User login
Perioperative statins: More than lipid-lowering?
Soon, the checklist for internists seeing patients about to undergo surgery may include prescribing one of the lipid-lowering hydroxymethylglutaryl-CoA reductase inhibitors, also called statins.
Statins? Not long ago, we were debating whether patients who take statins should stop taking them before surgery, based on the manufacturers’ recommendations.1 The discussion, however, has changed to whether patients who have never received a statin should be started on one before surgery to provide immediate prophylaxis against cardiac morbidity, and how much harm long-term statin users face if these drugs are withheld perioperatively.
The evidence is still very preliminary and based mostly on studies in animals and retrospective studies in people. However, an expanding body of indirect evidence suggests that these drugs are beneficial in this situation.
In this review, we discuss the mechanisms by which statins may protect the heart in the short term, drawing on data from animal and human studies of acute myocardial infarction, and we review the current (albeit limited) data from the perioperative setting.
FEW INTERVENTIONS DECREASE RISK
Each year, approximately 50,000 patients suffer a perioperative cardiovascular event; the incidence of myocardial infarction during or after noncardiac surgery is 2% to 3%.2 The primary goal of preoperative cardiovascular risk assessment is to predict and avert these events.
But short of canceling surgery, few interventions have been found to reduce a patient’s risk. For example, a landmark study in 2004 cast doubt on the efficacy of preoperative coronary revascularization.3 Similarly, although early studies of beta-blockers were promising4,5 and although most internists prescribe these drugs before surgery, more recent studies have cast doubt on their efficacy, particularly in patients at low risk undergoing intermediate-risk (rather than vascular) surgery.6–8
This changing clinical landscape has prompted a search for new strategies for perioperative risk-reduction. Several recent studies have placed statins in the spotlight.
POTENTIAL MECHANISMS OF SHORT-TERM BENEFIT
Statins have been proven to save lives when used long-term, but how could this class of drugs, designed to prevent the accumulation of arterial plaques by lowering low-density lipoprotein cholesterol (LDL-C) levels, have any short-term impact on operative outcomes? Although LDL-C reduction is the principal mechanism of action of statins, not all of the benefit can be ascribed to this mechanism.9 The answer may lie in their “pleiotropic” effects—ie, actions other than LDL-C reduction.
The more immediate pleiotropic effects of statins in the proinflammatory and prothrombotic environment of the perioperative period are thought to include improved endothelial function (both antithrombotic function and vasomotor function in response to ischemic stress), enhanced stability of atherosclerotic plaques, decreased oxidative stress, and decreased vascular inflammation.10–12
EVIDENCE FROM ANIMAL STUDIES
Experiments in animals suggest that statins, given shortly before or after a cardiovascular event, confer benefit before any changes in LDL-C are measurable.
Lefer et al13 found that simvastatin (Zocor), given 18 hours before an ischemic episode in rats, blunted the inflammatory response in cardiac reperfusion injury. Not only was reperfusion injury significantly less in the hearts of the rats that received simvastatin than in the saline control group, but the simvastatin-treated hearts also expressed fewer neutrophil adhesion molecules such as P-selectin, and they had more basal release of nitric oxide, the potent endothelial-derived vasodilator with antithrombotic, anti-inflammatory, and antiproliferative effects.14 These results suggest that statins may improve endothelial function acutely, particularly during ischemic stress.
Osborne et al15 fed rabbits a cholesterol-rich diet plus either lovastatin (Mevacor) or placebo. After 2 weeks, the rabbits underwent either surgery to induce a myocardial infarction or a sham procedure. Regardless of the pretreatment, biopsies of the aorta did not reveal any atherosclerosis; yet the lovastatin-treated rabbits sustained less myocardial ischemic damage and they had more endothelium-mediated vasodilatation.
Statin therapy also may improve cerebral ischemia outcomes in animal models.14,16
Sironi et al16 induced strokes in rats by occluding the middle cerebral artery. The rats received either simvastatin or vehicle for 3 days before the stroke or immediately afterwards. Even though simvastatin did not have enough time to affect the total cholesterol level, rats treated with simvastatin had smaller infarcts (as measured by magnetic resonance imaging) and produced more nitric oxide.
Comment. Taken together, these studies offer tantalizing evidence that statins have short-term, beneficial nonlipid effects and may reduce not only the likelihood of an ischemic event, but—should one occur—the degree of tissue damage that ensues.
EFFECTS OF STATINS IN ACUTE CORONARY SYNDROME
The National Registry of Myocardial Infarction17 is a prospective, observational database of all patients with acute myocardial infarction admitted to 1,230 participating hospitals throughout the United States. In an analysis from this cohort, patients were divided into four groups: those receiving statins before and after admission, those receiving statins only before admission, those receiving statins only after admission, and those who never received statins.
Compared with those who never received statins, fewer patients who received them both before and after admission died while in the hospital (unadjusted odds ratio 0.23, 95% confidence interval [CI] 0.22–0.25), and the odds ratio for those who received statins for the first time was 0.31 (95% CI 0.29–0.33). Patients who stopped receiving a statin on admission were more likely to die than were patients who never received statins (odds ratio 1.09, 95% CI 1.03–1.15). These trends held true even when adjustments were made for potential confounding factors.
Comment. Unmeasured confounding factors (such as the inability to take pills due to altered mental status or the different practice styles of the providers who chose to discontinue statins) might have affected the results. Nevertheless, these results suggest that the protective effects of statins stop almost immediately when these drugs are discontinued, and that there may even be an adverse “rebound” effect when patients who have been taking these drugs for a long time stop taking them temporarily.
The Platelet Receptor Inhibition in Ischemic Syndrome Management trial,18 in a subgroup analysis, had nearly identical findings. In the main part of this trial, patients with coronary artery disease and chest pain at rest or accelerating pain in the last 24 hours were randomized to receive tirofiban (Aggrastat) or heparin. Complete data on statin use were available for 1,616 (50%) of the 3,232 patients in this trial, and the rate of the primary end point (death, myocardial infarction, or recurrent ischemia) was analyzed on the basis of statin therapy in this subgroup.
Comment. Together, these data lead to the conclusion that, when admitted for either acute myocardial infarction or acute coronary syndrome, patients already receiving statins should not have them stopped, and those who had not been receiving statins should receive them immediately. The safety of these medications in the acute setting appears excellent: in the Myocardial Ischemia Reduction With Acute Cholesterol Lowering (MIRACL)12 and the Pravastatin or Atorvastatin Evaluation and Infection Therapy (PROVE-IT)11 trials, fewer than 5% of statin-treated patients had transient elevations in transaminase levels, and no cases of rhabdomyolysis were reported.
PERIOPERATIVE STATIN STUDIES
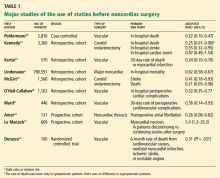
The data on perioperative statin use are mostly observational and retrospective and fall into essentially four surgical categories: coronary artery bypass grafting (CABG), carotid endarterectomy,19,20 noncardiac vascular surgery, and major noncardiac surgery. Two meta-analyses have also evaluated the data.21,22 The only randomized controlled trial (performed by Durazzo et al23) was small and was carried out at a single center in vascular surgery patients, and the event rate was low.
Current recommendations from the National Cholesterol Education Program (NCEP)24 say that patients who need CABG, have peripheral arterial disease, have an abdominal aortic aneurysm, or have cerebrovascular disease should already be on a statin to achieve an LDL-C goal level of less than 100 mg/dL, with an optional goal of less than 70 mg/dL, independent of surgery.
Since not all patients who should be on statins are actually on them, questions arise:
- Is it important (and safe) to start statin treatment preoperatively?
- Will patients with cardiovascular risk factors but without known cardiovascular disease benefit from statins perioperatively?
Noncardiac vascular surgery
Multiple retrospective studies have evaluated the effect of statins in patients undergoing major noncardiac vascular surgery.25–32
Kertai et al25 evaluated 570 patients in Holland who underwent elective open surgery for infrarenal abdominal aortic aneurysms between 1991 and 2001, looking for an association between statin use and the incidence of perioperative death from myocardial infarction. Only 162 of the 570 patients had been on long-term statin therapy before the surgery. The use of statins was only one of many known baseline characteristics that were significantly different between the two groups, including age, body mass index, known coronary artery disease, and use of angiotensin-converting enzyme inhibitors and beta-blockers. In univariate analysis, statins appeared to be protective: 6 (3.7%) of the patients in the statin group died of a myocardial infarction, compared with 45 (11%) of those in the nostatin group. A multivariate analysis yielded similar findings, with an odds ratio of 0.24 (95% CI 0.11–0.54).
Ward et al27 performed a very similar retrospective study, with similar findings. In 446 patients who underwent surgery for infrarenal abdominal aortic aneurysm, statin therapy was associated with a significantly lower incidence of the combined end point of death, myocardial infarction, stroke, and major peripheral vascular complications, with an adjusted odds ratio of 0.36 (95% CI 0.14–0.93).
Poldermans et al26 noted similar findings in a case-control study of noncardiac vascular surgery patients. Statin users had a much lower perioperative risk of death than did nonusers, with an adjusted odds ratio of 0.22 (95% CI 0.10–0.47).
O’Neil-Callahan et al,28 in a cohort study, found that statin users had fewer perioperative cardiac complications, with an adjusted odds ratio of 0.49 (95% CI 0.28–0.84, P = .009).
Dogma of withdrawing statins before major surgery is challenged
Le Manach et al33 reviewed the outcomes for all patients of a single hospital in Paris who underwent nonemergency infrarenal aortic procedures between January 2001 and December 2004. In January 2004, the hospital instituted guidelines to ensure that patients on statins continue taking them up to the evening before surgery and that statins be restarted on the first postoperative day (via nasogastric tube if necessary). Before 2004, there had been no specific guidelines, and patients on statins did not receive them for a median of 4 days postoperatively. Types of procedures were similar during the two time periods, as were the rates of beta-blocker use, preoperative revascularization, venous thromboembolism prophylaxis, and perioperative blood pressure control. After surgery, topononin I levels were measured in all patients as surveillance for cardiac events, and were defined as elevated when greater than 0.2 ng/mL.
Compared with patients not on statins at all, those treated with statins continuously throughout the perioperative period (after January 2004) had a lower rate of elevated troponin (relative risk 0.38). In contrast, those who had their statins transiently discontinued perioperatively (prior to 2004) had troponin elevations more often than those who had never been treated (relative risk 2.1). This suggested an over fivefold risk reduction (P < .001) conferred by not discontinuing statins in the immediate postoperative period. This finding was maintained after multivariate adjustment: statin withdrawal was associated with a 2.9-fold (95% CI 1.6–5.5) increase in the risk of cardiac enzyme elevations postoperatively. No fewer deaths were noted, but the study was not powered to detect a mortality difference.
Comment. Although secular trends cannot be entirely discounted as contributing to these findings, the prompt increase in cardiac events after just 4 days of statin withdrawal adds to the growing body of evidence suggesting that statin discontinuation can have harmful acute effects. It also brings up the question: Can starting statins benefit patients in the same time period?
Should statins be started before vascular surgery?
Schouten et al32 evaluated the effects of newly started or continued statin treatment in patients undergoing major elective vascular surgery. Patients were screened before surgery and started on statins if they were not already receiving them and their total cholesterol levels were elevated; new users received the medication for about 40 days before surgery. Of the 981 screened patients, 44 (5%) were newly started on statins and 182 (19%) were continued on their therapy. Perioperative death or myocardial infarction occurred in 22 (8.8%) of the statin users and 111 (14.7%) of the nonusers, a statistically significant difference. Temporary discontinuation (median 1 day) of statins in this study due to the inability to take an oral medication did not appear to affect the likelihood of a myocardial infarction.
Durazzo et al23 performed a single-center, randomized, prospective, placebo-controlled, double-blind clinical trial of atorvastatin (Lipitor) 20 mg daily vs placebo in 100 patients undergoing noncardiac arterial vascular surgery. Patients were excluded if they had previously used medications to treat dyslipidemia, recently had a cardiovascular event, or had contraindications to statin treatment such as a baseline creatinine level greater than 2.0 mg/dL or severe hepatic disease. The intervention group received atorvastatin starting at least 2 weeks before surgery for a total of 45 days. Patients were then continued or started on a statin after surgery if their LDL-C level was greater than 100 mg/dL. Beta-blocker use was recommended “on the basis of current guidelines.”
One month after surgery, the LDL-C level was statistically significantly lower in the atorvastatin group. Since most patients did not continue or start statin therapy after the 45-day treatment period, the LDL-C levels were not statistically different at 3 and 6 months after surgery.
At 6 months, the rate of the primary end point (death from cardiovascular causes, nonfatal acute myocardial infarction, ischemic stroke, or unstable angina) was 26.0% in the placebo group and 8.0% in the atorvastatin group, a statistically significant difference. Three patients in the atorvastatin group had cardiac events in the first 10 days after surgery, compared with 11 patients in the placebo group. Thirteen of the 17 total cardiac events took place within 10 days after surgery.
One of the atorvastatin patients developed rhabdomyolysis and elevated aminotransferase levels.
Major noncardiac surgery
Lindenauer et al2 performed a retrospective cohort study of surgical patients who were at least 18 years old and survived beyond the second hospital day. Patients were divided into a group receiving any form of lipid-lowering treatment (of whom more than 90% were taking statins) and a group that had never never received a lipid-lowering drug or only started one on the third day of the hospitalization or later. The period of study was from January 1, 2000, to December 31, 2001.
