User login
Chronic constipation may increase colorectal cancer risk
Chronic constipation may predispose affected patients to developing colorectal cancer and benign neoplasms, according to an analysis of data from a large retrospective U.S. claims database.
The risk of developing colorectal cancer was 1.78 times higher among 28,854 adults with chronic constipation than among 86,562 controls without chronic constipation, and the risk of developing benign neoplasms was 2.7 times higher in those with chronic constipation, Dr. Nicholas Talley reported in a poster at the annual meeting of the American College of Gastroenterology.
The risk of colorectal cancer and benign neoplasms among those with chronic constipation remained "consistently high" after researchers controlled for potential confounding factors, including age, gender, family history of malignancies, and other nongastrointestinal morbidities, said Dr. Talley of the University of Newcastle, Callaghan, New South Wales, Australia.
Patients included adults aged older than 18 years who received at least two diagnoses of chronic constipation 60-365 days apart between January 1999 and September 2011. Those with irritable bowel syndrome or diarrhea were excluded, as were those who did not remain enrolled in their health plans for at least 12 months from the date of their first eligible diagnosis of constipation.
The investigators matched control subjects, who had never been diagnosed with constipation and never had a prescription filled for a laxative during the observation period, with case patients in a 1:3 ratio based on year of birth, sex, and region of residence.
Patients and controls had a mean age of 61.9 years, and one-third were men. The mean observation period was nearly 4 years.
The prevalence of colorectal cancer in this study was 2.7% in the patients and 1.7% in the controls; the prevalence of benign neoplasms was 24.8% in the patients and 11.9% in the controls, Dr. Talley said.
Although the findings do not prove a causal link between chronic constipation and colorectal cancer or benign neoplasms, they do suggest a strong association, he said in a press statement.
"The postulated causal link is that longer transit times increase the duration of contact between the colonic mucosa and concentrated carcinogens such as bile acids in the lumen," he said.
This association deserves further investigation to more thoroughly explore and to better understand possible causal elements, he added.
This is particularly important because prospective cohort studies have failed to identify a similar association to that seen in this retrospective review, suggesting that those findings are affected by recall bias, he said.
While further study is needed, practitioners should be aware of the potential relationship between chronic constipation and development of colorectal cancer and benign neoplasms, and should monitor and treat patients accordingly, he concluded.
Dr. Talley received research support from Takeda Pharmaceuticals International, which supported the study. Coauthors were employed by Takeda or by Analysis Group Inc., which has received consulting fees from Takeda.
Chronic constipation may predispose affected patients to developing colorectal cancer and benign neoplasms, according to an analysis of data from a large retrospective U.S. claims database.
The risk of developing colorectal cancer was 1.78 times higher among 28,854 adults with chronic constipation than among 86,562 controls without chronic constipation, and the risk of developing benign neoplasms was 2.7 times higher in those with chronic constipation, Dr. Nicholas Talley reported in a poster at the annual meeting of the American College of Gastroenterology.
The risk of colorectal cancer and benign neoplasms among those with chronic constipation remained "consistently high" after researchers controlled for potential confounding factors, including age, gender, family history of malignancies, and other nongastrointestinal morbidities, said Dr. Talley of the University of Newcastle, Callaghan, New South Wales, Australia.
Patients included adults aged older than 18 years who received at least two diagnoses of chronic constipation 60-365 days apart between January 1999 and September 2011. Those with irritable bowel syndrome or diarrhea were excluded, as were those who did not remain enrolled in their health plans for at least 12 months from the date of their first eligible diagnosis of constipation.
The investigators matched control subjects, who had never been diagnosed with constipation and never had a prescription filled for a laxative during the observation period, with case patients in a 1:3 ratio based on year of birth, sex, and region of residence.
Patients and controls had a mean age of 61.9 years, and one-third were men. The mean observation period was nearly 4 years.
The prevalence of colorectal cancer in this study was 2.7% in the patients and 1.7% in the controls; the prevalence of benign neoplasms was 24.8% in the patients and 11.9% in the controls, Dr. Talley said.
Although the findings do not prove a causal link between chronic constipation and colorectal cancer or benign neoplasms, they do suggest a strong association, he said in a press statement.
"The postulated causal link is that longer transit times increase the duration of contact between the colonic mucosa and concentrated carcinogens such as bile acids in the lumen," he said.
This association deserves further investigation to more thoroughly explore and to better understand possible causal elements, he added.
This is particularly important because prospective cohort studies have failed to identify a similar association to that seen in this retrospective review, suggesting that those findings are affected by recall bias, he said.
While further study is needed, practitioners should be aware of the potential relationship between chronic constipation and development of colorectal cancer and benign neoplasms, and should monitor and treat patients accordingly, he concluded.
Dr. Talley received research support from Takeda Pharmaceuticals International, which supported the study. Coauthors were employed by Takeda or by Analysis Group Inc., which has received consulting fees from Takeda.
Chronic constipation may predispose affected patients to developing colorectal cancer and benign neoplasms, according to an analysis of data from a large retrospective U.S. claims database.
The risk of developing colorectal cancer was 1.78 times higher among 28,854 adults with chronic constipation than among 86,562 controls without chronic constipation, and the risk of developing benign neoplasms was 2.7 times higher in those with chronic constipation, Dr. Nicholas Talley reported in a poster at the annual meeting of the American College of Gastroenterology.
The risk of colorectal cancer and benign neoplasms among those with chronic constipation remained "consistently high" after researchers controlled for potential confounding factors, including age, gender, family history of malignancies, and other nongastrointestinal morbidities, said Dr. Talley of the University of Newcastle, Callaghan, New South Wales, Australia.
Patients included adults aged older than 18 years who received at least two diagnoses of chronic constipation 60-365 days apart between January 1999 and September 2011. Those with irritable bowel syndrome or diarrhea were excluded, as were those who did not remain enrolled in their health plans for at least 12 months from the date of their first eligible diagnosis of constipation.
The investigators matched control subjects, who had never been diagnosed with constipation and never had a prescription filled for a laxative during the observation period, with case patients in a 1:3 ratio based on year of birth, sex, and region of residence.
Patients and controls had a mean age of 61.9 years, and one-third were men. The mean observation period was nearly 4 years.
The prevalence of colorectal cancer in this study was 2.7% in the patients and 1.7% in the controls; the prevalence of benign neoplasms was 24.8% in the patients and 11.9% in the controls, Dr. Talley said.
Although the findings do not prove a causal link between chronic constipation and colorectal cancer or benign neoplasms, they do suggest a strong association, he said in a press statement.
"The postulated causal link is that longer transit times increase the duration of contact between the colonic mucosa and concentrated carcinogens such as bile acids in the lumen," he said.
This association deserves further investigation to more thoroughly explore and to better understand possible causal elements, he added.
This is particularly important because prospective cohort studies have failed to identify a similar association to that seen in this retrospective review, suggesting that those findings are affected by recall bias, he said.
While further study is needed, practitioners should be aware of the potential relationship between chronic constipation and development of colorectal cancer and benign neoplasms, and should monitor and treat patients accordingly, he concluded.
Dr. Talley received research support from Takeda Pharmaceuticals International, which supported the study. Coauthors were employed by Takeda or by Analysis Group Inc., which has received consulting fees from Takeda.
FROM THE ANNUAL MEETING OF THE AMERICAN COLLEGE OF GASTROENTEROLOGY
Major Finding: The risk of developing colorectal cancer was 1.78 times higher in 28,854 adults with chronic constipation than in 86,562 controls without chronic constipation, and the risk of developing benign neoplasms was 2.7 times higher in those with chronic constipation.
Data Source: A large retrospective U.S. claims database.
Disclosures: Dr. Talley received research support from Takeda Pharmaceuticals International, which supported the study. Coauthors were employed by Takeda or by Analysis Group Inc., which has received consulting fees from Takeda.
Teduglutide Trims Parenteral Support in Short Bowel Syndrome
Teduglutide significantly reduced the need for parenteral support in patients with short bowel syndrome and intestinal failure, based on data from 85 adults in a randomized, controlled multicenter trial. The findings were published in the December issue of Gastroenterology.
Patients with short bowel syndrome and intestinal failure (SBS-IF) have inadequate intestinal absorption and require parenteral support (PS) to maintain fluids, electrolytes, trace elements, vitamins, and nutrient balances, said Dr. Palle Bekker Jeppesen of Rigshospitalet in Copenhagen and colleagues.
Source: American Gastroenterological Association
Data from previous open-label studies suggest an association between teduglutide and clinically meaningful reductions in wet weight and energy, which may reduce the need for PS in these patients, the investigators noted.
The researchers randomized 86 adults with SBS-IF to either 0.05 mg/kg per day of teduglutide or a placebo. One patient was randomized in error; complete data were available for 42 teduglutide patients and 43 placebo patients.
Significantly more patients in the teduglutide group responded to treatment, compared with the placebo group (63% vs. 30%). This response was defined as sustaining a 20%-100% reduction from baseline in weekly PS volume during weeks 20-24. "Small bowel length did not appear to be a predictor of response," the researchers noted.
The high placebo response may be explained by examining the fluid composite effect, a measure of the combined effects of teduglutide on PS volume reduction as well as the ability to reduce oral fluid intake and increase urine output volume, the researchers noted.
"In the current study, where protocol modifications encouraged earlier and more aggressive PS reductions, significantly larger PS reductions were also achieved in patients receiving placebo, but subsequently these patients had to increase their oral fluid intake significantly to maintain urine production and hydration constant," they said.
After 24 weeks, overall PS volume was reduced by 32% from baseline in teduglutide patients, compared with 21% in placebo patients. Although no patients in either group were completely weaned from parenteral support at 24 weeks, the difference in PS volume reduction was significantly greater in the teduglutide group.
The average weekly PS volume in teduglutide patients decreased significantly from 12.5 L/wk at baseline to 8.1 L/wk at week 24. The placebo patients also had a significant decrease in average weekly PS volume, from 13.4 L/wk at baseline to 11.1 L/wk at week 24.
Treatment-ending adverse events were similar between the two groups; 5% of teduglutide patients and 7% of placebo patients discontinued treatment because of such events during the study period. The most frequently reported treatment-emergent adverse events included abdominal pain, abdominal distension, nausea, and gastrointestinal stoma complications.
Although the study did not specifically assess quality of life measures, significantly more teduglutide patients had at least 1 day off PS, compared with placebo patients, which could help to "liberate considerable time for unhindered daytime activities or undisturbed sleep," the researchers said.
The study did not address the possible benefit of teduglutide therapy earlier in the course of SBS, or the duration of effect after patients discontinued teduglutide, the researchers added.
However, the findings indicate that teduglutide was safe and well tolerated, and "could positively add to the limited treatment armamentarium" for patients with SBS-IF.
Dr. Jeppesen and several coauthors have served on the advisory board of and as consultants to NPS Pharmaceuticals, the company that funded the study. One author is an employee of NPS Pharmaceuticals.
Patients with short bowel syndrome whose absorption is insufficient to maintain nutritional or fluid autonomy have intestinal failure. These patients, particularly those with proximal jejunostomies, who may actually secrete more fluid than they ingest, are among the most complex and challenging to manage of patients with any gastrointestinal disease. Patients with short bowel syndrome and intestinal failure are dependent on parenteral nutrition and/or fluid support (PS) to maintain life. This therapy has substantial implications for employment, activities, sleep, and finances. Numerous, often life-threatening, complications develop.

|
|
A myriad of growth factors may be involved in the process of postresection intestinal adaptation, including glucagonlike peptide-2 (GLP-2), wherein intestinal epithelial growth is promoted. Teduglutide is a long-acting analog of native GLP-2 and is somewhat more resistant to enzymatic degradation in the enterocyte than is the native enzyme. Dr. Jeppesen and colleagues reported a sustained 20%-100% decrease in PS volume requirements during weeks 20-24 of treatment in 63% of patients who received teduglutide, compared with 30% of placebo-treated patients. The mean drop in weekly PS volume from baseline to week 24 totaled 4.4 L in patients who received teduglutide, which equates to a decrease of 1-2 nights of infusion weekly, a very profound improvement for individual patients. The PS weaning protocol used was similar to that used in most centers experienced in the care of these patients.
As would be expected in the SBS-IF patient population, there were many adverse events, although these were equally distributed across teduglutide and placebo groups. Stomal changes, primarily related to enlargement, were evident in a significant minority of patients in the teduglutide group, as would be expected given the hyperplastic effect of the medication on intestinal epithelial tissue.Concern has been raised about GLP-2’s potential to stimulate the development of colonic adenomas in rodent models. Although the risk for malignancy is hypothetical in humans, colonoscopy should be considered at baseline for those patients with residual colons and perhaps as frequently as annually while the patients are on therapy until more long-term safety data are available.
