User login
Rapper sings about living with sickle cell disease
MANCHESTER, ENGLAND – A London-based rapper known for his gospel-inspired music has now given a voice to patients with sickle cell disease. He is using one of his music videos to raise awareness and educate health care professionals about living with the condition.
One important aim of the video, he says, is to help educate health care professionals, some of whom have not come across this condition, he explained at a session during the annual meeting of the British Society for Haematology, held recently in Manchester, England.
“It’s kind of frustrating to feel like your safe space, when you’re in front of doctors and nurses and paramedics who are supposed to know what it is and react with treatment, [and they] don’t know what it is,” Mr. Gaspar said.
He recalled an occasion in which he was experiencing a crisis, and his wife called for an ambulance. The paramedics arrived and his wife asked them for “gas and air and morphine, and they were, like, no, we don’t want to give that to him.” She tried to explain that he has sickle cell disease, but the paramedics had not heard of the condition and were suspicious that the request for morphine was a sign of drug addiction.
Mr. Gaspar expressed his frustration over “constantly having to prove that you have something serious enough to need the treatment you are asking for.”
At the meeting, Mr. Gaspar was talking on the stage with hematologist Dr. Stephen Hibbs from Barts Health NHS Trust, London.
Mr. Gaspar explained that it took years before he eventually reached “a point where I understood that it’s something that affects me and affects many other people, and I didn’t want to hide it any more.”
Sickle cell disease, which occurs primarily in people of Afro-Caribbean background, is a taboo subject in his community, Mr. Gaspar elaborated in an interview.
The condition has been associated with a great deal of stigma, with young sufferers traditionally seen as “demonically possessed,” he commented.
“So there was always a shameful aspect around it when it came to African families speaking about it, especially back in Africa.”
But after his parents came to the United Kingdom, he was able to “do his research and understand that it’s just genetics.”
This knowledge, Mr. Gaspar said, “takes away the spiritual aspect” and allows people to “have the conversation about sickle cell with potential partners” and ask them to find out their genotype, which in turn helps to “break down the barriers and the stigma.”
Mr. Gaspar emphasizes that there is much more work still to do.
In the video, he appeals to the Black community to make blood donations.
He said that something that “haunts” him is that currently, only 1% of Black people in the United Kingdom give blood, “so I really want the song to move my community to take a step forward and make that difference.”
He has been in contact with NHS Blood and Transplant, which provides blood and transplantation service to the National Health Service. They “really liked” the song, Mr. Gaspar said, and helped him get access to a hospital ward in University College Hospital, London, for the video.
“I really wanted to make a video that made people uncomfortable when watching it,” he said. It shows him hospitalized for pain and breathlessness and recalling having to use a Zimmer frame at the age of 25.
“This is a side of sickle cell that normally people don’t know,” he said.
Since releasing the song and the video, Mr. Gaspar says he has been contacted by many fellow patients. They have told him that he is now their “voice”; when they are asked how the condition affects them, “they can show someone the Hidden Pain video and say: This is how it feels.”
Clinicians have also approached him, asking if they can show his video to illustrate to patients and their families how having the condition may affect their lives.
Preventable deaths
At the meeting, Dr. Hibbs highlighted the 2021 report No One’s Listening, which was issued by the Sickle Cell Society following an inquiry into avoidable deaths and failures of care for sickle cell patients.
The inquiry, published by an All-Party Parliamentary group, found “serious care failings” in acute services and evidence of attitudes underpinned by racism. There was evidence of substandard care for sickle cell patients who were admitted to general wards or to hospital accident and emergency departments, as well as low awareness of the condition among health care professionals.
The report noted that the care failings have led to patient deaths, some which could have been prevented, and that there have been many “near misses.”
Many patients with sickle cell disease said they are “not being listened to” or are not being understood, especially during that vulnerable period when they are “in a crisis.”
Mr. Gaspar said that the report, and also the deaths, really struck a chord with him and many in his community. “We felt like that was us. ... We’ve all been in that same position where we’ve been misunderstood and not heard by nurses, doctors, or paramedics.”
He emphasized the need for awareness of the condition and the need for timely treatment. Just 3 weeks ago, Mr. Gaspar attended the funeral of one of his friends who is in the Hidden Pain video, a fellow sickle cell disease patient, who died at 30 years of age.
Ignorance about the condition ‘all too common’
The lack of awareness about sickle cell disease, even among health care professionals, is “all too common,” says Dr. Subarna Chakravorty, consultant pediatric hematologist, King’s College Hospital, London.
Even in London, where there is a large Black community and the teaching hospitals have world-class expertise, patients with sickle cell disease are “still facing a lot of problems with knowledge” among health care professionals, she said in an interview.
“Often people are having to speak for their own condition; which is fine, except that sometimes they are not believed,” she commented.
“On the one hand, you rely on the patient to provide information about their disease, and then when you receive it, you don’t do anything about it. So [they’re] between a rock and a hard place.”
Why are sickle cell patients treated in this way?
For Dr. Chakravorty, there is “a lot to be said about racism and disparities” in treating patients “as morphine-seekers, opiate addicts, even in children.”
“So we really need to improve the knowledge and perceptions among nonspecialist staff,” she said, “and even among specialists.”
Mr. Gaspar aims to help with this effort and hopes that his song and video will be useful to health care professionals. Sickle cell disease “needs to be spoken about,” and more doctors and nurses need to “know what it is,” he said.
He said it is a relief to encounter health care professionals who are knowledgeable about his condition. There have been times when he has been “having a crisis at home, calling the ambulance, and the paramedic comes and says: ‘Mr. Gaspar, you have sickle cell...we believe that you usually have gas and air and morphine, is that correct?’”
“That gives me a sense of peace, to know that I don’t have to fight my case or convince someone I have sickle cell, and I need to start treatment. They already know.”
No relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – A London-based rapper known for his gospel-inspired music has now given a voice to patients with sickle cell disease. He is using one of his music videos to raise awareness and educate health care professionals about living with the condition.
One important aim of the video, he says, is to help educate health care professionals, some of whom have not come across this condition, he explained at a session during the annual meeting of the British Society for Haematology, held recently in Manchester, England.
“It’s kind of frustrating to feel like your safe space, when you’re in front of doctors and nurses and paramedics who are supposed to know what it is and react with treatment, [and they] don’t know what it is,” Mr. Gaspar said.
He recalled an occasion in which he was experiencing a crisis, and his wife called for an ambulance. The paramedics arrived and his wife asked them for “gas and air and morphine, and they were, like, no, we don’t want to give that to him.” She tried to explain that he has sickle cell disease, but the paramedics had not heard of the condition and were suspicious that the request for morphine was a sign of drug addiction.
Mr. Gaspar expressed his frustration over “constantly having to prove that you have something serious enough to need the treatment you are asking for.”
At the meeting, Mr. Gaspar was talking on the stage with hematologist Dr. Stephen Hibbs from Barts Health NHS Trust, London.
Mr. Gaspar explained that it took years before he eventually reached “a point where I understood that it’s something that affects me and affects many other people, and I didn’t want to hide it any more.”
Sickle cell disease, which occurs primarily in people of Afro-Caribbean background, is a taboo subject in his community, Mr. Gaspar elaborated in an interview.
The condition has been associated with a great deal of stigma, with young sufferers traditionally seen as “demonically possessed,” he commented.
“So there was always a shameful aspect around it when it came to African families speaking about it, especially back in Africa.”
But after his parents came to the United Kingdom, he was able to “do his research and understand that it’s just genetics.”
This knowledge, Mr. Gaspar said, “takes away the spiritual aspect” and allows people to “have the conversation about sickle cell with potential partners” and ask them to find out their genotype, which in turn helps to “break down the barriers and the stigma.”
Mr. Gaspar emphasizes that there is much more work still to do.
In the video, he appeals to the Black community to make blood donations.
He said that something that “haunts” him is that currently, only 1% of Black people in the United Kingdom give blood, “so I really want the song to move my community to take a step forward and make that difference.”
He has been in contact with NHS Blood and Transplant, which provides blood and transplantation service to the National Health Service. They “really liked” the song, Mr. Gaspar said, and helped him get access to a hospital ward in University College Hospital, London, for the video.
“I really wanted to make a video that made people uncomfortable when watching it,” he said. It shows him hospitalized for pain and breathlessness and recalling having to use a Zimmer frame at the age of 25.
“This is a side of sickle cell that normally people don’t know,” he said.
Since releasing the song and the video, Mr. Gaspar says he has been contacted by many fellow patients. They have told him that he is now their “voice”; when they are asked how the condition affects them, “they can show someone the Hidden Pain video and say: This is how it feels.”
Clinicians have also approached him, asking if they can show his video to illustrate to patients and their families how having the condition may affect their lives.
Preventable deaths
At the meeting, Dr. Hibbs highlighted the 2021 report No One’s Listening, which was issued by the Sickle Cell Society following an inquiry into avoidable deaths and failures of care for sickle cell patients.
The inquiry, published by an All-Party Parliamentary group, found “serious care failings” in acute services and evidence of attitudes underpinned by racism. There was evidence of substandard care for sickle cell patients who were admitted to general wards or to hospital accident and emergency departments, as well as low awareness of the condition among health care professionals.
The report noted that the care failings have led to patient deaths, some which could have been prevented, and that there have been many “near misses.”
Many patients with sickle cell disease said they are “not being listened to” or are not being understood, especially during that vulnerable period when they are “in a crisis.”
Mr. Gaspar said that the report, and also the deaths, really struck a chord with him and many in his community. “We felt like that was us. ... We’ve all been in that same position where we’ve been misunderstood and not heard by nurses, doctors, or paramedics.”
He emphasized the need for awareness of the condition and the need for timely treatment. Just 3 weeks ago, Mr. Gaspar attended the funeral of one of his friends who is in the Hidden Pain video, a fellow sickle cell disease patient, who died at 30 years of age.
Ignorance about the condition ‘all too common’
The lack of awareness about sickle cell disease, even among health care professionals, is “all too common,” says Dr. Subarna Chakravorty, consultant pediatric hematologist, King’s College Hospital, London.
Even in London, where there is a large Black community and the teaching hospitals have world-class expertise, patients with sickle cell disease are “still facing a lot of problems with knowledge” among health care professionals, she said in an interview.
“Often people are having to speak for their own condition; which is fine, except that sometimes they are not believed,” she commented.
“On the one hand, you rely on the patient to provide information about their disease, and then when you receive it, you don’t do anything about it. So [they’re] between a rock and a hard place.”
Why are sickle cell patients treated in this way?
For Dr. Chakravorty, there is “a lot to be said about racism and disparities” in treating patients “as morphine-seekers, opiate addicts, even in children.”
“So we really need to improve the knowledge and perceptions among nonspecialist staff,” she said, “and even among specialists.”
Mr. Gaspar aims to help with this effort and hopes that his song and video will be useful to health care professionals. Sickle cell disease “needs to be spoken about,” and more doctors and nurses need to “know what it is,” he said.
He said it is a relief to encounter health care professionals who are knowledgeable about his condition. There have been times when he has been “having a crisis at home, calling the ambulance, and the paramedic comes and says: ‘Mr. Gaspar, you have sickle cell...we believe that you usually have gas and air and morphine, is that correct?’”
“That gives me a sense of peace, to know that I don’t have to fight my case or convince someone I have sickle cell, and I need to start treatment. They already know.”
No relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
MANCHESTER, ENGLAND – A London-based rapper known for his gospel-inspired music has now given a voice to patients with sickle cell disease. He is using one of his music videos to raise awareness and educate health care professionals about living with the condition.
One important aim of the video, he says, is to help educate health care professionals, some of whom have not come across this condition, he explained at a session during the annual meeting of the British Society for Haematology, held recently in Manchester, England.
“It’s kind of frustrating to feel like your safe space, when you’re in front of doctors and nurses and paramedics who are supposed to know what it is and react with treatment, [and they] don’t know what it is,” Mr. Gaspar said.
He recalled an occasion in which he was experiencing a crisis, and his wife called for an ambulance. The paramedics arrived and his wife asked them for “gas and air and morphine, and they were, like, no, we don’t want to give that to him.” She tried to explain that he has sickle cell disease, but the paramedics had not heard of the condition and were suspicious that the request for morphine was a sign of drug addiction.
Mr. Gaspar expressed his frustration over “constantly having to prove that you have something serious enough to need the treatment you are asking for.”
At the meeting, Mr. Gaspar was talking on the stage with hematologist Dr. Stephen Hibbs from Barts Health NHS Trust, London.
Mr. Gaspar explained that it took years before he eventually reached “a point where I understood that it’s something that affects me and affects many other people, and I didn’t want to hide it any more.”
Sickle cell disease, which occurs primarily in people of Afro-Caribbean background, is a taboo subject in his community, Mr. Gaspar elaborated in an interview.
The condition has been associated with a great deal of stigma, with young sufferers traditionally seen as “demonically possessed,” he commented.
“So there was always a shameful aspect around it when it came to African families speaking about it, especially back in Africa.”
But after his parents came to the United Kingdom, he was able to “do his research and understand that it’s just genetics.”
This knowledge, Mr. Gaspar said, “takes away the spiritual aspect” and allows people to “have the conversation about sickle cell with potential partners” and ask them to find out their genotype, which in turn helps to “break down the barriers and the stigma.”
Mr. Gaspar emphasizes that there is much more work still to do.
In the video, he appeals to the Black community to make blood donations.
He said that something that “haunts” him is that currently, only 1% of Black people in the United Kingdom give blood, “so I really want the song to move my community to take a step forward and make that difference.”
He has been in contact with NHS Blood and Transplant, which provides blood and transplantation service to the National Health Service. They “really liked” the song, Mr. Gaspar said, and helped him get access to a hospital ward in University College Hospital, London, for the video.
“I really wanted to make a video that made people uncomfortable when watching it,” he said. It shows him hospitalized for pain and breathlessness and recalling having to use a Zimmer frame at the age of 25.
“This is a side of sickle cell that normally people don’t know,” he said.
Since releasing the song and the video, Mr. Gaspar says he has been contacted by many fellow patients. They have told him that he is now their “voice”; when they are asked how the condition affects them, “they can show someone the Hidden Pain video and say: This is how it feels.”
Clinicians have also approached him, asking if they can show his video to illustrate to patients and their families how having the condition may affect their lives.
Preventable deaths
At the meeting, Dr. Hibbs highlighted the 2021 report No One’s Listening, which was issued by the Sickle Cell Society following an inquiry into avoidable deaths and failures of care for sickle cell patients.
The inquiry, published by an All-Party Parliamentary group, found “serious care failings” in acute services and evidence of attitudes underpinned by racism. There was evidence of substandard care for sickle cell patients who were admitted to general wards or to hospital accident and emergency departments, as well as low awareness of the condition among health care professionals.
The report noted that the care failings have led to patient deaths, some which could have been prevented, and that there have been many “near misses.”
Many patients with sickle cell disease said they are “not being listened to” or are not being understood, especially during that vulnerable period when they are “in a crisis.”
Mr. Gaspar said that the report, and also the deaths, really struck a chord with him and many in his community. “We felt like that was us. ... We’ve all been in that same position where we’ve been misunderstood and not heard by nurses, doctors, or paramedics.”
He emphasized the need for awareness of the condition and the need for timely treatment. Just 3 weeks ago, Mr. Gaspar attended the funeral of one of his friends who is in the Hidden Pain video, a fellow sickle cell disease patient, who died at 30 years of age.
Ignorance about the condition ‘all too common’
The lack of awareness about sickle cell disease, even among health care professionals, is “all too common,” says Dr. Subarna Chakravorty, consultant pediatric hematologist, King’s College Hospital, London.
Even in London, where there is a large Black community and the teaching hospitals have world-class expertise, patients with sickle cell disease are “still facing a lot of problems with knowledge” among health care professionals, she said in an interview.
“Often people are having to speak for their own condition; which is fine, except that sometimes they are not believed,” she commented.
“On the one hand, you rely on the patient to provide information about their disease, and then when you receive it, you don’t do anything about it. So [they’re] between a rock and a hard place.”
Why are sickle cell patients treated in this way?
For Dr. Chakravorty, there is “a lot to be said about racism and disparities” in treating patients “as morphine-seekers, opiate addicts, even in children.”
“So we really need to improve the knowledge and perceptions among nonspecialist staff,” she said, “and even among specialists.”
Mr. Gaspar aims to help with this effort and hopes that his song and video will be useful to health care professionals. Sickle cell disease “needs to be spoken about,” and more doctors and nurses need to “know what it is,” he said.
He said it is a relief to encounter health care professionals who are knowledgeable about his condition. There have been times when he has been “having a crisis at home, calling the ambulance, and the paramedic comes and says: ‘Mr. Gaspar, you have sickle cell...we believe that you usually have gas and air and morphine, is that correct?’”
“That gives me a sense of peace, to know that I don’t have to fight my case or convince someone I have sickle cell, and I need to start treatment. They already know.”
No relevant financial relationships have been disclosed.
A version of this article first appeared on Medscape.com.
Some leukemias detectable up to 16 years before diagnosis?
Previous analyses showed that monoclonal B-cell lymphocytosis (MBL), a CLL precursor state, has been detected up to 6 years before CLL diagnosis, the investigators explained, noting that “[a]nother prognostically relevant immunogenetic feature of CLL concerns the stereotype of the B-cell receptor immunoglobulins (BcR IG).”
“Indeed, distinct stereotyped subsets can be defined by the expression of shared sequence motifs and are associated with particular presentation and outcomes,” P. Martijn Kolijn, PhD, a researcher in the department of immunology at Erasmus Medical Center, Rotterdam, the Netherlands, and colleagues wrote in a brief report published online in Blood. In an effort to “gain insight into the composition of the BcR IG repertoire during the early stages of CLL,” the investigators utilized next-generation sequencing to analyze 124 blood samples taken from healthy individuals up to 22 years before they received a diagnosis of CLL or small lymphocytic leukemia (SLL). An additional 118 matched control samples were also analyzed.
Study subjects were participants in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
“First, unsurprisingly, we observed a significant difference in the frequency of the dominant clonotype in CLL patients versus controls with a median frequency of 54.9%, compared to only 0.38% in controls,” they wrote.
Among 28 patients whose lymphocyte counts were measured at baseline, 10 showed evidence of lymphocytosis up to 8 years before CLL diagnosis.
This suggests undiagnosed instances of high-count MBL (cases with a cell count above 0.5x 109 cells/L, which can progress to CLL) or asymptomatic CLL, they explained.
“In contrast, next-generation sequencing results showed detectable skewing of the IGH gene repertoire in 21/28 patients up to 15 years before CLL diagnosis, often in the absence of elevated lymphocyte counts,” they wrote. “Remarkably, some patients with CLL requiring treatment and clinical transformation to an aggressive B-cell lymphoma displayed considerable skewing in the IGH gene repertoire even 16 years before CLL diagnosis.”
Patients with a prediagnostic IGHV-unmutated dominant clonotype had significantly shorter overall survival after CLL diagnosis than did those with an IGHV-mutated clonotype, they noted.
“Furthermore, at early timepoints (>10 years before diagnosis), patients with a high dominant clonotype frequency were more likely to be IGHV mutated, whereas closer to diagnosis this tendency was lost, indicating that the prediagnostic phase may be even longer than 16 years for [mutated] CLL patients,” they added.
The investigators also found that:
- Twenty-five patients carried stereotyped BcR IG up to 17 years prior to CLL diagnosis, and of these, 10 clonotypes were assigned to minor subsets and 15 to major CLL subsets. Among the latter, 14 of the 15 belonged to high-risk subsets, and most of those showed a trend for faster disease evolution.
- High frequency of the dominant clonotype was evident in samples obtained less than 6 years before diagnosis, whereas high-risk stereotyped clonotypes found longer before diagnosis (as early as 16 years) tended to have a lower dominant clonotype frequency (<20% of IGH gene repertoire)
- The stereotyped BcR IG matched the clonotype at diagnosis for both patients with diagnostic material.
- No stereotyped subsets were identified among the dominant clonotypes of the healthy controls.
“To our knowledge, the dynamics of the emergence of biclonality in an MBL patient and subsequent progression to CLL have never been captured in such a convincing manner,” they noted.
The findings “extend current knowledge on the evolution of the IGH repertoire prior to CLL diagnosis, highlighting that even high-risk CLL subtypes may display a prolonged indolent preclinical stage,” they added, speculating that “somatic genetic aberrations, (auto)stimulation, epigenetic and/or microenvironmental influences are required for the transformation into overt CLL.”
The investigators also noted that since the observed skewing in the IGH gene repertoire often occurs prior to B-cell lymphocytosis, they consider the findings “a novel extension to the characterization of MBL.”
“Further studies may prove invaluable in the clinical distinction between ‘progressing’ MBL versus ‘stable’ MBL. Notwithstanding the above, we emphasize that early detection is only warranted if it provides clear benefits to patient care,” they concluded.
In a related commentary, Gerald Marti, MD, PhD, of the National Heart, Lung, and Blood Institute, emphasized that the findings “represent the earliest detection of a clonotypic precursor cell for CLL.” .
They also raise new questions and point to new directions for research, Dr. Marti noted.
“Where do we go from here? CLL has a long evolutionary history in which early branching may start as an oligoclonal process (antigen stimulation) and include driver mutations,” he wrote. “A long-term analysis of the B-cell repertoire in familial CLL might shed light on this process. Further clarification of the mechanisms of age-related immune senescence is also of interest.”
The study authors and Dr. Marti reported having no competing financial interests.
Previous analyses showed that monoclonal B-cell lymphocytosis (MBL), a CLL precursor state, has been detected up to 6 years before CLL diagnosis, the investigators explained, noting that “[a]nother prognostically relevant immunogenetic feature of CLL concerns the stereotype of the B-cell receptor immunoglobulins (BcR IG).”
“Indeed, distinct stereotyped subsets can be defined by the expression of shared sequence motifs and are associated with particular presentation and outcomes,” P. Martijn Kolijn, PhD, a researcher in the department of immunology at Erasmus Medical Center, Rotterdam, the Netherlands, and colleagues wrote in a brief report published online in Blood. In an effort to “gain insight into the composition of the BcR IG repertoire during the early stages of CLL,” the investigators utilized next-generation sequencing to analyze 124 blood samples taken from healthy individuals up to 22 years before they received a diagnosis of CLL or small lymphocytic leukemia (SLL). An additional 118 matched control samples were also analyzed.
Study subjects were participants in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
“First, unsurprisingly, we observed a significant difference in the frequency of the dominant clonotype in CLL patients versus controls with a median frequency of 54.9%, compared to only 0.38% in controls,” they wrote.
Among 28 patients whose lymphocyte counts were measured at baseline, 10 showed evidence of lymphocytosis up to 8 years before CLL diagnosis.
This suggests undiagnosed instances of high-count MBL (cases with a cell count above 0.5x 109 cells/L, which can progress to CLL) or asymptomatic CLL, they explained.
“In contrast, next-generation sequencing results showed detectable skewing of the IGH gene repertoire in 21/28 patients up to 15 years before CLL diagnosis, often in the absence of elevated lymphocyte counts,” they wrote. “Remarkably, some patients with CLL requiring treatment and clinical transformation to an aggressive B-cell lymphoma displayed considerable skewing in the IGH gene repertoire even 16 years before CLL diagnosis.”
Patients with a prediagnostic IGHV-unmutated dominant clonotype had significantly shorter overall survival after CLL diagnosis than did those with an IGHV-mutated clonotype, they noted.
“Furthermore, at early timepoints (>10 years before diagnosis), patients with a high dominant clonotype frequency were more likely to be IGHV mutated, whereas closer to diagnosis this tendency was lost, indicating that the prediagnostic phase may be even longer than 16 years for [mutated] CLL patients,” they added.
The investigators also found that:
- Twenty-five patients carried stereotyped BcR IG up to 17 years prior to CLL diagnosis, and of these, 10 clonotypes were assigned to minor subsets and 15 to major CLL subsets. Among the latter, 14 of the 15 belonged to high-risk subsets, and most of those showed a trend for faster disease evolution.
- High frequency of the dominant clonotype was evident in samples obtained less than 6 years before diagnosis, whereas high-risk stereotyped clonotypes found longer before diagnosis (as early as 16 years) tended to have a lower dominant clonotype frequency (<20% of IGH gene repertoire)
- The stereotyped BcR IG matched the clonotype at diagnosis for both patients with diagnostic material.
- No stereotyped subsets were identified among the dominant clonotypes of the healthy controls.
“To our knowledge, the dynamics of the emergence of biclonality in an MBL patient and subsequent progression to CLL have never been captured in such a convincing manner,” they noted.
The findings “extend current knowledge on the evolution of the IGH repertoire prior to CLL diagnosis, highlighting that even high-risk CLL subtypes may display a prolonged indolent preclinical stage,” they added, speculating that “somatic genetic aberrations, (auto)stimulation, epigenetic and/or microenvironmental influences are required for the transformation into overt CLL.”
The investigators also noted that since the observed skewing in the IGH gene repertoire often occurs prior to B-cell lymphocytosis, they consider the findings “a novel extension to the characterization of MBL.”
“Further studies may prove invaluable in the clinical distinction between ‘progressing’ MBL versus ‘stable’ MBL. Notwithstanding the above, we emphasize that early detection is only warranted if it provides clear benefits to patient care,” they concluded.
In a related commentary, Gerald Marti, MD, PhD, of the National Heart, Lung, and Blood Institute, emphasized that the findings “represent the earliest detection of a clonotypic precursor cell for CLL.” .
They also raise new questions and point to new directions for research, Dr. Marti noted.
“Where do we go from here? CLL has a long evolutionary history in which early branching may start as an oligoclonal process (antigen stimulation) and include driver mutations,” he wrote. “A long-term analysis of the B-cell repertoire in familial CLL might shed light on this process. Further clarification of the mechanisms of age-related immune senescence is also of interest.”
The study authors and Dr. Marti reported having no competing financial interests.
