User login
Strong evidence for tuberculosis screening before psoriasis, PsA biologic therapy
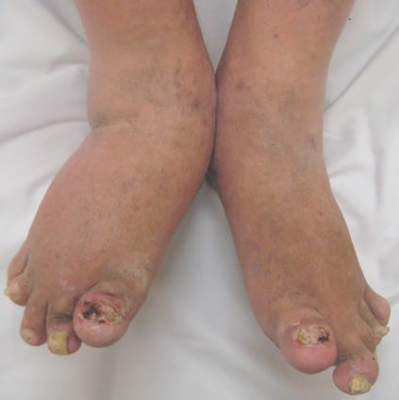
Tuberculosis is the only condition in patients with psoriasis or psoriatic arthritis who are being treated with systemic biologic agents for which there is strong evidence in favor of baseline screening, according to a literature review.
Analysis of 26 studies of systemic biologic treatments and screening tests – 13 of which included patients with hepatitis C, hepatitis B, or congestive heart failure – found the highest level evidence (grade B) in favor of tuberculin skin testing or interferon-gamma release assay, with the latter being preferred for its higher sensitivity and specificity.
“Based on the [U.S. Preventive Services Task Force] grading system, it is recommended this screen be provided because there is high certainty that the net benefit is moderate, or medium certainty that the net benefit is moderate to substantial,” wrote Dr. Christine S. Ahn of Wake Forest University, Winston-Salem, N.C., and coauthors in the Journal of the American Academy of Dermatology.
Screening for hepatitis B or C infection only was supported by grade-C evidence, and there was insufficient evidence for HIV screening, with the authors suggesting that selective screening should be performed based on professional judgment, clinical context, or patient preference (J Am Acad Dermatol. 2015 July 14 doi: 10.1016/j.jaad.2015.06.004).
Similarly, there was insufficient evidence to support complete blood cell count screening, and, given the low grade evidence on monitoring hepatic function, the authors suggested this should be performed at the clinician’s discretion, particularly among patients treated with infliximab.
Tuberculosis is the only condition in patients with psoriasis or psoriatic arthritis who are being treated with systemic biologic agents for which there is strong evidence in favor of baseline screening, according to a literature review.
Analysis of 26 studies of systemic biologic treatments and screening tests – 13 of which included patients with hepatitis C, hepatitis B, or congestive heart failure – found the highest level evidence (grade B) in favor of tuberculin skin testing or interferon-gamma release assay, with the latter being preferred for its higher sensitivity and specificity.
“Based on the [U.S. Preventive Services Task Force] grading system, it is recommended this screen be provided because there is high certainty that the net benefit is moderate, or medium certainty that the net benefit is moderate to substantial,” wrote Dr. Christine S. Ahn of Wake Forest University, Winston-Salem, N.C., and coauthors in the Journal of the American Academy of Dermatology.
Screening for hepatitis B or C infection only was supported by grade-C evidence, and there was insufficient evidence for HIV screening, with the authors suggesting that selective screening should be performed based on professional judgment, clinical context, or patient preference (J Am Acad Dermatol. 2015 July 14 doi: 10.1016/j.jaad.2015.06.004).
Similarly, there was insufficient evidence to support complete blood cell count screening, and, given the low grade evidence on monitoring hepatic function, the authors suggested this should be performed at the clinician’s discretion, particularly among patients treated with infliximab.
Tuberculosis is the only condition in patients with psoriasis or psoriatic arthritis who are being treated with systemic biologic agents for which there is strong evidence in favor of baseline screening, according to a literature review.
Analysis of 26 studies of systemic biologic treatments and screening tests – 13 of which included patients with hepatitis C, hepatitis B, or congestive heart failure – found the highest level evidence (grade B) in favor of tuberculin skin testing or interferon-gamma release assay, with the latter being preferred for its higher sensitivity and specificity.
“Based on the [U.S. Preventive Services Task Force] grading system, it is recommended this screen be provided because there is high certainty that the net benefit is moderate, or medium certainty that the net benefit is moderate to substantial,” wrote Dr. Christine S. Ahn of Wake Forest University, Winston-Salem, N.C., and coauthors in the Journal of the American Academy of Dermatology.
Screening for hepatitis B or C infection only was supported by grade-C evidence, and there was insufficient evidence for HIV screening, with the authors suggesting that selective screening should be performed based on professional judgment, clinical context, or patient preference (J Am Acad Dermatol. 2015 July 14 doi: 10.1016/j.jaad.2015.06.004).
Similarly, there was insufficient evidence to support complete blood cell count screening, and, given the low grade evidence on monitoring hepatic function, the authors suggested this should be performed at the clinician’s discretion, particularly among patients treated with infliximab.
FROM JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point:Tuberculosis is the only condition with strong evidence in favor of a baseline screening in patients with psoriasis or psoriatic arthritis being treated with systemic biologic agents.
Major finding: There is grade B–level evidence in favor of baseline tuberculosis screening in patients undergoing systemic biologic agent therapy, but only grade-C evidence for hepatitis B or C screening.
Data source: Analysis of 26 studies of systemic biologic treatments and screening tests.
Disclosures: One author declared consultancies, speaking engagements, grants, and stock options from a range of pharmaceutical companies. There were no other conflicts of interest declared.
Reciprocal risks found between psoriatic disease and uveitis
Researchers have uncovered a bidirectional relationship between psoriatic disease – psoriasis and psoriatic arthritis – and uveitis, with either condition significantly increasing the risk of the other.
A Danish nationwide cohort study of 74,129 individuals with psoriasis, including 6,735 with psoriatic arthritis, showed that patients with mild psoriasis had a 38% increased risk of uveitis, while those with psoriatic arthritis had a 2.5-fold increase in risk and those with psoriatic spondylitis had a greater than eight-fold increased risk, according to a paper published online in JAMA Dermatology.
There was a nonsignificant increase in the risk of uveitis in patients with severe psoriasis, but this failed to reach significance because of the small number of patients.
Similarly, patients with uveitis had a 59% greater risk of mild psoriasis, a twofold greater risk of severe psoriasis, a nearly fourfold increase in the risk of psoriatic arthritis, and an eightfold increase in the risk of psoriatic spondylitis (JAMA Dermatol. 2015 July 29 doi: 10.1001/jamadermatol.2015.1986).
The authors suggested that an increased focus on eye symptoms in patients with psoriasis and psoriatic arthritis and on skin and joint symptoms in patients with uveitis may be appropriate.
“The bidirectional relationship between psoriasis and psoriatic arthritis and uveitis suggests a shared pathogenic pathway, and increased systemic inflammation may contribute to the observed relationship,” wrote Dr. Alexander Egeberg of Pfizer and his coauthors. Dr. Egeberg was at the University of Copenhagen, Hellerup, when the study was conducted.
The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is employed by Eli Lilly. No other conflicts of interest were declared.
Researchers have uncovered a bidirectional relationship between psoriatic disease – psoriasis and psoriatic arthritis – and uveitis, with either condition significantly increasing the risk of the other.
A Danish nationwide cohort study of 74,129 individuals with psoriasis, including 6,735 with psoriatic arthritis, showed that patients with mild psoriasis had a 38% increased risk of uveitis, while those with psoriatic arthritis had a 2.5-fold increase in risk and those with psoriatic spondylitis had a greater than eight-fold increased risk, according to a paper published online in JAMA Dermatology.
There was a nonsignificant increase in the risk of uveitis in patients with severe psoriasis, but this failed to reach significance because of the small number of patients.
Similarly, patients with uveitis had a 59% greater risk of mild psoriasis, a twofold greater risk of severe psoriasis, a nearly fourfold increase in the risk of psoriatic arthritis, and an eightfold increase in the risk of psoriatic spondylitis (JAMA Dermatol. 2015 July 29 doi: 10.1001/jamadermatol.2015.1986).
The authors suggested that an increased focus on eye symptoms in patients with psoriasis and psoriatic arthritis and on skin and joint symptoms in patients with uveitis may be appropriate.
“The bidirectional relationship between psoriasis and psoriatic arthritis and uveitis suggests a shared pathogenic pathway, and increased systemic inflammation may contribute to the observed relationship,” wrote Dr. Alexander Egeberg of Pfizer and his coauthors. Dr. Egeberg was at the University of Copenhagen, Hellerup, when the study was conducted.
The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is employed by Eli Lilly. No other conflicts of interest were declared.
Researchers have uncovered a bidirectional relationship between psoriatic disease – psoriasis and psoriatic arthritis – and uveitis, with either condition significantly increasing the risk of the other.
A Danish nationwide cohort study of 74,129 individuals with psoriasis, including 6,735 with psoriatic arthritis, showed that patients with mild psoriasis had a 38% increased risk of uveitis, while those with psoriatic arthritis had a 2.5-fold increase in risk and those with psoriatic spondylitis had a greater than eight-fold increased risk, according to a paper published online in JAMA Dermatology.
There was a nonsignificant increase in the risk of uveitis in patients with severe psoriasis, but this failed to reach significance because of the small number of patients.
Similarly, patients with uveitis had a 59% greater risk of mild psoriasis, a twofold greater risk of severe psoriasis, a nearly fourfold increase in the risk of psoriatic arthritis, and an eightfold increase in the risk of psoriatic spondylitis (JAMA Dermatol. 2015 July 29 doi: 10.1001/jamadermatol.2015.1986).
The authors suggested that an increased focus on eye symptoms in patients with psoriasis and psoriatic arthritis and on skin and joint symptoms in patients with uveitis may be appropriate.
“The bidirectional relationship between psoriasis and psoriatic arthritis and uveitis suggests a shared pathogenic pathway, and increased systemic inflammation may contribute to the observed relationship,” wrote Dr. Alexander Egeberg of Pfizer and his coauthors. Dr. Egeberg was at the University of Copenhagen, Hellerup, when the study was conducted.
The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is employed by Eli Lilly. No other conflicts of interest were declared.
FROM JAMA DERMATOLOGY
Key clinical point:Patients with psoriasis or uveitis are at significantly greater risk of the other condition.
Major finding: Psoriatic arthritis more than doubles the risk of uveitis, and uveitis is associated with a two-gold greater risk of severe psoriasis.
