User login
Mediterranean diet for secondary prevention cuts all-cause mortality
ROME – Adherence to the traditional Mediterranean diet by patients with known heart or cerebrovascular disease was independently linked with a marked reduction in their risk of all-cause mortality in the Italian Moli-Sani Study, Giovanni de Gaetano, MD, reported at the annual congress of the European Society of Cardiology.
Subjects in the top tertile in terms of food intake consistent with the Mediterranean diet were 37% less likely to die during more than 7 years of follow-up than were those in the bottom tertile, according to Dr. de Gaetano, head of the department of epidemiology and prevention at the IRCCS Mediterranean Neurologic Institute in Pozzilli, Italy.
The Moli-Sani Study is an ongoing population-based epidemiologic study of 25,000 adults in the mountainous, heavily agricultural Molise region of southern Italy. Dr. de Gaetano, who directs the study, presented a substudy involving 1,197 participants with established coronary heart disease or cerebrovascular disease at entry. Their average age at enrollment was 66 years. Subjects with coronary heart disease outnumbered those with cerebrovascular disease by roughly 2:1.
Food intake was recorded using the European Prospective Investigation into Cancer (EPIC) food-frequency questionnaire. Dr. de Gaetano and his coinvestigators assessed adherence to the traditional Mediterranean diet using the Mediterranean Diet Score (MDS), a validated 0-9 scoring system developed as part of the famous Seven Countries Study pioneered by the late Ancel Keys.
During a median 7.3 years of prospective follow-up, 208 deaths occurred in the study population. A 2-point increase in the MDS was independently associated with a 21% reduction in the risk of mortality in a multivariate Cox proportional hazards analysis adjusted for “everything we could think of,” according to Dr. de Gaetano, including baseline demographics, socioeconomic status, energy intake, body mass index, leisure time physical activity, waist-to-hip ratio, smoking status, diabetes, standard cardiovascular risk factors, and use of cardiovascular medications.
Subjects in the top tertile for adherence to the Mediterranean diet, with an MDS of 6-9, had an adjusted 37% relative risk reduction in all-cause mortality, compared with those having an MDS of 0-3.
The number of deaths is too small at this point in the prospective study to permit analysis of specific causes of death in a statistically valid manner. With another 3 years or so of follow-up, that analysis can and will be done, Dr. de Gaetano said.
The traditional Mediterranean diet is an eating pattern characteristic of the Mediterranean basin, he explained. It encourages large intake of vegetables, legumes, fruits, nuts, and cereals, along with moderately high consumption of fish, olive oil as the primary fat source, and moderate alcohol intake during meals, but low-to-moderate intake of dairy products and low consumption of meat and poultry.
As a sobering aside, he said that in the Molise region, far and away the biggest obstacle to adherence to the Mediterranean diet is economic.
“There was no difference in adherence to the Mediterranean diet between different socioeconomic classes in the Moli-Sani Study until 2007-2008, when the Italian economic crisis began. Since then there are significant differences according to socioeconomic condition. Poor people are obliged to follow the Mediterranean diet less,” Dr. de Gaetano said.
Telling a patient with cardiovascular disease who is in a low-income family of four to eat fish at least twice per week is impractical advice, he explained.
The ongoing Moli-Sani Study is funded by the Italian government. Dr. de Gaetano reported having no financial conflicts of interest.
ROME – Adherence to the traditional Mediterranean diet by patients with known heart or cerebrovascular disease was independently linked with a marked reduction in their risk of all-cause mortality in the Italian Moli-Sani Study, Giovanni de Gaetano, MD, reported at the annual congress of the European Society of Cardiology.
Subjects in the top tertile in terms of food intake consistent with the Mediterranean diet were 37% less likely to die during more than 7 years of follow-up than were those in the bottom tertile, according to Dr. de Gaetano, head of the department of epidemiology and prevention at the IRCCS Mediterranean Neurologic Institute in Pozzilli, Italy.
The Moli-Sani Study is an ongoing population-based epidemiologic study of 25,000 adults in the mountainous, heavily agricultural Molise region of southern Italy. Dr. de Gaetano, who directs the study, presented a substudy involving 1,197 participants with established coronary heart disease or cerebrovascular disease at entry. Their average age at enrollment was 66 years. Subjects with coronary heart disease outnumbered those with cerebrovascular disease by roughly 2:1.
Food intake was recorded using the European Prospective Investigation into Cancer (EPIC) food-frequency questionnaire. Dr. de Gaetano and his coinvestigators assessed adherence to the traditional Mediterranean diet using the Mediterranean Diet Score (MDS), a validated 0-9 scoring system developed as part of the famous Seven Countries Study pioneered by the late Ancel Keys.
During a median 7.3 years of prospective follow-up, 208 deaths occurred in the study population. A 2-point increase in the MDS was independently associated with a 21% reduction in the risk of mortality in a multivariate Cox proportional hazards analysis adjusted for “everything we could think of,” according to Dr. de Gaetano, including baseline demographics, socioeconomic status, energy intake, body mass index, leisure time physical activity, waist-to-hip ratio, smoking status, diabetes, standard cardiovascular risk factors, and use of cardiovascular medications.
Subjects in the top tertile for adherence to the Mediterranean diet, with an MDS of 6-9, had an adjusted 37% relative risk reduction in all-cause mortality, compared with those having an MDS of 0-3.
The number of deaths is too small at this point in the prospective study to permit analysis of specific causes of death in a statistically valid manner. With another 3 years or so of follow-up, that analysis can and will be done, Dr. de Gaetano said.
The traditional Mediterranean diet is an eating pattern characteristic of the Mediterranean basin, he explained. It encourages large intake of vegetables, legumes, fruits, nuts, and cereals, along with moderately high consumption of fish, olive oil as the primary fat source, and moderate alcohol intake during meals, but low-to-moderate intake of dairy products and low consumption of meat and poultry.
As a sobering aside, he said that in the Molise region, far and away the biggest obstacle to adherence to the Mediterranean diet is economic.
“There was no difference in adherence to the Mediterranean diet between different socioeconomic classes in the Moli-Sani Study until 2007-2008, when the Italian economic crisis began. Since then there are significant differences according to socioeconomic condition. Poor people are obliged to follow the Mediterranean diet less,” Dr. de Gaetano said.
Telling a patient with cardiovascular disease who is in a low-income family of four to eat fish at least twice per week is impractical advice, he explained.
The ongoing Moli-Sani Study is funded by the Italian government. Dr. de Gaetano reported having no financial conflicts of interest.
ROME – Adherence to the traditional Mediterranean diet by patients with known heart or cerebrovascular disease was independently linked with a marked reduction in their risk of all-cause mortality in the Italian Moli-Sani Study, Giovanni de Gaetano, MD, reported at the annual congress of the European Society of Cardiology.
Subjects in the top tertile in terms of food intake consistent with the Mediterranean diet were 37% less likely to die during more than 7 years of follow-up than were those in the bottom tertile, according to Dr. de Gaetano, head of the department of epidemiology and prevention at the IRCCS Mediterranean Neurologic Institute in Pozzilli, Italy.
The Moli-Sani Study is an ongoing population-based epidemiologic study of 25,000 adults in the mountainous, heavily agricultural Molise region of southern Italy. Dr. de Gaetano, who directs the study, presented a substudy involving 1,197 participants with established coronary heart disease or cerebrovascular disease at entry. Their average age at enrollment was 66 years. Subjects with coronary heart disease outnumbered those with cerebrovascular disease by roughly 2:1.
Food intake was recorded using the European Prospective Investigation into Cancer (EPIC) food-frequency questionnaire. Dr. de Gaetano and his coinvestigators assessed adherence to the traditional Mediterranean diet using the Mediterranean Diet Score (MDS), a validated 0-9 scoring system developed as part of the famous Seven Countries Study pioneered by the late Ancel Keys.
During a median 7.3 years of prospective follow-up, 208 deaths occurred in the study population. A 2-point increase in the MDS was independently associated with a 21% reduction in the risk of mortality in a multivariate Cox proportional hazards analysis adjusted for “everything we could think of,” according to Dr. de Gaetano, including baseline demographics, socioeconomic status, energy intake, body mass index, leisure time physical activity, waist-to-hip ratio, smoking status, diabetes, standard cardiovascular risk factors, and use of cardiovascular medications.
Subjects in the top tertile for adherence to the Mediterranean diet, with an MDS of 6-9, had an adjusted 37% relative risk reduction in all-cause mortality, compared with those having an MDS of 0-3.
The number of deaths is too small at this point in the prospective study to permit analysis of specific causes of death in a statistically valid manner. With another 3 years or so of follow-up, that analysis can and will be done, Dr. de Gaetano said.
The traditional Mediterranean diet is an eating pattern characteristic of the Mediterranean basin, he explained. It encourages large intake of vegetables, legumes, fruits, nuts, and cereals, along with moderately high consumption of fish, olive oil as the primary fat source, and moderate alcohol intake during meals, but low-to-moderate intake of dairy products and low consumption of meat and poultry.
As a sobering aside, he said that in the Molise region, far and away the biggest obstacle to adherence to the Mediterranean diet is economic.
“There was no difference in adherence to the Mediterranean diet between different socioeconomic classes in the Moli-Sani Study until 2007-2008, when the Italian economic crisis began. Since then there are significant differences according to socioeconomic condition. Poor people are obliged to follow the Mediterranean diet less,” Dr. de Gaetano said.
Telling a patient with cardiovascular disease who is in a low-income family of four to eat fish at least twice per week is impractical advice, he explained.
The ongoing Moli-Sani Study is funded by the Italian government. Dr. de Gaetano reported having no financial conflicts of interest.
Key clinical point:
Major finding: Italian patients with known cardiovascular disease who were in the top third in terms of adherence to the traditional Mediterranean diet were a fully adjusted 37% less likely to die of any cause during follow-up than those in the bottom tertile.
Data source: This substudy of the prospective, observational Italian Moli-Sani Study included 1,197 participants with coronary heart disease or cerebrovascular disease at baseline who were followed for a median of 7.3 years.
Disclosures: The ongoing Moli-Sani Study is funded by the Italian government. The presenter reported having no financial conflicts of interest.
Recognizing anti-NMDA receptor encephalitis psychosis on the psych ward
VIENNA – Prominent psychiatric symptoms are common in patients with anti-N-methyl-D-asparate receptor (NMDAR) encephalitis and often occur prior to onset of obvious neurologic symptoms, Maarten J. Titulaer, MD, PhD, said at the annual congress of the European College of Neuropsychopharmacology.
Moreover, occasionally the psychiatric symptoms occur in isolation without neurologic involvement, as was the case in 4% of a series of 501 patients with confirmed anti-NMDAR encephalitis reported by Dr. Titulaer and coinvestigators. The most prominent symptoms included delusional thinking, aggression, and mood disturbances, which were usually manic (JAMA Neurol. 2013 Sep 1;70[9]:1133-9).
Anti-NMDAR encephalitis is an autoimmune disorder in which autoantibodies directed at NMDA receptors on neuronal plasma membranes induce severe neurologic and often psychiatric symptoms.
Red flags that raise the chance that a patient on the psychiatric ward with new-onset psychosis or mania might have primary anti-NMDAR encephalitis and should undergo diagnostic testing include autonomic disturbances such as tachycardia, fever, or hypertension, mild neurologic symptoms such as facial twitching, as well as catatonia, seizures, mutism, or development of extrapyramidal symptoms when placed on an antipsychotic agent. Anti-NMDAR encephalitis can have a relapsing course, so any behavioral change in a patient with a history of the disorder might signal relapse.
Certain cancers are strongly associated with anti-NMDAR encephalitis. New-onset psychotic or manic patients with a history of ovarian teratoma, small cell lung cancer, breast cancer, or thymoma should be tested for anti-NMDAR encephalitis. And conversely, screening for those tumors in occult form is warranted in patients with confirmed anti-NMDAR encephalitis, according to Dr. Titulaer.
Systematic screening for anti-NMDAR encephalitis should also be considered in women with severe acute psychosis during the postpartum period, particularly in the setting of extrapyramidal side effects of antipsychotic agents. Two of 96 consecutive women with acute-onset postpartum psychosis in a series reported by Dr. Titulaer and colleagues were antibody-positive for the disorder, and neither had an ovarian teratoma (Am J Psychiatry. 2015 Sep 1;172[9]:901-8).
If a patient hasn’t developed neurologic symptoms within 4 weeks after onset of psychiatric symptoms, anti-NMDAR psychosis becomes far less likely.
