User login
Consider ultraslow thrombolysis for mechanical valve thrombosis
SNOWMASS, COLO. – Ultraslow infusion of a very-low-dose thrombolytic agent for treatment of mechanical prosthetic valve thrombosis appears to be as effective as surgery – the former first-line therapy – and sports a far lower stroke risk, Rick A. Nishimura, MD, said at the Annual Cardiovascular Conference at Snowmass.
“I’m not saying you have to use this, but I think it’s reasonable to consider it, especially if the patient is at high risk for surgery and low risk for thrombolysis,” according to Dr. Nishimura, professor of cardiovascular diseases and hypertension at the Mayo Clinic in Rochester, Minn.
He added that he and his Mayo colleagues have begun using the novel therapy and are favorably impressed with the resultant complete normalization of valve gradients and low complication rate.
Dr. Nishimura was cochair of the writing committee for the current American College of Cardiology/American Heart Association guidelines for management of valvular heart disease (Circulation. 2014 Jun 10;129[23]:e521-643). Those guidelines state that emergency surgery is the treatment of choice for thrombosis of a left-sided mechanical heart valve. That strong recommendation was based on a dozen nonrandomized studies reported prior to 2013 which showed a 95% success rate with surgery compared with 75% with conventional large-bolus thrombolytic therapy, a high 10%-12% mortality with either form of therapy, and a stroke risk of 12%-14% with thrombolytic therapy, substantially higher than for surgery.
Since release of the ACC/AHA guidelines, however, there’s been an important new development: Three groups outside of the United States have pioneered ultraslow thrombolytic therapy for mechanical prosthetic valve thrombosis. The supporting evidence comes from cohort studies, with no randomized trials done to date. But the collective reported experience from these three research teams shows a 90%-95% success rate – comparable to surgery – along with stroke and mortality rates in the low single digits.
The Turkish group waits until the patient’s international normalized ratio (INR) is below 2.5, then administers 25 mg of tissue plasminogen activator guided by transesophageal echocardiography (TEE) over 25 hours.
“We traditionally give 90 mg over 1 hour, so this is very, very slow therapy,” Dr. Nishimura observed.
After the 24-hour infusion, TEE is repeated. If imaging shows the clot is not resolved, another 25 mg of tissue plasminogen activator is given over 24 hours. This process is repeated for up to 8 days as needed (Am Heart J. 2015 Aug;170[2]:409-18).
Dr. Nishimura advised reserving ultraslow thrombolytic therapy for patients who are hemodynamically stable; this treatment takes a while to work, so patients in severe heart failure should be sent straight away to surgery. The novel therapy is best suited for patients with recent-onset mechanical valve thrombosis, a low INR, TEE evidence that the clot isn’t huge, and/or when surgical expertise isn’t readily available.
Before you can treat a prosthetic mechanical valve thrombosis, however, you have to make the diagnosis. Here’s what Dr. Nishimura recommends: First, suspect the condition on the basis of clinical symptoms of heart failure and dull, muffled S1 and S2 sounds on auscultation, especially in a patient who presents with a low INR.
Next, prove that obstruction is present via Doppler echocardiographic evidence of an abnormal gradient across the mechanical valve.
Finally, determine if the mechanical valve shows abnormal disc motion with sticking leaflets. TEE is excellent for visualizing a mechanical mitral valve but isn’t helpful if it’s a mechanical aortic valve.
“Old-fashioned fluoroscopy is the best approach for looking at leaflet motion in the atrial valve and mitral valve. We’ve got 3-D cine now that provides beautiful images, but if you can get the same information with a quick fluoroscopy, go with the fluoroscopy,” the cardiologist suggested.
He reported having no financial conflicts of interest.
SNOWMASS, COLO. – Ultraslow infusion of a very-low-dose thrombolytic agent for treatment of mechanical prosthetic valve thrombosis appears to be as effective as surgery – the former first-line therapy – and sports a far lower stroke risk, Rick A. Nishimura, MD, said at the Annual Cardiovascular Conference at Snowmass.
“I’m not saying you have to use this, but I think it’s reasonable to consider it, especially if the patient is at high risk for surgery and low risk for thrombolysis,” according to Dr. Nishimura, professor of cardiovascular diseases and hypertension at the Mayo Clinic in Rochester, Minn.
He added that he and his Mayo colleagues have begun using the novel therapy and are favorably impressed with the resultant complete normalization of valve gradients and low complication rate.
Dr. Nishimura was cochair of the writing committee for the current American College of Cardiology/American Heart Association guidelines for management of valvular heart disease (Circulation. 2014 Jun 10;129[23]:e521-643). Those guidelines state that emergency surgery is the treatment of choice for thrombosis of a left-sided mechanical heart valve. That strong recommendation was based on a dozen nonrandomized studies reported prior to 2013 which showed a 95% success rate with surgery compared with 75% with conventional large-bolus thrombolytic therapy, a high 10%-12% mortality with either form of therapy, and a stroke risk of 12%-14% with thrombolytic therapy, substantially higher than for surgery.
Since release of the ACC/AHA guidelines, however, there’s been an important new development: Three groups outside of the United States have pioneered ultraslow thrombolytic therapy for mechanical prosthetic valve thrombosis. The supporting evidence comes from cohort studies, with no randomized trials done to date. But the collective reported experience from these three research teams shows a 90%-95% success rate – comparable to surgery – along with stroke and mortality rates in the low single digits.
The Turkish group waits until the patient’s international normalized ratio (INR) is below 2.5, then administers 25 mg of tissue plasminogen activator guided by transesophageal echocardiography (TEE) over 25 hours.
“We traditionally give 90 mg over 1 hour, so this is very, very slow therapy,” Dr. Nishimura observed.
After the 24-hour infusion, TEE is repeated. If imaging shows the clot is not resolved, another 25 mg of tissue plasminogen activator is given over 24 hours. This process is repeated for up to 8 days as needed (Am Heart J. 2015 Aug;170[2]:409-18).
Dr. Nishimura advised reserving ultraslow thrombolytic therapy for patients who are hemodynamically stable; this treatment takes a while to work, so patients in severe heart failure should be sent straight away to surgery. The novel therapy is best suited for patients with recent-onset mechanical valve thrombosis, a low INR, TEE evidence that the clot isn’t huge, and/or when surgical expertise isn’t readily available.
Before you can treat a prosthetic mechanical valve thrombosis, however, you have to make the diagnosis. Here’s what Dr. Nishimura recommends: First, suspect the condition on the basis of clinical symptoms of heart failure and dull, muffled S1 and S2 sounds on auscultation, especially in a patient who presents with a low INR.
Next, prove that obstruction is present via Doppler echocardiographic evidence of an abnormal gradient across the mechanical valve.
Finally, determine if the mechanical valve shows abnormal disc motion with sticking leaflets. TEE is excellent for visualizing a mechanical mitral valve but isn’t helpful if it’s a mechanical aortic valve.
“Old-fashioned fluoroscopy is the best approach for looking at leaflet motion in the atrial valve and mitral valve. We’ve got 3-D cine now that provides beautiful images, but if you can get the same information with a quick fluoroscopy, go with the fluoroscopy,” the cardiologist suggested.
He reported having no financial conflicts of interest.
SNOWMASS, COLO. – Ultraslow infusion of a very-low-dose thrombolytic agent for treatment of mechanical prosthetic valve thrombosis appears to be as effective as surgery – the former first-line therapy – and sports a far lower stroke risk, Rick A. Nishimura, MD, said at the Annual Cardiovascular Conference at Snowmass.
“I’m not saying you have to use this, but I think it’s reasonable to consider it, especially if the patient is at high risk for surgery and low risk for thrombolysis,” according to Dr. Nishimura, professor of cardiovascular diseases and hypertension at the Mayo Clinic in Rochester, Minn.
He added that he and his Mayo colleagues have begun using the novel therapy and are favorably impressed with the resultant complete normalization of valve gradients and low complication rate.
Dr. Nishimura was cochair of the writing committee for the current American College of Cardiology/American Heart Association guidelines for management of valvular heart disease (Circulation. 2014 Jun 10;129[23]:e521-643). Those guidelines state that emergency surgery is the treatment of choice for thrombosis of a left-sided mechanical heart valve. That strong recommendation was based on a dozen nonrandomized studies reported prior to 2013 which showed a 95% success rate with surgery compared with 75% with conventional large-bolus thrombolytic therapy, a high 10%-12% mortality with either form of therapy, and a stroke risk of 12%-14% with thrombolytic therapy, substantially higher than for surgery.
Since release of the ACC/AHA guidelines, however, there’s been an important new development: Three groups outside of the United States have pioneered ultraslow thrombolytic therapy for mechanical prosthetic valve thrombosis. The supporting evidence comes from cohort studies, with no randomized trials done to date. But the collective reported experience from these three research teams shows a 90%-95% success rate – comparable to surgery – along with stroke and mortality rates in the low single digits.
The Turkish group waits until the patient’s international normalized ratio (INR) is below 2.5, then administers 25 mg of tissue plasminogen activator guided by transesophageal echocardiography (TEE) over 25 hours.
“We traditionally give 90 mg over 1 hour, so this is very, very slow therapy,” Dr. Nishimura observed.
After the 24-hour infusion, TEE is repeated. If imaging shows the clot is not resolved, another 25 mg of tissue plasminogen activator is given over 24 hours. This process is repeated for up to 8 days as needed (Am Heart J. 2015 Aug;170[2]:409-18).
Dr. Nishimura advised reserving ultraslow thrombolytic therapy for patients who are hemodynamically stable; this treatment takes a while to work, so patients in severe heart failure should be sent straight away to surgery. The novel therapy is best suited for patients with recent-onset mechanical valve thrombosis, a low INR, TEE evidence that the clot isn’t huge, and/or when surgical expertise isn’t readily available.
Before you can treat a prosthetic mechanical valve thrombosis, however, you have to make the diagnosis. Here’s what Dr. Nishimura recommends: First, suspect the condition on the basis of clinical symptoms of heart failure and dull, muffled S1 and S2 sounds on auscultation, especially in a patient who presents with a low INR.
Next, prove that obstruction is present via Doppler echocardiographic evidence of an abnormal gradient across the mechanical valve.
Finally, determine if the mechanical valve shows abnormal disc motion with sticking leaflets. TEE is excellent for visualizing a mechanical mitral valve but isn’t helpful if it’s a mechanical aortic valve.
“Old-fashioned fluoroscopy is the best approach for looking at leaflet motion in the atrial valve and mitral valve. We’ve got 3-D cine now that provides beautiful images, but if you can get the same information with a quick fluoroscopy, go with the fluoroscopy,” the cardiologist suggested.
He reported having no financial conflicts of interest.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
The two faces of mitral regurgitation
SNOWMASS, COLO. – Primary mitral regurgitation and secondary mitral regurgitation may sound a lot alike, but they are in fact starkly different diseases, Blase A. Carabello, MD, said at the Annual Cardiovascular Conference at Snowmass.
“They are almost entirely different in their etiologies, in their pathophysiologies, and in their therapies,” according to Dr. Carabello, professor of medicine and chief of cardiology at East Carolina University in Greenville, N.C.
“On the other hand, if you fix the valve, you fix the disease,” he said.
In contrast, in secondary MR, the dysfunctional valve is a result of the ventricular problem, not its cause.
“In secondary MR, it’s the ventricle that made the valve sick. It’s the regional wall motion abnormalities, the displacement of the papillary muscles, and the dilation of the mitral annulus that pulls the mitral valve apart and prevents it from coapting,” he explained.
Dr. Carabello, who was on the writing committee for the current American College of Cardiology/American Heart Association guidelines on management of valvular heart disease (Circulation. 2014 Jun 10;129[23]:e521-643), highlighted other key distinctions between the two diseases.
Primary MR
“Primary MR, unlike aortic regurgitation, is not well tolerated. Early repair is key,” the cardiologist stressed.
The ACC/AHA guidelines emphasize the importance of early referral for surgery for primary MR because of surgery’s proven survival benefit. The triggers for surgery, as described in the guidelines, are easy to remember, namely, “symptoms/60/50/40.” That is, it’s time to move on to surgery when any of the following occurs: The patient becomes even mildly symptomatic, the left ventricular ejection fraction (LVEF) drops to 60%, the pulmonary artery pressure climbs to 50 mm Hg, or the LV end-systolic dimension reaches 40 mm.
Dr. Carabello said he believes those surgical thresholds are conservative, and it’s best to make the surgical referral when a patient approaches one or more of those triggers, but before actually reaching them.
“That’s the way I practice: Don’t wait for any of those things. Just get it done. One advantage to early repair is the patient can’t get lost to follow-up. They’re repaired, and they can’t take it out,” he said.
It’s possible that the next iteration of the guidelines will utilize stricter thresholds for surgery. French investigators have shown that surgery for primary MR achieves a significantly higher rate of normal LV function if the operation occurs when patients have an LVEF of 64% or more and an LV end-systolic dimension of less than 37 mm (Eur J Echocardiogr. 2011 Sep;12[9]:702-10). So far, though, there hasn’t been a confirmatory study.
“I think the normal EF in primary MR is about 70%. By the time the EF in a patient with primary MR gets down into the 50s, you’re looking at an extraordinarily sick ventricle,” according to Dr. Carabello.
The impetus for including the “symptoms/60/50/40” surgical triggers in the guidelines is to encourage physicians to make the surgical referral earlier than has often been the case. Too frequently, the surgical referral is delayed until damage to the ventricle is irreversible, with a resultant worsened prognosis.
“The natural history is such that, from the time a patient with severe primary MR enters your office to the annual mortality risk reaching 50% is only about 5 years. So if you’re going to do watchful waiting, fine, but remember: You don’t have all that long to watch and wait before something bad happens to these folks,” the cardiologist cautioned.
He added that it’s possible that, in the future, cardiac biomarkers will be utilized to help predict the long-term mortality risk of patients under medical management of their primary MR. In a recent study of 1,331 patients with primary MR, the investigators showed that the ratio of B-type natriuretic peptide (BNP) to the upper limit of normal BNP, adjusted for age and sex, was a powerful independent predictor of this risk (J Am Coll Cardiol. 2016 Sep 20;68[12]:1297-307).
The guidelines state that mitral valve repair is preferable to replacement as long as the heart team determines there’s at least a 95% chance of a durable repair. That’s because the operative mortality associated with replacement is significantly higher than with repair.
Secondary MR
Unlike in primary MR, it’s unclear whether surgery prolongs life for patients with secondary MR, or if mitral repair is superior to replacement. Thus, the current guidelines recommend surgery only for patients who are still severely symptomatic despite maximal guideline-directed medical therapy for heart failure as well as cardiac resynchronization therapy, provided they have a conduction system abnormality and qualify for the device therapy.
“If you’ve done all that and they’re still sick, I think that surgery or the MitraClip may benefit them very much,” Dr. Carabello said.
In the United States, the MitraClip transcatheter device is approved only for the treatment of primary MR in inoperable patients. But in the rest of the world, roughly three-quarters of these devices are used for treatment of secondary MR. That potential indication is currently under study in the United States in the phase III COAPT trial (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation).
“In the acute setting, the changes in hemodynamic variables and left ventricular performance that occur with the MitraClip are quite dramatic, with a marked decrease in wedge pressure and a markedly increased stroke volume, which is what it’s supposed to do. So I’m encouraged. We’ll wait for the results of the COAPT trial, which I believe is likely to demonstrate a reduction in hospitalizations and certainly an improvement in patient symptoms. But I doubt very much that we’ll ever see a change in lifespan because the ventricle is still sick, and we in 2017 have not found a way yet to revive a sick ventricle. It’s unlikely that fixing the MR will do so,” according to Dr. Carabello.
He reported serving on a data safety monitoring board for Edwards Lifesciences.
SNOWMASS, COLO. – Primary mitral regurgitation and secondary mitral regurgitation may sound a lot alike, but they are in fact starkly different diseases, Blase A. Carabello, MD, said at the Annual Cardiovascular Conference at Snowmass.
“They are almost entirely different in their etiologies, in their pathophysiologies, and in their therapies,” according to Dr. Carabello, professor of medicine and chief of cardiology at East Carolina University in Greenville, N.C.
“On the other hand, if you fix the valve, you fix the disease,” he said.
In contrast, in secondary MR, the dysfunctional valve is a result of the ventricular problem, not its cause.
“In secondary MR, it’s the ventricle that made the valve sick. It’s the regional wall motion abnormalities, the displacement of the papillary muscles, and the dilation of the mitral annulus that pulls the mitral valve apart and prevents it from coapting,” he explained.
