User login
VIDEO: RA linked to increased mortality in Nurses’ Health Study
BOSTON – Rheumatoid arthritis has been linked to increased risk for death in one of the first studies to directly compare RA patients with controls over a prolonged period of time. In women who developed RA after enrollment in the Nurses’ Health Study, seropositive disease was strongly associated with worse outcomes. Also, some surprising findings emerged about RA and the risk for fatal respiratory disease in women.
In our exclusive interview, one of the study’s lead authors, Dr. Jeffrey A. Sparks of Brigham and Women’s Hospital, Boston, discusses how the findings may begin to influence the clinical management of women with RA.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BOSTON – Rheumatoid arthritis has been linked to increased risk for death in one of the first studies to directly compare RA patients with controls over a prolonged period of time. In women who developed RA after enrollment in the Nurses’ Health Study, seropositive disease was strongly associated with worse outcomes. Also, some surprising findings emerged about RA and the risk for fatal respiratory disease in women.
In our exclusive interview, one of the study’s lead authors, Dr. Jeffrey A. Sparks of Brigham and Women’s Hospital, Boston, discusses how the findings may begin to influence the clinical management of women with RA.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
BOSTON – Rheumatoid arthritis has been linked to increased risk for death in one of the first studies to directly compare RA patients with controls over a prolonged period of time. In women who developed RA after enrollment in the Nurses’ Health Study, seropositive disease was strongly associated with worse outcomes. Also, some surprising findings emerged about RA and the risk for fatal respiratory disease in women.
In our exclusive interview, one of the study’s lead authors, Dr. Jeffrey A. Sparks of Brigham and Women’s Hospital, Boston, discusses how the findings may begin to influence the clinical management of women with RA.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT ACR 2014
Evidence builds for importance of CVD, new drug targets at ACR
It’s difficult to highlight the most striking studies or topic areas out of the bounty of research that will be presented at the annual meeting of the American College of Rheumatology, but a few noteworthy studies caught the eye of Dr. Eric L. Matteson, the meeting’s planning subcommittee abstract selection cochair for clinical research.
Some of the studies and topic areas that deserve attention include growing knowledge about the risk of cardiovascular disease in many rheumatic conditions; the conditions in which rheumatoid arthritis (RA) treatment can be cost-effective, and de-escalation of disease-modifying antirheumatic drugs (DMARDs) may be possible; promising clinical results with new drug targets; according to Dr. Matteson, professor of medicine and chair of the department of rheumatology at the Mayo Clinic, Rochester, Minn.
Cardiovascular disease in spondyloarthritis, RA
“One major area in rheumatic diseases is looking at the emerging field of cardiovascular disease. We’ve known that there’s increased risk for heart disease in rheumatoid arthritis patients, not necessarily what to do about it. Now increasingly we’re finding that there’s the same risk in some other inflammatory arthritis diseases,” Dr. Matteson said. A Canadian study to be presented on Tuesday (abstract 2829) is the first population-based cohort study to show that patients with spondyloarthritis are at increased risk for heart disease, although at 40%-50% increased risk of cardiovascular death in comparison to people without spondyloarthritis, it is not at the same magnitude of risk as what is observed for RA, where risk for CV death is doubled in some subgroups, such as women with rheumatoid factor-positive disease.
“I think that’s something that’s new and is significant,” Dr. Matteson said.
Another interesting study to be reported on Sunday provides some initial evidence to suggest that reducing disease activity in RA might reduce the risk of heart failure, which has been shown to be elevated in RA patients. The prospective, cross-sectional study (abstract 945) of patients from a clinic in Germany found progressively lower prevalence of heart failure as 28-joint Disease Activity Score declined, independent of treatment type.
De-escalating DMARDs in RA
Another “hot-button” issue in RA is whether it is possible to reduce therapy, Dr. Matteson noted. On Sunday, German investigators will present a multicenter, randomized controlled trial (abstract 940) showing that while DMARDs can be successfully de-escalated, it’s not clear whether starting the de-escalation process is better done first with a conventional DMARD or a biologic. The presence of anticitrullinated peptide antibodies turned out to be the only predictor of recurrent disease. Another study in the same session (abstract 941) indicated that an attempt to de-escalate biologic treatment was successful in 87% of the selected patients who tried to do so and provided substantial cost savings.
New drug targets in clinical research
One of the unique therapeutic approaches being tried is using autologous dendritic cell immunotherapy to treat RA patients, Dr. Matteson said. “The dendritic cell is an important immune-mediating cell actually not just in rheumatoid arthritis but a lot of other systemic inflammatory conditions including even giant cell arteritis.” A phase I, open-label study (abstract 946) that will be presented on Sunday demonstrated that five vaccinations with high or low doses of the cells over 2- to 4-week intervals in 12 RA patients yielded good to moderate European League Against Rheumatism (EULAR) responses in 7 patients while 12 adverse events occurred in 8 patients.
After not-so-encouraging studies of interleukin-17 as a drug target in RA, its potential in seronegative inflammatory arthritis appears much more promising. The results of a 1-year, phase III trial of secukinumab, a monoclonal antibody to IL-17A, in patients with psoriatic arthritis is “quite hopeful” because there were improvements in clinical parameters of disease and an indication of inhibition of joint erosion (abstract 954). Another phase III study of the drug showed “quite promising effects” in controlling ankylosing spondylitis over the course of 1 year (abstract 819). Both studies will be presented on Sunday.
Noteworthy plenary presentations
“One of the hot areas in lung disease in rheumatic diseases” involves controversy over the value of lung transplantation in patients with systemic sclerosis (SSc) and end-stage lung disease, Dr. Matteson said. A study to be presented in Monday’s plenary session suggests that new methods must be developed to determine which SSc patients can successfully undergo lung transplantation rather than to deny all patients with the condition the chance to undergo the procedure. While the study (abstract 1797) confirmed that patients with SSc have a nearly 50% increased risk of death at 1 year posttransplant when compared with interstitial lung disease patients without SSc, there was no difference in mortality for SSc patients, compared with patients with pulmonary arterial hypertension not caused by SSc.
In Tuesday’s plenary session, investigators will report on the cost-effectiveness of triple therapy, compared with biologics. The results of the study (abstract 2781) suggest that added costs associated with using etanercept prior to a triple-therapy regimen may not be ideal in RA patients for whom methotrexate is not working. Because etanercept has only a small probability of being cost-effective, compared with triple therapy, more health care dollars could be freed up, the authors argue, by adopting the triple regimen prior to etanercept.
Dr. Matteson has received research support from many companies marketing drugs for rheumatic diseases.
It’s difficult to highlight the most striking studies or topic areas out of the bounty of research that will be presented at the annual meeting of the American College of Rheumatology, but a few noteworthy studies caught the eye of Dr. Eric L. Matteson, the meeting’s planning subcommittee abstract selection cochair for clinical research.
Some of the studies and topic areas that deserve attention include growing knowledge about the risk of cardiovascular disease in many rheumatic conditions; the conditions in which rheumatoid arthritis (RA) treatment can be cost-effective, and de-escalation of disease-modifying antirheumatic drugs (DMARDs) may be possible; promising clinical results with new drug targets; according to Dr. Matteson, professor of medicine and chair of the department of rheumatology at the Mayo Clinic, Rochester, Minn.
Cardiovascular disease in spondyloarthritis, RA
“One major area in rheumatic diseases is looking at the emerging field of cardiovascular disease. We’ve known that there’s increased risk for heart disease in rheumatoid arthritis patients, not necessarily what to do about it. Now increasingly we’re finding that there’s the same risk in some other inflammatory arthritis diseases,” Dr. Matteson said. A Canadian study to be presented on Tuesday (abstract 2829) is the first population-based cohort study to show that patients with spondyloarthritis are at increased risk for heart disease, although at 40%-50% increased risk of cardiovascular death in comparison to people without spondyloarthritis, it is not at the same magnitude of risk as what is observed for RA, where risk for CV death is doubled in some subgroups, such as women with rheumatoid factor-positive disease.
“I think that’s something that’s new and is significant,” Dr. Matteson said.
Another interesting study to be reported on Sunday provides some initial evidence to suggest that reducing disease activity in RA might reduce the risk of heart failure, which has been shown to be elevated in RA patients. The prospective, cross-sectional study (abstract 945) of patients from a clinic in Germany found progressively lower prevalence of heart failure as 28-joint Disease Activity Score declined, independent of treatment type.
De-escalating DMARDs in RA
Another “hot-button” issue in RA is whether it is possible to reduce therapy, Dr. Matteson noted. On Sunday, German investigators will present a multicenter, randomized controlled trial (abstract 940) showing that while DMARDs can be successfully de-escalated, it’s not clear whether starting the de-escalation process is better done first with a conventional DMARD or a biologic. The presence of anticitrullinated peptide antibodies turned out to be the only predictor of recurrent disease. Another study in the same session (abstract 941) indicated that an attempt to de-escalate biologic treatment was successful in 87% of the selected patients who tried to do so and provided substantial cost savings.
New drug targets in clinical research
One of the unique therapeutic approaches being tried is using autologous dendritic cell immunotherapy to treat RA patients, Dr. Matteson said. “The dendritic cell is an important immune-mediating cell actually not just in rheumatoid arthritis but a lot of other systemic inflammatory conditions including even giant cell arteritis.” A phase I, open-label study (abstract 946) that will be presented on Sunday demonstrated that five vaccinations with high or low doses of the cells over 2- to 4-week intervals in 12 RA patients yielded good to moderate European League Against Rheumatism (EULAR) responses in 7 patients while 12 adverse events occurred in 8 patients.
After not-so-encouraging studies of interleukin-17 as a drug target in RA, its potential in seronegative inflammatory arthritis appears much more promising. The results of a 1-year, phase III trial of secukinumab, a monoclonal antibody to IL-17A, in patients with psoriatic arthritis is “quite hopeful” because there were improvements in clinical parameters of disease and an indication of inhibition of joint erosion (abstract 954). Another phase III study of the drug showed “quite promising effects” in controlling ankylosing spondylitis over the course of 1 year (abstract 819). Both studies will be presented on Sunday.
Noteworthy plenary presentations
“One of the hot areas in lung disease in rheumatic diseases” involves controversy over the value of lung transplantation in patients with systemic sclerosis (SSc) and end-stage lung disease, Dr. Matteson said. A study to be presented in Monday’s plenary session suggests that new methods must be developed to determine which SSc patients can successfully undergo lung transplantation rather than to deny all patients with the condition the chance to undergo the procedure. While the study (abstract 1797) confirmed that patients with SSc have a nearly 50% increased risk of death at 1 year posttransplant when compared with interstitial lung disease patients without SSc, there was no difference in mortality for SSc patients, compared with patients with pulmonary arterial hypertension not caused by SSc.