Data source: A Danish nationwide cohort study of 74,129 individuals with psoriasis.
Disclosures: The study was supported by Pfizer, the LEO Foundation, and the Novo Nordisk Foundation. One author is an employee of Pfizer and another is employed by Eli Lilly. No other conflicts of interest were declared.
PsA disease activity and treatment success defined
New criteria that define psoriatic arthritis disease activity and treatment response establish cutpoints and percentage changes on the Disease Activity Index for Psoriatic Arthritis scores, Dr. Monika M. Schoels of the Second Department of Internal Medicine at Hietzing Hospital, Vienna, and her coauthors reported.
Dr. Schoels and her colleagues collected 30 patient profiles from an observational data set and then surveyed 44 rheumatology experts (getting 33 responses) to assess the patients’ disease activity based on swollen joint count, tender joint count, global activity, C-reactive protein, and pain ratings. Based on the distribution of Disease Activity Index for Psoriatic Arthritis (DAPSA) scores in the patient population, the investigators then calculated the 25th and 75th percentiles to determine the thresholds for different disease states.
Based on the results, the investigators suggested cutoff values of 4 or less for remission, more than 4 and up to 14 for low disease activity, more than 14 and up to 28 for moderate disease activity, and greater than 28 for high disease activity.
They also found that percentage changes of 50%, 75%, and 85% on the DAPSA corresponded to minor, moderate, and major treatment responses based on American College of Rheumatology 20, 50, and 70 responses observed in three randomized trials.
The study findings provide a “definition of treatment targets … as well as the definition of inclusion criteria for clinical trials for patients in” moderate and high disease activity, Dr. Schoels and her colleagues wrote. “The new response levels will help assess treatment in many settings.”
Read the full study in Annals of the Rheumatic Diseases.
New criteria that define psoriatic arthritis disease activity and treatment response establish cutpoints and percentage changes on the Disease Activity Index for Psoriatic Arthritis scores, Dr. Monika M. Schoels of the Second Department of Internal Medicine at Hietzing Hospital, Vienna, and her coauthors reported.
Dr. Schoels and her colleagues collected 30 patient profiles from an observational data set and then surveyed 44 rheumatology experts (getting 33 responses) to assess the patients’ disease activity based on swollen joint count, tender joint count, global activity, C-reactive protein, and pain ratings. Based on the distribution of Disease Activity Index for Psoriatic Arthritis (DAPSA) scores in the patient population, the investigators then calculated the 25th and 75th percentiles to determine the thresholds for different disease states.
Based on the results, the investigators suggested cutoff values of 4 or less for remission, more than 4 and up to 14 for low disease activity, more than 14 and up to 28 for moderate disease activity, and greater than 28 for high disease activity.
They also found that percentage changes of 50%, 75%, and 85% on the DAPSA corresponded to minor, moderate, and major treatment responses based on American College of Rheumatology 20, 50, and 70 responses observed in three randomized trials.
The study findings provide a “definition of treatment targets … as well as the definition of inclusion criteria for clinical trials for patients in” moderate and high disease activity, Dr. Schoels and her colleagues wrote. “The new response levels will help assess treatment in many settings.”
Read the full study in Annals of the Rheumatic Diseases.
New criteria that define psoriatic arthritis disease activity and treatment response establish cutpoints and percentage changes on the Disease Activity Index for Psoriatic Arthritis scores, Dr. Monika M. Schoels of the Second Department of Internal Medicine at Hietzing Hospital, Vienna, and her coauthors reported.
Dr. Schoels and her colleagues collected 30 patient profiles from an observational data set and then surveyed 44 rheumatology experts (getting 33 responses) to assess the patients’ disease activity based on swollen joint count, tender joint count, global activity, C-reactive protein, and pain ratings. Based on the distribution of Disease Activity Index for Psoriatic Arthritis (DAPSA) scores in the patient population, the investigators then calculated the 25th and 75th percentiles to determine the thresholds for different disease states.
Based on the results, the investigators suggested cutoff values of 4 or less for remission, more than 4 and up to 14 for low disease activity, more than 14 and up to 28 for moderate disease activity, and greater than 28 for high disease activity.
They also found that percentage changes of 50%, 75%, and 85% on the DAPSA corresponded to minor, moderate, and major treatment responses based on American College of Rheumatology 20, 50, and 70 responses observed in three randomized trials.
The study findings provide a “definition of treatment targets … as well as the definition of inclusion criteria for clinical trials for patients in” moderate and high disease activity, Dr. Schoels and her colleagues wrote. “The new response levels will help assess treatment in many settings.”
Read the full study in Annals of the Rheumatic Diseases.
Treating Psoriasis in Pregnant Women
What do your patients need to know at the first visit?
Pregnant patients need to know that it is important to carefully monitor them throughout their pregnancy. Although many drugs are not contraindicated, it is still important for the dermatologist to consult with the patient’s obstetrician to discuss risks and benefits of different therapies. Some women see an improvement in the severity of their psoriasis during pregnancy, while others report their condition gets worse. Changes in severity of psoriasis vary by individual and from pregnancy to pregnancy.
What are your go-to treatments? What are the side effects?
UV therapy, particularly narrowband UVB, is a good choice during pregnancy. Excimer laser is another good choice. Topical therapies are standardly employed. Although many of these topical therapies are pregnancy category C, we still employ them regularly in consultation with the patient’s obstetrician. Most biologic therapies are pregnancy category B. We still use these drugs with caution in the setting of pregnancy. If a pregnant patient does wish to continue a biologic therapy, close monitoring and enrollment in a pregnancy registry (http: //www.pregnancystudies.org) would be good options. This registry is analyzing whether medications that are used to treat autoimmune diseases are safe to take during pregnancy. Specifically, the investigators are looking at medications used to treat Crohn disease, rheumatoid arthritis, psoriasis, psoriatic arthritis, and multiple sclerosis. Generally speaking, systemic and biologic drugs should be avoided while pregnant or breastfeeding unless there is a clear medical need. Women of childbearing potential should avoid oral retinoids, methotrexate, and cyclosporine due to a link to birth defects with each of those treatments.
How do you keep patients compliant with treatment?
We see the patients regularly, and I am always available to take telephone calls if any questions arise.
What do you do if patients refuse treatment?
We keep the dialogue ongoing and monitor their condition. They may change their attitude if their condition worsens.
What resources do you recommend to patients for more information?
Patients should consult the National Psoriasis Foundation (http://www.psoriasis.org).
What do your patients need to know at the first visit?
Pregnant patients need to know that it is important to carefully monitor them throughout their pregnancy. Although many drugs are not contraindicated, it is still important for the dermatologist to consult with the patient’s obstetrician to discuss risks and benefits of different therapies. Some women see an improvement in the severity of their psoriasis during pregnancy, while others report their condition gets worse. Changes in severity of psoriasis vary by individual and from pregnancy to pregnancy.
What are your go-to treatments? What are the side effects?
UV therapy, particularly narrowband UVB, is a good choice during pregnancy. Excimer laser is another good choice. Topical therapies are standardly employed. Although many of these topical therapies are pregnancy category C, we still employ them regularly in consultation with the patient’s obstetrician. Most biologic therapies are pregnancy category B. We still use these drugs with caution in the setting of pregnancy. If a pregnant patient does wish to continue a biologic therapy, close monitoring and enrollment in a pregnancy registry (http: //www.pregnancystudies.org) would be good options. This registry is analyzing whether medications that are used to treat autoimmune diseases are safe to take during pregnancy. Specifically, the investigators are looking at medications used to treat Crohn disease, rheumatoid arthritis, psoriasis, psoriatic arthritis, and multiple sclerosis. Generally speaking, systemic and biologic drugs should be avoided while pregnant or breastfeeding unless there is a clear medical need. Women of childbearing potential should avoid oral retinoids, methotrexate, and cyclosporine due to a link to birth defects with each of those treatments.
How do you keep patients compliant with treatment?
We see the patients regularly, and I am always available to take telephone calls if any questions arise.
What do you do if patients refuse treatment?
We keep the dialogue ongoing and monitor their condition. They may change their attitude if their condition worsens.
What resources do you recommend to patients for more information?
Patients should consult the National Psoriasis Foundation (http://www.psoriasis.org).
What do your patients need to know at the first visit?
Pregnant patients need to know that it is important to carefully monitor them throughout their pregnancy. Although many drugs are not contraindicated, it is still important for the dermatologist to consult with the patient’s obstetrician to discuss risks and benefits of different therapies. Some women see an improvement in the severity of their psoriasis during pregnancy, while others report their condition gets worse. Changes in severity of psoriasis vary by individual and from pregnancy to pregnancy.
What are your go-to treatments? What are the side effects?
UV therapy, particularly narrowband UVB, is a good choice during pregnancy. Excimer laser is another good choice. Topical therapies are standardly employed. Although many of these topical therapies are pregnancy category C, we still employ them regularly in consultation with the patient’s obstetrician. Most biologic therapies are pregnancy category B. We still use these drugs with caution in the setting of pregnancy. If a pregnant patient does wish to continue a biologic therapy, close monitoring and enrollment in a pregnancy registry (http: //www.pregnancystudies.org) would be good options. This registry is analyzing whether medications that are used to treat autoimmune diseases are safe to take during pregnancy. Specifically, the investigators are looking at medications used to treat Crohn disease, rheumatoid arthritis, psoriasis, psoriatic arthritis, and multiple sclerosis. Generally speaking, systemic and biologic drugs should be avoided while pregnant or breastfeeding unless there is a clear medical need. Women of childbearing potential should avoid oral retinoids, methotrexate, and cyclosporine due to a link to birth defects with each of those treatments.
How do you keep patients compliant with treatment?
We see the patients regularly, and I am always available to take telephone calls if any questions arise.
What do you do if patients refuse treatment?
We keep the dialogue ongoing and monitor their condition. They may change their attitude if their condition worsens.
What resources do you recommend to patients for more information?
Patients should consult the National Psoriasis Foundation (http://www.psoriasis.org).
Apremilast response durable at 52 weeks
If apremilast (Otezla) works initially for psoriasis, it’s likely to keep on helping for at least a year, according to phase III results published by the drug’s maker, Celgene, in the Journal of the American Academy of Dermatology.