Is teduglutide a "game changer"? The only patients who will be able to discontinue PS completely will be those who are on the borderline between nutritional autonomy and PS dependence. It is important to realize that teduglutide should be used to augment, not replace conventional management. What happens when teduglutide is stopped? Preliminary evidence suggests the effects on adaptation may be persistent, although earlier study noted that histologic changes trended toward baseline within 4 weeks of discontinuation. Perhaps longer treatment or maintenance will be required. The real future is an artificially grown and harvested intestine; even intestinal transplantation represents a bridge at best.
Dr. Alan L. Buchman is a former professor of medicine and surgery at the Feinberg School of Medicine at Northwestern University, Chicago. Within the past 12 months he has consulted for Takeda Pharmaceuticals and NPS Pharmaceuticals.
Patients with short bowel syndrome whose absorption is insufficient to maintain nutritional or fluid autonomy have intestinal failure. These patients, particularly those with proximal jejunostomies, who may actually secrete more fluid than they ingest, are among the most complex and challenging to manage of patients with any gastrointestinal disease. Patients with short bowel syndrome and intestinal failure are dependent on parenteral nutrition and/or fluid support (PS) to maintain life. This therapy has substantial implications for employment, activities, sleep, and finances. Numerous, often life-threatening, complications develop.

|
|
A myriad of growth factors may be involved in the process of postresection intestinal adaptation, including glucagonlike peptide-2 (GLP-2), wherein intestinal epithelial growth is promoted. Teduglutide is a long-acting analog of native GLP-2 and is somewhat more resistant to enzymatic degradation in the enterocyte than is the native enzyme. Dr. Jeppesen and colleagues reported a sustained 20%-100% decrease in PS volume requirements during weeks 20-24 of treatment in 63% of patients who received teduglutide, compared with 30% of placebo-treated patients. The mean drop in weekly PS volume from baseline to week 24 totaled 4.4 L in patients who received teduglutide, which equates to a decrease of 1-2 nights of infusion weekly, a very profound improvement for individual patients. The PS weaning protocol used was similar to that used in most centers experienced in the care of these patients.
As would be expected in the SBS-IF patient population, there were many adverse events, although these were equally distributed across teduglutide and placebo groups. Stomal changes, primarily related to enlargement, were evident in a significant minority of patients in the teduglutide group, as would be expected given the hyperplastic effect of the medication on intestinal epithelial tissue.Concern has been raised about GLP-2’s potential to stimulate the development of colonic adenomas in rodent models. Although the risk for malignancy is hypothetical in humans, colonoscopy should be considered at baseline for those patients with residual colons and perhaps as frequently as annually while the patients are on therapy until more long-term safety data are available.
Is teduglutide a "game changer"? The only patients who will be able to discontinue PS completely will be those who are on the borderline between nutritional autonomy and PS dependence. It is important to realize that teduglutide should be used to augment, not replace conventional management. What happens when teduglutide is stopped? Preliminary evidence suggests the effects on adaptation may be persistent, although earlier study noted that histologic changes trended toward baseline within 4 weeks of discontinuation. Perhaps longer treatment or maintenance will be required. The real future is an artificially grown and harvested intestine; even intestinal transplantation represents a bridge at best.
Dr. Alan L. Buchman is a former professor of medicine and surgery at the Feinberg School of Medicine at Northwestern University, Chicago. Within the past 12 months he has consulted for Takeda Pharmaceuticals and NPS Pharmaceuticals.
Patients with short bowel syndrome whose absorption is insufficient to maintain nutritional or fluid autonomy have intestinal failure. These patients, particularly those with proximal jejunostomies, who may actually secrete more fluid than they ingest, are among the most complex and challenging to manage of patients with any gastrointestinal disease. Patients with short bowel syndrome and intestinal failure are dependent on parenteral nutrition and/or fluid support (PS) to maintain life. This therapy has substantial implications for employment, activities, sleep, and finances. Numerous, often life-threatening, complications develop.

|
|
A myriad of growth factors may be involved in the process of postresection intestinal adaptation, including glucagonlike peptide-2 (GLP-2), wherein intestinal epithelial growth is promoted. Teduglutide is a long-acting analog of native GLP-2 and is somewhat more resistant to enzymatic degradation in the enterocyte than is the native enzyme. Dr. Jeppesen and colleagues reported a sustained 20%-100% decrease in PS volume requirements during weeks 20-24 of treatment in 63% of patients who received teduglutide, compared with 30% of placebo-treated patients. The mean drop in weekly PS volume from baseline to week 24 totaled 4.4 L in patients who received teduglutide, which equates to a decrease of 1-2 nights of infusion weekly, a very profound improvement for individual patients. The PS weaning protocol used was similar to that used in most centers experienced in the care of these patients.
As would be expected in the SBS-IF patient population, there were many adverse events, although these were equally distributed across teduglutide and placebo groups. Stomal changes, primarily related to enlargement, were evident in a significant minority of patients in the teduglutide group, as would be expected given the hyperplastic effect of the medication on intestinal epithelial tissue.Concern has been raised about GLP-2’s potential to stimulate the development of colonic adenomas in rodent models. Although the risk for malignancy is hypothetical in humans, colonoscopy should be considered at baseline for those patients with residual colons and perhaps as frequently as annually while the patients are on therapy until more long-term safety data are available.
Is teduglutide a "game changer"? The only patients who will be able to discontinue PS completely will be those who are on the borderline between nutritional autonomy and PS dependence. It is important to realize that teduglutide should be used to augment, not replace conventional management. What happens when teduglutide is stopped? Preliminary evidence suggests the effects on adaptation may be persistent, although earlier study noted that histologic changes trended toward baseline within 4 weeks of discontinuation. Perhaps longer treatment or maintenance will be required. The real future is an artificially grown and harvested intestine; even intestinal transplantation represents a bridge at best.
Dr. Alan L. Buchman is a former professor of medicine and surgery at the Feinberg School of Medicine at Northwestern University, Chicago. Within the past 12 months he has consulted for Takeda Pharmaceuticals and NPS Pharmaceuticals.
Teduglutide significantly reduced the need for parenteral support in patients with short bowel syndrome and intestinal failure, based on data from 85 adults in a randomized, controlled multicenter trial. The findings were published in the December issue of Gastroenterology.
Patients with short bowel syndrome and intestinal failure (SBS-IF) have inadequate intestinal absorption and require parenteral support (PS) to maintain fluids, electrolytes, trace elements, vitamins, and nutrient balances, said Dr. Palle Bekker Jeppesen of Rigshospitalet in Copenhagen and colleagues.
Source: American Gastroenterological Association
Data from previous open-label studies suggest an association between teduglutide and clinically meaningful reductions in wet weight and energy, which may reduce the need for PS in these patients, the investigators noted.
The researchers randomized 86 adults with SBS-IF to either 0.05 mg/kg per day of teduglutide or a placebo. One patient was randomized in error; complete data were available for 42 teduglutide patients and 43 placebo patients.
Significantly more patients in the teduglutide group responded to treatment, compared with the placebo group (63% vs. 30%). This response was defined as sustaining a 20%-100% reduction from baseline in weekly PS volume during weeks 20-24. "Small bowel length did not appear to be a predictor of response," the researchers noted.
The high placebo response may be explained by examining the fluid composite effect, a measure of the combined effects of teduglutide on PS volume reduction as well as the ability to reduce oral fluid intake and increase urine output volume, the researchers noted.
"In the current study, where protocol modifications encouraged earlier and more aggressive PS reductions, significantly larger PS reductions were also achieved in patients receiving placebo, but subsequently these patients had to increase their oral fluid intake significantly to maintain urine production and hydration constant," they said.
After 24 weeks, overall PS volume was reduced by 32% from baseline in teduglutide patients, compared with 21% in placebo patients. Although no patients in either group were completely weaned from parenteral support at 24 weeks, the difference in PS volume reduction was significantly greater in the teduglutide group.
The average weekly PS volume in teduglutide patients decreased significantly from 12.5 L/wk at baseline to 8.1 L/wk at week 24. The placebo patients also had a significant decrease in average weekly PS volume, from 13.4 L/wk at baseline to 11.1 L/wk at week 24.
Treatment-ending adverse events were similar between the two groups; 5% of teduglutide patients and 7% of placebo patients discontinued treatment because of such events during the study period. The most frequently reported treatment-emergent adverse events included abdominal pain, abdominal distension, nausea, and gastrointestinal stoma complications.
Although the study did not specifically assess quality of life measures, significantly more teduglutide patients had at least 1 day off PS, compared with placebo patients, which could help to "liberate considerable time for unhindered daytime activities or undisturbed sleep," the researchers said.
The study did not address the possible benefit of teduglutide therapy earlier in the course of SBS, or the duration of effect after patients discontinued teduglutide, the researchers added.
However, the findings indicate that teduglutide was safe and well tolerated, and "could positively add to the limited treatment armamentarium" for patients with SBS-IF.
Dr. Jeppesen and several coauthors have served on the advisory board of and as consultants to NPS Pharmaceuticals, the company that funded the study. One author is an employee of NPS Pharmaceuticals.
Teduglutide significantly reduced the need for parenteral support in patients with short bowel syndrome and intestinal failure, based on data from 85 adults in a randomized, controlled multicenter trial. The findings were published in the December issue of Gastroenterology.
Patients with short bowel syndrome and intestinal failure (SBS-IF) have inadequate intestinal absorption and require parenteral support (PS) to maintain fluids, electrolytes, trace elements, vitamins, and nutrient balances, said Dr. Palle Bekker Jeppesen of Rigshospitalet in Copenhagen and colleagues.
Source: American Gastroenterological Association
Data from previous open-label studies suggest an association between teduglutide and clinically meaningful reductions in wet weight and energy, which may reduce the need for PS in these patients, the investigators noted.
The researchers randomized 86 adults with SBS-IF to either 0.05 mg/kg per day of teduglutide or a placebo. One patient was randomized in error; complete data were available for 42 teduglutide patients and 43 placebo patients.
Significantly more patients in the teduglutide group responded to treatment, compared with the placebo group (63% vs. 30%). This response was defined as sustaining a 20%-100% reduction from baseline in weekly PS volume during weeks 20-24. "Small bowel length did not appear to be a predictor of response," the researchers noted.
The high placebo response may be explained by examining the fluid composite effect, a measure of the combined effects of teduglutide on PS volume reduction as well as the ability to reduce oral fluid intake and increase urine output volume, the researchers noted.
"In the current study, where protocol modifications encouraged earlier and more aggressive PS reductions, significantly larger PS reductions were also achieved in patients receiving placebo, but subsequently these patients had to increase their oral fluid intake significantly to maintain urine production and hydration constant," they said.
After 24 weeks, overall PS volume was reduced by 32% from baseline in teduglutide patients, compared with 21% in placebo patients. Although no patients in either group were completely weaned from parenteral support at 24 weeks, the difference in PS volume reduction was significantly greater in the teduglutide group.
The average weekly PS volume in teduglutide patients decreased significantly from 12.5 L/wk at baseline to 8.1 L/wk at week 24. The placebo patients also had a significant decrease in average weekly PS volume, from 13.4 L/wk at baseline to 11.1 L/wk at week 24.
Treatment-ending adverse events were similar between the two groups; 5% of teduglutide patients and 7% of placebo patients discontinued treatment because of such events during the study period. The most frequently reported treatment-emergent adverse events included abdominal pain, abdominal distension, nausea, and gastrointestinal stoma complications.
Although the study did not specifically assess quality of life measures, significantly more teduglutide patients had at least 1 day off PS, compared with placebo patients, which could help to "liberate considerable time for unhindered daytime activities or undisturbed sleep," the researchers said.
The study did not address the possible benefit of teduglutide therapy earlier in the course of SBS, or the duration of effect after patients discontinued teduglutide, the researchers added.
However, the findings indicate that teduglutide was safe and well tolerated, and "could positively add to the limited treatment armamentarium" for patients with SBS-IF.
Dr. Jeppesen and several coauthors have served on the advisory board of and as consultants to NPS Pharmaceuticals, the company that funded the study. One author is an employee of NPS Pharmaceuticals.
FROM GASTROENTEROLOGY
Major Finding: Significantly more patients who received 0.05 mg/kg per day of teduglutide had a sustained response to treatment during weeks 20-24, compared with the placebo group (63% vs. 30%).
Data Source: The data come from a randomized, controlled multicenter trial of 85 adults with short bowel syndrome and intestinal failure.
Disclosures: Dr. Jeppesen and several coauthors have served on the advisory board of and as consultants to NPS Pharmaceuticals, the company that funded the study. One author is an employee of NPS Pharmaceuticals.
