User login
Crohn's Responded to Ustekinumab
Ustekinumab induced a clinical response in patients with moderate to severe Crohn’s disease that was resistant to tumor necrosis factor antagonists, in a phase IIb clinical trial published online Oct. 17 in the New England Journal of Medicine.
However, the agent did not improve remission rates, compared with placebo, said Dr. William J. Sandborn, AGAF, who is professor of medicine and chief of the division of gastroenterology at the University of California San Diego, La Jolla, and his associates.
"A sizable proportion" of patients with moderate to severe Crohn’s disease do not respond to TNF antagonists, have an unsustained response, or must discontinue the medications because of adverse effects. After ustekinumab showed efficacy in such patients in a phase IIa clinical study, Dr. Sandborn and his colleagues performed a 36-week double-blind phase IIb trial in 526 adults at 153 medical centers in 12 countries.
Ustekinumab, a human IgG monoclonal antibody that inhibits the receptors for interleukin-12 and interleukin-23 on T cells, natural killer cells, and antigen-presenting cells, has Food and Drug Administration approval for use in plaque psoriasis. This clinical trial was sponsored by an affiliate of the manufacturer, Janssen Biotech.
During an 8-week induction phase, the study subjects were randomly assigned to receive intravenous placebo (132 patients) or ustekinumab in 1-mg/kg (131 patients), 3-mg/kg (132 patients), or 6-mg/kg (131 patients) doses. Then, during weeks 8-36, the study subjects who showed a response to induction therapy and those who did not show a response were separately randomized to receive either subcutaneous ustekinumab (90 mg) or placebo at week 8 and week 16, as maintenance therapy.
Treatment efficacy was assessed at week 22, and patients were followed through week 36 for a safety analysis. A total of 36.1% of the subjects discontinued the study before week 36.
The primary end point was a clinical response, defined as a decrease of 100 points or more on the Crohn’s Disease Activity Index (CDAI) score.
A total of 39.7% of patients receiving the 6-mg induction dose showed a clinical response, which was significantly greater than the 23.5% of patients receiving placebo, the investigators said (N. Engl. J. Med. 2012 [doi:10.1056/NEJMoa1203572]).
A greater number of patients receiving the lower doses of ustekinumab than receiving placebo showed a clinical response, but the differences between these low-dose groups and the placebo group did not reach statistical significance.
The 6-mg/kg dose was effective across most demographic and disease characteristics, judging from the findings of a subgroup analysis. It was consistently effective in patients who had failed on their first attempt at therapy with TNF antagonists, patients who had failed on two or more TNF antagonists, and patients who had only a transient response to TNF antagonists.
However, rates of clinical remission did not differ significantly between patients receiving ustekinumab and those receiving placebo, Dr. Sandborn and his associates said.
At all follow-up visits, the proportion of patients who had a 70-point clinical response was significantly higher, the reductions in mean CDAI scores were significantly greater, and the reductions in C-reactive protein levels were significantly greater in patients receiving 6 mg per kg of ustekinumab than in the placebo group.
As a maintenance therapy, 90 mg of subcutaneous ustekinumab appeared to be effective in patients who responded to the induction dose of the agent. The proportion of patients who showed a clinical response at week 22 was 69.4% in those receiving maintenance ustekinumab, significantly greater than the 42.5% response rate among those receiving maintenance placebo.
Among patients who responded to induction-phase ustekinumab, 41.7% of those who also received maintenance ustekinumab achieved clinical remission at week 22, compared with only 27.4% of those who received maintenance placebo.
Similarly, among patients who showed a response to induction ustekinumab, reductions in both CDAI scores and CRP levels were sustained if they continued on maintenance ustekinumab but were not sustained if they continued on placebo for the maintenance period.
However, patients who did not show a response to induction ustekinumab also did not benefit from additional ustekinumab in the maintenance phase of the study.
The results of the safety analysis were "somewhat limited" by the small sample size and the short duration of treatment. No deaths, serious opportunistic infections, or major adverse cardiovascular events were reported, "but large studies of longer duration are needed to assess uncommon adverse events," the investigators said.
Of note, one patient receiving ustekinumab as both induction and maintenance therapy developed a basal cell carcinoma. Among patients taking ustekinumab in the induction phase of the study, six developed serious infections: Clostridium difficile, viral gastroenteritis, UTI, anal abscess, vaginal abscess, and a staph infection of a central catheter.
Ustekinumab induced a clinical response in patients with moderate to severe Crohn’s disease that was resistant to tumor necrosis factor antagonists, in a phase IIb clinical trial published online Oct. 17 in the New England Journal of Medicine.
However, the agent did not improve remission rates, compared with placebo, said Dr. William J. Sandborn, AGAF, who is professor of medicine and chief of the division of gastroenterology at the University of California San Diego, La Jolla, and his associates.
"A sizable proportion" of patients with moderate to severe Crohn’s disease do not respond to TNF antagonists, have an unsustained response, or must discontinue the medications because of adverse effects. After ustekinumab showed efficacy in such patients in a phase IIa clinical study, Dr. Sandborn and his colleagues performed a 36-week double-blind phase IIb trial in 526 adults at 153 medical centers in 12 countries.
Ustekinumab, a human IgG monoclonal antibody that inhibits the receptors for interleukin-12 and interleukin-23 on T cells, natural killer cells, and antigen-presenting cells, has Food and Drug Administration approval for use in plaque psoriasis. This clinical trial was sponsored by an affiliate of the manufacturer, Janssen Biotech.
During an 8-week induction phase, the study subjects were randomly assigned to receive intravenous placebo (132 patients) or ustekinumab in 1-mg/kg (131 patients), 3-mg/kg (132 patients), or 6-mg/kg (131 patients) doses. Then, during weeks 8-36, the study subjects who showed a response to induction therapy and those who did not show a response were separately randomized to receive either subcutaneous ustekinumab (90 mg) or placebo at week 8 and week 16, as maintenance therapy.
Treatment efficacy was assessed at week 22, and patients were followed through week 36 for a safety analysis. A total of 36.1% of the subjects discontinued the study before week 36.
The primary end point was a clinical response, defined as a decrease of 100 points or more on the Crohn’s Disease Activity Index (CDAI) score.
A total of 39.7% of patients receiving the 6-mg induction dose showed a clinical response, which was significantly greater than the 23.5% of patients receiving placebo, the investigators said (N. Engl. J. Med. 2012 [doi:10.1056/NEJMoa1203572]).
A greater number of patients receiving the lower doses of ustekinumab than receiving placebo showed a clinical response, but the differences between these low-dose groups and the placebo group did not reach statistical significance.
The 6-mg/kg dose was effective across most demographic and disease characteristics, judging from the findings of a subgroup analysis. It was consistently effective in patients who had failed on their first attempt at therapy with TNF antagonists, patients who had failed on two or more TNF antagonists, and patients who had only a transient response to TNF antagonists.
However, rates of clinical remission did not differ significantly between patients receiving ustekinumab and those receiving placebo, Dr. Sandborn and his associates said.
At all follow-up visits, the proportion of patients who had a 70-point clinical response was significantly higher, the reductions in mean CDAI scores were significantly greater, and the reductions in C-reactive protein levels were significantly greater in patients receiving 6 mg per kg of ustekinumab than in the placebo group.
As a maintenance therapy, 90 mg of subcutaneous ustekinumab appeared to be effective in patients who responded to the induction dose of the agent. The proportion of patients who showed a clinical response at week 22 was 69.4% in those receiving maintenance ustekinumab, significantly greater than the 42.5% response rate among those receiving maintenance placebo.
Among patients who responded to induction-phase ustekinumab, 41.7% of those who also received maintenance ustekinumab achieved clinical remission at week 22, compared with only 27.4% of those who received maintenance placebo.
Similarly, among patients who showed a response to induction ustekinumab, reductions in both CDAI scores and CRP levels were sustained if they continued on maintenance ustekinumab but were not sustained if they continued on placebo for the maintenance period.
However, patients who did not show a response to induction ustekinumab also did not benefit from additional ustekinumab in the maintenance phase of the study.
The results of the safety analysis were "somewhat limited" by the small sample size and the short duration of treatment. No deaths, serious opportunistic infections, or major adverse cardiovascular events were reported, "but large studies of longer duration are needed to assess uncommon adverse events," the investigators said.
Of note, one patient receiving ustekinumab as both induction and maintenance therapy developed a basal cell carcinoma. Among patients taking ustekinumab in the induction phase of the study, six developed serious infections: Clostridium difficile, viral gastroenteritis, UTI, anal abscess, vaginal abscess, and a staph infection of a central catheter.
Ustekinumab induced a clinical response in patients with moderate to severe Crohn’s disease that was resistant to tumor necrosis factor antagonists, in a phase IIb clinical trial published online Oct. 17 in the New England Journal of Medicine.
However, the agent did not improve remission rates, compared with placebo, said Dr. William J. Sandborn, AGAF, who is professor of medicine and chief of the division of gastroenterology at the University of California San Diego, La Jolla, and his associates.
"A sizable proportion" of patients with moderate to severe Crohn’s disease do not respond to TNF antagonists, have an unsustained response, or must discontinue the medications because of adverse effects. After ustekinumab showed efficacy in such patients in a phase IIa clinical study, Dr. Sandborn and his colleagues performed a 36-week double-blind phase IIb trial in 526 adults at 153 medical centers in 12 countries.
Ustekinumab, a human IgG monoclonal antibody that inhibits the receptors for interleukin-12 and interleukin-23 on T cells, natural killer cells, and antigen-presenting cells, has Food and Drug Administration approval for use in plaque psoriasis. This clinical trial was sponsored by an affiliate of the manufacturer, Janssen Biotech.
During an 8-week induction phase, the study subjects were randomly assigned to receive intravenous placebo (132 patients) or ustekinumab in 1-mg/kg (131 patients), 3-mg/kg (132 patients), or 6-mg/kg (131 patients) doses. Then, during weeks 8-36, the study subjects who showed a response to induction therapy and those who did not show a response were separately randomized to receive either subcutaneous ustekinumab (90 mg) or placebo at week 8 and week 16, as maintenance therapy.
Treatment efficacy was assessed at week 22, and patients were followed through week 36 for a safety analysis. A total of 36.1% of the subjects discontinued the study before week 36.
The primary end point was a clinical response, defined as a decrease of 100 points or more on the Crohn’s Disease Activity Index (CDAI) score.
A total of 39.7% of patients receiving the 6-mg induction dose showed a clinical response, which was significantly greater than the 23.5% of patients receiving placebo, the investigators said (N. Engl. J. Med. 2012 [doi:10.1056/NEJMoa1203572]).
A greater number of patients receiving the lower doses of ustekinumab than receiving placebo showed a clinical response, but the differences between these low-dose groups and the placebo group did not reach statistical significance.
The 6-mg/kg dose was effective across most demographic and disease characteristics, judging from the findings of a subgroup analysis. It was consistently effective in patients who had failed on their first attempt at therapy with TNF antagonists, patients who had failed on two or more TNF antagonists, and patients who had only a transient response to TNF antagonists.
However, rates of clinical remission did not differ significantly between patients receiving ustekinumab and those receiving placebo, Dr. Sandborn and his associates said.
At all follow-up visits, the proportion of patients who had a 70-point clinical response was significantly higher, the reductions in mean CDAI scores were significantly greater, and the reductions in C-reactive protein levels were significantly greater in patients receiving 6 mg per kg of ustekinumab than in the placebo group.
As a maintenance therapy, 90 mg of subcutaneous ustekinumab appeared to be effective in patients who responded to the induction dose of the agent. The proportion of patients who showed a clinical response at week 22 was 69.4% in those receiving maintenance ustekinumab, significantly greater than the 42.5% response rate among those receiving maintenance placebo.
Among patients who responded to induction-phase ustekinumab, 41.7% of those who also received maintenance ustekinumab achieved clinical remission at week 22, compared with only 27.4% of those who received maintenance placebo.
Similarly, among patients who showed a response to induction ustekinumab, reductions in both CDAI scores and CRP levels were sustained if they continued on maintenance ustekinumab but were not sustained if they continued on placebo for the maintenance period.
However, patients who did not show a response to induction ustekinumab also did not benefit from additional ustekinumab in the maintenance phase of the study.
The results of the safety analysis were "somewhat limited" by the small sample size and the short duration of treatment. No deaths, serious opportunistic infections, or major adverse cardiovascular events were reported, "but large studies of longer duration are needed to assess uncommon adverse events," the investigators said.
Of note, one patient receiving ustekinumab as both induction and maintenance therapy developed a basal cell carcinoma. Among patients taking ustekinumab in the induction phase of the study, six developed serious infections: Clostridium difficile, viral gastroenteritis, UTI, anal abscess, vaginal abscess, and a staph infection of a central catheter.
Major Finding: Of patients with moderate to severe Crohn's disease who received ustekinumab (6 mg/kg), 39.7% showed a decrease of 100 points or more in CDAI score, compared with 23.5% of those who received placebo.
Data Source: The data come from a 36-week international phase IIb randomized clinical trial comparing 3 doses of ustekinumab with placebo in 526 adults who had refractory Crohn’ disease.
Disclosures: This study was sponsored by Janssen Research and Development; Janssen Biotech makes ustekinumab. Dr. Sandborn and his associates reported numerous ties to industry sources.
Panel Advises Approving Short Bowel Syndrome Drug
SILVER SPRING, MD. – A Food and Drug Administration advisory panel unanimously supported the approval of teduglutide, a recombinant analogue of human glucagon-like peptide-2 (GLP-2), as a treatment to improve the intestinal absorption of fluid and nutrients in adults with short bowel syndrome, with recommendations to follow the drug’s long-term safety, at a meeting on Oct. 16.
At a meeting in September, the FDA’s Gastrointestinal Drugs Advisory Committee voted 12 to 0 that the benefits of teduglutide outweighed the potential risks in patients with short bowel syndrome (SBS). In phase III studies of 169 adults with SBS, whose estimated small bowel length was a mean of 72 cm and who had been on parenteral nutrition and IV therapy for a mean of 7 years, teduglutide significantly reduced the volume of parenteral nutrition and IV fluids they needed after 6 months of treatment, compared with placebo.
While the panelists unanimously agreed that the results represented a clinically meaningful benefit for these patients and generally felt comfortable with the drug’s safety profile, they recommended that more safety data are needed, including determining whether the risk of colorectal cancer is increased with treatment.
Produced in the distal small intestine and proximal large intestine, GLP-2 is "an intestinotrophic peptide that stimulates mucosal epithelium," increasing absorption of fluids and nutrients, according to the manufacturer, NPS Pharmaceuticals. In studies, patients treated with teduglutide, administered subcutaneously once a week, had evidence of increased villus height after 21 days of treatment, according to the company.
In the main phase III study of 86 adults with SBS, 63% of those treated with 0.05 mg/kg daily had at least a 20% reduction in the volume of parenteral nutrition and IV fluids (the primary end point) required after 24 weeks of treatment, compared with 30% of those on placebo; this was a statistically significant difference. The volume of parenteral nutrition and IV fluids from baseline to the 24th week of treatment was reduced by a median of 4.4 L/week among patients on teduglutide, compared with 2.3 L/week for patients on placebo. Teduglutide treatment also led to a higher percentage of patients with at least one day less on therapy than did placebo (54% vs. 23%). An extension study indicated that this effect was maintained through 1 year of treatment.
While the FDA reviewers agreed that the drug had clinically meaningful effects in the studies, they raised some safety issues, mainly potential tumor-promoting effects, as well as potential GI side effects that included biliary and pancreatic disease and GI stenosis and obstruction. To date, there have been no reports of small bowel malignancies in treated patients; the three malignancies reported in treated patients have been a metastatic adenocarcinoma in a patient who had been on treatment for almost a year and lung cancer in two patients who had a history of smoking. Adenomas were seen in the bile duct and jejunum of rats at 700 times the human dose, findings that are "consistent with the pharmacological effects of the drug," according to the FDA.
