User login
VIDEO: Bioimpedance provides accurate assessment of Mohs surgical margins
SAN DIEGO – In assessing tumor-free margins during Mohs micrographic surgery for skin cancer, of histologic sections, in a single-center, pilot study of bioimpedance in 151 specimens from 50 consecutive patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
If the finding of high diagnostic accuracy using bioimpedance spectroscopy is confirmed in larger numbers of patients and specimens run at multiple sites, this approach could “potentially revolutionize what happens with the way Mohs sections are processed in the future” by potentially shaving many minutes off the duration of a standard procedure, Darrell S. Rigel, MD, said in a video interview during the annual meeting of the American Academy of Dermatology.
Usually, it takes 10-20 minutes to process and examine Mohs specimens at each stage of the surgical procedure to determine whether additional excision must remove residual cancer cells, said Dr. Rigel, a dermatologist at New York University. In contrast, assessment for residual cancer cells in the surgical field takes less than a minute using bioimpedance spectroscopy, which relies on differences in electrical conductivity between benign and cancerous cells to identify cancer cells remaining at the surgical margins.
The results of the study were presented in a poster at the meeting, by a research associate of Dr. Rigel’s, Ryan Svoboda, MD, of the National Society for Cutaneous Medicine, New York.
The researchers used a bioimpedance spectroscopy device made by NovaScan to assess 151 histology slides prepared during Mohs micrographic surgery on patients with nonmelanoma skin cancer, and compared the findings against the gold standard of histological slide examination. By this criterion, bioimpedance spectroscopy identified 105 true negatives and 2 false negatives, and 43 true positives and 1 false positive. Calculations showed that this equated to 95.6% sensitivity, 99.1% specificity, a 97.7% positive predictive value, and a 98.1% negative predictive value.
These may be underestimates of the accuracy of bioimpedance spectroscopy because the calculations presume that conventional histology is always correct, but Dr. Rigel noted that sometimes the histological diagnosis is wrong.
SOURCE: Svoboda R et al. Poster 7304.
SAN DIEGO – In assessing tumor-free margins during Mohs micrographic surgery for skin cancer, of histologic sections, in a single-center, pilot study of bioimpedance in 151 specimens from 50 consecutive patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
If the finding of high diagnostic accuracy using bioimpedance spectroscopy is confirmed in larger numbers of patients and specimens run at multiple sites, this approach could “potentially revolutionize what happens with the way Mohs sections are processed in the future” by potentially shaving many minutes off the duration of a standard procedure, Darrell S. Rigel, MD, said in a video interview during the annual meeting of the American Academy of Dermatology.
Usually, it takes 10-20 minutes to process and examine Mohs specimens at each stage of the surgical procedure to determine whether additional excision must remove residual cancer cells, said Dr. Rigel, a dermatologist at New York University. In contrast, assessment for residual cancer cells in the surgical field takes less than a minute using bioimpedance spectroscopy, which relies on differences in electrical conductivity between benign and cancerous cells to identify cancer cells remaining at the surgical margins.
The results of the study were presented in a poster at the meeting, by a research associate of Dr. Rigel’s, Ryan Svoboda, MD, of the National Society for Cutaneous Medicine, New York.
The researchers used a bioimpedance spectroscopy device made by NovaScan to assess 151 histology slides prepared during Mohs micrographic surgery on patients with nonmelanoma skin cancer, and compared the findings against the gold standard of histological slide examination. By this criterion, bioimpedance spectroscopy identified 105 true negatives and 2 false negatives, and 43 true positives and 1 false positive. Calculations showed that this equated to 95.6% sensitivity, 99.1% specificity, a 97.7% positive predictive value, and a 98.1% negative predictive value.
These may be underestimates of the accuracy of bioimpedance spectroscopy because the calculations presume that conventional histology is always correct, but Dr. Rigel noted that sometimes the histological diagnosis is wrong.
SOURCE: Svoboda R et al. Poster 7304.
SAN DIEGO – In assessing tumor-free margins during Mohs micrographic surgery for skin cancer, of histologic sections, in a single-center, pilot study of bioimpedance in 151 specimens from 50 consecutive patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
If the finding of high diagnostic accuracy using bioimpedance spectroscopy is confirmed in larger numbers of patients and specimens run at multiple sites, this approach could “potentially revolutionize what happens with the way Mohs sections are processed in the future” by potentially shaving many minutes off the duration of a standard procedure, Darrell S. Rigel, MD, said in a video interview during the annual meeting of the American Academy of Dermatology.
Usually, it takes 10-20 minutes to process and examine Mohs specimens at each stage of the surgical procedure to determine whether additional excision must remove residual cancer cells, said Dr. Rigel, a dermatologist at New York University. In contrast, assessment for residual cancer cells in the surgical field takes less than a minute using bioimpedance spectroscopy, which relies on differences in electrical conductivity between benign and cancerous cells to identify cancer cells remaining at the surgical margins.
The results of the study were presented in a poster at the meeting, by a research associate of Dr. Rigel’s, Ryan Svoboda, MD, of the National Society for Cutaneous Medicine, New York.
The researchers used a bioimpedance spectroscopy device made by NovaScan to assess 151 histology slides prepared during Mohs micrographic surgery on patients with nonmelanoma skin cancer, and compared the findings against the gold standard of histological slide examination. By this criterion, bioimpedance spectroscopy identified 105 true negatives and 2 false negatives, and 43 true positives and 1 false positive. Calculations showed that this equated to 95.6% sensitivity, 99.1% specificity, a 97.7% positive predictive value, and a 98.1% negative predictive value.
These may be underestimates of the accuracy of bioimpedance spectroscopy because the calculations presume that conventional histology is always correct, but Dr. Rigel noted that sometimes the histological diagnosis is wrong.
SOURCE: Svoboda R et al. Poster 7304.
REPORTING FROM AAD 18
Key clinical point: Bioimpedance spectroscopy showed excellent diagnostic accuracy for cancer cells on Mohs surgical margins.
Major finding: Bioimpedance spectroscopy had a sensitivity of 95.6% and specificity of 99.1% compared with Mohs histology.
Study details: A single-center pilot study with 151 Mohs surgical specimens taken from 50 patients.
Disclosures: The study was funded by NovaScan, the company developing the device tested in the study. Dr. Rigel has been a consultant to NovaScan and to Castle Biosciences, DermTech, Ferndale, Myriad, and Neutrogena, and has received research support from Castle and Neutrogena.
Source: Svoboda R et al. Poster 7304.
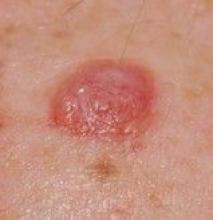
Evaluating Dermatology Apps for Patient Education
Topical imiquimod helps clear blurred lines in lentigo maligna excision
according to a study from the University of Utah.
Lentigo maligna is a subtype of melanoma in situ, usually occurring in the head and neck regions, the researchers said.
“Neoadjuvant topical imiquimod 5% cream applied 5 times weekly for 8 weeks was associated with decreased MDCs in LM treatment sites compared with the MDCs of negative control sites,” wrote Shadai Flores of the University of Utah, Salt Lake City, and her colleagues.
Previously, the ability to distinguish between the surgical border and surrounding background melanocytic hyperplasia was uncertain. Because of this uncertainty, LM removal required an average margin of 7.2 mm. Another study showed that topical imiquimod 5% cream enabled the removal of most LM tumors with 2-mm margins. This study “sought to evaluate MDCs in imiquimod-treated LM and negative control biopsy specimens to determine if there was a measurable difference in melanocyte density,” the researchers wrote in a research letter published in JAMA Dermatology.
The study prospectively followed 52 cases of LM treated with imiquimod 5% topical cream 5 days per week for 8 weeks followed by conservative staged excisions with 2-mm margins. Treatment with imiquimod 5% of LM was followed by a 2- to 4-month recuperation period before surgery could be performed. All patients in the study were treated by one Mohs surgeon at the Huntsman Cancer Institute at the university.
To establish an MDC baseline, a 10-mm long fusiform biopsy was taken as a negative control. The negative control sample site and the LM site were separated by approximately 6 cm, found on the same side of the body, and showed similar color changes. After a negative control was taken, an LM lesion was resected and subsequently quadrisected. The MDCs then were concurrently counted by the researchers and compared with the negative controls.
Of the 52 LM specimens, 44 (85%) exhibited decreases in MDCs, compared with the negative controls. The median MDC from post–imiquimod-treated sites was 14.4, with a range of 0.5-26.6. This showed marked improvement over the negative controls, which had a median MDC of 20.0 (range of 9.0-36.7). A 2-tailed paired t test revealed that the results displayed statistical significance (P less than .001). Residual LM was seen in the central areas of 9 (17%) specimens, but 43 (83%) had no indication of residual LM.
“The decreased melanocytic hyperplasia in imiquimod-treated sites reduced ambiguity in making a distinction between the border of the excised LM and background melanocytic hyperplasia,” noted Ms. Flores and her colleagues.
The authors had no conflicts of interest.
SOURCE: Flores S et al. JAMA Dermatol. 2018 Feb 16. doi: 10.1001/jamadermatol.2017.5632.
according to a study from the University of Utah.
Lentigo maligna is a subtype of melanoma in situ, usually occurring in the head and neck regions, the researchers said.
“Neoadjuvant topical imiquimod 5% cream applied 5 times weekly for 8 weeks was associated with decreased MDCs in LM treatment sites compared with the MDCs of negative control sites,” wrote Shadai Flores of the University of Utah, Salt Lake City, and her colleagues.
Previously, the ability to distinguish between the surgical border and surrounding background melanocytic hyperplasia was uncertain. Because of this uncertainty, LM removal required an average margin of 7.2 mm. Another study showed that topical imiquimod 5% cream enabled the removal of most LM tumors with 2-mm margins. This study “sought to evaluate MDCs in imiquimod-treated LM and negative control biopsy specimens to determine if there was a measurable difference in melanocyte density,” the researchers wrote in a research letter published in JAMA Dermatology.
The study prospectively followed 52 cases of LM treated with imiquimod 5% topical cream 5 days per week for 8 weeks followed by conservative staged excisions with 2-mm margins. Treatment with imiquimod 5% of LM was followed by a 2- to 4-month recuperation period before surgery could be performed. All patients in the study were treated by one Mohs surgeon at the Huntsman Cancer Institute at the university.
To establish an MDC baseline, a 10-mm long fusiform biopsy was taken as a negative control. The negative control sample site and the LM site were separated by approximately 6 cm, found on the same side of the body, and showed similar color changes. After a negative control was taken, an LM lesion was resected and subsequently quadrisected. The MDCs then were concurrently counted by the researchers and compared with the negative controls.
Of the 52 LM specimens, 44 (85%) exhibited decreases in MDCs, compared with the negative controls. The median MDC from post–imiquimod-treated sites was 14.4, with a range of 0.5-26.6. This showed marked improvement over the negative controls, which had a median MDC of 20.0 (range of 9.0-36.7). A 2-tailed paired t test revealed that the results displayed statistical significance (P less than .001). Residual LM was seen in the central areas of 9 (17%) specimens, but 43 (83%) had no indication of residual LM.
“The decreased melanocytic hyperplasia in imiquimod-treated sites reduced ambiguity in making a distinction between the border of the excised LM and background melanocytic hyperplasia,” noted Ms. Flores and her colleagues.
The authors had no conflicts of interest.
SOURCE: Flores S et al. JAMA Dermatol. 2018 Feb 16. doi: 10.1001/jamadermatol.2017.5632.
according to a study from the University of Utah.
Lentigo maligna is a subtype of melanoma in situ, usually occurring in the head and neck regions, the researchers said.
“Neoadjuvant topical imiquimod 5% cream applied 5 times weekly for 8 weeks was associated with decreased MDCs in LM treatment sites compared with the MDCs of negative control sites,” wrote Shadai Flores of the University of Utah, Salt Lake City, and her colleagues.
Previously, the ability to distinguish between the surgical border and surrounding background melanocytic hyperplasia was uncertain. Because of this uncertainty, LM removal required an average margin of 7.2 mm. Another study showed that topical imiquimod 5% cream enabled the removal of most LM tumors with 2-mm margins. This study “sought to evaluate MDCs in imiquimod-treated LM and negative control biopsy specimens to determine if there was a measurable difference in melanocyte density,” the researchers wrote in a research letter published in JAMA Dermatology.
The study prospectively followed 52 cases of LM treated with imiquimod 5% topical cream 5 days per week for 8 weeks followed by conservative staged excisions with 2-mm margins. Treatment with imiquimod 5% of LM was followed by a 2- to 4-month recuperation period before surgery could be performed. All patients in the study were treated by one Mohs surgeon at the Huntsman Cancer Institute at the university.
To establish an MDC baseline, a 10-mm long fusiform biopsy was taken as a negative control. The negative control sample site and the LM site were separated by approximately 6 cm, found on the same side of the body, and showed similar color changes. After a negative control was taken, an LM lesion was resected and subsequently quadrisected. The MDCs then were concurrently counted by the researchers and compared with the negative controls.
Of the 52 LM specimens, 44 (85%) exhibited decreases in MDCs, compared with the negative controls. The median MDC from post–imiquimod-treated sites was 14.4, with a range of 0.5-26.6. This showed marked improvement over the negative controls, which had a median MDC of 20.0 (range of 9.0-36.7). A 2-tailed paired t test revealed that the results displayed statistical significance (P less than .001). Residual LM was seen in the central areas of 9 (17%) specimens, but 43 (83%) had no indication of residual LM.
“The decreased melanocytic hyperplasia in imiquimod-treated sites reduced ambiguity in making a distinction between the border of the excised LM and background melanocytic hyperplasia,” noted Ms. Flores and her colleagues.
The authors had no conflicts of interest.
SOURCE: Flores S et al. JAMA Dermatol. 2018 Feb 16. doi: 10.1001/jamadermatol.2017.5632.
FROM JAMA DERMATOLOGY
Key clinical point: Neoadjuvant, topical imiquimod 5% cream is associated with a decrease in melanocyte density counts (MDC).
Major finding: Of 52 patient specimens, 44 (85%) exhibited decreases in MDCs, compared with the negative controls.
Study details: A prospective study of 52 cases of lentigo maligna treated with imiquimod 5% topical cream 5 days per week for 8 weeks, followed by conservative staged excisions with 2-mm margins.
Disclosures: The authors had no conflicts of interest.
Source: Flores S et al. JAMA Dermatol. 2018 Feb 16. doi: 10.1001/jamadermatol.2017.5632.
Concurrent ipilimumab and CMV colitis refractory to oral steroids
Immune checkpoint inhibitors, including anti-cytotoxic T-lymphocyte antigen 4 (anti-CTLA4) and anti-programmed cell death protein-1 (anti-PD-1) antibodies, have demonstrated clinical and survival benefits in a variety of malignancies, which has led to an expansion in their role in oncology. In melanoma, the anti-CTLA-4 antibody, ipilimumab, has demonstrated a survival benefit in patients with advanced metastatic melanoma and in patients with resectable disease with lymph node involvement.1,2
Ipilimumab exerts its effect by binding CTLA-4 on conventional and regulatory T cells, thus blocking inhibitory signals on T cells, which leads to an antitumor response.3 The increased immune response counteracts the immune-evading mechanisms of the tumor. With increased use of these agents, immune-related adverse events (irAEs) have become more prevalent. The most common irAEs secondary to ipilimumab are skin rash, colitis/diarrhea, hepatitis, pneumonitis, and various endocrinopathies.4 In a phase 3 trial of adjuvant ipilimumab in patients with resected stage III melanoma, grade 3 or 4 adverse events occurred in 54.1% of participants in the ipilimumab arm, the most common being diarrhea and colitis (9.8% and 6.8%, respectively).2Recognition and management of irAEs has led to the implementation of treatment guidelines.4,5 Management of irAEs includes checkpoint inhibitor discontinuation and reversal of the immune response by institution of immunosuppression with corticosteroids.
Case presentation and summary
A 40-year-old white woman with stage IIIB BRAF V600E-positive melanoma presented with diarrhea refractory to high-dose prednisone (1 mg/kg BID). She had recently undergone wide local excision and sentinel node biopsy and received her inaugural dose of ipilimumab (10 mg/kg).
The patient first presented with loose, watery stools that had begun 8 days after she had received her first dose of adjuvant ipilimumab. She was admitted to the hospital, and intravenous methylprednisolone was initiated along with empiric ciprofloxacin (400 mg, IVPB Q12h) and metronidazole (500 mg, IVPB Q8h) as infectious causes were concurrently ruled out. During this initial admission, the patient’s stool was negative for Clostridium difficile toxin, ova, and parasites, as well as enteric pathogens by culture. After infectious causes were excluded, she was diagnosed with ipilimumab-induced colitis. Antibiotics were discontinued, and the patient ultimately noted improvement in her symptoms. On hospital day 7, she was experiencing only 2 bowel movements a day and was discharged on 80 mg of prednisone twice daily.
After discharge the patient noted persistence of her symptoms. At her follow-up, 9 days after discharge, the patient noted continued symptoms of low-grade diarrhea. She failed a trial of steroid tapering due to exacerbation of her abdominal pain and frequency of diarrhea. Further investigation was negative for C. diff toxin and a computed-tomography scan was consistent with continuing colitis. The patient’s symptoms continued to worsen, with recurrence of grade 3 diarrhea, and she was ultimately readmitted 17 days after her earlier discharge (36 days after her first ipilimumab dosing).