Previous analyses showed that monoclonal B-cell lymphocytosis (MBL), a CLL precursor state, has been detected up to 6 years before CLL diagnosis, the investigators explained, noting that “[a]nother prognostically relevant immunogenetic feature of CLL concerns the stereotype of the B-cell receptor immunoglobulins (BcR IG).”
“Indeed, distinct stereotyped subsets can be defined by the expression of shared sequence motifs and are associated with particular presentation and outcomes,” P. Martijn Kolijn, PhD, a researcher in the department of immunology at Erasmus Medical Center, Rotterdam, the Netherlands, and colleagues wrote in a brief report published online in Blood. In an effort to “gain insight into the composition of the BcR IG repertoire during the early stages of CLL,” the investigators utilized next-generation sequencing to analyze 124 blood samples taken from healthy individuals up to 22 years before they received a diagnosis of CLL or small lymphocytic leukemia (SLL). An additional 118 matched control samples were also analyzed.
Study subjects were participants in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort.
“First, unsurprisingly, we observed a significant difference in the frequency of the dominant clonotype in CLL patients versus controls with a median frequency of 54.9%, compared to only 0.38% in controls,” they wrote.
Among 28 patients whose lymphocyte counts were measured at baseline, 10 showed evidence of lymphocytosis up to 8 years before CLL diagnosis.
This suggests undiagnosed instances of high-count MBL (cases with a cell count above 0.5x 109 cells/L, which can progress to CLL) or asymptomatic CLL, they explained.
“In contrast, next-generation sequencing results showed detectable skewing of the IGH gene repertoire in 21/28 patients up to 15 years before CLL diagnosis, often in the absence of elevated lymphocyte counts,” they wrote. “Remarkably, some patients with CLL requiring treatment and clinical transformation to an aggressive B-cell lymphoma displayed considerable skewing in the IGH gene repertoire even 16 years before CLL diagnosis.”
Patients with a prediagnostic IGHV-unmutated dominant clonotype had significantly shorter overall survival after CLL diagnosis than did those with an IGHV-mutated clonotype, they noted.
“Furthermore, at early timepoints (>10 years before diagnosis), patients with a high dominant clonotype frequency were more likely to be IGHV mutated, whereas closer to diagnosis this tendency was lost, indicating that the prediagnostic phase may be even longer than 16 years for [mutated] CLL patients,” they added.
The investigators also found that:
- Twenty-five patients carried stereotyped BcR IG up to 17 years prior to CLL diagnosis, and of these, 10 clonotypes were assigned to minor subsets and 15 to major CLL subsets. Among the latter, 14 of the 15 belonged to high-risk subsets, and most of those showed a trend for faster disease evolution.
- High frequency of the dominant clonotype was evident in samples obtained less than 6 years before diagnosis, whereas high-risk stereotyped clonotypes found longer before diagnosis (as early as 16 years) tended to have a lower dominant clonotype frequency (<20% of IGH gene repertoire)
- The stereotyped BcR IG matched the clonotype at diagnosis for both patients with diagnostic material.
- No stereotyped subsets were identified among the dominant clonotypes of the healthy controls.
“To our knowledge, the dynamics of the emergence of biclonality in an MBL patient and subsequent progression to CLL have never been captured in such a convincing manner,” they noted.
The findings “extend current knowledge on the evolution of the IGH repertoire prior to CLL diagnosis, highlighting that even high-risk CLL subtypes may display a prolonged indolent preclinical stage,” they added, speculating that “somatic genetic aberrations, (auto)stimulation, epigenetic and/or microenvironmental influences are required for the transformation into overt CLL.”
The investigators also noted that since the observed skewing in the IGH gene repertoire often occurs prior to B-cell lymphocytosis, they consider the findings “a novel extension to the characterization of MBL.”
“Further studies may prove invaluable in the clinical distinction between ‘progressing’ MBL versus ‘stable’ MBL. Notwithstanding the above, we emphasize that early detection is only warranted if it provides clear benefits to patient care,” they concluded.
In a related commentary, Gerald Marti, MD, PhD, of the National Heart, Lung, and Blood Institute, emphasized that the findings “represent the earliest detection of a clonotypic precursor cell for CLL.” .
They also raise new questions and point to new directions for research, Dr. Marti noted.
“Where do we go from here? CLL has a long evolutionary history in which early branching may start as an oligoclonal process (antigen stimulation) and include driver mutations,” he wrote. “A long-term analysis of the B-cell repertoire in familial CLL might shed light on this process. Further clarification of the mechanisms of age-related immune senescence is also of interest.”
The study authors and Dr. Marti reported having no competing financial interests.
FROM BLOOD
Cancer Data Trends 2022
Federal Practitioner, in collaboration with the Association of VA Hematology/Oncology (AVAHO), present the 2022 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- Exposure-Related Cancers
- Cancer in Women
- Genitourinary Cancers
- Gastrointestinal Cancers
- Telehealth in Oncology
- Precision Oncology
- Palliative and Hospice Care
- Alcohol and Cancer
- Lung Cancer
- Oropharyngeal Cancer
- Hematologic Cancers
Federal Practitioner and AVAHO would like to thank the following experts for their contributions to this issue:
Anita Aggarwal, DO, PhD; Sara Ahmed, PhD; Katherine Faricy-Anderson, MD; Apar Kishor Ganti, MD, MS; Solomon A Graf, MD; Kate Hendricks Thomas, PhD; Michael Kelley, MD; Mark Klein, MD, Gina McWhirter, MSN, MBA, RN; Bruce Montgomery, MD; Vida Almario Passero, MD, MBA; Thomas D Rodgers, MD; Vlad C Sandulache, MD, PhD; David H Wang, MD, PhD.
Federal Practitioner, in collaboration with the Association of VA Hematology/Oncology (AVAHO), present the 2022 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- Exposure-Related Cancers
- Cancer in Women
- Genitourinary Cancers
- Gastrointestinal Cancers
- Telehealth in Oncology
- Precision Oncology
- Palliative and Hospice Care
- Alcohol and Cancer
- Lung Cancer
- Oropharyngeal Cancer
- Hematologic Cancers
Federal Practitioner and AVAHO would like to thank the following experts for their contributions to this issue:
Anita Aggarwal, DO, PhD; Sara Ahmed, PhD; Katherine Faricy-Anderson, MD; Apar Kishor Ganti, MD, MS; Solomon A Graf, MD; Kate Hendricks Thomas, PhD; Michael Kelley, MD; Mark Klein, MD, Gina McWhirter, MSN, MBA, RN; Bruce Montgomery, MD; Vida Almario Passero, MD, MBA; Thomas D Rodgers, MD; Vlad C Sandulache, MD, PhD; David H Wang, MD, PhD.
Federal Practitioner, in collaboration with the Association of VA Hematology/Oncology (AVAHO), present the 2022 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- Exposure-Related Cancers
- Cancer in Women
- Genitourinary Cancers
- Gastrointestinal Cancers
- Telehealth in Oncology
- Precision Oncology
- Palliative and Hospice Care
- Alcohol and Cancer
- Lung Cancer
- Oropharyngeal Cancer
- Hematologic Cancers
Federal Practitioner and AVAHO would like to thank the following experts for their contributions to this issue:
Anita Aggarwal, DO, PhD; Sara Ahmed, PhD; Katherine Faricy-Anderson, MD; Apar Kishor Ganti, MD, MS; Solomon A Graf, MD; Kate Hendricks Thomas, PhD; Michael Kelley, MD; Mark Klein, MD, Gina McWhirter, MSN, MBA, RN; Bruce Montgomery, MD; Vida Almario Passero, MD, MBA; Thomas D Rodgers, MD; Vlad C Sandulache, MD, PhD; David H Wang, MD, PhD.
First-line CAR T-cell therapy could help cure some lymphomas
Results of the phase 2 ZUMA-12 trial suggest that axicabtagene ciloleucel (axi-cel), a chimeric antigen receptor (CAR) T-cell therapy approved to treat certain types of lymphoma, also shows promise as a treatment for another group of lymphoma patients – those with high-risk large B-cell lymphoma (LBCL) who failed two rounds of standard chemoimmunotherapy. In fact, a study author said, first-line treatment with this therapy could help usher some patients toward a cure.
The results appeared March 21, 2022, in Nature Medicine.
“The high efficacy with manageable safety profile suggest that further evaluation of axi-cel in first-line setting in patients with high-risk LBCL is warranted in a randomized, phase 3 trial comparing it to standard chemoimmunotherapy,” study lead author Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center, Houston, said in an interview.
According to Dr. Neelapu, “patients with high-risk LBCL include those with high-intermediate or high International Prognostic Index score and those with certain molecular subtypes such as double- or triple-hit lymphoma. These patients have lower response rates and lower progression-free and overall survival with standard chemoimmunotherapy.”
Treatment of these patients can be especially challenging because they are underrepresented in clinical research, hematologist Michael Dickinson, MBBS, of the Peter MacCallum Cancer Center in Melbourne, said in an interview. “They often have disease that requires urgent treatment, so there is no time to recruit them into trials. A feature of ZUMA-12 is that it allowed patients to be recruited after short exposure to chemotherapy, which means that higher-risk patients could successfully be recruited into the trial.”
Axi-cel is already Food and Drug Administration approved for treatment of relapsed or refractory LBCL after 2 or more lines of systemic therapy plus relapsed or refractory follicular lymphoma, also after two or more lines of systemic therapy, Dr. Neelapu said.
For this study, researchers administered the treatment to 40 subjects with high-risk disease from 2019-2020 (median age, 61 years; 68% male; 95% at disease stage III or IV).
The researchers reported that 78% of 37 patients in the primary efficacy analysis reached complete response rate (95% confidence interval, 62-90); the median time to first complete response rate was 30 days (range, 27-207). About 89% of these subjects reached the secondary endpoint of objective response rate (95% CI, 75-97); the median time to first objective response was 29 days (range, 27-207).
At a median follow-up of 15.9 months, 73% were still in objective response.
“This is quite remarkable,” Dr. Neelapu said. “The durability of more than 70% is far higher than what would be expected with standard chemoimmunotherapy in these patients – under 40% durability with standard chemoimmunotherapy. Also, axi-cel induces durable responses in about 40% of patients in second- and third-line setting. However, when used as part of first-line therapy in this study, durable responses were observed in more than 70% of patients, suggesting that the efficacy of axi-cel may be much higher when used in first-line setting.”
Dr. Neelapu added: “Although the follow-up is short, it is highly likely that the majority of the patients with ongoing response beyond 1 year will likely be cured of their lymphoma.”
As for side effects, no treatment-related grade 5 events occurred, but 18 patients (45%) experienced serious adverse events. Grade 3 or higher cytokine release syndrome occurred in three patients (8%) and nine experienced neurologic events (23%).
“The majority of the higher-grade adverse events observed were due to cytopenias, which were expected due to the conditioning therapy,” Dr. Neelapu said. “Such cytopenias would also have been expected if these patients had received standard chemoimmunotherapy.”
Six patients (15%) died, 4 of progressive disease after going forward to other therapies.
As for cost, Dr. Neelapu said it should be similar to that of axi-cel as an FDA-approved third-line therapy. Axi-cel is highly expensive. Research has suggested that CAR T-cell therapy can boost costs beyond standard chemotherapy by $350,000-$490,000 with gains of 2-8 years of life (J Med Econ. Jan-Dec 2021;24[1]:458-68).
The study was funded by Kite. The authors reported various disclosures.
Results of the phase 2 ZUMA-12 trial suggest that axicabtagene ciloleucel (axi-cel), a chimeric antigen receptor (CAR) T-cell therapy approved to treat certain types of lymphoma, also shows promise as a treatment for another group of lymphoma patients – those with high-risk large B-cell lymphoma (LBCL) who failed two rounds of standard chemoimmunotherapy. In fact, a study author said, first-line treatment with this therapy could help usher some patients toward a cure.
The results appeared March 21, 2022, in Nature Medicine.
“The high efficacy with manageable safety profile suggest that further evaluation of axi-cel in first-line setting in patients with high-risk LBCL is warranted in a randomized, phase 3 trial comparing it to standard chemoimmunotherapy,” study lead author Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center, Houston, said in an interview.
According to Dr. Neelapu, “patients with high-risk LBCL include those with high-intermediate or high International Prognostic Index score and those with certain molecular subtypes such as double- or triple-hit lymphoma. These patients have lower response rates and lower progression-free and overall survival with standard chemoimmunotherapy.”
Treatment of these patients can be especially challenging because they are underrepresented in clinical research, hematologist Michael Dickinson, MBBS, of the Peter MacCallum Cancer Center in Melbourne, said in an interview. “They often have disease that requires urgent treatment, so there is no time to recruit them into trials. A feature of ZUMA-12 is that it allowed patients to be recruited after short exposure to chemotherapy, which means that higher-risk patients could successfully be recruited into the trial.”
Axi-cel is already Food and Drug Administration approved for treatment of relapsed or refractory LBCL after 2 or more lines of systemic therapy plus relapsed or refractory follicular lymphoma, also after two or more lines of systemic therapy, Dr. Neelapu said.
For this study, researchers administered the treatment to 40 subjects with high-risk disease from 2019-2020 (median age, 61 years; 68% male; 95% at disease stage III or IV).
The researchers reported that 78% of 37 patients in the primary efficacy analysis reached complete response rate (95% confidence interval, 62-90); the median time to first complete response rate was 30 days (range, 27-207). About 89% of these subjects reached the secondary endpoint of objective response rate (95% CI, 75-97); the median time to first objective response was 29 days (range, 27-207).
At a median follow-up of 15.9 months, 73% were still in objective response.
“This is quite remarkable,” Dr. Neelapu said. “The durability of more than 70% is far higher than what would be expected with standard chemoimmunotherapy in these patients – under 40% durability with standard chemoimmunotherapy. Also, axi-cel induces durable responses in about 40% of patients in second- and third-line setting. However, when used as part of first-line therapy in this study, durable responses were observed in more than 70% of patients, suggesting that the efficacy of axi-cel may be much higher when used in first-line setting.”
Dr. Neelapu added: “Although the follow-up is short, it is highly likely that the majority of the patients with ongoing response beyond 1 year will likely be cured of their lymphoma.”
As for side effects, no treatment-related grade 5 events occurred, but 18 patients (45%) experienced serious adverse events. Grade 3 or higher cytokine release syndrome occurred in three patients (8%) and nine experienced neurologic events (23%).
“The majority of the higher-grade adverse events observed were due to cytopenias, which were expected due to the conditioning therapy,” Dr. Neelapu said. “Such cytopenias would also have been expected if these patients had received standard chemoimmunotherapy.”
Six patients (15%) died, 4 of progressive disease after going forward to other therapies.
As for cost, Dr. Neelapu said it should be similar to that of axi-cel as an FDA-approved third-line therapy. Axi-cel is highly expensive. Research has suggested that CAR T-cell therapy can boost costs beyond standard chemotherapy by $350,000-$490,000 with gains of 2-8 years of life (J Med Econ. Jan-Dec 2021;24[1]:458-68).
The study was funded by Kite. The authors reported various disclosures.
Results of the phase 2 ZUMA-12 trial suggest that axicabtagene ciloleucel (axi-cel), a chimeric antigen receptor (CAR) T-cell therapy approved to treat certain types of lymphoma, also shows promise as a treatment for another group of lymphoma patients – those with high-risk large B-cell lymphoma (LBCL) who failed two rounds of standard chemoimmunotherapy. In fact, a study author said, first-line treatment with this therapy could help usher some patients toward a cure.
The results appeared March 21, 2022, in Nature Medicine.
“The high efficacy with manageable safety profile suggest that further evaluation of axi-cel in first-line setting in patients with high-risk LBCL is warranted in a randomized, phase 3 trial comparing it to standard chemoimmunotherapy,” study lead author Sattva S. Neelapu, MD, of the University of Texas MD Anderson Cancer Center, Houston, said in an interview.
According to Dr. Neelapu, “patients with high-risk LBCL include those with high-intermediate or high International Prognostic Index score and those with certain molecular subtypes such as double- or triple-hit lymphoma. These patients have lower response rates and lower progression-free and overall survival with standard chemoimmunotherapy.”
Treatment of these patients can be especially challenging because they are underrepresented in clinical research, hematologist Michael Dickinson, MBBS, of the Peter MacCallum Cancer Center in Melbourne, said in an interview. “They often have disease that requires urgent treatment, so there is no time to recruit them into trials. A feature of ZUMA-12 is that it allowed patients to be recruited after short exposure to chemotherapy, which means that higher-risk patients could successfully be recruited into the trial.”
Axi-cel is already Food and Drug Administration approved for treatment of relapsed or refractory LBCL after 2 or more lines of systemic therapy plus relapsed or refractory follicular lymphoma, also after two or more lines of systemic therapy, Dr. Neelapu said.
For this study, researchers administered the treatment to 40 subjects with high-risk disease from 2019-2020 (median age, 61 years; 68% male; 95% at disease stage III or IV).
The researchers reported that 78% of 37 patients in the primary efficacy analysis reached complete response rate (95% confidence interval, 62-90); the median time to first complete response rate was 30 days (range, 27-207). About 89% of these subjects reached the secondary endpoint of objective response rate (95% CI, 75-97); the median time to first objective response was 29 days (range, 27-207).
At a median follow-up of 15.9 months, 73% were still in objective response.
“This is quite remarkable,” Dr. Neelapu said. “The durability of more than 70% is far higher than what would be expected with standard chemoimmunotherapy in these patients – under 40% durability with standard chemoimmunotherapy. Also, axi-cel induces durable responses in about 40% of patients in second- and third-line setting. However, when used as part of first-line therapy in this study, durable responses were observed in more than 70% of patients, suggesting that the efficacy of axi-cel may be much higher when used in first-line setting.”
Dr. Neelapu added: “Although the follow-up is short, it is highly likely that the majority of the patients with ongoing response beyond 1 year will likely be cured of their lymphoma.”
As for side effects, no treatment-related grade 5 events occurred, but 18 patients (45%) experienced serious adverse events. Grade 3 or higher cytokine release syndrome occurred in three patients (8%) and nine experienced neurologic events (23%).
“The majority of the higher-grade adverse events observed were due to cytopenias, which were expected due to the conditioning therapy,” Dr. Neelapu said. “Such cytopenias would also have been expected if these patients had received standard chemoimmunotherapy.”
Six patients (15%) died, 4 of progressive disease after going forward to other therapies.
As for cost, Dr. Neelapu said it should be similar to that of axi-cel as an FDA-approved third-line therapy. Axi-cel is highly expensive. Research has suggested that CAR T-cell therapy can boost costs beyond standard chemotherapy by $350,000-$490,000 with gains of 2-8 years of life (J Med Econ. Jan-Dec 2021;24[1]:458-68).
The study was funded by Kite. The authors reported various disclosures.
FROM NATURE MEDICINE
Eating olive oil may slow CLL disease progression
Olive oil is a major component of the Mediterranean diet, and olive phenols have been shown to convey antioxidant, anti-inflammatory, anticancer, neuroprotective, and antidiabetic effects by modulating various molecular pathways, Andrea Paola Rojas Gil, PhD, of the University of Peloponnese, Tripoli, Greece, and colleagues wrote.
In most patients, CLL is incurable, but those at the early stages do not need immediate therapy and may benefit from an intervention to prevent disease progression, the authors wrote. Previous research suggested that dietary intervention exerts a salutary effect on early CLL, and in vitro studies suggested that oleocanthal, a component of extra virgin olive oil, induced anticancer activity.
In a study published in Frontiers in Oncology, the researchers enrolled adults with early stage CLL who had not undergone chemotherapy or other treatment. All patients adhered to a Mediterranean-style diet.
After a washout period of 9-12 months, the researchers randomized 22 patients to extra virgin olive oil high in oleocanthal and oleacein (high OC/OL-EVOO). Patients in the intervention group consumed 40 mL/day of high OC/OL-EVOO before meals. Their average age was 71 years; 10 were women and 12 were men.
The primary outcomes included changes in hematological, biochemical, and apoptotic markers. After 6 months, patients in the intervention group showed a statistically significant reduction in white blood cells and lymphocyte count, compared with measurements taken 3 months before the intervention. The WBC decrease was greatest among patients with the highest WBC levels at baseline.
As for biochemical markers, the researchers observed a significant decrease in glucose levels during the intervention, but no significant effects on metabolic indexes or renal function.
After 3 months and also after 6 months of the olive oil intervention, patients showed a significant increase in the apoptotic markers ccK18 and Apo1-Fas (P ≤ .05 for both), as well as an increase in the cell cycle negative regulator p21. The dietary intervention also was associated with significant decreases in expression of the antiapoptotic protein survivin and in cyclin D, a positive cell cycle regulator protein.
Further, patients who had a high ccK18 level at baseline showed a significantly greater increase in ccK18 after the intervention, compared with those with lower ccK18 at baseline (P = .001).
Notably, “a negative correlation of the WBC at the end of the dietary intervention with the fluctuation of the protein expression of the apoptotic marker ccK18 (final – initial) was observed,” the researchers wrote in their discussion.
The study findings were limited by several factors including the small sample size, short intervention time, and pilot design, the researchers said. Other limitations include the possible effect of other unmeasured properties of olive oil.
However, the results reflect previous studies showing the benefits of a Mediterranean-type diet, and they represent the first clinical trial to indicate possible beneficial effects from oleocanthal and oleacein on the progression of CLL. Therefore, the authors concluded, the study is worthy of a large, multicenter trial.
Pilot data merit more research
In an interview, corresponding author Prokopios Magiatis, PhD, noted that CLL is “the most commonly diagnosed adult leukemia in Western countries and is responsible for about one in four cases of all leukemias.” CLL remains incurable in most patients, and ways to delay disease progression are needed.
“Oleocanthal is the active ingredient of early harvest olive oil with proven anticancer activities in vitro and in vivo,” Dr. Magiatis explained. “For this reason, it was a unique challenge to investigate the anticancer activity of this compound for the first time in humans through the dietary consumption of specifically selected olive oil.” He expressed surprise at the beneficial effects of high-oleocanthal olive oil, not only to the white blood cells, but also to glucose levels.
“It seems that oleocanthal can activate mechanisms related to the apoptosis of cancer cells, and also mechanisms related to blood glucose regulation without affecting any normal cells of the body,” he said. “All anticancer drugs usually have severe side effects, however the administration of 25 mg of oleocanthal through the dietary consumption of olive oil did not present any harmful effects for at least 6 months of everyday use.
“The addition of naturally produced high-oleocanthal olive oil in the diet of early-stage CLL patients at a dose of three tablespoons per day [40 mL] is a practice that may lower the cancerous white blood cells of the patients without any risk,” said Dr. Magiatis. “High-oleocanthal early-harvest olive oil has been consumed for centuries, and may be the key of longevity of several Mediterranean populations.
“In our study, the number of the white blood cells returned back to the number it was one year before the initiation of the study; this clearly shows that it could be a significant factor for the delay of the progress of the disease,” he said.
The current trial was a pilot study in one hospital with only 22 patients for 6 months, said Dr. Magiatis. “We are currently preparing the expansion of the study to other hospitals and other countries, and we aim to include at least 100 patients for at least 1 year, to validate the already-obtained beneficial results.”
The clinical trial is supported by the nonprofit organization World Olive Center for Health, he added.
The current study received no outside funding. The researchers had no financial conflicts to disclose.
Olive oil is a major component of the Mediterranean diet, and olive phenols have been shown to convey antioxidant, anti-inflammatory, anticancer, neuroprotective, and antidiabetic effects by modulating various molecular pathways, Andrea Paola Rojas Gil, PhD, of the University of Peloponnese, Tripoli, Greece, and colleagues wrote.
In most patients, CLL is incurable, but those at the early stages do not need immediate therapy and may benefit from an intervention to prevent disease progression, the authors wrote. Previous research suggested that dietary intervention exerts a salutary effect on early CLL, and in vitro studies suggested that oleocanthal, a component of extra virgin olive oil, induced anticancer activity.
In a study published in Frontiers in Oncology, the researchers enrolled adults with early stage CLL who had not undergone chemotherapy or other treatment. All patients adhered to a Mediterranean-style diet.
After a washout period of 9-12 months, the researchers randomized 22 patients to extra virgin olive oil high in oleocanthal and oleacein (high OC/OL-EVOO). Patients in the intervention group consumed 40 mL/day of high OC/OL-EVOO before meals. Their average age was 71 years; 10 were women and 12 were men.
The primary outcomes included changes in hematological, biochemical, and apoptotic markers. After 6 months, patients in the intervention group showed a statistically significant reduction in white blood cells and lymphocyte count, compared with measurements taken 3 months before the intervention. The WBC decrease was greatest among patients with the highest WBC levels at baseline.
As for biochemical markers, the researchers observed a significant decrease in glucose levels during the intervention, but no significant effects on metabolic indexes or renal function.
After 3 months and also after 6 months of the olive oil intervention, patients showed a significant increase in the apoptotic markers ccK18 and Apo1-Fas (P ≤ .05 for both), as well as an increase in the cell cycle negative regulator p21. The dietary intervention also was associated with significant decreases in expression of the antiapoptotic protein survivin and in cyclin D, a positive cell cycle regulator protein.
Further, patients who had a high ccK18 level at baseline showed a significantly greater increase in ccK18 after the intervention, compared with those with lower ccK18 at baseline (P = .001).
Notably, “a negative correlation of the WBC at the end of the dietary intervention with the fluctuation of the protein expression of the apoptotic marker ccK18 (final – initial) was observed,” the researchers wrote in their discussion.
The study findings were limited by several factors including the small sample size, short intervention time, and pilot design, the researchers said. Other limitations include the possible effect of other unmeasured properties of olive oil.
However, the results reflect previous studies showing the benefits of a Mediterranean-type diet, and they represent the first clinical trial to indicate possible beneficial effects from oleocanthal and oleacein on the progression of CLL. Therefore, the authors concluded, the study is worthy of a large, multicenter trial.
