User login
Can bariatric surgery ease psoriasis too?
LOS ANGELES – A provocative study opens the door for psoriasis and psoriatic arthritis to be added to the growing list of benefits following bariatric surgery.
“Although the natural history of psoriasis and psoriatic arthritis is typically chronic, the majority of patients experience improvement after bariatric surgery, Dr. Monica Sethi of New York University said at Obesity Week 2015.
At an average of 6 years after bariatric surgery, 55% of patients with psoriasis and 62% of those with psoriatic arthritis (PsA) reported subjective lessening of their disease.
Preoperative disease severity ratings on a 10-point scale significantly decreased at the most recent follow-up for psoriasis (5.6 vs. 3.3; P less than .01) and PsA (6.4 vs. 3.9; P = .02).
“Our results indicate an association between excess weight loss and symptomatic improvement in severe cases of psoriasis, and a possible improvement in psoriatic arthritis,” she said.
Although the effects of surgical weight loss on psoriasis and PsA are unknown, obesity is known to be more prevalent among patients with psoriasis and PsA. This correlation appears to be related to fat tissue–driven systemic inflammation, Dr. Sethi said.
The investigators surveyed 128 patients with a preoperative diagnosis of psoriasis identified from a single-center database of 9,073 bariatric surgeries performed between 2002 and 2013. A total of 86 patients completed the study, with 21 patients also having a preoperative diagnosis of PsA. Their mean preoperative weight was 288 pounds and preoperative body mass index 45.8 kg/m2. The average duration of psoriasis and PsA at the time of surgery was significant at 18.7 years, Dr. Sethi noted.
The mean time from surgery was 6.1 years, with a mean excess weight loss of 46.2% and total weight loss of 23.8%. Laparoscopic adjustable gastric banding was the most common surgery (91%), followed by Roux-en-Y bypass (7%).
In secondary analyses, a higher percent of excess weight loss at recent follow-up was significantly associated with an easing of psoriasis severity (59.5% vs. 43.5%; P = .046), while higher percent of excess weight loss at 1 year was associated with a trend in PsA improvement (55.4% vs. 43.8%; P = .47), Dr. Sethi said.
Easing of disease severity after surgery was associated with a higher rating of disease at the time of surgery (8.9 vs. 7.4; P less than .01) and older age at diagnosis (36.9 years vs. 25.9 years; P = .02), suggesting that these factors may be used to identify patients with a greater likelihood of improvement.
“Larger prospective studies are needed to further define the true effect of surgical weight loss on psoriasis and psoriatic arthritis,” she said at the meeting, presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery (ASMBS).
Session comoderator Dr. Peter T. Hallowell of the University of Virginia, Charlottesville, agreed that further studies are needed, but said the interesting thing is how bariatric surgery improves multiple problems.
“The results are pretty impressive with the moderate weight loss they got with laparoscopic gastric banding,” he said in an interview. “With gastric bypass or a sleeve, where we would expect greater weight loss and greater decrease in the inflammatory mediators, we may see an even greater improved outcome.”
A larger prospective study is being planned between New York University’s psoriasis and bariatric centers in about 60 patients with psoriasis and PsA undergoing bariatric surgery, mostly sleeve gastrectomy and gastric bypass, Dr. Sethi said in an interview.
LOS ANGELES – A provocative study opens the door for psoriasis and psoriatic arthritis to be added to the growing list of benefits following bariatric surgery.
“Although the natural history of psoriasis and psoriatic arthritis is typically chronic, the majority of patients experience improvement after bariatric surgery, Dr. Monica Sethi of New York University said at Obesity Week 2015.
At an average of 6 years after bariatric surgery, 55% of patients with psoriasis and 62% of those with psoriatic arthritis (PsA) reported subjective lessening of their disease.
Preoperative disease severity ratings on a 10-point scale significantly decreased at the most recent follow-up for psoriasis (5.6 vs. 3.3; P less than .01) and PsA (6.4 vs. 3.9; P = .02).
“Our results indicate an association between excess weight loss and symptomatic improvement in severe cases of psoriasis, and a possible improvement in psoriatic arthritis,” she said.
Although the effects of surgical weight loss on psoriasis and PsA are unknown, obesity is known to be more prevalent among patients with psoriasis and PsA. This correlation appears to be related to fat tissue–driven systemic inflammation, Dr. Sethi said.
The investigators surveyed 128 patients with a preoperative diagnosis of psoriasis identified from a single-center database of 9,073 bariatric surgeries performed between 2002 and 2013. A total of 86 patients completed the study, with 21 patients also having a preoperative diagnosis of PsA. Their mean preoperative weight was 288 pounds and preoperative body mass index 45.8 kg/m2. The average duration of psoriasis and PsA at the time of surgery was significant at 18.7 years, Dr. Sethi noted.
The mean time from surgery was 6.1 years, with a mean excess weight loss of 46.2% and total weight loss of 23.8%. Laparoscopic adjustable gastric banding was the most common surgery (91%), followed by Roux-en-Y bypass (7%).
In secondary analyses, a higher percent of excess weight loss at recent follow-up was significantly associated with an easing of psoriasis severity (59.5% vs. 43.5%; P = .046), while higher percent of excess weight loss at 1 year was associated with a trend in PsA improvement (55.4% vs. 43.8%; P = .47), Dr. Sethi said.
Easing of disease severity after surgery was associated with a higher rating of disease at the time of surgery (8.9 vs. 7.4; P less than .01) and older age at diagnosis (36.9 years vs. 25.9 years; P = .02), suggesting that these factors may be used to identify patients with a greater likelihood of improvement.
“Larger prospective studies are needed to further define the true effect of surgical weight loss on psoriasis and psoriatic arthritis,” she said at the meeting, presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery (ASMBS).
Session comoderator Dr. Peter T. Hallowell of the University of Virginia, Charlottesville, agreed that further studies are needed, but said the interesting thing is how bariatric surgery improves multiple problems.
“The results are pretty impressive with the moderate weight loss they got with laparoscopic gastric banding,” he said in an interview. “With gastric bypass or a sleeve, where we would expect greater weight loss and greater decrease in the inflammatory mediators, we may see an even greater improved outcome.”
A larger prospective study is being planned between New York University’s psoriasis and bariatric centers in about 60 patients with psoriasis and PsA undergoing bariatric surgery, mostly sleeve gastrectomy and gastric bypass, Dr. Sethi said in an interview.
LOS ANGELES – A provocative study opens the door for psoriasis and psoriatic arthritis to be added to the growing list of benefits following bariatric surgery.
“Although the natural history of psoriasis and psoriatic arthritis is typically chronic, the majority of patients experience improvement after bariatric surgery, Dr. Monica Sethi of New York University said at Obesity Week 2015.
At an average of 6 years after bariatric surgery, 55% of patients with psoriasis and 62% of those with psoriatic arthritis (PsA) reported subjective lessening of their disease.
Preoperative disease severity ratings on a 10-point scale significantly decreased at the most recent follow-up for psoriasis (5.6 vs. 3.3; P less than .01) and PsA (6.4 vs. 3.9; P = .02).
“Our results indicate an association between excess weight loss and symptomatic improvement in severe cases of psoriasis, and a possible improvement in psoriatic arthritis,” she said.
Although the effects of surgical weight loss on psoriasis and PsA are unknown, obesity is known to be more prevalent among patients with psoriasis and PsA. This correlation appears to be related to fat tissue–driven systemic inflammation, Dr. Sethi said.
The investigators surveyed 128 patients with a preoperative diagnosis of psoriasis identified from a single-center database of 9,073 bariatric surgeries performed between 2002 and 2013. A total of 86 patients completed the study, with 21 patients also having a preoperative diagnosis of PsA. Their mean preoperative weight was 288 pounds and preoperative body mass index 45.8 kg/m2. The average duration of psoriasis and PsA at the time of surgery was significant at 18.7 years, Dr. Sethi noted.
The mean time from surgery was 6.1 years, with a mean excess weight loss of 46.2% and total weight loss of 23.8%. Laparoscopic adjustable gastric banding was the most common surgery (91%), followed by Roux-en-Y bypass (7%).
In secondary analyses, a higher percent of excess weight loss at recent follow-up was significantly associated with an easing of psoriasis severity (59.5% vs. 43.5%; P = .046), while higher percent of excess weight loss at 1 year was associated with a trend in PsA improvement (55.4% vs. 43.8%; P = .47), Dr. Sethi said.
Easing of disease severity after surgery was associated with a higher rating of disease at the time of surgery (8.9 vs. 7.4; P less than .01) and older age at diagnosis (36.9 years vs. 25.9 years; P = .02), suggesting that these factors may be used to identify patients with a greater likelihood of improvement.
“Larger prospective studies are needed to further define the true effect of surgical weight loss on psoriasis and psoriatic arthritis,” she said at the meeting, presented by the Obesity Society and the American Society for Metabolic and Bariatric Surgery (ASMBS).
Session comoderator Dr. Peter T. Hallowell of the University of Virginia, Charlottesville, agreed that further studies are needed, but said the interesting thing is how bariatric surgery improves multiple problems.
“The results are pretty impressive with the moderate weight loss they got with laparoscopic gastric banding,” he said in an interview. “With gastric bypass or a sleeve, where we would expect greater weight loss and greater decrease in the inflammatory mediators, we may see an even greater improved outcome.”
A larger prospective study is being planned between New York University’s psoriasis and bariatric centers in about 60 patients with psoriasis and PsA undergoing bariatric surgery, mostly sleeve gastrectomy and gastric bypass, Dr. Sethi said in an interview.
AT OBESITY WEEK 2015
Key clinical point: Bariatric surgery in obese patients may ease the symptoms of psoriasis and psoriatic arthritis.
Major finding: Preoperative disease severity ratings declined at most recent follow-up for psoriasis (5.6 vs. 3.3; P less than .01) and psoriatic arthritis (6.4 vs. 3.9; P = .02).
Data source: Survey of 86 bariatric surgery patients with preoperative psoriasis alone or in combination with psoriatic arthritis.
Disclosures: Dr. Sethi reported having no disclosures. Three coauthors reported financial ties with Allergan Medical, one of whom is also a speaker for Apollo Endosurgery and one of whom is on the faculty for Gore.
Depression and Psoriasis
While psoriasis is a known risk factor for depression, depression can also exacerbate or trigger psoriasis. This relationship between depression and psoriasis, however, remains to be fully explored.
In an article published online on September 30 in JAMA Dermatology , Cohen et al examined the association between psoriasis and major depression in the US population. The authors conducted a population-based study that utilized individuals who were participating in the National Health and Nutrition Examination Survey from 2009 through 2012.
The authors identified 351 (2.8%) cases of psoriasis and 968 (7.8%) cases of major depression in the 12,382 participants included in the study. Of the patients with psoriasis, 58 (16.5%) met criteria for major depression. The mean (standard deviation) Patient Health Questionnaire-9 score was significantly higher among patients with a history of psoriasis than those without psoriasis (4.54 [5.7] vs 3.22 [4.3], P<.001). After adjustment for sex, age, race, body mass index, physical activity level, smoking history, alcohol use, history of myocardial infarction, history of stroke, and history of diabetes mellitus (odds ratio, 2.09 [95% confidence interval, 1.41–3.11], P<.001), psoriasis was significantly associated with major depression. Having a history of cardiovascular events did not modify the risk of major depression for patients with psoriasis. The investigators also found that the risk of major depression was not significantly different between patients with limited vs extensive psoriasis (odds ratio, 0.66 [95% confidence interval, 0.18–2.44], P=.53).
What’s the Issue?
We know that psoriasis is associated with depression. This study, however, has some surprising findings. The severity of psoriasis was unrelated to the risk of major depression. Additionally, cardiovascular events did not seem to impact major depression in participants with psoriasis. Therefore, all patients with psoriasis, regardless of severity, may be at risk for major depression. Will these findings impact your evaluation of psychological issues in individuals with psoriasis?
While psoriasis is a known risk factor for depression, depression can also exacerbate or trigger psoriasis. This relationship between depression and psoriasis, however, remains to be fully explored.
In an article published online on September 30 in JAMA Dermatology , Cohen et al examined the association between psoriasis and major depression in the US population. The authors conducted a population-based study that utilized individuals who were participating in the National Health and Nutrition Examination Survey from 2009 through 2012.
The authors identified 351 (2.8%) cases of psoriasis and 968 (7.8%) cases of major depression in the 12,382 participants included in the study. Of the patients with psoriasis, 58 (16.5%) met criteria for major depression. The mean (standard deviation) Patient Health Questionnaire-9 score was significantly higher among patients with a history of psoriasis than those without psoriasis (4.54 [5.7] vs 3.22 [4.3], P<.001). After adjustment for sex, age, race, body mass index, physical activity level, smoking history, alcohol use, history of myocardial infarction, history of stroke, and history of diabetes mellitus (odds ratio, 2.09 [95% confidence interval, 1.41–3.11], P<.001), psoriasis was significantly associated with major depression. Having a history of cardiovascular events did not modify the risk of major depression for patients with psoriasis. The investigators also found that the risk of major depression was not significantly different between patients with limited vs extensive psoriasis (odds ratio, 0.66 [95% confidence interval, 0.18–2.44], P=.53).
What’s the Issue?
We know that psoriasis is associated with depression. This study, however, has some surprising findings. The severity of psoriasis was unrelated to the risk of major depression. Additionally, cardiovascular events did not seem to impact major depression in participants with psoriasis. Therefore, all patients with psoriasis, regardless of severity, may be at risk for major depression. Will these findings impact your evaluation of psychological issues in individuals with psoriasis?