Dr. Carabello, who was on the writing committee for the current American College of Cardiology/American Heart Association guidelines on management of valvular heart disease (Circulation. 2014 Jun 10;129[23]:e521-643), highlighted other key distinctions between the two diseases.
Primary MR
“Primary MR, unlike aortic regurgitation, is not well tolerated. Early repair is key,” the cardiologist stressed.
The ACC/AHA guidelines emphasize the importance of early referral for surgery for primary MR because of surgery’s proven survival benefit. The triggers for surgery, as described in the guidelines, are easy to remember, namely, “symptoms/60/50/40.” That is, it’s time to move on to surgery when any of the following occurs: The patient becomes even mildly symptomatic, the left ventricular ejection fraction (LVEF) drops to 60%, the pulmonary artery pressure climbs to 50 mm Hg, or the LV end-systolic dimension reaches 40 mm.
Dr. Carabello said he believes those surgical thresholds are conservative, and it’s best to make the surgical referral when a patient approaches one or more of those triggers, but before actually reaching them.
“That’s the way I practice: Don’t wait for any of those things. Just get it done. One advantage to early repair is the patient can’t get lost to follow-up. They’re repaired, and they can’t take it out,” he said.
It’s possible that the next iteration of the guidelines will utilize stricter thresholds for surgery. French investigators have shown that surgery for primary MR achieves a significantly higher rate of normal LV function if the operation occurs when patients have an LVEF of 64% or more and an LV end-systolic dimension of less than 37 mm (Eur J Echocardiogr. 2011 Sep;12[9]:702-10). So far, though, there hasn’t been a confirmatory study.
“I think the normal EF in primary MR is about 70%. By the time the EF in a patient with primary MR gets down into the 50s, you’re looking at an extraordinarily sick ventricle,” according to Dr. Carabello.
The impetus for including the “symptoms/60/50/40” surgical triggers in the guidelines is to encourage physicians to make the surgical referral earlier than has often been the case. Too frequently, the surgical referral is delayed until damage to the ventricle is irreversible, with a resultant worsened prognosis.
“The natural history is such that, from the time a patient with severe primary MR enters your office to the annual mortality risk reaching 50% is only about 5 years. So if you’re going to do watchful waiting, fine, but remember: You don’t have all that long to watch and wait before something bad happens to these folks,” the cardiologist cautioned.
He added that it’s possible that, in the future, cardiac biomarkers will be utilized to help predict the long-term mortality risk of patients under medical management of their primary MR. In a recent study of 1,331 patients with primary MR, the investigators showed that the ratio of B-type natriuretic peptide (BNP) to the upper limit of normal BNP, adjusted for age and sex, was a powerful independent predictor of this risk (J Am Coll Cardiol. 2016 Sep 20;68[12]:1297-307).
The guidelines state that mitral valve repair is preferable to replacement as long as the heart team determines there’s at least a 95% chance of a durable repair. That’s because the operative mortality associated with replacement is significantly higher than with repair.
Secondary MR
Unlike in primary MR, it’s unclear whether surgery prolongs life for patients with secondary MR, or if mitral repair is superior to replacement. Thus, the current guidelines recommend surgery only for patients who are still severely symptomatic despite maximal guideline-directed medical therapy for heart failure as well as cardiac resynchronization therapy, provided they have a conduction system abnormality and qualify for the device therapy.
“If you’ve done all that and they’re still sick, I think that surgery or the MitraClip may benefit them very much,” Dr. Carabello said.
In the United States, the MitraClip transcatheter device is approved only for the treatment of primary MR in inoperable patients. But in the rest of the world, roughly three-quarters of these devices are used for treatment of secondary MR. That potential indication is currently under study in the United States in the phase III COAPT trial (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation).
“In the acute setting, the changes in hemodynamic variables and left ventricular performance that occur with the MitraClip are quite dramatic, with a marked decrease in wedge pressure and a markedly increased stroke volume, which is what it’s supposed to do. So I’m encouraged. We’ll wait for the results of the COAPT trial, which I believe is likely to demonstrate a reduction in hospitalizations and certainly an improvement in patient symptoms. But I doubt very much that we’ll ever see a change in lifespan because the ventricle is still sick, and we in 2017 have not found a way yet to revive a sick ventricle. It’s unlikely that fixing the MR will do so,” according to Dr. Carabello.
He reported serving on a data safety monitoring board for Edwards Lifesciences.
SNOWMASS, COLO. – Primary mitral regurgitation and secondary mitral regurgitation may sound a lot alike, but they are in fact starkly different diseases, Blase A. Carabello, MD, said at the Annual Cardiovascular Conference at Snowmass.
“They are almost entirely different in their etiologies, in their pathophysiologies, and in their therapies,” according to Dr. Carabello, professor of medicine and chief of cardiology at East Carolina University in Greenville, N.C.
“On the other hand, if you fix the valve, you fix the disease,” he said.
In contrast, in secondary MR, the dysfunctional valve is a result of the ventricular problem, not its cause.
“In secondary MR, it’s the ventricle that made the valve sick. It’s the regional wall motion abnormalities, the displacement of the papillary muscles, and the dilation of the mitral annulus that pulls the mitral valve apart and prevents it from coapting,” he explained.
Dr. Carabello, who was on the writing committee for the current American College of Cardiology/American Heart Association guidelines on management of valvular heart disease (Circulation. 2014 Jun 10;129[23]:e521-643), highlighted other key distinctions between the two diseases.
Primary MR
“Primary MR, unlike aortic regurgitation, is not well tolerated. Early repair is key,” the cardiologist stressed.
The ACC/AHA guidelines emphasize the importance of early referral for surgery for primary MR because of surgery’s proven survival benefit. The triggers for surgery, as described in the guidelines, are easy to remember, namely, “symptoms/60/50/40.” That is, it’s time to move on to surgery when any of the following occurs: The patient becomes even mildly symptomatic, the left ventricular ejection fraction (LVEF) drops to 60%, the pulmonary artery pressure climbs to 50 mm Hg, or the LV end-systolic dimension reaches 40 mm.
Dr. Carabello said he believes those surgical thresholds are conservative, and it’s best to make the surgical referral when a patient approaches one or more of those triggers, but before actually reaching them.
“That’s the way I practice: Don’t wait for any of those things. Just get it done. One advantage to early repair is the patient can’t get lost to follow-up. They’re repaired, and they can’t take it out,” he said.
It’s possible that the next iteration of the guidelines will utilize stricter thresholds for surgery. French investigators have shown that surgery for primary MR achieves a significantly higher rate of normal LV function if the operation occurs when patients have an LVEF of 64% or more and an LV end-systolic dimension of less than 37 mm (Eur J Echocardiogr. 2011 Sep;12[9]:702-10). So far, though, there hasn’t been a confirmatory study.
“I think the normal EF in primary MR is about 70%. By the time the EF in a patient with primary MR gets down into the 50s, you’re looking at an extraordinarily sick ventricle,” according to Dr. Carabello.
The impetus for including the “symptoms/60/50/40” surgical triggers in the guidelines is to encourage physicians to make the surgical referral earlier than has often been the case. Too frequently, the surgical referral is delayed until damage to the ventricle is irreversible, with a resultant worsened prognosis.
“The natural history is such that, from the time a patient with severe primary MR enters your office to the annual mortality risk reaching 50% is only about 5 years. So if you’re going to do watchful waiting, fine, but remember: You don’t have all that long to watch and wait before something bad happens to these folks,” the cardiologist cautioned.
He added that it’s possible that, in the future, cardiac biomarkers will be utilized to help predict the long-term mortality risk of patients under medical management of their primary MR. In a recent study of 1,331 patients with primary MR, the investigators showed that the ratio of B-type natriuretic peptide (BNP) to the upper limit of normal BNP, adjusted for age and sex, was a powerful independent predictor of this risk (J Am Coll Cardiol. 2016 Sep 20;68[12]:1297-307).
The guidelines state that mitral valve repair is preferable to replacement as long as the heart team determines there’s at least a 95% chance of a durable repair. That’s because the operative mortality associated with replacement is significantly higher than with repair.
Secondary MR
Unlike in primary MR, it’s unclear whether surgery prolongs life for patients with secondary MR, or if mitral repair is superior to replacement. Thus, the current guidelines recommend surgery only for patients who are still severely symptomatic despite maximal guideline-directed medical therapy for heart failure as well as cardiac resynchronization therapy, provided they have a conduction system abnormality and qualify for the device therapy.
“If you’ve done all that and they’re still sick, I think that surgery or the MitraClip may benefit them very much,” Dr. Carabello said.
In the United States, the MitraClip transcatheter device is approved only for the treatment of primary MR in inoperable patients. But in the rest of the world, roughly three-quarters of these devices are used for treatment of secondary MR. That potential indication is currently under study in the United States in the phase III COAPT trial (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation).
“In the acute setting, the changes in hemodynamic variables and left ventricular performance that occur with the MitraClip are quite dramatic, with a marked decrease in wedge pressure and a markedly increased stroke volume, which is what it’s supposed to do. So I’m encouraged. We’ll wait for the results of the COAPT trial, which I believe is likely to demonstrate a reduction in hospitalizations and certainly an improvement in patient symptoms. But I doubt very much that we’ll ever see a change in lifespan because the ventricle is still sick, and we in 2017 have not found a way yet to revive a sick ventricle. It’s unlikely that fixing the MR will do so,” according to Dr. Carabello.
He reported serving on a data safety monitoring board for Edwards Lifesciences.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Trials refine device therapy options for heart failure
SNOWMASS, COLO. – The indication for primary prophylactic implantable cardioverter-defibrillator therapy in patients with nonischemic heart failure is likely to be downgraded in the next iteration of the ACC/AHA heart failure guidelines as a consequence of the negative results of the DANISH trial, William T. Abraham, MD, predicted at the Annual Cardiovascular Conference at Snowmass.
In addition to outlining where the guideline recommendations for implantable cardioverter-defibrillator (ICD) therapy stand today, and how they’re likely to change in response to the DANISH findings, he highlighted the latest patient selection criteria for cardiac resynchronization therapy (CRT), which have grown considerably more complicated over time.
“Following the success of neurohormonal inhibitors and antagonists, our only other breakthroughs for the management of heart failure have been CRT and ICDs,” he noted.
The two device therapies are complementary, and indeed are often employed in combination.
“CRT makes patients feel better and saves lives, while ICDs prolong survival without an effect on improving heart failure per se,” the cardiologist explained.
To put the quality of life benefits of CRT into perspective, studies show that the device therapy results in a placebo-subtracted improvement on the Minnesota Living With Heart Failure Questionnaire of 9-10 points.
“This is a large and clinically meaningful improvement in quality of life. Our best drugs for heart failure – beta blockers and ACE inhibitors – improve this same measure by 4 or 5 points,” Dr. Abraham said.
Current American College of Cardiology/American Heart Association heart failure guidelines give a class I, level of evidence: A, recommendation for prophylactic ICD therapy in patients with an left ventricular ejection fraction (LVEF) of 35% or less and New York Heart Association functional class II or III symptoms despite optimal medical therapy, regardless of whether their heart failure is attributable to ischemic heart disease or nonischemic dilated cardiomyopathy.
The DANISH trial investigators looked at the evidence base for primary prevention ICDs in nonischemic heart failure and concluded it needed shoring up. The recommendation relied mainly on subgroup analyses of larger landmark trials done about 15 years ago, before major improvements in medical therapy had occurred. These reservations were the impetus for the DANISH trial, in which more than 1,100 patients with symptomatic systolic heart failure were randomized to an ICD or usual care.
The primary outcome in the DANISH trial – all-cause mortality – occurred in 21.6% of patients in the ICD group and 23.4% of controls during a median follow-up of 68 months, a nonsignificant difference (N Engl J Med. 2016 Sep 29;375[13]:1221-30).
Turning to the CRT guidelines, Dr. Abraham noted that the simple, broad, class I recommendation for this form of device therapy in patients with cardiac dyssynchrony as defined by a QRS duration greater than 120 msec contained in the 2005 ACC/AHA heart failure guidelines has been whittled down over time as new evidence has unfolded. The only class I recommendation in the current guidelines is in patients with an LVEF of 35% or less, sinus rhythm, left bundle branch block with a QRS duration of 150 msec or longer, and NYHA class II, III, or ambulatory IV symptoms while on guideline-directed medical therapy (Circulation. 2012 Oct 1;126:1784-800). “That’s the money group right there. That’s the group for whom we have the greatest confidence of producing the greatest benefit with the application of cardiac resynchronization therapy,” he explained.
Studies examining the use of CRT in heart failure patients with a non–left bundle branch morphology and a QRS duration of less than 150 msec have yielded negative findings. So have attempts to utilize echocardiographic evidence of mechanical dyssynchrony rather than ECG measurement of QRS duration to guide patient selection for CRT.
“In our practice, any patient with a left bundle branch block gets a CRT device. Our confidence in its efficacy is greater in patients with a QRS of at least 150 msec, but the studies demonstrate clear benefit for patients with left bundle branch block and a QRS of 120-149 msec as well,” according to the cardiologist.
Studies also show that patients who are dependent upon ventricular pacing benefit from CRT.
“If you have a patient who requires at least 40% or more ventricular pacing and also has reduced ejection fraction heart failure, that patient should have a CRT device rather than a dual chamber ICD or standard right-sided right ventricular pacemaker,” he said.
All of this presupposes that first and foremost the patient is already on optimized guideline-directed medical therapy.
“With optimal medical therapy, some of these patients may improve their left ventricular ejection fraction above 35%, or they may become asymptomatic and no longer have an indication for CRT,” Dr. Abraham added.
The rationale for utilizing CRT in combination with an ICD is a bit shaky, resting on a single older landmark study, the COMPANION trial (N Engl J Med. 2004; 350:2140-50).
“That study wasn’t powered to answer the question of whether CRT-D [a combined CRT/ICD device] is better than CRT. Really, this remains somewhat of an unanswered question. So where are we today? Essentially, if a patient has an indication for CRT and an indication for an ICD, we implant a combined device,” he said.
Dr. Abraham reported serving as a consultant to Abbott Vascular, Medtronic, Novartis, and St. Jude Medical.
SNOWMASS, COLO. – The indication for primary prophylactic implantable cardioverter-defibrillator therapy in patients with nonischemic heart failure is likely to be downgraded in the next iteration of the ACC/AHA heart failure guidelines as a consequence of the negative results of the DANISH trial, William T. Abraham, MD, predicted at the Annual Cardiovascular Conference at Snowmass.
In addition to outlining where the guideline recommendations for implantable cardioverter-defibrillator (ICD) therapy stand today, and how they’re likely to change in response to the DANISH findings, he highlighted the latest patient selection criteria for cardiac resynchronization therapy (CRT), which have grown considerably more complicated over time.
“Following the success of neurohormonal inhibitors and antagonists, our only other breakthroughs for the management of heart failure have been CRT and ICDs,” he noted.
The two device therapies are complementary, and indeed are often employed in combination.
“CRT makes patients feel better and saves lives, while ICDs prolong survival without an effect on improving heart failure per se,” the cardiologist explained.
To put the quality of life benefits of CRT into perspective, studies show that the device therapy results in a placebo-subtracted improvement on the Minnesota Living With Heart Failure Questionnaire of 9-10 points.
“This is a large and clinically meaningful improvement in quality of life. Our best drugs for heart failure – beta blockers and ACE inhibitors – improve this same measure by 4 or 5 points,” Dr. Abraham said.
Current American College of Cardiology/American Heart Association heart failure guidelines give a class I, level of evidence: A, recommendation for prophylactic ICD therapy in patients with an left ventricular ejection fraction (LVEF) of 35% or less and New York Heart Association functional class II or III symptoms despite optimal medical therapy, regardless of whether their heart failure is attributable to ischemic heart disease or nonischemic dilated cardiomyopathy.
The DANISH trial investigators looked at the evidence base for primary prevention ICDs in nonischemic heart failure and concluded it needed shoring up. The recommendation relied mainly on subgroup analyses of larger landmark trials done about 15 years ago, before major improvements in medical therapy had occurred. These reservations were the impetus for the DANISH trial, in which more than 1,100 patients with symptomatic systolic heart failure were randomized to an ICD or usual care.
The primary outcome in the DANISH trial – all-cause mortality – occurred in 21.6% of patients in the ICD group and 23.4% of controls during a median follow-up of 68 months, a nonsignificant difference (N Engl J Med. 2016 Sep 29;375[13]:1221-30).