The trial randomized 562 patients with moderate to severe plaque psoriasis to apremilast 30 mg twice daily, and 282 to placebo (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
At week 16, 33% of apremilast patients, but only 5.3% of placebo patients, achieved a 75% or greater reduction from their baseline Psoriasis Area and Severity Index (PASI-75) scores (P less than .0001).
The placebo group was next switched to apremilast so that all the subjects were on the drug from weeks 16 to 32. By week 32, patients switched from placebo caught up with their apremilast peers on PASI-75 response rates and improved pruritus scores.
The study continued past week 32 with 154 patients who had been on apremilast since baseline and had reached PASI-75; half were rerandomized to placebo, half to the drug. At week 52, 47 (61%) of apremilast patients had maintained their PASI-75 response, versus 9 (12%) of placebo patients.
“PASI response was maintained over 52 weeks with continued apremilast treatment. In addition, apremilast demonstrated improvements in nail and scalp psoriasis, both difficult-to-treat forms of psoriasis. Most patients rerandomized to placebo who lost PASI-75 response regained it after apremilast reinitiation,” said the authors, led by Dr. Kim Papp of Probity Medical Research in Waterloo, Ont.
Apremilast, an oral phosphodiesterase 4 inhibitor, was approved by the Food and Drug Administration in 2014 for moderate to severe plaque psoriasis and psoriatic arthritis. The 16-week results are included in the drug’s label.
The label warns of weight loss and depression with apremilast. The new report doesn’t mention depression but does note a mean weight loss of 2.08 kg with the drug, and that 19% of patients lost more than 5% of their body weight. However, no one left the study because of it.
During the first 16 weeks of the trial, the most common side effects were diarrhea (7.1% placebo versus 19% apremilast); nausea (6.7% placebo versus 16% apremilast); upper respiratory tract infection (7.4% versus 10%); nasopharyngitis (8.2% versus 7.3%); and tension headache (4.3% versus 7.3%).
Side effects tended to present early with apremilast, and the incidence didn’t increase as treatment continued. To minimize side effects, the investigators titrated the drug in 10-mg increments over the first week of treatment. Discontinuations due to side effects were low.
Serious adverse events occurred in 2.8% of placebo and 2.1% of apremilast subjects. In the apremilast group, they included three cases of coronary artery disease and three cases of nephrolithiasis, plus two cases each of urinary tract infections, acute myocardial infarctions, and chronic obstructive pulmonary disease.
The investigators excluded patients with major uncontrolled comorbidities, significant infections, active or incompletely treated tuberculosis, biologic use within 12-24 months, use of active topical agents within 2 weeks, and prolonged sun or ultraviolet exposure. Most of the subjects were white, two-thirds were men, and the average age in the study was 45 years. Weak or low-potency topical corticosteroids, coal tar shampoo and salicylic acid for scalp lesions, and unmedicated moisturizers were allowed in the study.
Celgene Corporation, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial relationships with the company.
If apremilast (Otezla) works initially for psoriasis, it’s likely to keep on helping for at least a year, according to phase III results published by the drug’s maker, Celgene, in the Journal of the American Academy of Dermatology.
The trial randomized 562 patients with moderate to severe plaque psoriasis to apremilast 30 mg twice daily, and 282 to placebo (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
At week 16, 33% of apremilast patients, but only 5.3% of placebo patients, achieved a 75% or greater reduction from their baseline Psoriasis Area and Severity Index (PASI-75) scores (P less than .0001).
The placebo group was next switched to apremilast so that all the subjects were on the drug from weeks 16 to 32. By week 32, patients switched from placebo caught up with their apremilast peers on PASI-75 response rates and improved pruritus scores.
The study continued past week 32 with 154 patients who had been on apremilast since baseline and had reached PASI-75; half were rerandomized to placebo, half to the drug. At week 52, 47 (61%) of apremilast patients had maintained their PASI-75 response, versus 9 (12%) of placebo patients.
“PASI response was maintained over 52 weeks with continued apremilast treatment. In addition, apremilast demonstrated improvements in nail and scalp psoriasis, both difficult-to-treat forms of psoriasis. Most patients rerandomized to placebo who lost PASI-75 response regained it after apremilast reinitiation,” said the authors, led by Dr. Kim Papp of Probity Medical Research in Waterloo, Ont.
Apremilast, an oral phosphodiesterase 4 inhibitor, was approved by the Food and Drug Administration in 2014 for moderate to severe plaque psoriasis and psoriatic arthritis. The 16-week results are included in the drug’s label.
The label warns of weight loss and depression with apremilast. The new report doesn’t mention depression but does note a mean weight loss of 2.08 kg with the drug, and that 19% of patients lost more than 5% of their body weight. However, no one left the study because of it.
During the first 16 weeks of the trial, the most common side effects were diarrhea (7.1% placebo versus 19% apremilast); nausea (6.7% placebo versus 16% apremilast); upper respiratory tract infection (7.4% versus 10%); nasopharyngitis (8.2% versus 7.3%); and tension headache (4.3% versus 7.3%).
Side effects tended to present early with apremilast, and the incidence didn’t increase as treatment continued. To minimize side effects, the investigators titrated the drug in 10-mg increments over the first week of treatment. Discontinuations due to side effects were low.
Serious adverse events occurred in 2.8% of placebo and 2.1% of apremilast subjects. In the apremilast group, they included three cases of coronary artery disease and three cases of nephrolithiasis, plus two cases each of urinary tract infections, acute myocardial infarctions, and chronic obstructive pulmonary disease.
The investigators excluded patients with major uncontrolled comorbidities, significant infections, active or incompletely treated tuberculosis, biologic use within 12-24 months, use of active topical agents within 2 weeks, and prolonged sun or ultraviolet exposure. Most of the subjects were white, two-thirds were men, and the average age in the study was 45 years. Weak or low-potency topical corticosteroids, coal tar shampoo and salicylic acid for scalp lesions, and unmedicated moisturizers were allowed in the study.
Celgene Corporation, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial relationships with the company.
If apremilast (Otezla) works initially for psoriasis, it’s likely to keep on helping for at least a year, according to phase III results published by the drug’s maker, Celgene, in the Journal of the American Academy of Dermatology.
The trial randomized 562 patients with moderate to severe plaque psoriasis to apremilast 30 mg twice daily, and 282 to placebo (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
At week 16, 33% of apremilast patients, but only 5.3% of placebo patients, achieved a 75% or greater reduction from their baseline Psoriasis Area and Severity Index (PASI-75) scores (P less than .0001).
The placebo group was next switched to apremilast so that all the subjects were on the drug from weeks 16 to 32. By week 32, patients switched from placebo caught up with their apremilast peers on PASI-75 response rates and improved pruritus scores.
The study continued past week 32 with 154 patients who had been on apremilast since baseline and had reached PASI-75; half were rerandomized to placebo, half to the drug. At week 52, 47 (61%) of apremilast patients had maintained their PASI-75 response, versus 9 (12%) of placebo patients.
“PASI response was maintained over 52 weeks with continued apremilast treatment. In addition, apremilast demonstrated improvements in nail and scalp psoriasis, both difficult-to-treat forms of psoriasis. Most patients rerandomized to placebo who lost PASI-75 response regained it after apremilast reinitiation,” said the authors, led by Dr. Kim Papp of Probity Medical Research in Waterloo, Ont.
Apremilast, an oral phosphodiesterase 4 inhibitor, was approved by the Food and Drug Administration in 2014 for moderate to severe plaque psoriasis and psoriatic arthritis. The 16-week results are included in the drug’s label.
The label warns of weight loss and depression with apremilast. The new report doesn’t mention depression but does note a mean weight loss of 2.08 kg with the drug, and that 19% of patients lost more than 5% of their body weight. However, no one left the study because of it.
During the first 16 weeks of the trial, the most common side effects were diarrhea (7.1% placebo versus 19% apremilast); nausea (6.7% placebo versus 16% apremilast); upper respiratory tract infection (7.4% versus 10%); nasopharyngitis (8.2% versus 7.3%); and tension headache (4.3% versus 7.3%).
Side effects tended to present early with apremilast, and the incidence didn’t increase as treatment continued. To minimize side effects, the investigators titrated the drug in 10-mg increments over the first week of treatment. Discontinuations due to side effects were low.
Serious adverse events occurred in 2.8% of placebo and 2.1% of apremilast subjects. In the apremilast group, they included three cases of coronary artery disease and three cases of nephrolithiasis, plus two cases each of urinary tract infections, acute myocardial infarctions, and chronic obstructive pulmonary disease.
The investigators excluded patients with major uncontrolled comorbidities, significant infections, active or incompletely treated tuberculosis, biologic use within 12-24 months, use of active topical agents within 2 weeks, and prolonged sun or ultraviolet exposure. Most of the subjects were white, two-thirds were men, and the average age in the study was 45 years. Weak or low-potency topical corticosteroids, coal tar shampoo and salicylic acid for scalp lesions, and unmedicated moisturizers were allowed in the study.
Celgene Corporation, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial relationships with the company.
FROM JAAD
Key clinical point: Apremilast doesn’t seem to lose effect over time.
Major finding: At week 52, 61% of apremilast patients had maintained their PASI-75 response, versus 12% of patients switched to placebo at week 32.
Data source: ESTEEM 1, a phase III trial of patients with moderate to severe plaque psoriasis.
Disclosures: Celgene, the maker of apremilast, funded the work. Three investigators are employees, and most of the rest reported financial ties with the company.
In psoriasis, low cholesterol efflux capacity may predict heart risk
Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, based on findings from a prospective cohort study.
High-density lipoprotein cholesterol efflux capacity (CEC) was inversely correlated with the noncalcified burden (NCB) of coronary atherosclerosis in baseline data from the first 101 patients enrolled in the 4-year study, Dr. Taufiq Salahuddin of the National Heart, Lung, and Blood Institute and colleagues reported online in the European Heart Journal.
