User login
Tofacitinib Lowered Disease Activity in Ulcerative Colitis
Tofacitinib was associated with greater clinical response and remission rates than was placebo in a double-blind, phase II trial of patients with moderately or severely active ulcerative colitis.
Current treatment of ulcerative colitis with mesalamine, glucocorticoids, azathioprine, and anti–tumor necrosis factor (anti-TNF) agents such as infliximab and adalimumab is not always effective, and may be associated with serious toxic effects (Gastroenterology 2006;130:940-87). Thus, "additional treatments are needed," said Dr. William J. Sandborn, chief of the division of gastroenterology and professor of medicine at the University of California, San Diego and his associates (N. Engl. J. Med. 2012;367:616-24).
Tofacitinib is a selective oral inhibitor of the Janus kinase (JAK) family of enzymes that includes JAK1 and JAK3, which mediate signal-transduction activity for multiple cytokines, including several that are integral to lymphocyte activation, function, and proliferation. Although the importance of the JAK family in the pathogenesis of ulcerative colitis is unclear, tofacitinib has shown efficacy against other immune-mediated conditions, including organ allograft rejection, rheumatoid arthritis, and psoriasis.
The 8-week trial was conducted at 51 centers in 17 countries from January 2009 through September 2010 with funding from Pfizer. Subjects had to be at least 18 years of age, with a confirmed diagnosis of ulcerative colitis of 3 months’ duration or longer, a score of 6-12 on the Mayo risk calculator for ulcerative colitis,and evidence on sigmoidoscopic examination of moderately or severely active disease. In most of the patients, the use of conventional therapy (mesalamine, glucocorticoids, immunosuppressants, or anti-TNF agents used as monotherapy or in some combination) had failed, according to the investigators.
During the study, patients could receive oral mesalamine or oral prednisone at a stable dosage of 30 mg or less per day. However, patients receiving azathioprine, 6-mercaptopurine, and methotrexate discontinued them immediately before initiating therapy with tofacitinib, and patients who had previously received anti-TNF therapy were required to discontinue it for at least 8 weeks before study entry.
Patients were randomly assigned to receive twice-daily oral tofacitinib doses of 0.5 mg, 3 mg, 10 mg, or 15 mg or placebo for 8 weeks, and were then followed for 4 weeks through 12 weeks. Of the 195 patients randomized, 194 received at least one dose of the study drug or placebo and 157 completed the full 8 weeks of treatment. Across all treatment groups, 131 patients (67.5%) received concomitant aminosalicylates, and 85 (43.8%) received concomitant glucocorticoids at some point during the study, they noted.
Clinical response was defined as a decrease from baseline in the total Mayo score (defined as an absolute decrease by at least 3 points and a relative decrease by at least 30%) with an accompanying decrease in the rectal bleeding subscore of at least 1 point or an absolute rectal bleeding subscore of 0 or 1.
The primary end point, clinical response at 8 weeks, occurred in 42% (20) of the patients who received placebo. The response was significantly greater than that seen only among the patients who received the highest doses of tofacitinib, 78% of those receiving 15 mg (P less than .001). For the lower doses, the clinical response rates were not significantly different from those with placebo (61%, 48% and 32%, respectively, for 10 mg, 3 mg and 0.5 mg).
Clinical remission, a secondary end point, was defined as a total Mayo score of 0-2, with no individual subscore exceeding 1. Clinical remission at 8 weeks occurred in 10% (5) of the placebo patients. Here, the difference from placebo with tofacitinib was significant for doses of 3 mg (33%; P = .01), 10 mg (48%; P less than .001), and 15 mg (41%; P less than .001).
Another secondary end point, endoscopic response, was defined as a decrease from baseline in the endoscopy subscore by at least 1.The secondary end point of endoscopic remission was defined as an endoscopy subscore of 0.
An endoscopic response at 8 weeks occurred in 46% with placebo, significantly lower than the responses with tofacitinib doses of 10 mg (67%; P = .07) and 15mg (78%; P = .001). Endoscopic remission at 8 weeks occurred in just 2% of the placebo group, vs. significantly greater proportions of 18% with 3 mg (P = .01), 30% with 10 mg (P less than .001), and 27% with 15 mg (P less than .001).
Rates of overall and serious adverse events, as well as adverse events from infection, were similar among the groups. Two patients receiving 10 mg of tofacitinib twice daily had serious adverse events from infection (a postoperative abscess in one and anal abscess in the other). There was a dose-dependent increase in both LDL and HDL cholesterol concentrations at 8 weeks with tofacitinib, which reversed after discontinuation of the study drug . During the study period, the absolute neutrophil count was less than 1,500 cells per cubic millimeter in three patients receiving tofacitinib, but was not less than 1,000 cells per cubic millimeter in any patient, Dr. Sandborn and his associates reported.
In previous studies of patients with rheumatoid arthritis, tofacitinib has been associated with increases in LDL cholesterol and serum creatinine, and decreases in absolute neutrophil count, whereas increased infection risk has been seen with the 15-mg, twice-daily dose. The small size and short duration of the current trial did not allow for a comprehensive assessment of the safety and tolerability of the drug in patients with ulcerative colitis, they noted.
This study was funded by Pfizer, from whom Dr. Sandborn has received grants and consulting fees. He has also served as a consultant and has additional ties to numerous other companies.
Tofacitinib was associated with greater clinical response and remission rates than was placebo in a double-blind, phase II trial of patients with moderately or severely active ulcerative colitis.
Current treatment of ulcerative colitis with mesalamine, glucocorticoids, azathioprine, and anti–tumor necrosis factor (anti-TNF) agents such as infliximab and adalimumab is not always effective, and may be associated with serious toxic effects (Gastroenterology 2006;130:940-87). Thus, "additional treatments are needed," said Dr. William J. Sandborn, chief of the division of gastroenterology and professor of medicine at the University of California, San Diego and his associates (N. Engl. J. Med. 2012;367:616-24).
Tofacitinib is a selective oral inhibitor of the Janus kinase (JAK) family of enzymes that includes JAK1 and JAK3, which mediate signal-transduction activity for multiple cytokines, including several that are integral to lymphocyte activation, function, and proliferation. Although the importance of the JAK family in the pathogenesis of ulcerative colitis is unclear, tofacitinib has shown efficacy against other immune-mediated conditions, including organ allograft rejection, rheumatoid arthritis, and psoriasis.
The 8-week trial was conducted at 51 centers in 17 countries from January 2009 through September 2010 with funding from Pfizer. Subjects had to be at least 18 years of age, with a confirmed diagnosis of ulcerative colitis of 3 months’ duration or longer, a score of 6-12 on the Mayo risk calculator for ulcerative colitis,and evidence on sigmoidoscopic examination of moderately or severely active disease. In most of the patients, the use of conventional therapy (mesalamine, glucocorticoids, immunosuppressants, or anti-TNF agents used as monotherapy or in some combination) had failed, according to the investigators.
During the study, patients could receive oral mesalamine or oral prednisone at a stable dosage of 30 mg or less per day. However, patients receiving azathioprine, 6-mercaptopurine, and methotrexate discontinued them immediately before initiating therapy with tofacitinib, and patients who had previously received anti-TNF therapy were required to discontinue it for at least 8 weeks before study entry.
Patients were randomly assigned to receive twice-daily oral tofacitinib doses of 0.5 mg, 3 mg, 10 mg, or 15 mg or placebo for 8 weeks, and were then followed for 4 weeks through 12 weeks. Of the 195 patients randomized, 194 received at least one dose of the study drug or placebo and 157 completed the full 8 weeks of treatment. Across all treatment groups, 131 patients (67.5%) received concomitant aminosalicylates, and 85 (43.8%) received concomitant glucocorticoids at some point during the study, they noted.
Clinical response was defined as a decrease from baseline in the total Mayo score (defined as an absolute decrease by at least 3 points and a relative decrease by at least 30%) with an accompanying decrease in the rectal bleeding subscore of at least 1 point or an absolute rectal bleeding subscore of 0 or 1.
The primary end point, clinical response at 8 weeks, occurred in 42% (20) of the patients who received placebo. The response was significantly greater than that seen only among the patients who received the highest doses of tofacitinib, 78% of those receiving 15 mg (P less than .001). For the lower doses, the clinical response rates were not significantly different from those with placebo (61%, 48% and 32%, respectively, for 10 mg, 3 mg and 0.5 mg).
Clinical remission, a secondary end point, was defined as a total Mayo score of 0-2, with no individual subscore exceeding 1. Clinical remission at 8 weeks occurred in 10% (5) of the placebo patients. Here, the difference from placebo with tofacitinib was significant for doses of 3 mg (33%; P = .01), 10 mg (48%; P less than .001), and 15 mg (41%; P less than .001).
Another secondary end point, endoscopic response, was defined as a decrease from baseline in the endoscopy subscore by at least 1.The secondary end point of endoscopic remission was defined as an endoscopy subscore of 0.
An endoscopic response at 8 weeks occurred in 46% with placebo, significantly lower than the responses with tofacitinib doses of 10 mg (67%; P = .07) and 15mg (78%; P = .001). Endoscopic remission at 8 weeks occurred in just 2% of the placebo group, vs. significantly greater proportions of 18% with 3 mg (P = .01), 30% with 10 mg (P less than .001), and 27% with 15 mg (P less than .001).
Rates of overall and serious adverse events, as well as adverse events from infection, were similar among the groups. Two patients receiving 10 mg of tofacitinib twice daily had serious adverse events from infection (a postoperative abscess in one and anal abscess in the other). There was a dose-dependent increase in both LDL and HDL cholesterol concentrations at 8 weeks with tofacitinib, which reversed after discontinuation of the study drug . During the study period, the absolute neutrophil count was less than 1,500 cells per cubic millimeter in three patients receiving tofacitinib, but was not less than 1,000 cells per cubic millimeter in any patient, Dr. Sandborn and his associates reported.
In previous studies of patients with rheumatoid arthritis, tofacitinib has been associated with increases in LDL cholesterol and serum creatinine, and decreases in absolute neutrophil count, whereas increased infection risk has been seen with the 15-mg, twice-daily dose. The small size and short duration of the current trial did not allow for a comprehensive assessment of the safety and tolerability of the drug in patients with ulcerative colitis, they noted.
This study was funded by Pfizer, from whom Dr. Sandborn has received grants and consulting fees. He has also served as a consultant and has additional ties to numerous other companies.
Tofacitinib was associated with greater clinical response and remission rates than was placebo in a double-blind, phase II trial of patients with moderately or severely active ulcerative colitis.
Current treatment of ulcerative colitis with mesalamine, glucocorticoids, azathioprine, and anti–tumor necrosis factor (anti-TNF) agents such as infliximab and adalimumab is not always effective, and may be associated with serious toxic effects (Gastroenterology 2006;130:940-87). Thus, "additional treatments are needed," said Dr. William J. Sandborn, chief of the division of gastroenterology and professor of medicine at the University of California, San Diego and his associates (N. Engl. J. Med. 2012;367:616-24).
Tofacitinib is a selective oral inhibitor of the Janus kinase (JAK) family of enzymes that includes JAK1 and JAK3, which mediate signal-transduction activity for multiple cytokines, including several that are integral to lymphocyte activation, function, and proliferation. Although the importance of the JAK family in the pathogenesis of ulcerative colitis is unclear, tofacitinib has shown efficacy against other immune-mediated conditions, including organ allograft rejection, rheumatoid arthritis, and psoriasis.
The 8-week trial was conducted at 51 centers in 17 countries from January 2009 through September 2010 with funding from Pfizer. Subjects had to be at least 18 years of age, with a confirmed diagnosis of ulcerative colitis of 3 months’ duration or longer, a score of 6-12 on the Mayo risk calculator for ulcerative colitis,and evidence on sigmoidoscopic examination of moderately or severely active disease. In most of the patients, the use of conventional therapy (mesalamine, glucocorticoids, immunosuppressants, or anti-TNF agents used as monotherapy or in some combination) had failed, according to the investigators.
During the study, patients could receive oral mesalamine or oral prednisone at a stable dosage of 30 mg or less per day. However, patients receiving azathioprine, 6-mercaptopurine, and methotrexate discontinued them immediately before initiating therapy with tofacitinib, and patients who had previously received anti-TNF therapy were required to discontinue it for at least 8 weeks before study entry.
Patients were randomly assigned to receive twice-daily oral tofacitinib doses of 0.5 mg, 3 mg, 10 mg, or 15 mg or placebo for 8 weeks, and were then followed for 4 weeks through 12 weeks. Of the 195 patients randomized, 194 received at least one dose of the study drug or placebo and 157 completed the full 8 weeks of treatment. Across all treatment groups, 131 patients (67.5%) received concomitant aminosalicylates, and 85 (43.8%) received concomitant glucocorticoids at some point during the study, they noted.
Clinical response was defined as a decrease from baseline in the total Mayo score (defined as an absolute decrease by at least 3 points and a relative decrease by at least 30%) with an accompanying decrease in the rectal bleeding subscore of at least 1 point or an absolute rectal bleeding subscore of 0 or 1.
The primary end point, clinical response at 8 weeks, occurred in 42% (20) of the patients who received placebo. The response was significantly greater than that seen only among the patients who received the highest doses of tofacitinib, 78% of those receiving 15 mg (P less than .001). For the lower doses, the clinical response rates were not significantly different from those with placebo (61%, 48% and 32%, respectively, for 10 mg, 3 mg and 0.5 mg).
