User login
Psoriasis cohort reveals high arthritis risk
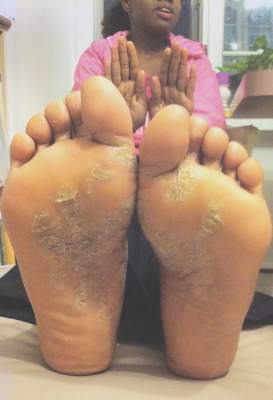
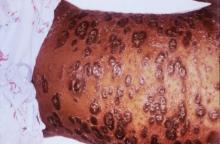
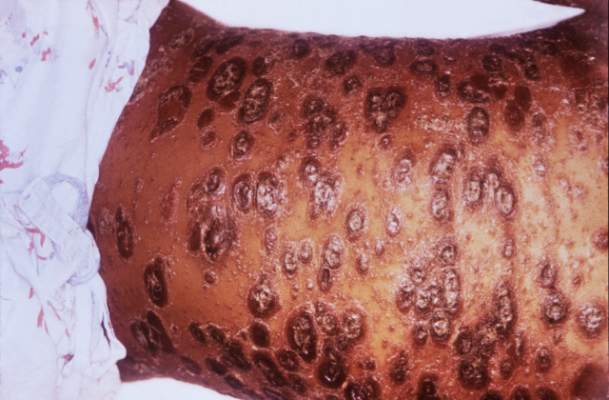
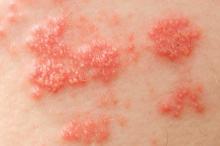
Psoriatic arthritis may occur more frequently among people with psoriasis than previously reported, and risk factors include having severe psoriasis, nail pitting, low education levels, and uveitis, according to findings from a Canadian cohort study.
Beginning in 2006, Dr. Lihi Eder of the University of Toronto and coinvestigators recruited 464 patients (mean age 47, 56% male, 77% white) mainly from phototherapy and dermatology outpatient clinics in Toronto, and followed them 8 years. All had psoriasis of varying type and severity at baseline, but not inflammatory arthritis or spondylitis (Arthritis Rheumatol. 2015 Nov 10 doi: 10.1002/art.39494).
During the 8-year follow-up, 51 patients developed rheumatologist-confirmed psoriatic arthritis (PsA). Dr. Eder and colleagues reported an annual incidence rate of 2.7 confirmed cases of psoriatic arthritis per 100 psoriasis patients per year, which is considerably higher than previous published estimates, the investigators noted. The independent predictors of confirmed psoriatic arthritis were severe psoriasis (relative risk, 5.4; P = .006), not finishing high school (vs. finishing college RR, 4.5, P = .005; and vs. finishing high school RR, 3.3; P = .049), and use of systemic retinoids (RR, 3.4; P = .02). Time-dependent predictive variables included psoriatic nail pitting (RR, 2.5; P = .002) and uveitis (RR, 31.5; P = .001). Disease severity and nail pitting have been found in previous studies to be associated with a higher risk of psoriatic arthritis.
This study confirmed this association and also identified low education levels and uveitis as predictors. Low education is a marker of socioeconomic status that has been associated with lifestyle habits and possibly occupations that may increase PsA risk, the study authors noted, but the link requires further investigation. The authors cautioned that only three uveitis cases occurred in the cohort and that confidence intervals were wide. They also noted as a limitation that most participants were recruited from dermatology clinics, leading to overrepresentation of moderate-severe psoriasis and possibly patients with longer disease duration. Nevertheless, it “is likely that the true incidence of PsA in patients with psoriasis, particularly those attending dermatology clinics, is higher than previously reported,” the investigators wrote. “This highlights the role of dermatologists as key players in identifying psoriasis patients who are at higher risk of developing PsA.”
Krembil Foundation, the Canadian Institutes of Health Research, and The Arthritis Society supported the study.
Psoriatic arthritis may occur more frequently among people with psoriasis than previously reported, and risk factors include having severe psoriasis, nail pitting, low education levels, and uveitis, according to findings from a Canadian cohort study.
Beginning in 2006, Dr. Lihi Eder of the University of Toronto and coinvestigators recruited 464 patients (mean age 47, 56% male, 77% white) mainly from phototherapy and dermatology outpatient clinics in Toronto, and followed them 8 years. All had psoriasis of varying type and severity at baseline, but not inflammatory arthritis or spondylitis (Arthritis Rheumatol. 2015 Nov 10 doi: 10.1002/art.39494).
During the 8-year follow-up, 51 patients developed rheumatologist-confirmed psoriatic arthritis (PsA). Dr. Eder and colleagues reported an annual incidence rate of 2.7 confirmed cases of psoriatic arthritis per 100 psoriasis patients per year, which is considerably higher than previous published estimates, the investigators noted. The independent predictors of confirmed psoriatic arthritis were severe psoriasis (relative risk, 5.4; P = .006), not finishing high school (vs. finishing college RR, 4.5, P = .005; and vs. finishing high school RR, 3.3; P = .049), and use of systemic retinoids (RR, 3.4; P = .02). Time-dependent predictive variables included psoriatic nail pitting (RR, 2.5; P = .002) and uveitis (RR, 31.5; P = .001). Disease severity and nail pitting have been found in previous studies to be associated with a higher risk of psoriatic arthritis.
This study confirmed this association and also identified low education levels and uveitis as predictors. Low education is a marker of socioeconomic status that has been associated with lifestyle habits and possibly occupations that may increase PsA risk, the study authors noted, but the link requires further investigation. The authors cautioned that only three uveitis cases occurred in the cohort and that confidence intervals were wide. They also noted as a limitation that most participants were recruited from dermatology clinics, leading to overrepresentation of moderate-severe psoriasis and possibly patients with longer disease duration. Nevertheless, it “is likely that the true incidence of PsA in patients with psoriasis, particularly those attending dermatology clinics, is higher than previously reported,” the investigators wrote. “This highlights the role of dermatologists as key players in identifying psoriasis patients who are at higher risk of developing PsA.”
Krembil Foundation, the Canadian Institutes of Health Research, and The Arthritis Society supported the study.
Psoriatic arthritis may occur more frequently among people with psoriasis than previously reported, and risk factors include having severe psoriasis, nail pitting, low education levels, and uveitis, according to findings from a Canadian cohort study.
Beginning in 2006, Dr. Lihi Eder of the University of Toronto and coinvestigators recruited 464 patients (mean age 47, 56% male, 77% white) mainly from phototherapy and dermatology outpatient clinics in Toronto, and followed them 8 years. All had psoriasis of varying type and severity at baseline, but not inflammatory arthritis or spondylitis (Arthritis Rheumatol. 2015 Nov 10 doi: 10.1002/art.39494).
During the 8-year follow-up, 51 patients developed rheumatologist-confirmed psoriatic arthritis (PsA). Dr. Eder and colleagues reported an annual incidence rate of 2.7 confirmed cases of psoriatic arthritis per 100 psoriasis patients per year, which is considerably higher than previous published estimates, the investigators noted. The independent predictors of confirmed psoriatic arthritis were severe psoriasis (relative risk, 5.4; P = .006), not finishing high school (vs. finishing college RR, 4.5, P = .005; and vs. finishing high school RR, 3.3; P = .049), and use of systemic retinoids (RR, 3.4; P = .02). Time-dependent predictive variables included psoriatic nail pitting (RR, 2.5; P = .002) and uveitis (RR, 31.5; P = .001). Disease severity and nail pitting have been found in previous studies to be associated with a higher risk of psoriatic arthritis.
This study confirmed this association and also identified low education levels and uveitis as predictors. Low education is a marker of socioeconomic status that has been associated with lifestyle habits and possibly occupations that may increase PsA risk, the study authors noted, but the link requires further investigation. The authors cautioned that only three uveitis cases occurred in the cohort and that confidence intervals were wide. They also noted as a limitation that most participants were recruited from dermatology clinics, leading to overrepresentation of moderate-severe psoriasis and possibly patients with longer disease duration. Nevertheless, it “is likely that the true incidence of PsA in patients with psoriasis, particularly those attending dermatology clinics, is higher than previously reported,” the investigators wrote. “This highlights the role of dermatologists as key players in identifying psoriasis patients who are at higher risk of developing PsA.”
Krembil Foundation, the Canadian Institutes of Health Research, and The Arthritis Society supported the study.
FROM ARTHRITIS & RHEUMATOLOGY
Key clinical point: Incidence of psoriatic arthritis is higher among psoriasis patients than previously estimated.
Major finding: Annual incidence rate was 2.7 (95% confidence interval 2.1, 3.6) PsA cases per 100 psoriasis patients; significant predictors included disease severity, nail pitting, low education, and uveitis.
Data source: A prospective cohort study of 464 psoriasis patients without arthritis at baseline, followed for 8 years.
Disclosures: Krembil Foundation, Canadian Institutes of Health Research, and The Arthritis Society sponsored the study.
EADV: New long-term data on biologics for pediatric psoriasis ‘encouraging’
COPENHAGEN – The longest-ever clinical trials of etanercept and adalimumab for pediatric psoriasis show reassuring maintenance of efficacy, coupled with safety and tolerability profiles similar to what has been seen in long-term trials in adults, investigators reported at the annual congress of the European Academy of Dermatology and Venereology.
Dr. Richard G. Langley presented outcomes from a 5-year open-label extension of an initial 12-week, multicenter, double-blind, randomized trial of etanercept or placebo in children and adolescents with moderate to severe chronic plaque psoriasis. The primary results were published more than 7 years ago (N Engl J Med. 2008 Jan 17;358[3]:241-51).
“This is the largest and longest follow-up of any biologic in children and adolescents to date. I think it’s encouraging data and should be reassuring to those of us who are managing this important population of pediatric patients in our conversations with parents and families,”said Dr. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
In the original 211-patient study, 57% of patients receiving etanercept (Enbrel) once weekly at 0.8 mg/kg to a maximum of 50 mg achieved a PASI 75 response at week 12, and 27% had a PASI 90. In the 69 patients who completed the full 264 weeks of follow-up, those response rates remained essentially unchanged.
Dr. Langley was quick to point out that this was an as-observed analysis, meaning results were counted only in those patients still participating at week 264. While conceding that the two-thirds dropout rate is an important study limitation, he added: “Notwithstanding that, what matters to us in the clinic are the patients we continue to treat and how they’re responding.”
Of note, most study discontinuations didn’t result from loss of response or adverse events, they were due to withdrawal of consent by families in which the patient began the study as a cooperative child and who as time went by turned into an independent and often willful teenager, he said.
No new safety signals arose over the course of 5 years. There were no opportunistic infections, no malignancies. The most common adverse events were the same ones seen in the original short-term study: upper respiratory infections, nasopharyngitis, and headaches. There was only one serious adverse event deemed by investigators as ‘possibly related’ to etanercept therapy: a case of cellulitis.
Separately, Dr. Diamant Thaci and Dr. Kim A. Papp presented 52-week outcomes for different aspects of the pivotal phase III randomized trial which earlier in 2015 earned adalimumab (Humira) European Commission marketing approval as the first biologic agent indicated for treatment of children as young as age 4 years, as well as for adolescents. The multi-arm trial included 114 patients aged 4-17 years with moderate to severe plaque psoriasis.
Dr. Thaci reported on the 37 subjects initially randomized double-blind to 16 weeks of oral methotrexate at 0.1-0.4 mg/kg weekly. The 19 patients (51%) who were deemed nonresponders to methotrexate because of inadequate PASI response at week 16 were then switched to open-label adalimumab at 0.8 mg/kg every other week for the remainder of the 52 weeks.
After 16 weeks on adalimumab, the methotrexate nonresponders had a PASI 75 of 90% and a PASI 90 of 74%, and 79% of the subjects were rated clear or almost clear by Physician’s Global Assessment. At week 52, the PASI 75 rate was 79%, the PASI 90 rate was 58%, and 68% of patients were rated clear or almost clear, according to Dr. Thaci of University Hospital Schleswig-Holstein, in L<scaps>ü</scaps>beck, Germany.
The side effects of methotrexate and adalimumab were similar to those seen in adults. There were no serious adverse events. The infections that occurred during adalimumab therapy were “very banal things,” mostly nasopharyngitis and upper respiratory tract infections, the dermatologist said. There were no opportunistic infections, malignancies, or cases of tuberculosis during the phase III study.
This was an important analysis because it recapitulates daily clinical practice, he explained. In most of the world, when dermatologists deem it time for systemic therapy, they generally start out with methotrexate, reserving biologics for second-line therapy because of the cost.
Dr. Papp reported on 39 patients on adalimumab at 0.4 mg/kg every other week, and 38 on 0.8 mg/kg every other week throughout the 52-week study. The response rate was essentially a flat line from week 16 to week 52, with PASI 75s of 44% at week 16 and 50% at week 52 in the low-dose group, and 58% and 56% in the high-dose group. Half of patients on 0.4 mg/kg were clear or almost clear at week 52, as were 56% on 0.8 mg/kg.
There was a nonsignificant trend for an increasing infection rate with greater exposure to adalimumab, which will require evaluation during the ongoing follow-up beyond 52 weeks, said Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
“I think what’s gratifying about this is we see that these children actually have a robust response and that response is maintained over a full year of treatment, which is reassuring because that reflects what we’ve seen in the adult population as well,” he said.
