User login
Cancer burden: Multiple metrics needed to clarify the big picture
A new analysis of 40 years of U.S. cancer data underscores the importance of looking at multiple metrics to discern the complex interplay of factors influencing cancer burden in the population. Findings showed that the epidemiologic signature – a composite of two or three key metrics – differed across cancer types and was favorable in some cases and unfavorable in others.
“Epidemiologic signatures that illustrate trends in population-based data on cancer burden provide insight into true cancer occurrence, overdiagnosis, and treatment advances,” explain the analysts, led by H. Gilbert Welch, MD, MPH, Center for Surgery and Public Health, Brigham and Women’s Hospital, Boston. “They are important indicators of the potential contribution of environmental exposures, primary preventive interventions, new treatments, and changing diagnostic and screening practices.”
Dr. Welch and colleagues analyzed data for the years 1975 through 2015, assessing juxtaposed trends in incidence, mortality, and, when available, metastatic incidence (cancer already metastatic at diagnosis) for 11 cancers individually and for all cancers combined. Incidence data combining invasive and in situ cancers were obtained from the original nine Surveillance, Epidemiology, and End Results (SEER) registries, and mortality data were obtained from the National Vital Statistics System.
The analysts then explored implications of the epidemiologic signatures as they pertain to true cancer occurrence (the underlying incidence of clinically meaningful cancer), overdiagnosis (detection of cancers that will not cause symptoms or death), and treatment advances.
Individual cancers
Findings of the analysis, published in a special report in the New England Journal of Medicine, revealed three broad categories of epidemiologic signatures having different implications for the public health and oncology fields.
Desirable signatures showed, for example, declining mortality against a backdrop of stable incidence over the 40-year period, signaling improved treatment, as seen for chronic myeloid leukemia following introduction of imatinib (Gleevec), according to the analysts. Lung cancer incidence and mortality rose and fell in tandem, reflecting an increase in smoking followed by a decrease in response to prevention efforts. Stomach, cervical, and colorectal cancers had both falling incidence – likely reflecting a true decline in occurrence related to prevention and/or screening detection and subsequent treatment of precancerous lesions – and falling mortality.
Undesirable signatures showed a rising incidence juxtaposed with stable mortality and stable or rising metastatic incidence, signaling likely overdiagnosis, Dr. Welch and colleagues proposed. Three cancers—thyroid cancer, kidney cancer, and melanoma—fell into this category; for thyroid cancer and melanoma, fairly recent upticks in metastatic incidence may reflect upstaging.
Finally, some signatures showed mixed signals, with rising incidence and falling mortality. Breast cancer incidence rose and stabilized, coinciding with introduction of screening mammography, and possibly reflecting an increase in true cancer occurrence or overdiagnosis (with stable metastatic incidence favoring the latter), the analysts speculate. Declining mortality since the 1990s may be due to improved treatment or screening, or both. Prostate cancer incidence rose sharply with introduction of prostate-specific antigen screening but then fell to initial levels, suggesting sensitivity of this cancer to diagnostic scrutiny. Falling metastatic incidence indicates screening leads to earlier diagnosis in some cases, while declining mortality starting in the 1990s may again reflect improved treatment or screening, or both.
All cancers
The epidemiologic signature for all cancers combined differed somewhat by sex. Women had a rising incidence during the 1980s that was mainly driven by lung and breast cancers, according to Dr. Welch and colleagues; a continued rise since the mid-1990s was largely driven by melanoma, kidney cancer, and thyroid cancer. Declining mortality since 1990 has been primarily due to reductions in deaths from breast and colorectal cancers, and, more recently, lung cancer.
Men had a “volatile pattern” in the incidence of all cancers combined that was attributable to prostate cancer trends; drops in lung and colorectal cancer incidences were offset by rises in melanoma and kidney cancer incidences, the analysts proposed. Declining mortality since 1990 was more marked than that among women and reflects a longer period of decline in lung cancer mortality, plus reductions in deaths from prostate cancer and colorectal cancer.
“Falling mortality means that there has been real progress against cancer in the past 40 years – largely reflecting improved treatment and the decline of a uniquely powerful causal factor: cigarette smoking,” Dr. Welch and colleagues noted. “The lack of an accompanying fall in incidence is an unfortunate side effect of early cancer-detection efforts.”
Dr. Welch reported that he had no relevant disclosures. The analysis did not receive any specific funding.
SOURCE: Welch HG et al. N Engl J Med. 2019;381:1378-86. doi: 10.1056/NEJMsr1905447.
A new analysis of 40 years of U.S. cancer data underscores the importance of looking at multiple metrics to discern the complex interplay of factors influencing cancer burden in the population. Findings showed that the epidemiologic signature – a composite of two or three key metrics – differed across cancer types and was favorable in some cases and unfavorable in others.
“Epidemiologic signatures that illustrate trends in population-based data on cancer burden provide insight into true cancer occurrence, overdiagnosis, and treatment advances,” explain the analysts, led by H. Gilbert Welch, MD, MPH, Center for Surgery and Public Health, Brigham and Women’s Hospital, Boston. “They are important indicators of the potential contribution of environmental exposures, primary preventive interventions, new treatments, and changing diagnostic and screening practices.”
Dr. Welch and colleagues analyzed data for the years 1975 through 2015, assessing juxtaposed trends in incidence, mortality, and, when available, metastatic incidence (cancer already metastatic at diagnosis) for 11 cancers individually and for all cancers combined. Incidence data combining invasive and in situ cancers were obtained from the original nine Surveillance, Epidemiology, and End Results (SEER) registries, and mortality data were obtained from the National Vital Statistics System.
The analysts then explored implications of the epidemiologic signatures as they pertain to true cancer occurrence (the underlying incidence of clinically meaningful cancer), overdiagnosis (detection of cancers that will not cause symptoms or death), and treatment advances.
Individual cancers
Findings of the analysis, published in a special report in the New England Journal of Medicine, revealed three broad categories of epidemiologic signatures having different implications for the public health and oncology fields.
Desirable signatures showed, for example, declining mortality against a backdrop of stable incidence over the 40-year period, signaling improved treatment, as seen for chronic myeloid leukemia following introduction of imatinib (Gleevec), according to the analysts. Lung cancer incidence and mortality rose and fell in tandem, reflecting an increase in smoking followed by a decrease in response to prevention efforts. Stomach, cervical, and colorectal cancers had both falling incidence – likely reflecting a true decline in occurrence related to prevention and/or screening detection and subsequent treatment of precancerous lesions – and falling mortality.
Undesirable signatures showed a rising incidence juxtaposed with stable mortality and stable or rising metastatic incidence, signaling likely overdiagnosis, Dr. Welch and colleagues proposed. Three cancers—thyroid cancer, kidney cancer, and melanoma—fell into this category; for thyroid cancer and melanoma, fairly recent upticks in metastatic incidence may reflect upstaging.
Finally, some signatures showed mixed signals, with rising incidence and falling mortality. Breast cancer incidence rose and stabilized, coinciding with introduction of screening mammography, and possibly reflecting an increase in true cancer occurrence or overdiagnosis (with stable metastatic incidence favoring the latter), the analysts speculate. Declining mortality since the 1990s may be due to improved treatment or screening, or both. Prostate cancer incidence rose sharply with introduction of prostate-specific antigen screening but then fell to initial levels, suggesting sensitivity of this cancer to diagnostic scrutiny. Falling metastatic incidence indicates screening leads to earlier diagnosis in some cases, while declining mortality starting in the 1990s may again reflect improved treatment or screening, or both.
All cancers
The epidemiologic signature for all cancers combined differed somewhat by sex. Women had a rising incidence during the 1980s that was mainly driven by lung and breast cancers, according to Dr. Welch and colleagues; a continued rise since the mid-1990s was largely driven by melanoma, kidney cancer, and thyroid cancer. Declining mortality since 1990 has been primarily due to reductions in deaths from breast and colorectal cancers, and, more recently, lung cancer.
Men had a “volatile pattern” in the incidence of all cancers combined that was attributable to prostate cancer trends; drops in lung and colorectal cancer incidences were offset by rises in melanoma and kidney cancer incidences, the analysts proposed. Declining mortality since 1990 was more marked than that among women and reflects a longer period of decline in lung cancer mortality, plus reductions in deaths from prostate cancer and colorectal cancer.
“Falling mortality means that there has been real progress against cancer in the past 40 years – largely reflecting improved treatment and the decline of a uniquely powerful causal factor: cigarette smoking,” Dr. Welch and colleagues noted. “The lack of an accompanying fall in incidence is an unfortunate side effect of early cancer-detection efforts.”
Dr. Welch reported that he had no relevant disclosures. The analysis did not receive any specific funding.
SOURCE: Welch HG et al. N Engl J Med. 2019;381:1378-86. doi: 10.1056/NEJMsr1905447.
A new analysis of 40 years of U.S. cancer data underscores the importance of looking at multiple metrics to discern the complex interplay of factors influencing cancer burden in the population. Findings showed that the epidemiologic signature – a composite of two or three key metrics – differed across cancer types and was favorable in some cases and unfavorable in others.
“Epidemiologic signatures that illustrate trends in population-based data on cancer burden provide insight into true cancer occurrence, overdiagnosis, and treatment advances,” explain the analysts, led by H. Gilbert Welch, MD, MPH, Center for Surgery and Public Health, Brigham and Women’s Hospital, Boston. “They are important indicators of the potential contribution of environmental exposures, primary preventive interventions, new treatments, and changing diagnostic and screening practices.”
Dr. Welch and colleagues analyzed data for the years 1975 through 2015, assessing juxtaposed trends in incidence, mortality, and, when available, metastatic incidence (cancer already metastatic at diagnosis) for 11 cancers individually and for all cancers combined. Incidence data combining invasive and in situ cancers were obtained from the original nine Surveillance, Epidemiology, and End Results (SEER) registries, and mortality data were obtained from the National Vital Statistics System.
The analysts then explored implications of the epidemiologic signatures as they pertain to true cancer occurrence (the underlying incidence of clinically meaningful cancer), overdiagnosis (detection of cancers that will not cause symptoms or death), and treatment advances.
Individual cancers
Findings of the analysis, published in a special report in the New England Journal of Medicine, revealed three broad categories of epidemiologic signatures having different implications for the public health and oncology fields.
Desirable signatures showed, for example, declining mortality against a backdrop of stable incidence over the 40-year period, signaling improved treatment, as seen for chronic myeloid leukemia following introduction of imatinib (Gleevec), according to the analysts. Lung cancer incidence and mortality rose and fell in tandem, reflecting an increase in smoking followed by a decrease in response to prevention efforts. Stomach, cervical, and colorectal cancers had both falling incidence – likely reflecting a true decline in occurrence related to prevention and/or screening detection and subsequent treatment of precancerous lesions – and falling mortality.
Undesirable signatures showed a rising incidence juxtaposed with stable mortality and stable or rising metastatic incidence, signaling likely overdiagnosis, Dr. Welch and colleagues proposed. Three cancers—thyroid cancer, kidney cancer, and melanoma—fell into this category; for thyroid cancer and melanoma, fairly recent upticks in metastatic incidence may reflect upstaging.
Finally, some signatures showed mixed signals, with rising incidence and falling mortality. Breast cancer incidence rose and stabilized, coinciding with introduction of screening mammography, and possibly reflecting an increase in true cancer occurrence or overdiagnosis (with stable metastatic incidence favoring the latter), the analysts speculate. Declining mortality since the 1990s may be due to improved treatment or screening, or both. Prostate cancer incidence rose sharply with introduction of prostate-specific antigen screening but then fell to initial levels, suggesting sensitivity of this cancer to diagnostic scrutiny. Falling metastatic incidence indicates screening leads to earlier diagnosis in some cases, while declining mortality starting in the 1990s may again reflect improved treatment or screening, or both.
All cancers
The epidemiologic signature for all cancers combined differed somewhat by sex. Women had a rising incidence during the 1980s that was mainly driven by lung and breast cancers, according to Dr. Welch and colleagues; a continued rise since the mid-1990s was largely driven by melanoma, kidney cancer, and thyroid cancer. Declining mortality since 1990 has been primarily due to reductions in deaths from breast and colorectal cancers, and, more recently, lung cancer.
Men had a “volatile pattern” in the incidence of all cancers combined that was attributable to prostate cancer trends; drops in lung and colorectal cancer incidences were offset by rises in melanoma and kidney cancer incidences, the analysts proposed. Declining mortality since 1990 was more marked than that among women and reflects a longer period of decline in lung cancer mortality, plus reductions in deaths from prostate cancer and colorectal cancer.
“Falling mortality means that there has been real progress against cancer in the past 40 years – largely reflecting improved treatment and the decline of a uniquely powerful causal factor: cigarette smoking,” Dr. Welch and colleagues noted. “The lack of an accompanying fall in incidence is an unfortunate side effect of early cancer-detection efforts.”
Dr. Welch reported that he had no relevant disclosures. The analysis did not receive any specific funding.
SOURCE: Welch HG et al. N Engl J Med. 2019;381:1378-86. doi: 10.1056/NEJMsr1905447.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Twitter Chat: Skin Cancer

Join us on Tuesday, October 8, 2019, at 8:00 pm EST on Twitter at #MDedgeChats as we discuss skin cancer, and what’s new in sunscreen, skin of color, and melanoma.
Special guests include physicians with expertise in dermatology and skin cancer, Anthony Rossi, MD (@DrAnthonyRossi), Julie Amthor Croley, MD, 15k followers on IG (@Drskinandsmiles), and Candrice Heath, MD (@DrCandriceHeath). Background information about the chat can be found below.
What will the conversation cover?
Q1: What are the most common types of skin cancer?
Q2: What recent research findings can better inform patients about skin cancer risks?
Q3: What’s the difference between melanoma in fair skin vs. darker skin?
Q4: How does the risk of skin cancer differ in people with darker skin?
Q5: Why should sunscreen be used even in the fall and winter?

Follow us here: @MDedgeDerm | @MDedgeTweets | #MDedgeChats
About Dr. Rossi:
Dr. Anthony Rossi (@DrAnthonyRossi) is a board-certified dermatologist with fellowship training in Mohs micrographic surgery, cosmetic and laser surgery, and advanced cutaneous oncology at the Memorial Sloan Kettering Cancer Center and Weill Cornell Medical College program, both in New York. He specializes in skin cancer surgery, cosmetic dermatologic surgery, and laser surgery.
His research includes quality of life in cancer survivors, the use of noninvasive imaging of the skin, and nonsurgical treatments of skin cancer. Additionally, Dr. Rossi is active in dermatologic organizations and advocacy for medicine.
Research and Publications by Dr. Rossi
About Dr. Heath:
Dr. Candrice Heath (@DrCandriceHeath) is Assistant Professor of Dermatology at the Lewis Katz School of Medicine at Temple University in Philadelphia, Pennsylvania with fellowship training in pediatric dermatology at Johns Hopkins University in Baltimore, Maryland. Dr. Heath is triple board certified in pediatrics, dermatology, and pediatric dermatology. She specializes in adult and pediatric dermatology, skin of color, acne, and eczema. Dr. Heath also enjoys educating primary care physicians on the front lines of health care and delivering easy to understand information to consumers.
Research and publications by Dr. Heath
Guest host of MDedge podcast: A sunscreen update with Dr. Vincent DeLeo.
About Dr. Croley:
Dr. Julie Amthor Croley (@Drskinandsmiles) also known as “Dr. Skin and Smiles” has 15,000 followers on Instagram, and is a Chief Dermatology Resident at the University of Texas Medical Branch in Galveston, Texas. She has a special interest in skin cancer and dermatological surgery and hopes to complete a fellowship in Mohs micrographic surgery after residency. In her free time, Dr. Croley enjoys spending time with her husband (an orthopedic surgeon), running and competing in marathons, and spending time on the beach.
Cutaneous melanoma is the most fatal form of skin cancer and is a considerable public health concern in the United States. Early detection and management of skin cancer can lead to decreased morbidity and mortality from skin cancer. As a result, the American Academy of Dermatology Association supports safe sun-protective practices and diligent self-screening for changing lesions.
Sunscreen use is an essential component of sun protection. New regulations from the US Food and Drug Administration (FDA) have left consumers concerned about the safety of sunscreens. According to a recent Cutis editorial from Vincent A. DeLeo, MD, “There is no question that, as physicians, we want to ‘first, do no harm,’ so we should all be interested in assuring our patients that our sunscreen recommendations are safe and we support the FDA proposal for additional data.”
Patients with skin of color experience disproportionately higher morbidity and mortality when diagnosed with melanoma. “Poor prognosis in patients with skin of color is multifactorial and may be due to poor use of sun protection, misconceptions about melanoma risk, atypical clinical presentation, impaired access to care, and delay in diagnosis,” according to a recent Cutis article.
