User login
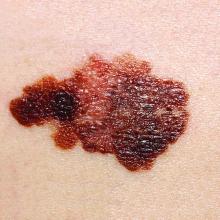
Second-melanoma risk higher with indoor tanning
than those who avoid indoor tanning, according to a retrospective study involving 434 melanoma patients.

The incidence of second melanomas over the entire 16-year course of the study was 25.2% among the tanning-bed users and 18.6% for nonusers. Among these study subjects – 27 with tanning-bed exposure and 61 without – median time to the second tumor was 225 days (0.62 years) for exposed patients and 1,280 days (3.50 years) for those with no exposure, the investigators reported.
This study, they wrote, is the first to show that “patients who had second primary melanoma diagnoses were more likely to have had” exposure to artificial UVR. The increased radiation intensity of tanning beds, “as opposed to UVR from ambient sunlight, in a physiologically vulnerable patient population [fair-skinned persons] at an early age contributes to our findings of decreased tumor lag time.”
SOURCE: Li Y et al. J Am Acad Dermatol. 2018;79(6):1101-8.
than those who avoid indoor tanning, according to a retrospective study involving 434 melanoma patients.

The incidence of second melanomas over the entire 16-year course of the study was 25.2% among the tanning-bed users and 18.6% for nonusers. Among these study subjects – 27 with tanning-bed exposure and 61 without – median time to the second tumor was 225 days (0.62 years) for exposed patients and 1,280 days (3.50 years) for those with no exposure, the investigators reported.
This study, they wrote, is the first to show that “patients who had second primary melanoma diagnoses were more likely to have had” exposure to artificial UVR. The increased radiation intensity of tanning beds, “as opposed to UVR from ambient sunlight, in a physiologically vulnerable patient population [fair-skinned persons] at an early age contributes to our findings of decreased tumor lag time.”
SOURCE: Li Y et al. J Am Acad Dermatol. 2018;79(6):1101-8.
than those who avoid indoor tanning, according to a retrospective study involving 434 melanoma patients.

The incidence of second melanomas over the entire 16-year course of the study was 25.2% among the tanning-bed users and 18.6% for nonusers. Among these study subjects – 27 with tanning-bed exposure and 61 without – median time to the second tumor was 225 days (0.62 years) for exposed patients and 1,280 days (3.50 years) for those with no exposure, the investigators reported.
This study, they wrote, is the first to show that “patients who had second primary melanoma diagnoses were more likely to have had” exposure to artificial UVR. The increased radiation intensity of tanning beds, “as opposed to UVR from ambient sunlight, in a physiologically vulnerable patient population [fair-skinned persons] at an early age contributes to our findings of decreased tumor lag time.”
SOURCE: Li Y et al. J Am Acad Dermatol. 2018;79(6):1101-8.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
SRS beats surgery in early control of brain mets, advantage fades with time
Stereotactic radiosurgery (SRS) provides better early local control of brain metastases than complete surgical resection, but this advantage fades with time, according to investigators.
By 6 months, lower risks associated with SRS shifted in favor of those who had surgical resection, reported lead author Thomas Churilla, MD, of Fox Chase Cancer Center in Philadelphia and his colleagues.
“Outside recognized indications for surgery such as establishing diagnosis or relieving mass effect, little evidence is available to guide the therapeutic choice of SRS vs. surgical resection in the treatment of patients with limited brain metastases,” the investigators wrote in JAMA Oncology.
The investigators performed an exploratory analysis of data from the European Organization for the Research and Treatment of Cancer (EORTC) 22952-26001 phase 3 trial, which was designed to evaluate whole-brain radiotherapy for patients with one to three brain metastases who had undergone SRS or complete surgical resection. The present analysis involved 268 patients, of whom 154 had SRS and 114 had complete surgical resection.
Primary tumors included lung, breast, colorectum, kidney, and melanoma. Initial analysis showed that patients undergoing surgical resection, compared with those who had SRS, typically had larger brain metastases (median, 28 mm vs. 20 mm) and more often had 1 brain metastasis (98.2% vs. 74.0%). Mass locality also differed between groups; compared with patients receiving SRS, surgical patients more often had metastases in the posterior fossa (26.3% vs. 7.8%) and less often in the parietal lobe (18.4% vs. 39.6%).
After median follow-up of 39.9 months, risks of local recurrence were similar between surgical and SRS groups (hazard ratio, 1.15). Stratifying by interval, however, showed that surgical patients were at much higher risk of local recurrence in the first 3 months following treatment (HR for 0-3 months, 5.94). Of note, this risk faded with time (HR for 3-6 months, 1.37; HR for 6-9 months, 0.75; HR for 9 months or longer, 0.36). From the 6-9 months interval onward, surgical patients had lower risk of recurrence, compared with SRS patients, and the risk even decreased after the 6-9 month interval.
“Prospective controlled trials are warranted to direct the optimal local approach for patients with brain metastases and to define whether any population may benefit from escalation in local therapy,” the investigators concluded.
The study was funded by the National Cancer Institute, National Institutes of Health, and Fonds Cancer in Belgium. One author reported receiving financial compensation from Pfizer via her institution.
SOURCE: Churilla T et al. JAMA Onc. 2018. doi: 10.1001/jamaoncol.2018.4610.
Stereotactic radiosurgery (SRS) provides better early local control of brain metastases than complete surgical resection, but this advantage fades with time, according to investigators.
By 6 months, lower risks associated with SRS shifted in favor of those who had surgical resection, reported lead author Thomas Churilla, MD, of Fox Chase Cancer Center in Philadelphia and his colleagues.
“Outside recognized indications for surgery such as establishing diagnosis or relieving mass effect, little evidence is available to guide the therapeutic choice of SRS vs. surgical resection in the treatment of patients with limited brain metastases,” the investigators wrote in JAMA Oncology.
The investigators performed an exploratory analysis of data from the European Organization for the Research and Treatment of Cancer (EORTC) 22952-26001 phase 3 trial, which was designed to evaluate whole-brain radiotherapy for patients with one to three brain metastases who had undergone SRS or complete surgical resection. The present analysis involved 268 patients, of whom 154 had SRS and 114 had complete surgical resection.
Primary tumors included lung, breast, colorectum, kidney, and melanoma. Initial analysis showed that patients undergoing surgical resection, compared with those who had SRS, typically had larger brain metastases (median, 28 mm vs. 20 mm) and more often had 1 brain metastasis (98.2% vs. 74.0%). Mass locality also differed between groups; compared with patients receiving SRS, surgical patients more often had metastases in the posterior fossa (26.3% vs. 7.8%) and less often in the parietal lobe (18.4% vs. 39.6%).
After median follow-up of 39.9 months, risks of local recurrence were similar between surgical and SRS groups (hazard ratio, 1.15). Stratifying by interval, however, showed that surgical patients were at much higher risk of local recurrence in the first 3 months following treatment (HR for 0-3 months, 5.94). Of note, this risk faded with time (HR for 3-6 months, 1.37; HR for 6-9 months, 0.75; HR for 9 months or longer, 0.36). From the 6-9 months interval onward, surgical patients had lower risk of recurrence, compared with SRS patients, and the risk even decreased after the 6-9 month interval.
“Prospective controlled trials are warranted to direct the optimal local approach for patients with brain metastases and to define whether any population may benefit from escalation in local therapy,” the investigators concluded.
The study was funded by the National Cancer Institute, National Institutes of Health, and Fonds Cancer in Belgium. One author reported receiving financial compensation from Pfizer via her institution.
SOURCE: Churilla T et al. JAMA Onc. 2018. doi: 10.1001/jamaoncol.2018.4610.
Stereotactic radiosurgery (SRS) provides better early local control of brain metastases than complete surgical resection, but this advantage fades with time, according to investigators.
By 6 months, lower risks associated with SRS shifted in favor of those who had surgical resection, reported lead author Thomas Churilla, MD, of Fox Chase Cancer Center in Philadelphia and his colleagues.
“Outside recognized indications for surgery such as establishing diagnosis or relieving mass effect, little evidence is available to guide the therapeutic choice of SRS vs. surgical resection in the treatment of patients with limited brain metastases,” the investigators wrote in JAMA Oncology.
The investigators performed an exploratory analysis of data from the European Organization for the Research and Treatment of Cancer (EORTC) 22952-26001 phase 3 trial, which was designed to evaluate whole-brain radiotherapy for patients with one to three brain metastases who had undergone SRS or complete surgical resection. The present analysis involved 268 patients, of whom 154 had SRS and 114 had complete surgical resection.
Primary tumors included lung, breast, colorectum, kidney, and melanoma. Initial analysis showed that patients undergoing surgical resection, compared with those who had SRS, typically had larger brain metastases (median, 28 mm vs. 20 mm) and more often had 1 brain metastasis (98.2% vs. 74.0%). Mass locality also differed between groups; compared with patients receiving SRS, surgical patients more often had metastases in the posterior fossa (26.3% vs. 7.8%) and less often in the parietal lobe (18.4% vs. 39.6%).
After median follow-up of 39.9 months, risks of local recurrence were similar between surgical and SRS groups (hazard ratio, 1.15). Stratifying by interval, however, showed that surgical patients were at much higher risk of local recurrence in the first 3 months following treatment (HR for 0-3 months, 5.94). Of note, this risk faded with time (HR for 3-6 months, 1.37; HR for 6-9 months, 0.75; HR for 9 months or longer, 0.36). From the 6-9 months interval onward, surgical patients had lower risk of recurrence, compared with SRS patients, and the risk even decreased after the 6-9 month interval.
“Prospective controlled trials are warranted to direct the optimal local approach for patients with brain metastases and to define whether any population may benefit from escalation in local therapy,” the investigators concluded.
The study was funded by the National Cancer Institute, National Institutes of Health, and Fonds Cancer in Belgium. One author reported receiving financial compensation from Pfizer via her institution.
SOURCE: Churilla T et al. JAMA Onc. 2018. doi: 10.1001/jamaoncol.2018.4610.
FROM JAMA ONCOLOGY
Key clinical point: Stereotactic radiosurgery (SRS) provides better early local control of brain metastases than surgical resection, but this advantage fades with time.
Major finding: Patients treated with surgery were more likely to have local recurrence in the first 3 months following treatment, compared with patients treated with SRS (hazard ratio, 5.94).
Study details: An exploratory analysis of data from the European Organization for the Research and Treatment of Cancer (EORTC) 22952-26001 phase 3 trial. Analysis involved 268 patients with one to three brain metastases who underwent whole-brain radiotherapy or observation after SRS (n = 154) or complete surgical resection (n = 114).
Disclosures: The study was funded by the National Cancer Institute, National Institutes of Health, and Fonds Cancer in Belgium. Dr. Handorf reported financial compensation from Pfizer, via her institution.
Source: Churilla T et al. JAMA Onc. 2018. doi: 10.1001/jamaoncol.2018.4610.