Turning to the CRT guidelines, Dr. Abraham noted that the simple, broad, class I recommendation for this form of device therapy in patients with cardiac dyssynchrony as defined by a QRS duration greater than 120 msec contained in the 2005 ACC/AHA heart failure guidelines has been whittled down over time as new evidence has unfolded. The only class I recommendation in the current guidelines is in patients with an LVEF of 35% or less, sinus rhythm, left bundle branch block with a QRS duration of 150 msec or longer, and NYHA class II, III, or ambulatory IV symptoms while on guideline-directed medical therapy (Circulation. 2012 Oct 1;126:1784-800). “That’s the money group right there. That’s the group for whom we have the greatest confidence of producing the greatest benefit with the application of cardiac resynchronization therapy,” he explained.
Studies examining the use of CRT in heart failure patients with a non–left bundle branch morphology and a QRS duration of less than 150 msec have yielded negative findings. So have attempts to utilize echocardiographic evidence of mechanical dyssynchrony rather than ECG measurement of QRS duration to guide patient selection for CRT.
“In our practice, any patient with a left bundle branch block gets a CRT device. Our confidence in its efficacy is greater in patients with a QRS of at least 150 msec, but the studies demonstrate clear benefit for patients with left bundle branch block and a QRS of 120-149 msec as well,” according to the cardiologist.
Studies also show that patients who are dependent upon ventricular pacing benefit from CRT.
“If you have a patient who requires at least 40% or more ventricular pacing and also has reduced ejection fraction heart failure, that patient should have a CRT device rather than a dual chamber ICD or standard right-sided right ventricular pacemaker,” he said.
All of this presupposes that first and foremost the patient is already on optimized guideline-directed medical therapy.
“With optimal medical therapy, some of these patients may improve their left ventricular ejection fraction above 35%, or they may become asymptomatic and no longer have an indication for CRT,” Dr. Abraham added.
The rationale for utilizing CRT in combination with an ICD is a bit shaky, resting on a single older landmark study, the COMPANION trial (N Engl J Med. 2004; 350:2140-50).
“That study wasn’t powered to answer the question of whether CRT-D [a combined CRT/ICD device] is better than CRT. Really, this remains somewhat of an unanswered question. So where are we today? Essentially, if a patient has an indication for CRT and an indication for an ICD, we implant a combined device,” he said.
Dr. Abraham reported serving as a consultant to Abbott Vascular, Medtronic, Novartis, and St. Jude Medical.
SNOWMASS, COLO. – The indication for primary prophylactic implantable cardioverter-defibrillator therapy in patients with nonischemic heart failure is likely to be downgraded in the next iteration of the ACC/AHA heart failure guidelines as a consequence of the negative results of the DANISH trial, William T. Abraham, MD, predicted at the Annual Cardiovascular Conference at Snowmass.
In addition to outlining where the guideline recommendations for implantable cardioverter-defibrillator (ICD) therapy stand today, and how they’re likely to change in response to the DANISH findings, he highlighted the latest patient selection criteria for cardiac resynchronization therapy (CRT), which have grown considerably more complicated over time.
“Following the success of neurohormonal inhibitors and antagonists, our only other breakthroughs for the management of heart failure have been CRT and ICDs,” he noted.
The two device therapies are complementary, and indeed are often employed in combination.
“CRT makes patients feel better and saves lives, while ICDs prolong survival without an effect on improving heart failure per se,” the cardiologist explained.
To put the quality of life benefits of CRT into perspective, studies show that the device therapy results in a placebo-subtracted improvement on the Minnesota Living With Heart Failure Questionnaire of 9-10 points.
“This is a large and clinically meaningful improvement in quality of life. Our best drugs for heart failure – beta blockers and ACE inhibitors – improve this same measure by 4 or 5 points,” Dr. Abraham said.
Current American College of Cardiology/American Heart Association heart failure guidelines give a class I, level of evidence: A, recommendation for prophylactic ICD therapy in patients with an left ventricular ejection fraction (LVEF) of 35% or less and New York Heart Association functional class II or III symptoms despite optimal medical therapy, regardless of whether their heart failure is attributable to ischemic heart disease or nonischemic dilated cardiomyopathy.
The DANISH trial investigators looked at the evidence base for primary prevention ICDs in nonischemic heart failure and concluded it needed shoring up. The recommendation relied mainly on subgroup analyses of larger landmark trials done about 15 years ago, before major improvements in medical therapy had occurred. These reservations were the impetus for the DANISH trial, in which more than 1,100 patients with symptomatic systolic heart failure were randomized to an ICD or usual care.
The primary outcome in the DANISH trial – all-cause mortality – occurred in 21.6% of patients in the ICD group and 23.4% of controls during a median follow-up of 68 months, a nonsignificant difference (N Engl J Med. 2016 Sep 29;375[13]:1221-30).
Turning to the CRT guidelines, Dr. Abraham noted that the simple, broad, class I recommendation for this form of device therapy in patients with cardiac dyssynchrony as defined by a QRS duration greater than 120 msec contained in the 2005 ACC/AHA heart failure guidelines has been whittled down over time as new evidence has unfolded. The only class I recommendation in the current guidelines is in patients with an LVEF of 35% or less, sinus rhythm, left bundle branch block with a QRS duration of 150 msec or longer, and NYHA class II, III, or ambulatory IV symptoms while on guideline-directed medical therapy (Circulation. 2012 Oct 1;126:1784-800). “That’s the money group right there. That’s the group for whom we have the greatest confidence of producing the greatest benefit with the application of cardiac resynchronization therapy,” he explained.
Studies examining the use of CRT in heart failure patients with a non–left bundle branch morphology and a QRS duration of less than 150 msec have yielded negative findings. So have attempts to utilize echocardiographic evidence of mechanical dyssynchrony rather than ECG measurement of QRS duration to guide patient selection for CRT.
“In our practice, any patient with a left bundle branch block gets a CRT device. Our confidence in its efficacy is greater in patients with a QRS of at least 150 msec, but the studies demonstrate clear benefit for patients with left bundle branch block and a QRS of 120-149 msec as well,” according to the cardiologist.
Studies also show that patients who are dependent upon ventricular pacing benefit from CRT.
“If you have a patient who requires at least 40% or more ventricular pacing and also has reduced ejection fraction heart failure, that patient should have a CRT device rather than a dual chamber ICD or standard right-sided right ventricular pacemaker,” he said.
All of this presupposes that first and foremost the patient is already on optimized guideline-directed medical therapy.
“With optimal medical therapy, some of these patients may improve their left ventricular ejection fraction above 35%, or they may become asymptomatic and no longer have an indication for CRT,” Dr. Abraham added.
The rationale for utilizing CRT in combination with an ICD is a bit shaky, resting on a single older landmark study, the COMPANION trial (N Engl J Med. 2004; 350:2140-50).
“That study wasn’t powered to answer the question of whether CRT-D [a combined CRT/ICD device] is better than CRT. Really, this remains somewhat of an unanswered question. So where are we today? Essentially, if a patient has an indication for CRT and an indication for an ICD, we implant a combined device,” he said.
Dr. Abraham reported serving as a consultant to Abbott Vascular, Medtronic, Novartis, and St. Jude Medical.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Smaller, intrapericardial LVAD noninferior to HeartMate II
A smaller, centrifugal-flow left ventricular assist device that lies entirely within the pericardial space was found noninferior to the HeartMate II axial-flow device in patients with advanced heart failure who weren’t eligible for heart transplant, according to a report published online Feb. 2 in the New England Journal of Medicine.
The two LVADs were compared in ENDURANCE (A Clinical Trial to Evaluate the HeartWare Ventricular Assist System), a prospective, randomized trial in 445 patients who were treated at 48 U.S. sites and followed for 2 years. The study participants had an LV ejection fraction of 25% or less and high prevalences of abnormal renal function and dependence on intravenous inotropic support, said Joseph G. Rogers, MD, of Duke University, Durham, N.C., and his associates.
The study participants were randomly assigned to receive the HeartWare, an investigational centrifugal-flow LVAD (297 patients) or the standard axial-flow HeartMate II LVAD (148 patients). In the intention-to-treat analysis, the primary endpoint – a composite of survival free from disabling stroke and no removal of the device for malfunction or failure – was 55.4% with the new device and 59.1% in the control group. The results were similar in the per-protocol and the as-treated analyses, demonstrating that the new device was noninferior but not superior to the axial-flow LVAD, the investigators said (N Engl J Med. 2017 Feb 2. doi: 10.1056/NEJMoa1602954).
There were significantly more cases of device malfunction or failure requiring urgent surgery in the control group than in the centrifugal-flow group (16.2% vs 8.8%), but significantly more cases of stroke (29.7% vs 12.1%), sepsis, and right heart failure. Rates of major bleeding, cardiac arrhythmia, renal dysfunction, and infection were similar between the two study groups. Overall survival* also was not significantly different (60.2% with the new LVAD and 67.6% in the control group).
Both study groups showed significant and comparable improvement after LVAD implantation. Functional status improved to New York Heart Association class I or II in roughly 80% of patients. Mean 6-minute walk distance improved from 100.2 to 199.4 meters with the new device and from 91.9 to 190.1 meters in the control group, a change that was noted within 3 months of surgery and persisted through the end of follow-up. Similarly, mean scores on the Kansas City Cardiomyopathy Questionnaire improved by 25.8 points and 25.3 points, respectively, and mean scores on the European Quality of Life 5 Dimensions scale improved by 22.5 points and 25.5 points, respectively.
This trial was sponsored by HeartWare, which was also involved in data management and analysis. Dr. Rogers reported having no relevant financial disclosures; his associates reported ties to HeartWare (Medtronic), Thoratec (St. Jude Medical), Novartis, and GE HealthCare.
*Correction 2/2/17: An earlier version of this article misidentified survival rates as mortality rates.
The smaller, fully intrapericardial, centrifugal-flow LVAD met one of its goals: Compared with the existing LVAD, it significantly reduced the need for urgent reoperation due to device malfunction or failure.
However, it did not resolve some of the most important problems with LVAD support. It didn’t reduce stroke risk; in fact, the overall risk of stroke was higher with the new device. It also failed to reduce the risk of bleeding, sepsis, or right heart failure.
It appears that no LVAD is fully superior to the others.
Roland Hetzer, MD, PhD, and Eva M. Delmo Walter, MD, PhD, of Cardio Centrum in Berlin, made these remarks in an accompanying editorial (N Engl J Med. 2017 Feb 2. doi: 10.1056/NEJMe1613755). They reported having no relevant financial disclosures.
The smaller, fully intrapericardial, centrifugal-flow LVAD met one of its goals: Compared with the existing LVAD, it significantly reduced the need for urgent reoperation due to device malfunction or failure.
However, it did not resolve some of the most important problems with LVAD support. It didn’t reduce stroke risk; in fact, the overall risk of stroke was higher with the new device. It also failed to reduce the risk of bleeding, sepsis, or right heart failure.
It appears that no LVAD is fully superior to the others.
Roland Hetzer, MD, PhD, and Eva M. Delmo Walter, MD, PhD, of Cardio Centrum in Berlin, made these remarks in an accompanying editorial (N Engl J Med. 2017 Feb 2. doi: 10.1056/NEJMe1613755). They reported having no relevant financial disclosures.
The smaller, fully intrapericardial, centrifugal-flow LVAD met one of its goals: Compared with the existing LVAD, it significantly reduced the need for urgent reoperation due to device malfunction or failure.
However, it did not resolve some of the most important problems with LVAD support. It didn’t reduce stroke risk; in fact, the overall risk of stroke was higher with the new device. It also failed to reduce the risk of bleeding, sepsis, or right heart failure.
It appears that no LVAD is fully superior to the others.
Roland Hetzer, MD, PhD, and Eva M. Delmo Walter, MD, PhD, of Cardio Centrum in Berlin, made these remarks in an accompanying editorial (N Engl J Med. 2017 Feb 2. doi: 10.1056/NEJMe1613755). They reported having no relevant financial disclosures.
A smaller, centrifugal-flow left ventricular assist device that lies entirely within the pericardial space was found noninferior to the HeartMate II axial-flow device in patients with advanced heart failure who weren’t eligible for heart transplant, according to a report published online Feb. 2 in the New England Journal of Medicine.
The two LVADs were compared in ENDURANCE (A Clinical Trial to Evaluate the HeartWare Ventricular Assist System), a prospective, randomized trial in 445 patients who were treated at 48 U.S. sites and followed for 2 years. The study participants had an LV ejection fraction of 25% or less and high prevalences of abnormal renal function and dependence on intravenous inotropic support, said Joseph G. Rogers, MD, of Duke University, Durham, N.C., and his associates.
The study participants were randomly assigned to receive the HeartWare, an investigational centrifugal-flow LVAD (297 patients) or the standard axial-flow HeartMate II LVAD (148 patients). In the intention-to-treat analysis, the primary endpoint – a composite of survival free from disabling stroke and no removal of the device for malfunction or failure – was 55.4% with the new device and 59.1% in the control group. The results were similar in the per-protocol and the as-treated analyses, demonstrating that the new device was noninferior but not superior to the axial-flow LVAD, the investigators said (N Engl J Med. 2017 Feb 2. doi: 10.1056/NEJMoa1602954).
There were significantly more cases of device malfunction or failure requiring urgent surgery in the control group than in the centrifugal-flow group (16.2% vs 8.8%), but significantly more cases of stroke (29.7% vs 12.1%), sepsis, and right heart failure. Rates of major bleeding, cardiac arrhythmia, renal dysfunction, and infection were similar between the two study groups. Overall survival* also was not significantly different (60.2% with the new LVAD and 67.6% in the control group).
Both study groups showed significant and comparable improvement after LVAD implantation. Functional status improved to New York Heart Association class I or II in roughly 80% of patients. Mean 6-minute walk distance improved from 100.2 to 199.4 meters with the new device and from 91.9 to 190.1 meters in the control group, a change that was noted within 3 months of surgery and persisted through the end of follow-up. Similarly, mean scores on the Kansas City Cardiomyopathy Questionnaire improved by 25.8 points and 25.3 points, respectively, and mean scores on the European Quality of Life 5 Dimensions scale improved by 22.5 points and 25.5 points, respectively.
This trial was sponsored by HeartWare, which was also involved in data management and analysis. Dr. Rogers reported having no relevant financial disclosures; his associates reported ties to HeartWare (Medtronic), Thoratec (St. Jude Medical), Novartis, and GE HealthCare.
*Correction 2/2/17: An earlier version of this article misidentified survival rates as mortality rates.
A smaller, centrifugal-flow left ventricular assist device that lies entirely within the pericardial space was found noninferior to the HeartMate II axial-flow device in patients with advanced heart failure who weren’t eligible for heart transplant, according to a report published online Feb. 2 in the New England Journal of Medicine.
The two LVADs were compared in ENDURANCE (A Clinical Trial to Evaluate the HeartWare Ventricular Assist System), a prospective, randomized trial in 445 patients who were treated at 48 U.S. sites and followed for 2 years. The study participants had an LV ejection fraction of 25% or less and high prevalences of abnormal renal function and dependence on intravenous inotropic support, said Joseph G. Rogers, MD, of Duke University, Durham, N.C., and his associates.
The study participants were randomly assigned to receive the HeartWare, an investigational centrifugal-flow LVAD (297 patients) or the standard axial-flow HeartMate II LVAD (148 patients). In the intention-to-treat analysis, the primary endpoint – a composite of survival free from disabling stroke and no removal of the device for malfunction or failure – was 55.4% with the new device and 59.1% in the control group. The results were similar in the per-protocol and the as-treated analyses, demonstrating that the new device was noninferior but not superior to the axial-flow LVAD, the investigators said (N Engl J Med. 2017 Feb 2. doi: 10.1056/NEJMoa1602954).
There were significantly more cases of device malfunction or failure requiring urgent surgery in the control group than in the centrifugal-flow group (16.2% vs 8.8%), but significantly more cases of stroke (29.7% vs 12.1%), sepsis, and right heart failure. Rates of major bleeding, cardiac arrhythmia, renal dysfunction, and infection were similar between the two study groups. Overall survival* also was not significantly different (60.2% with the new LVAD and 67.6% in the control group).
Both study groups showed significant and comparable improvement after LVAD implantation. Functional status improved to New York Heart Association class I or II in roughly 80% of patients. Mean 6-minute walk distance improved from 100.2 to 199.4 meters with the new device and from 91.9 to 190.1 meters in the control group, a change that was noted within 3 months of surgery and persisted through the end of follow-up. Similarly, mean scores on the Kansas City Cardiomyopathy Questionnaire improved by 25.8 points and 25.3 points, respectively, and mean scores on the European Quality of Life 5 Dimensions scale improved by 22.5 points and 25.5 points, respectively.
This trial was sponsored by HeartWare, which was also involved in data management and analysis. Dr. Rogers reported having no relevant financial disclosures; his associates reported ties to HeartWare (Medtronic), Thoratec (St. Jude Medical), Novartis, and GE HealthCare.
