User login
Lebrikizumab opens new door in atopic dermatitis therapy
VIENNA – The success of the interleukin-13 blocker lebrikizumab in the TREBLE trial provides a new avenue in the treatment of moderate-to-severe atopic dermatitis.
“This is another promising molecule in atopic dermatitis,” Eric L. Simpson, MD, declared in presenting the phase II TREBLE data at the annual congress of the European Academy of Dermatology and Venereology.
Interleukin-13 is known to be an especially potent promoter of type 1, IgE-mediated inflammation. For this reason, lebrikizumab is also under investigation in the treatment of severe asthma, where large phase III trials have been completed, as well as in idiopathic pulmonary fibrosis.
In mouse models of AD, topical anti-IL-13 therapy markedly reduces skin inflammation. This observation helped provide the rationale for investigating lebrikizumab as a novel therapy for AD.
TREBLE was a double-blind, dose-ranging study involving 209 adults with moderate-to-severe AD despite intensive topical corticosteroid therapy. Indeed, enrollment was restricted to patients who still had moderate or severe disease after 2 weeks of triamcinolone 0.1% cream BID. Continuation of that twice-daily topical steroid regimen was mandatory for the full 12-week study period that followed. Patients were randomized 1:1:1:1 to triamcinolone 0.1% BID plus either a single 125-mg subcutaneous dose of lebrikizumab at week 0, a single 250-mg dose of the biologic, 125 mg every 4 weeks, or placebo injections.
At baseline, after their 2 weeks of topical steroid monotherapy, patients had a median Eczema Area and Severity Index (EASI) score of 22 and a median SCORing Atopic Dermatitis (SCORAD) of about 56. They averaged 36 years of age. Slightly over 40% of their body surface area was affected.
The primary endpoint in TREBLE was the percentage of patients who achieved at least a 50% reduction from baseline on the EASI, or EASI 50. A dose-response effect was apparent: the EASI 50 rate was 62.3% in patients on placebo plus daily topical steroids, 69.2% with a single 125-mg dose of lebrikizumab, 69.8% with a single 250-mg dose, and 82.4% with 125 mg of lebrikizumab at weeks 0, 4, 8, and 12. Only the group with monthly dosing of the biologic plus daily triamcinolone 0.1% BID had an EASI 50 response rate significantly better than the controls on placebo plus topical steroid therapy.
A key secondary endpoint was the SCORAD 50 response. The rate was 26.4% in the control group, 34.6% in patients who received one 125-mg dose of lebrikizumab, 47.2% in those who got a single 250-mg dose, and 51% with monthly dosing. The SCORAD 50 rate was significantly greater than placebo in the latter two lebrikizumab arms.
The EASI 75 rate was significantly greater than placebo plus triamcinolone only in the group on monthly lebrikizumab plus topical steroids.
The EASI 50 and 75 response rates in the group on 125 mg of lebrikizumab every 4 weeks was still climbing with no plateau evident when the trial ended at 12 weeks. This suggests greater benefit might be achieved with longer treatment duration and/or a higher dosage, Dr. Simpson observed.
Turning to safety issues, he pointed out that the total number of adverse events and serious adverse events were similar across all four treatment arms. Herpes infections occurred in 2%-6% of patients who received lebrikizumab but in no controls. There was also a nonsignificant trend for more cases of conjunctivitis in the groups that received the biologic.
Audience member Andrew Blauvelt, MD, rose to take issue with the study design.
“This study is almost an advertisement for not using topical steroids in a biologic trial for atopic dermatitis. I think it just confuses everything. It especially doesn’t make sense when one of the inclusion criteria is inadequate control with topical steroids. Shouldn’t we be doing monotherapy trials in the new era of biologics for atopic dermatitis?” commented Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Dr. Simpson replied that when TREBLE was designed, the thinking was that in real-world clinical practice, physicians often prescribe a biologic for AD on top of topical therapy. The goal was to conduct a trial that mimics that situation. That being said, he agreed with Dr. Blauvelt’s critique.
“I think we’re realizing that topical steroids have a very strong effect, especially if you use them all the way through a trial or ad lib,” he said. “Topical steroids bring too many confounders. If you can remove potent topical steroid therapy I think you’re going to have a clearer result.”
“We’re not where psoriasis is,” Dr. Simpson added. “We don’t have any standardized methodologies in atopic dermatitis clinical trials yet, but I think we’re getting there.”
The TREBLE trial was sponsored by Genentech/Roche. Dr. Simpson reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
VIENNA – The success of the interleukin-13 blocker lebrikizumab in the TREBLE trial provides a new avenue in the treatment of moderate-to-severe atopic dermatitis.
“This is another promising molecule in atopic dermatitis,” Eric L. Simpson, MD, declared in presenting the phase II TREBLE data at the annual congress of the European Academy of Dermatology and Venereology.
Interleukin-13 is known to be an especially potent promoter of type 1, IgE-mediated inflammation. For this reason, lebrikizumab is also under investigation in the treatment of severe asthma, where large phase III trials have been completed, as well as in idiopathic pulmonary fibrosis.
In mouse models of AD, topical anti-IL-13 therapy markedly reduces skin inflammation. This observation helped provide the rationale for investigating lebrikizumab as a novel therapy for AD.
TREBLE was a double-blind, dose-ranging study involving 209 adults with moderate-to-severe AD despite intensive topical corticosteroid therapy. Indeed, enrollment was restricted to patients who still had moderate or severe disease after 2 weeks of triamcinolone 0.1% cream BID. Continuation of that twice-daily topical steroid regimen was mandatory for the full 12-week study period that followed. Patients were randomized 1:1:1:1 to triamcinolone 0.1% BID plus either a single 125-mg subcutaneous dose of lebrikizumab at week 0, a single 250-mg dose of the biologic, 125 mg every 4 weeks, or placebo injections.
At baseline, after their 2 weeks of topical steroid monotherapy, patients had a median Eczema Area and Severity Index (EASI) score of 22 and a median SCORing Atopic Dermatitis (SCORAD) of about 56. They averaged 36 years of age. Slightly over 40% of their body surface area was affected.
The primary endpoint in TREBLE was the percentage of patients who achieved at least a 50% reduction from baseline on the EASI, or EASI 50. A dose-response effect was apparent: the EASI 50 rate was 62.3% in patients on placebo plus daily topical steroids, 69.2% with a single 125-mg dose of lebrikizumab, 69.8% with a single 250-mg dose, and 82.4% with 125 mg of lebrikizumab at weeks 0, 4, 8, and 12. Only the group with monthly dosing of the biologic plus daily triamcinolone 0.1% BID had an EASI 50 response rate significantly better than the controls on placebo plus topical steroid therapy.
A key secondary endpoint was the SCORAD 50 response. The rate was 26.4% in the control group, 34.6% in patients who received one 125-mg dose of lebrikizumab, 47.2% in those who got a single 250-mg dose, and 51% with monthly dosing. The SCORAD 50 rate was significantly greater than placebo in the latter two lebrikizumab arms.
The EASI 75 rate was significantly greater than placebo plus triamcinolone only in the group on monthly lebrikizumab plus topical steroids.
The EASI 50 and 75 response rates in the group on 125 mg of lebrikizumab every 4 weeks was still climbing with no plateau evident when the trial ended at 12 weeks. This suggests greater benefit might be achieved with longer treatment duration and/or a higher dosage, Dr. Simpson observed.
Turning to safety issues, he pointed out that the total number of adverse events and serious adverse events were similar across all four treatment arms. Herpes infections occurred in 2%-6% of patients who received lebrikizumab but in no controls. There was also a nonsignificant trend for more cases of conjunctivitis in the groups that received the biologic.
Audience member Andrew Blauvelt, MD, rose to take issue with the study design.
“This study is almost an advertisement for not using topical steroids in a biologic trial for atopic dermatitis. I think it just confuses everything. It especially doesn’t make sense when one of the inclusion criteria is inadequate control with topical steroids. Shouldn’t we be doing monotherapy trials in the new era of biologics for atopic dermatitis?” commented Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Dr. Simpson replied that when TREBLE was designed, the thinking was that in real-world clinical practice, physicians often prescribe a biologic for AD on top of topical therapy. The goal was to conduct a trial that mimics that situation. That being said, he agreed with Dr. Blauvelt’s critique.
“I think we’re realizing that topical steroids have a very strong effect, especially if you use them all the way through a trial or ad lib,” he said. “Topical steroids bring too many confounders. If you can remove potent topical steroid therapy I think you’re going to have a clearer result.”
“We’re not where psoriasis is,” Dr. Simpson added. “We don’t have any standardized methodologies in atopic dermatitis clinical trials yet, but I think we’re getting there.”
The TREBLE trial was sponsored by Genentech/Roche. Dr. Simpson reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
VIENNA – The success of the interleukin-13 blocker lebrikizumab in the TREBLE trial provides a new avenue in the treatment of moderate-to-severe atopic dermatitis.
“This is another promising molecule in atopic dermatitis,” Eric L. Simpson, MD, declared in presenting the phase II TREBLE data at the annual congress of the European Academy of Dermatology and Venereology.
Interleukin-13 is known to be an especially potent promoter of type 1, IgE-mediated inflammation. For this reason, lebrikizumab is also under investigation in the treatment of severe asthma, where large phase III trials have been completed, as well as in idiopathic pulmonary fibrosis.
In mouse models of AD, topical anti-IL-13 therapy markedly reduces skin inflammation. This observation helped provide the rationale for investigating lebrikizumab as a novel therapy for AD.
TREBLE was a double-blind, dose-ranging study involving 209 adults with moderate-to-severe AD despite intensive topical corticosteroid therapy. Indeed, enrollment was restricted to patients who still had moderate or severe disease after 2 weeks of triamcinolone 0.1% cream BID. Continuation of that twice-daily topical steroid regimen was mandatory for the full 12-week study period that followed. Patients were randomized 1:1:1:1 to triamcinolone 0.1% BID plus either a single 125-mg subcutaneous dose of lebrikizumab at week 0, a single 250-mg dose of the biologic, 125 mg every 4 weeks, or placebo injections.
At baseline, after their 2 weeks of topical steroid monotherapy, patients had a median Eczema Area and Severity Index (EASI) score of 22 and a median SCORing Atopic Dermatitis (SCORAD) of about 56. They averaged 36 years of age. Slightly over 40% of their body surface area was affected.
The primary endpoint in TREBLE was the percentage of patients who achieved at least a 50% reduction from baseline on the EASI, or EASI 50. A dose-response effect was apparent: the EASI 50 rate was 62.3% in patients on placebo plus daily topical steroids, 69.2% with a single 125-mg dose of lebrikizumab, 69.8% with a single 250-mg dose, and 82.4% with 125 mg of lebrikizumab at weeks 0, 4, 8, and 12. Only the group with monthly dosing of the biologic plus daily triamcinolone 0.1% BID had an EASI 50 response rate significantly better than the controls on placebo plus topical steroid therapy.
A key secondary endpoint was the SCORAD 50 response. The rate was 26.4% in the control group, 34.6% in patients who received one 125-mg dose of lebrikizumab, 47.2% in those who got a single 250-mg dose, and 51% with monthly dosing. The SCORAD 50 rate was significantly greater than placebo in the latter two lebrikizumab arms.
The EASI 75 rate was significantly greater than placebo plus triamcinolone only in the group on monthly lebrikizumab plus topical steroids.
The EASI 50 and 75 response rates in the group on 125 mg of lebrikizumab every 4 weeks was still climbing with no plateau evident when the trial ended at 12 weeks. This suggests greater benefit might be achieved with longer treatment duration and/or a higher dosage, Dr. Simpson observed.
Turning to safety issues, he pointed out that the total number of adverse events and serious adverse events were similar across all four treatment arms. Herpes infections occurred in 2%-6% of patients who received lebrikizumab but in no controls. There was also a nonsignificant trend for more cases of conjunctivitis in the groups that received the biologic.
Audience member Andrew Blauvelt, MD, rose to take issue with the study design.
“This study is almost an advertisement for not using topical steroids in a biologic trial for atopic dermatitis. I think it just confuses everything. It especially doesn’t make sense when one of the inclusion criteria is inadequate control with topical steroids. Shouldn’t we be doing monotherapy trials in the new era of biologics for atopic dermatitis?” commented Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Dr. Simpson replied that when TREBLE was designed, the thinking was that in real-world clinical practice, physicians often prescribe a biologic for AD on top of topical therapy. The goal was to conduct a trial that mimics that situation. That being said, he agreed with Dr. Blauvelt’s critique.
“I think we’re realizing that topical steroids have a very strong effect, especially if you use them all the way through a trial or ad lib,” he said. “Topical steroids bring too many confounders. If you can remove potent topical steroid therapy I think you’re going to have a clearer result.”
“We’re not where psoriasis is,” Dr. Simpson added. “We don’t have any standardized methodologies in atopic dermatitis clinical trials yet, but I think we’re getting there.”
The TREBLE trial was sponsored by Genentech/Roche. Dr. Simpson reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point:
Major finding: Of atopic dermatitis patients randomized to 125 mg of lebrikizumab every 4 weeks on top of twice-daily potent topical steroids, 82% experienced at least a 50% reduction in Eczema Area and Severity Index scores at 12 weeks, compared with 62% on the same topical steroid regimen plus placebo injections.
Data source: The TREBLE trial was a phase II, double-blind, placebo-controlled, 12-week study including 209 adults with moderate-to-severe atopic dermatitis inadequately controlled by intensive topical steroid therapy.
Disclosures: The study was sponsored by Genentech/Roche. The presenter reported serving as an investigator for and consultant to that organization and numerous other pharmaceutical companies.
Topical anticholinergic for axillary hyperhidrosis hits marks in phase III
VIENNA – An investigational topical cholinergic receptor antagonist known as DRM04 achieved its efficacy and safety endpoints for the treatment of primary axillary hyperhidrosis in the pivotal phase III ATMOS-1 and ATMOS-2 trials.
“We haven’t had any good new treatment options for patients with hyperhidrosis for a long time,” Dr. David M. Pariser said, in presenting the pivotal trial outcomes data at the annual congress of the European Academy of Dermatology and Venereology.
ATMOS-1 and ATMOS-2 were identically designed 4-week, double-blind studies involving 697 patients with excessive underarm sweating who were randomized 2:1 to once daily use of DRM04 3.75% wipes or a vehicle control. The patients averaged 33 years of age, although as Dr. Pariser noted, primary axillary hyperhidrosis often begins in adolescence and patients as young as age 9 were enrolled. The majority of participants were female. Roughly two-thirds of subjects were grade 3 on the 4-point Hyperhidrosis Disease Severity Scale. The rest were grade 4.
The coprimary endpoints in ATMOS-1 and ATMOS-2 were a 4-point or greater improvement on the Axillary Sweating Daily Diary (ASDD) between baseline and week 4, and the absolute change from baseline in axillary sweat production measured gravimetrically.
The ASDD is a new patient-reported outcome measure developed specifically for the ATMOS trials. At baseline, participants scored a mean of 7.2 points on the 0-10 scale. A 4-point or greater improvement was seen at week 4 in 52.8% of ATMOS-1 participants on DRM04, compared with 28.3% of controls. In ATMOS-2, the spread was 66.1% vs. 26.9%.
Mean baseline sweat production was roughly 175 mg/5 min per armpit, a prodigious rate given that 50 mg/5 min is considered excessive. In ATMOS-1, the rate dropped by an adjusted average of 96.2 mg/5 min per armpit with active therapy, compared with 90.6 mg/5 min in the control group. In ATMOS-2, the DRM04 users had a mean drop in sweat production of 110.3 mg/5 min, compared with a reduction of 92.2 mg/5 min in the control group. Both of these differences were statistically significant and clinically meaningful, Dr. Pariser said.
