User login
Borderline personality disorder: Remember empathy and compassion
Oh, great!” a senior resident sardonically remarked with a smirk as they read up on the next patient in the clinic. “A borderline patient. Get ready for a rough one ... Ugh.”
Before ever stepping foot into the patient’s room, this resident had prematurely established and demonstrated an unfortunate dynamic for any student or trainee within earshot. This is an all-too-familiar occurrence when caring for individuals with borderline personality disorder (BPD), or any other patients deemed to be “difficult.” The patient, however, likely walked into the room with a traumatic past that they continue to suffer from, in addition to any other issues for which they were seeking care.
Consider what these patients have experienced
A typical profile of these resilient patients with BPD: They were born emotionally sensitive. They grew up in homes with caretakers who knowingly or unknowingly invalidated their complaints about having their feelings hurt, about being abused emotionally, sexually, or otherwise, or about their worries concerning their interactions with peers at school. These caretakers may have been frightening and unpredictable, randomly showing affection or arbitrarily punishing for any perceived misstep, which led these patients to develop (for their own safety’s sake) a hypersensitivity to the affect of others. Their wariness and distrust of their social surroundings may have led to a skeptical view of kindness from others. Over time, without any guidance from prior demonstrations of healthy coping skills or interpersonal outlets from their caregivers, the emotional pressure builds. This pressure finally erupts in the form of impulsivity, self-harm, desperation, and defensiveness—in other words, survival. This is often followed by these patients’ first experience with receiving some degree of appropriate response to their complaints—their first experience with feeling seen and heard by their caretakers. They learn that their needs are met only when they cry out in desperation.1-3
These patients typically bring these maladaptive coping skills with them into adulthood, which often leads to a series of intense, unhealthy, and short-lived interpersonal and professional connections. They desire healthy, lasting connections with others, but through no fault of their own are unable to appropriately manage the normal stressors therein.1 Often, these patients do not know of their eventual BPD diagnosis, or even reject it due to its ever-negative valence. For other patients, receiving a personality disorder diagnosis is incredibly validating because they are no longer alone regarding this type of suffering, and a doctor—a caretaker—is finally making sense of this tumultuous world.
The countertransference of frustration, anxiety, doubt, and annoyance we may feel when caring for patients with BPD pales in comparison to living in their shoes and carrying the weight of what they have had to endure before presenting to our care. As these resilient patients wait in the exam room for the chance to be heard, let this be a reminder to greet them with the patience, understanding, empathy, and compassion that physicians are known to embody.
Suggestions for working with ‘difficult’ patients
The following tips may be helpful for building rapport with patients with BPD or other “difficult” patients:
- validate their complaints, and the difficulties they cause
- be genuine and honest when discussing their complaints
- acknowledge your own mistakes and misunderstandings in their care
- don’t be defensive—accept criticism with an open mind
- practice listening with intent, and reflective listening
- set ground rules and stick to them (eg, time limits, prescribing expectations, patient-physician relationship boundaries)
- educate and support the patient and their loved ones.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:947.
2. Porter C, Palmier-Claus J, Branitsky A, et al. Childhood adversity and borderline personality disorder: a meta-analysis. Acta Psychiatr Scand. 2020;141(1):6-20.
3. Sansone RA, Sansone LA. Emotional hyper-reactivity in borderline personality disorder. Psychiatry (Edgmont). 2010;7(9):16-20.
Oh, great!” a senior resident sardonically remarked with a smirk as they read up on the next patient in the clinic. “A borderline patient. Get ready for a rough one ... Ugh.”
Before ever stepping foot into the patient’s room, this resident had prematurely established and demonstrated an unfortunate dynamic for any student or trainee within earshot. This is an all-too-familiar occurrence when caring for individuals with borderline personality disorder (BPD), or any other patients deemed to be “difficult.” The patient, however, likely walked into the room with a traumatic past that they continue to suffer from, in addition to any other issues for which they were seeking care.
Consider what these patients have experienced
A typical profile of these resilient patients with BPD: They were born emotionally sensitive. They grew up in homes with caretakers who knowingly or unknowingly invalidated their complaints about having their feelings hurt, about being abused emotionally, sexually, or otherwise, or about their worries concerning their interactions with peers at school. These caretakers may have been frightening and unpredictable, randomly showing affection or arbitrarily punishing for any perceived misstep, which led these patients to develop (for their own safety’s sake) a hypersensitivity to the affect of others. Their wariness and distrust of their social surroundings may have led to a skeptical view of kindness from others. Over time, without any guidance from prior demonstrations of healthy coping skills or interpersonal outlets from their caregivers, the emotional pressure builds. This pressure finally erupts in the form of impulsivity, self-harm, desperation, and defensiveness—in other words, survival. This is often followed by these patients’ first experience with receiving some degree of appropriate response to their complaints—their first experience with feeling seen and heard by their caretakers. They learn that their needs are met only when they cry out in desperation.1-3
These patients typically bring these maladaptive coping skills with them into adulthood, which often leads to a series of intense, unhealthy, and short-lived interpersonal and professional connections. They desire healthy, lasting connections with others, but through no fault of their own are unable to appropriately manage the normal stressors therein.1 Often, these patients do not know of their eventual BPD diagnosis, or even reject it due to its ever-negative valence. For other patients, receiving a personality disorder diagnosis is incredibly validating because they are no longer alone regarding this type of suffering, and a doctor—a caretaker—is finally making sense of this tumultuous world.
The countertransference of frustration, anxiety, doubt, and annoyance we may feel when caring for patients with BPD pales in comparison to living in their shoes and carrying the weight of what they have had to endure before presenting to our care. As these resilient patients wait in the exam room for the chance to be heard, let this be a reminder to greet them with the patience, understanding, empathy, and compassion that physicians are known to embody.
Suggestions for working with ‘difficult’ patients
The following tips may be helpful for building rapport with patients with BPD or other “difficult” patients:
- validate their complaints, and the difficulties they cause
- be genuine and honest when discussing their complaints
- acknowledge your own mistakes and misunderstandings in their care
- don’t be defensive—accept criticism with an open mind
- practice listening with intent, and reflective listening
- set ground rules and stick to them (eg, time limits, prescribing expectations, patient-physician relationship boundaries)
- educate and support the patient and their loved ones.
Oh, great!” a senior resident sardonically remarked with a smirk as they read up on the next patient in the clinic. “A borderline patient. Get ready for a rough one ... Ugh.”
Before ever stepping foot into the patient’s room, this resident had prematurely established and demonstrated an unfortunate dynamic for any student or trainee within earshot. This is an all-too-familiar occurrence when caring for individuals with borderline personality disorder (BPD), or any other patients deemed to be “difficult.” The patient, however, likely walked into the room with a traumatic past that they continue to suffer from, in addition to any other issues for which they were seeking care.
Consider what these patients have experienced
A typical profile of these resilient patients with BPD: They were born emotionally sensitive. They grew up in homes with caretakers who knowingly or unknowingly invalidated their complaints about having their feelings hurt, about being abused emotionally, sexually, or otherwise, or about their worries concerning their interactions with peers at school. These caretakers may have been frightening and unpredictable, randomly showing affection or arbitrarily punishing for any perceived misstep, which led these patients to develop (for their own safety’s sake) a hypersensitivity to the affect of others. Their wariness and distrust of their social surroundings may have led to a skeptical view of kindness from others. Over time, without any guidance from prior demonstrations of healthy coping skills or interpersonal outlets from their caregivers, the emotional pressure builds. This pressure finally erupts in the form of impulsivity, self-harm, desperation, and defensiveness—in other words, survival. This is often followed by these patients’ first experience with receiving some degree of appropriate response to their complaints—their first experience with feeling seen and heard by their caretakers. They learn that their needs are met only when they cry out in desperation.1-3
These patients typically bring these maladaptive coping skills with them into adulthood, which often leads to a series of intense, unhealthy, and short-lived interpersonal and professional connections. They desire healthy, lasting connections with others, but through no fault of their own are unable to appropriately manage the normal stressors therein.1 Often, these patients do not know of their eventual BPD diagnosis, or even reject it due to its ever-negative valence. For other patients, receiving a personality disorder diagnosis is incredibly validating because they are no longer alone regarding this type of suffering, and a doctor—a caretaker—is finally making sense of this tumultuous world.
The countertransference of frustration, anxiety, doubt, and annoyance we may feel when caring for patients with BPD pales in comparison to living in their shoes and carrying the weight of what they have had to endure before presenting to our care. As these resilient patients wait in the exam room for the chance to be heard, let this be a reminder to greet them with the patience, understanding, empathy, and compassion that physicians are known to embody.
Suggestions for working with ‘difficult’ patients
The following tips may be helpful for building rapport with patients with BPD or other “difficult” patients:
- validate their complaints, and the difficulties they cause
- be genuine and honest when discussing their complaints
- acknowledge your own mistakes and misunderstandings in their care
- don’t be defensive—accept criticism with an open mind
- practice listening with intent, and reflective listening
- set ground rules and stick to them (eg, time limits, prescribing expectations, patient-physician relationship boundaries)
- educate and support the patient and their loved ones.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:947.
2. Porter C, Palmier-Claus J, Branitsky A, et al. Childhood adversity and borderline personality disorder: a meta-analysis. Acta Psychiatr Scand. 2020;141(1):6-20.
3. Sansone RA, Sansone LA. Emotional hyper-reactivity in borderline personality disorder. Psychiatry (Edgmont). 2010;7(9):16-20.
1. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. American Psychiatric Association; 2013:947.
2. Porter C, Palmier-Claus J, Branitsky A, et al. Childhood adversity and borderline personality disorder: a meta-analysis. Acta Psychiatr Scand. 2020;141(1):6-20.
3. Sansone RA, Sansone LA. Emotional hyper-reactivity in borderline personality disorder. Psychiatry (Edgmont). 2010;7(9):16-20.
Antibiotics use and vaccine antibody levels
In this column I have previously discussed the microbiome and its importance to health, especially as it relates to infections in children. Given the appreciated connection between microbiome and immunity, my group in Rochester, N.Y., recently undertook a study of the effect of antibiotic usage on the immune response to routine early childhood vaccines. In mouse models, it was previously shown that antibiotic exposure induced a reduction in the abundance and diversity of gut microbiota that in turn negatively affected the generation and maintenance of vaccine-induced immunity.1,2 A study from Stanford University was the first experimental human trial of antibiotic effects on vaccine responses. Adult volunteers were given an antibiotic or not before seasonal influenza vaccination and the researchers identified specific bacteria in the gut that were reduced by the antibiotics given. Those normal bacteria in the gut microbiome were shown to provide positive immunity signals to the systemic immune system that potentiated vaccine responses.3
My group conducted the first-ever study in children to explore whether an association existed between antibiotic use and vaccine-induced antibody levels. In the May issue of Pediatrics we report results from 560 children studied.4 From these children, 11,888 serum antibody levels to vaccine antigens were measured. Vaccine-induced antibody levels were determined at various time points after primary vaccination at child age 2, 4, and 6 months and boosters at age 12-18 months for 10 antigens included in four vaccines: DTaP, Hib, IPV, and PCV. The antibody levels to vaccine components were measured to DTaP (diphtheria toxoid, pertussis toxoid, tetanus toxoid, pertactin, and filamentous hemagglutinin), Hib conjugate (polyribosylribitol phosphate), IPV (polio 2), and PCV (serotypes 6B, 14, and 23F). A total of 342 children with 1,678 antibiotic courses prescribed were compared with 218 children with no antibiotic exposures. The predominant antibiotics prescribed were amoxicillin, cefdinir, amoxicillin/clavulanate, and ceftriaxone, since most treatments were for acute otitis media.
Of possible high clinical relevance, we found that from 9 to 24 months of age, children with antibiotic exposure had a higher frequency of vaccine-induced antibody levels below protection compared with children with no antibiotic use, placing them at risk of contracting a vaccine-preventable infection for DTaP antigens DT, TT, and PT and for PCV serotype 14.
For time points where antibody levels were determined within 30 days of completion of a course of antibiotics (recent antibiotic use), individual antibiotics were analyzed for effect on antibody levels below protective levels. Across all vaccine antigens measured, we found that all antibiotics had a negative effect on antibody levels and percentage of children achieving the protective antibody level threshold. Amoxicillin use had a lower association with lower antibody levels than the broader spectrum antibiotics, amoxicillin clavulanate (Augmentin), cefdinir, and ceftriaxone. For children receiving amoxicillin/clavulanate prescriptions, it was possible to compare the effect of shorter versus longer courses and we found that a 5-day course was associated with subprotective antibody levels similar to 10 days of amoxicillin, whereas 10-day amoxicillin/clavulanate was associated with higher frequency of children having subprotective antibody levels (Figure).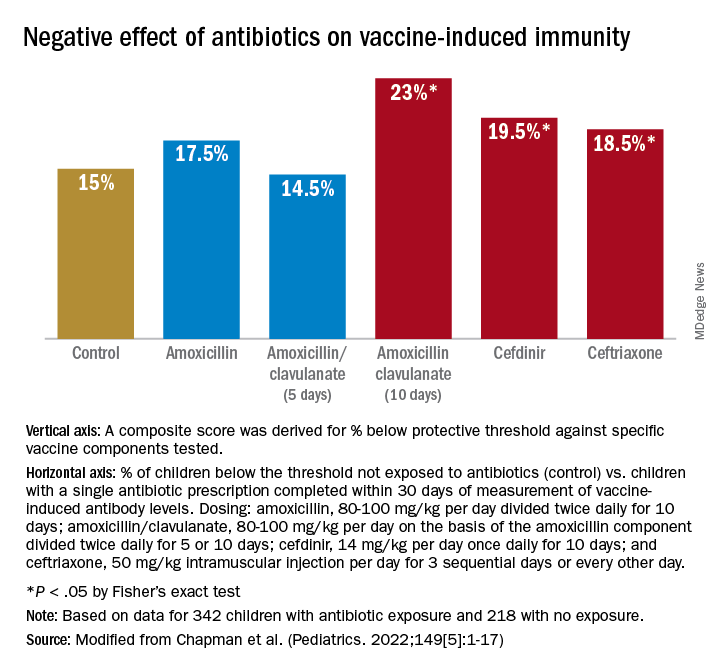
We examined whether accumulation of antibiotic courses in the first year of life had an association with subsequent vaccine-induced antibody levels and found that each antibiotic prescription was associated with a reduction in the median antibody level. For DTaP, each prescription was associated with 5.8% drop in antibody level to the vaccine components. For Hib the drop was 6.8%, IPV was 11.3%, and PCV was 10.4% – all statistically significant. To determine if booster vaccination influenced this association, a second analysis was performed using antibiotic prescriptions up to 15 months of age. We found each antibiotic prescription was associated with a reduction in median vaccine-induced antibody levels for DTaP by 18%, Hib by 21%, IPV by 19%, and PCV by 12% – all statistically significant.
Our study is the first in young children during the early age window where vaccine-induced immunity is established. Antibiotic use was associated with increased frequency of subprotective antibody levels for several vaccines used in children up to 2 years of age. The lower antibody levels could leave children vulnerable to vaccine preventable diseases. Perhaps outbreaks of vaccine-preventable diseases, such as pertussis, may be a consequence of multiple courses of antibiotics suppressing vaccine-induced immunity.
A goal of this study was to explore potential acute and long-term effects of antibiotic exposure on vaccine-induced antibody levels. Accumulated antibiotic courses up to booster immunization was associated with decreased vaccine antibody levels both before and after booster, suggesting that booster immunization was not sufficient to change the negative association with antibiotic exposure. The results were similar for all vaccines tested, suggesting that the specific vaccine formulation was not a factor.
The study has several limitations. The antibiotic prescription data and measurements of vaccine-induced antibody levels were recorded and measured prospectively; however, our analysis was done retrospectively. The group of study children was derived from my private practice in Rochester, N.Y., and may not be broadly representative of all children. The number of vaccine antibody measurements was limited by serum availability at some sampling time points in some children; and sometimes, the serum samples were collected far apart, which weakened our ability to perform longitudinal analyses. We did not collect stool samples from the children so we could not directly study the effect of antibiotic courses on the gut microbiome.
Our study adds new reasons to be cautious about overprescribing antibiotics on an individual child basis because an adverse effect extends to reduction in vaccine responses. This should be explained to parents requesting unnecessary antibiotics for colds and coughs. When antibiotics are necessary, the judicious choice of a narrow-spectrum antibiotic or a shorter duration of a broader spectrum antibiotic may reduce adverse effects on vaccine-induced immunity.
References
1. Valdez Y et al. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014;35(11):526-37.
2. Lynn MA et al. Early-life antibiotic-driven dysbiosis leads to dysregulated vaccine immune responses in mice. Cell Host Microbe. 2018;23(5):653-60.e5.
3. Hagan T et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178(6):1313-28.e13.
4. Chapman T et al. Antibiotic use and vaccine antibody levels. Pediatrics. 2022;149(5);1-17. doi: 10.1542/peds.2021-052061.
In this column I have previously discussed the microbiome and its importance to health, especially as it relates to infections in children. Given the appreciated connection between microbiome and immunity, my group in Rochester, N.Y., recently undertook a study of the effect of antibiotic usage on the immune response to routine early childhood vaccines. In mouse models, it was previously shown that antibiotic exposure induced a reduction in the abundance and diversity of gut microbiota that in turn negatively affected the generation and maintenance of vaccine-induced immunity.1,2 A study from Stanford University was the first experimental human trial of antibiotic effects on vaccine responses. Adult volunteers were given an antibiotic or not before seasonal influenza vaccination and the researchers identified specific bacteria in the gut that were reduced by the antibiotics given. Those normal bacteria in the gut microbiome were shown to provide positive immunity signals to the systemic immune system that potentiated vaccine responses.3
My group conducted the first-ever study in children to explore whether an association existed between antibiotic use and vaccine-induced antibody levels. In the May issue of Pediatrics we report results from 560 children studied.4 From these children, 11,888 serum antibody levels to vaccine antigens were measured. Vaccine-induced antibody levels were determined at various time points after primary vaccination at child age 2, 4, and 6 months and boosters at age 12-18 months for 10 antigens included in four vaccines: DTaP, Hib, IPV, and PCV. The antibody levels to vaccine components were measured to DTaP (diphtheria toxoid, pertussis toxoid, tetanus toxoid, pertactin, and filamentous hemagglutinin), Hib conjugate (polyribosylribitol phosphate), IPV (polio 2), and PCV (serotypes 6B, 14, and 23F). A total of 342 children with 1,678 antibiotic courses prescribed were compared with 218 children with no antibiotic exposures. The predominant antibiotics prescribed were amoxicillin, cefdinir, amoxicillin/clavulanate, and ceftriaxone, since most treatments were for acute otitis media.
Of possible high clinical relevance, we found that from 9 to 24 months of age, children with antibiotic exposure had a higher frequency of vaccine-induced antibody levels below protection compared with children with no antibiotic use, placing them at risk of contracting a vaccine-preventable infection for DTaP antigens DT, TT, and PT and for PCV serotype 14.
For time points where antibody levels were determined within 30 days of completion of a course of antibiotics (recent antibiotic use), individual antibiotics were analyzed for effect on antibody levels below protective levels. Across all vaccine antigens measured, we found that all antibiotics had a negative effect on antibody levels and percentage of children achieving the protective antibody level threshold. Amoxicillin use had a lower association with lower antibody levels than the broader spectrum antibiotics, amoxicillin clavulanate (Augmentin), cefdinir, and ceftriaxone. For children receiving amoxicillin/clavulanate prescriptions, it was possible to compare the effect of shorter versus longer courses and we found that a 5-day course was associated with subprotective antibody levels similar to 10 days of amoxicillin, whereas 10-day amoxicillin/clavulanate was associated with higher frequency of children having subprotective antibody levels (Figure).
We examined whether accumulation of antibiotic courses in the first year of life had an association with subsequent vaccine-induced antibody levels and found that each antibiotic prescription was associated with a reduction in the median antibody level. For DTaP, each prescription was associated with 5.8% drop in antibody level to the vaccine components. For Hib the drop was 6.8%, IPV was 11.3%, and PCV was 10.4% – all statistically significant. To determine if booster vaccination influenced this association, a second analysis was performed using antibiotic prescriptions up to 15 months of age. We found each antibiotic prescription was associated with a reduction in median vaccine-induced antibody levels for DTaP by 18%, Hib by 21%, IPV by 19%, and PCV by 12% – all statistically significant.
Our study is the first in young children during the early age window where vaccine-induced immunity is established. Antibiotic use was associated with increased frequency of subprotective antibody levels for several vaccines used in children up to 2 years of age. The lower antibody levels could leave children vulnerable to vaccine preventable diseases. Perhaps outbreaks of vaccine-preventable diseases, such as pertussis, may be a consequence of multiple courses of antibiotics suppressing vaccine-induced immunity.
A goal of this study was to explore potential acute and long-term effects of antibiotic exposure on vaccine-induced antibody levels. Accumulated antibiotic courses up to booster immunization was associated with decreased vaccine antibody levels both before and after booster, suggesting that booster immunization was not sufficient to change the negative association with antibiotic exposure. The results were similar for all vaccines tested, suggesting that the specific vaccine formulation was not a factor.
The study has several limitations. The antibiotic prescription data and measurements of vaccine-induced antibody levels were recorded and measured prospectively; however, our analysis was done retrospectively. The group of study children was derived from my private practice in Rochester, N.Y., and may not be broadly representative of all children. The number of vaccine antibody measurements was limited by serum availability at some sampling time points in some children; and sometimes, the serum samples were collected far apart, which weakened our ability to perform longitudinal analyses. We did not collect stool samples from the children so we could not directly study the effect of antibiotic courses on the gut microbiome.
Our study adds new reasons to be cautious about overprescribing antibiotics on an individual child basis because an adverse effect extends to reduction in vaccine responses. This should be explained to parents requesting unnecessary antibiotics for colds and coughs. When antibiotics are necessary, the judicious choice of a narrow-spectrum antibiotic or a shorter duration of a broader spectrum antibiotic may reduce adverse effects on vaccine-induced immunity.
References
1. Valdez Y et al. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014;35(11):526-37.
2. Lynn MA et al. Early-life antibiotic-driven dysbiosis leads to dysregulated vaccine immune responses in mice. Cell Host Microbe. 2018;23(5):653-60.e5.
3. Hagan T et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178(6):1313-28.e13.
4. Chapman T et al. Antibiotic use and vaccine antibody levels. Pediatrics. 2022;149(5);1-17. doi: 10.1542/peds.2021-052061.