There was also a low incidence of intestinal obstruction and stenosis, cholecystitis, and pancreatic disease in treated patients, compared with no cases among those on placebo.
NPS has proposed a registry of treated patients to evaluate the long-term safety and effectiveness of teduglutide, and a risk evaluation and mitigation strategy (REMS) aimed at educating prescribers about the potential and known risks of treatment. The REMS would include a letter to health care professionals and professional societies, including the American Gastroenterological Association and the American College of Gastroenterology; and drug labeling that includes contraindications in patients with a history of malignancy or with active or currently suspected malignancy, as well as warnings and precautions about the possible acceleration of neoplastic growth, colorectal polyps, and small bowel neoplasia.
The panel voted 10 to 1, with one abstention, that the company’s plans were adequate for addressing the safety concerns, although they pointed out that the number of patients in the studies was small and recommended close follow-up of treated patients.
"It seems like it’s a fairly safe drug," but follow-up is short term and fewer than 200 patients have been treated with teduglutide, said panelist Dr. Kevin Kelly, director of the division of solid tumor oncology at Thomas Jefferson University, Philadelphia. He and others recommended that patients be followed for up to 10 years.
"My gut feeling is that this is probably a very safe drug" that does not increase the risk of carcinogenesis, said another panelist, Dr. Ronald Fogel of the Digestive Health Center of Michigan in Chesterfield. He recommended more aggressive follow-up of treated patients than was proposed by the company, which he said could include serial colonoscopies at 2-year intervals with multiple biopsies at areas of dysplasia.
If teduglutide is approved, the company will market it as Gattex. About 10,000-15,000 adults in the United States with SBS are dependent on parenteral nutrition and IV fluids for fluid and nutrient replacement, according to NPS. Teduglutide was recently approved in Europe for the same indication. The FDA is expected to make a decision on approval by Dec. 30; if the drug is approved, the company plans to pursue studies in pediatric patients with SBS.
The two drugs currently approved by the FDA for people with SBS who are dependent on parenteral nutrition are somatropin rhGH (Zorbtive), a growth hormone approved in 2003, and L-glutamine powder for oral solution (Nutrestore), an adjunctive treatment approved in 2004.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – A Food and Drug Administration advisory panel unanimously supported the approval of teduglutide, a recombinant analogue of human glucagon-like peptide-2 (GLP-2), as a treatment to improve the intestinal absorption of fluid and nutrients in adults with short bowel syndrome, with recommendations to follow the drug’s long-term safety, at a meeting on Oct. 16.
At a meeting in September, the FDA’s Gastrointestinal Drugs Advisory Committee voted 12 to 0 that the benefits of teduglutide outweighed the potential risks in patients with short bowel syndrome (SBS). In phase III studies of 169 adults with SBS, whose estimated small bowel length was a mean of 72 cm and who had been on parenteral nutrition and IV therapy for a mean of 7 years, teduglutide significantly reduced the volume of parenteral nutrition and IV fluids they needed after 6 months of treatment, compared with placebo.
While the panelists unanimously agreed that the results represented a clinically meaningful benefit for these patients and generally felt comfortable with the drug’s safety profile, they recommended that more safety data are needed, including determining whether the risk of colorectal cancer is increased with treatment.
Produced in the distal small intestine and proximal large intestine, GLP-2 is "an intestinotrophic peptide that stimulates mucosal epithelium," increasing absorption of fluids and nutrients, according to the manufacturer, NPS Pharmaceuticals. In studies, patients treated with teduglutide, administered subcutaneously once a week, had evidence of increased villus height after 21 days of treatment, according to the company.
In the main phase III study of 86 adults with SBS, 63% of those treated with 0.05 mg/kg daily had at least a 20% reduction in the volume of parenteral nutrition and IV fluids (the primary end point) required after 24 weeks of treatment, compared with 30% of those on placebo; this was a statistically significant difference. The volume of parenteral nutrition and IV fluids from baseline to the 24th week of treatment was reduced by a median of 4.4 L/week among patients on teduglutide, compared with 2.3 L/week for patients on placebo. Teduglutide treatment also led to a higher percentage of patients with at least one day less on therapy than did placebo (54% vs. 23%). An extension study indicated that this effect was maintained through 1 year of treatment.
While the FDA reviewers agreed that the drug had clinically meaningful effects in the studies, they raised some safety issues, mainly potential tumor-promoting effects, as well as potential GI side effects that included biliary and pancreatic disease and GI stenosis and obstruction. To date, there have been no reports of small bowel malignancies in treated patients; the three malignancies reported in treated patients have been a metastatic adenocarcinoma in a patient who had been on treatment for almost a year and lung cancer in two patients who had a history of smoking. Adenomas were seen in the bile duct and jejunum of rats at 700 times the human dose, findings that are "consistent with the pharmacological effects of the drug," according to the FDA.
There was also a low incidence of intestinal obstruction and stenosis, cholecystitis, and pancreatic disease in treated patients, compared with no cases among those on placebo.
NPS has proposed a registry of treated patients to evaluate the long-term safety and effectiveness of teduglutide, and a risk evaluation and mitigation strategy (REMS) aimed at educating prescribers about the potential and known risks of treatment. The REMS would include a letter to health care professionals and professional societies, including the American Gastroenterological Association and the American College of Gastroenterology; and drug labeling that includes contraindications in patients with a history of malignancy or with active or currently suspected malignancy, as well as warnings and precautions about the possible acceleration of neoplastic growth, colorectal polyps, and small bowel neoplasia.
The panel voted 10 to 1, with one abstention, that the company’s plans were adequate for addressing the safety concerns, although they pointed out that the number of patients in the studies was small and recommended close follow-up of treated patients.
"It seems like it’s a fairly safe drug," but follow-up is short term and fewer than 200 patients have been treated with teduglutide, said panelist Dr. Kevin Kelly, director of the division of solid tumor oncology at Thomas Jefferson University, Philadelphia. He and others recommended that patients be followed for up to 10 years.
"My gut feeling is that this is probably a very safe drug" that does not increase the risk of carcinogenesis, said another panelist, Dr. Ronald Fogel of the Digestive Health Center of Michigan in Chesterfield. He recommended more aggressive follow-up of treated patients than was proposed by the company, which he said could include serial colonoscopies at 2-year intervals with multiple biopsies at areas of dysplasia.
If teduglutide is approved, the company will market it as Gattex. About 10,000-15,000 adults in the United States with SBS are dependent on parenteral nutrition and IV fluids for fluid and nutrient replacement, according to NPS. Teduglutide was recently approved in Europe for the same indication. The FDA is expected to make a decision on approval by Dec. 30; if the drug is approved, the company plans to pursue studies in pediatric patients with SBS.
The two drugs currently approved by the FDA for people with SBS who are dependent on parenteral nutrition are somatropin rhGH (Zorbtive), a growth hormone approved in 2003, and L-glutamine powder for oral solution (Nutrestore), an adjunctive treatment approved in 2004.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – A Food and Drug Administration advisory panel unanimously supported the approval of teduglutide, a recombinant analogue of human glucagon-like peptide-2 (GLP-2), as a treatment to improve the intestinal absorption of fluid and nutrients in adults with short bowel syndrome, with recommendations to follow the drug’s long-term safety, at a meeting on Oct. 16.
At a meeting in September, the FDA’s Gastrointestinal Drugs Advisory Committee voted 12 to 0 that the benefits of teduglutide outweighed the potential risks in patients with short bowel syndrome (SBS). In phase III studies of 169 adults with SBS, whose estimated small bowel length was a mean of 72 cm and who had been on parenteral nutrition and IV therapy for a mean of 7 years, teduglutide significantly reduced the volume of parenteral nutrition and IV fluids they needed after 6 months of treatment, compared with placebo.
While the panelists unanimously agreed that the results represented a clinically meaningful benefit for these patients and generally felt comfortable with the drug’s safety profile, they recommended that more safety data are needed, including determining whether the risk of colorectal cancer is increased with treatment.
Produced in the distal small intestine and proximal large intestine, GLP-2 is "an intestinotrophic peptide that stimulates mucosal epithelium," increasing absorption of fluids and nutrients, according to the manufacturer, NPS Pharmaceuticals. In studies, patients treated with teduglutide, administered subcutaneously once a week, had evidence of increased villus height after 21 days of treatment, according to the company.
In the main phase III study of 86 adults with SBS, 63% of those treated with 0.05 mg/kg daily had at least a 20% reduction in the volume of parenteral nutrition and IV fluids (the primary end point) required after 24 weeks of treatment, compared with 30% of those on placebo; this was a statistically significant difference. The volume of parenteral nutrition and IV fluids from baseline to the 24th week of treatment was reduced by a median of 4.4 L/week among patients on teduglutide, compared with 2.3 L/week for patients on placebo. Teduglutide treatment also led to a higher percentage of patients with at least one day less on therapy than did placebo (54% vs. 23%). An extension study indicated that this effect was maintained through 1 year of treatment.
While the FDA reviewers agreed that the drug had clinically meaningful effects in the studies, they raised some safety issues, mainly potential tumor-promoting effects, as well as potential GI side effects that included biliary and pancreatic disease and GI stenosis and obstruction. To date, there have been no reports of small bowel malignancies in treated patients; the three malignancies reported in treated patients have been a metastatic adenocarcinoma in a patient who had been on treatment for almost a year and lung cancer in two patients who had a history of smoking. Adenomas were seen in the bile duct and jejunum of rats at 700 times the human dose, findings that are "consistent with the pharmacological effects of the drug," according to the FDA.
There was also a low incidence of intestinal obstruction and stenosis, cholecystitis, and pancreatic disease in treated patients, compared with no cases among those on placebo.
NPS has proposed a registry of treated patients to evaluate the long-term safety and effectiveness of teduglutide, and a risk evaluation and mitigation strategy (REMS) aimed at educating prescribers about the potential and known risks of treatment. The REMS would include a letter to health care professionals and professional societies, including the American Gastroenterological Association and the American College of Gastroenterology; and drug labeling that includes contraindications in patients with a history of malignancy or with active or currently suspected malignancy, as well as warnings and precautions about the possible acceleration of neoplastic growth, colorectal polyps, and small bowel neoplasia.
The panel voted 10 to 1, with one abstention, that the company’s plans were adequate for addressing the safety concerns, although they pointed out that the number of patients in the studies was small and recommended close follow-up of treated patients.
"It seems like it’s a fairly safe drug," but follow-up is short term and fewer than 200 patients have been treated with teduglutide, said panelist Dr. Kevin Kelly, director of the division of solid tumor oncology at Thomas Jefferson University, Philadelphia. He and others recommended that patients be followed for up to 10 years.
"My gut feeling is that this is probably a very safe drug" that does not increase the risk of carcinogenesis, said another panelist, Dr. Ronald Fogel of the Digestive Health Center of Michigan in Chesterfield. He recommended more aggressive follow-up of treated patients than was proposed by the company, which he said could include serial colonoscopies at 2-year intervals with multiple biopsies at areas of dysplasia.
If teduglutide is approved, the company will market it as Gattex. About 10,000-15,000 adults in the United States with SBS are dependent on parenteral nutrition and IV fluids for fluid and nutrient replacement, according to NPS. Teduglutide was recently approved in Europe for the same indication. The FDA is expected to make a decision on approval by Dec. 30; if the drug is approved, the company plans to pursue studies in pediatric patients with SBS.
The two drugs currently approved by the FDA for people with SBS who are dependent on parenteral nutrition are somatropin rhGH (Zorbtive), a growth hormone approved in 2003, and L-glutamine powder for oral solution (Nutrestore), an adjunctive treatment approved in 2004.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
Anti-TNF Resistant Crohn's Disease May Respond to Ustekinumab
Ustekinumab induced a clinical response in patients with moderate to severe Crohn’s disease that was resistant to tumor necrosis factor antagonists, in a phase IIb clinical trial published online Oct. 17 in the New England Journal of Medicine.
However, the agent did not improve remission rates, compared with placebo, said Dr. William J. Sandborn, professor of medicine and chief of the division of gastroenterology at the University of California San Diego, La Jolla, and his associates.
"A sizable proportion" of patients with moderate to severe Crohn’s disease do not respond to TNF antagonists, have an unsustained response, or must discontinue the medications because of adverse effects. After ustekinumab showed efficacy in such patients in a phase IIa clinical study, Dr. Sandborn and his colleagues performed a 36-week double-blind phase II2b trial in 526 adults at 153 medical centers in 12 countries.
Ustekinumab, a human IgG monoclonal antibody that inhibits the receptors for interleukin-12 and interleukin-23 on T cells, natural killer cells, and antigen-presenting cells, has Food and Drug Administration approval for use in plaque psoriasis. This clinical trial was sponsored by an affiliate of the manufacturer, Janssen Biotech.
During an 8-week induction phase, the study subjects were randomly assigned to receive intravenous placebo (132 patients) or ustekinumab in 1-mg/kg (131 patients), 3-mg/kg (132 patients), or 6-mg/kg (131 patients) doses. Then, during weeks 8-36, the study subjects who showed a response to induction therapy and those who did not show a response were separately randomized to receive either subcutaneous ustekinumab (90 mg) or placebo at week 8 and week 16, as maintenance therapy.
Treatment efficacy was assessed at week 22, and patients were followed through week 36 for a safety analysis. A total of 36.1% of the subjects discontinued the study before week 36.
The primary end point was a clinical response, defined as a decrease of 100 points or more on the Crohn’s Disease Activity Index (CDAI) score.
A total of 39.7% of patients receiving the 6-mg induction dose showed a clinical response, which was significantly greater than the 23.5% of patients receiving placebo, the investigators said (New Engl. J. Med. 2012 [doi:10.1056/NEJMoa1203572]).
A greater number of patients receiving the lower doses of ustekinumab than receiving placebo showed a clinical response, but the differences between these low-dose groups and the placebo group did not reach statistical significance.
The 6-mg/kg dose was effective across most demographic and disease characteristics, judging from the findings of a subgroup analysis. It was consistently effective in patients who had failed on their first attempt at therapy with TNF antagonists, patients who had failed on two or more TNF antagonists, and patients who had only had a transient response to TNF antagonists.
However, rates of clinical remission did not differ significantly between patients receiving ustekinumab and those receiving placebo, Dr. Sandborn and his associates said.
At all follow-up visits, the proportion of patients who had a 70-point clinical response was significantly higher, the reductions in mean CDAI scores were significantly greater, and the reductions in C-reactive protein levels were significantly greater in patients receiving 6 mg per kg of ustekinumab than in the placebo group.
As a maintenance therapy, 90 mg of subcutaneous ustekinumab appeared to be effective in patients who responded to the induction dose of the agent. The proportion of patients who showed a clinical response at week 22 was 69.4% in those receiving maintenance ustekinumab, significantly greater than the 42.5% response rate among those receiving maintenance placebo.
Among patients who responded to induction-phase ustekinumab, 41.7% of those who also received maintenance ustekinumab achieved clinical remission at week 22, compared with only 27.4% of those who received maintenance placebo.
Similarly, among patients who showed a response to induction ustekinumab, reductions in both CDAI scores and CRP levels were sustained if they continued on maintenance ustekinumab but were not sustained if they continued on placebo for the maintenance period.
However, patients who did not show a response to induction ustekinumab also did not benefit from additional ustekinumab in the maintenance phase of the study.
The results of the safety analysis were "somewhat limited" by the small sample size and the short duration of treatment. No deaths, serious opportunistic infections, or major adverse cardiovascular events were reported, "but large studies of longer duration are needed to assess uncommon adverse events," the investigators said.
Of note, one patient receiving ustekinumab as both induction and maintenance therapy developed a basal cell carcinoma. Among patients taking ustekinumab in the induction phase of the study, six developed serious infections: Clostridium difficile, viral gastroenteritis, UTI, anal abscess, vaginal abscess, and a staph infection of a central catheter.