Population-based skin cancer screening performed exclusively by dermatologists is not practical. Primary care physicians and other experts in melanoma and public health need to be involved in reducing melanoma mortality.
In this chat, we will provide expert recommendations on the diagnosis of skin cancer, preventive measures, and the latest research discussed among physicians.
- “Doctor, Do I Need a Skin Check?”
- Assessing the effectiveness of knowledge-based interventions in skin of color populations.
- Melanoma in US Hispanics
- Podcast: Sunscreen update from Dr. Vincent DeLeo
- Windshield and UV exposure
- Racial, ethnic minorities often don’t practice sun-protective behaviors.
- Sunscreen regulations and advice for your patients.

Join us on Tuesday, October 8, 2019, at 8:00 pm EST on Twitter at #MDedgeChats as we discuss skin cancer, and what’s new in sunscreen, skin of color, and melanoma.
Special guests include physicians with expertise in dermatology and skin cancer, Anthony Rossi, MD (@DrAnthonyRossi), Julie Amthor Croley, MD, 15k followers on IG (@Drskinandsmiles), and Candrice Heath, MD (@DrCandriceHeath). Background information about the chat can be found below.
What will the conversation cover?
Q1: What are the most common types of skin cancer?
Q2: What recent research findings can better inform patients about skin cancer risks?
Q3: What’s the difference between melanoma in fair skin vs. darker skin?
Q4: How does the risk of skin cancer differ in people with darker skin?
Q5: Why should sunscreen be used even in the fall and winter?

Follow us here: @MDedgeDerm | @MDedgeTweets | #MDedgeChats
About Dr. Rossi:
Dr. Anthony Rossi (@DrAnthonyRossi) is a board-certified dermatologist with fellowship training in Mohs micrographic surgery, cosmetic and laser surgery, and advanced cutaneous oncology at the Memorial Sloan Kettering Cancer Center and Weill Cornell Medical College program, both in New York. He specializes in skin cancer surgery, cosmetic dermatologic surgery, and laser surgery.
His research includes quality of life in cancer survivors, the use of noninvasive imaging of the skin, and nonsurgical treatments of skin cancer. Additionally, Dr. Rossi is active in dermatologic organizations and advocacy for medicine.
Research and Publications by Dr. Rossi
About Dr. Heath:
Dr. Candrice Heath (@DrCandriceHeath) is Assistant Professor of Dermatology at the Lewis Katz School of Medicine at Temple University in Philadelphia, Pennsylvania with fellowship training in pediatric dermatology at Johns Hopkins University in Baltimore, Maryland. Dr. Heath is triple board certified in pediatrics, dermatology, and pediatric dermatology. She specializes in adult and pediatric dermatology, skin of color, acne, and eczema. Dr. Heath also enjoys educating primary care physicians on the front lines of health care and delivering easy to understand information to consumers.
Research and publications by Dr. Heath
Guest host of MDedge podcast: A sunscreen update with Dr. Vincent DeLeo.
About Dr. Croley:
Dr. Julie Amthor Croley (@Drskinandsmiles) also known as “Dr. Skin and Smiles” has 15,000 followers on Instagram, and is a Chief Dermatology Resident at the University of Texas Medical Branch in Galveston, Texas. She has a special interest in skin cancer and dermatological surgery and hopes to complete a fellowship in Mohs micrographic surgery after residency. In her free time, Dr. Croley enjoys spending time with her husband (an orthopedic surgeon), running and competing in marathons, and spending time on the beach.
Cutaneous melanoma is the most fatal form of skin cancer and is a considerable public health concern in the United States. Early detection and management of skin cancer can lead to decreased morbidity and mortality from skin cancer. As a result, the American Academy of Dermatology Association supports safe sun-protective practices and diligent self-screening for changing lesions.
Sunscreen use is an essential component of sun protection. New regulations from the US Food and Drug Administration (FDA) have left consumers concerned about the safety of sunscreens. According to a recent Cutis editorial from Vincent A. DeLeo, MD, “There is no question that, as physicians, we want to ‘first, do no harm,’ so we should all be interested in assuring our patients that our sunscreen recommendations are safe and we support the FDA proposal for additional data.”
Patients with skin of color experience disproportionately higher morbidity and mortality when diagnosed with melanoma. “Poor prognosis in patients with skin of color is multifactorial and may be due to poor use of sun protection, misconceptions about melanoma risk, atypical clinical presentation, impaired access to care, and delay in diagnosis,” according to a recent Cutis article.
Population-based skin cancer screening performed exclusively by dermatologists is not practical. Primary care physicians and other experts in melanoma and public health need to be involved in reducing melanoma mortality.
In this chat, we will provide expert recommendations on the diagnosis of skin cancer, preventive measures, and the latest research discussed among physicians.
- “Doctor, Do I Need a Skin Check?”
- Assessing the effectiveness of knowledge-based interventions in skin of color populations.
- Melanoma in US Hispanics
- Podcast: Sunscreen update from Dr. Vincent DeLeo
- Windshield and UV exposure
- Racial, ethnic minorities often don’t practice sun-protective behaviors.
- Sunscreen regulations and advice for your patients.

Join us on Tuesday, October 8, 2019, at 8:00 pm EST on Twitter at #MDedgeChats as we discuss skin cancer, and what’s new in sunscreen, skin of color, and melanoma.
Special guests include physicians with expertise in dermatology and skin cancer, Anthony Rossi, MD (@DrAnthonyRossi), Julie Amthor Croley, MD, 15k followers on IG (@Drskinandsmiles), and Candrice Heath, MD (@DrCandriceHeath). Background information about the chat can be found below.
What will the conversation cover?
Q1: What are the most common types of skin cancer?
Q2: What recent research findings can better inform patients about skin cancer risks?
Q3: What’s the difference between melanoma in fair skin vs. darker skin?
Q4: How does the risk of skin cancer differ in people with darker skin?
Q5: Why should sunscreen be used even in the fall and winter?

Follow us here: @MDedgeDerm | @MDedgeTweets | #MDedgeChats
About Dr. Rossi:
Dr. Anthony Rossi (@DrAnthonyRossi) is a board-certified dermatologist with fellowship training in Mohs micrographic surgery, cosmetic and laser surgery, and advanced cutaneous oncology at the Memorial Sloan Kettering Cancer Center and Weill Cornell Medical College program, both in New York. He specializes in skin cancer surgery, cosmetic dermatologic surgery, and laser surgery.
His research includes quality of life in cancer survivors, the use of noninvasive imaging of the skin, and nonsurgical treatments of skin cancer. Additionally, Dr. Rossi is active in dermatologic organizations and advocacy for medicine.
Research and Publications by Dr. Rossi
About Dr. Heath:
Dr. Candrice Heath (@DrCandriceHeath) is Assistant Professor of Dermatology at the Lewis Katz School of Medicine at Temple University in Philadelphia, Pennsylvania with fellowship training in pediatric dermatology at Johns Hopkins University in Baltimore, Maryland. Dr. Heath is triple board certified in pediatrics, dermatology, and pediatric dermatology. She specializes in adult and pediatric dermatology, skin of color, acne, and eczema. Dr. Heath also enjoys educating primary care physicians on the front lines of health care and delivering easy to understand information to consumers.
Research and publications by Dr. Heath
Guest host of MDedge podcast: A sunscreen update with Dr. Vincent DeLeo.
About Dr. Croley:
Dr. Julie Amthor Croley (@Drskinandsmiles) also known as “Dr. Skin and Smiles” has 15,000 followers on Instagram, and is a Chief Dermatology Resident at the University of Texas Medical Branch in Galveston, Texas. She has a special interest in skin cancer and dermatological surgery and hopes to complete a fellowship in Mohs micrographic surgery after residency. In her free time, Dr. Croley enjoys spending time with her husband (an orthopedic surgeon), running and competing in marathons, and spending time on the beach.
Cutaneous melanoma is the most fatal form of skin cancer and is a considerable public health concern in the United States. Early detection and management of skin cancer can lead to decreased morbidity and mortality from skin cancer. As a result, the American Academy of Dermatology Association supports safe sun-protective practices and diligent self-screening for changing lesions.
Sunscreen use is an essential component of sun protection. New regulations from the US Food and Drug Administration (FDA) have left consumers concerned about the safety of sunscreens. According to a recent Cutis editorial from Vincent A. DeLeo, MD, “There is no question that, as physicians, we want to ‘first, do no harm,’ so we should all be interested in assuring our patients that our sunscreen recommendations are safe and we support the FDA proposal for additional data.”
Patients with skin of color experience disproportionately higher morbidity and mortality when diagnosed with melanoma. “Poor prognosis in patients with skin of color is multifactorial and may be due to poor use of sun protection, misconceptions about melanoma risk, atypical clinical presentation, impaired access to care, and delay in diagnosis,” according to a recent Cutis article.
Population-based skin cancer screening performed exclusively by dermatologists is not practical. Primary care physicians and other experts in melanoma and public health need to be involved in reducing melanoma mortality.
In this chat, we will provide expert recommendations on the diagnosis of skin cancer, preventive measures, and the latest research discussed among physicians.
- “Doctor, Do I Need a Skin Check?”
- Assessing the effectiveness of knowledge-based interventions in skin of color populations.
- Melanoma in US Hispanics
- Podcast: Sunscreen update from Dr. Vincent DeLeo
- Windshield and UV exposure
- Racial, ethnic minorities often don’t practice sun-protective behaviors.
- Sunscreen regulations and advice for your patients.
Systemic sclerosis raises risk of breast cancer, lung cancer, melanoma
in a population-linked cohort study published in Arthritis Care & Research.
Kathleen Morrisroe, MBBS, PhD, of St. Vincent’s Hospital Melbourne and colleagues matched deidentified patient data in the Australian Scleroderma Cohort Study (ASCS) with patients’ respective state cancer registry data between January 2008 and December 2015. The researchers also used the Australian Medical Benefit Schedule (MBS) to track health care costs for hospital admissions, presentations to the ED, other health visits, pathology, and imaging, as well as other associated costs for care, in each state. Based on this information, Dr. Morrisroe and colleagues calculated standardized incidence ratios (SIR) and standardized mortality ratios (SMR) for these patients by comparing them with the general population in Australia.
The results included 1,727 patients with systemic sclerosis (SSc) and cancer in the cohort, which consisted of mostly white (92.1%) women (85.9%) who had limited cutaneous SSc (73.9%). They were a mean of 46.6 years old when they were diagnosed with SSc and had a mean disease duration of 10.9 years. The incidence of cancer was 1.3% per year, and the overall prevalence for the cohort was 14.2%, which was higher than the general Australian population (SIR, 2.15; 95% confidence interval, 1.84-2.49). Breast cancer, melanoma, hematologic cancer, and lung cancer were the most common types of cancers found in the cohort, with early breast cancer (SIR, 3.07; 95% CI, 1.47-5.64), lung cancer (SIR, 3.07; 95% CI, 1.21-3.44), and early melanoma (SIR, 3.40; 95% CI, 1.10-7.93) having a higher incidence than the general population.
Patients with RNA polymerase III (RNAP) autoantibody had a higher incidence of early onset cancer (odds ratio, 2.9; P = .044), defined as a cancer diagnosis within 5 years of SSc diagnosis. Interstitial lung disease was also linked to an increased risk of lung cancer (OR, 2.83; P = .031), which persisted after the researchers performed a multivariate analysis.
Another factor that increased the overall risk of cancer was calcium channel blockers (OR, 1.47; P = .016), which also increased the risk of breast (OR, 1.61; P = .051) and melanoma-specific cancers (OR, 2.01; P = .042), a finding the researchers said was “unexpected, but has been reported in the literature with conflicting results.”
“This association is hypothesized to be related to the role of calcium in cell apoptosis, such as activation of the caspase pathway, induction of endonuclease activity and mitochondrial permeation,” Dr. Morrisroe and colleagues wrote.
SSc patients had more than a doubling of risk of mortality with incident cancer in comparison with SSc patients who did not have cancer (hazard ratio, 2.85; 95% CI, 1.51-5.37; P = .001). The average cost of health care annually for an SSc patient with cancer was AUD $1,496 (P less than .001), the researchers said.
This study was funded in part by Scleroderma Australia, Arthritis Australia, Actelion Australia, Bayer, CSL Biotherapies, GlaxoSmithKline Australia, and Pfizer. Dr. Morrisroe reported receiving support from Arthritis Australia and Royal Australasian College of Physicians Research Establishment Fellowships. Another author reported receiving a fellowship from the National Health and Medical Research Council of Australia. The other authors reported no relevant conflicts of interest.
SOURCE: Morrisroe K et al. Arthritis Care Res. 2019 Sep 20. doi: 10.1002/acr.24076
in a population-linked cohort study published in Arthritis Care & Research.
Kathleen Morrisroe, MBBS, PhD, of St. Vincent’s Hospital Melbourne and colleagues matched deidentified patient data in the Australian Scleroderma Cohort Study (ASCS) with patients’ respective state cancer registry data between January 2008 and December 2015. The researchers also used the Australian Medical Benefit Schedule (MBS) to track health care costs for hospital admissions, presentations to the ED, other health visits, pathology, and imaging, as well as other associated costs for care, in each state. Based on this information, Dr. Morrisroe and colleagues calculated standardized incidence ratios (SIR) and standardized mortality ratios (SMR) for these patients by comparing them with the general population in Australia.
The results included 1,727 patients with systemic sclerosis (SSc) and cancer in the cohort, which consisted of mostly white (92.1%) women (85.9%) who had limited cutaneous SSc (73.9%). They were a mean of 46.6 years old when they were diagnosed with SSc and had a mean disease duration of 10.9 years. The incidence of cancer was 1.3% per year, and the overall prevalence for the cohort was 14.2%, which was higher than the general Australian population (SIR, 2.15; 95% confidence interval, 1.84-2.49). Breast cancer, melanoma, hematologic cancer, and lung cancer were the most common types of cancers found in the cohort, with early breast cancer (SIR, 3.07; 95% CI, 1.47-5.64), lung cancer (SIR, 3.07; 95% CI, 1.21-3.44), and early melanoma (SIR, 3.40; 95% CI, 1.10-7.93) having a higher incidence than the general population.
Patients with RNA polymerase III (RNAP) autoantibody had a higher incidence of early onset cancer (odds ratio, 2.9; P = .044), defined as a cancer diagnosis within 5 years of SSc diagnosis. Interstitial lung disease was also linked to an increased risk of lung cancer (OR, 2.83; P = .031), which persisted after the researchers performed a multivariate analysis.
Another factor that increased the overall risk of cancer was calcium channel blockers (OR, 1.47; P = .016), which also increased the risk of breast (OR, 1.61; P = .051) and melanoma-specific cancers (OR, 2.01; P = .042), a finding the researchers said was “unexpected, but has been reported in the literature with conflicting results.”
“This association is hypothesized to be related to the role of calcium in cell apoptosis, such as activation of the caspase pathway, induction of endonuclease activity and mitochondrial permeation,” Dr. Morrisroe and colleagues wrote.
SSc patients had more than a doubling of risk of mortality with incident cancer in comparison with SSc patients who did not have cancer (hazard ratio, 2.85; 95% CI, 1.51-5.37; P = .001). The average cost of health care annually for an SSc patient with cancer was AUD $1,496 (P less than .001), the researchers said.
This study was funded in part by Scleroderma Australia, Arthritis Australia, Actelion Australia, Bayer, CSL Biotherapies, GlaxoSmithKline Australia, and Pfizer. Dr. Morrisroe reported receiving support from Arthritis Australia and Royal Australasian College of Physicians Research Establishment Fellowships. Another author reported receiving a fellowship from the National Health and Medical Research Council of Australia. The other authors reported no relevant conflicts of interest.
SOURCE: Morrisroe K et al. Arthritis Care Res. 2019 Sep 20. doi: 10.1002/acr.24076
in a population-linked cohort study published in Arthritis Care & Research.
Kathleen Morrisroe, MBBS, PhD, of St. Vincent’s Hospital Melbourne and colleagues matched deidentified patient data in the Australian Scleroderma Cohort Study (ASCS) with patients’ respective state cancer registry data between January 2008 and December 2015. The researchers also used the Australian Medical Benefit Schedule (MBS) to track health care costs for hospital admissions, presentations to the ED, other health visits, pathology, and imaging, as well as other associated costs for care, in each state. Based on this information, Dr. Morrisroe and colleagues calculated standardized incidence ratios (SIR) and standardized mortality ratios (SMR) for these patients by comparing them with the general population in Australia.