Clinical remission, a secondary end point, was defined as a total Mayo score of 0-2, with no individual subscore exceeding 1. Clinical remission at 8 weeks occurred in 10% (5) of the placebo patients. Here, the difference from placebo with tofacitinib was significant for doses of 3 mg (33%; P = .01), 10 mg (48%; P less than .001), and 15 mg (41%; P less than .001).
Another secondary end point, endoscopic response, was defined as a decrease from baseline in the endoscopy subscore by at least 1.The secondary end point of endoscopic remission was defined as an endoscopy subscore of 0.
An endoscopic response at 8 weeks occurred in 46% with placebo, significantly lower than the responses with tofacitinib doses of 10 mg (67%; P = .07) and 15mg (78%; P = .001). Endoscopic remission at 8 weeks occurred in just 2% of the placebo group, vs. significantly greater proportions of 18% with 3 mg (P = .01), 30% with 10 mg (P less than .001), and 27% with 15 mg (P less than .001).
Rates of overall and serious adverse events, as well as adverse events from infection, were similar among the groups. Two patients receiving 10 mg of tofacitinib twice daily had serious adverse events from infection (a postoperative abscess in one and anal abscess in the other). There was a dose-dependent increase in both LDL and HDL cholesterol concentrations at 8 weeks with tofacitinib, which reversed after discontinuation of the study drug . During the study period, the absolute neutrophil count was less than 1,500 cells per cubic millimeter in three patients receiving tofacitinib, but was not less than 1,000 cells per cubic millimeter in any patient, Dr. Sandborn and his associates reported.
In previous studies of patients with rheumatoid arthritis, tofacitinib has been associated with increases in LDL cholesterol and serum creatinine, and decreases in absolute neutrophil count, whereas increased infection risk has been seen with the 15-mg, twice-daily dose. The small size and short duration of the current trial did not allow for a comprehensive assessment of the safety and tolerability of the drug in patients with ulcerative colitis, they noted.
This study was funded by Pfizer, from whom Dr. Sandborn has received grants and consulting fees. He has also served as a consultant and has additional ties to numerous other companies.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The primary end point, clinical response at 8 weeks, occurred in 42% (20) of the patients who received placebo. The response was significantly greater than that among the patients who received the highest doses of tofacitinib, 78% of those receiving 15 mg (P less than .001).
Data Source: The findings come from a randomized, double-blind phase II trial of 194 patients with moderately or severely active ulcerative colitis, most of whom had failed current therapies.
Disclosures: The study was funded by Pfizer, from whom Dr. Sandborn has received grants and consulting fees. He has also served as a consultant and has additional ties to numerous other companies.
Most IBD Patients Don't Meet Biologics Trial Criteria
Participants in randomized controlled trials of biologics for inflammatory bowel disease do not adequately represent real-world patients because of stringent inclusion criteria for the trials, reported Dr. Christina Ha and her colleagues in the September issue of Clinical Gastroenterology and Hepatology.
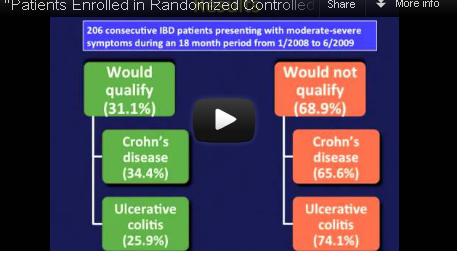
Indeed, among 125 Crohn’s disease (CD) patients seen in routine clinical practice at a tertiary care center, only 43 – just 34% – would have qualified for enrollment in at least one of seven randomized controlled trials (RCTs) for biologics, calling into question the generalizability of these trials’ results. The corresponding percentage for ulcerative colitis (UC) patients was only 25%.
Video Source: American Gastroenterological Association
Dr. Ha, of the division of gastroenterology at Johns Hopkins University, Baltimore, and her colleagues reviewed the medical records of 206 patients with moderate to severe CD or UC presenting to the Mount Sinai Medical Center in New York for adjustment of therapy during 2008-2009.
The researchers then looked at seven randomized controlled trials of biologics in CD as well as two trials of biologics in UC, and applied their inclusion and exclusion criteria to her real-world study population (Clin. Gastroenterol. Hepatol. 2012 September [doi: 10.1016/j.cgh.2012.02.004]).
Among the Crohn’s patients, "trial eligibility for the RCTs for infliximab, adalimumab, certolizumab pegol, and natalizumab ranged from 8% for the SONIC [Study of Biologic and Immunomodulator Naïve Patients in Crohn’s Disease] trial to 27% for the CHARM [Crohn’s Trial of the Fully Human Antibody Adalimumab for Remission Maintenance] and PRECISE [Pegylated Antibody Fragment Evaluation in Crohn’s Disease] trials," wrote the investigators.
Similarly, among the 81 UC patients, only 21 (25%) would have qualified for enrollment into the ACT (Active Ulcerative Colitis Trial) 1 or 2 trials.
Among the Crohn’s patients, the most common reasons for ineligibility were symptomatic strictures or abscesses (n = 51, 62.2%), recent exposure or prior nonresponse to anti–tumor necrosis factor drugs (n = 42, 51.2%), use of high-dose steroids (n = 15, 18.3%), and comorbid cardiovascular or pulmonary disease and malignancies (n = 21, 25.6%).
"The most common reason for trial ineligibility for UC was current rectal therapy usage (n = 34, 56.7%)," wrote the authors. Other reasons included steroid naivety (n = 27, 45.0%); new diagnoses of UC (n = 10, 16.7%); or need for colectomy due to age, comorbidity, or concomitant dysplasia found during colonoscopy (n = 9, 15.0%).
Finally, the authors assessed the outcomes of patients who would not have qualified for the biologics trials but initiated biologic therapy nevertheless. In the CD cohort, "ultimately, almost 50% of these ‘trial ineligible’ patients underwent surgery, either as a primary therapy or due to inadequate response to biologics or immunomodulators," wrote the authors. In the UC group, roughly one-quarter of would-be ineligible patients underwent colectomy at 4-12 weeks.
The authors conceded that if patients had been studied longitudinally, "with the inclusion and exclusion criteria applied at the time patients were first being considered for immunomodulators or biologics or at the time of diagnosis, a larger percentage of patients may have been eligible for trial participation."
Nevertheless, Dr. Ha and her colleagues concluded, "pragmatic trials of the major IBD therapeutics would not only serve to validate RCT findings, but provide additional insight regarding medication safety across a broader patient population."
Several of the authors disclosed financial relationships with pharmaceutical companies, including the makers of biologics. The authors stated that this study received no grant support.
Participants in randomized controlled trials of biologics for inflammatory bowel disease do not adequately represent real-world patients because of stringent inclusion criteria for the trials, reported Dr. Christina Ha and her colleagues in the September issue of Clinical Gastroenterology and Hepatology.
Indeed, among 125 Crohn’s disease (CD) patients seen in routine clinical practice at a tertiary care center, only 43 – just 34% – would have qualified for enrollment in at least one of seven randomized controlled trials (RCTs) for biologics, calling into question the generalizability of these trials’ results. The corresponding percentage for ulcerative colitis (UC) patients was only 25%.
Video Source: American Gastroenterological Association
Dr. Ha, of the division of gastroenterology at Johns Hopkins University, Baltimore, and her colleagues reviewed the medical records of 206 patients with moderate to severe CD or UC presenting to the Mount Sinai Medical Center in New York for adjustment of therapy during 2008-2009.
The researchers then looked at seven randomized controlled trials of biologics in CD as well as two trials of biologics in UC, and applied their inclusion and exclusion criteria to her real-world study population (Clin. Gastroenterol. Hepatol. 2012 September [doi: 10.1016/j.cgh.2012.02.004]).
Among the Crohn’s patients, "trial eligibility for the RCTs for infliximab, adalimumab, certolizumab pegol, and natalizumab ranged from 8% for the SONIC [Study of Biologic and Immunomodulator Naïve Patients in Crohn’s Disease] trial to 27% for the CHARM [Crohn’s Trial of the Fully Human Antibody Adalimumab for Remission Maintenance] and PRECISE [Pegylated Antibody Fragment Evaluation in Crohn’s Disease] trials," wrote the investigators.
Similarly, among the 81 UC patients, only 21 (25%) would have qualified for enrollment into the ACT (Active Ulcerative Colitis Trial) 1 or 2 trials.
Among the Crohn’s patients, the most common reasons for ineligibility were symptomatic strictures or abscesses (n = 51, 62.2%), recent exposure or prior nonresponse to anti–tumor necrosis factor drugs (n = 42, 51.2%), use of high-dose steroids (n = 15, 18.3%), and comorbid cardiovascular or pulmonary disease and malignancies (n = 21, 25.6%).
"The most common reason for trial ineligibility for UC was current rectal therapy usage (n = 34, 56.7%)," wrote the authors. Other reasons included steroid naivety (n = 27, 45.0%); new diagnoses of UC (n = 10, 16.7%); or need for colectomy due to age, comorbidity, or concomitant dysplasia found during colonoscopy (n = 9, 15.0%).
Finally, the authors assessed the outcomes of patients who would not have qualified for the biologics trials but initiated biologic therapy nevertheless. In the CD cohort, "ultimately, almost 50% of these ‘trial ineligible’ patients underwent surgery, either as a primary therapy or due to inadequate response to biologics or immunomodulators," wrote the authors. In the UC group, roughly one-quarter of would-be ineligible patients underwent colectomy at 4-12 weeks.
The authors conceded that if patients had been studied longitudinally, "with the inclusion and exclusion criteria applied at the time patients were first being considered for immunomodulators or biologics or at the time of diagnosis, a larger percentage of patients may have been eligible for trial participation."
Nevertheless, Dr. Ha and her colleagues concluded, "pragmatic trials of the major IBD therapeutics would not only serve to validate RCT findings, but provide additional insight regarding medication safety across a broader patient population."
Several of the authors disclosed financial relationships with pharmaceutical companies, including the makers of biologics. The authors stated that this study received no grant support.
Participants in randomized controlled trials of biologics for inflammatory bowel disease do not adequately represent real-world patients because of stringent inclusion criteria for the trials, reported Dr. Christina Ha and her colleagues in the September issue of Clinical Gastroenterology and Hepatology.
Indeed, among 125 Crohn’s disease (CD) patients seen in routine clinical practice at a tertiary care center, only 43 – just 34% – would have qualified for enrollment in at least one of seven randomized controlled trials (RCTs) for biologics, calling into question the generalizability of these trials’ results. The corresponding percentage for ulcerative colitis (UC) patients was only 25%.
Video Source: American Gastroenterological Association
Dr. Ha, of the division of gastroenterology at Johns Hopkins University, Baltimore, and her colleagues reviewed the medical records of 206 patients with moderate to severe CD or UC presenting to the Mount Sinai Medical Center in New York for adjustment of therapy during 2008-2009.
The researchers then looked at seven randomized controlled trials of biologics in CD as well as two trials of biologics in UC, and applied their inclusion and exclusion criteria to her real-world study population (Clin. Gastroenterol. Hepatol. 2012 September [doi: 10.1016/j.cgh.2012.02.004]).
Among the Crohn’s patients, "trial eligibility for the RCTs for infliximab, adalimumab, certolizumab pegol, and natalizumab ranged from 8% for the SONIC [Study of Biologic and Immunomodulator Naïve Patients in Crohn’s Disease] trial to 27% for the CHARM [Crohn’s Trial of the Fully Human Antibody Adalimumab for Remission Maintenance] and PRECISE [Pegylated Antibody Fragment Evaluation in Crohn’s Disease] trials," wrote the investigators.
Similarly, among the 81 UC patients, only 21 (25%) would have qualified for enrollment into the ACT (Active Ulcerative Colitis Trial) 1 or 2 trials.
Among the Crohn’s patients, the most common reasons for ineligibility were symptomatic strictures or abscesses (n = 51, 62.2%), recent exposure or prior nonresponse to anti–tumor necrosis factor drugs (n = 42, 51.2%), use of high-dose steroids (n = 15, 18.3%), and comorbid cardiovascular or pulmonary disease and malignancies (n = 21, 25.6%).
"The most common reason for trial ineligibility for UC was current rectal therapy usage (n = 34, 56.7%)," wrote the authors. Other reasons included steroid naivety (n = 27, 45.0%); new diagnoses of UC (n = 10, 16.7%); or need for colectomy due to age, comorbidity, or concomitant dysplasia found during colonoscopy (n = 9, 15.0%).
Finally, the authors assessed the outcomes of patients who would not have qualified for the biologics trials but initiated biologic therapy nevertheless. In the CD cohort, "ultimately, almost 50% of these ‘trial ineligible’ patients underwent surgery, either as a primary therapy or due to inadequate response to biologics or immunomodulators," wrote the authors. In the UC group, roughly one-quarter of would-be ineligible patients underwent colectomy at 4-12 weeks.
The authors conceded that if patients had been studied longitudinally, "with the inclusion and exclusion criteria applied at the time patients were first being considered for immunomodulators or biologics or at the time of diagnosis, a larger percentage of patients may have been eligible for trial participation."
Nevertheless, Dr. Ha and her colleagues concluded, "pragmatic trials of the major IBD therapeutics would not only serve to validate RCT findings, but provide additional insight regarding medication safety across a broader patient population."
Several of the authors disclosed financial relationships with pharmaceutical companies, including the makers of biologics. The authors stated that this study received no grant support.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Price Break Ahead for Fidaxomicin for C. Diff. Diarrhea
Hospitals will soon get a break on the price of fidaxomicin tablets for treatment of Clostridium difficile–associated diarrhea, thanks to a new add-on payment from Medicare.