This study was sponsored by Janssen Research and Development; Janssen Biotech makes ustekinumab. Dr. Sandborn and his associates reported numerous ties to industry sources.
Ustekinumab induced a clinical response in patients with moderate to severe Crohn’s disease that was resistant to tumor necrosis factor antagonists, in a phase IIb clinical trial published online Oct. 17 in the New England Journal of Medicine.
However, the agent did not improve remission rates, compared with placebo, said Dr. William J. Sandborn, professor of medicine and chief of the division of gastroenterology at the University of California San Diego, La Jolla, and his associates.
"A sizable proportion" of patients with moderate to severe Crohn’s disease do not respond to TNF antagonists, have an unsustained response, or must discontinue the medications because of adverse effects. After ustekinumab showed efficacy in such patients in a phase IIa clinical study, Dr. Sandborn and his colleagues performed a 36-week double-blind phase II2b trial in 526 adults at 153 medical centers in 12 countries.
Ustekinumab, a human IgG monoclonal antibody that inhibits the receptors for interleukin-12 and interleukin-23 on T cells, natural killer cells, and antigen-presenting cells, has Food and Drug Administration approval for use in plaque psoriasis. This clinical trial was sponsored by an affiliate of the manufacturer, Janssen Biotech.
During an 8-week induction phase, the study subjects were randomly assigned to receive intravenous placebo (132 patients) or ustekinumab in 1-mg/kg (131 patients), 3-mg/kg (132 patients), or 6-mg/kg (131 patients) doses. Then, during weeks 8-36, the study subjects who showed a response to induction therapy and those who did not show a response were separately randomized to receive either subcutaneous ustekinumab (90 mg) or placebo at week 8 and week 16, as maintenance therapy.
Treatment efficacy was assessed at week 22, and patients were followed through week 36 for a safety analysis. A total of 36.1% of the subjects discontinued the study before week 36.
The primary end point was a clinical response, defined as a decrease of 100 points or more on the Crohn’s Disease Activity Index (CDAI) score.
A total of 39.7% of patients receiving the 6-mg induction dose showed a clinical response, which was significantly greater than the 23.5% of patients receiving placebo, the investigators said (New Engl. J. Med. 2012 [doi:10.1056/NEJMoa1203572]).
A greater number of patients receiving the lower doses of ustekinumab than receiving placebo showed a clinical response, but the differences between these low-dose groups and the placebo group did not reach statistical significance.
The 6-mg/kg dose was effective across most demographic and disease characteristics, judging from the findings of a subgroup analysis. It was consistently effective in patients who had failed on their first attempt at therapy with TNF antagonists, patients who had failed on two or more TNF antagonists, and patients who had only had a transient response to TNF antagonists.
However, rates of clinical remission did not differ significantly between patients receiving ustekinumab and those receiving placebo, Dr. Sandborn and his associates said.
At all follow-up visits, the proportion of patients who had a 70-point clinical response was significantly higher, the reductions in mean CDAI scores were significantly greater, and the reductions in C-reactive protein levels were significantly greater in patients receiving 6 mg per kg of ustekinumab than in the placebo group.
As a maintenance therapy, 90 mg of subcutaneous ustekinumab appeared to be effective in patients who responded to the induction dose of the agent. The proportion of patients who showed a clinical response at week 22 was 69.4% in those receiving maintenance ustekinumab, significantly greater than the 42.5% response rate among those receiving maintenance placebo.
Among patients who responded to induction-phase ustekinumab, 41.7% of those who also received maintenance ustekinumab achieved clinical remission at week 22, compared with only 27.4% of those who received maintenance placebo.
Similarly, among patients who showed a response to induction ustekinumab, reductions in both CDAI scores and CRP levels were sustained if they continued on maintenance ustekinumab but were not sustained if they continued on placebo for the maintenance period.
However, patients who did not show a response to induction ustekinumab also did not benefit from additional ustekinumab in the maintenance phase of the study.
The results of the safety analysis were "somewhat limited" by the small sample size and the short duration of treatment. No deaths, serious opportunistic infections, or major adverse cardiovascular events were reported, "but large studies of longer duration are needed to assess uncommon adverse events," the investigators said.
Of note, one patient receiving ustekinumab as both induction and maintenance therapy developed a basal cell carcinoma. Among patients taking ustekinumab in the induction phase of the study, six developed serious infections: Clostridium difficile, viral gastroenteritis, UTI, anal abscess, vaginal abscess, and a staph infection of a central catheter.
This study was sponsored by Janssen Research and Development; Janssen Biotech makes ustekinumab. Dr. Sandborn and his associates reported numerous ties to industry sources.
Ustekinumab induced a clinical response in patients with moderate to severe Crohn’s disease that was resistant to tumor necrosis factor antagonists, in a phase IIb clinical trial published online Oct. 17 in the New England Journal of Medicine.
However, the agent did not improve remission rates, compared with placebo, said Dr. William J. Sandborn, professor of medicine and chief of the division of gastroenterology at the University of California San Diego, La Jolla, and his associates.
"A sizable proportion" of patients with moderate to severe Crohn’s disease do not respond to TNF antagonists, have an unsustained response, or must discontinue the medications because of adverse effects. After ustekinumab showed efficacy in such patients in a phase IIa clinical study, Dr. Sandborn and his colleagues performed a 36-week double-blind phase II2b trial in 526 adults at 153 medical centers in 12 countries.
Ustekinumab, a human IgG monoclonal antibody that inhibits the receptors for interleukin-12 and interleukin-23 on T cells, natural killer cells, and antigen-presenting cells, has Food and Drug Administration approval for use in plaque psoriasis. This clinical trial was sponsored by an affiliate of the manufacturer, Janssen Biotech.
During an 8-week induction phase, the study subjects were randomly assigned to receive intravenous placebo (132 patients) or ustekinumab in 1-mg/kg (131 patients), 3-mg/kg (132 patients), or 6-mg/kg (131 patients) doses. Then, during weeks 8-36, the study subjects who showed a response to induction therapy and those who did not show a response were separately randomized to receive either subcutaneous ustekinumab (90 mg) or placebo at week 8 and week 16, as maintenance therapy.
Treatment efficacy was assessed at week 22, and patients were followed through week 36 for a safety analysis. A total of 36.1% of the subjects discontinued the study before week 36.
The primary end point was a clinical response, defined as a decrease of 100 points or more on the Crohn’s Disease Activity Index (CDAI) score.
A total of 39.7% of patients receiving the 6-mg induction dose showed a clinical response, which was significantly greater than the 23.5% of patients receiving placebo, the investigators said (New Engl. J. Med. 2012 [doi:10.1056/NEJMoa1203572]).
A greater number of patients receiving the lower doses of ustekinumab than receiving placebo showed a clinical response, but the differences between these low-dose groups and the placebo group did not reach statistical significance.
The 6-mg/kg dose was effective across most demographic and disease characteristics, judging from the findings of a subgroup analysis. It was consistently effective in patients who had failed on their first attempt at therapy with TNF antagonists, patients who had failed on two or more TNF antagonists, and patients who had only had a transient response to TNF antagonists.
However, rates of clinical remission did not differ significantly between patients receiving ustekinumab and those receiving placebo, Dr. Sandborn and his associates said.
At all follow-up visits, the proportion of patients who had a 70-point clinical response was significantly higher, the reductions in mean CDAI scores were significantly greater, and the reductions in C-reactive protein levels were significantly greater in patients receiving 6 mg per kg of ustekinumab than in the placebo group.
As a maintenance therapy, 90 mg of subcutaneous ustekinumab appeared to be effective in patients who responded to the induction dose of the agent. The proportion of patients who showed a clinical response at week 22 was 69.4% in those receiving maintenance ustekinumab, significantly greater than the 42.5% response rate among those receiving maintenance placebo.
Among patients who responded to induction-phase ustekinumab, 41.7% of those who also received maintenance ustekinumab achieved clinical remission at week 22, compared with only 27.4% of those who received maintenance placebo.
Similarly, among patients who showed a response to induction ustekinumab, reductions in both CDAI scores and CRP levels were sustained if they continued on maintenance ustekinumab but were not sustained if they continued on placebo for the maintenance period.
However, patients who did not show a response to induction ustekinumab also did not benefit from additional ustekinumab in the maintenance phase of the study.
The results of the safety analysis were "somewhat limited" by the small sample size and the short duration of treatment. No deaths, serious opportunistic infections, or major adverse cardiovascular events were reported, "but large studies of longer duration are needed to assess uncommon adverse events," the investigators said.
Of note, one patient receiving ustekinumab as both induction and maintenance therapy developed a basal cell carcinoma. Among patients taking ustekinumab in the induction phase of the study, six developed serious infections: Clostridium difficile, viral gastroenteritis, UTI, anal abscess, vaginal abscess, and a staph infection of a central catheter.
This study was sponsored by Janssen Research and Development; Janssen Biotech makes ustekinumab. Dr. Sandborn and his associates reported numerous ties to industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Of patients with moderate to severe Crohn's disease who received ustekinumab (6 mg/kg), 39.7% showed a decrease of 100 points or more in CDAI score, compared with 23.5% of those who received placebo.
Data Source: The data come from a 36-week,international phase IIb randomized clinical trial comparing 3 doses of ustekinumab with placebo in 526 adults who had refractory Crohn’ disease.
Disclosures: This study was sponsored by Janssen Research and Development; Janssen Biotech makes ustekinumab. Dr. Sandborn and his associates reported numerous ties to industry sources.
Bedside Tools to ID Severe C. difficile Fall Short
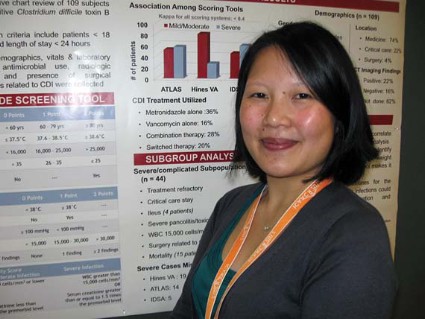
SAN FRANCISCO – A side-by-side comparison of three bedside tools used to identify severe cases of Clostridium difficile infection yielded no clear winner, a reminder that judgment at diagnosis is still the clinician’s best bet.
Criteria from the Infectious Diseases Society of America were more sensitive but the least specific than both the Hines Veterans Affairs (VA) and the ATLAS severity scoring systems, Thien-Ly Doan, Pharm.D. explained in an interview at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy.
The Hines VA system for stratifying patients missed 19 of 44 severe/complicated cases of C. difficile infection. The ATLAS scoring system (which incorporates five parameters: age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use) missed 14 of the 44 cases in a retrospective chart review of 109 patients hospitalized for more than a day with confirmed C. difficile infection.
The IDSA guidelines missed only 5 of the 44 severe/complicated infections, but they cast such a wide net that anyone with a white count above 15,000 cells/mm3 or an elevated creatinine (1.5 times or greater than the premorbid level) is considered to have severe C. difficile infection, she said.
Use of the IDSA guidelines could increase unnecessary use of vancomycin instead of metronidazole, said Dr. Doan, a clinical coordinator at Long Island Jewish Medical Center, New Hyde Park, N.Y.
The IDSA criteria suggested that nearly 60% of the 109 patients had severe infection. However, the 44 severe/complicated C. difficile patients comprised just 40% of the study population. They were defined in the study as patients who were in critical care or whose infections were refractory to treatment and who had ileus, severe pancolitis/toxic megacolon, a WBC of 15,000 cells/mL with hypotension, surgery related to C. difficile infection, or who had died from infection.
Dr. Doan and her associates compared the three stratification systems in evaluating the charts of adults with C. difficile infection at the medical center, who had a mean age of 71 years. A total of 74% of patients were on the medicine service, 22% were in critical care, and 4% were on the surgical service; 34% were female.
The Centers for Disease Control and Prevention also offer severity criteria, but these require the observation of clinical end points and thus are ineffective for assessing patients at initial presentation, she said in a poster presentation at the meeting, sponsored by the American Society for Microbiology.
The Hines VA scoring system, in addition to missing the most severe cases, also gives a great deal of weight to diagnostic imaging, which "makes it impractical at our institution," she said. The Hines VA tool incorporates temperature, the presence of ileus, systolic blood pressure, leukocytosis, and abnormal CT findings to stratify patients by severity.
"We’re going to continue relying on the clinician’s assessment at the bedside at the time of diagnosis to evaluate whether cases are severe or not severe, and not use any of these tools that are available," Dr. Doan said.
A good bedside tool sure would be nice, though, to have a good, objective way of identifying severe C. difficile infection, she added. In a large health system, order sets could be developed based on the tool’s findings "so that everybody would be on the same page in terms of treatment," she said. None of the current tools are good enough for that.
Severe cases of C. difficile are on the rise because of increasing prevalence of the hypervirulent NAP1/BI/027 strain, she noted.
A number of clinicians at the meeting approached her with their own versions of bedside tools for identifying severe C. difficile infection, which Dr. Doan and her associates may evaluate next. They also may compare the tools on different subpopulations of patients with severe infection, such as only patients whose death or surgery was related to C. difficile infection.
Dr. Doan reported having no financial disclosures.
Reported mortality from Clostridium difficile infection (CDI) in the United States has increased dramatically in recent years (Emerg. Infect. Dis. 2007;13: 1417-9). Current guidelines call for the use of oral vancomy-cin as first-line therapy in severe CDI while metronidazole may be used in milder disease (Infect. Control Hosp. Epidemiol. 2010;31:431-55). Thus, it becomes important for therapy to identify those with potentially severe CDI early in their clinical course. However, a systematic review published in 2012 that specifically looked at clinical prediction rules (CPRs) for poor outcomes in CDI concluded that the available tools are inadequate for the task (PLoS One 2012;7:e30258).
The study by Dr. Doan and colleagues assessed the utility of bedside severity-of-illness tools in the treatment of patients with CDI. This was a retrospective chart review of 109 patients hospitalized for more than a day with confirmed CDI. Three CPRs were assessed: The Hines VA system , ; the ATLAS scoring system; and the Infectious Diseases Society of America (IDSA) guidelines. . Sensitivity in detecting severe outcomes of CDI were 57%, 68%, and 89%, respectively. However, the most sensitive CPR, the IDSA guideline, showed poor specificity because it categorized 60% of all subjects as severe. Thus, the IDSA guideline will encourage more widespread use of oral vancomycin in CDI.
Therefore, we lack a risk-scoring system for severe CDI that is easy to use, sensitive, specific, and validated. Such a prediction tool is essential to allow us to follow the current CDI treatment guidelines.
CIARAN P. KELLY, M.D., is director of gastroenterology training and is medical director of the Celiac Center at Beth Israel Deaconess Medical Center, Boston. SAURABH SETHI, M.D., is a fellow in gastroenterology and hepatology at Beth Israe Deaconess. Dr. Kelly reported serving as a consultant or scientific advisor for, being a member of an advisory board for, or receiving research support from many companies developing drugs for C. difficile. Dr. Sethi had no relevant financial disclosures.
Reported mortality from Clostridium difficile infection (CDI) in the United States has increased dramatically in recent years (Emerg. Infect. Dis. 2007;13: 1417-9). Current guidelines call for the use of oral vancomy-cin as first-line therapy in severe CDI while metronidazole may be used in milder disease (Infect. Control Hosp. Epidemiol. 2010;31:431-55). Thus, it becomes important for therapy to identify those with potentially severe CDI early in their clinical course. However, a systematic review published in 2012 that specifically looked at clinical prediction rules (CPRs) for poor outcomes in CDI concluded that the available tools are inadequate for the task (PLoS One 2012;7:e30258).