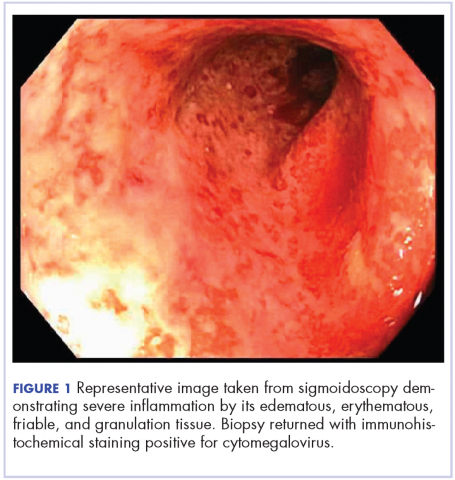
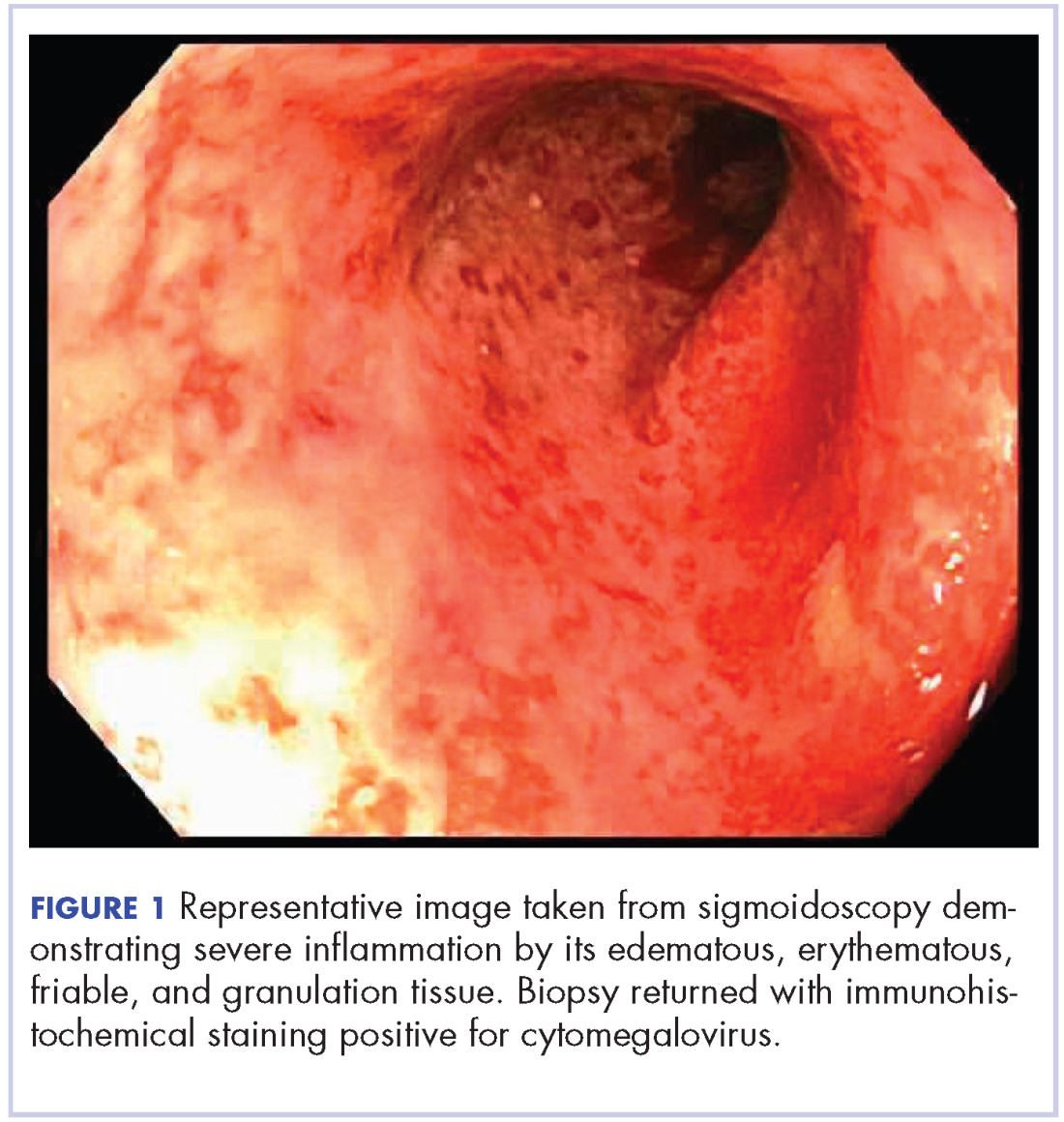
On re-admission, the patient was again given intravenous methylprednisolone and experienced interval improvement in the frequency of diarrhea. A gastroenterology expert was consulted, and the patient underwent a flexible sigmoidoscopy that demonstrated findings of diffuse and severe inflammation and biopsies were obtained (Figure 1). After several days of continued symptoms, the patient received infliximab 5 mg/kg for treatment of her adverse autoimmune reaction. After administration, the patient noted improvement in the frequency and volume of diarrhea, however, her symptoms still persisted.
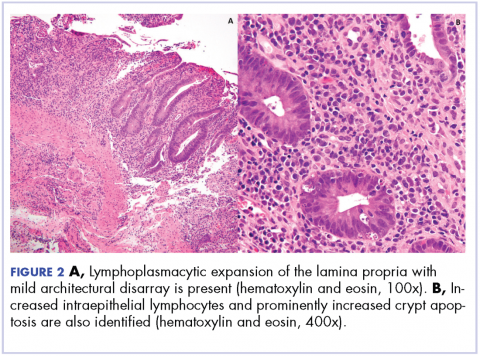
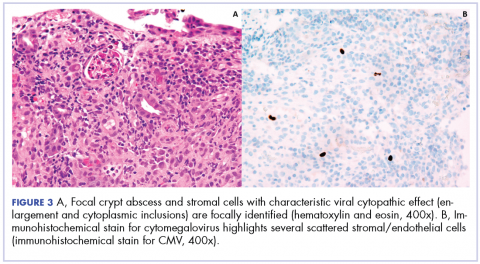
Biopsy results subsequently revealed findings compatible with ipilimumab-induced colitis, and immunohistochemical staining demonstrated positivity for cytomegalovirus (CMV). Specifically, histologic examination showed lymphoplasmacytic expansion of the lamina propria, some architectural distortion, and increased crypt apoptosis. Scattered cryptitis and crypt abscesses were also noted, as were rare stromal and endothelial cells with characteristic CMV inclusions (Figure 2 and Figure 3).
Serum CMV polymerase chain reaction (PCR) was also positive at 652,000 IU/mL (lower limit of detection 100 IU/mL). Induction dosing of ganciclovir (5 mg/kg IV Q12h) was initiated. The combined treatment with intravenous methylprednisone and ganciclovir led to an improvement in diarrhea frequency and resolution of blood in the stool. She was transitioned to oral prednisone, but it resulted in redevelopment of grade 3 diarrhea. The patient was therefore resumed on and discharged on daily intravenous methylprednisolone.
After discharge, the patient was started on budesonide 9 mg daily. Her serum CMV PCR level reduced and she was transitioned to oral valgancyclovir (900 mg daily) for maintenance. Another unsuccessful attempt was made to switch her to oral prednisone.
About 14 weeks after the initial ipilimumab dosing, the patient underwent another flexible sigmoidoscopy that again demonstrated severe colitis from the rectum to sigmoid colon. Biopsies were negative for CMV. Patient was readmitted for recurrence of diarrhea the following week. Treatment with IV methylprednisone (1mg/kg BID) and infliximab (5 mg/kg) again led to an improvement of symptoms. She was again discharged on IV methylprednisone (1 mg/kg BID) with a taper.
In the 15th week after her initial ipilimumab dose, the patient presented with a perforated bowel, requiring a subtotal colectomy and end ileostomy. She continued on a slow taper of oral prednisone (50 mg daily and decrease by 10 mg every 5 days).
At her last documented follow-up, 8 months after her first ipilimumab dose, she was having normal output from her ileostomy. She developed secondary adrenal insufficiency because of the long-term steroids and continued to take prednisone 5 mg daily.
Discussion
Diarrhea and colitis are common irAEs attributable to checkpoint-inhibitor therapy used for the treatment of melanoma. This case of ipilimumab-induced colitis refractory to high-dose oral steroids demonstrates the risks associated with management of anti-CTLA-4 induced colitis. In particular, the high-dose corticosteroids required to treat the autoimmune component of this patient’s colitis increased her susceptibility to CMV reactivation.
The diagnosis of colitis secondary to ipilimumab is made primarily in the appropriate clinical setting, and typically onsets during the induction period (within 12 weeks of initial dosing) and most resolve within 6-8 weeks.6 Histopathologically, there is lymphoplasmacytic expansion of lamina propria, increased intraepithelial lymphocytes, and increased epithelial apoptosis of crypts. One can also see acute cryptitis and crypt abscesses. Reactive epithelial changes with mucin depletion are also often seen in epithelial cells.
Findings from immunohistochemical studies have shown the increased intraepithelial lymphocytes to be predominantly CD8-positive T cells, while the lamina propria contains an increase in the mixture of CD4- and CD8-positive T cells. In addition, small intestinal samples show villous blunting. There is an absence of significant architectural distortion and well-developed basal lymphoplasmacytic infiltrates characteristic of chronic mucosal injury, such as idiopathic inflammatory bowel disease.7 Granulomas are also absent in most series, though they have been reported in some cases.8 The features are similar to those seen in autoimmune enteropathy, but goblet and endocrine cells remain preserved. Graft-versus-host disease has similar histologic features, however, the clinical setting usually makes the distinction between these obvious.
Current treatment algorithms for ipilimumab-related diarrhea, begin with immediate treatment with intravenous methylprednisolone (125 mg once). This is followed with oral prednisone at a dose of 1-2 mg/kg tapered over 4 to 8 weeks.4 In patients with persistent symptoms despite adequate doses of corticosteroids, infliximab (5 mg/kg every 2 weeks) is recommended until the resolution of symptoms, and a longer taper of prednisone is often necessary.
Institution of high-dose corticosteroids to treat grade 3 or 4 irAEs can increase the risk for infection, including opportunistic infections. One retrospective review of patients administered checkpoint inhibitors at a single institution revealed that 7.3% of 740 patients developed a severe infection that lead to hospitalization or treatment with intravenous antibiotics.9 In that patient cohort, only 0.6% had a serious infection secondary to a viral etiology, and 1 patient developed CMV enterocolitis. Most patients who developed an infection in this cohort had received corticosteroids (46/54 patients, 85%) and/or infliximab (13/54 patients, 24%).9
CMV is a member of the Herpesviridae family. After a primary infection, which can often go unrecognized in an immunocompetent host, CMV can persist in a latent state.10 In a study by Bate and colleagues, the age-adjusted seropositivity of CMV was found to be 50.4%.11 Based on those results, immunosuppression in a patient who has previously been infected with CMV can lead to a risk of reactivation or even reinfection. In the era of checkpoint-inhibitor therapy, reactivation of CMV has been described previously in a case of CMV hepatitis and a report of CMV colitis.12,13 Immunosuppression, such as that caused by corticosteroids, is a risk factor for CMV infection.14 Colitis caused by CMV usually presents with abdominal pain, diarrhea, and bloody diarrhea.15 In suspected cases of CMV colitis, endoscopy should be pursued with biopsy for tissue examination. A tissue diagnosis is required for CMV colitis because serum PCR can be negative in isolated cases of gastrointestinal CMV infection.15
Conclusion
Despite appropriate treatment with ganciclovir and the noted response in the patient’s serum CMV PCR, symptom exacerbation was observed with the transition to oral prednisone. The requirement for intravenous corticosteroids in the present case demonstrates the prolonged effects exerted by irAEs secondary to checkpoint-inhibitor therapy. Those effects are attributable to the design of the antibody – ipilimumab is a fully humanized monoclonal antibody and has a plasma half-life of about 15 days.1,4
By the identification of CMV histopathologically, this case, along with the case presented by Lankes and colleagues,13 illustrates the importance of considering CMV colitis in patients who are being treated with ipilimumab and who develop persistent or worsening diarrhea after initial treatment with high-dose steroids.
Early recognition of possible coexistent CMV colitis in patients with a history of treatment with ipilimumab can have important clinical consequences. It can lead to quicker implementation of proper antiviral therapy and minimization of immune suppression to levels required to maintain control of the patient’s symptoms.
1. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711-723.
2. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med. 2016;375(19):1845-1855.
3. Glassman PM, Balthasar JP. Mechanistic considerations for the use of monoclonal antibodies for cancer therapy. Cancer Biol Med. 2014;11(1):20-33.
4. Weber JS, Kahler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30(21):2691-2697.
5. Fecher LA, Agarwala SS, Hodi FS, Weber JS. Ipilimumab and its toxicities: a multidisciplinary approach. Oncologist. 2013;18(6):733-743.
6. Weber JS, Dummer R, de Pril V, Lebbe C, Hodi FS, Investigators MDX. Patterns of onset and resolution of immune-related adverse events of special interest with ipilimumab: detailed safety analysis from a phase 3 trial in patients with advanced melanoma. Cancer. 2013;119(9):1675-1682.
7. Oble DA, Mino-Kenudson M, Goldsmith J, et al. Alpha-CTLA-4 mAb-associated panenteritis: a histologic and immunohistochemical analysis. Am J Surg Pathol. 2008;32(8):1130-1137.
8. Beck KE, Blansfield JA, Tran KQ, et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol. 2006;24(15):2283-2289.
9. Del Castillo M, Romero FA, Arguello E, Kyi C, Postow MA, Redelman-Sidi G. The spectrum of serious infections among patients receiving immune checkpoint blockade for the treatment of melanoma. Clin Infect Dis. 2016;63(11):1490-1493.
10. Pillet S, Pozzetto B, Roblin X. Cytomegalovirus and ulcerative colitis: place of antiviral therapy. World J Gastroenterol. 2016;22(6):2030-2045.
11. Bate SL, Dollard SC, Cannon MJ. Cytomegalovirus seroprevalence in the United States: the national health and nutrition examination surveys, 1988-2004. Clin Infect Dis. 2010;50(11):1439-1447.
12. Uslu U, Agaimy A, Hundorfean G, Harrer T, Schuler G, Heinzerling L. autoimmune colitis and subsequent CMV-induced hepatitis after treatment with ipilimumab. J Immunother. 2015;38(5):212-215.
13. Lankes K, Hundorfean G, Harrer T, et al. Anti-TNF-refractory colitis after checkpoint inhibitor therapy: possible role of CMV-mediated immunopathogenesis. Oncoimmunology. 2016;5(6):e1128611.
14. Ko JH, Peck KR, Lee WJ, et al. Clinical presentation and risk factors for cytomegalovirus colitis in immunocompetent adult patients. Clin Infect Dis. 2015;60(6):e20-26.
15. You DM, Johnson MD. Cytomegalovirus infection and the gastrointestinal tract. Curr Gastroenterol Rep. 2012;14(4):334-342.
Immune checkpoint inhibitors, including anti-cytotoxic T-lymphocyte antigen 4 (anti-CTLA4) and anti-programmed cell death protein-1 (anti-PD-1) antibodies, have demonstrated clinical and survival benefits in a variety of malignancies, which has led to an expansion in their role in oncology. In melanoma, the anti-CTLA-4 antibody, ipilimumab, has demonstrated a survival benefit in patients with advanced metastatic melanoma and in patients with resectable disease with lymph node involvement.1,2
Ipilimumab exerts its effect by binding CTLA-4 on conventional and regulatory T cells, thus blocking inhibitory signals on T cells, which leads to an antitumor response.3 The increased immune response counteracts the immune-evading mechanisms of the tumor. With increased use of these agents, immune-related adverse events (irAEs) have become more prevalent. The most common irAEs secondary to ipilimumab are skin rash, colitis/diarrhea, hepatitis, pneumonitis, and various endocrinopathies.4 In a phase 3 trial of adjuvant ipilimumab in patients with resected stage III melanoma, grade 3 or 4 adverse events occurred in 54.1% of participants in the ipilimumab arm, the most common being diarrhea and colitis (9.8% and 6.8%, respectively).2Recognition and management of irAEs has led to the implementation of treatment guidelines.4,5 Management of irAEs includes checkpoint inhibitor discontinuation and reversal of the immune response by institution of immunosuppression with corticosteroids.
Case presentation and summary
A 40-year-old white woman with stage IIIB BRAF V600E-positive melanoma presented with diarrhea refractory to high-dose prednisone (1 mg/kg BID). She had recently undergone wide local excision and sentinel node biopsy and received her inaugural dose of ipilimumab (10 mg/kg).
The patient first presented with loose, watery stools that had begun 8 days after she had received her first dose of adjuvant ipilimumab. She was admitted to the hospital, and intravenous methylprednisolone was initiated along with empiric ciprofloxacin (400 mg, IVPB Q12h) and metronidazole (500 mg, IVPB Q8h) as infectious causes were concurrently ruled out. During this initial admission, the patient’s stool was negative for Clostridium difficile toxin, ova, and parasites, as well as enteric pathogens by culture. After infectious causes were excluded, she was diagnosed with ipilimumab-induced colitis. Antibiotics were discontinued, and the patient ultimately noted improvement in her symptoms. On hospital day 7, she was experiencing only 2 bowel movements a day and was discharged on 80 mg of prednisone twice daily.
After discharge the patient noted persistence of her symptoms. At her follow-up, 9 days after discharge, the patient noted continued symptoms of low-grade diarrhea. She failed a trial of steroid tapering due to exacerbation of her abdominal pain and frequency of diarrhea. Further investigation was negative for C. diff toxin and a computed-tomography scan was consistent with continuing colitis. The patient’s symptoms continued to worsen, with recurrence of grade 3 diarrhea, and she was ultimately readmitted 17 days after her earlier discharge (36 days after her first ipilimumab dosing).
On re-admission, the patient was again given intravenous methylprednisolone and experienced interval improvement in the frequency of diarrhea. A gastroenterology expert was consulted, and the patient underwent a flexible sigmoidoscopy that demonstrated findings of diffuse and severe inflammation and biopsies were obtained (Figure 1). After several days of continued symptoms, the patient received infliximab 5 mg/kg for treatment of her adverse autoimmune reaction. After administration, the patient noted improvement in the frequency and volume of diarrhea, however, her symptoms still persisted.
