User login
Herpes Zoster May Be a Marker for COVID-19 Infection During Pregnancy
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the most recently identified member of the zoonotic pathogens of coronaviruses. It caused an outbreak of pneumonia in December 2019 in Wuhan, China.1 Among all related acute respiratory syndromes (SARS-CoV, Middle East respiratory syndrome coronavirus), SARS-CoV-2 remains to be the most infectious, has the highest potential for human transmission, and can eventually result in acute respiratory distress syndrome.2,3
Only 15% of coronavirus disease 2019 (COVID-19) cases progress to pneumonia, and approximately 5% of these cases develop acute respiratory distress syndrome, septic shock, and/or multiple organ failure. The majority of cases only exhibit mild to moderate symptoms.4,5 A wide array of skin manifestations in COVID-19 infection have been reported, including maculopapular eruptions, morbilliform rashes, urticaria, chickenpoxlike lesions, livedo reticularis, COVID toes, erythema multiforme, pityriasis rosea, and several other patterns.6 We report a case of herpes zoster (HZ) complication in a COVID-19–positive woman who was 27 weeks pregnant.
Case Report
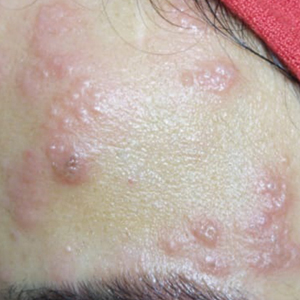
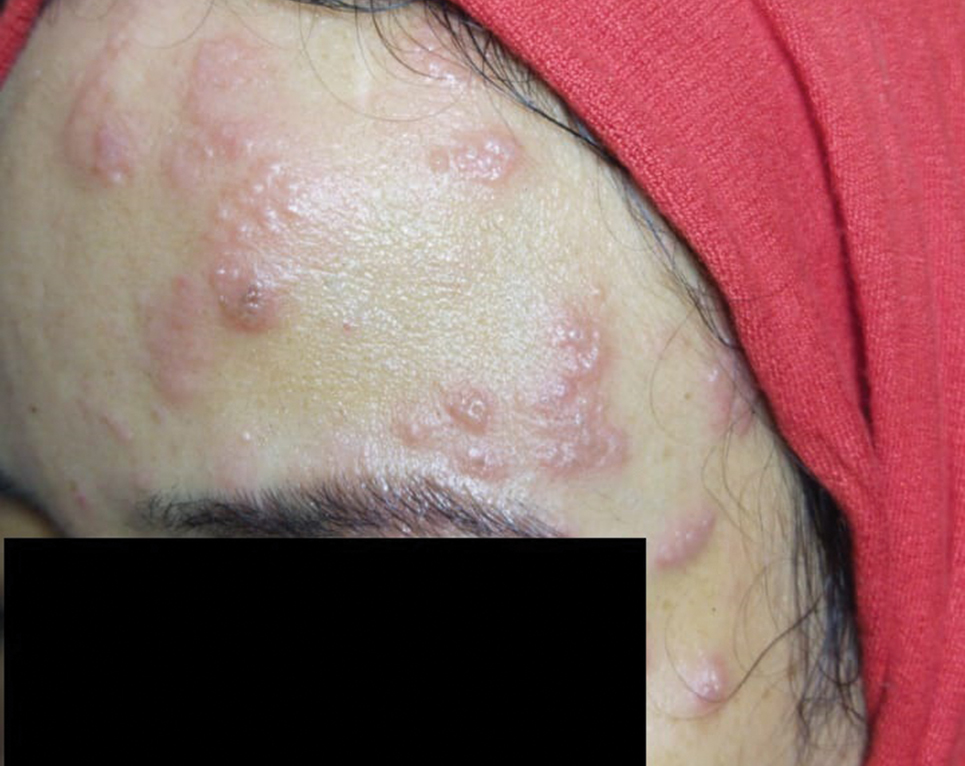
A 36-year-old woman who was 27 weeks pregnant was referred by her obstetrician to the dermatology clinic. She presented with a low-grade fever and a vesicular painful rash. Physical examination revealed painful, itchy, dysesthetic papules and vesicles on the left side of the forehead along with mild edema of the left upper eyelid but no watering of the eye or photophobia. She reported episodes of fever (temperature, 38.9°C), fatigue, and myalgia over the last week. She had bouts of dyspnea and tachycardia that she thought were related to being in the late second trimester of pregnancy. The area surrounding the vesicular eruption was tender to touch. No dry cough or any gastrointestinal or urinary tract symptoms were noted. She reported a burning sensation when splashing water on the face or when exposed to air currents. One week following the initial symptoms, she experienced a painful vesicular rash along the upper left forehead (Figure) associated with eyelid edema. Oral and ocular mucosae were free of any presentations. She had no relevant history and had not experienced any complications during pregnancy. A diagnosis of HZ was made, and she was prescribed valacyclovir 1 g 3 times daily for 7 days, acetaminophen for the fever, and calamine lotion. We recommended COVID-19 testing based on her symptoms. A chest radiograph and a positive nasopharyngeal smear were consistent with COVID-19 infection. She reported via telephone follow-up 1 week after presentation that her skin condition had improved following the treatment course and that the vesicles eventually dried, leaving a crusting appearance after 5 to 7 days. Regarding her SARS-CoV-2 condition, her oxygen saturation was 95% at presentation; she self-quarantined at home; and she was treated with oseltamivir 75 mg orally every 12 hours for 5 days, azithromycin 500 mg orally daily, acetaminophen, and vitamin C. Electronic fetal heart rate monitoring and ultrasound examinations were performed to assess the condition of the fetus and were reported normal. At the time of writing this article, she was 32 weeks pregnant and tested negative to 2 consecutive nasopharyngeal swabs for COVID-19 and was in good general condition. She continued her pregnancy according to her obstetrician’s recommendations.
Comment
The incubation time of COVID-19 can be up to 14 days. Fever, dry cough, fatigue, and diarrhea have been speculated to be clinical symptoms; however, many cases may be asymptomatic. Aside from a medical or travel history at risk for COVID-19, diagnosis can be confirmed by detection of viral RNA by reverse transcriptase–polymerase chain reaction for nasopharyngeal swabs or bronchoalveolar fluid. Patients who are immunocompromised, older, or male or who have a history of cardiovascular conditions or debilitating chronic conditions are at an increased risk for severe disease and poor outcome compared to younger healthy individuals.7
The vesicular rash of COVID-19 has been reported to have different forms of presentation. A diffuse widespread pattern resembling hand-foot-and-mouth disease and a localized monomorphic pattern resembling chickenpox but with predilection to the trunk has been described.8
Physiologic changes in the immune and cardiopulmonary systems during pregnancy (eg, diaphragm elevation, increased oxygen consumption, edema of the respiratory tract mucosae) make pregnant women intolerant to hypoxia. The mortality rate of the 1918 influenza pandemic was 2.6% in the overall population but 37% among pregnant women.9 In 2009, pregnant women were reported to be at an increased risk for complications from the H1N1 influenza virus pandemic, with a higher estimated rate of hospital admission than the general population.10 In 2003, approximately 50% of pregnant women who received a diagnosis of SARS-CoV were admitted to the intensive care unit, approximately 33% of pregnant women with SARS-CoV required mechanical ventilation, and the mortality rate was as high as 25% for these women.11 To date, data on the effects of COVID-19 in pregnancy are limited to small case series.12-15
It was confirmed that COVID-19 infection is accompanied by a reduction in lymphocytes, monocytes, and eosinophils, along with a notable reduction of CD4/CD8 T cells, B cells, and natural killer cells. It was further revealed that nonsurvivor COVID-19 patients continued to show a decrease in lymphocyte counts along the course of their disease until death.16-18
Different mechanisms for lymphocyte depletion and deficiency were speculated among COVID-19 patients and include direct lymphocyte death through coronavirus angiotensin-converting enzyme 2–lymphocyte-expressed receptors; direct damage to lymphatic organs, such as the thymus and spleen, but this theory needs to be further investigated; direct lymphocyte apoptosis mediated by tumor necrosis factor α, IL-6, and other proinflammatory cytokines; and direct inhibition of lymphocytes by metabolic upset, such as acidosis.19,20
These causes may precipitate lymphopenia and impaired antiviral responses.21 It also has been postulated that the functional damage of CD4+ T cells may predispose patients with COVID-19 to severe disease.22 Such immune changes can render a patient more susceptible to developing shingles by reactivating varicella-zoster virus, which could be a sign of undiagnosed COVID-19 infection in younger age groups.
Two earlier reports discussed HZ among COVID-19–diagnosed patients. Shors23 presented a case of a patient who developed varicella-zoster virus reactivation of the V2 dermatome during the course of COVID-19 infection. In addition, the patient developed severe acute herpetic neuralgia despite the early initiation of antiviral therapy.23 Elsaie et al24 described 2 cases of patients during the pandemic who first presented with HZ before later being diagnosed with COVID-19 infection.
New information and cutaneous manifestations possibly related to COVID-19 are emerging every day. We report a pregnant female presenting with HZ during the course of COVID-19 infection, which suggests that the clinical presentation of HZ at the time of the current pandemic, especially if associated with other signs of COVID-19 infection, should be carefully monitored and reported for further assessment.
Acknowledgment
The authors would like to thank all the health care workers who have been fighting COVID-19 in Egypt and worldwide.
- Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020;382:1199-1207.
- Zhang YZ, Holes EC. A genomic perspective on the origin and emergence of sars-cov-2. Cell. 2020;181:223-227.
- Prompetchara E, Ketloy C, Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. 2020;38:1‐9.
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan0, China. Lancet. 2020;395:497-506.
- Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422.
- Wollina U, Karadag˘ AS, Rowland-Payne C, et al. Cutaneous signs in COVID-19 patients: a review. Dermatol Ther. 2020;33:e13549.
- Lauer SA, Grantz KH, Bi Q, et al. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann Intern Med. 2020;172:577‐582.
- Fernandez-Nieto D, Ortega-Quijano D, Jimenez-Cauhe J, et al. Clinical and histological characterization of vesicular COVID-19 rashes: a prospective study in a tertiary care hospital. Clin Exp Dermatol. 2020;45:872-875.
- Gottfredsson M. The Spanish flu in Iceland 1918. Lessons in medicine and history [in Icelandic]. Laeknabladid. 2008;94:737-745.
- Jamieson D, Honein M, Rasmussen S, et al. H1N1 2009 influenza virus infection during pregnancy in the USA. Lancet. 2009;374:451-458.
- Ksiazek TG, Erdman D, Goldsmith CS. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953-1966.
- Chen H, Guo J, Wang C, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395:809‐815.
- Zhu H, Wang L, Fang C, et al. Clinical analysis of 10 neonates born to mothers with 2019-nCov pneumonia. Transl Pediatr. 2020;9:51-60.
- Liu Y, Chen H, Tang K, et al. Clinical manifestations and outcome of SARS-CoV-2 infection during pregnancy [published online March 4, 2020]. J Infect. doi:10.1016/j.jinf.2020.02.028.
- Zhang L, Jiang Y, Wei M, et al. Analysis of the pregnancy outcomes in pregnant women with COVID-19 in Hubei Province [in Chinese]. Zhonghua Fu Chan Ke Za Zhi. 2020;55:166-171.
- Henry BM, de Oliveira MHS, Benoit S, et al. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med. 2020;58:1021-1028.
- Cai Q, Huang D, Ou P, et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy. 2020;75:1742-1752.
- Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846-884.
- Kumar A, Anil A, Sharma P, et al. Clinical features of COVID-19 and factors associated with severe clinical course: a systematic review and meta-analysis [preprint]. SSRN. doi:10.2139/ssrn.3566166.
- Xu H, Zhong L, Deng J, et al. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. 2020;12. https://doi.org/10.1038/s41368-020-0074-x.
- Li H, Liu L, Zhang D, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395:1517-1520.
- Zheng M, Gao Y, Wang G, et al. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol Immunol. 2020;17:533-535.
- Shors AR. Herpes zoster and severe acute herpetic neuralgia as a complication of COVID-19 infection. JAAD Case Rep. 2020;6:656-657.
- Elsaie ML, Youssef EA, Nada HA. Herpes zoster might be an indicator for latent COVID 19 infection [published online May 23, 2020]. Dermatol Ther. doi:10.1111/dth.13666.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the most recently identified member of the zoonotic pathogens of coronaviruses. It caused an outbreak of pneumonia in December 2019 in Wuhan, China.1 Among all related acute respiratory syndromes (SARS-CoV, Middle East respiratory syndrome coronavirus), SARS-CoV-2 remains to be the most infectious, has the highest potential for human transmission, and can eventually result in acute respiratory distress syndrome.2,3
Only 15% of coronavirus disease 2019 (COVID-19) cases progress to pneumonia, and approximately 5% of these cases develop acute respiratory distress syndrome, septic shock, and/or multiple organ failure. The majority of cases only exhibit mild to moderate symptoms.4,5 A wide array of skin manifestations in COVID-19 infection have been reported, including maculopapular eruptions, morbilliform rashes, urticaria, chickenpoxlike lesions, livedo reticularis, COVID toes, erythema multiforme, pityriasis rosea, and several other patterns.6 We report a case of herpes zoster (HZ) complication in a COVID-19–positive woman who was 27 weeks pregnant.
Case Report
A 36-year-old woman who was 27 weeks pregnant was referred by her obstetrician to the dermatology clinic. She presented with a low-grade fever and a vesicular painful rash. Physical examination revealed painful, itchy, dysesthetic papules and vesicles on the left side of the forehead along with mild edema of the left upper eyelid but no watering of the eye or photophobia. She reported episodes of fever (temperature, 38.9°C), fatigue, and myalgia over the last week. She had bouts of dyspnea and tachycardia that she thought were related to being in the late second trimester of pregnancy. The area surrounding the vesicular eruption was tender to touch. No dry cough or any gastrointestinal or urinary tract symptoms were noted. She reported a burning sensation when splashing water on the face or when exposed to air currents. One week following the initial symptoms, she experienced a painful vesicular rash along the upper left forehead (Figure) associated with eyelid edema. Oral and ocular mucosae were free of any presentations. She had no relevant history and had not experienced any complications during pregnancy. A diagnosis of HZ was made, and she was prescribed valacyclovir 1 g 3 times daily for 7 days, acetaminophen for the fever, and calamine lotion. We recommended COVID-19 testing based on her symptoms. A chest radiograph and a positive nasopharyngeal smear were consistent with COVID-19 infection. She reported via telephone follow-up 1 week after presentation that her skin condition had improved following the treatment course and that the vesicles eventually dried, leaving a crusting appearance after 5 to 7 days. Regarding her SARS-CoV-2 condition, her oxygen saturation was 95% at presentation; she self-quarantined at home; and she was treated with oseltamivir 75 mg orally every 12 hours for 5 days, azithromycin 500 mg orally daily, acetaminophen, and vitamin C. Electronic fetal heart rate monitoring and ultrasound examinations were performed to assess the condition of the fetus and were reported normal. At the time of writing this article, she was 32 weeks pregnant and tested negative to 2 consecutive nasopharyngeal swabs for COVID-19 and was in good general condition. She continued her pregnancy according to her obstetrician’s recommendations.

Comment
The incubation time of COVID-19 can be up to 14 days. Fever, dry cough, fatigue, and diarrhea have been speculated to be clinical symptoms; however, many cases may be asymptomatic. Aside from a medical or travel history at risk for COVID-19, diagnosis can be confirmed by detection of viral RNA by reverse transcriptase–polymerase chain reaction for nasopharyngeal swabs or bronchoalveolar fluid. Patients who are immunocompromised, older, or male or who have a history of cardiovascular conditions or debilitating chronic conditions are at an increased risk for severe disease and poor outcome compared to younger healthy individuals.7
The vesicular rash of COVID-19 has been reported to have different forms of presentation. A diffuse widespread pattern resembling hand-foot-and-mouth disease and a localized monomorphic pattern resembling chickenpox but with predilection to the trunk has been described.8
Physiologic changes in the immune and cardiopulmonary systems during pregnancy (eg, diaphragm elevation, increased oxygen consumption, edema of the respiratory tract mucosae) make pregnant women intolerant to hypoxia. The mortality rate of the 1918 influenza pandemic was 2.6% in the overall population but 37% among pregnant women.9 In 2009, pregnant women were reported to be at an increased risk for complications from the H1N1 influenza virus pandemic, with a higher estimated rate of hospital admission than the general population.10 In 2003, approximately 50% of pregnant women who received a diagnosis of SARS-CoV were admitted to the intensive care unit, approximately 33% of pregnant women with SARS-CoV required mechanical ventilation, and the mortality rate was as high as 25% for these women.11 To date, data on the effects of COVID-19 in pregnancy are limited to small case series.12-15
It was confirmed that COVID-19 infection is accompanied by a reduction in lymphocytes, monocytes, and eosinophils, along with a notable reduction of CD4/CD8 T cells, B cells, and natural killer cells. It was further revealed that nonsurvivor COVID-19 patients continued to show a decrease in lymphocyte counts along the course of their disease until death.16-18
Different mechanisms for lymphocyte depletion and deficiency were speculated among COVID-19 patients and include direct lymphocyte death through coronavirus angiotensin-converting enzyme 2–lymphocyte-expressed receptors; direct damage to lymphatic organs, such as the thymus and spleen, but this theory needs to be further investigated; direct lymphocyte apoptosis mediated by tumor necrosis factor α, IL-6, and other proinflammatory cytokines; and direct inhibition of lymphocytes by metabolic upset, such as acidosis.19,20
These causes may precipitate lymphopenia and impaired antiviral responses.21 It also has been postulated that the functional damage of CD4+ T cells may predispose patients with COVID-19 to severe disease.22 Such immune changes can render a patient more susceptible to developing shingles by reactivating varicella-zoster virus, which could be a sign of undiagnosed COVID-19 infection in younger age groups.
Two earlier reports discussed HZ among COVID-19–diagnosed patients. Shors23 presented a case of a patient who developed varicella-zoster virus reactivation of the V2 dermatome during the course of COVID-19 infection. In addition, the patient developed severe acute herpetic neuralgia despite the early initiation of antiviral therapy.23 Elsaie et al24 described 2 cases of patients during the pandemic who first presented with HZ before later being diagnosed with COVID-19 infection.
New information and cutaneous manifestations possibly related to COVID-19 are emerging every day. We report a pregnant female presenting with HZ during the course of COVID-19 infection, which suggests that the clinical presentation of HZ at the time of the current pandemic, especially if associated with other signs of COVID-19 infection, should be carefully monitored and reported for further assessment.
Acknowledgment
The authors would like to thank all the health care workers who have been fighting COVID-19 in Egypt and worldwide.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the most recently identified member of the zoonotic pathogens of coronaviruses. It caused an outbreak of pneumonia in December 2019 in Wuhan, China.1 Among all related acute respiratory syndromes (SARS-CoV, Middle East respiratory syndrome coronavirus), SARS-CoV-2 remains to be the most infectious, has the highest potential for human transmission, and can eventually result in acute respiratory distress syndrome.2,3
Only 15% of coronavirus disease 2019 (COVID-19) cases progress to pneumonia, and approximately 5% of these cases develop acute respiratory distress syndrome, septic shock, and/or multiple organ failure. The majority of cases only exhibit mild to moderate symptoms.4,5 A wide array of skin manifestations in COVID-19 infection have been reported, including maculopapular eruptions, morbilliform rashes, urticaria, chickenpoxlike lesions, livedo reticularis, COVID toes, erythema multiforme, pityriasis rosea, and several other patterns.6 We report a case of herpes zoster (HZ) complication in a COVID-19–positive woman who was 27 weeks pregnant.