The study by Dr. Doan and colleagues assessed the utility of bedside severity-of-illness tools in the treatment of patients with CDI. This was a retrospective chart review of 109 patients hospitalized for more than a day with confirmed CDI. Three CPRs were assessed: The Hines VA system , ; the ATLAS scoring system; and the Infectious Diseases Society of America (IDSA) guidelines. . Sensitivity in detecting severe outcomes of CDI were 57%, 68%, and 89%, respectively. However, the most sensitive CPR, the IDSA guideline, showed poor specificity because it categorized 60% of all subjects as severe. Thus, the IDSA guideline will encourage more widespread use of oral vancomycin in CDI.
Therefore, we lack a risk-scoring system for severe CDI that is easy to use, sensitive, specific, and validated. Such a prediction tool is essential to allow us to follow the current CDI treatment guidelines.
CIARAN P. KELLY, M.D., is director of gastroenterology training and is medical director of the Celiac Center at Beth Israel Deaconess Medical Center, Boston. SAURABH SETHI, M.D., is a fellow in gastroenterology and hepatology at Beth Israe Deaconess. Dr. Kelly reported serving as a consultant or scientific advisor for, being a member of an advisory board for, or receiving research support from many companies developing drugs for C. difficile. Dr. Sethi had no relevant financial disclosures.
Reported mortality from Clostridium difficile infection (CDI) in the United States has increased dramatically in recent years (Emerg. Infect. Dis. 2007;13: 1417-9). Current guidelines call for the use of oral vancomy-cin as first-line therapy in severe CDI while metronidazole may be used in milder disease (Infect. Control Hosp. Epidemiol. 2010;31:431-55). Thus, it becomes important for therapy to identify those with potentially severe CDI early in their clinical course. However, a systematic review published in 2012 that specifically looked at clinical prediction rules (CPRs) for poor outcomes in CDI concluded that the available tools are inadequate for the task (PLoS One 2012;7:e30258).
The study by Dr. Doan and colleagues assessed the utility of bedside severity-of-illness tools in the treatment of patients with CDI. This was a retrospective chart review of 109 patients hospitalized for more than a day with confirmed CDI. Three CPRs were assessed: The Hines VA system , ; the ATLAS scoring system; and the Infectious Diseases Society of America (IDSA) guidelines. . Sensitivity in detecting severe outcomes of CDI were 57%, 68%, and 89%, respectively. However, the most sensitive CPR, the IDSA guideline, showed poor specificity because it categorized 60% of all subjects as severe. Thus, the IDSA guideline will encourage more widespread use of oral vancomycin in CDI.
Therefore, we lack a risk-scoring system for severe CDI that is easy to use, sensitive, specific, and validated. Such a prediction tool is essential to allow us to follow the current CDI treatment guidelines.
CIARAN P. KELLY, M.D., is director of gastroenterology training and is medical director of the Celiac Center at Beth Israel Deaconess Medical Center, Boston. SAURABH SETHI, M.D., is a fellow in gastroenterology and hepatology at Beth Israe Deaconess. Dr. Kelly reported serving as a consultant or scientific advisor for, being a member of an advisory board for, or receiving research support from many companies developing drugs for C. difficile. Dr. Sethi had no relevant financial disclosures.
SAN FRANCISCO – A side-by-side comparison of three bedside tools used to identify severe cases of Clostridium difficile infection yielded no clear winner, a reminder that judgment at diagnosis is still the clinician’s best bet.
Criteria from the Infectious Diseases Society of America were more sensitive but the least specific than both the Hines Veterans Affairs (VA) and the ATLAS severity scoring systems, Thien-Ly Doan, Pharm.D. explained in an interview at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy.
The Hines VA system for stratifying patients missed 19 of 44 severe/complicated cases of C. difficile infection. The ATLAS scoring system (which incorporates five parameters: age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use) missed 14 of the 44 cases in a retrospective chart review of 109 patients hospitalized for more than a day with confirmed C. difficile infection.
The IDSA guidelines missed only 5 of the 44 severe/complicated infections, but they cast such a wide net that anyone with a white count above 15,000 cells/mm3 or an elevated creatinine (1.5 times or greater than the premorbid level) is considered to have severe C. difficile infection, she said.
Use of the IDSA guidelines could increase unnecessary use of vancomycin instead of metronidazole, said Dr. Doan, a clinical coordinator at Long Island Jewish Medical Center, New Hyde Park, N.Y.
The IDSA criteria suggested that nearly 60% of the 109 patients had severe infection. However, the 44 severe/complicated C. difficile patients comprised just 40% of the study population. They were defined in the study as patients who were in critical care or whose infections were refractory to treatment and who had ileus, severe pancolitis/toxic megacolon, a WBC of 15,000 cells/mL with hypotension, surgery related to C. difficile infection, or who had died from infection.
Dr. Doan and her associates compared the three stratification systems in evaluating the charts of adults with C. difficile infection at the medical center, who had a mean age of 71 years. A total of 74% of patients were on the medicine service, 22% were in critical care, and 4% were on the surgical service; 34% were female.
The Centers for Disease Control and Prevention also offer severity criteria, but these require the observation of clinical end points and thus are ineffective for assessing patients at initial presentation, she said in a poster presentation at the meeting, sponsored by the American Society for Microbiology.
The Hines VA scoring system, in addition to missing the most severe cases, also gives a great deal of weight to diagnostic imaging, which "makes it impractical at our institution," she said. The Hines VA tool incorporates temperature, the presence of ileus, systolic blood pressure, leukocytosis, and abnormal CT findings to stratify patients by severity.
"We’re going to continue relying on the clinician’s assessment at the bedside at the time of diagnosis to evaluate whether cases are severe or not severe, and not use any of these tools that are available," Dr. Doan said.
A good bedside tool sure would be nice, though, to have a good, objective way of identifying severe C. difficile infection, she added. In a large health system, order sets could be developed based on the tool’s findings "so that everybody would be on the same page in terms of treatment," she said. None of the current tools are good enough for that.
Severe cases of C. difficile are on the rise because of increasing prevalence of the hypervirulent NAP1/BI/027 strain, she noted.
A number of clinicians at the meeting approached her with their own versions of bedside tools for identifying severe C. difficile infection, which Dr. Doan and her associates may evaluate next. They also may compare the tools on different subpopulations of patients with severe infection, such as only patients whose death or surgery was related to C. difficile infection.
Dr. Doan reported having no financial disclosures.
SAN FRANCISCO – A side-by-side comparison of three bedside tools used to identify severe cases of Clostridium difficile infection yielded no clear winner, a reminder that judgment at diagnosis is still the clinician’s best bet.
Criteria from the Infectious Diseases Society of America were more sensitive but the least specific than both the Hines Veterans Affairs (VA) and the ATLAS severity scoring systems, Thien-Ly Doan, Pharm.D. explained in an interview at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy.
The Hines VA system for stratifying patients missed 19 of 44 severe/complicated cases of C. difficile infection. The ATLAS scoring system (which incorporates five parameters: age, temperature, leukocytosis, albumin, and systemic concomitant antibiotic use) missed 14 of the 44 cases in a retrospective chart review of 109 patients hospitalized for more than a day with confirmed C. difficile infection.
The IDSA guidelines missed only 5 of the 44 severe/complicated infections, but they cast such a wide net that anyone with a white count above 15,000 cells/mm3 or an elevated creatinine (1.5 times or greater than the premorbid level) is considered to have severe C. difficile infection, she said.
Use of the IDSA guidelines could increase unnecessary use of vancomycin instead of metronidazole, said Dr. Doan, a clinical coordinator at Long Island Jewish Medical Center, New Hyde Park, N.Y.
The IDSA criteria suggested that nearly 60% of the 109 patients had severe infection. However, the 44 severe/complicated C. difficile patients comprised just 40% of the study population. They were defined in the study as patients who were in critical care or whose infections were refractory to treatment and who had ileus, severe pancolitis/toxic megacolon, a WBC of 15,000 cells/mL with hypotension, surgery related to C. difficile infection, or who had died from infection.
Dr. Doan and her associates compared the three stratification systems in evaluating the charts of adults with C. difficile infection at the medical center, who had a mean age of 71 years. A total of 74% of patients were on the medicine service, 22% were in critical care, and 4% were on the surgical service; 34% were female.
The Centers for Disease Control and Prevention also offer severity criteria, but these require the observation of clinical end points and thus are ineffective for assessing patients at initial presentation, she said in a poster presentation at the meeting, sponsored by the American Society for Microbiology.
The Hines VA scoring system, in addition to missing the most severe cases, also gives a great deal of weight to diagnostic imaging, which "makes it impractical at our institution," she said. The Hines VA tool incorporates temperature, the presence of ileus, systolic blood pressure, leukocytosis, and abnormal CT findings to stratify patients by severity.
"We’re going to continue relying on the clinician’s assessment at the bedside at the time of diagnosis to evaluate whether cases are severe or not severe, and not use any of these tools that are available," Dr. Doan said.
A good bedside tool sure would be nice, though, to have a good, objective way of identifying severe C. difficile infection, she added. In a large health system, order sets could be developed based on the tool’s findings "so that everybody would be on the same page in terms of treatment," she said. None of the current tools are good enough for that.
Severe cases of C. difficile are on the rise because of increasing prevalence of the hypervirulent NAP1/BI/027 strain, she noted.
A number of clinicians at the meeting approached her with their own versions of bedside tools for identifying severe C. difficile infection, which Dr. Doan and her associates may evaluate next. They also may compare the tools on different subpopulations of patients with severe infection, such as only patients whose death or surgery was related to C. difficile infection.
Dr. Doan reported having no financial disclosures.
AT THE ANNUAL INTERSCIENCE CONFERENCE ON ANTIMICROBIAL AGENTS AND CHEMOTHERAPY
Second TNF-Blocker Approved for Refractory Ulcerative Colitis
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced on Sept. 28.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating UC.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference. In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication. Panelists cited the need for more treatments for UC and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said. The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week. "The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced on Sept. 28.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating UC.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference. In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication. Panelists cited the need for more treatments for UC and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said. The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week. "The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced on Sept. 28.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating UC.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference. In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication. Panelists cited the need for more treatments for UC and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said. The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week. "The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
Open Surgery Tied to Small-Bowel Obstruction Risk
The risk of developing a small-bowel obstruction after open surgery is about fourfold higher when compared with laparoscopic surgery in nine commonly performed abdominal and pelvic procedures, including cholecystectomy, hysterectomy, and appendectomy, investigators reported.
Other factors such as patient age, , prior abdominal surgery, and comorbidities also contributed to the risk of SBO, the study showed. However, laparoscopy "exceeded other risk factors in reduction of the risk of SBO for most of the surgical procedures," wrote Dr. Eva Angenete and her colleagues (Arch. Surg. 2012;147:359-65).
"This study shows that, beyond important factors such as age, previous abdominal surgery, and comorbidity, the surgical technique is the most important factor related to SBO," the authors wrote. "Compared with laparoscopic surgery, open surgery seems to increase the risk of SBO at least four times."
It’s likely that the study results will hold up to further scrutiny because the study was population based and the sample size – 108,141 patients – is large, the authors said.
Still, laparoscopic surgery did not seem to reduce the incidence of SBO in some groups, including hysterectomy patients. "One hypothesis is that this result may be related to a limited dissection in the pelvis," but the small number of laparoscopic hysterectomy patients included in the study could have affected the study’s results, the authors wrote. In addition, there were no clear risk factors for SBO in patients undergoing bariatric surgery, they wrote.
The investigators used the Inpatient Register of the Swedish National Board of Health and Welfare to analyze the risk of SBO in nine procedures, including cholecystectomy, hysterectomy, salpingo-oophorectomy, bowel resection, anterior resection, abdominoperineal resection, rectopexy, appendectomy, and bariatric surgery performed from 2002 to 2004. The database included information on demographic characteristics, comorbidities, previous abdominal surgery, and deaths.
The rate of SBO was lowest after cholecystectomy, occurring in just 0.4% of all cases, and was highest, at 13.9%, in abdominoperineal resection patients. For most surgical procedures, patients who had SBO were older on average. SBO was also more common in patients with previous abdominal surgery.
In those who underwent cholecystectomy, bowel resection, or appendectomy, a higher level of comorbidity was associated with a greater incidence of SBO, the authors said. In the group of complicated cholecystectomy patients as well as the group of anterior resection patients, SBO was more common among men.
SBO was linked with an increased risk of death within 5 years, the study found.
"The aim of this study was to identify the incidence and risk factors for mechanical SBO after a number of common abdominal and pelvic procedures," the authors wrote. "Small-bowel obstruction is a substantial health care challenge, and correctly identified risk factors can provide improved tools to reduce the risk of SBO after an abdominal surgical procedure."
"The safety and the short-term benefits of laparoscopy are already known, and it is possible that laparoscopy should be regarded as the preferred technique in an attempt to further reduce the complications of surgery," the authors concluded.
The project was supported by grants from the Swedish Cancer Foundation, the Swedish Medical Association, the Gothenburg Medical Association, the Assar Gabrielsson Foundation, the Magn Berwall’s Foundation, and the Swedish Research Council. None of the funding sources had any role in the study or the preparation of the manuscript. The authors reported no financial conflicts of interest.
Replacing open abdominal surgery with laparoscopy when possible may offer an opportunity for improved quality of life and decreased morbidity for many patients, along with health care system cost savings, said Dr. Luke M. Funk and Dr. Stanley W. Ashley in an invited critique accompanying the study on small-bowel obstruction (SBO) risk (Arch Surg. 2012;147:365 [doi:10.1001/archsurg.2012.157]).
The finding that SBO risk was lower with laparoscopy even after accounting for patient factors such as age, comorbidities, and previous surgery, has important implications for both the quality and cost of surgical care, Dr. Funk and Dr. Ashley said in their commentary.
"For surgeons, it highlights another potential benefit of minimally invasive surgery and challenges us to continue to offer less invasive procedures whenever they are feasible," they wrote. "For payers and health care policy leaders, it suggests that substantial cost savings could be achieved if open surgery [were] replaced with laparoscopic surgery more often."
Inpatient expenses on adhesiolysis-related complications exceed $2 billion in the United States, they added.
However, to fully realize the benefits of laparoscopy, newer payment models such as bundled or episode-based payments would need to account for the higher initial cost of laparoscopy but lower long-term costs related to shorter hospitalizations, lower complication rates, fewer readmissions, and fewer reoperations, the two surgeons said.
Still, the study’s authors "have provided strong evidence that minimal invasion often results in maximal benefit," Dr. Funk and Dr. Ashley wrote.
Dr. Funk is a general surgeon at Brigham and Women’s Hospital in Boston. Dr. Ashley is vice chairman of the department of surgery at Brigham and Women’s.
Replacing open abdominal surgery with laparoscopy when possible may offer an opportunity for improved quality of life and decreased morbidity for many patients, along with health care system cost savings, said Dr. Luke M. Funk and Dr. Stanley W. Ashley in an invited critique accompanying the study on small-bowel obstruction (SBO) risk (Arch Surg. 2012;147:365 [doi:10.1001/archsurg.2012.157]).
The finding that SBO risk was lower with laparoscopy even after accounting for patient factors such as age, comorbidities, and previous surgery, has important implications for both the quality and cost of surgical care, Dr. Funk and Dr. Ashley said in their commentary.
"For surgeons, it highlights another potential benefit of minimally invasive surgery and challenges us to continue to offer less invasive procedures whenever they are feasible," they wrote. "For payers and health care policy leaders, it suggests that substantial cost savings could be achieved if open surgery [were] replaced with laparoscopic surgery more often."
Inpatient expenses on adhesiolysis-related complications exceed $2 billion in the United States, they added.
However, to fully realize the benefits of laparoscopy, newer payment models such as bundled or episode-based payments would need to account for the higher initial cost of laparoscopy but lower long-term costs related to shorter hospitalizations, lower complication rates, fewer readmissions, and fewer reoperations, the two surgeons said.