Some neurologists have suggested the presence of other autoimmune disorders in psychiatric patients is associated with increased likelihood that the psychiatric symptoms are secondary to anti-NMDAR encephalitis, but Dr. Titulaer doesn’t find the evidence to date persuasive.
The diagnosis of anti-NMDAR encephalitis hinges on the finding of IgG antibodies against the NR1 subunit of the NMDAR. But Dr. Titulaer and coinvestigators have shown there are testing pitfalls: The first-line commercially available cell-based serum assays have a sensitivity of roughly 75% along with 97%-99% specificity, so by relying solely on the cell-based assays a physician might miss one in four cases of anti-NMDAR encephalitis and wrongly diagnose the disease in 0.4%-3% of healthy individuals (Lancet Neurol. 2014 Feb;13[2]:167-77).
For this reason, a positive serum test should be confirmed by a cell-based assay of a cerebrospinal fluid (CSF) sample, which has 100% sensitivity and specificity. And if the serum assay is negative but anti-NMDAR is suspected based on clinical grounds or history, go ahead and test the CSF, the neurologist advised.
Other tools that can be helpful in making the diagnosis include the EEG, which is abnormal in 89% of patients with anti-NMDAR encephalitis. Thirty percent of affected patients will display a highly specific EEG abnormality called extreme delta brushes (Neurology. 2012 Sep 11;79[11]:1094-100).
Dr. Titulaer said that this extreme delta brushes pattern is not seen on the regular psychiatry ward, but only in the ICU, when the patient is severely ill. He has yet to see the first convincing extreme delta brushes pattern in a patient outside the ICU.
Brain MRI has proved “very disappointing,” as it’s abnormal in only one-third of patients with anti-NMDAR encephalitis, he continued.
First-line immunotherapy is corticosteroids, plasmapheresis, and/or intravenous immunoglobulin. In a series of 501 patients who received first-line immunotherapy or tumor removal, 53% improved within 4 weeks. Fifty-seven percent of those who didn’t then got second-line immunotherapy with rituximab (Rituxan) or cyclophosphamide. Outcomes continued to improve for up to 18 months following symptom onset. At 24 months of follow-up, just over 80% of patients in this observational study had a good outcome as defined by a modified Rankin scale score of 0-2, meaning they were living independently with no or minimal disability.
“Not bad, especially considering that the patients who didn’t improve on first-line therapy were in the ICU for a median of 6 weeks,” the neurologist observed.
“It’s important to diagnose patients with anti-NMDAR encephalitis,” he stressed. “Treatment might be difficult. You might need to be very aggressive. But in the end there are very good outcomes. It’s very rewarding to treat these patients.”
In multivariate analysis, Dr. Titulaer and coworkers identified earlier treatment and milder illness as reflected in no ICU admission as significant predictors of good outcome in the study population. Also, the use of second-line immunotherapy in nonresponders to first-line therapy was independently associated with a 2.69-fold increased likelihood of good outcome (Lancet Neurol. 2013 Feb;12[2]:157-65).
Twelve percent of patients experienced one or more relapses within 2 years.
In a separate study of 661 patients with anti-NMDAR encephalitis, only 31 were aged 45 years or older. They had less severe disease than the younger adults but a paradoxically worse outcome, possibly because their median time to diagnosis was twice as long. At 2 years, 60% of the patients aged 45 and up had full or substantial recovery (Neurology. 2013 Sep 17;81[12]:1058-63).
He stressed that treatment of anti-NMDAR encephalitis ought to be an interdisciplinary effort. Psychiatrists will typically not be the ones who administer the potent immunotherapy. But most patients will have behavioral problems in the very early and late phases that warrant psychiatric therapy. Dr. Titulaer suggested psychiatrists steer clear of haloperidol in these patients because it can exacerbate motor symptoms.
Asked if there are any specific patterns of movement disorders linked to anti-NMDAR encephalitis that might raise a psychiatrist’s index of suspicion, the neurologist replied no. Almost all the movement disorders have been seen in psychiatric patients with anti-NMDAR encephalitis. The one specific movement disorder that strongly suggests anti-NMDAR encephalitis is post–herpes simplex virus (HSV) encephalitis choreoathetosis. It appears that HSV encephalitis can trigger formation of NMDAR autoantibodies, resulting in onset of choreoathetosis 3-6 weeks after the HSV encephalopathy.
Dr. Titulaer reported having no financial conflicts of interest in regard to his presentation.
*This story was updated 1/26/2017.
VIENNA – Prominent psychiatric symptoms are common in patients with anti-N-methyl-D-asparate receptor (NMDAR) encephalitis and often occur prior to onset of obvious neurologic symptoms, Maarten J. Titulaer, MD, PhD, said at the annual congress of the European College of Neuropsychopharmacology.
Moreover, occasionally the psychiatric symptoms occur in isolation without neurologic involvement, as was the case in 4% of a series of 501 patients with confirmed anti-NMDAR encephalitis reported by Dr. Titulaer and coinvestigators. The most prominent symptoms included delusional thinking, aggression, and mood disturbances, which were usually manic (JAMA Neurol. 2013 Sep 1;70[9]:1133-9).
Anti-NMDAR encephalitis is an autoimmune disorder in which autoantibodies directed at NMDA receptors on neuronal plasma membranes induce severe neurologic and often psychiatric symptoms.
Red flags that raise the chance that a patient on the psychiatric ward with new-onset psychosis or mania might have primary anti-NMDAR encephalitis and should undergo diagnostic testing include autonomic disturbances such as tachycardia, fever, or hypertension, mild neurologic symptoms such as facial twitching, as well as catatonia, seizures, mutism, or development of extrapyramidal symptoms when placed on an antipsychotic agent. Anti-NMDAR encephalitis can have a relapsing course, so any behavioral change in a patient with a history of the disorder might signal relapse.
Certain cancers are strongly associated with anti-NMDAR encephalitis. New-onset psychotic or manic patients with a history of ovarian teratoma, small cell lung cancer, breast cancer, or thymoma should be tested for anti-NMDAR encephalitis. And conversely, screening for those tumors in occult form is warranted in patients with confirmed anti-NMDAR encephalitis, according to Dr. Titulaer.
Systematic screening for anti-NMDAR encephalitis should also be considered in women with severe acute psychosis during the postpartum period, particularly in the setting of extrapyramidal side effects of antipsychotic agents. Two of 96 consecutive women with acute-onset postpartum psychosis in a series reported by Dr. Titulaer and colleagues were antibody-positive for the disorder, and neither had an ovarian teratoma (Am J Psychiatry. 2015 Sep 1;172[9]:901-8).
If a patient hasn’t developed neurologic symptoms within 4 weeks after onset of psychiatric symptoms, anti-NMDAR psychosis becomes far less likely.
Some neurologists have suggested the presence of other autoimmune disorders in psychiatric patients is associated with increased likelihood that the psychiatric symptoms are secondary to anti-NMDAR encephalitis, but Dr. Titulaer doesn’t find the evidence to date persuasive.
The diagnosis of anti-NMDAR encephalitis hinges on the finding of IgG antibodies against the NR1 subunit of the NMDAR. But Dr. Titulaer and coinvestigators have shown there are testing pitfalls: The first-line commercially available cell-based serum assays have a sensitivity of roughly 75% along with 97%-99% specificity, so by relying solely on the cell-based assays a physician might miss one in four cases of anti-NMDAR encephalitis and wrongly diagnose the disease in 0.4%-3% of healthy individuals (Lancet Neurol. 2014 Feb;13[2]:167-77).
For this reason, a positive serum test should be confirmed by a cell-based assay of a cerebrospinal fluid (CSF) sample, which has 100% sensitivity and specificity. And if the serum assay is negative but anti-NMDAR is suspected based on clinical grounds or history, go ahead and test the CSF, the neurologist advised.
Other tools that can be helpful in making the diagnosis include the EEG, which is abnormal in 89% of patients with anti-NMDAR encephalitis. Thirty percent of affected patients will display a highly specific EEG abnormality called extreme delta brushes (Neurology. 2012 Sep 11;79[11]:1094-100).
Dr. Titulaer said that this extreme delta brushes pattern is not seen on the regular psychiatry ward, but only in the ICU, when the patient is severely ill. He has yet to see the first convincing extreme delta brushes pattern in a patient outside the ICU.
Brain MRI has proved “very disappointing,” as it’s abnormal in only one-third of patients with anti-NMDAR encephalitis, he continued.
First-line immunotherapy is corticosteroids, plasmapheresis, and/or intravenous immunoglobulin. In a series of 501 patients who received first-line immunotherapy or tumor removal, 53% improved within 4 weeks. Fifty-seven percent of those who didn’t then got second-line immunotherapy with rituximab (Rituxan) or cyclophosphamide. Outcomes continued to improve for up to 18 months following symptom onset. At 24 months of follow-up, just over 80% of patients in this observational study had a good outcome as defined by a modified Rankin scale score of 0-2, meaning they were living independently with no or minimal disability.
“Not bad, especially considering that the patients who didn’t improve on first-line therapy were in the ICU for a median of 6 weeks,” the neurologist observed.
“It’s important to diagnose patients with anti-NMDAR encephalitis,” he stressed. “Treatment might be difficult. You might need to be very aggressive. But in the end there are very good outcomes. It’s very rewarding to treat these patients.”
In multivariate analysis, Dr. Titulaer and coworkers identified earlier treatment and milder illness as reflected in no ICU admission as significant predictors of good outcome in the study population. Also, the use of second-line immunotherapy in nonresponders to first-line therapy was independently associated with a 2.69-fold increased likelihood of good outcome (Lancet Neurol. 2013 Feb;12[2]:157-65).
Twelve percent of patients experienced one or more relapses within 2 years.
In a separate study of 661 patients with anti-NMDAR encephalitis, only 31 were aged 45 years or older. They had less severe disease than the younger adults but a paradoxically worse outcome, possibly because their median time to diagnosis was twice as long. At 2 years, 60% of the patients aged 45 and up had full or substantial recovery (Neurology. 2013 Sep 17;81[12]:1058-63).
He stressed that treatment of anti-NMDAR encephalitis ought to be an interdisciplinary effort. Psychiatrists will typically not be the ones who administer the potent immunotherapy. But most patients will have behavioral problems in the very early and late phases that warrant psychiatric therapy. Dr. Titulaer suggested psychiatrists steer clear of haloperidol in these patients because it can exacerbate motor symptoms.
Asked if there are any specific patterns of movement disorders linked to anti-NMDAR encephalitis that might raise a psychiatrist’s index of suspicion, the neurologist replied no. Almost all the movement disorders have been seen in psychiatric patients with anti-NMDAR encephalitis. The one specific movement disorder that strongly suggests anti-NMDAR encephalitis is post–herpes simplex virus (HSV) encephalitis choreoathetosis. It appears that HSV encephalitis can trigger formation of NMDAR autoantibodies, resulting in onset of choreoathetosis 3-6 weeks after the HSV encephalopathy.
Dr. Titulaer reported having no financial conflicts of interest in regard to his presentation.
*This story was updated 1/26/2017.
VIENNA – Prominent psychiatric symptoms are common in patients with anti-N-methyl-D-asparate receptor (NMDAR) encephalitis and often occur prior to onset of obvious neurologic symptoms, Maarten J. Titulaer, MD, PhD, said at the annual congress of the European College of Neuropsychopharmacology.
Moreover, occasionally the psychiatric symptoms occur in isolation without neurologic involvement, as was the case in 4% of a series of 501 patients with confirmed anti-NMDAR encephalitis reported by Dr. Titulaer and coinvestigators. The most prominent symptoms included delusional thinking, aggression, and mood disturbances, which were usually manic (JAMA Neurol. 2013 Sep 1;70[9]:1133-9).
Anti-NMDAR encephalitis is an autoimmune disorder in which autoantibodies directed at NMDA receptors on neuronal plasma membranes induce severe neurologic and often psychiatric symptoms.
Red flags that raise the chance that a patient on the psychiatric ward with new-onset psychosis or mania might have primary anti-NMDAR encephalitis and should undergo diagnostic testing include autonomic disturbances such as tachycardia, fever, or hypertension, mild neurologic symptoms such as facial twitching, as well as catatonia, seizures, mutism, or development of extrapyramidal symptoms when placed on an antipsychotic agent. Anti-NMDAR encephalitis can have a relapsing course, so any behavioral change in a patient with a history of the disorder might signal relapse.
Certain cancers are strongly associated with anti-NMDAR encephalitis. New-onset psychotic or manic patients with a history of ovarian teratoma, small cell lung cancer, breast cancer, or thymoma should be tested for anti-NMDAR encephalitis. And conversely, screening for those tumors in occult form is warranted in patients with confirmed anti-NMDAR encephalitis, according to Dr. Titulaer.
