User login
Reassessing benzodiazepines: What role should this medication class play in psychiatry?
Many psychiatrists have had the grim experience of a newly referred patient explaining that her (and it is most often “her”) primary care doctor has been prescribing lorazepam 8 mg per day or alprazolam 6 mg per day and is sending her to you for help with ongoing anxiety. For conscientious psychiatrists, this means the beginning of a long tapering process along with a great deal of reassuring of a patient who is terrified of feeling overwhelmed with anxiety. The same problem occurs with patients taking large doses of sedatives who are still unable to sleep.
Mark Olfson and coauthors quantified benzodiazepine use in the United States in 2008 using a large prescription database, and found that 5.2% of adults between 18 and 80 years old were taking these drugs.1 The percentage increased with age, to 8.7% of those 65-80 years, in whom 31% received long-term prescriptions from a psychiatrist. Benzodiazepine use was twice as prevalent in women, compared with men. This occurs despite peer-reviewed publications and articles in the popular press regarding the risks of long-term benzodiazepine use in the elderly. Fang-Yu Lin and coauthors documented a 2.23-fold higher risk of hip fracture in zolpidem users that increased with age; elderly users had a 21-fold higher incidence of fracture, compared with younger users, and were twice as likely to sustain a fracture than elderly nonusers.2
Rashona Thomas and Edid Ramos-Rivas reviewed the risks of benzodiazepines in older patients with insomnia and document the increase in serious adverse events such as falls, fractures, and cognitive and behavioral changes.3 Many patients have ongoing prescriptions that make discontinuation difficult, given the potential for withdrawal agitation, seizures, insomnia, nightmares and even psychosis.
Greta Bushnell and coauthors pointed to the problem of simultaneous prescribing of a new antidepressant with a benzodiazepine by 10% of doctors initiating antidepressants.4 Over 12% of this group of patients continued benzodiazepines long term, even though there was no difference in the response to antidepressant treatment at 6 months. Those with long-term benzodiazepine use were also more likely to have recent prescriptions for opiates.
A Finnish research team found that 34% of middle-aged and 55% of elderly people developed long-term use of benzodiazepines after an initial prescription.5 Those who became long-term users were more often older male receivers of social benefits, with psychiatric comorbidities and substance abuse histories.
Kevin Xu and coauthors reviewed a National Health and Nutrition Examination Survey dataset from 1999 to 2015 with follow-up on over 5,000 individuals in that period.6 They found doubling of all-cause mortality in users of benzodiazepines with or without accompanying use of opiates, a statistically significant increase.
Perhaps most alarming is the increased risk for Alzheimer’s dementia diagnosis in users of benzodiazepines. Two separate studies (Billoti de Gage and colleagues and Ettcheto and colleagues7,8) provided reviews of evidence for the relationship between use of benzodiazepines and development of dementia, and repeated warnings about close monitoring of patients and the need for alternative treatments for anxiety and insomnia in the elderly.
Be alert to underlying issues
Overburdened primary practitioners faced with complaints about sleep and anxiety understandably turn to medication rather than taking time to discuss the reasons for these problems or to describe nonmedication approaches to relief of symptoms. Even insured patients may have very limited options for “covered” psychiatric consultation, as many competent psychiatrists have moved to a cash-only system. It is easier to renew prescriptions than to counsel patients or refer them, and many primary care practitioners have limited experience with diagnosing causes of anxiety and insomnia, much less alternative medication approaches.
Psychiatrists should be aware of the frequency of underlying mood disorders that include sleep and anxiety as prominent symptoms; in fact, these symptoms are often what motivates patients to pursue treatment. It is critical to obtain not only a personal history of symptoms beginning in childhood up to the present, but also a family history of mood and anxiety problems. Mood dysregulation disorders are highly hereditary and a family history of mania or psychosis should raise concern about the cause of symptoms in one’s patient. A strong personal and/or family history of alcohol abuse and dependence may cover underlying undiagnosed mood dysregulation. Primary care physicians may not recognize mood dysregulation unless a patient is clearly manic or psychotic.
There is a cohort of patients who do well on antidepressant medication, but anorgasmia, fatigue, and emotional blunting are common side effects that affect compliance. When patients have unexpected responses to SSRI medications such as euphoria, agitation, anxiety, insomnia, and more prominent mood swings, primary care physicians may add a benzodiazepine, expecting the problem to abate with time. Unfortunately, this often leads to ongoing use of benzodiazepines, since attempts to stop them causes withdrawal effects that are indistinguishable from the original anxiety symptoms.
Most psychiatrists are aware that some patients need mood stabilization rather than mood elevation to maintain an adequate baseline mood. Lithium, anticonvulsants, and second-generation antipsychotics may be effective without adding antidepressant medication. Managing dosing and side effects requires time for follow-up visits with patients after initiating treatment but leads to more stability and better outcomes.
Benzodiazepines are appropriate and helpful in situations that cause transient anxiety and with patients who have done poorly with other options. Intermittent use is key to avoiding tolerance and inevitable dose increases. Some individuals can take low daily doses that are harmless, though these likely only prevent withdrawal rather than preventing anxiety. The placebo effect of taking a pill is powerful. And some patients take more doses than they admit to. Most practitioners have heard stories about the alprazolam that was accidentally spilled into the sink or the prescription bottle of diazepam that was lost or the lorazepam supply that was stolen by the babysitter.
These concepts are illustrated in case examples below.
Case one
Ms. A, a 55-year-old married female business administrator, admitted to using zolpidem at 40 mg per night for the past several months. She began with the typical dose of 10 mg at bedtime prescribed by her internist, but after several weeks, needed an additional 10 mg at 2 a.m. to stay asleep. As weeks passed, she found that she needed an additional 20 mg when she awoke at 2 a.m. Within months, she needed 20 mg to initiate sleep and 20 mg to maintain sleep. She obtained extra zolpidem from her gynecologist and came for consultation when refill requests were refused.
Ms. A had a family history of high anxiety in her mother and depressed mood in multiple paternal relatives, including her father. She had trouble sleeping beginning in adolescence, significant premenstrual dysphoria, and postpartum depression that led to a prescription for sertraline. Instead of feeling better, Ms. A remembers being agitated and unable to sleep, so she stopped it. Ms. A was now perimenopausal, and insomnia was worse. She had gradually increased wine consumption to a bottle of wine each night after work to “settle down.” This allowed her to fall asleep, but she inevitably awoke within 4 hours. Her internist noted an elevation in ALT and asked Ms. A about alcohol consumption. She was alarmed and cut back to one glass of wine per night but again couldn’t sleep. Her internist started zolpidem at that point.
The psychiatrist explained the concepts of tolerance and addiction and a plan to slowly taper off zolpidem while using quetiapine for sleep. She decreased to 20 mg of zolpidem at bedtime with quetiapine 50 mg and was able to stay asleep. After 3 weeks, Ms. A took zolpidem 10 mg at bedtime with quetiapine 75 mg and again, was able to fall asleep and stay asleep. After another 3 weeks, she increased quetiapine to 100 mg and stopped zolpidem without difficulty. This dose of quetiapine has continued to work well without significant side effects.
Case two
Ms. B, a 70-year-old married housewife, was referred for help with longstanding anxiety when her primary care doctor recognized that lorazepam, initially helpful at 1 mg twice daily, had required titration to 2 mg three times daily. Ms. B was preoccupied with having lorazepam on hand and never missed a dose. She had little interest in activities beyond her home, rarely socialized, and had fallen twice. She napped for 2 hours each afternoon, and sometimes had trouble staying asleep through the night.
Ms. B was reluctant to talk about her childhood history of hostility and undermining by her mother, who clearly preferred her older brother and was competitive with Ms. B. Her father traveled for work during the week and had little time for her. Ms. B had always seen herself as stupid and unlovable, which interfered with making friends. She attended college for 1 year but dropped out to marry her husband. He was also anxious and had difficulty socializing, but they found reassurance in each other. Their only child, a son in his 40s, was estranged from them, having married a woman who disliked Ms. B. Ms. B felt hopeless about developing a relationship with her grandchildren who were rarely allowed to visit. Despite her initial shame in talking about these painful problems, Ms. B realized that she felt better and scheduled monthly visits to check in.
Ms. B understood the risks of using lorazepam and wanted to stop it but was terrified of becoming anxious again. We set up a very slow tapering schedule that lowered her total dose by 0.5 mg every 2 weeks. At the same time, she began escitalopram which was effective at 20 mg. Ms. B noted that she no longer felt anxious upon awakening but was still afraid to miss a dose of lorazepam. As she felt more confident and alert, Ms. B joined a painting class at a local community center and was gratified to find that she was good at working with watercolors. She invited her neighbors to come for dinner and was surprised at how friendly and open they were. Once she had tapered to 1 mg twice daily, Ms. B began walking for exercise as she now had enough energy that it felt good to move around. After 6 months, she was completely off lorazepam, and very grateful to have discovered her capacity to improve her pleasure in life.
Case three
Ms. C, a 48-year-old attorney was referred for help with anxiety and distress in the face of separation from her husband who had admitted to an affair after she heard him talking to his girlfriend from their basement. She was unsure whether she wanted to save the marriage or end it and was horrified at the thought of dating. She had never felt especially anxious or depressed and had a supportive circle of close friends. She was uncharacteristically unable to concentrate long enough to consider her options because of anxiety.
A dose of clonazepam 0.5 mg allowed her to stay alert but calm enough to reflect on her feelings. She used it intermittently over several months and maintained regular individual psychotherapy sessions that allowed her to review the situation thoroughly. On her psychiatrist’s recommendation, she contacted a colleague to represent her if she decided to initiate divorce proceedings. She attempted to engage her husband in marital therapy, and his reluctance made it clear to her that she could no longer trust him. Ms. C offered him the option of a dissolution if he was willing to cooperate, or to sue for divorce if not. Once Ms. C regained her confidence and recognized that she would survive this emotionally fraught situation, she no longer needed clonazepam.
Summary
The risks, which include cognitive slowing, falls and fractures, and withdrawal phenomena when abruptly stopped, make this class dangerous for all patients but particularly the elderly. Benzodiazepines are nonetheless useful medications for patients able to use them intermittently, whether on an alternating basis with other medications (for example, quetiapine alternating with clonazepam for chronic insomnia) or because symptoms of anxiety are intermittent. Psychiatrists treating tolerant patients should be familiar with the approach of tapering slowly while introducing more appropriate medications at adequate doses to manage symptoms.
Dr. Kaplan is training and supervising psychoanalyst at the Cincinnati Psychoanalytic Institute and volunteer professor of clinical psychiatry at the University of Cincinnati. The author reported no financial relationships with any companies whose products are mentioned in this article, or with manufacturers of competing products.
References
1. Olfson M et al. JAMA Psychiatry. 2015 Feb;72(2):136-42. doi: 10.1001/jamapsychiatry.2014.1763.
2. Lin FY et al. Sleep. 2014 Apr 1;37(4):673-9. doi: 10.5665/sleep.3566.
3. Thomas R and Ramos-Rivas E. Psychiatr Ann. 2018;48(6):266-70. doi: 10.3928/00485713-20180513-01.
4. Bushnell GA et al. JAMA Psychiatry. 2017 Jul 1;74(7):747-55. doi: 10.1001/jamapsychiatry.2017.1273.
5. Taipale H et al. JAMA Netw Open. 2020;3(10):e2019029. doi: 10.1001/jamanetworkopen.2020.19029.
6. Xu KY et al. JAMA Netw Open. 2020;3(12):e2028557. doi: 10.1001/jamanetworkopen.2020.28557.
7. Billioti de Gage S et al. BMJ. 2014;349:g5205. doi: 10.1136/bmj.g5205.
8. Ettcheto M et al. Front Aging Neurosci. 2020 Jan 8;11:344. doi: 10.3389/fnagi.2019.00344.
Many psychiatrists have had the grim experience of a newly referred patient explaining that her (and it is most often “her”) primary care doctor has been prescribing lorazepam 8 mg per day or alprazolam 6 mg per day and is sending her to you for help with ongoing anxiety. For conscientious psychiatrists, this means the beginning of a long tapering process along with a great deal of reassuring of a patient who is terrified of feeling overwhelmed with anxiety. The same problem occurs with patients taking large doses of sedatives who are still unable to sleep.
Mark Olfson and coauthors quantified benzodiazepine use in the United States in 2008 using a large prescription database, and found that 5.2% of adults between 18 and 80 years old were taking these drugs.1 The percentage increased with age, to 8.7% of those 65-80 years, in whom 31% received long-term prescriptions from a psychiatrist. Benzodiazepine use was twice as prevalent in women, compared with men. This occurs despite peer-reviewed publications and articles in the popular press regarding the risks of long-term benzodiazepine use in the elderly. Fang-Yu Lin and coauthors documented a 2.23-fold higher risk of hip fracture in zolpidem users that increased with age; elderly users had a 21-fold higher incidence of fracture, compared with younger users, and were twice as likely to sustain a fracture than elderly nonusers.2
Rashona Thomas and Edid Ramos-Rivas reviewed the risks of benzodiazepines in older patients with insomnia and document the increase in serious adverse events such as falls, fractures, and cognitive and behavioral changes.3 Many patients have ongoing prescriptions that make discontinuation difficult, given the potential for withdrawal agitation, seizures, insomnia, nightmares and even psychosis.
Greta Bushnell and coauthors pointed to the problem of simultaneous prescribing of a new antidepressant with a benzodiazepine by 10% of doctors initiating antidepressants.4 Over 12% of this group of patients continued benzodiazepines long term, even though there was no difference in the response to antidepressant treatment at 6 months. Those with long-term benzodiazepine use were also more likely to have recent prescriptions for opiates.
A Finnish research team found that 34% of middle-aged and 55% of elderly people developed long-term use of benzodiazepines after an initial prescription.5 Those who became long-term users were more often older male receivers of social benefits, with psychiatric comorbidities and substance abuse histories.
Kevin Xu and coauthors reviewed a National Health and Nutrition Examination Survey dataset from 1999 to 2015 with follow-up on over 5,000 individuals in that period.6 They found doubling of all-cause mortality in users of benzodiazepines with or without accompanying use of opiates, a statistically significant increase.
Perhaps most alarming is the increased risk for Alzheimer’s dementia diagnosis in users of benzodiazepines. Two separate studies (Billoti de Gage and colleagues and Ettcheto and colleagues7,8) provided reviews of evidence for the relationship between use of benzodiazepines and development of dementia, and repeated warnings about close monitoring of patients and the need for alternative treatments for anxiety and insomnia in the elderly.
Be alert to underlying issues
Overburdened primary practitioners faced with complaints about sleep and anxiety understandably turn to medication rather than taking time to discuss the reasons for these problems or to describe nonmedication approaches to relief of symptoms. Even insured patients may have very limited options for “covered” psychiatric consultation, as many competent psychiatrists have moved to a cash-only system. It is easier to renew prescriptions than to counsel patients or refer them, and many primary care practitioners have limited experience with diagnosing causes of anxiety and insomnia, much less alternative medication approaches.
Psychiatrists should be aware of the frequency of underlying mood disorders that include sleep and anxiety as prominent symptoms; in fact, these symptoms are often what motivates patients to pursue treatment. It is critical to obtain not only a personal history of symptoms beginning in childhood up to the present, but also a family history of mood and anxiety problems. Mood dysregulation disorders are highly hereditary and a family history of mania or psychosis should raise concern about the cause of symptoms in one’s patient. A strong personal and/or family history of alcohol abuse and dependence may cover underlying undiagnosed mood dysregulation. Primary care physicians may not recognize mood dysregulation unless a patient is clearly manic or psychotic.
There is a cohort of patients who do well on antidepressant medication, but anorgasmia, fatigue, and emotional blunting are common side effects that affect compliance. When patients have unexpected responses to SSRI medications such as euphoria, agitation, anxiety, insomnia, and more prominent mood swings, primary care physicians may add a benzodiazepine, expecting the problem to abate with time. Unfortunately, this often leads to ongoing use of benzodiazepines, since attempts to stop them causes withdrawal effects that are indistinguishable from the original anxiety symptoms.
Most psychiatrists are aware that some patients need mood stabilization rather than mood elevation to maintain an adequate baseline mood. Lithium, anticonvulsants, and second-generation antipsychotics may be effective without adding antidepressant medication. Managing dosing and side effects requires time for follow-up visits with patients after initiating treatment but leads to more stability and better outcomes.