Still, the study’s authors "have provided strong evidence that minimal invasion often results in maximal benefit," Dr. Funk and Dr. Ashley wrote.
Dr. Funk is a general surgeon at Brigham and Women’s Hospital in Boston. Dr. Ashley is vice chairman of the department of surgery at Brigham and Women’s.
Replacing open abdominal surgery with laparoscopy when possible may offer an opportunity for improved quality of life and decreased morbidity for many patients, along with health care system cost savings, said Dr. Luke M. Funk and Dr. Stanley W. Ashley in an invited critique accompanying the study on small-bowel obstruction (SBO) risk (Arch Surg. 2012;147:365 [doi:10.1001/archsurg.2012.157]).
The finding that SBO risk was lower with laparoscopy even after accounting for patient factors such as age, comorbidities, and previous surgery, has important implications for both the quality and cost of surgical care, Dr. Funk and Dr. Ashley said in their commentary.
"For surgeons, it highlights another potential benefit of minimally invasive surgery and challenges us to continue to offer less invasive procedures whenever they are feasible," they wrote. "For payers and health care policy leaders, it suggests that substantial cost savings could be achieved if open surgery [were] replaced with laparoscopic surgery more often."
Inpatient expenses on adhesiolysis-related complications exceed $2 billion in the United States, they added.
However, to fully realize the benefits of laparoscopy, newer payment models such as bundled or episode-based payments would need to account for the higher initial cost of laparoscopy but lower long-term costs related to shorter hospitalizations, lower complication rates, fewer readmissions, and fewer reoperations, the two surgeons said.
Still, the study’s authors "have provided strong evidence that minimal invasion often results in maximal benefit," Dr. Funk and Dr. Ashley wrote.
Dr. Funk is a general surgeon at Brigham and Women’s Hospital in Boston. Dr. Ashley is vice chairman of the department of surgery at Brigham and Women’s.
The risk of developing a small-bowel obstruction after open surgery is about fourfold higher when compared with laparoscopic surgery in nine commonly performed abdominal and pelvic procedures, including cholecystectomy, hysterectomy, and appendectomy, investigators reported.
Other factors such as patient age, , prior abdominal surgery, and comorbidities also contributed to the risk of SBO, the study showed. However, laparoscopy "exceeded other risk factors in reduction of the risk of SBO for most of the surgical procedures," wrote Dr. Eva Angenete and her colleagues (Arch. Surg. 2012;147:359-65).
"This study shows that, beyond important factors such as age, previous abdominal surgery, and comorbidity, the surgical technique is the most important factor related to SBO," the authors wrote. "Compared with laparoscopic surgery, open surgery seems to increase the risk of SBO at least four times."
It’s likely that the study results will hold up to further scrutiny because the study was population based and the sample size – 108,141 patients – is large, the authors said.
Still, laparoscopic surgery did not seem to reduce the incidence of SBO in some groups, including hysterectomy patients. "One hypothesis is that this result may be related to a limited dissection in the pelvis," but the small number of laparoscopic hysterectomy patients included in the study could have affected the study’s results, the authors wrote. In addition, there were no clear risk factors for SBO in patients undergoing bariatric surgery, they wrote.
The investigators used the Inpatient Register of the Swedish National Board of Health and Welfare to analyze the risk of SBO in nine procedures, including cholecystectomy, hysterectomy, salpingo-oophorectomy, bowel resection, anterior resection, abdominoperineal resection, rectopexy, appendectomy, and bariatric surgery performed from 2002 to 2004. The database included information on demographic characteristics, comorbidities, previous abdominal surgery, and deaths.
The rate of SBO was lowest after cholecystectomy, occurring in just 0.4% of all cases, and was highest, at 13.9%, in abdominoperineal resection patients. For most surgical procedures, patients who had SBO were older on average. SBO was also more common in patients with previous abdominal surgery.
In those who underwent cholecystectomy, bowel resection, or appendectomy, a higher level of comorbidity was associated with a greater incidence of SBO, the authors said. In the group of complicated cholecystectomy patients as well as the group of anterior resection patients, SBO was more common among men.
SBO was linked with an increased risk of death within 5 years, the study found.
"The aim of this study was to identify the incidence and risk factors for mechanical SBO after a number of common abdominal and pelvic procedures," the authors wrote. "Small-bowel obstruction is a substantial health care challenge, and correctly identified risk factors can provide improved tools to reduce the risk of SBO after an abdominal surgical procedure."
"The safety and the short-term benefits of laparoscopy are already known, and it is possible that laparoscopy should be regarded as the preferred technique in an attempt to further reduce the complications of surgery," the authors concluded.
The project was supported by grants from the Swedish Cancer Foundation, the Swedish Medical Association, the Gothenburg Medical Association, the Assar Gabrielsson Foundation, the Magn Berwall’s Foundation, and the Swedish Research Council. None of the funding sources had any role in the study or the preparation of the manuscript. The authors reported no financial conflicts of interest.
The risk of developing a small-bowel obstruction after open surgery is about fourfold higher when compared with laparoscopic surgery in nine commonly performed abdominal and pelvic procedures, including cholecystectomy, hysterectomy, and appendectomy, investigators reported.
Other factors such as patient age, , prior abdominal surgery, and comorbidities also contributed to the risk of SBO, the study showed. However, laparoscopy "exceeded other risk factors in reduction of the risk of SBO for most of the surgical procedures," wrote Dr. Eva Angenete and her colleagues (Arch. Surg. 2012;147:359-65).
"This study shows that, beyond important factors such as age, previous abdominal surgery, and comorbidity, the surgical technique is the most important factor related to SBO," the authors wrote. "Compared with laparoscopic surgery, open surgery seems to increase the risk of SBO at least four times."
It’s likely that the study results will hold up to further scrutiny because the study was population based and the sample size – 108,141 patients – is large, the authors said.
Still, laparoscopic surgery did not seem to reduce the incidence of SBO in some groups, including hysterectomy patients. "One hypothesis is that this result may be related to a limited dissection in the pelvis," but the small number of laparoscopic hysterectomy patients included in the study could have affected the study’s results, the authors wrote. In addition, there were no clear risk factors for SBO in patients undergoing bariatric surgery, they wrote.
The investigators used the Inpatient Register of the Swedish National Board of Health and Welfare to analyze the risk of SBO in nine procedures, including cholecystectomy, hysterectomy, salpingo-oophorectomy, bowel resection, anterior resection, abdominoperineal resection, rectopexy, appendectomy, and bariatric surgery performed from 2002 to 2004. The database included information on demographic characteristics, comorbidities, previous abdominal surgery, and deaths.
The rate of SBO was lowest after cholecystectomy, occurring in just 0.4% of all cases, and was highest, at 13.9%, in abdominoperineal resection patients. For most surgical procedures, patients who had SBO were older on average. SBO was also more common in patients with previous abdominal surgery.
In those who underwent cholecystectomy, bowel resection, or appendectomy, a higher level of comorbidity was associated with a greater incidence of SBO, the authors said. In the group of complicated cholecystectomy patients as well as the group of anterior resection patients, SBO was more common among men.
SBO was linked with an increased risk of death within 5 years, the study found.
"The aim of this study was to identify the incidence and risk factors for mechanical SBO after a number of common abdominal and pelvic procedures," the authors wrote. "Small-bowel obstruction is a substantial health care challenge, and correctly identified risk factors can provide improved tools to reduce the risk of SBO after an abdominal surgical procedure."
"The safety and the short-term benefits of laparoscopy are already known, and it is possible that laparoscopy should be regarded as the preferred technique in an attempt to further reduce the complications of surgery," the authors concluded.
The project was supported by grants from the Swedish Cancer Foundation, the Swedish Medical Association, the Gothenburg Medical Association, the Assar Gabrielsson Foundation, the Magn Berwall’s Foundation, and the Swedish Research Council. None of the funding sources had any role in the study or the preparation of the manuscript. The authors reported no financial conflicts of interest.
Major Finding: The risk of small-bowel obstruction is four times higher in patients who undergo open surgery than in patients who undergo laparoscopy for nine commonly performed procedures.
Data Source: Data were analyzed from 108,141 cases between 2002 and 2004 from the Inpatient Register of the Swedish National Board of Health and Welfare.
Disclosures: The project was supported by grants from the Swedish Cancer Foundation, the Swedish Medical Association, the Gothenburg Medical Association, the Assar Gabrielsson Foundation, the Magn Berwall’s Foundation, and the Swedish Research Council. None of the funding sources had any role in the study or the preparation of the manuscript. The authors reported no financial conflicts of interest.
Panel Backs Approval of Second Biologic for Ulcerative Colitis
SILVER SPRING, MD. – The approval of a second anti-tumor necrosis factor treatment option for ulcerative colitis is likely, now that the majority of a Food and Drug Administration advisory panel has recommended approval of adalimumab for treating people with moderately to severely active disease.
At a meeting on Aug. 28, the FDA’s Gastrointestinal Drugs Advisory Committee voted 15-2 that the expected benefits of adalimumab – a subcutaneously administered tumor necrosis factor (TNF) blocker – outweighed its potential risks as a treatment for patients with moderately to severely active ulcerative colitis (UC) who have not had an adequate response with conventional treatments. The panel’s recommendation for approval was based on the results of two studies comparing adalimumab against placebo in such patients. But their support for approval came with caveats. While most of the panel agreed that the dose used in the studies had been shown to be clinically effective, they agreed that the optimal dose for treating UC had not yet been determined and that post-approval studies were needed to address dosing, subpopulations of patients who may benefit most from treatment, and long-term safety.
Panel members voting in favor of approval cited the need for more treatments and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects. And although the differences in clinical remission rates between placebo and treatment at 8 weeks and at 1 year were less than 10% – one of the main issues raised by FDA reviewers – most of the panel members said that these differences still represented a clinically meaningful benefit. Infliximab (Remicade), an intravenous TNF blocker, is approved for treating UC and Crohn’s disease in pediatric and adult patients.
Like other panelists, Dr. Andelka LoSavio of the department of gastroenterology and nutrition, Loyola University, Maywood, Ill., said that the outstanding issues should not hold up approval of adalimumab for UC. "Once the drug is on the marketplace, we will have more insight into subpopulations where the drug may be more effective," which was the case with Crohn’s disease and adalimumab.
Although the remission rates in the studies were low, these are difficult-to-treat patients, and the responses were statistically significant, with evidence of a steroid-sparing effect, said panelist Dr. Marc Wishingrad, a gastroenterologist who practices in Santa Monica, Calif. "So it seems to me in a global sense that there is enough benefit here, that overall I would recommend that this drug be approved for this indication," he added.
Panelists supported the manufacturer’s recommendation that if a patient does not respond after 8 weeks of treatment, that treatment should be stopped.
The two statisticians on the panel did not support approval, voting no on the risk-benefit question for reasons that included the modest effects on clinical remission rates, uncertain durability of these effects, inadequate long-term data, missing data, and uncertainties about the dose.
Adalimumab – marketed as Humira by Abbott Laboratories – was first approved in 2002 for treating moderate to severe rheumatoid arthritis in adults, and was approved in 2007 for treating moderate to severe Crohn’s disease. It has also been approved for psoriasis, ankylosing spondylitis, and juvenile idiopathic arthritis indications.
The recommended dosing schedule for UC is one 160-mg starting dose, followed by 80 mg on day 15, and then 40 mg every other week starting at day 29, continuing treatment only in people who have responded within the first 8 weeks.
Abbott filed for approval in January 2011, but the FDA raised questions about whether a higher dose would be more effective and about the strength of the results in the two pivotal trials, including whether the differences in clinical remission rates at 8 and 52 weeks between placebo and treatment were clinically meaningful. The company resubmitted the application for approval, with a slightly different proposed indication: reducing the signs and symptoms, and achieving (instead of inducing and maintaining induction of) clinical remission in adults with moderately to severely active UC, who have had an inadequate response to conventional therapy.
The two double-blind, placebo-controlled, phase III studies compared adalimumab against placebo in 1,094 treatment-refractory patients with moderately to severely active UC with a total Mayo score of 6 to 12 points, and an endoscopy sub-score of 2 or 3, despite current or previous steroid and/or immunosuppressant therapy. In one 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference that was significant.
In the second study, which followed patients for 1 year and included some who had been treated with infliximab (40%), the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference that was statistically significant. At 52 weeks, the remission rate was 17.3% among those on adalimumab, vs. 8.5% among those on placebo, an 8.8% difference that also was statistically significant.
In different votes, the panel nearly unanimously agreed that the 8 week and 52 week clinical remission rates in the studies represented "clinically meaningful benefits."
However, in the second study, 8.5% of the patients on adalimumab were in clinical remission at both weeks 8 and 52, vs. 4.1% among those on placebo. The panel was less unanimous about whether the 4.4% difference between the two groups represented a clinically meaningful benefit of treatment, voting 10-6 with 1 abstention on this question.
Other findings in the 52-week study included a higher steroid discontinuation rate among those on adalimumab, a secondary endpoint: About 13% of those on adalimumab were able to go off steroid treatment before the 52nd week of treatment and had achieved a clinical remission, compared with 5.7% of those on placebo, a statistically significant difference. In addition, the patients on azathioprine or 6-MP at baseline in this study did not appear to benefit from adalimumab: In this subgroup, the remission rate at week 8 among those on adalimumab (12.9%) was similar to the rate among those on placebo (15%). But for those who were not on the immunomodulatory drugs at baseline, the remission rate was nearly 19% among those on adalimumab vs. 6.6% of those on placebo, the FDA reviewer pointed out. In the studies, the safety profile of adalimumab was similar to the placebo, other than a higher rate of injection site reactions among those on adalimumab, and no new safety signals for adalimumab were identified in the trials, according to Abbott.
If the FDA grants approval, adalimumab will be the only self-administered biologic therapy available for patients with UC, the company pointed out. In April 2012, adalimumab was approved in the European Union for treating adults with moderately to severely active UC who have had an inadequate response to conventional therapy, based on the same data submitted to the FDA for the UC indication, according to Abbott.
The company is planning an international registry of patients with UC treated with adalimumab in typical clinical settings to collect long-term safety data, which will include patients in the EU, and, if approved, in the United States. The FDA usually follows the recommendations of its advisory panels, which are not binding. The company expects a decision on approval by the end of the year, according to an Abbott spokesperson.
Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – The approval of a second anti-tumor necrosis factor treatment option for ulcerative colitis is likely, now that the majority of a Food and Drug Administration advisory panel has recommended approval of adalimumab for treating people with moderately to severely active disease.
At a meeting on Aug. 28, the FDA’s Gastrointestinal Drugs Advisory Committee voted 15-2 that the expected benefits of adalimumab – a subcutaneously administered tumor necrosis factor (TNF) blocker – outweighed its potential risks as a treatment for patients with moderately to severely active ulcerative colitis (UC) who have not had an adequate response with conventional treatments. The panel’s recommendation for approval was based on the results of two studies comparing adalimumab against placebo in such patients. But their support for approval came with caveats. While most of the panel agreed that the dose used in the studies had been shown to be clinically effective, they agreed that the optimal dose for treating UC had not yet been determined and that post-approval studies were needed to address dosing, subpopulations of patients who may benefit most from treatment, and long-term safety.
Panel members voting in favor of approval cited the need for more treatments and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects. And although the differences in clinical remission rates between placebo and treatment at 8 weeks and at 1 year were less than 10% – one of the main issues raised by FDA reviewers – most of the panel members said that these differences still represented a clinically meaningful benefit. Infliximab (Remicade), an intravenous TNF blocker, is approved for treating UC and Crohn’s disease in pediatric and adult patients.
Like other panelists, Dr. Andelka LoSavio of the department of gastroenterology and nutrition, Loyola University, Maywood, Ill., said that the outstanding issues should not hold up approval of adalimumab for UC. "Once the drug is on the marketplace, we will have more insight into subpopulations where the drug may be more effective," which was the case with Crohn’s disease and adalimumab.
