User login
IDSA panel updates guidelines on COVID molecular diagnostic tests
Saliva spit tests stack up well against the gold standard for molecular COVID-19 tests – the back-of-the-nose deep swab – without the discomfort and induced coughing or sneezing of the test taker, updated guidelines indicate.
In a press briefing on Jan. 6, the Infectious Diseases Society of America explained the findings of an expert panel that reviewed the literature since the IDSA released its first guidelines in May.
The panel found that saliva tests were especially effective if the test included instructions to cough or clear the throat before spitting into the tube, said panel chair Kimberly E. Hanson, MD, MHS, of University of Utah Health, Salt Lake City.
Throat swab alone less effective
Using a throat swab alone was less effective and missed more cases than the other methods, she said.
The IDSA has updated its recommendation: A saliva test or swabs from either the middle or front of the nose front are preferred to a throat swab alone.
A combination of saliva and swabs from the front and middle of the nose and throat together “looked pretty much equivalent” to the gold-standard deep swab, the panel found.
She acknowledged, however, that multiple swabs exacerbate already challenging supply issues.
Saliva samples do come with challenges, Dr. Hanson noted. A laboratory must validate that its systems can handle the stickier material. And asking a patient to cough necessitates more personal protective equipment for the health care professional.
Each center will have to tailor the specimen type it chooses, based on what resources it has available and the setting – whether in a hospital or a drive-through operation, for instance, she said.
Rapid testing vs. standard
Panel member Angela M. Caliendo, MD, PhD, of Brown University, Providence, R.I., said the panel preferred rapid polymerase chain reaction tests and standard, laboratory-based PCR tests over a rapid isothermal test.
The panel defined rapid tests as those for which results are available within an hour after a test provider has the specimen in hand. They excluded home tests for this category.
The only rapid isothermal test that had enough data on which to issue a recommendation was the ID NOW test (Abbott Labs), she noted.
Rapid PCR tests performed just as well as the standard laboratory-based tests, she said, with a high sensitivity of “97% on average and a very high specificity.”
But the rapid isothermal test had an average sensitivity of only about 80%, compared with the lab-based PCR test, Dr. Caliendo said, yielding a substantial number of false-negative results.
Testing centers will have to weigh the considerable advantages of having results in 15 minutes with a rapid isothermal test and being able to educate positive patients about immediate isolation against the potential for false negatives, which could send positive patients home thinking they don’t have the virus – and thus potentially spreading the disease.
And if a clinician gets a negative result with the rapid isothermal test, but has a strong suspicion the person has COVID or lives in an area with high prevalence, a backup test with a rapid PCR or laboratory-based test should be administered.
“You will miss a certain percentage of people using this rapid isothermal test,” she said.
However, Dr. Caliendo said, if the only available option is the isothermal test, “you should definitely use it because it’s certainly better than not testing at all.”
On a positive note, she said, all the varieties of tests have high specificity, so “you’re not going to see a lot of false-positive results.”
The guidelines back in May didn’t make recommendations on rapid tests, she said, because there weren’t enough data in the literature.
Dr. Caliendo noted that most of the available data were for symptomatic patients, but there are some data that show the amount of virus in the respiratory tract is similar for people with and without symptoms. The panel, therefore, expects that the performance of the various assays would be similar whether or not a person had symptoms.
Testing the immunocompromised
Dr. Hanson said the original recommendation in May was to do molecular testing for asymptomatic people who were awaiting a transplant or were waiting to start immunosuppressive therapy for cancer or an autoimmune disease. Now the current guidelines “make no recommendation for or against screening” in those cases.
Dr. Hanson added that the panel feels that patients awaiting bone marrow and solid organ transplants should have the testing because of the high risks that will result if patients have contracted the virus.
But for those with cancer or an autoimmune disease, the panel decided to leave it up to each physician to assess individual risk and determine whether the patient should be tested.
Home testing
The IDSA guidelines didn’t weigh in on home testing because the products are so new and studies so far have included fewer than 200 patients. But Dr. Caliendo said they clearly perform better earlier in the disease phase – the first 5-7 days – when the amount of the virus is higher.
Dr. Hanson and Dr. Caliendo also fielded a question about what the new virus variant, first discovered in the United Kingdom and now spreading to other countries (including the United States) means for diagnostic testing.
“So far we think with the majority of tests that are [emergency use] authorized, it doesn’t look like this new variant should really affect test performance,” Dr. Hanson said.
The variant has differences in the spike gene, and many of the current tests detect and identify SARS-CoV-2 without the spike gene so they wouldn’t be affected, she added.
Dr. Caliendo agreed: “I think the vast majority of our tests should be in good shape.”
Dr. Hanson and Dr. Caliendo disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Saliva spit tests stack up well against the gold standard for molecular COVID-19 tests – the back-of-the-nose deep swab – without the discomfort and induced coughing or sneezing of the test taker, updated guidelines indicate.
In a press briefing on Jan. 6, the Infectious Diseases Society of America explained the findings of an expert panel that reviewed the literature since the IDSA released its first guidelines in May.
The panel found that saliva tests were especially effective if the test included instructions to cough or clear the throat before spitting into the tube, said panel chair Kimberly E. Hanson, MD, MHS, of University of Utah Health, Salt Lake City.
Throat swab alone less effective
Using a throat swab alone was less effective and missed more cases than the other methods, she said.
The IDSA has updated its recommendation: A saliva test or swabs from either the middle or front of the nose front are preferred to a throat swab alone.
A combination of saliva and swabs from the front and middle of the nose and throat together “looked pretty much equivalent” to the gold-standard deep swab, the panel found.
She acknowledged, however, that multiple swabs exacerbate already challenging supply issues.
Saliva samples do come with challenges, Dr. Hanson noted. A laboratory must validate that its systems can handle the stickier material. And asking a patient to cough necessitates more personal protective equipment for the health care professional.
Each center will have to tailor the specimen type it chooses, based on what resources it has available and the setting – whether in a hospital or a drive-through operation, for instance, she said.
Rapid testing vs. standard
Panel member Angela M. Caliendo, MD, PhD, of Brown University, Providence, R.I., said the panel preferred rapid polymerase chain reaction tests and standard, laboratory-based PCR tests over a rapid isothermal test.
The panel defined rapid tests as those for which results are available within an hour after a test provider has the specimen in hand. They excluded home tests for this category.
The only rapid isothermal test that had enough data on which to issue a recommendation was the ID NOW test (Abbott Labs), she noted.
Rapid PCR tests performed just as well as the standard laboratory-based tests, she said, with a high sensitivity of “97% on average and a very high specificity.”
But the rapid isothermal test had an average sensitivity of only about 80%, compared with the lab-based PCR test, Dr. Caliendo said, yielding a substantial number of false-negative results.
Testing centers will have to weigh the considerable advantages of having results in 15 minutes with a rapid isothermal test and being able to educate positive patients about immediate isolation against the potential for false negatives, which could send positive patients home thinking they don’t have the virus – and thus potentially spreading the disease.
And if a clinician gets a negative result with the rapid isothermal test, but has a strong suspicion the person has COVID or lives in an area with high prevalence, a backup test with a rapid PCR or laboratory-based test should be administered.
“You will miss a certain percentage of people using this rapid isothermal test,” she said.
However, Dr. Caliendo said, if the only available option is the isothermal test, “you should definitely use it because it’s certainly better than not testing at all.”
On a positive note, she said, all the varieties of tests have high specificity, so “you’re not going to see a lot of false-positive results.”
The guidelines back in May didn’t make recommendations on rapid tests, she said, because there weren’t enough data in the literature.
Dr. Caliendo noted that most of the available data were for symptomatic patients, but there are some data that show the amount of virus in the respiratory tract is similar for people with and without symptoms. The panel, therefore, expects that the performance of the various assays would be similar whether or not a person had symptoms.
Testing the immunocompromised
Dr. Hanson said the original recommendation in May was to do molecular testing for asymptomatic people who were awaiting a transplant or were waiting to start immunosuppressive therapy for cancer or an autoimmune disease. Now the current guidelines “make no recommendation for or against screening” in those cases.
Dr. Hanson added that the panel feels that patients awaiting bone marrow and solid organ transplants should have the testing because of the high risks that will result if patients have contracted the virus.
But for those with cancer or an autoimmune disease, the panel decided to leave it up to each physician to assess individual risk and determine whether the patient should be tested.
Home testing
The IDSA guidelines didn’t weigh in on home testing because the products are so new and studies so far have included fewer than 200 patients. But Dr. Caliendo said they clearly perform better earlier in the disease phase – the first 5-7 days – when the amount of the virus is higher.
Dr. Hanson and Dr. Caliendo also fielded a question about what the new virus variant, first discovered in the United Kingdom and now spreading to other countries (including the United States) means for diagnostic testing.
“So far we think with the majority of tests that are [emergency use] authorized, it doesn’t look like this new variant should really affect test performance,” Dr. Hanson said.
The variant has differences in the spike gene, and many of the current tests detect and identify SARS-CoV-2 without the spike gene so they wouldn’t be affected, she added.
Dr. Caliendo agreed: “I think the vast majority of our tests should be in good shape.”
Dr. Hanson and Dr. Caliendo disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Saliva spit tests stack up well against the gold standard for molecular COVID-19 tests – the back-of-the-nose deep swab – without the discomfort and induced coughing or sneezing of the test taker, updated guidelines indicate.
In a press briefing on Jan. 6, the Infectious Diseases Society of America explained the findings of an expert panel that reviewed the literature since the IDSA released its first guidelines in May.
The panel found that saliva tests were especially effective if the test included instructions to cough or clear the throat before spitting into the tube, said panel chair Kimberly E. Hanson, MD, MHS, of University of Utah Health, Salt Lake City.
Throat swab alone less effective
Using a throat swab alone was less effective and missed more cases than the other methods, she said.
The IDSA has updated its recommendation: A saliva test or swabs from either the middle or front of the nose front are preferred to a throat swab alone.
A combination of saliva and swabs from the front and middle of the nose and throat together “looked pretty much equivalent” to the gold-standard deep swab, the panel found.
She acknowledged, however, that multiple swabs exacerbate already challenging supply issues.
Saliva samples do come with challenges, Dr. Hanson noted. A laboratory must validate that its systems can handle the stickier material. And asking a patient to cough necessitates more personal protective equipment for the health care professional.
Each center will have to tailor the specimen type it chooses, based on what resources it has available and the setting – whether in a hospital or a drive-through operation, for instance, she said.
Rapid testing vs. standard
Panel member Angela M. Caliendo, MD, PhD, of Brown University, Providence, R.I., said the panel preferred rapid polymerase chain reaction tests and standard, laboratory-based PCR tests over a rapid isothermal test.
The panel defined rapid tests as those for which results are available within an hour after a test provider has the specimen in hand. They excluded home tests for this category.
The only rapid isothermal test that had enough data on which to issue a recommendation was the ID NOW test (Abbott Labs), she noted.
Rapid PCR tests performed just as well as the standard laboratory-based tests, she said, with a high sensitivity of “97% on average and a very high specificity.”
But the rapid isothermal test had an average sensitivity of only about 80%, compared with the lab-based PCR test, Dr. Caliendo said, yielding a substantial number of false-negative results.
Testing centers will have to weigh the considerable advantages of having results in 15 minutes with a rapid isothermal test and being able to educate positive patients about immediate isolation against the potential for false negatives, which could send positive patients home thinking they don’t have the virus – and thus potentially spreading the disease.
And if a clinician gets a negative result with the rapid isothermal test, but has a strong suspicion the person has COVID or lives in an area with high prevalence, a backup test with a rapid PCR or laboratory-based test should be administered.
“You will miss a certain percentage of people using this rapid isothermal test,” she said.
However, Dr. Caliendo said, if the only available option is the isothermal test, “you should definitely use it because it’s certainly better than not testing at all.”
On a positive note, she said, all the varieties of tests have high specificity, so “you’re not going to see a lot of false-positive results.”
The guidelines back in May didn’t make recommendations on rapid tests, she said, because there weren’t enough data in the literature.
Dr. Caliendo noted that most of the available data were for symptomatic patients, but there are some data that show the amount of virus in the respiratory tract is similar for people with and without symptoms. The panel, therefore, expects that the performance of the various assays would be similar whether or not a person had symptoms.
Testing the immunocompromised
Dr. Hanson said the original recommendation in May was to do molecular testing for asymptomatic people who were awaiting a transplant or were waiting to start immunosuppressive therapy for cancer or an autoimmune disease. Now the current guidelines “make no recommendation for or against screening” in those cases.
Dr. Hanson added that the panel feels that patients awaiting bone marrow and solid organ transplants should have the testing because of the high risks that will result if patients have contracted the virus.
But for those with cancer or an autoimmune disease, the panel decided to leave it up to each physician to assess individual risk and determine whether the patient should be tested.
Home testing
The IDSA guidelines didn’t weigh in on home testing because the products are so new and studies so far have included fewer than 200 patients. But Dr. Caliendo said they clearly perform better earlier in the disease phase – the first 5-7 days – when the amount of the virus is higher.
Dr. Hanson and Dr. Caliendo also fielded a question about what the new virus variant, first discovered in the United Kingdom and now spreading to other countries (including the United States) means for diagnostic testing.
“So far we think with the majority of tests that are [emergency use] authorized, it doesn’t look like this new variant should really affect test performance,” Dr. Hanson said.
The variant has differences in the spike gene, and many of the current tests detect and identify SARS-CoV-2 without the spike gene so they wouldn’t be affected, she added.
Dr. Caliendo agreed: “I think the vast majority of our tests should be in good shape.”
Dr. Hanson and Dr. Caliendo disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Early use of high-titer plasma may prevent severe COVID-19
Administering convalescent plasma that has high levels of antibodies against SARS-CoV-2 within the first 3 days of symptoms was associated with significantly lower chances of progression to severe COVID-19, new evidence demonstrates.
In a trial of 160 older adults with COVID-19, half of whom were randomly assigned to receive plasma and half to receive placebo infusion, treatment with high-titer plasma lowered the relative risk for severe disease by 48% in an intent-to-treat analysis.
“We now have evidence, in the context of a small but well-designed study, that convalescent plasma with high titers of antibody against SARS-CoV-2 administered in the first 3 days of mild symptoms to infected elderly reduces progression of illness and the rate of severe presentations,” senior author Fernando Polack, MD, said in an interview.
“Not any plasma, not any time,” added Dr. Polack, an infectious disease specialist and scientific director at Fundacion INFANT and professor of pediatrics at the University of Buenos Aires. The key, he said, is to select plasma in the upper 28th percentile of IgG antibody concentrations and to administer therapy prior to disease progression.
The study was published online Jan. 6 in The New England Journal of Medicine.
“It’s a very good study and approaches a different population from the PlasmAr study,” Ventura Simonovich, MD, chief of the clinical pharmacology section, Medical Clinic Service, Hospital Italiano de Buenos Aires, said in an interview. “This is the first published randomized controlled trial that shows real benefit in this [older adult] population, the most vulnerable in this disease,” he said.
Dr. Simonovich, who was not affiliated with the current study, was lead author of the PlasmAr trial, which was published in The New England Journal of Medicine Nov. 24, 2020. In that trial, the researchers evaluated adults aged 18 years and older and found no significant benefit with convalescent plasma treatment over placebo for patients with COVID-19 and severe pneumonia.
“We know antibodies work best when given early and in high dose. This is one of the rare reports that validates it in the outpatient setting,” David Sullivan, MD, professor of molecular biology and immunology at Johns Hopkins Bloomberg School of Public Health, Baltimore, said in an interview when asked to comment.
Dr. Sullivan pointed out that most previous studies on convalescent plasma focused on patients with COVID-19 who had severe cases late in the disease course.
Regarding the current study, he said, “The striking thing is treating people within 3 days of illness.”
A more cautious interpretation may be warranted, one expert said. “The study demonstrates the benefit of early intervention. There was a dose-dependent effect, with higher titers providing a greater benefit,” Manoj Menon, MD, MPH, a hematologist and oncologist at the University of Washington, Seattle, said in an interview.
“Taken together, the findings have biologic plausibility and produce more data on the role of convalescent plasma to a relevant age cohort,” he added.
However, Dr. Menon said: “Given the limited sample size, I do not think this study, although well conducted, definitively addresses the role of convalescent plasma for COVID-19. But it does merit additional study.”
A search for clear answers
Treatments that target the early stages of COVID-19 “remain elusive. Few strategies provide benefit, several have failed, and others are being evaluated,” the researchers noted. “In hospitalized patients with COVID-19, the infusion of convalescent plasma against SARS-CoV-2 late in the course of illness has not shown clear benefits and, consequently, the most appropriate antibody concentrations for effective treatment are unclear.”
To learn more, Dr. Polack and colleagues included patients with PCR-confirmed COVID-19 who were aged 75 years or older, regardless of comorbidities. They also included patients aged 65-74 years who had at least one underlying condition. Participants were enrolled at clinical sites or geriatric units in Argentina. The mean age was 77 years, and 62% were women.
In an intent-to-treat analysis, the primary outcome – severe respiratory disease – occurred in 16% of the plasma recipients, vs. 31% of the group that received placebo. The relative risk was 0.52 (95% confidence interval, 0.29-0.94; P = .03).
The number needed to treat to avoid a severe respiratory disease episode was 7 (95% CI, 4-50).
Life-threatening respiratory disease, a secondary outcome, occurred in four people in the plasma group, compared with 10 in the placebo group. Two patients in the treatment group and four patients in the placebo group died.
The researchers also ran a modified intent-to-treat analysis that excluded six participants who experienced severe respiratory disease prior to receiving plasma or placebo. In this analysis, efficacy of plasma therapy increased to 60%.
“Again, this finding suggests that early intervention is critical for efficacy,” the investigators noted.
The investigators, who are based in Argentina, defined their primary endpoint as a respiratory rate of 30 or more breaths per minute and/or an oxygen saturation of less than 93% while breathing ambient air.
Dr. Sullivan pointed out that this is equivalent to the threshold commonly used for hospitalizing people with COVID-19 in the United States. “So it’s equivalent to avoiding hospitalizations. The take-home is high-titer plasma prevents respiratory distress, which equals hospitalization for us.”
Dr. Sullivan is conducting similar research in the United States regarding the use of plasma for treatment or prevention. He and colleagues are evaluating adults aged 18-90 years, “not just the ones at highest risk for going to the hospital,” he said. Enrollment is ongoing.
An inexpensive therapy with global potential?
“Although our trial lacked the statistical power to discern long-term outcomes, the convalescent plasma group appeared to have better outcomes than the placebo group with respect to all secondary endpoints,” the researchers wrote. “Our findings underscore the need to return to the classic approach of treating acute viral infections early, and they define IgG targets that facilitate donor selection.”
Dr. Polack said, “This is an inexpensive solution to mitigate the burden of severe illness in the population most vulnerable to the virus: the elderly. And it has the attraction of being applicable not only in industrialized countries but in many areas of the developing world.”