The results included 1,727 patients with systemic sclerosis (SSc) and cancer in the cohort, which consisted of mostly white (92.1%) women (85.9%) who had limited cutaneous SSc (73.9%). They were a mean of 46.6 years old when they were diagnosed with SSc and had a mean disease duration of 10.9 years. The incidence of cancer was 1.3% per year, and the overall prevalence for the cohort was 14.2%, which was higher than the general Australian population (SIR, 2.15; 95% confidence interval, 1.84-2.49). Breast cancer, melanoma, hematologic cancer, and lung cancer were the most common types of cancers found in the cohort, with early breast cancer (SIR, 3.07; 95% CI, 1.47-5.64), lung cancer (SIR, 3.07; 95% CI, 1.21-3.44), and early melanoma (SIR, 3.40; 95% CI, 1.10-7.93) having a higher incidence than the general population.
Patients with RNA polymerase III (RNAP) autoantibody had a higher incidence of early onset cancer (odds ratio, 2.9; P = .044), defined as a cancer diagnosis within 5 years of SSc diagnosis. Interstitial lung disease was also linked to an increased risk of lung cancer (OR, 2.83; P = .031), which persisted after the researchers performed a multivariate analysis.
Another factor that increased the overall risk of cancer was calcium channel blockers (OR, 1.47; P = .016), which also increased the risk of breast (OR, 1.61; P = .051) and melanoma-specific cancers (OR, 2.01; P = .042), a finding the researchers said was “unexpected, but has been reported in the literature with conflicting results.”
“This association is hypothesized to be related to the role of calcium in cell apoptosis, such as activation of the caspase pathway, induction of endonuclease activity and mitochondrial permeation,” Dr. Morrisroe and colleagues wrote.
SSc patients had more than a doubling of risk of mortality with incident cancer in comparison with SSc patients who did not have cancer (hazard ratio, 2.85; 95% CI, 1.51-5.37; P = .001). The average cost of health care annually for an SSc patient with cancer was AUD $1,496 (P less than .001), the researchers said.
This study was funded in part by Scleroderma Australia, Arthritis Australia, Actelion Australia, Bayer, CSL Biotherapies, GlaxoSmithKline Australia, and Pfizer. Dr. Morrisroe reported receiving support from Arthritis Australia and Royal Australasian College of Physicians Research Establishment Fellowships. Another author reported receiving a fellowship from the National Health and Medical Research Council of Australia. The other authors reported no relevant conflicts of interest.
SOURCE: Morrisroe K et al. Arthritis Care Res. 2019 Sep 20. doi: 10.1002/acr.24076
FROM ARTHRITIS CARE & RESEARCH
UV Radiation Exposure in Welders: Impact on the Skin and Eyes
Although solar radiation is the most commonly recognized source of UV radiation (UVR), occupational exposures can contribute due to the intensity and chronicity of exposure. Arc welding is a process whereby metal is fused together by heat produced from an electric arc. The electric arc that forms between the electrode and the base metal emits radiation in the full UV spectrum including UVA (400–315 nm), UVB (315–290 nm), and UVC (290–100 nm) wavelengths. Welders, therefore, have an increased risk for broad-spectrum, intense exposure to UVR, which may play a notable role in UV-related skin disease without proper protection. We report 3 welders with skin disease attributed to occupational exposure to UVR.
Case Reports
Patient 1
A 41-year-old man presented for evaluation of treatment-resistant cutaneous lupus. During the 10-year disease course, the patient was treated by both dermatologists and rheumatologists with frequent exacerbations and poor disease control. At the time of presentation, treatment with hydroxychloroquine 200 mg twice daily, azathioprine 50 mg twice daily, intramuscular methylprednisolone acetateinjectable suspension 40 mg, and prednisone 20 mg daily was failing. Physical examination revealed polycyclic erythematous plaques typical of subacute cutaneous lupus erythematosus. A skin biopsy confirmed the diagnosis. Upon further discussion of exacerbating risk factors, the patient noted UVR exposure while working as a welder. Although he had been previously told to avoid sunlight, he did not realize that this recommendation included all forms of UV light. Once this work exposure was eliminated, he was restarted on hydroxychloroquine 200 mg twice daily and topical steroids, and he responded with complete and sustained clearance of disease. When he returned to welding, utilization of sunscreen and sun-protective clothing enabled him to maintain control of his subacute cutaneous lupus erythematosus on oral hydroxychloroquine 200 mg twice daily and topical steroids.
Patient 2
A 55-year-old man presented with numerous actinic keratoses and persistent erythema in a well-demarcated area involving the forehead, temples, and lateral cheeks but sparing the periorbital area. The patient also experienced UVR exposure from welding (up to 4 to 5 times per week during his career spanning more than 20 years). He cited frequent burns in areas where his protective equipment did not cover his skin. He also reported that he often forgoes wearing protective equipment, even though it is available, and only uses safety goggles due to the extreme heat of the working environment as well as the awkwardness of wearing full protective gear while performing certain aspects of the job.
Patient 3
A 63-year-old man presented with a growth on the left side of the upper forehead. A biopsy revealed a squamous cell carcinoma, keratoacanthoma type. He worked as a welder for 40 years until retiring 1 year prior to presentation. He welded daily and always wore a tall face shield. Although the face shield covered most of his face, the scalp and some parts of the upper face were not well protected. In addition to the keratoacanthoma, which presented just outside of the area protected by the face shield, the patient had numerous actinic keratoses on the scalp.
Comment
Welding and UVR Exposure
Arc welders endure large amounts of UVR exposure, which is substantial enough to have notable health effects. The duration of exposure, electrical current used, angle of exposure, amount of ventilation, and the distance from the welding arc play a role in overall UVR exposure.1,2 Maximum permissible exposure (MPE) limits to UVR have been set by the International Commission on Non-Ionizing Radiation Protection and the National Institute for Occupational Safety and Health.3,4 The quantity of radiation produced by the arc allows for an exposure time of only a few seconds to minutes before surpassing MPE to UV light.1,5 Welders are exposed to total-body UVR doses up to 3000 times the MPE, and mean cumulative exposure calculated over an 8-hour workday can reach 9795 mJ/cm2.6
Workers in close proximity to welders also receive large UVR doses and may not be aware of its hazardous effects. Nearby nonwelders can be exposed to 13 times the MPE of UVR.6 At distances up to 10 m from the arc, the irradiance is large enough to reach MPE to UVR in less than 3 hours.1
Skin and Eye Damage From Welding
Exposure to UVR produced by the welding arc may lead to acute skin or eye reactions, chronic skin or eye disorders, or exacerbation of photosensitive diseases. Common acute problems are photokeratoconjunctivitis (welder’s flash) and skin erythema.7,8
Actinic elastosis, actinic keratoses, ocular melanoma, and photosensitive diseases represent a spectrum of disorders that can present from chronic UV exposure in welders. In a study by Emmett et al7 of 152 welders and 58 controls, actinic elastosis was found to be more frequent in welders than controls. Cases of basal cell carcinoma and squamous cell carcinoma also have been reported in welders.9,10 However, in the study by Emmett et al,7 a statistically significant correlation between welding and skin cancer was not documented. There were limitations in the study, such as small sample size and a young average age of welders.7 Future studies may be needed to further clarify the risk for skin cancer in welders.
Although there is no clear association with skin cancer, an increased risk of ocular melanoma in welders is more clearly established. A meta-analysis of 5 studies found that welding was a significant risk factor for ocular melanoma, with an odds ratio of 2.05 (95% confidence interval, 1.20-3.51).11 Other reported eye damage from chronic UVR exposure includes cataracts, chronic conjunctivitis, and retinal damage.12,13
Case reports of the following photosensitive diseases have been reported to be exacerbated or caused by UV light exposure in welders: discoid lupus erythematosus14; photodermatitis15; broadband photosensitivity with decreased minimal erythema dose to UVA, UVB, and UVC16; UVC-exacerbated atopic dermatitis17; polymorphous light eruption–like skin eruption18; and UVA-induced photoallergy to hydrochlorothiazide and ramipril.19
Prevention of Occupational Exposure to UVR
Occupational Safety and Health Administration guidelines protect workers from excessive exposure to UVR with personal protective equipment (PPE). In addition to UVR protection, PPE needs to protect welders from other risks including trauma from welding debris (slag), fires, electrical burns, and fumes. Online resources from the National Ag Safety Database,20 the American Welding Society,21 and Occupational Safety and Health Administration22,23 are available. These resources advise welders to work in ventilated areas with respirators specific for the metal being welded and to wear clothing and gloves that are not only fire retardant but also UV resistant.20-23 Additional PPE should protect the head, face, and eyes.
Unfortunately, even workers well trained in prevention guidelines may not adequately protect themselves. Some welders forego PPE due to heat, thus exposing themselves to UVR damage in areas that are normally covered. Welders also may forego equipment when working on jobs requiring more detailed welds where clothing, masks, and glasses may be overly bulky and inhibit the worker’s precision. Nontraditional welders, such as artisans or handymen, may not have workplace safety education to be aware of UVR emitted from welding and may not have readily available PPE.
The Figure portrays an amateur welder working without full PPE. Although he is wearing a face shield, he is not wearing fire-retardant clothing, lacks full protective garments, and has no ventilation system.

Conclusion
It is important to recognize welding as an occupation with notable exposure to UVR. Personal protective equipment should be the mainstay of prevention. Sunscreen is a useful adjunct but does not cover UVC that is emitted in the welding arc. Screens and welding blankets can be placed around welders to contain UVR and limit nonwelder exposure. Although UVR hazards should be regulated in the workplace as part of regular safety reviews, the clinician can play a role in recognizing this source of UVR in skin disease and in encouraging the use of PPE.
- Okuno T, Ojima J, Saito H. Ultraviolet radiation emitted by CO(2) arc welding. Ann Occup Hyg. 2001;45:597-601.
- Peng CY, Liu HH, Chang CP, et al. Evaluation and monitoring of UVR in shield metal ARC welding processing. Health Phys. 2007;93:101-108.
- The National Institute for Occupational Safety and Health. Criteria for a recommended standard: occupational exposure to ultraviolet radiation. DHHS (NIOSH) publication 73-11009. https://www.cdc.gov/niosh/docs/73-11009/. Updated June 6, 2014. Accessed September 6, 2019.
- International Commission on Non-Ionizing Radiation Protection. Guidelines on limits of exposure to ultraviolet radiation of wavelengths between 180 nm and 400 nm (incoherent optical radiation). Health Phys. 2004;87:171-186.
- Peng CY, Lan CH, Juang YJ, et al. Exposure assessment of aluminum arc welding radiation. Health Phys. 2007;93:298-306.
- Tenkate TD, Collins MJ. Personal ultraviolet radiation exposure of workers in a welding environment. Am Ind Hyg Assoc J. 1997;58:33-38.
- Emmett EA, Buncher CR, Suskind RB, et al. Skin and eye diseases among arc welders and those exposed to welding operations. J Occup Med. 1981;23:85-90.
- Bruze M, Hindsén M, Trulsson L. Dermatitis with an unusual explanation in a welder. Acta Derm Venereol. 1994;74:380-382.
- Donoghue AM, Sinclair MJ. Basal cell carcinoma after frequent episodes of cutaneous erythema and peeling induced by welding. Occup Environ Med. 1999;56:646.
- Currie CL, Monk BE. Welding and non-melanoma skin cancer. Clin Exp Dermatol. 2000;25:28-29.
- Shah CP, Weis E, Lajous M, et al. Intermittent and chronic ultraviolet light exposure and uveal melanoma: a meta-analysis. Ophthalmology. 2005;112:1599-1607.
- Yang X, Shao D, Ding X, et al. Chronic phototoxic maculopathy caused by welding arc in occupational welders. Can J Ophthalmol. 2012;47:45-50.
- Davies KG, Asanga U, Nku CO, et al. Effect of chronic exposure to welding light on Calabar welders. Niger J Physiol Sci. 2007;22:55-58.
- Wozniak KD. Erythematodes chronicus discoides as an occupational disease in an electric welder [in German]. Berufs-Dermatosen. 1971;19:187-196.
- Shehade SA, Roberts PJ, Diffey BL, et al. Photodermatitis due to spot welding. Br J Dermatol. 1987;117:117-119.
- Roelandts R, Huys I. Broad-band and persistent photosensitivity following accidental ultraviolet C overexposure. Photodermatol Photoimmunol Photomed. 1993;9:144-146.
- Elsner P, Hassam S. Occupational UVC-induced exacerbation of atopic dermatitis in a welder. Contact Dermatitis. 1996;35:180-181.
- Majoie IM, van Weelden H, Sybesma IM, et al. Polymorphous light eruption-like skin lesions in welders caused by ultraviolet C light. J Am Acad Dermatol. 2010;62:150-151.
- Wagner SN, Welke F, Goos M. Occupational UVA-induced allergic photodermatitis in a welder due to hydrochlorothiazide and ramipril. Contact Dermatitis. 2000;43:245-246.
- Fluegel L, Rein BK. Arc welding safety. National Ag Safety Database website. http://nasdonline.org/1083/d000873/arc-welding-safety.html. Published May 1989. Accessed September 6, 2019.
- American Welding Society. Personal protective equipment (PPE) for welding and cutting. Fact sheet no. 33-04/14. http://www.aws.org/technical/facts/FACT-33_2014.pdf. Published April 2014. Accessed September 6, 2019.
- Occupational Safety and Health Administration. Eye protection against radiant energy during welding and cutting in shipyard employment. https://www.osha.gov/Publications/OSHAfactsheet-eyeprotection-during-welding.pdf. Published January 2012. Accessed September 6, 2019.
- Occupational Safety and Health Administration. Welding, cutting, and brazing. https://www.osha.gov/SLTC/weldingcuttingbrazing/standards.html. Accessed September 10, 2019.
Although solar radiation is the most commonly recognized source of UV radiation (UVR), occupational exposures can contribute due to the intensity and chronicity of exposure. Arc welding is a process whereby metal is fused together by heat produced from an electric arc. The electric arc that forms between the electrode and the base metal emits radiation in the full UV spectrum including UVA (400–315 nm), UVB (315–290 nm), and UVC (290–100 nm) wavelengths. Welders, therefore, have an increased risk for broad-spectrum, intense exposure to UVR, which may play a notable role in UV-related skin disease without proper protection. We report 3 welders with skin disease attributed to occupational exposure to UVR.
Case Reports
Patient 1
A 41-year-old man presented for evaluation of treatment-resistant cutaneous lupus. During the 10-year disease course, the patient was treated by both dermatologists and rheumatologists with frequent exacerbations and poor disease control. At the time of presentation, treatment with hydroxychloroquine 200 mg twice daily, azathioprine 50 mg twice daily, intramuscular methylprednisolone acetateinjectable suspension 40 mg, and prednisone 20 mg daily was failing. Physical examination revealed polycyclic erythematous plaques typical of subacute cutaneous lupus erythematosus. A skin biopsy confirmed the diagnosis. Upon further discussion of exacerbating risk factors, the patient noted UVR exposure while working as a welder. Although he had been previously told to avoid sunlight, he did not realize that this recommendation included all forms of UV light. Once this work exposure was eliminated, he was restarted on hydroxychloroquine 200 mg twice daily and topical steroids, and he responded with complete and sustained clearance of disease. When he returned to welding, utilization of sunscreen and sun-protective clothing enabled him to maintain control of his subacute cutaneous lupus erythematosus on oral hydroxychloroquine 200 mg twice daily and topical steroids.
Patient 2
A 55-year-old man presented with numerous actinic keratoses and persistent erythema in a well-demarcated area involving the forehead, temples, and lateral cheeks but sparing the periorbital area. The patient also experienced UVR exposure from welding (up to 4 to 5 times per week during his career spanning more than 20 years). He cited frequent burns in areas where his protective equipment did not cover his skin. He also reported that he often forgoes wearing protective equipment, even though it is available, and only uses safety goggles due to the extreme heat of the working environment as well as the awkwardness of wearing full protective gear while performing certain aspects of the job.
Patient 3
A 63-year-old man presented with a growth on the left side of the upper forehead. A biopsy revealed a squamous cell carcinoma, keratoacanthoma type. He worked as a welder for 40 years until retiring 1 year prior to presentation. He welded daily and always wore a tall face shield. Although the face shield covered most of his face, the scalp and some parts of the upper face were not well protected. In addition to the keratoacanthoma, which presented just outside of the area protected by the face shield, the patient had numerous actinic keratoses on the scalp.