While psoriasis is a known risk factor for depression, depression can also exacerbate or trigger psoriasis. This relationship between depression and psoriasis, however, remains to be fully explored.
In an article published online on September 30 in JAMA Dermatology , Cohen et al examined the association between psoriasis and major depression in the US population. The authors conducted a population-based study that utilized individuals who were participating in the National Health and Nutrition Examination Survey from 2009 through 2012.
The authors identified 351 (2.8%) cases of psoriasis and 968 (7.8%) cases of major depression in the 12,382 participants included in the study. Of the patients with psoriasis, 58 (16.5%) met criteria for major depression. The mean (standard deviation) Patient Health Questionnaire-9 score was significantly higher among patients with a history of psoriasis than those without psoriasis (4.54 [5.7] vs 3.22 [4.3], P<.001). After adjustment for sex, age, race, body mass index, physical activity level, smoking history, alcohol use, history of myocardial infarction, history of stroke, and history of diabetes mellitus (odds ratio, 2.09 [95% confidence interval, 1.41–3.11], P<.001), psoriasis was significantly associated with major depression. Having a history of cardiovascular events did not modify the risk of major depression for patients with psoriasis. The investigators also found that the risk of major depression was not significantly different between patients with limited vs extensive psoriasis (odds ratio, 0.66 [95% confidence interval, 0.18–2.44], P=.53).
What’s the Issue?
We know that psoriasis is associated with depression. This study, however, has some surprising findings. The severity of psoriasis was unrelated to the risk of major depression. Additionally, cardiovascular events did not seem to impact major depression in participants with psoriasis. Therefore, all patients with psoriasis, regardless of severity, may be at risk for major depression. Will these findings impact your evaluation of psychological issues in individuals with psoriasis?
EADV: No cancer signal with ixekizumab
COPENHAGEN – Ixekizumab showed no hint of an increased malignancy risk of any sort in 4,208 patients with moderate to severe psoriasis who had 6,480 patient-years of exposure to the investigational interleukin-17A inhibitor in seven clinical trials.
“Patients were studied for an average of 18 months on ixekizumab. You may say that’s not enough time to assess the true risk for malignancy, and I would say you’re probably right. But 18 months isn’t shabby. From such an exposure, we can get some sense of the malignancy risk with this drug,” Dr. Bruce E. Strober asserted at the annual congress of the European Academy of Dermatology and Venereology.
That’s a key issue whenever a dermatologist prescribes a potent immune-altering anti-inflammatory medication for an indefinite period of time, he added.
In the randomized trials in which more than 2,300 patients who received 80 mg of ixekizumab every 2 or 4 weeks were compared with 739 patients assigned to etanercept (Enbrel) and 791 on placebo, malignancy rates were similarly low, at 0.1-0.3 cases per 100 patient-years across all four treatment arms, reported Dr. Strober, chair of the department of dermatology at the University of Connecticut, Farmington.
Moreover, in the long-term maintenance studies featuring 60 months of follow-up, the risks of both nonmelanoma skin cancer and other types of cancer were similarly low in patients randomized to ixekizumab at 80 mg every 2 weeks and those dosed every 4 weeks. That’s reassuring, because if a drug causes cancer one would expect to see that the more of the drug given, the higher the malignancy rate.
“You really don’t see any kind of a pattern or any difference between the various compared groups, including patients on etanercept, a drug that all of us feel very comfortable using in patients with psoriasis,” the dermatologist observed.
It’s thought that psoriasis patients have a higher background risk of malignancy than the general population due to the nature of the chronic disease, which involves immune dysregulation and chronic inflammation. In the studies to date, however, the malignancy risk in ixekizumab-treated patients is similar to those on placebo or etanercept.
“In my opinion, it’s comforting that the malignancy rates of the currently approved biologic agents for psoriasis are low and – one could argue – not above that which you would expect for the population of patients we’re treating. So I’ve always felt that monotherapy – and that’s an important point, not combination therapy with other immunosuppressive drugs – offers very little risk of de novo malignancy with the currently approved biologics,” Dr. Strober said.
Lumping together the results of the seven studies in his analysis, the incidence of squamous cell carcinoma was 0.1 cases per 100 patient-years of ixekizumab exposure, the risk of basal cell carcinoma was 0.3 cases per 100 patient-years, and the risk of malignancies other than nonmelanoma skin cancer was 0.5 per 100 patient-years of exposure to ixekizumab.
“That’s basically in line with every other biologic that I’ve examined with regard to malignancy rates: about 1 in 200 patients treated for 1 year gets a nonmelanoma skin cancer of some nature if you look at all data sources, including long-term registries and clinical trials. So it appears that this drug falls right in line with the other biologics in terms of rates of the more serious cancers, at least over the time period studied,” Dr. Strober commented.
He offered a caveat, however: “What will happen in patients given combination therapy with other immunosuppressive drugs? Or special populations, such as patients at higher risk for malignancy, be it skin cancer or other types of cancer? Or – and here’s the question we often have the most difficulty answering – what about people with a prior history of malignancy? What would be their risk of malignancy in being placed on a drug of this nature? Only time and really good registry data will allow us to answer these questions.”
Dr. Strober is a consultant to Eli Lilly, which sponsored the seven ixekizumab studies, as well as to numerous other pharmaceutical companies.
COPENHAGEN – Ixekizumab showed no hint of an increased malignancy risk of any sort in 4,208 patients with moderate to severe psoriasis who had 6,480 patient-years of exposure to the investigational interleukin-17A inhibitor in seven clinical trials.
“Patients were studied for an average of 18 months on ixekizumab. You may say that’s not enough time to assess the true risk for malignancy, and I would say you’re probably right. But 18 months isn’t shabby. From such an exposure, we can get some sense of the malignancy risk with this drug,” Dr. Bruce E. Strober asserted at the annual congress of the European Academy of Dermatology and Venereology.
That’s a key issue whenever a dermatologist prescribes a potent immune-altering anti-inflammatory medication for an indefinite period of time, he added.
In the randomized trials in which more than 2,300 patients who received 80 mg of ixekizumab every 2 or 4 weeks were compared with 739 patients assigned to etanercept (Enbrel) and 791 on placebo, malignancy rates were similarly low, at 0.1-0.3 cases per 100 patient-years across all four treatment arms, reported Dr. Strober, chair of the department of dermatology at the University of Connecticut, Farmington.
Moreover, in the long-term maintenance studies featuring 60 months of follow-up, the risks of both nonmelanoma skin cancer and other types of cancer were similarly low in patients randomized to ixekizumab at 80 mg every 2 weeks and those dosed every 4 weeks. That’s reassuring, because if a drug causes cancer one would expect to see that the more of the drug given, the higher the malignancy rate.
“You really don’t see any kind of a pattern or any difference between the various compared groups, including patients on etanercept, a drug that all of us feel very comfortable using in patients with psoriasis,” the dermatologist observed.
It’s thought that psoriasis patients have a higher background risk of malignancy than the general population due to the nature of the chronic disease, which involves immune dysregulation and chronic inflammation. In the studies to date, however, the malignancy risk in ixekizumab-treated patients is similar to those on placebo or etanercept.
“In my opinion, it’s comforting that the malignancy rates of the currently approved biologic agents for psoriasis are low and – one could argue – not above that which you would expect for the population of patients we’re treating. So I’ve always felt that monotherapy – and that’s an important point, not combination therapy with other immunosuppressive drugs – offers very little risk of de novo malignancy with the currently approved biologics,” Dr. Strober said.
Lumping together the results of the seven studies in his analysis, the incidence of squamous cell carcinoma was 0.1 cases per 100 patient-years of ixekizumab exposure, the risk of basal cell carcinoma was 0.3 cases per 100 patient-years, and the risk of malignancies other than nonmelanoma skin cancer was 0.5 per 100 patient-years of exposure to ixekizumab.
“That’s basically in line with every other biologic that I’ve examined with regard to malignancy rates: about 1 in 200 patients treated for 1 year gets a nonmelanoma skin cancer of some nature if you look at all data sources, including long-term registries and clinical trials. So it appears that this drug falls right in line with the other biologics in terms of rates of the more serious cancers, at least over the time period studied,” Dr. Strober commented.
He offered a caveat, however: “What will happen in patients given combination therapy with other immunosuppressive drugs? Or special populations, such as patients at higher risk for malignancy, be it skin cancer or other types of cancer? Or – and here’s the question we often have the most difficulty answering – what about people with a prior history of malignancy? What would be their risk of malignancy in being placed on a drug of this nature? Only time and really good registry data will allow us to answer these questions.”
Dr. Strober is a consultant to Eli Lilly, which sponsored the seven ixekizumab studies, as well as to numerous other pharmaceutical companies.
COPENHAGEN – Ixekizumab showed no hint of an increased malignancy risk of any sort in 4,208 patients with moderate to severe psoriasis who had 6,480 patient-years of exposure to the investigational interleukin-17A inhibitor in seven clinical trials.
“Patients were studied for an average of 18 months on ixekizumab. You may say that’s not enough time to assess the true risk for malignancy, and I would say you’re probably right. But 18 months isn’t shabby. From such an exposure, we can get some sense of the malignancy risk with this drug,” Dr. Bruce E. Strober asserted at the annual congress of the European Academy of Dermatology and Venereology.
That’s a key issue whenever a dermatologist prescribes a potent immune-altering anti-inflammatory medication for an indefinite period of time, he added.
In the randomized trials in which more than 2,300 patients who received 80 mg of ixekizumab every 2 or 4 weeks were compared with 739 patients assigned to etanercept (Enbrel) and 791 on placebo, malignancy rates were similarly low, at 0.1-0.3 cases per 100 patient-years across all four treatment arms, reported Dr. Strober, chair of the department of dermatology at the University of Connecticut, Farmington.
Moreover, in the long-term maintenance studies featuring 60 months of follow-up, the risks of both nonmelanoma skin cancer and other types of cancer were similarly low in patients randomized to ixekizumab at 80 mg every 2 weeks and those dosed every 4 weeks. That’s reassuring, because if a drug causes cancer one would expect to see that the more of the drug given, the higher the malignancy rate.
“You really don’t see any kind of a pattern or any difference between the various compared groups, including patients on etanercept, a drug that all of us feel very comfortable using in patients with psoriasis,” the dermatologist observed.
It’s thought that psoriasis patients have a higher background risk of malignancy than the general population due to the nature of the chronic disease, which involves immune dysregulation and chronic inflammation. In the studies to date, however, the malignancy risk in ixekizumab-treated patients is similar to those on placebo or etanercept.
“In my opinion, it’s comforting that the malignancy rates of the currently approved biologic agents for psoriasis are low and – one could argue – not above that which you would expect for the population of patients we’re treating. So I’ve always felt that monotherapy – and that’s an important point, not combination therapy with other immunosuppressive drugs – offers very little risk of de novo malignancy with the currently approved biologics,” Dr. Strober said.
Lumping together the results of the seven studies in his analysis, the incidence of squamous cell carcinoma was 0.1 cases per 100 patient-years of ixekizumab exposure, the risk of basal cell carcinoma was 0.3 cases per 100 patient-years, and the risk of malignancies other than nonmelanoma skin cancer was 0.5 per 100 patient-years of exposure to ixekizumab.
“That’s basically in line with every other biologic that I’ve examined with regard to malignancy rates: about 1 in 200 patients treated for 1 year gets a nonmelanoma skin cancer of some nature if you look at all data sources, including long-term registries and clinical trials. So it appears that this drug falls right in line with the other biologics in terms of rates of the more serious cancers, at least over the time period studied,” Dr. Strober commented.
He offered a caveat, however: “What will happen in patients given combination therapy with other immunosuppressive drugs? Or special populations, such as patients at higher risk for malignancy, be it skin cancer or other types of cancer? Or – and here’s the question we often have the most difficulty answering – what about people with a prior history of malignancy? What would be their risk of malignancy in being placed on a drug of this nature? Only time and really good registry data will allow us to answer these questions.”
Dr. Strober is a consultant to Eli Lilly, which sponsored the seven ixekizumab studies, as well as to numerous other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point: In studies conducted in psoriasis patients to date, ixekizumab doesn’t raise any malignancy concerns.
Major finding: The risk of malignancies of any type was 0.9 cases per 100 patient-years of exposure to the investigational interleukin-17A inhibitor ixekizumab, which is in line with rates associated with the currently approved biologic agents.
Data source: This was an analysis of treatment-emergent malignancies among participants in seven clinical psoriasis trials representing a total of 6,480 patient-years of exposure to ixekizumab.
Disclosures: The presenter of this analysis is a consultant to Eli Lilly, which sponsored the seven ixekizumab studies, as well as to numerous other pharmaceutical companies.
EADV: Secukinumab shows sustained high efficacy in SCULPTURE through 3 years
COPENHAGEN – Secukinumab maintained consistently high and previously unheard of long-term Psoriasis Area and Severity Index 90 and PASI 100 response rates in psoriasis patients through 3 years of prospective follow-up in an extension of the phase III SCULPTURE trial.
The key to maintaining these high response rates was to give secukinumab (Cosentyx) on a fixed schedule every 4 weeks. SCULPTURE tested the hypothesis that retreatment of initial responders only as needed based upon early evidence of relapse off treatment might be a reasonable alternative strategy. The answer turns out to be emphatically no.
