User login
Is stem-cell transplant curative for HIV infection?
DURBAN, SOUTH AFRICA – The 15 HIV-infected patients who have undergone allogeneic stem-cell transplant for life-threatening hematologic cancers under the auspices of the European EpiStem Consortium have uniformly demonstrated a profound and durable reduction in viral reservoir to a degree that hasn’t been approached by any other investigational cure strategy, Annemarie Wensing, MD, said at the 21st International AIDS Conference.
“We see an enormous reduction in the viral reservoir, and in two patients we cannot find any viable HIV in the blood using ultrasensitive tests. But we don’t know whether these patients are cured because they are still on antiretroviral therapy,” said Dr. Wensing of Utrecht (The Netherlands) University.
Non-Hodgkin’s lymphoma and Hodgkin’s lymphoma are 7-9 times more frequent in HIV-positive patients than in the general population. But allogeneic stem cell transplantation is an even higher-risk treatment in HIV-positive patients with life-threatening leukemia or lymphoma than in the HIV-negative population. Only 6 of the 15 EuroStem patients remain alive. Eight died within 4 months of the procedure and another died 2.5 years post-transplant, all from progression of their cancer or as a result of opportunistic infections arising during the immunosuppressive chemoablation that’s central to stem-cell transplantation. However, 3 of the 15 patients have survived longer than 3 years. In two of them, no HIV can be detected in blood or intestinal tissue using ultrasensitive tests, while in the third there is “only a slight trace,” according to Dr. Wensing, a clinical virologist.
EpiStem (the European Project to Guide and Investigate the Potential for HIV Cure by Stem-Cell Transplantation) is a multinational collaboration of European oncologists, infectious disease physicians, and other specialists. It was formed in response to the successful outcome of allogeneic stem cell transplantation for acute myeloid leukemia in HIV-positive Timothy Brown, more famously known as “the Berlin patient” (N Engl J Med. 2009 Feb 12;360(7):692-8). He has thus far survived 7 years off antiretroviral therapy.
Much has been made of the fact that Mr. Brown’s donor cells were homozygous for the CCR5 delta32 mutation, which confers natural resistance to HIV infection because it prevents the virus from infecting T cells. Only 1% or less of the population is homozygous for this mutation. But Dr. Wensing isn’t convinced that using donor cells with the mutation is a prerequisite for success. Indeed, while 4 of the 15 EpiStem patients received stem cells from donors homozygous for the mutation and another got donor cells heterozygous for the CCR5 delta32 mutation, the other 10 received stem cells capable of being infected by HIV – yet all 15 experienced an enormous reduction in their viral reservoir. And two of the three patients who have survived longer than 3 years got stem cells without the CCR5 delta32 mutation.
Dr. Wensing observed that a common denominator shared by Timothy Brown and the two EpiStem patients who have trace or undetectable HIV in blood or tissue samples more than 3 years post-transplant is that all three developed severe graft-versus-host disease in conjunction with their stem cell transplantation. She suspects this may have helped them to clear the infection, a hypothesis she intends to pursue further as EpiStem gathers more patients.
Eventually, if patients continue to test negative for HIV using ultrasensitive tests, it will be time to have a discussion with patients and their treating physicians as to whether they should continue on antiretroviral therapy.
“In the end it’s the patients’ decision, but they should be very well counseled because it can have medical and also psychological consequences if HIV returns,” she said.
EpiStem is funded by the American Foundation for AIDS Research Conssortium on HIV Eradication. Dr. Wensing reported having no financial conflicts regarding her presentation.
DURBAN, SOUTH AFRICA – The 15 HIV-infected patients who have undergone allogeneic stem-cell transplant for life-threatening hematologic cancers under the auspices of the European EpiStem Consortium have uniformly demonstrated a profound and durable reduction in viral reservoir to a degree that hasn’t been approached by any other investigational cure strategy, Annemarie Wensing, MD, said at the 21st International AIDS Conference.
“We see an enormous reduction in the viral reservoir, and in two patients we cannot find any viable HIV in the blood using ultrasensitive tests. But we don’t know whether these patients are cured because they are still on antiretroviral therapy,” said Dr. Wensing of Utrecht (The Netherlands) University.
Non-Hodgkin’s lymphoma and Hodgkin’s lymphoma are 7-9 times more frequent in HIV-positive patients than in the general population. But allogeneic stem cell transplantation is an even higher-risk treatment in HIV-positive patients with life-threatening leukemia or lymphoma than in the HIV-negative population. Only 6 of the 15 EuroStem patients remain alive. Eight died within 4 months of the procedure and another died 2.5 years post-transplant, all from progression of their cancer or as a result of opportunistic infections arising during the immunosuppressive chemoablation that’s central to stem-cell transplantation. However, 3 of the 15 patients have survived longer than 3 years. In two of them, no HIV can be detected in blood or intestinal tissue using ultrasensitive tests, while in the third there is “only a slight trace,” according to Dr. Wensing, a clinical virologist.
EpiStem (the European Project to Guide and Investigate the Potential for HIV Cure by Stem-Cell Transplantation) is a multinational collaboration of European oncologists, infectious disease physicians, and other specialists. It was formed in response to the successful outcome of allogeneic stem cell transplantation for acute myeloid leukemia in HIV-positive Timothy Brown, more famously known as “the Berlin patient” (N Engl J Med. 2009 Feb 12;360(7):692-8). He has thus far survived 7 years off antiretroviral therapy.
Much has been made of the fact that Mr. Brown’s donor cells were homozygous for the CCR5 delta32 mutation, which confers natural resistance to HIV infection because it prevents the virus from infecting T cells. Only 1% or less of the population is homozygous for this mutation. But Dr. Wensing isn’t convinced that using donor cells with the mutation is a prerequisite for success. Indeed, while 4 of the 15 EpiStem patients received stem cells from donors homozygous for the mutation and another got donor cells heterozygous for the CCR5 delta32 mutation, the other 10 received stem cells capable of being infected by HIV – yet all 15 experienced an enormous reduction in their viral reservoir. And two of the three patients who have survived longer than 3 years got stem cells without the CCR5 delta32 mutation.
Dr. Wensing observed that a common denominator shared by Timothy Brown and the two EpiStem patients who have trace or undetectable HIV in blood or tissue samples more than 3 years post-transplant is that all three developed severe graft-versus-host disease in conjunction with their stem cell transplantation. She suspects this may have helped them to clear the infection, a hypothesis she intends to pursue further as EpiStem gathers more patients.
Eventually, if patients continue to test negative for HIV using ultrasensitive tests, it will be time to have a discussion with patients and their treating physicians as to whether they should continue on antiretroviral therapy.
“In the end it’s the patients’ decision, but they should be very well counseled because it can have medical and also psychological consequences if HIV returns,” she said.
EpiStem is funded by the American Foundation for AIDS Research Conssortium on HIV Eradication. Dr. Wensing reported having no financial conflicts regarding her presentation.
DURBAN, SOUTH AFRICA – The 15 HIV-infected patients who have undergone allogeneic stem-cell transplant for life-threatening hematologic cancers under the auspices of the European EpiStem Consortium have uniformly demonstrated a profound and durable reduction in viral reservoir to a degree that hasn’t been approached by any other investigational cure strategy, Annemarie Wensing, MD, said at the 21st International AIDS Conference.
“We see an enormous reduction in the viral reservoir, and in two patients we cannot find any viable HIV in the blood using ultrasensitive tests. But we don’t know whether these patients are cured because they are still on antiretroviral therapy,” said Dr. Wensing of Utrecht (The Netherlands) University.
Non-Hodgkin’s lymphoma and Hodgkin’s lymphoma are 7-9 times more frequent in HIV-positive patients than in the general population. But allogeneic stem cell transplantation is an even higher-risk treatment in HIV-positive patients with life-threatening leukemia or lymphoma than in the HIV-negative population. Only 6 of the 15 EuroStem patients remain alive. Eight died within 4 months of the procedure and another died 2.5 years post-transplant, all from progression of their cancer or as a result of opportunistic infections arising during the immunosuppressive chemoablation that’s central to stem-cell transplantation. However, 3 of the 15 patients have survived longer than 3 years. In two of them, no HIV can be detected in blood or intestinal tissue using ultrasensitive tests, while in the third there is “only a slight trace,” according to Dr. Wensing, a clinical virologist.
EpiStem (the European Project to Guide and Investigate the Potential for HIV Cure by Stem-Cell Transplantation) is a multinational collaboration of European oncologists, infectious disease physicians, and other specialists. It was formed in response to the successful outcome of allogeneic stem cell transplantation for acute myeloid leukemia in HIV-positive Timothy Brown, more famously known as “the Berlin patient” (N Engl J Med. 2009 Feb 12;360(7):692-8). He has thus far survived 7 years off antiretroviral therapy.
Much has been made of the fact that Mr. Brown’s donor cells were homozygous for the CCR5 delta32 mutation, which confers natural resistance to HIV infection because it prevents the virus from infecting T cells. Only 1% or less of the population is homozygous for this mutation. But Dr. Wensing isn’t convinced that using donor cells with the mutation is a prerequisite for success. Indeed, while 4 of the 15 EpiStem patients received stem cells from donors homozygous for the mutation and another got donor cells heterozygous for the CCR5 delta32 mutation, the other 10 received stem cells capable of being infected by HIV – yet all 15 experienced an enormous reduction in their viral reservoir. And two of the three patients who have survived longer than 3 years got stem cells without the CCR5 delta32 mutation.
Dr. Wensing observed that a common denominator shared by Timothy Brown and the two EpiStem patients who have trace or undetectable HIV in blood or tissue samples more than 3 years post-transplant is that all three developed severe graft-versus-host disease in conjunction with their stem cell transplantation. She suspects this may have helped them to clear the infection, a hypothesis she intends to pursue further as EpiStem gathers more patients.
Eventually, if patients continue to test negative for HIV using ultrasensitive tests, it will be time to have a discussion with patients and their treating physicians as to whether they should continue on antiretroviral therapy.
“In the end it’s the patients’ decision, but they should be very well counseled because it can have medical and also psychological consequences if HIV returns,” she said.
EpiStem is funded by the American Foundation for AIDS Research Conssortium on HIV Eradication. Dr. Wensing reported having no financial conflicts regarding her presentation.
AT AIDS 2016
Key clinical point: It doesn’t appear to be necessary to use donor stem cells that are homozygous for the CCR5 delta32 mutation to achieve enormous sustained reductions in the viral reservoir in HIV-infected patients undergoing allogeneic stem cell transplantation for hematologic cancers.
Major finding: Two of three patients in a European series who have survived for longer than 3 years after stem-cell transplantation with undetectable or only trace HIV in their blood received donor cells lacking the rare CCR5 delta32 mutation.
Data source: EpiStem is an ongoing observational study of HIV-infected patients who undergo allogeneic stem cell transplantation for life-threatening hematologic cancers.
Disclosures: The EpiStem project is funded by the American Foundation for AIDS Research Conssortium on HIV Eradication. The presenter reported having no financial conflicts regarding her presentation.
Maraviroc shows potential for HIV PrEP in women
DURBAN, SOUTH AFRICA – Maraviroc-containing regimens for daily oral pre-exposure prophylaxis in women at risk for HIV infection showed good safety and tolerability in a phase 2 study, the first randomized trial of PrEP ever conducted in U.S. women, Roy M. Gulick, MD, reported at the 21st International AIDS Conference.
No new HIV infections occurred in the 188 women who participated in the 48-week, randomized, double-blind, placebo-controlled study known as the HPTN 069/ACTG A5305 trial.
However, the results shouldn’t be taken as evidence of efficacy. The relatively low 2% incidence of new STIs diagnosed during 48 weeks of close followup – three cases of chlamydia, one of gonorrhea – suggests that the study population probably wasn’t at high risk for acquiring HIV. Further, the study wasn’t powered to determine efficacy. That determination will have to await a larger phase 3 trial, observed Dr. Gulick, professor of medicine at Cornell University in New York.
Maraviroc (Selzentry) is categorized as an HIV entry inhibitor. It’s an antagonist of the CCR5 receptor found on the surface of T cells, which is the route of HIV infection. The rationale for exploring the drug for HIV PrEP, according to Dr. Gulick, is that it concentrates in both the genital tract and rectum, doesn’t select for drug-resistant viral strains, is well tolerated, and it isn’t commonly used for treatment of HIV infection.
Additional options for oral daily HIV PrEP are clearly desirable, he added. The only approved agent is Truvada (tenofovir/emtricitabine), which is often used in HIV therapy as well, and there is concern it may select for drug resistance. Plus, it has renal, GI, and bone side effects.
Study participants were HIV-negative adult women who were born female and considered at risk for HIV acquisition because of a history of condomless vaginal or anal intercourse with at least one HIV-positive or unknown status man within the previous 90 days. The women were randomized to one of four study arms: maraviroc at the standard dose of 300 mg/day plus two placebo pills; maraviroc plus emtricitabine at the standard dose of 200 mg/day plus one placebo pill; maraviroc plus tenofovir at 300 mg/day plus a placebo pill, or a control regimen of fixed-dose Truvada (tenofovir 300 mg/emtricitabine 200 mg) plus two placebo pills. Thus, everyone took three pills once daily.
The three-pill regimen might help explain the less than stellar patient adherence. Study drugs were detectable – and not necessarily at therapeutic levels – in the plasma of 65% of subjects at 24 weeks and 60% at 48 weeks, with no differences between the study arms.
Maraviroc alone was associated with fewer grade 2-4 adverse events than the other regimens.
There were 11 grade 3 or 4 adverse events deemed by investigators to be related to study drugs. They included abnormal weight loss, depression, hypophosphatemia, a rise in LDL cholesterol, headache, vitamin D deficiency, back pain, two spontaneous abortions, and two dissimilar cases of congenital anomaly, with no obvious differences between the study groups in rate or pattern. Rates of specific renal and GI toxicities were comparable across the four study arms.
Earlier in 2016, Dr. Gulick presented the results of the men’s arm of HPTN 069/ACTG A5305, a parallel 48-week randomized trial in 406 men who have sex with men. Five men in the maraviroc monotherapy arm seroconverted during the 48-week study, for an incidence of 1.4%. All had no or low plasma drug concentrations, and all five were infected with HIV lacking antiretroviral drug resistance.
Dr. Gulick and coinvestigators plan to present the findings of an analysis of rectal and vaginal biopsies from 42 women in the trial, along with a bone mineral density substudy in 200 men and 200 women men in the trial, plus detailed quality-of-life, behavioral, and adherence data in the full men’s and women’s cohorts.
The trial was sponsored by the HIV Prevention Trials Network and the AIDS Clinical Trials Group with funding from the National Institute of Allergic and Infectious Diseases. Dr. Gulick reported having no financial conflicts of interest.