Dr. Papp and Dr. Thaci receive research funding and serve as scientific advisers to AbbVie, which sponsored the adalimumab trial. They also have ties to other pharmaceutical companies.
Dr. Langley has served as principal investigator for and is on the scientific advisory boards of Amgen, which sponsored the etanercept pediatric psoriasis study. He also has ties to other pharmaceutical companies.
COPENHAGEN – The longest-ever clinical trials of etanercept and adalimumab for pediatric psoriasis show reassuring maintenance of efficacy, coupled with safety and tolerability profiles similar to what has been seen in long-term trials in adults, investigators reported at the annual congress of the European Academy of Dermatology and Venereology.
Dr. Richard G. Langley presented outcomes from a 5-year open-label extension of an initial 12-week, multicenter, double-blind, randomized trial of etanercept or placebo in children and adolescents with moderate to severe chronic plaque psoriasis. The primary results were published more than 7 years ago (N Engl J Med. 2008 Jan 17;358[3]:241-51).
“This is the largest and longest follow-up of any biologic in children and adolescents to date. I think it’s encouraging data and should be reassuring to those of us who are managing this important population of pediatric patients in our conversations with parents and families,”said Dr. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
In the original 211-patient study, 57% of patients receiving etanercept (Enbrel) once weekly at 0.8 mg/kg to a maximum of 50 mg achieved a PASI 75 response at week 12, and 27% had a PASI 90. In the 69 patients who completed the full 264 weeks of follow-up, those response rates remained essentially unchanged.
Dr. Langley was quick to point out that this was an as-observed analysis, meaning results were counted only in those patients still participating at week 264. While conceding that the two-thirds dropout rate is an important study limitation, he added: “Notwithstanding that, what matters to us in the clinic are the patients we continue to treat and how they’re responding.”
Of note, most study discontinuations didn’t result from loss of response or adverse events, they were due to withdrawal of consent by families in which the patient began the study as a cooperative child and who as time went by turned into an independent and often willful teenager, he said.
No new safety signals arose over the course of 5 years. There were no opportunistic infections, no malignancies. The most common adverse events were the same ones seen in the original short-term study: upper respiratory infections, nasopharyngitis, and headaches. There was only one serious adverse event deemed by investigators as ‘possibly related’ to etanercept therapy: a case of cellulitis.
Separately, Dr. Diamant Thaci and Dr. Kim A. Papp presented 52-week outcomes for different aspects of the pivotal phase III randomized trial which earlier in 2015 earned adalimumab (Humira) European Commission marketing approval as the first biologic agent indicated for treatment of children as young as age 4 years, as well as for adolescents. The multi-arm trial included 114 patients aged 4-17 years with moderate to severe plaque psoriasis.
Dr. Thaci reported on the 37 subjects initially randomized double-blind to 16 weeks of oral methotrexate at 0.1-0.4 mg/kg weekly. The 19 patients (51%) who were deemed nonresponders to methotrexate because of inadequate PASI response at week 16 were then switched to open-label adalimumab at 0.8 mg/kg every other week for the remainder of the 52 weeks.
After 16 weeks on adalimumab, the methotrexate nonresponders had a PASI 75 of 90% and a PASI 90 of 74%, and 79% of the subjects were rated clear or almost clear by Physician’s Global Assessment. At week 52, the PASI 75 rate was 79%, the PASI 90 rate was 58%, and 68% of patients were rated clear or almost clear, according to Dr. Thaci of University Hospital Schleswig-Holstein, in L<scaps>ü</scaps>beck, Germany.
The side effects of methotrexate and adalimumab were similar to those seen in adults. There were no serious adverse events. The infections that occurred during adalimumab therapy were “very banal things,” mostly nasopharyngitis and upper respiratory tract infections, the dermatologist said. There were no opportunistic infections, malignancies, or cases of tuberculosis during the phase III study.
This was an important analysis because it recapitulates daily clinical practice, he explained. In most of the world, when dermatologists deem it time for systemic therapy, they generally start out with methotrexate, reserving biologics for second-line therapy because of the cost.
Dr. Papp reported on 39 patients on adalimumab at 0.4 mg/kg every other week, and 38 on 0.8 mg/kg every other week throughout the 52-week study. The response rate was essentially a flat line from week 16 to week 52, with PASI 75s of 44% at week 16 and 50% at week 52 in the low-dose group, and 58% and 56% in the high-dose group. Half of patients on 0.4 mg/kg were clear or almost clear at week 52, as were 56% on 0.8 mg/kg.
There was a nonsignificant trend for an increasing infection rate with greater exposure to adalimumab, which will require evaluation during the ongoing follow-up beyond 52 weeks, said Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
“I think what’s gratifying about this is we see that these children actually have a robust response and that response is maintained over a full year of treatment, which is reassuring because that reflects what we’ve seen in the adult population as well,” he said.
Dr. Papp and Dr. Thaci receive research funding and serve as scientific advisers to AbbVie, which sponsored the adalimumab trial. They also have ties to other pharmaceutical companies.
Dr. Langley has served as principal investigator for and is on the scientific advisory boards of Amgen, which sponsored the etanercept pediatric psoriasis study. He also has ties to other pharmaceutical companies.
COPENHAGEN – The longest-ever clinical trials of etanercept and adalimumab for pediatric psoriasis show reassuring maintenance of efficacy, coupled with safety and tolerability profiles similar to what has been seen in long-term trials in adults, investigators reported at the annual congress of the European Academy of Dermatology and Venereology.
Dr. Richard G. Langley presented outcomes from a 5-year open-label extension of an initial 12-week, multicenter, double-blind, randomized trial of etanercept or placebo in children and adolescents with moderate to severe chronic plaque psoriasis. The primary results were published more than 7 years ago (N Engl J Med. 2008 Jan 17;358[3]:241-51).
“This is the largest and longest follow-up of any biologic in children and adolescents to date. I think it’s encouraging data and should be reassuring to those of us who are managing this important population of pediatric patients in our conversations with parents and families,”said Dr. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
In the original 211-patient study, 57% of patients receiving etanercept (Enbrel) once weekly at 0.8 mg/kg to a maximum of 50 mg achieved a PASI 75 response at week 12, and 27% had a PASI 90. In the 69 patients who completed the full 264 weeks of follow-up, those response rates remained essentially unchanged.
Dr. Langley was quick to point out that this was an as-observed analysis, meaning results were counted only in those patients still participating at week 264. While conceding that the two-thirds dropout rate is an important study limitation, he added: “Notwithstanding that, what matters to us in the clinic are the patients we continue to treat and how they’re responding.”
Of note, most study discontinuations didn’t result from loss of response or adverse events, they were due to withdrawal of consent by families in which the patient began the study as a cooperative child and who as time went by turned into an independent and often willful teenager, he said.
No new safety signals arose over the course of 5 years. There were no opportunistic infections, no malignancies. The most common adverse events were the same ones seen in the original short-term study: upper respiratory infections, nasopharyngitis, and headaches. There was only one serious adverse event deemed by investigators as ‘possibly related’ to etanercept therapy: a case of cellulitis.
Separately, Dr. Diamant Thaci and Dr. Kim A. Papp presented 52-week outcomes for different aspects of the pivotal phase III randomized trial which earlier in 2015 earned adalimumab (Humira) European Commission marketing approval as the first biologic agent indicated for treatment of children as young as age 4 years, as well as for adolescents. The multi-arm trial included 114 patients aged 4-17 years with moderate to severe plaque psoriasis.
Dr. Thaci reported on the 37 subjects initially randomized double-blind to 16 weeks of oral methotrexate at 0.1-0.4 mg/kg weekly. The 19 patients (51%) who were deemed nonresponders to methotrexate because of inadequate PASI response at week 16 were then switched to open-label adalimumab at 0.8 mg/kg every other week for the remainder of the 52 weeks.
After 16 weeks on adalimumab, the methotrexate nonresponders had a PASI 75 of 90% and a PASI 90 of 74%, and 79% of the subjects were rated clear or almost clear by Physician’s Global Assessment. At week 52, the PASI 75 rate was 79%, the PASI 90 rate was 58%, and 68% of patients were rated clear or almost clear, according to Dr. Thaci of University Hospital Schleswig-Holstein, in L<scaps>ü</scaps>beck, Germany.
The side effects of methotrexate and adalimumab were similar to those seen in adults. There were no serious adverse events. The infections that occurred during adalimumab therapy were “very banal things,” mostly nasopharyngitis and upper respiratory tract infections, the dermatologist said. There were no opportunistic infections, malignancies, or cases of tuberculosis during the phase III study.
This was an important analysis because it recapitulates daily clinical practice, he explained. In most of the world, when dermatologists deem it time for systemic therapy, they generally start out with methotrexate, reserving biologics for second-line therapy because of the cost.
Dr. Papp reported on 39 patients on adalimumab at 0.4 mg/kg every other week, and 38 on 0.8 mg/kg every other week throughout the 52-week study. The response rate was essentially a flat line from week 16 to week 52, with PASI 75s of 44% at week 16 and 50% at week 52 in the low-dose group, and 58% and 56% in the high-dose group. Half of patients on 0.4 mg/kg were clear or almost clear at week 52, as were 56% on 0.8 mg/kg.
There was a nonsignificant trend for an increasing infection rate with greater exposure to adalimumab, which will require evaluation during the ongoing follow-up beyond 52 weeks, said Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
“I think what’s gratifying about this is we see that these children actually have a robust response and that response is maintained over a full year of treatment, which is reassuring because that reflects what we’ve seen in the adult population as well,” he said.
Dr. Papp and Dr. Thaci receive research funding and serve as scientific advisers to AbbVie, which sponsored the adalimumab trial. They also have ties to other pharmaceutical companies.
Dr. Langley has served as principal investigator for and is on the scientific advisory boards of Amgen, which sponsored the etanercept pediatric psoriasis study. He also has ties to other pharmaceutical companies.
EXPERT ANALYSIS FROM THE EADV CONGRESS
EADV: Pediatric psoriasis called ‘grossly undertreated’
COPENHAGEN – Children and adolescents with moderate to severe psoriasis are generally undertreated, with resultant potential psychological impairment, social stigmatization, and isolation, psoriasis experts agreed at the annual congress of the European Academy of Dermatology and Venereology.
In a session featuring new long-term safety and efficacy data from extended pediatric clinical trials of etanercept and adalimumab, prominent clinical trial investigators who presented the findings asserted that many physicians and parents are overly timid about the use of systemic treatment options in children and adolescents when such therapy is clearly warranted.
“The burden of psoriasis is particularly acute in the pediatric population. I always ask patients, ‘How is psoriasis affecting you?’ And with children, it’s affecting them and it’s also affecting their parents. If it’s affecting their self-esteem – if it’s ruining your life and you can’t control it with topical agents – I think it’s time to think of other things,” declared Dr. Richard G. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
Yet all too often that doesn’t happen, as documented in the recently reported physician portion of the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey, which included 391 dermatologists and 390 rheumatologists in North America and Western Europe (J Eur Acad Dermatol Venereol. 2015 Oct;29[10]:2002-10).
Dr. Langley was a coinvestigator in MAPP, which found that 39% of dermatologists reported treating their moderate to severe psoriasis patients with conventional oral and/or biologic agents (19.5% and 19.6%, respectively). A total of 75% indicated they prescribe topical medications as monotherapy for their patients with moderate to severe psoriasis. In children, that rate was even higher, he said, despite the fact that topical monotherapy is clearly inadequate for treatment of more severe disease.
“I think there’s a fear about using systemics among parents and among some practitioners, which was one of the top reasons in the MAPP survey that people didn’t get appropriate treatment,” he said.
That’s a shortsighted attitude, the dermatologist continued: “The risk is not just the risk of the drug, but the risk of untreated disease, because untreated disease devastates patients.”
MAPP was the largest-ever survey of physician and patient perspectives regarding psoriasis and psoriatic arthritis. The patient perspective, derived from interviews with nearly 3,500 patients, was published earlier (J Am Acad Dermatol. 2014 May;70[5]:871-81).
One impediment to more widespread use of systemic therapies for pediatric psoriasis is that it’s almost entirely off-label prescribing. In the United States, no conventional oral agents or biologics are Food and Drug Administration approved for use in psoriasis patients under age 18 years. That was the case in Europe as well until earlier this year, when adalimumab (Humira) received European marketing approval for children ages 4 years and up with severe chronic plaque psoriasis with an inadequate response to topical agents and phototherapy or in whom such therapies are contraindicated.
Dr. Kim A. Papp, who presented new 52-week safety and efficacy data from a phase III study of adalimumab in pediatric psoriasis patients, said that even when dermatologists utilize biologics in pediatric patients, the medications are typically underdosed. In the phase III pediatric adalimumab trial, for example, most participants were overweight or obese, and efficacy dropped off with increasing body mass index, as has been the case in trials of most of the biologics.
“When I look at the doses of the biologic agents that are used in treating the pediatric population and what we know about how these molecules are distributed in the body, we can safely say we are grossly underdosing children. We’re doing that because we believe somehow they’re at greater risk from exposures that are comparable to those in the adult population. And it’s not true. What we’ve seen over 20 years of using the biologics is that these agents are very safe and very effective when used appropriately in adequate doses,” according to Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
Asked which biologics he’s most comfortable with in prescribing for pediatric patients, Dr. Papp replied, “I think the best biologic therapies for pediatric patients are the ones that have been studied in the pediatric population: etanercept, ustekinumab, and adalimumab. Those three are the ones we want to choose because we at least have some guidance and some assurance in terms of expectations of response and adverse events.”