*Correction 2/2/17: An earlier version of this article misidentified survival rates as mortality rates.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: A smaller, centrifugal-flow left ventricular assist device was found noninferior to the existing FDA-approved axial-flow device in patients with advanced heart failure who weren’t eligible for heart transplant.
Major finding: The primary endpoint – a composite of survival free from disabling stroke and no removal of the device for malfunction or failure – was 55.4% with the new device and 59.1% in the control group.
Data source: ENDURANCE, a prospective multicenter randomized trial in 445 patients followed for 2 years.
Disclosures: This trial was sponsored by HeartWare, which was also involved in data management and analysis. Dr. Rogers reported having no relevant financial disclosures; his associates reported ties to HeartWare (Medtronic), Thoratec (St. Jude Medical), Novartis, and GE HealthCare.
STICHES boosts CABG role in severe LV dysfunction
SNOWMASS, COLO. – Coronary artery bypass graft surgery in patients with severe ischemic left ventricular dysfunction is overdue for an upgrade in status in the American College of Cardiology/American Heart Association guidelines on the strength of the landmark STICH trial and its extended follow-up stage known as STICHES, according to Vinod H. Thourani, MD.
Currently, the guidelines give CABG in this large and growing population a class IIb recommendation, meaning it “might be considered.” This undervalues the study’s core lesson: “STICHES showed a clear survival benefit with CABG, so this most likely should become a class IIa recommendation,” Dr. Thourani said at the Annual Cardiovascular Conference at Snowmass.
He went on to describe how he applies the key study findings to individual patients.
At 5 and 10 years of follow-up, the probability of all-cause mortality was reduced by 14% and 16%, respectively, in the CABG group. The surgery provided on average an 18-month extension of life. The price paid for the CABG benefit was a 3.6% mortality rate at 30 days; however, this was overcome by the 2-year mark, at which point survival in the CABG group surpassed that in controls. Thereafter, the all-cause mortality gap between the two groups continued to widen for the duration of follow-up.
For the composite endpoint of all-cause mortality or cardiovascular hospitalization, the CABG group enjoyed a 26% relative risk reduction, compared with optimal medical management alone at 5 years, and a 28% reduction in risk at 10 years. The two study groups diverged in terms of risk of cardiovascular hospitalization after only 3 months.
CABG provided a reduction in the risk of cardiovascular death that was consistent across all ages. In contrast, the reduction in all-cause mortality was not, since a higher proportion of deaths in older patients came from cancer and other noncardiovascular causes (Circulation. 2016 Nov 1;134[18]:1314-24).
There have been no randomized, controlled trials of percutaneous coronary intervention in patients with heart failure.
“An interesting finding in STICHES was that medical therapy had a much higher 10-year all-cause mortality the younger the patient was. So CABG particularly benefits those who are at a younger age – in this study, age 60 or less. As you get older, say, at 80 years of age, I’m not sure there’s a huge benefit in all-cause mortality at that point,” said Dr. Thourani, professor of surgery and medicine, and codirector of the structural heart and valve center at Emory University in Atlanta.
In a STICH substudy, roughly half of participants underwent presurgical myocardial viability testing via single-photon emission CT and/or dobutamine echocardiography. The investigators found that the results didn’t predict mortality benefit for CABG (N Engl J Med. 2011 Apr 28;364[17]:1617-25).
More recently, however, other investigators have reported MRI to have prognostic value. For example, Belgian investigators showed that medical therapy in patients with ischemic heart failure and dysfunctional but viable myocardium on delayed-enhanced MRI was associated with a 4.56-fold increased likelihood of mortality during 3 years of follow-up, compared with complete revascularization via CABG (J Am Coll Cardiol. 2012 Feb 28;59[9]:825-35).
“This observation has been useful for me,” Dr. Thourani said. “My own personal practice is if I have good targets, I don’t do viability testing, but if I have really bad targets where I know I’m going to have a tough time sewing grafts, I try to get an MRI for viability testing.”
One important lesson of STICH is that all patients with heart failure and a low left ventricular ejection fraction should have a coronary angiogram, even if they are free of ischemia on noninvasive testing and have no angina. That’s because the patients enrolled in STICH had little or no angina, the surgeon continued.
These STICH-type patients will benefit greatly from a heart team assessment factoring in an individual’s Society of Thoracic Surgeons’ predicted risk score, based on age, comorbidities, and other factors. For example, if a patient’s STS risk score with CABG is 0.7%, that’s a strong argument for opting for the surgery, since the 30-day operative mortality in STICH was 3.6%. If, on the other hand, the STS score is greater than 7%, that’s a tougher call.
“I think it’s really important that a heart team assessment includes a noninvasive cardiologist as well as an interventional cardiologist and cardiac surgeon because I think interventionalists and cardiac surgeons sometimes get a little goofy in their assessment of these patients,” Dr. Thourani said.
Patients with a low ejection fraction and coronary artery disease who are deemed poor candidates for CABG should be evaluated for a mechanical circulatory support device or a heart transplant.
“I think that’s something we don’t think about enough, quite honestly,” he said.
Dr. Thourani reported serving as a consultant to Abbott Vascular, Edwards Lifesciences, and Gore, and receiving research grants from numerous companies.
SNOWMASS, COLO. – Coronary artery bypass graft surgery in patients with severe ischemic left ventricular dysfunction is overdue for an upgrade in status in the American College of Cardiology/American Heart Association guidelines on the strength of the landmark STICH trial and its extended follow-up stage known as STICHES, according to Vinod H. Thourani, MD.
Currently, the guidelines give CABG in this large and growing population a class IIb recommendation, meaning it “might be considered.” This undervalues the study’s core lesson: “STICHES showed a clear survival benefit with CABG, so this most likely should become a class IIa recommendation,” Dr. Thourani said at the Annual Cardiovascular Conference at Snowmass.
He went on to describe how he applies the key study findings to individual patients.
At 5 and 10 years of follow-up, the probability of all-cause mortality was reduced by 14% and 16%, respectively, in the CABG group. The surgery provided on average an 18-month extension of life. The price paid for the CABG benefit was a 3.6% mortality rate at 30 days; however, this was overcome by the 2-year mark, at which point survival in the CABG group surpassed that in controls. Thereafter, the all-cause mortality gap between the two groups continued to widen for the duration of follow-up.
For the composite endpoint of all-cause mortality or cardiovascular hospitalization, the CABG group enjoyed a 26% relative risk reduction, compared with optimal medical management alone at 5 years, and a 28% reduction in risk at 10 years. The two study groups diverged in terms of risk of cardiovascular hospitalization after only 3 months.
CABG provided a reduction in the risk of cardiovascular death that was consistent across all ages. In contrast, the reduction in all-cause mortality was not, since a higher proportion of deaths in older patients came from cancer and other noncardiovascular causes (Circulation. 2016 Nov 1;134[18]:1314-24).
There have been no randomized, controlled trials of percutaneous coronary intervention in patients with heart failure.
“An interesting finding in STICHES was that medical therapy had a much higher 10-year all-cause mortality the younger the patient was. So CABG particularly benefits those who are at a younger age – in this study, age 60 or less. As you get older, say, at 80 years of age, I’m not sure there’s a huge benefit in all-cause mortality at that point,” said Dr. Thourani, professor of surgery and medicine, and codirector of the structural heart and valve center at Emory University in Atlanta.
In a STICH substudy, roughly half of participants underwent presurgical myocardial viability testing via single-photon emission CT and/or dobutamine echocardiography. The investigators found that the results didn’t predict mortality benefit for CABG (N Engl J Med. 2011 Apr 28;364[17]:1617-25).
More recently, however, other investigators have reported MRI to have prognostic value. For example, Belgian investigators showed that medical therapy in patients with ischemic heart failure and dysfunctional but viable myocardium on delayed-enhanced MRI was associated with a 4.56-fold increased likelihood of mortality during 3 years of follow-up, compared with complete revascularization via CABG (J Am Coll Cardiol. 2012 Feb 28;59[9]:825-35).
“This observation has been useful for me,” Dr. Thourani said. “My own personal practice is if I have good targets, I don’t do viability testing, but if I have really bad targets where I know I’m going to have a tough time sewing grafts, I try to get an MRI for viability testing.”
One important lesson of STICH is that all patients with heart failure and a low left ventricular ejection fraction should have a coronary angiogram, even if they are free of ischemia on noninvasive testing and have no angina. That’s because the patients enrolled in STICH had little or no angina, the surgeon continued.
These STICH-type patients will benefit greatly from a heart team assessment factoring in an individual’s Society of Thoracic Surgeons’ predicted risk score, based on age, comorbidities, and other factors. For example, if a patient’s STS risk score with CABG is 0.7%, that’s a strong argument for opting for the surgery, since the 30-day operative mortality in STICH was 3.6%. If, on the other hand, the STS score is greater than 7%, that’s a tougher call.
“I think it’s really important that a heart team assessment includes a noninvasive cardiologist as well as an interventional cardiologist and cardiac surgeon because I think interventionalists and cardiac surgeons sometimes get a little goofy in their assessment of these patients,” Dr. Thourani said.
Patients with a low ejection fraction and coronary artery disease who are deemed poor candidates for CABG should be evaluated for a mechanical circulatory support device or a heart transplant.
“I think that’s something we don’t think about enough, quite honestly,” he said.
Dr. Thourani reported serving as a consultant to Abbott Vascular, Edwards Lifesciences, and Gore, and receiving research grants from numerous companies.
SNOWMASS, COLO. – Coronary artery bypass graft surgery in patients with severe ischemic left ventricular dysfunction is overdue for an upgrade in status in the American College of Cardiology/American Heart Association guidelines on the strength of the landmark STICH trial and its extended follow-up stage known as STICHES, according to Vinod H. Thourani, MD.
Currently, the guidelines give CABG in this large and growing population a class IIb recommendation, meaning it “might be considered.” This undervalues the study’s core lesson: “STICHES showed a clear survival benefit with CABG, so this most likely should become a class IIa recommendation,” Dr. Thourani said at the Annual Cardiovascular Conference at Snowmass.
He went on to describe how he applies the key study findings to individual patients.
At 5 and 10 years of follow-up, the probability of all-cause mortality was reduced by 14% and 16%, respectively, in the CABG group. The surgery provided on average an 18-month extension of life. The price paid for the CABG benefit was a 3.6% mortality rate at 30 days; however, this was overcome by the 2-year mark, at which point survival in the CABG group surpassed that in controls. Thereafter, the all-cause mortality gap between the two groups continued to widen for the duration of follow-up.
For the composite endpoint of all-cause mortality or cardiovascular hospitalization, the CABG group enjoyed a 26% relative risk reduction, compared with optimal medical management alone at 5 years, and a 28% reduction in risk at 10 years. The two study groups diverged in terms of risk of cardiovascular hospitalization after only 3 months.
CABG provided a reduction in the risk of cardiovascular death that was consistent across all ages. In contrast, the reduction in all-cause mortality was not, since a higher proportion of deaths in older patients came from cancer and other noncardiovascular causes (Circulation. 2016 Nov 1;134[18]:1314-24).
There have been no randomized, controlled trials of percutaneous coronary intervention in patients with heart failure.
“An interesting finding in STICHES was that medical therapy had a much higher 10-year all-cause mortality the younger the patient was. So CABG particularly benefits those who are at a younger age – in this study, age 60 or less. As you get older, say, at 80 years of age, I’m not sure there’s a huge benefit in all-cause mortality at that point,” said Dr. Thourani, professor of surgery and medicine, and codirector of the structural heart and valve center at Emory University in Atlanta.
In a STICH substudy, roughly half of participants underwent presurgical myocardial viability testing via single-photon emission CT and/or dobutamine echocardiography. The investigators found that the results didn’t predict mortality benefit for CABG (N Engl J Med. 2011 Apr 28;364[17]:1617-25).
More recently, however, other investigators have reported MRI to have prognostic value. For example, Belgian investigators showed that medical therapy in patients with ischemic heart failure and dysfunctional but viable myocardium on delayed-enhanced MRI was associated with a 4.56-fold increased likelihood of mortality during 3 years of follow-up, compared with complete revascularization via CABG (J Am Coll Cardiol. 2012 Feb 28;59[9]:825-35).
“This observation has been useful for me,” Dr. Thourani said. “My own personal practice is if I have good targets, I don’t do viability testing, but if I have really bad targets where I know I’m going to have a tough time sewing grafts, I try to get an MRI for viability testing.”
One important lesson of STICH is that all patients with heart failure and a low left ventricular ejection fraction should have a coronary angiogram, even if they are free of ischemia on noninvasive testing and have no angina. That’s because the patients enrolled in STICH had little or no angina, the surgeon continued.
These STICH-type patients will benefit greatly from a heart team assessment factoring in an individual’s Society of Thoracic Surgeons’ predicted risk score, based on age, comorbidities, and other factors. For example, if a patient’s STS risk score with CABG is 0.7%, that’s a strong argument for opting for the surgery, since the 30-day operative mortality in STICH was 3.6%. If, on the other hand, the STS score is greater than 7%, that’s a tougher call.
“I think it’s really important that a heart team assessment includes a noninvasive cardiologist as well as an interventional cardiologist and cardiac surgeon because I think interventionalists and cardiac surgeons sometimes get a little goofy in their assessment of these patients,” Dr. Thourani said.
Patients with a low ejection fraction and coronary artery disease who are deemed poor candidates for CABG should be evaluated for a mechanical circulatory support device or a heart transplant.
“I think that’s something we don’t think about enough, quite honestly,” he said.
Dr. Thourani reported serving as a consultant to Abbott Vascular, Edwards Lifesciences, and Gore, and receiving research grants from numerous companies.
EXPERT ANALYSIS AT THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Complex congenital heart conditions call for complex care in pregnancy
A new scientific statement from the American Heart Association (AHA) brings together recommendations for management of pregnancy for women with serious congenital heart disease. The 38-page document addresses a wide range of complex congenital heart conditions, presenting a newly unified set of recommendations for care that ranges from preconception counseling, through pregnancy, labor, and delivery, to the postpartum period.
Caring for women with complex congenital heart lesions is becoming more commonplace, as more infants undergo successful repairs of previously-unsurvivable cardiac anomalies. “More moms with congenital heart disease are showing up pregnant, having survived the tumultuous peripartum and neonatal period, and are now facing a new set of risks in pregnancy,” Michael Foley, MD, chair of the department of obstetrics and gynecology at the University of Arizona, Phoenix, said in an interview.
Joseph Kay, MD, a cardiologist and professor of medicine and pediatrics at the University of Colorado, Aurora, said that one big benefit of the new scientific statement is having a single reference point for care of these patients. “The scientific statement brings all of the information about caring for these patients together into one document. This will be a very valuable resource for trainees to get a sense of what’s important; it also represents a platform for new programs to understand the scope of services needed,” said Dr. Kay in an interview.
The document provides a thorough review of the physiologic changes of pregnancy and the intrapartum and postpartum periods, noting that the heterogeneity of congenital heart disease means that women who have different lesions carry different risks in pregnancy.
Examples of lesions presenting intermediate risk include most arrhythmias (category II), hypertrophic cardiomyopathy, and a repaired coarctation (both category II-III). The most severe lesions carry a contraindication for pregnancy; the WHO guidelines suggest discussing termination should women with a category IV lesion become pregnant. Severe mitral stenosis, severe symptomatic aortic stenosis, and severe systemic ventricular dysfunction all place women into category IV.
Beginning with pregnancy risk category III, the WHO guidelines recommend intensive cardiac and obstetric monitoring throughout pregnancy, childbirth, and the puerperium. Several maternal-fetal medicine specialists interviewed all agreed that an interdisciplinary team is a must for good obstetric care in this population.
How interdisciplinary care plays out can depend on geography and facility-dependent resources. Dr. Kay said that his facility is the referral site for pregnant women with complex congenital lesions in an area that spans the Canadian and the Mexican borders from north to south, and ranges from parts of Kansas to eastern Montana from east to west. Still, Dr. Kay said that even for patients with lower-risk lesions, “We will see patients at least once, at approximately the midpoint of pregnancy, and again during the third trimester if possible.” The specifics of care depend on “the nature of the lesion and the complexity of the disease,” said Dr. Kay.
In his facility, said Dr. Kay, telemetry is available for all of the labor and delivery unit beds. This means that the mother and infant can usually stay together and receive postpartum nursing and lactation care from a skilled staff.
In no circumstances should ob.gyns. go it alone, said Dr. Foley. “The conversation with the ob.gyn. needs to be about comanaging these patients, at the very least. Even the most learned maternal-fetal medicine specialist needs to be working with a cardiologist and an anesthesiologist to create a delivery plan that includes pain management, fluid management, and consideration for intrapartum hemodynamic monitoring,” he said.