Officials at the Centers for Medicare and Medicaid Services granted a new technology add-on payment for fidaxomicin tablets (Dificid) administered in the hospital. The new payment, which can be as high as $868 for a full course of the macrolide antibacterial drug, will begin on Oct. 1. The add-on payment is in addition to the standard Medicare payment for the treatment of C. difficile–associated diarrhea (CDAD).
The CMS included details about the add-on payment in the final regulation for the Inpatient Prospective Payment System.
Medicare typically only provides the add-on payment for new technologies associated with procedures, such grafts or stents. But after hearing about the clinical performance of the drug, Medicare officials agreed to grant the extra payment to hospitals. In the final rule, CMS officials wrote that the oral antibiotic has the potential "to decrease hospitalizations and physician office visits, and reduce the recurrence of CDAD, as well as to improve the quality of life for patients who have been diagnosed with CDAD."
The CMS provides add-on payments to hospitals to help subsidize the cost of treatments that are new and costly but that potentially offer a significant clinical benefit for Medicare beneficiaries. The payments are offered in addition to the standard DRG (diagnosis-related group) payment and can be up to 50% of the cost of the treatment for 2-3 years.
The CMS will issue guidance in the future on how hospitals can code to receive the add-on payment for Dificid.
Hospitals will soon get a break on the price of fidaxomicin tablets for treatment of Clostridium difficile–associated diarrhea, thanks to a new add-on payment from Medicare.
Officials at the Centers for Medicare and Medicaid Services granted a new technology add-on payment for fidaxomicin tablets (Dificid) administered in the hospital. The new payment, which can be as high as $868 for a full course of the macrolide antibacterial drug, will begin on Oct. 1. The add-on payment is in addition to the standard Medicare payment for the treatment of C. difficile–associated diarrhea (CDAD).
The CMS included details about the add-on payment in the final regulation for the Inpatient Prospective Payment System.
Medicare typically only provides the add-on payment for new technologies associated with procedures, such grafts or stents. But after hearing about the clinical performance of the drug, Medicare officials agreed to grant the extra payment to hospitals. In the final rule, CMS officials wrote that the oral antibiotic has the potential "to decrease hospitalizations and physician office visits, and reduce the recurrence of CDAD, as well as to improve the quality of life for patients who have been diagnosed with CDAD."
The CMS provides add-on payments to hospitals to help subsidize the cost of treatments that are new and costly but that potentially offer a significant clinical benefit for Medicare beneficiaries. The payments are offered in addition to the standard DRG (diagnosis-related group) payment and can be up to 50% of the cost of the treatment for 2-3 years.
The CMS will issue guidance in the future on how hospitals can code to receive the add-on payment for Dificid.
Hospitals will soon get a break on the price of fidaxomicin tablets for treatment of Clostridium difficile–associated diarrhea, thanks to a new add-on payment from Medicare.
Officials at the Centers for Medicare and Medicaid Services granted a new technology add-on payment for fidaxomicin tablets (Dificid) administered in the hospital. The new payment, which can be as high as $868 for a full course of the macrolide antibacterial drug, will begin on Oct. 1. The add-on payment is in addition to the standard Medicare payment for the treatment of C. difficile–associated diarrhea (CDAD).
The CMS included details about the add-on payment in the final regulation for the Inpatient Prospective Payment System.
Medicare typically only provides the add-on payment for new technologies associated with procedures, such grafts or stents. But after hearing about the clinical performance of the drug, Medicare officials agreed to grant the extra payment to hospitals. In the final rule, CMS officials wrote that the oral antibiotic has the potential "to decrease hospitalizations and physician office visits, and reduce the recurrence of CDAD, as well as to improve the quality of life for patients who have been diagnosed with CDAD."
The CMS provides add-on payments to hospitals to help subsidize the cost of treatments that are new and costly but that potentially offer a significant clinical benefit for Medicare beneficiaries. The payments are offered in addition to the standard DRG (diagnosis-related group) payment and can be up to 50% of the cost of the treatment for 2-3 years.
The CMS will issue guidance in the future on how hospitals can code to receive the add-on payment for Dificid.
Transfusion Rates Vary Widely at Academic Hospitals
Wide variations in perioperative blood transfusion rates among patients undergoing major noncardiac procedures across U.S. hospitals highlight the need to further investigate evidence-based "transfusion triggers" in this population of surgical patients, according to a study published ahead of print in Annals of Surgery.
"In light of the increased risk of mortality and major complications associated with blood transfusion, the extensive variability in hospital transfusion practice in noncardiac surgery may represent an important opportunity to improve surgical outcomes," wrote Feng Qian, Ph.D., of the University of Rochester (N.Y.), and associates.
The researchers used the University HealthSystem Consortium hospital database to compare transfusion rates of allogeneic red blood cells, fresh frozen plasma, and platelets in patients undergoing elective primary total hip replacement (54,405 patients), colectomy (21,334), or pancreaticoduodenectomy (7,929) at 77 hospitals between June 2006 and September 2010. Most of the hospitals were teaching hospitals with at least 500 beds.
Transfusion rates varied widely before and after adjustment for comorbidities and other patient risk factors. Patients who were treated in hospitals with high rates of transfusions were about twice as likely to receive a blood transfusion as were patients at hospitals with average transfusion rates (Ann. Surg. 2012 July 13[doi:10.1097/SLA.0b013e31825ffc37]).
In hospitals where the transfusion rate for one procedure was high, transfusion rates also tended to be high for the other two procedures. There was some evidence indicating that a higher volume of surgical cases was associated with lower transfusion rates.
After adjusting for patient risk factors, the authors determined that transfusion rates for the different blood components among those undergoing a total hip replacement ranged from 1.3% to almost 75% (red blood cells), from 0.1% to 7.7% (fresh frozen plasma), and from 0.1% to 2% (platelets). Among colectomy patients, transfusion rates ranged from 1.9% to 47.8% (RBCs), from 1.4% to 17.7% (fresh frozen plasma), and from 1.3% to 6.2% (platelets). Among those undergoing a pancreaticoduodenectomy, the rates ranged from 3% to 78.6% (RBCs), from 1% to 47% (fresh frozen plasma), and from 1.4% to 12.6% (platelets).
The variability, the authors said, "reflects, in part, the complexity of the medical decision-making process underlying transfusion therapy." Because the data included patients from 90% of academic medical centers in the United States, the results provide "a broad and contemporary picture of transfusion practices in academic surgical centers" and "reflect transfusion practices that are being taught to the next generation of academic and private-practice clinicians during residency training," they noted.
To the best of their knowledge, the authors said, there are no large randomized studies that have compared liberal and restrictive transfusion strategies in noncardiac surgery patients, and they believe that such trials are "urgently needed to better define evidence-based transfusion triggers for patients undergoing noncardiac surgery."
The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester. No disclosures were reported by the authors.
Wide variations in perioperative blood transfusion rates among patients undergoing major noncardiac procedures across U.S. hospitals highlight the need to further investigate evidence-based "transfusion triggers" in this population of surgical patients, according to a study published ahead of print in Annals of Surgery.
"In light of the increased risk of mortality and major complications associated with blood transfusion, the extensive variability in hospital transfusion practice in noncardiac surgery may represent an important opportunity to improve surgical outcomes," wrote Feng Qian, Ph.D., of the University of Rochester (N.Y.), and associates.
The researchers used the University HealthSystem Consortium hospital database to compare transfusion rates of allogeneic red blood cells, fresh frozen plasma, and platelets in patients undergoing elective primary total hip replacement (54,405 patients), colectomy (21,334), or pancreaticoduodenectomy (7,929) at 77 hospitals between June 2006 and September 2010. Most of the hospitals were teaching hospitals with at least 500 beds.
Transfusion rates varied widely before and after adjustment for comorbidities and other patient risk factors. Patients who were treated in hospitals with high rates of transfusions were about twice as likely to receive a blood transfusion as were patients at hospitals with average transfusion rates (Ann. Surg. 2012 July 13[doi:10.1097/SLA.0b013e31825ffc37]).
In hospitals where the transfusion rate for one procedure was high, transfusion rates also tended to be high for the other two procedures. There was some evidence indicating that a higher volume of surgical cases was associated with lower transfusion rates.
After adjusting for patient risk factors, the authors determined that transfusion rates for the different blood components among those undergoing a total hip replacement ranged from 1.3% to almost 75% (red blood cells), from 0.1% to 7.7% (fresh frozen plasma), and from 0.1% to 2% (platelets). Among colectomy patients, transfusion rates ranged from 1.9% to 47.8% (RBCs), from 1.4% to 17.7% (fresh frozen plasma), and from 1.3% to 6.2% (platelets). Among those undergoing a pancreaticoduodenectomy, the rates ranged from 3% to 78.6% (RBCs), from 1% to 47% (fresh frozen plasma), and from 1.4% to 12.6% (platelets).
The variability, the authors said, "reflects, in part, the complexity of the medical decision-making process underlying transfusion therapy." Because the data included patients from 90% of academic medical centers in the United States, the results provide "a broad and contemporary picture of transfusion practices in academic surgical centers" and "reflect transfusion practices that are being taught to the next generation of academic and private-practice clinicians during residency training," they noted.
To the best of their knowledge, the authors said, there are no large randomized studies that have compared liberal and restrictive transfusion strategies in noncardiac surgery patients, and they believe that such trials are "urgently needed to better define evidence-based transfusion triggers for patients undergoing noncardiac surgery."
The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester. No disclosures were reported by the authors.
Wide variations in perioperative blood transfusion rates among patients undergoing major noncardiac procedures across U.S. hospitals highlight the need to further investigate evidence-based "transfusion triggers" in this population of surgical patients, according to a study published ahead of print in Annals of Surgery.
"In light of the increased risk of mortality and major complications associated with blood transfusion, the extensive variability in hospital transfusion practice in noncardiac surgery may represent an important opportunity to improve surgical outcomes," wrote Feng Qian, Ph.D., of the University of Rochester (N.Y.), and associates.
The researchers used the University HealthSystem Consortium hospital database to compare transfusion rates of allogeneic red blood cells, fresh frozen plasma, and platelets in patients undergoing elective primary total hip replacement (54,405 patients), colectomy (21,334), or pancreaticoduodenectomy (7,929) at 77 hospitals between June 2006 and September 2010. Most of the hospitals were teaching hospitals with at least 500 beds.
Transfusion rates varied widely before and after adjustment for comorbidities and other patient risk factors. Patients who were treated in hospitals with high rates of transfusions were about twice as likely to receive a blood transfusion as were patients at hospitals with average transfusion rates (Ann. Surg. 2012 July 13[doi:10.1097/SLA.0b013e31825ffc37]).
In hospitals where the transfusion rate for one procedure was high, transfusion rates also tended to be high for the other two procedures. There was some evidence indicating that a higher volume of surgical cases was associated with lower transfusion rates.
After adjusting for patient risk factors, the authors determined that transfusion rates for the different blood components among those undergoing a total hip replacement ranged from 1.3% to almost 75% (red blood cells), from 0.1% to 7.7% (fresh frozen plasma), and from 0.1% to 2% (platelets). Among colectomy patients, transfusion rates ranged from 1.9% to 47.8% (RBCs), from 1.4% to 17.7% (fresh frozen plasma), and from 1.3% to 6.2% (platelets). Among those undergoing a pancreaticoduodenectomy, the rates ranged from 3% to 78.6% (RBCs), from 1% to 47% (fresh frozen plasma), and from 1.4% to 12.6% (platelets).
The variability, the authors said, "reflects, in part, the complexity of the medical decision-making process underlying transfusion therapy." Because the data included patients from 90% of academic medical centers in the United States, the results provide "a broad and contemporary picture of transfusion practices in academic surgical centers" and "reflect transfusion practices that are being taught to the next generation of academic and private-practice clinicians during residency training," they noted.
To the best of their knowledge, the authors said, there are no large randomized studies that have compared liberal and restrictive transfusion strategies in noncardiac surgery patients, and they believe that such trials are "urgently needed to better define evidence-based transfusion triggers for patients undergoing noncardiac surgery."
The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester. No disclosures were reported by the authors.
FROM THE ANNALS OF SURGERY
Major Finding: Transfusion rates of red blood cells, fresh frozen plasma, and platelets among patients undergoing noncardiac procedures varied widely across different U.S. academic-affiliated hospitals.
Data Source: Data from a national database of academic medical centers were used to compare transfusions in patients undergoing one of three elective noncardiac surgical procedures at 77 academic hospitals between June 2006 and September 2010.
Disclosures: The study was supported by a grant from the Agency for Healthcare and Quality Research and funding from the department of anesthesiology at the University of Rochester (N.Y.). The authors reported no disclosures.
'Weekend Effect' Seen for Diverticulitis Procedures
Patients who were admitted for emergency surgery on a weekend to treat left-sided diverticulitis experience more short-term complications and are markedly more likely to undergo a Hartmann procedure than are those admitted on weekdays, according to results from a large population-based study.
Longer hospital stays, significantly higher treatment costs, and higher rates of reoperations were also associated with weekend admission. However, no differences in mortality were observed between the patient groups.
Previous studies have shown worse outcomes for patients with gastrointestinal hemorrhage, kidney injury, myocardial infarction, pulmonary embolism, and intracerebral hemorrhage when they were admitted on weekends. Although the current study, led by Dr. Mathias Worni of Duke University Medical Center in Durham, N.C., and Bern (Switzerland) University Hospital, was not designed to isolate the cause of the "weekend effect" for left-sided diverticulitis patients, the authors noted that hospital staffing tends to be reduced on weekends – especially among specialists such as colorectal surgeons.