DURBAN, SOUTH AFRICA – Maraviroc-containing regimens for daily oral pre-exposure prophylaxis in women at risk for HIV infection showed good safety and tolerability in a phase 2 study, the first randomized trial of PrEP ever conducted in U.S. women, Roy M. Gulick, MD, reported at the 21st International AIDS Conference.
No new HIV infections occurred in the 188 women who participated in the 48-week, randomized, double-blind, placebo-controlled study known as the HPTN 069/ACTG A5305 trial.
However, the results shouldn’t be taken as evidence of efficacy. The relatively low 2% incidence of new STIs diagnosed during 48 weeks of close followup – three cases of chlamydia, one of gonorrhea – suggests that the study population probably wasn’t at high risk for acquiring HIV. Further, the study wasn’t powered to determine efficacy. That determination will have to await a larger phase 3 trial, observed Dr. Gulick, professor of medicine at Cornell University in New York.
Maraviroc (Selzentry) is categorized as an HIV entry inhibitor. It’s an antagonist of the CCR5 receptor found on the surface of T cells, which is the route of HIV infection. The rationale for exploring the drug for HIV PrEP, according to Dr. Gulick, is that it concentrates in both the genital tract and rectum, doesn’t select for drug-resistant viral strains, is well tolerated, and it isn’t commonly used for treatment of HIV infection.
Additional options for oral daily HIV PrEP are clearly desirable, he added. The only approved agent is Truvada (tenofovir/emtricitabine), which is often used in HIV therapy as well, and there is concern it may select for drug resistance. Plus, it has renal, GI, and bone side effects.
Study participants were HIV-negative adult women who were born female and considered at risk for HIV acquisition because of a history of condomless vaginal or anal intercourse with at least one HIV-positive or unknown status man within the previous 90 days. The women were randomized to one of four study arms: maraviroc at the standard dose of 300 mg/day plus two placebo pills; maraviroc plus emtricitabine at the standard dose of 200 mg/day plus one placebo pill; maraviroc plus tenofovir at 300 mg/day plus a placebo pill, or a control regimen of fixed-dose Truvada (tenofovir 300 mg/emtricitabine 200 mg) plus two placebo pills. Thus, everyone took three pills once daily.
The three-pill regimen might help explain the less than stellar patient adherence. Study drugs were detectable – and not necessarily at therapeutic levels – in the plasma of 65% of subjects at 24 weeks and 60% at 48 weeks, with no differences between the study arms.
Maraviroc alone was associated with fewer grade 2-4 adverse events than the other regimens.
There were 11 grade 3 or 4 adverse events deemed by investigators to be related to study drugs. They included abnormal weight loss, depression, hypophosphatemia, a rise in LDL cholesterol, headache, vitamin D deficiency, back pain, two spontaneous abortions, and two dissimilar cases of congenital anomaly, with no obvious differences between the study groups in rate or pattern. Rates of specific renal and GI toxicities were comparable across the four study arms.
Earlier in 2016, Dr. Gulick presented the results of the men’s arm of HPTN 069/ACTG A5305, a parallel 48-week randomized trial in 406 men who have sex with men. Five men in the maraviroc monotherapy arm seroconverted during the 48-week study, for an incidence of 1.4%. All had no or low plasma drug concentrations, and all five were infected with HIV lacking antiretroviral drug resistance.
Dr. Gulick and coinvestigators plan to present the findings of an analysis of rectal and vaginal biopsies from 42 women in the trial, along with a bone mineral density substudy in 200 men and 200 women men in the trial, plus detailed quality-of-life, behavioral, and adherence data in the full men’s and women’s cohorts.
The trial was sponsored by the HIV Prevention Trials Network and the AIDS Clinical Trials Group with funding from the National Institute of Allergic and Infectious Diseases. Dr. Gulick reported having no financial conflicts of interest.
DURBAN, SOUTH AFRICA – Maraviroc-containing regimens for daily oral pre-exposure prophylaxis in women at risk for HIV infection showed good safety and tolerability in a phase 2 study, the first randomized trial of PrEP ever conducted in U.S. women, Roy M. Gulick, MD, reported at the 21st International AIDS Conference.
No new HIV infections occurred in the 188 women who participated in the 48-week, randomized, double-blind, placebo-controlled study known as the HPTN 069/ACTG A5305 trial.
However, the results shouldn’t be taken as evidence of efficacy. The relatively low 2% incidence of new STIs diagnosed during 48 weeks of close followup – three cases of chlamydia, one of gonorrhea – suggests that the study population probably wasn’t at high risk for acquiring HIV. Further, the study wasn’t powered to determine efficacy. That determination will have to await a larger phase 3 trial, observed Dr. Gulick, professor of medicine at Cornell University in New York.
Maraviroc (Selzentry) is categorized as an HIV entry inhibitor. It’s an antagonist of the CCR5 receptor found on the surface of T cells, which is the route of HIV infection. The rationale for exploring the drug for HIV PrEP, according to Dr. Gulick, is that it concentrates in both the genital tract and rectum, doesn’t select for drug-resistant viral strains, is well tolerated, and it isn’t commonly used for treatment of HIV infection.
Additional options for oral daily HIV PrEP are clearly desirable, he added. The only approved agent is Truvada (tenofovir/emtricitabine), which is often used in HIV therapy as well, and there is concern it may select for drug resistance. Plus, it has renal, GI, and bone side effects.
Study participants were HIV-negative adult women who were born female and considered at risk for HIV acquisition because of a history of condomless vaginal or anal intercourse with at least one HIV-positive or unknown status man within the previous 90 days. The women were randomized to one of four study arms: maraviroc at the standard dose of 300 mg/day plus two placebo pills; maraviroc plus emtricitabine at the standard dose of 200 mg/day plus one placebo pill; maraviroc plus tenofovir at 300 mg/day plus a placebo pill, or a control regimen of fixed-dose Truvada (tenofovir 300 mg/emtricitabine 200 mg) plus two placebo pills. Thus, everyone took three pills once daily.
The three-pill regimen might help explain the less than stellar patient adherence. Study drugs were detectable – and not necessarily at therapeutic levels – in the plasma of 65% of subjects at 24 weeks and 60% at 48 weeks, with no differences between the study arms.
Maraviroc alone was associated with fewer grade 2-4 adverse events than the other regimens.
There were 11 grade 3 or 4 adverse events deemed by investigators to be related to study drugs. They included abnormal weight loss, depression, hypophosphatemia, a rise in LDL cholesterol, headache, vitamin D deficiency, back pain, two spontaneous abortions, and two dissimilar cases of congenital anomaly, with no obvious differences between the study groups in rate or pattern. Rates of specific renal and GI toxicities were comparable across the four study arms.
Earlier in 2016, Dr. Gulick presented the results of the men’s arm of HPTN 069/ACTG A5305, a parallel 48-week randomized trial in 406 men who have sex with men. Five men in the maraviroc monotherapy arm seroconverted during the 48-week study, for an incidence of 1.4%. All had no or low plasma drug concentrations, and all five were infected with HIV lacking antiretroviral drug resistance.
Dr. Gulick and coinvestigators plan to present the findings of an analysis of rectal and vaginal biopsies from 42 women in the trial, along with a bone mineral density substudy in 200 men and 200 women men in the trial, plus detailed quality-of-life, behavioral, and adherence data in the full men’s and women’s cohorts.
The trial was sponsored by the HIV Prevention Trials Network and the AIDS Clinical Trials Group with funding from the National Institute of Allergic and Infectious Diseases. Dr. Gulick reported having no financial conflicts of interest.
AT AIDS 2016
Key clinical point: Maraviroc shows promise as an alternative to Truvada for HIV pre-exposure prophylaxis.
Major finding: No new HIV infections occurred in at-risk women in a 48-week study of daily oral HIV pre-exposure prophylaxis comparing three maraviroc-containing regimens and tenofovir/emtricitabine.
Data source: This was a randomized, double-blind, placebo-controlled, multicenter, 48-week clinical trial involving 188 women at risk for HIV infection.
Disclosures: The trial was sponsored by the HIV Prevention Trials Network and the AIDS Clinical Trials Group with funding from the National Institute of Allergy and Infectious Diseases. The presenter reported having no financial conflicts of interest.
Novel treatment strategy to reduce infarct size in STEMI
ROME – Early administration of high-dose N-acetylcysteine and low-dose glyceryl trinitrate in ST-segment elevation acute myocardial infarction (STEMI) patients undergoing primary percutaneous coronary intervention (PCI) achieved a 30% reduction in myocardial infarct size, compared with placebo in a randomized, double-blind, multicenter clinical trial, Sivabaskari Pasupathy reported at the annual congress of the European Society of Cardiology.
The adjunctive regimen also resulted in a doubling of myocardial salvage, the secondary endpoint in the NACIAM (N-acetylcysteine in acute myocardial infarction) trial, said Ms. Pasupathy of the University of Adelaide, Australia.
NACIAM, which she stressed was a pilot study, included 75 randomized STEMI patients with follow-up cardiac MRIs obtained at about 5 days post-PCI, most of whom had another MRI at about 8 months.
All participants received IV glycerol trinitrate at a rate of 25 mcg/min for 48 hours starting in the emergency department. Patients in the active treatment arm received IV N-acetylcysteine (NAC) at 20 mg/min for the first hour and 10 mg/min for the next 47 hours. Controls got placebo at 40 mL/hour for the first hour and 20 mL/hour thereafter.
Cardiac MRI at 5 days showed a myocardial infarct size of 16.5% in the control group and 11% with active treatment. This absolute 5.5% reduction in infarct size in NAC recipients was achieved despite no difference between the two groups in myocardial area at risk by MRI, which was roughly 24%. Nor did the groups differ in rates of anterior MI or microvascular obstruction. The myocardial salvage rate was 60% in the NAC group and just 27% in controls.
Creatine kinase levels, measured hourly during the first 24 hours of hospitalization, were consistently lower in the NAC group, although the difference didn’t reach statistical significance.
In-hospital rates of hypotension, bleeding, or renal dysfunction were similar in the two study arms. However, at 2 years of follow-up the composite rate of death or cardiac rehospitalization was 27% in the control group, compared with 6% in patients who got NAC.
A key to the demonstrable success of the adjunctive NAC/nitrate treatment strategy was that it began early, in the emergency department, when patients initially presented with STEMI. The mean total ischemia time for study participants was 2.4 hours, and the shorter the duration of ischemia, the larger the reduction seen in infarct size, according to Ms. Pasupathy.
The rationale for this therapy is that NAC is thought to reduce oxidative stress by scavenging reactive oxygen species and inhibiting their release, thereby minimizing reperfusion injury. The Australian investigators also believe that NAC potentiates the effects of glycerol trinitrate in improving tissue reperfusion through enhanced vasodilation, inhibition of platelet aggregation, and dampening of inflammation. A reduction in reperfusion injury accompanied by increased tissue reperfusion could result in reduced infarct size, she explained.
John F. Beltrame, MD, lead investigator in the NACIAM study, noted that infarct size and myocardial salvage are surrogate endpoints. The investigators plan to conduct a larger trial with hard clinical outcomes.
“The key thing we need to do before we start using this in widespread fashion and changing practice guidelines is to look at clinical endpoints,” said Dr. Beltrame, professor of medicine at the University of Adelaide.
Nonetheless, he added, this therapy is given routinely in the setting of primary PCI for STEMI where he practices. That has been the case for 20 years because he and colleagues in his department did some of the early studies suggesting NAC was beneficial.
“Hospitals have different practices, and this is one of our particular practices,” the cardiologist said.
It all sounded pretty good to Jorge A. Belardi, MD, who co-chaired a press conference devoted to the NACIAM trial and other hotline presentations.
“It’s a very benign combination of medicines to use. So why not use it? We already use it to prevent contrast nephropathy. And there are no contraindications, to my knowledge,” said Dr. Belardi, director of the department of cardiology at the Buenos Aires Cardiovascular Institute.
Michel Ovize, MD, of the University of Lyon (France), the formal discussant designated for NACIAM, took a more flinty eyed view, noting that the study was small and previous studies of NAC during the past 2 decades have yielded conflicting results.
Ms. Pasupathy responded that a likely explanation for the disparate results is the fact that other investigators didn’t start NAC at the soonest possible time in the ED.
The NACIAM trial was funded by the Australian National Heart Foundation. Ms. Pasupathy and Dr. Beltrame reported having no financial conflicts of interest.
ROME – Early administration of high-dose N-acetylcysteine and low-dose glyceryl trinitrate in ST-segment elevation acute myocardial infarction (STEMI) patients undergoing primary percutaneous coronary intervention (PCI) achieved a 30% reduction in myocardial infarct size, compared with placebo in a randomized, double-blind, multicenter clinical trial, Sivabaskari Pasupathy reported at the annual congress of the European Society of Cardiology.
The adjunctive regimen also resulted in a doubling of myocardial salvage, the secondary endpoint in the NACIAM (N-acetylcysteine in acute myocardial infarction) trial, said Ms. Pasupathy of the University of Adelaide, Australia.
NACIAM, which she stressed was a pilot study, included 75 randomized STEMI patients with follow-up cardiac MRIs obtained at about 5 days post-PCI, most of whom had another MRI at about 8 months.
All participants received IV glycerol trinitrate at a rate of 25 mcg/min for 48 hours starting in the emergency department. Patients in the active treatment arm received IV N-acetylcysteine (NAC) at 20 mg/min for the first hour and 10 mg/min for the next 47 hours. Controls got placebo at 40 mL/hour for the first hour and 20 mL/hour thereafter.
Cardiac MRI at 5 days showed a myocardial infarct size of 16.5% in the control group and 11% with active treatment. This absolute 5.5% reduction in infarct size in NAC recipients was achieved despite no difference between the two groups in myocardial area at risk by MRI, which was roughly 24%. Nor did the groups differ in rates of anterior MI or microvascular obstruction. The myocardial salvage rate was 60% in the NAC group and just 27% in controls.
Creatine kinase levels, measured hourly during the first 24 hours of hospitalization, were consistently lower in the NAC group, although the difference didn’t reach statistical significance.
In-hospital rates of hypotension, bleeding, or renal dysfunction were similar in the two study arms. However, at 2 years of follow-up the composite rate of death or cardiac rehospitalization was 27% in the control group, compared with 6% in patients who got NAC.
A key to the demonstrable success of the adjunctive NAC/nitrate treatment strategy was that it began early, in the emergency department, when patients initially presented with STEMI. The mean total ischemia time for study participants was 2.4 hours, and the shorter the duration of ischemia, the larger the reduction seen in infarct size, according to Ms. Pasupathy.
The rationale for this therapy is that NAC is thought to reduce oxidative stress by scavenging reactive oxygen species and inhibiting their release, thereby minimizing reperfusion injury. The Australian investigators also believe that NAC potentiates the effects of glycerol trinitrate in improving tissue reperfusion through enhanced vasodilation, inhibition of platelet aggregation, and dampening of inflammation. A reduction in reperfusion injury accompanied by increased tissue reperfusion could result in reduced infarct size, she explained.