New pregnancy, genetic testing guidance added to AAD’s melanoma guidelines
Pregnancy does not necessarily increase a woman’s risk for melanoma, nor is it clear that becoming pregnant affects melanoma’s disease course, according to current evidence. This guidance is among several updates added to newly released guidelines for managing patients with primary cutaneous melanoma.
also addressed the burgeoning field of genetic testing for cancer in the guidelines, which were published online on Nov. 1. Although there may be a hereditary component to some melanomas, genetic testing may not be appropriate for all patients, and any formal genetic testing should be carried out only after individualized education and counseling, according to the updates.
However, the guidelines make it clear that all patients whose family history includes melanoma should be counseled about their genetic risk.
As with genetic testing, counseling regarding future pregnancies for women with melanoma, or a history of melanoma, should be personalized and account for individual history and melanoma risk, according to the new guidelines. Since evidence is lacking that pregnancy affects the course of melanoma, physicians caring for pregnant women with melanoma should first look at patient and the disease characteristics. The addition of detailed guidance regarding pregnancy reflects research showing that CM is the most common malignancy seen in pregnancy, amounting to nearly one-third of the malignancies that arise in pregnancy. “Although the incidence of CM is generally higher in men, it is higher in younger women than in men, most notably during women’s reproductive years,” wrote Susan M. Swetter, MD, and her guideline coauthors.
“Melanoma is the deadliest form of skin cancer, and we hope these guidelines will help dermatologists and other physicians enhance their delivery of life-saving treatment to patients,” Dr. Swetter said in a press release announcing the guideline updates. Dr. Swetter, professor of dermatology and director of the pigmented lesion and melanoma program at Stanford (Calif.) University Medical Center and Cancer Institute, led the working group that developed the guidelines. “In order to provide the best possible resource for practitioners, we reviewed the latest scientific data and addressed certain topics that weren’t covered in the AAD’s previous melanoma guidelines,” she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. Adjuvant topical therapies or radiation, say the guidelines, can be considered as second-line care, but only in limited situations in which surgery is not feasible. Staged excision techniques, such as Mohs surgery, also may be considered for certain types of melanoma and in certain body areas.
In an interview, Dr. Swetter also said that is critical that the updated guidelines have been harmonized with changes made in the American Joint Committee on Cancer’s 8th edition of its melanoma staging manual. Key points for dermatologists to understand that reporting of Breslow thickness to the nearest 1/10th decimal point (over the nearest 1/100th), such that a melanoma measuring 0.75-0.84 mm in thickness would be reported as 0.8 mm depth and one between 0.95-1.04 mm would be rounded to 1 mm.
The main changes regarding staging of thin (T1) melanoma – that is less than or equal to 1 mm – is that the 0.8 mm thickness is the threshold for a T1a melanoma (now classified as less than 0.8 mm without ulceration), whereas T1b is now 0.8 – 1.0 mm thickness with or w/out ulceration or less than 0.8 mm thickness with ulceration. A T1a melanoma generally is not considered appropriate for staging of the regional lymph nodes with sentinel lymph node biopsy (with exceptions noted in the guideline), whereas a T1b melanoma may be considered for SLNB staging – though rates of SLN positivity remain relatively low in the T1b group.”
Dr. Swetter also emphasized that histologic ulceration of the primary tumor was affirmed as an indicator of worse prognosis; mitotic rate, although removed from T1 staging, is still tracked by pathologists and still seen as an independent predictor of worse prognosis, according to the 8th edition, she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. “Mohs micrographic surgery and other staged excision techniques can provide exhaustive peripheral margin histologic assessment for melanoma in situ, lentigo maligna type and tissue sparing in anatomically constrained sites,” Dr. Swetter said. “Current data are insufficient to recommend Mohs surgery for invasive cutaneous melanoma, in which the use of surgical margins less than 1 cm has not been adequately studied,” she cautioned.
Reinforcing the importance of surgery as the primary treatment for melanoma, Dr. Swetter clarified that “Nonsurgical approaches (imiquimod and traditional forms of radiation therapy) should be considered [only] if surgery is impractical or contraindicated, and only for melanoma in situ, lentigo maligna type, as cure rates are lower.”
In terms of other therapies, the guideline working group found insufficient evidence to recommend electronic brachytherapy for melanoma.
Assessment of novel diagnostic and molecular imaging modalities was not the primary focus of the AAD guidelines, Dr. Swetter pointed out. Looking to the future, though, she added that the hope is “that these prebiopsy modalities can one day reduce unnecessary biopsies from being done” in the clinic.
Other knowledge gaps cited by the working group included several related to pathology, including determination of appropriate margin control in some lesion types, and the quest to reduce inter-reader variability in histopathologic diagnosis of melanocytic tissue samples. However, noted Dr. Swetter and her coauthors, the rapid pace of genomic medicine advances “may make many of the aforementioned issues obsolete” before the next guideline update.
In the interview, Dr. Swetter said that the guidelines reflect evolving thinking about melanoma in the context of a rapidly growing field. “Only in the last year have effective, more tolerable adjuvant therapies been [Food and Drug Administration] approved for patients with resected stage III melanoma, including patients with regional lymph node disease detected via sentinel lymph node biopsy. The hope is that less invasive procedures for melanoma will be performed in the future, and replaced by better drugs and novel techniques.”
Dr. Swetter reported that she had no relevant financial disclosures; several working group members reported multiple financial relationships with pharmaceutical, diagnostic, and imaging companies. Working group members were recused from discussion of guidelines where their particular relationships might pose a conflict of interest.
SOURCE: Swetter S. et al. J Am Acad Dermatol. 2011 Nov;65(5):1032-47.
Pregnancy does not necessarily increase a woman’s risk for melanoma, nor is it clear that becoming pregnant affects melanoma’s disease course, according to current evidence. This guidance is among several updates added to newly released guidelines for managing patients with primary cutaneous melanoma.
also addressed the burgeoning field of genetic testing for cancer in the guidelines, which were published online on Nov. 1. Although there may be a hereditary component to some melanomas, genetic testing may not be appropriate for all patients, and any formal genetic testing should be carried out only after individualized education and counseling, according to the updates.
However, the guidelines make it clear that all patients whose family history includes melanoma should be counseled about their genetic risk.
As with genetic testing, counseling regarding future pregnancies for women with melanoma, or a history of melanoma, should be personalized and account for individual history and melanoma risk, according to the new guidelines. Since evidence is lacking that pregnancy affects the course of melanoma, physicians caring for pregnant women with melanoma should first look at patient and the disease characteristics. The addition of detailed guidance regarding pregnancy reflects research showing that CM is the most common malignancy seen in pregnancy, amounting to nearly one-third of the malignancies that arise in pregnancy. “Although the incidence of CM is generally higher in men, it is higher in younger women than in men, most notably during women’s reproductive years,” wrote Susan M. Swetter, MD, and her guideline coauthors.
“Melanoma is the deadliest form of skin cancer, and we hope these guidelines will help dermatologists and other physicians enhance their delivery of life-saving treatment to patients,” Dr. Swetter said in a press release announcing the guideline updates. Dr. Swetter, professor of dermatology and director of the pigmented lesion and melanoma program at Stanford (Calif.) University Medical Center and Cancer Institute, led the working group that developed the guidelines. “In order to provide the best possible resource for practitioners, we reviewed the latest scientific data and addressed certain topics that weren’t covered in the AAD’s previous melanoma guidelines,” she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. Adjuvant topical therapies or radiation, say the guidelines, can be considered as second-line care, but only in limited situations in which surgery is not feasible. Staged excision techniques, such as Mohs surgery, also may be considered for certain types of melanoma and in certain body areas.
In an interview, Dr. Swetter also said that is critical that the updated guidelines have been harmonized with changes made in the American Joint Committee on Cancer’s 8th edition of its melanoma staging manual. Key points for dermatologists to understand that reporting of Breslow thickness to the nearest 1/10th decimal point (over the nearest 1/100th), such that a melanoma measuring 0.75-0.84 mm in thickness would be reported as 0.8 mm depth and one between 0.95-1.04 mm would be rounded to 1 mm.
The main changes regarding staging of thin (T1) melanoma – that is less than or equal to 1 mm – is that the 0.8 mm thickness is the threshold for a T1a melanoma (now classified as less than 0.8 mm without ulceration), whereas T1b is now 0.8 – 1.0 mm thickness with or w/out ulceration or less than 0.8 mm thickness with ulceration. A T1a melanoma generally is not considered appropriate for staging of the regional lymph nodes with sentinel lymph node biopsy (with exceptions noted in the guideline), whereas a T1b melanoma may be considered for SLNB staging – though rates of SLN positivity remain relatively low in the T1b group.”
Dr. Swetter also emphasized that histologic ulceration of the primary tumor was affirmed as an indicator of worse prognosis; mitotic rate, although removed from T1 staging, is still tracked by pathologists and still seen as an independent predictor of worse prognosis, according to the 8th edition, she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. “Mohs micrographic surgery and other staged excision techniques can provide exhaustive peripheral margin histologic assessment for melanoma in situ, lentigo maligna type and tissue sparing in anatomically constrained sites,” Dr. Swetter said. “Current data are insufficient to recommend Mohs surgery for invasive cutaneous melanoma, in which the use of surgical margins less than 1 cm has not been adequately studied,” she cautioned.
Reinforcing the importance of surgery as the primary treatment for melanoma, Dr. Swetter clarified that “Nonsurgical approaches (imiquimod and traditional forms of radiation therapy) should be considered [only] if surgery is impractical or contraindicated, and only for melanoma in situ, lentigo maligna type, as cure rates are lower.”
In terms of other therapies, the guideline working group found insufficient evidence to recommend electronic brachytherapy for melanoma.
Assessment of novel diagnostic and molecular imaging modalities was not the primary focus of the AAD guidelines, Dr. Swetter pointed out. Looking to the future, though, she added that the hope is “that these prebiopsy modalities can one day reduce unnecessary biopsies from being done” in the clinic.
Other knowledge gaps cited by the working group included several related to pathology, including determination of appropriate margin control in some lesion types, and the quest to reduce inter-reader variability in histopathologic diagnosis of melanocytic tissue samples. However, noted Dr. Swetter and her coauthors, the rapid pace of genomic medicine advances “may make many of the aforementioned issues obsolete” before the next guideline update.
In the interview, Dr. Swetter said that the guidelines reflect evolving thinking about melanoma in the context of a rapidly growing field. “Only in the last year have effective, more tolerable adjuvant therapies been [Food and Drug Administration] approved for patients with resected stage III melanoma, including patients with regional lymph node disease detected via sentinel lymph node biopsy. The hope is that less invasive procedures for melanoma will be performed in the future, and replaced by better drugs and novel techniques.”
Dr. Swetter reported that she had no relevant financial disclosures; several working group members reported multiple financial relationships with pharmaceutical, diagnostic, and imaging companies. Working group members were recused from discussion of guidelines where their particular relationships might pose a conflict of interest.
SOURCE: Swetter S. et al. J Am Acad Dermatol. 2011 Nov;65(5):1032-47.
