User login
Ukraine and PTSD: How psychiatry can help
The war in Ukraine is resulting in a devastating loss of life, catastrophic injuries, and physical destruction. But the war also will take an enormous mental health toll on millions of people, resulting in what I think will lead to an epidemic of posttraumatic stress disorder.
Think about the horrors that Ukrainians are experiencing. Millions of Ukrainians have been displaced to locations inside and outside of the country. People are being forced to leave behind family members, neighbors, and their pets and homes. In one recent news report, a Ukrainian woman who left Kyiv for Belgium reported having dreams in which she heard explosions. Smells, sounds, and even colors can trigger intrusive memories and a host of other problems. The mind can barely comprehend the scope of this human crisis.
Ukrainian soldiers are witnessing horrors that are unspeakable. Doctors, emergency service workers, and other medical professionals in Ukraine are being exposed to the catastrophe on a large scale. Children and youth are among the most affected victims, and it is difficult to predict the impact all of this upheaval is having on them.
The most important question for those of us who treat mental illness is “how will we help devastated people suffering from extreme trauma tied to death, dying, severe injuries, and torture by the invading soldiers?”
I have been treating patients with PTSD for many years. In my lifetime, the devastation in Ukraine will translate into what I expect will be the first overwhelming mass epidemic of PTSD – at least that I can recall. Yes, surely PTSD occurred during and after the Holocaust in the World War II era, but at that time, the mental health profession was not equipped to recognize it – even though the disorder most certainly existed. Even in ancient times, an Assyrian text from Mesopotamia (currently Iraq) described what we would define as PTSD symptoms in soldiers, such as sleep disturbances, flashbacks, and “low mood,” according to a 2014 article in the journal Early Science and Medicine.
The DSM-5 describes numerous criteria for PTSD mainly centering on trauma exposing a person to actual or threatened death, serious injury, or a variety of assaults, including direct exposure or witnessing the event. However, in my clinical experience, I’ve seen lesser events leading to PTSD. Much depends on how each individual processes what is occurring or has occurred.
What appears to be clear is that some key aspects of PTSD according to the DSM-5 – such as trauma-related thoughts or feelings, or trauma-related reminders, as well as nightmares and flashbacks – are likely occurring among Ukrainians. In addition, hypervigilance and exaggerated startle response seem to be key components of PTSD whether or not the cause is a major event or what one would perceive as less traumatic or dramatic.
I’ve certainly seen PTSD secondary to a hospitalization, especially in care involving ICUs or cardiac care units. In addition, I’ve had the occasion to note PTSD signs and symptoms after financial loss or divorce, situations in which some clinicians would never believe PTSD would occur, and would often diagnose as anxiety or depression. For me, again from a clinical point of view, it’s always been critical to assess how individuals process the event or events around them.
We know that there is already a shortage of mental health clinicians across the globe. This means that, in light of the hundreds of thousands – possibly millions – of Ukrainians affected by PTSD, a one-to-one approach will not do. For those Ukrainians who are able to find safe havens, I believe that PTSD symptoms can be debilitating, and the mental health community needs to begin putting supports in place now to address this trauma.
Specifically, proven cognitive-behavioral therapy (CBT) and guided imagery should be used to begin helping some of these people recover from the unbelievable trauma of war. For some, medication management might be helpful in those experiencing nightmares combined with anxiety and depression. But the main approach and first line of care should be CBT and guided imagery.
PTSD symptoms can make people feel like they are losing control, and prevent them from rebuilding their lives. We must do all we can in the mental health community to destigmatize care and develop support services to get ahead of this crisis. Only through medical, psychiatric, and health care organizations banding together using modern technology can the large number of people psychologically affected by this ongoing crisis be helped and saved.
Dr. London is a practicing psychiatrist who has been a newspaper columnist for 35 years, specializing in writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019). He has no conflicts of interest.
The war in Ukraine is resulting in a devastating loss of life, catastrophic injuries, and physical destruction. But the war also will take an enormous mental health toll on millions of people, resulting in what I think will lead to an epidemic of posttraumatic stress disorder.
Think about the horrors that Ukrainians are experiencing. Millions of Ukrainians have been displaced to locations inside and outside of the country. People are being forced to leave behind family members, neighbors, and their pets and homes. In one recent news report, a Ukrainian woman who left Kyiv for Belgium reported having dreams in which she heard explosions. Smells, sounds, and even colors can trigger intrusive memories and a host of other problems. The mind can barely comprehend the scope of this human crisis.
Ukrainian soldiers are witnessing horrors that are unspeakable. Doctors, emergency service workers, and other medical professionals in Ukraine are being exposed to the catastrophe on a large scale. Children and youth are among the most affected victims, and it is difficult to predict the impact all of this upheaval is having on them.
The most important question for those of us who treat mental illness is “how will we help devastated people suffering from extreme trauma tied to death, dying, severe injuries, and torture by the invading soldiers?”
I have been treating patients with PTSD for many years. In my lifetime, the devastation in Ukraine will translate into what I expect will be the first overwhelming mass epidemic of PTSD – at least that I can recall. Yes, surely PTSD occurred during and after the Holocaust in the World War II era, but at that time, the mental health profession was not equipped to recognize it – even though the disorder most certainly existed. Even in ancient times, an Assyrian text from Mesopotamia (currently Iraq) described what we would define as PTSD symptoms in soldiers, such as sleep disturbances, flashbacks, and “low mood,” according to a 2014 article in the journal Early Science and Medicine.
The DSM-5 describes numerous criteria for PTSD mainly centering on trauma exposing a person to actual or threatened death, serious injury, or a variety of assaults, including direct exposure or witnessing the event. However, in my clinical experience, I’ve seen lesser events leading to PTSD. Much depends on how each individual processes what is occurring or has occurred.
What appears to be clear is that some key aspects of PTSD according to the DSM-5 – such as trauma-related thoughts or feelings, or trauma-related reminders, as well as nightmares and flashbacks – are likely occurring among Ukrainians. In addition, hypervigilance and exaggerated startle response seem to be key components of PTSD whether or not the cause is a major event or what one would perceive as less traumatic or dramatic.
I’ve certainly seen PTSD secondary to a hospitalization, especially in care involving ICUs or cardiac care units. In addition, I’ve had the occasion to note PTSD signs and symptoms after financial loss or divorce, situations in which some clinicians would never believe PTSD would occur, and would often diagnose as anxiety or depression. For me, again from a clinical point of view, it’s always been critical to assess how individuals process the event or events around them.
We know that there is already a shortage of mental health clinicians across the globe. This means that, in light of the hundreds of thousands – possibly millions – of Ukrainians affected by PTSD, a one-to-one approach will not do. For those Ukrainians who are able to find safe havens, I believe that PTSD symptoms can be debilitating, and the mental health community needs to begin putting supports in place now to address this trauma.
Specifically, proven cognitive-behavioral therapy (CBT) and guided imagery should be used to begin helping some of these people recover from the unbelievable trauma of war. For some, medication management might be helpful in those experiencing nightmares combined with anxiety and depression. But the main approach and first line of care should be CBT and guided imagery.
PTSD symptoms can make people feel like they are losing control, and prevent them from rebuilding their lives. We must do all we can in the mental health community to destigmatize care and develop support services to get ahead of this crisis. Only through medical, psychiatric, and health care organizations banding together using modern technology can the large number of people psychologically affected by this ongoing crisis be helped and saved.
Dr. London is a practicing psychiatrist who has been a newspaper columnist for 35 years, specializing in writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019). He has no conflicts of interest.
The war in Ukraine is resulting in a devastating loss of life, catastrophic injuries, and physical destruction. But the war also will take an enormous mental health toll on millions of people, resulting in what I think will lead to an epidemic of posttraumatic stress disorder.
Think about the horrors that Ukrainians are experiencing. Millions of Ukrainians have been displaced to locations inside and outside of the country. People are being forced to leave behind family members, neighbors, and their pets and homes. In one recent news report, a Ukrainian woman who left Kyiv for Belgium reported having dreams in which she heard explosions. Smells, sounds, and even colors can trigger intrusive memories and a host of other problems. The mind can barely comprehend the scope of this human crisis.
Ukrainian soldiers are witnessing horrors that are unspeakable. Doctors, emergency service workers, and other medical professionals in Ukraine are being exposed to the catastrophe on a large scale. Children and youth are among the most affected victims, and it is difficult to predict the impact all of this upheaval is having on them.
The most important question for those of us who treat mental illness is “how will we help devastated people suffering from extreme trauma tied to death, dying, severe injuries, and torture by the invading soldiers?”
I have been treating patients with PTSD for many years. In my lifetime, the devastation in Ukraine will translate into what I expect will be the first overwhelming mass epidemic of PTSD – at least that I can recall. Yes, surely PTSD occurred during and after the Holocaust in the World War II era, but at that time, the mental health profession was not equipped to recognize it – even though the disorder most certainly existed. Even in ancient times, an Assyrian text from Mesopotamia (currently Iraq) described what we would define as PTSD symptoms in soldiers, such as sleep disturbances, flashbacks, and “low mood,” according to a 2014 article in the journal Early Science and Medicine.
The DSM-5 describes numerous criteria for PTSD mainly centering on trauma exposing a person to actual or threatened death, serious injury, or a variety of assaults, including direct exposure or witnessing the event. However, in my clinical experience, I’ve seen lesser events leading to PTSD. Much depends on how each individual processes what is occurring or has occurred.
What appears to be clear is that some key aspects of PTSD according to the DSM-5 – such as trauma-related thoughts or feelings, or trauma-related reminders, as well as nightmares and flashbacks – are likely occurring among Ukrainians. In addition, hypervigilance and exaggerated startle response seem to be key components of PTSD whether or not the cause is a major event or what one would perceive as less traumatic or dramatic.
I’ve certainly seen PTSD secondary to a hospitalization, especially in care involving ICUs or cardiac care units. In addition, I’ve had the occasion to note PTSD signs and symptoms after financial loss or divorce, situations in which some clinicians would never believe PTSD would occur, and would often diagnose as anxiety or depression. For me, again from a clinical point of view, it’s always been critical to assess how individuals process the event or events around them.
We know that there is already a shortage of mental health clinicians across the globe. This means that, in light of the hundreds of thousands – possibly millions – of Ukrainians affected by PTSD, a one-to-one approach will not do. For those Ukrainians who are able to find safe havens, I believe that PTSD symptoms can be debilitating, and the mental health community needs to begin putting supports in place now to address this trauma.
Specifically, proven cognitive-behavioral therapy (CBT) and guided imagery should be used to begin helping some of these people recover from the unbelievable trauma of war. For some, medication management might be helpful in those experiencing nightmares combined with anxiety and depression. But the main approach and first line of care should be CBT and guided imagery.
PTSD symptoms can make people feel like they are losing control, and prevent them from rebuilding their lives. We must do all we can in the mental health community to destigmatize care and develop support services to get ahead of this crisis. Only through medical, psychiatric, and health care organizations banding together using modern technology can the large number of people psychologically affected by this ongoing crisis be helped and saved.
Dr. London is a practicing psychiatrist who has been a newspaper columnist for 35 years, specializing in writing about short-term therapy, including cognitive-behavioral therapy and guided imagery. He is author of “Find Freedom Fast” (New York: Kettlehole Publishing, 2019). He has no conflicts of interest.
Unraveling primary ovarian insufficiency
In the presentation of secondary amenorrhea, pregnancy is the No. 1 differential diagnosis. Once this has been excluded, an algorithm is initiated to determine the etiology, including an assessment of the hypothalamic-pituitary-ovarian axis. While the early onset of ovarian failure can be physically and psychologically disrupting, the effect on fertility is an especially devastating event. Previously identified by terms including premature ovarian failure and premature menopause, “primary ovarian insufficiency” (POI) is now the preferred designation. This month’s article will address the diagnosis, evaluation, and management of POI.
The definition of POI is the development of primary hypogonadism before the age of 40 years. Spontaneous POI occurs in approximately 1 in 250 women by age 35 years and 1 in 100 by age 40 years. After excluding pregnancy, the clinician should determine signs and symptoms that can lead to expedited and cost-efficient testing.
Consequences
POI is an important risk factor for bone loss and osteoporosis, especially in young women who develop ovarian dysfunction before they achieve peak adult bone mass. At the time of diagnosis of POI, a bone density test (dual-energy x-ray absorptiometry) should be obtained. Women with POI may also develop depression and anxiety as well as experience an increased risk for cardiovascular morbidity and mortality, possibly related to endothelial dysfunction.
Young women with spontaneous POI are at increased risk of developing autoimmune adrenal insufficiency (AAI), a potentially fatal disorder. Consequently, to diagnose AAI, serum adrenal cortical and 21-hydroxylase antibodies should be measured in all women who have a karyotype of 46,XX and experience spontaneous POI. Women with AAI have a 50% risk of developing adrenal insufficiency. Despite initial normal adrenal function, women with positive adrenal cortical antibodies should be followed annually.
Causes (see table for a more complete list)
Iatrogenic
Known causes of POI include chemotherapy/radiation often in the setting of cancer treatment. The three most commonly used drugs, cyclophosphamide, cisplatin, and doxorubicin, cause POI by inducing death and/or accelerated activation of primordial follicles and increased atresia of growing follicles. The most damaging agents are alkylating drugs. A cyclophosphamide equivalent dose calculator has been established for ovarian failure risk stratification from chemotherapy based on the cumulative dose of alkylating agents received.
One study estimated the radiosensitivity of the oocyte to be less than 2 Gy. Based upon this estimate, the authors calculated the dose of radiotherapy that would result in immediate and permanent ovarian failure in 97.5% of patients as follows:
- 20.3 Gy at birth
- 18.4 Gy at age 10 years
- 16.5 Gy at age 20 years
- 14.3 Gy at age 30 years
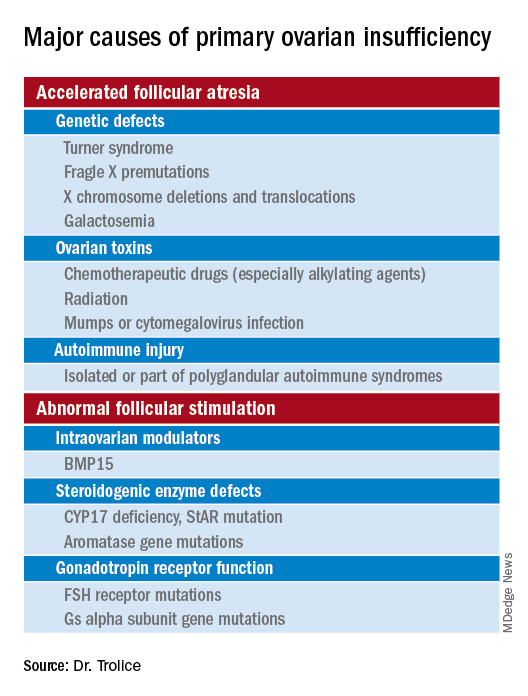
Genetic
Approximately 10% of cases are familial. A family history of POI raises concern for a fragile X premutation. Fragile X syndrome is an X-linked form of intellectual disability that is one of the most common causes of mental retardation worldwide. There is a strong relationship between age at menopause, including POI, and premutations for fragile X syndrome. The American College of Obstetricians and Gynecologists recommends that women with POI or an elevated follicle-stimulating hormone (FSH) level before age 40 years without known cause be screened for FMR1 premutations. Approximately 6% of cases of POI are associated with premutations in the FMR1 gene.
Turner syndrome is one of the most common causes of POI and results from the lack of a second X chromosome. The most common chromosomal defect in humans, TS occurs in up to 1.5% of conceptions, 10% of spontaneous abortions, and 1 of 2,500 live births.
Serum antiadrenal and/or anti–21-hydroxylase antibodies and antithyroid antiperoxidase antibodies, can aid in the diagnosis of adrenal gland, ovary, and thyroid autoimmune causes, which is found in 4% of women with spontaneous POI. Testing for the presence of 21-hydroxylase autoantibodies or adrenal autoantibodies is sufficient to make the diagnosis of autoimmune oophoritis in women with proven spontaneous POI.
The etiology of POI remains unknown in approximately 75%-90% of cases. However, studies using whole exome or whole genome sequencing have identified genetic variants in approximately 30%-35% of these patients.
Risk factors
Factors that are thought to play a role in determining the age of menopause, include genetics (e.g., FMR1 premutation and mosaic Turner syndrome), ethnicity (earlier among Hispanic women and later in Japanese American women when compared with White women), and smoking (reduced by approximately 2 years ).
Regarding ovarian aging, the holy grail of the reproductive life span is to predict menopause. While the definitive age eludes us, anti-Müllerian hormone levels appear to show promise. An ultrasensitive anti-Müllerian hormone assay (< 0.01 ng/mL) predicted a 79% probability of menopause within 12 months for women aged 51 and above; the probability was 51% for women below age 48.
Diagnosis
The three P’s of secondary amenorrhea are physiological, pharmacological, or pathological and can guide the clinician to a targeted evaluation. Physiological causes are pregnancy, the first 6 months of continuous breastfeeding (from elevated prolactin), and natural menopause. Pharmacological etiologies, excluding hormonal treatment that suppresses ovulation (combined oral contraceptives, gonadotropin-releasing hormone agonist/antagonist, or danazol), include agents that inhibit dopamine thereby increasing serum prolactin, such as metoclopramide; phenothiazine antipsychotics, such as haloperidol; and tardive dystonia dopamine-depleting medications, such as reserpine. Pathological causes include pituitary adenomas, thyroid disease, functional hypothalamic amenorrhea from changes in weight, exercise regimen, and stress.
Management
About 50%-75% of women with 46,XX spontaneous POI experience intermittent ovarian function and 5%-10% of women remain able to conceive. Anecdotally, a 32-year-old woman presented to me with primary infertility, secondary amenorrhea, and suspected POI based on vasomotor symptoms and elevated FSH levels. Pelvic ultrasound showed a hemorrhagic cyst, suspicious for a corpus luteum. Two weeks thereafter she reported a positive home urine human chorionic gonadotropin test and ultimately delivered twins. Her diagnosis of POI with amenorrhea remained postpartum.
Unless there is an absolute contraindication, estrogen therapy should be prescribed to women with POI to reduce the risk of osteoporosis, cardiovascular disease, and urogenital atrophy as well as to maintain sexual health and quality of life. For those with an intact uterus, women should receive progesterone because of the risk of endometrial hyperplasia from unopposed estrogen. Rather than oral estrogen, the use of transdermal or vaginal delivery of estrogen is a more physiological approach and provides lower risks of venous thromboembolism and gallbladder disease. Of note, standard postmenopausal hormone therapy, which has a much lower dose of estrogen than combined estrogen-progestin contraceptives, does not provide effective contraception. Per ACOG, systemic hormone treatment should be prescribed until age 50-51 years to all women with POI.
For fertility, women with spontaneous POI can be offered oocyte or embryo donation. The uterus does not age reproductively, unlike oocytes, therefore women can achieve reasonable pregnancy success rates through egg donation despite experiencing menopause.
Future potential options
Female germline stem cells have been isolated from neonatal mice and transplanted into sterile adult mice, who then were able to produce offspring. In a second study, oogonial stem cells were isolated from neonatal and adult mouse ovaries; pups were subsequently born from the oocytes. Further experiments are needed before the implications for humans can be determined.
Emotionally traumatic for most women, POI disrupts life plans, hopes, and dreams of raising a family. The approach to the patient with POI involves the above evidence-based testing along with empathy from the health care provider.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
In the presentation of secondary amenorrhea, pregnancy is the No. 1 differential diagnosis. Once this has been excluded, an algorithm is initiated to determine the etiology, including an assessment of the hypothalamic-pituitary-ovarian axis. While the early onset of ovarian failure can be physically and psychologically disrupting, the effect on fertility is an especially devastating event. Previously identified by terms including premature ovarian failure and premature menopause, “primary ovarian insufficiency” (POI) is now the preferred designation. This month’s article will address the diagnosis, evaluation, and management of POI.
The definition of POI is the development of primary hypogonadism before the age of 40 years. Spontaneous POI occurs in approximately 1 in 250 women by age 35 years and 1 in 100 by age 40 years. After excluding pregnancy, the clinician should determine signs and symptoms that can lead to expedited and cost-efficient testing.
Consequences
POI is an important risk factor for bone loss and osteoporosis, especially in young women who develop ovarian dysfunction before they achieve peak adult bone mass. At the time of diagnosis of POI, a bone density test (dual-energy x-ray absorptiometry) should be obtained. Women with POI may also develop depression and anxiety as well as experience an increased risk for cardiovascular morbidity and mortality, possibly related to endothelial dysfunction.
Young women with spontaneous POI are at increased risk of developing autoimmune adrenal insufficiency (AAI), a potentially fatal disorder. Consequently, to diagnose AAI, serum adrenal cortical and 21-hydroxylase antibodies should be measured in all women who have a karyotype of 46,XX and experience spontaneous POI. Women with AAI have a 50% risk of developing adrenal insufficiency. Despite initial normal adrenal function, women with positive adrenal cortical antibodies should be followed annually.
Causes (see table for a more complete list)
Iatrogenic
Known causes of POI include chemotherapy/radiation often in the setting of cancer treatment. The three most commonly used drugs, cyclophosphamide, cisplatin, and doxorubicin, cause POI by inducing death and/or accelerated activation of primordial follicles and increased atresia of growing follicles. The most damaging agents are alkylating drugs. A cyclophosphamide equivalent dose calculator has been established for ovarian failure risk stratification from chemotherapy based on the cumulative dose of alkylating agents received.
One study estimated the radiosensitivity of the oocyte to be less than 2 Gy. Based upon this estimate, the authors calculated the dose of radiotherapy that would result in immediate and permanent ovarian failure in 97.5% of patients as follows:
- 20.3 Gy at birth
- 18.4 Gy at age 10 years
- 16.5 Gy at age 20 years
- 14.3 Gy at age 30 years

Genetic
Approximately 10% of cases are familial. A family history of POI raises concern for a fragile X premutation. Fragile X syndrome is an X-linked form of intellectual disability that is one of the most common causes of mental retardation worldwide. There is a strong relationship between age at menopause, including POI, and premutations for fragile X syndrome. The American College of Obstetricians and Gynecologists recommends that women with POI or an elevated follicle-stimulating hormone (FSH) level before age 40 years without known cause be screened for FMR1 premutations. Approximately 6% of cases of POI are associated with premutations in the FMR1 gene.
Turner syndrome is one of the most common causes of POI and results from the lack of a second X chromosome. The most common chromosomal defect in humans, TS occurs in up to 1.5% of conceptions, 10% of spontaneous abortions, and 1 of 2,500 live births.
Serum antiadrenal and/or anti–21-hydroxylase antibodies and antithyroid antiperoxidase antibodies, can aid in the diagnosis of adrenal gland, ovary, and thyroid autoimmune causes, which is found in 4% of women with spontaneous POI. Testing for the presence of 21-hydroxylase autoantibodies or adrenal autoantibodies is sufficient to make the diagnosis of autoimmune oophoritis in women with proven spontaneous POI.
The etiology of POI remains unknown in approximately 75%-90% of cases. However, studies using whole exome or whole genome sequencing have identified genetic variants in approximately 30%-35% of these patients.
Risk factors
Factors that are thought to play a role in determining the age of menopause, include genetics (e.g., FMR1 premutation and mosaic Turner syndrome), ethnicity (earlier among Hispanic women and later in Japanese American women when compared with White women), and smoking (reduced by approximately 2 years ).
