User login
Large study identifies psoriasis as an independent risk factor for major depression
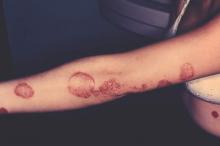
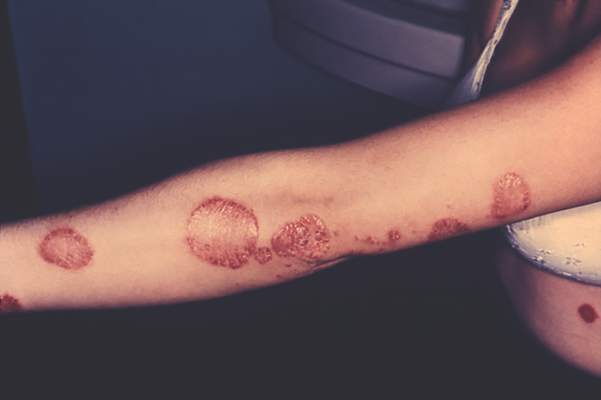
Self-reported psoriasis more than doubled the odds of major depression, even after accounting for demographic factors and comorbidities, reported the authors of a study that used data from the National Health and Nutrition Examination Survey.
In addition, depressive symptoms adversely affected the work and social lives of psoriasis patients in this first-in-kind study, said Dr. Brandon Cohen and his associates of the department of dermatology, New York University. “The risk of major depression was not associated with the self-reported degree of psoriasis. Therefore, our study supports that all patients with psoriasis, regardless of severity, are at risk for depressive symptoms and may benefit from depression screening,” they wrote. The study was published online on Sept. 30 in JAMA Dermatology (2015 Sep 30. doi: 10.1001/jamadermatol.2015.3605).
Psoriasis is known to increase the risk of major depression, as well as anxiety disorders, substance abuse, and suicidal ideation. To better understand whether disease severity or comorbidities affect the psoriasis-depression link, the researchers analyzed self-reported health data from 12,382 individuals who had responded to the National Health and Nutrition Examination Survey between 2009 and 2012. The investigators defined major depression as a score of at least 10 on the 27-point Patient Health Questionnaire-9 (PHQ-9). Psoriasis was self-reported, and some psoriasis patients were also asked to rate the severity of their skin disease.
After adjusting for factors such as age, sex, race, and history of myocardial infarction, stroke, and diabetes, the 351 individuals with psoriasis had more than twice the odds of major depression, compared with respondents without psoriasis (odds ratio, 2.09; 95% confidence interval, 1.41-3.11; P less than .001). Furthermore, the average PHQ-9 score among psoriasis patients was significantly higher than that among other respondents (4.54 vs. 3.22; P less than .001).
Neither severity of skin disease nor history of MI or stroke affected the relationship between psoriasis and depression. “The psychiatric burden of psoriasis may be more closely tied to patients’ perception of the social response to their appearance than objective disease severity,” the investigators said. They noted that as far as they know, this is the first study to use a nationally representative sample of patients to evaluate the association between psoriasis and major depression.
The investigators reported no funding sources and declared no competing interests.
Self-reported psoriasis more than doubled the odds of major depression, even after accounting for demographic factors and comorbidities, reported the authors of a study that used data from the National Health and Nutrition Examination Survey.
In addition, depressive symptoms adversely affected the work and social lives of psoriasis patients in this first-in-kind study, said Dr. Brandon Cohen and his associates of the department of dermatology, New York University. “The risk of major depression was not associated with the self-reported degree of psoriasis. Therefore, our study supports that all patients with psoriasis, regardless of severity, are at risk for depressive symptoms and may benefit from depression screening,” they wrote. The study was published online on Sept. 30 in JAMA Dermatology (2015 Sep 30. doi: 10.1001/jamadermatol.2015.3605).
Psoriasis is known to increase the risk of major depression, as well as anxiety disorders, substance abuse, and suicidal ideation. To better understand whether disease severity or comorbidities affect the psoriasis-depression link, the researchers analyzed self-reported health data from 12,382 individuals who had responded to the National Health and Nutrition Examination Survey between 2009 and 2012. The investigators defined major depression as a score of at least 10 on the 27-point Patient Health Questionnaire-9 (PHQ-9). Psoriasis was self-reported, and some psoriasis patients were also asked to rate the severity of their skin disease.
After adjusting for factors such as age, sex, race, and history of myocardial infarction, stroke, and diabetes, the 351 individuals with psoriasis had more than twice the odds of major depression, compared with respondents without psoriasis (odds ratio, 2.09; 95% confidence interval, 1.41-3.11; P less than .001). Furthermore, the average PHQ-9 score among psoriasis patients was significantly higher than that among other respondents (4.54 vs. 3.22; P less than .001).
Neither severity of skin disease nor history of MI or stroke affected the relationship between psoriasis and depression. “The psychiatric burden of psoriasis may be more closely tied to patients’ perception of the social response to their appearance than objective disease severity,” the investigators said. They noted that as far as they know, this is the first study to use a nationally representative sample of patients to evaluate the association between psoriasis and major depression.
The investigators reported no funding sources and declared no competing interests.
Self-reported psoriasis more than doubled the odds of major depression, even after accounting for demographic factors and comorbidities, reported the authors of a study that used data from the National Health and Nutrition Examination Survey.
In addition, depressive symptoms adversely affected the work and social lives of psoriasis patients in this first-in-kind study, said Dr. Brandon Cohen and his associates of the department of dermatology, New York University. “The risk of major depression was not associated with the self-reported degree of psoriasis. Therefore, our study supports that all patients with psoriasis, regardless of severity, are at risk for depressive symptoms and may benefit from depression screening,” they wrote. The study was published online on Sept. 30 in JAMA Dermatology (2015 Sep 30. doi: 10.1001/jamadermatol.2015.3605).
Psoriasis is known to increase the risk of major depression, as well as anxiety disorders, substance abuse, and suicidal ideation. To better understand whether disease severity or comorbidities affect the psoriasis-depression link, the researchers analyzed self-reported health data from 12,382 individuals who had responded to the National Health and Nutrition Examination Survey between 2009 and 2012. The investigators defined major depression as a score of at least 10 on the 27-point Patient Health Questionnaire-9 (PHQ-9). Psoriasis was self-reported, and some psoriasis patients were also asked to rate the severity of their skin disease.
After adjusting for factors such as age, sex, race, and history of myocardial infarction, stroke, and diabetes, the 351 individuals with psoriasis had more than twice the odds of major depression, compared with respondents without psoriasis (odds ratio, 2.09; 95% confidence interval, 1.41-3.11; P less than .001). Furthermore, the average PHQ-9 score among psoriasis patients was significantly higher than that among other respondents (4.54 vs. 3.22; P less than .001).
Neither severity of skin disease nor history of MI or stroke affected the relationship between psoriasis and depression. “The psychiatric burden of psoriasis may be more closely tied to patients’ perception of the social response to their appearance than objective disease severity,” the investigators said. They noted that as far as they know, this is the first study to use a nationally representative sample of patients to evaluate the association between psoriasis and major depression.
The investigators reported no funding sources and declared no competing interests.
FROM JAMA DERMATOLOGY
Key clinical point:Psoriasis was associated with a significantly increased risk of major depression in a large population-based U.S. study.
Major finding: Self-reported psoriasis more than doubled the odds of meeting criteria for major depression (OR, 2.09; P less than .001).
Data source: The cross-sectional study evaluated data from 12,382 National Health and Nutrition Examination Survey participants, who responded between 2009 and 2012.
Disclosures: The authors reported no funding sources and declared no competing interests.
Nail Psoriasis Diagnosis and Treatment Recommendations
Clinical diagnosis of nail psoriasis may be difficult, as many of its symptoms are seen in other nail disorders. To ensure accurate diagnosis, it is important to recognize the characteristic signs of nail psoriasis and clarify which part of the nail matrix is affected. Dr. Antonella Tosti discusses common symptoms of nail psoriasis and provides treatment and nail care recommendations for patients. She also notes that psoriasis of the nails should raise suspicion of possible joint involvement.
The psoriasis audiocast series is created in collaboration with Cutis® and the National Psoriasis Foundation®.
Clinical diagnosis of nail psoriasis may be difficult, as many of its symptoms are seen in other nail disorders. To ensure accurate diagnosis, it is important to recognize the characteristic signs of nail psoriasis and clarify which part of the nail matrix is affected. Dr. Antonella Tosti discusses common symptoms of nail psoriasis and provides treatment and nail care recommendations for patients. She also notes that psoriasis of the nails should raise suspicion of possible joint involvement.
The psoriasis audiocast series is created in collaboration with Cutis® and the National Psoriasis Foundation®.
Clinical diagnosis of nail psoriasis may be difficult, as many of its symptoms are seen in other nail disorders. To ensure accurate diagnosis, it is important to recognize the characteristic signs of nail psoriasis and clarify which part of the nail matrix is affected. Dr. Antonella Tosti discusses common symptoms of nail psoriasis and provides treatment and nail care recommendations for patients. She also notes that psoriasis of the nails should raise suspicion of possible joint involvement.
