User login
More comorbidity with PsA form of spondyloarthritis
Psoriatic arthritis in patients with spondyloarthritis conveys an increased risk of comorbidities, especially cardiovascular and metabolic diseases, as compared with spondyloarthritis alone.
The association persisted even after adjusting for demographic factors, disease duration, and length of treatment, Dr. Naba Haque of the University of Leuven (Belgium) and associates wrote in the Journal of Rheumatology (2015 Dec 15. doi: 10.3899/jrheum.141359).
In an analysis of 518 patients in the spondyloarthritis database at the university, over half (54%) had comorbidities, and those with psoriatic spondyloarthropathy had significantly more comorbidities than those without psoriatic arthropathy (P less than .001). Cardiovascular comorbidities were most common, with an increased prevalence of hypertension, coronary artery disease, and hyperlipidemia in the patients with psoriatic spondyloarthropathy (odds ratios, 1.81, 1.39, and 1.60, respectively; P less than .001). The differences persisted after adjusting for confounders such as age, sex, disease duration, and treatment.
Twice as many patients with spondyloarthritis and psoriatic arthropathy met the criteria for metabolic syndrome as did patients without psoriatric arthropathy (10% vs. 5%, P = .03). The regression model showed a significant difference in the prevalence of diabetes in the PsA patients (OR, 1.35; 95% confidence interval, 1.17-1.56; P less than .001). Psoriatic arthropathy also was linked to an increased risk of cancer (12% vs. 6%; P less than .05); however, the authors wrote that “we can only state a possible positive association … this observation needs to be studied in further detail.”
The study was supported by an unrestricted grant from the Abbvie Chair for psoriatic arthritis research (Dr. Rik J. Lories and Dr. Kurt de Vlam).
Psoriatic arthritis in patients with spondyloarthritis conveys an increased risk of comorbidities, especially cardiovascular and metabolic diseases, as compared with spondyloarthritis alone.
The association persisted even after adjusting for demographic factors, disease duration, and length of treatment, Dr. Naba Haque of the University of Leuven (Belgium) and associates wrote in the Journal of Rheumatology (2015 Dec 15. doi: 10.3899/jrheum.141359).
In an analysis of 518 patients in the spondyloarthritis database at the university, over half (54%) had comorbidities, and those with psoriatic spondyloarthropathy had significantly more comorbidities than those without psoriatic arthropathy (P less than .001). Cardiovascular comorbidities were most common, with an increased prevalence of hypertension, coronary artery disease, and hyperlipidemia in the patients with psoriatic spondyloarthropathy (odds ratios, 1.81, 1.39, and 1.60, respectively; P less than .001). The differences persisted after adjusting for confounders such as age, sex, disease duration, and treatment.
Twice as many patients with spondyloarthritis and psoriatic arthropathy met the criteria for metabolic syndrome as did patients without psoriatric arthropathy (10% vs. 5%, P = .03). The regression model showed a significant difference in the prevalence of diabetes in the PsA patients (OR, 1.35; 95% confidence interval, 1.17-1.56; P less than .001). Psoriatic arthropathy also was linked to an increased risk of cancer (12% vs. 6%; P less than .05); however, the authors wrote that “we can only state a possible positive association … this observation needs to be studied in further detail.”
The study was supported by an unrestricted grant from the Abbvie Chair for psoriatic arthritis research (Dr. Rik J. Lories and Dr. Kurt de Vlam).
Psoriatic arthritis in patients with spondyloarthritis conveys an increased risk of comorbidities, especially cardiovascular and metabolic diseases, as compared with spondyloarthritis alone.
The association persisted even after adjusting for demographic factors, disease duration, and length of treatment, Dr. Naba Haque of the University of Leuven (Belgium) and associates wrote in the Journal of Rheumatology (2015 Dec 15. doi: 10.3899/jrheum.141359).
In an analysis of 518 patients in the spondyloarthritis database at the university, over half (54%) had comorbidities, and those with psoriatic spondyloarthropathy had significantly more comorbidities than those without psoriatic arthropathy (P less than .001). Cardiovascular comorbidities were most common, with an increased prevalence of hypertension, coronary artery disease, and hyperlipidemia in the patients with psoriatic spondyloarthropathy (odds ratios, 1.81, 1.39, and 1.60, respectively; P less than .001). The differences persisted after adjusting for confounders such as age, sex, disease duration, and treatment.
Twice as many patients with spondyloarthritis and psoriatic arthropathy met the criteria for metabolic syndrome as did patients without psoriatric arthropathy (10% vs. 5%, P = .03). The regression model showed a significant difference in the prevalence of diabetes in the PsA patients (OR, 1.35; 95% confidence interval, 1.17-1.56; P less than .001). Psoriatic arthropathy also was linked to an increased risk of cancer (12% vs. 6%; P less than .05); however, the authors wrote that “we can only state a possible positive association … this observation needs to be studied in further detail.”
The study was supported by an unrestricted grant from the Abbvie Chair for psoriatic arthritis research (Dr. Rik J. Lories and Dr. Kurt de Vlam).
FROM THE JOURNAL OF RHEUMATOLOGY
Key clinical point: People with psoriatic arthritis (PsA) have a significantly increased risk of cardiovascular and metabolic diseases, compared with people with non-PsA forms of spondyloarthritis (SpA).
Major finding: After correcting for risk factors, spondyloarthritis plus psoriatic arthritis was linked with an increased prevalence of hypertension, coronary artery disease, and hyperlipidemia (OR, 1.81, 1.39, and 1.60 respectively, P less than .001).
Data source: Cross-sectional study of 518 spondyloarthritis patients in a database at a university rheumatology department.
Disclosures: The study was supported by an unrestricted grant from the Abbvie Chair for psoriatic arthritis research (Dr. Lories and Dr. de Vlam).
Secukinumab cut ankylosing spondylitis symptoms in MEASURE trials
Secukinumab, an interleukin 17-A inhibitor approved for the treatment of moderate to severe psoriasis, significantly reduced the signs and symptoms of ankylosing spondylitis in two phase III trials, researchers reported Dec. 23 in the New England Journal of Medicine.
The results of the double-blind MEASURE 1 and MEASURE 2 trials extend the positive results of the phase II study, according to Dr. Dominique Baeten of the Academic Medical Center at the University of Amsterdam and his colleagues (N Engl J Med. 2015 Dec 23. doi: 10.1056/NEJMoa1505066).
“Although head-to-head trials would be required to fully assess the efficacy and safety of secukinumab versus TNF-inhibitors, the [20% improvement in Assessment of Spondyloarthritis International Society (ASAS20) response criteria] response rates achieved with secukinumab at week 16 in our studies were similar to those reported in phase III studies of anti-TNF agents in which most of the patients had not received previous anti-TNF therapy (response rates of 58% to 64% at weeks 12 to 24), even though 30% to 40% of the patients in our studies had had no response to previous anti-TNF treatment,” the authors wrote.
“Thus, secukinumab not only is effective in patients who have not received TNF agents previously but also may be effective in patients in whom previous anti-TNF treatment failed,” they added.
In MEASURE 1, 371 patients received intravenous secukinumab (10 mg/kg of body weight) or matched placebo at weeks 0, 2, and 4, followed by subcutaneous secukinumab (150 mg or 75 mg) or matched placebo every 4 weeks starting at week 8.
The study’s primary endpoint of ASAS20 response rates at week 16 were 61%, 60%, and 29% for subcutaneous secukinumab at doses of 150 mg and 75 mg and for placebo, respectively, (P less than .001 for both comparisons with placebo).
In MEASURE 2, 219 patients received subcutaneous secukinumab (150 mg or 75 mg) or matched placebo at baseline; at weeks 1, 2, and 3; and every 4 weeks starting at week 4.
At week 16, patients in the placebo group were randomly reassigned to subcutaneous secukinumab at a dose of 150 mg or 75 mg.
In this trial, ASAS20 rates were 61%, 41%, and 28% for subcutaneous secukinumab at doses of 150 mg and 75 mg and for placebo, respectively (P less than .001 for the 150-mg dose and P = .10 for the 75-mg dose).
The researchers noted that the ineffectiveness of the 75-mg dose in MEASURE 2 suggests that the efficacy of secukinumab at the 75-mg dose in MEASURE 1 may have been due to the greater exposure at week 16 as a result of the intravenous loading regimen, not to the 75-mg subcutaneous maintenance dose.
The safety profile of secukinumab in the present studies was consistent with previous studies of secukinumab for ankylosing spondylitis and moderate-to-severe plaque psoriasis, Dr. Baeten and his associates said.
During the entire treatment period, pooled exposure-adjusted incidence rates of grade 3 or 4 neutropenia, candida infections, and Crohn’s disease were 0.7, 0.9, and 0.7 cases per 100 patient-years, respectively, in secukinumab-treated patients.
