User login
All Aboard
Ten years ago, Stephen Jencks, MD, MPH, was hospitalized after taking a nasty spill and rupturing a kidney, breaking two ribs, and fracturing two transverse processes. The independent healthcare safety and quality consultant based in Baltimore still laughs ruefully at what happened next.
Dr. Jencks was stabilized and given OxyContin to treat his considerable pain, and then he was discharged—without his wife or another caregiver present, with a prescription for nothing more than Tylenol, and without any instructions on what to do if his condition worsened. Twelve hours after returning home, his pain re-emerged with such a vengeance that he experienced severe muscle spasms.
Dr. Jencks suspects his doctor was so focused on his ruptured kidney that pain management and follow-up fell by the wayside. “I am not an unassertive individual, so why didn’t I say something?” he asks. “The simple answer is that, at least for me, if I’m taking OxyContin, there are no problems. People tend not to be at the very top of their game when they’re on opioids and traumatized.”
He made it through the night at home and received better pain medication in the morning, but his experience, he says, “beautifully illustrates” the chronic problem of less-than-graceful transfers of care that can lead to unnecessary hospital readmissions. If it nearly happened to him, it can happen to anyone.
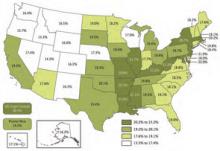
And, based on his research, it often does. In an influential 2009 New England Journal of Medicine study coauthored with Mark Williams, MD, FACP, FHM, professor and chief of the division of hospital medicine at Northwestern University’s Feinberg School of Medicine in Chicago, and principal investigator of SHM’s Project BOOST, and Eric Coleman, MD, MPH, FACP, associate professor of medicine and director of the care transitions program at the University of Colorado Denver, Dr. Jencks helped uncover some startling statistics: During a 15-month period from 2003 to 2004, nearly 20% of the roughly 12 million Medicare beneficiaries discharged from hospitals were readmitted within 30 days (see “State-by-State Breakdown of 30-Day Rehospitalizations of Medicare Beneficiaries,” p. 7).1 Of those patients discharged to the community and then rehospitalized, half had not seen their own primary-care physician (PCP) in the interim. In all, the authors estimated Medicare’s financial toll from unplanned rehospitalizations at $17.4 billion for 2004 alone.
Surprisingly, Dr. Jencks’ study and a 2007 Medicare Payment Advisory Commission report to Congress provided the first estimates of the overall burden of rehospitalization in nearly a quarter-century. Since then, however, the topic has been a mainstay in conversations about the kinds of interventions that could yield major improvements in healthcare.
“The thing that has propelled this to the front is the recognition that we really can do better,” Dr. Jencks says. “What had tended to be seen as just an evitable consequence of people being sick is now increasingly seen as often being the consequence of not having done as good a job as we should have.”
Beyond the potential for poor patient outcomes and wasted money, healthcare experts say excessive readmissions have the potential to undermine the reputations of hospitalists just as they are moving to center stage in national quality-improvement (QI) efforts.
“I see, basically every day, patients that come back to the hospital because the discharge process is broken,” says Eric Howell, MD, SFHM, director of the hospitalist division at Johns Hopkins Bayview Medical Center in Baltimore. Dr. Howell says communication difficulties between the hospital and a nursing home have plagued one “revolving door” case involving a patient with a stomach ulcer that requires surgical resection. Hospital surgeons have repeatedly arranged to see her as an outpatient and schedule the surgery, but before the surgery can take place, the patient vomits up blood and is rehospitalized.
Another contributing factor, Dr. Howell argues, is the lack of incentives for both hospitals and hospitalists to work hard at preventing the next readmission. Although Dr. Jencks’ study suggests readmissions might not always be profitable, Dr. Howell and others say the sizeable contribution of rehospitalizations to overall admission numbers and the single-digit profit margins of most hospitals offer little motivation to change the status quo. “I think there are good people who want to fix it,” says Dr. Howell, an SHM board member and Project BOOST mentor. But changing the reimbursement system so that hospitalists can better focus on reducing readmissions, he adds, “will really go a long way.”
A New Landscape
Change is in the air. As part of the federal Affordable Care Act of 2010, the Centers for Medicare & Medicaid Services (CMS) is expanding a pilot project on bundling payments to doctors and hospitals around episodes of care. Starting Jan. 1, 2013, the bundling pilot will define “episodes” as all medical services administered three days before a hospital admission until 30 days after discharge. A rehospitalization within that timeframe would net reduced reimbursements.
CMS also has begun accepting applications for what’s known as the Community-Based Care Transitions Program, with $500 million over five years authorized by the healthcare reform act to fund collaborative, readmission-reducing efforts between hospitals and community-based organizations. Linda Magno, CMS director of the Medicare Demonstrations Program Group (www.cms.gov/CMSLeadership/19_Office_ORDI.asp), says program participants will form a learning network so the agency can quickly deliver information about who’s doing well and what approaches are working better than others. The participating organizations, she says, can then help teach best practices to other hospitals around the country.
CMS has adopted public reporting requirements as another tactic. The “Hospital Compare” website (www.hospitalcompare.hhs.gov/) set up by CMS, for example, uses discharge data to publish rehospitalization rates for heart failure, acute myocardial infarction, and pneumonia. More published rates will be added soon. More importantly, Medicare will begin penalizing poorly performing institutions in October 2012 by withholding a percentage of their payments, starting at 1% and rising to 3% within three years, as part of the value-based purchasing initiative.
For hospitals, the looming deadline has prompted widespread concern about the potential financial impact. With a growing number of models and projects springing up around the country, however, hospitalists and other healthcare providers are finding encouraging signs that even relatively simple interventions might help profoundly change the trajectory of care transitions.
Rachel George, MD, MBA, FHM, regional medical director and vice president of operations for West Cogent Healthcare Inc., says Cogent has found success with one tactic—ensuring that all patients are called after being discharged. The call helps to verify that prescribed medications have been picked up and that other care-related questions have been answered. Even before discharge, Dr. George says, Cogent also tries to ensure that a follow-up appointment with every patient’s PCP is on the calendar.
Debbie White, project coordinator for the Little Rock, Ark.-based National Transitions of Care Coalition (NTOCC), says it helps to frame the entire process as a transition plan rather than a discharge. White says patients—and often their family caregivers—are the one constant in every transition. “Some older Americans, including the baby boomers, came from a culture where you don’t question your physician or even an RN,” White says. “So they’ve had a hard time speaking up and learning to ask for a list of their medications, or who’s going to make their next follow-up appointment.” Among its tools, NTOCC offers resources to teach patients how to take more responsibility for their own care (see “Patient Interaction,” p. 5).

On the other side of the equation, the most downloaded tool on the coalition’s website is an evaluation and implementation plan that helps healthcare professionals find the gaps in care transitions. Other tools, including case scenarios and checklists, help healthcare providers consider specific steps, and a compendium of evidence offers a look at successful models and projects.
Dr. Bradley M. Sherman, MD, FHM, chairman of the department of medicine at Glen Cove Hospital/North Shore-LIJ University Health System in New York, led one such project, sponsored by the Greater New York Hospital Association. Dr. Sherman targeted heart failure, the condition with the highest readmission rate for both Glen Cove Hospital and the North Shore/LIJ system. By placing special emphasis on medication compliance, dietary adherence, and physician follow-up, Dr. Sherman says, the hospital cut its readmission rates by more than half, to well below the national average.
Another effort led by Johns Hopkins’ Dr. Howell, known as Safe and Successful Transition of Elderly Patients (Safe STEP), used a collaborative staff approach in general medicine wards overseen by hospitalists to reduce 30-day readmission rates from 22% to 14%. The encouraging results, first reported at SHM’s annual meeting in 2008, provided the impetus for a project called Better Outcomes for Older Adults through Safe Transitions, or Project BOOST (www.hospitalmedicine.org/BOOST).
Developed by SHM, BOOST features a yearlong mentoring program to help sites implement the QI project. It began at six hospitals and has since spread to 62 active mentor sites. Enrollment may swell to between 100 and 120 sites by the end of 2011, according to project director Tina Budnitz, MPH. Data from the first phase revealed a 21% reduction in 30-day readmission rates at the six pilot sites, to 11.2% from 14.2%. Follow-up data from the larger cohort are expected this spring.
Eric Siegal, MD, SFHM, an SHM board member, past chair of SHM’s Public Policy Committee, and a clinical assistant professor of medicine at the University of Wisconsin School of Medicine and Public Health, says BOOST has benefited from being solidly in place at the right time, gaining momentum and garnering significant national attention as the focus on better care transitions has intensified.
“If BOOST demonstrates substantial and reproducible decreases in rehospitalizations, improvements in quality, and presumed projected cost reductions, I think that it’s going to go off like a bomb,” he says, “in a good way.”
Lakshmi Halasyamani, MD, SFHM, vice president for medical affairs for the Saint Joseph Mercy Health System in Michigan and an SHM board member, says BOOST encourages hospitalists to think about ways in which a discharge might fail. “And then we need to actively mitigate those risks,” she says.
National Collaborations
CMS has tapped a network of technical assistance and QI contractors in all 50 states, known as quality-improvement organizations (QIOs), for its own project addressing rehospitalizations. In 2008, these QIOs began working with communities in 14 states to implement what’s known as the Care Transitions Program.
The program has helped community leaders highlight three root causes of high readmission rates: patients’ lack of knowledge and understanding about their chronic conditions, lack of communication among providers, and the healthcare system’s lack of known standards.
The 14 communities, 70 hospitals, and 1.25 million Medicare beneficiaries being followed to date suggest that 30-day readmission rates can be significantly decreased, says Paul McGann, MD, CMS deputy chief medical officer. Preliminary data based on the number of readmissions per 1,000 Medicare beneficiaries, he says, show that participating communities have improved by an average of 4.7% over the first two years of the project, with the top performer improving 14% (for more information, visit www.cfmc.org/caretransitions).
Dr. Halasyamani says no single program has necessarily found the “secret sauce” to improve readmission rates across the board. “And we definitely haven’t figured out how to implement that in as cost-effective a way as possible,” she says.
But optimism is clearly building. With the initial focus on coaching low-performing institutions to improve their rates, Medicare could tap programs that demonstrate early promise as the main go-to teaching aids.
More importantly, hospitals around the country are finding what it takes to help their own patients.
“The question isn’t, ‘Is our number better than St. Elsewhere’s down the street?’ ” Dr. Jencks concludes. “The real question is, ‘Are there things we could reasonably have done for this patient and could do for the next patient that will keep this from happening to them?’ ” TH
Bryn Nelson is a freelance medical writer based in Seattle.
Reference
- Jencks SJ, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360(14):1418-1428.
Ten years ago, Stephen Jencks, MD, MPH, was hospitalized after taking a nasty spill and rupturing a kidney, breaking two ribs, and fracturing two transverse processes. The independent healthcare safety and quality consultant based in Baltimore still laughs ruefully at what happened next.
Dr. Jencks was stabilized and given OxyContin to treat his considerable pain, and then he was discharged—without his wife or another caregiver present, with a prescription for nothing more than Tylenol, and without any instructions on what to do if his condition worsened. Twelve hours after returning home, his pain re-emerged with such a vengeance that he experienced severe muscle spasms.
Dr. Jencks suspects his doctor was so focused on his ruptured kidney that pain management and follow-up fell by the wayside. “I am not an unassertive individual, so why didn’t I say something?” he asks. “The simple answer is that, at least for me, if I’m taking OxyContin, there are no problems. People tend not to be at the very top of their game when they’re on opioids and traumatized.”
He made it through the night at home and received better pain medication in the morning, but his experience, he says, “beautifully illustrates” the chronic problem of less-than-graceful transfers of care that can lead to unnecessary hospital readmissions. If it nearly happened to him, it can happen to anyone.
And, based on his research, it often does. In an influential 2009 New England Journal of Medicine study coauthored with Mark Williams, MD, FACP, FHM, professor and chief of the division of hospital medicine at Northwestern University’s Feinberg School of Medicine in Chicago, and principal investigator of SHM’s Project BOOST, and Eric Coleman, MD, MPH, FACP, associate professor of medicine and director of the care transitions program at the University of Colorado Denver, Dr. Jencks helped uncover some startling statistics: During a 15-month period from 2003 to 2004, nearly 20% of the roughly 12 million Medicare beneficiaries discharged from hospitals were readmitted within 30 days (see “State-by-State Breakdown of 30-Day Rehospitalizations of Medicare Beneficiaries,” p. 7).1 Of those patients discharged to the community and then rehospitalized, half had not seen their own primary-care physician (PCP) in the interim. In all, the authors estimated Medicare’s financial toll from unplanned rehospitalizations at $17.4 billion for 2004 alone.
Surprisingly, Dr. Jencks’ study and a 2007 Medicare Payment Advisory Commission report to Congress provided the first estimates of the overall burden of rehospitalization in nearly a quarter-century. Since then, however, the topic has been a mainstay in conversations about the kinds of interventions that could yield major improvements in healthcare.
“The thing that has propelled this to the front is the recognition that we really can do better,” Dr. Jencks says. “What had tended to be seen as just an evitable consequence of people being sick is now increasingly seen as often being the consequence of not having done as good a job as we should have.”
Beyond the potential for poor patient outcomes and wasted money, healthcare experts say excessive readmissions have the potential to undermine the reputations of hospitalists just as they are moving to center stage in national quality-improvement (QI) efforts.
“I see, basically every day, patients that come back to the hospital because the discharge process is broken,” says Eric Howell, MD, SFHM, director of the hospitalist division at Johns Hopkins Bayview Medical Center in Baltimore. Dr. Howell says communication difficulties between the hospital and a nursing home have plagued one “revolving door” case involving a patient with a stomach ulcer that requires surgical resection. Hospital surgeons have repeatedly arranged to see her as an outpatient and schedule the surgery, but before the surgery can take place, the patient vomits up blood and is rehospitalized.
Another contributing factor, Dr. Howell argues, is the lack of incentives for both hospitals and hospitalists to work hard at preventing the next readmission. Although Dr. Jencks’ study suggests readmissions might not always be profitable, Dr. Howell and others say the sizeable contribution of rehospitalizations to overall admission numbers and the single-digit profit margins of most hospitals offer little motivation to change the status quo. “I think there are good people who want to fix it,” says Dr. Howell, an SHM board member and Project BOOST mentor. But changing the reimbursement system so that hospitalists can better focus on reducing readmissions, he adds, “will really go a long way.”
A New Landscape
Change is in the air. As part of the federal Affordable Care Act of 2010, the Centers for Medicare & Medicaid Services (CMS) is expanding a pilot project on bundling payments to doctors and hospitals around episodes of care. Starting Jan. 1, 2013, the bundling pilot will define “episodes” as all medical services administered three days before a hospital admission until 30 days after discharge. A rehospitalization within that timeframe would net reduced reimbursements.
CMS also has begun accepting applications for what’s known as the Community-Based Care Transitions Program, with $500 million over five years authorized by the healthcare reform act to fund collaborative, readmission-reducing efforts between hospitals and community-based organizations. Linda Magno, CMS director of the Medicare Demonstrations Program Group (www.cms.gov/CMSLeadership/19_Office_ORDI.asp), says program participants will form a learning network so the agency can quickly deliver information about who’s doing well and what approaches are working better than others. The participating organizations, she says, can then help teach best practices to other hospitals around the country.
CMS has adopted public reporting requirements as another tactic. The “Hospital Compare” website (www.hospitalcompare.hhs.gov/) set up by CMS, for example, uses discharge data to publish rehospitalization rates for heart failure, acute myocardial infarction, and pneumonia. More published rates will be added soon. More importantly, Medicare will begin penalizing poorly performing institutions in October 2012 by withholding a percentage of their payments, starting at 1% and rising to 3% within three years, as part of the value-based purchasing initiative.
For hospitals, the looming deadline has prompted widespread concern about the potential financial impact. With a growing number of models and projects springing up around the country, however, hospitalists and other healthcare providers are finding encouraging signs that even relatively simple interventions might help profoundly change the trajectory of care transitions.
Rachel George, MD, MBA, FHM, regional medical director and vice president of operations for West Cogent Healthcare Inc., says Cogent has found success with one tactic—ensuring that all patients are called after being discharged. The call helps to verify that prescribed medications have been picked up and that other care-related questions have been answered. Even before discharge, Dr. George says, Cogent also tries to ensure that a follow-up appointment with every patient’s PCP is on the calendar.
Debbie White, project coordinator for the Little Rock, Ark.-based National Transitions of Care Coalition (NTOCC), says it helps to frame the entire process as a transition plan rather than a discharge. White says patients—and often their family caregivers—are the one constant in every transition. “Some older Americans, including the baby boomers, came from a culture where you don’t question your physician or even an RN,” White says. “So they’ve had a hard time speaking up and learning to ask for a list of their medications, or who’s going to make their next follow-up appointment.” Among its tools, NTOCC offers resources to teach patients how to take more responsibility for their own care (see “Patient Interaction,” p. 5).

On the other side of the equation, the most downloaded tool on the coalition’s website is an evaluation and implementation plan that helps healthcare professionals find the gaps in care transitions. Other tools, including case scenarios and checklists, help healthcare providers consider specific steps, and a compendium of evidence offers a look at successful models and projects.
Dr. Bradley M. Sherman, MD, FHM, chairman of the department of medicine at Glen Cove Hospital/North Shore-LIJ University Health System in New York, led one such project, sponsored by the Greater New York Hospital Association. Dr. Sherman targeted heart failure, the condition with the highest readmission rate for both Glen Cove Hospital and the North Shore/LIJ system. By placing special emphasis on medication compliance, dietary adherence, and physician follow-up, Dr. Sherman says, the hospital cut its readmission rates by more than half, to well below the national average.
Another effort led by Johns Hopkins’ Dr. Howell, known as Safe and Successful Transition of Elderly Patients (Safe STEP), used a collaborative staff approach in general medicine wards overseen by hospitalists to reduce 30-day readmission rates from 22% to 14%. The encouraging results, first reported at SHM’s annual meeting in 2008, provided the impetus for a project called Better Outcomes for Older Adults through Safe Transitions, or Project BOOST (www.hospitalmedicine.org/BOOST).
Developed by SHM, BOOST features a yearlong mentoring program to help sites implement the QI project. It began at six hospitals and has since spread to 62 active mentor sites. Enrollment may swell to between 100 and 120 sites by the end of 2011, according to project director Tina Budnitz, MPH. Data from the first phase revealed a 21% reduction in 30-day readmission rates at the six pilot sites, to 11.2% from 14.2%. Follow-up data from the larger cohort are expected this spring.
Eric Siegal, MD, SFHM, an SHM board member, past chair of SHM’s Public Policy Committee, and a clinical assistant professor of medicine at the University of Wisconsin School of Medicine and Public Health, says BOOST has benefited from being solidly in place at the right time, gaining momentum and garnering significant national attention as the focus on better care transitions has intensified.
“If BOOST demonstrates substantial and reproducible decreases in rehospitalizations, improvements in quality, and presumed projected cost reductions, I think that it’s going to go off like a bomb,” he says, “in a good way.”
Lakshmi Halasyamani, MD, SFHM, vice president for medical affairs for the Saint Joseph Mercy Health System in Michigan and an SHM board member, says BOOST encourages hospitalists to think about ways in which a discharge might fail. “And then we need to actively mitigate those risks,” she says.
National Collaborations
CMS has tapped a network of technical assistance and QI contractors in all 50 states, known as quality-improvement organizations (QIOs), for its own project addressing rehospitalizations. In 2008, these QIOs began working with communities in 14 states to implement what’s known as the Care Transitions Program.
The program has helped community leaders highlight three root causes of high readmission rates: patients’ lack of knowledge and understanding about their chronic conditions, lack of communication among providers, and the healthcare system’s lack of known standards.
The 14 communities, 70 hospitals, and 1.25 million Medicare beneficiaries being followed to date suggest that 30-day readmission rates can be significantly decreased, says Paul McGann, MD, CMS deputy chief medical officer. Preliminary data based on the number of readmissions per 1,000 Medicare beneficiaries, he says, show that participating communities have improved by an average of 4.7% over the first two years of the project, with the top performer improving 14% (for more information, visit www.cfmc.org/caretransitions).
Dr. Halasyamani says no single program has necessarily found the “secret sauce” to improve readmission rates across the board. “And we definitely haven’t figured out how to implement that in as cost-effective a way as possible,” she says.
But optimism is clearly building. With the initial focus on coaching low-performing institutions to improve their rates, Medicare could tap programs that demonstrate early promise as the main go-to teaching aids.
More importantly, hospitals around the country are finding what it takes to help their own patients.
“The question isn’t, ‘Is our number better than St. Elsewhere’s down the street?’ ” Dr. Jencks concludes. “The real question is, ‘Are there things we could reasonably have done for this patient and could do for the next patient that will keep this from happening to them?’ ” TH
Bryn Nelson is a freelance medical writer based in Seattle.
Reference
- Jencks SJ, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360(14):1418-1428.
Ten years ago, Stephen Jencks, MD, MPH, was hospitalized after taking a nasty spill and rupturing a kidney, breaking two ribs, and fracturing two transverse processes. The independent healthcare safety and quality consultant based in Baltimore still laughs ruefully at what happened next.
Dr. Jencks was stabilized and given OxyContin to treat his considerable pain, and then he was discharged—without his wife or another caregiver present, with a prescription for nothing more than Tylenol, and without any instructions on what to do if his condition worsened. Twelve hours after returning home, his pain re-emerged with such a vengeance that he experienced severe muscle spasms.
Dr. Jencks suspects his doctor was so focused on his ruptured kidney that pain management and follow-up fell by the wayside. “I am not an unassertive individual, so why didn’t I say something?” he asks. “The simple answer is that, at least for me, if I’m taking OxyContin, there are no problems. People tend not to be at the very top of their game when they’re on opioids and traumatized.”
He made it through the night at home and received better pain medication in the morning, but his experience, he says, “beautifully illustrates” the chronic problem of less-than-graceful transfers of care that can lead to unnecessary hospital readmissions. If it nearly happened to him, it can happen to anyone.
And, based on his research, it often does. In an influential 2009 New England Journal of Medicine study coauthored with Mark Williams, MD, FACP, FHM, professor and chief of the division of hospital medicine at Northwestern University’s Feinberg School of Medicine in Chicago, and principal investigator of SHM’s Project BOOST, and Eric Coleman, MD, MPH, FACP, associate professor of medicine and director of the care transitions program at the University of Colorado Denver, Dr. Jencks helped uncover some startling statistics: During a 15-month period from 2003 to 2004, nearly 20% of the roughly 12 million Medicare beneficiaries discharged from hospitals were readmitted within 30 days (see “State-by-State Breakdown of 30-Day Rehospitalizations of Medicare Beneficiaries,” p. 7).1 Of those patients discharged to the community and then rehospitalized, half had not seen their own primary-care physician (PCP) in the interim. In all, the authors estimated Medicare’s financial toll from unplanned rehospitalizations at $17.4 billion for 2004 alone.