Pregnancy does not necessarily increase a woman’s risk for melanoma, nor is it clear that becoming pregnant affects melanoma’s disease course, according to current evidence. This guidance is among several updates added to newly released guidelines for managing patients with primary cutaneous melanoma.
also addressed the burgeoning field of genetic testing for cancer in the guidelines, which were published online on Nov. 1. Although there may be a hereditary component to some melanomas, genetic testing may not be appropriate for all patients, and any formal genetic testing should be carried out only after individualized education and counseling, according to the updates.
However, the guidelines make it clear that all patients whose family history includes melanoma should be counseled about their genetic risk.
As with genetic testing, counseling regarding future pregnancies for women with melanoma, or a history of melanoma, should be personalized and account for individual history and melanoma risk, according to the new guidelines. Since evidence is lacking that pregnancy affects the course of melanoma, physicians caring for pregnant women with melanoma should first look at patient and the disease characteristics. The addition of detailed guidance regarding pregnancy reflects research showing that CM is the most common malignancy seen in pregnancy, amounting to nearly one-third of the malignancies that arise in pregnancy. “Although the incidence of CM is generally higher in men, it is higher in younger women than in men, most notably during women’s reproductive years,” wrote Susan M. Swetter, MD, and her guideline coauthors.
“Melanoma is the deadliest form of skin cancer, and we hope these guidelines will help dermatologists and other physicians enhance their delivery of life-saving treatment to patients,” Dr. Swetter said in a press release announcing the guideline updates. Dr. Swetter, professor of dermatology and director of the pigmented lesion and melanoma program at Stanford (Calif.) University Medical Center and Cancer Institute, led the working group that developed the guidelines. “In order to provide the best possible resource for practitioners, we reviewed the latest scientific data and addressed certain topics that weren’t covered in the AAD’s previous melanoma guidelines,” she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. Adjuvant topical therapies or radiation, say the guidelines, can be considered as second-line care, but only in limited situations in which surgery is not feasible. Staged excision techniques, such as Mohs surgery, also may be considered for certain types of melanoma and in certain body areas.
In an interview, Dr. Swetter also said that is critical that the updated guidelines have been harmonized with changes made in the American Joint Committee on Cancer’s 8th edition of its melanoma staging manual. Key points for dermatologists to understand that reporting of Breslow thickness to the nearest 1/10th decimal point (over the nearest 1/100th), such that a melanoma measuring 0.75-0.84 mm in thickness would be reported as 0.8 mm depth and one between 0.95-1.04 mm would be rounded to 1 mm.
The main changes regarding staging of thin (T1) melanoma – that is less than or equal to 1 mm – is that the 0.8 mm thickness is the threshold for a T1a melanoma (now classified as less than 0.8 mm without ulceration), whereas T1b is now 0.8 – 1.0 mm thickness with or w/out ulceration or less than 0.8 mm thickness with ulceration. A T1a melanoma generally is not considered appropriate for staging of the regional lymph nodes with sentinel lymph node biopsy (with exceptions noted in the guideline), whereas a T1b melanoma may be considered for SLNB staging – though rates of SLN positivity remain relatively low in the T1b group.”
Dr. Swetter also emphasized that histologic ulceration of the primary tumor was affirmed as an indicator of worse prognosis; mitotic rate, although removed from T1 staging, is still tracked by pathologists and still seen as an independent predictor of worse prognosis, according to the 8th edition, she said.
A cornerstone of cutaneous melanoma care remains unchanged in the guidelines: Surgical excision is still the preferred method for treating melanoma. “Mohs micrographic surgery and other staged excision techniques can provide exhaustive peripheral margin histologic assessment for melanoma in situ, lentigo maligna type and tissue sparing in anatomically constrained sites,” Dr. Swetter said. “Current data are insufficient to recommend Mohs surgery for invasive cutaneous melanoma, in which the use of surgical margins less than 1 cm has not been adequately studied,” she cautioned.
Reinforcing the importance of surgery as the primary treatment for melanoma, Dr. Swetter clarified that “Nonsurgical approaches (imiquimod and traditional forms of radiation therapy) should be considered [only] if surgery is impractical or contraindicated, and only for melanoma in situ, lentigo maligna type, as cure rates are lower.”
In terms of other therapies, the guideline working group found insufficient evidence to recommend electronic brachytherapy for melanoma.
Assessment of novel diagnostic and molecular imaging modalities was not the primary focus of the AAD guidelines, Dr. Swetter pointed out. Looking to the future, though, she added that the hope is “that these prebiopsy modalities can one day reduce unnecessary biopsies from being done” in the clinic.
Other knowledge gaps cited by the working group included several related to pathology, including determination of appropriate margin control in some lesion types, and the quest to reduce inter-reader variability in histopathologic diagnosis of melanocytic tissue samples. However, noted Dr. Swetter and her coauthors, the rapid pace of genomic medicine advances “may make many of the aforementioned issues obsolete” before the next guideline update.
In the interview, Dr. Swetter said that the guidelines reflect evolving thinking about melanoma in the context of a rapidly growing field. “Only in the last year have effective, more tolerable adjuvant therapies been [Food and Drug Administration] approved for patients with resected stage III melanoma, including patients with regional lymph node disease detected via sentinel lymph node biopsy. The hope is that less invasive procedures for melanoma will be performed in the future, and replaced by better drugs and novel techniques.”
Dr. Swetter reported that she had no relevant financial disclosures; several working group members reported multiple financial relationships with pharmaceutical, diagnostic, and imaging companies. Working group members were recused from discussion of guidelines where their particular relationships might pose a conflict of interest.
SOURCE: Swetter S. et al. J Am Acad Dermatol. 2011 Nov;65(5):1032-47.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Circulating tumor DNA identified by fragment size
Circulating tumor DNA could be effectively isolated from plasma by focusing on a particular range of fragment sizes, which paves the way for noninvasive genomic analysis of tumor DNA, new research suggests.
In a study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls, DNA fragment length could be used to distinguish circulating tumor DNA (ctDNA) from other cell-free DNA (cfDNA), investigators reported in Science Translational Medicine.
“We hypothesized that we could improve the sensitivity for noninvasive cancer genomics by selective sequencing of ctDNA fragments and by leveraging differences in the biology that determine DNA fragmentation,” wrote Florent Mouliere, PhD, from the Cancer Research UK Cambridge Institute, and coauthors.
Cell-free plasma fragments are often cleaved at around 167 base pairs in length and differences in length between circulating fetal and maternal DNA are already used for noninvasive prenatal diagnosis. However, the authors said that only a few studies, with conflicting results, have looked at the size distribution of tumor-derived cfDNA.
The study used two approaches to determining the size profile of mutant ctDNA. The first looked at tumor and nontumor cfDNA in mice with human ovarian cancer xenografts and the second approach used deep sequencing in 19 cancer patients. This revealed that tumor-derived cfDNA was most commonly found in fragments between 90-150 base pairs or 250-320 base pairs in size.
The researchers also noted that mutant circulating tumor DNA was generally more fragmented than nonmutant cfDNA and that patients with untreated advanced cancer showed consistently shorter lengths of mutant DNA.
The next question was whether size selection and other biological properties – such as somatic alterations – of the cfDNA could be used to enhance detection of ctDNA via machine learning technology.
Two models, designed to distinguish between healthy and cancerous samples, were developed using 153 samples, then validated on two datasets of 94 and 83 samples.
One of these models correctly classified cancerous samples in 94% of samples from patients with cancers known to have high levels of ctDNA – colorectal, cholangiocarcinoma, ovarian, breast, and melanoma – and in 65% of samples from low-ctDNA cancers – pancreatic, renal, and glioma.
Another model focused just on fragmentation patterns and was still able to distinguish cancer samples from those of healthy controls, although with slightly reduced area under the curve.
“Our results indicate that exploiting fundamental properties of cfDNA with fragment-specific analyses can allow more sensitive evaluation of ctDNA,” the authors wrote. “We identified features that could determine the presence and amount of ctDNA in plasma samples, without a prior knowledge of somatic aberrations.”
The authors pointed out that size selection of DNA fragments was relatively simple and cheap, and was also compatible with other genome-wide and targeted genomic analyses, “greatly increasing the potential value and utility of liquid biopsies as well as the cost-effectiveness of cfDNA sequencing.”
However, they cautioned that their catalogue had focused solely on double-stranded DNA and was subject to potential biases from the DNA extraction and sequencing methods they used in the study. They also commented that other biological effects could help refine the analysis of ctDNA.
“Other bodily fluids [urine, cerebrospinal fluid, and saliva], different nucleic acids and structures, altered mechanisms of release into circulation, or sample processing methods could exhibit varying fragment size signatures and could offer additional exploitable biological patterns for selective sequencing,” they wrote.
The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in ctDNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
SOURCE: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
Cell-free DNA analysis has tremendous diagnostic potential and so is a very active area of research. In this study, researchers were able to identify five variables and develop models for the detection of cancer following analysis of circulating tumor DNA. One of these models based on DNA fragmentation pattern performed very well, and so fragment length analyses could develop into a general test for the presence of cancer.
However confirmation of these findings in large, multicenter clinical trials is still needed. There is also the problem that size selection can result in a loss of circulating tumor DNA for analysis or may introduce biases. We also need to understand the mechanisms underpinning the different fragment size patterns seen in the study. But this study still substantially extends the potential of cell-free, DNA-based diagnostic tests.
Ellen Heitzer, PhD, and Michael R. Speicher, MD, are from the Medical University of Graz (Austria). These comments are taken from an accompanying editorial (Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aav3873). Both authors declared research funding from Servier and Dr. Heitzer declared laboratory research funding from Freenome and PreAnalytiX.
Cell-free DNA analysis has tremendous diagnostic potential and so is a very active area of research. In this study, researchers were able to identify five variables and develop models for the detection of cancer following analysis of circulating tumor DNA. One of these models based on DNA fragmentation pattern performed very well, and so fragment length analyses could develop into a general test for the presence of cancer.
However confirmation of these findings in large, multicenter clinical trials is still needed. There is also the problem that size selection can result in a loss of circulating tumor DNA for analysis or may introduce biases. We also need to understand the mechanisms underpinning the different fragment size patterns seen in the study. But this study still substantially extends the potential of cell-free, DNA-based diagnostic tests.
Ellen Heitzer, PhD, and Michael R. Speicher, MD, are from the Medical University of Graz (Austria). These comments are taken from an accompanying editorial (Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aav3873). Both authors declared research funding from Servier and Dr. Heitzer declared laboratory research funding from Freenome and PreAnalytiX.
Cell-free DNA analysis has tremendous diagnostic potential and so is a very active area of research. In this study, researchers were able to identify five variables and develop models for the detection of cancer following analysis of circulating tumor DNA. One of these models based on DNA fragmentation pattern performed very well, and so fragment length analyses could develop into a general test for the presence of cancer.
However confirmation of these findings in large, multicenter clinical trials is still needed. There is also the problem that size selection can result in a loss of circulating tumor DNA for analysis or may introduce biases. We also need to understand the mechanisms underpinning the different fragment size patterns seen in the study. But this study still substantially extends the potential of cell-free, DNA-based diagnostic tests.