Benzodiazepines are appropriate and helpful in situations that cause transient anxiety and with patients who have done poorly with other options. Intermittent use is key to avoiding tolerance and inevitable dose increases. Some individuals can take low daily doses that are harmless, though these likely only prevent withdrawal rather than preventing anxiety. The placebo effect of taking a pill is powerful. And some patients take more doses than they admit to. Most practitioners have heard stories about the alprazolam that was accidentally spilled into the sink or the prescription bottle of diazepam that was lost or the lorazepam supply that was stolen by the babysitter.
These concepts are illustrated in case examples below.
Case one
Ms. A, a 55-year-old married female business administrator, admitted to using zolpidem at 40 mg per night for the past several months. She began with the typical dose of 10 mg at bedtime prescribed by her internist, but after several weeks, needed an additional 10 mg at 2 a.m. to stay asleep. As weeks passed, she found that she needed an additional 20 mg when she awoke at 2 a.m. Within months, she needed 20 mg to initiate sleep and 20 mg to maintain sleep. She obtained extra zolpidem from her gynecologist and came for consultation when refill requests were refused.
Ms. A had a family history of high anxiety in her mother and depressed mood in multiple paternal relatives, including her father. She had trouble sleeping beginning in adolescence, significant premenstrual dysphoria, and postpartum depression that led to a prescription for sertraline. Instead of feeling better, Ms. A remembers being agitated and unable to sleep, so she stopped it. Ms. A was now perimenopausal, and insomnia was worse. She had gradually increased wine consumption to a bottle of wine each night after work to “settle down.” This allowed her to fall asleep, but she inevitably awoke within 4 hours. Her internist noted an elevation in ALT and asked Ms. A about alcohol consumption. She was alarmed and cut back to one glass of wine per night but again couldn’t sleep. Her internist started zolpidem at that point.
The psychiatrist explained the concepts of tolerance and addiction and a plan to slowly taper off zolpidem while using quetiapine for sleep. She decreased to 20 mg of zolpidem at bedtime with quetiapine 50 mg and was able to stay asleep. After 3 weeks, Ms. A took zolpidem 10 mg at bedtime with quetiapine 75 mg and again, was able to fall asleep and stay asleep. After another 3 weeks, she increased quetiapine to 100 mg and stopped zolpidem without difficulty. This dose of quetiapine has continued to work well without significant side effects.
Case two
Ms. B, a 70-year-old married housewife, was referred for help with longstanding anxiety when her primary care doctor recognized that lorazepam, initially helpful at 1 mg twice daily, had required titration to 2 mg three times daily. Ms. B was preoccupied with having lorazepam on hand and never missed a dose. She had little interest in activities beyond her home, rarely socialized, and had fallen twice. She napped for 2 hours each afternoon, and sometimes had trouble staying asleep through the night.
Ms. B was reluctant to talk about her childhood history of hostility and undermining by her mother, who clearly preferred her older brother and was competitive with Ms. B. Her father traveled for work during the week and had little time for her. Ms. B had always seen herself as stupid and unlovable, which interfered with making friends. She attended college for 1 year but dropped out to marry her husband. He was also anxious and had difficulty socializing, but they found reassurance in each other. Their only child, a son in his 40s, was estranged from them, having married a woman who disliked Ms. B. Ms. B felt hopeless about developing a relationship with her grandchildren who were rarely allowed to visit. Despite her initial shame in talking about these painful problems, Ms. B realized that she felt better and scheduled monthly visits to check in.
Ms. B understood the risks of using lorazepam and wanted to stop it but was terrified of becoming anxious again. We set up a very slow tapering schedule that lowered her total dose by 0.5 mg every 2 weeks. At the same time, she began escitalopram which was effective at 20 mg. Ms. B noted that she no longer felt anxious upon awakening but was still afraid to miss a dose of lorazepam. As she felt more confident and alert, Ms. B joined a painting class at a local community center and was gratified to find that she was good at working with watercolors. She invited her neighbors to come for dinner and was surprised at how friendly and open they were. Once she had tapered to 1 mg twice daily, Ms. B began walking for exercise as she now had enough energy that it felt good to move around. After 6 months, she was completely off lorazepam, and very grateful to have discovered her capacity to improve her pleasure in life.
Case three
Ms. C, a 48-year-old attorney was referred for help with anxiety and distress in the face of separation from her husband who had admitted to an affair after she heard him talking to his girlfriend from their basement. She was unsure whether she wanted to save the marriage or end it and was horrified at the thought of dating. She had never felt especially anxious or depressed and had a supportive circle of close friends. She was uncharacteristically unable to concentrate long enough to consider her options because of anxiety.
A dose of clonazepam 0.5 mg allowed her to stay alert but calm enough to reflect on her feelings. She used it intermittently over several months and maintained regular individual psychotherapy sessions that allowed her to review the situation thoroughly. On her psychiatrist’s recommendation, she contacted a colleague to represent her if she decided to initiate divorce proceedings. She attempted to engage her husband in marital therapy, and his reluctance made it clear to her that she could no longer trust him. Ms. C offered him the option of a dissolution if he was willing to cooperate, or to sue for divorce if not. Once Ms. C regained her confidence and recognized that she would survive this emotionally fraught situation, she no longer needed clonazepam.
Summary
The risks, which include cognitive slowing, falls and fractures, and withdrawal phenomena when abruptly stopped, make this class dangerous for all patients but particularly the elderly. Benzodiazepines are nonetheless useful medications for patients able to use them intermittently, whether on an alternating basis with other medications (for example, quetiapine alternating with clonazepam for chronic insomnia) or because symptoms of anxiety are intermittent. Psychiatrists treating tolerant patients should be familiar with the approach of tapering slowly while introducing more appropriate medications at adequate doses to manage symptoms.
Dr. Kaplan is training and supervising psychoanalyst at the Cincinnati Psychoanalytic Institute and volunteer professor of clinical psychiatry at the University of Cincinnati. The author reported no financial relationships with any companies whose products are mentioned in this article, or with manufacturers of competing products.
References
1. Olfson M et al. JAMA Psychiatry. 2015 Feb;72(2):136-42. doi: 10.1001/jamapsychiatry.2014.1763.
2. Lin FY et al. Sleep. 2014 Apr 1;37(4):673-9. doi: 10.5665/sleep.3566.
3. Thomas R and Ramos-Rivas E. Psychiatr Ann. 2018;48(6):266-70. doi: 10.3928/00485713-20180513-01.
4. Bushnell GA et al. JAMA Psychiatry. 2017 Jul 1;74(7):747-55. doi: 10.1001/jamapsychiatry.2017.1273.
5. Taipale H et al. JAMA Netw Open. 2020;3(10):e2019029. doi: 10.1001/jamanetworkopen.2020.19029.
6. Xu KY et al. JAMA Netw Open. 2020;3(12):e2028557. doi: 10.1001/jamanetworkopen.2020.28557.
7. Billioti de Gage S et al. BMJ. 2014;349:g5205. doi: 10.1136/bmj.g5205.
8. Ettcheto M et al. Front Aging Neurosci. 2020 Jan 8;11:344. doi: 10.3389/fnagi.2019.00344.
Many psychiatrists have had the grim experience of a newly referred patient explaining that her (and it is most often “her”) primary care doctor has been prescribing lorazepam 8 mg per day or alprazolam 6 mg per day and is sending her to you for help with ongoing anxiety. For conscientious psychiatrists, this means the beginning of a long tapering process along with a great deal of reassuring of a patient who is terrified of feeling overwhelmed with anxiety. The same problem occurs with patients taking large doses of sedatives who are still unable to sleep.
Mark Olfson and coauthors quantified benzodiazepine use in the United States in 2008 using a large prescription database, and found that 5.2% of adults between 18 and 80 years old were taking these drugs.1 The percentage increased with age, to 8.7% of those 65-80 years, in whom 31% received long-term prescriptions from a psychiatrist. Benzodiazepine use was twice as prevalent in women, compared with men. This occurs despite peer-reviewed publications and articles in the popular press regarding the risks of long-term benzodiazepine use in the elderly. Fang-Yu Lin and coauthors documented a 2.23-fold higher risk of hip fracture in zolpidem users that increased with age; elderly users had a 21-fold higher incidence of fracture, compared with younger users, and were twice as likely to sustain a fracture than elderly nonusers.2
Rashona Thomas and Edid Ramos-Rivas reviewed the risks of benzodiazepines in older patients with insomnia and document the increase in serious adverse events such as falls, fractures, and cognitive and behavioral changes.3 Many patients have ongoing prescriptions that make discontinuation difficult, given the potential for withdrawal agitation, seizures, insomnia, nightmares and even psychosis.
Greta Bushnell and coauthors pointed to the problem of simultaneous prescribing of a new antidepressant with a benzodiazepine by 10% of doctors initiating antidepressants.4 Over 12% of this group of patients continued benzodiazepines long term, even though there was no difference in the response to antidepressant treatment at 6 months. Those with long-term benzodiazepine use were also more likely to have recent prescriptions for opiates.
A Finnish research team found that 34% of middle-aged and 55% of elderly people developed long-term use of benzodiazepines after an initial prescription.5 Those who became long-term users were more often older male receivers of social benefits, with psychiatric comorbidities and substance abuse histories.
Kevin Xu and coauthors reviewed a National Health and Nutrition Examination Survey dataset from 1999 to 2015 with follow-up on over 5,000 individuals in that period.6 They found doubling of all-cause mortality in users of benzodiazepines with or without accompanying use of opiates, a statistically significant increase.
Perhaps most alarming is the increased risk for Alzheimer’s dementia diagnosis in users of benzodiazepines. Two separate studies (Billoti de Gage and colleagues and Ettcheto and colleagues7,8) provided reviews of evidence for the relationship between use of benzodiazepines and development of dementia, and repeated warnings about close monitoring of patients and the need for alternative treatments for anxiety and insomnia in the elderly.
Be alert to underlying issues
Overburdened primary practitioners faced with complaints about sleep and anxiety understandably turn to medication rather than taking time to discuss the reasons for these problems or to describe nonmedication approaches to relief of symptoms. Even insured patients may have very limited options for “covered” psychiatric consultation, as many competent psychiatrists have moved to a cash-only system. It is easier to renew prescriptions than to counsel patients or refer them, and many primary care practitioners have limited experience with diagnosing causes of anxiety and insomnia, much less alternative medication approaches.
Psychiatrists should be aware of the frequency of underlying mood disorders that include sleep and anxiety as prominent symptoms; in fact, these symptoms are often what motivates patients to pursue treatment. It is critical to obtain not only a personal history of symptoms beginning in childhood up to the present, but also a family history of mood and anxiety problems. Mood dysregulation disorders are highly hereditary and a family history of mania or psychosis should raise concern about the cause of symptoms in one’s patient. A strong personal and/or family history of alcohol abuse and dependence may cover underlying undiagnosed mood dysregulation. Primary care physicians may not recognize mood dysregulation unless a patient is clearly manic or psychotic.
There is a cohort of patients who do well on antidepressant medication, but anorgasmia, fatigue, and emotional blunting are common side effects that affect compliance. When patients have unexpected responses to SSRI medications such as euphoria, agitation, anxiety, insomnia, and more prominent mood swings, primary care physicians may add a benzodiazepine, expecting the problem to abate with time. Unfortunately, this often leads to ongoing use of benzodiazepines, since attempts to stop them causes withdrawal effects that are indistinguishable from the original anxiety symptoms.
Most psychiatrists are aware that some patients need mood stabilization rather than mood elevation to maintain an adequate baseline mood. Lithium, anticonvulsants, and second-generation antipsychotics may be effective without adding antidepressant medication. Managing dosing and side effects requires time for follow-up visits with patients after initiating treatment but leads to more stability and better outcomes.
Benzodiazepines are appropriate and helpful in situations that cause transient anxiety and with patients who have done poorly with other options. Intermittent use is key to avoiding tolerance and inevitable dose increases. Some individuals can take low daily doses that are harmless, though these likely only prevent withdrawal rather than preventing anxiety. The placebo effect of taking a pill is powerful. And some patients take more doses than they admit to. Most practitioners have heard stories about the alprazolam that was accidentally spilled into the sink or the prescription bottle of diazepam that was lost or the lorazepam supply that was stolen by the babysitter.
These concepts are illustrated in case examples below.
Case one
Ms. A, a 55-year-old married female business administrator, admitted to using zolpidem at 40 mg per night for the past several months. She began with the typical dose of 10 mg at bedtime prescribed by her internist, but after several weeks, needed an additional 10 mg at 2 a.m. to stay asleep. As weeks passed, she found that she needed an additional 20 mg when she awoke at 2 a.m. Within months, she needed 20 mg to initiate sleep and 20 mg to maintain sleep. She obtained extra zolpidem from her gynecologist and came for consultation when refill requests were refused.
Ms. A had a family history of high anxiety in her mother and depressed mood in multiple paternal relatives, including her father. She had trouble sleeping beginning in adolescence, significant premenstrual dysphoria, and postpartum depression that led to a prescription for sertraline. Instead of feeling better, Ms. A remembers being agitated and unable to sleep, so she stopped it. Ms. A was now perimenopausal, and insomnia was worse. She had gradually increased wine consumption to a bottle of wine each night after work to “settle down.” This allowed her to fall asleep, but she inevitably awoke within 4 hours. Her internist noted an elevation in ALT and asked Ms. A about alcohol consumption. She was alarmed and cut back to one glass of wine per night but again couldn’t sleep. Her internist started zolpidem at that point.
The psychiatrist explained the concepts of tolerance and addiction and a plan to slowly taper off zolpidem while using quetiapine for sleep. She decreased to 20 mg of zolpidem at bedtime with quetiapine 50 mg and was able to stay asleep. After 3 weeks, Ms. A took zolpidem 10 mg at bedtime with quetiapine 75 mg and again, was able to fall asleep and stay asleep. After another 3 weeks, she increased quetiapine to 100 mg and stopped zolpidem without difficulty. This dose of quetiapine has continued to work well without significant side effects.
Case two
Ms. B, a 70-year-old married housewife, was referred for help with longstanding anxiety when her primary care doctor recognized that lorazepam, initially helpful at 1 mg twice daily, had required titration to 2 mg three times daily. Ms. B was preoccupied with having lorazepam on hand and never missed a dose. She had little interest in activities beyond her home, rarely socialized, and had fallen twice. She napped for 2 hours each afternoon, and sometimes had trouble staying asleep through the night.
Ms. B was reluctant to talk about her childhood history of hostility and undermining by her mother, who clearly preferred her older brother and was competitive with Ms. B. Her father traveled for work during the week and had little time for her. Ms. B had always seen herself as stupid and unlovable, which interfered with making friends. She attended college for 1 year but dropped out to marry her husband. He was also anxious and had difficulty socializing, but they found reassurance in each other. Their only child, a son in his 40s, was estranged from them, having married a woman who disliked Ms. B. Ms. B felt hopeless about developing a relationship with her grandchildren who were rarely allowed to visit. Despite her initial shame in talking about these painful problems, Ms. B realized that she felt better and scheduled monthly visits to check in.
Ms. B understood the risks of using lorazepam and wanted to stop it but was terrified of becoming anxious again. We set up a very slow tapering schedule that lowered her total dose by 0.5 mg every 2 weeks. At the same time, she began escitalopram which was effective at 20 mg. Ms. B noted that she no longer felt anxious upon awakening but was still afraid to miss a dose of lorazepam. As she felt more confident and alert, Ms. B joined a painting class at a local community center and was gratified to find that she was good at working with watercolors. She invited her neighbors to come for dinner and was surprised at how friendly and open they were. Once she had tapered to 1 mg twice daily, Ms. B began walking for exercise as she now had enough energy that it felt good to move around. After 6 months, she was completely off lorazepam, and very grateful to have discovered her capacity to improve her pleasure in life.
Case three
Ms. C, a 48-year-old attorney was referred for help with anxiety and distress in the face of separation from her husband who had admitted to an affair after she heard him talking to his girlfriend from their basement. She was unsure whether she wanted to save the marriage or end it and was horrified at the thought of dating. She had never felt especially anxious or depressed and had a supportive circle of close friends. She was uncharacteristically unable to concentrate long enough to consider her options because of anxiety.
A dose of clonazepam 0.5 mg allowed her to stay alert but calm enough to reflect on her feelings. She used it intermittently over several months and maintained regular individual psychotherapy sessions that allowed her to review the situation thoroughly. On her psychiatrist’s recommendation, she contacted a colleague to represent her if she decided to initiate divorce proceedings. She attempted to engage her husband in marital therapy, and his reluctance made it clear to her that she could no longer trust him. Ms. C offered him the option of a dissolution if he was willing to cooperate, or to sue for divorce if not. Once Ms. C regained her confidence and recognized that she would survive this emotionally fraught situation, she no longer needed clonazepam.