And the team needs to be in place long before delivery, Dr. Foley pointed out. “In many hospitals, the care delivery gap may be the inability to have this consistent proactive approach. You can’t expect the best outcomes when you have to hurriedly assemble an unfamiliar ad hoc team when a woman with congenital heart disease presents in labor. Despite their best intentions, inconsistent team members may not have the knowledge and experience to provide the safest care for these patients,” he said.
Though an individualized labor and delivery plan is a must, and a multispecialty team should be assembled, maternal congenital heart disease doesn’t necessarily consign a woman to cesarean delivery. “Most women can and should have a vaginal delivery. It’s safer for them. If a natural delivery may increase risk of issues, we may consider a facilitated second stage of labor with epidural anesthesia and forceps- or vacuum-assisted delivery,” said Dr. Kay.
It’s important to understand the nuances of an individual patient’s health and risk status, said Dr. Norton. “A simplified view is often bad. It’s not the case that ‘it’s always better to deliver’ or ‘it’s always better to have a cesarean delivery.’”
Especially for women who need anticoagulation or who may have lesions that put them at great risk should pregnancy occur, preconception counseling is a vital part of their care, and guidance in the scientific statement can help specialists avoid the complications that can occur in the absence of evidence-based treatment. Said Dr. Kay, “I have seen an unfortunate case or two of patients whose anticoagulation was stopped or changed, contrary to guidelines, and who suffered strokes. I hope more people will see this document.”
Ms. Canobbio echoed the sentiment: “You don’t want to have to backpedal once a young woman presents with a pregnancy. Appropriate contraceptive counseling needs to be part of the conversation.”
One key concept underscored in the scientific statement is that elevated risk persists into the postpartum period. “Following delivery, the mother is still at risk for an extended period of time. The greatest risk for mortality in these patients is post delivery, when a large volume of blood is expelled from the uterus back into the maternal circulation,” said Ms. Canobbio. “These women need close follow-up; we can’t say they are home free until several weeks to 2 months after delivery. The need for vigilance and surveillance continues.”
Since the scientific statement is not a new set of guidelines, but rather a compilation of currently existing reference documents, the authors noted that management differences may exist in some cases, but did not assign greater value to one practice than another. “We addressed that there are differences between the European and the American guidelines. For example, with regard to anticoagulation, both would agree to use Lovenox [enoxaparin], but the difference is whether it should be used for the entire pregnancy or for parts of the pregnancy,” said Ms. Canobbio.
Looking forward, more women with complex congenital heart disease will bear children, but their future is not certain. Said Ms. Canobbio: “The data are growing that if the patient is clinically stable at the time of pregnancy, it’s likely we can get them through safely. What’s not yet known is whether the burden of pregnancy in a woman who is otherwise healthy will shorten her lifespan. However, early data are promising, and it’s looking like these women can fare well.”
Topics covered in the scientific statement include:
- Defining which patients are at increased risk in pregnancy.
- Physiological adaptations of pregnancy, the puerperium, and the postpartum period, with an emphasis on hemodynamic changes.
- Assessment and evaluation in the preconception and early prenatal periods.
- Pregnancy management, including appropriate testing.
- Medications in pregnancy, including a table of common cardiac drugs and their pregnancy categories and lactation risks.
- Breakdown of suggested prenatal care by trimester.
- Intrapartum care, including indications for fluid management, ECG and hemodynamic monitoring, and management of the second stage of delivery.
- Postpartum care, with attention to the very rapid increase in blood volume and concomitant leap in stroke volume and cardiac output.
- Considerations when choosing contraceptive method.
- Cardiac complications seen in pregnancy, including arrhythmias, managing mechanical valves and anticoagulation, heart failure, and cyanosis.
- Indications for and risks associated with interventional therapies during pregnancy.
- Detailed discussion of management of pregnancy for women with specific lesions.
None of the members of the writing committee for the scientific statement had relevant disclosures. Dr. Foley and Dr. Kay reported no disclosures. Dr. Norton reported that she has received research funding from Natera and Ultragenyx.
koakes@frontlinemedcom.com
On Twitter @karioakes
A new scientific statement from the American Heart Association (AHA) brings together recommendations for management of pregnancy for women with serious congenital heart disease. The 38-page document addresses a wide range of complex congenital heart conditions, presenting a newly unified set of recommendations for care that ranges from preconception counseling, through pregnancy, labor, and delivery, to the postpartum period.
Caring for women with complex congenital heart lesions is becoming more commonplace, as more infants undergo successful repairs of previously-unsurvivable cardiac anomalies. “More moms with congenital heart disease are showing up pregnant, having survived the tumultuous peripartum and neonatal period, and are now facing a new set of risks in pregnancy,” Michael Foley, MD, chair of the department of obstetrics and gynecology at the University of Arizona, Phoenix, said in an interview.
Joseph Kay, MD, a cardiologist and professor of medicine and pediatrics at the University of Colorado, Aurora, said that one big benefit of the new scientific statement is having a single reference point for care of these patients. “The scientific statement brings all of the information about caring for these patients together into one document. This will be a very valuable resource for trainees to get a sense of what’s important; it also represents a platform for new programs to understand the scope of services needed,” said Dr. Kay in an interview.
The document provides a thorough review of the physiologic changes of pregnancy and the intrapartum and postpartum periods, noting that the heterogeneity of congenital heart disease means that women who have different lesions carry different risks in pregnancy.
Examples of lesions presenting intermediate risk include most arrhythmias (category II), hypertrophic cardiomyopathy, and a repaired coarctation (both category II-III). The most severe lesions carry a contraindication for pregnancy; the WHO guidelines suggest discussing termination should women with a category IV lesion become pregnant. Severe mitral stenosis, severe symptomatic aortic stenosis, and severe systemic ventricular dysfunction all place women into category IV.
Beginning with pregnancy risk category III, the WHO guidelines recommend intensive cardiac and obstetric monitoring throughout pregnancy, childbirth, and the puerperium. Several maternal-fetal medicine specialists interviewed all agreed that an interdisciplinary team is a must for good obstetric care in this population.
How interdisciplinary care plays out can depend on geography and facility-dependent resources. Dr. Kay said that his facility is the referral site for pregnant women with complex congenital lesions in an area that spans the Canadian and the Mexican borders from north to south, and ranges from parts of Kansas to eastern Montana from east to west. Still, Dr. Kay said that even for patients with lower-risk lesions, “We will see patients at least once, at approximately the midpoint of pregnancy, and again during the third trimester if possible.” The specifics of care depend on “the nature of the lesion and the complexity of the disease,” said Dr. Kay.
In his facility, said Dr. Kay, telemetry is available for all of the labor and delivery unit beds. This means that the mother and infant can usually stay together and receive postpartum nursing and lactation care from a skilled staff.
In no circumstances should ob.gyns. go it alone, said Dr. Foley. “The conversation with the ob.gyn. needs to be about comanaging these patients, at the very least. Even the most learned maternal-fetal medicine specialist needs to be working with a cardiologist and an anesthesiologist to create a delivery plan that includes pain management, fluid management, and consideration for intrapartum hemodynamic monitoring,” he said.
And the team needs to be in place long before delivery, Dr. Foley pointed out. “In many hospitals, the care delivery gap may be the inability to have this consistent proactive approach. You can’t expect the best outcomes when you have to hurriedly assemble an unfamiliar ad hoc team when a woman with congenital heart disease presents in labor. Despite their best intentions, inconsistent team members may not have the knowledge and experience to provide the safest care for these patients,” he said.
Though an individualized labor and delivery plan is a must, and a multispecialty team should be assembled, maternal congenital heart disease doesn’t necessarily consign a woman to cesarean delivery. “Most women can and should have a vaginal delivery. It’s safer for them. If a natural delivery may increase risk of issues, we may consider a facilitated second stage of labor with epidural anesthesia and forceps- or vacuum-assisted delivery,” said Dr. Kay.
It’s important to understand the nuances of an individual patient’s health and risk status, said Dr. Norton. “A simplified view is often bad. It’s not the case that ‘it’s always better to deliver’ or ‘it’s always better to have a cesarean delivery.’”
Especially for women who need anticoagulation or who may have lesions that put them at great risk should pregnancy occur, preconception counseling is a vital part of their care, and guidance in the scientific statement can help specialists avoid the complications that can occur in the absence of evidence-based treatment. Said Dr. Kay, “I have seen an unfortunate case or two of patients whose anticoagulation was stopped or changed, contrary to guidelines, and who suffered strokes. I hope more people will see this document.”
Ms. Canobbio echoed the sentiment: “You don’t want to have to backpedal once a young woman presents with a pregnancy. Appropriate contraceptive counseling needs to be part of the conversation.”
One key concept underscored in the scientific statement is that elevated risk persists into the postpartum period. “Following delivery, the mother is still at risk for an extended period of time. The greatest risk for mortality in these patients is post delivery, when a large volume of blood is expelled from the uterus back into the maternal circulation,” said Ms. Canobbio. “These women need close follow-up; we can’t say they are home free until several weeks to 2 months after delivery. The need for vigilance and surveillance continues.”
Since the scientific statement is not a new set of guidelines, but rather a compilation of currently existing reference documents, the authors noted that management differences may exist in some cases, but did not assign greater value to one practice than another. “We addressed that there are differences between the European and the American guidelines. For example, with regard to anticoagulation, both would agree to use Lovenox [enoxaparin], but the difference is whether it should be used for the entire pregnancy or for parts of the pregnancy,” said Ms. Canobbio.
Looking forward, more women with complex congenital heart disease will bear children, but their future is not certain. Said Ms. Canobbio: “The data are growing that if the patient is clinically stable at the time of pregnancy, it’s likely we can get them through safely. What’s not yet known is whether the burden of pregnancy in a woman who is otherwise healthy will shorten her lifespan. However, early data are promising, and it’s looking like these women can fare well.”
Topics covered in the scientific statement include:
- Defining which patients are at increased risk in pregnancy.
- Physiological adaptations of pregnancy, the puerperium, and the postpartum period, with an emphasis on hemodynamic changes.
- Assessment and evaluation in the preconception and early prenatal periods.
- Pregnancy management, including appropriate testing.
- Medications in pregnancy, including a table of common cardiac drugs and their pregnancy categories and lactation risks.
- Breakdown of suggested prenatal care by trimester.
- Intrapartum care, including indications for fluid management, ECG and hemodynamic monitoring, and management of the second stage of delivery.
- Postpartum care, with attention to the very rapid increase in blood volume and concomitant leap in stroke volume and cardiac output.
- Considerations when choosing contraceptive method.
- Cardiac complications seen in pregnancy, including arrhythmias, managing mechanical valves and anticoagulation, heart failure, and cyanosis.
- Indications for and risks associated with interventional therapies during pregnancy.
- Detailed discussion of management of pregnancy for women with specific lesions.
None of the members of the writing committee for the scientific statement had relevant disclosures. Dr. Foley and Dr. Kay reported no disclosures. Dr. Norton reported that she has received research funding from Natera and Ultragenyx.
koakes@frontlinemedcom.com
On Twitter @karioakes
A new scientific statement from the American Heart Association (AHA) brings together recommendations for management of pregnancy for women with serious congenital heart disease. The 38-page document addresses a wide range of complex congenital heart conditions, presenting a newly unified set of recommendations for care that ranges from preconception counseling, through pregnancy, labor, and delivery, to the postpartum period.
Caring for women with complex congenital heart lesions is becoming more commonplace, as more infants undergo successful repairs of previously-unsurvivable cardiac anomalies. “More moms with congenital heart disease are showing up pregnant, having survived the tumultuous peripartum and neonatal period, and are now facing a new set of risks in pregnancy,” Michael Foley, MD, chair of the department of obstetrics and gynecology at the University of Arizona, Phoenix, said in an interview.
Joseph Kay, MD, a cardiologist and professor of medicine and pediatrics at the University of Colorado, Aurora, said that one big benefit of the new scientific statement is having a single reference point for care of these patients. “The scientific statement brings all of the information about caring for these patients together into one document. This will be a very valuable resource for trainees to get a sense of what’s important; it also represents a platform for new programs to understand the scope of services needed,” said Dr. Kay in an interview.
The document provides a thorough review of the physiologic changes of pregnancy and the intrapartum and postpartum periods, noting that the heterogeneity of congenital heart disease means that women who have different lesions carry different risks in pregnancy.
Examples of lesions presenting intermediate risk include most arrhythmias (category II), hypertrophic cardiomyopathy, and a repaired coarctation (both category II-III). The most severe lesions carry a contraindication for pregnancy; the WHO guidelines suggest discussing termination should women with a category IV lesion become pregnant. Severe mitral stenosis, severe symptomatic aortic stenosis, and severe systemic ventricular dysfunction all place women into category IV.
Beginning with pregnancy risk category III, the WHO guidelines recommend intensive cardiac and obstetric monitoring throughout pregnancy, childbirth, and the puerperium. Several maternal-fetal medicine specialists interviewed all agreed that an interdisciplinary team is a must for good obstetric care in this population.
How interdisciplinary care plays out can depend on geography and facility-dependent resources. Dr. Kay said that his facility is the referral site for pregnant women with complex congenital lesions in an area that spans the Canadian and the Mexican borders from north to south, and ranges from parts of Kansas to eastern Montana from east to west. Still, Dr. Kay said that even for patients with lower-risk lesions, “We will see patients at least once, at approximately the midpoint of pregnancy, and again during the third trimester if possible.” The specifics of care depend on “the nature of the lesion and the complexity of the disease,” said Dr. Kay.
In his facility, said Dr. Kay, telemetry is available for all of the labor and delivery unit beds. This means that the mother and infant can usually stay together and receive postpartum nursing and lactation care from a skilled staff.
In no circumstances should ob.gyns. go it alone, said Dr. Foley. “The conversation with the ob.gyn. needs to be about comanaging these patients, at the very least. Even the most learned maternal-fetal medicine specialist needs to be working with a cardiologist and an anesthesiologist to create a delivery plan that includes pain management, fluid management, and consideration for intrapartum hemodynamic monitoring,” he said.
And the team needs to be in place long before delivery, Dr. Foley pointed out. “In many hospitals, the care delivery gap may be the inability to have this consistent proactive approach. You can’t expect the best outcomes when you have to hurriedly assemble an unfamiliar ad hoc team when a woman with congenital heart disease presents in labor. Despite their best intentions, inconsistent team members may not have the knowledge and experience to provide the safest care for these patients,” he said.
Though an individualized labor and delivery plan is a must, and a multispecialty team should be assembled, maternal congenital heart disease doesn’t necessarily consign a woman to cesarean delivery. “Most women can and should have a vaginal delivery. It’s safer for them. If a natural delivery may increase risk of issues, we may consider a facilitated second stage of labor with epidural anesthesia and forceps- or vacuum-assisted delivery,” said Dr. Kay.
It’s important to understand the nuances of an individual patient’s health and risk status, said Dr. Norton. “A simplified view is often bad. It’s not the case that ‘it’s always better to deliver’ or ‘it’s always better to have a cesarean delivery.’”
Especially for women who need anticoagulation or who may have lesions that put them at great risk should pregnancy occur, preconception counseling is a vital part of their care, and guidance in the scientific statement can help specialists avoid the complications that can occur in the absence of evidence-based treatment. Said Dr. Kay, “I have seen an unfortunate case or two of patients whose anticoagulation was stopped or changed, contrary to guidelines, and who suffered strokes. I hope more people will see this document.”
Ms. Canobbio echoed the sentiment: “You don’t want to have to backpedal once a young woman presents with a pregnancy. Appropriate contraceptive counseling needs to be part of the conversation.”
One key concept underscored in the scientific statement is that elevated risk persists into the postpartum period. “Following delivery, the mother is still at risk for an extended period of time. The greatest risk for mortality in these patients is post delivery, when a large volume of blood is expelled from the uterus back into the maternal circulation,” said Ms. Canobbio. “These women need close follow-up; we can’t say they are home free until several weeks to 2 months after delivery. The need for vigilance and surveillance continues.”
Since the scientific statement is not a new set of guidelines, but rather a compilation of currently existing reference documents, the authors noted that management differences may exist in some cases, but did not assign greater value to one practice than another. “We addressed that there are differences between the European and the American guidelines. For example, with regard to anticoagulation, both would agree to use Lovenox [enoxaparin], but the difference is whether it should be used for the entire pregnancy or for parts of the pregnancy,” said Ms. Canobbio.
Looking forward, more women with complex congenital heart disease will bear children, but their future is not certain. Said Ms. Canobbio: “The data are growing that if the patient is clinically stable at the time of pregnancy, it’s likely we can get them through safely. What’s not yet known is whether the burden of pregnancy in a woman who is otherwise healthy will shorten her lifespan. However, early data are promising, and it’s looking like these women can fare well.”