Biopsy results subsequently revealed findings compatible with ipilimumab-induced colitis, and immunohistochemical staining demonstrated positivity for cytomegalovirus (CMV). Specifically, histologic examination showed lymphoplasmacytic expansion of the lamina propria, some architectural distortion, and increased crypt apoptosis. Scattered cryptitis and crypt abscesses were also noted, as were rare stromal and endothelial cells with characteristic CMV inclusions (Figure 2 and Figure 3).
Serum CMV polymerase chain reaction (PCR) was also positive at 652,000 IU/mL (lower limit of detection 100 IU/mL). Induction dosing of ganciclovir (5 mg/kg IV Q12h) was initiated. The combined treatment with intravenous methylprednisone and ganciclovir led to an improvement in diarrhea frequency and resolution of blood in the stool. She was transitioned to oral prednisone, but it resulted in redevelopment of grade 3 diarrhea. The patient was therefore resumed on and discharged on daily intravenous methylprednisolone.
After discharge, the patient was started on budesonide 9 mg daily. Her serum CMV PCR level reduced and she was transitioned to oral valgancyclovir (900 mg daily) for maintenance. Another unsuccessful attempt was made to switch her to oral prednisone.
About 14 weeks after the initial ipilimumab dosing, the patient underwent another flexible sigmoidoscopy that again demonstrated severe colitis from the rectum to sigmoid colon. Biopsies were negative for CMV. Patient was readmitted for recurrence of diarrhea the following week. Treatment with IV methylprednisone (1mg/kg BID) and infliximab (5 mg/kg) again led to an improvement of symptoms. She was again discharged on IV methylprednisone (1 mg/kg BID) with a taper.
In the 15th week after her initial ipilimumab dose, the patient presented with a perforated bowel, requiring a subtotal colectomy and end ileostomy. She continued on a slow taper of oral prednisone (50 mg daily and decrease by 10 mg every 5 days).
At her last documented follow-up, 8 months after her first ipilimumab dose, she was having normal output from her ileostomy. She developed secondary adrenal insufficiency because of the long-term steroids and continued to take prednisone 5 mg daily.
Discussion
Diarrhea and colitis are common irAEs attributable to checkpoint-inhibitor therapy used for the treatment of melanoma. This case of ipilimumab-induced colitis refractory to high-dose oral steroids demonstrates the risks associated with management of anti-CTLA-4 induced colitis. In particular, the high-dose corticosteroids required to treat the autoimmune component of this patient’s colitis increased her susceptibility to CMV reactivation.
The diagnosis of colitis secondary to ipilimumab is made primarily in the appropriate clinical setting, and typically onsets during the induction period (within 12 weeks of initial dosing) and most resolve within 6-8 weeks.6 Histopathologically, there is lymphoplasmacytic expansion of lamina propria, increased intraepithelial lymphocytes, and increased epithelial apoptosis of crypts. One can also see acute cryptitis and crypt abscesses. Reactive epithelial changes with mucin depletion are also often seen in epithelial cells.
Findings from immunohistochemical studies have shown the increased intraepithelial lymphocytes to be predominantly CD8-positive T cells, while the lamina propria contains an increase in the mixture of CD4- and CD8-positive T cells. In addition, small intestinal samples show villous blunting. There is an absence of significant architectural distortion and well-developed basal lymphoplasmacytic infiltrates characteristic of chronic mucosal injury, such as idiopathic inflammatory bowel disease.7 Granulomas are also absent in most series, though they have been reported in some cases.8 The features are similar to those seen in autoimmune enteropathy, but goblet and endocrine cells remain preserved. Graft-versus-host disease has similar histologic features, however, the clinical setting usually makes the distinction between these obvious.
Current treatment algorithms for ipilimumab-related diarrhea, begin with immediate treatment with intravenous methylprednisolone (125 mg once). This is followed with oral prednisone at a dose of 1-2 mg/kg tapered over 4 to 8 weeks.4 In patients with persistent symptoms despite adequate doses of corticosteroids, infliximab (5 mg/kg every 2 weeks) is recommended until the resolution of symptoms, and a longer taper of prednisone is often necessary.
Institution of high-dose corticosteroids to treat grade 3 or 4 irAEs can increase the risk for infection, including opportunistic infections. One retrospective review of patients administered checkpoint inhibitors at a single institution revealed that 7.3% of 740 patients developed a severe infection that lead to hospitalization or treatment with intravenous antibiotics.9 In that patient cohort, only 0.6% had a serious infection secondary to a viral etiology, and 1 patient developed CMV enterocolitis. Most patients who developed an infection in this cohort had received corticosteroids (46/54 patients, 85%) and/or infliximab (13/54 patients, 24%).9
CMV is a member of the Herpesviridae family. After a primary infection, which can often go unrecognized in an immunocompetent host, CMV can persist in a latent state.10 In a study by Bate and colleagues, the age-adjusted seropositivity of CMV was found to be 50.4%.11 Based on those results, immunosuppression in a patient who has previously been infected with CMV can lead to a risk of reactivation or even reinfection. In the era of checkpoint-inhibitor therapy, reactivation of CMV has been described previously in a case of CMV hepatitis and a report of CMV colitis.12,13 Immunosuppression, such as that caused by corticosteroids, is a risk factor for CMV infection.14 Colitis caused by CMV usually presents with abdominal pain, diarrhea, and bloody diarrhea.15 In suspected cases of CMV colitis, endoscopy should be pursued with biopsy for tissue examination. A tissue diagnosis is required for CMV colitis because serum PCR can be negative in isolated cases of gastrointestinal CMV infection.15
Conclusion
Despite appropriate treatment with ganciclovir and the noted response in the patient’s serum CMV PCR, symptom exacerbation was observed with the transition to oral prednisone. The requirement for intravenous corticosteroids in the present case demonstrates the prolonged effects exerted by irAEs secondary to checkpoint-inhibitor therapy. Those effects are attributable to the design of the antibody – ipilimumab is a fully humanized monoclonal antibody and has a plasma half-life of about 15 days.1,4
By the identification of CMV histopathologically, this case, along with the case presented by Lankes and colleagues,13 illustrates the importance of considering CMV colitis in patients who are being treated with ipilimumab and who develop persistent or worsening diarrhea after initial treatment with high-dose steroids.
Early recognition of possible coexistent CMV colitis in patients with a history of treatment with ipilimumab can have important clinical consequences. It can lead to quicker implementation of proper antiviral therapy and minimization of immune suppression to levels required to maintain control of the patient’s symptoms.
Immune checkpoint inhibitors, including anti-cytotoxic T-lymphocyte antigen 4 (anti-CTLA4) and anti-programmed cell death protein-1 (anti-PD-1) antibodies, have demonstrated clinical and survival benefits in a variety of malignancies, which has led to an expansion in their role in oncology. In melanoma, the anti-CTLA-4 antibody, ipilimumab, has demonstrated a survival benefit in patients with advanced metastatic melanoma and in patients with resectable disease with lymph node involvement.1,2
Ipilimumab exerts its effect by binding CTLA-4 on conventional and regulatory T cells, thus blocking inhibitory signals on T cells, which leads to an antitumor response.3 The increased immune response counteracts the immune-evading mechanisms of the tumor. With increased use of these agents, immune-related adverse events (irAEs) have become more prevalent. The most common irAEs secondary to ipilimumab are skin rash, colitis/diarrhea, hepatitis, pneumonitis, and various endocrinopathies.4 In a phase 3 trial of adjuvant ipilimumab in patients with resected stage III melanoma, grade 3 or 4 adverse events occurred in 54.1% of participants in the ipilimumab arm, the most common being diarrhea and colitis (9.8% and 6.8%, respectively).2Recognition and management of irAEs has led to the implementation of treatment guidelines.4,5 Management of irAEs includes checkpoint inhibitor discontinuation and reversal of the immune response by institution of immunosuppression with corticosteroids.
Case presentation and summary
A 40-year-old white woman with stage IIIB BRAF V600E-positive melanoma presented with diarrhea refractory to high-dose prednisone (1 mg/kg BID). She had recently undergone wide local excision and sentinel node biopsy and received her inaugural dose of ipilimumab (10 mg/kg).
The patient first presented with loose, watery stools that had begun 8 days after she had received her first dose of adjuvant ipilimumab. She was admitted to the hospital, and intravenous methylprednisolone was initiated along with empiric ciprofloxacin (400 mg, IVPB Q12h) and metronidazole (500 mg, IVPB Q8h) as infectious causes were concurrently ruled out. During this initial admission, the patient’s stool was negative for Clostridium difficile toxin, ova, and parasites, as well as enteric pathogens by culture. After infectious causes were excluded, she was diagnosed with ipilimumab-induced colitis. Antibiotics were discontinued, and the patient ultimately noted improvement in her symptoms. On hospital day 7, she was experiencing only 2 bowel movements a day and was discharged on 80 mg of prednisone twice daily.
After discharge the patient noted persistence of her symptoms. At her follow-up, 9 days after discharge, the patient noted continued symptoms of low-grade diarrhea. She failed a trial of steroid tapering due to exacerbation of her abdominal pain and frequency of diarrhea. Further investigation was negative for C. diff toxin and a computed-tomography scan was consistent with continuing colitis. The patient’s symptoms continued to worsen, with recurrence of grade 3 diarrhea, and she was ultimately readmitted 17 days after her earlier discharge (36 days after her first ipilimumab dosing).
On re-admission, the patient was again given intravenous methylprednisolone and experienced interval improvement in the frequency of diarrhea. A gastroenterology expert was consulted, and the patient underwent a flexible sigmoidoscopy that demonstrated findings of diffuse and severe inflammation and biopsies were obtained (Figure 1). After several days of continued symptoms, the patient received infliximab 5 mg/kg for treatment of her adverse autoimmune reaction. After administration, the patient noted improvement in the frequency and volume of diarrhea, however, her symptoms still persisted.
Biopsy results subsequently revealed findings compatible with ipilimumab-induced colitis, and immunohistochemical staining demonstrated positivity for cytomegalovirus (CMV). Specifically, histologic examination showed lymphoplasmacytic expansion of the lamina propria, some architectural distortion, and increased crypt apoptosis. Scattered cryptitis and crypt abscesses were also noted, as were rare stromal and endothelial cells with characteristic CMV inclusions (Figure 2 and Figure 3).
Serum CMV polymerase chain reaction (PCR) was also positive at 652,000 IU/mL (lower limit of detection 100 IU/mL). Induction dosing of ganciclovir (5 mg/kg IV Q12h) was initiated. The combined treatment with intravenous methylprednisone and ganciclovir led to an improvement in diarrhea frequency and resolution of blood in the stool. She was transitioned to oral prednisone, but it resulted in redevelopment of grade 3 diarrhea. The patient was therefore resumed on and discharged on daily intravenous methylprednisolone.
After discharge, the patient was started on budesonide 9 mg daily. Her serum CMV PCR level reduced and she was transitioned to oral valgancyclovir (900 mg daily) for maintenance. Another unsuccessful attempt was made to switch her to oral prednisone.
About 14 weeks after the initial ipilimumab dosing, the patient underwent another flexible sigmoidoscopy that again demonstrated severe colitis from the rectum to sigmoid colon. Biopsies were negative for CMV. Patient was readmitted for recurrence of diarrhea the following week. Treatment with IV methylprednisone (1mg/kg BID) and infliximab (5 mg/kg) again led to an improvement of symptoms. She was again discharged on IV methylprednisone (1 mg/kg BID) with a taper.
In the 15th week after her initial ipilimumab dose, the patient presented with a perforated bowel, requiring a subtotal colectomy and end ileostomy. She continued on a slow taper of oral prednisone (50 mg daily and decrease by 10 mg every 5 days).
At her last documented follow-up, 8 months after her first ipilimumab dose, she was having normal output from her ileostomy. She developed secondary adrenal insufficiency because of the long-term steroids and continued to take prednisone 5 mg daily.
Discussion
Diarrhea and colitis are common irAEs attributable to checkpoint-inhibitor therapy used for the treatment of melanoma. This case of ipilimumab-induced colitis refractory to high-dose oral steroids demonstrates the risks associated with management of anti-CTLA-4 induced colitis. In particular, the high-dose corticosteroids required to treat the autoimmune component of this patient’s colitis increased her susceptibility to CMV reactivation.
The diagnosis of colitis secondary to ipilimumab is made primarily in the appropriate clinical setting, and typically onsets during the induction period (within 12 weeks of initial dosing) and most resolve within 6-8 weeks.6 Histopathologically, there is lymphoplasmacytic expansion of lamina propria, increased intraepithelial lymphocytes, and increased epithelial apoptosis of crypts. One can also see acute cryptitis and crypt abscesses. Reactive epithelial changes with mucin depletion are also often seen in epithelial cells.
Findings from immunohistochemical studies have shown the increased intraepithelial lymphocytes to be predominantly CD8-positive T cells, while the lamina propria contains an increase in the mixture of CD4- and CD8-positive T cells. In addition, small intestinal samples show villous blunting. There is an absence of significant architectural distortion and well-developed basal lymphoplasmacytic infiltrates characteristic of chronic mucosal injury, such as idiopathic inflammatory bowel disease.7 Granulomas are also absent in most series, though they have been reported in some cases.8 The features are similar to those seen in autoimmune enteropathy, but goblet and endocrine cells remain preserved. Graft-versus-host disease has similar histologic features, however, the clinical setting usually makes the distinction between these obvious.
Current treatment algorithms for ipilimumab-related diarrhea, begin with immediate treatment with intravenous methylprednisolone (125 mg once). This is followed with oral prednisone at a dose of 1-2 mg/kg tapered over 4 to 8 weeks.4 In patients with persistent symptoms despite adequate doses of corticosteroids, infliximab (5 mg/kg every 2 weeks) is recommended until the resolution of symptoms, and a longer taper of prednisone is often necessary.
Institution of high-dose corticosteroids to treat grade 3 or 4 irAEs can increase the risk for infection, including opportunistic infections. One retrospective review of patients administered checkpoint inhibitors at a single institution revealed that 7.3% of 740 patients developed a severe infection that lead to hospitalization or treatment with intravenous antibiotics.9 In that patient cohort, only 0.6% had a serious infection secondary to a viral etiology, and 1 patient developed CMV enterocolitis. Most patients who developed an infection in this cohort had received corticosteroids (46/54 patients, 85%) and/or infliximab (13/54 patients, 24%).9
CMV is a member of the Herpesviridae family. After a primary infection, which can often go unrecognized in an immunocompetent host, CMV can persist in a latent state.10 In a study by Bate and colleagues, the age-adjusted seropositivity of CMV was found to be 50.4%.11 Based on those results, immunosuppression in a patient who has previously been infected with CMV can lead to a risk of reactivation or even reinfection. In the era of checkpoint-inhibitor therapy, reactivation of CMV has been described previously in a case of CMV hepatitis and a report of CMV colitis.12,13 Immunosuppression, such as that caused by corticosteroids, is a risk factor for CMV infection.14 Colitis caused by CMV usually presents with abdominal pain, diarrhea, and bloody diarrhea.15 In suspected cases of CMV colitis, endoscopy should be pursued with biopsy for tissue examination. A tissue diagnosis is required for CMV colitis because serum PCR can be negative in isolated cases of gastrointestinal CMV infection.15
Conclusion
Despite appropriate treatment with ganciclovir and the noted response in the patient’s serum CMV PCR, symptom exacerbation was observed with the transition to oral prednisone. The requirement for intravenous corticosteroids in the present case demonstrates the prolonged effects exerted by irAEs secondary to checkpoint-inhibitor therapy. Those effects are attributable to the design of the antibody – ipilimumab is a fully humanized monoclonal antibody and has a plasma half-life of about 15 days.1,4
By the identification of CMV histopathologically, this case, along with the case presented by Lankes and colleagues,13 illustrates the importance of considering CMV colitis in patients who are being treated with ipilimumab and who develop persistent or worsening diarrhea after initial treatment with high-dose steroids.
Early recognition of possible coexistent CMV colitis in patients with a history of treatment with ipilimumab can have important clinical consequences. It can lead to quicker implementation of proper antiviral therapy and minimization of immune suppression to levels required to maintain control of the patient’s symptoms.
1. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711-723.
2. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med. 2016;375(19):1845-1855.
3. Glassman PM, Balthasar JP. Mechanistic considerations for the use of monoclonal antibodies for cancer therapy. Cancer Biol Med. 2014;11(1):20-33.
4. Weber JS, Kahler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30(21):2691-2697.
5. Fecher LA, Agarwala SS, Hodi FS, Weber JS. Ipilimumab and its toxicities: a multidisciplinary approach. Oncologist. 2013;18(6):733-743.
6. Weber JS, Dummer R, de Pril V, Lebbe C, Hodi FS, Investigators MDX. Patterns of onset and resolution of immune-related adverse events of special interest with ipilimumab: detailed safety analysis from a phase 3 trial in patients with advanced melanoma. Cancer. 2013;119(9):1675-1682.
7. Oble DA, Mino-Kenudson M, Goldsmith J, et al. Alpha-CTLA-4 mAb-associated panenteritis: a histologic and immunohistochemical analysis. Am J Surg Pathol. 2008;32(8):1130-1137.
8. Beck KE, Blansfield JA, Tran KQ, et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol. 2006;24(15):2283-2289.
9. Del Castillo M, Romero FA, Arguello E, Kyi C, Postow MA, Redelman-Sidi G. The spectrum of serious infections among patients receiving immune checkpoint blockade for the treatment of melanoma. Clin Infect Dis. 2016;63(11):1490-1493.
10. Pillet S, Pozzetto B, Roblin X. Cytomegalovirus and ulcerative colitis: place of antiviral therapy. World J Gastroenterol. 2016;22(6):2030-2045.
11. Bate SL, Dollard SC, Cannon MJ. Cytomegalovirus seroprevalence in the United States: the national health and nutrition examination surveys, 1988-2004. Clin Infect Dis. 2010;50(11):1439-1447.
12. Uslu U, Agaimy A, Hundorfean G, Harrer T, Schuler G, Heinzerling L. autoimmune colitis and subsequent CMV-induced hepatitis after treatment with ipilimumab. J Immunother. 2015;38(5):212-215.
