User login
Reducing albumin improves kidney and heart function in people with type 2 diabetes
TOPLINE:
Reducing the urine albumin-to-creatinine ratio (UACR) significantly reduces kidney risk in people with type 2 diabetes, per new research in the Annals of Internal Medicine.
METHODOLOGY:
- Post hoc retrospective analysis of two phase 3 double-blind trials of finerenone in people with type 2 diabetes and chronic kidney disease
- Quantify the long-term health effects of reducing UACR within 4 months of taking finerenone by examining the records of 12,512 participants with an equal chance of receiving finerenone or placebo
- Isolate the impact of UACR reduction on kidney function and cardiovascular function by tracking health indicators related to the kidneys and the heart in participants for up to 4 years
TAKEAWAY:
- Over half of participants who received finerenone had reduced UACR by at least 30% from the baseline of 514 mg/g at the 4-month point after starting treatment, and the median UACR reduction in this group was 33%.
- By 4 months, a little over a quarter of participants who received the placebo had reduced their UACR levels by at least 30%, and the median UACR reduction in this group was 2.6%.
- A UACR reduction of at least 30% reduced kidney risk by 64%, as measured by reductions in kidney failure, sufficient glomerular filtration, and death from kidney disease.
- A UACR reduction of at least 30% reduced cardiovascular risk by 26%, as measured by fewer incidences of cardiovascular death, nonfatal infarction or stroke, and hospitalization for heart failure.
IN PRACTICE:
“Achieving early UACR reduction can lead to tangible benefits for kidney and cardiovascular health,” the authors note.
SOURCE:
The study was published in the Annals of Internal Medicine; the lead author is Rajiv Agarwal, MD, MS.
LIMITATIONS:
The study pertains only to finerenone, so the findings cannot be extrapolated to other drugs with different mechanisms of action.
DISCLOSURES:
Bayer AG Pharmaceuticals, which manufactures finerenone, was the primary funder of the study. The US National Institutes of Health and Veterans Administration also provided funding. Some study authors are full-time employees of Bayer AG. Many authors report consulting relationships with various pharmaceutical companies.
A version of this article appeared on Medscape.com.
TOPLINE:
Reducing the urine albumin-to-creatinine ratio (UACR) significantly reduces kidney risk in people with type 2 diabetes, per new research in the Annals of Internal Medicine.
METHODOLOGY:
- Post hoc retrospective analysis of two phase 3 double-blind trials of finerenone in people with type 2 diabetes and chronic kidney disease
- Quantify the long-term health effects of reducing UACR within 4 months of taking finerenone by examining the records of 12,512 participants with an equal chance of receiving finerenone or placebo
- Isolate the impact of UACR reduction on kidney function and cardiovascular function by tracking health indicators related to the kidneys and the heart in participants for up to 4 years
TAKEAWAY:
- Over half of participants who received finerenone had reduced UACR by at least 30% from the baseline of 514 mg/g at the 4-month point after starting treatment, and the median UACR reduction in this group was 33%.
- By 4 months, a little over a quarter of participants who received the placebo had reduced their UACR levels by at least 30%, and the median UACR reduction in this group was 2.6%.
- A UACR reduction of at least 30% reduced kidney risk by 64%, as measured by reductions in kidney failure, sufficient glomerular filtration, and death from kidney disease.
- A UACR reduction of at least 30% reduced cardiovascular risk by 26%, as measured by fewer incidences of cardiovascular death, nonfatal infarction or stroke, and hospitalization for heart failure.
IN PRACTICE:
“Achieving early UACR reduction can lead to tangible benefits for kidney and cardiovascular health,” the authors note.
SOURCE:
The study was published in the Annals of Internal Medicine; the lead author is Rajiv Agarwal, MD, MS.
LIMITATIONS:
The study pertains only to finerenone, so the findings cannot be extrapolated to other drugs with different mechanisms of action.
DISCLOSURES:
Bayer AG Pharmaceuticals, which manufactures finerenone, was the primary funder of the study. The US National Institutes of Health and Veterans Administration also provided funding. Some study authors are full-time employees of Bayer AG. Many authors report consulting relationships with various pharmaceutical companies.
A version of this article appeared on Medscape.com.
TOPLINE:
Reducing the urine albumin-to-creatinine ratio (UACR) significantly reduces kidney risk in people with type 2 diabetes, per new research in the Annals of Internal Medicine.
METHODOLOGY:
- Post hoc retrospective analysis of two phase 3 double-blind trials of finerenone in people with type 2 diabetes and chronic kidney disease
- Quantify the long-term health effects of reducing UACR within 4 months of taking finerenone by examining the records of 12,512 participants with an equal chance of receiving finerenone or placebo
- Isolate the impact of UACR reduction on kidney function and cardiovascular function by tracking health indicators related to the kidneys and the heart in participants for up to 4 years
TAKEAWAY:
- Over half of participants who received finerenone had reduced UACR by at least 30% from the baseline of 514 mg/g at the 4-month point after starting treatment, and the median UACR reduction in this group was 33%.
- By 4 months, a little over a quarter of participants who received the placebo had reduced their UACR levels by at least 30%, and the median UACR reduction in this group was 2.6%.
- A UACR reduction of at least 30% reduced kidney risk by 64%, as measured by reductions in kidney failure, sufficient glomerular filtration, and death from kidney disease.
- A UACR reduction of at least 30% reduced cardiovascular risk by 26%, as measured by fewer incidences of cardiovascular death, nonfatal infarction or stroke, and hospitalization for heart failure.
IN PRACTICE:
“Achieving early UACR reduction can lead to tangible benefits for kidney and cardiovascular health,” the authors note.
SOURCE:
The study was published in the Annals of Internal Medicine; the lead author is Rajiv Agarwal, MD, MS.
LIMITATIONS:
The study pertains only to finerenone, so the findings cannot be extrapolated to other drugs with different mechanisms of action.
DISCLOSURES:
Bayer AG Pharmaceuticals, which manufactures finerenone, was the primary funder of the study. The US National Institutes of Health and Veterans Administration also provided funding. Some study authors are full-time employees of Bayer AG. Many authors report consulting relationships with various pharmaceutical companies.
A version of this article appeared on Medscape.com.
Chronic Kidney Disease and Military Service in US Adults, 1999-2018
Chronic kidney disease (CKD) affects nearly 37 million people (11%) in the US and is a leading cause of death and morbidity. Due to their older age and higher prevalence of comorbid conditions, the prevalence of CKD among veterans is approximately 34% higher than in the general population and the fourth most common chronic disease diagnosed among US veterans.1,2 US veterans and those with prior military service (MS) may be at a particularly high risk for CKD and associated health care outcomes including increased hospitalization and death. The observed excess burden of CKD is not mirrored in the general population, and it is unclear whether prior MS confers a unique risk profile for CKD.
Current estimates of CKD burden among veterans or those with prior MS are widely variable and have been limited by unique regions, specific exposure profiles, or to single health care systems. As such, there remains a paucity of data examining CKD burden more broadly. We performed a study in the adult population of the US to quantify associations with the extent of CKD, enumerate temporal trends of CKD among those with prior MS, describe risk within subgroups, and compare heterogeneity of risk factors for CKD by MS.
Methods
The National Health and Nutrition Examination Survey (NHANES) is a suite of nationally representative, cross-sectional surveys of the noninstitutionalized US population. It is conducted by the National Center for Health Statistics and uses a stratified, clustered probability design, with surveys carried out without interruption, collated, and made accessible to the public at 2-year intervals.3 The survey consists of a questionnaire, physical examination, and laboratory data.
The inclusion criteria for our study were age ≥ 20 years along with serum creatinine and urinary albumin-creatinine measurements. The following definitions were used for the study:
• CKD: Estimated glomerular filtration rate < 60 mL/min/1.73 m2 calibrated to isotope dilution mass spectrometry (IDMS).
• Traceable: Creatinine-based CKD Epidemiology Collaboration formula or urinary albumin-creatine ratio ≥ 30 mg/g.
• MS: Positive response to the questions “Did you ever serve in the Armed Forces of the United States?” (1999 to 2010) or “Have you ever served on active duty in the US Armed Forces, military Reserves, or National Guard?” (2011 to 2018).
• Diabetes: Self-reported history, medication for diabetes, or glycated hemoglobin ≥ 7%.
• Hypertension: Blood pressure ≥ 140/90 or ≥ 130/40 mm Hg in the presence of diabetes, medication for hypertension, cardiovascular disease, or CKD, myocardial infarction, cardiac failure, or cerebrovascular disease by self-report.2,3
Analysis
Primary sampling unit, stratum, and weight variables were employed throughout to generate parameter estimates that are generalizable to the US population.4,5 The χ2 test and logistic regression, respectively, were employed for comparison of proportions and estimation of odds ratios. R Version 4.1.2 was employed for data analysis.
Results
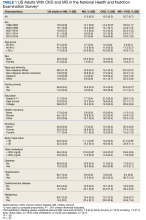
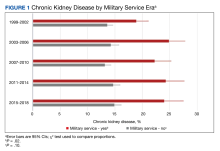
In the overall sample, the frequencies (95% standard error [SE]) of CKD and prior MS were 15.2% (0.3) and 11.5% (0.3) (Table 1). The proportion (SE) with CKD was significantly higher among those with prior MS vs the overall population: 22.7% (0.7) vs 15.2% (0.3) (P < .001). Significant associations with CKD were observed (P < .05) by age, sex, race and ethnicity, family poverty, school education, health insurance, smoking, body mass index, diabetes, hypertension, cardiovascular disease, and malignancy. Within those reporting prior MS, the proportion (SE) with CKD differed by era: 1999 to 2002, 18.9% (1.1); 2003 to 2006, 24.9% (1.5); 2007 to 2010, 22.3% (1.5); 2011 to 2014, 24.3% (1.7); and 2015 to 2018, 24.0% (1.8) (P = .02) (Figure 1).
Without covariate adjustment, prior MS was significantly associated with an increased risk of CKD (unadjusted odds ratio [OR], 1.78; 95% CI, 1.64-1.93; P < .05) (Table 2). Prior MS was significantly associated with CKD in the following subgroups: 2003 to 2006, 2011 to 2014, 2015 to 2018, age groups of 40 to 64 years and ≥ 65 years, male sex, non-Hispanic White and Hispanic ethnicity, school education of grade 0 to 11, and private or other health insurance. Additional comorbidities strongly associated with CKD included hypertension (OR, 6.37; 95% CI, 5.37-7.55), diabetes (OR, 4.16; 95% CI, 3.45-5.03), and cardiovascular disease (OR, 4.20; 95% CI, 3.57-4.95).
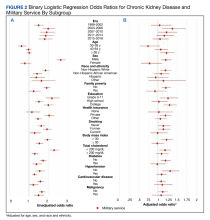
In the population reporting prior MS, the unadjusted OR of CKD vs 1999 to 2002 was greater for all other examined eras; with the greatest likelihood observed for the 2003 to 2006 era. Unadjusted ORs of CKD differed in groups with and without prior MS (P value for interaction < .05) for 2003 to 2006, those aged 40 to 64 years and ≥ 65 years, female sex, non-Hispanic African American and Hispanic race and ethnicity, family poverty, high school education, private health insurance, any smoking history, diabetes, hypertension, and cardiovascular disease (Figure 2A).
Following adjustment for age, sex, and race and ethnicity, MS was associated with a 17% higher likelihood of CKD (adjusted odds ratio [AOR], 1.17; 95% CI, 1.06-1.28; P < .01) (Table 3). Prior MS was significantly associated (P < .05) with CKD in the subgroups: age groups 40 to 64 years and ≥ 65 years, non-Hispanic African American, and body mass index ≥ 30. Among those with prior MS, comorbidities strongly associated with CKD in adjusted models included hypertension (AOR, 3.86; 95% CI, 3.18-4.69), diabetes (AOR, 3.05; 95% CI, 2.44-3.82), and cardiovascular disease (AOR, 2.51; 95% CI, 2.09-3.01). In the population with prior MS, the adjusted likelihood of CKD vs 1999 to 2002 was similar across all eras. Adjusted associations of CKD differed in groups with and without prior MS for age groups 40 to 64 years and ≥ 65 years, female sex, and family poverty (P < .05) (Figure 2B).
Discussion
We observed that prior MS was associated with CKD, all eras were associated with CKD in the subgroup with MS, and risk factors for CKD differed among many subgroups both with and without MS history, a finding that remained present in adjusted models. In addition, the finding of CKD was relatively common among those with prior MS (approximately 15%) and was most strongly associated with increasing age and comorbidities frequently associated with CKD.
Although many studies have demonstrated associations of US veteran status with various comorbidities, including hypertension, obesity, and diabetes, these studies often are limited to those both qualifying and receiving care within the US Department of Veterans Affairs (VA) health care system.6-9 The crude proportion of individuals reporting multiple chronic conditions, which included hypertension, diabetes, and weak or failing kidneys, was 49.7% for US veterans compared with 24.1% for nonveterans.2 Large-scale, nationally representative cohorts for use in this context have been limited by the heterogeneity of definitions of CKD applied with limited timeframes yielding variable estimates.1,10 Moreover, few studies have examined the clinical epidemiology of CKD more broadly in the US among those with prior MS. For example, a PubMed search on March 3, 2022, with the terms “epidemiology”, “military service”, and “chronic kidney disease” produced only 9 citations, one of which examined trends among a non-US cohort and quantifying disease burden another among adolescents.
Whether or not prior MS confers a unique risk profile for CKD is unknown. While our findings of an increased CKD burden among those reporting MS may partially reflect observed increases in baseline comorbidities, the observed excess CKD among those with MS remained across multiple categories even after adjustment for baseline demography. As several studies have demonstrated, enlistment into MS may select for a more diverse population; however those enlisted personnel may be of lower socioeconomic status and possibly at higher risk of CKD.11,12 Our findings of important differences in baseline determinants of health mirror this. The proportion of MS respondents with CKD vs CKD alone reporting a high school education or lower was higher (36.0% vs 21.8%) as well as among those with a history of family poverty (21.1% vs 18.0%).
Limitations
Our study has several limitations, including its cross-sectional study design, a lack of longitudinal data within individuals, and exclusion of institutionalized individuals. Limitations notwithstanding this study has several important aspects. As prior MS is highly variable, we were limited in our inability to stratify by service type or length of service. For example, veteran status is conferred to a “Reservist or member of the National Guard called to federal active duty or disabled from a disease or injury incurred or aggravated in line of duty or while in training status also qualify as a veteran” (13 CFR § 125.11). For the purposes of our study, prior MS would include all active-duty service (veterans) as well as reservists and National Guard members who have not been activated. This may be more representative of the overall effect of MS, as limitation to those receiving care within the VA may select for an older, more multimorbid population of patients, limiting generalizability.
In addition, more detailed information regarding service-related exposures and other service-connected conditions would allow for a more granular risk assessment by service type, era, and military conflict. Our finding of excess CKD burden among those with prior MS compared with the overall population is timely given the recent passage of the Promise to Address Comprehensive Toxics (PACT) Act. Exposure to and injury from Agent Orange—a known service-connected exposure associated with incident hypertension and diabetes—may be a significant contributor to CKD that may have a significant era effect. In addition, water contamination among those stationed in Camp Lejeune in North Carolina has notable genitourinary associations. Finally, burn pit exposures in more recent military conflicts may also have important associations with chronic disease, possibly including CKD. While similar attempts at the creation of large-scale US veteran cohorts have been limited by incomplete capture of creatinine, the large proportion of missing race data, and limited inclusion of additional markers of kidney disease, our use of a well-described, nationally representative survey along with standardized capture of clinical and laboratory elements mitigate the use of various societal or other codified definitions.1
Conclusions
Prior MS is associated with an increased risk of CKD overall and across several important subgroups. This finding was observed in various unadjusted and adjusted models and may constitute a unique risk profile of risk.
1. Ozieh MN, Gebregziabher M, Ward RC, Taber DJ, Egede LE. Creating a 13-year National Longitudinal Cohort of veterans with chronic kidney disease. BMC Nephrol. 2019;20(1):241. doi:10.1186/s12882-019-1430-y
2. Boersma P, Cohen RA, Zelaya CE, Moy E. Multiple chronic conditions among veterans and nonveterans: United States, 2015-2018. Natl Health Stat Report. 2021;(153):1-13.
3. Centers for Disease Control and Prevention, National Center for Health Statistics. National Health and Nutrition Survey. 2022. Accessed October 31, 2023. www.cdc.gov/nchs/nhanes/index.htm
4. Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604-612. doi:10.7326/0003-4819-150-9-200905050-00006
5. Selvin E, Manzi J, Stevens LA, et al. Calibration of serum creatinine in the National Health and Nutrition Examination Surveys (NHANES) 1988-1994, 1999-2004. Am J Kidney Dis. 2007;50(6):918-926. doi:10.1053/j.ajkd.2007.08.020
6. Smoley BA, Smith NL, Runkle GP. Hypertension in a population of active duty service members. J Am Board Fam Med. 2008;21(6):504-511. doi:10.3122/jabfm.2008.06.070182
7. Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129-139. doi:10.1056/NEJMoa0808431
8. Smith TJ, Marriott BP, Dotson L, et al. Overweight and obesity in military personnel: sociodemographic predictors. Obesity (Silver Spring). 2012;20(7):1534-1538. doi:10.1038/oby.2012.25
9. Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252-3257. doi:10.1001/archinte.160.21.3252
10. Saran R, Pearson A, Tilea A, et al. Burden and cost of caring for US veterans with CKD: initial findings from the VA Renal Information System (VA-REINS). Am J Kidney Dis. 2021;77(3):397-405. doi:10.1053/j.ajkd.2020.07.013
11. Wang L, Elder GH, Jr., Spence NJ. Status configurations, military service and higher education. Soc Forces. 2012;91(2):397-422. doi:10.1093/sf/sos174
12. Zeng X, Liu J, Tao S, Hong HG, Li Y, Fu P. Associations between socioeconomic status and chronic kidney disease: a meta-analysis. J Epidemiol Community Health. 2018;72(4):270-279. doi:10.1136/jech-2017-209815
Chronic kidney disease (CKD) affects nearly 37 million people (11%) in the US and is a leading cause of death and morbidity. Due to their older age and higher prevalence of comorbid conditions, the prevalence of CKD among veterans is approximately 34% higher than in the general population and the fourth most common chronic disease diagnosed among US veterans.1,2 US veterans and those with prior military service (MS) may be at a particularly high risk for CKD and associated health care outcomes including increased hospitalization and death. The observed excess burden of CKD is not mirrored in the general population, and it is unclear whether prior MS confers a unique risk profile for CKD.