FDA Reports Dabigatran Bleeds Don't Exceed Warfarin Bleeds
The Food and Drug Administration is satisfied that dabigatran’s bleeding risk is no greater than that of warfarin and will not change the drug’s label.
The rates of gastrointestinal and intracranial bleeding among patients who have been prescribed the anticoagulant dabigatran "do not appear to be higher" than the rates among patients who have been prescribed warfarin, according to an analysis of insurance claims and administrative data conducted by the agency.
The results of this analysis, conducted in response to postmarketing reports of bleeding among people treated with dabigatran, are "consistent with observations" in the RE-LY trial, the study of 18,000 patients that was the basis of the approval of the anticoagulant for reducing the risk of stroke and blood clots in patients with nonvalvular atrial fibrillation (AF), the FDA said in the MedWatch safety alert, released on Nov. 2. In the RE-LY study, the rates of serious bleeding was similar among those treated with dabigatran and those with warfarin (N. Engl. J. Med. 2009;361:1139-51).
The agency is evaluating different sources of data in its review of this safety issue, which is ongoing. Dabigatran, an orally administered direct thrombin inhibitor, was approved in October 2010 for reducing the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation and is marketed as Pradaxa by Boehringer Ingelheim.
The FDA’s analysis found that the rates of bleeding were actually higher among those on warfarin, although the statement does not point this out. The FDA analyzed data from a database of nearly 100 million patients and determined that the combined incidence of intracranial and gastrointestinal hemorrhages per 100,000 days at risk was 1.8-2.6 times higher for new users of warfarin than for new users of dabigatran. When they analyzed the two events separately, they found that the incidence rate of gastrointestinal hemorrhage events per 100,000 days at risk was 1.6-2.2 times higher for new users of warfarin than for new users of dabigatran. The incidence rate of intracranial hemorrhage events per 100,000 days at risk was 2.1-3.0 times higher for new users of warfarin than for those on dabigatran.
These estimates do not account for age, medical conditions, or other differences between the patients on warfarin and dabigatran that could affect bleeding outcomes, according to the FDA. In addition, although a "large" number of reports of bleeding in treated patients were submitted to the FDA’s Adverse Events Reporting System (FAERS) after dabigatran was approved, the agency believes that a "simple comparison" between the number of postmarketing bleeding events associated with dabigatran and warfarin is "misleading" because it is likely that bleeding events associated with warfarin are under reported, since the drug has been available for so long and bleeding is a well-recognized consequence of warfarin treatment.
At this time, the FDA is not changing any recommendations on the dabigatran label and is continuing to monitor postmarketing reports of bleeding in patients on dabigatran "for evidence of inappropriate dosing, use of interacting drugs, and other clinical factors that might lead to a bleeding event," according to the statement. The recommendations in the statement include advice to clinicians that they evaluate a patient’s renal function before prescribing dabigatran, which is eliminated by the kidneys, and the dosing regimens for patients with severe renal impairment and those with a creatinine clearance above 30 mL/min.
Click here for the Medwatch safety alert. Adverse events associated with dabigatran should be reported here or to the FDA at 800-332-0178.
The Food and Drug Administration is satisfied that dabigatran’s bleeding risk is no greater than that of warfarin and will not change the drug’s label.
The rates of gastrointestinal and intracranial bleeding among patients who have been prescribed the anticoagulant dabigatran "do not appear to be higher" than the rates among patients who have been prescribed warfarin, according to an analysis of insurance claims and administrative data conducted by the agency.
The results of this analysis, conducted in response to postmarketing reports of bleeding among people treated with dabigatran, are "consistent with observations" in the RE-LY trial, the study of 18,000 patients that was the basis of the approval of the anticoagulant for reducing the risk of stroke and blood clots in patients with nonvalvular atrial fibrillation (AF), the FDA said in the MedWatch safety alert, released on Nov. 2. In the RE-LY study, the rates of serious bleeding was similar among those treated with dabigatran and those with warfarin (N. Engl. J. Med. 2009;361:1139-51).
The agency is evaluating different sources of data in its review of this safety issue, which is ongoing. Dabigatran, an orally administered direct thrombin inhibitor, was approved in October 2010 for reducing the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation and is marketed as Pradaxa by Boehringer Ingelheim.
The FDA’s analysis found that the rates of bleeding were actually higher among those on warfarin, although the statement does not point this out. The FDA analyzed data from a database of nearly 100 million patients and determined that the combined incidence of intracranial and gastrointestinal hemorrhages per 100,000 days at risk was 1.8-2.6 times higher for new users of warfarin than for new users of dabigatran. When they analyzed the two events separately, they found that the incidence rate of gastrointestinal hemorrhage events per 100,000 days at risk was 1.6-2.2 times higher for new users of warfarin than for new users of dabigatran. The incidence rate of intracranial hemorrhage events per 100,000 days at risk was 2.1-3.0 times higher for new users of warfarin than for those on dabigatran.
These estimates do not account for age, medical conditions, or other differences between the patients on warfarin and dabigatran that could affect bleeding outcomes, according to the FDA. In addition, although a "large" number of reports of bleeding in treated patients were submitted to the FDA’s Adverse Events Reporting System (FAERS) after dabigatran was approved, the agency believes that a "simple comparison" between the number of postmarketing bleeding events associated with dabigatran and warfarin is "misleading" because it is likely that bleeding events associated with warfarin are under reported, since the drug has been available for so long and bleeding is a well-recognized consequence of warfarin treatment.
At this time, the FDA is not changing any recommendations on the dabigatran label and is continuing to monitor postmarketing reports of bleeding in patients on dabigatran "for evidence of inappropriate dosing, use of interacting drugs, and other clinical factors that might lead to a bleeding event," according to the statement. The recommendations in the statement include advice to clinicians that they evaluate a patient’s renal function before prescribing dabigatran, which is eliminated by the kidneys, and the dosing regimens for patients with severe renal impairment and those with a creatinine clearance above 30 mL/min.
Click here for the Medwatch safety alert. Adverse events associated with dabigatran should be reported here or to the FDA at 800-332-0178.
The Food and Drug Administration is satisfied that dabigatran’s bleeding risk is no greater than that of warfarin and will not change the drug’s label.
The rates of gastrointestinal and intracranial bleeding among patients who have been prescribed the anticoagulant dabigatran "do not appear to be higher" than the rates among patients who have been prescribed warfarin, according to an analysis of insurance claims and administrative data conducted by the agency.
The results of this analysis, conducted in response to postmarketing reports of bleeding among people treated with dabigatran, are "consistent with observations" in the RE-LY trial, the study of 18,000 patients that was the basis of the approval of the anticoagulant for reducing the risk of stroke and blood clots in patients with nonvalvular atrial fibrillation (AF), the FDA said in the MedWatch safety alert, released on Nov. 2. In the RE-LY study, the rates of serious bleeding was similar among those treated with dabigatran and those with warfarin (N. Engl. J. Med. 2009;361:1139-51).
The agency is evaluating different sources of data in its review of this safety issue, which is ongoing. Dabigatran, an orally administered direct thrombin inhibitor, was approved in October 2010 for reducing the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation and is marketed as Pradaxa by Boehringer Ingelheim.
The FDA’s analysis found that the rates of bleeding were actually higher among those on warfarin, although the statement does not point this out. The FDA analyzed data from a database of nearly 100 million patients and determined that the combined incidence of intracranial and gastrointestinal hemorrhages per 100,000 days at risk was 1.8-2.6 times higher for new users of warfarin than for new users of dabigatran. When they analyzed the two events separately, they found that the incidence rate of gastrointestinal hemorrhage events per 100,000 days at risk was 1.6-2.2 times higher for new users of warfarin than for new users of dabigatran. The incidence rate of intracranial hemorrhage events per 100,000 days at risk was 2.1-3.0 times higher for new users of warfarin than for those on dabigatran.
These estimates do not account for age, medical conditions, or other differences between the patients on warfarin and dabigatran that could affect bleeding outcomes, according to the FDA. In addition, although a "large" number of reports of bleeding in treated patients were submitted to the FDA’s Adverse Events Reporting System (FAERS) after dabigatran was approved, the agency believes that a "simple comparison" between the number of postmarketing bleeding events associated with dabigatran and warfarin is "misleading" because it is likely that bleeding events associated with warfarin are under reported, since the drug has been available for so long and bleeding is a well-recognized consequence of warfarin treatment.
At this time, the FDA is not changing any recommendations on the dabigatran label and is continuing to monitor postmarketing reports of bleeding in patients on dabigatran "for evidence of inappropriate dosing, use of interacting drugs, and other clinical factors that might lead to a bleeding event," according to the statement. The recommendations in the statement include advice to clinicians that they evaluate a patient’s renal function before prescribing dabigatran, which is eliminated by the kidneys, and the dosing regimens for patients with severe renal impairment and those with a creatinine clearance above 30 mL/min.
Click here for the Medwatch safety alert. Adverse events associated with dabigatran should be reported here or to the FDA at 800-332-0178.
Pneumonia Prevalence Highest of Health Care-Associated Infections
SAN DIEGO – The overall prevalence of health care–associated infections among inpatients in the United States stands at 4%, with the most common types of infections being a combination of pneumonia and lower respiratory infections.
Those are key preliminary findings from the Centers for Disease Control and Prevention’s first large-scale health care–associated infection (HAI) prevalence survey in more than 30 years, Dr. Shelley S. Magill reported during IDWeek 2012, the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The data "can help us better understand the factors that influence HAI prevalence," said Dr. Magill of the division of health care quality promotion at the Centers for Disease Control and Prevention, Atlanta. "We can also clarify the burden of different HAI types and pathogens across the hospital, which can suggest areas to target for prevention."
The phase 3 survey was conducted in 2011 in 183 hospitals in 10 states: California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee. Dr. Magill reported results from 11,282 patients who were surveyed in the 183 hospitals. Of these, 452 patients had HAIs, for a prevalence of 4%.
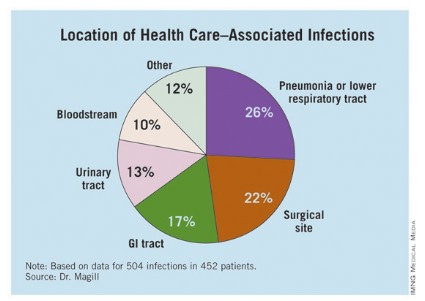
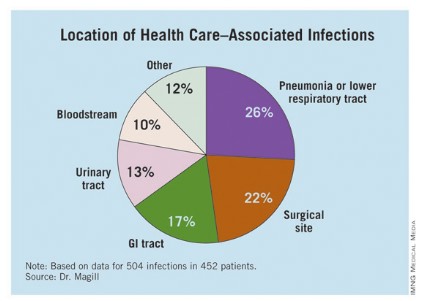
The researchers identified 504 HAIs in the 452 patients. Of these, the highest proportion (26%) were pneumonia or lower respiratory infections. "Of the pneumonia events, 39% were ventilator-associated infections," Dr. Magill said.
Surgical-site infections were the second most common infection type, representing 22% of all HAIs, followed by GI infections (17%), urinary tract infections (13%), and bloodstream infections (10%). "About two-third of UTIs were catheter associated and about 84% of the bloodstream infections were central-line associated," Dr. Magill noted.
Other infections made up the remaining 12% of HAIs.
Dr. Magill also reported that 56% of HAIs were attributed to non-ICU locations in the hospital while 53% were not directly associated with a device or with a procedure.
After multivariable regression analysis, patients with the following factors were at higher risk of having HAIs:
• Older age (risk ratio, 4.60 for patients older than age 77 compared with patients younger than 3 months old).
• Inpatient in a large hospital – defined as having 400 or more beds (RR, 1.24).
• Inpatient in a critical care unit (RR ,1.96 compared with all other units),
• Inpatient for more than 2 weeks at the time of the survey (RR, 26.09 compared with patients in the hospital for 3 days or fewer).
Pathogens were reported for 372 of the 504 HAIs. Clostridium difficile was the most common pathogen, accounting for 12% of all HAIs that were identified. Staphylococcus aureus was the second most common pathogen (11%; about half of these cases were methicillin-resistant S. aureus), followed by Klebsiella pneumoniae and Klebsiella oxytoca (10%).
Dr. Magill acknowledged certain limitations of the survey, including the fact that a small number of patients surveyed in each hospital "make results of limited use to individual facilities," she said. "We also had a relatively small number of hospitals in 10 states participate. This survey method provides a single snapshot of HAIs based on a retrospective review of medical record data, which is sometimes not complete. Finally, we were not able to collect a lot of detailed patient information, such as underlying illnesses and severity of illness."
She and her associates are currently planning a phase 4 survey, anticipated to occur in 2014.
Dr. Magill said she had no relevant financial conflicts to disclose.
