approach is simply to encourage patient engagement in the structured online program, I-CBT readily lends itself to use by primary care physicians and other nonspecialists, according to Dr. Rück, a psychiatrist at the Karolinska Institute in Stockholm.
“The vast majority of OCD patients have access to nothing. To me, I-CBT provides us with a unique opportunity to sustain quality of care but still make it very widely available. It’s sort of like serving a top-notch lobster meal at McDonald’s prices,” he said.
Over the past 15 years, I-CBT programs have been developed and proven effective in more than 150 randomized controlled trials for a wide range of psychiatric and medical conditions, including depression, panic disorder, social phobia, severe health anxiety and other anxiety disorders, erectile dysfunction, atrial fibrillation, fibromyalgia, irritable bowel syndrome, and insomnia.
More recently, Dr. Rück and his coinvestigators have pioneered the development of I-CBT as an evidence-based treatment for OCD. They now are in the process of doing the same for body dysmorphic disorder, a related condition that’s often particularly challenging to treat successfully.
The investigators’ first large randomized controlled trial of I-CBT for OCD included 101 patients with a mean 18-year history of the disorder. They were assigned to 10 weeks of the online therapy or to an online supportive care control arm. Mean scores on the Yale-Brown Obsessive-Compulsive Scale (Y-BOCS) improved from 21.4 at baseline to 12.9 upon completion of the I-CBT program and 12.6 at follow-up 4 months post treatment. Sixty percent of the I-CBT group demonstrated clinically significant improvement, compared with 6% of controls (Psychol Med. 2012 Oct;42[10]:2193-203).
The benefit proved durable, as demonstrated in an extension study with periodic follow-up out to 24 months post completion of the I-CBT program. The incorporation of an Internet-based booster program in one arm of the follow-up protocol protected against relapses (Psychol Med. 2014 Oct;44[13]2877-87).
Dr. Rück and his coinvestigators also have shown in a 128-patient, double-blind trial that d-cycloserine, a partial N-methyl-d-aspartate agonist that promotes fear extinction, augmented the response to I-CBT for OCD, but only in the subgroup not on concomitant antidepressant therapy. The remission rate was 60% at 3 months follow-up post I-CBT in antidepressant-free patients on d-cycloserine, compared with just 24% in d-cycloserine recipients taking antidepressants during I-CBT. Apparently, antidepressants blunt d-cycloserine’s fear-extinction effect (JAMA Psychiatry. 2015 Jul;72[7]:659-67).
The program content of I-CBT is the same as in conventional manualized CBT, but without the face-to-face contact. The I-CBT program consists of 10-15 weekly chapters or modules. There is homework, worksheets, and titrated exposures to fear-eliciting situations. Progress to the next module is contingent upon completion of the previous one, with the patient being required to successfully answer questions that show mastery of the material.
Most patients rate the program favorably. They like not having to show up in the therapist’s office week after week.
“One of the advantages of I-CBT is you can interact every day. If your exposure goes wrong you don’t have to wait 6 days until your next face-to-face appointment with your therapist; you can actually get treatment right away and in that way increase the speed of progress,” Dr. Rück observed.
Also, if the patient forgets a basic concept, he or she can go back and reread.
“Everything is saved. This makes it very easy to supervise. There’s nothing going on that can’t be seen. Isaac M. Marks, MD, an early OCD researcher at Imperial College London, once said that one of the greatest advantages of I-CBT is that the therapist can’t sleep with the patient,” Dr. Rück recalled.
His research group is now in the midst of a clinical trial designed to answer two key questions: Is I-CBT for OCD as effective as face-to-face CBT? And is the therapist really necessary in order to achieve optimal outcomes in I-CBT?
“It would be a kind of shock to us if all our therapist impact is for nothing, but we’ll find out soon,” he promised.