Summary
The risks, which include cognitive slowing, falls and fractures, and withdrawal phenomena when abruptly stopped, make this class dangerous for all patients but particularly the elderly. Benzodiazepines are nonetheless useful medications for patients able to use them intermittently, whether on an alternating basis with other medications (for example, quetiapine alternating with clonazepam for chronic insomnia) or because symptoms of anxiety are intermittent. Psychiatrists treating tolerant patients should be familiar with the approach of tapering slowly while introducing more appropriate medications at adequate doses to manage symptoms.
Dr. Kaplan is training and supervising psychoanalyst at the Cincinnati Psychoanalytic Institute and volunteer professor of clinical psychiatry at the University of Cincinnati. The author reported no financial relationships with any companies whose products are mentioned in this article, or with manufacturers of competing products.
References
1. Olfson M et al. JAMA Psychiatry. 2015 Feb;72(2):136-42. doi: 10.1001/jamapsychiatry.2014.1763.
2. Lin FY et al. Sleep. 2014 Apr 1;37(4):673-9. doi: 10.5665/sleep.3566.
3. Thomas R and Ramos-Rivas E. Psychiatr Ann. 2018;48(6):266-70. doi: 10.3928/00485713-20180513-01.
4. Bushnell GA et al. JAMA Psychiatry. 2017 Jul 1;74(7):747-55. doi: 10.1001/jamapsychiatry.2017.1273.
5. Taipale H et al. JAMA Netw Open. 2020;3(10):e2019029. doi: 10.1001/jamanetworkopen.2020.19029.
6. Xu KY et al. JAMA Netw Open. 2020;3(12):e2028557. doi: 10.1001/jamanetworkopen.2020.28557.
7. Billioti de Gage S et al. BMJ. 2014;349:g5205. doi: 10.1136/bmj.g5205.
8. Ettcheto M et al. Front Aging Neurosci. 2020 Jan 8;11:344. doi: 10.3389/fnagi.2019.00344.
Burnout and stress of today: How do we cope?
Interestingly, the group that seems to be least impacted by this was health care administrators (with 12% of them planning on leaving their jobs).
I couldn’t stop thinking about these percentages.
I am reminded every day of the commitment and excellence of my colleagues in the health care field, and I do not want to lose them. I am hoping the following information and my thoughts on this topic will be helpful for those thinking about leaving health care.
Surgeon general’s burnout report
The surgeon general recently released a report on addressing health care worker burnout.2 It includes several very interesting and appropriate observations. I will summarize the most important ones here:
1. Our health depends on the well-being of our health workforce.
2. Direct harm to health care workers can lead to anxiety, depression, insomnia, and interpersonal and relationship struggles.
3. Health care workers experience exhaustion from providing overwhelming care and empathy.
4. Health care workers spend less time with patients and too much time with EHRs.
5. There are health workforce shortages.
The report is comprehensive, and everything in it is correct. The real issue is how does it go from being a report to true actionable items that we as health care professionals benefit from? I think in regards to exhaustion from overwhelming care responsibilities, and empathy fatigue, we need better boundaries.
Those who go into medicine, and especially those who go into primary care, always put the patients’ needs first. When operating in a broken system, it stays broken when individuals cover for the deficiencies in the system. Adding four extra patients every day because there is no one to refer them to with availability is injurious to the health care provider, and those providers who accept these additional patients will eventually be part of the 23% who want to leave their jobs. It feels awful to say no, but until the system stops accommodating there will not be substantial change.
The empathy drain
One of the unreported stresses of open access for patients through EHR communications is the empathy drain on physicians. When I see a patient in clinic with chronic symptoms or issues, I spend important time making sure we have a plan and an agreed upon time frame.
With the EHR, patients frequently send multiple messages for the same symptoms between visits. It is okay to redirect the patient and share that these issues will be discussed at length at appointments. My reasoning on this is that I think it is better for me to better care for myself and stay as the doctor for my patients, than always say yes to limitless needs and soon be looking for the off ramp.
The following statistic in the surgeon general’s report really hit home. For every hour of direct patient care, physicians currently spend 2 hours on the EHR system. Most practices allow 10%-20% of time for catch up, where with statistics like this it should be 50%. This concept is fully lost on administrators, or ignored.
It is only when we refuse to continue to accept and follow a broken system that it will change. A minority of internal medicine and family doctors (4.5% in 2018) practice in direct primary care models, where these issues are addressed. Unfortunately, this model as it is currently available is not an option for lower income patients.
A major theme in the surgeon general’s report was that administrative burdens need to be reduced by 75% by 2025. When I look at the report, I see the suggestions, I just don’t see how it will be achieved. Despite almost all clinics moving to the EHR, paperwork in the form of faxes and forms has increased.
A sweeping reform would be needed to eliminate daily faxes from PT offices, visiting nurse services, prior authorization, patients reminders from insurance companies, and disability forms from patients. I am glad that there is acknowledgment of the problem, but this change will take more than 3 years.
Takeaways
So what do we do?
Be good to yourself, and your colleagues. The pandemic has isolated us, which accelerates burnout.
Reach out to people you care about.
We are all feeling this. Set boundaries that allow you to care for yourself, and accept that you are doing your best, even if you can’t meet the needs of all your patients all the time.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Sinsky CA et al. Covid-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021 Dec;5(6):1165-73.
2. Addressing health worker burnout. The U.S. Surgeon General’s advisory on building a thriving health workforce.
Interestingly, the group that seems to be least impacted by this was health care administrators (with 12% of them planning on leaving their jobs).
I couldn’t stop thinking about these percentages.
I am reminded every day of the commitment and excellence of my colleagues in the health care field, and I do not want to lose them. I am hoping the following information and my thoughts on this topic will be helpful for those thinking about leaving health care.
Surgeon general’s burnout report
The surgeon general recently released a report on addressing health care worker burnout.2 It includes several very interesting and appropriate observations. I will summarize the most important ones here:
1. Our health depends on the well-being of our health workforce.
2. Direct harm to health care workers can lead to anxiety, depression, insomnia, and interpersonal and relationship struggles.
3. Health care workers experience exhaustion from providing overwhelming care and empathy.
4. Health care workers spend less time with patients and too much time with EHRs.
5. There are health workforce shortages.
The report is comprehensive, and everything in it is correct. The real issue is how does it go from being a report to true actionable items that we as health care professionals benefit from? I think in regards to exhaustion from overwhelming care responsibilities, and empathy fatigue, we need better boundaries.
Those who go into medicine, and especially those who go into primary care, always put the patients’ needs first. When operating in a broken system, it stays broken when individuals cover for the deficiencies in the system. Adding four extra patients every day because there is no one to refer them to with availability is injurious to the health care provider, and those providers who accept these additional patients will eventually be part of the 23% who want to leave their jobs. It feels awful to say no, but until the system stops accommodating there will not be substantial change.
The empathy drain
One of the unreported stresses of open access for patients through EHR communications is the empathy drain on physicians. When I see a patient in clinic with chronic symptoms or issues, I spend important time making sure we have a plan and an agreed upon time frame.
With the EHR, patients frequently send multiple messages for the same symptoms between visits. It is okay to redirect the patient and share that these issues will be discussed at length at appointments. My reasoning on this is that I think it is better for me to better care for myself and stay as the doctor for my patients, than always say yes to limitless needs and soon be looking for the off ramp.
The following statistic in the surgeon general’s report really hit home. For every hour of direct patient care, physicians currently spend 2 hours on the EHR system. Most practices allow 10%-20% of time for catch up, where with statistics like this it should be 50%. This concept is fully lost on administrators, or ignored.
It is only when we refuse to continue to accept and follow a broken system that it will change. A minority of internal medicine and family doctors (4.5% in 2018) practice in direct primary care models, where these issues are addressed. Unfortunately, this model as it is currently available is not an option for lower income patients.
A major theme in the surgeon general’s report was that administrative burdens need to be reduced by 75% by 2025. When I look at the report, I see the suggestions, I just don’t see how it will be achieved. Despite almost all clinics moving to the EHR, paperwork in the form of faxes and forms has increased.
A sweeping reform would be needed to eliminate daily faxes from PT offices, visiting nurse services, prior authorization, patients reminders from insurance companies, and disability forms from patients. I am glad that there is acknowledgment of the problem, but this change will take more than 3 years.
Takeaways
So what do we do?
Be good to yourself, and your colleagues. The pandemic has isolated us, which accelerates burnout.
Reach out to people you care about.
We are all feeling this. Set boundaries that allow you to care for yourself, and accept that you are doing your best, even if you can’t meet the needs of all your patients all the time.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Sinsky CA et al. Covid-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021 Dec;5(6):1165-73.
2. Addressing health worker burnout. The U.S. Surgeon General’s advisory on building a thriving health workforce.
Interestingly, the group that seems to be least impacted by this was health care administrators (with 12% of them planning on leaving their jobs).
I couldn’t stop thinking about these percentages.
I am reminded every day of the commitment and excellence of my colleagues in the health care field, and I do not want to lose them. I am hoping the following information and my thoughts on this topic will be helpful for those thinking about leaving health care.
Surgeon general’s burnout report
The surgeon general recently released a report on addressing health care worker burnout.2 It includes several very interesting and appropriate observations. I will summarize the most important ones here:
1. Our health depends on the well-being of our health workforce.
2. Direct harm to health care workers can lead to anxiety, depression, insomnia, and interpersonal and relationship struggles.
3. Health care workers experience exhaustion from providing overwhelming care and empathy.
4. Health care workers spend less time with patients and too much time with EHRs.
5. There are health workforce shortages.
The report is comprehensive, and everything in it is correct. The real issue is how does it go from being a report to true actionable items that we as health care professionals benefit from? I think in regards to exhaustion from overwhelming care responsibilities, and empathy fatigue, we need better boundaries.
Those who go into medicine, and especially those who go into primary care, always put the patients’ needs first. When operating in a broken system, it stays broken when individuals cover for the deficiencies in the system. Adding four extra patients every day because there is no one to refer them to with availability is injurious to the health care provider, and those providers who accept these additional patients will eventually be part of the 23% who want to leave their jobs. It feels awful to say no, but until the system stops accommodating there will not be substantial change.
The empathy drain
One of the unreported stresses of open access for patients through EHR communications is the empathy drain on physicians. When I see a patient in clinic with chronic symptoms or issues, I spend important time making sure we have a plan and an agreed upon time frame.
With the EHR, patients frequently send multiple messages for the same symptoms between visits. It is okay to redirect the patient and share that these issues will be discussed at length at appointments. My reasoning on this is that I think it is better for me to better care for myself and stay as the doctor for my patients, than always say yes to limitless needs and soon be looking for the off ramp.
The following statistic in the surgeon general’s report really hit home. For every hour of direct patient care, physicians currently spend 2 hours on the EHR system. Most practices allow 10%-20% of time for catch up, where with statistics like this it should be 50%. This concept is fully lost on administrators, or ignored.
It is only when we refuse to continue to accept and follow a broken system that it will change. A minority of internal medicine and family doctors (4.5% in 2018) practice in direct primary care models, where these issues are addressed. Unfortunately, this model as it is currently available is not an option for lower income patients.
A major theme in the surgeon general’s report was that administrative burdens need to be reduced by 75% by 2025. When I look at the report, I see the suggestions, I just don’t see how it will be achieved. Despite almost all clinics moving to the EHR, paperwork in the form of faxes and forms has increased.
A sweeping reform would be needed to eliminate daily faxes from PT offices, visiting nurse services, prior authorization, patients reminders from insurance companies, and disability forms from patients. I am glad that there is acknowledgment of the problem, but this change will take more than 3 years.
Takeaways
So what do we do?
Be good to yourself, and your colleagues. The pandemic has isolated us, which accelerates burnout.
Reach out to people you care about.
We are all feeling this. Set boundaries that allow you to care for yourself, and accept that you are doing your best, even if you can’t meet the needs of all your patients all the time.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at imnews@mdedge.com.
References
1. Sinsky CA et al. Covid-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021 Dec;5(6):1165-73.
2. Addressing health worker burnout. The U.S. Surgeon General’s advisory on building a thriving health workforce.
The Best of DDW 2022: Feel the history
“The Best of DDW” elicits in the minds of most readers a compilation of the most important clinical and scientific content presented at DDW.
But I am not referring to that.
The “Best of DDW 2022” was the American Gastroenterological Association Presidential Plenary Session thanks to the humanity and vision of outgoing AGA President John Inadomi, MD.1 I sat in the audience, misty eyed, as each presenter addressed issues that strike deep into our humanity – the social determinants of health that have festered for far too long, leading to intolerable differences in health outcomes based on accidents of birth, and amplified by racism.
As the table on stage slowly filled in, an amazing picture took shape. A majority of the speakers were Black gastroenterologists and hepatologists, and among them many were young women. As I watched the video of a group of young Black gastroenterologists and hepatologists reaching out to the community, I asked myself “Has anything like this ever happened at a major national medical association meeting in the United States? Ever?” And then it occurred to me: “And just imagine, this exactly 2 days before the 2-year anniversary of the death of George Floyd.”
The plenary session happened on May 23, and I was conscious about the dates because I will never forget that George Floyd was killed on May 25, 2020 – my 55th birthday. The juxtaposition of his death and my birthday 2 years ago shook me profoundly, prompting me to write down my reflections and my hope that, in the national reactions that followed, we were seeing the beginning of true change.2 Two years later, despite our national divisions and serious challenges, I have reasons for hope.
On May 24, I ran into a colleague who was a Black woman. I have stopped being afraid to bring up previously untouchable subjects. I asked her what she thought about the remarkable AGA Plenary. She said she was glad that she is here to see it – that her parents never got the chance.
I admitted to her that I often ask myself what more I could and should be doing. I’m trying to do what I can in recruitment, education, in my personal life. What more? She said that one thing we really need is for people who look like me to amplify the message.
So here it is: Readers, listen to the plenary talks if you were not there. At minimum, behold the following line-up of speakers and topics. Feel the history.
This was the Best of DDW 2022:
- Julius Friedenwald Recognition of Timothy Wang. – John Inadomi.
- Presidential Address: Don’t Talk: Act. The relevance of DEI to gastroenterologists and hepatologists and the imperative for action. – John Inadomi.
- AGA Equity Project: Accomplishments and what lies ahead. – Byron L. Cryer, Sandra M. Quezada.
- The genesis and goals of the Association of Black Gastroenterologists and Hepatologists. – Sophie M. Balzora.
- Increasing racial and ethnic diversity in clinical trials: What we need to do. – Monica Webb Hooper.
- Reducing disparities in colorectal cancer. – Rachel Blankson Issaka.
- Reducing disparities in liver disease. – Lauren Nephew.
- Reducing disparities in IBD. – Fernando Velayos.
Uri Ladabaum, MD, MS, is with the division of gastroenterology and hepatology in the department of medicine at Stanford (Calif.) University. He reports serving on the advisory board for UniversalDx and Lean Medical and as a consultant for Medtronic, Clinical Genomics, Guardant Health, and Freenome. Dr. Ladabaum made these comments during the AGA Institute Presidential Plenary at the annual Digestive Disease Week®.
References
1. Inadomi JM. Gastroenterology. 2022 Jun;162(7):1855-7.
2. Ladabaum U. Ann Intern Med. 2020 Dec 1;173(11):938-9.
“The Best of DDW” elicits in the minds of most readers a compilation of the most important clinical and scientific content presented at DDW.
But I am not referring to that.
The “Best of DDW 2022” was the American Gastroenterological Association Presidential Plenary Session thanks to the humanity and vision of outgoing AGA President John Inadomi, MD.1 I sat in the audience, misty eyed, as each presenter addressed issues that strike deep into our humanity – the social determinants of health that have festered for far too long, leading to intolerable differences in health outcomes based on accidents of birth, and amplified by racism.
As the table on stage slowly filled in, an amazing picture took shape. A majority of the speakers were Black gastroenterologists and hepatologists, and among them many were young women. As I watched the video of a group of young Black gastroenterologists and hepatologists reaching out to the community, I asked myself “Has anything like this ever happened at a major national medical association meeting in the United States? Ever?” And then it occurred to me: “And just imagine, this exactly 2 days before the 2-year anniversary of the death of George Floyd.”