Systematic screening for anti-NMDAR encephalitis should also be considered in women with severe acute psychosis during the postpartum period, particularly in the setting of extrapyramidal side effects of antipsychotic agents. Two of 96 consecutive women with acute-onset postpartum psychosis in a series reported by Dr. Titulaer and colleagues were antibody-positive for the disorder, and neither had an ovarian teratoma (Am J Psychiatry. 2015 Sep 1;172[9]:901-8).
If a patient hasn’t developed neurologic symptoms within 4 weeks after onset of psychiatric symptoms, anti-NMDAR psychosis becomes far less likely.
Some neurologists have suggested the presence of other autoimmune disorders in psychiatric patients is associated with increased likelihood that the psychiatric symptoms are secondary to anti-NMDAR encephalitis, but Dr. Titulaer doesn’t find the evidence to date persuasive.
The diagnosis of anti-NMDAR encephalitis hinges on the finding of IgG antibodies against the NR1 subunit of the NMDAR. But Dr. Titulaer and coinvestigators have shown there are testing pitfalls: The first-line commercially available cell-based serum assays have a sensitivity of roughly 75% along with 97%-99% specificity, so by relying solely on the cell-based assays a physician might miss one in four cases of anti-NMDAR encephalitis and wrongly diagnose the disease in 0.4%-3% of healthy individuals (Lancet Neurol. 2014 Feb;13[2]:167-77).
For this reason, a positive serum test should be confirmed by a cell-based assay of a cerebrospinal fluid (CSF) sample, which has 100% sensitivity and specificity. And if the serum assay is negative but anti-NMDAR is suspected based on clinical grounds or history, go ahead and test the CSF, the neurologist advised.
Other tools that can be helpful in making the diagnosis include the EEG, which is abnormal in 89% of patients with anti-NMDAR encephalitis. Thirty percent of affected patients will display a highly specific EEG abnormality called extreme delta brushes (Neurology. 2012 Sep 11;79[11]:1094-100).
Dr. Titulaer said that this extreme delta brushes pattern is not seen on the regular psychiatry ward, but only in the ICU, when the patient is severely ill. He has yet to see the first convincing extreme delta brushes pattern in a patient outside the ICU.
Brain MRI has proved “very disappointing,” as it’s abnormal in only one-third of patients with anti-NMDAR encephalitis, he continued.
First-line immunotherapy is corticosteroids, plasmapheresis, and/or intravenous immunoglobulin. In a series of 501 patients who received first-line immunotherapy or tumor removal, 53% improved within 4 weeks. Fifty-seven percent of those who didn’t then got second-line immunotherapy with rituximab (Rituxan) or cyclophosphamide. Outcomes continued to improve for up to 18 months following symptom onset. At 24 months of follow-up, just over 80% of patients in this observational study had a good outcome as defined by a modified Rankin scale score of 0-2, meaning they were living independently with no or minimal disability.
“Not bad, especially considering that the patients who didn’t improve on first-line therapy were in the ICU for a median of 6 weeks,” the neurologist observed.
“It’s important to diagnose patients with anti-NMDAR encephalitis,” he stressed. “Treatment might be difficult. You might need to be very aggressive. But in the end there are very good outcomes. It’s very rewarding to treat these patients.”
In multivariate analysis, Dr. Titulaer and coworkers identified earlier treatment and milder illness as reflected in no ICU admission as significant predictors of good outcome in the study population. Also, the use of second-line immunotherapy in nonresponders to first-line therapy was independently associated with a 2.69-fold increased likelihood of good outcome (Lancet Neurol. 2013 Feb;12[2]:157-65).
Twelve percent of patients experienced one or more relapses within 2 years.
In a separate study of 661 patients with anti-NMDAR encephalitis, only 31 were aged 45 years or older. They had less severe disease than the younger adults but a paradoxically worse outcome, possibly because their median time to diagnosis was twice as long. At 2 years, 60% of the patients aged 45 and up had full or substantial recovery (Neurology. 2013 Sep 17;81[12]:1058-63).
He stressed that treatment of anti-NMDAR encephalitis ought to be an interdisciplinary effort. Psychiatrists will typically not be the ones who administer the potent immunotherapy. But most patients will have behavioral problems in the very early and late phases that warrant psychiatric therapy. Dr. Titulaer suggested psychiatrists steer clear of haloperidol in these patients because it can exacerbate motor symptoms.
Asked if there are any specific patterns of movement disorders linked to anti-NMDAR encephalitis that might raise a psychiatrist’s index of suspicion, the neurologist replied no. Almost all the movement disorders have been seen in psychiatric patients with anti-NMDAR encephalitis. The one specific movement disorder that strongly suggests anti-NMDAR encephalitis is post–herpes simplex virus (HSV) encephalitis choreoathetosis. It appears that HSV encephalitis can trigger formation of NMDAR autoantibodies, resulting in onset of choreoathetosis 3-6 weeks after the HSV encephalopathy.
Dr. Titulaer reported having no financial conflicts of interest in regard to his presentation.
*This story was updated 1/26/2017.
MIs in pregnancy have worse prognosis
ROME – It’s fortunate that pregnancy-associated acute MIs are infrequent, because the associated in-hospital mortality is markedly higher than in similar-age nonchildbearing women, Reza Masoomi, MD, said at the annual congress of the European Society of Cardiology.
One likely contributor to the disparity in outcome is that current management appears to feature underutilization of percutaneous intervention in women who experience pregnancy-associated MI, according to Dr. Masoomi of the University of Kansas in Kansas City.
He presented an analysis of the U.S. National Inpatient Sample database for the years 2008-2012. The NIS is a nationally representative sample of hospitalizations drawn from all of the country’s nonfederal acute-care hospitals.
A total of 55,315 hospitalizations with a discharge diagnosis of acute MI were recorded in women aged 15-54 years during the study years, of which 453 involved an ante- or postpartum MI. Extrapolating from those figures, nearly 262,000 women aged 15-54 years across the U.S. had an acute MI during the study years, of whom an estimated 2,153 experienced a pregnancy-associated MI.
In-hospital mortality among women with peripregnancy MI was 7.2%, significantly higher than the 5.2% rate in women who weren’t pregnant.
Women with peripregnancy MI had a significantly higher rate of ST-elevation MI (STEMI) than did nonpregnant women with MI in their reproductive years, by a margin of 35.3% to 32.8%. They were younger, too: an average age of 34.9 years, compared with 47.3 years in nonpregnant patients with an MI. Nearly two-thirds of women with peripregnancy MI were nonwhite, compared with 47.3% of the comparison group.
Regardless of whether women with peripregnancy MI had a STEMI or non-STEMI, they had significantly lower rates of diagnostic coronary angiography and percutaneous intervention. They were also far less likely to receive drug-eluting stents.
Diagnostic coronary angiography was performed in 59% of women with pregnancy-associated STEMI, compared with 73% of nonpregnant women with a STEMI. Only 34% of patients with peripregnancy STEMI underwent PCI, compared with 61% of nonpregnant women with a STEMI. Drug-eluting stents were implanted in 12% of peripregnancy STEMI patients and in 35% of nonpregnant patients. In contrast, 10% of patients with a pregnancy-related STEMI underwent coronary artery bypass surgery, compared with 5% of nonpregnant women with a STEMI.
The PCI rate among women with a peripregnancy non-STEMI was 7.8%, compared with 28.7% in nonpregnant women with a non-STEMI. However, CABG was utilized less frequently in the peripregnancy non-STEMI group, by a margin of 4.4% to 5.9%.
Dr. Masoomi reported having no financial conflicts regarding his study.
ROME – It’s fortunate that pregnancy-associated acute MIs are infrequent, because the associated in-hospital mortality is markedly higher than in similar-age nonchildbearing women, Reza Masoomi, MD, said at the annual congress of the European Society of Cardiology.
One likely contributor to the disparity in outcome is that current management appears to feature underutilization of percutaneous intervention in women who experience pregnancy-associated MI, according to Dr. Masoomi of the University of Kansas in Kansas City.
He presented an analysis of the U.S. National Inpatient Sample database for the years 2008-2012. The NIS is a nationally representative sample of hospitalizations drawn from all of the country’s nonfederal acute-care hospitals.
A total of 55,315 hospitalizations with a discharge diagnosis of acute MI were recorded in women aged 15-54 years during the study years, of which 453 involved an ante- or postpartum MI. Extrapolating from those figures, nearly 262,000 women aged 15-54 years across the U.S. had an acute MI during the study years, of whom an estimated 2,153 experienced a pregnancy-associated MI.
In-hospital mortality among women with peripregnancy MI was 7.2%, significantly higher than the 5.2% rate in women who weren’t pregnant.
Women with peripregnancy MI had a significantly higher rate of ST-elevation MI (STEMI) than did nonpregnant women with MI in their reproductive years, by a margin of 35.3% to 32.8%. They were younger, too: an average age of 34.9 years, compared with 47.3 years in nonpregnant patients with an MI. Nearly two-thirds of women with peripregnancy MI were nonwhite, compared with 47.3% of the comparison group.
Regardless of whether women with peripregnancy MI had a STEMI or non-STEMI, they had significantly lower rates of diagnostic coronary angiography and percutaneous intervention. They were also far less likely to receive drug-eluting stents.
Diagnostic coronary angiography was performed in 59% of women with pregnancy-associated STEMI, compared with 73% of nonpregnant women with a STEMI. Only 34% of patients with peripregnancy STEMI underwent PCI, compared with 61% of nonpregnant women with a STEMI. Drug-eluting stents were implanted in 12% of peripregnancy STEMI patients and in 35% of nonpregnant patients. In contrast, 10% of patients with a pregnancy-related STEMI underwent coronary artery bypass surgery, compared with 5% of nonpregnant women with a STEMI.
The PCI rate among women with a peripregnancy non-STEMI was 7.8%, compared with 28.7% in nonpregnant women with a non-STEMI. However, CABG was utilized less frequently in the peripregnancy non-STEMI group, by a margin of 4.4% to 5.9%.
Dr. Masoomi reported having no financial conflicts regarding his study.
ROME – It’s fortunate that pregnancy-associated acute MIs are infrequent, because the associated in-hospital mortality is markedly higher than in similar-age nonchildbearing women, Reza Masoomi, MD, said at the annual congress of the European Society of Cardiology.
One likely contributor to the disparity in outcome is that current management appears to feature underutilization of percutaneous intervention in women who experience pregnancy-associated MI, according to Dr. Masoomi of the University of Kansas in Kansas City.
He presented an analysis of the U.S. National Inpatient Sample database for the years 2008-2012. The NIS is a nationally representative sample of hospitalizations drawn from all of the country’s nonfederal acute-care hospitals.
A total of 55,315 hospitalizations with a discharge diagnosis of acute MI were recorded in women aged 15-54 years during the study years, of which 453 involved an ante- or postpartum MI. Extrapolating from those figures, nearly 262,000 women aged 15-54 years across the U.S. had an acute MI during the study years, of whom an estimated 2,153 experienced a pregnancy-associated MI.
In-hospital mortality among women with peripregnancy MI was 7.2%, significantly higher than the 5.2% rate in women who weren’t pregnant.
Women with peripregnancy MI had a significantly higher rate of ST-elevation MI (STEMI) than did nonpregnant women with MI in their reproductive years, by a margin of 35.3% to 32.8%. They were younger, too: an average age of 34.9 years, compared with 47.3 years in nonpregnant patients with an MI. Nearly two-thirds of women with peripregnancy MI were nonwhite, compared with 47.3% of the comparison group.
Regardless of whether women with peripregnancy MI had a STEMI or non-STEMI, they had significantly lower rates of diagnostic coronary angiography and percutaneous intervention. They were also far less likely to receive drug-eluting stents.
Diagnostic coronary angiography was performed in 59% of women with pregnancy-associated STEMI, compared with 73% of nonpregnant women with a STEMI. Only 34% of patients with peripregnancy STEMI underwent PCI, compared with 61% of nonpregnant women with a STEMI. Drug-eluting stents were implanted in 12% of peripregnancy STEMI patients and in 35% of nonpregnant patients. In contrast, 10% of patients with a pregnancy-related STEMI underwent coronary artery bypass surgery, compared with 5% of nonpregnant women with a STEMI.
The PCI rate among women with a peripregnancy non-STEMI was 7.8%, compared with 28.7% in nonpregnant women with a non-STEMI. However, CABG was utilized less frequently in the peripregnancy non-STEMI group, by a margin of 4.4% to 5.9%.
Dr. Masoomi reported having no financial conflicts regarding his study.