Dr. Papp and Dr. Langley have served as principal investigators of numerous clinical trials for and served as advisors to pharmaceutical companies developing dermatologic medications.
COPENHAGEN – Children and adolescents with moderate to severe psoriasis are generally undertreated, with resultant potential psychological impairment, social stigmatization, and isolation, psoriasis experts agreed at the annual congress of the European Academy of Dermatology and Venereology.
In a session featuring new long-term safety and efficacy data from extended pediatric clinical trials of etanercept and adalimumab, prominent clinical trial investigators who presented the findings asserted that many physicians and parents are overly timid about the use of systemic treatment options in children and adolescents when such therapy is clearly warranted.
“The burden of psoriasis is particularly acute in the pediatric population. I always ask patients, ‘How is psoriasis affecting you?’ And with children, it’s affecting them and it’s also affecting their parents. If it’s affecting their self-esteem – if it’s ruining your life and you can’t control it with topical agents – I think it’s time to think of other things,” declared Dr. Richard G. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
Yet all too often that doesn’t happen, as documented in the recently reported physician portion of the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey, which included 391 dermatologists and 390 rheumatologists in North America and Western Europe (J Eur Acad Dermatol Venereol. 2015 Oct;29[10]:2002-10).
Dr. Langley was a coinvestigator in MAPP, which found that 39% of dermatologists reported treating their moderate to severe psoriasis patients with conventional oral and/or biologic agents (19.5% and 19.6%, respectively). A total of 75% indicated they prescribe topical medications as monotherapy for their patients with moderate to severe psoriasis. In children, that rate was even higher, he said, despite the fact that topical monotherapy is clearly inadequate for treatment of more severe disease.
“I think there’s a fear about using systemics among parents and among some practitioners, which was one of the top reasons in the MAPP survey that people didn’t get appropriate treatment,” he said.
That’s a shortsighted attitude, the dermatologist continued: “The risk is not just the risk of the drug, but the risk of untreated disease, because untreated disease devastates patients.”
MAPP was the largest-ever survey of physician and patient perspectives regarding psoriasis and psoriatic arthritis. The patient perspective, derived from interviews with nearly 3,500 patients, was published earlier (J Am Acad Dermatol. 2014 May;70[5]:871-81).
One impediment to more widespread use of systemic therapies for pediatric psoriasis is that it’s almost entirely off-label prescribing. In the United States, no conventional oral agents or biologics are Food and Drug Administration approved for use in psoriasis patients under age 18 years. That was the case in Europe as well until earlier this year, when adalimumab (Humira) received European marketing approval for children ages 4 years and up with severe chronic plaque psoriasis with an inadequate response to topical agents and phototherapy or in whom such therapies are contraindicated.
Dr. Kim A. Papp, who presented new 52-week safety and efficacy data from a phase III study of adalimumab in pediatric psoriasis patients, said that even when dermatologists utilize biologics in pediatric patients, the medications are typically underdosed. In the phase III pediatric adalimumab trial, for example, most participants were overweight or obese, and efficacy dropped off with increasing body mass index, as has been the case in trials of most of the biologics.
“When I look at the doses of the biologic agents that are used in treating the pediatric population and what we know about how these molecules are distributed in the body, we can safely say we are grossly underdosing children. We’re doing that because we believe somehow they’re at greater risk from exposures that are comparable to those in the adult population. And it’s not true. What we’ve seen over 20 years of using the biologics is that these agents are very safe and very effective when used appropriately in adequate doses,” according to Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
Asked which biologics he’s most comfortable with in prescribing for pediatric patients, Dr. Papp replied, “I think the best biologic therapies for pediatric patients are the ones that have been studied in the pediatric population: etanercept, ustekinumab, and adalimumab. Those three are the ones we want to choose because we at least have some guidance and some assurance in terms of expectations of response and adverse events.”
Dr. Papp and Dr. Langley have served as principal investigators of numerous clinical trials for and served as advisors to pharmaceutical companies developing dermatologic medications.
COPENHAGEN – Children and adolescents with moderate to severe psoriasis are generally undertreated, with resultant potential psychological impairment, social stigmatization, and isolation, psoriasis experts agreed at the annual congress of the European Academy of Dermatology and Venereology.
In a session featuring new long-term safety and efficacy data from extended pediatric clinical trials of etanercept and adalimumab, prominent clinical trial investigators who presented the findings asserted that many physicians and parents are overly timid about the use of systemic treatment options in children and adolescents when such therapy is clearly warranted.
“The burden of psoriasis is particularly acute in the pediatric population. I always ask patients, ‘How is psoriasis affecting you?’ And with children, it’s affecting them and it’s also affecting their parents. If it’s affecting their self-esteem – if it’s ruining your life and you can’t control it with topical agents – I think it’s time to think of other things,” declared Dr. Richard G. Langley, professor of dermatology at Dalhousie University in Halifax, Nova Scotia.
Yet all too often that doesn’t happen, as documented in the recently reported physician portion of the Multinational Assessment of Psoriasis and Psoriatic Arthritis (MAPP) survey, which included 391 dermatologists and 390 rheumatologists in North America and Western Europe (J Eur Acad Dermatol Venereol. 2015 Oct;29[10]:2002-10).
Dr. Langley was a coinvestigator in MAPP, which found that 39% of dermatologists reported treating their moderate to severe psoriasis patients with conventional oral and/or biologic agents (19.5% and 19.6%, respectively). A total of 75% indicated they prescribe topical medications as monotherapy for their patients with moderate to severe psoriasis. In children, that rate was even higher, he said, despite the fact that topical monotherapy is clearly inadequate for treatment of more severe disease.
“I think there’s a fear about using systemics among parents and among some practitioners, which was one of the top reasons in the MAPP survey that people didn’t get appropriate treatment,” he said.
That’s a shortsighted attitude, the dermatologist continued: “The risk is not just the risk of the drug, but the risk of untreated disease, because untreated disease devastates patients.”
MAPP was the largest-ever survey of physician and patient perspectives regarding psoriasis and psoriatic arthritis. The patient perspective, derived from interviews with nearly 3,500 patients, was published earlier (J Am Acad Dermatol. 2014 May;70[5]:871-81).
One impediment to more widespread use of systemic therapies for pediatric psoriasis is that it’s almost entirely off-label prescribing. In the United States, no conventional oral agents or biologics are Food and Drug Administration approved for use in psoriasis patients under age 18 years. That was the case in Europe as well until earlier this year, when adalimumab (Humira) received European marketing approval for children ages 4 years and up with severe chronic plaque psoriasis with an inadequate response to topical agents and phototherapy or in whom such therapies are contraindicated.
Dr. Kim A. Papp, who presented new 52-week safety and efficacy data from a phase III study of adalimumab in pediatric psoriasis patients, said that even when dermatologists utilize biologics in pediatric patients, the medications are typically underdosed. In the phase III pediatric adalimumab trial, for example, most participants were overweight or obese, and efficacy dropped off with increasing body mass index, as has been the case in trials of most of the biologics.
“When I look at the doses of the biologic agents that are used in treating the pediatric population and what we know about how these molecules are distributed in the body, we can safely say we are grossly underdosing children. We’re doing that because we believe somehow they’re at greater risk from exposures that are comparable to those in the adult population. And it’s not true. What we’ve seen over 20 years of using the biologics is that these agents are very safe and very effective when used appropriately in adequate doses,” according to Dr. Papp, president of Probity Medical Research in Waterloo, Ont.
Asked which biologics he’s most comfortable with in prescribing for pediatric patients, Dr. Papp replied, “I think the best biologic therapies for pediatric patients are the ones that have been studied in the pediatric population: etanercept, ustekinumab, and adalimumab. Those three are the ones we want to choose because we at least have some guidance and some assurance in terms of expectations of response and adverse events.”
Dr. Papp and Dr. Langley have served as principal investigators of numerous clinical trials for and served as advisors to pharmaceutical companies developing dermatologic medications.
EXPERT ANALYSIS FROM THE EADV CONGRESS
ACR: Cardiovascular risk factors in psoriatic diseases are common, often go untreated
SAN FRANCISCO – Despite their frequent contact with the health care system, patients with psoriasis and psoriatic arthritis often receive no treatment for major cardiovascular risk factors, according to two large multicenter studies.
“We identified a gap in quality of care in terms of the primary prevention of cardiovascular risk factors in psoriatic arthritis and psoriasis. The next step will be to develop strategies to increase awareness and implement treatment recommendations among primary care physicians, dermatologists, and rheumatologists,” Dr. Lihi Eder of the University of Toronto said in an interview at the annual meeting of the American College of Rheumatology.
Psoriatic and cardiovascular diseases share an inflammatory etiology and often co-occur. In past studies, patients with psoriasis and psoriatic arthritis were about 50% more likely than average to have dyslipidemia and ischemic heart disease, and about 80%-90% more likely than usual to have hypertension and diabetes, Dr. Eder said.
She and her associates studied dyslipidemia and hypertension among 1,327 patients with psoriatic arthritis and 927 patients with psoriasis at eight sites in Canada, the United States, and Israel as part of the International Psoriasis & Arthritis Research Team (IPART). Based on medical and laboratory reports and self-reported data, the investigators assessed these comorbidities and whether treatment adhered to cholesterol and hypertension guidelines from the American College of Cardiology and the American Heart Association (Circulation. 2014 Jul 1;129:S1-45), and the Eighth Joint National Committee (JAMA. 2014 Feb 5;311[5]:507-20), respectively.
More than 80% of patients in the cohort had at least one modifiable cardiovascular risk factor, Dr. Eder said. While 6% had ischemic heart disease, 45% had hypertension, 71% had dyslipidemia, 13% had diabetes, 54% had central obesity, and 17% were current smokers. Furthermore, close to half of patients who had been diagnosed with hypertension had uncontrolled high blood pressure, and 57% were not receiving antihypertensive medications. Likewise, 58% of patients with dyslipidemia met criteria for statins, but only a third of these patients were receiving them.
Undertreatment was associated with having psoriatic arthritis or severe psoriasis and with having a high school or lower level of education, Dr. Eder added. “You have to remember that this study was conducted among specialists – these are supposed to be experts in the field,” she said. “If the treatment adherence is relatively low in these centers, then I would expect that for patients who are being followed in centers that do not specialize in psoriatic disease, adherence would be even lower.”
The second study detected significantly higher rates of cardiovascular risk factors among patients with psoriatic diseases, compared with controls from the Health Improvement Network, a medical records database that covers more than 9 million individuals in the United Kingdom. Patients with psoriatic arthritis or severe psoriasis were significantly more likely than were controls to develop hypertension, hyperlipidemia, obesity, or diabetes, with odds ratios ranging from 1.22 to 1.78, reported Dr. Kashif A. Jafri, who led the study while he was an internal medicine resident at the University of Pennsylvania in Philadelphia.
But despite their disproportionate risk, patients were treated at about the same rate as controls, Dr. Jafri said. About 15% of individuals with hypertension received no treatment, 30%-40% with hyperlipidemia went untreated, and nearly 60% with diabetes received no documented therapy. “The absence of a significant difference in receipt of appropriate therapy among the groups reflects a need for more careful attention to the management of cardiovascular risk factors in patients with inflammatory diseases,” Dr. Jafri emphasized. Because these risk factors can be successfully treated, it is “critical” to educate primary care providers about the need to do so, he said.
Rheumatologists also should periodically discuss cardiovascular risk factors with their patients as part of routine care, Dr. Jafri advised. “Although there are obviously time constraints during each office visit, this is a topic that dramatically influences the morbidity and mortality of our patient population, and rheumatologists have the unique ability to address this issue in the context of their long-term relationships with their patients,” he said.
Dr. Jafri is now a fellow in rheumatology at the University of California, San Francisco. His was supported by an Ephraim P. Engleman Endowed Resident Research Preceptorship Award from the Rheumatology Research Foundation. IPART is sponsored by the Krembil Foundation and the Canadian Institutes of Health Research. Dr. Jafri and Dr. Eder had no disclosures.
SAN FRANCISCO – Despite their frequent contact with the health care system, patients with psoriasis and psoriatic arthritis often receive no treatment for major cardiovascular risk factors, according to two large multicenter studies.
“We identified a gap in quality of care in terms of the primary prevention of cardiovascular risk factors in psoriatic arthritis and psoriasis. The next step will be to develop strategies to increase awareness and implement treatment recommendations among primary care physicians, dermatologists, and rheumatologists,” Dr. Lihi Eder of the University of Toronto said in an interview at the annual meeting of the American College of Rheumatology.
Psoriatic and cardiovascular diseases share an inflammatory etiology and often co-occur. In past studies, patients with psoriasis and psoriatic arthritis were about 50% more likely than average to have dyslipidemia and ischemic heart disease, and about 80%-90% more likely than usual to have hypertension and diabetes, Dr. Eder said.
She and her associates studied dyslipidemia and hypertension among 1,327 patients with psoriatic arthritis and 927 patients with psoriasis at eight sites in Canada, the United States, and Israel as part of the International Psoriasis & Arthritis Research Team (IPART). Based on medical and laboratory reports and self-reported data, the investigators assessed these comorbidities and whether treatment adhered to cholesterol and hypertension guidelines from the American College of Cardiology and the American Heart Association (Circulation. 2014 Jul 1;129:S1-45), and the Eighth Joint National Committee (JAMA. 2014 Feb 5;311[5]:507-20), respectively.