The relationship between CEC and NCB “suggests that higher CEC may promote reverse cholesterol transport from earlier, more lipid-rich plaques. … Others have found this noncalcified plaque to be the culprit lesion in acute coronary syndromes. Therefore, in the context of our study, there is strong biological plausibility for observing a relationship between CEC and NCB; psoriasis is associated with both increased future cardiovascular events and impaired HDL function, and our findings suggest that this may be due to predisposition toward formation of noncalcified plaque,” the researchers wrote.
The relationship between CEC (quantified using a cell-based ex vivo assay), and NCB plaque indexes (assessed by quantitative coronary computed tomography angiography) persisted after adjustment for cardiovascular risk factors, high-density lipoprotein cholesterol levels, and apolipoprotein A1 levels.
Of note, the relationship between CEC and NCB was stronger in women than in men, with a statistically significant gender interaction, the investigators found (Eur Heart J. 2015 Jul 18. doi.org/10.1093/eurheartj/ehv339).
Cholesterol efflux capacity has been shown in prior studies to predict future cardiovascular events, and since psoriasis both increases cardiovascular risk and impairs CEC, the investigators sought to assess the cross-sectional relationship between coronary plaque burden and CEC.
Study subjects were adults over age 18 years with moderate skin disease severity (median psoriasis area severity index [PASI] score, 6.2), and a low 10-year pooled cohort equation risk score (median, 2.7%). Traditional lipid profiles were within normal limits, and the median CEC was 0.94.
In addition to the inverse correlation between CEC and NCB at baseline, significant relationships were seen between CEC and PASI score, body surface area affected by psoriasis, and HDL cholesterol level, and between noncalcified burden and CEC, psoriasis severity, body surface area affected by psoriasis, and HDL cholesterol.
“Stratified by the sample’s median CEC value, patients with low CEC had greater total burden of coronary plaque and greater NCB than did patients with high CEC (0.0131 and 0.0127 mm2 in low CEC vs. 0.0106 and 0.0103 mm2 with high CEC),” they wrote.
No differences were seen based on psoriasis treatment status or statin use, although systemic/biologic therapy was associated with a reduction in noncalcified burden.
The findings with respect to gender differences may suggest that greater CEC is more protective against noncalcified plaque in women than it is in men, the investigators added, concluding that “ongoing follow-up of these patients will inform whether aggressive treatment of psoriasis and lifestyle measures improve CEC and ultimately noncalcified burden within the coronary arteries in psoriasis.”
This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, based on findings from a prospective cohort study.
High-density lipoprotein cholesterol efflux capacity (CEC) was inversely correlated with the noncalcified burden (NCB) of coronary atherosclerosis in baseline data from the first 101 patients enrolled in the 4-year study, Dr. Taufiq Salahuddin of the National Heart, Lung, and Blood Institute and colleagues reported online in the European Heart Journal.
The relationship between CEC and NCB “suggests that higher CEC may promote reverse cholesterol transport from earlier, more lipid-rich plaques. … Others have found this noncalcified plaque to be the culprit lesion in acute coronary syndromes. Therefore, in the context of our study, there is strong biological plausibility for observing a relationship between CEC and NCB; psoriasis is associated with both increased future cardiovascular events and impaired HDL function, and our findings suggest that this may be due to predisposition toward formation of noncalcified plaque,” the researchers wrote.
The relationship between CEC (quantified using a cell-based ex vivo assay), and NCB plaque indexes (assessed by quantitative coronary computed tomography angiography) persisted after adjustment for cardiovascular risk factors, high-density lipoprotein cholesterol levels, and apolipoprotein A1 levels.
Of note, the relationship between CEC and NCB was stronger in women than in men, with a statistically significant gender interaction, the investigators found (Eur Heart J. 2015 Jul 18. doi.org/10.1093/eurheartj/ehv339).
Cholesterol efflux capacity has been shown in prior studies to predict future cardiovascular events, and since psoriasis both increases cardiovascular risk and impairs CEC, the investigators sought to assess the cross-sectional relationship between coronary plaque burden and CEC.
Study subjects were adults over age 18 years with moderate skin disease severity (median psoriasis area severity index [PASI] score, 6.2), and a low 10-year pooled cohort equation risk score (median, 2.7%). Traditional lipid profiles were within normal limits, and the median CEC was 0.94.
In addition to the inverse correlation between CEC and NCB at baseline, significant relationships were seen between CEC and PASI score, body surface area affected by psoriasis, and HDL cholesterol level, and between noncalcified burden and CEC, psoriasis severity, body surface area affected by psoriasis, and HDL cholesterol.
“Stratified by the sample’s median CEC value, patients with low CEC had greater total burden of coronary plaque and greater NCB than did patients with high CEC (0.0131 and 0.0127 mm2 in low CEC vs. 0.0106 and 0.0103 mm2 with high CEC),” they wrote.
No differences were seen based on psoriasis treatment status or statin use, although systemic/biologic therapy was associated with a reduction in noncalcified burden.
The findings with respect to gender differences may suggest that greater CEC is more protective against noncalcified plaque in women than it is in men, the investigators added, concluding that “ongoing follow-up of these patients will inform whether aggressive treatment of psoriasis and lifestyle measures improve CEC and ultimately noncalcified burden within the coronary arteries in psoriasis.”
This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, based on findings from a prospective cohort study.
High-density lipoprotein cholesterol efflux capacity (CEC) was inversely correlated with the noncalcified burden (NCB) of coronary atherosclerosis in baseline data from the first 101 patients enrolled in the 4-year study, Dr. Taufiq Salahuddin of the National Heart, Lung, and Blood Institute and colleagues reported online in the European Heart Journal.
The relationship between CEC and NCB “suggests that higher CEC may promote reverse cholesterol transport from earlier, more lipid-rich plaques. … Others have found this noncalcified plaque to be the culprit lesion in acute coronary syndromes. Therefore, in the context of our study, there is strong biological plausibility for observing a relationship between CEC and NCB; psoriasis is associated with both increased future cardiovascular events and impaired HDL function, and our findings suggest that this may be due to predisposition toward formation of noncalcified plaque,” the researchers wrote.
The relationship between CEC (quantified using a cell-based ex vivo assay), and NCB plaque indexes (assessed by quantitative coronary computed tomography angiography) persisted after adjustment for cardiovascular risk factors, high-density lipoprotein cholesterol levels, and apolipoprotein A1 levels.
Of note, the relationship between CEC and NCB was stronger in women than in men, with a statistically significant gender interaction, the investigators found (Eur Heart J. 2015 Jul 18. doi.org/10.1093/eurheartj/ehv339).
Cholesterol efflux capacity has been shown in prior studies to predict future cardiovascular events, and since psoriasis both increases cardiovascular risk and impairs CEC, the investigators sought to assess the cross-sectional relationship between coronary plaque burden and CEC.
Study subjects were adults over age 18 years with moderate skin disease severity (median psoriasis area severity index [PASI] score, 6.2), and a low 10-year pooled cohort equation risk score (median, 2.7%). Traditional lipid profiles were within normal limits, and the median CEC was 0.94.
In addition to the inverse correlation between CEC and NCB at baseline, significant relationships were seen between CEC and PASI score, body surface area affected by psoriasis, and HDL cholesterol level, and between noncalcified burden and CEC, psoriasis severity, body surface area affected by psoriasis, and HDL cholesterol.
“Stratified by the sample’s median CEC value, patients with low CEC had greater total burden of coronary plaque and greater NCB than did patients with high CEC (0.0131 and 0.0127 mm2 in low CEC vs. 0.0106 and 0.0103 mm2 with high CEC),” they wrote.
No differences were seen based on psoriasis treatment status or statin use, although systemic/biologic therapy was associated with a reduction in noncalcified burden.
The findings with respect to gender differences may suggest that greater CEC is more protective against noncalcified plaque in women than it is in men, the investigators added, concluding that “ongoing follow-up of these patients will inform whether aggressive treatment of psoriasis and lifestyle measures improve CEC and ultimately noncalcified burden within the coronary arteries in psoriasis.”
This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
FROM THE EUROPEAN HEART JOURNAL
Key clinical point: Low cholesterol efflux capacity may be an important biomarker for subclinical coronary atherosclerosis in patients with psoriasis, according to findings from a prospective cohort study.
Major finding: Baseline data showed an inverse correlation between HDL cholesterol efflux capacity and noncalcified burden of coronary atherosclerosis (unadjusted beta-coefficient, –0.33).
Data source: The first 101 patients in a 4-year prospective cohort study.
Disclosures: This study was supported by a National Institutes of Health intramural grant, and the NIH Medical Research Scholars Program. The investigators reported having no relevant financial disclosures.
Manage Your Dermatology Practice: Answering Patient Questions About Diet
Patients often inquire if their diet has caused a dermatologic condition or if their diet makes it worse. Dr. Gary Goldenberg addresses how diet may impact acne, psoriasis, and urticaria. Ultimately, patient education by the dermatologist is needed to ensure patients are not relying on misinformation on the Internet regarding diets they should consider for their particular condition.
Patients often inquire if their diet has caused a dermatologic condition or if their diet makes it worse. Dr. Gary Goldenberg addresses how diet may impact acne, psoriasis, and urticaria. Ultimately, patient education by the dermatologist is needed to ensure patients are not relying on misinformation on the Internet regarding diets they should consider for their particular condition.
Patients often inquire if their diet has caused a dermatologic condition or if their diet makes it worse. Dr. Gary Goldenberg addresses how diet may impact acne, psoriasis, and urticaria. Ultimately, patient education by the dermatologist is needed to ensure patients are not relying on misinformation on the Internet regarding diets they should consider for their particular condition.
GRAPPA initiatives continue to chart the way for psoriasis, psoriatic arthritis
The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting was held in Stockholm this year on July 7 and 8. This year’s meeting was packed with updates regarding numerous projects, including, but not limited to, educational initiatives, research work streams, inclusion of patients in the research process in psoriasis and psoriatic arthritis (PsA), and updating of the core set of outcome measures to be included in both psoriasis and PsA clinical trials through Outcome Measures in Rheumatology (OMERACT) and International Dermatology Outcome Measures (IDEOM) initiatives.