“We can see from the 3-year data that retreatment as needed is probably not a good strategy over the long term. We see four times fewer patients with PASI 90s and 10 times fewer patients with PASI 100s if they are treated as needed. From the patient point of view, quality of life and satisfaction are not optimal with retreatment as needed,” Dr. Diamant Thaci said at the annual congress of the European Academy of Dermatology and Venereology.
The 3-year extension of SCULPTURE also provided reassuring safety data, he noted.
“It’s interesting that with long-term treatment out to 3 years, we don’t see any new safety signals. In fact, the signals we saw in the preapproval studies are slightly diluted here, and now that secukinumab is approved, we’re not seeing them as problems in real life clinical practice,” said Dr. Thaci of University Hospital in Lübeck, Germany.
The 52-week SCULPTURE results were published earlier this year (J Am Acad Dermatol. 2015 Jul;73[1]:27-36). Dr. Thaci’s update extended the findings from 52 weeks through 3 years.
The double-blind phase of the core SCULPTURE study began with 966 patients with moderate to severe plaque psoriasis and a baseline PASI score of about 23. After 12 weeks on subcutaneous secukinumab, the 842 subjects who had achieved 75% or more improvement from their baseline PASI score were randomized to one of four study arms: secukinumab at either 300 mg or 150 mg, administered either every 4 weeks or by retreatment as needed when an individual’s response dropped below PASI 75.
Picking up where the published 52-week results left off, Dr. Thaci focused special attention on the PASI 90 outcomes out to 3 years because he said he believes PASI 90 will become generally accepted as the new treatment target, now that secukinumab is on the market and other supereffective biologic agents are advancing through the developmental pipeline.
At 52 weeks, 68.5% of patients on fixed-dose secukinumab at 300 mg every 4 weeks had a PASI 90 response. At 3 years, after an additional 2 years on this schedule, the PASI 90 rate was still hovering around the two-thirds mark at 63.8%. In contrast, patients on retreatment with 300 mg as needed had a PASI 90 rate of 14.8% at 52 weeks and 13% at 3 years.
Although the Food and Drug Administration recommended the 300-mg dose when it approved secukinumab as a first-in-class biologic in January, the agency added that 150 mg may be acceptable in some patients. But the 150-mg dose was clearly inferior long term in the SCULPTURE extension. At 52 weeks, 50.3% of patients on secukinumab at 150 mg every 4 weeks had a PASI 90 response; at 3 years the rate was 36.8%. With retreatment as needed using 150 mg, the 52-week and 3-year PASI 90 rates were 12% and 11.8%, Dr. Thaci reported.
The PASI 100 (clear skin) response rate at 52 weeks with fixed-dose secukinumab at 300 mg was 43.8%. At 3 years it was 42.6%.
“These excellent responders kept their excellent response by continuing treatment for 3 years,” the dermatologist noted.
The PASI 100 rates with retreatment as needed were 5.3% and 3.8%.
In terms of safety through 3 years of treatment, rates of the adverse events of greatest interest with the use of immunomodulatory therapies – Candida infections, malignancies, serious infections, and major adverse cardiovascular events – were similarly low across all four treatment arms, ranging from 0 to 1.6 events per 100 subject-years. Thus, there was no safety advantage in using the retreatment as needed strategy.
“In the past we were all talking about the risk of Candida infection caused by inhibiting [interleukin]-17. But if you treat patients in real life clinical practice, you may see a signal of Candida infection in the beginning, yet in the long term you don’t see Candida as a significant side effect in our daily practice – and in this trial the incidence decreases,” Dr. Thaci said.
One audience member rose to criticize the way the analysis of the long-term extension of SCULPTURE was structured – namely, that only patients still on secukinumab at the 3-year mark were included in the results. This “as-observed” method inflates the efficacy results, he said. How many patients dropped out over time and weren’t counted? he asked.
Dr. Thaci replied that roughly 90% of patients in the trial at 52 weeks continued on secukinumab out to 3 years.
“It’s not a massive dropout. I wish it were a nonresponder imputation analysis, but in long-term studies, it’s very difficult to show that analysis because the vast majority of the trials that have been presented in the past have been ‘as observed.’ But I don’t think you’ll see a big difference between this as-observed analysis and a nonresponder imputation analysis. This is a very fair and convincing analysis,” he said.
Secukinumab is marketed by Novartis, which sponsored the study. Dr. Thaci serves as an investigator and consultant to Novartis and other pharmaceutical companies.
COPENHAGEN – Secukinumab maintained consistently high and previously unheard of long-term Psoriasis Area and Severity Index 90 and PASI 100 response rates in psoriasis patients through 3 years of prospective follow-up in an extension of the phase III SCULPTURE trial.
The key to maintaining these high response rates was to give secukinumab (Cosentyx) on a fixed schedule every 4 weeks. SCULPTURE tested the hypothesis that retreatment of initial responders only as needed based upon early evidence of relapse off treatment might be a reasonable alternative strategy. The answer turns out to be emphatically no.
“We can see from the 3-year data that retreatment as needed is probably not a good strategy over the long term. We see four times fewer patients with PASI 90s and 10 times fewer patients with PASI 100s if they are treated as needed. From the patient point of view, quality of life and satisfaction are not optimal with retreatment as needed,” Dr. Diamant Thaci said at the annual congress of the European Academy of Dermatology and Venereology.
The 3-year extension of SCULPTURE also provided reassuring safety data, he noted.
“It’s interesting that with long-term treatment out to 3 years, we don’t see any new safety signals. In fact, the signals we saw in the preapproval studies are slightly diluted here, and now that secukinumab is approved, we’re not seeing them as problems in real life clinical practice,” said Dr. Thaci of University Hospital in Lübeck, Germany.
The 52-week SCULPTURE results were published earlier this year (J Am Acad Dermatol. 2015 Jul;73[1]:27-36). Dr. Thaci’s update extended the findings from 52 weeks through 3 years.
The double-blind phase of the core SCULPTURE study began with 966 patients with moderate to severe plaque psoriasis and a baseline PASI score of about 23. After 12 weeks on subcutaneous secukinumab, the 842 subjects who had achieved 75% or more improvement from their baseline PASI score were randomized to one of four study arms: secukinumab at either 300 mg or 150 mg, administered either every 4 weeks or by retreatment as needed when an individual’s response dropped below PASI 75.
Picking up where the published 52-week results left off, Dr. Thaci focused special attention on the PASI 90 outcomes out to 3 years because he said he believes PASI 90 will become generally accepted as the new treatment target, now that secukinumab is on the market and other supereffective biologic agents are advancing through the developmental pipeline.
At 52 weeks, 68.5% of patients on fixed-dose secukinumab at 300 mg every 4 weeks had a PASI 90 response. At 3 years, after an additional 2 years on this schedule, the PASI 90 rate was still hovering around the two-thirds mark at 63.8%. In contrast, patients on retreatment with 300 mg as needed had a PASI 90 rate of 14.8% at 52 weeks and 13% at 3 years.
Although the Food and Drug Administration recommended the 300-mg dose when it approved secukinumab as a first-in-class biologic in January, the agency added that 150 mg may be acceptable in some patients. But the 150-mg dose was clearly inferior long term in the SCULPTURE extension. At 52 weeks, 50.3% of patients on secukinumab at 150 mg every 4 weeks had a PASI 90 response; at 3 years the rate was 36.8%. With retreatment as needed using 150 mg, the 52-week and 3-year PASI 90 rates were 12% and 11.8%, Dr. Thaci reported.
The PASI 100 (clear skin) response rate at 52 weeks with fixed-dose secukinumab at 300 mg was 43.8%. At 3 years it was 42.6%.
“These excellent responders kept their excellent response by continuing treatment for 3 years,” the dermatologist noted.
The PASI 100 rates with retreatment as needed were 5.3% and 3.8%.
In terms of safety through 3 years of treatment, rates of the adverse events of greatest interest with the use of immunomodulatory therapies – Candida infections, malignancies, serious infections, and major adverse cardiovascular events – were similarly low across all four treatment arms, ranging from 0 to 1.6 events per 100 subject-years. Thus, there was no safety advantage in using the retreatment as needed strategy.
“In the past we were all talking about the risk of Candida infection caused by inhibiting [interleukin]-17. But if you treat patients in real life clinical practice, you may see a signal of Candida infection in the beginning, yet in the long term you don’t see Candida as a significant side effect in our daily practice – and in this trial the incidence decreases,” Dr. Thaci said.
One audience member rose to criticize the way the analysis of the long-term extension of SCULPTURE was structured – namely, that only patients still on secukinumab at the 3-year mark were included in the results. This “as-observed” method inflates the efficacy results, he said. How many patients dropped out over time and weren’t counted? he asked.
Dr. Thaci replied that roughly 90% of patients in the trial at 52 weeks continued on secukinumab out to 3 years.
“It’s not a massive dropout. I wish it were a nonresponder imputation analysis, but in long-term studies, it’s very difficult to show that analysis because the vast majority of the trials that have been presented in the past have been ‘as observed.’ But I don’t think you’ll see a big difference between this as-observed analysis and a nonresponder imputation analysis. This is a very fair and convincing analysis,” he said.
Secukinumab is marketed by Novartis, which sponsored the study. Dr. Thaci serves as an investigator and consultant to Novartis and other pharmaceutical companies.
COPENHAGEN – Secukinumab maintained consistently high and previously unheard of long-term Psoriasis Area and Severity Index 90 and PASI 100 response rates in psoriasis patients through 3 years of prospective follow-up in an extension of the phase III SCULPTURE trial.
The key to maintaining these high response rates was to give secukinumab (Cosentyx) on a fixed schedule every 4 weeks. SCULPTURE tested the hypothesis that retreatment of initial responders only as needed based upon early evidence of relapse off treatment might be a reasonable alternative strategy. The answer turns out to be emphatically no.
“We can see from the 3-year data that retreatment as needed is probably not a good strategy over the long term. We see four times fewer patients with PASI 90s and 10 times fewer patients with PASI 100s if they are treated as needed. From the patient point of view, quality of life and satisfaction are not optimal with retreatment as needed,” Dr. Diamant Thaci said at the annual congress of the European Academy of Dermatology and Venereology.
The 3-year extension of SCULPTURE also provided reassuring safety data, he noted.
“It’s interesting that with long-term treatment out to 3 years, we don’t see any new safety signals. In fact, the signals we saw in the preapproval studies are slightly diluted here, and now that secukinumab is approved, we’re not seeing them as problems in real life clinical practice,” said Dr. Thaci of University Hospital in Lübeck, Germany.
The 52-week SCULPTURE results were published earlier this year (J Am Acad Dermatol. 2015 Jul;73[1]:27-36). Dr. Thaci’s update extended the findings from 52 weeks through 3 years.
The double-blind phase of the core SCULPTURE study began with 966 patients with moderate to severe plaque psoriasis and a baseline PASI score of about 23. After 12 weeks on subcutaneous secukinumab, the 842 subjects who had achieved 75% or more improvement from their baseline PASI score were randomized to one of four study arms: secukinumab at either 300 mg or 150 mg, administered either every 4 weeks or by retreatment as needed when an individual’s response dropped below PASI 75.
Picking up where the published 52-week results left off, Dr. Thaci focused special attention on the PASI 90 outcomes out to 3 years because he said he believes PASI 90 will become generally accepted as the new treatment target, now that secukinumab is on the market and other supereffective biologic agents are advancing through the developmental pipeline.
At 52 weeks, 68.5% of patients on fixed-dose secukinumab at 300 mg every 4 weeks had a PASI 90 response. At 3 years, after an additional 2 years on this schedule, the PASI 90 rate was still hovering around the two-thirds mark at 63.8%. In contrast, patients on retreatment with 300 mg as needed had a PASI 90 rate of 14.8% at 52 weeks and 13% at 3 years.
Although the Food and Drug Administration recommended the 300-mg dose when it approved secukinumab as a first-in-class biologic in January, the agency added that 150 mg may be acceptable in some patients. But the 150-mg dose was clearly inferior long term in the SCULPTURE extension. At 52 weeks, 50.3% of patients on secukinumab at 150 mg every 4 weeks had a PASI 90 response; at 3 years the rate was 36.8%. With retreatment as needed using 150 mg, the 52-week and 3-year PASI 90 rates were 12% and 11.8%, Dr. Thaci reported.
The PASI 100 (clear skin) response rate at 52 weeks with fixed-dose secukinumab at 300 mg was 43.8%. At 3 years it was 42.6%.
“These excellent responders kept their excellent response by continuing treatment for 3 years,” the dermatologist noted.
The PASI 100 rates with retreatment as needed were 5.3% and 3.8%.
In terms of safety through 3 years of treatment, rates of the adverse events of greatest interest with the use of immunomodulatory therapies – Candida infections, malignancies, serious infections, and major adverse cardiovascular events – were similarly low across all four treatment arms, ranging from 0 to 1.6 events per 100 subject-years. Thus, there was no safety advantage in using the retreatment as needed strategy.
“In the past we were all talking about the risk of Candida infection caused by inhibiting [interleukin]-17. But if you treat patients in real life clinical practice, you may see a signal of Candida infection in the beginning, yet in the long term you don’t see Candida as a significant side effect in our daily practice – and in this trial the incidence decreases,” Dr. Thaci said.
One audience member rose to criticize the way the analysis of the long-term extension of SCULPTURE was structured – namely, that only patients still on secukinumab at the 3-year mark were included in the results. This “as-observed” method inflates the efficacy results, he said. How many patients dropped out over time and weren’t counted? he asked.