John F. Beltrame, MD, lead investigator in the NACIAM study, noted that infarct size and myocardial salvage are surrogate endpoints. The investigators plan to conduct a larger trial with hard clinical outcomes.
“The key thing we need to do before we start using this in widespread fashion and changing practice guidelines is to look at clinical endpoints,” said Dr. Beltrame, professor of medicine at the University of Adelaide.
Nonetheless, he added, this therapy is given routinely in the setting of primary PCI for STEMI where he practices. That has been the case for 20 years because he and colleagues in his department did some of the early studies suggesting NAC was beneficial.
“Hospitals have different practices, and this is one of our particular practices,” the cardiologist said.
It all sounded pretty good to Jorge A. Belardi, MD, who co-chaired a press conference devoted to the NACIAM trial and other hotline presentations.
“It’s a very benign combination of medicines to use. So why not use it? We already use it to prevent contrast nephropathy. And there are no contraindications, to my knowledge,” said Dr. Belardi, director of the department of cardiology at the Buenos Aires Cardiovascular Institute.
Michel Ovize, MD, of the University of Lyon (France), the formal discussant designated for NACIAM, took a more flinty eyed view, noting that the study was small and previous studies of NAC during the past 2 decades have yielded conflicting results.
Ms. Pasupathy responded that a likely explanation for the disparate results is the fact that other investigators didn’t start NAC at the soonest possible time in the ED.
The NACIAM trial was funded by the Australian National Heart Foundation. Ms. Pasupathy and Dr. Beltrame reported having no financial conflicts of interest.
ROME – Early administration of high-dose N-acetylcysteine and low-dose glyceryl trinitrate in ST-segment elevation acute myocardial infarction (STEMI) patients undergoing primary percutaneous coronary intervention (PCI) achieved a 30% reduction in myocardial infarct size, compared with placebo in a randomized, double-blind, multicenter clinical trial, Sivabaskari Pasupathy reported at the annual congress of the European Society of Cardiology.
The adjunctive regimen also resulted in a doubling of myocardial salvage, the secondary endpoint in the NACIAM (N-acetylcysteine in acute myocardial infarction) trial, said Ms. Pasupathy of the University of Adelaide, Australia.
NACIAM, which she stressed was a pilot study, included 75 randomized STEMI patients with follow-up cardiac MRIs obtained at about 5 days post-PCI, most of whom had another MRI at about 8 months.
All participants received IV glycerol trinitrate at a rate of 25 mcg/min for 48 hours starting in the emergency department. Patients in the active treatment arm received IV N-acetylcysteine (NAC) at 20 mg/min for the first hour and 10 mg/min for the next 47 hours. Controls got placebo at 40 mL/hour for the first hour and 20 mL/hour thereafter.
Cardiac MRI at 5 days showed a myocardial infarct size of 16.5% in the control group and 11% with active treatment. This absolute 5.5% reduction in infarct size in NAC recipients was achieved despite no difference between the two groups in myocardial area at risk by MRI, which was roughly 24%. Nor did the groups differ in rates of anterior MI or microvascular obstruction. The myocardial salvage rate was 60% in the NAC group and just 27% in controls.
Creatine kinase levels, measured hourly during the first 24 hours of hospitalization, were consistently lower in the NAC group, although the difference didn’t reach statistical significance.
In-hospital rates of hypotension, bleeding, or renal dysfunction were similar in the two study arms. However, at 2 years of follow-up the composite rate of death or cardiac rehospitalization was 27% in the control group, compared with 6% in patients who got NAC.
A key to the demonstrable success of the adjunctive NAC/nitrate treatment strategy was that it began early, in the emergency department, when patients initially presented with STEMI. The mean total ischemia time for study participants was 2.4 hours, and the shorter the duration of ischemia, the larger the reduction seen in infarct size, according to Ms. Pasupathy.
The rationale for this therapy is that NAC is thought to reduce oxidative stress by scavenging reactive oxygen species and inhibiting their release, thereby minimizing reperfusion injury. The Australian investigators also believe that NAC potentiates the effects of glycerol trinitrate in improving tissue reperfusion through enhanced vasodilation, inhibition of platelet aggregation, and dampening of inflammation. A reduction in reperfusion injury accompanied by increased tissue reperfusion could result in reduced infarct size, she explained.
John F. Beltrame, MD, lead investigator in the NACIAM study, noted that infarct size and myocardial salvage are surrogate endpoints. The investigators plan to conduct a larger trial with hard clinical outcomes.
“The key thing we need to do before we start using this in widespread fashion and changing practice guidelines is to look at clinical endpoints,” said Dr. Beltrame, professor of medicine at the University of Adelaide.
Nonetheless, he added, this therapy is given routinely in the setting of primary PCI for STEMI where he practices. That has been the case for 20 years because he and colleagues in his department did some of the early studies suggesting NAC was beneficial.
“Hospitals have different practices, and this is one of our particular practices,” the cardiologist said.
It all sounded pretty good to Jorge A. Belardi, MD, who co-chaired a press conference devoted to the NACIAM trial and other hotline presentations.
“It’s a very benign combination of medicines to use. So why not use it? We already use it to prevent contrast nephropathy. And there are no contraindications, to my knowledge,” said Dr. Belardi, director of the department of cardiology at the Buenos Aires Cardiovascular Institute.
Michel Ovize, MD, of the University of Lyon (France), the formal discussant designated for NACIAM, took a more flinty eyed view, noting that the study was small and previous studies of NAC during the past 2 decades have yielded conflicting results.
Ms. Pasupathy responded that a likely explanation for the disparate results is the fact that other investigators didn’t start NAC at the soonest possible time in the ED.
The NACIAM trial was funded by the Australian National Heart Foundation. Ms. Pasupathy and Dr. Beltrame reported having no financial conflicts of interest.
AT THE ESC CONGRESS 2016
Key clinical point: Early use of N-acetylcysteine with glyceryl trinitrate resulted in reduced myocardial infarct size in STEMI patients undergoing primary PCI.
Major finding: Early use of N-acetylcysteine with glyceryl trinitrate in STEMI patients undergoing primary PCI resulted in a myocardial infarct size of 11% as assessed on day 5, compared with 16.5% in placebo-treated controls.
Data source: The NACIAM trial was a 75-patient, randomized, double-blind, multicenter study.
Disclosures: The study was supported by the Australian National Heart Foundation. The presenter reported having no financial conflicts of interest.
Long-acting all-injectable HIV therapy successful in phase 2b
DURBAN, SOUTH AFRICA – Long-acting injectable antiretroviral therapy with cabotegravir and rilpivirine in nanosuspension successfully suppressed HIV-infected patients’ plasma viral load to fewer than 50 copies/mL for 48 weeks as maintenance therapy in the LATTE-2 trial, David A. Margolis, MD, reported at the 21st International AIDS Conference.
The intramuscular gluteal injections given every 4 or 8 weeks also proved safe and extremely well tolerated. At week 48, more than 97% of patients on injectable maintenance antiretroviral therapy (ART) reported a high degree of satisfaction with their treatment regimen on a structured questionnaire and expressed a willingness to continue on injectable therapy in the future, according to Dr. Margolis of Viiv Healthcare in Research Triangle Park, N.C.
Cabotegravir is an HIV integrase inhibitor, rilpivirine a nonnucleoside reverse transcriptase inhibitor. Rilpivirine is already approved as Edurant, a once-daily 25-mg tablet for oral therapy. Cabotegravir is under development as a 30-mg once-daily tablet. As oral agents, each has a half-life of roughly 40 hours. The two-drug oral combination showed a high degree of safety and efficacy through 96 weeks of follow-up in the LATTE-1 study, previously reported by Dr. Margolis (Lancet Infect Dis. 2015 Oct;15[10]):1145-55) .
In addition, the two antiretroviral agents are being developed as long-acting injectables with half-lives of 20-90 days. LATTE-2 was a phase IIb, open-label, multicenter study that began with 309 HIV-positive, ART-naive subjects who participated in a 20-week induction phase during which they received 30 mg/day of oral cabotegravir plus abacavir/lamivudine to induce HIV viral suppression. During the last 4 weeks of the induction phase, participants also received oral rilpivirine at 25 mg once daily to ensure they could tolerate the drug.
At the end of the 20-week induction phase, 228 patients with an HIV viral load below 50 copies/mL were randomized 2:2:1 to maintenance therapy with intramuscular injections of long-acting cabotegravir at 200 mg/mL and long-acting rilpivirine at 300 mg/mL in nanosuspension every 4 or 8 weeks or to the oral once-daily versions of the two drugs.
After 48 weeks of maintenance therapy, an HIV RNA load of less than 50 copies/mL was present in 92% of subjects on injections every 8 weeks, 91% with injections every 4 weeks, and 89% of those on oral daily therapy. Moreover, of the eight virologic nonresponders at 48 weeks in the 8-week-injection schedule, six remained in the study and five of them have consistently achieved a viral load below 50 copies/mL in subsequent visits through week 72.
Viral failure defined as an HIV RNA viral load greater than 200 copies/mL on two occasions occurred in 1% of patients on the 8-week interval schedule, 1% of those on oral therapy, and no one on IM injections every 4 weeks. One patient on every-8-week schedule developed treatment-emergent viral resistance.
More than 80% of patients in both IM injection study arms experienced painful injection site reactions after their first treatment session, 82% of which were mild and 17% moderate. Ninety percent of these reactions resolved within 7 days; median duration was 3 days. Two patients withdrew from the study as a result of these reactions. After the first round of injections, the incidence of injection site reactions fell to 25%-30%.
Other adverse events consisted of fever, fatigue, headache, rash, and flu-like symptoms, each with an incidence of 2%-4%.
Based upon the LATTE-2 outcomes, the once-per-month injection regimen has been selected for the forthcoming pivotal phase III randomized clinical trials of long-acting cabotegravir/rilpivirine for ART, Dr. Margolis said.
The pharmaceutical industry is pursuing long-acting injectable ART both for pre-exposure prophylaxis (PrEP) and for treatment of HIV-infected patients. Clinicians and patients alike are eager for this option, especially for PrEP, where adherence to daily oral therapy has been suboptimal in many studies.
But there is a fly in the ointment. One of the most talked about studies presented at AIDS 2016 was Dr. Ian McGowan’s report that low levels of rilpivirine were found in plasma and female genital tract fluids more than 18 months after a single IM 1,200-mg dose of rilpivirine in seven participants in a long-acting PrEP study.
“There is certainly the possibility that extended periods of drug availability at perhaps subtherapeutic concentrations might increase the risk of ART resistance in individuals who seroconvert after exposure to long-acting PrEP,” cautioned Dr. McGowan, professor of medicine at the University of Pittsburgh.
No similar studies have as yet been done with other candidate long-acting injectables, he said.
“It’s clear, I think, that as we move forward with this exciting field of long-acting injectables we really need to better characterize the terminal half-life of these products so that we can better inform management of the pharmacokinetic tail and hopefully avoid the potential for ART resistance,” Dr. McGowan added.
His study was funded by the Bill and Melinda Gates Foundation.
Dr. Margolis is an employee of Viiv Healthcare, the LATTE-2 study sponsor.
DURBAN, SOUTH AFRICA – Long-acting injectable antiretroviral therapy with cabotegravir and rilpivirine in nanosuspension successfully suppressed HIV-infected patients’ plasma viral load to fewer than 50 copies/mL for 48 weeks as maintenance therapy in the LATTE-2 trial, David A. Margolis, MD, reported at the 21st International AIDS Conference.
The intramuscular gluteal injections given every 4 or 8 weeks also proved safe and extremely well tolerated. At week 48, more than 97% of patients on injectable maintenance antiretroviral therapy (ART) reported a high degree of satisfaction with their treatment regimen on a structured questionnaire and expressed a willingness to continue on injectable therapy in the future, according to Dr. Margolis of Viiv Healthcare in Research Triangle Park, N.C.
Cabotegravir is an HIV integrase inhibitor, rilpivirine a nonnucleoside reverse transcriptase inhibitor. Rilpivirine is already approved as Edurant, a once-daily 25-mg tablet for oral therapy. Cabotegravir is under development as a 30-mg once-daily tablet. As oral agents, each has a half-life of roughly 40 hours. The two-drug oral combination showed a high degree of safety and efficacy through 96 weeks of follow-up in the LATTE-1 study, previously reported by Dr. Margolis (Lancet Infect Dis. 2015 Oct;15[10]):1145-55) .
In addition, the two antiretroviral agents are being developed as long-acting injectables with half-lives of 20-90 days. LATTE-2 was a phase IIb, open-label, multicenter study that began with 309 HIV-positive, ART-naive subjects who participated in a 20-week induction phase during which they received 30 mg/day of oral cabotegravir plus abacavir/lamivudine to induce HIV viral suppression. During the last 4 weeks of the induction phase, participants also received oral rilpivirine at 25 mg once daily to ensure they could tolerate the drug.
At the end of the 20-week induction phase, 228 patients with an HIV viral load below 50 copies/mL were randomized 2:2:1 to maintenance therapy with intramuscular injections of long-acting cabotegravir at 200 mg/mL and long-acting rilpivirine at 300 mg/mL in nanosuspension every 4 or 8 weeks or to the oral once-daily versions of the two drugs.
After 48 weeks of maintenance therapy, an HIV RNA load of less than 50 copies/mL was present in 92% of subjects on injections every 8 weeks, 91% with injections every 4 weeks, and 89% of those on oral daily therapy. Moreover, of the eight virologic nonresponders at 48 weeks in the 8-week-injection schedule, six remained in the study and five of them have consistently achieved a viral load below 50 copies/mL in subsequent visits through week 72.
Viral failure defined as an HIV RNA viral load greater than 200 copies/mL on two occasions occurred in 1% of patients on the 8-week interval schedule, 1% of those on oral therapy, and no one on IM injections every 4 weeks. One patient on every-8-week schedule developed treatment-emergent viral resistance.
More than 80% of patients in both IM injection study arms experienced painful injection site reactions after their first treatment session, 82% of which were mild and 17% moderate. Ninety percent of these reactions resolved within 7 days; median duration was 3 days. Two patients withdrew from the study as a result of these reactions. After the first round of injections, the incidence of injection site reactions fell to 25%-30%.
Other adverse events consisted of fever, fatigue, headache, rash, and flu-like symptoms, each with an incidence of 2%-4%.
Based upon the LATTE-2 outcomes, the once-per-month injection regimen has been selected for the forthcoming pivotal phase III randomized clinical trials of long-acting cabotegravir/rilpivirine for ART, Dr. Margolis said.
The pharmaceutical industry is pursuing long-acting injectable ART both for pre-exposure prophylaxis (PrEP) and for treatment of HIV-infected patients. Clinicians and patients alike are eager for this option, especially for PrEP, where adherence to daily oral therapy has been suboptimal in many studies.