Current estimates of CKD burden among veterans or those with prior MS are widely variable and have been limited by unique regions, specific exposure profiles, or to single health care systems. As such, there remains a paucity of data examining CKD burden more broadly. We performed a study in the adult population of the US to quantify associations with the extent of CKD, enumerate temporal trends of CKD among those with prior MS, describe risk within subgroups, and compare heterogeneity of risk factors for CKD by MS.
Methods
The National Health and Nutrition Examination Survey (NHANES) is a suite of nationally representative, cross-sectional surveys of the noninstitutionalized US population. It is conducted by the National Center for Health Statistics and uses a stratified, clustered probability design, with surveys carried out without interruption, collated, and made accessible to the public at 2-year intervals.3 The survey consists of a questionnaire, physical examination, and laboratory data.
The inclusion criteria for our study were age ≥ 20 years along with serum creatinine and urinary albumin-creatinine measurements. The following definitions were used for the study:
• CKD: Estimated glomerular filtration rate < 60 mL/min/1.73 m2 calibrated to isotope dilution mass spectrometry (IDMS).
• Traceable: Creatinine-based CKD Epidemiology Collaboration formula or urinary albumin-creatine ratio ≥ 30 mg/g.
• MS: Positive response to the questions “Did you ever serve in the Armed Forces of the United States?” (1999 to 2010) or “Have you ever served on active duty in the US Armed Forces, military Reserves, or National Guard?” (2011 to 2018).
• Diabetes: Self-reported history, medication for diabetes, or glycated hemoglobin ≥ 7%.
• Hypertension: Blood pressure ≥ 140/90 or ≥ 130/40 mm Hg in the presence of diabetes, medication for hypertension, cardiovascular disease, or CKD, myocardial infarction, cardiac failure, or cerebrovascular disease by self-report.2,3
Analysis
Primary sampling unit, stratum, and weight variables were employed throughout to generate parameter estimates that are generalizable to the US population.4,5 The χ2 test and logistic regression, respectively, were employed for comparison of proportions and estimation of odds ratios. R Version 4.1.2 was employed for data analysis.
Results
In the overall sample, the frequencies (95% standard error [SE]) of CKD and prior MS were 15.2% (0.3) and 11.5% (0.3) (Table 1). The proportion (SE) with CKD was significantly higher among those with prior MS vs the overall population: 22.7% (0.7) vs 15.2% (0.3) (P < .001). Significant associations with CKD were observed (P < .05) by age, sex, race and ethnicity, family poverty, school education, health insurance, smoking, body mass index, diabetes, hypertension, cardiovascular disease, and malignancy. Within those reporting prior MS, the proportion (SE) with CKD differed by era: 1999 to 2002, 18.9% (1.1); 2003 to 2006, 24.9% (1.5); 2007 to 2010, 22.3% (1.5); 2011 to 2014, 24.3% (1.7); and 2015 to 2018, 24.0% (1.8) (P = .02) (Figure 1).
Without covariate adjustment, prior MS was significantly associated with an increased risk of CKD (unadjusted odds ratio [OR], 1.78; 95% CI, 1.64-1.93; P < .05) (Table 2). Prior MS was significantly associated with CKD in the following subgroups: 2003 to 2006, 2011 to 2014, 2015 to 2018, age groups of 40 to 64 years and ≥ 65 years, male sex, non-Hispanic White and Hispanic ethnicity, school education of grade 0 to 11, and private or other health insurance. Additional comorbidities strongly associated with CKD included hypertension (OR, 6.37; 95% CI, 5.37-7.55), diabetes (OR, 4.16; 95% CI, 3.45-5.03), and cardiovascular disease (OR, 4.20; 95% CI, 3.57-4.95).
In the population reporting prior MS, the unadjusted OR of CKD vs 1999 to 2002 was greater for all other examined eras; with the greatest likelihood observed for the 2003 to 2006 era. Unadjusted ORs of CKD differed in groups with and without prior MS (P value for interaction < .05) for 2003 to 2006, those aged 40 to 64 years and ≥ 65 years, female sex, non-Hispanic African American and Hispanic race and ethnicity, family poverty, high school education, private health insurance, any smoking history, diabetes, hypertension, and cardiovascular disease (Figure 2A).
Following adjustment for age, sex, and race and ethnicity, MS was associated with a 17% higher likelihood of CKD (adjusted odds ratio [AOR], 1.17; 95% CI, 1.06-1.28; P < .01) (Table 3). Prior MS was significantly associated (P < .05) with CKD in the subgroups: age groups 40 to 64 years and ≥ 65 years, non-Hispanic African American, and body mass index ≥ 30. Among those with prior MS, comorbidities strongly associated with CKD in adjusted models included hypertension (AOR, 3.86; 95% CI, 3.18-4.69), diabetes (AOR, 3.05; 95% CI, 2.44-3.82), and cardiovascular disease (AOR, 2.51; 95% CI, 2.09-3.01). In the population with prior MS, the adjusted likelihood of CKD vs 1999 to 2002 was similar across all eras. Adjusted associations of CKD differed in groups with and without prior MS for age groups 40 to 64 years and ≥ 65 years, female sex, and family poverty (P < .05) (Figure 2B).
Discussion
We observed that prior MS was associated with CKD, all eras were associated with CKD in the subgroup with MS, and risk factors for CKD differed among many subgroups both with and without MS history, a finding that remained present in adjusted models. In addition, the finding of CKD was relatively common among those with prior MS (approximately 15%) and was most strongly associated with increasing age and comorbidities frequently associated with CKD.
Although many studies have demonstrated associations of US veteran status with various comorbidities, including hypertension, obesity, and diabetes, these studies often are limited to those both qualifying and receiving care within the US Department of Veterans Affairs (VA) health care system.6-9 The crude proportion of individuals reporting multiple chronic conditions, which included hypertension, diabetes, and weak or failing kidneys, was 49.7% for US veterans compared with 24.1% for nonveterans.2 Large-scale, nationally representative cohorts for use in this context have been limited by the heterogeneity of definitions of CKD applied with limited timeframes yielding variable estimates.1,10 Moreover, few studies have examined the clinical epidemiology of CKD more broadly in the US among those with prior MS. For example, a PubMed search on March 3, 2022, with the terms “epidemiology”, “military service”, and “chronic kidney disease” produced only 9 citations, one of which examined trends among a non-US cohort and quantifying disease burden another among adolescents.
Whether or not prior MS confers a unique risk profile for CKD is unknown. While our findings of an increased CKD burden among those reporting MS may partially reflect observed increases in baseline comorbidities, the observed excess CKD among those with MS remained across multiple categories even after adjustment for baseline demography. As several studies have demonstrated, enlistment into MS may select for a more diverse population; however those enlisted personnel may be of lower socioeconomic status and possibly at higher risk of CKD.11,12 Our findings of important differences in baseline determinants of health mirror this. The proportion of MS respondents with CKD vs CKD alone reporting a high school education or lower was higher (36.0% vs 21.8%) as well as among those with a history of family poverty (21.1% vs 18.0%).
Limitations
Our study has several limitations, including its cross-sectional study design, a lack of longitudinal data within individuals, and exclusion of institutionalized individuals. Limitations notwithstanding this study has several important aspects. As prior MS is highly variable, we were limited in our inability to stratify by service type or length of service. For example, veteran status is conferred to a “Reservist or member of the National Guard called to federal active duty or disabled from a disease or injury incurred or aggravated in line of duty or while in training status also qualify as a veteran” (13 CFR § 125.11). For the purposes of our study, prior MS would include all active-duty service (veterans) as well as reservists and National Guard members who have not been activated. This may be more representative of the overall effect of MS, as limitation to those receiving care within the VA may select for an older, more multimorbid population of patients, limiting generalizability.
In addition, more detailed information regarding service-related exposures and other service-connected conditions would allow for a more granular risk assessment by service type, era, and military conflict. Our finding of excess CKD burden among those with prior MS compared with the overall population is timely given the recent passage of the Promise to Address Comprehensive Toxics (PACT) Act. Exposure to and injury from Agent Orange—a known service-connected exposure associated with incident hypertension and diabetes—may be a significant contributor to CKD that may have a significant era effect. In addition, water contamination among those stationed in Camp Lejeune in North Carolina has notable genitourinary associations. Finally, burn pit exposures in more recent military conflicts may also have important associations with chronic disease, possibly including CKD. While similar attempts at the creation of large-scale US veteran cohorts have been limited by incomplete capture of creatinine, the large proportion of missing race data, and limited inclusion of additional markers of kidney disease, our use of a well-described, nationally representative survey along with standardized capture of clinical and laboratory elements mitigate the use of various societal or other codified definitions.1
Conclusions
Prior MS is associated with an increased risk of CKD overall and across several important subgroups. This finding was observed in various unadjusted and adjusted models and may constitute a unique risk profile of risk.
Chronic kidney disease (CKD) affects nearly 37 million people (11%) in the US and is a leading cause of death and morbidity. Due to their older age and higher prevalence of comorbid conditions, the prevalence of CKD among veterans is approximately 34% higher than in the general population and the fourth most common chronic disease diagnosed among US veterans.1,2 US veterans and those with prior military service (MS) may be at a particularly high risk for CKD and associated health care outcomes including increased hospitalization and death. The observed excess burden of CKD is not mirrored in the general population, and it is unclear whether prior MS confers a unique risk profile for CKD.
Current estimates of CKD burden among veterans or those with prior MS are widely variable and have been limited by unique regions, specific exposure profiles, or to single health care systems. As such, there remains a paucity of data examining CKD burden more broadly. We performed a study in the adult population of the US to quantify associations with the extent of CKD, enumerate temporal trends of CKD among those with prior MS, describe risk within subgroups, and compare heterogeneity of risk factors for CKD by MS.
Methods
The National Health and Nutrition Examination Survey (NHANES) is a suite of nationally representative, cross-sectional surveys of the noninstitutionalized US population. It is conducted by the National Center for Health Statistics and uses a stratified, clustered probability design, with surveys carried out without interruption, collated, and made accessible to the public at 2-year intervals.3 The survey consists of a questionnaire, physical examination, and laboratory data.
The inclusion criteria for our study were age ≥ 20 years along with serum creatinine and urinary albumin-creatinine measurements. The following definitions were used for the study:
• CKD: Estimated glomerular filtration rate < 60 mL/min/1.73 m2 calibrated to isotope dilution mass spectrometry (IDMS).
• Traceable: Creatinine-based CKD Epidemiology Collaboration formula or urinary albumin-creatine ratio ≥ 30 mg/g.
• MS: Positive response to the questions “Did you ever serve in the Armed Forces of the United States?” (1999 to 2010) or “Have you ever served on active duty in the US Armed Forces, military Reserves, or National Guard?” (2011 to 2018).
• Diabetes: Self-reported history, medication for diabetes, or glycated hemoglobin ≥ 7%.
• Hypertension: Blood pressure ≥ 140/90 or ≥ 130/40 mm Hg in the presence of diabetes, medication for hypertension, cardiovascular disease, or CKD, myocardial infarction, cardiac failure, or cerebrovascular disease by self-report.2,3
Analysis
Primary sampling unit, stratum, and weight variables were employed throughout to generate parameter estimates that are generalizable to the US population.4,5 The χ2 test and logistic regression, respectively, were employed for comparison of proportions and estimation of odds ratios. R Version 4.1.2 was employed for data analysis.
Results
In the overall sample, the frequencies (95% standard error [SE]) of CKD and prior MS were 15.2% (0.3) and 11.5% (0.3) (Table 1). The proportion (SE) with CKD was significantly higher among those with prior MS vs the overall population: 22.7% (0.7) vs 15.2% (0.3) (P < .001). Significant associations with CKD were observed (P < .05) by age, sex, race and ethnicity, family poverty, school education, health insurance, smoking, body mass index, diabetes, hypertension, cardiovascular disease, and malignancy. Within those reporting prior MS, the proportion (SE) with CKD differed by era: 1999 to 2002, 18.9% (1.1); 2003 to 2006, 24.9% (1.5); 2007 to 2010, 22.3% (1.5); 2011 to 2014, 24.3% (1.7); and 2015 to 2018, 24.0% (1.8) (P = .02) (Figure 1).
Without covariate adjustment, prior MS was significantly associated with an increased risk of CKD (unadjusted odds ratio [OR], 1.78; 95% CI, 1.64-1.93; P < .05) (Table 2). Prior MS was significantly associated with CKD in the following subgroups: 2003 to 2006, 2011 to 2014, 2015 to 2018, age groups of 40 to 64 years and ≥ 65 years, male sex, non-Hispanic White and Hispanic ethnicity, school education of grade 0 to 11, and private or other health insurance. Additional comorbidities strongly associated with CKD included hypertension (OR, 6.37; 95% CI, 5.37-7.55), diabetes (OR, 4.16; 95% CI, 3.45-5.03), and cardiovascular disease (OR, 4.20; 95% CI, 3.57-4.95).
In the population reporting prior MS, the unadjusted OR of CKD vs 1999 to 2002 was greater for all other examined eras; with the greatest likelihood observed for the 2003 to 2006 era. Unadjusted ORs of CKD differed in groups with and without prior MS (P value for interaction < .05) for 2003 to 2006, those aged 40 to 64 years and ≥ 65 years, female sex, non-Hispanic African American and Hispanic race and ethnicity, family poverty, high school education, private health insurance, any smoking history, diabetes, hypertension, and cardiovascular disease (Figure 2A).
Following adjustment for age, sex, and race and ethnicity, MS was associated with a 17% higher likelihood of CKD (adjusted odds ratio [AOR], 1.17; 95% CI, 1.06-1.28; P < .01) (Table 3). Prior MS was significantly associated (P < .05) with CKD in the subgroups: age groups 40 to 64 years and ≥ 65 years, non-Hispanic African American, and body mass index ≥ 30. Among those with prior MS, comorbidities strongly associated with CKD in adjusted models included hypertension (AOR, 3.86; 95% CI, 3.18-4.69), diabetes (AOR, 3.05; 95% CI, 2.44-3.82), and cardiovascular disease (AOR, 2.51; 95% CI, 2.09-3.01). In the population with prior MS, the adjusted likelihood of CKD vs 1999 to 2002 was similar across all eras. Adjusted associations of CKD differed in groups with and without prior MS for age groups 40 to 64 years and ≥ 65 years, female sex, and family poverty (P < .05) (Figure 2B).
Discussion
We observed that prior MS was associated with CKD, all eras were associated with CKD in the subgroup with MS, and risk factors for CKD differed among many subgroups both with and without MS history, a finding that remained present in adjusted models. In addition, the finding of CKD was relatively common among those with prior MS (approximately 15%) and was most strongly associated with increasing age and comorbidities frequently associated with CKD.
Although many studies have demonstrated associations of US veteran status with various comorbidities, including hypertension, obesity, and diabetes, these studies often are limited to those both qualifying and receiving care within the US Department of Veterans Affairs (VA) health care system.6-9 The crude proportion of individuals reporting multiple chronic conditions, which included hypertension, diabetes, and weak or failing kidneys, was 49.7% for US veterans compared with 24.1% for nonveterans.2 Large-scale, nationally representative cohorts for use in this context have been limited by the heterogeneity of definitions of CKD applied with limited timeframes yielding variable estimates.1,10 Moreover, few studies have examined the clinical epidemiology of CKD more broadly in the US among those with prior MS. For example, a PubMed search on March 3, 2022, with the terms “epidemiology”, “military service”, and “chronic kidney disease” produced only 9 citations, one of which examined trends among a non-US cohort and quantifying disease burden another among adolescents.
Whether or not prior MS confers a unique risk profile for CKD is unknown. While our findings of an increased CKD burden among those reporting MS may partially reflect observed increases in baseline comorbidities, the observed excess CKD among those with MS remained across multiple categories even after adjustment for baseline demography. As several studies have demonstrated, enlistment into MS may select for a more diverse population; however those enlisted personnel may be of lower socioeconomic status and possibly at higher risk of CKD.11,12 Our findings of important differences in baseline determinants of health mirror this. The proportion of MS respondents with CKD vs CKD alone reporting a high school education or lower was higher (36.0% vs 21.8%) as well as among those with a history of family poverty (21.1% vs 18.0%).
Limitations
Our study has several limitations, including its cross-sectional study design, a lack of longitudinal data within individuals, and exclusion of institutionalized individuals. Limitations notwithstanding this study has several important aspects. As prior MS is highly variable, we were limited in our inability to stratify by service type or length of service. For example, veteran status is conferred to a “Reservist or member of the National Guard called to federal active duty or disabled from a disease or injury incurred or aggravated in line of duty or while in training status also qualify as a veteran” (13 CFR § 125.11). For the purposes of our study, prior MS would include all active-duty service (veterans) as well as reservists and National Guard members who have not been activated. This may be more representative of the overall effect of MS, as limitation to those receiving care within the VA may select for an older, more multimorbid population of patients, limiting generalizability.
In addition, more detailed information regarding service-related exposures and other service-connected conditions would allow for a more granular risk assessment by service type, era, and military conflict. Our finding of excess CKD burden among those with prior MS compared with the overall population is timely given the recent passage of the Promise to Address Comprehensive Toxics (PACT) Act. Exposure to and injury from Agent Orange—a known service-connected exposure associated with incident hypertension and diabetes—may be a significant contributor to CKD that may have a significant era effect. In addition, water contamination among those stationed in Camp Lejeune in North Carolina has notable genitourinary associations. Finally, burn pit exposures in more recent military conflicts may also have important associations with chronic disease, possibly including CKD. While similar attempts at the creation of large-scale US veteran cohorts have been limited by incomplete capture of creatinine, the large proportion of missing race data, and limited inclusion of additional markers of kidney disease, our use of a well-described, nationally representative survey along with standardized capture of clinical and laboratory elements mitigate the use of various societal or other codified definitions.1
Conclusions
Prior MS is associated with an increased risk of CKD overall and across several important subgroups. This finding was observed in various unadjusted and adjusted models and may constitute a unique risk profile of risk.
1. Ozieh MN, Gebregziabher M, Ward RC, Taber DJ, Egede LE. Creating a 13-year National Longitudinal Cohort of veterans with chronic kidney disease. BMC Nephrol. 2019;20(1):241. doi:10.1186/s12882-019-1430-y
2. Boersma P, Cohen RA, Zelaya CE, Moy E. Multiple chronic conditions among veterans and nonveterans: United States, 2015-2018. Natl Health Stat Report. 2021;(153):1-13.