The psoriasis audiocast series is created in collaboration with Cutis® and the National Psoriasis Foundation®.
PsA, PsC do not affect total hip replacement outcomes
Neither psoriatic arthritis (PsA) nor cutaneous psoriasis (PsC) is an independent predictor of poor postoperative pain or function following a total hip arthroplasty, according to the results of a case-control study by Dr. Lisa A. Mandl and her colleagues.
The study’s participants underwent surgery between May 1, 2007, and Dec. 31, 2010, in a center that performs more than 4,300 THAs annually. All subjects lived for at least 2 years after their operations. The researchers compared pre- and postoperative data from patients in the following three categories: those with PsA, those with PsC without evidence of inflammatory arthritis, and those with osteoarthritis (OA). Patients with OA comprised the control group, which excluded any patient who self-reported a history of PsA, rheumatoid arthritis, lupus erythematosus, or any other systematic rheumatic disease, or who had documentation of skin psoriasis. The researchers acquired postoperative self-report data from 47 PsA patients, 106 PsC patients, and 864 OA patients. Seventeen percent of patients submitted information on their status at 1 year, 69% at 2 years, and 14% at 3-5 years.
The primary outcomes of interest were postoperative pain and function, which were assessed via the Hip Osteoarthritis Outcome Score (HOOS), from which the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was derived.
There were no statistically significant differences in postoperative WOMAC pain or function scores between the three groups of patients (P = .78 and .96, respectively). The mean pain scores were 14.9, 6.1, and 15.8 for patients with PsA, PsC, and OA, respectively. These patients’ mean function scores were 16.3, 19.6, and 18.8 for the PsA, PsC and OA groups, respectively.
Overall levels of satisfaction with the surgery were similar among the three groups (P = .54). Ninety-three percent of the PsA patients, 79% of the PsC patients, and 84% of the OA patients were “very satisfied” with their total hip arthroplasty. Between 1% and 3% of each group reported being “very dissatisfied” with their surgery. The researchers found that extent of skin disease was not associated with worse postoperative pain or function.
“Further work needs to be done to better understand the interplay of disease activity and quality of life on the outcomes of [total hip arthroplasty] in PsA and PsC,” they wrote.
Read the report in Arthritis & Rheumatology (doi: 10.1002/art.39431).
Neither psoriatic arthritis (PsA) nor cutaneous psoriasis (PsC) is an independent predictor of poor postoperative pain or function following a total hip arthroplasty, according to the results of a case-control study by Dr. Lisa A. Mandl and her colleagues.
The study’s participants underwent surgery between May 1, 2007, and Dec. 31, 2010, in a center that performs more than 4,300 THAs annually. All subjects lived for at least 2 years after their operations. The researchers compared pre- and postoperative data from patients in the following three categories: those with PsA, those with PsC without evidence of inflammatory arthritis, and those with osteoarthritis (OA). Patients with OA comprised the control group, which excluded any patient who self-reported a history of PsA, rheumatoid arthritis, lupus erythematosus, or any other systematic rheumatic disease, or who had documentation of skin psoriasis. The researchers acquired postoperative self-report data from 47 PsA patients, 106 PsC patients, and 864 OA patients. Seventeen percent of patients submitted information on their status at 1 year, 69% at 2 years, and 14% at 3-5 years.
The primary outcomes of interest were postoperative pain and function, which were assessed via the Hip Osteoarthritis Outcome Score (HOOS), from which the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was derived.
There were no statistically significant differences in postoperative WOMAC pain or function scores between the three groups of patients (P = .78 and .96, respectively). The mean pain scores were 14.9, 6.1, and 15.8 for patients with PsA, PsC, and OA, respectively. These patients’ mean function scores were 16.3, 19.6, and 18.8 for the PsA, PsC and OA groups, respectively.
Overall levels of satisfaction with the surgery were similar among the three groups (P = .54). Ninety-three percent of the PsA patients, 79% of the PsC patients, and 84% of the OA patients were “very satisfied” with their total hip arthroplasty. Between 1% and 3% of each group reported being “very dissatisfied” with their surgery. The researchers found that extent of skin disease was not associated with worse postoperative pain or function.
“Further work needs to be done to better understand the interplay of disease activity and quality of life on the outcomes of [total hip arthroplasty] in PsA and PsC,” they wrote.
Read the report in Arthritis & Rheumatology (doi: 10.1002/art.39431).
Neither psoriatic arthritis (PsA) nor cutaneous psoriasis (PsC) is an independent predictor of poor postoperative pain or function following a total hip arthroplasty, according to the results of a case-control study by Dr. Lisa A. Mandl and her colleagues.
The study’s participants underwent surgery between May 1, 2007, and Dec. 31, 2010, in a center that performs more than 4,300 THAs annually. All subjects lived for at least 2 years after their operations. The researchers compared pre- and postoperative data from patients in the following three categories: those with PsA, those with PsC without evidence of inflammatory arthritis, and those with osteoarthritis (OA). Patients with OA comprised the control group, which excluded any patient who self-reported a history of PsA, rheumatoid arthritis, lupus erythematosus, or any other systematic rheumatic disease, or who had documentation of skin psoriasis. The researchers acquired postoperative self-report data from 47 PsA patients, 106 PsC patients, and 864 OA patients. Seventeen percent of patients submitted information on their status at 1 year, 69% at 2 years, and 14% at 3-5 years.
The primary outcomes of interest were postoperative pain and function, which were assessed via the Hip Osteoarthritis Outcome Score (HOOS), from which the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) was derived.
There were no statistically significant differences in postoperative WOMAC pain or function scores between the three groups of patients (P = .78 and .96, respectively). The mean pain scores were 14.9, 6.1, and 15.8 for patients with PsA, PsC, and OA, respectively. These patients’ mean function scores were 16.3, 19.6, and 18.8 for the PsA, PsC and OA groups, respectively.
Overall levels of satisfaction with the surgery were similar among the three groups (P = .54). Ninety-three percent of the PsA patients, 79% of the PsC patients, and 84% of the OA patients were “very satisfied” with their total hip arthroplasty. Between 1% and 3% of each group reported being “very dissatisfied” with their surgery. The researchers found that extent of skin disease was not associated with worse postoperative pain or function.
“Further work needs to be done to better understand the interplay of disease activity and quality of life on the outcomes of [total hip arthroplasty] in PsA and PsC,” they wrote.
Read the report in Arthritis & Rheumatology (doi: 10.1002/art.39431).
FROM ARTHRITIS & RHEUMATOLOGY
Screening Tests for Psoriasis Patients
Dermatologists should screen psoriasis patients for tuberculosis (TB) and hepatitis B virus (HBV) before beginning treatment with tumor necrosis factor (TNF) inhibitors or biologics, according to Jashin J. Wu, MD, Director of the Psoriasis Clinic, Department of Dermatology, Kaiser Permanente Los Angeles Medical Center, California, at the 2015 Summer Academy Meeting of the American Academy of Dermatology. Dr. Wu provided 10 psoriasis pearls in 10 minutes. Among them he reviewed important screenings for psoriasis patients.
The National Psoriasis Foundation’s consensus statement on screening for latent TB infection in patients with psoriasis treated with systemic and biologic agents (J Am Acad Dermatol. 2008;59:209-217) indicated that chronic immunosuppression is a known risk factor for allowing latent TB to transform into active TB. Therefore, immunosuppressive and immunomodulatory therapies for psoriasis and psoriatic arthritis may be associated with an increased rate of active TB in some patients receiving these therapies. The consensus statement reported, “It is, therefore, of utmost importance to appropriately screen all patients for latent TB infection prior to initiating any immunologic therapy. Delaying immunologic therapy until latent TB infection prophylaxis is completed is preferable.”
When screening for tuberculosis, Dr. Wu recommends the IFN-γ release assay instead of the purified protein derivative (tuberculin)(PPD) test because it is more sensitive and more specific than the PPD in diagnosing latent TB infection in immunocompetent individuals. Additionally, Dr. Wu reports that the IFN-γ release assay is more specific than the PPD in those who have received the BCG vaccine and is more convenient for patients.
For patients who may receive TNF inhibitors, Dr. Wu recommends screening for HBV using triple serology testing: hepatitis B surface antigen, hepatitis B surface antibody, and hepatitis B core antibody. He also recommends this screening for patients who may be on biologics including ustekinumab and secukinumab. If the patient is at risk for reactivation of HBV, dermatologists should check liver function tests, hepatitis B surface antibody, hepatitis B core e antigen, and HBV DNA counts. Routine follow-up with testing for reactivation should continue for at least 6 months after the TNF inhibitor is discontinued. If patients have chronic HBV and biologics are considered, etanercept is recommended as first-line therapy.