The secondary endpoint in the studies was change in the Dermatology Life Quality Index from baseline to week 4. The improvement in DRM04 users averaged 8.1 points in ATMOS-1 and 8.6 points in ATMOS-2, both significantly greater than the 4.3- and 5.0-point improvements in the control arms.
In ATMOS-1, 9.2% of patients in the DRM04 arm dropped out of the study, in many cases because of anticholinergic side effects, the most common of which included dry mouth, dry eyes, urinary hesitation or less frequently retention, and constipation. These were mostly mild to moderate in nature and were generally responsive to temporary treatment discontinuation, which the study protocol allowed. The dropout rate in the DRM04 arm of ATMOS-2 was 6.8%.
Seven percent of subjects in the DRM04 study arms experienced mydriasis, most often unilaterally. Dr. Pariser said this might be due to patients touching an eye while they still had DRM04 on their hands, a problem that can be readily addressed in the medication use instructions.
The dropout rates in the control groups were 2.6% and 5%.
More than 80% of ATMOS-1 and ATMOS-2 participants enrolled in ARIDO, the 48-week, open label, phase III extension study of DRM04. Dermira, which is developing DRM04, has announced it plans to file for marketing approval by the Food and Drug Administration in the second half of 2017.
Dr. Pariser reported serving as an investigator for and consultant to Dermira, Brickell Biotech, and TheraVida.
VIENNA – An investigational topical cholinergic receptor antagonist known as DRM04 achieved its efficacy and safety endpoints for the treatment of primary axillary hyperhidrosis in the pivotal phase III ATMOS-1 and ATMOS-2 trials.
“We haven’t had any good new treatment options for patients with hyperhidrosis for a long time,” Dr. David M. Pariser said, in presenting the pivotal trial outcomes data at the annual congress of the European Academy of Dermatology and Venereology.
ATMOS-1 and ATMOS-2 were identically designed 4-week, double-blind studies involving 697 patients with excessive underarm sweating who were randomized 2:1 to once daily use of DRM04 3.75% wipes or a vehicle control. The patients averaged 33 years of age, although as Dr. Pariser noted, primary axillary hyperhidrosis often begins in adolescence and patients as young as age 9 were enrolled. The majority of participants were female. Roughly two-thirds of subjects were grade 3 on the 4-point Hyperhidrosis Disease Severity Scale. The rest were grade 4.
The coprimary endpoints in ATMOS-1 and ATMOS-2 were a 4-point or greater improvement on the Axillary Sweating Daily Diary (ASDD) between baseline and week 4, and the absolute change from baseline in axillary sweat production measured gravimetrically.
The ASDD is a new patient-reported outcome measure developed specifically for the ATMOS trials. At baseline, participants scored a mean of 7.2 points on the 0-10 scale. A 4-point or greater improvement was seen at week 4 in 52.8% of ATMOS-1 participants on DRM04, compared with 28.3% of controls. In ATMOS-2, the spread was 66.1% vs. 26.9%.
Mean baseline sweat production was roughly 175 mg/5 min per armpit, a prodigious rate given that 50 mg/5 min is considered excessive. In ATMOS-1, the rate dropped by an adjusted average of 96.2 mg/5 min per armpit with active therapy, compared with 90.6 mg/5 min in the control group. In ATMOS-2, the DRM04 users had a mean drop in sweat production of 110.3 mg/5 min, compared with a reduction of 92.2 mg/5 min in the control group. Both of these differences were statistically significant and clinically meaningful, Dr. Pariser said.
The secondary endpoint in the studies was change in the Dermatology Life Quality Index from baseline to week 4. The improvement in DRM04 users averaged 8.1 points in ATMOS-1 and 8.6 points in ATMOS-2, both significantly greater than the 4.3- and 5.0-point improvements in the control arms.
In ATMOS-1, 9.2% of patients in the DRM04 arm dropped out of the study, in many cases because of anticholinergic side effects, the most common of which included dry mouth, dry eyes, urinary hesitation or less frequently retention, and constipation. These were mostly mild to moderate in nature and were generally responsive to temporary treatment discontinuation, which the study protocol allowed. The dropout rate in the DRM04 arm of ATMOS-2 was 6.8%.
Seven percent of subjects in the DRM04 study arms experienced mydriasis, most often unilaterally. Dr. Pariser said this might be due to patients touching an eye while they still had DRM04 on their hands, a problem that can be readily addressed in the medication use instructions.
The dropout rates in the control groups were 2.6% and 5%.
More than 80% of ATMOS-1 and ATMOS-2 participants enrolled in ARIDO, the 48-week, open label, phase III extension study of DRM04. Dermira, which is developing DRM04, has announced it plans to file for marketing approval by the Food and Drug Administration in the second half of 2017.
Dr. Pariser reported serving as an investigator for and consultant to Dermira, Brickell Biotech, and TheraVida.
VIENNA – An investigational topical cholinergic receptor antagonist known as DRM04 achieved its efficacy and safety endpoints for the treatment of primary axillary hyperhidrosis in the pivotal phase III ATMOS-1 and ATMOS-2 trials.
“We haven’t had any good new treatment options for patients with hyperhidrosis for a long time,” Dr. David M. Pariser said, in presenting the pivotal trial outcomes data at the annual congress of the European Academy of Dermatology and Venereology.
ATMOS-1 and ATMOS-2 were identically designed 4-week, double-blind studies involving 697 patients with excessive underarm sweating who were randomized 2:1 to once daily use of DRM04 3.75% wipes or a vehicle control. The patients averaged 33 years of age, although as Dr. Pariser noted, primary axillary hyperhidrosis often begins in adolescence and patients as young as age 9 were enrolled. The majority of participants were female. Roughly two-thirds of subjects were grade 3 on the 4-point Hyperhidrosis Disease Severity Scale. The rest were grade 4.
The coprimary endpoints in ATMOS-1 and ATMOS-2 were a 4-point or greater improvement on the Axillary Sweating Daily Diary (ASDD) between baseline and week 4, and the absolute change from baseline in axillary sweat production measured gravimetrically.
The ASDD is a new patient-reported outcome measure developed specifically for the ATMOS trials. At baseline, participants scored a mean of 7.2 points on the 0-10 scale. A 4-point or greater improvement was seen at week 4 in 52.8% of ATMOS-1 participants on DRM04, compared with 28.3% of controls. In ATMOS-2, the spread was 66.1% vs. 26.9%.
Mean baseline sweat production was roughly 175 mg/5 min per armpit, a prodigious rate given that 50 mg/5 min is considered excessive. In ATMOS-1, the rate dropped by an adjusted average of 96.2 mg/5 min per armpit with active therapy, compared with 90.6 mg/5 min in the control group. In ATMOS-2, the DRM04 users had a mean drop in sweat production of 110.3 mg/5 min, compared with a reduction of 92.2 mg/5 min in the control group. Both of these differences were statistically significant and clinically meaningful, Dr. Pariser said.
The secondary endpoint in the studies was change in the Dermatology Life Quality Index from baseline to week 4. The improvement in DRM04 users averaged 8.1 points in ATMOS-1 and 8.6 points in ATMOS-2, both significantly greater than the 4.3- and 5.0-point improvements in the control arms.
In ATMOS-1, 9.2% of patients in the DRM04 arm dropped out of the study, in many cases because of anticholinergic side effects, the most common of which included dry mouth, dry eyes, urinary hesitation or less frequently retention, and constipation. These were mostly mild to moderate in nature and were generally responsive to temporary treatment discontinuation, which the study protocol allowed. The dropout rate in the DRM04 arm of ATMOS-2 was 6.8%.
Seven percent of subjects in the DRM04 study arms experienced mydriasis, most often unilaterally. Dr. Pariser said this might be due to patients touching an eye while they still had DRM04 on their hands, a problem that can be readily addressed in the medication use instructions.
The dropout rates in the control groups were 2.6% and 5%.
More than 80% of ATMOS-1 and ATMOS-2 participants enrolled in ARIDO, the 48-week, open label, phase III extension study of DRM04. Dermira, which is developing DRM04, has announced it plans to file for marketing approval by the Food and Drug Administration in the second half of 2017.
Dr. Pariser reported serving as an investigator for and consultant to Dermira, Brickell Biotech, and TheraVida.
AT THE EADV CONGRESS
Key clinical point: A once-daily topical anticholinergic agent called DRM04 achieved positive outcomes for the treatment of primary axillary hyperhidrosis in two pivotal phase III trials.
Major finding: 53% and 66% of subjects on DRM04 in two large studies achieved clinically meaningful improvement in axillary sweating, compared with 28% and 27%, respectively, of controls.
Data source: Based on findings from ATMOS-1 and ATMOS-2, identically designed, 4-week, double-blind, vehicle-controlled clinical trials including a total of 687 patients with primary axillary hyperhidrosis.
Disclosures: The studies were funded by Dermira. The presenter reported serving as an investigator for and consultant to the company.
Guselkumab achieves highest-ever response rates in psoriasis
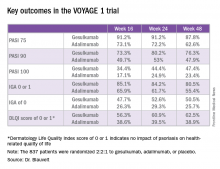
VIENNA – The investigational interleukin-23 inhibitor guselkumab decisively outperformed adalimumab in a head-to-head comparison for treatment of moderate or severe plaque psoriasis in the pivotal VOYAGE 1 study, Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
VOYAGE 1 was a 48-week, multicenter, international phase III trial in which 837 patients were randomized 2:2:1 to guselkumab, adalimumab (Humira), or placebo, with the placebo group switched to guselkumab at 16 weeks. Roughly three-quarters of patients had moderate psoriasis, the rest had severe disease. One in five had previously been treated with biologic agents; the only biologic disallowed was adalimumab.
The primary endpoints required by regulatory agencies involved efficacy comparisons between guselkumab and placebo at 16 weeks. Those results were a foregone conclusion. Far more arresting were the prespecified secondary endpoints comparing guselkumab to adalimumab at 24 and 48 weeks.
“These are very exciting results. We’re seeing efficacy in this trial that has not ever been seen before in a phase III study,” said Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Take, for example, an efficacy yardstick dermatologists are quite familiar with: the PASI 75 response, defined as at least a 75% improvement from baseline in the Psoriasis Area Severity Index score, which averaged 22 at baseline in this trial. The PASI 75 rate in guselkumab-treated patients was 91.2% at 16 weeks, remained at 91.2% at 24 weeks, and was 87.8% at week 48.
“To my knowledge this is the highest PASI 75 response rate that’s been seen in a phase III study of any biologic in psoriasis,” the dermatologist said.
The PASI 75 rates with adalimumab, a tumor necrosis factor–alpha blocker widely prescribed for psoriasis, were markedly lower, although just a few years ago they would have been considered stratospheric: 73.1% at 16 weeks, 72.2% at 24 weeks, and 62.6% at 48 weeks.
The same pattern held for PASI 90, PASI 100, Investigator’s Global Assessment (IGA), and quality-of-life measures.
“There is a clear early separation of guselkumab from adalimumab, sustained over time, curves staying flat, responses not dropping off,” Dr. Blauvelt said in summary.
Guselkumab was dosed at 100 mg subcutaneously at weeks 0 and 4, then every 8 weeks thereafter. Adalimumab was dosed subcutaneously at 80 mg at week 0, 40 mg at week 1, and then 40 mg every other week.
The two coprimary outcomes at week 16 in VOYAGE 1 were the guselkumab and placebo groups’ rates of clear or almost clear skin as defined by an IGA score of 0 or 1, and their PASI 90 response rates. An IGA of 0 or 1 was achieved by 85.1% of the guselkumab group compared with 6.9% on placebo. The week-16 PASI 90 rates – a “high bar” Dr. Blauvelt noted – were 73.3% and 2.9%, respectively.
“Clearly we’re now in an era where PASI 90 is the new PASI 75,” said session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
Guselkumab is a human monoclonal antibody directed at the p-19 subunit of interleukin-23, thereby preventing the inflammatory cytokine from binding to its receptor. In contrast, ustekinumab (Stelara) blocks both IL-23 and IL-12. Given that ustekinumab has established an excellent long-term safety record in PSOLAR, the Psoriasis Longitudinal Assessment and Registry, it stands to reason that guselkumab should have a favorable safety profile, too, since it targets only one of the two cytokines (J Drugs Dermatol. 2015 Jul;14[7]:706-14). And this indeed proved to be the case through 48 weeks in VOYAGE 1, according to Dr. Blauvelt.
Infections treated with antibiotics occurred in 6.1% of the guselkumab group, 7.2% of patients on adalimumab, and 7.5% on placebo. Mild to moderate injection site reactions occurred in 2.4% of patients on guselkumab and 7.5% on adalimumab. One patient on each of the biologics experienced an acute MI. Two malignancies occurred, both in the guselkumab group. One was prostate cancer, the other was a case of male breast cancer in a patient with a breast mass present at enrollment.
Results of two additional pivotal phase III trials, VOYAGE 2 and NAVIGATE, will be presented at future meetings. NAVIGATE is looking specifically at guselkumab’s performance in psoriasis patients with an inadequate response to ustekinumab.
“Those results look promising. It appears that patients who didn’t clear adequately on ustekinumab do well on guselkumab,” Dr. Blauvelt said in response to an audience question.
A phase II study of guselkumab in treating moderate to severe psoriatic arthritis is ongoing.
VOYAGE 1 was funded by Janssen, which is developing guselkumab. Dr. Blauvelt reported receiving research grants from and serving as a scientific consultant to Janssen and numerous other pharmaceutical companies.
VIENNA – The investigational interleukin-23 inhibitor guselkumab decisively outperformed adalimumab in a head-to-head comparison for treatment of moderate or severe plaque psoriasis in the pivotal VOYAGE 1 study, Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
VOYAGE 1 was a 48-week, multicenter, international phase III trial in which 837 patients were randomized 2:2:1 to guselkumab, adalimumab (Humira), or placebo, with the placebo group switched to guselkumab at 16 weeks. Roughly three-quarters of patients had moderate psoriasis, the rest had severe disease. One in five had previously been treated with biologic agents; the only biologic disallowed was adalimumab.
The primary endpoints required by regulatory agencies involved efficacy comparisons between guselkumab and placebo at 16 weeks. Those results were a foregone conclusion. Far more arresting were the prespecified secondary endpoints comparing guselkumab to adalimumab at 24 and 48 weeks.
“These are very exciting results. We’re seeing efficacy in this trial that has not ever been seen before in a phase III study,” said Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Take, for example, an efficacy yardstick dermatologists are quite familiar with: the PASI 75 response, defined as at least a 75% improvement from baseline in the Psoriasis Area Severity Index score, which averaged 22 at baseline in this trial. The PASI 75 rate in guselkumab-treated patients was 91.2% at 16 weeks, remained at 91.2% at 24 weeks, and was 87.8% at week 48.
“To my knowledge this is the highest PASI 75 response rate that’s been seen in a phase III study of any biologic in psoriasis,” the dermatologist said.
The PASI 75 rates with adalimumab, a tumor necrosis factor–alpha blocker widely prescribed for psoriasis, were markedly lower, although just a few years ago they would have been considered stratospheric: 73.1% at 16 weeks, 72.2% at 24 weeks, and 62.6% at 48 weeks.
The same pattern held for PASI 90, PASI 100, Investigator’s Global Assessment (IGA), and quality-of-life measures.
“There is a clear early separation of guselkumab from adalimumab, sustained over time, curves staying flat, responses not dropping off,” Dr. Blauvelt said in summary.
Guselkumab was dosed at 100 mg subcutaneously at weeks 0 and 4, then every 8 weeks thereafter. Adalimumab was dosed subcutaneously at 80 mg at week 0, 40 mg at week 1, and then 40 mg every other week.
The two coprimary outcomes at week 16 in VOYAGE 1 were the guselkumab and placebo groups’ rates of clear or almost clear skin as defined by an IGA score of 0 or 1, and their PASI 90 response rates. An IGA of 0 or 1 was achieved by 85.1% of the guselkumab group compared with 6.9% on placebo. The week-16 PASI 90 rates – a “high bar” Dr. Blauvelt noted – were 73.3% and 2.9%, respectively.