Key clinical point:
Major finding: In-hospital mortality among U.S. women with peripregnancy MI was 7.2% during 2008-2012, significantly higher than the 5.2% rate in women of reproductive age who weren’t pregnant.
Data source: This analysis of data from the U.S. National Inpatient Sample concluded that of an estimated 261,806 U.S. women aged 15-54 years who had an acute MI during 2008-2012, a total of 2,153 of them had an ante- or postpartum-associated MI.
Disclosures: The study presenter reported having no financial conflicts of interest.
Bias rampant in major depression treatment literature
VIENNA – Janneke A. Bastiaansen, PhD, has some advice for clinicians and researchers as they peruse the published literature on clinical trials of medication or psychotherapy for major depressive disorder: Don’t believe everything you read.
“Be critical. Use your critical mind,” she urged at the annual congress of the European College of Neuropsychopharmacology.
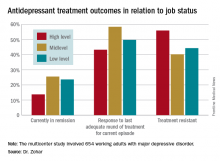
The results of her analysis of 105 clinical trials of antidepressant drugs and 142 studies of psychotherapy indicated that the literature is rife with four types of bias: publication, outcome reporting, spin, and citation bias.
She took a closer look at 105 clinical trials of antidepressant drugs registered with the U.S. National Institutes of Health at clinicaltrials.gov. Fifty-three reported positive findings, and 52 were negative. Fifty-two of the 53 positive trials were ultimately published, as were only 25 of the 52 negative studies. That’s a sterling example of publication bias.
Upon careful scrutiny of the 25 negative trials that were published, 10 were misleadingly reported as positive studies. The investigators either switched out the prespecified primary outcome previously filed with NIH and promoted a positive secondary outcome to primary outcome status because the original primary outcome was negative, or they omitted the negative outcomes altogether. That’s outcome-reporting bias.
Of the 15 published negative drug trials that were free of outcome-reporting bias, the authors of 10 of the studies employed “spin,” using phrases such as “the treatment was numerically superior.”
Thus, only 5 of the 25 published negative clinical trials unambiguously reported that the studied treatment was not effective.
“Here the message is that, when you read a paper, look at the results, come up with your own conclusion, and then compare it with the conclusion of the authors, because sometimes they’ve colored it in a more positive way,” Dr. Bastiaansen said in an interview.
Citation bias is the phenomenon whereby positive clinical trials are cited more frequently than published negative trials.
“As a clinician, if you look at the literature and print out every paper that’s out there on a given antidepressant drug for major depression, and you look at that pile, you’ll think: ‘Ah, the literature is really strong about this treatment effect,’ because positive papers selectively cite other positive papers,” she continued.
The pharmaceutical industry takes a lot of heat for selectively burying company-sponsored negative trials, but the literature on psychotherapy for major depression is actually more opaque.
“A lot of people aim their arrows at the pharmaceutical industry and say: ‘Everything’s bad about pharma,’ but actually, you see bias in every field. You see it in the trials of psychotherapy. It’s very important to know that it’s ubiquitous. The positive side of the antidepressant drug trials is that there’s this standard database [clinicaltrials.gov], and you can use it to check out what trial is published and what’s not. It’s not the case for psychotherapy trials. I think we need a mandatory registry for clinical trials of psychotherapy as well,” Dr. Bastiaansen said.
Of the 142 psychotherapy studies, 49 were negative, but the abstracts of only 12 of those 49 concluded that psychotherapy was not more effective than a control.
Dr. Bastiaansen declared having no financial conflicts regarding her university-funded study.
bjancin@frontlinemedcom.com
VIENNA – Janneke A. Bastiaansen, PhD, has some advice for clinicians and researchers as they peruse the published literature on clinical trials of medication or psychotherapy for major depressive disorder: Don’t believe everything you read.
“Be critical. Use your critical mind,” she urged at the annual congress of the European College of Neuropsychopharmacology.
The results of her analysis of 105 clinical trials of antidepressant drugs and 142 studies of psychotherapy indicated that the literature is rife with four types of bias: publication, outcome reporting, spin, and citation bias.
She took a closer look at 105 clinical trials of antidepressant drugs registered with the U.S. National Institutes of Health at clinicaltrials.gov. Fifty-three reported positive findings, and 52 were negative. Fifty-two of the 53 positive trials were ultimately published, as were only 25 of the 52 negative studies. That’s a sterling example of publication bias.
Upon careful scrutiny of the 25 negative trials that were published, 10 were misleadingly reported as positive studies. The investigators either switched out the prespecified primary outcome previously filed with NIH and promoted a positive secondary outcome to primary outcome status because the original primary outcome was negative, or they omitted the negative outcomes altogether. That’s outcome-reporting bias.
Of the 15 published negative drug trials that were free of outcome-reporting bias, the authors of 10 of the studies employed “spin,” using phrases such as “the treatment was numerically superior.”
Thus, only 5 of the 25 published negative clinical trials unambiguously reported that the studied treatment was not effective.
“Here the message is that, when you read a paper, look at the results, come up with your own conclusion, and then compare it with the conclusion of the authors, because sometimes they’ve colored it in a more positive way,” Dr. Bastiaansen said in an interview.
Citation bias is the phenomenon whereby positive clinical trials are cited more frequently than published negative trials.
“As a clinician, if you look at the literature and print out every paper that’s out there on a given antidepressant drug for major depression, and you look at that pile, you’ll think: ‘Ah, the literature is really strong about this treatment effect,’ because positive papers selectively cite other positive papers,” she continued.
The pharmaceutical industry takes a lot of heat for selectively burying company-sponsored negative trials, but the literature on psychotherapy for major depression is actually more opaque.
“A lot of people aim their arrows at the pharmaceutical industry and say: ‘Everything’s bad about pharma,’ but actually, you see bias in every field. You see it in the trials of psychotherapy. It’s very important to know that it’s ubiquitous. The positive side of the antidepressant drug trials is that there’s this standard database [clinicaltrials.gov], and you can use it to check out what trial is published and what’s not. It’s not the case for psychotherapy trials. I think we need a mandatory registry for clinical trials of psychotherapy as well,” Dr. Bastiaansen said.
Of the 142 psychotherapy studies, 49 were negative, but the abstracts of only 12 of those 49 concluded that psychotherapy was not more effective than a control.
Dr. Bastiaansen declared having no financial conflicts regarding her university-funded study.
bjancin@frontlinemedcom.com
VIENNA – Janneke A. Bastiaansen, PhD, has some advice for clinicians and researchers as they peruse the published literature on clinical trials of medication or psychotherapy for major depressive disorder: Don’t believe everything you read.
“Be critical. Use your critical mind,” she urged at the annual congress of the European College of Neuropsychopharmacology.
The results of her analysis of 105 clinical trials of antidepressant drugs and 142 studies of psychotherapy indicated that the literature is rife with four types of bias: publication, outcome reporting, spin, and citation bias.
She took a closer look at 105 clinical trials of antidepressant drugs registered with the U.S. National Institutes of Health at clinicaltrials.gov. Fifty-three reported positive findings, and 52 were negative. Fifty-two of the 53 positive trials were ultimately published, as were only 25 of the 52 negative studies. That’s a sterling example of publication bias.
Upon careful scrutiny of the 25 negative trials that were published, 10 were misleadingly reported as positive studies. The investigators either switched out the prespecified primary outcome previously filed with NIH and promoted a positive secondary outcome to primary outcome status because the original primary outcome was negative, or they omitted the negative outcomes altogether. That’s outcome-reporting bias.
Of the 15 published negative drug trials that were free of outcome-reporting bias, the authors of 10 of the studies employed “spin,” using phrases such as “the treatment was numerically superior.”
Thus, only 5 of the 25 published negative clinical trials unambiguously reported that the studied treatment was not effective.
“Here the message is that, when you read a paper, look at the results, come up with your own conclusion, and then compare it with the conclusion of the authors, because sometimes they’ve colored it in a more positive way,” Dr. Bastiaansen said in an interview.
Citation bias is the phenomenon whereby positive clinical trials are cited more frequently than published negative trials.
“As a clinician, if you look at the literature and print out every paper that’s out there on a given antidepressant drug for major depression, and you look at that pile, you’ll think: ‘Ah, the literature is really strong about this treatment effect,’ because positive papers selectively cite other positive papers,” she continued.
The pharmaceutical industry takes a lot of heat for selectively burying company-sponsored negative trials, but the literature on psychotherapy for major depression is actually more opaque.
“A lot of people aim their arrows at the pharmaceutical industry and say: ‘Everything’s bad about pharma,’ but actually, you see bias in every field. You see it in the trials of psychotherapy. It’s very important to know that it’s ubiquitous. The positive side of the antidepressant drug trials is that there’s this standard database [clinicaltrials.gov], and you can use it to check out what trial is published and what’s not. It’s not the case for psychotherapy trials. I think we need a mandatory registry for clinical trials of psychotherapy as well,” Dr. Bastiaansen said.
Of the 142 psychotherapy studies, 49 were negative, but the abstracts of only 12 of those 49 concluded that psychotherapy was not more effective than a control.
Dr. Bastiaansen declared having no financial conflicts regarding her university-funded study.
bjancin@frontlinemedcom.com
Key clinical point:
Major finding: Only 5 of 25 published negative clinical trials of antidepressant drug therapy unambiguously reported that the studied treatment was not effective.
Data source: For this study, 105 clinical trials of antidepressant drugs and 142 on psychotherapy for major depressive disorder were examined systematically for evidence of four types of bias that damage the quality of the evidence base.
Disclosures: The presenter reported having no financial conflicts of interest regarding her university-funded study.
Finger length ratio identifies women at increased risk for depression and anxiety
VIENNA – They say that in hula dancing, it’s the expressive hands, not the quaking hips, that tell the story.
And in Dutch women, a relatively short index finger on the left hand bespeaks an increased risk for depression and stress.
That’s right: The ratio of the length of the index finger to the ring finger, or 2D:4D digit ratio, of the left hand shows potential as a quick and dirty biomarker that could be used to screen patients for increased risk for depression. But only in women, Deborah De Kruijff reported at the annual congress of the European College of Neuropsychopharmacology.
She and her coinvestigators measured the lengths of the index and ring fingers on both hands of 124 male and 146 female Dutch college students using Vernier calipers accurate to within 0.01 mm. Participants completed the 21-item version of the Depression, Anxiety, and Stress Scale (DASS-21) and correlated the 2D:4D digit ratios with the DASS-21 total scores as well as the scores on the depression, anxiety, and stress subscales.
The 2D:4D digit ratio didn’t correlate with DASS-21 scores in men. But in women, the lower the 2D:4D ratio on the left hand, the higher their overall DASS-21 score as well as their scores on the depression and stress subscales. Each of these associations was highly statistically significant at the P = .002 to .005 level, according to Ms. De Kruijff, a PhD candidate in neuroscience at Utrecht (the Netherlands) University.
Finding correlates between the 2D:4D digit ratio and predispositions to various diseases, personality traits, and other human characteristics was a popular scientific pastime in the 1800s. After a long dry spell, it rebounded as a research area several decades ago. The 2D:4D ratio is a sexually dimorphic trait. It is thought to depend upon prenatal exposure to sex hormones. A low 2D:4D ratio is associated with in utero exposure to relatively higher levels of fetal testosterone than fetal estrogen. Thus, a greater proportion of men than women have index fingers that are shorter than the ring finger.
Other investigators have linked a low 2D:4D ratio to increased risks of prostate cancer, attention-deficit/hyperactivity disorder, and autism spectrum disorder in men, and to greater assertiveness and increased risk of anorexia nervosa in women.
Ms. De Kruijff said more research is needed to understand why only the finger length on the left hand of the women was predictive of increased risk of depression and stress.
She reported having no financial conflicts of interest regarding this university-funded study.
VIENNA – They say that in hula dancing, it’s the expressive hands, not the quaking hips, that tell the story.
And in Dutch women, a relatively short index finger on the left hand bespeaks an increased risk for depression and stress.
That’s right: The ratio of the length of the index finger to the ring finger, or 2D:4D digit ratio, of the left hand shows potential as a quick and dirty biomarker that could be used to screen patients for increased risk for depression. But only in women, Deborah De Kruijff reported at the annual congress of the European College of Neuropsychopharmacology.
She and her coinvestigators measured the lengths of the index and ring fingers on both hands of 124 male and 146 female Dutch college students using Vernier calipers accurate to within 0.01 mm. Participants completed the 21-item version of the Depression, Anxiety, and Stress Scale (DASS-21) and correlated the 2D:4D digit ratios with the DASS-21 total scores as well as the scores on the depression, anxiety, and stress subscales.