Dr. Rück and his coinvestigators recently developed a program for therapist-supervised I-CBT for patients with body dysmorphic disorder (BDD). In a single-blind pilot clinical trial involving 94 adults with BDD, the clinical response rate at 6 months of follow-up as defined by at least a 30% reduction from baseline in scores on the BDD–Y-BOCS was 56% in the I-CBT arm and 13% in controls assigned to online supportive care. The number needed to treat was impressively low, at 2.3. And those results were achieved by clinical psychology students having no prior experience with BDD, although they were closely supervised by an experienced clinician (BMJ. 2016 Feb 2;352:i241.
Dr. Rück has been disappointed to find that physicians, psychotherapists, and health care systems have been slow to embrace I-CBT for the conditions where there is solid supportive randomized trial evidence. I-CBT has been implemented within health care systems only in Australia and Stockholm County, the places where most of the research has been conducted.
“The literature has been around for about 15 years now, and we are noticing that the rollout of this is terribly slow. I’m starting to think that the health care system itself is one of the most problematic roadblocks, especially with regard to reimbursement issues. Maybe I-CBT should be offered outside of the health care system. Sometimes I think Google would do this better than a nationalized health care system,” the psychiatrist mused.
He reported having no financial conflicts of interest regarding his presentation.
EXPERT ANALYSIS At THE ECNP CONGRESS
CABG reduces cardiovascular mortality in ischemic heart failure regardless of age
ROME – Coronary artery bypass surgery reduces cardiovascular mortality in heart failure patients with ischemic cardiomyopathy to a consistent extent regardless of age at the time of surgery, according to a secondary analysis from the landmark STICH trial, Eric J. Velazquez, MD, reported at the annual congress of the European Society of Cardiology.
“Cardiologists and cardiac surgeons can confidently offer patients CABG in addition to optimal medical therapy with the knowledge that cardiovascular mortality is reduced by CABG to a similar extent across all age groups in this trial through 10 years of follow-up,” said Dr. Velazquez, professor of medicine at Duke University in Durham, N.C.
However, that’s only part of the story. Cardiovascular mortality was a secondary endpoint in STICH (Surgical Treatment for Ischemic Heart Failure). The primary endpoint was all-cause mortality. And CABG’s impact on all-cause mortality was diminished in older STICH participants because of their greater comorbidity burden and the competing risk of noncardiovascular death, he added.
The take-home message is that cardiologists and heart surgeons need to carefully assess competing mortality risks before pursuing CABG in older patients, according to Dr. Velazquez.
Session cochair Kim A. Williams, MD, professor and chief of cardiology at Rush University Medical Center in Chicago, posed a direct question: “Is there an age cutoff for your group for bypass surgery?”
No, Dr. Velazquez replied. He pointed out that cardiovascular mortality remained the No. 1 cause of mortality across all age groups.
“If the expectation is that the major cause of fatal events is going to be cardiovascular, and CABG plus medical therapy reduces that risk consistently regardless of age, we think that there really is no particular age cutoff. There is a point at which the noncardiovascular risk predominates, but in the population we studied we did not see that point,” Dr. Velazquez added.
STICH was a 22-nation trial in which 1,212 patients with a left ventricular ejection fraction of 35% or less and coronary artery disease amenable to CABG were randomized to CABG plus optimal medical therapy or optimal medical therapy alone and followed for a median of 9.8 years (JACC Heart Fail. 2013;1[5]:400-8). For purposes of this secondary analysis, participants were divided into quartiles according to baseline age: Quartile 1 patients were up to 54 years old; quartile 2 were ages 55-60; quartile 3 were ages 61-67; and quartile 4 were ages 68 and up.
Older subjects had more comorbidities. All-cause mortality was significantly higher in older than younger patients: for CABG, 68% vs. 48% in quartiles 4 and 1, respectively; for medical therapy, 79% vs. 60% in the same two quartiles. In contrast, cardiovascular mortality did not differ significantly by age: It was 39% in quartile 4 and 35% in quartile 1 in the CABG group, and 53%, compared with 49%, in medically managed patients in quartiles 4 and 1.
For the secondary composite endpoint of all-cause mortality or cardiovascular hospitalization, the benefit of CABG plus medical management over medical management alone was significantly greater in younger than in older patients.
The rate of noncardiovascular mortality was 5.8% in quartiles 1 and 2, then leapt to 14.7% in quartile 3 and 21.1% in quartile 4.
