User login
Bioactive lipid shows promise in atopic dermatitis
VIENNA – A novel oral agent known as DS107 showed promise as a safe and effective treatment for moderate-to-severe atopic dermatitis in a phase IIa proof-of-concept study, Diamant Thaçi, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Clear efficacy signals in the reduction of clinical symptoms of atopic dermatitis were detected within 2 weeks of treatment, with the maximum improvement in the endpoints observed between weeks 4 and 8,” said Dr. Thaçi, professor of dermatology and head of the Comprehensive Center for Inflammation Medicine at University Hospital Schleswig-Holstein, in Lübeck, Germany.
Dr. Thaçi presented the results of an 8-week, double-blind, randomized, placebo-controlled, multicenter phase IIa study that included 102 patients with moderate-to-severe AD. Participants averaged a baseline Investigator Global Assessment (IGA) score of 3.5 on the 0-5 scale. They were randomized to 2 g of oral DS107 once daily or an equal quantity of mineral oil as a placebo control.
Based upon the encouraging findings, a 300-patient phase IIb study will get underway soon. It will examine the effects of 1 g of DS107 per day as well as 2 g in the hope that the lower dose will cut down on the high rate of minor GI side effects seen at 2 g/day while preserving the efficacy of the higher dose.
The primary efficacy endpoint in the phase IIa study was at least a 2-point drop from baseline in IGA plus an end-of-treatment IGA of 0 or 1, meaning clear or almost clear. In the intent-to-treat analysis, this was achieved in 21.6% of the group assigned to DS107, compared with 11.8% of controls. In the 71 participants who actually completed 8 weeks of treatment – 35 in the DS107 arm, 36 controls – the composite efficacy endpoint was achieved in 31.4% of those on active treatment and 16.7% on mineral oil.
Significant separation between the active treatment and control arms in terms of itch visual analog scores was seen by week 4. This was a particularly encouraging finding, since patients report pruritis to be the most troublesome symptom of AD, Dr. Thaçi noted.
Significantly greater improvement in quality of life as measured by the Patient-Oriented Eczema Measure (POEM) was also seen by week 4 in the DS107 group as compared with controls.
No severe adverse events occurred in the study. However, more than one-quarter of subjects interrupted or discontinued participation because of mild nausea, loose stools, and/or abdominal pain. These issues were equally common in the DS107 and mineral oil groups, and Dr. Thaçi and his coinvestigators suspect that for many patients it was simply a matter of too much oil in the stomach. The GI symptoms resolved quickly without intervention after a brief halt of therapy, but some patients never returned to participation.
“This problem can be solved in the future with a different dosing design or even a different method of delivering the DGLA,” the dermatologist added.
Asked about the significant placebo response seen in the study, Dr. Thaçi shrugged it off as “quite understandable.”
“Placebo is not always a placebo. There is the feeling of fullness in the stomach, there is some emollient effect, the continuous contact with the physician. You see this in all the clinical trials in atopic dermatitis: in the beginning, the first 2-3 weeks, you have some influence of placebo,” he said.
The placebo effect was greatly diminished in patients with more severe AD. In the subset with a baseline IGA of 4 or 5, none of the control subjects achieved the primary efficacy endpoint, while more than 20% on DS107 did, Dr. Thaçi noted.
The mechanism of benefit of DGLA is not fully understood as yet. Animal studies point to an antibacterial effect, and DGLA also reduces levels of inflammatory cytokines. Eosinophilia was reduced to a much greater extent in the DS107 group than controls.
Dr. Thaçi reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
bjancin@frontlinemedcom.com
VIENNA – A novel oral agent known as DS107 showed promise as a safe and effective treatment for moderate-to-severe atopic dermatitis in a phase IIa proof-of-concept study, Diamant Thaçi, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Clear efficacy signals in the reduction of clinical symptoms of atopic dermatitis were detected within 2 weeks of treatment, with the maximum improvement in the endpoints observed between weeks 4 and 8,” said Dr. Thaçi, professor of dermatology and head of the Comprehensive Center for Inflammation Medicine at University Hospital Schleswig-Holstein, in Lübeck, Germany.
Dr. Thaçi presented the results of an 8-week, double-blind, randomized, placebo-controlled, multicenter phase IIa study that included 102 patients with moderate-to-severe AD. Participants averaged a baseline Investigator Global Assessment (IGA) score of 3.5 on the 0-5 scale. They were randomized to 2 g of oral DS107 once daily or an equal quantity of mineral oil as a placebo control.
Based upon the encouraging findings, a 300-patient phase IIb study will get underway soon. It will examine the effects of 1 g of DS107 per day as well as 2 g in the hope that the lower dose will cut down on the high rate of minor GI side effects seen at 2 g/day while preserving the efficacy of the higher dose.
The primary efficacy endpoint in the phase IIa study was at least a 2-point drop from baseline in IGA plus an end-of-treatment IGA of 0 or 1, meaning clear or almost clear. In the intent-to-treat analysis, this was achieved in 21.6% of the group assigned to DS107, compared with 11.8% of controls. In the 71 participants who actually completed 8 weeks of treatment – 35 in the DS107 arm, 36 controls – the composite efficacy endpoint was achieved in 31.4% of those on active treatment and 16.7% on mineral oil.
Significant separation between the active treatment and control arms in terms of itch visual analog scores was seen by week 4. This was a particularly encouraging finding, since patients report pruritis to be the most troublesome symptom of AD, Dr. Thaçi noted.
Significantly greater improvement in quality of life as measured by the Patient-Oriented Eczema Measure (POEM) was also seen by week 4 in the DS107 group as compared with controls.
No severe adverse events occurred in the study. However, more than one-quarter of subjects interrupted or discontinued participation because of mild nausea, loose stools, and/or abdominal pain. These issues were equally common in the DS107 and mineral oil groups, and Dr. Thaçi and his coinvestigators suspect that for many patients it was simply a matter of too much oil in the stomach. The GI symptoms resolved quickly without intervention after a brief halt of therapy, but some patients never returned to participation.
“This problem can be solved in the future with a different dosing design or even a different method of delivering the DGLA,” the dermatologist added.
Asked about the significant placebo response seen in the study, Dr. Thaçi shrugged it off as “quite understandable.”
“Placebo is not always a placebo. There is the feeling of fullness in the stomach, there is some emollient effect, the continuous contact with the physician. You see this in all the clinical trials in atopic dermatitis: in the beginning, the first 2-3 weeks, you have some influence of placebo,” he said.
The placebo effect was greatly diminished in patients with more severe AD. In the subset with a baseline IGA of 4 or 5, none of the control subjects achieved the primary efficacy endpoint, while more than 20% on DS107 did, Dr. Thaçi noted.
The mechanism of benefit of DGLA is not fully understood as yet. Animal studies point to an antibacterial effect, and DGLA also reduces levels of inflammatory cytokines. Eosinophilia was reduced to a much greater extent in the DS107 group than controls.
Dr. Thaçi reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
bjancin@frontlinemedcom.com
VIENNA – A novel oral agent known as DS107 showed promise as a safe and effective treatment for moderate-to-severe atopic dermatitis in a phase IIa proof-of-concept study, Diamant Thaçi, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
“Clear efficacy signals in the reduction of clinical symptoms of atopic dermatitis were detected within 2 weeks of treatment, with the maximum improvement in the endpoints observed between weeks 4 and 8,” said Dr. Thaçi, professor of dermatology and head of the Comprehensive Center for Inflammation Medicine at University Hospital Schleswig-Holstein, in Lübeck, Germany.
Dr. Thaçi presented the results of an 8-week, double-blind, randomized, placebo-controlled, multicenter phase IIa study that included 102 patients with moderate-to-severe AD. Participants averaged a baseline Investigator Global Assessment (IGA) score of 3.5 on the 0-5 scale. They were randomized to 2 g of oral DS107 once daily or an equal quantity of mineral oil as a placebo control.
Based upon the encouraging findings, a 300-patient phase IIb study will get underway soon. It will examine the effects of 1 g of DS107 per day as well as 2 g in the hope that the lower dose will cut down on the high rate of minor GI side effects seen at 2 g/day while preserving the efficacy of the higher dose.
The primary efficacy endpoint in the phase IIa study was at least a 2-point drop from baseline in IGA plus an end-of-treatment IGA of 0 or 1, meaning clear or almost clear. In the intent-to-treat analysis, this was achieved in 21.6% of the group assigned to DS107, compared with 11.8% of controls. In the 71 participants who actually completed 8 weeks of treatment – 35 in the DS107 arm, 36 controls – the composite efficacy endpoint was achieved in 31.4% of those on active treatment and 16.7% on mineral oil.
Significant separation between the active treatment and control arms in terms of itch visual analog scores was seen by week 4. This was a particularly encouraging finding, since patients report pruritis to be the most troublesome symptom of AD, Dr. Thaçi noted.
Significantly greater improvement in quality of life as measured by the Patient-Oriented Eczema Measure (POEM) was also seen by week 4 in the DS107 group as compared with controls.
No severe adverse events occurred in the study. However, more than one-quarter of subjects interrupted or discontinued participation because of mild nausea, loose stools, and/or abdominal pain. These issues were equally common in the DS107 and mineral oil groups, and Dr. Thaçi and his coinvestigators suspect that for many patients it was simply a matter of too much oil in the stomach. The GI symptoms resolved quickly without intervention after a brief halt of therapy, but some patients never returned to participation.
“This problem can be solved in the future with a different dosing design or even a different method of delivering the DGLA,” the dermatologist added.
Asked about the significant placebo response seen in the study, Dr. Thaçi shrugged it off as “quite understandable.”
“Placebo is not always a placebo. There is the feeling of fullness in the stomach, there is some emollient effect, the continuous contact with the physician. You see this in all the clinical trials in atopic dermatitis: in the beginning, the first 2-3 weeks, you have some influence of placebo,” he said.
The placebo effect was greatly diminished in patients with more severe AD. In the subset with a baseline IGA of 4 or 5, none of the control subjects achieved the primary efficacy endpoint, while more than 20% on DS107 did, Dr. Thaçi noted.
The mechanism of benefit of DGLA is not fully understood as yet. Animal studies point to an antibacterial effect, and DGLA also reduces levels of inflammatory cytokines. Eosinophilia was reduced to a much greater extent in the DS107 group than controls.
Dr. Thaçi reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
bjancin@frontlinemedcom.com
AT THE EADV CONGRESS
Key clinical point:
Major finding: Once-daily oral DS107, which contains a bioactive lipid, achieved significant improvement in moderate-to-severe atopic dermatitis in 31.4% of patients, compared with 16.7% on placebo.
Data source: This phase IIa, double-blind, randomized, placebo-controlled, multicenter 8-week trial included 102 adults with moderate-to-severe atopic dermatitis.
Disclosures: The study presenter reported serving as a consultant to DS Pharma, the privately held biopharmaceutical company that is developing oral DS107, as well as to numerous other pharmaceutical companies.
Cardiorespiratory fitness improves survival after depression
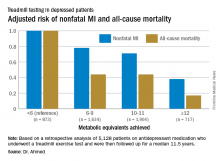
ROME – Cardiorespiratory fitness provided strong and graded protection against all-cause mortality and nonfatal MI in a study of more than 5,000 patients treated for depression, Amjad M. Ahmed, MD, reported at the annual congress of the European Society of Cardiology.
“These results highlight the importance of assessing fitness to identify risk as well as promoting an active lifestyle in patients with depression,” said Dr. Ahmed of Abdulaziz University for Health Sciences in Riyadh, Saudi Arabia.
This analysis focused on the 5,128 subjects who were on antidepressant medication at the time of their treadmill test. Their baseline cardiorespiratory fitness, as estimated by achieved peak metabolic equivalents (METs) on the treadmill, varied inversely with their risks of acute MI and all-cause mortality in the years to come. However, the less fit a patient was, the greater the burden of traditional cardiovascular risk factors. For example, the prevalence of hypertension was 86% in patients who achieved fewer than 6 METs, 75% in those who achieved 6-9 METs, 62% in depressed patients who reached 10-11 METs, and 51% in those who achieved 12 METs or more.
For this reason, Dr. Ahmed and coinvestigators performed a Cox multivariate regression analysis adjusted extensively for potential confounders, including age, sex, race, cardiovascular risk factors, known coronary artery disease, the use of cardiovascular medications, and the reason for the referral for stress testing.
When an achieved MET below 6 was used as the reference standard, for every 1 MET above 6 that patients achieved, their adjusted risk of all-cause mortality decreased by 18%, and the risk of nonfatal MI fell by 8%.
Session cochair Martin Halle, MD, pointed out what he viewed as a major limitation of the study.
“You didn’t follow their physical fitness over time, so you can’t say that increasing their METs would bring a better prognosis,” said Dr. Halle, professor and chairman of the department of preventive and rehabilitative sports medicine at the Technical University of Munich.
Dr. Ahmed reported having no financial conflicts of interest related to the Henry Ford FIT Project.
ROME – Cardiorespiratory fitness provided strong and graded protection against all-cause mortality and nonfatal MI in a study of more than 5,000 patients treated for depression, Amjad M. Ahmed, MD, reported at the annual congress of the European Society of Cardiology.
“These results highlight the importance of assessing fitness to identify risk as well as promoting an active lifestyle in patients with depression,” said Dr. Ahmed of Abdulaziz University for Health Sciences in Riyadh, Saudi Arabia.
This analysis focused on the 5,128 subjects who were on antidepressant medication at the time of their treadmill test. Their baseline cardiorespiratory fitness, as estimated by achieved peak metabolic equivalents (METs) on the treadmill, varied inversely with their risks of acute MI and all-cause mortality in the years to come. However, the less fit a patient was, the greater the burden of traditional cardiovascular risk factors. For example, the prevalence of hypertension was 86% in patients who achieved fewer than 6 METs, 75% in those who achieved 6-9 METs, 62% in depressed patients who reached 10-11 METs, and 51% in those who achieved 12 METs or more.
For this reason, Dr. Ahmed and coinvestigators performed a Cox multivariate regression analysis adjusted extensively for potential confounders, including age, sex, race, cardiovascular risk factors, known coronary artery disease, the use of cardiovascular medications, and the reason for the referral for stress testing.
When an achieved MET below 6 was used as the reference standard, for every 1 MET above 6 that patients achieved, their adjusted risk of all-cause mortality decreased by 18%, and the risk of nonfatal MI fell by 8%.
Session cochair Martin Halle, MD, pointed out what he viewed as a major limitation of the study.