13. Lankes K, Hundorfean G, Harrer T, et al. Anti-TNF-refractory colitis after checkpoint inhibitor therapy: possible role of CMV-mediated immunopathogenesis. Oncoimmunology. 2016;5(6):e1128611.
14. Ko JH, Peck KR, Lee WJ, et al. Clinical presentation and risk factors for cytomegalovirus colitis in immunocompetent adult patients. Clin Infect Dis. 2015;60(6):e20-26.
15. You DM, Johnson MD. Cytomegalovirus infection and the gastrointestinal tract. Curr Gastroenterol Rep. 2012;14(4):334-342.
1. Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711-723.
2. Eggermont AM, Chiarion-Sileni V, Grob JJ, et al. Prolonged survival in stage III melanoma with ipilimumab adjuvant therapy. N Engl J Med. 2016;375(19):1845-1855.
3. Glassman PM, Balthasar JP. Mechanistic considerations for the use of monoclonal antibodies for cancer therapy. Cancer Biol Med. 2014;11(1):20-33.
4. Weber JS, Kahler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30(21):2691-2697.
5. Fecher LA, Agarwala SS, Hodi FS, Weber JS. Ipilimumab and its toxicities: a multidisciplinary approach. Oncologist. 2013;18(6):733-743.
6. Weber JS, Dummer R, de Pril V, Lebbe C, Hodi FS, Investigators MDX. Patterns of onset and resolution of immune-related adverse events of special interest with ipilimumab: detailed safety analysis from a phase 3 trial in patients with advanced melanoma. Cancer. 2013;119(9):1675-1682.
7. Oble DA, Mino-Kenudson M, Goldsmith J, et al. Alpha-CTLA-4 mAb-associated panenteritis: a histologic and immunohistochemical analysis. Am J Surg Pathol. 2008;32(8):1130-1137.
8. Beck KE, Blansfield JA, Tran KQ, et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol. 2006;24(15):2283-2289.
9. Del Castillo M, Romero FA, Arguello E, Kyi C, Postow MA, Redelman-Sidi G. The spectrum of serious infections among patients receiving immune checkpoint blockade for the treatment of melanoma. Clin Infect Dis. 2016;63(11):1490-1493.
10. Pillet S, Pozzetto B, Roblin X. Cytomegalovirus and ulcerative colitis: place of antiviral therapy. World J Gastroenterol. 2016;22(6):2030-2045.
11. Bate SL, Dollard SC, Cannon MJ. Cytomegalovirus seroprevalence in the United States: the national health and nutrition examination surveys, 1988-2004. Clin Infect Dis. 2010;50(11):1439-1447.
12. Uslu U, Agaimy A, Hundorfean G, Harrer T, Schuler G, Heinzerling L. autoimmune colitis and subsequent CMV-induced hepatitis after treatment with ipilimumab. J Immunother. 2015;38(5):212-215.
13. Lankes K, Hundorfean G, Harrer T, et al. Anti-TNF-refractory colitis after checkpoint inhibitor therapy: possible role of CMV-mediated immunopathogenesis. Oncoimmunology. 2016;5(6):e1128611.
14. Ko JH, Peck KR, Lee WJ, et al. Clinical presentation and risk factors for cytomegalovirus colitis in immunocompetent adult patients. Clin Infect Dis. 2015;60(6):e20-26.
15. You DM, Johnson MD. Cytomegalovirus infection and the gastrointestinal tract. Curr Gastroenterol Rep. 2012;14(4):334-342.
VIDEO: Gene test guides need for sentinel node biopsy in elderly melanoma patients
SAN DIEGO – The results of a gene expression test, along with tumor thickness and patient age, can guide the need for sentinel lymph node biopsy, based on results from more than1,400 consecutively tested patients from 26 U.S. surgical oncology, medical oncology and dermatologic practices.
The findings, presented by John Vetto, MD, at the annual meeting of the American Academy of Dermatology, indicate the DecisionDx test correctly identified patients aged 65 and older with T1-T2 tumors whose risk of sentinel node metastasis was lower than 5%. The most recent melanoma guidelines from the National Comprehensive Cancer Network recommend that clinicians “discuss and offer” sentinel node biopsy if a patient has a greater than 10% likelihood of a positive node. If the likelihood is 5%-10%, the recommendation is to “discuss and consider” the procedure. But if the likelihood of a positive node is less than 5%, the guidelines recommend against a biopsy.
“Sentinel node biopsy (has) risks, especially in medically compromised older patients,” Dr. Vetto, professor of surgery at the Oregon Health and Sciences University, Portland, said in an interview, in which he discussed clinical use of the test. “This test offers us a good way to assess the risk/benefit ratio so we can better care for patients, and follow the newest guidelines about sentinel node biopsy.”
The DecisionDx Melanoma, developed by Castle Biosciences, tests for the expression of 28 genes know to play a role in melanoma metastasis, and three control genes. Tumors are stratified either as Class 1, with a 3% chance of spreading within 5 years, or Class 2, with a 69% risk of metastasis. There are two subclasses: 1A, which has an extremely low risk of progression, and 2b, which has an extremely high risk of progression.
For patients with T1-T2 tumors who had a Class 1A test result (lowest risk of recurrence), SLN positivity was 4.6% for all ages, 2.8% in patients 55 years and older, and 1.6% in patients 65 years and older. For patients with T1-T2 tumors who had a Class 2B test result (highest risk of recurrence), SLN positivity was 18.8% for all ages, 16.4% in patients 55 years and older and 11.9% in patients 65 years and older.
Dr. Vetto is a paid speaker for Castle Biosciences.
SOURCE: Vetto et al. AAD 2018 late-breaking research, Abstract 6805
SAN DIEGO – The results of a gene expression test, along with tumor thickness and patient age, can guide the need for sentinel lymph node biopsy, based on results from more than1,400 consecutively tested patients from 26 U.S. surgical oncology, medical oncology and dermatologic practices.
The findings, presented by John Vetto, MD, at the annual meeting of the American Academy of Dermatology, indicate the DecisionDx test correctly identified patients aged 65 and older with T1-T2 tumors whose risk of sentinel node metastasis was lower than 5%. The most recent melanoma guidelines from the National Comprehensive Cancer Network recommend that clinicians “discuss and offer” sentinel node biopsy if a patient has a greater than 10% likelihood of a positive node. If the likelihood is 5%-10%, the recommendation is to “discuss and consider” the procedure. But if the likelihood of a positive node is less than 5%, the guidelines recommend against a biopsy.
“Sentinel node biopsy (has) risks, especially in medically compromised older patients,” Dr. Vetto, professor of surgery at the Oregon Health and Sciences University, Portland, said in an interview, in which he discussed clinical use of the test. “This test offers us a good way to assess the risk/benefit ratio so we can better care for patients, and follow the newest guidelines about sentinel node biopsy.”
The DecisionDx Melanoma, developed by Castle Biosciences, tests for the expression of 28 genes know to play a role in melanoma metastasis, and three control genes. Tumors are stratified either as Class 1, with a 3% chance of spreading within 5 years, or Class 2, with a 69% risk of metastasis. There are two subclasses: 1A, which has an extremely low risk of progression, and 2b, which has an extremely high risk of progression.
For patients with T1-T2 tumors who had a Class 1A test result (lowest risk of recurrence), SLN positivity was 4.6% for all ages, 2.8% in patients 55 years and older, and 1.6% in patients 65 years and older. For patients with T1-T2 tumors who had a Class 2B test result (highest risk of recurrence), SLN positivity was 18.8% for all ages, 16.4% in patients 55 years and older and 11.9% in patients 65 years and older.
Dr. Vetto is a paid speaker for Castle Biosciences.
SOURCE: Vetto et al. AAD 2018 late-breaking research, Abstract 6805
SAN DIEGO – The results of a gene expression test, along with tumor thickness and patient age, can guide the need for sentinel lymph node biopsy, based on results from more than1,400 consecutively tested patients from 26 U.S. surgical oncology, medical oncology and dermatologic practices.
The findings, presented by John Vetto, MD, at the annual meeting of the American Academy of Dermatology, indicate the DecisionDx test correctly identified patients aged 65 and older with T1-T2 tumors whose risk of sentinel node metastasis was lower than 5%. The most recent melanoma guidelines from the National Comprehensive Cancer Network recommend that clinicians “discuss and offer” sentinel node biopsy if a patient has a greater than 10% likelihood of a positive node. If the likelihood is 5%-10%, the recommendation is to “discuss and consider” the procedure. But if the likelihood of a positive node is less than 5%, the guidelines recommend against a biopsy.
“Sentinel node biopsy (has) risks, especially in medically compromised older patients,” Dr. Vetto, professor of surgery at the Oregon Health and Sciences University, Portland, said in an interview, in which he discussed clinical use of the test. “This test offers us a good way to assess the risk/benefit ratio so we can better care for patients, and follow the newest guidelines about sentinel node biopsy.”
The DecisionDx Melanoma, developed by Castle Biosciences, tests for the expression of 28 genes know to play a role in melanoma metastasis, and three control genes. Tumors are stratified either as Class 1, with a 3% chance of spreading within 5 years, or Class 2, with a 69% risk of metastasis. There are two subclasses: 1A, which has an extremely low risk of progression, and 2b, which has an extremely high risk of progression.
For patients with T1-T2 tumors who had a Class 1A test result (lowest risk of recurrence), SLN positivity was 4.6% for all ages, 2.8% in patients 55 years and older, and 1.6% in patients 65 years and older. For patients with T1-T2 tumors who had a Class 2B test result (highest risk of recurrence), SLN positivity was 18.8% for all ages, 16.4% in patients 55 years and older and 11.9% in patients 65 years and older.
Dr. Vetto is a paid speaker for Castle Biosciences.
SOURCE: Vetto et al. AAD 2018 late-breaking research, Abstract 6805
REPORTING FROM AAD 18
Dermoscopy will help diagnose pediatric melanomas
Adding dermoscopy to the classic and modified melanoma ABCD criteria will help speed detection of melanoma in children, said Cristina Carrera, MD, of the University of Barcelona, and her associates.
Pediatric melanomas make up less than 3% of pediatric cancers and 1%-4% of all melanomas, occurring more commonly in adolescents than in children aged 5-9 years. These cancers often are thicker when first seen because of delay in diagnosis and/or differences in growth dynamics. Delay in diagnosis may occur because of the low incidence of pediatric melanoma and because classic melanoma criteria – ABCD: asymmetry, border irregularity, multiple colors, diameter (6 mm) – don’t always apply, they said.
A modified clinical ABCD rule of ‘‘amelanotic, bleeding bump, color uniformity, and de novo lesion of any diameter’’ was proposed to improve early detection of pediatric melanoma. And because dermoscopy improves melanoma diagnosis in adults, researchers decided to examine clinical and dermascopic findings in a group of pediatric melanoma cases.
Fifty-two pediatric melanoma cases from 51 patients were collected from pigmented lesion clinics across nine countries (Australia, Belgium, Brazil, France, Israel, Italy, Serbia, Spain, and the United States), and clinical and dermoscopic specimens were examined by two experienced reviewers.
The mean age of the patients was 15 years, and 26 of the patients were female. Histopathologically, 28% of the pediatric melanomas were classified as spitzoid and 72% as nonspitzoid. Patients with a spitzoid melanoma were significantly younger than those with nonspitzoid melanoma (12.5 vs. 16 years, P = .004). All of the spitzoid tumors were invasive, and they were significantly thicker than the nonspitzoid melanomas (2.6 vs. 1.2 mm, P = .004). Also, these lesions more frequently were ulcerated than nonspitzoid melanomas (29% vs. 8%, P = .06). Melanomas associated with a nevus more often were among nonspitzoid than spitzoid melanomas (62% vs. 27% P = .02), and the congenital type of nevus was linked most often with nonspitzoid melanomas (25 of 27 [92.6%]), Dr. Carrera and her associates reported.
The pediatric melanomas in this study appeared most frequently on the lower extremities (31%), followed by the back (27%). Spitzoid melanomas most often appeared on the limbs (73%); nonspitzoid melanomas were likely to occur on the torso (52%).
In the majority of cases (52%), the overall clinical morphology was considered to fulfill the classic melanoma ABCD criteria. The remaining lesions that did not fulfill the conventional ABCD criteria were called benign-appearing tumors or nodular/polypoid tumors. Spitzoid melanomas most often were nodular/ polypoid (47%), while most nonspitzoid melanomas (59%) were melanoma-like. Only 21% of the pediatric melanomas fulfilled the modified melanoma ABCD criteria, they noted.
On dermoscopy, which was available in 49 cases, a multicomponent pattern seen in 24 cases mostly was associated with nonspitzoid melanoma (88%, P less than.03); a nevus-like pattern in 9 cases was seen only among nonspitzoid melanomas. A vascular pink Spitz-like pattern in seven cases mostly was associated with spitzoid melanomas (86%, P less than .002). A pigmented Reed-like pattern seen in seven cases occurred more often among spitzoid melanomas, but this did not reach statistical significance, the investigators wrote.
In terms of appearance, “red and white colors, milky red areas, polymorphous vessels, and shiny white structures were associated with spitzoid melanoma. Dark brown color, atypical network, and structureless areas were associated with nonspitzoid melanomas,” Dr. Carrera and her associates said.
Dermoscopic patterns that were pink spitzoid or Reed-like were more likely to be histopathologically classified as spitzoid. These pediatric melanomas were linked with younger age, occurrence on the limbs, and de novo development. On the other hand, dermoscopic patterns that were multicomponent or nevus-like were likely to be histopathologically classified as nonspitzoid. These lesions were linked with older age, fair skin phenotype, family history of melanoma, and a preexisting nevus, the researchers said.
“In contrast to previous reports, the present study highlights the fact that the majority of melanomas diagnosed in patients younger than 20 years look similar to melanomas found in adults. Nonspitzoid melanomas were associated with adolescence and with the presence of melanoma risk factors. All nonspitzoid melanomas displayed dermoscopic features associated with melanoma. Spitzoid melanomas were associated with younger age, location on the extremities, and nodular/polypoid clinical morphology. Dermoscopy of spitzoid melanomas revealed atypical vessels and shiny white lines (if amelanotic) or an asymmetric starburst pattern (if pigmented),” Dr. Carrera and her associates concluded.
The investigators had no relevant financial disclosures. The study was supported in part through a grant from the National Institutes of Health/ National Cancer Institute. The research at the melanoma unit in Barcelona was partially funded by grants from the Spanish Fondo de Investigaciones Sanitarias and CIBER de Enfermedades Raras of the Instituto de Salud Carlos III, Spain; it was confinanced by numerous other grants.
SOURCE: Carrera C et al. J Am Acad Dermatol. 2018;78(2):278-88. doi: 10.1016/j.jaad.2017.09.065.
Adding dermoscopy to the classic and modified melanoma ABCD criteria will help speed detection of melanoma in children, said Cristina Carrera, MD, of the University of Barcelona, and her associates.
Pediatric melanomas make up less than 3% of pediatric cancers and 1%-4% of all melanomas, occurring more commonly in adolescents than in children aged 5-9 years. These cancers often are thicker when first seen because of delay in diagnosis and/or differences in growth dynamics. Delay in diagnosis may occur because of the low incidence of pediatric melanoma and because classic melanoma criteria – ABCD: asymmetry, border irregularity, multiple colors, diameter (6 mm) – don’t always apply, they said.
A modified clinical ABCD rule of ‘‘amelanotic, bleeding bump, color uniformity, and de novo lesion of any diameter’’ was proposed to improve early detection of pediatric melanoma. And because dermoscopy improves melanoma diagnosis in adults, researchers decided to examine clinical and dermascopic findings in a group of pediatric melanoma cases.
Fifty-two pediatric melanoma cases from 51 patients were collected from pigmented lesion clinics across nine countries (Australia, Belgium, Brazil, France, Israel, Italy, Serbia, Spain, and the United States), and clinical and dermoscopic specimens were examined by two experienced reviewers.
The mean age of the patients was 15 years, and 26 of the patients were female. Histopathologically, 28% of the pediatric melanomas were classified as spitzoid and 72% as nonspitzoid. Patients with a spitzoid melanoma were significantly younger than those with nonspitzoid melanoma (12.5 vs. 16 years, P = .004). All of the spitzoid tumors were invasive, and they were significantly thicker than the nonspitzoid melanomas (2.6 vs. 1.2 mm, P = .004). Also, these lesions more frequently were ulcerated than nonspitzoid melanomas (29% vs. 8%, P = .06). Melanomas associated with a nevus more often were among nonspitzoid than spitzoid melanomas (62% vs. 27% P = .02), and the congenital type of nevus was linked most often with nonspitzoid melanomas (25 of 27 [92.6%]), Dr. Carrera and her associates reported.
The pediatric melanomas in this study appeared most frequently on the lower extremities (31%), followed by the back (27%). Spitzoid melanomas most often appeared on the limbs (73%); nonspitzoid melanomas were likely to occur on the torso (52%).
In the majority of cases (52%), the overall clinical morphology was considered to fulfill the classic melanoma ABCD criteria. The remaining lesions that did not fulfill the conventional ABCD criteria were called benign-appearing tumors or nodular/polypoid tumors. Spitzoid melanomas most often were nodular/ polypoid (47%), while most nonspitzoid melanomas (59%) were melanoma-like. Only 21% of the pediatric melanomas fulfilled the modified melanoma ABCD criteria, they noted.
On dermoscopy, which was available in 49 cases, a multicomponent pattern seen in 24 cases mostly was associated with nonspitzoid melanoma (88%, P less than.03); a nevus-like pattern in 9 cases was seen only among nonspitzoid melanomas. A vascular pink Spitz-like pattern in seven cases mostly was associated with spitzoid melanomas (86%, P less than .002). A pigmented Reed-like pattern seen in seven cases occurred more often among spitzoid melanomas, but this did not reach statistical significance, the investigators wrote.