In Tuesday’s plenary session, investigators will report on the cost-effectiveness of triple therapy, compared with biologics. The results of the study (abstract 2781) suggest that added costs associated with using etanercept prior to a triple-therapy regimen may not be ideal in RA patients for whom methotrexate is not working. Because etanercept has only a small probability of being cost-effective, compared with triple therapy, more health care dollars could be freed up, the authors argue, by adopting the triple regimen prior to etanercept.
Dr. Matteson has received research support from many companies marketing drugs for rheumatic diseases.
It’s difficult to highlight the most striking studies or topic areas out of the bounty of research that will be presented at the annual meeting of the American College of Rheumatology, but a few noteworthy studies caught the eye of Dr. Eric L. Matteson, the meeting’s planning subcommittee abstract selection cochair for clinical research.
Some of the studies and topic areas that deserve attention include growing knowledge about the risk of cardiovascular disease in many rheumatic conditions; the conditions in which rheumatoid arthritis (RA) treatment can be cost-effective, and de-escalation of disease-modifying antirheumatic drugs (DMARDs) may be possible; promising clinical results with new drug targets; according to Dr. Matteson, professor of medicine and chair of the department of rheumatology at the Mayo Clinic, Rochester, Minn.
Cardiovascular disease in spondyloarthritis, RA
“One major area in rheumatic diseases is looking at the emerging field of cardiovascular disease. We’ve known that there’s increased risk for heart disease in rheumatoid arthritis patients, not necessarily what to do about it. Now increasingly we’re finding that there’s the same risk in some other inflammatory arthritis diseases,” Dr. Matteson said. A Canadian study to be presented on Tuesday (abstract 2829) is the first population-based cohort study to show that patients with spondyloarthritis are at increased risk for heart disease, although at 40%-50% increased risk of cardiovascular death in comparison to people without spondyloarthritis, it is not at the same magnitude of risk as what is observed for RA, where risk for CV death is doubled in some subgroups, such as women with rheumatoid factor-positive disease.
“I think that’s something that’s new and is significant,” Dr. Matteson said.
Another interesting study to be reported on Sunday provides some initial evidence to suggest that reducing disease activity in RA might reduce the risk of heart failure, which has been shown to be elevated in RA patients. The prospective, cross-sectional study (abstract 945) of patients from a clinic in Germany found progressively lower prevalence of heart failure as 28-joint Disease Activity Score declined, independent of treatment type.
De-escalating DMARDs in RA
Another “hot-button” issue in RA is whether it is possible to reduce therapy, Dr. Matteson noted. On Sunday, German investigators will present a multicenter, randomized controlled trial (abstract 940) showing that while DMARDs can be successfully de-escalated, it’s not clear whether starting the de-escalation process is better done first with a conventional DMARD or a biologic. The presence of anticitrullinated peptide antibodies turned out to be the only predictor of recurrent disease. Another study in the same session (abstract 941) indicated that an attempt to de-escalate biologic treatment was successful in 87% of the selected patients who tried to do so and provided substantial cost savings.
New drug targets in clinical research
One of the unique therapeutic approaches being tried is using autologous dendritic cell immunotherapy to treat RA patients, Dr. Matteson said. “The dendritic cell is an important immune-mediating cell actually not just in rheumatoid arthritis but a lot of other systemic inflammatory conditions including even giant cell arteritis.” A phase I, open-label study (abstract 946) that will be presented on Sunday demonstrated that five vaccinations with high or low doses of the cells over 2- to 4-week intervals in 12 RA patients yielded good to moderate European League Against Rheumatism (EULAR) responses in 7 patients while 12 adverse events occurred in 8 patients.
After not-so-encouraging studies of interleukin-17 as a drug target in RA, its potential in seronegative inflammatory arthritis appears much more promising. The results of a 1-year, phase III trial of secukinumab, a monoclonal antibody to IL-17A, in patients with psoriatic arthritis is “quite hopeful” because there were improvements in clinical parameters of disease and an indication of inhibition of joint erosion (abstract 954). Another phase III study of the drug showed “quite promising effects” in controlling ankylosing spondylitis over the course of 1 year (abstract 819). Both studies will be presented on Sunday.
Noteworthy plenary presentations
“One of the hot areas in lung disease in rheumatic diseases” involves controversy over the value of lung transplantation in patients with systemic sclerosis (SSc) and end-stage lung disease, Dr. Matteson said. A study to be presented in Monday’s plenary session suggests that new methods must be developed to determine which SSc patients can successfully undergo lung transplantation rather than to deny all patients with the condition the chance to undergo the procedure. While the study (abstract 1797) confirmed that patients with SSc have a nearly 50% increased risk of death at 1 year posttransplant when compared with interstitial lung disease patients without SSc, there was no difference in mortality for SSc patients, compared with patients with pulmonary arterial hypertension not caused by SSc.
In Tuesday’s plenary session, investigators will report on the cost-effectiveness of triple therapy, compared with biologics. The results of the study (abstract 2781) suggest that added costs associated with using etanercept prior to a triple-therapy regimen may not be ideal in RA patients for whom methotrexate is not working. Because etanercept has only a small probability of being cost-effective, compared with triple therapy, more health care dollars could be freed up, the authors argue, by adopting the triple regimen prior to etanercept.
Dr. Matteson has received research support from many companies marketing drugs for rheumatic diseases.
Children born to mothers with RA more likely to be born early
Children born to mothers with rheumatoid arthritis are more likely to be born prematurely and have a lower birth weight than are those born to mothers without the condition, according to a Danish nationwide cohort study.
The reduction in fetal growth, however, is small – a mean difference of 87 g – and unlikely to have any impact on the well being of the child, Ane Rom of Copenhagen University Hospital and her colleagues reported in Arthritis & Rheumatology.
Of 1,917,723 children born in Denmark during 1977-2008, 13,556 children were exposed to either maternal or preclinical RA. Children with mothers who had rheumatoid arthritis (RA) were almost one and half times more likely (odds ratio, 1.48; 95% confidence interval, 1.20-1.84) to be born preterm. The odds of preterm birth were slightly lower, but still elevated, in children with mothers who had preclinical signs of disease, compared with children not exposed to RA (OR, 1.32; 95% CI, 1.07-1.64).
Children exposed to RA had a similar length and head and abdominal circumference at birth, compared with children of mothers without RA, but their birth weight was, on average, 87 g lower (95% CI, –111.23 to –62.84) and their placenta weight was 14 g lower (95% CI, –21.46 to –5.43). The investigators found similar results for children exposed to preclinical RA (Arthritis Rheumatol. 2014 Nov. 13 [doi:10.1002/art.38874]).
Paternal RA was not associated with reduced fetal growth or preterm birth. Maternal RA may affect fetal growth either through fetal programming related to the effect that RA may have on the intrauterine environment, through genetic factors, or through medications taken in pregnancy. “For women with RA, it is reassuring that only a small reduction in fetal growth was found for most of their children, which will have little, if any, impact on perinatal conditions for the child,” Ms. Rom and her associates wrote.
“Clinicians should be aware of the increased risk of preterm birth not only in women diagnosed with RA but also in women with signs of preclinical RA,” they concluded.
Ms. Rom’s work on the study was supported by grants from the National Institutes of Health, the Danish Council for Independent Research, and the Augustinus Foundation. No conflicts of interest were reported.
Children born to mothers with rheumatoid arthritis are more likely to be born prematurely and have a lower birth weight than are those born to mothers without the condition, according to a Danish nationwide cohort study.
The reduction in fetal growth, however, is small – a mean difference of 87 g – and unlikely to have any impact on the well being of the child, Ane Rom of Copenhagen University Hospital and her colleagues reported in Arthritis & Rheumatology.
Of 1,917,723 children born in Denmark during 1977-2008, 13,556 children were exposed to either maternal or preclinical RA. Children with mothers who had rheumatoid arthritis (RA) were almost one and half times more likely (odds ratio, 1.48; 95% confidence interval, 1.20-1.84) to be born preterm. The odds of preterm birth were slightly lower, but still elevated, in children with mothers who had preclinical signs of disease, compared with children not exposed to RA (OR, 1.32; 95% CI, 1.07-1.64).
Children exposed to RA had a similar length and head and abdominal circumference at birth, compared with children of mothers without RA, but their birth weight was, on average, 87 g lower (95% CI, –111.23 to –62.84) and their placenta weight was 14 g lower (95% CI, –21.46 to –5.43). The investigators found similar results for children exposed to preclinical RA (Arthritis Rheumatol. 2014 Nov. 13 [doi:10.1002/art.38874]).
Paternal RA was not associated with reduced fetal growth or preterm birth. Maternal RA may affect fetal growth either through fetal programming related to the effect that RA may have on the intrauterine environment, through genetic factors, or through medications taken in pregnancy. “For women with RA, it is reassuring that only a small reduction in fetal growth was found for most of their children, which will have little, if any, impact on perinatal conditions for the child,” Ms. Rom and her associates wrote.
“Clinicians should be aware of the increased risk of preterm birth not only in women diagnosed with RA but also in women with signs of preclinical RA,” they concluded.
Ms. Rom’s work on the study was supported by grants from the National Institutes of Health, the Danish Council for Independent Research, and the Augustinus Foundation. No conflicts of interest were reported.
Children born to mothers with rheumatoid arthritis are more likely to be born prematurely and have a lower birth weight than are those born to mothers without the condition, according to a Danish nationwide cohort study.
The reduction in fetal growth, however, is small – a mean difference of 87 g – and unlikely to have any impact on the well being of the child, Ane Rom of Copenhagen University Hospital and her colleagues reported in Arthritis & Rheumatology.
Of 1,917,723 children born in Denmark during 1977-2008, 13,556 children were exposed to either maternal or preclinical RA. Children with mothers who had rheumatoid arthritis (RA) were almost one and half times more likely (odds ratio, 1.48; 95% confidence interval, 1.20-1.84) to be born preterm. The odds of preterm birth were slightly lower, but still elevated, in children with mothers who had preclinical signs of disease, compared with children not exposed to RA (OR, 1.32; 95% CI, 1.07-1.64).