SAN DIEGO – The overall prevalence of health care–associated infections among inpatients in the United States stands at 4%, with the most common types of infections being a combination of pneumonia and lower respiratory infections.
Those are key preliminary findings from the Centers for Disease Control and Prevention’s first large-scale health care–associated infection (HAI) prevalence survey in more than 30 years, Dr. Shelley S. Magill reported during IDWeek 2012, the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The data "can help us better understand the factors that influence HAI prevalence," said Dr. Magill of the division of health care quality promotion at the Centers for Disease Control and Prevention, Atlanta. "We can also clarify the burden of different HAI types and pathogens across the hospital, which can suggest areas to target for prevention."
The phase 3 survey was conducted in 2011 in 183 hospitals in 10 states: California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee. Dr. Magill reported results from 11,282 patients who were surveyed in the 183 hospitals. Of these, 452 patients had HAIs, for a prevalence of 4%.

The researchers identified 504 HAIs in the 452 patients. Of these, the highest proportion (26%) were pneumonia or lower respiratory infections. "Of the pneumonia events, 39% were ventilator-associated infections," Dr. Magill said.
Surgical-site infections were the second most common infection type, representing 22% of all HAIs, followed by GI infections (17%), urinary tract infections (13%), and bloodstream infections (10%). "About two-third of UTIs were catheter associated and about 84% of the bloodstream infections were central-line associated," Dr. Magill noted.
Other infections made up the remaining 12% of HAIs.
Dr. Magill also reported that 56% of HAIs were attributed to non-ICU locations in the hospital while 53% were not directly associated with a device or with a procedure.
After multivariable regression analysis, patients with the following factors were at higher risk of having HAIs:
• Older age (risk ratio, 4.60 for patients older than age 77 compared with patients younger than 3 months old).
• Inpatient in a large hospital – defined as having 400 or more beds (RR, 1.24).
• Inpatient in a critical care unit (RR ,1.96 compared with all other units),
• Inpatient for more than 2 weeks at the time of the survey (RR, 26.09 compared with patients in the hospital for 3 days or fewer).
Pathogens were reported for 372 of the 504 HAIs. Clostridium difficile was the most common pathogen, accounting for 12% of all HAIs that were identified. Staphylococcus aureus was the second most common pathogen (11%; about half of these cases were methicillin-resistant S. aureus), followed by Klebsiella pneumoniae and Klebsiella oxytoca (10%).
Dr. Magill acknowledged certain limitations of the survey, including the fact that a small number of patients surveyed in each hospital "make results of limited use to individual facilities," she said. "We also had a relatively small number of hospitals in 10 states participate. This survey method provides a single snapshot of HAIs based on a retrospective review of medical record data, which is sometimes not complete. Finally, we were not able to collect a lot of detailed patient information, such as underlying illnesses and severity of illness."
She and her associates are currently planning a phase 4 survey, anticipated to occur in 2014.
Dr. Magill said she had no relevant financial conflicts to disclose.
SAN DIEGO – The overall prevalence of health care–associated infections among inpatients in the United States stands at 4%, with the most common types of infections being a combination of pneumonia and lower respiratory infections.
Those are key preliminary findings from the Centers for Disease Control and Prevention’s first large-scale health care–associated infection (HAI) prevalence survey in more than 30 years, Dr. Shelley S. Magill reported during IDWeek 2012, the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
The data "can help us better understand the factors that influence HAI prevalence," said Dr. Magill of the division of health care quality promotion at the Centers for Disease Control and Prevention, Atlanta. "We can also clarify the burden of different HAI types and pathogens across the hospital, which can suggest areas to target for prevention."
The phase 3 survey was conducted in 2011 in 183 hospitals in 10 states: California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New Mexico, New York, Oregon, and Tennessee. Dr. Magill reported results from 11,282 patients who were surveyed in the 183 hospitals. Of these, 452 patients had HAIs, for a prevalence of 4%.

The researchers identified 504 HAIs in the 452 patients. Of these, the highest proportion (26%) were pneumonia or lower respiratory infections. "Of the pneumonia events, 39% were ventilator-associated infections," Dr. Magill said.
Surgical-site infections were the second most common infection type, representing 22% of all HAIs, followed by GI infections (17%), urinary tract infections (13%), and bloodstream infections (10%). "About two-third of UTIs were catheter associated and about 84% of the bloodstream infections were central-line associated," Dr. Magill noted.
Other infections made up the remaining 12% of HAIs.
Dr. Magill also reported that 56% of HAIs were attributed to non-ICU locations in the hospital while 53% were not directly associated with a device or with a procedure.
After multivariable regression analysis, patients with the following factors were at higher risk of having HAIs:
• Older age (risk ratio, 4.60 for patients older than age 77 compared with patients younger than 3 months old).
• Inpatient in a large hospital – defined as having 400 or more beds (RR, 1.24).
• Inpatient in a critical care unit (RR ,1.96 compared with all other units),
• Inpatient for more than 2 weeks at the time of the survey (RR, 26.09 compared with patients in the hospital for 3 days or fewer).
Pathogens were reported for 372 of the 504 HAIs. Clostridium difficile was the most common pathogen, accounting for 12% of all HAIs that were identified. Staphylococcus aureus was the second most common pathogen (11%; about half of these cases were methicillin-resistant S. aureus), followed by Klebsiella pneumoniae and Klebsiella oxytoca (10%).
Dr. Magill acknowledged certain limitations of the survey, including the fact that a small number of patients surveyed in each hospital "make results of limited use to individual facilities," she said. "We also had a relatively small number of hospitals in 10 states participate. This survey method provides a single snapshot of HAIs based on a retrospective review of medical record data, which is sometimes not complete. Finally, we were not able to collect a lot of detailed patient information, such as underlying illnesses and severity of illness."
She and her associates are currently planning a phase 4 survey, anticipated to occur in 2014.
Dr. Magill said she had no relevant financial conflicts to disclose.
AT IDWEEK 2012
Major Finding: The overall prevalence of health care–associated infections among hospitalized patients nationwide was 4%.
Data Source: Preliminary results were obtained from a 2011 survey of 11,282 inpatients at 183 hospitals located in 10 states.
Disclosures: The study was conducted by the Centers for Disease Control and Prevention. Dr. Magill said she had no relevant financial conflicts to disclose.
UV Light Beat Bleach for C. difficile Decontamination
SAN FRANCISCO – The M.D. Anderson Cancer Center is abandoning bleach for cleaning hospital rooms exposed to Clostridium difficile in favor of a new machine that kills the organism using ultraviolet light.
The machine reduced C. difficile counts as much as, or more than, bleach cleaning in a preliminary prospective trial in 30 hospital rooms previously occupied by patients infected with C. difficile. The machine is a bit more expensive than bleach at a cost of approximately $82,000 (or $3,000-$4,000 per month to lease), but it avoids damage to materials and the toxic environment for workers caused by the use of bleach or other corrosive chemicals, Dr. Shashank S. Ghantoji said in an interview at a poster presentation at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy.
Bleach treatment reduced the average number of colony-forming units of C. difficile from 2.39 before cleaning to 0.71, a 70% reduction in the contamination level. Treatment with the Pulsed Xenon UV machine (PX-UV) reduced the average number of colony-forming units from 22.97 to 1.10, a 95% reduction.
The postcleaning contamination levels were not statistically different between the bleach and PX-UV rooms, Dr. Ghantoji and his associates found. However, PX-UV decontamination is faster than using bleach, Dr. Ghantoji said. "It takes at least 45 minutes to clean a room with bleach, and it’s not good for the patients or the health care professionals," plus admissions staff usually are clamoring for the room to be ready as soon as possible, he said. Cleaning a room using the PX-UV method takes perhaps 15 minutes.
The PX-UV machine has been available for some time, but its adoption depends on how proactive hospital infection control teams are, he added. He said he is aware of at least two medical centers beyond M.D. Anderson that are also using the machine.
In the study, 298 samples were taken before and after cleaning from high-touch surfaces – the bathroom handrail, the bed control panel, the bed rail, the top of the bedside table, and the IV pole control panel or other equipment control panel – and analyzed for C. difficile endospores. Fifteen rooms were cleaned by the conventional method using a 1:10 solution of sodium hypochlorite (bleach), and 15 underwent a visual, nonbleach cleaning of surfaces followed by 15 minutes of treatment with the PX-UV.
With the PX-UV method, housekeeping workers clean the bathroom and place the remote-operated PX-UV in the bathroom with the door shut while they finish cleaning the rest of the room. Then the machine is placed on each side of the bed for 4 minutes of operation with workers gone. Sensors stop the machine if any movement is detected.
It works by emitting ultraviolet C light, which kills C. difficile. And here’s a bonus – it also kills vancomycin-resistant enterococci and methicillin-resistant Staphylococcus aureus, Dr. Ghantoji of M.D. Anderson, Houston, said at the meeting, sponsored by the American Society for Microbiology.
"The PX-UV method may be a promising alternative to the current standard of decontamination, bleach," he said. Future studies should look at whether the PX-UV method decreases not just endospore counts but transmission of C. difficile, he added.
C. difficile causes more than 300,000 health care–associated infections each year in the United States, incurring $2,500-$3,500 in costs per infection aside from any surgical costs, he estimated. Current guidelines recommend that rooms previously occupied by patients infected with C. difficile be cleaned with a disinfectant registered with the Environmental Protection Agency as effective against the organism.
Xenex Healthcare Services, which markets the PX-UV machine, funded the study, and two of the investigators are employees of the company. Dr. Ghantoji reported having no other relevant financial disclosures.
SAN FRANCISCO – The M.D. Anderson Cancer Center is abandoning bleach for cleaning hospital rooms exposed to Clostridium difficile in favor of a new machine that kills the organism using ultraviolet light.
The machine reduced C. difficile counts as much as, or more than, bleach cleaning in a preliminary prospective trial in 30 hospital rooms previously occupied by patients infected with C. difficile. The machine is a bit more expensive than bleach at a cost of approximately $82,000 (or $3,000-$4,000 per month to lease), but it avoids damage to materials and the toxic environment for workers caused by the use of bleach or other corrosive chemicals, Dr. Shashank S. Ghantoji said in an interview at a poster presentation at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy.
Bleach treatment reduced the average number of colony-forming units of C. difficile from 2.39 before cleaning to 0.71, a 70% reduction in the contamination level. Treatment with the Pulsed Xenon UV machine (PX-UV) reduced the average number of colony-forming units from 22.97 to 1.10, a 95% reduction.
The postcleaning contamination levels were not statistically different between the bleach and PX-UV rooms, Dr. Ghantoji and his associates found. However, PX-UV decontamination is faster than using bleach, Dr. Ghantoji said. "It takes at least 45 minutes to clean a room with bleach, and it’s not good for the patients or the health care professionals," plus admissions staff usually are clamoring for the room to be ready as soon as possible, he said. Cleaning a room using the PX-UV method takes perhaps 15 minutes.
The PX-UV machine has been available for some time, but its adoption depends on how proactive hospital infection control teams are, he added. He said he is aware of at least two medical centers beyond M.D. Anderson that are also using the machine.
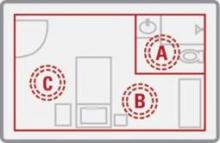
In the study, 298 samples were taken before and after cleaning from high-touch surfaces – the bathroom handrail, the bed control panel, the bed rail, the top of the bedside table, and the IV pole control panel or other equipment control panel – and analyzed for C. difficile endospores. Fifteen rooms were cleaned by the conventional method using a 1:10 solution of sodium hypochlorite (bleach), and 15 underwent a visual, nonbleach cleaning of surfaces followed by 15 minutes of treatment with the PX-UV.
With the PX-UV method, housekeeping workers clean the bathroom and place the remote-operated PX-UV in the bathroom with the door shut while they finish cleaning the rest of the room. Then the machine is placed on each side of the bed for 4 minutes of operation with workers gone. Sensors stop the machine if any movement is detected.
It works by emitting ultraviolet C light, which kills C. difficile. And here’s a bonus – it also kills vancomycin-resistant enterococci and methicillin-resistant Staphylococcus aureus, Dr. Ghantoji of M.D. Anderson, Houston, said at the meeting, sponsored by the American Society for Microbiology.
"The PX-UV method may be a promising alternative to the current standard of decontamination, bleach," he said. Future studies should look at whether the PX-UV method decreases not just endospore counts but transmission of C. difficile, he added.
C. difficile causes more than 300,000 health care–associated infections each year in the United States, incurring $2,500-$3,500 in costs per infection aside from any surgical costs, he estimated. Current guidelines recommend that rooms previously occupied by patients infected with C. difficile be cleaned with a disinfectant registered with the Environmental Protection Agency as effective against the organism.