Regarding ovarian aging, the holy grail of the reproductive life span is to predict menopause. While the definitive age eludes us, anti-Müllerian hormone levels appear to show promise. An ultrasensitive anti-Müllerian hormone assay (< 0.01 ng/mL) predicted a 79% probability of menopause within 12 months for women aged 51 and above; the probability was 51% for women below age 48.
Diagnosis
The three P’s of secondary amenorrhea are physiological, pharmacological, or pathological and can guide the clinician to a targeted evaluation. Physiological causes are pregnancy, the first 6 months of continuous breastfeeding (from elevated prolactin), and natural menopause. Pharmacological etiologies, excluding hormonal treatment that suppresses ovulation (combined oral contraceptives, gonadotropin-releasing hormone agonist/antagonist, or danazol), include agents that inhibit dopamine thereby increasing serum prolactin, such as metoclopramide; phenothiazine antipsychotics, such as haloperidol; and tardive dystonia dopamine-depleting medications, such as reserpine. Pathological causes include pituitary adenomas, thyroid disease, functional hypothalamic amenorrhea from changes in weight, exercise regimen, and stress.
Management
About 50%-75% of women with 46,XX spontaneous POI experience intermittent ovarian function and 5%-10% of women remain able to conceive. Anecdotally, a 32-year-old woman presented to me with primary infertility, secondary amenorrhea, and suspected POI based on vasomotor symptoms and elevated FSH levels. Pelvic ultrasound showed a hemorrhagic cyst, suspicious for a corpus luteum. Two weeks thereafter she reported a positive home urine human chorionic gonadotropin test and ultimately delivered twins. Her diagnosis of POI with amenorrhea remained postpartum.
Unless there is an absolute contraindication, estrogen therapy should be prescribed to women with POI to reduce the risk of osteoporosis, cardiovascular disease, and urogenital atrophy as well as to maintain sexual health and quality of life. For those with an intact uterus, women should receive progesterone because of the risk of endometrial hyperplasia from unopposed estrogen. Rather than oral estrogen, the use of transdermal or vaginal delivery of estrogen is a more physiological approach and provides lower risks of venous thromboembolism and gallbladder disease. Of note, standard postmenopausal hormone therapy, which has a much lower dose of estrogen than combined estrogen-progestin contraceptives, does not provide effective contraception. Per ACOG, systemic hormone treatment should be prescribed until age 50-51 years to all women with POI.
For fertility, women with spontaneous POI can be offered oocyte or embryo donation. The uterus does not age reproductively, unlike oocytes, therefore women can achieve reasonable pregnancy success rates through egg donation despite experiencing menopause.
Future potential options
Female germline stem cells have been isolated from neonatal mice and transplanted into sterile adult mice, who then were able to produce offspring. In a second study, oogonial stem cells were isolated from neonatal and adult mouse ovaries; pups were subsequently born from the oocytes. Further experiments are needed before the implications for humans can be determined.
Emotionally traumatic for most women, POI disrupts life plans, hopes, and dreams of raising a family. The approach to the patient with POI involves the above evidence-based testing along with empathy from the health care provider.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
In the presentation of secondary amenorrhea, pregnancy is the No. 1 differential diagnosis. Once this has been excluded, an algorithm is initiated to determine the etiology, including an assessment of the hypothalamic-pituitary-ovarian axis. While the early onset of ovarian failure can be physically and psychologically disrupting, the effect on fertility is an especially devastating event. Previously identified by terms including premature ovarian failure and premature menopause, “primary ovarian insufficiency” (POI) is now the preferred designation. This month’s article will address the diagnosis, evaluation, and management of POI.
The definition of POI is the development of primary hypogonadism before the age of 40 years. Spontaneous POI occurs in approximately 1 in 250 women by age 35 years and 1 in 100 by age 40 years. After excluding pregnancy, the clinician should determine signs and symptoms that can lead to expedited and cost-efficient testing.
Consequences
POI is an important risk factor for bone loss and osteoporosis, especially in young women who develop ovarian dysfunction before they achieve peak adult bone mass. At the time of diagnosis of POI, a bone density test (dual-energy x-ray absorptiometry) should be obtained. Women with POI may also develop depression and anxiety as well as experience an increased risk for cardiovascular morbidity and mortality, possibly related to endothelial dysfunction.
Young women with spontaneous POI are at increased risk of developing autoimmune adrenal insufficiency (AAI), a potentially fatal disorder. Consequently, to diagnose AAI, serum adrenal cortical and 21-hydroxylase antibodies should be measured in all women who have a karyotype of 46,XX and experience spontaneous POI. Women with AAI have a 50% risk of developing adrenal insufficiency. Despite initial normal adrenal function, women with positive adrenal cortical antibodies should be followed annually.
Causes (see table for a more complete list)
Iatrogenic
Known causes of POI include chemotherapy/radiation often in the setting of cancer treatment. The three most commonly used drugs, cyclophosphamide, cisplatin, and doxorubicin, cause POI by inducing death and/or accelerated activation of primordial follicles and increased atresia of growing follicles. The most damaging agents are alkylating drugs. A cyclophosphamide equivalent dose calculator has been established for ovarian failure risk stratification from chemotherapy based on the cumulative dose of alkylating agents received.
One study estimated the radiosensitivity of the oocyte to be less than 2 Gy. Based upon this estimate, the authors calculated the dose of radiotherapy that would result in immediate and permanent ovarian failure in 97.5% of patients as follows:
- 20.3 Gy at birth
- 18.4 Gy at age 10 years
- 16.5 Gy at age 20 years
- 14.3 Gy at age 30 years

Genetic
Approximately 10% of cases are familial. A family history of POI raises concern for a fragile X premutation. Fragile X syndrome is an X-linked form of intellectual disability that is one of the most common causes of mental retardation worldwide. There is a strong relationship between age at menopause, including POI, and premutations for fragile X syndrome. The American College of Obstetricians and Gynecologists recommends that women with POI or an elevated follicle-stimulating hormone (FSH) level before age 40 years without known cause be screened for FMR1 premutations. Approximately 6% of cases of POI are associated with premutations in the FMR1 gene.
Turner syndrome is one of the most common causes of POI and results from the lack of a second X chromosome. The most common chromosomal defect in humans, TS occurs in up to 1.5% of conceptions, 10% of spontaneous abortions, and 1 of 2,500 live births.
Serum antiadrenal and/or anti–21-hydroxylase antibodies and antithyroid antiperoxidase antibodies, can aid in the diagnosis of adrenal gland, ovary, and thyroid autoimmune causes, which is found in 4% of women with spontaneous POI. Testing for the presence of 21-hydroxylase autoantibodies or adrenal autoantibodies is sufficient to make the diagnosis of autoimmune oophoritis in women with proven spontaneous POI.
The etiology of POI remains unknown in approximately 75%-90% of cases. However, studies using whole exome or whole genome sequencing have identified genetic variants in approximately 30%-35% of these patients.
Risk factors
Factors that are thought to play a role in determining the age of menopause, include genetics (e.g., FMR1 premutation and mosaic Turner syndrome), ethnicity (earlier among Hispanic women and later in Japanese American women when compared with White women), and smoking (reduced by approximately 2 years ).
Regarding ovarian aging, the holy grail of the reproductive life span is to predict menopause. While the definitive age eludes us, anti-Müllerian hormone levels appear to show promise. An ultrasensitive anti-Müllerian hormone assay (< 0.01 ng/mL) predicted a 79% probability of menopause within 12 months for women aged 51 and above; the probability was 51% for women below age 48.
Diagnosis
The three P’s of secondary amenorrhea are physiological, pharmacological, or pathological and can guide the clinician to a targeted evaluation. Physiological causes are pregnancy, the first 6 months of continuous breastfeeding (from elevated prolactin), and natural menopause. Pharmacological etiologies, excluding hormonal treatment that suppresses ovulation (combined oral contraceptives, gonadotropin-releasing hormone agonist/antagonist, or danazol), include agents that inhibit dopamine thereby increasing serum prolactin, such as metoclopramide; phenothiazine antipsychotics, such as haloperidol; and tardive dystonia dopamine-depleting medications, such as reserpine. Pathological causes include pituitary adenomas, thyroid disease, functional hypothalamic amenorrhea from changes in weight, exercise regimen, and stress.
Management
About 50%-75% of women with 46,XX spontaneous POI experience intermittent ovarian function and 5%-10% of women remain able to conceive. Anecdotally, a 32-year-old woman presented to me with primary infertility, secondary amenorrhea, and suspected POI based on vasomotor symptoms and elevated FSH levels. Pelvic ultrasound showed a hemorrhagic cyst, suspicious for a corpus luteum. Two weeks thereafter she reported a positive home urine human chorionic gonadotropin test and ultimately delivered twins. Her diagnosis of POI with amenorrhea remained postpartum.
Unless there is an absolute contraindication, estrogen therapy should be prescribed to women with POI to reduce the risk of osteoporosis, cardiovascular disease, and urogenital atrophy as well as to maintain sexual health and quality of life. For those with an intact uterus, women should receive progesterone because of the risk of endometrial hyperplasia from unopposed estrogen. Rather than oral estrogen, the use of transdermal or vaginal delivery of estrogen is a more physiological approach and provides lower risks of venous thromboembolism and gallbladder disease. Of note, standard postmenopausal hormone therapy, which has a much lower dose of estrogen than combined estrogen-progestin contraceptives, does not provide effective contraception. Per ACOG, systemic hormone treatment should be prescribed until age 50-51 years to all women with POI.
For fertility, women with spontaneous POI can be offered oocyte or embryo donation. The uterus does not age reproductively, unlike oocytes, therefore women can achieve reasonable pregnancy success rates through egg donation despite experiencing menopause.
Future potential options
Female germline stem cells have been isolated from neonatal mice and transplanted into sterile adult mice, who then were able to produce offspring. In a second study, oogonial stem cells were isolated from neonatal and adult mouse ovaries; pups were subsequently born from the oocytes. Further experiments are needed before the implications for humans can be determined.
Emotionally traumatic for most women, POI disrupts life plans, hopes, and dreams of raising a family. The approach to the patient with POI involves the above evidence-based testing along with empathy from the health care provider.
Dr. Trolice is director of The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Depression in homeless patients: What can be done?
In a recent article published in JAMA Psychiatry, Joshua E. J. Buckman and coauthors described the results of a large research study which concludes that depression is harder to treat in those who are homeless or unemployed.
It is always good to get more data and this article adds to the literature about the social determinants of depression. A frustrating aspect is that this is no surprise at all, not least for anyone in the mental health field. We have known that intuitively for decades.
Again, data is always good to bolster intuition with science. But what are the actionable items to take from the paper?
However, there are a few policy and clinical points I would like to make, reflecting some of the chapters in a recently published book – edited by me and my colleague Maria D. Llorente – “Clinical Management of the Homeless Patient: Social Medical and Psychiatric Issues” (New York: Springer, May 2021).
The first is, if you really tackle homelessness, with a combination of federal, state, and local resources, you can make a difference. The Department of Veterans Affairs, under the leadership of former VA Secretary Eric Shinseki and others, has been markedly successful. Note, for instance, the Health Care for Homeless Veterans program , which conducts outreach to vulnerable veterans not currently receiving services and engages them in treatment and rehabilitative programs.
Secondly, there is a marked absence of shelters that can care for the homeless with medical problems. This leads to extended and extensive hospital stays. This is especially frustrating during the COVID era, when hospital beds are in such short supply. Having a safe place to discharge patients who still need wound or diabetes care would save money for the overall health care system and be best for the patient.
Third, it may be best to modify discharge regimens for those patients who are unhoused. For example, metformin, taken by mouth once a day, is more practical for unhoused patients with diabetes than insulin, which needs to be refrigerated and injected multiple times a day. While one can argue whether care for the homeless should differ from those who are housed, in practical terms, simplifying regimens is more likely to promote compliance.
My last take-home point is check the Feet. So many of our homeless patients who end up on hospital wards have been wearing ill-fitting or no shoes while they are out on the street. Their toenails may be long and thick. They may have cellulitis or ulcers. Or gangrene. Unfortunately, these medical issues can also cause surgical amputations of the lower extremities.
Back to the article by Buckman and colleagues. The data they provide is good to have. But we need more action to provide appropriate and compassionate care for those who are unhoused and ill – care that is good for them, good for the nation’s finances, and good for our moral standing in the world.
Dr. Ritchie is chair of psychiatry at Medstar Washington (D.C.) Hospital Center. She is a member of the Clinical Psychiatry News editorial advisory board, and has no conflicts of interest.
In a recent article published in JAMA Psychiatry, Joshua E. J. Buckman and coauthors described the results of a large research study which concludes that depression is harder to treat in those who are homeless or unemployed.
It is always good to get more data and this article adds to the literature about the social determinants of depression. A frustrating aspect is that this is no surprise at all, not least for anyone in the mental health field. We have known that intuitively for decades.
Again, data is always good to bolster intuition with science. But what are the actionable items to take from the paper?
However, there are a few policy and clinical points I would like to make, reflecting some of the chapters in a recently published book – edited by me and my colleague Maria D. Llorente – “Clinical Management of the Homeless Patient: Social Medical and Psychiatric Issues” (New York: Springer, May 2021).
The first is, if you really tackle homelessness, with a combination of federal, state, and local resources, you can make a difference. The Department of Veterans Affairs, under the leadership of former VA Secretary Eric Shinseki and others, has been markedly successful. Note, for instance, the Health Care for Homeless Veterans program , which conducts outreach to vulnerable veterans not currently receiving services and engages them in treatment and rehabilitative programs.
Secondly, there is a marked absence of shelters that can care for the homeless with medical problems. This leads to extended and extensive hospital stays. This is especially frustrating during the COVID era, when hospital beds are in such short supply. Having a safe place to discharge patients who still need wound or diabetes care would save money for the overall health care system and be best for the patient.
Third, it may be best to modify discharge regimens for those patients who are unhoused. For example, metformin, taken by mouth once a day, is more practical for unhoused patients with diabetes than insulin, which needs to be refrigerated and injected multiple times a day. While one can argue whether care for the homeless should differ from those who are housed, in practical terms, simplifying regimens is more likely to promote compliance.
My last take-home point is check the Feet. So many of our homeless patients who end up on hospital wards have been wearing ill-fitting or no shoes while they are out on the street. Their toenails may be long and thick. They may have cellulitis or ulcers. Or gangrene. Unfortunately, these medical issues can also cause surgical amputations of the lower extremities.
Back to the article by Buckman and colleagues. The data they provide is good to have. But we need more action to provide appropriate and compassionate care for those who are unhoused and ill – care that is good for them, good for the nation’s finances, and good for our moral standing in the world.
Dr. Ritchie is chair of psychiatry at Medstar Washington (D.C.) Hospital Center. She is a member of the Clinical Psychiatry News editorial advisory board, and has no conflicts of interest.
In a recent article published in JAMA Psychiatry, Joshua E. J. Buckman and coauthors described the results of a large research study which concludes that depression is harder to treat in those who are homeless or unemployed.
It is always good to get more data and this article adds to the literature about the social determinants of depression. A frustrating aspect is that this is no surprise at all, not least for anyone in the mental health field. We have known that intuitively for decades.
Again, data is always good to bolster intuition with science. But what are the actionable items to take from the paper?
However, there are a few policy and clinical points I would like to make, reflecting some of the chapters in a recently published book – edited by me and my colleague Maria D. Llorente – “Clinical Management of the Homeless Patient: Social Medical and Psychiatric Issues” (New York: Springer, May 2021).
The first is, if you really tackle homelessness, with a combination of federal, state, and local resources, you can make a difference. The Department of Veterans Affairs, under the leadership of former VA Secretary Eric Shinseki and others, has been markedly successful. Note, for instance, the Health Care for Homeless Veterans program , which conducts outreach to vulnerable veterans not currently receiving services and engages them in treatment and rehabilitative programs.
Secondly, there is a marked absence of shelters that can care for the homeless with medical problems. This leads to extended and extensive hospital stays. This is especially frustrating during the COVID era, when hospital beds are in such short supply. Having a safe place to discharge patients who still need wound or diabetes care would save money for the overall health care system and be best for the patient.
Third, it may be best to modify discharge regimens for those patients who are unhoused. For example, metformin, taken by mouth once a day, is more practical for unhoused patients with diabetes than insulin, which needs to be refrigerated and injected multiple times a day. While one can argue whether care for the homeless should differ from those who are housed, in practical terms, simplifying regimens is more likely to promote compliance.
My last take-home point is check the Feet. So many of our homeless patients who end up on hospital wards have been wearing ill-fitting or no shoes while they are out on the street. Their toenails may be long and thick. They may have cellulitis or ulcers. Or gangrene. Unfortunately, these medical issues can also cause surgical amputations of the lower extremities.
Back to the article by Buckman and colleagues. The data they provide is good to have. But we need more action to provide appropriate and compassionate care for those who are unhoused and ill – care that is good for them, good for the nation’s finances, and good for our moral standing in the world.
Dr. Ritchie is chair of psychiatry at Medstar Washington (D.C.) Hospital Center. She is a member of the Clinical Psychiatry News editorial advisory board, and has no conflicts of interest.
Weigh but don’t tell
Reports of long waiting times at mental health clinics and anecdotal observations by health care providers suggest the pandemic has generated a dramatic increase in the incidence of eating disorders among the pediatric population. Of course this should come as no surprise to pediatricians.
Eating disorders come in many different forms and a triggering event is sometimes difficult to define. Often the adolescent or preadolescent is searching for some sense of stability in a life tossed on a stormy sea roiled by hormonal and physical change. Wresting control of their bodies during a period of uncertainty may result in a downward spiral into dangerously unhealthy weight loss. If nothing else, the pandemic has been a period of dramatic uncertainty unlike what most children and few adults in this country have ever experienced.
With the unprecedented increase in eating disorder cases, providers in several disciplines are searching for novel strategies to ease the burden on their patients and their practices. I recently learned of a pediatric practice in California that is considering blinding all patients aged 12 and older to the body mass measurements obtained at their health maintenance visits.
Blind weight checks for children with eating disorders, particularly those who seem to be nearing recovery, has been a common and often helpful practice. However, I am unaware of any practice that has made it a universal office policy. I’m unsure of the rationale behind this practice’s policy, but on several fronts, suppressing body mass measurements in the age group most vulnerable to eating disorders makes some sense.
Universal blind weight checks could minimize the risk of in-office shaming. However, careful training of support staff and thoughtful placement of the scales could serve the same purpose. This new policy acknowledges not only the ubiquity of the problem but also that many, maybe even most, children with eating disorders appear normal. And of course, there is the unfortunate fact that body mass is a poor screening test for eating disorders.
As I thought more about this novel approach I came to see its educational value for patients, parents, and even physicians. I can envision how a 13-year-old’s first health maintenance visit would go after the roll-out of the new policy. “Dr. Smith, aren’t you going to tell us how much I (or my daughter Jenny) weigh(s)?” This could, or more likely, should launch a discussion about weight and body image. It might continue with questions like, “How much do you think you weigh?” Or, “Do you think you are too heavy or too thin?”
Or, the conversation could include the provider’s observations that weight is just one measure of health and in fact not a very good one. Other ingredients in a healthy life style, such as sleep and physical activity, are not as easy to measure as weight but in many cases are more important.
As my mind struggled to restructure a health maintenance schedule that included blind weight checks, I wondered why we should wait until age 12. Of course, it is unreasonable to expect parents to stick with a pediatric practice that seems to ignore their infant’s weight. I’m sure that, like me, you have always discouraged new parents from having a baby scale at home because in the first few months too-frequent weighings can usually cause more angst than good.
It might make sense to remove a within-earshot discussion of a child’s weight from the health maintenance visit as soon as the child can absorb and digest the discussion; say, around age 3 years. In a perfect world, the provider should have already elicited a history that suggested a young child’s vulnerability to obesity before the scale and the growth chart told the unfortunate story. But, neither you nor I are perfect providers and so we will always need the scale to document our concerns. However, when and how we report that one vital sign to the patient and his or her parents is a topic ripe for discussion and improvement.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Reports of long waiting times at mental health clinics and anecdotal observations by health care providers suggest the pandemic has generated a dramatic increase in the incidence of eating disorders among the pediatric population. Of course this should come as no surprise to pediatricians.
Eating disorders come in many different forms and a triggering event is sometimes difficult to define. Often the adolescent or preadolescent is searching for some sense of stability in a life tossed on a stormy sea roiled by hormonal and physical change. Wresting control of their bodies during a period of uncertainty may result in a downward spiral into dangerously unhealthy weight loss. If nothing else, the pandemic has been a period of dramatic uncertainty unlike what most children and few adults in this country have ever experienced.
With the unprecedented increase in eating disorder cases, providers in several disciplines are searching for novel strategies to ease the burden on their patients and their practices. I recently learned of a pediatric practice in California that is considering blinding all patients aged 12 and older to the body mass measurements obtained at their health maintenance visits.
Blind weight checks for children with eating disorders, particularly those who seem to be nearing recovery, has been a common and often helpful practice. However, I am unaware of any practice that has made it a universal office policy. I’m unsure of the rationale behind this practice’s policy, but on several fronts, suppressing body mass measurements in the age group most vulnerable to eating disorders makes some sense.
Universal blind weight checks could minimize the risk of in-office shaming. However, careful training of support staff and thoughtful placement of the scales could serve the same purpose. This new policy acknowledges not only the ubiquity of the problem but also that many, maybe even most, children with eating disorders appear normal. And of course, there is the unfortunate fact that body mass is a poor screening test for eating disorders.
As I thought more about this novel approach I came to see its educational value for patients, parents, and even physicians. I can envision how a 13-year-old’s first health maintenance visit would go after the roll-out of the new policy. “Dr. Smith, aren’t you going to tell us how much I (or my daughter Jenny) weigh(s)?” This could, or more likely, should launch a discussion about weight and body image. It might continue with questions like, “How much do you think you weigh?” Or, “Do you think you are too heavy or too thin?”
Or, the conversation could include the provider’s observations that weight is just one measure of health and in fact not a very good one. Other ingredients in a healthy life style, such as sleep and physical activity, are not as easy to measure as weight but in many cases are more important.
As my mind struggled to restructure a health maintenance schedule that included blind weight checks, I wondered why we should wait until age 12. Of course, it is unreasonable to expect parents to stick with a pediatric practice that seems to ignore their infant’s weight. I’m sure that, like me, you have always discouraged new parents from having a baby scale at home because in the first few months too-frequent weighings can usually cause more angst than good.
It might make sense to remove a within-earshot discussion of a child’s weight from the health maintenance visit as soon as the child can absorb and digest the discussion; say, around age 3 years. In a perfect world, the provider should have already elicited a history that suggested a young child’s vulnerability to obesity before the scale and the growth chart told the unfortunate story. But, neither you nor I are perfect providers and so we will always need the scale to document our concerns. However, when and how we report that one vital sign to the patient and his or her parents is a topic ripe for discussion and improvement.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
Reports of long waiting times at mental health clinics and anecdotal observations by health care providers suggest the pandemic has generated a dramatic increase in the incidence of eating disorders among the pediatric population. Of course this should come as no surprise to pediatricians.
Eating disorders come in many different forms and a triggering event is sometimes difficult to define. Often the adolescent or preadolescent is searching for some sense of stability in a life tossed on a stormy sea roiled by hormonal and physical change. Wresting control of their bodies during a period of uncertainty may result in a downward spiral into dangerously unhealthy weight loss. If nothing else, the pandemic has been a period of dramatic uncertainty unlike what most children and few adults in this country have ever experienced.