Case Report
A 36-year-old woman who was 27 weeks pregnant was referred by her obstetrician to the dermatology clinic. She presented with a low-grade fever and a vesicular painful rash. Physical examination revealed painful, itchy, dysesthetic papules and vesicles on the left side of the forehead along with mild edema of the left upper eyelid but no watering of the eye or photophobia. She reported episodes of fever (temperature, 38.9°C), fatigue, and myalgia over the last week. She had bouts of dyspnea and tachycardia that she thought were related to being in the late second trimester of pregnancy. The area surrounding the vesicular eruption was tender to touch. No dry cough or any gastrointestinal or urinary tract symptoms were noted. She reported a burning sensation when splashing water on the face or when exposed to air currents. One week following the initial symptoms, she experienced a painful vesicular rash along the upper left forehead (Figure) associated with eyelid edema. Oral and ocular mucosae were free of any presentations. She had no relevant history and had not experienced any complications during pregnancy. A diagnosis of HZ was made, and she was prescribed valacyclovir 1 g 3 times daily for 7 days, acetaminophen for the fever, and calamine lotion. We recommended COVID-19 testing based on her symptoms. A chest radiograph and a positive nasopharyngeal smear were consistent with COVID-19 infection. She reported via telephone follow-up 1 week after presentation that her skin condition had improved following the treatment course and that the vesicles eventually dried, leaving a crusting appearance after 5 to 7 days. Regarding her SARS-CoV-2 condition, her oxygen saturation was 95% at presentation; she self-quarantined at home; and she was treated with oseltamivir 75 mg orally every 12 hours for 5 days, azithromycin 500 mg orally daily, acetaminophen, and vitamin C. Electronic fetal heart rate monitoring and ultrasound examinations were performed to assess the condition of the fetus and were reported normal. At the time of writing this article, she was 32 weeks pregnant and tested negative to 2 consecutive nasopharyngeal swabs for COVID-19 and was in good general condition. She continued her pregnancy according to her obstetrician’s recommendations.

Comment
The incubation time of COVID-19 can be up to 14 days. Fever, dry cough, fatigue, and diarrhea have been speculated to be clinical symptoms; however, many cases may be asymptomatic. Aside from a medical or travel history at risk for COVID-19, diagnosis can be confirmed by detection of viral RNA by reverse transcriptase–polymerase chain reaction for nasopharyngeal swabs or bronchoalveolar fluid. Patients who are immunocompromised, older, or male or who have a history of cardiovascular conditions or debilitating chronic conditions are at an increased risk for severe disease and poor outcome compared to younger healthy individuals.7
The vesicular rash of COVID-19 has been reported to have different forms of presentation. A diffuse widespread pattern resembling hand-foot-and-mouth disease and a localized monomorphic pattern resembling chickenpox but with predilection to the trunk has been described.8
Physiologic changes in the immune and cardiopulmonary systems during pregnancy (eg, diaphragm elevation, increased oxygen consumption, edema of the respiratory tract mucosae) make pregnant women intolerant to hypoxia. The mortality rate of the 1918 influenza pandemic was 2.6% in the overall population but 37% among pregnant women.9 In 2009, pregnant women were reported to be at an increased risk for complications from the H1N1 influenza virus pandemic, with a higher estimated rate of hospital admission than the general population.10 In 2003, approximately 50% of pregnant women who received a diagnosis of SARS-CoV were admitted to the intensive care unit, approximately 33% of pregnant women with SARS-CoV required mechanical ventilation, and the mortality rate was as high as 25% for these women.11 To date, data on the effects of COVID-19 in pregnancy are limited to small case series.12-15
It was confirmed that COVID-19 infection is accompanied by a reduction in lymphocytes, monocytes, and eosinophils, along with a notable reduction of CD4/CD8 T cells, B cells, and natural killer cells. It was further revealed that nonsurvivor COVID-19 patients continued to show a decrease in lymphocyte counts along the course of their disease until death.16-18
Different mechanisms for lymphocyte depletion and deficiency were speculated among COVID-19 patients and include direct lymphocyte death through coronavirus angiotensin-converting enzyme 2–lymphocyte-expressed receptors; direct damage to lymphatic organs, such as the thymus and spleen, but this theory needs to be further investigated; direct lymphocyte apoptosis mediated by tumor necrosis factor α, IL-6, and other proinflammatory cytokines; and direct inhibition of lymphocytes by metabolic upset, such as acidosis.19,20
These causes may precipitate lymphopenia and impaired antiviral responses.21 It also has been postulated that the functional damage of CD4+ T cells may predispose patients with COVID-19 to severe disease.22 Such immune changes can render a patient more susceptible to developing shingles by reactivating varicella-zoster virus, which could be a sign of undiagnosed COVID-19 infection in younger age groups.
Two earlier reports discussed HZ among COVID-19–diagnosed patients. Shors23 presented a case of a patient who developed varicella-zoster virus reactivation of the V2 dermatome during the course of COVID-19 infection. In addition, the patient developed severe acute herpetic neuralgia despite the early initiation of antiviral therapy.23 Elsaie et al24 described 2 cases of patients during the pandemic who first presented with HZ before later being diagnosed with COVID-19 infection.
New information and cutaneous manifestations possibly related to COVID-19 are emerging every day. We report a pregnant female presenting with HZ during the course of COVID-19 infection, which suggests that the clinical presentation of HZ at the time of the current pandemic, especially if associated with other signs of COVID-19 infection, should be carefully monitored and reported for further assessment.
Acknowledgment
The authors would like to thank all the health care workers who have been fighting COVID-19 in Egypt and worldwide.
- Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020;382:1199-1207.
- Zhang YZ, Holes EC. A genomic perspective on the origin and emergence of sars-cov-2. Cell. 2020;181:223-227.
- Prompetchara E, Ketloy C, Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. 2020;38:1‐9.
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan0, China. Lancet. 2020;395:497-506.
- Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422.
- Wollina U, Karadag˘ AS, Rowland-Payne C, et al. Cutaneous signs in COVID-19 patients: a review. Dermatol Ther. 2020;33:e13549.
- Lauer SA, Grantz KH, Bi Q, et al. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann Intern Med. 2020;172:577‐582.
- Fernandez-Nieto D, Ortega-Quijano D, Jimenez-Cauhe J, et al. Clinical and histological characterization of vesicular COVID-19 rashes: a prospective study in a tertiary care hospital. Clin Exp Dermatol. 2020;45:872-875.
- Gottfredsson M. The Spanish flu in Iceland 1918. Lessons in medicine and history [in Icelandic]. Laeknabladid. 2008;94:737-745.
- Jamieson D, Honein M, Rasmussen S, et al. H1N1 2009 influenza virus infection during pregnancy in the USA. Lancet. 2009;374:451-458.
- Ksiazek TG, Erdman D, Goldsmith CS. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953-1966.
- Chen H, Guo J, Wang C, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395:809‐815.
- Zhu H, Wang L, Fang C, et al. Clinical analysis of 10 neonates born to mothers with 2019-nCov pneumonia. Transl Pediatr. 2020;9:51-60.
- Liu Y, Chen H, Tang K, et al. Clinical manifestations and outcome of SARS-CoV-2 infection during pregnancy [published online March 4, 2020]. J Infect. doi:10.1016/j.jinf.2020.02.028.
- Zhang L, Jiang Y, Wei M, et al. Analysis of the pregnancy outcomes in pregnant women with COVID-19 in Hubei Province [in Chinese]. Zhonghua Fu Chan Ke Za Zhi. 2020;55:166-171.
- Henry BM, de Oliveira MHS, Benoit S, et al. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med. 2020;58:1021-1028.
- Cai Q, Huang D, Ou P, et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy. 2020;75:1742-1752.
- Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846-884.
- Kumar A, Anil A, Sharma P, et al. Clinical features of COVID-19 and factors associated with severe clinical course: a systematic review and meta-analysis [preprint]. SSRN. doi:10.2139/ssrn.3566166.
- Xu H, Zhong L, Deng J, et al. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. 2020;12. https://doi.org/10.1038/s41368-020-0074-x.
- Li H, Liu L, Zhang D, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395:1517-1520.
- Zheng M, Gao Y, Wang G, et al. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol Immunol. 2020;17:533-535.
- Shors AR. Herpes zoster and severe acute herpetic neuralgia as a complication of COVID-19 infection. JAAD Case Rep. 2020;6:656-657.
- Elsaie ML, Youssef EA, Nada HA. Herpes zoster might be an indicator for latent COVID 19 infection [published online May 23, 2020]. Dermatol Ther. doi:10.1111/dth.13666.
- Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med. 2020;382:1199-1207.
- Zhang YZ, Holes EC. A genomic perspective on the origin and emergence of sars-cov-2. Cell. 2020;181:223-227.
- Prompetchara E, Ketloy C, Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. 2020;38:1‐9.
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan0, China. Lancet. 2020;395:497-506.
- Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8:420-422.
- Wollina U, Karadag˘ AS, Rowland-Payne C, et al. Cutaneous signs in COVID-19 patients: a review. Dermatol Ther. 2020;33:e13549.
- Lauer SA, Grantz KH, Bi Q, et al. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: estimation and application. Ann Intern Med. 2020;172:577‐582.
- Fernandez-Nieto D, Ortega-Quijano D, Jimenez-Cauhe J, et al. Clinical and histological characterization of vesicular COVID-19 rashes: a prospective study in a tertiary care hospital. Clin Exp Dermatol. 2020;45:872-875.
- Gottfredsson M. The Spanish flu in Iceland 1918. Lessons in medicine and history [in Icelandic]. Laeknabladid. 2008;94:737-745.
- Jamieson D, Honein M, Rasmussen S, et al. H1N1 2009 influenza virus infection during pregnancy in the USA. Lancet. 2009;374:451-458.
- Ksiazek TG, Erdman D, Goldsmith CS. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348:1953-1966.
- Chen H, Guo J, Wang C, et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395:809‐815.
- Zhu H, Wang L, Fang C, et al. Clinical analysis of 10 neonates born to mothers with 2019-nCov pneumonia. Transl Pediatr. 2020;9:51-60.
- Liu Y, Chen H, Tang K, et al. Clinical manifestations and outcome of SARS-CoV-2 infection during pregnancy [published online March 4, 2020]. J Infect. doi:10.1016/j.jinf.2020.02.028.
- Zhang L, Jiang Y, Wei M, et al. Analysis of the pregnancy outcomes in pregnant women with COVID-19 in Hubei Province [in Chinese]. Zhonghua Fu Chan Ke Za Zhi. 2020;55:166-171.
- Henry BM, de Oliveira MHS, Benoit S, et al. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med. 2020;58:1021-1028.
- Cai Q, Huang D, Ou P, et al. COVID-19 in a designated infectious diseases hospital outside Hubei Province, China. Allergy. 2020;75:1742-1752.
- Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846-884.
- Kumar A, Anil A, Sharma P, et al. Clinical features of COVID-19 and factors associated with severe clinical course: a systematic review and meta-analysis [preprint]. SSRN. doi:10.2139/ssrn.3566166.
- Xu H, Zhong L, Deng J, et al. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. 2020;12. https://doi.org/10.1038/s41368-020-0074-x.
- Li H, Liu L, Zhang D, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395:1517-1520.
- Zheng M, Gao Y, Wang G, et al. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol Immunol. 2020;17:533-535.
- Shors AR. Herpes zoster and severe acute herpetic neuralgia as a complication of COVID-19 infection. JAAD Case Rep. 2020;6:656-657.
- Elsaie ML, Youssef EA, Nada HA. Herpes zoster might be an indicator for latent COVID 19 infection [published online May 23, 2020]. Dermatol Ther. doi:10.1111/dth.13666.
Practice Points
- The vesicular rash of coronavirus disease 2019 (COVID-19) has been reported to have different forms of presentation.
- Pregnant women appear to be at increased risk for complications from COVID-19 infection.
- The clinical presentation of herpes zoster should be carefully monitored and reported for further assessment, especially if associated with other signs of COVID-19 infection.
Dermatology Battles COVID-19 With Comfort
We are in unprecedented times. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is attacking our communities and, as with any battle, we face unexpected challenges from the global pandemic. What can dermatologists, as highly skilled health care experts, do to support the fight against coronavirus disease 2019 (COVID-19)?
In early 2020, I became involved in a fulfilling and stimulating opportunity to contribute as a US Navy reservist, having just returned from a 3-month deployment. I served in the Medical Operations Center aboard the hospital ship USNS Comfort, which was docked in New York Harbor, as liaison to surrounding New York City hospitals. I also served as sole dermatologist on the ship, caring for the dermatologic needs of our team and consulting on numerous COVID-19 inpatients.
In May 2020, upon return to Virginia from New York City, I served as senior medical officer to medically clear other Navy Reserve health care workers returning from the field hospital at the Jacob K. Javits Convention Center of New York and from serving as embedded caregivers in existing New York City hospitals. I share 2 very important observations from my work there: First, COVID-19 is devastatingly real; second, we dermatologists can be valuable team members in the fight against this disease.
It is normal for us to feel scared, confused, and helpless; as 1% of the physician population, dermatologists represent a small focused fraction of the health care force. Nevertheless, we are all well-trained medical professionals who have taken the same Hippocratic Oath as other physicians. As members of the global health care team, we can each play a role in defeating COVID-19: We can be a trusted voice of reason, set an example, implement safe and effective distancing and hygiene precautions, and assist our local overburdened medical teams.
The magnitude and severity of COVID-19 can create a mass casualty–type phenomenon, overwhelming health care systems if the disease curve is not flattened. We can help flatten that curve by lengthening the pulse duration (to use dermatology jargon): that is, slowing the abrupt impact of cases to allow health care systems to triage, treat, and discharge in a more controlled manner.
How We Can Make a Difference
Despite representing a fraction of the health care team, we see a larger percentage of the population. On the Comfort, for example, dermatology visits accounted for approximately 20% of outpatient crew visits. We have an opportunity and a voice to reach a large percentage of the population directly. Whether we are now seeing patients face-to-face or virtually, we can spread the public health message and set an example. Wearing masks and social distancing do help to slow and markedly decrease the spread of SARS-CoV-2.
When you see patients in your office, consider the following:
• Have patients wait outside the office in their car and call the receptionist upon arrival.
• Have the receptionist call back the patient when the office is ready.
• Prescreen the patient before having him/her enter the clinic.
• Do not allow handshaking.
• Require everyone to wear a mask.
• Wear gloves.
• Have ample hand sanitizer openly available for all.
• Thoroughly clean or disinfect surfaces between patients.
Recalling the Difficult Experience of a Colleague-Patient
I think back to a crew member of Comfort who presented with new-onset pruritus and erythematous papules on the arms, legs, and torso. She was an intensive care unit nurse working 13-hour days, every day, for weeks on a COVID-positive unit—double-masked, gowned, wearing eye protection, in a warmer than usual intensive care unit, managing the most critically ill patients she’s ever cared for. Outside work, her life consisted of a commute on a government-chartered bus between Comfort and a contracted hotel while eating boxed meals. For 6 hours daily, she would—unsuccessfully—attempt to sleep with raging pruritus. Treating this routine case of eczema had a domino effect, improving her quality of life and thus allowing her to provide better care for the critically ill.
Let Us All Join in the Fight
As well-educated medical experts, we have the ability and the opportunity to reach outside our comfort zone and assist our medical colleagues. As I saw in New York City, the spectrum of specialists bravely worked together to meet overwhelming demand on the health care system and care for thousands of critically ill and dying patients. Dermatologists treated extensive eczema, ulcers, and other dermatoses on caretakers; triaged patients for appropriate allocation of care; and delivered care outside their comfort zone as physician extenders on inpatient and critical care units.
We are all in this together. I encourage all dermatologists who are in an area of need to ask your health care system how you can join the fight against SARS-CoV-2. Let’s step forward to help, in recognition of the oath we took to “prevent disease whenever we can.”
We are in unprecedented times. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is attacking our communities and, as with any battle, we face unexpected challenges from the global pandemic. What can dermatologists, as highly skilled health care experts, do to support the fight against coronavirus disease 2019 (COVID-19)?
In early 2020, I became involved in a fulfilling and stimulating opportunity to contribute as a US Navy reservist, having just returned from a 3-month deployment. I served in the Medical Operations Center aboard the hospital ship USNS Comfort, which was docked in New York Harbor, as liaison to surrounding New York City hospitals. I also served as sole dermatologist on the ship, caring for the dermatologic needs of our team and consulting on numerous COVID-19 inpatients.
In May 2020, upon return to Virginia from New York City, I served as senior medical officer to medically clear other Navy Reserve health care workers returning from the field hospital at the Jacob K. Javits Convention Center of New York and from serving as embedded caregivers in existing New York City hospitals. I share 2 very important observations from my work there: First, COVID-19 is devastatingly real; second, we dermatologists can be valuable team members in the fight against this disease.
It is normal for us to feel scared, confused, and helpless; as 1% of the physician population, dermatologists represent a small focused fraction of the health care force. Nevertheless, we are all well-trained medical professionals who have taken the same Hippocratic Oath as other physicians. As members of the global health care team, we can each play a role in defeating COVID-19: We can be a trusted voice of reason, set an example, implement safe and effective distancing and hygiene precautions, and assist our local overburdened medical teams.
The magnitude and severity of COVID-19 can create a mass casualty–type phenomenon, overwhelming health care systems if the disease curve is not flattened. We can help flatten that curve by lengthening the pulse duration (to use dermatology jargon): that is, slowing the abrupt impact of cases to allow health care systems to triage, treat, and discharge in a more controlled manner.
How We Can Make a Difference
Despite representing a fraction of the health care team, we see a larger percentage of the population. On the Comfort, for example, dermatology visits accounted for approximately 20% of outpatient crew visits. We have an opportunity and a voice to reach a large percentage of the population directly. Whether we are now seeing patients face-to-face or virtually, we can spread the public health message and set an example. Wearing masks and social distancing do help to slow and markedly decrease the spread of SARS-CoV-2.
When you see patients in your office, consider the following:
• Have patients wait outside the office in their car and call the receptionist upon arrival.
• Have the receptionist call back the patient when the office is ready.
• Prescreen the patient before having him/her enter the clinic.
• Do not allow handshaking.
• Require everyone to wear a mask.
• Wear gloves.
• Have ample hand sanitizer openly available for all.
• Thoroughly clean or disinfect surfaces between patients.
Recalling the Difficult Experience of a Colleague-Patient
I think back to a crew member of Comfort who presented with new-onset pruritus and erythematous papules on the arms, legs, and torso. She was an intensive care unit nurse working 13-hour days, every day, for weeks on a COVID-positive unit—double-masked, gowned, wearing eye protection, in a warmer than usual intensive care unit, managing the most critically ill patients she’s ever cared for. Outside work, her life consisted of a commute on a government-chartered bus between Comfort and a contracted hotel while eating boxed meals. For 6 hours daily, she would—unsuccessfully—attempt to sleep with raging pruritus. Treating this routine case of eczema had a domino effect, improving her quality of life and thus allowing her to provide better care for the critically ill.
Let Us All Join in the Fight
As well-educated medical experts, we have the ability and the opportunity to reach outside our comfort zone and assist our medical colleagues. As I saw in New York City, the spectrum of specialists bravely worked together to meet overwhelming demand on the health care system and care for thousands of critically ill and dying patients. Dermatologists treated extensive eczema, ulcers, and other dermatoses on caretakers; triaged patients for appropriate allocation of care; and delivered care outside their comfort zone as physician extenders on inpatient and critical care units.
We are all in this together. I encourage all dermatologists who are in an area of need to ask your health care system how you can join the fight against SARS-CoV-2. Let’s step forward to help, in recognition of the oath we took to “prevent disease whenever we can.”
We are in unprecedented times. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is attacking our communities and, as with any battle, we face unexpected challenges from the global pandemic. What can dermatologists, as highly skilled health care experts, do to support the fight against coronavirus disease 2019 (COVID-19)?