Xenex Healthcare Services, which markets the PX-UV machine, funded the study, and two of the investigators are employees of the company. Dr. Ghantoji reported having no other relevant financial disclosures.
SAN FRANCISCO – The M.D. Anderson Cancer Center is abandoning bleach for cleaning hospital rooms exposed to Clostridium difficile in favor of a new machine that kills the organism using ultraviolet light.
The machine reduced C. difficile counts as much as, or more than, bleach cleaning in a preliminary prospective trial in 30 hospital rooms previously occupied by patients infected with C. difficile. The machine is a bit more expensive than bleach at a cost of approximately $82,000 (or $3,000-$4,000 per month to lease), but it avoids damage to materials and the toxic environment for workers caused by the use of bleach or other corrosive chemicals, Dr. Shashank S. Ghantoji said in an interview at a poster presentation at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy.
Bleach treatment reduced the average number of colony-forming units of C. difficile from 2.39 before cleaning to 0.71, a 70% reduction in the contamination level. Treatment with the Pulsed Xenon UV machine (PX-UV) reduced the average number of colony-forming units from 22.97 to 1.10, a 95% reduction.
The postcleaning contamination levels were not statistically different between the bleach and PX-UV rooms, Dr. Ghantoji and his associates found. However, PX-UV decontamination is faster than using bleach, Dr. Ghantoji said. "It takes at least 45 minutes to clean a room with bleach, and it’s not good for the patients or the health care professionals," plus admissions staff usually are clamoring for the room to be ready as soon as possible, he said. Cleaning a room using the PX-UV method takes perhaps 15 minutes.
The PX-UV machine has been available for some time, but its adoption depends on how proactive hospital infection control teams are, he added. He said he is aware of at least two medical centers beyond M.D. Anderson that are also using the machine.
In the study, 298 samples were taken before and after cleaning from high-touch surfaces – the bathroom handrail, the bed control panel, the bed rail, the top of the bedside table, and the IV pole control panel or other equipment control panel – and analyzed for C. difficile endospores. Fifteen rooms were cleaned by the conventional method using a 1:10 solution of sodium hypochlorite (bleach), and 15 underwent a visual, nonbleach cleaning of surfaces followed by 15 minutes of treatment with the PX-UV.
With the PX-UV method, housekeeping workers clean the bathroom and place the remote-operated PX-UV in the bathroom with the door shut while they finish cleaning the rest of the room. Then the machine is placed on each side of the bed for 4 minutes of operation with workers gone. Sensors stop the machine if any movement is detected.
It works by emitting ultraviolet C light, which kills C. difficile. And here’s a bonus – it also kills vancomycin-resistant enterococci and methicillin-resistant Staphylococcus aureus, Dr. Ghantoji of M.D. Anderson, Houston, said at the meeting, sponsored by the American Society for Microbiology.
"The PX-UV method may be a promising alternative to the current standard of decontamination, bleach," he said. Future studies should look at whether the PX-UV method decreases not just endospore counts but transmission of C. difficile, he added.
C. difficile causes more than 300,000 health care–associated infections each year in the United States, incurring $2,500-$3,500 in costs per infection aside from any surgical costs, he estimated. Current guidelines recommend that rooms previously occupied by patients infected with C. difficile be cleaned with a disinfectant registered with the Environmental Protection Agency as effective against the organism.
Xenex Healthcare Services, which markets the PX-UV machine, funded the study, and two of the investigators are employees of the company. Dr. Ghantoji reported having no other relevant financial disclosures.
Major Finding: Bleach killed 70% of C. difficile spores in hospital rooms compared with 95% decontamination using nonbleach cleaning plus UV light treatment. The difference between groups was not statistically significant.
Data Source: A prospective comparison was performed of the two cleaning methods in 30 rooms after discharge of patients infected with C. difficile.
Disclosures: Xenex Healthcare Services, which markets the PX-UV machine, funded the study, and two of the investigators are employees of the company. Dr. Ghantoji reported having no other relevant financial disclosures.
UV-C Light Blasts 'Bad Bugs' in Hospital Rooms
SAN DIEGO – A portable device that emits ultraviolet C light destroyed vancomycin-resistant enterococci, Acinetobacter, and Clostridium difficile from hospital rooms where patients infected with those bacteria had been housed, results from a small study demonstrated.
"There is growing evidence that the environment can be a source for acquisition of bad bugs," lead study investigator Dr. Deverick J. Anderson said in an interview prior to IDWeek 2012, where the research was presented during a poster session.
"Our study further strengthens the data that no-touch systems like UV-C light kill important bacteria and can potentially help with current cleaning strategies. While several groups have demonstrated that UV-C light work in experimental conditions we are demonstrating that it works in a real-world hospital environment."
Dr. Anderson of the department of medicine in the division of infectious diseases at Duke University, Durham, N.C., and his associates analyzed 39 rooms at two tertiary care hospitals that had just housed a patient with one of the different bad bugs: vancomycin-resistant enterococci (VRE), Acinetobacter, and C. difficile. After the patient was discharged but prior to the regular cleaning, the investigators obtained 15 or more cultures from several different locations in the hospital rooms, including bed rails, remote controls, and toilets. Then they wheeled in the TRU-D, an automated mobile disinfection system manufactured by Lumalier that is about 6 feet tall and is equipped with 8 sensors and 16 bulbs that emit UV-C light.
"Each room was irradiated between 25 and 45 minutes in order to eradicate both bacteria and bacterial spores," Dr. Anderson explained during a premeeting telephone press conference. "We then went back into the rooms and cultured the environment from the same locations."
After comparing the number of colony-forming units (CFUs) before and after irradiation "we were able to demonstrate that we could achieve well over 90% reduction in each of those three bad bugs after using the UV light," said Dr. Anderson, who also chairs the antimicrobial stewardship and evaluation team at Duke University Medical Center. "This occurred in all locations sampled, in both direct and indirect light."
Specifically, the UV-C irradiation reduced CFUs of VRE by 98%, C. difficile by 93%, and Acinetobacter by 98%.
"Based on these results we came to the conclusion that UV-C light is indeed effective in killing VRE, C. difficile, and Acinetobacter from the real-world hospital environment," Dr. Anderson said during the telephone press conference. "The idea behind achieving bacterial irradiation in shadow is actually taking advantage of the reflective properties of UV light. It literally bounces around the room and ends up hitting areas in shadow. That’s how bacterial reduction occurs."
He acknowledged certain limitations of the study, including the fact that the researchers were able to evaluate onlytwo hospital rooms with Acinetobacter "because of how infrequently this organism causes infections. Regardless, we reduced the amount of Acinetobacter in both of those rooms."
The study was sponsored by the Centers for Disease Control and Prevention. Lumalier donated the machines used in the study but had no role in the trial design or in review of the data. Dr. Anderson said that he had no relevant financial conflicts to disclose.
IDWeek 2012 is the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
SAN DIEGO – A portable device that emits ultraviolet C light destroyed vancomycin-resistant enterococci, Acinetobacter, and Clostridium difficile from hospital rooms where patients infected with those bacteria had been housed, results from a small study demonstrated.
"There is growing evidence that the environment can be a source for acquisition of bad bugs," lead study investigator Dr. Deverick J. Anderson said in an interview prior to IDWeek 2012, where the research was presented during a poster session.
"Our study further strengthens the data that no-touch systems like UV-C light kill important bacteria and can potentially help with current cleaning strategies. While several groups have demonstrated that UV-C light work in experimental conditions we are demonstrating that it works in a real-world hospital environment."
Dr. Anderson of the department of medicine in the division of infectious diseases at Duke University, Durham, N.C., and his associates analyzed 39 rooms at two tertiary care hospitals that had just housed a patient with one of the different bad bugs: vancomycin-resistant enterococci (VRE), Acinetobacter, and C. difficile. After the patient was discharged but prior to the regular cleaning, the investigators obtained 15 or more cultures from several different locations in the hospital rooms, including bed rails, remote controls, and toilets. Then they wheeled in the TRU-D, an automated mobile disinfection system manufactured by Lumalier that is about 6 feet tall and is equipped with 8 sensors and 16 bulbs that emit UV-C light.
"Each room was irradiated between 25 and 45 minutes in order to eradicate both bacteria and bacterial spores," Dr. Anderson explained during a premeeting telephone press conference. "We then went back into the rooms and cultured the environment from the same locations."
After comparing the number of colony-forming units (CFUs) before and after irradiation "we were able to demonstrate that we could achieve well over 90% reduction in each of those three bad bugs after using the UV light," said Dr. Anderson, who also chairs the antimicrobial stewardship and evaluation team at Duke University Medical Center. "This occurred in all locations sampled, in both direct and indirect light."
Specifically, the UV-C irradiation reduced CFUs of VRE by 98%, C. difficile by 93%, and Acinetobacter by 98%.
"Based on these results we came to the conclusion that UV-C light is indeed effective in killing VRE, C. difficile, and Acinetobacter from the real-world hospital environment," Dr. Anderson said during the telephone press conference. "The idea behind achieving bacterial irradiation in shadow is actually taking advantage of the reflective properties of UV light. It literally bounces around the room and ends up hitting areas in shadow. That’s how bacterial reduction occurs."
He acknowledged certain limitations of the study, including the fact that the researchers were able to evaluate onlytwo hospital rooms with Acinetobacter "because of how infrequently this organism causes infections. Regardless, we reduced the amount of Acinetobacter in both of those rooms."
The study was sponsored by the Centers for Disease Control and Prevention. Lumalier donated the machines used in the study but had no role in the trial design or in review of the data. Dr. Anderson said that he had no relevant financial conflicts to disclose.
IDWeek 2012 is the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
SAN DIEGO – A portable device that emits ultraviolet C light destroyed vancomycin-resistant enterococci, Acinetobacter, and Clostridium difficile from hospital rooms where patients infected with those bacteria had been housed, results from a small study demonstrated.
"There is growing evidence that the environment can be a source for acquisition of bad bugs," lead study investigator Dr. Deverick J. Anderson said in an interview prior to IDWeek 2012, where the research was presented during a poster session.
"Our study further strengthens the data that no-touch systems like UV-C light kill important bacteria and can potentially help with current cleaning strategies. While several groups have demonstrated that UV-C light work in experimental conditions we are demonstrating that it works in a real-world hospital environment."
Dr. Anderson of the department of medicine in the division of infectious diseases at Duke University, Durham, N.C., and his associates analyzed 39 rooms at two tertiary care hospitals that had just housed a patient with one of the different bad bugs: vancomycin-resistant enterococci (VRE), Acinetobacter, and C. difficile. After the patient was discharged but prior to the regular cleaning, the investigators obtained 15 or more cultures from several different locations in the hospital rooms, including bed rails, remote controls, and toilets. Then they wheeled in the TRU-D, an automated mobile disinfection system manufactured by Lumalier that is about 6 feet tall and is equipped with 8 sensors and 16 bulbs that emit UV-C light.
"Each room was irradiated between 25 and 45 minutes in order to eradicate both bacteria and bacterial spores," Dr. Anderson explained during a premeeting telephone press conference. "We then went back into the rooms and cultured the environment from the same locations."
After comparing the number of colony-forming units (CFUs) before and after irradiation "we were able to demonstrate that we could achieve well over 90% reduction in each of those three bad bugs after using the UV light," said Dr. Anderson, who also chairs the antimicrobial stewardship and evaluation team at Duke University Medical Center. "This occurred in all locations sampled, in both direct and indirect light."
Specifically, the UV-C irradiation reduced CFUs of VRE by 98%, C. difficile by 93%, and Acinetobacter by 98%.
"Based on these results we came to the conclusion that UV-C light is indeed effective in killing VRE, C. difficile, and Acinetobacter from the real-world hospital environment," Dr. Anderson said during the telephone press conference. "The idea behind achieving bacterial irradiation in shadow is actually taking advantage of the reflective properties of UV light. It literally bounces around the room and ends up hitting areas in shadow. That’s how bacterial reduction occurs."
He acknowledged certain limitations of the study, including the fact that the researchers were able to evaluate onlytwo hospital rooms with Acinetobacter "because of how infrequently this organism causes infections. Regardless, we reduced the amount of Acinetobacter in both of those rooms."
The study was sponsored by the Centers for Disease Control and Prevention. Lumalier donated the machines used in the study but had no role in the trial design or in review of the data. Dr. Anderson said that he had no relevant financial conflicts to disclose.
IDWeek 2012 is the combined annual meetings of the Infectious Diseases Society of America, the Society for Healthcare Epidemiology of America, the HIV Medicine Association, and the Pediatric Infectious Diseases Society.
Major Finding: UV-C irradiation of hospital rooms with a portable disinfection system reduced colony-forming units of vancomycin-resistant enterococci by 98%, C. difficile by 93%, and Acinetobacter by 98%.