With the unprecedented increase in eating disorder cases, providers in several disciplines are searching for novel strategies to ease the burden on their patients and their practices. I recently learned of a pediatric practice in California that is considering blinding all patients aged 12 and older to the body mass measurements obtained at their health maintenance visits.
Blind weight checks for children with eating disorders, particularly those who seem to be nearing recovery, has been a common and often helpful practice. However, I am unaware of any practice that has made it a universal office policy. I’m unsure of the rationale behind this practice’s policy, but on several fronts, suppressing body mass measurements in the age group most vulnerable to eating disorders makes some sense.
Universal blind weight checks could minimize the risk of in-office shaming. However, careful training of support staff and thoughtful placement of the scales could serve the same purpose. This new policy acknowledges not only the ubiquity of the problem but also that many, maybe even most, children with eating disorders appear normal. And of course, there is the unfortunate fact that body mass is a poor screening test for eating disorders.
As I thought more about this novel approach I came to see its educational value for patients, parents, and even physicians. I can envision how a 13-year-old’s first health maintenance visit would go after the roll-out of the new policy. “Dr. Smith, aren’t you going to tell us how much I (or my daughter Jenny) weigh(s)?” This could, or more likely, should launch a discussion about weight and body image. It might continue with questions like, “How much do you think you weigh?” Or, “Do you think you are too heavy or too thin?”
Or, the conversation could include the provider’s observations that weight is just one measure of health and in fact not a very good one. Other ingredients in a healthy life style, such as sleep and physical activity, are not as easy to measure as weight but in many cases are more important.
As my mind struggled to restructure a health maintenance schedule that included blind weight checks, I wondered why we should wait until age 12. Of course, it is unreasonable to expect parents to stick with a pediatric practice that seems to ignore their infant’s weight. I’m sure that, like me, you have always discouraged new parents from having a baby scale at home because in the first few months too-frequent weighings can usually cause more angst than good.
It might make sense to remove a within-earshot discussion of a child’s weight from the health maintenance visit as soon as the child can absorb and digest the discussion; say, around age 3 years. In a perfect world, the provider should have already elicited a history that suggested a young child’s vulnerability to obesity before the scale and the growth chart told the unfortunate story. But, neither you nor I are perfect providers and so we will always need the scale to document our concerns. However, when and how we report that one vital sign to the patient and his or her parents is a topic ripe for discussion and improvement.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
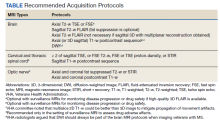
Harmonizing Magnetic Resonance Imaging Protocols for Veterans With Multiple Sclerosis
Multiple sclerosis (MS) is a lifelong disease that affects about a million people in the United States.1,2 Since 1998 more than 45,000 veterans have been diagnosed with MS and about 20,000 are evaluated in the Veterans Health Administration (VHA) annually.3
Magnetic resonance imaging (MRI) is a cornerstone for the assessment of persons with multiple sclerosis (pwMS).4-6 MRI assists with disease diagnosis, allowing for timely therapeutic interventions and withthe evaluation of its progression, treatment effect, and safety. 4,5 MRI-based outcomes also are used as primary endpoints in clinical trials.4,5
MS has its clinical onset in early adulthood in most individuals and is diagnosed at a mean age of 30 years.7 As a result, pwMS may receive care and MRIs in different facilities during their lifetime. Mitigating interscan variabilities that can challenge intra- and interperson comparisons is crucial for accurate care. Radiologists may find it difficult to compare scans acquired in different facilities, as dissimilarities in acquisition protocols may mask or uncover focal disease, creating false negative or false positive findings. Moreover, lack of a standardized method to report MRI changes may compromise neurologists’ ability to correctly interpret scans and disease progression.
Accordingly, in October 2019, an international task force of neurologists, radiologists, MRI technologists, and imaging scientists with expertise in MS, including representatives from the VHA, worked together to update guidelines for imaging the brain, spinal cord, and optic nerve in pwMS.8,9 Recognizing the importance of this effort, the VHA Multiple Sclerosis Centers of Excellence (MSCoE), in collaboration with a team of subject matter expert neuroradiologists promptly committed to this effort, advocating the updated consensus recommendations, and favoring their dissemination within the VHA.10
As part of this commitment and dissemination effort, in this report we summarize the core points of the newly proposed MRI guidelines and ways to adapt them for use within the VHA. We then discuss key elements for their successful implementation and dissemination, specifically regarding the clinical operations of VHA.
Updated Guidelines
MRI Scan at Different Timepoints of MS
There are 3 crucial milestones within a the lifespan of a pwMS that require an MRI to reach appropriate conclusions and avoid clinical errors. These include the initial diagnosis, the follow-up to monitor disease and/or treatment effect, and the assessment of medication safety.
In the interest of efficiency, MRI protocols may vary slightly depending on these clinical indications. The Table lists core sequences of the updated 2021 consensus recommendations at each timepoint along with the proposed alternatives or preferences from the VHA workgroup.
At the time of diagnosis, both brain and spine (cervical and thoracic) MRIs are recommended. Routine MRI of the optic nerve is considered optional at diagnosis. However, imaging the optic nerve may be useful in specific clinical scenarios when the optic nerve is selectively involved, and the diagnosis or etiology of an optic neuritis is not clear. A repeat brain MRI is advised every 6 to 12 months in patients with clinically or radiologically isolated syndrome who do not fulfill the diagnostic criteria of MS but present risk factors for conversion to MS or paraclinical features of it.
Once the diagnosis is established, brain MRI is recommended for follow-up and for surveillance of drug safety. Spinal cord and optic nerve MRIs are desirable but optional in the follow-up of pwMS and are not required for drug surveillance. Spinal cord MRIs are required at follow-up for patients whose progression cannot be explained by brain MRI features, or who manifest with recurrent spinal cord symptoms, or have spinal cord comorbidities. In these cases, spinal cord MRI also may assist with treatment decisions. Similarly, optic nerve MRI is necessary during follow-up only when optic nerve comorbidities are suspected or when there is progression or reoccurrence of optic nerve–related symptoms.
Brain MRIs are recommended for monitoring drug effect yearly (or at longer intervals, after a few years of disease stability). Conversely, a repeat brain MRI is advised after 6 months if nonsymptomatic radiological disease activity is discovered on surveillance scans.
Abbreviated but more frequent serial brain MRI protocols (eg, every 3 to 4 months) are recommended for pwMS treated with natalizumab and at high risk of developing progressive multifocal leukoencephalopathy (eg, pwMS who are John Cunningham virus [JCV]–positive, and have been treated with natalizumabfor ≥ 18 months, have a JCV antibody index > 0.9, or have a history of immunosuppression). A similar approach is recommended for carryover cases, such as those with high JCV antibody index who are switched to other immunosuppressive treatments.
MRI Field, Scan Resolution, and Coverage
Both 1.5-Tesla (1.5-T) and 3-T scans are believed to be equally effective in imaging pwMS, providing that the 1.5-T scans are good quality. Although imaging at < 1.5 T is not recommended due to suboptimal disease detection, the use of scanners > 3 T is equally discouraged outside the supervision of trained investigators. Signal-to-noise ratio and resolution are key factors impacting scan quality, and their optimization is prioritized over the number of sequences in the updated 2021 consensus recommendations. For brain imaging, a resolution of 1 mm3 isotropic is preferred for 3-dimensional (3D) imaging and slice thickness ≤ 3 mm without gap (≤ 5 mm with 10-30% gaps for diffusion-weighted imaging only) is recommended for 2D sequences. Images should cover the entire brain and as much of the cervical spine as possible; images should be prescribed axial for 2D or reformatted axial oblique for 3D using the subcallosal plane as reference. For spine imaging, sites should aim at an in-plane resolution of 1 mm2; using sagittal slices ≤ 3 mm thick and axial slices ≤ 5 mm thick, both with no gap. Scans should cover the entire cervical and thoracolumbar region inclusive of the conus. For the optic nerve images, slices should be ≤ 2 or 3 mm thick with an in-plane resolution of 1 mm2. Images should be aligned to the orientation of the optic nerve and chiasms, both of which should be entirely covered.
Postgadolinium Images Use
The discovery of the higher sensitivity of post-gadolinium (Gd) T1-weighted (T1-w) MRI relative to high iodine (88.1 g I) computed tomography scans in demonstrating contrast-enhancing MS lesions has revolutionized the way clinicians diagnose and monitor this disease.11 However, in recent years the role of postcontrast MRI has been debated, considering the potential safety concerns secondary to Gd tissue deposition. For this reason, an intentionally more judicious use of postcontrast MRI is proposed by the consensus recommendations. At disease diagnosis, the use of Gd is advisable to (1) show disease dissemination in time; (2) differentiate the diagnosis based on the Gd pattern; (3) predict short-term disease activity; and (4) characterize activity in the setting of progression. When monitoring pwMS, the use of Gd may be useful in the first year of follow-up, particularly if in the setting of low potency medications or for patients for whom the detection of one or more active lesions would lead to a change in disease-modifying agents. Gd also should be used to first, confirm a clinical exacerbation (if needed); second, further characterize a lesion suggestive of progressive multifocal encephalopathy or monitor this disease over time; and third, monitor lesion burden change in patients with large confluent lesions, the count of which otherwise may be difficult.
MRI During Pregnancy and Lactation
The consensus recommendations state that Gd contrast–enhanced MRI is not absolutely contraindicated during pregnancy, although its use should be limited to strictly necessary situations, particularly those involving differential diagnosis, such as cerebral venous thrombosis or monitoring of possibly enlarging lesion burden. The use of Gd is not contraindicated during lactation, as only a small proportion (< 0.4%) passes into the breast milk, leading to an exposure to < 1% of the permitted Gd dose for neonates.12,13
Harmonizing MRI Reports
The consensus recommendations propose reporting the exact lesion count on T2-weighted (T2-w) images when lesions are < 20, or specifying if the number of T2 lesions is between 20 and 50, between 50 and 100, or uncountable, eg, confluent large lesions. Similarly, for the spinal cord, the consensus recommendations propose reporting the exact lesion count on T2-w images when lesions are < 10, or otherwise report that > 10 lesions are seen.
The VHA workgroup proposed reporting a mild, moderate, or severe T2-lesion burden for a T2-lesion count < 20, between 20 and 50, and > 50, respectively. For follow-up MRIs, notation should be made if there is any change in lesion number, indicating the number of new lesions whenever possible. At each timepoint, the presence of active lesions on postcontrast images should be accurately defined.
Dissemination and Implementation
To implement and disseminate these proposed recommendations within the VHA, a workgroup of neurologists and radiologists was formed in late 2020. A review and discussion of the importance of each of the proposed MRI protocols for veterans with MS was held along with possible modifications to balance the intent of meeting standards of care with resources of individual US Department of Veterans Affairs (VA) medical centers and veterans’ needs. The final protocol recommendations were agreed on by group consensus.
In general, this VHA workgroup felt that the current adopted MRI protocols in several VA medical centers (based on previously proposed recommendations) were similar to the ones newly proposed and that implementing changes to meet the 2021 criteria would not be a major challenge.14,15 Possible regional and nonregional barriers were discussed. The result of these discussions led to a modified version of what could be considered more stringent guidelines to accommodate medical centers that had fewer imaging resources. This modified protocol offers a viable alternative that allows for minimizing heterogeneities while recognizing the capabilities of the available scanner fleet and meeting the needs of specific centers or veterans. Finally, the workgroup recognized a fundamental obstacle toward this harmonization process in the heterogeneity in vendors and scanner field strength, factors that have previously limited implementation.
The guidelines and proposed changes were then presented to the VA National Radiology Program Office, examined, and discussed for consensus. No changes were felt to be needed, and the recommendation to implement these guidelines in MS regional programs, whenever possible, was deemed appropriate.
At this time, a focused communication plan has been implemented to diffuse the use of this protocol at MS regional programs in the MSCoE network. We will work iteratively with individual sites to practically apply the guidelines, learn about challenges, and work through them to optimize local implementation.
Conclusions
Standardized MRI protocols are fundamental for the care of veterans with MS. Mitigating interscan variabilities should be recognized as a priority by scientific and clinical expert committees. Several guidelines have been developed over the years to standardize MRI acquisition protocols and interpretations, while updating the same to the latest discoveries.4,5,8,14,15 The VHA has been historically committed to these international efforts, with the goal to excel in the care of veterans with MS by providing access to state-of-the-art technologies. To this end, the initial Consortium of MS Centers MRI protocol was implemented in several MSCoE VA Regional Program sites a decade ago.14 Efforts continue to update protocol recommendations as needed and to promote their dissemination across the VHA enterprise.
This commentary is part of the continuous effort of the MSCoE to align with contemporary guidelines, apply the highest scientific standards, and achieve consistent outcomes for veterans with MS. For more important details of the clinical scenarios when additional/optional sequences or scans can be acquired, we advise the reader to refer to the 2021 MAGNIMS-CMSC-NAIMS Consensus Recommendations on the Use of MRI in Patients With Multiple Sclerosis.8
1. Wallin MT, Culpepper WJ, Campbell JD, et al. The prevalence of MS in the United States: A population-based estimate using health claims data. Neurology. 2019;92(10):e1029-e1040. doi:10.1212/WNL.0000000000007035
2. Nelson LM, Wallin MT, Marrie RA, et al. A new way to estimate neurologic disease prevalence in the United States: Illustrated with MS. Neurology. 2019;92(10):469-480. doi:10.1212/WNL.0000000000007044
3. Culpepper WJ, Wallin MT, Magder LS, et al. VHA Multiple Sclerosis Surveillance Registry and its similarities to other contemporary multiple sclerosis cohorts. J Rehabil Res Dev. 2015;52(3):263-272. doi:10.1682/JRRD.2014.07.0172
4. Wattjes MP, Rovira À, Miller D, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis--establishing disease prognosis and monitoring patients. Nat Rev Neurol. 2015;11(10):597-606. doi:10.1038/nrneurol.2015.157
5. Rovira À, Wattjes MP, Tintoré M, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis-clinical implementation in the diagnostic process. Nat Rev Neurol. 2015;11(8):471-482. doi:10.1038/nrneurol.2015.106
6. Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17(2):162-173. doi:10.1016/S1474-4422(17)30470-2
7. Reich DS, Lucchinetti CF, Calabresi PA. Multiple sclerosis. N Engl J Med. 2018;378(2):169-180. doi:10.1056/NEJMra1401483
8. Wattjes MP, Ciccarelli O, Reich DS, et al. 2021 MAGNIMS-CMSC-NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis. Lancet Neurol. 2021;20(8):653-670. doi:10.1016/S1474-4422(21)00095-8
9. Saslow L, Li DKB, Halper J, et al. An International Standardized Magnetic Resonance Imaging Protocol for Diagnosis and Follow-up of Patients with Multiple Sclerosis: Advocacy, Dissemination, and Implementation Strategies. Int J MS Care. 2020;22(5):226-232. doi:10.7224/1537-2073.2020-094
10. Cameron MH, Haselkorn JK, Wallin MT. The Multiple Sclerosis Centers of Excellence: a model of excellence in the VA. Fed Pract. 2020;37(suppl 1):S6-S10.
11. Grossman RI, Gonzalez-Scarano F, Atlas SW, Galetta S, Silberberg DH. Multiple sclerosis: gadolinium enhancement in MR imaging. Radiology. 1986;161(3):721-725. doi:10.1148/radiology.161.3.3786722
12. European Society of Urogenital Radiology. ESUR guidelines on contrast agent, 10.0. March 2018. Accessed March 11, 2022. https://www.esur.org/fileadmin/content/2019/ESUR_Guidelines_10.0_Final_Version.pdf
13. Sundgren PC, Leander P. Is administration of gadolinium-based contrast media to pregnant women and small children justified?. J Magn Reson Imaging. 2011;34(4):750-757. doi:10.1002/jmri.22413
14. Simon JH, Li D, Traboulsee A, et al. Standardized MR imaging protocol for multiple sclerosis: Consortium of MS Centers consensus guidelines. AJNR Am J Neuroradiol. 2006;27(2):455-461.
15. Traboulsee A, Simon JH, Stone L, et al. Revised Recommendations of the Consortium of MS Centers Task Force for a Standardized MRI Protocol and Clinical Guidelines for the Diagnosis and Follow-Up of Multiple Sclerosis. AJNR Am J Neuroradiol. 2016;37(3):394-401. doi:10.3174/ajnr.A4539
Multiple sclerosis (MS) is a lifelong disease that affects about a million people in the United States.1,2 Since 1998 more than 45,000 veterans have been diagnosed with MS and about 20,000 are evaluated in the Veterans Health Administration (VHA) annually.3
Magnetic resonance imaging (MRI) is a cornerstone for the assessment of persons with multiple sclerosis (pwMS).4-6 MRI assists with disease diagnosis, allowing for timely therapeutic interventions and withthe evaluation of its progression, treatment effect, and safety. 4,5 MRI-based outcomes also are used as primary endpoints in clinical trials.4,5
MS has its clinical onset in early adulthood in most individuals and is diagnosed at a mean age of 30 years.7 As a result, pwMS may receive care and MRIs in different facilities during their lifetime. Mitigating interscan variabilities that can challenge intra- and interperson comparisons is crucial for accurate care. Radiologists may find it difficult to compare scans acquired in different facilities, as dissimilarities in acquisition protocols may mask or uncover focal disease, creating false negative or false positive findings. Moreover, lack of a standardized method to report MRI changes may compromise neurologists’ ability to correctly interpret scans and disease progression.
Accordingly, in October 2019, an international task force of neurologists, radiologists, MRI technologists, and imaging scientists with expertise in MS, including representatives from the VHA, worked together to update guidelines for imaging the brain, spinal cord, and optic nerve in pwMS.8,9 Recognizing the importance of this effort, the VHA Multiple Sclerosis Centers of Excellence (MSCoE), in collaboration with a team of subject matter expert neuroradiologists promptly committed to this effort, advocating the updated consensus recommendations, and favoring their dissemination within the VHA.10
As part of this commitment and dissemination effort, in this report we summarize the core points of the newly proposed MRI guidelines and ways to adapt them for use within the VHA. We then discuss key elements for their successful implementation and dissemination, specifically regarding the clinical operations of VHA.
Updated Guidelines
MRI Scan at Different Timepoints of MS
There are 3 crucial milestones within a the lifespan of a pwMS that require an MRI to reach appropriate conclusions and avoid clinical errors. These include the initial diagnosis, the follow-up to monitor disease and/or treatment effect, and the assessment of medication safety.
In the interest of efficiency, MRI protocols may vary slightly depending on these clinical indications. The Table lists core sequences of the updated 2021 consensus recommendations at each timepoint along with the proposed alternatives or preferences from the VHA workgroup.
At the time of diagnosis, both brain and spine (cervical and thoracic) MRIs are recommended. Routine MRI of the optic nerve is considered optional at diagnosis. However, imaging the optic nerve may be useful in specific clinical scenarios when the optic nerve is selectively involved, and the diagnosis or etiology of an optic neuritis is not clear. A repeat brain MRI is advised every 6 to 12 months in patients with clinically or radiologically isolated syndrome who do not fulfill the diagnostic criteria of MS but present risk factors for conversion to MS or paraclinical features of it.
Once the diagnosis is established, brain MRI is recommended for follow-up and for surveillance of drug safety. Spinal cord and optic nerve MRIs are desirable but optional in the follow-up of pwMS and are not required for drug surveillance. Spinal cord MRIs are required at follow-up for patients whose progression cannot be explained by brain MRI features, or who manifest with recurrent spinal cord symptoms, or have spinal cord comorbidities. In these cases, spinal cord MRI also may assist with treatment decisions. Similarly, optic nerve MRI is necessary during follow-up only when optic nerve comorbidities are suspected or when there is progression or reoccurrence of optic nerve–related symptoms.
Brain MRIs are recommended for monitoring drug effect yearly (or at longer intervals, after a few years of disease stability). Conversely, a repeat brain MRI is advised after 6 months if nonsymptomatic radiological disease activity is discovered on surveillance scans.
Abbreviated but more frequent serial brain MRI protocols (eg, every 3 to 4 months) are recommended for pwMS treated with natalizumab and at high risk of developing progressive multifocal leukoencephalopathy (eg, pwMS who are John Cunningham virus [JCV]–positive, and have been treated with natalizumabfor ≥ 18 months, have a JCV antibody index > 0.9, or have a history of immunosuppression). A similar approach is recommended for carryover cases, such as those with high JCV antibody index who are switched to other immunosuppressive treatments.
MRI Field, Scan Resolution, and Coverage
Both 1.5-Tesla (1.5-T) and 3-T scans are believed to be equally effective in imaging pwMS, providing that the 1.5-T scans are good quality. Although imaging at < 1.5 T is not recommended due to suboptimal disease detection, the use of scanners > 3 T is equally discouraged outside the supervision of trained investigators. Signal-to-noise ratio and resolution are key factors impacting scan quality, and their optimization is prioritized over the number of sequences in the updated 2021 consensus recommendations. For brain imaging, a resolution of 1 mm3 isotropic is preferred for 3-dimensional (3D) imaging and slice thickness ≤ 3 mm without gap (≤ 5 mm with 10-30% gaps for diffusion-weighted imaging only) is recommended for 2D sequences. Images should cover the entire brain and as much of the cervical spine as possible; images should be prescribed axial for 2D or reformatted axial oblique for 3D using the subcallosal plane as reference. For spine imaging, sites should aim at an in-plane resolution of 1 mm2; using sagittal slices ≤ 3 mm thick and axial slices ≤ 5 mm thick, both with no gap. Scans should cover the entire cervical and thoracolumbar region inclusive of the conus. For the optic nerve images, slices should be ≤ 2 or 3 mm thick with an in-plane resolution of 1 mm2. Images should be aligned to the orientation of the optic nerve and chiasms, both of which should be entirely covered.
Postgadolinium Images Use
The discovery of the higher sensitivity of post-gadolinium (Gd) T1-weighted (T1-w) MRI relative to high iodine (88.1 g I) computed tomography scans in demonstrating contrast-enhancing MS lesions has revolutionized the way clinicians diagnose and monitor this disease.11 However, in recent years the role of postcontrast MRI has been debated, considering the potential safety concerns secondary to Gd tissue deposition. For this reason, an intentionally more judicious use of postcontrast MRI is proposed by the consensus recommendations. At disease diagnosis, the use of Gd is advisable to (1) show disease dissemination in time; (2) differentiate the diagnosis based on the Gd pattern; (3) predict short-term disease activity; and (4) characterize activity in the setting of progression. When monitoring pwMS, the use of Gd may be useful in the first year of follow-up, particularly if in the setting of low potency medications or for patients for whom the detection of one or more active lesions would lead to a change in disease-modifying agents. Gd also should be used to first, confirm a clinical exacerbation (if needed); second, further characterize a lesion suggestive of progressive multifocal encephalopathy or monitor this disease over time; and third, monitor lesion burden change in patients with large confluent lesions, the count of which otherwise may be difficult.