Children exposed to RA had a similar length and head and abdominal circumference at birth, compared with children of mothers without RA, but their birth weight was, on average, 87 g lower (95% CI, –111.23 to –62.84) and their placenta weight was 14 g lower (95% CI, –21.46 to –5.43). The investigators found similar results for children exposed to preclinical RA (Arthritis Rheumatol. 2014 Nov. 13 [doi:10.1002/art.38874]).
Paternal RA was not associated with reduced fetal growth or preterm birth. Maternal RA may affect fetal growth either through fetal programming related to the effect that RA may have on the intrauterine environment, through genetic factors, or through medications taken in pregnancy. “For women with RA, it is reassuring that only a small reduction in fetal growth was found for most of their children, which will have little, if any, impact on perinatal conditions for the child,” Ms. Rom and her associates wrote.
“Clinicians should be aware of the increased risk of preterm birth not only in women diagnosed with RA but also in women with signs of preclinical RA,” they concluded.
Ms. Rom’s work on the study was supported by grants from the National Institutes of Health, the Danish Council for Independent Research, and the Augustinus Foundation. No conflicts of interest were reported.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Keep aware that pregnant women with RA or signs of preclinical disease may be more likely to give birth preterm and have children with slightly lower birth weight.
Major finding: Children with mothers who had RA were almost one and half times more likely (OR, 1.48; 95% CI, 1.20-1.84) to be born preterm.
Data source: A nationwide cohort study of all singletons born in Denmark during 1977-2008.
Disclosures: Ms. Rom’s work on the study was supported by grants from the National Institutes of Health, the Danish Council for Independent Research, and the Augustinus Foundation. No conflicts of interest were reported.
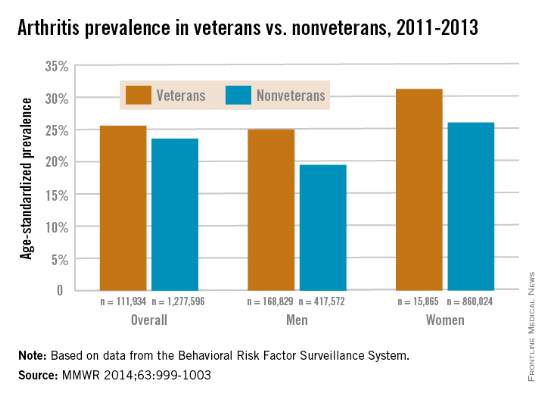
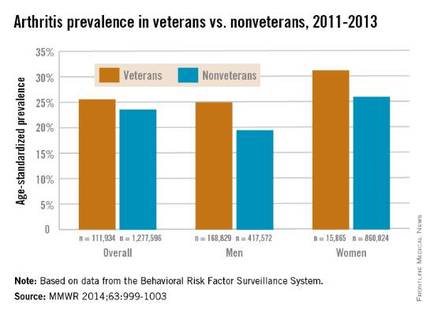
Veterans more likely to have arthritis at any age
Both male and female veterans were more likely to have arthritis than were nonveterans, according to a study from the Centers for Disease Control and Prevention.
For male veterans, the age-standardized arthritis rate for 2011-2013 was 25%, while for male nonveterans, the rate was 19.5%. Both female veterans and nonveterans had noticeably higher arthritis incidence than did the respective male group: 31.3% for veterans and 26.1% for nonveterans, the CDC found (MMWR 2014;63:999-1003).
Although arthritis rates were higher overall in middle-aged and older people, arthritis rates were consistently higher in younger veterans aged 18-44 years – 11.6% for males and 17.3% for females – compared with 6.9% in male nonveterans and 9.8% in female nonveterans. This suggests “that arthritis and its effects need to be addressed among male and female veterans of all ages,” the CDC researchers said.
Traumatic and overuse injuries were found to be common among active-duty military personnel in another study, the investigators noted, while pointing out that musculoskeletal injuries are a major risk factor for osteoarthritis, which “represents the largest portion of arthritis cases” among veterans.
The study used data collected by the Behavioral Risk Factor Surveillance System.
Both male and female veterans were more likely to have arthritis than were nonveterans, according to a study from the Centers for Disease Control and Prevention.
For male veterans, the age-standardized arthritis rate for 2011-2013 was 25%, while for male nonveterans, the rate was 19.5%. Both female veterans and nonveterans had noticeably higher arthritis incidence than did the respective male group: 31.3% for veterans and 26.1% for nonveterans, the CDC found (MMWR 2014;63:999-1003).
Although arthritis rates were higher overall in middle-aged and older people, arthritis rates were consistently higher in younger veterans aged 18-44 years – 11.6% for males and 17.3% for females – compared with 6.9% in male nonveterans and 9.8% in female nonveterans. This suggests “that arthritis and its effects need to be addressed among male and female veterans of all ages,” the CDC researchers said.
Traumatic and overuse injuries were found to be common among active-duty military personnel in another study, the investigators noted, while pointing out that musculoskeletal injuries are a major risk factor for osteoarthritis, which “represents the largest portion of arthritis cases” among veterans.
The study used data collected by the Behavioral Risk Factor Surveillance System.

Both male and female veterans were more likely to have arthritis than were nonveterans, according to a study from the Centers for Disease Control and Prevention.
For male veterans, the age-standardized arthritis rate for 2011-2013 was 25%, while for male nonveterans, the rate was 19.5%. Both female veterans and nonveterans had noticeably higher arthritis incidence than did the respective male group: 31.3% for veterans and 26.1% for nonveterans, the CDC found (MMWR 2014;63:999-1003).
Although arthritis rates were higher overall in middle-aged and older people, arthritis rates were consistently higher in younger veterans aged 18-44 years – 11.6% for males and 17.3% for females – compared with 6.9% in male nonveterans and 9.8% in female nonveterans. This suggests “that arthritis and its effects need to be addressed among male and female veterans of all ages,” the CDC researchers said.
Traumatic and overuse injuries were found to be common among active-duty military personnel in another study, the investigators noted, while pointing out that musculoskeletal injuries are a major risk factor for osteoarthritis, which “represents the largest portion of arthritis cases” among veterans.
The study used data collected by the Behavioral Risk Factor Surveillance System.

FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Chronic inflammatory disease patients at greater risk of major CV events
Patients with rheumatoid arthritis, psoriatic arthritis, or psoriasis are at an increased risk of major adverse cardiovascular events when compared with the general population, according to findings from a large cohort study.
All three diseases had statistically similar risks for major adverse cardiovascular events (MACE) after adjustment for age, gender, and traditional CV risk factors, Dr. Alexis Ogdie-Beatty of the University of Pennsylvania, Philadelphia, and her colleagues reported (Ann. Rheum. Dis. 2014 Oct. 30 [doi: 10.1136/annrheumdis-2014-205675]).
The investigators noted that most studies of CV risk in psoriatic arthritis (PsA) patients have been cross-sectional and that three previous population-based cohort studies have evaluated CV risk in psoriasis patients, with PsA patients as a subgroup; however, these three psoriasis studies did not include incident MACE and matched internal control patients with adjustments for traditional CV risk factors.
The investigators used data from the Health Improvement Network, a U.K. primary care medical record database, and compared the number of MACE (myocardial infarction, cerebrovascular accident, and CV death) that occurred during a mean 5 years of follow-up in 41,752 patients with rheumatoid arthritis (RA), 8,706 with PsA, 138,424 with psoriasis, and 81,573 matched controls. There was significant interaction between disease-modifying antirheumatic drug (DMARD) use and disease group (P < .001 for MACE and two components, CV death and cerebrovascular accident; and P = .01 for MI).
The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49). This risk was elevated in RA patients both with DMARD prescriptions (HR, 1.58; 95% CI, 1.46-1.70) and without (HR, 1.39; 95% CI, 1.28-1.50). Patients with severe psoriasis who were prescribed a DMARD had an HR of 1.42 (95% CI, 1.17-1.73), whereas psoriasis patients not prescribed a DMARD had an HR of 1.08 (95% CI, 1.02-1.15).
The results highlight a need for improved screening and management of traditional CV risk factors in patients with inflammatory diseases, the researchers said.
Study limitations included not being able to measure disease severity or the use of over-the-counter NSAIDs, as well as having few records on biologic medications and possibly missing DMARD prescriptions.
The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
Patients with rheumatoid arthritis, psoriatic arthritis, or psoriasis are at an increased risk of major adverse cardiovascular events when compared with the general population, according to findings from a large cohort study.
All three diseases had statistically similar risks for major adverse cardiovascular events (MACE) after adjustment for age, gender, and traditional CV risk factors, Dr. Alexis Ogdie-Beatty of the University of Pennsylvania, Philadelphia, and her colleagues reported (Ann. Rheum. Dis. 2014 Oct. 30 [doi: 10.1136/annrheumdis-2014-205675]).
The investigators noted that most studies of CV risk in psoriatic arthritis (PsA) patients have been cross-sectional and that three previous population-based cohort studies have evaluated CV risk in psoriasis patients, with PsA patients as a subgroup; however, these three psoriasis studies did not include incident MACE and matched internal control patients with adjustments for traditional CV risk factors.
The investigators used data from the Health Improvement Network, a U.K. primary care medical record database, and compared the number of MACE (myocardial infarction, cerebrovascular accident, and CV death) that occurred during a mean 5 years of follow-up in 41,752 patients with rheumatoid arthritis (RA), 8,706 with PsA, 138,424 with psoriasis, and 81,573 matched controls. There was significant interaction between disease-modifying antirheumatic drug (DMARD) use and disease group (P < .001 for MACE and two components, CV death and cerebrovascular accident; and P = .01 for MI).
The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49). This risk was elevated in RA patients both with DMARD prescriptions (HR, 1.58; 95% CI, 1.46-1.70) and without (HR, 1.39; 95% CI, 1.28-1.50). Patients with severe psoriasis who were prescribed a DMARD had an HR of 1.42 (95% CI, 1.17-1.73), whereas psoriasis patients not prescribed a DMARD had an HR of 1.08 (95% CI, 1.02-1.15).
The results highlight a need for improved screening and management of traditional CV risk factors in patients with inflammatory diseases, the researchers said.
Study limitations included not being able to measure disease severity or the use of over-the-counter NSAIDs, as well as having few records on biologic medications and possibly missing DMARD prescriptions.
The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
Patients with rheumatoid arthritis, psoriatic arthritis, or psoriasis are at an increased risk of major adverse cardiovascular events when compared with the general population, according to findings from a large cohort study.