“Clearly we’re now in an era where PASI 90 is the new PASI 75,” said session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
Guselkumab is a human monoclonal antibody directed at the p-19 subunit of interleukin-23, thereby preventing the inflammatory cytokine from binding to its receptor. In contrast, ustekinumab (Stelara) blocks both IL-23 and IL-12. Given that ustekinumab has established an excellent long-term safety record in PSOLAR, the Psoriasis Longitudinal Assessment and Registry, it stands to reason that guselkumab should have a favorable safety profile, too, since it targets only one of the two cytokines (J Drugs Dermatol. 2015 Jul;14[7]:706-14). And this indeed proved to be the case through 48 weeks in VOYAGE 1, according to Dr. Blauvelt.
Infections treated with antibiotics occurred in 6.1% of the guselkumab group, 7.2% of patients on adalimumab, and 7.5% on placebo. Mild to moderate injection site reactions occurred in 2.4% of patients on guselkumab and 7.5% on adalimumab. One patient on each of the biologics experienced an acute MI. Two malignancies occurred, both in the guselkumab group. One was prostate cancer, the other was a case of male breast cancer in a patient with a breast mass present at enrollment.
Results of two additional pivotal phase III trials, VOYAGE 2 and NAVIGATE, will be presented at future meetings. NAVIGATE is looking specifically at guselkumab’s performance in psoriasis patients with an inadequate response to ustekinumab.
“Those results look promising. It appears that patients who didn’t clear adequately on ustekinumab do well on guselkumab,” Dr. Blauvelt said in response to an audience question.
A phase II study of guselkumab in treating moderate to severe psoriatic arthritis is ongoing.
VOYAGE 1 was funded by Janssen, which is developing guselkumab. Dr. Blauvelt reported receiving research grants from and serving as a scientific consultant to Janssen and numerous other pharmaceutical companies.
VIENNA – The investigational interleukin-23 inhibitor guselkumab decisively outperformed adalimumab in a head-to-head comparison for treatment of moderate or severe plaque psoriasis in the pivotal VOYAGE 1 study, Andrew Blauvelt, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
VOYAGE 1 was a 48-week, multicenter, international phase III trial in which 837 patients were randomized 2:2:1 to guselkumab, adalimumab (Humira), or placebo, with the placebo group switched to guselkumab at 16 weeks. Roughly three-quarters of patients had moderate psoriasis, the rest had severe disease. One in five had previously been treated with biologic agents; the only biologic disallowed was adalimumab.
The primary endpoints required by regulatory agencies involved efficacy comparisons between guselkumab and placebo at 16 weeks. Those results were a foregone conclusion. Far more arresting were the prespecified secondary endpoints comparing guselkumab to adalimumab at 24 and 48 weeks.
“These are very exciting results. We’re seeing efficacy in this trial that has not ever been seen before in a phase III study,” said Dr. Blauvelt, president of the Oregon Medical Research Center in Portland.
Take, for example, an efficacy yardstick dermatologists are quite familiar with: the PASI 75 response, defined as at least a 75% improvement from baseline in the Psoriasis Area Severity Index score, which averaged 22 at baseline in this trial. The PASI 75 rate in guselkumab-treated patients was 91.2% at 16 weeks, remained at 91.2% at 24 weeks, and was 87.8% at week 48.
“To my knowledge this is the highest PASI 75 response rate that’s been seen in a phase III study of any biologic in psoriasis,” the dermatologist said.
The PASI 75 rates with adalimumab, a tumor necrosis factor–alpha blocker widely prescribed for psoriasis, were markedly lower, although just a few years ago they would have been considered stratospheric: 73.1% at 16 weeks, 72.2% at 24 weeks, and 62.6% at 48 weeks.
The same pattern held for PASI 90, PASI 100, Investigator’s Global Assessment (IGA), and quality-of-life measures.
“There is a clear early separation of guselkumab from adalimumab, sustained over time, curves staying flat, responses not dropping off,” Dr. Blauvelt said in summary.
Guselkumab was dosed at 100 mg subcutaneously at weeks 0 and 4, then every 8 weeks thereafter. Adalimumab was dosed subcutaneously at 80 mg at week 0, 40 mg at week 1, and then 40 mg every other week.
The two coprimary outcomes at week 16 in VOYAGE 1 were the guselkumab and placebo groups’ rates of clear or almost clear skin as defined by an IGA score of 0 or 1, and their PASI 90 response rates. An IGA of 0 or 1 was achieved by 85.1% of the guselkumab group compared with 6.9% on placebo. The week-16 PASI 90 rates – a “high bar” Dr. Blauvelt noted – were 73.3% and 2.9%, respectively.
“Clearly we’re now in an era where PASI 90 is the new PASI 75,” said session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
Guselkumab is a human monoclonal antibody directed at the p-19 subunit of interleukin-23, thereby preventing the inflammatory cytokine from binding to its receptor. In contrast, ustekinumab (Stelara) blocks both IL-23 and IL-12. Given that ustekinumab has established an excellent long-term safety record in PSOLAR, the Psoriasis Longitudinal Assessment and Registry, it stands to reason that guselkumab should have a favorable safety profile, too, since it targets only one of the two cytokines (J Drugs Dermatol. 2015 Jul;14[7]:706-14). And this indeed proved to be the case through 48 weeks in VOYAGE 1, according to Dr. Blauvelt.
Infections treated with antibiotics occurred in 6.1% of the guselkumab group, 7.2% of patients on adalimumab, and 7.5% on placebo. Mild to moderate injection site reactions occurred in 2.4% of patients on guselkumab and 7.5% on adalimumab. One patient on each of the biologics experienced an acute MI. Two malignancies occurred, both in the guselkumab group. One was prostate cancer, the other was a case of male breast cancer in a patient with a breast mass present at enrollment.
Results of two additional pivotal phase III trials, VOYAGE 2 and NAVIGATE, will be presented at future meetings. NAVIGATE is looking specifically at guselkumab’s performance in psoriasis patients with an inadequate response to ustekinumab.
“Those results look promising. It appears that patients who didn’t clear adequately on ustekinumab do well on guselkumab,” Dr. Blauvelt said in response to an audience question.
A phase II study of guselkumab in treating moderate to severe psoriatic arthritis is ongoing.
VOYAGE 1 was funded by Janssen, which is developing guselkumab. Dr. Blauvelt reported receiving research grants from and serving as a scientific consultant to Janssen and numerous other pharmaceutical companies.
Key clinical point:
Major finding: The PASI 90 response rate at 24 weeks was 80% in psoriasis patients on guselkumab compared with 53% in those on adalimumab.
Data source: A randomized, multinational, 48-week, pivotal phase III clinical trial involving 837 psoriasis patients assigned to guselkumab, adalimumab, or placebo.
Disclosures: The VOYAGE 1 trial was funded by Janssen, which is developing guselkumab. The study presenter reported receiving research grants from and serving as a scientific consultant to Janssen and numerous other pharmaceutical companies.
Birth outcomes unaffected by paternal immunosuppressive therapy
VIENNA – The use of classic systemic immunosuppressive agents by men in the months shortly before conception was not associated with increased risk of low birthweight, preterm birth, or congenital anomalies in their offspring in a large Danish national registry.
“We didn’t see any real safety signals,” Dr. Alexander Egeberg reported at the annual congress of the European Academy of Dermatology and Venereology.
He and his coinvestigators at the University of Copenhagen decided to examine this issue for a simple reason: “We know quite a lot from registry studies about the safety of these drugs when used by women during pregnancy, but very little about the safety of paternal use,” Dr. Egeberg explained.
Methotrexate, azathioprine, and cyclosporine are often prescribed for patients with moderate to severe psoriasis and psoriatic arthritis as well as other chronic inflammatory disorders. Female patients are typically told to stop using these medications if they’re trying to become pregnant, or as soon as they think they might be pregnant, but nearly half of all pregnancies are unintended.
Using linked comprehensive national Danish databases, the investigators scrutinized the medical records of all children born in Denmark during 2004-2010, as well as those of their parents. They identified 2,235 children whose fathers had been on immunosuppressive therapy for a medical condition at any time prior to conception. There were 1,246 fathers who had been on azathioprine, 848 on methotrexate, and 141 on cyclosporine.
Rates of preterm birth, congenital anomalies, and low birthweight were compared in children born to fathers using immunosuppression and in 415,589 children born to fathers with no history of exposure to the medications. These comparisons entailed multivariate regression analyses adjusted for maternal age, parity, smoking status, and the child’s gender. Dr. Egeberg and his colleagues also compared rates of these reproductive complications in the subgroup of children whose fathers had been on the medications within 3 months prior to the estimated time of conception and in children whose fathers had stopped taking the drugs by that point.
None of the adverse neonatal outcomes were significantly increased in ever or recent paternal users of the medications under study, with one exception. Paternal use of cyclosporine within the last 3 months prior to conception was associated with an adjusted 3.7-fold increased likelihood of having a baby with a congenital anomaly. Dr. Egeberg, however, was quick to state that this finding was based on small numbers of exposures: 18 paternal exposures and four affected offspring.
“The cyclosporine finding should be interpreted quite cautiously,” he emphasized.
The reproductive outcomes study was supported by Danish governmental research funds. Dr. Egeberg reported having received research funding from and serving as a consultant to Pfizer and Eli Lilly.
VIENNA – The use of classic systemic immunosuppressive agents by men in the months shortly before conception was not associated with increased risk of low birthweight, preterm birth, or congenital anomalies in their offspring in a large Danish national registry.
“We didn’t see any real safety signals,” Dr. Alexander Egeberg reported at the annual congress of the European Academy of Dermatology and Venereology.
He and his coinvestigators at the University of Copenhagen decided to examine this issue for a simple reason: “We know quite a lot from registry studies about the safety of these drugs when used by women during pregnancy, but very little about the safety of paternal use,” Dr. Egeberg explained.
Methotrexate, azathioprine, and cyclosporine are often prescribed for patients with moderate to severe psoriasis and psoriatic arthritis as well as other chronic inflammatory disorders. Female patients are typically told to stop using these medications if they’re trying to become pregnant, or as soon as they think they might be pregnant, but nearly half of all pregnancies are unintended.
Using linked comprehensive national Danish databases, the investigators scrutinized the medical records of all children born in Denmark during 2004-2010, as well as those of their parents. They identified 2,235 children whose fathers had been on immunosuppressive therapy for a medical condition at any time prior to conception. There were 1,246 fathers who had been on azathioprine, 848 on methotrexate, and 141 on cyclosporine.
Rates of preterm birth, congenital anomalies, and low birthweight were compared in children born to fathers using immunosuppression and in 415,589 children born to fathers with no history of exposure to the medications. These comparisons entailed multivariate regression analyses adjusted for maternal age, parity, smoking status, and the child’s gender. Dr. Egeberg and his colleagues also compared rates of these reproductive complications in the subgroup of children whose fathers had been on the medications within 3 months prior to the estimated time of conception and in children whose fathers had stopped taking the drugs by that point.
None of the adverse neonatal outcomes were significantly increased in ever or recent paternal users of the medications under study, with one exception. Paternal use of cyclosporine within the last 3 months prior to conception was associated with an adjusted 3.7-fold increased likelihood of having a baby with a congenital anomaly. Dr. Egeberg, however, was quick to state that this finding was based on small numbers of exposures: 18 paternal exposures and four affected offspring.
“The cyclosporine finding should be interpreted quite cautiously,” he emphasized.
The reproductive outcomes study was supported by Danish governmental research funds. Dr. Egeberg reported having received research funding from and serving as a consultant to Pfizer and Eli Lilly.
VIENNA – The use of classic systemic immunosuppressive agents by men in the months shortly before conception was not associated with increased risk of low birthweight, preterm birth, or congenital anomalies in their offspring in a large Danish national registry.
“We didn’t see any real safety signals,” Dr. Alexander Egeberg reported at the annual congress of the European Academy of Dermatology and Venereology.
He and his coinvestigators at the University of Copenhagen decided to examine this issue for a simple reason: “We know quite a lot from registry studies about the safety of these drugs when used by women during pregnancy, but very little about the safety of paternal use,” Dr. Egeberg explained.
Methotrexate, azathioprine, and cyclosporine are often prescribed for patients with moderate to severe psoriasis and psoriatic arthritis as well as other chronic inflammatory disorders. Female patients are typically told to stop using these medications if they’re trying to become pregnant, or as soon as they think they might be pregnant, but nearly half of all pregnancies are unintended.
Using linked comprehensive national Danish databases, the investigators scrutinized the medical records of all children born in Denmark during 2004-2010, as well as those of their parents. They identified 2,235 children whose fathers had been on immunosuppressive therapy for a medical condition at any time prior to conception. There were 1,246 fathers who had been on azathioprine, 848 on methotrexate, and 141 on cyclosporine.
Rates of preterm birth, congenital anomalies, and low birthweight were compared in children born to fathers using immunosuppression and in 415,589 children born to fathers with no history of exposure to the medications. These comparisons entailed multivariate regression analyses adjusted for maternal age, parity, smoking status, and the child’s gender. Dr. Egeberg and his colleagues also compared rates of these reproductive complications in the subgroup of children whose fathers had been on the medications within 3 months prior to the estimated time of conception and in children whose fathers had stopped taking the drugs by that point.
None of the adverse neonatal outcomes were significantly increased in ever or recent paternal users of the medications under study, with one exception. Paternal use of cyclosporine within the last 3 months prior to conception was associated with an adjusted 3.7-fold increased likelihood of having a baby with a congenital anomaly. Dr. Egeberg, however, was quick to state that this finding was based on small numbers of exposures: 18 paternal exposures and four affected offspring.
“The cyclosporine finding should be interpreted quite cautiously,” he emphasized.
The reproductive outcomes study was supported by Danish governmental research funds. Dr. Egeberg reported having received research funding from and serving as a consultant to Pfizer and Eli Lilly.
AT THE EADV CONGRESS
Key clinical point:
Major finding: Adjusted rates of congenital anomalies, preterm birth, and low birthweight are not increased in children with paternal use of azathioprine, methotrexate, or cyclosporine prior to conception.
Data source: This retrospective study utilized linked Danish national registries to compare rates of low birthweight, congenital anomalies, and preterm birth in all Danish children born in 2004-2010 depending upon whether or not the father had been on methotrexate, azathioprine, or cyclosporine prior to the pregnancy.
Disclosures: The study was supported by Danish governmental research funds. Dr. Egeberg reported having received research funding from, and serving as a consultant to, Pfizer and Eli Lilly.
Cariprazine shows efficacy for schizophrenia’s negative symptoms
VIENNA – Cariprazine appears to improve predominant negative symptoms of schizophrenia as well as functional status in affected patients, István Bitter, MD, PhD, DSci reported at the annual congress of the European College of Neuropsychopharmacology.
Cariprazine, a dopamine D3/D2 receptor partial agonist, was approved in the United States in 2015 as Vraylar for adults with bipolar disorder or schizophrenia. The drug remains investigational in Europe.
“There is very clearly an unmet need for the treatment of deficit schizophrenia, or schizophrenia with predominant and persistent negative symptoms,” the psychiatrist observed.
The strong efficacy signal for cariprazine in patients with predominant negative symptoms was identified in a post hoc analysis of a randomized, double-blind, phase III head-to-head comparison of cariprazine versus risperidone (Risperdal) in 461 affected adults. Dr. Bitter was a coinvestigator in the 26-week multicenter European study, which was preceded by a 4-week lead-in phase in which participants were uptitrated to a target dose of cariprazine at 4.5 mg/day or risperidone at 4 mg/day.