In all, 780,591 patients from 329 hospitals throughout the United States were included, of whom only 77,082 (9.9%) received lipid-lowering therapy. Eight percent of the patients underwent vascular surgery. Not surprisingly, the treated patients were more likely to have a history of hypertension, diabetes, ischemic heart disease, or hyperlipidemia. They also were more likely to have a vascular procedure performed, to have two or more cardiac risk factors (high-risk surgery, ischemic heart disease, congestive heart failure, cerebrovascular disease, renal insufficiency, or diabetes mellitus), and to be treated with beta-blockers and angiotensin-converting enzyme inhibitors, but they were less likely to have high-risk and emergency surgery performed.
The primary end point, perioperative death, occurred in 2.13% of the treated patients and 3.05% of the nontreated group. Compared with the rate in a propensity-matched cohort, the odds ratio adjusted for unbalanced covariates was 0.62 (95% CI 0.58–0.67) in favor of lipid treatment. Stratification by cardiac risk index revealed a number needed to treat of 186 for those with no risk factors, 60 for those with two risk factors, and 30 for those with four or more risk factors.
Unfortunately, this analysis was not able to take into account whether and for how long patients were receiving lipid-lowering therapy before hospitalization. It therefore does not answer the questions of whether starting lipid-lowering therapy before surgery is beneficial or whether stopping it is harmful. It also does not shed light on whether perioperative lipid-lowering increases the risk of rhabdomyolysis or liver disease.
Carotid endarterectomy
Two recent retrospective cohort studies evaluated the outcomes in patients undergoing carotid endarterectomy.19,20
Kennedy et al19 found that patients on a statin at the time of admission who had symptomatic carotid disease had lower rates of inhospital death (adjusted odds ratio 0.24, 95% CI 0.06–0.91) and ischemic stroke or death (adjusted odds ratio 0.55, 95% CI 0.31–0.97). However, cardiac outcomes among these symptomatic patients were not significantly improved (odds ratio 0.82, 95% CI 0.45–1.50), nor was there benefit for asymptomatic patients, raising the possibility that the positive findings were due to chance or that patients at lower baseline risk for vascular events may have less benefit.
McGirt et al20 performed a similar study; they did not, however, distinguish whether patients had symptomatic vs asymptomatic carotid disease. The 30-day risk of perioperative stroke was lower in patients treated with a statin, with an odds ratio of 0.41 (95% CI 0.18–0.93); the odds ratio for death was 0.21 (95% CI 0.05–0.96). Cardiac outcomes were not significantly affected.
Coronary artery bypass graft surgery
According to the NCEP recommendations, nearly all patients undergoing CABG should already be on a statin before surgery since they all have known coronary artery disease. Multiple observational studies have offered confirmatory evidence that statins are beneficial in this setting.34–38
Liakopoulos et al39 evaluated whether the anti-inflammatory effects of statins may, in part, account for their beneficial effect in the perioperative period. The authors prospectively matched 18 patients who were taking statins and were referred for elective CABG with 18 patients who were not prescribed statins previously. The only major measured baseline characteristic that differed between the two groups was a statistically significantly lower LDL-C level in the statin group. The operative characteristics did not differ, and cytokine levels at baseline were similar.
Tumor necrosis factor alpha levels increased significantly in the control group but did not change significantly in the statin group. Interleukin 8 increased in both groups by a similar amount. Interleukin 6 (the major inducer of C-reactive protein) increased from baseline in both groups but did not increase nearly as much in the statin group as in the control group; the intergroup difference was statistically significant. The anti-inflammatory cytokine interleukin 10 increased minimally from baseline in the control group, while the statin group’s levels increased significantly above baseline and those of the control group.
Christenson40 also found that inflammatory markers were improved with pre-CABG statin treatment in a small randomized trial in which patients received simvastatin 20 mg 4 weeks prior to CABG surgery vs no statin. Interestingly, far fewer statin-treated patients developed thrombocytosis (platelet count > 400 × 109/L) than did control patients (3% vs 81%, P < .0001).
RISKS OF PERIOPERATIVE STATINS
The risks associated with statin therapy in general appear low, but specific perioperative risks have not been well studied.
Baigent et al,41 in a meta-analysis of randomized trials of nonperioperative statin therapy, found that rhabdomyolysis occurred in 9 (0.023%) of 39,884 patients receiving statins vs 6 (0.015%) of the 39,817 controls, with a number needed to harm of 12,500. Moreover, the rates of nonvascular death and cancer did not increase. It is plausible that the risk is somewhat greater in the perioperative setting but is likely not enough to outweigh the potential benefits, especially since the risk of ischemic vascular events is particularly high then.
Some of the perioperative studies cited above specifically addressed potential risks. For example, in the study by Schouten et al,32 mild creatine kinase elevations were more common in the statin-treated group, but the incidence of moderate and severe creatine kinase elevations did not differ significantly. No case of rhabdomyolysis occurred, and length of surgery was the only predictor of myopathy. MIRACL and PROVE-IT revealed similar safety profiles; aminotransferase levels normalized when statins were stopped, and no cases of rhabdomyolysis occurred.11,12 In the vascular surgery study by Durazzo et al,23 1 (2%) of the 50 atorvastatin-treated patients developed both rhabdomyolysis and elevated aminotransferase levels that prompted discontinuation of the statin.
Overall, the observational studies do not indicate that statin continuation or treatment is harmful in perioperative patients. However, these studies did not specifically evaluate patients with acute insults from surgery such as sepsis, renal failure, or hepatitis. It is unknown what effect statin therapy would have in those patients and whether statins should be selectively discontinued in patients who develop major hepatic, musculoskeletal, or renal complications after surgery.
OUR RECOMMENDATIONS
Before CABG or vascular surgery
Given the NCEP recommendations, existing primary and secondary prevention studies, observational studies of CABG and noncardiac vascular surgery patients, and the one randomized trial of vascular surgery patients, data support the use of statins in nearly all patients undergoing cardiac or vascular surgery. We advocate starting statins in the perioperative period to take advantage of their rapid-acting pleiotropic effects, and continuing them long-term to take advantage of their lipid-lowering effects. This recommendation is in line with the recently released American College of Cardiology/American Heart Association (ACC/AHA) 2007 perioperative guidelines that state “for patients undergoing vascular surgery with or without clinical risk factors, statin use is reasonable.”42
Although the ideal time to start statins is not certain, the study by Durazzo et al23 suggests that they should be started at least 2 weeks before surgery if possible. Moreover, patients already taking statins should definitely not have their statins discontinued if at all possible.
Before major nonvascular surgery
For patients undergoing major nonvascular (intermediate-risk) surgery, physicians should first ascertain if the patient has an indication for statin therapy based on current nonsurgical lipid level recommendations. However, even if there is no clear indication for statin therapy based on NCEP guidelines, we endorse the recently released ACC/AHA perioperative guidelines that state that statin therapy can be considered in patients with a risk factor who are undergoing intermediate-risk procedures. Moreover, we wholeheartedly support the ACC/AHA’s strongest recommendation that patients who are already receiving statins and are undergoing noncardiac surgery should not have their statins discontinued.
When to discontinue statins?
The risk of harm overall appears to be minimal and certainly less than the likelihood of benefit. It is reasonable to observe patients postoperatively for adverse clinical events that may increase the risk of perioperative statin treatment, such as acute renal failure, hepatic failure, or sepsis, but whether statins should be stopped in patients with these complications remains unknown; we advocate individualizing the decision.
More studies needed
We need more data on whether moderate-risk patients undergoing moderate-risk surgery benefit from perioperative statin therapy, when therapy should be started, whether therapy should be started on the day of surgery if it was not started earlier, which statin and what doses are optimal, how long therapy should be continued, and what degree of risk is associated with perioperative statin therapy.
Fortunately, important data should be forthcoming in the next few years: the Dutch Echocardiographic Cardiac Risk Evaluation Applying Stress Echocardiography (DECREASE-IV) study43 is a 4-year two-by-two factorial placebo-controlled study evaluating the use of fluvastatin (Lescol) and bisoprolol (Zebeta, a beta-blocker) separately and together in patients who are older than 40 years, are undergoing elective noncardiac surgery, have an estimated risk of cardiovascular death of more than 1%, have not used statins previously, and do not have elevated cholesterol.
- Grant PJ, Kedia N. Should statins be discontinued preoperatively? IMPACT consults. Proceedings of the 2nd Annual Cleveland Clinic Perioperative Medicine Summit. Cleve Clin J Med 2006; 73 Electronic suppl 1:S9–S10.
- Lindenauer PK, Pekow P, Wang K, Gutierrez B, Benjamin EM. Lipid-lowering therapy and in-hospital mortality following major noncardiac surgery. JAMA 2004; 291:2092–2099.
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004; 351:2795–2804.
- Mangano DT, Layug EL, Wallace A, Tateo I. Effect of atenolol on mortality and cardiovascular morbidity after noncardiac surgery. Multicenter Study of Perioperative Ischemia Research Group. N Engl J Med 1996; 335:1713–1720.
- Poldermans D, Boersma E, Bax JJ, et al. The effect of bisoprolol on perioperative mortality and myocardial infarction in high-risk patients undergoing vascular surgery. Dutch Echocardiographic Cardiac Risk Evaluation Applying Stress Echocardiography Study Group. N Engl J Med 1999; 341:1789–1794.
- Brady AR, Gibbs JS, Greenhalgh RM, Powell JT, Sydes MR. Perioperative beta-blockade (POBBLE) for patients undergoing infrarenal vascular surgery: results of a randomized double-blind controlled trial. J Vasc Surg 2005; 41:602–609.
- Juul AB, Wetterslev J, Gluud C, et al. Effect of perioperative beta blockade in patients with diabetes undergoing major non-cardiac surgery: randomised placebo controlled, blinded multicentre trial. BMJ 2006; 332:1482.
- Yang H, Raymer K, Butler R, Parlow J, Roberts R. The effects of perioperative beta-blockade: results of the Metoprolol after Vascular Surgery (MaVS) study, a randomized controlled trial. Am Heart J 2006; 152:983–990.
- Ridker PM, Cannon CP, Morrow D, et al. C-reactive protein levels and outcomes after statin therapy. N Engl J Med 2005; 352:20–28.
- Ito MK, Talbert RL, Tsimikas S. Statin-associated pleiotropy: possible beneficial effects beyond cholesterol reduction. Pharmacotherapy 2006; 26:85S–97S.
- Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495–1504.
- Schwartz GG, Olsson AG, Ezekowitz MD, et al. Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA 2001; 285:1711–1718.
- Lefer AM, Campbell B, Shin YK, Scalia R, Hayward R, Lefer DJ. Simvastatin preserves the ischemic-reperfused myocardium in normocholesterolemic rat hearts. Circulation 1999; 100:178–184.
- Endres M, Laufs U, Liao JK, Moskowitz MA. Targeting eNOS for stroke protection. Trends Neurosci 2004; 27:283–289.
- Osborne JA, Lento PH, Siegfried MR, Stahl GL, Fusman B, Lefer AM. Cardiovascular effects of acute hypercholesterolemia in rabbits. Reversal with lovastatin treatment. J Clin Invest 1989; 83:465–473.
- Sironi L, Cimino M, Guerrini U, et al. Treatment with statins after induction of focal ischemia in rats reduces the extent of brain damage. Arterioscler Thromb Vasc Biol 2003; 23:322–327.
- Fonarow GC, Wright RS, Spencer FA, et al. Effect of statin use within the first 24 hours of admission for acute myocardial infarction on early morbidity and mortality. Am J Cardiol 2005; 96:611–616.
- Heeschen C, Hamm CW, Laufs U, Snapinn S, Bohm M, White HD. Withdrawal of statins increases event rates in patients with acute coronary syndromes. Circulation 2002; 105:1446–1452.
- Kennedy J, Quan H, Buchan AM, Ghali WA, Feasby TE. Statins are associated with better outcomes after carotid endarterectomy in symptomatic patients. Stroke 2005; 36:2072–2076.
- McGirt MJ, Perler BA, Brooke BS, et al. 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors reduce the risk of perioperative stroke and mortality after carotid endarterectomy. J Vasc Surg 2005; 42:829–836.
- Hindler K, Shaw AD, Samuels J, Fulton S, Collard CD, Riedel B. Improved postoperative outcomes associated with preoperative statin therapy. Anesthesiology 2006; 105:1260–1272.
- Kapoor AS, Kanji H, Buckingham J, Devereaux PJ, McAlister FA. Strength of evidence for perioperative use of statins to reduce cardiovascular risk: systematic review of controlled studies. BMJ 2006; 333:1149.
- Durazzo AE, Machado FS, Ikeoka DT, et al. Reduction in cardiovascular events after vascular surgery with atorvastatin: a randomized trial. J Vasc Surg 2004; 39:967–975.
- Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 2004; 110:227–239.
- Kertai MD, Boersma E, Westerhout CM, et al. A combination of statins and beta-blockers is independently associated with a reduction in the incidence of perioperative mortality and nonfatal myocardial infarction in patients undergoing abdominal aortic aneurysm surgery. Eur J Vasc Endovasc Surg 2004; 28:343–352.
- Poldermans D, Bax JJ, Kertai MD, et al. Statins are associated with a reduced incidence of perioperative mortality in patients undergoing major noncardiac vascular surgery. Circulation 2003; 107:1848–1851.
- Ward RP, Leeper NJ, Kirkpatrick JN, Lang RM, Sorrentino MJ, Williams KA. The effect of preoperative statin therapy on cardiovascular outcomes in patients undergoing infrainguinal vascular surgery. Int J Cardiol 2005; 104:264–268.