Although the main focus of Dr. Velazquez’s presentation was the impact of CABG with advancing age, he said he found an important lesson in the younger population as well.
“We saw roughly a 40% relative risk reduction in all-cause mortality with CABG in the youngest quartile, compared with the three older groups. My interpretation of that data is that it’s probably not appropriate to avoid CABG in favor of another strategy in a younger patient when you see this kind of mortality benefit,” the cardiologist said.
One limitation of the STICH analysis, said session cochair Stephan Achenbach, MD, is that the study population was relatively young overall. The oldest patients in STICH were roughly the same age as the average patients undergoing CABG for left ventricular systolic dysfunction today at most centers, according to Dr. Achenbach, professor of cardiology at the University of Erlangen-Nuremberg (Germany).
Dr. Velazquez agreed. “I can’t speak as to whether these trial results would apply to the very elderly, patients age 90 and above,” he said.
Simultaneously with the presentation , the new STICH analysis was published online (Circulation. 2016 Aug 29. doi: 10.1161/CIRCULATIONAHA.116.024800).
STICH was funded by the National Institutes of Health. Dr. Velazquez reported having no relevant financial conflicts.
ROME – Coronary artery bypass surgery reduces cardiovascular mortality in heart failure patients with ischemic cardiomyopathy to a consistent extent regardless of age at the time of surgery, according to a secondary analysis from the landmark STICH trial, Eric J. Velazquez, MD, reported at the annual congress of the European Society of Cardiology.
“Cardiologists and cardiac surgeons can confidently offer patients CABG in addition to optimal medical therapy with the knowledge that cardiovascular mortality is reduced by CABG to a similar extent across all age groups in this trial through 10 years of follow-up,” said Dr. Velazquez, professor of medicine at Duke University in Durham, N.C.
However, that’s only part of the story. Cardiovascular mortality was a secondary endpoint in STICH (Surgical Treatment for Ischemic Heart Failure). The primary endpoint was all-cause mortality. And CABG’s impact on all-cause mortality was diminished in older STICH participants because of their greater comorbidity burden and the competing risk of noncardiovascular death, he added.
The take-home message is that cardiologists and heart surgeons need to carefully assess competing mortality risks before pursuing CABG in older patients, according to Dr. Velazquez.
Session cochair Kim A. Williams, MD, professor and chief of cardiology at Rush University Medical Center in Chicago, posed a direct question: “Is there an age cutoff for your group for bypass surgery?”
No, Dr. Velazquez replied. He pointed out that cardiovascular mortality remained the No. 1 cause of mortality across all age groups.
“If the expectation is that the major cause of fatal events is going to be cardiovascular, and CABG plus medical therapy reduces that risk consistently regardless of age, we think that there really is no particular age cutoff. There is a point at which the noncardiovascular risk predominates, but in the population we studied we did not see that point,” Dr. Velazquez added.
STICH was a 22-nation trial in which 1,212 patients with a left ventricular ejection fraction of 35% or less and coronary artery disease amenable to CABG were randomized to CABG plus optimal medical therapy or optimal medical therapy alone and followed for a median of 9.8 years (JACC Heart Fail. 2013;1[5]:400-8). For purposes of this secondary analysis, participants were divided into quartiles according to baseline age: Quartile 1 patients were up to 54 years old; quartile 2 were ages 55-60; quartile 3 were ages 61-67; and quartile 4 were ages 68 and up.
Older subjects had more comorbidities. All-cause mortality was significantly higher in older than younger patients: for CABG, 68% vs. 48% in quartiles 4 and 1, respectively; for medical therapy, 79% vs. 60% in the same two quartiles. In contrast, cardiovascular mortality did not differ significantly by age: It was 39% in quartile 4 and 35% in quartile 1 in the CABG group, and 53%, compared with 49%, in medically managed patients in quartiles 4 and 1.
For the secondary composite endpoint of all-cause mortality or cardiovascular hospitalization, the benefit of CABG plus medical management over medical management alone was significantly greater in younger than in older patients.