Topics covered in the scientific statement include:
- Defining which patients are at increased risk in pregnancy.
- Physiological adaptations of pregnancy, the puerperium, and the postpartum period, with an emphasis on hemodynamic changes.
- Assessment and evaluation in the preconception and early prenatal periods.
- Pregnancy management, including appropriate testing.
- Medications in pregnancy, including a table of common cardiac drugs and their pregnancy categories and lactation risks.
- Breakdown of suggested prenatal care by trimester.
- Intrapartum care, including indications for fluid management, ECG and hemodynamic monitoring, and management of the second stage of delivery.
- Postpartum care, with attention to the very rapid increase in blood volume and concomitant leap in stroke volume and cardiac output.
- Considerations when choosing contraceptive method.
- Cardiac complications seen in pregnancy, including arrhythmias, managing mechanical valves and anticoagulation, heart failure, and cyanosis.
- Indications for and risks associated with interventional therapies during pregnancy.
- Detailed discussion of management of pregnancy for women with specific lesions.
None of the members of the writing committee for the scientific statement had relevant disclosures. Dr. Foley and Dr. Kay reported no disclosures. Dr. Norton reported that she has received research funding from Natera and Ultragenyx.
koakes@frontlinemedcom.com
On Twitter @karioakes
Curb AF recurrences through risk factor modification
SNOWMASS, COLO. – Overlooking the common modifiable risk factors in patients with atrial fibrillation is missing out on an excellent opportunity to help curb the growing global pandemic of the arrhythmia, Patrick T. O’Gara, MD, said at the Annual Cardiovascular Conference at Snowmass.
“My purpose here is a wake up call to improve screening for and treatment of modifiable risk factors in patients with atrial fibrillation,” declared Dr. O’Gara, professor of medicine at Harvard Medical School, Boston.
Overweight/obesity: Investigators at the University of Adelaide (Australia) demonstrated in the LEGACY trial that patients with atrial fibrillation (AF) and a BMI of 27 kg/m2 or more reduced their AF symptom burden in a dose-response fashion as they shed excess pounds as part of an intensive weight management program. Those who shed at least 10% of their baseline body weight had a 46% rate of 5-year freedom from AF without resort to rhythm control medications or ablation procedures of 46%. With 3%-9% weight loss, the rate was 22%. And with 3% weight loss, it was 13%.
The best results came from sustained linear weight loss. Weight fluctuations of greater than 5% – the classic yoyo dieting pattern – partially offset the overall benefit of weight loss with respect to recurrent AF (J Am Coll Cardiol. 2015 May 26;65(20):2159-69).
In a separate study, the same team of Australian investigators offered an opportunity to participate in a risk factor management program to patients with AF and a BMI of 27 kg/m2 or more who were undergoing radiofrequency ablation for their arrhythmia. Participants had significantly fewer repeat ablation procedures during followup and were also less likely to be on antiarrhythmic drugs than the patients who opted for usual care (J Am Coll Cardiol. 2014 Dec 2;64(21):2222-31).
Alcohol: The ‘holiday heart’ syndrome is well known, but alcohol consumption beyond binging can increase risk for AF. Dr. O’Gara noted that in a recent review article entitled “Alcohol and Atrial Fibrillation: A Sobering Review,” investigators at the University of Melbourne showed that while the relationship between the number of standard drinks per week and risk of cardiovascular mortality is J-shaped, with a nadir at 14-21 drinks per week in men and fewer in women, the risk of developing AF is linear over time and appears to increase incrementally with every additional drink per week (J Am Coll Cardiol. 2016 Dec 13;68(23):2567-76).
Also, a prospective study of nearly 80,000 Swedes free from AF at baseline, coupled with a meta-analysis of seven prospective studies found that for each additional drink per day consumed the risk of developing AF rose over time by roughly a further 10% compared to that of teetotalers (J Am Coll Cardiol. 2014; Jul 22;64(3):281-9).
Physical inactivity: In the prospective Tromso Study, in which more than 20,000 Norwegian adults were followed for 20 years, leisure time physical activity displayed a J-shaped relationship with the risk of developing AF. Moderately active subjects were an adjusted 19% less likely to develop AF than those with low physical activity, while the risk in subjects who regularly engaged in vigorous physical activity was 37% higher than in the low-activity group (Eur Heart J. 2016 Aug 1;37(29):2307-13).
“This effect of moderate exercise might be due to the associated weight loss, improved endothelial function, better sleep, perhaps a better balance between the sympathetic and parasympathetic nervous systems,” Dr. O’Gara observed.
How much physical activity is right for patients with AF? Dr. O’Gara said one of the best reviews he’s seen came from the University of Adelaide group (Circulation. 2016 Feb 2;133(5):457-9). They recommended a total of 120-200 minutes of exercise per week spread over three to five sessions. While the research base is strongest for moderate-intensity exercise, the Australians also noted the effectiveness and safety of a novel program of repeated 4-minute intervals of high-intensity exercise at 85%-95% of peak heart rate as demonstrated in a randomized controlled trial by investigators at the Norwegian University of Science and Technology in Trondheim. They showed this approach resulted in reduced time in AF and decreased AF symptoms coupled with improved quality of life and left atrial and ventricular function (Circulation. 2016 Feb 2;133(5):466-73).
“I think you could look at this review and feel very confident that there is some evidence base to substantiate your strong recommendation that patients actively engage in exercise as treatment for their atrial fibrillation,” the cardiologist said.
Sleep apnea: Investigators at Brigham and Women’s Hospital in Boston have demonstrated that effective treatment of sleep apnea with continuous positive airway pressure in patients with atrial fibrillation is associated with smaller atrial size and ventricular mass, lower blood pressure, and a significantly reduced risk of recurrent AF following an AF ablation procedure (J Am Heart Assoc. 2013 Nov 25;2(6):e000421).
“Sleep hygiene is one of the least attended aspects of cardiovascular health,” according to Dr. O’Gara. “We need to ask the partner or spouse, ‘How well does your partner sleep? Do you hear thrashing about, snoring, gagging, or notice restless legs?’ Heart failure folks are really tuned into this, but in the practice of seeing patients come into the emergency room with new-onset atrial fibrillation, it’s tenth on the list of five questions one would ask.”
Dr. O’Gara reported having no financial conflicts.
SNOWMASS, COLO. – Overlooking the common modifiable risk factors in patients with atrial fibrillation is missing out on an excellent opportunity to help curb the growing global pandemic of the arrhythmia, Patrick T. O’Gara, MD, said at the Annual Cardiovascular Conference at Snowmass.
“My purpose here is a wake up call to improve screening for and treatment of modifiable risk factors in patients with atrial fibrillation,” declared Dr. O’Gara, professor of medicine at Harvard Medical School, Boston.
Overweight/obesity: Investigators at the University of Adelaide (Australia) demonstrated in the LEGACY trial that patients with atrial fibrillation (AF) and a BMI of 27 kg/m2 or more reduced their AF symptom burden in a dose-response fashion as they shed excess pounds as part of an intensive weight management program. Those who shed at least 10% of their baseline body weight had a 46% rate of 5-year freedom from AF without resort to rhythm control medications or ablation procedures of 46%. With 3%-9% weight loss, the rate was 22%. And with 3% weight loss, it was 13%.
The best results came from sustained linear weight loss. Weight fluctuations of greater than 5% – the classic yoyo dieting pattern – partially offset the overall benefit of weight loss with respect to recurrent AF (J Am Coll Cardiol. 2015 May 26;65(20):2159-69).
In a separate study, the same team of Australian investigators offered an opportunity to participate in a risk factor management program to patients with AF and a BMI of 27 kg/m2 or more who were undergoing radiofrequency ablation for their arrhythmia. Participants had significantly fewer repeat ablation procedures during followup and were also less likely to be on antiarrhythmic drugs than the patients who opted for usual care (J Am Coll Cardiol. 2014 Dec 2;64(21):2222-31).
Alcohol: The ‘holiday heart’ syndrome is well known, but alcohol consumption beyond binging can increase risk for AF. Dr. O’Gara noted that in a recent review article entitled “Alcohol and Atrial Fibrillation: A Sobering Review,” investigators at the University of Melbourne showed that while the relationship between the number of standard drinks per week and risk of cardiovascular mortality is J-shaped, with a nadir at 14-21 drinks per week in men and fewer in women, the risk of developing AF is linear over time and appears to increase incrementally with every additional drink per week (J Am Coll Cardiol. 2016 Dec 13;68(23):2567-76).
Also, a prospective study of nearly 80,000 Swedes free from AF at baseline, coupled with a meta-analysis of seven prospective studies found that for each additional drink per day consumed the risk of developing AF rose over time by roughly a further 10% compared to that of teetotalers (J Am Coll Cardiol. 2014; Jul 22;64(3):281-9).
Physical inactivity: In the prospective Tromso Study, in which more than 20,000 Norwegian adults were followed for 20 years, leisure time physical activity displayed a J-shaped relationship with the risk of developing AF. Moderately active subjects were an adjusted 19% less likely to develop AF than those with low physical activity, while the risk in subjects who regularly engaged in vigorous physical activity was 37% higher than in the low-activity group (Eur Heart J. 2016 Aug 1;37(29):2307-13).
“This effect of moderate exercise might be due to the associated weight loss, improved endothelial function, better sleep, perhaps a better balance between the sympathetic and parasympathetic nervous systems,” Dr. O’Gara observed.
How much physical activity is right for patients with AF? Dr. O’Gara said one of the best reviews he’s seen came from the University of Adelaide group (Circulation. 2016 Feb 2;133(5):457-9). They recommended a total of 120-200 minutes of exercise per week spread over three to five sessions. While the research base is strongest for moderate-intensity exercise, the Australians also noted the effectiveness and safety of a novel program of repeated 4-minute intervals of high-intensity exercise at 85%-95% of peak heart rate as demonstrated in a randomized controlled trial by investigators at the Norwegian University of Science and Technology in Trondheim. They showed this approach resulted in reduced time in AF and decreased AF symptoms coupled with improved quality of life and left atrial and ventricular function (Circulation. 2016 Feb 2;133(5):466-73).
“I think you could look at this review and feel very confident that there is some evidence base to substantiate your strong recommendation that patients actively engage in exercise as treatment for their atrial fibrillation,” the cardiologist said.
Sleep apnea: Investigators at Brigham and Women’s Hospital in Boston have demonstrated that effective treatment of sleep apnea with continuous positive airway pressure in patients with atrial fibrillation is associated with smaller atrial size and ventricular mass, lower blood pressure, and a significantly reduced risk of recurrent AF following an AF ablation procedure (J Am Heart Assoc. 2013 Nov 25;2(6):e000421).
“Sleep hygiene is one of the least attended aspects of cardiovascular health,” according to Dr. O’Gara. “We need to ask the partner or spouse, ‘How well does your partner sleep? Do you hear thrashing about, snoring, gagging, or notice restless legs?’ Heart failure folks are really tuned into this, but in the practice of seeing patients come into the emergency room with new-onset atrial fibrillation, it’s tenth on the list of five questions one would ask.”
Dr. O’Gara reported having no financial conflicts.
SNOWMASS, COLO. – Overlooking the common modifiable risk factors in patients with atrial fibrillation is missing out on an excellent opportunity to help curb the growing global pandemic of the arrhythmia, Patrick T. O’Gara, MD, said at the Annual Cardiovascular Conference at Snowmass.
“My purpose here is a wake up call to improve screening for and treatment of modifiable risk factors in patients with atrial fibrillation,” declared Dr. O’Gara, professor of medicine at Harvard Medical School, Boston.
Overweight/obesity: Investigators at the University of Adelaide (Australia) demonstrated in the LEGACY trial that patients with atrial fibrillation (AF) and a BMI of 27 kg/m2 or more reduced their AF symptom burden in a dose-response fashion as they shed excess pounds as part of an intensive weight management program. Those who shed at least 10% of their baseline body weight had a 46% rate of 5-year freedom from AF without resort to rhythm control medications or ablation procedures of 46%. With 3%-9% weight loss, the rate was 22%. And with 3% weight loss, it was 13%.
The best results came from sustained linear weight loss. Weight fluctuations of greater than 5% – the classic yoyo dieting pattern – partially offset the overall benefit of weight loss with respect to recurrent AF (J Am Coll Cardiol. 2015 May 26;65(20):2159-69).
In a separate study, the same team of Australian investigators offered an opportunity to participate in a risk factor management program to patients with AF and a BMI of 27 kg/m2 or more who were undergoing radiofrequency ablation for their arrhythmia. Participants had significantly fewer repeat ablation procedures during followup and were also less likely to be on antiarrhythmic drugs than the patients who opted for usual care (J Am Coll Cardiol. 2014 Dec 2;64(21):2222-31).
Alcohol: The ‘holiday heart’ syndrome is well known, but alcohol consumption beyond binging can increase risk for AF. Dr. O’Gara noted that in a recent review article entitled “Alcohol and Atrial Fibrillation: A Sobering Review,” investigators at the University of Melbourne showed that while the relationship between the number of standard drinks per week and risk of cardiovascular mortality is J-shaped, with a nadir at 14-21 drinks per week in men and fewer in women, the risk of developing AF is linear over time and appears to increase incrementally with every additional drink per week (J Am Coll Cardiol. 2016 Dec 13;68(23):2567-76).
Also, a prospective study of nearly 80,000 Swedes free from AF at baseline, coupled with a meta-analysis of seven prospective studies found that for each additional drink per day consumed the risk of developing AF rose over time by roughly a further 10% compared to that of teetotalers (J Am Coll Cardiol. 2014; Jul 22;64(3):281-9).
Physical inactivity: In the prospective Tromso Study, in which more than 20,000 Norwegian adults were followed for 20 years, leisure time physical activity displayed a J-shaped relationship with the risk of developing AF. Moderately active subjects were an adjusted 19% less likely to develop AF than those with low physical activity, while the risk in subjects who regularly engaged in vigorous physical activity was 37% higher than in the low-activity group (Eur Heart J. 2016 Aug 1;37(29):2307-13).
“This effect of moderate exercise might be due to the associated weight loss, improved endothelial function, better sleep, perhaps a better balance between the sympathetic and parasympathetic nervous systems,” Dr. O’Gara observed.
How much physical activity is right for patients with AF? Dr. O’Gara said one of the best reviews he’s seen came from the University of Adelaide group (Circulation. 2016 Feb 2;133(5):457-9). They recommended a total of 120-200 minutes of exercise per week spread over three to five sessions. While the research base is strongest for moderate-intensity exercise, the Australians also noted the effectiveness and safety of a novel program of repeated 4-minute intervals of high-intensity exercise at 85%-95% of peak heart rate as demonstrated in a randomized controlled trial by investigators at the Norwegian University of Science and Technology in Trondheim. They showed this approach resulted in reduced time in AF and decreased AF symptoms coupled with improved quality of life and left atrial and ventricular function (Circulation. 2016 Feb 2;133(5):466-73).
“I think you could look at this review and feel very confident that there is some evidence base to substantiate your strong recommendation that patients actively engage in exercise as treatment for their atrial fibrillation,” the cardiologist said.
Sleep apnea: Investigators at Brigham and Women’s Hospital in Boston have demonstrated that effective treatment of sleep apnea with continuous positive airway pressure in patients with atrial fibrillation is associated with smaller atrial size and ventricular mass, lower blood pressure, and a significantly reduced risk of recurrent AF following an AF ablation procedure (J Am Heart Assoc. 2013 Nov 25;2(6):e000421).
“Sleep hygiene is one of the least attended aspects of cardiovascular health,” according to Dr. O’Gara. “We need to ask the partner or spouse, ‘How well does your partner sleep? Do you hear thrashing about, snoring, gagging, or notice restless legs?’ Heart failure folks are really tuned into this, but in the practice of seeing patients come into the emergency room with new-onset atrial fibrillation, it’s tenth on the list of five questions one would ask.”
Dr. O’Gara reported having no financial conflicts.
EXPERT ANALYSIS FROM THE CARDIOVASCULAR CONFERENCE AT SNOWMASS
Cardiovascular complications in pregnancy quickly boost future risk
NEW ORLEANS – Women who experience peripartum cardiomyopathy or any of a variety of hypertensive disorders in pregnancy are at sharply increased risk of acute MI, stroke, or new-onset heart failure beginning within just a few years, Rima Arnaout, MD, reported at the American Heart Association scientific sessions.
“Our study supports the idea that women who have cardiovascular complications in pregnancy really need to be monitored closely for potential primary prevention of cardiovascular events,” said Dr. Arnaout of the University of California, San Francisco.