In early 2020, I became involved in a fulfilling and stimulating opportunity to contribute as a US Navy reservist, having just returned from a 3-month deployment. I served in the Medical Operations Center aboard the hospital ship USNS Comfort, which was docked in New York Harbor, as liaison to surrounding New York City hospitals. I also served as sole dermatologist on the ship, caring for the dermatologic needs of our team and consulting on numerous COVID-19 inpatients.
In May 2020, upon return to Virginia from New York City, I served as senior medical officer to medically clear other Navy Reserve health care workers returning from the field hospital at the Jacob K. Javits Convention Center of New York and from serving as embedded caregivers in existing New York City hospitals. I share 2 very important observations from my work there: First, COVID-19 is devastatingly real; second, we dermatologists can be valuable team members in the fight against this disease.
It is normal for us to feel scared, confused, and helpless; as 1% of the physician population, dermatologists represent a small focused fraction of the health care force. Nevertheless, we are all well-trained medical professionals who have taken the same Hippocratic Oath as other physicians. As members of the global health care team, we can each play a role in defeating COVID-19: We can be a trusted voice of reason, set an example, implement safe and effective distancing and hygiene precautions, and assist our local overburdened medical teams.
The magnitude and severity of COVID-19 can create a mass casualty–type phenomenon, overwhelming health care systems if the disease curve is not flattened. We can help flatten that curve by lengthening the pulse duration (to use dermatology jargon): that is, slowing the abrupt impact of cases to allow health care systems to triage, treat, and discharge in a more controlled manner.
How We Can Make a Difference
Despite representing a fraction of the health care team, we see a larger percentage of the population. On the Comfort, for example, dermatology visits accounted for approximately 20% of outpatient crew visits. We have an opportunity and a voice to reach a large percentage of the population directly. Whether we are now seeing patients face-to-face or virtually, we can spread the public health message and set an example. Wearing masks and social distancing do help to slow and markedly decrease the spread of SARS-CoV-2.
When you see patients in your office, consider the following:
• Have patients wait outside the office in their car and call the receptionist upon arrival.
• Have the receptionist call back the patient when the office is ready.
• Prescreen the patient before having him/her enter the clinic.
• Do not allow handshaking.
• Require everyone to wear a mask.
• Wear gloves.
• Have ample hand sanitizer openly available for all.
• Thoroughly clean or disinfect surfaces between patients.
Recalling the Difficult Experience of a Colleague-Patient
I think back to a crew member of Comfort who presented with new-onset pruritus and erythematous papules on the arms, legs, and torso. She was an intensive care unit nurse working 13-hour days, every day, for weeks on a COVID-positive unit—double-masked, gowned, wearing eye protection, in a warmer than usual intensive care unit, managing the most critically ill patients she’s ever cared for. Outside work, her life consisted of a commute on a government-chartered bus between Comfort and a contracted hotel while eating boxed meals. For 6 hours daily, she would—unsuccessfully—attempt to sleep with raging pruritus. Treating this routine case of eczema had a domino effect, improving her quality of life and thus allowing her to provide better care for the critically ill.
Let Us All Join in the Fight
As well-educated medical experts, we have the ability and the opportunity to reach outside our comfort zone and assist our medical colleagues. As I saw in New York City, the spectrum of specialists bravely worked together to meet overwhelming demand on the health care system and care for thousands of critically ill and dying patients. Dermatologists treated extensive eczema, ulcers, and other dermatoses on caretakers; triaged patients for appropriate allocation of care; and delivered care outside their comfort zone as physician extenders on inpatient and critical care units.
We are all in this together. I encourage all dermatologists who are in an area of need to ask your health care system how you can join the fight against SARS-CoV-2. Let’s step forward to help, in recognition of the oath we took to “prevent disease whenever we can.”
Practice Points
- Be aware of and promote coronavirus disease 2019 guidelines and recommendations from the Centers for Disease Control and Prevention and your local health department.
- Be prepared to push the limits of your comfort zone in an effort to assist the health care community.
Skin Eruption and Gastrointestinal Symptoms as Presentation of COVID-19
In December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) started an outbreak of respiratory illnesses in Wuhan, China. The respiratory disease was termed coronavirus disease 2019 (COVID-19) and rapidly spread worldwide, resulting in a pandemic classification on March 11, 2020. 1 Recently, several cutaneous manifestations of COVID-19 have been reported. Skin manifestations have been reported to be similar to other common viral infections. 2 However, there is a paucity of published clinical images of more atypical presentations.
Case Report
A 52-year-old black man presented via urgent store-and-forward teledermatology consultation from his primary care provider with a self-described “vesicular,” highly pruritic rash of both arms and legs of 1 week’s duration without involvement of the trunk, axillae, groin, face, genitalia, or any mucous membranes. He noted nausea, loss of appetite, and nonbloody diarrhea 4 days later. He denied fever, chills, dry cough, shortness of breath, or dyspnea. He had a history of hypertension and type 2 diabetes mellitus. There were no changes in medications; no outdoor activities, gardening, or yard work; no exposure to plants or metals; and no use of new personal care products.
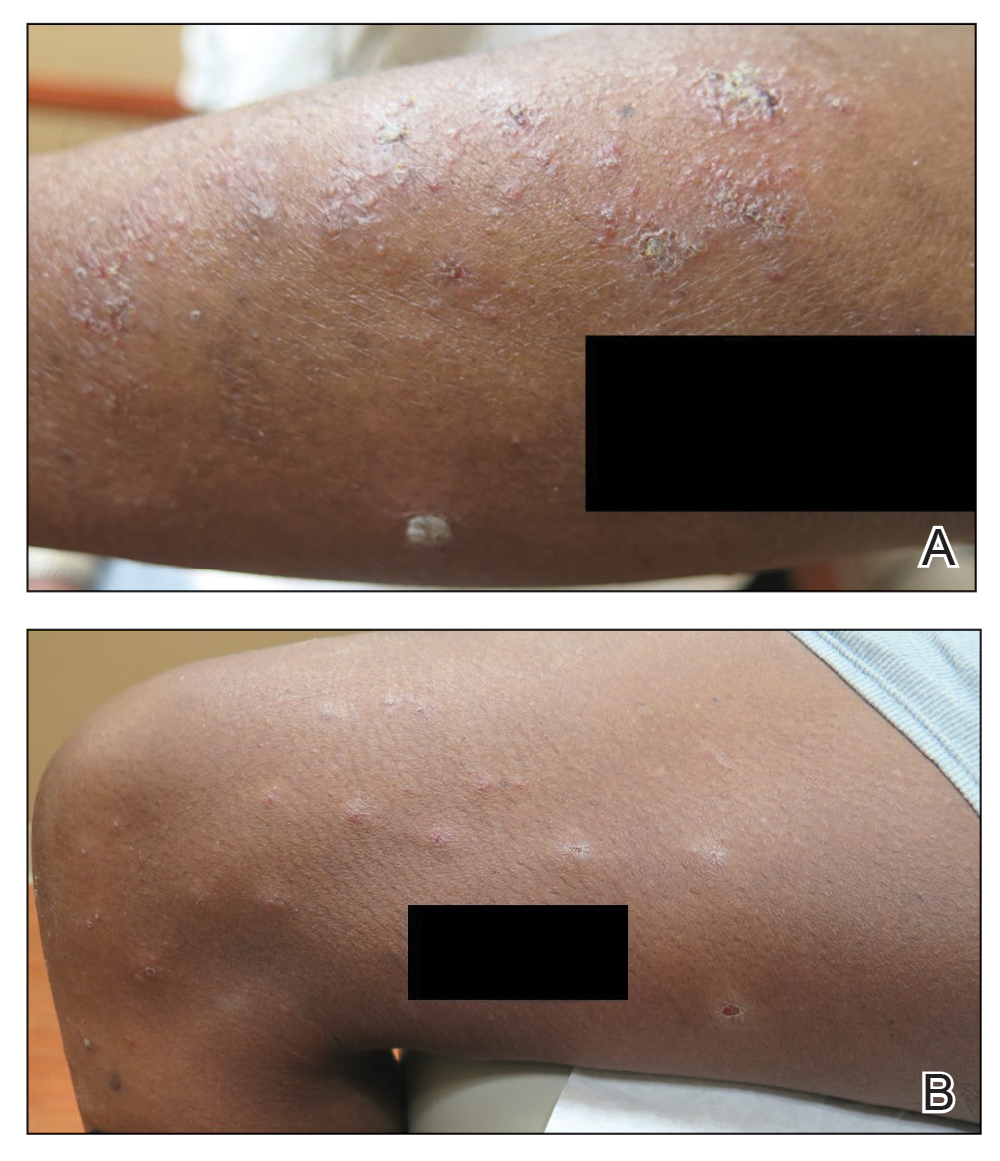
The digital images showed zones of flesh-colored to slightly erythematous, somewhat “juicy” papules with some coalescence into ill-defined plaques. There were scattered foci of scale and hemorrhagic crust that involved both palms, forearms (Figure, A), and legs (Figure, B). There were no intact vesicles, and a herald patch was not identified. Vital signs at the time of imaging were normal, with the exception of a low-grade fever (temperature, 37.3°C). Basic laboratory testing showed only mild leukocytosis with mild neutropenia and mild aspartate aminotransaminase elevation. A skin biopsy was not performed. Pulmonary imaging and workup were not performed because of the lack of respiratory symptoms.
The teledermatology differential diagnosis included a drug eruption, autosensitization eruption, unusual contact dermatitis, viral exanthem, secondary syphilis, and papular pityriasis rosea with an unusual distribution. The absence of changes in the patient’s medication regimen and the lack of outdoor activity in late winter made a drug eruption and contact dermatitis less likely, respectively. A rapid plasma reagin test drawn after disappearance of the rash was negative. Although the morphology of this eruption displayed some features of papular pityriasis rosea, this diagnosis was considered to be less likely given the presence of palmar involvement and the absence of any truncal lesions. This variant of pityriasis rosea is more commonly encountered in younger, darker-skinned patients.
Given the presence of an unusual rash on the extremities followed shortly by gastrointestinal (GI) symptoms and coupled with a low-grade fever, a nasopharyngeal swab was obtained to test for COVID-19 using a reverse transcriptase–polymerase chain reaction test. The results were positive.
The patient was treated with triamcinolone 0.1% slush (triamcinolone cream 0.1% mixed 1:1 with tap water) to the affected skin of the extremities 3 times daily, and he experienced a reduction in pruritus. He developed new lesions on the face and eyelids (not imaged) 2 days after teledermatology consultation. The facial involvement was treated with hydrocortisone cream 1%. During the following week, the GI symptoms and skin eruption completely resolved. However, postinflammatory hyperpigmentation was observed in areas of the resolved papules and plaques. Over the course of this illness, the patient reported no respiratory symptoms.
Comment
Coronavirus disease 2019 is caused by SARS-CoV2, an enveloped, nonsegmented, positive-sense RNA virus of the coronavirus family. It is currently believed that SARS-CoV-2 uses the angiotensin-converting enzyme 2 receptor to gain entry into human cells, leading to infection primarily affecting the lower respiratory tract.3 Patients suspected of COVID-19 infection most often present with fever, dry cough, dyspnea, and fatigue, while GI symptoms such as nausea, vomiting, and diarrhea are uncommon.4 More recently, several reports describe a variety of skin findings associated with COVID-19. A current theory suggests that the virus does not directly target keratinocytes but triggers a systemic immune response, leading to a diversity of skin morphologies.5 The main types of described cutaneous findings include pseudochilblains, overtly vesicular, urticarial, maculopapular, and livedo/necrosis.6 Others have described petechial7 and papulosquamous eruptions.8 Most of these patients initially presented with typical COVID-19 symptoms and frequently represented more severe cases of the disease. Additionally, the vesicular and papulosquamous eruptions reportedly occurred on the trunk and not the limbs, as in our case.
This confirmed COVID-19–positive patient presented with an ill-defined vesicular and papulosquamous-type eruption on the arms and legs and later developed only mild GI symptoms. By sharing this case, we report yet another skin manifestation of COVID-19 and propose the possible expansion of testing for SARS-CoV-2 in patients presenting with rash and GI symptoms, which holds the potential to increase the identification of COVID-19 in the population, thereby increasing strict contact tracing and slowing the spread of this pandemic.
- Ng OT, Marimuthu K, Chia PY, et al. SARS-CoV-2 infection among travelers returning from Wuhan, China. N Engl J Med. 2020;382:1476-1478.
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J Eur Acad Dermatol Venereol. 2020;34:E212-E213.
- Guo YR, Cao QD, Hong ZS, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak—an update on the status. Mil Med Res. 2020;7:11.
- Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-1720.
- Gianotti R, Zerbi P, Dodiuk-Gad RP. Clinical and histopathological study of skin dermatoses in patients affected by COVID-19 infection in the Northern part of Italy. J Dermatol Sci. 2020;98:141-143.
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77.
- Diaz-Guimaraens B, Dominguez-Santas M, Suarez-Valle A, et al. Petechial skin rash associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:820-822.
- Sanchez A, Sohier P, Benghanem S, et al. Digitate papulosquamous eruption associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:819-820.
In December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) started an outbreak of respiratory illnesses in Wuhan, China. The respiratory disease was termed coronavirus disease 2019 (COVID-19) and rapidly spread worldwide, resulting in a pandemic classification on March 11, 2020. 1 Recently, several cutaneous manifestations of COVID-19 have been reported. Skin manifestations have been reported to be similar to other common viral infections. 2 However, there is a paucity of published clinical images of more atypical presentations.
Case Report
A 52-year-old black man presented via urgent store-and-forward teledermatology consultation from his primary care provider with a self-described “vesicular,” highly pruritic rash of both arms and legs of 1 week’s duration without involvement of the trunk, axillae, groin, face, genitalia, or any mucous membranes. He noted nausea, loss of appetite, and nonbloody diarrhea 4 days later. He denied fever, chills, dry cough, shortness of breath, or dyspnea. He had a history of hypertension and type 2 diabetes mellitus. There were no changes in medications; no outdoor activities, gardening, or yard work; no exposure to plants or metals; and no use of new personal care products.
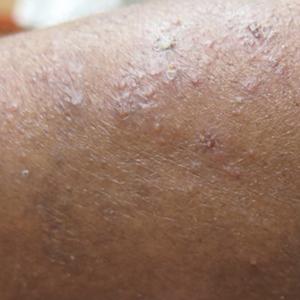
The digital images showed zones of flesh-colored to slightly erythematous, somewhat “juicy” papules with some coalescence into ill-defined plaques. There were scattered foci of scale and hemorrhagic crust that involved both palms, forearms (Figure, A), and legs (Figure, B). There were no intact vesicles, and a herald patch was not identified. Vital signs at the time of imaging were normal, with the exception of a low-grade fever (temperature, 37.3°C). Basic laboratory testing showed only mild leukocytosis with mild neutropenia and mild aspartate aminotransaminase elevation. A skin biopsy was not performed. Pulmonary imaging and workup were not performed because of the lack of respiratory symptoms.

The teledermatology differential diagnosis included a drug eruption, autosensitization eruption, unusual contact dermatitis, viral exanthem, secondary syphilis, and papular pityriasis rosea with an unusual distribution. The absence of changes in the patient’s medication regimen and the lack of outdoor activity in late winter made a drug eruption and contact dermatitis less likely, respectively. A rapid plasma reagin test drawn after disappearance of the rash was negative. Although the morphology of this eruption displayed some features of papular pityriasis rosea, this diagnosis was considered to be less likely given the presence of palmar involvement and the absence of any truncal lesions. This variant of pityriasis rosea is more commonly encountered in younger, darker-skinned patients.
Given the presence of an unusual rash on the extremities followed shortly by gastrointestinal (GI) symptoms and coupled with a low-grade fever, a nasopharyngeal swab was obtained to test for COVID-19 using a reverse transcriptase–polymerase chain reaction test. The results were positive.
The patient was treated with triamcinolone 0.1% slush (triamcinolone cream 0.1% mixed 1:1 with tap water) to the affected skin of the extremities 3 times daily, and he experienced a reduction in pruritus. He developed new lesions on the face and eyelids (not imaged) 2 days after teledermatology consultation. The facial involvement was treated with hydrocortisone cream 1%. During the following week, the GI symptoms and skin eruption completely resolved. However, postinflammatory hyperpigmentation was observed in areas of the resolved papules and plaques. Over the course of this illness, the patient reported no respiratory symptoms.
Comment
Coronavirus disease 2019 is caused by SARS-CoV2, an enveloped, nonsegmented, positive-sense RNA virus of the coronavirus family. It is currently believed that SARS-CoV-2 uses the angiotensin-converting enzyme 2 receptor to gain entry into human cells, leading to infection primarily affecting the lower respiratory tract.3 Patients suspected of COVID-19 infection most often present with fever, dry cough, dyspnea, and fatigue, while GI symptoms such as nausea, vomiting, and diarrhea are uncommon.4 More recently, several reports describe a variety of skin findings associated with COVID-19. A current theory suggests that the virus does not directly target keratinocytes but triggers a systemic immune response, leading to a diversity of skin morphologies.5 The main types of described cutaneous findings include pseudochilblains, overtly vesicular, urticarial, maculopapular, and livedo/necrosis.6 Others have described petechial7 and papulosquamous eruptions.8 Most of these patients initially presented with typical COVID-19 symptoms and frequently represented more severe cases of the disease. Additionally, the vesicular and papulosquamous eruptions reportedly occurred on the trunk and not the limbs, as in our case.
This confirmed COVID-19–positive patient presented with an ill-defined vesicular and papulosquamous-type eruption on the arms and legs and later developed only mild GI symptoms. By sharing this case, we report yet another skin manifestation of COVID-19 and propose the possible expansion of testing for SARS-CoV-2 in patients presenting with rash and GI symptoms, which holds the potential to increase the identification of COVID-19 in the population, thereby increasing strict contact tracing and slowing the spread of this pandemic.
In December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) started an outbreak of respiratory illnesses in Wuhan, China. The respiratory disease was termed coronavirus disease 2019 (COVID-19) and rapidly spread worldwide, resulting in a pandemic classification on March 11, 2020. 1 Recently, several cutaneous manifestations of COVID-19 have been reported. Skin manifestations have been reported to be similar to other common viral infections. 2 However, there is a paucity of published clinical images of more atypical presentations.
Case Report
A 52-year-old black man presented via urgent store-and-forward teledermatology consultation from his primary care provider with a self-described “vesicular,” highly pruritic rash of both arms and legs of 1 week’s duration without involvement of the trunk, axillae, groin, face, genitalia, or any mucous membranes. He noted nausea, loss of appetite, and nonbloody diarrhea 4 days later. He denied fever, chills, dry cough, shortness of breath, or dyspnea. He had a history of hypertension and type 2 diabetes mellitus. There were no changes in medications; no outdoor activities, gardening, or yard work; no exposure to plants or metals; and no use of new personal care products.
The digital images showed zones of flesh-colored to slightly erythematous, somewhat “juicy” papules with some coalescence into ill-defined plaques. There were scattered foci of scale and hemorrhagic crust that involved both palms, forearms (Figure, A), and legs (Figure, B). There were no intact vesicles, and a herald patch was not identified. Vital signs at the time of imaging were normal, with the exception of a low-grade fever (temperature, 37.3°C). Basic laboratory testing showed only mild leukocytosis with mild neutropenia and mild aspartate aminotransaminase elevation. A skin biopsy was not performed. Pulmonary imaging and workup were not performed because of the lack of respiratory symptoms.

The teledermatology differential diagnosis included a drug eruption, autosensitization eruption, unusual contact dermatitis, viral exanthem, secondary syphilis, and papular pityriasis rosea with an unusual distribution. The absence of changes in the patient’s medication regimen and the lack of outdoor activity in late winter made a drug eruption and contact dermatitis less likely, respectively. A rapid plasma reagin test drawn after disappearance of the rash was negative. Although the morphology of this eruption displayed some features of papular pityriasis rosea, this diagnosis was considered to be less likely given the presence of palmar involvement and the absence of any truncal lesions. This variant of pityriasis rosea is more commonly encountered in younger, darker-skinned patients.