Surprisingly, Dr. Jencks’ study and a 2007 Medicare Payment Advisory Commission report to Congress provided the first estimates of the overall burden of rehospitalization in nearly a quarter-century. Since then, however, the topic has been a mainstay in conversations about the kinds of interventions that could yield major improvements in healthcare.
“The thing that has propelled this to the front is the recognition that we really can do better,” Dr. Jencks says. “What had tended to be seen as just an evitable consequence of people being sick is now increasingly seen as often being the consequence of not having done as good a job as we should have.”
Beyond the potential for poor patient outcomes and wasted money, healthcare experts say excessive readmissions have the potential to undermine the reputations of hospitalists just as they are moving to center stage in national quality-improvement (QI) efforts.
“I see, basically every day, patients that come back to the hospital because the discharge process is broken,” says Eric Howell, MD, SFHM, director of the hospitalist division at Johns Hopkins Bayview Medical Center in Baltimore. Dr. Howell says communication difficulties between the hospital and a nursing home have plagued one “revolving door” case involving a patient with a stomach ulcer that requires surgical resection. Hospital surgeons have repeatedly arranged to see her as an outpatient and schedule the surgery, but before the surgery can take place, the patient vomits up blood and is rehospitalized.
Another contributing factor, Dr. Howell argues, is the lack of incentives for both hospitals and hospitalists to work hard at preventing the next readmission. Although Dr. Jencks’ study suggests readmissions might not always be profitable, Dr. Howell and others say the sizeable contribution of rehospitalizations to overall admission numbers and the single-digit profit margins of most hospitals offer little motivation to change the status quo. “I think there are good people who want to fix it,” says Dr. Howell, an SHM board member and Project BOOST mentor. But changing the reimbursement system so that hospitalists can better focus on reducing readmissions, he adds, “will really go a long way.”
A New Landscape
Change is in the air. As part of the federal Affordable Care Act of 2010, the Centers for Medicare & Medicaid Services (CMS) is expanding a pilot project on bundling payments to doctors and hospitals around episodes of care. Starting Jan. 1, 2013, the bundling pilot will define “episodes” as all medical services administered three days before a hospital admission until 30 days after discharge. A rehospitalization within that timeframe would net reduced reimbursements.
CMS also has begun accepting applications for what’s known as the Community-Based Care Transitions Program, with $500 million over five years authorized by the healthcare reform act to fund collaborative, readmission-reducing efforts between hospitals and community-based organizations. Linda Magno, CMS director of the Medicare Demonstrations Program Group (www.cms.gov/CMSLeadership/19_Office_ORDI.asp), says program participants will form a learning network so the agency can quickly deliver information about who’s doing well and what approaches are working better than others. The participating organizations, she says, can then help teach best practices to other hospitals around the country.
CMS has adopted public reporting requirements as another tactic. The “Hospital Compare” website (www.hospitalcompare.hhs.gov/) set up by CMS, for example, uses discharge data to publish rehospitalization rates for heart failure, acute myocardial infarction, and pneumonia. More published rates will be added soon. More importantly, Medicare will begin penalizing poorly performing institutions in October 2012 by withholding a percentage of their payments, starting at 1% and rising to 3% within three years, as part of the value-based purchasing initiative.
For hospitals, the looming deadline has prompted widespread concern about the potential financial impact. With a growing number of models and projects springing up around the country, however, hospitalists and other healthcare providers are finding encouraging signs that even relatively simple interventions might help profoundly change the trajectory of care transitions.
Rachel George, MD, MBA, FHM, regional medical director and vice president of operations for West Cogent Healthcare Inc., says Cogent has found success with one tactic—ensuring that all patients are called after being discharged. The call helps to verify that prescribed medications have been picked up and that other care-related questions have been answered. Even before discharge, Dr. George says, Cogent also tries to ensure that a follow-up appointment with every patient’s PCP is on the calendar.
Debbie White, project coordinator for the Little Rock, Ark.-based National Transitions of Care Coalition (NTOCC), says it helps to frame the entire process as a transition plan rather than a discharge. White says patients—and often their family caregivers—are the one constant in every transition. “Some older Americans, including the baby boomers, came from a culture where you don’t question your physician or even an RN,” White says. “So they’ve had a hard time speaking up and learning to ask for a list of their medications, or who’s going to make their next follow-up appointment.” Among its tools, NTOCC offers resources to teach patients how to take more responsibility for their own care (see “Patient Interaction,” p. 5).

On the other side of the equation, the most downloaded tool on the coalition’s website is an evaluation and implementation plan that helps healthcare professionals find the gaps in care transitions. Other tools, including case scenarios and checklists, help healthcare providers consider specific steps, and a compendium of evidence offers a look at successful models and projects.
Dr. Bradley M. Sherman, MD, FHM, chairman of the department of medicine at Glen Cove Hospital/North Shore-LIJ University Health System in New York, led one such project, sponsored by the Greater New York Hospital Association. Dr. Sherman targeted heart failure, the condition with the highest readmission rate for both Glen Cove Hospital and the North Shore/LIJ system. By placing special emphasis on medication compliance, dietary adherence, and physician follow-up, Dr. Sherman says, the hospital cut its readmission rates by more than half, to well below the national average.
Another effort led by Johns Hopkins’ Dr. Howell, known as Safe and Successful Transition of Elderly Patients (Safe STEP), used a collaborative staff approach in general medicine wards overseen by hospitalists to reduce 30-day readmission rates from 22% to 14%. The encouraging results, first reported at SHM’s annual meeting in 2008, provided the impetus for a project called Better Outcomes for Older Adults through Safe Transitions, or Project BOOST (www.hospitalmedicine.org/BOOST).
Developed by SHM, BOOST features a yearlong mentoring program to help sites implement the QI project. It began at six hospitals and has since spread to 62 active mentor sites. Enrollment may swell to between 100 and 120 sites by the end of 2011, according to project director Tina Budnitz, MPH. Data from the first phase revealed a 21% reduction in 30-day readmission rates at the six pilot sites, to 11.2% from 14.2%. Follow-up data from the larger cohort are expected this spring.
Eric Siegal, MD, SFHM, an SHM board member, past chair of SHM’s Public Policy Committee, and a clinical assistant professor of medicine at the University of Wisconsin School of Medicine and Public Health, says BOOST has benefited from being solidly in place at the right time, gaining momentum and garnering significant national attention as the focus on better care transitions has intensified.
“If BOOST demonstrates substantial and reproducible decreases in rehospitalizations, improvements in quality, and presumed projected cost reductions, I think that it’s going to go off like a bomb,” he says, “in a good way.”
Lakshmi Halasyamani, MD, SFHM, vice president for medical affairs for the Saint Joseph Mercy Health System in Michigan and an SHM board member, says BOOST encourages hospitalists to think about ways in which a discharge might fail. “And then we need to actively mitigate those risks,” she says.
National Collaborations
CMS has tapped a network of technical assistance and QI contractors in all 50 states, known as quality-improvement organizations (QIOs), for its own project addressing rehospitalizations. In 2008, these QIOs began working with communities in 14 states to implement what’s known as the Care Transitions Program.
The program has helped community leaders highlight three root causes of high readmission rates: patients’ lack of knowledge and understanding about their chronic conditions, lack of communication among providers, and the healthcare system’s lack of known standards.
The 14 communities, 70 hospitals, and 1.25 million Medicare beneficiaries being followed to date suggest that 30-day readmission rates can be significantly decreased, says Paul McGann, MD, CMS deputy chief medical officer. Preliminary data based on the number of readmissions per 1,000 Medicare beneficiaries, he says, show that participating communities have improved by an average of 4.7% over the first two years of the project, with the top performer improving 14% (for more information, visit www.cfmc.org/caretransitions).
Dr. Halasyamani says no single program has necessarily found the “secret sauce” to improve readmission rates across the board. “And we definitely haven’t figured out how to implement that in as cost-effective a way as possible,” she says.
But optimism is clearly building. With the initial focus on coaching low-performing institutions to improve their rates, Medicare could tap programs that demonstrate early promise as the main go-to teaching aids.
More importantly, hospitals around the country are finding what it takes to help their own patients.
“The question isn’t, ‘Is our number better than St. Elsewhere’s down the street?’ ” Dr. Jencks concludes. “The real question is, ‘Are there things we could reasonably have done for this patient and could do for the next patient that will keep this from happening to them?’ ” TH
Bryn Nelson is a freelance medical writer based in Seattle.
Reference
- Jencks SJ, Williams MV, Coleman EA. Rehospitalizations among patients in the Medicare fee-for-service program. N Engl J Med. 2009;360(14):1418-1428.
ONLINE EXCLUSIVE: Listen to Stephen Jencks, MD, and hospitalist Brad Sherman, MD, FHM, chat about readmission problems and solutions
Listen to Stephen Jencks, MD, MPH, discuss why the issue of hospital readmissions has caught the attention of doctors and professional societies.
Listen to veteran hospitalist Bradley Sherman, MD, FHM, chairman of the Department of Medicine at Glen Cove Hospital, part of the North Shore/LIJ Health System in New York, talk about what you can do both now and over the next year to help your institutions decrease readmission rates.
Listen to Stephen Jencks, MD, MPH, discuss why the issue of hospital readmissions has caught the attention of doctors and professional societies.
Listen to veteran hospitalist Bradley Sherman, MD, FHM, chairman of the Department of Medicine at Glen Cove Hospital, part of the North Shore/LIJ Health System in New York, talk about what you can do both now and over the next year to help your institutions decrease readmission rates.
Listen to Stephen Jencks, MD, MPH, discuss why the issue of hospital readmissions has caught the attention of doctors and professional societies.
Listen to veteran hospitalist Bradley Sherman, MD, FHM, chairman of the Department of Medicine at Glen Cove Hospital, part of the North Shore/LIJ Health System in New York, talk about what you can do both now and over the next year to help your institutions decrease readmission rates.
ONLINE EXCLUSIVE: The Exception or the Rule? Targeting the Right Patient Populations
Which patients are you most likely to see again? It’s a particularly vexing question for hospitalists amid the heightened focus on lowering hospital readmissions, and one that several recent studies have sought to address.
One Journal of Hospital Medicine analysis of more than 10,300 admissions found that unplanned rehospitalizations within 30 days of discharge were far more likely for African-American patients and those on high-risk medications like narcotics and corticosteroids.1 Patients with such chronic conditions as cancer, renal failure, and congestive heart failure also were at increased risk.
A second, smaller study of 142 inpatients who had been hospitalized within the preceding six months found that chronic disease, depression, and being underweight or obese all predicted a higher risk of another readmission within the next six months.2
And a third report in the Journal of Urban Health examined more than 36,000 Medicare patients admitted to urban public hospitals to assess which were most likely to return within the following year. Chronic medical conditions, substance abuse, and homelessness all contributed to increased odds.3

Most efforts aimed at reducing rehospitalizations, such as SHM’s Project BOOST, include a risk assessment that can point to potential trouble spots for individual patients. For certain populations, research has highlighted socioeconomic and racial disparities in access to healthcare that likely lead to unnecessary hospitalizations. But it’s one thing to identify the factors associated with higher rates, and quite another to actively manage them, especially when many crop up well beyond a hospital’s walls. Anxiety over these contributing factors is steadily building in anticipation of Medicare penalties for excessively high readmission rates set to begin in 2012.
“Whenever there is a program that has financial incentives, people always get concerned that they have patients who are somehow different,” says Lakshmi Halasyamani, MD, SFHM, SHM board member and vice president for medical affairs at Saint Joseph Mercy Health System in Ann Arbor, Mich. “Inherent in that assumption is: more difficult to manage or sicker or more complicated.”
Stephen Jencks, MD, MPH, an independent healthcare safety and quality consultant based in Baltimore, says he’s heard the same complaint for three decades. “It’s what we call the 'Lake Wobegon effect': All of our patients are sicker than average.
“I think it’s just a really poor way to go about what is a very human sort of question,” he adds. “If Mrs. Jones is back in the hospital because she didn’t understand the discharge instructions, the question is not ‘Does my population have more literacy problems than somebody else’s population of patients?’ The question is ‘What can we do for Mrs. Jones so she can understand this stuff?’ ” (For help communicating with patients, check out SHM's on-demand webinar, "Implementing Teach Back as a System-Wide Patient Communication Strategy.")
Healthcare experts say it’s not difficult to find challenges unique to particular urban areas or populations. Florida Hospital Association President Bruce Rueben, MBA, says many Floridians speak English as a second language, making clear communication critical. The state also has one of the highest percentages of elderly residents and is in a funding crisis that has required providers to do more with less. But instead of worrying about exceptions or anomalies, Rueben says, focusing on the best overall readmission-reducing approaches will help ensure that all patients are being treated and discharged effectively.
What about dealing with specific conditions? Paul McGann, MD, deputy chief medical officer at the Centers for Medicare & Medicaid Services (CMS), says good evidence exists for the effectiveness of interventions aimed at diseases ranging from congestive heart failure and cancer to chronic obstructive lung disease, ulcers, and stroke. But data from Medicare’s Care Transitions Program, he says, suggest that even if all hospitals pursued the dozens of disease-specific interventions collectively implemented by the program’s participants, they still wouldn’t address more than about half of the causes of readmission. Based on that finding, he says, project leaders have insisted on an all-cause focus.
Dr. Halasyamani says it’s only natural to sometimes focus on the exception rather than the rule. “And we’ve all had those experiences where, boy, you feel like you’ve done everything you can and the patient still comes back,” she says. “But having said that, we also have opportunities where we haven’t done everything that we can and the patient comes back. So I think we need to focus on that first, rather than say, ‘Well, this isn’t fixable based on all of the patient-level issues.’”
Rachel George, MD, MBA, FHM, regional medical director and vice president of operations for West Cogent Healthcare Inc., says it all comes down to perspective. “Instead of looking at what’s the percentage that we can’t deal with,” she says, “let’s look at the patient population that we can affect.”
Bryn Nelson is a freelance medical writer based in Seattle.
References
1. Allaudeen N, Vidyarthi A, Maselli J, Auerbach A. Redefining readmission risk factors for general medicine patients. J Hosp Med. 2011;6(2):54-60.
2. Mudge AM, Kasper KM, Clair, A, et al. Recurrent readmissions in medical patients: a prospective study. J Hosp Med. 2011;6(2):61-67.
3. Raven, MC, Billings, JC, Goldfrank LR, Manheimer ED, Gourevitch MN. Medicaid patients at high risk for frequent hospital admission: real-time identification and remediable risks. J Urb Health. 2009;86(2):230-241.
Which patients are you most likely to see again? It’s a particularly vexing question for hospitalists amid the heightened focus on lowering hospital readmissions, and one that several recent studies have sought to address.
One Journal of Hospital Medicine analysis of more than 10,300 admissions found that unplanned rehospitalizations within 30 days of discharge were far more likely for African-American patients and those on high-risk medications like narcotics and corticosteroids.1 Patients with such chronic conditions as cancer, renal failure, and congestive heart failure also were at increased risk.
A second, smaller study of 142 inpatients who had been hospitalized within the preceding six months found that chronic disease, depression, and being underweight or obese all predicted a higher risk of another readmission within the next six months.2
And a third report in the Journal of Urban Health examined more than 36,000 Medicare patients admitted to urban public hospitals to assess which were most likely to return within the following year. Chronic medical conditions, substance abuse, and homelessness all contributed to increased odds.3

Most efforts aimed at reducing rehospitalizations, such as SHM’s Project BOOST, include a risk assessment that can point to potential trouble spots for individual patients. For certain populations, research has highlighted socioeconomic and racial disparities in access to healthcare that likely lead to unnecessary hospitalizations. But it’s one thing to identify the factors associated with higher rates, and quite another to actively manage them, especially when many crop up well beyond a hospital’s walls. Anxiety over these contributing factors is steadily building in anticipation of Medicare penalties for excessively high readmission rates set to begin in 2012.
“Whenever there is a program that has financial incentives, people always get concerned that they have patients who are somehow different,” says Lakshmi Halasyamani, MD, SFHM, SHM board member and vice president for medical affairs at Saint Joseph Mercy Health System in Ann Arbor, Mich. “Inherent in that assumption is: more difficult to manage or sicker or more complicated.”
Stephen Jencks, MD, MPH, an independent healthcare safety and quality consultant based in Baltimore, says he’s heard the same complaint for three decades. “It’s what we call the 'Lake Wobegon effect': All of our patients are sicker than average.
“I think it’s just a really poor way to go about what is a very human sort of question,” he adds. “If Mrs. Jones is back in the hospital because she didn’t understand the discharge instructions, the question is not ‘Does my population have more literacy problems than somebody else’s population of patients?’ The question is ‘What can we do for Mrs. Jones so she can understand this stuff?’ ” (For help communicating with patients, check out SHM's on-demand webinar, "Implementing Teach Back as a System-Wide Patient Communication Strategy.")
Healthcare experts say it’s not difficult to find challenges unique to particular urban areas or populations. Florida Hospital Association President Bruce Rueben, MBA, says many Floridians speak English as a second language, making clear communication critical. The state also has one of the highest percentages of elderly residents and is in a funding crisis that has required providers to do more with less. But instead of worrying about exceptions or anomalies, Rueben says, focusing on the best overall readmission-reducing approaches will help ensure that all patients are being treated and discharged effectively.
What about dealing with specific conditions? Paul McGann, MD, deputy chief medical officer at the Centers for Medicare & Medicaid Services (CMS), says good evidence exists for the effectiveness of interventions aimed at diseases ranging from congestive heart failure and cancer to chronic obstructive lung disease, ulcers, and stroke. But data from Medicare’s Care Transitions Program, he says, suggest that even if all hospitals pursued the dozens of disease-specific interventions collectively implemented by the program’s participants, they still wouldn’t address more than about half of the causes of readmission. Based on that finding, he says, project leaders have insisted on an all-cause focus.
Dr. Halasyamani says it’s only natural to sometimes focus on the exception rather than the rule. “And we’ve all had those experiences where, boy, you feel like you’ve done everything you can and the patient still comes back,” she says. “But having said that, we also have opportunities where we haven’t done everything that we can and the patient comes back. So I think we need to focus on that first, rather than say, ‘Well, this isn’t fixable based on all of the patient-level issues.’”
Rachel George, MD, MBA, FHM, regional medical director and vice president of operations for West Cogent Healthcare Inc., says it all comes down to perspective. “Instead of looking at what’s the percentage that we can’t deal with,” she says, “let’s look at the patient population that we can affect.”
Bryn Nelson is a freelance medical writer based in Seattle.
References
1. Allaudeen N, Vidyarthi A, Maselli J, Auerbach A. Redefining readmission risk factors for general medicine patients. J Hosp Med. 2011;6(2):54-60.
2. Mudge AM, Kasper KM, Clair, A, et al. Recurrent readmissions in medical patients: a prospective study. J Hosp Med. 2011;6(2):61-67.
3. Raven, MC, Billings, JC, Goldfrank LR, Manheimer ED, Gourevitch MN. Medicaid patients at high risk for frequent hospital admission: real-time identification and remediable risks. J Urb Health. 2009;86(2):230-241.
Which patients are you most likely to see again? It’s a particularly vexing question for hospitalists amid the heightened focus on lowering hospital readmissions, and one that several recent studies have sought to address.
One Journal of Hospital Medicine analysis of more than 10,300 admissions found that unplanned rehospitalizations within 30 days of discharge were far more likely for African-American patients and those on high-risk medications like narcotics and corticosteroids.1 Patients with such chronic conditions as cancer, renal failure, and congestive heart failure also were at increased risk.
A second, smaller study of 142 inpatients who had been hospitalized within the preceding six months found that chronic disease, depression, and being underweight or obese all predicted a higher risk of another readmission within the next six months.2
And a third report in the Journal of Urban Health examined more than 36,000 Medicare patients admitted to urban public hospitals to assess which were most likely to return within the following year. Chronic medical conditions, substance abuse, and homelessness all contributed to increased odds.3

Most efforts aimed at reducing rehospitalizations, such as SHM’s Project BOOST, include a risk assessment that can point to potential trouble spots for individual patients. For certain populations, research has highlighted socioeconomic and racial disparities in access to healthcare that likely lead to unnecessary hospitalizations. But it’s one thing to identify the factors associated with higher rates, and quite another to actively manage them, especially when many crop up well beyond a hospital’s walls. Anxiety over these contributing factors is steadily building in anticipation of Medicare penalties for excessively high readmission rates set to begin in 2012.
“Whenever there is a program that has financial incentives, people always get concerned that they have patients who are somehow different,” says Lakshmi Halasyamani, MD, SFHM, SHM board member and vice president for medical affairs at Saint Joseph Mercy Health System in Ann Arbor, Mich. “Inherent in that assumption is: more difficult to manage or sicker or more complicated.”
Stephen Jencks, MD, MPH, an independent healthcare safety and quality consultant based in Baltimore, says he’s heard the same complaint for three decades. “It’s what we call the 'Lake Wobegon effect': All of our patients are sicker than average.
“I think it’s just a really poor way to go about what is a very human sort of question,” he adds. “If Mrs. Jones is back in the hospital because she didn’t understand the discharge instructions, the question is not ‘Does my population have more literacy problems than somebody else’s population of patients?’ The question is ‘What can we do for Mrs. Jones so she can understand this stuff?’ ” (For help communicating with patients, check out SHM's on-demand webinar, "Implementing Teach Back as a System-Wide Patient Communication Strategy.")
Healthcare experts say it’s not difficult to find challenges unique to particular urban areas or populations. Florida Hospital Association President Bruce Rueben, MBA, says many Floridians speak English as a second language, making clear communication critical. The state also has one of the highest percentages of elderly residents and is in a funding crisis that has required providers to do more with less. But instead of worrying about exceptions or anomalies, Rueben says, focusing on the best overall readmission-reducing approaches will help ensure that all patients are being treated and discharged effectively.
What about dealing with specific conditions? Paul McGann, MD, deputy chief medical officer at the Centers for Medicare & Medicaid Services (CMS), says good evidence exists for the effectiveness of interventions aimed at diseases ranging from congestive heart failure and cancer to chronic obstructive lung disease, ulcers, and stroke. But data from Medicare’s Care Transitions Program, he says, suggest that even if all hospitals pursued the dozens of disease-specific interventions collectively implemented by the program’s participants, they still wouldn’t address more than about half of the causes of readmission. Based on that finding, he says, project leaders have insisted on an all-cause focus.
Dr. Halasyamani says it’s only natural to sometimes focus on the exception rather than the rule. “And we’ve all had those experiences where, boy, you feel like you’ve done everything you can and the patient still comes back,” she says. “But having said that, we also have opportunities where we haven’t done everything that we can and the patient comes back. So I think we need to focus on that first, rather than say, ‘Well, this isn’t fixable based on all of the patient-level issues.’”