Dermatologists should screen psoriasis patients for tuberculosis (TB) and hepatitis B virus (HBV) before beginning treatment with tumor necrosis factor (TNF) inhibitors or biologics, according to Jashin J. Wu, MD, Director of the Psoriasis Clinic, Department of Dermatology, Kaiser Permanente Los Angeles Medical Center, California, at the 2015 Summer Academy Meeting of the American Academy of Dermatology. Dr. Wu provided 10 psoriasis pearls in 10 minutes. Among them he reviewed important screenings for psoriasis patients.
The National Psoriasis Foundation’s consensus statement on screening for latent TB infection in patients with psoriasis treated with systemic and biologic agents (J Am Acad Dermatol. 2008;59:209-217) indicated that chronic immunosuppression is a known risk factor for allowing latent TB to transform into active TB. Therefore, immunosuppressive and immunomodulatory therapies for psoriasis and psoriatic arthritis may be associated with an increased rate of active TB in some patients receiving these therapies. The consensus statement reported, “It is, therefore, of utmost importance to appropriately screen all patients for latent TB infection prior to initiating any immunologic therapy. Delaying immunologic therapy until latent TB infection prophylaxis is completed is preferable.”
When screening for tuberculosis, Dr. Wu recommends the IFN-γ release assay instead of the purified protein derivative (tuberculin)(PPD) test because it is more sensitive and more specific than the PPD in diagnosing latent TB infection in immunocompetent individuals. Additionally, Dr. Wu reports that the IFN-γ release assay is more specific than the PPD in those who have received the BCG vaccine and is more convenient for patients.
For patients who may receive TNF inhibitors, Dr. Wu recommends screening for HBV using triple serology testing: hepatitis B surface antigen, hepatitis B surface antibody, and hepatitis B core antibody. He also recommends this screening for patients who may be on biologics including ustekinumab and secukinumab. If the patient is at risk for reactivation of HBV, dermatologists should check liver function tests, hepatitis B surface antibody, hepatitis B core e antigen, and HBV DNA counts. Routine follow-up with testing for reactivation should continue for at least 6 months after the TNF inhibitor is discontinued. If patients have chronic HBV and biologics are considered, etanercept is recommended as first-line therapy.
Dermatologists should screen psoriasis patients for tuberculosis (TB) and hepatitis B virus (HBV) before beginning treatment with tumor necrosis factor (TNF) inhibitors or biologics, according to Jashin J. Wu, MD, Director of the Psoriasis Clinic, Department of Dermatology, Kaiser Permanente Los Angeles Medical Center, California, at the 2015 Summer Academy Meeting of the American Academy of Dermatology. Dr. Wu provided 10 psoriasis pearls in 10 minutes. Among them he reviewed important screenings for psoriasis patients.
The National Psoriasis Foundation’s consensus statement on screening for latent TB infection in patients with psoriasis treated with systemic and biologic agents (J Am Acad Dermatol. 2008;59:209-217) indicated that chronic immunosuppression is a known risk factor for allowing latent TB to transform into active TB. Therefore, immunosuppressive and immunomodulatory therapies for psoriasis and psoriatic arthritis may be associated with an increased rate of active TB in some patients receiving these therapies. The consensus statement reported, “It is, therefore, of utmost importance to appropriately screen all patients for latent TB infection prior to initiating any immunologic therapy. Delaying immunologic therapy until latent TB infection prophylaxis is completed is preferable.”
When screening for tuberculosis, Dr. Wu recommends the IFN-γ release assay instead of the purified protein derivative (tuberculin)(PPD) test because it is more sensitive and more specific than the PPD in diagnosing latent TB infection in immunocompetent individuals. Additionally, Dr. Wu reports that the IFN-γ release assay is more specific than the PPD in those who have received the BCG vaccine and is more convenient for patients.
For patients who may receive TNF inhibitors, Dr. Wu recommends screening for HBV using triple serology testing: hepatitis B surface antigen, hepatitis B surface antibody, and hepatitis B core antibody. He also recommends this screening for patients who may be on biologics including ustekinumab and secukinumab. If the patient is at risk for reactivation of HBV, dermatologists should check liver function tests, hepatitis B surface antibody, hepatitis B core e antigen, and HBV DNA counts. Routine follow-up with testing for reactivation should continue for at least 6 months after the TNF inhibitor is discontinued. If patients have chronic HBV and biologics are considered, etanercept is recommended as first-line therapy.
Dermatoses of Pregnancy
After, test your knowledge by answering the 5 practice questions.
Practice Questions
1. Which dermatosis of pregnancy occurs during the third trimester and is associated with multiple gestation pregnancies?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. intrahepatic cholestasis of pregnancy
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
2. Which dermatosis of pregnancy frequently flares after delivery?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. polymorphic eruption of pregnancy
d. prurigo gravidarum
e. prurigo of pregnancy
3. Which dermatosis of pregnancy has lesions that have a predilection for the abdominal striae?
a. cholestasis of pregnancy
b. gestational pemphigoid
c. prurigo gestationis
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
4. Which dermatosis of pregnancy has a risk for the development of hydatidiform moles and choriocarcinomas?
a. atopic eruption of pregnancy
b. cholestasis of pregnancy
c. gestational pemphigoid
d. pruritic urticarial papules and plaques of pregnancy
e. toxic erythema of pregnancy
5. Intrahepatic cholestasis of pregnancy has been associated with:
a. fetal mortality as high as 13%
b. jaundice in 20% of cases
c. onset in the third trimester of pregnancy
d. recurrence in subsequent pregnancies
e. all of the above
The answers appear on the next page.
1. Which dermatosis of pregnancy occurs during the third trimester and is associated with multiple gestation pregnancies?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. intrahepatic cholestasis of pregnancy
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
2. Which dermatosis of pregnancy frequently flares after delivery?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. polymorphic eruption of pregnancy
d. prurigo gravidarum
e. prurigo of pregnancy
3. Which dermatosis of pregnancy has lesions that have a predilection for the abdominal striae?
a. cholestasis of pregnancy
b. gestational pemphigoid
c. prurigo gestationis
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
4. Which dermatosis of pregnancy has a risk for the development of hydatidiform moles and choriocarcinomas?
a. atopic eruption of pregnancy
b. cholestasis of pregnancy
c. gestational pemphigoid
d. pruritic urticarial papules and plaques of pregnancy
e. toxic erythema of pregnancy
5. Intrahepatic cholestasis of pregnancy has been associated with:
a. fetal mortality as high as 13%
b. jaundice in 20% of cases
c. onset in the third trimester of pregnancy
d. recurrence in subsequent pregnancies
e. all of the above
After, test your knowledge by answering the 5 practice questions.
Practice Questions
1. Which dermatosis of pregnancy occurs during the third trimester and is associated with multiple gestation pregnancies?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. intrahepatic cholestasis of pregnancy
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
2. Which dermatosis of pregnancy frequently flares after delivery?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. polymorphic eruption of pregnancy
d. prurigo gravidarum
e. prurigo of pregnancy
3. Which dermatosis of pregnancy has lesions that have a predilection for the abdominal striae?
a. cholestasis of pregnancy
b. gestational pemphigoid
c. prurigo gestationis
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
4. Which dermatosis of pregnancy has a risk for the development of hydatidiform moles and choriocarcinomas?
a. atopic eruption of pregnancy
b. cholestasis of pregnancy
c. gestational pemphigoid
d. pruritic urticarial papules and plaques of pregnancy
e. toxic erythema of pregnancy
5. Intrahepatic cholestasis of pregnancy has been associated with:
a. fetal mortality as high as 13%
b. jaundice in 20% of cases
c. onset in the third trimester of pregnancy
d. recurrence in subsequent pregnancies
e. all of the above
The answers appear on the next page.
1. Which dermatosis of pregnancy occurs during the third trimester and is associated with multiple gestation pregnancies?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. intrahepatic cholestasis of pregnancy
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
2. Which dermatosis of pregnancy frequently flares after delivery?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. polymorphic eruption of pregnancy
d. prurigo gravidarum
e. prurigo of pregnancy
3. Which dermatosis of pregnancy has lesions that have a predilection for the abdominal striae?
a. cholestasis of pregnancy
b. gestational pemphigoid
c. prurigo gestationis
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
4. Which dermatosis of pregnancy has a risk for the development of hydatidiform moles and choriocarcinomas?
a. atopic eruption of pregnancy
b. cholestasis of pregnancy
c. gestational pemphigoid
d. pruritic urticarial papules and plaques of pregnancy
e. toxic erythema of pregnancy
5. Intrahepatic cholestasis of pregnancy has been associated with:
a. fetal mortality as high as 13%
b. jaundice in 20% of cases
c. onset in the third trimester of pregnancy
d. recurrence in subsequent pregnancies
e. all of the above
After, test your knowledge by answering the 5 practice questions.