“You didn’t follow their physical fitness over time, so you can’t say that increasing their METs would bring a better prognosis,” said Dr. Halle, professor and chairman of the department of preventive and rehabilitative sports medicine at the Technical University of Munich.
Dr. Ahmed reported having no financial conflicts of interest related to the Henry Ford FIT Project.
ROME – Cardiorespiratory fitness provided strong and graded protection against all-cause mortality and nonfatal MI in a study of more than 5,000 patients treated for depression, Amjad M. Ahmed, MD, reported at the annual congress of the European Society of Cardiology.
“These results highlight the importance of assessing fitness to identify risk as well as promoting an active lifestyle in patients with depression,” said Dr. Ahmed of Abdulaziz University for Health Sciences in Riyadh, Saudi Arabia.
This analysis focused on the 5,128 subjects who were on antidepressant medication at the time of their treadmill test. Their baseline cardiorespiratory fitness, as estimated by achieved peak metabolic equivalents (METs) on the treadmill, varied inversely with their risks of acute MI and all-cause mortality in the years to come. However, the less fit a patient was, the greater the burden of traditional cardiovascular risk factors. For example, the prevalence of hypertension was 86% in patients who achieved fewer than 6 METs, 75% in those who achieved 6-9 METs, 62% in depressed patients who reached 10-11 METs, and 51% in those who achieved 12 METs or more.
For this reason, Dr. Ahmed and coinvestigators performed a Cox multivariate regression analysis adjusted extensively for potential confounders, including age, sex, race, cardiovascular risk factors, known coronary artery disease, the use of cardiovascular medications, and the reason for the referral for stress testing.
When an achieved MET below 6 was used as the reference standard, for every 1 MET above 6 that patients achieved, their adjusted risk of all-cause mortality decreased by 18%, and the risk of nonfatal MI fell by 8%.
Session cochair Martin Halle, MD, pointed out what he viewed as a major limitation of the study.
“You didn’t follow their physical fitness over time, so you can’t say that increasing their METs would bring a better prognosis,” said Dr. Halle, professor and chairman of the department of preventive and rehabilitative sports medicine at the Technical University of Munich.
Dr. Ahmed reported having no financial conflicts of interest related to the Henry Ford FIT Project.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: For every 1-MET increase a patient on antidepressant medication achieved above 6 METs during a Bruce protocol treadmill exercise test, the risk of all-cause mortality during the subsequent 11.5 years decreased by an adjusted 18%.
Data source: A retrospective analysis of 5,128 patients on antidepressant medication who underwent a treadmill exercise test as part of the Henry Ford Exercise Testing Project and were then followed up for a median of 11.5 years.
Disclosures: The study presenter reported having no relevant financial conflicts.
Carotid stenting tied to cardiovascular events in real-world study
ROME – Carotid stenting was associated with a roughly 30% higher risk of cardiovascular events than that of carotid endarterectomy during 12 years of follow-up in a large, real-world, population-based cohort study, Mohamad A. Hussain, MD, reported at the annual congress of the European Society of Cardiology.
“Our data raise concerns about the external validity of randomized controlled trials of carotid endarterectomy versus stenting and question the potential interchangeability of carotid endarterectomy and stenting as stated in clinical practice guidelines,” said Dr. Hussain of the University of Toronto.
Major practice guidelines cite randomized trial evidence in suggesting that CEA and stenting can be used interchangeably in treating low- or average-risk patients with significant carotid artery disease. Dr. Hussain and his coinvestigators, suspicious that the generalizability of the randomized trial findings may be limited because of operator and institutional selection bias, decided to conduct a retrospective cohort study of all patients over age 40 years who underwent CEA or carotid stenting in the province of Ontario from April 2002 through March 2013.
Using validated chart abstraction software, they identified 12,529 patients who had CEA and 1,935 with carotid stenting. The two groups were similar in terms of most baseline characteristics. Notably, however, stent recipients were significantly more likely to have symptomatic carotid disease and also had more comorbid conditions as reflected in a higher Charlson Comorbidity Index score.
The primary outcome in the study was the 12-year rate of a composite comprising ischemic stroke, transient ischemic attack (TIA), MI, or death. The rate was 35.4% in the CEA group and 44.5% in the stent group. After adjustment for the baseline differences, the stent group still had a statistically significant 28% greater risk of the primary outcome.
“We found the difference remained significant in all of our subgroup analyses, regardless of age, sex, year of procedure, symptomatic or asymptomatic carotid artery disease, CAD [coronary artery disease] or no CAD, diabetes (type 1 or 2) or no diabetes. Outcomes with endarterectomy were always significantly better,” said Dr. Hussain.
“I think our study shows that in clinical practice we’re not quite seeing the outcomes reported in the clinical trials,” he added.
As for the individual components of the composite endpoint, the 12-year rate of ischemic stroke or TIA was 9% in the CEA group and 14% with stenting, for an adjusted 40% increased risk in the stent group. The 12-year all-cause mortality rate was 26% in the CEA group and 34% with stenting, for an adjusted 28% increased risk. The incidence of MI was 8% in both groups.
The investigators next conducted a confirmatory propensity-matched analysis in which 1,927 of the stented patients were closely matched to 3,844 surgical patients, eliminating baseline differences in the prevalence of symptomatic carotid artery disease and other disparities. In this matched cohort, the primary outcome occurred in 37.4% of the CEA group and 44.3% of stent patients, for an adjusted 32% increase in risk in the stented group.
The differences in outcome were driven by sharply higher periprocedural risk in the stented group. After the periprocedural period, the outcome curves remained parallel in the two treatment groups.
In that first 30 days post procedure, the primary composite outcome occurred in 5.4% of the CEA group and 10% of stented patients, for an adjusted 40% increase in relative risk in percutaneously treated patients. The 30-day rate of ischemic stroke or TIA was 3.4% in the surgical group compared with 6.4% in stented patients. Thirty-day mortality was 0.9% with CEA versus 3.3% with stenting.
Asked by the award panel for his thoughts on the disparity between the results of his real-world study and the major randomized trials of CEA versus stenting, Dr. Hussain replied, “It may be because the trials had high-volume operators at high-volume centers who are really experts in carotid stenting, while in the real world many physicians may not be selecting the right people for carotid stenting.”
Differences in sample size may also figure in the disparity, he continued. He noted that in the recent 10-year report from the Carotid Revascularization Endarterectomy versus Stenting Trial (CREST), the composite endpoint of stroke, MI, or death occurred in 9.9% of the CEA group compared with 11.8% of the stenting group (N Engl J Med. 2016 Mar 17;374[11]:1021-31), but this difference in favor of CEA didn’t achieve statistical significance because of the wide confidence intervals resulting from a smaller sample size than in the Ontario study.
Looking to the future, Dr. Hussain said he thinks the ongoing CREST-2 trial is “very important.” It is randomizing patients with asymptomatic high-grade carotid stenosis to uniform intensive medical management either alone or in combination with CEA or stenting with embolic protection.
“That study might end up showing us that medical therapy is as good as or even better than stenting or CEA, especially in asymptomatic patients,” he said.
Dr. Hussain reported having no financial conflicts regarding his academically funded study.
ROME – Carotid stenting was associated with a roughly 30% higher risk of cardiovascular events than that of carotid endarterectomy during 12 years of follow-up in a large, real-world, population-based cohort study, Mohamad A. Hussain, MD, reported at the annual congress of the European Society of Cardiology.
“Our data raise concerns about the external validity of randomized controlled trials of carotid endarterectomy versus stenting and question the potential interchangeability of carotid endarterectomy and stenting as stated in clinical practice guidelines,” said Dr. Hussain of the University of Toronto.
Major practice guidelines cite randomized trial evidence in suggesting that CEA and stenting can be used interchangeably in treating low- or average-risk patients with significant carotid artery disease. Dr. Hussain and his coinvestigators, suspicious that the generalizability of the randomized trial findings may be limited because of operator and institutional selection bias, decided to conduct a retrospective cohort study of all patients over age 40 years who underwent CEA or carotid stenting in the province of Ontario from April 2002 through March 2013.
Using validated chart abstraction software, they identified 12,529 patients who had CEA and 1,935 with carotid stenting. The two groups were similar in terms of most baseline characteristics. Notably, however, stent recipients were significantly more likely to have symptomatic carotid disease and also had more comorbid conditions as reflected in a higher Charlson Comorbidity Index score.
The primary outcome in the study was the 12-year rate of a composite comprising ischemic stroke, transient ischemic attack (TIA), MI, or death. The rate was 35.4% in the CEA group and 44.5% in the stent group. After adjustment for the baseline differences, the stent group still had a statistically significant 28% greater risk of the primary outcome.
“We found the difference remained significant in all of our subgroup analyses, regardless of age, sex, year of procedure, symptomatic or asymptomatic carotid artery disease, CAD [coronary artery disease] or no CAD, diabetes (type 1 or 2) or no diabetes. Outcomes with endarterectomy were always significantly better,” said Dr. Hussain.
“I think our study shows that in clinical practice we’re not quite seeing the outcomes reported in the clinical trials,” he added.
As for the individual components of the composite endpoint, the 12-year rate of ischemic stroke or TIA was 9% in the CEA group and 14% with stenting, for an adjusted 40% increased risk in the stent group. The 12-year all-cause mortality rate was 26% in the CEA group and 34% with stenting, for an adjusted 28% increased risk. The incidence of MI was 8% in both groups.
The investigators next conducted a confirmatory propensity-matched analysis in which 1,927 of the stented patients were closely matched to 3,844 surgical patients, eliminating baseline differences in the prevalence of symptomatic carotid artery disease and other disparities. In this matched cohort, the primary outcome occurred in 37.4% of the CEA group and 44.3% of stent patients, for an adjusted 32% increase in risk in the stented group.
The differences in outcome were driven by sharply higher periprocedural risk in the stented group. After the periprocedural period, the outcome curves remained parallel in the two treatment groups.
In that first 30 days post procedure, the primary composite outcome occurred in 5.4% of the CEA group and 10% of stented patients, for an adjusted 40% increase in relative risk in percutaneously treated patients. The 30-day rate of ischemic stroke or TIA was 3.4% in the surgical group compared with 6.4% in stented patients. Thirty-day mortality was 0.9% with CEA versus 3.3% with stenting.
Asked by the award panel for his thoughts on the disparity between the results of his real-world study and the major randomized trials of CEA versus stenting, Dr. Hussain replied, “It may be because the trials had high-volume operators at high-volume centers who are really experts in carotid stenting, while in the real world many physicians may not be selecting the right people for carotid stenting.”
Differences in sample size may also figure in the disparity, he continued. He noted that in the recent 10-year report from the Carotid Revascularization Endarterectomy versus Stenting Trial (CREST), the composite endpoint of stroke, MI, or death occurred in 9.9% of the CEA group compared with 11.8% of the stenting group (N Engl J Med. 2016 Mar 17;374[11]:1021-31), but this difference in favor of CEA didn’t achieve statistical significance because of the wide confidence intervals resulting from a smaller sample size than in the Ontario study.
Looking to the future, Dr. Hussain said he thinks the ongoing CREST-2 trial is “very important.” It is randomizing patients with asymptomatic high-grade carotid stenosis to uniform intensive medical management either alone or in combination with CEA or stenting with embolic protection.
“That study might end up showing us that medical therapy is as good as or even better than stenting or CEA, especially in asymptomatic patients,” he said.
Dr. Hussain reported having no financial conflicts regarding his academically funded study.
ROME – Carotid stenting was associated with a roughly 30% higher risk of cardiovascular events than that of carotid endarterectomy during 12 years of follow-up in a large, real-world, population-based cohort study, Mohamad A. Hussain, MD, reported at the annual congress of the European Society of Cardiology.
“Our data raise concerns about the external validity of randomized controlled trials of carotid endarterectomy versus stenting and question the potential interchangeability of carotid endarterectomy and stenting as stated in clinical practice guidelines,” said Dr. Hussain of the University of Toronto.
Major practice guidelines cite randomized trial evidence in suggesting that CEA and stenting can be used interchangeably in treating low- or average-risk patients with significant carotid artery disease. Dr. Hussain and his coinvestigators, suspicious that the generalizability of the randomized trial findings may be limited because of operator and institutional selection bias, decided to conduct a retrospective cohort study of all patients over age 40 years who underwent CEA or carotid stenting in the province of Ontario from April 2002 through March 2013.
Using validated chart abstraction software, they identified 12,529 patients who had CEA and 1,935 with carotid stenting. The two groups were similar in terms of most baseline characteristics. Notably, however, stent recipients were significantly more likely to have symptomatic carotid disease and also had more comorbid conditions as reflected in a higher Charlson Comorbidity Index score.
The primary outcome in the study was the 12-year rate of a composite comprising ischemic stroke, transient ischemic attack (TIA), MI, or death. The rate was 35.4% in the CEA group and 44.5% in the stent group. After adjustment for the baseline differences, the stent group still had a statistically significant 28% greater risk of the primary outcome.
“We found the difference remained significant in all of our subgroup analyses, regardless of age, sex, year of procedure, symptomatic or asymptomatic carotid artery disease, CAD [coronary artery disease] or no CAD, diabetes (type 1 or 2) or no diabetes. Outcomes with endarterectomy were always significantly better,” said Dr. Hussain.
“I think our study shows that in clinical practice we’re not quite seeing the outcomes reported in the clinical trials,” he added.
As for the individual components of the composite endpoint, the 12-year rate of ischemic stroke or TIA was 9% in the CEA group and 14% with stenting, for an adjusted 40% increased risk in the stent group. The 12-year all-cause mortality rate was 26% in the CEA group and 34% with stenting, for an adjusted 28% increased risk. The incidence of MI was 8% in both groups.
The investigators next conducted a confirmatory propensity-matched analysis in which 1,927 of the stented patients were closely matched to 3,844 surgical patients, eliminating baseline differences in the prevalence of symptomatic carotid artery disease and other disparities. In this matched cohort, the primary outcome occurred in 37.4% of the CEA group and 44.3% of stent patients, for an adjusted 32% increase in risk in the stented group.
The differences in outcome were driven by sharply higher periprocedural risk in the stented group. After the periprocedural period, the outcome curves remained parallel in the two treatment groups.
In that first 30 days post procedure, the primary composite outcome occurred in 5.4% of the CEA group and 10% of stented patients, for an adjusted 40% increase in relative risk in percutaneously treated patients. The 30-day rate of ischemic stroke or TIA was 3.4% in the surgical group compared with 6.4% in stented patients. Thirty-day mortality was 0.9% with CEA versus 3.3% with stenting.