Comment
Welding and UVR Exposure
Arc welders endure large amounts of UVR exposure, which is substantial enough to have notable health effects. The duration of exposure, electrical current used, angle of exposure, amount of ventilation, and the distance from the welding arc play a role in overall UVR exposure.1,2 Maximum permissible exposure (MPE) limits to UVR have been set by the International Commission on Non-Ionizing Radiation Protection and the National Institute for Occupational Safety and Health.3,4 The quantity of radiation produced by the arc allows for an exposure time of only a few seconds to minutes before surpassing MPE to UV light.1,5 Welders are exposed to total-body UVR doses up to 3000 times the MPE, and mean cumulative exposure calculated over an 8-hour workday can reach 9795 mJ/cm2.6
Workers in close proximity to welders also receive large UVR doses and may not be aware of its hazardous effects. Nearby nonwelders can be exposed to 13 times the MPE of UVR.6 At distances up to 10 m from the arc, the irradiance is large enough to reach MPE to UVR in less than 3 hours.1
Skin and Eye Damage From Welding
Exposure to UVR produced by the welding arc may lead to acute skin or eye reactions, chronic skin or eye disorders, or exacerbation of photosensitive diseases. Common acute problems are photokeratoconjunctivitis (welder’s flash) and skin erythema.7,8
Actinic elastosis, actinic keratoses, ocular melanoma, and photosensitive diseases represent a spectrum of disorders that can present from chronic UV exposure in welders. In a study by Emmett et al7 of 152 welders and 58 controls, actinic elastosis was found to be more frequent in welders than controls. Cases of basal cell carcinoma and squamous cell carcinoma also have been reported in welders.9,10 However, in the study by Emmett et al,7 a statistically significant correlation between welding and skin cancer was not documented. There were limitations in the study, such as small sample size and a young average age of welders.7 Future studies may be needed to further clarify the risk for skin cancer in welders.
Although there is no clear association with skin cancer, an increased risk of ocular melanoma in welders is more clearly established. A meta-analysis of 5 studies found that welding was a significant risk factor for ocular melanoma, with an odds ratio of 2.05 (95% confidence interval, 1.20-3.51).11 Other reported eye damage from chronic UVR exposure includes cataracts, chronic conjunctivitis, and retinal damage.12,13
Case reports of the following photosensitive diseases have been reported to be exacerbated or caused by UV light exposure in welders: discoid lupus erythematosus14; photodermatitis15; broadband photosensitivity with decreased minimal erythema dose to UVA, UVB, and UVC16; UVC-exacerbated atopic dermatitis17; polymorphous light eruption–like skin eruption18; and UVA-induced photoallergy to hydrochlorothiazide and ramipril.19
Prevention of Occupational Exposure to UVR
Occupational Safety and Health Administration guidelines protect workers from excessive exposure to UVR with personal protective equipment (PPE). In addition to UVR protection, PPE needs to protect welders from other risks including trauma from welding debris (slag), fires, electrical burns, and fumes. Online resources from the National Ag Safety Database,20 the American Welding Society,21 and Occupational Safety and Health Administration22,23 are available. These resources advise welders to work in ventilated areas with respirators specific for the metal being welded and to wear clothing and gloves that are not only fire retardant but also UV resistant.20-23 Additional PPE should protect the head, face, and eyes.
Unfortunately, even workers well trained in prevention guidelines may not adequately protect themselves. Some welders forego PPE due to heat, thus exposing themselves to UVR damage in areas that are normally covered. Welders also may forego equipment when working on jobs requiring more detailed welds where clothing, masks, and glasses may be overly bulky and inhibit the worker’s precision. Nontraditional welders, such as artisans or handymen, may not have workplace safety education to be aware of UVR emitted from welding and may not have readily available PPE.
The Figure portrays an amateur welder working without full PPE. Although he is wearing a face shield, he is not wearing fire-retardant clothing, lacks full protective garments, and has no ventilation system.

Conclusion
It is important to recognize welding as an occupation with notable exposure to UVR. Personal protective equipment should be the mainstay of prevention. Sunscreen is a useful adjunct but does not cover UVC that is emitted in the welding arc. Screens and welding blankets can be placed around welders to contain UVR and limit nonwelder exposure. Although UVR hazards should be regulated in the workplace as part of regular safety reviews, the clinician can play a role in recognizing this source of UVR in skin disease and in encouraging the use of PPE.
Although solar radiation is the most commonly recognized source of UV radiation (UVR), occupational exposures can contribute due to the intensity and chronicity of exposure. Arc welding is a process whereby metal is fused together by heat produced from an electric arc. The electric arc that forms between the electrode and the base metal emits radiation in the full UV spectrum including UVA (400–315 nm), UVB (315–290 nm), and UVC (290–100 nm) wavelengths. Welders, therefore, have an increased risk for broad-spectrum, intense exposure to UVR, which may play a notable role in UV-related skin disease without proper protection. We report 3 welders with skin disease attributed to occupational exposure to UVR.
Case Reports
Patient 1
A 41-year-old man presented for evaluation of treatment-resistant cutaneous lupus. During the 10-year disease course, the patient was treated by both dermatologists and rheumatologists with frequent exacerbations and poor disease control. At the time of presentation, treatment with hydroxychloroquine 200 mg twice daily, azathioprine 50 mg twice daily, intramuscular methylprednisolone acetateinjectable suspension 40 mg, and prednisone 20 mg daily was failing. Physical examination revealed polycyclic erythematous plaques typical of subacute cutaneous lupus erythematosus. A skin biopsy confirmed the diagnosis. Upon further discussion of exacerbating risk factors, the patient noted UVR exposure while working as a welder. Although he had been previously told to avoid sunlight, he did not realize that this recommendation included all forms of UV light. Once this work exposure was eliminated, he was restarted on hydroxychloroquine 200 mg twice daily and topical steroids, and he responded with complete and sustained clearance of disease. When he returned to welding, utilization of sunscreen and sun-protective clothing enabled him to maintain control of his subacute cutaneous lupus erythematosus on oral hydroxychloroquine 200 mg twice daily and topical steroids.
Patient 2
A 55-year-old man presented with numerous actinic keratoses and persistent erythema in a well-demarcated area involving the forehead, temples, and lateral cheeks but sparing the periorbital area. The patient also experienced UVR exposure from welding (up to 4 to 5 times per week during his career spanning more than 20 years). He cited frequent burns in areas where his protective equipment did not cover his skin. He also reported that he often forgoes wearing protective equipment, even though it is available, and only uses safety goggles due to the extreme heat of the working environment as well as the awkwardness of wearing full protective gear while performing certain aspects of the job.
Patient 3
A 63-year-old man presented with a growth on the left side of the upper forehead. A biopsy revealed a squamous cell carcinoma, keratoacanthoma type. He worked as a welder for 40 years until retiring 1 year prior to presentation. He welded daily and always wore a tall face shield. Although the face shield covered most of his face, the scalp and some parts of the upper face were not well protected. In addition to the keratoacanthoma, which presented just outside of the area protected by the face shield, the patient had numerous actinic keratoses on the scalp.
Comment
Welding and UVR Exposure
Arc welders endure large amounts of UVR exposure, which is substantial enough to have notable health effects. The duration of exposure, electrical current used, angle of exposure, amount of ventilation, and the distance from the welding arc play a role in overall UVR exposure.1,2 Maximum permissible exposure (MPE) limits to UVR have been set by the International Commission on Non-Ionizing Radiation Protection and the National Institute for Occupational Safety and Health.3,4 The quantity of radiation produced by the arc allows for an exposure time of only a few seconds to minutes before surpassing MPE to UV light.1,5 Welders are exposed to total-body UVR doses up to 3000 times the MPE, and mean cumulative exposure calculated over an 8-hour workday can reach 9795 mJ/cm2.6
Workers in close proximity to welders also receive large UVR doses and may not be aware of its hazardous effects. Nearby nonwelders can be exposed to 13 times the MPE of UVR.6 At distances up to 10 m from the arc, the irradiance is large enough to reach MPE to UVR in less than 3 hours.1
Skin and Eye Damage From Welding
Exposure to UVR produced by the welding arc may lead to acute skin or eye reactions, chronic skin or eye disorders, or exacerbation of photosensitive diseases. Common acute problems are photokeratoconjunctivitis (welder’s flash) and skin erythema.7,8
Actinic elastosis, actinic keratoses, ocular melanoma, and photosensitive diseases represent a spectrum of disorders that can present from chronic UV exposure in welders. In a study by Emmett et al7 of 152 welders and 58 controls, actinic elastosis was found to be more frequent in welders than controls. Cases of basal cell carcinoma and squamous cell carcinoma also have been reported in welders.9,10 However, in the study by Emmett et al,7 a statistically significant correlation between welding and skin cancer was not documented. There were limitations in the study, such as small sample size and a young average age of welders.7 Future studies may be needed to further clarify the risk for skin cancer in welders.
Although there is no clear association with skin cancer, an increased risk of ocular melanoma in welders is more clearly established. A meta-analysis of 5 studies found that welding was a significant risk factor for ocular melanoma, with an odds ratio of 2.05 (95% confidence interval, 1.20-3.51).11 Other reported eye damage from chronic UVR exposure includes cataracts, chronic conjunctivitis, and retinal damage.12,13
Case reports of the following photosensitive diseases have been reported to be exacerbated or caused by UV light exposure in welders: discoid lupus erythematosus14; photodermatitis15; broadband photosensitivity with decreased minimal erythema dose to UVA, UVB, and UVC16; UVC-exacerbated atopic dermatitis17; polymorphous light eruption–like skin eruption18; and UVA-induced photoallergy to hydrochlorothiazide and ramipril.19
Prevention of Occupational Exposure to UVR
Occupational Safety and Health Administration guidelines protect workers from excessive exposure to UVR with personal protective equipment (PPE). In addition to UVR protection, PPE needs to protect welders from other risks including trauma from welding debris (slag), fires, electrical burns, and fumes. Online resources from the National Ag Safety Database,20 the American Welding Society,21 and Occupational Safety and Health Administration22,23 are available. These resources advise welders to work in ventilated areas with respirators specific for the metal being welded and to wear clothing and gloves that are not only fire retardant but also UV resistant.20-23 Additional PPE should protect the head, face, and eyes.
Unfortunately, even workers well trained in prevention guidelines may not adequately protect themselves. Some welders forego PPE due to heat, thus exposing themselves to UVR damage in areas that are normally covered. Welders also may forego equipment when working on jobs requiring more detailed welds where clothing, masks, and glasses may be overly bulky and inhibit the worker’s precision. Nontraditional welders, such as artisans or handymen, may not have workplace safety education to be aware of UVR emitted from welding and may not have readily available PPE.
The Figure portrays an amateur welder working without full PPE. Although he is wearing a face shield, he is not wearing fire-retardant clothing, lacks full protective garments, and has no ventilation system.

Conclusion
It is important to recognize welding as an occupation with notable exposure to UVR. Personal protective equipment should be the mainstay of prevention. Sunscreen is a useful adjunct but does not cover UVC that is emitted in the welding arc. Screens and welding blankets can be placed around welders to contain UVR and limit nonwelder exposure. Although UVR hazards should be regulated in the workplace as part of regular safety reviews, the clinician can play a role in recognizing this source of UVR in skin disease and in encouraging the use of PPE.
- Okuno T, Ojima J, Saito H. Ultraviolet radiation emitted by CO(2) arc welding. Ann Occup Hyg. 2001;45:597-601.
- Peng CY, Liu HH, Chang CP, et al. Evaluation and monitoring of UVR in shield metal ARC welding processing. Health Phys. 2007;93:101-108.
- The National Institute for Occupational Safety and Health. Criteria for a recommended standard: occupational exposure to ultraviolet radiation. DHHS (NIOSH) publication 73-11009. https://www.cdc.gov/niosh/docs/73-11009/. Updated June 6, 2014. Accessed September 6, 2019.
- International Commission on Non-Ionizing Radiation Protection. Guidelines on limits of exposure to ultraviolet radiation of wavelengths between 180 nm and 400 nm (incoherent optical radiation). Health Phys. 2004;87:171-186.
- Peng CY, Lan CH, Juang YJ, et al. Exposure assessment of aluminum arc welding radiation. Health Phys. 2007;93:298-306.
- Tenkate TD, Collins MJ. Personal ultraviolet radiation exposure of workers in a welding environment. Am Ind Hyg Assoc J. 1997;58:33-38.
- Emmett EA, Buncher CR, Suskind RB, et al. Skin and eye diseases among arc welders and those exposed to welding operations. J Occup Med. 1981;23:85-90.
- Bruze M, Hindsén M, Trulsson L. Dermatitis with an unusual explanation in a welder. Acta Derm Venereol. 1994;74:380-382.
- Donoghue AM, Sinclair MJ. Basal cell carcinoma after frequent episodes of cutaneous erythema and peeling induced by welding. Occup Environ Med. 1999;56:646.
- Currie CL, Monk BE. Welding and non-melanoma skin cancer. Clin Exp Dermatol. 2000;25:28-29.
- Shah CP, Weis E, Lajous M, et al. Intermittent and chronic ultraviolet light exposure and uveal melanoma: a meta-analysis. Ophthalmology. 2005;112:1599-1607.
- Yang X, Shao D, Ding X, et al. Chronic phototoxic maculopathy caused by welding arc in occupational welders. Can J Ophthalmol. 2012;47:45-50.
- Davies KG, Asanga U, Nku CO, et al. Effect of chronic exposure to welding light on Calabar welders. Niger J Physiol Sci. 2007;22:55-58.
- Wozniak KD. Erythematodes chronicus discoides as an occupational disease in an electric welder [in German]. Berufs-Dermatosen. 1971;19:187-196.
- Shehade SA, Roberts PJ, Diffey BL, et al. Photodermatitis due to spot welding. Br J Dermatol. 1987;117:117-119.
- Roelandts R, Huys I. Broad-band and persistent photosensitivity following accidental ultraviolet C overexposure. Photodermatol Photoimmunol Photomed. 1993;9:144-146.
- Elsner P, Hassam S. Occupational UVC-induced exacerbation of atopic dermatitis in a welder. Contact Dermatitis. 1996;35:180-181.
- Majoie IM, van Weelden H, Sybesma IM, et al. Polymorphous light eruption-like skin lesions in welders caused by ultraviolet C light. J Am Acad Dermatol. 2010;62:150-151.
- Wagner SN, Welke F, Goos M. Occupational UVA-induced allergic photodermatitis in a welder due to hydrochlorothiazide and ramipril. Contact Dermatitis. 2000;43:245-246.
- Fluegel L, Rein BK. Arc welding safety. National Ag Safety Database website. http://nasdonline.org/1083/d000873/arc-welding-safety.html. Published May 1989. Accessed September 6, 2019.
- American Welding Society. Personal protective equipment (PPE) for welding and cutting. Fact sheet no. 33-04/14. http://www.aws.org/technical/facts/FACT-33_2014.pdf. Published April 2014. Accessed September 6, 2019.
- Occupational Safety and Health Administration. Eye protection against radiant energy during welding and cutting in shipyard employment. https://www.osha.gov/Publications/OSHAfactsheet-eyeprotection-during-welding.pdf. Published January 2012. Accessed September 6, 2019.
- Occupational Safety and Health Administration. Welding, cutting, and brazing. https://www.osha.gov/SLTC/weldingcuttingbrazing/standards.html. Accessed September 10, 2019.
- Okuno T, Ojima J, Saito H. Ultraviolet radiation emitted by CO(2) arc welding. Ann Occup Hyg. 2001;45:597-601.
- Peng CY, Liu HH, Chang CP, et al. Evaluation and monitoring of UVR in shield metal ARC welding processing. Health Phys. 2007;93:101-108.
- The National Institute for Occupational Safety and Health. Criteria for a recommended standard: occupational exposure to ultraviolet radiation. DHHS (NIOSH) publication 73-11009. https://www.cdc.gov/niosh/docs/73-11009/. Updated June 6, 2014. Accessed September 6, 2019.
- International Commission on Non-Ionizing Radiation Protection. Guidelines on limits of exposure to ultraviolet radiation of wavelengths between 180 nm and 400 nm (incoherent optical radiation). Health Phys. 2004;87:171-186.
- Peng CY, Lan CH, Juang YJ, et al. Exposure assessment of aluminum arc welding radiation. Health Phys. 2007;93:298-306.
- Tenkate TD, Collins MJ. Personal ultraviolet radiation exposure of workers in a welding environment. Am Ind Hyg Assoc J. 1997;58:33-38.
- Emmett EA, Buncher CR, Suskind RB, et al. Skin and eye diseases among arc welders and those exposed to welding operations. J Occup Med. 1981;23:85-90.
- Bruze M, Hindsén M, Trulsson L. Dermatitis with an unusual explanation in a welder. Acta Derm Venereol. 1994;74:380-382.
- Donoghue AM, Sinclair MJ. Basal cell carcinoma after frequent episodes of cutaneous erythema and peeling induced by welding. Occup Environ Med. 1999;56:646.
- Currie CL, Monk BE. Welding and non-melanoma skin cancer. Clin Exp Dermatol. 2000;25:28-29.
- Shah CP, Weis E, Lajous M, et al. Intermittent and chronic ultraviolet light exposure and uveal melanoma: a meta-analysis. Ophthalmology. 2005;112:1599-1607.
- Yang X, Shao D, Ding X, et al. Chronic phototoxic maculopathy caused by welding arc in occupational welders. Can J Ophthalmol. 2012;47:45-50.