Practice Questions
1. Which dermatosis of pregnancy occurs during the third trimester and is associated with multiple gestation pregnancies?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. intrahepatic cholestasis of pregnancy
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
2. Which dermatosis of pregnancy frequently flares after delivery?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. polymorphic eruption of pregnancy
d. prurigo gravidarum
e. prurigo of pregnancy
3. Which dermatosis of pregnancy has lesions that have a predilection for the abdominal striae?
a. cholestasis of pregnancy
b. gestational pemphigoid
c. prurigo gestationis
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
4. Which dermatosis of pregnancy has a risk for the development of hydatidiform moles and choriocarcinomas?
a. atopic eruption of pregnancy
b. cholestasis of pregnancy
c. gestational pemphigoid
d. pruritic urticarial papules and plaques of pregnancy
e. toxic erythema of pregnancy
5. Intrahepatic cholestasis of pregnancy has been associated with:
a. fetal mortality as high as 13%
b. jaundice in 20% of cases
c. onset in the third trimester of pregnancy
d. recurrence in subsequent pregnancies
e. all of the above
The answers appear on the next page.
1. Which dermatosis of pregnancy occurs during the third trimester and is associated with multiple gestation pregnancies?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. intrahepatic cholestasis of pregnancy
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
2. Which dermatosis of pregnancy frequently flares after delivery?
a. atopic eruption of pregnancy
b. gestational pemphigoid
c. polymorphic eruption of pregnancy
d. prurigo gravidarum
e. prurigo of pregnancy
3. Which dermatosis of pregnancy has lesions that have a predilection for the abdominal striae?
a. cholestasis of pregnancy
b. gestational pemphigoid
c. prurigo gestationis
d. prurigo of pregnancy
e. pruritic urticarial papules and plaques of pregnancy
4. Which dermatosis of pregnancy has a risk for the development of hydatidiform moles and choriocarcinomas?
a. atopic eruption of pregnancy
b. cholestasis of pregnancy
c. gestational pemphigoid
d. pruritic urticarial papules and plaques of pregnancy
e. toxic erythema of pregnancy
5. Intrahepatic cholestasis of pregnancy has been associated with:
a. fetal mortality as high as 13%
b. jaundice in 20% of cases
c. onset in the third trimester of pregnancy
d. recurrence in subsequent pregnancies
e. all of the above
Tissue-based treatment could improve psoriatic arthritis outcomes
Efforts underway to tailor psoriatic arthritis treatments to the various tissues involved in the pathogenesis of the disease could optimize therapeutic approaches, Dr. Iain McInnes said at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
“Psoriatic arthritis [PsA] is a disease whose treatment is evolving quickly, ” added Dr. McInnes, professor of medicine and director of the Institute of Infection, Immunity, and Inflammation at the University of Glasgow (Scotland). “We have new ideas about pathogenesis, emerging studies that inform the best strategic approach to disease management, including potential value in a treat-to-target approach, and new therapeutics that offer a range of options to allow us to optimize treatment.”
He pointed to recent evidence for a diverse pathogenesis of PsA subtypes. Synovial membranes in PsA have high levels of tumor necrosis factor (TNF)-alpha, while resident populations of interleukin (IL)-23 receptor–positive T cells are found in entheses and IL-17 is abundant in psoriatic lesions. Genome-wide association studies have underscored the role of the IL-23 and IL-17 cytokine axis, while other work has implicated Th17 cytokines in mesenchymal tissue changes.
Although drug development efforts have typically viewed rheumatoid arthritis and PsA as homogeneous, “we can appreciate the extent of articular, entheseal, and cutaneous involvement, and look to tailor therapies accordingly,” Dr. McInnes said.
So what will the next decade look like for PsA? Therapy will center around TNF-alpha inhibitors, new cytokine pathways, and small-molecule inhibitors, according to Dr. McInnes. While anti-TNF agents are not universally tolerated or effective, their “well-established safety profile” and positive effects on American College of Rheumatology response criteria and radiographic endpoints generally outweigh risks of infection and problems with decreasing long-term drug survival, he said. These agents also can improve disease activity, skin symptoms, physical function, fatigue, overall quality of life, enthesitis, and dactylitis, he noted.
Other recent additions to the PsA arsenal include the IL-12/IL-23 inhibitor ustekinumab (Stelara), which in the PSUMMIT 1 and 2 trials was associated with decreased rates of radiographic progression and dose-dependent improvements in enthesitis, Dr. McInnes said. The phosphodiesterase-4 inhibitor apremilast (Otezla) was associated with significantly better ACR 20 response at week 16 and significantly greater improvements in Maastricht Ankylosing Spondylitis Enthesis Score and measures of dactylitis at week 24 of the PALACE trials. Most recently, in the FUTURE1 and FUTURE2 trials, the IL-17A inhibitor secukinumab (Cosentyx) beat placebo in terms of ACR 20, ACR 50, ACR 70, resolution of dactylitis and enthesitis, and radiographic measures of disease progression.
The availability of effective biologics raises the possibility of a treat-to-target approach geared toward clinical remission or low disease activity, Dr. McInnes concluded. “Targets for low disease activity in PsA are now available. Composite measures of disease activity, such as minimal disease activity (MDA), encompass all clinically important aspects of PsA,” he emphasized. While studies still need to pinpoint the best therapeutic pathways for disease subtypes, the first treat-to-target study of PsA found that tailoring therapy based on remission or minimal disease activity led to significantly better ACR 20, ACR 50, and ACR 70 responses at week 48, compared with standard care, he noted.
Dr. McInnes reported receiving research funding or honoraria from Novartis, Janssen, AbbVie, UCB, Pfizer, and MSD Pharmaceuticals, all of which make medications for rheumatic diseases. Global Academy for Medical Education and this news organization are owned by the same parent company.
Efforts underway to tailor psoriatic arthritis treatments to the various tissues involved in the pathogenesis of the disease could optimize therapeutic approaches, Dr. Iain McInnes said at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
“Psoriatic arthritis [PsA] is a disease whose treatment is evolving quickly, ” added Dr. McInnes, professor of medicine and director of the Institute of Infection, Immunity, and Inflammation at the University of Glasgow (Scotland). “We have new ideas about pathogenesis, emerging studies that inform the best strategic approach to disease management, including potential value in a treat-to-target approach, and new therapeutics that offer a range of options to allow us to optimize treatment.”
He pointed to recent evidence for a diverse pathogenesis of PsA subtypes. Synovial membranes in PsA have high levels of tumor necrosis factor (TNF)-alpha, while resident populations of interleukin (IL)-23 receptor–positive T cells are found in entheses and IL-17 is abundant in psoriatic lesions. Genome-wide association studies have underscored the role of the IL-23 and IL-17 cytokine axis, while other work has implicated Th17 cytokines in mesenchymal tissue changes.
Although drug development efforts have typically viewed rheumatoid arthritis and PsA as homogeneous, “we can appreciate the extent of articular, entheseal, and cutaneous involvement, and look to tailor therapies accordingly,” Dr. McInnes said.
So what will the next decade look like for PsA? Therapy will center around TNF-alpha inhibitors, new cytokine pathways, and small-molecule inhibitors, according to Dr. McInnes. While anti-TNF agents are not universally tolerated or effective, their “well-established safety profile” and positive effects on American College of Rheumatology response criteria and radiographic endpoints generally outweigh risks of infection and problems with decreasing long-term drug survival, he said. These agents also can improve disease activity, skin symptoms, physical function, fatigue, overall quality of life, enthesitis, and dactylitis, he noted.
Other recent additions to the PsA arsenal include the IL-12/IL-23 inhibitor ustekinumab (Stelara), which in the PSUMMIT 1 and 2 trials was associated with decreased rates of radiographic progression and dose-dependent improvements in enthesitis, Dr. McInnes said. The phosphodiesterase-4 inhibitor apremilast (Otezla) was associated with significantly better ACR 20 response at week 16 and significantly greater improvements in Maastricht Ankylosing Spondylitis Enthesis Score and measures of dactylitis at week 24 of the PALACE trials. Most recently, in the FUTURE1 and FUTURE2 trials, the IL-17A inhibitor secukinumab (Cosentyx) beat placebo in terms of ACR 20, ACR 50, ACR 70, resolution of dactylitis and enthesitis, and radiographic measures of disease progression.
The availability of effective biologics raises the possibility of a treat-to-target approach geared toward clinical remission or low disease activity, Dr. McInnes concluded. “Targets for low disease activity in PsA are now available. Composite measures of disease activity, such as minimal disease activity (MDA), encompass all clinically important aspects of PsA,” he emphasized. While studies still need to pinpoint the best therapeutic pathways for disease subtypes, the first treat-to-target study of PsA found that tailoring therapy based on remission or minimal disease activity led to significantly better ACR 20, ACR 50, and ACR 70 responses at week 48, compared with standard care, he noted.
Dr. McInnes reported receiving research funding or honoraria from Novartis, Janssen, AbbVie, UCB, Pfizer, and MSD Pharmaceuticals, all of which make medications for rheumatic diseases. Global Academy for Medical Education and this news organization are owned by the same parent company.