All three diseases had statistically similar risks for major adverse cardiovascular events (MACE) after adjustment for age, gender, and traditional CV risk factors, Dr. Alexis Ogdie-Beatty of the University of Pennsylvania, Philadelphia, and her colleagues reported (Ann. Rheum. Dis. 2014 Oct. 30 [doi: 10.1136/annrheumdis-2014-205675]).
The investigators noted that most studies of CV risk in psoriatic arthritis (PsA) patients have been cross-sectional and that three previous population-based cohort studies have evaluated CV risk in psoriasis patients, with PsA patients as a subgroup; however, these three psoriasis studies did not include incident MACE and matched internal control patients with adjustments for traditional CV risk factors.
The investigators used data from the Health Improvement Network, a U.K. primary care medical record database, and compared the number of MACE (myocardial infarction, cerebrovascular accident, and CV death) that occurred during a mean 5 years of follow-up in 41,752 patients with rheumatoid arthritis (RA), 8,706 with PsA, 138,424 with psoriasis, and 81,573 matched controls. There was significant interaction between disease-modifying antirheumatic drug (DMARD) use and disease group (P < .001 for MACE and two components, CV death and cerebrovascular accident; and P = .01 for MI).
The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49). This risk was elevated in RA patients both with DMARD prescriptions (HR, 1.58; 95% CI, 1.46-1.70) and without (HR, 1.39; 95% CI, 1.28-1.50). Patients with severe psoriasis who were prescribed a DMARD had an HR of 1.42 (95% CI, 1.17-1.73), whereas psoriasis patients not prescribed a DMARD had an HR of 1.08 (95% CI, 1.02-1.15).
The results highlight a need for improved screening and management of traditional CV risk factors in patients with inflammatory diseases, the researchers said.
Study limitations included not being able to measure disease severity or the use of over-the-counter NSAIDs, as well as having few records on biologic medications and possibly missing DMARD prescriptions.
The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: Patients with RA, PsA, and psoriasis have similarly elevated risk for major adverse cardiovascular events when compared with the general population.
Major finding: The risk of MACE was higher in patients with PsA not prescribed a DMARD (hazard ratio, 1.24; 95% confidence interval, 1.03-1.49), compared with matched controls.
Data source: A population-based, longitudinal cohort study of 41,752 RA patients, 8,706 PsA patients, 138,424 psoriasis patients, and 81,573 matched controls.
Disclosures: The researchers were supported by various grants from the Rheumatology Research Foundation, the National Institutes of Health, the Doris Duke Charitable Foundation, and the Icelandic Research Fund. Several authors reported financial relationships with companies that market drugs for chronic inflammatory diseases.
Abatacept + methotrexate makes early drug-free RA remission possible
Patients with early rheumatoid arthritis who achieved remission after 1 year of treatment with abatacept and methotrexate had a small but significantly greater rate of sustained remission for another 6 months following withdrawal of all drugs when compared with patients who took methotrexate alone in a randomized phase IIIb study.
The AVERT (Assessing Very Early Rheumatoid Arthritis Treatment) study randomized 351 early rheumatoid arthritis (RA) patients who were methotrexate naive, tested positive for anticitrullinated peptide–2 antibody, and had a 28-joint Disease Activity Score using C-reactive protein (DAS28-CRP) of more than 3.2 to weekly doses of 125 mg abatacept plus methotrexate, abatacept monotherapy, or methotrexate alone. Methotrexate was initiated at 7.5 mg/wk and was then titrated to 15-20 mg/wk in 6-8 weeks, but the protocol allowed for 10 mg or less per week in patients who could not tolerate higher doses.
In one of two primary endpoints, significantly more patients in the combination arm achieved remission (DAS28-CRP <2.6) at 12 months (60.9%), compared with patients who took methotrexate alone (45.2%), for an odds ratio of 2.01 (P = .010). The rate was 42.5% in the abatacept monotherapy arm, Dr. Paul Emery of the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England) and his colleagues reported (Ann. Rheum. Dis. 2014 Nov. 3 [doi:10.1136/annrheumdis-2014-206106]).
At the end of the first 12 months, patients who had achieved a DAS28-CRP score of less than 3.2 entered a 12-month treatment withdrawal period. Remission at both 12 and 18 months – the second primary endpoint – occurred in 14.8% of patients on combination therapy and 7.8% of those on methotrexate alone (OR, 2.51; P = .045). A total of 12.4% of abatacept monotherapy patients achieved remission at both 12 and 18 months.
The results suggest that in early RA drug-free remission may be possible following treatment with abatacept, the researchers said.
In the 12-month treatment period, the serious adverse events were reported in 12.1% taking abatacept monotherapy, 6.7% taking combination therapy, and 7.8% taking methotrexate alone.
The study was sponsored by Bristol-Myers Squibb, which manufactures abatacept (Orencia). Dr. Emery reported receiving consulting fees and grant support from Bristol-Myers Squibb as well as other companies that market drugs for RA. Many of his coauthors reported similar disclosures. Two authors are employees of Bristol-Myers Squibb.
Patients with early rheumatoid arthritis who achieved remission after 1 year of treatment with abatacept and methotrexate had a small but significantly greater rate of sustained remission for another 6 months following withdrawal of all drugs when compared with patients who took methotrexate alone in a randomized phase IIIb study.
The AVERT (Assessing Very Early Rheumatoid Arthritis Treatment) study randomized 351 early rheumatoid arthritis (RA) patients who were methotrexate naive, tested positive for anticitrullinated peptide–2 antibody, and had a 28-joint Disease Activity Score using C-reactive protein (DAS28-CRP) of more than 3.2 to weekly doses of 125 mg abatacept plus methotrexate, abatacept monotherapy, or methotrexate alone. Methotrexate was initiated at 7.5 mg/wk and was then titrated to 15-20 mg/wk in 6-8 weeks, but the protocol allowed for 10 mg or less per week in patients who could not tolerate higher doses.
In one of two primary endpoints, significantly more patients in the combination arm achieved remission (DAS28-CRP <2.6) at 12 months (60.9%), compared with patients who took methotrexate alone (45.2%), for an odds ratio of 2.01 (P = .010). The rate was 42.5% in the abatacept monotherapy arm, Dr. Paul Emery of the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England) and his colleagues reported (Ann. Rheum. Dis. 2014 Nov. 3 [doi:10.1136/annrheumdis-2014-206106]).
At the end of the first 12 months, patients who had achieved a DAS28-CRP score of less than 3.2 entered a 12-month treatment withdrawal period. Remission at both 12 and 18 months – the second primary endpoint – occurred in 14.8% of patients on combination therapy and 7.8% of those on methotrexate alone (OR, 2.51; P = .045). A total of 12.4% of abatacept monotherapy patients achieved remission at both 12 and 18 months.
The results suggest that in early RA drug-free remission may be possible following treatment with abatacept, the researchers said.
In the 12-month treatment period, the serious adverse events were reported in 12.1% taking abatacept monotherapy, 6.7% taking combination therapy, and 7.8% taking methotrexate alone.
The study was sponsored by Bristol-Myers Squibb, which manufactures abatacept (Orencia). Dr. Emery reported receiving consulting fees and grant support from Bristol-Myers Squibb as well as other companies that market drugs for RA. Many of his coauthors reported similar disclosures. Two authors are employees of Bristol-Myers Squibb.
Patients with early rheumatoid arthritis who achieved remission after 1 year of treatment with abatacept and methotrexate had a small but significantly greater rate of sustained remission for another 6 months following withdrawal of all drugs when compared with patients who took methotrexate alone in a randomized phase IIIb study.
The AVERT (Assessing Very Early Rheumatoid Arthritis Treatment) study randomized 351 early rheumatoid arthritis (RA) patients who were methotrexate naive, tested positive for anticitrullinated peptide–2 antibody, and had a 28-joint Disease Activity Score using C-reactive protein (DAS28-CRP) of more than 3.2 to weekly doses of 125 mg abatacept plus methotrexate, abatacept monotherapy, or methotrexate alone. Methotrexate was initiated at 7.5 mg/wk and was then titrated to 15-20 mg/wk in 6-8 weeks, but the protocol allowed for 10 mg or less per week in patients who could not tolerate higher doses.
In one of two primary endpoints, significantly more patients in the combination arm achieved remission (DAS28-CRP <2.6) at 12 months (60.9%), compared with patients who took methotrexate alone (45.2%), for an odds ratio of 2.01 (P = .010). The rate was 42.5% in the abatacept monotherapy arm, Dr. Paul Emery of the Leeds Institute of Rheumatic and Musculoskeletal Medicine at the University of Leeds (England) and his colleagues reported (Ann. Rheum. Dis. 2014 Nov. 3 [doi:10.1136/annrheumdis-2014-206106]).
At the end of the first 12 months, patients who had achieved a DAS28-CRP score of less than 3.2 entered a 12-month treatment withdrawal period. Remission at both 12 and 18 months – the second primary endpoint – occurred in 14.8% of patients on combination therapy and 7.8% of those on methotrexate alone (OR, 2.51; P = .045). A total of 12.4% of abatacept monotherapy patients achieved remission at both 12 and 18 months.
The results suggest that in early RA drug-free remission may be possible following treatment with abatacept, the researchers said.
In the 12-month treatment period, the serious adverse events were reported in 12.1% taking abatacept monotherapy, 6.7% taking combination therapy, and 7.8% taking methotrexate alone.
The study was sponsored by Bristol-Myers Squibb, which manufactures abatacept (Orencia). Dr. Emery reported receiving consulting fees and grant support from Bristol-Myers Squibb as well as other companies that market drugs for RA. Many of his coauthors reported similar disclosures. Two authors are employees of Bristol-Myers Squibb.
FROM ANNALS OF THE RHEUMATIC DISEASES
Key clinical point: Early RA drug-free remission may be possible following treatment with abatacept.
Major finding: About 15% of patients who were in remission after taking abatacept and methotrexate for 1 year continued to be in remission for another 6 months after stopping all drugs, compared with about 8% of those taking methotrexate alone.
Data source: A phase IIIb randomized study of 351 early RA patients who were methotrexate naive, were anti-CCP2 positive, and had DAS28 scores of more than 3.2.
Disclosures: The study was sponsored by Bristol-Myers Squibb, which manufactures abatacept (Orencia). Dr. Emery reported receiving consulting fees and grant support from Bristol-Myers Squibb as well as other companies that market drugs for RA. Many of his coauthors reported similar disclosures. Two authors are employees of Bristol-Myers Squibb.