Rachel George, MD, MBA, FHM, regional medical director and vice president of operations for West Cogent Healthcare Inc., says it all comes down to perspective. “Instead of looking at what’s the percentage that we can’t deal with,” she says, “let’s look at the patient population that we can affect.”
Bryn Nelson is a freelance medical writer based in Seattle.
References
1. Allaudeen N, Vidyarthi A, Maselli J, Auerbach A. Redefining readmission risk factors for general medicine patients. J Hosp Med. 2011;6(2):54-60.
2. Mudge AM, Kasper KM, Clair, A, et al. Recurrent readmissions in medical patients: a prospective study. J Hosp Med. 2011;6(2):61-67.
3. Raven, MC, Billings, JC, Goldfrank LR, Manheimer ED, Gourevitch MN. Medicaid patients at high risk for frequent hospital admission: real-time identification and remediable risks. J Urb Health. 2009;86(2):230-241.
Infection-prevention professionals, hospital officials suggest steps to simplify and streamline HAI tracking system
Infection-prevention leaders and state hospital association representatives participated in regional meetings sponsored across the country during the summer of 2009 by the U.S. Department of Health and Human Services (HHS) and suggested ways that the Centers for Disease Control and Prevention’s (CDC) National Healthcare Safety Network (NHSN)—a leading federal system for tracking healthcare-associated infections (HAIs)—could be made easier for hospitals. Participants recommended steps to reduce data-collection burdens and to increase usefulness for hospital infection prevention and quality-improvement (QI) programs.
HHS convened the regional meetings to get stakeholders’ input into HHS’ Action Plan to Prevent Healthcare-Associated Infections (www.hhs.gov/ophs/initiatives/hai/index.html), as well as to hear about their experiences with the network. The action plan is a blueprint for HAI prevention and sets specific targets for monitoring and preventing HAIs nationally. Leaders from various HHS agencies, including the Agency for Healthcare Research and Quality (AHRQ), CDC, Centers for Medicare and Medicaid Services (CMS), the National Institutes of Health (NIH), and the Office of Public Health and Science joined me in discussing participants’ concerns at these meetings.
HHS uses NHSN data to help monitor progress toward the action plan goals. Twenty-eight states require hospitals to report HAIs publicly; most use the NHSN (www.cdc.gov/nhsn). Hospital enrollment in the NHSN has increased dramatically, to more than 2,700 hospitals in mid-2010 from 300 in 2005.
Daniel Pollock, MD, the surveillance branch chief for CDC’s Division of Healthcare Quality Promotion, and I reported on CDC’s efforts to update the NHSN and improve its ease of use. Simplifying and streamlining the system, and assuring sufficient technical capacity and user support, are top priorities. For example:
- CDC has instituted changes in data collection requirements for healthcare-associated urinary tract infections into the NHSN application;
- More NHSN staff were hired to perform comprehensive assessments and upgrades of the system’s technical infrastructure and usability, and to provide additional user support for enrollment and training;
- NHSN will begin migrating this fall to a new system of authenticating users; and
- NHSN Web pages are being redesigned to speed response times during peak use.
Dr. Pollock emphasized that the CDC is committed to accelerating the transition from manual to electronic case detection and reporting for the NHSN, and leveraging advances in health information technology as a primary strategy for enhancing the NHSN. As part of that effort, NHSN now is accepting electronic infection records submitted by hospitals that use commercial infection control surveillance systems.
The CDC continues to work closely with AHRQ, CMS, and the Office of the National Coordinator for Health Information Technology to coordinate integration efforts of federal information systems that provide HAI data. That is part of our commitment here in Washington to support hospitals’ efforts to reduce and eliminate HAIs, and to make healthcare safer for patients and families.
Don Wright, MD, MPH,
deputy assistant secretary for healthcare quality,
U.S. Department of Health and Human Services
Infection-prevention leaders and state hospital association representatives participated in regional meetings sponsored across the country during the summer of 2009 by the U.S. Department of Health and Human Services (HHS) and suggested ways that the Centers for Disease Control and Prevention’s (CDC) National Healthcare Safety Network (NHSN)—a leading federal system for tracking healthcare-associated infections (HAIs)—could be made easier for hospitals. Participants recommended steps to reduce data-collection burdens and to increase usefulness for hospital infection prevention and quality-improvement (QI) programs.
HHS convened the regional meetings to get stakeholders’ input into HHS’ Action Plan to Prevent Healthcare-Associated Infections (www.hhs.gov/ophs/initiatives/hai/index.html), as well as to hear about their experiences with the network. The action plan is a blueprint for HAI prevention and sets specific targets for monitoring and preventing HAIs nationally. Leaders from various HHS agencies, including the Agency for Healthcare Research and Quality (AHRQ), CDC, Centers for Medicare and Medicaid Services (CMS), the National Institutes of Health (NIH), and the Office of Public Health and Science joined me in discussing participants’ concerns at these meetings.
HHS uses NHSN data to help monitor progress toward the action plan goals. Twenty-eight states require hospitals to report HAIs publicly; most use the NHSN (www.cdc.gov/nhsn). Hospital enrollment in the NHSN has increased dramatically, to more than 2,700 hospitals in mid-2010 from 300 in 2005.
Daniel Pollock, MD, the surveillance branch chief for CDC’s Division of Healthcare Quality Promotion, and I reported on CDC’s efforts to update the NHSN and improve its ease of use. Simplifying and streamlining the system, and assuring sufficient technical capacity and user support, are top priorities. For example:
- CDC has instituted changes in data collection requirements for healthcare-associated urinary tract infections into the NHSN application;
- More NHSN staff were hired to perform comprehensive assessments and upgrades of the system’s technical infrastructure and usability, and to provide additional user support for enrollment and training;
- NHSN will begin migrating this fall to a new system of authenticating users; and
- NHSN Web pages are being redesigned to speed response times during peak use.
Dr. Pollock emphasized that the CDC is committed to accelerating the transition from manual to electronic case detection and reporting for the NHSN, and leveraging advances in health information technology as a primary strategy for enhancing the NHSN. As part of that effort, NHSN now is accepting electronic infection records submitted by hospitals that use commercial infection control surveillance systems.
The CDC continues to work closely with AHRQ, CMS, and the Office of the National Coordinator for Health Information Technology to coordinate integration efforts of federal information systems that provide HAI data. That is part of our commitment here in Washington to support hospitals’ efforts to reduce and eliminate HAIs, and to make healthcare safer for patients and families.
Don Wright, MD, MPH,
deputy assistant secretary for healthcare quality,
U.S. Department of Health and Human Services
Infection-prevention leaders and state hospital association representatives participated in regional meetings sponsored across the country during the summer of 2009 by the U.S. Department of Health and Human Services (HHS) and suggested ways that the Centers for Disease Control and Prevention’s (CDC) National Healthcare Safety Network (NHSN)—a leading federal system for tracking healthcare-associated infections (HAIs)—could be made easier for hospitals. Participants recommended steps to reduce data-collection burdens and to increase usefulness for hospital infection prevention and quality-improvement (QI) programs.
HHS convened the regional meetings to get stakeholders’ input into HHS’ Action Plan to Prevent Healthcare-Associated Infections (www.hhs.gov/ophs/initiatives/hai/index.html), as well as to hear about their experiences with the network. The action plan is a blueprint for HAI prevention and sets specific targets for monitoring and preventing HAIs nationally. Leaders from various HHS agencies, including the Agency for Healthcare Research and Quality (AHRQ), CDC, Centers for Medicare and Medicaid Services (CMS), the National Institutes of Health (NIH), and the Office of Public Health and Science joined me in discussing participants’ concerns at these meetings.
HHS uses NHSN data to help monitor progress toward the action plan goals. Twenty-eight states require hospitals to report HAIs publicly; most use the NHSN (www.cdc.gov/nhsn). Hospital enrollment in the NHSN has increased dramatically, to more than 2,700 hospitals in mid-2010 from 300 in 2005.
Daniel Pollock, MD, the surveillance branch chief for CDC’s Division of Healthcare Quality Promotion, and I reported on CDC’s efforts to update the NHSN and improve its ease of use. Simplifying and streamlining the system, and assuring sufficient technical capacity and user support, are top priorities. For example:
- CDC has instituted changes in data collection requirements for healthcare-associated urinary tract infections into the NHSN application;
- More NHSN staff were hired to perform comprehensive assessments and upgrades of the system’s technical infrastructure and usability, and to provide additional user support for enrollment and training;
- NHSN will begin migrating this fall to a new system of authenticating users; and
- NHSN Web pages are being redesigned to speed response times during peak use.
Dr. Pollock emphasized that the CDC is committed to accelerating the transition from manual to electronic case detection and reporting for the NHSN, and leveraging advances in health information technology as a primary strategy for enhancing the NHSN. As part of that effort, NHSN now is accepting electronic infection records submitted by hospitals that use commercial infection control surveillance systems.
The CDC continues to work closely with AHRQ, CMS, and the Office of the National Coordinator for Health Information Technology to coordinate integration efforts of federal information systems that provide HAI data. That is part of our commitment here in Washington to support hospitals’ efforts to reduce and eliminate HAIs, and to make healthcare safer for patients and families.
Don Wright, MD, MPH,
deputy assistant secretary for healthcare quality,
U.S. Department of Health and Human Services
Market Watch
New Generics
- Atomoxetine capsules (Strattera)1
- Clonidine transdermal system USP (catapres-TTS)2
- Enoxaparin sodium injection (Lovenox)3
- Naratriptan hydrochloride 2.5-mg tablets (Amerge)4
New Drugs, Devices, Indications, and Dosage Forms
- A combination tablet containing both aliskiren and amlodipine (Tekamlo) has been approved by the FDA for the treatment of hypertension (HTN).5 Four strengths for once-daily dosing are available.
- Antihemophilic factor VIII (recombinant) injection (Xyntha) for treatment of hemophilia A has been approved by the FDA in a pre-filled, dual-chamber syringe for intravenous (IV) infusion following reconstitution of the freeze-dried powder with 0.9% sodium chloride diluent (both supplied in the dosage form).6 The first dose will be available in the 3,000 international units strength (4 mL). Other dosages will be available in 2011.
- Bimatoprost ophthalmic solution 0.01% (Lumigan) has been approved by the FDA as a first-line treatment for reducing intraocular pressure in patients with open-angle glaucoma or ocular hypertension.7
- Buprenorphine/naloxone sublingual film (Suboxone sublingual) has been approved by the FDA for the treatment of opioid dependence.8
- Coagulation factor VIIa room temperature stable (recombinant) (NovoSeven RT) has been approved by the FDA in an 8-mg vial.9 This larger size allows rapid initiation and administration of this product for patients who need a larger dose. Additionally, this product is approved for an extended shelf life, for all vial sizes, to 36 months at room temperature.
- Donepezil 21-mg tablets (Aricept) have been approved by the FDA for the treatment of moderate to severe Alzheimer’s disease.10
- Glycopyrrolate cherry-flavored oral solution (Cuvposa) has been approved by the FDA as an orphan drug for treating chronic severe drooling in patients ages 3 to 16 with neurological conditions such as cerebral palsy.11
- Immune globulin subcutaneous (human) 20% liquid (Hizentra) has been approved by the FDA for a 24-month shelf life at room temperature when protected from light.12
- Miconazole 50-mg buccal tablets (Oravig) have been approved by the FDA to topically treat oropharyngeal candidiasis in patients 16 and older.13
- Niacin extended release/simvastatin tablets (Simcor) have been approved by the FDA in two new dosage strengths: 500 mg/40 mg and 1000 mg/40 mg.14
- Olmesartan medoxomil, amlodipine besylate, and hydrochlorothiazide tablets (HCTZ) (Tribenzor) have been approved by the FDA in a single tablet to treat hypertension (not initial therapy).15 This combination should not be used in patients with a creatinine clearance <30 mL/minute, or in patients with renal artery stenosis.
- STX-100 has received orphan drug status for treating idiopathic pulmonary fibrosis (IPF) for which there currently are no FDA-approved treatments.16 STX-100 is a humanized, monoclonal antibody that targets integrin vb6, which exhibited major antifibrotic activity in preclinical animal models of the lung and other organs. The FDA previously granted orphan drug designation for STX-100 for chronic allograft nephropathy. A Phase 2 trial in IPF is planned for 2011.
- Docetaxel injection concentrate (Taxotere) has been approved by the FDA in a one-vial system, which eliminates the dilution step.17
- Valganciclovir injection (Valcyte) is FDA-approved for an increased therapy length (200 days) in adult renal transplant patients at high risk of developing cytomegalovirus disease (CMV).18 This extends the length of therapy from 100 days.
Safety, Warnings, and Label Changes
- Carbidopa/levodopa and entacapone tablets (Stalevo) are undergoing a safety review in relation to a possible increased cardiovascular event risk, including myocardial infarction, stroke, and cardiovascular death, compared with patients only taking carbidopa/levodopa.19 An FDA meta-analysis of 15 clinical trials found a small increased risk of cardiovascular events. However, this meta-analysis was not specifically designed to assess cardiovascular safety, and most patients had pre-existing cardiovascular disease risk factors, so even small differences in the level of these risks could significantly affect the study outcomes. Additionally, most negative cardiovascular events occurred in a single trial. The FDA recommends regular evaluation of patients’ cardiovascular status.
- Tigecycline IV injection (Tygacil) has undergone a label change in its “Warnings” and “Precautions” in relation to an increased mortality risk.20 A pooled analysis compared tigecycline use to other similar antibacterials for managing different serious infections. Patients who had a greater increased risk of death were those with hospital-acquired pneumonia, ventilator-associated pneumonia, complicated skin and skin structure infections, diabetic foot infections, and complicated intra-abdominal infections.
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Actavis receives FDA approval of atomoxetine HCI capsules. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/199692.php. Accessed Sept. 2, 2010.
- Mylan receives approval for generic version of Catapres-TTS. Mylan Inc. website. Available at: http://investor.mylan.com/releasedetail.cfm?ReleaseID=489338. Accessed July 20, 2010.
- Riley K. FDA approves first generic enoxaparin sodium injection. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm220092.htm. Accessed Sept. 13, 2010.
- First-time generic approvals August 2010. Formulary website. Available at: http://formularyjournal.modernmedicine.com/formulary/Modern+Medicine+Now/First-time-generic-approvals-Aug.-2010/ArticleStandard/Article/detail/683182?contextCategoryId=44276. Accessed Sept. 7, 2010.
- Novartis receives FDA approval of Tekamlo, a single-pill combination of aliskiren and amlodipine to treat high blood pressure. Novartis Pharmaceuticals Corporation website. Available at: http://www.pharma.us.novartis.com/info/newsroom/press-release.jsp?PRID=2286. Accessed Sept. 13, 2010.
- Xyntha prefilled dual-chamber syringe approved for hemophilia A treatment. Monthly Prescribing Reference website. Available at: http://www.empr.com/xyntha-pre-filled-dual-chamber-syringe-approved-for-hemophilia-a-treatment/article/176666/. Accessed Aug.18, 2010.
- Allergan, Inc. receives FDA approval for Lumigan 0.01% as first-line therapy indicated for the reduction of elevated intraocular pressure in glaucoma patients. MarketWatch website. Available at: http://www.marketwatch.com/story/allergan-inc-receives-fda-approval-for-lumiganr-001-as-first-line-therapy-indicated-for-the-reduction-of-elevated-intraocular-pressure-in-glaucoma-patients-2010-08-31?reflink=MW_news_stmp. Accessed Sept. 2, 2010.
- MonoSol Rx announces Reckitt Benckiser FDA approval of Suboxone sublingual film for treatment of opioid dependence. PR Newswire website. Available at: http://www.prnewswire.com/news-releases/monosol-rx-announces-reckitt-benckiser-fda-approval-of-suboxone-sublingual-film-for-treatment-of-opioid-dependence-101874388.html. Accessed Sept. 14, 2010.
- NovoSeven RT 8mg vial approved for hemophilia A or B. Monthly Prescribing Reference website. Available at: http://www.empr.com/novoseven-rt-8mg-vial-approved-for-hemophilia-a-or-b/printarticle/176743/. Accessed Sept. 13, 2010.
- Eisai announces U.S. FDA approval for new higher dose Aricept 23 mg tablet for the treatment of moderate-to-severe Alzheimer’s disease. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/196410.php. Accessed Sept. 13, 2010.
- Cuvposa approved for chronic severe drooling associated with neurologic conditions. Monthly Prescribing Reference website. Available at: http://www.empr.com/cuvposa-approved-for-chronic-severe-drooling-associated-with-neurologic-conditions/article/175824/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Aug. 2, 2010.
- Shelf life of Hizentra extended from 18 to 24 months. Monthly Prescribing Reference website. Available at: http://www.empr.com/shelf-life-of-hizentra-extended-from-18-to-24-months/article/177068/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Aug. 23, 2010.
- Oravig available for oropharyngeal candidiasis. Monthly Prescribing Reference website. Available at: http://www.empr.com/oravig-available-for-oropharyngeal-candidiasis/article/177492/. Accessed Sept. 13, 2010.
- Additional dosage strengths of Simcor approved. Monthly Prescribing Reference website. Available at: http://www.empr.com/additional-dosage-strengths-of-simcor-approved/article/175825/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Sept. 13, 2010.
- Tribenzor 20/5/12.5mg. Monthly Prescribing Reference website. Available at: http://www.empr.com/tribenzor-205125mg/drugproduct/129/. Accessed Sept. 13, 2010.
- Stromedix receives FDA orphan drug designation for STX-100 for the treatment of idiopathic pulmonary fibrosis. Stromedix website. Available at: http://www.stromedix.com/Stromedix_STX-100_Orphan_Drug_IPF.pdf. Accessed Sept. 13, 2010.
- Dane L. Sanofi-Aventis garners FDA approval for one-vial formulation of Taxotere. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E0B4E517F06C4A9E94DC88EADBA079A8. Accessed Sept. 13, 2010.
- FDA approves longer use of Valcyte for adult kidney transplant patients at high risk of developing cytomegalovirus (CMV) disease. Genentech website. Available at: http://www.gene.com/gene/news/press-releases/display.do?method=detail&id=12907. Accessed Sept. 13, 2010.
- FDA drug safety communication: ongoing safety review of Stalevo and possible increased cardiovascular risk. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/ucm223060.htm. Accessed Sept. 13, 2010.
- FDA drug safety communication: increased risk of death with Tygacil (tigecycline) compared to other antibiotics used to treat similar infections. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/ucm224370.htm. Accessed Sept. 13, 2010.
New Generics
- Atomoxetine capsules (Strattera)1
- Clonidine transdermal system USP (catapres-TTS)2
- Enoxaparin sodium injection (Lovenox)3
- Naratriptan hydrochloride 2.5-mg tablets (Amerge)4
New Drugs, Devices, Indications, and Dosage Forms
- A combination tablet containing both aliskiren and amlodipine (Tekamlo) has been approved by the FDA for the treatment of hypertension (HTN).5 Four strengths for once-daily dosing are available.
- Antihemophilic factor VIII (recombinant) injection (Xyntha) for treatment of hemophilia A has been approved by the FDA in a pre-filled, dual-chamber syringe for intravenous (IV) infusion following reconstitution of the freeze-dried powder with 0.9% sodium chloride diluent (both supplied in the dosage form).6 The first dose will be available in the 3,000 international units strength (4 mL). Other dosages will be available in 2011.
- Bimatoprost ophthalmic solution 0.01% (Lumigan) has been approved by the FDA as a first-line treatment for reducing intraocular pressure in patients with open-angle glaucoma or ocular hypertension.7
- Buprenorphine/naloxone sublingual film (Suboxone sublingual) has been approved by the FDA for the treatment of opioid dependence.8
- Coagulation factor VIIa room temperature stable (recombinant) (NovoSeven RT) has been approved by the FDA in an 8-mg vial.9 This larger size allows rapid initiation and administration of this product for patients who need a larger dose. Additionally, this product is approved for an extended shelf life, for all vial sizes, to 36 months at room temperature.
- Donepezil 21-mg tablets (Aricept) have been approved by the FDA for the treatment of moderate to severe Alzheimer’s disease.10
- Glycopyrrolate cherry-flavored oral solution (Cuvposa) has been approved by the FDA as an orphan drug for treating chronic severe drooling in patients ages 3 to 16 with neurological conditions such as cerebral palsy.11
- Immune globulin subcutaneous (human) 20% liquid (Hizentra) has been approved by the FDA for a 24-month shelf life at room temperature when protected from light.12
- Miconazole 50-mg buccal tablets (Oravig) have been approved by the FDA to topically treat oropharyngeal candidiasis in patients 16 and older.13
- Niacin extended release/simvastatin tablets (Simcor) have been approved by the FDA in two new dosage strengths: 500 mg/40 mg and 1000 mg/40 mg.14
- Olmesartan medoxomil, amlodipine besylate, and hydrochlorothiazide tablets (HCTZ) (Tribenzor) have been approved by the FDA in a single tablet to treat hypertension (not initial therapy).15 This combination should not be used in patients with a creatinine clearance <30 mL/minute, or in patients with renal artery stenosis.
- STX-100 has received orphan drug status for treating idiopathic pulmonary fibrosis (IPF) for which there currently are no FDA-approved treatments.16 STX-100 is a humanized, monoclonal antibody that targets integrin vb6, which exhibited major antifibrotic activity in preclinical animal models of the lung and other organs. The FDA previously granted orphan drug designation for STX-100 for chronic allograft nephropathy. A Phase 2 trial in IPF is planned for 2011.
- Docetaxel injection concentrate (Taxotere) has been approved by the FDA in a one-vial system, which eliminates the dilution step.17
- Valganciclovir injection (Valcyte) is FDA-approved for an increased therapy length (200 days) in adult renal transplant patients at high risk of developing cytomegalovirus disease (CMV).18 This extends the length of therapy from 100 days.
Safety, Warnings, and Label Changes
- Carbidopa/levodopa and entacapone tablets (Stalevo) are undergoing a safety review in relation to a possible increased cardiovascular event risk, including myocardial infarction, stroke, and cardiovascular death, compared with patients only taking carbidopa/levodopa.19 An FDA meta-analysis of 15 clinical trials found a small increased risk of cardiovascular events. However, this meta-analysis was not specifically designed to assess cardiovascular safety, and most patients had pre-existing cardiovascular disease risk factors, so even small differences in the level of these risks could significantly affect the study outcomes. Additionally, most negative cardiovascular events occurred in a single trial. The FDA recommends regular evaluation of patients’ cardiovascular status.
- Tigecycline IV injection (Tygacil) has undergone a label change in its “Warnings” and “Precautions” in relation to an increased mortality risk.20 A pooled analysis compared tigecycline use to other similar antibacterials for managing different serious infections. Patients who had a greater increased risk of death were those with hospital-acquired pneumonia, ventilator-associated pneumonia, complicated skin and skin structure infections, diabetic foot infections, and complicated intra-abdominal infections.
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Actavis receives FDA approval of atomoxetine HCI capsules. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/199692.php. Accessed Sept. 2, 2010.