Among the most exciting initiatives was the review of the 2015 GRAPPA PsA Treatment Recommendations. The new treatment recommendations began 2 years ago and have included extensive literature reviews, small group discussions, and large group surveys. The final manuscript includes overarching principles and specific recommendations broken down by disease manifestations (for example, peripheral arthritis, axial disease, enthesitis, dactylitis, skin psoriasis, and nail disease). Changes from the 2009 GRAPPA Treatment Recommendations (Ann. Rheum. Dis. 2009;68:1387-94) include addition of new therapies (for example, phosphodiesterase-4 inhibitors, interleukin (IL) 12/23 inhibitors, IL-17 inhibitors) and recommendations for screening for comorbidities, as well as a table noting the influence of comorbidities on therapy selection. Each recommendation is supported as “weak” or “strong” depending on the level of evidence. Patient research partners were involved in all steps of the process. The final manuscript has been submitted for publication.
In addition to treating the disease, better defining the disease is also of critical importance. In particular, defining a “flare” of rheumatoid arthritis or PsA has been quite difficult, and there has been great interest in building an assessment tool to measure the occurrence of flare. During the meeting, Dr. Anna Moverley and Dr. Philip Helliwell, both from the NIHR Musculoskeletal Biomedical Research Unit, Leeds (England) Institute of Rheumatic & Musculoskeletal Medicine, presented their work to date on defining PsA flares. They began their work by conducting a qualitative analysis of 18 patient interviews to identify themes related to having a PsA flare. Domains (or themes) identified included physical, psychological, social withdrawal, fatigue, and loss of function. In addition, patients described flare triggers, timing of flares, preflare management, and flare management. The results of this study were recently published in a paper titled, “It’s not just the joints, it’s the whole thing: qualitative analysis of patients’ experience of flare in psoriatic arthritis” (Rheumatology 2015;54:1448-54). The team then used these themes to create a Delphi survey, which was sent to both patients and physicians. Working groups at GRAPPA were used to determine the next steps, in particular whether a physician or patient tool (or both) should be developed.
Care of the patient with PsA from a multispecialty perspective was discussed in the “combined clinics” presentation. Several academic centers across North America have developed combined dermatology-rheumatology clinics. The functioning of these clinics varies widely from monthly dual clinics in which the dermatologist and rheumatologist see the patient together for virtual combined clinics in which time is set aside for discussion of shared patients. The advantages of these dual clinics include rich educational opportunities for medical students, residents, and fellows; the ideal clinical care model for patients; enhanced patient satisfaction with their care; improved patient care and quicker transition to a disease-modifying agent when indicated; and physician satisfaction and learning opportunities through collaborative work. Institutions using these collaborative models have combined to form the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN). The group has had two meetings and has developed research initiatives that are moving forward. Another goal of the group is to assist others centers interested in creating similar collaborative clinics.
The GRAPPA PsA BioDam (Psoriatic Arthritis Biomarkers for Joint Damage) initiative was also discussed at the meeting. The goal of this initiative is to examine soluble biomarkers as predictors of structural damage in PsA. Blood samples are taken at enrollment and then patients are followed prospectively. At 24 months of follow-up, repeat radiographs are obtained to monitor the development of new erosions. In addition to plain film radiographs, three sites are now also including MRI to assess for erosions. The study team, led by Dr. Oliver FitzGerald, hopes to begin analyzing some of the results from the study in the next 1-2 years.
Dr. Ogdie-Beatty is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting was held in Stockholm this year on July 7 and 8. This year’s meeting was packed with updates regarding numerous projects, including, but not limited to, educational initiatives, research work streams, inclusion of patients in the research process in psoriasis and psoriatic arthritis (PsA), and updating of the core set of outcome measures to be included in both psoriasis and PsA clinical trials through Outcome Measures in Rheumatology (OMERACT) and International Dermatology Outcome Measures (IDEOM) initiatives.
Among the most exciting initiatives was the review of the 2015 GRAPPA PsA Treatment Recommendations. The new treatment recommendations began 2 years ago and have included extensive literature reviews, small group discussions, and large group surveys. The final manuscript includes overarching principles and specific recommendations broken down by disease manifestations (for example, peripheral arthritis, axial disease, enthesitis, dactylitis, skin psoriasis, and nail disease). Changes from the 2009 GRAPPA Treatment Recommendations (Ann. Rheum. Dis. 2009;68:1387-94) include addition of new therapies (for example, phosphodiesterase-4 inhibitors, interleukin (IL) 12/23 inhibitors, IL-17 inhibitors) and recommendations for screening for comorbidities, as well as a table noting the influence of comorbidities on therapy selection. Each recommendation is supported as “weak” or “strong” depending on the level of evidence. Patient research partners were involved in all steps of the process. The final manuscript has been submitted for publication.
In addition to treating the disease, better defining the disease is also of critical importance. In particular, defining a “flare” of rheumatoid arthritis or PsA has been quite difficult, and there has been great interest in building an assessment tool to measure the occurrence of flare. During the meeting, Dr. Anna Moverley and Dr. Philip Helliwell, both from the NIHR Musculoskeletal Biomedical Research Unit, Leeds (England) Institute of Rheumatic & Musculoskeletal Medicine, presented their work to date on defining PsA flares. They began their work by conducting a qualitative analysis of 18 patient interviews to identify themes related to having a PsA flare. Domains (or themes) identified included physical, psychological, social withdrawal, fatigue, and loss of function. In addition, patients described flare triggers, timing of flares, preflare management, and flare management. The results of this study were recently published in a paper titled, “It’s not just the joints, it’s the whole thing: qualitative analysis of patients’ experience of flare in psoriatic arthritis” (Rheumatology 2015;54:1448-54). The team then used these themes to create a Delphi survey, which was sent to both patients and physicians. Working groups at GRAPPA were used to determine the next steps, in particular whether a physician or patient tool (or both) should be developed.
Care of the patient with PsA from a multispecialty perspective was discussed in the “combined clinics” presentation. Several academic centers across North America have developed combined dermatology-rheumatology clinics. The functioning of these clinics varies widely from monthly dual clinics in which the dermatologist and rheumatologist see the patient together for virtual combined clinics in which time is set aside for discussion of shared patients. The advantages of these dual clinics include rich educational opportunities for medical students, residents, and fellows; the ideal clinical care model for patients; enhanced patient satisfaction with their care; improved patient care and quicker transition to a disease-modifying agent when indicated; and physician satisfaction and learning opportunities through collaborative work. Institutions using these collaborative models have combined to form the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN). The group has had two meetings and has developed research initiatives that are moving forward. Another goal of the group is to assist others centers interested in creating similar collaborative clinics.
The GRAPPA PsA BioDam (Psoriatic Arthritis Biomarkers for Joint Damage) initiative was also discussed at the meeting. The goal of this initiative is to examine soluble biomarkers as predictors of structural damage in PsA. Blood samples are taken at enrollment and then patients are followed prospectively. At 24 months of follow-up, repeat radiographs are obtained to monitor the development of new erosions. In addition to plain film radiographs, three sites are now also including MRI to assess for erosions. The study team, led by Dr. Oliver FitzGerald, hopes to begin analyzing some of the results from the study in the next 1-2 years.
Dr. Ogdie-Beatty is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
The Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) meeting was held in Stockholm this year on July 7 and 8. This year’s meeting was packed with updates regarding numerous projects, including, but not limited to, educational initiatives, research work streams, inclusion of patients in the research process in psoriasis and psoriatic arthritis (PsA), and updating of the core set of outcome measures to be included in both psoriasis and PsA clinical trials through Outcome Measures in Rheumatology (OMERACT) and International Dermatology Outcome Measures (IDEOM) initiatives.
Among the most exciting initiatives was the review of the 2015 GRAPPA PsA Treatment Recommendations. The new treatment recommendations began 2 years ago and have included extensive literature reviews, small group discussions, and large group surveys. The final manuscript includes overarching principles and specific recommendations broken down by disease manifestations (for example, peripheral arthritis, axial disease, enthesitis, dactylitis, skin psoriasis, and nail disease). Changes from the 2009 GRAPPA Treatment Recommendations (Ann. Rheum. Dis. 2009;68:1387-94) include addition of new therapies (for example, phosphodiesterase-4 inhibitors, interleukin (IL) 12/23 inhibitors, IL-17 inhibitors) and recommendations for screening for comorbidities, as well as a table noting the influence of comorbidities on therapy selection. Each recommendation is supported as “weak” or “strong” depending on the level of evidence. Patient research partners were involved in all steps of the process. The final manuscript has been submitted for publication.
In addition to treating the disease, better defining the disease is also of critical importance. In particular, defining a “flare” of rheumatoid arthritis or PsA has been quite difficult, and there has been great interest in building an assessment tool to measure the occurrence of flare. During the meeting, Dr. Anna Moverley and Dr. Philip Helliwell, both from the NIHR Musculoskeletal Biomedical Research Unit, Leeds (England) Institute of Rheumatic & Musculoskeletal Medicine, presented their work to date on defining PsA flares. They began their work by conducting a qualitative analysis of 18 patient interviews to identify themes related to having a PsA flare. Domains (or themes) identified included physical, psychological, social withdrawal, fatigue, and loss of function. In addition, patients described flare triggers, timing of flares, preflare management, and flare management. The results of this study were recently published in a paper titled, “It’s not just the joints, it’s the whole thing: qualitative analysis of patients’ experience of flare in psoriatic arthritis” (Rheumatology 2015;54:1448-54). The team then used these themes to create a Delphi survey, which was sent to both patients and physicians. Working groups at GRAPPA were used to determine the next steps, in particular whether a physician or patient tool (or both) should be developed.
Care of the patient with PsA from a multispecialty perspective was discussed in the “combined clinics” presentation. Several academic centers across North America have developed combined dermatology-rheumatology clinics. The functioning of these clinics varies widely from monthly dual clinics in which the dermatologist and rheumatologist see the patient together for virtual combined clinics in which time is set aside for discussion of shared patients. The advantages of these dual clinics include rich educational opportunities for medical students, residents, and fellows; the ideal clinical care model for patients; enhanced patient satisfaction with their care; improved patient care and quicker transition to a disease-modifying agent when indicated; and physician satisfaction and learning opportunities through collaborative work. Institutions using these collaborative models have combined to form the Psoriasis and Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN). The group has had two meetings and has developed research initiatives that are moving forward. Another goal of the group is to assist others centers interested in creating similar collaborative clinics.