Ellen Heitzer, PhD, and Michael R. Speicher, MD, are from the Medical University of Graz (Austria). These comments are taken from an accompanying editorial (Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aav3873). Both authors declared research funding from Servier and Dr. Heitzer declared laboratory research funding from Freenome and PreAnalytiX.
Circulating tumor DNA could be effectively isolated from plasma by focusing on a particular range of fragment sizes, which paves the way for noninvasive genomic analysis of tumor DNA, new research suggests.
In a study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls, DNA fragment length could be used to distinguish circulating tumor DNA (ctDNA) from other cell-free DNA (cfDNA), investigators reported in Science Translational Medicine.
“We hypothesized that we could improve the sensitivity for noninvasive cancer genomics by selective sequencing of ctDNA fragments and by leveraging differences in the biology that determine DNA fragmentation,” wrote Florent Mouliere, PhD, from the Cancer Research UK Cambridge Institute, and coauthors.
Cell-free plasma fragments are often cleaved at around 167 base pairs in length and differences in length between circulating fetal and maternal DNA are already used for noninvasive prenatal diagnosis. However, the authors said that only a few studies, with conflicting results, have looked at the size distribution of tumor-derived cfDNA.
The study used two approaches to determining the size profile of mutant ctDNA. The first looked at tumor and nontumor cfDNA in mice with human ovarian cancer xenografts and the second approach used deep sequencing in 19 cancer patients. This revealed that tumor-derived cfDNA was most commonly found in fragments between 90-150 base pairs or 250-320 base pairs in size.
The researchers also noted that mutant circulating tumor DNA was generally more fragmented than nonmutant cfDNA and that patients with untreated advanced cancer showed consistently shorter lengths of mutant DNA.
The next question was whether size selection and other biological properties – such as somatic alterations – of the cfDNA could be used to enhance detection of ctDNA via machine learning technology.
Two models, designed to distinguish between healthy and cancerous samples, were developed using 153 samples, then validated on two datasets of 94 and 83 samples.
One of these models correctly classified cancerous samples in 94% of samples from patients with cancers known to have high levels of ctDNA – colorectal, cholangiocarcinoma, ovarian, breast, and melanoma – and in 65% of samples from low-ctDNA cancers – pancreatic, renal, and glioma.
Another model focused just on fragmentation patterns and was still able to distinguish cancer samples from those of healthy controls, although with slightly reduced area under the curve.
“Our results indicate that exploiting fundamental properties of cfDNA with fragment-specific analyses can allow more sensitive evaluation of ctDNA,” the authors wrote. “We identified features that could determine the presence and amount of ctDNA in plasma samples, without a prior knowledge of somatic aberrations.”
The authors pointed out that size selection of DNA fragments was relatively simple and cheap, and was also compatible with other genome-wide and targeted genomic analyses, “greatly increasing the potential value and utility of liquid biopsies as well as the cost-effectiveness of cfDNA sequencing.”
However, they cautioned that their catalogue had focused solely on double-stranded DNA and was subject to potential biases from the DNA extraction and sequencing methods they used in the study. They also commented that other biological effects could help refine the analysis of ctDNA.
“Other bodily fluids [urine, cerebrospinal fluid, and saliva], different nucleic acids and structures, altered mechanisms of release into circulation, or sample processing methods could exhibit varying fragment size signatures and could offer additional exploitable biological patterns for selective sequencing,” they wrote.
The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in ctDNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
SOURCE: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
Circulating tumor DNA could be effectively isolated from plasma by focusing on a particular range of fragment sizes, which paves the way for noninvasive genomic analysis of tumor DNA, new research suggests.
In a study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls, DNA fragment length could be used to distinguish circulating tumor DNA (ctDNA) from other cell-free DNA (cfDNA), investigators reported in Science Translational Medicine.
“We hypothesized that we could improve the sensitivity for noninvasive cancer genomics by selective sequencing of ctDNA fragments and by leveraging differences in the biology that determine DNA fragmentation,” wrote Florent Mouliere, PhD, from the Cancer Research UK Cambridge Institute, and coauthors.
Cell-free plasma fragments are often cleaved at around 167 base pairs in length and differences in length between circulating fetal and maternal DNA are already used for noninvasive prenatal diagnosis. However, the authors said that only a few studies, with conflicting results, have looked at the size distribution of tumor-derived cfDNA.
The study used two approaches to determining the size profile of mutant ctDNA. The first looked at tumor and nontumor cfDNA in mice with human ovarian cancer xenografts and the second approach used deep sequencing in 19 cancer patients. This revealed that tumor-derived cfDNA was most commonly found in fragments between 90-150 base pairs or 250-320 base pairs in size.
The researchers also noted that mutant circulating tumor DNA was generally more fragmented than nonmutant cfDNA and that patients with untreated advanced cancer showed consistently shorter lengths of mutant DNA.
The next question was whether size selection and other biological properties – such as somatic alterations – of the cfDNA could be used to enhance detection of ctDNA via machine learning technology.
Two models, designed to distinguish between healthy and cancerous samples, were developed using 153 samples, then validated on two datasets of 94 and 83 samples.
One of these models correctly classified cancerous samples in 94% of samples from patients with cancers known to have high levels of ctDNA – colorectal, cholangiocarcinoma, ovarian, breast, and melanoma – and in 65% of samples from low-ctDNA cancers – pancreatic, renal, and glioma.
Another model focused just on fragmentation patterns and was still able to distinguish cancer samples from those of healthy controls, although with slightly reduced area under the curve.
“Our results indicate that exploiting fundamental properties of cfDNA with fragment-specific analyses can allow more sensitive evaluation of ctDNA,” the authors wrote. “We identified features that could determine the presence and amount of ctDNA in plasma samples, without a prior knowledge of somatic aberrations.”
The authors pointed out that size selection of DNA fragments was relatively simple and cheap, and was also compatible with other genome-wide and targeted genomic analyses, “greatly increasing the potential value and utility of liquid biopsies as well as the cost-effectiveness of cfDNA sequencing.”
However, they cautioned that their catalogue had focused solely on double-stranded DNA and was subject to potential biases from the DNA extraction and sequencing methods they used in the study. They also commented that other biological effects could help refine the analysis of ctDNA.
“Other bodily fluids [urine, cerebrospinal fluid, and saliva], different nucleic acids and structures, altered mechanisms of release into circulation, or sample processing methods could exhibit varying fragment size signatures and could offer additional exploitable biological patterns for selective sequencing,” they wrote.
The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in ctDNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
SOURCE: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
FROM SCIENCE TRANSLATIONAL MEDICINE
Key clinical point: The size of cell-free DNA could be used to single out circulating tumor DNA.
Major finding: Circulating tumor DNA fragments are more commonly found in the 90-150 base pair range.
Study details: A study of 344 plasma samples from 200 patients with 18 cancer types and 65 samples from healthy controls.
Disclosures: The study was supported by the University of Cambridge, Cancer Research UK, and the Engineering and Physical Sciences Research Council. Research supporting the study was also funded by the European Research Council, the National Institute for Health Research Cambridge, National Cancer Research Network, Cambridge Experimental Cancer Medicine Centre, Hutchison Whampoa, Target Ovarian Cancer, the Medical Research Council, and AstraZeneca. Three authors are cofounders, shareholders, and officers/consultants in a company specializing in circulating tumor DNA analysis. One author declared research funding and advisory board fees from private industry. Seven authors are listed on related patents.
Source: Mouliere F et al. Sci Transl Med. 2018 Nov 7. doi: 10.1126/scitranslmed.aat4921.
Nivo + ipi shows durable activity against metastatic melanoma
MUNICH – Four years on, the combination of the immune checkpoint inhibitors nivolumab and ipilimumab as well as nivolumab alone continue to show benefit as first-line therapies for patients with advanced malignant melanoma, compared with ipilimumab monotherapy, reported investigators in the CheckMate O67 trial.
Among 945 patients with previously untreated and unresectable stage III or IV malignant melanoma, median overall survival at a minimum of 48 months follow-up had not been reached for patients assigned to the combination of nivolumab (Opdivo) and ipilimumab (Yervoy), compared with 36.9 months for patients assigned to nivolumab alone, and 19.9 months for patients assigned to ipilimumab alone, reported F. Stephen Hodi Jr., MD, of Dana-Farber Cancer Institute, Boston, and his colleagues.
“There’s a durable, sustained clinical benefit that can be achieved with first-line nivo plus ipi combination or nivo alone in patients with advanced melanoma,” he said at the European Society for Medical Oncology Congress. The study results were published online in The Lancet Oncology to coincide with the presentation.
The benefit of immunotherapy also was seen in patients whose tumors had BRAF mutations, and both the combination and nivolumab alone showed improved efficacy compared with ipilimumab alone regardless of tumor expression of the programmed death ligand-1 (PD-L1), the investigators reported.
As previously reported, investigators in CheckMate 067 randomly assigned 945 previously untreated patients with unresectable stage III or IV melanoma to nivolumab 3 mg/kg every 2 weeks or nivolumab 1 mg/kg plus ipilimumab 3 mg/kg every 3 weeks for four doses then nivolumab 3 mg/kg every 2 weeks, or ipilimumab 3 mg/kg every 3 weeks for four doses. Patients were stratified at baseline by PD-L1 expression, BRAF status, and American Joint Commission on Cancer M stage.
Earlier results from the trial, reported at the 2015 annual meeting of the American Society of Clinical Oncology, showed that after a minimum of 9 months follow-up, the risk of disease progression or death was reduced by 43% with nivolumab versus ipilimumab (hazard ratio, 0.57; P less than .001) and by 58% with nivolumab plus ipilimumab vs. ipilimumab (HR, 0.42; P less than .001).
At ESMO 2018, Dr. Hodi presented 4-year follow-up results from the trial, with the analysis conducted at a minimum of 4 years after randomization of the last patient to be enrolled.
Median follow-up was 46.9 months for the nivolumab-plus-ipilimumab arm, 36 months in the nivolumab arm, and 18.6 months in the ipilimumab arm.
Median overall survival in the intention-to-treat population, a coprimary endpoint with progression-free survival (PFS), was as noted before. The HR for death with the combination compared with ipilimumab was 0.54 (P less than .0001) and for nivolumab versus ipilimumab it was 0.65 (P less than .0001).
The 4-year OS rates were 53% in the combination arm, 46% in the nivolumab-alone arm, and 30% in the ipilimumab-alone arm.
Median PFS was 11.5 months with the checkpoint inhibitor combination, 6.9 months in the nivolumab-alone arm, and 2.9 months in the ipilimumab arm.
The HR for PFS with the combination compared with ipilimumab was 0.42 (P less than .0001), and for nivolumab versus ipilimumab it was 0.53 (P less than .0001).
The safety analysis, conducted in all patients who received at least one dose of study drugs, showed that 59% of patients treated with the nivolumab/ipilimumab combination had treatment-related grade 3 or 4 adverse events, compared with 22% for patients treated with nivolumab alone, and 28% of those who received ipilimumab alone.