3. Centers for Disease Control and Prevention, National Center for Health Statistics. National Health and Nutrition Survey. 2022. Accessed October 31, 2023. www.cdc.gov/nchs/nhanes/index.htm
4. Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604-612. doi:10.7326/0003-4819-150-9-200905050-00006
5. Selvin E, Manzi J, Stevens LA, et al. Calibration of serum creatinine in the National Health and Nutrition Examination Surveys (NHANES) 1988-1994, 1999-2004. Am J Kidney Dis. 2007;50(6):918-926. doi:10.1053/j.ajkd.2007.08.020
6. Smoley BA, Smith NL, Runkle GP. Hypertension in a population of active duty service members. J Am Board Fam Med. 2008;21(6):504-511. doi:10.3122/jabfm.2008.06.070182
7. Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129-139. doi:10.1056/NEJMoa0808431
8. Smith TJ, Marriott BP, Dotson L, et al. Overweight and obesity in military personnel: sociodemographic predictors. Obesity (Silver Spring). 2012;20(7):1534-1538. doi:10.1038/oby.2012.25
9. Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252-3257. doi:10.1001/archinte.160.21.3252
10. Saran R, Pearson A, Tilea A, et al. Burden and cost of caring for US veterans with CKD: initial findings from the VA Renal Information System (VA-REINS). Am J Kidney Dis. 2021;77(3):397-405. doi:10.1053/j.ajkd.2020.07.013
11. Wang L, Elder GH, Jr., Spence NJ. Status configurations, military service and higher education. Soc Forces. 2012;91(2):397-422. doi:10.1093/sf/sos174
12. Zeng X, Liu J, Tao S, Hong HG, Li Y, Fu P. Associations between socioeconomic status and chronic kidney disease: a meta-analysis. J Epidemiol Community Health. 2018;72(4):270-279. doi:10.1136/jech-2017-209815
1. Ozieh MN, Gebregziabher M, Ward RC, Taber DJ, Egede LE. Creating a 13-year National Longitudinal Cohort of veterans with chronic kidney disease. BMC Nephrol. 2019;20(1):241. doi:10.1186/s12882-019-1430-y
2. Boersma P, Cohen RA, Zelaya CE, Moy E. Multiple chronic conditions among veterans and nonveterans: United States, 2015-2018. Natl Health Stat Report. 2021;(153):1-13.
3. Centers for Disease Control and Prevention, National Center for Health Statistics. National Health and Nutrition Survey. 2022. Accessed October 31, 2023. www.cdc.gov/nchs/nhanes/index.htm
4. Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604-612. doi:10.7326/0003-4819-150-9-200905050-00006
5. Selvin E, Manzi J, Stevens LA, et al. Calibration of serum creatinine in the National Health and Nutrition Examination Surveys (NHANES) 1988-1994, 1999-2004. Am J Kidney Dis. 2007;50(6):918-926. doi:10.1053/j.ajkd.2007.08.020
6. Smoley BA, Smith NL, Runkle GP. Hypertension in a population of active duty service members. J Am Board Fam Med. 2008;21(6):504-511. doi:10.3122/jabfm.2008.06.070182
7. Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med. 2009;360(2):129-139. doi:10.1056/NEJMoa0808431
8. Smith TJ, Marriott BP, Dotson L, et al. Overweight and obesity in military personnel: sociodemographic predictors. Obesity (Silver Spring). 2012;20(7):1534-1538. doi:10.1038/oby.2012.25
9. Agha Z, Lofgren RP, VanRuiswyk JV, Layde PM. Are patients at Veterans Affairs medical centers sicker? A comparative analysis of health status and medical resource use. Arch Intern Med. 2000;160(21):3252-3257. doi:10.1001/archinte.160.21.3252
10. Saran R, Pearson A, Tilea A, et al. Burden and cost of caring for US veterans with CKD: initial findings from the VA Renal Information System (VA-REINS). Am J Kidney Dis. 2021;77(3):397-405. doi:10.1053/j.ajkd.2020.07.013
11. Wang L, Elder GH, Jr., Spence NJ. Status configurations, military service and higher education. Soc Forces. 2012;91(2):397-422. doi:10.1093/sf/sos174
12. Zeng X, Liu J, Tao S, Hong HG, Li Y, Fu P. Associations between socioeconomic status and chronic kidney disease: a meta-analysis. J Epidemiol Community Health. 2018;72(4):270-279. doi:10.1136/jech-2017-209815
Low-dose methotrexate carries higher risk for older patients with CKD
TOPLINE:
The use of low-dose methotrexate among older adults with chronic kidney disease (CKD) was associated with a significantly increased risk at 90 days for serious adverse events requiring a hospital visit, compared with starting treatment with hydroxychloroquine.
METHODOLOGY:
- In a retrospective, population-based cohort study conducted in Ontario, researchers used linked administrative healthcare data to identify adults aged 66 years and older with CKD who were not undergoing dialysis and were new to medication; CKD was defined as an estimated glomerular filtration rate (eGFR) of less than 60 mL/min per 1.73 m2.
- The study population included 2,309 individuals who began treatment with low-dose methotrexate (5-35 mg/week); they were matched with 2,309 individuals who began treatment with hydroxychloroquine (200-400 mg/day). The median age was 76 years, 69% were women, and rheumatoid arthritis was the most common diagnosis (56%).
- The primary outcome was the risk of a hospital visit at 90 days for a composite of serious adverse events that included myelosuppression, sepsis, pneumotoxic effects, or hepatoxic effects.
TAKEAWAY:
- Overall, 3.55% of methotrexate patients and 1.73% of hydroxychloroquine patients met the primary outcome (risk ratio, 2.05); these events occurred at a median of 49 days and 43 days after starting the medications for the two groups, respectively.
- In an analysis by eGFR category, the risk of serious adverse events at 90 days increased among patients with eGFR levels less than 45 mL/min per 1.73 m2 (RR, 2.79).
- In a secondary comparison, the 90-day risk of serious adverse events was higher among methotrexate patients who began treatment with doses of 15-35 mg/week in comparison with those whose initial doses were 5 to less than 15 mg/week.
IN PRACTICE:
“Patients with CKD starting low-dose methotrexate should have active surveillance, including blood tests and chest radiographs performed regularly to monitor for signs of myelosuppression, infection, hepatotoxic effects, and pneumotoxic effects,” the researchers wrote.
SOURCE:
The lead author on the study was Flory T. Muanda, MD, of Western University, London, Ont. The study was published online in JAMA Network Open.
LIMITATIONS:
The observational design and lack of data on patients’ adherence to medications were among the limiting factors, as were the focus on older adults with CKD and the lack of assessment of the risk-benefit ratio of low-dose methotrexate.
DISCLOSURES:
The study was supported by the Institute for Clinical Evaluative Sciences. Dr. Muanda had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
The use of low-dose methotrexate among older adults with chronic kidney disease (CKD) was associated with a significantly increased risk at 90 days for serious adverse events requiring a hospital visit, compared with starting treatment with hydroxychloroquine.
METHODOLOGY:
- In a retrospective, population-based cohort study conducted in Ontario, researchers used linked administrative healthcare data to identify adults aged 66 years and older with CKD who were not undergoing dialysis and were new to medication; CKD was defined as an estimated glomerular filtration rate (eGFR) of less than 60 mL/min per 1.73 m2.
- The study population included 2,309 individuals who began treatment with low-dose methotrexate (5-35 mg/week); they were matched with 2,309 individuals who began treatment with hydroxychloroquine (200-400 mg/day). The median age was 76 years, 69% were women, and rheumatoid arthritis was the most common diagnosis (56%).
- The primary outcome was the risk of a hospital visit at 90 days for a composite of serious adverse events that included myelosuppression, sepsis, pneumotoxic effects, or hepatoxic effects.
TAKEAWAY:
- Overall, 3.55% of methotrexate patients and 1.73% of hydroxychloroquine patients met the primary outcome (risk ratio, 2.05); these events occurred at a median of 49 days and 43 days after starting the medications for the two groups, respectively.
- In an analysis by eGFR category, the risk of serious adverse events at 90 days increased among patients with eGFR levels less than 45 mL/min per 1.73 m2 (RR, 2.79).
- In a secondary comparison, the 90-day risk of serious adverse events was higher among methotrexate patients who began treatment with doses of 15-35 mg/week in comparison with those whose initial doses were 5 to less than 15 mg/week.
IN PRACTICE:
“Patients with CKD starting low-dose methotrexate should have active surveillance, including blood tests and chest radiographs performed regularly to monitor for signs of myelosuppression, infection, hepatotoxic effects, and pneumotoxic effects,” the researchers wrote.
SOURCE:
The lead author on the study was Flory T. Muanda, MD, of Western University, London, Ont. The study was published online in JAMA Network Open.
LIMITATIONS:
The observational design and lack of data on patients’ adherence to medications were among the limiting factors, as were the focus on older adults with CKD and the lack of assessment of the risk-benefit ratio of low-dose methotrexate.
DISCLOSURES:
The study was supported by the Institute for Clinical Evaluative Sciences. Dr. Muanda had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
The use of low-dose methotrexate among older adults with chronic kidney disease (CKD) was associated with a significantly increased risk at 90 days for serious adverse events requiring a hospital visit, compared with starting treatment with hydroxychloroquine.
METHODOLOGY:
- In a retrospective, population-based cohort study conducted in Ontario, researchers used linked administrative healthcare data to identify adults aged 66 years and older with CKD who were not undergoing dialysis and were new to medication; CKD was defined as an estimated glomerular filtration rate (eGFR) of less than 60 mL/min per 1.73 m2.
- The study population included 2,309 individuals who began treatment with low-dose methotrexate (5-35 mg/week); they were matched with 2,309 individuals who began treatment with hydroxychloroquine (200-400 mg/day). The median age was 76 years, 69% were women, and rheumatoid arthritis was the most common diagnosis (56%).
- The primary outcome was the risk of a hospital visit at 90 days for a composite of serious adverse events that included myelosuppression, sepsis, pneumotoxic effects, or hepatoxic effects.
TAKEAWAY:
- Overall, 3.55% of methotrexate patients and 1.73% of hydroxychloroquine patients met the primary outcome (risk ratio, 2.05); these events occurred at a median of 49 days and 43 days after starting the medications for the two groups, respectively.
- In an analysis by eGFR category, the risk of serious adverse events at 90 days increased among patients with eGFR levels less than 45 mL/min per 1.73 m2 (RR, 2.79).
- In a secondary comparison, the 90-day risk of serious adverse events was higher among methotrexate patients who began treatment with doses of 15-35 mg/week in comparison with those whose initial doses were 5 to less than 15 mg/week.
IN PRACTICE:
“Patients with CKD starting low-dose methotrexate should have active surveillance, including blood tests and chest radiographs performed regularly to monitor for signs of myelosuppression, infection, hepatotoxic effects, and pneumotoxic effects,” the researchers wrote.
SOURCE:
The lead author on the study was Flory T. Muanda, MD, of Western University, London, Ont. The study was published online in JAMA Network Open.
LIMITATIONS:
The observational design and lack of data on patients’ adherence to medications were among the limiting factors, as were the focus on older adults with CKD and the lack of assessment of the risk-benefit ratio of low-dose methotrexate.
DISCLOSURES:
The study was supported by the Institute for Clinical Evaluative Sciences. Dr. Muanda had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Albuminuria reduction fuels finerenone’s kidney benefits
PHILADELPHIA – Reducing albuminuria is a key mediator of the way finerenone (Kerendia, Bayer) reduces adverse renal and cardiovascular events in people with type 2 diabetes and chronic kidney disease (CKD), based on findings from two novel mediation analyses run on data from more than 12,000 people included in the two finerenone pivotal trials.
Results from these analyses showed that that finerenone treatment produced in the FIDELIO-DKD and FIGARO-DKD phase 3 trials. FIDELIO-DKD, which had protection against adverse kidney outcomes as its primary endpoint, supplied the data that led to finerenone’s approval in 2021 by the U.S. Food and Drug Administration for treating people with type 2 diabetes and CKD.
The findings of the mediation analyses underscore the important role that albuminuria plays in the nephropathy and related comorbidities associated with type 2 diabetes and CKD and highlight the importance of ongoing monitoring of albuminuria to guide treatments aimed at minimizing this pathology, said Rajiv Agarwal, MD, who presented a poster on the mediation analyses at Kidney Week 2023, organized by the American Society of Nephrology.
“My hope is that this [report] heightens awareness of UACR” as an important marker of both CKD and of the response by patients with CKD to their treatment, said Dr. Agarwal, a nephrologist and professor at Indiana University in Indianapolis.
“Only about half of people with type 2 diabetes get their UACR measured even though every guideline says measure UACR in people with diabetes. Our findings say that UACR is important not just for CKD diagnosis but also to give feedback” on whether management is working, Dr. Agarwal said in an interview.
Incorporate UACR into clinical decision-making
“My hope is that clinicians will look at UACR as something they should incorporate into clinical decision-making. I measure UACR in my patients [with CKD and type 2 diabetes] at every visit; it’s so inexpensive. Albuminuria is not a good sign. If it’s not reduced in a patient by at least 30% [the recommended minimum reduction by the American Diabetes Association for people who start with a UACR of at least 300 mg/g] clinicians should think of what else they could do to lower albuminuria”: Reduce salt intake, improve blood pressure control, make sure the patient is adherent to treatments, and add additional treatments, Dr. Agarwal advised.
Multiple efforts are now underway or will soon start to boost the rate at which at-risk people get their UACR measured, noted Leslie A. Inker, MD, in a separate talk during Kidney Week. These efforts include the National Kidney Foundation’s CKD Learning Collaborative, which aims to improve clinician awareness of CKD and improve routine testing for CKD. Early results during 2023 from this program in Missouri showed a nearly 8–percentage point increase in the screening rate for UACR levels in at-risk people, said Dr. Inker, professor and director of the Kidney and Blood Pressure Center at Tufts Medical Center in Boston.
A second advance was introduction in 2018 of the “kidney profile” lab order by the American College of Clinical Pathology that allows clinicians to order as a single test both an estimated glomerular filtration rate (eGFR) and a UACR.
Also, the Centers for Medicare & Medicaid Services and the National Committee for Quality Assurance have both taken steps to encourage UACR ordering. The NCQA established a new Healthcare Effectiveness Data and Information Set performance measure for U.S. physicians starting in 2023 that will track measurement of UACR and eGFR in people with diabetes. CMS also has made assessment of kidney health a measure of care quality in programs effective in 2023 and 2024, Dr. Inker noted.
Most subjects had elevated UACRs
The study run by Dr. Agarwal and his associates used data from 12,512 of the more than 13,000 people enrolled in either FIDELITY-DKD or FIGARO-DKD who had UACR measurements recorded at baseline, at 4 months into either study, or both. Their median UACR at the time they began on finerenone or placebo was 514 mg/g, with 67% having a UACR of at least 300 mg/g (macroalbuminuria) and 31% having a UACR of 30-299 mg/g (microalbuminuria). By design, virtually all patients in these two trials were on a renin-angiotensin system inhibitor (either an angiotensin-converting enzyme inhibitor or an angiotensin-receptor blocker), but given the time period when the two trials enrolled participants (during 2015-2018) only 7% of those enrolled were on a sodium-glucose cotransporter 2 inhibitor and only 7% were on a glucagonlike peptide–1 receptor agonist.
Four months after treatment began, 53% of those randomized to finerenone treatment and 27% of those in the placebo arm had their UACR reduced by at least 30% from baseline, the cutpoint chosen by Dr. Agarwal based on the American Diabetes Association guideline.
Kaplan-Meier analyses showed that the incidence of the primary kidney outcome – kidney failure, a sustained ≥ 57% decrease in eGFR from baseline, or kidney death – showed close correlation with at least a 30% reduction in UACR regardless of whether the patients in this subgroup received finerenone or placebo.
A different correlation was found in those with a less than 30% reduction in their UACR from baseline to 4 months, regardless of whether this happened on finerenone or placebo. People in the two finerenone trials who had a lesser reduction from baseline in their UACR also had a significantly higher rate of adverse kidney outcomes whether they received finerenone or placebo.
84% of finerenone’s kidney benefit linked to lowering of UACR
The causal-mediation analysis run by Dr. Agarwal quantified this observation, showing that 84% of finerenone’s effect on the kidney outcome was mediated by the reduction in UACR.
“It seems like the kidney benefit [from finerenone] travels through the level of albuminuria. This has broad implications for treatment of people with type 2 diabetes and CKD,” he said.
The link with reduction in albuminuria was weaker for the primary cardiovascular disease outcome: CV death, nonfatal myocardial infarction, nonfatal stroke, or hospitalization for heart failure. The strongest effect on this outcome was only seen in Kaplan-Meier analysis in those on finerenone who had at least a 30% reduction in their UACR. Those on placebo and with a similarly robust 4-month reduction in UACR showed a much more modest cardiovascular benefit that resembled those on either finerenone or placebo who had a smaller, less than 30% UACR reduction. The mediation analysis of these data showed that UACR reduction accounted for about 37% of the observed cardiovascular benefit seen during the trials.
“The effect of UACR is much stronger for the kidney outcomes,” summed up Dr. Agarwal. The results suggest that for cardiovascular outcomes finerenone works through factors other than lowering of UACR, but he admitted that no one currently knows what those other factors might be.
Treat aggressively to lower UACR by 30%
“I wouldn’t stop finerenone treatment in people who do not get a 30% reduction in their UACR” because these analyses suggest that a portion of the overall benefits from finerenone occurs via other mechanisms, he said. But in patients whose UACR is not reduced by at least 30% “be more aggressive on other measures to reduce UACR,” he advised.
The mediation analyses he ran are “the first time this has been done in nephrology,” producing a “groundbreaking” analysis and finding, Dr. Agarwal said. He also highlighted that the findings primarily relate to the importance of controlling UACR rather than an endorsement of finerenone as the best way to achieve this.
“All I care about is that people think about UACR as a modifiable risk factor. It doesn’t have to be treated with finerenone. It could be a renin-angiotensin system inhibitor, it could be chlorthalidone [a thiazide diuretic]. It just happened that we had a large dataset of people treated with finerenone or placebo.”
He said that future mediation analyses should look at the link between outcomes and UACR reductions produced by agents from the classes of sodium-glucose cotransporter 2 inhibitors and the glucagonlike peptide–1 receptor agonists.