Pilot data merit more research
In an interview, corresponding author Prokopios Magiatis, PhD, noted that CLL is “the most commonly diagnosed adult leukemia in Western countries and is responsible for about one in four cases of all leukemias.” CLL remains incurable in most patients, and ways to delay disease progression are needed.
“Oleocanthal is the active ingredient of early harvest olive oil with proven anticancer activities in vitro and in vivo,” Dr. Magiatis explained. “For this reason, it was a unique challenge to investigate the anticancer activity of this compound for the first time in humans through the dietary consumption of specifically selected olive oil.” He expressed surprise at the beneficial effects of high-oleocanthal olive oil, not only to the white blood cells, but also to glucose levels.
“It seems that oleocanthal can activate mechanisms related to the apoptosis of cancer cells, and also mechanisms related to blood glucose regulation without affecting any normal cells of the body,” he said. “All anticancer drugs usually have severe side effects, however the administration of 25 mg of oleocanthal through the dietary consumption of olive oil did not present any harmful effects for at least 6 months of everyday use.
“The addition of naturally produced high-oleocanthal olive oil in the diet of early-stage CLL patients at a dose of three tablespoons per day [40 mL] is a practice that may lower the cancerous white blood cells of the patients without any risk,” said Dr. Magiatis. “High-oleocanthal early-harvest olive oil has been consumed for centuries, and may be the key of longevity of several Mediterranean populations.
“In our study, the number of the white blood cells returned back to the number it was one year before the initiation of the study; this clearly shows that it could be a significant factor for the delay of the progress of the disease,” he said.
The current trial was a pilot study in one hospital with only 22 patients for 6 months, said Dr. Magiatis. “We are currently preparing the expansion of the study to other hospitals and other countries, and we aim to include at least 100 patients for at least 1 year, to validate the already-obtained beneficial results.”
The clinical trial is supported by the nonprofit organization World Olive Center for Health, he added.
The current study received no outside funding. The researchers had no financial conflicts to disclose.
Olive oil is a major component of the Mediterranean diet, and olive phenols have been shown to convey antioxidant, anti-inflammatory, anticancer, neuroprotective, and antidiabetic effects by modulating various molecular pathways, Andrea Paola Rojas Gil, PhD, of the University of Peloponnese, Tripoli, Greece, and colleagues wrote.
In most patients, CLL is incurable, but those at the early stages do not need immediate therapy and may benefit from an intervention to prevent disease progression, the authors wrote. Previous research suggested that dietary intervention exerts a salutary effect on early CLL, and in vitro studies suggested that oleocanthal, a component of extra virgin olive oil, induced anticancer activity.
In a study published in Frontiers in Oncology, the researchers enrolled adults with early stage CLL who had not undergone chemotherapy or other treatment. All patients adhered to a Mediterranean-style diet.
After a washout period of 9-12 months, the researchers randomized 22 patients to extra virgin olive oil high in oleocanthal and oleacein (high OC/OL-EVOO). Patients in the intervention group consumed 40 mL/day of high OC/OL-EVOO before meals. Their average age was 71 years; 10 were women and 12 were men.
The primary outcomes included changes in hematological, biochemical, and apoptotic markers. After 6 months, patients in the intervention group showed a statistically significant reduction in white blood cells and lymphocyte count, compared with measurements taken 3 months before the intervention. The WBC decrease was greatest among patients with the highest WBC levels at baseline.
As for biochemical markers, the researchers observed a significant decrease in glucose levels during the intervention, but no significant effects on metabolic indexes or renal function.
After 3 months and also after 6 months of the olive oil intervention, patients showed a significant increase in the apoptotic markers ccK18 and Apo1-Fas (P ≤ .05 for both), as well as an increase in the cell cycle negative regulator p21. The dietary intervention also was associated with significant decreases in expression of the antiapoptotic protein survivin and in cyclin D, a positive cell cycle regulator protein.
Further, patients who had a high ccK18 level at baseline showed a significantly greater increase in ccK18 after the intervention, compared with those with lower ccK18 at baseline (P = .001).
Notably, “a negative correlation of the WBC at the end of the dietary intervention with the fluctuation of the protein expression of the apoptotic marker ccK18 (final – initial) was observed,” the researchers wrote in their discussion.
The study findings were limited by several factors including the small sample size, short intervention time, and pilot design, the researchers said. Other limitations include the possible effect of other unmeasured properties of olive oil.
However, the results reflect previous studies showing the benefits of a Mediterranean-type diet, and they represent the first clinical trial to indicate possible beneficial effects from oleocanthal and oleacein on the progression of CLL. Therefore, the authors concluded, the study is worthy of a large, multicenter trial.
Pilot data merit more research
In an interview, corresponding author Prokopios Magiatis, PhD, noted that CLL is “the most commonly diagnosed adult leukemia in Western countries and is responsible for about one in four cases of all leukemias.” CLL remains incurable in most patients, and ways to delay disease progression are needed.
“Oleocanthal is the active ingredient of early harvest olive oil with proven anticancer activities in vitro and in vivo,” Dr. Magiatis explained. “For this reason, it was a unique challenge to investigate the anticancer activity of this compound for the first time in humans through the dietary consumption of specifically selected olive oil.” He expressed surprise at the beneficial effects of high-oleocanthal olive oil, not only to the white blood cells, but also to glucose levels.
“It seems that oleocanthal can activate mechanisms related to the apoptosis of cancer cells, and also mechanisms related to blood glucose regulation without affecting any normal cells of the body,” he said. “All anticancer drugs usually have severe side effects, however the administration of 25 mg of oleocanthal through the dietary consumption of olive oil did not present any harmful effects for at least 6 months of everyday use.
“The addition of naturally produced high-oleocanthal olive oil in the diet of early-stage CLL patients at a dose of three tablespoons per day [40 mL] is a practice that may lower the cancerous white blood cells of the patients without any risk,” said Dr. Magiatis. “High-oleocanthal early-harvest olive oil has been consumed for centuries, and may be the key of longevity of several Mediterranean populations.
“In our study, the number of the white blood cells returned back to the number it was one year before the initiation of the study; this clearly shows that it could be a significant factor for the delay of the progress of the disease,” he said.
The current trial was a pilot study in one hospital with only 22 patients for 6 months, said Dr. Magiatis. “We are currently preparing the expansion of the study to other hospitals and other countries, and we aim to include at least 100 patients for at least 1 year, to validate the already-obtained beneficial results.”
The clinical trial is supported by the nonprofit organization World Olive Center for Health, he added.
The current study received no outside funding. The researchers had no financial conflicts to disclose.
FROM FRONTIERS IN ONCOLOGY
A Practical and Cost-Effective Approach to the Diagnosis of Heparin-Induced Thrombocytopenia: A Single-Center Quality Improvement Study
From the Veterans Affairs Ann Arbor Healthcare System Medicine Service (Dr. Cusick), University of Michigan College of Pharmacy, Clinical Pharmacy Service, Michigan Medicine (Dr. Hanigan), Department of Internal Medicine Clinical Experience and Quality, Michigan Medicine (Linda Bashaw), Department of Internal Medicine, University of Michigan Medical School, Ann Arbor, MI (Dr. Heidemann), and the Operational Excellence Department, Sparrow Health System, Lansing, MI (Matthew Johnson).
Abstract
Background: Diagnosis of heparin-induced thrombocytopenia (HIT) requires completion of an enzyme-linked immunosorbent assay (ELISA)–based heparin-platelet factor 4 (PF4) antibody test. If this test is negative, HIT is excluded. If positive, a serotonin-release assay (SRA) test is indicated. The SRA is expensive and sometimes inappropriately ordered despite negative PF4 results, leading to unnecessary treatment with argatroban while awaiting SRA results.
Objectives: The primary objectives of this project were to reduce unnecessary SRA testing and argatroban utilization in patients with suspected HIT.
Methods: The authors implemented an intervention at a tertiary care academic hospital in November 2017 targeting patients hospitalized with suspected HIT. The intervention was controlled at the level of the laboratory and prevented ordering of SRA tests in the absence of a positive PF4 test. The number of SRA tests performed and argatroban bags administered were identified retrospectively via chart review before the intervention (January 2016 to November 2017) and post intervention (December 2017 to March 2020). Associated costs were calculated based on institutional SRA testing cost as well as the average wholesale price of argatroban.
Results: SRA testing decreased from an average of 3.7 SRA results per 1000 admissions before the intervention to an average of 0.6 results per 1000 admissions post intervention. The number of 50-mL argatroban bags used per 1000 admissions decreased from 18.8 prior to the intervention to 14.3 post intervention. Total estimated cost savings per 1000 admissions was $2361.20.
Conclusion: An evidence-based testing strategy for HIT can be effectively implemented at the level of the laboratory. This approach led to reductions in SRA testing and argatroban utilization with resultant cost savings.
Keywords: HIT, argatroban, anticoagulation, serotonin-release assay.
Thrombocytopenia is a common finding in hospitalized patients.1,2 Heparin-induced thrombocytopenia (HIT) is one of the many potential causes of thrombocytopenia in hospitalized patients and occurs when antibodies to the heparin-platelet factor 4 (PF4) complex develop after heparin exposure. This triggers a cascade of events, leading to platelet activation, platelet consumption, and thrombosis. While HIT is relatively rare, occurring in 0.3% to 0.5% of critically ill patients, many patients will be tested to rule out this potentially life-threatening cause of thrombocytopenia.3
The diagnosis of HIT utilizes a combination of both clinical suspicion and laboratory testing.4 The 4T score (Table) was developed to evaluate the clinical probability of HIT and involves assessing the degree and timing of thrombocytopenia, the presence or absence of thrombosis, and other potential causes of the thrombocytopenia.5 The 4T score is designed to be utilized to identify patients who require laboratory testing for HIT; however, it has low inter-rater agreement in patients undergoing evaluation for HIT,6 and, in our experience, completion of this scoring is time-consuming.
The enzyme-linked immunosorbent assay (ELISA) is a commonly used laboratory test to diagnose HIT that detects antibodies to the heparin-PF4 complex utilizing optical density (OD) units. When using an OD cutoff of 0.400, ELISA PF4 (PF4) tests have a sensitivity of 99.6%, but poor specificity at 69.3%.7 When the PF4 antibody test is positive with an OD ≥0.400, then a functional test is used to determine whether the antibodies detected will activate platelets. The serotonin-release assay (SRA) is a functional test that measures 14C-labeled serotonin release from donor platelets when mixed with patient serum or plasma containing HIT antibodies. In the correct clinical context, a positive ELISA PF4 antibody test along with a positive SRA is diagnostic of HIT.8
The process of diagnosing HIT in a timely and cost-effective manner is dependent on the clinician’s experience in diagnosing HIT as well as access to the laboratory testing necessary to confirm the diagnosis. PF4 antibody tests are time-consuming and not always available daily and/or are not available onsite. The SRA requires access to donor platelets and specialized radioactivity counting equipment, making it available only at particular centers.
The treatment of HIT is more straightforward and involves stopping all heparin products and starting a nonheparin anticoagulant. The direct thrombin inhibitor argatroban is one of the standard nonheparin anticoagulants used in patients with suspected HIT.4 While it is expensive, its short half-life and lack of renal clearance make it ideal for treatment of hospitalized patients with suspected HIT, many of whom need frequent procedures and/or have renal disease.
At our academic tertiary care center, we performed a retrospective analysis that showed inappropriate ordering of diagnostic HIT testing as well as unnecessary use of argatroban even when there was low suspicion for HIT based on laboratory findings. The aim of our project was to reduce unnecessary HIT testing and argatroban utilization without overburdening providers or interfering with established workflows.
Methods
Setting
The University of Michigan (UM) hospital is a 1000-bed tertiary care center in Ann Arbor, Michigan. The UM guidelines reflect evidence-based guidelines for the diagnosis and treatment of HIT.4 In 2016 the UM guidelines for laboratory testing included sending the PF4 antibody test first when there was clinical suspicion of HIT. The SRA was to be sent separately only when the PF4 returned positive (OD ≥ 0.400). Standard guidelines at UM also included switching patients with suspected HIT from heparin to a nonheparin anticoagulant and stopping all heparin products while awaiting the SRA results. The direct thrombin inhibitor argatroban is utilized at UM and monitored with anti-IIa levels. University of Michigan Hospital utilizes the Immucor PF4 IgG ELISA for detecting heparin-associated antibodies.9 In 2016, this PF4 test was performed in the UM onsite laboratory Monday through Friday. At UM the SRA is performed off site, with a turnaround time of 3 to 5 business days.
Baseline Data
We retrospectively reviewed PF4 and SRA testing as well as argatroban usage from December 2016 to May 2017. Despite the institutional guidelines, providers were sending PF4 and SRA simultaneously as soon as HIT was suspected; 62% of PF4 tests were ordered simultaneously with the SRA, but only 8% of these PF4 tests were positive with an OD ≥0.400. Of those patients with negative PF4 testing, argatroban was continued until the SRA returned negative, leading to many days of unnecessary argatroban usage. An informal survey of the anticoagulation pharmacists revealed that many recommended discontinuing argatroban when the PF4 test was negative, but providers routinely did not feel comfortable with this approach. This suggested many providers misunderstood the performance characteristics of the PF4 test.
Intervention
Our team consisted of hematology and internal medicine faculty, pharmacists, coagulation laboratory personnel, and quality improvement specialists. We designed and implemented an intervention in November 2017 focused on controlling the ordering of the SRA test. We chose to focus on this step due to the excellent sensitivity of the PF4 test with a cutoff of OD <0.400 and the significant expense of the SRA test. Under direction of the Coagulation Laboratory Director, a standard operating procedure was developed where the coagulation laboratory personnel did not send out the SRA until a positive PF4 test (OD ≥ 0.400) was reported. If the PF4 was negative, the SRA was canceled and the ordering provider received notification of the cancelled test via the electronic medical record, accompanied by education about HIT testing (Figure 1). In addition, the lab increased the availability of PF4 testing from 5 days to 7 days a week so there were no delays in tests ordered on Fridays or weekends.
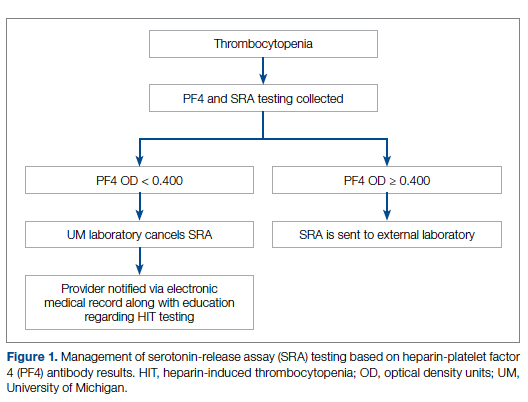
Outcomes
Our primary goals were to decrease both SRA testing and argatroban use. Secondarily, we examined the cost-effectiveness of this intervention. We hypothesized that controlling the SRA testing at the laboratory level would decrease both SRA testing and argatroban use.
Data Collection
Pre- and postintervention data were collected retrospectively. Pre-intervention data were from January 2016 through November 2017, and postintervention data were from December 2017 through March 2020. The number of SRA tests performed were identified retrospectively via review of electronic ordering records. All patients who had a hospital admission after January 1, 2016, were included. These patients were filtered to include only those who had a result for an SRA test. In order to calculate cost-savings, we identified both the number of SRA tests ordered retrospectively as well as patients who had both an SRA resulted and had been administered argatroban. Cost-savings were calculated based on our institutional cost of $357 per SRA test.
At our institution, argatroban is supplied in 50-mL bags; therefore, we utilized the number of bags to identify argatroban usage. Savings were calculated using the average wholesale price (AWP) of $292.50 per 50-mL bag. The amounts billed or collected for the SRA testing or argatroban treatment were not collected. Costs were estimated using only direct costs to the institution. Safety data were not collected. As the intent of our project was a quality improvement activity, this project did not require institutional review board regulation per our institutional guidance.
Results
During the pre-intervention period, the average number of admissions (adults and children) at UM was 5863 per month. Post intervention there was an average of 5842 admissions per month. A total of 1192 PF4 tests were ordered before the intervention and 1148 were ordered post intervention. Prior to the intervention, 481 SRA tests were completed, while post intervention 105 were completed. Serotonin-release testing decreased from an average of 3.7 SRA results per 1000 admissions during the pre-intervention period to an average of 0.6 per 1000 admissions post intervention (Figure 2). Cost-savings were $1045 per 1000 admissions.
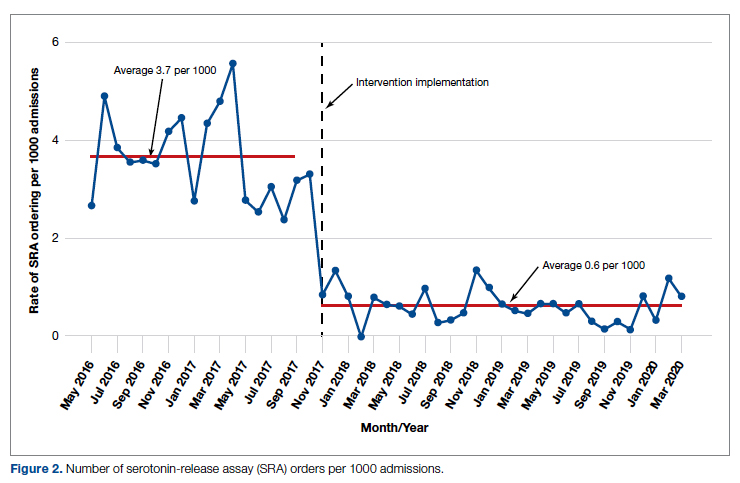
During the pre-intervention period, 2539 bags of argatroban were used, while 2337 bags were used post intervention. The number of 50-mL argatroban bags used per 1000 admissions decreased from 18.8 before the intervention to 14.3 post intervention. Cost-savings were $1316.20 per 1000 admissions. Figure 3 illustrates the monthly argatroban utilization per 1000 admissions during each quarter from January 2016 through March 2020.
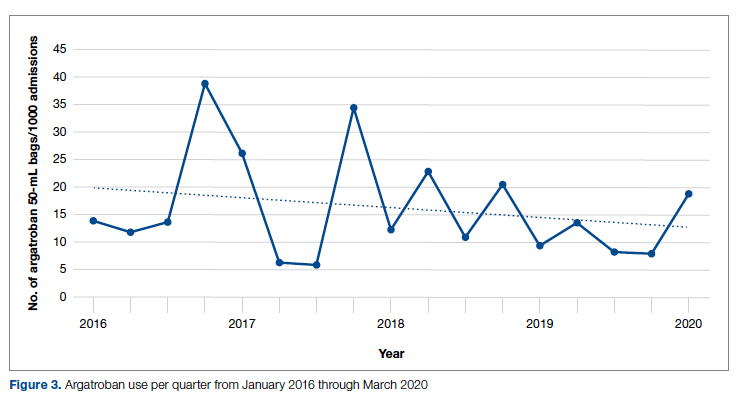
Discussion
We designed and implemented an evidence-based strategy for HIT at our academic institution which led to a decrease in unnecessary SRA testing and argatroban utilization, with associated cost savings. By focusing on a single point of intervention at the laboratory level where SRA tests were held and canceled if the PF4 test was negative, we helped offload the decision-making from the provider while simultaneously providing just-in-time education to the provider. This intervention was designed with input from multiple stakeholders, including physicians, quality improvement specialists, pharmacists, and coagulation laboratory personnel.
Serotonin-release testing dramatically decreased post intervention even though a similar number of PF4 tests were performed before and after the intervention. This suggests that the decrease in SRA testing was a direct consequence of our intervention. Post intervention the number of completed SRA tests was 9% of the number of PF4 tests sent. This is consistent with our baseline pre-intervention data showing that only 8% of all PF4 tests sent were positive.
While the absolute number of argatroban bags utilized did not dramatically decrease after the intervention, the quarterly rate did, particularly after 2018. Given that argatroban data were only drawn from patients with a concurrent SRA test, this decrease is clearly from decreased usage in patients with suspected HIT. We suspect the decrease occurred because argatroban was not being continued while awaiting an SRA test in patients with a negative PF4 test. Decreasing the utilization of argatroban not only saved money but also reduced days of exposure to argatroban. While we do not have data regarding adverse events related to argatroban prior to the intervention, it is logical to conclude that reducing unnecessary exposure to argatroban reduces the risk of adverse events related to bleeding. Future studies would ideally address specific safety outcome metrics such as adverse events, bleeding risk, or missed diagnoses of HIT.
Our institutional guidelines for the diagnosis of HIT are evidence-based and helpful but are rarely followed by busy inpatient providers. Controlling the utilization of the SRA at the laboratory level had several advantages. First, removing SRA decision-making from providers who are not experts in the diagnosis of HIT guaranteed adherence to evidence-based guidelines. Second, pharmacists could safely recommend discontinuing argatroban when the PF4 test was negative as there was no SRA pending. Third, with cancellation at the laboratory level there was no need to further burden providers with yet another alert in the electronic health record. Fourth, just-in-time education was provided to the providers with justification for why the SRA test was canceled. Last, ruling out HIT within 24 hours with the PF4 test alone allowed providers to evaluate patients for other causes of thrombocytopenia much earlier than the 3 to 5 business days before the SRA results returned.
A limitation of this study is that it was conducted at a single center. Our approach is also limited by the lack of universal applicability. At our institution we are fortunate to have PF4 testing available in our coagulation laboratory 7 days a week. In addition, the coagulation laboratory controls sending the SRA to the reference laboratory. The specific intervention of controlling the SRA testing is therefore applicable only to institutions similar to ours; however, the concept of removing control of specialized testing from the provider is not unique. Inpatient thrombophilia testing has been a successful target of this approach.11-13 While electronic alerts and education of individual providers can also be effective initially, the effectiveness of these interventions has been repeatedly shown to wane over time.14-16
Conclusion
At our institution we were able to implement practical, evidence-based testing for HIT by implementing control over SRA testing at the level of the laboratory. This approach led to decreased argatroban utilization and cost savings.
Corresponding author: Alice Cusick, MD; LTC Charles S Kettles VA Medical Center, 2215 Fuller Road, Ann Arbor, MI 48105; mccoyag@med.umich.edu
Disclosures: None reported.
doi: 10.12788/jcom.0087
1. Fountain E, Arepally GM. Thrombocytopenia in hospitalized non-ICU patients. Blood. 2015;126(23):1060. doi:10.1182/blood.v126.23.1060.1060
2. Hui P, Cook DJ, Lim W, Fraser GA, Arnold DM. The frequency and clinical significance of thrombocytopenia complicating critical illness: a systematic review. Chest. 2011;139(2):271-278. doi:10.1378/chest.10-2243
3. Warkentin TE. Heparin-induced thrombocytopenia. Curr Opin Crit Care. 2015;21(6):576-585. doi:10.1097/MCC.0000000000000259
4. Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv. 2018;2(22):3360-3392. doi:10.1182/bloodadvances.2018024489
5. Cuker A, Gimotty PA, Crowther MA, Warkentin TE. Predictive value of the 4Ts scoring system for heparin-induced thrombocytopenia: a systematic review and meta-analysis. Blood. 2012;120(20):4160-4167. doi:10.1182/blood-2012-07-443051
6. Northam KA, Parker WF, Chen S-L, et al. Evaluation of 4Ts score inter-rater agreement in patients undergoing evaluation for heparin-induced thrombocytopenia. Blood Coagul Fibrinolysis. 2021;32(5):328-334. doi:10.1097/MBC.0000000000001042
7. Raschke RA, Curry SC, Warkentin TE, Gerkin RD. Improving clinical interpretation of the anti-platelet factor 4/heparin enzyme-linked immunosorbent assay for the diagnosis of heparin-induced thrombocytopenia through the use of receiver operating characteristic analysis, stratum-specific likelihood ratios, and Bayes theorem. Chest. 2013;144(4):1269-1275. doi:10.1378/chest.12-2712
8. Warkentin TE, Arnold DM, Nazi I, Kelton JG. The platelet serotonin-release assay. Am J Hematol. 2015;90(6):564-572. doi:10.1002/ajh.24006
9. Use IFOR, Contents TOF. LIFECODES ® PF4 IgG assay:1-9.
10. Ancker JS, Edwards A, Nosal S, Hauser D, Mauer E, Kaushal R. Effects of workload, work complexity, and repeated alerts on alert fatigue in a clinical decision support system. BMC Med Inform Decis Mak. 2017;17(1):1-9. doi:10.1186/s12911-017-0430-8
11. O’Connor N, Carter-Johnson R. Effective screening of pathology tests controls costs: thrombophilia testing. J Clin Pathol. 2006;59(5):556. doi:10.1136/jcp.2005.030700
12. Lim MY, Greenberg CS. Inpatient thrombophilia testing: Impact of healthcare system technology and targeted clinician education on changing practice patterns. Vasc Med (United Kingdom). 2018;23(1):78-79. doi:10.1177/1358863X17742509
13. Cox JL, Shunkwiler SM, Koepsell SA. Requirement for a pathologist’s second signature limits inappropriate inpatient thrombophilia testing. Lab Med. 2017;48(4):367-371. doi:10.1093/labmed/lmx040
14. Kwang H, Mou E, Richman I, et al. Thrombophilia testing in the inpatient setting: impact of an educational intervention. BMC Med Inform Decis Mak. 2019;19(1):167. doi:10.1186/s12911-019-0889-6
15. Shah T, Patel-Teague S, Kroupa L, Meyer AND, Singh H. Impact of a national QI programme on reducing electronic health record notifications to clinicians. BMJ Qual Saf. 2019;28(1):10-14. doi:10.1136/bmjqs-2017-007447
16. Singh H, Spitzmueller C, Petersen NJ, Sawhney MK, Sittig DF. Information overload and missed test results in electronic health record-based settings. JAMA Intern Med. 2013;173(8):702-704. doi:10.1001/2013.jamainternmed.61
From the Veterans Affairs Ann Arbor Healthcare System Medicine Service (Dr. Cusick), University of Michigan College of Pharmacy, Clinical Pharmacy Service, Michigan Medicine (Dr. Hanigan), Department of Internal Medicine Clinical Experience and Quality, Michigan Medicine (Linda Bashaw), Department of Internal Medicine, University of Michigan Medical School, Ann Arbor, MI (Dr. Heidemann), and the Operational Excellence Department, Sparrow Health System, Lansing, MI (Matthew Johnson).