Efforts underway to tailor psoriatic arthritis treatments to the various tissues involved in the pathogenesis of the disease could optimize therapeutic approaches, Dr. Iain McInnes said at the annual Perspectives in Rheumatic Diseases held by Global Academy for Medical Education.
“Psoriatic arthritis [PsA] is a disease whose treatment is evolving quickly, ” added Dr. McInnes, professor of medicine and director of the Institute of Infection, Immunity, and Inflammation at the University of Glasgow (Scotland). “We have new ideas about pathogenesis, emerging studies that inform the best strategic approach to disease management, including potential value in a treat-to-target approach, and new therapeutics that offer a range of options to allow us to optimize treatment.”
He pointed to recent evidence for a diverse pathogenesis of PsA subtypes. Synovial membranes in PsA have high levels of tumor necrosis factor (TNF)-alpha, while resident populations of interleukin (IL)-23 receptor–positive T cells are found in entheses and IL-17 is abundant in psoriatic lesions. Genome-wide association studies have underscored the role of the IL-23 and IL-17 cytokine axis, while other work has implicated Th17 cytokines in mesenchymal tissue changes.
Although drug development efforts have typically viewed rheumatoid arthritis and PsA as homogeneous, “we can appreciate the extent of articular, entheseal, and cutaneous involvement, and look to tailor therapies accordingly,” Dr. McInnes said.
So what will the next decade look like for PsA? Therapy will center around TNF-alpha inhibitors, new cytokine pathways, and small-molecule inhibitors, according to Dr. McInnes. While anti-TNF agents are not universally tolerated or effective, their “well-established safety profile” and positive effects on American College of Rheumatology response criteria and radiographic endpoints generally outweigh risks of infection and problems with decreasing long-term drug survival, he said. These agents also can improve disease activity, skin symptoms, physical function, fatigue, overall quality of life, enthesitis, and dactylitis, he noted.
Other recent additions to the PsA arsenal include the IL-12/IL-23 inhibitor ustekinumab (Stelara), which in the PSUMMIT 1 and 2 trials was associated with decreased rates of radiographic progression and dose-dependent improvements in enthesitis, Dr. McInnes said. The phosphodiesterase-4 inhibitor apremilast (Otezla) was associated with significantly better ACR 20 response at week 16 and significantly greater improvements in Maastricht Ankylosing Spondylitis Enthesis Score and measures of dactylitis at week 24 of the PALACE trials. Most recently, in the FUTURE1 and FUTURE2 trials, the IL-17A inhibitor secukinumab (Cosentyx) beat placebo in terms of ACR 20, ACR 50, ACR 70, resolution of dactylitis and enthesitis, and radiographic measures of disease progression.
The availability of effective biologics raises the possibility of a treat-to-target approach geared toward clinical remission or low disease activity, Dr. McInnes concluded. “Targets for low disease activity in PsA are now available. Composite measures of disease activity, such as minimal disease activity (MDA), encompass all clinically important aspects of PsA,” he emphasized. While studies still need to pinpoint the best therapeutic pathways for disease subtypes, the first treat-to-target study of PsA found that tailoring therapy based on remission or minimal disease activity led to significantly better ACR 20, ACR 50, and ACR 70 responses at week 48, compared with standard care, he noted.
Dr. McInnes reported receiving research funding or honoraria from Novartis, Janssen, AbbVie, UCB, Pfizer, and MSD Pharmaceuticals, all of which make medications for rheumatic diseases. Global Academy for Medical Education and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM THE ANNUAL PERSPECTIVES IN RHEUMATIC DISEASES
Methotrexate ups risk of liver abnormalities, but not serious hepatic events
Methotrexate use more than doubled the risk of liver enzyme abnormalities but was not tied to serious hepatic outcomes among patients with rheumatic diseases, according to a meta-analysis of 32 randomized, controlled trials.
“Serious liver events were rare, with a suggestion of a trend towards fewer poor liver outcomes,” wrote Dr. Richard Conway of Galway (Ireland) University Hospitals. “The short duration of the included studies and the lack of liver biopsies are limitations, but the available data provide robust evidence for short-term safety” of methotrexate, wrote Dr. Conway and his associates.
Methotrexate is a first-line RA therapy with efficacy in several other inflammatory diseases, but concerns about toxicity have limited its use, the researchers noted. Clinicians have particularly debated its hepatic effects, in part because it can be “exceedingly difficult” to determine if liver disease is preexisting, methotrexate related, or caused by other medications or comorbidities, they said. To better assess the issue, they searched PubMed and the Cochrane Central Register of Controlled Trials for relevant double-blind, randomized, controlled trials published between 1990 and 2014. They found 32 such trials that included 13,177 patients with rheumatoid arthritis, psoriasis, psoriatic arthritis or inflammatory bowel disease (Semin Arthritis Rheum. 2015;45:156-62). Methotrexate use approximately doubled the risk of any adverse hepatic event (relative risk, 2.19; 95% confidence interval, 1.73-2.77) and of major and minor liver enzyme abnormalities (RR, 2.16; 95% CI, 1.67-2.79), but did not increase the risk of liver failure, cirrhosis, or liver-related mortality (RR, 0.12; 95% CI, 0.01-1.09), the investigators reported. “Clinical trials have inherent limitations, including short duration, selection bias, failure to collect information or reports that are not part of a prespecified protocol, and limited power to evaluate rare or unusual reports,” they cautioned. “While meta-analyses help overcome the last of these, the others raise some concern.”
But several other reports align with their results, the researchers noted. In an analysis of methotrexate studies lasting at least 2 years, about 10% of patients developed at least one minor liver enzyme abnormality, but less than 4% stopped methotrexate because of liver toxicity. Other controlled studies did not link methotrexate to severe fibrosis or cirrhosis – even after 4 years of treatment, they added. “It appears likely that serious liver-related complications due to methotrexate may take many years of treatment to manifest,” the investigators concluded.
The researchers reported no funding sources or conflicts of interest.
Methotrexate use more than doubled the risk of liver enzyme abnormalities but was not tied to serious hepatic outcomes among patients with rheumatic diseases, according to a meta-analysis of 32 randomized, controlled trials.
“Serious liver events were rare, with a suggestion of a trend towards fewer poor liver outcomes,” wrote Dr. Richard Conway of Galway (Ireland) University Hospitals. “The short duration of the included studies and the lack of liver biopsies are limitations, but the available data provide robust evidence for short-term safety” of methotrexate, wrote Dr. Conway and his associates.
Methotrexate is a first-line RA therapy with efficacy in several other inflammatory diseases, but concerns about toxicity have limited its use, the researchers noted. Clinicians have particularly debated its hepatic effects, in part because it can be “exceedingly difficult” to determine if liver disease is preexisting, methotrexate related, or caused by other medications or comorbidities, they said. To better assess the issue, they searched PubMed and the Cochrane Central Register of Controlled Trials for relevant double-blind, randomized, controlled trials published between 1990 and 2014. They found 32 such trials that included 13,177 patients with rheumatoid arthritis, psoriasis, psoriatic arthritis or inflammatory bowel disease (Semin Arthritis Rheum. 2015;45:156-62). Methotrexate use approximately doubled the risk of any adverse hepatic event (relative risk, 2.19; 95% confidence interval, 1.73-2.77) and of major and minor liver enzyme abnormalities (RR, 2.16; 95% CI, 1.67-2.79), but did not increase the risk of liver failure, cirrhosis, or liver-related mortality (RR, 0.12; 95% CI, 0.01-1.09), the investigators reported. “Clinical trials have inherent limitations, including short duration, selection bias, failure to collect information or reports that are not part of a prespecified protocol, and limited power to evaluate rare or unusual reports,” they cautioned. “While meta-analyses help overcome the last of these, the others raise some concern.”
But several other reports align with their results, the researchers noted. In an analysis of methotrexate studies lasting at least 2 years, about 10% of patients developed at least one minor liver enzyme abnormality, but less than 4% stopped methotrexate because of liver toxicity. Other controlled studies did not link methotrexate to severe fibrosis or cirrhosis – even after 4 years of treatment, they added. “It appears likely that serious liver-related complications due to methotrexate may take many years of treatment to manifest,” the investigators concluded.
The researchers reported no funding sources or conflicts of interest.
Methotrexate use more than doubled the risk of liver enzyme abnormalities but was not tied to serious hepatic outcomes among patients with rheumatic diseases, according to a meta-analysis of 32 randomized, controlled trials.
“Serious liver events were rare, with a suggestion of a trend towards fewer poor liver outcomes,” wrote Dr. Richard Conway of Galway (Ireland) University Hospitals. “The short duration of the included studies and the lack of liver biopsies are limitations, but the available data provide robust evidence for short-term safety” of methotrexate, wrote Dr. Conway and his associates.