More than 80% of patients in the cohort had at least one modifiable cardiovascular risk factor, Dr. Eder said. While 6% had ischemic heart disease, 45% had hypertension, 71% had dyslipidemia, 13% had diabetes, 54% had central obesity, and 17% were current smokers. Furthermore, close to half of patients who had been diagnosed with hypertension had uncontrolled high blood pressure, and 57% were not receiving antihypertensive medications. Likewise, 58% of patients with dyslipidemia met criteria for statins, but only a third of these patients were receiving them.
Undertreatment was associated with having psoriatic arthritis or severe psoriasis and with having a high school or lower level of education, Dr. Eder added. “You have to remember that this study was conducted among specialists – these are supposed to be experts in the field,” she said. “If the treatment adherence is relatively low in these centers, then I would expect that for patients who are being followed in centers that do not specialize in psoriatic disease, adherence would be even lower.”
The second study detected significantly higher rates of cardiovascular risk factors among patients with psoriatic diseases, compared with controls from the Health Improvement Network, a medical records database that covers more than 9 million individuals in the United Kingdom. Patients with psoriatic arthritis or severe psoriasis were significantly more likely than were controls to develop hypertension, hyperlipidemia, obesity, or diabetes, with odds ratios ranging from 1.22 to 1.78, reported Dr. Kashif A. Jafri, who led the study while he was an internal medicine resident at the University of Pennsylvania in Philadelphia.
But despite their disproportionate risk, patients were treated at about the same rate as controls, Dr. Jafri said. About 15% of individuals with hypertension received no treatment, 30%-40% with hyperlipidemia went untreated, and nearly 60% with diabetes received no documented therapy. “The absence of a significant difference in receipt of appropriate therapy among the groups reflects a need for more careful attention to the management of cardiovascular risk factors in patients with inflammatory diseases,” Dr. Jafri emphasized. Because these risk factors can be successfully treated, it is “critical” to educate primary care providers about the need to do so, he said.
Rheumatologists also should periodically discuss cardiovascular risk factors with their patients as part of routine care, Dr. Jafri advised. “Although there are obviously time constraints during each office visit, this is a topic that dramatically influences the morbidity and mortality of our patient population, and rheumatologists have the unique ability to address this issue in the context of their long-term relationships with their patients,” he said.
Dr. Jafri is now a fellow in rheumatology at the University of California, San Francisco. His was supported by an Ephraim P. Engleman Endowed Resident Research Preceptorship Award from the Rheumatology Research Foundation. IPART is sponsored by the Krembil Foundation and the Canadian Institutes of Health Research. Dr. Jafri and Dr. Eder had no disclosures.
SAN FRANCISCO – Despite their frequent contact with the health care system, patients with psoriasis and psoriatic arthritis often receive no treatment for major cardiovascular risk factors, according to two large multicenter studies.
“We identified a gap in quality of care in terms of the primary prevention of cardiovascular risk factors in psoriatic arthritis and psoriasis. The next step will be to develop strategies to increase awareness and implement treatment recommendations among primary care physicians, dermatologists, and rheumatologists,” Dr. Lihi Eder of the University of Toronto said in an interview at the annual meeting of the American College of Rheumatology.
Psoriatic and cardiovascular diseases share an inflammatory etiology and often co-occur. In past studies, patients with psoriasis and psoriatic arthritis were about 50% more likely than average to have dyslipidemia and ischemic heart disease, and about 80%-90% more likely than usual to have hypertension and diabetes, Dr. Eder said.
She and her associates studied dyslipidemia and hypertension among 1,327 patients with psoriatic arthritis and 927 patients with psoriasis at eight sites in Canada, the United States, and Israel as part of the International Psoriasis & Arthritis Research Team (IPART). Based on medical and laboratory reports and self-reported data, the investigators assessed these comorbidities and whether treatment adhered to cholesterol and hypertension guidelines from the American College of Cardiology and the American Heart Association (Circulation. 2014 Jul 1;129:S1-45), and the Eighth Joint National Committee (JAMA. 2014 Feb 5;311[5]:507-20), respectively.
More than 80% of patients in the cohort had at least one modifiable cardiovascular risk factor, Dr. Eder said. While 6% had ischemic heart disease, 45% had hypertension, 71% had dyslipidemia, 13% had diabetes, 54% had central obesity, and 17% were current smokers. Furthermore, close to half of patients who had been diagnosed with hypertension had uncontrolled high blood pressure, and 57% were not receiving antihypertensive medications. Likewise, 58% of patients with dyslipidemia met criteria for statins, but only a third of these patients were receiving them.
Undertreatment was associated with having psoriatic arthritis or severe psoriasis and with having a high school or lower level of education, Dr. Eder added. “You have to remember that this study was conducted among specialists – these are supposed to be experts in the field,” she said. “If the treatment adherence is relatively low in these centers, then I would expect that for patients who are being followed in centers that do not specialize in psoriatic disease, adherence would be even lower.”
The second study detected significantly higher rates of cardiovascular risk factors among patients with psoriatic diseases, compared with controls from the Health Improvement Network, a medical records database that covers more than 9 million individuals in the United Kingdom. Patients with psoriatic arthritis or severe psoriasis were significantly more likely than were controls to develop hypertension, hyperlipidemia, obesity, or diabetes, with odds ratios ranging from 1.22 to 1.78, reported Dr. Kashif A. Jafri, who led the study while he was an internal medicine resident at the University of Pennsylvania in Philadelphia.
But despite their disproportionate risk, patients were treated at about the same rate as controls, Dr. Jafri said. About 15% of individuals with hypertension received no treatment, 30%-40% with hyperlipidemia went untreated, and nearly 60% with diabetes received no documented therapy. “The absence of a significant difference in receipt of appropriate therapy among the groups reflects a need for more careful attention to the management of cardiovascular risk factors in patients with inflammatory diseases,” Dr. Jafri emphasized. Because these risk factors can be successfully treated, it is “critical” to educate primary care providers about the need to do so, he said.
Rheumatologists also should periodically discuss cardiovascular risk factors with their patients as part of routine care, Dr. Jafri advised. “Although there are obviously time constraints during each office visit, this is a topic that dramatically influences the morbidity and mortality of our patient population, and rheumatologists have the unique ability to address this issue in the context of their long-term relationships with their patients,” he said.
Dr. Jafri is now a fellow in rheumatology at the University of California, San Francisco. His was supported by an Ephraim P. Engleman Endowed Resident Research Preceptorship Award from the Rheumatology Research Foundation. IPART is sponsored by the Krembil Foundation and the Canadian Institutes of Health Research. Dr. Jafri and Dr. Eder had no disclosures.
AT THE ACR ANNUAL MEETING
Key clinical point: Patients with psoriatic diseases have high rates of modifiable cardiovascular risk factors that often go untreated, based on two large studies.
Major finding: These risk factors were untreated about one-third to one-half of the time.
Data source: The first study included 1,327 patients with psoriatic arthritis and 927 patients with psoriasis identified through the International Psoriasis & Arthritis Research Team (IPART). The second study analyzed data from The Health Improvement Network, including 211,832 patients with psoriatic disease and more than 1.3 million controls.
Disclosures: The IPART is sponsored by the Krembil Foundation and the Canadian Institutes of Health Research. The second study was supported by an Ephraim P. Engleman Endowed Resident Research Preceptorship Award from the Rheumatology Research Foundation. Dr. Eder and Dr. Jafri had no disclosures.
State laws, regulatory concerns complicate biosimilars landscape
SAN FRANCISCO – A growing number of states are passing laws that set the parameters for how originator biologics may be replaced with biosimilars, and not all of them are helpful to physicians trying to maintain some knowledge and control over how prescriptions for biologics are dispensed, according to speakers at the annual meeting of the American College of Rheumatology.
These laws – now passed by a total of 19 states and Puerto Rico, including 12 in 2015 – address a requirement built into the Biologics Price Competition and Innovation Act of 2009 (part of the Affordable Care Act) that introduced the term “interchangeability” into the biosimilars approval pathway, stating that an approved biosimilar “may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product.” The law also gives 1 year of exclusive marketing rights to the first biosimilar approved as being interchangeable with the reference product.
Although regulations on how the Food and Drug Administration will deem a biosimilar to be interchangeable have yet to be drafted, it’s important for physicians to play an active role in shaping the laws that address interchangeability, said Dr. J. Eugene Huffstutter, a rheumatologist in private practice in Hixson, Tenn., and clinical assistant professor of medicine at the University of Tennessee, Chattanooga.
Dr. Huffstutter encouraged rheumatologists and other physicians to work with their local state medical associations to contact their state legislators to explain to them the need for strict laws on how biosimilars can be dispensed. He suggested that good state legislation on biosimilars should contain provisions stating that:
• Only FDA-approved interchangeable biosimilars can be substituted.
• No substitution can be made with “dispense as written” prescription.
• The prescribing physician must be notified of substitution within 1-5 days by electronic record or fax.
• The pharmacy must maintain a record of the dispensed drug.
Organizations including the ACR, the Coalition of State Rheumatology Organizations, patient groups, electronic prescribers, state medical societies, and pharmaceutical companies (for the most part) support strong state laws on biosimilars, while the pharmacy lobby and insurance companies have opposed them, Dr. Huffstutter said.
The 19 states with biosimilars laws include California, Colorado, Delaware, Florida, Georgia, Idaho, Illinois, Indiana, Louisiana, Massachusetts, New Jersey, North Carolina, North Dakota, Oregon, Tennessee, Texas, Utah, Virginia, and Washington, along with Puerto Rico. However, the laws in Oregon and Virginia will sunset in 2016. Another seven states have 2015 or current session legislation on biosimilar substitution: Hawaii, Maryland, Michigan, Mississippi, Oklahoma, Pennsylvania, and Vermont. States with bills that failed or were not acted upon at adjournment include Arizona, Arkansas, Nevada, and Rhode Island, according to information provided to Dr. Huffstutter by the Coalition.
State laws may differ on substitution notification, he noted. In Tennessee, the law (H.B. 572) states that notification should occur in “a reasonable period of time” instead of a defined period. “What may be reasonable for one person may not be reasonable for another. I really think 5 days is the outside time, or 5 business days, because if you’re thinking about some of the biosimilars that are coming down the pike, they’ll be given on a weekly basis, so you’ll at least know before [patients] get their second shot what they’re receiving,” Dr. Huffstutter noted.
Idaho is unique in that it impaneled a board of pharmacy regulation on biosimilar substitution rather than a separate law. It states that “A pharmacist may substitute an interchangeable biosimilar product for a prescribed biological product if the biosimilar has been determined by the FDA to be interchangeable and published in the Purple Book; the prescriber does not indicate by any means that the prescribed biological product must be dispensed; and the name of the drug and the manufacturer or the NDC [National Drug Code] number is documented in the patient medical record.”
The situation may become trickier as more biosimilars are approved, noted Dr. Huffstutter, who is a member of the ACR’s Government Affairs Committee and is a liaison to the Committee on Rheumatologic Care. It could potentially be a problem when a patient is taking a biosimilar for a particular originator biologic and then other biosimilars for that originator join the marketplace. A patient could potentially be switched from one biosimilar to another when a prescription is filled if the company marketing the second biosimilar happens to win a competitive bid with an insurance company, he said.
The filgrastim biosimilar called Zarxio is the only biosimilar currently approved in the United States, but the FDA has received two applications for biosimilars for inflammatory diseases: one from Celltrion for infliximab biosimilar Remsima (August 2014) and one from Sandoz for an etanercept biosimilar (October 2015). In addition to two infliximab biosimilars that have been approved in other countries, one etanercept biosimilar that has been approved in South Korea, and one adalimumab biosimilar approved in India, there are many others in development to treat inflammatory diseases as of July 2015, including 12 for adalimumab, 9 for etanercept, 5 for infliximab, 2 for tocilizumab, and 7 for rituximab, according to Dr. Jonathan Kay, professor of medicine and director of clinical research in the division of rheumatology at the University of Massachusetts, Worcester.
Unresolved regulatory concerns
In separate presentations at the meeting, Dr. Kay described many of the nuances that define biosimilars, which go by different terminology in other countries, such as “follow-on biologic” in Japan, “subsequent-entry biological” in Canada, and “similar biological medicinal product” in the European Union. They are defined as a legitimate copy of an off-patent biopharmaceutical that has undergone rigorous analytical comparison and clinical assessment, in comparison to its reference product, and are approved by a regulatory agency according to a specific pathway for biosimilar evaluation. These differ from biomimetics, which are developed as a replica of a biopharmaceutical but have not been developed, assessed, or approved according to regulatory guidelines for biosimilars.
The FDA takes a “totality of the evidence” approach to establish biosimilarity. The biosimilar pathway for approval is different from the originator pathway by relying more on analytical and preclinical studies and less on the clinical pharmacology and clinical trial data to support biosimilarity.
However, the agency has not yet published final guidance for all steps in demonstrating biosimilarity, particularly for clinical pharmacology data. Draft guidance says the biosimilar must be analyzed and assigned to an assessment grade: not similar, similar, highly similar, and highly similar with fingerprintlike similarity.