Convalescent plasma “is a potentially inexpensive alternative to monoclonal antibodies,” the researchers added. Furthermore, “early infusions of convalescent plasma can provide a bridge to recovery for at-risk patients until vaccines become widely available.”
Dr. Polack said the study findings did not surprise him. “We always thought that, as it has been the case in the past with many therapeutic strategies against respiratory and other viral infections, the earlier you treat, the better.
“We just hoped that within 72 hours of symptoms we would be treating early enough – remember that there is a 4- to 5-day incubation period that the virus leverages before the first symptom – and with enough antibody,” he added.
“We are glad it worked,” he said.
The study was supported by the Bill and Melinda Gates Foundation and by the Fundación INFANT Pandemic Fund. Dr. Polack, Dr. Simonovich, and Dr. Sullivan have disclosed various financial relationships industry.
A version of this article first appeared on Medscape.com.
Administering convalescent plasma that has high levels of antibodies against SARS-CoV-2 within the first 3 days of symptoms was associated with significantly lower chances of progression to severe COVID-19, new evidence demonstrates.
In a trial of 160 older adults with COVID-19, half of whom were randomly assigned to receive plasma and half to receive placebo infusion, treatment with high-titer plasma lowered the relative risk for severe disease by 48% in an intent-to-treat analysis.
“We now have evidence, in the context of a small but well-designed study, that convalescent plasma with high titers of antibody against SARS-CoV-2 administered in the first 3 days of mild symptoms to infected elderly reduces progression of illness and the rate of severe presentations,” senior author Fernando Polack, MD, said in an interview.
“Not any plasma, not any time,” added Dr. Polack, an infectious disease specialist and scientific director at Fundacion INFANT and professor of pediatrics at the University of Buenos Aires. The key, he said, is to select plasma in the upper 28th percentile of IgG antibody concentrations and to administer therapy prior to disease progression.
The study was published online Jan. 6 in The New England Journal of Medicine.
“It’s a very good study and approaches a different population from the PlasmAr study,” Ventura Simonovich, MD, chief of the clinical pharmacology section, Medical Clinic Service, Hospital Italiano de Buenos Aires, said in an interview. “This is the first published randomized controlled trial that shows real benefit in this [older adult] population, the most vulnerable in this disease,” he said.
Dr. Simonovich, who was not affiliated with the current study, was lead author of the PlasmAr trial, which was published in The New England Journal of Medicine Nov. 24, 2020. In that trial, the researchers evaluated adults aged 18 years and older and found no significant benefit with convalescent plasma treatment over placebo for patients with COVID-19 and severe pneumonia.
“We know antibodies work best when given early and in high dose. This is one of the rare reports that validates it in the outpatient setting,” David Sullivan, MD, professor of molecular biology and immunology at Johns Hopkins Bloomberg School of Public Health, Baltimore, said in an interview when asked to comment.
Dr. Sullivan pointed out that most previous studies on convalescent plasma focused on patients with COVID-19 who had severe cases late in the disease course.
Regarding the current study, he said, “The striking thing is treating people within 3 days of illness.”
A more cautious interpretation may be warranted, one expert said. “The study demonstrates the benefit of early intervention. There was a dose-dependent effect, with higher titers providing a greater benefit,” Manoj Menon, MD, MPH, a hematologist and oncologist at the University of Washington, Seattle, said in an interview.
“Taken together, the findings have biologic plausibility and produce more data on the role of convalescent plasma to a relevant age cohort,” he added.
However, Dr. Menon said: “Given the limited sample size, I do not think this study, although well conducted, definitively addresses the role of convalescent plasma for COVID-19. But it does merit additional study.”
A search for clear answers
Treatments that target the early stages of COVID-19 “remain elusive. Few strategies provide benefit, several have failed, and others are being evaluated,” the researchers noted. “In hospitalized patients with COVID-19, the infusion of convalescent plasma against SARS-CoV-2 late in the course of illness has not shown clear benefits and, consequently, the most appropriate antibody concentrations for effective treatment are unclear.”
To learn more, Dr. Polack and colleagues included patients with PCR-confirmed COVID-19 who were aged 75 years or older, regardless of comorbidities. They also included patients aged 65-74 years who had at least one underlying condition. Participants were enrolled at clinical sites or geriatric units in Argentina. The mean age was 77 years, and 62% were women.
In an intent-to-treat analysis, the primary outcome – severe respiratory disease – occurred in 16% of the plasma recipients, vs. 31% of the group that received placebo. The relative risk was 0.52 (95% confidence interval, 0.29-0.94; P = .03).
The number needed to treat to avoid a severe respiratory disease episode was 7 (95% CI, 4-50).
Life-threatening respiratory disease, a secondary outcome, occurred in four people in the plasma group, compared with 10 in the placebo group. Two patients in the treatment group and four patients in the placebo group died.
The researchers also ran a modified intent-to-treat analysis that excluded six participants who experienced severe respiratory disease prior to receiving plasma or placebo. In this analysis, efficacy of plasma therapy increased to 60%.
“Again, this finding suggests that early intervention is critical for efficacy,” the investigators noted.
The investigators, who are based in Argentina, defined their primary endpoint as a respiratory rate of 30 or more breaths per minute and/or an oxygen saturation of less than 93% while breathing ambient air.
Dr. Sullivan pointed out that this is equivalent to the threshold commonly used for hospitalizing people with COVID-19 in the United States. “So it’s equivalent to avoiding hospitalizations. The take-home is high-titer plasma prevents respiratory distress, which equals hospitalization for us.”
Dr. Sullivan is conducting similar research in the United States regarding the use of plasma for treatment or prevention. He and colleagues are evaluating adults aged 18-90 years, “not just the ones at highest risk for going to the hospital,” he said. Enrollment is ongoing.
An inexpensive therapy with global potential?
“Although our trial lacked the statistical power to discern long-term outcomes, the convalescent plasma group appeared to have better outcomes than the placebo group with respect to all secondary endpoints,” the researchers wrote. “Our findings underscore the need to return to the classic approach of treating acute viral infections early, and they define IgG targets that facilitate donor selection.”
Dr. Polack said, “This is an inexpensive solution to mitigate the burden of severe illness in the population most vulnerable to the virus: the elderly. And it has the attraction of being applicable not only in industrialized countries but in many areas of the developing world.”
Convalescent plasma “is a potentially inexpensive alternative to monoclonal antibodies,” the researchers added. Furthermore, “early infusions of convalescent plasma can provide a bridge to recovery for at-risk patients until vaccines become widely available.”
Dr. Polack said the study findings did not surprise him. “We always thought that, as it has been the case in the past with many therapeutic strategies against respiratory and other viral infections, the earlier you treat, the better.
“We just hoped that within 72 hours of symptoms we would be treating early enough – remember that there is a 4- to 5-day incubation period that the virus leverages before the first symptom – and with enough antibody,” he added.
“We are glad it worked,” he said.
The study was supported by the Bill and Melinda Gates Foundation and by the Fundación INFANT Pandemic Fund. Dr. Polack, Dr. Simonovich, and Dr. Sullivan have disclosed various financial relationships industry.
A version of this article first appeared on Medscape.com.
Administering convalescent plasma that has high levels of antibodies against SARS-CoV-2 within the first 3 days of symptoms was associated with significantly lower chances of progression to severe COVID-19, new evidence demonstrates.
In a trial of 160 older adults with COVID-19, half of whom were randomly assigned to receive plasma and half to receive placebo infusion, treatment with high-titer plasma lowered the relative risk for severe disease by 48% in an intent-to-treat analysis.
“We now have evidence, in the context of a small but well-designed study, that convalescent plasma with high titers of antibody against SARS-CoV-2 administered in the first 3 days of mild symptoms to infected elderly reduces progression of illness and the rate of severe presentations,” senior author Fernando Polack, MD, said in an interview.
“Not any plasma, not any time,” added Dr. Polack, an infectious disease specialist and scientific director at Fundacion INFANT and professor of pediatrics at the University of Buenos Aires. The key, he said, is to select plasma in the upper 28th percentile of IgG antibody concentrations and to administer therapy prior to disease progression.
The study was published online Jan. 6 in The New England Journal of Medicine.
“It’s a very good study and approaches a different population from the PlasmAr study,” Ventura Simonovich, MD, chief of the clinical pharmacology section, Medical Clinic Service, Hospital Italiano de Buenos Aires, said in an interview. “This is the first published randomized controlled trial that shows real benefit in this [older adult] population, the most vulnerable in this disease,” he said.
Dr. Simonovich, who was not affiliated with the current study, was lead author of the PlasmAr trial, which was published in The New England Journal of Medicine Nov. 24, 2020. In that trial, the researchers evaluated adults aged 18 years and older and found no significant benefit with convalescent plasma treatment over placebo for patients with COVID-19 and severe pneumonia.
“We know antibodies work best when given early and in high dose. This is one of the rare reports that validates it in the outpatient setting,” David Sullivan, MD, professor of molecular biology and immunology at Johns Hopkins Bloomberg School of Public Health, Baltimore, said in an interview when asked to comment.
Dr. Sullivan pointed out that most previous studies on convalescent plasma focused on patients with COVID-19 who had severe cases late in the disease course.
Regarding the current study, he said, “The striking thing is treating people within 3 days of illness.”
A more cautious interpretation may be warranted, one expert said. “The study demonstrates the benefit of early intervention. There was a dose-dependent effect, with higher titers providing a greater benefit,” Manoj Menon, MD, MPH, a hematologist and oncologist at the University of Washington, Seattle, said in an interview.
“Taken together, the findings have biologic plausibility and produce more data on the role of convalescent plasma to a relevant age cohort,” he added.
However, Dr. Menon said: “Given the limited sample size, I do not think this study, although well conducted, definitively addresses the role of convalescent plasma for COVID-19. But it does merit additional study.”
A search for clear answers
Treatments that target the early stages of COVID-19 “remain elusive. Few strategies provide benefit, several have failed, and others are being evaluated,” the researchers noted. “In hospitalized patients with COVID-19, the infusion of convalescent plasma against SARS-CoV-2 late in the course of illness has not shown clear benefits and, consequently, the most appropriate antibody concentrations for effective treatment are unclear.”
To learn more, Dr. Polack and colleagues included patients with PCR-confirmed COVID-19 who were aged 75 years or older, regardless of comorbidities. They also included patients aged 65-74 years who had at least one underlying condition. Participants were enrolled at clinical sites or geriatric units in Argentina. The mean age was 77 years, and 62% were women.
In an intent-to-treat analysis, the primary outcome – severe respiratory disease – occurred in 16% of the plasma recipients, vs. 31% of the group that received placebo. The relative risk was 0.52 (95% confidence interval, 0.29-0.94; P = .03).
The number needed to treat to avoid a severe respiratory disease episode was 7 (95% CI, 4-50).
Life-threatening respiratory disease, a secondary outcome, occurred in four people in the plasma group, compared with 10 in the placebo group. Two patients in the treatment group and four patients in the placebo group died.
The researchers also ran a modified intent-to-treat analysis that excluded six participants who experienced severe respiratory disease prior to receiving plasma or placebo. In this analysis, efficacy of plasma therapy increased to 60%.
“Again, this finding suggests that early intervention is critical for efficacy,” the investigators noted.
The investigators, who are based in Argentina, defined their primary endpoint as a respiratory rate of 30 or more breaths per minute and/or an oxygen saturation of less than 93% while breathing ambient air.
Dr. Sullivan pointed out that this is equivalent to the threshold commonly used for hospitalizing people with COVID-19 in the United States. “So it’s equivalent to avoiding hospitalizations. The take-home is high-titer plasma prevents respiratory distress, which equals hospitalization for us.”
Dr. Sullivan is conducting similar research in the United States regarding the use of plasma for treatment or prevention. He and colleagues are evaluating adults aged 18-90 years, “not just the ones at highest risk for going to the hospital,” he said. Enrollment is ongoing.
An inexpensive therapy with global potential?
“Although our trial lacked the statistical power to discern long-term outcomes, the convalescent plasma group appeared to have better outcomes than the placebo group with respect to all secondary endpoints,” the researchers wrote. “Our findings underscore the need to return to the classic approach of treating acute viral infections early, and they define IgG targets that facilitate donor selection.”
Dr. Polack said, “This is an inexpensive solution to mitigate the burden of severe illness in the population most vulnerable to the virus: the elderly. And it has the attraction of being applicable not only in industrialized countries but in many areas of the developing world.”
Convalescent plasma “is a potentially inexpensive alternative to monoclonal antibodies,” the researchers added. Furthermore, “early infusions of convalescent plasma can provide a bridge to recovery for at-risk patients until vaccines become widely available.”
Dr. Polack said the study findings did not surprise him. “We always thought that, as it has been the case in the past with many therapeutic strategies against respiratory and other viral infections, the earlier you treat, the better.
“We just hoped that within 72 hours of symptoms we would be treating early enough – remember that there is a 4- to 5-day incubation period that the virus leverages before the first symptom – and with enough antibody,” he added.
“We are glad it worked,” he said.
The study was supported by the Bill and Melinda Gates Foundation and by the Fundación INFANT Pandemic Fund. Dr. Polack, Dr. Simonovich, and Dr. Sullivan have disclosed various financial relationships industry.
A version of this article first appeared on Medscape.com.
Guidance issued on COVID vaccine use in patients with dermal fillers
outlining the potential risk and clinical relevance.
The association is not surprising, since other vaccines, including the influenza vaccine, have also been associated with inflammatory reactions in patients with dermal fillers. A warning about inflammatory events from these and other immunologic triggers should be part of routine informed consent, according to Sue Ellen Cox, MD, a coauthor of the guidance and the ASDS president-elect.
“Patients who have had dermal filler should not be discouraged from receiving the vaccine, and those who have received the vaccine should not be discouraged from receiving dermal filler,” Dr. Cox, who practices in Chapel Hill, N.C., said in an interview.
The only available data to assess the risk came from the trial of the Moderna vaccine. Of a total of 15,184 participants who received at least one dose of mRNA-1273, three developed facial or lip swelling that was presumably related to dermal filler. In the placebo group, there were no comparable inflammatory events.
“This is a very small number, but there is no reliable information about the number of patients in either group who had dermal filler, so we do not know the denominator,” Dr. Cox said.
In all three cases, the swelling at the site of dermal filler was observed within 2 days of the vaccination. None were considered a serious adverse event and all resolved. The filler had been administered 2 weeks prior to vaccination in one case, 6 months prior in a second, and time of administration was unknown in the third.
The resolution of the inflammatory reactions associated with the SARS-CoV-2 vaccine is similar to those related to dermal fillers following other immunologic triggers, which not only include other vaccines, but viral or bacterial illnesses and dental procedures. Typically, they are readily controlled with oral corticosteroids, but also typically resolve even in the absence of treatment, according to Dr. Cox.
“The good news is that these will go away,” Dr. Cox said.
The ASDS guidance is meant to alert clinicians and patients to the potential association between inflammatory events and SARS-CoV-2 vaccination in patients with dermal filler, but Dr. Cox said that it will ultimately have very little effect on her own practice. She already employs an informed consent that includes language warning about the potential risk of local reactions to immunological triggers that include vaccines. SARS-CoV-2 vaccination can now be added to examples of potential triggers, but it does not change the importance of informing patients of such triggers, Dr. Cox explained.
Asked if patients should be informed specifically about the association between dermal filler inflammatory reactions and SARS-CoV-2 vaccine, the current ASDS president and first author of the guidance, Mathew Avram, MD, JD, suggested that they should. Although he emphasized that the side effect is clearly rare, he believes it deserves attention.
“We wanted dermatologists and other physicians to be aware of the potential. We focused on the available data but specifically decided not to provide any treatment recommendations at this time,” he said in an interview.
As new data become available, the Soft-Tissue Fillers Guideline Task Force of the ASDS, which provided the guidance, will continue to monitor the relationship between SARS-CoV-2 vaccinations and dermal filler reactions, including other SARS-CoV-2 vaccines and the relative risks for hyaluronic acid and non–hyaluronic acid types of fillers.
“Our guidance was based only on the trial data, but there will soon be tens of millions of patients exposed to several different SARS-CoV-2 vaccines. We may learn things we do not know now, and we plan to communicate to our membership and others any new information as events unfold,” said Dr. Avram, who is director of dermatologic surgery, Massachusetts General Hospital, Boston,
Based on her own expertise in the field, Dr. Cox suggested that administration of SARS-CoV-2 vaccine and administration of dermal filler should be separated by at least 2 weeks regardless of which comes first. Her recommendation is not based on controlled data, but she considers this a prudent interval even if it has not been tested in a controlled study.
The full ASDS guidance is scheduled to appear in an upcoming issue of Dermatologic Surgery.
As new data become available, the Soft-tissue Fillers Guideline Task Force of the ASDS, which provided the guidance, will continue to monitor the relationship between SARS-CoV-2 vaccinations and dermal filler reactions, including other types of vaccines and the relative risks for hyaluronic acid and non–hyaluronic acid types of fillers.
This article was updated 1/7/21.
outlining the potential risk and clinical relevance.
The association is not surprising, since other vaccines, including the influenza vaccine, have also been associated with inflammatory reactions in patients with dermal fillers. A warning about inflammatory events from these and other immunologic triggers should be part of routine informed consent, according to Sue Ellen Cox, MD, a coauthor of the guidance and the ASDS president-elect.
“Patients who have had dermal filler should not be discouraged from receiving the vaccine, and those who have received the vaccine should not be discouraged from receiving dermal filler,” Dr. Cox, who practices in Chapel Hill, N.C., said in an interview.
The only available data to assess the risk came from the trial of the Moderna vaccine. Of a total of 15,184 participants who received at least one dose of mRNA-1273, three developed facial or lip swelling that was presumably related to dermal filler. In the placebo group, there were no comparable inflammatory events.
“This is a very small number, but there is no reliable information about the number of patients in either group who had dermal filler, so we do not know the denominator,” Dr. Cox said.
In all three cases, the swelling at the site of dermal filler was observed within 2 days of the vaccination. None were considered a serious adverse event and all resolved. The filler had been administered 2 weeks prior to vaccination in one case, 6 months prior in a second, and time of administration was unknown in the third.
The resolution of the inflammatory reactions associated with the SARS-CoV-2 vaccine is similar to those related to dermal fillers following other immunologic triggers, which not only include other vaccines, but viral or bacterial illnesses and dental procedures. Typically, they are readily controlled with oral corticosteroids, but also typically resolve even in the absence of treatment, according to Dr. Cox.
“The good news is that these will go away,” Dr. Cox said.
The ASDS guidance is meant to alert clinicians and patients to the potential association between inflammatory events and SARS-CoV-2 vaccination in patients with dermal filler, but Dr. Cox said that it will ultimately have very little effect on her own practice. She already employs an informed consent that includes language warning about the potential risk of local reactions to immunological triggers that include vaccines. SARS-CoV-2 vaccination can now be added to examples of potential triggers, but it does not change the importance of informing patients of such triggers, Dr. Cox explained.