- Mylan receives approval for generic version of Catapres-TTS. Mylan Inc. website. Available at: http://investor.mylan.com/releasedetail.cfm?ReleaseID=489338. Accessed July 20, 2010.
- Riley K. FDA approves first generic enoxaparin sodium injection. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm220092.htm. Accessed Sept. 13, 2010.
- First-time generic approvals August 2010. Formulary website. Available at: http://formularyjournal.modernmedicine.com/formulary/Modern+Medicine+Now/First-time-generic-approvals-Aug.-2010/ArticleStandard/Article/detail/683182?contextCategoryId=44276. Accessed Sept. 7, 2010.
- Novartis receives FDA approval of Tekamlo, a single-pill combination of aliskiren and amlodipine to treat high blood pressure. Novartis Pharmaceuticals Corporation website. Available at: http://www.pharma.us.novartis.com/info/newsroom/press-release.jsp?PRID=2286. Accessed Sept. 13, 2010.
- Xyntha prefilled dual-chamber syringe approved for hemophilia A treatment. Monthly Prescribing Reference website. Available at: http://www.empr.com/xyntha-pre-filled-dual-chamber-syringe-approved-for-hemophilia-a-treatment/article/176666/. Accessed Aug.18, 2010.
- Allergan, Inc. receives FDA approval for Lumigan 0.01% as first-line therapy indicated for the reduction of elevated intraocular pressure in glaucoma patients. MarketWatch website. Available at: http://www.marketwatch.com/story/allergan-inc-receives-fda-approval-for-lumiganr-001-as-first-line-therapy-indicated-for-the-reduction-of-elevated-intraocular-pressure-in-glaucoma-patients-2010-08-31?reflink=MW_news_stmp. Accessed Sept. 2, 2010.
- MonoSol Rx announces Reckitt Benckiser FDA approval of Suboxone sublingual film for treatment of opioid dependence. PR Newswire website. Available at: http://www.prnewswire.com/news-releases/monosol-rx-announces-reckitt-benckiser-fda-approval-of-suboxone-sublingual-film-for-treatment-of-opioid-dependence-101874388.html. Accessed Sept. 14, 2010.
- NovoSeven RT 8mg vial approved for hemophilia A or B. Monthly Prescribing Reference website. Available at: http://www.empr.com/novoseven-rt-8mg-vial-approved-for-hemophilia-a-or-b/printarticle/176743/. Accessed Sept. 13, 2010.
- Eisai announces U.S. FDA approval for new higher dose Aricept 23 mg tablet for the treatment of moderate-to-severe Alzheimer’s disease. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/196410.php. Accessed Sept. 13, 2010.
- Cuvposa approved for chronic severe drooling associated with neurologic conditions. Monthly Prescribing Reference website. Available at: http://www.empr.com/cuvposa-approved-for-chronic-severe-drooling-associated-with-neurologic-conditions/article/175824/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Aug. 2, 2010.
- Shelf life of Hizentra extended from 18 to 24 months. Monthly Prescribing Reference website. Available at: http://www.empr.com/shelf-life-of-hizentra-extended-from-18-to-24-months/article/177068/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Aug. 23, 2010.
- Oravig available for oropharyngeal candidiasis. Monthly Prescribing Reference website. Available at: http://www.empr.com/oravig-available-for-oropharyngeal-candidiasis/article/177492/. Accessed Sept. 13, 2010.
- Additional dosage strengths of Simcor approved. Monthly Prescribing Reference website. Available at: http://www.empr.com/additional-dosage-strengths-of-simcor-approved/article/175825/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Sept. 13, 2010.
- Tribenzor 20/5/12.5mg. Monthly Prescribing Reference website. Available at: http://www.empr.com/tribenzor-205125mg/drugproduct/129/. Accessed Sept. 13, 2010.
- Stromedix receives FDA orphan drug designation for STX-100 for the treatment of idiopathic pulmonary fibrosis. Stromedix website. Available at: http://www.stromedix.com/Stromedix_STX-100_Orphan_Drug_IPF.pdf. Accessed Sept. 13, 2010.
- Dane L. Sanofi-Aventis garners FDA approval for one-vial formulation of Taxotere. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E0B4E517F06C4A9E94DC88EADBA079A8. Accessed Sept. 13, 2010.
- FDA approves longer use of Valcyte for adult kidney transplant patients at high risk of developing cytomegalovirus (CMV) disease. Genentech website. Available at: http://www.gene.com/gene/news/press-releases/display.do?method=detail&id=12907. Accessed Sept. 13, 2010.
- FDA drug safety communication: ongoing safety review of Stalevo and possible increased cardiovascular risk. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/ucm223060.htm. Accessed Sept. 13, 2010.
- FDA drug safety communication: increased risk of death with Tygacil (tigecycline) compared to other antibiotics used to treat similar infections. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/ucm224370.htm. Accessed Sept. 13, 2010.
New Generics
- Atomoxetine capsules (Strattera)1
- Clonidine transdermal system USP (catapres-TTS)2
- Enoxaparin sodium injection (Lovenox)3
- Naratriptan hydrochloride 2.5-mg tablets (Amerge)4
New Drugs, Devices, Indications, and Dosage Forms
- A combination tablet containing both aliskiren and amlodipine (Tekamlo) has been approved by the FDA for the treatment of hypertension (HTN).5 Four strengths for once-daily dosing are available.
- Antihemophilic factor VIII (recombinant) injection (Xyntha) for treatment of hemophilia A has been approved by the FDA in a pre-filled, dual-chamber syringe for intravenous (IV) infusion following reconstitution of the freeze-dried powder with 0.9% sodium chloride diluent (both supplied in the dosage form).6 The first dose will be available in the 3,000 international units strength (4 mL). Other dosages will be available in 2011.
- Bimatoprost ophthalmic solution 0.01% (Lumigan) has been approved by the FDA as a first-line treatment for reducing intraocular pressure in patients with open-angle glaucoma or ocular hypertension.7
- Buprenorphine/naloxone sublingual film (Suboxone sublingual) has been approved by the FDA for the treatment of opioid dependence.8
- Coagulation factor VIIa room temperature stable (recombinant) (NovoSeven RT) has been approved by the FDA in an 8-mg vial.9 This larger size allows rapid initiation and administration of this product for patients who need a larger dose. Additionally, this product is approved for an extended shelf life, for all vial sizes, to 36 months at room temperature.
- Donepezil 21-mg tablets (Aricept) have been approved by the FDA for the treatment of moderate to severe Alzheimer’s disease.10
- Glycopyrrolate cherry-flavored oral solution (Cuvposa) has been approved by the FDA as an orphan drug for treating chronic severe drooling in patients ages 3 to 16 with neurological conditions such as cerebral palsy.11
- Immune globulin subcutaneous (human) 20% liquid (Hizentra) has been approved by the FDA for a 24-month shelf life at room temperature when protected from light.12
- Miconazole 50-mg buccal tablets (Oravig) have been approved by the FDA to topically treat oropharyngeal candidiasis in patients 16 and older.13
- Niacin extended release/simvastatin tablets (Simcor) have been approved by the FDA in two new dosage strengths: 500 mg/40 mg and 1000 mg/40 mg.14
- Olmesartan medoxomil, amlodipine besylate, and hydrochlorothiazide tablets (HCTZ) (Tribenzor) have been approved by the FDA in a single tablet to treat hypertension (not initial therapy).15 This combination should not be used in patients with a creatinine clearance <30 mL/minute, or in patients with renal artery stenosis.
- STX-100 has received orphan drug status for treating idiopathic pulmonary fibrosis (IPF) for which there currently are no FDA-approved treatments.16 STX-100 is a humanized, monoclonal antibody that targets integrin vb6, which exhibited major antifibrotic activity in preclinical animal models of the lung and other organs. The FDA previously granted orphan drug designation for STX-100 for chronic allograft nephropathy. A Phase 2 trial in IPF is planned for 2011.
- Docetaxel injection concentrate (Taxotere) has been approved by the FDA in a one-vial system, which eliminates the dilution step.17
- Valganciclovir injection (Valcyte) is FDA-approved for an increased therapy length (200 days) in adult renal transplant patients at high risk of developing cytomegalovirus disease (CMV).18 This extends the length of therapy from 100 days.
Safety, Warnings, and Label Changes
- Carbidopa/levodopa and entacapone tablets (Stalevo) are undergoing a safety review in relation to a possible increased cardiovascular event risk, including myocardial infarction, stroke, and cardiovascular death, compared with patients only taking carbidopa/levodopa.19 An FDA meta-analysis of 15 clinical trials found a small increased risk of cardiovascular events. However, this meta-analysis was not specifically designed to assess cardiovascular safety, and most patients had pre-existing cardiovascular disease risk factors, so even small differences in the level of these risks could significantly affect the study outcomes. Additionally, most negative cardiovascular events occurred in a single trial. The FDA recommends regular evaluation of patients’ cardiovascular status.
- Tigecycline IV injection (Tygacil) has undergone a label change in its “Warnings” and “Precautions” in relation to an increased mortality risk.20 A pooled analysis compared tigecycline use to other similar antibacterials for managing different serious infections. Patients who had a greater increased risk of death were those with hospital-acquired pneumonia, ventilator-associated pneumonia, complicated skin and skin structure infections, diabetic foot infections, and complicated intra-abdominal infections.
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Actavis receives FDA approval of atomoxetine HCI capsules. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/199692.php. Accessed Sept. 2, 2010.
- Mylan receives approval for generic version of Catapres-TTS. Mylan Inc. website. Available at: http://investor.mylan.com/releasedetail.cfm?ReleaseID=489338. Accessed July 20, 2010.
- Riley K. FDA approves first generic enoxaparin sodium injection. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm220092.htm. Accessed Sept. 13, 2010.
- First-time generic approvals August 2010. Formulary website. Available at: http://formularyjournal.modernmedicine.com/formulary/Modern+Medicine+Now/First-time-generic-approvals-Aug.-2010/ArticleStandard/Article/detail/683182?contextCategoryId=44276. Accessed Sept. 7, 2010.
- Novartis receives FDA approval of Tekamlo, a single-pill combination of aliskiren and amlodipine to treat high blood pressure. Novartis Pharmaceuticals Corporation website. Available at: http://www.pharma.us.novartis.com/info/newsroom/press-release.jsp?PRID=2286. Accessed Sept. 13, 2010.
- Xyntha prefilled dual-chamber syringe approved for hemophilia A treatment. Monthly Prescribing Reference website. Available at: http://www.empr.com/xyntha-pre-filled-dual-chamber-syringe-approved-for-hemophilia-a-treatment/article/176666/. Accessed Aug.18, 2010.
- Allergan, Inc. receives FDA approval for Lumigan 0.01% as first-line therapy indicated for the reduction of elevated intraocular pressure in glaucoma patients. MarketWatch website. Available at: http://www.marketwatch.com/story/allergan-inc-receives-fda-approval-for-lumiganr-001-as-first-line-therapy-indicated-for-the-reduction-of-elevated-intraocular-pressure-in-glaucoma-patients-2010-08-31?reflink=MW_news_stmp. Accessed Sept. 2, 2010.
- MonoSol Rx announces Reckitt Benckiser FDA approval of Suboxone sublingual film for treatment of opioid dependence. PR Newswire website. Available at: http://www.prnewswire.com/news-releases/monosol-rx-announces-reckitt-benckiser-fda-approval-of-suboxone-sublingual-film-for-treatment-of-opioid-dependence-101874388.html. Accessed Sept. 14, 2010.
- NovoSeven RT 8mg vial approved for hemophilia A or B. Monthly Prescribing Reference website. Available at: http://www.empr.com/novoseven-rt-8mg-vial-approved-for-hemophilia-a-or-b/printarticle/176743/. Accessed Sept. 13, 2010.
- Eisai announces U.S. FDA approval for new higher dose Aricept 23 mg tablet for the treatment of moderate-to-severe Alzheimer’s disease. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/196410.php. Accessed Sept. 13, 2010.
- Cuvposa approved for chronic severe drooling associated with neurologic conditions. Monthly Prescribing Reference website. Available at: http://www.empr.com/cuvposa-approved-for-chronic-severe-drooling-associated-with-neurologic-conditions/article/175824/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Aug. 2, 2010.
- Shelf life of Hizentra extended from 18 to 24 months. Monthly Prescribing Reference website. Available at: http://www.empr.com/shelf-life-of-hizentra-extended-from-18-to-24-months/article/177068/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Aug. 23, 2010.
- Oravig available for oropharyngeal candidiasis. Monthly Prescribing Reference website. Available at: http://www.empr.com/oravig-available-for-oropharyngeal-candidiasis/article/177492/. Accessed Sept. 13, 2010.
- Additional dosage strengths of Simcor approved. Monthly Prescribing Reference website. Available at: http://www.empr.com/additional-dosage-strengths-of-simcor-approved/article/175825/?DCMP=EMC-MPR_WeeklyNewsbrief. Accessed Sept. 13, 2010.
- Tribenzor 20/5/12.5mg. Monthly Prescribing Reference website. Available at: http://www.empr.com/tribenzor-205125mg/drugproduct/129/. Accessed Sept. 13, 2010.
- Stromedix receives FDA orphan drug designation for STX-100 for the treatment of idiopathic pulmonary fibrosis. Stromedix website. Available at: http://www.stromedix.com/Stromedix_STX-100_Orphan_Drug_IPF.pdf. Accessed Sept. 13, 2010.
- Dane L. Sanofi-Aventis garners FDA approval for one-vial formulation of Taxotere. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E0B4E517F06C4A9E94DC88EADBA079A8. Accessed Sept. 13, 2010.
- FDA approves longer use of Valcyte for adult kidney transplant patients at high risk of developing cytomegalovirus (CMV) disease. Genentech website. Available at: http://www.gene.com/gene/news/press-releases/display.do?method=detail&id=12907. Accessed Sept. 13, 2010.
- FDA drug safety communication: ongoing safety review of Stalevo and possible increased cardiovascular risk. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/ucm223060.htm. Accessed Sept. 13, 2010.
- FDA drug safety communication: increased risk of death with Tygacil (tigecycline) compared to other antibiotics used to treat similar infections. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/ucm224370.htm. Accessed Sept. 13, 2010.
The Story of Us
From the outset, HM has been about efficiency. And there was nothing wrong with that, for value is quality divided by cost. But in our story, we found that mere efficiency was not enough: The lowering of the denominator (cost) had to be met with an escalation of the numerator (quality) to ensure value.
And see us as being born in the right place at the right time. For with the national focus turning to the need for quality and patient safety, hospital medicine was in the right place and the right time to heed the call to action: appropriately stepping up to enact efforts to make the slope of the line (on a chart of quality vs. cost) “STEEEPER” … finding systems innovations to make care Safe, Timely, Efficient, Effective, Equitable, and Patient-centered.
Of course, the story continued with the Affordable Care Act and healthcare reform, greatly accelerating our evolution as change agents. And now we find ourselves fully invested in a “change the system” mentality, perfectly positioned to meaningfully change healthcare for millions of people. But threats loom—specifically, the “R” in the STEEEPER mnemonic: the risks to quality in the face of healthcare reform.
So in the next chapter of our story, I present to you our challenge: how to overcome the threats to quality in the context of healthcare reform. The first three are presented here; in subsequent articles, I will address the remainder. Overcoming all threats will hinge on mastering the four truisms of cultural change:
- Systems drive function;
- Every system is perfectly designed to produce the outcomes that it does;
- This is not an issue of people needing to try harder; and
- The “no blame” culture begins with a paradigm shift from the “person at fault” to the “system at fault.”
Threat 1: Failure to Fund Quality
SHM elected to merge its annual State of Hospital Medicine survey with the MGMA. Though not without risk, this has resulted in the anticipated benefits. The MGMA collaboration brings greater leverage in working with the C-suite, which is pre-programmed to react to MGMA surveys. From the most recent MGMA survey comes good news: The financial compensation for hospitalists has increased. A sobering insight, however, is that this increase in compensation has been met with a corresponding increase in work intensity—RVUs. Further, the link between RVUs and compensation appears to be tightening, quantifying what has long been of concern: The time devoted to the nonclinical “value added” duties of the hospitalist is shrinking.
The threat to the culture of quality is captured in the single question: How many RVUs is a quality-improvement (QI) project worth? I’m not sure we have that answer. But without an answer, it is difficult to believe that meaningful QI can be expected without time to do so. And again, as the gap between compensation and RVUs narrows, one is left wondering if there will soon be a day where there is no value-added time remaining to perform QI at all.
Fortunately, the Affordable Care Act might provide some movement in the right direction via value-based purchasing. Linking quality outcomes to financial reimbursement is a big step forward in the hospitalist’s quest to leverage the C-suite in trading RVUs for devoted QI time. Although we still are left asking the question of how many RVUs a QI project is worth, value-based purchasing at least sets the stage for the conversation. But in the interim, it is still upon hospitalists to design these QI projects, and to learn the skills necessary to see the design to its fruition.
Threat 2: Quality Stops at Core Measures
It is hard to argue that fulfilling “core measures” is a bad thing. Nonetheless, the core measures were not meant to be quality; instead, they were meant as surrogate measures of quality. The presumption of the core-measures initiative is that the system would exist without direct attention to the core measures, operating as it ordinarily would with generic attention to meeting all standards of quality for all diseases. And at some point in time, the core measures would be assessed to give an overall assessment of the system’s quality.
What has evolved, however, is a concerted attention to meeting the core measures, with little regard to the overall culture of quality.
Let’s say you were tasked with improving the public school system in your state. As a measure of the improvement, you choose five of the 1,000 schools as “core measure” schools. The state board of education is told that the performance of these five sample schools will be assessed at the end of the year, and financial support for the system as a whole will hinge on their performance. The intended result is that attention would be paid to improving the performance of every school in the system, and this improvement would be reflected in the performance of the five sample schools. The board of education could take the route of improving all schools, but the more pragmatic route would be to funnel all resources into these five schools, to the detriment of resources for the other 995 schools. The performance of the core measure schools would dramatically improve, and funding would be secured. But ask yourself: Did the performance of the school system as a whole actually improve?
Such is the risk of the core measures in healthcare. The original intent of the core measures was to instill a culture of QI for all points of care. And this has been a valuable contribution to changing the consciousness of the healthcare system. The presumption was that the core measures would be “seeds,” and that by emphasizing these select measures, the QI culture eventually would spread to all aspects of patient care. But this plan hinged on the presumption that that there is an unlimited amount of mental energy and resources to be devoted to all tasks within healthcare. The reality is that there is a fixed amount of intellectual energy and resources to be devoted to the various aspects of healthcare. One wonders if the overemphasis on meeting the core measures might actually have taken the wind out of the sails for QI in other non-core-measure patient care.
The implications are twofold. By definition, a core measure has to be applicable to all healthcare systems, and with a fixed amount of mental energy and resources, there is a real risk that what portion is reserved for QI finds its way only to the core indicators, especially if they are overemphasized in the system. The second implication is captured in our experience with time to antibiotics. With meeting the core indicator as the priority, many systems instituted the “work-around”: Give antibiotics to every patient presenting to the ED, and you will be sure to have met the four-hour window in the core indicator. The result was an exponential increase in inappropriate antibiotic administration and radiographic tests, all because meeting the indicator became more important than the overall goal.
As stewards of the hospital system, it is upon us to ensure that the original intent of core measures remains secure: The core measures seed a culture of quality, but do not become ends in and of themselves. QI apart from the core measures must remain an equal priority, and it is the hospitalist who will be central in ensuring this comes to fruition.
Threat 3: Misplaced Incentives
There is an interesting anecdote in Steven Levitt and Stephen Dubner’s book Freakonomics.1 The story begins with a daycare center struggling with a problem: Some parents are showing up late to pick up their children at the end of the day, and this is costing the center in the way of overtime charges for the staff. To solve the problem, the center elects to institute a financial disincentive: Those showing up late to pick up their children will pay a modest financial penalty.
Fast-forward to months after the policy was put in place. The result? An exponential increase in the number of parents showing up late to pick up their children.
How do you explain worsening performance in spite of a financial disincentive? The answer resides in understanding human behavior. According to the authors, there are three primary motivations in life: financial, social, and moral. As ugly as it sounds, the decisions people make in life are driven by one of these three motivations. There is nothing wrong with providing incentives for behavior; incentives work.
But the danger arises when incentives are mismatched to behaviors. For example, if a financial outcome is the goal, then financial incentives make sense. If a social outcome (people should play better as a team) is the goal, the social incentives make sense (public recognition). But when the incentives get misaligned with their respective goals, trouble results.
What went wrong with the daycare’s plan is simple—most of the parents were motivated to pick up their children on time out of moral (“I gave my word”) or social (“I don’t want to be talked about by other parents”) incentives. But once a financial incentive was offered, the daycare center had essentially given the parents a way out in absolving their social and moral obligations. The parents had essentially cost-adjusted their behavior.
If you think this couldn’t happen to the healthcare system, let me ask you this. As a hospitalist, I see all of my patients early in the morning, because I see it as part of my obligation to the hospital team to discharge patients by 11 a.m. (social motivation).
But what if the CEO released this directive: “You will see all of your patients early in the morning, or you will take a $1,000 a year pay cut.” Is it possible that I might cost-adjust the $1,000 in exchange for sleeping in a little later and not having to deal with the morning traffic? I don’t know.
When it comes to financial incentives, there is a valley in the U-shaped curve. When the financial incentive is trivial, it is disregarded and the social/moral motivations of behavior persist (the kids are picked up on time; I persist in seeing patients early in the morning). When the financial incentive is huge, the financial incentive trumps all social/moral motivations, ensuring compliance with the goal behavior (every kid is picked up on time to avoid a penalty; I see all patients early in the day to avoid a larger penalty).
But in between is the risk zone: When the person feels they are paying an appropriate penance for not complying with the goal behavior, the financial disincentive absolves any social/moral guilt.
Healthcare reform is about incentives—and there is nothing wrong with that. But as the stewards of the inpatient healthcare system, it is upon us as hospitalists to ensure that the incentives remain matched to their intended goals, and that the untoward consequences of the incentives do not adversely affect the quality and safety of a patient’s care.
It is safe to say that the Affordable Care Act of 2010 moves us closer to a true environment of quality and patient safety. But it is equally safe to say that meaningful change will require more than what the law can provide. As stewards of the inpatient system, we have a responsibility to ensure that the healthcare system, particularly in how it responds to incentives, evolves to remain patient-centered, effective, and safe.
The next chapter in our story—the hospitalists’ story—will be one of accountability and responsibility. While there are things the government can do, the majority of what needs to be done will come directly from us. TH
Dr. Wiese is president of SHM.
Reference
- Levitt SD, Dubner SJ. Freakonomics: A Rogue Economist Explores the Hidden Side of Everything. New York City: William Morrow; 2005.
From the outset, HM has been about efficiency. And there was nothing wrong with that, for value is quality divided by cost. But in our story, we found that mere efficiency was not enough: The lowering of the denominator (cost) had to be met with an escalation of the numerator (quality) to ensure value.