In this column I have previously discussed the microbiome and its importance to health, especially as it relates to infections in children. Given the appreciated connection between microbiome and immunity, my group in Rochester, N.Y., recently undertook a study of the effect of antibiotic usage on the immune response to routine early childhood vaccines. In mouse models, it was previously shown that antibiotic exposure induced a reduction in the abundance and diversity of gut microbiota that in turn negatively affected the generation and maintenance of vaccine-induced immunity.1,2 A study from Stanford University was the first experimental human trial of antibiotic effects on vaccine responses. Adult volunteers were given an antibiotic or not before seasonal influenza vaccination and the researchers identified specific bacteria in the gut that were reduced by the antibiotics given. Those normal bacteria in the gut microbiome were shown to provide positive immunity signals to the systemic immune system that potentiated vaccine responses.3
My group conducted the first-ever study in children to explore whether an association existed between antibiotic use and vaccine-induced antibody levels. In the May issue of Pediatrics we report results from 560 children studied.4 From these children, 11,888 serum antibody levels to vaccine antigens were measured. Vaccine-induced antibody levels were determined at various time points after primary vaccination at child age 2, 4, and 6 months and boosters at age 12-18 months for 10 antigens included in four vaccines: DTaP, Hib, IPV, and PCV. The antibody levels to vaccine components were measured to DTaP (diphtheria toxoid, pertussis toxoid, tetanus toxoid, pertactin, and filamentous hemagglutinin), Hib conjugate (polyribosylribitol phosphate), IPV (polio 2), and PCV (serotypes 6B, 14, and 23F). A total of 342 children with 1,678 antibiotic courses prescribed were compared with 218 children with no antibiotic exposures. The predominant antibiotics prescribed were amoxicillin, cefdinir, amoxicillin/clavulanate, and ceftriaxone, since most treatments were for acute otitis media.
Of possible high clinical relevance, we found that from 9 to 24 months of age, children with antibiotic exposure had a higher frequency of vaccine-induced antibody levels below protection compared with children with no antibiotic use, placing them at risk of contracting a vaccine-preventable infection for DTaP antigens DT, TT, and PT and for PCV serotype 14.
For time points where antibody levels were determined within 30 days of completion of a course of antibiotics (recent antibiotic use), individual antibiotics were analyzed for effect on antibody levels below protective levels. Across all vaccine antigens measured, we found that all antibiotics had a negative effect on antibody levels and percentage of children achieving the protective antibody level threshold. Amoxicillin use had a lower association with lower antibody levels than the broader spectrum antibiotics, amoxicillin clavulanate (Augmentin), cefdinir, and ceftriaxone. For children receiving amoxicillin/clavulanate prescriptions, it was possible to compare the effect of shorter versus longer courses and we found that a 5-day course was associated with subprotective antibody levels similar to 10 days of amoxicillin, whereas 10-day amoxicillin/clavulanate was associated with higher frequency of children having subprotective antibody levels (Figure).
We examined whether accumulation of antibiotic courses in the first year of life had an association with subsequent vaccine-induced antibody levels and found that each antibiotic prescription was associated with a reduction in the median antibody level. For DTaP, each prescription was associated with 5.8% drop in antibody level to the vaccine components. For Hib the drop was 6.8%, IPV was 11.3%, and PCV was 10.4% – all statistically significant. To determine if booster vaccination influenced this association, a second analysis was performed using antibiotic prescriptions up to 15 months of age. We found each antibiotic prescription was associated with a reduction in median vaccine-induced antibody levels for DTaP by 18%, Hib by 21%, IPV by 19%, and PCV by 12% – all statistically significant.
Our study is the first in young children during the early age window where vaccine-induced immunity is established. Antibiotic use was associated with increased frequency of subprotective antibody levels for several vaccines used in children up to 2 years of age. The lower antibody levels could leave children vulnerable to vaccine preventable diseases. Perhaps outbreaks of vaccine-preventable diseases, such as pertussis, may be a consequence of multiple courses of antibiotics suppressing vaccine-induced immunity.
A goal of this study was to explore potential acute and long-term effects of antibiotic exposure on vaccine-induced antibody levels. Accumulated antibiotic courses up to booster immunization was associated with decreased vaccine antibody levels both before and after booster, suggesting that booster immunization was not sufficient to change the negative association with antibiotic exposure. The results were similar for all vaccines tested, suggesting that the specific vaccine formulation was not a factor.
The study has several limitations. The antibiotic prescription data and measurements of vaccine-induced antibody levels were recorded and measured prospectively; however, our analysis was done retrospectively. The group of study children was derived from my private practice in Rochester, N.Y., and may not be broadly representative of all children. The number of vaccine antibody measurements was limited by serum availability at some sampling time points in some children; and sometimes, the serum samples were collected far apart, which weakened our ability to perform longitudinal analyses. We did not collect stool samples from the children so we could not directly study the effect of antibiotic courses on the gut microbiome.
Our study adds new reasons to be cautious about overprescribing antibiotics on an individual child basis because an adverse effect extends to reduction in vaccine responses. This should be explained to parents requesting unnecessary antibiotics for colds and coughs. When antibiotics are necessary, the judicious choice of a narrow-spectrum antibiotic or a shorter duration of a broader spectrum antibiotic may reduce adverse effects on vaccine-induced immunity.
References
1. Valdez Y et al. Influence of the microbiota on vaccine effectiveness. Trends Immunol. 2014;35(11):526-37.
2. Lynn MA et al. Early-life antibiotic-driven dysbiosis leads to dysregulated vaccine immune responses in mice. Cell Host Microbe. 2018;23(5):653-60.e5.
3. Hagan T et al. Antibiotics-driven gut microbiome perturbation alters immunity to vaccines in humans. Cell. 2019;178(6):1313-28.e13.
4. Chapman T et al. Antibiotic use and vaccine antibody levels. Pediatrics. 2022;149(5);1-17. doi: 10.1542/peds.2021-052061.
San Diego, here we come
I attended my inaugural Digestive Disease Week® (DDW) in Orlando in 2013 as a first-year fellow, both excited and somewhat intimidated to be giving my first oral abstract presentation on an international stage. At that time, the only familiar faces at the conference were my co-fellows and faculty, along with a few welcoming faces from the fellowship interview trail. My attendings, on the other hand, couldn’t walk more than 100 feet in the conference hall without bumping into a smiling colleague! Now nearly a decade later, I am pleased to say that there are many more familiar faces in the crowd as I walk the halls of DDW. Each May, I look forward to meeting up with friends and colleagues across the country and the world while learning from an outstanding group of GI thought-leaders. While the COVID pandemic has disrupted this annual tradition in recent years, for the first time since 2019 those who feel comfortable will convene in San Diego to see old colleagues and meet new ones, learn about exciting, practice-changing innovations in our field, and enjoy the California sunshine. For those who cannot travel, robust DDW virtual offerings are planned as well.
A quick look at the 2022 conference schedule reveals an astonishing 3,300 oral abstract and poster presentations, 400 original lectures, and a variety of professional networking events on the agenda. The conference weekend will open with the AGA Post-Graduate Course, which offers a great opportunity to efficiently brush up on your clinical knowledge, guided by leading experts in the field. Monday, May 23 will feature the AGA Presidential Plenary, with AGA Institute President John Inadomi, MD, AGAF, and an exciting lineup of speakers discussing how best to address health care disparities impacting our patients and outlining AGA’s recent efforts to promote diversity, equity, and inclusion in our field. While it is nearly impossible to attend all sessions of interest during this whirlwind 4-day conference, you can rely on GI & Hepatology News to bring you key conference highlights over the coming months to ensure you don’t miss a beat!
Megan A. Adams, MD, JD, MSc
I attended my inaugural Digestive Disease Week® (DDW) in Orlando in 2013 as a first-year fellow, both excited and somewhat intimidated to be giving my first oral abstract presentation on an international stage. At that time, the only familiar faces at the conference were my co-fellows and faculty, along with a few welcoming faces from the fellowship interview trail. My attendings, on the other hand, couldn’t walk more than 100 feet in the conference hall without bumping into a smiling colleague! Now nearly a decade later, I am pleased to say that there are many more familiar faces in the crowd as I walk the halls of DDW. Each May, I look forward to meeting up with friends and colleagues across the country and the world while learning from an outstanding group of GI thought-leaders. While the COVID pandemic has disrupted this annual tradition in recent years, for the first time since 2019 those who feel comfortable will convene in San Diego to see old colleagues and meet new ones, learn about exciting, practice-changing innovations in our field, and enjoy the California sunshine. For those who cannot travel, robust DDW virtual offerings are planned as well.
A quick look at the 2022 conference schedule reveals an astonishing 3,300 oral abstract and poster presentations, 400 original lectures, and a variety of professional networking events on the agenda. The conference weekend will open with the AGA Post-Graduate Course, which offers a great opportunity to efficiently brush up on your clinical knowledge, guided by leading experts in the field. Monday, May 23 will feature the AGA Presidential Plenary, with AGA Institute President John Inadomi, MD, AGAF, and an exciting lineup of speakers discussing how best to address health care disparities impacting our patients and outlining AGA’s recent efforts to promote diversity, equity, and inclusion in our field. While it is nearly impossible to attend all sessions of interest during this whirlwind 4-day conference, you can rely on GI & Hepatology News to bring you key conference highlights over the coming months to ensure you don’t miss a beat!
Megan A. Adams, MD, JD, MSc
I attended my inaugural Digestive Disease Week® (DDW) in Orlando in 2013 as a first-year fellow, both excited and somewhat intimidated to be giving my first oral abstract presentation on an international stage. At that time, the only familiar faces at the conference were my co-fellows and faculty, along with a few welcoming faces from the fellowship interview trail. My attendings, on the other hand, couldn’t walk more than 100 feet in the conference hall without bumping into a smiling colleague! Now nearly a decade later, I am pleased to say that there are many more familiar faces in the crowd as I walk the halls of DDW. Each May, I look forward to meeting up with friends and colleagues across the country and the world while learning from an outstanding group of GI thought-leaders. While the COVID pandemic has disrupted this annual tradition in recent years, for the first time since 2019 those who feel comfortable will convene in San Diego to see old colleagues and meet new ones, learn about exciting, practice-changing innovations in our field, and enjoy the California sunshine. For those who cannot travel, robust DDW virtual offerings are planned as well.
A quick look at the 2022 conference schedule reveals an astonishing 3,300 oral abstract and poster presentations, 400 original lectures, and a variety of professional networking events on the agenda. The conference weekend will open with the AGA Post-Graduate Course, which offers a great opportunity to efficiently brush up on your clinical knowledge, guided by leading experts in the field. Monday, May 23 will feature the AGA Presidential Plenary, with AGA Institute President John Inadomi, MD, AGAF, and an exciting lineup of speakers discussing how best to address health care disparities impacting our patients and outlining AGA’s recent efforts to promote diversity, equity, and inclusion in our field. While it is nearly impossible to attend all sessions of interest during this whirlwind 4-day conference, you can rely on GI & Hepatology News to bring you key conference highlights over the coming months to ensure you don’t miss a beat!
Megan A. Adams, MD, JD, MSc
It’s time to shame the fat shamers
Fat shaming doesn’t work. If it did, obesity as we know it wouldn’t exist because if the one thing society ensures isn’t lacking for people with obesity, it’s shame. We know that fat shaming doesn’t lead to weight loss and that it’s actually correlated with weight gain: More shame leads to more gain (Puhl and Suh; Sutin and Terracciano; Tomiyama et al).
Shaming and weight stigma have far more concerning associations than weight gain. People who report experiencing more weight stigma have an increased risk for depression, anxiety, low self-esteem, poor body image, substance abuse, suicidality, unhealthy eating behaviors, disordered eating, increased caloric intake, exercise avoidance, decreased exercise motivation potentially due to heightened cortisol reactivity, elevated C-reactive protein, and elevated blood pressure.
Meanwhile, people with obesity – likely in part owing to negative weight-biased experiences in health care – are reluctant to discuss weight with their health care providers and are less likely to seek care at all for any conditions. When care is sought, people with obesity are more likely to receive substandard treatment, including receiving fewer preventive health screenings, decreased health education, and decreased time spent in appointments.
Remember that obesity is not a conscious choice
A fact that is conveniently forgotten by those who are most prone to fat shaming is that obesity, like every chronic noncommunicable disease, isn’t a choice that is consciously made by patients.
And yes, though there are lifestyle means that might affect weight, there are lifestyle means that might affect all chronic diseases – yet obesity is the only one we seem to moralize about. It’s also worth noting that other chronic diseases’ lifestyle levers tend not to be governed by thousands of genes and dozens of hormones; those trying to “lifestyle” their way out of obesity are swimming against strong physiologic currents that influence our most seminally important survival drive: eating.
But forgetting about physiologic currents, there is also staggering privilege associated with intentional perpetual behavior change around food and fitness in the name of health.
Whereas medicine and the world are right and quick to embrace the fights against racism, sexism, and homophobia, the push to confront weight bias is far rarer, despite the fact that it’s been shown to be rampant among health care professionals.
Protecting the rights of people with obesity
Perhaps though, times are changing. Movements are popping up to protect the rights of people with obesity while combating hate.
Of note, Brazil seems to have embraced a campaign to fight gordofobia — the Portuguese term used to describe weight-based discrimination. For instance, laws are being passed to ensure appropriate seating is supplied in schools for children with obesity, an annual day was formalized to promote the rights of people with obesity, preferential seating is provided on subways for people with obesity, and fines have been levied against at least one comedian for making fat jokes on the grounds of the state’s duty to protect minorities.
We need to take this fight to medicine. Given the incredibly depressing prevalence of weight bias among trainees, medical schools and residency programs should ensure countering weight bias is not only part of the curriculum but that it’s explicitly examined. National medical licensing examinations should include weight bias as well.
Though we’re closer than ever before to widely effective treatment options for obesity, it’s likely to still be decades before pharmaceutical options to treat obesity are as effective, accepted, and encouraged as medications to treat hypertension, dyslipidemia, diabetes, and more are today.
If you’re curious about your own implicit weight biases, consider taking Harvard’s Implicit Association Test for Weight. You might also want to take a few moments and review the Strategies to Overcome and Prevent Obesity Alliances’ Weight Can’t Wait guide for advice on the management of obesity in primary care.
Treat patients with obesity the same as you would those with any chronic condition.
Also, consider your physical office space. Do you have chairs suitable for patients with obesity (wide base and with arms to help patients rise)? A scale that measures up to high weights that’s in a private location? Appropriately sized blood pressure cuffs?
If not,
Examples include the family doctor who hadn’t checked my patient’s blood pressure in over a decade because he couldn’t be bothered buying an appropriately sized blood pressure cuff. Or the fertility doctor who told one of my patients that perhaps her weight reflected God’s will that she does not have children.
Finally, if reading this article about treating people with obesity the same as you would patients with other chronic, noncommunicable, lifestyle responsive diseases made you angry, there’s a great chance that you’re part of the problem.
Dr. Freedhoff, is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of The Diet Fix: Why Diets Fail and How to Make Yours Work. He has disclosed the following: He served as a director, officer, partner, employee, adviser, consultant, or trustee for Bariatric Medical Institute and Constant Health; has received research grant from Novo Nordisk, and has publicly shared opinions via Weighty Matters and social media. A version of this article first appeared on Medscape.com.
Fat shaming doesn’t work. If it did, obesity as we know it wouldn’t exist because if the one thing society ensures isn’t lacking for people with obesity, it’s shame. We know that fat shaming doesn’t lead to weight loss and that it’s actually correlated with weight gain: More shame leads to more gain (Puhl and Suh; Sutin and Terracciano; Tomiyama et al).
Shaming and weight stigma have far more concerning associations than weight gain. People who report experiencing more weight stigma have an increased risk for depression, anxiety, low self-esteem, poor body image, substance abuse, suicidality, unhealthy eating behaviors, disordered eating, increased caloric intake, exercise avoidance, decreased exercise motivation potentially due to heightened cortisol reactivity, elevated C-reactive protein, and elevated blood pressure.
Meanwhile, people with obesity – likely in part owing to negative weight-biased experiences in health care – are reluctant to discuss weight with their health care providers and are less likely to seek care at all for any conditions. When care is sought, people with obesity are more likely to receive substandard treatment, including receiving fewer preventive health screenings, decreased health education, and decreased time spent in appointments.
Remember that obesity is not a conscious choice
A fact that is conveniently forgotten by those who are most prone to fat shaming is that obesity, like every chronic noncommunicable disease, isn’t a choice that is consciously made by patients.
And yes, though there are lifestyle means that might affect weight, there are lifestyle means that might affect all chronic diseases – yet obesity is the only one we seem to moralize about. It’s also worth noting that other chronic diseases’ lifestyle levers tend not to be governed by thousands of genes and dozens of hormones; those trying to “lifestyle” their way out of obesity are swimming against strong physiologic currents that influence our most seminally important survival drive: eating.
But forgetting about physiologic currents, there is also staggering privilege associated with intentional perpetual behavior change around food and fitness in the name of health.
Whereas medicine and the world are right and quick to embrace the fights against racism, sexism, and homophobia, the push to confront weight bias is far rarer, despite the fact that it’s been shown to be rampant among health care professionals.
Protecting the rights of people with obesity
Perhaps though, times are changing. Movements are popping up to protect the rights of people with obesity while combating hate.
Of note, Brazil seems to have embraced a campaign to fight gordofobia — the Portuguese term used to describe weight-based discrimination. For instance, laws are being passed to ensure appropriate seating is supplied in schools for children with obesity, an annual day was formalized to promote the rights of people with obesity, preferential seating is provided on subways for people with obesity, and fines have been levied against at least one comedian for making fat jokes on the grounds of the state’s duty to protect minorities.
We need to take this fight to medicine. Given the incredibly depressing prevalence of weight bias among trainees, medical schools and residency programs should ensure countering weight bias is not only part of the curriculum but that it’s explicitly examined. National medical licensing examinations should include weight bias as well.
Though we’re closer than ever before to widely effective treatment options for obesity, it’s likely to still be decades before pharmaceutical options to treat obesity are as effective, accepted, and encouraged as medications to treat hypertension, dyslipidemia, diabetes, and more are today.
If you’re curious about your own implicit weight biases, consider taking Harvard’s Implicit Association Test for Weight. You might also want to take a few moments and review the Strategies to Overcome and Prevent Obesity Alliances’ Weight Can’t Wait guide for advice on the management of obesity in primary care.
Treat patients with obesity the same as you would those with any chronic condition.
Also, consider your physical office space. Do you have chairs suitable for patients with obesity (wide base and with arms to help patients rise)? A scale that measures up to high weights that’s in a private location? Appropriately sized blood pressure cuffs?
If not,
Examples include the family doctor who hadn’t checked my patient’s blood pressure in over a decade because he couldn’t be bothered buying an appropriately sized blood pressure cuff. Or the fertility doctor who told one of my patients that perhaps her weight reflected God’s will that she does not have children.
Finally, if reading this article about treating people with obesity the same as you would patients with other chronic, noncommunicable, lifestyle responsive diseases made you angry, there’s a great chance that you’re part of the problem.
Dr. Freedhoff, is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of The Diet Fix: Why Diets Fail and How to Make Yours Work. He has disclosed the following: He served as a director, officer, partner, employee, adviser, consultant, or trustee for Bariatric Medical Institute and Constant Health; has received research grant from Novo Nordisk, and has publicly shared opinions via Weighty Matters and social media. A version of this article first appeared on Medscape.com.
Fat shaming doesn’t work. If it did, obesity as we know it wouldn’t exist because if the one thing society ensures isn’t lacking for people with obesity, it’s shame. We know that fat shaming doesn’t lead to weight loss and that it’s actually correlated with weight gain: More shame leads to more gain (Puhl and Suh; Sutin and Terracciano; Tomiyama et al).
Shaming and weight stigma have far more concerning associations than weight gain. People who report experiencing more weight stigma have an increased risk for depression, anxiety, low self-esteem, poor body image, substance abuse, suicidality, unhealthy eating behaviors, disordered eating, increased caloric intake, exercise avoidance, decreased exercise motivation potentially due to heightened cortisol reactivity, elevated C-reactive protein, and elevated blood pressure.
Meanwhile, people with obesity – likely in part owing to negative weight-biased experiences in health care – are reluctant to discuss weight with their health care providers and are less likely to seek care at all for any conditions. When care is sought, people with obesity are more likely to receive substandard treatment, including receiving fewer preventive health screenings, decreased health education, and decreased time spent in appointments.
Remember that obesity is not a conscious choice
A fact that is conveniently forgotten by those who are most prone to fat shaming is that obesity, like every chronic noncommunicable disease, isn’t a choice that is consciously made by patients.
And yes, though there are lifestyle means that might affect weight, there are lifestyle means that might affect all chronic diseases – yet obesity is the only one we seem to moralize about. It’s also worth noting that other chronic diseases’ lifestyle levers tend not to be governed by thousands of genes and dozens of hormones; those trying to “lifestyle” their way out of obesity are swimming against strong physiologic currents that influence our most seminally important survival drive: eating.
But forgetting about physiologic currents, there is also staggering privilege associated with intentional perpetual behavior change around food and fitness in the name of health.
Whereas medicine and the world are right and quick to embrace the fights against racism, sexism, and homophobia, the push to confront weight bias is far rarer, despite the fact that it’s been shown to be rampant among health care professionals.
Protecting the rights of people with obesity
Perhaps though, times are changing. Movements are popping up to protect the rights of people with obesity while combating hate.
Of note, Brazil seems to have embraced a campaign to fight gordofobia — the Portuguese term used to describe weight-based discrimination. For instance, laws are being passed to ensure appropriate seating is supplied in schools for children with obesity, an annual day was formalized to promote the rights of people with obesity, preferential seating is provided on subways for people with obesity, and fines have been levied against at least one comedian for making fat jokes on the grounds of the state’s duty to protect minorities.
We need to take this fight to medicine. Given the incredibly depressing prevalence of weight bias among trainees, medical schools and residency programs should ensure countering weight bias is not only part of the curriculum but that it’s explicitly examined. National medical licensing examinations should include weight bias as well.
Though we’re closer than ever before to widely effective treatment options for obesity, it’s likely to still be decades before pharmaceutical options to treat obesity are as effective, accepted, and encouraged as medications to treat hypertension, dyslipidemia, diabetes, and more are today.
If you’re curious about your own implicit weight biases, consider taking Harvard’s Implicit Association Test for Weight. You might also want to take a few moments and review the Strategies to Overcome and Prevent Obesity Alliances’ Weight Can’t Wait guide for advice on the management of obesity in primary care.
Treat patients with obesity the same as you would those with any chronic condition.
Also, consider your physical office space. Do you have chairs suitable for patients with obesity (wide base and with arms to help patients rise)? A scale that measures up to high weights that’s in a private location? Appropriately sized blood pressure cuffs?
If not,
Examples include the family doctor who hadn’t checked my patient’s blood pressure in over a decade because he couldn’t be bothered buying an appropriately sized blood pressure cuff. Or the fertility doctor who told one of my patients that perhaps her weight reflected God’s will that she does not have children.
Finally, if reading this article about treating people with obesity the same as you would patients with other chronic, noncommunicable, lifestyle responsive diseases made you angry, there’s a great chance that you’re part of the problem.
Dr. Freedhoff, is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of The Diet Fix: Why Diets Fail and How to Make Yours Work. He has disclosed the following: He served as a director, officer, partner, employee, adviser, consultant, or trustee for Bariatric Medical Institute and Constant Health; has received research grant from Novo Nordisk, and has publicly shared opinions via Weighty Matters and social media. A version of this article first appeared on Medscape.com.
Career pivots: A new perspective on psychiatry
Psychiatrists practice a field of medicine that relies on one’s clinical perspective to interpret observable behaviors originating from the brains of others. In this manner, psychiatry and photography are similar. And digital technology has changed them both.