The plenary session happened on May 23, and I was conscious about the dates because I will never forget that George Floyd was killed on May 25, 2020 – my 55th birthday. The juxtaposition of his death and my birthday 2 years ago shook me profoundly, prompting me to write down my reflections and my hope that, in the national reactions that followed, we were seeing the beginning of true change.2 Two years later, despite our national divisions and serious challenges, I have reasons for hope.
On May 24, I ran into a colleague who was a Black woman. I have stopped being afraid to bring up previously untouchable subjects. I asked her what she thought about the remarkable AGA Plenary. She said she was glad that she is here to see it – that her parents never got the chance.
I admitted to her that I often ask myself what more I could and should be doing. I’m trying to do what I can in recruitment, education, in my personal life. What more? She said that one thing we really need is for people who look like me to amplify the message.
So here it is: Readers, listen to the plenary talks if you were not there. At minimum, behold the following line-up of speakers and topics. Feel the history.
This was the Best of DDW 2022:
- Julius Friedenwald Recognition of Timothy Wang. – John Inadomi.
- Presidential Address: Don’t Talk: Act. The relevance of DEI to gastroenterologists and hepatologists and the imperative for action. – John Inadomi.
- AGA Equity Project: Accomplishments and what lies ahead. – Byron L. Cryer, Sandra M. Quezada.
- The genesis and goals of the Association of Black Gastroenterologists and Hepatologists. – Sophie M. Balzora.
- Increasing racial and ethnic diversity in clinical trials: What we need to do. – Monica Webb Hooper.
- Reducing disparities in colorectal cancer. – Rachel Blankson Issaka.
- Reducing disparities in liver disease. – Lauren Nephew.
- Reducing disparities in IBD. – Fernando Velayos.
Uri Ladabaum, MD, MS, is with the division of gastroenterology and hepatology in the department of medicine at Stanford (Calif.) University. He reports serving on the advisory board for UniversalDx and Lean Medical and as a consultant for Medtronic, Clinical Genomics, Guardant Health, and Freenome. Dr. Ladabaum made these comments during the AGA Institute Presidential Plenary at the annual Digestive Disease Week®.
References
1. Inadomi JM. Gastroenterology. 2022 Jun;162(7):1855-7.
2. Ladabaum U. Ann Intern Med. 2020 Dec 1;173(11):938-9.
“The Best of DDW” elicits in the minds of most readers a compilation of the most important clinical and scientific content presented at DDW.
But I am not referring to that.
The “Best of DDW 2022” was the American Gastroenterological Association Presidential Plenary Session thanks to the humanity and vision of outgoing AGA President John Inadomi, MD.1 I sat in the audience, misty eyed, as each presenter addressed issues that strike deep into our humanity – the social determinants of health that have festered for far too long, leading to intolerable differences in health outcomes based on accidents of birth, and amplified by racism.
As the table on stage slowly filled in, an amazing picture took shape. A majority of the speakers were Black gastroenterologists and hepatologists, and among them many were young women. As I watched the video of a group of young Black gastroenterologists and hepatologists reaching out to the community, I asked myself “Has anything like this ever happened at a major national medical association meeting in the United States? Ever?” And then it occurred to me: “And just imagine, this exactly 2 days before the 2-year anniversary of the death of George Floyd.”
The plenary session happened on May 23, and I was conscious about the dates because I will never forget that George Floyd was killed on May 25, 2020 – my 55th birthday. The juxtaposition of his death and my birthday 2 years ago shook me profoundly, prompting me to write down my reflections and my hope that, in the national reactions that followed, we were seeing the beginning of true change.2 Two years later, despite our national divisions and serious challenges, I have reasons for hope.
On May 24, I ran into a colleague who was a Black woman. I have stopped being afraid to bring up previously untouchable subjects. I asked her what she thought about the remarkable AGA Plenary. She said she was glad that she is here to see it – that her parents never got the chance.
I admitted to her that I often ask myself what more I could and should be doing. I’m trying to do what I can in recruitment, education, in my personal life. What more? She said that one thing we really need is for people who look like me to amplify the message.
So here it is: Readers, listen to the plenary talks if you were not there. At minimum, behold the following line-up of speakers and topics. Feel the history.
This was the Best of DDW 2022:
- Julius Friedenwald Recognition of Timothy Wang. – John Inadomi.
- Presidential Address: Don’t Talk: Act. The relevance of DEI to gastroenterologists and hepatologists and the imperative for action. – John Inadomi.
- AGA Equity Project: Accomplishments and what lies ahead. – Byron L. Cryer, Sandra M. Quezada.
- The genesis and goals of the Association of Black Gastroenterologists and Hepatologists. – Sophie M. Balzora.
- Increasing racial and ethnic diversity in clinical trials: What we need to do. – Monica Webb Hooper.
- Reducing disparities in colorectal cancer. – Rachel Blankson Issaka.
- Reducing disparities in liver disease. – Lauren Nephew.
- Reducing disparities in IBD. – Fernando Velayos.
Uri Ladabaum, MD, MS, is with the division of gastroenterology and hepatology in the department of medicine at Stanford (Calif.) University. He reports serving on the advisory board for UniversalDx and Lean Medical and as a consultant for Medtronic, Clinical Genomics, Guardant Health, and Freenome. Dr. Ladabaum made these comments during the AGA Institute Presidential Plenary at the annual Digestive Disease Week®.
References
1. Inadomi JM. Gastroenterology. 2022 Jun;162(7):1855-7.
2. Ladabaum U. Ann Intern Med. 2020 Dec 1;173(11):938-9.
Reversing depression: A plethora of therapeutic strategies and mechanisms
Despite much progress, major depressive disorder (MDD) continues to be a challenging and life-threatening neuropsychiatric disorder. It is highly prevalent and afflicts tens of millions of Americans.
It is also ranked as the No. 1 disabling medical (not just psychiatric) condition by the World Health Organization.1 A significant proportion of patients with MDD do not respond adequately to several rounds of antidepressant medications,2 and many are labeled as having “treatment-resistant depression” (TRD).
In a previous article, I provocatively proposed that TRD is a myth.3 What I meant is that in a heterogeneous syndrome such as depression, failure to respond to 1, 2, or even 3 antidepressants should not imply TRD, because there is a “right treatment” that has not yet been identified for a given depressed patient. Most of those labeled as TRD have simply not yet received the pharmacotherapy or somatic therapy with the requisite mechanism of action for their variant of depression within a heterogeneous syndrome. IV ketamine, which, astonishingly, often reverses severe TRD of chronic duration within a few hours, is a prime example of why the term TRD is often used prematurely. Ketamine’s mechanism of action (immediate neuroplasticity via glutamate N-methyl-
Some clinicians may not be aware of the abundance of mechanisms of action currently available for the treatment of MDD as well as bipolar depression. Many practitioners, in both psychiatry and primary care, usually start the treatment of depression with a selective serotonin reuptake inhibitor, and if that does not produce a response or remission, they might switch to a serotonin-norepinephrine reuptake inhibitor. If that does not control the patient’s depressive symptoms, they start entertaining the notion that the patient may have TRD, not realizing that they have barely scratched the surface of the many therapeutic options and mechanisms of action, one of which could be the “best match” for a given patient.4
There will come a day when “precision psychiatry” finally arrives, and specific biomarkers will be developed to identify the “right” treatment for each patient within the heterogenous syndrome of depression.5 Until that day arrives, the treatment of depression will continue to be a process of trial and error, and hit or miss. But research will eventually discover genetic, neurochemical, neurophysiological, neuroimaging, or neuroimmune biomarkers that will rapidly guide clinicians to the correct treatment. This is critical to avoid inordinate delays in achieving remission and avert the ever-present risk of suicidal behavior.
The Table6 provides an overview of the numerous treatments currently available to manage depression. All increase brain-derived neurotrophic factor and restore healthy neuroplasticity and neurogenesis, which are impaired in MDD and currently believed to be a final common pathway for all depression treatments.7
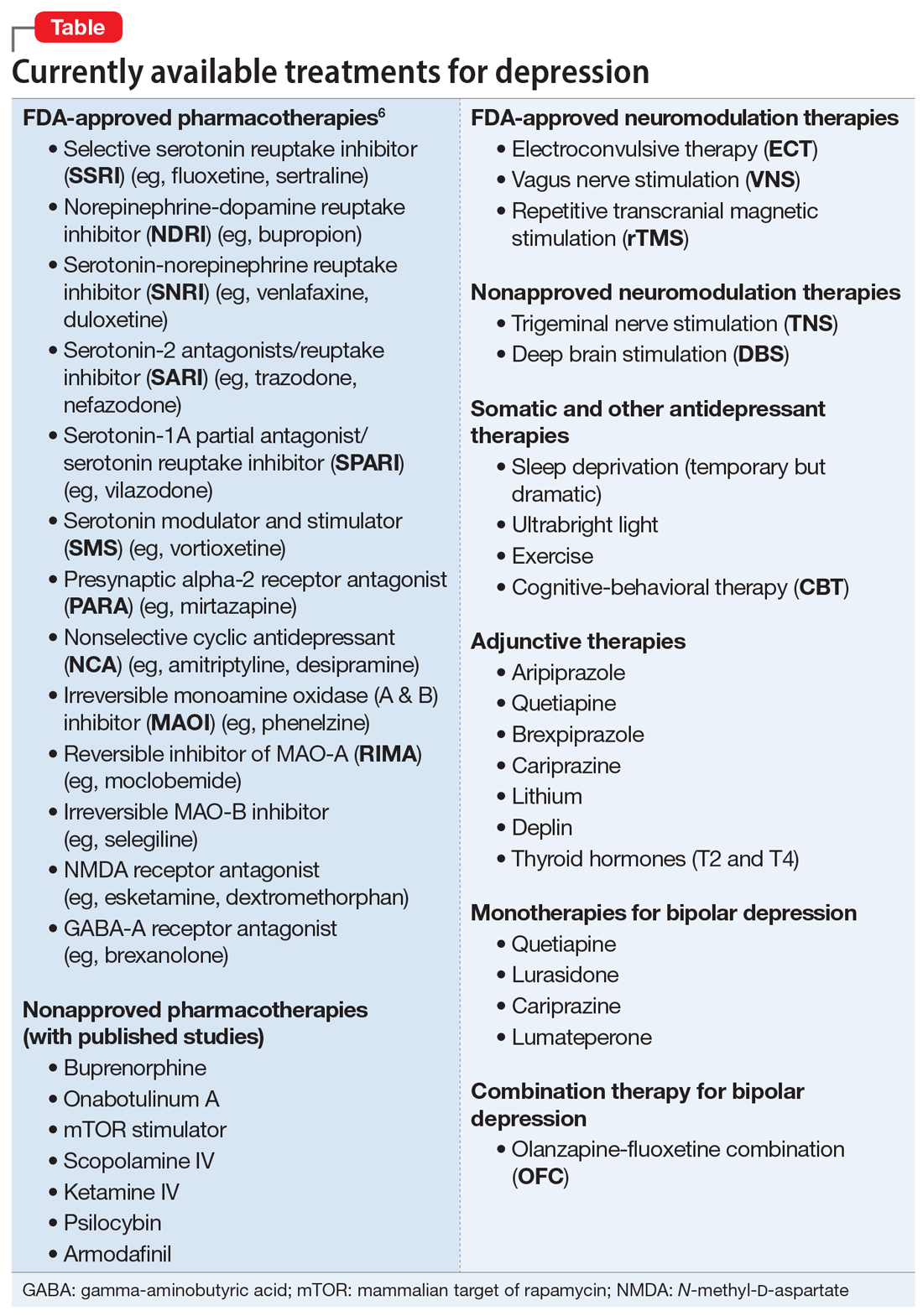
These 41 therapeutic approaches to treating MDD or bipolar depression reflect the heterogeneity of mechanisms of action to address an equally heterogeneous syndrome. This implies that clinicians have a wide array of on-label options to manage patients with depression, aiming for remission, not just a good response, which typically is defined as a ≥50% reduction in total score on one of the validated rating scales used to quantify depression severity, such as the Montgomery-Åsberg Depression Rating Scale, Hamilton Depression Rating Scale, or Calgary Depression Scale for Schizophrenia.
Continue to: When several FDA-approved pharmacotherapies...
When several FDA-approved pharmacotherapies fall short and produce a suboptimal response, clinicians can resort to other treatment options known to have a higher efficacy than oral antidepressants. These include electroconvulsive therapy, repetitive transcranial magnetic stimulation, and vagus nerve stimulation. Other on-label options include adjunctive therapy with one of the approved second-generation antipsychotic agents or with adjunctive esketamine.
But if the patient still does not improve, one of many emerging off-label treatment options may work. One of the exciting new discoveries is the hallucinogen psilocybin, whose mechanism of action is truly unique. Unlike standard antidepressant medications, which modulate neurotransmitters, psilocybin increases the brain’s network flexibility, decreases the modularity of several key brain networks (especially the default-brain network, or DMN), and alters the dark and distorted mental perspective of depression to a much healthier and optimistic outlook about the self and the world.8 Such novel breakthroughs in the treatment of severe depression will shed some unprecedented insights into the core neurobiology of depression, and may lead to early intervention and prevention.
As the saying goes, all roads lead to Rome. Psychiatric clinicians should rejoice that there are abundant approaches and therapeutic mechanisms to relieve their severely melancholic (and often suicidal) patients from the grips of this disabling and life-altering brain syndrome.
1. World Health Organization. Depression: let’s talk says WHO, as depression tops list of causes of ill health. March 30, 2017. Accessed July 5, 2022. www.who.int/news/item/30-03-2017--depression-let-s-talk-says-who-as-depression-tops-list-of-causes-of-ill-health
2. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Eng J Med. 2006;354(12)1243-1252.
3. Nasrallah HA. Treatment resistance is a myth! Current Psychiatry. 2021;20(3):14-16,28.
4. Nasrallah HA. 10 Recent paradigm shifts in the neurobiology and treatment of depression. Current Psychiatry. 2015;14(2):10-13.
5. Nasrallah HA. Biomarkers in neuropsychiatric disorders: translating research to clinical applications. Biomarkers in Neuropsychiatry. 2019;1:100001. doi:10.1016/j.bionps.2019.100001
6. Procyshyn RM, Bezchlibnyk-Butler KZ, Jeffries JJ. Clinical Handbook of Psychotropic Drugs. 23rd ed. Hogrefe; 2019.
7. Tartt AN, Mariani, MB, Hen R, et al. Dysregulation of adult hippocampal neuroplasticity in major depression: pathogenesis and therapeutic implications. Mol Psychiatry. 2022;27(6):2689-2699.
8. Lowe H, Toyang N, Steele B, et al. The therapeutic potential of psilocybin. Molecules. 2021;26(10):2948. doi: 10.3390/molecules26102948
Despite much progress, major depressive disorder (MDD) continues to be a challenging and life-threatening neuropsychiatric disorder. It is highly prevalent and afflicts tens of millions of Americans.
It is also ranked as the No. 1 disabling medical (not just psychiatric) condition by the World Health Organization.1 A significant proportion of patients with MDD do not respond adequately to several rounds of antidepressant medications,2 and many are labeled as having “treatment-resistant depression” (TRD).
In a previous article, I provocatively proposed that TRD is a myth.3 What I meant is that in a heterogeneous syndrome such as depression, failure to respond to 1, 2, or even 3 antidepressants should not imply TRD, because there is a “right treatment” that has not yet been identified for a given depressed patient. Most of those labeled as TRD have simply not yet received the pharmacotherapy or somatic therapy with the requisite mechanism of action for their variant of depression within a heterogeneous syndrome. IV ketamine, which, astonishingly, often reverses severe TRD of chronic duration within a few hours, is a prime example of why the term TRD is often used prematurely. Ketamine’s mechanism of action (immediate neuroplasticity via glutamate N-methyl-
Some clinicians may not be aware of the abundance of mechanisms of action currently available for the treatment of MDD as well as bipolar depression. Many practitioners, in both psychiatry and primary care, usually start the treatment of depression with a selective serotonin reuptake inhibitor, and if that does not produce a response or remission, they might switch to a serotonin-norepinephrine reuptake inhibitor. If that does not control the patient’s depressive symptoms, they start entertaining the notion that the patient may have TRD, not realizing that they have barely scratched the surface of the many therapeutic options and mechanisms of action, one of which could be the “best match” for a given patient.4
There will come a day when “precision psychiatry” finally arrives, and specific biomarkers will be developed to identify the “right” treatment for each patient within the heterogenous syndrome of depression.5 Until that day arrives, the treatment of depression will continue to be a process of trial and error, and hit or miss. But research will eventually discover genetic, neurochemical, neurophysiological, neuroimaging, or neuroimmune biomarkers that will rapidly guide clinicians to the correct treatment. This is critical to avoid inordinate delays in achieving remission and avert the ever-present risk of suicidal behavior.