Dr. Thaci replied that roughly 90% of patients in the trial at 52 weeks continued on secukinumab out to 3 years.
“It’s not a massive dropout. I wish it were a nonresponder imputation analysis, but in long-term studies, it’s very difficult to show that analysis because the vast majority of the trials that have been presented in the past have been ‘as observed.’ But I don’t think you’ll see a big difference between this as-observed analysis and a nonresponder imputation analysis. This is a very fair and convincing analysis,” he said.
Secukinumab is marketed by Novartis, which sponsored the study. Dr. Thaci serves as an investigator and consultant to Novartis and other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point: Three-year data from a phase III trial provide reassurance regarding the long-term safety and efficacy of secukinumab for psoriasis.
Major finding: The high level of improvement seen in the 52-week core study of secukinumab, at the dose of 300 mg every 4 weeks, in patients with psoriasis was largely maintained with 2 more years of therapy, with a 63.8% PASI-90 rate at 3 years.
Data source: A prospective 2-year extension of a 1-year, randomized double-blind trial of 842 psoriasis patients, who initially achieved at least a PASI-75 response to secukinumab.
Disclosures: The study was sponsored by Novartis. The presenter serves as a paid investigator and consultant for Novartis and other pharmaceutical companies.
EADV: Spotlight on alexithymia in psoriasis
COPENHAGEN – Alexithymia – difficulty in recognizing and describing one’s emotions – is exceptionally common among psoriasis patients and may represent a novel therapeutic target, according to Dr. Carle Paul.
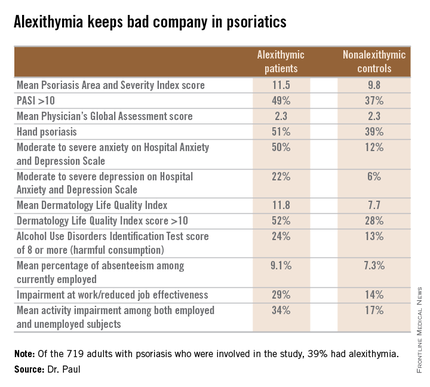
“We found a significant association between alexithymia and more severe psoriasis, anxiety, depression, decreased quality of life, harmful alcohol consumption, and work impairment,” reported Dr. Paul, professor and chairman of the department of dermatology at the University of Toulouse (France).
Alexithymia, a personality construct sometimes referred to as “emotional blindness,” was first described by psychologists in the 1970s. The previous glaring lack of data on the prevalence and consequences of alexithymia in psoriasis patients served as the impetus for the ongoing EPIDEPSO study (Epidemiological Study in Patients With Recently Diagnosed Psoriasis), a prospective 1-year international, epidemiologic, noninterventional observational study involving 719 adults with moderate to severe plaque psoriasis of less than 10 years’ duration. Dr. Paul presented the baseline findings at the at the annual congress of the European Academy of Dermatology and Venereology.
The first noteworthy finding was the strikingly high prevalence of alexithymia in this group of psoriasis patients: 39% of the 719 patients had alexithymia, as defined by a score of 61 or more on the validated, 20-item Toronto Alexithymia Scale.
Patients with alexithymia had slightly but significantly more severe psoriasis as evidenced by their mean Psoriasis Area and Severity Index score of 11.5, compared with 9.8 in unaffected patients. Hand psoriasis was more common in alexithymic patients by a margin of 51%-39%, although the prevalence of psoriasis of the face and neck was similar in the two groups.
Alexithymia was associated with significantly higher rates of several forms of psychiatric comorbidity and problems in living as assessed by validated tests. One comorbid condition stood out above the rest.
“The most striking feature is the very close relationship between alexithymia and anxiety,” according to Dr. Paul. “Among alexithymic patients, 50% had moderate to severe anxiety, as measured by the Hospital Anxiety and Depression Scale–A, whereas in psoriasis patients without alexithymia, this proportion was 12%.”
“Alexithymia identifies a patient population with a high burden of psoriasis, but at the same time, they have difficulty in expressing their emotions and feelings to their doctor. And I think this may explain the fact that some psoriasis patients have difficulties in interacting with doctors, because even though they have a high psoriasis burden, they cannot express how much they suffer from the disease,” the dermatologist continued.
Audience members at this standing-room-only EADV session on new research findings in psoriasis were clearly intrigued by the novel findings about a psychological condition unfamiliar to most. They wanted to know if the Toronto Alexithymia Scale is suitable for use in everyday clinical practice. The answer is yes, Dr. Paul replied, but the practical importance of identifying the large subgroup of patients with alexithymia has yet to be determined.
“We want to find out if we can modify alexithymia with interventions, but we don’t have the prospective data yet,” he added.
Even if alexithymia turns out to be a fixed characteristic not amenable to intervention, however, EPIDEPSO has shown that it could be useful as a red flag because it keeps company with several psychiatric conditions, which are anxiety, depression, and alcohol abuse.
What about causality? others asked. Does alexithymia cause anxiety and depression, or does the emotional toll of psoriasis promote anxiety and depression, which then causes alexithymia? Dr. Paul responded that it’s impossible to say at this point because these initial EPIDEPSO data are cross-sectional; however, the 1-year follow-up may yield insight.
EPIDEPSO is funded by Janssen. Dr. Paul reported receiving a research grant from the company to conduct the study.
COPENHAGEN – Alexithymia – difficulty in recognizing and describing one’s emotions – is exceptionally common among psoriasis patients and may represent a novel therapeutic target, according to Dr. Carle Paul.
“We found a significant association between alexithymia and more severe psoriasis, anxiety, depression, decreased quality of life, harmful alcohol consumption, and work impairment,” reported Dr. Paul, professor and chairman of the department of dermatology at the University of Toulouse (France).
Alexithymia, a personality construct sometimes referred to as “emotional blindness,” was first described by psychologists in the 1970s. The previous glaring lack of data on the prevalence and consequences of alexithymia in psoriasis patients served as the impetus for the ongoing EPIDEPSO study (Epidemiological Study in Patients With Recently Diagnosed Psoriasis), a prospective 1-year international, epidemiologic, noninterventional observational study involving 719 adults with moderate to severe plaque psoriasis of less than 10 years’ duration. Dr. Paul presented the baseline findings at the at the annual congress of the European Academy of Dermatology and Venereology.
The first noteworthy finding was the strikingly high prevalence of alexithymia in this group of psoriasis patients: 39% of the 719 patients had alexithymia, as defined by a score of 61 or more on the validated, 20-item Toronto Alexithymia Scale.
Patients with alexithymia had slightly but significantly more severe psoriasis as evidenced by their mean Psoriasis Area and Severity Index score of 11.5, compared with 9.8 in unaffected patients. Hand psoriasis was more common in alexithymic patients by a margin of 51%-39%, although the prevalence of psoriasis of the face and neck was similar in the two groups.
Alexithymia was associated with significantly higher rates of several forms of psychiatric comorbidity and problems in living as assessed by validated tests. One comorbid condition stood out above the rest.
“The most striking feature is the very close relationship between alexithymia and anxiety,” according to Dr. Paul. “Among alexithymic patients, 50% had moderate to severe anxiety, as measured by the Hospital Anxiety and Depression Scale–A, whereas in psoriasis patients without alexithymia, this proportion was 12%.”
“Alexithymia identifies a patient population with a high burden of psoriasis, but at the same time, they have difficulty in expressing their emotions and feelings to their doctor. And I think this may explain the fact that some psoriasis patients have difficulties in interacting with doctors, because even though they have a high psoriasis burden, they cannot express how much they suffer from the disease,” the dermatologist continued.

Audience members at this standing-room-only EADV session on new research findings in psoriasis were clearly intrigued by the novel findings about a psychological condition unfamiliar to most. They wanted to know if the Toronto Alexithymia Scale is suitable for use in everyday clinical practice. The answer is yes, Dr. Paul replied, but the practical importance of identifying the large subgroup of patients with alexithymia has yet to be determined.
“We want to find out if we can modify alexithymia with interventions, but we don’t have the prospective data yet,” he added.
Even if alexithymia turns out to be a fixed characteristic not amenable to intervention, however, EPIDEPSO has shown that it could be useful as a red flag because it keeps company with several psychiatric conditions, which are anxiety, depression, and alcohol abuse.
What about causality? others asked. Does alexithymia cause anxiety and depression, or does the emotional toll of psoriasis promote anxiety and depression, which then causes alexithymia? Dr. Paul responded that it’s impossible to say at this point because these initial EPIDEPSO data are cross-sectional; however, the 1-year follow-up may yield insight.
EPIDEPSO is funded by Janssen. Dr. Paul reported receiving a research grant from the company to conduct the study.
COPENHAGEN – Alexithymia – difficulty in recognizing and describing one’s emotions – is exceptionally common among psoriasis patients and may represent a novel therapeutic target, according to Dr. Carle Paul.
“We found a significant association between alexithymia and more severe psoriasis, anxiety, depression, decreased quality of life, harmful alcohol consumption, and work impairment,” reported Dr. Paul, professor and chairman of the department of dermatology at the University of Toulouse (France).
Alexithymia, a personality construct sometimes referred to as “emotional blindness,” was first described by psychologists in the 1970s. The previous glaring lack of data on the prevalence and consequences of alexithymia in psoriasis patients served as the impetus for the ongoing EPIDEPSO study (Epidemiological Study in Patients With Recently Diagnosed Psoriasis), a prospective 1-year international, epidemiologic, noninterventional observational study involving 719 adults with moderate to severe plaque psoriasis of less than 10 years’ duration. Dr. Paul presented the baseline findings at the at the annual congress of the European Academy of Dermatology and Venereology.
The first noteworthy finding was the strikingly high prevalence of alexithymia in this group of psoriasis patients: 39% of the 719 patients had alexithymia, as defined by a score of 61 or more on the validated, 20-item Toronto Alexithymia Scale.
Patients with alexithymia had slightly but significantly more severe psoriasis as evidenced by their mean Psoriasis Area and Severity Index score of 11.5, compared with 9.8 in unaffected patients. Hand psoriasis was more common in alexithymic patients by a margin of 51%-39%, although the prevalence of psoriasis of the face and neck was similar in the two groups.
Alexithymia was associated with significantly higher rates of several forms of psychiatric comorbidity and problems in living as assessed by validated tests. One comorbid condition stood out above the rest.
“The most striking feature is the very close relationship between alexithymia and anxiety,” according to Dr. Paul. “Among alexithymic patients, 50% had moderate to severe anxiety, as measured by the Hospital Anxiety and Depression Scale–A, whereas in psoriasis patients without alexithymia, this proportion was 12%.”
“Alexithymia identifies a patient population with a high burden of psoriasis, but at the same time, they have difficulty in expressing their emotions and feelings to their doctor. And I think this may explain the fact that some psoriasis patients have difficulties in interacting with doctors, because even though they have a high psoriasis burden, they cannot express how much they suffer from the disease,” the dermatologist continued.

Audience members at this standing-room-only EADV session on new research findings in psoriasis were clearly intrigued by the novel findings about a psychological condition unfamiliar to most. They wanted to know if the Toronto Alexithymia Scale is suitable for use in everyday clinical practice. The answer is yes, Dr. Paul replied, but the practical importance of identifying the large subgroup of patients with alexithymia has yet to be determined.
“We want to find out if we can modify alexithymia with interventions, but we don’t have the prospective data yet,” he added.
Even if alexithymia turns out to be a fixed characteristic not amenable to intervention, however, EPIDEPSO has shown that it could be useful as a red flag because it keeps company with several psychiatric conditions, which are anxiety, depression, and alcohol abuse.
What about causality? others asked. Does alexithymia cause anxiety and depression, or does the emotional toll of psoriasis promote anxiety and depression, which then causes alexithymia? Dr. Paul responded that it’s impossible to say at this point because these initial EPIDEPSO data are cross-sectional; however, the 1-year follow-up may yield insight.
EPIDEPSO is funded by Janssen. Dr. Paul reported receiving a research grant from the company to conduct the study.
AT THE EADV CONGRESS
Key clinical point: Alexithymia is strikingly common among psoriasis patients and is associated with multiple psychiatric comorbidities and problems in living.
Major finding: Thirty-nine percent of a large cohort of psoriasis patients met the criteria for alexithymia, an inability to identify and describe one’s emotions. Affected patients had markedly higher rates of anxiety, depression, problem drinking, and impairments of quality of life and work productivity.
Data source: The EPIDEPSO study, an ongoing 1-year prospective, observational international study involving 719 adults with moderate to severe psoriasis of less than 10 years’ duration.
Disclosures: The EPIDEPSO study is funded by Janssen, which provided the presenter with a research grant.
EADV: Long-term apremilast results show what to expect for psoriasis
COPENHAGEN – What can physicians and their psoriasis patients realistically expect from long-term apremilast therapy?
“I think the take-home number is a 50%-plus PASI-75 response after 1 year among patients with moderate to severe plaque psoriasis,” Dr. Kristian Reich said at the annual congress of the European Academy of Dermatology and Venereology.
He presented what he considers to be the first solid data addressing this key question. The data come from a new analysis of the 52-week results of the LIBERATE trial.
“Due to the randomized withdrawal design and long-term treatment only of responders in the original pivotal phase III ESTEEM program, I think we’ve had no real understanding from clinical trials of what the long-term efficacy of apremilast [Otezla] is. Of course, with psoriasis being a chronic disease, this is really what we want to know. And we can get this data very nicely from the LIBERATE study. I should point out that although LIBERATE is a relatively small study, every single patient is included here. So I think this is a very robust analysis of 1-year efficacy data,” said Dr. Reich, managing partner of Dermatologikum Hamburg (Germany).