- Davies KG, Asanga U, Nku CO, et al. Effect of chronic exposure to welding light on Calabar welders. Niger J Physiol Sci. 2007;22:55-58.
- Wozniak KD. Erythematodes chronicus discoides as an occupational disease in an electric welder [in German]. Berufs-Dermatosen. 1971;19:187-196.
- Shehade SA, Roberts PJ, Diffey BL, et al. Photodermatitis due to spot welding. Br J Dermatol. 1987;117:117-119.
- Roelandts R, Huys I. Broad-band and persistent photosensitivity following accidental ultraviolet C overexposure. Photodermatol Photoimmunol Photomed. 1993;9:144-146.
- Elsner P, Hassam S. Occupational UVC-induced exacerbation of atopic dermatitis in a welder. Contact Dermatitis. 1996;35:180-181.
- Majoie IM, van Weelden H, Sybesma IM, et al. Polymorphous light eruption-like skin lesions in welders caused by ultraviolet C light. J Am Acad Dermatol. 2010;62:150-151.
- Wagner SN, Welke F, Goos M. Occupational UVA-induced allergic photodermatitis in a welder due to hydrochlorothiazide and ramipril. Contact Dermatitis. 2000;43:245-246.
- Fluegel L, Rein BK. Arc welding safety. National Ag Safety Database website. http://nasdonline.org/1083/d000873/arc-welding-safety.html. Published May 1989. Accessed September 6, 2019.
- American Welding Society. Personal protective equipment (PPE) for welding and cutting. Fact sheet no. 33-04/14. http://www.aws.org/technical/facts/FACT-33_2014.pdf. Published April 2014. Accessed September 6, 2019.
- Occupational Safety and Health Administration. Eye protection against radiant energy during welding and cutting in shipyard employment. https://www.osha.gov/Publications/OSHAfactsheet-eyeprotection-during-welding.pdf. Published January 2012. Accessed September 6, 2019.
- Occupational Safety and Health Administration. Welding, cutting, and brazing. https://www.osha.gov/SLTC/weldingcuttingbrazing/standards.html. Accessed September 10, 2019.
Practice Points
- Arc welding can be a major source of UV radiation exposure.
- Welders should be advised to work with proper ventilation and with welding masks, clothing, and gloves that not only are fire retardant but also are UV resistant.
Prior antibiotic use lowers checkpoint inhibitor response and survival
Prior antibiotic use may be associated with a reduced treatment response to checkpoint inhibitors, and worse outcomes, in patients with cancer, according to investigators.
In a prospective cohort study, researchers followed 196 patients with cancer who were treated with immune checkpoint inhibitors in routine clinical practice.
A total of 22 patients had been treated with a 7-day or less course of broad-spectrum beta-lactam–based antibiotics in the 30 days prior to starting immune checkpoint inhibitor therapy, and 68 patients were concurrently taking broad-spectrum beta-lactam–based antibiotics with their checkpoint inhibitor therapy.
The analysis revealed that prior antibiotic therapy was associated with nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy (P less than .001) and significantly worse overall survival (2 vs. 26 months). Patients who had been on prior antibiotic therapy were also more likely to stop checkpoint inhibitor therapy because their disease had progressed, and were more likely to die of progressive disease while on checkpoint inhibitors.
However, concurrent antibiotic use did not appear to affect either treatment response to checkpoint inhibitors or overall survival.
The most common indication for both prior and concurrent antibiotic use was respiratory tract infections. Researchers examined whether cancer type might play a role in contributing to the association; for example, chronic airway disease in lung cancer might mean higher likelihood of antibiotic use but also lower treatment response and survival.
They found that the association between prior antibiotic therapy and overall survival was consistent across the 119 patients with non–small cell lung cancer, the 38 patients with melanoma, and the 39 patients with other tumor types.
The association was also independent of the class of antibiotic used, the patient’s performance status, and their corticosteroid use.
“Broad-spectrum ATB [antibiotic] use can cause prolonged disruption of the gut ecosystem and impair the effectiveness of the cytotoxic T-cell response against cancer, strengthening the biologic plausibility underlying the adverse effect of ATB therapy on immunotherapy outcomes,” wrote Dr. David J. Pinato, from Imperial College London, and coauthors in JAMA Oncology.
Addressing the question of whether comorbidities might be the mediating factor, the authors pointed out that the use of antibiotics during checkpoint inhibitor therapy – which was a potential indicator of patients’ status worsening during treatment – was not associated with reduced response to treatment or lower overall survival.
“Although provision of cATB [concurrent antibiotic] therapy appears to be safe in the context of immunotherapy, clinicians should carefully weigh the pros and cons of prescribing broad-spectrum ATBs prior to ICI [immune checkpoint inhibitor] treatment,” they wrote.
The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
SOURCE: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
Prior antibiotic use may be associated with a reduced treatment response to checkpoint inhibitors, and worse outcomes, in patients with cancer, according to investigators.
In a prospective cohort study, researchers followed 196 patients with cancer who were treated with immune checkpoint inhibitors in routine clinical practice.
A total of 22 patients had been treated with a 7-day or less course of broad-spectrum beta-lactam–based antibiotics in the 30 days prior to starting immune checkpoint inhibitor therapy, and 68 patients were concurrently taking broad-spectrum beta-lactam–based antibiotics with their checkpoint inhibitor therapy.
The analysis revealed that prior antibiotic therapy was associated with nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy (P less than .001) and significantly worse overall survival (2 vs. 26 months). Patients who had been on prior antibiotic therapy were also more likely to stop checkpoint inhibitor therapy because their disease had progressed, and were more likely to die of progressive disease while on checkpoint inhibitors.
However, concurrent antibiotic use did not appear to affect either treatment response to checkpoint inhibitors or overall survival.
The most common indication for both prior and concurrent antibiotic use was respiratory tract infections. Researchers examined whether cancer type might play a role in contributing to the association; for example, chronic airway disease in lung cancer might mean higher likelihood of antibiotic use but also lower treatment response and survival.
They found that the association between prior antibiotic therapy and overall survival was consistent across the 119 patients with non–small cell lung cancer, the 38 patients with melanoma, and the 39 patients with other tumor types.
The association was also independent of the class of antibiotic used, the patient’s performance status, and their corticosteroid use.
“Broad-spectrum ATB [antibiotic] use can cause prolonged disruption of the gut ecosystem and impair the effectiveness of the cytotoxic T-cell response against cancer, strengthening the biologic plausibility underlying the adverse effect of ATB therapy on immunotherapy outcomes,” wrote Dr. David J. Pinato, from Imperial College London, and coauthors in JAMA Oncology.
Addressing the question of whether comorbidities might be the mediating factor, the authors pointed out that the use of antibiotics during checkpoint inhibitor therapy – which was a potential indicator of patients’ status worsening during treatment – was not associated with reduced response to treatment or lower overall survival.
“Although provision of cATB [concurrent antibiotic] therapy appears to be safe in the context of immunotherapy, clinicians should carefully weigh the pros and cons of prescribing broad-spectrum ATBs prior to ICI [immune checkpoint inhibitor] treatment,” they wrote.
The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
SOURCE: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
Prior antibiotic use may be associated with a reduced treatment response to checkpoint inhibitors, and worse outcomes, in patients with cancer, according to investigators.
In a prospective cohort study, researchers followed 196 patients with cancer who were treated with immune checkpoint inhibitors in routine clinical practice.
A total of 22 patients had been treated with a 7-day or less course of broad-spectrum beta-lactam–based antibiotics in the 30 days prior to starting immune checkpoint inhibitor therapy, and 68 patients were concurrently taking broad-spectrum beta-lactam–based antibiotics with their checkpoint inhibitor therapy.
The analysis revealed that prior antibiotic therapy was associated with nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy (P less than .001) and significantly worse overall survival (2 vs. 26 months). Patients who had been on prior antibiotic therapy were also more likely to stop checkpoint inhibitor therapy because their disease had progressed, and were more likely to die of progressive disease while on checkpoint inhibitors.
However, concurrent antibiotic use did not appear to affect either treatment response to checkpoint inhibitors or overall survival.
The most common indication for both prior and concurrent antibiotic use was respiratory tract infections. Researchers examined whether cancer type might play a role in contributing to the association; for example, chronic airway disease in lung cancer might mean higher likelihood of antibiotic use but also lower treatment response and survival.
They found that the association between prior antibiotic therapy and overall survival was consistent across the 119 patients with non–small cell lung cancer, the 38 patients with melanoma, and the 39 patients with other tumor types.
The association was also independent of the class of antibiotic used, the patient’s performance status, and their corticosteroid use.
“Broad-spectrum ATB [antibiotic] use can cause prolonged disruption of the gut ecosystem and impair the effectiveness of the cytotoxic T-cell response against cancer, strengthening the biologic plausibility underlying the adverse effect of ATB therapy on immunotherapy outcomes,” wrote Dr. David J. Pinato, from Imperial College London, and coauthors in JAMA Oncology.
Addressing the question of whether comorbidities might be the mediating factor, the authors pointed out that the use of antibiotics during checkpoint inhibitor therapy – which was a potential indicator of patients’ status worsening during treatment – was not associated with reduced response to treatment or lower overall survival.
“Although provision of cATB [concurrent antibiotic] therapy appears to be safe in the context of immunotherapy, clinicians should carefully weigh the pros and cons of prescribing broad-spectrum ATBs prior to ICI [immune checkpoint inhibitor] treatment,” they wrote.
The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
SOURCE: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
FROM JAMA ONCOLOGY
Key clinical point: People who take antibiotics prior to checkpoint inhibitor therapy have lower treatment response and overall survival.
Major finding: Prior antibiotic use is associated with a nearly a 100% greater likelihood of poor response to checkpoint inhibitor therapy.
Study details: A prospective cohort study involving 196 patients receiving checkpoint inhibitor therapy for cancer.
Disclosures: The study was supported by the Imperial College National Institute for Health Research Biomedical Research Centre, the Imperial College Tissue Bank, the Imperial Cancer Research U.K. Centre, the National Institute for Health Research, and the Wellcome Trust Strategic Fund. Two authors reported receiving grant funding and personal fees from the pharmaceutical sector unrelated to the study.
Source: Pinato D et al. JAMA Oncol. 2019 Sep 12. doi: 10.1001/jamaoncol.2019.2785.
Sniffing Out Malignant Melanoma: A Case of Canine Olfactory Detection
To the Editor:
A 43-year-old woman presented with a mole on the central back that had been present since childhood and had changed and grown over the last few years. The patient reported that her 2-year-old rescue dog frequently sniffed the mole and would subsequently get agitated and try to scratch and bite the lesion. This behavior prompted the patient to visit a dermatologist.
She reported no personal history of melanoma or nonmelanoma skin cancer, tanning booth exposure, blistering sunburns, or use of immunosuppressant medications. Her family history was remarkable for basal cell carcinoma in her father but no family history of melanoma. Physical examination revealed a 1.2×1.5-cm brown patch along with a 1×1-cm ulcerated nodule on the lower aspect of the lesion (Figure 1). Dermoscopy showed a blue-white veil and an irregular vascular pattern (Figure 2). No cervical, axillary, or inguinal lymphadenopathy was appreciated on physical examination. Reflectance confocal microscopy showed pagetoid spread of atypical round melanocytes as well as melanocytes in the stratum corneum (Figure 3).
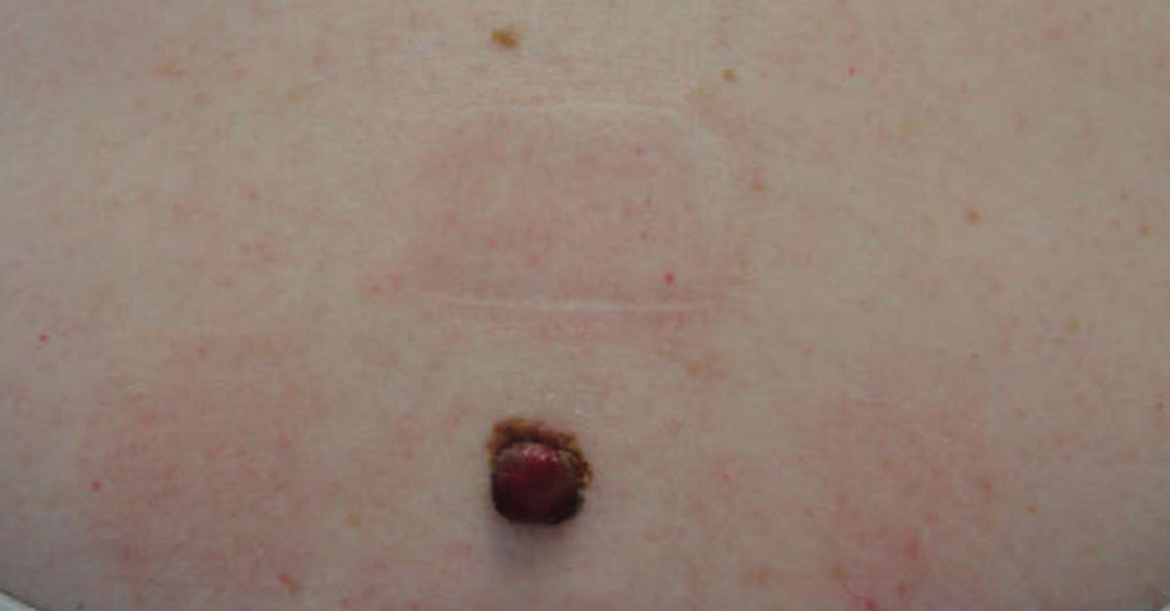
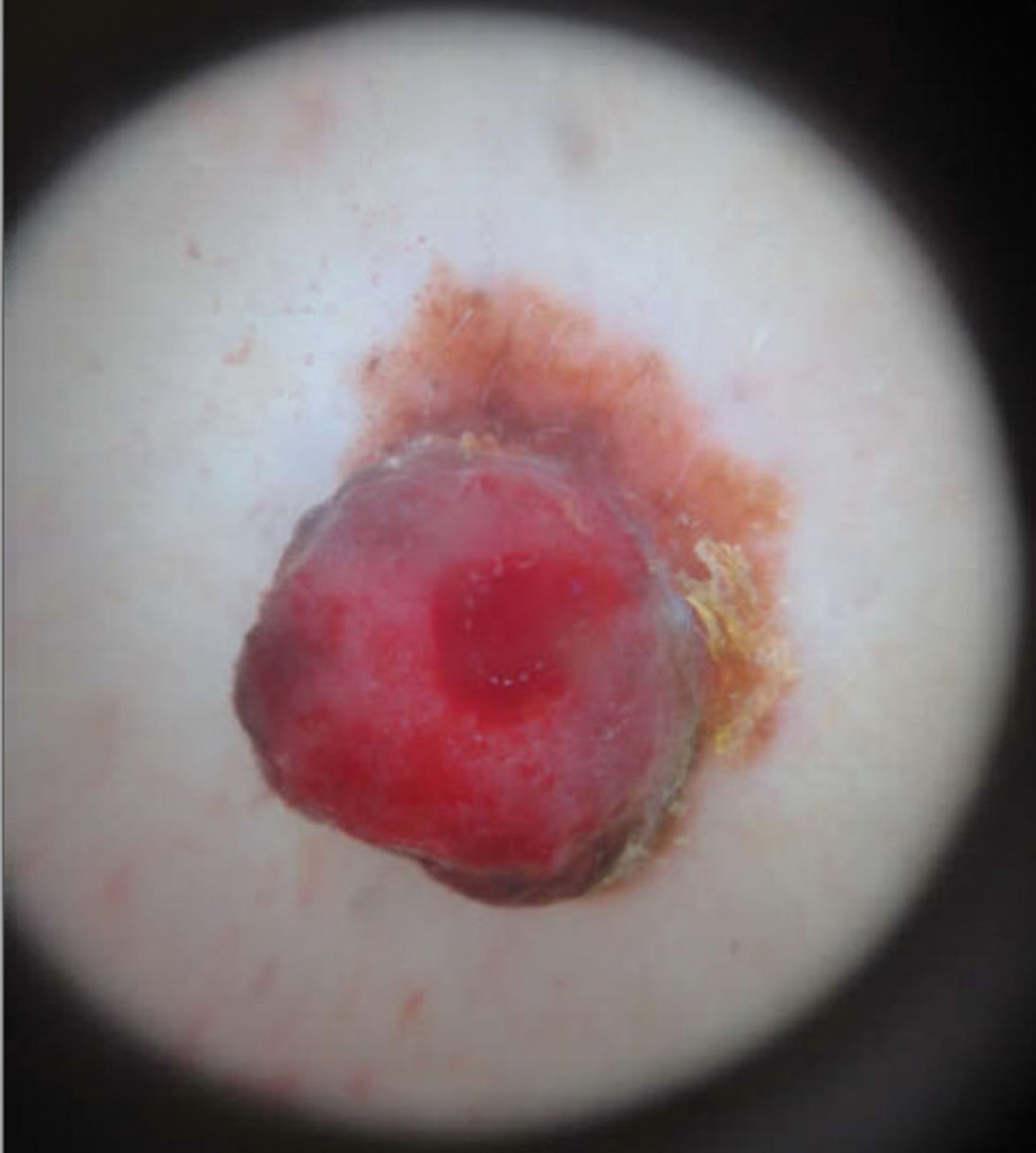

The patient was referred to a surgical oncologist for wide local excision and sentinel lymph node biopsy. Pathology showed a 4-mm-thick melanoma with numerous positive lymph nodes (Figure 4). The patient subsequently underwent a right axillary lymphadenectomy and was diagnosed with stage IIIB malignant melanoma. After surgery, the patient reported that her dog would now sniff her back and calmly rest his head in her lap.