At a session devoted to “big data” studies in cardiovascular medicine, she presented one of the biggest: a retrospective cohort study of 1.66 million pregnancies during 2005-2009 in California women without any history of congenital or valvular heart disease or prepregnancy cardiovascular events. The California database was created as part of the U.S. Agency for Healthcare Research and Quality’s comprehensive Healthcare Cost and Utilization Project, which included more than 95% of the state’s hospitals. Women who experienced an MI or a stroke, or who were diagnosed with heart failure, during a median of 2.7 years and a maximum of 6 years of follow-up post pregnancy were identified via ICD-9 codes.
There were 111,202 cases of various forms of hypertension in pregnancy, for a 6.9% incidence. Peripartum cardiomyopathy was diagnosed in 562 women, for a rate of 3.5 cases per 10,000 pregnancies.
Indeed, in a multivariate Cox proportional hazards analysis adjusted for numerous potential confounders, peripartum cardiomyopathy was associated with a 16-fold increased risk of acute MI during the relatively short follow-up period, as well as 29-fold increased risks of stroke and heart failure, compared with women with no cardiovascular issues during their pregnancy.
Chronic hypertension, regardless of whether it occurred alone or in combination with preeclampsia or gestational hypertension, was associated with roughly a twofold increased risk of each of the three study outcomes, compared with women who didn’t experience a cardiovascular complication during pregnancy. De novo preeclampsia was also associated with roughly a twofold increased risk of later MI, heart failure, or stroke.
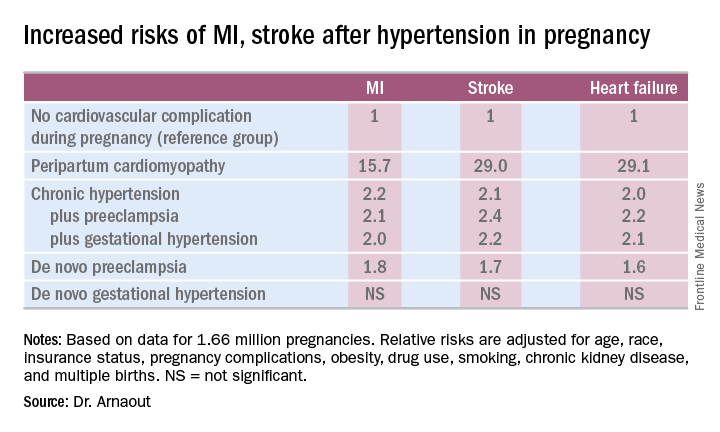
The only form of hypertension in pregnancy that wasn’t associated with a subsequent significantly increased risk of cardiovascular events was de novo gestational hypertension.
Audience member David C. Goff Jr., MD, head of the division of cardiovascular sciences at the National Heart, Lung, and Blood Institute in Bethesda, Md., rose to compliment Dr. Arnaout: “Great work and really important.”
He said that her findings are consistent with the notion that pregnancy constitutes a sort of early-life cardiovascular stress test. He said he wondered, however, just how comfortable Dr. Arnaout is in stating that gestational diabetes isn’t associated with increased subsequent cardiovascular risk, given the relatively short follow-up to date in this population of women who still remain several decades away from the age when cardiovascular event rates really start to climb.
“I completely agree with you,” she replied, noting that other investigators utilizing a different registry have reported an increased longer-term risk for women with gestational diabetes.
Dr. Arnaout said she and her coinvestigators plan to continue to follow the women who experienced peripartum cardiomyopathy or hypertension in pregnancy longer term. They’re also in the process of breaking down the data to look at the risks associated with specific subtypes of MI, stroke, and heart failure.
Dr. Arnaout reported having no financial conflicts regarding her study, which was supported by the American Heart Association and the Sarnoff Cardiovascular Research Foundation.
NEW ORLEANS – Women who experience peripartum cardiomyopathy or any of a variety of hypertensive disorders in pregnancy are at sharply increased risk of acute MI, stroke, or new-onset heart failure beginning within just a few years, Rima Arnaout, MD, reported at the American Heart Association scientific sessions.
“Our study supports the idea that women who have cardiovascular complications in pregnancy really need to be monitored closely for potential primary prevention of cardiovascular events,” said Dr. Arnaout of the University of California, San Francisco.
At a session devoted to “big data” studies in cardiovascular medicine, she presented one of the biggest: a retrospective cohort study of 1.66 million pregnancies during 2005-2009 in California women without any history of congenital or valvular heart disease or prepregnancy cardiovascular events. The California database was created as part of the U.S. Agency for Healthcare Research and Quality’s comprehensive Healthcare Cost and Utilization Project, which included more than 95% of the state’s hospitals. Women who experienced an MI or a stroke, or who were diagnosed with heart failure, during a median of 2.7 years and a maximum of 6 years of follow-up post pregnancy were identified via ICD-9 codes.
There were 111,202 cases of various forms of hypertension in pregnancy, for a 6.9% incidence. Peripartum cardiomyopathy was diagnosed in 562 women, for a rate of 3.5 cases per 10,000 pregnancies.
Indeed, in a multivariate Cox proportional hazards analysis adjusted for numerous potential confounders, peripartum cardiomyopathy was associated with a 16-fold increased risk of acute MI during the relatively short follow-up period, as well as 29-fold increased risks of stroke and heart failure, compared with women with no cardiovascular issues during their pregnancy.
Chronic hypertension, regardless of whether it occurred alone or in combination with preeclampsia or gestational hypertension, was associated with roughly a twofold increased risk of each of the three study outcomes, compared with women who didn’t experience a cardiovascular complication during pregnancy. De novo preeclampsia was also associated with roughly a twofold increased risk of later MI, heart failure, or stroke.

The only form of hypertension in pregnancy that wasn’t associated with a subsequent significantly increased risk of cardiovascular events was de novo gestational hypertension.
Audience member David C. Goff Jr., MD, head of the division of cardiovascular sciences at the National Heart, Lung, and Blood Institute in Bethesda, Md., rose to compliment Dr. Arnaout: “Great work and really important.”
He said that her findings are consistent with the notion that pregnancy constitutes a sort of early-life cardiovascular stress test. He said he wondered, however, just how comfortable Dr. Arnaout is in stating that gestational diabetes isn’t associated with increased subsequent cardiovascular risk, given the relatively short follow-up to date in this population of women who still remain several decades away from the age when cardiovascular event rates really start to climb.
“I completely agree with you,” she replied, noting that other investigators utilizing a different registry have reported an increased longer-term risk for women with gestational diabetes.
Dr. Arnaout said she and her coinvestigators plan to continue to follow the women who experienced peripartum cardiomyopathy or hypertension in pregnancy longer term. They’re also in the process of breaking down the data to look at the risks associated with specific subtypes of MI, stroke, and heart failure.
Dr. Arnaout reported having no financial conflicts regarding her study, which was supported by the American Heart Association and the Sarnoff Cardiovascular Research Foundation.
NEW ORLEANS – Women who experience peripartum cardiomyopathy or any of a variety of hypertensive disorders in pregnancy are at sharply increased risk of acute MI, stroke, or new-onset heart failure beginning within just a few years, Rima Arnaout, MD, reported at the American Heart Association scientific sessions.
“Our study supports the idea that women who have cardiovascular complications in pregnancy really need to be monitored closely for potential primary prevention of cardiovascular events,” said Dr. Arnaout of the University of California, San Francisco.
At a session devoted to “big data” studies in cardiovascular medicine, she presented one of the biggest: a retrospective cohort study of 1.66 million pregnancies during 2005-2009 in California women without any history of congenital or valvular heart disease or prepregnancy cardiovascular events. The California database was created as part of the U.S. Agency for Healthcare Research and Quality’s comprehensive Healthcare Cost and Utilization Project, which included more than 95% of the state’s hospitals. Women who experienced an MI or a stroke, or who were diagnosed with heart failure, during a median of 2.7 years and a maximum of 6 years of follow-up post pregnancy were identified via ICD-9 codes.
There were 111,202 cases of various forms of hypertension in pregnancy, for a 6.9% incidence. Peripartum cardiomyopathy was diagnosed in 562 women, for a rate of 3.5 cases per 10,000 pregnancies.
Indeed, in a multivariate Cox proportional hazards analysis adjusted for numerous potential confounders, peripartum cardiomyopathy was associated with a 16-fold increased risk of acute MI during the relatively short follow-up period, as well as 29-fold increased risks of stroke and heart failure, compared with women with no cardiovascular issues during their pregnancy.
Chronic hypertension, regardless of whether it occurred alone or in combination with preeclampsia or gestational hypertension, was associated with roughly a twofold increased risk of each of the three study outcomes, compared with women who didn’t experience a cardiovascular complication during pregnancy. De novo preeclampsia was also associated with roughly a twofold increased risk of later MI, heart failure, or stroke.

The only form of hypertension in pregnancy that wasn’t associated with a subsequent significantly increased risk of cardiovascular events was de novo gestational hypertension.
Audience member David C. Goff Jr., MD, head of the division of cardiovascular sciences at the National Heart, Lung, and Blood Institute in Bethesda, Md., rose to compliment Dr. Arnaout: “Great work and really important.”
He said that her findings are consistent with the notion that pregnancy constitutes a sort of early-life cardiovascular stress test. He said he wondered, however, just how comfortable Dr. Arnaout is in stating that gestational diabetes isn’t associated with increased subsequent cardiovascular risk, given the relatively short follow-up to date in this population of women who still remain several decades away from the age when cardiovascular event rates really start to climb.
“I completely agree with you,” she replied, noting that other investigators utilizing a different registry have reported an increased longer-term risk for women with gestational diabetes.
Dr. Arnaout said she and her coinvestigators plan to continue to follow the women who experienced peripartum cardiomyopathy or hypertension in pregnancy longer term. They’re also in the process of breaking down the data to look at the risks associated with specific subtypes of MI, stroke, and heart failure.
Dr. Arnaout reported having no financial conflicts regarding her study, which was supported by the American Heart Association and the Sarnoff Cardiovascular Research Foundation.
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point:
Major finding: California women who developed peripartum cardiomyopathy were at 16- to 29-fold increased risk of experiencing an acute MI, stroke, or heart failure during a median 2.7 years of follow-up.
Data source: A retrospective cohort study of more than 95% of pregnancies in California during 2005-2009.
Disclosures: The presenter reported having no financial conflicts regarding her study, which was supported by the American Heart Association and the Sarnoff Cardiovascular Research Foundation.
Options narrow for acute decompensated heart failure
Acute decompensated heart failure is a condition that clinicians want to prevent rather than treat.
The idea that patients hospitalized with acute decompensated heart failure can have a substantial change in their prognosis from an acute intervention given in the hospital seemed to finally hit a brick wall in November at the American Heart Association scientific sessions.
Treatment of acute heart failure patients with the vasodilating natriuretic peptide ularitide failed to cut long-term cardiovascular mortality or improve several other acute and mid-term outcomes in the TRUE-AHF trial. In a second report, ATHENA-HF, acute treatment with high-dose spironolactone during acute heart failure hospitalizations failed to improve a marker of heart failure severity, N-terminal pro B-type natriuretic peptide.
What alternative interventions are left? Dr. Yancy, as well as Milton Packer, MD, the cardiologist who led TRUE-HF, had somewhat similar answers.
In his discussion of TRUE-AHF, Dr. Yancy cited two possibilities: greater use of the relatively new drug formulation sacubitril plus valsartan (Entresto), which showed strong benefit treating patients with chronic heart failure with reduced ejection fraction (HFrEF); and expanded use of pulmonary-artery pressure (PAP) monitoring using an implanted device that gives clinicians early warning when a heart failure patient’s fluid volume moves into the danger zone that precedes by days or weeks the acute decompensation that produces dyspnea and drives a patient to the hospital.
Increasing experience with PAP monitoring “continues to endorse the notion that having early warning [of fluid overload] is important,” Dr. Yancy said in an interview. “The point of acuity in acute decompensated heart failure predates hospital admission, and this monitoring system allows us to catch this before it causes an emergency department visit. The data are very persuasive.”
“PAP usually rises 2-4 weeks before a heart failure hospitalization. Perhaps we need to focus more on long-term treatment rather than treatment in the hospital.” That’s especially true for patients with heart failure with preserved ejection fraction (HFpEF) because right now no treatment is clearly proven to improve HFpEF outcomes (although several experts make a persuasive case for spironolactone).
“Tight regulation of volume status is our best chance to improve outcomes in HFpEF,” said Dr. Stevenson, who helped pioneer the idea of PAP monitoring for heart failure patients. “Maintaining good volume is very important for HFpEF patients.”
But success with PAP monitoring requires more than gathering the pressure data. Producing benefit for patients “is predicated on having an infrastructure to accommodate the influx of PAP data, being nimble enough to respond to the data and being very precise about which patients you use this in,” cautioned Dr. Yancy. “The tool is not beneficial if the infrastructure is not there,”
The idea is still so new (the first implanted PAP monitor received U.S. approval in 2014) that at his institution, Northwestern Medicine in Chicago, about a dozen patients now have a monitor, he said.
“We’re looking to use it in patients with HFpEF, whom we can’t offer anything else. I’m intrigued by what I see,” in this first wave of Northwestern patients. Dr. Yancy’s anecdotal experience with PAP monitoring so far “helps endorse what the trial results suggested” about providing incremental benefit to heart failure patients.
“The solution is to prevent hospitalization in the first place with the medications we already have, but they’re not used. The medications we already have are enough, but they need to be used,” Dr. Packer told me in an interview during the meeting. He speculated that perhaps 10%-15% of HFrEF patients currently receive the full guideline-directed regimen of heart failure drugs. “That’s unbelievable,” he exclaimed. If clinicians diligently treated advanced HFrEF patients with these four agents, “you’d see a 60%, 70% drop in hospitalizations,” he suggested.
Dr. Packer put some of the blame for underuse of guideline-directed medications on the low financial incentive physicians have to vigorously apply this strategy.
He backed PAP monitoring as a good additional step for selected patients with unstable heart failure. “But as a general approach to managing patients with class III HFrEF, first put them on optimal medical therapy; then we can talk about an invasive procedure to place a PAP monitoring device.”
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Acute decompensated heart failure is a condition that clinicians want to prevent rather than treat.
The idea that patients hospitalized with acute decompensated heart failure can have a substantial change in their prognosis from an acute intervention given in the hospital seemed to finally hit a brick wall in November at the American Heart Association scientific sessions.
Treatment of acute heart failure patients with the vasodilating natriuretic peptide ularitide failed to cut long-term cardiovascular mortality or improve several other acute and mid-term outcomes in the TRUE-AHF trial. In a second report, ATHENA-HF, acute treatment with high-dose spironolactone during acute heart failure hospitalizations failed to improve a marker of heart failure severity, N-terminal pro B-type natriuretic peptide.
What alternative interventions are left? Dr. Yancy, as well as Milton Packer, MD, the cardiologist who led TRUE-HF, had somewhat similar answers.
In his discussion of TRUE-AHF, Dr. Yancy cited two possibilities: greater use of the relatively new drug formulation sacubitril plus valsartan (Entresto), which showed strong benefit treating patients with chronic heart failure with reduced ejection fraction (HFrEF); and expanded use of pulmonary-artery pressure (PAP) monitoring using an implanted device that gives clinicians early warning when a heart failure patient’s fluid volume moves into the danger zone that precedes by days or weeks the acute decompensation that produces dyspnea and drives a patient to the hospital.
Increasing experience with PAP monitoring “continues to endorse the notion that having early warning [of fluid overload] is important,” Dr. Yancy said in an interview. “The point of acuity in acute decompensated heart failure predates hospital admission, and this monitoring system allows us to catch this before it causes an emergency department visit. The data are very persuasive.”
“PAP usually rises 2-4 weeks before a heart failure hospitalization. Perhaps we need to focus more on long-term treatment rather than treatment in the hospital.” That’s especially true for patients with heart failure with preserved ejection fraction (HFpEF) because right now no treatment is clearly proven to improve HFpEF outcomes (although several experts make a persuasive case for spironolactone).
“Tight regulation of volume status is our best chance to improve outcomes in HFpEF,” said Dr. Stevenson, who helped pioneer the idea of PAP monitoring for heart failure patients. “Maintaining good volume is very important for HFpEF patients.”
But success with PAP monitoring requires more than gathering the pressure data. Producing benefit for patients “is predicated on having an infrastructure to accommodate the influx of PAP data, being nimble enough to respond to the data and being very precise about which patients you use this in,” cautioned Dr. Yancy. “The tool is not beneficial if the infrastructure is not there,”
The idea is still so new (the first implanted PAP monitor received U.S. approval in 2014) that at his institution, Northwestern Medicine in Chicago, about a dozen patients now have a monitor, he said.