The primary endpoint in the post hoc analysis was change from baseline on the Positive and Negative Symptoms Factor Score for Negative Symptoms (PANSS-FSNS). A significant difference in favor of cariprazine was seen by week 14, and the gap steadily enlarged thereafter through the remainder of the study. The difference was significant in five of the seven items on the PANSS-FSNS: blunted affect, emotional withdrawal, poor rapport, active social avoidance, and passive/apathetic social withdrawal. Cariprazine also outperformed risperidone on the other two elements of the scale – motor retardation, and lack of spontaneity and flow of conversation – but in those domains, the difference didn’t reach statistical significance.
Patients randomized to cariprazine also fared well on secondary endpoints. They displayed a significantly greater overall improvement in Marder factor scores over 26 weeks than the risperidone-treated group. This advantage was driven by a strong, statistically significant difference in the domain of difficulty in abstract thinking. However, the cariprazine group also showed nonsignificant trends for greater improvement in conceptual disorganization, mannerisms and posturing, disorientation, poor attention, and disturbed volition.
As evidence that cariprazine was exerting specific benefit on negative symptoms, Dr. Bitter cited the fact that the drug showed no significant difference from risperidone in change from baseline on the Marder factor scores for uncontrolled hostility/excitement and anxiety/depression, nor in the Calgary Depression Rating Scale for Schizophrenia.
Regulatory agencies in the United States and Europe have made it clear that, for a drug to obtain an indication for treatment of predominant negative symptoms of schizophrenia, it also has to show evidence of improved patient social function. The cariprazine group showed a significantly greater improvement on the PANSS-FSNS Personal and Social Performance Score. This advantage over risperidone-treated patients reached statistical significance at week 10 and broadened thereafter until study conclusion. Improvements in the subdomain scores for self-care, socially useful activities, and personal and social relationships led the way.
These positive results for cariprazine represent a sharp departure from the psychiatric field’s long-standing history of failed therapeutic attempts to improve negative symptoms. A comprehensive 2015 meta-analysis of all 168 randomized, placebo-controlled studies of various potential treatments for predominant negative symptoms of schizophrenia published through 2013 concluded that none of them provided evidence of clinically meaningful improvement (Schizophr Bull. 2015 Jul;41[4]:892-9).
Dr. Bitter observed that, since that discouraging meta-analysis, several additional novel agents for treatment of predominant negative symptoms of schizophrenia that showed “fantastic” promise in phase II studies subsequently went down in flames in advanced phase III trials. Among those were D-serine as an add-on to second-generation antipsychotics; encenicline, an alpha-7 nicotinic acetylcholine receptor antagonist; pomaglumetad methionil, a selective agonist for glutamate receptor subtypes mGluR2 and mGluR3; and bitopertin, a glycine reuptake inhibitor.
“This is basically a very negative message, that there is not much we can do about negative symptoms. But depending on who you ask, you can get a little bit more optimistic picture,” Dr. Bitter said.
That’s because there are considerable differences among the studies both in how negative symptoms are defined as well as in the measurement tools employed. For example, the Scale for Assessment of Negative Symptoms (SANS) includes items which aren’t strictly speaking part of the negative symptoms concept, so it yields skewed results. The Brief Psychiatric Rating Scale is a relic that has been abandoned by younger psychiatrists. So at present, the PANS-FSNS is the best available tool, and a reasonable common sense definition of predominant negative symptoms of schizophrenia is that, in an affected patient, the PANSS negative subscale score is higher than the positive one, he continued.
It’s noteworthy that none of the multitude of failed drugs for negative symptoms of schizophrenia target activity of dopamine-3 receptors. But cariprazine does.
“There is hope that dopamine-3 receptors might play a role in schizophrenia, especially in negative symptoms. According to experts, they are much older by a couple of million years than the D2 receptors. They don’t operate like D2. In fact, their function is not very clear. In animal studies, though, they help with cognition,” Dr. Bitter said.
“These cariprazine results open the door to some hope for the future,” commented Dr. Ferrara, professor of psychiatry at the University of Naples, Italy.
Even so, she added in an interview, most clinicians aren’t ready to handle a drug with an indication for treatment of negative symptoms. “We are not ready as clinicians to rate negative symptoms in our patients, or in many, many cases to even recognize them. In my opinion, several promising drugs failed because of the fact that the evaluations were not very well carried out in spite of training, but also because, so far, the assessment instruments have not been the best you could think of. Clinicians need training, and the assessment instruments need to be refined. We need instruments that provide more in-depth evaluation of the different aspects of negative symptoms and that are also more appealing to clinicians,” according to the psychiatrist.
She agreed with Dr. Bitter that, for now, the PANSS-FSNS is the best available assessment tool.
“The SANS is not a good tool for negative symptoms. We are all sure about that. We can and should do better,” Dr. Ferrara said.
The cariprazine study was funded by Gedeon Richter and Allergen. Dr. Bitter reported serving as an advisory board member and/or consultant to Gedeon Richter and eight other pharmaceutical companies.
VIENNA – Cariprazine appears to improve predominant negative symptoms of schizophrenia as well as functional status in affected patients, István Bitter, MD, PhD, DSci reported at the annual congress of the European College of Neuropsychopharmacology.
Cariprazine, a dopamine D3/D2 receptor partial agonist, was approved in the United States in 2015 as Vraylar for adults with bipolar disorder or schizophrenia. The drug remains investigational in Europe.
“There is very clearly an unmet need for the treatment of deficit schizophrenia, or schizophrenia with predominant and persistent negative symptoms,” the psychiatrist observed.
The strong efficacy signal for cariprazine in patients with predominant negative symptoms was identified in a post hoc analysis of a randomized, double-blind, phase III head-to-head comparison of cariprazine versus risperidone (Risperdal) in 461 affected adults. Dr. Bitter was a coinvestigator in the 26-week multicenter European study, which was preceded by a 4-week lead-in phase in which participants were uptitrated to a target dose of cariprazine at 4.5 mg/day or risperidone at 4 mg/day.
The primary endpoint in the post hoc analysis was change from baseline on the Positive and Negative Symptoms Factor Score for Negative Symptoms (PANSS-FSNS). A significant difference in favor of cariprazine was seen by week 14, and the gap steadily enlarged thereafter through the remainder of the study. The difference was significant in five of the seven items on the PANSS-FSNS: blunted affect, emotional withdrawal, poor rapport, active social avoidance, and passive/apathetic social withdrawal. Cariprazine also outperformed risperidone on the other two elements of the scale – motor retardation, and lack of spontaneity and flow of conversation – but in those domains, the difference didn’t reach statistical significance.
Patients randomized to cariprazine also fared well on secondary endpoints. They displayed a significantly greater overall improvement in Marder factor scores over 26 weeks than the risperidone-treated group. This advantage was driven by a strong, statistically significant difference in the domain of difficulty in abstract thinking. However, the cariprazine group also showed nonsignificant trends for greater improvement in conceptual disorganization, mannerisms and posturing, disorientation, poor attention, and disturbed volition.
As evidence that cariprazine was exerting specific benefit on negative symptoms, Dr. Bitter cited the fact that the drug showed no significant difference from risperidone in change from baseline on the Marder factor scores for uncontrolled hostility/excitement and anxiety/depression, nor in the Calgary Depression Rating Scale for Schizophrenia.
Regulatory agencies in the United States and Europe have made it clear that, for a drug to obtain an indication for treatment of predominant negative symptoms of schizophrenia, it also has to show evidence of improved patient social function. The cariprazine group showed a significantly greater improvement on the PANSS-FSNS Personal and Social Performance Score. This advantage over risperidone-treated patients reached statistical significance at week 10 and broadened thereafter until study conclusion. Improvements in the subdomain scores for self-care, socially useful activities, and personal and social relationships led the way.
These positive results for cariprazine represent a sharp departure from the psychiatric field’s long-standing history of failed therapeutic attempts to improve negative symptoms. A comprehensive 2015 meta-analysis of all 168 randomized, placebo-controlled studies of various potential treatments for predominant negative symptoms of schizophrenia published through 2013 concluded that none of them provided evidence of clinically meaningful improvement (Schizophr Bull. 2015 Jul;41[4]:892-9).
Dr. Bitter observed that, since that discouraging meta-analysis, several additional novel agents for treatment of predominant negative symptoms of schizophrenia that showed “fantastic” promise in phase II studies subsequently went down in flames in advanced phase III trials. Among those were D-serine as an add-on to second-generation antipsychotics; encenicline, an alpha-7 nicotinic acetylcholine receptor antagonist; pomaglumetad methionil, a selective agonist for glutamate receptor subtypes mGluR2 and mGluR3; and bitopertin, a glycine reuptake inhibitor.
“This is basically a very negative message, that there is not much we can do about negative symptoms. But depending on who you ask, you can get a little bit more optimistic picture,” Dr. Bitter said.
That’s because there are considerable differences among the studies both in how negative symptoms are defined as well as in the measurement tools employed. For example, the Scale for Assessment of Negative Symptoms (SANS) includes items which aren’t strictly speaking part of the negative symptoms concept, so it yields skewed results. The Brief Psychiatric Rating Scale is a relic that has been abandoned by younger psychiatrists. So at present, the PANS-FSNS is the best available tool, and a reasonable common sense definition of predominant negative symptoms of schizophrenia is that, in an affected patient, the PANSS negative subscale score is higher than the positive one, he continued.
It’s noteworthy that none of the multitude of failed drugs for negative symptoms of schizophrenia target activity of dopamine-3 receptors. But cariprazine does.
“There is hope that dopamine-3 receptors might play a role in schizophrenia, especially in negative symptoms. According to experts, they are much older by a couple of million years than the D2 receptors. They don’t operate like D2. In fact, their function is not very clear. In animal studies, though, they help with cognition,” Dr. Bitter said.
“These cariprazine results open the door to some hope for the future,” commented Dr. Ferrara, professor of psychiatry at the University of Naples, Italy.
Even so, she added in an interview, most clinicians aren’t ready to handle a drug with an indication for treatment of negative symptoms. “We are not ready as clinicians to rate negative symptoms in our patients, or in many, many cases to even recognize them. In my opinion, several promising drugs failed because of the fact that the evaluations were not very well carried out in spite of training, but also because, so far, the assessment instruments have not been the best you could think of. Clinicians need training, and the assessment instruments need to be refined. We need instruments that provide more in-depth evaluation of the different aspects of negative symptoms and that are also more appealing to clinicians,” according to the psychiatrist.
She agreed with Dr. Bitter that, for now, the PANSS-FSNS is the best available assessment tool.
“The SANS is not a good tool for negative symptoms. We are all sure about that. We can and should do better,” Dr. Ferrara said.
The cariprazine study was funded by Gedeon Richter and Allergen. Dr. Bitter reported serving as an advisory board member and/or consultant to Gedeon Richter and eight other pharmaceutical companies.
VIENNA – Cariprazine appears to improve predominant negative symptoms of schizophrenia as well as functional status in affected patients, István Bitter, MD, PhD, DSci reported at the annual congress of the European College of Neuropsychopharmacology.
Cariprazine, a dopamine D3/D2 receptor partial agonist, was approved in the United States in 2015 as Vraylar for adults with bipolar disorder or schizophrenia. The drug remains investigational in Europe.
“There is very clearly an unmet need for the treatment of deficit schizophrenia, or schizophrenia with predominant and persistent negative symptoms,” the psychiatrist observed.
The strong efficacy signal for cariprazine in patients with predominant negative symptoms was identified in a post hoc analysis of a randomized, double-blind, phase III head-to-head comparison of cariprazine versus risperidone (Risperdal) in 461 affected adults. Dr. Bitter was a coinvestigator in the 26-week multicenter European study, which was preceded by a 4-week lead-in phase in which participants were uptitrated to a target dose of cariprazine at 4.5 mg/day or risperidone at 4 mg/day.
The primary endpoint in the post hoc analysis was change from baseline on the Positive and Negative Symptoms Factor Score for Negative Symptoms (PANSS-FSNS). A significant difference in favor of cariprazine was seen by week 14, and the gap steadily enlarged thereafter through the remainder of the study. The difference was significant in five of the seven items on the PANSS-FSNS: blunted affect, emotional withdrawal, poor rapport, active social avoidance, and passive/apathetic social withdrawal. Cariprazine also outperformed risperidone on the other two elements of the scale – motor retardation, and lack of spontaneity and flow of conversation – but in those domains, the difference didn’t reach statistical significance.
Patients randomized to cariprazine also fared well on secondary endpoints. They displayed a significantly greater overall improvement in Marder factor scores over 26 weeks than the risperidone-treated group. This advantage was driven by a strong, statistically significant difference in the domain of difficulty in abstract thinking. However, the cariprazine group also showed nonsignificant trends for greater improvement in conceptual disorganization, mannerisms and posturing, disorientation, poor attention, and disturbed volition.
As evidence that cariprazine was exerting specific benefit on negative symptoms, Dr. Bitter cited the fact that the drug showed no significant difference from risperidone in change from baseline on the Marder factor scores for uncontrolled hostility/excitement and anxiety/depression, nor in the Calgary Depression Rating Scale for Schizophrenia.
Regulatory agencies in the United States and Europe have made it clear that, for a drug to obtain an indication for treatment of predominant negative symptoms of schizophrenia, it also has to show evidence of improved patient social function. The cariprazine group showed a significantly greater improvement on the PANSS-FSNS Personal and Social Performance Score. This advantage over risperidone-treated patients reached statistical significance at week 10 and broadened thereafter until study conclusion. Improvements in the subdomain scores for self-care, socially useful activities, and personal and social relationships led the way.
These positive results for cariprazine represent a sharp departure from the psychiatric field’s long-standing history of failed therapeutic attempts to improve negative symptoms. A comprehensive 2015 meta-analysis of all 168 randomized, placebo-controlled studies of various potential treatments for predominant negative symptoms of schizophrenia published through 2013 concluded that none of them provided evidence of clinically meaningful improvement (Schizophr Bull. 2015 Jul;41[4]:892-9).
Dr. Bitter observed that, since that discouraging meta-analysis, several additional novel agents for treatment of predominant negative symptoms of schizophrenia that showed “fantastic” promise in phase II studies subsequently went down in flames in advanced phase III trials. Among those were D-serine as an add-on to second-generation antipsychotics; encenicline, an alpha-7 nicotinic acetylcholine receptor antagonist; pomaglumetad methionil, a selective agonist for glutamate receptor subtypes mGluR2 and mGluR3; and bitopertin, a glycine reuptake inhibitor.
“This is basically a very negative message, that there is not much we can do about negative symptoms. But depending on who you ask, you can get a little bit more optimistic picture,” Dr. Bitter said.
That’s because there are considerable differences among the studies both in how negative symptoms are defined as well as in the measurement tools employed. For example, the Scale for Assessment of Negative Symptoms (SANS) includes items which aren’t strictly speaking part of the negative symptoms concept, so it yields skewed results. The Brief Psychiatric Rating Scale is a relic that has been abandoned by younger psychiatrists. So at present, the PANS-FSNS is the best available tool, and a reasonable common sense definition of predominant negative symptoms of schizophrenia is that, in an affected patient, the PANSS negative subscale score is higher than the positive one, he continued.
It’s noteworthy that none of the multitude of failed drugs for negative symptoms of schizophrenia target activity of dopamine-3 receptors. But cariprazine does.
“There is hope that dopamine-3 receptors might play a role in schizophrenia, especially in negative symptoms. According to experts, they are much older by a couple of million years than the D2 receptors. They don’t operate like D2. In fact, their function is not very clear. In animal studies, though, they help with cognition,” Dr. Bitter said.
“These cariprazine results open the door to some hope for the future,” commented Dr. Ferrara, professor of psychiatry at the University of Naples, Italy.