Methotrexate is a first-line RA therapy with efficacy in several other inflammatory diseases, but concerns about toxicity have limited its use, the researchers noted. Clinicians have particularly debated its hepatic effects, in part because it can be “exceedingly difficult” to determine if liver disease is preexisting, methotrexate related, or caused by other medications or comorbidities, they said. To better assess the issue, they searched PubMed and the Cochrane Central Register of Controlled Trials for relevant double-blind, randomized, controlled trials published between 1990 and 2014. They found 32 such trials that included 13,177 patients with rheumatoid arthritis, psoriasis, psoriatic arthritis or inflammatory bowel disease (Semin Arthritis Rheum. 2015;45:156-62). Methotrexate use approximately doubled the risk of any adverse hepatic event (relative risk, 2.19; 95% confidence interval, 1.73-2.77) and of major and minor liver enzyme abnormalities (RR, 2.16; 95% CI, 1.67-2.79), but did not increase the risk of liver failure, cirrhosis, or liver-related mortality (RR, 0.12; 95% CI, 0.01-1.09), the investigators reported. “Clinical trials have inherent limitations, including short duration, selection bias, failure to collect information or reports that are not part of a prespecified protocol, and limited power to evaluate rare or unusual reports,” they cautioned. “While meta-analyses help overcome the last of these, the others raise some concern.”
But several other reports align with their results, the researchers noted. In an analysis of methotrexate studies lasting at least 2 years, about 10% of patients developed at least one minor liver enzyme abnormality, but less than 4% stopped methotrexate because of liver toxicity. Other controlled studies did not link methotrexate to severe fibrosis or cirrhosis – even after 4 years of treatment, they added. “It appears likely that serious liver-related complications due to methotrexate may take many years of treatment to manifest,” the investigators concluded.
The researchers reported no funding sources or conflicts of interest.
FROM SEMINARS IN ARTHRITIS AND RHEUMATISM
Key clinical point: Use of methotrexate increased the risk of hepatic enzyme abnormalities but not serious liver-related events among patients with inflammatory diseases.
Major finding: Methotrexate use approximately doubled the risk of any adverse liver event (RR, 2.19) but did not increase the risk of cirrhosis, liver failure, or liver-related death.
Data source: A meta-analysis of 32 randomized, controlled trials of methotrexate in rheumatoid arthritis, psoriasis, psoriatic arthritis, and inflammatory bowel disease.
Disclosures: The investigators reported no funding sources or conflicts of interest.
WCD: Look for TNF inhibitor–induced psoriasis in kids
VANCOUVER – Tumor necrosis factor inhibitor–induced psoriasiform dermatitis can occur in pediatric patients after any length of treatment, with documented cases emerging after the very first dose and as late as 63 months into anti-TNF therapy, Dr. Amy S. Paller said at the World Congress of Dermatology.
Histopathologically, this medication-induced condition is psoriasis. But it tends to follow a distinctive pattern, favoring the scalp, dorsal hands and feet, nails, and periorificial skin. Palmoplantar pustulosis is not uncommon. The lesions are often secondarily infected with Staphylococcus aureus, according to Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University in Chicago.
The phenomenon was first described in adults. But in the past several years, as the use of tumor necrosis factor (TNF) antagonists has gained increasing traction for treatment of pediatric inflammatory bowel disease and rheumatologic diseases, the dermatologic disorder has become better characterized in youths. In a retrospective study at McMaster Children’s Hospital in Hamilton, Ont., 17 of 172 (10%) infliximab-treated patients with Crohn’s disease developed new-onset psoriasis and another (0.6%) experienced worsening of preexisting psoriasis after anywhere from 1 to 25 infusions. Most patients responded well to topical steroids; however, three discontinued the biologic because of this complication (J Pediatr Gastroenterol Nutr. 2013 May;56[5]:512-8).
It’s now clear that the emergence of TNF inhibitor–induced psoriasis does not adversely affect the response of a patient’s inflammatory bowel disease or juvenile arthritis to the biologic. Also, the risk of recurrent psoriatic eruption is not reduced by concurrent methotrexate.
Researchers find TNF inhibitor–induced psoriasis to be an intriguing puzzle because of its paradoxical nature. After all, the TNF inhibitors are a highly effective treatment for moderate to severe plaque psoriasis. The leading theory as to the underlying basis for TNF inhibitor–induced psoriasis is that it may have a genetic basis, Dr. Paller noted.
The McMaster group found that their pediatric Crohn’s disease patients who developed psoriasis in conjunction with infliximab (Remicade) therapy were more likely than disease-matched controls to be homozygous for one of several specific polymorphisms in the interleukin-23R gene. And investigators at the University of Helsinki have reported that children with inflammatory bowel disease who developed psoriasiform dermatitis while on infliximab only rarely possessed the HLA-Cw*0602 genotype, which is commonly associated with psoriasis (Inflamm Bowel Dis. 2014 Aug;20[8]:1309-15).
The possibility of streptococcal or staphylococcal infection serving as a trigger for TNF inhibitor–induced psoriasis is also being explored, according to Dr. Paller.
She has received research funding from LEO Pharma and Amgen and serves as a consultant to AbbVie.
VANCOUVER – Tumor necrosis factor inhibitor–induced psoriasiform dermatitis can occur in pediatric patients after any length of treatment, with documented cases emerging after the very first dose and as late as 63 months into anti-TNF therapy, Dr. Amy S. Paller said at the World Congress of Dermatology.
Histopathologically, this medication-induced condition is psoriasis. But it tends to follow a distinctive pattern, favoring the scalp, dorsal hands and feet, nails, and periorificial skin. Palmoplantar pustulosis is not uncommon. The lesions are often secondarily infected with Staphylococcus aureus, according to Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University in Chicago.
The phenomenon was first described in adults. But in the past several years, as the use of tumor necrosis factor (TNF) antagonists has gained increasing traction for treatment of pediatric inflammatory bowel disease and rheumatologic diseases, the dermatologic disorder has become better characterized in youths. In a retrospective study at McMaster Children’s Hospital in Hamilton, Ont., 17 of 172 (10%) infliximab-treated patients with Crohn’s disease developed new-onset psoriasis and another (0.6%) experienced worsening of preexisting psoriasis after anywhere from 1 to 25 infusions. Most patients responded well to topical steroids; however, three discontinued the biologic because of this complication (J Pediatr Gastroenterol Nutr. 2013 May;56[5]:512-8).
It’s now clear that the emergence of TNF inhibitor–induced psoriasis does not adversely affect the response of a patient’s inflammatory bowel disease or juvenile arthritis to the biologic. Also, the risk of recurrent psoriatic eruption is not reduced by concurrent methotrexate.
Researchers find TNF inhibitor–induced psoriasis to be an intriguing puzzle because of its paradoxical nature. After all, the TNF inhibitors are a highly effective treatment for moderate to severe plaque psoriasis. The leading theory as to the underlying basis for TNF inhibitor–induced psoriasis is that it may have a genetic basis, Dr. Paller noted.
The McMaster group found that their pediatric Crohn’s disease patients who developed psoriasis in conjunction with infliximab (Remicade) therapy were more likely than disease-matched controls to be homozygous for one of several specific polymorphisms in the interleukin-23R gene. And investigators at the University of Helsinki have reported that children with inflammatory bowel disease who developed psoriasiform dermatitis while on infliximab only rarely possessed the HLA-Cw*0602 genotype, which is commonly associated with psoriasis (Inflamm Bowel Dis. 2014 Aug;20[8]:1309-15).
The possibility of streptococcal or staphylococcal infection serving as a trigger for TNF inhibitor–induced psoriasis is also being explored, according to Dr. Paller.
She has received research funding from LEO Pharma and Amgen and serves as a consultant to AbbVie.
VANCOUVER – Tumor necrosis factor inhibitor–induced psoriasiform dermatitis can occur in pediatric patients after any length of treatment, with documented cases emerging after the very first dose and as late as 63 months into anti-TNF therapy, Dr. Amy S. Paller said at the World Congress of Dermatology.
Histopathologically, this medication-induced condition is psoriasis. But it tends to follow a distinctive pattern, favoring the scalp, dorsal hands and feet, nails, and periorificial skin. Palmoplantar pustulosis is not uncommon. The lesions are often secondarily infected with Staphylococcus aureus, according to Dr. Paller, professor and chair of the department of dermatology and professor of pediatrics at Northwestern University in Chicago.
The phenomenon was first described in adults. But in the past several years, as the use of tumor necrosis factor (TNF) antagonists has gained increasing traction for treatment of pediatric inflammatory bowel disease and rheumatologic diseases, the dermatologic disorder has become better characterized in youths. In a retrospective study at McMaster Children’s Hospital in Hamilton, Ont., 17 of 172 (10%) infliximab-treated patients with Crohn’s disease developed new-onset psoriasis and another (0.6%) experienced worsening of preexisting psoriasis after anywhere from 1 to 25 infusions. Most patients responded well to topical steroids; however, three discontinued the biologic because of this complication (J Pediatr Gastroenterol Nutr. 2013 May;56[5]:512-8).