Asked by the award panel for his thoughts on the disparity between the results of his real-world study and the major randomized trials of CEA versus stenting, Dr. Hussain replied, “It may be because the trials had high-volume operators at high-volume centers who are really experts in carotid stenting, while in the real world many physicians may not be selecting the right people for carotid stenting.”
Differences in sample size may also figure in the disparity, he continued. He noted that in the recent 10-year report from the Carotid Revascularization Endarterectomy versus Stenting Trial (CREST), the composite endpoint of stroke, MI, or death occurred in 9.9% of the CEA group compared with 11.8% of the stenting group (N Engl J Med. 2016 Mar 17;374[11]:1021-31), but this difference in favor of CEA didn’t achieve statistical significance because of the wide confidence intervals resulting from a smaller sample size than in the Ontario study.
Looking to the future, Dr. Hussain said he thinks the ongoing CREST-2 trial is “very important.” It is randomizing patients with asymptomatic high-grade carotid stenosis to uniform intensive medical management either alone or in combination with CEA or stenting with embolic protection.
“That study might end up showing us that medical therapy is as good as or even better than stenting or CEA, especially in asymptomatic patients,” he said.
Dr. Hussain reported having no financial conflicts regarding his academically funded study.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: The composite rate of ischemic stroke, TIA, MI, or death over 12 years of follow-up occurred in 35.4% of patients with carotid artery disease treated by carotid endarterectomy and 44.5% of those who underwent stenting.
Data source: A retrospective population-based cohort study comprising all 14,464 patients who underwent carotid endarterectomy or stenting for carotid artery disease in Ontario during 2002-2013.
Disclosures: The presenter reported having no financial conflicts of interest regarding his academically funded study.
How to reduce early readmissions after bariatric surgery
NEW ORLEANS – A couple of home nursing visits early after bariatric surgery halved emergency department visits and hospital readmissions within the first 30 days in a case-control study, Linden A. Karas, MD, reported at Obesity Week 2016.
“Home nursing visits during the month following surgery, between doctor’s office visits, are a simple and inexpensive intervention that drastically decreases hospital revisits,” she declared at the meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
These early revisits to the hospital following bariatric surgery are not only common, they carry a substantial cost. An ED visit for hydration runs about $3,000 and a 2-day inpatient admission about $18,000. In contrast, the price tag for two home nursing visits in this study was $300, noted Dr. Karas, a surgeon at Mercy Catholic Medical Center in Philadelphia.
The triad of dehydration, nausea, and vomiting in addition to abdominal pain account for up to 40% of early readmissions following bariatric surgery. Because most hospital revisits for these reasons entail very little intervention other than IV fluids, Dr. Karas and her coinvestigators decided to investigate whether preemptive structured home visits by nurses prepared to administer IV fluids on the spot would cut down on hospital revisits. They found that indeed this strategy was successful.
Mercy Catholic Medical Center serves a largely low-income Medicare/Medicaid population. Part of the local culture is for many patients to obtain their primary care in the ED, so bariatric surgery patients are generally not reluctant to show up there if they don’t feel well in the first few weeks post surgery. That’s why in the year prior to the study the 30-day readmission rate was 23.6%, she explained.
The study included 193 consecutive prospectively followed adult participants in the home nursing program who underwent Roux-en-Y gastric bypass, laparoscopic adjustable banding, or revision surgery in 2014 and a control group of 267 matched bariatric surgery patients who underwent the same procedures in 2013, before introduction of the home nursing visits.
The first home visit occurred roughly 3 days after hospital discharge. The second one bridged the period between the first scheduled postoperative office visit at 2 weeks and the next office visit at 4 weeks. At discharge, patients were given a checklist concerning the signs and symptoms of dehydration, dietary recommendations, and lifestyle choices. Patients were encouraged to use the checklist to self-evaluate for dehydration and hand it over to the visiting nurses. The same checklist was provided to the home nurses. If the nurses found that patients met three or more criteria on the dehydration checklist, which included dry mucus membranes, heart rate greater than 90 beats/minute, headache, going more than 8 hours without urination, weakness, abdominal pain, and roughly a dozen other well-known criteria, they were instructed to initiate home hydration therapy with a banana bag and 2 L of normal saline.
If 1-2 days of home hydration failed to resolve the symptoms, the home care nurse was supposed to refer the patient to the ED. If the patient improved in response to home hydration therapy, the home nurse informed the surgeon’s office, which then set up a follow-up office visit to take place within the next 24 hours. Also, if the patient’s responses on the nutrition and behavioral health questionnaire raised any red flags regarding inadequate food and fluid intake, nonadherence to the recommended daily minimum of 30 minutes of physical activity, or depressive symptoms, the nurse was supposed to notify the surgeon to schedule a behavioral or nutrition appointment at the patient’s next scheduled visit.
The 30-day rate of all-cause hospital revisits in the home nursing care group was 11.9%, compared with 23.6% in controls. The rate of hospital revisits specifically for dehydration was 6.2% in the home visit group vs. 8.6% in controls, a 28% relative risk reduction in the home treatment group, which didn’t achieve statistical significance. However, in hindsight, it was evident that some of the visiting nurses didn’t fully understand the treatment protocol; they sent patients straight to the ED without first providing home hydration therapy. The visiting nurses were subcontracted out and weren’t part of the hospital’s bariatric surgery program.
Among those patients who did receive the therapy, however, it prevented readmission in 77% of cases. The 23% of patients who didn’t improve were referred to the hospital for further workup and treatment.
Patients with early hospital readmission had an average of 4.38 comorbid conditions, significantly more than the average 3.46 comorbidities in patients who weren’t readmitted. Several specific comorbid conditions were independently associated with significantly increased risk of hospital readmission: asthma, chronic obstructive pulmonary disease, anxiety, heart failure, and gastroesophageal reflux disease.
“Patients with those comorbidities are the ones to watch aggressively in the postop period,” according to Dr. Karas.
Insurers have paid for the home nursing visits without hesitation.
Her presentation met with an enthusiastic audience reception.
“I think it’s important that you’ve shown a return on investment for this initiative,” one surgeon commented.
Session cochair John J. Kelly, MD, chief of general and minimally invasive surgery at the University of Massachusetts in Worcester, declared, “I think this is a very-important paper. We’re obviously all faced with this dilemma in terms of readmission rates. If you can prevent them, it’s to be commended.”
Dr. Karas reported having no financial conflicts of interest regarding her study.
NEW ORLEANS – A couple of home nursing visits early after bariatric surgery halved emergency department visits and hospital readmissions within the first 30 days in a case-control study, Linden A. Karas, MD, reported at Obesity Week 2016.
“Home nursing visits during the month following surgery, between doctor’s office visits, are a simple and inexpensive intervention that drastically decreases hospital revisits,” she declared at the meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
These early revisits to the hospital following bariatric surgery are not only common, they carry a substantial cost. An ED visit for hydration runs about $3,000 and a 2-day inpatient admission about $18,000. In contrast, the price tag for two home nursing visits in this study was $300, noted Dr. Karas, a surgeon at Mercy Catholic Medical Center in Philadelphia.
The triad of dehydration, nausea, and vomiting in addition to abdominal pain account for up to 40% of early readmissions following bariatric surgery. Because most hospital revisits for these reasons entail very little intervention other than IV fluids, Dr. Karas and her coinvestigators decided to investigate whether preemptive structured home visits by nurses prepared to administer IV fluids on the spot would cut down on hospital revisits. They found that indeed this strategy was successful.
Mercy Catholic Medical Center serves a largely low-income Medicare/Medicaid population. Part of the local culture is for many patients to obtain their primary care in the ED, so bariatric surgery patients are generally not reluctant to show up there if they don’t feel well in the first few weeks post surgery. That’s why in the year prior to the study the 30-day readmission rate was 23.6%, she explained.
The study included 193 consecutive prospectively followed adult participants in the home nursing program who underwent Roux-en-Y gastric bypass, laparoscopic adjustable banding, or revision surgery in 2014 and a control group of 267 matched bariatric surgery patients who underwent the same procedures in 2013, before introduction of the home nursing visits.
The first home visit occurred roughly 3 days after hospital discharge. The second one bridged the period between the first scheduled postoperative office visit at 2 weeks and the next office visit at 4 weeks. At discharge, patients were given a checklist concerning the signs and symptoms of dehydration, dietary recommendations, and lifestyle choices. Patients were encouraged to use the checklist to self-evaluate for dehydration and hand it over to the visiting nurses. The same checklist was provided to the home nurses. If the nurses found that patients met three or more criteria on the dehydration checklist, which included dry mucus membranes, heart rate greater than 90 beats/minute, headache, going more than 8 hours without urination, weakness, abdominal pain, and roughly a dozen other well-known criteria, they were instructed to initiate home hydration therapy with a banana bag and 2 L of normal saline.
If 1-2 days of home hydration failed to resolve the symptoms, the home care nurse was supposed to refer the patient to the ED. If the patient improved in response to home hydration therapy, the home nurse informed the surgeon’s office, which then set up a follow-up office visit to take place within the next 24 hours. Also, if the patient’s responses on the nutrition and behavioral health questionnaire raised any red flags regarding inadequate food and fluid intake, nonadherence to the recommended daily minimum of 30 minutes of physical activity, or depressive symptoms, the nurse was supposed to notify the surgeon to schedule a behavioral or nutrition appointment at the patient’s next scheduled visit.
The 30-day rate of all-cause hospital revisits in the home nursing care group was 11.9%, compared with 23.6% in controls. The rate of hospital revisits specifically for dehydration was 6.2% in the home visit group vs. 8.6% in controls, a 28% relative risk reduction in the home treatment group, which didn’t achieve statistical significance. However, in hindsight, it was evident that some of the visiting nurses didn’t fully understand the treatment protocol; they sent patients straight to the ED without first providing home hydration therapy. The visiting nurses were subcontracted out and weren’t part of the hospital’s bariatric surgery program.
Among those patients who did receive the therapy, however, it prevented readmission in 77% of cases. The 23% of patients who didn’t improve were referred to the hospital for further workup and treatment.
Patients with early hospital readmission had an average of 4.38 comorbid conditions, significantly more than the average 3.46 comorbidities in patients who weren’t readmitted. Several specific comorbid conditions were independently associated with significantly increased risk of hospital readmission: asthma, chronic obstructive pulmonary disease, anxiety, heart failure, and gastroesophageal reflux disease.
“Patients with those comorbidities are the ones to watch aggressively in the postop period,” according to Dr. Karas.
Insurers have paid for the home nursing visits without hesitation.
Her presentation met with an enthusiastic audience reception.
“I think it’s important that you’ve shown a return on investment for this initiative,” one surgeon commented.
Session cochair John J. Kelly, MD, chief of general and minimally invasive surgery at the University of Massachusetts in Worcester, declared, “I think this is a very-important paper. We’re obviously all faced with this dilemma in terms of readmission rates. If you can prevent them, it’s to be commended.”
Dr. Karas reported having no financial conflicts of interest regarding her study.
NEW ORLEANS – A couple of home nursing visits early after bariatric surgery halved emergency department visits and hospital readmissions within the first 30 days in a case-control study, Linden A. Karas, MD, reported at Obesity Week 2016.
“Home nursing visits during the month following surgery, between doctor’s office visits, are a simple and inexpensive intervention that drastically decreases hospital revisits,” she declared at the meeting presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery.
These early revisits to the hospital following bariatric surgery are not only common, they carry a substantial cost. An ED visit for hydration runs about $3,000 and a 2-day inpatient admission about $18,000. In contrast, the price tag for two home nursing visits in this study was $300, noted Dr. Karas, a surgeon at Mercy Catholic Medical Center in Philadelphia.
The triad of dehydration, nausea, and vomiting in addition to abdominal pain account for up to 40% of early readmissions following bariatric surgery. Because most hospital revisits for these reasons entail very little intervention other than IV fluids, Dr. Karas and her coinvestigators decided to investigate whether preemptive structured home visits by nurses prepared to administer IV fluids on the spot would cut down on hospital revisits. They found that indeed this strategy was successful.
Mercy Catholic Medical Center serves a largely low-income Medicare/Medicaid population. Part of the local culture is for many patients to obtain their primary care in the ED, so bariatric surgery patients are generally not reluctant to show up there if they don’t feel well in the first few weeks post surgery. That’s why in the year prior to the study the 30-day readmission rate was 23.6%, she explained.
The study included 193 consecutive prospectively followed adult participants in the home nursing program who underwent Roux-en-Y gastric bypass, laparoscopic adjustable banding, or revision surgery in 2014 and a control group of 267 matched bariatric surgery patients who underwent the same procedures in 2013, before introduction of the home nursing visits.
The first home visit occurred roughly 3 days after hospital discharge. The second one bridged the period between the first scheduled postoperative office visit at 2 weeks and the next office visit at 4 weeks. At discharge, patients were given a checklist concerning the signs and symptoms of dehydration, dietary recommendations, and lifestyle choices. Patients were encouraged to use the checklist to self-evaluate for dehydration and hand it over to the visiting nurses. The same checklist was provided to the home nurses. If the nurses found that patients met three or more criteria on the dehydration checklist, which included dry mucus membranes, heart rate greater than 90 beats/minute, headache, going more than 8 hours without urination, weakness, abdominal pain, and roughly a dozen other well-known criteria, they were instructed to initiate home hydration therapy with a banana bag and 2 L of normal saline.
If 1-2 days of home hydration failed to resolve the symptoms, the home care nurse was supposed to refer the patient to the ED. If the patient improved in response to home hydration therapy, the home nurse informed the surgeon’s office, which then set up a follow-up office visit to take place within the next 24 hours. Also, if the patient’s responses on the nutrition and behavioral health questionnaire raised any red flags regarding inadequate food and fluid intake, nonadherence to the recommended daily minimum of 30 minutes of physical activity, or depressive symptoms, the nurse was supposed to notify the surgeon to schedule a behavioral or nutrition appointment at the patient’s next scheduled visit.
The 30-day rate of all-cause hospital revisits in the home nursing care group was 11.9%, compared with 23.6% in controls. The rate of hospital revisits specifically for dehydration was 6.2% in the home visit group vs. 8.6% in controls, a 28% relative risk reduction in the home treatment group, which didn’t achieve statistical significance. However, in hindsight, it was evident that some of the visiting nurses didn’t fully understand the treatment protocol; they sent patients straight to the ED without first providing home hydration therapy. The visiting nurses were subcontracted out and weren’t part of the hospital’s bariatric surgery program.