And see us as being born in the right place at the right time. For with the national focus turning to the need for quality and patient safety, hospital medicine was in the right place and the right time to heed the call to action: appropriately stepping up to enact efforts to make the slope of the line (on a chart of quality vs. cost) “STEEEPER” … finding systems innovations to make care Safe, Timely, Efficient, Effective, Equitable, and Patient-centered.
Of course, the story continued with the Affordable Care Act and healthcare reform, greatly accelerating our evolution as change agents. And now we find ourselves fully invested in a “change the system” mentality, perfectly positioned to meaningfully change healthcare for millions of people. But threats loom—specifically, the “R” in the STEEEPER mnemonic: the risks to quality in the face of healthcare reform.
So in the next chapter of our story, I present to you our challenge: how to overcome the threats to quality in the context of healthcare reform. The first three are presented here; in subsequent articles, I will address the remainder. Overcoming all threats will hinge on mastering the four truisms of cultural change:
- Systems drive function;
- Every system is perfectly designed to produce the outcomes that it does;
- This is not an issue of people needing to try harder; and
- The “no blame” culture begins with a paradigm shift from the “person at fault” to the “system at fault.”
Threat 1: Failure to Fund Quality
SHM elected to merge its annual State of Hospital Medicine survey with the MGMA. Though not without risk, this has resulted in the anticipated benefits. The MGMA collaboration brings greater leverage in working with the C-suite, which is pre-programmed to react to MGMA surveys. From the most recent MGMA survey comes good news: The financial compensation for hospitalists has increased. A sobering insight, however, is that this increase in compensation has been met with a corresponding increase in work intensity—RVUs. Further, the link between RVUs and compensation appears to be tightening, quantifying what has long been of concern: The time devoted to the nonclinical “value added” duties of the hospitalist is shrinking.
The threat to the culture of quality is captured in the single question: How many RVUs is a quality-improvement (QI) project worth? I’m not sure we have that answer. But without an answer, it is difficult to believe that meaningful QI can be expected without time to do so. And again, as the gap between compensation and RVUs narrows, one is left wondering if there will soon be a day where there is no value-added time remaining to perform QI at all.
Fortunately, the Affordable Care Act might provide some movement in the right direction via value-based purchasing. Linking quality outcomes to financial reimbursement is a big step forward in the hospitalist’s quest to leverage the C-suite in trading RVUs for devoted QI time. Although we still are left asking the question of how many RVUs a QI project is worth, value-based purchasing at least sets the stage for the conversation. But in the interim, it is still upon hospitalists to design these QI projects, and to learn the skills necessary to see the design to its fruition.
Threat 2: Quality Stops at Core Measures
It is hard to argue that fulfilling “core measures” is a bad thing. Nonetheless, the core measures were not meant to be quality; instead, they were meant as surrogate measures of quality. The presumption of the core-measures initiative is that the system would exist without direct attention to the core measures, operating as it ordinarily would with generic attention to meeting all standards of quality for all diseases. And at some point in time, the core measures would be assessed to give an overall assessment of the system’s quality.
What has evolved, however, is a concerted attention to meeting the core measures, with little regard to the overall culture of quality.
Let’s say you were tasked with improving the public school system in your state. As a measure of the improvement, you choose five of the 1,000 schools as “core measure” schools. The state board of education is told that the performance of these five sample schools will be assessed at the end of the year, and financial support for the system as a whole will hinge on their performance. The intended result is that attention would be paid to improving the performance of every school in the system, and this improvement would be reflected in the performance of the five sample schools. The board of education could take the route of improving all schools, but the more pragmatic route would be to funnel all resources into these five schools, to the detriment of resources for the other 995 schools. The performance of the core measure schools would dramatically improve, and funding would be secured. But ask yourself: Did the performance of the school system as a whole actually improve?
Such is the risk of the core measures in healthcare. The original intent of the core measures was to instill a culture of QI for all points of care. And this has been a valuable contribution to changing the consciousness of the healthcare system. The presumption was that the core measures would be “seeds,” and that by emphasizing these select measures, the QI culture eventually would spread to all aspects of patient care. But this plan hinged on the presumption that that there is an unlimited amount of mental energy and resources to be devoted to all tasks within healthcare. The reality is that there is a fixed amount of intellectual energy and resources to be devoted to the various aspects of healthcare. One wonders if the overemphasis on meeting the core measures might actually have taken the wind out of the sails for QI in other non-core-measure patient care.
The implications are twofold. By definition, a core measure has to be applicable to all healthcare systems, and with a fixed amount of mental energy and resources, there is a real risk that what portion is reserved for QI finds its way only to the core indicators, especially if they are overemphasized in the system. The second implication is captured in our experience with time to antibiotics. With meeting the core indicator as the priority, many systems instituted the “work-around”: Give antibiotics to every patient presenting to the ED, and you will be sure to have met the four-hour window in the core indicator. The result was an exponential increase in inappropriate antibiotic administration and radiographic tests, all because meeting the indicator became more important than the overall goal.
As stewards of the hospital system, it is upon us to ensure that the original intent of core measures remains secure: The core measures seed a culture of quality, but do not become ends in and of themselves. QI apart from the core measures must remain an equal priority, and it is the hospitalist who will be central in ensuring this comes to fruition.
Threat 3: Misplaced Incentives
There is an interesting anecdote in Steven Levitt and Stephen Dubner’s book Freakonomics.1 The story begins with a daycare center struggling with a problem: Some parents are showing up late to pick up their children at the end of the day, and this is costing the center in the way of overtime charges for the staff. To solve the problem, the center elects to institute a financial disincentive: Those showing up late to pick up their children will pay a modest financial penalty.
Fast-forward to months after the policy was put in place. The result? An exponential increase in the number of parents showing up late to pick up their children.
How do you explain worsening performance in spite of a financial disincentive? The answer resides in understanding human behavior. According to the authors, there are three primary motivations in life: financial, social, and moral. As ugly as it sounds, the decisions people make in life are driven by one of these three motivations. There is nothing wrong with providing incentives for behavior; incentives work.
But the danger arises when incentives are mismatched to behaviors. For example, if a financial outcome is the goal, then financial incentives make sense. If a social outcome (people should play better as a team) is the goal, the social incentives make sense (public recognition). But when the incentives get misaligned with their respective goals, trouble results.
What went wrong with the daycare’s plan is simple—most of the parents were motivated to pick up their children on time out of moral (“I gave my word”) or social (“I don’t want to be talked about by other parents”) incentives. But once a financial incentive was offered, the daycare center had essentially given the parents a way out in absolving their social and moral obligations. The parents had essentially cost-adjusted their behavior.
If you think this couldn’t happen to the healthcare system, let me ask you this. As a hospitalist, I see all of my patients early in the morning, because I see it as part of my obligation to the hospital team to discharge patients by 11 a.m. (social motivation).
But what if the CEO released this directive: “You will see all of your patients early in the morning, or you will take a $1,000 a year pay cut.” Is it possible that I might cost-adjust the $1,000 in exchange for sleeping in a little later and not having to deal with the morning traffic? I don’t know.
When it comes to financial incentives, there is a valley in the U-shaped curve. When the financial incentive is trivial, it is disregarded and the social/moral motivations of behavior persist (the kids are picked up on time; I persist in seeing patients early in the morning). When the financial incentive is huge, the financial incentive trumps all social/moral motivations, ensuring compliance with the goal behavior (every kid is picked up on time to avoid a penalty; I see all patients early in the day to avoid a larger penalty).
But in between is the risk zone: When the person feels they are paying an appropriate penance for not complying with the goal behavior, the financial disincentive absolves any social/moral guilt.
Healthcare reform is about incentives—and there is nothing wrong with that. But as the stewards of the inpatient healthcare system, it is upon us as hospitalists to ensure that the incentives remain matched to their intended goals, and that the untoward consequences of the incentives do not adversely affect the quality and safety of a patient’s care.
It is safe to say that the Affordable Care Act of 2010 moves us closer to a true environment of quality and patient safety. But it is equally safe to say that meaningful change will require more than what the law can provide. As stewards of the inpatient system, we have a responsibility to ensure that the healthcare system, particularly in how it responds to incentives, evolves to remain patient-centered, effective, and safe.
The next chapter in our story—the hospitalists’ story—will be one of accountability and responsibility. While there are things the government can do, the majority of what needs to be done will come directly from us. TH
Dr. Wiese is president of SHM.
Reference
- Levitt SD, Dubner SJ. Freakonomics: A Rogue Economist Explores the Hidden Side of Everything. New York City: William Morrow; 2005.
From the outset, HM has been about efficiency. And there was nothing wrong with that, for value is quality divided by cost. But in our story, we found that mere efficiency was not enough: The lowering of the denominator (cost) had to be met with an escalation of the numerator (quality) to ensure value.
And see us as being born in the right place at the right time. For with the national focus turning to the need for quality and patient safety, hospital medicine was in the right place and the right time to heed the call to action: appropriately stepping up to enact efforts to make the slope of the line (on a chart of quality vs. cost) “STEEEPER” … finding systems innovations to make care Safe, Timely, Efficient, Effective, Equitable, and Patient-centered.
Of course, the story continued with the Affordable Care Act and healthcare reform, greatly accelerating our evolution as change agents. And now we find ourselves fully invested in a “change the system” mentality, perfectly positioned to meaningfully change healthcare for millions of people. But threats loom—specifically, the “R” in the STEEEPER mnemonic: the risks to quality in the face of healthcare reform.
So in the next chapter of our story, I present to you our challenge: how to overcome the threats to quality in the context of healthcare reform. The first three are presented here; in subsequent articles, I will address the remainder. Overcoming all threats will hinge on mastering the four truisms of cultural change:
- Systems drive function;
- Every system is perfectly designed to produce the outcomes that it does;
- This is not an issue of people needing to try harder; and
- The “no blame” culture begins with a paradigm shift from the “person at fault” to the “system at fault.”
Threat 1: Failure to Fund Quality
SHM elected to merge its annual State of Hospital Medicine survey with the MGMA. Though not without risk, this has resulted in the anticipated benefits. The MGMA collaboration brings greater leverage in working with the C-suite, which is pre-programmed to react to MGMA surveys. From the most recent MGMA survey comes good news: The financial compensation for hospitalists has increased. A sobering insight, however, is that this increase in compensation has been met with a corresponding increase in work intensity—RVUs. Further, the link between RVUs and compensation appears to be tightening, quantifying what has long been of concern: The time devoted to the nonclinical “value added” duties of the hospitalist is shrinking.
The threat to the culture of quality is captured in the single question: How many RVUs is a quality-improvement (QI) project worth? I’m not sure we have that answer. But without an answer, it is difficult to believe that meaningful QI can be expected without time to do so. And again, as the gap between compensation and RVUs narrows, one is left wondering if there will soon be a day where there is no value-added time remaining to perform QI at all.
Fortunately, the Affordable Care Act might provide some movement in the right direction via value-based purchasing. Linking quality outcomes to financial reimbursement is a big step forward in the hospitalist’s quest to leverage the C-suite in trading RVUs for devoted QI time. Although we still are left asking the question of how many RVUs a QI project is worth, value-based purchasing at least sets the stage for the conversation. But in the interim, it is still upon hospitalists to design these QI projects, and to learn the skills necessary to see the design to its fruition.
Threat 2: Quality Stops at Core Measures
It is hard to argue that fulfilling “core measures” is a bad thing. Nonetheless, the core measures were not meant to be quality; instead, they were meant as surrogate measures of quality. The presumption of the core-measures initiative is that the system would exist without direct attention to the core measures, operating as it ordinarily would with generic attention to meeting all standards of quality for all diseases. And at some point in time, the core measures would be assessed to give an overall assessment of the system’s quality.
What has evolved, however, is a concerted attention to meeting the core measures, with little regard to the overall culture of quality.
Let’s say you were tasked with improving the public school system in your state. As a measure of the improvement, you choose five of the 1,000 schools as “core measure” schools. The state board of education is told that the performance of these five sample schools will be assessed at the end of the year, and financial support for the system as a whole will hinge on their performance. The intended result is that attention would be paid to improving the performance of every school in the system, and this improvement would be reflected in the performance of the five sample schools. The board of education could take the route of improving all schools, but the more pragmatic route would be to funnel all resources into these five schools, to the detriment of resources for the other 995 schools. The performance of the core measure schools would dramatically improve, and funding would be secured. But ask yourself: Did the performance of the school system as a whole actually improve?
Such is the risk of the core measures in healthcare. The original intent of the core measures was to instill a culture of QI for all points of care. And this has been a valuable contribution to changing the consciousness of the healthcare system. The presumption was that the core measures would be “seeds,” and that by emphasizing these select measures, the QI culture eventually would spread to all aspects of patient care. But this plan hinged on the presumption that that there is an unlimited amount of mental energy and resources to be devoted to all tasks within healthcare. The reality is that there is a fixed amount of intellectual energy and resources to be devoted to the various aspects of healthcare. One wonders if the overemphasis on meeting the core measures might actually have taken the wind out of the sails for QI in other non-core-measure patient care.
The implications are twofold. By definition, a core measure has to be applicable to all healthcare systems, and with a fixed amount of mental energy and resources, there is a real risk that what portion is reserved for QI finds its way only to the core indicators, especially if they are overemphasized in the system. The second implication is captured in our experience with time to antibiotics. With meeting the core indicator as the priority, many systems instituted the “work-around”: Give antibiotics to every patient presenting to the ED, and you will be sure to have met the four-hour window in the core indicator. The result was an exponential increase in inappropriate antibiotic administration and radiographic tests, all because meeting the indicator became more important than the overall goal.
As stewards of the hospital system, it is upon us to ensure that the original intent of core measures remains secure: The core measures seed a culture of quality, but do not become ends in and of themselves. QI apart from the core measures must remain an equal priority, and it is the hospitalist who will be central in ensuring this comes to fruition.
Threat 3: Misplaced Incentives
There is an interesting anecdote in Steven Levitt and Stephen Dubner’s book Freakonomics.1 The story begins with a daycare center struggling with a problem: Some parents are showing up late to pick up their children at the end of the day, and this is costing the center in the way of overtime charges for the staff. To solve the problem, the center elects to institute a financial disincentive: Those showing up late to pick up their children will pay a modest financial penalty.
Fast-forward to months after the policy was put in place. The result? An exponential increase in the number of parents showing up late to pick up their children.
How do you explain worsening performance in spite of a financial disincentive? The answer resides in understanding human behavior. According to the authors, there are three primary motivations in life: financial, social, and moral. As ugly as it sounds, the decisions people make in life are driven by one of these three motivations. There is nothing wrong with providing incentives for behavior; incentives work.
But the danger arises when incentives are mismatched to behaviors. For example, if a financial outcome is the goal, then financial incentives make sense. If a social outcome (people should play better as a team) is the goal, the social incentives make sense (public recognition). But when the incentives get misaligned with their respective goals, trouble results.
What went wrong with the daycare’s plan is simple—most of the parents were motivated to pick up their children on time out of moral (“I gave my word”) or social (“I don’t want to be talked about by other parents”) incentives. But once a financial incentive was offered, the daycare center had essentially given the parents a way out in absolving their social and moral obligations. The parents had essentially cost-adjusted their behavior.
If you think this couldn’t happen to the healthcare system, let me ask you this. As a hospitalist, I see all of my patients early in the morning, because I see it as part of my obligation to the hospital team to discharge patients by 11 a.m. (social motivation).
But what if the CEO released this directive: “You will see all of your patients early in the morning, or you will take a $1,000 a year pay cut.” Is it possible that I might cost-adjust the $1,000 in exchange for sleeping in a little later and not having to deal with the morning traffic? I don’t know.
When it comes to financial incentives, there is a valley in the U-shaped curve. When the financial incentive is trivial, it is disregarded and the social/moral motivations of behavior persist (the kids are picked up on time; I persist in seeing patients early in the morning). When the financial incentive is huge, the financial incentive trumps all social/moral motivations, ensuring compliance with the goal behavior (every kid is picked up on time to avoid a penalty; I see all patients early in the day to avoid a larger penalty).
But in between is the risk zone: When the person feels they are paying an appropriate penance for not complying with the goal behavior, the financial disincentive absolves any social/moral guilt.
Healthcare reform is about incentives—and there is nothing wrong with that. But as the stewards of the inpatient healthcare system, it is upon us as hospitalists to ensure that the incentives remain matched to their intended goals, and that the untoward consequences of the incentives do not adversely affect the quality and safety of a patient’s care.
It is safe to say that the Affordable Care Act of 2010 moves us closer to a true environment of quality and patient safety. But it is equally safe to say that meaningful change will require more than what the law can provide. As stewards of the inpatient system, we have a responsibility to ensure that the healthcare system, particularly in how it responds to incentives, evolves to remain patient-centered, effective, and safe.
The next chapter in our story—the hospitalists’ story—will be one of accountability and responsibility. While there are things the government can do, the majority of what needs to be done will come directly from us. TH
Dr. Wiese is president of SHM.
Reference
- Levitt SD, Dubner SJ. Freakonomics: A Rogue Economist Explores the Hidden Side of Everything. New York City: William Morrow; 2005.
Gettin’ Dirty
Several months ago, my toilet broke. You should also know that I’m not particularly handy. So when I first realized that the toilet bowl seemed to fill constantly, I got a little stressed out.
How much was it going cost to call in a plumber on the weekend?
What kind of a water bill was I going to have?
Was this a serious problem?
I took a quick peek in the tank, but that just made me more confused. I was paralyzed by a lack of know-how.
Normally, I would have just Googled a local plumber. But that day, I decided to do something different. Maybe it was because it was the fantasy football offseason. Maybe it was because my wife had started to ask my father-in-law to change light bulbs around the house. Or, maybe, I wanted to learn to actually fix the problem. A few hours later, after an Internet lesson in toilet physiology, a $4.12 trip to Home Depot, and a wet pair of hands, I had replaced my first toilet flapper.
This wasn’t the rebuilding of a car engine, but it was a clear DIY step toward self-improvement. Easily the most memorable moment here was my sense of accomplishment.
I felt empowered.
One Part Science, One Part Art
It’s taken me a while to realize this, but I’ve begun to take advantage of improvement opportunities at work as well. No, I haven’t been moonlighting as a plumber for my hospital. I’ve just been fortunate to be part of a trifecta of rewarding quality-improvement (QI) projects over the past year. Before I’d gotten my hands dirty with these, my understanding of QI was fairly naive. I’d heard about Plan-Do-Study-Act many times. I had listened to a talk at a national conference. And I had kept up with the general medical literature on the subject.
But none of those activities had truly prepared me for experience of actually doing the work on my own.
By taking on a project, an ambitious attempt to reduce continuous pulse oximetry use, I experienced a crash course in both the science and the art of process improvement. I was forced to overcome my “I don’t know how” inertia. And with expert guidance in the form of a clinical safety and effectiveness class, I learned the importance of run charts (science) and a well-crafted multidisciplinary team (art) in changing established but inefficient behavior.
Our rates of continuous pulse oximetry usage dropped by 50%, and cost savings were $12,000 per year on one unit. These results made my prior attempts at change—years of complaining about ingrained nursing culture—look infantile. (OK, maybe it was ineffective, but who hasn’t complained about the overuse of continuous monitoring?)
I haven’t met a pediatric hospitalist who wouldn’t understand the symbolic importance of this success. But I know of many hospitalists who have not yet participated in meaningful QI project. Imagine calling a plumber who grasped the flush and fill mechanism of a toilet but had never touched real porcelain. Here’s an even better analogy: What if doctors could get licensed without having touched real patients?
If pediatric hospitalists are to transform the care delivery of hospitalized children, and quality learning only comes through hands-on training, then we need some more hands in the pot.
Discharge Improvement
On the heels of my first project, I was fortunate enough to augment my education through another effort—this time with a cohort of fellow pediatric hospitalists. This was a national collaborative to improve discharge handoffs, and I will admit that, at the outset, I was as puzzled as the first time I pulled the lid off the tank of the toilet. There were just too many permutations on PCP communication at the participating institutions, and some felt our aim of timely discharge handoffs was unattainable.
What carried me through, however, was the collective and infectious DIY—no, QIY (Quality Improve-it-Yourself) attitude of the group. We were all learning, and regular participation in the collaborative essentially guaranteed improvement. We achieved our aim of 90% communication with PCPs within two days of discharge. The secret was simple: The more you do, the more you learn.
Pediatric hospitalists can transform care delivery through a focus on safe and quality care, but the tools to accomplish this must come through post-residency, on-the-job learning. This QI know-how must efficiently spread among our ranks through practical and project-based educational efforts. It’s “see one, do one, teach one,” but we’re not talking about lumbar punctures anymore.
This is a journey in which we all take on the responsibility of rolling up our sleeves and simply learn by doing. And here is where the third leg of my as-yet-unfinished QI course unfolds.
Through my involvement with the Value in Inpatient Pediatrics (VIP) Network, I’ve gained a newfound vision for what the future might hold. VIP has evolved from a benchmarking project focused on bronchiolitis to an improvement network that will incorporate projects similar to the discharge handoff collaborative above.
In the process, a model for how to rapidly spread QI learning has emerged. The capacity lies in the network’s rapidly growing connectivity. The power comes from the individuals: motivated, card-carrying pediatric hospitalists from a wide array of sites. Collaborative learning harbors the potential to exponentially increase the pace at which we improve.
The future of our quality care is bright. I see an open network of improvement doers and learners. I see collaboration on quality and safety initiatives in all manner of hospitals and communities. I see that this will all be built upon a foundation of hard work and a QIY attitude.
You, too, will play a role.
Just don’t be afraid to get your hands a little dirty. TH
Dr. Shen is medical director of hospital medicine at Dell Children’s Medical Center in Austin, Texas. He is pediatric editor of The Hospitalist.
Several months ago, my toilet broke. You should also know that I’m not particularly handy. So when I first realized that the toilet bowl seemed to fill constantly, I got a little stressed out.
How much was it going cost to call in a plumber on the weekend?
What kind of a water bill was I going to have?
Was this a serious problem?
I took a quick peek in the tank, but that just made me more confused. I was paralyzed by a lack of know-how.
Normally, I would have just Googled a local plumber. But that day, I decided to do something different. Maybe it was because it was the fantasy football offseason. Maybe it was because my wife had started to ask my father-in-law to change light bulbs around the house. Or, maybe, I wanted to learn to actually fix the problem. A few hours later, after an Internet lesson in toilet physiology, a $4.12 trip to Home Depot, and a wet pair of hands, I had replaced my first toilet flapper.
This wasn’t the rebuilding of a car engine, but it was a clear DIY step toward self-improvement. Easily the most memorable moment here was my sense of accomplishment.
I felt empowered.
One Part Science, One Part Art
It’s taken me a while to realize this, but I’ve begun to take advantage of improvement opportunities at work as well. No, I haven’t been moonlighting as a plumber for my hospital. I’ve just been fortunate to be part of a trifecta of rewarding quality-improvement (QI) projects over the past year. Before I’d gotten my hands dirty with these, my understanding of QI was fairly naive. I’d heard about Plan-Do-Study-Act many times. I had listened to a talk at a national conference. And I had kept up with the general medical literature on the subject.