Control of early RA maintained with reduced-dose etanercept plus methotrexate
For patients with early rheumatoid arthritis who achieved remission on full-dose etanercept plus methotrexate, continuing this combination treatment at a reduced dose controlled the disease better than did methotrexate alone or placebo in the randomized, controlled PRIZE trial.
The results of the PRIZE (Productivity and Remission in a Randomized Controlled Trial of Etanercept vs. Standard of Care in Early Rheumatoid Arthritis) trial, which was designed and funded by Pfizer, also showed that maintenance therapy with etanercept plus methotrexate increased the number of patients with sustained remission and extended the duration of remission after treatment was discontinued. However, it did not alter the radiographic progression of RA, said Dr. Paul Emery of the Leeds Institute of Rheumatic and Musculoskeletal Medicine at Leeds (England) University, and his associates.
The trial, conducted at 57 medical centers in Europe and Asia during 2009-2012, included 193 patients who responded to the combination therapy during a 1-year, open-label phase (N. Engl. J. Med. 2014;371:1781-92).
This open-label phase consisted of subcutaneous injections of 50 mg etanercept plus oral methotrexate initiated at a dose of 10 mg/week and adjusted up to a maximum dose of 25 mg/week during the first 8 weeks. Patients who did not have low disease activity, defined as a 28-joint Disease Activity Score (DAS28) of 3.2 or less, at week 13 or 26 received glucocorticoids as rescue medication unless contraindicated or if they had unacceptable side effects.
The patients who met the criteria for response during the open-label phase by having a DAS28 of 3.2 or less at week 39 and a DAS28 of less than 2.6 at 1 year were randomly assigned in a double-blind fashion to 39 weeks of 25 mg etanercept plus methotrexate (63 participants), placebo injection plus methotrexate (65 participants), or a double placebo (65 participants). Afterward, the study treatments were withdrawn and 131 participants were followed to week 65.
The primary efficacy endpoint of the trial was the proportion of patients who had sustained remission at the end of the double-blind maintenance phase. With DAS28-defined remission, this percentage was significantly higher for patients who received etanercept plus methotrexate (79%) than for those who received methotrexate alone (54%) or placebo alone (38%). When remission was defined by the stricter American College of Rheumatology and European League Against Rheumatism (ACR-EULAR) criteria, the differences were still significant: 68%, 46%, and 23%, respectively.
At this time, 78% of patients who received combination therapy achieved a normal score on the Health Assessment Questionnaire-Disability Index (HAQ-DI), compared with 72% of those who received methotrexate alone and 45% of those who received placebo. The difference was statistically significant between the combination-therapy group and the placebo group, the investigators said.
At week 65, which was 26 weeks after all study treatments had been withdrawn, a significantly higher percentage of patients in the combination-therapy group than in the methotrexate group or the placebo group met both sets of remission criteria and still had a normal HAQ-DI score, although the percentages between the two active treatment groups were not significantly different when examining only those who met DAS28 remission criteria or those who had a normal HAQ-DI score.
But patients in the combination-therapy group remained in remission significantly longer than did those in the other study groups, and their mean DAS28 scores were significantly lower.
However, there were no differences among the three study groups in radiographic progression of RA measured at the hands, wrists, and feet at the end of the double-blind phase.
Rates of serious adverse events were 5% with combination therapy, 3% with methotrexate alone, and 3% with placebo during the maintenance phase of the trial and 0%, 0%, and 6%, respectively, during the treatment-withdrawal phase.
This study was limited in that it included only patients newly diagnosed as having RA who had not received any other treatments. Therefore the findings may not be generalizable to patients with RA of longer duration and those who have undergone other treatments, Dr. Emery and his associates said.
This study was funded by Pfizer, maker of etanercept (Enbrel); Pfizer also designed the study, collected and analyzed the data, and participated in writing the report. Dr. Emery reported ties to AbbVie, Bristol-Myers Squibb, Pfizer, UCB, Merck Sharpe & Dohme, Roche, and Takeda, and his associates reported ties to numerous industry sources.
For patients with early rheumatoid arthritis who achieved remission on full-dose etanercept plus methotrexate, continuing this combination treatment at a reduced dose controlled the disease better than did methotrexate alone or placebo in the randomized, controlled PRIZE trial.
The results of the PRIZE (Productivity and Remission in a Randomized Controlled Trial of Etanercept vs. Standard of Care in Early Rheumatoid Arthritis) trial, which was designed and funded by Pfizer, also showed that maintenance therapy with etanercept plus methotrexate increased the number of patients with sustained remission and extended the duration of remission after treatment was discontinued. However, it did not alter the radiographic progression of RA, said Dr. Paul Emery of the Leeds Institute of Rheumatic and Musculoskeletal Medicine at Leeds (England) University, and his associates.
The trial, conducted at 57 medical centers in Europe and Asia during 2009-2012, included 193 patients who responded to the combination therapy during a 1-year, open-label phase (N. Engl. J. Med. 2014;371:1781-92).
This open-label phase consisted of subcutaneous injections of 50 mg etanercept plus oral methotrexate initiated at a dose of 10 mg/week and adjusted up to a maximum dose of 25 mg/week during the first 8 weeks. Patients who did not have low disease activity, defined as a 28-joint Disease Activity Score (DAS28) of 3.2 or less, at week 13 or 26 received glucocorticoids as rescue medication unless contraindicated or if they had unacceptable side effects.
The patients who met the criteria for response during the open-label phase by having a DAS28 of 3.2 or less at week 39 and a DAS28 of less than 2.6 at 1 year were randomly assigned in a double-blind fashion to 39 weeks of 25 mg etanercept plus methotrexate (63 participants), placebo injection plus methotrexate (65 participants), or a double placebo (65 participants). Afterward, the study treatments were withdrawn and 131 participants were followed to week 65.
The primary efficacy endpoint of the trial was the proportion of patients who had sustained remission at the end of the double-blind maintenance phase. With DAS28-defined remission, this percentage was significantly higher for patients who received etanercept plus methotrexate (79%) than for those who received methotrexate alone (54%) or placebo alone (38%). When remission was defined by the stricter American College of Rheumatology and European League Against Rheumatism (ACR-EULAR) criteria, the differences were still significant: 68%, 46%, and 23%, respectively.
At this time, 78% of patients who received combination therapy achieved a normal score on the Health Assessment Questionnaire-Disability Index (HAQ-DI), compared with 72% of those who received methotrexate alone and 45% of those who received placebo. The difference was statistically significant between the combination-therapy group and the placebo group, the investigators said.
At week 65, which was 26 weeks after all study treatments had been withdrawn, a significantly higher percentage of patients in the combination-therapy group than in the methotrexate group or the placebo group met both sets of remission criteria and still had a normal HAQ-DI score, although the percentages between the two active treatment groups were not significantly different when examining only those who met DAS28 remission criteria or those who had a normal HAQ-DI score.
But patients in the combination-therapy group remained in remission significantly longer than did those in the other study groups, and their mean DAS28 scores were significantly lower.
However, there were no differences among the three study groups in radiographic progression of RA measured at the hands, wrists, and feet at the end of the double-blind phase.
Rates of serious adverse events were 5% with combination therapy, 3% with methotrexate alone, and 3% with placebo during the maintenance phase of the trial and 0%, 0%, and 6%, respectively, during the treatment-withdrawal phase.
This study was limited in that it included only patients newly diagnosed as having RA who had not received any other treatments. Therefore the findings may not be generalizable to patients with RA of longer duration and those who have undergone other treatments, Dr. Emery and his associates said.
This study was funded by Pfizer, maker of etanercept (Enbrel); Pfizer also designed the study, collected and analyzed the data, and participated in writing the report. Dr. Emery reported ties to AbbVie, Bristol-Myers Squibb, Pfizer, UCB, Merck Sharpe & Dohme, Roche, and Takeda, and his associates reported ties to numerous industry sources.
For patients with early rheumatoid arthritis who achieved remission on full-dose etanercept plus methotrexate, continuing this combination treatment at a reduced dose controlled the disease better than did methotrexate alone or placebo in the randomized, controlled PRIZE trial.
The results of the PRIZE (Productivity and Remission in a Randomized Controlled Trial of Etanercept vs. Standard of Care in Early Rheumatoid Arthritis) trial, which was designed and funded by Pfizer, also showed that maintenance therapy with etanercept plus methotrexate increased the number of patients with sustained remission and extended the duration of remission after treatment was discontinued. However, it did not alter the radiographic progression of RA, said Dr. Paul Emery of the Leeds Institute of Rheumatic and Musculoskeletal Medicine at Leeds (England) University, and his associates.
The trial, conducted at 57 medical centers in Europe and Asia during 2009-2012, included 193 patients who responded to the combination therapy during a 1-year, open-label phase (N. Engl. J. Med. 2014;371:1781-92).
This open-label phase consisted of subcutaneous injections of 50 mg etanercept plus oral methotrexate initiated at a dose of 10 mg/week and adjusted up to a maximum dose of 25 mg/week during the first 8 weeks. Patients who did not have low disease activity, defined as a 28-joint Disease Activity Score (DAS28) of 3.2 or less, at week 13 or 26 received glucocorticoids as rescue medication unless contraindicated or if they had unacceptable side effects.
The patients who met the criteria for response during the open-label phase by having a DAS28 of 3.2 or less at week 39 and a DAS28 of less than 2.6 at 1 year were randomly assigned in a double-blind fashion to 39 weeks of 25 mg etanercept plus methotrexate (63 participants), placebo injection plus methotrexate (65 participants), or a double placebo (65 participants). Afterward, the study treatments were withdrawn and 131 participants were followed to week 65.
The primary efficacy endpoint of the trial was the proportion of patients who had sustained remission at the end of the double-blind maintenance phase. With DAS28-defined remission, this percentage was significantly higher for patients who received etanercept plus methotrexate (79%) than for those who received methotrexate alone (54%) or placebo alone (38%). When remission was defined by the stricter American College of Rheumatology and European League Against Rheumatism (ACR-EULAR) criteria, the differences were still significant: 68%, 46%, and 23%, respectively.
At this time, 78% of patients who received combination therapy achieved a normal score on the Health Assessment Questionnaire-Disability Index (HAQ-DI), compared with 72% of those who received methotrexate alone and 45% of those who received placebo. The difference was statistically significant between the combination-therapy group and the placebo group, the investigators said.