Although the remission rates in the studies were low, these are difficult-to-treat patients, and the responses were statistically significant, with evidence of a steroid-sparing effect, said panelist Dr. Marc Wishingrad, a gastroenterologist who practices in Santa Monica, Calif. "So it seems to me in a global sense that there is enough benefit here, that overall I would recommend that this drug be approved for this indication," he added.
Panelists supported the manufacturer’s recommendation that if a patient does not respond after 8 weeks of treatment, that treatment should be stopped.
The two statisticians on the panel did not support approval, voting no on the risk-benefit question for reasons that included the modest effects on clinical remission rates, uncertain durability of these effects, inadequate long-term data, missing data, and uncertainties about the dose.
Adalimumab – marketed as Humira by Abbott Laboratories – was first approved in 2002 for treating moderate to severe rheumatoid arthritis in adults, and was approved in 2007 for treating moderate to severe Crohn’s disease. It has also been approved for psoriasis, ankylosing spondylitis, and juvenile idiopathic arthritis indications.
The recommended dosing schedule for UC is one 160-mg starting dose, followed by 80 mg on day 15, and then 40 mg every other week starting at day 29, continuing treatment only in people who have responded within the first 8 weeks.
Abbott filed for approval in January 2011, but the FDA raised questions about whether a higher dose would be more effective and about the strength of the results in the two pivotal trials, including whether the differences in clinical remission rates at 8 and 52 weeks between placebo and treatment were clinically meaningful. The company resubmitted the application for approval, with a slightly different proposed indication: reducing the signs and symptoms, and achieving (instead of inducing and maintaining induction of) clinical remission in adults with moderately to severely active UC, who have had an inadequate response to conventional therapy.
The two double-blind, placebo-controlled, phase III studies compared adalimumab against placebo in 1,094 treatment-refractory patients with moderately to severely active UC with a total Mayo score of 6 to 12 points, and an endoscopy sub-score of 2 or 3, despite current or previous steroid and/or immunosuppressant therapy. In one 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference that was significant.
In the second study, which followed patients for 1 year and included some who had been treated with infliximab (40%), the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference that was statistically significant. At 52 weeks, the remission rate was 17.3% among those on adalimumab, vs. 8.5% among those on placebo, an 8.8% difference that also was statistically significant.
In different votes, the panel nearly unanimously agreed that the 8 week and 52 week clinical remission rates in the studies represented "clinically meaningful benefits."
However, in the second study, 8.5% of the patients on adalimumab were in clinical remission at both weeks 8 and 52, vs. 4.1% among those on placebo. The panel was less unanimous about whether the 4.4% difference between the two groups represented a clinically meaningful benefit of treatment, voting 10-6 with 1 abstention on this question.
Other findings in the 52-week study included a higher steroid discontinuation rate among those on adalimumab, a secondary endpoint: About 13% of those on adalimumab were able to go off steroid treatment before the 52nd week of treatment and had achieved a clinical remission, compared with 5.7% of those on placebo, a statistically significant difference. In addition, the patients on azathioprine or 6-MP at baseline in this study did not appear to benefit from adalimumab: In this subgroup, the remission rate at week 8 among those on adalimumab (12.9%) was similar to the rate among those on placebo (15%). But for those who were not on the immunomodulatory drugs at baseline, the remission rate was nearly 19% among those on adalimumab vs. 6.6% of those on placebo, the FDA reviewer pointed out. In the studies, the safety profile of adalimumab was similar to the placebo, other than a higher rate of injection site reactions among those on adalimumab, and no new safety signals for adalimumab were identified in the trials, according to Abbott.
If the FDA grants approval, adalimumab will be the only self-administered biologic therapy available for patients with UC, the company pointed out. In April 2012, adalimumab was approved in the European Union for treating adults with moderately to severely active UC who have had an inadequate response to conventional therapy, based on the same data submitted to the FDA for the UC indication, according to Abbott.
The company is planning an international registry of patients with UC treated with adalimumab in typical clinical settings to collect long-term safety data, which will include patients in the EU, and, if approved, in the United States. The FDA usually follows the recommendations of its advisory panels, which are not binding. The company expects a decision on approval by the end of the year, according to an Abbott spokesperson.
Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
SILVER SPRING, MD. – The approval of a second anti-tumor necrosis factor treatment option for ulcerative colitis is likely, now that the majority of a Food and Drug Administration advisory panel has recommended approval of adalimumab for treating people with moderately to severely active disease.
At a meeting on Aug. 28, the FDA’s Gastrointestinal Drugs Advisory Committee voted 15-2 that the expected benefits of adalimumab – a subcutaneously administered tumor necrosis factor (TNF) blocker – outweighed its potential risks as a treatment for patients with moderately to severely active ulcerative colitis (UC) who have not had an adequate response with conventional treatments. The panel’s recommendation for approval was based on the results of two studies comparing adalimumab against placebo in such patients. But their support for approval came with caveats. While most of the panel agreed that the dose used in the studies had been shown to be clinically effective, they agreed that the optimal dose for treating UC had not yet been determined and that post-approval studies were needed to address dosing, subpopulations of patients who may benefit most from treatment, and long-term safety.
Panel members voting in favor of approval cited the need for more treatments and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects. And although the differences in clinical remission rates between placebo and treatment at 8 weeks and at 1 year were less than 10% – one of the main issues raised by FDA reviewers – most of the panel members said that these differences still represented a clinically meaningful benefit. Infliximab (Remicade), an intravenous TNF blocker, is approved for treating UC and Crohn’s disease in pediatric and adult patients.
Like other panelists, Dr. Andelka LoSavio of the department of gastroenterology and nutrition, Loyola University, Maywood, Ill., said that the outstanding issues should not hold up approval of adalimumab for UC. "Once the drug is on the marketplace, we will have more insight into subpopulations where the drug may be more effective," which was the case with Crohn’s disease and adalimumab.
Although the remission rates in the studies were low, these are difficult-to-treat patients, and the responses were statistically significant, with evidence of a steroid-sparing effect, said panelist Dr. Marc Wishingrad, a gastroenterologist who practices in Santa Monica, Calif. "So it seems to me in a global sense that there is enough benefit here, that overall I would recommend that this drug be approved for this indication," he added.
Panelists supported the manufacturer’s recommendation that if a patient does not respond after 8 weeks of treatment, that treatment should be stopped.
The two statisticians on the panel did not support approval, voting no on the risk-benefit question for reasons that included the modest effects on clinical remission rates, uncertain durability of these effects, inadequate long-term data, missing data, and uncertainties about the dose.
Adalimumab – marketed as Humira by Abbott Laboratories – was first approved in 2002 for treating moderate to severe rheumatoid arthritis in adults, and was approved in 2007 for treating moderate to severe Crohn’s disease. It has also been approved for psoriasis, ankylosing spondylitis, and juvenile idiopathic arthritis indications.
The recommended dosing schedule for UC is one 160-mg starting dose, followed by 80 mg on day 15, and then 40 mg every other week starting at day 29, continuing treatment only in people who have responded within the first 8 weeks.
Abbott filed for approval in January 2011, but the FDA raised questions about whether a higher dose would be more effective and about the strength of the results in the two pivotal trials, including whether the differences in clinical remission rates at 8 and 52 weeks between placebo and treatment were clinically meaningful. The company resubmitted the application for approval, with a slightly different proposed indication: reducing the signs and symptoms, and achieving (instead of inducing and maintaining induction of) clinical remission in adults with moderately to severely active UC, who have had an inadequate response to conventional therapy.
The two double-blind, placebo-controlled, phase III studies compared adalimumab against placebo in 1,094 treatment-refractory patients with moderately to severely active UC with a total Mayo score of 6 to 12 points, and an endoscopy sub-score of 2 or 3, despite current or previous steroid and/or immunosuppressant therapy. In one 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference that was significant.
In the second study, which followed patients for 1 year and included some who had been treated with infliximab (40%), the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference that was statistically significant. At 52 weeks, the remission rate was 17.3% among those on adalimumab, vs. 8.5% among those on placebo, an 8.8% difference that also was statistically significant.
In different votes, the panel nearly unanimously agreed that the 8 week and 52 week clinical remission rates in the studies represented "clinically meaningful benefits."
However, in the second study, 8.5% of the patients on adalimumab were in clinical remission at both weeks 8 and 52, vs. 4.1% among those on placebo. The panel was less unanimous about whether the 4.4% difference between the two groups represented a clinically meaningful benefit of treatment, voting 10-6 with 1 abstention on this question.
Other findings in the 52-week study included a higher steroid discontinuation rate among those on adalimumab, a secondary endpoint: About 13% of those on adalimumab were able to go off steroid treatment before the 52nd week of treatment and had achieved a clinical remission, compared with 5.7% of those on placebo, a statistically significant difference. In addition, the patients on azathioprine or 6-MP at baseline in this study did not appear to benefit from adalimumab: In this subgroup, the remission rate at week 8 among those on adalimumab (12.9%) was similar to the rate among those on placebo (15%). But for those who were not on the immunomodulatory drugs at baseline, the remission rate was nearly 19% among those on adalimumab vs. 6.6% of those on placebo, the FDA reviewer pointed out. In the studies, the safety profile of adalimumab was similar to the placebo, other than a higher rate of injection site reactions among those on adalimumab, and no new safety signals for adalimumab were identified in the trials, according to Abbott.
If the FDA grants approval, adalimumab will be the only self-administered biologic therapy available for patients with UC, the company pointed out. In April 2012, adalimumab was approved in the European Union for treating adults with moderately to severely active UC who have had an inadequate response to conventional therapy, based on the same data submitted to the FDA for the UC indication, according to Abbott.
The company is planning an international registry of patients with UC treated with adalimumab in typical clinical settings to collect long-term safety data, which will include patients in the EU, and, if approved, in the United States. The FDA usually follows the recommendations of its advisory panels, which are not binding. The company expects a decision on approval by the end of the year, according to an Abbott spokesperson.
Panelists have been cleared of potential conflicts of interest related to the topic of the meeting. Occasionally, a panelist may be given a waiver, but not at this meeting.
Noroviruses Affecting Diverse Populations
SAN FRANCISCO – As the availability of more sensitive diagnostic methods such as reverse transcription-polymerase chain reaction testing become more widespread, noroviruses are increasingly being recognized as important enteric pathogens in diverse populations.
"In the past, much of our knowledge about noroviruses has been hindered because we don’t have a good animal model or method for culturing norovirus," Dr. Hoonmo L. Koo said at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. "The majority of our current understanding has come from studying human outbreaks, volunteer challenge studies, and evaluating surrogate caliciviruses such as feline and murine caliciviruses, which don’t cause human infection but can be cultured."
Noroviruses (NoVs) are classified into five genetic groups based on their RNA capsid sequences, with genogroup I (GI) and genogroup II (GII) NoVs causing the most human NoV infections. GII.4 NoV strains are the predominant circulating genotype in the United States and worldwide, said Dr. Koo of Baylor College of Medicine, Houston. As reverse transcription-polymerase chain reaction testing (RT-PCR) has become more available in recent years, NoVs "are now recognized as the most definable common cause of acute nonbacterial gastroenteritis worldwide," he said. "They account for approximately half of all food-borne illness in the United States.
"Each year, NoVs cause about 21 million cases of infection in the United States. They occur throughout the year, but they peak in the winter season."
NoV outbreaks are common in children, travelers, restaurant patrons, military personnel, patients, and health care staff at hospitals, nursing homes, and other medical facilities. Dr. Koo and his associates conducted an 8.5-year surveillance study at Texas Children’s Hospital (TCH) investigating NoV, rotavirus (RV), and adenovirus prevalence at the facility before and after introduction of the RV vaccine in 2006. The study evaluated 8,173 stool samples from inpatients and outpatients at TCH from February 2002 to June 2010. The samples were evaluated for RV by antigen detection or electron microscopy and adenoviruses by electron microscopy. In addition, a subset of 3,222 stools were evaluated for NoV by RT-PCR (J. Ped. Infect. Dis. 2012 Aug. 3 [doi:10.1093/jpids/pis070]).
"We found that RV prevalence decreased significantly after the introduction of the RV vaccine in 2006," Dr. Koo said at the meeting, which was sponsored by the American Society for Microbiology. "In more recent years, it decreased from about 9% in 2007 to 3% in 2010." At the same time, he continued, "NoV prevalence increased in 2004 and was consistently between 11% and 17% from 2004 to 2010. There was no significant increase in NoV prevalence after the RV vaccine was introduced in 2006."
The researchers concluded that NoVs have emerged as the most common viral gastroenteritis pathogen at TCH, which is one of the largest pediatric hospitals in the United States. "We believe that as RV prevalence continues to decline with vaccination, NoVs will soon eclipse rotaviruses as the most important cause of pediatric gastroenteritis in the United States and other countries where the RV vaccine is successfully administered," Dr. Koo said.
In a separate study, he and his associates evaluated stools from 571 international travelers who acquired diarrhea in Guatemala, India, and Mexico (J. Clin. Microbiol. 2010;48:1673-6). NoVs were identified in 10% of cases of travelers’ diarrhea, and overall were the second most common pathogen following diarrheagenic Escherichia coli. "We concluded that NoVs are important pathogens of travelers’ diarrhea in multiple developing regions of the world," Dr. Koo said. "However, there was significant variation evident in the prevalence of NoV diarrhea and in the predominant genogroup infecting the travelers, depending on the specific geographic location and time period we looked at."
Immunocompromised patients also have been affected by NoVs. One report described 12 hematopoietic stem cell transplant patients with NoV gastroenteritis who were hospitalized for a median of 73 days (Clin. Infect. Dis. 2009;49:1069-71). Half of the patients required supplemental feeding with enteral or parenteral nutrition, and two patients died: one secondary to malnutrition and chronic NoV gastroenteritis. "Future areas of NoV study include further defining the burden of disease in pediatric and immunocompromised populations," Dr. Koo said. "We need development of sensitive diagnostic assays that can be used by clinical laboratories. Unfortunately, the ELISA assay that we have now for NoV detection is relatively insensitive, and most clinical laboratories cannot perform RT-PCR. We need effective therapeutic agents to be developed, and we need an effective NoV vaccine."
Results from a recent randomized, double-blind, placebo-controlled trial of an investigational NoV vaccine found a significantly lower frequency of viral gastroenteritis among vaccine recipients, compared with placebo recipients (37% vs. 69%, respectively; P = .006). It was found to be safe and well tolerated, and no severe adverse events were reported (N. Engl. J. Med. 2011;365:2178-87). "The vaccine was also found to be immunogenic, with 70% of vaccine recipients producing a fourfold rise in serum total antibody and serum IgA," Dr. Koo said. "However, there are significant challenges to the development of NoV vaccines. In the NEJM study, the frequency and magnitude of serum antibody response with the vaccine was lower than what’s been observed with natural infection. Previous studies have shown that acquired immunity with natural infection may be short-lived: less than 2 years. So how long will the vaccine protection last?"
Another challenge, he said, is that antibodies are only cross-protective within the same genogroup. "Future NoV vaccines will need to be composed of both GI and GII virus–like particles (VLPs)," he said. "Finally, significant antigenic diversity of noroviruses and the constant antigenic drift may require active surveillance similar to what we have for influenza viruses, where we are constantly surveying what strains are in the community and changing the composition of vaccine VLP strains to represent these important community strains."
Dr. Koo disclosed that he has received research support from the National Institute of Diabetes and Digestive and Kidney Diseases, and the Baylor College of Medicine Center for Globalization.
SAN FRANCISCO – As the availability of more sensitive diagnostic methods such as reverse transcription-polymerase chain reaction testing become more widespread, noroviruses are increasingly being recognized as important enteric pathogens in diverse populations.