Asked if patients should be informed specifically about the association between dermal filler inflammatory reactions and SARS-CoV-2 vaccine, the current ASDS president and first author of the guidance, Mathew Avram, MD, JD, suggested that they should. Although he emphasized that the side effect is clearly rare, he believes it deserves attention.
“We wanted dermatologists and other physicians to be aware of the potential. We focused on the available data but specifically decided not to provide any treatment recommendations at this time,” he said in an interview.
As new data become available, the Soft-Tissue Fillers Guideline Task Force of the ASDS, which provided the guidance, will continue to monitor the relationship between SARS-CoV-2 vaccinations and dermal filler reactions, including other SARS-CoV-2 vaccines and the relative risks for hyaluronic acid and non–hyaluronic acid types of fillers.
“Our guidance was based only on the trial data, but there will soon be tens of millions of patients exposed to several different SARS-CoV-2 vaccines. We may learn things we do not know now, and we plan to communicate to our membership and others any new information as events unfold,” said Dr. Avram, who is director of dermatologic surgery, Massachusetts General Hospital, Boston,
Based on her own expertise in the field, Dr. Cox suggested that administration of SARS-CoV-2 vaccine and administration of dermal filler should be separated by at least 2 weeks regardless of which comes first. Her recommendation is not based on controlled data, but she considers this a prudent interval even if it has not been tested in a controlled study.
The full ASDS guidance is scheduled to appear in an upcoming issue of Dermatologic Surgery.
As new data become available, the Soft-tissue Fillers Guideline Task Force of the ASDS, which provided the guidance, will continue to monitor the relationship between SARS-CoV-2 vaccinations and dermal filler reactions, including other types of vaccines and the relative risks for hyaluronic acid and non–hyaluronic acid types of fillers.
This article was updated 1/7/21.
outlining the potential risk and clinical relevance.
The association is not surprising, since other vaccines, including the influenza vaccine, have also been associated with inflammatory reactions in patients with dermal fillers. A warning about inflammatory events from these and other immunologic triggers should be part of routine informed consent, according to Sue Ellen Cox, MD, a coauthor of the guidance and the ASDS president-elect.
“Patients who have had dermal filler should not be discouraged from receiving the vaccine, and those who have received the vaccine should not be discouraged from receiving dermal filler,” Dr. Cox, who practices in Chapel Hill, N.C., said in an interview.
The only available data to assess the risk came from the trial of the Moderna vaccine. Of a total of 15,184 participants who received at least one dose of mRNA-1273, three developed facial or lip swelling that was presumably related to dermal filler. In the placebo group, there were no comparable inflammatory events.
“This is a very small number, but there is no reliable information about the number of patients in either group who had dermal filler, so we do not know the denominator,” Dr. Cox said.
In all three cases, the swelling at the site of dermal filler was observed within 2 days of the vaccination. None were considered a serious adverse event and all resolved. The filler had been administered 2 weeks prior to vaccination in one case, 6 months prior in a second, and time of administration was unknown in the third.
The resolution of the inflammatory reactions associated with the SARS-CoV-2 vaccine is similar to those related to dermal fillers following other immunologic triggers, which not only include other vaccines, but viral or bacterial illnesses and dental procedures. Typically, they are readily controlled with oral corticosteroids, but also typically resolve even in the absence of treatment, according to Dr. Cox.
“The good news is that these will go away,” Dr. Cox said.
The ASDS guidance is meant to alert clinicians and patients to the potential association between inflammatory events and SARS-CoV-2 vaccination in patients with dermal filler, but Dr. Cox said that it will ultimately have very little effect on her own practice. She already employs an informed consent that includes language warning about the potential risk of local reactions to immunological triggers that include vaccines. SARS-CoV-2 vaccination can now be added to examples of potential triggers, but it does not change the importance of informing patients of such triggers, Dr. Cox explained.
Asked if patients should be informed specifically about the association between dermal filler inflammatory reactions and SARS-CoV-2 vaccine, the current ASDS president and first author of the guidance, Mathew Avram, MD, JD, suggested that they should. Although he emphasized that the side effect is clearly rare, he believes it deserves attention.
“We wanted dermatologists and other physicians to be aware of the potential. We focused on the available data but specifically decided not to provide any treatment recommendations at this time,” he said in an interview.
As new data become available, the Soft-Tissue Fillers Guideline Task Force of the ASDS, which provided the guidance, will continue to monitor the relationship between SARS-CoV-2 vaccinations and dermal filler reactions, including other SARS-CoV-2 vaccines and the relative risks for hyaluronic acid and non–hyaluronic acid types of fillers.
“Our guidance was based only on the trial data, but there will soon be tens of millions of patients exposed to several different SARS-CoV-2 vaccines. We may learn things we do not know now, and we plan to communicate to our membership and others any new information as events unfold,” said Dr. Avram, who is director of dermatologic surgery, Massachusetts General Hospital, Boston,
Based on her own expertise in the field, Dr. Cox suggested that administration of SARS-CoV-2 vaccine and administration of dermal filler should be separated by at least 2 weeks regardless of which comes first. Her recommendation is not based on controlled data, but she considers this a prudent interval even if it has not been tested in a controlled study.
The full ASDS guidance is scheduled to appear in an upcoming issue of Dermatologic Surgery.
As new data become available, the Soft-tissue Fillers Guideline Task Force of the ASDS, which provided the guidance, will continue to monitor the relationship between SARS-CoV-2 vaccinations and dermal filler reactions, including other types of vaccines and the relative risks for hyaluronic acid and non–hyaluronic acid types of fillers.
This article was updated 1/7/21.
No increase seen in children’s cumulative COVID-19 burden
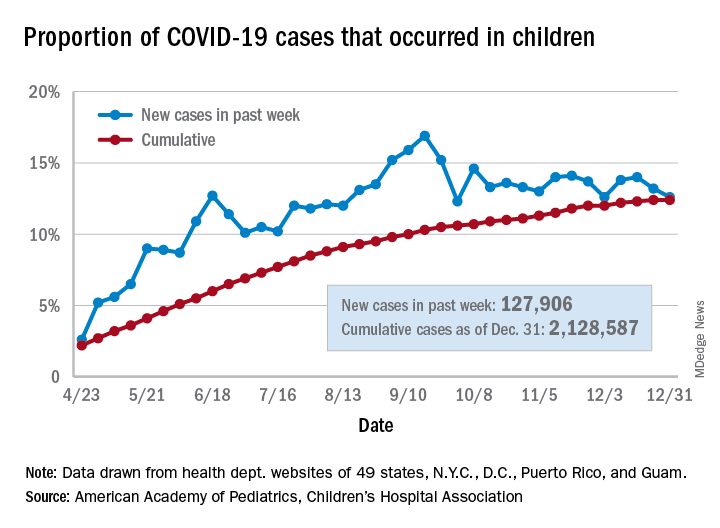
Children’s share of the cumulative COVID-19 burden remained at 12.4% for a second consecutive week, the AAP and CHA said in their weekly report. The last full week of 2020 also marked the second consecutive drop in new cases, although that may be holiday related.
There were almost 128,000 new cases of COVID-19 reported in children for the week, down from 179,000 cases the week before (Dec. 24) and down from the pandemic high of 182,000 reported 2 weeks earlier (Dec.17), based on data from 49 state health departments (excluding New York), along with the District of Columbia, New York City, Puerto Rico, and Guam.
Children’s proportion of new cases for the week, 12.6%, is at its lowest point since early October after dropping for the second week in a row. The cumulative rate of COVID-19 infection, however, is now 2,828 cases per 100,000 children, up from 2,658 the previous week, the AAP and CHA said.
State-level metrics show that North Dakota has the highest cumulative rate at 7,851 per 100,000 children and Hawaii the lowest at 828. Wyoming’s cumulative proportion of child cases, 20.3%, is the highest in the country, while Florida, which uses an age range of 0-14 years for children, is the lowest at 7.1%. California’s total of 268,000 cases is almost double the number of second-place Illinois (138,000), the AAP/CHA data show.
Cumulative child deaths from COVID-19 are up to 179 in the jurisdictions reporting such data (43 states and New York City). That represents just 0.6% of all coronavirus-related deaths and has changed little over the last several months – never rising higher than 0.7% or dropping below 0.6% since early July, according to the report.

Children’s share of the cumulative COVID-19 burden remained at 12.4% for a second consecutive week, the AAP and CHA said in their weekly report. The last full week of 2020 also marked the second consecutive drop in new cases, although that may be holiday related.
There were almost 128,000 new cases of COVID-19 reported in children for the week, down from 179,000 cases the week before (Dec. 24) and down from the pandemic high of 182,000 reported 2 weeks earlier (Dec.17), based on data from 49 state health departments (excluding New York), along with the District of Columbia, New York City, Puerto Rico, and Guam.
Children’s proportion of new cases for the week, 12.6%, is at its lowest point since early October after dropping for the second week in a row. The cumulative rate of COVID-19 infection, however, is now 2,828 cases per 100,000 children, up from 2,658 the previous week, the AAP and CHA said.
State-level metrics show that North Dakota has the highest cumulative rate at 7,851 per 100,000 children and Hawaii the lowest at 828. Wyoming’s cumulative proportion of child cases, 20.3%, is the highest in the country, while Florida, which uses an age range of 0-14 years for children, is the lowest at 7.1%. California’s total of 268,000 cases is almost double the number of second-place Illinois (138,000), the AAP/CHA data show.
Cumulative child deaths from COVID-19 are up to 179 in the jurisdictions reporting such data (43 states and New York City). That represents just 0.6% of all coronavirus-related deaths and has changed little over the last several months – never rising higher than 0.7% or dropping below 0.6% since early July, according to the report.

Children’s share of the cumulative COVID-19 burden remained at 12.4% for a second consecutive week, the AAP and CHA said in their weekly report. The last full week of 2020 also marked the second consecutive drop in new cases, although that may be holiday related.
There were almost 128,000 new cases of COVID-19 reported in children for the week, down from 179,000 cases the week before (Dec. 24) and down from the pandemic high of 182,000 reported 2 weeks earlier (Dec.17), based on data from 49 state health departments (excluding New York), along with the District of Columbia, New York City, Puerto Rico, and Guam.
Children’s proportion of new cases for the week, 12.6%, is at its lowest point since early October after dropping for the second week in a row. The cumulative rate of COVID-19 infection, however, is now 2,828 cases per 100,000 children, up from 2,658 the previous week, the AAP and CHA said.
State-level metrics show that North Dakota has the highest cumulative rate at 7,851 per 100,000 children and Hawaii the lowest at 828. Wyoming’s cumulative proportion of child cases, 20.3%, is the highest in the country, while Florida, which uses an age range of 0-14 years for children, is the lowest at 7.1%. California’s total of 268,000 cases is almost double the number of second-place Illinois (138,000), the AAP/CHA data show.
Cumulative child deaths from COVID-19 are up to 179 in the jurisdictions reporting such data (43 states and New York City). That represents just 0.6% of all coronavirus-related deaths and has changed little over the last several months – never rising higher than 0.7% or dropping below 0.6% since early July, according to the report.
COVID-19 vaccines: The rollout, the risks, and the reason to still wear a mask
REFERENCES
- Oliver SE, Gargano JW, Marin M; et al. The Advisory Committee on Immunization Practices’ interim recommendation for use of Pfizer-BioNTech COVID-19 vaccine—United States, December 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1922-1924. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/69/wr/mm6950e2.htm
- 2. Oliver SE, Gargano JW, Marin M; et al. The Advisory Committee on Immunization Practices’ interim recommendation for use of Moderna COVID-19 vaccine—United States, December 2020. MMWR Morbid Mortal Wkly Rep. 2021;69:1653-1656. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/69/wr/mm695152e1.htm
- CDC. COVID-19 vaccines: update on allergic reactions, contraindications, and precautions [webinar]. December 30, 2020. Accessed January 6, 2021. https://emergency.cdc.gov/coca/calls/2020/callinfo_123020.asp
- CDC. What clinicians need to know about the Pfizer-BioNTech and Moderna COVID-19 vaccines [webinar]. December 18, 2020. Accessed January 6, 2021. https://emergency.cdc.gov/coca/calls/2020/callinfo_121820.asp
- CDC COVID-19 Response Team; Food and Drug Administration. Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine—United States, December 14-23, 2020. MMWR Morb Mortal Wkly Rep. ePub: January 6, 2021. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/70/wr/mm7002e1.htm
REFERENCES
- Oliver SE, Gargano JW, Marin M; et al. The Advisory Committee on Immunization Practices’ interim recommendation for use of Pfizer-BioNTech COVID-19 vaccine—United States, December 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1922-1924. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/69/wr/mm6950e2.htm
- 2. Oliver SE, Gargano JW, Marin M; et al. The Advisory Committee on Immunization Practices’ interim recommendation for use of Moderna COVID-19 vaccine—United States, December 2020. MMWR Morbid Mortal Wkly Rep. 2021;69:1653-1656. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/69/wr/mm695152e1.htm
- CDC. COVID-19 vaccines: update on allergic reactions, contraindications, and precautions [webinar]. December 30, 2020. Accessed January 6, 2021. https://emergency.cdc.gov/coca/calls/2020/callinfo_123020.asp
- CDC. What clinicians need to know about the Pfizer-BioNTech and Moderna COVID-19 vaccines [webinar]. December 18, 2020. Accessed January 6, 2021. https://emergency.cdc.gov/coca/calls/2020/callinfo_121820.asp
- CDC COVID-19 Response Team; Food and Drug Administration. Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine—United States, December 14-23, 2020. MMWR Morb Mortal Wkly Rep. ePub: January 6, 2021. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/70/wr/mm7002e1.htm
REFERENCES
- Oliver SE, Gargano JW, Marin M; et al. The Advisory Committee on Immunization Practices’ interim recommendation for use of Pfizer-BioNTech COVID-19 vaccine—United States, December 2020. MMWR Morbid Mortal Wkly Rep. 2020;69:1922-1924. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/69/wr/mm6950e2.htm
- 2. Oliver SE, Gargano JW, Marin M; et al. The Advisory Committee on Immunization Practices’ interim recommendation for use of Moderna COVID-19 vaccine—United States, December 2020. MMWR Morbid Mortal Wkly Rep. 2021;69:1653-1656. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/69/wr/mm695152e1.htm
- CDC. COVID-19 vaccines: update on allergic reactions, contraindications, and precautions [webinar]. December 30, 2020. Accessed January 6, 2021. https://emergency.cdc.gov/coca/calls/2020/callinfo_123020.asp
- CDC. What clinicians need to know about the Pfizer-BioNTech and Moderna COVID-19 vaccines [webinar]. December 18, 2020. Accessed January 6, 2021. https://emergency.cdc.gov/coca/calls/2020/callinfo_121820.asp
- CDC COVID-19 Response Team; Food and Drug Administration. Allergic reactions including anaphylaxis after receipt of the first dose of Pfizer-BioNTech COVID-19 vaccine—United States, December 14-23, 2020. MMWR Morb Mortal Wkly Rep. ePub: January 6, 2021. Accessed January 13, 2021. www.cdc.gov/mmwr/volumes/70/wr/mm7002e1.htm
Is it safe to be pregnant during the COVID-19 pandemic?
Pregnant women, or women considering pregnancy, want to know—is pregnancy safe in the midst of the coronavirus disease 2019 (COVID-19) pandemic? In this article, I tackle common questions facing reproductive-aged or pregnant women and their providers.
1. What are the risks of COVID-19 in pregnancy?
A large, national prospective cohort study of outpatient pregnant and recently postpartum women with the diagnosis of suspected or confirmed COVID-19 demonstrated that many affected women have mild illnesses, with typical symptoms including cough, sore throat, body aches, fever, and headache.1 Although symptoms were most common within the first 3 weeks of presentation, approximately 25% of women had a protracted course of symptoms (8 or more weeks). As this cohort disproportionately enrolled outpatients, it is important to note that many women had mild illnesses, which is the most likely course of infection in otherwise healthy, young women.
Data on the impact of COVID-19 on rates of miscarriage and birth defects are limited, yet the published reports are reassuring, with no increased risks of miscarriage, and no clear signal for birth defects.2
In a prospective cohort study across 3 New York City institutions when universal severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) testing was recommended upon admission for delivery, approximately 80% of women who were positive were asymptomatic.3 Maternal outcomes generally were reassuring, with no patients experiencing severe or critical illness. There were no differences in preterm delivery rates by SARS-CoV-2 status, but the rate of cesarean delivery was higher among women with COVID-19, for unclear reasons. Most notably, the rate of postpartum complications was 13% among women with COVID-19, versus 2.5% among women without COVID-19. These complications included readmission for worsening COVID-19, postpartum hypoxia, and postpartum fever.
A recent prospective cohort study from 1 institution in Texas similarly demonstrated favorable maternal outcomes with COVID-19, with 95% of women with asymptomatic or mild illness, and no differences in adverse pregnancy outcomes between COVID-19–positive and COVID-19–negative women, including cesarean delivery rate.4
Finally, certain characteristics increase the risk of COVID-19 among pregnant women and nonpregnant individuals alike. In a nationwide prospective cohort from the United Kingdom, medical comorbidities including obesity, diabetes (gestational or pregestational), hypertension, as well as Black or other minority ethnicities are associated with COVID-19.5 This is particularly notable given universal health insurance in the United Kingdom. Other data have also confirmed that women with comorbidities, women of Black or Hispanic ethnicity, and women with lower socioeconomic status, are at increased risk of COVID-19.3,6,7
2. Is COVID-19 worse in pregnancy?
Given the well-documented risks of COVID-19 outside of pregnancy, is COVID-19 worse in a pregnant woman than in a nonpregnant woman? The most recent guidance from the Centers for Disease Control and Prevention (CDC) from November 2020 suggests that pregnant women are at increased risk for severe illness.8 However, it is important to understand the design of this study in order to appreciate its implications. Laboratory confirmed SARS-CoV-2 in the United States is systematically reported to the CDC. Among women aged 15–44 years with such confirmation, data on pregnancy status were available for 35.5%, almost 90% of whom were symptomatic. Within this cohort of largely symptomatic pregnant women, risks of intensive care unit (ICU) admission, invasive ventilation, and use of extracorporeal membrane oxygenation (ECMO) were approximately 2 to 3 times higher for pregnant women than for nonpregnant women. The absolute risks, however, were low. The risk of ICU admission for symptomatic pregnant women was approximately 1%; the risk of invasive ventilation, 0.3%; and the risk of ECMO, 0.1%.
Moreover, the lack of uniform data capture on pregnancy status for all women ages 15–44 years may skew the population with known pregnancy status to be sicker and, thus, may bias the results toward increased risks. Nevertheless, there is consistency in several publications with different data sources, all of which suggest pregnancy is an independent risk factor for increased severity of COVID-19.9-11 Additionally, women with medical comorbidities (such as pregestational or gestational diabetes or obesity) are more likely to have severe COVID-19.