- O’Neil-Callahan K, Katsimaglis G, Tepper MR, et al. Statins decrease perioperative cardiac complications in patients undergoing non-cardiac vascular surgery: the Statins for Risk Reduction in Surgery (StaRRS) study. J Am Coll Cardiol 2005; 45:336–342.
- Abbruzzese TA, Havens J, Belkin M, et al. Statin therapy is associated with improved patency of autogenous infrainguinal bypass grafts. J Vasc Surg 2004; 39:1178–1185.
- Boersma E, Poldermans D, Bax JJ, et al. Predictors of cardiac events after major vascular surgery: role of clinical characteristics, dobutamine echocardiography, and beta-blocker therapy. JAMA 2001; 285:1865–1873.
- Landesberg G, Mosseri M, Wolf YG, et al. Preoperative thallium scanning, selective coronary revascularization, and long-term survival after major vascular surgery. Circulation 2003; 108:177–183.
- Schouten O, Kertai MD, Bax JJ, et al. Safety of perioperative statin use in high-risk patients undergoing major vascular surgery. Am J Cardiol 2005; 95:658–660.
- Le Manach Y, Godet G, Coriat P, et al. The impact of postoperative discontinuation or continuation of chronic statin therapy on cardiac outcome after major vascular surgery. Anesth Analg 2007; 104:1326–1333.
- Ali IS, Buth KJ. Preoperative statin use and outcomes following cardiac surgery. Int J Cardiol 2005; 103:12–18.
- Clark LL, Ikonomidis JS, Crawford FA, et al. Preoperative statin treatment is associated with reduced postoperative mortality and morbidity in patients undergoing cardiac surgery: an 8-year retrospective cohort study. J Thorac Cardiovasc Surg 2006; 131:679–685.
- Pan W, Pintar T, Anton J, Lee VV, Vaughn WK, Collard CD. Statins are associated with a reduced incidence of perioperative mortality after coronary artery bypass graft surgery. Circulation 2004; 110(suppl 2):II45–II49.
- Pascual DA, Arribas JM, Tornel PL, et al. Preoperative statin therapy and troponin T predict early complications of coronary artery surgery. Ann Thorac Surg 2006; 81:78–83.
- Dotani MI, Elnicki DM, Jain AC, Gibson CM. Effect of preoperative statin therapy and cardiac outcomes after coronary artery bypass grafting. Am J Cardiol 2000; 86:1128–1130.
- Liakopoulos OJ, Dorge H, Schmitto JD, Nagorsnik U, Grabedunkel J, Schoendube FA. Effects of preoperative statin therapy on cytokines after cardiac surgery. Thorac Cardiovasc Surg 2006; 54:250–254.
- Christenson JT. Preoperative lipid-control with simvastatin reduces the risk of postoperative thrombocytosis and thrombotic complications following CABG. Eur J Cardiothorac Surg 1999; 15:394–399.
- Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 2005; 366:1267–1278.
- Fleisher LA, Beckman JA, Brown KA, et al. ACC/AHA 2007 Guidelines on Perioperative Cardiovascular Evaluation and Care for Noncardiac Surgery. A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation 2007; 116:e418–e499.
- Schouten O, Poldermans D, Visser L, et al. Fluvastatin and bisoprolol for the reduction of perioperative cardiac mortality and morbidity in high-risk patients undergoing non-cardiac surgery: rationale and design of the DECREASE-IV study. Am Heart J 2004; 148:1047–1052.
- Amar D, Zhang H, Heerdt PM, Park B, Fleisher M, Thaler HT. Statin use is associated with a reduction in atrial fibrillation after noncardiac thoracic surgery independent of C-reactive protein. Chest 2005; 128:3421–3427.
Soon, the checklist for internists seeing patients about to undergo surgery may include prescribing one of the lipid-lowering hydroxymethylglutaryl-CoA reductase inhibitors, also called statins.
Statins? Not long ago, we were debating whether patients who take statins should stop taking them before surgery, based on the manufacturers’ recommendations.1 The discussion, however, has changed to whether patients who have never received a statin should be started on one before surgery to provide immediate prophylaxis against cardiac morbidity, and how much harm long-term statin users face if these drugs are withheld perioperatively.
The evidence is still very preliminary and based mostly on studies in animals and retrospective studies in people. However, an expanding body of indirect evidence suggests that these drugs are beneficial in this situation.
In this review, we discuss the mechanisms by which statins may protect the heart in the short term, drawing on data from animal and human studies of acute myocardial infarction, and we review the current (albeit limited) data from the perioperative setting.
FEW INTERVENTIONS DECREASE RISK
Each year, approximately 50,000 patients suffer a perioperative cardiovascular event; the incidence of myocardial infarction during or after noncardiac surgery is 2% to 3%.2 The primary goal of preoperative cardiovascular risk assessment is to predict and avert these events.
But short of canceling surgery, few interventions have been found to reduce a patient’s risk. For example, a landmark study in 2004 cast doubt on the efficacy of preoperative coronary revascularization.3 Similarly, although early studies of beta-blockers were promising4,5 and although most internists prescribe these drugs before surgery, more recent studies have cast doubt on their efficacy, particularly in patients at low risk undergoing intermediate-risk (rather than vascular) surgery.6–8
This changing clinical landscape has prompted a search for new strategies for perioperative risk-reduction. Several recent studies have placed statins in the spotlight.
POTENTIAL MECHANISMS OF SHORT-TERM BENEFIT
Statins have been proven to save lives when used long-term, but how could this class of drugs, designed to prevent the accumulation of arterial plaques by lowering low-density lipoprotein cholesterol (LDL-C) levels, have any short-term impact on operative outcomes? Although LDL-C reduction is the principal mechanism of action of statins, not all of the benefit can be ascribed to this mechanism.9 The answer may lie in their “pleiotropic” effects—ie, actions other than LDL-C reduction.
The more immediate pleiotropic effects of statins in the proinflammatory and prothrombotic environment of the perioperative period are thought to include improved endothelial function (both antithrombotic function and vasomotor function in response to ischemic stress), enhanced stability of atherosclerotic plaques, decreased oxidative stress, and decreased vascular inflammation.10–12
EVIDENCE FROM ANIMAL STUDIES
Experiments in animals suggest that statins, given shortly before or after a cardiovascular event, confer benefit before any changes in LDL-C are measurable.
Lefer et al13 found that simvastatin (Zocor), given 18 hours before an ischemic episode in rats, blunted the inflammatory response in cardiac reperfusion injury. Not only was reperfusion injury significantly less in the hearts of the rats that received simvastatin than in the saline control group, but the simvastatin-treated hearts also expressed fewer neutrophil adhesion molecules such as P-selectin, and they had more basal release of nitric oxide, the potent endothelial-derived vasodilator with antithrombotic, anti-inflammatory, and antiproliferative effects.14 These results suggest that statins may improve endothelial function acutely, particularly during ischemic stress.
Osborne et al15 fed rabbits a cholesterol-rich diet plus either lovastatin (Mevacor) or placebo. After 2 weeks, the rabbits underwent either surgery to induce a myocardial infarction or a sham procedure. Regardless of the pretreatment, biopsies of the aorta did not reveal any atherosclerosis; yet the lovastatin-treated rabbits sustained less myocardial ischemic damage and they had more endothelium-mediated vasodilatation.
Statin therapy also may improve cerebral ischemia outcomes in animal models.14,16
Sironi et al16 induced strokes in rats by occluding the middle cerebral artery. The rats received either simvastatin or vehicle for 3 days before the stroke or immediately afterwards. Even though simvastatin did not have enough time to affect the total cholesterol level, rats treated with simvastatin had smaller infarcts (as measured by magnetic resonance imaging) and produced more nitric oxide.
Comment. Taken together, these studies offer tantalizing evidence that statins have short-term, beneficial nonlipid effects and may reduce not only the likelihood of an ischemic event, but—should one occur—the degree of tissue damage that ensues.
EFFECTS OF STATINS IN ACUTE CORONARY SYNDROME
The National Registry of Myocardial Infarction17 is a prospective, observational database of all patients with acute myocardial infarction admitted to 1,230 participating hospitals throughout the United States. In an analysis from this cohort, patients were divided into four groups: those receiving statins before and after admission, those receiving statins only before admission, those receiving statins only after admission, and those who never received statins.
Compared with those who never received statins, fewer patients who received them both before and after admission died while in the hospital (unadjusted odds ratio 0.23, 95% confidence interval [CI] 0.22–0.25), and the odds ratio for those who received statins for the first time was 0.31 (95% CI 0.29–0.33). Patients who stopped receiving a statin on admission were more likely to die than were patients who never received statins (odds ratio 1.09, 95% CI 1.03–1.15). These trends held true even when adjustments were made for potential confounding factors.
Comment. Unmeasured confounding factors (such as the inability to take pills due to altered mental status or the different practice styles of the providers who chose to discontinue statins) might have affected the results. Nevertheless, these results suggest that the protective effects of statins stop almost immediately when these drugs are discontinued, and that there may even be an adverse “rebound” effect when patients who have been taking these drugs for a long time stop taking them temporarily.
The Platelet Receptor Inhibition in Ischemic Syndrome Management trial,18 in a subgroup analysis, had nearly identical findings. In the main part of this trial, patients with coronary artery disease and chest pain at rest or accelerating pain in the last 24 hours were randomized to receive tirofiban (Aggrastat) or heparin. Complete data on statin use were available for 1,616 (50%) of the 3,232 patients in this trial, and the rate of the primary end point (death, myocardial infarction, or recurrent ischemia) was analyzed on the basis of statin therapy in this subgroup.
Comment. Together, these data lead to the conclusion that, when admitted for either acute myocardial infarction or acute coronary syndrome, patients already receiving statins should not have them stopped, and those who had not been receiving statins should receive them immediately. The safety of these medications in the acute setting appears excellent: in the Myocardial Ischemia Reduction With Acute Cholesterol Lowering (MIRACL)12 and the Pravastatin or Atorvastatin Evaluation and Infection Therapy (PROVE-IT)11 trials, fewer than 5% of statin-treated patients had transient elevations in transaminase levels, and no cases of rhabdomyolysis were reported.
PERIOPERATIVE STATIN STUDIES
The data on perioperative statin use are mostly observational and retrospective and fall into essentially four surgical categories: coronary artery bypass grafting (CABG), carotid endarterectomy,19,20 noncardiac vascular surgery, and major noncardiac surgery. Two meta-analyses have also evaluated the data.21,22 The only randomized controlled trial (performed by Durazzo et al23) was small and was carried out at a single center in vascular surgery patients, and the event rate was low.
Current recommendations from the National Cholesterol Education Program (NCEP)24 say that patients who need CABG, have peripheral arterial disease, have an abdominal aortic aneurysm, or have cerebrovascular disease should already be on a statin to achieve an LDL-C goal level of less than 100 mg/dL, with an optional goal of less than 70 mg/dL, independent of surgery.
Since not all patients who should be on statins are actually on them, questions arise:
- Is it important (and safe) to start statin treatment preoperatively?
- Will patients with cardiovascular risk factors but without known cardiovascular disease benefit from statins perioperatively?
Noncardiac vascular surgery
Multiple retrospective studies have evaluated the effect of statins in patients undergoing major noncardiac vascular surgery.25–32
Kertai et al25 evaluated 570 patients in Holland who underwent elective open surgery for infrarenal abdominal aortic aneurysms between 1991 and 2001, looking for an association between statin use and the incidence of perioperative death from myocardial infarction. Only 162 of the 570 patients had been on long-term statin therapy before the surgery. The use of statins was only one of many known baseline characteristics that were significantly different between the two groups, including age, body mass index, known coronary artery disease, and use of angiotensin-converting enzyme inhibitors and beta-blockers. In univariate analysis, statins appeared to be protective: 6 (3.7%) of the patients in the statin group died of a myocardial infarction, compared with 45 (11%) of those in the nostatin group. A multivariate analysis yielded similar findings, with an odds ratio of 0.24 (95% CI 0.11–0.54).
Ward et al27 performed a very similar retrospective study, with similar findings. In 446 patients who underwent surgery for infrarenal abdominal aortic aneurysm, statin therapy was associated with a significantly lower incidence of the combined end point of death, myocardial infarction, stroke, and major peripheral vascular complications, with an adjusted odds ratio of 0.36 (95% CI 0.14–0.93).
Poldermans et al26 noted similar findings in a case-control study of noncardiac vascular surgery patients. Statin users had a much lower perioperative risk of death than did nonusers, with an adjusted odds ratio of 0.22 (95% CI 0.10–0.47).
O’Neil-Callahan et al,28 in a cohort study, found that statin users had fewer perioperative cardiac complications, with an adjusted odds ratio of 0.49 (95% CI 0.28–0.84, P = .009).
Dogma of withdrawing statins before major surgery is challenged
Le Manach et al33 reviewed the outcomes for all patients of a single hospital in Paris who underwent nonemergency infrarenal aortic procedures between January 2001 and December 2004. In January 2004, the hospital instituted guidelines to ensure that patients on statins continue taking them up to the evening before surgery and that statins be restarted on the first postoperative day (via nasogastric tube if necessary). Before 2004, there had been no specific guidelines, and patients on statins did not receive them for a median of 4 days postoperatively. Types of procedures were similar during the two time periods, as were the rates of beta-blocker use, preoperative revascularization, venous thromboembolism prophylaxis, and perioperative blood pressure control. After surgery, topononin I levels were measured in all patients as surveillance for cardiac events, and were defined as elevated when greater than 0.2 ng/mL.