But there is a fly in the ointment. One of the most talked about studies presented at AIDS 2016 was Dr. Ian McGowan’s report that low levels of rilpivirine were found in plasma and female genital tract fluids more than 18 months after a single IM 1,200-mg dose of rilpivirine in seven participants in a long-acting PrEP study.
“There is certainly the possibility that extended periods of drug availability at perhaps subtherapeutic concentrations might increase the risk of ART resistance in individuals who seroconvert after exposure to long-acting PrEP,” cautioned Dr. McGowan, professor of medicine at the University of Pittsburgh.
No similar studies have as yet been done with other candidate long-acting injectables, he said.
“It’s clear, I think, that as we move forward with this exciting field of long-acting injectables we really need to better characterize the terminal half-life of these products so that we can better inform management of the pharmacokinetic tail and hopefully avoid the potential for ART resistance,” Dr. McGowan added.
His study was funded by the Bill and Melinda Gates Foundation.
Dr. Margolis is an employee of Viiv Healthcare, the LATTE-2 study sponsor.
DURBAN, SOUTH AFRICA – Long-acting injectable antiretroviral therapy with cabotegravir and rilpivirine in nanosuspension successfully suppressed HIV-infected patients’ plasma viral load to fewer than 50 copies/mL for 48 weeks as maintenance therapy in the LATTE-2 trial, David A. Margolis, MD, reported at the 21st International AIDS Conference.
The intramuscular gluteal injections given every 4 or 8 weeks also proved safe and extremely well tolerated. At week 48, more than 97% of patients on injectable maintenance antiretroviral therapy (ART) reported a high degree of satisfaction with their treatment regimen on a structured questionnaire and expressed a willingness to continue on injectable therapy in the future, according to Dr. Margolis of Viiv Healthcare in Research Triangle Park, N.C.
Cabotegravir is an HIV integrase inhibitor, rilpivirine a nonnucleoside reverse transcriptase inhibitor. Rilpivirine is already approved as Edurant, a once-daily 25-mg tablet for oral therapy. Cabotegravir is under development as a 30-mg once-daily tablet. As oral agents, each has a half-life of roughly 40 hours. The two-drug oral combination showed a high degree of safety and efficacy through 96 weeks of follow-up in the LATTE-1 study, previously reported by Dr. Margolis (Lancet Infect Dis. 2015 Oct;15[10]):1145-55) .
In addition, the two antiretroviral agents are being developed as long-acting injectables with half-lives of 20-90 days. LATTE-2 was a phase IIb, open-label, multicenter study that began with 309 HIV-positive, ART-naive subjects who participated in a 20-week induction phase during which they received 30 mg/day of oral cabotegravir plus abacavir/lamivudine to induce HIV viral suppression. During the last 4 weeks of the induction phase, participants also received oral rilpivirine at 25 mg once daily to ensure they could tolerate the drug.
At the end of the 20-week induction phase, 228 patients with an HIV viral load below 50 copies/mL were randomized 2:2:1 to maintenance therapy with intramuscular injections of long-acting cabotegravir at 200 mg/mL and long-acting rilpivirine at 300 mg/mL in nanosuspension every 4 or 8 weeks or to the oral once-daily versions of the two drugs.
After 48 weeks of maintenance therapy, an HIV RNA load of less than 50 copies/mL was present in 92% of subjects on injections every 8 weeks, 91% with injections every 4 weeks, and 89% of those on oral daily therapy. Moreover, of the eight virologic nonresponders at 48 weeks in the 8-week-injection schedule, six remained in the study and five of them have consistently achieved a viral load below 50 copies/mL in subsequent visits through week 72.
Viral failure defined as an HIV RNA viral load greater than 200 copies/mL on two occasions occurred in 1% of patients on the 8-week interval schedule, 1% of those on oral therapy, and no one on IM injections every 4 weeks. One patient on every-8-week schedule developed treatment-emergent viral resistance.
More than 80% of patients in both IM injection study arms experienced painful injection site reactions after their first treatment session, 82% of which were mild and 17% moderate. Ninety percent of these reactions resolved within 7 days; median duration was 3 days. Two patients withdrew from the study as a result of these reactions. After the first round of injections, the incidence of injection site reactions fell to 25%-30%.
Other adverse events consisted of fever, fatigue, headache, rash, and flu-like symptoms, each with an incidence of 2%-4%.
Based upon the LATTE-2 outcomes, the once-per-month injection regimen has been selected for the forthcoming pivotal phase III randomized clinical trials of long-acting cabotegravir/rilpivirine for ART, Dr. Margolis said.
The pharmaceutical industry is pursuing long-acting injectable ART both for pre-exposure prophylaxis (PrEP) and for treatment of HIV-infected patients. Clinicians and patients alike are eager for this option, especially for PrEP, where adherence to daily oral therapy has been suboptimal in many studies.
But there is a fly in the ointment. One of the most talked about studies presented at AIDS 2016 was Dr. Ian McGowan’s report that low levels of rilpivirine were found in plasma and female genital tract fluids more than 18 months after a single IM 1,200-mg dose of rilpivirine in seven participants in a long-acting PrEP study.
“There is certainly the possibility that extended periods of drug availability at perhaps subtherapeutic concentrations might increase the risk of ART resistance in individuals who seroconvert after exposure to long-acting PrEP,” cautioned Dr. McGowan, professor of medicine at the University of Pittsburgh.
No similar studies have as yet been done with other candidate long-acting injectables, he said.
“It’s clear, I think, that as we move forward with this exciting field of long-acting injectables we really need to better characterize the terminal half-life of these products so that we can better inform management of the pharmacokinetic tail and hopefully avoid the potential for ART resistance,” Dr. McGowan added.
His study was funded by the Bill and Melinda Gates Foundation.
Dr. Margolis is an employee of Viiv Healthcare, the LATTE-2 study sponsor.
AT AIDS 2016
Key clinical point: The era of all-injectable, long-acting antiretroviral therapy for HIV may be drawing closer.
Major finding: More than 90% of HIV-infected patients maintained a viral load below 50 copies/mL throughout 48 weeks of maintenance therapy with intramuscular injections of cabotegravir and rilpivirine regardless of whether given every 4 or 8 weeks.
Data source: LATTE-2 was a phase IIb, open-label, multicenter study in which 228 HIV-infected patients with an HIV RNA viral load below 50 copies/mL on oral therapy at baseline were randomized to long-acting injectable antiretroviral therapy either every 4 or 8 weeks or to daily oral therapy.
Disclosures: The presenter is an employee of ViiV Healthcare, the study sponsor.
Extended-release granisetron superior antiemetic for cisplatin chemotherapy
SAN DIEGO – An investigational extended-release formulation of granisetron outperformed ondansetron as a component of triple-drug antiemetic therapy for patients on cisplatin-based highly emetogenic chemotherapy, Dr. Lee Schwartzberg reported at the annual meeting of the Society of Gynecologic Oncology.
He presented a post hoc subgroup analysis drawn from the previously reported pivotal phase III 942-patient MAGIC trial of extended-release granisetron (Sustol), formerly known as APF530, versus ondansetron (Zofran) as part of the triple-drug antiemetic regimen recommended in National Community Cancer Network guidelines for the prevention of chemotherapy-induced nausea and vomiting (CINV) in patients on highly emetogenic chemotherapy .
Dr. Schwartzberg and coinvestigators focused on the 251 MAGIC participants on high-dose cisplatin-based chemotherapy. The reason? Few studies have examined the management of CINV in patients on platinum-based regimens, commonly used in treating gynecologic cancers, explained Dr. Schwartzberg, medical director of the West Clinic in Memphis.
Sustol consists of 2% granisetron in a proprietary slow-release vehicle. A single 500-mg subcutaneous dose containing 10 mg of granisetron maintains therapeutic blood levels of granisetron for 5 days or longer. These pharmacokinetics make APF530 an attractive strategy for preventing both acute and delayed chemotherapy-induced nausea and vomiting. The delayed phase, which affects many cancer patients 2-5 days following administration of chemotherapy, has traditionally been a particularly difficult management challenge.
National Community Cancer Network guidelines recommend that patients on highly emetogenic chemotherapy receive a potent three-drug antiemetic regimen consisting of a 5-hydroxytryptoamine-3 receptor antagonist – ondansetron and granisetron fall within this class – along with a neurokinin-1 receptor antagonist and dexamethasone.
MAGIC was a double-blind, prospective, multicenter trial in which 942 patients on highly emetogenic chemotherapy received either 500 mg of subcutaneous extended-release granisetron or intravenous ondansetron at 0.15 mg/kg. In addition, all participants concomitantly got 150 mg of the neurokinin-1 receptor antagonist fosaprepitant (Emend) plus 12 mg of IV dexamethasone on day 1, followed by 8 mg of oral dexamethasone once on day 2 and 8 mg twice daily on days 3 and 4.
In the overall trial, extended-release granisetron significantly outperformed ondansetron. The primary endpoint – complete response defined as no emesis and no use of rescue medication for CINV during the delayed phase – was achieved in 64.7% of the Sustol group, compared with 56.6% on ondansetron.
Results in Dr. Schwartzberg’s post hoc analysis confined to the 251 randomized patients on high-dose cisplatin-based chemotherapy paralleled those in the overall trial: a 64.8% complete response rate in the delayed phase in the granisetron group versus 56.3% with ondansetron. That absolute 8.5% difference favoring the investigational agent translates to a number needed to treat (NNT) of 12.
In the 112 women on highly emetogenic cisplatin-based chemotherapy, the complete response rates in the delayed phase were 61.6% with extended-release granisetron and 53.2% with ondansetron, for an absolute 10.3% difference and an NNT of 10.
Heron Therapeutics, which is developing extended-release granisetron, is awaiting word from the Food and Drug Administration regarding the company’s application for marketing approval.
The MAGIC trial was sponsored by Heron Therapeutics. Dr. Schwartzberg is a consultant to the company.
SAN DIEGO – An investigational extended-release formulation of granisetron outperformed ondansetron as a component of triple-drug antiemetic therapy for patients on cisplatin-based highly emetogenic chemotherapy, Dr. Lee Schwartzberg reported at the annual meeting of the Society of Gynecologic Oncology.
He presented a post hoc subgroup analysis drawn from the previously reported pivotal phase III 942-patient MAGIC trial of extended-release granisetron (Sustol), formerly known as APF530, versus ondansetron (Zofran) as part of the triple-drug antiemetic regimen recommended in National Community Cancer Network guidelines for the prevention of chemotherapy-induced nausea and vomiting (CINV) in patients on highly emetogenic chemotherapy .
Dr. Schwartzberg and coinvestigators focused on the 251 MAGIC participants on high-dose cisplatin-based chemotherapy. The reason? Few studies have examined the management of CINV in patients on platinum-based regimens, commonly used in treating gynecologic cancers, explained Dr. Schwartzberg, medical director of the West Clinic in Memphis.
Sustol consists of 2% granisetron in a proprietary slow-release vehicle. A single 500-mg subcutaneous dose containing 10 mg of granisetron maintains therapeutic blood levels of granisetron for 5 days or longer. These pharmacokinetics make APF530 an attractive strategy for preventing both acute and delayed chemotherapy-induced nausea and vomiting. The delayed phase, which affects many cancer patients 2-5 days following administration of chemotherapy, has traditionally been a particularly difficult management challenge.
National Community Cancer Network guidelines recommend that patients on highly emetogenic chemotherapy receive a potent three-drug antiemetic regimen consisting of a 5-hydroxytryptoamine-3 receptor antagonist – ondansetron and granisetron fall within this class – along with a neurokinin-1 receptor antagonist and dexamethasone.
MAGIC was a double-blind, prospective, multicenter trial in which 942 patients on highly emetogenic chemotherapy received either 500 mg of subcutaneous extended-release granisetron or intravenous ondansetron at 0.15 mg/kg. In addition, all participants concomitantly got 150 mg of the neurokinin-1 receptor antagonist fosaprepitant (Emend) plus 12 mg of IV dexamethasone on day 1, followed by 8 mg of oral dexamethasone once on day 2 and 8 mg twice daily on days 3 and 4.
In the overall trial, extended-release granisetron significantly outperformed ondansetron. The primary endpoint – complete response defined as no emesis and no use of rescue medication for CINV during the delayed phase – was achieved in 64.7% of the Sustol group, compared with 56.6% on ondansetron.
Results in Dr. Schwartzberg’s post hoc analysis confined to the 251 randomized patients on high-dose cisplatin-based chemotherapy paralleled those in the overall trial: a 64.8% complete response rate in the delayed phase in the granisetron group versus 56.3% with ondansetron. That absolute 8.5% difference favoring the investigational agent translates to a number needed to treat (NNT) of 12.
In the 112 women on highly emetogenic cisplatin-based chemotherapy, the complete response rates in the delayed phase were 61.6% with extended-release granisetron and 53.2% with ondansetron, for an absolute 10.3% difference and an NNT of 10.
Heron Therapeutics, which is developing extended-release granisetron, is awaiting word from the Food and Drug Administration regarding the company’s application for marketing approval.
The MAGIC trial was sponsored by Heron Therapeutics. Dr. Schwartzberg is a consultant to the company.
SAN DIEGO – An investigational extended-release formulation of granisetron outperformed ondansetron as a component of triple-drug antiemetic therapy for patients on cisplatin-based highly emetogenic chemotherapy, Dr. Lee Schwartzberg reported at the annual meeting of the Society of Gynecologic Oncology.
He presented a post hoc subgroup analysis drawn from the previously reported pivotal phase III 942-patient MAGIC trial of extended-release granisetron (Sustol), formerly known as APF530, versus ondansetron (Zofran) as part of the triple-drug antiemetic regimen recommended in National Community Cancer Network guidelines for the prevention of chemotherapy-induced nausea and vomiting (CINV) in patients on highly emetogenic chemotherapy .
Dr. Schwartzberg and coinvestigators focused on the 251 MAGIC participants on high-dose cisplatin-based chemotherapy. The reason? Few studies have examined the management of CINV in patients on platinum-based regimens, commonly used in treating gynecologic cancers, explained Dr. Schwartzberg, medical director of the West Clinic in Memphis.
Sustol consists of 2% granisetron in a proprietary slow-release vehicle. A single 500-mg subcutaneous dose containing 10 mg of granisetron maintains therapeutic blood levels of granisetron for 5 days or longer. These pharmacokinetics make APF530 an attractive strategy for preventing both acute and delayed chemotherapy-induced nausea and vomiting. The delayed phase, which affects many cancer patients 2-5 days following administration of chemotherapy, has traditionally been a particularly difficult management challenge.
National Community Cancer Network guidelines recommend that patients on highly emetogenic chemotherapy receive a potent three-drug antiemetic regimen consisting of a 5-hydroxytryptoamine-3 receptor antagonist – ondansetron and granisetron fall within this class – along with a neurokinin-1 receptor antagonist and dexamethasone.