Given the presence of an unusual rash on the extremities followed shortly by gastrointestinal (GI) symptoms and coupled with a low-grade fever, a nasopharyngeal swab was obtained to test for COVID-19 using a reverse transcriptase–polymerase chain reaction test. The results were positive.
The patient was treated with triamcinolone 0.1% slush (triamcinolone cream 0.1% mixed 1:1 with tap water) to the affected skin of the extremities 3 times daily, and he experienced a reduction in pruritus. He developed new lesions on the face and eyelids (not imaged) 2 days after teledermatology consultation. The facial involvement was treated with hydrocortisone cream 1%. During the following week, the GI symptoms and skin eruption completely resolved. However, postinflammatory hyperpigmentation was observed in areas of the resolved papules and plaques. Over the course of this illness, the patient reported no respiratory symptoms.
Comment
Coronavirus disease 2019 is caused by SARS-CoV2, an enveloped, nonsegmented, positive-sense RNA virus of the coronavirus family. It is currently believed that SARS-CoV-2 uses the angiotensin-converting enzyme 2 receptor to gain entry into human cells, leading to infection primarily affecting the lower respiratory tract.3 Patients suspected of COVID-19 infection most often present with fever, dry cough, dyspnea, and fatigue, while GI symptoms such as nausea, vomiting, and diarrhea are uncommon.4 More recently, several reports describe a variety of skin findings associated with COVID-19. A current theory suggests that the virus does not directly target keratinocytes but triggers a systemic immune response, leading to a diversity of skin morphologies.5 The main types of described cutaneous findings include pseudochilblains, overtly vesicular, urticarial, maculopapular, and livedo/necrosis.6 Others have described petechial7 and papulosquamous eruptions.8 Most of these patients initially presented with typical COVID-19 symptoms and frequently represented more severe cases of the disease. Additionally, the vesicular and papulosquamous eruptions reportedly occurred on the trunk and not the limbs, as in our case.
This confirmed COVID-19–positive patient presented with an ill-defined vesicular and papulosquamous-type eruption on the arms and legs and later developed only mild GI symptoms. By sharing this case, we report yet another skin manifestation of COVID-19 and propose the possible expansion of testing for SARS-CoV-2 in patients presenting with rash and GI symptoms, which holds the potential to increase the identification of COVID-19 in the population, thereby increasing strict contact tracing and slowing the spread of this pandemic.
- Ng OT, Marimuthu K, Chia PY, et al. SARS-CoV-2 infection among travelers returning from Wuhan, China. N Engl J Med. 2020;382:1476-1478.
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J Eur Acad Dermatol Venereol. 2020;34:E212-E213.
- Guo YR, Cao QD, Hong ZS, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak—an update on the status. Mil Med Res. 2020;7:11.
- Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-1720.
- Gianotti R, Zerbi P, Dodiuk-Gad RP. Clinical and histopathological study of skin dermatoses in patients affected by COVID-19 infection in the Northern part of Italy. J Dermatol Sci. 2020;98:141-143.
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77.
- Diaz-Guimaraens B, Dominguez-Santas M, Suarez-Valle A, et al. Petechial skin rash associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:820-822.
- Sanchez A, Sohier P, Benghanem S, et al. Digitate papulosquamous eruption associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:819-820.
- Ng OT, Marimuthu K, Chia PY, et al. SARS-CoV-2 infection among travelers returning from Wuhan, China. N Engl J Med. 2020;382:1476-1478.
- Recalcati S. Cutaneous manifestations in COVID-19: a first perspective. J Eur Acad Dermatol Venereol. 2020;34:E212-E213.
- Guo YR, Cao QD, Hong ZS, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak—an update on the status. Mil Med Res. 2020;7:11.
- Guan WJ, Ni ZY, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708-1720.
- Gianotti R, Zerbi P, Dodiuk-Gad RP. Clinical and histopathological study of skin dermatoses in patients affected by COVID-19 infection in the Northern part of Italy. J Dermatol Sci. 2020;98:141-143.
- Galván Casas C, Català A, Carretero Hernández G, et al. Classification of the cutaneous manifestations of COVID-19: a rapid prospective nationwide consensus study in Spain with 375 cases. Br J Dermatol. 2020;183:71-77.
- Diaz-Guimaraens B, Dominguez-Santas M, Suarez-Valle A, et al. Petechial skin rash associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:820-822.
- Sanchez A, Sohier P, Benghanem S, et al. Digitate papulosquamous eruption associated with severe acute respiratory syndrome coronavirus 2 infection. JAMA Dermatol. 2020;156:819-820.
Practice Points
- Patients with coronavirus disease 2019 (COVID-19) typically present with fever, dry cough, dyspnea, and fatigue, but cutaneous manifestations also have been reported.
- Awareness of atypical presentations of COVID-19, including uncommon cutaneous manifestations, may identify more cases and help slow the expansion of this pandemic.
Dermatology and Vaccines: We Must Do Better
Vaccines work. They are powerful tools that have saved millions of lives worldwide; however, a robust antivaccine movement has taken hold in the United States and worldwide despite overwhelming data in support of vaccination. In fact, vaccine hesitancy—the reluctance or refusal to vaccinate despite the availability of vaccines—was listed by the World Health Organization as one of the top 10 global health threats in 2019.1
Several vaccines have a role in dermatology, including the human papillomavirus (HPV) vaccine (Gardasil 9 [Merck Sharp & Dohme Corp]), the herpes zoster vaccines (Zostavax [Merck Sharp & Dohme Corp] and Shingrix [GlaxoSmithKline Biologicals]), and the measles-mumps-rubella vaccine, among others. These vaccinations are necessary for children and many adults alike, and they play a critical role in protecting both healthy and immunosuppressed patients.
Vaccine hesitancy is a growing threat to individual and public health that requires a response from all physicians. In our experience, dermatologists have been somewhat passive in advocating for vaccinations, possibly due to knowledge barriers or time constraints; however, this stance must change. Dermatologists must join the front lines in advocating for vaccinations, which are a proven and effective modality in promoting public health.
Dermatologists can employ the following practical tips to improve vaccination compliance among patients:
• Familiarize yourself with the Centers for Disease Control and Prevention immunization schedules and vaccination information sheets (https://www.cdc.gov/vaccines/hcp/vis/current-vis.html). Printed copies of informational handouts should be readily available to provide to patients in the office. The Centers for Disease Control and Prevention also offers tip sheets to guide conversations with patients (https://www.cdc.gov/vaccines/hcp/conversations/index.html).
• Prior to starting an immunosuppressive medication, confirm the patient’s immunization status. You should know which vaccines are live (containing an attenuated pathogen) and which are inactivated. Live vaccines typically are not administered to immunosuppressed patients.
• Use electronic medical records to help provide reminders to prompt administration of any necessary vaccines.
• Know the facts, especially regarding purported vaccine controversies, and be able to cite data on vaccine safety and efficacy. For example, when having a conversation with a patient you could state that vaccination against HPV, which can cause genital warts and certain cancers, has decreased the number of HPV infections by more than 70% in young women and 80% in teenaged girls.2 Cervical precancers were reduced by 40% in women vaccinated against HPV. Twelve years of monitoring data validates the safety and efficacy of the HPV vaccine—it is safe and effective, with benefits that outweigh any potential risks.2
• Tailor counseling based on the patient’s age and focus on benefits that directly impact the patient. For example, consider showing young adults photographs of genital warts while educating them that the HPV vaccine can help prevent this kind of infection in the future.
• Emphasize that vaccines are a routine part of comprehensive patient care and support this point by providing data and specific reasons for recommending vaccines.3 Avoid phrases such as, “Do you want the vaccine?” or “You could consider receiving the vaccine today,” which can imply that the vaccine is not necessary.
• Offer vaccines in your office or provide clear printed informational sheets directing patients to nearby primary care clinics, infectious disease clinics, or pharmacies where vaccinations are offered.
• Consider using social media to promote the benefits of vaccination among patients.
The recent coronavirus disease 2019 pandemic has brought the topic of vaccination into the limelight while highlighting that rampant misinformation can lead to distrust of health care workers. Dermatologists, along with all physicians, should be trusted advisors and advocates for public health. In addition to being knowledgeable, dermatologists must remain open-minded in having conversations with skeptical patients. Physicians must take the time and effort to promote vaccinations—the health of patients and the general public depends on it.
- Akbar R. Ten threats to global health in 2019. World Health Organization website. https://www.who.int/emergencies/ten-threats-to-global-health-in-2019. Published March 21, 2019. Accessed November 11, 2020.
- HPV vaccination is safe and effective. Centers for Disease Control and Prevention website. https://www.cdc.gov/hpv/parents/vaccinesafety.html. Updated April 29, 2019. Accessed November 11, 2020.
- How to give a strong recommendation to adult patients who require vaccination. Medscape website. https://www.medscape.com/viewarticle/842874. Published April 16, 2015. Accessed November 11, 2020.
Vaccines work. They are powerful tools that have saved millions of lives worldwide; however, a robust antivaccine movement has taken hold in the United States and worldwide despite overwhelming data in support of vaccination. In fact, vaccine hesitancy—the reluctance or refusal to vaccinate despite the availability of vaccines—was listed by the World Health Organization as one of the top 10 global health threats in 2019.1
Several vaccines have a role in dermatology, including the human papillomavirus (HPV) vaccine (Gardasil 9 [Merck Sharp & Dohme Corp]), the herpes zoster vaccines (Zostavax [Merck Sharp & Dohme Corp] and Shingrix [GlaxoSmithKline Biologicals]), and the measles-mumps-rubella vaccine, among others. These vaccinations are necessary for children and many adults alike, and they play a critical role in protecting both healthy and immunosuppressed patients.
Vaccine hesitancy is a growing threat to individual and public health that requires a response from all physicians. In our experience, dermatologists have been somewhat passive in advocating for vaccinations, possibly due to knowledge barriers or time constraints; however, this stance must change. Dermatologists must join the front lines in advocating for vaccinations, which are a proven and effective modality in promoting public health.
Dermatologists can employ the following practical tips to improve vaccination compliance among patients:
• Familiarize yourself with the Centers for Disease Control and Prevention immunization schedules and vaccination information sheets (https://www.cdc.gov/vaccines/hcp/vis/current-vis.html). Printed copies of informational handouts should be readily available to provide to patients in the office. The Centers for Disease Control and Prevention also offers tip sheets to guide conversations with patients (https://www.cdc.gov/vaccines/hcp/conversations/index.html).
• Prior to starting an immunosuppressive medication, confirm the patient’s immunization status. You should know which vaccines are live (containing an attenuated pathogen) and which are inactivated. Live vaccines typically are not administered to immunosuppressed patients.
• Use electronic medical records to help provide reminders to prompt administration of any necessary vaccines.
• Know the facts, especially regarding purported vaccine controversies, and be able to cite data on vaccine safety and efficacy. For example, when having a conversation with a patient you could state that vaccination against HPV, which can cause genital warts and certain cancers, has decreased the number of HPV infections by more than 70% in young women and 80% in teenaged girls.2 Cervical precancers were reduced by 40% in women vaccinated against HPV. Twelve years of monitoring data validates the safety and efficacy of the HPV vaccine—it is safe and effective, with benefits that outweigh any potential risks.2
• Tailor counseling based on the patient’s age and focus on benefits that directly impact the patient. For example, consider showing young adults photographs of genital warts while educating them that the HPV vaccine can help prevent this kind of infection in the future.
• Emphasize that vaccines are a routine part of comprehensive patient care and support this point by providing data and specific reasons for recommending vaccines.3 Avoid phrases such as, “Do you want the vaccine?” or “You could consider receiving the vaccine today,” which can imply that the vaccine is not necessary.
• Offer vaccines in your office or provide clear printed informational sheets directing patients to nearby primary care clinics, infectious disease clinics, or pharmacies where vaccinations are offered.
• Consider using social media to promote the benefits of vaccination among patients.
The recent coronavirus disease 2019 pandemic has brought the topic of vaccination into the limelight while highlighting that rampant misinformation can lead to distrust of health care workers. Dermatologists, along with all physicians, should be trusted advisors and advocates for public health. In addition to being knowledgeable, dermatologists must remain open-minded in having conversations with skeptical patients. Physicians must take the time and effort to promote vaccinations—the health of patients and the general public depends on it.
Vaccines work. They are powerful tools that have saved millions of lives worldwide; however, a robust antivaccine movement has taken hold in the United States and worldwide despite overwhelming data in support of vaccination. In fact, vaccine hesitancy—the reluctance or refusal to vaccinate despite the availability of vaccines—was listed by the World Health Organization as one of the top 10 global health threats in 2019.1
Several vaccines have a role in dermatology, including the human papillomavirus (HPV) vaccine (Gardasil 9 [Merck Sharp & Dohme Corp]), the herpes zoster vaccines (Zostavax [Merck Sharp & Dohme Corp] and Shingrix [GlaxoSmithKline Biologicals]), and the measles-mumps-rubella vaccine, among others. These vaccinations are necessary for children and many adults alike, and they play a critical role in protecting both healthy and immunosuppressed patients.
Vaccine hesitancy is a growing threat to individual and public health that requires a response from all physicians. In our experience, dermatologists have been somewhat passive in advocating for vaccinations, possibly due to knowledge barriers or time constraints; however, this stance must change. Dermatologists must join the front lines in advocating for vaccinations, which are a proven and effective modality in promoting public health.
Dermatologists can employ the following practical tips to improve vaccination compliance among patients:
• Familiarize yourself with the Centers for Disease Control and Prevention immunization schedules and vaccination information sheets (https://www.cdc.gov/vaccines/hcp/vis/current-vis.html). Printed copies of informational handouts should be readily available to provide to patients in the office. The Centers for Disease Control and Prevention also offers tip sheets to guide conversations with patients (https://www.cdc.gov/vaccines/hcp/conversations/index.html).
• Prior to starting an immunosuppressive medication, confirm the patient’s immunization status. You should know which vaccines are live (containing an attenuated pathogen) and which are inactivated. Live vaccines typically are not administered to immunosuppressed patients.
• Use electronic medical records to help provide reminders to prompt administration of any necessary vaccines.
• Know the facts, especially regarding purported vaccine controversies, and be able to cite data on vaccine safety and efficacy. For example, when having a conversation with a patient you could state that vaccination against HPV, which can cause genital warts and certain cancers, has decreased the number of HPV infections by more than 70% in young women and 80% in teenaged girls.2 Cervical precancers were reduced by 40% in women vaccinated against HPV. Twelve years of monitoring data validates the safety and efficacy of the HPV vaccine—it is safe and effective, with benefits that outweigh any potential risks.2
• Tailor counseling based on the patient’s age and focus on benefits that directly impact the patient. For example, consider showing young adults photographs of genital warts while educating them that the HPV vaccine can help prevent this kind of infection in the future.
• Emphasize that vaccines are a routine part of comprehensive patient care and support this point by providing data and specific reasons for recommending vaccines.3 Avoid phrases such as, “Do you want the vaccine?” or “You could consider receiving the vaccine today,” which can imply that the vaccine is not necessary.
• Offer vaccines in your office or provide clear printed informational sheets directing patients to nearby primary care clinics, infectious disease clinics, or pharmacies where vaccinations are offered.
• Consider using social media to promote the benefits of vaccination among patients.
The recent coronavirus disease 2019 pandemic has brought the topic of vaccination into the limelight while highlighting that rampant misinformation can lead to distrust of health care workers. Dermatologists, along with all physicians, should be trusted advisors and advocates for public health. In addition to being knowledgeable, dermatologists must remain open-minded in having conversations with skeptical patients. Physicians must take the time and effort to promote vaccinations—the health of patients and the general public depends on it.
- Akbar R. Ten threats to global health in 2019. World Health Organization website. https://www.who.int/emergencies/ten-threats-to-global-health-in-2019. Published March 21, 2019. Accessed November 11, 2020.
- HPV vaccination is safe and effective. Centers for Disease Control and Prevention website. https://www.cdc.gov/hpv/parents/vaccinesafety.html. Updated April 29, 2019. Accessed November 11, 2020.
- How to give a strong recommendation to adult patients who require vaccination. Medscape website. https://www.medscape.com/viewarticle/842874. Published April 16, 2015. Accessed November 11, 2020.
- Akbar R. Ten threats to global health in 2019. World Health Organization website. https://www.who.int/emergencies/ten-threats-to-global-health-in-2019. Published March 21, 2019. Accessed November 11, 2020.
- HPV vaccination is safe and effective. Centers for Disease Control and Prevention website. https://www.cdc.gov/hpv/parents/vaccinesafety.html. Updated April 29, 2019. Accessed November 11, 2020.
- How to give a strong recommendation to adult patients who require vaccination. Medscape website. https://www.medscape.com/viewarticle/842874. Published April 16, 2015. Accessed November 11, 2020.
COVID-19–related outcomes poor for patients with hematologic disease in ASH registry
Patients with hematologic disease who develop COVID-19 may experience substantial morbidity and mortality related to SARS-CoV-2 infection, according to recent registry data reported at the all-virtual annual meeting of the American Society of Hematology.
Overall mortality was 28% for the first 250 patients entered into the ASH Research Collaborative COVID-19 Registry for Hematology, researchers reported in an abstract of their study findings.
However, the burden of death and moderate-to-severe COVID-19 outcomes was highest in patients with poorer prognosis and those with relapsed/refractory hematological disease, they added.
The most commonly represented malignancies were acute leukemia, non-Hodgkin lymphoma, and myeloma or amyloidosis, according to the report.
Taken together, the findings do support an “emerging consensus” that COVID-19 related morbidity and mortality is significant in these patients, authors said – however, the current findings may not be reason enough to support a change in treatment course for the underlying disease.
“We see no reason, based on our data, to withhold intensive therapies from patients with underlying hematologic malignancies and favorable prognoses, if aggressive supportive care is consistent with patient preferences,” wrote the researchers.
ASH President Stephanie Lee, MD, MPH, said these registry findings are important to better understand how SARS-CoV-2 is affecting not only patients with hematologic diseases, but also individuals who experience COVID-19-related hematologic complications.
However, the findings are limited due to the heterogeneity of diseases, symptoms, and treatments represented in the registry, said Dr. Lee, associate director of the clinical research division at Fred Hutchinson Cancer Center in Seattle.
“More data will be coming in, but I think this is an example of trying to harness real-world information to try to learn things until we get more controlled studies,” Dr. Lee said in a media briefing held in advance of the ASH meeting.
Comorbidities and more
Patients with blood cancers are often older and may have comorbidities such as diabetes or hypertension that have been linked to poor COVID-19 outcomes, according to the authors of the report, led by William A. Wood, MD, MPH, associate professor of medicine with the UNC Lineberger Comprehensive Cancer Center in Chapel Hill, N.C.
Moreover, these patients may have underlying immune dysfunction and may receive chemotherapy or immunotherapy that is “profoundly immunosuppressive,” Dr. Wood and coauthors said in their report.
To date, however, risks of morbidity and mortality related to SARS-CoV-2 infection have not been well defined in this patient population, authors said.
More data is emerging now from the ASH Research Collaborative COVID-19 Registry for Hematology, which includes data on patients positive for COVID-19 who have a past or present hematologic condition or have experienced a hematologic complication related to COVID-19.
All data from the registry is being made available through a dashboard on the ASH Research Collaborative website, which as of Dec. 1, 2020, included 693 complete cases.
The data cut in the ASH abstract includes the first 250 patients enrolled at 74 sites around the world, the authors said. The most common malignancies included acute leukemia in 33%, non-Hodgkin lymphoma in 27%, and myeloma or amyloidosis in 16%.
The most frequently reported symptoms included fever in 73%, cough in 67%, dyspnea in 50%, and fatigue in 40%, according to that report.
At the time of this data snapshot, treatment with COVID-19-directed therapies including hydroxychloroquine or azithromycin were common, reported in 76 and 59 patients, respectively, in the cohort.