The 2D:4D digit ratio didn’t correlate with DASS-21 scores in men. But in women, the lower the 2D:4D ratio on the left hand, the higher their overall DASS-21 score as well as their scores on the depression and stress subscales. Each of these associations was highly statistically significant at the P = .002 to .005 level, according to Ms. De Kruijff, a PhD candidate in neuroscience at Utrecht (the Netherlands) University.
Finding correlates between the 2D:4D digit ratio and predispositions to various diseases, personality traits, and other human characteristics was a popular scientific pastime in the 1800s. After a long dry spell, it rebounded as a research area several decades ago. The 2D:4D ratio is a sexually dimorphic trait. It is thought to depend upon prenatal exposure to sex hormones. A low 2D:4D ratio is associated with in utero exposure to relatively higher levels of fetal testosterone than fetal estrogen. Thus, a greater proportion of men than women have index fingers that are shorter than the ring finger.
Other investigators have linked a low 2D:4D ratio to increased risks of prostate cancer, attention-deficit/hyperactivity disorder, and autism spectrum disorder in men, and to greater assertiveness and increased risk of anorexia nervosa in women.
Ms. De Kruijff said more research is needed to understand why only the finger length on the left hand of the women was predictive of increased risk of depression and stress.
She reported having no financial conflicts of interest regarding this university-funded study.
VIENNA – They say that in hula dancing, it’s the expressive hands, not the quaking hips, that tell the story.
And in Dutch women, a relatively short index finger on the left hand bespeaks an increased risk for depression and stress.
That’s right: The ratio of the length of the index finger to the ring finger, or 2D:4D digit ratio, of the left hand shows potential as a quick and dirty biomarker that could be used to screen patients for increased risk for depression. But only in women, Deborah De Kruijff reported at the annual congress of the European College of Neuropsychopharmacology.
She and her coinvestigators measured the lengths of the index and ring fingers on both hands of 124 male and 146 female Dutch college students using Vernier calipers accurate to within 0.01 mm. Participants completed the 21-item version of the Depression, Anxiety, and Stress Scale (DASS-21) and correlated the 2D:4D digit ratios with the DASS-21 total scores as well as the scores on the depression, anxiety, and stress subscales.
The 2D:4D digit ratio didn’t correlate with DASS-21 scores in men. But in women, the lower the 2D:4D ratio on the left hand, the higher their overall DASS-21 score as well as their scores on the depression and stress subscales. Each of these associations was highly statistically significant at the P = .002 to .005 level, according to Ms. De Kruijff, a PhD candidate in neuroscience at Utrecht (the Netherlands) University.
Finding correlates between the 2D:4D digit ratio and predispositions to various diseases, personality traits, and other human characteristics was a popular scientific pastime in the 1800s. After a long dry spell, it rebounded as a research area several decades ago. The 2D:4D ratio is a sexually dimorphic trait. It is thought to depend upon prenatal exposure to sex hormones. A low 2D:4D ratio is associated with in utero exposure to relatively higher levels of fetal testosterone than fetal estrogen. Thus, a greater proportion of men than women have index fingers that are shorter than the ring finger.
Other investigators have linked a low 2D:4D ratio to increased risks of prostate cancer, attention-deficit/hyperactivity disorder, and autism spectrum disorder in men, and to greater assertiveness and increased risk of anorexia nervosa in women.
Ms. De Kruijff said more research is needed to understand why only the finger length on the left hand of the women was predictive of increased risk of depression and stress.
She reported having no financial conflicts of interest regarding this university-funded study.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: The lower the ratio of the length of the index finger to the ring finger on the left hand in women, the higher they scored on a validated measure of depression and stress.
Data source: A cross-sectional study involving 124 male and 146 female university students who completed the 21-item version of the Depression, Anxiety, and Stress Scale and were measured for the lengths of their index and ring fingers on both hands.
Disclosures: The presenter reported having no financial conflicts of interest regarding this university-funded study.
Early change in emotional processing predicts antidepressant response
VIENNA – A novel method of individualizing antidepressant drug therapy while drastically shortening the time required to figure out whether a given agent will be effective in a depressed patient is undergoing its definitive evaluation in five European countries.
“I think this study will provide a critical test of whether we can use these kinds of correlations with emotional processing of information to actually improve the treatment of depression,” Catherine J. Harmer, DPhil, said at the annual congress of the European College of Neuropsychopharmacology.
As early as 2009, she and her coinvestigators demonstrated that depression is associated with a measurable negative bias in the processing of emotional information. Depressed patients selectively pay more attention to and better remember negative information. For example, when depressed patients taking the Facial Expression Recognition Test are quickly shown a photo of a smiling face, they are more likely to describe it as “sad.” Similarly, in a word recall test that includes “positive” adjectives such as cheerful, poised, original, and optimistic and “negative” words such as mean, hostile, domineering, and untidy, they recall fewer positive words than nondepressed people.
This negative emotional bias is a key factor in maintenance of depression. Many weeks before patients report feeling improvement in their mood and clinical symptoms of depression in response to effective antidepressant medication, the drug produces a favorable effect on their cognitive biases in emotional processing, explained Dr. Harmer, professor of cognitive neuroscience and director of the Psychopharmacology and Emotional Research Lab at the University of Oxford (England).
Dr. Harmer hypothesized that antidepressants don’t necessarily act as direct mood enhancers, but instead change the balance away from negative to more positive emotional processing, resulting in neural modulation in limbic and prefrontal circuitry. These neural changes take time to reach the subjective conscious mind, which is why improvement in clinical symptoms of depression doesn’t manifest until 2-3 weeks into therapy, with the drug’s full effects not seen until 6-7 weeks (Br J Psychiatry. 2009 Aug;195[2]:102-8).
“Antidepressants can target cognitive biases surprisingly early on in treatment, before patients report any change in their clinical symptoms. This could explain the delay in antidepressant effect. You need life events, stressors, and environmental stimuli before a change in bias would be expected to result in a change in clinical state,” she said.
Together with her colleagues, she employed functional MRI to study changes in the brain associated with the improvement in negative biases in emotional processing that occur when depressed patients go on antidepressant medication. Forty-two unmedicated depressed patients were randomized to 10 mg of escitalopram (Lexapro) daily for 7 days or placebo. At baseline, all subjects demonstrated amygdala hyperactivity in response to fearful facial expressions, a response that didn’t occur in healthy controls. After 7 days of escitalopram – weeks before any improvement in depressed mood – amygdala activity was normalized in the active treatment group but not in placebo-treated controls (Psychol Med. 2012 Dec;42[12]:2609-17).
Dr. Harmer and a colleague expanded on her theory of depression in a review article aptly titled, “It’s the way that you look at it” (Philos Trans R Soc Lond B Biol Sci. 2013 Feb 25;368[1615]:20120407).
A measurable improvement in emotional processing can be seen within a few hours after a depressed patient takes the first dose of an effective antidepressant. Thus, early change in negative emotional processing is predictive of subsequent clinical outcome. Lack of an early shift to positive emotional processing has been associated in multiple studies by Dr. Harmer and others with a high likelihood that an antidepressant won’t provide significant improvement in depressive symptoms at week 6.
The predictive accuracy of tests of emotional processing is higher when testing is done after a patient has been on an antidepressant medication for a few days rather than after the very first dose. Based upon Dr. Harmer’s work, pharmaceutical companies are now using tests of change in emotional processing at 1 week to help screen and select novel treatments for depression and anxiety.
In PReDicT, depressed patients being treated in primary care clinics across Europe will undergo emotional processing testing at baseline. In the active intervention arm, participants will be retested after 1 week on antidepressant therapy in order to identify those who are unlikely to have a favorable clinical response to that drug, enabling physicians to accelerate decision making about the appropriate next treatment. Treatment decisions in the control group will be made without the emotional processing results, mirroring current everyday practice.
Instead of waiting 4-6 weeks before concluding that a switch to another antidepressant with a different mechanism of action is warranted, as is now routine, participating PReDicT physicians whose patients are in the active intervention arm can make an informed change after just 1 week. The study hypothesis is that participants randomized to this study arm will take less time to respond to antidepressant therapy, because their physicians will be able to find the right drug faster than in the control group. The primary study endpoint will be the percentage of patients in the two study arms showing at least a 50% reduction in their Quick Inventory of Depressive Symptomatology (QIDS SR-16) score at 8 weeks. Secondary endpoints will focus on cumulative health care costs at weeks 24 and 48.
The PReDicT study is being run by P1vital Products Ltd., an Oxfordshire health care company that will use its proprietary Internet-based eHealth Emotional Test Battery to assess early changes in emotional processing. The test battery, which is classified as a medical device, is based upon Dr. Harmer’s earlier work. She is an investigator in PReDicT.
Her research funding comes from the U.K. Medical Research Council, Eli Lilly, and the EU’s Horizon 2020 program.
VIENNA – A novel method of individualizing antidepressant drug therapy while drastically shortening the time required to figure out whether a given agent will be effective in a depressed patient is undergoing its definitive evaluation in five European countries.
“I think this study will provide a critical test of whether we can use these kinds of correlations with emotional processing of information to actually improve the treatment of depression,” Catherine J. Harmer, DPhil, said at the annual congress of the European College of Neuropsychopharmacology.
As early as 2009, she and her coinvestigators demonstrated that depression is associated with a measurable negative bias in the processing of emotional information. Depressed patients selectively pay more attention to and better remember negative information. For example, when depressed patients taking the Facial Expression Recognition Test are quickly shown a photo of a smiling face, they are more likely to describe it as “sad.” Similarly, in a word recall test that includes “positive” adjectives such as cheerful, poised, original, and optimistic and “negative” words such as mean, hostile, domineering, and untidy, they recall fewer positive words than nondepressed people.
This negative emotional bias is a key factor in maintenance of depression. Many weeks before patients report feeling improvement in their mood and clinical symptoms of depression in response to effective antidepressant medication, the drug produces a favorable effect on their cognitive biases in emotional processing, explained Dr. Harmer, professor of cognitive neuroscience and director of the Psychopharmacology and Emotional Research Lab at the University of Oxford (England).
Dr. Harmer hypothesized that antidepressants don’t necessarily act as direct mood enhancers, but instead change the balance away from negative to more positive emotional processing, resulting in neural modulation in limbic and prefrontal circuitry. These neural changes take time to reach the subjective conscious mind, which is why improvement in clinical symptoms of depression doesn’t manifest until 2-3 weeks into therapy, with the drug’s full effects not seen until 6-7 weeks (Br J Psychiatry. 2009 Aug;195[2]:102-8).
“Antidepressants can target cognitive biases surprisingly early on in treatment, before patients report any change in their clinical symptoms. This could explain the delay in antidepressant effect. You need life events, stressors, and environmental stimuli before a change in bias would be expected to result in a change in clinical state,” she said.
Together with her colleagues, she employed functional MRI to study changes in the brain associated with the improvement in negative biases in emotional processing that occur when depressed patients go on antidepressant medication. Forty-two unmedicated depressed patients were randomized to 10 mg of escitalopram (Lexapro) daily for 7 days or placebo. At baseline, all subjects demonstrated amygdala hyperactivity in response to fearful facial expressions, a response that didn’t occur in healthy controls. After 7 days of escitalopram – weeks before any improvement in depressed mood – amygdala activity was normalized in the active treatment group but not in placebo-treated controls (Psychol Med. 2012 Dec;42[12]:2609-17).
Dr. Harmer and a colleague expanded on her theory of depression in a review article aptly titled, “It’s the way that you look at it” (Philos Trans R Soc Lond B Biol Sci. 2013 Feb 25;368[1615]:20120407).
A measurable improvement in emotional processing can be seen within a few hours after a depressed patient takes the first dose of an effective antidepressant. Thus, early change in negative emotional processing is predictive of subsequent clinical outcome. Lack of an early shift to positive emotional processing has been associated in multiple studies by Dr. Harmer and others with a high likelihood that an antidepressant won’t provide significant improvement in depressive symptoms at week 6.
The predictive accuracy of tests of emotional processing is higher when testing is done after a patient has been on an antidepressant medication for a few days rather than after the very first dose. Based upon Dr. Harmer’s work, pharmaceutical companies are now using tests of change in emotional processing at 1 week to help screen and select novel treatments for depression and anxiety.
In PReDicT, depressed patients being treated in primary care clinics across Europe will undergo emotional processing testing at baseline. In the active intervention arm, participants will be retested after 1 week on antidepressant therapy in order to identify those who are unlikely to have a favorable clinical response to that drug, enabling physicians to accelerate decision making about the appropriate next treatment. Treatment decisions in the control group will be made without the emotional processing results, mirroring current everyday practice.