During production, both originator and biosimilar biopharmaceuticals can have protein-folding variants, misfolding, aggregation, enzymatic cleavage, and degradation that can lead to inactivation of the protein or increased immunogenicity. Even so, most biosimilars are not identical to the originator because of post-translational modifications, but they do not matter as long as they are not clinically meaningful. Even originator products can drift over time because small changes in manufacturing processes can lead to gradual changes in the molecule, Dr. Kay said.
Extrapolation of indications from the originator to a biosimilar is another area of concern, Dr. Kay said. The FDA will consider the extrapolation of data from a clinical trial of a biosimilar conducted in one disease to support approval for additional indications for which the originator product is already licensed, but not across indications with different mechanisms of action, such as between rheumatoid arthritis and non-Hodgkin’s lymphoma for rituximab. But for which inflammatory diseases should a biosimilar be studied to provide adequate information for extrapolation of indications? Dr. Kay asked. This situation brings about questions of whether dermatologists would be comfortable using a biosimilar that has been studied for rheumatoid arthritis to treat psoriasis or gastroenterologists using a drug with data only from psoriatic arthritis to treat Crohn’s disease or ulcerative colitis.
Both the Coalition of State Rheumatology Organizations and the ACR oppose extrapolation of indications from the clinical trial data of a biosimilar in one disease to support approval of additional indications for which the originator product is already licensed, but Dr. Kay thought that “extrapolation of indications makes perfect sense” because it won’t be possible to review a biosimilar for indications not already approved for the originator and it’s natural to extrapolate to already approved indications once the protein is shown to function nearly identically to the originator.
Ideally, the nomenclature system for biosimilars should clearly identify each product to improve pharmacovigilance and to differentiate between products that have not been determined to be interchangeable, Dr. Kay said. In August, the FDA announced draft guidance proposing that all biopharmaceutical products (originator and biosimilars) would have a nonproprietary name that includes a suffix of four lowercase letters that is devoid of meaning. For example, all products that share a core name such as replicamab would be named replicamab-cznm, replicamab-hixf, and so on, Dr. Kay said. Some people have voiced concern that it would be better for names to have some meaning, such as using an abbreviated name for the developing company along with the nonproprietary name of the originator product, but others noted that method could be problematic when companies merge or are acquired by others.
Dr. Huffstutter disclosed relationships with Janssen, UCB, Lilly, Pfizer, Genentech, Bristol-Myers Squibb, and Celgene. Dr. Kay has received grant and research support from many pharmaceutical companies and has served as a consultant or on an advisory board for many pharmaceutical companies, including those developing biosimilars.
SAN FRANCISCO – A growing number of states are passing laws that set the parameters for how originator biologics may be replaced with biosimilars, and not all of them are helpful to physicians trying to maintain some knowledge and control over how prescriptions for biologics are dispensed, according to speakers at the annual meeting of the American College of Rheumatology.
These laws – now passed by a total of 19 states and Puerto Rico, including 12 in 2015 – address a requirement built into the Biologics Price Competition and Innovation Act of 2009 (part of the Affordable Care Act) that introduced the term “interchangeability” into the biosimilars approval pathway, stating that an approved biosimilar “may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product.” The law also gives 1 year of exclusive marketing rights to the first biosimilar approved as being interchangeable with the reference product.
Although regulations on how the Food and Drug Administration will deem a biosimilar to be interchangeable have yet to be drafted, it’s important for physicians to play an active role in shaping the laws that address interchangeability, said Dr. J. Eugene Huffstutter, a rheumatologist in private practice in Hixson, Tenn., and clinical assistant professor of medicine at the University of Tennessee, Chattanooga.
Dr. Huffstutter encouraged rheumatologists and other physicians to work with their local state medical associations to contact their state legislators to explain to them the need for strict laws on how biosimilars can be dispensed. He suggested that good state legislation on biosimilars should contain provisions stating that:
• Only FDA-approved interchangeable biosimilars can be substituted.
• No substitution can be made with “dispense as written” prescription.
• The prescribing physician must be notified of substitution within 1-5 days by electronic record or fax.
• The pharmacy must maintain a record of the dispensed drug.
Organizations including the ACR, the Coalition of State Rheumatology Organizations, patient groups, electronic prescribers, state medical societies, and pharmaceutical companies (for the most part) support strong state laws on biosimilars, while the pharmacy lobby and insurance companies have opposed them, Dr. Huffstutter said.
The 19 states with biosimilars laws include California, Colorado, Delaware, Florida, Georgia, Idaho, Illinois, Indiana, Louisiana, Massachusetts, New Jersey, North Carolina, North Dakota, Oregon, Tennessee, Texas, Utah, Virginia, and Washington, along with Puerto Rico. However, the laws in Oregon and Virginia will sunset in 2016. Another seven states have 2015 or current session legislation on biosimilar substitution: Hawaii, Maryland, Michigan, Mississippi, Oklahoma, Pennsylvania, and Vermont. States with bills that failed or were not acted upon at adjournment include Arizona, Arkansas, Nevada, and Rhode Island, according to information provided to Dr. Huffstutter by the Coalition.
State laws may differ on substitution notification, he noted. In Tennessee, the law (H.B. 572) states that notification should occur in “a reasonable period of time” instead of a defined period. “What may be reasonable for one person may not be reasonable for another. I really think 5 days is the outside time, or 5 business days, because if you’re thinking about some of the biosimilars that are coming down the pike, they’ll be given on a weekly basis, so you’ll at least know before [patients] get their second shot what they’re receiving,” Dr. Huffstutter noted.
Idaho is unique in that it impaneled a board of pharmacy regulation on biosimilar substitution rather than a separate law. It states that “A pharmacist may substitute an interchangeable biosimilar product for a prescribed biological product if the biosimilar has been determined by the FDA to be interchangeable and published in the Purple Book; the prescriber does not indicate by any means that the prescribed biological product must be dispensed; and the name of the drug and the manufacturer or the NDC [National Drug Code] number is documented in the patient medical record.”
The situation may become trickier as more biosimilars are approved, noted Dr. Huffstutter, who is a member of the ACR’s Government Affairs Committee and is a liaison to the Committee on Rheumatologic Care. It could potentially be a problem when a patient is taking a biosimilar for a particular originator biologic and then other biosimilars for that originator join the marketplace. A patient could potentially be switched from one biosimilar to another when a prescription is filled if the company marketing the second biosimilar happens to win a competitive bid with an insurance company, he said.
The filgrastim biosimilar called Zarxio is the only biosimilar currently approved in the United States, but the FDA has received two applications for biosimilars for inflammatory diseases: one from Celltrion for infliximab biosimilar Remsima (August 2014) and one from Sandoz for an etanercept biosimilar (October 2015). In addition to two infliximab biosimilars that have been approved in other countries, one etanercept biosimilar that has been approved in South Korea, and one adalimumab biosimilar approved in India, there are many others in development to treat inflammatory diseases as of July 2015, including 12 for adalimumab, 9 for etanercept, 5 for infliximab, 2 for tocilizumab, and 7 for rituximab, according to Dr. Jonathan Kay, professor of medicine and director of clinical research in the division of rheumatology at the University of Massachusetts, Worcester.
Unresolved regulatory concerns
In separate presentations at the meeting, Dr. Kay described many of the nuances that define biosimilars, which go by different terminology in other countries, such as “follow-on biologic” in Japan, “subsequent-entry biological” in Canada, and “similar biological medicinal product” in the European Union. They are defined as a legitimate copy of an off-patent biopharmaceutical that has undergone rigorous analytical comparison and clinical assessment, in comparison to its reference product, and are approved by a regulatory agency according to a specific pathway for biosimilar evaluation. These differ from biomimetics, which are developed as a replica of a biopharmaceutical but have not been developed, assessed, or approved according to regulatory guidelines for biosimilars.
The FDA takes a “totality of the evidence” approach to establish biosimilarity. The biosimilar pathway for approval is different from the originator pathway by relying more on analytical and preclinical studies and less on the clinical pharmacology and clinical trial data to support biosimilarity.
However, the agency has not yet published final guidance for all steps in demonstrating biosimilarity, particularly for clinical pharmacology data. Draft guidance says the biosimilar must be analyzed and assigned to an assessment grade: not similar, similar, highly similar, and highly similar with fingerprintlike similarity.
During production, both originator and biosimilar biopharmaceuticals can have protein-folding variants, misfolding, aggregation, enzymatic cleavage, and degradation that can lead to inactivation of the protein or increased immunogenicity. Even so, most biosimilars are not identical to the originator because of post-translational modifications, but they do not matter as long as they are not clinically meaningful. Even originator products can drift over time because small changes in manufacturing processes can lead to gradual changes in the molecule, Dr. Kay said.
Extrapolation of indications from the originator to a biosimilar is another area of concern, Dr. Kay said. The FDA will consider the extrapolation of data from a clinical trial of a biosimilar conducted in one disease to support approval for additional indications for which the originator product is already licensed, but not across indications with different mechanisms of action, such as between rheumatoid arthritis and non-Hodgkin’s lymphoma for rituximab. But for which inflammatory diseases should a biosimilar be studied to provide adequate information for extrapolation of indications? Dr. Kay asked. This situation brings about questions of whether dermatologists would be comfortable using a biosimilar that has been studied for rheumatoid arthritis to treat psoriasis or gastroenterologists using a drug with data only from psoriatic arthritis to treat Crohn’s disease or ulcerative colitis.
Both the Coalition of State Rheumatology Organizations and the ACR oppose extrapolation of indications from the clinical trial data of a biosimilar in one disease to support approval of additional indications for which the originator product is already licensed, but Dr. Kay thought that “extrapolation of indications makes perfect sense” because it won’t be possible to review a biosimilar for indications not already approved for the originator and it’s natural to extrapolate to already approved indications once the protein is shown to function nearly identically to the originator.
Ideally, the nomenclature system for biosimilars should clearly identify each product to improve pharmacovigilance and to differentiate between products that have not been determined to be interchangeable, Dr. Kay said. In August, the FDA announced draft guidance proposing that all biopharmaceutical products (originator and biosimilars) would have a nonproprietary name that includes a suffix of four lowercase letters that is devoid of meaning. For example, all products that share a core name such as replicamab would be named replicamab-cznm, replicamab-hixf, and so on, Dr. Kay said. Some people have voiced concern that it would be better for names to have some meaning, such as using an abbreviated name for the developing company along with the nonproprietary name of the originator product, but others noted that method could be problematic when companies merge or are acquired by others.
Dr. Huffstutter disclosed relationships with Janssen, UCB, Lilly, Pfizer, Genentech, Bristol-Myers Squibb, and Celgene. Dr. Kay has received grant and research support from many pharmaceutical companies and has served as a consultant or on an advisory board for many pharmaceutical companies, including those developing biosimilars.
SAN FRANCISCO – A growing number of states are passing laws that set the parameters for how originator biologics may be replaced with biosimilars, and not all of them are helpful to physicians trying to maintain some knowledge and control over how prescriptions for biologics are dispensed, according to speakers at the annual meeting of the American College of Rheumatology.
These laws – now passed by a total of 19 states and Puerto Rico, including 12 in 2015 – address a requirement built into the Biologics Price Competition and Innovation Act of 2009 (part of the Affordable Care Act) that introduced the term “interchangeability” into the biosimilars approval pathway, stating that an approved biosimilar “may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product.” The law also gives 1 year of exclusive marketing rights to the first biosimilar approved as being interchangeable with the reference product.
Although regulations on how the Food and Drug Administration will deem a biosimilar to be interchangeable have yet to be drafted, it’s important for physicians to play an active role in shaping the laws that address interchangeability, said Dr. J. Eugene Huffstutter, a rheumatologist in private practice in Hixson, Tenn., and clinical assistant professor of medicine at the University of Tennessee, Chattanooga.
Dr. Huffstutter encouraged rheumatologists and other physicians to work with their local state medical associations to contact their state legislators to explain to them the need for strict laws on how biosimilars can be dispensed. He suggested that good state legislation on biosimilars should contain provisions stating that:
• Only FDA-approved interchangeable biosimilars can be substituted.
• No substitution can be made with “dispense as written” prescription.
• The prescribing physician must be notified of substitution within 1-5 days by electronic record or fax.
• The pharmacy must maintain a record of the dispensed drug.
Organizations including the ACR, the Coalition of State Rheumatology Organizations, patient groups, electronic prescribers, state medical societies, and pharmaceutical companies (for the most part) support strong state laws on biosimilars, while the pharmacy lobby and insurance companies have opposed them, Dr. Huffstutter said.
The 19 states with biosimilars laws include California, Colorado, Delaware, Florida, Georgia, Idaho, Illinois, Indiana, Louisiana, Massachusetts, New Jersey, North Carolina, North Dakota, Oregon, Tennessee, Texas, Utah, Virginia, and Washington, along with Puerto Rico. However, the laws in Oregon and Virginia will sunset in 2016. Another seven states have 2015 or current session legislation on biosimilar substitution: Hawaii, Maryland, Michigan, Mississippi, Oklahoma, Pennsylvania, and Vermont. States with bills that failed or were not acted upon at adjournment include Arizona, Arkansas, Nevada, and Rhode Island, according to information provided to Dr. Huffstutter by the Coalition.