The rate of noncardiovascular mortality was 5.8% in quartiles 1 and 2, then leapt to 14.7% in quartile 3 and 21.1% in quartile 4.
Although the main focus of Dr. Velazquez’s presentation was the impact of CABG with advancing age, he said he found an important lesson in the younger population as well.
“We saw roughly a 40% relative risk reduction in all-cause mortality with CABG in the youngest quartile, compared with the three older groups. My interpretation of that data is that it’s probably not appropriate to avoid CABG in favor of another strategy in a younger patient when you see this kind of mortality benefit,” the cardiologist said.
One limitation of the STICH analysis, said session cochair Stephan Achenbach, MD, is that the study population was relatively young overall. The oldest patients in STICH were roughly the same age as the average patients undergoing CABG for left ventricular systolic dysfunction today at most centers, according to Dr. Achenbach, professor of cardiology at the University of Erlangen-Nuremberg (Germany).
Dr. Velazquez agreed. “I can’t speak as to whether these trial results would apply to the very elderly, patients age 90 and above,” he said.
Simultaneously with the presentation , the new STICH analysis was published online (Circulation. 2016 Aug 29. doi: 10.1161/CIRCULATIONAHA.116.024800).
STICH was funded by the National Institutes of Health. Dr. Velazquez reported having no relevant financial conflicts.
ROME – Coronary artery bypass surgery reduces cardiovascular mortality in heart failure patients with ischemic cardiomyopathy to a consistent extent regardless of age at the time of surgery, according to a secondary analysis from the landmark STICH trial, Eric J. Velazquez, MD, reported at the annual congress of the European Society of Cardiology.
“Cardiologists and cardiac surgeons can confidently offer patients CABG in addition to optimal medical therapy with the knowledge that cardiovascular mortality is reduced by CABG to a similar extent across all age groups in this trial through 10 years of follow-up,” said Dr. Velazquez, professor of medicine at Duke University in Durham, N.C.
However, that’s only part of the story. Cardiovascular mortality was a secondary endpoint in STICH (Surgical Treatment for Ischemic Heart Failure). The primary endpoint was all-cause mortality. And CABG’s impact on all-cause mortality was diminished in older STICH participants because of their greater comorbidity burden and the competing risk of noncardiovascular death, he added.
The take-home message is that cardiologists and heart surgeons need to carefully assess competing mortality risks before pursuing CABG in older patients, according to Dr. Velazquez.
Session cochair Kim A. Williams, MD, professor and chief of cardiology at Rush University Medical Center in Chicago, posed a direct question: “Is there an age cutoff for your group for bypass surgery?”
No, Dr. Velazquez replied. He pointed out that cardiovascular mortality remained the No. 1 cause of mortality across all age groups.
“If the expectation is that the major cause of fatal events is going to be cardiovascular, and CABG plus medical therapy reduces that risk consistently regardless of age, we think that there really is no particular age cutoff. There is a point at which the noncardiovascular risk predominates, but in the population we studied we did not see that point,” Dr. Velazquez added.
STICH was a 22-nation trial in which 1,212 patients with a left ventricular ejection fraction of 35% or less and coronary artery disease amenable to CABG were randomized to CABG plus optimal medical therapy or optimal medical therapy alone and followed for a median of 9.8 years (JACC Heart Fail. 2013;1[5]:400-8). For purposes of this secondary analysis, participants were divided into quartiles according to baseline age: Quartile 1 patients were up to 54 years old; quartile 2 were ages 55-60; quartile 3 were ages 61-67; and quartile 4 were ages 68 and up.
Older subjects had more comorbidities. All-cause mortality was significantly higher in older than younger patients: for CABG, 68% vs. 48% in quartiles 4 and 1, respectively; for medical therapy, 79% vs. 60% in the same two quartiles. In contrast, cardiovascular mortality did not differ significantly by age: It was 39% in quartile 4 and 35% in quartile 1 in the CABG group, and 53%, compared with 49%, in medically managed patients in quartiles 4 and 1.