In contrast to the inclusive analysis performed in LIBERATE, in the pivotal ESTEEM I trial a mere 77 of 562 psoriasis patients randomized to apremilast were on the oral phosphodiesterase type 4 inhibitor for the full 52-week study period, with 61% of that superselect subgroup showing a PASI-75 response (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
LIBERATE included 250 psoriasis patients. Their baseline Psoriasis Area and Severity Index (PASI) score was about 20, with 27% body surface area involved, a body mass index of 29 kg/m2, 89 kg of body weight, and a mean Dermatology Life Quality Index (DLQI) score of 13. Seventy percent to 80% of subjects had previously used conventional systemic therapies, but no one was allowed to have prior use of biologic therapies.
Patients were randomized to one of three study arms: 16 weeks of oral apremilast at the approved dose of 30 mg twice daily plus a weekly placebo injection, subcutaneous etanercept (Enbrel) at 50 mg once weekly plus a placebo tablet, or dual placebos. At 16 weeks, all patients were switched to apremilast at 30 mg twice daily with no placebos through week 52.
The PASI-75 response rate at week 16 was 11.9% in the placebo group, 39.8% with apremilast, and 48.2% with etanercept.
“I am surprised to see in the more recent studies the high response to etanercept. This is fantastic data. Etanercept seems to ripen like old wine. It’s getting better the longer we have it,” the dermatologist commented.
The PASI-75 rates at 1 year were 46.4% with placebo/apremilast, 50.6% with apremilast throughout, and 55.4% with etanercept/apremilast.
The mean improvement in DLQI from baseline to 52 weeks – a secondary endpoint – was 6.6 points with placebo/apremilast, 8.0 with apremilast/apremilast, and 8.9 points with etanercept/apremilast. A noteworthy finding was that during the first 16 weeks apremilast brought a clinically meaningful improvement in quality of life significantly faster than etanercept did, with a significant difference seen between the two study arms even in the first 1-2 weeks.
The likely explanation for this benefit is that the mean reduction in pruritus visual analog scale scores was significantly greater with apremilast than etanercept through the first 8 weeks. Although pruritus is traditionally thought of more in the context of atopic dermatitis, it’s actually also the No. 1 complaint among psoriasis patients, according to Dr. Reich.
“This is a special thing with apremilast, that the pruritus really goes down very significantly early on. It raises an interesting question about what the role of phosphodiesterase-4 inhibition might be in pruritus. I couldn’t give you a molecular explanation, but I think because pruritus is so annoying and it definitely affects quality of life, this could be a possible explanation for why there is more rapid improvement in quality of life independent of the PASI improvement,” he said.
Switching from etanercept to apremilast didn’t result in any clinically significant safety findings through week 52. No meaningful laboratory changes occurred during 52 weeks of monitoring. There were no cases of suicidal ideation, no increase in serious infections, and only a single cardiac event. The most common apremilast-associated side effects seen in LIBERATE were loose stools in 8%-15% of patients, nausea in 10%, and headache in 13%. These adverse events were mild to moderate in nature and decreased in prevalence over time. The maximum weight loss noted over the course of 52 weeks occurred in the placebo-to-apremilast group, with a mean 1.3-kg reduction.
“I would call the LIBERATE results a validation of the earlier data showing a very clean safety profile with this drug,” Dr. Reich said.
This is reflected in the product labeling, which unlike other systemic therapies for psoriasis includes no requirements for laboratory monitoring or tuberculosis testing.
Both the LIBERATE and the ESTEEM trials were sponsored by Celgene. Dr. Reich received research grants as an investigator in both programs.
COPENHAGEN – What can physicians and their psoriasis patients realistically expect from long-term apremilast therapy?
“I think the take-home number is a 50%-plus PASI-75 response after 1 year among patients with moderate to severe plaque psoriasis,” Dr. Kristian Reich said at the annual congress of the European Academy of Dermatology and Venereology.
He presented what he considers to be the first solid data addressing this key question. The data come from a new analysis of the 52-week results of the LIBERATE trial.
“Due to the randomized withdrawal design and long-term treatment only of responders in the original pivotal phase III ESTEEM program, I think we’ve had no real understanding from clinical trials of what the long-term efficacy of apremilast [Otezla] is. Of course, with psoriasis being a chronic disease, this is really what we want to know. And we can get this data very nicely from the LIBERATE study. I should point out that although LIBERATE is a relatively small study, every single patient is included here. So I think this is a very robust analysis of 1-year efficacy data,” said Dr. Reich, managing partner of Dermatologikum Hamburg (Germany).
In contrast to the inclusive analysis performed in LIBERATE, in the pivotal ESTEEM I trial a mere 77 of 562 psoriasis patients randomized to apremilast were on the oral phosphodiesterase type 4 inhibitor for the full 52-week study period, with 61% of that superselect subgroup showing a PASI-75 response (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
LIBERATE included 250 psoriasis patients. Their baseline Psoriasis Area and Severity Index (PASI) score was about 20, with 27% body surface area involved, a body mass index of 29 kg/m2, 89 kg of body weight, and a mean Dermatology Life Quality Index (DLQI) score of 13. Seventy percent to 80% of subjects had previously used conventional systemic therapies, but no one was allowed to have prior use of biologic therapies.
Patients were randomized to one of three study arms: 16 weeks of oral apremilast at the approved dose of 30 mg twice daily plus a weekly placebo injection, subcutaneous etanercept (Enbrel) at 50 mg once weekly plus a placebo tablet, or dual placebos. At 16 weeks, all patients were switched to apremilast at 30 mg twice daily with no placebos through week 52.
The PASI-75 response rate at week 16 was 11.9% in the placebo group, 39.8% with apremilast, and 48.2% with etanercept.
“I am surprised to see in the more recent studies the high response to etanercept. This is fantastic data. Etanercept seems to ripen like old wine. It’s getting better the longer we have it,” the dermatologist commented.
The PASI-75 rates at 1 year were 46.4% with placebo/apremilast, 50.6% with apremilast throughout, and 55.4% with etanercept/apremilast.
The mean improvement in DLQI from baseline to 52 weeks – a secondary endpoint – was 6.6 points with placebo/apremilast, 8.0 with apremilast/apremilast, and 8.9 points with etanercept/apremilast. A noteworthy finding was that during the first 16 weeks apremilast brought a clinically meaningful improvement in quality of life significantly faster than etanercept did, with a significant difference seen between the two study arms even in the first 1-2 weeks.
The likely explanation for this benefit is that the mean reduction in pruritus visual analog scale scores was significantly greater with apremilast than etanercept through the first 8 weeks. Although pruritus is traditionally thought of more in the context of atopic dermatitis, it’s actually also the No. 1 complaint among psoriasis patients, according to Dr. Reich.
“This is a special thing with apremilast, that the pruritus really goes down very significantly early on. It raises an interesting question about what the role of phosphodiesterase-4 inhibition might be in pruritus. I couldn’t give you a molecular explanation, but I think because pruritus is so annoying and it definitely affects quality of life, this could be a possible explanation for why there is more rapid improvement in quality of life independent of the PASI improvement,” he said.
Switching from etanercept to apremilast didn’t result in any clinically significant safety findings through week 52. No meaningful laboratory changes occurred during 52 weeks of monitoring. There were no cases of suicidal ideation, no increase in serious infections, and only a single cardiac event. The most common apremilast-associated side effects seen in LIBERATE were loose stools in 8%-15% of patients, nausea in 10%, and headache in 13%. These adverse events were mild to moderate in nature and decreased in prevalence over time. The maximum weight loss noted over the course of 52 weeks occurred in the placebo-to-apremilast group, with a mean 1.3-kg reduction.
“I would call the LIBERATE results a validation of the earlier data showing a very clean safety profile with this drug,” Dr. Reich said.
This is reflected in the product labeling, which unlike other systemic therapies for psoriasis includes no requirements for laboratory monitoring or tuberculosis testing.
Both the LIBERATE and the ESTEEM trials were sponsored by Celgene. Dr. Reich received research grants as an investigator in both programs.
COPENHAGEN – What can physicians and their psoriasis patients realistically expect from long-term apremilast therapy?
“I think the take-home number is a 50%-plus PASI-75 response after 1 year among patients with moderate to severe plaque psoriasis,” Dr. Kristian Reich said at the annual congress of the European Academy of Dermatology and Venereology.
He presented what he considers to be the first solid data addressing this key question. The data come from a new analysis of the 52-week results of the LIBERATE trial.
“Due to the randomized withdrawal design and long-term treatment only of responders in the original pivotal phase III ESTEEM program, I think we’ve had no real understanding from clinical trials of what the long-term efficacy of apremilast [Otezla] is. Of course, with psoriasis being a chronic disease, this is really what we want to know. And we can get this data very nicely from the LIBERATE study. I should point out that although LIBERATE is a relatively small study, every single patient is included here. So I think this is a very robust analysis of 1-year efficacy data,” said Dr. Reich, managing partner of Dermatologikum Hamburg (Germany).
In contrast to the inclusive analysis performed in LIBERATE, in the pivotal ESTEEM I trial a mere 77 of 562 psoriasis patients randomized to apremilast were on the oral phosphodiesterase type 4 inhibitor for the full 52-week study period, with 61% of that superselect subgroup showing a PASI-75 response (J Am Acad Dermatol. 2015 Jul;73[1]:37-49).
LIBERATE included 250 psoriasis patients. Their baseline Psoriasis Area and Severity Index (PASI) score was about 20, with 27% body surface area involved, a body mass index of 29 kg/m2, 89 kg of body weight, and a mean Dermatology Life Quality Index (DLQI) score of 13. Seventy percent to 80% of subjects had previously used conventional systemic therapies, but no one was allowed to have prior use of biologic therapies.
Patients were randomized to one of three study arms: 16 weeks of oral apremilast at the approved dose of 30 mg twice daily plus a weekly placebo injection, subcutaneous etanercept (Enbrel) at 50 mg once weekly plus a placebo tablet, or dual placebos. At 16 weeks, all patients were switched to apremilast at 30 mg twice daily with no placebos through week 52.
The PASI-75 response rate at week 16 was 11.9% in the placebo group, 39.8% with apremilast, and 48.2% with etanercept.
“I am surprised to see in the more recent studies the high response to etanercept. This is fantastic data. Etanercept seems to ripen like old wine. It’s getting better the longer we have it,” the dermatologist commented.
The PASI-75 rates at 1 year were 46.4% with placebo/apremilast, 50.6% with apremilast throughout, and 55.4% with etanercept/apremilast.
The mean improvement in DLQI from baseline to 52 weeks – a secondary endpoint – was 6.6 points with placebo/apremilast, 8.0 with apremilast/apremilast, and 8.9 points with etanercept/apremilast. A noteworthy finding was that during the first 16 weeks apremilast brought a clinically meaningful improvement in quality of life significantly faster than etanercept did, with a significant difference seen between the two study arms even in the first 1-2 weeks.
The likely explanation for this benefit is that the mean reduction in pruritus visual analog scale scores was significantly greater with apremilast than etanercept through the first 8 weeks. Although pruritus is traditionally thought of more in the context of atopic dermatitis, it’s actually also the No. 1 complaint among psoriasis patients, according to Dr. Reich.
“This is a special thing with apremilast, that the pruritus really goes down very significantly early on. It raises an interesting question about what the role of phosphodiesterase-4 inhibition might be in pruritus. I couldn’t give you a molecular explanation, but I think because pruritus is so annoying and it definitely affects quality of life, this could be a possible explanation for why there is more rapid improvement in quality of life independent of the PASI improvement,” he said.
Switching from etanercept to apremilast didn’t result in any clinically significant safety findings through week 52. No meaningful laboratory changes occurred during 52 weeks of monitoring. There were no cases of suicidal ideation, no increase in serious infections, and only a single cardiac event. The most common apremilast-associated side effects seen in LIBERATE were loose stools in 8%-15% of patients, nausea in 10%, and headache in 13%. These adverse events were mild to moderate in nature and decreased in prevalence over time. The maximum weight loss noted over the course of 52 weeks occurred in the placebo-to-apremilast group, with a mean 1.3-kg reduction.
“I would call the LIBERATE results a validation of the earlier data showing a very clean safety profile with this drug,” Dr. Reich said.
This is reflected in the product labeling, which unlike other systemic therapies for psoriasis includes no requirements for laboratory monitoring or tuberculosis testing.
Both the LIBERATE and the ESTEEM trials were sponsored by Celgene. Dr. Reich received research grants as an investigator in both programs.
AT THE EADV CONGRESS
Key clinical point: Patients on apremilast for moderate to severe plaque psoriasis can reasonably anticipate a 50%-plus likelihood of a PASI-75 response after 52 weeks of treatment.
Major finding: A 75% or greater improvement in Psoriasis Area and Severity Index scores was documented in 50.6% of patients after 52 weeks on apremilast and in 55.4% who underwent a protocol-mandated switch from etanercept to apremilast after 16 weeks on the injectable biologic agent.
Data source: A prospective 52-week study randomizing 250 patients with moderate to severe psoriasis to 16 weeks of oral apremilast, etanercept, or placebo followed by 36 weeks of apremilast for all.
Disclosures: The LIBERATE trial was sponsored by Celgene. The presenter received a research grant from the company.