MAGIC was a double-blind, prospective, multicenter trial in which 942 patients on highly emetogenic chemotherapy received either 500 mg of subcutaneous extended-release granisetron or intravenous ondansetron at 0.15 mg/kg. In addition, all participants concomitantly got 150 mg of the neurokinin-1 receptor antagonist fosaprepitant (Emend) plus 12 mg of IV dexamethasone on day 1, followed by 8 mg of oral dexamethasone once on day 2 and 8 mg twice daily on days 3 and 4.
In the overall trial, extended-release granisetron significantly outperformed ondansetron. The primary endpoint – complete response defined as no emesis and no use of rescue medication for CINV during the delayed phase – was achieved in 64.7% of the Sustol group, compared with 56.6% on ondansetron.
Results in Dr. Schwartzberg’s post hoc analysis confined to the 251 randomized patients on high-dose cisplatin-based chemotherapy paralleled those in the overall trial: a 64.8% complete response rate in the delayed phase in the granisetron group versus 56.3% with ondansetron. That absolute 8.5% difference favoring the investigational agent translates to a number needed to treat (NNT) of 12.
In the 112 women on highly emetogenic cisplatin-based chemotherapy, the complete response rates in the delayed phase were 61.6% with extended-release granisetron and 53.2% with ondansetron, for an absolute 10.3% difference and an NNT of 10.
Heron Therapeutics, which is developing extended-release granisetron, is awaiting word from the Food and Drug Administration regarding the company’s application for marketing approval.
The MAGIC trial was sponsored by Heron Therapeutics. Dr. Schwartzberg is a consultant to the company.
AT THE ANNUAL MEETING ON WOMEN’S CANCER
Key clinical point: Investigational agent outperforms ondansetron during delayed phase of highly emetogenic cisplatin-based chemotherapy-induced nausea and vomiting.
Major finding: Complete response rate during delayed phase was 64.8% when triple antiemetic therapy included extended-release granisetron and 56.3% when the regimen included ondansetron.
Data source: This post hoc subgroup analysis of the 942-patient pivotal phase III MAGIC trial focused specifically on the 251 participants on highly emetogenic cisplatin-based chemotherapy.
Disclosures: The study presenter reported serving as a consultant to Heron Therapeutics, which sponsored the MAGIC trial.
With HIV PrEP, benefits outweigh resistance risk
DURBAN, SOUTH AFRICA – For physicians who are leery of prescribing oral daily pre-exposure prophylaxis (PrEP) against HIV infection because of concern that it will promote drug-resistant viral strains, Robert Grant, MD, has a reassuring message.
It’s a message based upon his systematic review of published resistance testing results in all the randomized, placebo-controlled PrEP trials.
“For clinicians who are anxious, I can say based on this excellent work that if your primary concern is drug resistance – and I’m not suggesting that it should be – but if you are very concerned about drug resistance, then you should be a PrEP advocate, because in the end, your population will have less drug resistance,” Dr. Grant said at the 21st International AIDS Conference.
The overall rate of resistance to emtricitabine or tenofovir – the antiretrovirals contained in fixed-dose combination in Truvada, the only FDA-approved agent for HIV PrEP – was five cases in 9,222 trial participants, for a risk of 0.05%. The number needed to treat in order to prevent one HIV infection was 13-60, depending upon the adherence rate in a given trial.
In contrast, the number needed to harm by causing emergence of a drug-resistant strain of HIV was 1,844, reported Dr. Grant, professor of medicine at the University of California, San Francisco.
Drug resistance during PrEP is rare. It occurs mainly when PrEP is started or restarted during an acute HIV infection. Almost all of the resistance is to emtricitabine. Those emtricitabine-resistant HIV infections are treatable.
The randomized trials of PrEP used several methods of screening for acute HIV infection: rapid second- and third-generation antibody assays, highly sensitive HIV nucleic acid genotypic assays capable of detecting resistant viral variants present in very low abundance, and a clinical screen.
The use of one or the other of the laboratory approaches is common practice in the United States and Europe. But these tools are much less frequently available in sub-Saharan Africa and elsewhere the HIV epidemic is hitting hardest. That is a setting where a clinical screening program could be of great value.
“The majority of people with acute HIV infection will have some sort of symptoms of an acute viral syndrome. They’re nonspecific symptoms: a flu-like illness, fever, sore throat, headache, a rash,” he observed.
Dr. Grant was first author of the iPrEx OLE study, the only published trial to examine a strategy of clinical screening for acute viral syndromes and acute HIV infection in PrEP candidates (Lancet Infect Dis. 2014 Sep;14[9]:820-9).
Thirty of 1,603 PrEP candidates (1.9%) who underwent clinical screening had PrEP deferred because of an acute viral syndrome. Two of those 30 patients subsequently proved to have acute HIV infection on laboratory testing, 25 of the HIV-negative patients had a delayed start of PrEP, and 3 of the 30 never started PrEP. Thus, clinical screening had 100% sensitivity, 98% specificity, 100% negative predictive value, and a 6.7% positive predictive value, according to Dr. Grant.
The World Health Organization sponsored his systematic review. Dr. Grant reported having no financial conflicts of interest.
DURBAN, SOUTH AFRICA – For physicians who are leery of prescribing oral daily pre-exposure prophylaxis (PrEP) against HIV infection because of concern that it will promote drug-resistant viral strains, Robert Grant, MD, has a reassuring message.
It’s a message based upon his systematic review of published resistance testing results in all the randomized, placebo-controlled PrEP trials.
“For clinicians who are anxious, I can say based on this excellent work that if your primary concern is drug resistance – and I’m not suggesting that it should be – but if you are very concerned about drug resistance, then you should be a PrEP advocate, because in the end, your population will have less drug resistance,” Dr. Grant said at the 21st International AIDS Conference.
The overall rate of resistance to emtricitabine or tenofovir – the antiretrovirals contained in fixed-dose combination in Truvada, the only FDA-approved agent for HIV PrEP – was five cases in 9,222 trial participants, for a risk of 0.05%. The number needed to treat in order to prevent one HIV infection was 13-60, depending upon the adherence rate in a given trial.
In contrast, the number needed to harm by causing emergence of a drug-resistant strain of HIV was 1,844, reported Dr. Grant, professor of medicine at the University of California, San Francisco.
Drug resistance during PrEP is rare. It occurs mainly when PrEP is started or restarted during an acute HIV infection. Almost all of the resistance is to emtricitabine. Those emtricitabine-resistant HIV infections are treatable.
The randomized trials of PrEP used several methods of screening for acute HIV infection: rapid second- and third-generation antibody assays, highly sensitive HIV nucleic acid genotypic assays capable of detecting resistant viral variants present in very low abundance, and a clinical screen.
The use of one or the other of the laboratory approaches is common practice in the United States and Europe. But these tools are much less frequently available in sub-Saharan Africa and elsewhere the HIV epidemic is hitting hardest. That is a setting where a clinical screening program could be of great value.
“The majority of people with acute HIV infection will have some sort of symptoms of an acute viral syndrome. They’re nonspecific symptoms: a flu-like illness, fever, sore throat, headache, a rash,” he observed.
Dr. Grant was first author of the iPrEx OLE study, the only published trial to examine a strategy of clinical screening for acute viral syndromes and acute HIV infection in PrEP candidates (Lancet Infect Dis. 2014 Sep;14[9]:820-9).
Thirty of 1,603 PrEP candidates (1.9%) who underwent clinical screening had PrEP deferred because of an acute viral syndrome. Two of those 30 patients subsequently proved to have acute HIV infection on laboratory testing, 25 of the HIV-negative patients had a delayed start of PrEP, and 3 of the 30 never started PrEP. Thus, clinical screening had 100% sensitivity, 98% specificity, 100% negative predictive value, and a 6.7% positive predictive value, according to Dr. Grant.
The World Health Organization sponsored his systematic review. Dr. Grant reported having no financial conflicts of interest.
DURBAN, SOUTH AFRICA – For physicians who are leery of prescribing oral daily pre-exposure prophylaxis (PrEP) against HIV infection because of concern that it will promote drug-resistant viral strains, Robert Grant, MD, has a reassuring message.
It’s a message based upon his systematic review of published resistance testing results in all the randomized, placebo-controlled PrEP trials.
“For clinicians who are anxious, I can say based on this excellent work that if your primary concern is drug resistance – and I’m not suggesting that it should be – but if you are very concerned about drug resistance, then you should be a PrEP advocate, because in the end, your population will have less drug resistance,” Dr. Grant said at the 21st International AIDS Conference.
The overall rate of resistance to emtricitabine or tenofovir – the antiretrovirals contained in fixed-dose combination in Truvada, the only FDA-approved agent for HIV PrEP – was five cases in 9,222 trial participants, for a risk of 0.05%. The number needed to treat in order to prevent one HIV infection was 13-60, depending upon the adherence rate in a given trial.
In contrast, the number needed to harm by causing emergence of a drug-resistant strain of HIV was 1,844, reported Dr. Grant, professor of medicine at the University of California, San Francisco.
Drug resistance during PrEP is rare. It occurs mainly when PrEP is started or restarted during an acute HIV infection. Almost all of the resistance is to emtricitabine. Those emtricitabine-resistant HIV infections are treatable.
The randomized trials of PrEP used several methods of screening for acute HIV infection: rapid second- and third-generation antibody assays, highly sensitive HIV nucleic acid genotypic assays capable of detecting resistant viral variants present in very low abundance, and a clinical screen.
The use of one or the other of the laboratory approaches is common practice in the United States and Europe. But these tools are much less frequently available in sub-Saharan Africa and elsewhere the HIV epidemic is hitting hardest. That is a setting where a clinical screening program could be of great value.
“The majority of people with acute HIV infection will have some sort of symptoms of an acute viral syndrome. They’re nonspecific symptoms: a flu-like illness, fever, sore throat, headache, a rash,” he observed.
Dr. Grant was first author of the iPrEx OLE study, the only published trial to examine a strategy of clinical screening for acute viral syndromes and acute HIV infection in PrEP candidates (Lancet Infect Dis. 2014 Sep;14[9]:820-9).
Thirty of 1,603 PrEP candidates (1.9%) who underwent clinical screening had PrEP deferred because of an acute viral syndrome. Two of those 30 patients subsequently proved to have acute HIV infection on laboratory testing, 25 of the HIV-negative patients had a delayed start of PrEP, and 3 of the 30 never started PrEP. Thus, clinical screening had 100% sensitivity, 98% specificity, 100% negative predictive value, and a 6.7% positive predictive value, according to Dr. Grant.
The World Health Organization sponsored his systematic review. Dr. Grant reported having no financial conflicts of interest.
AT AIDS 2016
Key clinical point: The benefit of oral daily chemoprophylaxis against HIV infection in at-risk individuals far outweighs the risk.
Major finding: The number needed to treat with oral daily HIV pre-exposure prophylaxis in order to prevent one HIV infection is 13-60; the number needed to harm by causing a case of drug-resistant HIV is 1,844.
Data source: This was a systematic review of six published randomized, placebo-controlled trials of HIV pre-exposure prophylaxis totaling 15,894 participants.
Disclosures: The World Health Organization sponsored the review. Dr. Grant reported having no financial conflicts of interest.
Ombitasvir/paritaprevir-based HCV regimens perform well in HIV coinfection
DURBAN, SOUTH AFRICA – The all-oral, interferon-free, triple- or double-direct-acting antiviral regimens informally known as 3D and 2D proved effective and safe for treatment of chronic hepatitis C due to HCV genotypes 1 and 4 in HIV-coinfected patients taking various antiretroviral combinations in the phase III, multicenter TURQUOISE-1, Part 2 trial, Jürgen K. Rockstroh, MD, reported at the 21st International AIDS Conference.
TURQUOISE-1, Part 2 included 228 patients coinfected with HCV/HIV. Of the 154 with HCV genotype 1a and 44 with genotype 1b, 21 of whom had compensated cirrhosis, 98% of subjects achieved a sustained virologic response (SVR) 12 weeks after conclusion of the 3D regimen, or SVR12, in a modified intent-to-treat analysis. A handful of patients with missing data, or those who stopped treatment for reasons other than virologic failure, were excluded from the analysis.
In the 28 patients coinfected with HCV genotype 4, none of whom had cirrhosis, the modified SVR12 rate was 100%.
The so-called 3D regimen consists of the NS5A inhibitor ombitasvir coformulated in once-daily, fixed-dose fashion with the NS3/4A protease inhibitor paritaprevir boosted with ritonavir, along with twice-daily dasabuvir, a nonnucleoside NS5B RNA polymerase inhibitor. This combination has received Food and Drug Administration approval and is marketed as Viekira Pak for treatment of HCV genotype 1 in HIV-coinfected patients on the basis of the TURQUOISE-1, Part 1a trial (JAMA 2015;313[12]:1223-32). But this study was confined to 63 U.S. patients, and European investigators wanted to see how 3D worked in a larger, more diverse population, explained Dr. Rockstroh, professor of medicine and head of the outpatient HIV clinic at the University of Bonn (Germany).
In TURQUOISE-1, Part 2, patients with HCV genotype 1a without cirrhosis received 12 weeks of the 3D regimen plus ribavirin, while those with compensated cirrhosis got 24 weeks. Patients with genotype 1b received 12 weeks of 3D without ribavirin, regardless of whether or not they had compensated cirrhosis.
Because dasabuvir is ineffective against HCV genotype 4, those patients got 12 weeks of the 2D regimen – the coformulated ombitasvir/paritaprevir/ritonavir – along with ribavirin.
All subjects were on antiretroviral regimens based upon either darunavir (Prezista), atazanavir (Reyataz), raltegravir (Isentress), or dolutegravir (Tivicay). Nearly all (97%) of the patients had an HIV RNA viral load of less than 40 copies/mL at baseline. Those on ritonavir-boosted antiretroviral therapy discontinued the ritonavir and received it instead via their 3D or 2D hepatitis C therapy.
HIV suppression was well maintained throughout the study. Intermittent episodes of HIV viremia affected 4% of participants, but in all cases the peak was less than 200 copies/mL. None of the episodes met standard criteria for HIV genotypic resistance testing, according to Dr. Rockstroh.
In contrast to the former standard therapy consisting of pegylated interferon and ribavirin, which had an SVR of about 30% in HCV/HIV coinfected patients and was fraught with serious side effects, no one discontinued treatment due to adverse events in TURQUOISE-1, Part 2. No cases of hepatic decompensation occurred. The only serious adverse event deemed possibly treatment related was a single case of depression. Mild fatigue, nausea, diarrhea, insomnia, headache, or itching each occurred in 10%-20% of subjects.