Compared with patients not on statins at all, those treated with statins continuously throughout the perioperative period (after January 2004) had a lower rate of elevated troponin (relative risk 0.38). In contrast, those who had their statins transiently discontinued perioperatively (prior to 2004) had troponin elevations more often than those who had never been treated (relative risk 2.1). This suggested an over fivefold risk reduction (P < .001) conferred by not discontinuing statins in the immediate postoperative period. This finding was maintained after multivariate adjustment: statin withdrawal was associated with a 2.9-fold (95% CI 1.6–5.5) increase in the risk of cardiac enzyme elevations postoperatively. No fewer deaths were noted, but the study was not powered to detect a mortality difference.
Comment. Although secular trends cannot be entirely discounted as contributing to these findings, the prompt increase in cardiac events after just 4 days of statin withdrawal adds to the growing body of evidence suggesting that statin discontinuation can have harmful acute effects. It also brings up the question: Can starting statins benefit patients in the same time period?
Should statins be started before vascular surgery?
Schouten et al32 evaluated the effects of newly started or continued statin treatment in patients undergoing major elective vascular surgery. Patients were screened before surgery and started on statins if they were not already receiving them and their total cholesterol levels were elevated; new users received the medication for about 40 days before surgery. Of the 981 screened patients, 44 (5%) were newly started on statins and 182 (19%) were continued on their therapy. Perioperative death or myocardial infarction occurred in 22 (8.8%) of the statin users and 111 (14.7%) of the nonusers, a statistically significant difference. Temporary discontinuation (median 1 day) of statins in this study due to the inability to take an oral medication did not appear to affect the likelihood of a myocardial infarction.
Durazzo et al23 performed a single-center, randomized, prospective, placebo-controlled, double-blind clinical trial of atorvastatin (Lipitor) 20 mg daily vs placebo in 100 patients undergoing noncardiac arterial vascular surgery. Patients were excluded if they had previously used medications to treat dyslipidemia, recently had a cardiovascular event, or had contraindications to statin treatment such as a baseline creatinine level greater than 2.0 mg/dL or severe hepatic disease. The intervention group received atorvastatin starting at least 2 weeks before surgery for a total of 45 days. Patients were then continued or started on a statin after surgery if their LDL-C level was greater than 100 mg/dL. Beta-blocker use was recommended “on the basis of current guidelines.”
One month after surgery, the LDL-C level was statistically significantly lower in the atorvastatin group. Since most patients did not continue or start statin therapy after the 45-day treatment period, the LDL-C levels were not statistically different at 3 and 6 months after surgery.
At 6 months, the rate of the primary end point (death from cardiovascular causes, nonfatal acute myocardial infarction, ischemic stroke, or unstable angina) was 26.0% in the placebo group and 8.0% in the atorvastatin group, a statistically significant difference. Three patients in the atorvastatin group had cardiac events in the first 10 days after surgery, compared with 11 patients in the placebo group. Thirteen of the 17 total cardiac events took place within 10 days after surgery.
One of the atorvastatin patients developed rhabdomyolysis and elevated aminotransferase levels.
Major noncardiac surgery
Lindenauer et al2 performed a retrospective cohort study of surgical patients who were at least 18 years old and survived beyond the second hospital day. Patients were divided into a group receiving any form of lipid-lowering treatment (of whom more than 90% were taking statins) and a group that had never never received a lipid-lowering drug or only started one on the third day of the hospitalization or later. The period of study was from January 1, 2000, to December 31, 2001.
In all, 780,591 patients from 329 hospitals throughout the United States were included, of whom only 77,082 (9.9%) received lipid-lowering therapy. Eight percent of the patients underwent vascular surgery. Not surprisingly, the treated patients were more likely to have a history of hypertension, diabetes, ischemic heart disease, or hyperlipidemia. They also were more likely to have a vascular procedure performed, to have two or more cardiac risk factors (high-risk surgery, ischemic heart disease, congestive heart failure, cerebrovascular disease, renal insufficiency, or diabetes mellitus), and to be treated with beta-blockers and angiotensin-converting enzyme inhibitors, but they were less likely to have high-risk and emergency surgery performed.
The primary end point, perioperative death, occurred in 2.13% of the treated patients and 3.05% of the nontreated group. Compared with the rate in a propensity-matched cohort, the odds ratio adjusted for unbalanced covariates was 0.62 (95% CI 0.58–0.67) in favor of lipid treatment. Stratification by cardiac risk index revealed a number needed to treat of 186 for those with no risk factors, 60 for those with two risk factors, and 30 for those with four or more risk factors.
Unfortunately, this analysis was not able to take into account whether and for how long patients were receiving lipid-lowering therapy before hospitalization. It therefore does not answer the questions of whether starting lipid-lowering therapy before surgery is beneficial or whether stopping it is harmful. It also does not shed light on whether perioperative lipid-lowering increases the risk of rhabdomyolysis or liver disease.
Carotid endarterectomy
Two recent retrospective cohort studies evaluated the outcomes in patients undergoing carotid endarterectomy.19,20
Kennedy et al19 found that patients on a statin at the time of admission who had symptomatic carotid disease had lower rates of inhospital death (adjusted odds ratio 0.24, 95% CI 0.06–0.91) and ischemic stroke or death (adjusted odds ratio 0.55, 95% CI 0.31–0.97). However, cardiac outcomes among these symptomatic patients were not significantly improved (odds ratio 0.82, 95% CI 0.45–1.50), nor was there benefit for asymptomatic patients, raising the possibility that the positive findings were due to chance or that patients at lower baseline risk for vascular events may have less benefit.
McGirt et al20 performed a similar study; they did not, however, distinguish whether patients had symptomatic vs asymptomatic carotid disease. The 30-day risk of perioperative stroke was lower in patients treated with a statin, with an odds ratio of 0.41 (95% CI 0.18–0.93); the odds ratio for death was 0.21 (95% CI 0.05–0.96). Cardiac outcomes were not significantly affected.
Coronary artery bypass graft surgery
According to the NCEP recommendations, nearly all patients undergoing CABG should already be on a statin before surgery since they all have known coronary artery disease. Multiple observational studies have offered confirmatory evidence that statins are beneficial in this setting.34–38
Liakopoulos et al39 evaluated whether the anti-inflammatory effects of statins may, in part, account for their beneficial effect in the perioperative period. The authors prospectively matched 18 patients who were taking statins and were referred for elective CABG with 18 patients who were not prescribed statins previously. The only major measured baseline characteristic that differed between the two groups was a statistically significantly lower LDL-C level in the statin group. The operative characteristics did not differ, and cytokine levels at baseline were similar.
Tumor necrosis factor alpha levels increased significantly in the control group but did not change significantly in the statin group. Interleukin 8 increased in both groups by a similar amount. Interleukin 6 (the major inducer of C-reactive protein) increased from baseline in both groups but did not increase nearly as much in the statin group as in the control group; the intergroup difference was statistically significant. The anti-inflammatory cytokine interleukin 10 increased minimally from baseline in the control group, while the statin group’s levels increased significantly above baseline and those of the control group.
Christenson40 also found that inflammatory markers were improved with pre-CABG statin treatment in a small randomized trial in which patients received simvastatin 20 mg 4 weeks prior to CABG surgery vs no statin. Interestingly, far fewer statin-treated patients developed thrombocytosis (platelet count > 400 × 109/L) than did control patients (3% vs 81%, P < .0001).
RISKS OF PERIOPERATIVE STATINS
The risks associated with statin therapy in general appear low, but specific perioperative risks have not been well studied.
Baigent et al,41 in a meta-analysis of randomized trials of nonperioperative statin therapy, found that rhabdomyolysis occurred in 9 (0.023%) of 39,884 patients receiving statins vs 6 (0.015%) of the 39,817 controls, with a number needed to harm of 12,500. Moreover, the rates of nonvascular death and cancer did not increase. It is plausible that the risk is somewhat greater in the perioperative setting but is likely not enough to outweigh the potential benefits, especially since the risk of ischemic vascular events is particularly high then.
Some of the perioperative studies cited above specifically addressed potential risks. For example, in the study by Schouten et al,32 mild creatine kinase elevations were more common in the statin-treated group, but the incidence of moderate and severe creatine kinase elevations did not differ significantly. No case of rhabdomyolysis occurred, and length of surgery was the only predictor of myopathy. MIRACL and PROVE-IT revealed similar safety profiles; aminotransferase levels normalized when statins were stopped, and no cases of rhabdomyolysis occurred.11,12 In the vascular surgery study by Durazzo et al,23 1 (2%) of the 50 atorvastatin-treated patients developed both rhabdomyolysis and elevated aminotransferase levels that prompted discontinuation of the statin.
Overall, the observational studies do not indicate that statin continuation or treatment is harmful in perioperative patients. However, these studies did not specifically evaluate patients with acute insults from surgery such as sepsis, renal failure, or hepatitis. It is unknown what effect statin therapy would have in those patients and whether statins should be selectively discontinued in patients who develop major hepatic, musculoskeletal, or renal complications after surgery.
OUR RECOMMENDATIONS
Before CABG or vascular surgery
Given the NCEP recommendations, existing primary and secondary prevention studies, observational studies of CABG and noncardiac vascular surgery patients, and the one randomized trial of vascular surgery patients, data support the use of statins in nearly all patients undergoing cardiac or vascular surgery. We advocate starting statins in the perioperative period to take advantage of their rapid-acting pleiotropic effects, and continuing them long-term to take advantage of their lipid-lowering effects. This recommendation is in line with the recently released American College of Cardiology/American Heart Association (ACC/AHA) 2007 perioperative guidelines that state “for patients undergoing vascular surgery with or without clinical risk factors, statin use is reasonable.”42
Although the ideal time to start statins is not certain, the study by Durazzo et al23 suggests that they should be started at least 2 weeks before surgery if possible. Moreover, patients already taking statins should definitely not have their statins discontinued if at all possible.
Before major nonvascular surgery
For patients undergoing major nonvascular (intermediate-risk) surgery, physicians should first ascertain if the patient has an indication for statin therapy based on current nonsurgical lipid level recommendations. However, even if there is no clear indication for statin therapy based on NCEP guidelines, we endorse the recently released ACC/AHA perioperative guidelines that state that statin therapy can be considered in patients with a risk factor who are undergoing intermediate-risk procedures. Moreover, we wholeheartedly support the ACC/AHA’s strongest recommendation that patients who are already receiving statins and are undergoing noncardiac surgery should not have their statins discontinued.
When to discontinue statins?
The risk of harm overall appears to be minimal and certainly less than the likelihood of benefit. It is reasonable to observe patients postoperatively for adverse clinical events that may increase the risk of perioperative statin treatment, such as acute renal failure, hepatic failure, or sepsis, but whether statins should be stopped in patients with these complications remains unknown; we advocate individualizing the decision.
More studies needed
We need more data on whether moderate-risk patients undergoing moderate-risk surgery benefit from perioperative statin therapy, when therapy should be started, whether therapy should be started on the day of surgery if it was not started earlier, which statin and what doses are optimal, how long therapy should be continued, and what degree of risk is associated with perioperative statin therapy.
Fortunately, important data should be forthcoming in the next few years: the Dutch Echocardiographic Cardiac Risk Evaluation Applying Stress Echocardiography (DECREASE-IV) study43 is a 4-year two-by-two factorial placebo-controlled study evaluating the use of fluvastatin (Lescol) and bisoprolol (Zebeta, a beta-blocker) separately and together in patients who are older than 40 years, are undergoing elective noncardiac surgery, have an estimated risk of cardiovascular death of more than 1%, have not used statins previously, and do not have elevated cholesterol.
Soon, the checklist for internists seeing patients about to undergo surgery may include prescribing one of the lipid-lowering hydroxymethylglutaryl-CoA reductase inhibitors, also called statins.
Statins? Not long ago, we were debating whether patients who take statins should stop taking them before surgery, based on the manufacturers’ recommendations.1 The discussion, however, has changed to whether patients who have never received a statin should be started on one before surgery to provide immediate prophylaxis against cardiac morbidity, and how much harm long-term statin users face if these drugs are withheld perioperatively.
The evidence is still very preliminary and based mostly on studies in animals and retrospective studies in people. However, an expanding body of indirect evidence suggests that these drugs are beneficial in this situation.
In this review, we discuss the mechanisms by which statins may protect the heart in the short term, drawing on data from animal and human studies of acute myocardial infarction, and we review the current (albeit limited) data from the perioperative setting.
FEW INTERVENTIONS DECREASE RISK
Each year, approximately 50,000 patients suffer a perioperative cardiovascular event; the incidence of myocardial infarction during or after noncardiac surgery is 2% to 3%.2 The primary goal of preoperative cardiovascular risk assessment is to predict and avert these events.
But short of canceling surgery, few interventions have been found to reduce a patient’s risk. For example, a landmark study in 2004 cast doubt on the efficacy of preoperative coronary revascularization.3 Similarly, although early studies of beta-blockers were promising4,5 and although most internists prescribe these drugs before surgery, more recent studies have cast doubt on their efficacy, particularly in patients at low risk undergoing intermediate-risk (rather than vascular) surgery.6–8
This changing clinical landscape has prompted a search for new strategies for perioperative risk-reduction. Several recent studies have placed statins in the spotlight.