For the secondary composite endpoint of all-cause mortality or cardiovascular hospitalization, the benefit of CABG plus medical management over medical management alone was significantly greater in younger than in older patients.
The rate of noncardiovascular mortality was 5.8% in quartiles 1 and 2, then leapt to 14.7% in quartile 3 and 21.1% in quartile 4.
Although the main focus of Dr. Velazquez’s presentation was the impact of CABG with advancing age, he said he found an important lesson in the younger population as well.
“We saw roughly a 40% relative risk reduction in all-cause mortality with CABG in the youngest quartile, compared with the three older groups. My interpretation of that data is that it’s probably not appropriate to avoid CABG in favor of another strategy in a younger patient when you see this kind of mortality benefit,” the cardiologist said.
One limitation of the STICH analysis, said session cochair Stephan Achenbach, MD, is that the study population was relatively young overall. The oldest patients in STICH were roughly the same age as the average patients undergoing CABG for left ventricular systolic dysfunction today at most centers, according to Dr. Achenbach, professor of cardiology at the University of Erlangen-Nuremberg (Germany).
Dr. Velazquez agreed. “I can’t speak as to whether these trial results would apply to the very elderly, patients age 90 and above,” he said.
Simultaneously with the presentation , the new STICH analysis was published online (Circulation. 2016 Aug 29. doi: 10.1161/CIRCULATIONAHA.116.024800).
STICH was funded by the National Institutes of Health. Dr. Velazquez reported having no relevant financial conflicts.
AT THE ESC CONGRESS 2016
Key clinical point: There should be no age cutoff in offering CABG to older patients with ischemic heart failure.
Major finding: CABG provided an absolute 14.4% reduction in cardiovascular mortality, compared with medical management, in both the youngest and oldest quartiles of patients with heart failure due to ischemic cardiomyopathy.
Data source: A secondary analysis of the STICH trial, in which 1,212 heart failure patients with ischemic cardiomyopathy were randomized to CABG plus medical therapy or medical therapy alone and followed for nearly 10 years.
Disclosures: The study was funded by the National Institutes of Health. The presenter reported having no relevant financial conflicts.
Oral HIV PrEP also protects against herpes
DURBAN, SOUTH AFRICA – Oral antiretroviral pre-exposure prophylaxis (PrEP) against HIV also reduces the risk of acquiring herpes simplex virus type 2, according to research presented at the 21st International AIDS Conference.
“Given the limited interventions for primary prevention of HSV-2, efficacy against HSV-2 provides additional benefit to oral PrEP,” observed Connie Celum, MD, professor of global health and medicine at the University of Washington, Seattle.
HSV-2 is, however, the only non-HIV sexually transmitted infection whose incidence is reduced by PrEP with emtricitabine/tenofovir (Truvada), the sole approved agent for oral HIV PrEP, she added.
Dr. Celum was lead author in a report from the landmark Partners PrEP study of HIV serodiscordant heterosexual Kenyan and Ugandan couples, which demonstrated that oral PrEP provided a 33% reduction in the risk of HSV-2 infection in participants with a known HSV-2-positive partner (Ann Intern Med. 2014 Jul 1;161[1]:11-9. doi: 10.7326/M13-2471).
Tenofovir has been shown to have in vitro activity against HSV-2, providing biologic plausibility to the Partners PrEP study finding, but the 90% effective concentration of the drug required to achieve strong anti-HSV-2 activity in the laboratory makes it likely that good adherence to daily oral PrEP is necessary to see the clinical benefit in terms of reduced HSV-2 acquisition, Dr. Celum said.
She also addressed other questions about the interaction between PrEP and non-HIV STIs she often receives from physicians who provide care for HIV-infected or at-risk patients.
Do STIs reduce the efficacy of oral PrEP? “My answer would be no,” Dr. Celum said. She noted that no difference in PrEP efficacy was seen between patients with and without STIs in Partners PrEP or the French IPERGAY study.