MRI During Pregnancy and Lactation
The consensus recommendations state that Gd contrast–enhanced MRI is not absolutely contraindicated during pregnancy, although its use should be limited to strictly necessary situations, particularly those involving differential diagnosis, such as cerebral venous thrombosis or monitoring of possibly enlarging lesion burden. The use of Gd is not contraindicated during lactation, as only a small proportion (< 0.4%) passes into the breast milk, leading to an exposure to < 1% of the permitted Gd dose for neonates.12,13
Harmonizing MRI Reports
The consensus recommendations propose reporting the exact lesion count on T2-weighted (T2-w) images when lesions are < 20, or specifying if the number of T2 lesions is between 20 and 50, between 50 and 100, or uncountable, eg, confluent large lesions. Similarly, for the spinal cord, the consensus recommendations propose reporting the exact lesion count on T2-w images when lesions are < 10, or otherwise report that > 10 lesions are seen.
The VHA workgroup proposed reporting a mild, moderate, or severe T2-lesion burden for a T2-lesion count < 20, between 20 and 50, and > 50, respectively. For follow-up MRIs, notation should be made if there is any change in lesion number, indicating the number of new lesions whenever possible. At each timepoint, the presence of active lesions on postcontrast images should be accurately defined.
Dissemination and Implementation
To implement and disseminate these proposed recommendations within the VHA, a workgroup of neurologists and radiologists was formed in late 2020. A review and discussion of the importance of each of the proposed MRI protocols for veterans with MS was held along with possible modifications to balance the intent of meeting standards of care with resources of individual US Department of Veterans Affairs (VA) medical centers and veterans’ needs. The final protocol recommendations were agreed on by group consensus.
In general, this VHA workgroup felt that the current adopted MRI protocols in several VA medical centers (based on previously proposed recommendations) were similar to the ones newly proposed and that implementing changes to meet the 2021 criteria would not be a major challenge.14,15 Possible regional and nonregional barriers were discussed. The result of these discussions led to a modified version of what could be considered more stringent guidelines to accommodate medical centers that had fewer imaging resources. This modified protocol offers a viable alternative that allows for minimizing heterogeneities while recognizing the capabilities of the available scanner fleet and meeting the needs of specific centers or veterans. Finally, the workgroup recognized a fundamental obstacle toward this harmonization process in the heterogeneity in vendors and scanner field strength, factors that have previously limited implementation.
The guidelines and proposed changes were then presented to the VA National Radiology Program Office, examined, and discussed for consensus. No changes were felt to be needed, and the recommendation to implement these guidelines in MS regional programs, whenever possible, was deemed appropriate.
At this time, a focused communication plan has been implemented to diffuse the use of this protocol at MS regional programs in the MSCoE network. We will work iteratively with individual sites to practically apply the guidelines, learn about challenges, and work through them to optimize local implementation.
Conclusions
Standardized MRI protocols are fundamental for the care of veterans with MS. Mitigating interscan variabilities should be recognized as a priority by scientific and clinical expert committees. Several guidelines have been developed over the years to standardize MRI acquisition protocols and interpretations, while updating the same to the latest discoveries.4,5,8,14,15 The VHA has been historically committed to these international efforts, with the goal to excel in the care of veterans with MS by providing access to state-of-the-art technologies. To this end, the initial Consortium of MS Centers MRI protocol was implemented in several MSCoE VA Regional Program sites a decade ago.14 Efforts continue to update protocol recommendations as needed and to promote their dissemination across the VHA enterprise.
This commentary is part of the continuous effort of the MSCoE to align with contemporary guidelines, apply the highest scientific standards, and achieve consistent outcomes for veterans with MS. For more important details of the clinical scenarios when additional/optional sequences or scans can be acquired, we advise the reader to refer to the 2021 MAGNIMS-CMSC-NAIMS Consensus Recommendations on the Use of MRI in Patients With Multiple Sclerosis.8
Multiple sclerosis (MS) is a lifelong disease that affects about a million people in the United States.1,2 Since 1998 more than 45,000 veterans have been diagnosed with MS and about 20,000 are evaluated in the Veterans Health Administration (VHA) annually.3
Magnetic resonance imaging (MRI) is a cornerstone for the assessment of persons with multiple sclerosis (pwMS).4-6 MRI assists with disease diagnosis, allowing for timely therapeutic interventions and withthe evaluation of its progression, treatment effect, and safety. 4,5 MRI-based outcomes also are used as primary endpoints in clinical trials.4,5
MS has its clinical onset in early adulthood in most individuals and is diagnosed at a mean age of 30 years.7 As a result, pwMS may receive care and MRIs in different facilities during their lifetime. Mitigating interscan variabilities that can challenge intra- and interperson comparisons is crucial for accurate care. Radiologists may find it difficult to compare scans acquired in different facilities, as dissimilarities in acquisition protocols may mask or uncover focal disease, creating false negative or false positive findings. Moreover, lack of a standardized method to report MRI changes may compromise neurologists’ ability to correctly interpret scans and disease progression.
Accordingly, in October 2019, an international task force of neurologists, radiologists, MRI technologists, and imaging scientists with expertise in MS, including representatives from the VHA, worked together to update guidelines for imaging the brain, spinal cord, and optic nerve in pwMS.8,9 Recognizing the importance of this effort, the VHA Multiple Sclerosis Centers of Excellence (MSCoE), in collaboration with a team of subject matter expert neuroradiologists promptly committed to this effort, advocating the updated consensus recommendations, and favoring their dissemination within the VHA.10
As part of this commitment and dissemination effort, in this report we summarize the core points of the newly proposed MRI guidelines and ways to adapt them for use within the VHA. We then discuss key elements for their successful implementation and dissemination, specifically regarding the clinical operations of VHA.
Updated Guidelines
MRI Scan at Different Timepoints of MS
There are 3 crucial milestones within a the lifespan of a pwMS that require an MRI to reach appropriate conclusions and avoid clinical errors. These include the initial diagnosis, the follow-up to monitor disease and/or treatment effect, and the assessment of medication safety.
In the interest of efficiency, MRI protocols may vary slightly depending on these clinical indications. The Table lists core sequences of the updated 2021 consensus recommendations at each timepoint along with the proposed alternatives or preferences from the VHA workgroup.
At the time of diagnosis, both brain and spine (cervical and thoracic) MRIs are recommended. Routine MRI of the optic nerve is considered optional at diagnosis. However, imaging the optic nerve may be useful in specific clinical scenarios when the optic nerve is selectively involved, and the diagnosis or etiology of an optic neuritis is not clear. A repeat brain MRI is advised every 6 to 12 months in patients with clinically or radiologically isolated syndrome who do not fulfill the diagnostic criteria of MS but present risk factors for conversion to MS or paraclinical features of it.
Once the diagnosis is established, brain MRI is recommended for follow-up and for surveillance of drug safety. Spinal cord and optic nerve MRIs are desirable but optional in the follow-up of pwMS and are not required for drug surveillance. Spinal cord MRIs are required at follow-up for patients whose progression cannot be explained by brain MRI features, or who manifest with recurrent spinal cord symptoms, or have spinal cord comorbidities. In these cases, spinal cord MRI also may assist with treatment decisions. Similarly, optic nerve MRI is necessary during follow-up only when optic nerve comorbidities are suspected or when there is progression or reoccurrence of optic nerve–related symptoms.
Brain MRIs are recommended for monitoring drug effect yearly (or at longer intervals, after a few years of disease stability). Conversely, a repeat brain MRI is advised after 6 months if nonsymptomatic radiological disease activity is discovered on surveillance scans.
Abbreviated but more frequent serial brain MRI protocols (eg, every 3 to 4 months) are recommended for pwMS treated with natalizumab and at high risk of developing progressive multifocal leukoencephalopathy (eg, pwMS who are John Cunningham virus [JCV]–positive, and have been treated with natalizumabfor ≥ 18 months, have a JCV antibody index > 0.9, or have a history of immunosuppression). A similar approach is recommended for carryover cases, such as those with high JCV antibody index who are switched to other immunosuppressive treatments.
MRI Field, Scan Resolution, and Coverage
Both 1.5-Tesla (1.5-T) and 3-T scans are believed to be equally effective in imaging pwMS, providing that the 1.5-T scans are good quality. Although imaging at < 1.5 T is not recommended due to suboptimal disease detection, the use of scanners > 3 T is equally discouraged outside the supervision of trained investigators. Signal-to-noise ratio and resolution are key factors impacting scan quality, and their optimization is prioritized over the number of sequences in the updated 2021 consensus recommendations. For brain imaging, a resolution of 1 mm3 isotropic is preferred for 3-dimensional (3D) imaging and slice thickness ≤ 3 mm without gap (≤ 5 mm with 10-30% gaps for diffusion-weighted imaging only) is recommended for 2D sequences. Images should cover the entire brain and as much of the cervical spine as possible; images should be prescribed axial for 2D or reformatted axial oblique for 3D using the subcallosal plane as reference. For spine imaging, sites should aim at an in-plane resolution of 1 mm2; using sagittal slices ≤ 3 mm thick and axial slices ≤ 5 mm thick, both with no gap. Scans should cover the entire cervical and thoracolumbar region inclusive of the conus. For the optic nerve images, slices should be ≤ 2 or 3 mm thick with an in-plane resolution of 1 mm2. Images should be aligned to the orientation of the optic nerve and chiasms, both of which should be entirely covered.
Postgadolinium Images Use
The discovery of the higher sensitivity of post-gadolinium (Gd) T1-weighted (T1-w) MRI relative to high iodine (88.1 g I) computed tomography scans in demonstrating contrast-enhancing MS lesions has revolutionized the way clinicians diagnose and monitor this disease.11 However, in recent years the role of postcontrast MRI has been debated, considering the potential safety concerns secondary to Gd tissue deposition. For this reason, an intentionally more judicious use of postcontrast MRI is proposed by the consensus recommendations. At disease diagnosis, the use of Gd is advisable to (1) show disease dissemination in time; (2) differentiate the diagnosis based on the Gd pattern; (3) predict short-term disease activity; and (4) characterize activity in the setting of progression. When monitoring pwMS, the use of Gd may be useful in the first year of follow-up, particularly if in the setting of low potency medications or for patients for whom the detection of one or more active lesions would lead to a change in disease-modifying agents. Gd also should be used to first, confirm a clinical exacerbation (if needed); second, further characterize a lesion suggestive of progressive multifocal encephalopathy or monitor this disease over time; and third, monitor lesion burden change in patients with large confluent lesions, the count of which otherwise may be difficult.
MRI During Pregnancy and Lactation
The consensus recommendations state that Gd contrast–enhanced MRI is not absolutely contraindicated during pregnancy, although its use should be limited to strictly necessary situations, particularly those involving differential diagnosis, such as cerebral venous thrombosis or monitoring of possibly enlarging lesion burden. The use of Gd is not contraindicated during lactation, as only a small proportion (< 0.4%) passes into the breast milk, leading to an exposure to < 1% of the permitted Gd dose for neonates.12,13
Harmonizing MRI Reports
The consensus recommendations propose reporting the exact lesion count on T2-weighted (T2-w) images when lesions are < 20, or specifying if the number of T2 lesions is between 20 and 50, between 50 and 100, or uncountable, eg, confluent large lesions. Similarly, for the spinal cord, the consensus recommendations propose reporting the exact lesion count on T2-w images when lesions are < 10, or otherwise report that > 10 lesions are seen.
The VHA workgroup proposed reporting a mild, moderate, or severe T2-lesion burden for a T2-lesion count < 20, between 20 and 50, and > 50, respectively. For follow-up MRIs, notation should be made if there is any change in lesion number, indicating the number of new lesions whenever possible. At each timepoint, the presence of active lesions on postcontrast images should be accurately defined.
Dissemination and Implementation
To implement and disseminate these proposed recommendations within the VHA, a workgroup of neurologists and radiologists was formed in late 2020. A review and discussion of the importance of each of the proposed MRI protocols for veterans with MS was held along with possible modifications to balance the intent of meeting standards of care with resources of individual US Department of Veterans Affairs (VA) medical centers and veterans’ needs. The final protocol recommendations were agreed on by group consensus.
In general, this VHA workgroup felt that the current adopted MRI protocols in several VA medical centers (based on previously proposed recommendations) were similar to the ones newly proposed and that implementing changes to meet the 2021 criteria would not be a major challenge.14,15 Possible regional and nonregional barriers were discussed. The result of these discussions led to a modified version of what could be considered more stringent guidelines to accommodate medical centers that had fewer imaging resources. This modified protocol offers a viable alternative that allows for minimizing heterogeneities while recognizing the capabilities of the available scanner fleet and meeting the needs of specific centers or veterans. Finally, the workgroup recognized a fundamental obstacle toward this harmonization process in the heterogeneity in vendors and scanner field strength, factors that have previously limited implementation.
The guidelines and proposed changes were then presented to the VA National Radiology Program Office, examined, and discussed for consensus. No changes were felt to be needed, and the recommendation to implement these guidelines in MS regional programs, whenever possible, was deemed appropriate.
At this time, a focused communication plan has been implemented to diffuse the use of this protocol at MS regional programs in the MSCoE network. We will work iteratively with individual sites to practically apply the guidelines, learn about challenges, and work through them to optimize local implementation.
Conclusions
Standardized MRI protocols are fundamental for the care of veterans with MS. Mitigating interscan variabilities should be recognized as a priority by scientific and clinical expert committees. Several guidelines have been developed over the years to standardize MRI acquisition protocols and interpretations, while updating the same to the latest discoveries.4,5,8,14,15 The VHA has been historically committed to these international efforts, with the goal to excel in the care of veterans with MS by providing access to state-of-the-art technologies. To this end, the initial Consortium of MS Centers MRI protocol was implemented in several MSCoE VA Regional Program sites a decade ago.14 Efforts continue to update protocol recommendations as needed and to promote their dissemination across the VHA enterprise.
This commentary is part of the continuous effort of the MSCoE to align with contemporary guidelines, apply the highest scientific standards, and achieve consistent outcomes for veterans with MS. For more important details of the clinical scenarios when additional/optional sequences or scans can be acquired, we advise the reader to refer to the 2021 MAGNIMS-CMSC-NAIMS Consensus Recommendations on the Use of MRI in Patients With Multiple Sclerosis.8
1. Wallin MT, Culpepper WJ, Campbell JD, et al. The prevalence of MS in the United States: A population-based estimate using health claims data. Neurology. 2019;92(10):e1029-e1040. doi:10.1212/WNL.0000000000007035
2. Nelson LM, Wallin MT, Marrie RA, et al. A new way to estimate neurologic disease prevalence in the United States: Illustrated with MS. Neurology. 2019;92(10):469-480. doi:10.1212/WNL.0000000000007044
3. Culpepper WJ, Wallin MT, Magder LS, et al. VHA Multiple Sclerosis Surveillance Registry and its similarities to other contemporary multiple sclerosis cohorts. J Rehabil Res Dev. 2015;52(3):263-272. doi:10.1682/JRRD.2014.07.0172
4. Wattjes MP, Rovira À, Miller D, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis--establishing disease prognosis and monitoring patients. Nat Rev Neurol. 2015;11(10):597-606. doi:10.1038/nrneurol.2015.157
5. Rovira À, Wattjes MP, Tintoré M, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis-clinical implementation in the diagnostic process. Nat Rev Neurol. 2015;11(8):471-482. doi:10.1038/nrneurol.2015.106
6. Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17(2):162-173. doi:10.1016/S1474-4422(17)30470-2
7. Reich DS, Lucchinetti CF, Calabresi PA. Multiple sclerosis. N Engl J Med. 2018;378(2):169-180. doi:10.1056/NEJMra1401483
8. Wattjes MP, Ciccarelli O, Reich DS, et al. 2021 MAGNIMS-CMSC-NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis. Lancet Neurol. 2021;20(8):653-670. doi:10.1016/S1474-4422(21)00095-8
9. Saslow L, Li DKB, Halper J, et al. An International Standardized Magnetic Resonance Imaging Protocol for Diagnosis and Follow-up of Patients with Multiple Sclerosis: Advocacy, Dissemination, and Implementation Strategies. Int J MS Care. 2020;22(5):226-232. doi:10.7224/1537-2073.2020-094
10. Cameron MH, Haselkorn JK, Wallin MT. The Multiple Sclerosis Centers of Excellence: a model of excellence in the VA. Fed Pract. 2020;37(suppl 1):S6-S10.
11. Grossman RI, Gonzalez-Scarano F, Atlas SW, Galetta S, Silberberg DH. Multiple sclerosis: gadolinium enhancement in MR imaging. Radiology. 1986;161(3):721-725. doi:10.1148/radiology.161.3.3786722
12. European Society of Urogenital Radiology. ESUR guidelines on contrast agent, 10.0. March 2018. Accessed March 11, 2022. https://www.esur.org/fileadmin/content/2019/ESUR_Guidelines_10.0_Final_Version.pdf
13. Sundgren PC, Leander P. Is administration of gadolinium-based contrast media to pregnant women and small children justified?. J Magn Reson Imaging. 2011;34(4):750-757. doi:10.1002/jmri.22413
14. Simon JH, Li D, Traboulsee A, et al. Standardized MR imaging protocol for multiple sclerosis: Consortium of MS Centers consensus guidelines. AJNR Am J Neuroradiol. 2006;27(2):455-461.
15. Traboulsee A, Simon JH, Stone L, et al. Revised Recommendations of the Consortium of MS Centers Task Force for a Standardized MRI Protocol and Clinical Guidelines for the Diagnosis and Follow-Up of Multiple Sclerosis. AJNR Am J Neuroradiol. 2016;37(3):394-401. doi:10.3174/ajnr.A4539
1. Wallin MT, Culpepper WJ, Campbell JD, et al. The prevalence of MS in the United States: A population-based estimate using health claims data. Neurology. 2019;92(10):e1029-e1040. doi:10.1212/WNL.0000000000007035
2. Nelson LM, Wallin MT, Marrie RA, et al. A new way to estimate neurologic disease prevalence in the United States: Illustrated with MS. Neurology. 2019;92(10):469-480. doi:10.1212/WNL.0000000000007044
3. Culpepper WJ, Wallin MT, Magder LS, et al. VHA Multiple Sclerosis Surveillance Registry and its similarities to other contemporary multiple sclerosis cohorts. J Rehabil Res Dev. 2015;52(3):263-272. doi:10.1682/JRRD.2014.07.0172
4. Wattjes MP, Rovira À, Miller D, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis--establishing disease prognosis and monitoring patients. Nat Rev Neurol. 2015;11(10):597-606. doi:10.1038/nrneurol.2015.157
5. Rovira À, Wattjes MP, Tintoré M, et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis-clinical implementation in the diagnostic process. Nat Rev Neurol. 2015;11(8):471-482. doi:10.1038/nrneurol.2015.106
6. Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17(2):162-173. doi:10.1016/S1474-4422(17)30470-2
7. Reich DS, Lucchinetti CF, Calabresi PA. Multiple sclerosis. N Engl J Med. 2018;378(2):169-180. doi:10.1056/NEJMra1401483
8. Wattjes MP, Ciccarelli O, Reich DS, et al. 2021 MAGNIMS-CMSC-NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis. Lancet Neurol. 2021;20(8):653-670. doi:10.1016/S1474-4422(21)00095-8
9. Saslow L, Li DKB, Halper J, et al. An International Standardized Magnetic Resonance Imaging Protocol for Diagnosis and Follow-up of Patients with Multiple Sclerosis: Advocacy, Dissemination, and Implementation Strategies. Int J MS Care. 2020;22(5):226-232. doi:10.7224/1537-2073.2020-094
10. Cameron MH, Haselkorn JK, Wallin MT. The Multiple Sclerosis Centers of Excellence: a model of excellence in the VA. Fed Pract. 2020;37(suppl 1):S6-S10.
11. Grossman RI, Gonzalez-Scarano F, Atlas SW, Galetta S, Silberberg DH. Multiple sclerosis: gadolinium enhancement in MR imaging. Radiology. 1986;161(3):721-725. doi:10.1148/radiology.161.3.3786722
12. European Society of Urogenital Radiology. ESUR guidelines on contrast agent, 10.0. March 2018. Accessed March 11, 2022. https://www.esur.org/fileadmin/content/2019/ESUR_Guidelines_10.0_Final_Version.pdf
13. Sundgren PC, Leander P. Is administration of gadolinium-based contrast media to pregnant women and small children justified?. J Magn Reson Imaging. 2011;34(4):750-757. doi:10.1002/jmri.22413
14. Simon JH, Li D, Traboulsee A, et al. Standardized MR imaging protocol for multiple sclerosis: Consortium of MS Centers consensus guidelines. AJNR Am J Neuroradiol. 2006;27(2):455-461.
15. Traboulsee A, Simon JH, Stone L, et al. Revised Recommendations of the Consortium of MS Centers Task Force for a Standardized MRI Protocol and Clinical Guidelines for the Diagnosis and Follow-Up of Multiple Sclerosis. AJNR Am J Neuroradiol. 2016;37(3):394-401. doi:10.3174/ajnr.A4539
No Veteran Leaves Alone: Ensuring Veterans Receive a Hero’s Final Salute
It is a great honor and privilege to care for the men and women who have bravely served our country, and to give a hero’s Final Salute in recognition of the veteran’s service and sacrifices. US Department of Veterans Affairs (VA) and other non-VA health care facilities caring for veterans find meaning and take pride in providing a Final Salute to veterans who spend their last days of life at their facilities. The Final Salute aligns with the mission of the VA: To fulfill President Lincoln’s promise “To care for him who shall have borne the battle, and for his widow, and his orphan” by serving and honoring the people who are America’s veterans.1 As health care professionals, we feel and grieve the loss when a veteran dies within our facilities. While some VA and community health care facilities honor veterans at the time of death, others have yet to implement a Final Salute program.2 How can we ensure that veterans at the time of death receive a hero’s Final Salute?
There are 26 million veterans alive today, representing about 8% of the total US adult population.3 Yet more than 1800 veterans die every day, representing about a quarter of all US deaths.4,5 Most veterans die in the community; only 4% of veteran deaths occur in VA facilities.5,6 This article highlights the unique tradition that a few VA and community health care facilities have launched to honor veterans whose journeys end under their care. This article also is a call to action to raise awareness of the importance of instituting the Final Salute program that is part of the end-of-life protocol for veterans.
A Final Salute ceremony (also called Honors Escort or Honor Walk) takes place when a veteran who dies in the hospital or nursing home is transported on the gurney from the location of their passing to the funeral home vehicle or the morgue. Staff, family members, visitors, and other veterans silently line the hallways from the veteran’s room to the health care facility exit and pay their respects to the deceased veteran. A Final Salute is a quiet, yet profound and powerful way for care teams to ensure that the deceased veteran does not leave alone.
VA-Based Ceremonies
There are many acts of remembrance at the bedside from the time of death to the time when the veteran’s body approaches the funeral home vehicle or the doors of the morgue. Tonya Ross, social worker and Honors Escort program manager at the Robert J. Dole VA Medical Center (VAMC) in Wichita, Kansas, reported that following the death of a veteran, there is a bedside remembrance that begins with a flag ceremony. Afterward, the veteran’s gurney is draped with the American flag, and as the procession moves through the medical center, the veterans salute, and all others place their hands over their hearts
Chaplain Michael Halyard at the Ozarks VAMC in Fayetteville, Arkansas, reported that following the death of a veteran, the chaplain greets family members with condolences and allows them to grieve and reflect on their life with the deceased veteran. On arrival of the funeral home team, an announcement for an Honor Walk is made. Staff, visitors, and family are lined up on the first floor of the hospital waiting to pay their final respects to the veteran. A slow processional of the veteran covered by a handmade quilt is escorted by a VA police officer and the chaplain. The processional stops in the middle and the chaplain announces, “Let us pause for a moment of silence as we honor one of our own US Army veterans who has completed the journey of life.”