But none of those activities had truly prepared me for experience of actually doing the work on my own.
By taking on a project, an ambitious attempt to reduce continuous pulse oximetry use, I experienced a crash course in both the science and the art of process improvement. I was forced to overcome my “I don’t know how” inertia. And with expert guidance in the form of a clinical safety and effectiveness class, I learned the importance of run charts (science) and a well-crafted multidisciplinary team (art) in changing established but inefficient behavior.
Our rates of continuous pulse oximetry usage dropped by 50%, and cost savings were $12,000 per year on one unit. These results made my prior attempts at change—years of complaining about ingrained nursing culture—look infantile. (OK, maybe it was ineffective, but who hasn’t complained about the overuse of continuous monitoring?)
I haven’t met a pediatric hospitalist who wouldn’t understand the symbolic importance of this success. But I know of many hospitalists who have not yet participated in meaningful QI project. Imagine calling a plumber who grasped the flush and fill mechanism of a toilet but had never touched real porcelain. Here’s an even better analogy: What if doctors could get licensed without having touched real patients?
If pediatric hospitalists are to transform the care delivery of hospitalized children, and quality learning only comes through hands-on training, then we need some more hands in the pot.
Discharge Improvement
On the heels of my first project, I was fortunate enough to augment my education through another effort—this time with a cohort of fellow pediatric hospitalists. This was a national collaborative to improve discharge handoffs, and I will admit that, at the outset, I was as puzzled as the first time I pulled the lid off the tank of the toilet. There were just too many permutations on PCP communication at the participating institutions, and some felt our aim of timely discharge handoffs was unattainable.
What carried me through, however, was the collective and infectious DIY—no, QIY (Quality Improve-it-Yourself) attitude of the group. We were all learning, and regular participation in the collaborative essentially guaranteed improvement. We achieved our aim of 90% communication with PCPs within two days of discharge. The secret was simple: The more you do, the more you learn.
Pediatric hospitalists can transform care delivery through a focus on safe and quality care, but the tools to accomplish this must come through post-residency, on-the-job learning. This QI know-how must efficiently spread among our ranks through practical and project-based educational efforts. It’s “see one, do one, teach one,” but we’re not talking about lumbar punctures anymore.
This is a journey in which we all take on the responsibility of rolling up our sleeves and simply learn by doing. And here is where the third leg of my as-yet-unfinished QI course unfolds.
Through my involvement with the Value in Inpatient Pediatrics (VIP) Network, I’ve gained a newfound vision for what the future might hold. VIP has evolved from a benchmarking project focused on bronchiolitis to an improvement network that will incorporate projects similar to the discharge handoff collaborative above.
In the process, a model for how to rapidly spread QI learning has emerged. The capacity lies in the network’s rapidly growing connectivity. The power comes from the individuals: motivated, card-carrying pediatric hospitalists from a wide array of sites. Collaborative learning harbors the potential to exponentially increase the pace at which we improve.
The future of our quality care is bright. I see an open network of improvement doers and learners. I see collaboration on quality and safety initiatives in all manner of hospitals and communities. I see that this will all be built upon a foundation of hard work and a QIY attitude.
You, too, will play a role.
Just don’t be afraid to get your hands a little dirty. TH
Dr. Shen is medical director of hospital medicine at Dell Children’s Medical Center in Austin, Texas. He is pediatric editor of The Hospitalist.
Several months ago, my toilet broke. You should also know that I’m not particularly handy. So when I first realized that the toilet bowl seemed to fill constantly, I got a little stressed out.
How much was it going cost to call in a plumber on the weekend?
What kind of a water bill was I going to have?
Was this a serious problem?
I took a quick peek in the tank, but that just made me more confused. I was paralyzed by a lack of know-how.
Normally, I would have just Googled a local plumber. But that day, I decided to do something different. Maybe it was because it was the fantasy football offseason. Maybe it was because my wife had started to ask my father-in-law to change light bulbs around the house. Or, maybe, I wanted to learn to actually fix the problem. A few hours later, after an Internet lesson in toilet physiology, a $4.12 trip to Home Depot, and a wet pair of hands, I had replaced my first toilet flapper.
This wasn’t the rebuilding of a car engine, but it was a clear DIY step toward self-improvement. Easily the most memorable moment here was my sense of accomplishment.
I felt empowered.
One Part Science, One Part Art
It’s taken me a while to realize this, but I’ve begun to take advantage of improvement opportunities at work as well. No, I haven’t been moonlighting as a plumber for my hospital. I’ve just been fortunate to be part of a trifecta of rewarding quality-improvement (QI) projects over the past year. Before I’d gotten my hands dirty with these, my understanding of QI was fairly naive. I’d heard about Plan-Do-Study-Act many times. I had listened to a talk at a national conference. And I had kept up with the general medical literature on the subject.
But none of those activities had truly prepared me for experience of actually doing the work on my own.
By taking on a project, an ambitious attempt to reduce continuous pulse oximetry use, I experienced a crash course in both the science and the art of process improvement. I was forced to overcome my “I don’t know how” inertia. And with expert guidance in the form of a clinical safety and effectiveness class, I learned the importance of run charts (science) and a well-crafted multidisciplinary team (art) in changing established but inefficient behavior.
Our rates of continuous pulse oximetry usage dropped by 50%, and cost savings were $12,000 per year on one unit. These results made my prior attempts at change—years of complaining about ingrained nursing culture—look infantile. (OK, maybe it was ineffective, but who hasn’t complained about the overuse of continuous monitoring?)
I haven’t met a pediatric hospitalist who wouldn’t understand the symbolic importance of this success. But I know of many hospitalists who have not yet participated in meaningful QI project. Imagine calling a plumber who grasped the flush and fill mechanism of a toilet but had never touched real porcelain. Here’s an even better analogy: What if doctors could get licensed without having touched real patients?
If pediatric hospitalists are to transform the care delivery of hospitalized children, and quality learning only comes through hands-on training, then we need some more hands in the pot.
Discharge Improvement
On the heels of my first project, I was fortunate enough to augment my education through another effort—this time with a cohort of fellow pediatric hospitalists. This was a national collaborative to improve discharge handoffs, and I will admit that, at the outset, I was as puzzled as the first time I pulled the lid off the tank of the toilet. There were just too many permutations on PCP communication at the participating institutions, and some felt our aim of timely discharge handoffs was unattainable.
What carried me through, however, was the collective and infectious DIY—no, QIY (Quality Improve-it-Yourself) attitude of the group. We were all learning, and regular participation in the collaborative essentially guaranteed improvement. We achieved our aim of 90% communication with PCPs within two days of discharge. The secret was simple: The more you do, the more you learn.
Pediatric hospitalists can transform care delivery through a focus on safe and quality care, but the tools to accomplish this must come through post-residency, on-the-job learning. This QI know-how must efficiently spread among our ranks through practical and project-based educational efforts. It’s “see one, do one, teach one,” but we’re not talking about lumbar punctures anymore.
This is a journey in which we all take on the responsibility of rolling up our sleeves and simply learn by doing. And here is where the third leg of my as-yet-unfinished QI course unfolds.
Through my involvement with the Value in Inpatient Pediatrics (VIP) Network, I’ve gained a newfound vision for what the future might hold. VIP has evolved from a benchmarking project focused on bronchiolitis to an improvement network that will incorporate projects similar to the discharge handoff collaborative above.
In the process, a model for how to rapidly spread QI learning has emerged. The capacity lies in the network’s rapidly growing connectivity. The power comes from the individuals: motivated, card-carrying pediatric hospitalists from a wide array of sites. Collaborative learning harbors the potential to exponentially increase the pace at which we improve.
The future of our quality care is bright. I see an open network of improvement doers and learners. I see collaboration on quality and safety initiatives in all manner of hospitals and communities. I see that this will all be built upon a foundation of hard work and a QIY attitude.
You, too, will play a role.
Just don’t be afraid to get your hands a little dirty. TH
Dr. Shen is medical director of hospital medicine at Dell Children’s Medical Center in Austin, Texas. He is pediatric editor of The Hospitalist.
Designed to Harm
If every system is perfectly designed to get the results it gets …
And if 15 million patients are harmed every year from medical care …
And if as many as 98,000 people die every year due to medical errors in hospitals …
Then what does that say about the system we have designed?
A System Designed to Competently Hurt Many
By now you’ve no doubt heard, read, and possibly even uttered the above facts and figures yourself. I think we all have our opinions about the veracity of these numbers, but I don’t think any of us would argue with the sentiment. The U.S. healthcare system comprises the most competent, compassionate, well-meaning, and caring professionals on this planet—who harm, maim, and kill countless people every year.
What a discomforting paradox.
Equity: The Overlooked Quality Domain
Many years back, the Institute of Medicine (IOM) published a list of six “domains” of healthcare quality. You’ve no doubt stumbled across the IOM’s Safe … Timely … Effective … Efficient … Equitable … Patient-Centered mnemonic recipe—STEEEP—for high-quality care.1 In fact, it’s hard to read a journal, attend a medical presentation, or open a local newspaper without finding reference to these domains. It’s all the rage to talk about wrong-site surgery (safe), access to care (timely), comparative-effectiveness research (effective), lean concepts (efficient), and individualized medicine (patient-centered). However, the sixth domain often seems to get the Jan Brady treatment—minimized, marginalized, and oft-forgotten.
The IOM defines equitable care as that which “does not vary in quality because of personal characteristics such as gender, ethnicity, geographic location, and socioeconomic status.”1 To be sure, there is some hum at the national level about issues of equity, especially around healthcare coverage for all. And this is important. However, what appears lost in the rant surrounding the inherent inequities in our tiered health insurance system is that we have blantant inequalities baked into the everyday machinery of our hospitals. And they affect all, regardless of skin color, gender, or insurance status.
Think for a moment about your hospital. Are the type, level, and access to care equal at all times? Does the level of care change when the streetlights come on? How about on weekends and holidays? Do your operating rooms run on Saturdays and Sundays? Can patients get chemotherapy on the weekends? Does your hospital alter its nursing staff ratios after hours? Can you get an ultrasound at midnight? How about a urology or neurosurgery consult at 2 a.m.? How about getting interventional radiology to place an IVC filter on a holiday?
Now scratch a bit closer to home. Does your hospitalist group downstaff on weekends and holidays, even though the volumes probably warrant more coverage? Are your night providers part of your group, or are they moonlighters? Do your after-hours providers cross-cover and admit a reasonable number of patients, or are they frequently overwhelmed? Do they cover patients or admit for services that they don’t typically care for during the day (e.g. ICU, neurosurgical, subspecialty cardiology or oncology patients)? For the intensely ill patients admitted to U.S. hospitals today, should the type and availability of care differ when it’s delivered at 3 p.m. or 3 a.m., Sunday or Monday?
Disregarding the macro-inequities in our societal approach to healthcare, can we even ensure equitable care within our own hospital walls 24 hours a day, seven days a week?
The Answer: An Unfortunate “No”
For the record, I hate working nights, abhor working weekends, and resent working holidays. But the thing I’d hate even more than working nights, weekends, and holidays would be being a patient admitted during a night, weekend, or holiday. To understand why, I have to look no further than my hospital parking lot. During bankers’ hours, I can barely find a parking spot on the top floor of our multilevel parking structure.
Fast-forward to Saturday, and I have my pick of empty football fields’ worth of spots on all floors. Ditto Sundays, nights, and holidays.
Why is it that nationally, a collectively near-trillion-dollar hospital enterprise finds it acceptable to effectively shutter itself for a quarter to a third of the week? Especially when doing so seems to counter their primary mission of providing safe, timely, effective, efficient, equitable, and patient-centered care.
The Weekend Effect
There are, of course, economic and operational reasons to downshift during off hours—some hospitals don’t have the elective procedures to run operating rooms seven days a week, and very few patients want to have their elective colonoscopy at 11 p.m. or their chemotherapy during Thanksgiving dinner. However, in most cases, the reasons for doing so center on hospital staff and physician satisfaction. Most us of just don’t like working off hours. As a result, studies have shown significantly less access to such high-level care as coronary angiography and percutaneous coronary intervention on weekends.2,3
And the effects of this “weekend effect” can be devastating.
A recent paper in the New England Journal of Medicine reported that for every 1,000 patients admitted with a myocardial infarction on a weekend, nine more would die than a comparable group admitted during the week.4 Their offense? Having the misfortune to get ill on a Saturday morning. The authors concluded that this higher mortality was secondary to a lower rate of invasive cardiac procedures, presumably because they were less available. And the weekend effect isn’t just limited to coronary care. Poorer outcomes, including higher mortality rates, have been reported for weekend admissions to the neonatal ICU and adult ICU, as well as admission for epiglottitis, ruptured abdominal aortic aneurysms, and pulmonary embolism.5,6,7
So let’s connect the dots: A system designed with inequal access to lifesaving therapies and appropriate staffing results in worse outcomes, more harm, more deaths.
To be clear, 98,000 people don’t die every year because of my disdain for working nights and weekends. This is a much deeper problem that hinges on many unsatisfactory systems working unsatisfactorily in tandem (e.g. see the other five IOM domains of quality care). Furthermore, I don’t mean to suggest that hospitalists necessarily have a lot of say in cardiac catheterization schedules. Yet we do control our own systems of care—how many patients we admit and cover during a shift, how strongly we advocate for timely testing and consultation, how we staff weekends and cover patients at night.
And, more and more, we are in a position to enhance the care delivery systems of the hospital and its providers that surround us. With that comes a responsibility to ensure that these systems are highly functioning and equitable, regardless of the time of day, day of the week.
If we are going to fundamentally enhance the quality of care, then we have to design safer systems of care. It will take time and resources to alter many of our bruised systems of care, but we can begin by at least ensuring equity in how we deliver care at our own institutions. That is, unless we are comfortable with a system perfectly designed to harm. TH
Dr. Glasheen is associate professor of medicine at the University of Colorado Denver, where he serves as director of the Hospital Medicine Program and the Hospitalist Training Program, and as associate program director of the Internal Medicine Residency Program.
References
- Institute of Medicine (IOM). Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, D.C.: National Academy Press; 2001.
- Bell CM, Redelmeier DA. Waiting for urgent procedures on the weekend among emergently hospitalized patients. Am J Med. 2004:117(3):175-181.
- Magid DJ, Wang Y, Herrin J, et al. Relationship between time of day, day of week, timeliness of reperfusion, and in-hospital mortality for patients with acute ST-segment elevation myocardial infarction. JAMA. 2005;294(7):803-812.
- Kostis WJ, Demissie K, Marcella SW, Shao YH, Wilson AC, Moreyra AE. Weekend versus weekday admission and mortality from myocardial infarction. N Eng J Med. 2007;356 (11):1099-1109.
- Hendry RA. The weekend—a dangerous time to be born? Br J Obstet Gynaecol. 1981;88(12):1200-1203.
- Barnett MJ, Kaboli PJ, Sirio CA, Rosenthal GE. Day of the week of intensive care admission and patient outcomes: a multisite regional evaluation. Med Care. 2002;40(6):530-539.
- Bell CM, Redelmeier DA. Mortality among patients admitted to hospitals on weekends as compared with weekdays. N Eng J Med. 2001;345(9):663-668.
If every system is perfectly designed to get the results it gets …
And if 15 million patients are harmed every year from medical care …
And if as many as 98,000 people die every year due to medical errors in hospitals …
Then what does that say about the system we have designed?
A System Designed to Competently Hurt Many
By now you’ve no doubt heard, read, and possibly even uttered the above facts and figures yourself. I think we all have our opinions about the veracity of these numbers, but I don’t think any of us would argue with the sentiment. The U.S. healthcare system comprises the most competent, compassionate, well-meaning, and caring professionals on this planet—who harm, maim, and kill countless people every year.
What a discomforting paradox.
Equity: The Overlooked Quality Domain
Many years back, the Institute of Medicine (IOM) published a list of six “domains” of healthcare quality. You’ve no doubt stumbled across the IOM’s Safe … Timely … Effective … Efficient … Equitable … Patient-Centered mnemonic recipe—STEEEP—for high-quality care.1 In fact, it’s hard to read a journal, attend a medical presentation, or open a local newspaper without finding reference to these domains. It’s all the rage to talk about wrong-site surgery (safe), access to care (timely), comparative-effectiveness research (effective), lean concepts (efficient), and individualized medicine (patient-centered). However, the sixth domain often seems to get the Jan Brady treatment—minimized, marginalized, and oft-forgotten.
The IOM defines equitable care as that which “does not vary in quality because of personal characteristics such as gender, ethnicity, geographic location, and socioeconomic status.”1 To be sure, there is some hum at the national level about issues of equity, especially around healthcare coverage for all. And this is important. However, what appears lost in the rant surrounding the inherent inequities in our tiered health insurance system is that we have blantant inequalities baked into the everyday machinery of our hospitals. And they affect all, regardless of skin color, gender, or insurance status.
Think for a moment about your hospital. Are the type, level, and access to care equal at all times? Does the level of care change when the streetlights come on? How about on weekends and holidays? Do your operating rooms run on Saturdays and Sundays? Can patients get chemotherapy on the weekends? Does your hospital alter its nursing staff ratios after hours? Can you get an ultrasound at midnight? How about a urology or neurosurgery consult at 2 a.m.? How about getting interventional radiology to place an IVC filter on a holiday?
Now scratch a bit closer to home. Does your hospitalist group downstaff on weekends and holidays, even though the volumes probably warrant more coverage? Are your night providers part of your group, or are they moonlighters? Do your after-hours providers cross-cover and admit a reasonable number of patients, or are they frequently overwhelmed? Do they cover patients or admit for services that they don’t typically care for during the day (e.g. ICU, neurosurgical, subspecialty cardiology or oncology patients)? For the intensely ill patients admitted to U.S. hospitals today, should the type and availability of care differ when it’s delivered at 3 p.m. or 3 a.m., Sunday or Monday?
Disregarding the macro-inequities in our societal approach to healthcare, can we even ensure equitable care within our own hospital walls 24 hours a day, seven days a week?
The Answer: An Unfortunate “No”
For the record, I hate working nights, abhor working weekends, and resent working holidays. But the thing I’d hate even more than working nights, weekends, and holidays would be being a patient admitted during a night, weekend, or holiday. To understand why, I have to look no further than my hospital parking lot. During bankers’ hours, I can barely find a parking spot on the top floor of our multilevel parking structure.
Fast-forward to Saturday, and I have my pick of empty football fields’ worth of spots on all floors. Ditto Sundays, nights, and holidays.
Why is it that nationally, a collectively near-trillion-dollar hospital enterprise finds it acceptable to effectively shutter itself for a quarter to a third of the week? Especially when doing so seems to counter their primary mission of providing safe, timely, effective, efficient, equitable, and patient-centered care.
The Weekend Effect
There are, of course, economic and operational reasons to downshift during off hours—some hospitals don’t have the elective procedures to run operating rooms seven days a week, and very few patients want to have their elective colonoscopy at 11 p.m. or their chemotherapy during Thanksgiving dinner. However, in most cases, the reasons for doing so center on hospital staff and physician satisfaction. Most us of just don’t like working off hours. As a result, studies have shown significantly less access to such high-level care as coronary angiography and percutaneous coronary intervention on weekends.2,3
And the effects of this “weekend effect” can be devastating.
A recent paper in the New England Journal of Medicine reported that for every 1,000 patients admitted with a myocardial infarction on a weekend, nine more would die than a comparable group admitted during the week.4 Their offense? Having the misfortune to get ill on a Saturday morning. The authors concluded that this higher mortality was secondary to a lower rate of invasive cardiac procedures, presumably because they were less available. And the weekend effect isn’t just limited to coronary care. Poorer outcomes, including higher mortality rates, have been reported for weekend admissions to the neonatal ICU and adult ICU, as well as admission for epiglottitis, ruptured abdominal aortic aneurysms, and pulmonary embolism.5,6,7
So let’s connect the dots: A system designed with inequal access to lifesaving therapies and appropriate staffing results in worse outcomes, more harm, more deaths.
To be clear, 98,000 people don’t die every year because of my disdain for working nights and weekends. This is a much deeper problem that hinges on many unsatisfactory systems working unsatisfactorily in tandem (e.g. see the other five IOM domains of quality care). Furthermore, I don’t mean to suggest that hospitalists necessarily have a lot of say in cardiac catheterization schedules. Yet we do control our own systems of care—how many patients we admit and cover during a shift, how strongly we advocate for timely testing and consultation, how we staff weekends and cover patients at night.
And, more and more, we are in a position to enhance the care delivery systems of the hospital and its providers that surround us. With that comes a responsibility to ensure that these systems are highly functioning and equitable, regardless of the time of day, day of the week.
If we are going to fundamentally enhance the quality of care, then we have to design safer systems of care. It will take time and resources to alter many of our bruised systems of care, but we can begin by at least ensuring equity in how we deliver care at our own institutions. That is, unless we are comfortable with a system perfectly designed to harm. TH
Dr. Glasheen is associate professor of medicine at the University of Colorado Denver, where he serves as director of the Hospital Medicine Program and the Hospitalist Training Program, and as associate program director of the Internal Medicine Residency Program.
References
- Institute of Medicine (IOM). Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, D.C.: National Academy Press; 2001.
- Bell CM, Redelmeier DA. Waiting for urgent procedures on the weekend among emergently hospitalized patients. Am J Med. 2004:117(3):175-181.
- Magid DJ, Wang Y, Herrin J, et al. Relationship between time of day, day of week, timeliness of reperfusion, and in-hospital mortality for patients with acute ST-segment elevation myocardial infarction. JAMA. 2005;294(7):803-812.
- Kostis WJ, Demissie K, Marcella SW, Shao YH, Wilson AC, Moreyra AE. Weekend versus weekday admission and mortality from myocardial infarction. N Eng J Med. 2007;356 (11):1099-1109.
- Hendry RA. The weekend—a dangerous time to be born? Br J Obstet Gynaecol. 1981;88(12):1200-1203.
- Barnett MJ, Kaboli PJ, Sirio CA, Rosenthal GE. Day of the week of intensive care admission and patient outcomes: a multisite regional evaluation. Med Care. 2002;40(6):530-539.
- Bell CM, Redelmeier DA. Mortality among patients admitted to hospitals on weekends as compared with weekdays. N Eng J Med. 2001;345(9):663-668.
If every system is perfectly designed to get the results it gets …
And if 15 million patients are harmed every year from medical care …
And if as many as 98,000 people die every year due to medical errors in hospitals …
Then what does that say about the system we have designed?
A System Designed to Competently Hurt Many
By now you’ve no doubt heard, read, and possibly even uttered the above facts and figures yourself. I think we all have our opinions about the veracity of these numbers, but I don’t think any of us would argue with the sentiment. The U.S. healthcare system comprises the most competent, compassionate, well-meaning, and caring professionals on this planet—who harm, maim, and kill countless people every year.