Continue to: 3. What are newborn outcomes if COVID-19 is diagnosed during pregnancy?...
3. What are newborn outcomes if COVID-19 is diagnosed during pregnancy?
Two large cohorts of newborns, disproportionately term infants, from the first wave of the pandemic in New York City, have reassuring news. In one cohort of 101 infants born at 2 New York City institutions to SARS-CoV-2–positive mothers, 2 neonates were diagnosed with SARS-CoV-2 during the immediate postnatal period.12 Neither infant demonstrated clinical COVID-19. In another cohort of 120 infants born at 3 other New York City institutions to SARS-CoV-2–positive mothers and tested systematically within 24 hours of life, 5–7 days of life, and 14 days of life, there were no neonates who tested positive for SARS-CoV-2 at the initial time point. Among the 79 infants who had testing at 5–7 days of life and the 72 tested at 14 days of life, there were no infants positive for SARS-CoV-2.13 It is important to note that case reports and small case series have demonstrated some convincing evidence of vertical transmission. However, the overwhelming evidence suggests this risk is very low.
4. What is a reasonable outpatient setting–approach to managing COVID-19 in a pregnant woman?
Women should be counseled to quarantine for 10 to 14 days from symptom onset or, if asymptomatic, from positive polymerase chain reaction (PCR) test. Warning signs of worsening COVID-19 disease should be reviewed. Serial telemedicine follow-up for 10 to 14 days is recommended to ensure clinical stability and continued management as an outpatient. A home pulse oximeter is also recommended. Women should be advised to check their oxygen saturation daily and to call if oxygen saturation becomes less than 93%. Supportive care is recommended.
If delay in obstetric care may result in adverse pregnancy outcomes (for instance, postponing indicated fetal surveillance), obstetric care should be delivered, with appropriate personal protective equipment for health care workers and minimization of exposure of other pregnant women to the infected patient. Appointments should be scheduled at the end of the day.
During influenza season, women should receive empiric oseltamivir treatment (75 mg twice a day) per CDC guidelines for symptoms that may also be consistent with influenza, regardless of testing.
Prophylactic anticoagulation is not indicated for pregnant antepartum women who do not require inpatient care.
If inpatient care is required, management is individualized.
The approach to prenatal care after resolution of COVID-19 is not evidence-based. At my institution, all patients have a detailed mid-trimester anatomic evaluation, but if this is not routine, a detailed anatomic ultrasound (Current Procedural Terminology code 76811) may be considered. Additionally, for women with COVID-19 we perform one third-trimester growth ultrasound to screen for fetal growth restriction, on the basis of several placental studies demonstrating clots on the fetal or maternal side of the placenta.3,14 Routine antenatal testing in the absence of growth restriction, or other comorbid conditions for which testing occurs, is not recommended.
Continue to: 5. What if asymptomatic or mild COVID-19 is diagnosed at the time of delivery?...
5. What if asymptomatic or mild COVID-19 is diagnosed at the time of delivery? What is reasonable management?
Asymptomatic or mildly symptomatic COVID-19 should not alter obstetric management, beyond appropriate use of personal protective equipment. Delayed cord clamping is also reasonable, if there are no other contraindications, as there is no documented harm associated with this practice among women with COVID-19.
Women with COVID-19 may be at higher risk for venous thromboembolic events in the postpartum period. At my institution, prophylactic postpartum anticoagulation is recommended for 2 weeks after vaginal delivery, and 6 weeks after cesarean delivery.
During the postpartum hospitalization, given reassuring data about vertical transmission and postnatal horizontal transmission risks, babies may room in with mothers in a single private room, if rooming-in is the current standard of care—as long as the mother and newborn do not require higher levels of care. Mothers should wear a mask and use hand hygiene when in contact with the baby. Skin-to-skin and breastfeeding or infant feeding of breast milk are appropriate practices to continue. There is no evidence to suggest that transmission of COVID-19 can occur via breastmilk; however, given the close contact inherent in breastfeeding, transmission through direct contact or maternal respiratory droplets is possible, and thus maternal use of masks and hand hygiene is recommended. When not feeding, the infant should be 6 feet away, and if possible, in an isolette.
6. When can individuals with COVID-19 discontinue transmission precautions or “home quarantine”?
For women with mildly symptomatic COVID-19 and without immunocompromise, home quarantine can be discontinued 10 days after onset of symptoms as long as there has been symptom improvement and no fever for at least 24 hours without the use of antipyretics. For immunocompetent women with incidentally diagnosed asymptomatic COVID-19, home quarantine can be discontinued 10 days after the positive test was obtained. Pregnancy in and of itself is not an immunocompromising condition.15,16
For women with severe or critical COVID-19, who were hospitalized due to their clinical status, home quarantine can be discontinued when at least 10 days, and up to 20 days, after onset of symptoms and with symptom improvement and with no fever for at least 24 hours, without the use of antipyretics. Local hospital infection control experts may be able to guide the recommended practice for your site better, based on local information.15,16
Repeating a PCR test to discontinue home quarantine is not recommended in most circumstances, as individuals may have prolonged shedding of noninfectious particles in their nasopharynx. Immunocompromise may be one exception to this general guidance, but consultation with local hospital infection control experts will help guide management.15,16
7. Should women get pregnant during the COVID-19 pandemic?
Every pandemic has its own set of implications for the health of the mother, fetus, or both, and COVID-19 is no exception. While there are risks, described above, to mother and fetus, these risks are not so catastrophic as to strongly and directively recommend a patient not become pregnant.17 Moreover, the last several months of the pandemic have demonstrated that consistent mask usage, social distancing, and hand hygiene, are effective methods of preventing the acquisition of COVID-19. All of these risk-reducing strategies are available to pregnant women. Finally, accessing care during a pandemic in a hospital setting does not also pose a risk for acquisition of SARS-CoV-2.18
Continue to: 8. Is the COVID-19 vaccine safe for pregnant or postpartum/lactating women?...
8. Is the COVID-19 vaccine safe for pregnant or postpartum/lactating women?
On December 11, 2020, the US Food and Drug Administration (FDA) issued emergency use authorization (EUA) for the Pfizer-BioNtech mRNA vaccine (BNT 162b2) against COVID-19, for individuals aged 16 and older as a 2-dose series given 21 days apart. Among the more than 40,000 individuals in the trial that led to this EUA, vaccine efficacy was 95%.19 Adverse effects included fatigue and headache most commonly, with 16% of vaccine recipients experiencing fever after the second dose. Follow-up regarding safety is planned for 2 years by the manufacturer, in addition to safety monitoring by pre-existing national systems.
On December 18, 2020, the FDA announced EUA for Moderna’s mRNA-based vaccine, mRNA-1273, in men and women aged 18 and older. This is a 2-dose series given 28 days apart. The vaccine efficacy has been reported at 94.5%, with the most common adverse effects being injection site pain, tiredness, headache, muscle pain, chills, joint pain, swollen lymph nodes in the same arm as the injection, nausea and vomiting, and fever.20,21 The phase 3 trial is ongoing.
Despite the speed with which these effective vaccines were developed, it is important to note that all regulatory and safety steps mandated for the development of any vaccine were met for these two, as well as for other COVID-19 vaccinations that will similarly receive EUA from the FDA.
In the EUA for BNT 162b2, the specific language regarding pregnant and lactating women recommends that patients and providers have an individualized conversation about vaccination. In the data presented to the FDA for the Pfizer-BioNtech mRNA vaccine, a limited number of pregnant women received either the vaccine (12 women) or placebo (11 women), with no long-term follow-up data available to characterize either maternal or fetal benefits and risks. The mechanism of action of an mRNA vaccine is to induce the cytoplasmic machinery within cells to create the coronavirus spike protein, which then allows the body’s immune system to create antibodies against this protein and confer protection accordingly. While the above mechanism is not theorized to result in different outcomes or different efficacy, the safety for the pregnant woman and fetus are unknown. It is not believed that vaccination during lactation would cause any adverse outcomes to a neonate, and lactating women do not need to interrupt or discontinue breast milk production in order to receive the vaccine.
The American College of Obstetricians and Gynecologists (ACOG) released a Practice Advisory on December 13, 2020, regarding their recommendations.22 ACOG recommends that vaccines against COVID-19 not be withheld from pregnant or lactating women, if they might otherwise meet criteria for and have access to vaccination. Currently, the CDC’s Advisory Committee on Immunization Practices (ACIP) stated that health care workers and long-term care facility residents represent priority groups to vaccinate in the initial phases of vaccination, given limitations in supply.23 This recommendation is likely to be updated frequently as additional vaccines become available. Shared decision-making between patient and provider may help the patient to make the best decision for herself, but provider input is not required prior to a pregnant woman being vaccinated.
Additional animal data evaluating adverse effects on the reproductive system from developmental and reproductive toxicity (DART) studies for both mRNA vaccines should be available in the coming weeks, which may aid in the counseling of reproductive-aged women.
Vaccine trials to specifically enroll pregnant women are set to begin in early 2021, and more data will certainly inform the conversation between patient and provider regarding risks and benefits.
Conclusions
While the absolute risks of COVID-19 to mothers, fetuses, and neonates is low, pregnancy is a risk factor for severe disease. Many pregnant women with COVID-19 can be safely followed as outpatients via telemedicine, and supportive care is recommended. Inpatient care should be individualized. Pregnancy during the COVID-19 pandemic should be not be absolutely discouraged; instead, a conversation about risk mitigation should be undertaken. The COVID-19 vaccine is available to pregnant and lactating women, and the decision to choose vaccination in pregnancy is in the purview of the patient, in consultation with her physician. ●
- Afshar Y, Gaw SL, Flaherman VJ, et al. Clinical presentation of coronavirus disease 2019 (COVID-19) in pregnant and recently pregnant people. Obstet Gynecol. 2020;128:1117-1125.
- Cosma S, Carosso AR, Cusato J, et al. Coronavirus disease 2019 and first-trimester spontaneous abortion: a casecontrol study of 225 pregnant patients. Am J Obstet Gynecol. 2020;S0002-9378:31177-7. doi: 10.1016/j.ajog.2020.10.005.
- Prabhu M, Cagino K, Matthews KC, et al. Pregnancy and postpartum outcomes in a universally tested population for SARS-CoV-2 in New York City: a prospective cohort study. BJOG. 2020;127:1548-1556.
- Adhikari E, Moreno W, Zofkie AC, et al. Pregnancy outcomes among women with and without severe acute respiratory syndrome coronavirus 2 infection. JAMA Netw Open. 2020;3:e2029256.
- Knight M, Bunch K, Vousden B, et al; UK Obstetric Suveillance System SARS-CoV-2 Infection in Pregnancy Collaborative Group. Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107.
- Emeruwa UN, Ona S, Shaman JL, et al. Associations between built environment, neighborhood socioeconomic status, and SARS-CoV-2 infection among pregnant women in New York City. JAMA. 2020;324:390-392.
- Emeruwa UN, Spiegelman J, Ona S, et al. Influence of race and ethnicity on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection rates and clinical outcomes in pregnancy. Obstet Gynecol. 2020;126:1040-1043.
- Zambrano LD, Ellington S, Strid P, et al; CDC COVID-19 response pregnancy and infant linked outcomes team. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status–United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647.
- Badr DA, Mattern J, Carlin A, et al. Are clinical outcomes worse for pregnant women at ≥20 weeks’ gestation infected with coronavirus disease 2019? A multicenter case control study with propensity score matching. Am J Obstet Gynecol. 2020;223:764-768.
- DeBolt CA, Bianco A, Limaye MA, et al. Pregnant women with severe or critical COVID-19 have increased composite morbidity compared with nonpregnant matched controls. Am J Obstet Gynecol. 2020;S0002-9378:31312-0.
- Collin J, Byström E, Carnahan A, et al. Public Health Agency of Sweden’s Brief Report: pregnant and postpartum women with severe acute respiratory syndrome coronavirus 2 infection in intensive care in Sweden. Acta Obstet Gynecol Scand. 2020;99: 819-822.
- Dumitriu D, Emeruwa UN, Hanft E, et al. Outcomes of neonates born to mothers with severe acute respiratory syndrome coronavirus 2 infection at a large medical center in New York City. JAMA Pediatr. 2020;e204298.
- Salvatore CM, Han JY, Acker KP, et al. Neonatal management and outcomes during the COVID-19 pandemic: an observational cohort study. Lancet Child Adolesc Health. 2020;4: 721-727.
- Shanes ED, Mithal LB, Otero S, et al. Placental pathology in COVID-19. Am J Clin Path. 2020;154:23-32.
- Centers for Disease Control and Prevention. Duration of isolation and precautions for adults with COVID-19. Updated October 19, 2020. https://www.cdc.gov/corona virus/2019-ncov/hcp/duration-isolation.html?CDC _AA_refVal=https%3A%2F%2Fwww.cdc.gov%2F coronavirus%2F2019-ncov%2Fcommunity%2Fstrategy -discontinue-isolation.html. Accessed December 15, 2020.
- Centers for Disease Control and Prevention. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings. Updated August 10, 2020. https://www.cdc.gov /coronavirus/2019-ncov/hcp/disposition-hospitalized -patients.html. Accessed December 15, 2020.
- Rasmussen SA, Lyerly AD, Jamieson DJ. Delaying pregnancy during a public health crisis–examining public health recommendations for COVID-19 and beyond. N Engl J Med. 2020;383:2097-2099.
- Reale SC, Field KG, Lumbreras-Marquez MI, et al. Association between number of in-person health care visits and SARS-CoV-2 infection in obstetrical patients. JAMA. 2020;324: 1210-1212.
- Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT 162b2 mRNA Covid-19 vaccine. N Engl J Med. December 10, 2020. doi: 10.1056/NEJMoa2034577.
- Widge AT, Rouphael NG, Jackson LA, et al. Durability of responses after SARS-CoV-2 mRNA-1273 vaccination. December 3, 2020. doi: 10.1056/NEJMc2032195.
- US Food and Drug Administration. FDA takes additional action in fight against COVID-19 by issuing emergency use authorization for second COVID-19 vaccine. December 18, 2020. https://www.fda.gov/news-events/press-announcements /fda-takes-additional-action-fight-against-covid-19-issuing -emergency-use-authorization-second-covid. Accessed December 22, 2020.
- American College of Obstetricians and Gynecologists. Practice advisory: vaccinating pregnancy and lactating patients against COVID-19. https://www.acog.org/clinical/clinical -guidance/practice-advisory/articles/2020/12/vaccinating -pregnant-and-lactating-patients-against-covid-19. Last updated December 21, 2020. Accessed December 21, 2020.
- Dooling K, McClung N, Chamberland M, et al. The Advisory Committee on Immunization Practices’ interim recommendation for allocating initial supplies of COVID-19 vaccine–United States, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1857-1859.

Pregnant women, or women considering pregnancy, want to know—is pregnancy safe in the midst of the coronavirus disease 2019 (COVID-19) pandemic? In this article, I tackle common questions facing reproductive-aged or pregnant women and their providers.
1. What are the risks of COVID-19 in pregnancy?
A large, national prospective cohort study of outpatient pregnant and recently postpartum women with the diagnosis of suspected or confirmed COVID-19 demonstrated that many affected women have mild illnesses, with typical symptoms including cough, sore throat, body aches, fever, and headache.1 Although symptoms were most common within the first 3 weeks of presentation, approximately 25% of women had a protracted course of symptoms (8 or more weeks). As this cohort disproportionately enrolled outpatients, it is important to note that many women had mild illnesses, which is the most likely course of infection in otherwise healthy, young women.
Data on the impact of COVID-19 on rates of miscarriage and birth defects are limited, yet the published reports are reassuring, with no increased risks of miscarriage, and no clear signal for birth defects.2
In a prospective cohort study across 3 New York City institutions when universal severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) testing was recommended upon admission for delivery, approximately 80% of women who were positive were asymptomatic.3 Maternal outcomes generally were reassuring, with no patients experiencing severe or critical illness. There were no differences in preterm delivery rates by SARS-CoV-2 status, but the rate of cesarean delivery was higher among women with COVID-19, for unclear reasons. Most notably, the rate of postpartum complications was 13% among women with COVID-19, versus 2.5% among women without COVID-19. These complications included readmission for worsening COVID-19, postpartum hypoxia, and postpartum fever.
A recent prospective cohort study from 1 institution in Texas similarly demonstrated favorable maternal outcomes with COVID-19, with 95% of women with asymptomatic or mild illness, and no differences in adverse pregnancy outcomes between COVID-19–positive and COVID-19–negative women, including cesarean delivery rate.4
Finally, certain characteristics increase the risk of COVID-19 among pregnant women and nonpregnant individuals alike. In a nationwide prospective cohort from the United Kingdom, medical comorbidities including obesity, diabetes (gestational or pregestational), hypertension, as well as Black or other minority ethnicities are associated with COVID-19.5 This is particularly notable given universal health insurance in the United Kingdom. Other data have also confirmed that women with comorbidities, women of Black or Hispanic ethnicity, and women with lower socioeconomic status, are at increased risk of COVID-19.3,6,7
2. Is COVID-19 worse in pregnancy?
Given the well-documented risks of COVID-19 outside of pregnancy, is COVID-19 worse in a pregnant woman than in a nonpregnant woman? The most recent guidance from the Centers for Disease Control and Prevention (CDC) from November 2020 suggests that pregnant women are at increased risk for severe illness.8 However, it is important to understand the design of this study in order to appreciate its implications. Laboratory confirmed SARS-CoV-2 in the United States is systematically reported to the CDC. Among women aged 15–44 years with such confirmation, data on pregnancy status were available for 35.5%, almost 90% of whom were symptomatic. Within this cohort of largely symptomatic pregnant women, risks of intensive care unit (ICU) admission, invasive ventilation, and use of extracorporeal membrane oxygenation (ECMO) were approximately 2 to 3 times higher for pregnant women than for nonpregnant women. The absolute risks, however, were low. The risk of ICU admission for symptomatic pregnant women was approximately 1%; the risk of invasive ventilation, 0.3%; and the risk of ECMO, 0.1%.
Moreover, the lack of uniform data capture on pregnancy status for all women ages 15–44 years may skew the population with known pregnancy status to be sicker and, thus, may bias the results toward increased risks. Nevertheless, there is consistency in several publications with different data sources, all of which suggest pregnancy is an independent risk factor for increased severity of COVID-19.9-11 Additionally, women with medical comorbidities (such as pregestational or gestational diabetes or obesity) are more likely to have severe COVID-19.
Continue to: 3. What are newborn outcomes if COVID-19 is diagnosed during pregnancy?...