Overall, the results suggest that interleukin-17A plays a role in the pathogenesis of ankylosing spondylitis, and they validate inhibition of this cytokine as a potential therapeutic approach, the study authors concluded.
The study was sponsored by Novartis Pharma. Dr. Baeten has received a grant from Novartis to study the impact of IL-17A blockade in experimental models of spondyloarthritis. He also has been a consultant for Novartis for the design and conduct of the secukinumab program in ankylosing spondylitis and psoriatic arthritis.
Secukinumab, an interleukin 17-A inhibitor approved for the treatment of moderate to severe psoriasis, significantly reduced the signs and symptoms of ankylosing spondylitis in two phase III trials, researchers reported Dec. 23 in the New England Journal of Medicine.
The results of the double-blind MEASURE 1 and MEASURE 2 trials extend the positive results of the phase II study, according to Dr. Dominique Baeten of the Academic Medical Center at the University of Amsterdam and his colleagues (N Engl J Med. 2015 Dec 23. doi: 10.1056/NEJMoa1505066).
“Although head-to-head trials would be required to fully assess the efficacy and safety of secukinumab versus TNF-inhibitors, the [20% improvement in Assessment of Spondyloarthritis International Society (ASAS20) response criteria] response rates achieved with secukinumab at week 16 in our studies were similar to those reported in phase III studies of anti-TNF agents in which most of the patients had not received previous anti-TNF therapy (response rates of 58% to 64% at weeks 12 to 24), even though 30% to 40% of the patients in our studies had had no response to previous anti-TNF treatment,” the authors wrote.
“Thus, secukinumab not only is effective in patients who have not received TNF agents previously but also may be effective in patients in whom previous anti-TNF treatment failed,” they added.
In MEASURE 1, 371 patients received intravenous secukinumab (10 mg/kg of body weight) or matched placebo at weeks 0, 2, and 4, followed by subcutaneous secukinumab (150 mg or 75 mg) or matched placebo every 4 weeks starting at week 8.
The study’s primary endpoint of ASAS20 response rates at week 16 were 61%, 60%, and 29% for subcutaneous secukinumab at doses of 150 mg and 75 mg and for placebo, respectively, (P less than .001 for both comparisons with placebo).
In MEASURE 2, 219 patients received subcutaneous secukinumab (150 mg or 75 mg) or matched placebo at baseline; at weeks 1, 2, and 3; and every 4 weeks starting at week 4.
At week 16, patients in the placebo group were randomly reassigned to subcutaneous secukinumab at a dose of 150 mg or 75 mg.
In this trial, ASAS20 rates were 61%, 41%, and 28% for subcutaneous secukinumab at doses of 150 mg and 75 mg and for placebo, respectively (P less than .001 for the 150-mg dose and P = .10 for the 75-mg dose).
The researchers noted that the ineffectiveness of the 75-mg dose in MEASURE 2 suggests that the efficacy of secukinumab at the 75-mg dose in MEASURE 1 may have been due to the greater exposure at week 16 as a result of the intravenous loading regimen, not to the 75-mg subcutaneous maintenance dose.
The safety profile of secukinumab in the present studies was consistent with previous studies of secukinumab for ankylosing spondylitis and moderate-to-severe plaque psoriasis, Dr. Baeten and his associates said.
During the entire treatment period, pooled exposure-adjusted incidence rates of grade 3 or 4 neutropenia, candida infections, and Crohn’s disease were 0.7, 0.9, and 0.7 cases per 100 patient-years, respectively, in secukinumab-treated patients.
Overall, the results suggest that interleukin-17A plays a role in the pathogenesis of ankylosing spondylitis, and they validate inhibition of this cytokine as a potential therapeutic approach, the study authors concluded.
The study was sponsored by Novartis Pharma. Dr. Baeten has received a grant from Novartis to study the impact of IL-17A blockade in experimental models of spondyloarthritis. He also has been a consultant for Novartis for the design and conduct of the secukinumab program in ankylosing spondylitis and psoriatic arthritis.
Secukinumab, an interleukin 17-A inhibitor approved for the treatment of moderate to severe psoriasis, significantly reduced the signs and symptoms of ankylosing spondylitis in two phase III trials, researchers reported Dec. 23 in the New England Journal of Medicine.
The results of the double-blind MEASURE 1 and MEASURE 2 trials extend the positive results of the phase II study, according to Dr. Dominique Baeten of the Academic Medical Center at the University of Amsterdam and his colleagues (N Engl J Med. 2015 Dec 23. doi: 10.1056/NEJMoa1505066).
“Although head-to-head trials would be required to fully assess the efficacy and safety of secukinumab versus TNF-inhibitors, the [20% improvement in Assessment of Spondyloarthritis International Society (ASAS20) response criteria] response rates achieved with secukinumab at week 16 in our studies were similar to those reported in phase III studies of anti-TNF agents in which most of the patients had not received previous anti-TNF therapy (response rates of 58% to 64% at weeks 12 to 24), even though 30% to 40% of the patients in our studies had had no response to previous anti-TNF treatment,” the authors wrote.
“Thus, secukinumab not only is effective in patients who have not received TNF agents previously but also may be effective in patients in whom previous anti-TNF treatment failed,” they added.
In MEASURE 1, 371 patients received intravenous secukinumab (10 mg/kg of body weight) or matched placebo at weeks 0, 2, and 4, followed by subcutaneous secukinumab (150 mg or 75 mg) or matched placebo every 4 weeks starting at week 8.
The study’s primary endpoint of ASAS20 response rates at week 16 were 61%, 60%, and 29% for subcutaneous secukinumab at doses of 150 mg and 75 mg and for placebo, respectively, (P less than .001 for both comparisons with placebo).
In MEASURE 2, 219 patients received subcutaneous secukinumab (150 mg or 75 mg) or matched placebo at baseline; at weeks 1, 2, and 3; and every 4 weeks starting at week 4.
At week 16, patients in the placebo group were randomly reassigned to subcutaneous secukinumab at a dose of 150 mg or 75 mg.
In this trial, ASAS20 rates were 61%, 41%, and 28% for subcutaneous secukinumab at doses of 150 mg and 75 mg and for placebo, respectively (P less than .001 for the 150-mg dose and P = .10 for the 75-mg dose).
The researchers noted that the ineffectiveness of the 75-mg dose in MEASURE 2 suggests that the efficacy of secukinumab at the 75-mg dose in MEASURE 1 may have been due to the greater exposure at week 16 as a result of the intravenous loading regimen, not to the 75-mg subcutaneous maintenance dose.
The safety profile of secukinumab in the present studies was consistent with previous studies of secukinumab for ankylosing spondylitis and moderate-to-severe plaque psoriasis, Dr. Baeten and his associates said.
During the entire treatment period, pooled exposure-adjusted incidence rates of grade 3 or 4 neutropenia, candida infections, and Crohn’s disease were 0.7, 0.9, and 0.7 cases per 100 patient-years, respectively, in secukinumab-treated patients.
Overall, the results suggest that interleukin-17A plays a role in the pathogenesis of ankylosing spondylitis, and they validate inhibition of this cytokine as a potential therapeutic approach, the study authors concluded.
The study was sponsored by Novartis Pharma. Dr. Baeten has received a grant from Novartis to study the impact of IL-17A blockade in experimental models of spondyloarthritis. He also has been a consultant for Novartis for the design and conduct of the secukinumab program in ankylosing spondylitis and psoriatic arthritis.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Interleukin-17A may play a role in ankylosing spondylitis, and secukinumab may prove to be an effective therapy for these patients.
Major finding: The primary endpoint of Assessment of Spondyloarthritis International Society (ASAS20) response rates at week 16 was met in both secukinumab groups in MEASURE 1 and in the group that received 150 mg of secukinumab subcutaneously in MEASURE 2.
Data source: Two double-blind, phase III studies: MEASURE 1 involving 371 patients with AS and MEASURE 2 involving 371 patients.
Disclosures: The studies were funded by Novartis Pharma. Dr. Baeten has received a grant from Novartis to study the impact of IL-17A blockade in experimental models of spondyloarthritis. He also has been a consultant for Novartis for the design and conduct of the secukinumab program in ankylosing spondylitis and psoriatic arthritis.
Aerosol foam product found effective for psoriasis vulgaris
An alcohol-free aerosol foam that contains a fixed combination of calcipotriene 0.005% plus betamethasone dipropionate 0.064% provided rapid itch relief and was well tolerated in patients with psoriasis vulgaris, according to results from a randomized, phase III study.
In earlier phase II studies, the product, which is being developed by Denmark-based LEO Pharma, was shown to be effective and had a safe tolerability profile, with no clinically relevant impact on the hypothalamic-pituitary-adrenal axis or on calcium metabolism. In an effort to confirm the efficacy and safety profile seen in the phase II trials, researchers led by Dr. Craig Leonardi, a dermatologist at Saint Louis (Mo.) University, performed the phase III study to compare the aerosol foam product with vehicle when applied once daily for up to 4 weeks in patients with psoriasis vulgaris.