Abstract
Background: Diagnosis of heparin-induced thrombocytopenia (HIT) requires completion of an enzyme-linked immunosorbent assay (ELISA)–based heparin-platelet factor 4 (PF4) antibody test. If this test is negative, HIT is excluded. If positive, a serotonin-release assay (SRA) test is indicated. The SRA is expensive and sometimes inappropriately ordered despite negative PF4 results, leading to unnecessary treatment with argatroban while awaiting SRA results.
Objectives: The primary objectives of this project were to reduce unnecessary SRA testing and argatroban utilization in patients with suspected HIT.
Methods: The authors implemented an intervention at a tertiary care academic hospital in November 2017 targeting patients hospitalized with suspected HIT. The intervention was controlled at the level of the laboratory and prevented ordering of SRA tests in the absence of a positive PF4 test. The number of SRA tests performed and argatroban bags administered were identified retrospectively via chart review before the intervention (January 2016 to November 2017) and post intervention (December 2017 to March 2020). Associated costs were calculated based on institutional SRA testing cost as well as the average wholesale price of argatroban.
Results: SRA testing decreased from an average of 3.7 SRA results per 1000 admissions before the intervention to an average of 0.6 results per 1000 admissions post intervention. The number of 50-mL argatroban bags used per 1000 admissions decreased from 18.8 prior to the intervention to 14.3 post intervention. Total estimated cost savings per 1000 admissions was $2361.20.
Conclusion: An evidence-based testing strategy for HIT can be effectively implemented at the level of the laboratory. This approach led to reductions in SRA testing and argatroban utilization with resultant cost savings.
Keywords: HIT, argatroban, anticoagulation, serotonin-release assay.
Thrombocytopenia is a common finding in hospitalized patients.1,2 Heparin-induced thrombocytopenia (HIT) is one of the many potential causes of thrombocytopenia in hospitalized patients and occurs when antibodies to the heparin-platelet factor 4 (PF4) complex develop after heparin exposure. This triggers a cascade of events, leading to platelet activation, platelet consumption, and thrombosis. While HIT is relatively rare, occurring in 0.3% to 0.5% of critically ill patients, many patients will be tested to rule out this potentially life-threatening cause of thrombocytopenia.3
The diagnosis of HIT utilizes a combination of both clinical suspicion and laboratory testing.4 The 4T score (Table) was developed to evaluate the clinical probability of HIT and involves assessing the degree and timing of thrombocytopenia, the presence or absence of thrombosis, and other potential causes of the thrombocytopenia.5 The 4T score is designed to be utilized to identify patients who require laboratory testing for HIT; however, it has low inter-rater agreement in patients undergoing evaluation for HIT,6 and, in our experience, completion of this scoring is time-consuming.
The enzyme-linked immunosorbent assay (ELISA) is a commonly used laboratory test to diagnose HIT that detects antibodies to the heparin-PF4 complex utilizing optical density (OD) units. When using an OD cutoff of 0.400, ELISA PF4 (PF4) tests have a sensitivity of 99.6%, but poor specificity at 69.3%.7 When the PF4 antibody test is positive with an OD ≥0.400, then a functional test is used to determine whether the antibodies detected will activate platelets. The serotonin-release assay (SRA) is a functional test that measures 14C-labeled serotonin release from donor platelets when mixed with patient serum or plasma containing HIT antibodies. In the correct clinical context, a positive ELISA PF4 antibody test along with a positive SRA is diagnostic of HIT.8
The process of diagnosing HIT in a timely and cost-effective manner is dependent on the clinician’s experience in diagnosing HIT as well as access to the laboratory testing necessary to confirm the diagnosis. PF4 antibody tests are time-consuming and not always available daily and/or are not available onsite. The SRA requires access to donor platelets and specialized radioactivity counting equipment, making it available only at particular centers.
The treatment of HIT is more straightforward and involves stopping all heparin products and starting a nonheparin anticoagulant. The direct thrombin inhibitor argatroban is one of the standard nonheparin anticoagulants used in patients with suspected HIT.4 While it is expensive, its short half-life and lack of renal clearance make it ideal for treatment of hospitalized patients with suspected HIT, many of whom need frequent procedures and/or have renal disease.
At our academic tertiary care center, we performed a retrospective analysis that showed inappropriate ordering of diagnostic HIT testing as well as unnecessary use of argatroban even when there was low suspicion for HIT based on laboratory findings. The aim of our project was to reduce unnecessary HIT testing and argatroban utilization without overburdening providers or interfering with established workflows.
Methods
Setting
The University of Michigan (UM) hospital is a 1000-bed tertiary care center in Ann Arbor, Michigan. The UM guidelines reflect evidence-based guidelines for the diagnosis and treatment of HIT.4 In 2016 the UM guidelines for laboratory testing included sending the PF4 antibody test first when there was clinical suspicion of HIT. The SRA was to be sent separately only when the PF4 returned positive (OD ≥ 0.400). Standard guidelines at UM also included switching patients with suspected HIT from heparin to a nonheparin anticoagulant and stopping all heparin products while awaiting the SRA results. The direct thrombin inhibitor argatroban is utilized at UM and monitored with anti-IIa levels. University of Michigan Hospital utilizes the Immucor PF4 IgG ELISA for detecting heparin-associated antibodies.9 In 2016, this PF4 test was performed in the UM onsite laboratory Monday through Friday. At UM the SRA is performed off site, with a turnaround time of 3 to 5 business days.
Baseline Data
We retrospectively reviewed PF4 and SRA testing as well as argatroban usage from December 2016 to May 2017. Despite the institutional guidelines, providers were sending PF4 and SRA simultaneously as soon as HIT was suspected; 62% of PF4 tests were ordered simultaneously with the SRA, but only 8% of these PF4 tests were positive with an OD ≥0.400. Of those patients with negative PF4 testing, argatroban was continued until the SRA returned negative, leading to many days of unnecessary argatroban usage. An informal survey of the anticoagulation pharmacists revealed that many recommended discontinuing argatroban when the PF4 test was negative, but providers routinely did not feel comfortable with this approach. This suggested many providers misunderstood the performance characteristics of the PF4 test.
Intervention
Our team consisted of hematology and internal medicine faculty, pharmacists, coagulation laboratory personnel, and quality improvement specialists. We designed and implemented an intervention in November 2017 focused on controlling the ordering of the SRA test. We chose to focus on this step due to the excellent sensitivity of the PF4 test with a cutoff of OD <0.400 and the significant expense of the SRA test. Under direction of the Coagulation Laboratory Director, a standard operating procedure was developed where the coagulation laboratory personnel did not send out the SRA until a positive PF4 test (OD ≥ 0.400) was reported. If the PF4 was negative, the SRA was canceled and the ordering provider received notification of the cancelled test via the electronic medical record, accompanied by education about HIT testing (Figure 1). In addition, the lab increased the availability of PF4 testing from 5 days to 7 days a week so there were no delays in tests ordered on Fridays or weekends.

Outcomes
Our primary goals were to decrease both SRA testing and argatroban use. Secondarily, we examined the cost-effectiveness of this intervention. We hypothesized that controlling the SRA testing at the laboratory level would decrease both SRA testing and argatroban use.
Data Collection
Pre- and postintervention data were collected retrospectively. Pre-intervention data were from January 2016 through November 2017, and postintervention data were from December 2017 through March 2020. The number of SRA tests performed were identified retrospectively via review of electronic ordering records. All patients who had a hospital admission after January 1, 2016, were included. These patients were filtered to include only those who had a result for an SRA test. In order to calculate cost-savings, we identified both the number of SRA tests ordered retrospectively as well as patients who had both an SRA resulted and had been administered argatroban. Cost-savings were calculated based on our institutional cost of $357 per SRA test.
At our institution, argatroban is supplied in 50-mL bags; therefore, we utilized the number of bags to identify argatroban usage. Savings were calculated using the average wholesale price (AWP) of $292.50 per 50-mL bag. The amounts billed or collected for the SRA testing or argatroban treatment were not collected. Costs were estimated using only direct costs to the institution. Safety data were not collected. As the intent of our project was a quality improvement activity, this project did not require institutional review board regulation per our institutional guidance.
Results
During the pre-intervention period, the average number of admissions (adults and children) at UM was 5863 per month. Post intervention there was an average of 5842 admissions per month. A total of 1192 PF4 tests were ordered before the intervention and 1148 were ordered post intervention. Prior to the intervention, 481 SRA tests were completed, while post intervention 105 were completed. Serotonin-release testing decreased from an average of 3.7 SRA results per 1000 admissions during the pre-intervention period to an average of 0.6 per 1000 admissions post intervention (Figure 2). Cost-savings were $1045 per 1000 admissions.

During the pre-intervention period, 2539 bags of argatroban were used, while 2337 bags were used post intervention. The number of 50-mL argatroban bags used per 1000 admissions decreased from 18.8 before the intervention to 14.3 post intervention. Cost-savings were $1316.20 per 1000 admissions. Figure 3 illustrates the monthly argatroban utilization per 1000 admissions during each quarter from January 2016 through March 2020.

Discussion
We designed and implemented an evidence-based strategy for HIT at our academic institution which led to a decrease in unnecessary SRA testing and argatroban utilization, with associated cost savings. By focusing on a single point of intervention at the laboratory level where SRA tests were held and canceled if the PF4 test was negative, we helped offload the decision-making from the provider while simultaneously providing just-in-time education to the provider. This intervention was designed with input from multiple stakeholders, including physicians, quality improvement specialists, pharmacists, and coagulation laboratory personnel.
Serotonin-release testing dramatically decreased post intervention even though a similar number of PF4 tests were performed before and after the intervention. This suggests that the decrease in SRA testing was a direct consequence of our intervention. Post intervention the number of completed SRA tests was 9% of the number of PF4 tests sent. This is consistent with our baseline pre-intervention data showing that only 8% of all PF4 tests sent were positive.
While the absolute number of argatroban bags utilized did not dramatically decrease after the intervention, the quarterly rate did, particularly after 2018. Given that argatroban data were only drawn from patients with a concurrent SRA test, this decrease is clearly from decreased usage in patients with suspected HIT. We suspect the decrease occurred because argatroban was not being continued while awaiting an SRA test in patients with a negative PF4 test. Decreasing the utilization of argatroban not only saved money but also reduced days of exposure to argatroban. While we do not have data regarding adverse events related to argatroban prior to the intervention, it is logical to conclude that reducing unnecessary exposure to argatroban reduces the risk of adverse events related to bleeding. Future studies would ideally address specific safety outcome metrics such as adverse events, bleeding risk, or missed diagnoses of HIT.
Our institutional guidelines for the diagnosis of HIT are evidence-based and helpful but are rarely followed by busy inpatient providers. Controlling the utilization of the SRA at the laboratory level had several advantages. First, removing SRA decision-making from providers who are not experts in the diagnosis of HIT guaranteed adherence to evidence-based guidelines. Second, pharmacists could safely recommend discontinuing argatroban when the PF4 test was negative as there was no SRA pending. Third, with cancellation at the laboratory level there was no need to further burden providers with yet another alert in the electronic health record. Fourth, just-in-time education was provided to the providers with justification for why the SRA test was canceled. Last, ruling out HIT within 24 hours with the PF4 test alone allowed providers to evaluate patients for other causes of thrombocytopenia much earlier than the 3 to 5 business days before the SRA results returned.
A limitation of this study is that it was conducted at a single center. Our approach is also limited by the lack of universal applicability. At our institution we are fortunate to have PF4 testing available in our coagulation laboratory 7 days a week. In addition, the coagulation laboratory controls sending the SRA to the reference laboratory. The specific intervention of controlling the SRA testing is therefore applicable only to institutions similar to ours; however, the concept of removing control of specialized testing from the provider is not unique. Inpatient thrombophilia testing has been a successful target of this approach.11-13 While electronic alerts and education of individual providers can also be effective initially, the effectiveness of these interventions has been repeatedly shown to wane over time.14-16
Conclusion
At our institution we were able to implement practical, evidence-based testing for HIT by implementing control over SRA testing at the level of the laboratory. This approach led to decreased argatroban utilization and cost savings.
Corresponding author: Alice Cusick, MD; LTC Charles S Kettles VA Medical Center, 2215 Fuller Road, Ann Arbor, MI 48105; mccoyag@med.umich.edu
Disclosures: None reported.
doi: 10.12788/jcom.0087
From the Veterans Affairs Ann Arbor Healthcare System Medicine Service (Dr. Cusick), University of Michigan College of Pharmacy, Clinical Pharmacy Service, Michigan Medicine (Dr. Hanigan), Department of Internal Medicine Clinical Experience and Quality, Michigan Medicine (Linda Bashaw), Department of Internal Medicine, University of Michigan Medical School, Ann Arbor, MI (Dr. Heidemann), and the Operational Excellence Department, Sparrow Health System, Lansing, MI (Matthew Johnson).
Abstract
Background: Diagnosis of heparin-induced thrombocytopenia (HIT) requires completion of an enzyme-linked immunosorbent assay (ELISA)–based heparin-platelet factor 4 (PF4) antibody test. If this test is negative, HIT is excluded. If positive, a serotonin-release assay (SRA) test is indicated. The SRA is expensive and sometimes inappropriately ordered despite negative PF4 results, leading to unnecessary treatment with argatroban while awaiting SRA results.
Objectives: The primary objectives of this project were to reduce unnecessary SRA testing and argatroban utilization in patients with suspected HIT.
Methods: The authors implemented an intervention at a tertiary care academic hospital in November 2017 targeting patients hospitalized with suspected HIT. The intervention was controlled at the level of the laboratory and prevented ordering of SRA tests in the absence of a positive PF4 test. The number of SRA tests performed and argatroban bags administered were identified retrospectively via chart review before the intervention (January 2016 to November 2017) and post intervention (December 2017 to March 2020). Associated costs were calculated based on institutional SRA testing cost as well as the average wholesale price of argatroban.
Results: SRA testing decreased from an average of 3.7 SRA results per 1000 admissions before the intervention to an average of 0.6 results per 1000 admissions post intervention. The number of 50-mL argatroban bags used per 1000 admissions decreased from 18.8 prior to the intervention to 14.3 post intervention. Total estimated cost savings per 1000 admissions was $2361.20.
Conclusion: An evidence-based testing strategy for HIT can be effectively implemented at the level of the laboratory. This approach led to reductions in SRA testing and argatroban utilization with resultant cost savings.
Keywords: HIT, argatroban, anticoagulation, serotonin-release assay.
Thrombocytopenia is a common finding in hospitalized patients.1,2 Heparin-induced thrombocytopenia (HIT) is one of the many potential causes of thrombocytopenia in hospitalized patients and occurs when antibodies to the heparin-platelet factor 4 (PF4) complex develop after heparin exposure. This triggers a cascade of events, leading to platelet activation, platelet consumption, and thrombosis. While HIT is relatively rare, occurring in 0.3% to 0.5% of critically ill patients, many patients will be tested to rule out this potentially life-threatening cause of thrombocytopenia.3
The diagnosis of HIT utilizes a combination of both clinical suspicion and laboratory testing.4 The 4T score (Table) was developed to evaluate the clinical probability of HIT and involves assessing the degree and timing of thrombocytopenia, the presence or absence of thrombosis, and other potential causes of the thrombocytopenia.5 The 4T score is designed to be utilized to identify patients who require laboratory testing for HIT; however, it has low inter-rater agreement in patients undergoing evaluation for HIT,6 and, in our experience, completion of this scoring is time-consuming.
The enzyme-linked immunosorbent assay (ELISA) is a commonly used laboratory test to diagnose HIT that detects antibodies to the heparin-PF4 complex utilizing optical density (OD) units. When using an OD cutoff of 0.400, ELISA PF4 (PF4) tests have a sensitivity of 99.6%, but poor specificity at 69.3%.7 When the PF4 antibody test is positive with an OD ≥0.400, then a functional test is used to determine whether the antibodies detected will activate platelets. The serotonin-release assay (SRA) is a functional test that measures 14C-labeled serotonin release from donor platelets when mixed with patient serum or plasma containing HIT antibodies. In the correct clinical context, a positive ELISA PF4 antibody test along with a positive SRA is diagnostic of HIT.8
The process of diagnosing HIT in a timely and cost-effective manner is dependent on the clinician’s experience in diagnosing HIT as well as access to the laboratory testing necessary to confirm the diagnosis. PF4 antibody tests are time-consuming and not always available daily and/or are not available onsite. The SRA requires access to donor platelets and specialized radioactivity counting equipment, making it available only at particular centers.
The treatment of HIT is more straightforward and involves stopping all heparin products and starting a nonheparin anticoagulant. The direct thrombin inhibitor argatroban is one of the standard nonheparin anticoagulants used in patients with suspected HIT.4 While it is expensive, its short half-life and lack of renal clearance make it ideal for treatment of hospitalized patients with suspected HIT, many of whom need frequent procedures and/or have renal disease.
At our academic tertiary care center, we performed a retrospective analysis that showed inappropriate ordering of diagnostic HIT testing as well as unnecessary use of argatroban even when there was low suspicion for HIT based on laboratory findings. The aim of our project was to reduce unnecessary HIT testing and argatroban utilization without overburdening providers or interfering with established workflows.
Methods
Setting
The University of Michigan (UM) hospital is a 1000-bed tertiary care center in Ann Arbor, Michigan. The UM guidelines reflect evidence-based guidelines for the diagnosis and treatment of HIT.4 In 2016 the UM guidelines for laboratory testing included sending the PF4 antibody test first when there was clinical suspicion of HIT. The SRA was to be sent separately only when the PF4 returned positive (OD ≥ 0.400). Standard guidelines at UM also included switching patients with suspected HIT from heparin to a nonheparin anticoagulant and stopping all heparin products while awaiting the SRA results. The direct thrombin inhibitor argatroban is utilized at UM and monitored with anti-IIa levels. University of Michigan Hospital utilizes the Immucor PF4 IgG ELISA for detecting heparin-associated antibodies.9 In 2016, this PF4 test was performed in the UM onsite laboratory Monday through Friday. At UM the SRA is performed off site, with a turnaround time of 3 to 5 business days.
Baseline Data
We retrospectively reviewed PF4 and SRA testing as well as argatroban usage from December 2016 to May 2017. Despite the institutional guidelines, providers were sending PF4 and SRA simultaneously as soon as HIT was suspected; 62% of PF4 tests were ordered simultaneously with the SRA, but only 8% of these PF4 tests were positive with an OD ≥0.400. Of those patients with negative PF4 testing, argatroban was continued until the SRA returned negative, leading to many days of unnecessary argatroban usage. An informal survey of the anticoagulation pharmacists revealed that many recommended discontinuing argatroban when the PF4 test was negative, but providers routinely did not feel comfortable with this approach. This suggested many providers misunderstood the performance characteristics of the PF4 test.
Intervention
Our team consisted of hematology and internal medicine faculty, pharmacists, coagulation laboratory personnel, and quality improvement specialists. We designed and implemented an intervention in November 2017 focused on controlling the ordering of the SRA test. We chose to focus on this step due to the excellent sensitivity of the PF4 test with a cutoff of OD <0.400 and the significant expense of the SRA test. Under direction of the Coagulation Laboratory Director, a standard operating procedure was developed where the coagulation laboratory personnel did not send out the SRA until a positive PF4 test (OD ≥ 0.400) was reported. If the PF4 was negative, the SRA was canceled and the ordering provider received notification of the cancelled test via the electronic medical record, accompanied by education about HIT testing (Figure 1). In addition, the lab increased the availability of PF4 testing from 5 days to 7 days a week so there were no delays in tests ordered on Fridays or weekends.

Outcomes
Our primary goals were to decrease both SRA testing and argatroban use. Secondarily, we examined the cost-effectiveness of this intervention. We hypothesized that controlling the SRA testing at the laboratory level would decrease both SRA testing and argatroban use.
Data Collection
Pre- and postintervention data were collected retrospectively. Pre-intervention data were from January 2016 through November 2017, and postintervention data were from December 2017 through March 2020. The number of SRA tests performed were identified retrospectively via review of electronic ordering records. All patients who had a hospital admission after January 1, 2016, were included. These patients were filtered to include only those who had a result for an SRA test. In order to calculate cost-savings, we identified both the number of SRA tests ordered retrospectively as well as patients who had both an SRA resulted and had been administered argatroban. Cost-savings were calculated based on our institutional cost of $357 per SRA test.
At our institution, argatroban is supplied in 50-mL bags; therefore, we utilized the number of bags to identify argatroban usage. Savings were calculated using the average wholesale price (AWP) of $292.50 per 50-mL bag. The amounts billed or collected for the SRA testing or argatroban treatment were not collected. Costs were estimated using only direct costs to the institution. Safety data were not collected. As the intent of our project was a quality improvement activity, this project did not require institutional review board regulation per our institutional guidance.
Results
During the pre-intervention period, the average number of admissions (adults and children) at UM was 5863 per month. Post intervention there was an average of 5842 admissions per month. A total of 1192 PF4 tests were ordered before the intervention and 1148 were ordered post intervention. Prior to the intervention, 481 SRA tests were completed, while post intervention 105 were completed. Serotonin-release testing decreased from an average of 3.7 SRA results per 1000 admissions during the pre-intervention period to an average of 0.6 per 1000 admissions post intervention (Figure 2). Cost-savings were $1045 per 1000 admissions.

During the pre-intervention period, 2539 bags of argatroban were used, while 2337 bags were used post intervention. The number of 50-mL argatroban bags used per 1000 admissions decreased from 18.8 before the intervention to 14.3 post intervention. Cost-savings were $1316.20 per 1000 admissions. Figure 3 illustrates the monthly argatroban utilization per 1000 admissions during each quarter from January 2016 through March 2020.

Discussion
We designed and implemented an evidence-based strategy for HIT at our academic institution which led to a decrease in unnecessary SRA testing and argatroban utilization, with associated cost savings. By focusing on a single point of intervention at the laboratory level where SRA tests were held and canceled if the PF4 test was negative, we helped offload the decision-making from the provider while simultaneously providing just-in-time education to the provider. This intervention was designed with input from multiple stakeholders, including physicians, quality improvement specialists, pharmacists, and coagulation laboratory personnel.
Serotonin-release testing dramatically decreased post intervention even though a similar number of PF4 tests were performed before and after the intervention. This suggests that the decrease in SRA testing was a direct consequence of our intervention. Post intervention the number of completed SRA tests was 9% of the number of PF4 tests sent. This is consistent with our baseline pre-intervention data showing that only 8% of all PF4 tests sent were positive.
While the absolute number of argatroban bags utilized did not dramatically decrease after the intervention, the quarterly rate did, particularly after 2018. Given that argatroban data were only drawn from patients with a concurrent SRA test, this decrease is clearly from decreased usage in patients with suspected HIT. We suspect the decrease occurred because argatroban was not being continued while awaiting an SRA test in patients with a negative PF4 test. Decreasing the utilization of argatroban not only saved money but also reduced days of exposure to argatroban. While we do not have data regarding adverse events related to argatroban prior to the intervention, it is logical to conclude that reducing unnecessary exposure to argatroban reduces the risk of adverse events related to bleeding. Future studies would ideally address specific safety outcome metrics such as adverse events, bleeding risk, or missed diagnoses of HIT.
Our institutional guidelines for the diagnosis of HIT are evidence-based and helpful but are rarely followed by busy inpatient providers. Controlling the utilization of the SRA at the laboratory level had several advantages. First, removing SRA decision-making from providers who are not experts in the diagnosis of HIT guaranteed adherence to evidence-based guidelines. Second, pharmacists could safely recommend discontinuing argatroban when the PF4 test was negative as there was no SRA pending. Third, with cancellation at the laboratory level there was no need to further burden providers with yet another alert in the electronic health record. Fourth, just-in-time education was provided to the providers with justification for why the SRA test was canceled. Last, ruling out HIT within 24 hours with the PF4 test alone allowed providers to evaluate patients for other causes of thrombocytopenia much earlier than the 3 to 5 business days before the SRA results returned.
A limitation of this study is that it was conducted at a single center. Our approach is also limited by the lack of universal applicability. At our institution we are fortunate to have PF4 testing available in our coagulation laboratory 7 days a week. In addition, the coagulation laboratory controls sending the SRA to the reference laboratory. The specific intervention of controlling the SRA testing is therefore applicable only to institutions similar to ours; however, the concept of removing control of specialized testing from the provider is not unique. Inpatient thrombophilia testing has been a successful target of this approach.11-13 While electronic alerts and education of individual providers can also be effective initially, the effectiveness of these interventions has been repeatedly shown to wane over time.14-16
Conclusion
At our institution we were able to implement practical, evidence-based testing for HIT by implementing control over SRA testing at the level of the laboratory. This approach led to decreased argatroban utilization and cost savings.