"In the past, much of our knowledge about noroviruses has been hindered because we don’t have a good animal model or method for culturing norovirus," Dr. Hoonmo L. Koo said at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. "The majority of our current understanding has come from studying human outbreaks, volunteer challenge studies, and evaluating surrogate caliciviruses such as feline and murine caliciviruses, which don’t cause human infection but can be cultured."
Noroviruses (NoVs) are classified into five genetic groups based on their RNA capsid sequences, with genogroup I (GI) and genogroup II (GII) NoVs causing the most human NoV infections. GII.4 NoV strains are the predominant circulating genotype in the United States and worldwide, said Dr. Koo of Baylor College of Medicine, Houston. As reverse transcription-polymerase chain reaction testing (RT-PCR) has become more available in recent years, NoVs "are now recognized as the most definable common cause of acute nonbacterial gastroenteritis worldwide," he said. "They account for approximately half of all food-borne illness in the United States.
"Each year, NoVs cause about 21 million cases of infection in the United States. They occur throughout the year, but they peak in the winter season."
NoV outbreaks are common in children, travelers, restaurant patrons, military personnel, patients, and health care staff at hospitals, nursing homes, and other medical facilities. Dr. Koo and his associates conducted an 8.5-year surveillance study at Texas Children’s Hospital (TCH) investigating NoV, rotavirus (RV), and adenovirus prevalence at the facility before and after introduction of the RV vaccine in 2006. The study evaluated 8,173 stool samples from inpatients and outpatients at TCH from February 2002 to June 2010. The samples were evaluated for RV by antigen detection or electron microscopy and adenoviruses by electron microscopy. In addition, a subset of 3,222 stools were evaluated for NoV by RT-PCR (J. Ped. Infect. Dis. 2012 Aug. 3 [doi:10.1093/jpids/pis070]).
"We found that RV prevalence decreased significantly after the introduction of the RV vaccine in 2006," Dr. Koo said at the meeting, which was sponsored by the American Society for Microbiology. "In more recent years, it decreased from about 9% in 2007 to 3% in 2010." At the same time, he continued, "NoV prevalence increased in 2004 and was consistently between 11% and 17% from 2004 to 2010. There was no significant increase in NoV prevalence after the RV vaccine was introduced in 2006."
The researchers concluded that NoVs have emerged as the most common viral gastroenteritis pathogen at TCH, which is one of the largest pediatric hospitals in the United States. "We believe that as RV prevalence continues to decline with vaccination, NoVs will soon eclipse rotaviruses as the most important cause of pediatric gastroenteritis in the United States and other countries where the RV vaccine is successfully administered," Dr. Koo said.
In a separate study, he and his associates evaluated stools from 571 international travelers who acquired diarrhea in Guatemala, India, and Mexico (J. Clin. Microbiol. 2010;48:1673-6). NoVs were identified in 10% of cases of travelers’ diarrhea, and overall were the second most common pathogen following diarrheagenic Escherichia coli. "We concluded that NoVs are important pathogens of travelers’ diarrhea in multiple developing regions of the world," Dr. Koo said. "However, there was significant variation evident in the prevalence of NoV diarrhea and in the predominant genogroup infecting the travelers, depending on the specific geographic location and time period we looked at."
Immunocompromised patients also have been affected by NoVs. One report described 12 hematopoietic stem cell transplant patients with NoV gastroenteritis who were hospitalized for a median of 73 days (Clin. Infect. Dis. 2009;49:1069-71). Half of the patients required supplemental feeding with enteral or parenteral nutrition, and two patients died: one secondary to malnutrition and chronic NoV gastroenteritis. "Future areas of NoV study include further defining the burden of disease in pediatric and immunocompromised populations," Dr. Koo said. "We need development of sensitive diagnostic assays that can be used by clinical laboratories. Unfortunately, the ELISA assay that we have now for NoV detection is relatively insensitive, and most clinical laboratories cannot perform RT-PCR. We need effective therapeutic agents to be developed, and we need an effective NoV vaccine."
Results from a recent randomized, double-blind, placebo-controlled trial of an investigational NoV vaccine found a significantly lower frequency of viral gastroenteritis among vaccine recipients, compared with placebo recipients (37% vs. 69%, respectively; P = .006). It was found to be safe and well tolerated, and no severe adverse events were reported (N. Engl. J. Med. 2011;365:2178-87). "The vaccine was also found to be immunogenic, with 70% of vaccine recipients producing a fourfold rise in serum total antibody and serum IgA," Dr. Koo said. "However, there are significant challenges to the development of NoV vaccines. In the NEJM study, the frequency and magnitude of serum antibody response with the vaccine was lower than what’s been observed with natural infection. Previous studies have shown that acquired immunity with natural infection may be short-lived: less than 2 years. So how long will the vaccine protection last?"
Another challenge, he said, is that antibodies are only cross-protective within the same genogroup. "Future NoV vaccines will need to be composed of both GI and GII virus–like particles (VLPs)," he said. "Finally, significant antigenic diversity of noroviruses and the constant antigenic drift may require active surveillance similar to what we have for influenza viruses, where we are constantly surveying what strains are in the community and changing the composition of vaccine VLP strains to represent these important community strains."
Dr. Koo disclosed that he has received research support from the National Institute of Diabetes and Digestive and Kidney Diseases, and the Baylor College of Medicine Center for Globalization.
SAN FRANCISCO – As the availability of more sensitive diagnostic methods such as reverse transcription-polymerase chain reaction testing become more widespread, noroviruses are increasingly being recognized as important enteric pathogens in diverse populations.
"In the past, much of our knowledge about noroviruses has been hindered because we don’t have a good animal model or method for culturing norovirus," Dr. Hoonmo L. Koo said at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. "The majority of our current understanding has come from studying human outbreaks, volunteer challenge studies, and evaluating surrogate caliciviruses such as feline and murine caliciviruses, which don’t cause human infection but can be cultured."
Noroviruses (NoVs) are classified into five genetic groups based on their RNA capsid sequences, with genogroup I (GI) and genogroup II (GII) NoVs causing the most human NoV infections. GII.4 NoV strains are the predominant circulating genotype in the United States and worldwide, said Dr. Koo of Baylor College of Medicine, Houston. As reverse transcription-polymerase chain reaction testing (RT-PCR) has become more available in recent years, NoVs "are now recognized as the most definable common cause of acute nonbacterial gastroenteritis worldwide," he said. "They account for approximately half of all food-borne illness in the United States.
"Each year, NoVs cause about 21 million cases of infection in the United States. They occur throughout the year, but they peak in the winter season."
NoV outbreaks are common in children, travelers, restaurant patrons, military personnel, patients, and health care staff at hospitals, nursing homes, and other medical facilities. Dr. Koo and his associates conducted an 8.5-year surveillance study at Texas Children’s Hospital (TCH) investigating NoV, rotavirus (RV), and adenovirus prevalence at the facility before and after introduction of the RV vaccine in 2006. The study evaluated 8,173 stool samples from inpatients and outpatients at TCH from February 2002 to June 2010. The samples were evaluated for RV by antigen detection or electron microscopy and adenoviruses by electron microscopy. In addition, a subset of 3,222 stools were evaluated for NoV by RT-PCR (J. Ped. Infect. Dis. 2012 Aug. 3 [doi:10.1093/jpids/pis070]).
"We found that RV prevalence decreased significantly after the introduction of the RV vaccine in 2006," Dr. Koo said at the meeting, which was sponsored by the American Society for Microbiology. "In more recent years, it decreased from about 9% in 2007 to 3% in 2010." At the same time, he continued, "NoV prevalence increased in 2004 and was consistently between 11% and 17% from 2004 to 2010. There was no significant increase in NoV prevalence after the RV vaccine was introduced in 2006."
The researchers concluded that NoVs have emerged as the most common viral gastroenteritis pathogen at TCH, which is one of the largest pediatric hospitals in the United States. "We believe that as RV prevalence continues to decline with vaccination, NoVs will soon eclipse rotaviruses as the most important cause of pediatric gastroenteritis in the United States and other countries where the RV vaccine is successfully administered," Dr. Koo said.
In a separate study, he and his associates evaluated stools from 571 international travelers who acquired diarrhea in Guatemala, India, and Mexico (J. Clin. Microbiol. 2010;48:1673-6). NoVs were identified in 10% of cases of travelers’ diarrhea, and overall were the second most common pathogen following diarrheagenic Escherichia coli. "We concluded that NoVs are important pathogens of travelers’ diarrhea in multiple developing regions of the world," Dr. Koo said. "However, there was significant variation evident in the prevalence of NoV diarrhea and in the predominant genogroup infecting the travelers, depending on the specific geographic location and time period we looked at."
Immunocompromised patients also have been affected by NoVs. One report described 12 hematopoietic stem cell transplant patients with NoV gastroenteritis who were hospitalized for a median of 73 days (Clin. Infect. Dis. 2009;49:1069-71). Half of the patients required supplemental feeding with enteral or parenteral nutrition, and two patients died: one secondary to malnutrition and chronic NoV gastroenteritis. "Future areas of NoV study include further defining the burden of disease in pediatric and immunocompromised populations," Dr. Koo said. "We need development of sensitive diagnostic assays that can be used by clinical laboratories. Unfortunately, the ELISA assay that we have now for NoV detection is relatively insensitive, and most clinical laboratories cannot perform RT-PCR. We need effective therapeutic agents to be developed, and we need an effective NoV vaccine."
Results from a recent randomized, double-blind, placebo-controlled trial of an investigational NoV vaccine found a significantly lower frequency of viral gastroenteritis among vaccine recipients, compared with placebo recipients (37% vs. 69%, respectively; P = .006). It was found to be safe and well tolerated, and no severe adverse events were reported (N. Engl. J. Med. 2011;365:2178-87). "The vaccine was also found to be immunogenic, with 70% of vaccine recipients producing a fourfold rise in serum total antibody and serum IgA," Dr. Koo said. "However, there are significant challenges to the development of NoV vaccines. In the NEJM study, the frequency and magnitude of serum antibody response with the vaccine was lower than what’s been observed with natural infection. Previous studies have shown that acquired immunity with natural infection may be short-lived: less than 2 years. So how long will the vaccine protection last?"
Another challenge, he said, is that antibodies are only cross-protective within the same genogroup. "Future NoV vaccines will need to be composed of both GI and GII virus–like particles (VLPs)," he said. "Finally, significant antigenic diversity of noroviruses and the constant antigenic drift may require active surveillance similar to what we have for influenza viruses, where we are constantly surveying what strains are in the community and changing the composition of vaccine VLP strains to represent these important community strains."
Dr. Koo disclosed that he has received research support from the National Institute of Diabetes and Digestive and Kidney Diseases, and the Baylor College of Medicine Center for Globalization.
AT THE ANNUAL INTERSCIENCE CONFERENCE ON ANTIMICROBIAL AGENTS AND CHEMOTHERAPY
Promising C. difficile Antibiotic in Pipeline
SAN FRANCISCO – Those desperate for new treatments for Clostridium difficile infection may want to keep an eye on the experimental oral antibiotic cadazolid, which looked promising in an early-phase trial, according to Daniela Baldoni, Pharm.D.
Cadazolid produced low systemic exposure with high concentrations at the desired site – the colon – and was well tolerated in 64 healthy men who received up to 3,000 mg b.i.d. for 10 days, she reported in a poster presentation at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. Dr. Baldoni is employed by Actelion Pharmaceuticals, the company that is developing cadazolid.
The study randomized nonsmoking men to either the drug (single or multiple doses of cadazolid) or placebo. All subjects remained in the clinic for observation for 120 hours after the last dose.
In the single-dose group, 30 fasting subjects received a single dose of 30, 100, 300, 1,000, or 3,000 mg cadazolid and 10 subjects received matching placebo. After a wash-out period of 8-15 days, the six subjects who had taken 300 mg received a second dose of 300 mg after eating instead of after fasting. In the multiple-dose group, 18 subjects took 300, 1,000, or 3,000 mg of cadazolid twice a day and 6 received matching placebo for 10 days.
Taking cadazolid with food appeared to increase the rate and extent of drug absorption by two- to fivefold. Blood samples showed low systemic exposure after single or multiple doses, with a minor, twofold increase in cadazolid in plasma after 10 days for all doses in the twice-a-day group, Dr. Baldoni reported at the meeting, sponsored by the American Society for Microbiology.
Negligible recovery of unchanged cadazolid in urine samples and the fact that most of the dose was recovered in feces suggests that treatment produced high concentrations at the colon. In fecal samples collected during days 6 and 10 of treatment, the mean cumulative recovery of unchanged cadazolid in feces was between 87% and 94%.
There were no significant changes in vital signs, ECG results, or laboratory parameters compared with baseline measurements, and no drug-related serious adverse events. All subjects completed the study except one man in the 100-mg single-dose subgroup who withdrew consent for reasons unrelated to adverse events.
The dose or duration of treatment did not seem to affect the number of adverse events.
In the single-dose group, 8 of 30 subjects on cadazolid (27%) and 4 of 10 on placebo (40%) reported adverse events, most commonly headache and diarrhea. Four subjects on cadazolid (13%) and two on placebo (20%) reported headache, and three subjects on cadazolid (10%) and one on placebo (10%) reported diarrhea.
In the twice-a-day dosing group, 7 of 18 subjects on cadazolid (39%) and 1 of 6 subjects on placebo (17%) reported adverse events, most commonly headache in 5 subjects on cadazolid (28%) and 1 subject on placebo (17%).
Subjects were aged 45-60 years and had a body mass index of 18-32 kg/m2.
C. difficile is the most common cause of antibiotic-associated infectious diarrhea, especially among the elderly. The mainstays of treating C. difficile–associated infection, metronidazole or vancomycin, fail in approximately 20%-45% of patients.
Cadazolid is in the oxazolidinone class of antibiotics. Its mechanism of action consists mainly of bacterial protein-synthesis inhibition.
Dr. Baldoni and most of her coinvestigators are employees of Actelion Pharmaceuticals, which funded the study.
SAN FRANCISCO – Those desperate for new treatments for Clostridium difficile infection may want to keep an eye on the experimental oral antibiotic cadazolid, which looked promising in an early-phase trial, according to Daniela Baldoni, Pharm.D.
Cadazolid produced low systemic exposure with high concentrations at the desired site – the colon – and was well tolerated in 64 healthy men who received up to 3,000 mg b.i.d. for 10 days, she reported in a poster presentation at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. Dr. Baldoni is employed by Actelion Pharmaceuticals, the company that is developing cadazolid.
The study randomized nonsmoking men to either the drug (single or multiple doses of cadazolid) or placebo. All subjects remained in the clinic for observation for 120 hours after the last dose.
In the single-dose group, 30 fasting subjects received a single dose of 30, 100, 300, 1,000, or 3,000 mg cadazolid and 10 subjects received matching placebo. After a wash-out period of 8-15 days, the six subjects who had taken 300 mg received a second dose of 300 mg after eating instead of after fasting. In the multiple-dose group, 18 subjects took 300, 1,000, or 3,000 mg of cadazolid twice a day and 6 received matching placebo for 10 days.
Taking cadazolid with food appeared to increase the rate and extent of drug absorption by two- to fivefold. Blood samples showed low systemic exposure after single or multiple doses, with a minor, twofold increase in cadazolid in plasma after 10 days for all doses in the twice-a-day group, Dr. Baldoni reported at the meeting, sponsored by the American Society for Microbiology.
Negligible recovery of unchanged cadazolid in urine samples and the fact that most of the dose was recovered in feces suggests that treatment produced high concentrations at the colon. In fecal samples collected during days 6 and 10 of treatment, the mean cumulative recovery of unchanged cadazolid in feces was between 87% and 94%.
There were no significant changes in vital signs, ECG results, or laboratory parameters compared with baseline measurements, and no drug-related serious adverse events. All subjects completed the study except one man in the 100-mg single-dose subgroup who withdrew consent for reasons unrelated to adverse events.
The dose or duration of treatment did not seem to affect the number of adverse events.