3. What are newborn outcomes if COVID-19 is diagnosed during pregnancy?
Two large cohorts of newborns, disproportionately term infants, from the first wave of the pandemic in New York City, have reassuring news. In one cohort of 101 infants born at 2 New York City institutions to SARS-CoV-2–positive mothers, 2 neonates were diagnosed with SARS-CoV-2 during the immediate postnatal period.12 Neither infant demonstrated clinical COVID-19. In another cohort of 120 infants born at 3 other New York City institutions to SARS-CoV-2–positive mothers and tested systematically within 24 hours of life, 5–7 days of life, and 14 days of life, there were no neonates who tested positive for SARS-CoV-2 at the initial time point. Among the 79 infants who had testing at 5–7 days of life and the 72 tested at 14 days of life, there were no infants positive for SARS-CoV-2.13 It is important to note that case reports and small case series have demonstrated some convincing evidence of vertical transmission. However, the overwhelming evidence suggests this risk is very low.
4. What is a reasonable outpatient setting–approach to managing COVID-19 in a pregnant woman?
Women should be counseled to quarantine for 10 to 14 days from symptom onset or, if asymptomatic, from positive polymerase chain reaction (PCR) test. Warning signs of worsening COVID-19 disease should be reviewed. Serial telemedicine follow-up for 10 to 14 days is recommended to ensure clinical stability and continued management as an outpatient. A home pulse oximeter is also recommended. Women should be advised to check their oxygen saturation daily and to call if oxygen saturation becomes less than 93%. Supportive care is recommended.
If delay in obstetric care may result in adverse pregnancy outcomes (for instance, postponing indicated fetal surveillance), obstetric care should be delivered, with appropriate personal protective equipment for health care workers and minimization of exposure of other pregnant women to the infected patient. Appointments should be scheduled at the end of the day.
During influenza season, women should receive empiric oseltamivir treatment (75 mg twice a day) per CDC guidelines for symptoms that may also be consistent with influenza, regardless of testing.
Prophylactic anticoagulation is not indicated for pregnant antepartum women who do not require inpatient care.
If inpatient care is required, management is individualized.
The approach to prenatal care after resolution of COVID-19 is not evidence-based. At my institution, all patients have a detailed mid-trimester anatomic evaluation, but if this is not routine, a detailed anatomic ultrasound (Current Procedural Terminology code 76811) may be considered. Additionally, for women with COVID-19 we perform one third-trimester growth ultrasound to screen for fetal growth restriction, on the basis of several placental studies demonstrating clots on the fetal or maternal side of the placenta.3,14 Routine antenatal testing in the absence of growth restriction, or other comorbid conditions for which testing occurs, is not recommended.
Continue to: 5. What if asymptomatic or mild COVID-19 is diagnosed at the time of delivery?...
5. What if asymptomatic or mild COVID-19 is diagnosed at the time of delivery? What is reasonable management?
Asymptomatic or mildly symptomatic COVID-19 should not alter obstetric management, beyond appropriate use of personal protective equipment. Delayed cord clamping is also reasonable, if there are no other contraindications, as there is no documented harm associated with this practice among women with COVID-19.
Women with COVID-19 may be at higher risk for venous thromboembolic events in the postpartum period. At my institution, prophylactic postpartum anticoagulation is recommended for 2 weeks after vaginal delivery, and 6 weeks after cesarean delivery.
During the postpartum hospitalization, given reassuring data about vertical transmission and postnatal horizontal transmission risks, babies may room in with mothers in a single private room, if rooming-in is the current standard of care—as long as the mother and newborn do not require higher levels of care. Mothers should wear a mask and use hand hygiene when in contact with the baby. Skin-to-skin and breastfeeding or infant feeding of breast milk are appropriate practices to continue. There is no evidence to suggest that transmission of COVID-19 can occur via breastmilk; however, given the close contact inherent in breastfeeding, transmission through direct contact or maternal respiratory droplets is possible, and thus maternal use of masks and hand hygiene is recommended. When not feeding, the infant should be 6 feet away, and if possible, in an isolette.
6. When can individuals with COVID-19 discontinue transmission precautions or “home quarantine”?
For women with mildly symptomatic COVID-19 and without immunocompromise, home quarantine can be discontinued 10 days after onset of symptoms as long as there has been symptom improvement and no fever for at least 24 hours without the use of antipyretics. For immunocompetent women with incidentally diagnosed asymptomatic COVID-19, home quarantine can be discontinued 10 days after the positive test was obtained. Pregnancy in and of itself is not an immunocompromising condition.15,16
For women with severe or critical COVID-19, who were hospitalized due to their clinical status, home quarantine can be discontinued when at least 10 days, and up to 20 days, after onset of symptoms and with symptom improvement and with no fever for at least 24 hours, without the use of antipyretics. Local hospital infection control experts may be able to guide the recommended practice for your site better, based on local information.15,16
Repeating a PCR test to discontinue home quarantine is not recommended in most circumstances, as individuals may have prolonged shedding of noninfectious particles in their nasopharynx. Immunocompromise may be one exception to this general guidance, but consultation with local hospital infection control experts will help guide management.15,16
7. Should women get pregnant during the COVID-19 pandemic?
Every pandemic has its own set of implications for the health of the mother, fetus, or both, and COVID-19 is no exception. While there are risks, described above, to mother and fetus, these risks are not so catastrophic as to strongly and directively recommend a patient not become pregnant.17 Moreover, the last several months of the pandemic have demonstrated that consistent mask usage, social distancing, and hand hygiene, are effective methods of preventing the acquisition of COVID-19. All of these risk-reducing strategies are available to pregnant women. Finally, accessing care during a pandemic in a hospital setting does not also pose a risk for acquisition of SARS-CoV-2.18
Continue to: 8. Is the COVID-19 vaccine safe for pregnant or postpartum/lactating women?...
8. Is the COVID-19 vaccine safe for pregnant or postpartum/lactating women?
On December 11, 2020, the US Food and Drug Administration (FDA) issued emergency use authorization (EUA) for the Pfizer-BioNtech mRNA vaccine (BNT 162b2) against COVID-19, for individuals aged 16 and older as a 2-dose series given 21 days apart. Among the more than 40,000 individuals in the trial that led to this EUA, vaccine efficacy was 95%.19 Adverse effects included fatigue and headache most commonly, with 16% of vaccine recipients experiencing fever after the second dose. Follow-up regarding safety is planned for 2 years by the manufacturer, in addition to safety monitoring by pre-existing national systems.
On December 18, 2020, the FDA announced EUA for Moderna’s mRNA-based vaccine, mRNA-1273, in men and women aged 18 and older. This is a 2-dose series given 28 days apart. The vaccine efficacy has been reported at 94.5%, with the most common adverse effects being injection site pain, tiredness, headache, muscle pain, chills, joint pain, swollen lymph nodes in the same arm as the injection, nausea and vomiting, and fever.20,21 The phase 3 trial is ongoing.
Despite the speed with which these effective vaccines were developed, it is important to note that all regulatory and safety steps mandated for the development of any vaccine were met for these two, as well as for other COVID-19 vaccinations that will similarly receive EUA from the FDA.
In the EUA for BNT 162b2, the specific language regarding pregnant and lactating women recommends that patients and providers have an individualized conversation about vaccination. In the data presented to the FDA for the Pfizer-BioNtech mRNA vaccine, a limited number of pregnant women received either the vaccine (12 women) or placebo (11 women), with no long-term follow-up data available to characterize either maternal or fetal benefits and risks. The mechanism of action of an mRNA vaccine is to induce the cytoplasmic machinery within cells to create the coronavirus spike protein, which then allows the body’s immune system to create antibodies against this protein and confer protection accordingly. While the above mechanism is not theorized to result in different outcomes or different efficacy, the safety for the pregnant woman and fetus are unknown. It is not believed that vaccination during lactation would cause any adverse outcomes to a neonate, and lactating women do not need to interrupt or discontinue breast milk production in order to receive the vaccine.
The American College of Obstetricians and Gynecologists (ACOG) released a Practice Advisory on December 13, 2020, regarding their recommendations.22 ACOG recommends that vaccines against COVID-19 not be withheld from pregnant or lactating women, if they might otherwise meet criteria for and have access to vaccination. Currently, the CDC’s Advisory Committee on Immunization Practices (ACIP) stated that health care workers and long-term care facility residents represent priority groups to vaccinate in the initial phases of vaccination, given limitations in supply.23 This recommendation is likely to be updated frequently as additional vaccines become available. Shared decision-making between patient and provider may help the patient to make the best decision for herself, but provider input is not required prior to a pregnant woman being vaccinated.
Additional animal data evaluating adverse effects on the reproductive system from developmental and reproductive toxicity (DART) studies for both mRNA vaccines should be available in the coming weeks, which may aid in the counseling of reproductive-aged women.
Vaccine trials to specifically enroll pregnant women are set to begin in early 2021, and more data will certainly inform the conversation between patient and provider regarding risks and benefits.
Conclusions
While the absolute risks of COVID-19 to mothers, fetuses, and neonates is low, pregnancy is a risk factor for severe disease. Many pregnant women with COVID-19 can be safely followed as outpatients via telemedicine, and supportive care is recommended. Inpatient care should be individualized. Pregnancy during the COVID-19 pandemic should be not be absolutely discouraged; instead, a conversation about risk mitigation should be undertaken. The COVID-19 vaccine is available to pregnant and lactating women, and the decision to choose vaccination in pregnancy is in the purview of the patient, in consultation with her physician. ●

Pregnant women, or women considering pregnancy, want to know—is pregnancy safe in the midst of the coronavirus disease 2019 (COVID-19) pandemic? In this article, I tackle common questions facing reproductive-aged or pregnant women and their providers.
1. What are the risks of COVID-19 in pregnancy?
A large, national prospective cohort study of outpatient pregnant and recently postpartum women with the diagnosis of suspected or confirmed COVID-19 demonstrated that many affected women have mild illnesses, with typical symptoms including cough, sore throat, body aches, fever, and headache.1 Although symptoms were most common within the first 3 weeks of presentation, approximately 25% of women had a protracted course of symptoms (8 or more weeks). As this cohort disproportionately enrolled outpatients, it is important to note that many women had mild illnesses, which is the most likely course of infection in otherwise healthy, young women.
Data on the impact of COVID-19 on rates of miscarriage and birth defects are limited, yet the published reports are reassuring, with no increased risks of miscarriage, and no clear signal for birth defects.2
In a prospective cohort study across 3 New York City institutions when universal severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) testing was recommended upon admission for delivery, approximately 80% of women who were positive were asymptomatic.3 Maternal outcomes generally were reassuring, with no patients experiencing severe or critical illness. There were no differences in preterm delivery rates by SARS-CoV-2 status, but the rate of cesarean delivery was higher among women with COVID-19, for unclear reasons. Most notably, the rate of postpartum complications was 13% among women with COVID-19, versus 2.5% among women without COVID-19. These complications included readmission for worsening COVID-19, postpartum hypoxia, and postpartum fever.
A recent prospective cohort study from 1 institution in Texas similarly demonstrated favorable maternal outcomes with COVID-19, with 95% of women with asymptomatic or mild illness, and no differences in adverse pregnancy outcomes between COVID-19–positive and COVID-19–negative women, including cesarean delivery rate.4
Finally, certain characteristics increase the risk of COVID-19 among pregnant women and nonpregnant individuals alike. In a nationwide prospective cohort from the United Kingdom, medical comorbidities including obesity, diabetes (gestational or pregestational), hypertension, as well as Black or other minority ethnicities are associated with COVID-19.5 This is particularly notable given universal health insurance in the United Kingdom. Other data have also confirmed that women with comorbidities, women of Black or Hispanic ethnicity, and women with lower socioeconomic status, are at increased risk of COVID-19.3,6,7
2. Is COVID-19 worse in pregnancy?
Given the well-documented risks of COVID-19 outside of pregnancy, is COVID-19 worse in a pregnant woman than in a nonpregnant woman? The most recent guidance from the Centers for Disease Control and Prevention (CDC) from November 2020 suggests that pregnant women are at increased risk for severe illness.8 However, it is important to understand the design of this study in order to appreciate its implications. Laboratory confirmed SARS-CoV-2 in the United States is systematically reported to the CDC. Among women aged 15–44 years with such confirmation, data on pregnancy status were available for 35.5%, almost 90% of whom were symptomatic. Within this cohort of largely symptomatic pregnant women, risks of intensive care unit (ICU) admission, invasive ventilation, and use of extracorporeal membrane oxygenation (ECMO) were approximately 2 to 3 times higher for pregnant women than for nonpregnant women. The absolute risks, however, were low. The risk of ICU admission for symptomatic pregnant women was approximately 1%; the risk of invasive ventilation, 0.3%; and the risk of ECMO, 0.1%.
Moreover, the lack of uniform data capture on pregnancy status for all women ages 15–44 years may skew the population with known pregnancy status to be sicker and, thus, may bias the results toward increased risks. Nevertheless, there is consistency in several publications with different data sources, all of which suggest pregnancy is an independent risk factor for increased severity of COVID-19.9-11 Additionally, women with medical comorbidities (such as pregestational or gestational diabetes or obesity) are more likely to have severe COVID-19.
Continue to: 3. What are newborn outcomes if COVID-19 is diagnosed during pregnancy?...
3. What are newborn outcomes if COVID-19 is diagnosed during pregnancy?
Two large cohorts of newborns, disproportionately term infants, from the first wave of the pandemic in New York City, have reassuring news. In one cohort of 101 infants born at 2 New York City institutions to SARS-CoV-2–positive mothers, 2 neonates were diagnosed with SARS-CoV-2 during the immediate postnatal period.12 Neither infant demonstrated clinical COVID-19. In another cohort of 120 infants born at 3 other New York City institutions to SARS-CoV-2–positive mothers and tested systematically within 24 hours of life, 5–7 days of life, and 14 days of life, there were no neonates who tested positive for SARS-CoV-2 at the initial time point. Among the 79 infants who had testing at 5–7 days of life and the 72 tested at 14 days of life, there were no infants positive for SARS-CoV-2.13 It is important to note that case reports and small case series have demonstrated some convincing evidence of vertical transmission. However, the overwhelming evidence suggests this risk is very low.
4. What is a reasonable outpatient setting–approach to managing COVID-19 in a pregnant woman?
Women should be counseled to quarantine for 10 to 14 days from symptom onset or, if asymptomatic, from positive polymerase chain reaction (PCR) test. Warning signs of worsening COVID-19 disease should be reviewed. Serial telemedicine follow-up for 10 to 14 days is recommended to ensure clinical stability and continued management as an outpatient. A home pulse oximeter is also recommended. Women should be advised to check their oxygen saturation daily and to call if oxygen saturation becomes less than 93%. Supportive care is recommended.
If delay in obstetric care may result in adverse pregnancy outcomes (for instance, postponing indicated fetal surveillance), obstetric care should be delivered, with appropriate personal protective equipment for health care workers and minimization of exposure of other pregnant women to the infected patient. Appointments should be scheduled at the end of the day.
During influenza season, women should receive empiric oseltamivir treatment (75 mg twice a day) per CDC guidelines for symptoms that may also be consistent with influenza, regardless of testing.
Prophylactic anticoagulation is not indicated for pregnant antepartum women who do not require inpatient care.
If inpatient care is required, management is individualized.
The approach to prenatal care after resolution of COVID-19 is not evidence-based. At my institution, all patients have a detailed mid-trimester anatomic evaluation, but if this is not routine, a detailed anatomic ultrasound (Current Procedural Terminology code 76811) may be considered. Additionally, for women with COVID-19 we perform one third-trimester growth ultrasound to screen for fetal growth restriction, on the basis of several placental studies demonstrating clots on the fetal or maternal side of the placenta.3,14 Routine antenatal testing in the absence of growth restriction, or other comorbid conditions for which testing occurs, is not recommended.
Continue to: 5. What if asymptomatic or mild COVID-19 is diagnosed at the time of delivery?...
5. What if asymptomatic or mild COVID-19 is diagnosed at the time of delivery? What is reasonable management?
Asymptomatic or mildly symptomatic COVID-19 should not alter obstetric management, beyond appropriate use of personal protective equipment. Delayed cord clamping is also reasonable, if there are no other contraindications, as there is no documented harm associated with this practice among women with COVID-19.
Women with COVID-19 may be at higher risk for venous thromboembolic events in the postpartum period. At my institution, prophylactic postpartum anticoagulation is recommended for 2 weeks after vaginal delivery, and 6 weeks after cesarean delivery.
During the postpartum hospitalization, given reassuring data about vertical transmission and postnatal horizontal transmission risks, babies may room in with mothers in a single private room, if rooming-in is the current standard of care—as long as the mother and newborn do not require higher levels of care. Mothers should wear a mask and use hand hygiene when in contact with the baby. Skin-to-skin and breastfeeding or infant feeding of breast milk are appropriate practices to continue. There is no evidence to suggest that transmission of COVID-19 can occur via breastmilk; however, given the close contact inherent in breastfeeding, transmission through direct contact or maternal respiratory droplets is possible, and thus maternal use of masks and hand hygiene is recommended. When not feeding, the infant should be 6 feet away, and if possible, in an isolette.
6. When can individuals with COVID-19 discontinue transmission precautions or “home quarantine”?
For women with mildly symptomatic COVID-19 and without immunocompromise, home quarantine can be discontinued 10 days after onset of symptoms as long as there has been symptom improvement and no fever for at least 24 hours without the use of antipyretics. For immunocompetent women with incidentally diagnosed asymptomatic COVID-19, home quarantine can be discontinued 10 days after the positive test was obtained. Pregnancy in and of itself is not an immunocompromising condition.15,16
For women with severe or critical COVID-19, who were hospitalized due to their clinical status, home quarantine can be discontinued when at least 10 days, and up to 20 days, after onset of symptoms and with symptom improvement and with no fever for at least 24 hours, without the use of antipyretics. Local hospital infection control experts may be able to guide the recommended practice for your site better, based on local information.15,16
Repeating a PCR test to discontinue home quarantine is not recommended in most circumstances, as individuals may have prolonged shedding of noninfectious particles in their nasopharynx. Immunocompromise may be one exception to this general guidance, but consultation with local hospital infection control experts will help guide management.15,16
7. Should women get pregnant during the COVID-19 pandemic?
Every pandemic has its own set of implications for the health of the mother, fetus, or both, and COVID-19 is no exception. While there are risks, described above, to mother and fetus, these risks are not so catastrophic as to strongly and directively recommend a patient not become pregnant.17 Moreover, the last several months of the pandemic have demonstrated that consistent mask usage, social distancing, and hand hygiene, are effective methods of preventing the acquisition of COVID-19. All of these risk-reducing strategies are available to pregnant women. Finally, accessing care during a pandemic in a hospital setting does not also pose a risk for acquisition of SARS-CoV-2.18
Continue to: 8. Is the COVID-19 vaccine safe for pregnant or postpartum/lactating women?...