Corresponding author: Alice Cusick, MD; LTC Charles S Kettles VA Medical Center, 2215 Fuller Road, Ann Arbor, MI 48105; mccoyag@med.umich.edu
Disclosures: None reported.
doi: 10.12788/jcom.0087
1. Fountain E, Arepally GM. Thrombocytopenia in hospitalized non-ICU patients. Blood. 2015;126(23):1060. doi:10.1182/blood.v126.23.1060.1060
2. Hui P, Cook DJ, Lim W, Fraser GA, Arnold DM. The frequency and clinical significance of thrombocytopenia complicating critical illness: a systematic review. Chest. 2011;139(2):271-278. doi:10.1378/chest.10-2243
3. Warkentin TE. Heparin-induced thrombocytopenia. Curr Opin Crit Care. 2015;21(6):576-585. doi:10.1097/MCC.0000000000000259
4. Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv. 2018;2(22):3360-3392. doi:10.1182/bloodadvances.2018024489
5. Cuker A, Gimotty PA, Crowther MA, Warkentin TE. Predictive value of the 4Ts scoring system for heparin-induced thrombocytopenia: a systematic review and meta-analysis. Blood. 2012;120(20):4160-4167. doi:10.1182/blood-2012-07-443051
6. Northam KA, Parker WF, Chen S-L, et al. Evaluation of 4Ts score inter-rater agreement in patients undergoing evaluation for heparin-induced thrombocytopenia. Blood Coagul Fibrinolysis. 2021;32(5):328-334. doi:10.1097/MBC.0000000000001042
7. Raschke RA, Curry SC, Warkentin TE, Gerkin RD. Improving clinical interpretation of the anti-platelet factor 4/heparin enzyme-linked immunosorbent assay for the diagnosis of heparin-induced thrombocytopenia through the use of receiver operating characteristic analysis, stratum-specific likelihood ratios, and Bayes theorem. Chest. 2013;144(4):1269-1275. doi:10.1378/chest.12-2712
8. Warkentin TE, Arnold DM, Nazi I, Kelton JG. The platelet serotonin-release assay. Am J Hematol. 2015;90(6):564-572. doi:10.1002/ajh.24006
9. Use IFOR, Contents TOF. LIFECODES ® PF4 IgG assay:1-9.
10. Ancker JS, Edwards A, Nosal S, Hauser D, Mauer E, Kaushal R. Effects of workload, work complexity, and repeated alerts on alert fatigue in a clinical decision support system. BMC Med Inform Decis Mak. 2017;17(1):1-9. doi:10.1186/s12911-017-0430-8
11. O’Connor N, Carter-Johnson R. Effective screening of pathology tests controls costs: thrombophilia testing. J Clin Pathol. 2006;59(5):556. doi:10.1136/jcp.2005.030700
12. Lim MY, Greenberg CS. Inpatient thrombophilia testing: Impact of healthcare system technology and targeted clinician education on changing practice patterns. Vasc Med (United Kingdom). 2018;23(1):78-79. doi:10.1177/1358863X17742509
13. Cox JL, Shunkwiler SM, Koepsell SA. Requirement for a pathologist’s second signature limits inappropriate inpatient thrombophilia testing. Lab Med. 2017;48(4):367-371. doi:10.1093/labmed/lmx040
14. Kwang H, Mou E, Richman I, et al. Thrombophilia testing in the inpatient setting: impact of an educational intervention. BMC Med Inform Decis Mak. 2019;19(1):167. doi:10.1186/s12911-019-0889-6
15. Shah T, Patel-Teague S, Kroupa L, Meyer AND, Singh H. Impact of a national QI programme on reducing electronic health record notifications to clinicians. BMJ Qual Saf. 2019;28(1):10-14. doi:10.1136/bmjqs-2017-007447
16. Singh H, Spitzmueller C, Petersen NJ, Sawhney MK, Sittig DF. Information overload and missed test results in electronic health record-based settings. JAMA Intern Med. 2013;173(8):702-704. doi:10.1001/2013.jamainternmed.61
1. Fountain E, Arepally GM. Thrombocytopenia in hospitalized non-ICU patients. Blood. 2015;126(23):1060. doi:10.1182/blood.v126.23.1060.1060
2. Hui P, Cook DJ, Lim W, Fraser GA, Arnold DM. The frequency and clinical significance of thrombocytopenia complicating critical illness: a systematic review. Chest. 2011;139(2):271-278. doi:10.1378/chest.10-2243
3. Warkentin TE. Heparin-induced thrombocytopenia. Curr Opin Crit Care. 2015;21(6):576-585. doi:10.1097/MCC.0000000000000259
4. Cuker A, Arepally GM, Chong BH, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: heparin-induced thrombocytopenia. Blood Adv. 2018;2(22):3360-3392. doi:10.1182/bloodadvances.2018024489
5. Cuker A, Gimotty PA, Crowther MA, Warkentin TE. Predictive value of the 4Ts scoring system for heparin-induced thrombocytopenia: a systematic review and meta-analysis. Blood. 2012;120(20):4160-4167. doi:10.1182/blood-2012-07-443051
6. Northam KA, Parker WF, Chen S-L, et al. Evaluation of 4Ts score inter-rater agreement in patients undergoing evaluation for heparin-induced thrombocytopenia. Blood Coagul Fibrinolysis. 2021;32(5):328-334. doi:10.1097/MBC.0000000000001042
7. Raschke RA, Curry SC, Warkentin TE, Gerkin RD. Improving clinical interpretation of the anti-platelet factor 4/heparin enzyme-linked immunosorbent assay for the diagnosis of heparin-induced thrombocytopenia through the use of receiver operating characteristic analysis, stratum-specific likelihood ratios, and Bayes theorem. Chest. 2013;144(4):1269-1275. doi:10.1378/chest.12-2712
8. Warkentin TE, Arnold DM, Nazi I, Kelton JG. The platelet serotonin-release assay. Am J Hematol. 2015;90(6):564-572. doi:10.1002/ajh.24006
9. Use IFOR, Contents TOF. LIFECODES ® PF4 IgG assay:1-9.
10. Ancker JS, Edwards A, Nosal S, Hauser D, Mauer E, Kaushal R. Effects of workload, work complexity, and repeated alerts on alert fatigue in a clinical decision support system. BMC Med Inform Decis Mak. 2017;17(1):1-9. doi:10.1186/s12911-017-0430-8
11. O’Connor N, Carter-Johnson R. Effective screening of pathology tests controls costs: thrombophilia testing. J Clin Pathol. 2006;59(5):556. doi:10.1136/jcp.2005.030700
12. Lim MY, Greenberg CS. Inpatient thrombophilia testing: Impact of healthcare system technology and targeted clinician education on changing practice patterns. Vasc Med (United Kingdom). 2018;23(1):78-79. doi:10.1177/1358863X17742509
13. Cox JL, Shunkwiler SM, Koepsell SA. Requirement for a pathologist’s second signature limits inappropriate inpatient thrombophilia testing. Lab Med. 2017;48(4):367-371. doi:10.1093/labmed/lmx040
14. Kwang H, Mou E, Richman I, et al. Thrombophilia testing in the inpatient setting: impact of an educational intervention. BMC Med Inform Decis Mak. 2019;19(1):167. doi:10.1186/s12911-019-0889-6
15. Shah T, Patel-Teague S, Kroupa L, Meyer AND, Singh H. Impact of a national QI programme on reducing electronic health record notifications to clinicians. BMJ Qual Saf. 2019;28(1):10-14. doi:10.1136/bmjqs-2017-007447
16. Singh H, Spitzmueller C, Petersen NJ, Sawhney MK, Sittig DF. Information overload and missed test results in electronic health record-based settings. JAMA Intern Med. 2013;173(8):702-704. doi:10.1001/2013.jamainternmed.61
Hematocrit, White Blood Cells, and Thrombotic Events in the Veteran Population With Polycythemia Vera
Polycythemia vera (PV) is a rare myeloproliferative neoplasm affecting 44 to 57 individuals per 100,000 in the United States.1,2 It is characterized by somatic mutations in the hematopoietic stem cell, resulting in hyperproliferation of mature myeloid lineage cells.2 Sustained erythrocytosis is a hallmark of PV, although many patients also have leukocytosis and thrombocytosis.2,3 These patients have increased inherent thrombotic risk with arterial events reported to occur at rates of 7 to 21/1000 person-years and venous thrombotic events at 5 to 20/1000 person-years.4-7 Thrombotic and cardiovascular events are leading causes of morbidity and mortality, resulting in a reduced overall survival of patients with PV compared with the general population.3,8-10
Blood Cell Counts and Thrombotic Events in PV
Treatment strategies for patients with PV mainly aim to prevent or manage thrombotic and bleeding complications through normalization of blood counts.11 Hematocrit (Hct) control has been reported to be associated with reduced thrombotic risk in patients with PV. This was shown and popularized by the prospective, randomized Cytoreductive Therapy in Polycythemia Vera (CYTO-PV) trial in which participants were randomized 1:1 to maintaining either a low (< 45%) or high (45%-50%) Hct for 5 years to examine the long-term effects of more- or less-intensive cytoreductive therapy.12 Patients in the low-Hct group were found to have a lower rate of death from cardiovascular events or major thrombosis (1.1/100 person-years in the low-Hct group vs 4.4 in the high-Hct group; hazard ratio [HR], 3.91; 95% confidence interval [CI], 1.45-10.53; P = .007). Likewise, cardiovascular events occurred at a lower rate in patients in the low-Hct group compared with the high-Hct group (4.4% vs 10.9% of patients, respectively; HR, 2.69; 95% CI, 1.19-6.12; P = .02).12
Leukocytosis has also been linked to elevated risk for vascular events as shown in several studies, including the real-world European Collaboration on Low-Dose Aspirin in PV (ECLAP) observational study and a post hoc subanalysis of the CYTO-PV study.13,14 In a multivariate, time-dependent analysis in ECLAP, patients with white blood cell (WBC) counts > 15 × 109/L had a significant increase in the risk of thrombosis compared with those who had lower WBC counts, with higher WBC count more strongly associated with arterial than venous thromboembolism.13 In CYTO-PV, a significant correlation between elevated WBC count (≥ 11 × 109/L vs reference level of < 7 × 109/L) and time-dependent risk of major thrombosis was shown (HR, 3.9; 95% CI, 1.24-12.3; P = .02).14 Likewise, WBC count ≥ 11 × 109/L was found to be a predictor of subsequent venous events in a separate single-center multivariate analysis of patients with PV.8
Although CYTO-PV remains one of the largest prospective landmark studies in PV demonstrating the impact of Hct control on thrombosis, it is worthwhile to note that the patients in the high-Hct group who received less frequent myelosuppressive therapy with hydroxyurea than the low-Hct group also had higher WBC counts.12,15 Work is needed to determine the relative effects of high Hct and high WBC counts on PV independent of each other.
The Veteran Population with PV
Two recently published retrospective analyses from Parasuraman and colleagues used data from the Veterans Health Administration (VHA), the largest integrated health care system in the US, with an aim to replicate findings from CYTO-PV in a real-world population.16,17 The 2 analyses focused independently on the effects of Hct control and WBC count on the risk of a thrombotic event in patients with PV.
In the first retrospective analysis, 213 patients with PV and no prior thrombosis were placed into groups based on whether Hct levels were consistently either < 45% or ≥ 45% throughout the study period.17 The mean follow-up time was 2.3 years, during which 44.1% of patients experienced a thrombotic event (Figure 1). Patients with Hct levels < 45% had a lower rate of thrombotic events compared to those with levels ≥ 45% (40.3% vs 54.2%, respectively; HR, 1.61; 95% CI, 1.03-2.51; P = .04). In a sensitivity analysis that included patients with pre-index thrombotic events (N = 342), similar results were noted (55.6% vs 76.9% between the < 45% and ≥ 45% groups, respectively; HR, 1.95; 95% CI, 1.46-2.61; P < .001).
In the second analysis, the authors investigated the relationship between WBC counts and thrombotic events.16 Evaluable patients (N = 1565) were grouped into 1 of 4 cohorts based on the last WBC measurement taken during the study period before a thrombotic event or through the end of follow-up: (1) WBC < 7.0 × 109/L, (2) 7.0 to 8.4 × 109/L, (3) 8.5 to < 11.0 × 109/L, or (4) ≥ 11.0 × 109/L. Mean follow-up time ranged from 3.6 to 4.5 years among WBC count cohorts, during which 24.9% of patients experienced a thrombotic event. Compared with the reference cohort (WBC < 7.0 × 109/L), a significant positive association between WBC counts and thrombotic event occurrence was observed among patients with WBC counts of 8.5 to < 11.0 × 109/L (HR, 1.47; 95% CI, 1.10-1.96; P < .01) and ≥ 11 × 109/L (HR, 1.87; 95% CI, 1.44-2.43; P < .001) (Figure 2).16 When including all patients in a sensitivity analysis regardless of whether they experienced thrombotic events before the index date (N = 1876), similar results were obtained (7.0-8.4 × 109/L group: HR, 1.22; 95% CI, 0.97-1.55; P = .0959; 8.5 - 11.0 × 109/L group: HR, 1.41; 95% CI, 1.10-1.81; P = .0062; ≥ 11.0 × 109/L group: HR, 1.53; 95% CI, 1.23-1.91; P < .001; compared with < 7.0 × 109/L reference group). Rates of phlebotomy and cytoreductive treatments were similar across groups.16
Some limitations to these studies are attributable to their retrospective design, reliance on health records, and the VHA population characteristics, which differ from the general population. For example, in this analysis, patients with PV in the VHA population had significantly increased risk of thrombotic events, even at a lower WBC count threshold (≥ 8.5 × 109/L) compared with those reported in CYTO-PV (≥ 11 × 109/L). Furthermore, approximately one-third of patients had elevated WBC levels, compared with 25.5% in the CYTO-PV study.14,16 This is most likely due to the unique nature of the VHA patient population, who are predominantly older adult men and generally have a higher comorbidity burden. A notable pre-index comorbidity burden was reported in the VHA population in the Hct analysis, even when compared to patients with PV in the general US population (Charlson Comorbidity Index score, 1.3 vs 0.8).6,17 Comorbid conditions such as hypertension, diabetes, and tobacco use, which are most common among the VHA population, are independently associated with higher risk of cardiovascular and thrombotic events.18,19 However, whether these higher levels of comorbidities affected the type of treatments they received was not elucidated, and the effectiveness of treatments to maintain target Hct levels was not addressed in the study.
Current PV Management and Future Implications
The National Comprehensive Cancer Network (NCCN) clinical practice guidelines in oncology in myeloproliferative neoplasms recommend maintaining Hct levels < 45% in patients with PV.11 Patients with high-risk disease (age ≥ 60 years and/or history of thrombosis) are monitored for new thrombosis or bleeding and are managed for their cardiovascular risk factors. In addition, they receive low-dose aspirin (81-100 mg/day), undergo phlebotomy to maintain an Hct < 45%, and are managed with pharmacologic cytoreductive therapy. Cytoreductive therapy primarily consists of hydroxyurea or peginterferon alfa-2a for younger patients. Ruxolitinib, a Janus kinase (JAK1)/JAK2 inhibitor, is now approved by the US Food and Drug Administration as second-line treatment for those with PV that is intolerant or unresponsive to hydroxyurea or peginterferon alfa-2a treatments.11,20 However, the role of cytoreductive therapy is not clear for patients with low-risk disease (age < 60 years and no history of thrombosis). These patients are managed for their cardiovascular risk factors, undergo phlebotomy to maintain an Hct < 45%, are maintained on low-dose aspirin (81-100 mg/day), and are monitored for indications for cytoreductive therapy, which include any new thrombosis or disease-related major bleeding, frequent or persistent need for phlebotomy with poor tolerance for the procedure, splenomegaly, thrombocytosis, leukocytosis, and disease-related symptoms (eg, aquagenic pruritus, night sweats, fatigue).
Even though the current guidelines recommend maintaining a target Hct of < 45% in patients with high-risk PV, the role of Hct as the main determinant of thrombotic risk in patients with PV is still debated.21 In JAK2V617F-positive essential thrombocythemia, Hct levels are usually normal but risk of thrombosis is nevertheless still significant.22 The risk of thrombosis is significantly lower in primary familial and congenital polycythemia and much lower in secondary erythrocytosis such as cyanotic heart disease, long-term native dwellers of high altitude, and those with high-oxygen–affinity hemoglobins.21,23 In secondary erythrocytosis from hypoxia or upregulated hypoxic pathway such as hypoxia inducible factor-2α (HIF-2α) mutation and Chuvash erythrocytosis, the risk of thrombosis is more associated with the upregulated HIF pathway and its downstream consequences, rather than the elevated Hct level.24
However, most current literature supports the association of increased risk of thrombosis with higher Hct and high WBC count in patients with PV. In addition, the underlying mechanism of thrombogenesis still remains elusive; it is likely a complex process that involves interactions among multiple components, including elevated blood counts arising from clonal hematopoiesis, JAK2V617F allele burden, and platelet and WBC activation and their interaction with endothelial cells and inflammatory cytokines.25
Nevertheless, Hct control and aspirin use are current standard of care for patients with PV to mitigate thrombotic risk, and the results from the 2 analyses by Parasuraman and colleagues, using real-world data from the VHA, support the current practice guidelines to maintain Hct < 45% in these patients. They also provide additional support for considering WBC counts when determining patient risk and treatment plans. Although treatment response criteria from the European LeukemiaNet include achieving normal WBC levels to decrease the risk of thrombosis, current NCCN guidelines do not include WBC counts as a component for establishing patient risk or provide a target WBC count to guide patient management.11,26,27 Updates to these practice guidelines may be warranted. In addition, further study is needed to understand the mechanism of thrombogenesis in PV and other myeloproliferative disorders in order to develop novel therapeutic targets and improve patient outcomes.
Acknowledgments
Writing assistance was provided by Tania Iqbal, PhD, an employee of ICON (North Wales, PA), and was funded by Incyte Corporation (Wilmington, DE).
1. Mehta J, Wang H, Iqbal SU, Mesa R. Epidemiology of myeloproliferative neoplasms in the United States. Leuk Lymphoma. 2014;55(3):595-600. doi:10.3109/10428194.2013.813500
2. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391-2405. doi:10.1182/blood-2016-03-643544
3. Tefferi A, Rumi E, Finazzi G, et al. Survival and prognosis among 1545 patients with contemporary polycythemia vera: an international study. Leukemia. 2013;27(9):1874-1881. doi:10.1038/leu.2013.163
4. Marchioli R, Finazzi G, Landolfi R, et al. Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J Clin Oncol. 2005;23(10):2224-2232. doi:10.1200/JCO.2005.07.062
5. Vannucchi AM, Antonioli E, Guglielmelli P, et al. Clinical profile of homozygous JAK2 617V>F mutation in patients with polycythemia vera or essential thrombocythemia. Blood. 2007;110(3):840-846. doi:10.1182/blood-2006-12-064287
6. Goyal RK, Davis KL, Cote I, Mounedji N, Kaye JA. Increased incidence of thromboembolic event rates in patients diagnosed with polycythemia vera: results from an observational cohort study. Blood (ASH Annual Meeting Abstracts). 2014;124:4840. doi:10.1182/blood.V124.21.4840.4840
7. Barbui T, Carobbio A, Rumi E, et al. In contemporary patients with polycythemia vera, rates of thrombosis and risk factors delineate a new clinical epidemiology. Blood. 2014;124(19):3021-3023. doi:10.1182/blood-2014-07-591610 8. Cerquozzi S, Barraco D, Lasho T, et al. Risk factors for arterial versus venous thrombosis in polycythemia vera: a single center experience in 587 patients. Blood Cancer J. 2017;7(12):662. doi:10.1038/s41408-017-0035-6
9. Stein BL, Moliterno AR, Tiu RV. Polycythemia vera disease burden: contributing factors, impact on quality of life, and emerging treatment options. Ann Hematol. 2014;93(12):1965-1976. doi:10.1007/s00277-014-2205-y
10. Hultcrantz M, Kristinsson SY, Andersson TM-L, et al. Patterns of survival among patients with myeloproliferative neoplasms diagnosed in Sweden from 1973 to 2008: a population-based study. J Clin Oncol. 2012;30(24):2995-3001. doi:10.1200/JCO.2012.42.1925
11. National Comprehensive Cancer Network. NCCN clinical practice guidelines in myeloproliferative neoplasms (Version 1.2020). Accessed March 3, 2022. https://www.nccn.org/professionals/physician_gls/pdf/mpn.pdf
12. Marchioli R, Finazzi G, Specchia G, et al. Cardiovascular events and intensity of treatment in polycythemia vera. N Engl J Med. 2013;368(1):22-33. doi:10.1056/NEJMoa1208500
13. Landolfi R, Di Gennaro L, Barbui T, et al. Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood. 2007;109(6):2446-2452. doi:10.1182/blood-2006-08-042515
14. Barbui T, Masciulli A, Marfisi MR, et al. White blood cell counts and thrombosis in polycythemia vera: a subanalysis of the CYTO-PV study. Blood. 2015;126(4):560-561. doi:10.1182/blood-2015-04-638593
15. Prchal JT, Gordeuk VR. Treatment target in polycythemia vera. N Engl J Med. 2013;368(16):1555-1556. doi:10.1056/NEJMc1301262
16. Parasuraman S, Yu J, Paranagama D, et al. Elevated white blood cell levels and thrombotic events in patients with polycythemia vera: a real-world analysis of Veterans Health Administration data. Clin Lymphoma Myeloma Leuk. 2020;20(2):63-69. doi:10.1016/j.clml.2019.11.010
17. Parasuraman S, Yu J, Paranagama D, et al. Hematocrit levels and thrombotic events in patients with polycythemia vera: an analysis of Veterans Health Administration data. Ann Hematol. 2019;98(11):2533-2539. doi:10.1007/s00277-019-03793-w
18. WHO CVD Risk Chart Working Group. World Health Organization cardiovascular disease risk charts: revised models to estimate risk in 21 global regions. Lancet Glob Health. 2019;7(10):e1332-e1345. doi:10.1016/S2214-109X(19)30318-3.
19. D’Agostino RB Sr, Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. 2008;117(6):743-753. doi:10.1161/CIRCULATIONAHA.107.699579
20. Jakafi. Package insert. Incyte Corporation; 2020.
21. Gordeuk VR, Key NS, Prchal JT. Re-evaluation of hematocrit as a determinant of thrombotic risk in erythrocytosis. Haematologica. 2019;104(4):653-658. doi:10.3324/haematol.2018.210732
22. Carobbio A, Thiele J, Passamonti F, et al. Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: an international study of 891 patients. Blood. 2011;117(22):5857-5859. doi:10.1182/blood-2011-02-339002
23. Perloff JK, Marelli AJ, Miner PD. Risk of stroke in adults with cyanotic congenital heart disease. Circulation. 1993;87(6):1954-1959. doi:10.1161/01.cir.87.6.1954
24. Gordeuk VR, Miasnikova GY, Sergueeva AI, et al. Thrombotic risk in congenital erythrocytosis due to up-regulated hypoxia sensing is not associated with elevated hematocrit. Haematologica. 2020;105(3):e87-e90. doi:10.3324/haematol.2019.216267
25. Kroll MH, Michaelis LC, Verstovsek S. Mechanisms of thrombogenesis in polycythemia vera. Blood Rev. 2015;29(4):215-221. doi:10.1016/j.blre.2014.12.002
26. Barbui T, Tefferi A, Vannucchi AM, et al. Philadelphia chromosome-negative classical myeloproliferative neoplasms: revised management recommendations from European LeukemiaNet. Leukemia. 2018;32(5):1057-1069. doi:10.1038/s41375-018-0077-1
27. Barosi G, Mesa R, Finazzi G, et al. Revised response criteria for polycythemia vera and essential thrombocythemia: an ELN and IWG-MRT consensus project. Blood. 2013;121(23):4778-4781. doi:10.1182/blood-2013-01-478891
Polycythemia vera (PV) is a rare myeloproliferative neoplasm affecting 44 to 57 individuals per 100,000 in the United States.1,2 It is characterized by somatic mutations in the hematopoietic stem cell, resulting in hyperproliferation of mature myeloid lineage cells.2 Sustained erythrocytosis is a hallmark of PV, although many patients also have leukocytosis and thrombocytosis.2,3 These patients have increased inherent thrombotic risk with arterial events reported to occur at rates of 7 to 21/1000 person-years and venous thrombotic events at 5 to 20/1000 person-years.4-7 Thrombotic and cardiovascular events are leading causes of morbidity and mortality, resulting in a reduced overall survival of patients with PV compared with the general population.3,8-10
Blood Cell Counts and Thrombotic Events in PV
Treatment strategies for patients with PV mainly aim to prevent or manage thrombotic and bleeding complications through normalization of blood counts.11 Hematocrit (Hct) control has been reported to be associated with reduced thrombotic risk in patients with PV. This was shown and popularized by the prospective, randomized Cytoreductive Therapy in Polycythemia Vera (CYTO-PV) trial in which participants were randomized 1:1 to maintaining either a low (< 45%) or high (45%-50%) Hct for 5 years to examine the long-term effects of more- or less-intensive cytoreductive therapy.12 Patients in the low-Hct group were found to have a lower rate of death from cardiovascular events or major thrombosis (1.1/100 person-years in the low-Hct group vs 4.4 in the high-Hct group; hazard ratio [HR], 3.91; 95% confidence interval [CI], 1.45-10.53; P = .007). Likewise, cardiovascular events occurred at a lower rate in patients in the low-Hct group compared with the high-Hct group (4.4% vs 10.9% of patients, respectively; HR, 2.69; 95% CI, 1.19-6.12; P = .02).12
Leukocytosis has also been linked to elevated risk for vascular events as shown in several studies, including the real-world European Collaboration on Low-Dose Aspirin in PV (ECLAP) observational study and a post hoc subanalysis of the CYTO-PV study.13,14 In a multivariate, time-dependent analysis in ECLAP, patients with white blood cell (WBC) counts > 15 × 109/L had a significant increase in the risk of thrombosis compared with those who had lower WBC counts, with higher WBC count more strongly associated with arterial than venous thromboembolism.13 In CYTO-PV, a significant correlation between elevated WBC count (≥ 11 × 109/L vs reference level of < 7 × 109/L) and time-dependent risk of major thrombosis was shown (HR, 3.9; 95% CI, 1.24-12.3; P = .02).14 Likewise, WBC count ≥ 11 × 109/L was found to be a predictor of subsequent venous events in a separate single-center multivariate analysis of patients with PV.8
Although CYTO-PV remains one of the largest prospective landmark studies in PV demonstrating the impact of Hct control on thrombosis, it is worthwhile to note that the patients in the high-Hct group who received less frequent myelosuppressive therapy with hydroxyurea than the low-Hct group also had higher WBC counts.12,15 Work is needed to determine the relative effects of high Hct and high WBC counts on PV independent of each other.