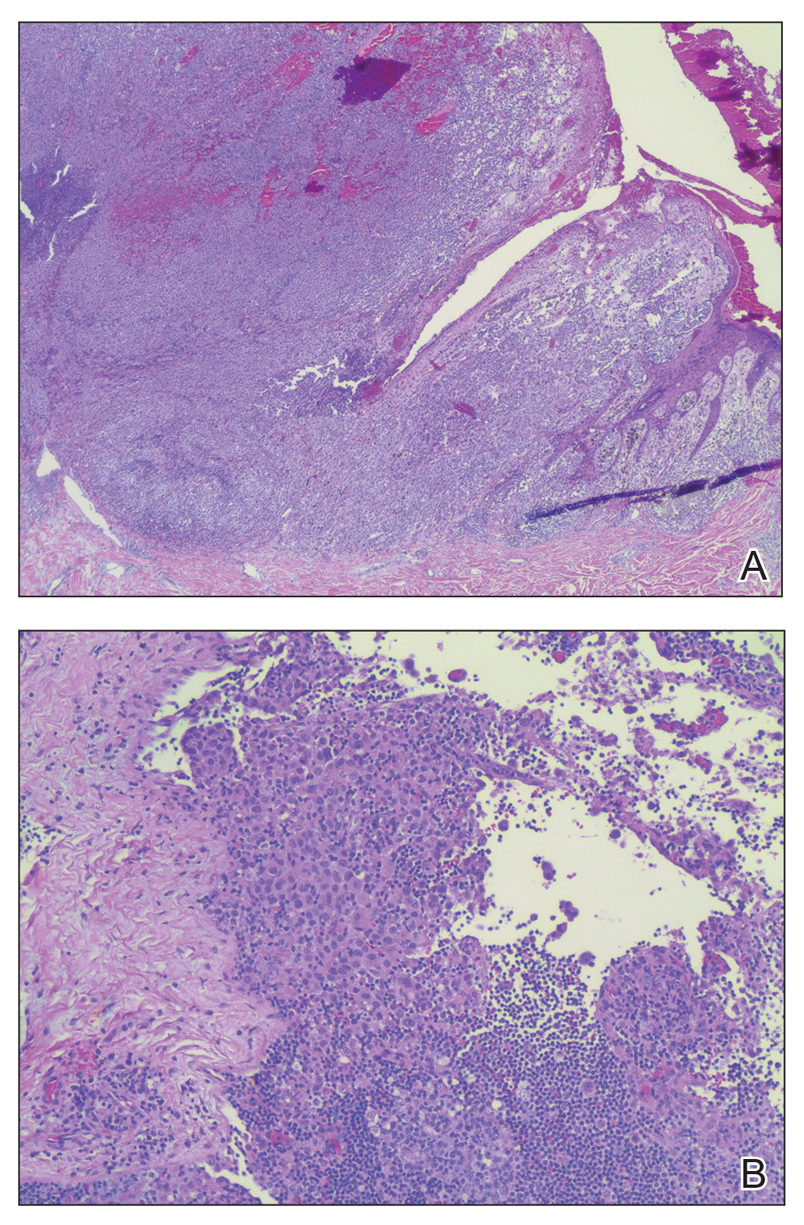
She was treated with ipilimumab but subsequently developed panhypopituitarism, so she was taken off the ipilimumab. Currently, the patient is doing well. She follows up annually for full-body skin examinations and has not had any recurrence in the last 7 years. The patient credits her dog for prompting her to see a dermatologist and saving her life.
Both anecdotal and systematic evidence have emerged on the role of canine olfaction in the detection of lung, breast, colorectal, ovarian, prostate, and skin cancers, including malignant melanoma.1-6 A 1989 case report described a woman who was prompted to seek dermatologic evaluation of a pigmented lesion because her dog consistently targeted the lesion. Excision and subsequent histopathologic examination of the lesion revealed that it was malignant melanoma.5 Another case report described a patient whose dog, which was not trained to detect cancers in humans, persistently licked a lesion behind the patient’s ear that eventually was found to be malignant melanoma.6 These reports have inspired considerable research interest regarding canine olfaction as a potential method to noninvasively screen for and even diagnose malignant melanomas in humans.
Both physiologic and pathologic metabolic processes result in the production of volatile organic compounds (VOCs), or small odorant molecules that evaporate at normal temperatures and pressures.1 Individual cells release VOCs in extremely low concentrations into the blood, urine, feces, and breath, as well as onto the skin’s surface, but there are methods for detecting these VOCs, including gas chromatography–mass spectrometry and canine olfaction.7,8 Pathologic processes, such as infection and malignancy, result in irregular protein synthesis and metabolism, producing new VOCs or differing concentrations of VOCs as compared to normal processes.1
Dimethyl disulfide and dimethyl trisulfide compounds have been identified in malignant melanoma, and these compounds are not produced by normal melanocytes.7 Furthermore, malignant melanoma produces differing quantities of these compounds as compared to normal melanocytes, including isovaleric acid, 2-methylbutyric acid, isoamyl alcohol (3-methyl-1-butanol), and 2-methyl-1-butanol, resulting in a distinct odorant profile that previously has been detected via canine olfaction.7 Canine olfaction can identify odorant molecules at up to 1 part per trillion (a magnitude more sensitive than the currently available gas chromatography–mass spectrometry technologies) and can detect the production of new VOCs or altered VOC ratios due to pathologic processes.1 Systematic studies with dogs that are trained to detect cancers in humans have shown that canine olfaction correctly identified malignant melanomas against healthy skin, benign nevi, and even basal cell carcinomas at higher rates than what would have been expected by chance alone.2,3
Canine olfaction can identify new or altered ratios of odorant VOCs associated with pathologic metabolic processes, and canines can be trained to target odor profiles associated with specific diseases.1 Canine olfaction for melanoma screening and diagnosis may seem appealing, as it provides an easily transportable, real-time, low-cost method compared to other techniques such as gas chromatography–mass spectrometry.1 Although preliminary results have shown that canine olfaction detects melanoma at higher rates than would be expected by chance alone, these findings have not approached clinical utility for the widespread use of canine olfaction as a screening method for melanoma.2,3,9 Further studies are needed to understand the role of canine olfaction in melanoma screening and diagnosis as well as to explore methods to optimize sensitivity and specificity. Until then, patients and dermatologists should not ignore the behavior of dogs toward skin lesions. Dogs may be beneficial in the detection of melanoma and help save lives, as was seen in our case.
- Angle C, Waggoner LP, Ferrando A, et al. Canine detection of the volatilome: a review of implications for pathogen and disease detection. Front Vet Sci. 2016;3:47.
- Pickel D, Mauncy GP, Walker DB, et al. Evidence for canine olfactory detection of melanoma. Applied Animal Behaviour Science. 2004;89:107-116.
- Willis CM, Britton LE, Swindells MA, et al. Invasive melanoma in vivo can be distinguished from basal cell carcinoma, benign naevi and healthy skin by canine olfaction: a proof‐of‐principle study of differential volatile organic compound emission. Br J Dermatol. 2016;175:1020-1029.
- Jezierski T, Walczak M, Ligor T, et al. Study of the art: canine olfaction used for cancer detection on the basis of breath odour. perspectives and limitations. J Breath Res. 2015;9:027001.
- Williams H, Pembroke A. Sniffer dogs in the melanoma clinic? Lancet. 1989;1:734.
- Campbell LF, Farmery L, George SM, et al. Canine olfactory detection of malignant melanoma. BMJ Case Rep. 2013. doi:10.1136/bcr-2013-008566.
- Kwak J, Gallagher M, Ozdener MH, et al. Volatile biomarkers from human melanoma cells. J Chromotogr B Analyt Technol Biomed Life Sci. 2013;931:90-96.
- D’Amico A, Bono R, Pennazza G, et al. Identification of melanoma with a gas sensor array. Skin Res Technol. 2008;14:226-236.
- Elliker KR, Williams HC. Detection of skin cancer odours using dogs: a step forward in melanoma detection training and research methodologies. Br J Dermatol. 2016;175:851-852.
To the Editor:
A 43-year-old woman presented with a mole on the central back that had been present since childhood and had changed and grown over the last few years. The patient reported that her 2-year-old rescue dog frequently sniffed the mole and would subsequently get agitated and try to scratch and bite the lesion. This behavior prompted the patient to visit a dermatologist.
She reported no personal history of melanoma or nonmelanoma skin cancer, tanning booth exposure, blistering sunburns, or use of immunosuppressant medications. Her family history was remarkable for basal cell carcinoma in her father but no family history of melanoma. Physical examination revealed a 1.2×1.5-cm brown patch along with a 1×1-cm ulcerated nodule on the lower aspect of the lesion (Figure 1). Dermoscopy showed a blue-white veil and an irregular vascular pattern (Figure 2). No cervical, axillary, or inguinal lymphadenopathy was appreciated on physical examination. Reflectance confocal microscopy showed pagetoid spread of atypical round melanocytes as well as melanocytes in the stratum corneum (Figure 3).



The patient was referred to a surgical oncologist for wide local excision and sentinel lymph node biopsy. Pathology showed a 4-mm-thick melanoma with numerous positive lymph nodes (Figure 4). The patient subsequently underwent a right axillary lymphadenectomy and was diagnosed with stage IIIB malignant melanoma. After surgery, the patient reported that her dog would now sniff her back and calmly rest his head in her lap.

She was treated with ipilimumab but subsequently developed panhypopituitarism, so she was taken off the ipilimumab. Currently, the patient is doing well. She follows up annually for full-body skin examinations and has not had any recurrence in the last 7 years. The patient credits her dog for prompting her to see a dermatologist and saving her life.
Both anecdotal and systematic evidence have emerged on the role of canine olfaction in the detection of lung, breast, colorectal, ovarian, prostate, and skin cancers, including malignant melanoma.1-6 A 1989 case report described a woman who was prompted to seek dermatologic evaluation of a pigmented lesion because her dog consistently targeted the lesion. Excision and subsequent histopathologic examination of the lesion revealed that it was malignant melanoma.5 Another case report described a patient whose dog, which was not trained to detect cancers in humans, persistently licked a lesion behind the patient’s ear that eventually was found to be malignant melanoma.6 These reports have inspired considerable research interest regarding canine olfaction as a potential method to noninvasively screen for and even diagnose malignant melanomas in humans.
Both physiologic and pathologic metabolic processes result in the production of volatile organic compounds (VOCs), or small odorant molecules that evaporate at normal temperatures and pressures.1 Individual cells release VOCs in extremely low concentrations into the blood, urine, feces, and breath, as well as onto the skin’s surface, but there are methods for detecting these VOCs, including gas chromatography–mass spectrometry and canine olfaction.7,8 Pathologic processes, such as infection and malignancy, result in irregular protein synthesis and metabolism, producing new VOCs or differing concentrations of VOCs as compared to normal processes.1
Dimethyl disulfide and dimethyl trisulfide compounds have been identified in malignant melanoma, and these compounds are not produced by normal melanocytes.7 Furthermore, malignant melanoma produces differing quantities of these compounds as compared to normal melanocytes, including isovaleric acid, 2-methylbutyric acid, isoamyl alcohol (3-methyl-1-butanol), and 2-methyl-1-butanol, resulting in a distinct odorant profile that previously has been detected via canine olfaction.7 Canine olfaction can identify odorant molecules at up to 1 part per trillion (a magnitude more sensitive than the currently available gas chromatography–mass spectrometry technologies) and can detect the production of new VOCs or altered VOC ratios due to pathologic processes.1 Systematic studies with dogs that are trained to detect cancers in humans have shown that canine olfaction correctly identified malignant melanomas against healthy skin, benign nevi, and even basal cell carcinomas at higher rates than what would have been expected by chance alone.2,3
Canine olfaction can identify new or altered ratios of odorant VOCs associated with pathologic metabolic processes, and canines can be trained to target odor profiles associated with specific diseases.1 Canine olfaction for melanoma screening and diagnosis may seem appealing, as it provides an easily transportable, real-time, low-cost method compared to other techniques such as gas chromatography–mass spectrometry.1 Although preliminary results have shown that canine olfaction detects melanoma at higher rates than would be expected by chance alone, these findings have not approached clinical utility for the widespread use of canine olfaction as a screening method for melanoma.2,3,9 Further studies are needed to understand the role of canine olfaction in melanoma screening and diagnosis as well as to explore methods to optimize sensitivity and specificity. Until then, patients and dermatologists should not ignore the behavior of dogs toward skin lesions. Dogs may be beneficial in the detection of melanoma and help save lives, as was seen in our case.
To the Editor:
A 43-year-old woman presented with a mole on the central back that had been present since childhood and had changed and grown over the last few years. The patient reported that her 2-year-old rescue dog frequently sniffed the mole and would subsequently get agitated and try to scratch and bite the lesion. This behavior prompted the patient to visit a dermatologist.
She reported no personal history of melanoma or nonmelanoma skin cancer, tanning booth exposure, blistering sunburns, or use of immunosuppressant medications. Her family history was remarkable for basal cell carcinoma in her father but no family history of melanoma. Physical examination revealed a 1.2×1.5-cm brown patch along with a 1×1-cm ulcerated nodule on the lower aspect of the lesion (Figure 1). Dermoscopy showed a blue-white veil and an irregular vascular pattern (Figure 2). No cervical, axillary, or inguinal lymphadenopathy was appreciated on physical examination. Reflectance confocal microscopy showed pagetoid spread of atypical round melanocytes as well as melanocytes in the stratum corneum (Figure 3).



The patient was referred to a surgical oncologist for wide local excision and sentinel lymph node biopsy. Pathology showed a 4-mm-thick melanoma with numerous positive lymph nodes (Figure 4). The patient subsequently underwent a right axillary lymphadenectomy and was diagnosed with stage IIIB malignant melanoma. After surgery, the patient reported that her dog would now sniff her back and calmly rest his head in her lap.

She was treated with ipilimumab but subsequently developed panhypopituitarism, so she was taken off the ipilimumab. Currently, the patient is doing well. She follows up annually for full-body skin examinations and has not had any recurrence in the last 7 years. The patient credits her dog for prompting her to see a dermatologist and saving her life.
Both anecdotal and systematic evidence have emerged on the role of canine olfaction in the detection of lung, breast, colorectal, ovarian, prostate, and skin cancers, including malignant melanoma.1-6 A 1989 case report described a woman who was prompted to seek dermatologic evaluation of a pigmented lesion because her dog consistently targeted the lesion. Excision and subsequent histopathologic examination of the lesion revealed that it was malignant melanoma.5 Another case report described a patient whose dog, which was not trained to detect cancers in humans, persistently licked a lesion behind the patient’s ear that eventually was found to be malignant melanoma.6 These reports have inspired considerable research interest regarding canine olfaction as a potential method to noninvasively screen for and even diagnose malignant melanomas in humans.