The Final Salute at the VA Wilkes-Barre Community Living Center (CLC) in Pennsylvania begins with a bedside flag ceremony. Afterward, the veteran’s gurney is draped with the flag, and as the procession moves through the CLC, all who are standing along the route offer their respects. Throughout the ceremony, a team member remains with the family of the deceased, providing comfort and support. Once the ceremony is completed, the team member remains with the family to ensure all issues are addressed and all questions or concerns are answered.
Residents of the Philadelphia VAMC CLC in Pennsylvania have found a way to say a last goodbye to fellow veterans in a unique and dignified manner. Bettyanne Corkery, nurse manager for the Heroes’ Crossing hospice and palliative care unit explains, “Our Honor Guard evolved from our residents’ requests. We used to drape a flag over the body of veterans leaving us for the last time, but our residents came to us and said they wanted to do more.” CLC residents wanted to form an Honor Guard and say goodbye with dignity and grace. Gerry Donlon, a US Army Vietnam veteran and president of the residents council and chief program coordinator, explained that Honor Guard members are called to the deceased’s room and stand guard until the hearse comes. Donlon adds, “We proceed forward, along with the family, and the speaker system for the hospital plays patriotic songs, including Taps. When we get to the lobby, we stop, and I say a prayer. We fold the flag military style and hand it over to the family members, we render a Final Salute, and then the veteran is taken to the hearse.”7
Community Cermeonies
Texas Health Arlington Memorial Hospital (THAM) has honored 531 veterans with Final Salutes since 2015. Before the official procession begins, designated employees drape the patient’s body with the flag. Physicians, nurses, and volunteers escort the body in a silent procession along with the family. On leaving, the veteran’s family receives the flag in honor of their loved one. A specially designed medallion has been placed in the lobby floor at the location where the Final Salute is rendered. Christi Evans, RN, BSN, ACM, manager for care
coordination at AnMed Health, Anderson, South Carolina, witnessed a Final Salute at THAM for a relative and took the idea to Mike Johnston, Director of Spiritual Care to establish the program at AnMed Health, which has provided 118 Final Salutes since 2018.
Central Maine Healthcare (CMH), which operates 3 hospitals, provides 2 ceremonies. The Final Salute occurs prior to the veteran’s passing and the Honor Walk gathers hospital personnel outside the patient’s room as they are moved. During the Final Salute, with the approval of a veteran’s family, a veteran employed by CMF presents the veteran with a folded flag and certificate and thanks them for their service and hospital employee salute. After the veteran dies, staff members gather in the hallway for the Honor Walk. Ascension Sacred Heart (ASH), Florida, where on average 260 veterans look for treatment every month, has taken the Final Salute to all 4 of their hospitals. Sabrina Granese, BSN, RN, Military Service Line Director at ASH explains, “Patients that are active duty or veterans are identified at the time of admission. When a veteran passes away, with the approval of a veteran’s family, ‘Code veteran’ will be heard over the hospital intercom. Staff members will have 5 minutes to make their way to the main hospital entrance for the Honor Walk.” Similarly, the skilled nursing facilities operated by Bethesda Health Group, St. Louis, Missouri, have implemented the Veteran Escort Ceremony. Employees, volunteers, family members, and residents line the hallways during the procession to salute and honor the passing of the veteran’s body.
Closure For Families
Simple yet magnificent, a Final Salute shows that a veteran is “gone but not forgotten” and also shows families they are not alone as they too made sacrifices to allow their loved ones to serve in the Armed Forces; it signals the hope of healing and closure.8 “The staff came to pay their respects,” recalled Cindy Roberts, a social worker at the VA Bay Pines, when her relative died at the Ozarks VAMC. She explained, I wasn’t expecting as much because it was 2 AM. I have never in my life had an experience like that. I wish there were words to describe it; I wish every VAMC in the country did that.”
Hope Danishanko, social worker at the VA Wilkes-Barre CLC, said veterans are appreciative of the program. “I have had many CLC residents tell me that the Honors Escort allows them to have closure. They also feel it provides respect to the veteran who has passed.”
Bettyanne Corkery noted that the Philadelphia CLC Honor Guard program is unique because it is veteran driven. “They have sessions in which they talk about what works and what doesn’t, and they recruit new volunteers themselves,” she said. “It has evolved into the most beautiful ceremony, and they are constantly tweaking it.” According to Gerry Donlon, “When you see all 8 members of the Honor Guard get a call at 2 AM, and everyone shows up, you know there’s personal satisfaction. I’d like to see every CLC [throughout VA] do this. I really would.”7
“Family members tell us they feel blessed and honored to be a part of the program. They are so grateful for the way we pay tribute to their veteran loved one,” says Leslie Schaeffer, support services manager and bereavement coordinator and coordinator of the Veteran Escort Ceremony at Bethesda Health Group communities.
Privileged and humbled—that is how staff and family members describe feeling after participating in a Final Salute. Its impact on the families has been amazing. Between the tears, there are thanks for the recognition of the sacrifices their loved ones made. When one family was informed of the ceremony by Reverend Tricia Lytle, Manager of Spiritual Care at AnMed Health, the “whole family responded by explaining how much that meant at such a difficult time. They began sharing stories about his service and how proud he was to be a veteran,” she reported. “As I [Rev. Lytle] leaned over to present the flag at the bedside, the wife reached up and took hold as she tearfully accepted it and embraced it close to her heart. The staff in the hallway looked on respectfully also in tears.”
Conclusions
The Final Salute is a brief ceremonial procession demonstrating that the mission to care for America’s veterans does not end at the bedside. It ensures that no veteran’s body is alone when led out of the health facility room to the exit. With these Final Salute practices, I hope that the rest of VA and community health facilities caring for veterans will implement a Final Salute program to better honor veterans who depart in their care.
Acknowledgments
The author would like to express gratitude to everyone who so openly shared their stories—your insight, advice, and encouragement are inspiring and invaluable. Thank you to all the facilities that consented to be featured in this article.
1. US Department of Veteran Affairs. About VA: mission, vision, core values & goals. Updated September 30, 2021. Accessed September 30, 2021. https://www.va.gov /about_va/mission.asp
2. Kuznik R. Hospital program presentation, 2021 national convention. Accessed September 30, 2021. https:// vfwauxiliary.org/wp-content/uploads/2021.2022-National -Hospital-Ambassador-Presentation-Notes.pdf
3. US Department of Veteran Affairs, National Center for Veterans Analysis and Statistics. Veteran population projections 2017-2037. Published 2016. Accessed September 30, 2021. https://www.va.gov/vetdata/docs /Demographics/New_Vetpop_Model/Vetpop_Infographic _Final31.pdf
4. Calkins H. Psychologists, veterans and end-of-life care. Good Practice. Winter 2018. Accessed September 30, 2021. https://www.apaservices.org/practice/good -practice/veterans-end-of-life.pdf
5. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Accessed September 30, 2021. http://www.va.gov/vetdata
6. Grassman D. Veterans: an underserved population. Published 2007. Accessed September 30, 2021. https:// www.wehonorveterans.org/wp-content/uploads/2020/02 /WHVP_Toolkit.pdf
7. US Department of Veterans Affairs, VA Healthcare-VISN 4. An honorable procession: Philadelphia’s Honor Guard provides veterans a dignified farewell. 2015. Updated December 15, 2015. Accessed Semptember 30, 2021. https://www.visn4.va.gov/VISN4/news/vision/issue21 /honors-escort.asp
8. Nathan S, Dunn KM. Gone but not forgotten: how VA remembers. Federal Practitioner. 2019;36(6):254-256.
It is a great honor and privilege to care for the men and women who have bravely served our country, and to give a hero’s Final Salute in recognition of the veteran’s service and sacrifices. US Department of Veterans Affairs (VA) and other non-VA health care facilities caring for veterans find meaning and take pride in providing a Final Salute to veterans who spend their last days of life at their facilities. The Final Salute aligns with the mission of the VA: To fulfill President Lincoln’s promise “To care for him who shall have borne the battle, and for his widow, and his orphan” by serving and honoring the people who are America’s veterans.1 As health care professionals, we feel and grieve the loss when a veteran dies within our facilities. While some VA and community health care facilities honor veterans at the time of death, others have yet to implement a Final Salute program.2 How can we ensure that veterans at the time of death receive a hero’s Final Salute?
There are 26 million veterans alive today, representing about 8% of the total US adult population.3 Yet more than 1800 veterans die every day, representing about a quarter of all US deaths.4,5 Most veterans die in the community; only 4% of veteran deaths occur in VA facilities.5,6 This article highlights the unique tradition that a few VA and community health care facilities have launched to honor veterans whose journeys end under their care. This article also is a call to action to raise awareness of the importance of instituting the Final Salute program that is part of the end-of-life protocol for veterans.
A Final Salute ceremony (also called Honors Escort or Honor Walk) takes place when a veteran who dies in the hospital or nursing home is transported on the gurney from the location of their passing to the funeral home vehicle or the morgue. Staff, family members, visitors, and other veterans silently line the hallways from the veteran’s room to the health care facility exit and pay their respects to the deceased veteran. A Final Salute is a quiet, yet profound and powerful way for care teams to ensure that the deceased veteran does not leave alone.
VA-Based Ceremonies
There are many acts of remembrance at the bedside from the time of death to the time when the veteran’s body approaches the funeral home vehicle or the doors of the morgue. Tonya Ross, social worker and Honors Escort program manager at the Robert J. Dole VA Medical Center (VAMC) in Wichita, Kansas, reported that following the death of a veteran, there is a bedside remembrance that begins with a flag ceremony. Afterward, the veteran’s gurney is draped with the American flag, and as the procession moves through the medical center, the veterans salute, and all others place their hands over their hearts
Chaplain Michael Halyard at the Ozarks VAMC in Fayetteville, Arkansas, reported that following the death of a veteran, the chaplain greets family members with condolences and allows them to grieve and reflect on their life with the deceased veteran. On arrival of the funeral home team, an announcement for an Honor Walk is made. Staff, visitors, and family are lined up on the first floor of the hospital waiting to pay their final respects to the veteran. A slow processional of the veteran covered by a handmade quilt is escorted by a VA police officer and the chaplain. The processional stops in the middle and the chaplain announces, “Let us pause for a moment of silence as we honor one of our own US Army veterans who has completed the journey of life.”
The Final Salute at the VA Wilkes-Barre Community Living Center (CLC) in Pennsylvania begins with a bedside flag ceremony. Afterward, the veteran’s gurney is draped with the flag, and as the procession moves through the CLC, all who are standing along the route offer their respects. Throughout the ceremony, a team member remains with the family of the deceased, providing comfort and support. Once the ceremony is completed, the team member remains with the family to ensure all issues are addressed and all questions or concerns are answered.
Residents of the Philadelphia VAMC CLC in Pennsylvania have found a way to say a last goodbye to fellow veterans in a unique and dignified manner. Bettyanne Corkery, nurse manager for the Heroes’ Crossing hospice and palliative care unit explains, “Our Honor Guard evolved from our residents’ requests. We used to drape a flag over the body of veterans leaving us for the last time, but our residents came to us and said they wanted to do more.” CLC residents wanted to form an Honor Guard and say goodbye with dignity and grace. Gerry Donlon, a US Army Vietnam veteran and president of the residents council and chief program coordinator, explained that Honor Guard members are called to the deceased’s room and stand guard until the hearse comes. Donlon adds, “We proceed forward, along with the family, and the speaker system for the hospital plays patriotic songs, including Taps. When we get to the lobby, we stop, and I say a prayer. We fold the flag military style and hand it over to the family members, we render a Final Salute, and then the veteran is taken to the hearse.”7
Community Cermeonies
Texas Health Arlington Memorial Hospital (THAM) has honored 531 veterans with Final Salutes since 2015. Before the official procession begins, designated employees drape the patient’s body with the flag. Physicians, nurses, and volunteers escort the body in a silent procession along with the family. On leaving, the veteran’s family receives the flag in honor of their loved one. A specially designed medallion has been placed in the lobby floor at the location where the Final Salute is rendered. Christi Evans, RN, BSN, ACM, manager for care
coordination at AnMed Health, Anderson, South Carolina, witnessed a Final Salute at THAM for a relative and took the idea to Mike Johnston, Director of Spiritual Care to establish the program at AnMed Health, which has provided 118 Final Salutes since 2018.
Central Maine Healthcare (CMH), which operates 3 hospitals, provides 2 ceremonies. The Final Salute occurs prior to the veteran’s passing and the Honor Walk gathers hospital personnel outside the patient’s room as they are moved. During the Final Salute, with the approval of a veteran’s family, a veteran employed by CMF presents the veteran with a folded flag and certificate and thanks them for their service and hospital employee salute. After the veteran dies, staff members gather in the hallway for the Honor Walk. Ascension Sacred Heart (ASH), Florida, where on average 260 veterans look for treatment every month, has taken the Final Salute to all 4 of their hospitals. Sabrina Granese, BSN, RN, Military Service Line Director at ASH explains, “Patients that are active duty or veterans are identified at the time of admission. When a veteran passes away, with the approval of a veteran’s family, ‘Code veteran’ will be heard over the hospital intercom. Staff members will have 5 minutes to make their way to the main hospital entrance for the Honor Walk.” Similarly, the skilled nursing facilities operated by Bethesda Health Group, St. Louis, Missouri, have implemented the Veteran Escort Ceremony. Employees, volunteers, family members, and residents line the hallways during the procession to salute and honor the passing of the veteran’s body.
Closure For Families
Simple yet magnificent, a Final Salute shows that a veteran is “gone but not forgotten” and also shows families they are not alone as they too made sacrifices to allow their loved ones to serve in the Armed Forces; it signals the hope of healing and closure.8 “The staff came to pay their respects,” recalled Cindy Roberts, a social worker at the VA Bay Pines, when her relative died at the Ozarks VAMC. She explained, I wasn’t expecting as much because it was 2 AM. I have never in my life had an experience like that. I wish there were words to describe it; I wish every VAMC in the country did that.”
Hope Danishanko, social worker at the VA Wilkes-Barre CLC, said veterans are appreciative of the program. “I have had many CLC residents tell me that the Honors Escort allows them to have closure. They also feel it provides respect to the veteran who has passed.”
Bettyanne Corkery noted that the Philadelphia CLC Honor Guard program is unique because it is veteran driven. “They have sessions in which they talk about what works and what doesn’t, and they recruit new volunteers themselves,” she said. “It has evolved into the most beautiful ceremony, and they are constantly tweaking it.” According to Gerry Donlon, “When you see all 8 members of the Honor Guard get a call at 2 AM, and everyone shows up, you know there’s personal satisfaction. I’d like to see every CLC [throughout VA] do this. I really would.”7
“Family members tell us they feel blessed and honored to be a part of the program. They are so grateful for the way we pay tribute to their veteran loved one,” says Leslie Schaeffer, support services manager and bereavement coordinator and coordinator of the Veteran Escort Ceremony at Bethesda Health Group communities.
Privileged and humbled—that is how staff and family members describe feeling after participating in a Final Salute. Its impact on the families has been amazing. Between the tears, there are thanks for the recognition of the sacrifices their loved ones made. When one family was informed of the ceremony by Reverend Tricia Lytle, Manager of Spiritual Care at AnMed Health, the “whole family responded by explaining how much that meant at such a difficult time. They began sharing stories about his service and how proud he was to be a veteran,” she reported. “As I [Rev. Lytle] leaned over to present the flag at the bedside, the wife reached up and took hold as she tearfully accepted it and embraced it close to her heart. The staff in the hallway looked on respectfully also in tears.”
Conclusions
The Final Salute is a brief ceremonial procession demonstrating that the mission to care for America’s veterans does not end at the bedside. It ensures that no veteran’s body is alone when led out of the health facility room to the exit. With these Final Salute practices, I hope that the rest of VA and community health facilities caring for veterans will implement a Final Salute program to better honor veterans who depart in their care.
Acknowledgments
The author would like to express gratitude to everyone who so openly shared their stories—your insight, advice, and encouragement are inspiring and invaluable. Thank you to all the facilities that consented to be featured in this article.
It is a great honor and privilege to care for the men and women who have bravely served our country, and to give a hero’s Final Salute in recognition of the veteran’s service and sacrifices. US Department of Veterans Affairs (VA) and other non-VA health care facilities caring for veterans find meaning and take pride in providing a Final Salute to veterans who spend their last days of life at their facilities. The Final Salute aligns with the mission of the VA: To fulfill President Lincoln’s promise “To care for him who shall have borne the battle, and for his widow, and his orphan” by serving and honoring the people who are America’s veterans.1 As health care professionals, we feel and grieve the loss when a veteran dies within our facilities. While some VA and community health care facilities honor veterans at the time of death, others have yet to implement a Final Salute program.2 How can we ensure that veterans at the time of death receive a hero’s Final Salute?
There are 26 million veterans alive today, representing about 8% of the total US adult population.3 Yet more than 1800 veterans die every day, representing about a quarter of all US deaths.4,5 Most veterans die in the community; only 4% of veteran deaths occur in VA facilities.5,6 This article highlights the unique tradition that a few VA and community health care facilities have launched to honor veterans whose journeys end under their care. This article also is a call to action to raise awareness of the importance of instituting the Final Salute program that is part of the end-of-life protocol for veterans.
A Final Salute ceremony (also called Honors Escort or Honor Walk) takes place when a veteran who dies in the hospital or nursing home is transported on the gurney from the location of their passing to the funeral home vehicle or the morgue. Staff, family members, visitors, and other veterans silently line the hallways from the veteran’s room to the health care facility exit and pay their respects to the deceased veteran. A Final Salute is a quiet, yet profound and powerful way for care teams to ensure that the deceased veteran does not leave alone.
VA-Based Ceremonies
There are many acts of remembrance at the bedside from the time of death to the time when the veteran’s body approaches the funeral home vehicle or the doors of the morgue. Tonya Ross, social worker and Honors Escort program manager at the Robert J. Dole VA Medical Center (VAMC) in Wichita, Kansas, reported that following the death of a veteran, there is a bedside remembrance that begins with a flag ceremony. Afterward, the veteran’s gurney is draped with the American flag, and as the procession moves through the medical center, the veterans salute, and all others place their hands over their hearts
Chaplain Michael Halyard at the Ozarks VAMC in Fayetteville, Arkansas, reported that following the death of a veteran, the chaplain greets family members with condolences and allows them to grieve and reflect on their life with the deceased veteran. On arrival of the funeral home team, an announcement for an Honor Walk is made. Staff, visitors, and family are lined up on the first floor of the hospital waiting to pay their final respects to the veteran. A slow processional of the veteran covered by a handmade quilt is escorted by a VA police officer and the chaplain. The processional stops in the middle and the chaplain announces, “Let us pause for a moment of silence as we honor one of our own US Army veterans who has completed the journey of life.”
The Final Salute at the VA Wilkes-Barre Community Living Center (CLC) in Pennsylvania begins with a bedside flag ceremony. Afterward, the veteran’s gurney is draped with the flag, and as the procession moves through the CLC, all who are standing along the route offer their respects. Throughout the ceremony, a team member remains with the family of the deceased, providing comfort and support. Once the ceremony is completed, the team member remains with the family to ensure all issues are addressed and all questions or concerns are answered.
Residents of the Philadelphia VAMC CLC in Pennsylvania have found a way to say a last goodbye to fellow veterans in a unique and dignified manner. Bettyanne Corkery, nurse manager for the Heroes’ Crossing hospice and palliative care unit explains, “Our Honor Guard evolved from our residents’ requests. We used to drape a flag over the body of veterans leaving us for the last time, but our residents came to us and said they wanted to do more.” CLC residents wanted to form an Honor Guard and say goodbye with dignity and grace. Gerry Donlon, a US Army Vietnam veteran and president of the residents council and chief program coordinator, explained that Honor Guard members are called to the deceased’s room and stand guard until the hearse comes. Donlon adds, “We proceed forward, along with the family, and the speaker system for the hospital plays patriotic songs, including Taps. When we get to the lobby, we stop, and I say a prayer. We fold the flag military style and hand it over to the family members, we render a Final Salute, and then the veteran is taken to the hearse.”7
Community Cermeonies
Texas Health Arlington Memorial Hospital (THAM) has honored 531 veterans with Final Salutes since 2015. Before the official procession begins, designated employees drape the patient’s body with the flag. Physicians, nurses, and volunteers escort the body in a silent procession along with the family. On leaving, the veteran’s family receives the flag in honor of their loved one. A specially designed medallion has been placed in the lobby floor at the location where the Final Salute is rendered. Christi Evans, RN, BSN, ACM, manager for care
coordination at AnMed Health, Anderson, South Carolina, witnessed a Final Salute at THAM for a relative and took the idea to Mike Johnston, Director of Spiritual Care to establish the program at AnMed Health, which has provided 118 Final Salutes since 2018.
Central Maine Healthcare (CMH), which operates 3 hospitals, provides 2 ceremonies. The Final Salute occurs prior to the veteran’s passing and the Honor Walk gathers hospital personnel outside the patient’s room as they are moved. During the Final Salute, with the approval of a veteran’s family, a veteran employed by CMF presents the veteran with a folded flag and certificate and thanks them for their service and hospital employee salute. After the veteran dies, staff members gather in the hallway for the Honor Walk. Ascension Sacred Heart (ASH), Florida, where on average 260 veterans look for treatment every month, has taken the Final Salute to all 4 of their hospitals. Sabrina Granese, BSN, RN, Military Service Line Director at ASH explains, “Patients that are active duty or veterans are identified at the time of admission. When a veteran passes away, with the approval of a veteran’s family, ‘Code veteran’ will be heard over the hospital intercom. Staff members will have 5 minutes to make their way to the main hospital entrance for the Honor Walk.” Similarly, the skilled nursing facilities operated by Bethesda Health Group, St. Louis, Missouri, have implemented the Veteran Escort Ceremony. Employees, volunteers, family members, and residents line the hallways during the procession to salute and honor the passing of the veteran’s body.
Closure For Families
Simple yet magnificent, a Final Salute shows that a veteran is “gone but not forgotten” and also shows families they are not alone as they too made sacrifices to allow their loved ones to serve in the Armed Forces; it signals the hope of healing and closure.8 “The staff came to pay their respects,” recalled Cindy Roberts, a social worker at the VA Bay Pines, when her relative died at the Ozarks VAMC. She explained, I wasn’t expecting as much because it was 2 AM. I have never in my life had an experience like that. I wish there were words to describe it; I wish every VAMC in the country did that.”
Hope Danishanko, social worker at the VA Wilkes-Barre CLC, said veterans are appreciative of the program. “I have had many CLC residents tell me that the Honors Escort allows them to have closure. They also feel it provides respect to the veteran who has passed.”
Bettyanne Corkery noted that the Philadelphia CLC Honor Guard program is unique because it is veteran driven. “They have sessions in which they talk about what works and what doesn’t, and they recruit new volunteers themselves,” she said. “It has evolved into the most beautiful ceremony, and they are constantly tweaking it.” According to Gerry Donlon, “When you see all 8 members of the Honor Guard get a call at 2 AM, and everyone shows up, you know there’s personal satisfaction. I’d like to see every CLC [throughout VA] do this. I really would.”7
“Family members tell us they feel blessed and honored to be a part of the program. They are so grateful for the way we pay tribute to their veteran loved one,” says Leslie Schaeffer, support services manager and bereavement coordinator and coordinator of the Veteran Escort Ceremony at Bethesda Health Group communities.