The GRAPPA PsA BioDam (Psoriatic Arthritis Biomarkers for Joint Damage) initiative was also discussed at the meeting. The goal of this initiative is to examine soluble biomarkers as predictors of structural damage in PsA. Blood samples are taken at enrollment and then patients are followed prospectively. At 24 months of follow-up, repeat radiographs are obtained to monitor the development of new erosions. In addition to plain film radiographs, three sites are now also including MRI to assess for erosions. The study team, led by Dr. Oliver FitzGerald, hopes to begin analyzing some of the results from the study in the next 1-2 years.
Dr. Ogdie-Beatty is director of the Penn Psoriatic Arthritis Clinic at the University of Pennsylvania, Philadelphia, and is a member of the GRAPPA Steering Committee.
A call to action on metabolic syndrome and pediatric psoriasis
VANCOUVER – Dermatologists and primary care physicians working collaboratively have a golden opportunity to improve the long-term health of pediatric psoriasis patients by addressing their predisposition to components of the metabolic syndrome, Dr. Amy S. Paller declared at the World Congress of Dermatology.
“I think we as dermatologists should be in touch with the primary care doctors of every one of our children with psoriasis. Together, we should be thinking about whether the child has metabolic issues and working jointly to most effectively counsel and evaluate these children for their potential risk for these metabolic disorders,” said Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Pediatric psoriasis is commonly associated with other comorbid conditions in addition to metabolic disorders. But the metabolic syndrome has recently become the focus of increasing attention given that cardiovascular disease is the No. 1 cause of death in the United States, and it appears that children with psoriasis may be getting a jump start on the atherosclerotic process.
By now, it’s well established that plaque psoriasis in adults is strongly associated with increased risks of diabetes, obesity, dyslipidemia, the metabolic syndrome, and cardiovascular disease. Mounting evidence indicates children and adolescents with psoriasis face the same risks.
Everyone knows how difficult it can be to make the long-term lifestyle changes that reverse obesity and its related metabolic disorders. But dermatologists, pediatricians, and family physicians have some leverage when it comes to pediatric psoriasis.
“Think about the fact that 30% of children with psoriasis have a first-degree relative with psoriasis, usually a parent. I think we need to think about counseling young adults with psoriasis early on, especially if that adult is overweight or obese, about the need for adopting a healthy lifestyle. If they do that, it’s not just for themselves but for their children, and we just might prevent pediatric psoriasis in that family or temper its severity through that healthy lifestyle intervention,” Dr. Paller continued.
The hope is that effectively addressing the metabolic comorbidities of pediatric psoriasis will modulate and improve the skin disease; in other words, that weight loss could improve psoriasis. As yet, however, that’s just a hope, as there is no persuasive supporting evidence.
“We’re looking towards ongoing adult trials to give us some clues about whether that’s the case,” she said.
Evidence for comorbidities
Some of the key evidence regarding the metabolic comorbidities of pediatric psoriasis comes from a landmark German epidemiologic study involving 33,981 pediatric psoriasis patients. The prevalence of psoriasis in German youth rose linearly from 0.12% at age 1 year to 1.2% at age 18. Pediatric psoriasis patients had significantly higher rates of diabetes, hyperlipidemia, obesity, and hypertension than did nonpsoriatic controls (Br. J. Dermatol. 2010;162:633-6).
A Kaiser Permanente study of nearly 711,000 youths aged 2-19 years showed that those who were overweight were 2.8-fold more likely than normal-weight youth to have severe or widespread psoriasis, while those who were moderately obese were at 2.9-fold increased risk and extremely obese youth were at 4.2-fold increased risk. Among adolescents, having psoriasis was associated with significantly higher mean total and LDL cholesterol, triglycerides, and alanine aminotransferase levels (J. Pediatr. 2011;159:577-83).
Recent evidence suggests that even before increased levels of LDL cholesterol and triglycerides are apparent in children with psoriasis, abnormalities in lipid function are present and may potentially serve as a novel marker for early cardiovascular risk. Dr. Paller cited a study presented by Dr. Wynnis L. Tom of Rady Children’s Hospital, San Diego, at the 2015 annual meeting of the Society for Investigative Dermatology. The case-control study included 50 children with psoriasis and 50 matched controls with a mean age of 13 years.
Like other investigators, Dr. Tom found that the psoriatic children had higher waist/hip ratios and more insulin resistance. While fasting lipid levels didn’t differ between the two groups, the psoriasis patients had significantly higher levels of atherogenic apolipoprotein B, fewer of the particularly cardioprotective large-size HDL particles, and reduced HDL efflux capacity. Stay tuned regarding these potential early markers, Dr. Paller advised.
She was lead author of a 409-patient international study that showed the risks of obesity and a high waist circumference rise with greater severity of pediatric psoriasis. Children with severe psoriasis were at 4.92-fold increased risk of obesity, compared with controls, while even those with mild psoriasis were at 3.6-fold increased risk (JAMA Dermatol. 2013;149:166-76).
Which comes first?
The question arises: Which comes first in children, the excess adiposity or the psoriasis? Dr. Paller said that although the final word isn’t in, she and her coworkers found in a pilot study of 27 overweight or obese children with psoriasis that excess adiposity typically came first. Moreover, among the roughly one-half of children with a family history of obesity, onset of psoriasis occurred a full 3 years earlier than in those without a positive family history (JAMA Dermatol. 2014;150:573-4).
In another small study, this by investigators at Tufts University, Boston, 6 of 20 children with psoriasis (30%) met criteria for the metabolic syndrome, compared with just 1 of 20 matched nonpsoriatic controls (Pediatr. Dermatol. 2013;30:700-5).
Dr. Paller said that if dermatologists and primary care physicians are to successfully collaborate in tackling the comorbid metabolic disorders associated with pediatric psoriasis, a prerequisite is that dermatologists are going to have to do a better job of educating their primary care colleagues about the skin disease as manifest in children.
“I think it’s very important that pediatricians are aware that psoriasis is a risk factor for metabolic syndrome. But pediatric psoriasis is often misdiagnosed by primary care physicians who mistake it for eczema or tinea infection or contact dermatitis,” according to the pediatric dermatologist.
In one eye-catching Australian study, she noted, a mere 9% of patients with pediatric psoriasis were correctly diagnosed before referral to a dermatologist (Australas. J. Dermatol. 2012;53:98-105).
Pediatric psoriasis: not just skin deep
In addition to the increased risk of metabolic disorders faced by pediatric psoriasis patients, other common comorbidities include depression, anxiety disorders, impaired self-esteem and quality of life, arthritis, and Crohn’s disease, Dr. Paller observed.
• Quality of life. “The quality of life impact of psoriasis is profound. It’s a highly visible disorder, which affects the development of self-esteem and social relationships,” Dr. Paller said.
Investigators at Texas A&M University applied the Pediatric Quality of Life Inventory Version 4.0 to 208 patients aged 2-17 years with moderate to severe psoriasis and compared the results to published data on children with arthritis, asthma, diabetes, and psychiatric disorders. Health-related quality of life turned out to be more impaired in the psoriasis patients than in those with diabetes. The quality-of-life impairment associated with pediatric psoriasis was comparable to that of having asthma or arthritis, albeit not as severe as for pediatric psychiatric disorders (Eur. J. Pediatr. 2012;171:485-92).
• Psychiatric disorders. A study of more than 7,400 pediatric psoriasis patients concluded they had an adjusted 25% increased risk of developing depression, compared with psoriasis-free controls, as well as a 32% increased risk of anxiety disorders and a 55% greater risk of bipolar disorder (J. Am. Acad. Dermatol. 2012;67:651-7.e2).
• Psoriatic arthritis. An estimated 1 in 10 U.S. children with psoriasis report having arthritis, often classified as juvenile idiopathic arthritis (JAMA Dermatol. 2013;149:1180-5).
• Crohn’s disease. A large German epidemiologic study concluded that psoriasis was associated with a 3.69-fold increased risk of Crohn’s disease. There was no increased risk of ulcerative colitis (Br. J. Dermatol. 2010;162:633-6).
Dr. Paller reported receiving research grants from Amgen and Leo and serving as a consultant to AbbVie.
VANCOUVER – Dermatologists and primary care physicians working collaboratively have a golden opportunity to improve the long-term health of pediatric psoriasis patients by addressing their predisposition to components of the metabolic syndrome, Dr. Amy S. Paller declared at the World Congress of Dermatology.
“I think we as dermatologists should be in touch with the primary care doctors of every one of our children with psoriasis. Together, we should be thinking about whether the child has metabolic issues and working jointly to most effectively counsel and evaluate these children for their potential risk for these metabolic disorders,” said Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Pediatric psoriasis is commonly associated with other comorbid conditions in addition to metabolic disorders. But the metabolic syndrome has recently become the focus of increasing attention given that cardiovascular disease is the No. 1 cause of death in the United States, and it appears that children with psoriasis may be getting a jump start on the atherosclerotic process.
By now, it’s well established that plaque psoriasis in adults is strongly associated with increased risks of diabetes, obesity, dyslipidemia, the metabolic syndrome, and cardiovascular disease. Mounting evidence indicates children and adolescents with psoriasis face the same risks.
Everyone knows how difficult it can be to make the long-term lifestyle changes that reverse obesity and its related metabolic disorders. But dermatologists, pediatricians, and family physicians have some leverage when it comes to pediatric psoriasis.
“Think about the fact that 30% of children with psoriasis have a first-degree relative with psoriasis, usually a parent. I think we need to think about counseling young adults with psoriasis early on, especially if that adult is overweight or obese, about the need for adopting a healthy lifestyle. If they do that, it’s not just for themselves but for their children, and we just might prevent pediatric psoriasis in that family or temper its severity through that healthy lifestyle intervention,” Dr. Paller continued.
The hope is that effectively addressing the metabolic comorbidities of pediatric psoriasis will modulate and improve the skin disease; in other words, that weight loss could improve psoriasis. As yet, however, that’s just a hope, as there is no persuasive supporting evidence.
“We’re looking towards ongoing adult trials to give us some clues about whether that’s the case,” she said.