Dr. Worni and his colleagues looked at records from the Nationwide Inpatient Sample between January 2002 and December 2008. Of the 31,832 patients who were treated surgically for left-sided diverticulitis, 7,066 (22.2%) were admitted on weekends and 24,766 (77.8%) on weekdays. Patients’ mean age was 60.8 years, and more than half were women.
Among patients who were admitted on a Saturday or Sunday, a Hartmann procedure was performed on 64.8% (n = 4,580), compared with only 53.9% (n = 13,351) for those admitted on a weekday (Arch. Surg. 2012;147:649-55). The Hartmann procedure – which involves formation of a colostomy – has long been the standard surgery for people presenting with left-sided diverticulitis, but is associated with long-term complications and a low rate of reversals.
Primary anastomosis, in which colostomy is avoided, is increasingly preferred, but only 35.2% of patients who were admitted on weekends underwent primary anastomosis, compared with 46.1% of patients admitted on weekdays.
The investigators found that patients admitted on weekends had significantly higher risk for any postoperative complication (odds ratio, 1.10; P = .005), compared with patients admitted on weekdays. Risk of reoperation was also higher among weekend admissions (OR, 1.50; P less than .001).
Furthermore, median total hospital charges were $3,734 higher among patients treated on weekends, and the median length of hospital stay was 0.5 days longer (P less than .001). The authors observed that these findings should motivate improvements in the quality of weekend care.
"Physicians working on weekends are thought to be less experienced than teams working during the week," they wrote. Experienced and specialized colorectal surgeons have been shown to perform more primary anastomoses, compared with trainees or general surgeons (Arch. Surg. 2010;145:79-86; Dis. Colon Rectum 2003;46:1461-8).
Limitations of the study include the fact that it did not capture long-term outcomes or severity of disease at presentation. The latter could be of potential importance: "Some patients, especially those with milder symptoms, may prefer weekend or weekday admission and may time their admission accordingly," the investigators noted.
In an invited critique that accompanied the article, Dr. Juerg Metzger, a surgeon at Lucerne (Switzerland) Cantonal Hospital, wrote that a disparity in experience among weekday and weekend surgical staff likely accounted for the higher rate of Hartmann procedures and complications following weekend admissions.
"Work-hour restrictions do not seem to have a negative influence on mortality and morbidity in surgical patients," Dr. Metzger wrote. "However, reduced experience owing to restricted working hours may negatively influence the practical skills of younger surgeons, resulting in more limited surgery [for example, a Hartmann procedure being performed instead of a primary anastomosis] and an increase in complications related to that surgery."
In the end, Dr. Metzger wrote, "quality is expensive, and our society has to decide if it is desirable and necessary to have the best surgical quality available all the time, especially when considering that health care costs will dramatically increase. It would be relevant to analyze additional large databases, asking similar questions about the outcomes of other common diseases [for example, appendicitis, cholecystitis, and strangulated hernias] and studying the effect of weekend admission on these illnesses."
Dr. Worni’s and colleagues’ was funded by a grant from the Swiss National Science Foundation. None of the investigators declared conflicts of interest. Dr. Metzger declared that he had no conflicts of interest related to his critique.
Patients who were admitted for emergency surgery on a weekend to treat left-sided diverticulitis experience more short-term complications and are markedly more likely to undergo a Hartmann procedure than are those admitted on weekdays, according to results from a large population-based study.
Longer hospital stays, significantly higher treatment costs, and higher rates of reoperations were also associated with weekend admission. However, no differences in mortality were observed between the patient groups.
Previous studies have shown worse outcomes for patients with gastrointestinal hemorrhage, kidney injury, myocardial infarction, pulmonary embolism, and intracerebral hemorrhage when they were admitted on weekends. Although the current study, led by Dr. Mathias Worni of Duke University Medical Center in Durham, N.C., and Bern (Switzerland) University Hospital, was not designed to isolate the cause of the "weekend effect" for left-sided diverticulitis patients, the authors noted that hospital staffing tends to be reduced on weekends – especially among specialists such as colorectal surgeons.
Dr. Worni and his colleagues looked at records from the Nationwide Inpatient Sample between January 2002 and December 2008. Of the 31,832 patients who were treated surgically for left-sided diverticulitis, 7,066 (22.2%) were admitted on weekends and 24,766 (77.8%) on weekdays. Patients’ mean age was 60.8 years, and more than half were women.
Among patients who were admitted on a Saturday or Sunday, a Hartmann procedure was performed on 64.8% (n = 4,580), compared with only 53.9% (n = 13,351) for those admitted on a weekday (Arch. Surg. 2012;147:649-55). The Hartmann procedure – which involves formation of a colostomy – has long been the standard surgery for people presenting with left-sided diverticulitis, but is associated with long-term complications and a low rate of reversals.
Primary anastomosis, in which colostomy is avoided, is increasingly preferred, but only 35.2% of patients who were admitted on weekends underwent primary anastomosis, compared with 46.1% of patients admitted on weekdays.
The investigators found that patients admitted on weekends had significantly higher risk for any postoperative complication (odds ratio, 1.10; P = .005), compared with patients admitted on weekdays. Risk of reoperation was also higher among weekend admissions (OR, 1.50; P less than .001).
Furthermore, median total hospital charges were $3,734 higher among patients treated on weekends, and the median length of hospital stay was 0.5 days longer (P less than .001). The authors observed that these findings should motivate improvements in the quality of weekend care.
"Physicians working on weekends are thought to be less experienced than teams working during the week," they wrote. Experienced and specialized colorectal surgeons have been shown to perform more primary anastomoses, compared with trainees or general surgeons (Arch. Surg. 2010;145:79-86; Dis. Colon Rectum 2003;46:1461-8).
Limitations of the study include the fact that it did not capture long-term outcomes or severity of disease at presentation. The latter could be of potential importance: "Some patients, especially those with milder symptoms, may prefer weekend or weekday admission and may time their admission accordingly," the investigators noted.
In an invited critique that accompanied the article, Dr. Juerg Metzger, a surgeon at Lucerne (Switzerland) Cantonal Hospital, wrote that a disparity in experience among weekday and weekend surgical staff likely accounted for the higher rate of Hartmann procedures and complications following weekend admissions.
"Work-hour restrictions do not seem to have a negative influence on mortality and morbidity in surgical patients," Dr. Metzger wrote. "However, reduced experience owing to restricted working hours may negatively influence the practical skills of younger surgeons, resulting in more limited surgery [for example, a Hartmann procedure being performed instead of a primary anastomosis] and an increase in complications related to that surgery."
In the end, Dr. Metzger wrote, "quality is expensive, and our society has to decide if it is desirable and necessary to have the best surgical quality available all the time, especially when considering that health care costs will dramatically increase. It would be relevant to analyze additional large databases, asking similar questions about the outcomes of other common diseases [for example, appendicitis, cholecystitis, and strangulated hernias] and studying the effect of weekend admission on these illnesses."
Dr. Worni’s and colleagues’ was funded by a grant from the Swiss National Science Foundation. None of the investigators declared conflicts of interest. Dr. Metzger declared that he had no conflicts of interest related to his critique.
Patients who were admitted for emergency surgery on a weekend to treat left-sided diverticulitis experience more short-term complications and are markedly more likely to undergo a Hartmann procedure than are those admitted on weekdays, according to results from a large population-based study.
Longer hospital stays, significantly higher treatment costs, and higher rates of reoperations were also associated with weekend admission. However, no differences in mortality were observed between the patient groups.
Previous studies have shown worse outcomes for patients with gastrointestinal hemorrhage, kidney injury, myocardial infarction, pulmonary embolism, and intracerebral hemorrhage when they were admitted on weekends. Although the current study, led by Dr. Mathias Worni of Duke University Medical Center in Durham, N.C., and Bern (Switzerland) University Hospital, was not designed to isolate the cause of the "weekend effect" for left-sided diverticulitis patients, the authors noted that hospital staffing tends to be reduced on weekends – especially among specialists such as colorectal surgeons.
Dr. Worni and his colleagues looked at records from the Nationwide Inpatient Sample between January 2002 and December 2008. Of the 31,832 patients who were treated surgically for left-sided diverticulitis, 7,066 (22.2%) were admitted on weekends and 24,766 (77.8%) on weekdays. Patients’ mean age was 60.8 years, and more than half were women.
Among patients who were admitted on a Saturday or Sunday, a Hartmann procedure was performed on 64.8% (n = 4,580), compared with only 53.9% (n = 13,351) for those admitted on a weekday (Arch. Surg. 2012;147:649-55). The Hartmann procedure – which involves formation of a colostomy – has long been the standard surgery for people presenting with left-sided diverticulitis, but is associated with long-term complications and a low rate of reversals.
Primary anastomosis, in which colostomy is avoided, is increasingly preferred, but only 35.2% of patients who were admitted on weekends underwent primary anastomosis, compared with 46.1% of patients admitted on weekdays.
The investigators found that patients admitted on weekends had significantly higher risk for any postoperative complication (odds ratio, 1.10; P = .005), compared with patients admitted on weekdays. Risk of reoperation was also higher among weekend admissions (OR, 1.50; P less than .001).
Furthermore, median total hospital charges were $3,734 higher among patients treated on weekends, and the median length of hospital stay was 0.5 days longer (P less than .001). The authors observed that these findings should motivate improvements in the quality of weekend care.
"Physicians working on weekends are thought to be less experienced than teams working during the week," they wrote. Experienced and specialized colorectal surgeons have been shown to perform more primary anastomoses, compared with trainees or general surgeons (Arch. Surg. 2010;145:79-86; Dis. Colon Rectum 2003;46:1461-8).
Limitations of the study include the fact that it did not capture long-term outcomes or severity of disease at presentation. The latter could be of potential importance: "Some patients, especially those with milder symptoms, may prefer weekend or weekday admission and may time their admission accordingly," the investigators noted.
In an invited critique that accompanied the article, Dr. Juerg Metzger, a surgeon at Lucerne (Switzerland) Cantonal Hospital, wrote that a disparity in experience among weekday and weekend surgical staff likely accounted for the higher rate of Hartmann procedures and complications following weekend admissions.
"Work-hour restrictions do not seem to have a negative influence on mortality and morbidity in surgical patients," Dr. Metzger wrote. "However, reduced experience owing to restricted working hours may negatively influence the practical skills of younger surgeons, resulting in more limited surgery [for example, a Hartmann procedure being performed instead of a primary anastomosis] and an increase in complications related to that surgery."
In the end, Dr. Metzger wrote, "quality is expensive, and our society has to decide if it is desirable and necessary to have the best surgical quality available all the time, especially when considering that health care costs will dramatically increase. It would be relevant to analyze additional large databases, asking similar questions about the outcomes of other common diseases [for example, appendicitis, cholecystitis, and strangulated hernias] and studying the effect of weekend admission on these illnesses."
Dr. Worni’s and colleagues’ was funded by a grant from the Swiss National Science Foundation. None of the investigators declared conflicts of interest. Dr. Metzger declared that he had no conflicts of interest related to his critique.
FROM ARCHIVES OF SURGERY
Major Finding: Weekend admission to the hospital for diverticulitis posed a significantly higher risk for any postoperative complication (OR, 1.10; P = .005) and risk of reoperation (OR, 1.50; P less than .001), compared with weekday admission.
Data Source: The findings are based on an analysis of NIS records for 31,832 patients who were treated surgically for left-sided diverticulitis.
Disclosures: Dr. Worni’s and colleagues’ study was funded by a grant from the Swiss National Science Foundation. None of the investigators declared conflicts of interest. Dr. Metzger declared that he had no conflicts of interest related to his critique.
Antegrade Beats Retrograde Enteroscopy in Small Bowel Disease
Antegrade enteroscopy had a significantly greater diagnostic and therapeutic yield in small bowel disease, compared with retrograde enteroscopy, reported Dr. Madhusudhan R. Sanaka and colleagues in the August issue of Clinical Gastroenterology and Hepatology.
Moreover, antegrade enteroscopy had a significantly shorter mean duration, with a greater mean depth of maximal insertion, the authors added.
In what the researchers called "the first study ... to compare the efficacy of all three available enteroscopy systems between antegrade and retrograde approach" in small bowel disease, Dr. Sanaka, of the Digestive Disease Institute at the Cleveland Clinic, studied 250 such procedures performed at that institution between January 2008 and August 2009.
A total of 182 procedures were antegrade (91 with a single-balloon enteroscope, 52 with a double-balloon enteroscope, and 39 with a spiral enteroscope), and 68 were retrograde (23 with a single balloon, 37 with a double balloon, and 8 with a spiral enteroscope).
The mean age of all participants was 61.5 years, and the antegrade and retrograde groups did not differ significantly on any of the demographic factors or history of prior capsule endoscopies.
Although obscure gastrointestinal bleeding was the most common indication in both groups, "abdominal pain or suspected Crohn’s disease was a much more common indication for antegrade enteroscopy when compared to retrograde (18.7% vs. 4.4%, P less than .001)," wrote the authors.
Overall, the diagnostic yield of antegrade enteroscopy was significantly greater, at 63.7%, than the yield of the retrograde procedures (39.7%), with P less than .001 (Clin. Gastroenterol. Hepatol. 2012 [doi: 10.1016/j.cgh.2012.04.020]).
The investigators then looked at the therapeutic yield of the two procedures. "With the antegrade approach, in 59 procedures (32.4%), a therapeutic intervention was performed," including argon plasma coagulation in 52 cases (28.6%), dilatation in 1 (0.6%), and polypectomy in 4 cases (2.2%).
With the retrograde approach, therapies were initiated in just 14.7% of cases, which was significantly lower than the percentage for the antegrade approach (P less than .001).