In photography, there are many technical aspects for one to master when framing and capturing a shot. The length of exposure. The amount of light needed. The speed of the film, which is its sensitivity to light. The aperture that controls how much light falls on the film. The movement of the subject across the film during the exposure. Despite the fact that physical film has mostly yielded to electronic sensors over the past couple decades, these basic aspects of photography remain.
But perspective is the critical ingredient. This is what brings the greatest impact to photography. The composition, or the subject of the photograph and how its elements – foreground, background, shapes, patterns, texture, shadow, motion, leading lines, and focal points – are arranged. The most powerful way to improve the composition – more powerful than fancy camera bells and whistles – is to move. One step to the left or right, one step forward or back. Stand on your toes, or crouch to your knees. Pivot this way or that. A simple change in perspective dramatically changes the nature and the energy of the captured image.
In fact, many physicians are changing what they actually do for a living. Pivoting their clinical perspectives. And applying those perspectives to other areas. The latest catalyst fueling these career pivots, these changes in perspectives, has been the incredible global impact of the tiny little coronavirus known as SARS-CoV-2. The COVID-19 pandemic that began two years ago has disrupted the entire planet. The virus has caused us all to change our perspective, to see our world differently, and our place in it.
The virus has exposed defects in our health care delivery system. And physicians have necessarily reacted, injecting changes in what they do and how they do it. Many of these changes rely on digital technology, building upon the groundwork laid over the past couple of decades to convert our paper processes into electronic processes, and our manual work flows into digital work flows. This groundwork is no small thing, as it relies on conventions and standards, such as DICOM, LOINC, AES, CDA, UMLS, FHIR, ICD, NDC, USCDI, and SNOMED-CT. Establishing, maintaining, and evolving health care standards requires organized groups of people to come together to share their diverse perspectives. This is but one of many places where physicians are using their unique clinical perspective to share what they see with others.
This column will focus on these professional pivots that physicians make when they take a step to the left or right to change their perspective and share their viewpoints in different settings with diverse groups of people. Some of these pivots are small, while others are career changing. But the theme that knits them together is about taking what one has learned while helping others achieve better health, and using that perspective to make a difference.
Dr. Daviss is chief medical officer for Optum Maryland and immediate past president of the Maryland-DC Society of Addiction Medicine, and former medical director and senior medical advisor at SAMHSA. He is coauthor of the 2011 book, Shrink Rap: Three Psychiatrists Explain Their Work. Psychiatrists and other physicians may share their own experience with pivots they have made with Dr. Daviss via email (drdaviss@gmail.com) or Twitter (@HITshrink). The opinions expressed are solely those of the author and do not necessarily reflect those of his employer or organizations with which he is associated.
Psychiatrists practice a field of medicine that relies on one’s clinical perspective to interpret observable behaviors originating from the brains of others. In this manner, psychiatry and photography are similar. And digital technology has changed them both.
In photography, there are many technical aspects for one to master when framing and capturing a shot. The length of exposure. The amount of light needed. The speed of the film, which is its sensitivity to light. The aperture that controls how much light falls on the film. The movement of the subject across the film during the exposure. Despite the fact that physical film has mostly yielded to electronic sensors over the past couple decades, these basic aspects of photography remain.
But perspective is the critical ingredient. This is what brings the greatest impact to photography. The composition, or the subject of the photograph and how its elements – foreground, background, shapes, patterns, texture, shadow, motion, leading lines, and focal points – are arranged. The most powerful way to improve the composition – more powerful than fancy camera bells and whistles – is to move. One step to the left or right, one step forward or back. Stand on your toes, or crouch to your knees. Pivot this way or that. A simple change in perspective dramatically changes the nature and the energy of the captured image.
In fact, many physicians are changing what they actually do for a living. Pivoting their clinical perspectives. And applying those perspectives to other areas. The latest catalyst fueling these career pivots, these changes in perspectives, has been the incredible global impact of the tiny little coronavirus known as SARS-CoV-2. The COVID-19 pandemic that began two years ago has disrupted the entire planet. The virus has caused us all to change our perspective, to see our world differently, and our place in it.
The virus has exposed defects in our health care delivery system. And physicians have necessarily reacted, injecting changes in what they do and how they do it. Many of these changes rely on digital technology, building upon the groundwork laid over the past couple of decades to convert our paper processes into electronic processes, and our manual work flows into digital work flows. This groundwork is no small thing, as it relies on conventions and standards, such as DICOM, LOINC, AES, CDA, UMLS, FHIR, ICD, NDC, USCDI, and SNOMED-CT. Establishing, maintaining, and evolving health care standards requires organized groups of people to come together to share their diverse perspectives. This is but one of many places where physicians are using their unique clinical perspective to share what they see with others.
This column will focus on these professional pivots that physicians make when they take a step to the left or right to change their perspective and share their viewpoints in different settings with diverse groups of people. Some of these pivots are small, while others are career changing. But the theme that knits them together is about taking what one has learned while helping others achieve better health, and using that perspective to make a difference.
Dr. Daviss is chief medical officer for Optum Maryland and immediate past president of the Maryland-DC Society of Addiction Medicine, and former medical director and senior medical advisor at SAMHSA. He is coauthor of the 2011 book, Shrink Rap: Three Psychiatrists Explain Their Work. Psychiatrists and other physicians may share their own experience with pivots they have made with Dr. Daviss via email (drdaviss@gmail.com) or Twitter (@HITshrink). The opinions expressed are solely those of the author and do not necessarily reflect those of his employer or organizations with which he is associated.
Psychiatrists practice a field of medicine that relies on one’s clinical perspective to interpret observable behaviors originating from the brains of others. In this manner, psychiatry and photography are similar. And digital technology has changed them both.
In photography, there are many technical aspects for one to master when framing and capturing a shot. The length of exposure. The amount of light needed. The speed of the film, which is its sensitivity to light. The aperture that controls how much light falls on the film. The movement of the subject across the film during the exposure. Despite the fact that physical film has mostly yielded to electronic sensors over the past couple decades, these basic aspects of photography remain.
But perspective is the critical ingredient. This is what brings the greatest impact to photography. The composition, or the subject of the photograph and how its elements – foreground, background, shapes, patterns, texture, shadow, motion, leading lines, and focal points – are arranged. The most powerful way to improve the composition – more powerful than fancy camera bells and whistles – is to move. One step to the left or right, one step forward or back. Stand on your toes, or crouch to your knees. Pivot this way or that. A simple change in perspective dramatically changes the nature and the energy of the captured image.
In fact, many physicians are changing what they actually do for a living. Pivoting their clinical perspectives. And applying those perspectives to other areas. The latest catalyst fueling these career pivots, these changes in perspectives, has been the incredible global impact of the tiny little coronavirus known as SARS-CoV-2. The COVID-19 pandemic that began two years ago has disrupted the entire planet. The virus has caused us all to change our perspective, to see our world differently, and our place in it.
The virus has exposed defects in our health care delivery system. And physicians have necessarily reacted, injecting changes in what they do and how they do it. Many of these changes rely on digital technology, building upon the groundwork laid over the past couple of decades to convert our paper processes into electronic processes, and our manual work flows into digital work flows. This groundwork is no small thing, as it relies on conventions and standards, such as DICOM, LOINC, AES, CDA, UMLS, FHIR, ICD, NDC, USCDI, and SNOMED-CT. Establishing, maintaining, and evolving health care standards requires organized groups of people to come together to share their diverse perspectives. This is but one of many places where physicians are using their unique clinical perspective to share what they see with others.
This column will focus on these professional pivots that physicians make when they take a step to the left or right to change their perspective and share their viewpoints in different settings with diverse groups of people. Some of these pivots are small, while others are career changing. But the theme that knits them together is about taking what one has learned while helping others achieve better health, and using that perspective to make a difference.
Dr. Daviss is chief medical officer for Optum Maryland and immediate past president of the Maryland-DC Society of Addiction Medicine, and former medical director and senior medical advisor at SAMHSA. He is coauthor of the 2011 book, Shrink Rap: Three Psychiatrists Explain Their Work. Psychiatrists and other physicians may share their own experience with pivots they have made with Dr. Daviss via email (drdaviss@gmail.com) or Twitter (@HITshrink). The opinions expressed are solely those of the author and do not necessarily reflect those of his employer or organizations with which he is associated.
Screening for anxiety in young children
On April 12, 2022, the U.S. Preventive Services Task Force released the draft of a recommendation statement titled Screening for Anxiety in Children and Adolescents. Based on their observation that 7.8% of children and adolescents have a current anxiety disorder and their analysis of the magnitude of the net benefit, the Task Force plans on recommending that children ages 8-18 years be screened for the condition. However, the group could not find evidence to support screening for children 7 years and younger.
Over more than 4 decades of general pediatric practice, it became obvious to me that anxiety was driving a high percentage of my office visits. Most often in young children it was parental anxiety that was prompting the phone call or office visit. In older childhood and adolescence it was patient anxiety that began to play a larger role.
Over the last 2 decades the level of anxiety in all age groups has seemed to increase. How large a role the events of Sept. 11, 2001, and other terrorist attacks were playing in this phenomenon is unclear to me. However, I suspect they were significant. More recently the pandemic and the failure of both political parties to forge a working arrangement have fueled even more anxiety in many demographic segments. It may be safe to say that everyone is anxious to one degree or another.
Broad-based anxiety in the general population and the incidence of anxiety disorders severe enough to disrupt a child’s life are certainly two different kettles of fish. However, the factors that have raised the level of anxiety across all age groups certainly hasn’t made things any easier for the child who has inherited or developed an anxiety disorder.
Glancing at the 600-page evidence synthesis that accompanies the task force’s report it is clear that they have taken their challenge seriously. However, I wonder whether looking at the 7-and-under age group with a different lens might have resulted in the inclusion of younger children in their recommendation.
I understand that to support their recommendations the U.S. Preventive Services Task Forces must rely on data from peer-reviewed studies that have looked at quantifiable outcomes. However, I suspect the task force would agree that its recommendations shouldn’t prevent the rest of us from using our own observations and intuition when deciding whether to selectively screen our younger patients for anxiety disorders.
Although it may not generate a measurable data point, providing the parents of a 5-year-old whose troubling behavior is in part the result of an anxiety disorder is invaluable. Do we need to screen all 5-year-olds? The task force says probably not given the current state of our knowledge and I agree. But, the fact that almost 8% of the pediatric population carries the diagnosis and my anecdotal observations suggest that as pediatricians we should be learning more about anxiety disorders and their wide variety of presentations. Then we should selectively screen more of our patients. In fact, I suspect we might help our patients and ourselves by questioning more parents about their own mental health histories even before we have any inkling that their child has a problem. While the degree to which anxiety disorders are inheritable and the exact mechanism is far from clear, I think this history might be a valuable piece of information to learn as early as the prenatal get-acquainted visit. A simple question to a new or expecting parent about what worries them most about becoming a parent would be a good opener. Your reassurance that you expect parents to be worried and welcome hearing about their concerns should be a step in building a strong foundation for a family-provider relationship.
Anxiety happens and unfortunately so do anxiety disorders. We need to be doing a better job of acknowledging and responding to these two realities.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
*This column was updated on 5/4/2022.
On April 12, 2022, the U.S. Preventive Services Task Force released the draft of a recommendation statement titled Screening for Anxiety in Children and Adolescents. Based on their observation that 7.8% of children and adolescents have a current anxiety disorder and their analysis of the magnitude of the net benefit, the Task Force plans on recommending that children ages 8-18 years be screened for the condition. However, the group could not find evidence to support screening for children 7 years and younger.
Over more than 4 decades of general pediatric practice, it became obvious to me that anxiety was driving a high percentage of my office visits. Most often in young children it was parental anxiety that was prompting the phone call or office visit. In older childhood and adolescence it was patient anxiety that began to play a larger role.
Over the last 2 decades the level of anxiety in all age groups has seemed to increase. How large a role the events of Sept. 11, 2001, and other terrorist attacks were playing in this phenomenon is unclear to me. However, I suspect they were significant. More recently the pandemic and the failure of both political parties to forge a working arrangement have fueled even more anxiety in many demographic segments. It may be safe to say that everyone is anxious to one degree or another.
Broad-based anxiety in the general population and the incidence of anxiety disorders severe enough to disrupt a child’s life are certainly two different kettles of fish. However, the factors that have raised the level of anxiety across all age groups certainly hasn’t made things any easier for the child who has inherited or developed an anxiety disorder.
Glancing at the 600-page evidence synthesis that accompanies the task force’s report it is clear that they have taken their challenge seriously. However, I wonder whether looking at the 7-and-under age group with a different lens might have resulted in the inclusion of younger children in their recommendation.
I understand that to support their recommendations the U.S. Preventive Services Task Forces must rely on data from peer-reviewed studies that have looked at quantifiable outcomes. However, I suspect the task force would agree that its recommendations shouldn’t prevent the rest of us from using our own observations and intuition when deciding whether to selectively screen our younger patients for anxiety disorders.
Although it may not generate a measurable data point, providing the parents of a 5-year-old whose troubling behavior is in part the result of an anxiety disorder is invaluable. Do we need to screen all 5-year-olds? The task force says probably not given the current state of our knowledge and I agree. But, the fact that almost 8% of the pediatric population carries the diagnosis and my anecdotal observations suggest that as pediatricians we should be learning more about anxiety disorders and their wide variety of presentations. Then we should selectively screen more of our patients. In fact, I suspect we might help our patients and ourselves by questioning more parents about their own mental health histories even before we have any inkling that their child has a problem. While the degree to which anxiety disorders are inheritable and the exact mechanism is far from clear, I think this history might be a valuable piece of information to learn as early as the prenatal get-acquainted visit. A simple question to a new or expecting parent about what worries them most about becoming a parent would be a good opener. Your reassurance that you expect parents to be worried and welcome hearing about their concerns should be a step in building a strong foundation for a family-provider relationship.
Anxiety happens and unfortunately so do anxiety disorders. We need to be doing a better job of acknowledging and responding to these two realities.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
*This column was updated on 5/4/2022.
On April 12, 2022, the U.S. Preventive Services Task Force released the draft of a recommendation statement titled Screening for Anxiety in Children and Adolescents. Based on their observation that 7.8% of children and adolescents have a current anxiety disorder and their analysis of the magnitude of the net benefit, the Task Force plans on recommending that children ages 8-18 years be screened for the condition. However, the group could not find evidence to support screening for children 7 years and younger.
Over more than 4 decades of general pediatric practice, it became obvious to me that anxiety was driving a high percentage of my office visits. Most often in young children it was parental anxiety that was prompting the phone call or office visit. In older childhood and adolescence it was patient anxiety that began to play a larger role.
Over the last 2 decades the level of anxiety in all age groups has seemed to increase. How large a role the events of Sept. 11, 2001, and other terrorist attacks were playing in this phenomenon is unclear to me. However, I suspect they were significant. More recently the pandemic and the failure of both political parties to forge a working arrangement have fueled even more anxiety in many demographic segments. It may be safe to say that everyone is anxious to one degree or another.
Broad-based anxiety in the general population and the incidence of anxiety disorders severe enough to disrupt a child’s life are certainly two different kettles of fish. However, the factors that have raised the level of anxiety across all age groups certainly hasn’t made things any easier for the child who has inherited or developed an anxiety disorder.
Glancing at the 600-page evidence synthesis that accompanies the task force’s report it is clear that they have taken their challenge seriously. However, I wonder whether looking at the 7-and-under age group with a different lens might have resulted in the inclusion of younger children in their recommendation.
I understand that to support their recommendations the U.S. Preventive Services Task Forces must rely on data from peer-reviewed studies that have looked at quantifiable outcomes. However, I suspect the task force would agree that its recommendations shouldn’t prevent the rest of us from using our own observations and intuition when deciding whether to selectively screen our younger patients for anxiety disorders.
Although it may not generate a measurable data point, providing the parents of a 5-year-old whose troubling behavior is in part the result of an anxiety disorder is invaluable. Do we need to screen all 5-year-olds? The task force says probably not given the current state of our knowledge and I agree. But, the fact that almost 8% of the pediatric population carries the diagnosis and my anecdotal observations suggest that as pediatricians we should be learning more about anxiety disorders and their wide variety of presentations. Then we should selectively screen more of our patients. In fact, I suspect we might help our patients and ourselves by questioning more parents about their own mental health histories even before we have any inkling that their child has a problem. While the degree to which anxiety disorders are inheritable and the exact mechanism is far from clear, I think this history might be a valuable piece of information to learn as early as the prenatal get-acquainted visit. A simple question to a new or expecting parent about what worries them most about becoming a parent would be a good opener. Your reassurance that you expect parents to be worried and welcome hearing about their concerns should be a step in building a strong foundation for a family-provider relationship.
Anxiety happens and unfortunately so do anxiety disorders. We need to be doing a better job of acknowledging and responding to these two realities.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at pdnews@mdedge.com.
*This column was updated on 5/4/2022.
Smartphone diagnosis in infant seizures could be highly effective
This video transcript has been edited for clarity.
Andrew N. Wilner, MD: Welcome to Medscape. I’m Dr Andrew Wilner, reporting from the American Epilepsy Society meeting.
Today, I have the pleasure of speaking with Dr. Chethan Rao, a child and adolescent neurology resident from the Mayo Clinic in Jacksonville, Fla. Dr. Rao has a particular interest in pediatric epilepsy. Welcome, Dr. Rao.
Chethan Rao, DO: Thank you, Dr. Wilner. It’s a pleasure to be here, and thanks for taking the time to highlight our work.
Dr. Wilner: You had a very interesting paper at the meeting that I wanted to talk about, focused on infantile spasms and smartphone video. Before we dive into the paper, tell us: What are infantile spasms, and why is it important to diagnose them early?
Dr. Rao: Infantile spasms, also known as epileptic spasms, are 1- to 2-second seizures, and they typically consist of sudden stiffening of the body with brief bending forward or backward of the arms, legs, and head. They usually happen around age 3-8 months, and they typically occur in clusters, most often after awakening from sleep.
The incidence is about 1 in 2,000-3,000 children. Many kids with spasms go on to develop seizures that are very difficult to treat, like Lennox-Gastaut epilepsy, and many go on to have developmental delays as well.
Dr. Wilner: Are these subtle? In other words, could a parent have a child like that and not really recognize that this is something abnormal? Or are they so dramatic that parents say: “We’re going to the emergency room?”
Dr. Rao: One of the problems that we encounter often is that in this age group of infants, they have benign sleep myoclonus; they have Sandifer syndrome related to reflux. Those can be very difficult mimics of spasms. They’re not the most clear-cut, but they look usually different enough from normal baby movements that they get parents to seek medical attention.
Dr. Wilner: You mentioned that the infantile spasms really are a type of epilepsy and symptomatic, usually, of some underlying neurologic condition. Why is it so important to diagnose them early?
Dr. Rao: Great question. Many studies have looked at developmental outcomes based on when spasms were diagnosed and treated, and all of them have replicated time over time that the earlier you get to treatment for the spasms, the better the outcomes are for seizure control and for development.
For this reason, infantile spasm is considered a neurologic urgency in our world. Like I said, accurate diagnosis is often complicated by these potential mimics. Prompt EEG is one of the most important things for confirmation of diagnosis.
Dr. Wilner: But to get that EEG, it has to get all the way to the neurologist, right? It’s not something they’re going to do in the ER. I saw a statistic: There are millions, if not billions, of smartphones out there. Where does the smartphone come in?
Dr. Rao: Absolutely. One of the things that we have on our side these days is that almost everyone has a smartphone at their disposal. One of the recent polls in 2021 showed that more than 95% of adults of childbearing age have smartphones with video access. As some other studies have shown in the adult world, we all really have an epilepsy monitoring unit minus the EEG in our own pockets.
It’s definitely a useful tool, as that first screening video can be used in adjunct to history and physical. There have been many of studies on the adult epilepsy side showing the predictive value of smartphone video for differentiating things like epileptic seizures and nonepileptic spells. What we wanted to do is use smartphone video to pin the diagnosis early of infantile spasms and get it treated as quickly as possible.
Dr. Wilner: I’m a fan. Every now and then, I do have a patient who brings in a video of some spell. I’m an adult neurologist. The patient had a spell, and you ask them – of course they don’t remember – and you ask the witness, who usually is not a trained observer. There have been one or two occasions where I thought: “Well, I don’t know if that was really a seizure.” Then they show me the video and it’s like, “Wow, that is definitely a convulsion.” A picture definitely can be worth a thousand words.
You studied this systematically for your poster. Tell me about what you did.
Dr. Rao: Since the poster, we’ve actually expanded the study, so I’ll give you the updated version. We looked at 101 infants retrospectively at two large children’s health care centers: Nemours Children’s, associated with Mayo Clinic in Jacksonville, Fla., and Texas Children’s Hospital in Houston. We narrowed it down to 80 patients whom we included. Of these, 43 had smartphone video capture when they first presented and 37 had no video when they first presented.
We found a 17-day difference by median in the time to diagnosis and treatment. In other words, the video group was diagnosed and treated 17 days by median, compared with the no-video group. Although 17 days may not sound like a big number, in this context it can make a huge difference. That’s been shown by one of these key studies in our field called the UK Infantile Spasms Study. The 2-week difference made about a 10-point difference on the developmental scale that they use – so pretty significant.
Dr. Wilner: Let me think about this for a minute. Was that because the parents brought the child in with their video and the doctor said, “Hey, that’s infantile spasms. Here’s your shot of ACTH [or whatever they’re using these days].” Or was it because the parents who were attentive enough to use video brought their kids in sooner?
Or was this the time from when they brought the child in to treatment? Is that the time you looked at? So it wasn’t just that these were more attentive parents and more likely to use the video – you’re looking at the time from presentation with or without video until treatment, is that right?
Dr. Rao: We looked to the time from the start of the spasms, as reported by the parents, to the time of diagnosis and then the start of spasms to the time of treatment. What you asked was a fantastic question. We wanted to know who these parents are who are taking videos versus the ones that are not.
We looked at the race/ethnicity data and socioeconomic status data. There were no significant differences between the video and nonvideo group. That would not explain the difference in our results here.
Dr. Wilner: Do you have plans to follow these approximately 40 children 5 years from now and see who’s riding a bicycle and who’s still stuck in the stroller? Is there going to be a difference?
Dr. Rao: Because time to diagnosis and time to treatment were our primary outcomes, long-term follow-up may not really help as much in this study. We did have a couple of other ideas for future studies. One that we wanted to look at was kids who have risk factors for developing spasms, such as trisomy 21, tuberous sclerosis, and congenital cortical malformations; those kids are at a much higher risk for developing spasms around 3-8 months of life.
In giving targeted counseling to those families about how they can use smartphone video to minimize the time to diagnosis and treatment, we think we may be able to learn more and maybe do that prospectively.
The other interesting idea is using artificial intelligence technology for spasm detection in some of these smartphone videos. They’re already using it for different seizure types. It could be an efficient first pass when we get a whole bunch of smartphone videos to determine which ones we need to pursue further steps – to see whether we need to get long-term EEG monitoring or not.
Dr. Wilner: As an epileptologist, I was going to say that we have smartphone EKG. All we need now is smartphone EEG, and then you’ll have all the information you need on day one. It may be a ways away.