Batch submissions from sites with high incidence of COVID-19 infection are ongoing. The registry has been expanded to include nonmalignant hematologic diseases, and the registry will continue to accumulate data as a resource for the hematology community.
Overall mortality was 28% at the time, according to the abstract, with nearly all of the deaths occurring in patients classified as having COVID-19 that was moderate (i.e., requiring hospitalization) or severe (i.e., requiring ICU admission).
“In some instances, death occurred after a decision was made to forgo ICU admission in favor of a palliative approach,” said Dr. Wood and coauthors in their report.
Dr. Wood reported research funding from Pfizer, consultancy with Teladoc/Best Doctors, and honoraria from the ASH Research Collaborative. Coauthors provided disclosures related to Celgene, Madrigal Pharmaceuticals, Pharmacyclics, and Amgen, among others.
SOURCE: Wood WA et al. ASH 2020, Abstract 215.
Patients with hematologic disease who develop COVID-19 may experience substantial morbidity and mortality related to SARS-CoV-2 infection, according to recent registry data reported at the all-virtual annual meeting of the American Society of Hematology.
Overall mortality was 28% for the first 250 patients entered into the ASH Research Collaborative COVID-19 Registry for Hematology, researchers reported in an abstract of their study findings.
However, the burden of death and moderate-to-severe COVID-19 outcomes was highest in patients with poorer prognosis and those with relapsed/refractory hematological disease, they added.
The most commonly represented malignancies were acute leukemia, non-Hodgkin lymphoma, and myeloma or amyloidosis, according to the report.
Taken together, the findings do support an “emerging consensus” that COVID-19 related morbidity and mortality is significant in these patients, authors said – however, the current findings may not be reason enough to support a change in treatment course for the underlying disease.
“We see no reason, based on our data, to withhold intensive therapies from patients with underlying hematologic malignancies and favorable prognoses, if aggressive supportive care is consistent with patient preferences,” wrote the researchers.
ASH President Stephanie Lee, MD, MPH, said these registry findings are important to better understand how SARS-CoV-2 is affecting not only patients with hematologic diseases, but also individuals who experience COVID-19-related hematologic complications.
However, the findings are limited due to the heterogeneity of diseases, symptoms, and treatments represented in the registry, said Dr. Lee, associate director of the clinical research division at Fred Hutchinson Cancer Center in Seattle.
“More data will be coming in, but I think this is an example of trying to harness real-world information to try to learn things until we get more controlled studies,” Dr. Lee said in a media briefing held in advance of the ASH meeting.
Comorbidities and more
Patients with blood cancers are often older and may have comorbidities such as diabetes or hypertension that have been linked to poor COVID-19 outcomes, according to the authors of the report, led by William A. Wood, MD, MPH, associate professor of medicine with the UNC Lineberger Comprehensive Cancer Center in Chapel Hill, N.C.
Moreover, these patients may have underlying immune dysfunction and may receive chemotherapy or immunotherapy that is “profoundly immunosuppressive,” Dr. Wood and coauthors said in their report.
To date, however, risks of morbidity and mortality related to SARS-CoV-2 infection have not been well defined in this patient population, authors said.
More data is emerging now from the ASH Research Collaborative COVID-19 Registry for Hematology, which includes data on patients positive for COVID-19 who have a past or present hematologic condition or have experienced a hematologic complication related to COVID-19.
All data from the registry is being made available through a dashboard on the ASH Research Collaborative website, which as of Dec. 1, 2020, included 693 complete cases.
The data cut in the ASH abstract includes the first 250 patients enrolled at 74 sites around the world, the authors said. The most common malignancies included acute leukemia in 33%, non-Hodgkin lymphoma in 27%, and myeloma or amyloidosis in 16%.
The most frequently reported symptoms included fever in 73%, cough in 67%, dyspnea in 50%, and fatigue in 40%, according to that report.
At the time of this data snapshot, treatment with COVID-19-directed therapies including hydroxychloroquine or azithromycin were common, reported in 76 and 59 patients, respectively, in the cohort.
Batch submissions from sites with high incidence of COVID-19 infection are ongoing. The registry has been expanded to include nonmalignant hematologic diseases, and the registry will continue to accumulate data as a resource for the hematology community.
Overall mortality was 28% at the time, according to the abstract, with nearly all of the deaths occurring in patients classified as having COVID-19 that was moderate (i.e., requiring hospitalization) or severe (i.e., requiring ICU admission).
“In some instances, death occurred after a decision was made to forgo ICU admission in favor of a palliative approach,” said Dr. Wood and coauthors in their report.
Dr. Wood reported research funding from Pfizer, consultancy with Teladoc/Best Doctors, and honoraria from the ASH Research Collaborative. Coauthors provided disclosures related to Celgene, Madrigal Pharmaceuticals, Pharmacyclics, and Amgen, among others.
SOURCE: Wood WA et al. ASH 2020, Abstract 215.
Patients with hematologic disease who develop COVID-19 may experience substantial morbidity and mortality related to SARS-CoV-2 infection, according to recent registry data reported at the all-virtual annual meeting of the American Society of Hematology.
Overall mortality was 28% for the first 250 patients entered into the ASH Research Collaborative COVID-19 Registry for Hematology, researchers reported in an abstract of their study findings.
However, the burden of death and moderate-to-severe COVID-19 outcomes was highest in patients with poorer prognosis and those with relapsed/refractory hematological disease, they added.
The most commonly represented malignancies were acute leukemia, non-Hodgkin lymphoma, and myeloma or amyloidosis, according to the report.
Taken together, the findings do support an “emerging consensus” that COVID-19 related morbidity and mortality is significant in these patients, authors said – however, the current findings may not be reason enough to support a change in treatment course for the underlying disease.
“We see no reason, based on our data, to withhold intensive therapies from patients with underlying hematologic malignancies and favorable prognoses, if aggressive supportive care is consistent with patient preferences,” wrote the researchers.
ASH President Stephanie Lee, MD, MPH, said these registry findings are important to better understand how SARS-CoV-2 is affecting not only patients with hematologic diseases, but also individuals who experience COVID-19-related hematologic complications.
However, the findings are limited due to the heterogeneity of diseases, symptoms, and treatments represented in the registry, said Dr. Lee, associate director of the clinical research division at Fred Hutchinson Cancer Center in Seattle.
“More data will be coming in, but I think this is an example of trying to harness real-world information to try to learn things until we get more controlled studies,” Dr. Lee said in a media briefing held in advance of the ASH meeting.
Comorbidities and more
Patients with blood cancers are often older and may have comorbidities such as diabetes or hypertension that have been linked to poor COVID-19 outcomes, according to the authors of the report, led by William A. Wood, MD, MPH, associate professor of medicine with the UNC Lineberger Comprehensive Cancer Center in Chapel Hill, N.C.
Moreover, these patients may have underlying immune dysfunction and may receive chemotherapy or immunotherapy that is “profoundly immunosuppressive,” Dr. Wood and coauthors said in their report.
To date, however, risks of morbidity and mortality related to SARS-CoV-2 infection have not been well defined in this patient population, authors said.
More data is emerging now from the ASH Research Collaborative COVID-19 Registry for Hematology, which includes data on patients positive for COVID-19 who have a past or present hematologic condition or have experienced a hematologic complication related to COVID-19.
All data from the registry is being made available through a dashboard on the ASH Research Collaborative website, which as of Dec. 1, 2020, included 693 complete cases.
The data cut in the ASH abstract includes the first 250 patients enrolled at 74 sites around the world, the authors said. The most common malignancies included acute leukemia in 33%, non-Hodgkin lymphoma in 27%, and myeloma or amyloidosis in 16%.
The most frequently reported symptoms included fever in 73%, cough in 67%, dyspnea in 50%, and fatigue in 40%, according to that report.
At the time of this data snapshot, treatment with COVID-19-directed therapies including hydroxychloroquine or azithromycin were common, reported in 76 and 59 patients, respectively, in the cohort.
Batch submissions from sites with high incidence of COVID-19 infection are ongoing. The registry has been expanded to include nonmalignant hematologic diseases, and the registry will continue to accumulate data as a resource for the hematology community.
Overall mortality was 28% at the time, according to the abstract, with nearly all of the deaths occurring in patients classified as having COVID-19 that was moderate (i.e., requiring hospitalization) or severe (i.e., requiring ICU admission).
“In some instances, death occurred after a decision was made to forgo ICU admission in favor of a palliative approach,” said Dr. Wood and coauthors in their report.
Dr. Wood reported research funding from Pfizer, consultancy with Teladoc/Best Doctors, and honoraria from the ASH Research Collaborative. Coauthors provided disclosures related to Celgene, Madrigal Pharmaceuticals, Pharmacyclics, and Amgen, among others.
SOURCE: Wood WA et al. ASH 2020, Abstract 215.
FROM ASH 2020
Infant’s COVID-19–related myocardial injury reversed
Reports of signs of heart failure in adults with COVID-19 have been rare – just four such cases have been published since the outbreak started in China – and now a team of pediatric cardiologists in New York have reported a case of acute but reversible myocardial injury in an infant with COVID-19.
and right upper lobe atelectasis.
The 2-month-old infant went home after more than 2 weeks in the hospital with no apparent lingering cardiac effects of the illness and not needing any oral heart failure medications, Madhu Sharma, MD, of the Children’s Hospital and Montefiore in New York and colleagues reported in JACC Case Reports. With close follow-up, the child’s left ventricle size and systolic function have remained normal and mitral regurgitation resolved. The case report didn’t mention the infant’s gender.
But before the straightforward postdischarge course emerged, the infant was in a precarious state, and Dr. Sharma and her team were challenged to diagnose the underlying causes.
The child, who was born about 7 weeks premature, first came to the hospital having turned blue after choking on food. Nonrebreather mask ventilation was initiated in the ED, and an examination detected a holosystolic murmur. A test for COVID-19 was negative, but a later test was positive, and a chest x-ray exhibited cardiomegaly and signs of fluid and inflammation in the lungs.
An electrocardiogram detected sinus tachycardia, ST-segment depression and other anomalies in cardiac function. Further investigation with a transthoracic ECG showed severely depressed left ventricle systolic function with an ejection fraction of 30%, severe mitral regurgitation, and normal right ventricular systolic function.
Treatment included remdesivir and intravenous antibiotics. Through the hospital course, the patient was extubated to noninvasive ventilation, reintubated, put on intravenous steroid (methylprednisolone) and low-molecular-weight heparin, extubated, and tested throughout for cardiac function.
By day 14, left ventricle size and function normalized, and while the mitral regurgitation remained severe, it improved later without HF therapies. Left ventricle ejection fraction had recovered to 60%, and key cardiac biomarkers had normalized. On day 16, milrinone was discontinued, and the care team determined the patient no longer needed oral heart failure therapies.
“Most children with COVID-19 are either asymptomatic or have mild symptoms, but our case shows the potential for reversible myocardial injury in infants with COVID-19,” said Dr. Sharma. “Testing for COVID-19 in children presenting with signs and symptoms of heart failure is very important as we learn more about the impact of this virus.”
Dr. Sharma and coauthors have no relevant financial relationships to disclose.
SOURCE: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
Reports of signs of heart failure in adults with COVID-19 have been rare – just four such cases have been published since the outbreak started in China – and now a team of pediatric cardiologists in New York have reported a case of acute but reversible myocardial injury in an infant with COVID-19.
and right upper lobe atelectasis.
The 2-month-old infant went home after more than 2 weeks in the hospital with no apparent lingering cardiac effects of the illness and not needing any oral heart failure medications, Madhu Sharma, MD, of the Children’s Hospital and Montefiore in New York and colleagues reported in JACC Case Reports. With close follow-up, the child’s left ventricle size and systolic function have remained normal and mitral regurgitation resolved. The case report didn’t mention the infant’s gender.
But before the straightforward postdischarge course emerged, the infant was in a precarious state, and Dr. Sharma and her team were challenged to diagnose the underlying causes.
The child, who was born about 7 weeks premature, first came to the hospital having turned blue after choking on food. Nonrebreather mask ventilation was initiated in the ED, and an examination detected a holosystolic murmur. A test for COVID-19 was negative, but a later test was positive, and a chest x-ray exhibited cardiomegaly and signs of fluid and inflammation in the lungs.
An electrocardiogram detected sinus tachycardia, ST-segment depression and other anomalies in cardiac function. Further investigation with a transthoracic ECG showed severely depressed left ventricle systolic function with an ejection fraction of 30%, severe mitral regurgitation, and normal right ventricular systolic function.
Treatment included remdesivir and intravenous antibiotics. Through the hospital course, the patient was extubated to noninvasive ventilation, reintubated, put on intravenous steroid (methylprednisolone) and low-molecular-weight heparin, extubated, and tested throughout for cardiac function.
By day 14, left ventricle size and function normalized, and while the mitral regurgitation remained severe, it improved later without HF therapies. Left ventricle ejection fraction had recovered to 60%, and key cardiac biomarkers had normalized. On day 16, milrinone was discontinued, and the care team determined the patient no longer needed oral heart failure therapies.
“Most children with COVID-19 are either asymptomatic or have mild symptoms, but our case shows the potential for reversible myocardial injury in infants with COVID-19,” said Dr. Sharma. “Testing for COVID-19 in children presenting with signs and symptoms of heart failure is very important as we learn more about the impact of this virus.”
Dr. Sharma and coauthors have no relevant financial relationships to disclose.
SOURCE: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
Reports of signs of heart failure in adults with COVID-19 have been rare – just four such cases have been published since the outbreak started in China – and now a team of pediatric cardiologists in New York have reported a case of acute but reversible myocardial injury in an infant with COVID-19.
and right upper lobe atelectasis.
The 2-month-old infant went home after more than 2 weeks in the hospital with no apparent lingering cardiac effects of the illness and not needing any oral heart failure medications, Madhu Sharma, MD, of the Children’s Hospital and Montefiore in New York and colleagues reported in JACC Case Reports. With close follow-up, the child’s left ventricle size and systolic function have remained normal and mitral regurgitation resolved. The case report didn’t mention the infant’s gender.
But before the straightforward postdischarge course emerged, the infant was in a precarious state, and Dr. Sharma and her team were challenged to diagnose the underlying causes.
The child, who was born about 7 weeks premature, first came to the hospital having turned blue after choking on food. Nonrebreather mask ventilation was initiated in the ED, and an examination detected a holosystolic murmur. A test for COVID-19 was negative, but a later test was positive, and a chest x-ray exhibited cardiomegaly and signs of fluid and inflammation in the lungs.
An electrocardiogram detected sinus tachycardia, ST-segment depression and other anomalies in cardiac function. Further investigation with a transthoracic ECG showed severely depressed left ventricle systolic function with an ejection fraction of 30%, severe mitral regurgitation, and normal right ventricular systolic function.
Treatment included remdesivir and intravenous antibiotics. Through the hospital course, the patient was extubated to noninvasive ventilation, reintubated, put on intravenous steroid (methylprednisolone) and low-molecular-weight heparin, extubated, and tested throughout for cardiac function.
By day 14, left ventricle size and function normalized, and while the mitral regurgitation remained severe, it improved later without HF therapies. Left ventricle ejection fraction had recovered to 60%, and key cardiac biomarkers had normalized. On day 16, milrinone was discontinued, and the care team determined the patient no longer needed oral heart failure therapies.
“Most children with COVID-19 are either asymptomatic or have mild symptoms, but our case shows the potential for reversible myocardial injury in infants with COVID-19,” said Dr. Sharma. “Testing for COVID-19 in children presenting with signs and symptoms of heart failure is very important as we learn more about the impact of this virus.”
Dr. Sharma and coauthors have no relevant financial relationships to disclose.
SOURCE: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
FROM JACC CASE REPORTS
Key clinical point: Children presenting with COVID-19 should be tested for heart failure.
Major finding: A 2-month-old infant with COVID-19 had acute but reversible myocardial injury.
Study details: Single case report.
Disclosures: Dr. Sharma, MD, has no relevant financial relationships to disclose.
Source: Sharma M et al. JACC Case Rep. 2020. doi: 10.1016/j.jaccas.2020.09.031.
Obesity, hypoxia predict severity in children with COVID-19
based on data from 281 patients at 8 locations.
Manifestations of COVID-19 in children include respiratory disease similar to that seen in adults, but the full spectrum of disease in children has been studied mainly in single settings or with a focus on one clinical manifestation, wrote Danielle M. Fernandes, MD, of Albert Einstein College of Medicine, New York, and colleagues.
In a study published in the Journal of Pediatrics, the researchers identified 281 children hospitalized with COVID-19 and/or multisystem inflammatory syndrome in children (MIS-C) at 8 sites in Connecticut, New Jersey, and New York. A total of 143 (51%) had respiratory disease, 69 (25%) had MIS-C, and 69 (25%) had other manifestations of illness including 32 patients with gastrointestinal problems, 21 infants with fever, 6 cases of neurologic disease, 6 cases of diabetic ketoacidosis, and 4 patients with other indications. The median age of the patients was 10 years, 60% were male, 51% were Hispanic, and 23% were non-Hispanic Black. The most common comorbidities were obesity (34%) and asthma (14%).
Independent predictors of disease severity in children found
After controlling for multiple variables, obesity and hypoxia at hospital admission were significant independent predictors of severe respiratory disease, with odds ratios of 3.39 and 4.01, respectively. In addition, lower absolute lymphocyte count (OR, 8.33 per unit decrease in 109 cells/L) and higher C-reactive protein (OR, 1.06 per unit increase in mg/dL) were significantly predictive of severe MIS-C (P = .001 and P = .017, respectively).
“The association between weight and severe respiratory COVID-19 is consistent with the adult literature; however, the mechanisms of this association require further study,” Dr. Fernandes and associates noted.
Overall, children with MIS-C were significantly more likely to be non-Hispanic Black, compared with children with respiratory disease, an 18% difference. However, neither race/ethnicity nor socioeconomic status were significant predictors of disease severity, the researchers wrote.
During the study period, 7 patients (2%) died and 114 (41%) were admitted to the ICU.
“We found a wide array of clinical manifestations in children and youth hospitalized with SARS-CoV-2,” Dr. Fernandes and associates wrote. Notably, gastrointestinal symptoms, ocular symptoms, and dermatologic symptoms have rarely been noted in adults with COVID-19, but occurred in more than 30% of the pediatric patients.
“We also found that SARS-CoV-2 can be an incidental finding in a substantial number of hospitalized pediatric patients,” the researchers said.
The findings were limited by several factors including a population of patients only from Connecticut, New Jersey, and New York, and the possibility that decisions on hospital and ICU admission may have varied by location, the researchers said. In addition, approaches may have varied in the absence of data on the optimal treatment of MIS-C.
“This study builds on the growing body of evidence showing that mortality in hospitalized pediatric patients is low, compared with adults,” Dr. Fernandes and associates said. “However, it highlights that the young population is not universally spared from morbidity, and that even previously healthy children and youth can develop severe disease requiring supportive therapy.”
Findings confirm other clinical experience
The study was important to show that, “although most children are spared severe illness from COVID-19, some children are hospitalized both with acute COVID-19 respiratory disease, with MIS-C and with a range of other complications,” Adrienne Randolph, MD, of Boston Children’s Hospital and Harvard Medical School, Boston, said in an interview.
Dr. Randolph said she was not surprised by the study findings, “as we are also seeing these types of complications at Boston Children’s Hospital where I work.”
Additional research is needed on the outcomes of these patients, “especially the longer-term sequelae of having COVID-19 or MIS-C early in life,” she emphasized.
The take-home message to clinicians from the findings at this time is to be aware that children and adolescents can become severely ill from COVID-19–related complications, said Dr. Randolph. “Some of the laboratory values on presentation appear to be associated with disease severity.”
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Randolph disclosed funding from the Centers for Disease Control and Prevention to lead the Overcoming COVID-19 Study in U.S. Children and Adults.