FIDELIO-DKD and FIGARO-DKD were both sponsored by Bayer, the company that markets finerenone. Dr. Agarwal has received personal fees and nonfinancial support from Bayer. He has also received personal fees and nonfinancial support from Akebia Therapeutics, AstraZeneca, Boehringer Ingelheim, Eli Lilly, and Vifor Pharma, and he is a member of data safety monitoring committees for Chinook and Vertex. Dr. Inker is a consultant to Diamtrix, and her department receives research funding from Chinook, Omeros, Reata, and Tricida.
PHILADELPHIA – Reducing albuminuria is a key mediator of the way finerenone (Kerendia, Bayer) reduces adverse renal and cardiovascular events in people with type 2 diabetes and chronic kidney disease (CKD), based on findings from two novel mediation analyses run on data from more than 12,000 people included in the two finerenone pivotal trials.
Results from these analyses showed that that finerenone treatment produced in the FIDELIO-DKD and FIGARO-DKD phase 3 trials. FIDELIO-DKD, which had protection against adverse kidney outcomes as its primary endpoint, supplied the data that led to finerenone’s approval in 2021 by the U.S. Food and Drug Administration for treating people with type 2 diabetes and CKD.
The findings of the mediation analyses underscore the important role that albuminuria plays in the nephropathy and related comorbidities associated with type 2 diabetes and CKD and highlight the importance of ongoing monitoring of albuminuria to guide treatments aimed at minimizing this pathology, said Rajiv Agarwal, MD, who presented a poster on the mediation analyses at Kidney Week 2023, organized by the American Society of Nephrology.
“My hope is that this [report] heightens awareness of UACR” as an important marker of both CKD and of the response by patients with CKD to their treatment, said Dr. Agarwal, a nephrologist and professor at Indiana University in Indianapolis.
“Only about half of people with type 2 diabetes get their UACR measured even though every guideline says measure UACR in people with diabetes. Our findings say that UACR is important not just for CKD diagnosis but also to give feedback” on whether management is working, Dr. Agarwal said in an interview.
Incorporate UACR into clinical decision-making
“My hope is that clinicians will look at UACR as something they should incorporate into clinical decision-making. I measure UACR in my patients [with CKD and type 2 diabetes] at every visit; it’s so inexpensive. Albuminuria is not a good sign. If it’s not reduced in a patient by at least 30% [the recommended minimum reduction by the American Diabetes Association for people who start with a UACR of at least 300 mg/g] clinicians should think of what else they could do to lower albuminuria”: Reduce salt intake, improve blood pressure control, make sure the patient is adherent to treatments, and add additional treatments, Dr. Agarwal advised.
Multiple efforts are now underway or will soon start to boost the rate at which at-risk people get their UACR measured, noted Leslie A. Inker, MD, in a separate talk during Kidney Week. These efforts include the National Kidney Foundation’s CKD Learning Collaborative, which aims to improve clinician awareness of CKD and improve routine testing for CKD. Early results during 2023 from this program in Missouri showed a nearly 8–percentage point increase in the screening rate for UACR levels in at-risk people, said Dr. Inker, professor and director of the Kidney and Blood Pressure Center at Tufts Medical Center in Boston.
A second advance was introduction in 2018 of the “kidney profile” lab order by the American College of Clinical Pathology that allows clinicians to order as a single test both an estimated glomerular filtration rate (eGFR) and a UACR.
Also, the Centers for Medicare & Medicaid Services and the National Committee for Quality Assurance have both taken steps to encourage UACR ordering. The NCQA established a new Healthcare Effectiveness Data and Information Set performance measure for U.S. physicians starting in 2023 that will track measurement of UACR and eGFR in people with diabetes. CMS also has made assessment of kidney health a measure of care quality in programs effective in 2023 and 2024, Dr. Inker noted.
Most subjects had elevated UACRs
The study run by Dr. Agarwal and his associates used data from 12,512 of the more than 13,000 people enrolled in either FIDELITY-DKD or FIGARO-DKD who had UACR measurements recorded at baseline, at 4 months into either study, or both. Their median UACR at the time they began on finerenone or placebo was 514 mg/g, with 67% having a UACR of at least 300 mg/g (macroalbuminuria) and 31% having a UACR of 30-299 mg/g (microalbuminuria). By design, virtually all patients in these two trials were on a renin-angiotensin system inhibitor (either an angiotensin-converting enzyme inhibitor or an angiotensin-receptor blocker), but given the time period when the two trials enrolled participants (during 2015-2018) only 7% of those enrolled were on a sodium-glucose cotransporter 2 inhibitor and only 7% were on a glucagonlike peptide–1 receptor agonist.
Four months after treatment began, 53% of those randomized to finerenone treatment and 27% of those in the placebo arm had their UACR reduced by at least 30% from baseline, the cutpoint chosen by Dr. Agarwal based on the American Diabetes Association guideline.
Kaplan-Meier analyses showed that the incidence of the primary kidney outcome – kidney failure, a sustained ≥ 57% decrease in eGFR from baseline, or kidney death – showed close correlation with at least a 30% reduction in UACR regardless of whether the patients in this subgroup received finerenone or placebo.
A different correlation was found in those with a less than 30% reduction in their UACR from baseline to 4 months, regardless of whether this happened on finerenone or placebo. People in the two finerenone trials who had a lesser reduction from baseline in their UACR also had a significantly higher rate of adverse kidney outcomes whether they received finerenone or placebo.
84% of finerenone’s kidney benefit linked to lowering of UACR
The causal-mediation analysis run by Dr. Agarwal quantified this observation, showing that 84% of finerenone’s effect on the kidney outcome was mediated by the reduction in UACR.
“It seems like the kidney benefit [from finerenone] travels through the level of albuminuria. This has broad implications for treatment of people with type 2 diabetes and CKD,” he said.
The link with reduction in albuminuria was weaker for the primary cardiovascular disease outcome: CV death, nonfatal myocardial infarction, nonfatal stroke, or hospitalization for heart failure. The strongest effect on this outcome was only seen in Kaplan-Meier analysis in those on finerenone who had at least a 30% reduction in their UACR. Those on placebo and with a similarly robust 4-month reduction in UACR showed a much more modest cardiovascular benefit that resembled those on either finerenone or placebo who had a smaller, less than 30% UACR reduction. The mediation analysis of these data showed that UACR reduction accounted for about 37% of the observed cardiovascular benefit seen during the trials.
“The effect of UACR is much stronger for the kidney outcomes,” summed up Dr. Agarwal. The results suggest that for cardiovascular outcomes finerenone works through factors other than lowering of UACR, but he admitted that no one currently knows what those other factors might be.
Treat aggressively to lower UACR by 30%
“I wouldn’t stop finerenone treatment in people who do not get a 30% reduction in their UACR” because these analyses suggest that a portion of the overall benefits from finerenone occurs via other mechanisms, he said. But in patients whose UACR is not reduced by at least 30% “be more aggressive on other measures to reduce UACR,” he advised.
The mediation analyses he ran are “the first time this has been done in nephrology,” producing a “groundbreaking” analysis and finding, Dr. Agarwal said. He also highlighted that the findings primarily relate to the importance of controlling UACR rather than an endorsement of finerenone as the best way to achieve this.
“All I care about is that people think about UACR as a modifiable risk factor. It doesn’t have to be treated with finerenone. It could be a renin-angiotensin system inhibitor, it could be chlorthalidone [a thiazide diuretic]. It just happened that we had a large dataset of people treated with finerenone or placebo.”
He said that future mediation analyses should look at the link between outcomes and UACR reductions produced by agents from the classes of sodium-glucose cotransporter 2 inhibitors and the glucagonlike peptide–1 receptor agonists.
FIDELIO-DKD and FIGARO-DKD were both sponsored by Bayer, the company that markets finerenone. Dr. Agarwal has received personal fees and nonfinancial support from Bayer. He has also received personal fees and nonfinancial support from Akebia Therapeutics, AstraZeneca, Boehringer Ingelheim, Eli Lilly, and Vifor Pharma, and he is a member of data safety monitoring committees for Chinook and Vertex. Dr. Inker is a consultant to Diamtrix, and her department receives research funding from Chinook, Omeros, Reata, and Tricida.
PHILADELPHIA – Reducing albuminuria is a key mediator of the way finerenone (Kerendia, Bayer) reduces adverse renal and cardiovascular events in people with type 2 diabetes and chronic kidney disease (CKD), based on findings from two novel mediation analyses run on data from more than 12,000 people included in the two finerenone pivotal trials.
Results from these analyses showed that that finerenone treatment produced in the FIDELIO-DKD and FIGARO-DKD phase 3 trials. FIDELIO-DKD, which had protection against adverse kidney outcomes as its primary endpoint, supplied the data that led to finerenone’s approval in 2021 by the U.S. Food and Drug Administration for treating people with type 2 diabetes and CKD.
The findings of the mediation analyses underscore the important role that albuminuria plays in the nephropathy and related comorbidities associated with type 2 diabetes and CKD and highlight the importance of ongoing monitoring of albuminuria to guide treatments aimed at minimizing this pathology, said Rajiv Agarwal, MD, who presented a poster on the mediation analyses at Kidney Week 2023, organized by the American Society of Nephrology.
“My hope is that this [report] heightens awareness of UACR” as an important marker of both CKD and of the response by patients with CKD to their treatment, said Dr. Agarwal, a nephrologist and professor at Indiana University in Indianapolis.
“Only about half of people with type 2 diabetes get their UACR measured even though every guideline says measure UACR in people with diabetes. Our findings say that UACR is important not just for CKD diagnosis but also to give feedback” on whether management is working, Dr. Agarwal said in an interview.
Incorporate UACR into clinical decision-making
“My hope is that clinicians will look at UACR as something they should incorporate into clinical decision-making. I measure UACR in my patients [with CKD and type 2 diabetes] at every visit; it’s so inexpensive. Albuminuria is not a good sign. If it’s not reduced in a patient by at least 30% [the recommended minimum reduction by the American Diabetes Association for people who start with a UACR of at least 300 mg/g] clinicians should think of what else they could do to lower albuminuria”: Reduce salt intake, improve blood pressure control, make sure the patient is adherent to treatments, and add additional treatments, Dr. Agarwal advised.
Multiple efforts are now underway or will soon start to boost the rate at which at-risk people get their UACR measured, noted Leslie A. Inker, MD, in a separate talk during Kidney Week. These efforts include the National Kidney Foundation’s CKD Learning Collaborative, which aims to improve clinician awareness of CKD and improve routine testing for CKD. Early results during 2023 from this program in Missouri showed a nearly 8–percentage point increase in the screening rate for UACR levels in at-risk people, said Dr. Inker, professor and director of the Kidney and Blood Pressure Center at Tufts Medical Center in Boston.
A second advance was introduction in 2018 of the “kidney profile” lab order by the American College of Clinical Pathology that allows clinicians to order as a single test both an estimated glomerular filtration rate (eGFR) and a UACR.
Also, the Centers for Medicare & Medicaid Services and the National Committee for Quality Assurance have both taken steps to encourage UACR ordering. The NCQA established a new Healthcare Effectiveness Data and Information Set performance measure for U.S. physicians starting in 2023 that will track measurement of UACR and eGFR in people with diabetes. CMS also has made assessment of kidney health a measure of care quality in programs effective in 2023 and 2024, Dr. Inker noted.
Most subjects had elevated UACRs
The study run by Dr. Agarwal and his associates used data from 12,512 of the more than 13,000 people enrolled in either FIDELITY-DKD or FIGARO-DKD who had UACR measurements recorded at baseline, at 4 months into either study, or both. Their median UACR at the time they began on finerenone or placebo was 514 mg/g, with 67% having a UACR of at least 300 mg/g (macroalbuminuria) and 31% having a UACR of 30-299 mg/g (microalbuminuria). By design, virtually all patients in these two trials were on a renin-angiotensin system inhibitor (either an angiotensin-converting enzyme inhibitor or an angiotensin-receptor blocker), but given the time period when the two trials enrolled participants (during 2015-2018) only 7% of those enrolled were on a sodium-glucose cotransporter 2 inhibitor and only 7% were on a glucagonlike peptide–1 receptor agonist.
Four months after treatment began, 53% of those randomized to finerenone treatment and 27% of those in the placebo arm had their UACR reduced by at least 30% from baseline, the cutpoint chosen by Dr. Agarwal based on the American Diabetes Association guideline.
Kaplan-Meier analyses showed that the incidence of the primary kidney outcome – kidney failure, a sustained ≥ 57% decrease in eGFR from baseline, or kidney death – showed close correlation with at least a 30% reduction in UACR regardless of whether the patients in this subgroup received finerenone or placebo.
A different correlation was found in those with a less than 30% reduction in their UACR from baseline to 4 months, regardless of whether this happened on finerenone or placebo. People in the two finerenone trials who had a lesser reduction from baseline in their UACR also had a significantly higher rate of adverse kidney outcomes whether they received finerenone or placebo.
84% of finerenone’s kidney benefit linked to lowering of UACR
The causal-mediation analysis run by Dr. Agarwal quantified this observation, showing that 84% of finerenone’s effect on the kidney outcome was mediated by the reduction in UACR.
“It seems like the kidney benefit [from finerenone] travels through the level of albuminuria. This has broad implications for treatment of people with type 2 diabetes and CKD,” he said.
The link with reduction in albuminuria was weaker for the primary cardiovascular disease outcome: CV death, nonfatal myocardial infarction, nonfatal stroke, or hospitalization for heart failure. The strongest effect on this outcome was only seen in Kaplan-Meier analysis in those on finerenone who had at least a 30% reduction in their UACR. Those on placebo and with a similarly robust 4-month reduction in UACR showed a much more modest cardiovascular benefit that resembled those on either finerenone or placebo who had a smaller, less than 30% UACR reduction. The mediation analysis of these data showed that UACR reduction accounted for about 37% of the observed cardiovascular benefit seen during the trials.
“The effect of UACR is much stronger for the kidney outcomes,” summed up Dr. Agarwal. The results suggest that for cardiovascular outcomes finerenone works through factors other than lowering of UACR, but he admitted that no one currently knows what those other factors might be.
Treat aggressively to lower UACR by 30%
“I wouldn’t stop finerenone treatment in people who do not get a 30% reduction in their UACR” because these analyses suggest that a portion of the overall benefits from finerenone occurs via other mechanisms, he said. But in patients whose UACR is not reduced by at least 30% “be more aggressive on other measures to reduce UACR,” he advised.
The mediation analyses he ran are “the first time this has been done in nephrology,” producing a “groundbreaking” analysis and finding, Dr. Agarwal said. He also highlighted that the findings primarily relate to the importance of controlling UACR rather than an endorsement of finerenone as the best way to achieve this.
“All I care about is that people think about UACR as a modifiable risk factor. It doesn’t have to be treated with finerenone. It could be a renin-angiotensin system inhibitor, it could be chlorthalidone [a thiazide diuretic]. It just happened that we had a large dataset of people treated with finerenone or placebo.”
He said that future mediation analyses should look at the link between outcomes and UACR reductions produced by agents from the classes of sodium-glucose cotransporter 2 inhibitors and the glucagonlike peptide–1 receptor agonists.
FIDELIO-DKD and FIGARO-DKD were both sponsored by Bayer, the company that markets finerenone. Dr. Agarwal has received personal fees and nonfinancial support from Bayer. He has also received personal fees and nonfinancial support from Akebia Therapeutics, AstraZeneca, Boehringer Ingelheim, Eli Lilly, and Vifor Pharma, and he is a member of data safety monitoring committees for Chinook and Vertex. Dr. Inker is a consultant to Diamtrix, and her department receives research funding from Chinook, Omeros, Reata, and Tricida.
AT KIDNEY WEEK 2023
T2D: Real benefits of new oral antidiabetic drugs
Cardiovascular disease is the most common cause of death in people living with type 2 diabetes (T2D). It is true that patient prognoses have improved with the use of metformin and by addressing cardiovascular risk factors. But the new oral antidiabetic drugs, SGLT2 (sodium glucose cotransporter-2) inhibitors (SGLT2i), and glucagon-like peptide-1 receptor agonists (GLP-1Ra) offer fresh therapeutic approaches.
A cohort of more than 2 million patients with T2D
What about in the real world, far away from the ideal conditions of randomized trials? Could combining SGLT2 inhibitors with GLP-1R agonists be even more effective?
These are the questions answered by a large retrospective cohort study in which 2.2 million patients with T2D receiving insulin were initially enrolled and monitored at 85 specialist centers spread throughout three countries (Denmark, the United Kingdom, and the United States).
Three groups were formed from this cohort according to whether they received monotherapy or combination treatments: SGLT2i (n = 143,600), GLP-1Ra (n = 186,841), and SGLT2i + GLP-1Ra (n = 108,5040). A control group received none of these treatments.
Propensity score matching took into account the following relevant variables: age, sex, ischemic heart disease, hypertension, chronic kidney disease, heart failure, and glycated hemoglobin. The data was analyzed using the Cox’s proportional hazards model, with follow-up at 5 years.
Real-world benefits – Even better when combined
The inter-group comparison suggests that oral antidiabetic agents are effective when taking into account three major events:
All-cause mortality: SGLT2i (hazard ratio, 0.49; confidence interval 95% 0.48-0.50); GLP-1Ra (HR, 0.47; CI 95% 0.46-0.48); SGLT2i + GLP-1Ra (HR, 0.25; CI 95% 0.24-0.26).
Admissions rate: respectively HR: 0.73 (0.72-0.74); 0.69 (0.68-0.69); 0.60 (0.59-0.61).
Myocardial infarction rate: respectively HR: 0.75 (0.72-0.78); 0.70 (0.68-0.73); 0.63 (0.60-0.66).
A complementary sub-analysis also revealed a more significant reduction in all-cause mortality in the event of exposure to the combination of two antidiabetic drugs versus SGLT2i alone (HR, 0.53 [0.50-0.55]) and GLP-1Ra as monotherapy (HR, 0.56 [0.54-0.59]).
This real-world retrospective cohort study involves a large sample size: more than 400,000 patients with T2D treated with new oral antidiabetic drugs and as many control patients. It suggests that SGLT2 inhibitors and GLP-1R agonists have a significant effect on overall mortality, as well as on the risk of myocardial infarction and the admissions rate. Yes, it is retrospective, but its findings are in line with those from the most recent and conclusive randomized trials that suggest a cardio- and nephroprotective effect, at least with regard to SGLT2 inhibitors.
This article was translated from JIM and a version appeared on Medscape.com.