Among those patients who did receive the therapy, however, it prevented readmission in 77% of cases. The 23% of patients who didn’t improve were referred to the hospital for further workup and treatment.
Patients with early hospital readmission had an average of 4.38 comorbid conditions, significantly more than the average 3.46 comorbidities in patients who weren’t readmitted. Several specific comorbid conditions were independently associated with significantly increased risk of hospital readmission: asthma, chronic obstructive pulmonary disease, anxiety, heart failure, and gastroesophageal reflux disease.
“Patients with those comorbidities are the ones to watch aggressively in the postop period,” according to Dr. Karas.
Insurers have paid for the home nursing visits without hesitation.
Her presentation met with an enthusiastic audience reception.
“I think it’s important that you’ve shown a return on investment for this initiative,” one surgeon commented.
Session cochair John J. Kelly, MD, chief of general and minimally invasive surgery at the University of Massachusetts in Worcester, declared, “I think this is a very-important paper. We’re obviously all faced with this dilemma in terms of readmission rates. If you can prevent them, it’s to be commended.”
Dr. Karas reported having no financial conflicts of interest regarding her study.
AT OBESITY WEEK 2016
Key clinical point:
Major finding: The combined rate of ED visits and hospital readmissions within 30 days post–bariatric surgery was 11.9% in a group of patients who received two home nursing visits that included home hydration therapy as indicated, compared with 23.6% in bariatric surgery patients who weren’t involved in the home visit program.
Data source: This study compared 30-day hospital readmission rates in 193 consecutive prospectively followed bariatric surgery patients who received two structured home nursing visits in the first 4 postoperative weeks to readmissions in 267 matched controls whose surgery occurred prior to implementation of the home nursing program.
Disclosures: The presenter reported having no financial conflicts of interest regarding the study.
Weight loss procedure is linked to subsequent substance misuse
NEW ORLEANS – Severely obese patients who undergo Roux-en-Y gastric bypass surgery are subsequently at sharply increased risk for new-onset alcohol use disorder as well as for treatment of substance use disorder, compared with others who opt for a laparoscopic adjustable banding procedure for weight loss, Wendy C. King, PhD, reported at a meeting presented by the Obesity Society of America and the American Society for Metabolic and Bariatric Surgery.
This new finding from the NIH-sponsored Longitudinal Assessment of Bariatric Surgery–2 study (LABS-2) has important implications for clinical practice.
“Patients considering bariatric surgery really should be informed of this surgery-specific risk. Also, alcohol use disorder screening, evaluation, intervention, and referral should be incorporated as part of regular presurgical and definitely also postoperative care. And because many patients don’t return to their surgeon for long-term postoperative care, it’s important that clinicians in primary care and other specialties are really looking for this problem in long-term follow-up,” said Dr. King, an epidemiologist at the University of Pittsburgh.
LABS-2 is an observational cohort study of patients undergoing first-time bariatric surgery at 10 participating U.S. hospitals, all of which have academic ties and are rated as bariatric surgery centers of excellence. Dr. King presented 5-year postsurgical follow-up data on 1,481 patients who had Roux-en-Y gastric bypass (RYGB) and 522 with laparoscopic adjustable gastric banding (LAGB). Independently of their regular clinical care visits, participants were assessed annually for their alcohol use and its consequences using the Alcohol Use Disorders Identification Test (AUDIT), use of illicit drugs within the past year, and whether they had undergone hospitalization or counseling for alcohol or drug problems. A score of 8 or more points on the AUDIT was deemed an indication of symptoms of alcohol use disorder (AUD),
After eliminating from consideration the 7% of patients with AUD symptoms at baseline, the cumulative incidence of AUD symptoms in the RYGB patients climbed from zero to 20.8% by the end of the fifth year of follow-up. Treatment for a substance use disorder occurred in 3.5% of RYGB patients during their first 5 years postsurgery, and 7.5% admitted to illicit drug use, said Dr. King.
In contrast, the cumulative incidence of AUD symptoms through 5 years in the LAGB patients was only 11.3%, less than 1% underwent treatment for a substance use disorder, and 4.9% said they had used illicit drugs.
But LABS-2 is not a randomized trial. Patients chose their bariatric procedure together with their surgeon. For this reason, it was important to perform a multivariate regression analysis adjusted for sociodemographics, social support, psychiatric treatment, lifetime history of psychiatric hospitalization, baseline smoking and alcohol consumption, and other potential confounders.
After performing this statistical exercise, the RYGB patients remained at an adjusted 2.05-fold increased risk of AUD symptoms, compared with the LAGB patients, as well as at 3.83-fold greater risk of treatment for a substance use disorder.
The 1.6-fold increased rate of illicit drug use in the RYGB group didn’t achieve statistical significance. Moreover, on closer examination, most of this illicit drug use involved marijuana, and its use in the post–bariatric surgery population appeared to mirror secular trends in the United States as a whole, according to Dr. King.
With her coinvestigators, Dr. King searched for presurgical risk factors that might predict postsurgical substance misuse. Perhaps the most interesting finding concerned the factors that weren’t predictive, including education, unemployment, score on the Beck Depression Inventory, SF-36 mental component summary score, race, marital status, binge eating, loss of control eating, and body mass index.
Lower social support prior to surgery was associated with increased risk for developing AUD symptoms during the first 5 years after bariatric surgery. Younger age and smoking at baseline were associated with increased rates of postoperative AUD symptoms, substance use disorder treatment, and illicit drug use. A history of psychiatric treatment was associated with increased rates of substance use disorder treatment and illicit drug use.
“That could indicate greater medical surveillance among those patients or greater willingness to get treatment, since they’d had treatment for other psychiatric issues in the past,” Dr. King speculated.
She described the study’s strengths as its large size, geographically diverse patient population, unusually high retention over time, compared with other bariatric surgery studies, and the use of AUDIT, a validated and reliable screening tool. The major limitations are that investigators didn’t inquire about illicit use of opioids and benzodiazepines, and recipients of gastric sleeve procedures weren’t included in the long-term follow-up analysis because LABS-2 began before the gastric sleeve boomed in popularity.
John M. Morton, MD, a former president of the American Society for Metabolic and Bariatric Surgery, predicted that a similar study that included gastric sleeve patients would show them to have the same unremarkable postoperative rates of substance misuse as the LAGB group.
“I want to emphasize that this increased incidence of alcohol problems in the Roux-en-Y gastric bypass patients is maybe not so much a psychological issue as it is a physiologic one,” added Dr. Morton, chief of bariatric and minimally invasive surgery at Stanford (Calif.) School of Medicine.
Dr. King agreed. “Just in the last year and a half there have been some great pharmacokinetic studies showing that the Roux-en-Y affects alcohol metabolism and absorption, as well as studies in rodent models that suggest alcohol produces increased neurobiologic reward,” she noted.
The LABS-2 study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. King reported having no relevant financial interests.
NEW ORLEANS – Severely obese patients who undergo Roux-en-Y gastric bypass surgery are subsequently at sharply increased risk for new-onset alcohol use disorder as well as for treatment of substance use disorder, compared with others who opt for a laparoscopic adjustable banding procedure for weight loss, Wendy C. King, PhD, reported at a meeting presented by the Obesity Society of America and the American Society for Metabolic and Bariatric Surgery.
This new finding from the NIH-sponsored Longitudinal Assessment of Bariatric Surgery–2 study (LABS-2) has important implications for clinical practice.
“Patients considering bariatric surgery really should be informed of this surgery-specific risk. Also, alcohol use disorder screening, evaluation, intervention, and referral should be incorporated as part of regular presurgical and definitely also postoperative care. And because many patients don’t return to their surgeon for long-term postoperative care, it’s important that clinicians in primary care and other specialties are really looking for this problem in long-term follow-up,” said Dr. King, an epidemiologist at the University of Pittsburgh.
LABS-2 is an observational cohort study of patients undergoing first-time bariatric surgery at 10 participating U.S. hospitals, all of which have academic ties and are rated as bariatric surgery centers of excellence. Dr. King presented 5-year postsurgical follow-up data on 1,481 patients who had Roux-en-Y gastric bypass (RYGB) and 522 with laparoscopic adjustable gastric banding (LAGB). Independently of their regular clinical care visits, participants were assessed annually for their alcohol use and its consequences using the Alcohol Use Disorders Identification Test (AUDIT), use of illicit drugs within the past year, and whether they had undergone hospitalization or counseling for alcohol or drug problems. A score of 8 or more points on the AUDIT was deemed an indication of symptoms of alcohol use disorder (AUD),
After eliminating from consideration the 7% of patients with AUD symptoms at baseline, the cumulative incidence of AUD symptoms in the RYGB patients climbed from zero to 20.8% by the end of the fifth year of follow-up. Treatment for a substance use disorder occurred in 3.5% of RYGB patients during their first 5 years postsurgery, and 7.5% admitted to illicit drug use, said Dr. King.
In contrast, the cumulative incidence of AUD symptoms through 5 years in the LAGB patients was only 11.3%, less than 1% underwent treatment for a substance use disorder, and 4.9% said they had used illicit drugs.
But LABS-2 is not a randomized trial. Patients chose their bariatric procedure together with their surgeon. For this reason, it was important to perform a multivariate regression analysis adjusted for sociodemographics, social support, psychiatric treatment, lifetime history of psychiatric hospitalization, baseline smoking and alcohol consumption, and other potential confounders.
After performing this statistical exercise, the RYGB patients remained at an adjusted 2.05-fold increased risk of AUD symptoms, compared with the LAGB patients, as well as at 3.83-fold greater risk of treatment for a substance use disorder.
The 1.6-fold increased rate of illicit drug use in the RYGB group didn’t achieve statistical significance. Moreover, on closer examination, most of this illicit drug use involved marijuana, and its use in the post–bariatric surgery population appeared to mirror secular trends in the United States as a whole, according to Dr. King.
With her coinvestigators, Dr. King searched for presurgical risk factors that might predict postsurgical substance misuse. Perhaps the most interesting finding concerned the factors that weren’t predictive, including education, unemployment, score on the Beck Depression Inventory, SF-36 mental component summary score, race, marital status, binge eating, loss of control eating, and body mass index.
Lower social support prior to surgery was associated with increased risk for developing AUD symptoms during the first 5 years after bariatric surgery. Younger age and smoking at baseline were associated with increased rates of postoperative AUD symptoms, substance use disorder treatment, and illicit drug use. A history of psychiatric treatment was associated with increased rates of substance use disorder treatment and illicit drug use.
“That could indicate greater medical surveillance among those patients or greater willingness to get treatment, since they’d had treatment for other psychiatric issues in the past,” Dr. King speculated.
She described the study’s strengths as its large size, geographically diverse patient population, unusually high retention over time, compared with other bariatric surgery studies, and the use of AUDIT, a validated and reliable screening tool. The major limitations are that investigators didn’t inquire about illicit use of opioids and benzodiazepines, and recipients of gastric sleeve procedures weren’t included in the long-term follow-up analysis because LABS-2 began before the gastric sleeve boomed in popularity.
John M. Morton, MD, a former president of the American Society for Metabolic and Bariatric Surgery, predicted that a similar study that included gastric sleeve patients would show them to have the same unremarkable postoperative rates of substance misuse as the LAGB group.
“I want to emphasize that this increased incidence of alcohol problems in the Roux-en-Y gastric bypass patients is maybe not so much a psychological issue as it is a physiologic one,” added Dr. Morton, chief of bariatric and minimally invasive surgery at Stanford (Calif.) School of Medicine.
Dr. King agreed. “Just in the last year and a half there have been some great pharmacokinetic studies showing that the Roux-en-Y affects alcohol metabolism and absorption, as well as studies in rodent models that suggest alcohol produces increased neurobiologic reward,” she noted.
The LABS-2 study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. King reported having no relevant financial interests.
NEW ORLEANS – Severely obese patients who undergo Roux-en-Y gastric bypass surgery are subsequently at sharply increased risk for new-onset alcohol use disorder as well as for treatment of substance use disorder, compared with others who opt for a laparoscopic adjustable banding procedure for weight loss, Wendy C. King, PhD, reported at a meeting presented by the Obesity Society of America and the American Society for Metabolic and Bariatric Surgery.
This new finding from the NIH-sponsored Longitudinal Assessment of Bariatric Surgery–2 study (LABS-2) has important implications for clinical practice.
“Patients considering bariatric surgery really should be informed of this surgery-specific risk. Also, alcohol use disorder screening, evaluation, intervention, and referral should be incorporated as part of regular presurgical and definitely also postoperative care. And because many patients don’t return to their surgeon for long-term postoperative care, it’s important that clinicians in primary care and other specialties are really looking for this problem in long-term follow-up,” said Dr. King, an epidemiologist at the University of Pittsburgh.
LABS-2 is an observational cohort study of patients undergoing first-time bariatric surgery at 10 participating U.S. hospitals, all of which have academic ties and are rated as bariatric surgery centers of excellence. Dr. King presented 5-year postsurgical follow-up data on 1,481 patients who had Roux-en-Y gastric bypass (RYGB) and 522 with laparoscopic adjustable gastric banding (LAGB). Independently of their regular clinical care visits, participants were assessed annually for their alcohol use and its consequences using the Alcohol Use Disorders Identification Test (AUDIT), use of illicit drugs within the past year, and whether they had undergone hospitalization or counseling for alcohol or drug problems. A score of 8 or more points on the AUDIT was deemed an indication of symptoms of alcohol use disorder (AUD),
After eliminating from consideration the 7% of patients with AUD symptoms at baseline, the cumulative incidence of AUD symptoms in the RYGB patients climbed from zero to 20.8% by the end of the fifth year of follow-up. Treatment for a substance use disorder occurred in 3.5% of RYGB patients during their first 5 years postsurgery, and 7.5% admitted to illicit drug use, said Dr. King.
In contrast, the cumulative incidence of AUD symptoms through 5 years in the LAGB patients was only 11.3%, less than 1% underwent treatment for a substance use disorder, and 4.9% said they had used illicit drugs.
But LABS-2 is not a randomized trial. Patients chose their bariatric procedure together with their surgeon. For this reason, it was important to perform a multivariate regression analysis adjusted for sociodemographics, social support, psychiatric treatment, lifetime history of psychiatric hospitalization, baseline smoking and alcohol consumption, and other potential confounders.
After performing this statistical exercise, the RYGB patients remained at an adjusted 2.05-fold increased risk of AUD symptoms, compared with the LAGB patients, as well as at 3.83-fold greater risk of treatment for a substance use disorder.