Reporting in the December 2015 issue of the Journal of Drugs in Dermatology, Dr. Leonardi and his associates at 27 outpatient sites enrolled 426 patients aged 18 years and older between June and October of 2013 in a trial known as PSO-FAST (Cal/BD Foam in Psoriasis Vulgaris, a Four-Week, Vehicle-Controlled, Efficacy, and Safety Trial). The primary outcome was the proportion of patients who achieved treatment success at week 4, based on the physician’s global assessment, which was defined as clear or almost clear (for patients with at least moderate disease at baseline) or clear (for patients who had mild disease at baseline). Secondary outcomes included a modified (excluding head) Psoriasis Area and Severity Index (mPASI) and patient’s assessment of itch based on a visual analog scale(J. Drugs Dermatol. 2015;14[12]:1468-77).
Of the 426 patients, 323 received the aerosol foam product, while 103 received the vehicle. Their median age was 51 years, 59% were male, and their mean body mass index was 32.3 kg/m2. At week 4, the researchers found that significantly more patients in the aerosol foam group achieved treatment success, compared with those in the vehicle group (53.3% vs. 4.8%, respectively; odds ratio 30.3; P less than .001).
In addition, the mean mPASI score was significantly lower among patients in the aerosol foam group, compared with those in the vehicle group (a score of 2.0 vs. 5.5, for an adjusted difference of –3.3; P less than .001).
Among the 96% of patients who reported any level of itch at baseline, 36.8% in the aerosol foam group reported a 70% reduction in itch at day 3, compared with 24% of those in the vehicle group (OR 1.9; P = .018).
“This trial also demonstrated itch alleviation led to significant reductions in sleep loss, with 70.8% of patients using Cal/BD aerosol foam reporting a 70% reduction in itch-related sleep loss by week 4,” the researchers wrote.
No clinically significant changes in mean albumin-corrected serum calcium or urinary calcium to creatinine ratio was observed in either group. The researchers concluded that the Cal/DB aerosol foam product “may be a beneficial treatment option for those patients who are candidates for therapy with a superpotent steroid, but desire a therapeutic safety profile similar to that of a less potent steroid.”
An alcohol-free aerosol foam that contains a fixed combination of calcipotriene 0.005% plus betamethasone dipropionate 0.064% provided rapid itch relief and was well tolerated in patients with psoriasis vulgaris, according to results from a randomized, phase III study.
In earlier phase II studies, the product, which is being developed by Denmark-based LEO Pharma, was shown to be effective and had a safe tolerability profile, with no clinically relevant impact on the hypothalamic-pituitary-adrenal axis or on calcium metabolism. In an effort to confirm the efficacy and safety profile seen in the phase II trials, researchers led by Dr. Craig Leonardi, a dermatologist at Saint Louis (Mo.) University, performed the phase III study to compare the aerosol foam product with vehicle when applied once daily for up to 4 weeks in patients with psoriasis vulgaris.
Reporting in the December 2015 issue of the Journal of Drugs in Dermatology, Dr. Leonardi and his associates at 27 outpatient sites enrolled 426 patients aged 18 years and older between June and October of 2013 in a trial known as PSO-FAST (Cal/BD Foam in Psoriasis Vulgaris, a Four-Week, Vehicle-Controlled, Efficacy, and Safety Trial). The primary outcome was the proportion of patients who achieved treatment success at week 4, based on the physician’s global assessment, which was defined as clear or almost clear (for patients with at least moderate disease at baseline) or clear (for patients who had mild disease at baseline). Secondary outcomes included a modified (excluding head) Psoriasis Area and Severity Index (mPASI) and patient’s assessment of itch based on a visual analog scale(J. Drugs Dermatol. 2015;14[12]:1468-77).
Of the 426 patients, 323 received the aerosol foam product, while 103 received the vehicle. Their median age was 51 years, 59% were male, and their mean body mass index was 32.3 kg/m2. At week 4, the researchers found that significantly more patients in the aerosol foam group achieved treatment success, compared with those in the vehicle group (53.3% vs. 4.8%, respectively; odds ratio 30.3; P less than .001).
In addition, the mean mPASI score was significantly lower among patients in the aerosol foam group, compared with those in the vehicle group (a score of 2.0 vs. 5.5, for an adjusted difference of –3.3; P less than .001).
Among the 96% of patients who reported any level of itch at baseline, 36.8% in the aerosol foam group reported a 70% reduction in itch at day 3, compared with 24% of those in the vehicle group (OR 1.9; P = .018).
“This trial also demonstrated itch alleviation led to significant reductions in sleep loss, with 70.8% of patients using Cal/BD aerosol foam reporting a 70% reduction in itch-related sleep loss by week 4,” the researchers wrote.
No clinically significant changes in mean albumin-corrected serum calcium or urinary calcium to creatinine ratio was observed in either group. The researchers concluded that the Cal/DB aerosol foam product “may be a beneficial treatment option for those patients who are candidates for therapy with a superpotent steroid, but desire a therapeutic safety profile similar to that of a less potent steroid.”
An alcohol-free aerosol foam that contains a fixed combination of calcipotriene 0.005% plus betamethasone dipropionate 0.064% provided rapid itch relief and was well tolerated in patients with psoriasis vulgaris, according to results from a randomized, phase III study.
In earlier phase II studies, the product, which is being developed by Denmark-based LEO Pharma, was shown to be effective and had a safe tolerability profile, with no clinically relevant impact on the hypothalamic-pituitary-adrenal axis or on calcium metabolism. In an effort to confirm the efficacy and safety profile seen in the phase II trials, researchers led by Dr. Craig Leonardi, a dermatologist at Saint Louis (Mo.) University, performed the phase III study to compare the aerosol foam product with vehicle when applied once daily for up to 4 weeks in patients with psoriasis vulgaris.
Reporting in the December 2015 issue of the Journal of Drugs in Dermatology, Dr. Leonardi and his associates at 27 outpatient sites enrolled 426 patients aged 18 years and older between June and October of 2013 in a trial known as PSO-FAST (Cal/BD Foam in Psoriasis Vulgaris, a Four-Week, Vehicle-Controlled, Efficacy, and Safety Trial). The primary outcome was the proportion of patients who achieved treatment success at week 4, based on the physician’s global assessment, which was defined as clear or almost clear (for patients with at least moderate disease at baseline) or clear (for patients who had mild disease at baseline). Secondary outcomes included a modified (excluding head) Psoriasis Area and Severity Index (mPASI) and patient’s assessment of itch based on a visual analog scale(J. Drugs Dermatol. 2015;14[12]:1468-77).
Of the 426 patients, 323 received the aerosol foam product, while 103 received the vehicle. Their median age was 51 years, 59% were male, and their mean body mass index was 32.3 kg/m2. At week 4, the researchers found that significantly more patients in the aerosol foam group achieved treatment success, compared with those in the vehicle group (53.3% vs. 4.8%, respectively; odds ratio 30.3; P less than .001).
In addition, the mean mPASI score was significantly lower among patients in the aerosol foam group, compared with those in the vehicle group (a score of 2.0 vs. 5.5, for an adjusted difference of –3.3; P less than .001).
Among the 96% of patients who reported any level of itch at baseline, 36.8% in the aerosol foam group reported a 70% reduction in itch at day 3, compared with 24% of those in the vehicle group (OR 1.9; P = .018).
“This trial also demonstrated itch alleviation led to significant reductions in sleep loss, with 70.8% of patients using Cal/BD aerosol foam reporting a 70% reduction in itch-related sleep loss by week 4,” the researchers wrote.
No clinically significant changes in mean albumin-corrected serum calcium or urinary calcium to creatinine ratio was observed in either group. The researchers concluded that the Cal/DB aerosol foam product “may be a beneficial treatment option for those patients who are candidates for therapy with a superpotent steroid, but desire a therapeutic safety profile similar to that of a less potent steroid.”
FROM JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point: A foam product containing calcipotriene 0.005% plus betamethasone dipropionate 0.064% was found to be safe and effective for patients with psoriasis vulgaris.
Major finding: At week 4, a significantly greater number of patients in the aerosol foam group achieved treatment success, compared with those in the vehicle group (53.3% vs. 4.8%, respectively; OR 30.3; P less than .001).
Data source: A randomized, phase III trial compared an aerosol foam product containing calcipotriene 0.005% plus betamethasone dipropionate 0.064% with vehicle, applied once daily for up to 4 weeks in 426 patients with psoriasis vulgaris.