State laws may differ on substitution notification, he noted. In Tennessee, the law (H.B. 572) states that notification should occur in “a reasonable period of time” instead of a defined period. “What may be reasonable for one person may not be reasonable for another. I really think 5 days is the outside time, or 5 business days, because if you’re thinking about some of the biosimilars that are coming down the pike, they’ll be given on a weekly basis, so you’ll at least know before [patients] get their second shot what they’re receiving,” Dr. Huffstutter noted.
Idaho is unique in that it impaneled a board of pharmacy regulation on biosimilar substitution rather than a separate law. It states that “A pharmacist may substitute an interchangeable biosimilar product for a prescribed biological product if the biosimilar has been determined by the FDA to be interchangeable and published in the Purple Book; the prescriber does not indicate by any means that the prescribed biological product must be dispensed; and the name of the drug and the manufacturer or the NDC [National Drug Code] number is documented in the patient medical record.”
The situation may become trickier as more biosimilars are approved, noted Dr. Huffstutter, who is a member of the ACR’s Government Affairs Committee and is a liaison to the Committee on Rheumatologic Care. It could potentially be a problem when a patient is taking a biosimilar for a particular originator biologic and then other biosimilars for that originator join the marketplace. A patient could potentially be switched from one biosimilar to another when a prescription is filled if the company marketing the second biosimilar happens to win a competitive bid with an insurance company, he said.
The filgrastim biosimilar called Zarxio is the only biosimilar currently approved in the United States, but the FDA has received two applications for biosimilars for inflammatory diseases: one from Celltrion for infliximab biosimilar Remsima (August 2014) and one from Sandoz for an etanercept biosimilar (October 2015). In addition to two infliximab biosimilars that have been approved in other countries, one etanercept biosimilar that has been approved in South Korea, and one adalimumab biosimilar approved in India, there are many others in development to treat inflammatory diseases as of July 2015, including 12 for adalimumab, 9 for etanercept, 5 for infliximab, 2 for tocilizumab, and 7 for rituximab, according to Dr. Jonathan Kay, professor of medicine and director of clinical research in the division of rheumatology at the University of Massachusetts, Worcester.
Unresolved regulatory concerns
In separate presentations at the meeting, Dr. Kay described many of the nuances that define biosimilars, which go by different terminology in other countries, such as “follow-on biologic” in Japan, “subsequent-entry biological” in Canada, and “similar biological medicinal product” in the European Union. They are defined as a legitimate copy of an off-patent biopharmaceutical that has undergone rigorous analytical comparison and clinical assessment, in comparison to its reference product, and are approved by a regulatory agency according to a specific pathway for biosimilar evaluation. These differ from biomimetics, which are developed as a replica of a biopharmaceutical but have not been developed, assessed, or approved according to regulatory guidelines for biosimilars.
The FDA takes a “totality of the evidence” approach to establish biosimilarity. The biosimilar pathway for approval is different from the originator pathway by relying more on analytical and preclinical studies and less on the clinical pharmacology and clinical trial data to support biosimilarity.
However, the agency has not yet published final guidance for all steps in demonstrating biosimilarity, particularly for clinical pharmacology data. Draft guidance says the biosimilar must be analyzed and assigned to an assessment grade: not similar, similar, highly similar, and highly similar with fingerprintlike similarity.
During production, both originator and biosimilar biopharmaceuticals can have protein-folding variants, misfolding, aggregation, enzymatic cleavage, and degradation that can lead to inactivation of the protein or increased immunogenicity. Even so, most biosimilars are not identical to the originator because of post-translational modifications, but they do not matter as long as they are not clinically meaningful. Even originator products can drift over time because small changes in manufacturing processes can lead to gradual changes in the molecule, Dr. Kay said.
Extrapolation of indications from the originator to a biosimilar is another area of concern, Dr. Kay said. The FDA will consider the extrapolation of data from a clinical trial of a biosimilar conducted in one disease to support approval for additional indications for which the originator product is already licensed, but not across indications with different mechanisms of action, such as between rheumatoid arthritis and non-Hodgkin’s lymphoma for rituximab. But for which inflammatory diseases should a biosimilar be studied to provide adequate information for extrapolation of indications? Dr. Kay asked. This situation brings about questions of whether dermatologists would be comfortable using a biosimilar that has been studied for rheumatoid arthritis to treat psoriasis or gastroenterologists using a drug with data only from psoriatic arthritis to treat Crohn’s disease or ulcerative colitis.
Both the Coalition of State Rheumatology Organizations and the ACR oppose extrapolation of indications from the clinical trial data of a biosimilar in one disease to support approval of additional indications for which the originator product is already licensed, but Dr. Kay thought that “extrapolation of indications makes perfect sense” because it won’t be possible to review a biosimilar for indications not already approved for the originator and it’s natural to extrapolate to already approved indications once the protein is shown to function nearly identically to the originator.
Ideally, the nomenclature system for biosimilars should clearly identify each product to improve pharmacovigilance and to differentiate between products that have not been determined to be interchangeable, Dr. Kay said. In August, the FDA announced draft guidance proposing that all biopharmaceutical products (originator and biosimilars) would have a nonproprietary name that includes a suffix of four lowercase letters that is devoid of meaning. For example, all products that share a core name such as replicamab would be named replicamab-cznm, replicamab-hixf, and so on, Dr. Kay said. Some people have voiced concern that it would be better for names to have some meaning, such as using an abbreviated name for the developing company along with the nonproprietary name of the originator product, but others noted that method could be problematic when companies merge or are acquired by others.
Dr. Huffstutter disclosed relationships with Janssen, UCB, Lilly, Pfizer, Genentech, Bristol-Myers Squibb, and Celgene. Dr. Kay has received grant and research support from many pharmaceutical companies and has served as a consultant or on an advisory board for many pharmaceutical companies, including those developing biosimilars.
EXPERT ANALYSIS FROM THE ACR ANNUAL MEETING
Hope for Hidradenitis Suppurativa
In September 2015, the US Food and Drug Administration approved adalimumab, the well-known injectable tumor necrosis factor (TNF)–α inhibitor indicated for psoriasis and other inflammatory conditions, for treatment of moderate to severe hidradenitis suppurativa (HS), classifying it as the first and only US Food and Drug Administration–approved therapy for adults with HS.
Pivotal studies (PIONEER/HS-I and -II, phase 3, double-blind) evaluated 633 patients (307 in HS-I and 326 in HS-II) with moderate to severe HS who were randomized to adalimumab versus placebo for 12 weeks. There was significant clinical response (at least 50% reduction in abscess and inflammatory nodule count, defined as hidradenitis suppurativa clinical response, HiSCR) in the adalimumab group (42% vs 26% in HS-I, 59% vs. 28% in HS-II, P<.001), reduction in pain (significant in the HS-II trial, 45.7% vs 20.7%, P<.001; HS-I 27.9% vs 24.8%, P>.05), and no new safety concerns when compared to other adalimumab dosages and indications. Some HS-II study patients (19.3%) were permitted to use oral antibiotics during the study.
For the indication of adult HS (moderate to severe disease), adalimumab should be administered subcutaneously at a dosing regimen of 160 mg/4 syringes on day 1 (or 80 mg/2 syringes on days 1 and 2), followed by 80 mg/2 syringes on day 15, then 40 mg/1 syringe on day 29, and every 7 days thereafter for an indefinite treatment period.
What’s the Issue?
It sits well with dermatologists when the indications for a medication with which we have great familiarity are broadened to include new disease entities; it is even better when the new entity is a condition for which every dermatologist pines for efficacious treatment options. Despite the disease burden of HS, which can include pain, scarring, disfigurement, social exclusion, and/or embarrassment, as well as the wasteful and burdensome effect that HS has on health care resources, such as injudicious use of antibiotics and unnecessary emergency department visits and inpatient hospital stays,1 there is nonetheless a wide-open and inviting playing field for effective therapies.
Although a much higher dosage of adalimumab is required in the treatment of HS compared to what is indicated for psoriasis patients, its safety concerns and side effect profile were unchanged in HS, and therefore its monitoring guidelines remain the same. Not all patients in these studies showed a notable reduction in lesion count, but given that HS classically is unresponsive to most medication regimens, will you embrace this therapy option for your patients with HS?
Reference
1. Khalsa A, Liu G, Kirby JS. Increased utilization of emergency department and inpatient care by patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73:609-614.
In September 2015, the US Food and Drug Administration approved adalimumab, the well-known injectable tumor necrosis factor (TNF)–α inhibitor indicated for psoriasis and other inflammatory conditions, for treatment of moderate to severe hidradenitis suppurativa (HS), classifying it as the first and only US Food and Drug Administration–approved therapy for adults with HS.
Pivotal studies (PIONEER/HS-I and -II, phase 3, double-blind) evaluated 633 patients (307 in HS-I and 326 in HS-II) with moderate to severe HS who were randomized to adalimumab versus placebo for 12 weeks. There was significant clinical response (at least 50% reduction in abscess and inflammatory nodule count, defined as hidradenitis suppurativa clinical response, HiSCR) in the adalimumab group (42% vs 26% in HS-I, 59% vs. 28% in HS-II, P<.001), reduction in pain (significant in the HS-II trial, 45.7% vs 20.7%, P<.001; HS-I 27.9% vs 24.8%, P>.05), and no new safety concerns when compared to other adalimumab dosages and indications. Some HS-II study patients (19.3%) were permitted to use oral antibiotics during the study.
For the indication of adult HS (moderate to severe disease), adalimumab should be administered subcutaneously at a dosing regimen of 160 mg/4 syringes on day 1 (or 80 mg/2 syringes on days 1 and 2), followed by 80 mg/2 syringes on day 15, then 40 mg/1 syringe on day 29, and every 7 days thereafter for an indefinite treatment period.
What’s the Issue?
It sits well with dermatologists when the indications for a medication with which we have great familiarity are broadened to include new disease entities; it is even better when the new entity is a condition for which every dermatologist pines for efficacious treatment options. Despite the disease burden of HS, which can include pain, scarring, disfigurement, social exclusion, and/or embarrassment, as well as the wasteful and burdensome effect that HS has on health care resources, such as injudicious use of antibiotics and unnecessary emergency department visits and inpatient hospital stays,1 there is nonetheless a wide-open and inviting playing field for effective therapies.
Although a much higher dosage of adalimumab is required in the treatment of HS compared to what is indicated for psoriasis patients, its safety concerns and side effect profile were unchanged in HS, and therefore its monitoring guidelines remain the same. Not all patients in these studies showed a notable reduction in lesion count, but given that HS classically is unresponsive to most medication regimens, will you embrace this therapy option for your patients with HS?
In September 2015, the US Food and Drug Administration approved adalimumab, the well-known injectable tumor necrosis factor (TNF)–α inhibitor indicated for psoriasis and other inflammatory conditions, for treatment of moderate to severe hidradenitis suppurativa (HS), classifying it as the first and only US Food and Drug Administration–approved therapy for adults with HS.
Pivotal studies (PIONEER/HS-I and -II, phase 3, double-blind) evaluated 633 patients (307 in HS-I and 326 in HS-II) with moderate to severe HS who were randomized to adalimumab versus placebo for 12 weeks. There was significant clinical response (at least 50% reduction in abscess and inflammatory nodule count, defined as hidradenitis suppurativa clinical response, HiSCR) in the adalimumab group (42% vs 26% in HS-I, 59% vs. 28% in HS-II, P<.001), reduction in pain (significant in the HS-II trial, 45.7% vs 20.7%, P<.001; HS-I 27.9% vs 24.8%, P>.05), and no new safety concerns when compared to other adalimumab dosages and indications. Some HS-II study patients (19.3%) were permitted to use oral antibiotics during the study.
For the indication of adult HS (moderate to severe disease), adalimumab should be administered subcutaneously at a dosing regimen of 160 mg/4 syringes on day 1 (or 80 mg/2 syringes on days 1 and 2), followed by 80 mg/2 syringes on day 15, then 40 mg/1 syringe on day 29, and every 7 days thereafter for an indefinite treatment period.
What’s the Issue?
It sits well with dermatologists when the indications for a medication with which we have great familiarity are broadened to include new disease entities; it is even better when the new entity is a condition for which every dermatologist pines for efficacious treatment options. Despite the disease burden of HS, which can include pain, scarring, disfigurement, social exclusion, and/or embarrassment, as well as the wasteful and burdensome effect that HS has on health care resources, such as injudicious use of antibiotics and unnecessary emergency department visits and inpatient hospital stays,1 there is nonetheless a wide-open and inviting playing field for effective therapies.
Although a much higher dosage of adalimumab is required in the treatment of HS compared to what is indicated for psoriasis patients, its safety concerns and side effect profile were unchanged in HS, and therefore its monitoring guidelines remain the same. Not all patients in these studies showed a notable reduction in lesion count, but given that HS classically is unresponsive to most medication regimens, will you embrace this therapy option for your patients with HS?
Reference
1. Khalsa A, Liu G, Kirby JS. Increased utilization of emergency department and inpatient care by patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73:609-614.
Reference
1. Khalsa A, Liu G, Kirby JS. Increased utilization of emergency department and inpatient care by patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73:609-614.
Common sense drives risk factor screening for systemic psoriasis therapy
LAS VEGAS – For a physician starting a patient on a systemic agent to treat psoriasis, deciding on risk factor screening and management strategies can be confusing. Who gets checked for what? Why? How often?