The 1.6-fold increased rate of illicit drug use in the RYGB group didn’t achieve statistical significance. Moreover, on closer examination, most of this illicit drug use involved marijuana, and its use in the post–bariatric surgery population appeared to mirror secular trends in the United States as a whole, according to Dr. King.
With her coinvestigators, Dr. King searched for presurgical risk factors that might predict postsurgical substance misuse. Perhaps the most interesting finding concerned the factors that weren’t predictive, including education, unemployment, score on the Beck Depression Inventory, SF-36 mental component summary score, race, marital status, binge eating, loss of control eating, and body mass index.
Lower social support prior to surgery was associated with increased risk for developing AUD symptoms during the first 5 years after bariatric surgery. Younger age and smoking at baseline were associated with increased rates of postoperative AUD symptoms, substance use disorder treatment, and illicit drug use. A history of psychiatric treatment was associated with increased rates of substance use disorder treatment and illicit drug use.
“That could indicate greater medical surveillance among those patients or greater willingness to get treatment, since they’d had treatment for other psychiatric issues in the past,” Dr. King speculated.
She described the study’s strengths as its large size, geographically diverse patient population, unusually high retention over time, compared with other bariatric surgery studies, and the use of AUDIT, a validated and reliable screening tool. The major limitations are that investigators didn’t inquire about illicit use of opioids and benzodiazepines, and recipients of gastric sleeve procedures weren’t included in the long-term follow-up analysis because LABS-2 began before the gastric sleeve boomed in popularity.
John M. Morton, MD, a former president of the American Society for Metabolic and Bariatric Surgery, predicted that a similar study that included gastric sleeve patients would show them to have the same unremarkable postoperative rates of substance misuse as the LAGB group.
“I want to emphasize that this increased incidence of alcohol problems in the Roux-en-Y gastric bypass patients is maybe not so much a psychological issue as it is a physiologic one,” added Dr. Morton, chief of bariatric and minimally invasive surgery at Stanford (Calif.) School of Medicine.
Dr. King agreed. “Just in the last year and a half there have been some great pharmacokinetic studies showing that the Roux-en-Y affects alcohol metabolism and absorption, as well as studies in rodent models that suggest alcohol produces increased neurobiologic reward,” she noted.
The LABS-2 study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. King reported having no relevant financial interests.
AT OBESITY WEEK 2016
Key clinical point:
Major finding: In the first 5 years following bariatric surgery, patients who underwent Roux-en-Y gastric bypass were twice as likely to develop new-onset alcohol use disorder and nearly four times more likely to be treated for substance use disorder, compared with recipients of laparoscopic gastric banding.
Data source: The LABS-2 study is an observational cohort study involving more than 2,000 patients in long-term follow-up after undergoing Roux-en-Y gastric bypass or laparoscopic adjustable banding.
Disclosures: LABS-2 is funded by the National Institute of Diabetes and Digestive and Kidney Diseases. The presenter reported having no relevant financial interests.
Rheumatoid arthritis increases heart failure risk
ROME – Rheumatoid arthritis is associated with a significantly increased risk of hospitalization for heart failure, according to a nationwide Danish study, Usman Khalid, MD, reported at the annual congress of the European Society of Cardiology.
As a chronic systemic inflammatory disease, rheumatoid arthritis (RA) has been associated with increased risk of a variety of comorbid conditions, including cardiovascular disease. But RA’s relationship specifically with heart failure hasn’t previously been looked at in the comprehensive way that’s possible in Denmark, where linked national registries enable researchers to follow health issues in the entire population from birth to death, noted Dr. Khalid of the University of Copenhagen.
In an analysis adjusted for age, sex, and calendar year, individuals with RA were 83% more likely to be hospitalized for heart failure than were the non-RA Danish population. In a fully adjusted analysis that controlled for those potential confounders as well as comorbid conditions, smoking, alcohol intake, socioeconomic status, and prescription medications, patients with RA remained at a statistically significant and clinically meaningful 38% increased risk of heart failure hospitalization.
Further studies are planned to determine the underlying mechanisms of this association, Dr. Khalid added.
He reported having no financial conflicts of interest regarding this study, which was supported by an unrestricted grant from Leo Pharma.
ROME – Rheumatoid arthritis is associated with a significantly increased risk of hospitalization for heart failure, according to a nationwide Danish study, Usman Khalid, MD, reported at the annual congress of the European Society of Cardiology.
As a chronic systemic inflammatory disease, rheumatoid arthritis (RA) has been associated with increased risk of a variety of comorbid conditions, including cardiovascular disease. But RA’s relationship specifically with heart failure hasn’t previously been looked at in the comprehensive way that’s possible in Denmark, where linked national registries enable researchers to follow health issues in the entire population from birth to death, noted Dr. Khalid of the University of Copenhagen.
In an analysis adjusted for age, sex, and calendar year, individuals with RA were 83% more likely to be hospitalized for heart failure than were the non-RA Danish population. In a fully adjusted analysis that controlled for those potential confounders as well as comorbid conditions, smoking, alcohol intake, socioeconomic status, and prescription medications, patients with RA remained at a statistically significant and clinically meaningful 38% increased risk of heart failure hospitalization.
Further studies are planned to determine the underlying mechanisms of this association, Dr. Khalid added.
He reported having no financial conflicts of interest regarding this study, which was supported by an unrestricted grant from Leo Pharma.
ROME – Rheumatoid arthritis is associated with a significantly increased risk of hospitalization for heart failure, according to a nationwide Danish study, Usman Khalid, MD, reported at the annual congress of the European Society of Cardiology.
As a chronic systemic inflammatory disease, rheumatoid arthritis (RA) has been associated with increased risk of a variety of comorbid conditions, including cardiovascular disease. But RA’s relationship specifically with heart failure hasn’t previously been looked at in the comprehensive way that’s possible in Denmark, where linked national registries enable researchers to follow health issues in the entire population from birth to death, noted Dr. Khalid of the University of Copenhagen.
In an analysis adjusted for age, sex, and calendar year, individuals with RA were 83% more likely to be hospitalized for heart failure than were the non-RA Danish population. In a fully adjusted analysis that controlled for those potential confounders as well as comorbid conditions, smoking, alcohol intake, socioeconomic status, and prescription medications, patients with RA remained at a statistically significant and clinically meaningful 38% increased risk of heart failure hospitalization.
Further studies are planned to determine the underlying mechanisms of this association, Dr. Khalid added.
He reported having no financial conflicts of interest regarding this study, which was supported by an unrestricted grant from Leo Pharma.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: The incidence rate for heart failure hospitalization was 7.37 per 1,000 person-years in Danish adults with rheumatoid arthritis, compared with 2.45 per 1,000 in the general population.
Data source: This study utilized Danish comprehensive national registries to determine the rate at which 13,800 Danes with rheumatoid arthritis and no baseline history of heart failure were hospitalized for heart failure during 15 years of follow-up.
Disclosures: The study was supported by an unrestricted grant from Leo Pharma. The presenter reported having no financial conflicts of interest.
Topical crisaborole boosts quality of life in atopic dermatitis
VIENNA – Topical crisaborole 2% ointment administered twice a day was consistently associated with clinically meaningful quality of life improvement scores on multiple measures in the two pivotal phase III, randomized, controlled trials of atopic dermatitis (AD) patients aged 2 years old through adulthood, Amy S. Paller, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
AD in children and adolescents is infamous for the adverse quality of life impact it imposes upon the patients’ parents, family, and caregivers. So the significant improvement seen with crisaborole, compared with its vehicle on the Dermatitis Family Impact (DFI) Questionnaire was particularly gratifying. The DFI questionnaire assesses quality of life in key domains, including family, parent, and caregiver sleep, emotional distress, relationships, family leisure, and ability to do housework or go shopping.
“If approved, crisaborole ... could improve the quality of life for patients with mild to moderate atopic dermatitis and, very importantly, for their families as well,” declared Dr. Paller, professor of dermatology and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Crisaborole’s developer, Anacor Pharmaceuticals, has filed an application for approval for treating mild to moderate AD in patients aged 2 years and older, now under review at the Food and Drug Administration.
In a separate presentation at the EADV congress, Lawrence F. Eichenfield, MD, presented the results of a long-term, open-label crisaborole safety study of 48-52 weeks duration. The long-term study involved 517 participants in the two pivotal phase III trials. There were no serious adverse events and no long-term cutaneous adverse events such as the skin atrophy or telangiectasias that can occur with topical steroids. The safety profile was favorable for long-term treatment of patients 2 years of age or older with mild to moderate AD.
Crisaborole 2% topical ointment is a novel, boron-based, nonsteroidal inhibitor of phosphodiesterase 4 (PDE-4). AD is marked by overactivity of PDE-4, which results in decreased levels of cyclic AMP and resultant increased release of inflammatory cytokines.
Dr. Paller noted that, in the previously reported efficacy results of the two pivotal, double-blind, 28-day, phase III trials, crisaborole treatment reduced global disease severity and provided early and sustained improvement in itch severity. She presented the prespecified quality of life results for the two identically designed, parallel pivotal trials, which totaled 1,016 patients on crisaborole and 506 on its vehicle. At baseline, 39% of subjects had mild AD, and 61% had moderate AD. The mean body surface area affected was 18%. Participants’ mean age was 12.3 years, and 14% were aged 8 years old or older.
The structure of the long-term safety study suggests how crisaborole might be used in clinical practice. During the year-long, open-label study, patients were evaluated every 28 days. If their skin was deemed clear or almost clear on the basis of an Investigator’s Static Global Assessment (ISGA) score of 0 or 1, they were taken off crisaborole and could use only emollients for the next 28 days, at which time they would be reevaluated. At that point, if they had an ISGA of 2 or more, they went back on crisaborole twice a day for 28 days until their next evaluation.
Dr. Eichenfield reported that 10.2% of participants in the long-term safety study reported treatment-related adverse events, which were mild to moderate. The most frequently reported of these were mild to moderate flares of AD during a 28-day off-treatment period in 1.3% of patients, application site pain in 2.3%, and application site infection in 1.2%.
Dr. Paller and Dr. Eichenfield reported serving as consultants to Anacor Pharmaceuticals.
The FDA review of crisaborole is expected to be completed by early January 2017, according to Anacor.
VIENNA – Topical crisaborole 2% ointment administered twice a day was consistently associated with clinically meaningful quality of life improvement scores on multiple measures in the two pivotal phase III, randomized, controlled trials of atopic dermatitis (AD) patients aged 2 years old through adulthood, Amy S. Paller, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
AD in children and adolescents is infamous for the adverse quality of life impact it imposes upon the patients’ parents, family, and caregivers. So the significant improvement seen with crisaborole, compared with its vehicle on the Dermatitis Family Impact (DFI) Questionnaire was particularly gratifying. The DFI questionnaire assesses quality of life in key domains, including family, parent, and caregiver sleep, emotional distress, relationships, family leisure, and ability to do housework or go shopping.
“If approved, crisaborole ... could improve the quality of life for patients with mild to moderate atopic dermatitis and, very importantly, for their families as well,” declared Dr. Paller, professor of dermatology and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Crisaborole’s developer, Anacor Pharmaceuticals, has filed an application for approval for treating mild to moderate AD in patients aged 2 years and older, now under review at the Food and Drug Administration.
In a separate presentation at the EADV congress, Lawrence F. Eichenfield, MD, presented the results of a long-term, open-label crisaborole safety study of 48-52 weeks duration. The long-term study involved 517 participants in the two pivotal phase III trials. There were no serious adverse events and no long-term cutaneous adverse events such as the skin atrophy or telangiectasias that can occur with topical steroids. The safety profile was favorable for long-term treatment of patients 2 years of age or older with mild to moderate AD.
Crisaborole 2% topical ointment is a novel, boron-based, nonsteroidal inhibitor of phosphodiesterase 4 (PDE-4). AD is marked by overactivity of PDE-4, which results in decreased levels of cyclic AMP and resultant increased release of inflammatory cytokines.
Dr. Paller noted that, in the previously reported efficacy results of the two pivotal, double-blind, 28-day, phase III trials, crisaborole treatment reduced global disease severity and provided early and sustained improvement in itch severity. She presented the prespecified quality of life results for the two identically designed, parallel pivotal trials, which totaled 1,016 patients on crisaborole and 506 on its vehicle. At baseline, 39% of subjects had mild AD, and 61% had moderate AD. The mean body surface area affected was 18%. Participants’ mean age was 12.3 years, and 14% were aged 8 years old or older.
The structure of the long-term safety study suggests how crisaborole might be used in clinical practice. During the year-long, open-label study, patients were evaluated every 28 days. If their skin was deemed clear or almost clear on the basis of an Investigator’s Static Global Assessment (ISGA) score of 0 or 1, they were taken off crisaborole and could use only emollients for the next 28 days, at which time they would be reevaluated. At that point, if they had an ISGA of 2 or more, they went back on crisaborole twice a day for 28 days until their next evaluation.
Dr. Eichenfield reported that 10.2% of participants in the long-term safety study reported treatment-related adverse events, which were mild to moderate. The most frequently reported of these were mild to moderate flares of AD during a 28-day off-treatment period in 1.3% of patients, application site pain in 2.3%, and application site infection in 1.2%.
Dr. Paller and Dr. Eichenfield reported serving as consultants to Anacor Pharmaceuticals.
The FDA review of crisaborole is expected to be completed by early January 2017, according to Anacor.
VIENNA – Topical crisaborole 2% ointment administered twice a day was consistently associated with clinically meaningful quality of life improvement scores on multiple measures in the two pivotal phase III, randomized, controlled trials of atopic dermatitis (AD) patients aged 2 years old through adulthood, Amy S. Paller, MD, reported at the annual congress of the European Academy of Dermatology and Venereology.
AD in children and adolescents is infamous for the adverse quality of life impact it imposes upon the patients’ parents, family, and caregivers. So the significant improvement seen with crisaborole, compared with its vehicle on the Dermatitis Family Impact (DFI) Questionnaire was particularly gratifying. The DFI questionnaire assesses quality of life in key domains, including family, parent, and caregiver sleep, emotional distress, relationships, family leisure, and ability to do housework or go shopping.
“If approved, crisaborole ... could improve the quality of life for patients with mild to moderate atopic dermatitis and, very importantly, for their families as well,” declared Dr. Paller, professor of dermatology and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Crisaborole’s developer, Anacor Pharmaceuticals, has filed an application for approval for treating mild to moderate AD in patients aged 2 years and older, now under review at the Food and Drug Administration.