Privileged and humbled—that is how staff and family members describe feeling after participating in a Final Salute. Its impact on the families has been amazing. Between the tears, there are thanks for the recognition of the sacrifices their loved ones made. When one family was informed of the ceremony by Reverend Tricia Lytle, Manager of Spiritual Care at AnMed Health, the “whole family responded by explaining how much that meant at such a difficult time. They began sharing stories about his service and how proud he was to be a veteran,” she reported. “As I [Rev. Lytle] leaned over to present the flag at the bedside, the wife reached up and took hold as she tearfully accepted it and embraced it close to her heart. The staff in the hallway looked on respectfully also in tears.”
Conclusions
The Final Salute is a brief ceremonial procession demonstrating that the mission to care for America’s veterans does not end at the bedside. It ensures that no veteran’s body is alone when led out of the health facility room to the exit. With these Final Salute practices, I hope that the rest of VA and community health facilities caring for veterans will implement a Final Salute program to better honor veterans who depart in their care.
Acknowledgments
The author would like to express gratitude to everyone who so openly shared their stories—your insight, advice, and encouragement are inspiring and invaluable. Thank you to all the facilities that consented to be featured in this article.
1. US Department of Veteran Affairs. About VA: mission, vision, core values & goals. Updated September 30, 2021. Accessed September 30, 2021. https://www.va.gov /about_va/mission.asp
2. Kuznik R. Hospital program presentation, 2021 national convention. Accessed September 30, 2021. https:// vfwauxiliary.org/wp-content/uploads/2021.2022-National -Hospital-Ambassador-Presentation-Notes.pdf
3. US Department of Veteran Affairs, National Center for Veterans Analysis and Statistics. Veteran population projections 2017-2037. Published 2016. Accessed September 30, 2021. https://www.va.gov/vetdata/docs /Demographics/New_Vetpop_Model/Vetpop_Infographic _Final31.pdf
4. Calkins H. Psychologists, veterans and end-of-life care. Good Practice. Winter 2018. Accessed September 30, 2021. https://www.apaservices.org/practice/good -practice/veterans-end-of-life.pdf
5. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Accessed September 30, 2021. http://www.va.gov/vetdata
6. Grassman D. Veterans: an underserved population. Published 2007. Accessed September 30, 2021. https:// www.wehonorveterans.org/wp-content/uploads/2020/02 /WHVP_Toolkit.pdf
7. US Department of Veterans Affairs, VA Healthcare-VISN 4. An honorable procession: Philadelphia’s Honor Guard provides veterans a dignified farewell. 2015. Updated December 15, 2015. Accessed Semptember 30, 2021. https://www.visn4.va.gov/VISN4/news/vision/issue21 /honors-escort.asp
8. Nathan S, Dunn KM. Gone but not forgotten: how VA remembers. Federal Practitioner. 2019;36(6):254-256.
1. US Department of Veteran Affairs. About VA: mission, vision, core values & goals. Updated September 30, 2021. Accessed September 30, 2021. https://www.va.gov /about_va/mission.asp
2. Kuznik R. Hospital program presentation, 2021 national convention. Accessed September 30, 2021. https:// vfwauxiliary.org/wp-content/uploads/2021.2022-National -Hospital-Ambassador-Presentation-Notes.pdf
3. US Department of Veteran Affairs, National Center for Veterans Analysis and Statistics. Veteran population projections 2017-2037. Published 2016. Accessed September 30, 2021. https://www.va.gov/vetdata/docs /Demographics/New_Vetpop_Model/Vetpop_Infographic _Final31.pdf
4. Calkins H. Psychologists, veterans and end-of-life care. Good Practice. Winter 2018. Accessed September 30, 2021. https://www.apaservices.org/practice/good -practice/veterans-end-of-life.pdf
5. US Department of Veterans Affairs, National Center for Veterans Analysis and Statistics. Accessed September 30, 2021. http://www.va.gov/vetdata
6. Grassman D. Veterans: an underserved population. Published 2007. Accessed September 30, 2021. https:// www.wehonorveterans.org/wp-content/uploads/2020/02 /WHVP_Toolkit.pdf
7. US Department of Veterans Affairs, VA Healthcare-VISN 4. An honorable procession: Philadelphia’s Honor Guard provides veterans a dignified farewell. 2015. Updated December 15, 2015. Accessed Semptember 30, 2021. https://www.visn4.va.gov/VISN4/news/vision/issue21 /honors-escort.asp
8. Nathan S, Dunn KM. Gone but not forgotten: how VA remembers. Federal Practitioner. 2019;36(6):254-256.
“Provider” Etymology is Unclear, but Still Wrong for Health Care
I am grateful for the opportunity to clarify and correct my recent commentary.1 I wrote that the word provider was first used to refer to health care professionals during the 1930s in Nazi Germany, when Jewish physicians were termed Behandler. The cited manuscript stated that Behandler was “freely translated” as “provider.”2 However, after reading social media comments that claimed this was a mistranslation, I sought to verify the translation.
Online German-English dictionaries yielded perplexing results. The dictionary Reverso translates Behandler as “dentist,” “practitioner,” or “therapist.”3 The Past Tenses Dictionary translates Behandler as “handlers.”4 Although a distasteful way to refer to a clinician-patient relationship, it still doesn’t translate as “provider.” The Collins and Cambridge dictionaries do not include Behandler, and the Langenscheidt dictionary does not provide a translation, instead noting that the translation “is missing” and that they are “verifying the word in question.”5-7 Conversely, Anbieter appears to be the commonly provided German translation for provider.
The author of the original manuscript acknowledged that although Behandler is not listed as a translation for provider, it “comes close.”2 He added that Behandler is not used anymore in German medicine because of the Nazi past (Saenger P, personal communication, February 9, 2022). A native German and Professor of German Studies at the University of Kentucky shared that “My best guess is that the term Behandler was used as a short form of Krankenbehandler, the designation for Jewish doctors in Nazi Germany who were still allowed to treat Jewish patients after withdrawal of their medical license. The best translations would be (health) practitioner or health care provider.” (Hobusch H, personal communication, 2022). However, Krankenbehandler has also been translated as “practitioner of the sick.”8
Given this ambiguity, it is ultimately unclear whether or to what extent Behandler can be translated as provider. Despite this uncertainty, my original argument remains unchanged. It is best to refer to all health care professionals (eg, psychotherapists, physicians, nurses, phlebotomists, pharmacists, physician assistants, social workers, physical therapists, dentists, optometrists) by their credentials. Overarching terms such as clinicians, practitioners, or health care professionals also are reasonable. This ensures accurate terminology, respects individuals’ unique training and degrees, and avoids confusion within multidisciplinary health care settings.
I thank Paul Saenger, MD, and Harald Höbusch, PhD, for their helpful insights, and those individuals who raised this concern on social media.
Correction: Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446- 448. The translation of the German word Behandler is unclear; therefore, the word “provider” should not be directly associated with the Nazi regime and its treatment of Jewish physicians.1. Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446-448. doi:10.12788/fp.0188
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.reverso.net/german-english /behandler/forced
4. Past Tenses. Translate behandler in English. Accessed March 4, 2022. https://pasttenses.com/behandler-german-english
5. Collins Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://www.collinsdictionary.com /dictionary/german-english/behandeln
6. Cambridge Dictionary, German-–English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.cambridge.org /spellcheck/german-english/?q=behandler
7. Langenscheidt Dictionary, German-English. Behandler. Accessed March 4, 2022. https://en.langenscheidt .com/german-english/search?term=behandler&q _cat=%2Fgerman-english%2F
8. Von Villiez A [trans, Kummer I]. The disenfranchisement of Jewish physicians in Hamburg during National Socialism. In: Institut fur die Geschichte der Deutschen Juden [Institute for the history of German Jews]. Key Documents of German-Jewish History. Updated September 16, 2016. Accessed March 16, 2022. doi: 10.23691/jgo:article-156.en.v1
I am grateful for the opportunity to clarify and correct my recent commentary.1 I wrote that the word provider was first used to refer to health care professionals during the 1930s in Nazi Germany, when Jewish physicians were termed Behandler. The cited manuscript stated that Behandler was “freely translated” as “provider.”2 However, after reading social media comments that claimed this was a mistranslation, I sought to verify the translation.
Online German-English dictionaries yielded perplexing results. The dictionary Reverso translates Behandler as “dentist,” “practitioner,” or “therapist.”3 The Past Tenses Dictionary translates Behandler as “handlers.”4 Although a distasteful way to refer to a clinician-patient relationship, it still doesn’t translate as “provider.” The Collins and Cambridge dictionaries do not include Behandler, and the Langenscheidt dictionary does not provide a translation, instead noting that the translation “is missing” and that they are “verifying the word in question.”5-7 Conversely, Anbieter appears to be the commonly provided German translation for provider.
The author of the original manuscript acknowledged that although Behandler is not listed as a translation for provider, it “comes close.”2 He added that Behandler is not used anymore in German medicine because of the Nazi past (Saenger P, personal communication, February 9, 2022). A native German and Professor of German Studies at the University of Kentucky shared that “My best guess is that the term Behandler was used as a short form of Krankenbehandler, the designation for Jewish doctors in Nazi Germany who were still allowed to treat Jewish patients after withdrawal of their medical license. The best translations would be (health) practitioner or health care provider.” (Hobusch H, personal communication, 2022). However, Krankenbehandler has also been translated as “practitioner of the sick.”8
Given this ambiguity, it is ultimately unclear whether or to what extent Behandler can be translated as provider. Despite this uncertainty, my original argument remains unchanged. It is best to refer to all health care professionals (eg, psychotherapists, physicians, nurses, phlebotomists, pharmacists, physician assistants, social workers, physical therapists, dentists, optometrists) by their credentials. Overarching terms such as clinicians, practitioners, or health care professionals also are reasonable. This ensures accurate terminology, respects individuals’ unique training and degrees, and avoids confusion within multidisciplinary health care settings.
I thank Paul Saenger, MD, and Harald Höbusch, PhD, for their helpful insights, and those individuals who raised this concern on social media.
Correction: Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446- 448. The translation of the German word Behandler is unclear; therefore, the word “provider” should not be directly associated with the Nazi regime and its treatment of Jewish physicians.I am grateful for the opportunity to clarify and correct my recent commentary.1 I wrote that the word provider was first used to refer to health care professionals during the 1930s in Nazi Germany, when Jewish physicians were termed Behandler. The cited manuscript stated that Behandler was “freely translated” as “provider.”2 However, after reading social media comments that claimed this was a mistranslation, I sought to verify the translation.
Online German-English dictionaries yielded perplexing results. The dictionary Reverso translates Behandler as “dentist,” “practitioner,” or “therapist.”3 The Past Tenses Dictionary translates Behandler as “handlers.”4 Although a distasteful way to refer to a clinician-patient relationship, it still doesn’t translate as “provider.” The Collins and Cambridge dictionaries do not include Behandler, and the Langenscheidt dictionary does not provide a translation, instead noting that the translation “is missing” and that they are “verifying the word in question.”5-7 Conversely, Anbieter appears to be the commonly provided German translation for provider.
The author of the original manuscript acknowledged that although Behandler is not listed as a translation for provider, it “comes close.”2 He added that Behandler is not used anymore in German medicine because of the Nazi past (Saenger P, personal communication, February 9, 2022). A native German and Professor of German Studies at the University of Kentucky shared that “My best guess is that the term Behandler was used as a short form of Krankenbehandler, the designation for Jewish doctors in Nazi Germany who were still allowed to treat Jewish patients after withdrawal of their medical license. The best translations would be (health) practitioner or health care provider.” (Hobusch H, personal communication, 2022). However, Krankenbehandler has also been translated as “practitioner of the sick.”8
Given this ambiguity, it is ultimately unclear whether or to what extent Behandler can be translated as provider. Despite this uncertainty, my original argument remains unchanged. It is best to refer to all health care professionals (eg, psychotherapists, physicians, nurses, phlebotomists, pharmacists, physician assistants, social workers, physical therapists, dentists, optometrists) by their credentials. Overarching terms such as clinicians, practitioners, or health care professionals also are reasonable. This ensures accurate terminology, respects individuals’ unique training and degrees, and avoids confusion within multidisciplinary health care settings.
I thank Paul Saenger, MD, and Harald Höbusch, PhD, for their helpful insights, and those individuals who raised this concern on social media.
Correction: Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446- 448. The translation of the German word Behandler is unclear; therefore, the word “provider” should not be directly associated with the Nazi regime and its treatment of Jewish physicians.1. Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446-448. doi:10.12788/fp.0188
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.reverso.net/german-english /behandler/forced
4. Past Tenses. Translate behandler in English. Accessed March 4, 2022. https://pasttenses.com/behandler-german-english
5. Collins Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://www.collinsdictionary.com /dictionary/german-english/behandeln
6. Cambridge Dictionary, German-–English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.cambridge.org /spellcheck/german-english/?q=behandler
7. Langenscheidt Dictionary, German-English. Behandler. Accessed March 4, 2022. https://en.langenscheidt .com/german-english/search?term=behandler&q _cat=%2Fgerman-english%2F
8. Von Villiez A [trans, Kummer I]. The disenfranchisement of Jewish physicians in Hamburg during National Socialism. In: Institut fur die Geschichte der Deutschen Juden [Institute for the history of German Jews]. Key Documents of German-Jewish History. Updated September 16, 2016. Accessed March 16, 2022. doi: 10.23691/jgo:article-156.en.v1
1. Scarff JR. What’s in a name? The problematic term “provider”. Fed Pract. 2021;38(10):446-448. doi:10.12788/fp.0188
2. Saenger P. Jewish pediatricians in Nazi Germany: victims of persecution. Isr Med Assoc J. 2006;8(5):324-328.
3. Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.reverso.net/german-english /behandler/forced
4. Past Tenses. Translate behandler in English. Accessed March 4, 2022. https://pasttenses.com/behandler-german-english
5. Collins Reverso German-English Dictionary. Behandler. Accessed March 16, 2022. https://www.collinsdictionary.com /dictionary/german-english/behandeln
6. Cambridge Dictionary, German-–English Dictionary. Behandler. Accessed March 16, 2022. https://dictionary.cambridge.org /spellcheck/german-english/?q=behandler
7. Langenscheidt Dictionary, German-English. Behandler. Accessed March 4, 2022. https://en.langenscheidt .com/german-english/search?term=behandler&q _cat=%2Fgerman-english%2F
8. Von Villiez A [trans, Kummer I]. The disenfranchisement of Jewish physicians in Hamburg during National Socialism. In: Institut fur die Geschichte der Deutschen Juden [Institute for the history of German Jews]. Key Documents of German-Jewish History. Updated September 16, 2016. Accessed March 16, 2022. doi: 10.23691/jgo:article-156.en.v1
Are all medical errors now crimes? The Nurse Vaught verdict
This video transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome! I’m Dr Robert Glatter, medical advisor for Medscape Emergency Medicine. Today we have a distinguished panel joining us to discuss an important legal decision resulting in a criminal conviction, involving a medical error due to administration of the wrong medication by a critical care nurse that led to a patient’s death.
Joining us to discuss this case is Dr. Megan Ranney, professor of emergency medicine and the academic dean at Brown University School of Public Health. Also joining us is Dr. Jane Barnsteiner, emeritus professor at the University of Pennsylvania School of Nursing and an expert on patient safety, quality improvement, and system modeling. Welcome to both of you.
Jane Barnsteiner, PhD, RN: Thank you.
Megan L. Ranney, MD, MPH: Thank you. It’s a joy to be with you.
Dr. Glatter: Let’s discuss this very tragic case involving RaDonda Vaught, who was an ICU nurse who was recently convicted in Tennessee of criminally negligent homicide and gross neglect of an impaired adult. She accidentally administered a paralytic medication, vecuronium, instead of a sedative, Versed, which was ordered to sedate a 75-year-old patient who had a brain bleed and TBI. She was scheduled to have a PET scan. After receiving the wrong medication and not really being monitored in any true way, just being in the care of an MRI tech, she suffered cardiac arrest and subsequently died.
Dr. Ranney, I want to begin with you. I saw on Twitter that you had written something that really stuck with me. I’ll quote you. “A culture of safety is one in which the system that allowed the mistake to happen is changed, not one in which the individual is scapegoated. And a culture of safety correlates with better patient outcomes that we know. This verdict is the opposite.”
I’ll let you explain from here. The system issue is the medication dispensing cabinet, in my mind, and there was a medication override. The question is, how was this override allowed to occur in the first place?
Dr. Ranney: My goodness, overrides happen every single day across this country, dozens of times a day in any particular shift. I would think of the system as being much bigger than just the Pyxis or that kind of automated dispensing cabinet, but around the larger system of the verbal orders, the time pressures that the nurse is under, the fact that the nurses are with a trainee, the fact that they’re being asked to operate outside of their normal environment by going down to MRI. There’s a series of issues.
Just as we thought about the Swiss cheese model for COVID-19, that model originated when we talked about patient safety and medical errors. It is a Swiss cheese of circumstances that allows this type of tragic error to occur.
Many of us have worked for years on trying to change the system from one of punishing people, changing it from that punitive system, to rather a system where we can do root-cause analysis, allow people to disclose errors, and allow us to inquire as to what are those series of Swiss cheese holes that allowed this mistake or any other to happen.
When you punish people, you lead them to hide their mistakes instead of allowing them to disclose them and allowing that important inquiry to happen. That’s why this is just so harmful to that culture of safety that so many of us are trying to create.
Dr. Glatter: It’s a chilling verdict in so many ways. I’m right on the same page with you, having worked for so long in the emergency department and seeing nurses that are overtaxed, overburdened, but also on patient floors. This goes to an ICU-type environment where this woman was having a nonemergent head scan and required some sedation.
The question I want to get to is how the system allowed the nurse to dispense this medication —though she was distracted, she’ll admit that. Jane, I want to get to you on this. How can we avoid this? What are the system checks that can be done in some fashion to make this safer and to avoid this tragic error?
Dr. Barnsteiner: First of all, I would say that you do not put in a major change, as they were doing with their EPIC system, as a big bank where you do the change through the entire organization. You do it in one area where you get the whole system smoothed out and all the errors taken care of so that you’re not having a problem like they had through their entire organization, which required overrides multiple times a day.
One of the things that’s been recommended is that these systems, like the Pyxis system, require the first five letters of a medication to be entered into the system so that when you have multiple medications where the first two letters are the same, the chances of pulling out the wrong medication are much smaller.
There’s a question of whether this medication, vecuronium, should have even been in this machine. You can have high-alert medications like this in baggies that have written on the front of the bag, “This is a high-alert medication. It requires two independent double checks.” These are all the things that will help alert the fatigued or distracted nurse or physician and will make things safer. There are many things that can be put into place.
Dr. Glatter: It’s almost like a hard stop. This is a different class of medication. Even if the nurse had a lapse and didn’t realize that, there should have been a hard stop asking whether you want this class. A sedative and a paralytic are two very different medications.
I’m not trying to assign any blame here. I’m just trying to look at mechanics of what happened and how we can put in place methods to avoid these types of errors where a system clearly is overtaxed and overburdened. Is it an artificial intelligence alert? Is it a pharmacy alert that goes out? Is it a Vocera message that gets triggered? It’s something to stop the nurse from doing something where they know better.
She’s used Versed before, apparently, and knows it’s a liquid and doesn’t have to be reconstituted. In my mind, as a practicing doctor for a long time, I see this and I see how it can happen. There are ways I think we can address it. Megan, I want to bring you into this and get your viewpoint.
Dr. Ranney: We’re working in an environment right now — and obviously, this happened pre-COVID — where medicines are constantly in short supply and we’re constantly dealing with substitutions of one for another. This has worsened during COVID, but it existed in the pre-COVID era as well. We’d have time periods where, like today, we’re out of D50 and we have to use D10, or we have a different formulation of a common antibiotic.
I could totally imagine that this nurse had been exposed to multiple medication substitution and so they were rushing; they thought, well, they just put one thing in instead of another and didn’t make that kind of cognitive connection.
What we know so well from our studies of human factors, engineering, and the way that systems work is that when someone is cognitively overloaded and constantly having to think outside the box and make decisions, particularly when they’re exposed to a new system for ordering medicine, there’s only so much that the brain can do at a time. This person was set up for this type of error.
Again, not to say that they didn’t do something wrong. That’s why we have a civil system. That’s why we have licensing. That’s why we have malpractice. To call this a criminal error when they were working within a system that had all these other problems where they were constantly having to make do for system failures, it’s almost inevitable that at some point something really horrible happened.
I’m sorry that it was this nurse, and how horrible for the patient and the family. I’m not excusing that. You can totally imagine, as a practicing physician, nurse, or anyone else in the healthcare system, how this happened.
Dr. Barnsteiner: The other part of it was that they did not have in place, at this time, the barcoding system in this particular patient area. What nurses are used to doing is when they have to pull a medication, they’re using the barcoding system to coordinate with what’s in the electronic health record, with the medication, and with the person’s ID band.
Those are all well-known safety checks that obviously were used to being used by this nurse in the critical care unit but that weren’t available in this MRI area. That is something that absolutely is a system failure. Those kinds of safety systems have to be available at any place in a health system where medications are being delivered.
Dr. Glatter: I think that’s an important point. Here, we have a technology that can supersede the ability of a human to make a mistake, and to have that in place is very critical. I want to go back to the idea of medical malpractice vs homicide charges.
Megan, you made a point of this. This nurse is now an example of someone who went to trial and was convicted, and it could have a chilling effect on healthcare providers. Pre-COVID, post-COVID, it is just chilling. It makes people want to leave the field. It causes PTSD. The psychiatric downstream effects of such an error are just immense.
I don’t know how the district attorney went for criminal charges here. I’m not an attorney and we don’t have a legal expert with us. For this to have happened is just setting precedent that it’s okay to have the effect of making so many people leave the field.
Dr. Ranney: I’m not a lawyer, but I’ve certainly been on the front lines, not only for the past 2 years during COVID but for almost 20 years prior to that. I will say that these types of errors are never-events that sit with our colleagues and friends for their entire career. No one goes into medicine intending to hurt someone. The system fails us and fails the patient.
There are certainly examples of intentional harm, and those people deserve to be prosecuted. This type of thing where a system let them down, again, should require an inquiry of the system. Don’t punish the individuals to the point of putting them in jail.
I think about my last few months working in the emergency department and what my nurses, in particular, have said to me. They worry that they’re going to lose their license and their ability to practice because of the horrific circumstances that we’ve been working in — the understaffing, the lack of access to standard medications, the long wait times, and on and on. They’re not able to take care of patients the way that they’ve been taught to do.
They’re worried already about the downstream effects on their sense of self, as well as on their ability to maintain their livelihood. When you put something like this on top of it, where again, an unintentional error that was potentiated by a somewhat broken system or by a series of Swiss cheese holes that just happened to line up, what message does that send to my nursing colleagues who have stayed on the front lines and who know that they have not been able to provide the standard of care that they’re used to?
Dr. Barnsteiner: On Friday, I did a program on fair and just culture with three health systems and a university school of nursing. Already, some of the faculty reported that students are talking about transferring to another major outside of the School of Nursing because of their worry about this particular guilty verdict.
The other thing is that we already have a tremendous shortage of nurses. We’ve seen many people leave the profession or retire in the past couple of years, and this is only going to compound it further. It is a sobering message that the public can’t afford to have, actually, because this will impact the quality of care and the safety of care that can be delivered to people and families as a result of not having sufficient numbers of professionals to deliver care.