In terms of appearance, “red and white colors, milky red areas, polymorphous vessels, and shiny white structures were associated with spitzoid melanoma. Dark brown color, atypical network, and structureless areas were associated with nonspitzoid melanomas,” Dr. Carrera and her associates said.
Dermoscopic patterns that were pink spitzoid or Reed-like were more likely to be histopathologically classified as spitzoid. These pediatric melanomas were linked with younger age, occurrence on the limbs, and de novo development. On the other hand, dermoscopic patterns that were multicomponent or nevus-like were likely to be histopathologically classified as nonspitzoid. These lesions were linked with older age, fair skin phenotype, family history of melanoma, and a preexisting nevus, the researchers said.
“In contrast to previous reports, the present study highlights the fact that the majority of melanomas diagnosed in patients younger than 20 years look similar to melanomas found in adults. Nonspitzoid melanomas were associated with adolescence and with the presence of melanoma risk factors. All nonspitzoid melanomas displayed dermoscopic features associated with melanoma. Spitzoid melanomas were associated with younger age, location on the extremities, and nodular/polypoid clinical morphology. Dermoscopy of spitzoid melanomas revealed atypical vessels and shiny white lines (if amelanotic) or an asymmetric starburst pattern (if pigmented),” Dr. Carrera and her associates concluded.
The investigators had no relevant financial disclosures. The study was supported in part through a grant from the National Institutes of Health/ National Cancer Institute. The research at the melanoma unit in Barcelona was partially funded by grants from the Spanish Fondo de Investigaciones Sanitarias and CIBER de Enfermedades Raras of the Instituto de Salud Carlos III, Spain; it was confinanced by numerous other grants.
SOURCE: Carrera C et al. J Am Acad Dermatol. 2018;78(2):278-88. doi: 10.1016/j.jaad.2017.09.065.
Adding dermoscopy to the classic and modified melanoma ABCD criteria will help speed detection of melanoma in children, said Cristina Carrera, MD, of the University of Barcelona, and her associates.
Pediatric melanomas make up less than 3% of pediatric cancers and 1%-4% of all melanomas, occurring more commonly in adolescents than in children aged 5-9 years. These cancers often are thicker when first seen because of delay in diagnosis and/or differences in growth dynamics. Delay in diagnosis may occur because of the low incidence of pediatric melanoma and because classic melanoma criteria – ABCD: asymmetry, border irregularity, multiple colors, diameter (6 mm) – don’t always apply, they said.
A modified clinical ABCD rule of ‘‘amelanotic, bleeding bump, color uniformity, and de novo lesion of any diameter’’ was proposed to improve early detection of pediatric melanoma. And because dermoscopy improves melanoma diagnosis in adults, researchers decided to examine clinical and dermascopic findings in a group of pediatric melanoma cases.
Fifty-two pediatric melanoma cases from 51 patients were collected from pigmented lesion clinics across nine countries (Australia, Belgium, Brazil, France, Israel, Italy, Serbia, Spain, and the United States), and clinical and dermoscopic specimens were examined by two experienced reviewers.
The mean age of the patients was 15 years, and 26 of the patients were female. Histopathologically, 28% of the pediatric melanomas were classified as spitzoid and 72% as nonspitzoid. Patients with a spitzoid melanoma were significantly younger than those with nonspitzoid melanoma (12.5 vs. 16 years, P = .004). All of the spitzoid tumors were invasive, and they were significantly thicker than the nonspitzoid melanomas (2.6 vs. 1.2 mm, P = .004). Also, these lesions more frequently were ulcerated than nonspitzoid melanomas (29% vs. 8%, P = .06). Melanomas associated with a nevus more often were among nonspitzoid than spitzoid melanomas (62% vs. 27% P = .02), and the congenital type of nevus was linked most often with nonspitzoid melanomas (25 of 27 [92.6%]), Dr. Carrera and her associates reported.
The pediatric melanomas in this study appeared most frequently on the lower extremities (31%), followed by the back (27%). Spitzoid melanomas most often appeared on the limbs (73%); nonspitzoid melanomas were likely to occur on the torso (52%).
In the majority of cases (52%), the overall clinical morphology was considered to fulfill the classic melanoma ABCD criteria. The remaining lesions that did not fulfill the conventional ABCD criteria were called benign-appearing tumors or nodular/polypoid tumors. Spitzoid melanomas most often were nodular/ polypoid (47%), while most nonspitzoid melanomas (59%) were melanoma-like. Only 21% of the pediatric melanomas fulfilled the modified melanoma ABCD criteria, they noted.
On dermoscopy, which was available in 49 cases, a multicomponent pattern seen in 24 cases mostly was associated with nonspitzoid melanoma (88%, P less than.03); a nevus-like pattern in 9 cases was seen only among nonspitzoid melanomas. A vascular pink Spitz-like pattern in seven cases mostly was associated with spitzoid melanomas (86%, P less than .002). A pigmented Reed-like pattern seen in seven cases occurred more often among spitzoid melanomas, but this did not reach statistical significance, the investigators wrote.
In terms of appearance, “red and white colors, milky red areas, polymorphous vessels, and shiny white structures were associated with spitzoid melanoma. Dark brown color, atypical network, and structureless areas were associated with nonspitzoid melanomas,” Dr. Carrera and her associates said.
Dermoscopic patterns that were pink spitzoid or Reed-like were more likely to be histopathologically classified as spitzoid. These pediatric melanomas were linked with younger age, occurrence on the limbs, and de novo development. On the other hand, dermoscopic patterns that were multicomponent or nevus-like were likely to be histopathologically classified as nonspitzoid. These lesions were linked with older age, fair skin phenotype, family history of melanoma, and a preexisting nevus, the researchers said.
“In contrast to previous reports, the present study highlights the fact that the majority of melanomas diagnosed in patients younger than 20 years look similar to melanomas found in adults. Nonspitzoid melanomas were associated with adolescence and with the presence of melanoma risk factors. All nonspitzoid melanomas displayed dermoscopic features associated with melanoma. Spitzoid melanomas were associated with younger age, location on the extremities, and nodular/polypoid clinical morphology. Dermoscopy of spitzoid melanomas revealed atypical vessels and shiny white lines (if amelanotic) or an asymmetric starburst pattern (if pigmented),” Dr. Carrera and her associates concluded.
The investigators had no relevant financial disclosures. The study was supported in part through a grant from the National Institutes of Health/ National Cancer Institute. The research at the melanoma unit in Barcelona was partially funded by grants from the Spanish Fondo de Investigaciones Sanitarias and CIBER de Enfermedades Raras of the Instituto de Salud Carlos III, Spain; it was confinanced by numerous other grants.
SOURCE: Carrera C et al. J Am Acad Dermatol. 2018;78(2):278-88. doi: 10.1016/j.jaad.2017.09.065.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Key clinical point:
Major finding: On dermoscopy, a multicomponent pattern seen in 24 cases mostly was associated with nonspitzoid melanoma (88%, P less than.03).
Data source: Fifty-two pediatric melanoma cases from 51 patients were collected from pigmented lesion clinics across 9 countries.
Disclosures: The investigators had no relevant financial disclosures. The study was supported in part through a grant from the National Institutes of Health/ National Cancer Institute. The research at the melanoma unit in Barcelona was partially funded by grants from the Spanish Fondo de Investigaciones Sanitarias and CIBER de Enfermedades Raras of the Instituto de Salud Carlos III, Spain; it was cofinanced by numerous other grants.
Source: Carrera C et al. J Am Acad Dermatol. 2018;78(2):278-88.
Melanoma incidence increased in older non-Hispanic whites
reported Dawn M. Holman, MPH, of the division of cancer prevention and control at the Centers for Disease Control and Prevention, and her coauthors.
In non-Hispanic white females aged 15 years and older, 131,976 melanomas were diagnosed between January 2010 and December 2014. In non-Hispanic white males, 192,979 melanomas were diagnosed during this time period. More than 70% of melanomas were diagnosed in patients aged 55 years or older, the authors reported. In females, melanoma incidence rates ranged from 4.5/100,000 population in those aged 15-24 years to 60.9/100,000 population in those aged 85 years and older. For males, melanoma incidence ranged from 2.0/100,000 population in those aged 15-24 years to 198.3/100,000 population in men aged 85 years and older.
Investigators analyzed data from the CDC National Program of Cancer Registries and the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program. Melanoma incidence rates and average annual counts by 10-year age groups were calculated from January 2010 to December 2014, as was average annual percent change (AAPC) by 10-year age groups from January 2005 to December 2014.
Overall, the increase in melanoma incidence was statistically significant for both males and females 15 years of age and older (AAPC, 1.4; P less than .05). However, melanoma incidence decreased significantly in younger patients aged 15-24 years, 25-34 years, and 35-44 years (AAPC, –5.1, –1.7, and –0.5 respectively; P less than .05), and increased significantly in those aged 55-64 years, 65-74 years, 75-84 years, and older than 85 years (AAPC, 1.3, 2.5, 3.6, and 4.6 respectively; P less than .05). The increase in melanoma incidence was statistically significant in men older than 54 years and in women older than 44 years, Ms. Holman and her associates reported in a research letter in JAMA Dermatology.
The findings suggest that recent decreases in indoor tanning and sunburn prevalence may account for the reduction in melanoma incidence over time, especially in adolescents and young adults, they said.
“Although primary skin cancer prevention efforts have often focused on children, adolescents, and young adults, the steady increase in melanoma incidence rates among older adults indicates a need for efforts that promote skin cancer preventive behaviors throughout adulthood. Such efforts could focus on groups at high risk, such as outdoor workers and intentional tanners,” Ms. Holman and her associates concluded.
The authors did not disclose any conflicts of interest. The study was partially supported by appointments to the CDC Research Participation Program through an interagency agreement with the U.S. Department of Energy.
SOURCE: Holman DM et al. JAMA Dermatol. 2018 Jan 31. doi: 10.1001/jamadermatol.2017.5541.
reported Dawn M. Holman, MPH, of the division of cancer prevention and control at the Centers for Disease Control and Prevention, and her coauthors.
In non-Hispanic white females aged 15 years and older, 131,976 melanomas were diagnosed between January 2010 and December 2014. In non-Hispanic white males, 192,979 melanomas were diagnosed during this time period. More than 70% of melanomas were diagnosed in patients aged 55 years or older, the authors reported. In females, melanoma incidence rates ranged from 4.5/100,000 population in those aged 15-24 years to 60.9/100,000 population in those aged 85 years and older. For males, melanoma incidence ranged from 2.0/100,000 population in those aged 15-24 years to 198.3/100,000 population in men aged 85 years and older.
Investigators analyzed data from the CDC National Program of Cancer Registries and the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program. Melanoma incidence rates and average annual counts by 10-year age groups were calculated from January 2010 to December 2014, as was average annual percent change (AAPC) by 10-year age groups from January 2005 to December 2014.
Overall, the increase in melanoma incidence was statistically significant for both males and females 15 years of age and older (AAPC, 1.4; P less than .05). However, melanoma incidence decreased significantly in younger patients aged 15-24 years, 25-34 years, and 35-44 years (AAPC, –5.1, –1.7, and –0.5 respectively; P less than .05), and increased significantly in those aged 55-64 years, 65-74 years, 75-84 years, and older than 85 years (AAPC, 1.3, 2.5, 3.6, and 4.6 respectively; P less than .05). The increase in melanoma incidence was statistically significant in men older than 54 years and in women older than 44 years, Ms. Holman and her associates reported in a research letter in JAMA Dermatology.
The findings suggest that recent decreases in indoor tanning and sunburn prevalence may account for the reduction in melanoma incidence over time, especially in adolescents and young adults, they said.
“Although primary skin cancer prevention efforts have often focused on children, adolescents, and young adults, the steady increase in melanoma incidence rates among older adults indicates a need for efforts that promote skin cancer preventive behaviors throughout adulthood. Such efforts could focus on groups at high risk, such as outdoor workers and intentional tanners,” Ms. Holman and her associates concluded.
The authors did not disclose any conflicts of interest. The study was partially supported by appointments to the CDC Research Participation Program through an interagency agreement with the U.S. Department of Energy.
SOURCE: Holman DM et al. JAMA Dermatol. 2018 Jan 31. doi: 10.1001/jamadermatol.2017.5541.
reported Dawn M. Holman, MPH, of the division of cancer prevention and control at the Centers for Disease Control and Prevention, and her coauthors.
In non-Hispanic white females aged 15 years and older, 131,976 melanomas were diagnosed between January 2010 and December 2014. In non-Hispanic white males, 192,979 melanomas were diagnosed during this time period. More than 70% of melanomas were diagnosed in patients aged 55 years or older, the authors reported. In females, melanoma incidence rates ranged from 4.5/100,000 population in those aged 15-24 years to 60.9/100,000 population in those aged 85 years and older. For males, melanoma incidence ranged from 2.0/100,000 population in those aged 15-24 years to 198.3/100,000 population in men aged 85 years and older.
Investigators analyzed data from the CDC National Program of Cancer Registries and the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program. Melanoma incidence rates and average annual counts by 10-year age groups were calculated from January 2010 to December 2014, as was average annual percent change (AAPC) by 10-year age groups from January 2005 to December 2014.
Overall, the increase in melanoma incidence was statistically significant for both males and females 15 years of age and older (AAPC, 1.4; P less than .05). However, melanoma incidence decreased significantly in younger patients aged 15-24 years, 25-34 years, and 35-44 years (AAPC, –5.1, –1.7, and –0.5 respectively; P less than .05), and increased significantly in those aged 55-64 years, 65-74 years, 75-84 years, and older than 85 years (AAPC, 1.3, 2.5, 3.6, and 4.6 respectively; P less than .05). The increase in melanoma incidence was statistically significant in men older than 54 years and in women older than 44 years, Ms. Holman and her associates reported in a research letter in JAMA Dermatology.
The findings suggest that recent decreases in indoor tanning and sunburn prevalence may account for the reduction in melanoma incidence over time, especially in adolescents and young adults, they said.
“Although primary skin cancer prevention efforts have often focused on children, adolescents, and young adults, the steady increase in melanoma incidence rates among older adults indicates a need for efforts that promote skin cancer preventive behaviors throughout adulthood. Such efforts could focus on groups at high risk, such as outdoor workers and intentional tanners,” Ms. Holman and her associates concluded.
The authors did not disclose any conflicts of interest. The study was partially supported by appointments to the CDC Research Participation Program through an interagency agreement with the U.S. Department of Energy.
SOURCE: Holman DM et al. JAMA Dermatol. 2018 Jan 31. doi: 10.1001/jamadermatol.2017.5541.
FROM JAMA DERMATOLOGY
Key clinical point: Melanoma incidence has increased in older non-Hispanic whites, but decreased in younger members of this population.
Major finding: Overall increase in melanoma incidence was statistically significant for both males and females 15 years of age and older (AAPC = 1.4; P less than .05).
Study details: An analysis of data from the CDC’s National Program of Cancer Registries and the National Cancer Institute’s Surveillance, Epidemiology, and End Results Program from January 2010 to December 2014 (for incidence and rates) and from January 2005 to December 2014 (for trends over time).
Disclosures: The authors did not disclose any conflicts of interest. The study was partially supported by appointments to the CDC Research Participation Program through an interagency agreement with the U.S. Department of Energy.
Source: Holman DM et al. JAMA Dermatol. 2018 Jan 31. doi: 10.1001/jamadermatol.2017.5541.
Be alert for BAP1 mutations in hereditary melanomas
MIAMI – Although rare, patients who present with one or more skin cancers characteristic of those associated with loss of the BAP1 tumor suppressor protein may be at elevated risk for more aggressive uveal melanomas and other cancers such as kidney cancer and mesothelioma. For this reason, dermatologists who recognize the lesions and telltale pattern of this inherited mutation within families can do a great service, encouraging education, genetic counseling, and referral of patients to a nearby cancer center, according to Hensin Tsao, MD, PhD.
“ Dr. Tsao said at the 2018 Orlando Dermatology Aesthetic and Clinical Conference.
The BAP1-associated skin lesions can emerge when patients are relatively young, even as teenagers. The melanoma and renal cell cancers also can have an early onset, said Dr. Tsao, director of the melanoma genetics program at Massachusetts General Hospital, Boston. The skin lesion itself can be a tipoff for a BAP1 germline mutation. In general, they are small, dome shaped – not flat like a superficial basal cell – rarely pigmented and appear “orangey translucent.” Dr. Tsao added: “When you start seeing them, you’ll recognize them. However, to be sure, you’re going to have to biopsy to know what is going on.”
In one patient he described, the pattern of malignancies in the patient’s family was a hint that she had a BAP1 mutation, Dr. Tsao said. The proband had melanoma starting at age 31 years, a squamous cell carcinoma at 35 years, and basal cell carcinoma at age 40 years. “She had nine ‘nevoid melanomas’ over the years. Nevoid melanomas are rare, and with nine in a row, you know something odd is going on.” Dr. Tsao and his team performed a series of sentinel lymph node biopsies that ruled out metastasis. “What is also interesting is the father had ocular melanoma, which is what got us to thinking about BAP1 mutations in this family.” A sister who developed melanoma and a brother who also was diagnosed with melanoma plus kidney cancer at age 45 years were further clues to the germline mutation.
No longer ‘condemned proteins’
Under normal circumstances, BAP1 is a tumor suppressor protein involved in cellular process called “ubiquitination.” Often, ubiquitination serves to identify proteins “condemned” for destruction by the proteasome system. The BAP1 protein acts through a molecular relay and removes ubiquitin polypeptide groups on the protein. “In the absence of BAP1, proteins often linger longer because they accumulate ubiquitin groups, or alternatively, the protein’s function is somehow altered by mechanisms we don’t quite understand yet,” Dr. Tsao explained.
Once a dermatologist suspects a BAP1 mutation–associated cancer, they can order a BAP1 nuclear stain to confirm diagnosis. Formal documentation of a germline mutation, however, requires genetic testing of blood DNA.