In a separate presentation at the EADV congress, Lawrence F. Eichenfield, MD, presented the results of a long-term, open-label crisaborole safety study of 48-52 weeks duration. The long-term study involved 517 participants in the two pivotal phase III trials. There were no serious adverse events and no long-term cutaneous adverse events such as the skin atrophy or telangiectasias that can occur with topical steroids. The safety profile was favorable for long-term treatment of patients 2 years of age or older with mild to moderate AD.
Crisaborole 2% topical ointment is a novel, boron-based, nonsteroidal inhibitor of phosphodiesterase 4 (PDE-4). AD is marked by overactivity of PDE-4, which results in decreased levels of cyclic AMP and resultant increased release of inflammatory cytokines.
Dr. Paller noted that, in the previously reported efficacy results of the two pivotal, double-blind, 28-day, phase III trials, crisaborole treatment reduced global disease severity and provided early and sustained improvement in itch severity. She presented the prespecified quality of life results for the two identically designed, parallel pivotal trials, which totaled 1,016 patients on crisaborole and 506 on its vehicle. At baseline, 39% of subjects had mild AD, and 61% had moderate AD. The mean body surface area affected was 18%. Participants’ mean age was 12.3 years, and 14% were aged 8 years old or older.
The structure of the long-term safety study suggests how crisaborole might be used in clinical practice. During the year-long, open-label study, patients were evaluated every 28 days. If their skin was deemed clear or almost clear on the basis of an Investigator’s Static Global Assessment (ISGA) score of 0 or 1, they were taken off crisaborole and could use only emollients for the next 28 days, at which time they would be reevaluated. At that point, if they had an ISGA of 2 or more, they went back on crisaborole twice a day for 28 days until their next evaluation.
Dr. Eichenfield reported that 10.2% of participants in the long-term safety study reported treatment-related adverse events, which were mild to moderate. The most frequently reported of these were mild to moderate flares of AD during a 28-day off-treatment period in 1.3% of patients, application site pain in 2.3%, and application site infection in 1.2%.
Dr. Paller and Dr. Eichenfield reported serving as consultants to Anacor Pharmaceuticals.
The FDA review of crisaborole is expected to be completed by early January 2017, according to Anacor.
EXPERT ANALYSIS FROM THE EADV CONGRESS
An enlightened approach to weight loss using liraglutide
NEW ORLEANS – Early weight loss on liraglutide – specifically, dropping at least 4% of body weight at 16 weeks – is a strong and clinically useful identifier of patients with a high likelihood of significant weight loss 13 months into treatment, with an accompanying improvement in cardiometabolic risk factors, according to Ken Fujioka, MD.
Conversely, patients who aren’t early responders to subcutaneous liraglutide at 3 mg/day are unlikely to achieve at least 5% weight loss after a full year on the drug, the regulatory benchmark for clinically meaningful weight loss, added Dr. Fujioka, an internist and director of the center for weight management at the Scripps Clinic in La Jolla, Calif.
“Using this early response criterion at week 16 to predict long-term weight loss is to me a very valuable tool. Obesity is an odd disease because it has so many different causes. Finding the right drug is tough, and how long to keep trying with a particular medication is something we haven’t known. So I think the biggest change in obesity medicine is the creation of stopping rules that allow you to say, ‘OK, maybe this isn’t going to work. There’s some other reason you’re gaining weight, so let’s move on to something else,’” Dr. Fujioka said at the meeting presented by the Obesity Society of America and the American Society for Metabolic and Bariatric Surgery.
“When you stop the medication, you improve the risk-benefit ratio by removing all risk. That’s a win-win to me, and I applaud the FDA for getting on the pharmaceutical companies to make sure they put stopping rules in their medication labels,” he added.
He presented a post hoc pooled analysis of two previously published large, double-blind phase III clinical trials of subcutaneous liraglutide at 3 mg/day (Saxenda) or placebo in combination with a diet and exercise intervention for weight loss: the 3,731-patient SCALE Obesity and Prediabetes trial and the 846-patient SCALE Diabetes trial. In both trials liraglutide was started at a dose of 0.6 mg and titrated to 3.0 mg by week 4. The lifestyle intervention entailed a 500-kcal/day deficit diet and a minimum of 150 minutes of physical activity per week.
The purpose of the pooled analysis was to identify the best early predictor of response status at 56 weeks by examining the impact of 3%, 4%, and 5% weight loss after 8, 12, and 16 weeks of treatment as cut points. This post hoc analysis was prespecified at the request of the Food and Drug Administration before the trials were completed.
The bottom line: The best predictor of long-term outcome on liraglutide, a glucagonlike peptide–1 analog, proved to be a weight loss of 4% or greater at 16 weeks. It had an 81% positive predictive value and a 76% negative predictive value for at least a 5% weight loss at 56 weeks. It correctly predicted weight outcomes at 56 weeks in 80.1% of patients, the highest success rate of all the combinations studied. This finding was the impetus for the current product labeling, which contains the stopping rule. Dr. Fujioka shared study data not included in the labeling; namely, the marked contrast in how early responders and early nonresponders fared at 56 weeks.
The mean weight loss at 56 weeks in nondiabetic early responders to liraglutide was 10.8%, compared with only 3% in early nondiabetic nonresponders. Diabetic early responders averaged an 8.5% weight loss at 56 weeks, while early nonresponders had a mean 3.1% weight loss.
In the SCALE Obesity and Prediabetes trial, 50% of early responders to liraglutide ended up with a greater than 10% weight loss at 56 weeks, and 21% had more than 15% weight loss, compared with rates of 6% and 2%, respectively, in early nonresponders.
In the SCALE Diabetes study, 38% of early responders had greater than 10% weight loss long term, a rate nearly fourfold higher than in early nonresponders. Moreover, 10% of early responder diabetic patients had greater than 15% weight loss, versus a mere 2% of early nonresponders, the internist continued.
The ratio of early responders to early nonresponders in the nondiabetic population was 77%:23%. In diabetic patients, it was 63%:37%.
Turning to cardiometabolic endpoints, Dr. Fujioka noted that early responders in the SCALE Obesity and Prediabetes trial went on to show a mean reduction in systolic blood pressure of 5.1 mm Hg at 56 weeks, compared with a 2–mm Hg decrease in early nonresponders. Early responders also averaged a 10.5-cm shrinkage in waist circumference from a baseline of 115 cm, which was more than twice that observed at 56 weeks in early nonresponders. HDL-cholesterol level rose by 3.9% in early responders but remained unchanged over time in early nonresponders.
Diabetic patients who were early responders to liraglutide 3.0 mg/day had a mean 44.2-mg/dL reduction in fasting plasma glucose at 56 weeks from a baseline of 158 mg/dL, compared with a 30.1-mg/dL decrease in early nonresponders.
“The drop in fasting blood glucose is very quick – within a matter of weeks – so if you already have diabetic patients on drugs that are going to bring their blood sugar down, you may have to back titrate those other drugs really quickly. You don’t want to make your patients hypoglycemic,” the physician said.
Mean hemoglobin A1c values in early responder diabetic patients fell by 1.6% from a baseline of 7.9%, a full 0.5% greater reduction than in early nonresponders.
By far the most frequent adverse events in the two SCALE trials were gastrointestinal, with nausea leading the way. Rates were modestly higher in the early responders.
This analysis and the clinical trials on which it was based were sponsored by Novo Nordisk, which markets liraglutide under the brand names Saxenda and Voctoza. The presenter reported receiving research grants from and serving as a consultant to Novo Nordisk and other pharmaceutical companies.
NEW ORLEANS – Early weight loss on liraglutide – specifically, dropping at least 4% of body weight at 16 weeks – is a strong and clinically useful identifier of patients with a high likelihood of significant weight loss 13 months into treatment, with an accompanying improvement in cardiometabolic risk factors, according to Ken Fujioka, MD.
Conversely, patients who aren’t early responders to subcutaneous liraglutide at 3 mg/day are unlikely to achieve at least 5% weight loss after a full year on the drug, the regulatory benchmark for clinically meaningful weight loss, added Dr. Fujioka, an internist and director of the center for weight management at the Scripps Clinic in La Jolla, Calif.
“Using this early response criterion at week 16 to predict long-term weight loss is to me a very valuable tool. Obesity is an odd disease because it has so many different causes. Finding the right drug is tough, and how long to keep trying with a particular medication is something we haven’t known. So I think the biggest change in obesity medicine is the creation of stopping rules that allow you to say, ‘OK, maybe this isn’t going to work. There’s some other reason you’re gaining weight, so let’s move on to something else,’” Dr. Fujioka said at the meeting presented by the Obesity Society of America and the American Society for Metabolic and Bariatric Surgery.
“When you stop the medication, you improve the risk-benefit ratio by removing all risk. That’s a win-win to me, and I applaud the FDA for getting on the pharmaceutical companies to make sure they put stopping rules in their medication labels,” he added.
He presented a post hoc pooled analysis of two previously published large, double-blind phase III clinical trials of subcutaneous liraglutide at 3 mg/day (Saxenda) or placebo in combination with a diet and exercise intervention for weight loss: the 3,731-patient SCALE Obesity and Prediabetes trial and the 846-patient SCALE Diabetes trial. In both trials liraglutide was started at a dose of 0.6 mg and titrated to 3.0 mg by week 4. The lifestyle intervention entailed a 500-kcal/day deficit diet and a minimum of 150 minutes of physical activity per week.
The purpose of the pooled analysis was to identify the best early predictor of response status at 56 weeks by examining the impact of 3%, 4%, and 5% weight loss after 8, 12, and 16 weeks of treatment as cut points. This post hoc analysis was prespecified at the request of the Food and Drug Administration before the trials were completed.
The bottom line: The best predictor of long-term outcome on liraglutide, a glucagonlike peptide–1 analog, proved to be a weight loss of 4% or greater at 16 weeks. It had an 81% positive predictive value and a 76% negative predictive value for at least a 5% weight loss at 56 weeks. It correctly predicted weight outcomes at 56 weeks in 80.1% of patients, the highest success rate of all the combinations studied. This finding was the impetus for the current product labeling, which contains the stopping rule. Dr. Fujioka shared study data not included in the labeling; namely, the marked contrast in how early responders and early nonresponders fared at 56 weeks.
The mean weight loss at 56 weeks in nondiabetic early responders to liraglutide was 10.8%, compared with only 3% in early nondiabetic nonresponders. Diabetic early responders averaged an 8.5% weight loss at 56 weeks, while early nonresponders had a mean 3.1% weight loss.
In the SCALE Obesity and Prediabetes trial, 50% of early responders to liraglutide ended up with a greater than 10% weight loss at 56 weeks, and 21% had more than 15% weight loss, compared with rates of 6% and 2%, respectively, in early nonresponders.
In the SCALE Diabetes study, 38% of early responders had greater than 10% weight loss long term, a rate nearly fourfold higher than in early nonresponders. Moreover, 10% of early responder diabetic patients had greater than 15% weight loss, versus a mere 2% of early nonresponders, the internist continued.
The ratio of early responders to early nonresponders in the nondiabetic population was 77%:23%. In diabetic patients, it was 63%:37%.
Turning to cardiometabolic endpoints, Dr. Fujioka noted that early responders in the SCALE Obesity and Prediabetes trial went on to show a mean reduction in systolic blood pressure of 5.1 mm Hg at 56 weeks, compared with a 2–mm Hg decrease in early nonresponders. Early responders also averaged a 10.5-cm shrinkage in waist circumference from a baseline of 115 cm, which was more than twice that observed at 56 weeks in early nonresponders. HDL-cholesterol level rose by 3.9% in early responders but remained unchanged over time in early nonresponders.
Diabetic patients who were early responders to liraglutide 3.0 mg/day had a mean 44.2-mg/dL reduction in fasting plasma glucose at 56 weeks from a baseline of 158 mg/dL, compared with a 30.1-mg/dL decrease in early nonresponders.
“The drop in fasting blood glucose is very quick – within a matter of weeks – so if you already have diabetic patients on drugs that are going to bring their blood sugar down, you may have to back titrate those other drugs really quickly. You don’t want to make your patients hypoglycemic,” the physician said.
Mean hemoglobin A1c values in early responder diabetic patients fell by 1.6% from a baseline of 7.9%, a full 0.5% greater reduction than in early nonresponders.
By far the most frequent adverse events in the two SCALE trials were gastrointestinal, with nausea leading the way. Rates were modestly higher in the early responders.
This analysis and the clinical trials on which it was based were sponsored by Novo Nordisk, which markets liraglutide under the brand names Saxenda and Voctoza. The presenter reported receiving research grants from and serving as a consultant to Novo Nordisk and other pharmaceutical companies.
NEW ORLEANS – Early weight loss on liraglutide – specifically, dropping at least 4% of body weight at 16 weeks – is a strong and clinically useful identifier of patients with a high likelihood of significant weight loss 13 months into treatment, with an accompanying improvement in cardiometabolic risk factors, according to Ken Fujioka, MD.
Conversely, patients who aren’t early responders to subcutaneous liraglutide at 3 mg/day are unlikely to achieve at least 5% weight loss after a full year on the drug, the regulatory benchmark for clinically meaningful weight loss, added Dr. Fujioka, an internist and director of the center for weight management at the Scripps Clinic in La Jolla, Calif.
“Using this early response criterion at week 16 to predict long-term weight loss is to me a very valuable tool. Obesity is an odd disease because it has so many different causes. Finding the right drug is tough, and how long to keep trying with a particular medication is something we haven’t known. So I think the biggest change in obesity medicine is the creation of stopping rules that allow you to say, ‘OK, maybe this isn’t going to work. There’s some other reason you’re gaining weight, so let’s move on to something else,’” Dr. Fujioka said at the meeting presented by the Obesity Society of America and the American Society for Metabolic and Bariatric Surgery.
“When you stop the medication, you improve the risk-benefit ratio by removing all risk. That’s a win-win to me, and I applaud the FDA for getting on the pharmaceutical companies to make sure they put stopping rules in their medication labels,” he added.
He presented a post hoc pooled analysis of two previously published large, double-blind phase III clinical trials of subcutaneous liraglutide at 3 mg/day (Saxenda) or placebo in combination with a diet and exercise intervention for weight loss: the 3,731-patient SCALE Obesity and Prediabetes trial and the 846-patient SCALE Diabetes trial. In both trials liraglutide was started at a dose of 0.6 mg and titrated to 3.0 mg by week 4. The lifestyle intervention entailed a 500-kcal/day deficit diet and a minimum of 150 minutes of physical activity per week.