Cardiovascular disease is the most common cause of death in people living with type 2 diabetes (T2D). It is true that patient prognoses have improved with the use of metformin and by addressing cardiovascular risk factors. But the new oral antidiabetic drugs, SGLT2 (sodium glucose cotransporter-2) inhibitors (SGLT2i), and glucagon-like peptide-1 receptor agonists (GLP-1Ra) offer fresh therapeutic approaches.
A cohort of more than 2 million patients with T2D
What about in the real world, far away from the ideal conditions of randomized trials? Could combining SGLT2 inhibitors with GLP-1R agonists be even more effective?
These are the questions answered by a large retrospective cohort study in which 2.2 million patients with T2D receiving insulin were initially enrolled and monitored at 85 specialist centers spread throughout three countries (Denmark, the United Kingdom, and the United States).
Three groups were formed from this cohort according to whether they received monotherapy or combination treatments: SGLT2i (n = 143,600), GLP-1Ra (n = 186,841), and SGLT2i + GLP-1Ra (n = 108,5040). A control group received none of these treatments.
Propensity score matching took into account the following relevant variables: age, sex, ischemic heart disease, hypertension, chronic kidney disease, heart failure, and glycated hemoglobin. The data was analyzed using the Cox’s proportional hazards model, with follow-up at 5 years.
Real-world benefits – Even better when combined
The inter-group comparison suggests that oral antidiabetic agents are effective when taking into account three major events:
All-cause mortality: SGLT2i (hazard ratio, 0.49; confidence interval 95% 0.48-0.50); GLP-1Ra (HR, 0.47; CI 95% 0.46-0.48); SGLT2i + GLP-1Ra (HR, 0.25; CI 95% 0.24-0.26).
Admissions rate: respectively HR: 0.73 (0.72-0.74); 0.69 (0.68-0.69); 0.60 (0.59-0.61).
Myocardial infarction rate: respectively HR: 0.75 (0.72-0.78); 0.70 (0.68-0.73); 0.63 (0.60-0.66).
A complementary sub-analysis also revealed a more significant reduction in all-cause mortality in the event of exposure to the combination of two antidiabetic drugs versus SGLT2i alone (HR, 0.53 [0.50-0.55]) and GLP-1Ra as monotherapy (HR, 0.56 [0.54-0.59]).
This real-world retrospective cohort study involves a large sample size: more than 400,000 patients with T2D treated with new oral antidiabetic drugs and as many control patients. It suggests that SGLT2 inhibitors and GLP-1R agonists have a significant effect on overall mortality, as well as on the risk of myocardial infarction and the admissions rate. Yes, it is retrospective, but its findings are in line with those from the most recent and conclusive randomized trials that suggest a cardio- and nephroprotective effect, at least with regard to SGLT2 inhibitors.
This article was translated from JIM and a version appeared on Medscape.com.
Cardiovascular disease is the most common cause of death in people living with type 2 diabetes (T2D). It is true that patient prognoses have improved with the use of metformin and by addressing cardiovascular risk factors. But the new oral antidiabetic drugs, SGLT2 (sodium glucose cotransporter-2) inhibitors (SGLT2i), and glucagon-like peptide-1 receptor agonists (GLP-1Ra) offer fresh therapeutic approaches.
A cohort of more than 2 million patients with T2D
What about in the real world, far away from the ideal conditions of randomized trials? Could combining SGLT2 inhibitors with GLP-1R agonists be even more effective?
These are the questions answered by a large retrospective cohort study in which 2.2 million patients with T2D receiving insulin were initially enrolled and monitored at 85 specialist centers spread throughout three countries (Denmark, the United Kingdom, and the United States).
Three groups were formed from this cohort according to whether they received monotherapy or combination treatments: SGLT2i (n = 143,600), GLP-1Ra (n = 186,841), and SGLT2i + GLP-1Ra (n = 108,5040). A control group received none of these treatments.
Propensity score matching took into account the following relevant variables: age, sex, ischemic heart disease, hypertension, chronic kidney disease, heart failure, and glycated hemoglobin. The data was analyzed using the Cox’s proportional hazards model, with follow-up at 5 years.
Real-world benefits – Even better when combined
The inter-group comparison suggests that oral antidiabetic agents are effective when taking into account three major events:
All-cause mortality: SGLT2i (hazard ratio, 0.49; confidence interval 95% 0.48-0.50); GLP-1Ra (HR, 0.47; CI 95% 0.46-0.48); SGLT2i + GLP-1Ra (HR, 0.25; CI 95% 0.24-0.26).
Admissions rate: respectively HR: 0.73 (0.72-0.74); 0.69 (0.68-0.69); 0.60 (0.59-0.61).
Myocardial infarction rate: respectively HR: 0.75 (0.72-0.78); 0.70 (0.68-0.73); 0.63 (0.60-0.66).
A complementary sub-analysis also revealed a more significant reduction in all-cause mortality in the event of exposure to the combination of two antidiabetic drugs versus SGLT2i alone (HR, 0.53 [0.50-0.55]) and GLP-1Ra as monotherapy (HR, 0.56 [0.54-0.59]).
This real-world retrospective cohort study involves a large sample size: more than 400,000 patients with T2D treated with new oral antidiabetic drugs and as many control patients. It suggests that SGLT2 inhibitors and GLP-1R agonists have a significant effect on overall mortality, as well as on the risk of myocardial infarction and the admissions rate. Yes, it is retrospective, but its findings are in line with those from the most recent and conclusive randomized trials that suggest a cardio- and nephroprotective effect, at least with regard to SGLT2 inhibitors.
This article was translated from JIM and a version appeared on Medscape.com.
CKD-EPI eGFR formula surpasses alternatives in young adults
PHILADELPHIA –
The two alternative formulas for calculating eGFR, the CKiD U25 (Chronic Kidney Disease in Children under 25) and the European Kidney Function Consortium equations, showed higher levels of bias that resulted in underestimates of kidney function, particularly in younger adults 18-25 years old and in those with higher eGFR values, Leslie A. Inker, MD, said at Kidney Week 2023, organized by the American Society of Nephrology.
However, for young adults with a history of childhood CKD and especially for those who continue under the care of pediatric clinicians even after they become young adults, use of the CKiD U25 equation, remains a reasonable option, said Dr. Inker, professor and director of the Kidney and Blood Pressure Center at Tufts Medical Center in Boston. The CKiD U25 equation is intended for people aged 1-25 years and came out in late 2020.
Pediatric nephrologists use the CKiD U25 but the results of the 2021 CKD-EPI race-free equation is what U.S. clinical labs routinely report for people aged 18 or older. “Our findings support the current practice of pediatric nephrologists” who may opt to use the CKiD U25 even when a patient turns 18 years or older, Dr. Inker said in an interview.
But the new results also support the current practice of U.S. labs, which is to focus on calculating eGFR in anyone at least 18 years old using the 2021 equation developed by the CKD-EPI, work led by Dr. Inker. The new findings confirm routine use of the 2021 CKD-EPI equation in adults as young as 18 years old, especially when they’re having their eGFR calculated for the first time, she said.
Discontinuity changing from CKiD U25 to CKD-EPI is ‘not huge’
“It’s important to understand that the 2021 CKD-EPI equation works in young adults,” noted Josef Coresh, MD, PhD, professor of clinical epidemiology at Johns Hopkins University, Baltimore, who collaborated on both the current study and on developing the 2021 CKD-EPI equation.
The new data show that the discontinuity in eGFR produced by switching from the CKiD U25 formula to the 2021 CKD-EPI formula “is not huge, maybe about 5 mL/min per 1.73 m2 higher. People should focus on the new baseline and subsequent trends, not the modest difference between equations,” Dr. Coresh advised in an interview.
The study run by Dr. Inker and her associates used 1,491 people aged 18-40 years and enrolled in a cohort created by the CKD-EPI. They compared measured GFR levels in each subject with the estimates generated by the 2021 race-free CKD-EPI equation, the CKiD U25 equation, and a third equation developed by the EKFC and introduced in 2021.
Less bias with the 2021 CKD-EPI equation
The researchers compared the three eGFR equations with their respective measured GFR values by two metrics: bias, defined as the median difference between measured and estimated GFR; and the percentage of eGFR values that fell within 30% of the corresponding measured GFR value.
The results showed that bias was lowest using the 2021 CKD-EPI equation, with an overall median difference of 0.5 mL/min per 1.73 m2. This compared with median differences of 7.2 with the CKiD U25 equation and 4.9 mL/min per 1.73 m2 with the EKFC equation. The disparity in bias was greatest among those with eGFR values in the range of 60-90 mL/min per 1.73 m2 and was also greatest for those 18-25 years old.
The CKD-EPI equation results also showed the greatest consistency of bias across the entire 18- to 40-year-old range. Between-group differences were small for the percentage of eGFR values that fell within 30% of measured GFR, with all three equations scoring in the range of 88%-90%.
The study received no commercial funding. Dr. Inker is a consultant to Diamtrix and her department receives research funding from Chinook, Omeros, Reata, and Tricida. Dr. Coresh is a consultant to Healthy.io and SomaLogic and he has an ownership interest in Healthy.io.
PHILADELPHIA –
The two alternative formulas for calculating eGFR, the CKiD U25 (Chronic Kidney Disease in Children under 25) and the European Kidney Function Consortium equations, showed higher levels of bias that resulted in underestimates of kidney function, particularly in younger adults 18-25 years old and in those with higher eGFR values, Leslie A. Inker, MD, said at Kidney Week 2023, organized by the American Society of Nephrology.
However, for young adults with a history of childhood CKD and especially for those who continue under the care of pediatric clinicians even after they become young adults, use of the CKiD U25 equation, remains a reasonable option, said Dr. Inker, professor and director of the Kidney and Blood Pressure Center at Tufts Medical Center in Boston. The CKiD U25 equation is intended for people aged 1-25 years and came out in late 2020.
Pediatric nephrologists use the CKiD U25 but the results of the 2021 CKD-EPI race-free equation is what U.S. clinical labs routinely report for people aged 18 or older. “Our findings support the current practice of pediatric nephrologists” who may opt to use the CKiD U25 even when a patient turns 18 years or older, Dr. Inker said in an interview.
But the new results also support the current practice of U.S. labs, which is to focus on calculating eGFR in anyone at least 18 years old using the 2021 equation developed by the CKD-EPI, work led by Dr. Inker. The new findings confirm routine use of the 2021 CKD-EPI equation in adults as young as 18 years old, especially when they’re having their eGFR calculated for the first time, she said.
Discontinuity changing from CKiD U25 to CKD-EPI is ‘not huge’
“It’s important to understand that the 2021 CKD-EPI equation works in young adults,” noted Josef Coresh, MD, PhD, professor of clinical epidemiology at Johns Hopkins University, Baltimore, who collaborated on both the current study and on developing the 2021 CKD-EPI equation.
The new data show that the discontinuity in eGFR produced by switching from the CKiD U25 formula to the 2021 CKD-EPI formula “is not huge, maybe about 5 mL/min per 1.73 m2 higher. People should focus on the new baseline and subsequent trends, not the modest difference between equations,” Dr. Coresh advised in an interview.
The study run by Dr. Inker and her associates used 1,491 people aged 18-40 years and enrolled in a cohort created by the CKD-EPI. They compared measured GFR levels in each subject with the estimates generated by the 2021 race-free CKD-EPI equation, the CKiD U25 equation, and a third equation developed by the EKFC and introduced in 2021.
Less bias with the 2021 CKD-EPI equation
The researchers compared the three eGFR equations with their respective measured GFR values by two metrics: bias, defined as the median difference between measured and estimated GFR; and the percentage of eGFR values that fell within 30% of the corresponding measured GFR value.
The results showed that bias was lowest using the 2021 CKD-EPI equation, with an overall median difference of 0.5 mL/min per 1.73 m2. This compared with median differences of 7.2 with the CKiD U25 equation and 4.9 mL/min per 1.73 m2 with the EKFC equation. The disparity in bias was greatest among those with eGFR values in the range of 60-90 mL/min per 1.73 m2 and was also greatest for those 18-25 years old.
The CKD-EPI equation results also showed the greatest consistency of bias across the entire 18- to 40-year-old range. Between-group differences were small for the percentage of eGFR values that fell within 30% of measured GFR, with all three equations scoring in the range of 88%-90%.
The study received no commercial funding. Dr. Inker is a consultant to Diamtrix and her department receives research funding from Chinook, Omeros, Reata, and Tricida. Dr. Coresh is a consultant to Healthy.io and SomaLogic and he has an ownership interest in Healthy.io.
PHILADELPHIA –
The two alternative formulas for calculating eGFR, the CKiD U25 (Chronic Kidney Disease in Children under 25) and the European Kidney Function Consortium equations, showed higher levels of bias that resulted in underestimates of kidney function, particularly in younger adults 18-25 years old and in those with higher eGFR values, Leslie A. Inker, MD, said at Kidney Week 2023, organized by the American Society of Nephrology.
However, for young adults with a history of childhood CKD and especially for those who continue under the care of pediatric clinicians even after they become young adults, use of the CKiD U25 equation, remains a reasonable option, said Dr. Inker, professor and director of the Kidney and Blood Pressure Center at Tufts Medical Center in Boston. The CKiD U25 equation is intended for people aged 1-25 years and came out in late 2020.
Pediatric nephrologists use the CKiD U25 but the results of the 2021 CKD-EPI race-free equation is what U.S. clinical labs routinely report for people aged 18 or older. “Our findings support the current practice of pediatric nephrologists” who may opt to use the CKiD U25 even when a patient turns 18 years or older, Dr. Inker said in an interview.
But the new results also support the current practice of U.S. labs, which is to focus on calculating eGFR in anyone at least 18 years old using the 2021 equation developed by the CKD-EPI, work led by Dr. Inker. The new findings confirm routine use of the 2021 CKD-EPI equation in adults as young as 18 years old, especially when they’re having their eGFR calculated for the first time, she said.
Discontinuity changing from CKiD U25 to CKD-EPI is ‘not huge’
“It’s important to understand that the 2021 CKD-EPI equation works in young adults,” noted Josef Coresh, MD, PhD, professor of clinical epidemiology at Johns Hopkins University, Baltimore, who collaborated on both the current study and on developing the 2021 CKD-EPI equation.
The new data show that the discontinuity in eGFR produced by switching from the CKiD U25 formula to the 2021 CKD-EPI formula “is not huge, maybe about 5 mL/min per 1.73 m2 higher. People should focus on the new baseline and subsequent trends, not the modest difference between equations,” Dr. Coresh advised in an interview.
The study run by Dr. Inker and her associates used 1,491 people aged 18-40 years and enrolled in a cohort created by the CKD-EPI. They compared measured GFR levels in each subject with the estimates generated by the 2021 race-free CKD-EPI equation, the CKiD U25 equation, and a third equation developed by the EKFC and introduced in 2021.
Less bias with the 2021 CKD-EPI equation
The researchers compared the three eGFR equations with their respective measured GFR values by two metrics: bias, defined as the median difference between measured and estimated GFR; and the percentage of eGFR values that fell within 30% of the corresponding measured GFR value.
The results showed that bias was lowest using the 2021 CKD-EPI equation, with an overall median difference of 0.5 mL/min per 1.73 m2. This compared with median differences of 7.2 with the CKiD U25 equation and 4.9 mL/min per 1.73 m2 with the EKFC equation. The disparity in bias was greatest among those with eGFR values in the range of 60-90 mL/min per 1.73 m2 and was also greatest for those 18-25 years old.
The CKD-EPI equation results also showed the greatest consistency of bias across the entire 18- to 40-year-old range. Between-group differences were small for the percentage of eGFR values that fell within 30% of measured GFR, with all three equations scoring in the range of 88%-90%.
The study received no commercial funding. Dr. Inker is a consultant to Diamtrix and her department receives research funding from Chinook, Omeros, Reata, and Tricida. Dr. Coresh is a consultant to Healthy.io and SomaLogic and he has an ownership interest in Healthy.io.
AT KIDNEY WEEK 2023
Blood pressure lowering reduces dementia risk
Results of a trial using an intensive, 4-year program aimed at blood pressure lowering showed that intervention reduced not only blood pressure, but also significantly reduced the risk of total dementia over that period.
and cognitive impairment no dementia (CIND), a secondary outcome, was also significantly reduced by 16%.
“Blood pressure reduction is effective in reducing the risk of dementia in patients with hypertension,” concluded Jiang He, MD, PhD, professor of epidemiology and medicine and director of Tulane University’s Translational Science Institute, New Orleans. “This proven, effective intervention should be widely scaled up to reduce the global burden of dementia.”
He presented these results from the China Rural Hypertension Control Project (CRHCP) at the annual scientific sessions of the American Heart Association.
Target organ damage
Keith Ferdinand, MD, also from Tulane University, commented on the findings during a press conference at the meeting, noting that the result “opens our opportunity to recognize that the target organ damage of hypertension also now includes dementia.”
The researchers were able to “rigorously lower blood pressure from 157 to 127.6 in the intervention, 155 to 147 in the controls – 22 mg Hg – and if you look at the P values for all the various outcomes, they were very robust,” Dr. Ferdinand said.
Another interesting feature about the strategy used in this trial is that “this was true team-based care,” he pointed out. The trained interventionists in the study, called village doctors, collaborated with primary care physicians and initiated medications. “They stayed on a simple treatment protocol, and they were able to assist patients to ensure they had free medications, health coaching for lifestyle, home blood pressure measurement, and ensuring adherence.”
So, Dr. Ferdinand added, “one of the questions is whether this is a model we can use in other places around the globe, in places with low resources, and in the United States in disadvantaged populations.”
Public health priority
It’s estimated that the global number of those living with dementia will increase from 57.4 million in 2019 to 152.8 million by 2050, Dr. He said. “In the absence of curative treatment, the primary prevention of dementia through risk factor reduction, such as blood pressure lowering, becomes a public health priority.”
Previous randomized trials have lacked sample size and duration but have reported a nonsignificant reduction in dementia associated with antihypertensive treatment in patients with hypertension or a history of stroke, Dr. He noted.
This new trial aimed to test the effectiveness of intensive BP intervention to reduce the risk of all-cause dementia and cognitive impairment over a 48-month intervention period versus usual care.
It was an open-label, blinded-endpoint, cluster-randomized trial, and included 33,995 individual patients from 325 villages in China, aged 40 years and older, with untreated hypertension. The villages were randomly assigned to an intervention group or usual care, stratified by province, county, and township.