The authors also compared the technical aspects of the different procedure types. In this study, antegrade enteroscopies lasted 44.3 minutes on average, versus 58.9 minutes for the retrograde procedures (P less than .001).
Antegrade procedures also achieved a significantly greater depth of maximal insertion on average, at 231.8 cm, compared with 103.4 cm for retrograde procedures (P less than .001).
The authors conceded that the study had several limitations. Not only was it retrospective, they wrote, "there was no randomization and hence there could have been a significant bias in patient selection and use of a particular enteroscopy approach in individual cases, particularly in patients in whom the source of small bowel disorder was not known."
Nevertheless, "our findings of higher diagnostic and therapeutic yields with antegrade enteroscopy compared to retrograde enteroscopy support the expert opinion to consider antegrade enteroscopy as a default initial approach for suspected small bowel disease," the authors concluded.
"Retrograde enteroscopy may be considered when the antegrade enteroscopy is either nondiagnostic or if the abnormalities identified are unlikely to account for the patient’s symptoms," or when capsule endoscopy or radiologic imaging studies indicate that distal small bowel disease is likely, such as in suspected Crohn’s disease.
One of the authors, Dr. John Vargo, declared that he is a consultant for Olympus America, maker of enteroscopes and other devices. The authors stated that there was no outside funding.
Antegrade enteroscopy had a significantly greater diagnostic and therapeutic yield in small bowel disease, compared with retrograde enteroscopy, reported Dr. Madhusudhan R. Sanaka and colleagues in the August issue of Clinical Gastroenterology and Hepatology.
Moreover, antegrade enteroscopy had a significantly shorter mean duration, with a greater mean depth of maximal insertion, the authors added.
In what the researchers called "the first study ... to compare the efficacy of all three available enteroscopy systems between antegrade and retrograde approach" in small bowel disease, Dr. Sanaka, of the Digestive Disease Institute at the Cleveland Clinic, studied 250 such procedures performed at that institution between January 2008 and August 2009.
A total of 182 procedures were antegrade (91 with a single-balloon enteroscope, 52 with a double-balloon enteroscope, and 39 with a spiral enteroscope), and 68 were retrograde (23 with a single balloon, 37 with a double balloon, and 8 with a spiral enteroscope).
The mean age of all participants was 61.5 years, and the antegrade and retrograde groups did not differ significantly on any of the demographic factors or history of prior capsule endoscopies.
Although obscure gastrointestinal bleeding was the most common indication in both groups, "abdominal pain or suspected Crohn’s disease was a much more common indication for antegrade enteroscopy when compared to retrograde (18.7% vs. 4.4%, P less than .001)," wrote the authors.
Overall, the diagnostic yield of antegrade enteroscopy was significantly greater, at 63.7%, than the yield of the retrograde procedures (39.7%), with P less than .001 (Clin. Gastroenterol. Hepatol. 2012 [doi: 10.1016/j.cgh.2012.04.020]).
The investigators then looked at the therapeutic yield of the two procedures. "With the antegrade approach, in 59 procedures (32.4%), a therapeutic intervention was performed," including argon plasma coagulation in 52 cases (28.6%), dilatation in 1 (0.6%), and polypectomy in 4 cases (2.2%).
With the retrograde approach, therapies were initiated in just 14.7% of cases, which was significantly lower than the percentage for the antegrade approach (P less than .001).
The authors also compared the technical aspects of the different procedure types. In this study, antegrade enteroscopies lasted 44.3 minutes on average, versus 58.9 minutes for the retrograde procedures (P less than .001).
Antegrade procedures also achieved a significantly greater depth of maximal insertion on average, at 231.8 cm, compared with 103.4 cm for retrograde procedures (P less than .001).
The authors conceded that the study had several limitations. Not only was it retrospective, they wrote, "there was no randomization and hence there could have been a significant bias in patient selection and use of a particular enteroscopy approach in individual cases, particularly in patients in whom the source of small bowel disorder was not known."
Nevertheless, "our findings of higher diagnostic and therapeutic yields with antegrade enteroscopy compared to retrograde enteroscopy support the expert opinion to consider antegrade enteroscopy as a default initial approach for suspected small bowel disease," the authors concluded.
"Retrograde enteroscopy may be considered when the antegrade enteroscopy is either nondiagnostic or if the abnormalities identified are unlikely to account for the patient’s symptoms," or when capsule endoscopy or radiologic imaging studies indicate that distal small bowel disease is likely, such as in suspected Crohn’s disease.
One of the authors, Dr. John Vargo, declared that he is a consultant for Olympus America, maker of enteroscopes and other devices. The authors stated that there was no outside funding.
Antegrade enteroscopy had a significantly greater diagnostic and therapeutic yield in small bowel disease, compared with retrograde enteroscopy, reported Dr. Madhusudhan R. Sanaka and colleagues in the August issue of Clinical Gastroenterology and Hepatology.
Moreover, antegrade enteroscopy had a significantly shorter mean duration, with a greater mean depth of maximal insertion, the authors added.
In what the researchers called "the first study ... to compare the efficacy of all three available enteroscopy systems between antegrade and retrograde approach" in small bowel disease, Dr. Sanaka, of the Digestive Disease Institute at the Cleveland Clinic, studied 250 such procedures performed at that institution between January 2008 and August 2009.
A total of 182 procedures were antegrade (91 with a single-balloon enteroscope, 52 with a double-balloon enteroscope, and 39 with a spiral enteroscope), and 68 were retrograde (23 with a single balloon, 37 with a double balloon, and 8 with a spiral enteroscope).
The mean age of all participants was 61.5 years, and the antegrade and retrograde groups did not differ significantly on any of the demographic factors or history of prior capsule endoscopies.
Although obscure gastrointestinal bleeding was the most common indication in both groups, "abdominal pain or suspected Crohn’s disease was a much more common indication for antegrade enteroscopy when compared to retrograde (18.7% vs. 4.4%, P less than .001)," wrote the authors.
Overall, the diagnostic yield of antegrade enteroscopy was significantly greater, at 63.7%, than the yield of the retrograde procedures (39.7%), with P less than .001 (Clin. Gastroenterol. Hepatol. 2012 [doi: 10.1016/j.cgh.2012.04.020]).
The investigators then looked at the therapeutic yield of the two procedures. "With the antegrade approach, in 59 procedures (32.4%), a therapeutic intervention was performed," including argon plasma coagulation in 52 cases (28.6%), dilatation in 1 (0.6%), and polypectomy in 4 cases (2.2%).
With the retrograde approach, therapies were initiated in just 14.7% of cases, which was significantly lower than the percentage for the antegrade approach (P less than .001).
The authors also compared the technical aspects of the different procedure types. In this study, antegrade enteroscopies lasted 44.3 minutes on average, versus 58.9 minutes for the retrograde procedures (P less than .001).
Antegrade procedures also achieved a significantly greater depth of maximal insertion on average, at 231.8 cm, compared with 103.4 cm for retrograde procedures (P less than .001).
The authors conceded that the study had several limitations. Not only was it retrospective, they wrote, "there was no randomization and hence there could have been a significant bias in patient selection and use of a particular enteroscopy approach in individual cases, particularly in patients in whom the source of small bowel disorder was not known."
Nevertheless, "our findings of higher diagnostic and therapeutic yields with antegrade enteroscopy compared to retrograde enteroscopy support the expert opinion to consider antegrade enteroscopy as a default initial approach for suspected small bowel disease," the authors concluded.
"Retrograde enteroscopy may be considered when the antegrade enteroscopy is either nondiagnostic or if the abnormalities identified are unlikely to account for the patient’s symptoms," or when capsule endoscopy or radiologic imaging studies indicate that distal small bowel disease is likely, such as in suspected Crohn’s disease.
One of the authors, Dr. John Vargo, declared that he is a consultant for Olympus America, maker of enteroscopes and other devices. The authors stated that there was no outside funding.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Early Data Find No Adalimumab Teratogenicity
BALTIMORE – Exposure to adalimumab was not associated with any specific pattern of minor or major birth defects in women with rheumatoid arthritis taking the biologic drug during pregnancy, according to preliminary data from an ongoing prospective cohort study.
Between November 2004 and January 2012, 312 pregnant women in the United States and Canada – 69 women with RA exposed to adalimumab, 80 women with RA who had not taken adalimumab, and 163 healthy controls – were enrolled before 20 weeks’ gestation. Their mean age was 32-33 years, and about two-thirds were white.
Major birth defects among the live births were identified in 5% of the babies born to women exposed to adalimumab, compared with about 4% among disease-matched controls who did not take adalimumab, and about 7% among healthy controls, Christina Chambers, Ph.D., of the University of California, San Diego, reported at the annual meeting of the Teratology Society.
The rate of minor structural abnormalities was similar in the three groups, at about 22%-24%, and there was no pattern of major or minor structural defects noted among the adalimumab-exposed group. (The three major malformations in the adalimumab-exposed group were one ventricular septal defect, one unilateral cryptorchidism, and one case of microcephaly.)
There were no stillbirths. The rate of spontaneous abortions was not significantly different between the three groups, nor were the rates of preterm delivery or birth weights, said Dr. Chambers, director of the California Teratogen Information Service and Clinical Research Program.
Through 1-year of follow-up, there were no malignancies among the infants and the rates of serious infections in the three groups were similar (about 3% in the two RA groups and 2% in the healthy comparison group).
The teratogenic effects of adalimumab, a tumor necrosis factor blocker, are being evaluated in the pregnancy registry, which is part of the Organization of Teratology Information Specialists (OTIS) Autoimmune Diseases in Pregnancy Project.
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved in the United States in 2002 as a treatment for people with moderately to severely active RA, and has since been approved for other autoimmune diseases, including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and psoriasis.
The registry study is comparing outcomes in women with RA who are treated with adalimumab during pregnancy, in women with RA not treated with adalimumab during pregnancy, and in women who do not have an autoimmune disease and have not been exposed to adalimumab or any known teratogenic drug during pregnancy. The study includes medical record reviews, examination of infants for major and minor structural abnormalities, and follow-up for 1 year post partum. It is expected to continue through 2017; the pregnant women are recruited from OTIS member services and from rheumatologists, and other clinicians who care for these patients.
Although little to no placental transfer of adalimumab is expected during early pregnancy, limited information on the safety of adalimumab during pregnancy has been published, Dr. Chambers said.
Abbott Laboratories is among the sponsors of the OTIS Autoimmune Diseases in Pregnancy Project, which is also evaluating safety of medications in women with ankylosing spondylitis, psoriasis and psoriatic arthritis, and Crohn’s disease. Dr. Chambers and her coauthors have received or receive grant funding for research on medications for autoimmune diseases from Abbott and other manufacturers: Amgen, Bristol Myers Squibb, Roche Genentech, Sanofi, Teva, Par, Sandoz, and Apotex.
Information for women and clinicians interested in enrolling in the OTIS Autoimmune Diseases in Pregnancy Project is available at www.otispregnancy.org/autoimmune-studies-s13049.
BALTIMORE – Exposure to adalimumab was not associated with any specific pattern of minor or major birth defects in women with rheumatoid arthritis taking the biologic drug during pregnancy, according to preliminary data from an ongoing prospective cohort study.
Between November 2004 and January 2012, 312 pregnant women in the United States and Canada – 69 women with RA exposed to adalimumab, 80 women with RA who had not taken adalimumab, and 163 healthy controls – were enrolled before 20 weeks’ gestation. Their mean age was 32-33 years, and about two-thirds were white.
Major birth defects among the live births were identified in 5% of the babies born to women exposed to adalimumab, compared with about 4% among disease-matched controls who did not take adalimumab, and about 7% among healthy controls, Christina Chambers, Ph.D., of the University of California, San Diego, reported at the annual meeting of the Teratology Society.
The rate of minor structural abnormalities was similar in the three groups, at about 22%-24%, and there was no pattern of major or minor structural defects noted among the adalimumab-exposed group. (The three major malformations in the adalimumab-exposed group were one ventricular septal defect, one unilateral cryptorchidism, and one case of microcephaly.)
There were no stillbirths. The rate of spontaneous abortions was not significantly different between the three groups, nor were the rates of preterm delivery or birth weights, said Dr. Chambers, director of the California Teratogen Information Service and Clinical Research Program.
Through 1-year of follow-up, there were no malignancies among the infants and the rates of serious infections in the three groups were similar (about 3% in the two RA groups and 2% in the healthy comparison group).
The teratogenic effects of adalimumab, a tumor necrosis factor blocker, are being evaluated in the pregnancy registry, which is part of the Organization of Teratology Information Specialists (OTIS) Autoimmune Diseases in Pregnancy Project.
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved in the United States in 2002 as a treatment for people with moderately to severely active RA, and has since been approved for other autoimmune diseases, including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and psoriasis.
The registry study is comparing outcomes in women with RA who are treated with adalimumab during pregnancy, in women with RA not treated with adalimumab during pregnancy, and in women who do not have an autoimmune disease and have not been exposed to adalimumab or any known teratogenic drug during pregnancy. The study includes medical record reviews, examination of infants for major and minor structural abnormalities, and follow-up for 1 year post partum. It is expected to continue through 2017; the pregnant women are recruited from OTIS member services and from rheumatologists, and other clinicians who care for these patients.
Although little to no placental transfer of adalimumab is expected during early pregnancy, limited information on the safety of adalimumab during pregnancy has been published, Dr. Chambers said.
Abbott Laboratories is among the sponsors of the OTIS Autoimmune Diseases in Pregnancy Project, which is also evaluating safety of medications in women with ankylosing spondylitis, psoriasis and psoriatic arthritis, and Crohn’s disease. Dr. Chambers and her coauthors have received or receive grant funding for research on medications for autoimmune diseases from Abbott and other manufacturers: Amgen, Bristol Myers Squibb, Roche Genentech, Sanofi, Teva, Par, Sandoz, and Apotex.