At week 65, which was 26 weeks after all study treatments had been withdrawn, a significantly higher percentage of patients in the combination-therapy group than in the methotrexate group or the placebo group met both sets of remission criteria and still had a normal HAQ-DI score, although the percentages between the two active treatment groups were not significantly different when examining only those who met DAS28 remission criteria or those who had a normal HAQ-DI score.
But patients in the combination-therapy group remained in remission significantly longer than did those in the other study groups, and their mean DAS28 scores were significantly lower.
However, there were no differences among the three study groups in radiographic progression of RA measured at the hands, wrists, and feet at the end of the double-blind phase.
Rates of serious adverse events were 5% with combination therapy, 3% with methotrexate alone, and 3% with placebo during the maintenance phase of the trial and 0%, 0%, and 6%, respectively, during the treatment-withdrawal phase.
This study was limited in that it included only patients newly diagnosed as having RA who had not received any other treatments. Therefore the findings may not be generalizable to patients with RA of longer duration and those who have undergone other treatments, Dr. Emery and his associates said.
This study was funded by Pfizer, maker of etanercept (Enbrel); Pfizer also designed the study, collected and analyzed the data, and participated in writing the report. Dr. Emery reported ties to AbbVie, Bristol-Myers Squibb, Pfizer, UCB, Merck Sharpe & Dohme, Roche, and Takeda, and his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Maintenance therapy with reduced-dose etanercept plus methotrexate controls RA better than methotrexate alone or placebo.
Major finding: The primary efficacy endpoint – the proportion of patients with sustained remission at 39 weeks – was significantly higher for patients who received etanercept plus methotrexate (79%) than for those who received methotrexate alone (54%) or placebo (38%) using DAS28 criteria.
Data source: The randomized, controlled PRIZE trial assessing the efficacy of reduced-dose etanercept as maintenance therapy in 193 adults with RA followed for 65 weeks.
Disclosures: This study was funded by Pfizer, maker of etanercept (Enbrel); Pfizer also designed the study, collected and analyzed the data, and participated in writing the report. Dr. Emery reported ties to AbbVie, Bristol-Myers Squibb, Pfizer, UCB, Merck Sharpe & Dohme, Roche, and Takeda, and his associates reported ties to numerous industry sources.
Current use of COX-2 inhibitors linked to increased mortality after ischemic stroke
Current use of cyclooxygenase-2 inhibitors was associated with an increase in 30-day mortality after ischemic stroke in a population-based cohort study published Nov. 5 in Neurology.
Since the association between COX-2 inhibitors and ischemic stroke mortality was associated only with current use and not former use, the researchers, led by Dr. Morten Schmidt of Aarhus (Denmark) University Hospital, believe that alternative treatment options – such as nonselective NSAIDs, which did not have an impact on overall mortality after ischemic stroke – may be more suitable for treating potential ischemic stroke patients, such as those with atrial fibrillation and a high CHA2DS2-VASc score.
If the association is truly causal, it constitutes a strong argument for increasing the efforts to ensure that patients with a high predicted risk of arterial thromboembolism are not prescribed COX-2 inhibitors when alternative treatment options are available,” Dr. Schmidt and his associates wrote.
In order to determine whether COX-2 inhibitors influenced 30-day mortality at the time of hospitalization for stroke, the researchers examined records of 100,243 people hospitalized for a first-time stroke in Denmark during 2004-2012 and deaths within 1 month after the stroke (Neurology 2014 Nov. 5 [doi:10.1212/WNL.0000000000001024]).
The hazard ratio for ischemic stroke was 1.19 (95% confidence interval, 1.02-1.38) for current users of COX-2 inhibitors, while current users of nonselective NSAIDs had an HR of 1.00 (95% CI, 0.87-1.15), compared with nonusers.
The COX-2 inhibitors in the study included diclofenac, etodolac, nabumeton, and meloxicam, as well as coxibs including celecoxib and rofecoxib. The nonselective NSAIDs in the study were ibuprofen, naproxen, ketoprofen, dexibuprofen, piroxicam, tolfenamic acid, and indomethacin.
Though the researchers acknowledged more studies are needed to truly examine the effects of COX-2 inhibitors on stroke mortality, they hypothesized that the increased mortality rate may be caused by COX-2 inhibition interfering with the pathophysiologic response to a stroke, or unwanted effects from the thromboembolic properties of COX-2 inhibitors.
“Our study adds to the increasing body of evidence concerning the vascular risk and prognostic impact associated with use of COX-2 inhibitors,” Dr. Schmidt and his associates wrote.
The study was funded by several Danish research foundations and the Program for Clinical Research Infrastructure, which was established by the Lundbeck Foundation and the Novo Nordisk Foundation. The authors reported no relevant disclosures.
Current use of cyclooxygenase-2 inhibitors was associated with an increase in 30-day mortality after ischemic stroke in a population-based cohort study published Nov. 5 in Neurology.
Since the association between COX-2 inhibitors and ischemic stroke mortality was associated only with current use and not former use, the researchers, led by Dr. Morten Schmidt of Aarhus (Denmark) University Hospital, believe that alternative treatment options – such as nonselective NSAIDs, which did not have an impact on overall mortality after ischemic stroke – may be more suitable for treating potential ischemic stroke patients, such as those with atrial fibrillation and a high CHA2DS2-VASc score.
If the association is truly causal, it constitutes a strong argument for increasing the efforts to ensure that patients with a high predicted risk of arterial thromboembolism are not prescribed COX-2 inhibitors when alternative treatment options are available,” Dr. Schmidt and his associates wrote.
In order to determine whether COX-2 inhibitors influenced 30-day mortality at the time of hospitalization for stroke, the researchers examined records of 100,243 people hospitalized for a first-time stroke in Denmark during 2004-2012 and deaths within 1 month after the stroke (Neurology 2014 Nov. 5 [doi:10.1212/WNL.0000000000001024]).
The hazard ratio for ischemic stroke was 1.19 (95% confidence interval, 1.02-1.38) for current users of COX-2 inhibitors, while current users of nonselective NSAIDs had an HR of 1.00 (95% CI, 0.87-1.15), compared with nonusers.
The COX-2 inhibitors in the study included diclofenac, etodolac, nabumeton, and meloxicam, as well as coxibs including celecoxib and rofecoxib. The nonselective NSAIDs in the study were ibuprofen, naproxen, ketoprofen, dexibuprofen, piroxicam, tolfenamic acid, and indomethacin.
Though the researchers acknowledged more studies are needed to truly examine the effects of COX-2 inhibitors on stroke mortality, they hypothesized that the increased mortality rate may be caused by COX-2 inhibition interfering with the pathophysiologic response to a stroke, or unwanted effects from the thromboembolic properties of COX-2 inhibitors.
“Our study adds to the increasing body of evidence concerning the vascular risk and prognostic impact associated with use of COX-2 inhibitors,” Dr. Schmidt and his associates wrote.
The study was funded by several Danish research foundations and the Program for Clinical Research Infrastructure, which was established by the Lundbeck Foundation and the Novo Nordisk Foundation. The authors reported no relevant disclosures.
Current use of cyclooxygenase-2 inhibitors was associated with an increase in 30-day mortality after ischemic stroke in a population-based cohort study published Nov. 5 in Neurology.
Since the association between COX-2 inhibitors and ischemic stroke mortality was associated only with current use and not former use, the researchers, led by Dr. Morten Schmidt of Aarhus (Denmark) University Hospital, believe that alternative treatment options – such as nonselective NSAIDs, which did not have an impact on overall mortality after ischemic stroke – may be more suitable for treating potential ischemic stroke patients, such as those with atrial fibrillation and a high CHA2DS2-VASc score.
If the association is truly causal, it constitutes a strong argument for increasing the efforts to ensure that patients with a high predicted risk of arterial thromboembolism are not prescribed COX-2 inhibitors when alternative treatment options are available,” Dr. Schmidt and his associates wrote.
In order to determine whether COX-2 inhibitors influenced 30-day mortality at the time of hospitalization for stroke, the researchers examined records of 100,243 people hospitalized for a first-time stroke in Denmark during 2004-2012 and deaths within 1 month after the stroke (Neurology 2014 Nov. 5 [doi:10.1212/WNL.0000000000001024]).
The hazard ratio for ischemic stroke was 1.19 (95% confidence interval, 1.02-1.38) for current users of COX-2 inhibitors, while current users of nonselective NSAIDs had an HR of 1.00 (95% CI, 0.87-1.15), compared with nonusers.
The COX-2 inhibitors in the study included diclofenac, etodolac, nabumeton, and meloxicam, as well as coxibs including celecoxib and rofecoxib. The nonselective NSAIDs in the study were ibuprofen, naproxen, ketoprofen, dexibuprofen, piroxicam, tolfenamic acid, and indomethacin.
Though the researchers acknowledged more studies are needed to truly examine the effects of COX-2 inhibitors on stroke mortality, they hypothesized that the increased mortality rate may be caused by COX-2 inhibition interfering with the pathophysiologic response to a stroke, or unwanted effects from the thromboembolic properties of COX-2 inhibitors.
“Our study adds to the increasing body of evidence concerning the vascular risk and prognostic impact associated with use of COX-2 inhibitors,” Dr. Schmidt and his associates wrote.
The study was funded by several Danish research foundations and the Program for Clinical Research Infrastructure, which was established by the Lundbeck Foundation and the Novo Nordisk Foundation. The authors reported no relevant disclosures.
FROM NEUROLOGY
Key clinical point:Consideration should be given to using treatment options other than COX-2 inhibitors in patients with a high future risk of arterial thromboembolism.
Major finding: Preadmission use of COX-2 inhibitors was associated with increased 30-day mortality after ischemic stroke 1.19 (95% CI, 1.02-1.38), compared with nonusers.
Data source:Population-based cohort study of 100,043 patients from Denmark with first-time stroke.
Disclosures:The study was funded by several Danish research foundations and the Program for Clinical Research Infrastructure, which was established by the Lundbeck Foundation and the Novo Nordisk Foundation. The authors reported no relevant disclosures.