As a bottom line, would it be fair to say that parents should not hesitate to take a video of any suspiciously abnormal behavior and bring it to their family doctor or pediatric neurologist?
Dr. Rao: Yes. I was happy to see the Tuberous Sclerosis Alliance put out a promotional video that had some steps for when parents see things that are suspicious for spasms, and they do recommend using smartphone video and promptly showing it to their doctors. I think the difference that we hope to provide in this study is that we can now quantify the effect of having that smartphone video when they first present.
My takeaway from this study that I would like to show is encouraging the use of smartphone video as an adjunct tool and for providers to ask for the videos, but also for these pediatric centers to develop an infrastructure – either a secure, monitored email address like we have at our center or a patient portal – where parents can submit video concerning for spasms.
Dr. Wilner: Save the trip to the doctor. Get that video out there first.
Dr. Rao: Especially in the pandemic world, right?
Dr. Wilner: Yes. I understand that you are a neurology resident. To wrap up, what’s the next step for you?
Dr. Rao: I’m finishing up my child neurology residency this year, and I’m moving out to Stanford for pediatric epilepsy fellowship. We’re preparing this project we’re talking about for submission soon, and we’re working on another project, which is a systematic review of genetic testing and the presurgical workup for pediatric drug-resistant focal epilepsy.
Dr. Wilner: Excellent. That’s pretty exciting. Good luck to you. I want to thank you very much for telling us about your research.
Dr. Rao: It was a pleasure speaking with you, and I look forward to the next time.
Dr. Wilner: I’m Dr Andrew Wilner, reporting for Medscape. Thanks for watching.
A version of this article first appeared on Medscape.com.
This video transcript has been edited for clarity.
Andrew N. Wilner, MD: Welcome to Medscape. I’m Dr Andrew Wilner, reporting from the American Epilepsy Society meeting.
Today, I have the pleasure of speaking with Dr. Chethan Rao, a child and adolescent neurology resident from the Mayo Clinic in Jacksonville, Fla. Dr. Rao has a particular interest in pediatric epilepsy. Welcome, Dr. Rao.
Chethan Rao, DO: Thank you, Dr. Wilner. It’s a pleasure to be here, and thanks for taking the time to highlight our work.
Dr. Wilner: You had a very interesting paper at the meeting that I wanted to talk about, focused on infantile spasms and smartphone video. Before we dive into the paper, tell us: What are infantile spasms, and why is it important to diagnose them early?
Dr. Rao: Infantile spasms, also known as epileptic spasms, are 1- to 2-second seizures, and they typically consist of sudden stiffening of the body with brief bending forward or backward of the arms, legs, and head. They usually happen around age 3-8 months, and they typically occur in clusters, most often after awakening from sleep.
The incidence is about 1 in 2,000-3,000 children. Many kids with spasms go on to develop seizures that are very difficult to treat, like Lennox-Gastaut epilepsy, and many go on to have developmental delays as well.
Dr. Wilner: Are these subtle? In other words, could a parent have a child like that and not really recognize that this is something abnormal? Or are they so dramatic that parents say: “We’re going to the emergency room?”
Dr. Rao: One of the problems that we encounter often is that in this age group of infants, they have benign sleep myoclonus; they have Sandifer syndrome related to reflux. Those can be very difficult mimics of spasms. They’re not the most clear-cut, but they look usually different enough from normal baby movements that they get parents to seek medical attention.
Dr. Wilner: You mentioned that the infantile spasms really are a type of epilepsy and symptomatic, usually, of some underlying neurologic condition. Why is it so important to diagnose them early?
Dr. Rao: Great question. Many studies have looked at developmental outcomes based on when spasms were diagnosed and treated, and all of them have replicated time over time that the earlier you get to treatment for the spasms, the better the outcomes are for seizure control and for development.
For this reason, infantile spasm is considered a neurologic urgency in our world. Like I said, accurate diagnosis is often complicated by these potential mimics. Prompt EEG is one of the most important things for confirmation of diagnosis.
Dr. Wilner: But to get that EEG, it has to get all the way to the neurologist, right? It’s not something they’re going to do in the ER. I saw a statistic: There are millions, if not billions, of smartphones out there. Where does the smartphone come in?
Dr. Rao: Absolutely. One of the things that we have on our side these days is that almost everyone has a smartphone at their disposal. One of the recent polls in 2021 showed that more than 95% of adults of childbearing age have smartphones with video access. As some other studies have shown in the adult world, we all really have an epilepsy monitoring unit minus the EEG in our own pockets.
It’s definitely a useful tool, as that first screening video can be used in adjunct to history and physical. There have been many of studies on the adult epilepsy side showing the predictive value of smartphone video for differentiating things like epileptic seizures and nonepileptic spells. What we wanted to do is use smartphone video to pin the diagnosis early of infantile spasms and get it treated as quickly as possible.
Dr. Wilner: I’m a fan. Every now and then, I do have a patient who brings in a video of some spell. I’m an adult neurologist. The patient had a spell, and you ask them – of course they don’t remember – and you ask the witness, who usually is not a trained observer. There have been one or two occasions where I thought: “Well, I don’t know if that was really a seizure.” Then they show me the video and it’s like, “Wow, that is definitely a convulsion.” A picture definitely can be worth a thousand words.
You studied this systematically for your poster. Tell me about what you did.
Dr. Rao: Since the poster, we’ve actually expanded the study, so I’ll give you the updated version. We looked at 101 infants retrospectively at two large children’s health care centers: Nemours Children’s, associated with Mayo Clinic in Jacksonville, Fla., and Texas Children’s Hospital in Houston. We narrowed it down to 80 patients whom we included. Of these, 43 had smartphone video capture when they first presented and 37 had no video when they first presented.
We found a 17-day difference by median in the time to diagnosis and treatment. In other words, the video group was diagnosed and treated 17 days by median, compared with the no-video group. Although 17 days may not sound like a big number, in this context it can make a huge difference. That’s been shown by one of these key studies in our field called the UK Infantile Spasms Study. The 2-week difference made about a 10-point difference on the developmental scale that they use – so pretty significant.
Dr. Wilner: Let me think about this for a minute. Was that because the parents brought the child in with their video and the doctor said, “Hey, that’s infantile spasms. Here’s your shot of ACTH [or whatever they’re using these days].” Or was it because the parents who were attentive enough to use video brought their kids in sooner?
Or was this the time from when they brought the child in to treatment? Is that the time you looked at? So it wasn’t just that these were more attentive parents and more likely to use the video – you’re looking at the time from presentation with or without video until treatment, is that right?
Dr. Rao: We looked to the time from the start of the spasms, as reported by the parents, to the time of diagnosis and then the start of spasms to the time of treatment. What you asked was a fantastic question. We wanted to know who these parents are who are taking videos versus the ones that are not.
We looked at the race/ethnicity data and socioeconomic status data. There were no significant differences between the video and nonvideo group. That would not explain the difference in our results here.
Dr. Wilner: Do you have plans to follow these approximately 40 children 5 years from now and see who’s riding a bicycle and who’s still stuck in the stroller? Is there going to be a difference?
Dr. Rao: Because time to diagnosis and time to treatment were our primary outcomes, long-term follow-up may not really help as much in this study. We did have a couple of other ideas for future studies. One that we wanted to look at was kids who have risk factors for developing spasms, such as trisomy 21, tuberous sclerosis, and congenital cortical malformations; those kids are at a much higher risk for developing spasms around 3-8 months of life.
In giving targeted counseling to those families about how they can use smartphone video to minimize the time to diagnosis and treatment, we think we may be able to learn more and maybe do that prospectively.
The other interesting idea is using artificial intelligence technology for spasm detection in some of these smartphone videos. They’re already using it for different seizure types. It could be an efficient first pass when we get a whole bunch of smartphone videos to determine which ones we need to pursue further steps – to see whether we need to get long-term EEG monitoring or not.
Dr. Wilner: As an epileptologist, I was going to say that we have smartphone EKG. All we need now is smartphone EEG, and then you’ll have all the information you need on day one. It may be a ways away.
As a bottom line, would it be fair to say that parents should not hesitate to take a video of any suspiciously abnormal behavior and bring it to their family doctor or pediatric neurologist?
Dr. Rao: Yes. I was happy to see the Tuberous Sclerosis Alliance put out a promotional video that had some steps for when parents see things that are suspicious for spasms, and they do recommend using smartphone video and promptly showing it to their doctors. I think the difference that we hope to provide in this study is that we can now quantify the effect of having that smartphone video when they first present.
My takeaway from this study that I would like to show is encouraging the use of smartphone video as an adjunct tool and for providers to ask for the videos, but also for these pediatric centers to develop an infrastructure – either a secure, monitored email address like we have at our center or a patient portal – where parents can submit video concerning for spasms.
Dr. Wilner: Save the trip to the doctor. Get that video out there first.
Dr. Rao: Especially in the pandemic world, right?
Dr. Wilner: Yes. I understand that you are a neurology resident. To wrap up, what’s the next step for you?
Dr. Rao: I’m finishing up my child neurology residency this year, and I’m moving out to Stanford for pediatric epilepsy fellowship. We’re preparing this project we’re talking about for submission soon, and we’re working on another project, which is a systematic review of genetic testing and the presurgical workup for pediatric drug-resistant focal epilepsy.
Dr. Wilner: Excellent. That’s pretty exciting. Good luck to you. I want to thank you very much for telling us about your research.
Dr. Rao: It was a pleasure speaking with you, and I look forward to the next time.
Dr. Wilner: I’m Dr Andrew Wilner, reporting for Medscape. Thanks for watching.
A version of this article first appeared on Medscape.com.
This video transcript has been edited for clarity.
Andrew N. Wilner, MD: Welcome to Medscape. I’m Dr Andrew Wilner, reporting from the American Epilepsy Society meeting.
Today, I have the pleasure of speaking with Dr. Chethan Rao, a child and adolescent neurology resident from the Mayo Clinic in Jacksonville, Fla. Dr. Rao has a particular interest in pediatric epilepsy. Welcome, Dr. Rao.
Chethan Rao, DO: Thank you, Dr. Wilner. It’s a pleasure to be here, and thanks for taking the time to highlight our work.
Dr. Wilner: You had a very interesting paper at the meeting that I wanted to talk about, focused on infantile spasms and smartphone video. Before we dive into the paper, tell us: What are infantile spasms, and why is it important to diagnose them early?
Dr. Rao: Infantile spasms, also known as epileptic spasms, are 1- to 2-second seizures, and they typically consist of sudden stiffening of the body with brief bending forward or backward of the arms, legs, and head. They usually happen around age 3-8 months, and they typically occur in clusters, most often after awakening from sleep.
The incidence is about 1 in 2,000-3,000 children. Many kids with spasms go on to develop seizures that are very difficult to treat, like Lennox-Gastaut epilepsy, and many go on to have developmental delays as well.
Dr. Wilner: Are these subtle? In other words, could a parent have a child like that and not really recognize that this is something abnormal? Or are they so dramatic that parents say: “We’re going to the emergency room?”
Dr. Rao: One of the problems that we encounter often is that in this age group of infants, they have benign sleep myoclonus; they have Sandifer syndrome related to reflux. Those can be very difficult mimics of spasms. They’re not the most clear-cut, but they look usually different enough from normal baby movements that they get parents to seek medical attention.
Dr. Wilner: You mentioned that the infantile spasms really are a type of epilepsy and symptomatic, usually, of some underlying neurologic condition. Why is it so important to diagnose them early?
Dr. Rao: Great question. Many studies have looked at developmental outcomes based on when spasms were diagnosed and treated, and all of them have replicated time over time that the earlier you get to treatment for the spasms, the better the outcomes are for seizure control and for development.
For this reason, infantile spasm is considered a neurologic urgency in our world. Like I said, accurate diagnosis is often complicated by these potential mimics. Prompt EEG is one of the most important things for confirmation of diagnosis.
Dr. Wilner: But to get that EEG, it has to get all the way to the neurologist, right? It’s not something they’re going to do in the ER. I saw a statistic: There are millions, if not billions, of smartphones out there. Where does the smartphone come in?
Dr. Rao: Absolutely. One of the things that we have on our side these days is that almost everyone has a smartphone at their disposal. One of the recent polls in 2021 showed that more than 95% of adults of childbearing age have smartphones with video access. As some other studies have shown in the adult world, we all really have an epilepsy monitoring unit minus the EEG in our own pockets.
It’s definitely a useful tool, as that first screening video can be used in adjunct to history and physical. There have been many of studies on the adult epilepsy side showing the predictive value of smartphone video for differentiating things like epileptic seizures and nonepileptic spells. What we wanted to do is use smartphone video to pin the diagnosis early of infantile spasms and get it treated as quickly as possible.
Dr. Wilner: I’m a fan. Every now and then, I do have a patient who brings in a video of some spell. I’m an adult neurologist. The patient had a spell, and you ask them – of course they don’t remember – and you ask the witness, who usually is not a trained observer. There have been one or two occasions where I thought: “Well, I don’t know if that was really a seizure.” Then they show me the video and it’s like, “Wow, that is definitely a convulsion.” A picture definitely can be worth a thousand words.
You studied this systematically for your poster. Tell me about what you did.
Dr. Rao: Since the poster, we’ve actually expanded the study, so I’ll give you the updated version. We looked at 101 infants retrospectively at two large children’s health care centers: Nemours Children’s, associated with Mayo Clinic in Jacksonville, Fla., and Texas Children’s Hospital in Houston. We narrowed it down to 80 patients whom we included. Of these, 43 had smartphone video capture when they first presented and 37 had no video when they first presented.
We found a 17-day difference by median in the time to diagnosis and treatment. In other words, the video group was diagnosed and treated 17 days by median, compared with the no-video group. Although 17 days may not sound like a big number, in this context it can make a huge difference. That’s been shown by one of these key studies in our field called the UK Infantile Spasms Study. The 2-week difference made about a 10-point difference on the developmental scale that they use – so pretty significant.
Dr. Wilner: Let me think about this for a minute. Was that because the parents brought the child in with their video and the doctor said, “Hey, that’s infantile spasms. Here’s your shot of ACTH [or whatever they’re using these days].” Or was it because the parents who were attentive enough to use video brought their kids in sooner?
Or was this the time from when they brought the child in to treatment? Is that the time you looked at? So it wasn’t just that these were more attentive parents and more likely to use the video – you’re looking at the time from presentation with or without video until treatment, is that right?
Dr. Rao: We looked to the time from the start of the spasms, as reported by the parents, to the time of diagnosis and then the start of spasms to the time of treatment. What you asked was a fantastic question. We wanted to know who these parents are who are taking videos versus the ones that are not.
We looked at the race/ethnicity data and socioeconomic status data. There were no significant differences between the video and nonvideo group. That would not explain the difference in our results here.
Dr. Wilner: Do you have plans to follow these approximately 40 children 5 years from now and see who’s riding a bicycle and who’s still stuck in the stroller? Is there going to be a difference?
Dr. Rao: Because time to diagnosis and time to treatment were our primary outcomes, long-term follow-up may not really help as much in this study. We did have a couple of other ideas for future studies. One that we wanted to look at was kids who have risk factors for developing spasms, such as trisomy 21, tuberous sclerosis, and congenital cortical malformations; those kids are at a much higher risk for developing spasms around 3-8 months of life.
In giving targeted counseling to those families about how they can use smartphone video to minimize the time to diagnosis and treatment, we think we may be able to learn more and maybe do that prospectively.
The other interesting idea is using artificial intelligence technology for spasm detection in some of these smartphone videos. They’re already using it for different seizure types. It could be an efficient first pass when we get a whole bunch of smartphone videos to determine which ones we need to pursue further steps – to see whether we need to get long-term EEG monitoring or not.
Dr. Wilner: As an epileptologist, I was going to say that we have smartphone EKG. All we need now is smartphone EEG, and then you’ll have all the information you need on day one. It may be a ways away.
As a bottom line, would it be fair to say that parents should not hesitate to take a video of any suspiciously abnormal behavior and bring it to their family doctor or pediatric neurologist?
Dr. Rao: Yes. I was happy to see the Tuberous Sclerosis Alliance put out a promotional video that had some steps for when parents see things that are suspicious for spasms, and they do recommend using smartphone video and promptly showing it to their doctors. I think the difference that we hope to provide in this study is that we can now quantify the effect of having that smartphone video when they first present.
My takeaway from this study that I would like to show is encouraging the use of smartphone video as an adjunct tool and for providers to ask for the videos, but also for these pediatric centers to develop an infrastructure – either a secure, monitored email address like we have at our center or a patient portal – where parents can submit video concerning for spasms.
Dr. Wilner: Save the trip to the doctor. Get that video out there first.
Dr. Rao: Especially in the pandemic world, right?
Dr. Wilner: Yes. I understand that you are a neurology resident. To wrap up, what’s the next step for you?
Dr. Rao: I’m finishing up my child neurology residency this year, and I’m moving out to Stanford for pediatric epilepsy fellowship. We’re preparing this project we’re talking about for submission soon, and we’re working on another project, which is a systematic review of genetic testing and the presurgical workup for pediatric drug-resistant focal epilepsy.
Dr. Wilner: Excellent. That’s pretty exciting. Good luck to you. I want to thank you very much for telling us about your research.
Dr. Rao: It was a pleasure speaking with you, and I look forward to the next time.
Dr. Wilner: I’m Dr Andrew Wilner, reporting for Medscape. Thanks for watching.
A version of this article first appeared on Medscape.com.
Neurotransmitter-based diagnosis and treatment: A hypothesis (Part 1)
It is unfortunate that, in some clinical areas, medical conditions are still treated by name and not based on the underlying pathological process. It would be odd in 2022 to treat “dropsy” instead of heart or kidney disease (2 very different causes of edema). Similarly, if the FDA had been approving drugs 150 years ago, we would have medications on label for “dementia praecox,” not schizophrenia or Alzheimer disease. With the help of DSM-5, psychiatry still resides in the descriptive symptomatic world of disorders.
In the United States, thanks to Freud, psychiatric symptoms became separated from medical symptoms, which made it more difficult to associate psychiatric manifestations with the underlying pathophysiology. Though the physical manifestations that parallel emotional symptoms—such as the dry mouth of anxiety, the tremor and leg weakness of fear, the constipation and blurry vision of depression, the breathing difficulty of anger, the abdominal pain of stress, the blushing of shyness, the palpitations of flashbacks, and endless others—are well known, the present classification of psychiatric disorders is blind to it. Neurochemical causes of gastrointestinal spasm or muscle tension are better researched than underlying central neurochemistry, though the same neurotransmitters drive them.
Can the biochemistry of psychiatric symptoms be judged on the basis of peripheral symptoms? Can the mental manifestations be connected to biological causation, and vice versa? Would psychiatrists be better off selecting treatments by recognizing involved neurotransmitters instead of addressing descriptive “depression, anxiety, and psychosis”? Each of these clinical syndromes may be caused by entirely different underlying neuronal mechanisms. Such mechanisms could be suggested if medical symptoms (which are measurable and objective) would become part of the psychiatric diagnosis. Is treating the “cough” sufficient, or would recognition that tuberculosis caused the cough guide better treatment? Is it time to abandon descriptive conditions and replace them with a specific “mechanism-based” viewpoint?
Ample research has shown that serotonin, dopamine, norepinephrine, endorphins, glutamate, and gamma aminobutyric acid (GABA) are the neurotransmitters most responsible in the process of both psychiatric disorders and chronic pain. These neurotransmitters are involved in much more than emotions (including the feeling of pain). An abundance of medical symptom clusters point toward which neurotransmitter dysfunction may be leading in specific cases of distinct types of depression, psychosis, anxiety, or “chronic pain.” Even presently, there are medications available (both for FDA-approved indications and off-label) that can be used to regulate these neurotransmitters, allowing practitioners to target the possible biological underlining of psychiatric or pain pathology. Hopefully, in the not-so-distant future, there will be specific medications for serotonin, dopamine, and noradrenergic depression as well as for GABAergic anxiety, endorphin psychosis, noradrenergic insomnia, and similar conditions.
Numerous neurotransmitters may be connected to both depression and pain in all their forms. These include (but are not limited to) prostaglandins, bradykinins, substance P, potassium, magnesium, calcium, histamine, adenosine triphosphate, calcitonin gene-related peptide (CGRP), nitric oxide (NO), cholecystokinin 7 (CCK7), neurotrophic growth factor (NGF), neurotensin, acetylcholine (Ach), oxytocin, cannabinoids, and others. These have not been researched sufficiently to identify their clinical presentation of excessive or insufficient availability at the sites of neurotransmission. It is difficult to draw conclusions about what kind of clinical symptoms they may cause (outside of pain), and therefore, they are not addressed in this article.
Both high and low levels of certain neurotransmitters may be associated with psychiatric conditions and chronic pain. Too much is as bad as too little.1 This applies to both quantity of neurotransmitters as well as quality of the corresponding receptor activity. An astute clinician may judge which neurotransmitter is dysfunctional based on the patient’s presentation. Reading indirect signs of bodily functions is a basic clinical skill that should not be forgotten, even in the time of advanced technology.
A different way of viewing psychiatric disorders
In this article, we present 4 hypothetical clinical cases to emphasize a possible way of analyzing symptoms to identify underlying pathology and guide more effective treatment. In no way do these descriptions reflect the entire set of symptoms caused by neurotransmitters; we created them based on what is presently known or suspected, and extensive research is required to confirm or disprove what we describe here.
Continue to: There are no well-recognized...
There are no well-recognized, well-established, reliable, or validated syndromes described in our work. Our goal is to suggest an alternative way of looking at psychiatric disorders by viewing syndromal presentation through the lens of specific neurotransmitters. The collection of symptoms associated with various neurotransmitters as presented in this hypothesis is not complete. We have assembled what is described in the literature as a suggestion for specific future research. We simplified these clinical presentations by omitting scenarios in which a specific neurotransmitter increases in one area but not another. For example, all the symptoms of dopamine excess we describe would not have to occur concurrently in the same patient, but they may develop in certain patients depending on which dopaminergic pathway is exhibiting excess activity. Such distinctions may be established only by exhaustive research not yet conducted.
Our proposal may seem radical, but it truly is not. For example, if we know that dopamine excess may cause seizures, psychosis, and blood pressure elevation, why not consider dopamine excess as an underlying cause in a patient with depression who exhibits these symptoms simultaneously? And why not call it “dopamine excess syndrome”? We already have “serotonin syndrome” for a patient experiencing a serotonin storm. However, using the same logic, it should be called “serotonin excess syndrome.” And if we know of “serotonin excess syndrome,” why not consider “serotonin deficiency syndrome”?