Patients were eligible if they had mean untreated systolic BP greater than 140 mm Hg and/or diastolic BP greater than 90 mm Hg or mean treated systolic BP of greater than 130 and/or diastolic greater than 80 mm Hg. Patients with a history of cardiovascular disease, chronic kidney disease, or diabetes and a mean systolic BP greater than 130 mm Hg and/or diastolic BP greater than 80 mm Hg from six measures on two different days were also eligible.
All were enrolled in the China New Rural Cooperative Medical Scheme, which covers 99% of rural residents for health care services, Dr. He noted.
The intervention was a simple stepped-care protocol for hypertension treatment, aimed at achieving a target systolic BP of less than 130 mm Hg and diastolic of less than 80 mm Hg.
Village doctors started and titrated antihypertensive treatment based on a protocol and were able to deliver discounted and free medications to patients. They also did health coaching on lifestyle modification and adherence to medication, and instructed patients on home BP monitoring.
Patients were provided training, supervision, and consultation by primary care physicians and hypertension specialists.
At the month 48 follow-up visit, the participants were assessed by neurologists who were blinded to randomization assignments. Neurologists did a variety of tests and assessments including collecting data on the patient’s medical and psychiatric history and risk factors for dementia, as well as neurologic assessment using the Mini-Mental State Examination, the Functional Activities Questionnaire, and the Quick Dementia Rating System.
The primary outcome was all-cause dementia, defined according to recommendations from the National Institute on Aging–Alzheimer’s Association work groups on diagnostic guidelines for Alzheimer’s disease.
Secondary outcomes included CIND, a composite outcome of dementia or CIND, and a composite of dementia or deaths.
The final diagnosis of all-cause dementia or CIND was made by an expert adjudication panel blinded to the intervention assignment.
At 48 months, 91.3% of patients completed the follow-up for clinical outcomes. Participants were an average of 63 years of age, 61% were female, and 23% had less than a primary school education, Dr. He noted.
The net group differences in systolic and diastolic BP reduction were 22 and 9.3 mm Hg, respectively (P < .0001).
Significant differences were also seen between the groups in the primary outcome of all-cause dementia, as well as secondary outcomes of CIND, dementia or cognitive impairment, or dementia or deaths.
Serious adverse events were more common in the usual care group, and there was no difference between groups in the occurrence of falls or syncope.
The effect was consistent across subgroups, Dr. He said, including age, sex, education, cigarette smoking, body mass index, systolic BP, and fasting plasma glucose at baseline.
First definitive evidence
Invited discussant for the trial, Daniel W. Jones, MD, University of Mississippi Medical Center, Jackson, and past president of the AHA, pointed out that previous results from CRHCP on cardiovascular outcomes, reported earlier in 2023 in The Lancet, showed that, similar to results of the large SPRINT trial, lowering systolic BP to a goal of less than 130 mm Hg reduced a composite endpoint of MI, stroke, heart failure requiring hospitalization, and cardiovascular disease death over the 36-month follow-up.
The SPRINT findings also suggested a possible reduction in dementia, Dr. Jones said.
Now, in these new CRHCP results, “there was a clear benefit for intensive BP control in reducing risk for dementia and cognitive dysfunction,” he said. “This is, importantly, the first definitive evidence of dementia risk reduction demonstrated in a randomized controlled clinical trial. This outcome supports observational data that shows a strong relationship between BP and dementia.”
Since it is the first of its kind though, replication of the results will be important, he noted.
The study also showed that the intervention, using minimally trained village doctors, sustained BP control for 48 months. “This model could be used in any setting with modifications, including in the United States,” Dr. Jones said.
The study was supported by the Ministry of Science and Technology of China; U.S. investigators did not receive financial support from this study. The researchers and Dr. Jones disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Results of a trial using an intensive, 4-year program aimed at blood pressure lowering showed that intervention reduced not only blood pressure, but also significantly reduced the risk of total dementia over that period.
and cognitive impairment no dementia (CIND), a secondary outcome, was also significantly reduced by 16%.
“Blood pressure reduction is effective in reducing the risk of dementia in patients with hypertension,” concluded Jiang He, MD, PhD, professor of epidemiology and medicine and director of Tulane University’s Translational Science Institute, New Orleans. “This proven, effective intervention should be widely scaled up to reduce the global burden of dementia.”
He presented these results from the China Rural Hypertension Control Project (CRHCP) at the annual scientific sessions of the American Heart Association.
Target organ damage
Keith Ferdinand, MD, also from Tulane University, commented on the findings during a press conference at the meeting, noting that the result “opens our opportunity to recognize that the target organ damage of hypertension also now includes dementia.”
The researchers were able to “rigorously lower blood pressure from 157 to 127.6 in the intervention, 155 to 147 in the controls – 22 mg Hg – and if you look at the P values for all the various outcomes, they were very robust,” Dr. Ferdinand said.
Another interesting feature about the strategy used in this trial is that “this was true team-based care,” he pointed out. The trained interventionists in the study, called village doctors, collaborated with primary care physicians and initiated medications. “They stayed on a simple treatment protocol, and they were able to assist patients to ensure they had free medications, health coaching for lifestyle, home blood pressure measurement, and ensuring adherence.”
So, Dr. Ferdinand added, “one of the questions is whether this is a model we can use in other places around the globe, in places with low resources, and in the United States in disadvantaged populations.”
Public health priority
It’s estimated that the global number of those living with dementia will increase from 57.4 million in 2019 to 152.8 million by 2050, Dr. He said. “In the absence of curative treatment, the primary prevention of dementia through risk factor reduction, such as blood pressure lowering, becomes a public health priority.”
Previous randomized trials have lacked sample size and duration but have reported a nonsignificant reduction in dementia associated with antihypertensive treatment in patients with hypertension or a history of stroke, Dr. He noted.
This new trial aimed to test the effectiveness of intensive BP intervention to reduce the risk of all-cause dementia and cognitive impairment over a 48-month intervention period versus usual care.
It was an open-label, blinded-endpoint, cluster-randomized trial, and included 33,995 individual patients from 325 villages in China, aged 40 years and older, with untreated hypertension. The villages were randomly assigned to an intervention group or usual care, stratified by province, county, and township.
Patients were eligible if they had mean untreated systolic BP greater than 140 mm Hg and/or diastolic BP greater than 90 mm Hg or mean treated systolic BP of greater than 130 and/or diastolic greater than 80 mm Hg. Patients with a history of cardiovascular disease, chronic kidney disease, or diabetes and a mean systolic BP greater than 130 mm Hg and/or diastolic BP greater than 80 mm Hg from six measures on two different days were also eligible.
All were enrolled in the China New Rural Cooperative Medical Scheme, which covers 99% of rural residents for health care services, Dr. He noted.
The intervention was a simple stepped-care protocol for hypertension treatment, aimed at achieving a target systolic BP of less than 130 mm Hg and diastolic of less than 80 mm Hg.
Village doctors started and titrated antihypertensive treatment based on a protocol and were able to deliver discounted and free medications to patients. They also did health coaching on lifestyle modification and adherence to medication, and instructed patients on home BP monitoring.
Patients were provided training, supervision, and consultation by primary care physicians and hypertension specialists.
At the month 48 follow-up visit, the participants were assessed by neurologists who were blinded to randomization assignments. Neurologists did a variety of tests and assessments including collecting data on the patient’s medical and psychiatric history and risk factors for dementia, as well as neurologic assessment using the Mini-Mental State Examination, the Functional Activities Questionnaire, and the Quick Dementia Rating System.
The primary outcome was all-cause dementia, defined according to recommendations from the National Institute on Aging–Alzheimer’s Association work groups on diagnostic guidelines for Alzheimer’s disease.
Secondary outcomes included CIND, a composite outcome of dementia or CIND, and a composite of dementia or deaths.
The final diagnosis of all-cause dementia or CIND was made by an expert adjudication panel blinded to the intervention assignment.
At 48 months, 91.3% of patients completed the follow-up for clinical outcomes. Participants were an average of 63 years of age, 61% were female, and 23% had less than a primary school education, Dr. He noted.
The net group differences in systolic and diastolic BP reduction were 22 and 9.3 mm Hg, respectively (P < .0001).
Significant differences were also seen between the groups in the primary outcome of all-cause dementia, as well as secondary outcomes of CIND, dementia or cognitive impairment, or dementia or deaths.
Serious adverse events were more common in the usual care group, and there was no difference between groups in the occurrence of falls or syncope.
The effect was consistent across subgroups, Dr. He said, including age, sex, education, cigarette smoking, body mass index, systolic BP, and fasting plasma glucose at baseline.
First definitive evidence
Invited discussant for the trial, Daniel W. Jones, MD, University of Mississippi Medical Center, Jackson, and past president of the AHA, pointed out that previous results from CRHCP on cardiovascular outcomes, reported earlier in 2023 in The Lancet, showed that, similar to results of the large SPRINT trial, lowering systolic BP to a goal of less than 130 mm Hg reduced a composite endpoint of MI, stroke, heart failure requiring hospitalization, and cardiovascular disease death over the 36-month follow-up.
The SPRINT findings also suggested a possible reduction in dementia, Dr. Jones said.
Now, in these new CRHCP results, “there was a clear benefit for intensive BP control in reducing risk for dementia and cognitive dysfunction,” he said. “This is, importantly, the first definitive evidence of dementia risk reduction demonstrated in a randomized controlled clinical trial. This outcome supports observational data that shows a strong relationship between BP and dementia.”
Since it is the first of its kind though, replication of the results will be important, he noted.
The study also showed that the intervention, using minimally trained village doctors, sustained BP control for 48 months. “This model could be used in any setting with modifications, including in the United States,” Dr. Jones said.
The study was supported by the Ministry of Science and Technology of China; U.S. investigators did not receive financial support from this study. The researchers and Dr. Jones disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Results of a trial using an intensive, 4-year program aimed at blood pressure lowering showed that intervention reduced not only blood pressure, but also significantly reduced the risk of total dementia over that period.
and cognitive impairment no dementia (CIND), a secondary outcome, was also significantly reduced by 16%.
“Blood pressure reduction is effective in reducing the risk of dementia in patients with hypertension,” concluded Jiang He, MD, PhD, professor of epidemiology and medicine and director of Tulane University’s Translational Science Institute, New Orleans. “This proven, effective intervention should be widely scaled up to reduce the global burden of dementia.”
He presented these results from the China Rural Hypertension Control Project (CRHCP) at the annual scientific sessions of the American Heart Association.
Target organ damage
Keith Ferdinand, MD, also from Tulane University, commented on the findings during a press conference at the meeting, noting that the result “opens our opportunity to recognize that the target organ damage of hypertension also now includes dementia.”
The researchers were able to “rigorously lower blood pressure from 157 to 127.6 in the intervention, 155 to 147 in the controls – 22 mg Hg – and if you look at the P values for all the various outcomes, they were very robust,” Dr. Ferdinand said.
Another interesting feature about the strategy used in this trial is that “this was true team-based care,” he pointed out. The trained interventionists in the study, called village doctors, collaborated with primary care physicians and initiated medications. “They stayed on a simple treatment protocol, and they were able to assist patients to ensure they had free medications, health coaching for lifestyle, home blood pressure measurement, and ensuring adherence.”
So, Dr. Ferdinand added, “one of the questions is whether this is a model we can use in other places around the globe, in places with low resources, and in the United States in disadvantaged populations.”
Public health priority
It’s estimated that the global number of those living with dementia will increase from 57.4 million in 2019 to 152.8 million by 2050, Dr. He said. “In the absence of curative treatment, the primary prevention of dementia through risk factor reduction, such as blood pressure lowering, becomes a public health priority.”
Previous randomized trials have lacked sample size and duration but have reported a nonsignificant reduction in dementia associated with antihypertensive treatment in patients with hypertension or a history of stroke, Dr. He noted.
This new trial aimed to test the effectiveness of intensive BP intervention to reduce the risk of all-cause dementia and cognitive impairment over a 48-month intervention period versus usual care.
It was an open-label, blinded-endpoint, cluster-randomized trial, and included 33,995 individual patients from 325 villages in China, aged 40 years and older, with untreated hypertension. The villages were randomly assigned to an intervention group or usual care, stratified by province, county, and township.
Patients were eligible if they had mean untreated systolic BP greater than 140 mm Hg and/or diastolic BP greater than 90 mm Hg or mean treated systolic BP of greater than 130 and/or diastolic greater than 80 mm Hg. Patients with a history of cardiovascular disease, chronic kidney disease, or diabetes and a mean systolic BP greater than 130 mm Hg and/or diastolic BP greater than 80 mm Hg from six measures on two different days were also eligible.
All were enrolled in the China New Rural Cooperative Medical Scheme, which covers 99% of rural residents for health care services, Dr. He noted.
The intervention was a simple stepped-care protocol for hypertension treatment, aimed at achieving a target systolic BP of less than 130 mm Hg and diastolic of less than 80 mm Hg.
Village doctors started and titrated antihypertensive treatment based on a protocol and were able to deliver discounted and free medications to patients. They also did health coaching on lifestyle modification and adherence to medication, and instructed patients on home BP monitoring.
Patients were provided training, supervision, and consultation by primary care physicians and hypertension specialists.
At the month 48 follow-up visit, the participants were assessed by neurologists who were blinded to randomization assignments. Neurologists did a variety of tests and assessments including collecting data on the patient’s medical and psychiatric history and risk factors for dementia, as well as neurologic assessment using the Mini-Mental State Examination, the Functional Activities Questionnaire, and the Quick Dementia Rating System.
The primary outcome was all-cause dementia, defined according to recommendations from the National Institute on Aging–Alzheimer’s Association work groups on diagnostic guidelines for Alzheimer’s disease.
Secondary outcomes included CIND, a composite outcome of dementia or CIND, and a composite of dementia or deaths.
The final diagnosis of all-cause dementia or CIND was made by an expert adjudication panel blinded to the intervention assignment.
At 48 months, 91.3% of patients completed the follow-up for clinical outcomes. Participants were an average of 63 years of age, 61% were female, and 23% had less than a primary school education, Dr. He noted.
The net group differences in systolic and diastolic BP reduction were 22 and 9.3 mm Hg, respectively (P < .0001).
Significant differences were also seen between the groups in the primary outcome of all-cause dementia, as well as secondary outcomes of CIND, dementia or cognitive impairment, or dementia or deaths.
Serious adverse events were more common in the usual care group, and there was no difference between groups in the occurrence of falls or syncope.
The effect was consistent across subgroups, Dr. He said, including age, sex, education, cigarette smoking, body mass index, systolic BP, and fasting plasma glucose at baseline.
First definitive evidence
Invited discussant for the trial, Daniel W. Jones, MD, University of Mississippi Medical Center, Jackson, and past president of the AHA, pointed out that previous results from CRHCP on cardiovascular outcomes, reported earlier in 2023 in The Lancet, showed that, similar to results of the large SPRINT trial, lowering systolic BP to a goal of less than 130 mm Hg reduced a composite endpoint of MI, stroke, heart failure requiring hospitalization, and cardiovascular disease death over the 36-month follow-up.
The SPRINT findings also suggested a possible reduction in dementia, Dr. Jones said.
Now, in these new CRHCP results, “there was a clear benefit for intensive BP control in reducing risk for dementia and cognitive dysfunction,” he said. “This is, importantly, the first definitive evidence of dementia risk reduction demonstrated in a randomized controlled clinical trial. This outcome supports observational data that shows a strong relationship between BP and dementia.”
Since it is the first of its kind though, replication of the results will be important, he noted.
The study also showed that the intervention, using minimally trained village doctors, sustained BP control for 48 months. “This model could be used in any setting with modifications, including in the United States,” Dr. Jones said.
The study was supported by the Ministry of Science and Technology of China; U.S. investigators did not receive financial support from this study. The researchers and Dr. Jones disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM AHA 2023
Obinutuzumab promotes renal preservation in lupus nephritis
TOPLINE:
Adults with lupus nephritis (LN) who received obinutuzumab (Gazyva) plus standard of care therapy experienced significantly improved kidney function and fewer flares compared with those given a placebo plus standard of care.
METHODOLOGY:
- Researchers conducted a post hoc analysis of the phase 2 NOBILITY study, a randomized trial in which 63 adults received 1,000 mg of obinutuzumab or placebo by infusion on day 1 and at weeks 2, 24, and 26.
- Outcomes were time to an unfavorable kidney outcome, defined by the first of any of the following events: treatment failure, doubling of serum creatinine, or death; researchers also measured LN flare outcomes including the first 30% and 40% declines in estimated glomerular filtration rate (eGFR) from baseline, chronic eGFR slope, and how many patients achieved complete renal response (CRR) on no more than 7.5 mg of prednisone.
TAKEAWAY:
- Adding obinutuzumab to the treatment of patients with LN reduced the risk of the composite outcome by 60% and reduced the risk for LN flare by 57%.
- The risk of first eGFR 30% and 40% decline was reduced by 80% and 91%, respectively, with obinutuzumab, and patients who took obinutuzumab had a significantly slower eGFR decline than with placebo (annualized eGFR slope advantage, 4.1 mL/min/1.73 m2 /year).
- At 76 weeks (1.5 years), 38% of patients receiving obinutuzumab achieved CRR on 7.5 mg or less of daily prednisone, compared with 16% of placebo patients, but this difference was not statistically significant at 104 weeks (2 years).
- The total numbers of unfavorable kidney outcomes for obinutuzumab vs. placebo were 12 vs. 24 for treatment failure, 1 vs. 6 for creatinine doubling, and 1 vs. 4 for death, respectively.
IN PRACTICE:
“By reducing flare risk, obinutuzumab should decrease the accumulation of chronic parenchymal kidney damage,” the authors wrote.
SOURCE:
The study was presented at the American College of Rheumatology (ACR) 2023 annual meeting and was published online in Arthritis & Rheumatology. The lead author was Brad H. Rovin, MD, of The Ohio State University in Columbus.
LIMITATIONS:
The analyses were post hoc and not prespecified, and the number of events was small, which prevented statistical confirmation, but the analyses are being repeated in an ongoing phase 3 study.
DISCLOSURES:
The study was supported by F. Hoffman–La Roche. Dr. Rovin reported receiving personal fees from F. Hoffman–La Roche during the conduct of the original trial. Several coauthors are F. Hoffman–La Roche employees.
A version of this article first appeared on Medscape.com.
TOPLINE:
Adults with lupus nephritis (LN) who received obinutuzumab (Gazyva) plus standard of care therapy experienced significantly improved kidney function and fewer flares compared with those given a placebo plus standard of care.