EADV: Ixekizumab promising for psoriatic arthritis
COPENHAGEN – Ixekizumab not only shows considerable promise for the treatment of moderate to severe psoriasis, it looks like it may be a winner for comorbid psoriatic arthritis, too.
The investigational IgG4 humanized monoclonal antibody directed against interleukin-17A brought marked improvements in joint pain, systemic inflammatory burden, and quality of life as well as skin disease in patients with both psoriasis and self-reported psoriatic arthritis in a combined analysis of three phase III clinical trials, Dr. Alice B. Gottlieb reported at the annual congress of the European Academy of Dermatology and Venereology.
Of the 3,126 patients with moderate to severe psoriasis who participated in the 12-week, phase III UNCOVER-1, -2, and -3 trials, 751 (24%) also had self-reported psoriatic arthritis; her analysis focused on them.
She was quick to note that the UNCOVER trials were primarily psoriasis studies that relied upon patient self-report of psoriatic arthritis. Nevertheless, it seems likely that the great majority of self-reported psoriatic arthritis patients really did have the rheumatologic disease, since the mean baseline C-reactive protein (CRP) level in that group was 8.43 mg/L, a level far higher than expected in patients with psoriasis only.
In any case, more-rigorous phase III studies of ixekizumab conducted specifically in patients with formally rheumatologist-diagnosed psoriatic arthritis and treated in rheumatology practices are due to be presented at the annual meeting of the American College of Rheumatology in November. And while Dr. Gottlieb wasn’t at liberty to discuss those results, she did hint that the data will be strongly positive.
“If you’re happy about these UNCOVER findings, you’ll be ecstatic about those,” predicted Dr. Gottlieb, professor of dermatology and dermatologist in chief at Tufts Medical Center, Boston.
Also coming up at the American College of Rheumatology meeting will be the results of the first-ever head-to-head comparison of an IL-17 inhibitor versus a tumor necrosis factor–alpha blocker in psoriatic arthritis patients. While at present most physicians consider a TNF inhibitor to be the treatment of choice in patients with psoriatic arthritis, that view may change as a result of the forthcoming comparative study, according to the dermatologist.
In each of the three phase III UNCOVER studies, patients were randomized to 12 weeks of subcutaneous ixekizumab at 80 mg every 2 or 4 weeks following a 160-mg loading dose, or to placebo. At baseline, the subgroup with self-reported psoriatic arthritis had a mean Psoriasis Area and Severity Index ( PASI) 0f about 21, a self-rated joint pain severity of 50 on a 0-100 scale, a CRP of 8.43 mg/L, and a Dermatology Life Quality Index (DLQI) score of 14.
Joint pain decreased dramatically in the two ixekizumab groups as early at 2 weeks into the trial, at which point, patients on treatment every 2 weeks averaged a 13.1-point reduction from baseline, with a similar 14.1-point drop noted in those on an every 4 weeks schedule. At week 12, the mean reductions from baseline were 25.2 and 26.8 points, compared with a 1.1-point increase in joint pain among placebo-treated controls.
Inflammatory burden plunged quickly, as evidenced by mean reductions in CRP of 4.63 mg/L and 4.33 mg/L at week 1 with biweekly and monthly dosing, respectively. These reductions were then maintained through week 12.
In terms of improvement in skin symptoms, with ixekizumab dosed every 2 weeks, the PASI 75 response was 89.8% at 12 weeks, the PASI 90 response was 69.3%, and the PASI 100 response (clear skin) was 37.1%. In patients treated every 4 weeks, the rates were 81.1%, 60.8%, and 34.7%.
“There’s good news in both groups, but I think the news is even better in the every-2-weeks group,” Dr. Gottlieb commented.
The mean 12-week decrease from baseline in DLQI was 11.8 points with biweekly dosing and 10.5 with 4-week dosing, compared with 0.8 points in controls. That’s impressive given that a 5-point reduction in DLQI is deemed clinically meaningful, the dermatologist observed. At 12 weeks, 56.5% of patients on ixekizumab every 2 weeks had a DLQI of 0 or 1, as did 54% on monthly dosing and 1.5% of controls.
The ixekizumab-treated groups also showed what Dr. Gottlieb described as “dramatic” improvements – in the 4+ to 5+ point range – in both the mental and physical component scores on the SF-36, another widely used quality of life measure.
Improvements in skin and self-reported joint symptoms appeared to correlate. Patients with a PASI 50 to less-than PASI 75 response had a mean 17-point reduction in joint pain scores, while those with a PASI 75 to less-than PASI 90 response averaged a 25.1-point improvement in joint pain, patients with a PASI 90 to less-than PASI 100 skin response averaged a 27.6-point reduction, and PASI 100 responders had a mean 30.3-point reduction in joint pain scores.
“Obviously, one needs to look at this more carefully in a phase III psoriatic arthritis study. That’ll provide a more robust answer. But this gives a hint,” the dermatologist said.
The UNCOVER program was sponsored by Eli Lilly. Dr. Gottlieb serves as an adviser to Lilly and numerous other pharmaceutical companies.
COPENHAGEN – Ixekizumab not only shows considerable promise for the treatment of moderate to severe psoriasis, it looks like it may be a winner for comorbid psoriatic arthritis, too.
The investigational IgG4 humanized monoclonal antibody directed against interleukin-17A brought marked improvements in joint pain, systemic inflammatory burden, and quality of life as well as skin disease in patients with both psoriasis and self-reported psoriatic arthritis in a combined analysis of three phase III clinical trials, Dr. Alice B. Gottlieb reported at the annual congress of the European Academy of Dermatology and Venereology.
Of the 3,126 patients with moderate to severe psoriasis who participated in the 12-week, phase III UNCOVER-1, -2, and -3 trials, 751 (24%) also had self-reported psoriatic arthritis; her analysis focused on them.
She was quick to note that the UNCOVER trials were primarily psoriasis studies that relied upon patient self-report of psoriatic arthritis. Nevertheless, it seems likely that the great majority of self-reported psoriatic arthritis patients really did have the rheumatologic disease, since the mean baseline C-reactive protein (CRP) level in that group was 8.43 mg/L, a level far higher than expected in patients with psoriasis only.
In any case, more-rigorous phase III studies of ixekizumab conducted specifically in patients with formally rheumatologist-diagnosed psoriatic arthritis and treated in rheumatology practices are due to be presented at the annual meeting of the American College of Rheumatology in November. And while Dr. Gottlieb wasn’t at liberty to discuss those results, she did hint that the data will be strongly positive.
“If you’re happy about these UNCOVER findings, you’ll be ecstatic about those,” predicted Dr. Gottlieb, professor of dermatology and dermatologist in chief at Tufts Medical Center, Boston.
Also coming up at the American College of Rheumatology meeting will be the results of the first-ever head-to-head comparison of an IL-17 inhibitor versus a tumor necrosis factor–alpha blocker in psoriatic arthritis patients. While at present most physicians consider a TNF inhibitor to be the treatment of choice in patients with psoriatic arthritis, that view may change as a result of the forthcoming comparative study, according to the dermatologist.
In each of the three phase III UNCOVER studies, patients were randomized to 12 weeks of subcutaneous ixekizumab at 80 mg every 2 or 4 weeks following a 160-mg loading dose, or to placebo. At baseline, the subgroup with self-reported psoriatic arthritis had a mean Psoriasis Area and Severity Index ( PASI) 0f about 21, a self-rated joint pain severity of 50 on a 0-100 scale, a CRP of 8.43 mg/L, and a Dermatology Life Quality Index (DLQI) score of 14.
Joint pain decreased dramatically in the two ixekizumab groups as early at 2 weeks into the trial, at which point, patients on treatment every 2 weeks averaged a 13.1-point reduction from baseline, with a similar 14.1-point drop noted in those on an every 4 weeks schedule. At week 12, the mean reductions from baseline were 25.2 and 26.8 points, compared with a 1.1-point increase in joint pain among placebo-treated controls.
Inflammatory burden plunged quickly, as evidenced by mean reductions in CRP of 4.63 mg/L and 4.33 mg/L at week 1 with biweekly and monthly dosing, respectively. These reductions were then maintained through week 12.
In terms of improvement in skin symptoms, with ixekizumab dosed every 2 weeks, the PASI 75 response was 89.8% at 12 weeks, the PASI 90 response was 69.3%, and the PASI 100 response (clear skin) was 37.1%. In patients treated every 4 weeks, the rates were 81.1%, 60.8%, and 34.7%.
“There’s good news in both groups, but I think the news is even better in the every-2-weeks group,” Dr. Gottlieb commented.
The mean 12-week decrease from baseline in DLQI was 11.8 points with biweekly dosing and 10.5 with 4-week dosing, compared with 0.8 points in controls. That’s impressive given that a 5-point reduction in DLQI is deemed clinically meaningful, the dermatologist observed. At 12 weeks, 56.5% of patients on ixekizumab every 2 weeks had a DLQI of 0 or 1, as did 54% on monthly dosing and 1.5% of controls.
The ixekizumab-treated groups also showed what Dr. Gottlieb described as “dramatic” improvements – in the 4+ to 5+ point range – in both the mental and physical component scores on the SF-36, another widely used quality of life measure.
Improvements in skin and self-reported joint symptoms appeared to correlate. Patients with a PASI 50 to less-than PASI 75 response had a mean 17-point reduction in joint pain scores, while those with a PASI 75 to less-than PASI 90 response averaged a 25.1-point improvement in joint pain, patients with a PASI 90 to less-than PASI 100 skin response averaged a 27.6-point reduction, and PASI 100 responders had a mean 30.3-point reduction in joint pain scores.
“Obviously, one needs to look at this more carefully in a phase III psoriatic arthritis study. That’ll provide a more robust answer. But this gives a hint,” the dermatologist said.
The UNCOVER program was sponsored by Eli Lilly. Dr. Gottlieb serves as an adviser to Lilly and numerous other pharmaceutical companies.
COPENHAGEN – Ixekizumab not only shows considerable promise for the treatment of moderate to severe psoriasis, it looks like it may be a winner for comorbid psoriatic arthritis, too.
The investigational IgG4 humanized monoclonal antibody directed against interleukin-17A brought marked improvements in joint pain, systemic inflammatory burden, and quality of life as well as skin disease in patients with both psoriasis and self-reported psoriatic arthritis in a combined analysis of three phase III clinical trials, Dr. Alice B. Gottlieb reported at the annual congress of the European Academy of Dermatology and Venereology.
Of the 3,126 patients with moderate to severe psoriasis who participated in the 12-week, phase III UNCOVER-1, -2, and -3 trials, 751 (24%) also had self-reported psoriatic arthritis; her analysis focused on them.
She was quick to note that the UNCOVER trials were primarily psoriasis studies that relied upon patient self-report of psoriatic arthritis. Nevertheless, it seems likely that the great majority of self-reported psoriatic arthritis patients really did have the rheumatologic disease, since the mean baseline C-reactive protein (CRP) level in that group was 8.43 mg/L, a level far higher than expected in patients with psoriasis only.
In any case, more-rigorous phase III studies of ixekizumab conducted specifically in patients with formally rheumatologist-diagnosed psoriatic arthritis and treated in rheumatology practices are due to be presented at the annual meeting of the American College of Rheumatology in November. And while Dr. Gottlieb wasn’t at liberty to discuss those results, she did hint that the data will be strongly positive.
“If you’re happy about these UNCOVER findings, you’ll be ecstatic about those,” predicted Dr. Gottlieb, professor of dermatology and dermatologist in chief at Tufts Medical Center, Boston.
Also coming up at the American College of Rheumatology meeting will be the results of the first-ever head-to-head comparison of an IL-17 inhibitor versus a tumor necrosis factor–alpha blocker in psoriatic arthritis patients. While at present most physicians consider a TNF inhibitor to be the treatment of choice in patients with psoriatic arthritis, that view may change as a result of the forthcoming comparative study, according to the dermatologist.
In each of the three phase III UNCOVER studies, patients were randomized to 12 weeks of subcutaneous ixekizumab at 80 mg every 2 or 4 weeks following a 160-mg loading dose, or to placebo. At baseline, the subgroup with self-reported psoriatic arthritis had a mean Psoriasis Area and Severity Index ( PASI) 0f about 21, a self-rated joint pain severity of 50 on a 0-100 scale, a CRP of 8.43 mg/L, and a Dermatology Life Quality Index (DLQI) score of 14.
Joint pain decreased dramatically in the two ixekizumab groups as early at 2 weeks into the trial, at which point, patients on treatment every 2 weeks averaged a 13.1-point reduction from baseline, with a similar 14.1-point drop noted in those on an every 4 weeks schedule. At week 12, the mean reductions from baseline were 25.2 and 26.8 points, compared with a 1.1-point increase in joint pain among placebo-treated controls.
Inflammatory burden plunged quickly, as evidenced by mean reductions in CRP of 4.63 mg/L and 4.33 mg/L at week 1 with biweekly and monthly dosing, respectively. These reductions were then maintained through week 12.
In terms of improvement in skin symptoms, with ixekizumab dosed every 2 weeks, the PASI 75 response was 89.8% at 12 weeks, the PASI 90 response was 69.3%, and the PASI 100 response (clear skin) was 37.1%. In patients treated every 4 weeks, the rates were 81.1%, 60.8%, and 34.7%.
“There’s good news in both groups, but I think the news is even better in the every-2-weeks group,” Dr. Gottlieb commented.