As effective antiretroviral therapy enables HIV-infected patients to have a lifespan approaching normal, HCV coinfection has emerged as a major issue. Roughly one-third of HIV-positive individuals are coinfected with HCV, and the rate of death due to HCV-related liver disease in coinfected patients exceeds the rate of death due to HIV. HCV genotype 1 accounts for three-quarters of chronic hepatitis C in the United States, while genotype 4 predominates in Africa, where the bulk of HIV infections occur.
During the AIDS 2016 conference, the Food and Drug Administration approved Viekira XR, an extended-release coformulation of the 3D regimen. Each tablet contains 200 mg of dasabuvir, 8.33 mg of ombitasvir, 50 mg of paritaprevir, and 33.33 mg of ritonavir. The standard dosing is three oral tablets taken once daily.
The TURQUOISE-1, Part 2 trial was sponsored by AbbVie. Dr. Rockstroh is a consultant to AbbVie and seven other pharmaceutical companies.
DURBAN, SOUTH AFRICA – The all-oral, interferon-free, triple- or double-direct-acting antiviral regimens informally known as 3D and 2D proved effective and safe for treatment of chronic hepatitis C due to HCV genotypes 1 and 4 in HIV-coinfected patients taking various antiretroviral combinations in the phase III, multicenter TURQUOISE-1, Part 2 trial, Jürgen K. Rockstroh, MD, reported at the 21st International AIDS Conference.
TURQUOISE-1, Part 2 included 228 patients coinfected with HCV/HIV. Of the 154 with HCV genotype 1a and 44 with genotype 1b, 21 of whom had compensated cirrhosis, 98% of subjects achieved a sustained virologic response (SVR) 12 weeks after conclusion of the 3D regimen, or SVR12, in a modified intent-to-treat analysis. A handful of patients with missing data, or those who stopped treatment for reasons other than virologic failure, were excluded from the analysis.
In the 28 patients coinfected with HCV genotype 4, none of whom had cirrhosis, the modified SVR12 rate was 100%.
The so-called 3D regimen consists of the NS5A inhibitor ombitasvir coformulated in once-daily, fixed-dose fashion with the NS3/4A protease inhibitor paritaprevir boosted with ritonavir, along with twice-daily dasabuvir, a nonnucleoside NS5B RNA polymerase inhibitor. This combination has received Food and Drug Administration approval and is marketed as Viekira Pak for treatment of HCV genotype 1 in HIV-coinfected patients on the basis of the TURQUOISE-1, Part 1a trial (JAMA 2015;313[12]:1223-32). But this study was confined to 63 U.S. patients, and European investigators wanted to see how 3D worked in a larger, more diverse population, explained Dr. Rockstroh, professor of medicine and head of the outpatient HIV clinic at the University of Bonn (Germany).
In TURQUOISE-1, Part 2, patients with HCV genotype 1a without cirrhosis received 12 weeks of the 3D regimen plus ribavirin, while those with compensated cirrhosis got 24 weeks. Patients with genotype 1b received 12 weeks of 3D without ribavirin, regardless of whether or not they had compensated cirrhosis.
Because dasabuvir is ineffective against HCV genotype 4, those patients got 12 weeks of the 2D regimen – the coformulated ombitasvir/paritaprevir/ritonavir – along with ribavirin.
All subjects were on antiretroviral regimens based upon either darunavir (Prezista), atazanavir (Reyataz), raltegravir (Isentress), or dolutegravir (Tivicay). Nearly all (97%) of the patients had an HIV RNA viral load of less than 40 copies/mL at baseline. Those on ritonavir-boosted antiretroviral therapy discontinued the ritonavir and received it instead via their 3D or 2D hepatitis C therapy.
HIV suppression was well maintained throughout the study. Intermittent episodes of HIV viremia affected 4% of participants, but in all cases the peak was less than 200 copies/mL. None of the episodes met standard criteria for HIV genotypic resistance testing, according to Dr. Rockstroh.
In contrast to the former standard therapy consisting of pegylated interferon and ribavirin, which had an SVR of about 30% in HCV/HIV coinfected patients and was fraught with serious side effects, no one discontinued treatment due to adverse events in TURQUOISE-1, Part 2. No cases of hepatic decompensation occurred. The only serious adverse event deemed possibly treatment related was a single case of depression. Mild fatigue, nausea, diarrhea, insomnia, headache, or itching each occurred in 10%-20% of subjects.
As effective antiretroviral therapy enables HIV-infected patients to have a lifespan approaching normal, HCV coinfection has emerged as a major issue. Roughly one-third of HIV-positive individuals are coinfected with HCV, and the rate of death due to HCV-related liver disease in coinfected patients exceeds the rate of death due to HIV. HCV genotype 1 accounts for three-quarters of chronic hepatitis C in the United States, while genotype 4 predominates in Africa, where the bulk of HIV infections occur.
During the AIDS 2016 conference, the Food and Drug Administration approved Viekira XR, an extended-release coformulation of the 3D regimen. Each tablet contains 200 mg of dasabuvir, 8.33 mg of ombitasvir, 50 mg of paritaprevir, and 33.33 mg of ritonavir. The standard dosing is three oral tablets taken once daily.
The TURQUOISE-1, Part 2 trial was sponsored by AbbVie. Dr. Rockstroh is a consultant to AbbVie and seven other pharmaceutical companies.
DURBAN, SOUTH AFRICA – The all-oral, interferon-free, triple- or double-direct-acting antiviral regimens informally known as 3D and 2D proved effective and safe for treatment of chronic hepatitis C due to HCV genotypes 1 and 4 in HIV-coinfected patients taking various antiretroviral combinations in the phase III, multicenter TURQUOISE-1, Part 2 trial, Jürgen K. Rockstroh, MD, reported at the 21st International AIDS Conference.
TURQUOISE-1, Part 2 included 228 patients coinfected with HCV/HIV. Of the 154 with HCV genotype 1a and 44 with genotype 1b, 21 of whom had compensated cirrhosis, 98% of subjects achieved a sustained virologic response (SVR) 12 weeks after conclusion of the 3D regimen, or SVR12, in a modified intent-to-treat analysis. A handful of patients with missing data, or those who stopped treatment for reasons other than virologic failure, were excluded from the analysis.
In the 28 patients coinfected with HCV genotype 4, none of whom had cirrhosis, the modified SVR12 rate was 100%.
The so-called 3D regimen consists of the NS5A inhibitor ombitasvir coformulated in once-daily, fixed-dose fashion with the NS3/4A protease inhibitor paritaprevir boosted with ritonavir, along with twice-daily dasabuvir, a nonnucleoside NS5B RNA polymerase inhibitor. This combination has received Food and Drug Administration approval and is marketed as Viekira Pak for treatment of HCV genotype 1 in HIV-coinfected patients on the basis of the TURQUOISE-1, Part 1a trial (JAMA 2015;313[12]:1223-32). But this study was confined to 63 U.S. patients, and European investigators wanted to see how 3D worked in a larger, more diverse population, explained Dr. Rockstroh, professor of medicine and head of the outpatient HIV clinic at the University of Bonn (Germany).
In TURQUOISE-1, Part 2, patients with HCV genotype 1a without cirrhosis received 12 weeks of the 3D regimen plus ribavirin, while those with compensated cirrhosis got 24 weeks. Patients with genotype 1b received 12 weeks of 3D without ribavirin, regardless of whether or not they had compensated cirrhosis.
Because dasabuvir is ineffective against HCV genotype 4, those patients got 12 weeks of the 2D regimen – the coformulated ombitasvir/paritaprevir/ritonavir – along with ribavirin.
All subjects were on antiretroviral regimens based upon either darunavir (Prezista), atazanavir (Reyataz), raltegravir (Isentress), or dolutegravir (Tivicay). Nearly all (97%) of the patients had an HIV RNA viral load of less than 40 copies/mL at baseline. Those on ritonavir-boosted antiretroviral therapy discontinued the ritonavir and received it instead via their 3D or 2D hepatitis C therapy.
HIV suppression was well maintained throughout the study. Intermittent episodes of HIV viremia affected 4% of participants, but in all cases the peak was less than 200 copies/mL. None of the episodes met standard criteria for HIV genotypic resistance testing, according to Dr. Rockstroh.
In contrast to the former standard therapy consisting of pegylated interferon and ribavirin, which had an SVR of about 30% in HCV/HIV coinfected patients and was fraught with serious side effects, no one discontinued treatment due to adverse events in TURQUOISE-1, Part 2. No cases of hepatic decompensation occurred. The only serious adverse event deemed possibly treatment related was a single case of depression. Mild fatigue, nausea, diarrhea, insomnia, headache, or itching each occurred in 10%-20% of subjects.
As effective antiretroviral therapy enables HIV-infected patients to have a lifespan approaching normal, HCV coinfection has emerged as a major issue. Roughly one-third of HIV-positive individuals are coinfected with HCV, and the rate of death due to HCV-related liver disease in coinfected patients exceeds the rate of death due to HIV. HCV genotype 1 accounts for three-quarters of chronic hepatitis C in the United States, while genotype 4 predominates in Africa, where the bulk of HIV infections occur.
During the AIDS 2016 conference, the Food and Drug Administration approved Viekira XR, an extended-release coformulation of the 3D regimen. Each tablet contains 200 mg of dasabuvir, 8.33 mg of ombitasvir, 50 mg of paritaprevir, and 33.33 mg of ritonavir. The standard dosing is three oral tablets taken once daily.
The TURQUOISE-1, Part 2 trial was sponsored by AbbVie. Dr. Rockstroh is a consultant to AbbVie and seven other pharmaceutical companies.
AT AIDS 2016
Key clinical point: All-oral regimens featuring two or three direct-acting-antiviral agents proved safe, effective, and well tolerated for treatment of chronic hepatitis C due to genotypes 1 or 4 in patients coinfected with HIV.
Major finding: The sustained virologic response rate 12 weeks after completion of an all-oral, triple-direct-acting antiviral regimen for chronic hepatitis C due to genotype 1 in HIV-coinfected patients was 98%, while the corresponding rate was 100% in coinfected patients with genotype 4 disease.
Data source: The TURQUOISE-1, Part 2 trial, a phase III multicenter study of all-oral, triple- or double-direct-acting antiviral regimens in 228 patients coinfected with HIV and hepatitis C genotypes 1 or 4.
Disclosures: The TURQUOISE-1, Part 2 trial was sponsored by AbbVie. The presenter is a consultant to the company.
Anal cancer in HIV-infected patients: to screen or not?
DURBAN, SOUTH AFRICA – Screening for anal cancer in HIV-infected men or women should not be part of routine clinical practice at this time, Andrew Grulich, MBBS, PhD, declared at the 21st International AIDS Conference.
Some experts recommend anal cytologic screening or high-resolution anoscopy for HIV-positive men and women, but it’s worth noting that strategy hasn’t been incorporated into any national practice guidelines.
“And for very good reason: When we have a condition with a prevalence that’s so high and a treatment with recurrence rates that are so high, I think we need to question our approach,” said Dr. Grulich, professor of medicine and head of the HIV epidemiology and prevention program at the University of New South Wales in Sydney, Australia.
Screening proponents point to the high incidence of anal cancer in persons with HIV infection. It’s the fourth most common cancer in HIV patients in the United States, behind the AIDS-defining cancers and lung cancer. Indeed, the anal cancer rate is 10-fold greater in HIV-positive women, heterosexual men, and injection drug users than in the HIV-negative general population, and 50-fold higher in HIV-positive gay and bisexual men. Screening proponents also draw an analogy between anal cancer screening and the screening and treatment of cervical intraepithelial neoplasia (CIN), which has been enormously successful in preventing cervical cancer. But Dr. Grulich said he believes the cervical cancer screening analogy is faulty.
Colposcopy has a mean 90% specificity for diagnosis of HPV-related high-grade squamous intraepithelial lesions (HSIL) or cervical cancer, while high-resolution anoscopy as a diagnostic test has a specificity as low as 37% in HIV-positive persons. The prevalence of HSIL is 30%-40% in anal samples from HIV-infected homosexual men, compared with 1%-2% in cervical samples from HIV-negative women.
The rate of progression from CIN-3 to cervical cancer in women in the general population is about 1 in 80 per year. In contrast, the rate of progression from anal intraepithelial neoplasia (AIN)-2 or AIN-3 to anal cancer in HIV-infected homosexual men is estimated at only 1 in 400-600 per year, probably because regression of anal lesions is quite common.
Moreover, while a single treatment of high-grade CIN is typically curative and entails little morbidity, destruction of AIN by means of heat, cold, or electricity has a 70% failure rate, carries substantial morbidity, and is not supported by any evidence that it actually reduces the incidence of anal cancer, he continued.
“We’re in a bit of a quandary regarding what to do about anal cancer prevention. We really need research in order to move this field forward,” Dr. Grulich said.
He added that it’s worth keeping an eye on two ongoing studies addressing key questions surrounding anal cancer in HIV-positive persons. The U.S. National Cancer Institute–funded randomized ANCHOR trial is examining ablative therapy versus watchful waiting in HIV-infected patients with anal HSIL lesions; however, results of this large study aren’t expected until 2022 or 2023. And Dr. Grulich heads the Study of the Prevention of Anal Cancer, aimed at identifying biomarkers that predict persistence of HSIL as a marker of anal cancer risk.
A study he would very much like to see funded is a randomized, placebo-controlled, adequately powered trial of the 9-valent HPV vaccine in HIV-infected gay or bisexual men over age 26. At the 2016 meeting of the Conference on Retroviruses and Opportunistic Infections (CROI), Timothy J. Wilkin, MD, of Cornell University, New York, presented the results of the phase III ACTG A5298 trial of the quadrivalent HPV vaccine in HIV-infected adults over age 26. The vaccine group had a 27% reduction in risk of persistent anal HPV compared with placebo, which wasn’t statistically significant because of the small study size. The 9-valent vaccine would prevent a broader range of oncogenic HPV types.
Dr. Grulich reported receiving research funding from CSL Australia, Gilead Sciences, Viiv, and Hologic.
DURBAN, SOUTH AFRICA – Screening for anal cancer in HIV-infected men or women should not be part of routine clinical practice at this time, Andrew Grulich, MBBS, PhD, declared at the 21st International AIDS Conference.
Some experts recommend anal cytologic screening or high-resolution anoscopy for HIV-positive men and women, but it’s worth noting that strategy hasn’t been incorporated into any national practice guidelines.
“And for very good reason: When we have a condition with a prevalence that’s so high and a treatment with recurrence rates that are so high, I think we need to question our approach,” said Dr. Grulich, professor of medicine and head of the HIV epidemiology and prevention program at the University of New South Wales in Sydney, Australia.
Screening proponents point to the high incidence of anal cancer in persons with HIV infection. It’s the fourth most common cancer in HIV patients in the United States, behind the AIDS-defining cancers and lung cancer. Indeed, the anal cancer rate is 10-fold greater in HIV-positive women, heterosexual men, and injection drug users than in the HIV-negative general population, and 50-fold higher in HIV-positive gay and bisexual men. Screening proponents also draw an analogy between anal cancer screening and the screening and treatment of cervical intraepithelial neoplasia (CIN), which has been enormously successful in preventing cervical cancer. But Dr. Grulich said he believes the cervical cancer screening analogy is faulty.