METHODOLOGY:
- Researchers conducted a post hoc analysis of the phase 2 NOBILITY study, a randomized trial in which 63 adults received 1,000 mg of obinutuzumab or placebo by infusion on day 1 and at weeks 2, 24, and 26.
- Outcomes were time to an unfavorable kidney outcome, defined by the first of any of the following events: treatment failure, doubling of serum creatinine, or death; researchers also measured LN flare outcomes including the first 30% and 40% declines in estimated glomerular filtration rate (eGFR) from baseline, chronic eGFR slope, and how many patients achieved complete renal response (CRR) on no more than 7.5 mg of prednisone.
TAKEAWAY:
- Adding obinutuzumab to the treatment of patients with LN reduced the risk of the composite outcome by 60% and reduced the risk for LN flare by 57%.
- The risk of first eGFR 30% and 40% decline was reduced by 80% and 91%, respectively, with obinutuzumab, and patients who took obinutuzumab had a significantly slower eGFR decline than with placebo (annualized eGFR slope advantage, 4.1 mL/min/1.73 m2 /year).
- At 76 weeks (1.5 years), 38% of patients receiving obinutuzumab achieved CRR on 7.5 mg or less of daily prednisone, compared with 16% of placebo patients, but this difference was not statistically significant at 104 weeks (2 years).
- The total numbers of unfavorable kidney outcomes for obinutuzumab vs. placebo were 12 vs. 24 for treatment failure, 1 vs. 6 for creatinine doubling, and 1 vs. 4 for death, respectively.
IN PRACTICE:
“By reducing flare risk, obinutuzumab should decrease the accumulation of chronic parenchymal kidney damage,” the authors wrote.
SOURCE:
The study was presented at the American College of Rheumatology (ACR) 2023 annual meeting and was published online in Arthritis & Rheumatology. The lead author was Brad H. Rovin, MD, of The Ohio State University in Columbus.
LIMITATIONS:
The analyses were post hoc and not prespecified, and the number of events was small, which prevented statistical confirmation, but the analyses are being repeated in an ongoing phase 3 study.
DISCLOSURES:
The study was supported by F. Hoffman–La Roche. Dr. Rovin reported receiving personal fees from F. Hoffman–La Roche during the conduct of the original trial. Several coauthors are F. Hoffman–La Roche employees.
A version of this article first appeared on Medscape.com.
TOPLINE:
Adults with lupus nephritis (LN) who received obinutuzumab (Gazyva) plus standard of care therapy experienced significantly improved kidney function and fewer flares compared with those given a placebo plus standard of care.
METHODOLOGY:
- Researchers conducted a post hoc analysis of the phase 2 NOBILITY study, a randomized trial in which 63 adults received 1,000 mg of obinutuzumab or placebo by infusion on day 1 and at weeks 2, 24, and 26.
- Outcomes were time to an unfavorable kidney outcome, defined by the first of any of the following events: treatment failure, doubling of serum creatinine, or death; researchers also measured LN flare outcomes including the first 30% and 40% declines in estimated glomerular filtration rate (eGFR) from baseline, chronic eGFR slope, and how many patients achieved complete renal response (CRR) on no more than 7.5 mg of prednisone.
TAKEAWAY:
- Adding obinutuzumab to the treatment of patients with LN reduced the risk of the composite outcome by 60% and reduced the risk for LN flare by 57%.
- The risk of first eGFR 30% and 40% decline was reduced by 80% and 91%, respectively, with obinutuzumab, and patients who took obinutuzumab had a significantly slower eGFR decline than with placebo (annualized eGFR slope advantage, 4.1 mL/min/1.73 m2 /year).
- At 76 weeks (1.5 years), 38% of patients receiving obinutuzumab achieved CRR on 7.5 mg or less of daily prednisone, compared with 16% of placebo patients, but this difference was not statistically significant at 104 weeks (2 years).
- The total numbers of unfavorable kidney outcomes for obinutuzumab vs. placebo were 12 vs. 24 for treatment failure, 1 vs. 6 for creatinine doubling, and 1 vs. 4 for death, respectively.
IN PRACTICE:
“By reducing flare risk, obinutuzumab should decrease the accumulation of chronic parenchymal kidney damage,” the authors wrote.
SOURCE:
The study was presented at the American College of Rheumatology (ACR) 2023 annual meeting and was published online in Arthritis & Rheumatology. The lead author was Brad H. Rovin, MD, of The Ohio State University in Columbus.
LIMITATIONS:
The analyses were post hoc and not prespecified, and the number of events was small, which prevented statistical confirmation, but the analyses are being repeated in an ongoing phase 3 study.
DISCLOSURES:
The study was supported by F. Hoffman–La Roche. Dr. Rovin reported receiving personal fees from F. Hoffman–La Roche during the conduct of the original trial. Several coauthors are F. Hoffman–La Roche employees.
A version of this article first appeared on Medscape.com.
Risk calculator for early-stage CKD may soon enter U.S. market
PHILADELPHIA – The analyses offer the possibility of focusing intensified medical management of early-stage CKD on those patients who could potentially receive the most benefit.
The Klinrisk model predicts the risk of an adult with early-stage CKD developing either a 40% or greater drop in estimated glomerular filtration rate or kidney failure. It calculates risk based on 20 lab-measured variables that include serum creatinine, urine albumin-to-creatinine ratio, and other values taken from routinely ordered tests such as complete blood cell counts, chemistry panels, comprehensive metabolic panels, and urinalysis.
In the most recent and largest external validation study using data from 4.6 million American adults enrolled in commercial and Medicare insurance plans, the results showed Klinrisk correctly predicted CKD progression in 80%-83% of individuals over 2 years and in 78%-83% of individuals over 5 years, depending on the insurance provider, Navdeep Tangri, MD, PhD, reported at the annual meeting of the American Society of Nephrology. When urinalysis data were available, the model correctly predicted CKD progression in 81%-87% of individuals over 2 years and in 80%-87% of individuals over 5 years. These results follow prior reports of several other successful validations of Klinrisk.
‘Ready to implement’
“The Klinrisk model is ready to implement by any payer, health system, or clinic where the needed lab data are available,” said Dr. Tangri, a nephrologist and professor at the University of Manitoba, Winnipeg, and founder of Klinrisk Inc., the company developing and commercializing the Klinrisk assessment tool.
For the time being, Dr. Tangri sees Klinrisk as a population health device that can allow insurers and health systems to track management quality and quality improvement and to target patients who stand to benefit most from relatively expensive resources. This includes prescriptions for finerenone (Kerendia, Bayer) for people who also have type 2 diabetes, and agents from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors such as dapagliflozin (Farxiga, AstraZeneca) and empagliflozin (Jardiance, Boehringer Ingelheim and Lilly).
He has also begun discussions with the Food and Drug Administration about the data the agency will need to consider Klinrisk for potential approval as a new medical device, perhaps in 2025. That’s how he envisions getting a Klinrisk assessment into the hands of caregivers that they could use with individual patients to create an appropriate treatment plan.
Results from his new analysis showed that “all the kidney disease action is in the 10%-20% of people with the highest risk on Klinrisk, while not much happens in those in the bottom half,” Dr. Tangri said during his presentation.
“We’re trying to find the patients who get the largest [absolute] benefit from intensified treatment,” he added in an interview. “Klinrisk finds people with high-risk kidney disease early on, when kidney function is still normal or near normal. High-risk patients are often completely unrecognized. Risk-based management” that identifies the early-stage CKD patients who would benefit most from treatment with an SGLT2 inhibitor, finerenone, and other foundational treatments to slow CKD progression “is better than the free-for-all that occurs today.”
Simplified data collection
“Klinrisk is very effective,” but requires follow-up by clinicians and health systems to implement its findings, commented Josef Coresh, MD, a professor of clinical epidemiology at Johns Hopkins Bloomberg, Baltimore. Dr. Coresh compared it with a free equation that estimates a person’s risk for a 40% drop in kidney function over the next 3 years developed by Dr. Tangri, Dr. Coresh, and many collaborators led by Morgan C. Grams, MD, PhD, of New York University that they published in 2022, and posted on a website of the CKD Prognosis Consortium.
The CKD Prognosis Consortium formula “takes a different approach” from Klinrisk. The commercial formula “is simpler, only using lab measures, and avoids inputs taken from physical examination such as systolic blood pressure and body mass index and health history data such as smoking, noted Dr. Coresh. He also speculated that “a commercial formula that must be paid for may counterintuitively result in better follow-up for making management changes if it uses some of the resources for education and system changes.”
Using data from multiple sources, like the CKD Prognosis Consortium equation, can create implementation challenges, said Dr. Tangri. “Lab results don’t vary much,” which makes Klinrisk “quite an improvement for implementation. It’s easier to implement.”
Other findings from the newest validation study that Dr. Tangri presented were that the people studied with Klinrisk scores in the top 10% had, over the next 2 years of follow-up and compared with people in the bottom half for Klinrisk staging, a 3- to 5-fold higher rate of all-cause medical costs, a 13-30-fold increase in CKD-related costs, and a 5- to 10-fold increase in hospitalizations and ED visits.
Early identification of CKD and early initiation of intensified treatment for high-risk patients can reduce the rate of progression to dialysis, reduce hospitalizations for heart failure, and lower the cost of care, Dr. Tangri said.
The validation study in 4.6 million Americans was sponsored by Boehringer Ingelheim. Dr. Tangri founded and has an ownership interest in Klinrisk. He has also received honoraria from, has ownership interests in, and has been a consultant to multiple pharmaceutical companies. Dr. Coresh had no disclosures.
PHILADELPHIA – The analyses offer the possibility of focusing intensified medical management of early-stage CKD on those patients who could potentially receive the most benefit.
The Klinrisk model predicts the risk of an adult with early-stage CKD developing either a 40% or greater drop in estimated glomerular filtration rate or kidney failure. It calculates risk based on 20 lab-measured variables that include serum creatinine, urine albumin-to-creatinine ratio, and other values taken from routinely ordered tests such as complete blood cell counts, chemistry panels, comprehensive metabolic panels, and urinalysis.
In the most recent and largest external validation study using data from 4.6 million American adults enrolled in commercial and Medicare insurance plans, the results showed Klinrisk correctly predicted CKD progression in 80%-83% of individuals over 2 years and in 78%-83% of individuals over 5 years, depending on the insurance provider, Navdeep Tangri, MD, PhD, reported at the annual meeting of the American Society of Nephrology. When urinalysis data were available, the model correctly predicted CKD progression in 81%-87% of individuals over 2 years and in 80%-87% of individuals over 5 years. These results follow prior reports of several other successful validations of Klinrisk.
‘Ready to implement’
“The Klinrisk model is ready to implement by any payer, health system, or clinic where the needed lab data are available,” said Dr. Tangri, a nephrologist and professor at the University of Manitoba, Winnipeg, and founder of Klinrisk Inc., the company developing and commercializing the Klinrisk assessment tool.
For the time being, Dr. Tangri sees Klinrisk as a population health device that can allow insurers and health systems to track management quality and quality improvement and to target patients who stand to benefit most from relatively expensive resources. This includes prescriptions for finerenone (Kerendia, Bayer) for people who also have type 2 diabetes, and agents from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors such as dapagliflozin (Farxiga, AstraZeneca) and empagliflozin (Jardiance, Boehringer Ingelheim and Lilly).
He has also begun discussions with the Food and Drug Administration about the data the agency will need to consider Klinrisk for potential approval as a new medical device, perhaps in 2025. That’s how he envisions getting a Klinrisk assessment into the hands of caregivers that they could use with individual patients to create an appropriate treatment plan.
Results from his new analysis showed that “all the kidney disease action is in the 10%-20% of people with the highest risk on Klinrisk, while not much happens in those in the bottom half,” Dr. Tangri said during his presentation.
“We’re trying to find the patients who get the largest [absolute] benefit from intensified treatment,” he added in an interview. “Klinrisk finds people with high-risk kidney disease early on, when kidney function is still normal or near normal. High-risk patients are often completely unrecognized. Risk-based management” that identifies the early-stage CKD patients who would benefit most from treatment with an SGLT2 inhibitor, finerenone, and other foundational treatments to slow CKD progression “is better than the free-for-all that occurs today.”
Simplified data collection
“Klinrisk is very effective,” but requires follow-up by clinicians and health systems to implement its findings, commented Josef Coresh, MD, a professor of clinical epidemiology at Johns Hopkins Bloomberg, Baltimore. Dr. Coresh compared it with a free equation that estimates a person’s risk for a 40% drop in kidney function over the next 3 years developed by Dr. Tangri, Dr. Coresh, and many collaborators led by Morgan C. Grams, MD, PhD, of New York University that they published in 2022, and posted on a website of the CKD Prognosis Consortium.
The CKD Prognosis Consortium formula “takes a different approach” from Klinrisk. The commercial formula “is simpler, only using lab measures, and avoids inputs taken from physical examination such as systolic blood pressure and body mass index and health history data such as smoking, noted Dr. Coresh. He also speculated that “a commercial formula that must be paid for may counterintuitively result in better follow-up for making management changes if it uses some of the resources for education and system changes.”
Using data from multiple sources, like the CKD Prognosis Consortium equation, can create implementation challenges, said Dr. Tangri. “Lab results don’t vary much,” which makes Klinrisk “quite an improvement for implementation. It’s easier to implement.”
Other findings from the newest validation study that Dr. Tangri presented were that the people studied with Klinrisk scores in the top 10% had, over the next 2 years of follow-up and compared with people in the bottom half for Klinrisk staging, a 3- to 5-fold higher rate of all-cause medical costs, a 13-30-fold increase in CKD-related costs, and a 5- to 10-fold increase in hospitalizations and ED visits.
Early identification of CKD and early initiation of intensified treatment for high-risk patients can reduce the rate of progression to dialysis, reduce hospitalizations for heart failure, and lower the cost of care, Dr. Tangri said.
The validation study in 4.6 million Americans was sponsored by Boehringer Ingelheim. Dr. Tangri founded and has an ownership interest in Klinrisk. He has also received honoraria from, has ownership interests in, and has been a consultant to multiple pharmaceutical companies. Dr. Coresh had no disclosures.
PHILADELPHIA – The analyses offer the possibility of focusing intensified medical management of early-stage CKD on those patients who could potentially receive the most benefit.
The Klinrisk model predicts the risk of an adult with early-stage CKD developing either a 40% or greater drop in estimated glomerular filtration rate or kidney failure. It calculates risk based on 20 lab-measured variables that include serum creatinine, urine albumin-to-creatinine ratio, and other values taken from routinely ordered tests such as complete blood cell counts, chemistry panels, comprehensive metabolic panels, and urinalysis.
In the most recent and largest external validation study using data from 4.6 million American adults enrolled in commercial and Medicare insurance plans, the results showed Klinrisk correctly predicted CKD progression in 80%-83% of individuals over 2 years and in 78%-83% of individuals over 5 years, depending on the insurance provider, Navdeep Tangri, MD, PhD, reported at the annual meeting of the American Society of Nephrology. When urinalysis data were available, the model correctly predicted CKD progression in 81%-87% of individuals over 2 years and in 80%-87% of individuals over 5 years. These results follow prior reports of several other successful validations of Klinrisk.
‘Ready to implement’
“The Klinrisk model is ready to implement by any payer, health system, or clinic where the needed lab data are available,” said Dr. Tangri, a nephrologist and professor at the University of Manitoba, Winnipeg, and founder of Klinrisk Inc., the company developing and commercializing the Klinrisk assessment tool.
For the time being, Dr. Tangri sees Klinrisk as a population health device that can allow insurers and health systems to track management quality and quality improvement and to target patients who stand to benefit most from relatively expensive resources. This includes prescriptions for finerenone (Kerendia, Bayer) for people who also have type 2 diabetes, and agents from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors such as dapagliflozin (Farxiga, AstraZeneca) and empagliflozin (Jardiance, Boehringer Ingelheim and Lilly).
He has also begun discussions with the Food and Drug Administration about the data the agency will need to consider Klinrisk for potential approval as a new medical device, perhaps in 2025. That’s how he envisions getting a Klinrisk assessment into the hands of caregivers that they could use with individual patients to create an appropriate treatment plan.
Results from his new analysis showed that “all the kidney disease action is in the 10%-20% of people with the highest risk on Klinrisk, while not much happens in those in the bottom half,” Dr. Tangri said during his presentation.
“We’re trying to find the patients who get the largest [absolute] benefit from intensified treatment,” he added in an interview. “Klinrisk finds people with high-risk kidney disease early on, when kidney function is still normal or near normal. High-risk patients are often completely unrecognized. Risk-based management” that identifies the early-stage CKD patients who would benefit most from treatment with an SGLT2 inhibitor, finerenone, and other foundational treatments to slow CKD progression “is better than the free-for-all that occurs today.”
Simplified data collection
“Klinrisk is very effective,” but requires follow-up by clinicians and health systems to implement its findings, commented Josef Coresh, MD, a professor of clinical epidemiology at Johns Hopkins Bloomberg, Baltimore. Dr. Coresh compared it with a free equation that estimates a person’s risk for a 40% drop in kidney function over the next 3 years developed by Dr. Tangri, Dr. Coresh, and many collaborators led by Morgan C. Grams, MD, PhD, of New York University that they published in 2022, and posted on a website of the CKD Prognosis Consortium.
The CKD Prognosis Consortium formula “takes a different approach” from Klinrisk. The commercial formula “is simpler, only using lab measures, and avoids inputs taken from physical examination such as systolic blood pressure and body mass index and health history data such as smoking, noted Dr. Coresh. He also speculated that “a commercial formula that must be paid for may counterintuitively result in better follow-up for making management changes if it uses some of the resources for education and system changes.”
Using data from multiple sources, like the CKD Prognosis Consortium equation, can create implementation challenges, said Dr. Tangri. “Lab results don’t vary much,” which makes Klinrisk “quite an improvement for implementation. It’s easier to implement.”
Other findings from the newest validation study that Dr. Tangri presented were that the people studied with Klinrisk scores in the top 10% had, over the next 2 years of follow-up and compared with people in the bottom half for Klinrisk staging, a 3- to 5-fold higher rate of all-cause medical costs, a 13-30-fold increase in CKD-related costs, and a 5- to 10-fold increase in hospitalizations and ED visits.
Early identification of CKD and early initiation of intensified treatment for high-risk patients can reduce the rate of progression to dialysis, reduce hospitalizations for heart failure, and lower the cost of care, Dr. Tangri said.