SOURCE: Fernandes DM et al. J Pediatr. 2020 Nov 13. doi: 10.1016/j.jpeds.2020.11.016.
based on data from 281 patients at 8 locations.
Manifestations of COVID-19 in children include respiratory disease similar to that seen in adults, but the full spectrum of disease in children has been studied mainly in single settings or with a focus on one clinical manifestation, wrote Danielle M. Fernandes, MD, of Albert Einstein College of Medicine, New York, and colleagues.
In a study published in the Journal of Pediatrics, the researchers identified 281 children hospitalized with COVID-19 and/or multisystem inflammatory syndrome in children (MIS-C) at 8 sites in Connecticut, New Jersey, and New York. A total of 143 (51%) had respiratory disease, 69 (25%) had MIS-C, and 69 (25%) had other manifestations of illness including 32 patients with gastrointestinal problems, 21 infants with fever, 6 cases of neurologic disease, 6 cases of diabetic ketoacidosis, and 4 patients with other indications. The median age of the patients was 10 years, 60% were male, 51% were Hispanic, and 23% were non-Hispanic Black. The most common comorbidities were obesity (34%) and asthma (14%).
Independent predictors of disease severity in children found
After controlling for multiple variables, obesity and hypoxia at hospital admission were significant independent predictors of severe respiratory disease, with odds ratios of 3.39 and 4.01, respectively. In addition, lower absolute lymphocyte count (OR, 8.33 per unit decrease in 109 cells/L) and higher C-reactive protein (OR, 1.06 per unit increase in mg/dL) were significantly predictive of severe MIS-C (P = .001 and P = .017, respectively).
“The association between weight and severe respiratory COVID-19 is consistent with the adult literature; however, the mechanisms of this association require further study,” Dr. Fernandes and associates noted.
Overall, children with MIS-C were significantly more likely to be non-Hispanic Black, compared with children with respiratory disease, an 18% difference. However, neither race/ethnicity nor socioeconomic status were significant predictors of disease severity, the researchers wrote.
During the study period, 7 patients (2%) died and 114 (41%) were admitted to the ICU.
“We found a wide array of clinical manifestations in children and youth hospitalized with SARS-CoV-2,” Dr. Fernandes and associates wrote. Notably, gastrointestinal symptoms, ocular symptoms, and dermatologic symptoms have rarely been noted in adults with COVID-19, but occurred in more than 30% of the pediatric patients.
“We also found that SARS-CoV-2 can be an incidental finding in a substantial number of hospitalized pediatric patients,” the researchers said.
The findings were limited by several factors including a population of patients only from Connecticut, New Jersey, and New York, and the possibility that decisions on hospital and ICU admission may have varied by location, the researchers said. In addition, approaches may have varied in the absence of data on the optimal treatment of MIS-C.
“This study builds on the growing body of evidence showing that mortality in hospitalized pediatric patients is low, compared with adults,” Dr. Fernandes and associates said. “However, it highlights that the young population is not universally spared from morbidity, and that even previously healthy children and youth can develop severe disease requiring supportive therapy.”
Findings confirm other clinical experience
The study was important to show that, “although most children are spared severe illness from COVID-19, some children are hospitalized both with acute COVID-19 respiratory disease, with MIS-C and with a range of other complications,” Adrienne Randolph, MD, of Boston Children’s Hospital and Harvard Medical School, Boston, said in an interview.
Dr. Randolph said she was not surprised by the study findings, “as we are also seeing these types of complications at Boston Children’s Hospital where I work.”
Additional research is needed on the outcomes of these patients, “especially the longer-term sequelae of having COVID-19 or MIS-C early in life,” she emphasized.
The take-home message to clinicians from the findings at this time is to be aware that children and adolescents can become severely ill from COVID-19–related complications, said Dr. Randolph. “Some of the laboratory values on presentation appear to be associated with disease severity.”
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Randolph disclosed funding from the Centers for Disease Control and Prevention to lead the Overcoming COVID-19 Study in U.S. Children and Adults.
SOURCE: Fernandes DM et al. J Pediatr. 2020 Nov 13. doi: 10.1016/j.jpeds.2020.11.016.
based on data from 281 patients at 8 locations.
Manifestations of COVID-19 in children include respiratory disease similar to that seen in adults, but the full spectrum of disease in children has been studied mainly in single settings or with a focus on one clinical manifestation, wrote Danielle M. Fernandes, MD, of Albert Einstein College of Medicine, New York, and colleagues.
In a study published in the Journal of Pediatrics, the researchers identified 281 children hospitalized with COVID-19 and/or multisystem inflammatory syndrome in children (MIS-C) at 8 sites in Connecticut, New Jersey, and New York. A total of 143 (51%) had respiratory disease, 69 (25%) had MIS-C, and 69 (25%) had other manifestations of illness including 32 patients with gastrointestinal problems, 21 infants with fever, 6 cases of neurologic disease, 6 cases of diabetic ketoacidosis, and 4 patients with other indications. The median age of the patients was 10 years, 60% were male, 51% were Hispanic, and 23% were non-Hispanic Black. The most common comorbidities were obesity (34%) and asthma (14%).
Independent predictors of disease severity in children found
After controlling for multiple variables, obesity and hypoxia at hospital admission were significant independent predictors of severe respiratory disease, with odds ratios of 3.39 and 4.01, respectively. In addition, lower absolute lymphocyte count (OR, 8.33 per unit decrease in 109 cells/L) and higher C-reactive protein (OR, 1.06 per unit increase in mg/dL) were significantly predictive of severe MIS-C (P = .001 and P = .017, respectively).
“The association between weight and severe respiratory COVID-19 is consistent with the adult literature; however, the mechanisms of this association require further study,” Dr. Fernandes and associates noted.
Overall, children with MIS-C were significantly more likely to be non-Hispanic Black, compared with children with respiratory disease, an 18% difference. However, neither race/ethnicity nor socioeconomic status were significant predictors of disease severity, the researchers wrote.
During the study period, 7 patients (2%) died and 114 (41%) were admitted to the ICU.
“We found a wide array of clinical manifestations in children and youth hospitalized with SARS-CoV-2,” Dr. Fernandes and associates wrote. Notably, gastrointestinal symptoms, ocular symptoms, and dermatologic symptoms have rarely been noted in adults with COVID-19, but occurred in more than 30% of the pediatric patients.
“We also found that SARS-CoV-2 can be an incidental finding in a substantial number of hospitalized pediatric patients,” the researchers said.
The findings were limited by several factors including a population of patients only from Connecticut, New Jersey, and New York, and the possibility that decisions on hospital and ICU admission may have varied by location, the researchers said. In addition, approaches may have varied in the absence of data on the optimal treatment of MIS-C.
“This study builds on the growing body of evidence showing that mortality in hospitalized pediatric patients is low, compared with adults,” Dr. Fernandes and associates said. “However, it highlights that the young population is not universally spared from morbidity, and that even previously healthy children and youth can develop severe disease requiring supportive therapy.”
Findings confirm other clinical experience
The study was important to show that, “although most children are spared severe illness from COVID-19, some children are hospitalized both with acute COVID-19 respiratory disease, with MIS-C and with a range of other complications,” Adrienne Randolph, MD, of Boston Children’s Hospital and Harvard Medical School, Boston, said in an interview.
Dr. Randolph said she was not surprised by the study findings, “as we are also seeing these types of complications at Boston Children’s Hospital where I work.”
Additional research is needed on the outcomes of these patients, “especially the longer-term sequelae of having COVID-19 or MIS-C early in life,” she emphasized.
The take-home message to clinicians from the findings at this time is to be aware that children and adolescents can become severely ill from COVID-19–related complications, said Dr. Randolph. “Some of the laboratory values on presentation appear to be associated with disease severity.”
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Randolph disclosed funding from the Centers for Disease Control and Prevention to lead the Overcoming COVID-19 Study in U.S. Children and Adults.
SOURCE: Fernandes DM et al. J Pediatr. 2020 Nov 13. doi: 10.1016/j.jpeds.2020.11.016.
FROM THE JOURNAL OF PEDIATRICS
Leading hospitalists during a pandemic
As I write this, we are entering the third surge of the COVID-19 pandemic, with new cases, hospitalizations, and deaths from COVID-19 skyrocketing around the country. Worst of all, this surge has been most severely affecting areas of the nation least prepared to handle it (rural) and populations already marginalized by the health care system (Latinx and Black). Despite the onslaught of COVID-19, “pandemic fatigue” has begun to set in amongst colleagues, friends, and family, leading to challenges in adhering to social distancing and other infection-control measures, both at work and home.
In the face of the pandemic’s onslaught, hospitalists – who have faced the brunt of caring for patients with COVID-19, despite the absence of reporting about the subspecialty’s role – are faced with mustering the grit to respond with resolve, coordinated action, and empathy. Luckily, hospitalists are equipped with the very characteristics needed to lead teams, groups, and hospitals through the crisis of this pandemic. Ask yourself, why did you become a hospitalist? If you wanted steady predictability and control, there were many office-based specialties you could have chosen. You chose to become a hospitalist because you seek the challenges of clinical variety, problem-solving, systems improvement, and you are a natural team leader, whether you have been designated as such or not. In the words of John Quincy Adams, “if your actions inspire others to dream more, learn more, do more, and become more, you are a leader.”
As a leader, how can you lead your team through the series of trials and tribulations that this year has thrown at you? From COVID-19 to racism directed against Black and Latinx people to the behavioral health crisis, 2020 has likely made you feel as if you’re stuck in a ghoulish carnival fun house without an exit.
Yet this is where some leaders hit their stride, in what Bennis and Thomas describe as the “crucible of leadership.”1 There are many types of “crucibles of leadership,” according to Bennis and Thomas, and this year has thrown most of these at us: prejudice/bias, physical fatigue and illness, sudden elevation of responsibility to lead new processes, not to mention family stressors. Leaders who succeed in guiding their colleagues through these challenges have manifested critical skills: engaging others in shared meaning, having a distinctive and compelling voice, displaying integrity, and having adaptive capacity.
What exactly is adaptive capacity, the most important of these, in my opinion? Adaptive capacity requires understanding the new context of a crisis and how it has shifted team members’ needs and perceptions. It also requires what Bennis and Thomas call hardiness and what I call grit – the ability to face adversity, get knocked down, get up, and do it again.
There is probably no better example of a crisis leader with extraordinary adaptive capacity than Anglo-Irish explorer Sir Ernest Shackleton. Bitten by the bug of exploration, Shackleton failed at reaching the South Pole (1908-1909) but subsequently attempted to cross the Antarctic, departing South Georgia Island on Dec. 5, 1914. Depressingly for Shackleton, his ship, the Endurance, became stuck in sea ice on Jan. 19, 1915 before even reaching the continent. Drifting with the ice floe, his crew had set up a winter station hoping to be released from the ice later, but the Endurance was crushed by the pressure of sea ice and sank on Nov. 21, 1915. From there, Shackleton hoped to drift north to Paulet Island, 250 miles away, but eventually was forced to take his crew on lifeboats to the nearest land, Elephant Island, 346 miles from where the Endurance sank. He then took five of his men on an open boat, 828-mile journey to South Georgia Island. Encountering hurricane-force winds, the team landed on South Georgia Island 15 days later, only to face a climb of 32 miles over mountainous terrain to reach a whaling station. Shackleton eventually organized his men’s rescue on Elephant Island, reaching them on Aug. 30, 1916, 4½ months after he had set out for South Georgia Island. His entire crew survived, only to have two of them killed later in World War I.
You might consider Shackleton a failure for not even coming close to his original goal, but his success in saving his crew is regarded as the epitome of crisis leadership. As Harvard Business School professor Nancy F. Koehn, PhD, whose case study of Shackleton is one of the most popular at HBS, stated, “He thought he was going to be an entrepreneur of exploration, but he became an entrepreneur of survival.”2 Upon realizing the futility of his original mission, he pivoted immediately to the survival of his crew. “A man must shape himself to a new mark directly the old one goes to ground,” wrote Shackleton in his diary.3
Realizing that preserving his crew’s morale was critical, he maintained the crew’s everyday activities, despite the prospect of dying on the ice. He realized that he needed to keep up his own courage and confidence as well as that of his crew. Despite his ability to share the strategic focus of getting to safety with his men, he didn’t lose sight of day-to-day needs, such as keeping the crew entertained. When he encountered crew members who seemed problematic to his mission goals, he assigned them to his own tent.
Despite the extreme cold, his decision-making did not freeze – he acted decisively. He took risks when he thought appropriate, twice needing to abandon his efforts to drag a lifeboat full of supplies with his men toward the sea. “You can’t be afraid to make smart mistakes,” says Dr. Koehn. “That’s something we have no training in.”4 Most importantly, Shackleton took ultimate responsibility for his men’s survival, never resting until they had all been rescued. And he modeled a culture of shared responsibility for one another5 – he had once offered his only biscuit of the day on a prior expedition to his fellow explorer Frank Wild.
As winter arrives in 2020 and deepens into 2021, we will all be faced with leading our teams across the ice and to the safety of spring, and hopefully a vaccine. Whether we can get there with our entire crew depends on effective crisis leadership. But we can draw on the lessons provided by Shackleton and other crisis leaders in the past to guide us in the present.
Author disclosure: I studied the HBS case study “Leadership in Crisis: Ernest Shackleton and the Epic Voyage of the Endurance” as part of a 12-month certificate course in Safety, Quality, Informatics, and Leadership (SQIL) offered by Harvard Medical School.
Dr. Chang is chief of pediatric hospital medicine at Baystate Children’s Hospital in Springfield, Mass., and associate professor of pediatrics at the University of Massachusetts, also in Springfield.
References
1. HBR’s 10 must reads on leadership. Boston: Harvard Business Review Press, 2011.
2. Lagace M. Shackleton: An entrepreneur of survival. Harvard Business School. Working Knowledge website. Published 2003. Accessed 2020 Nov 19.
3. Koehn N. Leadership lessons from the Shackleton Expedition. The New York Times. 2011 Dec 25.
4. Potier B. Shackleton in business school. Harvard Public Affairs and Communications. The Harvard Gazette website. Published 2004. Accessed 2020 Nov 19.
5. Perkins D. 4 Lessons in crisis leadership from Shackleton’s expedition. In Leadership Essentials by HarpersCollins Leadership. Vol 2020. New York: HarpersCollins, 2020.
As I write this, we are entering the third surge of the COVID-19 pandemic, with new cases, hospitalizations, and deaths from COVID-19 skyrocketing around the country. Worst of all, this surge has been most severely affecting areas of the nation least prepared to handle it (rural) and populations already marginalized by the health care system (Latinx and Black). Despite the onslaught of COVID-19, “pandemic fatigue” has begun to set in amongst colleagues, friends, and family, leading to challenges in adhering to social distancing and other infection-control measures, both at work and home.
In the face of the pandemic’s onslaught, hospitalists – who have faced the brunt of caring for patients with COVID-19, despite the absence of reporting about the subspecialty’s role – are faced with mustering the grit to respond with resolve, coordinated action, and empathy. Luckily, hospitalists are equipped with the very characteristics needed to lead teams, groups, and hospitals through the crisis of this pandemic. Ask yourself, why did you become a hospitalist? If you wanted steady predictability and control, there were many office-based specialties you could have chosen. You chose to become a hospitalist because you seek the challenges of clinical variety, problem-solving, systems improvement, and you are a natural team leader, whether you have been designated as such or not. In the words of John Quincy Adams, “if your actions inspire others to dream more, learn more, do more, and become more, you are a leader.”
As a leader, how can you lead your team through the series of trials and tribulations that this year has thrown at you? From COVID-19 to racism directed against Black and Latinx people to the behavioral health crisis, 2020 has likely made you feel as if you’re stuck in a ghoulish carnival fun house without an exit.
Yet this is where some leaders hit their stride, in what Bennis and Thomas describe as the “crucible of leadership.”1 There are many types of “crucibles of leadership,” according to Bennis and Thomas, and this year has thrown most of these at us: prejudice/bias, physical fatigue and illness, sudden elevation of responsibility to lead new processes, not to mention family stressors. Leaders who succeed in guiding their colleagues through these challenges have manifested critical skills: engaging others in shared meaning, having a distinctive and compelling voice, displaying integrity, and having adaptive capacity.
What exactly is adaptive capacity, the most important of these, in my opinion? Adaptive capacity requires understanding the new context of a crisis and how it has shifted team members’ needs and perceptions. It also requires what Bennis and Thomas call hardiness and what I call grit – the ability to face adversity, get knocked down, get up, and do it again.
There is probably no better example of a crisis leader with extraordinary adaptive capacity than Anglo-Irish explorer Sir Ernest Shackleton. Bitten by the bug of exploration, Shackleton failed at reaching the South Pole (1908-1909) but subsequently attempted to cross the Antarctic, departing South Georgia Island on Dec. 5, 1914. Depressingly for Shackleton, his ship, the Endurance, became stuck in sea ice on Jan. 19, 1915 before even reaching the continent. Drifting with the ice floe, his crew had set up a winter station hoping to be released from the ice later, but the Endurance was crushed by the pressure of sea ice and sank on Nov. 21, 1915. From there, Shackleton hoped to drift north to Paulet Island, 250 miles away, but eventually was forced to take his crew on lifeboats to the nearest land, Elephant Island, 346 miles from where the Endurance sank. He then took five of his men on an open boat, 828-mile journey to South Georgia Island. Encountering hurricane-force winds, the team landed on South Georgia Island 15 days later, only to face a climb of 32 miles over mountainous terrain to reach a whaling station. Shackleton eventually organized his men’s rescue on Elephant Island, reaching them on Aug. 30, 1916, 4½ months after he had set out for South Georgia Island. His entire crew survived, only to have two of them killed later in World War I.
You might consider Shackleton a failure for not even coming close to his original goal, but his success in saving his crew is regarded as the epitome of crisis leadership. As Harvard Business School professor Nancy F. Koehn, PhD, whose case study of Shackleton is one of the most popular at HBS, stated, “He thought he was going to be an entrepreneur of exploration, but he became an entrepreneur of survival.”2 Upon realizing the futility of his original mission, he pivoted immediately to the survival of his crew. “A man must shape himself to a new mark directly the old one goes to ground,” wrote Shackleton in his diary.3
Realizing that preserving his crew’s morale was critical, he maintained the crew’s everyday activities, despite the prospect of dying on the ice. He realized that he needed to keep up his own courage and confidence as well as that of his crew. Despite his ability to share the strategic focus of getting to safety with his men, he didn’t lose sight of day-to-day needs, such as keeping the crew entertained. When he encountered crew members who seemed problematic to his mission goals, he assigned them to his own tent.
Despite the extreme cold, his decision-making did not freeze – he acted decisively. He took risks when he thought appropriate, twice needing to abandon his efforts to drag a lifeboat full of supplies with his men toward the sea. “You can’t be afraid to make smart mistakes,” says Dr. Koehn. “That’s something we have no training in.”4 Most importantly, Shackleton took ultimate responsibility for his men’s survival, never resting until they had all been rescued. And he modeled a culture of shared responsibility for one another5 – he had once offered his only biscuit of the day on a prior expedition to his fellow explorer Frank Wild.
As winter arrives in 2020 and deepens into 2021, we will all be faced with leading our teams across the ice and to the safety of spring, and hopefully a vaccine. Whether we can get there with our entire crew depends on effective crisis leadership. But we can draw on the lessons provided by Shackleton and other crisis leaders in the past to guide us in the present.
Author disclosure: I studied the HBS case study “Leadership in Crisis: Ernest Shackleton and the Epic Voyage of the Endurance” as part of a 12-month certificate course in Safety, Quality, Informatics, and Leadership (SQIL) offered by Harvard Medical School.
Dr. Chang is chief of pediatric hospital medicine at Baystate Children’s Hospital in Springfield, Mass., and associate professor of pediatrics at the University of Massachusetts, also in Springfield.