Evidence for comorbidities
Some of the key evidence regarding the metabolic comorbidities of pediatric psoriasis comes from a landmark German epidemiologic study involving 33,981 pediatric psoriasis patients. The prevalence of psoriasis in German youth rose linearly from 0.12% at age 1 year to 1.2% at age 18. Pediatric psoriasis patients had significantly higher rates of diabetes, hyperlipidemia, obesity, and hypertension than did nonpsoriatic controls (Br. J. Dermatol. 2010;162:633-6).
A Kaiser Permanente study of nearly 711,000 youths aged 2-19 years showed that those who were overweight were 2.8-fold more likely than normal-weight youth to have severe or widespread psoriasis, while those who were moderately obese were at 2.9-fold increased risk and extremely obese youth were at 4.2-fold increased risk. Among adolescents, having psoriasis was associated with significantly higher mean total and LDL cholesterol, triglycerides, and alanine aminotransferase levels (J. Pediatr. 2011;159:577-83).
Recent evidence suggests that even before increased levels of LDL cholesterol and triglycerides are apparent in children with psoriasis, abnormalities in lipid function are present and may potentially serve as a novel marker for early cardiovascular risk. Dr. Paller cited a study presented by Dr. Wynnis L. Tom of Rady Children’s Hospital, San Diego, at the 2015 annual meeting of the Society for Investigative Dermatology. The case-control study included 50 children with psoriasis and 50 matched controls with a mean age of 13 years.
Like other investigators, Dr. Tom found that the psoriatic children had higher waist/hip ratios and more insulin resistance. While fasting lipid levels didn’t differ between the two groups, the psoriasis patients had significantly higher levels of atherogenic apolipoprotein B, fewer of the particularly cardioprotective large-size HDL particles, and reduced HDL efflux capacity. Stay tuned regarding these potential early markers, Dr. Paller advised.
She was lead author of a 409-patient international study that showed the risks of obesity and a high waist circumference rise with greater severity of pediatric psoriasis. Children with severe psoriasis were at 4.92-fold increased risk of obesity, compared with controls, while even those with mild psoriasis were at 3.6-fold increased risk (JAMA Dermatol. 2013;149:166-76).
Which comes first?
The question arises: Which comes first in children, the excess adiposity or the psoriasis? Dr. Paller said that although the final word isn’t in, she and her coworkers found in a pilot study of 27 overweight or obese children with psoriasis that excess adiposity typically came first. Moreover, among the roughly one-half of children with a family history of obesity, onset of psoriasis occurred a full 3 years earlier than in those without a positive family history (JAMA Dermatol. 2014;150:573-4).
In another small study, this by investigators at Tufts University, Boston, 6 of 20 children with psoriasis (30%) met criteria for the metabolic syndrome, compared with just 1 of 20 matched nonpsoriatic controls (Pediatr. Dermatol. 2013;30:700-5).
Dr. Paller said that if dermatologists and primary care physicians are to successfully collaborate in tackling the comorbid metabolic disorders associated with pediatric psoriasis, a prerequisite is that dermatologists are going to have to do a better job of educating their primary care colleagues about the skin disease as manifest in children.
“I think it’s very important that pediatricians are aware that psoriasis is a risk factor for metabolic syndrome. But pediatric psoriasis is often misdiagnosed by primary care physicians who mistake it for eczema or tinea infection or contact dermatitis,” according to the pediatric dermatologist.
In one eye-catching Australian study, she noted, a mere 9% of patients with pediatric psoriasis were correctly diagnosed before referral to a dermatologist (Australas. J. Dermatol. 2012;53:98-105).
Pediatric psoriasis: not just skin deep
In addition to the increased risk of metabolic disorders faced by pediatric psoriasis patients, other common comorbidities include depression, anxiety disorders, impaired self-esteem and quality of life, arthritis, and Crohn’s disease, Dr. Paller observed.
• Quality of life. “The quality of life impact of psoriasis is profound. It’s a highly visible disorder, which affects the development of self-esteem and social relationships,” Dr. Paller said.
Investigators at Texas A&M University applied the Pediatric Quality of Life Inventory Version 4.0 to 208 patients aged 2-17 years with moderate to severe psoriasis and compared the results to published data on children with arthritis, asthma, diabetes, and psychiatric disorders. Health-related quality of life turned out to be more impaired in the psoriasis patients than in those with diabetes. The quality-of-life impairment associated with pediatric psoriasis was comparable to that of having asthma or arthritis, albeit not as severe as for pediatric psychiatric disorders (Eur. J. Pediatr. 2012;171:485-92).
• Psychiatric disorders. A study of more than 7,400 pediatric psoriasis patients concluded they had an adjusted 25% increased risk of developing depression, compared with psoriasis-free controls, as well as a 32% increased risk of anxiety disorders and a 55% greater risk of bipolar disorder (J. Am. Acad. Dermatol. 2012;67:651-7.e2).
• Psoriatic arthritis. An estimated 1 in 10 U.S. children with psoriasis report having arthritis, often classified as juvenile idiopathic arthritis (JAMA Dermatol. 2013;149:1180-5).
• Crohn’s disease. A large German epidemiologic study concluded that psoriasis was associated with a 3.69-fold increased risk of Crohn’s disease. There was no increased risk of ulcerative colitis (Br. J. Dermatol. 2010;162:633-6).
Dr. Paller reported receiving research grants from Amgen and Leo and serving as a consultant to AbbVie.
VANCOUVER – Dermatologists and primary care physicians working collaboratively have a golden opportunity to improve the long-term health of pediatric psoriasis patients by addressing their predisposition to components of the metabolic syndrome, Dr. Amy S. Paller declared at the World Congress of Dermatology.
“I think we as dermatologists should be in touch with the primary care doctors of every one of our children with psoriasis. Together, we should be thinking about whether the child has metabolic issues and working jointly to most effectively counsel and evaluate these children for their potential risk for these metabolic disorders,” said Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University, Chicago.
Pediatric psoriasis is commonly associated with other comorbid conditions in addition to metabolic disorders. But the metabolic syndrome has recently become the focus of increasing attention given that cardiovascular disease is the No. 1 cause of death in the United States, and it appears that children with psoriasis may be getting a jump start on the atherosclerotic process.
By now, it’s well established that plaque psoriasis in adults is strongly associated with increased risks of diabetes, obesity, dyslipidemia, the metabolic syndrome, and cardiovascular disease. Mounting evidence indicates children and adolescents with psoriasis face the same risks.
Everyone knows how difficult it can be to make the long-term lifestyle changes that reverse obesity and its related metabolic disorders. But dermatologists, pediatricians, and family physicians have some leverage when it comes to pediatric psoriasis.
“Think about the fact that 30% of children with psoriasis have a first-degree relative with psoriasis, usually a parent. I think we need to think about counseling young adults with psoriasis early on, especially if that adult is overweight or obese, about the need for adopting a healthy lifestyle. If they do that, it’s not just for themselves but for their children, and we just might prevent pediatric psoriasis in that family or temper its severity through that healthy lifestyle intervention,” Dr. Paller continued.
The hope is that effectively addressing the metabolic comorbidities of pediatric psoriasis will modulate and improve the skin disease; in other words, that weight loss could improve psoriasis. As yet, however, that’s just a hope, as there is no persuasive supporting evidence.
“We’re looking towards ongoing adult trials to give us some clues about whether that’s the case,” she said.
Evidence for comorbidities
Some of the key evidence regarding the metabolic comorbidities of pediatric psoriasis comes from a landmark German epidemiologic study involving 33,981 pediatric psoriasis patients. The prevalence of psoriasis in German youth rose linearly from 0.12% at age 1 year to 1.2% at age 18. Pediatric psoriasis patients had significantly higher rates of diabetes, hyperlipidemia, obesity, and hypertension than did nonpsoriatic controls (Br. J. Dermatol. 2010;162:633-6).
A Kaiser Permanente study of nearly 711,000 youths aged 2-19 years showed that those who were overweight were 2.8-fold more likely than normal-weight youth to have severe or widespread psoriasis, while those who were moderately obese were at 2.9-fold increased risk and extremely obese youth were at 4.2-fold increased risk. Among adolescents, having psoriasis was associated with significantly higher mean total and LDL cholesterol, triglycerides, and alanine aminotransferase levels (J. Pediatr. 2011;159:577-83).
Recent evidence suggests that even before increased levels of LDL cholesterol and triglycerides are apparent in children with psoriasis, abnormalities in lipid function are present and may potentially serve as a novel marker for early cardiovascular risk. Dr. Paller cited a study presented by Dr. Wynnis L. Tom of Rady Children’s Hospital, San Diego, at the 2015 annual meeting of the Society for Investigative Dermatology. The case-control study included 50 children with psoriasis and 50 matched controls with a mean age of 13 years.
Like other investigators, Dr. Tom found that the psoriatic children had higher waist/hip ratios and more insulin resistance. While fasting lipid levels didn’t differ between the two groups, the psoriasis patients had significantly higher levels of atherogenic apolipoprotein B, fewer of the particularly cardioprotective large-size HDL particles, and reduced HDL efflux capacity. Stay tuned regarding these potential early markers, Dr. Paller advised.
She was lead author of a 409-patient international study that showed the risks of obesity and a high waist circumference rise with greater severity of pediatric psoriasis. Children with severe psoriasis were at 4.92-fold increased risk of obesity, compared with controls, while even those with mild psoriasis were at 3.6-fold increased risk (JAMA Dermatol. 2013;149:166-76).
Which comes first?
The question arises: Which comes first in children, the excess adiposity or the psoriasis? Dr. Paller said that although the final word isn’t in, she and her coworkers found in a pilot study of 27 overweight or obese children with psoriasis that excess adiposity typically came first. Moreover, among the roughly one-half of children with a family history of obesity, onset of psoriasis occurred a full 3 years earlier than in those without a positive family history (JAMA Dermatol. 2014;150:573-4).
In another small study, this by investigators at Tufts University, Boston, 6 of 20 children with psoriasis (30%) met criteria for the metabolic syndrome, compared with just 1 of 20 matched nonpsoriatic controls (Pediatr. Dermatol. 2013;30:700-5).