Data Source: Results were taken from a study conducted in 39 hospital rooms at two tertiary medical centers.
Disclosures: The study was sponsored by the Centers for Disease Control and Prevention. Lumalier donated the machines used in the study but had no role in the trial design or in review of the data. Dr. Anderson said that he had no relevant financial conflicts to disclose.
Promising C. difficile Antibiotic in Pipeline
SAN FRANCISCO – Those desperate for new treatments for Clostridium difficile infection may want to keep an eye on the experimental oral antibiotic cadazolid, which looked promising in an early-phase trial, according to Daniela Baldoni, Pharm.D.
Cadazolid is in the oxazolidinone class of antibiotics. Its mechanism of action consists mainly of bacterial protein-synthesis inhibition.
Cadazolid produced low systemic exposure with high concentrations at the desired site – the colon – and was well tolerated in 64 healthy men who received up to 3,000 mg b.i.d. for 10 days, she reported in a poster presentation at the conference. Dr. Baldoni is employed by Actelion Pharmaceuticals, the company that is developing cadazolid.
The study randomized nonsmoking men aged 45-60 years and a body mass index of 18-32 kg/m2 to single or multiple doses of cadazolid or placebo.
In the single-dose group, 30 fasting subjects received a single dose of 30, 100, 300, 1,000, or 3,000 mg cadazolid and 10 subjects received matching placebo. After a wash-out period of 8-15 days, the six subjects who had taken 300 mg received a second dose of 300 mg after eating instead of after fasting. In the multiple-dose group, 18 subjects took 300, 1,000, or 3,000 mg of cadazolid twice a day and 6 received matching placebo for 10 days.
Taking cadazolid with food appeared to increase the rate and extent of drug absorption by two- to fivefold. Blood samples showed low systemic exposure after single or multiple doses, with a minor, twofold increase in cadazolid in plasma after 10 days for all doses in the twice-a-day group, Dr. Baldoni reported at the meeting, sponsored by the American Society for Microbiology.
The dose or duration of treatment did not seem to affect the number of adverse events (none of which were serious). They occurred in 27%-39% of cadazolid-treated subjects and in 17%-40% taking placebo and were mostly headache or diarrhea.
All subjects completed the study except one man in the 100-mg single-dose subgroup who withdrew consent for reasons unrelated to adverse events.
SAN FRANCISCO – Those desperate for new treatments for Clostridium difficile infection may want to keep an eye on the experimental oral antibiotic cadazolid, which looked promising in an early-phase trial, according to Daniela Baldoni, Pharm.D.
Cadazolid is in the oxazolidinone class of antibiotics. Its mechanism of action consists mainly of bacterial protein-synthesis inhibition.
Cadazolid produced low systemic exposure with high concentrations at the desired site – the colon – and was well tolerated in 64 healthy men who received up to 3,000 mg b.i.d. for 10 days, she reported in a poster presentation at the conference. Dr. Baldoni is employed by Actelion Pharmaceuticals, the company that is developing cadazolid.
The study randomized nonsmoking men aged 45-60 years and a body mass index of 18-32 kg/m2 to single or multiple doses of cadazolid or placebo.
In the single-dose group, 30 fasting subjects received a single dose of 30, 100, 300, 1,000, or 3,000 mg cadazolid and 10 subjects received matching placebo. After a wash-out period of 8-15 days, the six subjects who had taken 300 mg received a second dose of 300 mg after eating instead of after fasting. In the multiple-dose group, 18 subjects took 300, 1,000, or 3,000 mg of cadazolid twice a day and 6 received matching placebo for 10 days.
Taking cadazolid with food appeared to increase the rate and extent of drug absorption by two- to fivefold. Blood samples showed low systemic exposure after single or multiple doses, with a minor, twofold increase in cadazolid in plasma after 10 days for all doses in the twice-a-day group, Dr. Baldoni reported at the meeting, sponsored by the American Society for Microbiology.
The dose or duration of treatment did not seem to affect the number of adverse events (none of which were serious). They occurred in 27%-39% of cadazolid-treated subjects and in 17%-40% taking placebo and were mostly headache or diarrhea.
All subjects completed the study except one man in the 100-mg single-dose subgroup who withdrew consent for reasons unrelated to adverse events.
SAN FRANCISCO – Those desperate for new treatments for Clostridium difficile infection may want to keep an eye on the experimental oral antibiotic cadazolid, which looked promising in an early-phase trial, according to Daniela Baldoni, Pharm.D.
Cadazolid is in the oxazolidinone class of antibiotics. Its mechanism of action consists mainly of bacterial protein-synthesis inhibition.
Cadazolid produced low systemic exposure with high concentrations at the desired site – the colon – and was well tolerated in 64 healthy men who received up to 3,000 mg b.i.d. for 10 days, she reported in a poster presentation at the conference. Dr. Baldoni is employed by Actelion Pharmaceuticals, the company that is developing cadazolid.
The study randomized nonsmoking men aged 45-60 years and a body mass index of 18-32 kg/m2 to single or multiple doses of cadazolid or placebo.
In the single-dose group, 30 fasting subjects received a single dose of 30, 100, 300, 1,000, or 3,000 mg cadazolid and 10 subjects received matching placebo. After a wash-out period of 8-15 days, the six subjects who had taken 300 mg received a second dose of 300 mg after eating instead of after fasting. In the multiple-dose group, 18 subjects took 300, 1,000, or 3,000 mg of cadazolid twice a day and 6 received matching placebo for 10 days.
Taking cadazolid with food appeared to increase the rate and extent of drug absorption by two- to fivefold. Blood samples showed low systemic exposure after single or multiple doses, with a minor, twofold increase in cadazolid in plasma after 10 days for all doses in the twice-a-day group, Dr. Baldoni reported at the meeting, sponsored by the American Society for Microbiology.
The dose or duration of treatment did not seem to affect the number of adverse events (none of which were serious). They occurred in 27%-39% of cadazolid-treated subjects and in 17%-40% taking placebo and were mostly headache or diarrhea.
All subjects completed the study except one man in the 100-mg single-dose subgroup who withdrew consent for reasons unrelated to adverse events.
Major Finding: The experimental antibiotic cadazolid concentrated in feces with low systemic exposure and few side effects after single doses or twice-a-day dosing for 10 days.
Data Source: Data are from a randomized, placebo-controlled study in 64 healthy, nonsmoking men.
Disclosures: Dr. Baldoni and most of her coinvestigators are employees of Actelion Pharmaceuticals, which funded the study.
Higher Dose for Severe C. difficile Speeds Response
SAN FRANCISCO – Two small studies suggest that treating severe Clostridium difficile infection with a higher initial dose of vancomycin may work better than the recommended dose of 125 mg every 6 hours.
The most recent study, presented in a poster at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy, surprised the investigators.
"Pharmacodynamically, the concentrations in stool of the standard dose of 125 mg are about 500-1,000 times greater than the MIC [minimum inhibitory concentration]," Yleana T. Garcia, Pharm.D., said in an interview. "So, we have enough concentration in the stool. We wanted to see that we have similar outcomes with patients who are treated with standard doses as with higher doses."
Instead, the retrospective review of 62 patients with severe diarrhea who received oral vancomycin for at least 3 days found that symptoms resolved significantly quicker in 19 patients who got 250 mg every 6 hours compared with 43 patients who got 125 mg every 6 hours, she and her associates reported.
Symptoms resolved by day 3 in nine patients (47%) on the high dose and six patients (15%) on the conventional dose, said Dr. Garcia, a palliative care fellow at the James J. Peters Veterans Affairs Medical Center, Bronx, N.Y.
There also were statistically nonsignificant trends toward a higher likelihood of clinical cure, shorter length of stay, and reduced risk of recurrence in patients with the higher dose. An increased death rate in the higher-dose group also was not statistically significant, and might be due to greater severity of illness at baseline in patients who got the higher dose of vancomycin, she said at the meeting, sponsored by the American Society for Microbiology.
The findings support those of a small prospective study that analyzed levels of vancomycin in feces collected from 15 patients with presumed or confirmed C. difficile infection. Drug concentrations were high in patients who got 250 or 500 mg q.i.d. but were inadequate in at least one patient on the first day of treatment with 125 mg q.i.d. (BMC Infect. Dis. 2010;10:363).<< http://www.biomedcentral.com/1471-2334/10/363 >>
"Higher doses like 250-500 mg may be warranted to reach adequate concentrations in the stool in the first 24-48 hours," Dr. Garcia said. "I’m not saying to use 250 for the whole treatment course, but there may be a role for a loading dose of 250 mg q6 for the first 24-48 hours, and then switching to 125 mg q6 for the remainder of the treatment course. We know that 125 does have adequate fecal concentration; it just may not be adequate on day 1."
The 2010 update to clinical practice guidelines for C. difficile infection in adults recommends treating severe C. difficile infection with oral vancomycin 125 mg every 6 hours or using 500 mg every 6 hours for patients with severe disease complicated by ileus, megacolon, or hypotension (Infect. Control Hosp. Epidemiol. 2010;31:431-55).
Severe C. difficile infection generally is defined as the presence of the organism plus leukocytosis with a white blood cell count of 15,000 cells/microL or greater, or a serum creatinine level at least 1.5 times baseline. The study reviewed records of patients who received vancomycin for these indications or hypotension, shock, ileus, megacolon, or evidence of colitis. The study excluded patients who were treated with any other medication besides metronidazole.
The study is continuing in order to increase the number of patients reviewed and the power of the findings.
Dr. Garcia reported having no financial disclosures.
SAN FRANCISCO – Two small studies suggest that treating severe Clostridium difficile infection with a higher initial dose of vancomycin may work better than the recommended dose of 125 mg every 6 hours.
The most recent study, presented in a poster at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy, surprised the investigators.
"Pharmacodynamically, the concentrations in stool of the standard dose of 125 mg are about 500-1,000 times greater than the MIC [minimum inhibitory concentration]," Yleana T. Garcia, Pharm.D., said in an interview. "So, we have enough concentration in the stool. We wanted to see that we have similar outcomes with patients who are treated with standard doses as with higher doses."
Instead, the retrospective review of 62 patients with severe diarrhea who received oral vancomycin for at least 3 days found that symptoms resolved significantly quicker in 19 patients who got 250 mg every 6 hours compared with 43 patients who got 125 mg every 6 hours, she and her associates reported.
Symptoms resolved by day 3 in nine patients (47%) on the high dose and six patients (15%) on the conventional dose, said Dr. Garcia, a palliative care fellow at the James J. Peters Veterans Affairs Medical Center, Bronx, N.Y.
There also were statistically nonsignificant trends toward a higher likelihood of clinical cure, shorter length of stay, and reduced risk of recurrence in patients with the higher dose. An increased death rate in the higher-dose group also was not statistically significant, and might be due to greater severity of illness at baseline in patients who got the higher dose of vancomycin, she said at the meeting, sponsored by the American Society for Microbiology.
The findings support those of a small prospective study that analyzed levels of vancomycin in feces collected from 15 patients with presumed or confirmed C. difficile infection. Drug concentrations were high in patients who got 250 or 500 mg q.i.d. but were inadequate in at least one patient on the first day of treatment with 125 mg q.i.d. (BMC Infect. Dis. 2010;10:363).<< http://www.biomedcentral.com/1471-2334/10/363 >>
"Higher doses like 250-500 mg may be warranted to reach adequate concentrations in the stool in the first 24-48 hours," Dr. Garcia said. "I’m not saying to use 250 for the whole treatment course, but there may be a role for a loading dose of 250 mg q6 for the first 24-48 hours, and then switching to 125 mg q6 for the remainder of the treatment course. We know that 125 does have adequate fecal concentration; it just may not be adequate on day 1."
The 2010 update to clinical practice guidelines for C. difficile infection in adults recommends treating severe C. difficile infection with oral vancomycin 125 mg every 6 hours or using 500 mg every 6 hours for patients with severe disease complicated by ileus, megacolon, or hypotension (Infect. Control Hosp. Epidemiol. 2010;31:431-55).
Severe C. difficile infection generally is defined as the presence of the organism plus leukocytosis with a white blood cell count of 15,000 cells/microL or greater, or a serum creatinine level at least 1.5 times baseline. The study reviewed records of patients who received vancomycin for these indications or hypotension, shock, ileus, megacolon, or evidence of colitis. The study excluded patients who were treated with any other medication besides metronidazole.
The study is continuing in order to increase the number of patients reviewed and the power of the findings.
Dr. Garcia reported having no financial disclosures.
SAN FRANCISCO – Two small studies suggest that treating severe Clostridium difficile infection with a higher initial dose of vancomycin may work better than the recommended dose of 125 mg every 6 hours.