The validation study in 4.6 million Americans was sponsored by Boehringer Ingelheim. Dr. Tangri founded and has an ownership interest in Klinrisk. He has also received honoraria from, has ownership interests in, and has been a consultant to multiple pharmaceutical companies. Dr. Coresh had no disclosures.
AT KIDNEY WEEK 2023
Aprocitentan reduces resistant hypertension in CKD
PHILADELPHIA – (CKD). The results come from a prespecified subgroup analysis of data collected in the drug’s pivotal trial, PRECISION.
The findings provide support for potentially using aprocitentan, if approved for U.S. marketing in 2024, in patients with blood pressure that remains elevated despite treatment with three established antihypertensive drug classes and with stage 3 CKD with an estimated glomerular filtration rate of 30-59 mL/min per 1.73 m2. This is a key group of patients because “chronic kidney disease is the most common comorbidity in patients with resistant hypertension,” said George Bakris, MD, who presented the subgroup analysis at Kidney Week 2023, organized by the American Society of Nephrology.
The CKD subgroup analysis showed “good evidence for safety and evidence in stage 3 CKD,” a subgroup of 141 patients among the total 730 enrolled in PRECISION, said Dr. Bakris. Professor and director of the Comprehensive Hypertension Center at the University of Chicago, he acknowledged that while the results also showed a signal for safety and efficacy in the 21 enrolled patients with stage 4 hypertension, 15-29 mL/min per 1.73m2, this number of stage 4 patients was too small to allow definitive conclusions.
Nephrologist Nishigandha Pradhan, MD, who cochaired the session with this report, agreed. “Resistant hypertension is a particularly intractable problem in patients with CKD, and the risk is greatest with stage 4 CKD. If studies could show that aprocitentan is safe in people with stage 4 CKD, that would be a big plus, but we need more data,” commented Dr. Pradhan in an interview.
Incremental blood pressure reductions
The parallel-group, phase 3 PRECISION trial investigated the safety and short-term antihypertensive effect of aprocitentan in patients with resistant hypertension. The study’s primary efficacy endpoint was blood pressure reduction from baseline in 730 randomized people with persistent systolic hypertension despite treatment with three established antihypertensive agents including a diuretic. The study ran during June 2018–April 2022 at 191 sites in 22 countries.
The primary outcome after 4 weeks on treatment was a least-square mean reduction in office-measured systolic blood pressure, compared with placebo, of 3.8 mm Hg with a 12.5-mg daily oral dose of aprocitentan and 3.7 mm Hg with a 25-mg daily oral dose. Both significant differences were first reported in 2022. Twenty-four–hour ambulatory systolic blood pressures after 4 weeks of treatment fell by an average of 4.2 mm Hg on the lower dose compared with placebo and by an average of 5.9 mm Hg on the higher daily dose, compared with placebo.
Consistent blood pressure reductions occurred in the CKD subgroups. Among people with stage 3 CKD, daytime ambulatory blood pressure at 4 weeks fell by about 10 mm Hg on both the 12.5-mg daily and 25-mg daily doses, compared with placebo.
Among the small number of people with stage 4 CKD, the incremental nighttime systolic blood pressure on aprocitentan, compared with placebo, was even greater, with about a 15–mm Hg incremental reduction on 12.5 mg daily and about a 17–mm Hg incremental reduction on the higher dose.
“This is the first evidence for a change in nocturnal blood pressure in people with stage 4 CKD [and treatment-resistant hypertension], but it was just 21 patients so not yet a big deal,” Dr. Bakris noted.
Increased rates of fluid retention
Although aprocitentan was generally well tolerated, the most common adverse effect was edema or fluid retention, mainly during the first 4 weeks of treatment. In the full PRECISION cohort, this adverse event occurred in 2.1% of people treated with placebo, 9.1% of those on the 12.5-mg daily dose, and in 18.4% of those on the higher dose during the initial 4-week phase of treatment.
Among all stage 3 and 4 CKD patients on aprocitentan, edema or fluid retention occurred in 21% during the first 4 weeks, and in 27% during an additional 32 weeks of treatment with 25 mg aprocitentan daily. A majority of these patients started a diuretic to address their excess fluid, with only two discontinuing aprocitentan treatment.
“Fluid retention is an issue with aprocitentan,” Dr. Bakris acknowledged. But he also highlighted than only 6 of the 162 patients with CKD required hospitalization for heart failure during the study, and one of these cases had placebo treatment. Among the five with acute heart failure while on aprocitentan, none had to stop their treatment, and two had a clear prior history of heart failure.
The companies developing aprocitentan, Janssen and Idorsia, used the PRECISION results as the centerpiece in filing for a new drug approval to the FDA, with a March 2024 goal for the FDA‘s decision. Dr. Bakris called the application “a solid case for approval.” But he added that approval will likely require that all treatment candidates first undergo testing of their heart function or fluid volume, such as a measure of their blood level of N-terminal pro-B-type natriuretic peptide, with treatment withheld when the level is too high.
The upside of aprocitentan compared with current drug options for treating resistant hypertension is that it has not appeared to cause any increase in blood potassium levels, which is an issue with the current top agent for resistant hypertension, spironolactone.
“The problem with spironolactone is the risk for hyperkalemia, which keeps us looking for something with lower risk,” commented Dr. Pradhan, a nephrologist with University Hospitals in Cleveland. Hyperkalemia is an even greater risk for people with CKD. Although the PRECISION trial identified the issue of fluid retention with aprocitentan, titrating an effective dose of a loop diuretic for treated patients may effectively blunt the edema risk, Dr. Pradhan said.
Endothelin has a potent vasoconstrictive effect and is “implicated in the pathogenesis of hypertension,” Dr. Bakris explained. Aprocitentan antagonizes both the endothelin A and B receptors. The subgroup analyses also showed that in people with CKD, treatment with aprocitentan led to roughly a halving of the baseline level of urine albumin-to-creatinine ratio, a small and stable decrease in estimated glomerular filtration rate, and a modest and stable increase in blood levels of N-terminal pro-B-type natriuretic hormone.
The PRECISION trial was sponsored by Janssen Pharmaceuticals and Idorsia Pharmaceuticals, the companies jointly developing aprocitentan. Dr. Bakris has been a consultant to Janssen, and also a consultant to or honoraria recipient of Alnylam, AstraZeneca, Bayer, Dia Medica Therapeutics, Ionis, inREGEN, KBP Biosciences, Merck, Novo Nordisk, and Quantum Genomics. Dr. Pradhan had no disclosures.
PHILADELPHIA – (CKD). The results come from a prespecified subgroup analysis of data collected in the drug’s pivotal trial, PRECISION.
The findings provide support for potentially using aprocitentan, if approved for U.S. marketing in 2024, in patients with blood pressure that remains elevated despite treatment with three established antihypertensive drug classes and with stage 3 CKD with an estimated glomerular filtration rate of 30-59 mL/min per 1.73 m2. This is a key group of patients because “chronic kidney disease is the most common comorbidity in patients with resistant hypertension,” said George Bakris, MD, who presented the subgroup analysis at Kidney Week 2023, organized by the American Society of Nephrology.
The CKD subgroup analysis showed “good evidence for safety and evidence in stage 3 CKD,” a subgroup of 141 patients among the total 730 enrolled in PRECISION, said Dr. Bakris. Professor and director of the Comprehensive Hypertension Center at the University of Chicago, he acknowledged that while the results also showed a signal for safety and efficacy in the 21 enrolled patients with stage 4 hypertension, 15-29 mL/min per 1.73m2, this number of stage 4 patients was too small to allow definitive conclusions.
Nephrologist Nishigandha Pradhan, MD, who cochaired the session with this report, agreed. “Resistant hypertension is a particularly intractable problem in patients with CKD, and the risk is greatest with stage 4 CKD. If studies could show that aprocitentan is safe in people with stage 4 CKD, that would be a big plus, but we need more data,” commented Dr. Pradhan in an interview.
Incremental blood pressure reductions
The parallel-group, phase 3 PRECISION trial investigated the safety and short-term antihypertensive effect of aprocitentan in patients with resistant hypertension. The study’s primary efficacy endpoint was blood pressure reduction from baseline in 730 randomized people with persistent systolic hypertension despite treatment with three established antihypertensive agents including a diuretic. The study ran during June 2018–April 2022 at 191 sites in 22 countries.
The primary outcome after 4 weeks on treatment was a least-square mean reduction in office-measured systolic blood pressure, compared with placebo, of 3.8 mm Hg with a 12.5-mg daily oral dose of aprocitentan and 3.7 mm Hg with a 25-mg daily oral dose. Both significant differences were first reported in 2022. Twenty-four–hour ambulatory systolic blood pressures after 4 weeks of treatment fell by an average of 4.2 mm Hg on the lower dose compared with placebo and by an average of 5.9 mm Hg on the higher daily dose, compared with placebo.
Consistent blood pressure reductions occurred in the CKD subgroups. Among people with stage 3 CKD, daytime ambulatory blood pressure at 4 weeks fell by about 10 mm Hg on both the 12.5-mg daily and 25-mg daily doses, compared with placebo.
Among the small number of people with stage 4 CKD, the incremental nighttime systolic blood pressure on aprocitentan, compared with placebo, was even greater, with about a 15–mm Hg incremental reduction on 12.5 mg daily and about a 17–mm Hg incremental reduction on the higher dose.
“This is the first evidence for a change in nocturnal blood pressure in people with stage 4 CKD [and treatment-resistant hypertension], but it was just 21 patients so not yet a big deal,” Dr. Bakris noted.
Increased rates of fluid retention
Although aprocitentan was generally well tolerated, the most common adverse effect was edema or fluid retention, mainly during the first 4 weeks of treatment. In the full PRECISION cohort, this adverse event occurred in 2.1% of people treated with placebo, 9.1% of those on the 12.5-mg daily dose, and in 18.4% of those on the higher dose during the initial 4-week phase of treatment.
Among all stage 3 and 4 CKD patients on aprocitentan, edema or fluid retention occurred in 21% during the first 4 weeks, and in 27% during an additional 32 weeks of treatment with 25 mg aprocitentan daily. A majority of these patients started a diuretic to address their excess fluid, with only two discontinuing aprocitentan treatment.
“Fluid retention is an issue with aprocitentan,” Dr. Bakris acknowledged. But he also highlighted than only 6 of the 162 patients with CKD required hospitalization for heart failure during the study, and one of these cases had placebo treatment. Among the five with acute heart failure while on aprocitentan, none had to stop their treatment, and two had a clear prior history of heart failure.
The companies developing aprocitentan, Janssen and Idorsia, used the PRECISION results as the centerpiece in filing for a new drug approval to the FDA, with a March 2024 goal for the FDA‘s decision. Dr. Bakris called the application “a solid case for approval.” But he added that approval will likely require that all treatment candidates first undergo testing of their heart function or fluid volume, such as a measure of their blood level of N-terminal pro-B-type natriuretic peptide, with treatment withheld when the level is too high.
The upside of aprocitentan compared with current drug options for treating resistant hypertension is that it has not appeared to cause any increase in blood potassium levels, which is an issue with the current top agent for resistant hypertension, spironolactone.
“The problem with spironolactone is the risk for hyperkalemia, which keeps us looking for something with lower risk,” commented Dr. Pradhan, a nephrologist with University Hospitals in Cleveland. Hyperkalemia is an even greater risk for people with CKD. Although the PRECISION trial identified the issue of fluid retention with aprocitentan, titrating an effective dose of a loop diuretic for treated patients may effectively blunt the edema risk, Dr. Pradhan said.
Endothelin has a potent vasoconstrictive effect and is “implicated in the pathogenesis of hypertension,” Dr. Bakris explained. Aprocitentan antagonizes both the endothelin A and B receptors. The subgroup analyses also showed that in people with CKD, treatment with aprocitentan led to roughly a halving of the baseline level of urine albumin-to-creatinine ratio, a small and stable decrease in estimated glomerular filtration rate, and a modest and stable increase in blood levels of N-terminal pro-B-type natriuretic hormone.
The PRECISION trial was sponsored by Janssen Pharmaceuticals and Idorsia Pharmaceuticals, the companies jointly developing aprocitentan. Dr. Bakris has been a consultant to Janssen, and also a consultant to or honoraria recipient of Alnylam, AstraZeneca, Bayer, Dia Medica Therapeutics, Ionis, inREGEN, KBP Biosciences, Merck, Novo Nordisk, and Quantum Genomics. Dr. Pradhan had no disclosures.
PHILADELPHIA – (CKD). The results come from a prespecified subgroup analysis of data collected in the drug’s pivotal trial, PRECISION.
The findings provide support for potentially using aprocitentan, if approved for U.S. marketing in 2024, in patients with blood pressure that remains elevated despite treatment with three established antihypertensive drug classes and with stage 3 CKD with an estimated glomerular filtration rate of 30-59 mL/min per 1.73 m2. This is a key group of patients because “chronic kidney disease is the most common comorbidity in patients with resistant hypertension,” said George Bakris, MD, who presented the subgroup analysis at Kidney Week 2023, organized by the American Society of Nephrology.
The CKD subgroup analysis showed “good evidence for safety and evidence in stage 3 CKD,” a subgroup of 141 patients among the total 730 enrolled in PRECISION, said Dr. Bakris. Professor and director of the Comprehensive Hypertension Center at the University of Chicago, he acknowledged that while the results also showed a signal for safety and efficacy in the 21 enrolled patients with stage 4 hypertension, 15-29 mL/min per 1.73m2, this number of stage 4 patients was too small to allow definitive conclusions.
Nephrologist Nishigandha Pradhan, MD, who cochaired the session with this report, agreed. “Resistant hypertension is a particularly intractable problem in patients with CKD, and the risk is greatest with stage 4 CKD. If studies could show that aprocitentan is safe in people with stage 4 CKD, that would be a big plus, but we need more data,” commented Dr. Pradhan in an interview.
Incremental blood pressure reductions
The parallel-group, phase 3 PRECISION trial investigated the safety and short-term antihypertensive effect of aprocitentan in patients with resistant hypertension. The study’s primary efficacy endpoint was blood pressure reduction from baseline in 730 randomized people with persistent systolic hypertension despite treatment with three established antihypertensive agents including a diuretic. The study ran during June 2018–April 2022 at 191 sites in 22 countries.
The primary outcome after 4 weeks on treatment was a least-square mean reduction in office-measured systolic blood pressure, compared with placebo, of 3.8 mm Hg with a 12.5-mg daily oral dose of aprocitentan and 3.7 mm Hg with a 25-mg daily oral dose. Both significant differences were first reported in 2022. Twenty-four–hour ambulatory systolic blood pressures after 4 weeks of treatment fell by an average of 4.2 mm Hg on the lower dose compared with placebo and by an average of 5.9 mm Hg on the higher daily dose, compared with placebo.
Consistent blood pressure reductions occurred in the CKD subgroups. Among people with stage 3 CKD, daytime ambulatory blood pressure at 4 weeks fell by about 10 mm Hg on both the 12.5-mg daily and 25-mg daily doses, compared with placebo.
Among the small number of people with stage 4 CKD, the incremental nighttime systolic blood pressure on aprocitentan, compared with placebo, was even greater, with about a 15–mm Hg incremental reduction on 12.5 mg daily and about a 17–mm Hg incremental reduction on the higher dose.
“This is the first evidence for a change in nocturnal blood pressure in people with stage 4 CKD [and treatment-resistant hypertension], but it was just 21 patients so not yet a big deal,” Dr. Bakris noted.
Increased rates of fluid retention
Although aprocitentan was generally well tolerated, the most common adverse effect was edema or fluid retention, mainly during the first 4 weeks of treatment. In the full PRECISION cohort, this adverse event occurred in 2.1% of people treated with placebo, 9.1% of those on the 12.5-mg daily dose, and in 18.4% of those on the higher dose during the initial 4-week phase of treatment.
Among all stage 3 and 4 CKD patients on aprocitentan, edema or fluid retention occurred in 21% during the first 4 weeks, and in 27% during an additional 32 weeks of treatment with 25 mg aprocitentan daily. A majority of these patients started a diuretic to address their excess fluid, with only two discontinuing aprocitentan treatment.
“Fluid retention is an issue with aprocitentan,” Dr. Bakris acknowledged. But he also highlighted than only 6 of the 162 patients with CKD required hospitalization for heart failure during the study, and one of these cases had placebo treatment. Among the five with acute heart failure while on aprocitentan, none had to stop their treatment, and two had a clear prior history of heart failure.
The companies developing aprocitentan, Janssen and Idorsia, used the PRECISION results as the centerpiece in filing for a new drug approval to the FDA, with a March 2024 goal for the FDA‘s decision. Dr. Bakris called the application “a solid case for approval.” But he added that approval will likely require that all treatment candidates first undergo testing of their heart function or fluid volume, such as a measure of their blood level of N-terminal pro-B-type natriuretic peptide, with treatment withheld when the level is too high.
The upside of aprocitentan compared with current drug options for treating resistant hypertension is that it has not appeared to cause any increase in blood potassium levels, which is an issue with the current top agent for resistant hypertension, spironolactone.
“The problem with spironolactone is the risk for hyperkalemia, which keeps us looking for something with lower risk,” commented Dr. Pradhan, a nephrologist with University Hospitals in Cleveland. Hyperkalemia is an even greater risk for people with CKD. Although the PRECISION trial identified the issue of fluid retention with aprocitentan, titrating an effective dose of a loop diuretic for treated patients may effectively blunt the edema risk, Dr. Pradhan said.
Endothelin has a potent vasoconstrictive effect and is “implicated in the pathogenesis of hypertension,” Dr. Bakris explained. Aprocitentan antagonizes both the endothelin A and B receptors. The subgroup analyses also showed that in people with CKD, treatment with aprocitentan led to roughly a halving of the baseline level of urine albumin-to-creatinine ratio, a small and stable decrease in estimated glomerular filtration rate, and a modest and stable increase in blood levels of N-terminal pro-B-type natriuretic hormone.
The PRECISION trial was sponsored by Janssen Pharmaceuticals and Idorsia Pharmaceuticals, the companies jointly developing aprocitentan. Dr. Bakris has been a consultant to Janssen, and also a consultant to or honoraria recipient of Alnylam, AstraZeneca, Bayer, Dia Medica Therapeutics, Ionis, inREGEN, KBP Biosciences, Merck, Novo Nordisk, and Quantum Genomics. Dr. Pradhan had no disclosures.
AT KIDNEY WEEK 2023