Instead of waiting 4-6 weeks before concluding that a switch to another antidepressant with a different mechanism of action is warranted, as is now routine, participating PReDicT physicians whose patients are in the active intervention arm can make an informed change after just 1 week. The study hypothesis is that participants randomized to this study arm will take less time to respond to antidepressant therapy, because their physicians will be able to find the right drug faster than in the control group. The primary study endpoint will be the percentage of patients in the two study arms showing at least a 50% reduction in their Quick Inventory of Depressive Symptomatology (QIDS SR-16) score at 8 weeks. Secondary endpoints will focus on cumulative health care costs at weeks 24 and 48.
The PReDicT study is being run by P1vital Products Ltd., an Oxfordshire health care company that will use its proprietary Internet-based eHealth Emotional Test Battery to assess early changes in emotional processing. The test battery, which is classified as a medical device, is based upon Dr. Harmer’s earlier work. She is an investigator in PReDicT.
Her research funding comes from the U.K. Medical Research Council, Eli Lilly, and the EU’s Horizon 2020 program.
VIENNA – A novel method of individualizing antidepressant drug therapy while drastically shortening the time required to figure out whether a given agent will be effective in a depressed patient is undergoing its definitive evaluation in five European countries.
“I think this study will provide a critical test of whether we can use these kinds of correlations with emotional processing of information to actually improve the treatment of depression,” Catherine J. Harmer, DPhil, said at the annual congress of the European College of Neuropsychopharmacology.
As early as 2009, she and her coinvestigators demonstrated that depression is associated with a measurable negative bias in the processing of emotional information. Depressed patients selectively pay more attention to and better remember negative information. For example, when depressed patients taking the Facial Expression Recognition Test are quickly shown a photo of a smiling face, they are more likely to describe it as “sad.” Similarly, in a word recall test that includes “positive” adjectives such as cheerful, poised, original, and optimistic and “negative” words such as mean, hostile, domineering, and untidy, they recall fewer positive words than nondepressed people.
This negative emotional bias is a key factor in maintenance of depression. Many weeks before patients report feeling improvement in their mood and clinical symptoms of depression in response to effective antidepressant medication, the drug produces a favorable effect on their cognitive biases in emotional processing, explained Dr. Harmer, professor of cognitive neuroscience and director of the Psychopharmacology and Emotional Research Lab at the University of Oxford (England).
Dr. Harmer hypothesized that antidepressants don’t necessarily act as direct mood enhancers, but instead change the balance away from negative to more positive emotional processing, resulting in neural modulation in limbic and prefrontal circuitry. These neural changes take time to reach the subjective conscious mind, which is why improvement in clinical symptoms of depression doesn’t manifest until 2-3 weeks into therapy, with the drug’s full effects not seen until 6-7 weeks (Br J Psychiatry. 2009 Aug;195[2]:102-8).
“Antidepressants can target cognitive biases surprisingly early on in treatment, before patients report any change in their clinical symptoms. This could explain the delay in antidepressant effect. You need life events, stressors, and environmental stimuli before a change in bias would be expected to result in a change in clinical state,” she said.
Together with her colleagues, she employed functional MRI to study changes in the brain associated with the improvement in negative biases in emotional processing that occur when depressed patients go on antidepressant medication. Forty-two unmedicated depressed patients were randomized to 10 mg of escitalopram (Lexapro) daily for 7 days or placebo. At baseline, all subjects demonstrated amygdala hyperactivity in response to fearful facial expressions, a response that didn’t occur in healthy controls. After 7 days of escitalopram – weeks before any improvement in depressed mood – amygdala activity was normalized in the active treatment group but not in placebo-treated controls (Psychol Med. 2012 Dec;42[12]:2609-17).
Dr. Harmer and a colleague expanded on her theory of depression in a review article aptly titled, “It’s the way that you look at it” (Philos Trans R Soc Lond B Biol Sci. 2013 Feb 25;368[1615]:20120407).
A measurable improvement in emotional processing can be seen within a few hours after a depressed patient takes the first dose of an effective antidepressant. Thus, early change in negative emotional processing is predictive of subsequent clinical outcome. Lack of an early shift to positive emotional processing has been associated in multiple studies by Dr. Harmer and others with a high likelihood that an antidepressant won’t provide significant improvement in depressive symptoms at week 6.
The predictive accuracy of tests of emotional processing is higher when testing is done after a patient has been on an antidepressant medication for a few days rather than after the very first dose. Based upon Dr. Harmer’s work, pharmaceutical companies are now using tests of change in emotional processing at 1 week to help screen and select novel treatments for depression and anxiety.
In PReDicT, depressed patients being treated in primary care clinics across Europe will undergo emotional processing testing at baseline. In the active intervention arm, participants will be retested after 1 week on antidepressant therapy in order to identify those who are unlikely to have a favorable clinical response to that drug, enabling physicians to accelerate decision making about the appropriate next treatment. Treatment decisions in the control group will be made without the emotional processing results, mirroring current everyday practice.
Instead of waiting 4-6 weeks before concluding that a switch to another antidepressant with a different mechanism of action is warranted, as is now routine, participating PReDicT physicians whose patients are in the active intervention arm can make an informed change after just 1 week. The study hypothesis is that participants randomized to this study arm will take less time to respond to antidepressant therapy, because their physicians will be able to find the right drug faster than in the control group. The primary study endpoint will be the percentage of patients in the two study arms showing at least a 50% reduction in their Quick Inventory of Depressive Symptomatology (QIDS SR-16) score at 8 weeks. Secondary endpoints will focus on cumulative health care costs at weeks 24 and 48.
The PReDicT study is being run by P1vital Products Ltd., an Oxfordshire health care company that will use its proprietary Internet-based eHealth Emotional Test Battery to assess early changes in emotional processing. The test battery, which is classified as a medical device, is based upon Dr. Harmer’s earlier work. She is an investigator in PReDicT.
Her research funding comes from the U.K. Medical Research Council, Eli Lilly, and the EU’s Horizon 2020 program.
High-status occupation spells worse response to antidepressant therapy
VIENNA – Depressed patients with a high-status job appear to be significantly less responsive to antidepressant therapy than other workers, Joseph Zohar, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
He presented a multicenter study of 654 Austrian, Israeli, Belgian, and Italian working adults with major depressive disorder. Those categorized as having a high occupational level were significantly less likely to have responded to the most recent trial of antidepressant medication for their current episode. They also had a significantly higher rate of treatment-resistant depression as defined by nonresponse to at least two previous adequate treatment trials, compared with the patients with mid- or low-level occupations.
“These results show that the need for precise prescribing is not only related to the symptoms and genetics but also to occupational level. One might need to prescribe different medication for the same disorder and need to take into account the occupational level in order to reach optimum effect,” he continued.
He and his coinvestigators categorized occupational level according to Hollingshead’s Occupational Scale, a tool widely used by sociologists. Occupational level is a different concept from socioeconomic status. It incorporates prestige, entry qualifications, job responsibilities, and skill requirements – as well as earnings. Examples of high occupational level jobs in this construct include physicians, engineers, architects, scientists, lawyers, CEOs, and professors.
Fifty-one percent of the study participants were classified as having high-status occupations, with the rest evenly divided between mid- and low-level occupations. Only 43.2% of patients in the high-level group had responded to their last treatment episode based upon clinical judgment and a Hamilton Depression Rating Scale score of 17 or less, compared with 58.4% of patients with mid- and 49.7% with low-level jobs.
At this point the explanation for the observed relationship between occupation level and antidepressant treatment response remains speculative. This is the first large study of its kind, and the results require confirmation. If the findings hold up, the results could have important implications for employers, who might want to discourage patients with high-status jobs and a history of treatment-resistant depression from particularly high work stress environments in order to reduce depression-related disability in the workplace, according to the psychiatrist.
Potential explanations for the findings include the possibility that depressed patients in high-level occupations are more likely to have poor illness acceptance, be less adherent to prescribed medications, or possess certain personality, cognitive, or behavioral differences that predispose to poor treatment outcome.
The study was funded by the ECNP’s Expert Platform on Mental Health – Focus on Depression. Dr. Zohar reported having no relevant financial conflicts.
VIENNA – Depressed patients with a high-status job appear to be significantly less responsive to antidepressant therapy than other workers, Joseph Zohar, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
He presented a multicenter study of 654 Austrian, Israeli, Belgian, and Italian working adults with major depressive disorder. Those categorized as having a high occupational level were significantly less likely to have responded to the most recent trial of antidepressant medication for their current episode. They also had a significantly higher rate of treatment-resistant depression as defined by nonresponse to at least two previous adequate treatment trials, compared with the patients with mid- or low-level occupations.
“These results show that the need for precise prescribing is not only related to the symptoms and genetics but also to occupational level. One might need to prescribe different medication for the same disorder and need to take into account the occupational level in order to reach optimum effect,” he continued.
He and his coinvestigators categorized occupational level according to Hollingshead’s Occupational Scale, a tool widely used by sociologists. Occupational level is a different concept from socioeconomic status. It incorporates prestige, entry qualifications, job responsibilities, and skill requirements – as well as earnings. Examples of high occupational level jobs in this construct include physicians, engineers, architects, scientists, lawyers, CEOs, and professors.
Fifty-one percent of the study participants were classified as having high-status occupations, with the rest evenly divided between mid- and low-level occupations. Only 43.2% of patients in the high-level group had responded to their last treatment episode based upon clinical judgment and a Hamilton Depression Rating Scale score of 17 or less, compared with 58.4% of patients with mid- and 49.7% with low-level jobs.
At this point the explanation for the observed relationship between occupation level and antidepressant treatment response remains speculative. This is the first large study of its kind, and the results require confirmation. If the findings hold up, the results could have important implications for employers, who might want to discourage patients with high-status jobs and a history of treatment-resistant depression from particularly high work stress environments in order to reduce depression-related disability in the workplace, according to the psychiatrist.
Potential explanations for the findings include the possibility that depressed patients in high-level occupations are more likely to have poor illness acceptance, be less adherent to prescribed medications, or possess certain personality, cognitive, or behavioral differences that predispose to poor treatment outcome.
The study was funded by the ECNP’s Expert Platform on Mental Health – Focus on Depression. Dr. Zohar reported having no relevant financial conflicts.
VIENNA – Depressed patients with a high-status job appear to be significantly less responsive to antidepressant therapy than other workers, Joseph Zohar, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
He presented a multicenter study of 654 Austrian, Israeli, Belgian, and Italian working adults with major depressive disorder. Those categorized as having a high occupational level were significantly less likely to have responded to the most recent trial of antidepressant medication for their current episode. They also had a significantly higher rate of treatment-resistant depression as defined by nonresponse to at least two previous adequate treatment trials, compared with the patients with mid- or low-level occupations.
“These results show that the need for precise prescribing is not only related to the symptoms and genetics but also to occupational level. One might need to prescribe different medication for the same disorder and need to take into account the occupational level in order to reach optimum effect,” he continued.
He and his coinvestigators categorized occupational level according to Hollingshead’s Occupational Scale, a tool widely used by sociologists. Occupational level is a different concept from socioeconomic status. It incorporates prestige, entry qualifications, job responsibilities, and skill requirements – as well as earnings. Examples of high occupational level jobs in this construct include physicians, engineers, architects, scientists, lawyers, CEOs, and professors.
Fifty-one percent of the study participants were classified as having high-status occupations, with the rest evenly divided between mid- and low-level occupations. Only 43.2% of patients in the high-level group had responded to their last treatment episode based upon clinical judgment and a Hamilton Depression Rating Scale score of 17 or less, compared with 58.4% of patients with mid- and 49.7% with low-level jobs.
At this point the explanation for the observed relationship between occupation level and antidepressant treatment response remains speculative. This is the first large study of its kind, and the results require confirmation. If the findings hold up, the results could have important implications for employers, who might want to discourage patients with high-status jobs and a history of treatment-resistant depression from particularly high work stress environments in order to reduce depression-related disability in the workplace, according to the psychiatrist.
Potential explanations for the findings include the possibility that depressed patients in high-level occupations are more likely to have poor illness acceptance, be less adherent to prescribed medications, or possess certain personality, cognitive, or behavioral differences that predispose to poor treatment outcome.
The study was funded by the ECNP’s Expert Platform on Mental Health – Focus on Depression. Dr. Zohar reported having no relevant financial conflicts.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Fifty-six percent of patients with high-status occupations who were treated for major depression were classified as treatment resistant, compared with 40% of patients with midlevel occupations and 44% with low-level occupations.