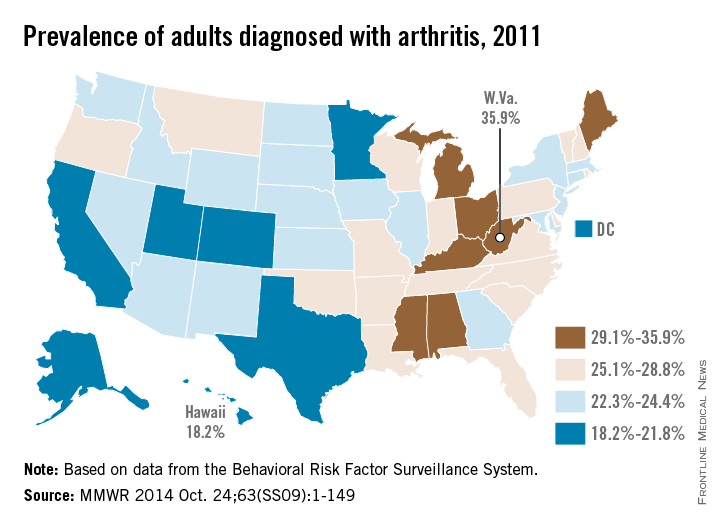
Eastern United States has the highest arthritis rates
The prevalence of arthritis tended to be higher in eastern U.S. states than in western states in 2011, according to a report from the Centers for Disease Control and Prevention.
All seven states with an arthritis rate below 22% were located west of the Mississippi River, with Hawaii having the lowest rate at 18.2%, followed by Utah (19.8%) and Texas (20.2%). The District of Columbia, which is in the East, had an arthritis rate of 20.9%.
The eight states with an arthritis rate greater than 29% were all east of the Mississippi, with West Virginia having the highest prevalence (35.9%), followed by Kentucky (31.9%) and Michigan (31.0%), according to the report (MMWR 2014;63[SS09]:1-149).
Of 198 reported metropolitan and micropolitan statistical areas (MMSAs), 29 had an arthritis prevalence lower than 20%. Lawrence, Kan., had the lowest rate at 13.5%. Of the 29, only Atlanta; Knoxville, Tenn.; and Raleigh, N.C., are located entirely east of the Mississippi River. Kingsport-Bristol, in Tennessee and Virginia, had the highest arthritis rate at 37%. Of the 16 MMSAs with an arthritis rate greater than 30%, North Platte, Neb., was the only one west of the Mississippi, according to data from the Behavioral Risk Factor Surveillance System.
The prevalence of arthritis tended to be higher in eastern U.S. states than in western states in 2011, according to a report from the Centers for Disease Control and Prevention.
All seven states with an arthritis rate below 22% were located west of the Mississippi River, with Hawaii having the lowest rate at 18.2%, followed by Utah (19.8%) and Texas (20.2%). The District of Columbia, which is in the East, had an arthritis rate of 20.9%.
The eight states with an arthritis rate greater than 29% were all east of the Mississippi, with West Virginia having the highest prevalence (35.9%), followed by Kentucky (31.9%) and Michigan (31.0%), according to the report (MMWR 2014;63[SS09]:1-149).
Of 198 reported metropolitan and micropolitan statistical areas (MMSAs), 29 had an arthritis prevalence lower than 20%. Lawrence, Kan., had the lowest rate at 13.5%. Of the 29, only Atlanta; Knoxville, Tenn.; and Raleigh, N.C., are located entirely east of the Mississippi River. Kingsport-Bristol, in Tennessee and Virginia, had the highest arthritis rate at 37%. Of the 16 MMSAs with an arthritis rate greater than 30%, North Platte, Neb., was the only one west of the Mississippi, according to data from the Behavioral Risk Factor Surveillance System.
The prevalence of arthritis tended to be higher in eastern U.S. states than in western states in 2011, according to a report from the Centers for Disease Control and Prevention.
All seven states with an arthritis rate below 22% were located west of the Mississippi River, with Hawaii having the lowest rate at 18.2%, followed by Utah (19.8%) and Texas (20.2%). The District of Columbia, which is in the East, had an arthritis rate of 20.9%.
The eight states with an arthritis rate greater than 29% were all east of the Mississippi, with West Virginia having the highest prevalence (35.9%), followed by Kentucky (31.9%) and Michigan (31.0%), according to the report (MMWR 2014;63[SS09]:1-149).
Of 198 reported metropolitan and micropolitan statistical areas (MMSAs), 29 had an arthritis prevalence lower than 20%. Lawrence, Kan., had the lowest rate at 13.5%. Of the 29, only Atlanta; Knoxville, Tenn.; and Raleigh, N.C., are located entirely east of the Mississippi River. Kingsport-Bristol, in Tennessee and Virginia, had the highest arthritis rate at 37%. Of the 16 MMSAs with an arthritis rate greater than 30%, North Platte, Neb., was the only one west of the Mississippi, according to data from the Behavioral Risk Factor Surveillance System.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Disease activity, not lipid levels, appear to affect CV risk with tocilizumab
Rheumatoid arthritis disease activity during treatment with tocilizumab plays a significant role in patients’ risk for major adverse cardiovascular events, rather than increases in lipid levels observed during treatment with the interleukin-6 receptor inhibitor, according to a new analysis of phase III trials and extensions studies of the drug.
The study of 3,986 rheumatoid arthritis (RA) patients treated with tocilizumab for a mean of 3.68 years indicated that reductions in disease activity based on lower 28-joint disease activity score (DAS28) were significantly associated with fewer major adverse cardiovascular events (MACE), defined as definite or probable nonfatal myocardial infarction, nonfatal stroke, or death from a cardiovascular cause.
Conversely, risk for MACE rose with poorer response to treatment, as measured by increases in DAS28 and total swollen and tender joints (Arthritis Rheumatol. 2014 [doi:10.1002/art.38920]).
In addition to DAS28, some traditional risk factors at baseline – older age, history of cardiac disorders, and high ratio of total cholesterol to HDL cholesterol (known as the atherogenic index) – were independent predictors of MACE during treatment, reported Dr. Vijay U. Rao and his colleagues. Dr. Rao was a research fellow with Genentech during the study but is now a cardiologist in private practice in Indianapolis.
The presence of a cardiac disorder increased the risk of MACE, with a hazard ratio of 2.32, and for each 1-unit increase in DAS28, age, and atherogenic index, MACE risk rose by 36%, 7%, and 33%, respectively.
Predictors of future risk of MACE following the trials’ initial 24-week treatment period remained largely the same as baseline predictors for MACE during initial treatment, including measures of higher disease activity.
In contrast, baseline and 24-week levels of systemic markers of inflammation such as erythrocyte sedimentation rate (ESR) and C-reactive protein, as well as changes in ESR and interleukin-6 (IL-6) receptor levels and changes in lipid levels during 24 weeks of tocilizumab treatment, did not predict future risk of MACE.
The IL-6 receptor pathway is thought to play a role in cardiovascular disease, and tocilizumab’s action as a monoclonal antibody against that receptor suggested that its effects might reduce CV risk. However, baseline levels of IL-6 and IL-6 receptor, or levels of IL-6 receptor at 24 weeks, were not associated with MACE.
The investigators found 50 confirmed cases of MACE in pooled data from five randomized, controlled trials and their extension studies that enrolled 3,986 patients with moderate to severe RA who received tocilizumab 4 mg/kg or 8 mg/kg intravenously every 4 weeks in combination with methotrexate or other disease-modifying antirheumatic drugs. The MACE rate was 3.4/1,000 patient-years of exposure, and there were no obvious time trends observed.
Because the trials used in the post hoc analyses were not specifically powered for analyzing MACE risk, the findings should be viewed as “hypothesis generating,” the investigators said.
They also noted that because 31% of patients in the trials discontinued treatment and MACE cases that occurred beyond 6 months after discontinuation were not recorded, there is a chance that those who withdrew were at a different risk for MACE than were those who remained on treatment. They also did not evaluate hypertension and use of statins, antiplatelet medications, or glucocorticoids.
The findings support a previous registry study in which MI risk was halved in responders to antitumor necrosis factor-alpha therapy, compared with nonresponders, according to the authors, as well as in several observational studies in which control of disease activity with nonbiologic DMARDs was associated with lower risk for MI.
The study was funded by F. Hoffmann-La Roche, of which the manufacturer of tocilizumab, Genentech, is a subsidiary. Five of the nine authors were employed by or received funding from Roche or Genentech at the time of the study, two were consultants or received speaking fees from Roche or Genentech, and the remaining two had no disclosures.

|
Dr. Katherine P. Liao |
Dr. Rao and his colleagues’ analysis is “reassuring” in showing a lack of cardiovascular events seen in the pooled data for tocilizumab, but it “cannot provide definitive answers regarding the safety of tocilizumab,” according to Dr. Katherine P. Liao and Dr. Daniel H. Solomon.
An ongoing, randomized, open-label study of CV events in rheumatoid arthritis patients on tocilizumab and etanercept, as well as observational studies comparing tocilizumab and other biologic DMARDs, should provide more important safety information, they noted.
The pooled data analysis highlights additional evidence to support RA disease activity as an important CV risk factor. And it provides additional evidence showing that total cholesterol and LDL cholesterol “may be suboptimal measures to guide CV risk estimation and preventative interventions, since levels appear to fluctuate with therapy in an inverse manner,” Dr. Liao and Dr. Solomon wrote.

|
Dr. Daniel H. Solomon |
Studies that use advanced lipoprotein tests and those that more closely examine the “underlying mechanisms by which inflammation elevates CV risk in RA” have the potential to improve the accuracy of CV risk assessment in RA, they wrote.
Dr. Liao and Dr. Solomon are with the division of rheumatology, immunology, and allergy at Brigham and Women’s Hospital, Boston. Their comments are taken from an editorial accompanying the study by Dr. Rao and his associates (Arthritis Rheumatol. 2014 [doi:10.1002/art.38925]). Dr. Liao said that she has no disclosures. Dr. Solomon reported receiving research funding through Brigham and Women’s Hospital from Amgen, Lilly, and Pfizer, and has unpaid roles on trials funded by Bristol-Myers Squibb and Pfizer.

|
Dr. Katherine P. Liao |
Dr. Rao and his colleagues’ analysis is “reassuring” in showing a lack of cardiovascular events seen in the pooled data for tocilizumab, but it “cannot provide definitive answers regarding the safety of tocilizumab,” according to Dr. Katherine P. Liao and Dr. Daniel H. Solomon.
An ongoing, randomized, open-label study of CV events in rheumatoid arthritis patients on tocilizumab and etanercept, as well as observational studies comparing tocilizumab and other biologic DMARDs, should provide more important safety information, they noted.
The pooled data analysis highlights additional evidence to support RA disease activity as an important CV risk factor. And it provides additional evidence showing that total cholesterol and LDL cholesterol “may be suboptimal measures to guide CV risk estimation and preventative interventions, since levels appear to fluctuate with therapy in an inverse manner,” Dr. Liao and Dr. Solomon wrote.