The mean 12-week decrease from baseline in DLQI was 11.8 points with biweekly dosing and 10.5 with 4-week dosing, compared with 0.8 points in controls. That’s impressive given that a 5-point reduction in DLQI is deemed clinically meaningful, the dermatologist observed. At 12 weeks, 56.5% of patients on ixekizumab every 2 weeks had a DLQI of 0 or 1, as did 54% on monthly dosing and 1.5% of controls.
The ixekizumab-treated groups also showed what Dr. Gottlieb described as “dramatic” improvements – in the 4+ to 5+ point range – in both the mental and physical component scores on the SF-36, another widely used quality of life measure.
Improvements in skin and self-reported joint symptoms appeared to correlate. Patients with a PASI 50 to less-than PASI 75 response had a mean 17-point reduction in joint pain scores, while those with a PASI 75 to less-than PASI 90 response averaged a 25.1-point improvement in joint pain, patients with a PASI 90 to less-than PASI 100 skin response averaged a 27.6-point reduction, and PASI 100 responders had a mean 30.3-point reduction in joint pain scores.
“Obviously, one needs to look at this more carefully in a phase III psoriatic arthritis study. That’ll provide a more robust answer. But this gives a hint,” the dermatologist said.
The UNCOVER program was sponsored by Eli Lilly. Dr. Gottlieb serves as an adviser to Lilly and numerous other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point: The IL-17A inhibitor ixekizumab appears to be dramatically effective in psoriatic arthritis as well as in psoriasis.
Major finding: Self-reported joint pain scores in psoriatic arthritis patients were cut in half after 12 weeks of ixekizumab while remaining unmoved in placebo-treated controls.
Data source: This was an analysis of treatment outcomes in 751 self-reported psoriatic arthritis patients who participated in three 12-week phase III randomized clinical trials of ixekizumab vs. placebo.
Disclosures: The UNCOVER clinical trials program was sponsored by Eli Lilly. The presenter serves as an adviser to Lilly and numerous other pharmaceutical companies.
EADV: Investigational biologic rocks psoriasis world
COPENHAGEN – An investigational biologic agent that selectively inhibits the p19 subunit of interleukin-23 was the talk of the 2015 EADV congress based upon its striking outperformance of ustekinumab in a head-to-head phase II study in psoriasis patients.
Not only did the investigational agent, known for now as BI 655066, achieve substantially higher rates of PASI 90 – clear or almost clear skin – and PASI 100, but it did so much faster and maintained those stellar results far longer off treatment than with ustekinumab (Stelara), Dr. Kim A. Papp reported at the annual congress of the European Academy of Dermatology and Venereology.
“These data make an irrefutable case for IL-23 as instrumental in the expression of psoriasis,” declared Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
“What these data show is that selective blockade of IL-23 p19 clearly provides a remarkable response, whether we measure it as PASI 75, PASI 90, or PASI 100, compared with ustekinumab, which provides a very robust response itself. And when we look at the safety profile – admittedly in a small number of patients and for a short period of time – we see there are no real differences in safety signals. Obviously, we’re very encouraged to move forward with long-term phase III investigations,” Dr. Papp added.
The multicenter, double-blind phase II study included 166 patients with moderate to severe plaque psoriasis who were randomized to one of four treatment arms: BI 655066 at 90 or 180 mg given subcutaneously at weeks 0, 4, and 16; weight-based ustekinumab at 45 or 90 mg given on the same schedule; or a single 18-mg dose of BI 655066. Dr. Papp presented the results at 36 weeks, fully 20 weeks after the final dose was given.
Significantly more patients taking the investigational agent achieved PASI targets than did those taking ustekinumab, a highly effective medication. Here are the eye-popping 36-week results:
• PASI 90: 81% with BI 655066 at 180 mg, 69% with the lower dose, 30% with ustekinumab, and 7% with a single 18-mg dose of BI 655066.
• PASI 100: 54% and 43% with high- and low-dose BI 655066, respectively, 15% with ustekinumab, and 0% with single-dose BI 655066.
• PASI 75: 93% and 88% with high- and low-dose BI 655066; 55% with ustekinumab; and 19% after single-dose BI 655066.
One of the most intriguing study findings, according to Dr. Papp, is that the slope of the PASI 100 response curve in BI 655066–treated patients was still rising at week 20, a month after the final dose.
“It suggests that if you were to continue treating these patients, you might anticipate an even higher response,” the dermatologist said.
Another notable finding was the accelerated speed of response to the investigational agent: 57 days after the first dose of BI 655066, 50% of patients had a PASI 90 response.
“That means from the time you initiate therapy, you can expect half of your patients to get to PASI 90 in less than 2 months time,” Dr. Papp observed.
In contrast, it took 117 days – basically twice as long – for half of the ustekinumab group to achieve PASI 90.
Turning to the durability of response, Dr. Papp noted that it took 169 days for half of patients in the ustekinumab group to lose their PASI 90 response after treatment stopped, compared with 225 days in the BI 655066 90-mg group. That endpoint was never reached in the BI 655066 180-mg group before the study’s end.
Audience reaction to the study results was “wow.”
“Obviously, the data are terrific. Your mother couldn’t have invented better data,” declared Dr. Alice B. Gottlieb, professor of dermatology and dermatologist in chief at Tufts Medical Center, Boston.
She posed a question: Why are the BI 655066 response rates so much higher and longer lasting than with other biologics, including ustekinumab, which inhibits both IL-23 and IL-12?
“I think one key factor in the durability of the response is the pathway,” Dr. Papp replied. “Exactly why blockade of IL-23 p19 leads to this durable clinical response is unclear, but we can posit that we’re depopulating the Th-17 cells. IL-23 is necessary for Th-17 cell survival. So if you block the water, you ain’t going to grow the grass. I’m speculating here, but I think we’re removing that clone of cells, and it’s only once we start to allow them to repopulate that we then see recurrence of the disease.”
BI 655066 is being developed by Boehringer Ingelheim. Dr. Papp serves as an investigator for and consultant to Boehringer Ingelheim and numerous other pharmaceutical companies.
COPENHAGEN – An investigational biologic agent that selectively inhibits the p19 subunit of interleukin-23 was the talk of the 2015 EADV congress based upon its striking outperformance of ustekinumab in a head-to-head phase II study in psoriasis patients.
Not only did the investigational agent, known for now as BI 655066, achieve substantially higher rates of PASI 90 – clear or almost clear skin – and PASI 100, but it did so much faster and maintained those stellar results far longer off treatment than with ustekinumab (Stelara), Dr. Kim A. Papp reported at the annual congress of the European Academy of Dermatology and Venereology.
“These data make an irrefutable case for IL-23 as instrumental in the expression of psoriasis,” declared Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
“What these data show is that selective blockade of IL-23 p19 clearly provides a remarkable response, whether we measure it as PASI 75, PASI 90, or PASI 100, compared with ustekinumab, which provides a very robust response itself. And when we look at the safety profile – admittedly in a small number of patients and for a short period of time – we see there are no real differences in safety signals. Obviously, we’re very encouraged to move forward with long-term phase III investigations,” Dr. Papp added.
The multicenter, double-blind phase II study included 166 patients with moderate to severe plaque psoriasis who were randomized to one of four treatment arms: BI 655066 at 90 or 180 mg given subcutaneously at weeks 0, 4, and 16; weight-based ustekinumab at 45 or 90 mg given on the same schedule; or a single 18-mg dose of BI 655066. Dr. Papp presented the results at 36 weeks, fully 20 weeks after the final dose was given.
Significantly more patients taking the investigational agent achieved PASI targets than did those taking ustekinumab, a highly effective medication. Here are the eye-popping 36-week results:
• PASI 90: 81% with BI 655066 at 180 mg, 69% with the lower dose, 30% with ustekinumab, and 7% with a single 18-mg dose of BI 655066.
• PASI 100: 54% and 43% with high- and low-dose BI 655066, respectively, 15% with ustekinumab, and 0% with single-dose BI 655066.
• PASI 75: 93% and 88% with high- and low-dose BI 655066; 55% with ustekinumab; and 19% after single-dose BI 655066.
One of the most intriguing study findings, according to Dr. Papp, is that the slope of the PASI 100 response curve in BI 655066–treated patients was still rising at week 20, a month after the final dose.
“It suggests that if you were to continue treating these patients, you might anticipate an even higher response,” the dermatologist said.
Another notable finding was the accelerated speed of response to the investigational agent: 57 days after the first dose of BI 655066, 50% of patients had a PASI 90 response.
“That means from the time you initiate therapy, you can expect half of your patients to get to PASI 90 in less than 2 months time,” Dr. Papp observed.
In contrast, it took 117 days – basically twice as long – for half of the ustekinumab group to achieve PASI 90.
Turning to the durability of response, Dr. Papp noted that it took 169 days for half of patients in the ustekinumab group to lose their PASI 90 response after treatment stopped, compared with 225 days in the BI 655066 90-mg group. That endpoint was never reached in the BI 655066 180-mg group before the study’s end.
Audience reaction to the study results was “wow.”
“Obviously, the data are terrific. Your mother couldn’t have invented better data,” declared Dr. Alice B. Gottlieb, professor of dermatology and dermatologist in chief at Tufts Medical Center, Boston.
She posed a question: Why are the BI 655066 response rates so much higher and longer lasting than with other biologics, including ustekinumab, which inhibits both IL-23 and IL-12?
“I think one key factor in the durability of the response is the pathway,” Dr. Papp replied. “Exactly why blockade of IL-23 p19 leads to this durable clinical response is unclear, but we can posit that we’re depopulating the Th-17 cells. IL-23 is necessary for Th-17 cell survival. So if you block the water, you ain’t going to grow the grass. I’m speculating here, but I think we’re removing that clone of cells, and it’s only once we start to allow them to repopulate that we then see recurrence of the disease.”
BI 655066 is being developed by Boehringer Ingelheim. Dr. Papp serves as an investigator for and consultant to Boehringer Ingelheim and numerous other pharmaceutical companies.
COPENHAGEN – An investigational biologic agent that selectively inhibits the p19 subunit of interleukin-23 was the talk of the 2015 EADV congress based upon its striking outperformance of ustekinumab in a head-to-head phase II study in psoriasis patients.
Not only did the investigational agent, known for now as BI 655066, achieve substantially higher rates of PASI 90 – clear or almost clear skin – and PASI 100, but it did so much faster and maintained those stellar results far longer off treatment than with ustekinumab (Stelara), Dr. Kim A. Papp reported at the annual congress of the European Academy of Dermatology and Venereology.
“These data make an irrefutable case for IL-23 as instrumental in the expression of psoriasis,” declared Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
“What these data show is that selective blockade of IL-23 p19 clearly provides a remarkable response, whether we measure it as PASI 75, PASI 90, or PASI 100, compared with ustekinumab, which provides a very robust response itself. And when we look at the safety profile – admittedly in a small number of patients and for a short period of time – we see there are no real differences in safety signals. Obviously, we’re very encouraged to move forward with long-term phase III investigations,” Dr. Papp added.
The multicenter, double-blind phase II study included 166 patients with moderate to severe plaque psoriasis who were randomized to one of four treatment arms: BI 655066 at 90 or 180 mg given subcutaneously at weeks 0, 4, and 16; weight-based ustekinumab at 45 or 90 mg given on the same schedule; or a single 18-mg dose of BI 655066. Dr. Papp presented the results at 36 weeks, fully 20 weeks after the final dose was given.
Significantly more patients taking the investigational agent achieved PASI targets than did those taking ustekinumab, a highly effective medication. Here are the eye-popping 36-week results:
• PASI 90: 81% with BI 655066 at 180 mg, 69% with the lower dose, 30% with ustekinumab, and 7% with a single 18-mg dose of BI 655066.
• PASI 100: 54% and 43% with high- and low-dose BI 655066, respectively, 15% with ustekinumab, and 0% with single-dose BI 655066.
• PASI 75: 93% and 88% with high- and low-dose BI 655066; 55% with ustekinumab; and 19% after single-dose BI 655066.
One of the most intriguing study findings, according to Dr. Papp, is that the slope of the PASI 100 response curve in BI 655066–treated patients was still rising at week 20, a month after the final dose.
“It suggests that if you were to continue treating these patients, you might anticipate an even higher response,” the dermatologist said.
Another notable finding was the accelerated speed of response to the investigational agent: 57 days after the first dose of BI 655066, 50% of patients had a PASI 90 response.
“That means from the time you initiate therapy, you can expect half of your patients to get to PASI 90 in less than 2 months time,” Dr. Papp observed.
In contrast, it took 117 days – basically twice as long – for half of the ustekinumab group to achieve PASI 90.
Turning to the durability of response, Dr. Papp noted that it took 169 days for half of patients in the ustekinumab group to lose their PASI 90 response after treatment stopped, compared with 225 days in the BI 655066 90-mg group. That endpoint was never reached in the BI 655066 180-mg group before the study’s end.
Audience reaction to the study results was “wow.”
“Obviously, the data are terrific. Your mother couldn’t have invented better data,” declared Dr. Alice B. Gottlieb, professor of dermatology and dermatologist in chief at Tufts Medical Center, Boston.
She posed a question: Why are the BI 655066 response rates so much higher and longer lasting than with other biologics, including ustekinumab, which inhibits both IL-23 and IL-12?
“I think one key factor in the durability of the response is the pathway,” Dr. Papp replied. “Exactly why blockade of IL-23 p19 leads to this durable clinical response is unclear, but we can posit that we’re depopulating the Th-17 cells. IL-23 is necessary for Th-17 cell survival. So if you block the water, you ain’t going to grow the grass. I’m speculating here, but I think we’re removing that clone of cells, and it’s only once we start to allow them to repopulate that we then see recurrence of the disease.”