“We’re looking to use it in patients with HFpEF, whom we can’t offer anything else. I’m intrigued by what I see,” in this first wave of Northwestern patients. Dr. Yancy’s anecdotal experience with PAP monitoring so far “helps endorse what the trial results suggested” about providing incremental benefit to heart failure patients.
“The solution is to prevent hospitalization in the first place with the medications we already have, but they’re not used. The medications we already have are enough, but they need to be used,” Dr. Packer told me in an interview during the meeting. He speculated that perhaps 10%-15% of HFrEF patients currently receive the full guideline-directed regimen of heart failure drugs. “That’s unbelievable,” he exclaimed. If clinicians diligently treated advanced HFrEF patients with these four agents, “you’d see a 60%, 70% drop in hospitalizations,” he suggested.
Dr. Packer put some of the blame for underuse of guideline-directed medications on the low financial incentive physicians have to vigorously apply this strategy.
He backed PAP monitoring as a good additional step for selected patients with unstable heart failure. “But as a general approach to managing patients with class III HFrEF, first put them on optimal medical therapy; then we can talk about an invasive procedure to place a PAP monitoring device.”
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Acute decompensated heart failure is a condition that clinicians want to prevent rather than treat.
The idea that patients hospitalized with acute decompensated heart failure can have a substantial change in their prognosis from an acute intervention given in the hospital seemed to finally hit a brick wall in November at the American Heart Association scientific sessions.
Treatment of acute heart failure patients with the vasodilating natriuretic peptide ularitide failed to cut long-term cardiovascular mortality or improve several other acute and mid-term outcomes in the TRUE-AHF trial. In a second report, ATHENA-HF, acute treatment with high-dose spironolactone during acute heart failure hospitalizations failed to improve a marker of heart failure severity, N-terminal pro B-type natriuretic peptide.
What alternative interventions are left? Dr. Yancy, as well as Milton Packer, MD, the cardiologist who led TRUE-HF, had somewhat similar answers.
In his discussion of TRUE-AHF, Dr. Yancy cited two possibilities: greater use of the relatively new drug formulation sacubitril plus valsartan (Entresto), which showed strong benefit treating patients with chronic heart failure with reduced ejection fraction (HFrEF); and expanded use of pulmonary-artery pressure (PAP) monitoring using an implanted device that gives clinicians early warning when a heart failure patient’s fluid volume moves into the danger zone that precedes by days or weeks the acute decompensation that produces dyspnea and drives a patient to the hospital.
Increasing experience with PAP monitoring “continues to endorse the notion that having early warning [of fluid overload] is important,” Dr. Yancy said in an interview. “The point of acuity in acute decompensated heart failure predates hospital admission, and this monitoring system allows us to catch this before it causes an emergency department visit. The data are very persuasive.”
“PAP usually rises 2-4 weeks before a heart failure hospitalization. Perhaps we need to focus more on long-term treatment rather than treatment in the hospital.” That’s especially true for patients with heart failure with preserved ejection fraction (HFpEF) because right now no treatment is clearly proven to improve HFpEF outcomes (although several experts make a persuasive case for spironolactone).
“Tight regulation of volume status is our best chance to improve outcomes in HFpEF,” said Dr. Stevenson, who helped pioneer the idea of PAP monitoring for heart failure patients. “Maintaining good volume is very important for HFpEF patients.”
But success with PAP monitoring requires more than gathering the pressure data. Producing benefit for patients “is predicated on having an infrastructure to accommodate the influx of PAP data, being nimble enough to respond to the data and being very precise about which patients you use this in,” cautioned Dr. Yancy. “The tool is not beneficial if the infrastructure is not there,”
The idea is still so new (the first implanted PAP monitor received U.S. approval in 2014) that at his institution, Northwestern Medicine in Chicago, about a dozen patients now have a monitor, he said.
“We’re looking to use it in patients with HFpEF, whom we can’t offer anything else. I’m intrigued by what I see,” in this first wave of Northwestern patients. Dr. Yancy’s anecdotal experience with PAP monitoring so far “helps endorse what the trial results suggested” about providing incremental benefit to heart failure patients.
“The solution is to prevent hospitalization in the first place with the medications we already have, but they’re not used. The medications we already have are enough, but they need to be used,” Dr. Packer told me in an interview during the meeting. He speculated that perhaps 10%-15% of HFrEF patients currently receive the full guideline-directed regimen of heart failure drugs. “That’s unbelievable,” he exclaimed. If clinicians diligently treated advanced HFrEF patients with these four agents, “you’d see a 60%, 70% drop in hospitalizations,” he suggested.
Dr. Packer put some of the blame for underuse of guideline-directed medications on the low financial incentive physicians have to vigorously apply this strategy.
He backed PAP monitoring as a good additional step for selected patients with unstable heart failure. “But as a general approach to managing patients with class III HFrEF, first put them on optimal medical therapy; then we can talk about an invasive procedure to place a PAP monitoring device.”
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Entresto cuts LV mass in hypertensive patients
NEW ORLEANS – Hypertensive patients without heart failure treated with the heart failure formulation sacubitril plus valsartan had a significantly greater drop in their left ventricular mass than did patients treated with olmesartan in a randomized trial with 114 patients.
Treatment with sacubitril plus valsartan for both 3 and 12 months led to roughly twice as much reduction in left ventricular (LV) mass as did treatment with olmesartan, and this difference persisted after adjustment for between-group differences in blood pressure reduction, Roland E. Schmieder, MD, reported at the American Heart Association Scientific Sessions.
During clinical development of sacubitril plus valsartan (Entresto), the company that owns this compound, Novartis, ran a few small studies using the formulation to treat hypertension, but eventually those studies stopped, he said in an interview. “I hope this pushes development of other neprilysin inhibitor formulations for their blood pressure effects. I think this finding helps us understand why sacubitril plus valsartan was so effective for treating heart failure.” (N Engl J Med. 2014 Sep 11;371[11]:933-1004.)
The study enrolled patients with mild or moderate hypertension; the average blood pressure of enrolled patients was 155/92 mm Hg. They averaged 60 years of age, two-thirds were men, and their average LV mass index at baseline was 72 g/m2. The study excluded patients with heart failure. Patients randomized to receive sacubitril plus valsartan began on a dose of 200 mg/day, and after 2 weeks this rose to 400 mg/day, the maximum recommended dosage in the labeling. Patients randomized to olmesartan began on 20 mg/day, and after 2 weeks their dosage increased to 40 mg/day. Patients in both arms were also eligible to receive amlodipine for additional blood pressure lowering if deemed necessary by the treating physician.
The sacubitril plus valsartan group patients had an average cut in systolic blood pressure of about 26 mm Hg, both 3 and 12 months after the start of treatment. Patients in the olmesartan arm had decreases of 23 mm Hg and 21 mm Hg, respectively, at the two follow-up times. These between-group differences were statistically significant, Dr. Schmieder said.
Measurement of LV mass index using MRI scans showed an average reduction of LV mass index, compared with baseline of 6.4 g/m2 and 6.8 g/m2 after 3 and 12 months of treatment with sacubitril plus valsartan, and average reductions from baseline of 2.3 g/m2 and 3.5 g/m2 at the two follow-up examinations for patients treated with olmesartan. These statistically significant differences remained after adjustment for degree of blood pressure reduction at 3 and 12 months.
Additional measurements showed no between-group differences in aortic distensibility, but central pulse pressure also showed a significantly greater reduction with sacubitril plus valsartan, compared with olmesartan.
The trial was investigator initiated and received funding from Novartis. Dr. Schmieder has received honoraria from Novartis and also from AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and Servier.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
This study produced very interesting and convincing data. The results suggested that treating hypertension with sacubitril plus valsartan produced a significant, incremental improvement in left ventricular mass beyond the formulation’s blood pressure effect. This could potentially have importance when treating patients with hypertension and left ventricular hypertrophy.
Olmesartan was a fair comparator to use. It arguably is the most potent angiotensin receptor blocker for reducing blood pressure. However, in routine practice we generally combine an angiotensin receptor blocker with a diuretic to get maximum blood pressure lowering. In addition, it is not new to show that blood pressure lowering reduces left ventricular size.
These data are too limited and the cost for prescribing sacubitril plus valsartan is so high, compared with most other antihypertensive drugs, that I would like to see additional study results documenting this effect before I’d be willing to prescribe sacubitril plus valsartan to patients with hypertension but no heart failure.
Dan J. Fintel, MD , a cardiologist and professor of medicine at Northwestern University in Chicago, made these comments in an interview. He has been a speaker on behalf of AstraZeneca, BMS, Daiichi Sankyo, Janssen, Merck, and Pfizer.
This study produced very interesting and convincing data. The results suggested that treating hypertension with sacubitril plus valsartan produced a significant, incremental improvement in left ventricular mass beyond the formulation’s blood pressure effect. This could potentially have importance when treating patients with hypertension and left ventricular hypertrophy.
Olmesartan was a fair comparator to use. It arguably is the most potent angiotensin receptor blocker for reducing blood pressure. However, in routine practice we generally combine an angiotensin receptor blocker with a diuretic to get maximum blood pressure lowering. In addition, it is not new to show that blood pressure lowering reduces left ventricular size.
These data are too limited and the cost for prescribing sacubitril plus valsartan is so high, compared with most other antihypertensive drugs, that I would like to see additional study results documenting this effect before I’d be willing to prescribe sacubitril plus valsartan to patients with hypertension but no heart failure.
Dan J. Fintel, MD , a cardiologist and professor of medicine at Northwestern University in Chicago, made these comments in an interview. He has been a speaker on behalf of AstraZeneca, BMS, Daiichi Sankyo, Janssen, Merck, and Pfizer.
This study produced very interesting and convincing data. The results suggested that treating hypertension with sacubitril plus valsartan produced a significant, incremental improvement in left ventricular mass beyond the formulation’s blood pressure effect. This could potentially have importance when treating patients with hypertension and left ventricular hypertrophy.
Olmesartan was a fair comparator to use. It arguably is the most potent angiotensin receptor blocker for reducing blood pressure. However, in routine practice we generally combine an angiotensin receptor blocker with a diuretic to get maximum blood pressure lowering. In addition, it is not new to show that blood pressure lowering reduces left ventricular size.
These data are too limited and the cost for prescribing sacubitril plus valsartan is so high, compared with most other antihypertensive drugs, that I would like to see additional study results documenting this effect before I’d be willing to prescribe sacubitril plus valsartan to patients with hypertension but no heart failure.
Dan J. Fintel, MD , a cardiologist and professor of medicine at Northwestern University in Chicago, made these comments in an interview. He has been a speaker on behalf of AstraZeneca, BMS, Daiichi Sankyo, Janssen, Merck, and Pfizer.
NEW ORLEANS – Hypertensive patients without heart failure treated with the heart failure formulation sacubitril plus valsartan had a significantly greater drop in their left ventricular mass than did patients treated with olmesartan in a randomized trial with 114 patients.
Treatment with sacubitril plus valsartan for both 3 and 12 months led to roughly twice as much reduction in left ventricular (LV) mass as did treatment with olmesartan, and this difference persisted after adjustment for between-group differences in blood pressure reduction, Roland E. Schmieder, MD, reported at the American Heart Association Scientific Sessions.
During clinical development of sacubitril plus valsartan (Entresto), the company that owns this compound, Novartis, ran a few small studies using the formulation to treat hypertension, but eventually those studies stopped, he said in an interview. “I hope this pushes development of other neprilysin inhibitor formulations for their blood pressure effects. I think this finding helps us understand why sacubitril plus valsartan was so effective for treating heart failure.” (N Engl J Med. 2014 Sep 11;371[11]:933-1004.)
The study enrolled patients with mild or moderate hypertension; the average blood pressure of enrolled patients was 155/92 mm Hg. They averaged 60 years of age, two-thirds were men, and their average LV mass index at baseline was 72 g/m2. The study excluded patients with heart failure. Patients randomized to receive sacubitril plus valsartan began on a dose of 200 mg/day, and after 2 weeks this rose to 400 mg/day, the maximum recommended dosage in the labeling. Patients randomized to olmesartan began on 20 mg/day, and after 2 weeks their dosage increased to 40 mg/day. Patients in both arms were also eligible to receive amlodipine for additional blood pressure lowering if deemed necessary by the treating physician.
The sacubitril plus valsartan group patients had an average cut in systolic blood pressure of about 26 mm Hg, both 3 and 12 months after the start of treatment. Patients in the olmesartan arm had decreases of 23 mm Hg and 21 mm Hg, respectively, at the two follow-up times. These between-group differences were statistically significant, Dr. Schmieder said.
Measurement of LV mass index using MRI scans showed an average reduction of LV mass index, compared with baseline of 6.4 g/m2 and 6.8 g/m2 after 3 and 12 months of treatment with sacubitril plus valsartan, and average reductions from baseline of 2.3 g/m2 and 3.5 g/m2 at the two follow-up examinations for patients treated with olmesartan. These statistically significant differences remained after adjustment for degree of blood pressure reduction at 3 and 12 months.
Additional measurements showed no between-group differences in aortic distensibility, but central pulse pressure also showed a significantly greater reduction with sacubitril plus valsartan, compared with olmesartan.
The trial was investigator initiated and received funding from Novartis. Dr. Schmieder has received honoraria from Novartis and also from AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and Servier.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
NEW ORLEANS – Hypertensive patients without heart failure treated with the heart failure formulation sacubitril plus valsartan had a significantly greater drop in their left ventricular mass than did patients treated with olmesartan in a randomized trial with 114 patients.
Treatment with sacubitril plus valsartan for both 3 and 12 months led to roughly twice as much reduction in left ventricular (LV) mass as did treatment with olmesartan, and this difference persisted after adjustment for between-group differences in blood pressure reduction, Roland E. Schmieder, MD, reported at the American Heart Association Scientific Sessions.
During clinical development of sacubitril plus valsartan (Entresto), the company that owns this compound, Novartis, ran a few small studies using the formulation to treat hypertension, but eventually those studies stopped, he said in an interview. “I hope this pushes development of other neprilysin inhibitor formulations for their blood pressure effects. I think this finding helps us understand why sacubitril plus valsartan was so effective for treating heart failure.” (N Engl J Med. 2014 Sep 11;371[11]:933-1004.)
The study enrolled patients with mild or moderate hypertension; the average blood pressure of enrolled patients was 155/92 mm Hg. They averaged 60 years of age, two-thirds were men, and their average LV mass index at baseline was 72 g/m2. The study excluded patients with heart failure. Patients randomized to receive sacubitril plus valsartan began on a dose of 200 mg/day, and after 2 weeks this rose to 400 mg/day, the maximum recommended dosage in the labeling. Patients randomized to olmesartan began on 20 mg/day, and after 2 weeks their dosage increased to 40 mg/day. Patients in both arms were also eligible to receive amlodipine for additional blood pressure lowering if deemed necessary by the treating physician.
The sacubitril plus valsartan group patients had an average cut in systolic blood pressure of about 26 mm Hg, both 3 and 12 months after the start of treatment. Patients in the olmesartan arm had decreases of 23 mm Hg and 21 mm Hg, respectively, at the two follow-up times. These between-group differences were statistically significant, Dr. Schmieder said.
Measurement of LV mass index using MRI scans showed an average reduction of LV mass index, compared with baseline of 6.4 g/m2 and 6.8 g/m2 after 3 and 12 months of treatment with sacubitril plus valsartan, and average reductions from baseline of 2.3 g/m2 and 3.5 g/m2 at the two follow-up examinations for patients treated with olmesartan. These statistically significant differences remained after adjustment for degree of blood pressure reduction at 3 and 12 months.
Additional measurements showed no between-group differences in aortic distensibility, but central pulse pressure also showed a significantly greater reduction with sacubitril plus valsartan, compared with olmesartan.
The trial was investigator initiated and received funding from Novartis. Dr. Schmieder has received honoraria from Novartis and also from AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and Servier.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE AHA SCIENTIFIC SESSIONS
Key clinical point: .
Major finding: After 1 year, average left ventricular mass index fell 6.8 g/m2 from baseline with sacubitril/valsartan and 3.5 g/m2 with olmesartan.
Data source: A multicenter, randomized trial with 114 patients with mild or moderate hypertension.
Disclosures: The trial was investigator initiated and received funding from Novartis, the company that markets sacubitril plus valsartan (Entresto). Dr. Schmieder has received honoraria from Novartis and also from AstraZeneca, Boehringer Ingelheim, Daiichi Sankyo, and Servier.