Even so, she added in an interview, most clinicians aren’t ready to handle a drug with an indication for treatment of negative symptoms. “We are not ready as clinicians to rate negative symptoms in our patients, or in many, many cases to even recognize them. In my opinion, several promising drugs failed because of the fact that the evaluations were not very well carried out in spite of training, but also because, so far, the assessment instruments have not been the best you could think of. Clinicians need training, and the assessment instruments need to be refined. We need instruments that provide more in-depth evaluation of the different aspects of negative symptoms and that are also more appealing to clinicians,” according to the psychiatrist.
She agreed with Dr. Bitter that, for now, the PANSS-FSNS is the best available assessment tool.
“The SANS is not a good tool for negative symptoms. We are all sure about that. We can and should do better,” Dr. Ferrara said.
The cariprazine study was funded by Gedeon Richter and Allergen. Dr. Bitter reported serving as an advisory board member and/or consultant to Gedeon Richter and eight other pharmaceutical companies.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Cariprazine resulted in significantly greater improvement than risperidone in negative symptoms of schizophrenia as well as in social functioning.
Data source: This was a post hoc analysis of a 28-week, randomized, multicenter, phase III head-to-head comparative trial of cariprazine versus risperidone in 461 schizophrenia patients with predominant negative symptoms.
Disclosures: The study was funded by Gedeon Richter and Allergen. The presenter reported serving as an advisory board member and/or consultant to Gedeon Richter and eight other pharmaceutical companies.
Reassuring findings on neurodevelopmental outcomes in HIV-exposed children
DURBAN, SOUTH AFRICA – Children exposed to HIV in utero but uninfected at birth have neurodevelopmental test scores at age 24 months that are comparable with those of unexposed children, based on a study conducted in Botswana and presented by Jean Leidner at the 21st International AIDS Conference.
“These results provide reassurance regarding the potential effects of in-utero HIV and antiretroviral exposure,” declared Ms. Leidner, CEO of Goodtables Data Consulting in Norman, Okla., and the Botswana Harvard AIDS Institute Partnership.
The two groups of children had virtually identical scores on the cognitive, gross motor, fine motor, expressive language, and receptive language domains measured in the Bayley-III. The same was true for scores on the fine motor, locomotor, language, and personal-social elements of the Developmental Milestone Checklist.
The two groups of children differed in other ways; 17% of the uninfected children exposed to HIV in utero and 8% of the controls were low birth weight. The HIV-exposed children are being raised in a more challenging environment: just 49% have electricity in the home, compared with 64% of control families. Moreover, 53% of the HIV-exposed children and 33% of the controls live under conditions of moderate-to-severe food uncertainty.
Only 8% of the HIV-infected mothers breastfed, whereas breastfeeding was universal among the control group.
More than 99% of the HIV-infected mothers took antiretroviral medication antenatally. Roughly two-thirds were on zidovudine (Retrovir) monotherapy, the rest on a three-drug regimen of nevirapine (Viramune) plus lamivudine/zidovudine (Combivir). These are older antiretrovirals. Additional neurodevelopmental studies are warranted in children with in-utero exposure to newer agents, as well as in older children, Ms. Leidner said.
She reported having no financial conflicts regarding this study, which was funded by the National Institute of Mental Health.
DURBAN, SOUTH AFRICA – Children exposed to HIV in utero but uninfected at birth have neurodevelopmental test scores at age 24 months that are comparable with those of unexposed children, based on a study conducted in Botswana and presented by Jean Leidner at the 21st International AIDS Conference.
“These results provide reassurance regarding the potential effects of in-utero HIV and antiretroviral exposure,” declared Ms. Leidner, CEO of Goodtables Data Consulting in Norman, Okla., and the Botswana Harvard AIDS Institute Partnership.
The two groups of children had virtually identical scores on the cognitive, gross motor, fine motor, expressive language, and receptive language domains measured in the Bayley-III. The same was true for scores on the fine motor, locomotor, language, and personal-social elements of the Developmental Milestone Checklist.
The two groups of children differed in other ways; 17% of the uninfected children exposed to HIV in utero and 8% of the controls were low birth weight. The HIV-exposed children are being raised in a more challenging environment: just 49% have electricity in the home, compared with 64% of control families. Moreover, 53% of the HIV-exposed children and 33% of the controls live under conditions of moderate-to-severe food uncertainty.
Only 8% of the HIV-infected mothers breastfed, whereas breastfeeding was universal among the control group.
More than 99% of the HIV-infected mothers took antiretroviral medication antenatally. Roughly two-thirds were on zidovudine (Retrovir) monotherapy, the rest on a three-drug regimen of nevirapine (Viramune) plus lamivudine/zidovudine (Combivir). These are older antiretrovirals. Additional neurodevelopmental studies are warranted in children with in-utero exposure to newer agents, as well as in older children, Ms. Leidner said.
She reported having no financial conflicts regarding this study, which was funded by the National Institute of Mental Health.
DURBAN, SOUTH AFRICA – Children exposed to HIV in utero but uninfected at birth have neurodevelopmental test scores at age 24 months that are comparable with those of unexposed children, based on a study conducted in Botswana and presented by Jean Leidner at the 21st International AIDS Conference.
“These results provide reassurance regarding the potential effects of in-utero HIV and antiretroviral exposure,” declared Ms. Leidner, CEO of Goodtables Data Consulting in Norman, Okla., and the Botswana Harvard AIDS Institute Partnership.
The two groups of children had virtually identical scores on the cognitive, gross motor, fine motor, expressive language, and receptive language domains measured in the Bayley-III. The same was true for scores on the fine motor, locomotor, language, and personal-social elements of the Developmental Milestone Checklist.
The two groups of children differed in other ways; 17% of the uninfected children exposed to HIV in utero and 8% of the controls were low birth weight. The HIV-exposed children are being raised in a more challenging environment: just 49% have electricity in the home, compared with 64% of control families. Moreover, 53% of the HIV-exposed children and 33% of the controls live under conditions of moderate-to-severe food uncertainty.
Only 8% of the HIV-infected mothers breastfed, whereas breastfeeding was universal among the control group.
More than 99% of the HIV-infected mothers took antiretroviral medication antenatally. Roughly two-thirds were on zidovudine (Retrovir) monotherapy, the rest on a three-drug regimen of nevirapine (Viramune) plus lamivudine/zidovudine (Combivir). These are older antiretrovirals. Additional neurodevelopmental studies are warranted in children with in-utero exposure to newer agents, as well as in older children, Ms. Leidner said.
She reported having no financial conflicts regarding this study, which was funded by the National Institute of Mental Health.
Key clinical point:
Major finding: In-utero exposure to maternal HIV and antiretroviral drugs had no measurable adverse neurodevelopmental effects at age 24 months in uninfected children.
Data source: 337 uninfected children exposed to HIV in-utero and 387 children unexposed to HIV in utero.
Disclosures: The National Institute of Mental Health funded the study. The presenter reported having no financial conflicts of interest.
Atopic dermatitis: Pivotal dupilumab results create sensation
VIENNA – The marquee event at this year’s annual congress of the European Academy of Dermatology and Venereology – the one everyone was eagerly awaiting – was the first presentation of two large, international, pivotal phase III randomized trials of dupilumab for treatment of inadequately controlled moderate to severe atopic dermatitis in adults.
Attendees at EADV 2016 understood that, if positive, these studies, known as SOLO 1 and SOLO 2, would be transformative. They would herald a new era of highly effective targeted biologic therapy for this common and often debilitating chronic relapsing skin disease, akin to what occurred in psoriasis therapy well over a decade ago.
The results did not disappoint.
“Dual targeting of interleukin-4 and -13 represents a therapeutic option for patients with moderate to severe atopic dermatitis,” added Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
These results have implications extending beyond atopic dermatitis. Asthma, chronic sinusitis with nasal polyposis, and eosinophilic esophagitis are other conditions where the type 2 inflammatory cytokines IL-4 and -13 are believed to be important drivers of disease activity. Clinical trials of dupilumab in those diseases are underway.
Dupilumab, a fully human monoclonal antibody that binds specifically to the shared alpha chain subunit of the IL-4 and -13 receptors, hit all of its primary and secondary outcome measures in SOLO 1 and SOLO 2. Moreover, some of these “secondary” endpoints are consistently reported in patient surveys to be among what they consider to be the most troublesome aspects of atopic dermatitis, including intense itching, disrupted sleep, clinically significant anxiety and/or depression, and generally diminished quality of life.
SOLO 1 and SOLO 2 were identically designed, independent, randomized, double-blind, placebo-controlled clinical trials of 16 weeks’ duration. Conducted in North America, Europe, and Asia, they included a total of 1,379 patients, split roughly 50/50 between those with moderate or severe atopic dermatitis. Their average disease duration was 26 years. Participants were randomized to subcutaneous injection of dupilumab at 300 mg once weekly or every 2 weeks or to matching placebo.
The primary endpoint was a score of clear or almost clear – 0 or 1 – on the Investigator’s Global Assessment (IGA) at week 16 accompanied by a reduction of at least 2 points from baseline. A key secondary endpoint was at least a 75% improvement in the Eczema Area and Severity Index (EASI-75), considered a coprimary endpoint by regulators in Japan and the European Union.
The use of topical agents for atopic dermatitis was not permitted except as rescue therapy for uncontrolled symptoms. An IGA of 0 or 1 with at least a 2-point drop from baseline was a high bar to reach, given that a median of 50% of participants’ body surface area was affected. But in SOLO 1, that target was achieved in 37.9% of subjects on dupilumab every other week, 37.2% with weekly therapy, and just 10.3% of placebo-treated controls. Similarly, in SOLO 2, the rates were 36.1%, 36.4%, and 8.5%, respectively.
Of note, there were essentially no differences in outcomes across the board with weekly versus biweekly dosing of dupilumab.
From a median baseline EASI score of 30, an EASI-75 was achieved at 16 weeks in 51.3% of patients on dupilumab every other week, 52.5% on weekly injections, and 14.7% of controls in SOLO 1. In SOLO 2, the corresponding EASI-75 rates were 44.2%, 48.1%, and 11.9%, respectively.
Itch is described by most patients with moderate to severe atopic dermatitis as their No. 1 issue. From a baseline median peak score of 7.7 on a 0-10 numerical rating scale for pruritus, week 16 scores dropped by a median of 51% in patients on dupilumab every 2 weeks, 48.9% with weekly therapy, and 26.1% with placebo in SOLO 1. Results in SOLO 2 mirrored those in SOLO 1.
Particularly noteworthy was the finding that a significant reduction in itch severity was documented by week 2 in both dupilumab treatment arms, Dr. Simpson observed.
Just under half of study participants had a baseline score of 8 or more on the Hospital Anxiety and Depression Scale Anxiety subscale or HADS Depression subscale, considered the cutoff for a clinically significant mood disorder. Among affected patients, a score of less than 8 was achieved at 16 weeks without the use of psychotropic medications in 12.4% of SOLO 1 participants on placebo, 41% on biweekly dupilumab, and 36.3% with weekly dupilumab. In SOLO 2, the rates were 6.1% with placebo, 39.5% with biweekly dupilumab, and 41.2% with once-weekly dupilumab.
The median baseline Dermatology Life Quality Index score was 15 across the two parallel trials. The collective proportion of patients who experienced at least a 4-point improvement, which is considered a clinically meaningful response, was 29.1% in controls, compared with 68.6% in patients dupilumab every other week and 60.2% with weekly dupilumab.
On the Patient-Oriented Eczema Measure, a composite yardstick that emphasizes sleep symptoms, the median baseline score was 22 out of a possible 28. An improvement of 4 points or more, defined as a minimal clinically important difference, was achieved in a collective 25.6% of controls, 69.6% of patients on biweekly dupilumab, and 63.6% on weekly dupilumab.
Regarding safety, no increase in infections was seen with dupilumab. In fact, only two adverse events were more frequent than with placebo. One was injection-site reactions, which were two- to threefold more common than in controls, and all of which were mild to moderate. The other safety issue was conjunctivitis, which occurred in three patients in the control arms of SOLO 1 and 2, compared with 36 in the dupilumab arms.
Asked about the mechanism of this conjunctivitis, Dr. Simpson said it remains unknown. There was no signal of an issue in the phase II studies.
“Ongoing studies are attempting to further characterize the affected patients. I would say the comforting thing is that most cases have been mild to moderate and have responded to topical steroids or topical cyclosporine. Only one patient had to discontinue dupilumab,” according to the dermatologist.
In any event, 16 weeks of treatment is not sufficient to determine the safety of long-term therapy. Long-term extension studies of SOLO 1 and 2 are well underway, as are earlier stage clinical trials in pediatric patients with moderate to severe atopic dermatitis.
In response to another audience question, Dr. Simpson said he and his coinvestigators plan to drill down into the data to see if patients with severe atopic dermatitis obtained significantly more benefits from weekly as compared with biweekly therapy, or if treatment every 2 weeks was as good as weekly therapy across the board. It’s an important question, but the study finished so recently that the investigators haven’t yet had time to conduct the analysis.
The pivotal phase III dupilumab findings met with an enthusiastic reception.
“Biologic therapy for atopic dermatitis is the light at the end of the tunnel,” declared session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
“Seminal work,” commented David M. Pariser, MD, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Dr. Simpson’s presentation of the pivotal dupilumab studies was but one of the highlights of a horn-of-plenty late-breaking clinical trials session held on the final full day of EADV 2016. As attendees mingled in the hall afterward, a palpable sense of pride in their profession was evident. It was borne of the knowledge that their field not only includes basic and translational scientists capable of unraveling the inflammatory pathways involved in a challenging disease like atopic dermatitis, where there is a long-standing unmet need for new therapies, but also that their specialty includes experienced clinical trialists who can put those novel targeted therapies to the test.
There was also a sense of satisfaction that, although dermatology is a small specialty, these accomplishments are drawing favorable attention throughout the broader medical community. Pivotal trials of novel treatments for important dermatologic diseases are regularly getting published in prominent nondermatology journals. For instance, simultaneous with Dr. Simpson’s presentation in Vienna at EADV 2016, the SOLO 1 and 2 results were published online in the New England Journal of Medicine (doi. 10.1056/NEJMoa1610020).
“The online publication occurred a few minutes ago, at the start of my presentation. I didn’t say anything then because I didn’t want everybody looking at their cell phones,” he quipped.
The Food and Drug Administration has granted dupilumab a breakthrough therapy designation; a decision on the application for approval is expected by March 29, 2017.
The phase III dupilumab trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Simpson reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
VIENNA – The marquee event at this year’s annual congress of the European Academy of Dermatology and Venereology – the one everyone was eagerly awaiting – was the first presentation of two large, international, pivotal phase III randomized trials of dupilumab for treatment of inadequately controlled moderate to severe atopic dermatitis in adults.
Attendees at EADV 2016 understood that, if positive, these studies, known as SOLO 1 and SOLO 2, would be transformative. They would herald a new era of highly effective targeted biologic therapy for this common and often debilitating chronic relapsing skin disease, akin to what occurred in psoriasis therapy well over a decade ago.
The results did not disappoint.
“Dual targeting of interleukin-4 and -13 represents a therapeutic option for patients with moderate to severe atopic dermatitis,” added Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
These results have implications extending beyond atopic dermatitis. Asthma, chronic sinusitis with nasal polyposis, and eosinophilic esophagitis are other conditions where the type 2 inflammatory cytokines IL-4 and -13 are believed to be important drivers of disease activity. Clinical trials of dupilumab in those diseases are underway.
Dupilumab, a fully human monoclonal antibody that binds specifically to the shared alpha chain subunit of the IL-4 and -13 receptors, hit all of its primary and secondary outcome measures in SOLO 1 and SOLO 2. Moreover, some of these “secondary” endpoints are consistently reported in patient surveys to be among what they consider to be the most troublesome aspects of atopic dermatitis, including intense itching, disrupted sleep, clinically significant anxiety and/or depression, and generally diminished quality of life.