The purpose of the pooled analysis was to identify the best early predictor of response status at 56 weeks by examining the impact of 3%, 4%, and 5% weight loss after 8, 12, and 16 weeks of treatment as cut points. This post hoc analysis was prespecified at the request of the Food and Drug Administration before the trials were completed.
The bottom line: The best predictor of long-term outcome on liraglutide, a glucagonlike peptide–1 analog, proved to be a weight loss of 4% or greater at 16 weeks. It had an 81% positive predictive value and a 76% negative predictive value for at least a 5% weight loss at 56 weeks. It correctly predicted weight outcomes at 56 weeks in 80.1% of patients, the highest success rate of all the combinations studied. This finding was the impetus for the current product labeling, which contains the stopping rule. Dr. Fujioka shared study data not included in the labeling; namely, the marked contrast in how early responders and early nonresponders fared at 56 weeks.
The mean weight loss at 56 weeks in nondiabetic early responders to liraglutide was 10.8%, compared with only 3% in early nondiabetic nonresponders. Diabetic early responders averaged an 8.5% weight loss at 56 weeks, while early nonresponders had a mean 3.1% weight loss.
In the SCALE Obesity and Prediabetes trial, 50% of early responders to liraglutide ended up with a greater than 10% weight loss at 56 weeks, and 21% had more than 15% weight loss, compared with rates of 6% and 2%, respectively, in early nonresponders.
In the SCALE Diabetes study, 38% of early responders had greater than 10% weight loss long term, a rate nearly fourfold higher than in early nonresponders. Moreover, 10% of early responder diabetic patients had greater than 15% weight loss, versus a mere 2% of early nonresponders, the internist continued.
The ratio of early responders to early nonresponders in the nondiabetic population was 77%:23%. In diabetic patients, it was 63%:37%.
Turning to cardiometabolic endpoints, Dr. Fujioka noted that early responders in the SCALE Obesity and Prediabetes trial went on to show a mean reduction in systolic blood pressure of 5.1 mm Hg at 56 weeks, compared with a 2–mm Hg decrease in early nonresponders. Early responders also averaged a 10.5-cm shrinkage in waist circumference from a baseline of 115 cm, which was more than twice that observed at 56 weeks in early nonresponders. HDL-cholesterol level rose by 3.9% in early responders but remained unchanged over time in early nonresponders.
Diabetic patients who were early responders to liraglutide 3.0 mg/day had a mean 44.2-mg/dL reduction in fasting plasma glucose at 56 weeks from a baseline of 158 mg/dL, compared with a 30.1-mg/dL decrease in early nonresponders.
“The drop in fasting blood glucose is very quick – within a matter of weeks – so if you already have diabetic patients on drugs that are going to bring their blood sugar down, you may have to back titrate those other drugs really quickly. You don’t want to make your patients hypoglycemic,” the physician said.
Mean hemoglobin A1c values in early responder diabetic patients fell by 1.6% from a baseline of 7.9%, a full 0.5% greater reduction than in early nonresponders.
By far the most frequent adverse events in the two SCALE trials were gastrointestinal, with nausea leading the way. Rates were modestly higher in the early responders.
This analysis and the clinical trials on which it was based were sponsored by Novo Nordisk, which markets liraglutide under the brand names Saxenda and Voctoza. The presenter reported receiving research grants from and serving as a consultant to Novo Nordisk and other pharmaceutical companies.
AT OBESITY WEEK 2016
Key clinical point:
Major finding: A 4% or greater weight loss at week 16 on liraglutide 3 mg/day had an 81.4% positive predictive value and a 76% negative predictive value for achievement of at least 5% weight loss at week 56 on the drug.
Data source: This was a prespecified post hoc analysis of two phase III randomized, double-blind clinical trials of liraglutide 3 mg/day for weight loss in more than 4,500 subjects.
Disclosures: This analysis and the clinical trials on which it is based were sponsored by Novo Nordisk, which markets liraglutide under the brand names Saxenda and Voctoza. The presenter reported receiving research grants from and serving as a consultant to Novo Nordisk and other pharmaceutical companies.
14% of ASCVD patients need a PCSK9 inhibitor to reach LDL goal
ROME – An estimated 14% of Americans with atherosclerotic cardiovascular disease can’t reach the LDL cholesterol goal of less than 70 mg/dL on maximal intensified oral lipid-lowering therapy and thus are candidates for a PCSK9 inhibitor such as alirocumab, Christopher P. Cannon, MD, reported at the annual congress of the European Society of Cardiology.
After adding alirocumab (Praluent) at 75 mg by subcutaneous injection every 2 weeks, that figure drops to 2%. And by increasing the alirocumab dose to 150 mg in that 2%, the result is that fewer than 1% of patients with atherosclerotic cardiovascular disease (ASCVD) will have an LDL cholesterol level of 70 mg/dL or more, assuming no tolerability issues along the way, added Dr. Cannon, professor of medicine at Harvard Medical School, Boston.
This Monte Carlo simulation relies on lipid-lowering treatment outcome rates from published landmark clinical trials such as IMPROVE-IT (N Engl J Med. 2015 Jun 18;372[25]:2387-97), for which Dr. Cannon was a lead investigator, as well as data from the ongoing ODYSSEY program of alirocumab studies. Importantly, the model doesn’t factor in drug intolerance.
In this model, the average age of the hypothetical 1 million ASCVD patients was 66.5 years and 54.6% were men. The distribution of ASCVD diagnoses was representative of the real-world experience: 70% had coronary heart disease, 25% had ischemic cerebrovascular disease, 35% had peripheral artery disease, and 5% had experienced an acute coronary syndrome within the past 12 months.
Current guidelines would strongly recommend that all of these patients be on lipid-lowering therapy, yet only 53% were at baseline. Guideline-recommended lipid-lowering strategies would suggest that those patients not on a lipid-lowering drug be placed on atorvastatin at 20 mg/day; by that step, 50% of the 1 million ASCVD patients would be at the goal of an LDL cholesterol level below 70 mg/dL.
For the other 50%, a reasonable next step would be a high-intensity statin: say, atorvastatin at 80 mg/day instead of 20. That would leave only 21% of the original ASCVD population with an LDL cholesterol level of 70 mg/dL or higher. The next step for those patients, as established in IMPROVE-IT, would be to add ezetimibe (Zetia). That constitutes maximal oral lipid-lowering therapy, and 14% of the original ASCVD population would still have an LDL cholesterol level of 70 mg/dL or more on that multidrug regimen.
On the basis of the results of the ODYSSEY trials, adding alirocumab at 75 mg would drop that figure from 14% down to 2%. And by switching to alirocumab at 150 mg every 2 weeks in those outliers, less than 1% of the 1 million patients with ASCVD would still have an LDL cholesterol level of 70 mg/dL or more. The mean LDL cholesterol level would be 52.0 mg/dL in patients on full treatment intensification with a high-dose statin, ezetimibe, and alirocumab.
If future studies were to establish that the new LDL cholesterol level goal for patients with known ASCVD was less than 55 mg/dL, the simulation indicates that just under 59% of patients on full-on treatment intensification including alirocumab would achieve it, according to Dr. Cannon.
He reported receiving research grants from and/or serving as a consultant to well over a dozen pharmaceutical companies, including Sanofi and Regeneron, which sponsored this analysis.
ROME – An estimated 14% of Americans with atherosclerotic cardiovascular disease can’t reach the LDL cholesterol goal of less than 70 mg/dL on maximal intensified oral lipid-lowering therapy and thus are candidates for a PCSK9 inhibitor such as alirocumab, Christopher P. Cannon, MD, reported at the annual congress of the European Society of Cardiology.
After adding alirocumab (Praluent) at 75 mg by subcutaneous injection every 2 weeks, that figure drops to 2%. And by increasing the alirocumab dose to 150 mg in that 2%, the result is that fewer than 1% of patients with atherosclerotic cardiovascular disease (ASCVD) will have an LDL cholesterol level of 70 mg/dL or more, assuming no tolerability issues along the way, added Dr. Cannon, professor of medicine at Harvard Medical School, Boston.
This Monte Carlo simulation relies on lipid-lowering treatment outcome rates from published landmark clinical trials such as IMPROVE-IT (N Engl J Med. 2015 Jun 18;372[25]:2387-97), for which Dr. Cannon was a lead investigator, as well as data from the ongoing ODYSSEY program of alirocumab studies. Importantly, the model doesn’t factor in drug intolerance.
In this model, the average age of the hypothetical 1 million ASCVD patients was 66.5 years and 54.6% were men. The distribution of ASCVD diagnoses was representative of the real-world experience: 70% had coronary heart disease, 25% had ischemic cerebrovascular disease, 35% had peripheral artery disease, and 5% had experienced an acute coronary syndrome within the past 12 months.
Current guidelines would strongly recommend that all of these patients be on lipid-lowering therapy, yet only 53% were at baseline. Guideline-recommended lipid-lowering strategies would suggest that those patients not on a lipid-lowering drug be placed on atorvastatin at 20 mg/day; by that step, 50% of the 1 million ASCVD patients would be at the goal of an LDL cholesterol level below 70 mg/dL.
For the other 50%, a reasonable next step would be a high-intensity statin: say, atorvastatin at 80 mg/day instead of 20. That would leave only 21% of the original ASCVD population with an LDL cholesterol level of 70 mg/dL or higher. The next step for those patients, as established in IMPROVE-IT, would be to add ezetimibe (Zetia). That constitutes maximal oral lipid-lowering therapy, and 14% of the original ASCVD population would still have an LDL cholesterol level of 70 mg/dL or more on that multidrug regimen.
On the basis of the results of the ODYSSEY trials, adding alirocumab at 75 mg would drop that figure from 14% down to 2%. And by switching to alirocumab at 150 mg every 2 weeks in those outliers, less than 1% of the 1 million patients with ASCVD would still have an LDL cholesterol level of 70 mg/dL or more. The mean LDL cholesterol level would be 52.0 mg/dL in patients on full treatment intensification with a high-dose statin, ezetimibe, and alirocumab.
If future studies were to establish that the new LDL cholesterol level goal for patients with known ASCVD was less than 55 mg/dL, the simulation indicates that just under 59% of patients on full-on treatment intensification including alirocumab would achieve it, according to Dr. Cannon.
He reported receiving research grants from and/or serving as a consultant to well over a dozen pharmaceutical companies, including Sanofi and Regeneron, which sponsored this analysis.
ROME – An estimated 14% of Americans with atherosclerotic cardiovascular disease can’t reach the LDL cholesterol goal of less than 70 mg/dL on maximal intensified oral lipid-lowering therapy and thus are candidates for a PCSK9 inhibitor such as alirocumab, Christopher P. Cannon, MD, reported at the annual congress of the European Society of Cardiology.
After adding alirocumab (Praluent) at 75 mg by subcutaneous injection every 2 weeks, that figure drops to 2%. And by increasing the alirocumab dose to 150 mg in that 2%, the result is that fewer than 1% of patients with atherosclerotic cardiovascular disease (ASCVD) will have an LDL cholesterol level of 70 mg/dL or more, assuming no tolerability issues along the way, added Dr. Cannon, professor of medicine at Harvard Medical School, Boston.
This Monte Carlo simulation relies on lipid-lowering treatment outcome rates from published landmark clinical trials such as IMPROVE-IT (N Engl J Med. 2015 Jun 18;372[25]:2387-97), for which Dr. Cannon was a lead investigator, as well as data from the ongoing ODYSSEY program of alirocumab studies. Importantly, the model doesn’t factor in drug intolerance.
In this model, the average age of the hypothetical 1 million ASCVD patients was 66.5 years and 54.6% were men. The distribution of ASCVD diagnoses was representative of the real-world experience: 70% had coronary heart disease, 25% had ischemic cerebrovascular disease, 35% had peripheral artery disease, and 5% had experienced an acute coronary syndrome within the past 12 months.
Current guidelines would strongly recommend that all of these patients be on lipid-lowering therapy, yet only 53% were at baseline. Guideline-recommended lipid-lowering strategies would suggest that those patients not on a lipid-lowering drug be placed on atorvastatin at 20 mg/day; by that step, 50% of the 1 million ASCVD patients would be at the goal of an LDL cholesterol level below 70 mg/dL.
For the other 50%, a reasonable next step would be a high-intensity statin: say, atorvastatin at 80 mg/day instead of 20. That would leave only 21% of the original ASCVD population with an LDL cholesterol level of 70 mg/dL or higher. The next step for those patients, as established in IMPROVE-IT, would be to add ezetimibe (Zetia). That constitutes maximal oral lipid-lowering therapy, and 14% of the original ASCVD population would still have an LDL cholesterol level of 70 mg/dL or more on that multidrug regimen.
On the basis of the results of the ODYSSEY trials, adding alirocumab at 75 mg would drop that figure from 14% down to 2%. And by switching to alirocumab at 150 mg every 2 weeks in those outliers, less than 1% of the 1 million patients with ASCVD would still have an LDL cholesterol level of 70 mg/dL or more. The mean LDL cholesterol level would be 52.0 mg/dL in patients on full treatment intensification with a high-dose statin, ezetimibe, and alirocumab.
If future studies were to establish that the new LDL cholesterol level goal for patients with known ASCVD was less than 55 mg/dL, the simulation indicates that just under 59% of patients on full-on treatment intensification including alirocumab would achieve it, according to Dr. Cannon.
He reported receiving research grants from and/or serving as a consultant to well over a dozen pharmaceutical companies, including Sanofi and Regeneron, which sponsored this analysis.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: The combination of a high-intensity statin, ezetimibe, and alirocumab should enable more than 99% of Americans with atherosclerotic cardiovascular disease to achieve an LDL cholesterol level below 70 mg/dL.
Data source: This Monte Carlo simulation model created a hypothetical cohort of 1 million Americans with ASCVD and utilized outcome data from landmark clinical trials to estimate the patients’ ability to achieve a guideline-recommended LDL cholesterol level below 70 mg/dL in response to various intensities of lipid-lowering therapy.
Disclosures: This analysis was funded by Sanofi and Regeneron. The presenter reported receiving research grants from and serving as a consultant to those pharmaceutical companies and more than a dozen others.
Interrupting oral anticoagulation in AF carries high thromboembolic cost
ROME – Temporary interruption of oral anticoagulation for stroke prevention in patients with atrial fibrillation occurs often and is associated with substantially increased risk of both cardioembolic events and all-cause mortality, according to a new prespecified secondary analysis of the ENGAGE-AF TIMI 48 trial.