The Table6 provides an overview of the numerous treatments currently available to manage depression. All increase brain-derived neurotrophic factor and restore healthy neuroplasticity and neurogenesis, which are impaired in MDD and currently believed to be a final common pathway for all depression treatments.7

These 41 therapeutic approaches to treating MDD or bipolar depression reflect the heterogeneity of mechanisms of action to address an equally heterogeneous syndrome. This implies that clinicians have a wide array of on-label options to manage patients with depression, aiming for remission, not just a good response, which typically is defined as a ≥50% reduction in total score on one of the validated rating scales used to quantify depression severity, such as the Montgomery-Åsberg Depression Rating Scale, Hamilton Depression Rating Scale, or Calgary Depression Scale for Schizophrenia.
Continue to: When several FDA-approved pharmacotherapies...
When several FDA-approved pharmacotherapies fall short and produce a suboptimal response, clinicians can resort to other treatment options known to have a higher efficacy than oral antidepressants. These include electroconvulsive therapy, repetitive transcranial magnetic stimulation, and vagus nerve stimulation. Other on-label options include adjunctive therapy with one of the approved second-generation antipsychotic agents or with adjunctive esketamine.
But if the patient still does not improve, one of many emerging off-label treatment options may work. One of the exciting new discoveries is the hallucinogen psilocybin, whose mechanism of action is truly unique. Unlike standard antidepressant medications, which modulate neurotransmitters, psilocybin increases the brain’s network flexibility, decreases the modularity of several key brain networks (especially the default-brain network, or DMN), and alters the dark and distorted mental perspective of depression to a much healthier and optimistic outlook about the self and the world.8 Such novel breakthroughs in the treatment of severe depression will shed some unprecedented insights into the core neurobiology of depression, and may lead to early intervention and prevention.
As the saying goes, all roads lead to Rome. Psychiatric clinicians should rejoice that there are abundant approaches and therapeutic mechanisms to relieve their severely melancholic (and often suicidal) patients from the grips of this disabling and life-altering brain syndrome.
Despite much progress, major depressive disorder (MDD) continues to be a challenging and life-threatening neuropsychiatric disorder. It is highly prevalent and afflicts tens of millions of Americans.
It is also ranked as the No. 1 disabling medical (not just psychiatric) condition by the World Health Organization.1 A significant proportion of patients with MDD do not respond adequately to several rounds of antidepressant medications,2 and many are labeled as having “treatment-resistant depression” (TRD).
In a previous article, I provocatively proposed that TRD is a myth.3 What I meant is that in a heterogeneous syndrome such as depression, failure to respond to 1, 2, or even 3 antidepressants should not imply TRD, because there is a “right treatment” that has not yet been identified for a given depressed patient. Most of those labeled as TRD have simply not yet received the pharmacotherapy or somatic therapy with the requisite mechanism of action for their variant of depression within a heterogeneous syndrome. IV ketamine, which, astonishingly, often reverses severe TRD of chronic duration within a few hours, is a prime example of why the term TRD is often used prematurely. Ketamine’s mechanism of action (immediate neuroplasticity via glutamate N-methyl-
Some clinicians may not be aware of the abundance of mechanisms of action currently available for the treatment of MDD as well as bipolar depression. Many practitioners, in both psychiatry and primary care, usually start the treatment of depression with a selective serotonin reuptake inhibitor, and if that does not produce a response or remission, they might switch to a serotonin-norepinephrine reuptake inhibitor. If that does not control the patient’s depressive symptoms, they start entertaining the notion that the patient may have TRD, not realizing that they have barely scratched the surface of the many therapeutic options and mechanisms of action, one of which could be the “best match” for a given patient.4
There will come a day when “precision psychiatry” finally arrives, and specific biomarkers will be developed to identify the “right” treatment for each patient within the heterogenous syndrome of depression.5 Until that day arrives, the treatment of depression will continue to be a process of trial and error, and hit or miss. But research will eventually discover genetic, neurochemical, neurophysiological, neuroimaging, or neuroimmune biomarkers that will rapidly guide clinicians to the correct treatment. This is critical to avoid inordinate delays in achieving remission and avert the ever-present risk of suicidal behavior.
The Table6 provides an overview of the numerous treatments currently available to manage depression. All increase brain-derived neurotrophic factor and restore healthy neuroplasticity and neurogenesis, which are impaired in MDD and currently believed to be a final common pathway for all depression treatments.7

These 41 therapeutic approaches to treating MDD or bipolar depression reflect the heterogeneity of mechanisms of action to address an equally heterogeneous syndrome. This implies that clinicians have a wide array of on-label options to manage patients with depression, aiming for remission, not just a good response, which typically is defined as a ≥50% reduction in total score on one of the validated rating scales used to quantify depression severity, such as the Montgomery-Åsberg Depression Rating Scale, Hamilton Depression Rating Scale, or Calgary Depression Scale for Schizophrenia.
Continue to: When several FDA-approved pharmacotherapies...
When several FDA-approved pharmacotherapies fall short and produce a suboptimal response, clinicians can resort to other treatment options known to have a higher efficacy than oral antidepressants. These include electroconvulsive therapy, repetitive transcranial magnetic stimulation, and vagus nerve stimulation. Other on-label options include adjunctive therapy with one of the approved second-generation antipsychotic agents or with adjunctive esketamine.
But if the patient still does not improve, one of many emerging off-label treatment options may work. One of the exciting new discoveries is the hallucinogen psilocybin, whose mechanism of action is truly unique. Unlike standard antidepressant medications, which modulate neurotransmitters, psilocybin increases the brain’s network flexibility, decreases the modularity of several key brain networks (especially the default-brain network, or DMN), and alters the dark and distorted mental perspective of depression to a much healthier and optimistic outlook about the self and the world.8 Such novel breakthroughs in the treatment of severe depression will shed some unprecedented insights into the core neurobiology of depression, and may lead to early intervention and prevention.
As the saying goes, all roads lead to Rome. Psychiatric clinicians should rejoice that there are abundant approaches and therapeutic mechanisms to relieve their severely melancholic (and often suicidal) patients from the grips of this disabling and life-altering brain syndrome.
1. World Health Organization. Depression: let’s talk says WHO, as depression tops list of causes of ill health. March 30, 2017. Accessed July 5, 2022. www.who.int/news/item/30-03-2017--depression-let-s-talk-says-who-as-depression-tops-list-of-causes-of-ill-health
2. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Eng J Med. 2006;354(12)1243-1252.
3. Nasrallah HA. Treatment resistance is a myth! Current Psychiatry. 2021;20(3):14-16,28.
4. Nasrallah HA. 10 Recent paradigm shifts in the neurobiology and treatment of depression. Current Psychiatry. 2015;14(2):10-13.
5. Nasrallah HA. Biomarkers in neuropsychiatric disorders: translating research to clinical applications. Biomarkers in Neuropsychiatry. 2019;1:100001. doi:10.1016/j.bionps.2019.100001
6. Procyshyn RM, Bezchlibnyk-Butler KZ, Jeffries JJ. Clinical Handbook of Psychotropic Drugs. 23rd ed. Hogrefe; 2019.
7. Tartt AN, Mariani, MB, Hen R, et al. Dysregulation of adult hippocampal neuroplasticity in major depression: pathogenesis and therapeutic implications. Mol Psychiatry. 2022;27(6):2689-2699.
8. Lowe H, Toyang N, Steele B, et al. The therapeutic potential of psilocybin. Molecules. 2021;26(10):2948. doi: 10.3390/molecules26102948
1. World Health Organization. Depression: let’s talk says WHO, as depression tops list of causes of ill health. March 30, 2017. Accessed July 5, 2022. www.who.int/news/item/30-03-2017--depression-let-s-talk-says-who-as-depression-tops-list-of-causes-of-ill-health
2. Trivedi MH, Fava M, Wisniewski SR, et al. Medication augmentation after the failure of SSRIs for depression. N Eng J Med. 2006;354(12)1243-1252.
3. Nasrallah HA. Treatment resistance is a myth! Current Psychiatry. 2021;20(3):14-16,28.
4. Nasrallah HA. 10 Recent paradigm shifts in the neurobiology and treatment of depression. Current Psychiatry. 2015;14(2):10-13.
5. Nasrallah HA. Biomarkers in neuropsychiatric disorders: translating research to clinical applications. Biomarkers in Neuropsychiatry. 2019;1:100001. doi:10.1016/j.bionps.2019.100001
6. Procyshyn RM, Bezchlibnyk-Butler KZ, Jeffries JJ. Clinical Handbook of Psychotropic Drugs. 23rd ed. Hogrefe; 2019.
7. Tartt AN, Mariani, MB, Hen R, et al. Dysregulation of adult hippocampal neuroplasticity in major depression: pathogenesis and therapeutic implications. Mol Psychiatry. 2022;27(6):2689-2699.
8. Lowe H, Toyang N, Steele B, et al. The therapeutic potential of psilocybin. Molecules. 2021;26(10):2948. doi: 10.3390/molecules26102948
Hope, help, and humor when facing a life-threatening illness
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
My father, Morty Sosland, MD, was a psychiatrist in a community health setting when he was diagnosed with amyotrophic lateral sclerosis (ALS; Lou Gehrig’s disease) in April 2020. He continued to work until February 2021 and credits his ongoing resilience to what he refers to as “the 3 Hs”: hope, help, and humor. Although he can no longer speak, I was able to interview him over the advanced technology that is text messaging.
Sarah: Hi, Dad.
Morty: It’s Doctor Dad to you.
Sarah: I guess we are starting with humor, then?
Humor
Research has demonstrated that humor can have serious health benefits, such as decreasing stress-making hormones and altering dopamine activity.1 For individuals facing a life-threatening illness, humor can help them gain a sense of perspective in a situation that would otherwise feel overwhelming.
Sarah: I feel like a lot of the humor you used with patients was to help them gain perspective.
Continue to: Morty
Morty: Yes. I’d have to know the client well enough, though—and timing is important. My patients would come to me with a long list of challenges they had faced in the week, and I would say, “But besides that, everything’s good?”
Sarah: And besides the ALS, everything’s good?
Morty: Exactly. I’d also use magic or math tricks to make kids like coming to therapy or to reinforce important concepts.
Sarah: How has humor helped you cope?
Morty: Thinking about things in humorous ways has always been helpful. I used to say my Olympic sport was walking to the dining room with my walker. Unfortunately, I can’t do that anymore, so now my Olympic sport is getting out of bed. It’s a team sport.
Sarah: And that’s a good segue to…
Help
Countless studies have shown the impact of social support on health. Good social support can increase resilience, protect against mental illness, and even increase life expectancy.2 Support becomes even more critical when you are physically dependent on others due to illness.
Continue to: Sarah
Sarah: Was it difficult for you to accept help at first?
Morty: I would say yes—but at the same time, I accepted it because the illness was so shocking. I learned early on this was a fight that my family would also fight alongside me.
Sarah: I remember you would quote Fred Rogers.
Morty: Actually, it was Fred Rogers’ mother. She would tell her son during hard times, “Look for the helpers. You will always find people who are helping.” Helpers can be family members, friends, doctors, and aides, as well as others who have the same illness.
Hope
In the face of all life’s challenges, hope is important, but in the face of a life-threatening illness, hope must be multifaceted.3 In addition to hope for a cure, patients may focus their hopes on deepening relationships, maintaining dignity, or living each day to its fullest.
Morty: Early on in this illness, I chose to set a positive tone when I told people. I would say I have the top doctors and there is more research now than ever. Years ago, I wrote a children’s book with the mantra, “I say I can, I make a plan, I get right to it and then I do it.”4 My plan is to be around for at least 30 more years.
Sarah: Do you think it’s possible to hold acceptance and hope at the same time?
Morty: Acceptance and hope are not easy, but possible. I get down about this illness. In my dreams, I walk and talk, and most mornings I wake up and see my wheelchair and I think this is absurd or a different choice word. But I focus on the things I still can do, and that gives me a feeling of hope. I can read the latest research, I can enjoy moments of laughter, and I can spend time with my family and close friends.
1. Yim J. Therapeutic benefits of laughter in mental health: a theoretical review. Tohoku J Exp Med. 2016;239(3):243-249. doi:10.1620/tjem.239.243
2. Ozbay F, Johnson DC, Dimoulas E, et al. Social support and resilience to stress: from neurobiology to clinical practice. Psychiatry (Edgmont). 2007;4(5):35-40.
3. Hill DL, Feudnter C. Hope in the midst of terminal illness. In: Gallagher MW, Lopez SJ, eds. The Oxford Handbook of Hope. Oxford University Press; 2018:191-206.
4. Sosland MD. The Can Do Duck: A Story About Believing in Yourself. Can Do Duck Publishing; 2019.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
My father, Morty Sosland, MD, was a psychiatrist in a community health setting when he was diagnosed with amyotrophic lateral sclerosis (ALS; Lou Gehrig’s disease) in April 2020. He continued to work until February 2021 and credits his ongoing resilience to what he refers to as “the 3 Hs”: hope, help, and humor. Although he can no longer speak, I was able to interview him over the advanced technology that is text messaging.
Sarah: Hi, Dad.
Morty: It’s Doctor Dad to you.
Sarah: I guess we are starting with humor, then?
Humor
Research has demonstrated that humor can have serious health benefits, such as decreasing stress-making hormones and altering dopamine activity.1 For individuals facing a life-threatening illness, humor can help them gain a sense of perspective in a situation that would otherwise feel overwhelming.
Sarah: I feel like a lot of the humor you used with patients was to help them gain perspective.
Continue to: Morty
Morty: Yes. I’d have to know the client well enough, though—and timing is important. My patients would come to me with a long list of challenges they had faced in the week, and I would say, “But besides that, everything’s good?”
Sarah: And besides the ALS, everything’s good?
Morty: Exactly. I’d also use magic or math tricks to make kids like coming to therapy or to reinforce important concepts.
Sarah: How has humor helped you cope?
Morty: Thinking about things in humorous ways has always been helpful. I used to say my Olympic sport was walking to the dining room with my walker. Unfortunately, I can’t do that anymore, so now my Olympic sport is getting out of bed. It’s a team sport.
Sarah: And that’s a good segue to…
Help
Countless studies have shown the impact of social support on health. Good social support can increase resilience, protect against mental illness, and even increase life expectancy.2 Support becomes even more critical when you are physically dependent on others due to illness.
Continue to: Sarah
Sarah: Was it difficult for you to accept help at first?
Morty: I would say yes—but at the same time, I accepted it because the illness was so shocking. I learned early on this was a fight that my family would also fight alongside me.
Sarah: I remember you would quote Fred Rogers.
Morty: Actually, it was Fred Rogers’ mother. She would tell her son during hard times, “Look for the helpers. You will always find people who are helping.” Helpers can be family members, friends, doctors, and aides, as well as others who have the same illness.
Hope
In the face of all life’s challenges, hope is important, but in the face of a life-threatening illness, hope must be multifaceted.3 In addition to hope for a cure, patients may focus their hopes on deepening relationships, maintaining dignity, or living each day to its fullest.
Morty: Early on in this illness, I chose to set a positive tone when I told people. I would say I have the top doctors and there is more research now than ever. Years ago, I wrote a children’s book with the mantra, “I say I can, I make a plan, I get right to it and then I do it.”4 My plan is to be around for at least 30 more years.
Sarah: Do you think it’s possible to hold acceptance and hope at the same time?
Morty: Acceptance and hope are not easy, but possible. I get down about this illness. In my dreams, I walk and talk, and most mornings I wake up and see my wheelchair and I think this is absurd or a different choice word. But I focus on the things I still can do, and that gives me a feeling of hope. I can read the latest research, I can enjoy moments of laughter, and I can spend time with my family and close friends.
Editor’s note: Readers’ Forum is a department for correspondence from readers that is not in response to articles published in
My father, Morty Sosland, MD, was a psychiatrist in a community health setting when he was diagnosed with amyotrophic lateral sclerosis (ALS; Lou Gehrig’s disease) in April 2020. He continued to work until February 2021 and credits his ongoing resilience to what he refers to as “the 3 Hs”: hope, help, and humor. Although he can no longer speak, I was able to interview him over the advanced technology that is text messaging.