The Veteran Population with PV
Two recently published retrospective analyses from Parasuraman and colleagues used data from the Veterans Health Administration (VHA), the largest integrated health care system in the US, with an aim to replicate findings from CYTO-PV in a real-world population.16,17 The 2 analyses focused independently on the effects of Hct control and WBC count on the risk of a thrombotic event in patients with PV.
In the first retrospective analysis, 213 patients with PV and no prior thrombosis were placed into groups based on whether Hct levels were consistently either < 45% or ≥ 45% throughout the study period.17 The mean follow-up time was 2.3 years, during which 44.1% of patients experienced a thrombotic event (Figure 1). Patients with Hct levels < 45% had a lower rate of thrombotic events compared to those with levels ≥ 45% (40.3% vs 54.2%, respectively; HR, 1.61; 95% CI, 1.03-2.51; P = .04). In a sensitivity analysis that included patients with pre-index thrombotic events (N = 342), similar results were noted (55.6% vs 76.9% between the < 45% and ≥ 45% groups, respectively; HR, 1.95; 95% CI, 1.46-2.61; P < .001).
In the second analysis, the authors investigated the relationship between WBC counts and thrombotic events.16 Evaluable patients (N = 1565) were grouped into 1 of 4 cohorts based on the last WBC measurement taken during the study period before a thrombotic event or through the end of follow-up: (1) WBC < 7.0 × 109/L, (2) 7.0 to 8.4 × 109/L, (3) 8.5 to < 11.0 × 109/L, or (4) ≥ 11.0 × 109/L. Mean follow-up time ranged from 3.6 to 4.5 years among WBC count cohorts, during which 24.9% of patients experienced a thrombotic event. Compared with the reference cohort (WBC < 7.0 × 109/L), a significant positive association between WBC counts and thrombotic event occurrence was observed among patients with WBC counts of 8.5 to < 11.0 × 109/L (HR, 1.47; 95% CI, 1.10-1.96; P < .01) and ≥ 11 × 109/L (HR, 1.87; 95% CI, 1.44-2.43; P < .001) (Figure 2).16 When including all patients in a sensitivity analysis regardless of whether they experienced thrombotic events before the index date (N = 1876), similar results were obtained (7.0-8.4 × 109/L group: HR, 1.22; 95% CI, 0.97-1.55; P = .0959; 8.5 - 11.0 × 109/L group: HR, 1.41; 95% CI, 1.10-1.81; P = .0062; ≥ 11.0 × 109/L group: HR, 1.53; 95% CI, 1.23-1.91; P < .001; compared with < 7.0 × 109/L reference group). Rates of phlebotomy and cytoreductive treatments were similar across groups.16
Some limitations to these studies are attributable to their retrospective design, reliance on health records, and the VHA population characteristics, which differ from the general population. For example, in this analysis, patients with PV in the VHA population had significantly increased risk of thrombotic events, even at a lower WBC count threshold (≥ 8.5 × 109/L) compared with those reported in CYTO-PV (≥ 11 × 109/L). Furthermore, approximately one-third of patients had elevated WBC levels, compared with 25.5% in the CYTO-PV study.14,16 This is most likely due to the unique nature of the VHA patient population, who are predominantly older adult men and generally have a higher comorbidity burden. A notable pre-index comorbidity burden was reported in the VHA population in the Hct analysis, even when compared to patients with PV in the general US population (Charlson Comorbidity Index score, 1.3 vs 0.8).6,17 Comorbid conditions such as hypertension, diabetes, and tobacco use, which are most common among the VHA population, are independently associated with higher risk of cardiovascular and thrombotic events.18,19 However, whether these higher levels of comorbidities affected the type of treatments they received was not elucidated, and the effectiveness of treatments to maintain target Hct levels was not addressed in the study.
Current PV Management and Future Implications
The National Comprehensive Cancer Network (NCCN) clinical practice guidelines in oncology in myeloproliferative neoplasms recommend maintaining Hct levels < 45% in patients with PV.11 Patients with high-risk disease (age ≥ 60 years and/or history of thrombosis) are monitored for new thrombosis or bleeding and are managed for their cardiovascular risk factors. In addition, they receive low-dose aspirin (81-100 mg/day), undergo phlebotomy to maintain an Hct < 45%, and are managed with pharmacologic cytoreductive therapy. Cytoreductive therapy primarily consists of hydroxyurea or peginterferon alfa-2a for younger patients. Ruxolitinib, a Janus kinase (JAK1)/JAK2 inhibitor, is now approved by the US Food and Drug Administration as second-line treatment for those with PV that is intolerant or unresponsive to hydroxyurea or peginterferon alfa-2a treatments.11,20 However, the role of cytoreductive therapy is not clear for patients with low-risk disease (age < 60 years and no history of thrombosis). These patients are managed for their cardiovascular risk factors, undergo phlebotomy to maintain an Hct < 45%, are maintained on low-dose aspirin (81-100 mg/day), and are monitored for indications for cytoreductive therapy, which include any new thrombosis or disease-related major bleeding, frequent or persistent need for phlebotomy with poor tolerance for the procedure, splenomegaly, thrombocytosis, leukocytosis, and disease-related symptoms (eg, aquagenic pruritus, night sweats, fatigue).
Even though the current guidelines recommend maintaining a target Hct of < 45% in patients with high-risk PV, the role of Hct as the main determinant of thrombotic risk in patients with PV is still debated.21 In JAK2V617F-positive essential thrombocythemia, Hct levels are usually normal but risk of thrombosis is nevertheless still significant.22 The risk of thrombosis is significantly lower in primary familial and congenital polycythemia and much lower in secondary erythrocytosis such as cyanotic heart disease, long-term native dwellers of high altitude, and those with high-oxygen–affinity hemoglobins.21,23 In secondary erythrocytosis from hypoxia or upregulated hypoxic pathway such as hypoxia inducible factor-2α (HIF-2α) mutation and Chuvash erythrocytosis, the risk of thrombosis is more associated with the upregulated HIF pathway and its downstream consequences, rather than the elevated Hct level.24
However, most current literature supports the association of increased risk of thrombosis with higher Hct and high WBC count in patients with PV. In addition, the underlying mechanism of thrombogenesis still remains elusive; it is likely a complex process that involves interactions among multiple components, including elevated blood counts arising from clonal hematopoiesis, JAK2V617F allele burden, and platelet and WBC activation and their interaction with endothelial cells and inflammatory cytokines.25
Nevertheless, Hct control and aspirin use are current standard of care for patients with PV to mitigate thrombotic risk, and the results from the 2 analyses by Parasuraman and colleagues, using real-world data from the VHA, support the current practice guidelines to maintain Hct < 45% in these patients. They also provide additional support for considering WBC counts when determining patient risk and treatment plans. Although treatment response criteria from the European LeukemiaNet include achieving normal WBC levels to decrease the risk of thrombosis, current NCCN guidelines do not include WBC counts as a component for establishing patient risk or provide a target WBC count to guide patient management.11,26,27 Updates to these practice guidelines may be warranted. In addition, further study is needed to understand the mechanism of thrombogenesis in PV and other myeloproliferative disorders in order to develop novel therapeutic targets and improve patient outcomes.
Acknowledgments
Writing assistance was provided by Tania Iqbal, PhD, an employee of ICON (North Wales, PA), and was funded by Incyte Corporation (Wilmington, DE).
Polycythemia vera (PV) is a rare myeloproliferative neoplasm affecting 44 to 57 individuals per 100,000 in the United States.1,2 It is characterized by somatic mutations in the hematopoietic stem cell, resulting in hyperproliferation of mature myeloid lineage cells.2 Sustained erythrocytosis is a hallmark of PV, although many patients also have leukocytosis and thrombocytosis.2,3 These patients have increased inherent thrombotic risk with arterial events reported to occur at rates of 7 to 21/1000 person-years and venous thrombotic events at 5 to 20/1000 person-years.4-7 Thrombotic and cardiovascular events are leading causes of morbidity and mortality, resulting in a reduced overall survival of patients with PV compared with the general population.3,8-10
Blood Cell Counts and Thrombotic Events in PV
Treatment strategies for patients with PV mainly aim to prevent or manage thrombotic and bleeding complications through normalization of blood counts.11 Hematocrit (Hct) control has been reported to be associated with reduced thrombotic risk in patients with PV. This was shown and popularized by the prospective, randomized Cytoreductive Therapy in Polycythemia Vera (CYTO-PV) trial in which participants were randomized 1:1 to maintaining either a low (< 45%) or high (45%-50%) Hct for 5 years to examine the long-term effects of more- or less-intensive cytoreductive therapy.12 Patients in the low-Hct group were found to have a lower rate of death from cardiovascular events or major thrombosis (1.1/100 person-years in the low-Hct group vs 4.4 in the high-Hct group; hazard ratio [HR], 3.91; 95% confidence interval [CI], 1.45-10.53; P = .007). Likewise, cardiovascular events occurred at a lower rate in patients in the low-Hct group compared with the high-Hct group (4.4% vs 10.9% of patients, respectively; HR, 2.69; 95% CI, 1.19-6.12; P = .02).12
Leukocytosis has also been linked to elevated risk for vascular events as shown in several studies, including the real-world European Collaboration on Low-Dose Aspirin in PV (ECLAP) observational study and a post hoc subanalysis of the CYTO-PV study.13,14 In a multivariate, time-dependent analysis in ECLAP, patients with white blood cell (WBC) counts > 15 × 109/L had a significant increase in the risk of thrombosis compared with those who had lower WBC counts, with higher WBC count more strongly associated with arterial than venous thromboembolism.13 In CYTO-PV, a significant correlation between elevated WBC count (≥ 11 × 109/L vs reference level of < 7 × 109/L) and time-dependent risk of major thrombosis was shown (HR, 3.9; 95% CI, 1.24-12.3; P = .02).14 Likewise, WBC count ≥ 11 × 109/L was found to be a predictor of subsequent venous events in a separate single-center multivariate analysis of patients with PV.8
Although CYTO-PV remains one of the largest prospective landmark studies in PV demonstrating the impact of Hct control on thrombosis, it is worthwhile to note that the patients in the high-Hct group who received less frequent myelosuppressive therapy with hydroxyurea than the low-Hct group also had higher WBC counts.12,15 Work is needed to determine the relative effects of high Hct and high WBC counts on PV independent of each other.
The Veteran Population with PV
Two recently published retrospective analyses from Parasuraman and colleagues used data from the Veterans Health Administration (VHA), the largest integrated health care system in the US, with an aim to replicate findings from CYTO-PV in a real-world population.16,17 The 2 analyses focused independently on the effects of Hct control and WBC count on the risk of a thrombotic event in patients with PV.
In the first retrospective analysis, 213 patients with PV and no prior thrombosis were placed into groups based on whether Hct levels were consistently either < 45% or ≥ 45% throughout the study period.17 The mean follow-up time was 2.3 years, during which 44.1% of patients experienced a thrombotic event (Figure 1). Patients with Hct levels < 45% had a lower rate of thrombotic events compared to those with levels ≥ 45% (40.3% vs 54.2%, respectively; HR, 1.61; 95% CI, 1.03-2.51; P = .04). In a sensitivity analysis that included patients with pre-index thrombotic events (N = 342), similar results were noted (55.6% vs 76.9% between the < 45% and ≥ 45% groups, respectively; HR, 1.95; 95% CI, 1.46-2.61; P < .001).
In the second analysis, the authors investigated the relationship between WBC counts and thrombotic events.16 Evaluable patients (N = 1565) were grouped into 1 of 4 cohorts based on the last WBC measurement taken during the study period before a thrombotic event or through the end of follow-up: (1) WBC < 7.0 × 109/L, (2) 7.0 to 8.4 × 109/L, (3) 8.5 to < 11.0 × 109/L, or (4) ≥ 11.0 × 109/L. Mean follow-up time ranged from 3.6 to 4.5 years among WBC count cohorts, during which 24.9% of patients experienced a thrombotic event. Compared with the reference cohort (WBC < 7.0 × 109/L), a significant positive association between WBC counts and thrombotic event occurrence was observed among patients with WBC counts of 8.5 to < 11.0 × 109/L (HR, 1.47; 95% CI, 1.10-1.96; P < .01) and ≥ 11 × 109/L (HR, 1.87; 95% CI, 1.44-2.43; P < .001) (Figure 2).16 When including all patients in a sensitivity analysis regardless of whether they experienced thrombotic events before the index date (N = 1876), similar results were obtained (7.0-8.4 × 109/L group: HR, 1.22; 95% CI, 0.97-1.55; P = .0959; 8.5 - 11.0 × 109/L group: HR, 1.41; 95% CI, 1.10-1.81; P = .0062; ≥ 11.0 × 109/L group: HR, 1.53; 95% CI, 1.23-1.91; P < .001; compared with < 7.0 × 109/L reference group). Rates of phlebotomy and cytoreductive treatments were similar across groups.16
Some limitations to these studies are attributable to their retrospective design, reliance on health records, and the VHA population characteristics, which differ from the general population. For example, in this analysis, patients with PV in the VHA population had significantly increased risk of thrombotic events, even at a lower WBC count threshold (≥ 8.5 × 109/L) compared with those reported in CYTO-PV (≥ 11 × 109/L). Furthermore, approximately one-third of patients had elevated WBC levels, compared with 25.5% in the CYTO-PV study.14,16 This is most likely due to the unique nature of the VHA patient population, who are predominantly older adult men and generally have a higher comorbidity burden. A notable pre-index comorbidity burden was reported in the VHA population in the Hct analysis, even when compared to patients with PV in the general US population (Charlson Comorbidity Index score, 1.3 vs 0.8).6,17 Comorbid conditions such as hypertension, diabetes, and tobacco use, which are most common among the VHA population, are independently associated with higher risk of cardiovascular and thrombotic events.18,19 However, whether these higher levels of comorbidities affected the type of treatments they received was not elucidated, and the effectiveness of treatments to maintain target Hct levels was not addressed in the study.
Current PV Management and Future Implications
The National Comprehensive Cancer Network (NCCN) clinical practice guidelines in oncology in myeloproliferative neoplasms recommend maintaining Hct levels < 45% in patients with PV.11 Patients with high-risk disease (age ≥ 60 years and/or history of thrombosis) are monitored for new thrombosis or bleeding and are managed for their cardiovascular risk factors. In addition, they receive low-dose aspirin (81-100 mg/day), undergo phlebotomy to maintain an Hct < 45%, and are managed with pharmacologic cytoreductive therapy. Cytoreductive therapy primarily consists of hydroxyurea or peginterferon alfa-2a for younger patients. Ruxolitinib, a Janus kinase (JAK1)/JAK2 inhibitor, is now approved by the US Food and Drug Administration as second-line treatment for those with PV that is intolerant or unresponsive to hydroxyurea or peginterferon alfa-2a treatments.11,20 However, the role of cytoreductive therapy is not clear for patients with low-risk disease (age < 60 years and no history of thrombosis). These patients are managed for their cardiovascular risk factors, undergo phlebotomy to maintain an Hct < 45%, are maintained on low-dose aspirin (81-100 mg/day), and are monitored for indications for cytoreductive therapy, which include any new thrombosis or disease-related major bleeding, frequent or persistent need for phlebotomy with poor tolerance for the procedure, splenomegaly, thrombocytosis, leukocytosis, and disease-related symptoms (eg, aquagenic pruritus, night sweats, fatigue).
Even though the current guidelines recommend maintaining a target Hct of < 45% in patients with high-risk PV, the role of Hct as the main determinant of thrombotic risk in patients with PV is still debated.21 In JAK2V617F-positive essential thrombocythemia, Hct levels are usually normal but risk of thrombosis is nevertheless still significant.22 The risk of thrombosis is significantly lower in primary familial and congenital polycythemia and much lower in secondary erythrocytosis such as cyanotic heart disease, long-term native dwellers of high altitude, and those with high-oxygen–affinity hemoglobins.21,23 In secondary erythrocytosis from hypoxia or upregulated hypoxic pathway such as hypoxia inducible factor-2α (HIF-2α) mutation and Chuvash erythrocytosis, the risk of thrombosis is more associated with the upregulated HIF pathway and its downstream consequences, rather than the elevated Hct level.24
However, most current literature supports the association of increased risk of thrombosis with higher Hct and high WBC count in patients with PV. In addition, the underlying mechanism of thrombogenesis still remains elusive; it is likely a complex process that involves interactions among multiple components, including elevated blood counts arising from clonal hematopoiesis, JAK2V617F allele burden, and platelet and WBC activation and their interaction with endothelial cells and inflammatory cytokines.25
Nevertheless, Hct control and aspirin use are current standard of care for patients with PV to mitigate thrombotic risk, and the results from the 2 analyses by Parasuraman and colleagues, using real-world data from the VHA, support the current practice guidelines to maintain Hct < 45% in these patients. They also provide additional support for considering WBC counts when determining patient risk and treatment plans. Although treatment response criteria from the European LeukemiaNet include achieving normal WBC levels to decrease the risk of thrombosis, current NCCN guidelines do not include WBC counts as a component for establishing patient risk or provide a target WBC count to guide patient management.11,26,27 Updates to these practice guidelines may be warranted. In addition, further study is needed to understand the mechanism of thrombogenesis in PV and other myeloproliferative disorders in order to develop novel therapeutic targets and improve patient outcomes.
Acknowledgments
Writing assistance was provided by Tania Iqbal, PhD, an employee of ICON (North Wales, PA), and was funded by Incyte Corporation (Wilmington, DE).
1. Mehta J, Wang H, Iqbal SU, Mesa R. Epidemiology of myeloproliferative neoplasms in the United States. Leuk Lymphoma. 2014;55(3):595-600. doi:10.3109/10428194.2013.813500
2. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391-2405. doi:10.1182/blood-2016-03-643544
3. Tefferi A, Rumi E, Finazzi G, et al. Survival and prognosis among 1545 patients with contemporary polycythemia vera: an international study. Leukemia. 2013;27(9):1874-1881. doi:10.1038/leu.2013.163
4. Marchioli R, Finazzi G, Landolfi R, et al. Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J Clin Oncol. 2005;23(10):2224-2232. doi:10.1200/JCO.2005.07.062
5. Vannucchi AM, Antonioli E, Guglielmelli P, et al. Clinical profile of homozygous JAK2 617V>F mutation in patients with polycythemia vera or essential thrombocythemia. Blood. 2007;110(3):840-846. doi:10.1182/blood-2006-12-064287
6. Goyal RK, Davis KL, Cote I, Mounedji N, Kaye JA. Increased incidence of thromboembolic event rates in patients diagnosed with polycythemia vera: results from an observational cohort study. Blood (ASH Annual Meeting Abstracts). 2014;124:4840. doi:10.1182/blood.V124.21.4840.4840
7. Barbui T, Carobbio A, Rumi E, et al. In contemporary patients with polycythemia vera, rates of thrombosis and risk factors delineate a new clinical epidemiology. Blood. 2014;124(19):3021-3023. doi:10.1182/blood-2014-07-591610 8. Cerquozzi S, Barraco D, Lasho T, et al. Risk factors for arterial versus venous thrombosis in polycythemia vera: a single center experience in 587 patients. Blood Cancer J. 2017;7(12):662. doi:10.1038/s41408-017-0035-6
9. Stein BL, Moliterno AR, Tiu RV. Polycythemia vera disease burden: contributing factors, impact on quality of life, and emerging treatment options. Ann Hematol. 2014;93(12):1965-1976. doi:10.1007/s00277-014-2205-y
10. Hultcrantz M, Kristinsson SY, Andersson TM-L, et al. Patterns of survival among patients with myeloproliferative neoplasms diagnosed in Sweden from 1973 to 2008: a population-based study. J Clin Oncol. 2012;30(24):2995-3001. doi:10.1200/JCO.2012.42.1925
11. National Comprehensive Cancer Network. NCCN clinical practice guidelines in myeloproliferative neoplasms (Version 1.2020). Accessed March 3, 2022. https://www.nccn.org/professionals/physician_gls/pdf/mpn.pdf
12. Marchioli R, Finazzi G, Specchia G, et al. Cardiovascular events and intensity of treatment in polycythemia vera. N Engl J Med. 2013;368(1):22-33. doi:10.1056/NEJMoa1208500
13. Landolfi R, Di Gennaro L, Barbui T, et al. Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood. 2007;109(6):2446-2452. doi:10.1182/blood-2006-08-042515
14. Barbui T, Masciulli A, Marfisi MR, et al. White blood cell counts and thrombosis in polycythemia vera: a subanalysis of the CYTO-PV study. Blood. 2015;126(4):560-561. doi:10.1182/blood-2015-04-638593
15. Prchal JT, Gordeuk VR. Treatment target in polycythemia vera. N Engl J Med. 2013;368(16):1555-1556. doi:10.1056/NEJMc1301262
16. Parasuraman S, Yu J, Paranagama D, et al. Elevated white blood cell levels and thrombotic events in patients with polycythemia vera: a real-world analysis of Veterans Health Administration data. Clin Lymphoma Myeloma Leuk. 2020;20(2):63-69. doi:10.1016/j.clml.2019.11.010
17. Parasuraman S, Yu J, Paranagama D, et al. Hematocrit levels and thrombotic events in patients with polycythemia vera: an analysis of Veterans Health Administration data. Ann Hematol. 2019;98(11):2533-2539. doi:10.1007/s00277-019-03793-w
18. WHO CVD Risk Chart Working Group. World Health Organization cardiovascular disease risk charts: revised models to estimate risk in 21 global regions. Lancet Glob Health. 2019;7(10):e1332-e1345. doi:10.1016/S2214-109X(19)30318-3.
19. D’Agostino RB Sr, Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. 2008;117(6):743-753. doi:10.1161/CIRCULATIONAHA.107.699579
20. Jakafi. Package insert. Incyte Corporation; 2020.
21. Gordeuk VR, Key NS, Prchal JT. Re-evaluation of hematocrit as a determinant of thrombotic risk in erythrocytosis. Haematologica. 2019;104(4):653-658. doi:10.3324/haematol.2018.210732
22. Carobbio A, Thiele J, Passamonti F, et al. Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: an international study of 891 patients. Blood. 2011;117(22):5857-5859. doi:10.1182/blood-2011-02-339002
23. Perloff JK, Marelli AJ, Miner PD. Risk of stroke in adults with cyanotic congenital heart disease. Circulation. 1993;87(6):1954-1959. doi:10.1161/01.cir.87.6.1954
24. Gordeuk VR, Miasnikova GY, Sergueeva AI, et al. Thrombotic risk in congenital erythrocytosis due to up-regulated hypoxia sensing is not associated with elevated hematocrit. Haematologica. 2020;105(3):e87-e90. doi:10.3324/haematol.2019.216267
25. Kroll MH, Michaelis LC, Verstovsek S. Mechanisms of thrombogenesis in polycythemia vera. Blood Rev. 2015;29(4):215-221. doi:10.1016/j.blre.2014.12.002
26. Barbui T, Tefferi A, Vannucchi AM, et al. Philadelphia chromosome-negative classical myeloproliferative neoplasms: revised management recommendations from European LeukemiaNet. Leukemia. 2018;32(5):1057-1069. doi:10.1038/s41375-018-0077-1
27. Barosi G, Mesa R, Finazzi G, et al. Revised response criteria for polycythemia vera and essential thrombocythemia: an ELN and IWG-MRT consensus project. Blood. 2013;121(23):4778-4781. doi:10.1182/blood-2013-01-478891
1. Mehta J, Wang H, Iqbal SU, Mesa R. Epidemiology of myeloproliferative neoplasms in the United States. Leuk Lymphoma. 2014;55(3):595-600. doi:10.3109/10428194.2013.813500
2. Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391-2405. doi:10.1182/blood-2016-03-643544
3. Tefferi A, Rumi E, Finazzi G, et al. Survival and prognosis among 1545 patients with contemporary polycythemia vera: an international study. Leukemia. 2013;27(9):1874-1881. doi:10.1038/leu.2013.163
4. Marchioli R, Finazzi G, Landolfi R, et al. Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J Clin Oncol. 2005;23(10):2224-2232. doi:10.1200/JCO.2005.07.062
5. Vannucchi AM, Antonioli E, Guglielmelli P, et al. Clinical profile of homozygous JAK2 617V>F mutation in patients with polycythemia vera or essential thrombocythemia. Blood. 2007;110(3):840-846. doi:10.1182/blood-2006-12-064287
6. Goyal RK, Davis KL, Cote I, Mounedji N, Kaye JA. Increased incidence of thromboembolic event rates in patients diagnosed with polycythemia vera: results from an observational cohort study. Blood (ASH Annual Meeting Abstracts). 2014;124:4840. doi:10.1182/blood.V124.21.4840.4840
7. Barbui T, Carobbio A, Rumi E, et al. In contemporary patients with polycythemia vera, rates of thrombosis and risk factors delineate a new clinical epidemiology. Blood. 2014;124(19):3021-3023. doi:10.1182/blood-2014-07-591610 8. Cerquozzi S, Barraco D, Lasho T, et al. Risk factors for arterial versus venous thrombosis in polycythemia vera: a single center experience in 587 patients. Blood Cancer J. 2017;7(12):662. doi:10.1038/s41408-017-0035-6
9. Stein BL, Moliterno AR, Tiu RV. Polycythemia vera disease burden: contributing factors, impact on quality of life, and emerging treatment options. Ann Hematol. 2014;93(12):1965-1976. doi:10.1007/s00277-014-2205-y
10. Hultcrantz M, Kristinsson SY, Andersson TM-L, et al. Patterns of survival among patients with myeloproliferative neoplasms diagnosed in Sweden from 1973 to 2008: a population-based study. J Clin Oncol. 2012;30(24):2995-3001. doi:10.1200/JCO.2012.42.1925
11. National Comprehensive Cancer Network. NCCN clinical practice guidelines in myeloproliferative neoplasms (Version 1.2020). Accessed March 3, 2022. https://www.nccn.org/professionals/physician_gls/pdf/mpn.pdf
12. Marchioli R, Finazzi G, Specchia G, et al. Cardiovascular events and intensity of treatment in polycythemia vera. N Engl J Med. 2013;368(1):22-33. doi:10.1056/NEJMoa1208500
13. Landolfi R, Di Gennaro L, Barbui T, et al. Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood. 2007;109(6):2446-2452. doi:10.1182/blood-2006-08-042515
14. Barbui T, Masciulli A, Marfisi MR, et al. White blood cell counts and thrombosis in polycythemia vera: a subanalysis of the CYTO-PV study. Blood. 2015;126(4):560-561. doi:10.1182/blood-2015-04-638593
15. Prchal JT, Gordeuk VR. Treatment target in polycythemia vera. N Engl J Med. 2013;368(16):1555-1556. doi:10.1056/NEJMc1301262
16. Parasuraman S, Yu J, Paranagama D, et al. Elevated white blood cell levels and thrombotic events in patients with polycythemia vera: a real-world analysis of Veterans Health Administration data. Clin Lymphoma Myeloma Leuk. 2020;20(2):63-69. doi:10.1016/j.clml.2019.11.010
17. Parasuraman S, Yu J, Paranagama D, et al. Hematocrit levels and thrombotic events in patients with polycythemia vera: an analysis of Veterans Health Administration data. Ann Hematol. 2019;98(11):2533-2539. doi:10.1007/s00277-019-03793-w
18. WHO CVD Risk Chart Working Group. World Health Organization cardiovascular disease risk charts: revised models to estimate risk in 21 global regions. Lancet Glob Health. 2019;7(10):e1332-e1345. doi:10.1016/S2214-109X(19)30318-3.