Data source: A study of 654 working adults with major depression in four countries.
Disclosures: The study was funded by the Expert Platform on Mental Health Focus on Depression. The presenter reported having no relevant financial conflicts.
Children on antipsychotics show more early weight gain than adults
VIENNA – Antipsychotic-naive children and adolescents are at greater risk for clinically significant weight gain during their first 6 months on a second-generation antipsychotic agent than adults, Covadonga M. Diaz-Caneja, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
She presented what she believes to be the first prospective observational study comparing weight gain between antipsychotic-naive children and adults during their first 6 months on second-generation antipsychotics. Weight gains were significantly greater with olanzapine (Zyprexa) than risperidone (Risperdal) or quetiapine (Seroquel) in both age groups, but the magnitude and trajectory of olanzapine-related weight gain was significantly more impressive in the pediatric patients.
The study included 226 pediatric patients with a mean age of 15.2 years and 135 adults, average age 39 years. All were antipsychotic naive when placed on one of the three second-generation antipsychotics at clinician discretion. Fifty percent of the youths and 60% of adults had a psychosis diagnosis. Sixty-four percent of the pediatric group and 55% of the adults were male.
Weight gain during the first 6 months of therapy was expressed as change in body mass index z score. An increase in BMI z score of 0.5 or more is considered clinically significant.
The least weight gain at 6 months occurred with quetiapine in both age groups. However, the BMI z score increased by 0.532 in the pediatric patients on the drug, more than twice the 0.236 increase in adults.
Risperidone was the only second-generation antipsychotic where the weight gain trajectories in pediatric and adult patients were not significantly different.
The study was funded by the Spanish Ministry of Economy and Competitiveness, and the European Commission. Dr. Diaz-Caneja reported having no financial conflicts of interest.
VIENNA – Antipsychotic-naive children and adolescents are at greater risk for clinically significant weight gain during their first 6 months on a second-generation antipsychotic agent than adults, Covadonga M. Diaz-Caneja, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
She presented what she believes to be the first prospective observational study comparing weight gain between antipsychotic-naive children and adults during their first 6 months on second-generation antipsychotics. Weight gains were significantly greater with olanzapine (Zyprexa) than risperidone (Risperdal) or quetiapine (Seroquel) in both age groups, but the magnitude and trajectory of olanzapine-related weight gain was significantly more impressive in the pediatric patients.
The study included 226 pediatric patients with a mean age of 15.2 years and 135 adults, average age 39 years. All were antipsychotic naive when placed on one of the three second-generation antipsychotics at clinician discretion. Fifty percent of the youths and 60% of adults had a psychosis diagnosis. Sixty-four percent of the pediatric group and 55% of the adults were male.
Weight gain during the first 6 months of therapy was expressed as change in body mass index z score. An increase in BMI z score of 0.5 or more is considered clinically significant.
The least weight gain at 6 months occurred with quetiapine in both age groups. However, the BMI z score increased by 0.532 in the pediatric patients on the drug, more than twice the 0.236 increase in adults.
Risperidone was the only second-generation antipsychotic where the weight gain trajectories in pediatric and adult patients were not significantly different.
The study was funded by the Spanish Ministry of Economy and Competitiveness, and the European Commission. Dr. Diaz-Caneja reported having no financial conflicts of interest.
VIENNA – Antipsychotic-naive children and adolescents are at greater risk for clinically significant weight gain during their first 6 months on a second-generation antipsychotic agent than adults, Covadonga M. Diaz-Caneja, MD, reported at the annual congress of the European College of Neuropsychopharmacology.
She presented what she believes to be the first prospective observational study comparing weight gain between antipsychotic-naive children and adults during their first 6 months on second-generation antipsychotics. Weight gains were significantly greater with olanzapine (Zyprexa) than risperidone (Risperdal) or quetiapine (Seroquel) in both age groups, but the magnitude and trajectory of olanzapine-related weight gain was significantly more impressive in the pediatric patients.
The study included 226 pediatric patients with a mean age of 15.2 years and 135 adults, average age 39 years. All were antipsychotic naive when placed on one of the three second-generation antipsychotics at clinician discretion. Fifty percent of the youths and 60% of adults had a psychosis diagnosis. Sixty-four percent of the pediatric group and 55% of the adults were male.
Weight gain during the first 6 months of therapy was expressed as change in body mass index z score. An increase in BMI z score of 0.5 or more is considered clinically significant.
The least weight gain at 6 months occurred with quetiapine in both age groups. However, the BMI z score increased by 0.532 in the pediatric patients on the drug, more than twice the 0.236 increase in adults.
Risperidone was the only second-generation antipsychotic where the weight gain trajectories in pediatric and adult patients were not significantly different.
The study was funded by the Spanish Ministry of Economy and Competitiveness, and the European Commission. Dr. Diaz-Caneja reported having no financial conflicts of interest.
Key clinical point:
Major finding: The BMI z score increased by 1.535 in pediatric patients during their first 6 months on olanzapine – the most weight unfriendly of the second-generation antipsychotics – compared with 0.724 in adults.
Data source: This prospective, observational study examined weight gain in 361 antipsychotic-naive children, adolescents, and adults during their first 6 months on second-generation antipsychotic agents.
Disclosures: The study was funded by the Spanish Ministry of Economy and Competitiveness and the European Commission.
CEQUEL: Combo therapy a win in bipolar depression
VIENNA – Psychiatrists have received a welcome gift in the form of CEQUEL, a randomized trial that offers much needed guidance on how to proceed when quetiapine for bipolar depression isn’t achieving sufficient response, Eduard Vieta, MD, said at the annual congress of the European College of Neuropsychopharmacology.
The answer provided by CEQUEL is to add lamotrigine.
“Quetiapine is indicated for treatment of symptoms of bipolar depression, but many clinicians – including myself – are a bit underwhelmed about the practical results. In the clinical trials, quetiapine looks great, but when we use it in practice we’re not so happy with the results. The trials are consistently positive with a large effect size, whereas in clinical practice many patients respond only partially to quetiapine. The question has been what to do next,” observed Dr. Vieta, professor of psychiatry and head of the bipolar disorders program at the University of Barcelona.
Increasing the dose of quetiapine (Seroquel) above the recommended 300 mg/day in an effort to boost efficacy is problematic. Patients become extremely sedated. The CEQUEL (Comparative Evaluation of Quetiapine plus Lamotrigine) investigators decided to examine combination therapy with two drugs having completely different mechanisms of action.
A total of 202 patients with bipolar depression at 27 U.K. sites were placed on quetiapine and randomized double-blind to lamotrigine or placebo. Lamotrigine (Lamictal) was started at 25 mg/day and gradually increased to 200 mg daily. Because there is some limited evidence that folic acid is effective in unipolar depression, the investigators employed a 2x2 factorial design, further randomizing participants to 500 mcg/day of folic acid or placebo.
The primary endpoint was improvement in depressive symptoms at 12 weeks, with 52-week outcomes as a secondary endpoint. The group on combination therapy with quetiapine and lamotrigine showed significantly greater improvement both on mean depressive symptoms as measured with the Quick Inventory of Depressive Symptomatology-self report version 16 (QIDS-SR16), and on rates of remission at 12 and 52 weeks, compared with patients on quetiapine and placebo. The combination also reduced the risk of relapse in patients with bipolar I disorder with predominantly depressive symptoms.
Folic acid proved to be not only ineffective, it actually interfered with lamotrigine’s effectiveness. The mean reduction from baseline to 12 weeks on the QIDS-SR16 was 4.4 points in patients who received lamotrigine without folic acid, compared with a 0.12-point increase in those receiving lamotrigine plus folic acid (Lancet Psychiatry. 2016 Jan;3[1]:31-9).
Dr. Vieta wasn’t involved in CEQUEL, but he considers the study one of the highlights of the year in the field of affective disorders and urged his colleagues to consider lamotrigine/quetiapine combination therapy for bipolar depression.
“Quetiapine and lamotrigine are both now generic drugs. CEQUEL showed both are superior to quetiapine alone. It’s good news – a not-expensive treatment combining quetiapine with a drug, lamotrigine, that’s quite well tolerated,” he said.
CEQUEL was funded by the Medical Research Council. Dr. Vieta reported having no financial conflicts of interest.
VIENNA – Psychiatrists have received a welcome gift in the form of CEQUEL, a randomized trial that offers much needed guidance on how to proceed when quetiapine for bipolar depression isn’t achieving sufficient response, Eduard Vieta, MD, said at the annual congress of the European College of Neuropsychopharmacology.
The answer provided by CEQUEL is to add lamotrigine.
“Quetiapine is indicated for treatment of symptoms of bipolar depression, but many clinicians – including myself – are a bit underwhelmed about the practical results. In the clinical trials, quetiapine looks great, but when we use it in practice we’re not so happy with the results. The trials are consistently positive with a large effect size, whereas in clinical practice many patients respond only partially to quetiapine. The question has been what to do next,” observed Dr. Vieta, professor of psychiatry and head of the bipolar disorders program at the University of Barcelona.
Increasing the dose of quetiapine (Seroquel) above the recommended 300 mg/day in an effort to boost efficacy is problematic. Patients become extremely sedated. The CEQUEL (Comparative Evaluation of Quetiapine plus Lamotrigine) investigators decided to examine combination therapy with two drugs having completely different mechanisms of action.
A total of 202 patients with bipolar depression at 27 U.K. sites were placed on quetiapine and randomized double-blind to lamotrigine or placebo. Lamotrigine (Lamictal) was started at 25 mg/day and gradually increased to 200 mg daily. Because there is some limited evidence that folic acid is effective in unipolar depression, the investigators employed a 2x2 factorial design, further randomizing participants to 500 mcg/day of folic acid or placebo.
The primary endpoint was improvement in depressive symptoms at 12 weeks, with 52-week outcomes as a secondary endpoint. The group on combination therapy with quetiapine and lamotrigine showed significantly greater improvement both on mean depressive symptoms as measured with the Quick Inventory of Depressive Symptomatology-self report version 16 (QIDS-SR16), and on rates of remission at 12 and 52 weeks, compared with patients on quetiapine and placebo. The combination also reduced the risk of relapse in patients with bipolar I disorder with predominantly depressive symptoms.
Folic acid proved to be not only ineffective, it actually interfered with lamotrigine’s effectiveness. The mean reduction from baseline to 12 weeks on the QIDS-SR16 was 4.4 points in patients who received lamotrigine without folic acid, compared with a 0.12-point increase in those receiving lamotrigine plus folic acid (Lancet Psychiatry. 2016 Jan;3[1]:31-9).
Dr. Vieta wasn’t involved in CEQUEL, but he considers the study one of the highlights of the year in the field of affective disorders and urged his colleagues to consider lamotrigine/quetiapine combination therapy for bipolar depression.
“Quetiapine and lamotrigine are both now generic drugs. CEQUEL showed both are superior to quetiapine alone. It’s good news – a not-expensive treatment combining quetiapine with a drug, lamotrigine, that’s quite well tolerated,” he said.
CEQUEL was funded by the Medical Research Council. Dr. Vieta reported having no financial conflicts of interest.
VIENNA – Psychiatrists have received a welcome gift in the form of CEQUEL, a randomized trial that offers much needed guidance on how to proceed when quetiapine for bipolar depression isn’t achieving sufficient response, Eduard Vieta, MD, said at the annual congress of the European College of Neuropsychopharmacology.
The answer provided by CEQUEL is to add lamotrigine.
“Quetiapine is indicated for treatment of symptoms of bipolar depression, but many clinicians – including myself – are a bit underwhelmed about the practical results. In the clinical trials, quetiapine looks great, but when we use it in practice we’re not so happy with the results. The trials are consistently positive with a large effect size, whereas in clinical practice many patients respond only partially to quetiapine. The question has been what to do next,” observed Dr. Vieta, professor of psychiatry and head of the bipolar disorders program at the University of Barcelona.
Increasing the dose of quetiapine (Seroquel) above the recommended 300 mg/day in an effort to boost efficacy is problematic. Patients become extremely sedated. The CEQUEL (Comparative Evaluation of Quetiapine plus Lamotrigine) investigators decided to examine combination therapy with two drugs having completely different mechanisms of action.
A total of 202 patients with bipolar depression at 27 U.K. sites were placed on quetiapine and randomized double-blind to lamotrigine or placebo. Lamotrigine (Lamictal) was started at 25 mg/day and gradually increased to 200 mg daily. Because there is some limited evidence that folic acid is effective in unipolar depression, the investigators employed a 2x2 factorial design, further randomizing participants to 500 mcg/day of folic acid or placebo.