Does PrEP increase the rate of other STIs? The concern here has been that PrEP’s beneficial effect in reducing the risk of HIV infection could be counteracted by a compensatory increase in unsafe sexual practices among PrEP users, with a resultant increase in other STIs. That didn’t occur, however, in the recently reported UK PROUD randomized study of 544 men who have sex with men (MSM), which showed no increase in other STIs with the addition of daily oral PrEP (Lancet. 2016 Jan 2;387[10013]:53-60. doi: 10.1016/S0140-6736[15]00056-2).
Are STIs useful in selecting patients for PrEP by serving as a marker of increased risk for HIV? The answer is a strong yes for MSM. The iPrEX study documented that the number of MSM and transgender women who needed to be treated with PrEP for 1 year to prevent one additional case of HIV infection dropped from 62 for the group as a whole to 36 for those self-reporting condomless receptive anal intercourse and 41 for those with another STI (Lancet Infect Dis. 2014 Jun;14[6]:468-75. doi: 10.1016/S1473-3099[14]70025-8). To cast a wide net for potential beneficiaries, most PrEP programs targeting MSM offer PrEP to those with other STIs or who report engaging in condomless anal sex, Dr. Celum said.
Can PrEP programs reduce STIs through engagement in care? “I think there’s a great opportunity here,” she said. “Most programs are rolling out PrEP with quarterly visits for refills. And I think particularly in MSM and probably in young women, STI testing at those visits provides an opportunity for earlier diagnosis and treatment of STIs as well as for partner notification.”
Dr. Celum reported having no financial conflicts regarding her presentation.
DURBAN, SOUTH AFRICA – Oral antiretroviral pre-exposure prophylaxis (PrEP) against HIV also reduces the risk of acquiring herpes simplex virus type 2, according to research presented at the 21st International AIDS Conference.
“Given the limited interventions for primary prevention of HSV-2, efficacy against HSV-2 provides additional benefit to oral PrEP,” observed Connie Celum, MD, professor of global health and medicine at the University of Washington, Seattle.
HSV-2 is, however, the only non-HIV sexually transmitted infection whose incidence is reduced by PrEP with emtricitabine/tenofovir (Truvada), the sole approved agent for oral HIV PrEP, she added.
Dr. Celum was lead author in a report from the landmark Partners PrEP study of HIV serodiscordant heterosexual Kenyan and Ugandan couples, which demonstrated that oral PrEP provided a 33% reduction in the risk of HSV-2 infection in participants with a known HSV-2-positive partner (Ann Intern Med. 2014 Jul 1;161[1]:11-9. doi: 10.7326/M13-2471).
Tenofovir has been shown to have in vitro activity against HSV-2, providing biologic plausibility to the Partners PrEP study finding, but the 90% effective concentration of the drug required to achieve strong anti-HSV-2 activity in the laboratory makes it likely that good adherence to daily oral PrEP is necessary to see the clinical benefit in terms of reduced HSV-2 acquisition, Dr. Celum said.
She also addressed other questions about the interaction between PrEP and non-HIV STIs she often receives from physicians who provide care for HIV-infected or at-risk patients.
Do STIs reduce the efficacy of oral PrEP? “My answer would be no,” Dr. Celum said. She noted that no difference in PrEP efficacy was seen between patients with and without STIs in Partners PrEP or the French IPERGAY study.
Does PrEP increase the rate of other STIs? The concern here has been that PrEP’s beneficial effect in reducing the risk of HIV infection could be counteracted by a compensatory increase in unsafe sexual practices among PrEP users, with a resultant increase in other STIs. That didn’t occur, however, in the recently reported UK PROUD randomized study of 544 men who have sex with men (MSM), which showed no increase in other STIs with the addition of daily oral PrEP (Lancet. 2016 Jan 2;387[10013]:53-60. doi: 10.1016/S0140-6736[15]00056-2).
Are STIs useful in selecting patients for PrEP by serving as a marker of increased risk for HIV? The answer is a strong yes for MSM. The iPrEX study documented that the number of MSM and transgender women who needed to be treated with PrEP for 1 year to prevent one additional case of HIV infection dropped from 62 for the group as a whole to 36 for those self-reporting condomless receptive anal intercourse and 41 for those with another STI (Lancet Infect Dis. 2014 Jun;14[6]:468-75. doi: 10.1016/S1473-3099[14]70025-8). To cast a wide net for potential beneficiaries, most PrEP programs targeting MSM offer PrEP to those with other STIs or who report engaging in condomless anal sex, Dr. Celum said.