Information for women and clinicians interested in enrolling in the OTIS Autoimmune Diseases in Pregnancy Project is available at www.otispregnancy.org/autoimmune-studies-s13049.
BALTIMORE – Exposure to adalimumab was not associated with any specific pattern of minor or major birth defects in women with rheumatoid arthritis taking the biologic drug during pregnancy, according to preliminary data from an ongoing prospective cohort study.
Between November 2004 and January 2012, 312 pregnant women in the United States and Canada – 69 women with RA exposed to adalimumab, 80 women with RA who had not taken adalimumab, and 163 healthy controls – were enrolled before 20 weeks’ gestation. Their mean age was 32-33 years, and about two-thirds were white.
Major birth defects among the live births were identified in 5% of the babies born to women exposed to adalimumab, compared with about 4% among disease-matched controls who did not take adalimumab, and about 7% among healthy controls, Christina Chambers, Ph.D., of the University of California, San Diego, reported at the annual meeting of the Teratology Society.
The rate of minor structural abnormalities was similar in the three groups, at about 22%-24%, and there was no pattern of major or minor structural defects noted among the adalimumab-exposed group. (The three major malformations in the adalimumab-exposed group were one ventricular septal defect, one unilateral cryptorchidism, and one case of microcephaly.)
There were no stillbirths. The rate of spontaneous abortions was not significantly different between the three groups, nor were the rates of preterm delivery or birth weights, said Dr. Chambers, director of the California Teratogen Information Service and Clinical Research Program.
Through 1-year of follow-up, there were no malignancies among the infants and the rates of serious infections in the three groups were similar (about 3% in the two RA groups and 2% in the healthy comparison group).
The teratogenic effects of adalimumab, a tumor necrosis factor blocker, are being evaluated in the pregnancy registry, which is part of the Organization of Teratology Information Specialists (OTIS) Autoimmune Diseases in Pregnancy Project.
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved in the United States in 2002 as a treatment for people with moderately to severely active RA, and has since been approved for other autoimmune diseases, including psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, and psoriasis.
The registry study is comparing outcomes in women with RA who are treated with adalimumab during pregnancy, in women with RA not treated with adalimumab during pregnancy, and in women who do not have an autoimmune disease and have not been exposed to adalimumab or any known teratogenic drug during pregnancy. The study includes medical record reviews, examination of infants for major and minor structural abnormalities, and follow-up for 1 year post partum. It is expected to continue through 2017; the pregnant women are recruited from OTIS member services and from rheumatologists, and other clinicians who care for these patients.
Although little to no placental transfer of adalimumab is expected during early pregnancy, limited information on the safety of adalimumab during pregnancy has been published, Dr. Chambers said.
Abbott Laboratories is among the sponsors of the OTIS Autoimmune Diseases in Pregnancy Project, which is also evaluating safety of medications in women with ankylosing spondylitis, psoriasis and psoriatic arthritis, and Crohn’s disease. Dr. Chambers and her coauthors have received or receive grant funding for research on medications for autoimmune diseases from Abbott and other manufacturers: Amgen, Bristol Myers Squibb, Roche Genentech, Sanofi, Teva, Par, Sandoz, and Apotex.
Information for women and clinicians interested in enrolling in the OTIS Autoimmune Diseases in Pregnancy Project is available at www.otispregnancy.org/autoimmune-studies-s13049.
AT THE ANNUAL MEETING OF THE TERATOLOGY SOCIETY
Spondyloarthropathy Common a Decade After IBD Diagnosis
BERLIN – The cumulative incidence of spondyloarthropathy in the first decade after diagnosis of inflammatory bowel disease is about 1 in 50 patients, climbing to 1 in 25 at 20 years and 1 in 14 at 30 years, according to the first population-based study to scrutinize the issue.
The incidence of spondyloarthropathy following diagnosis of ulcerative colitis is about half that seen in Crohn’s disease patients, said Dr. Eric L. Matteson at the Annual European Congress of Rheumatology.
Dr. Matteson, a professor of medicine and chair of the department of rheumatology at the Mayo Clinic, Rochester, Minn., and his coinvestigators analyzed the medical records of Olmsted County, Minn., residents who had been diagnosed with inflammatory bowel disease (IBD) during 1970-2004. The 311 patients diagnosed with Crohn’s disease and 365 with ulcerative colitis were followed longitudinally through June 2011. The median age at diagnosis of IBD was 33 years.
The prevalence of diagnosed spondyloarthropathy prior to diagnosis of IBD was 1%. The cumulative incidence of spondyloarthropathy following an established IBD diagnosis was 2.3% at 10 years, 4.2% at 20 years, and 7.1% at 30 years. The overall cumulative incidence of ankylosing spondylitis was 0.2% at 10 years, rising to 0.7% at 20 years.
The 10-, 20- and 30-year cumulative incidence rates for spondyloarthropathy after diagnosis of Crohn’s disease were 2.6%, 6.1%, and 9.8%, respectively, as compared with 1.9%, 2.8%, and 4.9% following diagnosis of ulcerative colitis.
Clinical features of spondyloarthropathy that appeared only after diagnosis of IBD included oligoarthritis in 3.1%, polyarthritis in 1.3%, sacroiliitis in 1.6%, psoriasis in 2.2%, uveitis in 2.9%, and plantar fasciitis in 5%.
Dr. Matteson reported having no financial conflicts.
BERLIN – The cumulative incidence of spondyloarthropathy in the first decade after diagnosis of inflammatory bowel disease is about 1 in 50 patients, climbing to 1 in 25 at 20 years and 1 in 14 at 30 years, according to the first population-based study to scrutinize the issue.
The incidence of spondyloarthropathy following diagnosis of ulcerative colitis is about half that seen in Crohn’s disease patients, said Dr. Eric L. Matteson at the Annual European Congress of Rheumatology.
Dr. Matteson, a professor of medicine and chair of the department of rheumatology at the Mayo Clinic, Rochester, Minn., and his coinvestigators analyzed the medical records of Olmsted County, Minn., residents who had been diagnosed with inflammatory bowel disease (IBD) during 1970-2004. The 311 patients diagnosed with Crohn’s disease and 365 with ulcerative colitis were followed longitudinally through June 2011. The median age at diagnosis of IBD was 33 years.
The prevalence of diagnosed spondyloarthropathy prior to diagnosis of IBD was 1%. The cumulative incidence of spondyloarthropathy following an established IBD diagnosis was 2.3% at 10 years, 4.2% at 20 years, and 7.1% at 30 years. The overall cumulative incidence of ankylosing spondylitis was 0.2% at 10 years, rising to 0.7% at 20 years.
The 10-, 20- and 30-year cumulative incidence rates for spondyloarthropathy after diagnosis of Crohn’s disease were 2.6%, 6.1%, and 9.8%, respectively, as compared with 1.9%, 2.8%, and 4.9% following diagnosis of ulcerative colitis.
Clinical features of spondyloarthropathy that appeared only after diagnosis of IBD included oligoarthritis in 3.1%, polyarthritis in 1.3%, sacroiliitis in 1.6%, psoriasis in 2.2%, uveitis in 2.9%, and plantar fasciitis in 5%.
Dr. Matteson reported having no financial conflicts.
BERLIN – The cumulative incidence of spondyloarthropathy in the first decade after diagnosis of inflammatory bowel disease is about 1 in 50 patients, climbing to 1 in 25 at 20 years and 1 in 14 at 30 years, according to the first population-based study to scrutinize the issue.
The incidence of spondyloarthropathy following diagnosis of ulcerative colitis is about half that seen in Crohn’s disease patients, said Dr. Eric L. Matteson at the Annual European Congress of Rheumatology.
Dr. Matteson, a professor of medicine and chair of the department of rheumatology at the Mayo Clinic, Rochester, Minn., and his coinvestigators analyzed the medical records of Olmsted County, Minn., residents who had been diagnosed with inflammatory bowel disease (IBD) during 1970-2004. The 311 patients diagnosed with Crohn’s disease and 365 with ulcerative colitis were followed longitudinally through June 2011. The median age at diagnosis of IBD was 33 years.
The prevalence of diagnosed spondyloarthropathy prior to diagnosis of IBD was 1%. The cumulative incidence of spondyloarthropathy following an established IBD diagnosis was 2.3% at 10 years, 4.2% at 20 years, and 7.1% at 30 years. The overall cumulative incidence of ankylosing spondylitis was 0.2% at 10 years, rising to 0.7% at 20 years.
The 10-, 20- and 30-year cumulative incidence rates for spondyloarthropathy after diagnosis of Crohn’s disease were 2.6%, 6.1%, and 9.8%, respectively, as compared with 1.9%, 2.8%, and 4.9% following diagnosis of ulcerative colitis.
Clinical features of spondyloarthropathy that appeared only after diagnosis of IBD included oligoarthritis in 3.1%, polyarthritis in 1.3%, sacroiliitis in 1.6%, psoriasis in 2.2%, uveitis in 2.9%, and plantar fasciitis in 5%.
Dr. Matteson reported having no financial conflicts.
AT THE ANNUAL EUROPEAN CONGRESS OF RHEUMATOLOGY
Major Finding: Roughly 1in 50 patients will be diagnosed with spondyloarthropathy during the first decade after receiving a diagnosis of inflammatory bowel disease.
Data Source: This finding comes from a population-based longitudinal study conducted in Olmsted County, Minn., residents diagnosed with ulcerative colitis or Crohn’s disease.
Disclosures: The investigators reported having no financial conflicts.
Vedolizumab Scores on Safety, Efficacy for Ulcerative Colitis
SAN DIEGO – Vedolizumab, a novel drug that selectively blocks lymphocyte trafficking to the gut, was safe and highly effective for inducing and maintaining clinical remission in patients with moderate to severe ulcerative colitis in a pivotal phase III trial.
"If the findings hold up with more detailed analysis, it looks like we’ll have the efficacy of a biologic agent without some of the toxicity issues that we’ve seen with anti-TNF [tumor necrosis factor] drugs," said Dr. William J. Sandborn, professor of medicine and chief of gastroenterology at the University of California, San Diego.
"This potentially could be a first-line drug," for treating moderate to severe ulcerative colitis, said Dr. Sandborn, a coinvestigator in the study.
"Vedolizumab was more effective than placebo for induction and maintenance treatment, including both anti-TNF–exposed and naive patients," Dr. Brian G. Feagan, the study’s lead investigator, said at the annual Digestive Disease Week. In addition, there was little difference between the vedolizumab and placebo groups in terms of adverse events, serious adverse events, and serious infections, said Dr. Feagan, professor of medicine at Western University in London, Ont.
At the end of a year of maintenance treatment, patients kept on the more frequent vedolizumab dosage tested, 300 mg delivered intravenously once every 4 weeks, showed a potent efficacy effect, surpassing the placebo group in corticosteroid-free clinical remissions by 31 percentage points (45% vs. 14%). "Nothing else is that good," Dr. Sandborn said in an interview. "Steroid-free remission with a delta over placebo of 30% is very impressive, especially when you factor in that many of the patients had failed anti-TNF treatment.
"We thought that vedolizumab would be safer than systemic immunosuppression, and I think the data are consistent with that. This will be a first-line treatment," Dr. Feagan said.
Equally important, total worldwide experience with vedolizumab, which now includes about 2,500 patients, has not yet resulted in a single case of progressive multifocal leukoencephalopathy (PML), an adverse effect produced by vedolizumab’s cousin drug, natalizumab (Tysarbi), ’approved for U.S. marketing to treat multiple sclerosis and Crohns disease.
Vedolizumab is a humanized monoclonal antibody that specifically binds to the alpha-4 beta-7 integrin protein that helps move leukocytes into the gut. Natalizumab is a less specific integrin antagonist that affects the protein’s actions and immune-cell trafficking to a variety of body sites, including the brain. Natalizumab "essentially blocks immune surveillance in the brain, and that allows for the JC virus [carried by about 50% of people] to cause PML," explained Dr. Sandborn. In contrast, vedolizumab does not cause "a systemic blockade of lymphocytes trafficking; it only affects a small fraction of lymphocytes, and leaves the brain completely unaffected."
Patients most at risk for PML are those who previously received immunosuppressive treatment with a drug such as azathioprine, methotrexate, or an anti-TNF drug. Patients with that history who received natalizumab for 1-2 years have about a 1 in 500-600 risk for PML, and those who got natalizumab for more than 2 years have a 1% risk.
By comparison, vedolizomab’s clean record based on 2,500 recipients "looks like it might have a very nice safety profile. If people can get comfortable with vedolizumab being different from natalizumab, then it has the potential to be first-line therapy for patients who have the worst prognosis," those who don’t respond to treatment with mesalamine.
The GEMINI I trial enrolled patients at 105 international sites with ulcerative colitis who had a Mayo score of at least 6 and an endoscopic subscore of at least 2 (indicating moderate disease) despite standard treatments. Patients were an average age of 40 years, their average duration of disease was 7 years, and their average Mayo score at entry was 8.5. Roughly 40% had previously received an anti-TNF treatment, about a third had failed on an anti-TNF drug, and just over half of the patients entered the study on a corticosteroid.
The trial included an induction phase that randomized 225 patients to a 6-week regimen with vedolizumab infusions and 149 patients to placebo. The researchers started another 521 ulcerative colitis patients on an open-label induction regimen, and then randomized 373 patients who responded after 6 weeks to maintenance infusion with vedolizumab every 4 weeks, a vedolizumab infusion every 8 weeks, or placebo.