8. Is the COVID-19 vaccine safe for pregnant or postpartum/lactating women?
On December 11, 2020, the US Food and Drug Administration (FDA) issued emergency use authorization (EUA) for the Pfizer-BioNtech mRNA vaccine (BNT 162b2) against COVID-19, for individuals aged 16 and older as a 2-dose series given 21 days apart. Among the more than 40,000 individuals in the trial that led to this EUA, vaccine efficacy was 95%.19 Adverse effects included fatigue and headache most commonly, with 16% of vaccine recipients experiencing fever after the second dose. Follow-up regarding safety is planned for 2 years by the manufacturer, in addition to safety monitoring by pre-existing national systems.
On December 18, 2020, the FDA announced EUA for Moderna’s mRNA-based vaccine, mRNA-1273, in men and women aged 18 and older. This is a 2-dose series given 28 days apart. The vaccine efficacy has been reported at 94.5%, with the most common adverse effects being injection site pain, tiredness, headache, muscle pain, chills, joint pain, swollen lymph nodes in the same arm as the injection, nausea and vomiting, and fever.20,21 The phase 3 trial is ongoing.
Despite the speed with which these effective vaccines were developed, it is important to note that all regulatory and safety steps mandated for the development of any vaccine were met for these two, as well as for other COVID-19 vaccinations that will similarly receive EUA from the FDA.
In the EUA for BNT 162b2, the specific language regarding pregnant and lactating women recommends that patients and providers have an individualized conversation about vaccination. In the data presented to the FDA for the Pfizer-BioNtech mRNA vaccine, a limited number of pregnant women received either the vaccine (12 women) or placebo (11 women), with no long-term follow-up data available to characterize either maternal or fetal benefits and risks. The mechanism of action of an mRNA vaccine is to induce the cytoplasmic machinery within cells to create the coronavirus spike protein, which then allows the body’s immune system to create antibodies against this protein and confer protection accordingly. While the above mechanism is not theorized to result in different outcomes or different efficacy, the safety for the pregnant woman and fetus are unknown. It is not believed that vaccination during lactation would cause any adverse outcomes to a neonate, and lactating women do not need to interrupt or discontinue breast milk production in order to receive the vaccine.
The American College of Obstetricians and Gynecologists (ACOG) released a Practice Advisory on December 13, 2020, regarding their recommendations.22 ACOG recommends that vaccines against COVID-19 not be withheld from pregnant or lactating women, if they might otherwise meet criteria for and have access to vaccination. Currently, the CDC’s Advisory Committee on Immunization Practices (ACIP) stated that health care workers and long-term care facility residents represent priority groups to vaccinate in the initial phases of vaccination, given limitations in supply.23 This recommendation is likely to be updated frequently as additional vaccines become available. Shared decision-making between patient and provider may help the patient to make the best decision for herself, but provider input is not required prior to a pregnant woman being vaccinated.
Additional animal data evaluating adverse effects on the reproductive system from developmental and reproductive toxicity (DART) studies for both mRNA vaccines should be available in the coming weeks, which may aid in the counseling of reproductive-aged women.
Vaccine trials to specifically enroll pregnant women are set to begin in early 2021, and more data will certainly inform the conversation between patient and provider regarding risks and benefits.
Conclusions
While the absolute risks of COVID-19 to mothers, fetuses, and neonates is low, pregnancy is a risk factor for severe disease. Many pregnant women with COVID-19 can be safely followed as outpatients via telemedicine, and supportive care is recommended. Inpatient care should be individualized. Pregnancy during the COVID-19 pandemic should be not be absolutely discouraged; instead, a conversation about risk mitigation should be undertaken. The COVID-19 vaccine is available to pregnant and lactating women, and the decision to choose vaccination in pregnancy is in the purview of the patient, in consultation with her physician. ●
- Afshar Y, Gaw SL, Flaherman VJ, et al. Clinical presentation of coronavirus disease 2019 (COVID-19) in pregnant and recently pregnant people. Obstet Gynecol. 2020;128:1117-1125.
- Cosma S, Carosso AR, Cusato J, et al. Coronavirus disease 2019 and first-trimester spontaneous abortion: a casecontrol study of 225 pregnant patients. Am J Obstet Gynecol. 2020;S0002-9378:31177-7. doi: 10.1016/j.ajog.2020.10.005.
- Prabhu M, Cagino K, Matthews KC, et al. Pregnancy and postpartum outcomes in a universally tested population for SARS-CoV-2 in New York City: a prospective cohort study. BJOG. 2020;127:1548-1556.
- Adhikari E, Moreno W, Zofkie AC, et al. Pregnancy outcomes among women with and without severe acute respiratory syndrome coronavirus 2 infection. JAMA Netw Open. 2020;3:e2029256.
- Knight M, Bunch K, Vousden B, et al; UK Obstetric Suveillance System SARS-CoV-2 Infection in Pregnancy Collaborative Group. Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107.
- Emeruwa UN, Ona S, Shaman JL, et al. Associations between built environment, neighborhood socioeconomic status, and SARS-CoV-2 infection among pregnant women in New York City. JAMA. 2020;324:390-392.
- Emeruwa UN, Spiegelman J, Ona S, et al. Influence of race and ethnicity on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection rates and clinical outcomes in pregnancy. Obstet Gynecol. 2020;126:1040-1043.
- Zambrano LD, Ellington S, Strid P, et al; CDC COVID-19 response pregnancy and infant linked outcomes team. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status–United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647.
- Badr DA, Mattern J, Carlin A, et al. Are clinical outcomes worse for pregnant women at ≥20 weeks’ gestation infected with coronavirus disease 2019? A multicenter case control study with propensity score matching. Am J Obstet Gynecol. 2020;223:764-768.
- DeBolt CA, Bianco A, Limaye MA, et al. Pregnant women with severe or critical COVID-19 have increased composite morbidity compared with nonpregnant matched controls. Am J Obstet Gynecol. 2020;S0002-9378:31312-0.
- Collin J, Byström E, Carnahan A, et al. Public Health Agency of Sweden’s Brief Report: pregnant and postpartum women with severe acute respiratory syndrome coronavirus 2 infection in intensive care in Sweden. Acta Obstet Gynecol Scand. 2020;99: 819-822.
- Dumitriu D, Emeruwa UN, Hanft E, et al. Outcomes of neonates born to mothers with severe acute respiratory syndrome coronavirus 2 infection at a large medical center in New York City. JAMA Pediatr. 2020;e204298.
- Salvatore CM, Han JY, Acker KP, et al. Neonatal management and outcomes during the COVID-19 pandemic: an observational cohort study. Lancet Child Adolesc Health. 2020;4: 721-727.
- Shanes ED, Mithal LB, Otero S, et al. Placental pathology in COVID-19. Am J Clin Path. 2020;154:23-32.
- Centers for Disease Control and Prevention. Duration of isolation and precautions for adults with COVID-19. Updated October 19, 2020. https://www.cdc.gov/corona virus/2019-ncov/hcp/duration-isolation.html?CDC _AA_refVal=https%3A%2F%2Fwww.cdc.gov%2F coronavirus%2F2019-ncov%2Fcommunity%2Fstrategy -discontinue-isolation.html. Accessed December 15, 2020.
- Centers for Disease Control and Prevention. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings. Updated August 10, 2020. https://www.cdc.gov /coronavirus/2019-ncov/hcp/disposition-hospitalized -patients.html. Accessed December 15, 2020.
- Rasmussen SA, Lyerly AD, Jamieson DJ. Delaying pregnancy during a public health crisis–examining public health recommendations for COVID-19 and beyond. N Engl J Med. 2020;383:2097-2099.
- Reale SC, Field KG, Lumbreras-Marquez MI, et al. Association between number of in-person health care visits and SARS-CoV-2 infection in obstetrical patients. JAMA. 2020;324: 1210-1212.
- Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT 162b2 mRNA Covid-19 vaccine. N Engl J Med. December 10, 2020. doi: 10.1056/NEJMoa2034577.
- Widge AT, Rouphael NG, Jackson LA, et al. Durability of responses after SARS-CoV-2 mRNA-1273 vaccination. December 3, 2020. doi: 10.1056/NEJMc2032195.
- US Food and Drug Administration. FDA takes additional action in fight against COVID-19 by issuing emergency use authorization for second COVID-19 vaccine. December 18, 2020. https://www.fda.gov/news-events/press-announcements /fda-takes-additional-action-fight-against-covid-19-issuing -emergency-use-authorization-second-covid. Accessed December 22, 2020.
- American College of Obstetricians and Gynecologists. Practice advisory: vaccinating pregnancy and lactating patients against COVID-19. https://www.acog.org/clinical/clinical -guidance/practice-advisory/articles/2020/12/vaccinating -pregnant-and-lactating-patients-against-covid-19. Last updated December 21, 2020. Accessed December 21, 2020.
- Dooling K, McClung N, Chamberland M, et al. The Advisory Committee on Immunization Practices’ interim recommendation for allocating initial supplies of COVID-19 vaccine–United States, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1857-1859.
- Afshar Y, Gaw SL, Flaherman VJ, et al. Clinical presentation of coronavirus disease 2019 (COVID-19) in pregnant and recently pregnant people. Obstet Gynecol. 2020;128:1117-1125.
- Cosma S, Carosso AR, Cusato J, et al. Coronavirus disease 2019 and first-trimester spontaneous abortion: a casecontrol study of 225 pregnant patients. Am J Obstet Gynecol. 2020;S0002-9378:31177-7. doi: 10.1016/j.ajog.2020.10.005.
- Prabhu M, Cagino K, Matthews KC, et al. Pregnancy and postpartum outcomes in a universally tested population for SARS-CoV-2 in New York City: a prospective cohort study. BJOG. 2020;127:1548-1556.
- Adhikari E, Moreno W, Zofkie AC, et al. Pregnancy outcomes among women with and without severe acute respiratory syndrome coronavirus 2 infection. JAMA Netw Open. 2020;3:e2029256.
- Knight M, Bunch K, Vousden B, et al; UK Obstetric Suveillance System SARS-CoV-2 Infection in Pregnancy Collaborative Group. Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107.
- Emeruwa UN, Ona S, Shaman JL, et al. Associations between built environment, neighborhood socioeconomic status, and SARS-CoV-2 infection among pregnant women in New York City. JAMA. 2020;324:390-392.
- Emeruwa UN, Spiegelman J, Ona S, et al. Influence of race and ethnicity on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection rates and clinical outcomes in pregnancy. Obstet Gynecol. 2020;126:1040-1043.
- Zambrano LD, Ellington S, Strid P, et al; CDC COVID-19 response pregnancy and infant linked outcomes team. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status–United States, January 22-October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647.
- Badr DA, Mattern J, Carlin A, et al. Are clinical outcomes worse for pregnant women at ≥20 weeks’ gestation infected with coronavirus disease 2019? A multicenter case control study with propensity score matching. Am J Obstet Gynecol. 2020;223:764-768.
- DeBolt CA, Bianco A, Limaye MA, et al. Pregnant women with severe or critical COVID-19 have increased composite morbidity compared with nonpregnant matched controls. Am J Obstet Gynecol. 2020;S0002-9378:31312-0.
- Collin J, Byström E, Carnahan A, et al. Public Health Agency of Sweden’s Brief Report: pregnant and postpartum women with severe acute respiratory syndrome coronavirus 2 infection in intensive care in Sweden. Acta Obstet Gynecol Scand. 2020;99: 819-822.
- Dumitriu D, Emeruwa UN, Hanft E, et al. Outcomes of neonates born to mothers with severe acute respiratory syndrome coronavirus 2 infection at a large medical center in New York City. JAMA Pediatr. 2020;e204298.
- Salvatore CM, Han JY, Acker KP, et al. Neonatal management and outcomes during the COVID-19 pandemic: an observational cohort study. Lancet Child Adolesc Health. 2020;4: 721-727.
- Shanes ED, Mithal LB, Otero S, et al. Placental pathology in COVID-19. Am J Clin Path. 2020;154:23-32.
- Centers for Disease Control and Prevention. Duration of isolation and precautions for adults with COVID-19. Updated October 19, 2020. https://www.cdc.gov/corona virus/2019-ncov/hcp/duration-isolation.html?CDC _AA_refVal=https%3A%2F%2Fwww.cdc.gov%2F coronavirus%2F2019-ncov%2Fcommunity%2Fstrategy -discontinue-isolation.html. Accessed December 15, 2020.
- Centers for Disease Control and Prevention. Discontinuation of transmission-based precautions and disposition of patients with COVID-19 in healthcare settings. Updated August 10, 2020. https://www.cdc.gov /coronavirus/2019-ncov/hcp/disposition-hospitalized -patients.html. Accessed December 15, 2020.
- Rasmussen SA, Lyerly AD, Jamieson DJ. Delaying pregnancy during a public health crisis–examining public health recommendations for COVID-19 and beyond. N Engl J Med. 2020;383:2097-2099.
- Reale SC, Field KG, Lumbreras-Marquez MI, et al. Association between number of in-person health care visits and SARS-CoV-2 infection in obstetrical patients. JAMA. 2020;324: 1210-1212.
- Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT 162b2 mRNA Covid-19 vaccine. N Engl J Med. December 10, 2020. doi: 10.1056/NEJMoa2034577.
- Widge AT, Rouphael NG, Jackson LA, et al. Durability of responses after SARS-CoV-2 mRNA-1273 vaccination. December 3, 2020. doi: 10.1056/NEJMc2032195.
- US Food and Drug Administration. FDA takes additional action in fight against COVID-19 by issuing emergency use authorization for second COVID-19 vaccine. December 18, 2020. https://www.fda.gov/news-events/press-announcements /fda-takes-additional-action-fight-against-covid-19-issuing -emergency-use-authorization-second-covid. Accessed December 22, 2020.
- American College of Obstetricians and Gynecologists. Practice advisory: vaccinating pregnancy and lactating patients against COVID-19. https://www.acog.org/clinical/clinical -guidance/practice-advisory/articles/2020/12/vaccinating -pregnant-and-lactating-patients-against-covid-19. Last updated December 21, 2020. Accessed December 21, 2020.
- Dooling K, McClung N, Chamberland M, et al. The Advisory Committee on Immunization Practices’ interim recommendation for allocating initial supplies of COVID-19 vaccine–United States, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1857-1859.
FDA warns about risk for false negatives from Curative COVID test
which is being used in Los Angeles and other large metropolitan areas in the United States.
The real-time reverse transcription polymerase chain reaction (PCR) test was developed by Menlo Park, Calif.–based health care start-up Curative. Results are analyzed by the company’s clinical lab, KorvaLabs. The test, which is authorized for prescription use only, received emergency-use authorization from the FDA on April 16, 2020. By Nov. 9, the company had processed 6 million test results, according to the company.
The FDA alert cautions that false negative results from any COVID-19 test can lead to delays in or the lack of supportive treatment and increase the risk for viral spread.
To mitigate the risk for false negatives, the agency advises clinicians to perform the Curative test as described in the product’s Fact Sheet for Healthcare Providers. This includes limiting its use to people who have had COVID-19 symptoms for 14 days or less. “Consider retesting your patients using a different test if you suspect an inaccurate result was given recently by the Curative SARS-Cov-2 test,” the FDA alert stated. “If testing was performed more than 2 weeks ago, and there is no reason to suspect current SARS-CoV-2 infection, it is not necessary to retest.”
The alert also notes that a negative result from the Curative PCR test “does not rule out COVID-19 and should not be used as the sole basis for treatment or patient management decisions. A negative result does not exclude the possibility of COVID-19.”
According to a press release issued by Curative on Oct. 7, its PCR test is being used by the Department of Defense, as well as the states of Alaska, California, Colorado, Delaware, Florida, Georgia (Atlanta and Savannah), Illinois (Chicago), Louisiana, Texas, and Wyoming. The company also operates Clinical Laboratory Improvement Amendments–certified laboratories in San Dimas, Calif.; Washington, D.C.; and Pflugerville, Tex.
A version of this article first appeared on Medscape.com.
which is being used in Los Angeles and other large metropolitan areas in the United States.
The real-time reverse transcription polymerase chain reaction (PCR) test was developed by Menlo Park, Calif.–based health care start-up Curative. Results are analyzed by the company’s clinical lab, KorvaLabs. The test, which is authorized for prescription use only, received emergency-use authorization from the FDA on April 16, 2020. By Nov. 9, the company had processed 6 million test results, according to the company.
The FDA alert cautions that false negative results from any COVID-19 test can lead to delays in or the lack of supportive treatment and increase the risk for viral spread.
To mitigate the risk for false negatives, the agency advises clinicians to perform the Curative test as described in the product’s Fact Sheet for Healthcare Providers. This includes limiting its use to people who have had COVID-19 symptoms for 14 days or less. “Consider retesting your patients using a different test if you suspect an inaccurate result was given recently by the Curative SARS-Cov-2 test,” the FDA alert stated. “If testing was performed more than 2 weeks ago, and there is no reason to suspect current SARS-CoV-2 infection, it is not necessary to retest.”
The alert also notes that a negative result from the Curative PCR test “does not rule out COVID-19 and should not be used as the sole basis for treatment or patient management decisions. A negative result does not exclude the possibility of COVID-19.”
According to a press release issued by Curative on Oct. 7, its PCR test is being used by the Department of Defense, as well as the states of Alaska, California, Colorado, Delaware, Florida, Georgia (Atlanta and Savannah), Illinois (Chicago), Louisiana, Texas, and Wyoming. The company also operates Clinical Laboratory Improvement Amendments–certified laboratories in San Dimas, Calif.; Washington, D.C.; and Pflugerville, Tex.
A version of this article first appeared on Medscape.com.
which is being used in Los Angeles and other large metropolitan areas in the United States.
The real-time reverse transcription polymerase chain reaction (PCR) test was developed by Menlo Park, Calif.–based health care start-up Curative. Results are analyzed by the company’s clinical lab, KorvaLabs. The test, which is authorized for prescription use only, received emergency-use authorization from the FDA on April 16, 2020. By Nov. 9, the company had processed 6 million test results, according to the company.
The FDA alert cautions that false negative results from any COVID-19 test can lead to delays in or the lack of supportive treatment and increase the risk for viral spread.
To mitigate the risk for false negatives, the agency advises clinicians to perform the Curative test as described in the product’s Fact Sheet for Healthcare Providers. This includes limiting its use to people who have had COVID-19 symptoms for 14 days or less. “Consider retesting your patients using a different test if you suspect an inaccurate result was given recently by the Curative SARS-Cov-2 test,” the FDA alert stated. “If testing was performed more than 2 weeks ago, and there is no reason to suspect current SARS-CoV-2 infection, it is not necessary to retest.”
The alert also notes that a negative result from the Curative PCR test “does not rule out COVID-19 and should not be used as the sole basis for treatment or patient management decisions. A negative result does not exclude the possibility of COVID-19.”