Dr. Glatter: That’s such an important point. In any high-reliability organization, a culture of safety is key. There are tenets we try to adhere to. When we have people leaving the field after seeing a case like this, it’s chilling. We have to re-educate the public and we need to have a realignment of how errors are handled.
This is just the beginning. Her sentencing is going to be in about a month, and we’ll see what happens on reckless homicide charges and neglect. I think there’s going to be a follow-up to this and we’re going to need to discuss this more.
I just wanted to get a couple of takeaways for our audience to just really sear in the brain what we can learn from such an event.
Dr. Ranney: The big takeaway, to me, is the importance of us both continuing to use our voices and working across professional boundaries to help to create this culture of safety, one in which we all feel safe and supported in advocating for systems that work for us. We cannot ask nurses, respiratory technicians, radiology technicians, physicians, or anyone else within the healthcare system to work unsupported, and we have to recognize the degree to which we are all interdependent. My biggest takeaway is for us to use our voices together.
Dr. Barnsteiner: The takeaway that I would have from this, and what I’m working with a number of health systems on, is to have the chair of the board, the CEO of the hospital, the chief medical officer, and the chief nursing officer together promulgate a statement that is sent out to all employees to discuss this verdict and to say what they’re doing to promote a high-reliability organization and a fair and just culture. They should also ask for open conversation and for employees to let the top leadership know any concerns that they have about vulnerabilities in the system. It’s extremely important right now with this verdict that the leaders in healthcare settings, as well as in education settings, let people know what they’ll be doing to protect their employees.
Dr. Glatter: Jane and Megan, I want to thank you so much for such an important discussion that was very informative. I think there’s going to be a follow-up to this that’ll be very, very important. Thanks again.
Robert D. Glatter, MD, is assistant professor of emergency medicine at Lenox Hill Hospital in New York City and at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. He is an editorial advisor and hosts the Hot Topics in EM series on Medscape. He is also a medical contributor for Forbes. Dr. Glatter has disclosed no relevant financial relationships.
Megan Ranney, MD, MPH, is professor of emergency medicine and the academic dean at Brown University School of Public Health in Providence, Rhode Island. She is the director and founder of the Brown Emergency Digital Health Innovation (eDHI) program. She is also chief research officer for the American Foundation for Firearm Injury Reduction in Medicine, the country’s only nonprofit committed to reducing firearm injury through the public health approach, and a founding partner of GetUsPPE.org, dedicated to matching donors to health systems in need of protective equipment. Dr. Ranney has disclosed the following relevant financial relationships: Serve(d) as a speaker or a member of a speakers bureau for: Medscape; Merck.
Jane Barnsteiner, PhD, RN, is an emeritus professor at the University of Pennsylvania School of Nursing and an expert on patient safety, quality improvement, and system modeling. In addition to her teaching responsibilities, she was director of translational research at the Hospital of the University of Pennsylvania. Jane was one of the developers of the Quality and Safety in Education for Nurses (QSEN) initiative and is co-editor of Quality and Safety in Nursing: A Competency Based Approach to Improving Outcomes, published by Wiley. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This video transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome! I’m Dr Robert Glatter, medical advisor for Medscape Emergency Medicine. Today we have a distinguished panel joining us to discuss an important legal decision resulting in a criminal conviction, involving a medical error due to administration of the wrong medication by a critical care nurse that led to a patient’s death.
Joining us to discuss this case is Dr. Megan Ranney, professor of emergency medicine and the academic dean at Brown University School of Public Health. Also joining us is Dr. Jane Barnsteiner, emeritus professor at the University of Pennsylvania School of Nursing and an expert on patient safety, quality improvement, and system modeling. Welcome to both of you.
Jane Barnsteiner, PhD, RN: Thank you.
Megan L. Ranney, MD, MPH: Thank you. It’s a joy to be with you.
Dr. Glatter: Let’s discuss this very tragic case involving RaDonda Vaught, who was an ICU nurse who was recently convicted in Tennessee of criminally negligent homicide and gross neglect of an impaired adult. She accidentally administered a paralytic medication, vecuronium, instead of a sedative, Versed, which was ordered to sedate a 75-year-old patient who had a brain bleed and TBI. She was scheduled to have a PET scan. After receiving the wrong medication and not really being monitored in any true way, just being in the care of an MRI tech, she suffered cardiac arrest and subsequently died.
Dr. Ranney, I want to begin with you. I saw on Twitter that you had written something that really stuck with me. I’ll quote you. “A culture of safety is one in which the system that allowed the mistake to happen is changed, not one in which the individual is scapegoated. And a culture of safety correlates with better patient outcomes that we know. This verdict is the opposite.”
I’ll let you explain from here. The system issue is the medication dispensing cabinet, in my mind, and there was a medication override. The question is, how was this override allowed to occur in the first place?
Dr. Ranney: My goodness, overrides happen every single day across this country, dozens of times a day in any particular shift. I would think of the system as being much bigger than just the Pyxis or that kind of automated dispensing cabinet, but around the larger system of the verbal orders, the time pressures that the nurse is under, the fact that the nurses are with a trainee, the fact that they’re being asked to operate outside of their normal environment by going down to MRI. There’s a series of issues.
Just as we thought about the Swiss cheese model for COVID-19, that model originated when we talked about patient safety and medical errors. It is a Swiss cheese of circumstances that allows this type of tragic error to occur.
Many of us have worked for years on trying to change the system from one of punishing people, changing it from that punitive system, to rather a system where we can do root-cause analysis, allow people to disclose errors, and allow us to inquire as to what are those series of Swiss cheese holes that allowed this mistake or any other to happen.
When you punish people, you lead them to hide their mistakes instead of allowing them to disclose them and allowing that important inquiry to happen. That’s why this is just so harmful to that culture of safety that so many of us are trying to create.
Dr. Glatter: It’s a chilling verdict in so many ways. I’m right on the same page with you, having worked for so long in the emergency department and seeing nurses that are overtaxed, overburdened, but also on patient floors. This goes to an ICU-type environment where this woman was having a nonemergent head scan and required some sedation.
The question I want to get to is how the system allowed the nurse to dispense this medication —though she was distracted, she’ll admit that. Jane, I want to get to you on this. How can we avoid this? What are the system checks that can be done in some fashion to make this safer and to avoid this tragic error?
Dr. Barnsteiner: First of all, I would say that you do not put in a major change, as they were doing with their EPIC system, as a big bank where you do the change through the entire organization. You do it in one area where you get the whole system smoothed out and all the errors taken care of so that you’re not having a problem like they had through their entire organization, which required overrides multiple times a day.
One of the things that’s been recommended is that these systems, like the Pyxis system, require the first five letters of a medication to be entered into the system so that when you have multiple medications where the first two letters are the same, the chances of pulling out the wrong medication are much smaller.
There’s a question of whether this medication, vecuronium, should have even been in this machine. You can have high-alert medications like this in baggies that have written on the front of the bag, “This is a high-alert medication. It requires two independent double checks.” These are all the things that will help alert the fatigued or distracted nurse or physician and will make things safer. There are many things that can be put into place.
Dr. Glatter: It’s almost like a hard stop. This is a different class of medication. Even if the nurse had a lapse and didn’t realize that, there should have been a hard stop asking whether you want this class. A sedative and a paralytic are two very different medications.
I’m not trying to assign any blame here. I’m just trying to look at mechanics of what happened and how we can put in place methods to avoid these types of errors where a system clearly is overtaxed and overburdened. Is it an artificial intelligence alert? Is it a pharmacy alert that goes out? Is it a Vocera message that gets triggered? It’s something to stop the nurse from doing something where they know better.
She’s used Versed before, apparently, and knows it’s a liquid and doesn’t have to be reconstituted. In my mind, as a practicing doctor for a long time, I see this and I see how it can happen. There are ways I think we can address it. Megan, I want to bring you into this and get your viewpoint.
Dr. Ranney: We’re working in an environment right now — and obviously, this happened pre-COVID — where medicines are constantly in short supply and we’re constantly dealing with substitutions of one for another. This has worsened during COVID, but it existed in the pre-COVID era as well. We’d have time periods where, like today, we’re out of D50 and we have to use D10, or we have a different formulation of a common antibiotic.
I could totally imagine that this nurse had been exposed to multiple medication substitution and so they were rushing; they thought, well, they just put one thing in instead of another and didn’t make that kind of cognitive connection.
What we know so well from our studies of human factors, engineering, and the way that systems work is that when someone is cognitively overloaded and constantly having to think outside the box and make decisions, particularly when they’re exposed to a new system for ordering medicine, there’s only so much that the brain can do at a time. This person was set up for this type of error.
Again, not to say that they didn’t do something wrong. That’s why we have a civil system. That’s why we have licensing. That’s why we have malpractice. To call this a criminal error when they were working within a system that had all these other problems where they were constantly having to make do for system failures, it’s almost inevitable that at some point something really horrible happened.
I’m sorry that it was this nurse, and how horrible for the patient and the family. I’m not excusing that. You can totally imagine, as a practicing physician, nurse, or anyone else in the healthcare system, how this happened.
Dr. Barnsteiner: The other part of it was that they did not have in place, at this time, the barcoding system in this particular patient area. What nurses are used to doing is when they have to pull a medication, they’re using the barcoding system to coordinate with what’s in the electronic health record, with the medication, and with the person’s ID band.
Those are all well-known safety checks that obviously were used to being used by this nurse in the critical care unit but that weren’t available in this MRI area. That is something that absolutely is a system failure. Those kinds of safety systems have to be available at any place in a health system where medications are being delivered.
Dr. Glatter: I think that’s an important point. Here, we have a technology that can supersede the ability of a human to make a mistake, and to have that in place is very critical. I want to go back to the idea of medical malpractice vs homicide charges.
Megan, you made a point of this. This nurse is now an example of someone who went to trial and was convicted, and it could have a chilling effect on healthcare providers. Pre-COVID, post-COVID, it is just chilling. It makes people want to leave the field. It causes PTSD. The psychiatric downstream effects of such an error are just immense.
I don’t know how the district attorney went for criminal charges here. I’m not an attorney and we don’t have a legal expert with us. For this to have happened is just setting precedent that it’s okay to have the effect of making so many people leave the field.
Dr. Ranney: I’m not a lawyer, but I’ve certainly been on the front lines, not only for the past 2 years during COVID but for almost 20 years prior to that. I will say that these types of errors are never-events that sit with our colleagues and friends for their entire career. No one goes into medicine intending to hurt someone. The system fails us and fails the patient.
There are certainly examples of intentional harm, and those people deserve to be prosecuted. This type of thing where a system let them down, again, should require an inquiry of the system. Don’t punish the individuals to the point of putting them in jail.
I think about my last few months working in the emergency department and what my nurses, in particular, have said to me. They worry that they’re going to lose their license and their ability to practice because of the horrific circumstances that we’ve been working in — the understaffing, the lack of access to standard medications, the long wait times, and on and on. They’re not able to take care of patients the way that they’ve been taught to do.
They’re worried already about the downstream effects on their sense of self, as well as on their ability to maintain their livelihood. When you put something like this on top of it, where again, an unintentional error that was potentiated by a somewhat broken system or by a series of Swiss cheese holes that just happened to line up, what message does that send to my nursing colleagues who have stayed on the front lines and who know that they have not been able to provide the standard of care that they’re used to?
Dr. Barnsteiner: On Friday, I did a program on fair and just culture with three health systems and a university school of nursing. Already, some of the faculty reported that students are talking about transferring to another major outside of the School of Nursing because of their worry about this particular guilty verdict.
The other thing is that we already have a tremendous shortage of nurses. We’ve seen many people leave the profession or retire in the past couple of years, and this is only going to compound it further. It is a sobering message that the public can’t afford to have, actually, because this will impact the quality of care and the safety of care that can be delivered to people and families as a result of not having sufficient numbers of professionals to deliver care.
Dr. Glatter: That’s such an important point. In any high-reliability organization, a culture of safety is key. There are tenets we try to adhere to. When we have people leaving the field after seeing a case like this, it’s chilling. We have to re-educate the public and we need to have a realignment of how errors are handled.
This is just the beginning. Her sentencing is going to be in about a month, and we’ll see what happens on reckless homicide charges and neglect. I think there’s going to be a follow-up to this and we’re going to need to discuss this more.
I just wanted to get a couple of takeaways for our audience to just really sear in the brain what we can learn from such an event.
Dr. Ranney: The big takeaway, to me, is the importance of us both continuing to use our voices and working across professional boundaries to help to create this culture of safety, one in which we all feel safe and supported in advocating for systems that work for us. We cannot ask nurses, respiratory technicians, radiology technicians, physicians, or anyone else within the healthcare system to work unsupported, and we have to recognize the degree to which we are all interdependent. My biggest takeaway is for us to use our voices together.
Dr. Barnsteiner: The takeaway that I would have from this, and what I’m working with a number of health systems on, is to have the chair of the board, the CEO of the hospital, the chief medical officer, and the chief nursing officer together promulgate a statement that is sent out to all employees to discuss this verdict and to say what they’re doing to promote a high-reliability organization and a fair and just culture. They should also ask for open conversation and for employees to let the top leadership know any concerns that they have about vulnerabilities in the system. It’s extremely important right now with this verdict that the leaders in healthcare settings, as well as in education settings, let people know what they’ll be doing to protect their employees.
Dr. Glatter: Jane and Megan, I want to thank you so much for such an important discussion that was very informative. I think there’s going to be a follow-up to this that’ll be very, very important. Thanks again.
Robert D. Glatter, MD, is assistant professor of emergency medicine at Lenox Hill Hospital in New York City and at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. He is an editorial advisor and hosts the Hot Topics in EM series on Medscape. He is also a medical contributor for Forbes. Dr. Glatter has disclosed no relevant financial relationships.
Megan Ranney, MD, MPH, is professor of emergency medicine and the academic dean at Brown University School of Public Health in Providence, Rhode Island. She is the director and founder of the Brown Emergency Digital Health Innovation (eDHI) program. She is also chief research officer for the American Foundation for Firearm Injury Reduction in Medicine, the country’s only nonprofit committed to reducing firearm injury through the public health approach, and a founding partner of GetUsPPE.org, dedicated to matching donors to health systems in need of protective equipment. Dr. Ranney has disclosed the following relevant financial relationships: Serve(d) as a speaker or a member of a speakers bureau for: Medscape; Merck.
Jane Barnsteiner, PhD, RN, is an emeritus professor at the University of Pennsylvania School of Nursing and an expert on patient safety, quality improvement, and system modeling. In addition to her teaching responsibilities, she was director of translational research at the Hospital of the University of Pennsylvania. Jane was one of the developers of the Quality and Safety in Education for Nurses (QSEN) initiative and is co-editor of Quality and Safety in Nursing: A Competency Based Approach to Improving Outcomes, published by Wiley. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This video transcript has been edited for clarity.
Robert D. Glatter, MD: Welcome! I’m Dr Robert Glatter, medical advisor for Medscape Emergency Medicine. Today we have a distinguished panel joining us to discuss an important legal decision resulting in a criminal conviction, involving a medical error due to administration of the wrong medication by a critical care nurse that led to a patient’s death.
Joining us to discuss this case is Dr. Megan Ranney, professor of emergency medicine and the academic dean at Brown University School of Public Health. Also joining us is Dr. Jane Barnsteiner, emeritus professor at the University of Pennsylvania School of Nursing and an expert on patient safety, quality improvement, and system modeling. Welcome to both of you.
Jane Barnsteiner, PhD, RN: Thank you.
Megan L. Ranney, MD, MPH: Thank you. It’s a joy to be with you.
Dr. Glatter: Let’s discuss this very tragic case involving RaDonda Vaught, who was an ICU nurse who was recently convicted in Tennessee of criminally negligent homicide and gross neglect of an impaired adult. She accidentally administered a paralytic medication, vecuronium, instead of a sedative, Versed, which was ordered to sedate a 75-year-old patient who had a brain bleed and TBI. She was scheduled to have a PET scan. After receiving the wrong medication and not really being monitored in any true way, just being in the care of an MRI tech, she suffered cardiac arrest and subsequently died.
Dr. Ranney, I want to begin with you. I saw on Twitter that you had written something that really stuck with me. I’ll quote you. “A culture of safety is one in which the system that allowed the mistake to happen is changed, not one in which the individual is scapegoated. And a culture of safety correlates with better patient outcomes that we know. This verdict is the opposite.”
I’ll let you explain from here. The system issue is the medication dispensing cabinet, in my mind, and there was a medication override. The question is, how was this override allowed to occur in the first place?
Dr. Ranney: My goodness, overrides happen every single day across this country, dozens of times a day in any particular shift. I would think of the system as being much bigger than just the Pyxis or that kind of automated dispensing cabinet, but around the larger system of the verbal orders, the time pressures that the nurse is under, the fact that the nurses are with a trainee, the fact that they’re being asked to operate outside of their normal environment by going down to MRI. There’s a series of issues.
Just as we thought about the Swiss cheese model for COVID-19, that model originated when we talked about patient safety and medical errors. It is a Swiss cheese of circumstances that allows this type of tragic error to occur.
Many of us have worked for years on trying to change the system from one of punishing people, changing it from that punitive system, to rather a system where we can do root-cause analysis, allow people to disclose errors, and allow us to inquire as to what are those series of Swiss cheese holes that allowed this mistake or any other to happen.
When you punish people, you lead them to hide their mistakes instead of allowing them to disclose them and allowing that important inquiry to happen. That’s why this is just so harmful to that culture of safety that so many of us are trying to create.
Dr. Glatter: It’s a chilling verdict in so many ways. I’m right on the same page with you, having worked for so long in the emergency department and seeing nurses that are overtaxed, overburdened, but also on patient floors. This goes to an ICU-type environment where this woman was having a nonemergent head scan and required some sedation.
The question I want to get to is how the system allowed the nurse to dispense this medication —though she was distracted, she’ll admit that. Jane, I want to get to you on this. How can we avoid this? What are the system checks that can be done in some fashion to make this safer and to avoid this tragic error?
Dr. Barnsteiner: First of all, I would say that you do not put in a major change, as they were doing with their EPIC system, as a big bank where you do the change through the entire organization. You do it in one area where you get the whole system smoothed out and all the errors taken care of so that you’re not having a problem like they had through their entire organization, which required overrides multiple times a day.
One of the things that’s been recommended is that these systems, like the Pyxis system, require the first five letters of a medication to be entered into the system so that when you have multiple medications where the first two letters are the same, the chances of pulling out the wrong medication are much smaller.
There’s a question of whether this medication, vecuronium, should have even been in this machine. You can have high-alert medications like this in baggies that have written on the front of the bag, “This is a high-alert medication. It requires two independent double checks.” These are all the things that will help alert the fatigued or distracted nurse or physician and will make things safer. There are many things that can be put into place.
Dr. Glatter: It’s almost like a hard stop. This is a different class of medication. Even if the nurse had a lapse and didn’t realize that, there should have been a hard stop asking whether you want this class. A sedative and a paralytic are two very different medications.
I’m not trying to assign any blame here. I’m just trying to look at mechanics of what happened and how we can put in place methods to avoid these types of errors where a system clearly is overtaxed and overburdened. Is it an artificial intelligence alert? Is it a pharmacy alert that goes out? Is it a Vocera message that gets triggered? It’s something to stop the nurse from doing something where they know better.
She’s used Versed before, apparently, and knows it’s a liquid and doesn’t have to be reconstituted. In my mind, as a practicing doctor for a long time, I see this and I see how it can happen. There are ways I think we can address it. Megan, I want to bring you into this and get your viewpoint.
Dr. Ranney: We’re working in an environment right now — and obviously, this happened pre-COVID — where medicines are constantly in short supply and we’re constantly dealing with substitutions of one for another. This has worsened during COVID, but it existed in the pre-COVID era as well. We’d have time periods where, like today, we’re out of D50 and we have to use D10, or we have a different formulation of a common antibiotic.
I could totally imagine that this nurse had been exposed to multiple medication substitution and so they were rushing; they thought, well, they just put one thing in instead of another and didn’t make that kind of cognitive connection.
What we know so well from our studies of human factors, engineering, and the way that systems work is that when someone is cognitively overloaded and constantly having to think outside the box and make decisions, particularly when they’re exposed to a new system for ordering medicine, there’s only so much that the brain can do at a time. This person was set up for this type of error.
Again, not to say that they didn’t do something wrong. That’s why we have a civil system. That’s why we have licensing. That’s why we have malpractice. To call this a criminal error when they were working within a system that had all these other problems where they were constantly having to make do for system failures, it’s almost inevitable that at some point something really horrible happened.
I’m sorry that it was this nurse, and how horrible for the patient and the family. I’m not excusing that. You can totally imagine, as a practicing physician, nurse, or anyone else in the healthcare system, how this happened.
Dr. Barnsteiner: The other part of it was that they did not have in place, at this time, the barcoding system in this particular patient area. What nurses are used to doing is when they have to pull a medication, they’re using the barcoding system to coordinate with what’s in the electronic health record, with the medication, and with the person’s ID band.
Those are all well-known safety checks that obviously were used to being used by this nurse in the critical care unit but that weren’t available in this MRI area. That is something that absolutely is a system failure. Those kinds of safety systems have to be available at any place in a health system where medications are being delivered.
Dr. Glatter: I think that’s an important point. Here, we have a technology that can supersede the ability of a human to make a mistake, and to have that in place is very critical. I want to go back to the idea of medical malpractice vs homicide charges.
Megan, you made a point of this. This nurse is now an example of someone who went to trial and was convicted, and it could have a chilling effect on healthcare providers. Pre-COVID, post-COVID, it is just chilling. It makes people want to leave the field. It causes PTSD. The psychiatric downstream effects of such an error are just immense.
I don’t know how the district attorney went for criminal charges here. I’m not an attorney and we don’t have a legal expert with us. For this to have happened is just setting precedent that it’s okay to have the effect of making so many people leave the field.
Dr. Ranney: I’m not a lawyer, but I’ve certainly been on the front lines, not only for the past 2 years during COVID but for almost 20 years prior to that. I will say that these types of errors are never-events that sit with our colleagues and friends for their entire career. No one goes into medicine intending to hurt someone. The system fails us and fails the patient.
There are certainly examples of intentional harm, and those people deserve to be prosecuted. This type of thing where a system let them down, again, should require an inquiry of the system. Don’t punish the individuals to the point of putting them in jail.
I think about my last few months working in the emergency department and what my nurses, in particular, have said to me. They worry that they’re going to lose their license and their ability to practice because of the horrific circumstances that we’ve been working in — the understaffing, the lack of access to standard medications, the long wait times, and on and on. They’re not able to take care of patients the way that they’ve been taught to do.
They’re worried already about the downstream effects on their sense of self, as well as on their ability to maintain their livelihood. When you put something like this on top of it, where again, an unintentional error that was potentiated by a somewhat broken system or by a series of Swiss cheese holes that just happened to line up, what message does that send to my nursing colleagues who have stayed on the front lines and who know that they have not been able to provide the standard of care that they’re used to?
Dr. Barnsteiner: On Friday, I did a program on fair and just culture with three health systems and a university school of nursing. Already, some of the faculty reported that students are talking about transferring to another major outside of the School of Nursing because of their worry about this particular guilty verdict.
The other thing is that we already have a tremendous shortage of nurses. We’ve seen many people leave the profession or retire in the past couple of years, and this is only going to compound it further. It is a sobering message that the public can’t afford to have, actually, because this will impact the quality of care and the safety of care that can be delivered to people and families as a result of not having sufficient numbers of professionals to deliver care.