It’s now clear that the emergence of TNF inhibitor–induced psoriasis does not adversely affect the response of a patient’s inflammatory bowel disease or juvenile arthritis to the biologic. Also, the risk of recurrent psoriatic eruption is not reduced by concurrent methotrexate.
Researchers find TNF inhibitor–induced psoriasis to be an intriguing puzzle because of its paradoxical nature. After all, the TNF inhibitors are a highly effective treatment for moderate to severe plaque psoriasis. The leading theory as to the underlying basis for TNF inhibitor–induced psoriasis is that it may have a genetic basis, Dr. Paller noted.
The McMaster group found that their pediatric Crohn’s disease patients who developed psoriasis in conjunction with infliximab (Remicade) therapy were more likely than disease-matched controls to be homozygous for one of several specific polymorphisms in the interleukin-23R gene. And investigators at the University of Helsinki have reported that children with inflammatory bowel disease who developed psoriasiform dermatitis while on infliximab only rarely possessed the HLA-Cw*0602 genotype, which is commonly associated with psoriasis (Inflamm Bowel Dis. 2014 Aug;20[8]:1309-15).
The possibility of streptococcal or staphylococcal infection serving as a trigger for TNF inhibitor–induced psoriasis is also being explored, according to Dr. Paller.
She has received research funding from LEO Pharma and Amgen and serves as a consultant to AbbVie.
EXPERT ANALYSIS FROM WCD 2015
Nail psoriasis therapies lack supporting evidence
VANCOUVER – Evidence-based therapy for nail psoriasis is in a sorry state because of a lack of consensus on a reliable nail psoriasis scoring system for use in clinical trials, according to a coauthor of the Cochrane systematic review of interventions for nail psoriasis.
“The last 12 randomized clinical trials used 21 ways of scoring the results of treatment, so comparing the studies means comparing apples to oranges. Which is the most effective treatment? What should we advise our patients? We don’t know. Comparison is impossible,” Dr. Marcel C. Pasch said at the World Congress of Dermatology.
The Cochrane report (Cochrane Database Syst Rev. 2013 Jan 31;1:CD007633) deemed the evidence for topical therapies as “inconclusive and weak,” even though topicals are the treatment mainstay for this localized expression of psoriasis. Indeed, Dr. Pasch and his coauthors found that no topical therapy has been shown effective in improving nail psoriasis. The Cochrane group concluded that just five therapies rise to the standard of being evidence based in terms of efficacy: the tumor necrosis factor (TNF) inhibitors infliximab (Remicade) and golimumab (Simponi), superficial radiation therapy, Grenz rays, and electron beam therapy. All five are strikingly impractical for use in clinical practice.
“The findings are quite disappointing because nobody sends a patient with psoriasis to the radiotherapist, and while giving an anti-TNF biologic only for the nails will be effective, at least in my country it won’t be reimbursed,” wrote Dr. Pasch, a dermatologist at Radboud University Nijmegen (the Netherlands) Medical Centre.
The presence and severity of nail psoriasis is unrelated to the severity of cutaneous psoriasis. Moreover, nail psoriasis without cutaneous involvement occurs in 5%-10% of psoriasis patients.
Since publication of the Cochrane systematic review, 12 new randomized controlled trials of treatments for nail psoriasis have appeared. Six focused on biologics: the anti-TNF agents certolizumab (Cimzia), etanercept (Enbrel), and adalimumab (Humira); the anti–interleukin-12/23 agent ustekinumab (Stelara); and the interleukin-17A inhibitor secukiumab (Cosentyx). Dr. Pasch said in his opinion all five biologics were supported by convincing studies and now can be added to the short list of evidence-based nail psoriasis therapies.
Of the six recent studies of topical therapies, two provided persuasive evidence of efficacy, in his view: tacrolimus ointment and indigo naturalis extract in oil (Lindioil), a variant of a traditional Chinese medicine therapy, which at this time isn’t commercially available.
In contrast, studies of clobetasol nail lacquer, pulsed dye laser therapy, a nail lacquer based upon chitin from crab shells, and a study of calcitriol ointment versus betamethasone dipropionate ointment failed to be convincing either because of methodologic problems or lack of efficacy, he continued.
These 12 recent randomized clinical trials utilized 21 different nail psoriasis scoring systems.
“Which scoring system is best? The answer is, we don’t know,” Dr. Pasch said.
He and his coinvestigators compared eight different scoring systems in a prospective study and concluded that the Nijmegen–Nail Psoriasis Activity Index Tool (N-NAIL), which Dr. Pasch helped develop, best reflected the clinical severity of nail psoriasis (J Am Acad Dermatol. 2014 Jun;70[6]:1061-6).
However, he added that at present there is no validated scoring system for nail psoriasis. And creation of a single validated scoring system that researchers can agree on as the standard is a prerequisite for making major advances in the treatment of nail psoriasis, in Dr. Pasch’s view.
He is so convinced of this that he has created an organization whose goal is to achieve consensus on one reliable, validated nail psoriasis scoring system for use in clinical trials. At the World Congress of Dermatology, he invited stakeholders – including academic and community dermatologists, patient organizations, and the pharmaceutical industry – to join (www.nailinitiative.org).
Session chair Dr. Peter van de Kerkhof, chairman of dermatology at Radboud University, said he sees the NAPSI (Nail Psoriasis Severity Index) being used in lots of clinical trials in psoriasis. What’s wrong with building a consensus around NAPSI? he asked.
“The problem is not the NAPSI score,” Dr. Pasch replied. “The problem is that in each trial a modified NAPSI score is used, but they are all modified in different ways. We have the single-hand NAPSI, the eight-finger NAPSI, the 10-finger NAPSI, the target NAPSI. The NAPSI doesn’t exist anymore.”
He reported receiving research grants from Pfizer and Janssen-Cilag.
VANCOUVER – Evidence-based therapy for nail psoriasis is in a sorry state because of a lack of consensus on a reliable nail psoriasis scoring system for use in clinical trials, according to a coauthor of the Cochrane systematic review of interventions for nail psoriasis.
“The last 12 randomized clinical trials used 21 ways of scoring the results of treatment, so comparing the studies means comparing apples to oranges. Which is the most effective treatment? What should we advise our patients? We don’t know. Comparison is impossible,” Dr. Marcel C. Pasch said at the World Congress of Dermatology.
The Cochrane report (Cochrane Database Syst Rev. 2013 Jan 31;1:CD007633) deemed the evidence for topical therapies as “inconclusive and weak,” even though topicals are the treatment mainstay for this localized expression of psoriasis. Indeed, Dr. Pasch and his coauthors found that no topical therapy has been shown effective in improving nail psoriasis. The Cochrane group concluded that just five therapies rise to the standard of being evidence based in terms of efficacy: the tumor necrosis factor (TNF) inhibitors infliximab (Remicade) and golimumab (Simponi), superficial radiation therapy, Grenz rays, and electron beam therapy. All five are strikingly impractical for use in clinical practice.
“The findings are quite disappointing because nobody sends a patient with psoriasis to the radiotherapist, and while giving an anti-TNF biologic only for the nails will be effective, at least in my country it won’t be reimbursed,” wrote Dr. Pasch, a dermatologist at Radboud University Nijmegen (the Netherlands) Medical Centre.
The presence and severity of nail psoriasis is unrelated to the severity of cutaneous psoriasis. Moreover, nail psoriasis without cutaneous involvement occurs in 5%-10% of psoriasis patients.
Since publication of the Cochrane systematic review, 12 new randomized controlled trials of treatments for nail psoriasis have appeared. Six focused on biologics: the anti-TNF agents certolizumab (Cimzia), etanercept (Enbrel), and adalimumab (Humira); the anti–interleukin-12/23 agent ustekinumab (Stelara); and the interleukin-17A inhibitor secukiumab (Cosentyx). Dr. Pasch said in his opinion all five biologics were supported by convincing studies and now can be added to the short list of evidence-based nail psoriasis therapies.
Of the six recent studies of topical therapies, two provided persuasive evidence of efficacy, in his view: tacrolimus ointment and indigo naturalis extract in oil (Lindioil), a variant of a traditional Chinese medicine therapy, which at this time isn’t commercially available.
In contrast, studies of clobetasol nail lacquer, pulsed dye laser therapy, a nail lacquer based upon chitin from crab shells, and a study of calcitriol ointment versus betamethasone dipropionate ointment failed to be convincing either because of methodologic problems or lack of efficacy, he continued.
These 12 recent randomized clinical trials utilized 21 different nail psoriasis scoring systems.
“Which scoring system is best? The answer is, we don’t know,” Dr. Pasch said.
He and his coinvestigators compared eight different scoring systems in a prospective study and concluded that the Nijmegen–Nail Psoriasis Activity Index Tool (N-NAIL), which Dr. Pasch helped develop, best reflected the clinical severity of nail psoriasis (J Am Acad Dermatol. 2014 Jun;70[6]:1061-6).