Colposcopy has a mean 90% specificity for diagnosis of HPV-related high-grade squamous intraepithelial lesions (HSIL) or cervical cancer, while high-resolution anoscopy as a diagnostic test has a specificity as low as 37% in HIV-positive persons. The prevalence of HSIL is 30%-40% in anal samples from HIV-infected homosexual men, compared with 1%-2% in cervical samples from HIV-negative women.
The rate of progression from CIN-3 to cervical cancer in women in the general population is about 1 in 80 per year. In contrast, the rate of progression from anal intraepithelial neoplasia (AIN)-2 or AIN-3 to anal cancer in HIV-infected homosexual men is estimated at only 1 in 400-600 per year, probably because regression of anal lesions is quite common.
Moreover, while a single treatment of high-grade CIN is typically curative and entails little morbidity, destruction of AIN by means of heat, cold, or electricity has a 70% failure rate, carries substantial morbidity, and is not supported by any evidence that it actually reduces the incidence of anal cancer, he continued.
“We’re in a bit of a quandary regarding what to do about anal cancer prevention. We really need research in order to move this field forward,” Dr. Grulich said.
He added that it’s worth keeping an eye on two ongoing studies addressing key questions surrounding anal cancer in HIV-positive persons. The U.S. National Cancer Institute–funded randomized ANCHOR trial is examining ablative therapy versus watchful waiting in HIV-infected patients with anal HSIL lesions; however, results of this large study aren’t expected until 2022 or 2023. And Dr. Grulich heads the Study of the Prevention of Anal Cancer, aimed at identifying biomarkers that predict persistence of HSIL as a marker of anal cancer risk.
A study he would very much like to see funded is a randomized, placebo-controlled, adequately powered trial of the 9-valent HPV vaccine in HIV-infected gay or bisexual men over age 26. At the 2016 meeting of the Conference on Retroviruses and Opportunistic Infections (CROI), Timothy J. Wilkin, MD, of Cornell University, New York, presented the results of the phase III ACTG A5298 trial of the quadrivalent HPV vaccine in HIV-infected adults over age 26. The vaccine group had a 27% reduction in risk of persistent anal HPV compared with placebo, which wasn’t statistically significant because of the small study size. The 9-valent vaccine would prevent a broader range of oncogenic HPV types.
Dr. Grulich reported receiving research funding from CSL Australia, Gilead Sciences, Viiv, and Hologic.
DURBAN, SOUTH AFRICA – Screening for anal cancer in HIV-infected men or women should not be part of routine clinical practice at this time, Andrew Grulich, MBBS, PhD, declared at the 21st International AIDS Conference.
Some experts recommend anal cytologic screening or high-resolution anoscopy for HIV-positive men and women, but it’s worth noting that strategy hasn’t been incorporated into any national practice guidelines.
“And for very good reason: When we have a condition with a prevalence that’s so high and a treatment with recurrence rates that are so high, I think we need to question our approach,” said Dr. Grulich, professor of medicine and head of the HIV epidemiology and prevention program at the University of New South Wales in Sydney, Australia.
Screening proponents point to the high incidence of anal cancer in persons with HIV infection. It’s the fourth most common cancer in HIV patients in the United States, behind the AIDS-defining cancers and lung cancer. Indeed, the anal cancer rate is 10-fold greater in HIV-positive women, heterosexual men, and injection drug users than in the HIV-negative general population, and 50-fold higher in HIV-positive gay and bisexual men. Screening proponents also draw an analogy between anal cancer screening and the screening and treatment of cervical intraepithelial neoplasia (CIN), which has been enormously successful in preventing cervical cancer. But Dr. Grulich said he believes the cervical cancer screening analogy is faulty.
Colposcopy has a mean 90% specificity for diagnosis of HPV-related high-grade squamous intraepithelial lesions (HSIL) or cervical cancer, while high-resolution anoscopy as a diagnostic test has a specificity as low as 37% in HIV-positive persons. The prevalence of HSIL is 30%-40% in anal samples from HIV-infected homosexual men, compared with 1%-2% in cervical samples from HIV-negative women.
The rate of progression from CIN-3 to cervical cancer in women in the general population is about 1 in 80 per year. In contrast, the rate of progression from anal intraepithelial neoplasia (AIN)-2 or AIN-3 to anal cancer in HIV-infected homosexual men is estimated at only 1 in 400-600 per year, probably because regression of anal lesions is quite common.
Moreover, while a single treatment of high-grade CIN is typically curative and entails little morbidity, destruction of AIN by means of heat, cold, or electricity has a 70% failure rate, carries substantial morbidity, and is not supported by any evidence that it actually reduces the incidence of anal cancer, he continued.
“We’re in a bit of a quandary regarding what to do about anal cancer prevention. We really need research in order to move this field forward,” Dr. Grulich said.
He added that it’s worth keeping an eye on two ongoing studies addressing key questions surrounding anal cancer in HIV-positive persons. The U.S. National Cancer Institute–funded randomized ANCHOR trial is examining ablative therapy versus watchful waiting in HIV-infected patients with anal HSIL lesions; however, results of this large study aren’t expected until 2022 or 2023. And Dr. Grulich heads the Study of the Prevention of Anal Cancer, aimed at identifying biomarkers that predict persistence of HSIL as a marker of anal cancer risk.
A study he would very much like to see funded is a randomized, placebo-controlled, adequately powered trial of the 9-valent HPV vaccine in HIV-infected gay or bisexual men over age 26. At the 2016 meeting of the Conference on Retroviruses and Opportunistic Infections (CROI), Timothy J. Wilkin, MD, of Cornell University, New York, presented the results of the phase III ACTG A5298 trial of the quadrivalent HPV vaccine in HIV-infected adults over age 26. The vaccine group had a 27% reduction in risk of persistent anal HPV compared with placebo, which wasn’t statistically significant because of the small study size. The 9-valent vaccine would prevent a broader range of oncogenic HPV types.
Dr. Grulich reported receiving research funding from CSL Australia, Gilead Sciences, Viiv, and Hologic.
EXPERT ANALYSIS FROM AIDS 2016
Cancer trends shifting in HIV-positive patients
DURBAN, SOUTH AFRICA – The rates of the AIDS-defining cancers Kaposi’s sarcoma and non-Hodgkin’s lymphoma have plummeted in the antiretroviral era, yet they are still the two top cancers in terms of cumulative incidence in HIV-infected patients by age 75, Benigno Rodriguez, MD, said at the 21st International AIDS Conference.
Lung cancer also remains a major concern in the HIV-infected population. Each of these three cancers carries about a 1 in 25 lifetime risk to age 75, the estimated lifespan of HIV-positive patients on combination antiretroviral therapy (ART), according to Dr. Rodriguez of Case Western Reserve University in Cleveland.
ART has resulted in a marked change in cancer trends among HIV-infected patients. The incidence of AIDS-defining cancers has decreased “massively,” Dr. Rodriguez observed; but because ART has extended the lifespan, patients are now living long enough to get other cancers. And they do so at a higher rate than that of the general population because of their impaired immune function, higher rate of risk factors such as smoking, and greater prevalence of oncogenic viral coinfections such as hepatitis B and C and Epstein-Barr virus.
The increase in the incidence and risk of colorectal, liver, and anal cancers among HIV-positive individuals since the introduction of combination ART can largely be explained by the population’s longer exposure to risk due to increasing survival, he said.
Dr. Rodriguez presented highlights of an analysis of cancer trends in North America during 1996-2009. The study was based on 86,620 HIV-infected persons with 475,660 person-years of follow-up and 196,987 subjects without HIV infection and with more than 1.8 million person-years of follow-up. Trends over time were assessed for nine cancers: Kaposi’s sarcoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and melanoma, as well as anal, lung, colorectal, liver, and oropharyngeal cancers (Ann Intern Med. 2015;163:507-18).
Examining trends in three periods – 1996-1999, 2000-2004, and 2005-2009 – the investigators looked at the impact of ART over time on rates of AIDS-related and non–AIDS-related cancers in HIV-infected patients and compared them to results in the general HIV-uninfected population.
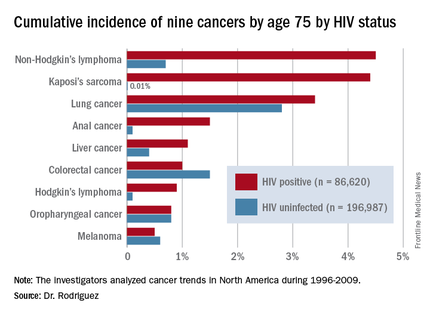
By conducting a competing risk analysis, Dr. Rodriguez and his coworkers were able to estimate the cumulative lifetime risk of the various cancers by age 75, a metric that provides readily understandable information for counseling HIV-infected patients about their long-term cancer risk.
The measure “is more intuitive than using incidence rates,” Dr. Rodriguez said. In a study of 1,578 HIV-infected patients who received the hepatitis B vaccine, for example, those patients whose immune function did not to respond to the vaccine were more likely to be among the 6% of patients who subsequently developed cancer during up to 20 years of follow-up.
The findings on cancer trends in the ART era have clinical implications for cancer screening and prevention in HIV-infected patients. The high rates of smoking and lung cancer in this population make HIV-positive smokers a logical target for lung cancer screening. The rising risk of colorectal cancer – the cumulative lifetime risk to age 75 was 0.4% in 1996-1999, 0.7% in 2000-2004, and 1.3% in 2005-2009 – suggests a need for increased colorectal cancer screening in the older HIV-positive population.
Early and sustained HIV suppression with combination ART remains the only known method of preventing AIDS-defining cancers. Dr. Rodriguez and his coinvestigators in the Centers for AIDS Research Network of Integrated Clinical Systems demonstrated the crucial role of suppressing HIV in a study of 6,036 HIV-infected patients who started on ART and were followed for more than 21,000 person-years. Compared with HIV-infected patients with a 3-month lagged HIV viremia of no more than 50 copies/mL, patients’ risk of developing non-Hodgkin’s lymphoma was 2.1-fold greater if their 3-month lagged HIV viremia was 51-500 copies/mL and 3.56-fold greater if it exceeded 500 copies/mL (Clin Infect Dis. 2014 Jun;58[11]:1599-606).
The study on cancer trends over time was funded by the National Institutes of Health. Dr. Rodriguez reported receiving honoraria from Gilead Sciences.
DURBAN, SOUTH AFRICA – The rates of the AIDS-defining cancers Kaposi’s sarcoma and non-Hodgkin’s lymphoma have plummeted in the antiretroviral era, yet they are still the two top cancers in terms of cumulative incidence in HIV-infected patients by age 75, Benigno Rodriguez, MD, said at the 21st International AIDS Conference.
Lung cancer also remains a major concern in the HIV-infected population. Each of these three cancers carries about a 1 in 25 lifetime risk to age 75, the estimated lifespan of HIV-positive patients on combination antiretroviral therapy (ART), according to Dr. Rodriguez of Case Western Reserve University in Cleveland.
ART has resulted in a marked change in cancer trends among HIV-infected patients. The incidence of AIDS-defining cancers has decreased “massively,” Dr. Rodriguez observed; but because ART has extended the lifespan, patients are now living long enough to get other cancers. And they do so at a higher rate than that of the general population because of their impaired immune function, higher rate of risk factors such as smoking, and greater prevalence of oncogenic viral coinfections such as hepatitis B and C and Epstein-Barr virus.
The increase in the incidence and risk of colorectal, liver, and anal cancers among HIV-positive individuals since the introduction of combination ART can largely be explained by the population’s longer exposure to risk due to increasing survival, he said.
Dr. Rodriguez presented highlights of an analysis of cancer trends in North America during 1996-2009. The study was based on 86,620 HIV-infected persons with 475,660 person-years of follow-up and 196,987 subjects without HIV infection and with more than 1.8 million person-years of follow-up. Trends over time were assessed for nine cancers: Kaposi’s sarcoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and melanoma, as well as anal, lung, colorectal, liver, and oropharyngeal cancers (Ann Intern Med. 2015;163:507-18).
Examining trends in three periods – 1996-1999, 2000-2004, and 2005-2009 – the investigators looked at the impact of ART over time on rates of AIDS-related and non–AIDS-related cancers in HIV-infected patients and compared them to results in the general HIV-uninfected population.

By conducting a competing risk analysis, Dr. Rodriguez and his coworkers were able to estimate the cumulative lifetime risk of the various cancers by age 75, a metric that provides readily understandable information for counseling HIV-infected patients about their long-term cancer risk.
The measure “is more intuitive than using incidence rates,” Dr. Rodriguez said. In a study of 1,578 HIV-infected patients who received the hepatitis B vaccine, for example, those patients whose immune function did not to respond to the vaccine were more likely to be among the 6% of patients who subsequently developed cancer during up to 20 years of follow-up.
The findings on cancer trends in the ART era have clinical implications for cancer screening and prevention in HIV-infected patients. The high rates of smoking and lung cancer in this population make HIV-positive smokers a logical target for lung cancer screening. The rising risk of colorectal cancer – the cumulative lifetime risk to age 75 was 0.4% in 1996-1999, 0.7% in 2000-2004, and 1.3% in 2005-2009 – suggests a need for increased colorectal cancer screening in the older HIV-positive population.
Early and sustained HIV suppression with combination ART remains the only known method of preventing AIDS-defining cancers. Dr. Rodriguez and his coinvestigators in the Centers for AIDS Research Network of Integrated Clinical Systems demonstrated the crucial role of suppressing HIV in a study of 6,036 HIV-infected patients who started on ART and were followed for more than 21,000 person-years. Compared with HIV-infected patients with a 3-month lagged HIV viremia of no more than 50 copies/mL, patients’ risk of developing non-Hodgkin’s lymphoma was 2.1-fold greater if their 3-month lagged HIV viremia was 51-500 copies/mL and 3.56-fold greater if it exceeded 500 copies/mL (Clin Infect Dis. 2014 Jun;58[11]:1599-606).
The study on cancer trends over time was funded by the National Institutes of Health. Dr. Rodriguez reported receiving honoraria from Gilead Sciences.
DURBAN, SOUTH AFRICA – The rates of the AIDS-defining cancers Kaposi’s sarcoma and non-Hodgkin’s lymphoma have plummeted in the antiretroviral era, yet they are still the two top cancers in terms of cumulative incidence in HIV-infected patients by age 75, Benigno Rodriguez, MD, said at the 21st International AIDS Conference.
Lung cancer also remains a major concern in the HIV-infected population. Each of these three cancers carries about a 1 in 25 lifetime risk to age 75, the estimated lifespan of HIV-positive patients on combination antiretroviral therapy (ART), according to Dr. Rodriguez of Case Western Reserve University in Cleveland.