Disclosures: Dr. Leonardi reported that he has been a consultant for LEO Pharma. All of the other authors disclosed having current or former ties to LEO Pharma, including two who are currently employed by the company. Dr. Leonardi and many of the other study authors also reported having numerous ties to other pharmaceutical companies.
‘Cytokine converter’ cells alleviate psoriasis in mice
Implantation of cells designed to sense the presence of proinflammatory cytokines and subsequently produce and deliver anti-inflammatory molecules prevented flares of psoriasis in mice, according to Lina Schukur of the department of Biosystems Science and Engineering, ETH Zurich, in Basel, Switzerland, and associates.
The investigators designed and engineered human cells that they explain “sequentially detected elevated TNF [tumor necrosis factor] and IL-22 [interleukin-22] levels from a psoriatic flare and, in response, produced therapeutic doses of IL-4 [interleukin-4] and IL-10 [interleukin-10].” TNF and IL-22 are proinflammatory cytokines, which have been found to be upregulated in patients with active psoriasis; and IL-4 and IL-10 are immunomodulatory cytokines that have been shown to induce rapid improvements in people with psoriasis in phase II clinical trials, they noted.
The researchers implanted these designer cells in mice with psoriasis-like lesions, which showed that this “antipsoriatic cytokine converter network” was able to improve skin lesions “and restore dermal tissue morphology.” They also found that the cells were responsive in blood samples from psoriasis patients, which suggested that the cytokine converter “is sufficiently sensitive to detect circulating TNF and IL-22 in humans.”
“These synthetic circuits, which program designer cells to process complex metabolic information, open the door to autonomously prevent, attenuate, or reset acute or chronic medical conditions without constant injections of drugs or cumbersome dosing schedules, and thus provide a new opportunity for personalized medicine,” the investigators wrote.
An editor’s summary of the study states that, “in demonstrating that the converter cells were responsive to blood from psoriasis patients, the authors suggest that synthetic biology may be ready to autonomously flip therapeutic switches in people and later take on other diseases with defined disease indicators.”
The full study, published on Dec. 16, 2015, is available in Science Translational Medicine at http://stm.sciencemag.org/content/7/318/318ra201.
Implantation of cells designed to sense the presence of proinflammatory cytokines and subsequently produce and deliver anti-inflammatory molecules prevented flares of psoriasis in mice, according to Lina Schukur of the department of Biosystems Science and Engineering, ETH Zurich, in Basel, Switzerland, and associates.
The investigators designed and engineered human cells that they explain “sequentially detected elevated TNF [tumor necrosis factor] and IL-22 [interleukin-22] levels from a psoriatic flare and, in response, produced therapeutic doses of IL-4 [interleukin-4] and IL-10 [interleukin-10].” TNF and IL-22 are proinflammatory cytokines, which have been found to be upregulated in patients with active psoriasis; and IL-4 and IL-10 are immunomodulatory cytokines that have been shown to induce rapid improvements in people with psoriasis in phase II clinical trials, they noted.
The researchers implanted these designer cells in mice with psoriasis-like lesions, which showed that this “antipsoriatic cytokine converter network” was able to improve skin lesions “and restore dermal tissue morphology.” They also found that the cells were responsive in blood samples from psoriasis patients, which suggested that the cytokine converter “is sufficiently sensitive to detect circulating TNF and IL-22 in humans.”
“These synthetic circuits, which program designer cells to process complex metabolic information, open the door to autonomously prevent, attenuate, or reset acute or chronic medical conditions without constant injections of drugs or cumbersome dosing schedules, and thus provide a new opportunity for personalized medicine,” the investigators wrote.
An editor’s summary of the study states that, “in demonstrating that the converter cells were responsive to blood from psoriasis patients, the authors suggest that synthetic biology may be ready to autonomously flip therapeutic switches in people and later take on other diseases with defined disease indicators.”
The full study, published on Dec. 16, 2015, is available in Science Translational Medicine at http://stm.sciencemag.org/content/7/318/318ra201.
Implantation of cells designed to sense the presence of proinflammatory cytokines and subsequently produce and deliver anti-inflammatory molecules prevented flares of psoriasis in mice, according to Lina Schukur of the department of Biosystems Science and Engineering, ETH Zurich, in Basel, Switzerland, and associates.
The investigators designed and engineered human cells that they explain “sequentially detected elevated TNF [tumor necrosis factor] and IL-22 [interleukin-22] levels from a psoriatic flare and, in response, produced therapeutic doses of IL-4 [interleukin-4] and IL-10 [interleukin-10].” TNF and IL-22 are proinflammatory cytokines, which have been found to be upregulated in patients with active psoriasis; and IL-4 and IL-10 are immunomodulatory cytokines that have been shown to induce rapid improvements in people with psoriasis in phase II clinical trials, they noted.
The researchers implanted these designer cells in mice with psoriasis-like lesions, which showed that this “antipsoriatic cytokine converter network” was able to improve skin lesions “and restore dermal tissue morphology.” They also found that the cells were responsive in blood samples from psoriasis patients, which suggested that the cytokine converter “is sufficiently sensitive to detect circulating TNF and IL-22 in humans.”
“These synthetic circuits, which program designer cells to process complex metabolic information, open the door to autonomously prevent, attenuate, or reset acute or chronic medical conditions without constant injections of drugs or cumbersome dosing schedules, and thus provide a new opportunity for personalized medicine,” the investigators wrote.
An editor’s summary of the study states that, “in demonstrating that the converter cells were responsive to blood from psoriasis patients, the authors suggest that synthetic biology may be ready to autonomously flip therapeutic switches in people and later take on other diseases with defined disease indicators.”
The full study, published on Dec. 16, 2015, is available in Science Translational Medicine at http://stm.sciencemag.org/content/7/318/318ra201.
FROM SCIENCE TRANSLATIONAL MEDICINE
Poor response to third anti-TNF agent seen in most psoriatic arthritis patients
Patients with psoriatic arthritis who don’t respond to or cannot tolerate two different anti–tumor necrosis factor (TNF) agents are likely to respond poorly, if at all, to a third one, according to findings from a prospective, open-label, longitudinal study.
Dr. Lars Erik Kristensen of the department of rheumatology, Parker Institute, Copenhagen, and the department of rheumatology, Lund (Sweden) University Hospital, and his coinvestigators assessed treatment responses in patients treated at 11 European rheumatology centers during a 9-year period to build on the “rather sparse” data concerning second or third courses of anti-TNF treatment in psoriatic arthritis patients. “Our results suggest that other therapeutic options be considered after two courses of anti-TNF treatment have failed,” such as biological disease-modifying antirheumatic drugs that have different modes of action, they wrote.
The study participants were 217 patients with psoriatic arthritis who were switching from one anti-TNF agent to another and 57 who had tried two anti-TNF agents and were switching to a third. The drugs included etanercept, adalimumab, certolizumab pegol, golimumab, and infliximab.
In general, the treatment response rates among patients trying their second agent were markedly greater than those of patients trying their third. Nearly half (47%) of first-time switchers met the primary outcome measure – an ACR 20 response at 3 months – compared with only 22% of second-time switchers, the investigators said (J Rheumatol. 2015 Dec 1. doi: 10.3899/jrheum.150744).
The median drug survival (time on treatment) was 64 months for the first group, compared with only 14 months for the second group. The estimated 5-year drug survival was 51% for patients trying their second anti-TNF agent, compared with only 23% for patients trying their third.
This study was supported by the Osterlund Foundation, the Kock Foundation, the King Gustav V 80-Year Fund, Lund University Hospital, the Reumatikerforbundet, and the Oak Foundation. No information was available regarding Dr. Kristensen’s and his associates’ financial disclosures.
Patients with psoriatic arthritis who don’t respond to or cannot tolerate two different anti–tumor necrosis factor (TNF) agents are likely to respond poorly, if at all, to a third one, according to findings from a prospective, open-label, longitudinal study.
Dr. Lars Erik Kristensen of the department of rheumatology, Parker Institute, Copenhagen, and the department of rheumatology, Lund (Sweden) University Hospital, and his coinvestigators assessed treatment responses in patients treated at 11 European rheumatology centers during a 9-year period to build on the “rather sparse” data concerning second or third courses of anti-TNF treatment in psoriatic arthritis patients. “Our results suggest that other therapeutic options be considered after two courses of anti-TNF treatment have failed,” such as biological disease-modifying antirheumatic drugs that have different modes of action, they wrote.
The study participants were 217 patients with psoriatic arthritis who were switching from one anti-TNF agent to another and 57 who had tried two anti-TNF agents and were switching to a third. The drugs included etanercept, adalimumab, certolizumab pegol, golimumab, and infliximab.
In general, the treatment response rates among patients trying their second agent were markedly greater than those of patients trying their third. Nearly half (47%) of first-time switchers met the primary outcome measure – an ACR 20 response at 3 months – compared with only 22% of second-time switchers, the investigators said (J Rheumatol. 2015 Dec 1. doi: 10.3899/jrheum.150744).