Sarah: Hi, Dad.
Morty: It’s Doctor Dad to you.
Sarah: I guess we are starting with humor, then?
Humor
Research has demonstrated that humor can have serious health benefits, such as decreasing stress-making hormones and altering dopamine activity.1 For individuals facing a life-threatening illness, humor can help them gain a sense of perspective in a situation that would otherwise feel overwhelming.
Sarah: I feel like a lot of the humor you used with patients was to help them gain perspective.
Continue to: Morty
Morty: Yes. I’d have to know the client well enough, though—and timing is important. My patients would come to me with a long list of challenges they had faced in the week, and I would say, “But besides that, everything’s good?”
Sarah: And besides the ALS, everything’s good?
Morty: Exactly. I’d also use magic or math tricks to make kids like coming to therapy or to reinforce important concepts.
Sarah: How has humor helped you cope?
Morty: Thinking about things in humorous ways has always been helpful. I used to say my Olympic sport was walking to the dining room with my walker. Unfortunately, I can’t do that anymore, so now my Olympic sport is getting out of bed. It’s a team sport.
Sarah: And that’s a good segue to…
Help
Countless studies have shown the impact of social support on health. Good social support can increase resilience, protect against mental illness, and even increase life expectancy.2 Support becomes even more critical when you are physically dependent on others due to illness.
Continue to: Sarah
Sarah: Was it difficult for you to accept help at first?
Morty: I would say yes—but at the same time, I accepted it because the illness was so shocking. I learned early on this was a fight that my family would also fight alongside me.
Sarah: I remember you would quote Fred Rogers.
Morty: Actually, it was Fred Rogers’ mother. She would tell her son during hard times, “Look for the helpers. You will always find people who are helping.” Helpers can be family members, friends, doctors, and aides, as well as others who have the same illness.
Hope
In the face of all life’s challenges, hope is important, but in the face of a life-threatening illness, hope must be multifaceted.3 In addition to hope for a cure, patients may focus their hopes on deepening relationships, maintaining dignity, or living each day to its fullest.
Morty: Early on in this illness, I chose to set a positive tone when I told people. I would say I have the top doctors and there is more research now than ever. Years ago, I wrote a children’s book with the mantra, “I say I can, I make a plan, I get right to it and then I do it.”4 My plan is to be around for at least 30 more years.
Sarah: Do you think it’s possible to hold acceptance and hope at the same time?
Morty: Acceptance and hope are not easy, but possible. I get down about this illness. In my dreams, I walk and talk, and most mornings I wake up and see my wheelchair and I think this is absurd or a different choice word. But I focus on the things I still can do, and that gives me a feeling of hope. I can read the latest research, I can enjoy moments of laughter, and I can spend time with my family and close friends.
1. Yim J. Therapeutic benefits of laughter in mental health: a theoretical review. Tohoku J Exp Med. 2016;239(3):243-249. doi:10.1620/tjem.239.243
2. Ozbay F, Johnson DC, Dimoulas E, et al. Social support and resilience to stress: from neurobiology to clinical practice. Psychiatry (Edgmont). 2007;4(5):35-40.
3. Hill DL, Feudnter C. Hope in the midst of terminal illness. In: Gallagher MW, Lopez SJ, eds. The Oxford Handbook of Hope. Oxford University Press; 2018:191-206.
4. Sosland MD. The Can Do Duck: A Story About Believing in Yourself. Can Do Duck Publishing; 2019.
1. Yim J. Therapeutic benefits of laughter in mental health: a theoretical review. Tohoku J Exp Med. 2016;239(3):243-249. doi:10.1620/tjem.239.243
2. Ozbay F, Johnson DC, Dimoulas E, et al. Social support and resilience to stress: from neurobiology to clinical practice. Psychiatry (Edgmont). 2007;4(5):35-40.
3. Hill DL, Feudnter C. Hope in the midst of terminal illness. In: Gallagher MW, Lopez SJ, eds. The Oxford Handbook of Hope. Oxford University Press; 2018:191-206.
4. Sosland MD. The Can Do Duck: A Story About Believing in Yourself. Can Do Duck Publishing; 2019.
The impact of COVID-19 on adolescents’ mental health
While the COVID-19 pandemic has impacted the mental health of a wide range of individuals, its adverse effects have been particularly detrimental to adolescents. In this article, I discuss evidence that shows the effects of the pandemic on adolescent patients, potential reasons for this increased distress, and what types of coping mechanisms adolescents have used to counter these effects.
Increases in multiple measures of psychopathology
Multiple online surveys and other studies have documented the pandemic’s impact on younger individuals. In the United States, visits to emergency departments by pediatric patients increased in the months after the first lockdown period.1 Several studies found increased rates of anxiety and depression among adolescents during the COVID-19 pandemic.2,3 In an online survey of 359 children and 3,254 adolescents in China, 22% of respondents reported that they experienced depressive symptoms.3 In an online survey of 1,054 Canadian adolescents, 43% said they were “very concerned” about the pandemic.4 In an online survey of 7,353 adolescents in the United States, 37% reported suicidal ideation during the pandemic compared to 17% in 2017.5 A Chinese study found that smartphone and internet addiction was significantly associated with increased levels of depressive symptoms during the pandemic.3 In a survey in the Philippines, 16.3% of adolescents reported moderate-to-severe psychological impairment during the pandemic; the rates of COVID-19–related anxiety were higher among girls vs boys.6 Alcohol and cannabis use increased among Canadian adolescents during the pandemic, according to an online survey.7 Adolescents with anorexia nervosa reported a 70% increase in poor eating habits and more thoughts associated with eating disorders during the pandemic.8 A Danish study found that children and adolescents newly diagnosed with obsessive-compulsive disorder (OCD) or who had completed treatment exhibited worsening OCD, anxiety, and depressive symptoms during the pandemic.9 An online survey of 6,196 Chinese adolescents found that those with a higher number of pre-pandemic adverse childhood experiences, such as abuse and neglect, had elevated posttraumatic stress symptoms and anxiety during the onset of the pandemic.10
Underlying causes of pandemic-induced distress
Limited social connectedness during the pandemic is a major reason for distress among adolescents. A review of 80 studies found that social isolation and loneliness as a result of social distancing and quarantining were associated with an increased risk of depression, anxiety, suicidal ideation, and self-harm.11 Parents’ stress about the risks of COVID-19 was correlated with worsening mental health in their adolescent children.12 A Chinese study found that the amount of time students spent on smartphones and social media doubled during the pandemic.13 In an online survey of 7,890 Chinese adolescents, greater social media, internet, and smartphone use was associated with increased anxiety and depression.14 This may be in part the result of adolescents spending time reading COVID-related news.
Coping mechanisms to increase well-being
Researchers have identified several positive coping mechanisms adolescents employed during the pandemic. Although some data suggest that increased internet use raises the risk of COVID-related distress, for certain adolescents, using social media to stay connected with friends and relatives was a buffer for feelings of loneliness and might have increased mental well-being.15 Other common coping mechanisms include relying on faith, volunteering, and starting new hobbies.16 During the pandemic, there were higher rates of playing outside and increased physical activity, which correlated with positive mental health outcomes.16 An online survey of 1,040 adolescents found that those who looked to the future optimistically and confidently had a higher health-related quality of life.17
Continuing an emphasis on adolescent well-being
Although data are limited, adolescents can continue to use these coping mechanisms to maintain their well-being, even if COVID-related restrictions are lifted or reimplemented. During these difficult times, it is imperative for adolescents to get the mental health services they need, and for psychiatric clinicians to continue to find avenues to promote resilience and mental wellness among young patients.
1. Leeb RT, Bitsko RH, Radhakrishnan L, et al. Mental health–related emergency department visits among children aged <18 years during the COVID-19 pandemic—United States, January 1-October 17, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(45):1675-1680. doi:10.15585/mmwr.mm6945a3
2. Oosterhoff B, Palmer CA, Wilson J, et al. Adolescents’ motivations to engage in social distancing during the COVID-19 pandemic: associations with mental and social health. J Adolesc Health. 2020;67(2):179-185. doi:10.1016/j.jadohealth.2020.05.004
3. Duan L, Shao X, Wang Y, et al. An investigation of mental health status of children and adolescents in China during the outbreak of COVID-19. J Affect Disord. 2020;275:112-118. doi:10.1016/j.jad.2020.06.029
4. Ellis WE, Dumas TM, Forbes LM. Physically isolated but socially connected: psychological adjustment and stress among adolescents during the initial COVID-19 crisis. Can J Behav Sci. 2020;52(3):177-187. doi:10.1037/cbs0000215
5. Murata S, Rezeppa T, Thoma B, et al. The psychiatric sequelae of the COVID-19 pandemic in adolescents, adults, and health care workers. Depress Anxiety. 2021;38(2):233-246. doi:10.1002/da.23120
6. Tee ML, Tee CA, Anlacan JP, et al. Psychological impact of COVID-19 pandemic in the Philippines. J Affect Disord. 2020;277:379-391. doi:10.1016/j.jad.2020.08.043
7. Dumas TM, Ellis W, Litt DM. What does adolescent substance use look like during the COVID-19 pandemic? Examining changes in frequency, social contexts, and pandemic-related predictors. J Adolesc Health. 2020;67(3):354-361. doi:10.1016/j.jadohealth.2020.06.018
8. Schlegl S, Maier J, Meule A, et al. Eating disorders in times of the COVID-19 pandemic—results from an online survey of patients with anorexia nervosa. Int J Eat Disord. 2020;53:1791-1800. doi:10.1002/eat.23374.
9. Nissen JB, Højgaard D, Thomsen PH. The immediate effect of COVID-19 pandemic on children and adolescents with obsessive compulsive disorder. BMC Psychiatry. 2020;20(1):511. doi:10.1186/s12888-020-02905-5
10. Guo J, Fu M, Liu D, et al. Is the psychological impact of exposure to COVID-19 stronger in adolescents with pre-pandemic maltreatment experiences? A survey of rural Chinese adolescents. Child Abuse Negl. 2020;110(Pt 2):104667. doi:10.1016/j.chiabu.2020.104667
11. Loades ME, Chatburn E, Higson-Sweeney N, et al. Rapid Systematic Review: The impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19. J Am Acad Child Adolesc Psychiatry. 2020;59(11):1218-1239.e3. doi:10.1016/j.jaac.2020.05.009
12. Spinelli M, Lionetti F, Setti A, et al. Parenting stress during the COVID-19 outbreak: socioeconomic and environmental risk factors and implications for children emotion regulation. Fam Process. 2021;60(2):639-653. doi:10.1111/famp.12601
13. Chen IH, Chen CY, Pakpour AH, et al. Internet-related behaviors and psychological distress among schoolchildren during COVID-19 school suspension. J Am Acad Child Adolesc Psychiatry. 2020;59(10):1099-1102.e1. doi:10.1016/j.jaac.2020.06.007
14. Li W, Zhang Y, Wang J, et al. Association of home quarantine and mental health among teenagers in Wuhan, China, during the COVID-19 pandemic. JAMA Pediatr. 2021;175(3):313-316. doi:10.1001/jamapediatrics.2020.5499
15. Janssen, LHC, Kullberg, MJ, Verkuil B, et al. Does the COVID-19 pandemic impact parents’ and adolescents’ well-being? An EMA-study on daily affect and parenting. PLoS One. 2020;15(10):e0240962. doi:10.1371/journal.pone.0240962
16. Banati P, Jones N, Youssef S. Intersecting vulnerabilities: the impacts of COVID-19 on the psycho-emotional lives of young people in low- and middle-income countries. Eur J Dev Res. 2020;32(5):1613-1638. doi:10.1057/s41287-020-00325-5
17. Ravens-Sieberer U, Kaman A, Otto C, et al. Mental health and quality of life in children and adolescents during the COVID-19 pandemic—results of the COPSY study. Dtsch Arztebl Int. 2020;117(48):828-829. doi:10.3238/arztebl.2020.0828
While the COVID-19 pandemic has impacted the mental health of a wide range of individuals, its adverse effects have been particularly detrimental to adolescents. In this article, I discuss evidence that shows the effects of the pandemic on adolescent patients, potential reasons for this increased distress, and what types of coping mechanisms adolescents have used to counter these effects.
Increases in multiple measures of psychopathology
Multiple online surveys and other studies have documented the pandemic’s impact on younger individuals. In the United States, visits to emergency departments by pediatric patients increased in the months after the first lockdown period.1 Several studies found increased rates of anxiety and depression among adolescents during the COVID-19 pandemic.2,3 In an online survey of 359 children and 3,254 adolescents in China, 22% of respondents reported that they experienced depressive symptoms.3 In an online survey of 1,054 Canadian adolescents, 43% said they were “very concerned” about the pandemic.4 In an online survey of 7,353 adolescents in the United States, 37% reported suicidal ideation during the pandemic compared to 17% in 2017.5 A Chinese study found that smartphone and internet addiction was significantly associated with increased levels of depressive symptoms during the pandemic.3 In a survey in the Philippines, 16.3% of adolescents reported moderate-to-severe psychological impairment during the pandemic; the rates of COVID-19–related anxiety were higher among girls vs boys.6 Alcohol and cannabis use increased among Canadian adolescents during the pandemic, according to an online survey.7 Adolescents with anorexia nervosa reported a 70% increase in poor eating habits and more thoughts associated with eating disorders during the pandemic.8 A Danish study found that children and adolescents newly diagnosed with obsessive-compulsive disorder (OCD) or who had completed treatment exhibited worsening OCD, anxiety, and depressive symptoms during the pandemic.9 An online survey of 6,196 Chinese adolescents found that those with a higher number of pre-pandemic adverse childhood experiences, such as abuse and neglect, had elevated posttraumatic stress symptoms and anxiety during the onset of the pandemic.10
Underlying causes of pandemic-induced distress
Limited social connectedness during the pandemic is a major reason for distress among adolescents. A review of 80 studies found that social isolation and loneliness as a result of social distancing and quarantining were associated with an increased risk of depression, anxiety, suicidal ideation, and self-harm.11 Parents’ stress about the risks of COVID-19 was correlated with worsening mental health in their adolescent children.12 A Chinese study found that the amount of time students spent on smartphones and social media doubled during the pandemic.13 In an online survey of 7,890 Chinese adolescents, greater social media, internet, and smartphone use was associated with increased anxiety and depression.14 This may be in part the result of adolescents spending time reading COVID-related news.
Coping mechanisms to increase well-being
Researchers have identified several positive coping mechanisms adolescents employed during the pandemic. Although some data suggest that increased internet use raises the risk of COVID-related distress, for certain adolescents, using social media to stay connected with friends and relatives was a buffer for feelings of loneliness and might have increased mental well-being.15 Other common coping mechanisms include relying on faith, volunteering, and starting new hobbies.16 During the pandemic, there were higher rates of playing outside and increased physical activity, which correlated with positive mental health outcomes.16 An online survey of 1,040 adolescents found that those who looked to the future optimistically and confidently had a higher health-related quality of life.17
Continuing an emphasis on adolescent well-being
Although data are limited, adolescents can continue to use these coping mechanisms to maintain their well-being, even if COVID-related restrictions are lifted or reimplemented. During these difficult times, it is imperative for adolescents to get the mental health services they need, and for psychiatric clinicians to continue to find avenues to promote resilience and mental wellness among young patients.
While the COVID-19 pandemic has impacted the mental health of a wide range of individuals, its adverse effects have been particularly detrimental to adolescents. In this article, I discuss evidence that shows the effects of the pandemic on adolescent patients, potential reasons for this increased distress, and what types of coping mechanisms adolescents have used to counter these effects.