With a dearth of evidence to guide them, physicians should use what they know about psoriasis itself, as well as patient risk factors and the inherent risks of a given systemic therapy to guide screening, according to Dr. Kristina Callis Duffin of the department of dermatology, University of Utah, Salt Lake City.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar, Dr. Duffin said that in her practice, this means that all patients being considered for systemic therapy get baseline labs as recommended by the drug manufacturer, and risk-focused monitoring for cardiovascular disease, infection, and cancer. Basically, she noted, the strategy is, “How can we be effective at monitoring and surveying for those ‘bad guys’– those complications of therapy?”
“Some of this is really about using common sense and being a good physician,” she added.
To obtain a pertinent history, it’s worthwhile to formulate a directed intake questionnaire, Dr. Duffin said. Screening items should include history of recurrent infections; hepatitis, HIV, and TB status and risk; travel history; cardiovascular history, including lipid status; history of any liver or kidney problems; history and risk of diabetes mellitus; depression; personal or family history of multiple sclerosis; and history of cancer, including skin cancer, and blood disorders.
The social history should include pregnancy status and birth control method for women (as appropriate); and alcohol use for all patients. Patients should be current with age-appropriate recommended immunizations and cancer screenings.
Obtaining baseline lab tests for all patients before they start treatment with a systemic agent affords the opportunity to detect common metabolic abnormalities, such as diabetes, liver or kidney disease, and dyslipidemia, as well as rare but serious blood abnormalities, Dr. Duffin said. This approach respects the fact that “there is no question that cardiovascular risk factors are overrepresented in the moderate to severe psoriasis population.”
Published TB screening guidelines have universally recommended screening before initiating a biologic agent with a tuberculin skin test, unless the patient has had the BCG vaccine or is immunosuppressed. From there, interpretation of the tine test and further screening requires knowing the patient: “It’s all about pretest probability,” said Dr. Duffin, adding that physicians should not be afraid to use infectious disease consultants to help them sort out tricky cases.
In terms of ongoing management, communication and coordination are key. Continue to ask patients about symptoms of psoriatic arthritis, and make sure that you work with the patient’s primary care provider so that cardiovascular risk management, malignancy screening, and immunizations don’t fall through the cracks, she advised.
“Have a high index of suspicion for infection,” including deep fungal infections, she added. Though the literature does not report frequent cases of serious fungal infections, “there’s no question cases are unreported or numbers can be unclear in the registries.” Patients should know early signs of infection and know to seek care right away.
Patients should know to defer live vaccines, and the physician managing the care of psoriasis should be included in the planning and management process for any type of surgery, to oversee biologic administration. While probably slight, the risk of infection should be weighed against the risk of loss of efficacy in psoriasis or psoriatic arthritis treatment, Dr. Duffin said.
The baseline risk of solid tumors is elevated in patients with psoriasis, but “is not definitively elevated in patients on biologics,” she commented. The risk of nonmelanoma skin cancer, however, is definitely elevated for patients on biologic therapies, so an annual skin survey is a must for these patients, she said.
“Should we be discussing immunizations with our patients? Absolutely,”she added. All patients need the influenza vaccine and the pneumococcal vaccine, and for live virus immunizations, the dermatologist should coordinate administration with the primary care physician.
Managing these risks effectively can make a big difference in quality of life for patients who are able to start and stay on systemic therapy, said Dr. Duffin. The many risks of not treating psoriasis appropriately include not only the significant psychosocial impact, but also progression of psoriatic arthritis, progression of skin disease, and increased risk of heart disease and other cardiovascular comorbidities. “Our job is to treat to patient satisfaction and the best possible quality of life. A lot of this is about anticipation and communication,” she said.
Dr. Duffin reported receiving research support from and consulting for Amgen, Eli Lilly, Janssen, Stiefel, AbbVie, Bristol-Myers Squibb, Celgene, Novartis, and XenoPort; consulting and being on the scientific advisory board for Pfizer; and being on the scientific advisory board for Novartis, Eli Lilly, Janssen, Celgene, and XenoPort.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
LAS VEGAS – For a physician starting a patient on a systemic agent to treat psoriasis, deciding on risk factor screening and management strategies can be confusing. Who gets checked for what? Why? How often?
With a dearth of evidence to guide them, physicians should use what they know about psoriasis itself, as well as patient risk factors and the inherent risks of a given systemic therapy to guide screening, according to Dr. Kristina Callis Duffin of the department of dermatology, University of Utah, Salt Lake City.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar, Dr. Duffin said that in her practice, this means that all patients being considered for systemic therapy get baseline labs as recommended by the drug manufacturer, and risk-focused monitoring for cardiovascular disease, infection, and cancer. Basically, she noted, the strategy is, “How can we be effective at monitoring and surveying for those ‘bad guys’– those complications of therapy?”
“Some of this is really about using common sense and being a good physician,” she added.
To obtain a pertinent history, it’s worthwhile to formulate a directed intake questionnaire, Dr. Duffin said. Screening items should include history of recurrent infections; hepatitis, HIV, and TB status and risk; travel history; cardiovascular history, including lipid status; history of any liver or kidney problems; history and risk of diabetes mellitus; depression; personal or family history of multiple sclerosis; and history of cancer, including skin cancer, and blood disorders.
The social history should include pregnancy status and birth control method for women (as appropriate); and alcohol use for all patients. Patients should be current with age-appropriate recommended immunizations and cancer screenings.
Obtaining baseline lab tests for all patients before they start treatment with a systemic agent affords the opportunity to detect common metabolic abnormalities, such as diabetes, liver or kidney disease, and dyslipidemia, as well as rare but serious blood abnormalities, Dr. Duffin said. This approach respects the fact that “there is no question that cardiovascular risk factors are overrepresented in the moderate to severe psoriasis population.”
Published TB screening guidelines have universally recommended screening before initiating a biologic agent with a tuberculin skin test, unless the patient has had the BCG vaccine or is immunosuppressed. From there, interpretation of the tine test and further screening requires knowing the patient: “It’s all about pretest probability,” said Dr. Duffin, adding that physicians should not be afraid to use infectious disease consultants to help them sort out tricky cases.
In terms of ongoing management, communication and coordination are key. Continue to ask patients about symptoms of psoriatic arthritis, and make sure that you work with the patient’s primary care provider so that cardiovascular risk management, malignancy screening, and immunizations don’t fall through the cracks, she advised.
“Have a high index of suspicion for infection,” including deep fungal infections, she added. Though the literature does not report frequent cases of serious fungal infections, “there’s no question cases are unreported or numbers can be unclear in the registries.” Patients should know early signs of infection and know to seek care right away.
Patients should know to defer live vaccines, and the physician managing the care of psoriasis should be included in the planning and management process for any type of surgery, to oversee biologic administration. While probably slight, the risk of infection should be weighed against the risk of loss of efficacy in psoriasis or psoriatic arthritis treatment, Dr. Duffin said.
The baseline risk of solid tumors is elevated in patients with psoriasis, but “is not definitively elevated in patients on biologics,” she commented. The risk of nonmelanoma skin cancer, however, is definitely elevated for patients on biologic therapies, so an annual skin survey is a must for these patients, she said.
“Should we be discussing immunizations with our patients? Absolutely,”she added. All patients need the influenza vaccine and the pneumococcal vaccine, and for live virus immunizations, the dermatologist should coordinate administration with the primary care physician.
Managing these risks effectively can make a big difference in quality of life for patients who are able to start and stay on systemic therapy, said Dr. Duffin. The many risks of not treating psoriasis appropriately include not only the significant psychosocial impact, but also progression of psoriatic arthritis, progression of skin disease, and increased risk of heart disease and other cardiovascular comorbidities. “Our job is to treat to patient satisfaction and the best possible quality of life. A lot of this is about anticipation and communication,” she said.
Dr. Duffin reported receiving research support from and consulting for Amgen, Eli Lilly, Janssen, Stiefel, AbbVie, Bristol-Myers Squibb, Celgene, Novartis, and XenoPort; consulting and being on the scientific advisory board for Pfizer; and being on the scientific advisory board for Novartis, Eli Lilly, Janssen, Celgene, and XenoPort.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
LAS VEGAS – For a physician starting a patient on a systemic agent to treat psoriasis, deciding on risk factor screening and management strategies can be confusing. Who gets checked for what? Why? How often?
With a dearth of evidence to guide them, physicians should use what they know about psoriasis itself, as well as patient risk factors and the inherent risks of a given systemic therapy to guide screening, according to Dr. Kristina Callis Duffin of the department of dermatology, University of Utah, Salt Lake City.
Speaking at the Skin Disease Education Foundation’s annual Las Vegas dermatology seminar, Dr. Duffin said that in her practice, this means that all patients being considered for systemic therapy get baseline labs as recommended by the drug manufacturer, and risk-focused monitoring for cardiovascular disease, infection, and cancer. Basically, she noted, the strategy is, “How can we be effective at monitoring and surveying for those ‘bad guys’– those complications of therapy?”
“Some of this is really about using common sense and being a good physician,” she added.
To obtain a pertinent history, it’s worthwhile to formulate a directed intake questionnaire, Dr. Duffin said. Screening items should include history of recurrent infections; hepatitis, HIV, and TB status and risk; travel history; cardiovascular history, including lipid status; history of any liver or kidney problems; history and risk of diabetes mellitus; depression; personal or family history of multiple sclerosis; and history of cancer, including skin cancer, and blood disorders.
The social history should include pregnancy status and birth control method for women (as appropriate); and alcohol use for all patients. Patients should be current with age-appropriate recommended immunizations and cancer screenings.
Obtaining baseline lab tests for all patients before they start treatment with a systemic agent affords the opportunity to detect common metabolic abnormalities, such as diabetes, liver or kidney disease, and dyslipidemia, as well as rare but serious blood abnormalities, Dr. Duffin said. This approach respects the fact that “there is no question that cardiovascular risk factors are overrepresented in the moderate to severe psoriasis population.”
Published TB screening guidelines have universally recommended screening before initiating a biologic agent with a tuberculin skin test, unless the patient has had the BCG vaccine or is immunosuppressed. From there, interpretation of the tine test and further screening requires knowing the patient: “It’s all about pretest probability,” said Dr. Duffin, adding that physicians should not be afraid to use infectious disease consultants to help them sort out tricky cases.
In terms of ongoing management, communication and coordination are key. Continue to ask patients about symptoms of psoriatic arthritis, and make sure that you work with the patient’s primary care provider so that cardiovascular risk management, malignancy screening, and immunizations don’t fall through the cracks, she advised.
“Have a high index of suspicion for infection,” including deep fungal infections, she added. Though the literature does not report frequent cases of serious fungal infections, “there’s no question cases are unreported or numbers can be unclear in the registries.” Patients should know early signs of infection and know to seek care right away.
Patients should know to defer live vaccines, and the physician managing the care of psoriasis should be included in the planning and management process for any type of surgery, to oversee biologic administration. While probably slight, the risk of infection should be weighed against the risk of loss of efficacy in psoriasis or psoriatic arthritis treatment, Dr. Duffin said.
The baseline risk of solid tumors is elevated in patients with psoriasis, but “is not definitively elevated in patients on biologics,” she commented. The risk of nonmelanoma skin cancer, however, is definitely elevated for patients on biologic therapies, so an annual skin survey is a must for these patients, she said.
“Should we be discussing immunizations with our patients? Absolutely,”she added. All patients need the influenza vaccine and the pneumococcal vaccine, and for live virus immunizations, the dermatologist should coordinate administration with the primary care physician.
Managing these risks effectively can make a big difference in quality of life for patients who are able to start and stay on systemic therapy, said Dr. Duffin. The many risks of not treating psoriasis appropriately include not only the significant psychosocial impact, but also progression of psoriatic arthritis, progression of skin disease, and increased risk of heart disease and other cardiovascular comorbidities. “Our job is to treat to patient satisfaction and the best possible quality of life. A lot of this is about anticipation and communication,” she said.
Dr. Duffin reported receiving research support from and consulting for Amgen, Eli Lilly, Janssen, Stiefel, AbbVie, Bristol-Myers Squibb, Celgene, Novartis, and XenoPort; consulting and being on the scientific advisory board for Pfizer; and being on the scientific advisory board for Novartis, Eli Lilly, Janssen, Celgene, and XenoPort.
SDEF and this news organization are owned by the same parent company.
On Twitter @karioakes
EXPERT ANALYSIS FROM SDEF LAS VEGAS DERMATOLOGY SEMINAR
VIDEO: Psoriasis, psoriatic arthritis improve with bariatric surgery
SAN FRANCISCO – It might be time to add psoriasis to the list of comorbidities bariatric surgery is likely to help.
New York University investigators have found a marked improvement in psoriasis and psoriatic arthritis following bariatric surgery, especially with severe disease. The more weight people lose, the better they do.
In an interview at the annual meeting of the American College of Rheumatology, investigator Dr. Soumya Reddy, codirector of NYU’s Psoriatic Arthritis Center in Manhattan, explained how the findings can be used in the clinic and their potential impact on bariatric surgery authorization.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO – It might be time to add psoriasis to the list of comorbidities bariatric surgery is likely to help.
New York University investigators have found a marked improvement in psoriasis and psoriatic arthritis following bariatric surgery, especially with severe disease. The more weight people lose, the better they do.