19. D’Agostino RB Sr, Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation. 2008;117(6):743-753. doi:10.1161/CIRCULATIONAHA.107.699579
20. Jakafi. Package insert. Incyte Corporation; 2020.
21. Gordeuk VR, Key NS, Prchal JT. Re-evaluation of hematocrit as a determinant of thrombotic risk in erythrocytosis. Haematologica. 2019;104(4):653-658. doi:10.3324/haematol.2018.210732
22. Carobbio A, Thiele J, Passamonti F, et al. Risk factors for arterial and venous thrombosis in WHO-defined essential thrombocythemia: an international study of 891 patients. Blood. 2011;117(22):5857-5859. doi:10.1182/blood-2011-02-339002
23. Perloff JK, Marelli AJ, Miner PD. Risk of stroke in adults with cyanotic congenital heart disease. Circulation. 1993;87(6):1954-1959. doi:10.1161/01.cir.87.6.1954
24. Gordeuk VR, Miasnikova GY, Sergueeva AI, et al. Thrombotic risk in congenital erythrocytosis due to up-regulated hypoxia sensing is not associated with elevated hematocrit. Haematologica. 2020;105(3):e87-e90. doi:10.3324/haematol.2019.216267
25. Kroll MH, Michaelis LC, Verstovsek S. Mechanisms of thrombogenesis in polycythemia vera. Blood Rev. 2015;29(4):215-221. doi:10.1016/j.blre.2014.12.002
26. Barbui T, Tefferi A, Vannucchi AM, et al. Philadelphia chromosome-negative classical myeloproliferative neoplasms: revised management recommendations from European LeukemiaNet. Leukemia. 2018;32(5):1057-1069. doi:10.1038/s41375-018-0077-1
27. Barosi G, Mesa R, Finazzi G, et al. Revised response criteria for polycythemia vera and essential thrombocythemia: an ELN and IWG-MRT consensus project. Blood. 2013;121(23):4778-4781. doi:10.1182/blood-2013-01-478891
Rare pediatric cancers persist 63 years after nuclear accident
Chernobyl. Fukushima. Three Mile Island.
The world knows these names all too well because of accidents there: complete or partial meltdowns of nuclear reactors that released massive amounts of cancer-causing radiation into the air, soil, and water.
The Santa Susana Field Lab is far less well-known, but no less infamous for what took place at this former rocket engine and nuclear energy test site just 28 miles northwest of downtown Los Angeles.
In July 1959, an accident involving one of 10 experimental nuclear reactors at the SSFL site released a cloud of harmful radiation and toxic chemicals over the surrounding area, including Simi Valley, San Gabriel Valley, Chatsworth, and Canoga Park. The small reactor had no containment vessel.
This accident resulted in a release of radioactive iodine estimated to be as much as 250 times that of the partial meltdown that would occur 2 decades later at Three Mile Island, a much larger commercial reactor that had a containment vessel.
Decades-long cover-up
In 1959, the public knew nothing about what happened at the site.
According to John Pace, then an employee at SSFL, the accident was covered up. Mr. Pace recounted the cover-up in the documentary “In the Dark of the Valley,” which first aired in November 2021 on MSNBC.
In fact, the accident at SSFL remained under wraps for 2 decades, according to Daniel Hirsch, former director of the Program on Environmental and Nuclear Policy at the University of California, Santa Cruz, and now president of Committee to Bridge the Gap, a nuclear policy nongovernmental organization.
“Students working with me while I was teaching at UCLA in 1979 uncovered these Atomic Energy Commission reports from Atomics International,” he said in an interview. “We had to order the documents from the annex to the UCLA Engineering Library. They were stored offsite, and it took a few days, and when we got them, we opened them up, and there were these fold-out photographs of the fuel [rods]. As we folded out the photographs further, we saw one photo with an arrow labeled ‘longitudinal cracks,’ and then other arrows showing other kinds of cracks, and then another arrow labeled ‘melted blob.’ ”
Mr. Hirsch and his students found that other accidents had occurred at SSFL, including a fuel fabrication system that leached plutonium, fires in a “hot” lab where irradiated nuclear fuel from around the United States was handled, and open-air burn pits where radioactive and toxic chemical wastes were illegally torched.
According to the Committee to Bridge the Gap, when the 2,800-acre SSFL site was being developed under the name Rocketdyne by aircraft maker North American Aviation, the area was sparsely populated, with nearly as many grazing animals as people in its hills and valleys.
North American Aviation later became part of Rockwell International, which in turn sold its aerospace and defense business units to the Boeing Company in 1996. Boeing, now in charge of the site and the cleanup efforts, is doing everything in its power to shirk or diminish its responsibility, Mr. Hirsch and other critics say.
Parents against SSFL
Today, more than 150,000 people live within 5 miles of SSFL, and more than half a million live within 10 miles.
Melissa Bumstead is one of those residents. She and her family live 3.7 miles from the Santa Susana site. When her toddler Grace was diagnosed with a rare form of leukemia in 2014, doctors told Ms. Bumstead there were no known links between her daughter’s cancer and environmental contamination.
But during Grace’s treatment at Children’s Hospital Los Angeles, her mother began meeting other parents who lived near her and had children facing equally rare cancers.
Lauren Hammersley, whose daughter Hazel was diagnosed with a rare brain tumor called neuroblastoma at age 2, lived about 10 miles from Ms. Bumstead on the other side of a mountain and just over 4 miles from SSFL.
On her street alone, Ms. Bumstead discovered three cases of pediatric cancer, including two children in adjacent homes who had the same rare brain tumor as Hazel Hammersley.
As Ms. Bumstead told Los Angeles National Public Radio station KCRW in 2021, “I started to panic because I knew that childhood cancer is extremely rare. There’s only 15,000 new cases every year out of 72 million children in America. So, the chance of knowing your neighbors, especially at an internationally renowned hospital like Children’s Hospital Los Angeles – we knew something wasn’t right.”
After a relapse of her tumor, Hazel died in 2018, a few months after her seventh birthday.
Cancer clusters
Hoping to understand why their kids were getting so sick, Ms. Bumstead and the other parents formed a Facebook group. They plotted their homes on Google Maps and found that they all lived within roughly 10 miles of one another. It would take another year for them to realize that the SSFL site was at the center of the circle.
Once they realized that being close to SSFL could be their common thread, Ms. Bumstead and parents in her group began to gradually piece together the story, linking unusual or unexplained illnesses in their families to potential radiation or toxic chemical exposures from the lab.
“What really convinced me that this was absolutely a problem was when I learned about the epidemiological study by Dr. Hal Morgenstern that found that residents living within 2 miles of the Santa Susana Field Lab actually had a 60% higher cancer incidence rate and that over 1,500 workers have been diagnosed with cancer just from the Santa Susana Field Lab,” she told KCRW.
In 2015, Ms. Bumstead and other parents formed Parents Against Santa Susana Field Lab to hold SSFL site owner Boeing accountable for radiologic and toxic contamination and to ensure that Boeing cleans the site and surrounding areas. The group “seeks to reduce, to the greatest extent possible, the number of local families who have to hear the words, ‘Your child has cancer.’ ”
No longer quite so rare
Dr. Morgenstern, now retired from the University of Michigan, declined to be interviewed for this article. But as he and colleagues reported to the Centers for Disease Control and Prevention’s Agency for Toxic Substances and Disease Registry in 2007, there were strong signs of a link between contamination of the site and cancer.
The researchers compared cancer rates of adults living within 2 miles and 2-5 miles from SSFL with those of adults living more than 5 miles away, in Ventura and Los Angeles counties. They found that from 1988 through 1995, residents living within 2 miles of SSFL had a 60% higher rate of cancers than the control group. These included cancers of the thyroid, oral and nasal cavities, pharynx, larynx, esophagus, and bladder, as well as blood cancers such as leukemia, lymphoma, and multiple myeloma.
In separate studies, the investigators found higher rates of certain cancers among workers at SSFL who were exposed to radiation and to hydrazine, a chemical in rocket fuel.
In an interview, Dr. Saro Armenian, a pediatric hematologist-oncologist who was not involved in the studies, said the 60% increase in cancer incidence, which translated into a 1.6-fold increase in risk, merits more investigation.
“In epidemiologic studies, a 1.6-fold risk is actually a pretty strong signal because typically, most signals that you get are somewhere around 1.1- to 1.2-fold increased risk,” noted Dr. Armenian, a specialist in pediatric cancer survivorship and outcomes at City of Hope National Medical Center in Duarte, Calif.
However, Dr. Thomas Mack, former director of the Los Angeles County Cancer Surveillance Program, contends that there is insufficient evidence to support a direct link between the 1959 reactor accident and recent incident cancers. Dr. Mack is currently a professor of preventive medicine and pathology at the University of Southern California in Los Angeles.
“I have evaluated concerns about local excesses of cancer at least 100 times, usually from county residents, but for a while I represented the CDC and the California cancer registry,” Dr. Mack said, in response to an emailed request for comment.
“So far I have seen no evidence of carcinogenic radionucleotides or chemical carcinogens from Santa Susana found in any meaningful amount in any nearby community, but if someone has such evidence that would constitute evidence, that needs a response,” Dr. Mack added.
Boeing and California
Boeing has said problems at SSFL were not responsible for the high cancer rates among children in the community.
In April 2007, in a statement opposing a bill before the California State Legislature that would compel Boeing to pay for SSFL site cleanup, the company said that “in contrast to the accusations made against The Boeing Company that falsely claim increased cancer rates in the communities surrounding SSFL, a recent study conducted by the University of Michigan School of Public Health just concluded the opposite.”
Yet as Dr. Morgenstern wrote in 2007 to California state Sen. Joe Simitian, then chair of the Committee on Environmental Quality: “For the period 1996 through 2002, we found that the incidence rate of thyroid cancer was more than 60% greater among residents living within 2 miles of SSFL than for residents living more than 5 miles from SSFL. The magnitude and consistency of the thyroid finding for both periods is especially provocative because of evidence from other studies linking thyroid cancer with environmental exposures originating at SSFL and found in the surrounding communities.”
Boeing chose to ignore the results and instead focused on the methods used in the study, where the authors acknowledged that they measured distance from the site rather than environmental exposures and thus could not conclusively link excess cancer rates to exposures arising from SSFL.
But Dr. Morgenstern emphasized the conclusion of the report: “Despite the methodologic limitations of this study, the findings suggest there may be elevated incidence rates of certain cancers near SSFL that have been linked in previous studies with hazardous substances used at Rocketdyne, some of which have been observed or projected to exist offsite.”
Failure to come clean
In 2008, a law that set standards for cleanup of the site was passed. But the law was overturned in 2014 after a legal challenge by Boeing.
That left in place a 2007 order of consent between Boeing, NASA, the U.S. Department of Energy, and the California Department of Toxic Substances Control (DTSC) that required cleanup of SSFL to a much less stringent standard.
As of last year, Boeing and DTSC had begun confidential, nonbinding agreements regarding the 2007 order of consent, according to Parents Against SSFL.
Among the contaminants lingering at the site are radioactive particles, chemical compounds, heavy metals, and polluted water.
“In fact, over 300 contaminants of concern have been found at the site, and they are refusing to clean it,” Mr. Hirsch said. “This company releases large amounts of carcinogens, and perhaps significant numbers of people get sick with cancer, and the company doesn’t go to prison. They get more federal contracts.”
A version of this article first appeared on WebMD.com.
April 20, 2022 – Editor’s note: This article has been updated to include an interview with Dr. Thomas Mack, former director of the Los Angeles County Cancer Surveillance Program, who contends that there is insufficient evidence to support a direct link between the 1959 reactor accident and recent incident cancers.
Chernobyl. Fukushima. Three Mile Island.
The world knows these names all too well because of accidents there: complete or partial meltdowns of nuclear reactors that released massive amounts of cancer-causing radiation into the air, soil, and water.
The Santa Susana Field Lab is far less well-known, but no less infamous for what took place at this former rocket engine and nuclear energy test site just 28 miles northwest of downtown Los Angeles.
In July 1959, an accident involving one of 10 experimental nuclear reactors at the SSFL site released a cloud of harmful radiation and toxic chemicals over the surrounding area, including Simi Valley, San Gabriel Valley, Chatsworth, and Canoga Park. The small reactor had no containment vessel.
This accident resulted in a release of radioactive iodine estimated to be as much as 250 times that of the partial meltdown that would occur 2 decades later at Three Mile Island, a much larger commercial reactor that had a containment vessel.
Decades-long cover-up
In 1959, the public knew nothing about what happened at the site.
According to John Pace, then an employee at SSFL, the accident was covered up. Mr. Pace recounted the cover-up in the documentary “In the Dark of the Valley,” which first aired in November 2021 on MSNBC.
In fact, the accident at SSFL remained under wraps for 2 decades, according to Daniel Hirsch, former director of the Program on Environmental and Nuclear Policy at the University of California, Santa Cruz, and now president of Committee to Bridge the Gap, a nuclear policy nongovernmental organization.
“Students working with me while I was teaching at UCLA in 1979 uncovered these Atomic Energy Commission reports from Atomics International,” he said in an interview. “We had to order the documents from the annex to the UCLA Engineering Library. They were stored offsite, and it took a few days, and when we got them, we opened them up, and there were these fold-out photographs of the fuel [rods]. As we folded out the photographs further, we saw one photo with an arrow labeled ‘longitudinal cracks,’ and then other arrows showing other kinds of cracks, and then another arrow labeled ‘melted blob.’ ”
Mr. Hirsch and his students found that other accidents had occurred at SSFL, including a fuel fabrication system that leached plutonium, fires in a “hot” lab where irradiated nuclear fuel from around the United States was handled, and open-air burn pits where radioactive and toxic chemical wastes were illegally torched.
According to the Committee to Bridge the Gap, when the 2,800-acre SSFL site was being developed under the name Rocketdyne by aircraft maker North American Aviation, the area was sparsely populated, with nearly as many grazing animals as people in its hills and valleys.
North American Aviation later became part of Rockwell International, which in turn sold its aerospace and defense business units to the Boeing Company in 1996. Boeing, now in charge of the site and the cleanup efforts, is doing everything in its power to shirk or diminish its responsibility, Mr. Hirsch and other critics say.
Parents against SSFL
Today, more than 150,000 people live within 5 miles of SSFL, and more than half a million live within 10 miles.
Melissa Bumstead is one of those residents. She and her family live 3.7 miles from the Santa Susana site. When her toddler Grace was diagnosed with a rare form of leukemia in 2014, doctors told Ms. Bumstead there were no known links between her daughter’s cancer and environmental contamination.
But during Grace’s treatment at Children’s Hospital Los Angeles, her mother began meeting other parents who lived near her and had children facing equally rare cancers.
Lauren Hammersley, whose daughter Hazel was diagnosed with a rare brain tumor called neuroblastoma at age 2, lived about 10 miles from Ms. Bumstead on the other side of a mountain and just over 4 miles from SSFL.
On her street alone, Ms. Bumstead discovered three cases of pediatric cancer, including two children in adjacent homes who had the same rare brain tumor as Hazel Hammersley.
As Ms. Bumstead told Los Angeles National Public Radio station KCRW in 2021, “I started to panic because I knew that childhood cancer is extremely rare. There’s only 15,000 new cases every year out of 72 million children in America. So, the chance of knowing your neighbors, especially at an internationally renowned hospital like Children’s Hospital Los Angeles – we knew something wasn’t right.”
After a relapse of her tumor, Hazel died in 2018, a few months after her seventh birthday.
Cancer clusters
Hoping to understand why their kids were getting so sick, Ms. Bumstead and the other parents formed a Facebook group. They plotted their homes on Google Maps and found that they all lived within roughly 10 miles of one another. It would take another year for them to realize that the SSFL site was at the center of the circle.
Once they realized that being close to SSFL could be their common thread, Ms. Bumstead and parents in her group began to gradually piece together the story, linking unusual or unexplained illnesses in their families to potential radiation or toxic chemical exposures from the lab.
“What really convinced me that this was absolutely a problem was when I learned about the epidemiological study by Dr. Hal Morgenstern that found that residents living within 2 miles of the Santa Susana Field Lab actually had a 60% higher cancer incidence rate and that over 1,500 workers have been diagnosed with cancer just from the Santa Susana Field Lab,” she told KCRW.
In 2015, Ms. Bumstead and other parents formed Parents Against Santa Susana Field Lab to hold SSFL site owner Boeing accountable for radiologic and toxic contamination and to ensure that Boeing cleans the site and surrounding areas. The group “seeks to reduce, to the greatest extent possible, the number of local families who have to hear the words, ‘Your child has cancer.’ ”
No longer quite so rare
Dr. Morgenstern, now retired from the University of Michigan, declined to be interviewed for this article. But as he and colleagues reported to the Centers for Disease Control and Prevention’s Agency for Toxic Substances and Disease Registry in 2007, there were strong signs of a link between contamination of the site and cancer.
The researchers compared cancer rates of adults living within 2 miles and 2-5 miles from SSFL with those of adults living more than 5 miles away, in Ventura and Los Angeles counties. They found that from 1988 through 1995, residents living within 2 miles of SSFL had a 60% higher rate of cancers than the control group. These included cancers of the thyroid, oral and nasal cavities, pharynx, larynx, esophagus, and bladder, as well as blood cancers such as leukemia, lymphoma, and multiple myeloma.
In separate studies, the investigators found higher rates of certain cancers among workers at SSFL who were exposed to radiation and to hydrazine, a chemical in rocket fuel.
In an interview, Dr. Saro Armenian, a pediatric hematologist-oncologist who was not involved in the studies, said the 60% increase in cancer incidence, which translated into a 1.6-fold increase in risk, merits more investigation.
“In epidemiologic studies, a 1.6-fold risk is actually a pretty strong signal because typically, most signals that you get are somewhere around 1.1- to 1.2-fold increased risk,” noted Dr. Armenian, a specialist in pediatric cancer survivorship and outcomes at City of Hope National Medical Center in Duarte, Calif.
However, Dr. Thomas Mack, former director of the Los Angeles County Cancer Surveillance Program, contends that there is insufficient evidence to support a direct link between the 1959 reactor accident and recent incident cancers. Dr. Mack is currently a professor of preventive medicine and pathology at the University of Southern California in Los Angeles.
“I have evaluated concerns about local excesses of cancer at least 100 times, usually from county residents, but for a while I represented the CDC and the California cancer registry,” Dr. Mack said, in response to an emailed request for comment.
“So far I have seen no evidence of carcinogenic radionucleotides or chemical carcinogens from Santa Susana found in any meaningful amount in any nearby community, but if someone has such evidence that would constitute evidence, that needs a response,” Dr. Mack added.
Boeing and California
Boeing has said problems at SSFL were not responsible for the high cancer rates among children in the community.
In April 2007, in a statement opposing a bill before the California State Legislature that would compel Boeing to pay for SSFL site cleanup, the company said that “in contrast to the accusations made against The Boeing Company that falsely claim increased cancer rates in the communities surrounding SSFL, a recent study conducted by the University of Michigan School of Public Health just concluded the opposite.”
Yet as Dr. Morgenstern wrote in 2007 to California state Sen. Joe Simitian, then chair of the Committee on Environmental Quality: “For the period 1996 through 2002, we found that the incidence rate of thyroid cancer was more than 60% greater among residents living within 2 miles of SSFL than for residents living more than 5 miles from SSFL. The magnitude and consistency of the thyroid finding for both periods is especially provocative because of evidence from other studies linking thyroid cancer with environmental exposures originating at SSFL and found in the surrounding communities.”
Boeing chose to ignore the results and instead focused on the methods used in the study, where the authors acknowledged that they measured distance from the site rather than environmental exposures and thus could not conclusively link excess cancer rates to exposures arising from SSFL.
But Dr. Morgenstern emphasized the conclusion of the report: “Despite the methodologic limitations of this study, the findings suggest there may be elevated incidence rates of certain cancers near SSFL that have been linked in previous studies with hazardous substances used at Rocketdyne, some of which have been observed or projected to exist offsite.”
Failure to come clean
In 2008, a law that set standards for cleanup of the site was passed. But the law was overturned in 2014 after a legal challenge by Boeing.
That left in place a 2007 order of consent between Boeing, NASA, the U.S. Department of Energy, and the California Department of Toxic Substances Control (DTSC) that required cleanup of SSFL to a much less stringent standard.
As of last year, Boeing and DTSC had begun confidential, nonbinding agreements regarding the 2007 order of consent, according to Parents Against SSFL.
Among the contaminants lingering at the site are radioactive particles, chemical compounds, heavy metals, and polluted water.
“In fact, over 300 contaminants of concern have been found at the site, and they are refusing to clean it,” Mr. Hirsch said. “This company releases large amounts of carcinogens, and perhaps significant numbers of people get sick with cancer, and the company doesn’t go to prison. They get more federal contracts.”
A version of this article first appeared on WebMD.com.
April 20, 2022 – Editor’s note: This article has been updated to include an interview with Dr. Thomas Mack, former director of the Los Angeles County Cancer Surveillance Program, who contends that there is insufficient evidence to support a direct link between the 1959 reactor accident and recent incident cancers.
Chernobyl. Fukushima. Three Mile Island.
The world knows these names all too well because of accidents there: complete or partial meltdowns of nuclear reactors that released massive amounts of cancer-causing radiation into the air, soil, and water.
The Santa Susana Field Lab is far less well-known, but no less infamous for what took place at this former rocket engine and nuclear energy test site just 28 miles northwest of downtown Los Angeles.
In July 1959, an accident involving one of 10 experimental nuclear reactors at the SSFL site released a cloud of harmful radiation and toxic chemicals over the surrounding area, including Simi Valley, San Gabriel Valley, Chatsworth, and Canoga Park. The small reactor had no containment vessel.
This accident resulted in a release of radioactive iodine estimated to be as much as 250 times that of the partial meltdown that would occur 2 decades later at Three Mile Island, a much larger commercial reactor that had a containment vessel.
Decades-long cover-up
In 1959, the public knew nothing about what happened at the site.
According to John Pace, then an employee at SSFL, the accident was covered up. Mr. Pace recounted the cover-up in the documentary “In the Dark of the Valley,” which first aired in November 2021 on MSNBC.
In fact, the accident at SSFL remained under wraps for 2 decades, according to Daniel Hirsch, former director of the Program on Environmental and Nuclear Policy at the University of California, Santa Cruz, and now president of Committee to Bridge the Gap, a nuclear policy nongovernmental organization.
“Students working with me while I was teaching at UCLA in 1979 uncovered these Atomic Energy Commission reports from Atomics International,” he said in an interview. “We had to order the documents from the annex to the UCLA Engineering Library. They were stored offsite, and it took a few days, and when we got them, we opened them up, and there were these fold-out photographs of the fuel [rods]. As we folded out the photographs further, we saw one photo with an arrow labeled ‘longitudinal cracks,’ and then other arrows showing other kinds of cracks, and then another arrow labeled ‘melted blob.’ ”
Mr. Hirsch and his students found that other accidents had occurred at SSFL, including a fuel fabrication system that leached plutonium, fires in a “hot” lab where irradiated nuclear fuel from around the United States was handled, and open-air burn pits where radioactive and toxic chemical wastes were illegally torched.
According to the Committee to Bridge the Gap, when the 2,800-acre SSFL site was being developed under the name Rocketdyne by aircraft maker North American Aviation, the area was sparsely populated, with nearly as many grazing animals as people in its hills and valleys.
North American Aviation later became part of Rockwell International, which in turn sold its aerospace and defense business units to the Boeing Company in 1996. Boeing, now in charge of the site and the cleanup efforts, is doing everything in its power to shirk or diminish its responsibility, Mr. Hirsch and other critics say.
Parents against SSFL
Today, more than 150,000 people live within 5 miles of SSFL, and more than half a million live within 10 miles.
Melissa Bumstead is one of those residents. She and her family live 3.7 miles from the Santa Susana site. When her toddler Grace was diagnosed with a rare form of leukemia in 2014, doctors told Ms. Bumstead there were no known links between her daughter’s cancer and environmental contamination.