POTENTIAL MECHANISMS OF SHORT-TERM BENEFIT
Statins have been proven to save lives when used long-term, but how could this class of drugs, designed to prevent the accumulation of arterial plaques by lowering low-density lipoprotein cholesterol (LDL-C) levels, have any short-term impact on operative outcomes? Although LDL-C reduction is the principal mechanism of action of statins, not all of the benefit can be ascribed to this mechanism.9 The answer may lie in their “pleiotropic” effects—ie, actions other than LDL-C reduction.
The more immediate pleiotropic effects of statins in the proinflammatory and prothrombotic environment of the perioperative period are thought to include improved endothelial function (both antithrombotic function and vasomotor function in response to ischemic stress), enhanced stability of atherosclerotic plaques, decreased oxidative stress, and decreased vascular inflammation.10–12
EVIDENCE FROM ANIMAL STUDIES
Experiments in animals suggest that statins, given shortly before or after a cardiovascular event, confer benefit before any changes in LDL-C are measurable.
Lefer et al13 found that simvastatin (Zocor), given 18 hours before an ischemic episode in rats, blunted the inflammatory response in cardiac reperfusion injury. Not only was reperfusion injury significantly less in the hearts of the rats that received simvastatin than in the saline control group, but the simvastatin-treated hearts also expressed fewer neutrophil adhesion molecules such as P-selectin, and they had more basal release of nitric oxide, the potent endothelial-derived vasodilator with antithrombotic, anti-inflammatory, and antiproliferative effects.14 These results suggest that statins may improve endothelial function acutely, particularly during ischemic stress.
Osborne et al15 fed rabbits a cholesterol-rich diet plus either lovastatin (Mevacor) or placebo. After 2 weeks, the rabbits underwent either surgery to induce a myocardial infarction or a sham procedure. Regardless of the pretreatment, biopsies of the aorta did not reveal any atherosclerosis; yet the lovastatin-treated rabbits sustained less myocardial ischemic damage and they had more endothelium-mediated vasodilatation.
Statin therapy also may improve cerebral ischemia outcomes in animal models.14,16
Sironi et al16 induced strokes in rats by occluding the middle cerebral artery. The rats received either simvastatin or vehicle for 3 days before the stroke or immediately afterwards. Even though simvastatin did not have enough time to affect the total cholesterol level, rats treated with simvastatin had smaller infarcts (as measured by magnetic resonance imaging) and produced more nitric oxide.
Comment. Taken together, these studies offer tantalizing evidence that statins have short-term, beneficial nonlipid effects and may reduce not only the likelihood of an ischemic event, but—should one occur—the degree of tissue damage that ensues.
EFFECTS OF STATINS IN ACUTE CORONARY SYNDROME
The National Registry of Myocardial Infarction17 is a prospective, observational database of all patients with acute myocardial infarction admitted to 1,230 participating hospitals throughout the United States. In an analysis from this cohort, patients were divided into four groups: those receiving statins before and after admission, those receiving statins only before admission, those receiving statins only after admission, and those who never received statins.
Compared with those who never received statins, fewer patients who received them both before and after admission died while in the hospital (unadjusted odds ratio 0.23, 95% confidence interval [CI] 0.22–0.25), and the odds ratio for those who received statins for the first time was 0.31 (95% CI 0.29–0.33). Patients who stopped receiving a statin on admission were more likely to die than were patients who never received statins (odds ratio 1.09, 95% CI 1.03–1.15). These trends held true even when adjustments were made for potential confounding factors.
Comment. Unmeasured confounding factors (such as the inability to take pills due to altered mental status or the different practice styles of the providers who chose to discontinue statins) might have affected the results. Nevertheless, these results suggest that the protective effects of statins stop almost immediately when these drugs are discontinued, and that there may even be an adverse “rebound” effect when patients who have been taking these drugs for a long time stop taking them temporarily.
The Platelet Receptor Inhibition in Ischemic Syndrome Management trial,18 in a subgroup analysis, had nearly identical findings. In the main part of this trial, patients with coronary artery disease and chest pain at rest or accelerating pain in the last 24 hours were randomized to receive tirofiban (Aggrastat) or heparin. Complete data on statin use were available for 1,616 (50%) of the 3,232 patients in this trial, and the rate of the primary end point (death, myocardial infarction, or recurrent ischemia) was analyzed on the basis of statin therapy in this subgroup.
Comment. Together, these data lead to the conclusion that, when admitted for either acute myocardial infarction or acute coronary syndrome, patients already receiving statins should not have them stopped, and those who had not been receiving statins should receive them immediately. The safety of these medications in the acute setting appears excellent: in the Myocardial Ischemia Reduction With Acute Cholesterol Lowering (MIRACL)12 and the Pravastatin or Atorvastatin Evaluation and Infection Therapy (PROVE-IT)11 trials, fewer than 5% of statin-treated patients had transient elevations in transaminase levels, and no cases of rhabdomyolysis were reported.
PERIOPERATIVE STATIN STUDIES
The data on perioperative statin use are mostly observational and retrospective and fall into essentially four surgical categories: coronary artery bypass grafting (CABG), carotid endarterectomy,19,20 noncardiac vascular surgery, and major noncardiac surgery. Two meta-analyses have also evaluated the data.21,22 The only randomized controlled trial (performed by Durazzo et al23) was small and was carried out at a single center in vascular surgery patients, and the event rate was low.
Current recommendations from the National Cholesterol Education Program (NCEP)24 say that patients who need CABG, have peripheral arterial disease, have an abdominal aortic aneurysm, or have cerebrovascular disease should already be on a statin to achieve an LDL-C goal level of less than 100 mg/dL, with an optional goal of less than 70 mg/dL, independent of surgery.
Since not all patients who should be on statins are actually on them, questions arise:
- Is it important (and safe) to start statin treatment preoperatively?
- Will patients with cardiovascular risk factors but without known cardiovascular disease benefit from statins perioperatively?
Noncardiac vascular surgery
Multiple retrospective studies have evaluated the effect of statins in patients undergoing major noncardiac vascular surgery.25–32
Kertai et al25 evaluated 570 patients in Holland who underwent elective open surgery for infrarenal abdominal aortic aneurysms between 1991 and 2001, looking for an association between statin use and the incidence of perioperative death from myocardial infarction. Only 162 of the 570 patients had been on long-term statin therapy before the surgery. The use of statins was only one of many known baseline characteristics that were significantly different between the two groups, including age, body mass index, known coronary artery disease, and use of angiotensin-converting enzyme inhibitors and beta-blockers. In univariate analysis, statins appeared to be protective: 6 (3.7%) of the patients in the statin group died of a myocardial infarction, compared with 45 (11%) of those in the nostatin group. A multivariate analysis yielded similar findings, with an odds ratio of 0.24 (95% CI 0.11–0.54).
Ward et al27 performed a very similar retrospective study, with similar findings. In 446 patients who underwent surgery for infrarenal abdominal aortic aneurysm, statin therapy was associated with a significantly lower incidence of the combined end point of death, myocardial infarction, stroke, and major peripheral vascular complications, with an adjusted odds ratio of 0.36 (95% CI 0.14–0.93).
Poldermans et al26 noted similar findings in a case-control study of noncardiac vascular surgery patients. Statin users had a much lower perioperative risk of death than did nonusers, with an adjusted odds ratio of 0.22 (95% CI 0.10–0.47).
O’Neil-Callahan et al,28 in a cohort study, found that statin users had fewer perioperative cardiac complications, with an adjusted odds ratio of 0.49 (95% CI 0.28–0.84, P = .009).
Dogma of withdrawing statins before major surgery is challenged
Le Manach et al33 reviewed the outcomes for all patients of a single hospital in Paris who underwent nonemergency infrarenal aortic procedures between January 2001 and December 2004. In January 2004, the hospital instituted guidelines to ensure that patients on statins continue taking them up to the evening before surgery and that statins be restarted on the first postoperative day (via nasogastric tube if necessary). Before 2004, there had been no specific guidelines, and patients on statins did not receive them for a median of 4 days postoperatively. Types of procedures were similar during the two time periods, as were the rates of beta-blocker use, preoperative revascularization, venous thromboembolism prophylaxis, and perioperative blood pressure control. After surgery, topononin I levels were measured in all patients as surveillance for cardiac events, and were defined as elevated when greater than 0.2 ng/mL.
Compared with patients not on statins at all, those treated with statins continuously throughout the perioperative period (after January 2004) had a lower rate of elevated troponin (relative risk 0.38). In contrast, those who had their statins transiently discontinued perioperatively (prior to 2004) had troponin elevations more often than those who had never been treated (relative risk 2.1). This suggested an over fivefold risk reduction (P < .001) conferred by not discontinuing statins in the immediate postoperative period. This finding was maintained after multivariate adjustment: statin withdrawal was associated with a 2.9-fold (95% CI 1.6–5.5) increase in the risk of cardiac enzyme elevations postoperatively. No fewer deaths were noted, but the study was not powered to detect a mortality difference.
Comment. Although secular trends cannot be entirely discounted as contributing to these findings, the prompt increase in cardiac events after just 4 days of statin withdrawal adds to the growing body of evidence suggesting that statin discontinuation can have harmful acute effects. It also brings up the question: Can starting statins benefit patients in the same time period?
Should statins be started before vascular surgery?
Schouten et al32 evaluated the effects of newly started or continued statin treatment in patients undergoing major elective vascular surgery. Patients were screened before surgery and started on statins if they were not already receiving them and their total cholesterol levels were elevated; new users received the medication for about 40 days before surgery. Of the 981 screened patients, 44 (5%) were newly started on statins and 182 (19%) were continued on their therapy. Perioperative death or myocardial infarction occurred in 22 (8.8%) of the statin users and 111 (14.7%) of the nonusers, a statistically significant difference. Temporary discontinuation (median 1 day) of statins in this study due to the inability to take an oral medication did not appear to affect the likelihood of a myocardial infarction.
Durazzo et al23 performed a single-center, randomized, prospective, placebo-controlled, double-blind clinical trial of atorvastatin (Lipitor) 20 mg daily vs placebo in 100 patients undergoing noncardiac arterial vascular surgery. Patients were excluded if they had previously used medications to treat dyslipidemia, recently had a cardiovascular event, or had contraindications to statin treatment such as a baseline creatinine level greater than 2.0 mg/dL or severe hepatic disease. The intervention group received atorvastatin starting at least 2 weeks before surgery for a total of 45 days. Patients were then continued or started on a statin after surgery if their LDL-C level was greater than 100 mg/dL. Beta-blocker use was recommended “on the basis of current guidelines.”
One month after surgery, the LDL-C level was statistically significantly lower in the atorvastatin group. Since most patients did not continue or start statin therapy after the 45-day treatment period, the LDL-C levels were not statistically different at 3 and 6 months after surgery.
At 6 months, the rate of the primary end point (death from cardiovascular causes, nonfatal acute myocardial infarction, ischemic stroke, or unstable angina) was 26.0% in the placebo group and 8.0% in the atorvastatin group, a statistically significant difference. Three patients in the atorvastatin group had cardiac events in the first 10 days after surgery, compared with 11 patients in the placebo group. Thirteen of the 17 total cardiac events took place within 10 days after surgery.
One of the atorvastatin patients developed rhabdomyolysis and elevated aminotransferase levels.
Major noncardiac surgery
Lindenauer et al2 performed a retrospective cohort study of surgical patients who were at least 18 years old and survived beyond the second hospital day. Patients were divided into a group receiving any form of lipid-lowering treatment (of whom more than 90% were taking statins) and a group that had never never received a lipid-lowering drug or only started one on the third day of the hospitalization or later. The period of study was from January 1, 2000, to December 31, 2001.
In all, 780,591 patients from 329 hospitals throughout the United States were included, of whom only 77,082 (9.9%) received lipid-lowering therapy. Eight percent of the patients underwent vascular surgery. Not surprisingly, the treated patients were more likely to have a history of hypertension, diabetes, ischemic heart disease, or hyperlipidemia. They also were more likely to have a vascular procedure performed, to have two or more cardiac risk factors (high-risk surgery, ischemic heart disease, congestive heart failure, cerebrovascular disease, renal insufficiency, or diabetes mellitus), and to be treated with beta-blockers and angiotensin-converting enzyme inhibitors, but they were less likely to have high-risk and emergency surgery performed.
The primary end point, perioperative death, occurred in 2.13% of the treated patients and 3.05% of the nontreated group. Compared with the rate in a propensity-matched cohort, the odds ratio adjusted for unbalanced covariates was 0.62 (95% CI 0.58–0.67) in favor of lipid treatment. Stratification by cardiac risk index revealed a number needed to treat of 186 for those with no risk factors, 60 for those with two risk factors, and 30 for those with four or more risk factors.
Unfortunately, this analysis was not able to take into account whether and for how long patients were receiving lipid-lowering therapy before hospitalization. It therefore does not answer the questions of whether starting lipid-lowering therapy before surgery is beneficial or whether stopping it is harmful. It also does not shed light on whether perioperative lipid-lowering increases the risk of rhabdomyolysis or liver disease.
Carotid endarterectomy
Two recent retrospective cohort studies evaluated the outcomes in patients undergoing carotid endarterectomy.19,20
Kennedy et al19 found that patients on a statin at the time of admission who had symptomatic carotid disease had lower rates of inhospital death (adjusted odds ratio 0.24, 95% CI 0.06–0.91) and ischemic stroke or death (adjusted odds ratio 0.55, 95% CI 0.31–0.97). However, cardiac outcomes among these symptomatic patients were not significantly improved (odds ratio 0.82, 95% CI 0.45–1.50), nor was there benefit for asymptomatic patients, raising the possibility that the positive findings were due to chance or that patients at lower baseline risk for vascular events may have less benefit.