The most recent study, presented in a poster at the annual Interscience Conference on Antimicrobial Agents and Chemotherapy, surprised the investigators.
"Pharmacodynamically, the concentrations in stool of the standard dose of 125 mg are about 500-1,000 times greater than the MIC [minimum inhibitory concentration]," Yleana T. Garcia, Pharm.D., said in an interview. "So, we have enough concentration in the stool. We wanted to see that we have similar outcomes with patients who are treated with standard doses as with higher doses."
Instead, the retrospective review of 62 patients with severe diarrhea who received oral vancomycin for at least 3 days found that symptoms resolved significantly quicker in 19 patients who got 250 mg every 6 hours compared with 43 patients who got 125 mg every 6 hours, she and her associates reported.
Symptoms resolved by day 3 in nine patients (47%) on the high dose and six patients (15%) on the conventional dose, said Dr. Garcia, a palliative care fellow at the James J. Peters Veterans Affairs Medical Center, Bronx, N.Y.
There also were statistically nonsignificant trends toward a higher likelihood of clinical cure, shorter length of stay, and reduced risk of recurrence in patients with the higher dose. An increased death rate in the higher-dose group also was not statistically significant, and might be due to greater severity of illness at baseline in patients who got the higher dose of vancomycin, she said at the meeting, sponsored by the American Society for Microbiology.
The findings support those of a small prospective study that analyzed levels of vancomycin in feces collected from 15 patients with presumed or confirmed C. difficile infection. Drug concentrations were high in patients who got 250 or 500 mg q.i.d. but were inadequate in at least one patient on the first day of treatment with 125 mg q.i.d. (BMC Infect. Dis. 2010;10:363).<< http://www.biomedcentral.com/1471-2334/10/363 >>
"Higher doses like 250-500 mg may be warranted to reach adequate concentrations in the stool in the first 24-48 hours," Dr. Garcia said. "I’m not saying to use 250 for the whole treatment course, but there may be a role for a loading dose of 250 mg q6 for the first 24-48 hours, and then switching to 125 mg q6 for the remainder of the treatment course. We know that 125 does have adequate fecal concentration; it just may not be adequate on day 1."
The 2010 update to clinical practice guidelines for C. difficile infection in adults recommends treating severe C. difficile infection with oral vancomycin 125 mg every 6 hours or using 500 mg every 6 hours for patients with severe disease complicated by ileus, megacolon, or hypotension (Infect. Control Hosp. Epidemiol. 2010;31:431-55).
Severe C. difficile infection generally is defined as the presence of the organism plus leukocytosis with a white blood cell count of 15,000 cells/microL or greater, or a serum creatinine level at least 1.5 times baseline. The study reviewed records of patients who received vancomycin for these indications or hypotension, shock, ileus, megacolon, or evidence of colitis. The study excluded patients who were treated with any other medication besides metronidazole.
The study is continuing in order to increase the number of patients reviewed and the power of the findings.
Dr. Garcia reported having no financial disclosures.
Major Finding: Symptoms of severe C. difficile infection resolved by day 3 of oral vancomycin treatment in 9 of 19 patients treated with 250 mg every 6 hours (47%) compared with 6 of 43 patients on 125 mg every 6 hours (15%).
Data Source: Retrospective review of records on 62 adults at one institution treated for at least 3 days with oral vancomycin for severe C. difficile infection.
Disclosures: Dr. Garcia reported having no financial disclosures.
Analysis Details the GI Disease Burden in U.S.
Clostridium difficile contributes mightily to the overall burden of gastrointestinal disease in the United States and was associated with a 237% increase in hospitalizations in the last decade.
Researchers who examined the latest data on the nationwide toll of GI and liver disease also found a 314% rise in hospitalizations related to morbid obesity and a continuing national health burden exacted by reflux symptoms, Barrett’s esophagus, and colorectal cancer.
Video from the American Gastroenterological Association (http://www.youtube.com/amergastroassn)
"We compiled the most recently available statistics on GI symptoms, quality of life, outpatient diagnoses, hospitalizations, costs, mortality, and endoscopic utilization from a variety of publicly and privately held databases," Dr. Anne F. Peery of the University of North Carolina, Chapel Hill, and her colleagues reported in the November issue of Gastroenterology (doi:10.1053/j.gastro.2012.08.002).
"Payers, policy makers, clinicians, and others interested in resource utilization may use these statistics to better understand evolving disease trends, and the best way to meet the challenge of these diseases."
The findings are based on data for 2009, the most recent year for which complete information was available, from the National Ambulatory Medical Care Survey, sponsored by the U.S. Centers for Disease Control and Prevention; the United States National Health and Wellness Survey, sponsored by the private company Kantar Health; the Nationwide Inpatient Sample, sponsored by the Agency for Healthcare Research and Quality; the Surveillance, Epidemiology, and End Results database of the National Cancer Institute; the National Vital Statistics System, sponsored by the National Center for Health Statistics and the CDC; and the Thomson Reuters MarketScan’s databases of commercial, Medicare, and Medicaid records.
Among the findings:
• C. difficile hospitalizations have increased 237% since 2000 and were associated with 4% in-hospital mortality. Now the ninth leading GI cause of mortality, with an absolute increase of 230% in the number of C. difficile–related deaths since 2002, the infection also markedly impairs quality of life and the capacity for work and other activities.
• Hospitalizations related to obesity remained relatively stable since 2000, but those associated with morbid obesity rose by 314%, and many were likely caused by the marked increase in bariatric surgery.
• Gastroesophageal reflux remains the most common GI-associated diagnosis in primary care, accounting for 9 million outpatient visits in 2009, and the most common GI-associated discharge diagnosis, with 4.4 million such diagnoses in 2009. Obesity was associated with 1.7 million discharge diagnoses and constipation with 1 million.
• Barrett’s esophagus accounted for almost half a million outpatient visits in 2009, when an estimated 3.3 million Americans had this diagnosis. Given that endoscopic surveillance is recommended every 3-5 years, Barrett’s contributes substantially to resource utilization.
• Colorectal cancer, with an estimated 147,000 patients diagnosed in 2008, accounts for more than half of all GI cancer diagnoses and continues to be the primary cause of GI-associated mortality. Pancreatic and hepatobiliary cancers are the next most frequently diagnosed GI cancers.
• Of the approximately 2.5 million deaths in the United States in 2009, 10% were attributed to an underlying GI cause. Chronic liver disease and cirrhosis are the 12th leading causes of death in the country.
• The total outpatient cost for GI endoscopy in 2009 was estimated to be $32.4 billion, which is higher than previously published estimates. An estimated 6.9 million upper endoscopies, 11.5 million lower endoscopies, and 228,000 biliary endoscopies were performed in the United States in 2009.
• Chronic liver disease and viral hepatitis were associated with 6% mortality and cost an estimated $1.8 billion per year in inpatient cost.
• Hospitalizations for nonalcoholic fatty liver disease increased 97% since 2000.
This study was supported in part by the National Institutes of Health. No financial conflicts of interest were reported.
Digestive (GI and liver) diseases constitute a substantial and growing burden in the United States. As detailed in the report by Dr. Peery and colleagues, there were over 46 million outpatient encounters associated with the top 20 digestive disease diagnoses in 2009, with approximately 10% of deaths nationwide with an underlying digestive disease cause. The observed increased prevalence of hospitalizations for many diagnoses (e.g., a 14% increase with principal discharge diagnosis of chronic liver disease with viral hepatitis) and procedures (e.g., a 17% increase in lower GI endoscopies among commercially insured patients) between 2000 and 2009 is expected given population growth and aging. In addition, dramatic increases in hospitalizations associated with C. difficile and morbid obesity were also noted.
The report provides crucial information for diverse constituencies, including healthcare planners, clinicians and researchers. However, as acknowledged by the authors, there are some important caveats with respect to coverage or quality for some data sources to bear in mind when interpreting these results. This is particularly relevant for non-alcoholic fatty liver disease, which is likely underestimated because of well-known problems in diagnostic code specificity and use.
Several factors suggest that the prevalence and costs of digestive diseases will increase substantially during the next decade. These include: an aging population with the number of people aged 65 years or older projected to be greater than 54 million by 2020;the estimated tens of millions of individuals with newly available healthcare coverage as of 2014 as part of the Affordable Care Act; continued increases in obesity rates; and the recent CDC recommendation that all baby-boomers be screened for hepatitis C. This report will facilitate timely planning and also serve as benchmark to help measure the impact of these forces on the scope and burden of digestive diseases and their clinical management.
DONNA L. WHITE, PH.D., MPH, is an investigator in the Clinical Epidemiology and Outcomes Program in the Houston VA Health Services Research and Development Center of Excellence at the Michael E. DeBakey VA Medical Center, Houston. She also is an assistant professor in the department of medicine at Baylor College of Medicine, Houston.
Digestive (GI and liver) diseases constitute a substantial and growing burden in the United States. As detailed in the report by Dr. Peery and colleagues, there were over 46 million outpatient encounters associated with the top 20 digestive disease diagnoses in 2009, with approximately 10% of deaths nationwide with an underlying digestive disease cause. The observed increased prevalence of hospitalizations for many diagnoses (e.g., a 14% increase with principal discharge diagnosis of chronic liver disease with viral hepatitis) and procedures (e.g., a 17% increase in lower GI endoscopies among commercially insured patients) between 2000 and 2009 is expected given population growth and aging. In addition, dramatic increases in hospitalizations associated with C. difficile and morbid obesity were also noted.
The report provides crucial information for diverse constituencies, including healthcare planners, clinicians and researchers. However, as acknowledged by the authors, there are some important caveats with respect to coverage or quality for some data sources to bear in mind when interpreting these results. This is particularly relevant for non-alcoholic fatty liver disease, which is likely underestimated because of well-known problems in diagnostic code specificity and use.
Several factors suggest that the prevalence and costs of digestive diseases will increase substantially during the next decade. These include: an aging population with the number of people aged 65 years or older projected to be greater than 54 million by 2020;the estimated tens of millions of individuals with newly available healthcare coverage as of 2014 as part of the Affordable Care Act; continued increases in obesity rates; and the recent CDC recommendation that all baby-boomers be screened for hepatitis C. This report will facilitate timely planning and also serve as benchmark to help measure the impact of these forces on the scope and burden of digestive diseases and their clinical management.
DONNA L. WHITE, PH.D., MPH, is an investigator in the Clinical Epidemiology and Outcomes Program in the Houston VA Health Services Research and Development Center of Excellence at the Michael E. DeBakey VA Medical Center, Houston. She also is an assistant professor in the department of medicine at Baylor College of Medicine, Houston.
Digestive (GI and liver) diseases constitute a substantial and growing burden in the United States. As detailed in the report by Dr. Peery and colleagues, there were over 46 million outpatient encounters associated with the top 20 digestive disease diagnoses in 2009, with approximately 10% of deaths nationwide with an underlying digestive disease cause. The observed increased prevalence of hospitalizations for many diagnoses (e.g., a 14% increase with principal discharge diagnosis of chronic liver disease with viral hepatitis) and procedures (e.g., a 17% increase in lower GI endoscopies among commercially insured patients) between 2000 and 2009 is expected given population growth and aging. In addition, dramatic increases in hospitalizations associated with C. difficile and morbid obesity were also noted.
The report provides crucial information for diverse constituencies, including healthcare planners, clinicians and researchers. However, as acknowledged by the authors, there are some important caveats with respect to coverage or quality for some data sources to bear in mind when interpreting these results. This is particularly relevant for non-alcoholic fatty liver disease, which is likely underestimated because of well-known problems in diagnostic code specificity and use.
Several factors suggest that the prevalence and costs of digestive diseases will increase substantially during the next decade. These include: an aging population with the number of people aged 65 years or older projected to be greater than 54 million by 2020;the estimated tens of millions of individuals with newly available healthcare coverage as of 2014 as part of the Affordable Care Act; continued increases in obesity rates; and the recent CDC recommendation that all baby-boomers be screened for hepatitis C. This report will facilitate timely planning and also serve as benchmark to help measure the impact of these forces on the scope and burden of digestive diseases and their clinical management.
DONNA L. WHITE, PH.D., MPH, is an investigator in the Clinical Epidemiology and Outcomes Program in the Houston VA Health Services Research and Development Center of Excellence at the Michael E. DeBakey VA Medical Center, Houston. She also is an assistant professor in the department of medicine at Baylor College of Medicine, Houston.
Clostridium difficile contributes mightily to the overall burden of gastrointestinal disease in the United States and was associated with a 237% increase in hospitalizations in the last decade.
Researchers who examined the latest data on the nationwide toll of GI and liver disease also found a 314% rise in hospitalizations related to morbid obesity and a continuing national health burden exacted by reflux symptoms, Barrett’s esophagus, and colorectal cancer.