The primary end point of the induction phase was clinical response, defined as a drop in the Mayo score of at least 3 points and at least 30%, plus a drop in the rectal bleeding score of at least 1 point or an absolute rectal bleeding subscore of 1 point or less. Achievement of this end point occurred in 47% of patients on vedolizumab and in 26% of those on placebo, a statistically significant difference. Among patients previously treated with an anti-TNF drug, 39% had a clinical response after 6 weeks on vedolizumab compared with 21% in the placebo arm, a significant difference. Among anti-TNF–naive patients, the rates were 53% and 26%.
The primary end point of the maintenance phase was clinical remission at 52 weeks, defined as a total Mayo score of 2 or less and no subscore greater than 1 point. This end point was met by 45% of patients who received vedolizumab every 4 weeks, by 42% who received the drug every 8 weeks, and by 16% of the placebo patients. Clinical remission without corticosteroid treatment occurred in 45% of patients who got vedolizumab every 4 weeks, 31% who got the drug every 8 weeks, and 14% who took placebo. Among patients with a history of anti-TNF treatment, clinical remission after 1 year occurred in 35% of patients maintained with the drug every 4 weeks, 37% who got the drug every 8 weeks, and 5% who took placebo. A total of 209 patients remained in the study through the 52-week assessment.
On May 11, Millennium Pharmaceuticals, the company developing vedolizumab, announced that the drug significantly surpassed placebo for the treatment of Crohn’s disease in the pivotal, phase III trial for that indication. The company said that it will soon report the full Crohn’s disease results.
The GEMINI I study was funded by Millennium. Dr. Feagan said he has been a consultant to Millennium and to several other drug companies. Dr. Sandborn said he has been a consultant to several drug companies but has no relationship with Millennium.
Vedolizumab is a biological therapy that blocks alpha-4/beta-7 integrins. This mechanism is novel within our armamentarium of available therapies for ulcerative colitis, and vedolizumab is also the first non-systemically acting biologic therapy. The induction and maintenance results of this phase III trial of vedolizumab in moderate to severe ulcerative colitis are very encouraging and represent a major advance in our field. It appears that the impressive safety profile (infection rate was similar to placebo) seen in this trial is likely due to its gut-specific activity.
Furthermore, it is of interest that despite inhibition of lymphocyte trafficking in the gut, there does not appear to be an increased risk of GI infections. If these impressive results pan out in clinical practice, vedolizumab undoubtedly will become a significant option for the management of our ulcerative colitis patients, and will have a prominent place in our treatment algorithms. We eagerly await FDA review and further studies, including the Crohn’s disease trial results.
David T. Rubin, M.D., AGAF, is Professor of Medicine, Associate Section Chief for Education, and Co-Director, Inflammatory Bowel Disease Center, University of Chicago.
Vedolizumab is a biological therapy that blocks alpha-4/beta-7 integrins. This mechanism is novel within our armamentarium of available therapies for ulcerative colitis, and vedolizumab is also the first non-systemically acting biologic therapy. The induction and maintenance results of this phase III trial of vedolizumab in moderate to severe ulcerative colitis are very encouraging and represent a major advance in our field. It appears that the impressive safety profile (infection rate was similar to placebo) seen in this trial is likely due to its gut-specific activity.
Furthermore, it is of interest that despite inhibition of lymphocyte trafficking in the gut, there does not appear to be an increased risk of GI infections. If these impressive results pan out in clinical practice, vedolizumab undoubtedly will become a significant option for the management of our ulcerative colitis patients, and will have a prominent place in our treatment algorithms. We eagerly await FDA review and further studies, including the Crohn’s disease trial results.
David T. Rubin, M.D., AGAF, is Professor of Medicine, Associate Section Chief for Education, and Co-Director, Inflammatory Bowel Disease Center, University of Chicago.
Vedolizumab is a biological therapy that blocks alpha-4/beta-7 integrins. This mechanism is novel within our armamentarium of available therapies for ulcerative colitis, and vedolizumab is also the first non-systemically acting biologic therapy. The induction and maintenance results of this phase III trial of vedolizumab in moderate to severe ulcerative colitis are very encouraging and represent a major advance in our field. It appears that the impressive safety profile (infection rate was similar to placebo) seen in this trial is likely due to its gut-specific activity.
Furthermore, it is of interest that despite inhibition of lymphocyte trafficking in the gut, there does not appear to be an increased risk of GI infections. If these impressive results pan out in clinical practice, vedolizumab undoubtedly will become a significant option for the management of our ulcerative colitis patients, and will have a prominent place in our treatment algorithms. We eagerly await FDA review and further studies, including the Crohn’s disease trial results.
David T. Rubin, M.D., AGAF, is Professor of Medicine, Associate Section Chief for Education, and Co-Director, Inflammatory Bowel Disease Center, University of Chicago.
SAN DIEGO – Vedolizumab, a novel drug that selectively blocks lymphocyte trafficking to the gut, was safe and highly effective for inducing and maintaining clinical remission in patients with moderate to severe ulcerative colitis in a pivotal phase III trial.
"If the findings hold up with more detailed analysis, it looks like we’ll have the efficacy of a biologic agent without some of the toxicity issues that we’ve seen with anti-TNF [tumor necrosis factor] drugs," said Dr. William J. Sandborn, professor of medicine and chief of gastroenterology at the University of California, San Diego.
"This potentially could be a first-line drug," for treating moderate to severe ulcerative colitis, said Dr. Sandborn, a coinvestigator in the study.
"Vedolizumab was more effective than placebo for induction and maintenance treatment, including both anti-TNF–exposed and naive patients," Dr. Brian G. Feagan, the study’s lead investigator, said at the annual Digestive Disease Week. In addition, there was little difference between the vedolizumab and placebo groups in terms of adverse events, serious adverse events, and serious infections, said Dr. Feagan, professor of medicine at Western University in London, Ont.
At the end of a year of maintenance treatment, patients kept on the more frequent vedolizumab dosage tested, 300 mg delivered intravenously once every 4 weeks, showed a potent efficacy effect, surpassing the placebo group in corticosteroid-free clinical remissions by 31 percentage points (45% vs. 14%). "Nothing else is that good," Dr. Sandborn said in an interview. "Steroid-free remission with a delta over placebo of 30% is very impressive, especially when you factor in that many of the patients had failed anti-TNF treatment.
"We thought that vedolizumab would be safer than systemic immunosuppression, and I think the data are consistent with that. This will be a first-line treatment," Dr. Feagan said.
Equally important, total worldwide experience with vedolizumab, which now includes about 2,500 patients, has not yet resulted in a single case of progressive multifocal leukoencephalopathy (PML), an adverse effect produced by vedolizumab’s cousin drug, natalizumab (Tysarbi), ’approved for U.S. marketing to treat multiple sclerosis and Crohns disease.
Vedolizumab is a humanized monoclonal antibody that specifically binds to the alpha-4 beta-7 integrin protein that helps move leukocytes into the gut. Natalizumab is a less specific integrin antagonist that affects the protein’s actions and immune-cell trafficking to a variety of body sites, including the brain. Natalizumab "essentially blocks immune surveillance in the brain, and that allows for the JC virus [carried by about 50% of people] to cause PML," explained Dr. Sandborn. In contrast, vedolizumab does not cause "a systemic blockade of lymphocytes trafficking; it only affects a small fraction of lymphocytes, and leaves the brain completely unaffected."
Patients most at risk for PML are those who previously received immunosuppressive treatment with a drug such as azathioprine, methotrexate, or an anti-TNF drug. Patients with that history who received natalizumab for 1-2 years have about a 1 in 500-600 risk for PML, and those who got natalizumab for more than 2 years have a 1% risk.
By comparison, vedolizomab’s clean record based on 2,500 recipients "looks like it might have a very nice safety profile. If people can get comfortable with vedolizumab being different from natalizumab, then it has the potential to be first-line therapy for patients who have the worst prognosis," those who don’t respond to treatment with mesalamine.
The GEMINI I trial enrolled patients at 105 international sites with ulcerative colitis who had a Mayo score of at least 6 and an endoscopic subscore of at least 2 (indicating moderate disease) despite standard treatments. Patients were an average age of 40 years, their average duration of disease was 7 years, and their average Mayo score at entry was 8.5. Roughly 40% had previously received an anti-TNF treatment, about a third had failed on an anti-TNF drug, and just over half of the patients entered the study on a corticosteroid.
The trial included an induction phase that randomized 225 patients to a 6-week regimen with vedolizumab infusions and 149 patients to placebo. The researchers started another 521 ulcerative colitis patients on an open-label induction regimen, and then randomized 373 patients who responded after 6 weeks to maintenance infusion with vedolizumab every 4 weeks, a vedolizumab infusion every 8 weeks, or placebo.
The primary end point of the induction phase was clinical response, defined as a drop in the Mayo score of at least 3 points and at least 30%, plus a drop in the rectal bleeding score of at least 1 point or an absolute rectal bleeding subscore of 1 point or less. Achievement of this end point occurred in 47% of patients on vedolizumab and in 26% of those on placebo, a statistically significant difference. Among patients previously treated with an anti-TNF drug, 39% had a clinical response after 6 weeks on vedolizumab compared with 21% in the placebo arm, a significant difference. Among anti-TNF–naive patients, the rates were 53% and 26%.
The primary end point of the maintenance phase was clinical remission at 52 weeks, defined as a total Mayo score of 2 or less and no subscore greater than 1 point. This end point was met by 45% of patients who received vedolizumab every 4 weeks, by 42% who received the drug every 8 weeks, and by 16% of the placebo patients. Clinical remission without corticosteroid treatment occurred in 45% of patients who got vedolizumab every 4 weeks, 31% who got the drug every 8 weeks, and 14% who took placebo. Among patients with a history of anti-TNF treatment, clinical remission after 1 year occurred in 35% of patients maintained with the drug every 4 weeks, 37% who got the drug every 8 weeks, and 5% who took placebo. A total of 209 patients remained in the study through the 52-week assessment.
On May 11, Millennium Pharmaceuticals, the company developing vedolizumab, announced that the drug significantly surpassed placebo for the treatment of Crohn’s disease in the pivotal, phase III trial for that indication. The company said that it will soon report the full Crohn’s disease results.
The GEMINI I study was funded by Millennium. Dr. Feagan said he has been a consultant to Millennium and to several other drug companies. Dr. Sandborn said he has been a consultant to several drug companies but has no relationship with Millennium.
SAN DIEGO – Vedolizumab, a novel drug that selectively blocks lymphocyte trafficking to the gut, was safe and highly effective for inducing and maintaining clinical remission in patients with moderate to severe ulcerative colitis in a pivotal phase III trial.
"If the findings hold up with more detailed analysis, it looks like we’ll have the efficacy of a biologic agent without some of the toxicity issues that we’ve seen with anti-TNF [tumor necrosis factor] drugs," said Dr. William J. Sandborn, professor of medicine and chief of gastroenterology at the University of California, San Diego.
"This potentially could be a first-line drug," for treating moderate to severe ulcerative colitis, said Dr. Sandborn, a coinvestigator in the study.
"Vedolizumab was more effective than placebo for induction and maintenance treatment, including both anti-TNF–exposed and naive patients," Dr. Brian G. Feagan, the study’s lead investigator, said at the annual Digestive Disease Week. In addition, there was little difference between the vedolizumab and placebo groups in terms of adverse events, serious adverse events, and serious infections, said Dr. Feagan, professor of medicine at Western University in London, Ont.
At the end of a year of maintenance treatment, patients kept on the more frequent vedolizumab dosage tested, 300 mg delivered intravenously once every 4 weeks, showed a potent efficacy effect, surpassing the placebo group in corticosteroid-free clinical remissions by 31 percentage points (45% vs. 14%). "Nothing else is that good," Dr. Sandborn said in an interview. "Steroid-free remission with a delta over placebo of 30% is very impressive, especially when you factor in that many of the patients had failed anti-TNF treatment.
"We thought that vedolizumab would be safer than systemic immunosuppression, and I think the data are consistent with that. This will be a first-line treatment," Dr. Feagan said.
Equally important, total worldwide experience with vedolizumab, which now includes about 2,500 patients, has not yet resulted in a single case of progressive multifocal leukoencephalopathy (PML), an adverse effect produced by vedolizumab’s cousin drug, natalizumab (Tysarbi), ’approved for U.S. marketing to treat multiple sclerosis and Crohns disease.
Vedolizumab is a humanized monoclonal antibody that specifically binds to the alpha-4 beta-7 integrin protein that helps move leukocytes into the gut. Natalizumab is a less specific integrin antagonist that affects the protein’s actions and immune-cell trafficking to a variety of body sites, including the brain. Natalizumab "essentially blocks immune surveillance in the brain, and that allows for the JC virus [carried by about 50% of people] to cause PML," explained Dr. Sandborn. In contrast, vedolizumab does not cause "a systemic blockade of lymphocytes trafficking; it only affects a small fraction of lymphocytes, and leaves the brain completely unaffected."
Patients most at risk for PML are those who previously received immunosuppressive treatment with a drug such as azathioprine, methotrexate, or an anti-TNF drug. Patients with that history who received natalizumab for 1-2 years have about a 1 in 500-600 risk for PML, and those who got natalizumab for more than 2 years have a 1% risk.
By comparison, vedolizomab’s clean record based on 2,500 recipients "looks like it might have a very nice safety profile. If people can get comfortable with vedolizumab being different from natalizumab, then it has the potential to be first-line therapy for patients who have the worst prognosis," those who don’t respond to treatment with mesalamine.