Dr. Paller said that if dermatologists and primary care physicians are to successfully collaborate in tackling the comorbid metabolic disorders associated with pediatric psoriasis, a prerequisite is that dermatologists are going to have to do a better job of educating their primary care colleagues about the skin disease as manifest in children.
“I think it’s very important that pediatricians are aware that psoriasis is a risk factor for metabolic syndrome. But pediatric psoriasis is often misdiagnosed by primary care physicians who mistake it for eczema or tinea infection or contact dermatitis,” according to the pediatric dermatologist.
In one eye-catching Australian study, she noted, a mere 9% of patients with pediatric psoriasis were correctly diagnosed before referral to a dermatologist (Australas. J. Dermatol. 2012;53:98-105).
Pediatric psoriasis: not just skin deep
In addition to the increased risk of metabolic disorders faced by pediatric psoriasis patients, other common comorbidities include depression, anxiety disorders, impaired self-esteem and quality of life, arthritis, and Crohn’s disease, Dr. Paller observed.
• Quality of life. “The quality of life impact of psoriasis is profound. It’s a highly visible disorder, which affects the development of self-esteem and social relationships,” Dr. Paller said.
Investigators at Texas A&M University applied the Pediatric Quality of Life Inventory Version 4.0 to 208 patients aged 2-17 years with moderate to severe psoriasis and compared the results to published data on children with arthritis, asthma, diabetes, and psychiatric disorders. Health-related quality of life turned out to be more impaired in the psoriasis patients than in those with diabetes. The quality-of-life impairment associated with pediatric psoriasis was comparable to that of having asthma or arthritis, albeit not as severe as for pediatric psychiatric disorders (Eur. J. Pediatr. 2012;171:485-92).
• Psychiatric disorders. A study of more than 7,400 pediatric psoriasis patients concluded they had an adjusted 25% increased risk of developing depression, compared with psoriasis-free controls, as well as a 32% increased risk of anxiety disorders and a 55% greater risk of bipolar disorder (J. Am. Acad. Dermatol. 2012;67:651-7.e2).
• Psoriatic arthritis. An estimated 1 in 10 U.S. children with psoriasis report having arthritis, often classified as juvenile idiopathic arthritis (JAMA Dermatol. 2013;149:1180-5).
• Crohn’s disease. A large German epidemiologic study concluded that psoriasis was associated with a 3.69-fold increased risk of Crohn’s disease. There was no increased risk of ulcerative colitis (Br. J. Dermatol. 2010;162:633-6).
Dr. Paller reported receiving research grants from Amgen and Leo and serving as a consultant to AbbVie.
EXPERT ANALYSIS FROM WCD 2015
Secukinumab proves successful against psoriatic arthritis in FUTURE 2 trial
Treatment of psoriatic arthritis over 24 weeks with the human anti-interleukin-17A monoclonal antibody secukinumab allowed a significantly higher percentage of patients who received 150-mg and 300-mg doses to achieve at least 20% improvement in the American College of Rheumatology response criteria than did placebo.
The results of the multicenter, double-blind, randomized, placebo-controlled FUTURE 2 trial hinted at the effectiveness of the 300-mg dosing regimen in treating patients who previously had not responded to treatment with tumor necrosis factor (TNF) inhibitors and did not show differences in effectiveness when used together with methotrexate. The drug did not have any particularly worrisome treatment-related adverse events, Dr. Iain McInnes of the University of Glasgow (Scotland) and his colleagues reported (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61134-5]).
At week 24, ACR20 responses occurred in 54 of 100 patients who received secukinumab 300 mg, 51 of 100 who took the 150-mg dosing regimen, 29 of 99 on the 75-mg dosing regimen, and 15 of 98 on placebo. The odds for achieving ACR20 at 24 weeks were nearly sevenfold higher for the 300-mg dose (odds ratio, 6.81; 95% confidence interval, 3.42-13.56; P < .0001) and 150-mg doses (OR, 6.52; 95% CI, 3.25-13.08; P < .0001) than with placebo.
In an editorial accompanying the report, Dr. Philip Helliwell and Dr. Laura Coates of the University of Leeds (England) said that it is important to note that responses at week 24 were similar to those observed at weeks 12 and 16 so that patients and physicians can “get an idea of response (or non-response) at an early timepoint” (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61170-9]).
Although radiographic progression wasn’t assessed in the FUTURE 2 trial, Dr. Helliwell and Dr. Coates noted that secukinumab inhibited radiographic damage in the unpublished FUTURE-1 trial, so “in view of its position in the cytokine hierarchy, interleukin-17 inhibition might prevent both of these features, and future studies with this class of drug should investigate this issue in detail.”
The lower response that seemed to have occurred in the secukinumab-treated patients who had previously used TNF inhibitors vs. those who had not “is in keeping with data for other treatments such as ustekinumab,” they wrote. The fact that there appeared to be a dose-response effect between the patients who received 150 mg secukinumab vs. 300 mg and had previously used TNF inhibitors suggests that the 300-mg dose might be more appropriate for patients who did not respond to TNF inhibitors and/or have moderate to severe psoriasis, for whom the 300-mg dose is already approved and recommended.
Read our meeting coverage of the FUTURE 2 trial at ACR 2014 here.
Treatment of psoriatic arthritis over 24 weeks with the human anti-interleukin-17A monoclonal antibody secukinumab allowed a significantly higher percentage of patients who received 150-mg and 300-mg doses to achieve at least 20% improvement in the American College of Rheumatology response criteria than did placebo.
The results of the multicenter, double-blind, randomized, placebo-controlled FUTURE 2 trial hinted at the effectiveness of the 300-mg dosing regimen in treating patients who previously had not responded to treatment with tumor necrosis factor (TNF) inhibitors and did not show differences in effectiveness when used together with methotrexate. The drug did not have any particularly worrisome treatment-related adverse events, Dr. Iain McInnes of the University of Glasgow (Scotland) and his colleagues reported (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61134-5]).
At week 24, ACR20 responses occurred in 54 of 100 patients who received secukinumab 300 mg, 51 of 100 who took the 150-mg dosing regimen, 29 of 99 on the 75-mg dosing regimen, and 15 of 98 on placebo. The odds for achieving ACR20 at 24 weeks were nearly sevenfold higher for the 300-mg dose (odds ratio, 6.81; 95% confidence interval, 3.42-13.56; P < .0001) and 150-mg doses (OR, 6.52; 95% CI, 3.25-13.08; P < .0001) than with placebo.
In an editorial accompanying the report, Dr. Philip Helliwell and Dr. Laura Coates of the University of Leeds (England) said that it is important to note that responses at week 24 were similar to those observed at weeks 12 and 16 so that patients and physicians can “get an idea of response (or non-response) at an early timepoint” (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61170-9]).
Although radiographic progression wasn’t assessed in the FUTURE 2 trial, Dr. Helliwell and Dr. Coates noted that secukinumab inhibited radiographic damage in the unpublished FUTURE-1 trial, so “in view of its position in the cytokine hierarchy, interleukin-17 inhibition might prevent both of these features, and future studies with this class of drug should investigate this issue in detail.”
The lower response that seemed to have occurred in the secukinumab-treated patients who had previously used TNF inhibitors vs. those who had not “is in keeping with data for other treatments such as ustekinumab,” they wrote. The fact that there appeared to be a dose-response effect between the patients who received 150 mg secukinumab vs. 300 mg and had previously used TNF inhibitors suggests that the 300-mg dose might be more appropriate for patients who did not respond to TNF inhibitors and/or have moderate to severe psoriasis, for whom the 300-mg dose is already approved and recommended.
Read our meeting coverage of the FUTURE 2 trial at ACR 2014 here.
Treatment of psoriatic arthritis over 24 weeks with the human anti-interleukin-17A monoclonal antibody secukinumab allowed a significantly higher percentage of patients who received 150-mg and 300-mg doses to achieve at least 20% improvement in the American College of Rheumatology response criteria than did placebo.
The results of the multicenter, double-blind, randomized, placebo-controlled FUTURE 2 trial hinted at the effectiveness of the 300-mg dosing regimen in treating patients who previously had not responded to treatment with tumor necrosis factor (TNF) inhibitors and did not show differences in effectiveness when used together with methotrexate. The drug did not have any particularly worrisome treatment-related adverse events, Dr. Iain McInnes of the University of Glasgow (Scotland) and his colleagues reported (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61134-5]).
At week 24, ACR20 responses occurred in 54 of 100 patients who received secukinumab 300 mg, 51 of 100 who took the 150-mg dosing regimen, 29 of 99 on the 75-mg dosing regimen, and 15 of 98 on placebo. The odds for achieving ACR20 at 24 weeks were nearly sevenfold higher for the 300-mg dose (odds ratio, 6.81; 95% confidence interval, 3.42-13.56; P < .0001) and 150-mg doses (OR, 6.52; 95% CI, 3.25-13.08; P < .0001) than with placebo.
In an editorial accompanying the report, Dr. Philip Helliwell and Dr. Laura Coates of the University of Leeds (England) said that it is important to note that responses at week 24 were similar to those observed at weeks 12 and 16 so that patients and physicians can “get an idea of response (or non-response) at an early timepoint” (Lancet 2015 June 28 [doi:10.1016/S0140-6736(15)61170-9]).
Although radiographic progression wasn’t assessed in the FUTURE 2 trial, Dr. Helliwell and Dr. Coates noted that secukinumab inhibited radiographic damage in the unpublished FUTURE-1 trial, so “in view of its position in the cytokine hierarchy, interleukin-17 inhibition might prevent both of these features, and future studies with this class of drug should investigate this issue in detail.”
The lower response that seemed to have occurred in the secukinumab-treated patients who had previously used TNF inhibitors vs. those who had not “is in keeping with data for other treatments such as ustekinumab,” they wrote. The fact that there appeared to be a dose-response effect between the patients who received 150 mg secukinumab vs. 300 mg and had previously used TNF inhibitors suggests that the 300-mg dose might be more appropriate for patients who did not respond to TNF inhibitors and/or have moderate to severe psoriasis, for whom the 300-mg dose is already approved and recommended.
Read our meeting coverage of the FUTURE 2 trial at ACR 2014 here.