A family history lesson
Ask patients not only about history of melanoma in their family, including if any close relative was diagnosed with eye melanoma, Dr. Tsao suggested. “We had an opportunity to look at cutaneous and ocular melanoma families. Overall, if your family has an ocular melanoma along with cutaneous melanoma, the risk of being a BAP1 mutation–bearing family is greater.” In addition, he and his colleagues did a case control study with Ivana K. Kim, MD, at the Massachusetts Eye and Ear Infirmary in Boston, and found people with metastatic ocular melanoma were more likely to have BAP1 mutations, compared with those with nonmetastatic ocular melanoma.
“The fear is, of course, patient who are BAP1 mutation carriers might be predisposed to more lethal variants of uveal melanoma.”
Although taking a family history is essential, some patients may be unfamiliar with mesothelioma. “So ask about any unusual lung cancers or eye cancers,” Dr. Tsao suggested. “And if it looks like there is an aggregation of rare tumors, get them to a nearby cancer center [for further work-up]. Mesothelioma is difficult to treat and a horrible disease,” he added. “So if there is any chance you can [catch] the mesothelioma early, that’s good.”
He also cautioned against over interpretation of patient reports about family malignancies, in part because lung and breast cancers are relatively common. “Sometimes, when you see a family with lung or breast cancers, it could just be a chance association since these are quite common in the general population.” In other words, determining if a lung cancer in a family with melanoma is an association beyond chance can take some “pretty large numbers to prove.”
In contrast, “the number of kidney cancers among BAP1 families I do believe are out of proportion with normal population expectations,” Dr. Tsao added.
Follow-up and genetic counseling
There is no standard protocol for follow-up once a patient is identified with a BAP1 mutation. “I refer them for uveal, kidney, and/or lung cancer evaluation and see them back two to four times a year for skin checks.”
A meeting attendee suggested that management of a patient with melanoma might not differ based on genetic-testing results. “I agree with you that I don’t need to know the genetic status within these families to help with their cutaneous melanomas,” Dr. Tsao replied. “But the question becomes, are there other internal malignancies you’re not screening for appropriately?”
Another attendee asked about genetic counseling. “I encourage genetic counseling since dermatologists often don’t have time to take at detailed family history of all cancers and ages of onset,” Dr. Tsao said. “Genetic counselors can help sort out the strength of the genetic pedigree in a family. My residents usually ask if someone has a history of melanoma in their family, and that’s it. But there is a big difference between having a cousin with melanoma and three brothers with melanoma.”
MIAMI – Although rare, patients who present with one or more skin cancers characteristic of those associated with loss of the BAP1 tumor suppressor protein may be at elevated risk for more aggressive uveal melanomas and other cancers such as kidney cancer and mesothelioma. For this reason, dermatologists who recognize the lesions and telltale pattern of this inherited mutation within families can do a great service, encouraging education, genetic counseling, and referral of patients to a nearby cancer center, according to Hensin Tsao, MD, PhD.
“ Dr. Tsao said at the 2018 Orlando Dermatology Aesthetic and Clinical Conference.
The BAP1-associated skin lesions can emerge when patients are relatively young, even as teenagers. The melanoma and renal cell cancers also can have an early onset, said Dr. Tsao, director of the melanoma genetics program at Massachusetts General Hospital, Boston. The skin lesion itself can be a tipoff for a BAP1 germline mutation. In general, they are small, dome shaped – not flat like a superficial basal cell – rarely pigmented and appear “orangey translucent.” Dr. Tsao added: “When you start seeing them, you’ll recognize them. However, to be sure, you’re going to have to biopsy to know what is going on.”
In one patient he described, the pattern of malignancies in the patient’s family was a hint that she had a BAP1 mutation, Dr. Tsao said. The proband had melanoma starting at age 31 years, a squamous cell carcinoma at 35 years, and basal cell carcinoma at age 40 years. “She had nine ‘nevoid melanomas’ over the years. Nevoid melanomas are rare, and with nine in a row, you know something odd is going on.” Dr. Tsao and his team performed a series of sentinel lymph node biopsies that ruled out metastasis. “What is also interesting is the father had ocular melanoma, which is what got us to thinking about BAP1 mutations in this family.” A sister who developed melanoma and a brother who also was diagnosed with melanoma plus kidney cancer at age 45 years were further clues to the germline mutation.
No longer ‘condemned proteins’
Under normal circumstances, BAP1 is a tumor suppressor protein involved in cellular process called “ubiquitination.” Often, ubiquitination serves to identify proteins “condemned” for destruction by the proteasome system. The BAP1 protein acts through a molecular relay and removes ubiquitin polypeptide groups on the protein. “In the absence of BAP1, proteins often linger longer because they accumulate ubiquitin groups, or alternatively, the protein’s function is somehow altered by mechanisms we don’t quite understand yet,” Dr. Tsao explained.
Once a dermatologist suspects a BAP1 mutation–associated cancer, they can order a BAP1 nuclear stain to confirm diagnosis. Formal documentation of a germline mutation, however, requires genetic testing of blood DNA.
A family history lesson
Ask patients not only about history of melanoma in their family, including if any close relative was diagnosed with eye melanoma, Dr. Tsao suggested. “We had an opportunity to look at cutaneous and ocular melanoma families. Overall, if your family has an ocular melanoma along with cutaneous melanoma, the risk of being a BAP1 mutation–bearing family is greater.” In addition, he and his colleagues did a case control study with Ivana K. Kim, MD, at the Massachusetts Eye and Ear Infirmary in Boston, and found people with metastatic ocular melanoma were more likely to have BAP1 mutations, compared with those with nonmetastatic ocular melanoma.
“The fear is, of course, patient who are BAP1 mutation carriers might be predisposed to more lethal variants of uveal melanoma.”
Although taking a family history is essential, some patients may be unfamiliar with mesothelioma. “So ask about any unusual lung cancers or eye cancers,” Dr. Tsao suggested. “And if it looks like there is an aggregation of rare tumors, get them to a nearby cancer center [for further work-up]. Mesothelioma is difficult to treat and a horrible disease,” he added. “So if there is any chance you can [catch] the mesothelioma early, that’s good.”
He also cautioned against over interpretation of patient reports about family malignancies, in part because lung and breast cancers are relatively common. “Sometimes, when you see a family with lung or breast cancers, it could just be a chance association since these are quite common in the general population.” In other words, determining if a lung cancer in a family with melanoma is an association beyond chance can take some “pretty large numbers to prove.”
In contrast, “the number of kidney cancers among BAP1 families I do believe are out of proportion with normal population expectations,” Dr. Tsao added.
Follow-up and genetic counseling
There is no standard protocol for follow-up once a patient is identified with a BAP1 mutation. “I refer them for uveal, kidney, and/or lung cancer evaluation and see them back two to four times a year for skin checks.”
A meeting attendee suggested that management of a patient with melanoma might not differ based on genetic-testing results. “I agree with you that I don’t need to know the genetic status within these families to help with their cutaneous melanomas,” Dr. Tsao replied. “But the question becomes, are there other internal malignancies you’re not screening for appropriately?”
Another attendee asked about genetic counseling. “I encourage genetic counseling since dermatologists often don’t have time to take at detailed family history of all cancers and ages of onset,” Dr. Tsao said. “Genetic counselors can help sort out the strength of the genetic pedigree in a family. My residents usually ask if someone has a history of melanoma in their family, and that’s it. But there is a big difference between having a cousin with melanoma and three brothers with melanoma.”
MIAMI – Although rare, patients who present with one or more skin cancers characteristic of those associated with loss of the BAP1 tumor suppressor protein may be at elevated risk for more aggressive uveal melanomas and other cancers such as kidney cancer and mesothelioma. For this reason, dermatologists who recognize the lesions and telltale pattern of this inherited mutation within families can do a great service, encouraging education, genetic counseling, and referral of patients to a nearby cancer center, according to Hensin Tsao, MD, PhD.
“ Dr. Tsao said at the 2018 Orlando Dermatology Aesthetic and Clinical Conference.
The BAP1-associated skin lesions can emerge when patients are relatively young, even as teenagers. The melanoma and renal cell cancers also can have an early onset, said Dr. Tsao, director of the melanoma genetics program at Massachusetts General Hospital, Boston. The skin lesion itself can be a tipoff for a BAP1 germline mutation. In general, they are small, dome shaped – not flat like a superficial basal cell – rarely pigmented and appear “orangey translucent.” Dr. Tsao added: “When you start seeing them, you’ll recognize them. However, to be sure, you’re going to have to biopsy to know what is going on.”
In one patient he described, the pattern of malignancies in the patient’s family was a hint that she had a BAP1 mutation, Dr. Tsao said. The proband had melanoma starting at age 31 years, a squamous cell carcinoma at 35 years, and basal cell carcinoma at age 40 years. “She had nine ‘nevoid melanomas’ over the years. Nevoid melanomas are rare, and with nine in a row, you know something odd is going on.” Dr. Tsao and his team performed a series of sentinel lymph node biopsies that ruled out metastasis. “What is also interesting is the father had ocular melanoma, which is what got us to thinking about BAP1 mutations in this family.” A sister who developed melanoma and a brother who also was diagnosed with melanoma plus kidney cancer at age 45 years were further clues to the germline mutation.
No longer ‘condemned proteins’
Under normal circumstances, BAP1 is a tumor suppressor protein involved in cellular process called “ubiquitination.” Often, ubiquitination serves to identify proteins “condemned” for destruction by the proteasome system. The BAP1 protein acts through a molecular relay and removes ubiquitin polypeptide groups on the protein. “In the absence of BAP1, proteins often linger longer because they accumulate ubiquitin groups, or alternatively, the protein’s function is somehow altered by mechanisms we don’t quite understand yet,” Dr. Tsao explained.
Once a dermatologist suspects a BAP1 mutation–associated cancer, they can order a BAP1 nuclear stain to confirm diagnosis. Formal documentation of a germline mutation, however, requires genetic testing of blood DNA.
A family history lesson
Ask patients not only about history of melanoma in their family, including if any close relative was diagnosed with eye melanoma, Dr. Tsao suggested. “We had an opportunity to look at cutaneous and ocular melanoma families. Overall, if your family has an ocular melanoma along with cutaneous melanoma, the risk of being a BAP1 mutation–bearing family is greater.” In addition, he and his colleagues did a case control study with Ivana K. Kim, MD, at the Massachusetts Eye and Ear Infirmary in Boston, and found people with metastatic ocular melanoma were more likely to have BAP1 mutations, compared with those with nonmetastatic ocular melanoma.
“The fear is, of course, patient who are BAP1 mutation carriers might be predisposed to more lethal variants of uveal melanoma.”
Although taking a family history is essential, some patients may be unfamiliar with mesothelioma. “So ask about any unusual lung cancers or eye cancers,” Dr. Tsao suggested. “And if it looks like there is an aggregation of rare tumors, get them to a nearby cancer center [for further work-up]. Mesothelioma is difficult to treat and a horrible disease,” he added. “So if there is any chance you can [catch] the mesothelioma early, that’s good.”
He also cautioned against over interpretation of patient reports about family malignancies, in part because lung and breast cancers are relatively common. “Sometimes, when you see a family with lung or breast cancers, it could just be a chance association since these are quite common in the general population.” In other words, determining if a lung cancer in a family with melanoma is an association beyond chance can take some “pretty large numbers to prove.”
In contrast, “the number of kidney cancers among BAP1 families I do believe are out of proportion with normal population expectations,” Dr. Tsao added.
Follow-up and genetic counseling
There is no standard protocol for follow-up once a patient is identified with a BAP1 mutation. “I refer them for uveal, kidney, and/or lung cancer evaluation and see them back two to four times a year for skin checks.”
A meeting attendee suggested that management of a patient with melanoma might not differ based on genetic-testing results. “I agree with you that I don’t need to know the genetic status within these families to help with their cutaneous melanomas,” Dr. Tsao replied. “But the question becomes, are there other internal malignancies you’re not screening for appropriately?”
Another attendee asked about genetic counseling. “I encourage genetic counseling since dermatologists often don’t have time to take at detailed family history of all cancers and ages of onset,” Dr. Tsao said. “Genetic counselors can help sort out the strength of the genetic pedigree in a family. My residents usually ask if someone has a history of melanoma in their family, and that’s it. But there is a big difference between having a cousin with melanoma and three brothers with melanoma.”
REPORTING FROM ODAC 2018
Mobile Medical Apps for Patient Education: A Graded Review of Available Dermatology Apps
According to industry estimates, roughly 64% of US adults were smartphone users in 2015.1 Smartphones enable users to utilize mobile applications (apps) that can perform a variety of functions in many categories, including business, music, photography, entertainment, education, social networking, travel, and lifestyle. The widespread adoption and use of mobile apps has implications for medical practice. Mobile apps have the capability to serve as information sources for patients, educational tools for students, and diagnostic aids for physicians.2 Consequently, a number of medical and health care–oriented apps have already been developed3 and are increasingly utilized by patients and providers.4
Given its visual nature, dermatology is particularly amenable to the integration of mobile medical apps. A study by Brewer et al5 identified more than 229 dermatology-related apps in categories ranging from general dermatology reference, self-surveillance and diagnosis, disease guides, educational aids, sunscreen and UV recommendations, and teledermatology. Patients served as the target audience and principal consumers of more than half of these dermatology apps.5
Mobile medical and health care apps demonstrate great potential for serving as valuable information sources for patients with dermatologic conditions; however, the content, functions, accuracy, and educational value of dermatology mobile apps are not well characterized, making it difficult for patients and health care providers to select and recommend appropriate apps.6 In this study, we created a rubric to objectively grade 44 publicly available mobile dermatology apps with the primary focus of patient education.
Methods
We conducted a search of dermatology-related educational mobile apps that were publicly available via the App Store (Apple Inc) from January 2016 to November 2016. (The pricing, availability, and other features of these apps may have changed since the study period.) The following search terms were used: dermatology, dermoscopy, melanoma, skin cancer, psoriasis, rosacea, acne, eczema, dermal fillers, and Mohs surgery. We excluded apps that were not in English; had a solely commercial focus; were mobile textbooks or scientific journals; were used to provide teledermatology services with no educational purpose; were solely focused on homeopathic, alternative, and/or complementary medicine; or were intended primarily as a reference for students or health care professionals. Our search yielded 44 apps with patient education as a primary objective. The apps were divided into 6 categories based on their focus: general dermatology, cosmetic dermatology, acne, eczema, psoriasis, and skin cancer.
Each app was reviewed using a quantified grading rubric developed by the researchers. In a prior evaluation, Handel7 reviewed 35 health and wellness mobile apps utilizing the categories of ease of use, reliability, quality, scope of information, and aesthetics.4 These criteria were modified and adapted for the purposes of this study, and a 4-point scale was applied to each criterion. The final criteria were (1) educational objectives, (2) content, (3) accuracy, (4) design, and (5) conflict of interest. The quantified grading rubric is described in Table 1.
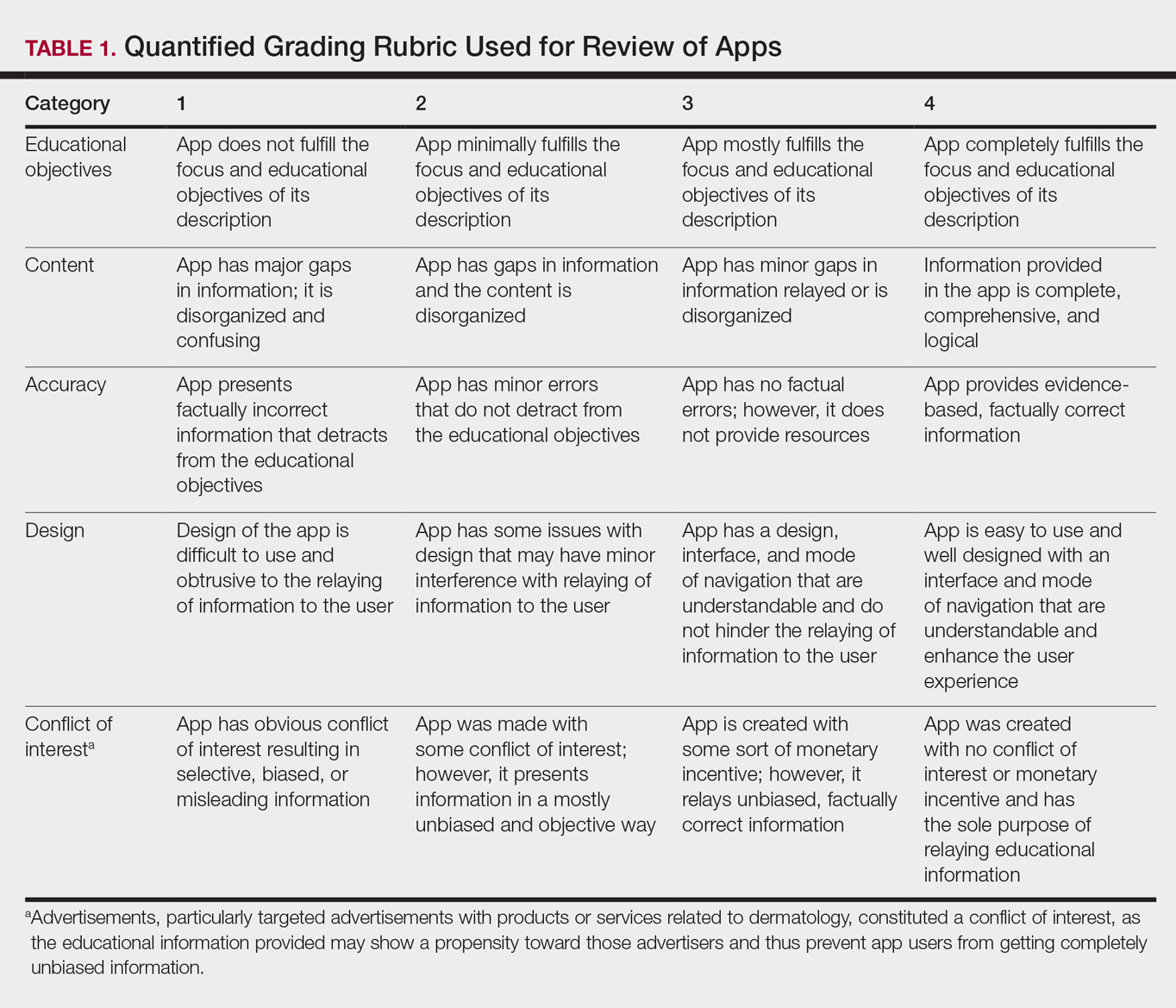
Results
The possible range of scores based on the grading rubric was 5 to 20. The actual range of scores was 8 to 19 (Table 2). The 44 reviewed apps were categorized by topic as acne, cosmetic dermatology, eczema, general dermatology, psoriasis, or skin cancer. A sample of 15 apps selected to represent the distribution of scores and their grading on the rubric are presented in Table 3.