SOLO 1 and SOLO 2 were identically designed, independent, randomized, double-blind, placebo-controlled clinical trials of 16 weeks’ duration. Conducted in North America, Europe, and Asia, they included a total of 1,379 patients, split roughly 50/50 between those with moderate or severe atopic dermatitis. Their average disease duration was 26 years. Participants were randomized to subcutaneous injection of dupilumab at 300 mg once weekly or every 2 weeks or to matching placebo.
The primary endpoint was a score of clear or almost clear – 0 or 1 – on the Investigator’s Global Assessment (IGA) at week 16 accompanied by a reduction of at least 2 points from baseline. A key secondary endpoint was at least a 75% improvement in the Eczema Area and Severity Index (EASI-75), considered a coprimary endpoint by regulators in Japan and the European Union.
The use of topical agents for atopic dermatitis was not permitted except as rescue therapy for uncontrolled symptoms. An IGA of 0 or 1 with at least a 2-point drop from baseline was a high bar to reach, given that a median of 50% of participants’ body surface area was affected. But in SOLO 1, that target was achieved in 37.9% of subjects on dupilumab every other week, 37.2% with weekly therapy, and just 10.3% of placebo-treated controls. Similarly, in SOLO 2, the rates were 36.1%, 36.4%, and 8.5%, respectively.
Of note, there were essentially no differences in outcomes across the board with weekly versus biweekly dosing of dupilumab.
From a median baseline EASI score of 30, an EASI-75 was achieved at 16 weeks in 51.3% of patients on dupilumab every other week, 52.5% on weekly injections, and 14.7% of controls in SOLO 1. In SOLO 2, the corresponding EASI-75 rates were 44.2%, 48.1%, and 11.9%, respectively.
Itch is described by most patients with moderate to severe atopic dermatitis as their No. 1 issue. From a baseline median peak score of 7.7 on a 0-10 numerical rating scale for pruritus, week 16 scores dropped by a median of 51% in patients on dupilumab every 2 weeks, 48.9% with weekly therapy, and 26.1% with placebo in SOLO 1. Results in SOLO 2 mirrored those in SOLO 1.
Particularly noteworthy was the finding that a significant reduction in itch severity was documented by week 2 in both dupilumab treatment arms, Dr. Simpson observed.
Just under half of study participants had a baseline score of 8 or more on the Hospital Anxiety and Depression Scale Anxiety subscale or HADS Depression subscale, considered the cutoff for a clinically significant mood disorder. Among affected patients, a score of less than 8 was achieved at 16 weeks without the use of psychotropic medications in 12.4% of SOLO 1 participants on placebo, 41% on biweekly dupilumab, and 36.3% with weekly dupilumab. In SOLO 2, the rates were 6.1% with placebo, 39.5% with biweekly dupilumab, and 41.2% with once-weekly dupilumab.
The median baseline Dermatology Life Quality Index score was 15 across the two parallel trials. The collective proportion of patients who experienced at least a 4-point improvement, which is considered a clinically meaningful response, was 29.1% in controls, compared with 68.6% in patients dupilumab every other week and 60.2% with weekly dupilumab.
On the Patient-Oriented Eczema Measure, a composite yardstick that emphasizes sleep symptoms, the median baseline score was 22 out of a possible 28. An improvement of 4 points or more, defined as a minimal clinically important difference, was achieved in a collective 25.6% of controls, 69.6% of patients on biweekly dupilumab, and 63.6% on weekly dupilumab.
Regarding safety, no increase in infections was seen with dupilumab. In fact, only two adverse events were more frequent than with placebo. One was injection-site reactions, which were two- to threefold more common than in controls, and all of which were mild to moderate. The other safety issue was conjunctivitis, which occurred in three patients in the control arms of SOLO 1 and 2, compared with 36 in the dupilumab arms.
Asked about the mechanism of this conjunctivitis, Dr. Simpson said it remains unknown. There was no signal of an issue in the phase II studies.
“Ongoing studies are attempting to further characterize the affected patients. I would say the comforting thing is that most cases have been mild to moderate and have responded to topical steroids or topical cyclosporine. Only one patient had to discontinue dupilumab,” according to the dermatologist.
In any event, 16 weeks of treatment is not sufficient to determine the safety of long-term therapy. Long-term extension studies of SOLO 1 and 2 are well underway, as are earlier stage clinical trials in pediatric patients with moderate to severe atopic dermatitis.
In response to another audience question, Dr. Simpson said he and his coinvestigators plan to drill down into the data to see if patients with severe atopic dermatitis obtained significantly more benefits from weekly as compared with biweekly therapy, or if treatment every 2 weeks was as good as weekly therapy across the board. It’s an important question, but the study finished so recently that the investigators haven’t yet had time to conduct the analysis.
The pivotal phase III dupilumab findings met with an enthusiastic reception.
“Biologic therapy for atopic dermatitis is the light at the end of the tunnel,” declared session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
“Seminal work,” commented David M. Pariser, MD, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Dr. Simpson’s presentation of the pivotal dupilumab studies was but one of the highlights of a horn-of-plenty late-breaking clinical trials session held on the final full day of EADV 2016. As attendees mingled in the hall afterward, a palpable sense of pride in their profession was evident. It was borne of the knowledge that their field not only includes basic and translational scientists capable of unraveling the inflammatory pathways involved in a challenging disease like atopic dermatitis, where there is a long-standing unmet need for new therapies, but also that their specialty includes experienced clinical trialists who can put those novel targeted therapies to the test.
There was also a sense of satisfaction that, although dermatology is a small specialty, these accomplishments are drawing favorable attention throughout the broader medical community. Pivotal trials of novel treatments for important dermatologic diseases are regularly getting published in prominent nondermatology journals. For instance, simultaneous with Dr. Simpson’s presentation in Vienna at EADV 2016, the SOLO 1 and 2 results were published online in the New England Journal of Medicine (doi. 10.1056/NEJMoa1610020).
“The online publication occurred a few minutes ago, at the start of my presentation. I didn’t say anything then because I didn’t want everybody looking at their cell phones,” he quipped.
The Food and Drug Administration has granted dupilumab a breakthrough therapy designation; a decision on the application for approval is expected by March 29, 2017.
The phase III dupilumab trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Simpson reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
VIENNA – The marquee event at this year’s annual congress of the European Academy of Dermatology and Venereology – the one everyone was eagerly awaiting – was the first presentation of two large, international, pivotal phase III randomized trials of dupilumab for treatment of inadequately controlled moderate to severe atopic dermatitis in adults.
Attendees at EADV 2016 understood that, if positive, these studies, known as SOLO 1 and SOLO 2, would be transformative. They would herald a new era of highly effective targeted biologic therapy for this common and often debilitating chronic relapsing skin disease, akin to what occurred in psoriasis therapy well over a decade ago.
The results did not disappoint.
“Dual targeting of interleukin-4 and -13 represents a therapeutic option for patients with moderate to severe atopic dermatitis,” added Dr. Simpson, professor of dermatology at Oregon Health and Science University, Portland.
These results have implications extending beyond atopic dermatitis. Asthma, chronic sinusitis with nasal polyposis, and eosinophilic esophagitis are other conditions where the type 2 inflammatory cytokines IL-4 and -13 are believed to be important drivers of disease activity. Clinical trials of dupilumab in those diseases are underway.
Dupilumab, a fully human monoclonal antibody that binds specifically to the shared alpha chain subunit of the IL-4 and -13 receptors, hit all of its primary and secondary outcome measures in SOLO 1 and SOLO 2. Moreover, some of these “secondary” endpoints are consistently reported in patient surveys to be among what they consider to be the most troublesome aspects of atopic dermatitis, including intense itching, disrupted sleep, clinically significant anxiety and/or depression, and generally diminished quality of life.
SOLO 1 and SOLO 2 were identically designed, independent, randomized, double-blind, placebo-controlled clinical trials of 16 weeks’ duration. Conducted in North America, Europe, and Asia, they included a total of 1,379 patients, split roughly 50/50 between those with moderate or severe atopic dermatitis. Their average disease duration was 26 years. Participants were randomized to subcutaneous injection of dupilumab at 300 mg once weekly or every 2 weeks or to matching placebo.
The primary endpoint was a score of clear or almost clear – 0 or 1 – on the Investigator’s Global Assessment (IGA) at week 16 accompanied by a reduction of at least 2 points from baseline. A key secondary endpoint was at least a 75% improvement in the Eczema Area and Severity Index (EASI-75), considered a coprimary endpoint by regulators in Japan and the European Union.
The use of topical agents for atopic dermatitis was not permitted except as rescue therapy for uncontrolled symptoms. An IGA of 0 or 1 with at least a 2-point drop from baseline was a high bar to reach, given that a median of 50% of participants’ body surface area was affected. But in SOLO 1, that target was achieved in 37.9% of subjects on dupilumab every other week, 37.2% with weekly therapy, and just 10.3% of placebo-treated controls. Similarly, in SOLO 2, the rates were 36.1%, 36.4%, and 8.5%, respectively.
Of note, there were essentially no differences in outcomes across the board with weekly versus biweekly dosing of dupilumab.
From a median baseline EASI score of 30, an EASI-75 was achieved at 16 weeks in 51.3% of patients on dupilumab every other week, 52.5% on weekly injections, and 14.7% of controls in SOLO 1. In SOLO 2, the corresponding EASI-75 rates were 44.2%, 48.1%, and 11.9%, respectively.
Itch is described by most patients with moderate to severe atopic dermatitis as their No. 1 issue. From a baseline median peak score of 7.7 on a 0-10 numerical rating scale for pruritus, week 16 scores dropped by a median of 51% in patients on dupilumab every 2 weeks, 48.9% with weekly therapy, and 26.1% with placebo in SOLO 1. Results in SOLO 2 mirrored those in SOLO 1.
Particularly noteworthy was the finding that a significant reduction in itch severity was documented by week 2 in both dupilumab treatment arms, Dr. Simpson observed.
Just under half of study participants had a baseline score of 8 or more on the Hospital Anxiety and Depression Scale Anxiety subscale or HADS Depression subscale, considered the cutoff for a clinically significant mood disorder. Among affected patients, a score of less than 8 was achieved at 16 weeks without the use of psychotropic medications in 12.4% of SOLO 1 participants on placebo, 41% on biweekly dupilumab, and 36.3% with weekly dupilumab. In SOLO 2, the rates were 6.1% with placebo, 39.5% with biweekly dupilumab, and 41.2% with once-weekly dupilumab.
The median baseline Dermatology Life Quality Index score was 15 across the two parallel trials. The collective proportion of patients who experienced at least a 4-point improvement, which is considered a clinically meaningful response, was 29.1% in controls, compared with 68.6% in patients dupilumab every other week and 60.2% with weekly dupilumab.
On the Patient-Oriented Eczema Measure, a composite yardstick that emphasizes sleep symptoms, the median baseline score was 22 out of a possible 28. An improvement of 4 points or more, defined as a minimal clinically important difference, was achieved in a collective 25.6% of controls, 69.6% of patients on biweekly dupilumab, and 63.6% on weekly dupilumab.
Regarding safety, no increase in infections was seen with dupilumab. In fact, only two adverse events were more frequent than with placebo. One was injection-site reactions, which were two- to threefold more common than in controls, and all of which were mild to moderate. The other safety issue was conjunctivitis, which occurred in three patients in the control arms of SOLO 1 and 2, compared with 36 in the dupilumab arms.
Asked about the mechanism of this conjunctivitis, Dr. Simpson said it remains unknown. There was no signal of an issue in the phase II studies.
“Ongoing studies are attempting to further characterize the affected patients. I would say the comforting thing is that most cases have been mild to moderate and have responded to topical steroids or topical cyclosporine. Only one patient had to discontinue dupilumab,” according to the dermatologist.
In any event, 16 weeks of treatment is not sufficient to determine the safety of long-term therapy. Long-term extension studies of SOLO 1 and 2 are well underway, as are earlier stage clinical trials in pediatric patients with moderate to severe atopic dermatitis.
In response to another audience question, Dr. Simpson said he and his coinvestigators plan to drill down into the data to see if patients with severe atopic dermatitis obtained significantly more benefits from weekly as compared with biweekly therapy, or if treatment every 2 weeks was as good as weekly therapy across the board. It’s an important question, but the study finished so recently that the investigators haven’t yet had time to conduct the analysis.
The pivotal phase III dupilumab findings met with an enthusiastic reception.
“Biologic therapy for atopic dermatitis is the light at the end of the tunnel,” declared session cochair Lajos Kemény, MD, professor and chairman of the department of dermatology and allergology at the University of Szeged, Hungary.
“Seminal work,” commented David M. Pariser, MD, professor of dermatology at Eastern Virginia Medical School in Norfolk.
Dr. Simpson’s presentation of the pivotal dupilumab studies was but one of the highlights of a horn-of-plenty late-breaking clinical trials session held on the final full day of EADV 2016. As attendees mingled in the hall afterward, a palpable sense of pride in their profession was evident. It was borne of the knowledge that their field not only includes basic and translational scientists capable of unraveling the inflammatory pathways involved in a challenging disease like atopic dermatitis, where there is a long-standing unmet need for new therapies, but also that their specialty includes experienced clinical trialists who can put those novel targeted therapies to the test.
There was also a sense of satisfaction that, although dermatology is a small specialty, these accomplishments are drawing favorable attention throughout the broader medical community. Pivotal trials of novel treatments for important dermatologic diseases are regularly getting published in prominent nondermatology journals. For instance, simultaneous with Dr. Simpson’s presentation in Vienna at EADV 2016, the SOLO 1 and 2 results were published online in the New England Journal of Medicine (doi. 10.1056/NEJMoa1610020).
“The online publication occurred a few minutes ago, at the start of my presentation. I didn’t say anything then because I didn’t want everybody looking at their cell phones,” he quipped.
The Food and Drug Administration has granted dupilumab a breakthrough therapy designation; a decision on the application for approval is expected by March 29, 2017.
The phase III dupilumab trials were funded by Sanofi and Regeneron Pharmaceuticals. Dr. Simpson reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
Key clinical point:
Major finding: After 16 weeks of weekly or biweekly subcutaneous injections of dupilumab, 36%-38% of patients with baseline moderate or severe atopic dermatitis were clear or almost clear, compared with 8%-10% of placebo-treated controls.
Data source: The SOLO 1 and SOLO 2 pivotal phase III randomized, double-blind, placebo-controlled clinical trials included a total of 1,379 adults with inadequately controlled moderate or severe atopic dermatitis on three continents.
Disclosures: The trials were funded by Sanofi and Regeneron Pharmaceuticals. The presenter reported having received research grants from and serving as a consultant to Regeneron and more than a dozen other pharmaceutical companies.
Algorithm for suspected pulmonary embolism safely cut CT rate
ROME – A newly validated, simplified algorithm for the management of patients with suspected acute pulmonary embolism enables physicians to safely exclude the disorder in roughly half of patients without resorting to CT pulmonary angiography, Tom van der Hulle, MD, reported at the annual congress of the European Society of Cardiology.
“This is the largest study ever performed in the diagnostic management of suspected pulmonary embolism. Based on our results, I think the YEARS algorithm is ready to be used in daily clinical practice,” declared Dr. van der Hulle of the department of thrombosis and hemostasis at Leiden (the Netherlands) University Medical Center.
Using the YEARS algorithm, PE was reliably ruled out without need for CT pulmonary angiography – considered the standard in the diagnosis of PE – in 48% of patients. In contrast, adherence to the Wells rule would have meant that 62% of patients would have gotten a CT scan to rule out PE with a comparably high degree of accuracy.
But that 62% figure underestimates the actual CT rate in clinical practice. The reality is that although the guideline-recommended Wells rule and revised Geneva score have been shown to be safe and accurate, they are so complex, cumbersome, and out of sync with the flow of routine clinical practice that many physicians skip the algorithms and go straight to CT, Dr. van der Hulle said. This approach results in many unnecessary CTs, needlessly exposing patients to the risks of radiation and intravenous contrast material while driving up health care costs, he added.