In the single-dose group, 8 of 30 subjects on cadazolid (27%) and 4 of 10 on placebo (40%) reported adverse events, most commonly headache and diarrhea. Four subjects on cadazolid (13%) and two on placebo (20%) reported headache, and three subjects on cadazolid (10%) and one on placebo (10%) reported diarrhea.
In the twice-a-day dosing group, 7 of 18 subjects on cadazolid (39%) and 1 of 6 subjects on placebo (17%) reported adverse events, most commonly headache in 5 subjects on cadazolid (28%) and 1 subject on placebo (17%).
Subjects were aged 45-60 years and had a body mass index of 18-32 kg/m2.
C. difficile is the most common cause of antibiotic-associated infectious diarrhea, especially among the elderly. The mainstays of treating C. difficile–associated infection, metronidazole or vancomycin, fail in approximately 20%-45% of patients.
Cadazolid is in the oxazolidinone class of antibiotics. Its mechanism of action consists mainly of bacterial protein-synthesis inhibition.
Dr. Baldoni and most of her coinvestigators are employees of Actelion Pharmaceuticals, which funded the study.
SAN FRANCISCO – Those desperate for new treatments for Clostridium difficile infection may want to keep an eye on the experimental oral antibiotic cadazolid, which looked promising in an early-phase trial, according to Daniela Baldoni, Pharm.D.
Cadazolid produced low systemic exposure with high concentrations at the desired site – the colon – and was well tolerated in 64 healthy men who received up to 3,000 mg b.i.d. for 10 days, she reported in a poster presentation at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy. Dr. Baldoni is employed by Actelion Pharmaceuticals, the company that is developing cadazolid.
The study randomized nonsmoking men to either the drug (single or multiple doses of cadazolid) or placebo. All subjects remained in the clinic for observation for 120 hours after the last dose.
In the single-dose group, 30 fasting subjects received a single dose of 30, 100, 300, 1,000, or 3,000 mg cadazolid and 10 subjects received matching placebo. After a wash-out period of 8-15 days, the six subjects who had taken 300 mg received a second dose of 300 mg after eating instead of after fasting. In the multiple-dose group, 18 subjects took 300, 1,000, or 3,000 mg of cadazolid twice a day and 6 received matching placebo for 10 days.
Taking cadazolid with food appeared to increase the rate and extent of drug absorption by two- to fivefold. Blood samples showed low systemic exposure after single or multiple doses, with a minor, twofold increase in cadazolid in plasma after 10 days for all doses in the twice-a-day group, Dr. Baldoni reported at the meeting, sponsored by the American Society for Microbiology.
Negligible recovery of unchanged cadazolid in urine samples and the fact that most of the dose was recovered in feces suggests that treatment produced high concentrations at the colon. In fecal samples collected during days 6 and 10 of treatment, the mean cumulative recovery of unchanged cadazolid in feces was between 87% and 94%.
There were no significant changes in vital signs, ECG results, or laboratory parameters compared with baseline measurements, and no drug-related serious adverse events. All subjects completed the study except one man in the 100-mg single-dose subgroup who withdrew consent for reasons unrelated to adverse events.
The dose or duration of treatment did not seem to affect the number of adverse events.
In the single-dose group, 8 of 30 subjects on cadazolid (27%) and 4 of 10 on placebo (40%) reported adverse events, most commonly headache and diarrhea. Four subjects on cadazolid (13%) and two on placebo (20%) reported headache, and three subjects on cadazolid (10%) and one on placebo (10%) reported diarrhea.
In the twice-a-day dosing group, 7 of 18 subjects on cadazolid (39%) and 1 of 6 subjects on placebo (17%) reported adverse events, most commonly headache in 5 subjects on cadazolid (28%) and 1 subject on placebo (17%).
Subjects were aged 45-60 years and had a body mass index of 18-32 kg/m2.
C. difficile is the most common cause of antibiotic-associated infectious diarrhea, especially among the elderly. The mainstays of treating C. difficile–associated infection, metronidazole or vancomycin, fail in approximately 20%-45% of patients.
Cadazolid is in the oxazolidinone class of antibiotics. Its mechanism of action consists mainly of bacterial protein-synthesis inhibition.
Dr. Baldoni and most of her coinvestigators are employees of Actelion Pharmaceuticals, which funded the study.
AT THE ANNUAL INTERSCIENCE CONFERENCE ON ANTIMICROBIAL AGENTS AND CHEMOTHERAPY
Major Finding: The experimental antibiotic cadazolid concentrated in feces with low systemic exposure and few side effects after single doses or twice-a-day dosing for 10 days.
Data Source: Data are from a randomized, placebo-controlled study in 64 healthy, nonsmoking men.
Disclosures: Dr. Baldoni and most of her coinvestigators are employees of Actelion Pharmaceuticals, which funded the study.
FDA Approves Linaclotide for Constipation Conditions
The Food and Drug Administration approved linaclotide on Aug. 30 to treat two conditions: chronic idiopathic constipation and irritable bowel syndrome with constipation in adults.
Linaclotide (Linzess) is administered as a capsule taken once daily on an empty stomach, at least 30 minutes before the first meal of the day. This agent helps relieve constipation by increasing the frequency of bowel movements. In irritable bowel syndrome with constipation (IBS-C), linaclotide has been shown to reduce abdominal pain, according to a statement from the FDA.
The drug is approved with a boxed warning to alert patients and health care professionals that linaclotide should not be used in patients 16 years of age and younger. The most common side effect reported during the clinical studies was diarrhea, the statement said.
According to the FDA, the safety and effectiveness of linaclotide for the management of IBS-C were established in two double-blind studies (Gastroenterology 2011;140:S138 and Gastroenterology 2011;140:S135). A total of 1,604 patients were randomly assigned to take 290 mcg of linaclotide or a placebo for at least 12 weeks. Linaclotide was more effective in reducing abdominal pain and increasing the number of complete spontaneous bowel movements, compared with placebo, in both trials.
The safety and effectiveness of linaclotide for the management of chronic idiopathic constipation also were established in two double-blind studies (N. Engl. J. Med. 2011;365:527-36). A total of 1,272 patients were randomly assigned to take 145 mcg or 290 mcg linaclotide or a placebo for 12 weeks. Patients on linaclotide had more complete spontaneous bowel movements than did those taking the placebo. The 290-mcg dose is not approved for chronic constipation because the data showed that it was no more effective than the 145-mcg dose.
Linzess is marketed by Ironwood Pharmaceuticals Inc.
Linaclotide is currently the only FDA-approved medication indicated for increasing bowel movements and decreasing abdominal pain in men and women with irritable bowel syndrome with constipation (IBS-C). It has been shown to be efficacious in relieving abdominal pain and constipation in patients with IBS-C, and constipation in those with chronic idiopathic constipation (CIC). The drug is a peripherally-acting agent that activates guanylate cyclase-C (GC-C) on intestinal epithelial cells resulting in increased intracellular and extracellular concentrations of cyclic guanosine monophosphate (cGMP).
Relief of constipation symptoms in IBS-C and CIC is believed to be due to an increase in intracellular cGMP resulting in chloride and fluid secretion through the cystic fibrosis transmembrane conductance regulator (CFTR) ion channel and acceleration of colonic transit. Linaclotide’s effect on reducing abdominal pain in IBS-C is thought to be due to increased extracellular cGMP, which has been shown to decrease firing of sensory nerves within the bowel wall in preclinical animal studies.
Patients with CIC who responded to linaclotide had at least three complete spontaneous bowel movements (CSBMs) per week and an increase in one CSBM for at least 9 out of 12 weeks. The 145 mcg and 290 mcg daily doses showed a statistically significant benefit over placebo; the FDA has approved only the lower dose for CIC. The efficacy of linaclotide was sustained throughout the 12 weeks of the trials.
The dose of 290 mcg per day was approved for the treatment of IBS-C, which is usually differentiated from CIC by the presence of predominant abdominal pain associated with constipation. The significant improvement in CSBMs occurred within the first week of treatment. The decrease in abdominal pain was more gradual and appeared to reach its maximum effect at 8 weeks. The significant effect of linaclotide on abdominal pain may be due to an additional independent effect beyond relief of constipation, but further studies are needed to better understand linaclotide’s effect on abdominal pain.
LIN CHANG, M.D., is co-director of the Oppenheimer Family Center for Neurobiology of Stress and director of the Digestive Health and Nutrition Clinic at the University of California, Los Angeles. She is a consultant for Ironwood Pharmaceuticals and Forest Laboratories and has received grant support from Ironwood Pharmaceuticals.
Linaclotide is currently the only FDA-approved medication indicated for increasing bowel movements and decreasing abdominal pain in men and women with irritable bowel syndrome with constipation (IBS-C). It has been shown to be efficacious in relieving abdominal pain and constipation in patients with IBS-C, and constipation in those with chronic idiopathic constipation (CIC). The drug is a peripherally-acting agent that activates guanylate cyclase-C (GC-C) on intestinal epithelial cells resulting in increased intracellular and extracellular concentrations of cyclic guanosine monophosphate (cGMP).
Relief of constipation symptoms in IBS-C and CIC is believed to be due to an increase in intracellular cGMP resulting in chloride and fluid secretion through the cystic fibrosis transmembrane conductance regulator (CFTR) ion channel and acceleration of colonic transit. Linaclotide’s effect on reducing abdominal pain in IBS-C is thought to be due to increased extracellular cGMP, which has been shown to decrease firing of sensory nerves within the bowel wall in preclinical animal studies.
Patients with CIC who responded to linaclotide had at least three complete spontaneous bowel movements (CSBMs) per week and an increase in one CSBM for at least 9 out of 12 weeks. The 145 mcg and 290 mcg daily doses showed a statistically significant benefit over placebo; the FDA has approved only the lower dose for CIC. The efficacy of linaclotide was sustained throughout the 12 weeks of the trials.
The dose of 290 mcg per day was approved for the treatment of IBS-C, which is usually differentiated from CIC by the presence of predominant abdominal pain associated with constipation. The significant improvement in CSBMs occurred within the first week of treatment. The decrease in abdominal pain was more gradual and appeared to reach its maximum effect at 8 weeks. The significant effect of linaclotide on abdominal pain may be due to an additional independent effect beyond relief of constipation, but further studies are needed to better understand linaclotide’s effect on abdominal pain.
LIN CHANG, M.D., is co-director of the Oppenheimer Family Center for Neurobiology of Stress and director of the Digestive Health and Nutrition Clinic at the University of California, Los Angeles. She is a consultant for Ironwood Pharmaceuticals and Forest Laboratories and has received grant support from Ironwood Pharmaceuticals.
Linaclotide is currently the only FDA-approved medication indicated for increasing bowel movements and decreasing abdominal pain in men and women with irritable bowel syndrome with constipation (IBS-C). It has been shown to be efficacious in relieving abdominal pain and constipation in patients with IBS-C, and constipation in those with chronic idiopathic constipation (CIC). The drug is a peripherally-acting agent that activates guanylate cyclase-C (GC-C) on intestinal epithelial cells resulting in increased intracellular and extracellular concentrations of cyclic guanosine monophosphate (cGMP).
Relief of constipation symptoms in IBS-C and CIC is believed to be due to an increase in intracellular cGMP resulting in chloride and fluid secretion through the cystic fibrosis transmembrane conductance regulator (CFTR) ion channel and acceleration of colonic transit. Linaclotide’s effect on reducing abdominal pain in IBS-C is thought to be due to increased extracellular cGMP, which has been shown to decrease firing of sensory nerves within the bowel wall in preclinical animal studies.
Patients with CIC who responded to linaclotide had at least three complete spontaneous bowel movements (CSBMs) per week and an increase in one CSBM for at least 9 out of 12 weeks. The 145 mcg and 290 mcg daily doses showed a statistically significant benefit over placebo; the FDA has approved only the lower dose for CIC. The efficacy of linaclotide was sustained throughout the 12 weeks of the trials.
The dose of 290 mcg per day was approved for the treatment of IBS-C, which is usually differentiated from CIC by the presence of predominant abdominal pain associated with constipation. The significant improvement in CSBMs occurred within the first week of treatment. The decrease in abdominal pain was more gradual and appeared to reach its maximum effect at 8 weeks. The significant effect of linaclotide on abdominal pain may be due to an additional independent effect beyond relief of constipation, but further studies are needed to better understand linaclotide’s effect on abdominal pain.
LIN CHANG, M.D., is co-director of the Oppenheimer Family Center for Neurobiology of Stress and director of the Digestive Health and Nutrition Clinic at the University of California, Los Angeles. She is a consultant for Ironwood Pharmaceuticals and Forest Laboratories and has received grant support from Ironwood Pharmaceuticals.
The Food and Drug Administration approved linaclotide on Aug. 30 to treat two conditions: chronic idiopathic constipation and irritable bowel syndrome with constipation in adults.
Linaclotide (Linzess) is administered as a capsule taken once daily on an empty stomach, at least 30 minutes before the first meal of the day. This agent helps relieve constipation by increasing the frequency of bowel movements. In irritable bowel syndrome with constipation (IBS-C), linaclotide has been shown to reduce abdominal pain, according to a statement from the FDA.
The drug is approved with a boxed warning to alert patients and health care professionals that linaclotide should not be used in patients 16 years of age and younger. The most common side effect reported during the clinical studies was diarrhea, the statement said.
According to the FDA, the safety and effectiveness of linaclotide for the management of IBS-C were established in two double-blind studies (Gastroenterology 2011;140:S138 and Gastroenterology 2011;140:S135). A total of 1,604 patients were randomly assigned to take 290 mcg of linaclotide or a placebo for at least 12 weeks. Linaclotide was more effective in reducing abdominal pain and increasing the number of complete spontaneous bowel movements, compared with placebo, in both trials.
The safety and effectiveness of linaclotide for the management of chronic idiopathic constipation also were established in two double-blind studies (N. Engl. J. Med. 2011;365:527-36). A total of 1,272 patients were randomly assigned to take 145 mcg or 290 mcg linaclotide or a placebo for 12 weeks. Patients on linaclotide had more complete spontaneous bowel movements than did those taking the placebo. The 290-mcg dose is not approved for chronic constipation because the data showed that it was no more effective than the 145-mcg dose.
Linzess is marketed by Ironwood Pharmaceuticals Inc.
The Food and Drug Administration approved linaclotide on Aug. 30 to treat two conditions: chronic idiopathic constipation and irritable bowel syndrome with constipation in adults.
Linaclotide (Linzess) is administered as a capsule taken once daily on an empty stomach, at least 30 minutes before the first meal of the day. This agent helps relieve constipation by increasing the frequency of bowel movements. In irritable bowel syndrome with constipation (IBS-C), linaclotide has been shown to reduce abdominal pain, according to a statement from the FDA.
The drug is approved with a boxed warning to alert patients and health care professionals that linaclotide should not be used in patients 16 years of age and younger. The most common side effect reported during the clinical studies was diarrhea, the statement said.
According to the FDA, the safety and effectiveness of linaclotide for the management of IBS-C were established in two double-blind studies (Gastroenterology 2011;140:S138 and Gastroenterology 2011;140:S135). A total of 1,604 patients were randomly assigned to take 290 mcg of linaclotide or a placebo for at least 12 weeks. Linaclotide was more effective in reducing abdominal pain and increasing the number of complete spontaneous bowel movements, compared with placebo, in both trials.
The safety and effectiveness of linaclotide for the management of chronic idiopathic constipation also were established in two double-blind studies (N. Engl. J. Med. 2011;365:527-36). A total of 1,272 patients were randomly assigned to take 145 mcg or 290 mcg linaclotide or a placebo for 12 weeks. Patients on linaclotide had more complete spontaneous bowel movements than did those taking the placebo. The 290-mcg dose is not approved for chronic constipation because the data showed that it was no more effective than the 145-mcg dose.
Linzess is marketed by Ironwood Pharmaceuticals Inc.