BI 655066 is being developed by Boehringer Ingelheim. Dr. Papp serves as an investigator for and consultant to Boehringer Ingelheim and numerous other pharmaceutical companies.
AT THE EADV CONGRESS
Key clinical point: An investigational inhibitor of the p19 subunit of interleukin-23 is generating efficacy results never before seen in psoriasis.
Major finding: The PASI 90 response rate was 81% fully 20 weeks after the third and final dose of BI 655066 at 180 mg, compared with 30% for ustekinumab.
Data source: This prospective, multicenter, double-blind study included 166 patients with moderate to severe plaque psoriasis who were randomized to various doses of ustekinumab or BI 655066 and followed for 36 weeks.
Disclosures: The study was sponsored by Boehringer Ingelheim. The presenter serves as an investigator for and consultant to Boehringer Ingelheim and numerous other pharmaceutical companies.
Infections from endemic fungi, mycobacteria rare in patients on TNFIs
The development of infections from mycobacteria and fungi endemic to U.S. regions in patients taking tumor necrosis factor–alpha inhibitors (TNFIs) is rare and is not influenced by prescreening of targeted infections, research suggests.
A case-control study of 30,772 patients taking TNFIs showed that only 158 (0.51%) patients developed the fungal and/or mycobacterial infections targeted in this study, with tuberculosis and histoplasmosis being the most common infections.
Targeted infections were nontuberculous mycobacterial infection, blastomycosis, coccidioidomyocosis, cryptococcal infection, histoplasmosis, pneumocystosis, tuberculosis disease, and unspecified fungal infection.
Prednisone was the only predictive factor for infection and was associated with a twofold increase in the likelihood of patients seeking medical attention for a fungal or mycobacterial infection, which the authors said was supported by previous research, according to a paper published online in Arthritis & Rheumatology.
“Thus, the question remains if the increased infection rates are related solely to the use of the glucocorticoids or the active disease for which the medication is being prescribed,” wrote Elizabeth Salt, Ph.D., of the University of Kentucky, Lexington, and coauthors (Arthritis Rheumatol. 2015 Oct 16 doi: 10.1002/art.39462).
Researchers also noted that sulfamethoxazole-trimethoprim was associated with a nonsignificant 45% increase in the likelihood of requiring medical care, compared with controls.
“It is possible that providers recognized the infectious risk of this population and made attempts at controlling infectious processes among those most vulnerable.”
The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
The development of infections from mycobacteria and fungi endemic to U.S. regions in patients taking tumor necrosis factor–alpha inhibitors (TNFIs) is rare and is not influenced by prescreening of targeted infections, research suggests.
A case-control study of 30,772 patients taking TNFIs showed that only 158 (0.51%) patients developed the fungal and/or mycobacterial infections targeted in this study, with tuberculosis and histoplasmosis being the most common infections.
Targeted infections were nontuberculous mycobacterial infection, blastomycosis, coccidioidomyocosis, cryptococcal infection, histoplasmosis, pneumocystosis, tuberculosis disease, and unspecified fungal infection.
Prednisone was the only predictive factor for infection and was associated with a twofold increase in the likelihood of patients seeking medical attention for a fungal or mycobacterial infection, which the authors said was supported by previous research, according to a paper published online in Arthritis & Rheumatology.
“Thus, the question remains if the increased infection rates are related solely to the use of the glucocorticoids or the active disease for which the medication is being prescribed,” wrote Elizabeth Salt, Ph.D., of the University of Kentucky, Lexington, and coauthors (Arthritis Rheumatol. 2015 Oct 16 doi: 10.1002/art.39462).
Researchers also noted that sulfamethoxazole-trimethoprim was associated with a nonsignificant 45% increase in the likelihood of requiring medical care, compared with controls.
“It is possible that providers recognized the infectious risk of this population and made attempts at controlling infectious processes among those most vulnerable.”
The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
The development of infections from mycobacteria and fungi endemic to U.S. regions in patients taking tumor necrosis factor–alpha inhibitors (TNFIs) is rare and is not influenced by prescreening of targeted infections, research suggests.
A case-control study of 30,772 patients taking TNFIs showed that only 158 (0.51%) patients developed the fungal and/or mycobacterial infections targeted in this study, with tuberculosis and histoplasmosis being the most common infections.
Targeted infections were nontuberculous mycobacterial infection, blastomycosis, coccidioidomyocosis, cryptococcal infection, histoplasmosis, pneumocystosis, tuberculosis disease, and unspecified fungal infection.
Prednisone was the only predictive factor for infection and was associated with a twofold increase in the likelihood of patients seeking medical attention for a fungal or mycobacterial infection, which the authors said was supported by previous research, according to a paper published online in Arthritis & Rheumatology.
“Thus, the question remains if the increased infection rates are related solely to the use of the glucocorticoids or the active disease for which the medication is being prescribed,” wrote Elizabeth Salt, Ph.D., of the University of Kentucky, Lexington, and coauthors (Arthritis Rheumatol. 2015 Oct 16 doi: 10.1002/art.39462).
Researchers also noted that sulfamethoxazole-trimethoprim was associated with a nonsignificant 45% increase in the likelihood of requiring medical care, compared with controls.
“It is possible that providers recognized the infectious risk of this population and made attempts at controlling infectious processes among those most vulnerable.”
The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: The incidence of select mycobacterial and fungal infections in patients taking TNFIs is low.
Major finding: Only 0.51% of patients taking TNFIs developed the mycobacterial and fungal infections targeted in this study.
Data source: A case-control study of 30,772 patients taking TNFIs.
Disclosures: The study was supported by the National Institutes of Health. There were no conflicts of interest declared.
Topical calcipotriene-betamethasone combination approved for plaque psoriasis
A combination of calcipotriene and betamethasone dipropionate in a foam formulation has been approved by the Food and Drug Administration for the topical treatment of plaque psoriasis in adults, according to the manufacturer.
The product will be marketed as Enstilar (calcipotriene and betamethasone dipropionate) foam, 0.005%/0.064%, by Leo Pharma.
Each gram contains 52.2 mcg calcipotriene hydrate (equivalent to 50 mcg of calcipotriene) and 0.643 mg of betamethasone dipropionate (equivalent to 0.5 mg of betamethasone); it is applied to affected areas once a day for up to 4 weeks, according to the prescribing information, which states that patient should not use more than 60 g every 4 days.
The vitamin D analog-corticosteroid combination product was compared with treatment with the two individual components and/or a vehicle only, in more than 700 patients with plaque psoriasis in two studies. At baseline, about two-thirds of the patients had moderate disease, 14%-15% had mild disease, and 10% had moderately severe disease, based on a 5-point Investigator’s Global Assessment scale. The proportion of patients who were considered clear or almost clear at 4 weeks, based on IGA scale results, was the efficacy endpoint defining treatment success. Among patients with mild disease at baseline, treatment was considered successful if they were considered clear at 4 weeks.
In one study of 302 patients, 45% treated with the combination product had achieved treatment success at week 4 vs. 30.7% of those treated with betamethasone and 14.9% treated with calcipotriene, according to the prescribing information. In the second study, 323 patients were treated with the combination product and 103 received the vehicle. At 4 weeks, 53.3% of those treated with the foam met the efficacy endpoint vs. 4.8% of those in the vehicle group.
Application site irritation, application site pruritus, folliculitis, skin hypopigmentation, hypercalcemia, urticaria, and psoriasis exacerbations were among the adverse reactions reported in fewer than 1% of those treated with the combination product. The warnings and precautions section of the prescribing information includes a statement noting that the product contains propellants that are flammable, and that patients should be advised to “avoid fire, flame, or smoking during and immediately after using this product.”
This approval is for an adult population. As a postmarketing commitment, the FDA is requiring that the company conduct a study in pediatric patients with plaque psoriasis, according to the agency’s approval letter, dated Oct. 16. That study is described as an open-label study to assess the effect of the product on calcium metabolism in pediatric patients aged 12-16 years, with plaque psoriasis of the scalp and body. In a subgroup of these children, pharmacokinetics and hypothalamic-pituitary axis suppression also will be assessed, the letter says.
Serious adverse events associated with this product should be reported to the FDA’s MedWatch program at 800-332-1088, or http://www.fda.gov/Safety/MedWatch/default.htm.
A combination of calcipotriene and betamethasone dipropionate in a foam formulation has been approved by the Food and Drug Administration for the topical treatment of plaque psoriasis in adults, according to the manufacturer.
The product will be marketed as Enstilar (calcipotriene and betamethasone dipropionate) foam, 0.005%/0.064%, by Leo Pharma.
Each gram contains 52.2 mcg calcipotriene hydrate (equivalent to 50 mcg of calcipotriene) and 0.643 mg of betamethasone dipropionate (equivalent to 0.5 mg of betamethasone); it is applied to affected areas once a day for up to 4 weeks, according to the prescribing information, which states that patient should not use more than 60 g every 4 days.
The vitamin D analog-corticosteroid combination product was compared with treatment with the two individual components and/or a vehicle only, in more than 700 patients with plaque psoriasis in two studies. At baseline, about two-thirds of the patients had moderate disease, 14%-15% had mild disease, and 10% had moderately severe disease, based on a 5-point Investigator’s Global Assessment scale. The proportion of patients who were considered clear or almost clear at 4 weeks, based on IGA scale results, was the efficacy endpoint defining treatment success. Among patients with mild disease at baseline, treatment was considered successful if they were considered clear at 4 weeks.
In one study of 302 patients, 45% treated with the combination product had achieved treatment success at week 4 vs. 30.7% of those treated with betamethasone and 14.9% treated with calcipotriene, according to the prescribing information. In the second study, 323 patients were treated with the combination product and 103 received the vehicle. At 4 weeks, 53.3% of those treated with the foam met the efficacy endpoint vs. 4.8% of those in the vehicle group.
Application site irritation, application site pruritus, folliculitis, skin hypopigmentation, hypercalcemia, urticaria, and psoriasis exacerbations were among the adverse reactions reported in fewer than 1% of those treated with the combination product. The warnings and precautions section of the prescribing information includes a statement noting that the product contains propellants that are flammable, and that patients should be advised to “avoid fire, flame, or smoking during and immediately after using this product.”
This approval is for an adult population. As a postmarketing commitment, the FDA is requiring that the company conduct a study in pediatric patients with plaque psoriasis, according to the agency’s approval letter, dated Oct. 16. That study is described as an open-label study to assess the effect of the product on calcium metabolism in pediatric patients aged 12-16 years, with plaque psoriasis of the scalp and body. In a subgroup of these children, pharmacokinetics and hypothalamic-pituitary axis suppression also will be assessed, the letter says.
Serious adverse events associated with this product should be reported to the FDA’s MedWatch program at 800-332-1088, or http://www.fda.gov/Safety/MedWatch/default.htm.
A combination of calcipotriene and betamethasone dipropionate in a foam formulation has been approved by the Food and Drug Administration for the topical treatment of plaque psoriasis in adults, according to the manufacturer.
The product will be marketed as Enstilar (calcipotriene and betamethasone dipropionate) foam, 0.005%/0.064%, by Leo Pharma.
Each gram contains 52.2 mcg calcipotriene hydrate (equivalent to 50 mcg of calcipotriene) and 0.643 mg of betamethasone dipropionate (equivalent to 0.5 mg of betamethasone); it is applied to affected areas once a day for up to 4 weeks, according to the prescribing information, which states that patient should not use more than 60 g every 4 days.
The vitamin D analog-corticosteroid combination product was compared with treatment with the two individual components and/or a vehicle only, in more than 700 patients with plaque psoriasis in two studies. At baseline, about two-thirds of the patients had moderate disease, 14%-15% had mild disease, and 10% had moderately severe disease, based on a 5-point Investigator’s Global Assessment scale. The proportion of patients who were considered clear or almost clear at 4 weeks, based on IGA scale results, was the efficacy endpoint defining treatment success. Among patients with mild disease at baseline, treatment was considered successful if they were considered clear at 4 weeks.
In one study of 302 patients, 45% treated with the combination product had achieved treatment success at week 4 vs. 30.7% of those treated with betamethasone and 14.9% treated with calcipotriene, according to the prescribing information. In the second study, 323 patients were treated with the combination product and 103 received the vehicle. At 4 weeks, 53.3% of those treated with the foam met the efficacy endpoint vs. 4.8% of those in the vehicle group.
Application site irritation, application site pruritus, folliculitis, skin hypopigmentation, hypercalcemia, urticaria, and psoriasis exacerbations were among the adverse reactions reported in fewer than 1% of those treated with the combination product. The warnings and precautions section of the prescribing information includes a statement noting that the product contains propellants that are flammable, and that patients should be advised to “avoid fire, flame, or smoking during and immediately after using this product.”
This approval is for an adult population. As a postmarketing commitment, the FDA is requiring that the company conduct a study in pediatric patients with plaque psoriasis, according to the agency’s approval letter, dated Oct. 16. That study is described as an open-label study to assess the effect of the product on calcium metabolism in pediatric patients aged 12-16 years, with plaque psoriasis of the scalp and body. In a subgroup of these children, pharmacokinetics and hypothalamic-pituitary axis suppression also will be assessed, the letter says.
Serious adverse events associated with this product should be reported to the FDA’s MedWatch program at 800-332-1088, or http://www.fda.gov/Safety/MedWatch/default.htm.