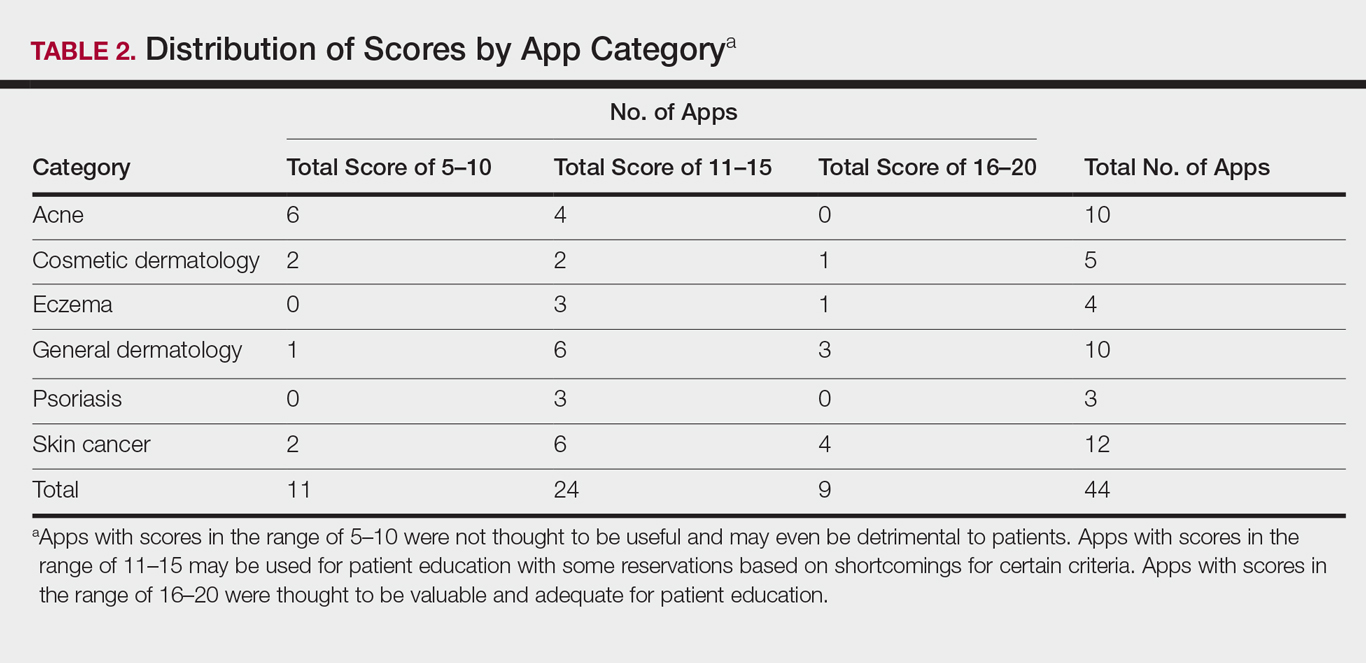
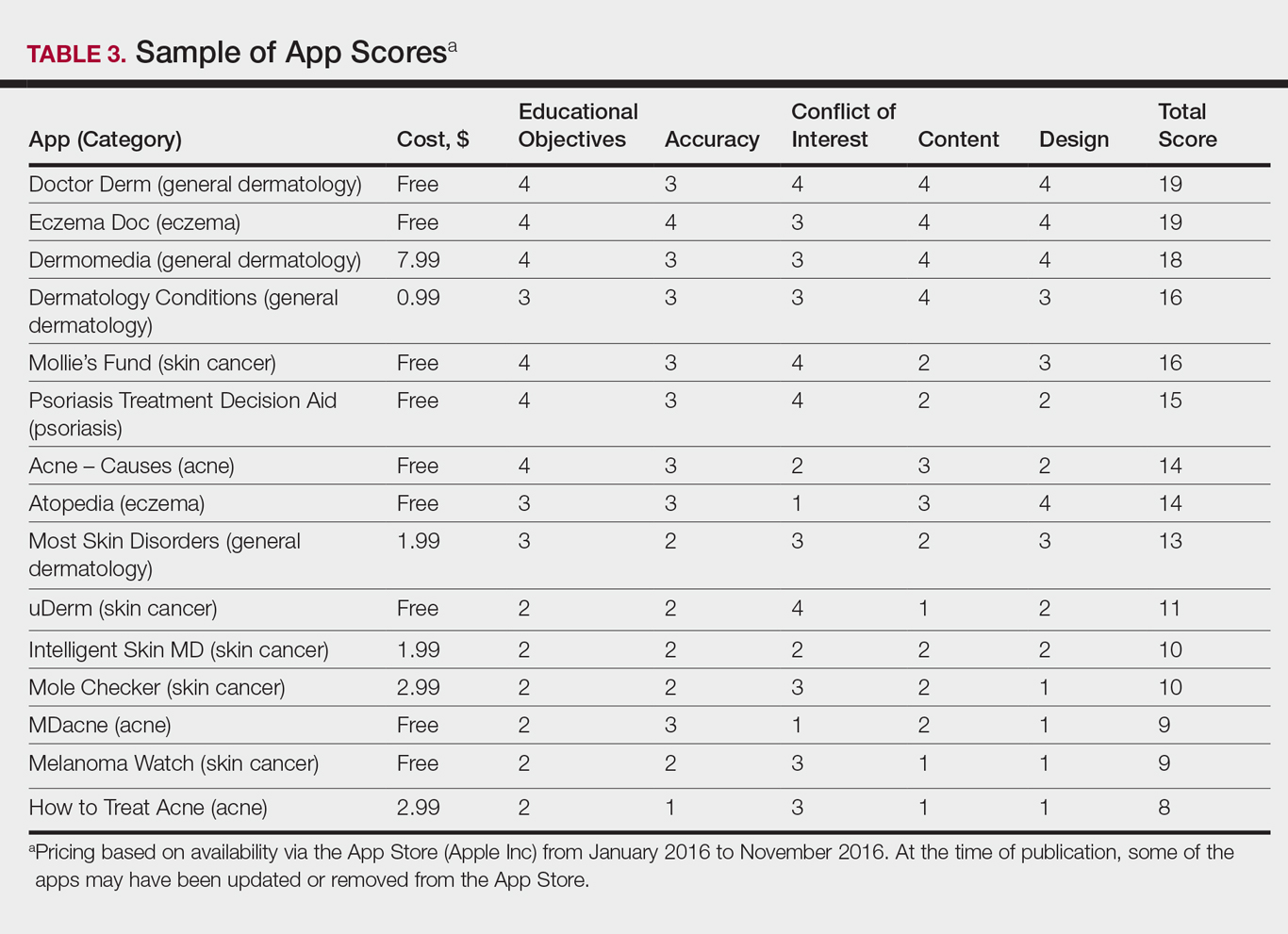
Comment
The number of dermatology-related apps available to mobile users continues to grow at an increasing rate.8 The apps vary in many aspects, including their purpose, scope, intended audience, and goals of the app publisher. In turn, more individuals are turning to mobile apps for medical information,4 especially in dermatology, thus it is necessary to create a systematic way to evaluate the quality and utility of each app to assist users in making informed decisions about which apps will best meet their needs in the midst of a wide array of choices.
For the purpose of this study, an objective rubric was created that can be used to evaluate the quality of medical apps for patient education in dermatology. An app’s adequacy and usefulness for patient education was thought to depend on 3 possible score ranges into which the app could fall based on the grading rubric. An app with a total score in the range of 5 to 10 was not thought to be useful and may even be detrimental to patients. An app with a total score in the range of 11 to 15 may be used for patient education with some reservations based on shortcomings for certain criteria. An app with a score in the range of 16 to 20 was thought to be valuable and adequate for patient education. For example, the How to Treat Acne app received a total score of 8 and therefore would not be recommended to patients based on the grading rubric used in this study. This particular app provided sparse and sometimes inaccurate information, had a confusing user interface, and contained many obstructive advertisements. In contrast, the Eczema Doc app received a total score of 19, which indicates a quality app deemed to be useful for patient information based on the established rubric. This app met all the objectives that it advertised, contained accurate information with verified citation of sources, and was very easy for users to navigate.
Of the 44 graded apps, only 9 (20.5%) received scores in the highest range of 16 to 20, which indicates a need for improvements in mobile dermatology apps intended for patient education. Adopting the grading rubric developed in this study as a standard in the creation of medical apps could have beneficial implications in disseminating accurate, safe, unbiased, and easy-to-understand information to patients.
- Smith A. U.S. smartphone use in 2015. Pew Research Center website. http://www.pewinternet.org/2015/04/01/us-smartphone-use-in-2015. Published April 1, 2015. Accessed August 29, 2017.
- Nilsen W, Kumar S, Shar A, et al. Advancing the science of mHealth. J Health Commun. 2012;17(suppl 1):5-10.
- West DM. How mobile devices are transforming healthcare issues in technology innovation. Issues Technol Innov. 2012;18:1-14.
- Boudreaux ED, Waring ME, Hayes RB, et al. Evaluating and selecting mobile health apps: strategies for healthcare providers and healthcare organizations. Transl Behav Med. 2014;4:363-371.
- Brewer AC, Endly DC, Henley J, et al. Mobile applications in dermatology. JAMA Dermatol. 2013;149:1300-1304.
- Cummings E, Borycki E, Roehrer E. Issues and considerations for healthcare consumers using mobile applications. Stud Health Technol Inform. 2013;183:227-231.
- Handel MJ. mHealth (mobile health)-using apps for health and wellness. Explore. 2011;7:256-261.
- Boulos MN, Brewer AC, Karimkhani C, et al. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J Public Health Inform. 2014;5:229.
According to industry estimates, roughly 64% of US adults were smartphone users in 2015.1 Smartphones enable users to utilize mobile applications (apps) that can perform a variety of functions in many categories, including business, music, photography, entertainment, education, social networking, travel, and lifestyle. The widespread adoption and use of mobile apps has implications for medical practice. Mobile apps have the capability to serve as information sources for patients, educational tools for students, and diagnostic aids for physicians.2 Consequently, a number of medical and health care–oriented apps have already been developed3 and are increasingly utilized by patients and providers.4
Given its visual nature, dermatology is particularly amenable to the integration of mobile medical apps. A study by Brewer et al5 identified more than 229 dermatology-related apps in categories ranging from general dermatology reference, self-surveillance and diagnosis, disease guides, educational aids, sunscreen and UV recommendations, and teledermatology. Patients served as the target audience and principal consumers of more than half of these dermatology apps.5
Mobile medical and health care apps demonstrate great potential for serving as valuable information sources for patients with dermatologic conditions; however, the content, functions, accuracy, and educational value of dermatology mobile apps are not well characterized, making it difficult for patients and health care providers to select and recommend appropriate apps.6 In this study, we created a rubric to objectively grade 44 publicly available mobile dermatology apps with the primary focus of patient education.
Methods
We conducted a search of dermatology-related educational mobile apps that were publicly available via the App Store (Apple Inc) from January 2016 to November 2016. (The pricing, availability, and other features of these apps may have changed since the study period.) The following search terms were used: dermatology, dermoscopy, melanoma, skin cancer, psoriasis, rosacea, acne, eczema, dermal fillers, and Mohs surgery. We excluded apps that were not in English; had a solely commercial focus; were mobile textbooks or scientific journals; were used to provide teledermatology services with no educational purpose; were solely focused on homeopathic, alternative, and/or complementary medicine; or were intended primarily as a reference for students or health care professionals. Our search yielded 44 apps with patient education as a primary objective. The apps were divided into 6 categories based on their focus: general dermatology, cosmetic dermatology, acne, eczema, psoriasis, and skin cancer.
Each app was reviewed using a quantified grading rubric developed by the researchers. In a prior evaluation, Handel7 reviewed 35 health and wellness mobile apps utilizing the categories of ease of use, reliability, quality, scope of information, and aesthetics.4 These criteria were modified and adapted for the purposes of this study, and a 4-point scale was applied to each criterion. The final criteria were (1) educational objectives, (2) content, (3) accuracy, (4) design, and (5) conflict of interest. The quantified grading rubric is described in Table 1.

Results
The possible range of scores based on the grading rubric was 5 to 20. The actual range of scores was 8 to 19 (Table 2). The 44 reviewed apps were categorized by topic as acne, cosmetic dermatology, eczema, general dermatology, psoriasis, or skin cancer. A sample of 15 apps selected to represent the distribution of scores and their grading on the rubric are presented in Table 3.


Comment
The number of dermatology-related apps available to mobile users continues to grow at an increasing rate.8 The apps vary in many aspects, including their purpose, scope, intended audience, and goals of the app publisher. In turn, more individuals are turning to mobile apps for medical information,4 especially in dermatology, thus it is necessary to create a systematic way to evaluate the quality and utility of each app to assist users in making informed decisions about which apps will best meet their needs in the midst of a wide array of choices.
For the purpose of this study, an objective rubric was created that can be used to evaluate the quality of medical apps for patient education in dermatology. An app’s adequacy and usefulness for patient education was thought to depend on 3 possible score ranges into which the app could fall based on the grading rubric. An app with a total score in the range of 5 to 10 was not thought to be useful and may even be detrimental to patients. An app with a total score in the range of 11 to 15 may be used for patient education with some reservations based on shortcomings for certain criteria. An app with a score in the range of 16 to 20 was thought to be valuable and adequate for patient education. For example, the How to Treat Acne app received a total score of 8 and therefore would not be recommended to patients based on the grading rubric used in this study. This particular app provided sparse and sometimes inaccurate information, had a confusing user interface, and contained many obstructive advertisements. In contrast, the Eczema Doc app received a total score of 19, which indicates a quality app deemed to be useful for patient information based on the established rubric. This app met all the objectives that it advertised, contained accurate information with verified citation of sources, and was very easy for users to navigate.
Of the 44 graded apps, only 9 (20.5%) received scores in the highest range of 16 to 20, which indicates a need for improvements in mobile dermatology apps intended for patient education. Adopting the grading rubric developed in this study as a standard in the creation of medical apps could have beneficial implications in disseminating accurate, safe, unbiased, and easy-to-understand information to patients.
According to industry estimates, roughly 64% of US adults were smartphone users in 2015.1 Smartphones enable users to utilize mobile applications (apps) that can perform a variety of functions in many categories, including business, music, photography, entertainment, education, social networking, travel, and lifestyle. The widespread adoption and use of mobile apps has implications for medical practice. Mobile apps have the capability to serve as information sources for patients, educational tools for students, and diagnostic aids for physicians.2 Consequently, a number of medical and health care–oriented apps have already been developed3 and are increasingly utilized by patients and providers.4
Given its visual nature, dermatology is particularly amenable to the integration of mobile medical apps. A study by Brewer et al5 identified more than 229 dermatology-related apps in categories ranging from general dermatology reference, self-surveillance and diagnosis, disease guides, educational aids, sunscreen and UV recommendations, and teledermatology. Patients served as the target audience and principal consumers of more than half of these dermatology apps.5
Mobile medical and health care apps demonstrate great potential for serving as valuable information sources for patients with dermatologic conditions; however, the content, functions, accuracy, and educational value of dermatology mobile apps are not well characterized, making it difficult for patients and health care providers to select and recommend appropriate apps.6 In this study, we created a rubric to objectively grade 44 publicly available mobile dermatology apps with the primary focus of patient education.
Methods
We conducted a search of dermatology-related educational mobile apps that were publicly available via the App Store (Apple Inc) from January 2016 to November 2016. (The pricing, availability, and other features of these apps may have changed since the study period.) The following search terms were used: dermatology, dermoscopy, melanoma, skin cancer, psoriasis, rosacea, acne, eczema, dermal fillers, and Mohs surgery. We excluded apps that were not in English; had a solely commercial focus; were mobile textbooks or scientific journals; were used to provide teledermatology services with no educational purpose; were solely focused on homeopathic, alternative, and/or complementary medicine; or were intended primarily as a reference for students or health care professionals. Our search yielded 44 apps with patient education as a primary objective. The apps were divided into 6 categories based on their focus: general dermatology, cosmetic dermatology, acne, eczema, psoriasis, and skin cancer.
Each app was reviewed using a quantified grading rubric developed by the researchers. In a prior evaluation, Handel7 reviewed 35 health and wellness mobile apps utilizing the categories of ease of use, reliability, quality, scope of information, and aesthetics.4 These criteria were modified and adapted for the purposes of this study, and a 4-point scale was applied to each criterion. The final criteria were (1) educational objectives, (2) content, (3) accuracy, (4) design, and (5) conflict of interest. The quantified grading rubric is described in Table 1.

Results
The possible range of scores based on the grading rubric was 5 to 20. The actual range of scores was 8 to 19 (Table 2). The 44 reviewed apps were categorized by topic as acne, cosmetic dermatology, eczema, general dermatology, psoriasis, or skin cancer. A sample of 15 apps selected to represent the distribution of scores and their grading on the rubric are presented in Table 3.


Comment
The number of dermatology-related apps available to mobile users continues to grow at an increasing rate.8 The apps vary in many aspects, including their purpose, scope, intended audience, and goals of the app publisher. In turn, more individuals are turning to mobile apps for medical information,4 especially in dermatology, thus it is necessary to create a systematic way to evaluate the quality and utility of each app to assist users in making informed decisions about which apps will best meet their needs in the midst of a wide array of choices.