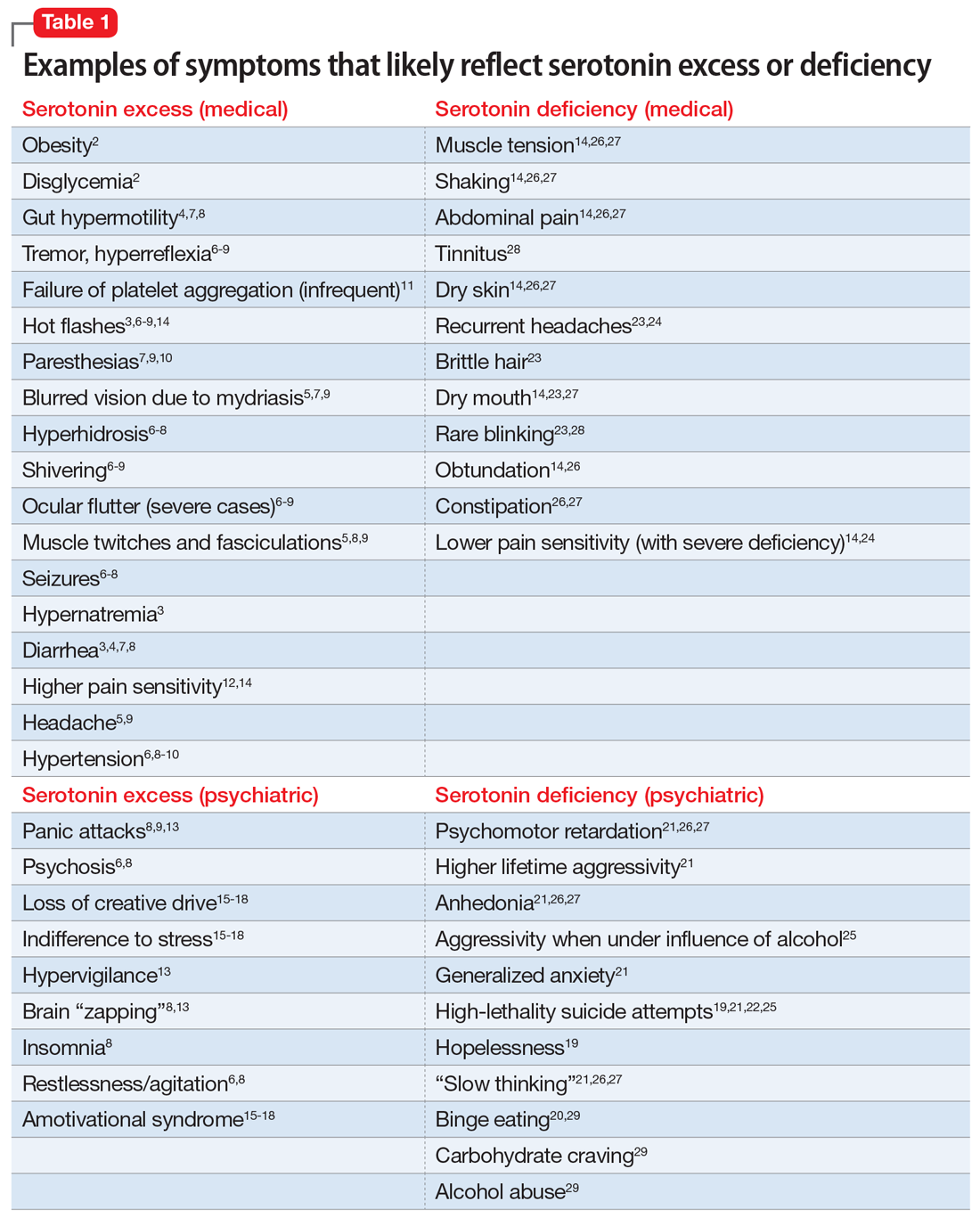
In Part 1 of this article, we discuss serotonin and dopamine. Table 1 outlines medical and psychiatric symptoms that likely reflect serotonin excess2-18 and deficiency,14,19-29 and Table 2 lists symptoms that likely reflect dopamine excess14,30-41 and deficiency.4,14,20,38,40-43 In Part 2 we will touch on endorphins and norepinephrine, and in Part 3 we will conclude by looking at GABA and glutamate.
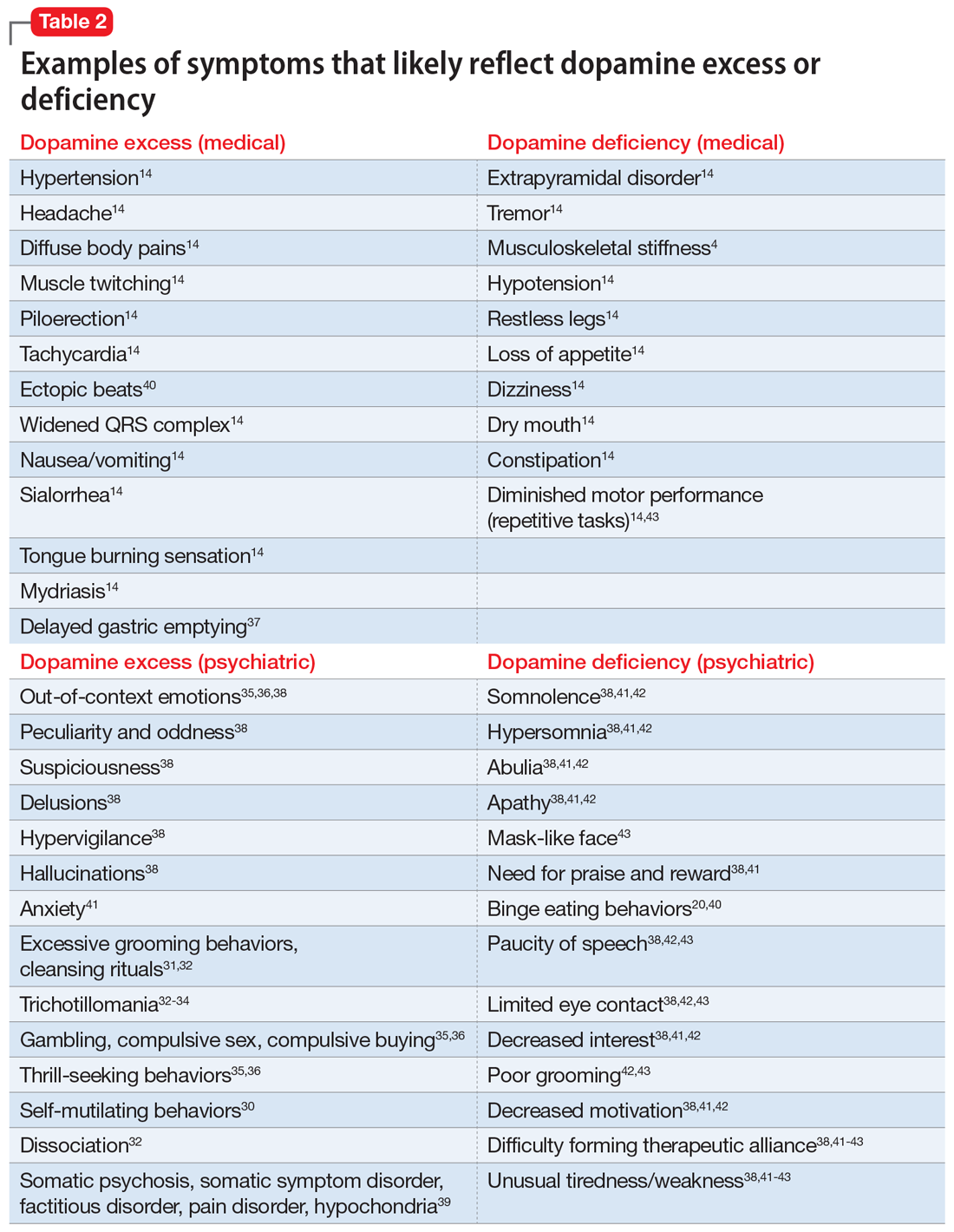
Serotonin excess (Table 12-18)
On a recent office visit, Ms. H reports that most of the time she does not feel much of anything, but she still experiences panic attacks8,9,13,15 and is easily agitated.6,8 Her mother died recently, and Ms. H is concerned that she did not grieve.15-18 She failed her last semester in college and was indifferent to her failure.18 She sleeps poorly,8 is failing her creative classes, and wonders why she has lost her artistic inclination.16-18 Ms. H has difficulty with amotivation, planning, social interactions, and speech.16,17 All of those symptoms worsened after she was prescribed fluoxetine approximately 1 year ago for her “blues.” Ms. H is obese and continues to gain weight,2 though she frequently has diarrhea,3,4,7,8 loud peristalsis, and abdominal cramps.4,7,8 She sweats easily6-8 and her heart frequently races.8,9 Additionally, Ms. H’s primary care physician told her that she has “borderline diabetes.”2 She is prone to frequent bruising11 and is easy to shake, even when she is experiencing minimal anxiety.6-9 Ms. H had consulted with a neurologist because of unusual electrical “zapping” in her brain and muscle twitches.5,8,9,13 She had experienced a seizure as a child, but this was possibly related to hypernatremia,2 and she has not taken any anticonvulsant medication for several years.8 She exhibits hyperactive deep tendon reflexes and tremors5,7,9 and blinks frequently.6,9 She experiences hot flashes,3,6-8,14 does not tolerate heat, and prefers cooler weather.8,9 Her pains and aches,12,14 to which she has been prone all of her life, have recently become much worse, and she was diagnosed with fibromyalgia in part because she frequently feels stiff all over.10 She complains of strange tingling and prickling sensations in her hands and feet, especially when anxious.7,9,10 Her headaches also worsened and may be precipitated by bright light, as her pupils are usually dilated.5,7,9 Her hypertension is fairly controlled with medication.6,8-10 Ms. H says she experienced a psychotic episode when she was in her mid-teens,6,8 but reassures you that “she is not that bad now,” although she remains hypervigilant.13 Also while in her teens, Ms. H was treated with paroxetine and experienced restlessness, agitation, delirium, tachycardia, fluctuating blood pressure, diaphoresis, diarrhea, and neuromuscular excitation, which prompted discontinuation of the antidepressant.5-7,9,10
Impression. Ms. H exhibits symptoms associated with serotonin hyperactivity. Discontinuing and avoiding selective serotonin reuptake inhibitors (SSRIs) would be prudent; prescribing an anticonvulsant would be reasonable. Using a GABAergic medication to suppress serotonin (eg, baclofen) is likely to help. Avoiding dopaminergic medications is a must. Antidepressive antipsychotics would be logical to use. The use of serotonin-suppressing medications may be considered. One may argue for the use of beta-blockers in such a patient.
Continue to: Serotonin deficiency
Serotonin deficiency (Table 114,19-29)
Mr. A is chronically depressed, hopeless,19 and easily angered.21 He does not believe anyone can help him.19 You are concerned for his safety because he had attempted to end his life by shooting himself in the chest.19,21,22,25 Even when he’s not particularly depressed, Mr. A does not enjoy much of anything.21,26,27 He becomes particularly agitated when he drinks alcohol,25 which unfortunately is common for him.29 He engages in binge eating to feel better; he knows this is not healthy but he cannot control his behavior.20,29 Mr. A is poorly compliant with his medications, even with a blood thinner, which he was prescribed due to an episode of deep vein thrombosis. He complains of chronic daily headaches and episodic migraines.23,24 He rarely blinks,23,28 his skin is dry and cool, his hair is brittle,23 his mouth is dry,14,23,27 and he constantly licks his chapped lips.14,26,27 Mr. A frequently has general body pain26,31 but is dismissive of his body aches and completely stops reporting pain when his depression gets particularly severe. When depressed, he is slow in movement and thinking.14,21,26,27 He is more concerned with anxiety than depression.21 Mr. A is plagued by constipation, abdominal pain, muscle tension, and episodes of shaking.14,26,27 He also frequently complains about chronic tinnitus.28
Impression. Mr. A shows symptoms associated with serotonin hypoactivity. SSRIs and any other antidepressants with serotonin activity would be an obvious choice for treatment. A mood-stabilizing antipsychotic with serotonin activity would be welcome in treatment. Thyroid hormone supplementation may be of value, especially if thyroid stimulating hormone level is high. Light therapy, a diet with food that contains tryptophan, psychotherapy, and exercise are desirable. Avoiding benzodiazepines would be a good idea.
Dopamine excess (Table 214,30-41)
Ms. L presents with complaints of “fibromyalgia” and “daily headaches,”14 and also dissociation (finding herself in places when she does not know how she got there) and “out-of-body experiences.”32 She is odd, and states that people do not understand her and that she is “different.”38 Her friend, who is present at the appointment, elaborates on Ms. L’s bizarreness and oddness in behavior, out-of-context emotions, suspiciousness, paranoia, and possible hallucinations.35,36,38 Ms. L discloses frequent diffuse body pains, headaches, nausea, excessive salivation, and tongue burning, as well as muscle twitching.14 Sex worsens her headaches and body pain. She reports seizures that are not registered on EEG. In the office, she is suspicious, exhibits odd posturing, tends to misinterpret your words, and makes you feel uncomfortable. Anxiety38 and multiple obsessive-compulsive symptoms, especially excessive cleaning and grooming, complicate Ms. L’s life.31,32,34 On examination, she is hypertensive, and she has scars caused by self-cutting and skin picking on her arms.30-32 An electrocardiogram shows an elevated heart rate, widened QRS complex, and ectopic heartbeats.14 Ms. L has experienced trichotillomania since adolescence32-34 and her fingernails are bitten to the skin.34 She has difficulty with impulse control, and thrill-seeking is a prominent part of her life, mainly via gambling, compulsive sex, and compulsive buying.35,36 She also says she experiences indigestion and delayed gastric emptying.37
Impression. Ms. L exhibits multiple symptoms associated with dopamine excess. Dopamine antagonists should be considered and may help not only with her psychiatric symptoms but also with her pain symptoms. Bupropion (as a dopamine agonist), caffeine, and stimulants should be avoided.
Excessive dopamine is, in extreme cases, associated with somatic psychosis, somatic symptom disorder, factitious disorder, pain disorder, and hypochondria.39 It may come with odd and bizarre/peculiar symptoms out of proportion with objectively identified pathology. These symptoms are common in chronic pain and headache patients, and need to be addressed by appropriate use of dopamine antagonizing medications.39
Continue to: Dopamine deficiency
Dopamine deficiency (Table 24,14,20,38,40-43)
Mr. W experiences widespread pain, including chronic back pain, headaches, and abdominal pain. He also has substantial anhedonia, lack of interest, procrastination, and hypersomnia.41,42 He is apathetic and has difficulty getting up in the morning.41,42 Unusual tiredness and weakness drive him to overuse caffeine; he states that 5 Mountain Dews and 4 cups of regular coffee a day make his headaches bearable.38,41-43 Sex also improves his headaches. Since childhood, he has taken stimulants for attention-deficit/hyperactivity disorder. He reports that occasional use of cocaine helps ease his pain and depression. Mr. W’s wife is concerned with her husband’s low sexual drive and alcohol consumption, and discloses that he has periodic trouble with gambling. Mr. W was forced into psychotherapy but never was able to work productively with his therapist.38,41-43 He loves eating and cannot control his weight.40 This contrasts with episodic anorexia he experienced when he was younger.20 His face is usually emotionless.43 Mr. W is prone to constipation.14 His restless leg syndrome and periodic limb movement disorder are so bad that his wife refuses to share a bed with him.14 He is clumsy and has a problem with repetitive motor tasks.43 A paucity of speech, limited eye contact, poor grooming, and difficulty forming therapeutic alliances have long been part of Mr. W’s history.38,42,43 On physical examination, he has a dry mouth; he is stiff, tremulous, and hypotensive.14
Impression. Mr. W shows multiple symptoms associated with dopamine deficiency. Bupropion may be reasonable to consider. Dopamine augmentation via the use of stimulants is warranted in such patients, especially if stimulants had not been tried before (lisdexamfetamine would be a good choice to minimize addictive potential). For a patient with dopamine deficiency, levodopa may improve more than just restless legs. Amantadine may improve dopaminergic signaling through the accelerated dopamine release and decrease in presynaptic uptake, so this medication may be carefully tried.44 Pain treatment would not be successful for Mr. W without simultaneous treatment for his substance use.
Bottom Line
Both high and low levels of serotonin and dopamine may be associated with certain psychiatric and medical symptoms and disorders. An astute clinician may judge which neurotransmitter is dysfunctional based on the patient’s presentation, and tailor treatment accordingly.
Related Resources
- Abell SR, El-Mallakh RS. Serotonin-mediated anxiety: How to recognize and treat it. Current Psychiatry. 2021;20(11):37-40. doi:10.12788/cp.0168
Drug Brand Names
Amantadine • Gocovri
Baclofen • Ozobax
Bupropion • Wellbutrin
Fluoxetine • Prozac
Lisdexamfetamine • Vyvanse
Paroxetine • Paxil
1. Stahl SM. Dazzled by the dominions of dopamine: clinical roles of D3, D2, and D1 receptors. CNS Spectr. 2017;22(4):305-311.
2. Young RL, Lumsden AL, Martin AM, et al. Augmented capacity for peripheral serotonin release in human obesity. Int J Obes (Lond). 2018;42(11):1880-1889.
3. Ahlman H. Serotonin and carcinoid tumors. J Cardiovasc Pharmacol. 1985;7(Suppl 7):S79-S85.
4. Terry N, Margolis KG. Serotonergic mechanisms regulating the GI tract: experimental evidence and therapeutic relevance. Handb Exp Pharmacol. 2017;239:319-342.
5. Prakash S, Belani P, Trivedi A. Headache as a presenting feature in patients with serotonin syndrome: a case series. Cephalalgia. 2014;34(2):148-153.
6. van Ewijk CE, Jacobs GE, Girbes ARJ. Unsuspected serotonin toxicity in the ICU. Ann Intensive Care. 2016;6(1):85.
7. Pedavally S, Fugate JE, Rabinstein AA. Serotonin syndrome in the intensive care unit: clinical presentations and precipitating medications. Neurocrit Care. 2014;21(1):108-113.
8. Nguyen H, Pan A, Smollin C, et al. An 11-year retrospective review of cyproheptadine use in serotonin syndrome cases reported to the California Poison Control System. J Clin Pharm Ther. 2019;44(2):327-334.
9. Ansari H, Kouti L. Drug interaction and serotonin toxicity with opioid use: another reason to avoid opioids in headache and migraine treatment. Curr Pain Headache Rep. 2016;20(8):50.
10. Ott M, Mannchen JK, Jamshidi F, et al. Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques. Ther Adv Psychopharmacol. 2019;9:2045125318818814. doi:10.1177/2045125318818814
11. Cerrito F, Lazzaro MP, Gaudio E, et al. 5HT2-receptors and serotonin release: their role in human platelet aggregation. Life Sci. 1993;53(3):209-215.
12. Ohayon MM. Pain sensitivity, depression, and sleep deprivation: links with serotoninergic dysfunction. J Psychiatr Res. 2009;43(16):1243-1245.
13. Maron E, Shlik J. Serotonin function in panic disorder: important, but why? Neuropsychopharmacology. 2006;31(1):1-11.
14. Hall JE, Guyton AC. Textbook of Medical Physiology. 12th ed. Spanish version. Elsevier; 2011:120,199,201-204,730-740.
15. Garland EJ, Baerg EA. Amotivational syndrome associated with selective serotonin reuptake inhibitors in children and adolescents. J Child Adolesc Psychopharmacol. 2001;11(2):181-186.
16. George MS, Trimble MR. A fluvoxamine-induced frontal lobe syndrome in a patient with comorbid Gilles de la Tourette’s syndrome and obsessive compulsive disorder. J Clin Psychiatry. 1992;53(10):379-380.
17. Hoehn-Saric R, Harris GJ, Pearlson GD, et al. A fluoxetine-induced frontal lobe syndrome in an obsessive compulsive patient. J Clin Psychiatry. 1991;52(3):131-133.
18. Hoehn-Saric R, Lipsey JR, McLeod DR. Apathy and indifference in patients on fluvoxamine and fluoxetine. J Clin Psychopharmacol. 1990;10(5):343-345.
19. Samuelsson M, Jokinen J, Nordström AL, et al. CSF 5-HIAA, suicide intent and hopelessness in the prediction of early suicide in male high-risk suicide attempters. Acta Psychiatr Scand. 2006;113(1):44-47.
20. Brewerton TD. Clinical Handbook of Eating Disorders: An Integrated Approach. CRC Press; 2004:257-281.
21. Mann JJ, Oquendo M, Underwood MD, et al. The neurobiology of suicide risk: a review for the clinician. J Clin Psychiatry. 1999;60 Suppl 2:7-116.
22. Mann JJ, Malone KM. Cerebrospinal fluid amines and higher-lethality suicide attempts in depressed inpatients. Biol Psychiatry. 1997;41(2):162-171.
23. Joseph R, Welch KM, D’Andrea G. Serotonergic hypofunction in migraine: a synthesis of evidence based on platelet dense body dysfunction. Cephalalgia. 1989;9(4):293-299.
24. Pakalnis A, Splaingard M, Splaingard D, et al. Serotonin effects on sleep and emotional disorders in adolescent migraine. Headache. 2009;49(10):1486-1492.
25. Virkkunen M, Goldman D, Nielsen DA, et al. Low brain serotonin turnover rate (low CSF 5-HIAA) and impulsive violence. J Psychiatry Neurosci. 1995;20(4):271-275.
26. Liu Y, Zhao J, Fan X, et al. Dysfunction in serotonergic and noradrenergic systems and somatic symptoms in psychiatric disorders. Front Psychiatry. 2019;10:286.
27. Ginsburg GS, Riddle MA, Davies M. Somatic symptoms in children and adolescents with anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2006;45(10):1179-1187.
28. O’Malley PG, Jackson JL, Santoro J, et al. Antidepressant therapy for unexplained symptoms and symptom syndromes. J Fam Pract. 1999;48(12):980-990.
29. Fortuna JL. Sweet preference, sugar addiction and the familial history of alcohol dependence: shared neural pathways and genes. J Psychoactive Drugs. 2010;42(2):147-151.
30. Stanley B, Sher L, Wilson S, et al. Non-suicidal self-injurious behavior, endogenous opioids and monoamine neurotransmitters. J Affect Disord. 2010;124(1-2):134-140.
31. Graybiel AM, Saka E. A genetic basis for obsessive grooming. Neuron. 2002;33(1):1-2.
32. Tse W, Hälbig TD. Skin picking in Parkinson’s disease: a behavioral side-effect of dopaminergic treatment? Psychiatry Clin Neurosci. 2010;64(2):214.
33. Ayaydın H. Probable emergence of symptoms of trichotillomania by atomoxetine: a case report. Psychiatry and Clinical Psychopharmacology. 2019;29(2)220-222.
34. Paholpak P, Mendez MF. Trichotillomania as a manifestation of dementia. Case Rep Psychiatry. 2016;2016:9782702. doi:10.1155/2016/9782702
35. Clark CA, Dagher A. The role of dopamine in risk taking: a specific look at Parkinson’s disease and gambling. Front Behav Neurosci. 2014;8:196.
36. Norbury A, Husain M. Sensation-seeking: dopaminergic modulation and risk for psychopathology. Behav Brain Res. 2015;288:79-93.
37. Chen TS, Chang FY. Elevated serum dopamine increases while coffee consumption decreases the occurrence of reddish streaks in the intact stomach. J Gastroenterol Hepatol. 2013;28(12):1810-1814.