Increases in multiple measures of psychopathology
Multiple online surveys and other studies have documented the pandemic’s impact on younger individuals. In the United States, visits to emergency departments by pediatric patients increased in the months after the first lockdown period.1 Several studies found increased rates of anxiety and depression among adolescents during the COVID-19 pandemic.2,3 In an online survey of 359 children and 3,254 adolescents in China, 22% of respondents reported that they experienced depressive symptoms.3 In an online survey of 1,054 Canadian adolescents, 43% said they were “very concerned” about the pandemic.4 In an online survey of 7,353 adolescents in the United States, 37% reported suicidal ideation during the pandemic compared to 17% in 2017.5 A Chinese study found that smartphone and internet addiction was significantly associated with increased levels of depressive symptoms during the pandemic.3 In a survey in the Philippines, 16.3% of adolescents reported moderate-to-severe psychological impairment during the pandemic; the rates of COVID-19–related anxiety were higher among girls vs boys.6 Alcohol and cannabis use increased among Canadian adolescents during the pandemic, according to an online survey.7 Adolescents with anorexia nervosa reported a 70% increase in poor eating habits and more thoughts associated with eating disorders during the pandemic.8 A Danish study found that children and adolescents newly diagnosed with obsessive-compulsive disorder (OCD) or who had completed treatment exhibited worsening OCD, anxiety, and depressive symptoms during the pandemic.9 An online survey of 6,196 Chinese adolescents found that those with a higher number of pre-pandemic adverse childhood experiences, such as abuse and neglect, had elevated posttraumatic stress symptoms and anxiety during the onset of the pandemic.10
Underlying causes of pandemic-induced distress
Limited social connectedness during the pandemic is a major reason for distress among adolescents. A review of 80 studies found that social isolation and loneliness as a result of social distancing and quarantining were associated with an increased risk of depression, anxiety, suicidal ideation, and self-harm.11 Parents’ stress about the risks of COVID-19 was correlated with worsening mental health in their adolescent children.12 A Chinese study found that the amount of time students spent on smartphones and social media doubled during the pandemic.13 In an online survey of 7,890 Chinese adolescents, greater social media, internet, and smartphone use was associated with increased anxiety and depression.14 This may be in part the result of adolescents spending time reading COVID-related news.
Coping mechanisms to increase well-being
Researchers have identified several positive coping mechanisms adolescents employed during the pandemic. Although some data suggest that increased internet use raises the risk of COVID-related distress, for certain adolescents, using social media to stay connected with friends and relatives was a buffer for feelings of loneliness and might have increased mental well-being.15 Other common coping mechanisms include relying on faith, volunteering, and starting new hobbies.16 During the pandemic, there were higher rates of playing outside and increased physical activity, which correlated with positive mental health outcomes.16 An online survey of 1,040 adolescents found that those who looked to the future optimistically and confidently had a higher health-related quality of life.17
Continuing an emphasis on adolescent well-being
Although data are limited, adolescents can continue to use these coping mechanisms to maintain their well-being, even if COVID-related restrictions are lifted or reimplemented. During these difficult times, it is imperative for adolescents to get the mental health services they need, and for psychiatric clinicians to continue to find avenues to promote resilience and mental wellness among young patients.
1. Leeb RT, Bitsko RH, Radhakrishnan L, et al. Mental health–related emergency department visits among children aged <18 years during the COVID-19 pandemic—United States, January 1-October 17, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(45):1675-1680. doi:10.15585/mmwr.mm6945a3
2. Oosterhoff B, Palmer CA, Wilson J, et al. Adolescents’ motivations to engage in social distancing during the COVID-19 pandemic: associations with mental and social health. J Adolesc Health. 2020;67(2):179-185. doi:10.1016/j.jadohealth.2020.05.004
3. Duan L, Shao X, Wang Y, et al. An investigation of mental health status of children and adolescents in China during the outbreak of COVID-19. J Affect Disord. 2020;275:112-118. doi:10.1016/j.jad.2020.06.029
4. Ellis WE, Dumas TM, Forbes LM. Physically isolated but socially connected: psychological adjustment and stress among adolescents during the initial COVID-19 crisis. Can J Behav Sci. 2020;52(3):177-187. doi:10.1037/cbs0000215
5. Murata S, Rezeppa T, Thoma B, et al. The psychiatric sequelae of the COVID-19 pandemic in adolescents, adults, and health care workers. Depress Anxiety. 2021;38(2):233-246. doi:10.1002/da.23120
6. Tee ML, Tee CA, Anlacan JP, et al. Psychological impact of COVID-19 pandemic in the Philippines. J Affect Disord. 2020;277:379-391. doi:10.1016/j.jad.2020.08.043
7. Dumas TM, Ellis W, Litt DM. What does adolescent substance use look like during the COVID-19 pandemic? Examining changes in frequency, social contexts, and pandemic-related predictors. J Adolesc Health. 2020;67(3):354-361. doi:10.1016/j.jadohealth.2020.06.018
8. Schlegl S, Maier J, Meule A, et al. Eating disorders in times of the COVID-19 pandemic—results from an online survey of patients with anorexia nervosa. Int J Eat Disord. 2020;53:1791-1800. doi:10.1002/eat.23374.
9. Nissen JB, Højgaard D, Thomsen PH. The immediate effect of COVID-19 pandemic on children and adolescents with obsessive compulsive disorder. BMC Psychiatry. 2020;20(1):511. doi:10.1186/s12888-020-02905-5
10. Guo J, Fu M, Liu D, et al. Is the psychological impact of exposure to COVID-19 stronger in adolescents with pre-pandemic maltreatment experiences? A survey of rural Chinese adolescents. Child Abuse Negl. 2020;110(Pt 2):104667. doi:10.1016/j.chiabu.2020.104667
11. Loades ME, Chatburn E, Higson-Sweeney N, et al. Rapid Systematic Review: The impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19. J Am Acad Child Adolesc Psychiatry. 2020;59(11):1218-1239.e3. doi:10.1016/j.jaac.2020.05.009
12. Spinelli M, Lionetti F, Setti A, et al. Parenting stress during the COVID-19 outbreak: socioeconomic and environmental risk factors and implications for children emotion regulation. Fam Process. 2021;60(2):639-653. doi:10.1111/famp.12601
13. Chen IH, Chen CY, Pakpour AH, et al. Internet-related behaviors and psychological distress among schoolchildren during COVID-19 school suspension. J Am Acad Child Adolesc Psychiatry. 2020;59(10):1099-1102.e1. doi:10.1016/j.jaac.2020.06.007
14. Li W, Zhang Y, Wang J, et al. Association of home quarantine and mental health among teenagers in Wuhan, China, during the COVID-19 pandemic. JAMA Pediatr. 2021;175(3):313-316. doi:10.1001/jamapediatrics.2020.5499
15. Janssen, LHC, Kullberg, MJ, Verkuil B, et al. Does the COVID-19 pandemic impact parents’ and adolescents’ well-being? An EMA-study on daily affect and parenting. PLoS One. 2020;15(10):e0240962. doi:10.1371/journal.pone.0240962
16. Banati P, Jones N, Youssef S. Intersecting vulnerabilities: the impacts of COVID-19 on the psycho-emotional lives of young people in low- and middle-income countries. Eur J Dev Res. 2020;32(5):1613-1638. doi:10.1057/s41287-020-00325-5
17. Ravens-Sieberer U, Kaman A, Otto C, et al. Mental health and quality of life in children and adolescents during the COVID-19 pandemic—results of the COPSY study. Dtsch Arztebl Int. 2020;117(48):828-829. doi:10.3238/arztebl.2020.0828
1. Leeb RT, Bitsko RH, Radhakrishnan L, et al. Mental health–related emergency department visits among children aged <18 years during the COVID-19 pandemic—United States, January 1-October 17, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(45):1675-1680. doi:10.15585/mmwr.mm6945a3
2. Oosterhoff B, Palmer CA, Wilson J, et al. Adolescents’ motivations to engage in social distancing during the COVID-19 pandemic: associations with mental and social health. J Adolesc Health. 2020;67(2):179-185. doi:10.1016/j.jadohealth.2020.05.004
3. Duan L, Shao X, Wang Y, et al. An investigation of mental health status of children and adolescents in China during the outbreak of COVID-19. J Affect Disord. 2020;275:112-118. doi:10.1016/j.jad.2020.06.029
4. Ellis WE, Dumas TM, Forbes LM. Physically isolated but socially connected: psychological adjustment and stress among adolescents during the initial COVID-19 crisis. Can J Behav Sci. 2020;52(3):177-187. doi:10.1037/cbs0000215
5. Murata S, Rezeppa T, Thoma B, et al. The psychiatric sequelae of the COVID-19 pandemic in adolescents, adults, and health care workers. Depress Anxiety. 2021;38(2):233-246. doi:10.1002/da.23120
6. Tee ML, Tee CA, Anlacan JP, et al. Psychological impact of COVID-19 pandemic in the Philippines. J Affect Disord. 2020;277:379-391. doi:10.1016/j.jad.2020.08.043
7. Dumas TM, Ellis W, Litt DM. What does adolescent substance use look like during the COVID-19 pandemic? Examining changes in frequency, social contexts, and pandemic-related predictors. J Adolesc Health. 2020;67(3):354-361. doi:10.1016/j.jadohealth.2020.06.018
8. Schlegl S, Maier J, Meule A, et al. Eating disorders in times of the COVID-19 pandemic—results from an online survey of patients with anorexia nervosa. Int J Eat Disord. 2020;53:1791-1800. doi:10.1002/eat.23374.
9. Nissen JB, Højgaard D, Thomsen PH. The immediate effect of COVID-19 pandemic on children and adolescents with obsessive compulsive disorder. BMC Psychiatry. 2020;20(1):511. doi:10.1186/s12888-020-02905-5
10. Guo J, Fu M, Liu D, et al. Is the psychological impact of exposure to COVID-19 stronger in adolescents with pre-pandemic maltreatment experiences? A survey of rural Chinese adolescents. Child Abuse Negl. 2020;110(Pt 2):104667. doi:10.1016/j.chiabu.2020.104667
11. Loades ME, Chatburn E, Higson-Sweeney N, et al. Rapid Systematic Review: The impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19. J Am Acad Child Adolesc Psychiatry. 2020;59(11):1218-1239.e3. doi:10.1016/j.jaac.2020.05.009
12. Spinelli M, Lionetti F, Setti A, et al. Parenting stress during the COVID-19 outbreak: socioeconomic and environmental risk factors and implications for children emotion regulation. Fam Process. 2021;60(2):639-653. doi:10.1111/famp.12601
13. Chen IH, Chen CY, Pakpour AH, et al. Internet-related behaviors and psychological distress among schoolchildren during COVID-19 school suspension. J Am Acad Child Adolesc Psychiatry. 2020;59(10):1099-1102.e1. doi:10.1016/j.jaac.2020.06.007
14. Li W, Zhang Y, Wang J, et al. Association of home quarantine and mental health among teenagers in Wuhan, China, during the COVID-19 pandemic. JAMA Pediatr. 2021;175(3):313-316. doi:10.1001/jamapediatrics.2020.5499
15. Janssen, LHC, Kullberg, MJ, Verkuil B, et al. Does the COVID-19 pandemic impact parents’ and adolescents’ well-being? An EMA-study on daily affect and parenting. PLoS One. 2020;15(10):e0240962. doi:10.1371/journal.pone.0240962
16. Banati P, Jones N, Youssef S. Intersecting vulnerabilities: the impacts of COVID-19 on the psycho-emotional lives of young people in low- and middle-income countries. Eur J Dev Res. 2020;32(5):1613-1638. doi:10.1057/s41287-020-00325-5
17. Ravens-Sieberer U, Kaman A, Otto C, et al. Mental health and quality of life in children and adolescents during the COVID-19 pandemic—results of the COPSY study. Dtsch Arztebl Int. 2020;117(48):828-829. doi:10.3238/arztebl.2020.0828
More on stigma
I just finished reading your editorial “A PSYCHIATRIC MANIFESTO: Stigma is hate speech and a hate crime” (
Our son went from an honor roll student before the pandemic to a child I barely recognized. Approximately 6 months into the pandemic, he was using drugs, vaping nicotine, destroying our property, and eloping at night. The journey of watching his decline and getting him help was agonizing. But the stigma around what was happening to him was an entirely separate animal.
Our society vilifies, ridicules, dismisses, and often makes fun of those with mental health issues. I experience it daily with my son and am on constant guard to shoot down any comments and to calmly teach those who say such cruel things. But the shame my son feels is the most devastating part. Although we keep reminding him that his condition is a medical condition like diabetes or heart disease, for a teenage boy, that makes no sense. He just wants to be “normal.” And living in a world that rarely represents mental illness this way, it’s almost a lost cause to get him to let go of this shame. All we can do is love him, be there for him, support him, and do what we can to educate those around us about the stigma of mental illness.
What a powerful and accurate article. Thank you for putting into words what I have been thinking and feeling, and for being as outraged as we are at how this vulnerable population is treated. My husband is a psychiatrist and we live in an affluent urban area, so we are not in the middle of nowhere with no knowledge of what is happening to our son. And despite that, we still suffer from the stigma.
Thank you, Dr. Nasrallah.
Name withheld
I need to take a moment to thank you for your editorial about stigma being hate speech and a hate crime. I really agree with you, and I think the way you formulated and articulated this message is very compelling.
I have focused on normalizing mental health differences among entrepreneurs as a destigmatization strategy (see https://www.sciencedirect.com/science/article/pii/S0883902622000027 and https://link.springer.com/article/10.1007/s11187-018-0059-8). Entrepreneurs clearly illustrate the fallacy of stigma. As a simple example, Elon Musk—the wealthiest person in the world—talks openly about being autistic, and possibly bipolar. These mental health differences help him create jobs and contribute to our shared prosperity. Nothing to be ashamed of there.
Thanks again for being such an effective advocate.
Michael A. Freeman, MD
Kentfield, California
Continue to: Thank you...
Thank you so much for your “Psychiatric Manifesto.” I will do my best to disseminate it amongst colleagues, patients, friends, family, and as many others as possible.
Daniel N. Pistone, MD
San Francisco, California
Once again, your words hit the pin on the head.
Robert W. Pollack, MD, ABPN, DLFAPA
Fort Myers, Florida
I just finished reading your editorial “A PSYCHIATRIC MANIFESTO: Stigma is hate speech and a hate crime” (
Our son went from an honor roll student before the pandemic to a child I barely recognized. Approximately 6 months into the pandemic, he was using drugs, vaping nicotine, destroying our property, and eloping at night. The journey of watching his decline and getting him help was agonizing. But the stigma around what was happening to him was an entirely separate animal.
Our society vilifies, ridicules, dismisses, and often makes fun of those with mental health issues. I experience it daily with my son and am on constant guard to shoot down any comments and to calmly teach those who say such cruel things. But the shame my son feels is the most devastating part. Although we keep reminding him that his condition is a medical condition like diabetes or heart disease, for a teenage boy, that makes no sense. He just wants to be “normal.” And living in a world that rarely represents mental illness this way, it’s almost a lost cause to get him to let go of this shame. All we can do is love him, be there for him, support him, and do what we can to educate those around us about the stigma of mental illness.
What a powerful and accurate article. Thank you for putting into words what I have been thinking and feeling, and for being as outraged as we are at how this vulnerable population is treated. My husband is a psychiatrist and we live in an affluent urban area, so we are not in the middle of nowhere with no knowledge of what is happening to our son. And despite that, we still suffer from the stigma.
Thank you, Dr. Nasrallah.
Name withheld
I need to take a moment to thank you for your editorial about stigma being hate speech and a hate crime. I really agree with you, and I think the way you formulated and articulated this message is very compelling.
I have focused on normalizing mental health differences among entrepreneurs as a destigmatization strategy (see https://www.sciencedirect.com/science/article/pii/S0883902622000027 and https://link.springer.com/article/10.1007/s11187-018-0059-8). Entrepreneurs clearly illustrate the fallacy of stigma. As a simple example, Elon Musk—the wealthiest person in the world—talks openly about being autistic, and possibly bipolar. These mental health differences help him create jobs and contribute to our shared prosperity. Nothing to be ashamed of there.
Thanks again for being such an effective advocate.
Michael A. Freeman, MD
Kentfield, California
Continue to: Thank you...
Thank you so much for your “Psychiatric Manifesto.” I will do my best to disseminate it amongst colleagues, patients, friends, family, and as many others as possible.
Daniel N. Pistone, MD
San Francisco, California
Once again, your words hit the pin on the head.
Robert W. Pollack, MD, ABPN, DLFAPA
Fort Myers, Florida
I just finished reading your editorial “A PSYCHIATRIC MANIFESTO: Stigma is hate speech and a hate crime” (
Our son went from an honor roll student before the pandemic to a child I barely recognized. Approximately 6 months into the pandemic, he was using drugs, vaping nicotine, destroying our property, and eloping at night. The journey of watching his decline and getting him help was agonizing. But the stigma around what was happening to him was an entirely separate animal.
Our society vilifies, ridicules, dismisses, and often makes fun of those with mental health issues. I experience it daily with my son and am on constant guard to shoot down any comments and to calmly teach those who say such cruel things. But the shame my son feels is the most devastating part. Although we keep reminding him that his condition is a medical condition like diabetes or heart disease, for a teenage boy, that makes no sense. He just wants to be “normal.” And living in a world that rarely represents mental illness this way, it’s almost a lost cause to get him to let go of this shame. All we can do is love him, be there for him, support him, and do what we can to educate those around us about the stigma of mental illness.