According to a press release issued by Curative on Oct. 7, its PCR test is being used by the Department of Defense, as well as the states of Alaska, California, Colorado, Delaware, Florida, Georgia (Atlanta and Savannah), Illinois (Chicago), Louisiana, Texas, and Wyoming. The company also operates Clinical Laboratory Improvement Amendments–certified laboratories in San Dimas, Calif.; Washington, D.C.; and Pflugerville, Tex.
A version of this article first appeared on Medscape.com.
Social isolation at the time of social distancing
Implications of loneliness and suggested management strategies in hospitalized patients with COVID-19
During a busy morning of rounds, our patient, Mrs. M., appeared distraught. She was diagnosed with COVID-19 2 weeks prior and remained inpatient because of medicosocial reasons. Since admission she remained on the same ward, in the same room, cared for by the same group of providers donned in masks, gowns, gloves, and face shields. The personal protective equipment helped to shield us from the virus, but it also shielded Mrs. M. from us.
During initial interaction, Mrs. M. appeared anxious, tearful, and detached. It seemed that she recognized a new voice; however, she did not express much interest in engaging during the visit. When she realized that she was not being discharged, Mrs. M. appeared to lose further interest. She wanted to go home. Her outpatient dialysis arrangements were not complete, and that precluded hospital discharge. Prescribed anxiolytics were doing little to relieve her symptoms.
The next day, Mrs. M. continued to ask if she could go home. She stated that there was nothing for her to do while in the hospital. She was tired of watching TV, she was unable to call her friends, and was not able to see her family. Because of COVID-19 status, Mrs. M was not permitted to leave her hospital room, and she was transported to the dialysis unit via stretcher, being unable to walk. The more we talked, the more engaged Mrs. M. had become. When it was time to complete the encounter, Mrs. M. started pleading with us to “stay a little longer, please don’t leave.”
Throughout her hospitalization, Mrs. M. had an extremely limited number of human encounters. Those encounters were fragmented and brief, centered on the infection mitigation. The chaplain was not permitted to enter her room, and she was unwilling to use the phone. The subspecialty consultants utilized telemedicine visits. As a result, Mrs. M. felt isolated and lonely. Social distancing in the hospital makes human interactions particularly challenging and contributes to the development of isolation, loneliness, and fear.
Loneliness is real
Loneliness is the “subjective experience of involuntary social isolation.”1 As the COVID-19 pandemic began to entrap the world in early 2020, many people have faced new challenges – loneliness and its impact on physical and mental health. The prevalence of loneliness nearly tripled in the early months of the pandemic, leading to psychological distress and reopening conversations on ethical issues.2
Ethical implications of loneliness
Social distancing challenges all four main ethical principles: autonomy, beneficence, nonmaleficence, and justice. How do we reconcile these principles from the standpoint of each affected individual, their caregivers, health care providers, and public health at large? How can we continue to mitigate the spread of COVID-19, but also remain attentive to our patients who are still in need of human interactions to recover and thrive?
Social distancing is important, but so is social interaction. What strategies do we have in place to combat loneliness? How do we help our hospitalized patients who feel connected to the “outside world?” Is battling loneliness worth the risks of additional exposure to COVID-19? These dilemmas cannot be easily resolved. However, it is important for us to recognize the negative impacts of loneliness and identify measures to help our patients.
In our mission to fulfill the beneficence and nonmaleficence principles of caring for patients affected by COVID-19, patients like Mrs. M. lose much of their autonomy during hospital admission. Despite our best efforts, our isolated patients during the pandemic, remain alone, which further heightens their feeling of loneliness.
Clinical implications of loneliness
With the advancements in technology, our capabilities to substitute personal human interactions have grown exponentially. The use of telemedicine, video- and audio-conferencing communications have changed the landscape of our capacities to exchange information. This could be a blessing and a curse. While the use of digital platforms for virtual communication is tempting, we should preserve human interactions as much as possible, particularly when caring for patients affected by COVID-19. Interpersonal “connectedness” plays a crucial role in providing psychological and psychotherapeutic support, particularly when the number of human encounters is already limited.
Social distancing requirements have magnified loneliness. Several studies demonstrate that the perception of loneliness leads to poor health outcomes, including lower immunity, increased peripheral vascular resistance,3 and higher overall mortality.4 Loneliness can lead to functional impairment, such as poor social skills, and even increased inflammation.5 The negative emotional impact of SARS-CoV-2 echoes the experiences of patients affected by the severe acute respiratory syndrome (SARS) outbreak in 2003. However, with COVID-19, we are witnessing the amplified effects of loneliness on a global scale. The majority of affected patients during the 2003 SARS outbreak in Canada reported loneliness, fear, aggression, and boredom: They had concerns about the impacts of the infection on loved ones, and psychological support was required for many patients with mild to moderate SARS disease.6
Nonpharmacological management strategies for battling loneliness
Utilization of early supportive services has been well described in literature and includes extending additional resources such as books, newspapers and, most importantly, additional in-person time to our patients.6 Maintaining rapport with patients’ families is also helpful in reducing anxiety and fear. The following measures have been suggested to prevent the negative impacts of loneliness and should be considered when caring for hospitalized patients diagnosed with COVID-19.7
- Screen patients for depression and delirium and utilize delirium prevention measures throughout the hospitalization.
- Educate patients about the signs and symptoms of loneliness, fear, and anxiety.
- Extend additional resources to patients, including books, magazines, and newspapers.
- Keep the patient’s cell or hospital phone within their reach.
- Adequately manage pain and prevent insomnia.
- Communicate frequently, utilizing audio- and visual-teleconferencing platforms that simultaneously include the patient and their loved ones.
- For patients who continue to exhibit feelings of loneliness despite the above interventions, consider consultations with psychiatry to offer additional coping strategies.
- Ensure a multidisciplinary approach when applicable – proactive consultation with the members of a palliative care team, ethics, spiritual health, social and ancillary services.
It is important to recognize how vulnerable our patients are. Diagnosed with COVID-19, and caught in the midst of the current pandemic, not only do they suffer from the physical effects of this novel disease, but they also have to endure prolonged confinement, social isolation, and uncertainty – all wrapped in a cloak of loneliness and fear.
With our main focus being on the management of a largely unknown viral illness, patients’ personal experiences can be easily overlooked. It is vital for us as health care providers on the front lines to recognize, reflect, and reform to ease our patients’ journey through COVID-19.
Dr. Burklin is an assistant professor of medicine, division of hospital medicine, at the department of medicine, Emory University, Atlanta. Dr. Wiley is an assistant professor of medicine, division of infectious disease, at the department of Medicine, Emory University, Atlanta.
References
1. Schlomann A et al. Use of information and communication technology (ICT) devices among the oldest-old: Loneliness, anomie, and autonomy. Innov Aging. 2020 Jan 1;4(2):igz050.
2. McGinty E et al. Psychological distress and loneliness reported by U.S. adults in 2018 and April 2020. JAMA. 2020 Jun 3. doi: 10.1001/jama.2020.9740. 3. Wang J et al. Associations between loneliness and perceived social support and outcomes of mental health problems: A systematic review. BMC Psychiatry. 2018 May 29;18(1):156.
4. Luo Y et al. Loneliness, health, and mortality in old age: A national longitudinal study. Soc Sci Med. 2012 Mar;74(6):907-14.
5. Smith KJ et al. The association between loneliness, social isolation, and inflammation: A systematic review and meta-analysis. Neurosci Biobehav Rev. 2020 Feb 21; 112:519-41.
6. Maunder R et al. The immediate psychological and occupational impact of the 2003 SARS outbreak in a teaching hospital. CMAJ. 2003 May 13;168(10):1245-51.
7. Masi CM et al. A meta-analysis of interventions to reduce loneliness. Pers Soc Psychol Rev. 2011 Aug;15(3):219-66.
Implications of loneliness and suggested management strategies in hospitalized patients with COVID-19
Implications of loneliness and suggested management strategies in hospitalized patients with COVID-19
During a busy morning of rounds, our patient, Mrs. M., appeared distraught. She was diagnosed with COVID-19 2 weeks prior and remained inpatient because of medicosocial reasons. Since admission she remained on the same ward, in the same room, cared for by the same group of providers donned in masks, gowns, gloves, and face shields. The personal protective equipment helped to shield us from the virus, but it also shielded Mrs. M. from us.
During initial interaction, Mrs. M. appeared anxious, tearful, and detached. It seemed that she recognized a new voice; however, she did not express much interest in engaging during the visit. When she realized that she was not being discharged, Mrs. M. appeared to lose further interest. She wanted to go home. Her outpatient dialysis arrangements were not complete, and that precluded hospital discharge. Prescribed anxiolytics were doing little to relieve her symptoms.
The next day, Mrs. M. continued to ask if she could go home. She stated that there was nothing for her to do while in the hospital. She was tired of watching TV, she was unable to call her friends, and was not able to see her family. Because of COVID-19 status, Mrs. M was not permitted to leave her hospital room, and she was transported to the dialysis unit via stretcher, being unable to walk. The more we talked, the more engaged Mrs. M. had become. When it was time to complete the encounter, Mrs. M. started pleading with us to “stay a little longer, please don’t leave.”
Throughout her hospitalization, Mrs. M. had an extremely limited number of human encounters. Those encounters were fragmented and brief, centered on the infection mitigation. The chaplain was not permitted to enter her room, and she was unwilling to use the phone. The subspecialty consultants utilized telemedicine visits. As a result, Mrs. M. felt isolated and lonely. Social distancing in the hospital makes human interactions particularly challenging and contributes to the development of isolation, loneliness, and fear.
Loneliness is real
Loneliness is the “subjective experience of involuntary social isolation.”1 As the COVID-19 pandemic began to entrap the world in early 2020, many people have faced new challenges – loneliness and its impact on physical and mental health. The prevalence of loneliness nearly tripled in the early months of the pandemic, leading to psychological distress and reopening conversations on ethical issues.2
Ethical implications of loneliness
Social distancing challenges all four main ethical principles: autonomy, beneficence, nonmaleficence, and justice. How do we reconcile these principles from the standpoint of each affected individual, their caregivers, health care providers, and public health at large? How can we continue to mitigate the spread of COVID-19, but also remain attentive to our patients who are still in need of human interactions to recover and thrive?
Social distancing is important, but so is social interaction. What strategies do we have in place to combat loneliness? How do we help our hospitalized patients who feel connected to the “outside world?” Is battling loneliness worth the risks of additional exposure to COVID-19? These dilemmas cannot be easily resolved. However, it is important for us to recognize the negative impacts of loneliness and identify measures to help our patients.
In our mission to fulfill the beneficence and nonmaleficence principles of caring for patients affected by COVID-19, patients like Mrs. M. lose much of their autonomy during hospital admission. Despite our best efforts, our isolated patients during the pandemic, remain alone, which further heightens their feeling of loneliness.
Clinical implications of loneliness
With the advancements in technology, our capabilities to substitute personal human interactions have grown exponentially. The use of telemedicine, video- and audio-conferencing communications have changed the landscape of our capacities to exchange information. This could be a blessing and a curse. While the use of digital platforms for virtual communication is tempting, we should preserve human interactions as much as possible, particularly when caring for patients affected by COVID-19. Interpersonal “connectedness” plays a crucial role in providing psychological and psychotherapeutic support, particularly when the number of human encounters is already limited.
Social distancing requirements have magnified loneliness. Several studies demonstrate that the perception of loneliness leads to poor health outcomes, including lower immunity, increased peripheral vascular resistance,3 and higher overall mortality.4 Loneliness can lead to functional impairment, such as poor social skills, and even increased inflammation.5 The negative emotional impact of SARS-CoV-2 echoes the experiences of patients affected by the severe acute respiratory syndrome (SARS) outbreak in 2003. However, with COVID-19, we are witnessing the amplified effects of loneliness on a global scale. The majority of affected patients during the 2003 SARS outbreak in Canada reported loneliness, fear, aggression, and boredom: They had concerns about the impacts of the infection on loved ones, and psychological support was required for many patients with mild to moderate SARS disease.6
Nonpharmacological management strategies for battling loneliness
Utilization of early supportive services has been well described in literature and includes extending additional resources such as books, newspapers and, most importantly, additional in-person time to our patients.6 Maintaining rapport with patients’ families is also helpful in reducing anxiety and fear. The following measures have been suggested to prevent the negative impacts of loneliness and should be considered when caring for hospitalized patients diagnosed with COVID-19.7
- Screen patients for depression and delirium and utilize delirium prevention measures throughout the hospitalization.
- Educate patients about the signs and symptoms of loneliness, fear, and anxiety.
- Extend additional resources to patients, including books, magazines, and newspapers.
- Keep the patient’s cell or hospital phone within their reach.
- Adequately manage pain and prevent insomnia.
- Communicate frequently, utilizing audio- and visual-teleconferencing platforms that simultaneously include the patient and their loved ones.
- For patients who continue to exhibit feelings of loneliness despite the above interventions, consider consultations with psychiatry to offer additional coping strategies.
- Ensure a multidisciplinary approach when applicable – proactive consultation with the members of a palliative care team, ethics, spiritual health, social and ancillary services.
It is important to recognize how vulnerable our patients are. Diagnosed with COVID-19, and caught in the midst of the current pandemic, not only do they suffer from the physical effects of this novel disease, but they also have to endure prolonged confinement, social isolation, and uncertainty – all wrapped in a cloak of loneliness and fear.
With our main focus being on the management of a largely unknown viral illness, patients’ personal experiences can be easily overlooked. It is vital for us as health care providers on the front lines to recognize, reflect, and reform to ease our patients’ journey through COVID-19.
Dr. Burklin is an assistant professor of medicine, division of hospital medicine, at the department of medicine, Emory University, Atlanta. Dr. Wiley is an assistant professor of medicine, division of infectious disease, at the department of Medicine, Emory University, Atlanta.
References
1. Schlomann A et al. Use of information and communication technology (ICT) devices among the oldest-old: Loneliness, anomie, and autonomy. Innov Aging. 2020 Jan 1;4(2):igz050.
2. McGinty E et al. Psychological distress and loneliness reported by U.S. adults in 2018 and April 2020. JAMA. 2020 Jun 3. doi: 10.1001/jama.2020.9740. 3. Wang J et al. Associations between loneliness and perceived social support and outcomes of mental health problems: A systematic review. BMC Psychiatry. 2018 May 29;18(1):156.
4. Luo Y et al. Loneliness, health, and mortality in old age: A national longitudinal study. Soc Sci Med. 2012 Mar;74(6):907-14.
5. Smith KJ et al. The association between loneliness, social isolation, and inflammation: A systematic review and meta-analysis. Neurosci Biobehav Rev. 2020 Feb 21; 112:519-41.
6. Maunder R et al. The immediate psychological and occupational impact of the 2003 SARS outbreak in a teaching hospital. CMAJ. 2003 May 13;168(10):1245-51.
7. Masi CM et al. A meta-analysis of interventions to reduce loneliness. Pers Soc Psychol Rev. 2011 Aug;15(3):219-66.
During a busy morning of rounds, our patient, Mrs. M., appeared distraught. She was diagnosed with COVID-19 2 weeks prior and remained inpatient because of medicosocial reasons. Since admission she remained on the same ward, in the same room, cared for by the same group of providers donned in masks, gowns, gloves, and face shields. The personal protective equipment helped to shield us from the virus, but it also shielded Mrs. M. from us.
During initial interaction, Mrs. M. appeared anxious, tearful, and detached. It seemed that she recognized a new voice; however, she did not express much interest in engaging during the visit. When she realized that she was not being discharged, Mrs. M. appeared to lose further interest. She wanted to go home. Her outpatient dialysis arrangements were not complete, and that precluded hospital discharge. Prescribed anxiolytics were doing little to relieve her symptoms.
The next day, Mrs. M. continued to ask if she could go home. She stated that there was nothing for her to do while in the hospital. She was tired of watching TV, she was unable to call her friends, and was not able to see her family. Because of COVID-19 status, Mrs. M was not permitted to leave her hospital room, and she was transported to the dialysis unit via stretcher, being unable to walk. The more we talked, the more engaged Mrs. M. had become. When it was time to complete the encounter, Mrs. M. started pleading with us to “stay a little longer, please don’t leave.”
Throughout her hospitalization, Mrs. M. had an extremely limited number of human encounters. Those encounters were fragmented and brief, centered on the infection mitigation. The chaplain was not permitted to enter her room, and she was unwilling to use the phone. The subspecialty consultants utilized telemedicine visits. As a result, Mrs. M. felt isolated and lonely. Social distancing in the hospital makes human interactions particularly challenging and contributes to the development of isolation, loneliness, and fear.
Loneliness is real
Loneliness is the “subjective experience of involuntary social isolation.”1 As the COVID-19 pandemic began to entrap the world in early 2020, many people have faced new challenges – loneliness and its impact on physical and mental health. The prevalence of loneliness nearly tripled in the early months of the pandemic, leading to psychological distress and reopening conversations on ethical issues.2
Ethical implications of loneliness
Social distancing challenges all four main ethical principles: autonomy, beneficence, nonmaleficence, and justice. How do we reconcile these principles from the standpoint of each affected individual, their caregivers, health care providers, and public health at large? How can we continue to mitigate the spread of COVID-19, but also remain attentive to our patients who are still in need of human interactions to recover and thrive?
Social distancing is important, but so is social interaction. What strategies do we have in place to combat loneliness? How do we help our hospitalized patients who feel connected to the “outside world?” Is battling loneliness worth the risks of additional exposure to COVID-19? These dilemmas cannot be easily resolved. However, it is important for us to recognize the negative impacts of loneliness and identify measures to help our patients.
In our mission to fulfill the beneficence and nonmaleficence principles of caring for patients affected by COVID-19, patients like Mrs. M. lose much of their autonomy during hospital admission. Despite our best efforts, our isolated patients during the pandemic, remain alone, which further heightens their feeling of loneliness.
Clinical implications of loneliness
With the advancements in technology, our capabilities to substitute personal human interactions have grown exponentially. The use of telemedicine, video- and audio-conferencing communications have changed the landscape of our capacities to exchange information. This could be a blessing and a curse. While the use of digital platforms for virtual communication is tempting, we should preserve human interactions as much as possible, particularly when caring for patients affected by COVID-19. Interpersonal “connectedness” plays a crucial role in providing psychological and psychotherapeutic support, particularly when the number of human encounters is already limited.
Social distancing requirements have magnified loneliness. Several studies demonstrate that the perception of loneliness leads to poor health outcomes, including lower immunity, increased peripheral vascular resistance,3 and higher overall mortality.4 Loneliness can lead to functional impairment, such as poor social skills, and even increased inflammation.5 The negative emotional impact of SARS-CoV-2 echoes the experiences of patients affected by the severe acute respiratory syndrome (SARS) outbreak in 2003. However, with COVID-19, we are witnessing the amplified effects of loneliness on a global scale. The majority of affected patients during the 2003 SARS outbreak in Canada reported loneliness, fear, aggression, and boredom: They had concerns about the impacts of the infection on loved ones, and psychological support was required for many patients with mild to moderate SARS disease.6
Nonpharmacological management strategies for battling loneliness
Utilization of early supportive services has been well described in literature and includes extending additional resources such as books, newspapers and, most importantly, additional in-person time to our patients.6 Maintaining rapport with patients’ families is also helpful in reducing anxiety and fear. The following measures have been suggested to prevent the negative impacts of loneliness and should be considered when caring for hospitalized patients diagnosed with COVID-19.7
- Screen patients for depression and delirium and utilize delirium prevention measures throughout the hospitalization.