McGirt et al20 performed a similar study; they did not, however, distinguish whether patients had symptomatic vs asymptomatic carotid disease. The 30-day risk of perioperative stroke was lower in patients treated with a statin, with an odds ratio of 0.41 (95% CI 0.18–0.93); the odds ratio for death was 0.21 (95% CI 0.05–0.96). Cardiac outcomes were not significantly affected.
Coronary artery bypass graft surgery
According to the NCEP recommendations, nearly all patients undergoing CABG should already be on a statin before surgery since they all have known coronary artery disease. Multiple observational studies have offered confirmatory evidence that statins are beneficial in this setting.34–38
Liakopoulos et al39 evaluated whether the anti-inflammatory effects of statins may, in part, account for their beneficial effect in the perioperative period. The authors prospectively matched 18 patients who were taking statins and were referred for elective CABG with 18 patients who were not prescribed statins previously. The only major measured baseline characteristic that differed between the two groups was a statistically significantly lower LDL-C level in the statin group. The operative characteristics did not differ, and cytokine levels at baseline were similar.
Tumor necrosis factor alpha levels increased significantly in the control group but did not change significantly in the statin group. Interleukin 8 increased in both groups by a similar amount. Interleukin 6 (the major inducer of C-reactive protein) increased from baseline in both groups but did not increase nearly as much in the statin group as in the control group; the intergroup difference was statistically significant. The anti-inflammatory cytokine interleukin 10 increased minimally from baseline in the control group, while the statin group’s levels increased significantly above baseline and those of the control group.
Christenson40 also found that inflammatory markers were improved with pre-CABG statin treatment in a small randomized trial in which patients received simvastatin 20 mg 4 weeks prior to CABG surgery vs no statin. Interestingly, far fewer statin-treated patients developed thrombocytosis (platelet count > 400 × 109/L) than did control patients (3% vs 81%, P < .0001).
RISKS OF PERIOPERATIVE STATINS
The risks associated with statin therapy in general appear low, but specific perioperative risks have not been well studied.
Baigent et al,41 in a meta-analysis of randomized trials of nonperioperative statin therapy, found that rhabdomyolysis occurred in 9 (0.023%) of 39,884 patients receiving statins vs 6 (0.015%) of the 39,817 controls, with a number needed to harm of 12,500. Moreover, the rates of nonvascular death and cancer did not increase. It is plausible that the risk is somewhat greater in the perioperative setting but is likely not enough to outweigh the potential benefits, especially since the risk of ischemic vascular events is particularly high then.
Some of the perioperative studies cited above specifically addressed potential risks. For example, in the study by Schouten et al,32 mild creatine kinase elevations were more common in the statin-treated group, but the incidence of moderate and severe creatine kinase elevations did not differ significantly. No case of rhabdomyolysis occurred, and length of surgery was the only predictor of myopathy. MIRACL and PROVE-IT revealed similar safety profiles; aminotransferase levels normalized when statins were stopped, and no cases of rhabdomyolysis occurred.11,12 In the vascular surgery study by Durazzo et al,23 1 (2%) of the 50 atorvastatin-treated patients developed both rhabdomyolysis and elevated aminotransferase levels that prompted discontinuation of the statin.
Overall, the observational studies do not indicate that statin continuation or treatment is harmful in perioperative patients. However, these studies did not specifically evaluate patients with acute insults from surgery such as sepsis, renal failure, or hepatitis. It is unknown what effect statin therapy would have in those patients and whether statins should be selectively discontinued in patients who develop major hepatic, musculoskeletal, or renal complications after surgery.
OUR RECOMMENDATIONS
Before CABG or vascular surgery
Given the NCEP recommendations, existing primary and secondary prevention studies, observational studies of CABG and noncardiac vascular surgery patients, and the one randomized trial of vascular surgery patients, data support the use of statins in nearly all patients undergoing cardiac or vascular surgery. We advocate starting statins in the perioperative period to take advantage of their rapid-acting pleiotropic effects, and continuing them long-term to take advantage of their lipid-lowering effects. This recommendation is in line with the recently released American College of Cardiology/American Heart Association (ACC/AHA) 2007 perioperative guidelines that state “for patients undergoing vascular surgery with or without clinical risk factors, statin use is reasonable.”42
Although the ideal time to start statins is not certain, the study by Durazzo et al23 suggests that they should be started at least 2 weeks before surgery if possible. Moreover, patients already taking statins should definitely not have their statins discontinued if at all possible.
Before major nonvascular surgery
For patients undergoing major nonvascular (intermediate-risk) surgery, physicians should first ascertain if the patient has an indication for statin therapy based on current nonsurgical lipid level recommendations. However, even if there is no clear indication for statin therapy based on NCEP guidelines, we endorse the recently released ACC/AHA perioperative guidelines that state that statin therapy can be considered in patients with a risk factor who are undergoing intermediate-risk procedures. Moreover, we wholeheartedly support the ACC/AHA’s strongest recommendation that patients who are already receiving statins and are undergoing noncardiac surgery should not have their statins discontinued.
When to discontinue statins?
The risk of harm overall appears to be minimal and certainly less than the likelihood of benefit. It is reasonable to observe patients postoperatively for adverse clinical events that may increase the risk of perioperative statin treatment, such as acute renal failure, hepatic failure, or sepsis, but whether statins should be stopped in patients with these complications remains unknown; we advocate individualizing the decision.
More studies needed
We need more data on whether moderate-risk patients undergoing moderate-risk surgery benefit from perioperative statin therapy, when therapy should be started, whether therapy should be started on the day of surgery if it was not started earlier, which statin and what doses are optimal, how long therapy should be continued, and what degree of risk is associated with perioperative statin therapy.
Fortunately, important data should be forthcoming in the next few years: the Dutch Echocardiographic Cardiac Risk Evaluation Applying Stress Echocardiography (DECREASE-IV) study43 is a 4-year two-by-two factorial placebo-controlled study evaluating the use of fluvastatin (Lescol) and bisoprolol (Zebeta, a beta-blocker) separately and together in patients who are older than 40 years, are undergoing elective noncardiac surgery, have an estimated risk of cardiovascular death of more than 1%, have not used statins previously, and do not have elevated cholesterol.
- Grant PJ, Kedia N. Should statins be discontinued preoperatively? IMPACT consults. Proceedings of the 2nd Annual Cleveland Clinic Perioperative Medicine Summit. Cleve Clin J Med 2006; 73 Electronic suppl 1:S9–S10.
- Lindenauer PK, Pekow P, Wang K, Gutierrez B, Benjamin EM. Lipid-lowering therapy and in-hospital mortality following major noncardiac surgery. JAMA 2004; 291:2092–2099.
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004; 351:2795–2804.
- Mangano DT, Layug EL, Wallace A, Tateo I. Effect of atenolol on mortality and cardiovascular morbidity after noncardiac surgery. Multicenter Study of Perioperative Ischemia Research Group. N Engl J Med 1996; 335:1713–1720.
- Poldermans D, Boersma E, Bax JJ, et al. The effect of bisoprolol on perioperative mortality and myocardial infarction in high-risk patients undergoing vascular surgery. Dutch Echocardiographic Cardiac Risk Evaluation Applying Stress Echocardiography Study Group. N Engl J Med 1999; 341:1789–1794.
- Brady AR, Gibbs JS, Greenhalgh RM, Powell JT, Sydes MR. Perioperative beta-blockade (POBBLE) for patients undergoing infrarenal vascular surgery: results of a randomized double-blind controlled trial. J Vasc Surg 2005; 41:602–609.
- Juul AB, Wetterslev J, Gluud C, et al. Effect of perioperative beta blockade in patients with diabetes undergoing major non-cardiac surgery: randomised placebo controlled, blinded multicentre trial. BMJ 2006; 332:1482.
- Yang H, Raymer K, Butler R, Parlow J, Roberts R. The effects of perioperative beta-blockade: results of the Metoprolol after Vascular Surgery (MaVS) study, a randomized controlled trial. Am Heart J 2006; 152:983–990.
- Ridker PM, Cannon CP, Morrow D, et al. C-reactive protein levels and outcomes after statin therapy. N Engl J Med 2005; 352:20–28.
- Ito MK, Talbert RL, Tsimikas S. Statin-associated pleiotropy: possible beneficial effects beyond cholesterol reduction. Pharmacotherapy 2006; 26:85S–97S.
- Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495–1504.
- Schwartz GG, Olsson AG, Ezekowitz MD, et al. Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA 2001; 285:1711–1718.
- Lefer AM, Campbell B, Shin YK, Scalia R, Hayward R, Lefer DJ. Simvastatin preserves the ischemic-reperfused myocardium in normocholesterolemic rat hearts. Circulation 1999; 100:178–184.
- Endres M, Laufs U, Liao JK, Moskowitz MA. Targeting eNOS for stroke protection. Trends Neurosci 2004; 27:283–289.
- Osborne JA, Lento PH, Siegfried MR, Stahl GL, Fusman B, Lefer AM. Cardiovascular effects of acute hypercholesterolemia in rabbits. Reversal with lovastatin treatment. J Clin Invest 1989; 83:465–473.
- Sironi L, Cimino M, Guerrini U, et al. Treatment with statins after induction of focal ischemia in rats reduces the extent of brain damage. Arterioscler Thromb Vasc Biol 2003; 23:322–327.
- Fonarow GC, Wright RS, Spencer FA, et al. Effect of statin use within the first 24 hours of admission for acute myocardial infarction on early morbidity and mortality. Am J Cardiol 2005; 96:611–616.
- Heeschen C, Hamm CW, Laufs U, Snapinn S, Bohm M, White HD. Withdrawal of statins increases event rates in patients with acute coronary syndromes. Circulation 2002; 105:1446–1452.
- Kennedy J, Quan H, Buchan AM, Ghali WA, Feasby TE. Statins are associated with better outcomes after carotid endarterectomy in symptomatic patients. Stroke 2005; 36:2072–2076.
- McGirt MJ, Perler BA, Brooke BS, et al. 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors reduce the risk of perioperative stroke and mortality after carotid endarterectomy. J Vasc Surg 2005; 42:829–836.
- Hindler K, Shaw AD, Samuels J, Fulton S, Collard CD, Riedel B. Improved postoperative outcomes associated with preoperative statin therapy. Anesthesiology 2006; 105:1260–1272.
- Kapoor AS, Kanji H, Buckingham J, Devereaux PJ, McAlister FA. Strength of evidence for perioperative use of statins to reduce cardiovascular risk: systematic review of controlled studies. BMJ 2006; 333:1149.
- Durazzo AE, Machado FS, Ikeoka DT, et al. Reduction in cardiovascular events after vascular surgery with atorvastatin: a randomized trial. J Vasc Surg 2004; 39:967–975.
- Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 2004; 110:227–239.
- Kertai MD, Boersma E, Westerhout CM, et al. A combination of statins and beta-blockers is independently associated with a reduction in the incidence of perioperative mortality and nonfatal myocardial infarction in patients undergoing abdominal aortic aneurysm surgery. Eur J Vasc Endovasc Surg 2004; 28:343–352.
- Poldermans D, Bax JJ, Kertai MD, et al. Statins are associated with a reduced incidence of perioperative mortality in patients undergoing major noncardiac vascular surgery. Circulation 2003; 107:1848–1851.
- Ward RP, Leeper NJ, Kirkpatrick JN, Lang RM, Sorrentino MJ, Williams KA. The effect of preoperative statin therapy on cardiovascular outcomes in patients undergoing infrainguinal vascular surgery. Int J Cardiol 2005; 104:264–268.
- O’Neil-Callahan K, Katsimaglis G, Tepper MR, et al. Statins decrease perioperative cardiac complications in patients undergoing non-cardiac vascular surgery: the Statins for Risk Reduction in Surgery (StaRRS) study. J Am Coll Cardiol 2005; 45:336–342.
- Abbruzzese TA, Havens J, Belkin M, et al. Statin therapy is associated with improved patency of autogenous infrainguinal bypass grafts. J Vasc Surg 2004; 39:1178–1185.
- Boersma E, Poldermans D, Bax JJ, et al. Predictors of cardiac events after major vascular surgery: role of clinical characteristics, dobutamine echocardiography, and beta-blocker therapy. JAMA 2001; 285:1865–1873.
- Landesberg G, Mosseri M, Wolf YG, et al. Preoperative thallium scanning, selective coronary revascularization, and long-term survival after major vascular surgery. Circulation 2003; 108:177–183.
- Schouten O, Kertai MD, Bax JJ, et al. Safety of perioperative statin use in high-risk patients undergoing major vascular surgery. Am J Cardiol 2005; 95:658–660.
- Le Manach Y, Godet G, Coriat P, et al. The impact of postoperative discontinuation or continuation of chronic statin therapy on cardiac outcome after major vascular surgery. Anesth Analg 2007; 104:1326–1333.
- Ali IS, Buth KJ. Preoperative statin use and outcomes following cardiac surgery. Int J Cardiol 2005; 103:12–18.
- Clark LL, Ikonomidis JS, Crawford FA, et al. Preoperative statin treatment is associated with reduced postoperative mortality and morbidity in patients undergoing cardiac surgery: an 8-year retrospective cohort study. J Thorac Cardiovasc Surg 2006; 131:679–685.
- Pan W, Pintar T, Anton J, Lee VV, Vaughn WK, Collard CD. Statins are associated with a reduced incidence of perioperative mortality after coronary artery bypass graft surgery. Circulation 2004; 110(suppl 2):II45–II49.