But during Grace’s treatment at Children’s Hospital Los Angeles, her mother began meeting other parents who lived near her and had children facing equally rare cancers.
Lauren Hammersley, whose daughter Hazel was diagnosed with a rare brain tumor called neuroblastoma at age 2, lived about 10 miles from Ms. Bumstead on the other side of a mountain and just over 4 miles from SSFL.
On her street alone, Ms. Bumstead discovered three cases of pediatric cancer, including two children in adjacent homes who had the same rare brain tumor as Hazel Hammersley.
As Ms. Bumstead told Los Angeles National Public Radio station KCRW in 2021, “I started to panic because I knew that childhood cancer is extremely rare. There’s only 15,000 new cases every year out of 72 million children in America. So, the chance of knowing your neighbors, especially at an internationally renowned hospital like Children’s Hospital Los Angeles – we knew something wasn’t right.”
After a relapse of her tumor, Hazel died in 2018, a few months after her seventh birthday.
Cancer clusters
Hoping to understand why their kids were getting so sick, Ms. Bumstead and the other parents formed a Facebook group. They plotted their homes on Google Maps and found that they all lived within roughly 10 miles of one another. It would take another year for them to realize that the SSFL site was at the center of the circle.
Once they realized that being close to SSFL could be their common thread, Ms. Bumstead and parents in her group began to gradually piece together the story, linking unusual or unexplained illnesses in their families to potential radiation or toxic chemical exposures from the lab.
“What really convinced me that this was absolutely a problem was when I learned about the epidemiological study by Dr. Hal Morgenstern that found that residents living within 2 miles of the Santa Susana Field Lab actually had a 60% higher cancer incidence rate and that over 1,500 workers have been diagnosed with cancer just from the Santa Susana Field Lab,” she told KCRW.
In 2015, Ms. Bumstead and other parents formed Parents Against Santa Susana Field Lab to hold SSFL site owner Boeing accountable for radiologic and toxic contamination and to ensure that Boeing cleans the site and surrounding areas. The group “seeks to reduce, to the greatest extent possible, the number of local families who have to hear the words, ‘Your child has cancer.’ ”
No longer quite so rare
Dr. Morgenstern, now retired from the University of Michigan, declined to be interviewed for this article. But as he and colleagues reported to the Centers for Disease Control and Prevention’s Agency for Toxic Substances and Disease Registry in 2007, there were strong signs of a link between contamination of the site and cancer.
The researchers compared cancer rates of adults living within 2 miles and 2-5 miles from SSFL with those of adults living more than 5 miles away, in Ventura and Los Angeles counties. They found that from 1988 through 1995, residents living within 2 miles of SSFL had a 60% higher rate of cancers than the control group. These included cancers of the thyroid, oral and nasal cavities, pharynx, larynx, esophagus, and bladder, as well as blood cancers such as leukemia, lymphoma, and multiple myeloma.
In separate studies, the investigators found higher rates of certain cancers among workers at SSFL who were exposed to radiation and to hydrazine, a chemical in rocket fuel.
In an interview, Dr. Saro Armenian, a pediatric hematologist-oncologist who was not involved in the studies, said the 60% increase in cancer incidence, which translated into a 1.6-fold increase in risk, merits more investigation.
“In epidemiologic studies, a 1.6-fold risk is actually a pretty strong signal because typically, most signals that you get are somewhere around 1.1- to 1.2-fold increased risk,” noted Dr. Armenian, a specialist in pediatric cancer survivorship and outcomes at City of Hope National Medical Center in Duarte, Calif.
However, Dr. Thomas Mack, former director of the Los Angeles County Cancer Surveillance Program, contends that there is insufficient evidence to support a direct link between the 1959 reactor accident and recent incident cancers. Dr. Mack is currently a professor of preventive medicine and pathology at the University of Southern California in Los Angeles.
“I have evaluated concerns about local excesses of cancer at least 100 times, usually from county residents, but for a while I represented the CDC and the California cancer registry,” Dr. Mack said, in response to an emailed request for comment.
“So far I have seen no evidence of carcinogenic radionucleotides or chemical carcinogens from Santa Susana found in any meaningful amount in any nearby community, but if someone has such evidence that would constitute evidence, that needs a response,” Dr. Mack added.
Boeing and California
Boeing has said problems at SSFL were not responsible for the high cancer rates among children in the community.
In April 2007, in a statement opposing a bill before the California State Legislature that would compel Boeing to pay for SSFL site cleanup, the company said that “in contrast to the accusations made against The Boeing Company that falsely claim increased cancer rates in the communities surrounding SSFL, a recent study conducted by the University of Michigan School of Public Health just concluded the opposite.”
Yet as Dr. Morgenstern wrote in 2007 to California state Sen. Joe Simitian, then chair of the Committee on Environmental Quality: “For the period 1996 through 2002, we found that the incidence rate of thyroid cancer was more than 60% greater among residents living within 2 miles of SSFL than for residents living more than 5 miles from SSFL. The magnitude and consistency of the thyroid finding for both periods is especially provocative because of evidence from other studies linking thyroid cancer with environmental exposures originating at SSFL and found in the surrounding communities.”
Boeing chose to ignore the results and instead focused on the methods used in the study, where the authors acknowledged that they measured distance from the site rather than environmental exposures and thus could not conclusively link excess cancer rates to exposures arising from SSFL.
But Dr. Morgenstern emphasized the conclusion of the report: “Despite the methodologic limitations of this study, the findings suggest there may be elevated incidence rates of certain cancers near SSFL that have been linked in previous studies with hazardous substances used at Rocketdyne, some of which have been observed or projected to exist offsite.”
Failure to come clean
In 2008, a law that set standards for cleanup of the site was passed. But the law was overturned in 2014 after a legal challenge by Boeing.
That left in place a 2007 order of consent between Boeing, NASA, the U.S. Department of Energy, and the California Department of Toxic Substances Control (DTSC) that required cleanup of SSFL to a much less stringent standard.
As of last year, Boeing and DTSC had begun confidential, nonbinding agreements regarding the 2007 order of consent, according to Parents Against SSFL.
Among the contaminants lingering at the site are radioactive particles, chemical compounds, heavy metals, and polluted water.
“In fact, over 300 contaminants of concern have been found at the site, and they are refusing to clean it,” Mr. Hirsch said. “This company releases large amounts of carcinogens, and perhaps significant numbers of people get sick with cancer, and the company doesn’t go to prison. They get more federal contracts.”
A version of this article first appeared on WebMD.com.
April 20, 2022 – Editor’s note: This article has been updated to include an interview with Dr. Thomas Mack, former director of the Los Angeles County Cancer Surveillance Program, who contends that there is insufficient evidence to support a direct link between the 1959 reactor accident and recent incident cancers.
Complex link between gut microbiome and immunotherapy response in advanced melanoma
A large-scale than previously thought.
Overall, researchers identified a panel of species, including Roseburia spp. and Akkermansia muciniphila, associated with responses to ICI therapy. However, no single species was a “fully consistent biomarker” across the studies, the authors explain.
This “machine learning analysis confirmed the link between the microbiome and overall response rates (ORRs) and progression-free survival (PFS) with ICIs but also revealed limited reproducibility of microbiome-based signatures across cohorts,” Karla A. Lee, PhD, a clinical research fellow at King’s College London, and colleagues report. The results suggest that “the microbiome is predictive of response in some, but not all, cohorts.”
The findings were published online Feb. 28 in Nature Medicine.
Despite recent advances in targeted therapies for melanoma, less than half of the those who receive a single-agent ICI respond, and those who receive combination ICI therapy often suffer from severe drug toxicity problems. That is why finding patients more likely to respond to a single-agent ICI has become a priority.
Previous studies have identified the gut microbiome as “a potential biomarker of response, as well as a therapeutic target” in melanoma and other malignancies, but “little consensus exists on which microbiome characteristics are associated with treatment responses in the human setting,” the authors explain.
To further clarify the microbiome–immunotherapy relationship, the researchers performed metagenomic sequencing of stool samples collected from 165 ICI-naive patients with unresectable stage III or IV cutaneous melanoma from 5 observational cohorts in the Netherlands, United Kingdom, and Spain. These data were integrated with 147 samples from publicly available datasets.
First, the authors highlighted the variability in findings across these observational studies. For instance, they analyzed stool samples from one UK-based observational study of patients with melanoma (PRIMM-UK) and found a small but statistically significant difference in the microbiome composition of immunotherapy responders versus nonresponders (P = .05) but did not find such an association in a parallel study in the Netherlands (PRIMM-NL, P = .61).
The investigators also explored biomarkers of response across different cohorts and found several standouts. In trials using ORR as an endpoint, two uncultivated Roseburia species (CAG:182 and CAG:471) were associated with responses to ICIs. For patients with available PFS data, Phascolarctobacterium succinatutens and Lactobacillus vaginalis were “enriched in responders” across 7 datasets and significant in 3 of the 8 meta-analysis approaches. A muciniphila and Dorea formicigenerans were also associated with ORR and PFS at 12 months in several meta-analyses.
However, “no single bacterium was a fully consistent biomarker of response across all datasets,” the authors wrote.
Still, the findings could have important implications for the more than 50% of patients with advanced melanoma who don’t respond to single-agent ICI therapy.
“Our study shows that studying the microbiome is important to improve and personalize immunotherapy treatments for melanoma,” study coauthor Nicola Segata, PhD, principal investigator in the Laboratory of Computational Metagenomics, University of Trento, Italy, said in a press release. “However, it also suggests that because of the person-to-person variability of the gut microbiome, even larger studies must be carried out to understand the specific gut microbial features that are more likely to lead to a positive response to immunotherapy.”
Coauthor Tim Spector, PhD, head of the Department of Twin Research & Genetic Epidemiology at King’s College London, added that “the ultimate goal is to identify which specific features of the microbiome are directly influencing the clinical benefits of immunotherapy to exploit these features in new personalized approaches to support cancer immunotherapy.”
In the meantime, he said, “this study highlights the potential impact of good diet and gut health on chances of survival in patients undergoing immunotherapy.”
This study was coordinated by King’s College London, CIBIO Department of the University of Trento and European Institute of Oncology in Italy, and the University of Groningen in the Netherlands, and was funded by the Seerave Foundation. Dr. Lee, Dr. Segata, and Dr. Spector have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A large-scale than previously thought.
Overall, researchers identified a panel of species, including Roseburia spp. and Akkermansia muciniphila, associated with responses to ICI therapy. However, no single species was a “fully consistent biomarker” across the studies, the authors explain.
This “machine learning analysis confirmed the link between the microbiome and overall response rates (ORRs) and progression-free survival (PFS) with ICIs but also revealed limited reproducibility of microbiome-based signatures across cohorts,” Karla A. Lee, PhD, a clinical research fellow at King’s College London, and colleagues report. The results suggest that “the microbiome is predictive of response in some, but not all, cohorts.”
The findings were published online Feb. 28 in Nature Medicine.
Despite recent advances in targeted therapies for melanoma, less than half of the those who receive a single-agent ICI respond, and those who receive combination ICI therapy often suffer from severe drug toxicity problems. That is why finding patients more likely to respond to a single-agent ICI has become a priority.
Previous studies have identified the gut microbiome as “a potential biomarker of response, as well as a therapeutic target” in melanoma and other malignancies, but “little consensus exists on which microbiome characteristics are associated with treatment responses in the human setting,” the authors explain.
To further clarify the microbiome–immunotherapy relationship, the researchers performed metagenomic sequencing of stool samples collected from 165 ICI-naive patients with unresectable stage III or IV cutaneous melanoma from 5 observational cohorts in the Netherlands, United Kingdom, and Spain. These data were integrated with 147 samples from publicly available datasets.
First, the authors highlighted the variability in findings across these observational studies. For instance, they analyzed stool samples from one UK-based observational study of patients with melanoma (PRIMM-UK) and found a small but statistically significant difference in the microbiome composition of immunotherapy responders versus nonresponders (P = .05) but did not find such an association in a parallel study in the Netherlands (PRIMM-NL, P = .61).
The investigators also explored biomarkers of response across different cohorts and found several standouts. In trials using ORR as an endpoint, two uncultivated Roseburia species (CAG:182 and CAG:471) were associated with responses to ICIs. For patients with available PFS data, Phascolarctobacterium succinatutens and Lactobacillus vaginalis were “enriched in responders” across 7 datasets and significant in 3 of the 8 meta-analysis approaches. A muciniphila and Dorea formicigenerans were also associated with ORR and PFS at 12 months in several meta-analyses.
However, “no single bacterium was a fully consistent biomarker of response across all datasets,” the authors wrote.
Still, the findings could have important implications for the more than 50% of patients with advanced melanoma who don’t respond to single-agent ICI therapy.
“Our study shows that studying the microbiome is important to improve and personalize immunotherapy treatments for melanoma,” study coauthor Nicola Segata, PhD, principal investigator in the Laboratory of Computational Metagenomics, University of Trento, Italy, said in a press release. “However, it also suggests that because of the person-to-person variability of the gut microbiome, even larger studies must be carried out to understand the specific gut microbial features that are more likely to lead to a positive response to immunotherapy.”
Coauthor Tim Spector, PhD, head of the Department of Twin Research & Genetic Epidemiology at King’s College London, added that “the ultimate goal is to identify which specific features of the microbiome are directly influencing the clinical benefits of immunotherapy to exploit these features in new personalized approaches to support cancer immunotherapy.”
In the meantime, he said, “this study highlights the potential impact of good diet and gut health on chances of survival in patients undergoing immunotherapy.”
This study was coordinated by King’s College London, CIBIO Department of the University of Trento and European Institute of Oncology in Italy, and the University of Groningen in the Netherlands, and was funded by the Seerave Foundation. Dr. Lee, Dr. Segata, and Dr. Spector have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A large-scale than previously thought.
Overall, researchers identified a panel of species, including Roseburia spp. and Akkermansia muciniphila, associated with responses to ICI therapy. However, no single species was a “fully consistent biomarker” across the studies, the authors explain.
This “machine learning analysis confirmed the link between the microbiome and overall response rates (ORRs) and progression-free survival (PFS) with ICIs but also revealed limited reproducibility of microbiome-based signatures across cohorts,” Karla A. Lee, PhD, a clinical research fellow at King’s College London, and colleagues report. The results suggest that “the microbiome is predictive of response in some, but not all, cohorts.”
The findings were published online Feb. 28 in Nature Medicine.
Despite recent advances in targeted therapies for melanoma, less than half of the those who receive a single-agent ICI respond, and those who receive combination ICI therapy often suffer from severe drug toxicity problems. That is why finding patients more likely to respond to a single-agent ICI has become a priority.
Previous studies have identified the gut microbiome as “a potential biomarker of response, as well as a therapeutic target” in melanoma and other malignancies, but “little consensus exists on which microbiome characteristics are associated with treatment responses in the human setting,” the authors explain.
To further clarify the microbiome–immunotherapy relationship, the researchers performed metagenomic sequencing of stool samples collected from 165 ICI-naive patients with unresectable stage III or IV cutaneous melanoma from 5 observational cohorts in the Netherlands, United Kingdom, and Spain. These data were integrated with 147 samples from publicly available datasets.
First, the authors highlighted the variability in findings across these observational studies. For instance, they analyzed stool samples from one UK-based observational study of patients with melanoma (PRIMM-UK) and found a small but statistically significant difference in the microbiome composition of immunotherapy responders versus nonresponders (P = .05) but did not find such an association in a parallel study in the Netherlands (PRIMM-NL, P = .61).
The investigators also explored biomarkers of response across different cohorts and found several standouts. In trials using ORR as an endpoint, two uncultivated Roseburia species (CAG:182 and CAG:471) were associated with responses to ICIs. For patients with available PFS data, Phascolarctobacterium succinatutens and Lactobacillus vaginalis were “enriched in responders” across 7 datasets and significant in 3 of the 8 meta-analysis approaches. A muciniphila and Dorea formicigenerans were also associated with ORR and PFS at 12 months in several meta-analyses.
However, “no single bacterium was a fully consistent biomarker of response across all datasets,” the authors wrote.
Still, the findings could have important implications for the more than 50% of patients with advanced melanoma who don’t respond to single-agent ICI therapy.
“Our study shows that studying the microbiome is important to improve and personalize immunotherapy treatments for melanoma,” study coauthor Nicola Segata, PhD, principal investigator in the Laboratory of Computational Metagenomics, University of Trento, Italy, said in a press release. “However, it also suggests that because of the person-to-person variability of the gut microbiome, even larger studies must be carried out to understand the specific gut microbial features that are more likely to lead to a positive response to immunotherapy.”
Coauthor Tim Spector, PhD, head of the Department of Twin Research & Genetic Epidemiology at King’s College London, added that “the ultimate goal is to identify which specific features of the microbiome are directly influencing the clinical benefits of immunotherapy to exploit these features in new personalized approaches to support cancer immunotherapy.”
In the meantime, he said, “this study highlights the potential impact of good diet and gut health on chances of survival in patients undergoing immunotherapy.”
This study was coordinated by King’s College London, CIBIO Department of the University of Trento and European Institute of Oncology in Italy, and the University of Groningen in the Netherlands, and was funded by the Seerave Foundation. Dr. Lee, Dr. Segata, and Dr. Spector have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA approves new CAR T-cell treatment for multiple myeloma
A new treatment option for patients with refractory/relapsed multiple myeloma who have already tried four or more therapies has been approved by the U.S. Food and Drug Administration.
There are already two other therapies on the market that target BCMA – another CAR T cell, idecabtagene vicleucel (Abecma), which was approved by the FDA in March 2021, and a drug conjugate, belantamab mafodotin (Blenrep), which was approved in August 2020.
The approval of cilta-cel was based on clinical data from the CARTITUDE-1 study, which were initially presented in December 2020 at the annual meeting of the American Society of Hematology, as reported at the time by this news organization.
The trial involved 97 patients with relapsed/refractory multiple myeloma who had already received a median of six previous treatments (range, three to 18), including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.
“The treatment journey for the majority of patients living with multiple myeloma is a relentless cycle of remission and relapse, with fewer patients achieving a deep response as they progress through later lines of therapy,” commented Sundar Jagannath, MBBS, professor of medicine, hematology, and medical oncology at Mount Sinai, who was a principal investigator on the pivotal study.
“That is why I have been really excited about the results from the CARTITUDE-1 study, which has demonstrated that cilta-cel can provide deep and durable responses and long-term treatment-free intervals, even in this heavily pretreated multiple myeloma patient population,” he said.
“Today’s approval of Carvykti helps address a great unmet need for these patients,” he commented in a press release from the manufacturer.
Like other CAR T-cell therapies, ciltacabtagene autoleucel is a one-time treatment. It involves collecting blood from the patient, extracting T cells, genetically engineering them, then transfusing them back to the patient, who in the meantime has undergone conditioning.
The results from CARTITUDE-1 show that this one-time treatment resulted in deep and durable responses.
The overall response rate was 98%, and the majority of patients (78%) achieved a stringent complete response, in which physicians are unable to observe any signs or symptoms of disease via imaging or other tests after treatment.
At a median of 18 months’ follow-up, the median duration of response was 21.8 months.
“The responses in the CARTITUDE-1 study showed durability over time and resulted in the majority of heavily pretreated patients achieving deep responses after 18-month follow-up,” commented Mr. Jagannath.
“The approval of cilta-cel provides physicians an immunotherapy treatment option that offers patients an opportunity to be free from anti-myeloma therapies for a period of time,” he added.
As with other CAR T-cell therapies, there were serious side effects, and these products are available only through restricted programs under a risk evaluation and mitigation strategy.
The product information for Cartykti includes a boxed warning that mentions cytokine release syndrome (CRS), immune effector cell–associated neurotoxicity syndrome, parkinsonism, Guillain-Barré syndrome, hemophagocytic lymphohistiocytosis/macrophage activation syndrome, and prolonged and/or recurrent cytopenias.
The most common adverse reactions (reported in greater than or equal to 20% of patients) are pyrexia, CRS, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections–pathogens unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting.
A version of this article first appeared on Medscape.com.
A new treatment option for patients with refractory/relapsed multiple myeloma who have already tried four or more therapies has been approved by the U.S. Food and Drug Administration.
There are already two other therapies on the market that target BCMA – another CAR T cell, idecabtagene vicleucel (Abecma), which was approved by the FDA in March 2021, and a drug conjugate, belantamab mafodotin (Blenrep), which was approved in August 2020.
The approval of cilta-cel was based on clinical data from the CARTITUDE-1 study, which were initially presented in December 2020 at the annual meeting of the American Society of Hematology, as reported at the time by this news organization.
The trial involved 97 patients with relapsed/refractory multiple myeloma who had already received a median of six previous treatments (range, three to 18), including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.
“The treatment journey for the majority of patients living with multiple myeloma is a relentless cycle of remission and relapse, with fewer patients achieving a deep response as they progress through later lines of therapy,” commented Sundar Jagannath, MBBS, professor of medicine, hematology, and medical oncology at Mount Sinai, who was a principal investigator on the pivotal study.
“That is why I have been really excited about the results from the CARTITUDE-1 study, which has demonstrated that cilta-cel can provide deep and durable responses and long-term treatment-free intervals, even in this heavily pretreated multiple myeloma patient population,” he said.
“Today’s approval of Carvykti helps address a great unmet need for these patients,” he commented in a press release from the manufacturer.
Like other CAR T-cell therapies, ciltacabtagene autoleucel is a one-time treatment. It involves collecting blood from the patient, extracting T cells, genetically engineering them, then transfusing them back to the patient, who in the meantime has undergone conditioning.
The results from CARTITUDE-1 show that this one-time treatment resulted in deep and durable responses.
The overall response rate was 98%, and the majority of patients (78%) achieved a stringent complete response, in which physicians are unable to observe any signs or symptoms of disease via imaging or other tests after treatment.
At a median of 18 months’ follow-up, the median duration of response was 21.8 months.
“The responses in the CARTITUDE-1 study showed durability over time and resulted in the majority of heavily pretreated patients achieving deep responses after 18-month follow-up,” commented Mr. Jagannath.
“The approval of cilta-cel provides physicians an immunotherapy treatment option that offers patients an opportunity to be free from anti-myeloma therapies for a period of time,” he added.
As with other CAR T-cell therapies, there were serious side effects, and these products are available only through restricted programs under a risk evaluation and mitigation strategy.
The product information for Cartykti includes a boxed warning that mentions cytokine release syndrome (CRS), immune effector cell–associated neurotoxicity syndrome, parkinsonism, Guillain-Barré syndrome, hemophagocytic lymphohistiocytosis/macrophage activation syndrome, and prolonged and/or recurrent cytopenias.
The most common adverse reactions (reported in greater than or equal to 20% of patients) are pyrexia, CRS, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections–pathogens unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting.
A version of this article first appeared on Medscape.com.
A new treatment option for patients with refractory/relapsed multiple myeloma who have already tried four or more therapies has been approved by the U.S. Food and Drug Administration.
There are already two other therapies on the market that target BCMA – another CAR T cell, idecabtagene vicleucel (Abecma), which was approved by the FDA in March 2021, and a drug conjugate, belantamab mafodotin (Blenrep), which was approved in August 2020.
The approval of cilta-cel was based on clinical data from the CARTITUDE-1 study, which were initially presented in December 2020 at the annual meeting of the American Society of Hematology, as reported at the time by this news organization.
The trial involved 97 patients with relapsed/refractory multiple myeloma who had already received a median of six previous treatments (range, three to 18), including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.
“The treatment journey for the majority of patients living with multiple myeloma is a relentless cycle of remission and relapse, with fewer patients achieving a deep response as they progress through later lines of therapy,” commented Sundar Jagannath, MBBS, professor of medicine, hematology, and medical oncology at Mount Sinai, who was a principal investigator on the pivotal study.
“That is why I have been really excited about the results from the CARTITUDE-1 study, which has demonstrated that cilta-cel can provide deep and durable responses and long-term treatment-free intervals, even in this heavily pretreated multiple myeloma patient population,” he said.
“Today’s approval of Carvykti helps address a great unmet need for these patients,” he commented in a press release from the manufacturer.
Like other CAR T-cell therapies, ciltacabtagene autoleucel is a one-time treatment. It involves collecting blood from the patient, extracting T cells, genetically engineering them, then transfusing them back to the patient, who in the meantime has undergone conditioning.
The results from CARTITUDE-1 show that this one-time treatment resulted in deep and durable responses.
The overall response rate was 98%, and the majority of patients (78%) achieved a stringent complete response, in which physicians are unable to observe any signs or symptoms of disease via imaging or other tests after treatment.
At a median of 18 months’ follow-up, the median duration of response was 21.8 months.
“The responses in the CARTITUDE-1 study showed durability over time and resulted in the majority of heavily pretreated patients achieving deep responses after 18-month follow-up,” commented Mr. Jagannath.
“The approval of cilta-cel provides physicians an immunotherapy treatment option that offers patients an opportunity to be free from anti-myeloma therapies for a period of time,” he added.
As with other CAR T-cell therapies, there were serious side effects, and these products are available only through restricted programs under a risk evaluation and mitigation strategy.
The product information for Cartykti includes a boxed warning that mentions cytokine release syndrome (CRS), immune effector cell–associated neurotoxicity syndrome, parkinsonism, Guillain-Barré syndrome, hemophagocytic lymphohistiocytosis/macrophage activation syndrome, and prolonged and/or recurrent cytopenias.
The most common adverse reactions (reported in greater than or equal to 20% of patients) are pyrexia, CRS, hypogammaglobulinemia, hypotension, musculoskeletal pain, fatigue, infections–pathogens unspecified, cough, chills, diarrhea, nausea, encephalopathy, decreased appetite, upper respiratory tract infection, headache, tachycardia, dizziness, dyspnea, edema, viral infections, coagulopathy, constipation, and vomiting.
A version of this article first appeared on Medscape.com.