- Educate patients about the signs and symptoms of loneliness, fear, and anxiety.
- Extend additional resources to patients, including books, magazines, and newspapers.
- Keep the patient’s cell or hospital phone within their reach.
- Adequately manage pain and prevent insomnia.
- Communicate frequently, utilizing audio- and visual-teleconferencing platforms that simultaneously include the patient and their loved ones.
- For patients who continue to exhibit feelings of loneliness despite the above interventions, consider consultations with psychiatry to offer additional coping strategies.
- Ensure a multidisciplinary approach when applicable – proactive consultation with the members of a palliative care team, ethics, spiritual health, social and ancillary services.
It is important to recognize how vulnerable our patients are. Diagnosed with COVID-19, and caught in the midst of the current pandemic, not only do they suffer from the physical effects of this novel disease, but they also have to endure prolonged confinement, social isolation, and uncertainty – all wrapped in a cloak of loneliness and fear.
With our main focus being on the management of a largely unknown viral illness, patients’ personal experiences can be easily overlooked. It is vital for us as health care providers on the front lines to recognize, reflect, and reform to ease our patients’ journey through COVID-19.
Dr. Burklin is an assistant professor of medicine, division of hospital medicine, at the department of medicine, Emory University, Atlanta. Dr. Wiley is an assistant professor of medicine, division of infectious disease, at the department of Medicine, Emory University, Atlanta.
References
1. Schlomann A et al. Use of information and communication technology (ICT) devices among the oldest-old: Loneliness, anomie, and autonomy. Innov Aging. 2020 Jan 1;4(2):igz050.
2. McGinty E et al. Psychological distress and loneliness reported by U.S. adults in 2018 and April 2020. JAMA. 2020 Jun 3. doi: 10.1001/jama.2020.9740. 3. Wang J et al. Associations between loneliness and perceived social support and outcomes of mental health problems: A systematic review. BMC Psychiatry. 2018 May 29;18(1):156.
4. Luo Y et al. Loneliness, health, and mortality in old age: A national longitudinal study. Soc Sci Med. 2012 Mar;74(6):907-14.
5. Smith KJ et al. The association between loneliness, social isolation, and inflammation: A systematic review and meta-analysis. Neurosci Biobehav Rev. 2020 Feb 21; 112:519-41.
6. Maunder R et al. The immediate psychological and occupational impact of the 2003 SARS outbreak in a teaching hospital. CMAJ. 2003 May 13;168(10):1245-51.
7. Masi CM et al. A meta-analysis of interventions to reduce loneliness. Pers Soc Psychol Rev. 2011 Aug;15(3):219-66.
Pandemic packed a year of distress into 1 month
The first month of the coronavirus pandemic created almost as much psychological distress among American adults as they had experienced in the year before February 2019, according to the results of two representative surveys.
“The 30-day prevalence of SD [serious distress] in May 2020 did not differ from the past-year prevalence of SD assessed with the same instrument [the Kessler-6 distress scale] in February 2019. In other words, equal numbers of people experienced SD in 30-days during the pandemic as experienced SD over an entire year prior to the pandemic,” Joshua Breslau, PhD, and associates at the Rand Corporation wrote in Preventive Medicine.
In May of 2020, the prevalence of SD was 10.1% in the previous month among 1,870 adults aged 20 years and older who had participated in the two Rand American Life Panel surveys, the first occurring in February 2019. In that earlier poll, 10.9% of the 2,555 respondents said that they experienced SD in the worst month of the previous year, the investigators said.
The prevalence of overall psychological distress increased by 12.8% from February 2019 to May 2020, with increases higher among women (17.7%) than men (10.6%); adults under age 60 years, compared with those over 60 (see graph); and Hispanics, compared with other races/ethnicities. Disparities also were seen among income groups: Distress rose 10.2% for those earning over $100,000, compared with 15.4% for those making less than $35,000 and 18.2% for Americans earning between $35,000 and $60,000, the researchers reported.
A high level of stress in the prepandemic survey strongly predicted serious distress during the pandemic. “Risk for SD during the pandemic among those with SD during a year before the pandemic was almost 3 times higher than among those reporting mild/moderate distress and 15 times higher than among those reporting no/low distress during the prepandemic year,” they noted.
Distress levels often return to normal after a disaster, Dr. Breslau and associates pointed out, but “the pandemic’s influence on economic stressors, disruption of usual activities and subsequent effects on population health may continue for an extended period and affect different regions of the country at different points in time.”
SOURCE: Breslau J et al. Prev Med. 2020 Dec 31. doi: 10.1016/j.ypmed.2020.106362.
The first month of the coronavirus pandemic created almost as much psychological distress among American adults as they had experienced in the year before February 2019, according to the results of two representative surveys.

“The 30-day prevalence of SD [serious distress] in May 2020 did not differ from the past-year prevalence of SD assessed with the same instrument [the Kessler-6 distress scale] in February 2019. In other words, equal numbers of people experienced SD in 30-days during the pandemic as experienced SD over an entire year prior to the pandemic,” Joshua Breslau, PhD, and associates at the Rand Corporation wrote in Preventive Medicine.
In May of 2020, the prevalence of SD was 10.1% in the previous month among 1,870 adults aged 20 years and older who had participated in the two Rand American Life Panel surveys, the first occurring in February 2019. In that earlier poll, 10.9% of the 2,555 respondents said that they experienced SD in the worst month of the previous year, the investigators said.
The prevalence of overall psychological distress increased by 12.8% from February 2019 to May 2020, with increases higher among women (17.7%) than men (10.6%); adults under age 60 years, compared with those over 60 (see graph); and Hispanics, compared with other races/ethnicities. Disparities also were seen among income groups: Distress rose 10.2% for those earning over $100,000, compared with 15.4% for those making less than $35,000 and 18.2% for Americans earning between $35,000 and $60,000, the researchers reported.
A high level of stress in the prepandemic survey strongly predicted serious distress during the pandemic. “Risk for SD during the pandemic among those with SD during a year before the pandemic was almost 3 times higher than among those reporting mild/moderate distress and 15 times higher than among those reporting no/low distress during the prepandemic year,” they noted.
Distress levels often return to normal after a disaster, Dr. Breslau and associates pointed out, but “the pandemic’s influence on economic stressors, disruption of usual activities and subsequent effects on population health may continue for an extended period and affect different regions of the country at different points in time.”
SOURCE: Breslau J et al. Prev Med. 2020 Dec 31. doi: 10.1016/j.ypmed.2020.106362.
The first month of the coronavirus pandemic created almost as much psychological distress among American adults as they had experienced in the year before February 2019, according to the results of two representative surveys.

“The 30-day prevalence of SD [serious distress] in May 2020 did not differ from the past-year prevalence of SD assessed with the same instrument [the Kessler-6 distress scale] in February 2019. In other words, equal numbers of people experienced SD in 30-days during the pandemic as experienced SD over an entire year prior to the pandemic,” Joshua Breslau, PhD, and associates at the Rand Corporation wrote in Preventive Medicine.
In May of 2020, the prevalence of SD was 10.1% in the previous month among 1,870 adults aged 20 years and older who had participated in the two Rand American Life Panel surveys, the first occurring in February 2019. In that earlier poll, 10.9% of the 2,555 respondents said that they experienced SD in the worst month of the previous year, the investigators said.
The prevalence of overall psychological distress increased by 12.8% from February 2019 to May 2020, with increases higher among women (17.7%) than men (10.6%); adults under age 60 years, compared with those over 60 (see graph); and Hispanics, compared with other races/ethnicities. Disparities also were seen among income groups: Distress rose 10.2% for those earning over $100,000, compared with 15.4% for those making less than $35,000 and 18.2% for Americans earning between $35,000 and $60,000, the researchers reported.
A high level of stress in the prepandemic survey strongly predicted serious distress during the pandemic. “Risk for SD during the pandemic among those with SD during a year before the pandemic was almost 3 times higher than among those reporting mild/moderate distress and 15 times higher than among those reporting no/low distress during the prepandemic year,” they noted.
Distress levels often return to normal after a disaster, Dr. Breslau and associates pointed out, but “the pandemic’s influence on economic stressors, disruption of usual activities and subsequent effects on population health may continue for an extended period and affect different regions of the country at different points in time.”
SOURCE: Breslau J et al. Prev Med. 2020 Dec 31. doi: 10.1016/j.ypmed.2020.106362.
FROM PREVENTIVE MEDICINE
Microvascular injury of brain, olfactory bulb seen in COVID-19
new research suggests.
Postmortem MRI brain scans of 13 patients who died from COVID-19 showed abnormalities in 10 of the participants. Of these, nine showed punctate hyperintensities, “which represented areas of microvascular injury and fibrinogen leakage,” the investigators reported. Immunostaining also showed a thinning of the basal lamina in five of these patients.
Further analyses showed punctate hypointensities linked to congested blood vessels in 10 patients. These areas were “interpreted as microhemorrhages,” the researchers noted.
There was no evidence of viral infection, including SARS-CoV-2.
“These findings may inform the interpretation of changes observed on [MRI] of punctate hyperintensities and linear hypointensities in patients with COVID-19,” wrote Myoung-Hwa Lee, PhD, a research fellow at the National Institute of Neurological Disorders and Stroke, and colleagues. The findings were published online Dec. 30 in a “correspondence” piece in the New England Journal of Medicine.
Interpret with caution
The investigators examined brains from a convenience sample of 19 patients (mean age, 50 years), all of whom died from COVID-19 between March and July 2020.
An 11.7-tesla scanner was used to obtain magnetic resonance microscopy images for 13 of the patients. In order to scan the olfactory bulb, the scanner was set at a resolution of 25 mcm; for the brain, it was set at 100 mcm.
Chromogenic immunostaining was used to assess brain abnormalities found in 10 of the patients. Multiplex fluorescence imaging was also used for some of the patients.
For 18 study participants, a histopathological brain examination was performed. In the patients who also had medical histories available to the researchers, five had mild respiratory syndrome, four had acute respiratory distress syndrome, two had pulmonary embolism, one had delirium, and three had unknown symptoms.
The punctate hyperintensities found on magnetic resonance microscopy were also found on histopathological exam. Collagen IV immunostaining showed a thinning in the basal lamina of endothelial cells in these areas.
In addition to congested blood vessels, punctate hypointensities were linked to areas of fibrinogen leakage – but also to “relatively intact vasculature,” the investigators reported.
“There was minimal perivascular inflammation in the specimens examined, but there was no vascular occlusion,” they added.
SARS-CoV-2 was also not found in any of the participants. “It is possible that the virus was cleared by the time of death or that viral copy numbers were below the level of detection by our assays,” the researchers noted.
In 13 of the patients, hypertrophic astrocytes, macrophage infiltrates, and perivascular-activated microglia were found. Eight patients showed CD3+ and CD8+ T cells in spaces and lumens next to endothelial cells.
Finally, five patients showed activated microglia next to neurons. This is “suggestive of neuronophagia in the olfactory bulb, substantial nigra, dorsal motor nucleus of the vagal nerve, and the pre-Bötzinger complex in the medulla, which is involved in the generation of spontaneous rhythmic breathing,” wrote the investigators.
In summary, vascular pathology was found in 10 cases, perivascular infiltrates were present in 13 cases, acute ischemic hypoxic neurons were present in 6 cases, and changes suggestive of neuronophagia were present in 5 cases.
The researchers noted that, although the study findings may be helpful when interpreting brain changes on MRI scan in this patient population, availability of clinical information for the participants was limited.
Therefore, “no conclusions can be drawn in relation to neurologic features of COVID-19,” they wrote.
The study was funded by NINDS. Dr. Lee and all but one of the other investigators reported no relevant financial relationships; the remaining investigator reported having received grants from NINDS during the conduct of this study.
A version of this article first appeared on Medscape.com.
new research suggests.
Postmortem MRI brain scans of 13 patients who died from COVID-19 showed abnormalities in 10 of the participants. Of these, nine showed punctate hyperintensities, “which represented areas of microvascular injury and fibrinogen leakage,” the investigators reported. Immunostaining also showed a thinning of the basal lamina in five of these patients.
Further analyses showed punctate hypointensities linked to congested blood vessels in 10 patients. These areas were “interpreted as microhemorrhages,” the researchers noted.
There was no evidence of viral infection, including SARS-CoV-2.
“These findings may inform the interpretation of changes observed on [MRI] of punctate hyperintensities and linear hypointensities in patients with COVID-19,” wrote Myoung-Hwa Lee, PhD, a research fellow at the National Institute of Neurological Disorders and Stroke, and colleagues. The findings were published online Dec. 30 in a “correspondence” piece in the New England Journal of Medicine.
Interpret with caution
The investigators examined brains from a convenience sample of 19 patients (mean age, 50 years), all of whom died from COVID-19 between March and July 2020.
An 11.7-tesla scanner was used to obtain magnetic resonance microscopy images for 13 of the patients. In order to scan the olfactory bulb, the scanner was set at a resolution of 25 mcm; for the brain, it was set at 100 mcm.
Chromogenic immunostaining was used to assess brain abnormalities found in 10 of the patients. Multiplex fluorescence imaging was also used for some of the patients.
For 18 study participants, a histopathological brain examination was performed. In the patients who also had medical histories available to the researchers, five had mild respiratory syndrome, four had acute respiratory distress syndrome, two had pulmonary embolism, one had delirium, and three had unknown symptoms.
The punctate hyperintensities found on magnetic resonance microscopy were also found on histopathological exam. Collagen IV immunostaining showed a thinning in the basal lamina of endothelial cells in these areas.
In addition to congested blood vessels, punctate hypointensities were linked to areas of fibrinogen leakage – but also to “relatively intact vasculature,” the investigators reported.
“There was minimal perivascular inflammation in the specimens examined, but there was no vascular occlusion,” they added.
SARS-CoV-2 was also not found in any of the participants. “It is possible that the virus was cleared by the time of death or that viral copy numbers were below the level of detection by our assays,” the researchers noted.
In 13 of the patients, hypertrophic astrocytes, macrophage infiltrates, and perivascular-activated microglia were found. Eight patients showed CD3+ and CD8+ T cells in spaces and lumens next to endothelial cells.
Finally, five patients showed activated microglia next to neurons. This is “suggestive of neuronophagia in the olfactory bulb, substantial nigra, dorsal motor nucleus of the vagal nerve, and the pre-Bötzinger complex in the medulla, which is involved in the generation of spontaneous rhythmic breathing,” wrote the investigators.
In summary, vascular pathology was found in 10 cases, perivascular infiltrates were present in 13 cases, acute ischemic hypoxic neurons were present in 6 cases, and changes suggestive of neuronophagia were present in 5 cases.
The researchers noted that, although the study findings may be helpful when interpreting brain changes on MRI scan in this patient population, availability of clinical information for the participants was limited.
Therefore, “no conclusions can be drawn in relation to neurologic features of COVID-19,” they wrote.
The study was funded by NINDS. Dr. Lee and all but one of the other investigators reported no relevant financial relationships; the remaining investigator reported having received grants from NINDS during the conduct of this study.
A version of this article first appeared on Medscape.com.
new research suggests.
Postmortem MRI brain scans of 13 patients who died from COVID-19 showed abnormalities in 10 of the participants. Of these, nine showed punctate hyperintensities, “which represented areas of microvascular injury and fibrinogen leakage,” the investigators reported. Immunostaining also showed a thinning of the basal lamina in five of these patients.
Further analyses showed punctate hypointensities linked to congested blood vessels in 10 patients. These areas were “interpreted as microhemorrhages,” the researchers noted.
There was no evidence of viral infection, including SARS-CoV-2.
“These findings may inform the interpretation of changes observed on [MRI] of punctate hyperintensities and linear hypointensities in patients with COVID-19,” wrote Myoung-Hwa Lee, PhD, a research fellow at the National Institute of Neurological Disorders and Stroke, and colleagues. The findings were published online Dec. 30 in a “correspondence” piece in the New England Journal of Medicine.
Interpret with caution
The investigators examined brains from a convenience sample of 19 patients (mean age, 50 years), all of whom died from COVID-19 between March and July 2020.
An 11.7-tesla scanner was used to obtain magnetic resonance microscopy images for 13 of the patients. In order to scan the olfactory bulb, the scanner was set at a resolution of 25 mcm; for the brain, it was set at 100 mcm.
Chromogenic immunostaining was used to assess brain abnormalities found in 10 of the patients. Multiplex fluorescence imaging was also used for some of the patients.
For 18 study participants, a histopathological brain examination was performed. In the patients who also had medical histories available to the researchers, five had mild respiratory syndrome, four had acute respiratory distress syndrome, two had pulmonary embolism, one had delirium, and three had unknown symptoms.
The punctate hyperintensities found on magnetic resonance microscopy were also found on histopathological exam. Collagen IV immunostaining showed a thinning in the basal lamina of endothelial cells in these areas.
In addition to congested blood vessels, punctate hypointensities were linked to areas of fibrinogen leakage – but also to “relatively intact vasculature,” the investigators reported.
“There was minimal perivascular inflammation in the specimens examined, but there was no vascular occlusion,” they added.
SARS-CoV-2 was also not found in any of the participants. “It is possible that the virus was cleared by the time of death or that viral copy numbers were below the level of detection by our assays,” the researchers noted.
In 13 of the patients, hypertrophic astrocytes, macrophage infiltrates, and perivascular-activated microglia were found. Eight patients showed CD3+ and CD8+ T cells in spaces and lumens next to endothelial cells.
Finally, five patients showed activated microglia next to neurons. This is “suggestive of neuronophagia in the olfactory bulb, substantial nigra, dorsal motor nucleus of the vagal nerve, and the pre-Bötzinger complex in the medulla, which is involved in the generation of spontaneous rhythmic breathing,” wrote the investigators.
In summary, vascular pathology was found in 10 cases, perivascular infiltrates were present in 13 cases, acute ischemic hypoxic neurons were present in 6 cases, and changes suggestive of neuronophagia were present in 5 cases.
The researchers noted that, although the study findings may be helpful when interpreting brain changes on MRI scan in this patient population, availability of clinical information for the participants was limited.
Therefore, “no conclusions can be drawn in relation to neurologic features of COVID-19,” they wrote.
The study was funded by NINDS. Dr. Lee and all but one of the other investigators reported no relevant financial relationships; the remaining investigator reported having received grants from NINDS during the conduct of this study.
A version of this article first appeared on Medscape.com.