- Pascual DA, Arribas JM, Tornel PL, et al. Preoperative statin therapy and troponin T predict early complications of coronary artery surgery. Ann Thorac Surg 2006; 81:78–83.
- Dotani MI, Elnicki DM, Jain AC, Gibson CM. Effect of preoperative statin therapy and cardiac outcomes after coronary artery bypass grafting. Am J Cardiol 2000; 86:1128–1130.
- Liakopoulos OJ, Dorge H, Schmitto JD, Nagorsnik U, Grabedunkel J, Schoendube FA. Effects of preoperative statin therapy on cytokines after cardiac surgery. Thorac Cardiovasc Surg 2006; 54:250–254.
- Christenson JT. Preoperative lipid-control with simvastatin reduces the risk of postoperative thrombocytosis and thrombotic complications following CABG. Eur J Cardiothorac Surg 1999; 15:394–399.
- Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 2005; 366:1267–1278.
- Fleisher LA, Beckman JA, Brown KA, et al. ACC/AHA 2007 Guidelines on Perioperative Cardiovascular Evaluation and Care for Noncardiac Surgery. A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation 2007; 116:e418–e499.
- Schouten O, Poldermans D, Visser L, et al. Fluvastatin and bisoprolol for the reduction of perioperative cardiac mortality and morbidity in high-risk patients undergoing non-cardiac surgery: rationale and design of the DECREASE-IV study. Am Heart J 2004; 148:1047–1052.
- Amar D, Zhang H, Heerdt PM, Park B, Fleisher M, Thaler HT. Statin use is associated with a reduction in atrial fibrillation after noncardiac thoracic surgery independent of C-reactive protein. Chest 2005; 128:3421–3427.
- Grant PJ, Kedia N. Should statins be discontinued preoperatively? IMPACT consults. Proceedings of the 2nd Annual Cleveland Clinic Perioperative Medicine Summit. Cleve Clin J Med 2006; 73 Electronic suppl 1:S9–S10.
- Lindenauer PK, Pekow P, Wang K, Gutierrez B, Benjamin EM. Lipid-lowering therapy and in-hospital mortality following major noncardiac surgery. JAMA 2004; 291:2092–2099.
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004; 351:2795–2804.
- Mangano DT, Layug EL, Wallace A, Tateo I. Effect of atenolol on mortality and cardiovascular morbidity after noncardiac surgery. Multicenter Study of Perioperative Ischemia Research Group. N Engl J Med 1996; 335:1713–1720.
- Poldermans D, Boersma E, Bax JJ, et al. The effect of bisoprolol on perioperative mortality and myocardial infarction in high-risk patients undergoing vascular surgery. Dutch Echocardiographic Cardiac Risk Evaluation Applying Stress Echocardiography Study Group. N Engl J Med 1999; 341:1789–1794.
- Brady AR, Gibbs JS, Greenhalgh RM, Powell JT, Sydes MR. Perioperative beta-blockade (POBBLE) for patients undergoing infrarenal vascular surgery: results of a randomized double-blind controlled trial. J Vasc Surg 2005; 41:602–609.
- Juul AB, Wetterslev J, Gluud C, et al. Effect of perioperative beta blockade in patients with diabetes undergoing major non-cardiac surgery: randomised placebo controlled, blinded multicentre trial. BMJ 2006; 332:1482.
- Yang H, Raymer K, Butler R, Parlow J, Roberts R. The effects of perioperative beta-blockade: results of the Metoprolol after Vascular Surgery (MaVS) study, a randomized controlled trial. Am Heart J 2006; 152:983–990.
- Ridker PM, Cannon CP, Morrow D, et al. C-reactive protein levels and outcomes after statin therapy. N Engl J Med 2005; 352:20–28.
- Ito MK, Talbert RL, Tsimikas S. Statin-associated pleiotropy: possible beneficial effects beyond cholesterol reduction. Pharmacotherapy 2006; 26:85S–97S.
- Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495–1504.
- Schwartz GG, Olsson AG, Ezekowitz MD, et al. Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA 2001; 285:1711–1718.
- Lefer AM, Campbell B, Shin YK, Scalia R, Hayward R, Lefer DJ. Simvastatin preserves the ischemic-reperfused myocardium in normocholesterolemic rat hearts. Circulation 1999; 100:178–184.
- Endres M, Laufs U, Liao JK, Moskowitz MA. Targeting eNOS for stroke protection. Trends Neurosci 2004; 27:283–289.
- Osborne JA, Lento PH, Siegfried MR, Stahl GL, Fusman B, Lefer AM. Cardiovascular effects of acute hypercholesterolemia in rabbits. Reversal with lovastatin treatment. J Clin Invest 1989; 83:465–473.
- Sironi L, Cimino M, Guerrini U, et al. Treatment with statins after induction of focal ischemia in rats reduces the extent of brain damage. Arterioscler Thromb Vasc Biol 2003; 23:322–327.
- Fonarow GC, Wright RS, Spencer FA, et al. Effect of statin use within the first 24 hours of admission for acute myocardial infarction on early morbidity and mortality. Am J Cardiol 2005; 96:611–616.
- Heeschen C, Hamm CW, Laufs U, Snapinn S, Bohm M, White HD. Withdrawal of statins increases event rates in patients with acute coronary syndromes. Circulation 2002; 105:1446–1452.
- Kennedy J, Quan H, Buchan AM, Ghali WA, Feasby TE. Statins are associated with better outcomes after carotid endarterectomy in symptomatic patients. Stroke 2005; 36:2072–2076.
- McGirt MJ, Perler BA, Brooke BS, et al. 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors reduce the risk of perioperative stroke and mortality after carotid endarterectomy. J Vasc Surg 2005; 42:829–836.
- Hindler K, Shaw AD, Samuels J, Fulton S, Collard CD, Riedel B. Improved postoperative outcomes associated with preoperative statin therapy. Anesthesiology 2006; 105:1260–1272.
- Kapoor AS, Kanji H, Buckingham J, Devereaux PJ, McAlister FA. Strength of evidence for perioperative use of statins to reduce cardiovascular risk: systematic review of controlled studies. BMJ 2006; 333:1149.
- Durazzo AE, Machado FS, Ikeoka DT, et al. Reduction in cardiovascular events after vascular surgery with atorvastatin: a randomized trial. J Vasc Surg 2004; 39:967–975.
- Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III guidelines. Circulation 2004; 110:227–239.
- Kertai MD, Boersma E, Westerhout CM, et al. A combination of statins and beta-blockers is independently associated with a reduction in the incidence of perioperative mortality and nonfatal myocardial infarction in patients undergoing abdominal aortic aneurysm surgery. Eur J Vasc Endovasc Surg 2004; 28:343–352.
- Poldermans D, Bax JJ, Kertai MD, et al. Statins are associated with a reduced incidence of perioperative mortality in patients undergoing major noncardiac vascular surgery. Circulation 2003; 107:1848–1851.
- Ward RP, Leeper NJ, Kirkpatrick JN, Lang RM, Sorrentino MJ, Williams KA. The effect of preoperative statin therapy on cardiovascular outcomes in patients undergoing infrainguinal vascular surgery. Int J Cardiol 2005; 104:264–268.
- O’Neil-Callahan K, Katsimaglis G, Tepper MR, et al. Statins decrease perioperative cardiac complications in patients undergoing non-cardiac vascular surgery: the Statins for Risk Reduction in Surgery (StaRRS) study. J Am Coll Cardiol 2005; 45:336–342.
- Abbruzzese TA, Havens J, Belkin M, et al. Statin therapy is associated with improved patency of autogenous infrainguinal bypass grafts. J Vasc Surg 2004; 39:1178–1185.
- Boersma E, Poldermans D, Bax JJ, et al. Predictors of cardiac events after major vascular surgery: role of clinical characteristics, dobutamine echocardiography, and beta-blocker therapy. JAMA 2001; 285:1865–1873.
- Landesberg G, Mosseri M, Wolf YG, et al. Preoperative thallium scanning, selective coronary revascularization, and long-term survival after major vascular surgery. Circulation 2003; 108:177–183.
- Schouten O, Kertai MD, Bax JJ, et al. Safety of perioperative statin use in high-risk patients undergoing major vascular surgery. Am J Cardiol 2005; 95:658–660.
- Le Manach Y, Godet G, Coriat P, et al. The impact of postoperative discontinuation or continuation of chronic statin therapy on cardiac outcome after major vascular surgery. Anesth Analg 2007; 104:1326–1333.
- Ali IS, Buth KJ. Preoperative statin use and outcomes following cardiac surgery. Int J Cardiol 2005; 103:12–18.
- Clark LL, Ikonomidis JS, Crawford FA, et al. Preoperative statin treatment is associated with reduced postoperative mortality and morbidity in patients undergoing cardiac surgery: an 8-year retrospective cohort study. J Thorac Cardiovasc Surg 2006; 131:679–685.
- Pan W, Pintar T, Anton J, Lee VV, Vaughn WK, Collard CD. Statins are associated with a reduced incidence of perioperative mortality after coronary artery bypass graft surgery. Circulation 2004; 110(suppl 2):II45–II49.
- Pascual DA, Arribas JM, Tornel PL, et al. Preoperative statin therapy and troponin T predict early complications of coronary artery surgery. Ann Thorac Surg 2006; 81:78–83.
- Dotani MI, Elnicki DM, Jain AC, Gibson CM. Effect of preoperative statin therapy and cardiac outcomes after coronary artery bypass grafting. Am J Cardiol 2000; 86:1128–1130.
- Liakopoulos OJ, Dorge H, Schmitto JD, Nagorsnik U, Grabedunkel J, Schoendube FA. Effects of preoperative statin therapy on cytokines after cardiac surgery. Thorac Cardiovasc Surg 2006; 54:250–254.
- Christenson JT. Preoperative lipid-control with simvastatin reduces the risk of postoperative thrombocytosis and thrombotic complications following CABG. Eur J Cardiothorac Surg 1999; 15:394–399.
- Baigent C, Keech A, Kearney PM, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 2005; 366:1267–1278.
- Fleisher LA, Beckman JA, Brown KA, et al. ACC/AHA 2007 Guidelines on Perioperative Cardiovascular Evaluation and Care for Noncardiac Surgery. A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery). Circulation 2007; 116:e418–e499.
- Schouten O, Poldermans D, Visser L, et al. Fluvastatin and bisoprolol for the reduction of perioperative cardiac mortality and morbidity in high-risk patients undergoing non-cardiac surgery: rationale and design of the DECREASE-IV study. Am Heart J 2004; 148:1047–1052.
- Amar D, Zhang H, Heerdt PM, Park B, Fleisher M, Thaler HT. Statin use is associated with a reduction in atrial fibrillation after noncardiac thoracic surgery independent of C-reactive protein. Chest 2005; 128:3421–3427.
KEY POINTS
- Experiments in animals suggest that statins, given shortly before or after a cardiovascular event, confer benefit before any changes in lipids are measurable.
- Retrospective and prospective studies indicate that patients with either acute myocardial infarction or acute coronary syndrome who are already receiving statins should not have them stopped, and those who had not been receiving statins should receive them immediately.
- Most patients undergoing coronary artery bypass grafting or noncardiac vascular surgery should already be receiving a statin. These drugs can also be considered in patients undergoing intermediate-risk nonvascular surgery. Patients who have been receiving statins prior to surgery should not have them stopped for surgery.
And then there were none? An internist’s reflections
I observe with sadness the decreasing number of our brightest medical students entering into internal medicine careers and other “cognitive” subspecialties. Much effort has been spent on many fronts to understand and reverse this trend, with limited success.
At the other end of their careers, physicians seem to be looking for ways to retire earlier or to withdraw from their usual and customary practice of internal medicine. Hearing these senior physicians’ reasons for withdrawing from clinical practice evokes an even stronger response in me, especially when the physician is a really good one, a role model for the next generation of our internists currently in training.
In an essay in this issue, Dr. Thomas Lansdale, internist and former chairman of medicine at a community teaching hospital, eloquently expresses a common theme: medicine just isn’t that much fun anymore. We don’t generally run this type of article in the Journal. But Dr. Lansdale’s words reflect an undercurrent that is changing the landscape of American medicine. We would like to hear responses from our readers, but not to simply agree or disagree with Dr. Lansdale. Rather, we’d like to hear some solutions, which we hope to print in a future issue.
I have known Dr. Lansdale for over 20 years; we trained together as residents at the University of Pennsylvania. He was a year or so behind me, and over the years I have had the opportunity to follow his clinical career from afar and occasionally to discuss patient care and education issues. He was (and is) a thoughtful and extremely insightful internist, devoted and capable of delivering the highest quality of care to his patients. He has always approached medicine, his trainees, and his patients in a serious and respectful manner. His words should prompt some serious self-reflection.
Send your comments to ccjm@ccf.org. Please note that sending your comments constitutes permission to publish them, and also that we cannot respond to or publish all submissions.
I observe with sadness the decreasing number of our brightest medical students entering into internal medicine careers and other “cognitive” subspecialties. Much effort has been spent on many fronts to understand and reverse this trend, with limited success.
At the other end of their careers, physicians seem to be looking for ways to retire earlier or to withdraw from their usual and customary practice of internal medicine. Hearing these senior physicians’ reasons for withdrawing from clinical practice evokes an even stronger response in me, especially when the physician is a really good one, a role model for the next generation of our internists currently in training.