What a powerful and accurate article. Thank you for putting into words what I have been thinking and feeling, and for being as outraged as we are at how this vulnerable population is treated. My husband is a psychiatrist and we live in an affluent urban area, so we are not in the middle of nowhere with no knowledge of what is happening to our son. And despite that, we still suffer from the stigma.
Thank you, Dr. Nasrallah.
Name withheld
I need to take a moment to thank you for your editorial about stigma being hate speech and a hate crime. I really agree with you, and I think the way you formulated and articulated this message is very compelling.
I have focused on normalizing mental health differences among entrepreneurs as a destigmatization strategy (see https://www.sciencedirect.com/science/article/pii/S0883902622000027 and https://link.springer.com/article/10.1007/s11187-018-0059-8). Entrepreneurs clearly illustrate the fallacy of stigma. As a simple example, Elon Musk—the wealthiest person in the world—talks openly about being autistic, and possibly bipolar. These mental health differences help him create jobs and contribute to our shared prosperity. Nothing to be ashamed of there.
Thanks again for being such an effective advocate.
Michael A. Freeman, MD
Kentfield, California
Continue to: Thank you...
Thank you so much for your “Psychiatric Manifesto.” I will do my best to disseminate it amongst colleagues, patients, friends, family, and as many others as possible.
Daniel N. Pistone, MD
San Francisco, California
Once again, your words hit the pin on the head.
Robert W. Pollack, MD, ABPN, DLFAPA
Fort Myers, Florida
Then and now: Inflammatory bowel diseases
(IBD) creating a whole new landscape for the disease.
In 2007, IBD seemed to be primarily a disease of Caucasian and Jewish ancestry. While prevalence of IBD is still highest in the Western world, there is now increasing incidence, even accounting for detection bias, in people of all other ancestries globally. Incidence of IBD in children under the age of 18 years is also rising. Patients with IBD are living longer and, despite the notion that IBD is a disease primarily of younger adults, nearly one-third of Americans with IBD are 60 years and older.
“Adalimumab aids in Crohn’s disease” read the front page of the inaugural issue of GI & Hepatology News in January 2007. The article highlighted the GAIN study, which demonstrated that patients who lost response to infliximab responded to adalimumab, the second anti-tumor necrosis factor (TNF) agent approved for the treatment of Crohn’s disease and subsequently ulcerative colitis. Over the subsequent 15 years, the armamentarium of treatment options for Crohn’s disease and ulcerative colitis have rapidly proliferated: there are now four anti–tumor necrosis factor (TNF) agents, two anti-integrin agents, two anti-interleukin agents, two Janus kinase inhibitors and a sphingosine-1 receptor modulator approved for the treatment of IBD. Many more promising treatment options are in trials. Other mechanisms are under investigation as well, including antimicrobial therapies for ulcerative colitis and stem cell therapeutics for the treatment of refractory perianal fistulizing Crohn’s disease. Perhaps even more novel – dietary therapies are more rigorously under investigation.
“Ulcerative colitis guidelines endorse combined therapy” reads another headline from the inaugural GI & Hepatology News issue. The article discusses the European Crohn’s and Colitis Organisation’s consensus guideline that topical and systemic agents used together are superior to either used alone, referring specifically to mesalamine, both systemic and topical as well as the additional of topical corticosteroid to systemic mesalamine. Combination therapy has a completely new meaning in modern times. With the publication of the SONIC trial in 2010, combination therapy referred to an anti-TNF in combination with an immunomodulator for the ensuing decade. However, in this new era of IBD treatment, combination therapy could also mean a biologic with a small molecule or even combination biologics, for which there is an ongoing randomized controlled trial. On the topic of treatment strategies, one of the biggest shifts in the IBD treatment paradigm is the bottom-up versus top-down approach of treatment, with increasing evidence to suggest that early biologic initiation is more effective, especially in patients with Crohn’s disease. Therapeutic drug monitoring is mainstream. Treat-to-target strategies to achieve more stringent outcomes, such as biomarker, endoscopic and histologic normalization, especially in ulcerative colitis, have evolved to become the norm in 2022.
The combination of increased treatment options, decreased reliance on corticosteroids and stringent treatment strategies have resulted in improved outcomes: IBD-related hospitalizations, surgeries, and even mortality have declined since 2007. The growing recognition and focus on extra-intestinal manifestations, including fatigue, and the gut-brain axis are important steps to improving the overall quality of life of patients with IBD. Beyond treating the disease, we are now learning how to treat the patient. We will be developing personalized strategies to identify the right patient for the right treatment, including patient-level clinical and biologic markers. We need to identify those who are at risk for IBD to prevent the disease at a preclinical phase. Concomitantly, we must continue the quest to cure the disease!
Dr. Kochar is a gastroenterologist and inflammatory bowel disease specialist at Massachusetts General Hospital and a physician investigator in the clinical and translational epidemiology unit at The Mongan Institute, both in Boston. She has no relevant disclosures.
(IBD) creating a whole new landscape for the disease.
In 2007, IBD seemed to be primarily a disease of Caucasian and Jewish ancestry. While prevalence of IBD is still highest in the Western world, there is now increasing incidence, even accounting for detection bias, in people of all other ancestries globally. Incidence of IBD in children under the age of 18 years is also rising. Patients with IBD are living longer and, despite the notion that IBD is a disease primarily of younger adults, nearly one-third of Americans with IBD are 60 years and older.
“Adalimumab aids in Crohn’s disease” read the front page of the inaugural issue of GI & Hepatology News in January 2007. The article highlighted the GAIN study, which demonstrated that patients who lost response to infliximab responded to adalimumab, the second anti-tumor necrosis factor (TNF) agent approved for the treatment of Crohn’s disease and subsequently ulcerative colitis. Over the subsequent 15 years, the armamentarium of treatment options for Crohn’s disease and ulcerative colitis have rapidly proliferated: there are now four anti–tumor necrosis factor (TNF) agents, two anti-integrin agents, two anti-interleukin agents, two Janus kinase inhibitors and a sphingosine-1 receptor modulator approved for the treatment of IBD. Many more promising treatment options are in trials. Other mechanisms are under investigation as well, including antimicrobial therapies for ulcerative colitis and stem cell therapeutics for the treatment of refractory perianal fistulizing Crohn’s disease. Perhaps even more novel – dietary therapies are more rigorously under investigation.
“Ulcerative colitis guidelines endorse combined therapy” reads another headline from the inaugural GI & Hepatology News issue. The article discusses the European Crohn’s and Colitis Organisation’s consensus guideline that topical and systemic agents used together are superior to either used alone, referring specifically to mesalamine, both systemic and topical as well as the additional of topical corticosteroid to systemic mesalamine. Combination therapy has a completely new meaning in modern times. With the publication of the SONIC trial in 2010, combination therapy referred to an anti-TNF in combination with an immunomodulator for the ensuing decade. However, in this new era of IBD treatment, combination therapy could also mean a biologic with a small molecule or even combination biologics, for which there is an ongoing randomized controlled trial. On the topic of treatment strategies, one of the biggest shifts in the IBD treatment paradigm is the bottom-up versus top-down approach of treatment, with increasing evidence to suggest that early biologic initiation is more effective, especially in patients with Crohn’s disease. Therapeutic drug monitoring is mainstream. Treat-to-target strategies to achieve more stringent outcomes, such as biomarker, endoscopic and histologic normalization, especially in ulcerative colitis, have evolved to become the norm in 2022.
The combination of increased treatment options, decreased reliance on corticosteroids and stringent treatment strategies have resulted in improved outcomes: IBD-related hospitalizations, surgeries, and even mortality have declined since 2007. The growing recognition and focus on extra-intestinal manifestations, including fatigue, and the gut-brain axis are important steps to improving the overall quality of life of patients with IBD. Beyond treating the disease, we are now learning how to treat the patient. We will be developing personalized strategies to identify the right patient for the right treatment, including patient-level clinical and biologic markers. We need to identify those who are at risk for IBD to prevent the disease at a preclinical phase. Concomitantly, we must continue the quest to cure the disease!
Dr. Kochar is a gastroenterologist and inflammatory bowel disease specialist at Massachusetts General Hospital and a physician investigator in the clinical and translational epidemiology unit at The Mongan Institute, both in Boston. She has no relevant disclosures.
(IBD) creating a whole new landscape for the disease.
In 2007, IBD seemed to be primarily a disease of Caucasian and Jewish ancestry. While prevalence of IBD is still highest in the Western world, there is now increasing incidence, even accounting for detection bias, in people of all other ancestries globally. Incidence of IBD in children under the age of 18 years is also rising. Patients with IBD are living longer and, despite the notion that IBD is a disease primarily of younger adults, nearly one-third of Americans with IBD are 60 years and older.
“Adalimumab aids in Crohn’s disease” read the front page of the inaugural issue of GI & Hepatology News in January 2007. The article highlighted the GAIN study, which demonstrated that patients who lost response to infliximab responded to adalimumab, the second anti-tumor necrosis factor (TNF) agent approved for the treatment of Crohn’s disease and subsequently ulcerative colitis. Over the subsequent 15 years, the armamentarium of treatment options for Crohn’s disease and ulcerative colitis have rapidly proliferated: there are now four anti–tumor necrosis factor (TNF) agents, two anti-integrin agents, two anti-interleukin agents, two Janus kinase inhibitors and a sphingosine-1 receptor modulator approved for the treatment of IBD. Many more promising treatment options are in trials. Other mechanisms are under investigation as well, including antimicrobial therapies for ulcerative colitis and stem cell therapeutics for the treatment of refractory perianal fistulizing Crohn’s disease. Perhaps even more novel – dietary therapies are more rigorously under investigation.
“Ulcerative colitis guidelines endorse combined therapy” reads another headline from the inaugural GI & Hepatology News issue. The article discusses the European Crohn’s and Colitis Organisation’s consensus guideline that topical and systemic agents used together are superior to either used alone, referring specifically to mesalamine, both systemic and topical as well as the additional of topical corticosteroid to systemic mesalamine. Combination therapy has a completely new meaning in modern times. With the publication of the SONIC trial in 2010, combination therapy referred to an anti-TNF in combination with an immunomodulator for the ensuing decade. However, in this new era of IBD treatment, combination therapy could also mean a biologic with a small molecule or even combination biologics, for which there is an ongoing randomized controlled trial. On the topic of treatment strategies, one of the biggest shifts in the IBD treatment paradigm is the bottom-up versus top-down approach of treatment, with increasing evidence to suggest that early biologic initiation is more effective, especially in patients with Crohn’s disease. Therapeutic drug monitoring is mainstream. Treat-to-target strategies to achieve more stringent outcomes, such as biomarker, endoscopic and histologic normalization, especially in ulcerative colitis, have evolved to become the norm in 2022.
The combination of increased treatment options, decreased reliance on corticosteroids and stringent treatment strategies have resulted in improved outcomes: IBD-related hospitalizations, surgeries, and even mortality have declined since 2007. The growing recognition and focus on extra-intestinal manifestations, including fatigue, and the gut-brain axis are important steps to improving the overall quality of life of patients with IBD. Beyond treating the disease, we are now learning how to treat the patient. We will be developing personalized strategies to identify the right patient for the right treatment, including patient-level clinical and biologic markers. We need to identify those who are at risk for IBD to prevent the disease at a preclinical phase. Concomitantly, we must continue the quest to cure the disease!
Dr. Kochar is a gastroenterologist and inflammatory bowel disease specialist at Massachusetts General Hospital and a physician investigator in the clinical and translational epidemiology unit at The Mongan Institute, both in Boston. She has no relevant disclosures.
A valuable learning experience
It was a pleasure to serve as editor-in-chief (EIC) of GI & Hepatology News from 2011 to 2016. As the second EIC of the newspaper, I was preceded by Dr. Charles Lightdale – big shoes to fill! I was fortunate to attract a strong group of associate editors who covered many key areas of interest for the paper’s readership. With the enthusiastic support of American Gastroenterological Association staff members, we published once-monthly and received generally positive feedback from readers – predominantly U.S.-based AGA members.
Serving as EIC was also a learning opportunity for me. A number of potentially newsworthy items were brought to my attention – some of which I would not otherwise have seen. Although not all were of direct relevance to the readership, I believe that most of those we published were of value.
One rewarding aspect of the editorship was the opportunity to liaise with those experts from whom I solicited commentaries on some of our featured items. These busy individuals were consistently generous with their time and expertise, and I believe that their contributions added to the paper’s overall appeal.
I initiated the inclusion of two DDSEP questions per edition, and am pleased that this feature continues. One less successful venture was the attempt at a Correspondence section, which ultimately proved too cumbersome to maintain.
I congratulate the AGA on the 15th anniversary of GIHN, and I wish the current EIC, Dr. Megan Adams, and her editorial colleagues continued success in providing this benefit to AGA members.
Colin W. Howden, MD, AGAF, is professor emeritus in the division of gastroenterology, department of medicine, at the University of Tennessee, Memphis. He is a consultant for Allakos, Ironwood, Phathom, and RedHill Biopharma. He is a member of speakers’ bureaus for Alnylam, RedHill Biopharma, and Sanofi/Genzyme. He owns stock in Antibe Therapeutics.
It was a pleasure to serve as editor-in-chief (EIC) of GI & Hepatology News from 2011 to 2016. As the second EIC of the newspaper, I was preceded by Dr. Charles Lightdale – big shoes to fill! I was fortunate to attract a strong group of associate editors who covered many key areas of interest for the paper’s readership. With the enthusiastic support of American Gastroenterological Association staff members, we published once-monthly and received generally positive feedback from readers – predominantly U.S.-based AGA members.
Serving as EIC was also a learning opportunity for me. A number of potentially newsworthy items were brought to my attention – some of which I would not otherwise have seen. Although not all were of direct relevance to the readership, I believe that most of those we published were of value.
One rewarding aspect of the editorship was the opportunity to liaise with those experts from whom I solicited commentaries on some of our featured items. These busy individuals were consistently generous with their time and expertise, and I believe that their contributions added to the paper’s overall appeal.
I initiated the inclusion of two DDSEP questions per edition, and am pleased that this feature continues. One less successful venture was the attempt at a Correspondence section, which ultimately proved too cumbersome to maintain.
I congratulate the AGA on the 15th anniversary of GIHN, and I wish the current EIC, Dr. Megan Adams, and her editorial colleagues continued success in providing this benefit to AGA members.
Colin W. Howden, MD, AGAF, is professor emeritus in the division of gastroenterology, department of medicine, at the University of Tennessee, Memphis. He is a consultant for Allakos, Ironwood, Phathom, and RedHill Biopharma. He is a member of speakers’ bureaus for Alnylam, RedHill Biopharma, and Sanofi/Genzyme. He owns stock in Antibe Therapeutics.
It was a pleasure to serve as editor-in-chief (EIC) of GI & Hepatology News from 2011 to 2016. As the second EIC of the newspaper, I was preceded by Dr. Charles Lightdale – big shoes to fill! I was fortunate to attract a strong group of associate editors who covered many key areas of interest for the paper’s readership. With the enthusiastic support of American Gastroenterological Association staff members, we published once-monthly and received generally positive feedback from readers – predominantly U.S.-based AGA members.
Serving as EIC was also a learning opportunity for me. A number of potentially newsworthy items were brought to my attention – some of which I would not otherwise have seen. Although not all were of direct relevance to the readership, I believe that most of those we published were of value.
One rewarding aspect of the editorship was the opportunity to liaise with those experts from whom I solicited commentaries on some of our featured items. These busy individuals were consistently generous with their time and expertise, and I believe that their contributions added to the paper’s overall appeal.
I initiated the inclusion of two DDSEP questions per edition, and am pleased that this feature continues. One less successful venture was the attempt at a Correspondence section, which ultimately proved too cumbersome to maintain.
I congratulate the AGA on the 15th anniversary of GIHN, and I wish the current EIC, Dr. Megan Adams, and her editorial colleagues continued success in providing this benefit to AGA members.
Colin W. Howden, MD, AGAF, is professor emeritus in the division of gastroenterology, department of medicine, at the University of Tennessee, Memphis. He is a consultant for Allakos, Ironwood, Phathom, and RedHill Biopharma. He is a member of speakers’ bureaus for Alnylam, RedHill Biopharma, and Sanofi/Genzyme. He owns stock in Antibe Therapeutics.