|
Dr. Daniel H. Solomon |
Studies that use advanced lipoprotein tests and those that more closely examine the “underlying mechanisms by which inflammation elevates CV risk in RA” have the potential to improve the accuracy of CV risk assessment in RA, they wrote.
Dr. Liao and Dr. Solomon are with the division of rheumatology, immunology, and allergy at Brigham and Women’s Hospital, Boston. Their comments are taken from an editorial accompanying the study by Dr. Rao and his associates (Arthritis Rheumatol. 2014 [doi:10.1002/art.38925]). Dr. Liao said that she has no disclosures. Dr. Solomon reported receiving research funding through Brigham and Women’s Hospital from Amgen, Lilly, and Pfizer, and has unpaid roles on trials funded by Bristol-Myers Squibb and Pfizer.

|
Dr. Katherine P. Liao |
Dr. Rao and his colleagues’ analysis is “reassuring” in showing a lack of cardiovascular events seen in the pooled data for tocilizumab, but it “cannot provide definitive answers regarding the safety of tocilizumab,” according to Dr. Katherine P. Liao and Dr. Daniel H. Solomon.
An ongoing, randomized, open-label study of CV events in rheumatoid arthritis patients on tocilizumab and etanercept, as well as observational studies comparing tocilizumab and other biologic DMARDs, should provide more important safety information, they noted.
The pooled data analysis highlights additional evidence to support RA disease activity as an important CV risk factor. And it provides additional evidence showing that total cholesterol and LDL cholesterol “may be suboptimal measures to guide CV risk estimation and preventative interventions, since levels appear to fluctuate with therapy in an inverse manner,” Dr. Liao and Dr. Solomon wrote.

|
Dr. Daniel H. Solomon |
Studies that use advanced lipoprotein tests and those that more closely examine the “underlying mechanisms by which inflammation elevates CV risk in RA” have the potential to improve the accuracy of CV risk assessment in RA, they wrote.
Dr. Liao and Dr. Solomon are with the division of rheumatology, immunology, and allergy at Brigham and Women’s Hospital, Boston. Their comments are taken from an editorial accompanying the study by Dr. Rao and his associates (Arthritis Rheumatol. 2014 [doi:10.1002/art.38925]). Dr. Liao said that she has no disclosures. Dr. Solomon reported receiving research funding through Brigham and Women’s Hospital from Amgen, Lilly, and Pfizer, and has unpaid roles on trials funded by Bristol-Myers Squibb and Pfizer.
Rheumatoid arthritis disease activity during treatment with tocilizumab plays a significant role in patients’ risk for major adverse cardiovascular events, rather than increases in lipid levels observed during treatment with the interleukin-6 receptor inhibitor, according to a new analysis of phase III trials and extensions studies of the drug.
The study of 3,986 rheumatoid arthritis (RA) patients treated with tocilizumab for a mean of 3.68 years indicated that reductions in disease activity based on lower 28-joint disease activity score (DAS28) were significantly associated with fewer major adverse cardiovascular events (MACE), defined as definite or probable nonfatal myocardial infarction, nonfatal stroke, or death from a cardiovascular cause.
Conversely, risk for MACE rose with poorer response to treatment, as measured by increases in DAS28 and total swollen and tender joints (Arthritis Rheumatol. 2014 [doi:10.1002/art.38920]).
In addition to DAS28, some traditional risk factors at baseline – older age, history of cardiac disorders, and high ratio of total cholesterol to HDL cholesterol (known as the atherogenic index) – were independent predictors of MACE during treatment, reported Dr. Vijay U. Rao and his colleagues. Dr. Rao was a research fellow with Genentech during the study but is now a cardiologist in private practice in Indianapolis.
The presence of a cardiac disorder increased the risk of MACE, with a hazard ratio of 2.32, and for each 1-unit increase in DAS28, age, and atherogenic index, MACE risk rose by 36%, 7%, and 33%, respectively.
Predictors of future risk of MACE following the trials’ initial 24-week treatment period remained largely the same as baseline predictors for MACE during initial treatment, including measures of higher disease activity.
In contrast, baseline and 24-week levels of systemic markers of inflammation such as erythrocyte sedimentation rate (ESR) and C-reactive protein, as well as changes in ESR and interleukin-6 (IL-6) receptor levels and changes in lipid levels during 24 weeks of tocilizumab treatment, did not predict future risk of MACE.
The IL-6 receptor pathway is thought to play a role in cardiovascular disease, and tocilizumab’s action as a monoclonal antibody against that receptor suggested that its effects might reduce CV risk. However, baseline levels of IL-6 and IL-6 receptor, or levels of IL-6 receptor at 24 weeks, were not associated with MACE.
The investigators found 50 confirmed cases of MACE in pooled data from five randomized, controlled trials and their extension studies that enrolled 3,986 patients with moderate to severe RA who received tocilizumab 4 mg/kg or 8 mg/kg intravenously every 4 weeks in combination with methotrexate or other disease-modifying antirheumatic drugs. The MACE rate was 3.4/1,000 patient-years of exposure, and there were no obvious time trends observed.
Because the trials used in the post hoc analyses were not specifically powered for analyzing MACE risk, the findings should be viewed as “hypothesis generating,” the investigators said.
They also noted that because 31% of patients in the trials discontinued treatment and MACE cases that occurred beyond 6 months after discontinuation were not recorded, there is a chance that those who withdrew were at a different risk for MACE than were those who remained on treatment. They also did not evaluate hypertension and use of statins, antiplatelet medications, or glucocorticoids.
The findings support a previous registry study in which MI risk was halved in responders to antitumor necrosis factor-alpha therapy, compared with nonresponders, according to the authors, as well as in several observational studies in which control of disease activity with nonbiologic DMARDs was associated with lower risk for MI.
The study was funded by F. Hoffmann-La Roche, of which the manufacturer of tocilizumab, Genentech, is a subsidiary. Five of the nine authors were employed by or received funding from Roche or Genentech at the time of the study, two were consultants or received speaking fees from Roche or Genentech, and the remaining two had no disclosures.
Rheumatoid arthritis disease activity during treatment with tocilizumab plays a significant role in patients’ risk for major adverse cardiovascular events, rather than increases in lipid levels observed during treatment with the interleukin-6 receptor inhibitor, according to a new analysis of phase III trials and extensions studies of the drug.
The study of 3,986 rheumatoid arthritis (RA) patients treated with tocilizumab for a mean of 3.68 years indicated that reductions in disease activity based on lower 28-joint disease activity score (DAS28) were significantly associated with fewer major adverse cardiovascular events (MACE), defined as definite or probable nonfatal myocardial infarction, nonfatal stroke, or death from a cardiovascular cause.
Conversely, risk for MACE rose with poorer response to treatment, as measured by increases in DAS28 and total swollen and tender joints (Arthritis Rheumatol. 2014 [doi:10.1002/art.38920]).
In addition to DAS28, some traditional risk factors at baseline – older age, history of cardiac disorders, and high ratio of total cholesterol to HDL cholesterol (known as the atherogenic index) – were independent predictors of MACE during treatment, reported Dr. Vijay U. Rao and his colleagues. Dr. Rao was a research fellow with Genentech during the study but is now a cardiologist in private practice in Indianapolis.
The presence of a cardiac disorder increased the risk of MACE, with a hazard ratio of 2.32, and for each 1-unit increase in DAS28, age, and atherogenic index, MACE risk rose by 36%, 7%, and 33%, respectively.
Predictors of future risk of MACE following the trials’ initial 24-week treatment period remained largely the same as baseline predictors for MACE during initial treatment, including measures of higher disease activity.
In contrast, baseline and 24-week levels of systemic markers of inflammation such as erythrocyte sedimentation rate (ESR) and C-reactive protein, as well as changes in ESR and interleukin-6 (IL-6) receptor levels and changes in lipid levels during 24 weeks of tocilizumab treatment, did not predict future risk of MACE.
The IL-6 receptor pathway is thought to play a role in cardiovascular disease, and tocilizumab’s action as a monoclonal antibody against that receptor suggested that its effects might reduce CV risk. However, baseline levels of IL-6 and IL-6 receptor, or levels of IL-6 receptor at 24 weeks, were not associated with MACE.
The investigators found 50 confirmed cases of MACE in pooled data from five randomized, controlled trials and their extension studies that enrolled 3,986 patients with moderate to severe RA who received tocilizumab 4 mg/kg or 8 mg/kg intravenously every 4 weeks in combination with methotrexate or other disease-modifying antirheumatic drugs. The MACE rate was 3.4/1,000 patient-years of exposure, and there were no obvious time trends observed.
Because the trials used in the post hoc analyses were not specifically powered for analyzing MACE risk, the findings should be viewed as “hypothesis generating,” the investigators said.
They also noted that because 31% of patients in the trials discontinued treatment and MACE cases that occurred beyond 6 months after discontinuation were not recorded, there is a chance that those who withdrew were at a different risk for MACE than were those who remained on treatment. They also did not evaluate hypertension and use of statins, antiplatelet medications, or glucocorticoids.
The findings support a previous registry study in which MI risk was halved in responders to antitumor necrosis factor-alpha therapy, compared with nonresponders, according to the authors, as well as in several observational studies in which control of disease activity with nonbiologic DMARDs was associated with lower risk for MI.
The study was funded by F. Hoffmann-La Roche, of which the manufacturer of tocilizumab, Genentech, is a subsidiary. Five of the nine authors were employed by or received funding from Roche or Genentech at the time of the study, two were consultants or received speaking fees from Roche or Genentech, and the remaining two had no disclosures.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Disease activity, rather than lipid levels, is an independent predictor of cardiovascular event risk during treatment with tocilizumab.
Major finding: The presence of a cardiac disorder increased the risk of MACE, with a hazard ratio of 2.32; for each 1-unit increase in DAS28, age, and atherogenic index, MACE risk rose by 36%, 7%, and 33%, respectively.
Data source: A post hoc analysis of 3,986 patients with rheumatoid arthritis from five tocilizumab randomized, controlled trials and extension studies.
Disclosures: The study was funded by F. Hoffmann-La Roche, of which the manufacturer of tocilizumab, Genentech, is a subsidiary. Five of the nine authors were employed by or received funding from Roche or Genentech at the time of the study, two were consultants or received speaking fees from Roche or Genentech, and the remaining two had no disclosures.