Using the Wells rule or revised Geneva score, the patient evaluation begins with an assessment of the clinical probability of PE based upon a risk score involving seven or eight factors. Only patients with a low or intermediate clinical probability of PE get a D-dimer test; those with a high clinical probability go straight to CT.
The YEARS algorithm is much simpler than that, Dr. van der Hulle explained. Everyone who presents with suspected acute PE gets a D-dimer test while the physician simultaneously applies a brief, three-item clinical prediction rule. These three items were selected by the Dutch investigators because they were the three strongest predictors of PE out of the original seven in the Wells rule. They are hemoptysis, clinical signs of deep vein thrombosis such as leg swelling or hyperpigmentation, and the clinician’s global impression of PE as being the most likely diagnosis.
In the YEARS algorithm, the threshold for a positive D-dimer test warranting CT pulmonary angiography depends upon whether any of the three clinical predictors is present. If none is present, the threshold is 1,000 ng/mL or above; if one or more is present, the threshold for a positive D-dimer test drops to 500 ng/mL.
Using these criteria, PE was excluded without resort to CT in 1,306 patients with none of the three YEARS items and a D-dimer test result below 1,000 ng/mL, as well as in another 327 patients with one or more YEARS items present but a D-dimer below 500 ng/mL. Those two groups were left untreated and followed prospectively for 3 months.
The 964 patients with one or more YEARS predictors present and a D-dimer score of at least 500 ng/mL underwent CT imaging, as did the 352 with no YEARS items and a D-dimer of at least 1,000 ng/mL.
The prevalence of CT-confirmed PE in the study was 13.2%. Affected patients were treated with anticoagulants.
The primary study endpoint was the total rate of deep vein thrombosis during 3 months of follow-up after PE had been excluded. The rate was 0.61%, including a fatal PE rate of 0.20%. The rate in patients managed without CT was 0.43%, including a 0.12% rate of fatal PE. In patients managed with diagnostic CT, the deep vein thrombosis rate was 0.84%, with a fatal PE rate of 0.30%.
“I think these results are completely comparable to those in previous studies using the standard algorithms,” Dr. van der Hulle commented.
The study’s main limitation is that it wasn’t a randomized, controlled trial. But given the tiny event rates, detecting any small differences between management strategies would require an unrealistically huge sample size, he added.
Asked if he thinks physicians will actually use the new tool, Dr. van der Hulle replied that some physicians feel driven to be 100% sure that a patient doesn’t have PE, and they will probably keep overordering CT scans. But others will embrace the YEARS algorithm because it reduces wasted resources and minimizes radiation exposure, a particularly compelling consideration in young female patients.
Discussant Marion Delcroix, MD, had reservations. She said she appreciated the appeal of a simple algorithm, but she asked, “Couldn’t we do better with a bit more sophistication, perhaps by adjusting the D-dimer cutoff for age and also adding some other items, like oxygen saturation and estrogen use?
“My concern is about the applicability. The age of the study cohort is relatively young, at a mean of 53 years. The peak age of PE in a very large contemporary German database is 70-80 years. We don’t know if the YEARS score is any good in this older population,” asserted Dr. Delcroix, professor of medicine and respiratory physiology and head of the center for pulmonary vascular diseases at University Hospital in Leuven, Belgium.
“If the aim is to decrease the number of CT pulmonary angiograms for safety reasons, why not reintroduce compression ultrasound of the lower limbs in the diagnostic algorithm?” she continued. “It has been shown to effectively reduce the need for further imaging.”
Dr. Delcroix predicted that the YEARS algorithm study will prove “too optimistic” regarding the number of CT scans avoided, particularly in elderly patients.
The YEARS study was funded by the trial’s 12 participating Dutch hospitals. Dr. van der Hulle reported having no financial conflicts of interest.
ROME – A newly validated, simplified algorithm for the management of patients with suspected acute pulmonary embolism enables physicians to safely exclude the disorder in roughly half of patients without resorting to CT pulmonary angiography, Tom van der Hulle, MD, reported at the annual congress of the European Society of Cardiology.
“This is the largest study ever performed in the diagnostic management of suspected pulmonary embolism. Based on our results, I think the YEARS algorithm is ready to be used in daily clinical practice,” declared Dr. van der Hulle of the department of thrombosis and hemostasis at Leiden (the Netherlands) University Medical Center.
Using the YEARS algorithm, PE was reliably ruled out without need for CT pulmonary angiography – considered the standard in the diagnosis of PE – in 48% of patients. In contrast, adherence to the Wells rule would have meant that 62% of patients would have gotten a CT scan to rule out PE with a comparably high degree of accuracy.
But that 62% figure underestimates the actual CT rate in clinical practice. The reality is that although the guideline-recommended Wells rule and revised Geneva score have been shown to be safe and accurate, they are so complex, cumbersome, and out of sync with the flow of routine clinical practice that many physicians skip the algorithms and go straight to CT, Dr. van der Hulle said. This approach results in many unnecessary CTs, needlessly exposing patients to the risks of radiation and intravenous contrast material while driving up health care costs, he added.
Using the Wells rule or revised Geneva score, the patient evaluation begins with an assessment of the clinical probability of PE based upon a risk score involving seven or eight factors. Only patients with a low or intermediate clinical probability of PE get a D-dimer test; those with a high clinical probability go straight to CT.
The YEARS algorithm is much simpler than that, Dr. van der Hulle explained. Everyone who presents with suspected acute PE gets a D-dimer test while the physician simultaneously applies a brief, three-item clinical prediction rule. These three items were selected by the Dutch investigators because they were the three strongest predictors of PE out of the original seven in the Wells rule. They are hemoptysis, clinical signs of deep vein thrombosis such as leg swelling or hyperpigmentation, and the clinician’s global impression of PE as being the most likely diagnosis.
In the YEARS algorithm, the threshold for a positive D-dimer test warranting CT pulmonary angiography depends upon whether any of the three clinical predictors is present. If none is present, the threshold is 1,000 ng/mL or above; if one or more is present, the threshold for a positive D-dimer test drops to 500 ng/mL.
Using these criteria, PE was excluded without resort to CT in 1,306 patients with none of the three YEARS items and a D-dimer test result below 1,000 ng/mL, as well as in another 327 patients with one or more YEARS items present but a D-dimer below 500 ng/mL. Those two groups were left untreated and followed prospectively for 3 months.
The 964 patients with one or more YEARS predictors present and a D-dimer score of at least 500 ng/mL underwent CT imaging, as did the 352 with no YEARS items and a D-dimer of at least 1,000 ng/mL.
The prevalence of CT-confirmed PE in the study was 13.2%. Affected patients were treated with anticoagulants.
The primary study endpoint was the total rate of deep vein thrombosis during 3 months of follow-up after PE had been excluded. The rate was 0.61%, including a fatal PE rate of 0.20%. The rate in patients managed without CT was 0.43%, including a 0.12% rate of fatal PE. In patients managed with diagnostic CT, the deep vein thrombosis rate was 0.84%, with a fatal PE rate of 0.30%.
“I think these results are completely comparable to those in previous studies using the standard algorithms,” Dr. van der Hulle commented.
The study’s main limitation is that it wasn’t a randomized, controlled trial. But given the tiny event rates, detecting any small differences between management strategies would require an unrealistically huge sample size, he added.
Asked if he thinks physicians will actually use the new tool, Dr. van der Hulle replied that some physicians feel driven to be 100% sure that a patient doesn’t have PE, and they will probably keep overordering CT scans. But others will embrace the YEARS algorithm because it reduces wasted resources and minimizes radiation exposure, a particularly compelling consideration in young female patients.
Discussant Marion Delcroix, MD, had reservations. She said she appreciated the appeal of a simple algorithm, but she asked, “Couldn’t we do better with a bit more sophistication, perhaps by adjusting the D-dimer cutoff for age and also adding some other items, like oxygen saturation and estrogen use?
“My concern is about the applicability. The age of the study cohort is relatively young, at a mean of 53 years. The peak age of PE in a very large contemporary German database is 70-80 years. We don’t know if the YEARS score is any good in this older population,” asserted Dr. Delcroix, professor of medicine and respiratory physiology and head of the center for pulmonary vascular diseases at University Hospital in Leuven, Belgium.
“If the aim is to decrease the number of CT pulmonary angiograms for safety reasons, why not reintroduce compression ultrasound of the lower limbs in the diagnostic algorithm?” she continued. “It has been shown to effectively reduce the need for further imaging.”
Dr. Delcroix predicted that the YEARS algorithm study will prove “too optimistic” regarding the number of CT scans avoided, particularly in elderly patients.
The YEARS study was funded by the trial’s 12 participating Dutch hospitals. Dr. van der Hulle reported having no financial conflicts of interest.
ROME – A newly validated, simplified algorithm for the management of patients with suspected acute pulmonary embolism enables physicians to safely exclude the disorder in roughly half of patients without resorting to CT pulmonary angiography, Tom van der Hulle, MD, reported at the annual congress of the European Society of Cardiology.
“This is the largest study ever performed in the diagnostic management of suspected pulmonary embolism. Based on our results, I think the YEARS algorithm is ready to be used in daily clinical practice,” declared Dr. van der Hulle of the department of thrombosis and hemostasis at Leiden (the Netherlands) University Medical Center.
Using the YEARS algorithm, PE was reliably ruled out without need for CT pulmonary angiography – considered the standard in the diagnosis of PE – in 48% of patients. In contrast, adherence to the Wells rule would have meant that 62% of patients would have gotten a CT scan to rule out PE with a comparably high degree of accuracy.
But that 62% figure underestimates the actual CT rate in clinical practice. The reality is that although the guideline-recommended Wells rule and revised Geneva score have been shown to be safe and accurate, they are so complex, cumbersome, and out of sync with the flow of routine clinical practice that many physicians skip the algorithms and go straight to CT, Dr. van der Hulle said. This approach results in many unnecessary CTs, needlessly exposing patients to the risks of radiation and intravenous contrast material while driving up health care costs, he added.
Using the Wells rule or revised Geneva score, the patient evaluation begins with an assessment of the clinical probability of PE based upon a risk score involving seven or eight factors. Only patients with a low or intermediate clinical probability of PE get a D-dimer test; those with a high clinical probability go straight to CT.
The YEARS algorithm is much simpler than that, Dr. van der Hulle explained. Everyone who presents with suspected acute PE gets a D-dimer test while the physician simultaneously applies a brief, three-item clinical prediction rule. These three items were selected by the Dutch investigators because they were the three strongest predictors of PE out of the original seven in the Wells rule. They are hemoptysis, clinical signs of deep vein thrombosis such as leg swelling or hyperpigmentation, and the clinician’s global impression of PE as being the most likely diagnosis.
In the YEARS algorithm, the threshold for a positive D-dimer test warranting CT pulmonary angiography depends upon whether any of the three clinical predictors is present. If none is present, the threshold is 1,000 ng/mL or above; if one or more is present, the threshold for a positive D-dimer test drops to 500 ng/mL.
Using these criteria, PE was excluded without resort to CT in 1,306 patients with none of the three YEARS items and a D-dimer test result below 1,000 ng/mL, as well as in another 327 patients with one or more YEARS items present but a D-dimer below 500 ng/mL. Those two groups were left untreated and followed prospectively for 3 months.
The 964 patients with one or more YEARS predictors present and a D-dimer score of at least 500 ng/mL underwent CT imaging, as did the 352 with no YEARS items and a D-dimer of at least 1,000 ng/mL.
The prevalence of CT-confirmed PE in the study was 13.2%. Affected patients were treated with anticoagulants.
The primary study endpoint was the total rate of deep vein thrombosis during 3 months of follow-up after PE had been excluded. The rate was 0.61%, including a fatal PE rate of 0.20%. The rate in patients managed without CT was 0.43%, including a 0.12% rate of fatal PE. In patients managed with diagnostic CT, the deep vein thrombosis rate was 0.84%, with a fatal PE rate of 0.30%.
“I think these results are completely comparable to those in previous studies using the standard algorithms,” Dr. van der Hulle commented.
The study’s main limitation is that it wasn’t a randomized, controlled trial. But given the tiny event rates, detecting any small differences between management strategies would require an unrealistically huge sample size, he added.
Asked if he thinks physicians will actually use the new tool, Dr. van der Hulle replied that some physicians feel driven to be 100% sure that a patient doesn’t have PE, and they will probably keep overordering CT scans. But others will embrace the YEARS algorithm because it reduces wasted resources and minimizes radiation exposure, a particularly compelling consideration in young female patients.
Discussant Marion Delcroix, MD, had reservations. She said she appreciated the appeal of a simple algorithm, but she asked, “Couldn’t we do better with a bit more sophistication, perhaps by adjusting the D-dimer cutoff for age and also adding some other items, like oxygen saturation and estrogen use?
“My concern is about the applicability. The age of the study cohort is relatively young, at a mean of 53 years. The peak age of PE in a very large contemporary German database is 70-80 years. We don’t know if the YEARS score is any good in this older population,” asserted Dr. Delcroix, professor of medicine and respiratory physiology and head of the center for pulmonary vascular diseases at University Hospital in Leuven, Belgium.
“If the aim is to decrease the number of CT pulmonary angiograms for safety reasons, why not reintroduce compression ultrasound of the lower limbs in the diagnostic algorithm?” she continued. “It has been shown to effectively reduce the need for further imaging.”
Dr. Delcroix predicted that the YEARS algorithm study will prove “too optimistic” regarding the number of CT scans avoided, particularly in elderly patients.
The YEARS study was funded by the trial’s 12 participating Dutch hospitals. Dr. van der Hulle reported having no financial conflicts of interest.
Key clinical point:
Major finding: Applying the YEARS algorithm to a large population of patients with suspected PE, the 3-month incidence of deep vein thrombosis after PE had been excluded was 0.61%.
Data source: This was a prospective study of clinical outcomes in nearly 3,000 consecutive Dutch patients who presented with suspected acute PE and were managed in accord with the YEARS algorithm.
Disclosures: The YEARS algorithm validation study was funded by the trial’s 12 participating Dutch hospitals. The study presenter reported having no financial conflicts of interest.
October 2016: Click for Credit
Here are 5 articles in the October issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Autism Follow-up Screening by PCPs Yields High Accuracy
To take the posttest, go to: http://bit.ly/2bTLhFS
Expires August 19, 2017
VITALS
Key clinical point:
Primary care providers can conduct the M-CHAT/F following a positive M-CHAT screening for autism spectrum disorders.
Major finding:
Primary care providers and trained interviewers agreed 86.6% of the time on the screening results of the M-CHAT/F for ASDs.
Data source:
A cohort study of 5,071 children, mean age 23 months, screened with the M-CHAT, and a subsequent 197 children screened with the M-CHAT/F in 22 Maryland primary care practices.
Disclosures:
The National Institutes of Mental Health funded the research. Dr. Sturner is director of Total Child Health (TCH), a for-profit subsidiary of the Center for Promotion of Child Development through Primary Care, which conducted the study. Barbara Howard, MD, is president of TCH. Tanya Morrel, PhD, is an employee of and stockholder in TCH, and Paul Bergmann has consulted for the company. The remaining authors had no relevant disclosures.
2. Gallstone Disease Boosts Heart Risk
To take the posttest, go to: http://bit.ly/2c7TP7D
Expires August 18, 2017
VITALS
Key clinical point:
Gallstone disease is associated with an increased risk for coronary heart disease; preventing the former can help mitigate chances of developing the latter.
Major finding:
A meta-analysis revealed a 23% increased chance of CHD in gallstone disease patients.
Data source:
A meta-analysis of seven studies involving 842,553 subjects, and a prospective cohort study of 269,142 participants in three separate studies that took place from 1980 to 2011.
Disclosures:
Funding provided by NIH, Boston Obesity Nutrition Research Center, and United States-Israel Binational Science Foundation. The authors had no relevant financial disclosures.