The primary endpoint was improvement in depressive symptoms at 12 weeks, with 52-week outcomes as a secondary endpoint. The group on combination therapy with quetiapine and lamotrigine showed significantly greater improvement both on mean depressive symptoms as measured with the Quick Inventory of Depressive Symptomatology-self report version 16 (QIDS-SR16), and on rates of remission at 12 and 52 weeks, compared with patients on quetiapine and placebo. The combination also reduced the risk of relapse in patients with bipolar I disorder with predominantly depressive symptoms.
Folic acid proved to be not only ineffective, it actually interfered with lamotrigine’s effectiveness. The mean reduction from baseline to 12 weeks on the QIDS-SR16 was 4.4 points in patients who received lamotrigine without folic acid, compared with a 0.12-point increase in those receiving lamotrigine plus folic acid (Lancet Psychiatry. 2016 Jan;3[1]:31-9).
Dr. Vieta wasn’t involved in CEQUEL, but he considers the study one of the highlights of the year in the field of affective disorders and urged his colleagues to consider lamotrigine/quetiapine combination therapy for bipolar depression.
“Quetiapine and lamotrigine are both now generic drugs. CEQUEL showed both are superior to quetiapine alone. It’s good news – a not-expensive treatment combining quetiapine with a drug, lamotrigine, that’s quite well tolerated,” he said.
CEQUEL was funded by the Medical Research Council. Dr. Vieta reported having no financial conflicts of interest.
Ketamine curbs suicidal ideation independent of antidepressant effect
VIENNA – Repeated infusions of ketamine appear to have a direct effect in quelling suicidal ideation independent of the drug’s impact on depressive symptoms, Pierre Blier, MD, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
“Our preliminary results indicate that ketamine reduced suicidal ideation even in patients who failed to show a reduction in depression severity with repeated infusions,” observed Dr. Blier, professor of psychiatry at the University of Ottawa.
If the findings from this proof-of-concept study are confirmed, it would be very good news indeed.
“Suicidal ideation often requires rapid intervention, but there are very few treatments that are effective in reducing suicidal ideation, and none are fast acting,” the psychiatrist noted.
Therapeutic interest in ketamine is running high because subanesthetic doses of the drug have been shown to reduce depressive symptoms within a matter of hours, albeit transiently, in patients with treatment-resistant depression. If the drug acts as quickly in decreasing suicidal ideation as it does for depression, it would have major implications for clinical practice. An estimated 1 million people die each year worldwide from suicide, and two-thirds of them have major depressive disorder.
Dr. Blier reported on 27 patients with treatment-resistant depression, defined as failure to respond to at least two antidepressant medications from different pharmacologic classes at adequate dosages for at least 6 weeks each, along with two augmentation strategies. Their mean baseline score on the Montgomery-Asberg Depression Rating Scale (MADRS) was roughly 35. Five of the 27 had made one or more prior suicide attempts. Twenty-six of the 27 had suicidal ideation at baseline as defined by a MADRS suicidal ideation score (MADRS-SI) of more than 0. The MADRS-SI is based upon item 10 on the MADRS and is scored 0-6. Eight patients reported marked suicidal ideation as defined by a baseline MADRS-SI score of 4 or higher.
Study participants received a total of seven intravenous infusions of ketamine over roughly 3 weeks. Patients were retested 2-3 days after the seventh infusion, at which time 16 patients were categorized as treatment responders on the basis of at least a 50% reduction in their total MADRS score. Indeed, they averaged a 20.7-point reduction in the non–suicide-related MADRS score – that is, items 1-9, which reflect symptoms of depression – compared with a mere 3.9-point decrease in the 11 ketamine nonresponders. This 59% response rate for depressive symptoms is consistent with reported results of other studies of ketamine in patients with treatment-resistant depression, according to Dr. Blier, also professor of cellular/molecular medicines at the university.
The baseline MADRS-SI score averaged 2.9 in treatment responders and was similar at 2.5 in the nonresponders. But after seven infusions of ketamine, 21 of 27 patients had a MADRS-SI score of 0 or 1. The key study finding was that both the ketamine responders and nonresponders in terms of depressive symptoms experienced significant reductions in suicidal ideation. The 16 responders averaged a 2.4-point decrease from baseline in MADRS-SI scores, while the 11 nonresponders averaged a 1.5-point reduction. The magnitude of the reduction in MADRS-SI was significantly greater in the antidepressant responders than the nonresponders, but the improvement in suicidal ideation was statistically significant and clinically meaningful in both groups.
Seven of the 8 patients with baseline marked suicidal ideation and a MADRS-SI of 4 or greater improved to a score of 2 or less after their seven ketamine infusions, meaning they were having at most only fleeting suicidal thoughts.
Dr. Blier and coinvestigators are now expanding the ketamine study, enrolling up to 40 additional patients with treatment-resistant depression and suicidality in order to confirm the preliminary findings.
Dr. Blier reported having no financial conflicts of interest regarding this study, which was funded by the Canadian Institutes of Health Research. He noted that he receives research grants from and/or serves on advisory boards for more than a dozen pharmaceutical companies.
VIENNA – Repeated infusions of ketamine appear to have a direct effect in quelling suicidal ideation independent of the drug’s impact on depressive symptoms, Pierre Blier, MD, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
“Our preliminary results indicate that ketamine reduced suicidal ideation even in patients who failed to show a reduction in depression severity with repeated infusions,” observed Dr. Blier, professor of psychiatry at the University of Ottawa.
If the findings from this proof-of-concept study are confirmed, it would be very good news indeed.
“Suicidal ideation often requires rapid intervention, but there are very few treatments that are effective in reducing suicidal ideation, and none are fast acting,” the psychiatrist noted.
Therapeutic interest in ketamine is running high because subanesthetic doses of the drug have been shown to reduce depressive symptoms within a matter of hours, albeit transiently, in patients with treatment-resistant depression. If the drug acts as quickly in decreasing suicidal ideation as it does for depression, it would have major implications for clinical practice. An estimated 1 million people die each year worldwide from suicide, and two-thirds of them have major depressive disorder.
Dr. Blier reported on 27 patients with treatment-resistant depression, defined as failure to respond to at least two antidepressant medications from different pharmacologic classes at adequate dosages for at least 6 weeks each, along with two augmentation strategies. Their mean baseline score on the Montgomery-Asberg Depression Rating Scale (MADRS) was roughly 35. Five of the 27 had made one or more prior suicide attempts. Twenty-six of the 27 had suicidal ideation at baseline as defined by a MADRS suicidal ideation score (MADRS-SI) of more than 0. The MADRS-SI is based upon item 10 on the MADRS and is scored 0-6. Eight patients reported marked suicidal ideation as defined by a baseline MADRS-SI score of 4 or higher.
Study participants received a total of seven intravenous infusions of ketamine over roughly 3 weeks. Patients were retested 2-3 days after the seventh infusion, at which time 16 patients were categorized as treatment responders on the basis of at least a 50% reduction in their total MADRS score. Indeed, they averaged a 20.7-point reduction in the non–suicide-related MADRS score – that is, items 1-9, which reflect symptoms of depression – compared with a mere 3.9-point decrease in the 11 ketamine nonresponders. This 59% response rate for depressive symptoms is consistent with reported results of other studies of ketamine in patients with treatment-resistant depression, according to Dr. Blier, also professor of cellular/molecular medicines at the university.
The baseline MADRS-SI score averaged 2.9 in treatment responders and was similar at 2.5 in the nonresponders. But after seven infusions of ketamine, 21 of 27 patients had a MADRS-SI score of 0 or 1. The key study finding was that both the ketamine responders and nonresponders in terms of depressive symptoms experienced significant reductions in suicidal ideation. The 16 responders averaged a 2.4-point decrease from baseline in MADRS-SI scores, while the 11 nonresponders averaged a 1.5-point reduction. The magnitude of the reduction in MADRS-SI was significantly greater in the antidepressant responders than the nonresponders, but the improvement in suicidal ideation was statistically significant and clinically meaningful in both groups.
Seven of the 8 patients with baseline marked suicidal ideation and a MADRS-SI of 4 or greater improved to a score of 2 or less after their seven ketamine infusions, meaning they were having at most only fleeting suicidal thoughts.
Dr. Blier and coinvestigators are now expanding the ketamine study, enrolling up to 40 additional patients with treatment-resistant depression and suicidality in order to confirm the preliminary findings.
Dr. Blier reported having no financial conflicts of interest regarding this study, which was funded by the Canadian Institutes of Health Research. He noted that he receives research grants from and/or serves on advisory boards for more than a dozen pharmaceutical companies.
VIENNA – Repeated infusions of ketamine appear to have a direct effect in quelling suicidal ideation independent of the drug’s impact on depressive symptoms, Pierre Blier, MD, PhD, reported at the annual congress of the European College of Neuropsychopharmacology.
“Our preliminary results indicate that ketamine reduced suicidal ideation even in patients who failed to show a reduction in depression severity with repeated infusions,” observed Dr. Blier, professor of psychiatry at the University of Ottawa.
If the findings from this proof-of-concept study are confirmed, it would be very good news indeed.
“Suicidal ideation often requires rapid intervention, but there are very few treatments that are effective in reducing suicidal ideation, and none are fast acting,” the psychiatrist noted.
Therapeutic interest in ketamine is running high because subanesthetic doses of the drug have been shown to reduce depressive symptoms within a matter of hours, albeit transiently, in patients with treatment-resistant depression. If the drug acts as quickly in decreasing suicidal ideation as it does for depression, it would have major implications for clinical practice. An estimated 1 million people die each year worldwide from suicide, and two-thirds of them have major depressive disorder.
Dr. Blier reported on 27 patients with treatment-resistant depression, defined as failure to respond to at least two antidepressant medications from different pharmacologic classes at adequate dosages for at least 6 weeks each, along with two augmentation strategies. Their mean baseline score on the Montgomery-Asberg Depression Rating Scale (MADRS) was roughly 35. Five of the 27 had made one or more prior suicide attempts. Twenty-six of the 27 had suicidal ideation at baseline as defined by a MADRS suicidal ideation score (MADRS-SI) of more than 0. The MADRS-SI is based upon item 10 on the MADRS and is scored 0-6. Eight patients reported marked suicidal ideation as defined by a baseline MADRS-SI score of 4 or higher.
Study participants received a total of seven intravenous infusions of ketamine over roughly 3 weeks. Patients were retested 2-3 days after the seventh infusion, at which time 16 patients were categorized as treatment responders on the basis of at least a 50% reduction in their total MADRS score. Indeed, they averaged a 20.7-point reduction in the non–suicide-related MADRS score – that is, items 1-9, which reflect symptoms of depression – compared with a mere 3.9-point decrease in the 11 ketamine nonresponders. This 59% response rate for depressive symptoms is consistent with reported results of other studies of ketamine in patients with treatment-resistant depression, according to Dr. Blier, also professor of cellular/molecular medicines at the university.
The baseline MADRS-SI score averaged 2.9 in treatment responders and was similar at 2.5 in the nonresponders. But after seven infusions of ketamine, 21 of 27 patients had a MADRS-SI score of 0 or 1. The key study finding was that both the ketamine responders and nonresponders in terms of depressive symptoms experienced significant reductions in suicidal ideation. The 16 responders averaged a 2.4-point decrease from baseline in MADRS-SI scores, while the 11 nonresponders averaged a 1.5-point reduction. The magnitude of the reduction in MADRS-SI was significantly greater in the antidepressant responders than the nonresponders, but the improvement in suicidal ideation was statistically significant and clinically meaningful in both groups.
Seven of the 8 patients with baseline marked suicidal ideation and a MADRS-SI of 4 or greater improved to a score of 2 or less after their seven ketamine infusions, meaning they were having at most only fleeting suicidal thoughts.
Dr. Blier and coinvestigators are now expanding the ketamine study, enrolling up to 40 additional patients with treatment-resistant depression and suicidality in order to confirm the preliminary findings.
Dr. Blier reported having no financial conflicts of interest regarding this study, which was funded by the Canadian Institutes of Health Research. He noted that he receives research grants from and/or serves on advisory boards for more than a dozen pharmaceutical companies.
AT THE ECNP CONGRESS
Key clinical point:
Major finding: Ketamine infusions significantly reduced suicidal ideation even in patients whose treatment-resistant depression did not respond to the drug.
Data source: This ongoing observational study included 27 patients with treatment-resistant depression who received seven intravenous infusions of ketamine over roughly a 3-week period.
Disclosures: The presenter reported having no financial conflicts of interest regarding this study, which was funded by the Canadian Institutes of Health Research.