The median drug survival (time on treatment) was 64 months for the first group, compared with only 14 months for the second group. The estimated 5-year drug survival was 51% for patients trying their second anti-TNF agent, compared with only 23% for patients trying their third.
This study was supported by the Osterlund Foundation, the Kock Foundation, the King Gustav V 80-Year Fund, Lund University Hospital, the Reumatikerforbundet, and the Oak Foundation. No information was available regarding Dr. Kristensen’s and his associates’ financial disclosures.
Patients with psoriatic arthritis who don’t respond to or cannot tolerate two different anti–tumor necrosis factor (TNF) agents are likely to respond poorly, if at all, to a third one, according to findings from a prospective, open-label, longitudinal study.
Dr. Lars Erik Kristensen of the department of rheumatology, Parker Institute, Copenhagen, and the department of rheumatology, Lund (Sweden) University Hospital, and his coinvestigators assessed treatment responses in patients treated at 11 European rheumatology centers during a 9-year period to build on the “rather sparse” data concerning second or third courses of anti-TNF treatment in psoriatic arthritis patients. “Our results suggest that other therapeutic options be considered after two courses of anti-TNF treatment have failed,” such as biological disease-modifying antirheumatic drugs that have different modes of action, they wrote.
The study participants were 217 patients with psoriatic arthritis who were switching from one anti-TNF agent to another and 57 who had tried two anti-TNF agents and were switching to a third. The drugs included etanercept, adalimumab, certolizumab pegol, golimumab, and infliximab.
In general, the treatment response rates among patients trying their second agent were markedly greater than those of patients trying their third. Nearly half (47%) of first-time switchers met the primary outcome measure – an ACR 20 response at 3 months – compared with only 22% of second-time switchers, the investigators said (J Rheumatol. 2015 Dec 1. doi: 10.3899/jrheum.150744).
The median drug survival (time on treatment) was 64 months for the first group, compared with only 14 months for the second group. The estimated 5-year drug survival was 51% for patients trying their second anti-TNF agent, compared with only 23% for patients trying their third.
This study was supported by the Osterlund Foundation, the Kock Foundation, the King Gustav V 80-Year Fund, Lund University Hospital, the Reumatikerforbundet, and the Oak Foundation. No information was available regarding Dr. Kristensen’s and his associates’ financial disclosures.
FROM JOURNAL OF RHEUMATOLOGY
Key clinical point: Patients with psoriatic arthritis who don’t respond to or cannot tolerate two different anti-TNF agents are likely to respond poorly, if at all, to a third one.
Major finding: 47% of first-time switchers met the primary outcome measure – an ACR 20 response at 3 months – compared with only 22% of second-time switchers.
Data source: A prospective, open-label, longitudinal study involving 217 patients who switched anti-TNF therapy once and 57 who switched twice during a 9-year period.
Disclosures: This study was supported by the Osterlund Foundation, the Kock Foundation, the King Gustav V 80-Year Fund, Lund University Hospital, the Reumatikerforbundet, and the Oak Foundation. No information was available regarding Dr. Kristensen’s and his associates’ financial disclosures.
Genetic differences may help predict progression of PsC to PsA
Dermatology researchers have uncovered differences in the genetic architecture of psoriatic arthritis (PsA) and cutaneous psoriasis (PsC).
Dr. Philip E. Stuart of the University of Michigan, Ann Arbor, and his colleagues from 32 institutions in the United States, Europe, and Canada, carried out a genome-wide association study (GWAS) of 1,430 patients with psoriatic arthritis and 1,417 healthy controls. They then combined results of their GWAS with five published studies of psoriasis associations (three GWASs and two targeted studies), comprising 3,061 psoriatic arthritis patients, 3,110 cutaneous-only psoriasis patients, and 9,273 psoriasis vulgaris patients, and 13,670 healthy controls of European descent.
The work, published in the American Journal of Human Genetics, identified five regions of significance in the PsA GWAS. These were all in known risk regions for PsA, with the strongest signals near HLA-B, IL12B, TRAF3IP2, TNIP1, and TYK2.
Looking across all studies, researchers detected 10 regions associated with PsA and 11 with PsC at genome-wide significance. Several of these association signals (IFNLR1, IFIH1, NFKBIA for PsA, and TJFRSF9, LCE3C/B, TRAF3IP2, IL23A, and NFKBIA for PsC) had not previously achieved such significance. Researchers also identified a genetic variant associated with psoriasis vulgaris (PsV) near CDKAL1, and other variants that were more strongly associated with either PsA or PsC.
“These results provide insights into the pathogenic similarities and differences between PsC and PsA,” the authors wrote. Identifying the causative variants driving the observed associations “will aid prediction and therapy of psoriasis and its cutaneous and joint manifestations.”
Read the article in the American Journal of Human Genetics (2015 Dec 3;97:816-36).
Dermatology researchers have uncovered differences in the genetic architecture of psoriatic arthritis (PsA) and cutaneous psoriasis (PsC).
Dr. Philip E. Stuart of the University of Michigan, Ann Arbor, and his colleagues from 32 institutions in the United States, Europe, and Canada, carried out a genome-wide association study (GWAS) of 1,430 patients with psoriatic arthritis and 1,417 healthy controls. They then combined results of their GWAS with five published studies of psoriasis associations (three GWASs and two targeted studies), comprising 3,061 psoriatic arthritis patients, 3,110 cutaneous-only psoriasis patients, and 9,273 psoriasis vulgaris patients, and 13,670 healthy controls of European descent.
The work, published in the American Journal of Human Genetics, identified five regions of significance in the PsA GWAS. These were all in known risk regions for PsA, with the strongest signals near HLA-B, IL12B, TRAF3IP2, TNIP1, and TYK2.
Looking across all studies, researchers detected 10 regions associated with PsA and 11 with PsC at genome-wide significance. Several of these association signals (IFNLR1, IFIH1, NFKBIA for PsA, and TJFRSF9, LCE3C/B, TRAF3IP2, IL23A, and NFKBIA for PsC) had not previously achieved such significance. Researchers also identified a genetic variant associated with psoriasis vulgaris (PsV) near CDKAL1, and other variants that were more strongly associated with either PsA or PsC.
“These results provide insights into the pathogenic similarities and differences between PsC and PsA,” the authors wrote. Identifying the causative variants driving the observed associations “will aid prediction and therapy of psoriasis and its cutaneous and joint manifestations.”
Read the article in the American Journal of Human Genetics (2015 Dec 3;97:816-36).
Dermatology researchers have uncovered differences in the genetic architecture of psoriatic arthritis (PsA) and cutaneous psoriasis (PsC).
Dr. Philip E. Stuart of the University of Michigan, Ann Arbor, and his colleagues from 32 institutions in the United States, Europe, and Canada, carried out a genome-wide association study (GWAS) of 1,430 patients with psoriatic arthritis and 1,417 healthy controls. They then combined results of their GWAS with five published studies of psoriasis associations (three GWASs and two targeted studies), comprising 3,061 psoriatic arthritis patients, 3,110 cutaneous-only psoriasis patients, and 9,273 psoriasis vulgaris patients, and 13,670 healthy controls of European descent.
The work, published in the American Journal of Human Genetics, identified five regions of significance in the PsA GWAS. These were all in known risk regions for PsA, with the strongest signals near HLA-B, IL12B, TRAF3IP2, TNIP1, and TYK2.
Looking across all studies, researchers detected 10 regions associated with PsA and 11 with PsC at genome-wide significance. Several of these association signals (IFNLR1, IFIH1, NFKBIA for PsA, and TJFRSF9, LCE3C/B, TRAF3IP2, IL23A, and NFKBIA for PsC) had not previously achieved such significance. Researchers also identified a genetic variant associated with psoriasis vulgaris (PsV) near CDKAL1, and other variants that were more strongly associated with either PsA or PsC.
“These results provide insights into the pathogenic similarities and differences between PsC and PsA,” the authors wrote. Identifying the causative variants driving the observed associations “will aid prediction and therapy of psoriasis and its cutaneous and joint manifestations.”
Read the article in the American Journal of Human Genetics (2015 Dec 3;97:816-36).
FROM AMERICAN JOURNAL OF HUMAN GENETICS
EADV: Pursuing ‘clear’ in psoriasis worthwhile, expert says
COPENHAGEN – It is worthwhile to push harder for complete skin clearance in a psoriasis patient whose treatment regimen provides a Physician Global Assessment score of 1, or “almost clear,” according to Dr. Kristian Reich.