For the purpose of this study, an objective rubric was created that can be used to evaluate the quality of medical apps for patient education in dermatology. An app’s adequacy and usefulness for patient education was thought to depend on 3 possible score ranges into which the app could fall based on the grading rubric. An app with a total score in the range of 5 to 10 was not thought to be useful and may even be detrimental to patients. An app with a total score in the range of 11 to 15 may be used for patient education with some reservations based on shortcomings for certain criteria. An app with a score in the range of 16 to 20 was thought to be valuable and adequate for patient education. For example, the How to Treat Acne app received a total score of 8 and therefore would not be recommended to patients based on the grading rubric used in this study. This particular app provided sparse and sometimes inaccurate information, had a confusing user interface, and contained many obstructive advertisements. In contrast, the Eczema Doc app received a total score of 19, which indicates a quality app deemed to be useful for patient information based on the established rubric. This app met all the objectives that it advertised, contained accurate information with verified citation of sources, and was very easy for users to navigate.
Of the 44 graded apps, only 9 (20.5%) received scores in the highest range of 16 to 20, which indicates a need for improvements in mobile dermatology apps intended for patient education. Adopting the grading rubric developed in this study as a standard in the creation of medical apps could have beneficial implications in disseminating accurate, safe, unbiased, and easy-to-understand information to patients.
- Smith A. U.S. smartphone use in 2015. Pew Research Center website. http://www.pewinternet.org/2015/04/01/us-smartphone-use-in-2015. Published April 1, 2015. Accessed August 29, 2017.
- Nilsen W, Kumar S, Shar A, et al. Advancing the science of mHealth. J Health Commun. 2012;17(suppl 1):5-10.
- West DM. How mobile devices are transforming healthcare issues in technology innovation. Issues Technol Innov. 2012;18:1-14.
- Boudreaux ED, Waring ME, Hayes RB, et al. Evaluating and selecting mobile health apps: strategies for healthcare providers and healthcare organizations. Transl Behav Med. 2014;4:363-371.
- Brewer AC, Endly DC, Henley J, et al. Mobile applications in dermatology. JAMA Dermatol. 2013;149:1300-1304.
- Cummings E, Borycki E, Roehrer E. Issues and considerations for healthcare consumers using mobile applications. Stud Health Technol Inform. 2013;183:227-231.
- Handel MJ. mHealth (mobile health)-using apps for health and wellness. Explore. 2011;7:256-261.
- Boulos MN, Brewer AC, Karimkhani C, et al. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J Public Health Inform. 2014;5:229.
- Smith A. U.S. smartphone use in 2015. Pew Research Center website. http://www.pewinternet.org/2015/04/01/us-smartphone-use-in-2015. Published April 1, 2015. Accessed August 29, 2017.
- Nilsen W, Kumar S, Shar A, et al. Advancing the science of mHealth. J Health Commun. 2012;17(suppl 1):5-10.
- West DM. How mobile devices are transforming healthcare issues in technology innovation. Issues Technol Innov. 2012;18:1-14.
- Boudreaux ED, Waring ME, Hayes RB, et al. Evaluating and selecting mobile health apps: strategies for healthcare providers and healthcare organizations. Transl Behav Med. 2014;4:363-371.
- Brewer AC, Endly DC, Henley J, et al. Mobile applications in dermatology. JAMA Dermatol. 2013;149:1300-1304.
- Cummings E, Borycki E, Roehrer E. Issues and considerations for healthcare consumers using mobile applications. Stud Health Technol Inform. 2013;183:227-231.
- Handel MJ. mHealth (mobile health)-using apps for health and wellness. Explore. 2011;7:256-261.
- Boulos MN, Brewer AC, Karimkhani C, et al. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J Public Health Inform. 2014;5:229.
Practice Points
- Mobile dermatology apps for educational purposes should be objectively reviewed before being used by patients.
- In our study, only 9 (20.5%) of the 44 dermatology apps evaluated were considered adequate for patient information based on our grading criteria.
Neoadjuvant dabrafenib and trametinib improves event-free survival in resectable melanoma
For patients with surgically resectable, BRAF-mutated melanoma, neoadjuvant and adjuvant treatment with the combination of dabrafenib and trametinib resulted in significantly longer event-free survival compared with standard care, according to results of a randomized study.
The trial was closed early because of the “large difference” in event-free survival favoring the neoadjuvant approach, the authors wrote (Lancet Oncol. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
While early closure limits interpretation of results, they do provide important proof-of-concept and data for future studies, wrote the authors, led by Rodabe N Amaria, MD, of the department of medical oncology, University of Texas MD Anderson Cancer Center, Houston.
“The clinical and translational results strongly support the rationale for further assessment of neoadjuvant therapy in patients with high-risk, surgically resectable melanoma,” Dr. Amaria and colleagues said in the report on the randomized trial, believed to be the first to evaluate the role of neoadjuvant therapy versus standard care in BRAF-mutated melanoma.
Dabrafenib and trametinib combination therapy is approved as a treatment for patients with unresectable or metastatic stage IV melanoma and a BRAFV600 mutation, which is found in about half of cutaneous melanomas, authors said.
To evaluate the combination in earlier stage disease, Dr. Amaria and coinvestigators conducted a single-center, open-label, randomized, phase 2 trial of 21 patients with surgically resectable clinical stage III or oligometastatic stage IV melanoma with BRAFV600E or BRAFV600K mutations.
Patients were randomized 2:1 to receive the neoadjuvant/adjuvant treatment or to standard of care, which consisted of standard surgery plus consideration for adjuvant therapy, the authors said. Those patients assigned to the targeted therapy arm received 8 weeks of neoadjuvant dabrafenib and trametinib followed by surgery, then adjuvant dabrafenib and trametinib for up to 44 weeks.
Event-free survival, the primary endpoint of the trial, was a median of 19.7 months for neoadjuvant plus adjuvant dabrafenib and trametinib, versus 2.9 months for standard care (P less than .0001), the investigators reported.
Dabrafenib and trametinib combination therapy was well tolerated as neoadjuvant and adjuvant therapy, with no grade 4 adverse events or treatment related deaths, according to the investigators. The most common grade 3 adverse event seen with the combination was diarrhea, occurring in 2 patients (15%).
The trial is continuing as a single-arm study of neoadjuvant plus adjuvant dabrafenib and trametinib.
Dr. Amaria and colleagues reported individual disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others, including Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study.
SOURCE: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9
Although results of the study by Amaria et al. are “promising,” the role of neoadjuvant therapy in treatment of stage III–IV oligometastatic melanoma in clinical practice “is unclear for now,” melanoma specialists Paolo A. Ascierto, MD, and Alexander M. M. Eggermont, MD, PhD, wrote in an editorial.
Amaria et al. have presented results of the first randomized trial to evaluate neoadjuvant therapy versus standard care in patients with high-risk resectable BRAF-mutated melanoma.
Patients who received both neoadjuvant and adjuvant treatment with the dabrafenib/trametinib combination had superior event-free survival versus standard surgery and consideration for adjuvant therapy, published results show.
However, previous studies have already shown good results on this, that adjuvant dabrafenib plus trametinib (as well as nivolumab monotherapy) in this setting, “raising the question of whether a neoadjuvant approach is really needed, especially given a possible reduction of the role of surgery in the future,” Dr. Ascierto and Dr. Eggermont wrote.
Alternatively, adjuvant therapy with newer, more effective agents may be a “better way forward,” they said, noting that three patients in the trial by Amaria et al. who progressed after neoadjuvant/adjuvant dabrafenib and trametinib relapsed at first with brain metastases, raising the question of whether the treatment “might induce a resistant phenotype predisposed to the development of CNS metastases.”
That said, effectively combining neoadjuvant with adjuvant therapy could reduce the extent of surgery, make radiotherapy redundant, or increase distant metastasis-free survival and overall survival, among other benefits.
“The next generation of adjuvant trials should aim to address these outstanding questions,” they concluded.
Dr. Ascierto is with Istituto Nazionale Tumori Fondazione “G Pascale,” Napoli, Italy, and Dr. Eggermont is with Cancer Institute Gustave Roussy, University Paris-Sud, France. This commentary is based on their editorial appearing in The Lancet Oncology (2018 Jan 17. doi: 10.1016/S1470-2045[18]30016-0). The authors reported disclosures related to Novartis, Merck Serono, Bristol-Myers Squibb, Amgen, and others.
Although results of the study by Amaria et al. are “promising,” the role of neoadjuvant therapy in treatment of stage III–IV oligometastatic melanoma in clinical practice “is unclear for now,” melanoma specialists Paolo A. Ascierto, MD, and Alexander M. M. Eggermont, MD, PhD, wrote in an editorial.
Amaria et al. have presented results of the first randomized trial to evaluate neoadjuvant therapy versus standard care in patients with high-risk resectable BRAF-mutated melanoma.
Patients who received both neoadjuvant and adjuvant treatment with the dabrafenib/trametinib combination had superior event-free survival versus standard surgery and consideration for adjuvant therapy, published results show.
However, previous studies have already shown good results on this, that adjuvant dabrafenib plus trametinib (as well as nivolumab monotherapy) in this setting, “raising the question of whether a neoadjuvant approach is really needed, especially given a possible reduction of the role of surgery in the future,” Dr. Ascierto and Dr. Eggermont wrote.
Alternatively, adjuvant therapy with newer, more effective agents may be a “better way forward,” they said, noting that three patients in the trial by Amaria et al. who progressed after neoadjuvant/adjuvant dabrafenib and trametinib relapsed at first with brain metastases, raising the question of whether the treatment “might induce a resistant phenotype predisposed to the development of CNS metastases.”
That said, effectively combining neoadjuvant with adjuvant therapy could reduce the extent of surgery, make radiotherapy redundant, or increase distant metastasis-free survival and overall survival, among other benefits.
“The next generation of adjuvant trials should aim to address these outstanding questions,” they concluded.
Dr. Ascierto is with Istituto Nazionale Tumori Fondazione “G Pascale,” Napoli, Italy, and Dr. Eggermont is with Cancer Institute Gustave Roussy, University Paris-Sud, France. This commentary is based on their editorial appearing in The Lancet Oncology (2018 Jan 17. doi: 10.1016/S1470-2045[18]30016-0). The authors reported disclosures related to Novartis, Merck Serono, Bristol-Myers Squibb, Amgen, and others.
Although results of the study by Amaria et al. are “promising,” the role of neoadjuvant therapy in treatment of stage III–IV oligometastatic melanoma in clinical practice “is unclear for now,” melanoma specialists Paolo A. Ascierto, MD, and Alexander M. M. Eggermont, MD, PhD, wrote in an editorial.
Amaria et al. have presented results of the first randomized trial to evaluate neoadjuvant therapy versus standard care in patients with high-risk resectable BRAF-mutated melanoma.
Patients who received both neoadjuvant and adjuvant treatment with the dabrafenib/trametinib combination had superior event-free survival versus standard surgery and consideration for adjuvant therapy, published results show.
However, previous studies have already shown good results on this, that adjuvant dabrafenib plus trametinib (as well as nivolumab monotherapy) in this setting, “raising the question of whether a neoadjuvant approach is really needed, especially given a possible reduction of the role of surgery in the future,” Dr. Ascierto and Dr. Eggermont wrote.
Alternatively, adjuvant therapy with newer, more effective agents may be a “better way forward,” they said, noting that three patients in the trial by Amaria et al. who progressed after neoadjuvant/adjuvant dabrafenib and trametinib relapsed at first with brain metastases, raising the question of whether the treatment “might induce a resistant phenotype predisposed to the development of CNS metastases.”
That said, effectively combining neoadjuvant with adjuvant therapy could reduce the extent of surgery, make radiotherapy redundant, or increase distant metastasis-free survival and overall survival, among other benefits.
“The next generation of adjuvant trials should aim to address these outstanding questions,” they concluded.
Dr. Ascierto is with Istituto Nazionale Tumori Fondazione “G Pascale,” Napoli, Italy, and Dr. Eggermont is with Cancer Institute Gustave Roussy, University Paris-Sud, France. This commentary is based on their editorial appearing in The Lancet Oncology (2018 Jan 17. doi: 10.1016/S1470-2045[18]30016-0). The authors reported disclosures related to Novartis, Merck Serono, Bristol-Myers Squibb, Amgen, and others.
For patients with surgically resectable, BRAF-mutated melanoma, neoadjuvant and adjuvant treatment with the combination of dabrafenib and trametinib resulted in significantly longer event-free survival compared with standard care, according to results of a randomized study.
The trial was closed early because of the “large difference” in event-free survival favoring the neoadjuvant approach, the authors wrote (Lancet Oncol. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
While early closure limits interpretation of results, they do provide important proof-of-concept and data for future studies, wrote the authors, led by Rodabe N Amaria, MD, of the department of medical oncology, University of Texas MD Anderson Cancer Center, Houston.
“The clinical and translational results strongly support the rationale for further assessment of neoadjuvant therapy in patients with high-risk, surgically resectable melanoma,” Dr. Amaria and colleagues said in the report on the randomized trial, believed to be the first to evaluate the role of neoadjuvant therapy versus standard care in BRAF-mutated melanoma.
Dabrafenib and trametinib combination therapy is approved as a treatment for patients with unresectable or metastatic stage IV melanoma and a BRAFV600 mutation, which is found in about half of cutaneous melanomas, authors said.
To evaluate the combination in earlier stage disease, Dr. Amaria and coinvestigators conducted a single-center, open-label, randomized, phase 2 trial of 21 patients with surgically resectable clinical stage III or oligometastatic stage IV melanoma with BRAFV600E or BRAFV600K mutations.
Patients were randomized 2:1 to receive the neoadjuvant/adjuvant treatment or to standard of care, which consisted of standard surgery plus consideration for adjuvant therapy, the authors said. Those patients assigned to the targeted therapy arm received 8 weeks of neoadjuvant dabrafenib and trametinib followed by surgery, then adjuvant dabrafenib and trametinib for up to 44 weeks.
Event-free survival, the primary endpoint of the trial, was a median of 19.7 months for neoadjuvant plus adjuvant dabrafenib and trametinib, versus 2.9 months for standard care (P less than .0001), the investigators reported.
Dabrafenib and trametinib combination therapy was well tolerated as neoadjuvant and adjuvant therapy, with no grade 4 adverse events or treatment related deaths, according to the investigators. The most common grade 3 adverse event seen with the combination was diarrhea, occurring in 2 patients (15%).
The trial is continuing as a single-arm study of neoadjuvant plus adjuvant dabrafenib and trametinib.
Dr. Amaria and colleagues reported individual disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others, including Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study.
SOURCE: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9
For patients with surgically resectable, BRAF-mutated melanoma, neoadjuvant and adjuvant treatment with the combination of dabrafenib and trametinib resulted in significantly longer event-free survival compared with standard care, according to results of a randomized study.
The trial was closed early because of the “large difference” in event-free survival favoring the neoadjuvant approach, the authors wrote (Lancet Oncol. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.
While early closure limits interpretation of results, they do provide important proof-of-concept and data for future studies, wrote the authors, led by Rodabe N Amaria, MD, of the department of medical oncology, University of Texas MD Anderson Cancer Center, Houston.
“The clinical and translational results strongly support the rationale for further assessment of neoadjuvant therapy in patients with high-risk, surgically resectable melanoma,” Dr. Amaria and colleagues said in the report on the randomized trial, believed to be the first to evaluate the role of neoadjuvant therapy versus standard care in BRAF-mutated melanoma.
Dabrafenib and trametinib combination therapy is approved as a treatment for patients with unresectable or metastatic stage IV melanoma and a BRAFV600 mutation, which is found in about half of cutaneous melanomas, authors said.
To evaluate the combination in earlier stage disease, Dr. Amaria and coinvestigators conducted a single-center, open-label, randomized, phase 2 trial of 21 patients with surgically resectable clinical stage III or oligometastatic stage IV melanoma with BRAFV600E or BRAFV600K mutations.
Patients were randomized 2:1 to receive the neoadjuvant/adjuvant treatment or to standard of care, which consisted of standard surgery plus consideration for adjuvant therapy, the authors said. Those patients assigned to the targeted therapy arm received 8 weeks of neoadjuvant dabrafenib and trametinib followed by surgery, then adjuvant dabrafenib and trametinib for up to 44 weeks.
Event-free survival, the primary endpoint of the trial, was a median of 19.7 months for neoadjuvant plus adjuvant dabrafenib and trametinib, versus 2.9 months for standard care (P less than .0001), the investigators reported.
Dabrafenib and trametinib combination therapy was well tolerated as neoadjuvant and adjuvant therapy, with no grade 4 adverse events or treatment related deaths, according to the investigators. The most common grade 3 adverse event seen with the combination was diarrhea, occurring in 2 patients (15%).
The trial is continuing as a single-arm study of neoadjuvant plus adjuvant dabrafenib and trametinib.
Dr. Amaria and colleagues reported individual disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others, including Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study.
SOURCE: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9
FROM THE LANCET ONCOLOGY
Key clinical point: In patients with high-risk, surgically resectable, clinical stage III-IV melanoma, dabrafenib and trametinib given in both the neoadjuvant and adjuvant setting improved event-free survival compared with standard care.
Major finding: Median event-free survival was 19.7 months for neoadjuvant/adjuvant dabrafenib and trametinib versus 2.9 months for standard upfront surgery including consideration for standard adjuvant therapy (P less than .0001).
Data source: A single-center, open-label, randomized, phase 2 trial including 21 patients with surgically resectable clinical stage III or oligometastatic stage IV BRAF-mutated melanoma.
Disclosures: Investigators reported ties to Novartis Pharmaceuticals Corp., which supplied drugs and funded clinical aspects of the study, and disclosures related to Merck, Bristol-Myers Squibb, Array Biopharma, and others.
Source: Amaria et al. 2018 Jan 17. doi: 10.1016/S1470-2045(18)30015-9.