Both physiologic and pathologic metabolic processes result in the production of volatile organic compounds (VOCs), or small odorant molecules that evaporate at normal temperatures and pressures.1 Individual cells release VOCs in extremely low concentrations into the blood, urine, feces, and breath, as well as onto the skin’s surface, but there are methods for detecting these VOCs, including gas chromatography–mass spectrometry and canine olfaction.7,8 Pathologic processes, such as infection and malignancy, result in irregular protein synthesis and metabolism, producing new VOCs or differing concentrations of VOCs as compared to normal processes.1
Dimethyl disulfide and dimethyl trisulfide compounds have been identified in malignant melanoma, and these compounds are not produced by normal melanocytes.7 Furthermore, malignant melanoma produces differing quantities of these compounds as compared to normal melanocytes, including isovaleric acid, 2-methylbutyric acid, isoamyl alcohol (3-methyl-1-butanol), and 2-methyl-1-butanol, resulting in a distinct odorant profile that previously has been detected via canine olfaction.7 Canine olfaction can identify odorant molecules at up to 1 part per trillion (a magnitude more sensitive than the currently available gas chromatography–mass spectrometry technologies) and can detect the production of new VOCs or altered VOC ratios due to pathologic processes.1 Systematic studies with dogs that are trained to detect cancers in humans have shown that canine olfaction correctly identified malignant melanomas against healthy skin, benign nevi, and even basal cell carcinomas at higher rates than what would have been expected by chance alone.2,3
Canine olfaction can identify new or altered ratios of odorant VOCs associated with pathologic metabolic processes, and canines can be trained to target odor profiles associated with specific diseases.1 Canine olfaction for melanoma screening and diagnosis may seem appealing, as it provides an easily transportable, real-time, low-cost method compared to other techniques such as gas chromatography–mass spectrometry.1 Although preliminary results have shown that canine olfaction detects melanoma at higher rates than would be expected by chance alone, these findings have not approached clinical utility for the widespread use of canine olfaction as a screening method for melanoma.2,3,9 Further studies are needed to understand the role of canine olfaction in melanoma screening and diagnosis as well as to explore methods to optimize sensitivity and specificity. Until then, patients and dermatologists should not ignore the behavior of dogs toward skin lesions. Dogs may be beneficial in the detection of melanoma and help save lives, as was seen in our case.
- Angle C, Waggoner LP, Ferrando A, et al. Canine detection of the volatilome: a review of implications for pathogen and disease detection. Front Vet Sci. 2016;3:47.
- Pickel D, Mauncy GP, Walker DB, et al. Evidence for canine olfactory detection of melanoma. Applied Animal Behaviour Science. 2004;89:107-116.
- Willis CM, Britton LE, Swindells MA, et al. Invasive melanoma in vivo can be distinguished from basal cell carcinoma, benign naevi and healthy skin by canine olfaction: a proof‐of‐principle study of differential volatile organic compound emission. Br J Dermatol. 2016;175:1020-1029.
- Jezierski T, Walczak M, Ligor T, et al. Study of the art: canine olfaction used for cancer detection on the basis of breath odour. perspectives and limitations. J Breath Res. 2015;9:027001.
- Williams H, Pembroke A. Sniffer dogs in the melanoma clinic? Lancet. 1989;1:734.
- Campbell LF, Farmery L, George SM, et al. Canine olfactory detection of malignant melanoma. BMJ Case Rep. 2013. doi:10.1136/bcr-2013-008566.
- Kwak J, Gallagher M, Ozdener MH, et al. Volatile biomarkers from human melanoma cells. J Chromotogr B Analyt Technol Biomed Life Sci. 2013;931:90-96.
- D’Amico A, Bono R, Pennazza G, et al. Identification of melanoma with a gas sensor array. Skin Res Technol. 2008;14:226-236.
- Elliker KR, Williams HC. Detection of skin cancer odours using dogs: a step forward in melanoma detection training and research methodologies. Br J Dermatol. 2016;175:851-852.
- Angle C, Waggoner LP, Ferrando A, et al. Canine detection of the volatilome: a review of implications for pathogen and disease detection. Front Vet Sci. 2016;3:47.
- Pickel D, Mauncy GP, Walker DB, et al. Evidence for canine olfactory detection of melanoma. Applied Animal Behaviour Science. 2004;89:107-116.
- Willis CM, Britton LE, Swindells MA, et al. Invasive melanoma in vivo can be distinguished from basal cell carcinoma, benign naevi and healthy skin by canine olfaction: a proof‐of‐principle study of differential volatile organic compound emission. Br J Dermatol. 2016;175:1020-1029.
- Jezierski T, Walczak M, Ligor T, et al. Study of the art: canine olfaction used for cancer detection on the basis of breath odour. perspectives and limitations. J Breath Res. 2015;9:027001.
- Williams H, Pembroke A. Sniffer dogs in the melanoma clinic? Lancet. 1989;1:734.
- Campbell LF, Farmery L, George SM, et al. Canine olfactory detection of malignant melanoma. BMJ Case Rep. 2013. doi:10.1136/bcr-2013-008566.
- Kwak J, Gallagher M, Ozdener MH, et al. Volatile biomarkers from human melanoma cells. J Chromotogr B Analyt Technol Biomed Life Sci. 2013;931:90-96.
- D’Amico A, Bono R, Pennazza G, et al. Identification of melanoma with a gas sensor array. Skin Res Technol. 2008;14:226-236.
- Elliker KR, Williams HC. Detection of skin cancer odours using dogs: a step forward in melanoma detection training and research methodologies. Br J Dermatol. 2016;175:851-852.
Practice Points
- Physiologic and pathologic processes produce volatile organic compounds in the skin and other tissues.
- Malignant melanocytes release unique volatile organic compounds (VOCs) as well as differing combinations and quantities of VOCs as compared to normal melanocytes.
- Volatile organic compounds released at the skin’s surface can be detected by various methods, including canine olfaction; therefore, unusual canine behavior toward skin lesions should not be ignored.
Characterization of Adverse Reactions to ‘4-week’ Nivolumab Dosing
Background: Nivolumab was recently approved for a new flat-dose schedule 480 mg IV every 4 weeks (“480 Q4w”) using data from pharmacokinetics simulations without being first tested directly in humans. We noted several unusual adverse drug reactions (ADRs) using the new dosing and hypothesized that this new dose schedule might generate more ADRs than prior dosing schedules.
Methods: This study attempts to summarize and characterize the types of ADRs seen on the new 480 Q4w dosing. We conducted a retrospective, descriptive chart review and case series including patients at the San Antonio VA Hematology/Oncology clinic treated with at least one dose of Nivolumab 480 mg between 2/1/18 and 10/1/18. We tracked whether these patients developed ADRs, and if so, the highest CTCAE 4.03 grade of reaction, the number of treatments before the reaction developed, and whether the reaction influenced treatment (hold treatment, stop treatment, dose change).
Results: 18 patients matched this criterion (all male, average age 67.6 years). 6 patients experienced an ADR during treatment with the 480 Q4w dose. Grade 1 toxicities included pruritis, abdominal pain, skin rash, fatigue, fever, cramping, myalgia, and diarrhea. There was a Grade 3 case of encephalopathy and a Grade 2 case of diplopia. Of the 6 patients who experienced an adverse drug reaction, 2 (with only Grade 1 toxicities) continued treatment at their same dose frequency; the others changed to 240 mg Q2w. All 4 patients who experienced an ADR and had their dose changed to 240 mg Q2w experienced resolution or improvement in their symptoms except for 1 patient’s complaint of abdominal pain.
Conclusion: 480 Q4w dosing of Nivolumab may have a different ADR profile from prior dose regimens; further quantitative analysis will be required to answer this question. Dose frequency change may present an opportunity to relieve toxicities while allowing patients to continue treatment.
Background: Nivolumab was recently approved for a new flat-dose schedule 480 mg IV every 4 weeks (“480 Q4w”) using data from pharmacokinetics simulations without being first tested directly in humans. We noted several unusual adverse drug reactions (ADRs) using the new dosing and hypothesized that this new dose schedule might generate more ADRs than prior dosing schedules.
Methods: This study attempts to summarize and characterize the types of ADRs seen on the new 480 Q4w dosing. We conducted a retrospective, descriptive chart review and case series including patients at the San Antonio VA Hematology/Oncology clinic treated with at least one dose of Nivolumab 480 mg between 2/1/18 and 10/1/18. We tracked whether these patients developed ADRs, and if so, the highest CTCAE 4.03 grade of reaction, the number of treatments before the reaction developed, and whether the reaction influenced treatment (hold treatment, stop treatment, dose change).
Results: 18 patients matched this criterion (all male, average age 67.6 years). 6 patients experienced an ADR during treatment with the 480 Q4w dose. Grade 1 toxicities included pruritis, abdominal pain, skin rash, fatigue, fever, cramping, myalgia, and diarrhea. There was a Grade 3 case of encephalopathy and a Grade 2 case of diplopia. Of the 6 patients who experienced an adverse drug reaction, 2 (with only Grade 1 toxicities) continued treatment at their same dose frequency; the others changed to 240 mg Q2w. All 4 patients who experienced an ADR and had their dose changed to 240 mg Q2w experienced resolution or improvement in their symptoms except for 1 patient’s complaint of abdominal pain.
Conclusion: 480 Q4w dosing of Nivolumab may have a different ADR profile from prior dose regimens; further quantitative analysis will be required to answer this question. Dose frequency change may present an opportunity to relieve toxicities while allowing patients to continue treatment.
Background: Nivolumab was recently approved for a new flat-dose schedule 480 mg IV every 4 weeks (“480 Q4w”) using data from pharmacokinetics simulations without being first tested directly in humans. We noted several unusual adverse drug reactions (ADRs) using the new dosing and hypothesized that this new dose schedule might generate more ADRs than prior dosing schedules.
Methods: This study attempts to summarize and characterize the types of ADRs seen on the new 480 Q4w dosing. We conducted a retrospective, descriptive chart review and case series including patients at the San Antonio VA Hematology/Oncology clinic treated with at least one dose of Nivolumab 480 mg between 2/1/18 and 10/1/18. We tracked whether these patients developed ADRs, and if so, the highest CTCAE 4.03 grade of reaction, the number of treatments before the reaction developed, and whether the reaction influenced treatment (hold treatment, stop treatment, dose change).
Results: 18 patients matched this criterion (all male, average age 67.6 years). 6 patients experienced an ADR during treatment with the 480 Q4w dose. Grade 1 toxicities included pruritis, abdominal pain, skin rash, fatigue, fever, cramping, myalgia, and diarrhea. There was a Grade 3 case of encephalopathy and a Grade 2 case of diplopia. Of the 6 patients who experienced an adverse drug reaction, 2 (with only Grade 1 toxicities) continued treatment at their same dose frequency; the others changed to 240 mg Q2w. All 4 patients who experienced an ADR and had their dose changed to 240 mg Q2w experienced resolution or improvement in their symptoms except for 1 patient’s complaint of abdominal pain.
Conclusion: 480 Q4w dosing of Nivolumab may have a different ADR profile from prior dose regimens; further quantitative analysis will be required to answer this question. Dose frequency change may present an opportunity to relieve toxicities while allowing patients to continue treatment.
Melanoma of Unknown Primary Presenting as a Parotid Gland Mass
Background: Malignant melanoma is an aggressive malignancy that can present as a poorly differentiated neoplasm. Loss of S100 and melanA antigenicity can make pathologic identification difficult, especially in those patients who lack a cutaneous primary lesion. Immunostaining with SOX10, a key nuclear transcription factor in the differentiation of neural crest progenitor cells to melanocytes, has a high reported sensitivity and specificity for pathologic identification of melanoma in difficult cases.
Case Report: A 69-year-old male with a history of heavy tobacco use presented to the otolaryngology clinic with a left parotid mass. He underwent a parotid gland biopsy, which was significant for a high grade, poorly differentiated malignancy of unclear primary source. A staging PET/CT demonstrated localized hypermetabolic activity in the draining left cervical lymph node basins. He underwent a left modified radical neck dissection and parotidectomy. Pathologic assessment demonstrated a 3.9 × 1.6 × 1.6 cm3 poorly differentiated carcinoma with perineural invasion and 8/85 lymph nodes involved. Morphologically, it had features of a high grade epithelioid tumor with spindle cell features. Immunohistochemical (IHC) stains were negative for epithelial markers (AE1/3, EMA, CK5/6, CAM5.2), smooth muscle actin, CD34, S100, and melanA. Given the concern for a spindle cell melanoma that lost its antigenicity for S100 and melanA, a SOX10 IHC stain was performed.
The SOX10 immunostain demonstrated strong, diffuse positivity which secured the diagnosis of malignant melanoma. Molecular testing for BRAF and KIT mutations was negative. The nal diagnosis was a stage IVA (pT2pN2bM0) malignant melanoma of the parotid gland without a cutaneous primary lesion. The patient received a course of adjuvant radiation to a total dose of 66Gy and will complete one year of adjuvant immunotherapy with Nivolumab.
Conclusion: Malignant melanoma can present as a poorly differentiated malignancy and may be difficult to diagnose by providers, especially in the absence of a typical clinical history and a primary cutaneous lesion. In cases where the standard melanoma immunostains are negative, IHC staining with SOX10 can help secure the diagnosis with high sensitivity and specificity.
Background: Malignant melanoma is an aggressive malignancy that can present as a poorly differentiated neoplasm. Loss of S100 and melanA antigenicity can make pathologic identification difficult, especially in those patients who lack a cutaneous primary lesion. Immunostaining with SOX10, a key nuclear transcription factor in the differentiation of neural crest progenitor cells to melanocytes, has a high reported sensitivity and specificity for pathologic identification of melanoma in difficult cases.
Case Report: A 69-year-old male with a history of heavy tobacco use presented to the otolaryngology clinic with a left parotid mass. He underwent a parotid gland biopsy, which was significant for a high grade, poorly differentiated malignancy of unclear primary source. A staging PET/CT demonstrated localized hypermetabolic activity in the draining left cervical lymph node basins. He underwent a left modified radical neck dissection and parotidectomy. Pathologic assessment demonstrated a 3.9 × 1.6 × 1.6 cm3 poorly differentiated carcinoma with perineural invasion and 8/85 lymph nodes involved. Morphologically, it had features of a high grade epithelioid tumor with spindle cell features. Immunohistochemical (IHC) stains were negative for epithelial markers (AE1/3, EMA, CK5/6, CAM5.2), smooth muscle actin, CD34, S100, and melanA. Given the concern for a spindle cell melanoma that lost its antigenicity for S100 and melanA, a SOX10 IHC stain was performed.
The SOX10 immunostain demonstrated strong, diffuse positivity which secured the diagnosis of malignant melanoma. Molecular testing for BRAF and KIT mutations was negative. The nal diagnosis was a stage IVA (pT2pN2bM0) malignant melanoma of the parotid gland without a cutaneous primary lesion. The patient received a course of adjuvant radiation to a total dose of 66Gy and will complete one year of adjuvant immunotherapy with Nivolumab.
Conclusion: Malignant melanoma can present as a poorly differentiated malignancy and may be difficult to diagnose by providers, especially in the absence of a typical clinical history and a primary cutaneous lesion. In cases where the standard melanoma immunostains are negative, IHC staining with SOX10 can help secure the diagnosis with high sensitivity and specificity.
Background: Malignant melanoma is an aggressive malignancy that can present as a poorly differentiated neoplasm. Loss of S100 and melanA antigenicity can make pathologic identification difficult, especially in those patients who lack a cutaneous primary lesion. Immunostaining with SOX10, a key nuclear transcription factor in the differentiation of neural crest progenitor cells to melanocytes, has a high reported sensitivity and specificity for pathologic identification of melanoma in difficult cases.
Case Report: A 69-year-old male with a history of heavy tobacco use presented to the otolaryngology clinic with a left parotid mass. He underwent a parotid gland biopsy, which was significant for a high grade, poorly differentiated malignancy of unclear primary source. A staging PET/CT demonstrated localized hypermetabolic activity in the draining left cervical lymph node basins. He underwent a left modified radical neck dissection and parotidectomy. Pathologic assessment demonstrated a 3.9 × 1.6 × 1.6 cm3 poorly differentiated carcinoma with perineural invasion and 8/85 lymph nodes involved. Morphologically, it had features of a high grade epithelioid tumor with spindle cell features. Immunohistochemical (IHC) stains were negative for epithelial markers (AE1/3, EMA, CK5/6, CAM5.2), smooth muscle actin, CD34, S100, and melanA. Given the concern for a spindle cell melanoma that lost its antigenicity for S100 and melanA, a SOX10 IHC stain was performed.
The SOX10 immunostain demonstrated strong, diffuse positivity which secured the diagnosis of malignant melanoma. Molecular testing for BRAF and KIT mutations was negative. The nal diagnosis was a stage IVA (pT2pN2bM0) malignant melanoma of the parotid gland without a cutaneous primary lesion. The patient received a course of adjuvant radiation to a total dose of 66Gy and will complete one year of adjuvant immunotherapy with Nivolumab.
Conclusion: Malignant melanoma can present as a poorly differentiated malignancy and may be difficult to diagnose by providers, especially in the absence of a typical clinical history and a primary cutaneous lesion. In cases where the standard melanoma immunostains are negative, IHC staining with SOX10 can help secure the diagnosis with high sensitivity and specificity.
Severe lipodystrophy linked to pembrolizumab
according to a case study.
A 47-year-old woman received pembrolizumab to treat metastatic melanoma and developed immune-related generalized acquired lipodystrophy. The condition has persisted 12 months after she stopped taking pembrolizumab.
Julie Delyon, MD, PhD, of Saint-Louis Hospital in Paris, and colleagues described this case in the British Journal of Dermatology.