However, he added that at present there is no validated scoring system for nail psoriasis. And creation of a single validated scoring system that researchers can agree on as the standard is a prerequisite for making major advances in the treatment of nail psoriasis, in Dr. Pasch’s view.
He is so convinced of this that he has created an organization whose goal is to achieve consensus on one reliable, validated nail psoriasis scoring system for use in clinical trials. At the World Congress of Dermatology, he invited stakeholders – including academic and community dermatologists, patient organizations, and the pharmaceutical industry – to join (www.nailinitiative.org).
Session chair Dr. Peter van de Kerkhof, chairman of dermatology at Radboud University, said he sees the NAPSI (Nail Psoriasis Severity Index) being used in lots of clinical trials in psoriasis. What’s wrong with building a consensus around NAPSI? he asked.
“The problem is not the NAPSI score,” Dr. Pasch replied. “The problem is that in each trial a modified NAPSI score is used, but they are all modified in different ways. We have the single-hand NAPSI, the eight-finger NAPSI, the 10-finger NAPSI, the target NAPSI. The NAPSI doesn’t exist anymore.”
He reported receiving research grants from Pfizer and Janssen-Cilag.
VANCOUVER – Evidence-based therapy for nail psoriasis is in a sorry state because of a lack of consensus on a reliable nail psoriasis scoring system for use in clinical trials, according to a coauthor of the Cochrane systematic review of interventions for nail psoriasis.
“The last 12 randomized clinical trials used 21 ways of scoring the results of treatment, so comparing the studies means comparing apples to oranges. Which is the most effective treatment? What should we advise our patients? We don’t know. Comparison is impossible,” Dr. Marcel C. Pasch said at the World Congress of Dermatology.
The Cochrane report (Cochrane Database Syst Rev. 2013 Jan 31;1:CD007633) deemed the evidence for topical therapies as “inconclusive and weak,” even though topicals are the treatment mainstay for this localized expression of psoriasis. Indeed, Dr. Pasch and his coauthors found that no topical therapy has been shown effective in improving nail psoriasis. The Cochrane group concluded that just five therapies rise to the standard of being evidence based in terms of efficacy: the tumor necrosis factor (TNF) inhibitors infliximab (Remicade) and golimumab (Simponi), superficial radiation therapy, Grenz rays, and electron beam therapy. All five are strikingly impractical for use in clinical practice.
“The findings are quite disappointing because nobody sends a patient with psoriasis to the radiotherapist, and while giving an anti-TNF biologic only for the nails will be effective, at least in my country it won’t be reimbursed,” wrote Dr. Pasch, a dermatologist at Radboud University Nijmegen (the Netherlands) Medical Centre.
The presence and severity of nail psoriasis is unrelated to the severity of cutaneous psoriasis. Moreover, nail psoriasis without cutaneous involvement occurs in 5%-10% of psoriasis patients.
Since publication of the Cochrane systematic review, 12 new randomized controlled trials of treatments for nail psoriasis have appeared. Six focused on biologics: the anti-TNF agents certolizumab (Cimzia), etanercept (Enbrel), and adalimumab (Humira); the anti–interleukin-12/23 agent ustekinumab (Stelara); and the interleukin-17A inhibitor secukiumab (Cosentyx). Dr. Pasch said in his opinion all five biologics were supported by convincing studies and now can be added to the short list of evidence-based nail psoriasis therapies.
Of the six recent studies of topical therapies, two provided persuasive evidence of efficacy, in his view: tacrolimus ointment and indigo naturalis extract in oil (Lindioil), a variant of a traditional Chinese medicine therapy, which at this time isn’t commercially available.
In contrast, studies of clobetasol nail lacquer, pulsed dye laser therapy, a nail lacquer based upon chitin from crab shells, and a study of calcitriol ointment versus betamethasone dipropionate ointment failed to be convincing either because of methodologic problems or lack of efficacy, he continued.
These 12 recent randomized clinical trials utilized 21 different nail psoriasis scoring systems.
“Which scoring system is best? The answer is, we don’t know,” Dr. Pasch said.
He and his coinvestigators compared eight different scoring systems in a prospective study and concluded that the Nijmegen–Nail Psoriasis Activity Index Tool (N-NAIL), which Dr. Pasch helped develop, best reflected the clinical severity of nail psoriasis (J Am Acad Dermatol. 2014 Jun;70[6]:1061-6).
However, he added that at present there is no validated scoring system for nail psoriasis. And creation of a single validated scoring system that researchers can agree on as the standard is a prerequisite for making major advances in the treatment of nail psoriasis, in Dr. Pasch’s view.
He is so convinced of this that he has created an organization whose goal is to achieve consensus on one reliable, validated nail psoriasis scoring system for use in clinical trials. At the World Congress of Dermatology, he invited stakeholders – including academic and community dermatologists, patient organizations, and the pharmaceutical industry – to join (www.nailinitiative.org).
Session chair Dr. Peter van de Kerkhof, chairman of dermatology at Radboud University, said he sees the NAPSI (Nail Psoriasis Severity Index) being used in lots of clinical trials in psoriasis. What’s wrong with building a consensus around NAPSI? he asked.
“The problem is not the NAPSI score,” Dr. Pasch replied. “The problem is that in each trial a modified NAPSI score is used, but they are all modified in different ways. We have the single-hand NAPSI, the eight-finger NAPSI, the 10-finger NAPSI, the target NAPSI. The NAPSI doesn’t exist anymore.”
He reported receiving research grants from Pfizer and Janssen-Cilag.
EXPERT ANALYSIS FROM WCD 2015
Psoriasis patients more likely to have type D personalities
Incidence of type D personality was significantly more common in patients with moderate to severe psoriasis, compared with a healthy control group, according to Dr. Alejandro Molina-Leyva of Hospital Torrecardenas, Almeria, Spain, and his associates.
People with type D, or distressed, personality tend to be more worried and irritable, and tend to display more negative emotions than do others. Of the 90 patients with moderate to severe psoriasis included in the study, 39% had type D personality, compared with 24% of the 82 members of the control group. The odds ratio for psoriasis patients developing type D personality was 2.1.
Psoriasis patients with type D personalities had significantly worse general, sexual, and psoriasis-related health-related quality of life, compared with psoriasis patients without type D personality. In addition, type D personality psoriasis patients were much more likely to experience anxiety or depression than were healthy people with type D personality, with an OR of 3.2.
“It may be that the higher prevalence of type D personality in moderate to severe psoriasis is, at least in part, the result of accumulated psychic damage over years of evolution of the disease. It is important to conduct prospective studies with incident cases of psoriasis to clarify the relationship between type D personality and psoriasis,” the investigators noted.
Find the full study here in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.12960).
Incidence of type D personality was significantly more common in patients with moderate to severe psoriasis, compared with a healthy control group, according to Dr. Alejandro Molina-Leyva of Hospital Torrecardenas, Almeria, Spain, and his associates.
People with type D, or distressed, personality tend to be more worried and irritable, and tend to display more negative emotions than do others. Of the 90 patients with moderate to severe psoriasis included in the study, 39% had type D personality, compared with 24% of the 82 members of the control group. The odds ratio for psoriasis patients developing type D personality was 2.1.
Psoriasis patients with type D personalities had significantly worse general, sexual, and psoriasis-related health-related quality of life, compared with psoriasis patients without type D personality. In addition, type D personality psoriasis patients were much more likely to experience anxiety or depression than were healthy people with type D personality, with an OR of 3.2.
“It may be that the higher prevalence of type D personality in moderate to severe psoriasis is, at least in part, the result of accumulated psychic damage over years of evolution of the disease. It is important to conduct prospective studies with incident cases of psoriasis to clarify the relationship between type D personality and psoriasis,” the investigators noted.
Find the full study here in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.12960).
Incidence of type D personality was significantly more common in patients with moderate to severe psoriasis, compared with a healthy control group, according to Dr. Alejandro Molina-Leyva of Hospital Torrecardenas, Almeria, Spain, and his associates.
People with type D, or distressed, personality tend to be more worried and irritable, and tend to display more negative emotions than do others. Of the 90 patients with moderate to severe psoriasis included in the study, 39% had type D personality, compared with 24% of the 82 members of the control group. The odds ratio for psoriasis patients developing type D personality was 2.1.
Psoriasis patients with type D personalities had significantly worse general, sexual, and psoriasis-related health-related quality of life, compared with psoriasis patients without type D personality. In addition, type D personality psoriasis patients were much more likely to experience anxiety or depression than were healthy people with type D personality, with an OR of 3.2.
“It may be that the higher prevalence of type D personality in moderate to severe psoriasis is, at least in part, the result of accumulated psychic damage over years of evolution of the disease. It is important to conduct prospective studies with incident cases of psoriasis to clarify the relationship between type D personality and psoriasis,” the investigators noted.
Find the full study here in the Journal of the European Academy of Dermatology and Venereology (doi: 10.1111/jdv.12960).
FROM THE JOURNAL OF THE EUROPEAN ACADEMY OF DERMATOLOGY AND VENEREOLOGY