The most common treatment-related grade 3 adverse events were diarrhea in the combination and nivolumab-alone arms, and colitis in the ipilimumab group. In all three study arms the most common grade 4 adverse event was increased lipase.
Over the 4 years of follow-up, four patients died from treatment-related causes: one patient from cardiomyopathy and one from liver necrosis in the combination group, one from neutropenia in the nivolumab group, and one from colon perforation in the ipilimumab group. All of the deaths occurred within the first 3 years of the follow-up.
The investigators did not report on serious adverse events in the current analysis.
Invited discussant Reinhard Dummer, MD, of University Hospital Zurich Skin Cancer Center in Switzerland, said that while the study shows improved response rates and duration of response and longer PFS and OS with the combination, it’s premature to state conclusively that the combination is superior, because the study was not powered to compare efficacy between the two nivolumab-containing arms.
“So unfortunately, we have results, but we are not really convinced that the combination is so much better,” he said.
He added that the 4-year overall survival results for each arm show a consistent difference in the curves between the nivolumab and ipilimumab-alone arms. He also pointed to encouraging data showing that among patients alive at 4 years, 71% in the combination group did not require subsequent therapy, compared with 50% in the nivolumab group, and 39% in the ipilimumab group.
Dr. Hodi has received grant/research support from, and is a nonpaid consultant to, Bristol-Myers Squibb, which supported Checkmate 067. Dr. Dummer reported advising/consulting roles with the company.
SOURCE: Hodi FS et al. Lancet Oncol. 2018 Oct 22. doi: 10.1016/S1470-2045(18)30700-9.
MUNICH – Four years on, the combination of the immune checkpoint inhibitors nivolumab and ipilimumab as well as nivolumab alone continue to show benefit as first-line therapies for patients with advanced malignant melanoma, compared with ipilimumab monotherapy, reported investigators in the CheckMate O67 trial.
Among 945 patients with previously untreated and unresectable stage III or IV malignant melanoma, median overall survival at a minimum of 48 months follow-up had not been reached for patients assigned to the combination of nivolumab (Opdivo) and ipilimumab (Yervoy), compared with 36.9 months for patients assigned to nivolumab alone, and 19.9 months for patients assigned to ipilimumab alone, reported F. Stephen Hodi Jr., MD, of Dana-Farber Cancer Institute, Boston, and his colleagues.
“There’s a durable, sustained clinical benefit that can be achieved with first-line nivo plus ipi combination or nivo alone in patients with advanced melanoma,” he said at the European Society for Medical Oncology Congress. The study results were published online in The Lancet Oncology to coincide with the presentation.
The benefit of immunotherapy also was seen in patients whose tumors had BRAF mutations, and both the combination and nivolumab alone showed improved efficacy compared with ipilimumab alone regardless of tumor expression of the programmed death ligand-1 (PD-L1), the investigators reported.
As previously reported, investigators in CheckMate 067 randomly assigned 945 previously untreated patients with unresectable stage III or IV melanoma to nivolumab 3 mg/kg every 2 weeks or nivolumab 1 mg/kg plus ipilimumab 3 mg/kg every 3 weeks for four doses then nivolumab 3 mg/kg every 2 weeks, or ipilimumab 3 mg/kg every 3 weeks for four doses. Patients were stratified at baseline by PD-L1 expression, BRAF status, and American Joint Commission on Cancer M stage.
Earlier results from the trial, reported at the 2015 annual meeting of the American Society of Clinical Oncology, showed that after a minimum of 9 months follow-up, the risk of disease progression or death was reduced by 43% with nivolumab versus ipilimumab (hazard ratio, 0.57; P less than .001) and by 58% with nivolumab plus ipilimumab vs. ipilimumab (HR, 0.42; P less than .001).
At ESMO 2018, Dr. Hodi presented 4-year follow-up results from the trial, with the analysis conducted at a minimum of 4 years after randomization of the last patient to be enrolled.
Median follow-up was 46.9 months for the nivolumab-plus-ipilimumab arm, 36 months in the nivolumab arm, and 18.6 months in the ipilimumab arm.
Median overall survival in the intention-to-treat population, a coprimary endpoint with progression-free survival (PFS), was as noted before. The HR for death with the combination compared with ipilimumab was 0.54 (P less than .0001) and for nivolumab versus ipilimumab it was 0.65 (P less than .0001).
The 4-year OS rates were 53% in the combination arm, 46% in the nivolumab-alone arm, and 30% in the ipilimumab-alone arm.
Median PFS was 11.5 months with the checkpoint inhibitor combination, 6.9 months in the nivolumab-alone arm, and 2.9 months in the ipilimumab arm.
The HR for PFS with the combination compared with ipilimumab was 0.42 (P less than .0001), and for nivolumab versus ipilimumab it was 0.53 (P less than .0001).
The safety analysis, conducted in all patients who received at least one dose of study drugs, showed that 59% of patients treated with the nivolumab/ipilimumab combination had treatment-related grade 3 or 4 adverse events, compared with 22% for patients treated with nivolumab alone, and 28% of those who received ipilimumab alone.
The most common treatment-related grade 3 adverse events were diarrhea in the combination and nivolumab-alone arms, and colitis in the ipilimumab group. In all three study arms the most common grade 4 adverse event was increased lipase.
Over the 4 years of follow-up, four patients died from treatment-related causes: one patient from cardiomyopathy and one from liver necrosis in the combination group, one from neutropenia in the nivolumab group, and one from colon perforation in the ipilimumab group. All of the deaths occurred within the first 3 years of the follow-up.
The investigators did not report on serious adverse events in the current analysis.
Invited discussant Reinhard Dummer, MD, of University Hospital Zurich Skin Cancer Center in Switzerland, said that while the study shows improved response rates and duration of response and longer PFS and OS with the combination, it’s premature to state conclusively that the combination is superior, because the study was not powered to compare efficacy between the two nivolumab-containing arms.
“So unfortunately, we have results, but we are not really convinced that the combination is so much better,” he said.
He added that the 4-year overall survival results for each arm show a consistent difference in the curves between the nivolumab and ipilimumab-alone arms. He also pointed to encouraging data showing that among patients alive at 4 years, 71% in the combination group did not require subsequent therapy, compared with 50% in the nivolumab group, and 39% in the ipilimumab group.
Dr. Hodi has received grant/research support from, and is a nonpaid consultant to, Bristol-Myers Squibb, which supported Checkmate 067. Dr. Dummer reported advising/consulting roles with the company.
SOURCE: Hodi FS et al. Lancet Oncol. 2018 Oct 22. doi: 10.1016/S1470-2045(18)30700-9.
MUNICH – Four years on, the combination of the immune checkpoint inhibitors nivolumab and ipilimumab as well as nivolumab alone continue to show benefit as first-line therapies for patients with advanced malignant melanoma, compared with ipilimumab monotherapy, reported investigators in the CheckMate O67 trial.
Among 945 patients with previously untreated and unresectable stage III or IV malignant melanoma, median overall survival at a minimum of 48 months follow-up had not been reached for patients assigned to the combination of nivolumab (Opdivo) and ipilimumab (Yervoy), compared with 36.9 months for patients assigned to nivolumab alone, and 19.9 months for patients assigned to ipilimumab alone, reported F. Stephen Hodi Jr., MD, of Dana-Farber Cancer Institute, Boston, and his colleagues.
“There’s a durable, sustained clinical benefit that can be achieved with first-line nivo plus ipi combination or nivo alone in patients with advanced melanoma,” he said at the European Society for Medical Oncology Congress. The study results were published online in The Lancet Oncology to coincide with the presentation.
The benefit of immunotherapy also was seen in patients whose tumors had BRAF mutations, and both the combination and nivolumab alone showed improved efficacy compared with ipilimumab alone regardless of tumor expression of the programmed death ligand-1 (PD-L1), the investigators reported.
As previously reported, investigators in CheckMate 067 randomly assigned 945 previously untreated patients with unresectable stage III or IV melanoma to nivolumab 3 mg/kg every 2 weeks or nivolumab 1 mg/kg plus ipilimumab 3 mg/kg every 3 weeks for four doses then nivolumab 3 mg/kg every 2 weeks, or ipilimumab 3 mg/kg every 3 weeks for four doses. Patients were stratified at baseline by PD-L1 expression, BRAF status, and American Joint Commission on Cancer M stage.
Earlier results from the trial, reported at the 2015 annual meeting of the American Society of Clinical Oncology, showed that after a minimum of 9 months follow-up, the risk of disease progression or death was reduced by 43% with nivolumab versus ipilimumab (hazard ratio, 0.57; P less than .001) and by 58% with nivolumab plus ipilimumab vs. ipilimumab (HR, 0.42; P less than .001).
At ESMO 2018, Dr. Hodi presented 4-year follow-up results from the trial, with the analysis conducted at a minimum of 4 years after randomization of the last patient to be enrolled.
Median follow-up was 46.9 months for the nivolumab-plus-ipilimumab arm, 36 months in the nivolumab arm, and 18.6 months in the ipilimumab arm.
Median overall survival in the intention-to-treat population, a coprimary endpoint with progression-free survival (PFS), was as noted before. The HR for death with the combination compared with ipilimumab was 0.54 (P less than .0001) and for nivolumab versus ipilimumab it was 0.65 (P less than .0001).
The 4-year OS rates were 53% in the combination arm, 46% in the nivolumab-alone arm, and 30% in the ipilimumab-alone arm.
Median PFS was 11.5 months with the checkpoint inhibitor combination, 6.9 months in the nivolumab-alone arm, and 2.9 months in the ipilimumab arm.
The HR for PFS with the combination compared with ipilimumab was 0.42 (P less than .0001), and for nivolumab versus ipilimumab it was 0.53 (P less than .0001).
The safety analysis, conducted in all patients who received at least one dose of study drugs, showed that 59% of patients treated with the nivolumab/ipilimumab combination had treatment-related grade 3 or 4 adverse events, compared with 22% for patients treated with nivolumab alone, and 28% of those who received ipilimumab alone.
The most common treatment-related grade 3 adverse events were diarrhea in the combination and nivolumab-alone arms, and colitis in the ipilimumab group. In all three study arms the most common grade 4 adverse event was increased lipase.
Over the 4 years of follow-up, four patients died from treatment-related causes: one patient from cardiomyopathy and one from liver necrosis in the combination group, one from neutropenia in the nivolumab group, and one from colon perforation in the ipilimumab group. All of the deaths occurred within the first 3 years of the follow-up.
The investigators did not report on serious adverse events in the current analysis.
Invited discussant Reinhard Dummer, MD, of University Hospital Zurich Skin Cancer Center in Switzerland, said that while the study shows improved response rates and duration of response and longer PFS and OS with the combination, it’s premature to state conclusively that the combination is superior, because the study was not powered to compare efficacy between the two nivolumab-containing arms.
“So unfortunately, we have results, but we are not really convinced that the combination is so much better,” he said.