ART has resulted in a marked change in cancer trends among HIV-infected patients. The incidence of AIDS-defining cancers has decreased “massively,” Dr. Rodriguez observed; but because ART has extended the lifespan, patients are now living long enough to get other cancers. And they do so at a higher rate than that of the general population because of their impaired immune function, higher rate of risk factors such as smoking, and greater prevalence of oncogenic viral coinfections such as hepatitis B and C and Epstein-Barr virus.
The increase in the incidence and risk of colorectal, liver, and anal cancers among HIV-positive individuals since the introduction of combination ART can largely be explained by the population’s longer exposure to risk due to increasing survival, he said.
Dr. Rodriguez presented highlights of an analysis of cancer trends in North America during 1996-2009. The study was based on 86,620 HIV-infected persons with 475,660 person-years of follow-up and 196,987 subjects without HIV infection and with more than 1.8 million person-years of follow-up. Trends over time were assessed for nine cancers: Kaposi’s sarcoma, non-Hodgkin’s lymphoma, Hodgkin’s lymphoma, and melanoma, as well as anal, lung, colorectal, liver, and oropharyngeal cancers (Ann Intern Med. 2015;163:507-18).
Examining trends in three periods – 1996-1999, 2000-2004, and 2005-2009 – the investigators looked at the impact of ART over time on rates of AIDS-related and non–AIDS-related cancers in HIV-infected patients and compared them to results in the general HIV-uninfected population.

By conducting a competing risk analysis, Dr. Rodriguez and his coworkers were able to estimate the cumulative lifetime risk of the various cancers by age 75, a metric that provides readily understandable information for counseling HIV-infected patients about their long-term cancer risk.
The measure “is more intuitive than using incidence rates,” Dr. Rodriguez said. In a study of 1,578 HIV-infected patients who received the hepatitis B vaccine, for example, those patients whose immune function did not to respond to the vaccine were more likely to be among the 6% of patients who subsequently developed cancer during up to 20 years of follow-up.
The findings on cancer trends in the ART era have clinical implications for cancer screening and prevention in HIV-infected patients. The high rates of smoking and lung cancer in this population make HIV-positive smokers a logical target for lung cancer screening. The rising risk of colorectal cancer – the cumulative lifetime risk to age 75 was 0.4% in 1996-1999, 0.7% in 2000-2004, and 1.3% in 2005-2009 – suggests a need for increased colorectal cancer screening in the older HIV-positive population.
Early and sustained HIV suppression with combination ART remains the only known method of preventing AIDS-defining cancers. Dr. Rodriguez and his coinvestigators in the Centers for AIDS Research Network of Integrated Clinical Systems demonstrated the crucial role of suppressing HIV in a study of 6,036 HIV-infected patients who started on ART and were followed for more than 21,000 person-years. Compared with HIV-infected patients with a 3-month lagged HIV viremia of no more than 50 copies/mL, patients’ risk of developing non-Hodgkin’s lymphoma was 2.1-fold greater if their 3-month lagged HIV viremia was 51-500 copies/mL and 3.56-fold greater if it exceeded 500 copies/mL (Clin Infect Dis. 2014 Jun;58[11]:1599-606).
The study on cancer trends over time was funded by the National Institutes of Health. Dr. Rodriguez reported receiving honoraria from Gilead Sciences.
AT AIDS 2016
Key clinical point: HIV-infected persons in North America have roughly a 1 in 25 cumulative lifetime risk of developing lung cancer, Kaposi’s sarcoma, or non-Hodgkin’s lymphoma.
Major finding: The cumulative lifetime risk of developing non-Hodgkin’s lymphoma in HIV-infected patients on combination antiretroviral therapy is sevenfold greater than the risk in the general population.
Data source: A competing risk analysis for nine cancers based upon 86,620 HIV-infected persons followed for 475,660 person-years and 196,987 subjects not infected with HIV and with more than 1.8 million person-years of follow-up.
Disclosures: The National Institutes of Health funded the study. The presenter reported receiving honoraria from Gilead Sciences.
Aortomitral continuity calcification predicts new atrial fib after TAVR
PARIS – Aortomitral continuity calcification, a common finding on CT in patients undergoing transcatheter aortic valve replacement, predicts new-onset atrial fibrillation and the need for permanent pacemaker insertion, Marco Spaziano, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Increased surveillance for arrhythmias in the 30 days post TAVR is warranted in patients with aortomitral continuity calcification,” declared Dr. Spaziano of the Paris South Cardiovascular Institute in Massy, France.
He presented a single-center retrospective study of 524 patients undergoing TAVR with a self-expandable or balloon-expandable device. Aortomitral continuity calcification (AMCC) was found on CT in 15.8% of them. Dr. Spaziano defined AMCC as the presence of calcium in the curtain linking the aortic and mitral valve annuli. The clinical implications of this common finding were unknown prior to this study.
The 83 patients with AMCC did not differ significantly from the 441 without that CT finding in terms of baseline demographics, Society of Thoracic Surgeons risk score, prevalence of peripheral vascular disease, QRS duration, left ventricular ejection fraction, complete left or right bundle branch block, or aortic valve calcification volume. The prevalence of atrial fibrillation at baseline was 25.6% in the AMCC group and closely similar at 26.3% in the group without AMCC. Sixteen percent of subjects in each group had a previous pacemaker.
Similarly, the two groups didn’t differ in terms of procedural characteristics, including device type, size, or depth of implantation, or need for a second valve, or annular rupture.
However, excluding from consideration the patients with prior AF, the incidence of new AF in the 30 days post-TAVR was 22.7% in patients with AMCC compared with just 7.6% in the no-AMCC group. In addition, 33% of patients with AMCC received a new permanent pacemaker, as did 21% of those with no AMCC.
Other key 30-day outcomes didn’t differ between the two populations, including rates of death, stroke, vascular complications, and moderate or severe paravalvular regurgitation.
In a multivariate regression analysis adjusted for age, sex, device type and implantation depth, preexisting right bundle branch block, and surgical risk score, AMCC was associated with a statistically significant 1.8-fold increased likelihood of new pacemaker insertion and a 3.4-fold greater risk of new AF.
Dr. Spaziano said that in brainstorming with electrophysiology and echocardiography colleagues, the group came up with two hypotheses to explain the study findings. One is that AMCC might be a biologic marker for concomitant mitral stenosis, a known strong predictor of AF.
“Oftentimes it’s very difficult to diagnose mitral stenosis when there is aortic stenosis, because of left ventricular compliance issues, so potentially the patients with this calcium ridge may also have mitral stenosis,” he observed.
The other proposed hypothesis is that AMCC reflects increased calcification and fibrosis in the electrical system of both the AV node and atrium, with a resultant increased risk of developing new AF after the TAVR procedure.
Session chair Mohammad Abdelghani, MD, wasn’t buying either hypothesis. If either were correct, the group with AMCC would be expected to have a higher baseline rate of AF preprocedurally, observed Dr. Abdelghani of the Academic Medical Center at Amsterdam.
He suggested an alternative explanation on the basis of a German study that showed patients with significant calcification of the left coronary cusp were at sixfold greater risk for pacemaker implantation post TAVR. He proposed that calcification in the left sector of the valve landing zone causes the device to end up being positioned a bit off-line.
“I think the device protrudes away from the calcium and towards the right coronary artery commisure, compressing the conduction system that we know lies there,” Dr. Abdelghani said.
Dr. Spaziano reported having no financial conflicts of interest regarding his study.
PARIS – Aortomitral continuity calcification, a common finding on CT in patients undergoing transcatheter aortic valve replacement, predicts new-onset atrial fibrillation and the need for permanent pacemaker insertion, Marco Spaziano, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Increased surveillance for arrhythmias in the 30 days post TAVR is warranted in patients with aortomitral continuity calcification,” declared Dr. Spaziano of the Paris South Cardiovascular Institute in Massy, France.
He presented a single-center retrospective study of 524 patients undergoing TAVR with a self-expandable or balloon-expandable device. Aortomitral continuity calcification (AMCC) was found on CT in 15.8% of them. Dr. Spaziano defined AMCC as the presence of calcium in the curtain linking the aortic and mitral valve annuli. The clinical implications of this common finding were unknown prior to this study.
The 83 patients with AMCC did not differ significantly from the 441 without that CT finding in terms of baseline demographics, Society of Thoracic Surgeons risk score, prevalence of peripheral vascular disease, QRS duration, left ventricular ejection fraction, complete left or right bundle branch block, or aortic valve calcification volume. The prevalence of atrial fibrillation at baseline was 25.6% in the AMCC group and closely similar at 26.3% in the group without AMCC. Sixteen percent of subjects in each group had a previous pacemaker.
Similarly, the two groups didn’t differ in terms of procedural characteristics, including device type, size, or depth of implantation, or need for a second valve, or annular rupture.
However, excluding from consideration the patients with prior AF, the incidence of new AF in the 30 days post-TAVR was 22.7% in patients with AMCC compared with just 7.6% in the no-AMCC group. In addition, 33% of patients with AMCC received a new permanent pacemaker, as did 21% of those with no AMCC.
Other key 30-day outcomes didn’t differ between the two populations, including rates of death, stroke, vascular complications, and moderate or severe paravalvular regurgitation.
In a multivariate regression analysis adjusted for age, sex, device type and implantation depth, preexisting right bundle branch block, and surgical risk score, AMCC was associated with a statistically significant 1.8-fold increased likelihood of new pacemaker insertion and a 3.4-fold greater risk of new AF.
Dr. Spaziano said that in brainstorming with electrophysiology and echocardiography colleagues, the group came up with two hypotheses to explain the study findings. One is that AMCC might be a biologic marker for concomitant mitral stenosis, a known strong predictor of AF.
“Oftentimes it’s very difficult to diagnose mitral stenosis when there is aortic stenosis, because of left ventricular compliance issues, so potentially the patients with this calcium ridge may also have mitral stenosis,” he observed.
The other proposed hypothesis is that AMCC reflects increased calcification and fibrosis in the electrical system of both the AV node and atrium, with a resultant increased risk of developing new AF after the TAVR procedure.
Session chair Mohammad Abdelghani, MD, wasn’t buying either hypothesis. If either were correct, the group with AMCC would be expected to have a higher baseline rate of AF preprocedurally, observed Dr. Abdelghani of the Academic Medical Center at Amsterdam.
He suggested an alternative explanation on the basis of a German study that showed patients with significant calcification of the left coronary cusp were at sixfold greater risk for pacemaker implantation post TAVR. He proposed that calcification in the left sector of the valve landing zone causes the device to end up being positioned a bit off-line.
“I think the device protrudes away from the calcium and towards the right coronary artery commisure, compressing the conduction system that we know lies there,” Dr. Abdelghani said.
Dr. Spaziano reported having no financial conflicts of interest regarding his study.
PARIS – Aortomitral continuity calcification, a common finding on CT in patients undergoing transcatheter aortic valve replacement, predicts new-onset atrial fibrillation and the need for permanent pacemaker insertion, Marco Spaziano, MD, reported at the annual congress of the European Association of Percutaneous Cardiovascular Interventions.
“Increased surveillance for arrhythmias in the 30 days post TAVR is warranted in patients with aortomitral continuity calcification,” declared Dr. Spaziano of the Paris South Cardiovascular Institute in Massy, France.
He presented a single-center retrospective study of 524 patients undergoing TAVR with a self-expandable or balloon-expandable device. Aortomitral continuity calcification (AMCC) was found on CT in 15.8% of them. Dr. Spaziano defined AMCC as the presence of calcium in the curtain linking the aortic and mitral valve annuli. The clinical implications of this common finding were unknown prior to this study.
The 83 patients with AMCC did not differ significantly from the 441 without that CT finding in terms of baseline demographics, Society of Thoracic Surgeons risk score, prevalence of peripheral vascular disease, QRS duration, left ventricular ejection fraction, complete left or right bundle branch block, or aortic valve calcification volume. The prevalence of atrial fibrillation at baseline was 25.6% in the AMCC group and closely similar at 26.3% in the group without AMCC. Sixteen percent of subjects in each group had a previous pacemaker.
Similarly, the two groups didn’t differ in terms of procedural characteristics, including device type, size, or depth of implantation, or need for a second valve, or annular rupture.
However, excluding from consideration the patients with prior AF, the incidence of new AF in the 30 days post-TAVR was 22.7% in patients with AMCC compared with just 7.6% in the no-AMCC group. In addition, 33% of patients with AMCC received a new permanent pacemaker, as did 21% of those with no AMCC.
Other key 30-day outcomes didn’t differ between the two populations, including rates of death, stroke, vascular complications, and moderate or severe paravalvular regurgitation.
In a multivariate regression analysis adjusted for age, sex, device type and implantation depth, preexisting right bundle branch block, and surgical risk score, AMCC was associated with a statistically significant 1.8-fold increased likelihood of new pacemaker insertion and a 3.4-fold greater risk of new AF.
Dr. Spaziano said that in brainstorming with electrophysiology and echocardiography colleagues, the group came up with two hypotheses to explain the study findings. One is that AMCC might be a biologic marker for concomitant mitral stenosis, a known strong predictor of AF.
“Oftentimes it’s very difficult to diagnose mitral stenosis when there is aortic stenosis, because of left ventricular compliance issues, so potentially the patients with this calcium ridge may also have mitral stenosis,” he observed.
The other proposed hypothesis is that AMCC reflects increased calcification and fibrosis in the electrical system of both the AV node and atrium, with a resultant increased risk of developing new AF after the TAVR procedure.
Session chair Mohammad Abdelghani, MD, wasn’t buying either hypothesis. If either were correct, the group with AMCC would be expected to have a higher baseline rate of AF preprocedurally, observed Dr. Abdelghani of the Academic Medical Center at Amsterdam.
He suggested an alternative explanation on the basis of a German study that showed patients with significant calcification of the left coronary cusp were at sixfold greater risk for pacemaker implantation post TAVR. He proposed that calcification in the left sector of the valve landing zone causes the device to end up being positioned a bit off-line.
“I think the device protrudes away from the calcium and towards the right coronary artery commisure, compressing the conduction system that we know lies there,” Dr. Abdelghani said.
Dr. Spaziano reported having no financial conflicts of interest regarding his study.
AT europcr 2016
Key clinical point: Aortomitral continuity calcification is associated with a markedly increased risk of new atrial fibrillation in patients undergoing transcatheter aortic valve replacement.
Major finding: The CT finding of aortomitral continuity calcification in patients undergoing transcatheter aortic valve replacement was associated with a 3.4-fold increased likelihood of new atrial fibrillation arising during the first 30 days post procedure.
Data source: A retrospective single-center study in 524 patients undergoing transcatheter aortic valve replacement, nearly 16% of whom were found to have aortomitral continuity calcification.
Disclosures: The presenter reported having no financial conflicts of interest regarding his study.