Dr. Glatter: That’s such an important point. In any high-reliability organization, a culture of safety is key. There are tenets we try to adhere to. When we have people leaving the field after seeing a case like this, it’s chilling. We have to re-educate the public and we need to have a realignment of how errors are handled.
This is just the beginning. Her sentencing is going to be in about a month, and we’ll see what happens on reckless homicide charges and neglect. I think there’s going to be a follow-up to this and we’re going to need to discuss this more.
I just wanted to get a couple of takeaways for our audience to just really sear in the brain what we can learn from such an event.
Dr. Ranney: The big takeaway, to me, is the importance of us both continuing to use our voices and working across professional boundaries to help to create this culture of safety, one in which we all feel safe and supported in advocating for systems that work for us. We cannot ask nurses, respiratory technicians, radiology technicians, physicians, or anyone else within the healthcare system to work unsupported, and we have to recognize the degree to which we are all interdependent. My biggest takeaway is for us to use our voices together.
Dr. Barnsteiner: The takeaway that I would have from this, and what I’m working with a number of health systems on, is to have the chair of the board, the CEO of the hospital, the chief medical officer, and the chief nursing officer together promulgate a statement that is sent out to all employees to discuss this verdict and to say what they’re doing to promote a high-reliability organization and a fair and just culture. They should also ask for open conversation and for employees to let the top leadership know any concerns that they have about vulnerabilities in the system. It’s extremely important right now with this verdict that the leaders in healthcare settings, as well as in education settings, let people know what they’ll be doing to protect their employees.
Dr. Glatter: Jane and Megan, I want to thank you so much for such an important discussion that was very informative. I think there’s going to be a follow-up to this that’ll be very, very important. Thanks again.
Robert D. Glatter, MD, is assistant professor of emergency medicine at Lenox Hill Hospital in New York City and at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. He is an editorial advisor and hosts the Hot Topics in EM series on Medscape. He is also a medical contributor for Forbes. Dr. Glatter has disclosed no relevant financial relationships.
Megan Ranney, MD, MPH, is professor of emergency medicine and the academic dean at Brown University School of Public Health in Providence, Rhode Island. She is the director and founder of the Brown Emergency Digital Health Innovation (eDHI) program. She is also chief research officer for the American Foundation for Firearm Injury Reduction in Medicine, the country’s only nonprofit committed to reducing firearm injury through the public health approach, and a founding partner of GetUsPPE.org, dedicated to matching donors to health systems in need of protective equipment. Dr. Ranney has disclosed the following relevant financial relationships: Serve(d) as a speaker or a member of a speakers bureau for: Medscape; Merck.
Jane Barnsteiner, PhD, RN, is an emeritus professor at the University of Pennsylvania School of Nursing and an expert on patient safety, quality improvement, and system modeling. In addition to her teaching responsibilities, she was director of translational research at the Hospital of the University of Pennsylvania. Jane was one of the developers of the Quality and Safety in Education for Nurses (QSEN) initiative and is co-editor of Quality and Safety in Nursing: A Competency Based Approach to Improving Outcomes, published by Wiley. She has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Has the anti-benzodiazepine backlash gone too far?
When benzodiazepines were first introduced, they were greeted with enthusiasm. Librium came first, in 1960, followed by Valium in 1962, and they were seen as an improvement over barbiturates for the treatment of anxiety, insomnia, and seizures. From 1968 to 1982, Valium (diazepam) was the No. 1–selling U.S. pharmaceutical: 2.3 billion tablets of Valium were sold in 1978 alone. Valium was even the subject of a 1966 Rolling Stones hit, “Mother’s Little Helper.”
By the 1980s, it became apparent that there was a downside to these medications: patients became tolerant, dependent, and some became addicted to the medications. In older patients an association was noted with falls and cognitive impairment. And while safe in overdoses when they are the only agent, combined with alcohol or opioids, benzodiazepines can be lethal and have played a significant role in the current overdose crisis.
Because of the problems that are associated with their use, benzodiazepines and their relatives, the Z-drugs used for sleep, have become stigmatized, as have the patients who use them and perhaps even the doctors who prescribe them. Still, there are circumstances where patients find these medications to be helpful, where other medications don’t work, or don’t work quickly enough. They provide fast relief in conditions where there are not always good alternatives.
In the Facebook group, “Psychiatry for All Physicians,” it’s not uncommon for physicians to ask what to do with older patients who are transferred to them on therapeutic doses of benzodiazepines or zolpidem. These are outpatients coming for routine care, and they find the medications helpful and don’t want to discontinue them. They have tried other medications that were not helpful. I’ve been surprised at how often the respondents insist the patient should be told he must taper off the medication. “Just say no,” is often the advice, and perhaps it’s more about the doctor’s discomfort than it is about the individual patient. For sleep issues, cognitive-behavioral therapy is given as the gold-standard treatment, while in my practice I have found it difficult to motivate patients to engage in it, and of those who do, it is sometimes helpful, but not a panacea. Severe anxiety and sleepless nights, however, are not benign conditions.
This “just say no, hold the line” sentiment has me wondering if our pendulum has swung too far with respect to prescribing benzodiazepines. Is this just one more issue that has become strongly polarized? Certainly the literature would support that idea, with some physicians writing about how benzodiazepines are underused, and others urging avoidance.
I posted a poll on Twitter: Has the anti-benzo movement gone too far? In addition, I started a Twitter thread of my own thoughts about prescribing and deprescribing these medications and will give a synopsis of those ideas here.
Clearly, benzodiazepines are harmful to some patients, they have side effects, can be difficult to stop because of withdrawal symptoms, and they carry the risk of addiction. That’s not in question. Many medications, however, have the potential to do more harm than good, for example ibuprofen can cause bleeding or renal problems, and Fosamax, used to treat osteoporosis, can cause osteonecrosis of the jaw and femur, to name just two.
It would be so much easier if we could know in advance who benzodiazepines will harm, just as it would be good if we could know in advance who will get tardive dyskinesia or dyslipidemia from antipsychotic medications, or who will have life-threatening adverse reactions from cancer chemotherapy with no tumor response. There are risks to both starting and stopping sedatives, and if we insist a patient stop a medication because of potential risk, then we are cutting them off from being a partner in their own care. It also creates an adversarial relationship that can be draining for the doctor and upsetting for the patient.
By definition, if someone needs hospitalization for a psychiatric condition, their outpatient benzodiazepine is not keeping them stable and stopping it may be a good idea. If someone is seen in an ED for a fall, it’s common to blame the benzodiazepine, but older people who are not on these medications also fall and have memory problems. In his book, “Being Mortal: Illness, Medicine, and What Matters Most in the End” (New York: Picador, 2014), Atul Gawande, MD, makes the point that taking more than four prescriptions medications increases the risk for falls in the elderly. Still, no one is suggesting patients be taken off their antidepressants, antihypertensives, or blood thinners.
Finally, the question is not should we be giving benzodiazepines out without careful consideration – the answer is clearly no. Physicians don’t pass out benzodiazepines “like candy” for all the above reasons. They are initiated because the patient is suffering and sometimes desperate. Anxiety, panic, intractable insomnia, and severe agitation are all miserable, and alternative treatments may take weeks to work, or not work at all. Yet these subjective symptoms may be dismissed by physicians.
So what do I do in my own practice? I don’t encourage patients to take potentially addictive medications, but I do sometimes use them. I give ‘as needed’ benzodiazepines to people in distress who don’t have a history of misusing them. I never plan to start them as a permanent standing medication, though once in a while that ends up happening. As with other medications, it is best to use the minimally effective dose.
There is some controversy as to whether it is best to use anxiety medications on an “as-needed” basis or as a standing dosage. Psychiatrists who prescribe benzodiazepines more liberally often feel it’s better to give standing doses and prevent breakthrough anxiety. Patients may appear to be ‘medication seeking’ not because they are addicted, but because the doses used are too low to adequately treat their anxiety.
My hope is that there is less risk of tolerance, dependence, or addiction with less-frequent dosing, and I prescribe as-needed benzodiazepines for panic attacks, agitated major depression while we wait for the antidepressant to “kick in,” insomnia during manic episodes, and to people who get very anxious in specific situations such as flying or for medical procedures. I sometimes prescribe them for people with insomnia that does not respond to other treatments, or for disabling generalized anxiety.
For patients who have taken benzodiazepines for many years, I continue to discuss the risks, but often they are not looking to fix something that isn’t broken, or to live a risk-free life. A few of the patients who have come to me on low standing doses of sedatives are now in their 80’s, yet they remain active, live independently, drive, travel, and have busy social lives. One could argue either that the medications are working, or that the patient has become dependent on them and needs them to prevent withdrawal.
These medications present a quandary: by denying patients treatment with benzodiazepines, we are sparing some people addictions (this is good, we should be careful), but we are leaving some people to suffer. There is no perfect answer.
What I do know is that doctors should think carefully and consider the patient in front of them. “No Benzos Ever For Anyone” or “you must come off because there is risk and people will think I am a bad doctor for prescribing them to you” can be done by a robot.
So, yes, I think the pendulum has swung a bit too far; there is a place for these medications in acute treatment for those at low risk of addiction, and there are people who benefit from them over the long run. At times, they provide immense relief to someone who is really struggling.
So what was the result of my Twitter poll? Of the 219 voters, 34.2% voted: “No, the pendulum has not swung too far, and these medications are harmful”; 65.8% voted: “Yes, these medications are helpful.” There were many comments expressing a wide variety of sentiments. Of those who had taken prescription benzodiazepines, some felt they had been harmed and wished they had never been started on them, and others continue to find them helpful. Psychiatrists, it seems, see them from the vantage point of the populations they treat.
People who are uncomfortable search for answers, and those answers may come in the form of meditation or exercise, medicines, or illicit drugs. It’s interesting that these same patients can now easily obtain “medical” marijuana, and the Rolling Stones’ “Mother’s Little Helper” is often replaced by a gin and tonic.
Dr. Dinah Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. Dr. Miller has no conflicts of interest.
When benzodiazepines were first introduced, they were greeted with enthusiasm. Librium came first, in 1960, followed by Valium in 1962, and they were seen as an improvement over barbiturates for the treatment of anxiety, insomnia, and seizures. From 1968 to 1982, Valium (diazepam) was the No. 1–selling U.S. pharmaceutical: 2.3 billion tablets of Valium were sold in 1978 alone. Valium was even the subject of a 1966 Rolling Stones hit, “Mother’s Little Helper.”
By the 1980s, it became apparent that there was a downside to these medications: patients became tolerant, dependent, and some became addicted to the medications. In older patients an association was noted with falls and cognitive impairment. And while safe in overdoses when they are the only agent, combined with alcohol or opioids, benzodiazepines can be lethal and have played a significant role in the current overdose crisis.
Because of the problems that are associated with their use, benzodiazepines and their relatives, the Z-drugs used for sleep, have become stigmatized, as have the patients who use them and perhaps even the doctors who prescribe them. Still, there are circumstances where patients find these medications to be helpful, where other medications don’t work, or don’t work quickly enough. They provide fast relief in conditions where there are not always good alternatives.
In the Facebook group, “Psychiatry for All Physicians,” it’s not uncommon for physicians to ask what to do with older patients who are transferred to them on therapeutic doses of benzodiazepines or zolpidem. These are outpatients coming for routine care, and they find the medications helpful and don’t want to discontinue them. They have tried other medications that were not helpful. I’ve been surprised at how often the respondents insist the patient should be told he must taper off the medication. “Just say no,” is often the advice, and perhaps it’s more about the doctor’s discomfort than it is about the individual patient. For sleep issues, cognitive-behavioral therapy is given as the gold-standard treatment, while in my practice I have found it difficult to motivate patients to engage in it, and of those who do, it is sometimes helpful, but not a panacea. Severe anxiety and sleepless nights, however, are not benign conditions.
This “just say no, hold the line” sentiment has me wondering if our pendulum has swung too far with respect to prescribing benzodiazepines. Is this just one more issue that has become strongly polarized? Certainly the literature would support that idea, with some physicians writing about how benzodiazepines are underused, and others urging avoidance.
I posted a poll on Twitter: Has the anti-benzo movement gone too far? In addition, I started a Twitter thread of my own thoughts about prescribing and deprescribing these medications and will give a synopsis of those ideas here.
Clearly, benzodiazepines are harmful to some patients, they have side effects, can be difficult to stop because of withdrawal symptoms, and they carry the risk of addiction. That’s not in question. Many medications, however, have the potential to do more harm than good, for example ibuprofen can cause bleeding or renal problems, and Fosamax, used to treat osteoporosis, can cause osteonecrosis of the jaw and femur, to name just two.
It would be so much easier if we could know in advance who benzodiazepines will harm, just as it would be good if we could know in advance who will get tardive dyskinesia or dyslipidemia from antipsychotic medications, or who will have life-threatening adverse reactions from cancer chemotherapy with no tumor response. There are risks to both starting and stopping sedatives, and if we insist a patient stop a medication because of potential risk, then we are cutting them off from being a partner in their own care. It also creates an adversarial relationship that can be draining for the doctor and upsetting for the patient.
By definition, if someone needs hospitalization for a psychiatric condition, their outpatient benzodiazepine is not keeping them stable and stopping it may be a good idea. If someone is seen in an ED for a fall, it’s common to blame the benzodiazepine, but older people who are not on these medications also fall and have memory problems. In his book, “Being Mortal: Illness, Medicine, and What Matters Most in the End” (New York: Picador, 2014), Atul Gawande, MD, makes the point that taking more than four prescriptions medications increases the risk for falls in the elderly. Still, no one is suggesting patients be taken off their antidepressants, antihypertensives, or blood thinners.
Finally, the question is not should we be giving benzodiazepines out without careful consideration – the answer is clearly no. Physicians don’t pass out benzodiazepines “like candy” for all the above reasons. They are initiated because the patient is suffering and sometimes desperate. Anxiety, panic, intractable insomnia, and severe agitation are all miserable, and alternative treatments may take weeks to work, or not work at all. Yet these subjective symptoms may be dismissed by physicians.
So what do I do in my own practice? I don’t encourage patients to take potentially addictive medications, but I do sometimes use them. I give ‘as needed’ benzodiazepines to people in distress who don’t have a history of misusing them. I never plan to start them as a permanent standing medication, though once in a while that ends up happening. As with other medications, it is best to use the minimally effective dose.
There is some controversy as to whether it is best to use anxiety medications on an “as-needed” basis or as a standing dosage. Psychiatrists who prescribe benzodiazepines more liberally often feel it’s better to give standing doses and prevent breakthrough anxiety. Patients may appear to be ‘medication seeking’ not because they are addicted, but because the doses used are too low to adequately treat their anxiety.
My hope is that there is less risk of tolerance, dependence, or addiction with less-frequent dosing, and I prescribe as-needed benzodiazepines for panic attacks, agitated major depression while we wait for the antidepressant to “kick in,” insomnia during manic episodes, and to people who get very anxious in specific situations such as flying or for medical procedures. I sometimes prescribe them for people with insomnia that does not respond to other treatments, or for disabling generalized anxiety.
For patients who have taken benzodiazepines for many years, I continue to discuss the risks, but often they are not looking to fix something that isn’t broken, or to live a risk-free life. A few of the patients who have come to me on low standing doses of sedatives are now in their 80’s, yet they remain active, live independently, drive, travel, and have busy social lives. One could argue either that the medications are working, or that the patient has become dependent on them and needs them to prevent withdrawal.
These medications present a quandary: by denying patients treatment with benzodiazepines, we are sparing some people addictions (this is good, we should be careful), but we are leaving some people to suffer. There is no perfect answer.
What I do know is that doctors should think carefully and consider the patient in front of them. “No Benzos Ever For Anyone” or “you must come off because there is risk and people will think I am a bad doctor for prescribing them to you” can be done by a robot.
So, yes, I think the pendulum has swung a bit too far; there is a place for these medications in acute treatment for those at low risk of addiction, and there are people who benefit from them over the long run. At times, they provide immense relief to someone who is really struggling.
So what was the result of my Twitter poll? Of the 219 voters, 34.2% voted: “No, the pendulum has not swung too far, and these medications are harmful”; 65.8% voted: “Yes, these medications are helpful.” There were many comments expressing a wide variety of sentiments. Of those who had taken prescription benzodiazepines, some felt they had been harmed and wished they had never been started on them, and others continue to find them helpful. Psychiatrists, it seems, see them from the vantage point of the populations they treat.
People who are uncomfortable search for answers, and those answers may come in the form of meditation or exercise, medicines, or illicit drugs. It’s interesting that these same patients can now easily obtain “medical” marijuana, and the Rolling Stones’ “Mother’s Little Helper” is often replaced by a gin and tonic.
Dr. Dinah Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. Dr. Miller has no conflicts of interest.
When benzodiazepines were first introduced, they were greeted with enthusiasm. Librium came first, in 1960, followed by Valium in 1962, and they were seen as an improvement over barbiturates for the treatment of anxiety, insomnia, and seizures. From 1968 to 1982, Valium (diazepam) was the No. 1–selling U.S. pharmaceutical: 2.3 billion tablets of Valium were sold in 1978 alone. Valium was even the subject of a 1966 Rolling Stones hit, “Mother’s Little Helper.”
By the 1980s, it became apparent that there was a downside to these medications: patients became tolerant, dependent, and some became addicted to the medications. In older patients an association was noted with falls and cognitive impairment. And while safe in overdoses when they are the only agent, combined with alcohol or opioids, benzodiazepines can be lethal and have played a significant role in the current overdose crisis.
Because of the problems that are associated with their use, benzodiazepines and their relatives, the Z-drugs used for sleep, have become stigmatized, as have the patients who use them and perhaps even the doctors who prescribe them. Still, there are circumstances where patients find these medications to be helpful, where other medications don’t work, or don’t work quickly enough. They provide fast relief in conditions where there are not always good alternatives.
In the Facebook group, “Psychiatry for All Physicians,” it’s not uncommon for physicians to ask what to do with older patients who are transferred to them on therapeutic doses of benzodiazepines or zolpidem. These are outpatients coming for routine care, and they find the medications helpful and don’t want to discontinue them. They have tried other medications that were not helpful. I’ve been surprised at how often the respondents insist the patient should be told he must taper off the medication. “Just say no,” is often the advice, and perhaps it’s more about the doctor’s discomfort than it is about the individual patient. For sleep issues, cognitive-behavioral therapy is given as the gold-standard treatment, while in my practice I have found it difficult to motivate patients to engage in it, and of those who do, it is sometimes helpful, but not a panacea. Severe anxiety and sleepless nights, however, are not benign conditions.
This “just say no, hold the line” sentiment has me wondering if our pendulum has swung too far with respect to prescribing benzodiazepines. Is this just one more issue that has become strongly polarized? Certainly the literature would support that idea, with some physicians writing about how benzodiazepines are underused, and others urging avoidance.
I posted a poll on Twitter: Has the anti-benzo movement gone too far? In addition, I started a Twitter thread of my own thoughts about prescribing and deprescribing these medications and will give a synopsis of those ideas here.
Clearly, benzodiazepines are harmful to some patients, they have side effects, can be difficult to stop because of withdrawal symptoms, and they carry the risk of addiction. That’s not in question. Many medications, however, have the potential to do more harm than good, for example ibuprofen can cause bleeding or renal problems, and Fosamax, used to treat osteoporosis, can cause osteonecrosis of the jaw and femur, to name just two.
It would be so much easier if we could know in advance who benzodiazepines will harm, just as it would be good if we could know in advance who will get tardive dyskinesia or dyslipidemia from antipsychotic medications, or who will have life-threatening adverse reactions from cancer chemotherapy with no tumor response. There are risks to both starting and stopping sedatives, and if we insist a patient stop a medication because of potential risk, then we are cutting them off from being a partner in their own care. It also creates an adversarial relationship that can be draining for the doctor and upsetting for the patient.
By definition, if someone needs hospitalization for a psychiatric condition, their outpatient benzodiazepine is not keeping them stable and stopping it may be a good idea. If someone is seen in an ED for a fall, it’s common to blame the benzodiazepine, but older people who are not on these medications also fall and have memory problems. In his book, “Being Mortal: Illness, Medicine, and What Matters Most in the End” (New York: Picador, 2014), Atul Gawande, MD, makes the point that taking more than four prescriptions medications increases the risk for falls in the elderly. Still, no one is suggesting patients be taken off their antidepressants, antihypertensives, or blood thinners.
Finally, the question is not should we be giving benzodiazepines out without careful consideration – the answer is clearly no. Physicians don’t pass out benzodiazepines “like candy” for all the above reasons. They are initiated because the patient is suffering and sometimes desperate. Anxiety, panic, intractable insomnia, and severe agitation are all miserable, and alternative treatments may take weeks to work, or not work at all. Yet these subjective symptoms may be dismissed by physicians.
So what do I do in my own practice? I don’t encourage patients to take potentially addictive medications, but I do sometimes use them. I give ‘as needed’ benzodiazepines to people in distress who don’t have a history of misusing them. I never plan to start them as a permanent standing medication, though once in a while that ends up happening. As with other medications, it is best to use the minimally effective dose.
There is some controversy as to whether it is best to use anxiety medications on an “as-needed” basis or as a standing dosage. Psychiatrists who prescribe benzodiazepines more liberally often feel it’s better to give standing doses and prevent breakthrough anxiety. Patients may appear to be ‘medication seeking’ not because they are addicted, but because the doses used are too low to adequately treat their anxiety.
My hope is that there is less risk of tolerance, dependence, or addiction with less-frequent dosing, and I prescribe as-needed benzodiazepines for panic attacks, agitated major depression while we wait for the antidepressant to “kick in,” insomnia during manic episodes, and to people who get very anxious in specific situations such as flying or for medical procedures. I sometimes prescribe them for people with insomnia that does not respond to other treatments, or for disabling generalized anxiety.
For patients who have taken benzodiazepines for many years, I continue to discuss the risks, but often they are not looking to fix something that isn’t broken, or to live a risk-free life. A few of the patients who have come to me on low standing doses of sedatives are now in their 80’s, yet they remain active, live independently, drive, travel, and have busy social lives. One could argue either that the medications are working, or that the patient has become dependent on them and needs them to prevent withdrawal.
These medications present a quandary: by denying patients treatment with benzodiazepines, we are sparing some people addictions (this is good, we should be careful), but we are leaving some people to suffer. There is no perfect answer.
What I do know is that doctors should think carefully and consider the patient in front of them. “No Benzos Ever For Anyone” or “you must come off because there is risk and people will think I am a bad doctor for prescribing them to you” can be done by a robot.
So, yes, I think the pendulum has swung a bit too far; there is a place for these medications in acute treatment for those at low risk of addiction, and there are people who benefit from them over the long run. At times, they provide immense relief to someone who is really struggling.
So what was the result of my Twitter poll? Of the 219 voters, 34.2% voted: “No, the pendulum has not swung too far, and these medications are harmful”; 65.8% voted: “Yes, these medications are helpful.” There were many comments expressing a wide variety of sentiments. Of those who had taken prescription benzodiazepines, some felt they had been harmed and wished they had never been started on them, and others continue to find them helpful. Psychiatrists, it seems, see them from the vantage point of the populations they treat.
People who are uncomfortable search for answers, and those answers may come in the form of meditation or exercise, medicines, or illicit drugs. It’s interesting that these same patients can now easily obtain “medical” marijuana, and the Rolling Stones’ “Mother’s Little Helper” is often replaced by a gin and tonic.
Dr. Dinah Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. Dr. Miller has no conflicts of interest.