What a discomforting paradox.
Equity: The Overlooked Quality Domain
Many years back, the Institute of Medicine (IOM) published a list of six “domains” of healthcare quality. You’ve no doubt stumbled across the IOM’s Safe … Timely … Effective … Efficient … Equitable … Patient-Centered mnemonic recipe—STEEEP—for high-quality care.1 In fact, it’s hard to read a journal, attend a medical presentation, or open a local newspaper without finding reference to these domains. It’s all the rage to talk about wrong-site surgery (safe), access to care (timely), comparative-effectiveness research (effective), lean concepts (efficient), and individualized medicine (patient-centered). However, the sixth domain often seems to get the Jan Brady treatment—minimized, marginalized, and oft-forgotten.
The IOM defines equitable care as that which “does not vary in quality because of personal characteristics such as gender, ethnicity, geographic location, and socioeconomic status.”1 To be sure, there is some hum at the national level about issues of equity, especially around healthcare coverage for all. And this is important. However, what appears lost in the rant surrounding the inherent inequities in our tiered health insurance system is that we have blantant inequalities baked into the everyday machinery of our hospitals. And they affect all, regardless of skin color, gender, or insurance status.
Think for a moment about your hospital. Are the type, level, and access to care equal at all times? Does the level of care change when the streetlights come on? How about on weekends and holidays? Do your operating rooms run on Saturdays and Sundays? Can patients get chemotherapy on the weekends? Does your hospital alter its nursing staff ratios after hours? Can you get an ultrasound at midnight? How about a urology or neurosurgery consult at 2 a.m.? How about getting interventional radiology to place an IVC filter on a holiday?
Now scratch a bit closer to home. Does your hospitalist group downstaff on weekends and holidays, even though the volumes probably warrant more coverage? Are your night providers part of your group, or are they moonlighters? Do your after-hours providers cross-cover and admit a reasonable number of patients, or are they frequently overwhelmed? Do they cover patients or admit for services that they don’t typically care for during the day (e.g. ICU, neurosurgical, subspecialty cardiology or oncology patients)? For the intensely ill patients admitted to U.S. hospitals today, should the type and availability of care differ when it’s delivered at 3 p.m. or 3 a.m., Sunday or Monday?
Disregarding the macro-inequities in our societal approach to healthcare, can we even ensure equitable care within our own hospital walls 24 hours a day, seven days a week?
The Answer: An Unfortunate “No”
For the record, I hate working nights, abhor working weekends, and resent working holidays. But the thing I’d hate even more than working nights, weekends, and holidays would be being a patient admitted during a night, weekend, or holiday. To understand why, I have to look no further than my hospital parking lot. During bankers’ hours, I can barely find a parking spot on the top floor of our multilevel parking structure.
Fast-forward to Saturday, and I have my pick of empty football fields’ worth of spots on all floors. Ditto Sundays, nights, and holidays.
Why is it that nationally, a collectively near-trillion-dollar hospital enterprise finds it acceptable to effectively shutter itself for a quarter to a third of the week? Especially when doing so seems to counter their primary mission of providing safe, timely, effective, efficient, equitable, and patient-centered care.
The Weekend Effect
There are, of course, economic and operational reasons to downshift during off hours—some hospitals don’t have the elective procedures to run operating rooms seven days a week, and very few patients want to have their elective colonoscopy at 11 p.m. or their chemotherapy during Thanksgiving dinner. However, in most cases, the reasons for doing so center on hospital staff and physician satisfaction. Most us of just don’t like working off hours. As a result, studies have shown significantly less access to such high-level care as coronary angiography and percutaneous coronary intervention on weekends.2,3
And the effects of this “weekend effect” can be devastating.
A recent paper in the New England Journal of Medicine reported that for every 1,000 patients admitted with a myocardial infarction on a weekend, nine more would die than a comparable group admitted during the week.4 Their offense? Having the misfortune to get ill on a Saturday morning. The authors concluded that this higher mortality was secondary to a lower rate of invasive cardiac procedures, presumably because they were less available. And the weekend effect isn’t just limited to coronary care. Poorer outcomes, including higher mortality rates, have been reported for weekend admissions to the neonatal ICU and adult ICU, as well as admission for epiglottitis, ruptured abdominal aortic aneurysms, and pulmonary embolism.5,6,7
So let’s connect the dots: A system designed with inequal access to lifesaving therapies and appropriate staffing results in worse outcomes, more harm, more deaths.
To be clear, 98,000 people don’t die every year because of my disdain for working nights and weekends. This is a much deeper problem that hinges on many unsatisfactory systems working unsatisfactorily in tandem (e.g. see the other five IOM domains of quality care). Furthermore, I don’t mean to suggest that hospitalists necessarily have a lot of say in cardiac catheterization schedules. Yet we do control our own systems of care—how many patients we admit and cover during a shift, how strongly we advocate for timely testing and consultation, how we staff weekends and cover patients at night.
And, more and more, we are in a position to enhance the care delivery systems of the hospital and its providers that surround us. With that comes a responsibility to ensure that these systems are highly functioning and equitable, regardless of the time of day, day of the week.
If we are going to fundamentally enhance the quality of care, then we have to design safer systems of care. It will take time and resources to alter many of our bruised systems of care, but we can begin by at least ensuring equity in how we deliver care at our own institutions. That is, unless we are comfortable with a system perfectly designed to harm. TH
Dr. Glasheen is associate professor of medicine at the University of Colorado Denver, where he serves as director of the Hospital Medicine Program and the Hospitalist Training Program, and as associate program director of the Internal Medicine Residency Program.
References
- Institute of Medicine (IOM). Crossing the Quality Chasm: A New Health System for the 21st Century. Washington, D.C.: National Academy Press; 2001.
- Bell CM, Redelmeier DA. Waiting for urgent procedures on the weekend among emergently hospitalized patients. Am J Med. 2004:117(3):175-181.
- Magid DJ, Wang Y, Herrin J, et al. Relationship between time of day, day of week, timeliness of reperfusion, and in-hospital mortality for patients with acute ST-segment elevation myocardial infarction. JAMA. 2005;294(7):803-812.
- Kostis WJ, Demissie K, Marcella SW, Shao YH, Wilson AC, Moreyra AE. Weekend versus weekday admission and mortality from myocardial infarction. N Eng J Med. 2007;356 (11):1099-1109.
- Hendry RA. The weekend—a dangerous time to be born? Br J Obstet Gynaecol. 1981;88(12):1200-1203.
- Barnett MJ, Kaboli PJ, Sirio CA, Rosenthal GE. Day of the week of intensive care admission and patient outcomes: a multisite regional evaluation. Med Care. 2002;40(6):530-539.
- Bell CM, Redelmeier DA. Mortality among patients admitted to hospitals on weekends as compared with weekdays. N Eng J Med. 2001;345(9):663-668.
Market Watch
New Drugs, Devices, Indications, and Approvals
- Alglucosidase alfa (Lumizyme) has been approved by the FDA for the treatment of patients older than 8 with late (noninfantile) onset Pompe disease (GAA deficiency) who do not have evidence of cardiac hypertrophy.1
- Dutasteride and tamsulosin (Jalyn) has been approved by the FDA in a fixed-dose combination product for treating symptomatic benign prostatic hypertrophy.2
- Gatifloxacin ophthalmic solution 0.5% (Zymaxid) has been approved by the FDA to treat bacterial conjunctivitis caused by susceptible bacterial strains.3 It has the highest concentration of gatifloxacin ophthalmic on the U.S. market.
- Memantine hydrochloride (Namenda XR) has been approved by the FDA as an extended-release product to treat moderate to severe dementia of the Alzheimer's type.4 It is available as a 28 mg, once-daily dosage form. The immediate-release product is dosed twice daily.
- Mesenchymal stem cells for intravenous infusion (MSCs, Prochymal) formulated into Prochymal has been granted orphan drug status to treat Type 1 diabetes mellitus (T1DM).5 This agent is in Phase 2 clinical trials in a collaboration with the Juvenile Diabetes Research Foundation as a treatment for patients with newly diagnosed T1DM.
- Mometasone furoate/formoterol fumarate (Dulera) has been approved by the FDA as a combination treatment for asthmatics aged 12 and older.6
- Naproxen/esomeprazole (Vimovo) has been approved by the FDA as a fixed combination for patients with arthritis who are at risk for developing gastric ulcers.7
- Pioglitazone/metformin (ACTOplus met XR) has been approved by the FDA in an extended-release fixed combination for treating Type 2 diabetes mellitus (T2DM) as an adjunct to diet and exercise.8
- Ranibizumab (Lucentis) has received a new indication from the FDA for treating retinal vein occlusion.9 The original approval of ranibizumab was for wet age-related macular degeneration.
- Tramadol HCl, orally disintegrating tablets (Rybix ODT), have been approved by the FDA for treating moderate to moderately severe pain in patients 16 years of age and older.10
Pipeline
- Dronedarone (Multaq) is being investigated as to whether it can reduce major cardiovascular events in patients being treated with the agent for atrial fibrillation.11 Study endpoints will include a reduction in a major cardiovascular event such as stroke or myocardial infarction, or a reduction in cardiovascular hospitalization or death.
- The combination product ibuprofen and famotidine (to be known as Duexa) has been filed as a new drug application (NDA) for reducing the risk of developing upper gastrointestinal (GI) ulcers in patients with pain and arthritis.12 Two Phase 3 trials showed about a 50% reduction in GI ulcers in combination treatment compared with ibuprofen alone.
- Rifaximin (Xifaxan) has been submitted to the FDA at its higher dose (550 mg) for treatment of nonconstipation irritable bowel syndrome (Non-C-IBS) and IBS-related bloating.13 Rifaximin already has approval from the FDA for hepatic encephalopathy and diarrhea.
- TC-5214 has begun Phase 3 clinical trials.14 It is a nicotinic channel blocker for the adjunctive treatment of major depressive disorder in adults who have had an inadequate response to selective serotonin reuptake inhibitors (SSRIs) or serotonin/norepinephrine reuptake inhibitors (SNRIs). A Phase 2 study evaluating TC-5214 as a second-line (“switch”) monotherapy is planned for this year.
- An NDA for vilazodone has been filed for treating major depressive disorder.15 It is a dual-acting potent and selective serotonin reuptake inhibitor and a 5-HT1A receptor partial agonist.
Safety, Warnings, and Label Changes
- Tramadol and tramadol/acetaminophen have undergone a label change related to strengthened warnings of the risk of suicide for patients who are addiction-prone, taking tranquilizers, drinking alcohol, or taking other central-nervous-system-active drugs.16 Addictive effects might occur when tramadol is combined with alcohol, other opioids, or illicit drugs that have central-nervous-system-depressive effects. Serious potential consequences of overdosage are CNS and/or respiratory depression, and death.
- Earlier this year, the FDA once again updated the warning related to severe liver injury related to the use of propylthiouracil (PTU).17 This time, PTU has garnered a boxed warning, which includes reports of severe hepatotoxicity and acute liver failure in both adults and children. Some of these reactions were fatal. Use of PTU should be reserved for patients who do not tolerate other treatments for hyperthyroidism, such as methimazole, radioactive iodine, or are not surgery candidates. PTU might be preferred over methimazole just before and/or during the first trimester of pregnancy due to the occurrence of birth defects with methimazole during this timeframe. A medication guide has been developed and is to be given to patients when they fill PTU prescriptions. The guide alerts patients to the signs and symptoms of hepatotoxicity. TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Genzyme receives FDA approval for lumizyme for Pompe disease. Business Wire website. Available at: www.businesswire.com/portal/site/home/email/alert/?ndmViewId=news_view&newsLang=en&newsId=20100525006514. Accessed June 30, 2010.
- Dennis M. FDA approves GlaxoSmithKline’s combination BPH drug Jalyn. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=B5F703CADB0347DF970A7B7542D99982&logRowId=369156. Accessed June 30, 2010.
- Allergan receives FDA approval for Zymaxid ophthalmic solution. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/189397.php. Accessed June 29, 2010.
- Forest and Merz announce FDA approval of Namenda XR for the treatment of moderate to severe dementia of the Alzheimer’s type. Forest Laboratories website. Available at: http://www.frx.com/news/PressRelease.aspx?ID=1440385. Accessed June 30, 2010.
- Osiris Therapeutics receives FDA orphan drug designation for stem cell treatment for Type 1 diabetes. Osiris Therapeutics website. Available at: http://osiris.com/pdf/2010-05-04%20T1D%20Orphan%20Drug%20Designation.pdf. Accessed June 30, 2010.
- FDA approves Merck’s new lung drug Dulera. The Economic Times website. Available at: http://economictimes.indiatimes.com/articleshow/6087079.cms. Accessed June 30, 2010.
- Dane L. FDA approves AstraZeneca, Pozen’s Vimovo. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=0C2915EE4D2D47D287D41D90DFE4240D. Accessed June 30, 2010.
- ACTOplus met XR available for diabetes. Monthly Prescribing Reference website. Available at: http://www.empr.com/actoplus-met-xr-available-for-diabetes/article/172985/. Accessed June 30, 2010.
- Dennis M. Roche’s Lucentis garners expanded FDA approval for macular oedema. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=5146664F5A4C48108209D15EA744A614&logRowId=370643. Accessed June 30, 2010.
- Rybix ODT launched for moderate to moderately severe pain. Monthly Prescribing Reference website. Available at: http://www.empr.com/rybix-odt-launched-for-moderate-to-moderately-severe-pain/article/172743/. Accessed June 30, 3010.
- Dennis M. Sanofi-Aventis begins late-stage study of Multaq in patients with permanent AF. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E43104A2DB604649A812E69C58F89304&logRowId=364228. Accessed July 7, 2010.
- FDA accepts NDA for Duexa for ulcer risk reduction in arthritis patients. Monthly Prescribing Reference website. Available at: http://www.empr.com/fda-accepts-nda-for-duexa-for-ulcer-risk-reduction-in-arthritis-patients/article/171054/. Accessed July 7, 2010.
- Salix Pharmaceuticals announces NDA submission for Xifaxan550 for treatment of non-constipation irritable bowel syndrome. Salix Pharmaceuticals website. Available at: http://salix.com/news/stories/20100608.aspx. Accessed June 9, 2010.
- AstraZeneca and Targacept initiate Phase 3 clinical development of TC-5214 as an adjunct treatment for major depressive disorder. Targacept website. Available at: http://www.targacept.com/wt/page/pr_1277240993. Accessed June 29, 2010.
- Clinical Data, Inc. announces FDA acceptance of new drug application for Vilazodone for the treatment of major depressive disorder. Clinical Data Inc. website. Available at: http://clda.com/uploads/CLDA%20NDA%20acceptance%20FINAL.pdf. Accessed July 7, 2010.
- Ultram (tramadol hydrochloride), Ultracet (tramadol hydrochloride/acetaminophen): Label change. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm213264.htm. Accessed July 8, 2010.
- FDA Drug Safety Communication: New boxed warning on severe liver injury with propylthiouracil. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm209023.htm. Accessed July 8, 2010.
New Drugs, Devices, Indications, and Approvals
- Alglucosidase alfa (Lumizyme) has been approved by the FDA for the treatment of patients older than 8 with late (noninfantile) onset Pompe disease (GAA deficiency) who do not have evidence of cardiac hypertrophy.1
- Dutasteride and tamsulosin (Jalyn) has been approved by the FDA in a fixed-dose combination product for treating symptomatic benign prostatic hypertrophy.2
- Gatifloxacin ophthalmic solution 0.5% (Zymaxid) has been approved by the FDA to treat bacterial conjunctivitis caused by susceptible bacterial strains.3 It has the highest concentration of gatifloxacin ophthalmic on the U.S. market.
- Memantine hydrochloride (Namenda XR) has been approved by the FDA as an extended-release product to treat moderate to severe dementia of the Alzheimer's type.4 It is available as a 28 mg, once-daily dosage form. The immediate-release product is dosed twice daily.
- Mesenchymal stem cells for intravenous infusion (MSCs, Prochymal) formulated into Prochymal has been granted orphan drug status to treat Type 1 diabetes mellitus (T1DM).5 This agent is in Phase 2 clinical trials in a collaboration with the Juvenile Diabetes Research Foundation as a treatment for patients with newly diagnosed T1DM.
- Mometasone furoate/formoterol fumarate (Dulera) has been approved by the FDA as a combination treatment for asthmatics aged 12 and older.6
- Naproxen/esomeprazole (Vimovo) has been approved by the FDA as a fixed combination for patients with arthritis who are at risk for developing gastric ulcers.7
- Pioglitazone/metformin (ACTOplus met XR) has been approved by the FDA in an extended-release fixed combination for treating Type 2 diabetes mellitus (T2DM) as an adjunct to diet and exercise.8
- Ranibizumab (Lucentis) has received a new indication from the FDA for treating retinal vein occlusion.9 The original approval of ranibizumab was for wet age-related macular degeneration.
- Tramadol HCl, orally disintegrating tablets (Rybix ODT), have been approved by the FDA for treating moderate to moderately severe pain in patients 16 years of age and older.10
Pipeline
- Dronedarone (Multaq) is being investigated as to whether it can reduce major cardiovascular events in patients being treated with the agent for atrial fibrillation.11 Study endpoints will include a reduction in a major cardiovascular event such as stroke or myocardial infarction, or a reduction in cardiovascular hospitalization or death.
- The combination product ibuprofen and famotidine (to be known as Duexa) has been filed as a new drug application (NDA) for reducing the risk of developing upper gastrointestinal (GI) ulcers in patients with pain and arthritis.12 Two Phase 3 trials showed about a 50% reduction in GI ulcers in combination treatment compared with ibuprofen alone.
- Rifaximin (Xifaxan) has been submitted to the FDA at its higher dose (550 mg) for treatment of nonconstipation irritable bowel syndrome (Non-C-IBS) and IBS-related bloating.13 Rifaximin already has approval from the FDA for hepatic encephalopathy and diarrhea.
- TC-5214 has begun Phase 3 clinical trials.14 It is a nicotinic channel blocker for the adjunctive treatment of major depressive disorder in adults who have had an inadequate response to selective serotonin reuptake inhibitors (SSRIs) or serotonin/norepinephrine reuptake inhibitors (SNRIs). A Phase 2 study evaluating TC-5214 as a second-line (“switch”) monotherapy is planned for this year.
- An NDA for vilazodone has been filed for treating major depressive disorder.15 It is a dual-acting potent and selective serotonin reuptake inhibitor and a 5-HT1A receptor partial agonist.
Safety, Warnings, and Label Changes
- Tramadol and tramadol/acetaminophen have undergone a label change related to strengthened warnings of the risk of suicide for patients who are addiction-prone, taking tranquilizers, drinking alcohol, or taking other central-nervous-system-active drugs.16 Addictive effects might occur when tramadol is combined with alcohol, other opioids, or illicit drugs that have central-nervous-system-depressive effects. Serious potential consequences of overdosage are CNS and/or respiratory depression, and death.
- Earlier this year, the FDA once again updated the warning related to severe liver injury related to the use of propylthiouracil (PTU).17 This time, PTU has garnered a boxed warning, which includes reports of severe hepatotoxicity and acute liver failure in both adults and children. Some of these reactions were fatal. Use of PTU should be reserved for patients who do not tolerate other treatments for hyperthyroidism, such as methimazole, radioactive iodine, or are not surgery candidates. PTU might be preferred over methimazole just before and/or during the first trimester of pregnancy due to the occurrence of birth defects with methimazole during this timeframe. A medication guide has been developed and is to be given to patients when they fill PTU prescriptions. The guide alerts patients to the signs and symptoms of hepatotoxicity. TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Genzyme receives FDA approval for lumizyme for Pompe disease. Business Wire website. Available at: www.businesswire.com/portal/site/home/email/alert/?ndmViewId=news_view&newsLang=en&newsId=20100525006514. Accessed June 30, 2010.
- Dennis M. FDA approves GlaxoSmithKline’s combination BPH drug Jalyn. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=B5F703CADB0347DF970A7B7542D99982&logRowId=369156. Accessed June 30, 2010.
- Allergan receives FDA approval for Zymaxid ophthalmic solution. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/189397.php. Accessed June 29, 2010.
- Forest and Merz announce FDA approval of Namenda XR for the treatment of moderate to severe dementia of the Alzheimer’s type. Forest Laboratories website. Available at: http://www.frx.com/news/PressRelease.aspx?ID=1440385. Accessed June 30, 2010.
- Osiris Therapeutics receives FDA orphan drug designation for stem cell treatment for Type 1 diabetes. Osiris Therapeutics website. Available at: http://osiris.com/pdf/2010-05-04%20T1D%20Orphan%20Drug%20Designation.pdf. Accessed June 30, 2010.
- FDA approves Merck’s new lung drug Dulera. The Economic Times website. Available at: http://economictimes.indiatimes.com/articleshow/6087079.cms. Accessed June 30, 2010.
- Dane L. FDA approves AstraZeneca, Pozen’s Vimovo. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=0C2915EE4D2D47D287D41D90DFE4240D. Accessed June 30, 2010.
- ACTOplus met XR available for diabetes. Monthly Prescribing Reference website. Available at: http://www.empr.com/actoplus-met-xr-available-for-diabetes/article/172985/. Accessed June 30, 2010.
- Dennis M. Roche’s Lucentis garners expanded FDA approval for macular oedema. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=5146664F5A4C48108209D15EA744A614&logRowId=370643. Accessed June 30, 2010.
- Rybix ODT launched for moderate to moderately severe pain. Monthly Prescribing Reference website. Available at: http://www.empr.com/rybix-odt-launched-for-moderate-to-moderately-severe-pain/article/172743/. Accessed June 30, 3010.
- Dennis M. Sanofi-Aventis begins late-stage study of Multaq in patients with permanent AF. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E43104A2DB604649A812E69C58F89304&logRowId=364228. Accessed July 7, 2010.
- FDA accepts NDA for Duexa for ulcer risk reduction in arthritis patients. Monthly Prescribing Reference website. Available at: http://www.empr.com/fda-accepts-nda-for-duexa-for-ulcer-risk-reduction-in-arthritis-patients/article/171054/. Accessed July 7, 2010.
- Salix Pharmaceuticals announces NDA submission for Xifaxan550 for treatment of non-constipation irritable bowel syndrome. Salix Pharmaceuticals website. Available at: http://salix.com/news/stories/20100608.aspx. Accessed June 9, 2010.
- AstraZeneca and Targacept initiate Phase 3 clinical development of TC-5214 as an adjunct treatment for major depressive disorder. Targacept website. Available at: http://www.targacept.com/wt/page/pr_1277240993. Accessed June 29, 2010.
- Clinical Data, Inc. announces FDA acceptance of new drug application for Vilazodone for the treatment of major depressive disorder. Clinical Data Inc. website. Available at: http://clda.com/uploads/CLDA%20NDA%20acceptance%20FINAL.pdf. Accessed July 7, 2010.