He presented an analysis of pooled data from the active treatment arms of three 12-week phase III clinical trials of the investigational interleukin-17 inhibitor brodalumab. The 1,260 patients with a static Physician Global Assessment (PGA) score of 1, or almost clear, at week 12 had significantly greater residual disease in terms of involved body surface area, fewer symptom-free days, and greater health-related quality of life impairment than did the 1,097 participants who achieved a static PGA of 0.
“These results suggest that clear versus almost clear represents meaningfully different levels of disease,” Dr. Reich of Dermatologikum Hamburg (Germany) said at the annual congress of the European Academy of Dermatology and Venereology.
The newer-generation biologics that target IL-17 or IL-23 provide a significantly greater rate of complete skin clearance than do the older anti–tumor necrosis factor biologic agents. But the newer biologics are also typically costlier and have a shorter safety record. So the question arises: Does the residual disease that defines a static PGA of 1 matter to patients in terms of quality of life and days free of symptoms? Or do they want even better results, if achievable? This is what Dr. Reich and his coworkers set out to answer.
Thirty percent of patients with a static PGA of 1 had clinically significant residual disease at week 12 as defined by a body surface area involvement of 6% or greater, as did 0.3% of those with a static PGA of 0. Fourteen percent of patients with a static PGA of 1 had a Dermatology Life Quality Index score of 6 or more, indicative of at least moderate impairment in quality of life, compared with 6.4% of psoriasis patients with a static PGA of 0.
Moreover, 42% of patients with a static PGA of 0 were 100% symptom free every day from week 10 to week 12 of the study as defined by a score of 0 on the eight-item Psoriasis Symptom Inventory, compared with 12% of patients with a static PGA of 1.
Dr. Reich reported receiving research support from Amgen, which funded the study.
COPENHAGEN – It is worthwhile to push harder for complete skin clearance in a psoriasis patient whose treatment regimen provides a Physician Global Assessment score of 1, or “almost clear,” according to Dr. Kristian Reich.
He presented an analysis of pooled data from the active treatment arms of three 12-week phase III clinical trials of the investigational interleukin-17 inhibitor brodalumab. The 1,260 patients with a static Physician Global Assessment (PGA) score of 1, or almost clear, at week 12 had significantly greater residual disease in terms of involved body surface area, fewer symptom-free days, and greater health-related quality of life impairment than did the 1,097 participants who achieved a static PGA of 0.
“These results suggest that clear versus almost clear represents meaningfully different levels of disease,” Dr. Reich of Dermatologikum Hamburg (Germany) said at the annual congress of the European Academy of Dermatology and Venereology.
The newer-generation biologics that target IL-17 or IL-23 provide a significantly greater rate of complete skin clearance than do the older anti–tumor necrosis factor biologic agents. But the newer biologics are also typically costlier and have a shorter safety record. So the question arises: Does the residual disease that defines a static PGA of 1 matter to patients in terms of quality of life and days free of symptoms? Or do they want even better results, if achievable? This is what Dr. Reich and his coworkers set out to answer.
Thirty percent of patients with a static PGA of 1 had clinically significant residual disease at week 12 as defined by a body surface area involvement of 6% or greater, as did 0.3% of those with a static PGA of 0. Fourteen percent of patients with a static PGA of 1 had a Dermatology Life Quality Index score of 6 or more, indicative of at least moderate impairment in quality of life, compared with 6.4% of psoriasis patients with a static PGA of 0.
Moreover, 42% of patients with a static PGA of 0 were 100% symptom free every day from week 10 to week 12 of the study as defined by a score of 0 on the eight-item Psoriasis Symptom Inventory, compared with 12% of patients with a static PGA of 1.
Dr. Reich reported receiving research support from Amgen, which funded the study.
COPENHAGEN – It is worthwhile to push harder for complete skin clearance in a psoriasis patient whose treatment regimen provides a Physician Global Assessment score of 1, or “almost clear,” according to Dr. Kristian Reich.
He presented an analysis of pooled data from the active treatment arms of three 12-week phase III clinical trials of the investigational interleukin-17 inhibitor brodalumab. The 1,260 patients with a static Physician Global Assessment (PGA) score of 1, or almost clear, at week 12 had significantly greater residual disease in terms of involved body surface area, fewer symptom-free days, and greater health-related quality of life impairment than did the 1,097 participants who achieved a static PGA of 0.
“These results suggest that clear versus almost clear represents meaningfully different levels of disease,” Dr. Reich of Dermatologikum Hamburg (Germany) said at the annual congress of the European Academy of Dermatology and Venereology.
The newer-generation biologics that target IL-17 or IL-23 provide a significantly greater rate of complete skin clearance than do the older anti–tumor necrosis factor biologic agents. But the newer biologics are also typically costlier and have a shorter safety record. So the question arises: Does the residual disease that defines a static PGA of 1 matter to patients in terms of quality of life and days free of symptoms? Or do they want even better results, if achievable? This is what Dr. Reich and his coworkers set out to answer.
Thirty percent of patients with a static PGA of 1 had clinically significant residual disease at week 12 as defined by a body surface area involvement of 6% or greater, as did 0.3% of those with a static PGA of 0. Fourteen percent of patients with a static PGA of 1 had a Dermatology Life Quality Index score of 6 or more, indicative of at least moderate impairment in quality of life, compared with 6.4% of psoriasis patients with a static PGA of 0.
Moreover, 42% of patients with a static PGA of 0 were 100% symptom free every day from week 10 to week 12 of the study as defined by a score of 0 on the eight-item Psoriasis Symptom Inventory, compared with 12% of patients with a static PGA of 1.
Dr. Reich reported receiving research support from Amgen, which funded the study.
AT THE EADV CONGRESS
Key clinical point: A Physician Global Assessment of “clear” is worth striving for in psoriasis patients because it represents a clinically important lesser level of disease than “almost clear.”
Major finding: Fourteen percent of psoriasis patients with an on-treatment physician assessment of “almost clear” had at least moderately impaired health care quality of life, compared with 6.4% of those rated “clear.”
Data source: A secondary pooled analysis of quality of life–related outcomes among psoriasis patients rated “clear” as opposed to “almost clear” at week 12 of three phase III double-blind randomized trials of the interleukin-17 inhibitor brodalumab.
Disclosures: The presenter reported receiving research support from Amgen, which sponsored the study.
Anti-TNF therapy can continue for IBD patients with skin lesions
Inflammatory bowel disease (IBD) patients who experience skin lesions during anti–tumor necrosis factor therapy do not usually need to stop treatment, according to Isabelle Cleynen, Ph.D., of the University of Leuven (Belgium) and her associates.
In their retrospective study of 917 IBD patients who started treatment with infliximab at the University Hospitals Leuven between December 1994 and January 2009, 264 developed skin lesions during the follow-up period. The most common type was psoriasiform eczema, in 30.6% of the patients with lesions. Other common types included eczema (in 23.5%), xerosis cutis (10.6%), palmoplantar pustulosis (5.3%), and psoriasis (3.8%). Median cumulative doses and trough levels of infliximab were similar in people who developed skin lesions and those who did not.
Just over half of patients with skin lesions received only topical treatment, 1.9% received only systemic treatment, 28% received both, and 19.3% of patients required no specific treatment. Almost 11% of patients who developed skin lesions were forced to stop therapy. Reasons for stopping treatment included an intolerable location of lesions, concomitant itching or pain, recurring episodes, and concomitant arthralgia.
“Knowledge of the diagnostic and therapeutic criteria and the clinical course of these lesions should assist in their management. With referral to a dedicated dermatologist, most lesions can be treated and the need for interruption of anti-TNF therapy is rare,” the investigators concluded.
Find the full study in Annals of Internal Medicine (doi: 10.7326/M15-0729).
Inflammatory bowel disease (IBD) patients who experience skin lesions during anti–tumor necrosis factor therapy do not usually need to stop treatment, according to Isabelle Cleynen, Ph.D., of the University of Leuven (Belgium) and her associates.
In their retrospective study of 917 IBD patients who started treatment with infliximab at the University Hospitals Leuven between December 1994 and January 2009, 264 developed skin lesions during the follow-up period. The most common type was psoriasiform eczema, in 30.6% of the patients with lesions. Other common types included eczema (in 23.5%), xerosis cutis (10.6%), palmoplantar pustulosis (5.3%), and psoriasis (3.8%). Median cumulative doses and trough levels of infliximab were similar in people who developed skin lesions and those who did not.
Just over half of patients with skin lesions received only topical treatment, 1.9% received only systemic treatment, 28% received both, and 19.3% of patients required no specific treatment. Almost 11% of patients who developed skin lesions were forced to stop therapy. Reasons for stopping treatment included an intolerable location of lesions, concomitant itching or pain, recurring episodes, and concomitant arthralgia.
“Knowledge of the diagnostic and therapeutic criteria and the clinical course of these lesions should assist in their management. With referral to a dedicated dermatologist, most lesions can be treated and the need for interruption of anti-TNF therapy is rare,” the investigators concluded.