Can PrEP programs reduce STIs through engagement in care? “I think there’s a great opportunity here,” she said. “Most programs are rolling out PrEP with quarterly visits for refills. And I think particularly in MSM and probably in young women, STI testing at those visits provides an opportunity for earlier diagnosis and treatment of STIs as well as for partner notification.”
Dr. Celum reported having no financial conflicts regarding her presentation.
DURBAN, SOUTH AFRICA – Oral antiretroviral pre-exposure prophylaxis (PrEP) against HIV also reduces the risk of acquiring herpes simplex virus type 2, according to research presented at the 21st International AIDS Conference.
“Given the limited interventions for primary prevention of HSV-2, efficacy against HSV-2 provides additional benefit to oral PrEP,” observed Connie Celum, MD, professor of global health and medicine at the University of Washington, Seattle.
HSV-2 is, however, the only non-HIV sexually transmitted infection whose incidence is reduced by PrEP with emtricitabine/tenofovir (Truvada), the sole approved agent for oral HIV PrEP, she added.
Dr. Celum was lead author in a report from the landmark Partners PrEP study of HIV serodiscordant heterosexual Kenyan and Ugandan couples, which demonstrated that oral PrEP provided a 33% reduction in the risk of HSV-2 infection in participants with a known HSV-2-positive partner (Ann Intern Med. 2014 Jul 1;161[1]:11-9. doi: 10.7326/M13-2471).
Tenofovir has been shown to have in vitro activity against HSV-2, providing biologic plausibility to the Partners PrEP study finding, but the 90% effective concentration of the drug required to achieve strong anti-HSV-2 activity in the laboratory makes it likely that good adherence to daily oral PrEP is necessary to see the clinical benefit in terms of reduced HSV-2 acquisition, Dr. Celum said.
She also addressed other questions about the interaction between PrEP and non-HIV STIs she often receives from physicians who provide care for HIV-infected or at-risk patients.
Do STIs reduce the efficacy of oral PrEP? “My answer would be no,” Dr. Celum said. She noted that no difference in PrEP efficacy was seen between patients with and without STIs in Partners PrEP or the French IPERGAY study.
Does PrEP increase the rate of other STIs? The concern here has been that PrEP’s beneficial effect in reducing the risk of HIV infection could be counteracted by a compensatory increase in unsafe sexual practices among PrEP users, with a resultant increase in other STIs. That didn’t occur, however, in the recently reported UK PROUD randomized study of 544 men who have sex with men (MSM), which showed no increase in other STIs with the addition of daily oral PrEP (Lancet. 2016 Jan 2;387[10013]:53-60. doi: 10.1016/S0140-6736[15]00056-2).
Are STIs useful in selecting patients for PrEP by serving as a marker of increased risk for HIV? The answer is a strong yes for MSM. The iPrEX study documented that the number of MSM and transgender women who needed to be treated with PrEP for 1 year to prevent one additional case of HIV infection dropped from 62 for the group as a whole to 36 for those self-reporting condomless receptive anal intercourse and 41 for those with another STI (Lancet Infect Dis. 2014 Jun;14[6]:468-75. doi: 10.1016/S1473-3099[14]70025-8). To cast a wide net for potential beneficiaries, most PrEP programs targeting MSM offer PrEP to those with other STIs or who report engaging in condomless anal sex, Dr. Celum said.
Can PrEP programs reduce STIs through engagement in care? “I think there’s a great opportunity here,” she said. “Most programs are rolling out PrEP with quarterly visits for refills. And I think particularly in MSM and probably in young women, STI testing at those visits provides an opportunity for earlier diagnosis and treatment of STIs as well as for partner notification.”
Dr. Celum reported having no financial conflicts regarding her presentation.
EXPERT ANALYSIS FROM AIDS 2016





