38. Wong-Riley MT. Neuroscience Secrets. 1st edition. Spanish version. Hanley & Belfus; 1999:420-429.
39. Arbuck DM. Antipsychotics, dopamine, and pain. Current Psychiatry. 2020;19(1):25-29,31.
40. Bello NT, Hajnal A. Dopamine and binge eating behaviors. Pharmacol Biochem Behav. 2010;97(1):25-33.
41. Velligan DI, Weiden PJ, Sajatovic M, et al; Expert Consensus Panel on Adherence Problems in Serious and Persistent Mental Illness. The expert consensus guideline series: adherence problems in patients with serious and persistent mental illness. J Clin Psychiatry. 2009;70 Suppl 4:1-46.
42. Milev P, Ho BC, Arndt S, et al. Predictive values of neurocognition and negative symptoms on functional outcome in schizophrenia: a longitudinal first-episode study with 7-year follow-up. Am J Psychiatry. 2005;162(3):495-506.
43. Gepshtein S, Li X, Snider J, et al. Dopamine function and the efficiency of human movement. J Cogn Neurosci. 2014;26(3):645-657.
44. Scarff JR. The ABCDs of treating tardive dyskinesia. Current Psychiatry. 2020;19(4):21,55.
It is unfortunate that, in some clinical areas, medical conditions are still treated by name and not based on the underlying pathological process. It would be odd in 2022 to treat “dropsy” instead of heart or kidney disease (2 very different causes of edema). Similarly, if the FDA had been approving drugs 150 years ago, we would have medications on label for “dementia praecox,” not schizophrenia or Alzheimer disease. With the help of DSM-5, psychiatry still resides in the descriptive symptomatic world of disorders.
In the United States, thanks to Freud, psychiatric symptoms became separated from medical symptoms, which made it more difficult to associate psychiatric manifestations with the underlying pathophysiology. Though the physical manifestations that parallel emotional symptoms—such as the dry mouth of anxiety, the tremor and leg weakness of fear, the constipation and blurry vision of depression, the breathing difficulty of anger, the abdominal pain of stress, the blushing of shyness, the palpitations of flashbacks, and endless others—are well known, the present classification of psychiatric disorders is blind to it. Neurochemical causes of gastrointestinal spasm or muscle tension are better researched than underlying central neurochemistry, though the same neurotransmitters drive them.
Can the biochemistry of psychiatric symptoms be judged on the basis of peripheral symptoms? Can the mental manifestations be connected to biological causation, and vice versa? Would psychiatrists be better off selecting treatments by recognizing involved neurotransmitters instead of addressing descriptive “depression, anxiety, and psychosis”? Each of these clinical syndromes may be caused by entirely different underlying neuronal mechanisms. Such mechanisms could be suggested if medical symptoms (which are measurable and objective) would become part of the psychiatric diagnosis. Is treating the “cough” sufficient, or would recognition that tuberculosis caused the cough guide better treatment? Is it time to abandon descriptive conditions and replace them with a specific “mechanism-based” viewpoint?
Ample research has shown that serotonin, dopamine, norepinephrine, endorphins, glutamate, and gamma aminobutyric acid (GABA) are the neurotransmitters most responsible in the process of both psychiatric disorders and chronic pain. These neurotransmitters are involved in much more than emotions (including the feeling of pain). An abundance of medical symptom clusters point toward which neurotransmitter dysfunction may be leading in specific cases of distinct types of depression, psychosis, anxiety, or “chronic pain.” Even presently, there are medications available (both for FDA-approved indications and off-label) that can be used to regulate these neurotransmitters, allowing practitioners to target the possible biological underlining of psychiatric or pain pathology. Hopefully, in the not-so-distant future, there will be specific medications for serotonin, dopamine, and noradrenergic depression as well as for GABAergic anxiety, endorphin psychosis, noradrenergic insomnia, and similar conditions.
Numerous neurotransmitters may be connected to both depression and pain in all their forms. These include (but are not limited to) prostaglandins, bradykinins, substance P, potassium, magnesium, calcium, histamine, adenosine triphosphate, calcitonin gene-related peptide (CGRP), nitric oxide (NO), cholecystokinin 7 (CCK7), neurotrophic growth factor (NGF), neurotensin, acetylcholine (Ach), oxytocin, cannabinoids, and others. These have not been researched sufficiently to identify their clinical presentation of excessive or insufficient availability at the sites of neurotransmission. It is difficult to draw conclusions about what kind of clinical symptoms they may cause (outside of pain), and therefore, they are not addressed in this article.
Both high and low levels of certain neurotransmitters may be associated with psychiatric conditions and chronic pain. Too much is as bad as too little.1 This applies to both quantity of neurotransmitters as well as quality of the corresponding receptor activity. An astute clinician may judge which neurotransmitter is dysfunctional based on the patient’s presentation. Reading indirect signs of bodily functions is a basic clinical skill that should not be forgotten, even in the time of advanced technology.
A different way of viewing psychiatric disorders
In this article, we present 4 hypothetical clinical cases to emphasize a possible way of analyzing symptoms to identify underlying pathology and guide more effective treatment. In no way do these descriptions reflect the entire set of symptoms caused by neurotransmitters; we created them based on what is presently known or suspected, and extensive research is required to confirm or disprove what we describe here.
Continue to: There are no well-recognized...
There are no well-recognized, well-established, reliable, or validated syndromes described in our work. Our goal is to suggest an alternative way of looking at psychiatric disorders by viewing syndromal presentation through the lens of specific neurotransmitters. The collection of symptoms associated with various neurotransmitters as presented in this hypothesis is not complete. We have assembled what is described in the literature as a suggestion for specific future research. We simplified these clinical presentations by omitting scenarios in which a specific neurotransmitter increases in one area but not another. For example, all the symptoms of dopamine excess we describe would not have to occur concurrently in the same patient, but they may develop in certain patients depending on which dopaminergic pathway is exhibiting excess activity. Such distinctions may be established only by exhaustive research not yet conducted.
Our proposal may seem radical, but it truly is not. For example, if we know that dopamine excess may cause seizures, psychosis, and blood pressure elevation, why not consider dopamine excess as an underlying cause in a patient with depression who exhibits these symptoms simultaneously? And why not call it “dopamine excess syndrome”? We already have “serotonin syndrome” for a patient experiencing a serotonin storm. However, using the same logic, it should be called “serotonin excess syndrome.” And if we know of “serotonin excess syndrome,” why not consider “serotonin deficiency syndrome”?

In Part 1 of this article, we discuss serotonin and dopamine. Table 1 outlines medical and psychiatric symptoms that likely reflect serotonin excess2-18 and deficiency,14,19-29 and Table 2 lists symptoms that likely reflect dopamine excess14,30-41 and deficiency.4,14,20,38,40-43 In Part 2 we will touch on endorphins and norepinephrine, and in Part 3 we will conclude by looking at GABA and glutamate.

Serotonin excess (Table 12-18)
On a recent office visit, Ms. H reports that most of the time she does not feel much of anything, but she still experiences panic attacks8,9,13,15 and is easily agitated.6,8 Her mother died recently, and Ms. H is concerned that she did not grieve.15-18 She failed her last semester in college and was indifferent to her failure.18 She sleeps poorly,8 is failing her creative classes, and wonders why she has lost her artistic inclination.16-18 Ms. H has difficulty with amotivation, planning, social interactions, and speech.16,17 All of those symptoms worsened after she was prescribed fluoxetine approximately 1 year ago for her “blues.” Ms. H is obese and continues to gain weight,2 though she frequently has diarrhea,3,4,7,8 loud peristalsis, and abdominal cramps.4,7,8 She sweats easily6-8 and her heart frequently races.8,9 Additionally, Ms. H’s primary care physician told her that she has “borderline diabetes.”2 She is prone to frequent bruising11 and is easy to shake, even when she is experiencing minimal anxiety.6-9 Ms. H had consulted with a neurologist because of unusual electrical “zapping” in her brain and muscle twitches.5,8,9,13 She had experienced a seizure as a child, but this was possibly related to hypernatremia,2 and she has not taken any anticonvulsant medication for several years.8 She exhibits hyperactive deep tendon reflexes and tremors5,7,9 and blinks frequently.6,9 She experiences hot flashes,3,6-8,14 does not tolerate heat, and prefers cooler weather.8,9 Her pains and aches,12,14 to which she has been prone all of her life, have recently become much worse, and she was diagnosed with fibromyalgia in part because she frequently feels stiff all over.10 She complains of strange tingling and prickling sensations in her hands and feet, especially when anxious.7,9,10 Her headaches also worsened and may be precipitated by bright light, as her pupils are usually dilated.5,7,9 Her hypertension is fairly controlled with medication.6,8-10 Ms. H says she experienced a psychotic episode when she was in her mid-teens,6,8 but reassures you that “she is not that bad now,” although she remains hypervigilant.13 Also while in her teens, Ms. H was treated with paroxetine and experienced restlessness, agitation, delirium, tachycardia, fluctuating blood pressure, diaphoresis, diarrhea, and neuromuscular excitation, which prompted discontinuation of the antidepressant.5-7,9,10
Impression. Ms. H exhibits symptoms associated with serotonin hyperactivity. Discontinuing and avoiding selective serotonin reuptake inhibitors (SSRIs) would be prudent; prescribing an anticonvulsant would be reasonable. Using a GABAergic medication to suppress serotonin (eg, baclofen) is likely to help. Avoiding dopaminergic medications is a must. Antidepressive antipsychotics would be logical to use. The use of serotonin-suppressing medications may be considered. One may argue for the use of beta-blockers in such a patient.
Continue to: Serotonin deficiency
Serotonin deficiency (Table 114,19-29)
Mr. A is chronically depressed, hopeless,19 and easily angered.21 He does not believe anyone can help him.19 You are concerned for his safety because he had attempted to end his life by shooting himself in the chest.19,21,22,25 Even when he’s not particularly depressed, Mr. A does not enjoy much of anything.21,26,27 He becomes particularly agitated when he drinks alcohol,25 which unfortunately is common for him.29 He engages in binge eating to feel better; he knows this is not healthy but he cannot control his behavior.20,29 Mr. A is poorly compliant with his medications, even with a blood thinner, which he was prescribed due to an episode of deep vein thrombosis. He complains of chronic daily headaches and episodic migraines.23,24 He rarely blinks,23,28 his skin is dry and cool, his hair is brittle,23 his mouth is dry,14,23,27 and he constantly licks his chapped lips.14,26,27 Mr. A frequently has general body pain26,31 but is dismissive of his body aches and completely stops reporting pain when his depression gets particularly severe. When depressed, he is slow in movement and thinking.14,21,26,27 He is more concerned with anxiety than depression.21 Mr. A is plagued by constipation, abdominal pain, muscle tension, and episodes of shaking.14,26,27 He also frequently complains about chronic tinnitus.28
Impression. Mr. A shows symptoms associated with serotonin hypoactivity. SSRIs and any other antidepressants with serotonin activity would be an obvious choice for treatment. A mood-stabilizing antipsychotic with serotonin activity would be welcome in treatment. Thyroid hormone supplementation may be of value, especially if thyroid stimulating hormone level is high. Light therapy, a diet with food that contains tryptophan, psychotherapy, and exercise are desirable. Avoiding benzodiazepines would be a good idea.
Dopamine excess (Table 214,30-41)
Ms. L presents with complaints of “fibromyalgia” and “daily headaches,”14 and also dissociation (finding herself in places when she does not know how she got there) and “out-of-body experiences.”32 She is odd, and states that people do not understand her and that she is “different.”38 Her friend, who is present at the appointment, elaborates on Ms. L’s bizarreness and oddness in behavior, out-of-context emotions, suspiciousness, paranoia, and possible hallucinations.35,36,38 Ms. L discloses frequent diffuse body pains, headaches, nausea, excessive salivation, and tongue burning, as well as muscle twitching.14 Sex worsens her headaches and body pain. She reports seizures that are not registered on EEG. In the office, she is suspicious, exhibits odd posturing, tends to misinterpret your words, and makes you feel uncomfortable. Anxiety38 and multiple obsessive-compulsive symptoms, especially excessive cleaning and grooming, complicate Ms. L’s life.31,32,34 On examination, she is hypertensive, and she has scars caused by self-cutting and skin picking on her arms.30-32 An electrocardiogram shows an elevated heart rate, widened QRS complex, and ectopic heartbeats.14 Ms. L has experienced trichotillomania since adolescence32-34 and her fingernails are bitten to the skin.34 She has difficulty with impulse control, and thrill-seeking is a prominent part of her life, mainly via gambling, compulsive sex, and compulsive buying.35,36 She also says she experiences indigestion and delayed gastric emptying.37
Impression. Ms. L exhibits multiple symptoms associated with dopamine excess. Dopamine antagonists should be considered and may help not only with her psychiatric symptoms but also with her pain symptoms. Bupropion (as a dopamine agonist), caffeine, and stimulants should be avoided.
Excessive dopamine is, in extreme cases, associated with somatic psychosis, somatic symptom disorder, factitious disorder, pain disorder, and hypochondria.39 It may come with odd and bizarre/peculiar symptoms out of proportion with objectively identified pathology. These symptoms are common in chronic pain and headache patients, and need to be addressed by appropriate use of dopamine antagonizing medications.39
Continue to: Dopamine deficiency
Dopamine deficiency (Table 24,14,20,38,40-43)
Mr. W experiences widespread pain, including chronic back pain, headaches, and abdominal pain. He also has substantial anhedonia, lack of interest, procrastination, and hypersomnia.41,42 He is apathetic and has difficulty getting up in the morning.41,42 Unusual tiredness and weakness drive him to overuse caffeine; he states that 5 Mountain Dews and 4 cups of regular coffee a day make his headaches bearable.38,41-43 Sex also improves his headaches. Since childhood, he has taken stimulants for attention-deficit/hyperactivity disorder. He reports that occasional use of cocaine helps ease his pain and depression. Mr. W’s wife is concerned with her husband’s low sexual drive and alcohol consumption, and discloses that he has periodic trouble with gambling. Mr. W was forced into psychotherapy but never was able to work productively with his therapist.38,41-43 He loves eating and cannot control his weight.40 This contrasts with episodic anorexia he experienced when he was younger.20 His face is usually emotionless.43 Mr. W is prone to constipation.14 His restless leg syndrome and periodic limb movement disorder are so bad that his wife refuses to share a bed with him.14 He is clumsy and has a problem with repetitive motor tasks.43 A paucity of speech, limited eye contact, poor grooming, and difficulty forming therapeutic alliances have long been part of Mr. W’s history.38,42,43 On physical examination, he has a dry mouth; he is stiff, tremulous, and hypotensive.14
Impression. Mr. W shows multiple symptoms associated with dopamine deficiency. Bupropion may be reasonable to consider. Dopamine augmentation via the use of stimulants is warranted in such patients, especially if stimulants had not been tried before (lisdexamfetamine would be a good choice to minimize addictive potential). For a patient with dopamine deficiency, levodopa may improve more than just restless legs. Amantadine may improve dopaminergic signaling through the accelerated dopamine release and decrease in presynaptic uptake, so this medication may be carefully tried.44 Pain treatment would not be successful for Mr. W without simultaneous treatment for his substance use.
Bottom Line
Both high and low levels of serotonin and dopamine may be associated with certain psychiatric and medical symptoms and disorders. An astute clinician may judge which neurotransmitter is dysfunctional based on the patient’s presentation, and tailor treatment accordingly.
Related Resources
- Abell SR, El-Mallakh RS. Serotonin-mediated anxiety: How to recognize and treat it. Current Psychiatry. 2021;20(11):37-40. doi:10.12788/cp.0168
Drug Brand Names
Amantadine • Gocovri
Baclofen • Ozobax
Bupropion • Wellbutrin
Fluoxetine • Prozac
Lisdexamfetamine • Vyvanse
Paroxetine • Paxil
It is unfortunate that, in some clinical areas, medical conditions are still treated by name and not based on the underlying pathological process. It would be odd in 2022 to treat “dropsy” instead of heart or kidney disease (2 very different causes of edema). Similarly, if the FDA had been approving drugs 150 years ago, we would have medications on label for “dementia praecox,” not schizophrenia or Alzheimer disease. With the help of DSM-5, psychiatry still resides in the descriptive symptomatic world of disorders.
In the United States, thanks to Freud, psychiatric symptoms became separated from medical symptoms, which made it more difficult to associate psychiatric manifestations with the underlying pathophysiology. Though the physical manifestations that parallel emotional symptoms—such as the dry mouth of anxiety, the tremor and leg weakness of fear, the constipation and blurry vision of depression, the breathing difficulty of anger, the abdominal pain of stress, the blushing of shyness, the palpitations of flashbacks, and endless others—are well known, the present classification of psychiatric disorders is blind to it. Neurochemical causes of gastrointestinal spasm or muscle tension are better researched than underlying central neurochemistry, though the same neurotransmitters drive them.
Can the biochemistry of psychiatric symptoms be judged on the basis of peripheral symptoms? Can the mental manifestations be connected to biological causation, and vice versa? Would psychiatrists be better off selecting treatments by recognizing involved neurotransmitters instead of addressing descriptive “depression, anxiety, and psychosis”? Each of these clinical syndromes may be caused by entirely different underlying neuronal mechanisms. Such mechanisms could be suggested if medical symptoms (which are measurable and objective) would become part of the psychiatric diagnosis. Is treating the “cough” sufficient, or would recognition that tuberculosis caused the cough guide better treatment? Is it time to abandon descriptive conditions and replace them with a specific “mechanism-based” viewpoint?
Ample research has shown that serotonin, dopamine, norepinephrine, endorphins, glutamate, and gamma aminobutyric acid (GABA) are the neurotransmitters most responsible in the process of both psychiatric disorders and chronic pain. These neurotransmitters are involved in much more than emotions (including the feeling of pain). An abundance of medical symptom clusters point toward which neurotransmitter dysfunction may be leading in specific cases of distinct types of depression, psychosis, anxiety, or “chronic pain.” Even presently, there are medications available (both for FDA-approved indications and off-label) that can be used to regulate these neurotransmitters, allowing practitioners to target the possible biological underlining of psychiatric or pain pathology. Hopefully, in the not-so-distant future, there will be specific medications for serotonin, dopamine, and noradrenergic depression as well as for GABAergic anxiety, endorphin psychosis, noradrenergic insomnia, and similar conditions.
Numerous neurotransmitters may be connected to both depression and pain in all their forms. These include (but are not limited to) prostaglandins, bradykinins, substance P, potassium, magnesium, calcium, histamine, adenosine triphosphate, calcitonin gene-related peptide (CGRP), nitric oxide (NO), cholecystokinin 7 (CCK7), neurotrophic growth factor (NGF), neurotensin, acetylcholine (Ach), oxytocin, cannabinoids, and others. These have not been researched sufficiently to identify their clinical presentation of excessive or insufficient availability at the sites of neurotransmission. It is difficult to draw conclusions about what kind of clinical symptoms they may cause (outside of pain), and therefore, they are not addressed in this article.
Both high and low levels of certain neurotransmitters may be associated with psychiatric conditions and chronic pain. Too much is as bad as too little.1 This applies to both quantity of neurotransmitters as well as quality of the corresponding receptor activity. An astute clinician may judge which neurotransmitter is dysfunctional based on the patient’s presentation. Reading indirect signs of bodily functions is a basic clinical skill that should not be forgotten, even in the time of advanced technology.
A different way of viewing psychiatric disorders
In this article, we present 4 hypothetical clinical cases to emphasize a possible way of analyzing symptoms to identify underlying pathology and guide more effective treatment. In no way do these descriptions reflect the entire set of symptoms caused by neurotransmitters; we created them based on what is presently known or suspected, and extensive research is required to confirm or disprove what we describe here.
Continue to: There are no well-recognized...
There are no well-recognized, well-established, reliable, or validated syndromes described in our work. Our goal is to suggest an alternative way of looking at psychiatric disorders by viewing syndromal presentation through the lens of specific neurotransmitters. The collection of symptoms associated with various neurotransmitters as presented in this hypothesis is not complete. We have assembled what is described in the literature as a suggestion for specific future research. We simplified these clinical presentations by omitting scenarios in which a specific neurotransmitter increases in one area but not another. For example, all the symptoms of dopamine excess we describe would not have to occur concurrently in the same patient, but they may develop in certain patients depending on which dopaminergic pathway is exhibiting excess activity. Such distinctions may be established only by exhaustive research not yet conducted.
Our proposal may seem radical, but it truly is not. For example, if we know that dopamine excess may cause seizures, psychosis, and blood pressure elevation, why not consider dopamine excess as an underlying cause in a patient with depression who exhibits these symptoms simultaneously? And why not call it “dopamine excess syndrome”? We already have “serotonin syndrome” for a patient experiencing a serotonin storm. However, using the same logic, it should be called “serotonin excess syndrome.” And if we know of “serotonin excess syndrome,” why not consider “serotonin deficiency syndrome”?

In Part 1 of this article, we discuss serotonin and dopamine. Table 1 outlines medical and psychiatric symptoms that likely reflect serotonin excess2-18 and deficiency,14,19-29 and Table 2 lists symptoms that likely reflect dopamine excess14,30-41 and deficiency.4,14,20,38,40-43 In Part 2 we will touch on endorphins and norepinephrine, and in Part 3 we will conclude by looking at GABA and glutamate.

Serotonin excess (Table 12-18)
On a recent office visit, Ms. H reports that most of the time she does not feel much of anything, but she still experiences panic attacks8,9,13,15 and is easily agitated.6,8 Her mother died recently, and Ms. H is concerned that she did not grieve.15-18 She failed her last semester in college and was indifferent to her failure.18 She sleeps poorly,8 is failing her creative classes, and wonders why she has lost her artistic inclination.16-18 Ms. H has difficulty with amotivation, planning, social interactions, and speech.16,17 All of those symptoms worsened after she was prescribed fluoxetine approximately 1 year ago for her “blues.” Ms. H is obese and continues to gain weight,2 though she frequently has diarrhea,3,4,7,8 loud peristalsis, and abdominal cramps.4,7,8 She sweats easily6-8 and her heart frequently races.8,9 Additionally, Ms. H’s primary care physician told her that she has “borderline diabetes.”2 She is prone to frequent bruising11 and is easy to shake, even when she is experiencing minimal anxiety.6-9 Ms. H had consulted with a neurologist because of unusual electrical “zapping” in her brain and muscle twitches.5,8,9,13 She had experienced a seizure as a child, but this was possibly related to hypernatremia,2 and she has not taken any anticonvulsant medication for several years.8 She exhibits hyperactive deep tendon reflexes and tremors5,7,9 and blinks frequently.6,9 She experiences hot flashes,3,6-8,14 does not tolerate heat, and prefers cooler weather.8,9 Her pains and aches,12,14 to which she has been prone all of her life, have recently become much worse, and she was diagnosed with fibromyalgia in part because she frequently feels stiff all over.10 She complains of strange tingling and prickling sensations in her hands and feet, especially when anxious.7,9,10 Her headaches also worsened and may be precipitated by bright light, as her pupils are usually dilated.5,7,9 Her hypertension is fairly controlled with medication.6,8-10 Ms. H says she experienced a psychotic episode when she was in her mid-teens,6,8 but reassures you that “she is not that bad now,” although she remains hypervigilant.13 Also while in her teens, Ms. H was treated with paroxetine and experienced restlessness, agitation, delirium, tachycardia, fluctuating blood pressure, diaphoresis, diarrhea, and neuromuscular excitation, which prompted discontinuation of the antidepressant.5-7,9,10
Impression. Ms. H exhibits symptoms associated with serotonin hyperactivity. Discontinuing and avoiding selective serotonin reuptake inhibitors (SSRIs) would be prudent; prescribing an anticonvulsant would be reasonable. Using a GABAergic medication to suppress serotonin (eg, baclofen) is likely to help. Avoiding dopaminergic medications is a must. Antidepressive antipsychotics would be logical to use. The use of serotonin-suppressing medications may be considered. One may argue for the use of beta-blockers in such a patient.