The analysis showed that many of these treatment interruptions occur in response to nonserious adverse events such as minor bleeding, planned dental procedures, or simply because of patient wishes. The new ENGAGE-AF TIMI 48 findings should encourage physicians and patients to think twice before interrupting anticoagulant therapy for such reasons, which look pretty flimsy in light of the new evidence of the potentially serious consequences, Ilaria Cavallari, MD, said at the annual congress of the European Society of Cardiology.
The ENGAGE-AF TIMI 48 study was the pivotal phase III, double-blind, 21,105-patient clinical trial that led to Food and Drug Administration and European approval of the direct oral factor Xa inhibitor edoxaban (Savaysa) for stroke prevention in moderate- to high-risk patients with AF (N Engl J Med. 2013 Nov 28;369[22]:2093-104). The study showed that edoxaban at what later became the approved dose of 60 mg/day, or 30 mg/day in patients with impaired renal function, body weight of 60 kg or less, or on concomitant therapy with a platelet glycoprotein inhibitor, resulted in a 21% reduction in the risk of stroke or systemic embolism and a 20% reduction in major bleeding, compared with warfarin over 2.8 years of follow-up.
Dr. Cavallari presented a prespecified secondary retrospective analysis that focused on treatment interruptions: the reasons and the price paid in terms of thromboembolic events.
One or more treatment interruptions lasting for longer than 3 days occurred in 63% of patients during a median 2.8 years of follow-up. Since these were participants in a clinical trial with relatively close patient contact, it’s likely that the true interruption rate in real-world clinical practice is even higher, she said.
Interruptions were more significantly frequent in patients assigned to warfarin than among the groups assigned to edoxaban. The median duration of treatment interruptions was 9 days.
After excluding patients who were on any other anticoagulant during their interruption – low-molecular-weight heparin being the most common – investigators were left with 9,148 patients.
The endpoints of interest in this analysis were the major adverse events occurring during a time window lasting from 4 days after their last dose of oral anticoagulant until day 34 or when they resumed their study drug. The 30-day incidence of ischemic stroke or systemic embolism was 1.27%. The rate of a composite composed of cardiovascular death, MI, and ischemic stroke or systemic embolism was 4.99%. And the 30-day rate of an endpoint Dr. Cavallari termed primary net clinical outcome – a composite of stroke or systemic embolism, major bleeding, and all-cause mortality – was 7.16%.
These 30-day event rates among treatment interrupters are extremely high, compared with the 1-year rates in patients who didn’t interrupt oral anticoagulant therapy: 0.26% for ischemic stroke or systemic embolism; 0.36% for the composite of cardiovascular death, MI, and ischemic stroke; and 0.56% for the primary net clinical outcome, she continued.
The most common reason for treatment interruptions was adverse events, which accounted for 41% of the interruptions.
Drilling deeper into the types of adverse events that triggered treatment interruption, 1.5% of interrupters did so because of an on-treatment ischemic stroke or systemic embolism, 4.7% did so because of major bleeding, 8% had minor and clinically relevant nonmajor bleeding, another 30% interrupted treatment for other serious or nonserious adverse events.
Interrupting therapy because of an adverse event often had serious consequences, as reflected in an adjusted 3.94-fold increased risk of 30-day all-cause mortality, compared with patients who stopped for other reasons. Patients who stopped treatment because of a stroke, transient ischemic attack, or systemic embolism had a 30-day all-cause mortality of 29.3%. Those who interrupted treatment because of a major bleeding event had an 8.8% 30-day mortality. When minor or clinically relevant nonmajor bleeding was the impetus for a treatment interruption, the associated 30-day mortality was 3.4%.
Almost a third (29%) of treatment interruptions were the result of physician decisions in response to an upcoming invasive procedure, most often dental work.
The 30-day rates of ischemic stroke/systemic embolism and primary net clinical outcome didn’t differ significantly between patients who interrupted warfarin versus edoxaban at the approved dose. Nonetheless, this new secondary analysis from ENGAGE-AF TIMI 48 supports the parent study’s conclusion that edoxaban is preferable to warfarin in patients with AF, according to Dr. Cavallari.
“In light of the increased risk of ischemic events after interruption of oral anticoagulation, NOACs [new oral anticoagulants] represent an attractive alternative to vitamin K antagonists, given their faster onset of action, better adherence rates, safety, and tolerability profiles,” she concluded.
ENGAGE AF-TIMI 48 was funded by Daiichi Sankyo. Dr. Cavallari reported having no financial conflicts of interest regarding her presentation.
ROME – Temporary interruption of oral anticoagulation for stroke prevention in patients with atrial fibrillation occurs often and is associated with substantially increased risk of both cardioembolic events and all-cause mortality, according to a new prespecified secondary analysis of the ENGAGE-AF TIMI 48 trial.
The analysis showed that many of these treatment interruptions occur in response to nonserious adverse events such as minor bleeding, planned dental procedures, or simply because of patient wishes. The new ENGAGE-AF TIMI 48 findings should encourage physicians and patients to think twice before interrupting anticoagulant therapy for such reasons, which look pretty flimsy in light of the new evidence of the potentially serious consequences, Ilaria Cavallari, MD, said at the annual congress of the European Society of Cardiology.
The ENGAGE-AF TIMI 48 study was the pivotal phase III, double-blind, 21,105-patient clinical trial that led to Food and Drug Administration and European approval of the direct oral factor Xa inhibitor edoxaban (Savaysa) for stroke prevention in moderate- to high-risk patients with AF (N Engl J Med. 2013 Nov 28;369[22]:2093-104). The study showed that edoxaban at what later became the approved dose of 60 mg/day, or 30 mg/day in patients with impaired renal function, body weight of 60 kg or less, or on concomitant therapy with a platelet glycoprotein inhibitor, resulted in a 21% reduction in the risk of stroke or systemic embolism and a 20% reduction in major bleeding, compared with warfarin over 2.8 years of follow-up.
Dr. Cavallari presented a prespecified secondary retrospective analysis that focused on treatment interruptions: the reasons and the price paid in terms of thromboembolic events.
One or more treatment interruptions lasting for longer than 3 days occurred in 63% of patients during a median 2.8 years of follow-up. Since these were participants in a clinical trial with relatively close patient contact, it’s likely that the true interruption rate in real-world clinical practice is even higher, she said.
Interruptions were more significantly frequent in patients assigned to warfarin than among the groups assigned to edoxaban. The median duration of treatment interruptions was 9 days.
After excluding patients who were on any other anticoagulant during their interruption – low-molecular-weight heparin being the most common – investigators were left with 9,148 patients.
The endpoints of interest in this analysis were the major adverse events occurring during a time window lasting from 4 days after their last dose of oral anticoagulant until day 34 or when they resumed their study drug. The 30-day incidence of ischemic stroke or systemic embolism was 1.27%. The rate of a composite composed of cardiovascular death, MI, and ischemic stroke or systemic embolism was 4.99%. And the 30-day rate of an endpoint Dr. Cavallari termed primary net clinical outcome – a composite of stroke or systemic embolism, major bleeding, and all-cause mortality – was 7.16%.
These 30-day event rates among treatment interrupters are extremely high, compared with the 1-year rates in patients who didn’t interrupt oral anticoagulant therapy: 0.26% for ischemic stroke or systemic embolism; 0.36% for the composite of cardiovascular death, MI, and ischemic stroke; and 0.56% for the primary net clinical outcome, she continued.
The most common reason for treatment interruptions was adverse events, which accounted for 41% of the interruptions.
Drilling deeper into the types of adverse events that triggered treatment interruption, 1.5% of interrupters did so because of an on-treatment ischemic stroke or systemic embolism, 4.7% did so because of major bleeding, 8% had minor and clinically relevant nonmajor bleeding, another 30% interrupted treatment for other serious or nonserious adverse events.
Interrupting therapy because of an adverse event often had serious consequences, as reflected in an adjusted 3.94-fold increased risk of 30-day all-cause mortality, compared with patients who stopped for other reasons. Patients who stopped treatment because of a stroke, transient ischemic attack, or systemic embolism had a 30-day all-cause mortality of 29.3%. Those who interrupted treatment because of a major bleeding event had an 8.8% 30-day mortality. When minor or clinically relevant nonmajor bleeding was the impetus for a treatment interruption, the associated 30-day mortality was 3.4%.
Almost a third (29%) of treatment interruptions were the result of physician decisions in response to an upcoming invasive procedure, most often dental work.
The 30-day rates of ischemic stroke/systemic embolism and primary net clinical outcome didn’t differ significantly between patients who interrupted warfarin versus edoxaban at the approved dose. Nonetheless, this new secondary analysis from ENGAGE-AF TIMI 48 supports the parent study’s conclusion that edoxaban is preferable to warfarin in patients with AF, according to Dr. Cavallari.
“In light of the increased risk of ischemic events after interruption of oral anticoagulation, NOACs [new oral anticoagulants] represent an attractive alternative to vitamin K antagonists, given their faster onset of action, better adherence rates, safety, and tolerability profiles,” she concluded.
ENGAGE AF-TIMI 48 was funded by Daiichi Sankyo. Dr. Cavallari reported having no financial conflicts of interest regarding her presentation.
ROME – Temporary interruption of oral anticoagulation for stroke prevention in patients with atrial fibrillation occurs often and is associated with substantially increased risk of both cardioembolic events and all-cause mortality, according to a new prespecified secondary analysis of the ENGAGE-AF TIMI 48 trial.
The analysis showed that many of these treatment interruptions occur in response to nonserious adverse events such as minor bleeding, planned dental procedures, or simply because of patient wishes. The new ENGAGE-AF TIMI 48 findings should encourage physicians and patients to think twice before interrupting anticoagulant therapy for such reasons, which look pretty flimsy in light of the new evidence of the potentially serious consequences, Ilaria Cavallari, MD, said at the annual congress of the European Society of Cardiology.
The ENGAGE-AF TIMI 48 study was the pivotal phase III, double-blind, 21,105-patient clinical trial that led to Food and Drug Administration and European approval of the direct oral factor Xa inhibitor edoxaban (Savaysa) for stroke prevention in moderate- to high-risk patients with AF (N Engl J Med. 2013 Nov 28;369[22]:2093-104). The study showed that edoxaban at what later became the approved dose of 60 mg/day, or 30 mg/day in patients with impaired renal function, body weight of 60 kg or less, or on concomitant therapy with a platelet glycoprotein inhibitor, resulted in a 21% reduction in the risk of stroke or systemic embolism and a 20% reduction in major bleeding, compared with warfarin over 2.8 years of follow-up.
Dr. Cavallari presented a prespecified secondary retrospective analysis that focused on treatment interruptions: the reasons and the price paid in terms of thromboembolic events.
One or more treatment interruptions lasting for longer than 3 days occurred in 63% of patients during a median 2.8 years of follow-up. Since these were participants in a clinical trial with relatively close patient contact, it’s likely that the true interruption rate in real-world clinical practice is even higher, she said.
Interruptions were more significantly frequent in patients assigned to warfarin than among the groups assigned to edoxaban. The median duration of treatment interruptions was 9 days.
After excluding patients who were on any other anticoagulant during their interruption – low-molecular-weight heparin being the most common – investigators were left with 9,148 patients.
The endpoints of interest in this analysis were the major adverse events occurring during a time window lasting from 4 days after their last dose of oral anticoagulant until day 34 or when they resumed their study drug. The 30-day incidence of ischemic stroke or systemic embolism was 1.27%. The rate of a composite composed of cardiovascular death, MI, and ischemic stroke or systemic embolism was 4.99%. And the 30-day rate of an endpoint Dr. Cavallari termed primary net clinical outcome – a composite of stroke or systemic embolism, major bleeding, and all-cause mortality – was 7.16%.
These 30-day event rates among treatment interrupters are extremely high, compared with the 1-year rates in patients who didn’t interrupt oral anticoagulant therapy: 0.26% for ischemic stroke or systemic embolism; 0.36% for the composite of cardiovascular death, MI, and ischemic stroke; and 0.56% for the primary net clinical outcome, she continued.
The most common reason for treatment interruptions was adverse events, which accounted for 41% of the interruptions.
Drilling deeper into the types of adverse events that triggered treatment interruption, 1.5% of interrupters did so because of an on-treatment ischemic stroke or systemic embolism, 4.7% did so because of major bleeding, 8% had minor and clinically relevant nonmajor bleeding, another 30% interrupted treatment for other serious or nonserious adverse events.
Interrupting therapy because of an adverse event often had serious consequences, as reflected in an adjusted 3.94-fold increased risk of 30-day all-cause mortality, compared with patients who stopped for other reasons. Patients who stopped treatment because of a stroke, transient ischemic attack, or systemic embolism had a 30-day all-cause mortality of 29.3%. Those who interrupted treatment because of a major bleeding event had an 8.8% 30-day mortality. When minor or clinically relevant nonmajor bleeding was the impetus for a treatment interruption, the associated 30-day mortality was 3.4%.
Almost a third (29%) of treatment interruptions were the result of physician decisions in response to an upcoming invasive procedure, most often dental work.
The 30-day rates of ischemic stroke/systemic embolism and primary net clinical outcome didn’t differ significantly between patients who interrupted warfarin versus edoxaban at the approved dose. Nonetheless, this new secondary analysis from ENGAGE-AF TIMI 48 supports the parent study’s conclusion that edoxaban is preferable to warfarin in patients with AF, according to Dr. Cavallari.
“In light of the increased risk of ischemic events after interruption of oral anticoagulation, NOACs [new oral anticoagulants] represent an attractive alternative to vitamin K antagonists, given their faster onset of action, better adherence rates, safety, and tolerability profiles,” she concluded.
ENGAGE AF-TIMI 48 was funded by Daiichi Sankyo. Dr. Cavallari reported having no financial conflicts of interest regarding her presentation.
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: Atrial fibrillation patients who interrupted oral anticoagulation with edoxaban or warfarin for stroke prevention for longer than 3 days had a 127-fold increase in 30-day risk of the composite of stroke or systemic embolism, major bleeding, or all-cause mortality, compared with patients who didn’t interrupt treatment after 1 year.
Data source: A prespecified secondary analysis of 9,148 patients with atrial fibrillation who interrupted oral anticoagulation therapy with warfarin or edoxaban for longer than 3 days during a median follow-up of 2.8 years in the phase III, randomized, double-blind ENGAGE AF-TIMI 48 trial, and the consequences thereof.
Disclosures: The ENGAGE AF-TIMI 48 trial was funded by Daiichi Sankyo, which markets edoxaban. However, the presenter of this secondary analysis reported having no relevant financial interests.