Video from the American Gastroenterological Association (http://www.youtube.com/amergastroassn)
"We compiled the most recently available statistics on GI symptoms, quality of life, outpatient diagnoses, hospitalizations, costs, mortality, and endoscopic utilization from a variety of publicly and privately held databases," Dr. Anne F. Peery of the University of North Carolina, Chapel Hill, and her colleagues reported in the November issue of Gastroenterology (doi:10.1053/j.gastro.2012.08.002).
"Payers, policy makers, clinicians, and others interested in resource utilization may use these statistics to better understand evolving disease trends, and the best way to meet the challenge of these diseases."
The findings are based on data for 2009, the most recent year for which complete information was available, from the National Ambulatory Medical Care Survey, sponsored by the U.S. Centers for Disease Control and Prevention; the United States National Health and Wellness Survey, sponsored by the private company Kantar Health; the Nationwide Inpatient Sample, sponsored by the Agency for Healthcare Research and Quality; the Surveillance, Epidemiology, and End Results database of the National Cancer Institute; the National Vital Statistics System, sponsored by the National Center for Health Statistics and the CDC; and the Thomson Reuters MarketScan’s databases of commercial, Medicare, and Medicaid records.
Among the findings:
• C. difficile hospitalizations have increased 237% since 2000 and were associated with 4% in-hospital mortality. Now the ninth leading GI cause of mortality, with an absolute increase of 230% in the number of C. difficile–related deaths since 2002, the infection also markedly impairs quality of life and the capacity for work and other activities.
• Hospitalizations related to obesity remained relatively stable since 2000, but those associated with morbid obesity rose by 314%, and many were likely caused by the marked increase in bariatric surgery.
• Gastroesophageal reflux remains the most common GI-associated diagnosis in primary care, accounting for 9 million outpatient visits in 2009, and the most common GI-associated discharge diagnosis, with 4.4 million such diagnoses in 2009. Obesity was associated with 1.7 million discharge diagnoses and constipation with 1 million.
• Barrett’s esophagus accounted for almost half a million outpatient visits in 2009, when an estimated 3.3 million Americans had this diagnosis. Given that endoscopic surveillance is recommended every 3-5 years, Barrett’s contributes substantially to resource utilization.
• Colorectal cancer, with an estimated 147,000 patients diagnosed in 2008, accounts for more than half of all GI cancer diagnoses and continues to be the primary cause of GI-associated mortality. Pancreatic and hepatobiliary cancers are the next most frequently diagnosed GI cancers.
• Of the approximately 2.5 million deaths in the United States in 2009, 10% were attributed to an underlying GI cause. Chronic liver disease and cirrhosis are the 12th leading causes of death in the country.
• The total outpatient cost for GI endoscopy in 2009 was estimated to be $32.4 billion, which is higher than previously published estimates. An estimated 6.9 million upper endoscopies, 11.5 million lower endoscopies, and 228,000 biliary endoscopies were performed in the United States in 2009.
• Chronic liver disease and viral hepatitis were associated with 6% mortality and cost an estimated $1.8 billion per year in inpatient cost.
• Hospitalizations for nonalcoholic fatty liver disease increased 97% since 2000.
This study was supported in part by the National Institutes of Health. No financial conflicts of interest were reported.
Clostridium difficile contributes mightily to the overall burden of gastrointestinal disease in the United States and was associated with a 237% increase in hospitalizations in the last decade.
Researchers who examined the latest data on the nationwide toll of GI and liver disease also found a 314% rise in hospitalizations related to morbid obesity and a continuing national health burden exacted by reflux symptoms, Barrett’s esophagus, and colorectal cancer.
Video from the American Gastroenterological Association (http://www.youtube.com/amergastroassn)
"We compiled the most recently available statistics on GI symptoms, quality of life, outpatient diagnoses, hospitalizations, costs, mortality, and endoscopic utilization from a variety of publicly and privately held databases," Dr. Anne F. Peery of the University of North Carolina, Chapel Hill, and her colleagues reported in the November issue of Gastroenterology (doi:10.1053/j.gastro.2012.08.002).
"Payers, policy makers, clinicians, and others interested in resource utilization may use these statistics to better understand evolving disease trends, and the best way to meet the challenge of these diseases."
The findings are based on data for 2009, the most recent year for which complete information was available, from the National Ambulatory Medical Care Survey, sponsored by the U.S. Centers for Disease Control and Prevention; the United States National Health and Wellness Survey, sponsored by the private company Kantar Health; the Nationwide Inpatient Sample, sponsored by the Agency for Healthcare Research and Quality; the Surveillance, Epidemiology, and End Results database of the National Cancer Institute; the National Vital Statistics System, sponsored by the National Center for Health Statistics and the CDC; and the Thomson Reuters MarketScan’s databases of commercial, Medicare, and Medicaid records.
Among the findings:
• C. difficile hospitalizations have increased 237% since 2000 and were associated with 4% in-hospital mortality. Now the ninth leading GI cause of mortality, with an absolute increase of 230% in the number of C. difficile–related deaths since 2002, the infection also markedly impairs quality of life and the capacity for work and other activities.
• Hospitalizations related to obesity remained relatively stable since 2000, but those associated with morbid obesity rose by 314%, and many were likely caused by the marked increase in bariatric surgery.
• Gastroesophageal reflux remains the most common GI-associated diagnosis in primary care, accounting for 9 million outpatient visits in 2009, and the most common GI-associated discharge diagnosis, with 4.4 million such diagnoses in 2009. Obesity was associated with 1.7 million discharge diagnoses and constipation with 1 million.
• Barrett’s esophagus accounted for almost half a million outpatient visits in 2009, when an estimated 3.3 million Americans had this diagnosis. Given that endoscopic surveillance is recommended every 3-5 years, Barrett’s contributes substantially to resource utilization.
• Colorectal cancer, with an estimated 147,000 patients diagnosed in 2008, accounts for more than half of all GI cancer diagnoses and continues to be the primary cause of GI-associated mortality. Pancreatic and hepatobiliary cancers are the next most frequently diagnosed GI cancers.
• Of the approximately 2.5 million deaths in the United States in 2009, 10% were attributed to an underlying GI cause. Chronic liver disease and cirrhosis are the 12th leading causes of death in the country.
• The total outpatient cost for GI endoscopy in 2009 was estimated to be $32.4 billion, which is higher than previously published estimates. An estimated 6.9 million upper endoscopies, 11.5 million lower endoscopies, and 228,000 biliary endoscopies were performed in the United States in 2009.
• Chronic liver disease and viral hepatitis were associated with 6% mortality and cost an estimated $1.8 billion per year in inpatient cost.
• Hospitalizations for nonalcoholic fatty liver disease increased 97% since 2000.
This study was supported in part by the National Institutes of Health. No financial conflicts of interest were reported.
FROM GASTROENTEROLOGY
Second TNF Blocker Approved for Refractory UC
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating ulcerative colitis.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference.
In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication – adults with moderately to severely active ulcerative colitis who have not had an adequate response to conventional treatment.
Panelists cited the need for more treatments for ulcerative colitis and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said.
The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week.
"The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
It is good news for patients who have ulcerative colitis and the health care providers who take care of them that a second anti-TNF agent is now available. The FDA’s approval of Humira (adalimumab) for moderate to severe ulcer-ative colitis provides us with an additional option that we definitely need because there are many patients who suffer from ulcerative colitis and fail to respond to the "conventional" treatments with aminosalicylates or steroids and thiopurines, or lose response to infliximab. Adalimumab is an option for many of these types of patients and may be the first choice anti–tumor necrosis factor agent for some patients or providers, due to the injectable delivery method for this therapy.
Despite this good news, however, we need to acknowledge that 8-week remission rates of 18.5% and 16.5% leave a lot of room for improvement. Future studies with adalimumab in ulcerative colitis will focus on how to optimize this therapy and will explore adjustable dosing schedules, combination therapies, and other important longer-term outcomes such as sustained remission and mucosal healing.
David T. Rubin, M.D., AGAF, is a professor of medicine and co-director of the Inflammatory Bowel Disease Center at the University of Chicago. He has served as a consultant for Janssen and for Abbott.
It is good news for patients who have ulcerative colitis and the health care providers who take care of them that a second anti-TNF agent is now available. The FDA’s approval of Humira (adalimumab) for moderate to severe ulcer-ative colitis provides us with an additional option that we definitely need because there are many patients who suffer from ulcerative colitis and fail to respond to the "conventional" treatments with aminosalicylates or steroids and thiopurines, or lose response to infliximab. Adalimumab is an option for many of these types of patients and may be the first choice anti–tumor necrosis factor agent for some patients or providers, due to the injectable delivery method for this therapy.
Despite this good news, however, we need to acknowledge that 8-week remission rates of 18.5% and 16.5% leave a lot of room for improvement. Future studies with adalimumab in ulcerative colitis will focus on how to optimize this therapy and will explore adjustable dosing schedules, combination therapies, and other important longer-term outcomes such as sustained remission and mucosal healing.
David T. Rubin, M.D., AGAF, is a professor of medicine and co-director of the Inflammatory Bowel Disease Center at the University of Chicago. He has served as a consultant for Janssen and for Abbott.
It is good news for patients who have ulcerative colitis and the health care providers who take care of them that a second anti-TNF agent is now available. The FDA’s approval of Humira (adalimumab) for moderate to severe ulcer-ative colitis provides us with an additional option that we definitely need because there are many patients who suffer from ulcerative colitis and fail to respond to the "conventional" treatments with aminosalicylates or steroids and thiopurines, or lose response to infliximab. Adalimumab is an option for many of these types of patients and may be the first choice anti–tumor necrosis factor agent for some patients or providers, due to the injectable delivery method for this therapy.
Despite this good news, however, we need to acknowledge that 8-week remission rates of 18.5% and 16.5% leave a lot of room for improvement. Future studies with adalimumab in ulcerative colitis will focus on how to optimize this therapy and will explore adjustable dosing schedules, combination therapies, and other important longer-term outcomes such as sustained remission and mucosal healing.
David T. Rubin, M.D., AGAF, is a professor of medicine and co-director of the Inflammatory Bowel Disease Center at the University of Chicago. He has served as a consultant for Janssen and for Abbott.
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating ulcerative colitis.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference.
In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication – adults with moderately to severely active ulcerative colitis who have not had an adequate response to conventional treatment.
Panelists cited the need for more treatments for ulcerative colitis and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said.
The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week.
"The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.
Adalimumab, a subcutaneously administered tumor necrosis factor blocker, has been approved for treating adults with moderately to severely active ulcerative colitis who have not had an adequate response with conventional treatments, the Food and Drug Administration announced.
The safety and effectiveness of adalimumab for this patient population was established in two clinical studies of 908 patients with moderately to severely active ulcerative colitis (UC).
Adalimumab, marketed as Humira by Abbott Laboratories, was first approved for treating rheumatoid arthritis in 2002, followed by psoriatic arthritis in 2005, ankylosing spondylitis in 2006, Crohn’s disease in 2007, and plaque psoriasis and juvenile idiopathic arthritis in 2008.
Adalimumab is the second TNF blocker to be approved for ulcerative colitis; infliximab (Remicade), an intravenous TNF blocker, was previously approved for treating ulcerative colitis.
Clinical remission rates in the two studies were significantly greater among patients treated with infliximab than among those who received placebo: In an 8-week study, which did not include patients who had previously been treated with a TNF blocker, the clinical remission rate at 8 weeks was 18.5% among those on adalimumab vs. 9.2% in those on placebo, a 9.3% difference.
In the second study, which followed patients for 1 year and included some who had been treated with infliximab, the clinical remission rate at 8 weeks was 16.5% among those on adalimumab, vs. 9.3% among those on placebo, a 7.2% difference.
At a meeting on Aug. 28 held to review these data, the majority of the FDA’s Gastrointestinal Drugs Advisory Committee agreed that these differences represented clinically meaningful benefits and supported approval of adalimumab for this indication – adults with moderately to severely active ulcerative colitis who have not had an adequate response to conventional treatment.
Panelists cited the need for more treatments for ulcerative colitis and for a subcutaneous TNF blocker for these patients, as well as its potential steroid-sparing effects.
In the studies, no new side effects were identified, the agency said.
The FDA statement points out that the effectiveness of adalimumab "has not been established in patients with ulcerative colitis who have lost response to or were intolerant to TNF blockers."
The approved dosing regimen for adalimumab is a starting dose of 160 mg, followed by a second dose of 80 mg 2 weeks later and then a maintenance dose of 40 mg every other week.
"The drug should only continue to be used in patients who have shown evidence of clinical remission by 8 weeks of therapy," according to the FDA statement.
Adalimumab is the first self-administered biologic treatment for ulcerative colitis to be approved.