Continue to: Serotonin deficiency
Serotonin deficiency (Table 114,19-29)
Mr. A is chronically depressed, hopeless,19 and easily angered.21 He does not believe anyone can help him.19 You are concerned for his safety because he had attempted to end his life by shooting himself in the chest.19,21,22,25 Even when he’s not particularly depressed, Mr. A does not enjoy much of anything.21,26,27 He becomes particularly agitated when he drinks alcohol,25 which unfortunately is common for him.29 He engages in binge eating to feel better; he knows this is not healthy but he cannot control his behavior.20,29 Mr. A is poorly compliant with his medications, even with a blood thinner, which he was prescribed due to an episode of deep vein thrombosis. He complains of chronic daily headaches and episodic migraines.23,24 He rarely blinks,23,28 his skin is dry and cool, his hair is brittle,23 his mouth is dry,14,23,27 and he constantly licks his chapped lips.14,26,27 Mr. A frequently has general body pain26,31 but is dismissive of his body aches and completely stops reporting pain when his depression gets particularly severe. When depressed, he is slow in movement and thinking.14,21,26,27 He is more concerned with anxiety than depression.21 Mr. A is plagued by constipation, abdominal pain, muscle tension, and episodes of shaking.14,26,27 He also frequently complains about chronic tinnitus.28
Impression. Mr. A shows symptoms associated with serotonin hypoactivity. SSRIs and any other antidepressants with serotonin activity would be an obvious choice for treatment. A mood-stabilizing antipsychotic with serotonin activity would be welcome in treatment. Thyroid hormone supplementation may be of value, especially if thyroid stimulating hormone level is high. Light therapy, a diet with food that contains tryptophan, psychotherapy, and exercise are desirable. Avoiding benzodiazepines would be a good idea.
Dopamine excess (Table 214,30-41)
Ms. L presents with complaints of “fibromyalgia” and “daily headaches,”14 and also dissociation (finding herself in places when she does not know how she got there) and “out-of-body experiences.”32 She is odd, and states that people do not understand her and that she is “different.”38 Her friend, who is present at the appointment, elaborates on Ms. L’s bizarreness and oddness in behavior, out-of-context emotions, suspiciousness, paranoia, and possible hallucinations.35,36,38 Ms. L discloses frequent diffuse body pains, headaches, nausea, excessive salivation, and tongue burning, as well as muscle twitching.14 Sex worsens her headaches and body pain. She reports seizures that are not registered on EEG. In the office, she is suspicious, exhibits odd posturing, tends to misinterpret your words, and makes you feel uncomfortable. Anxiety38 and multiple obsessive-compulsive symptoms, especially excessive cleaning and grooming, complicate Ms. L’s life.31,32,34 On examination, she is hypertensive, and she has scars caused by self-cutting and skin picking on her arms.30-32 An electrocardiogram shows an elevated heart rate, widened QRS complex, and ectopic heartbeats.14 Ms. L has experienced trichotillomania since adolescence32-34 and her fingernails are bitten to the skin.34 She has difficulty with impulse control, and thrill-seeking is a prominent part of her life, mainly via gambling, compulsive sex, and compulsive buying.35,36 She also says she experiences indigestion and delayed gastric emptying.37
Impression. Ms. L exhibits multiple symptoms associated with dopamine excess. Dopamine antagonists should be considered and may help not only with her psychiatric symptoms but also with her pain symptoms. Bupropion (as a dopamine agonist), caffeine, and stimulants should be avoided.
Excessive dopamine is, in extreme cases, associated with somatic psychosis, somatic symptom disorder, factitious disorder, pain disorder, and hypochondria.39 It may come with odd and bizarre/peculiar symptoms out of proportion with objectively identified pathology. These symptoms are common in chronic pain and headache patients, and need to be addressed by appropriate use of dopamine antagonizing medications.39
Continue to: Dopamine deficiency
Dopamine deficiency (Table 24,14,20,38,40-43)
Mr. W experiences widespread pain, including chronic back pain, headaches, and abdominal pain. He also has substantial anhedonia, lack of interest, procrastination, and hypersomnia.41,42 He is apathetic and has difficulty getting up in the morning.41,42 Unusual tiredness and weakness drive him to overuse caffeine; he states that 5 Mountain Dews and 4 cups of regular coffee a day make his headaches bearable.38,41-43 Sex also improves his headaches. Since childhood, he has taken stimulants for attention-deficit/hyperactivity disorder. He reports that occasional use of cocaine helps ease his pain and depression. Mr. W’s wife is concerned with her husband’s low sexual drive and alcohol consumption, and discloses that he has periodic trouble with gambling. Mr. W was forced into psychotherapy but never was able to work productively with his therapist.38,41-43 He loves eating and cannot control his weight.40 This contrasts with episodic anorexia he experienced when he was younger.20 His face is usually emotionless.43 Mr. W is prone to constipation.14 His restless leg syndrome and periodic limb movement disorder are so bad that his wife refuses to share a bed with him.14 He is clumsy and has a problem with repetitive motor tasks.43 A paucity of speech, limited eye contact, poor grooming, and difficulty forming therapeutic alliances have long been part of Mr. W’s history.38,42,43 On physical examination, he has a dry mouth; he is stiff, tremulous, and hypotensive.14
Impression. Mr. W shows multiple symptoms associated with dopamine deficiency. Bupropion may be reasonable to consider. Dopamine augmentation via the use of stimulants is warranted in such patients, especially if stimulants had not been tried before (lisdexamfetamine would be a good choice to minimize addictive potential). For a patient with dopamine deficiency, levodopa may improve more than just restless legs. Amantadine may improve dopaminergic signaling through the accelerated dopamine release and decrease in presynaptic uptake, so this medication may be carefully tried.44 Pain treatment would not be successful for Mr. W without simultaneous treatment for his substance use.
Bottom Line
Both high and low levels of serotonin and dopamine may be associated with certain psychiatric and medical symptoms and disorders. An astute clinician may judge which neurotransmitter is dysfunctional based on the patient’s presentation, and tailor treatment accordingly.
Related Resources
- Abell SR, El-Mallakh RS. Serotonin-mediated anxiety: How to recognize and treat it. Current Psychiatry. 2021;20(11):37-40. doi:10.12788/cp.0168
Drug Brand Names
Amantadine • Gocovri
Baclofen • Ozobax
Bupropion • Wellbutrin
Fluoxetine • Prozac
Lisdexamfetamine • Vyvanse
Paroxetine • Paxil
1. Stahl SM. Dazzled by the dominions of dopamine: clinical roles of D3, D2, and D1 receptors. CNS Spectr. 2017;22(4):305-311.
2. Young RL, Lumsden AL, Martin AM, et al. Augmented capacity for peripheral serotonin release in human obesity. Int J Obes (Lond). 2018;42(11):1880-1889.
3. Ahlman H. Serotonin and carcinoid tumors. J Cardiovasc Pharmacol. 1985;7(Suppl 7):S79-S85.
4. Terry N, Margolis KG. Serotonergic mechanisms regulating the GI tract: experimental evidence and therapeutic relevance. Handb Exp Pharmacol. 2017;239:319-342.
5. Prakash S, Belani P, Trivedi A. Headache as a presenting feature in patients with serotonin syndrome: a case series. Cephalalgia. 2014;34(2):148-153.
6. van Ewijk CE, Jacobs GE, Girbes ARJ. Unsuspected serotonin toxicity in the ICU. Ann Intensive Care. 2016;6(1):85.
7. Pedavally S, Fugate JE, Rabinstein AA. Serotonin syndrome in the intensive care unit: clinical presentations and precipitating medications. Neurocrit Care. 2014;21(1):108-113.
8. Nguyen H, Pan A, Smollin C, et al. An 11-year retrospective review of cyproheptadine use in serotonin syndrome cases reported to the California Poison Control System. J Clin Pharm Ther. 2019;44(2):327-334.
9. Ansari H, Kouti L. Drug interaction and serotonin toxicity with opioid use: another reason to avoid opioids in headache and migraine treatment. Curr Pain Headache Rep. 2016;20(8):50.
10. Ott M, Mannchen JK, Jamshidi F, et al. Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques. Ther Adv Psychopharmacol. 2019;9:2045125318818814. doi:10.1177/2045125318818814
11. Cerrito F, Lazzaro MP, Gaudio E, et al. 5HT2-receptors and serotonin release: their role in human platelet aggregation. Life Sci. 1993;53(3):209-215.
12. Ohayon MM. Pain sensitivity, depression, and sleep deprivation: links with serotoninergic dysfunction. J Psychiatr Res. 2009;43(16):1243-1245.
13. Maron E, Shlik J. Serotonin function in panic disorder: important, but why? Neuropsychopharmacology. 2006;31(1):1-11.
14. Hall JE, Guyton AC. Textbook of Medical Physiology. 12th ed. Spanish version. Elsevier; 2011:120,199,201-204,730-740.
15. Garland EJ, Baerg EA. Amotivational syndrome associated with selective serotonin reuptake inhibitors in children and adolescents. J Child Adolesc Psychopharmacol. 2001;11(2):181-186.
16. George MS, Trimble MR. A fluvoxamine-induced frontal lobe syndrome in a patient with comorbid Gilles de la Tourette’s syndrome and obsessive compulsive disorder. J Clin Psychiatry. 1992;53(10):379-380.
17. Hoehn-Saric R, Harris GJ, Pearlson GD, et al. A fluoxetine-induced frontal lobe syndrome in an obsessive compulsive patient. J Clin Psychiatry. 1991;52(3):131-133.
18. Hoehn-Saric R, Lipsey JR, McLeod DR. Apathy and indifference in patients on fluvoxamine and fluoxetine. J Clin Psychopharmacol. 1990;10(5):343-345.
19. Samuelsson M, Jokinen J, Nordström AL, et al. CSF 5-HIAA, suicide intent and hopelessness in the prediction of early suicide in male high-risk suicide attempters. Acta Psychiatr Scand. 2006;113(1):44-47.
20. Brewerton TD. Clinical Handbook of Eating Disorders: An Integrated Approach. CRC Press; 2004:257-281.
21. Mann JJ, Oquendo M, Underwood MD, et al. The neurobiology of suicide risk: a review for the clinician. J Clin Psychiatry. 1999;60 Suppl 2:7-116.
22. Mann JJ, Malone KM. Cerebrospinal fluid amines and higher-lethality suicide attempts in depressed inpatients. Biol Psychiatry. 1997;41(2):162-171.
23. Joseph R, Welch KM, D’Andrea G. Serotonergic hypofunction in migraine: a synthesis of evidence based on platelet dense body dysfunction. Cephalalgia. 1989;9(4):293-299.
24. Pakalnis A, Splaingard M, Splaingard D, et al. Serotonin effects on sleep and emotional disorders in adolescent migraine. Headache. 2009;49(10):1486-1492.
25. Virkkunen M, Goldman D, Nielsen DA, et al. Low brain serotonin turnover rate (low CSF 5-HIAA) and impulsive violence. J Psychiatry Neurosci. 1995;20(4):271-275.
26. Liu Y, Zhao J, Fan X, et al. Dysfunction in serotonergic and noradrenergic systems and somatic symptoms in psychiatric disorders. Front Psychiatry. 2019;10:286.
27. Ginsburg GS, Riddle MA, Davies M. Somatic symptoms in children and adolescents with anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2006;45(10):1179-1187.
28. O’Malley PG, Jackson JL, Santoro J, et al. Antidepressant therapy for unexplained symptoms and symptom syndromes. J Fam Pract. 1999;48(12):980-990.
29. Fortuna JL. Sweet preference, sugar addiction and the familial history of alcohol dependence: shared neural pathways and genes. J Psychoactive Drugs. 2010;42(2):147-151.
30. Stanley B, Sher L, Wilson S, et al. Non-suicidal self-injurious behavior, endogenous opioids and monoamine neurotransmitters. J Affect Disord. 2010;124(1-2):134-140.
31. Graybiel AM, Saka E. A genetic basis for obsessive grooming. Neuron. 2002;33(1):1-2.
32. Tse W, Hälbig TD. Skin picking in Parkinson’s disease: a behavioral side-effect of dopaminergic treatment? Psychiatry Clin Neurosci. 2010;64(2):214.
33. Ayaydın H. Probable emergence of symptoms of trichotillomania by atomoxetine: a case report. Psychiatry and Clinical Psychopharmacology. 2019;29(2)220-222.
34. Paholpak P, Mendez MF. Trichotillomania as a manifestation of dementia. Case Rep Psychiatry. 2016;2016:9782702. doi:10.1155/2016/9782702
35. Clark CA, Dagher A. The role of dopamine in risk taking: a specific look at Parkinson’s disease and gambling. Front Behav Neurosci. 2014;8:196.
36. Norbury A, Husain M. Sensation-seeking: dopaminergic modulation and risk for psychopathology. Behav Brain Res. 2015;288:79-93.
37. Chen TS, Chang FY. Elevated serum dopamine increases while coffee consumption decreases the occurrence of reddish streaks in the intact stomach. J Gastroenterol Hepatol. 2013;28(12):1810-1814.
38. Wong-Riley MT. Neuroscience Secrets. 1st edition. Spanish version. Hanley & Belfus; 1999:420-429.
39. Arbuck DM. Antipsychotics, dopamine, and pain. Current Psychiatry. 2020;19(1):25-29,31.
40. Bello NT, Hajnal A. Dopamine and binge eating behaviors. Pharmacol Biochem Behav. 2010;97(1):25-33.
41. Velligan DI, Weiden PJ, Sajatovic M, et al; Expert Consensus Panel on Adherence Problems in Serious and Persistent Mental Illness. The expert consensus guideline series: adherence problems in patients with serious and persistent mental illness. J Clin Psychiatry. 2009;70 Suppl 4:1-46.
42. Milev P, Ho BC, Arndt S, et al. Predictive values of neurocognition and negative symptoms on functional outcome in schizophrenia: a longitudinal first-episode study with 7-year follow-up. Am J Psychiatry. 2005;162(3):495-506.
43. Gepshtein S, Li X, Snider J, et al. Dopamine function and the efficiency of human movement. J Cogn Neurosci. 2014;26(3):645-657.
44. Scarff JR. The ABCDs of treating tardive dyskinesia. Current Psychiatry. 2020;19(4):21,55.
1. Stahl SM. Dazzled by the dominions of dopamine: clinical roles of D3, D2, and D1 receptors. CNS Spectr. 2017;22(4):305-311.
2. Young RL, Lumsden AL, Martin AM, et al. Augmented capacity for peripheral serotonin release in human obesity. Int J Obes (Lond). 2018;42(11):1880-1889.
3. Ahlman H. Serotonin and carcinoid tumors. J Cardiovasc Pharmacol. 1985;7(Suppl 7):S79-S85.
4. Terry N, Margolis KG. Serotonergic mechanisms regulating the GI tract: experimental evidence and therapeutic relevance. Handb Exp Pharmacol. 2017;239:319-342.
5. Prakash S, Belani P, Trivedi A. Headache as a presenting feature in patients with serotonin syndrome: a case series. Cephalalgia. 2014;34(2):148-153.
6. van Ewijk CE, Jacobs GE, Girbes ARJ. Unsuspected serotonin toxicity in the ICU. Ann Intensive Care. 2016;6(1):85.
7. Pedavally S, Fugate JE, Rabinstein AA. Serotonin syndrome in the intensive care unit: clinical presentations and precipitating medications. Neurocrit Care. 2014;21(1):108-113.
8. Nguyen H, Pan A, Smollin C, et al. An 11-year retrospective review of cyproheptadine use in serotonin syndrome cases reported to the California Poison Control System. J Clin Pharm Ther. 2019;44(2):327-334.
9. Ansari H, Kouti L. Drug interaction and serotonin toxicity with opioid use: another reason to avoid opioids in headache and migraine treatment. Curr Pain Headache Rep. 2016;20(8):50.
10. Ott M, Mannchen JK, Jamshidi F, et al. Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques. Ther Adv Psychopharmacol. 2019;9:2045125318818814. doi:10.1177/2045125318818814
11. Cerrito F, Lazzaro MP, Gaudio E, et al. 5HT2-receptors and serotonin release: their role in human platelet aggregation. Life Sci. 1993;53(3):209-215.
12. Ohayon MM. Pain sensitivity, depression, and sleep deprivation: links with serotoninergic dysfunction. J Psychiatr Res. 2009;43(16):1243-1245.
13. Maron E, Shlik J. Serotonin function in panic disorder: important, but why? Neuropsychopharmacology. 2006;31(1):1-11.
14. Hall JE, Guyton AC. Textbook of Medical Physiology. 12th ed. Spanish version. Elsevier; 2011:120,199,201-204,730-740.
15. Garland EJ, Baerg EA. Amotivational syndrome associated with selective serotonin reuptake inhibitors in children and adolescents. J Child Adolesc Psychopharmacol. 2001;11(2):181-186.
16. George MS, Trimble MR. A fluvoxamine-induced frontal lobe syndrome in a patient with comorbid Gilles de la Tourette’s syndrome and obsessive compulsive disorder. J Clin Psychiatry. 1992;53(10):379-380.
17. Hoehn-Saric R, Harris GJ, Pearlson GD, et al. A fluoxetine-induced frontal lobe syndrome in an obsessive compulsive patient. J Clin Psychiatry. 1991;52(3):131-133.
18. Hoehn-Saric R, Lipsey JR, McLeod DR. Apathy and indifference in patients on fluvoxamine and fluoxetine. J Clin Psychopharmacol. 1990;10(5):343-345.
19. Samuelsson M, Jokinen J, Nordström AL, et al. CSF 5-HIAA, suicide intent and hopelessness in the prediction of early suicide in male high-risk suicide attempters. Acta Psychiatr Scand. 2006;113(1):44-47.
20. Brewerton TD. Clinical Handbook of Eating Disorders: An Integrated Approach. CRC Press; 2004:257-281.
21. Mann JJ, Oquendo M, Underwood MD, et al. The neurobiology of suicide risk: a review for the clinician. J Clin Psychiatry. 1999;60 Suppl 2:7-116.
22. Mann JJ, Malone KM. Cerebrospinal fluid amines and higher-lethality suicide attempts in depressed inpatients. Biol Psychiatry. 1997;41(2):162-171.
23. Joseph R, Welch KM, D’Andrea G. Serotonergic hypofunction in migraine: a synthesis of evidence based on platelet dense body dysfunction. Cephalalgia. 1989;9(4):293-299.
24. Pakalnis A, Splaingard M, Splaingard D, et al. Serotonin effects on sleep and emotional disorders in adolescent migraine. Headache. 2009;49(10):1486-1492.
25. Virkkunen M, Goldman D, Nielsen DA, et al. Low brain serotonin turnover rate (low CSF 5-HIAA) and impulsive violence. J Psychiatry Neurosci. 1995;20(4):271-275.
26. Liu Y, Zhao J, Fan X, et al. Dysfunction in serotonergic and noradrenergic systems and somatic symptoms in psychiatric disorders. Front Psychiatry. 2019;10:286.
27. Ginsburg GS, Riddle MA, Davies M. Somatic symptoms in children and adolescents with anxiety disorders. J Am Acad Child Adolesc Psychiatry. 2006;45(10):1179-1187.
28. O’Malley PG, Jackson JL, Santoro J, et al. Antidepressant therapy for unexplained symptoms and symptom syndromes. J Fam Pract. 1999;48(12):980-990.
29. Fortuna JL. Sweet preference, sugar addiction and the familial history of alcohol dependence: shared neural pathways and genes. J Psychoactive Drugs. 2010;42(2):147-151.
30. Stanley B, Sher L, Wilson S, et al. Non-suicidal self-injurious behavior, endogenous opioids and monoamine neurotransmitters. J Affect Disord. 2010;124(1-2):134-140.
31. Graybiel AM, Saka E. A genetic basis for obsessive grooming. Neuron. 2002;33(1):1-2.
32. Tse W, Hälbig TD. Skin picking in Parkinson’s disease: a behavioral side-effect of dopaminergic treatment? Psychiatry Clin Neurosci. 2010;64(2):214.
33. Ayaydın H. Probable emergence of symptoms of trichotillomania by atomoxetine: a case report. Psychiatry and Clinical Psychopharmacology. 2019;29(2)220-222.
34. Paholpak P, Mendez MF. Trichotillomania as a manifestation of dementia. Case Rep Psychiatry. 2016;2016:9782702. doi:10.1155/2016/9782702
35. Clark CA, Dagher A. The role of dopamine in risk taking: a specific look at Parkinson’s disease and gambling. Front Behav Neurosci. 2014;8:196.
36. Norbury A, Husain M. Sensation-seeking: dopaminergic modulation and risk for psychopathology. Behav Brain Res. 2015;288:79-93.
37. Chen TS, Chang FY. Elevated serum dopamine increases while coffee consumption decreases the occurrence of reddish streaks in the intact stomach. J Gastroenterol Hepatol. 2013;28(12):1810-1814.
38. Wong-Riley MT. Neuroscience Secrets. 1st edition. Spanish version. Hanley & Belfus; 1999:420-429.
39. Arbuck DM. Antipsychotics, dopamine, and pain. Current Psychiatry. 2020;19(1):25-29,31.
40. Bello NT, Hajnal A. Dopamine and binge eating behaviors. Pharmacol Biochem Behav. 2010;97(1):25-33.
41. Velligan DI, Weiden PJ, Sajatovic M, et al; Expert Consensus Panel on Adherence Problems in Serious and Persistent Mental Illness. The expert consensus guideline series: adherence problems in patients with serious and persistent mental illness. J Clin Psychiatry. 2009;70 Suppl 4:1-46.
42. Milev P, Ho BC, Arndt S, et al. Predictive values of neurocognition and negative symptoms on functional outcome in schizophrenia: a longitudinal first-episode study with 7-year follow-up. Am J Psychiatry. 2005;162(3):495-506.
43. Gepshtein S, Li X, Snider J, et al. Dopamine function and the efficiency of human movement. J Cogn Neurosci. 2014;26(3):645-657.