The patient was diagnosed with BRAF-mutated, stage IV melanoma with bone and lymph node metastases. She received pembrolizumab at 2 mg/kg every 3 weeks as first-line treatment. She achieved a complete response and was still in remission at last follow-up.
The patient was obese at baseline, with a body mass index of 40 kg/m2, but she did not have diabetes, hepatic steatosis, or dyslipidemia.
Within 2 months of starting pembrolizumab, the patient observed “some major changes to her physical appearance,” according to Dr. Delyon and colleagues. At 10 months, the patient had severe lipodystrophy.
The patient experienced a change in fat distribution – namely, severe peripheral lipoatrophy and an accumulation of trunk fat. Imaging revealed a decrease in subcutaneous adipose tissue, which contrasted with an increase in visceral fat observed from baseline.
The patient also developed “moderate hirsutism, facial and limb atrophy, and prominent forearm and leg muscles and veins,” according to the authors. She had impaired glucose tolerance with insulin resistance, reduced concentrations of leptin and adiponectin, hypertriglyceridemia, a low level of HDL cholesterol, and hepatic steatosis.
Analyses of subcutaneous fat revealed adipose tissue atrophy with edema, lipophages, and CD3+/CD4+ T-cell infiltration of the fat and vessel walls. This suggested that the lipodystrophy had an autoimmune origin, according to the authors.
The patient had no family history of autoimmune disease or lipodystrophy. She tested negative for mutations in 23 genes associated with generalized lipodystrophy.
The patient also tested negative for HIV, antinuclear antibodies, native anti-DNA, and anti-insulin receptor antibodies. There were no signs of panniculitis, and the authors noted that “there were no arguments in favor of hyperthyroidism, Cushing syndrome, or acromegaly.”
In an attempt to reverse the lipodystrophy, the researchers stopped pembrolizumab treatment. The patient was treated for diabetes and hypertriglyceridemia as well. She could not receive corticosteroids because of the risk of severe metabolic complications, and she didn’t receive metreleptin because it wasn’t available. The patient still had lipodystrophy 12 months after stopping pembrolizumab.
Dr. Delyon and colleagues wrote that this case suggests pembrolizumab, and perhaps other anti–programmed death 1 therapies, may cause lipodystrophy with severe metabolic complications. “The long-term side effects of such metabolic adverse events, although rare, are unknown and will probably become a topic of utmost importance, considering the increasing rate of remission following ICIs and their use in the adjuvant setting.”
Two coauthors reported relationships with Merck, which markets pembrolizumab as Keytruda. The authors also reported relationships with Bristol-Myers Squibb, Pierre Fabre, Takeda, Innate Pharma, LEO Pharma, Roche, GlaxoSmithKline, Novartis, and Amgen.
SOURCE: Delyon J et al. Br J Dermatol. 2019 May 11. doi: 10.1111/bjd.18124.
according to a case study.
A 47-year-old woman received pembrolizumab to treat metastatic melanoma and developed immune-related generalized acquired lipodystrophy. The condition has persisted 12 months after she stopped taking pembrolizumab.
Julie Delyon, MD, PhD, of Saint-Louis Hospital in Paris, and colleagues described this case in the British Journal of Dermatology.
The patient was diagnosed with BRAF-mutated, stage IV melanoma with bone and lymph node metastases. She received pembrolizumab at 2 mg/kg every 3 weeks as first-line treatment. She achieved a complete response and was still in remission at last follow-up.
The patient was obese at baseline, with a body mass index of 40 kg/m2, but she did not have diabetes, hepatic steatosis, or dyslipidemia.
Within 2 months of starting pembrolizumab, the patient observed “some major changes to her physical appearance,” according to Dr. Delyon and colleagues. At 10 months, the patient had severe lipodystrophy.
The patient experienced a change in fat distribution – namely, severe peripheral lipoatrophy and an accumulation of trunk fat. Imaging revealed a decrease in subcutaneous adipose tissue, which contrasted with an increase in visceral fat observed from baseline.
The patient also developed “moderate hirsutism, facial and limb atrophy, and prominent forearm and leg muscles and veins,” according to the authors. She had impaired glucose tolerance with insulin resistance, reduced concentrations of leptin and adiponectin, hypertriglyceridemia, a low level of HDL cholesterol, and hepatic steatosis.
Analyses of subcutaneous fat revealed adipose tissue atrophy with edema, lipophages, and CD3+/CD4+ T-cell infiltration of the fat and vessel walls. This suggested that the lipodystrophy had an autoimmune origin, according to the authors.
The patient had no family history of autoimmune disease or lipodystrophy. She tested negative for mutations in 23 genes associated with generalized lipodystrophy.
The patient also tested negative for HIV, antinuclear antibodies, native anti-DNA, and anti-insulin receptor antibodies. There were no signs of panniculitis, and the authors noted that “there were no arguments in favor of hyperthyroidism, Cushing syndrome, or acromegaly.”
In an attempt to reverse the lipodystrophy, the researchers stopped pembrolizumab treatment. The patient was treated for diabetes and hypertriglyceridemia as well. She could not receive corticosteroids because of the risk of severe metabolic complications, and she didn’t receive metreleptin because it wasn’t available. The patient still had lipodystrophy 12 months after stopping pembrolizumab.
Dr. Delyon and colleagues wrote that this case suggests pembrolizumab, and perhaps other anti–programmed death 1 therapies, may cause lipodystrophy with severe metabolic complications. “The long-term side effects of such metabolic adverse events, although rare, are unknown and will probably become a topic of utmost importance, considering the increasing rate of remission following ICIs and their use in the adjuvant setting.”
Two coauthors reported relationships with Merck, which markets pembrolizumab as Keytruda. The authors also reported relationships with Bristol-Myers Squibb, Pierre Fabre, Takeda, Innate Pharma, LEO Pharma, Roche, GlaxoSmithKline, Novartis, and Amgen.
SOURCE: Delyon J et al. Br J Dermatol. 2019 May 11. doi: 10.1111/bjd.18124.
according to a case study.
A 47-year-old woman received pembrolizumab to treat metastatic melanoma and developed immune-related generalized acquired lipodystrophy. The condition has persisted 12 months after she stopped taking pembrolizumab.
Julie Delyon, MD, PhD, of Saint-Louis Hospital in Paris, and colleagues described this case in the British Journal of Dermatology.
The patient was diagnosed with BRAF-mutated, stage IV melanoma with bone and lymph node metastases. She received pembrolizumab at 2 mg/kg every 3 weeks as first-line treatment. She achieved a complete response and was still in remission at last follow-up.
The patient was obese at baseline, with a body mass index of 40 kg/m2, but she did not have diabetes, hepatic steatosis, or dyslipidemia.
Within 2 months of starting pembrolizumab, the patient observed “some major changes to her physical appearance,” according to Dr. Delyon and colleagues. At 10 months, the patient had severe lipodystrophy.
The patient experienced a change in fat distribution – namely, severe peripheral lipoatrophy and an accumulation of trunk fat. Imaging revealed a decrease in subcutaneous adipose tissue, which contrasted with an increase in visceral fat observed from baseline.
The patient also developed “moderate hirsutism, facial and limb atrophy, and prominent forearm and leg muscles and veins,” according to the authors. She had impaired glucose tolerance with insulin resistance, reduced concentrations of leptin and adiponectin, hypertriglyceridemia, a low level of HDL cholesterol, and hepatic steatosis.
Analyses of subcutaneous fat revealed adipose tissue atrophy with edema, lipophages, and CD3+/CD4+ T-cell infiltration of the fat and vessel walls. This suggested that the lipodystrophy had an autoimmune origin, according to the authors.
The patient had no family history of autoimmune disease or lipodystrophy. She tested negative for mutations in 23 genes associated with generalized lipodystrophy.
The patient also tested negative for HIV, antinuclear antibodies, native anti-DNA, and anti-insulin receptor antibodies. There were no signs of panniculitis, and the authors noted that “there were no arguments in favor of hyperthyroidism, Cushing syndrome, or acromegaly.”
In an attempt to reverse the lipodystrophy, the researchers stopped pembrolizumab treatment. The patient was treated for diabetes and hypertriglyceridemia as well. She could not receive corticosteroids because of the risk of severe metabolic complications, and she didn’t receive metreleptin because it wasn’t available. The patient still had lipodystrophy 12 months after stopping pembrolizumab.
Dr. Delyon and colleagues wrote that this case suggests pembrolizumab, and perhaps other anti–programmed death 1 therapies, may cause lipodystrophy with severe metabolic complications. “The long-term side effects of such metabolic adverse events, although rare, are unknown and will probably become a topic of utmost importance, considering the increasing rate of remission following ICIs and their use in the adjuvant setting.”
Two coauthors reported relationships with Merck, which markets pembrolizumab as Keytruda. The authors also reported relationships with Bristol-Myers Squibb, Pierre Fabre, Takeda, Innate Pharma, LEO Pharma, Roche, GlaxoSmithKline, Novartis, and Amgen.
SOURCE: Delyon J et al. Br J Dermatol. 2019 May 11. doi: 10.1111/bjd.18124.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Dabrafenib plus trametinib yields long-term benefit in melanoma patients
Dabrafenib plus trametinib treatment was associated with a 5-year overall survival rate of 34% in patients with melanoma harboring a BRAF V600E or V600K mutation, according to a combined analysis of two trials.
The 5-year progression-free survival rate was 19% in the long-term, pooled analysis of the COMBI-d and COMBI-v trials, which included at total of 563 patients with previously untreated, unresectable or metastatic melanoma who received combined treatment with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib.
Previously reported 5-year progression-free survival rates for patients treated with anti–programmed death-1 checkpoint inhibitors, either nivolumab or pembrolizumab, “appear to be similar” to these results for dabrafenib plus trametinib, investigators said in a report on the analysis appearing in the New England Journal of Medicine.
To date, however, 5-year survival data have not been reported for other BRAF-targeted therapies, according to the investigators, who were led by Caroline Robert, MD, PhD, of Institut Gustave Roussy and Paris-Sud-Paris-Saclay University, Villejuif, France.
“These data will be critical to assess the potential of therapy to exert long-term disease control through analysis of survival plateaus and to understand factors predictive of long-term survival,” Dr. Robert and coauthors wrote in their report.
A total of 211 patients in the COMBI-d trial were randomly allocated to receive the combination of dabrafenib plus trametinib, while in COMBI-v, 352 received this combination therapy, according to investigators.
Notably, the survival curves for dabrafenib plus trametinib appear to plateau starting at 3 years, investigators reported. In a previously published report on pooled COMBI-d and COMBI-v data, the 3-year progression-free survival rate was 23%, and the 3-year overall survival rate was 44%.
In this more recent analysis, progression-free survival rates were 21% at 4 years and 19% at 5 years, while overall survival rates were 37% at 4 years and 34% at 5 years.
“This finding suggests stabilization of rates of progression-free survival and overall survival over time in this population,” Dr. Robert and colleagues wrote.
Survival rates were higher in patients with normal lactate dehydrogenase (LDH) levels at baseline, and they were especially high in those with normal LDH and three or fewer disease sites at baseline, according to the report. Specifically, the reported 5-year rates of progression-free and overall survival were 31% and 55%, respectively.
Other factors associated with prolonged progression-free survival included female sex, older age, better performance status, and BRAF V600E genotype, according to results of a multivariate analysis that investigators said confirmed findings from the previously reported 3-year data.
The study was supported by GlaxoSmithKline and Novartis. Dr. Robert provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
SOURCE: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
Dabrafenib plus trametinib treatment was associated with a 5-year overall survival rate of 34% in patients with melanoma harboring a BRAF V600E or V600K mutation, according to a combined analysis of two trials.
The 5-year progression-free survival rate was 19% in the long-term, pooled analysis of the COMBI-d and COMBI-v trials, which included at total of 563 patients with previously untreated, unresectable or metastatic melanoma who received combined treatment with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib.
Previously reported 5-year progression-free survival rates for patients treated with anti–programmed death-1 checkpoint inhibitors, either nivolumab or pembrolizumab, “appear to be similar” to these results for dabrafenib plus trametinib, investigators said in a report on the analysis appearing in the New England Journal of Medicine.
To date, however, 5-year survival data have not been reported for other BRAF-targeted therapies, according to the investigators, who were led by Caroline Robert, MD, PhD, of Institut Gustave Roussy and Paris-Sud-Paris-Saclay University, Villejuif, France.
“These data will be critical to assess the potential of therapy to exert long-term disease control through analysis of survival plateaus and to understand factors predictive of long-term survival,” Dr. Robert and coauthors wrote in their report.
A total of 211 patients in the COMBI-d trial were randomly allocated to receive the combination of dabrafenib plus trametinib, while in COMBI-v, 352 received this combination therapy, according to investigators.
Notably, the survival curves for dabrafenib plus trametinib appear to plateau starting at 3 years, investigators reported. In a previously published report on pooled COMBI-d and COMBI-v data, the 3-year progression-free survival rate was 23%, and the 3-year overall survival rate was 44%.
In this more recent analysis, progression-free survival rates were 21% at 4 years and 19% at 5 years, while overall survival rates were 37% at 4 years and 34% at 5 years.
“This finding suggests stabilization of rates of progression-free survival and overall survival over time in this population,” Dr. Robert and colleagues wrote.
Survival rates were higher in patients with normal lactate dehydrogenase (LDH) levels at baseline, and they were especially high in those with normal LDH and three or fewer disease sites at baseline, according to the report. Specifically, the reported 5-year rates of progression-free and overall survival were 31% and 55%, respectively.
Other factors associated with prolonged progression-free survival included female sex, older age, better performance status, and BRAF V600E genotype, according to results of a multivariate analysis that investigators said confirmed findings from the previously reported 3-year data.
The study was supported by GlaxoSmithKline and Novartis. Dr. Robert provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
SOURCE: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
Dabrafenib plus trametinib treatment was associated with a 5-year overall survival rate of 34% in patients with melanoma harboring a BRAF V600E or V600K mutation, according to a combined analysis of two trials.
The 5-year progression-free survival rate was 19% in the long-term, pooled analysis of the COMBI-d and COMBI-v trials, which included at total of 563 patients with previously untreated, unresectable or metastatic melanoma who received combined treatment with the BRAF inhibitor dabrafenib and the MEK inhibitor trametinib.
Previously reported 5-year progression-free survival rates for patients treated with anti–programmed death-1 checkpoint inhibitors, either nivolumab or pembrolizumab, “appear to be similar” to these results for dabrafenib plus trametinib, investigators said in a report on the analysis appearing in the New England Journal of Medicine.
To date, however, 5-year survival data have not been reported for other BRAF-targeted therapies, according to the investigators, who were led by Caroline Robert, MD, PhD, of Institut Gustave Roussy and Paris-Sud-Paris-Saclay University, Villejuif, France.
“These data will be critical to assess the potential of therapy to exert long-term disease control through analysis of survival plateaus and to understand factors predictive of long-term survival,” Dr. Robert and coauthors wrote in their report.
A total of 211 patients in the COMBI-d trial were randomly allocated to receive the combination of dabrafenib plus trametinib, while in COMBI-v, 352 received this combination therapy, according to investigators.
Notably, the survival curves for dabrafenib plus trametinib appear to plateau starting at 3 years, investigators reported. In a previously published report on pooled COMBI-d and COMBI-v data, the 3-year progression-free survival rate was 23%, and the 3-year overall survival rate was 44%.
In this more recent analysis, progression-free survival rates were 21% at 4 years and 19% at 5 years, while overall survival rates were 37% at 4 years and 34% at 5 years.
“This finding suggests stabilization of rates of progression-free survival and overall survival over time in this population,” Dr. Robert and colleagues wrote.
Survival rates were higher in patients with normal lactate dehydrogenase (LDH) levels at baseline, and they were especially high in those with normal LDH and three or fewer disease sites at baseline, according to the report. Specifically, the reported 5-year rates of progression-free and overall survival were 31% and 55%, respectively.
Other factors associated with prolonged progression-free survival included female sex, older age, better performance status, and BRAF V600E genotype, according to results of a multivariate analysis that investigators said confirmed findings from the previously reported 3-year data.
The study was supported by GlaxoSmithKline and Novartis. Dr. Robert provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
SOURCE: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: A long-term survival benefit was seen in about a third of patients with metastatic or unresectable melanoma who underwent first-line treatment with dabrafenib and trametinib.
Major finding: The 5-year rates of progression-free survival and overall survival were 19% and 34%, respectively.
Study details: Pooled analysis including 563 patients randomly allocated to the combination treatment in two randomized trials (COMBI-d and COMBI-v).
Disclosures: The study was supported by GlaxoSmithKline and Novartis. The first author provided disclosures related to BMS, Pierre Fabre, Novartis, Amgen, Merck, Roche, MSD, and Sanofi.
Source: Robert C et al. N Engl J Med. 2019 Aug 15. doi: 10.1056/NEJMoa1904059