References
1. HBR’s 10 must reads on leadership. Boston: Harvard Business Review Press, 2011.
2. Lagace M. Shackleton: An entrepreneur of survival. Harvard Business School. Working Knowledge website. Published 2003. Accessed 2020 Nov 19.
3. Koehn N. Leadership lessons from the Shackleton Expedition. The New York Times. 2011 Dec 25.
4. Potier B. Shackleton in business school. Harvard Public Affairs and Communications. The Harvard Gazette website. Published 2004. Accessed 2020 Nov 19.
5. Perkins D. 4 Lessons in crisis leadership from Shackleton’s expedition. In Leadership Essentials by HarpersCollins Leadership. Vol 2020. New York: HarpersCollins, 2020.
As I write this, we are entering the third surge of the COVID-19 pandemic, with new cases, hospitalizations, and deaths from COVID-19 skyrocketing around the country. Worst of all, this surge has been most severely affecting areas of the nation least prepared to handle it (rural) and populations already marginalized by the health care system (Latinx and Black). Despite the onslaught of COVID-19, “pandemic fatigue” has begun to set in amongst colleagues, friends, and family, leading to challenges in adhering to social distancing and other infection-control measures, both at work and home.
In the face of the pandemic’s onslaught, hospitalists – who have faced the brunt of caring for patients with COVID-19, despite the absence of reporting about the subspecialty’s role – are faced with mustering the grit to respond with resolve, coordinated action, and empathy. Luckily, hospitalists are equipped with the very characteristics needed to lead teams, groups, and hospitals through the crisis of this pandemic. Ask yourself, why did you become a hospitalist? If you wanted steady predictability and control, there were many office-based specialties you could have chosen. You chose to become a hospitalist because you seek the challenges of clinical variety, problem-solving, systems improvement, and you are a natural team leader, whether you have been designated as such or not. In the words of John Quincy Adams, “if your actions inspire others to dream more, learn more, do more, and become more, you are a leader.”
As a leader, how can you lead your team through the series of trials and tribulations that this year has thrown at you? From COVID-19 to racism directed against Black and Latinx people to the behavioral health crisis, 2020 has likely made you feel as if you’re stuck in a ghoulish carnival fun house without an exit.
Yet this is where some leaders hit their stride, in what Bennis and Thomas describe as the “crucible of leadership.”1 There are many types of “crucibles of leadership,” according to Bennis and Thomas, and this year has thrown most of these at us: prejudice/bias, physical fatigue and illness, sudden elevation of responsibility to lead new processes, not to mention family stressors. Leaders who succeed in guiding their colleagues through these challenges have manifested critical skills: engaging others in shared meaning, having a distinctive and compelling voice, displaying integrity, and having adaptive capacity.
What exactly is adaptive capacity, the most important of these, in my opinion? Adaptive capacity requires understanding the new context of a crisis and how it has shifted team members’ needs and perceptions. It also requires what Bennis and Thomas call hardiness and what I call grit – the ability to face adversity, get knocked down, get up, and do it again.
There is probably no better example of a crisis leader with extraordinary adaptive capacity than Anglo-Irish explorer Sir Ernest Shackleton. Bitten by the bug of exploration, Shackleton failed at reaching the South Pole (1908-1909) but subsequently attempted to cross the Antarctic, departing South Georgia Island on Dec. 5, 1914. Depressingly for Shackleton, his ship, the Endurance, became stuck in sea ice on Jan. 19, 1915 before even reaching the continent. Drifting with the ice floe, his crew had set up a winter station hoping to be released from the ice later, but the Endurance was crushed by the pressure of sea ice and sank on Nov. 21, 1915. From there, Shackleton hoped to drift north to Paulet Island, 250 miles away, but eventually was forced to take his crew on lifeboats to the nearest land, Elephant Island, 346 miles from where the Endurance sank. He then took five of his men on an open boat, 828-mile journey to South Georgia Island. Encountering hurricane-force winds, the team landed on South Georgia Island 15 days later, only to face a climb of 32 miles over mountainous terrain to reach a whaling station. Shackleton eventually organized his men’s rescue on Elephant Island, reaching them on Aug. 30, 1916, 4½ months after he had set out for South Georgia Island. His entire crew survived, only to have two of them killed later in World War I.
You might consider Shackleton a failure for not even coming close to his original goal, but his success in saving his crew is regarded as the epitome of crisis leadership. As Harvard Business School professor Nancy F. Koehn, PhD, whose case study of Shackleton is one of the most popular at HBS, stated, “He thought he was going to be an entrepreneur of exploration, but he became an entrepreneur of survival.”2 Upon realizing the futility of his original mission, he pivoted immediately to the survival of his crew. “A man must shape himself to a new mark directly the old one goes to ground,” wrote Shackleton in his diary.3
Realizing that preserving his crew’s morale was critical, he maintained the crew’s everyday activities, despite the prospect of dying on the ice. He realized that he needed to keep up his own courage and confidence as well as that of his crew. Despite his ability to share the strategic focus of getting to safety with his men, he didn’t lose sight of day-to-day needs, such as keeping the crew entertained. When he encountered crew members who seemed problematic to his mission goals, he assigned them to his own tent.
Despite the extreme cold, his decision-making did not freeze – he acted decisively. He took risks when he thought appropriate, twice needing to abandon his efforts to drag a lifeboat full of supplies with his men toward the sea. “You can’t be afraid to make smart mistakes,” says Dr. Koehn. “That’s something we have no training in.”4 Most importantly, Shackleton took ultimate responsibility for his men’s survival, never resting until they had all been rescued. And he modeled a culture of shared responsibility for one another5 – he had once offered his only biscuit of the day on a prior expedition to his fellow explorer Frank Wild.
As winter arrives in 2020 and deepens into 2021, we will all be faced with leading our teams across the ice and to the safety of spring, and hopefully a vaccine. Whether we can get there with our entire crew depends on effective crisis leadership. But we can draw on the lessons provided by Shackleton and other crisis leaders in the past to guide us in the present.
Author disclosure: I studied the HBS case study “Leadership in Crisis: Ernest Shackleton and the Epic Voyage of the Endurance” as part of a 12-month certificate course in Safety, Quality, Informatics, and Leadership (SQIL) offered by Harvard Medical School.
Dr. Chang is chief of pediatric hospital medicine at Baystate Children’s Hospital in Springfield, Mass., and associate professor of pediatrics at the University of Massachusetts, also in Springfield.
References
1. HBR’s 10 must reads on leadership. Boston: Harvard Business Review Press, 2011.
2. Lagace M. Shackleton: An entrepreneur of survival. Harvard Business School. Working Knowledge website. Published 2003. Accessed 2020 Nov 19.
3. Koehn N. Leadership lessons from the Shackleton Expedition. The New York Times. 2011 Dec 25.
4. Potier B. Shackleton in business school. Harvard Public Affairs and Communications. The Harvard Gazette website. Published 2004. Accessed 2020 Nov 19.
5. Perkins D. 4 Lessons in crisis leadership from Shackleton’s expedition. In Leadership Essentials by HarpersCollins Leadership. Vol 2020. New York: HarpersCollins, 2020.
Several strategies can prevent pandemic-related weight gain
If you have been faithfully following the COVID-19 stay-at-home restrictive orders, you may have become a victim of “COVID-15,” the additional, unexpected, unwanted 10- to 15-pound weight gain that is making your clothes not fit so well any more.
A change in routine; being home in comfy, stretchable clothing in front of the TV; and having unhealthy, processed foods ready to grab have set us up to lose the battle with COVID-15. We are set up to gain the weight because of excessive or unhealthful eating, taking an extra daily shot of alcohol, and being inactive, bored, depressed, anxious, and isolated from coworkers and family. Beware – weight gain can be “catching”; we tend to adopt the same poor eating habits and eat the same junk foods as those around us.
Since psychiatry can be a sedentary profession, I’ve (R.W.C.) kept myself very active and physically fit. Prior to the pandemic, I played tennis and ran every day. I was obese only once in my life. I had not realized that I had gained a lot of weight.
Thankfully, a physician called me “obese.” Initially, I was angry at the doctor, however, I realized that he did me the biggest favor of my life. I changed my diet and eating habits, and for the past 20 years, kept my weight between 135-140 pounds and my BMI at 23 consistently – until the pandemic stress caused me to fall into the same bad eating habits that have caused many others to gain the COVID-15.
I was surprised to see that when I weighed myself, and I had gained 12 pounds! I immediately modified my diet and increased my physical activity. I have now lost the extra 12 pounds and will offer suggestions that may help you and your patients exceed your prepandemic physical condition.
Possible solutions
1. Keep a food journal. Write down what you eat, the amount of food you eat, the time you are eating, and your mood at that moment. Keeping a small notebook to record what and when you eat is important because upon review, it will make you face reality and be accountable for what you put in your mouth. Until you review your journal, you may have underestimated the amount, as well as the kinds, of food and drinks you actually consume. A food journal can show your areas of struggle and unhealthy eating habits and help you make necessary changes in your habits and diet to eventually lose weight. You will be less likely to eat junk food or have an extra serving of food. If you do not want to use paper and pencil, you can download an app on your phone, such as myplate tracker to keep track of your food and calorie intake. Do your journaling immediately after you eat and include snacks; do not wait until night time to record your food and journal. Include your mood or how you felt during your meal or snack (for example, were you bored, sad, or anxious) since this information will indicate why you may be overeating.
2. Develop healthful eating habits. Eat a maximum of three meals and three snacks per day but eat only when you are hungry (that is, when your stomach growls or you feel light headed). Limiting yourself to eating only when you are hungry will help eliminate emotional eating to fill a loss in your life or to deal with feelings of stress, anxiety, sadness, or isolation, which have been exacerbated by the pandemic. Buy eat only healthful foods and not items with empty calories, such as chips, cake, and items with sugar. When you are eating, devote yourself to that activity only, eat slowly, and savor each bite. Do not watch television during your meal time.
3. Record the amount and type of exercise you engage in each day and determine the number of calories burned. Walk, run, or bicycle outside, or exercise inside with stretching, weights, or an exercycle. You may use a website, such as diet tool on WebMD.com to calculate daily calories burned. To lose weight, calories burned during a day must exceed caloric intake. You may want to invest in a Fitbit or an Apple Watch and use the health section to determine your caloric intake versus output. Analyzing your caloric data will provide a concrete measure of your progress.
4. Do not overconsume calories or underconsume protein. Protein plays a key role in the creation and maintenance of every cell of your body, and because the body does not store protein, it is important to consume it every day. To meet basic protein requirements, the DRI (Dietary Reference Intake) recommends 0.36 grams of protein per pound (0.8 grams per kg) of body weight. This amounts to: 56 grams per day for the average sedentary man, and 46 grams per day for the average sedentary woman. There is also an app entitled the Protein Tracker that can simplify your calculations.
5. Drink water. It is important to be hydrated to regulate body temperature, keep joints lubricated, prevent infections, deliver nutrients to cells, and keep organs functioning properly. Being well hydrated also improves sleep, cognition, and mood. Your daily water intake by ounce should be equal to your weight in pounds multiplied by two-thirds (or 67%) to determine the amount of water to drink daily. For example, if you weigh 175 pounds, you would multiply 175 by two-thirds and learn that you should be drinking about 117 ounces of water every day. You can also meet some of your daily water requirements by consuming fruits and vegetables, such as tomatoes, watermelon, lettuce, etc.
Also, drink 2 cups (16 oz.) of water before every meal: Often when you feel hungry, it is because your body simply needs water. Science has proven that drinking 2 cups of water before every meal helps you to eat less during meal time and lose weight. If you do this three times daily – at breakfast, lunch, and dinner – you have already consumed 48 ounces of water.
6. Keep track of your progress. In addition to keeping and analyzing your food journal, weigh yourself once or twice a week. Do not weigh yourself every day; you will not see any results on a day-to-day basis, but once a week gives your body time to regulate and show progress. Always calibrate/zero your scale before each use, and weigh yourself at the same time of the day (preferably after you first wake up in the morning) while wearing the same type of clothing. Keep a record of your weight in your journal to track your progress. Do not panic if the scale indicates you gained 1, 2, or 3 pounds, your weight can fluctuate because of glycogen storage, sodium retention, human bias, reporting or recall errors, and home scales can have a plus or minus 3 pound margin of error. Look at your weight trend over time. You may prefer buying a scale that indicates both weight and body mass index.
7. Celebrate and reward yourself with nonfood items. A healthful fitness and diet regime requires energy and dedication, so if you are able to follow a healthful routine, reward yourself with nonfood rewards for your good choices and new habits as an incentive to maintain your healthful behavior.
8. Don’t buy it if you can’t stop eating it. The biggest decision you make is when you decide what you are going to buy. Don’t lie to yourself in the store that you will only eat one at a time. Only buy what you can afford to binge eat if you can’t stop yourself from eating any particular type of food.
9. Have someone hide the food you can’t resist. You can’t eat what you can’t find. If you can’t avoid having irresistible food around, ask another adult to hide the food from you.
10. Learn what harm foods can cause in your body. Read about the effects of high blood sugar and high blood pressure can cause in your body. Find out which foods boost your immune system. Demonize the bad foods in your mind. Make up your mind before you go into the store that you are going to read food labels and find the best quality food with the lowest amount of sugar or saturated fat. Appreciate the flavor of vegetables and fruit.
11. Treat sugar as if it were an addictive drug. You can’t have just one. If you reduce your craving for sugar by slowly reducing your intake of sugar, you will find that you don’t crave sugar any more. This won’t be easy, but once it is done, you will be preventing many of the ravages that sugar takes on your body over time. But you can’t have one piece of pie because the craving will come back. At some point, it may be more likely that you find that piece of pie too sweet.
Here are a few other ideas: Buy a gift for yourself or new clothes, makeup, a plant or flowers, running shoes, exercise clothes, fitness tracker, water bottle, book, movie or network subscription. Improve your home décor. Or treat yourself to online lessons for painting, music, and so on. Or you might adopt a dog, donate food to a shelter or food bank; or organize and declutter your home since staying busy will give you a reason not to eat. In nice weather, enjoy the outdoors by going for a walk, run, bikeride or by gardening.
We are all worried about getting COVID-19. Preventing COVID-15 will go a long way toward boosting our immune systems to help protect us from the coronavirus.
Dr. Cohen is board-certified in psychiatry and has had a private practice in Philadelphia for more than 35 years. His areas of specialty include sports psychiatry, agoraphobia, depression, and substance abuse. In addition, Dr. Cohen is a former professor of psychiatry, family medicine, and otolaryngology at Thomas Jefferson University in Philadelphia. He has no conflicts of interest. Ms. Cohen holds an MBA from Temple University in Philadelphia with a focus on health care administration. Previously, Ms. Cohen was an associate administrator at Hahnemann University Hospital and an executive at the Health Services Council, both in Philadelphia. She currently writes biographical summaries of notable 18th- and 19th-century women. Ms. Cohen has no conflicts of interest.
If you have been faithfully following the COVID-19 stay-at-home restrictive orders, you may have become a victim of “COVID-15,” the additional, unexpected, unwanted 10- to 15-pound weight gain that is making your clothes not fit so well any more.
A change in routine; being home in comfy, stretchable clothing in front of the TV; and having unhealthy, processed foods ready to grab have set us up to lose the battle with COVID-15. We are set up to gain the weight because of excessive or unhealthful eating, taking an extra daily shot of alcohol, and being inactive, bored, depressed, anxious, and isolated from coworkers and family. Beware – weight gain can be “catching”; we tend to adopt the same poor eating habits and eat the same junk foods as those around us.
Since psychiatry can be a sedentary profession, I’ve (R.W.C.) kept myself very active and physically fit. Prior to the pandemic, I played tennis and ran every day. I was obese only once in my life. I had not realized that I had gained a lot of weight.
Thankfully, a physician called me “obese.” Initially, I was angry at the doctor, however, I realized that he did me the biggest favor of my life. I changed my diet and eating habits, and for the past 20 years, kept my weight between 135-140 pounds and my BMI at 23 consistently – until the pandemic stress caused me to fall into the same bad eating habits that have caused many others to gain the COVID-15.
I was surprised to see that when I weighed myself, and I had gained 12 pounds! I immediately modified my diet and increased my physical activity. I have now lost the extra 12 pounds and will offer suggestions that may help you and your patients exceed your prepandemic physical condition.
Possible solutions
1. Keep a food journal. Write down what you eat, the amount of food you eat, the time you are eating, and your mood at that moment. Keeping a small notebook to record what and when you eat is important because upon review, it will make you face reality and be accountable for what you put in your mouth. Until you review your journal, you may have underestimated the amount, as well as the kinds, of food and drinks you actually consume. A food journal can show your areas of struggle and unhealthy eating habits and help you make necessary changes in your habits and diet to eventually lose weight. You will be less likely to eat junk food or have an extra serving of food. If you do not want to use paper and pencil, you can download an app on your phone, such as myplate tracker to keep track of your food and calorie intake. Do your journaling immediately after you eat and include snacks; do not wait until night time to record your food and journal. Include your mood or how you felt during your meal or snack (for example, were you bored, sad, or anxious) since this information will indicate why you may be overeating.
2. Develop healthful eating habits. Eat a maximum of three meals and three snacks per day but eat only when you are hungry (that is, when your stomach growls or you feel light headed). Limiting yourself to eating only when you are hungry will help eliminate emotional eating to fill a loss in your life or to deal with feelings of stress, anxiety, sadness, or isolation, which have been exacerbated by the pandemic. Buy eat only healthful foods and not items with empty calories, such as chips, cake, and items with sugar. When you are eating, devote yourself to that activity only, eat slowly, and savor each bite. Do not watch television during your meal time.
3. Record the amount and type of exercise you engage in each day and determine the number of calories burned. Walk, run, or bicycle outside, or exercise inside with stretching, weights, or an exercycle. You may use a website, such as diet tool on WebMD.com to calculate daily calories burned. To lose weight, calories burned during a day must exceed caloric intake. You may want to invest in a Fitbit or an Apple Watch and use the health section to determine your caloric intake versus output. Analyzing your caloric data will provide a concrete measure of your progress.
4. Do not overconsume calories or underconsume protein. Protein plays a key role in the creation and maintenance of every cell of your body, and because the body does not store protein, it is important to consume it every day. To meet basic protein requirements, the DRI (Dietary Reference Intake) recommends 0.36 grams of protein per pound (0.8 grams per kg) of body weight. This amounts to: 56 grams per day for the average sedentary man, and 46 grams per day for the average sedentary woman. There is also an app entitled the Protein Tracker that can simplify your calculations.
5. Drink water. It is important to be hydrated to regulate body temperature, keep joints lubricated, prevent infections, deliver nutrients to cells, and keep organs functioning properly. Being well hydrated also improves sleep, cognition, and mood. Your daily water intake by ounce should be equal to your weight in pounds multiplied by two-thirds (or 67%) to determine the amount of water to drink daily. For example, if you weigh 175 pounds, you would multiply 175 by two-thirds and learn that you should be drinking about 117 ounces of water every day. You can also meet some of your daily water requirements by consuming fruits and vegetables, such as tomatoes, watermelon, lettuce, etc.
Also, drink 2 cups (16 oz.) of water before every meal: Often when you feel hungry, it is because your body simply needs water. Science has proven that drinking 2 cups of water before every meal helps you to eat less during meal time and lose weight. If you do this three times daily – at breakfast, lunch, and dinner – you have already consumed 48 ounces of water.
6. Keep track of your progress. In addition to keeping and analyzing your food journal, weigh yourself once or twice a week. Do not weigh yourself every day; you will not see any results on a day-to-day basis, but once a week gives your body time to regulate and show progress. Always calibrate/zero your scale before each use, and weigh yourself at the same time of the day (preferably after you first wake up in the morning) while wearing the same type of clothing. Keep a record of your weight in your journal to track your progress. Do not panic if the scale indicates you gained 1, 2, or 3 pounds, your weight can fluctuate because of glycogen storage, sodium retention, human bias, reporting or recall errors, and home scales can have a plus or minus 3 pound margin of error. Look at your weight trend over time. You may prefer buying a scale that indicates both weight and body mass index.