Find the full study in Annals of Internal Medicine (doi: 10.7326/M15-0729).
Inflammatory bowel disease (IBD) patients who experience skin lesions during anti–tumor necrosis factor therapy do not usually need to stop treatment, according to Isabelle Cleynen, Ph.D., of the University of Leuven (Belgium) and her associates.
In their retrospective study of 917 IBD patients who started treatment with infliximab at the University Hospitals Leuven between December 1994 and January 2009, 264 developed skin lesions during the follow-up period. The most common type was psoriasiform eczema, in 30.6% of the patients with lesions. Other common types included eczema (in 23.5%), xerosis cutis (10.6%), palmoplantar pustulosis (5.3%), and psoriasis (3.8%). Median cumulative doses and trough levels of infliximab were similar in people who developed skin lesions and those who did not.
Just over half of patients with skin lesions received only topical treatment, 1.9% received only systemic treatment, 28% received both, and 19.3% of patients required no specific treatment. Almost 11% of patients who developed skin lesions were forced to stop therapy. Reasons for stopping treatment included an intolerable location of lesions, concomitant itching or pain, recurring episodes, and concomitant arthralgia.
“Knowledge of the diagnostic and therapeutic criteria and the clinical course of these lesions should assist in their management. With referral to a dedicated dermatologist, most lesions can be treated and the need for interruption of anti-TNF therapy is rare,” the investigators concluded.
Find the full study in Annals of Internal Medicine (doi: 10.7326/M15-0729).
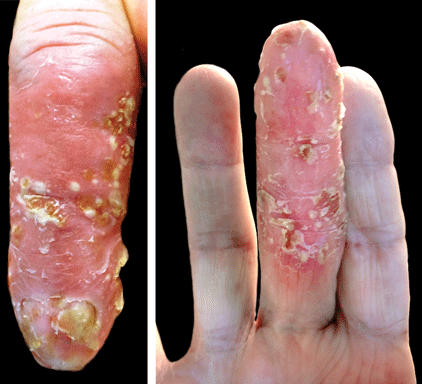
Scaly Plaque With Pustules and Anonychia on the Middle Finger
The Diagnosis: Acrodermatitis Continua of Hallopeau
Acrodermatitis continua of Hallopeau (ACH) is considered to be a form of acropustular psoriasis that presents as a sterile, pustular eruption initially affecting the fingertips and/or toes.1 The slow-growing pustules typically progress locally and can lead to onychodystrophy and/or osteolysis of the underlying bone.2,3 Most commonly affecting adult women, ACH often begins following local trauma to or infection of a single digit.4 As the disease progresses proximally, the small pustules burst, leaving a shiny, erythematous surface on which new pustules can develop. These pustules have a tendency to amalgamate, leading to the characteristic clinical finding of lakes of pus. Pustules frequently appear on the nail matrix and nail bed presenting as severe onychodystrophy and ultimately anonychia.5,6 Rarely, ACH can be associated with generalized pustular psoriasis as well as conjunctivitis, balanitis, and fissuring or annulus migrans of the tongue.2,7
Diagnosis can be established based on clinical findings, biopsy, and bacterial and fungal cultures revealing sterile pustules.8,9 Histologic findings are similar to those seen in pustular psoriasis, demonstrating subcorneal neutrophilic pustules, Munro microabscesses, and dilated blood vessels with lymphocytic infiltrate in the papillary dermis.10
Due to the refractory nature of the disease, there are no recommended guidelines for treatment of ACH. Most successful treatment regimens consist of topical psoriasis medications combined with systemic psoriatic therapies such as cyclosporine, methotrexate, acitretin, or biologic therapy.8,11-16 Our patient achieved satisfactory clinical improvement with clobetasol propionate ointment 0.05% twice daily alternating with calcipotriene cream 0.005% twice daily.
- Suchanek J. Relation of Hallopeau’s acrodermatitis continua to psoriasis. Przegl Dermatol. 1951;1:165-181.
- Adam BA, Loh CL. Acropustulosis (acrodermatitis continua) with resorption of terminal phalanges. Med J Malaysia. 1972;27:30-32.
- Mrowietz U. Pustular eruptions of palms and soles. In: Wolff K, Goldsmith LS, Katz SI, et al, eds. Fitzpatrick’s Dermatology in General Medicine. 7th ed. New York, NY: McGraw-Hill; 2007:215-218.
- Yerushalmi J, Grunwald MH, Hallel-Halevy D, et al. Chronic pustular eruption of the thumbs. diagnosis: acrodermatitis continue of Hallopeau (ACH). Arch Dermatol. 2000:136:925-930.
- Granelli U. Impetigo herpetiformis; acrodermatitis continue of Hallopeau and pustular psoriasis; etiology and pathogenesis and differential diagnosis. Minerva Dermatol. 1956;31:120-126.
- Mobini N, Toussaint S, Kamino H. Noninfectious erythematous, papular, and squamous diseases. In: Elder DE, Elenitsas R, Johnson B, et al, eds. Lever’s Histopathology of the Skin. 9th ed. Philadelphia, PA: Lippincott, Williams & Wilkins; 2005:174-210.
- Radcliff-Crocker H. Diseases of the Skin: Their Descriptions, Pathology, Diagnosis and Treatment. Philadelphia, PA: P. Blakiston, Son, & Co; 1888.
- Sehgal VN, Verma P, Sharma S, et al. Review: acrodermatitis continua of Hallopeau: evolution of treatment options. Int J Dermatol. 2011;50:1195-1211.
- Post CF, Hopper ME. Dermatitis repens: a report of two cases with bacteriologic studies. AMA Arc Derm Syphilol. 1951;63:220-223.
- Sehgal VN, Sharma S. The significance of Gram’s stain smear, potassium hydroxide mount, culture and microscopic pathology in the diagnosis of acrodermatitis continua of Hallopeau. Skinmed. 2011;9:260-261.
- Mosser G, Pillekamp H, Peter RU. Suppurative acrodermatitis continua of Hallopeau. a differential diagnosis of paronychia. Dtsch Med Wochenschr. 1998;123:386-390.
- Piquero-Casals J, Fonseca de Mello AP, Dal Coleto C, et al. Using oral tetracycline and topical betamethasone valerate to treat acrodermatitis continua of Hallopeau. Cutis. 2002;70:106-108.
- Tsuji T, Nishimura M. Topically administered fluorouracil in acrodermatitis continua of Hallopeau. Arch Dermatol. 1991;127:27-28.
- Van de Kerkhof PCM. In vivo effects of vitamin D3 analogs. J Dermatolog Treat. 1998;(suppl 3):S25-S29.
- Kokelj F, Plozzer C, Trevisan G. Uselessness of topical calcipotriol as monotherapy for acrodermatitis continua of Hallopeau. Acta Derm Venereol. 2001;81:153.
- Schneider LA, Hinrichs R, Scharffetter-Kochanek K. Phototherapy and photochemotherapy. Clin Dermatol. 2008;26:464-476.
The Diagnosis: Acrodermatitis Continua of Hallopeau
Acrodermatitis continua of Hallopeau (ACH) is considered to be a form of acropustular psoriasis that presents as a sterile, pustular eruption initially affecting the fingertips and/or toes.1 The slow-growing pustules typically progress locally and can lead to onychodystrophy and/or osteolysis of the underlying bone.2,3 Most commonly affecting adult women, ACH often begins following local trauma to or infection of a single digit.4 As the disease progresses proximally, the small pustules burst, leaving a shiny, erythematous surface on which new pustules can develop. These pustules have a tendency to amalgamate, leading to the characteristic clinical finding of lakes of pus. Pustules frequently appear on the nail matrix and nail bed presenting as severe onychodystrophy and ultimately anonychia.5,6 Rarely, ACH can be associated with generalized pustular psoriasis as well as conjunctivitis, balanitis, and fissuring or annulus migrans of the tongue.2,7
Diagnosis can be established based on clinical findings, biopsy, and bacterial and fungal cultures revealing sterile pustules.8,9 Histologic findings are similar to those seen in pustular psoriasis, demonstrating subcorneal neutrophilic pustules, Munro microabscesses, and dilated blood vessels with lymphocytic infiltrate in the papillary dermis.10
Due to the refractory nature of the disease, there are no recommended guidelines for treatment of ACH. Most successful treatment regimens consist of topical psoriasis medications combined with systemic psoriatic therapies such as cyclosporine, methotrexate, acitretin, or biologic therapy.8,11-16 Our patient achieved satisfactory clinical improvement with clobetasol propionate ointment 0.05% twice daily alternating with calcipotriene cream 0.005% twice daily.