- Ultram (tramadol hydrochloride), Ultracet (tramadol hydrochloride/acetaminophen): Label change. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm213264.htm. Accessed July 8, 2010.
- FDA Drug Safety Communication: New boxed warning on severe liver injury with propylthiouracil. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm209023.htm. Accessed July 8, 2010.
New Drugs, Devices, Indications, and Approvals
- Alglucosidase alfa (Lumizyme) has been approved by the FDA for the treatment of patients older than 8 with late (noninfantile) onset Pompe disease (GAA deficiency) who do not have evidence of cardiac hypertrophy.1
- Dutasteride and tamsulosin (Jalyn) has been approved by the FDA in a fixed-dose combination product for treating symptomatic benign prostatic hypertrophy.2
- Gatifloxacin ophthalmic solution 0.5% (Zymaxid) has been approved by the FDA to treat bacterial conjunctivitis caused by susceptible bacterial strains.3 It has the highest concentration of gatifloxacin ophthalmic on the U.S. market.
- Memantine hydrochloride (Namenda XR) has been approved by the FDA as an extended-release product to treat moderate to severe dementia of the Alzheimer's type.4 It is available as a 28 mg, once-daily dosage form. The immediate-release product is dosed twice daily.
- Mesenchymal stem cells for intravenous infusion (MSCs, Prochymal) formulated into Prochymal has been granted orphan drug status to treat Type 1 diabetes mellitus (T1DM).5 This agent is in Phase 2 clinical trials in a collaboration with the Juvenile Diabetes Research Foundation as a treatment for patients with newly diagnosed T1DM.
- Mometasone furoate/formoterol fumarate (Dulera) has been approved by the FDA as a combination treatment for asthmatics aged 12 and older.6
- Naproxen/esomeprazole (Vimovo) has been approved by the FDA as a fixed combination for patients with arthritis who are at risk for developing gastric ulcers.7
- Pioglitazone/metformin (ACTOplus met XR) has been approved by the FDA in an extended-release fixed combination for treating Type 2 diabetes mellitus (T2DM) as an adjunct to diet and exercise.8
- Ranibizumab (Lucentis) has received a new indication from the FDA for treating retinal vein occlusion.9 The original approval of ranibizumab was for wet age-related macular degeneration.
- Tramadol HCl, orally disintegrating tablets (Rybix ODT), have been approved by the FDA for treating moderate to moderately severe pain in patients 16 years of age and older.10
Pipeline
- Dronedarone (Multaq) is being investigated as to whether it can reduce major cardiovascular events in patients being treated with the agent for atrial fibrillation.11 Study endpoints will include a reduction in a major cardiovascular event such as stroke or myocardial infarction, or a reduction in cardiovascular hospitalization or death.
- The combination product ibuprofen and famotidine (to be known as Duexa) has been filed as a new drug application (NDA) for reducing the risk of developing upper gastrointestinal (GI) ulcers in patients with pain and arthritis.12 Two Phase 3 trials showed about a 50% reduction in GI ulcers in combination treatment compared with ibuprofen alone.
- Rifaximin (Xifaxan) has been submitted to the FDA at its higher dose (550 mg) for treatment of nonconstipation irritable bowel syndrome (Non-C-IBS) and IBS-related bloating.13 Rifaximin already has approval from the FDA for hepatic encephalopathy and diarrhea.
- TC-5214 has begun Phase 3 clinical trials.14 It is a nicotinic channel blocker for the adjunctive treatment of major depressive disorder in adults who have had an inadequate response to selective serotonin reuptake inhibitors (SSRIs) or serotonin/norepinephrine reuptake inhibitors (SNRIs). A Phase 2 study evaluating TC-5214 as a second-line (“switch”) monotherapy is planned for this year.
- An NDA for vilazodone has been filed for treating major depressive disorder.15 It is a dual-acting potent and selective serotonin reuptake inhibitor and a 5-HT1A receptor partial agonist.
Safety, Warnings, and Label Changes
- Tramadol and tramadol/acetaminophen have undergone a label change related to strengthened warnings of the risk of suicide for patients who are addiction-prone, taking tranquilizers, drinking alcohol, or taking other central-nervous-system-active drugs.16 Addictive effects might occur when tramadol is combined with alcohol, other opioids, or illicit drugs that have central-nervous-system-depressive effects. Serious potential consequences of overdosage are CNS and/or respiratory depression, and death.
- Earlier this year, the FDA once again updated the warning related to severe liver injury related to the use of propylthiouracil (PTU).17 This time, PTU has garnered a boxed warning, which includes reports of severe hepatotoxicity and acute liver failure in both adults and children. Some of these reactions were fatal. Use of PTU should be reserved for patients who do not tolerate other treatments for hyperthyroidism, such as methimazole, radioactive iodine, or are not surgery candidates. PTU might be preferred over methimazole just before and/or during the first trimester of pregnancy due to the occurrence of birth defects with methimazole during this timeframe. A medication guide has been developed and is to be given to patients when they fill PTU prescriptions. The guide alerts patients to the signs and symptoms of hepatotoxicity. TH
Michele B. Kaufman, PharmD, BSc, RPh, is a freelance medical writer based in New York City and a clinical pharmacist at New York Downtown Hospital.
References
- Genzyme receives FDA approval for lumizyme for Pompe disease. Business Wire website. Available at: www.businesswire.com/portal/site/home/email/alert/?ndmViewId=news_view&newsLang=en&newsId=20100525006514. Accessed June 30, 2010.
- Dennis M. FDA approves GlaxoSmithKline’s combination BPH drug Jalyn. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=B5F703CADB0347DF970A7B7542D99982&logRowId=369156. Accessed June 30, 2010.
- Allergan receives FDA approval for Zymaxid ophthalmic solution. Medical News Today website. Available at: http://www.medicalnewstoday.com/articles/189397.php. Accessed June 29, 2010.
- Forest and Merz announce FDA approval of Namenda XR for the treatment of moderate to severe dementia of the Alzheimer’s type. Forest Laboratories website. Available at: http://www.frx.com/news/PressRelease.aspx?ID=1440385. Accessed June 30, 2010.
- Osiris Therapeutics receives FDA orphan drug designation for stem cell treatment for Type 1 diabetes. Osiris Therapeutics website. Available at: http://osiris.com/pdf/2010-05-04%20T1D%20Orphan%20Drug%20Designation.pdf. Accessed June 30, 2010.
- FDA approves Merck’s new lung drug Dulera. The Economic Times website. Available at: http://economictimes.indiatimes.com/articleshow/6087079.cms. Accessed June 30, 2010.
- Dane L. FDA approves AstraZeneca, Pozen’s Vimovo. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=0C2915EE4D2D47D287D41D90DFE4240D. Accessed June 30, 2010.
- ACTOplus met XR available for diabetes. Monthly Prescribing Reference website. Available at: http://www.empr.com/actoplus-met-xr-available-for-diabetes/article/172985/. Accessed June 30, 2010.
- Dennis M. Roche’s Lucentis garners expanded FDA approval for macular oedema. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=5146664F5A4C48108209D15EA744A614&logRowId=370643. Accessed June 30, 2010.
- Rybix ODT launched for moderate to moderately severe pain. Monthly Prescribing Reference website. Available at: http://www.empr.com/rybix-odt-launched-for-moderate-to-moderately-severe-pain/article/172743/. Accessed June 30, 3010.
- Dennis M. Sanofi-Aventis begins late-stage study of Multaq in patients with permanent AF. FirstWord website. Available at: http://www.firstwordplus.com/Fws.do?articleid=E43104A2DB604649A812E69C58F89304&logRowId=364228. Accessed July 7, 2010.
- FDA accepts NDA for Duexa for ulcer risk reduction in arthritis patients. Monthly Prescribing Reference website. Available at: http://www.empr.com/fda-accepts-nda-for-duexa-for-ulcer-risk-reduction-in-arthritis-patients/article/171054/. Accessed July 7, 2010.
- Salix Pharmaceuticals announces NDA submission for Xifaxan550 for treatment of non-constipation irritable bowel syndrome. Salix Pharmaceuticals website. Available at: http://salix.com/news/stories/20100608.aspx. Accessed June 9, 2010.
- AstraZeneca and Targacept initiate Phase 3 clinical development of TC-5214 as an adjunct treatment for major depressive disorder. Targacept website. Available at: http://www.targacept.com/wt/page/pr_1277240993. Accessed June 29, 2010.
- Clinical Data, Inc. announces FDA acceptance of new drug application for Vilazodone for the treatment of major depressive disorder. Clinical Data Inc. website. Available at: http://clda.com/uploads/CLDA%20NDA%20acceptance%20FINAL.pdf. Accessed July 7, 2010.
- Ultram (tramadol hydrochloride), Ultracet (tramadol hydrochloride/acetaminophen): Label change. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm213264.htm. Accessed July 8, 2010.
- FDA Drug Safety Communication: New boxed warning on severe liver injury with propylthiouracil. U.S. Food and Drug Administration website. Available at: http://www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm209023.htm. Accessed July 8, 2010.
Innovative Use of Scribes in the Inpatient Setting
Over the past couple of decades, the ever-increasing medical-legal, financial, and patient-safety demands have led to a mushrooming of the amount of documentation needed during patient care, especially in the inpatient setting. With the advent of HM, that burden simply shifted to hospitalists, along with rapidly expanding patient loads as PCPs started delegating inpatient care. The problem was further compounded in teaching hospitals by a gradual decline in resident support as patient caps and work-hour limits were instituted.
Not surprisingly, the quality and accuracy of physicians’ progress notes started to suffer. It became frequently difficult to remember the details of encounters from earlier in the day, and fatigue sometimes led to details essential to continuity of care being left out completely.
A potential—and very intriguing—solution presented itself, however, as we were designing family-centered team rounds. Apparently, the ED group at the adjacent Abbott-Northwestern Hospital in Minneapolis had been using scribes for several years. The scribes captured the providers’ notes during each encounter, and the group had developed a polished system of scribe recruitment and training (mostly pre-med students awaiting acceptance to medical schools).
But would this model work in the inpatient setting where workflow was completely different? This was a total unknown, given that there has been no precedent in the literature about inpatient scribe utilization. Although relatively meager in number, the published articles about outpatient use of scribes were generally favorable, with the authors noting improved productivity and increased patient and staff satisfaction.1,2,3,4
Operationally, the main difference between inpatient and outpatient scribe use lied in the rounding mechanism itself; it was not just a provider-patient interaction, as in the ED, but rather a sometimes cacophonous collection of multiple inputs from families, providers, students, residents, nurses, and ancillary staff. The challenge here was to train the scribes (natural ability played a significant role) to produce a coherent document out of this interaction; the document had to accurately reflect current status and plans of care. The first step was to create a template for the daily progress notes with all the necessary headings, as well as age-appropriate normal exams, with the ability to directly import from the EMR such dynamic data as vital signs, medication lists, and lab results.
Each morning prior to rounds, the scribe was provided a list of patients to be rounded on, and the scribe spent about 30 minutes creating partial notes on each patient, as well as a brief patient identification statement. This proved to be a significant time-saver during rounds and allowed the scribes to keep up with the care team. Each note was pre-pended with the following statements, as recommended by the hospital’s risk-management team:
- I, (scribe name), am serving as a scribe to document services personally performed by (hospitalist name), MD, based on my observation and the provider’s statements to me.
- I, (hospitalist name), MD, attest that (scribe name) is acting in a scribe capacity, has observed my performance of the services, and has documented them in accordance with my direction.
A computer-on-wheels (COW) station was assigned to the scribes and accompanied the rounding team into the patient rooms, making it possible for them to follow the interactions in real time. The scribes were encouraged to hold their hands up and interrupt the conversation at any time if they needed clarification, something that happened rather infrequently. On the other hand, the care team was directed to speak clearly and the residents were instructed to present their cases using the same format as the progress-note template, thus minimizing guesswork on the part of the scribes. The only part of the interaction the scribes could not transcribe directly was the physical examination, and typically the attending physician dictated abnormal findings as well as systems not assessed, if any, at the end. All the scribe-generated notes were forwarded to the rounding provider’s EMR inbox and reviewed by the latter, amended when necessary, and signed electronically. The review of the scribe-generated notes proved to be initially a time-consuming process, typically two hours. However, as the scribes gained experience, the time commitment on the part of the providers decreased rapidly; after about three months, the time spent was about 30 minutes on busy days, even less otherwise, with a concomitant increase in satisfaction with the scribing process.
Not too unexpectedly, our transcription service initially pushed back on this idea because of their fear of job loss. However, this issue was resolved by eliminating the need for outsourcing overflow dictations. In addition, the relative terseness of the scribed notes did not sit well initially with some of our colleagues used to excessive verbosity, and this issue again gradually faded away as physicians realized that all the necessary information for care was preserved.
Does a scribe program make sense in all situations? Not likely. This kind of setup will work best in busy tertiary-care centers with residency programs and family-centered team rounds. Furthermore, a nearby college or university is a must if cost-efficient hiring of pre-med students is to be possible.
Walid Maalouli, MD, FAAP,
medical director, hospitalist department,
Children’s Hospitals & Clinics of Minnesota, Minneapolis
References
- Arya R, Salovich DM, Ohman-Strickland P, Merlin MA. Impact of scribes on performance indicators in the emergency department. Acad Emerg Med. 2010;17(5):490- 494.
- Koshy S, Feustel PJ, Hong M, Kogan BA. Scribes in an ambulatory urology practice: patient and physician satisfaction. J Urol. 2010;184(1):258-262.
- Conn J. Getting it in writing. Docs using scribes to ease the transition to EHRs. Mod Healthc. 2010;40(6):30,32.
- Expanded scribe role boosts staff morale. ED Manag. 2009;21(7):75-77.
Over the past couple of decades, the ever-increasing medical-legal, financial, and patient-safety demands have led to a mushrooming of the amount of documentation needed during patient care, especially in the inpatient setting. With the advent of HM, that burden simply shifted to hospitalists, along with rapidly expanding patient loads as PCPs started delegating inpatient care. The problem was further compounded in teaching hospitals by a gradual decline in resident support as patient caps and work-hour limits were instituted.
Not surprisingly, the quality and accuracy of physicians’ progress notes started to suffer. It became frequently difficult to remember the details of encounters from earlier in the day, and fatigue sometimes led to details essential to continuity of care being left out completely.
A potential—and very intriguing—solution presented itself, however, as we were designing family-centered team rounds. Apparently, the ED group at the adjacent Abbott-Northwestern Hospital in Minneapolis had been using scribes for several years. The scribes captured the providers’ notes during each encounter, and the group had developed a polished system of scribe recruitment and training (mostly pre-med students awaiting acceptance to medical schools).
But would this model work in the inpatient setting where workflow was completely different? This was a total unknown, given that there has been no precedent in the literature about inpatient scribe utilization. Although relatively meager in number, the published articles about outpatient use of scribes were generally favorable, with the authors noting improved productivity and increased patient and staff satisfaction.1,2,3,4
Operationally, the main difference between inpatient and outpatient scribe use lied in the rounding mechanism itself; it was not just a provider-patient interaction, as in the ED, but rather a sometimes cacophonous collection of multiple inputs from families, providers, students, residents, nurses, and ancillary staff. The challenge here was to train the scribes (natural ability played a significant role) to produce a coherent document out of this interaction; the document had to accurately reflect current status and plans of care. The first step was to create a template for the daily progress notes with all the necessary headings, as well as age-appropriate normal exams, with the ability to directly import from the EMR such dynamic data as vital signs, medication lists, and lab results.
Each morning prior to rounds, the scribe was provided a list of patients to be rounded on, and the scribe spent about 30 minutes creating partial notes on each patient, as well as a brief patient identification statement. This proved to be a significant time-saver during rounds and allowed the scribes to keep up with the care team. Each note was pre-pended with the following statements, as recommended by the hospital’s risk-management team:
- I, (scribe name), am serving as a scribe to document services personally performed by (hospitalist name), MD, based on my observation and the provider’s statements to me.
- I, (hospitalist name), MD, attest that (scribe name) is acting in a scribe capacity, has observed my performance of the services, and has documented them in accordance with my direction.
A computer-on-wheels (COW) station was assigned to the scribes and accompanied the rounding team into the patient rooms, making it possible for them to follow the interactions in real time. The scribes were encouraged to hold their hands up and interrupt the conversation at any time if they needed clarification, something that happened rather infrequently. On the other hand, the care team was directed to speak clearly and the residents were instructed to present their cases using the same format as the progress-note template, thus minimizing guesswork on the part of the scribes. The only part of the interaction the scribes could not transcribe directly was the physical examination, and typically the attending physician dictated abnormal findings as well as systems not assessed, if any, at the end. All the scribe-generated notes were forwarded to the rounding provider’s EMR inbox and reviewed by the latter, amended when necessary, and signed electronically. The review of the scribe-generated notes proved to be initially a time-consuming process, typically two hours. However, as the scribes gained experience, the time commitment on the part of the providers decreased rapidly; after about three months, the time spent was about 30 minutes on busy days, even less otherwise, with a concomitant increase in satisfaction with the scribing process.
Not too unexpectedly, our transcription service initially pushed back on this idea because of their fear of job loss. However, this issue was resolved by eliminating the need for outsourcing overflow dictations. In addition, the relative terseness of the scribed notes did not sit well initially with some of our colleagues used to excessive verbosity, and this issue again gradually faded away as physicians realized that all the necessary information for care was preserved.
Does a scribe program make sense in all situations? Not likely. This kind of setup will work best in busy tertiary-care centers with residency programs and family-centered team rounds. Furthermore, a nearby college or university is a must if cost-efficient hiring of pre-med students is to be possible.
Walid Maalouli, MD, FAAP,
medical director, hospitalist department,
Children’s Hospitals & Clinics of Minnesota, Minneapolis
References
- Arya R, Salovich DM, Ohman-Strickland P, Merlin MA. Impact of scribes on performance indicators in the emergency department. Acad Emerg Med. 2010;17(5):490- 494.
- Koshy S, Feustel PJ, Hong M, Kogan BA. Scribes in an ambulatory urology practice: patient and physician satisfaction. J Urol. 2010;184(1):258-262.
- Conn J. Getting it in writing. Docs using scribes to ease the transition to EHRs. Mod Healthc. 2010;40(6):30,32.
- Expanded scribe role boosts staff morale. ED Manag. 2009;21(7):75-77.
Over the past couple of decades, the ever-increasing medical-legal, financial, and patient-safety demands have led to a mushrooming of the amount of documentation needed during patient care, especially in the inpatient setting. With the advent of HM, that burden simply shifted to hospitalists, along with rapidly expanding patient loads as PCPs started delegating inpatient care. The problem was further compounded in teaching hospitals by a gradual decline in resident support as patient caps and work-hour limits were instituted.
Not surprisingly, the quality and accuracy of physicians’ progress notes started to suffer. It became frequently difficult to remember the details of encounters from earlier in the day, and fatigue sometimes led to details essential to continuity of care being left out completely.
A potential—and very intriguing—solution presented itself, however, as we were designing family-centered team rounds. Apparently, the ED group at the adjacent Abbott-Northwestern Hospital in Minneapolis had been using scribes for several years. The scribes captured the providers’ notes during each encounter, and the group had developed a polished system of scribe recruitment and training (mostly pre-med students awaiting acceptance to medical schools).
But would this model work in the inpatient setting where workflow was completely different? This was a total unknown, given that there has been no precedent in the literature about inpatient scribe utilization. Although relatively meager in number, the published articles about outpatient use of scribes were generally favorable, with the authors noting improved productivity and increased patient and staff satisfaction.1,2,3,4
Operationally, the main difference between inpatient and outpatient scribe use lied in the rounding mechanism itself; it was not just a provider-patient interaction, as in the ED, but rather a sometimes cacophonous collection of multiple inputs from families, providers, students, residents, nurses, and ancillary staff. The challenge here was to train the scribes (natural ability played a significant role) to produce a coherent document out of this interaction; the document had to accurately reflect current status and plans of care. The first step was to create a template for the daily progress notes with all the necessary headings, as well as age-appropriate normal exams, with the ability to directly import from the EMR such dynamic data as vital signs, medication lists, and lab results.
Each morning prior to rounds, the scribe was provided a list of patients to be rounded on, and the scribe spent about 30 minutes creating partial notes on each patient, as well as a brief patient identification statement. This proved to be a significant time-saver during rounds and allowed the scribes to keep up with the care team. Each note was pre-pended with the following statements, as recommended by the hospital’s risk-management team:
- I, (scribe name), am serving as a scribe to document services personally performed by (hospitalist name), MD, based on my observation and the provider’s statements to me.
- I, (hospitalist name), MD, attest that (scribe name) is acting in a scribe capacity, has observed my performance of the services, and has documented them in accordance with my direction.
A computer-on-wheels (COW) station was assigned to the scribes and accompanied the rounding team into the patient rooms, making it possible for them to follow the interactions in real time. The scribes were encouraged to hold their hands up and interrupt the conversation at any time if they needed clarification, something that happened rather infrequently. On the other hand, the care team was directed to speak clearly and the residents were instructed to present their cases using the same format as the progress-note template, thus minimizing guesswork on the part of the scribes. The only part of the interaction the scribes could not transcribe directly was the physical examination, and typically the attending physician dictated abnormal findings as well as systems not assessed, if any, at the end. All the scribe-generated notes were forwarded to the rounding provider’s EMR inbox and reviewed by the latter, amended when necessary, and signed electronically. The review of the scribe-generated notes proved to be initially a time-consuming process, typically two hours. However, as the scribes gained experience, the time commitment on the part of the providers decreased rapidly; after about three months, the time spent was about 30 minutes on busy days, even less otherwise, with a concomitant increase in satisfaction with the scribing process.
Not too unexpectedly, our transcription service initially pushed back on this idea because of their fear of job loss. However, this issue was resolved by eliminating the need for outsourcing overflow dictations. In addition, the relative terseness of the scribed notes did not sit well initially with some of our colleagues used to excessive verbosity, and this issue again gradually faded away as physicians realized that all the necessary information for care was preserved.
Does a scribe program make sense in all situations? Not likely. This kind of setup will work best in busy tertiary-care centers with residency programs and family-centered team rounds. Furthermore, a nearby college or university is a must if cost-efficient hiring of pre-med students is to be possible.
Walid Maalouli, MD, FAAP,
medical director, hospitalist department,
Children’s Hospitals & Clinics of Minnesota, Minneapolis
References
- Arya R, Salovich DM, Ohman-Strickland P, Merlin MA. Impact of scribes on performance indicators in the emergency department. Acad Emerg Med. 2010;17(5):490- 494.
- Koshy S, Feustel PJ, Hong M, Kogan BA. Scribes in an ambulatory urology practice: patient and physician satisfaction. J Urol. 2010;184(1):258-262.
- Conn J. Getting it in writing. Docs using scribes to ease the transition to EHRs. Mod Healthc. 2010;40(6):30,32.
- Expanded scribe role boosts staff morale. ED Manag. 2009;21(7):75-77.