7. Celebrate and reward yourself with nonfood items. A healthful fitness and diet regime requires energy and dedication, so if you are able to follow a healthful routine, reward yourself with nonfood rewards for your good choices and new habits as an incentive to maintain your healthful behavior.
8. Don’t buy it if you can’t stop eating it. The biggest decision you make is when you decide what you are going to buy. Don’t lie to yourself in the store that you will only eat one at a time. Only buy what you can afford to binge eat if you can’t stop yourself from eating any particular type of food.
9. Have someone hide the food you can’t resist. You can’t eat what you can’t find. If you can’t avoid having irresistible food around, ask another adult to hide the food from you.
10. Learn what harm foods can cause in your body. Read about the effects of high blood sugar and high blood pressure can cause in your body. Find out which foods boost your immune system. Demonize the bad foods in your mind. Make up your mind before you go into the store that you are going to read food labels and find the best quality food with the lowest amount of sugar or saturated fat. Appreciate the flavor of vegetables and fruit.
11. Treat sugar as if it were an addictive drug. You can’t have just one. If you reduce your craving for sugar by slowly reducing your intake of sugar, you will find that you don’t crave sugar any more. This won’t be easy, but once it is done, you will be preventing many of the ravages that sugar takes on your body over time. But you can’t have one piece of pie because the craving will come back. At some point, it may be more likely that you find that piece of pie too sweet.
Here are a few other ideas: Buy a gift for yourself or new clothes, makeup, a plant or flowers, running shoes, exercise clothes, fitness tracker, water bottle, book, movie or network subscription. Improve your home décor. Or treat yourself to online lessons for painting, music, and so on. Or you might adopt a dog, donate food to a shelter or food bank; or organize and declutter your home since staying busy will give you a reason not to eat. In nice weather, enjoy the outdoors by going for a walk, run, bikeride or by gardening.
We are all worried about getting COVID-19. Preventing COVID-15 will go a long way toward boosting our immune systems to help protect us from the coronavirus.
Dr. Cohen is board-certified in psychiatry and has had a private practice in Philadelphia for more than 35 years. His areas of specialty include sports psychiatry, agoraphobia, depression, and substance abuse. In addition, Dr. Cohen is a former professor of psychiatry, family medicine, and otolaryngology at Thomas Jefferson University in Philadelphia. He has no conflicts of interest. Ms. Cohen holds an MBA from Temple University in Philadelphia with a focus on health care administration. Previously, Ms. Cohen was an associate administrator at Hahnemann University Hospital and an executive at the Health Services Council, both in Philadelphia. She currently writes biographical summaries of notable 18th- and 19th-century women. Ms. Cohen has no conflicts of interest.
If you have been faithfully following the COVID-19 stay-at-home restrictive orders, you may have become a victim of “COVID-15,” the additional, unexpected, unwanted 10- to 15-pound weight gain that is making your clothes not fit so well any more.
A change in routine; being home in comfy, stretchable clothing in front of the TV; and having unhealthy, processed foods ready to grab have set us up to lose the battle with COVID-15. We are set up to gain the weight because of excessive or unhealthful eating, taking an extra daily shot of alcohol, and being inactive, bored, depressed, anxious, and isolated from coworkers and family. Beware – weight gain can be “catching”; we tend to adopt the same poor eating habits and eat the same junk foods as those around us.
Since psychiatry can be a sedentary profession, I’ve (R.W.C.) kept myself very active and physically fit. Prior to the pandemic, I played tennis and ran every day. I was obese only once in my life. I had not realized that I had gained a lot of weight.
Thankfully, a physician called me “obese.” Initially, I was angry at the doctor, however, I realized that he did me the biggest favor of my life. I changed my diet and eating habits, and for the past 20 years, kept my weight between 135-140 pounds and my BMI at 23 consistently – until the pandemic stress caused me to fall into the same bad eating habits that have caused many others to gain the COVID-15.
I was surprised to see that when I weighed myself, and I had gained 12 pounds! I immediately modified my diet and increased my physical activity. I have now lost the extra 12 pounds and will offer suggestions that may help you and your patients exceed your prepandemic physical condition.
Possible solutions
1. Keep a food journal. Write down what you eat, the amount of food you eat, the time you are eating, and your mood at that moment. Keeping a small notebook to record what and when you eat is important because upon review, it will make you face reality and be accountable for what you put in your mouth. Until you review your journal, you may have underestimated the amount, as well as the kinds, of food and drinks you actually consume. A food journal can show your areas of struggle and unhealthy eating habits and help you make necessary changes in your habits and diet to eventually lose weight. You will be less likely to eat junk food or have an extra serving of food. If you do not want to use paper and pencil, you can download an app on your phone, such as myplate tracker to keep track of your food and calorie intake. Do your journaling immediately after you eat and include snacks; do not wait until night time to record your food and journal. Include your mood or how you felt during your meal or snack (for example, were you bored, sad, or anxious) since this information will indicate why you may be overeating.
2. Develop healthful eating habits. Eat a maximum of three meals and three snacks per day but eat only when you are hungry (that is, when your stomach growls or you feel light headed). Limiting yourself to eating only when you are hungry will help eliminate emotional eating to fill a loss in your life or to deal with feelings of stress, anxiety, sadness, or isolation, which have been exacerbated by the pandemic. Buy eat only healthful foods and not items with empty calories, such as chips, cake, and items with sugar. When you are eating, devote yourself to that activity only, eat slowly, and savor each bite. Do not watch television during your meal time.
3. Record the amount and type of exercise you engage in each day and determine the number of calories burned. Walk, run, or bicycle outside, or exercise inside with stretching, weights, or an exercycle. You may use a website, such as diet tool on WebMD.com to calculate daily calories burned. To lose weight, calories burned during a day must exceed caloric intake. You may want to invest in a Fitbit or an Apple Watch and use the health section to determine your caloric intake versus output. Analyzing your caloric data will provide a concrete measure of your progress.
4. Do not overconsume calories or underconsume protein. Protein plays a key role in the creation and maintenance of every cell of your body, and because the body does not store protein, it is important to consume it every day. To meet basic protein requirements, the DRI (Dietary Reference Intake) recommends 0.36 grams of protein per pound (0.8 grams per kg) of body weight. This amounts to: 56 grams per day for the average sedentary man, and 46 grams per day for the average sedentary woman. There is also an app entitled the Protein Tracker that can simplify your calculations.
5. Drink water. It is important to be hydrated to regulate body temperature, keep joints lubricated, prevent infections, deliver nutrients to cells, and keep organs functioning properly. Being well hydrated also improves sleep, cognition, and mood. Your daily water intake by ounce should be equal to your weight in pounds multiplied by two-thirds (or 67%) to determine the amount of water to drink daily. For example, if you weigh 175 pounds, you would multiply 175 by two-thirds and learn that you should be drinking about 117 ounces of water every day. You can also meet some of your daily water requirements by consuming fruits and vegetables, such as tomatoes, watermelon, lettuce, etc.
Also, drink 2 cups (16 oz.) of water before every meal: Often when you feel hungry, it is because your body simply needs water. Science has proven that drinking 2 cups of water before every meal helps you to eat less during meal time and lose weight. If you do this three times daily – at breakfast, lunch, and dinner – you have already consumed 48 ounces of water.
6. Keep track of your progress. In addition to keeping and analyzing your food journal, weigh yourself once or twice a week. Do not weigh yourself every day; you will not see any results on a day-to-day basis, but once a week gives your body time to regulate and show progress. Always calibrate/zero your scale before each use, and weigh yourself at the same time of the day (preferably after you first wake up in the morning) while wearing the same type of clothing. Keep a record of your weight in your journal to track your progress. Do not panic if the scale indicates you gained 1, 2, or 3 pounds, your weight can fluctuate because of glycogen storage, sodium retention, human bias, reporting or recall errors, and home scales can have a plus or minus 3 pound margin of error. Look at your weight trend over time. You may prefer buying a scale that indicates both weight and body mass index.
7. Celebrate and reward yourself with nonfood items. A healthful fitness and diet regime requires energy and dedication, so if you are able to follow a healthful routine, reward yourself with nonfood rewards for your good choices and new habits as an incentive to maintain your healthful behavior.
8. Don’t buy it if you can’t stop eating it. The biggest decision you make is when you decide what you are going to buy. Don’t lie to yourself in the store that you will only eat one at a time. Only buy what you can afford to binge eat if you can’t stop yourself from eating any particular type of food.
9. Have someone hide the food you can’t resist. You can’t eat what you can’t find. If you can’t avoid having irresistible food around, ask another adult to hide the food from you.
10. Learn what harm foods can cause in your body. Read about the effects of high blood sugar and high blood pressure can cause in your body. Find out which foods boost your immune system. Demonize the bad foods in your mind. Make up your mind before you go into the store that you are going to read food labels and find the best quality food with the lowest amount of sugar or saturated fat. Appreciate the flavor of vegetables and fruit.
11. Treat sugar as if it were an addictive drug. You can’t have just one. If you reduce your craving for sugar by slowly reducing your intake of sugar, you will find that you don’t crave sugar any more. This won’t be easy, but once it is done, you will be preventing many of the ravages that sugar takes on your body over time. But you can’t have one piece of pie because the craving will come back. At some point, it may be more likely that you find that piece of pie too sweet.
Here are a few other ideas: Buy a gift for yourself or new clothes, makeup, a plant or flowers, running shoes, exercise clothes, fitness tracker, water bottle, book, movie or network subscription. Improve your home décor. Or treat yourself to online lessons for painting, music, and so on. Or you might adopt a dog, donate food to a shelter or food bank; or organize and declutter your home since staying busy will give you a reason not to eat. In nice weather, enjoy the outdoors by going for a walk, run, bikeride or by gardening.
We are all worried about getting COVID-19. Preventing COVID-15 will go a long way toward boosting our immune systems to help protect us from the coronavirus.
Dr. Cohen is board-certified in psychiatry and has had a private practice in Philadelphia for more than 35 years. His areas of specialty include sports psychiatry, agoraphobia, depression, and substance abuse. In addition, Dr. Cohen is a former professor of psychiatry, family medicine, and otolaryngology at Thomas Jefferson University in Philadelphia. He has no conflicts of interest. Ms. Cohen holds an MBA from Temple University in Philadelphia with a focus on health care administration. Previously, Ms. Cohen was an associate administrator at Hahnemann University Hospital and an executive at the Health Services Council, both in Philadelphia. She currently writes biographical summaries of notable 18th- and 19th-century women. Ms. Cohen has no conflicts of interest.
Diabetic retinopathy may predict greater risk of COVID-19 severity
Risk of intubation for COVID-19 in very sick hospitalized patients was increased over fivefold in those with diabetic retinopathy, compared with those without, in a small single-center study from the United Kingdom.
Importantly, the risk of intubation was independent of conventional risk factors for poor COVID-19 outcomes.
“People with preexisting diabetes-related vascular damage, such as retinopathy, might be predisposed to a more severe form of COVID-19 requiring ventilation in the intensive therapy unit,” said lead investigator Janaka Karalliedde, MBBS, PhD.
Dr. Karalliedde and colleagues note that this is “the first description of diabetic retinopathy as a potential risk factor for poor COVID-19 outcomes.”
“For this reason, looking for the presence or history of retinopathy or other vascular complications of diabetes may help health care professionals identify patients at high risk of severe COVID-19,” added Dr. Karalliedde, of Guy’s and St Thomas’ NHS Foundation Trust, London.
The study was published online in Diabetes Research and Clinical Practice.
Preexisting diabetic retinopathy and COVID-19 outcomes
The prevalence of diabetic retinopathy is thought to be around 55% in people with type 1 diabetes and 30% in people with type 2 diabetes, on average.
Dr. Karalliedde is part of a research group at King’s College London that has been focused on how vascular disease may predispose to more severe COVID-19.
“COVID-19 affects the blood vessels all over the body,” he said, so they wondered whether having preexisting retinopathy “would predispose to a severe manifestation of COVID-19.”
The observational study included 187 patients with diabetes (179 patients with type 2 diabetes and 8 patients with type 1 diabetes) hospitalized with COVID-19 at Guy’s and St Thomas’ NHS Foundation Trust between March 12 and April 7 (the peak of the first wave of the pandemic in the United Kingdom).
“It was an ethnically diverse population who were very sick and provides a clinical observation of real life,” Dr. Karalliedde said.
Nearly half of patients were African Caribbean (44%), 39% were White, and 17% were of other ethnicities, including 8% who were Asian. The mean age of the cohort was 68 years (range, 22-97 years), and 60% were men.
Diabetic retinopathy was reported in 67 (36%) patients, of whom 80% had background retinopathy and 20% had more advanced retinopathy.
They then looked at whether the presence of retinopathy was associated with a more severe manifestation of COVID-19 as defined by the need for tracheal intubation.
Of the 187 patients, 26% were intubated and 45% of these patients had diabetic retinopathy.
The analysis showed those with diabetic retinopathy had an over-fivefold increased risk for intubation (odds ratio, 5.81; 95% confidence interval, 1.37-24.66).
Of the entire cohort, 32% of patients died, although no association was observed between retinopathy and mortality.
“A greater number of diabetes patients with COVID-19 ended up on the intensive therapy unit. Upon multivariate analysis, we found retinopathy was independently associated with ending up on the intensive therapy unit,” stressed Dr. Karalliedde.
However, they noted that, “due to the cross-sectional design of our study, we cannot prove causality [between retinopathy and intubation]. Further studies are required to understand the mechanisms that explain the associations between retinopathy and other indices of microangiopathy with severe COVID-19.”
A version of this article originally appeared on Medscape.com.
Risk of intubation for COVID-19 in very sick hospitalized patients was increased over fivefold in those with diabetic retinopathy, compared with those without, in a small single-center study from the United Kingdom.
Importantly, the risk of intubation was independent of conventional risk factors for poor COVID-19 outcomes.
“People with preexisting diabetes-related vascular damage, such as retinopathy, might be predisposed to a more severe form of COVID-19 requiring ventilation in the intensive therapy unit,” said lead investigator Janaka Karalliedde, MBBS, PhD.
Dr. Karalliedde and colleagues note that this is “the first description of diabetic retinopathy as a potential risk factor for poor COVID-19 outcomes.”
“For this reason, looking for the presence or history of retinopathy or other vascular complications of diabetes may help health care professionals identify patients at high risk of severe COVID-19,” added Dr. Karalliedde, of Guy’s and St Thomas’ NHS Foundation Trust, London.
The study was published online in Diabetes Research and Clinical Practice.
Preexisting diabetic retinopathy and COVID-19 outcomes
The prevalence of diabetic retinopathy is thought to be around 55% in people with type 1 diabetes and 30% in people with type 2 diabetes, on average.
Dr. Karalliedde is part of a research group at King’s College London that has been focused on how vascular disease may predispose to more severe COVID-19.
“COVID-19 affects the blood vessels all over the body,” he said, so they wondered whether having preexisting retinopathy “would predispose to a severe manifestation of COVID-19.”
The observational study included 187 patients with diabetes (179 patients with type 2 diabetes and 8 patients with type 1 diabetes) hospitalized with COVID-19 at Guy’s and St Thomas’ NHS Foundation Trust between March 12 and April 7 (the peak of the first wave of the pandemic in the United Kingdom).
“It was an ethnically diverse population who were very sick and provides a clinical observation of real life,” Dr. Karalliedde said.
Nearly half of patients were African Caribbean (44%), 39% were White, and 17% were of other ethnicities, including 8% who were Asian. The mean age of the cohort was 68 years (range, 22-97 years), and 60% were men.
Diabetic retinopathy was reported in 67 (36%) patients, of whom 80% had background retinopathy and 20% had more advanced retinopathy.
They then looked at whether the presence of retinopathy was associated with a more severe manifestation of COVID-19 as defined by the need for tracheal intubation.
Of the 187 patients, 26% were intubated and 45% of these patients had diabetic retinopathy.
The analysis showed those with diabetic retinopathy had an over-fivefold increased risk for intubation (odds ratio, 5.81; 95% confidence interval, 1.37-24.66).
Of the entire cohort, 32% of patients died, although no association was observed between retinopathy and mortality.
“A greater number of diabetes patients with COVID-19 ended up on the intensive therapy unit. Upon multivariate analysis, we found retinopathy was independently associated with ending up on the intensive therapy unit,” stressed Dr. Karalliedde.
However, they noted that, “due to the cross-sectional design of our study, we cannot prove causality [between retinopathy and intubation]. Further studies are required to understand the mechanisms that explain the associations between retinopathy and other indices of microangiopathy with severe COVID-19.”
A version of this article originally appeared on Medscape.com.
Risk of intubation for COVID-19 in very sick hospitalized patients was increased over fivefold in those with diabetic retinopathy, compared with those without, in a small single-center study from the United Kingdom.
Importantly, the risk of intubation was independent of conventional risk factors for poor COVID-19 outcomes.
“People with preexisting diabetes-related vascular damage, such as retinopathy, might be predisposed to a more severe form of COVID-19 requiring ventilation in the intensive therapy unit,” said lead investigator Janaka Karalliedde, MBBS, PhD.
Dr. Karalliedde and colleagues note that this is “the first description of diabetic retinopathy as a potential risk factor for poor COVID-19 outcomes.”
“For this reason, looking for the presence or history of retinopathy or other vascular complications of diabetes may help health care professionals identify patients at high risk of severe COVID-19,” added Dr. Karalliedde, of Guy’s and St Thomas’ NHS Foundation Trust, London.
The study was published online in Diabetes Research and Clinical Practice.
Preexisting diabetic retinopathy and COVID-19 outcomes
The prevalence of diabetic retinopathy is thought to be around 55% in people with type 1 diabetes and 30% in people with type 2 diabetes, on average.
Dr. Karalliedde is part of a research group at King’s College London that has been focused on how vascular disease may predispose to more severe COVID-19.
“COVID-19 affects the blood vessels all over the body,” he said, so they wondered whether having preexisting retinopathy “would predispose to a severe manifestation of COVID-19.”
The observational study included 187 patients with diabetes (179 patients with type 2 diabetes and 8 patients with type 1 diabetes) hospitalized with COVID-19 at Guy’s and St Thomas’ NHS Foundation Trust between March 12 and April 7 (the peak of the first wave of the pandemic in the United Kingdom).
“It was an ethnically diverse population who were very sick and provides a clinical observation of real life,” Dr. Karalliedde said.
Nearly half of patients were African Caribbean (44%), 39% were White, and 17% were of other ethnicities, including 8% who were Asian. The mean age of the cohort was 68 years (range, 22-97 years), and 60% were men.
Diabetic retinopathy was reported in 67 (36%) patients, of whom 80% had background retinopathy and 20% had more advanced retinopathy.
They then looked at whether the presence of retinopathy was associated with a more severe manifestation of COVID-19 as defined by the need for tracheal intubation.
Of the 187 patients, 26% were intubated and 45% of these patients had diabetic retinopathy.
The analysis showed those with diabetic retinopathy had an over-fivefold increased risk for intubation (odds ratio, 5.81; 95% confidence interval, 1.37-24.66).
Of the entire cohort, 32% of patients died, although no association was observed between retinopathy and mortality.
“A greater number of diabetes patients with COVID-19 ended up on the intensive therapy unit. Upon multivariate analysis, we found retinopathy was independently associated with ending up on the intensive therapy unit,” stressed Dr. Karalliedde.
However, they noted that, “due to the cross-sectional design of our study, we cannot prove causality [between retinopathy and intubation]. Further studies are required to understand the mechanisms that explain the associations between retinopathy and other indices of microangiopathy with severe COVID-19.”
A version of this article originally appeared on Medscape.com.