He added that the 4-year overall survival results for each arm show a consistent difference in the curves between the nivolumab and ipilimumab-alone arms. He also pointed to encouraging data showing that among patients alive at 4 years, 71% in the combination group did not require subsequent therapy, compared with 50% in the nivolumab group, and 39% in the ipilimumab group.
Dr. Hodi has received grant/research support from, and is a nonpaid consultant to, Bristol-Myers Squibb, which supported Checkmate 067. Dr. Dummer reported advising/consulting roles with the company.
SOURCE: Hodi FS et al. Lancet Oncol. 2018 Oct 22. doi: 10.1016/S1470-2045(18)30700-9.
REPORTING FROM ESMO 2018
Key clinical point: Nivolumab and ipilimumab combined provide superior progression-free and overall survival compared with nivolumab or ipilimumab alone.
Major finding: At 4-year minimum follow-up the median overall survival with the combination had not be reached, vs. 36.9 months for nivolumab and 19.9 months for ipilimumab.
Study details: Randomized phase 3 trial of 945 patients with previously untreated stage III or IV malignant melanoma.
Disclosures: Dr. Hodi has received grant/research support from, and is a nonpaid consultant to, Bristol-Myers Squibb, which supported Checkmate 067. Dr. Dummer reported advising/consulting roles with the company.
Source: Hodi FS et al. Lancet Oncol. 2018 Oct 22. doi: 10.1016/S1470-2045(18)30700-9.
Checkpoint inhibitor doubles 3-year survival rate of BRAF wild-type advanced melanoma
For previously untreated patients with BRAF wild-type advanced melanoma, the antiprogrammed death-ligand 1 agent nivolumab dramatically improved overall survival (OS), compared with standard first-line chemotherapy, according to new data from the CheckMate 066 trial.
At the 3-year follow-up, more than twice as many nivolumab-treated patients were alive, compared with those who received dacarbazine, reported lead author Paolo A. Ascierto, MD, director of the unit of melanoma, cancer immunotherapy, and innovative therapy at the Istituto Nazionale Tumori Fondazione Pascale in Naples, Italy, and his colleagues. Longer median progression-free survival (PFS) and higher complete response rates were also reported.
“The results of this 3-year follow-up analysis provided evidence for a durable survival benefit with nivolumab monotherapy in patients with previously untreated BRAF wild-type advanced melanoma,” the investigators wrote in JAMA Oncology.
The double-blind, phase 3 trial involved 418 patients with unresectable, previously untreated stage III or IV melanoma not exhibiting a BRAF mutation. Patients were randomized to receive either nivolumab (n = 210; 3 mg/kg every 2 weeks plus placebo every 3 weeks) or dacarbazine (n = 208; 1,000 mg/m2 every 3 weeks plus placebo every 2 weeks). Treatment continued until unacceptable toxicity or disease progression occurred.
The results showed dramatic benefits, measured by OS and PFS, when patients were treated with nivolumab. The 3-year OS rate was 51.2% in the nivolumab group, compared with 21.6% in the dacarbazine group, an approximate 130% difference. Median OS and median PFS were also multiplied by the checkpoint inhibitor, showing 230% and 130% improvements, respectively (OS, 37.5 months vs. 11.2 months; PFS, 5.1 months vs. 2.2 months; P less than .001). Grade 3 or higher treatment-related adverse events (AEs) were comparable between treatment arms (nivolumab, 15.0% vs. dacarbazine, 17.6%).
“Responses to nivolumab were long lasting in many patients who discontinued treatment, with most patients who stopped treatment still alive and without disease progression at the time of the last assessment,” the investigators wrote.
“Collectively, our results showed durable responses and long-term survival with nivolumab monotherapy, with no new AEs developing at late time points,” they concluded.
The study was funded by Bristol-Myers Squibb. The authors reported financial relationships with Bristol-Myers Squibb, Roche/Genentech, Novartis, Amgen, and others.
SOURCE: Ascierto PA et al. JAMA Oncol. 2018 Oct 25. doi: 10.1001/jamaoncol.2018.4514.
For previously untreated patients with BRAF wild-type advanced melanoma, the antiprogrammed death-ligand 1 agent nivolumab dramatically improved overall survival (OS), compared with standard first-line chemotherapy, according to new data from the CheckMate 066 trial.
At the 3-year follow-up, more than twice as many nivolumab-treated patients were alive, compared with those who received dacarbazine, reported lead author Paolo A. Ascierto, MD, director of the unit of melanoma, cancer immunotherapy, and innovative therapy at the Istituto Nazionale Tumori Fondazione Pascale in Naples, Italy, and his colleagues. Longer median progression-free survival (PFS) and higher complete response rates were also reported.
“The results of this 3-year follow-up analysis provided evidence for a durable survival benefit with nivolumab monotherapy in patients with previously untreated BRAF wild-type advanced melanoma,” the investigators wrote in JAMA Oncology.
The double-blind, phase 3 trial involved 418 patients with unresectable, previously untreated stage III or IV melanoma not exhibiting a BRAF mutation. Patients were randomized to receive either nivolumab (n = 210; 3 mg/kg every 2 weeks plus placebo every 3 weeks) or dacarbazine (n = 208; 1,000 mg/m2 every 3 weeks plus placebo every 2 weeks). Treatment continued until unacceptable toxicity or disease progression occurred.
The results showed dramatic benefits, measured by OS and PFS, when patients were treated with nivolumab. The 3-year OS rate was 51.2% in the nivolumab group, compared with 21.6% in the dacarbazine group, an approximate 130% difference. Median OS and median PFS were also multiplied by the checkpoint inhibitor, showing 230% and 130% improvements, respectively (OS, 37.5 months vs. 11.2 months; PFS, 5.1 months vs. 2.2 months; P less than .001). Grade 3 or higher treatment-related adverse events (AEs) were comparable between treatment arms (nivolumab, 15.0% vs. dacarbazine, 17.6%).
“Responses to nivolumab were long lasting in many patients who discontinued treatment, with most patients who stopped treatment still alive and without disease progression at the time of the last assessment,” the investigators wrote.
“Collectively, our results showed durable responses and long-term survival with nivolumab monotherapy, with no new AEs developing at late time points,” they concluded.
The study was funded by Bristol-Myers Squibb. The authors reported financial relationships with Bristol-Myers Squibb, Roche/Genentech, Novartis, Amgen, and others.
SOURCE: Ascierto PA et al. JAMA Oncol. 2018 Oct 25. doi: 10.1001/jamaoncol.2018.4514.
For previously untreated patients with BRAF wild-type advanced melanoma, the antiprogrammed death-ligand 1 agent nivolumab dramatically improved overall survival (OS), compared with standard first-line chemotherapy, according to new data from the CheckMate 066 trial.
At the 3-year follow-up, more than twice as many nivolumab-treated patients were alive, compared with those who received dacarbazine, reported lead author Paolo A. Ascierto, MD, director of the unit of melanoma, cancer immunotherapy, and innovative therapy at the Istituto Nazionale Tumori Fondazione Pascale in Naples, Italy, and his colleagues. Longer median progression-free survival (PFS) and higher complete response rates were also reported.
“The results of this 3-year follow-up analysis provided evidence for a durable survival benefit with nivolumab monotherapy in patients with previously untreated BRAF wild-type advanced melanoma,” the investigators wrote in JAMA Oncology.
The double-blind, phase 3 trial involved 418 patients with unresectable, previously untreated stage III or IV melanoma not exhibiting a BRAF mutation. Patients were randomized to receive either nivolumab (n = 210; 3 mg/kg every 2 weeks plus placebo every 3 weeks) or dacarbazine (n = 208; 1,000 mg/m2 every 3 weeks plus placebo every 2 weeks). Treatment continued until unacceptable toxicity or disease progression occurred.
The results showed dramatic benefits, measured by OS and PFS, when patients were treated with nivolumab. The 3-year OS rate was 51.2% in the nivolumab group, compared with 21.6% in the dacarbazine group, an approximate 130% difference. Median OS and median PFS were also multiplied by the checkpoint inhibitor, showing 230% and 130% improvements, respectively (OS, 37.5 months vs. 11.2 months; PFS, 5.1 months vs. 2.2 months; P less than .001). Grade 3 or higher treatment-related adverse events (AEs) were comparable between treatment arms (nivolumab, 15.0% vs. dacarbazine, 17.6%).
“Responses to nivolumab were long lasting in many patients who discontinued treatment, with most patients who stopped treatment still alive and without disease progression at the time of the last assessment,” the investigators wrote.
“Collectively, our results showed durable responses and long-term survival with nivolumab monotherapy, with no new AEs developing at late time points,” they concluded.
The study was funded by Bristol-Myers Squibb. The authors reported financial relationships with Bristol-Myers Squibb, Roche/Genentech, Novartis, Amgen, and others.
SOURCE: Ascierto PA et al. JAMA Oncol. 2018 Oct 25. doi: 10.1001/jamaoncol.2018.4514.
FROM JAMA ONCOLOGY
Key clinical point: For previously untreated patients with BRAF wild-type advanced melanoma, the checkpoint inhibitor nivolumab dramatically improved overall survival, compared with standard first-line chemotherapy.
Major finding: The 3-year overall survival rate for patients treated with nivolumab was 51.2%, compared with 21.6% for those who received dacarbazine.
Study details: CheckMate 066 is an ongoing, phase 3, double-blind trial involving 418 patients with BRAF wild-type advanced melanoma.
Disclosures: The study was funded by Bristol-Myers Squibb. The authors reported financial relationships with Bristol-Myers Squibb, Roche/Genentech, Novartis, Amgen, and others.
Source: Ascierto PA et al. JAMA Oncol. 2018 Oct 25. doi: 10.1001/jamaoncol.2018.4514.
Checkpoint inhibitor seems safe and effective for patients with HIV
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
MUNICH – Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent, investigators found.
The retrospective study also showed that viral load and CD4 status were largely unchanged by immunotherapy, lead author Aurélien Gobert, MD, of Groupe Hospitalier Pitié Salpêtrière, Paris, reported at the European Society for Medical Oncology Congress.
HIV increases risks of certain cancer types, Dr. Gobert said in a press release. “These patients are at higher risk for a number of cancers: AIDS-defining forms, the diagnosis of which results in the categorization of a person as suffering from AIDS, but also various other types that they are two to three times more likely to develop than in the general population, such as anal, skin, head and neck, and lung cancer,” he said.
Despite the increased risks, few studies have evaluated cancer treatments for patients with HIV due to exclusions from most clinical trials. As HIV is an immune-based disease, concerns have arisen surrounding the safety and efficacy of using anti-neoplastic immunotherapies for HIV-positive patients. Considering that millions of people worldwide are HIV positive, research in this area can have real-world consequences.