44. Scarff JR. The ABCDs of treating tardive dyskinesia. Current Psychiatry. 2020;19(4):21,55.
Review of new drugs that may be used during pregnancy
In 2021, the Food and Drug Administration approved 50 new drugs, but 24 will not be described here because they would probably not be used in pregnancy. The 24 are Aduhelm (aducanumab) to treat Alzheimer’s disease; Azstarys (serdexmethylphenidate and dexmethylphenidate), a combination CNS stimulant indicated for the treatment of ADHD; Cabenuva (cabotegravir and rilpivirine) to treat HIV; Voxzogo (vosoritide) for children with achondroplasia and open epiphyses; Qelbree (viloxazine) used in children aged 6-17 years to treat ADHD; and Pylarify (piflufolastat) for prostate cancer. Other anticancer drugs that will not be covered are Cosela (trilaciclib), Cytalux (pafolacianine), Exkivity (mobocertinib); Fotivda (tivozanib), Jemperli (dostarlimab-gxly), Lumakras (sotorasib), Pepaxto (melphalan flufenamide), Rybrevant (amivantamab-vmjw), Rylaze (asparaginase erwinia chrysanthemi), Scemblix (asciminib), Tepmetko (tepotinib), Tivdak (tisotumab vedotin-tftv), Truseltiq (infigratinib), Ukoniq (umbralisib), and Zynlonta (loncastuximab tesirine-lpyl).
Skytrofa (lonapegsomatropin-tcgd) will not be described below because it is indicated to treat short stature and is unlikely to be used in pregnancy. Nextstellis (drospirenone and estetrol) is used to prevent pregnancy.
Typically, for new drugs there will be no published reports describing their use in pregnant women. That information will come much later. In the sections below, the indications, effects on pregnant animals, and the potential for harm of a fetus/embryo are described. However, the relevance of animal data to human pregnancies is not great.
Adbry (tralokinumab) (molecular weight [MW], 147 kilodaltons), is indicated for the treatment of moderate to severe atopic dermatitis in adult patients whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. The drug did not harm fetal monkeys at doses that were 10 times the maximum recommended human dose.
Besremi (ropeginterferon alfa-2b-njft) (MW, 60 kDa) is an interferon alfa-2b indicated for the treatment of adults with polycythemia vera. It is given by subcutaneous injection every 2 weeks. Animal studies assessing reproductive toxicity have not been conducted. The manufacturer states that the drug may cause fetal harm and should be assumed to have abortifacient potential.
Brexafemme (ibrexafungerp) (MW, 922) is indicated for the treatment of vulvovaginal candidiasis. The drug was teratogenic in pregnant rabbits but not in pregnant rats. The manufacturer recommends females with reproductive potential should use effective contraception during treatment and for 4 days after the final dose.
Bylvay (odevixibat) (MW unknown) is indicated for the treatment of pruritus in patients aged 3 months and older. There are no human data regarding its use in pregnant women. The drug was teratogenic in pregnant rabbits. Although there are no data, the drug has low absorption following oral administration and breastfeeding is not expected to result in exposure of the infant.
Empaveli (pegcetacoplan) (MW, 44 kDa) is used to treat paroxysmal nocturnal hemoglobinuria. When the drug was given to pregnant cynomolgus monkeys there was an increase in abortions and stillbirths.
Evkeeza (evinacumab-dgnb) (MW, 146k) is used to treat homozygous familial hypercholesterolemia. The drug was teratogenic in rabbits but not rats.
Fexinidazole (MW not specified) is indicated to treat human African trypanosomiasis caused by the parasite Trypanosoma brucei gambiense. Additional information not available.
Kerendia (finerenone) (MW, 378), is indicated to reduce the risk of kidney and heart complications in chronic kidney disease associated with type 2 diabetes. The drug was teratogenic in rats.
Korsuva (difelikefalin) (MW, 679) is a kappa opioid–receptor agonist indicated for the treatment of moderate to severe pruritus associated with chronic kidney disease in adults undergoing hemodialysis. No adverse effects were observed in pregnant rats and rabbits. The limited human data on use of Korsuva in pregnant women are not sufficient to evaluate a drug associated risk for major birth defects or miscarriage.
Leqvio (inclisiran) (MW, 17,285) is indicated to treat heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease as an add-on therapy. The drug was not teratogenic in rats and rabbits.
Livmarli (maralixibat) (MW, 710) is indicated for the treatment of cholestatic pruritus associated with Alagille syndrome. Because systemic absorption is low, the recommended clinical dose is not expected to result in measurable fetal exposure. No effects on fetal rats were observed.
Livtencity (maribavir) (MW, 376) is used to treat posttransplant cytomegalovirus infection that has not responded to other treatment. Embryo/fetal survival was reduced in rats but not in rabbits at doses less then the human dose.
Lupkynis (voclosporin) (MW, 1,215) is used to treat nephritis. Avoid use of Lupkynis in pregnant women because of the alcohol content of the drug formulation. The drug was embryocidal and feticidal in rats and rabbits but with no treatment-related fetal malformations or variations.
Lybalvi (olanzapine and samidorphan) (MW, 312 and 505) is a combination drug used to treat schizophrenia and bipolar disorder. It was fetal toxic in pregnant rats and rabbits but with no evidence of malformations. There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to atypical antipsychotics, including this drug, during pregnancy. Health care providers are encouraged to register patients by contacting the National Pregnancy Registry for Atypical Antipsychotics at 1-866-961-2388 or visit the Reproductive Psychiatry Resource and Information Center of the MGH Center for Women’s Mental Health.
Nexviazyme (avalglucosidase alfa-ngpt) (MW, 124k) is a hydrolytic lysosomal glycogen-specific enzyme indicated for the treatment of patients aged 1 year and older with late-onset Pompe disease. The drug was not teratogenic in mice and rabbits.
Nulibry (fosdenopterin) (MW, 480) is used to reduce the risk of mortality in molybdenum cofactor deficiency type A. Studies have not been conducted in pregnant animals.
Ponvory (ponesimod) (MW, 461) is used to treat relapsing forms of multiple sclerosis. The drug caused severe adverse effects in pregnant rats and rabbits.
Qulipta (atogepant) (MW, 604) is indicated to prevent episodic migraines. It is embryo/fetal toxic in rats and rabbits.
Saphnelo (anifrolumab-fnia) (MW, 148k) is used to treat moderate to severe systemic lupus erythematosus along with standard therapy. In pregnant cynomolgus monkeys, there was no evidence of embryotoxicity or fetal malformations with exposures up to approximately 28 times the exposure at the maximum recommended human dose.
Tavneos (avacopan) (MW, 582) is indicated to treat severe active antineutrophil cytoplasmic autoantibody–associated vasculitis in combination with standard therapy including glucocorticoids. There appears to be an increased risk for hepatotoxicity. The drug caused no defects in hamsters and rabbits, but in rabbits there was an increase in abortions.
Tezspire (tezepelumab-ekko) (MW, 147k) is indicated to treat severe asthma as an add-on maintenance therapy. No adverse fetal effects were observed in pregnant cynomolgus monkeys.
Verquvo (vericiguat) (MW, 426) is used to mitigate the risk of cardiovascular death and hospitalization for chronic heart failure. The drug was teratogenic in pregnant rabbits but not rats.
Vyvgart (efgartigimod alfa-fcab) (MW, 54k) is indicated to treat generalized myasthenia gravis. The drug did not cause birth defects in rats and rabbits.
Welireg (belzutifan) (MW, 383) is used to treat von Hippel–Lindau disease. In pregnant rats, the drug caused embryo-fetal lethality, reduced fetal body weight, and caused fetal skeletal malformations at maternal exposures of at least 0.2 times the human exposures.
Zegalogue (dasiglucagon) (MW, 3,382) is used to treat severe hypoglycemia. The drug did not cause birth defects in pregnant rats and rabbits.
Breastfeeding
It is not known if the above drugs will be in breast milk, but the safest course for an infant is to not breast feed if the mother is taking any of the above drugs.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at obnews@mdedge.com.
In 2021, the Food and Drug Administration approved 50 new drugs, but 24 will not be described here because they would probably not be used in pregnancy. The 24 are Aduhelm (aducanumab) to treat Alzheimer’s disease; Azstarys (serdexmethylphenidate and dexmethylphenidate), a combination CNS stimulant indicated for the treatment of ADHD; Cabenuva (cabotegravir and rilpivirine) to treat HIV; Voxzogo (vosoritide) for children with achondroplasia and open epiphyses; Qelbree (viloxazine) used in children aged 6-17 years to treat ADHD; and Pylarify (piflufolastat) for prostate cancer. Other anticancer drugs that will not be covered are Cosela (trilaciclib), Cytalux (pafolacianine), Exkivity (mobocertinib); Fotivda (tivozanib), Jemperli (dostarlimab-gxly), Lumakras (sotorasib), Pepaxto (melphalan flufenamide), Rybrevant (amivantamab-vmjw), Rylaze (asparaginase erwinia chrysanthemi), Scemblix (asciminib), Tepmetko (tepotinib), Tivdak (tisotumab vedotin-tftv), Truseltiq (infigratinib), Ukoniq (umbralisib), and Zynlonta (loncastuximab tesirine-lpyl).
Skytrofa (lonapegsomatropin-tcgd) will not be described below because it is indicated to treat short stature and is unlikely to be used in pregnancy. Nextstellis (drospirenone and estetrol) is used to prevent pregnancy.
Typically, for new drugs there will be no published reports describing their use in pregnant women. That information will come much later. In the sections below, the indications, effects on pregnant animals, and the potential for harm of a fetus/embryo are described. However, the relevance of animal data to human pregnancies is not great.
Adbry (tralokinumab) (molecular weight [MW], 147 kilodaltons), is indicated for the treatment of moderate to severe atopic dermatitis in adult patients whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. The drug did not harm fetal monkeys at doses that were 10 times the maximum recommended human dose.
Besremi (ropeginterferon alfa-2b-njft) (MW, 60 kDa) is an interferon alfa-2b indicated for the treatment of adults with polycythemia vera. It is given by subcutaneous injection every 2 weeks. Animal studies assessing reproductive toxicity have not been conducted. The manufacturer states that the drug may cause fetal harm and should be assumed to have abortifacient potential.
Brexafemme (ibrexafungerp) (MW, 922) is indicated for the treatment of vulvovaginal candidiasis. The drug was teratogenic in pregnant rabbits but not in pregnant rats. The manufacturer recommends females with reproductive potential should use effective contraception during treatment and for 4 days after the final dose.
Bylvay (odevixibat) (MW unknown) is indicated for the treatment of pruritus in patients aged 3 months and older. There are no human data regarding its use in pregnant women. The drug was teratogenic in pregnant rabbits. Although there are no data, the drug has low absorption following oral administration and breastfeeding is not expected to result in exposure of the infant.
Empaveli (pegcetacoplan) (MW, 44 kDa) is used to treat paroxysmal nocturnal hemoglobinuria. When the drug was given to pregnant cynomolgus monkeys there was an increase in abortions and stillbirths.
Evkeeza (evinacumab-dgnb) (MW, 146k) is used to treat homozygous familial hypercholesterolemia. The drug was teratogenic in rabbits but not rats.
Fexinidazole (MW not specified) is indicated to treat human African trypanosomiasis caused by the parasite Trypanosoma brucei gambiense. Additional information not available.
Kerendia (finerenone) (MW, 378), is indicated to reduce the risk of kidney and heart complications in chronic kidney disease associated with type 2 diabetes. The drug was teratogenic in rats.
Korsuva (difelikefalin) (MW, 679) is a kappa opioid–receptor agonist indicated for the treatment of moderate to severe pruritus associated with chronic kidney disease in adults undergoing hemodialysis. No adverse effects were observed in pregnant rats and rabbits. The limited human data on use of Korsuva in pregnant women are not sufficient to evaluate a drug associated risk for major birth defects or miscarriage.
Leqvio (inclisiran) (MW, 17,285) is indicated to treat heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease as an add-on therapy. The drug was not teratogenic in rats and rabbits.
Livmarli (maralixibat) (MW, 710) is indicated for the treatment of cholestatic pruritus associated with Alagille syndrome. Because systemic absorption is low, the recommended clinical dose is not expected to result in measurable fetal exposure. No effects on fetal rats were observed.
Livtencity (maribavir) (MW, 376) is used to treat posttransplant cytomegalovirus infection that has not responded to other treatment. Embryo/fetal survival was reduced in rats but not in rabbits at doses less then the human dose.
Lupkynis (voclosporin) (MW, 1,215) is used to treat nephritis. Avoid use of Lupkynis in pregnant women because of the alcohol content of the drug formulation. The drug was embryocidal and feticidal in rats and rabbits but with no treatment-related fetal malformations or variations.
Lybalvi (olanzapine and samidorphan) (MW, 312 and 505) is a combination drug used to treat schizophrenia and bipolar disorder. It was fetal toxic in pregnant rats and rabbits but with no evidence of malformations. There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to atypical antipsychotics, including this drug, during pregnancy. Health care providers are encouraged to register patients by contacting the National Pregnancy Registry for Atypical Antipsychotics at 1-866-961-2388 or visit the Reproductive Psychiatry Resource and Information Center of the MGH Center for Women’s Mental Health.
Nexviazyme (avalglucosidase alfa-ngpt) (MW, 124k) is a hydrolytic lysosomal glycogen-specific enzyme indicated for the treatment of patients aged 1 year and older with late-onset Pompe disease. The drug was not teratogenic in mice and rabbits.
Nulibry (fosdenopterin) (MW, 480) is used to reduce the risk of mortality in molybdenum cofactor deficiency type A. Studies have not been conducted in pregnant animals.
Ponvory (ponesimod) (MW, 461) is used to treat relapsing forms of multiple sclerosis. The drug caused severe adverse effects in pregnant rats and rabbits.
Qulipta (atogepant) (MW, 604) is indicated to prevent episodic migraines. It is embryo/fetal toxic in rats and rabbits.
Saphnelo (anifrolumab-fnia) (MW, 148k) is used to treat moderate to severe systemic lupus erythematosus along with standard therapy. In pregnant cynomolgus monkeys, there was no evidence of embryotoxicity or fetal malformations with exposures up to approximately 28 times the exposure at the maximum recommended human dose.
Tavneos (avacopan) (MW, 582) is indicated to treat severe active antineutrophil cytoplasmic autoantibody–associated vasculitis in combination with standard therapy including glucocorticoids. There appears to be an increased risk for hepatotoxicity. The drug caused no defects in hamsters and rabbits, but in rabbits there was an increase in abortions.
Tezspire (tezepelumab-ekko) (MW, 147k) is indicated to treat severe asthma as an add-on maintenance therapy. No adverse fetal effects were observed in pregnant cynomolgus monkeys.
Verquvo (vericiguat) (MW, 426) is used to mitigate the risk of cardiovascular death and hospitalization for chronic heart failure. The drug was teratogenic in pregnant rabbits but not rats.
Vyvgart (efgartigimod alfa-fcab) (MW, 54k) is indicated to treat generalized myasthenia gravis. The drug did not cause birth defects in rats and rabbits.
Welireg (belzutifan) (MW, 383) is used to treat von Hippel–Lindau disease. In pregnant rats, the drug caused embryo-fetal lethality, reduced fetal body weight, and caused fetal skeletal malformations at maternal exposures of at least 0.2 times the human exposures.
Zegalogue (dasiglucagon) (MW, 3,382) is used to treat severe hypoglycemia. The drug did not cause birth defects in pregnant rats and rabbits.
Breastfeeding
It is not known if the above drugs will be in breast milk, but the safest course for an infant is to not breast feed if the mother is taking any of the above drugs.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at obnews@mdedge.com.
In 2021, the Food and Drug Administration approved 50 new drugs, but 24 will not be described here because they would probably not be used in pregnancy. The 24 are Aduhelm (aducanumab) to treat Alzheimer’s disease; Azstarys (serdexmethylphenidate and dexmethylphenidate), a combination CNS stimulant indicated for the treatment of ADHD; Cabenuva (cabotegravir and rilpivirine) to treat HIV; Voxzogo (vosoritide) for children with achondroplasia and open epiphyses; Qelbree (viloxazine) used in children aged 6-17 years to treat ADHD; and Pylarify (piflufolastat) for prostate cancer. Other anticancer drugs that will not be covered are Cosela (trilaciclib), Cytalux (pafolacianine), Exkivity (mobocertinib); Fotivda (tivozanib), Jemperli (dostarlimab-gxly), Lumakras (sotorasib), Pepaxto (melphalan flufenamide), Rybrevant (amivantamab-vmjw), Rylaze (asparaginase erwinia chrysanthemi), Scemblix (asciminib), Tepmetko (tepotinib), Tivdak (tisotumab vedotin-tftv), Truseltiq (infigratinib), Ukoniq (umbralisib), and Zynlonta (loncastuximab tesirine-lpyl).
Skytrofa (lonapegsomatropin-tcgd) will not be described below because it is indicated to treat short stature and is unlikely to be used in pregnancy. Nextstellis (drospirenone and estetrol) is used to prevent pregnancy.
Typically, for new drugs there will be no published reports describing their use in pregnant women. That information will come much later. In the sections below, the indications, effects on pregnant animals, and the potential for harm of a fetus/embryo are described. However, the relevance of animal data to human pregnancies is not great.
Adbry (tralokinumab) (molecular weight [MW], 147 kilodaltons), is indicated for the treatment of moderate to severe atopic dermatitis in adult patients whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. The drug did not harm fetal monkeys at doses that were 10 times the maximum recommended human dose.
Besremi (ropeginterferon alfa-2b-njft) (MW, 60 kDa) is an interferon alfa-2b indicated for the treatment of adults with polycythemia vera. It is given by subcutaneous injection every 2 weeks. Animal studies assessing reproductive toxicity have not been conducted. The manufacturer states that the drug may cause fetal harm and should be assumed to have abortifacient potential.
Brexafemme (ibrexafungerp) (MW, 922) is indicated for the treatment of vulvovaginal candidiasis. The drug was teratogenic in pregnant rabbits but not in pregnant rats. The manufacturer recommends females with reproductive potential should use effective contraception during treatment and for 4 days after the final dose.
Bylvay (odevixibat) (MW unknown) is indicated for the treatment of pruritus in patients aged 3 months and older. There are no human data regarding its use in pregnant women. The drug was teratogenic in pregnant rabbits. Although there are no data, the drug has low absorption following oral administration and breastfeeding is not expected to result in exposure of the infant.
Empaveli (pegcetacoplan) (MW, 44 kDa) is used to treat paroxysmal nocturnal hemoglobinuria. When the drug was given to pregnant cynomolgus monkeys there was an increase in abortions and stillbirths.
Evkeeza (evinacumab-dgnb) (MW, 146k) is used to treat homozygous familial hypercholesterolemia. The drug was teratogenic in rabbits but not rats.
Fexinidazole (MW not specified) is indicated to treat human African trypanosomiasis caused by the parasite Trypanosoma brucei gambiense. Additional information not available.
Kerendia (finerenone) (MW, 378), is indicated to reduce the risk of kidney and heart complications in chronic kidney disease associated with type 2 diabetes. The drug was teratogenic in rats.
Korsuva (difelikefalin) (MW, 679) is a kappa opioid–receptor agonist indicated for the treatment of moderate to severe pruritus associated with chronic kidney disease in adults undergoing hemodialysis. No adverse effects were observed in pregnant rats and rabbits. The limited human data on use of Korsuva in pregnant women are not sufficient to evaluate a drug associated risk for major birth defects or miscarriage.
Leqvio (inclisiran) (MW, 17,285) is indicated to treat heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease as an add-on therapy. The drug was not teratogenic in rats and rabbits.
Livmarli (maralixibat) (MW, 710) is indicated for the treatment of cholestatic pruritus associated with Alagille syndrome. Because systemic absorption is low, the recommended clinical dose is not expected to result in measurable fetal exposure. No effects on fetal rats were observed.
Livtencity (maribavir) (MW, 376) is used to treat posttransplant cytomegalovirus infection that has not responded to other treatment. Embryo/fetal survival was reduced in rats but not in rabbits at doses less then the human dose.
Lupkynis (voclosporin) (MW, 1,215) is used to treat nephritis. Avoid use of Lupkynis in pregnant women because of the alcohol content of the drug formulation. The drug was embryocidal and feticidal in rats and rabbits but with no treatment-related fetal malformations or variations.
Lybalvi (olanzapine and samidorphan) (MW, 312 and 505) is a combination drug used to treat schizophrenia and bipolar disorder. It was fetal toxic in pregnant rats and rabbits but with no evidence of malformations. There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to atypical antipsychotics, including this drug, during pregnancy. Health care providers are encouraged to register patients by contacting the National Pregnancy Registry for Atypical Antipsychotics at 1-866-961-2388 or visit the Reproductive Psychiatry Resource and Information Center of the MGH Center for Women’s Mental Health.
Nexviazyme (avalglucosidase alfa-ngpt) (MW, 124k) is a hydrolytic lysosomal glycogen-specific enzyme indicated for the treatment of patients aged 1 year and older with late-onset Pompe disease. The drug was not teratogenic in mice and rabbits.
Nulibry (fosdenopterin) (MW, 480) is used to reduce the risk of mortality in molybdenum cofactor deficiency type A. Studies have not been conducted in pregnant animals.
Ponvory (ponesimod) (MW, 461) is used to treat relapsing forms of multiple sclerosis. The drug caused severe adverse effects in pregnant rats and rabbits.
Qulipta (atogepant) (MW, 604) is indicated to prevent episodic migraines. It is embryo/fetal toxic in rats and rabbits.
Saphnelo (anifrolumab-fnia) (MW, 148k) is used to treat moderate to severe systemic lupus erythematosus along with standard therapy. In pregnant cynomolgus monkeys, there was no evidence of embryotoxicity or fetal malformations with exposures up to approximately 28 times the exposure at the maximum recommended human dose.
Tavneos (avacopan) (MW, 582) is indicated to treat severe active antineutrophil cytoplasmic autoantibody–associated vasculitis in combination with standard therapy including glucocorticoids. There appears to be an increased risk for hepatotoxicity. The drug caused no defects in hamsters and rabbits, but in rabbits there was an increase in abortions.
Tezspire (tezepelumab-ekko) (MW, 147k) is indicated to treat severe asthma as an add-on maintenance therapy. No adverse fetal effects were observed in pregnant cynomolgus monkeys.
Verquvo (vericiguat) (MW, 426) is used to mitigate the risk of cardiovascular death and hospitalization for chronic heart failure. The drug was teratogenic in pregnant rabbits but not rats.
Vyvgart (efgartigimod alfa-fcab) (MW, 54k) is indicated to treat generalized myasthenia gravis. The drug did not cause birth defects in rats and rabbits.
Welireg (belzutifan) (MW, 383) is used to treat von Hippel–Lindau disease. In pregnant rats, the drug caused embryo-fetal lethality, reduced fetal body weight, and caused fetal skeletal malformations at maternal exposures of at least 0.2 times the human exposures.
Zegalogue (dasiglucagon) (MW, 3,382) is used to treat severe hypoglycemia. The drug did not cause birth defects in pregnant rats and rabbits.
Breastfeeding
It is not known if the above drugs will be in breast milk, but the safest course for an infant is to not breast feed if the mother is taking any of the above drugs.
Mr. Briggs is clinical professor of pharmacy at the University of California, San Francisco, and adjunct professor of pharmacy at the University of Southern California, Los Angeles, as well as at Washington State University, Spokane. Mr. Briggs said he had no relevant financial disclosures. Email him at obnews@mdedge.com.