The GEMINI I trial enrolled patients at 105 international sites with ulcerative colitis who had a Mayo score of at least 6 and an endoscopic subscore of at least 2 (indicating moderate disease) despite standard treatments. Patients were an average age of 40 years, their average duration of disease was 7 years, and their average Mayo score at entry was 8.5. Roughly 40% had previously received an anti-TNF treatment, about a third had failed on an anti-TNF drug, and just over half of the patients entered the study on a corticosteroid.
The trial included an induction phase that randomized 225 patients to a 6-week regimen with vedolizumab infusions and 149 patients to placebo. The researchers started another 521 ulcerative colitis patients on an open-label induction regimen, and then randomized 373 patients who responded after 6 weeks to maintenance infusion with vedolizumab every 4 weeks, a vedolizumab infusion every 8 weeks, or placebo.
The primary end point of the induction phase was clinical response, defined as a drop in the Mayo score of at least 3 points and at least 30%, plus a drop in the rectal bleeding score of at least 1 point or an absolute rectal bleeding subscore of 1 point or less. Achievement of this end point occurred in 47% of patients on vedolizumab and in 26% of those on placebo, a statistically significant difference. Among patients previously treated with an anti-TNF drug, 39% had a clinical response after 6 weeks on vedolizumab compared with 21% in the placebo arm, a significant difference. Among anti-TNF–naive patients, the rates were 53% and 26%.
The primary end point of the maintenance phase was clinical remission at 52 weeks, defined as a total Mayo score of 2 or less and no subscore greater than 1 point. This end point was met by 45% of patients who received vedolizumab every 4 weeks, by 42% who received the drug every 8 weeks, and by 16% of the placebo patients. Clinical remission without corticosteroid treatment occurred in 45% of patients who got vedolizumab every 4 weeks, 31% who got the drug every 8 weeks, and 14% who took placebo. Among patients with a history of anti-TNF treatment, clinical remission after 1 year occurred in 35% of patients maintained with the drug every 4 weeks, 37% who got the drug every 8 weeks, and 5% who took placebo. A total of 209 patients remained in the study through the 52-week assessment.
On May 11, Millennium Pharmaceuticals, the company developing vedolizumab, announced that the drug significantly surpassed placebo for the treatment of Crohn’s disease in the pivotal, phase III trial for that indication. The company said that it will soon report the full Crohn’s disease results.
The GEMINI I study was funded by Millennium. Dr. Feagan said he has been a consultant to Millennium and to several other drug companies. Dr. Sandborn said he has been a consultant to several drug companies but has no relationship with Millennium.
FROM THE ANNUAL DIGESTIVE DISEASE WEEK
Major Finding: Maintenance therapy with vedolizumab produced a 45% steroid-free clinical remission rate compared with a 14% rate for patients taking placebo.
Data Source: The data came from GEMINI I, an international randomized, controlled phase III trial with 209 patients who remained in the study for 1 year.
Disclosures: The GEMINI I study was funded by Millennium Pharmaceuticals. Dr. Feagan said he has been a consultant to Millennium and to several other drug companies. Dr. Sandborn said he has been a consultant to several drug companies, but has no relationship with Millennium.
Lymphadenectomy Underused in GI Cancer Surgery
SAN DIEGO – Lymph node removal during gastrointestinal cancer surgery remains underperformed in a large proportion of patients in the United States, although the median number of resected nodes increased from 1998 to 2009.
Those are the key findings of a 10-year analysis of medical records from the National Cancer Institute’s Surveillance Epidemiology and End Results (SEER) database.
Several reports in the literature show a correlation between long-term survival and the removal of possibly metastatic lymph nodes along with the cancerous organ during surgery, Dr. Attila Dubecz explained in an interview at the annual Digestive Disease Week. There are also survival differences based on sex, race or poverty status, and differences in lymph node removal between these groups in certain cancer types, he said. "We wanted to determine if these differences are more related to cancer types therefore the type of operation, for example or to these underprivileged groups."
Using SEER data from 1998 to 2009, Dr. Dubecz of Klinikum Nürnberg (Germany) and his colleagues identified 326,243 patients with a surgically treated GI malignancy. This included 13,165 malignancies in the esophagus, 18,588 in the stomach, 7,666 in the small bowel, 232,345 in the colon, 42,338 in the rectum, and 12,141 in the pancreas.
Adequate lymphadenectomy was defined as removal of at least 15 lymph nodes for cancer of the esophagus and the stomach; at least 12 for cancer of the small bowel, colon, and rectum; and at least 15 for cancer of the pancreas. The researchers evaluated the median number of lymph nodes removed and the prevalence of adequate and/or no lymphadenectomy for each cancer type over the 10-year period. They used multivariate logistic regression analysis to identify factors predicting adequate lymphadenectomy.
Dr. Dubecz, a surgeon, reported that the median number of excised nodes improved over the 10-year period in all types of cancer: from 7 to 13 in esophageal cancer, 8 to 12 in stomach cancer, 2 to 7 in small bowel cancer, 9 to 16 in colon cancer, 8 to 13 in rectal cancer, and 7 to 13 in pancreatic cancer.
In addition, the percentage of patients with an adequate lymphadenectomy (a median of 49% for all types) steadily increased and those with zero nodes removed (a median of 6% for all types) steadily decreased in all types of cancer, "although both remained far from ideal," the researchers wrote.
By 2009, the percentage of patients with adequate lymphadenectomy was 43% for esophageal cancer, 42% for stomach cancer, 35% for small bowel cancer, 77% for colon cancer, 61% for rectal cancer and 42% for pancreatic cancer. Men, patients older than age 65, or those undergoing surgical therapy earlier in the study period and living in areas with high poverty rates were significantly less likely to receive adequate lymphadenectomy (P less than .0001 for all groups).
"The main surprise was that race was an insignificant factor, and gender, age, and socioeconomic differences between the groups with adequate versus inadequate lymph node dissection were also much less [than] between the groups of different cancer types," Dr. Dubecz said at the annual meeting of the Digestive Disease Week.
Dr. Dubecz acknowledged certain limitations of the study, including the potential for misclassification of patient information in the SEER database. "Furthermore, despite being advocated by several practice organizations and consensus panels, the definitions of adequate lymphadenectomy used in this study are not universally accepted," he noted. "Third, our analyses are limited to the available variables in the SEER database with no information regarding patient insurance status, comorbidities, body mass index, or [neo]adjuvant chemotherapy, which could influence lymph node dissection and the disparities."
Dr. Dubecz said he had no relevant financial disclosures.
SAN DIEGO – Lymph node removal during gastrointestinal cancer surgery remains underperformed in a large proportion of patients in the United States, although the median number of resected nodes increased from 1998 to 2009.
Those are the key findings of a 10-year analysis of medical records from the National Cancer Institute’s Surveillance Epidemiology and End Results (SEER) database.
Several reports in the literature show a correlation between long-term survival and the removal of possibly metastatic lymph nodes along with the cancerous organ during surgery, Dr. Attila Dubecz explained in an interview at the annual Digestive Disease Week. There are also survival differences based on sex, race or poverty status, and differences in lymph node removal between these groups in certain cancer types, he said. "We wanted to determine if these differences are more related to cancer types therefore the type of operation, for example or to these underprivileged groups."
Using SEER data from 1998 to 2009, Dr. Dubecz of Klinikum Nürnberg (Germany) and his colleagues identified 326,243 patients with a surgically treated GI malignancy. This included 13,165 malignancies in the esophagus, 18,588 in the stomach, 7,666 in the small bowel, 232,345 in the colon, 42,338 in the rectum, and 12,141 in the pancreas.
Adequate lymphadenectomy was defined as removal of at least 15 lymph nodes for cancer of the esophagus and the stomach; at least 12 for cancer of the small bowel, colon, and rectum; and at least 15 for cancer of the pancreas. The researchers evaluated the median number of lymph nodes removed and the prevalence of adequate and/or no lymphadenectomy for each cancer type over the 10-year period. They used multivariate logistic regression analysis to identify factors predicting adequate lymphadenectomy.
Dr. Dubecz, a surgeon, reported that the median number of excised nodes improved over the 10-year period in all types of cancer: from 7 to 13 in esophageal cancer, 8 to 12 in stomach cancer, 2 to 7 in small bowel cancer, 9 to 16 in colon cancer, 8 to 13 in rectal cancer, and 7 to 13 in pancreatic cancer.
In addition, the percentage of patients with an adequate lymphadenectomy (a median of 49% for all types) steadily increased and those with zero nodes removed (a median of 6% for all types) steadily decreased in all types of cancer, "although both remained far from ideal," the researchers wrote.
By 2009, the percentage of patients with adequate lymphadenectomy was 43% for esophageal cancer, 42% for stomach cancer, 35% for small bowel cancer, 77% for colon cancer, 61% for rectal cancer and 42% for pancreatic cancer. Men, patients older than age 65, or those undergoing surgical therapy earlier in the study period and living in areas with high poverty rates were significantly less likely to receive adequate lymphadenectomy (P less than .0001 for all groups).
"The main surprise was that race was an insignificant factor, and gender, age, and socioeconomic differences between the groups with adequate versus inadequate lymph node dissection were also much less [than] between the groups of different cancer types," Dr. Dubecz said at the annual meeting of the Digestive Disease Week.
Dr. Dubecz acknowledged certain limitations of the study, including the potential for misclassification of patient information in the SEER database. "Furthermore, despite being advocated by several practice organizations and consensus panels, the definitions of adequate lymphadenectomy used in this study are not universally accepted," he noted. "Third, our analyses are limited to the available variables in the SEER database with no information regarding patient insurance status, comorbidities, body mass index, or [neo]adjuvant chemotherapy, which could influence lymph node dissection and the disparities."
Dr. Dubecz said he had no relevant financial disclosures.
SAN DIEGO – Lymph node removal during gastrointestinal cancer surgery remains underperformed in a large proportion of patients in the United States, although the median number of resected nodes increased from 1998 to 2009.
Those are the key findings of a 10-year analysis of medical records from the National Cancer Institute’s Surveillance Epidemiology and End Results (SEER) database.
Several reports in the literature show a correlation between long-term survival and the removal of possibly metastatic lymph nodes along with the cancerous organ during surgery, Dr. Attila Dubecz explained in an interview at the annual Digestive Disease Week. There are also survival differences based on sex, race or poverty status, and differences in lymph node removal between these groups in certain cancer types, he said. "We wanted to determine if these differences are more related to cancer types therefore the type of operation, for example or to these underprivileged groups."
Using SEER data from 1998 to 2009, Dr. Dubecz of Klinikum Nürnberg (Germany) and his colleagues identified 326,243 patients with a surgically treated GI malignancy. This included 13,165 malignancies in the esophagus, 18,588 in the stomach, 7,666 in the small bowel, 232,345 in the colon, 42,338 in the rectum, and 12,141 in the pancreas.
Adequate lymphadenectomy was defined as removal of at least 15 lymph nodes for cancer of the esophagus and the stomach; at least 12 for cancer of the small bowel, colon, and rectum; and at least 15 for cancer of the pancreas. The researchers evaluated the median number of lymph nodes removed and the prevalence of adequate and/or no lymphadenectomy for each cancer type over the 10-year period. They used multivariate logistic regression analysis to identify factors predicting adequate lymphadenectomy.
Dr. Dubecz, a surgeon, reported that the median number of excised nodes improved over the 10-year period in all types of cancer: from 7 to 13 in esophageal cancer, 8 to 12 in stomach cancer, 2 to 7 in small bowel cancer, 9 to 16 in colon cancer, 8 to 13 in rectal cancer, and 7 to 13 in pancreatic cancer.
In addition, the percentage of patients with an adequate lymphadenectomy (a median of 49% for all types) steadily increased and those with zero nodes removed (a median of 6% for all types) steadily decreased in all types of cancer, "although both remained far from ideal," the researchers wrote.
By 2009, the percentage of patients with adequate lymphadenectomy was 43% for esophageal cancer, 42% for stomach cancer, 35% for small bowel cancer, 77% for colon cancer, 61% for rectal cancer and 42% for pancreatic cancer. Men, patients older than age 65, or those undergoing surgical therapy earlier in the study period and living in areas with high poverty rates were significantly less likely to receive adequate lymphadenectomy (P less than .0001 for all groups).
"The main surprise was that race was an insignificant factor, and gender, age, and socioeconomic differences between the groups with adequate versus inadequate lymph node dissection were also much less [than] between the groups of different cancer types," Dr. Dubecz said at the annual meeting of the Digestive Disease Week.
Dr. Dubecz acknowledged certain limitations of the study, including the potential for misclassification of patient information in the SEER database. "Furthermore, despite being advocated by several practice organizations and consensus panels, the definitions of adequate lymphadenectomy used in this study are not universally accepted," he noted. "Third, our analyses are limited to the available variables in the SEER database with no information regarding patient insurance status, comorbidities, body mass index, or [neo]adjuvant chemotherapy, which could influence lymph node dissection and the disparities."
Dr. Dubecz said he had no relevant financial disclosures.
FROM THE ANNUAL DIGESTIVE DISEASE WEEK
Major Finding: By 2009, the percentage of patients with adequate lymphadenectomy during surgery for gastrointestinal cancer was 43% for esophageal cancer, 42% for stomach cancer, 35% for small bowel cancer, 77% for colon cancer, 61% for rectal cancer, and 42% for pancreatic cancer.
Data Source: Findings are based on a 10-year analysis of medical records from 326,243 patients in the National Cancer Institute’s Surveillance Epidemiology and End Results (SEER) database.
Disclosures: Dr. Dubecz said he had no relevant financial disclosures.