Dr. Gobert and his colleagues analyzed data from CANCERVIH, a French national database of patients with cancer and HIV. Since May 2014, nivolumab has been administered to 20 patients. Nineteen had metastatic non–small-cell lung cancer and 1 had metastatic melanoma. At diagnosis, the median CD4 count was 338.5 per cubic millimeter. Seventeen patients had undetectable viral load, two had fewer than 40 copies per millimeter, and one patient’s viral load was unknown. Dr. Gobert described the population as “demographically homogenous,” with “most patients being males around 60 years old.”
Analysis showed that nivolumab had little impact on CD4 count or viral load. One patient had a decreased CD4 count and an increased viral load, but this occurred during an interruption to antiretroviral therapy, which clouds potential associations with nivolumab. No immune-related adverse events or deaths due to drug toxicity occurred. Efficacy was assessed in 17 patients: Four (24%) showed a partial response, 2 (12%) had stable disease, and 11 (64%) had disease progression.
“Based on these preliminary data, treatment with anti-PD-1 ... seems to be feasible in people with HIV,” Dr. Gobert reported. He added that “antiretroviral therapy should not be interrupted.”
In a comment for ESMO, John Haanen, PhD, of the Netherlands Cancer Institute, Amsterdam, said that the results “confirm those of other, smaller cohorts in showing that while on antiretroviral therapy, cancer patients living with HIV can safely receive anti-PD-1 treatment. The efficacy data also suggests that the overall response rate of HIV-positive patients seems to be similar to that of other cancer patients. These promising results need to be confirmed in larger studies – ideally, in a prospective clinical trial.”
Principal investigator Jean-Philippe Spano, MD, PhD, disclosed relationships with Gilead, Roche, BMS, and others.
SOURCE: Gobert et al. ESMO 2018, Abstract 1213P_PR.
REPORTING FROM ESMO 2018
Key clinical point: Patients with HIV who are treated with nivolumab, a programmed death-1 (PD-1) inhibitor, appear to have similar safety and efficacy outcomes compared with HIV-negative patients treated with the same agent.
Major finding: No immune-related adverse events or deaths due to drug toxicity occurred.
Study details: A retrospective analysis of 20 patients with HIV who received nivolumab immunotherapy.
Disclosures: Jean-Philippe Spano, principal investigator, disclosed relationships with Gilead, Roche, BMS, and others.
Source: Gobert et al. ESMO 2018, Abstract 1213P_PR.
Cross-contamination of Pathology Specimens: A Cautionary Tale
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.
The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.
For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.
Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.

The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.

For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.

Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
Cross-contamination of pathology specimens is a rare but nonnegligible source of potential morbidity in clinical practice. Contaminant tissue fragments, colloquially referred to as floaters, typically are readily identifiable based on obvious cytomorphologic differences, especially if the tissues arise from different organs; however, one cannot rely on such distinctions in a pathology laboratory dedicated to a single organ system (eg, dermatopathology). The inability to identify quickly and confidently the presence of a contaminant puts the patient at risk for misdiagnosis, which can lead to unnecessary morbidity or even mortality in the case of cancer misdiagnosis. Studies that have been conducted to estimate the incidence of this type of error have suggested an overall incidence rate between approximately 1% and 3%.1,2 Awareness of this phenomenon and careful scrutiny when the histopathologic evidence diverges considerably from the clinical impression is critical for minimizing the negative outcomes that could result from the presence of contaminant tissue. We present a case in which cross-contamination of a pathology specimen led to an initial erroneous diagnosis of an aggressive cutaneous melanoma in a patient with a benign adnexal neoplasm.
Case Report
A 72-year-old man was referred to the Pigmented Lesion and Melanoma Program at Stanford University Medical Center and Cancer Institute (Palo Alto, California) for evaluation and treatment of a presumed stage IIB melanoma on the right preauricular cheek based on a shave biopsy that had been performed (<1 month prior) by his local dermatology provider and subsequently read by an affiliated out-of-state dermatopathology laboratory. Per the clinical history that was gathered at the current presentation, neither the patient nor his wife had noticed the lesion prior to his dermatology provider pointing it out on the day of the biopsy. Additionally, he denied associated pain, bleeding, or ulceration. According to outside medical records, the referring dermatology provider described the lesion as a 4-mm pink pearly papule with telangiectasia favoring a diagnosis of basal cell carcinoma, and a diagnostic shave biopsy was performed. On presentation to our clinic, physical examination of the right preauricular cheek revealed a 4×3-mm depressed erythematous scar with no evidence of residual pigmentation or nodularity (Figure 1). There was no clinically appreciable regional lymphadenopathy.

The original dermatopathology report indicated an invasive melanoma with the following pathologic characteristics: superficial spreading type, Breslow depth of at least 2.16 mm, ulceration, and a mitotic index of 8 mitotic figures/mm2 with transection of the invasive component at the peripheral and deep margins. There was no evidence of regression, perineural invasion, lymphovascular invasion, or microsatellites. Interestingly, the report indicated that there also was a basaloid proliferation with features of cylindroma in the same pathology slide adjacent to the aggressive invasive melanoma that was described. Given the complexity of cases referred to our academic center, the standard of care includes internal dermatopathology review of all outside pathology specimens. This review proved critical to this patient’s care in light of the considerable divergence of the initial pathologic diagnosis and the reported clinical features of the lesion.
Internal review of the single pathology slide received from the referring provider showed a total of 4 sections, 3 of which are shown here (Figure 2A). Three sections, including the one not shown, were all consistent with a diagnosis of cylindroma and showed no evidence of a melanocytic proliferation (Figure 2B). However, the fourth section demonstrated marked morphologic dissimilarity compared to the other 3 sections. This outlier section showed a thick cutaneous melanoma with a Breslow depth of at least 2.1 mm, ulceration, a mitotic rate of 12 mitotic figures/mm2, and broad transection of the invasive component at the peripheral and deep margins (Figures 2C and 2D). Correlation with the gross description of tissue processing on the original pathology report indicating that the specimen had been trisected raised suspicion that the fourth and very dissimilar section could be a contaminant from another source that was incorporated into our patient’s histologic sections during processing. Taken together, these discrepancies made the diagnosis of cylindroma alone far more likely than cutaneous melanoma, but we needed conclusive evidence given the dramatic difference in prognosis and management between a cylindroma and an aggressive cutaneous melanoma.

For further diagnostic clarification, we performed polymorphic short tandem repeat (STR) analysis, a well-described forensic pathology technique, to determine if the melanoma and cylindroma specimens derived from different patients, as we hypothesized. This analysis revealed differences in all but one DNA locus tested between the cylindroma specimen and the melanoma specimen, confirming our hypothesis (Figure 3). Subsequent discussion of the case with staff from the dermatopathology laboratory that processed this specimen provided further support for our suspicion that the invasive melanoma specimen was part of a case processed prior to our patient’s benign lesion. Therefore, the wide local excision for treatment of the suspected melanoma fortunately was canceled, and the patient did not require further treatment of the benign cylindroma. The patient expressed relief and gratitude for this critical clarification and change in management.

Comment
Shah et al3 reported a similar case in which a benign granuloma of the lung masqueraded as a squamous cell carcinoma due to histopathologic contamination. Although few similar cases have been described in the literature, the risk posed by such contamination is remarkable, regardless of whether it occurs during specimen grossing, embedding, sectioning, or staining.1,4,5 This risk is amplified in facilities that process specimens originating predominantly from a single organ system or tissue type, as is often the case in dedicated dermatopathology laboratories. In this scenario, it is unlikely that one could use the presence of tissues from 2 different organ systems on a single slide as a way of easily recognizing the presence of a contaminant and rectifying the error. Additionally, the presence of malignant cells in the contaminant further complicates the problem and requires an investigation that can conclusively distinguish the contaminant from the patient’s actual tissue.
In our case, our dermatology and dermatopathology teams partnered with our molecular pathology team to find a solution. Polymorphic STR analysis via polymerase chain reaction amplification is a sensitive method employed commonly in forensic DNA laboratories for determining whether a sample submitted as evidence belongs to a given suspect.6 Although much more commonly used in forensics, STR analysis does have known roles in clinical medicine, such as chimerism testing after bone marrow or allogeneic stem cell transplantation.7 Given the relatively short period of time it takes along with the convenience of commercially available kits, a high discriminative ability, and well-validated interpretation procedures, STR analysis is an excellent method for determining if a given tissue sample came from a given patient, which is what was needed in our case.
The combined clinical, histopathologic, and molecular data in our case allowed for confident clarification of our patient’s diagnosis, sparing him the morbidity of wide local excision on the face, sentinel lymph node biopsy, and emotional distress associated with a diagnosis of aggressive cutaneous melanoma. Our case highlights the critical importance of internal review of pathology specimens in ensuring proper diagnosis and management and reminds us that, though rare, accidental contamination during processing of pathology specimens is a potential adverse event that must be considered, especially when a pathologic finding diverges considerably from what is anticipated based on the patient’s history and physical examination.
Acknowledgment
The authors express gratitude to the patient described herein who graciously provided permission for us to publish his case and clinical photography.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
- Gephardt GN, Zarbo RJ. Extraneous tissue in surgical pathology: a College of American Pathologists Q-Probes study of 275 laboratories. Arch Pathol Lab Med. 1996;120:1009-1014.
- Alam M, Shah AD, Ali S, et al. Floaters in Mohs micrographic surgery [published online June 27, 2013]. Dermatol Surg. 2013;39:1317-1322.
- Shah PA, Prat MP, Hostler DC. Benign granuloma masquerading as squamous cell carcinoma due to a “floater.” Hawaii J Med Public Health. 2017;76(11, suppl 2):19-21.
- Platt E, Sommer P, McDonald L, et al. Tissue floaters and contaminants in the histology laboratory. Arch Pathol Lab Med. 2009;133:973-978.
- Layfield LJ, Witt BL, Metzger KG, et al. Extraneous tissue: a potential source for diagnostic error in surgical pathology. Am J Clin Pathol. 2011;136:767-772.
- Butler JM. Forensic DNA testing. Cold Spring Harb Protoc. 2011;2011:1438-1450.
- Manasatienkij C, Ra-ngabpai C. Clinical application of forensic DNA analysis: a literature review. J Med Assoc Thai. 2012;95:1357-1363.
Resident Pearl
- Although cross-contamination of pathology specimens is rare, it does occur and can impact diagnosis and management if detected early.
Which Patients Have the Best Chance With Checkpoint Inhibitors?
Checkpoint inhibitors are so new that not enough patients have received them to allow clinicians to predict who will benefit most. But researchers from the National Cancer Institute, Center for Cancer Institute; Harvard University in Cambridge, Massachusetts; University of Pennsylvania in Philadelphia; and University of Maryland in College Park may have found a clue: A gene expression predictor.
They began by looking at neuroblastoma cases where the immune system seemed to mount “an unprompted, successful immune response” to cancer, causing spontaneous tumor regression. The researchers were able to define gene expression features that separated regressing from nonregressing disease.
The researchers then computed Immuno-PREdictive Scores (IMPRES) for each patient sample. The higher the score, the more likely was spontaneous regression. Analyzing 297 samples from several studies, they found the predictor identified nearly all patients who responded to the inhibitors and more than half of those who did not. “Importantly,” the researchers say, their predictor was accurate across many different melanoma patient datasets.