3. New HER2-testing Guidelines Result in More Women Eligible for Directed Treatment
To take the posttest, go to: http://bit.ly/2cd9llO
Expires July 25, 2017
VITALS
Key clinical point:
New IHC and FISH pathology guidelines categorize more breast cancers as "equivocal" regarding HER2 positivity and ultimately lead to identifying more of them as HER2 positive.
Major finding:
By using 2013 guidelines, 358 additional tumors were interpreted as positive, compared with the 2007 guidelines and 298 additional tumors were considered positive, compared with the FDA criteria.
Data source:
A cohort study involving 2,851 breast cancer samples analyzed according to three different pathology guidelines during a 1-year period.
Disclosures:
This study was supported by the Mayo Clinic. Dr. Shah reported having no relevant financial disclosures; his associates reported ties to Merck, Hospira, Ariad Pharmaceuticals, Abbott Molecular, and Genome Diagnostics.
4. Extreme Alcohol Use Worsens HIV Disease
To take the posttest, go to: http://bit.ly/2coIzG3
Expires August 14, 2017
VITALS
Key clinical point:
A pattern of heavy alcohol use over time in HIV-infected patients was associated with accelerated HIV disease progression.
Major finding:
Long-term heavy alcohol use by middle-aged, HIV-infected military veterans was associated with a 1.83-fold increased likelihood of also being in the highest-risk group for accelerated progression of HIV disease.
Data source:
This study included 3,539 U.S. military veterans receiving care for HIV infection at eight VA centers. The impact of their long-term pattern of alcohol use on HIV disease progression was assessed over an 8-year period by annual assessments using validated instruments.
Disclosures:
The presenter reported having no financial conflicts of interest regarding the study, funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute of Allergy and Infectious Diseases.
5. Weight Loss Boosts TNFis' Psoriatic Arthritis Efficacy
To take the posttest, go to: http://bit.ly/2chD4M1
Expires July 23, 2017
Here are 5 articles in the October issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Autism Follow-up Screening by PCPs Yields High Accuracy
To take the posttest, go to: http://bit.ly/2bTLhFS
Expires August 19, 2017
VITALS
Key clinical point:
Primary care providers can conduct the M-CHAT/F following a positive M-CHAT screening for autism spectrum disorders.
Major finding:
Primary care providers and trained interviewers agreed 86.6% of the time on the screening results of the M-CHAT/F for ASDs.
Data source:
A cohort study of 5,071 children, mean age 23 months, screened with the M-CHAT, and a subsequent 197 children screened with the M-CHAT/F in 22 Maryland primary care practices.
Disclosures:
The National Institutes of Mental Health funded the research. Dr. Sturner is director of Total Child Health (TCH), a for-profit subsidiary of the Center for Promotion of Child Development through Primary Care, which conducted the study. Barbara Howard, MD, is president of TCH. Tanya Morrel, PhD, is an employee of and stockholder in TCH, and Paul Bergmann has consulted for the company. The remaining authors had no relevant disclosures.
2. Gallstone Disease Boosts Heart Risk
To take the posttest, go to: http://bit.ly/2c7TP7D
Expires August 18, 2017
VITALS
Key clinical point:
Gallstone disease is associated with an increased risk for coronary heart disease; preventing the former can help mitigate chances of developing the latter.
Major finding:
A meta-analysis revealed a 23% increased chance of CHD in gallstone disease patients.
Data source:
A meta-analysis of seven studies involving 842,553 subjects, and a prospective cohort study of 269,142 participants in three separate studies that took place from 1980 to 2011.
Disclosures:
Funding provided by NIH, Boston Obesity Nutrition Research Center, and United States-Israel Binational Science Foundation. The authors had no relevant financial disclosures.
3. New HER2-testing Guidelines Result in More Women Eligible for Directed Treatment
To take the posttest, go to: http://bit.ly/2cd9llO
Expires July 25, 2017
VITALS
Key clinical point:
New IHC and FISH pathology guidelines categorize more breast cancers as "equivocal" regarding HER2 positivity and ultimately lead to identifying more of them as HER2 positive.
Major finding:
By using 2013 guidelines, 358 additional tumors were interpreted as positive, compared with the 2007 guidelines and 298 additional tumors were considered positive, compared with the FDA criteria.
Data source:
A cohort study involving 2,851 breast cancer samples analyzed according to three different pathology guidelines during a 1-year period.
Disclosures:
This study was supported by the Mayo Clinic. Dr. Shah reported having no relevant financial disclosures; his associates reported ties to Merck, Hospira, Ariad Pharmaceuticals, Abbott Molecular, and Genome Diagnostics.
4. Extreme Alcohol Use Worsens HIV Disease
To take the posttest, go to: http://bit.ly/2coIzG3
Expires August 14, 2017
VITALS
Key clinical point:
A pattern of heavy alcohol use over time in HIV-infected patients was associated with accelerated HIV disease progression.
Major finding:
Long-term heavy alcohol use by middle-aged, HIV-infected military veterans was associated with a 1.83-fold increased likelihood of also being in the highest-risk group for accelerated progression of HIV disease.
Data source:
This study included 3,539 U.S. military veterans receiving care for HIV infection at eight VA centers. The impact of their long-term pattern of alcohol use on HIV disease progression was assessed over an 8-year period by annual assessments using validated instruments.
Disclosures:
The presenter reported having no financial conflicts of interest regarding the study, funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute of Allergy and Infectious Diseases.
5. Weight Loss Boosts TNFis' Psoriatic Arthritis Efficacy
To take the posttest, go to: http://bit.ly/2chD4M1
Expires July 23, 2017
Here are 5 articles in the October issue of Clinician Reviews (individual articles are valid for one year from date of publication—expiration dates below):
1. Autism Follow-up Screening by PCPs Yields High Accuracy
To take the posttest, go to: http://bit.ly/2bTLhFS
Expires August 19, 2017
VITALS
Key clinical point:
Primary care providers can conduct the M-CHAT/F following a positive M-CHAT screening for autism spectrum disorders.
Major finding:
Primary care providers and trained interviewers agreed 86.6% of the time on the screening results of the M-CHAT/F for ASDs.
Data source:
A cohort study of 5,071 children, mean age 23 months, screened with the M-CHAT, and a subsequent 197 children screened with the M-CHAT/F in 22 Maryland primary care practices.
Disclosures:
The National Institutes of Mental Health funded the research. Dr. Sturner is director of Total Child Health (TCH), a for-profit subsidiary of the Center for Promotion of Child Development through Primary Care, which conducted the study. Barbara Howard, MD, is president of TCH. Tanya Morrel, PhD, is an employee of and stockholder in TCH, and Paul Bergmann has consulted for the company. The remaining authors had no relevant disclosures.
2. Gallstone Disease Boosts Heart Risk
To take the posttest, go to: http://bit.ly/2c7TP7D
Expires August 18, 2017
VITALS
Key clinical point:
Gallstone disease is associated with an increased risk for coronary heart disease; preventing the former can help mitigate chances of developing the latter.
Major finding:
A meta-analysis revealed a 23% increased chance of CHD in gallstone disease patients.
Data source:
A meta-analysis of seven studies involving 842,553 subjects, and a prospective cohort study of 269,142 participants in three separate studies that took place from 1980 to 2011.
Disclosures:
Funding provided by NIH, Boston Obesity Nutrition Research Center, and United States-Israel Binational Science Foundation. The authors had no relevant financial disclosures.
3. New HER2-testing Guidelines Result in More Women Eligible for Directed Treatment
To take the posttest, go to: http://bit.ly/2cd9llO
Expires July 25, 2017
VITALS
Key clinical point:
New IHC and FISH pathology guidelines categorize more breast cancers as "equivocal" regarding HER2 positivity and ultimately lead to identifying more of them as HER2 positive.
Major finding:
By using 2013 guidelines, 358 additional tumors were interpreted as positive, compared with the 2007 guidelines and 298 additional tumors were considered positive, compared with the FDA criteria.
Data source:
A cohort study involving 2,851 breast cancer samples analyzed according to three different pathology guidelines during a 1-year period.
Disclosures:
This study was supported by the Mayo Clinic. Dr. Shah reported having no relevant financial disclosures; his associates reported ties to Merck, Hospira, Ariad Pharmaceuticals, Abbott Molecular, and Genome Diagnostics.
4. Extreme Alcohol Use Worsens HIV Disease
To take the posttest, go to: http://bit.ly/2coIzG3
Expires August 14, 2017
VITALS
Key clinical point:
A pattern of heavy alcohol use over time in HIV-infected patients was associated with accelerated HIV disease progression.
Major finding:
Long-term heavy alcohol use by middle-aged, HIV-infected military veterans was associated with a 1.83-fold increased likelihood of also being in the highest-risk group for accelerated progression of HIV disease.
Data source:
This study included 3,539 U.S. military veterans receiving care for HIV infection at eight VA centers. The impact of their long-term pattern of alcohol use on HIV disease progression was assessed over an 8-year period by annual assessments using validated instruments.
Disclosures:
The presenter reported having no financial conflicts of interest regarding the study, funded by the National Institute on Alcohol Abuse and Alcoholism and the National Institute of Allergy and Infectious Diseases.
5. Weight Loss Boosts TNFis' Psoriatic Arthritis Efficacy
To take the posttest, go to: http://bit.ly/2chD4M1
Expires July 23, 2017
Early epilepsy increases risk of later comorbid ADHD in autism
VIENNA – Early-onset idiopathic epilepsy occurring before age 7 years nearly doubles the likelihood that a child with autism spectrum disorder will later develop comorbid attention-deficit/hyperactivity disorder, Johnny Downs, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Comorbid ADHD is common in the setting of autism spectrum disorder (ASD). In a search for risk factors for the comorbid condition, he and his coinvestigators reviewed the physical health records prior to age 7 years of 3,032 patients with ASD referred at ages 3-17 years to child and adolescent mental health services clinics serving South London.
“That’s information that often doesn’t make it into the clinical psychiatric record,” noted Dr. Downs, a child psychiatrist at King’s College London.
Half of the 3,032 subjects were diagnosed with ASD at age 6-12 years and another 39% at age 13-17. During 5 years of prospective follow-up after being diagnosed with ASD in this longitudinal observational study, 25.5% of patients were diagnosed with comorbid ADHD. Looking back through the early physical health records, 114 (3.76%) of study participants had experienced early-onset epilepsy before age 7 years.
This large sample size allowed for robust multivariate adjustment for potential confounders. In a multivariate analysis, ASD patients with a history of early-onset epilepsy were at a significant 1.75-fold increased risk for subsequent comorbid ADHD. The analysis was adjusted for family history of epilepsy, sociodemographic factors, intellectual disability, previous head injury, perinatal complications, central nervous system tumors, early meningitis, and other confounders.
“The take-home message would be if you’ve got social and communication difficulties in a young child appearing at the age of 5, 6, or 7 [years], and there’s a history of seizures, we are seeing from observational data that the child is at increased risk of ADHD over the age of 7,” Dr. Downs said in an interview.
Compared with white subjects with ASD, the risk of developing comorbid ADHD was reduced by 37% in black and by 52% in Asian patients with ASD.
He plans further studies aimed at determining whether conventional ADHD management strategies have the same risk/benefit ratios in children with ASD and comorbid ADHD as in those with ADHD alone.
Dr. Downs reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
VIENNA – Early-onset idiopathic epilepsy occurring before age 7 years nearly doubles the likelihood that a child with autism spectrum disorder will later develop comorbid attention-deficit/hyperactivity disorder, Johnny Downs, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Comorbid ADHD is common in the setting of autism spectrum disorder (ASD). In a search for risk factors for the comorbid condition, he and his coinvestigators reviewed the physical health records prior to age 7 years of 3,032 patients with ASD referred at ages 3-17 years to child and adolescent mental health services clinics serving South London.
“That’s information that often doesn’t make it into the clinical psychiatric record,” noted Dr. Downs, a child psychiatrist at King’s College London.
Half of the 3,032 subjects were diagnosed with ASD at age 6-12 years and another 39% at age 13-17. During 5 years of prospective follow-up after being diagnosed with ASD in this longitudinal observational study, 25.5% of patients were diagnosed with comorbid ADHD. Looking back through the early physical health records, 114 (3.76%) of study participants had experienced early-onset epilepsy before age 7 years.
This large sample size allowed for robust multivariate adjustment for potential confounders. In a multivariate analysis, ASD patients with a history of early-onset epilepsy were at a significant 1.75-fold increased risk for subsequent comorbid ADHD. The analysis was adjusted for family history of epilepsy, sociodemographic factors, intellectual disability, previous head injury, perinatal complications, central nervous system tumors, early meningitis, and other confounders.
“The take-home message would be if you’ve got social and communication difficulties in a young child appearing at the age of 5, 6, or 7 [years], and there’s a history of seizures, we are seeing from observational data that the child is at increased risk of ADHD over the age of 7,” Dr. Downs said in an interview.
Compared with white subjects with ASD, the risk of developing comorbid ADHD was reduced by 37% in black and by 52% in Asian patients with ASD.
He plans further studies aimed at determining whether conventional ADHD management strategies have the same risk/benefit ratios in children with ASD and comorbid ADHD as in those with ADHD alone.
Dr. Downs reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
VIENNA – Early-onset idiopathic epilepsy occurring before age 7 years nearly doubles the likelihood that a child with autism spectrum disorder will later develop comorbid attention-deficit/hyperactivity disorder, Johnny Downs, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
Comorbid ADHD is common in the setting of autism spectrum disorder (ASD). In a search for risk factors for the comorbid condition, he and his coinvestigators reviewed the physical health records prior to age 7 years of 3,032 patients with ASD referred at ages 3-17 years to child and adolescent mental health services clinics serving South London.
“That’s information that often doesn’t make it into the clinical psychiatric record,” noted Dr. Downs, a child psychiatrist at King’s College London.
Half of the 3,032 subjects were diagnosed with ASD at age 6-12 years and another 39% at age 13-17. During 5 years of prospective follow-up after being diagnosed with ASD in this longitudinal observational study, 25.5% of patients were diagnosed with comorbid ADHD. Looking back through the early physical health records, 114 (3.76%) of study participants had experienced early-onset epilepsy before age 7 years.
This large sample size allowed for robust multivariate adjustment for potential confounders. In a multivariate analysis, ASD patients with a history of early-onset epilepsy were at a significant 1.75-fold increased risk for subsequent comorbid ADHD. The analysis was adjusted for family history of epilepsy, sociodemographic factors, intellectual disability, previous head injury, perinatal complications, central nervous system tumors, early meningitis, and other confounders.
“The take-home message would be if you’ve got social and communication difficulties in a young child appearing at the age of 5, 6, or 7 [years], and there’s a history of seizures, we are seeing from observational data that the child is at increased risk of ADHD over the age of 7,” Dr. Downs said in an interview.
Compared with white subjects with ASD, the risk of developing comorbid ADHD was reduced by 37% in black and by 52% in Asian patients with ASD.
He plans further studies aimed at determining whether conventional ADHD management strategies have the same risk/benefit ratios in children with ASD and comorbid ADHD as in those with ADHD alone.
Dr. Downs reported having no financial conflicts of interest regarding his study, which was conducted free of commercial support.
AT THE ECNP CONGRESS
Key clinical point: Youths with autism spectrum disorder and a history of early-onset epilepsy before age 7 years are at an increased risk of subsequent comorbid ADHD.
Major finding: Youths with autism spectrum disorder who have a history of early-onset epilepsy before age 7 years are at 1.75-fold increased likelihood of subsequently developing comorbid ADHD.
Data source: This longitudinal study included 3,032 children and adolescents with autism spectrum disorder, 26% of whom developed ADHD during 5 years of prospective follow-up.
Disclosures: The presenter reported having no financial conflicts of interest regarding this study, which was conducted free of commercial support.