The Diagnosis: Acrodermatitis Continua of Hallopeau
Acrodermatitis continua of Hallopeau (ACH) is considered to be a form of acropustular psoriasis that presents as a sterile, pustular eruption initially affecting the fingertips and/or toes.1 The slow-growing pustules typically progress locally and can lead to onychodystrophy and/or osteolysis of the underlying bone.2,3 Most commonly affecting adult women, ACH often begins following local trauma to or infection of a single digit.4 As the disease progresses proximally, the small pustules burst, leaving a shiny, erythematous surface on which new pustules can develop. These pustules have a tendency to amalgamate, leading to the characteristic clinical finding of lakes of pus. Pustules frequently appear on the nail matrix and nail bed presenting as severe onychodystrophy and ultimately anonychia.5,6 Rarely, ACH can be associated with generalized pustular psoriasis as well as conjunctivitis, balanitis, and fissuring or annulus migrans of the tongue.2,7
Diagnosis can be established based on clinical findings, biopsy, and bacterial and fungal cultures revealing sterile pustules.8,9 Histologic findings are similar to those seen in pustular psoriasis, demonstrating subcorneal neutrophilic pustules, Munro microabscesses, and dilated blood vessels with lymphocytic infiltrate in the papillary dermis.10
Due to the refractory nature of the disease, there are no recommended guidelines for treatment of ACH. Most successful treatment regimens consist of topical psoriasis medications combined with systemic psoriatic therapies such as cyclosporine, methotrexate, acitretin, or biologic therapy.8,11-16 Our patient achieved satisfactory clinical improvement with clobetasol propionate ointment 0.05% twice daily alternating with calcipotriene cream 0.005% twice daily.
- Suchanek J. Relation of Hallopeau’s acrodermatitis continua to psoriasis. Przegl Dermatol. 1951;1:165-181.
- Adam BA, Loh CL. Acropustulosis (acrodermatitis continua) with resorption of terminal phalanges. Med J Malaysia. 1972;27:30-32.
- Mrowietz U. Pustular eruptions of palms and soles. In: Wolff K, Goldsmith LS, Katz SI, et al, eds. Fitzpatrick’s Dermatology in General Medicine. 7th ed. New York, NY: McGraw-Hill; 2007:215-218.
- Yerushalmi J, Grunwald MH, Hallel-Halevy D, et al. Chronic pustular eruption of the thumbs. diagnosis: acrodermatitis continue of Hallopeau (ACH). Arch Dermatol. 2000:136:925-930.
- Granelli U. Impetigo herpetiformis; acrodermatitis continue of Hallopeau and pustular psoriasis; etiology and pathogenesis and differential diagnosis. Minerva Dermatol. 1956;31:120-126.
- Mobini N, Toussaint S, Kamino H. Noninfectious erythematous, papular, and squamous diseases. In: Elder DE, Elenitsas R, Johnson B, et al, eds. Lever’s Histopathology of the Skin. 9th ed. Philadelphia, PA: Lippincott, Williams & Wilkins; 2005:174-210.
- Radcliff-Crocker H. Diseases of the Skin: Their Descriptions, Pathology, Diagnosis and Treatment. Philadelphia, PA: P. Blakiston, Son, & Co; 1888.
- Sehgal VN, Verma P, Sharma S, et al. Review: acrodermatitis continua of Hallopeau: evolution of treatment options. Int J Dermatol. 2011;50:1195-1211.
- Post CF, Hopper ME. Dermatitis repens: a report of two cases with bacteriologic studies. AMA Arc Derm Syphilol. 1951;63:220-223.
- Sehgal VN, Sharma S. The significance of Gram’s stain smear, potassium hydroxide mount, culture and microscopic pathology in the diagnosis of acrodermatitis continua of Hallopeau. Skinmed. 2011;9:260-261.
- Mosser G, Pillekamp H, Peter RU. Suppurative acrodermatitis continua of Hallopeau. a differential diagnosis of paronychia. Dtsch Med Wochenschr. 1998;123:386-390.
- Piquero-Casals J, Fonseca de Mello AP, Dal Coleto C, et al. Using oral tetracycline and topical betamethasone valerate to treat acrodermatitis continua of Hallopeau. Cutis. 2002;70:106-108.
- Tsuji T, Nishimura M. Topically administered fluorouracil in acrodermatitis continua of Hallopeau. Arch Dermatol. 1991;127:27-28.
- Van de Kerkhof PCM. In vivo effects of vitamin D3 analogs. J Dermatolog Treat. 1998;(suppl 3):S25-S29.
- Kokelj F, Plozzer C, Trevisan G. Uselessness of topical calcipotriol as monotherapy for acrodermatitis continua of Hallopeau. Acta Derm Venereol. 2001;81:153.
- Schneider LA, Hinrichs R, Scharffetter-Kochanek K. Phototherapy and photochemotherapy. Clin Dermatol. 2008;26:464-476.
- Suchanek J. Relation of Hallopeau’s acrodermatitis continua to psoriasis. Przegl Dermatol. 1951;1:165-181.
- Adam BA, Loh CL. Acropustulosis (acrodermatitis continua) with resorption of terminal phalanges. Med J Malaysia. 1972;27:30-32.
- Mrowietz U. Pustular eruptions of palms and soles. In: Wolff K, Goldsmith LS, Katz SI, et al, eds. Fitzpatrick’s Dermatology in General Medicine. 7th ed. New York, NY: McGraw-Hill; 2007:215-218.
- Yerushalmi J, Grunwald MH, Hallel-Halevy D, et al. Chronic pustular eruption of the thumbs. diagnosis: acrodermatitis continue of Hallopeau (ACH). Arch Dermatol. 2000:136:925-930.
- Granelli U. Impetigo herpetiformis; acrodermatitis continue of Hallopeau and pustular psoriasis; etiology and pathogenesis and differential diagnosis. Minerva Dermatol. 1956;31:120-126.
- Mobini N, Toussaint S, Kamino H. Noninfectious erythematous, papular, and squamous diseases. In: Elder DE, Elenitsas R, Johnson B, et al, eds. Lever’s Histopathology of the Skin. 9th ed. Philadelphia, PA: Lippincott, Williams & Wilkins; 2005:174-210.
- Radcliff-Crocker H. Diseases of the Skin: Their Descriptions, Pathology, Diagnosis and Treatment. Philadelphia, PA: P. Blakiston, Son, & Co; 1888.
- Sehgal VN, Verma P, Sharma S, et al. Review: acrodermatitis continua of Hallopeau: evolution of treatment options. Int J Dermatol. 2011;50:1195-1211.
- Post CF, Hopper ME. Dermatitis repens: a report of two cases with bacteriologic studies. AMA Arc Derm Syphilol. 1951;63:220-223.
- Sehgal VN, Sharma S. The significance of Gram’s stain smear, potassium hydroxide mount, culture and microscopic pathology in the diagnosis of acrodermatitis continua of Hallopeau. Skinmed. 2011;9:260-261.
- Mosser G, Pillekamp H, Peter RU. Suppurative acrodermatitis continua of Hallopeau. a differential diagnosis of paronychia. Dtsch Med Wochenschr. 1998;123:386-390.
- Piquero-Casals J, Fonseca de Mello AP, Dal Coleto C, et al. Using oral tetracycline and topical betamethasone valerate to treat acrodermatitis continua of Hallopeau. Cutis. 2002;70:106-108.
- Tsuji T, Nishimura M. Topically administered fluorouracil in acrodermatitis continua of Hallopeau. Arch Dermatol. 1991;127:27-28.
- Van de Kerkhof PCM. In vivo effects of vitamin D3 analogs. J Dermatolog Treat. 1998;(suppl 3):S25-S29.
- Kokelj F, Plozzer C, Trevisan G. Uselessness of topical calcipotriol as monotherapy for acrodermatitis continua of Hallopeau. Acta Derm Venereol. 2001;81:153.
- Schneider LA, Hinrichs R, Scharffetter-Kochanek K. Phototherapy and photochemotherapy. Clin Dermatol. 2008;26:464-476.
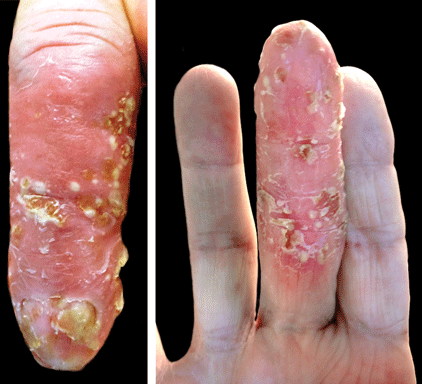
A 69-year-old man presented to our dermatology clinic with a persistent rash on the right middle finger of 5 years’ duration (left). Physical examination revealed a well-demarcated scaly plaque with pustules and anonychia localized to the right middle finger (right). Fungal and bacterial cultures revealed sterile pustules. The patient was successfully treated with an occluded superpotent topical steroid alternating with a topical vitamin D analogue.