In an interview at the annual meeting of the American College of Rheumatology, investigator Dr. Soumya Reddy, codirector of NYU’s Psoriatic Arthritis Center in Manhattan, explained how the findings can be used in the clinic and their potential impact on bariatric surgery authorization.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO – It might be time to add psoriasis to the list of comorbidities bariatric surgery is likely to help.
New York University investigators have found a marked improvement in psoriasis and psoriatic arthritis following bariatric surgery, especially with severe disease. The more weight people lose, the better they do.
In an interview at the annual meeting of the American College of Rheumatology, investigator Dr. Soumya Reddy, codirector of NYU’s Psoriatic Arthritis Center in Manhattan, explained how the findings can be used in the clinic and their potential impact on bariatric surgery authorization.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE ACR ANNUAL MEETING
Stroke risk rose in autoimmune disease patients after herpes zoster
Stroke risk was 50% higher in the month after patients with autoimmune diseases developed herpes zoster, compared with the next 2-6 years, according to Dr. Leonard H. Calabrese.
“These data provide urgency for developing strategies to reduce the risk of varicella zoster virus in vulnerable immunosuppressed patients,” said Dr. Calabrese of the department of rheumatic and immunologic diseases at the Cleveland Clinic.
Immunosuppressive therapies increase the frequency and complexity of herpes zoster, which is a known risk factor for stroke. To examine the temporal relationship between herpes zoster and stroke among immunosuppressed patients, Dr. Calabrese and his associates studied Medicare data for almost 51,000 patients with new-onset herpes zoster who also had physician-diagnosed ankylosing spondylitis, inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis. The researchers excluded patients with a history of stroke.
In the multivariable analysis, stroke was 1.5 times more likely during the 6 months immediately after herpes zoster than in the 2-6 years after herpes zoster (95% confidence interval, 1.06-2.12). During this 6-month window, there were 9.8 strokes per 1,000 person-years, compared with 8.7 per 1,000 person-years in the 2-6 years after herpes zoster. Stroke risk also remained somewhat elevated during the entire year after herpes zoster (incidence rate ratio, 1.3; 95% CI, 1.05-1.61).
In general, stroke was more likely to occur among patients who were older, were receiving high-dose glucocorticoids, or had diabetes, hypertension, atrial fibrillation, or a history of transient ischemic attack, he said at the annual meeting of the American College of Rheumatology in San Francisco.
Dr. Calabrese disclosed relationships with Bristol-Myers Squibb, Crescendo, AbbVie, Genentech, Biogen, Pfizer, Sanofi-Aventis Pharmaceutical, and Johnson & Johnson. Two coauthors also disclosed relationships with several pharmaceutical companies. The other four coinvestigators had no disclosures.
Stroke risk was 50% higher in the month after patients with autoimmune diseases developed herpes zoster, compared with the next 2-6 years, according to Dr. Leonard H. Calabrese.
“These data provide urgency for developing strategies to reduce the risk of varicella zoster virus in vulnerable immunosuppressed patients,” said Dr. Calabrese of the department of rheumatic and immunologic diseases at the Cleveland Clinic.
Immunosuppressive therapies increase the frequency and complexity of herpes zoster, which is a known risk factor for stroke. To examine the temporal relationship between herpes zoster and stroke among immunosuppressed patients, Dr. Calabrese and his associates studied Medicare data for almost 51,000 patients with new-onset herpes zoster who also had physician-diagnosed ankylosing spondylitis, inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis. The researchers excluded patients with a history of stroke.
In the multivariable analysis, stroke was 1.5 times more likely during the 6 months immediately after herpes zoster than in the 2-6 years after herpes zoster (95% confidence interval, 1.06-2.12). During this 6-month window, there were 9.8 strokes per 1,000 person-years, compared with 8.7 per 1,000 person-years in the 2-6 years after herpes zoster. Stroke risk also remained somewhat elevated during the entire year after herpes zoster (incidence rate ratio, 1.3; 95% CI, 1.05-1.61).
In general, stroke was more likely to occur among patients who were older, were receiving high-dose glucocorticoids, or had diabetes, hypertension, atrial fibrillation, or a history of transient ischemic attack, he said at the annual meeting of the American College of Rheumatology in San Francisco.
Dr. Calabrese disclosed relationships with Bristol-Myers Squibb, Crescendo, AbbVie, Genentech, Biogen, Pfizer, Sanofi-Aventis Pharmaceutical, and Johnson & Johnson. Two coauthors also disclosed relationships with several pharmaceutical companies. The other four coinvestigators had no disclosures.
Stroke risk was 50% higher in the month after patients with autoimmune diseases developed herpes zoster, compared with the next 2-6 years, according to Dr. Leonard H. Calabrese.
“These data provide urgency for developing strategies to reduce the risk of varicella zoster virus in vulnerable immunosuppressed patients,” said Dr. Calabrese of the department of rheumatic and immunologic diseases at the Cleveland Clinic.
Immunosuppressive therapies increase the frequency and complexity of herpes zoster, which is a known risk factor for stroke. To examine the temporal relationship between herpes zoster and stroke among immunosuppressed patients, Dr. Calabrese and his associates studied Medicare data for almost 51,000 patients with new-onset herpes zoster who also had physician-diagnosed ankylosing spondylitis, inflammatory bowel disease, psoriasis, psoriatic arthritis, or rheumatoid arthritis. The researchers excluded patients with a history of stroke.
In the multivariable analysis, stroke was 1.5 times more likely during the 6 months immediately after herpes zoster than in the 2-6 years after herpes zoster (95% confidence interval, 1.06-2.12). During this 6-month window, there were 9.8 strokes per 1,000 person-years, compared with 8.7 per 1,000 person-years in the 2-6 years after herpes zoster. Stroke risk also remained somewhat elevated during the entire year after herpes zoster (incidence rate ratio, 1.3; 95% CI, 1.05-1.61).
In general, stroke was more likely to occur among patients who were older, were receiving high-dose glucocorticoids, or had diabetes, hypertension, atrial fibrillation, or a history of transient ischemic attack, he said at the annual meeting of the American College of Rheumatology in San Francisco.
Dr. Calabrese disclosed relationships with Bristol-Myers Squibb, Crescendo, AbbVie, Genentech, Biogen, Pfizer, Sanofi-Aventis Pharmaceutical, and Johnson & Johnson. Two coauthors also disclosed relationships with several pharmaceutical companies. The other four coinvestigators had no disclosures.
FROM THE ACR ANNUAL MEETING
Key clinical point: Stroke risk increased by 50% in the month after patients with autoimmune diseases had an episode of herpes zoster.
Major finding: The risk of stroke was 50% higher in the 6 months immediately after incident herpes zoster than 2-6 years after herpes zoster (95% CI, 1.06- 2.12).
Data source: An analysis of Medicare data for 50,929 patients with autoimmune disease and incident herpes zoster between 2006 and 2012.
Disclosures: Dr. Calabrese disclosed relationships with Bristol-Myers Squibb, Crescendo, AbbVie, Genentech, Biogen, Pfizer, Sanofi-Aventis Pharmaceutical, and Johnson & Johnson. Two coauthors also disclosed relationships with several pharmaceutical companies. The other four coinvestigators had no disclosures.
Shingles vaccine protection lasted 5-6 years in autoimmune disease patients
Protection against shingles appeared to wane between the fifth and sixth years after patients with autoimmune diseases received the live herpes zoster vaccine, according to a large retrospective cohort study presented at the annual meeting of the American College of Rheumatology.
In contrast, shingles risk remained fairly constant among unvaccinated patients, reported Dr. Huifeng Yun, assistant professor of epidemiology at the University of Alabama, Birmingham. Based on the findings, clinicians could consider revaccinating patients with autoimmune diseases about 5 years after their first live herpes zoster vaccine, she said.*
The long-term Shingles Prevention Study recently showed that the live herpes zoster vaccine is effective for about a decade among healthy older individuals, but the duration of protection for patients with autoimmune diseases was unclear, the investigators said. Therefore, they retrospectively studied Medicare data for more than 130,000 such patients between 2006 and 2012. About one-third of patients were vaccinated against herpes zoster, and 47% had rheumatoid arthritis, 32% had psoriasis, 21% had inflammatory bowel disease, 5% had psoriatic arthritis, and 1% had ankylosing spondylitis.
Rates of herpes zoster among vaccinated patients rose from 0.75 per 100 person-years during the first year after vaccination to 1.36 during the sixth year. In the adjusted analysis, vaccinated patients had about half the risk of herpes zoster, compared with unvaccinated patients, during year 1 (relative risk, 0.52; 95% confidence interval, 0.45-0.61) and remained substantially less likely to develop shingles until year 6, when the gap in risk between vaccinated and unvaccinated patients essentially closed (RR, 0.92; 95% CI, 0.45-1.86). There was no overall change in risk among unvaccinated patients during the study period, the investigators noted.
Dr. Yun disclosed a financial relationship with Amgen. The senior author and two coauthors also disclosed relationships with several pharmaceutical companies. Two investigators had no disclosures.
* Correction, 11/13/2015: The article previously misstated Dr. Yun's gender.
Protection against shingles appeared to wane between the fifth and sixth years after patients with autoimmune diseases received the live herpes zoster vaccine, according to a large retrospective cohort study presented at the annual meeting of the American College of Rheumatology.
In contrast, shingles risk remained fairly constant among unvaccinated patients, reported Dr. Huifeng Yun, assistant professor of epidemiology at the University of Alabama, Birmingham. Based on the findings, clinicians could consider revaccinating patients with autoimmune diseases about 5 years after their first live herpes zoster vaccine, she said.*
The long-term Shingles Prevention Study recently showed that the live herpes zoster vaccine is effective for about a decade among healthy older individuals, but the duration of protection for patients with autoimmune diseases was unclear, the investigators said. Therefore, they retrospectively studied Medicare data for more than 130,000 such patients between 2006 and 2012. About one-third of patients were vaccinated against herpes zoster, and 47% had rheumatoid arthritis, 32% had psoriasis, 21% had inflammatory bowel disease, 5% had psoriatic arthritis, and 1% had ankylosing spondylitis.
Rates of herpes zoster among vaccinated patients rose from 0.75 per 100 person-years during the first year after vaccination to 1.36 during the sixth year. In the adjusted analysis, vaccinated patients had about half the risk of herpes zoster, compared with unvaccinated patients, during year 1 (relative risk, 0.52; 95% confidence interval, 0.45-0.61) and remained substantially less likely to develop shingles until year 6, when the gap in risk between vaccinated and unvaccinated patients essentially closed (RR, 0.92; 95% CI, 0.45-1.86). There was no overall change in risk among unvaccinated patients during the study period, the investigators noted.
Dr. Yun disclosed a financial relationship with Amgen. The senior author and two coauthors also disclosed relationships with several pharmaceutical companies. Two investigators had no disclosures.
* Correction, 11/13/2015: The article previously misstated Dr. Yun's gender.
Protection against shingles appeared to wane between the fifth and sixth years after patients with autoimmune diseases received the live herpes zoster vaccine, according to a large retrospective cohort study presented at the annual meeting of the American College of Rheumatology.
In contrast, shingles risk remained fairly constant among unvaccinated patients, reported Dr. Huifeng Yun, assistant professor of epidemiology at the University of Alabama, Birmingham. Based on the findings, clinicians could consider revaccinating patients with autoimmune diseases about 5 years after their first live herpes zoster vaccine, she said.*
The long-term Shingles Prevention Study recently showed that the live herpes zoster vaccine is effective for about a decade among healthy older individuals, but the duration of protection for patients with autoimmune diseases was unclear, the investigators said. Therefore, they retrospectively studied Medicare data for more than 130,000 such patients between 2006 and 2012. About one-third of patients were vaccinated against herpes zoster, and 47% had rheumatoid arthritis, 32% had psoriasis, 21% had inflammatory bowel disease, 5% had psoriatic arthritis, and 1% had ankylosing spondylitis.
Rates of herpes zoster among vaccinated patients rose from 0.75 per 100 person-years during the first year after vaccination to 1.36 during the sixth year. In the adjusted analysis, vaccinated patients had about half the risk of herpes zoster, compared with unvaccinated patients, during year 1 (relative risk, 0.52; 95% confidence interval, 0.45-0.61) and remained substantially less likely to develop shingles until year 6, when the gap in risk between vaccinated and unvaccinated patients essentially closed (RR, 0.92; 95% CI, 0.45-1.86). There was no overall change in risk among unvaccinated patients during the study period, the investigators noted.
Dr. Yun disclosed a financial relationship with Amgen. The senior author and two coauthors also disclosed relationships with several pharmaceutical companies. Two investigators had no disclosures.
* Correction, 11/13/2015: The article previously misstated Dr. Yun's gender.
FROM THE ACR ANNUAL MEETING
Key clinical point: Protection against shingles appeared to wane between the fifth and sixth years after patients with autoimmune diseases received the live herpes zoster vaccine.
Major finding: The rate of herpes zoster rose from 0.75 per 100 person-years in the first year after vaccination to 1.36 in the sixth year, when it approached the rate among unvaccinated patients.
Data source: Retrospective analysis of 130,107 Medicare patients with autoimmune diseases between 2006 and 2012.
Disclosures: Dr. Yun disclosed a financial relationship with Amgen. The senior author and two coauthors also disclosed relationships with several pharmaceutical companies. Two investigators had no disclosures.