Checkpoint inhibitors are so new that not enough patients have received them to allow clinicians to predict who will benefit most. But researchers from the National Cancer Institute, Center for Cancer Institute; Harvard University in Cambridge, Massachusetts; University of Pennsylvania in Philadelphia; and University of Maryland in College Park may have found a clue: A gene expression predictor.
They began by looking at neuroblastoma cases where the immune system seemed to mount “an unprompted, successful immune response” to cancer, causing spontaneous tumor regression. The researchers were able to define gene expression features that separated regressing from nonregressing disease.
The researchers then computed Immuno-PREdictive Scores (IMPRES) for each patient sample. The higher the score, the more likely was spontaneous regression. Analyzing 297 samples from several studies, they found the predictor identified nearly all patients who responded to the inhibitors and more than half of those who did not. “Importantly,” the researchers say, their predictor was accurate across many different melanoma patient datasets.
Checkpoint inhibitors are so new that not enough patients have received them to allow clinicians to predict who will benefit most. But researchers from the National Cancer Institute, Center for Cancer Institute; Harvard University in Cambridge, Massachusetts; University of Pennsylvania in Philadelphia; and University of Maryland in College Park may have found a clue: A gene expression predictor.
They began by looking at neuroblastoma cases where the immune system seemed to mount “an unprompted, successful immune response” to cancer, causing spontaneous tumor regression. The researchers were able to define gene expression features that separated regressing from nonregressing disease.
The researchers then computed Immuno-PREdictive Scores (IMPRES) for each patient sample. The higher the score, the more likely was spontaneous regression. Analyzing 297 samples from several studies, they found the predictor identified nearly all patients who responded to the inhibitors and more than half of those who did not. “Importantly,” the researchers say, their predictor was accurate across many different melanoma patient datasets.
Epacadostat plus pembrolizumab shows promise in advanced solid tumors
Epacadostat, a highly selective oral inhibitor of the indoleamine 2,3-dioxygenase 1 (IDO1) enzyme, was well tolerated when combined with pembrolizumab and demonstrated encouraging antitumor activity in multiple types of advanced solid tumors, according to the results of a phase l/ll trial.
Tumors may evade immunosurveillance through upregulation of the IDO1 enzyme, and thus there is a great interest in developing combination therapies that can target various immune evasion pathways to improve therapeutic response and outcomes. In this study, the authors evaluated the investigational agent epacadostat combined with pembrolizumab in 62 patients with advanced solid tumors.
In the dose escalation phase, patents received increasing doses of oral epacadostat (25, 50, 100, or 300 mg) twice per day plus intravenous pembrolizumab 2 mg/kg or 200 mg every 3 weeks. During the safety expansion, epacadostat at 50, 100, or 300 mg was given twice per day, plus pembrolizumab 200 mg every 3 weeks. The maximum tolerated dose of epacadostat in combination with pembrolizumab was not reached.
Objective responses (per Response Evaluation Criteria in Solid Tumors [RECIST] version 1.1) occurred in 12 (55%) of 22 patients with melanoma and in patients with non–small-cell lung cancer, renal cell carcinoma, endometrial adenocarcinoma, urothelial carcinoma, and squamous cell carcinoma of the head and neck, reported Tara C. Mitchell, MD, of the Abramson Cancer Center, University of Pennsylvania, Philadelphia, and her colleagues. The report is in the Journal of Clinical Oncology.
The authors observed that there was antitumor activity at all epacadostat doses and in several tumor types. A complete response was achieved by 8 patients (treatment naive melanoma [5 patients] and previously treated for advanced/ metastatic melanoma, endometrial adenocarcinoma [EA], or urothelial carcinoma [UC] [1 patient each]), while 17 patients achieved a partial response (treatment-naive melanoma [6 patients], non–small cell lung cancer [NSCLC] [5 patients], renal cell carcinoma [RCC] and UC [2 patients each], and EA and squamous cell carcinoma of the head and neck [1 patient each]).
Most patients (n = 52, 84%) experienced treatment-related adverse events (TRAEs), the most frequently observed being fatigue (36%), rash (36%), arthralgia (24%), pruritus (23%), and nausea (21%). Grade 3/4 TRAEs occurred in 24% of patients, and 7 patients (11%) discontinued their treatment because of TRAEs. There were no deaths associated with TRAEs.
“The safety profile observed with epacadostat plus pembrolizumab compares favorably with studies of other combination immunotherapies,” wrote Dr. Mitchell and her colleagues. “Although not powered to evaluate efficacy, the phase I portion of this study showed that epacadostat plus pembrolizumab had encouraging and durable antitumor activity,” they said.
SOURCE: Mitchell TC et al. J Clin Oncol. 2018 Sep 28. doi: 10.1200/JCO.2018.78.9602.
Epacadostat, a highly selective oral inhibitor of the indoleamine 2,3-dioxygenase 1 (IDO1) enzyme, was well tolerated when combined with pembrolizumab and demonstrated encouraging antitumor activity in multiple types of advanced solid tumors, according to the results of a phase l/ll trial.
Tumors may evade immunosurveillance through upregulation of the IDO1 enzyme, and thus there is a great interest in developing combination therapies that can target various immune evasion pathways to improve therapeutic response and outcomes. In this study, the authors evaluated the investigational agent epacadostat combined with pembrolizumab in 62 patients with advanced solid tumors.
In the dose escalation phase, patents received increasing doses of oral epacadostat (25, 50, 100, or 300 mg) twice per day plus intravenous pembrolizumab 2 mg/kg or 200 mg every 3 weeks. During the safety expansion, epacadostat at 50, 100, or 300 mg was given twice per day, plus pembrolizumab 200 mg every 3 weeks. The maximum tolerated dose of epacadostat in combination with pembrolizumab was not reached.
Objective responses (per Response Evaluation Criteria in Solid Tumors [RECIST] version 1.1) occurred in 12 (55%) of 22 patients with melanoma and in patients with non–small-cell lung cancer, renal cell carcinoma, endometrial adenocarcinoma, urothelial carcinoma, and squamous cell carcinoma of the head and neck, reported Tara C. Mitchell, MD, of the Abramson Cancer Center, University of Pennsylvania, Philadelphia, and her colleagues. The report is in the Journal of Clinical Oncology.
The authors observed that there was antitumor activity at all epacadostat doses and in several tumor types. A complete response was achieved by 8 patients (treatment naive melanoma [5 patients] and previously treated for advanced/ metastatic melanoma, endometrial adenocarcinoma [EA], or urothelial carcinoma [UC] [1 patient each]), while 17 patients achieved a partial response (treatment-naive melanoma [6 patients], non–small cell lung cancer [NSCLC] [5 patients], renal cell carcinoma [RCC] and UC [2 patients each], and EA and squamous cell carcinoma of the head and neck [1 patient each]).
Most patients (n = 52, 84%) experienced treatment-related adverse events (TRAEs), the most frequently observed being fatigue (36%), rash (36%), arthralgia (24%), pruritus (23%), and nausea (21%). Grade 3/4 TRAEs occurred in 24% of patients, and 7 patients (11%) discontinued their treatment because of TRAEs. There were no deaths associated with TRAEs.
“The safety profile observed with epacadostat plus pembrolizumab compares favorably with studies of other combination immunotherapies,” wrote Dr. Mitchell and her colleagues. “Although not powered to evaluate efficacy, the phase I portion of this study showed that epacadostat plus pembrolizumab had encouraging and durable antitumor activity,” they said.
SOURCE: Mitchell TC et al. J Clin Oncol. 2018 Sep 28. doi: 10.1200/JCO.2018.78.9602.
Epacadostat, a highly selective oral inhibitor of the indoleamine 2,3-dioxygenase 1 (IDO1) enzyme, was well tolerated when combined with pembrolizumab and demonstrated encouraging antitumor activity in multiple types of advanced solid tumors, according to the results of a phase l/ll trial.
Tumors may evade immunosurveillance through upregulation of the IDO1 enzyme, and thus there is a great interest in developing combination therapies that can target various immune evasion pathways to improve therapeutic response and outcomes. In this study, the authors evaluated the investigational agent epacadostat combined with pembrolizumab in 62 patients with advanced solid tumors.
In the dose escalation phase, patents received increasing doses of oral epacadostat (25, 50, 100, or 300 mg) twice per day plus intravenous pembrolizumab 2 mg/kg or 200 mg every 3 weeks. During the safety expansion, epacadostat at 50, 100, or 300 mg was given twice per day, plus pembrolizumab 200 mg every 3 weeks. The maximum tolerated dose of epacadostat in combination with pembrolizumab was not reached.
Objective responses (per Response Evaluation Criteria in Solid Tumors [RECIST] version 1.1) occurred in 12 (55%) of 22 patients with melanoma and in patients with non–small-cell lung cancer, renal cell carcinoma, endometrial adenocarcinoma, urothelial carcinoma, and squamous cell carcinoma of the head and neck, reported Tara C. Mitchell, MD, of the Abramson Cancer Center, University of Pennsylvania, Philadelphia, and her colleagues. The report is in the Journal of Clinical Oncology.
The authors observed that there was antitumor activity at all epacadostat doses and in several tumor types. A complete response was achieved by 8 patients (treatment naive melanoma [5 patients] and previously treated for advanced/ metastatic melanoma, endometrial adenocarcinoma [EA], or urothelial carcinoma [UC] [1 patient each]), while 17 patients achieved a partial response (treatment-naive melanoma [6 patients], non–small cell lung cancer [NSCLC] [5 patients], renal cell carcinoma [RCC] and UC [2 patients each], and EA and squamous cell carcinoma of the head and neck [1 patient each]).
Most patients (n = 52, 84%) experienced treatment-related adverse events (TRAEs), the most frequently observed being fatigue (36%), rash (36%), arthralgia (24%), pruritus (23%), and nausea (21%). Grade 3/4 TRAEs occurred in 24% of patients, and 7 patients (11%) discontinued their treatment because of TRAEs. There were no deaths associated with TRAEs.
“The safety profile observed with epacadostat plus pembrolizumab compares favorably with studies of other combination immunotherapies,” wrote Dr. Mitchell and her colleagues. “Although not powered to evaluate efficacy, the phase I portion of this study showed that epacadostat plus pembrolizumab had encouraging and durable antitumor activity,” they said.
SOURCE: Mitchell TC et al. J Clin Oncol. 2018 Sep 28. doi: 10.1200/JCO.2018.78.9602.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Epacadostat plus pembrolizumab showed antitumor activity and tolerability in patients with advanced solid tumors.
Major finding: Among 62 patients, 25 achieved an objective response.
Study details: Phase l/ll clinical trial of 62 patients with advanced solid tumors.
Disclosures: Incyte and Merck funded the study. All of the authors have disclosed relationships with industry, including the study sponsor.
Source: Mitchell TC et al. J Clin Oncol. 2018 Sep 28. doi: 10.1200/JCO.2018.78.9602.