In an essay in this issue, Dr. Thomas Lansdale, internist and former chairman of medicine at a community teaching hospital, eloquently expresses a common theme: medicine just isn’t that much fun anymore. We don’t generally run this type of article in the Journal. But Dr. Lansdale’s words reflect an undercurrent that is changing the landscape of American medicine. We would like to hear responses from our readers, but not to simply agree or disagree with Dr. Lansdale. Rather, we’d like to hear some solutions, which we hope to print in a future issue.
I have known Dr. Lansdale for over 20 years; we trained together as residents at the University of Pennsylvania. He was a year or so behind me, and over the years I have had the opportunity to follow his clinical career from afar and occasionally to discuss patient care and education issues. He was (and is) a thoughtful and extremely insightful internist, devoted and capable of delivering the highest quality of care to his patients. He has always approached medicine, his trainees, and his patients in a serious and respectful manner. His words should prompt some serious self-reflection.
Send your comments to ccjm@ccf.org. Please note that sending your comments constitutes permission to publish them, and also that we cannot respond to or publish all submissions.
I observe with sadness the decreasing number of our brightest medical students entering into internal medicine careers and other “cognitive” subspecialties. Much effort has been spent on many fronts to understand and reverse this trend, with limited success.
At the other end of their careers, physicians seem to be looking for ways to retire earlier or to withdraw from their usual and customary practice of internal medicine. Hearing these senior physicians’ reasons for withdrawing from clinical practice evokes an even stronger response in me, especially when the physician is a really good one, a role model for the next generation of our internists currently in training.
In an essay in this issue, Dr. Thomas Lansdale, internist and former chairman of medicine at a community teaching hospital, eloquently expresses a common theme: medicine just isn’t that much fun anymore. We don’t generally run this type of article in the Journal. But Dr. Lansdale’s words reflect an undercurrent that is changing the landscape of American medicine. We would like to hear responses from our readers, but not to simply agree or disagree with Dr. Lansdale. Rather, we’d like to hear some solutions, which we hope to print in a future issue.
I have known Dr. Lansdale for over 20 years; we trained together as residents at the University of Pennsylvania. He was a year or so behind me, and over the years I have had the opportunity to follow his clinical career from afar and occasionally to discuss patient care and education issues. He was (and is) a thoughtful and extremely insightful internist, devoted and capable of delivering the highest quality of care to his patients. He has always approached medicine, his trainees, and his patients in a serious and respectful manner. His words should prompt some serious self-reflection.
Send your comments to ccjm@ccf.org. Please note that sending your comments constitutes permission to publish them, and also that we cannot respond to or publish all submissions.
A case of refractory diarrhea
A 68-year-old white woman with irritable bowel syndrome has had worsening symptoms of right-sided abdominal pain, excessive bloating, and loose stools. Her bowel movements have increased from one a day to two or three a day. She has not noted any mucus or blood in the stool. She cannot identify any alleviating or aggravating factors, and the pain is not related to eating.
She consumes a normal diet, including meat and dairy. Over-the-counter antidiarrheal medications do not relieve the symptoms. She has had no fevers, chills, or night sweats, and she has not lost weight over the past year.
Her medical history includes breast cancer (in remission), alcohol abuse (in remission), and hypothyroidism, osteoporosis, and supraventricular tachycardia, all controlled with treatment as noted below. She has never undergone abdominal surgery.
A general review of systems is normal. Her current medications include oxybutynin (available as Ditropan, others), calcium polycarbophil (FiberCon, others), risedronate (Actonel), levothyroxine (Synthroid, others), simethicone (Maalox Anti-Gas, others), atenolol (Tenormin), trazodone (Desyrel), a calcium supplement, and aspirin. She began taking duloxetine (Cymbalta) 18 months ago, and the dose was increased from 60 mg to 90 mg 1 week before this visit.
She has never smoked, and she has abstained from alcohol for 10 years. She has no family history of colon cancer, celiac disease, or inflammatory bowel disease. She has not traveled outside the country in the past several years, and she notes no change in her source of drinking water.
On physical examination, she does not appear to be in acute distress. Her pulse is 64 and her blood pressure is 112/78 mm Hg. The cardiopulmonary examination is normal. Her abdomen is soft, symmetrical, nondistended, and nontender. Bowel sounds are normal. No abdominal masses, palpable organomegaly, or abdominal bruits are noted.
Results of basic laboratory tests, including thyroid-stimulating hormone (TSH), complete blood count, blood chemistries, renal function, and liver function, are normal. Colonoscopy shows normal mucosa as far as the cecum.
DIFFERENTIAL DIAGNOSIS
1. In addition to irritable bowel syndrome, which of these can explain her symptoms?
- Ulcerative colitis
- Celiac disease
- Microscopic colitis
- Hyperthyroidism
- Lactase deficiency
Ulcerative colitis typically presents with blood and mucus in the stool and gross abnormalities on colonoscopy, none of which is present in this patient.
Hyperthyroidism can be ruled out by the normal TSH level.
Lactase deficiency or lactose intolerance is unlikely because it is present in only 15% of people of northern European descent (compared with 80% of blacks and Hispanics and up to 100% of Native Americans and Asians).1 Furthermore, her pain is apparently not related to consuming dairy products.
The hydrogen breath test can aid in the diagnosis of lactase deficiency. This test relies on the breakdown of malabsorbed lactose by colonic flora. This is the most widely used test for this deficiency, but its high false-negative rate of 25% means that a negative result does not exclude the diagnosis and should not be relied on in working up a patient with chronic diarrhea.2 Simply noting whether symptoms develop after ingesting 50 g of lactose is clinically useful when lactase deficiency is suspected.
Based on the information so far, it is reasonable in this patient to evaluate for celiac disease and for microscopic colitis.
Celiac disease, also called gluten-sensitive enteropathy, has a varied presentation that includes nonspecific symptoms such as those in this patient. Classically, it causes diarrhea, but patients may present with a single nutrient deficiency and no diarrhea.
This patient lacks the elevated alkaline phosphatase or evidence of vitamin deficiencies characteristic of malabsorption in celiac disease (ie, vitamins A, B12, D, K, and folate)3. She also lacks evidence of malnutrition, such as iron deficiency anemia, weight loss, or low serum albumin. Finally, she does not have the dermatitis herpetiformis rash to suggest autoimmune gluten-sensitive enteropathy, nor does she have evidence of follicular hyperplasia or petechiae due to vitamin malabsorption.3
Because no single serologic test is ideal for diagnosing gluten-sensitive enteropathy, several tests are typically used: immunoglobulin A (IgA) antigliadin antibody, IgG antigliadin antibody, IgA antitransglutaminase antibody, and IgA antiendomysial antibody. IgA antitransglutaminase antibody is 92% to 98% sensitive and 91% to 100% specific for celiac disease. IgG antigliadin antibody is 92% to 97% sensitive and 99% specific. The positive predictive value of the IgA and IgG antigliadin antibody tests is less than 2% in the general population, whereas the positive predictive value for antiendomysial antibody and antitransglutaminase antibody are 15.7% and 21.8%, respectively.4 A positive serologic test for antiendomysial antibody is nearly 100% specific.
Our patient’s entire celiac antibody panel is negative, and thus celiac disease is unlikely.
Case continued: Features of microscopic colitis
In our patient, colonic biopsy reveals a mildly expanded lamina propria, intraepithelial lymphocytes, and a patchy but prominent thickening of the subepithelial collagen table. This set of features is consistent with collagenous colitis, a variant of microscopic colitis. Histologic signs on biopsy specimens are fairly specific for the disease.5
Chronic, intermittent, secretory diarrhea without bleeding is the hallmark of microscopic colitis. Associated symptoms may include abdominal pain, weight loss, and fatigue. If biopsies are not taken at the time of the initial evaluation, and the colonic pathology is overlooked, patients with collagenous colitis may be diagnosed with irritable bowel syndrome with diarrhea.6 The sedimentation rate is often elevated, and the antinuclear antibody test can be positive.7 Steatorrhea or protein-losing enteropathy can occur, and fecal leukocytes are present in more than 50% of patients.8
This patient fits well the demographics of the typical collagenous colitis patient: ie, a middle-aged woman in her 6th decade in otherwise good general health. The female-to-male ratio is 15:1 overall, although the relative frequency of collagenous colitis in women is greater than that of lymphocytic colitis.9 In a population-based study, the incidence of collagenous colitis was 5.1 per 100,000 per year, with a prevalence of 36 per 100,000; the incidence of lymphocytic colitis was 9.8 per 100,000 per year, with a prevalence of 64 per 100,000.10
Symptoms are typically vague and range from an annoyance to more than 20 non-bloody stools per day. The course of the disease also varies. Case series have reported a spontaneous remission rate of 15% to 20%,11 though flare-ups are common. Microscopic colitis is largely a benign disease. It does not increase a person’s risk of colon cancer.
CAUSES OF COLLAGENOUS COLITIS
2. What causes of collagenous colitis have been identified?
- Alcohol abuse
- Previous gastrointestinal surgery
- Drug-induced injury to colon
Neither alcohol use nor previous gastrointestinal surgery has been associated with the development of collagenous colitis.
Collagenous colitis has, however, been linked to several causes. Abnormal collagen metabolism has been demonstrated in patients as a result of increased expression of procollagen I and metalloproteinase inhibitor TIMP-1.12 Bacterial toxins and a bile-acid malabsorption defect in the terminal ileum and subsequent exposure of the colon to high concentrations of bile acids have also been linked to the development of collagenous colitis.
Many drugs have been linked to the development of collagenous colitis. Damage to the large intestine related to the use of non-steroidal anti-inflammatory drugs has been attributed to the blockage of prostaglandin synthesis.13 Simvastatin (Zocor), lansoprazole (Prilosec), and ticlopidine (Ticlid) have been linked to collagenous colitis; ticlopidine, flutamide (Eulexin), gold salts, lansoprazole, and sertraline (Zoloft) have been linked to the development of lymphocytic colitis.14 In one small series, patients developed colitis after switching from omeprazole (Prevacid) to lansoprazole. All patients had their symptoms and biopsy findings resolve within 1 week of stopping the drug.15
WHICH DRUG IS BEST?
3. Which drug is best for microscopic colitis, based on the current evidence?
- Bismuth (eg, Kaopectate, Pepto-Bismol)
- Sulfasalazine (Sulfazine)
- Budesonide (Entocort)
- Prednisolone
Studies have evaluated bismuth subsalicylate, Boswellia serrata extract, probiotics, prednisolone, budesonide, and other drugs for treating collagenous colitis.16
Bismuth trials have been small. In an open-label study of bismuth,17 symptoms improved in 11 of 12 patients.
Prednisolone recipients had a trend towards clinical response with treatment vs placebo, but it was not statistically significant, and there was incomplete remission of disease.18
Boswellia serrataextract19 and probiotics20 showed no clinical improvement.
Cholestyramine has been shown to be helpful when used in conjunction with an anti-inflammatory agent,21 and it may be helpful when used alone.
Aminosalicylate compounds have not been tested in prospective randomized trials, even though they are the cornerstone of treatment for ulcerative colitis. Retrospective trials have been equivocal.22
Budesonide currently has the best evidence of efficacy in collagenous colitis,23,24 and some evidence suggests it is also effective for other variants of microscopic colitis.
A total of 94 patients were enrolled in three placebo-controlled trials of budesonide at 9 mg daily or on a tapering schedule for 6 to 8 weeks. The pooled odds ratio for clinical response to treatment with budesonide was 12.32 (95% confidence interval 5.53–27.46), with a number needed to treat of 1.58. Significant histologic improvement with treatment was noted in all three trials.23
Quality of life has also been studied in patients with microscopic colitis who take budesonide. Symptoms, emotional functioning, and physical functioning are improved. Budesonide also improved stool consistency and significantly reduced the mean stool frequency compared with placebo.24
Compared with cortisol, budesonide has a 200 times greater affinity for the glucocorticoid receptor, and a 1,000 times greater topical anti-inflammatory potency. It is also well absorbed in the gastrointestinal tract but is substantially modified into very weak metabolites as a result of first-pass metabolism in the liver.25 This localized effect further supports the use of budesonide in patients with any form of microscopic colitis.
Although studies have shown budesonide to be effective, not every patient with a histologic diagnosis of microscopic colitis needs it. It is reasonable to try antidiarrheal agents, bismuth, or both as a first step because they are inexpensive and have few side effects. If budesonide is used, it should be given for 6 to 8 weeks, then stopped, and the patient should then be monitored for symptom recurrence. If a flare does occur, budesonide can be restarted and continued as maintenance therapy.
KEY CONSIDERATIONS
Microscopic colitis is diagnosed histologically, while irritable bowel syndrome is a clinical diagnosis. In population-based cohorts of histologically confirmed microscopic colitis, 50% to 70% met symptom-based Rome criteria for the diagnosis of irritable bowel syndrome. The clinical symptom-based criteria for irritable bowel syndrome are not specific enough to rule out the diagnosis of microscopic colitis. Therefore, patients with suspected diarrhea-predominant irritable bowel syndrome should undergo colonoscopy with biopsy to investigate microscopic colitis if symptoms are not well controlled by antidiarrheal therapy.26 The patient’s management may be very different depending on whether colonoscopy is done.
Management of microscopic colitis should include stopping any drugs associated with it. Simple antidiarrheal agents should be tried first to manage symptoms. If symptoms persist, patients can be treated with budesonide (Entocort EC) 9 mg by mouth daily for 8 weeks to induce remission, or 6 mg by mouth daily for 3 months as maintenance therapy.
OUR PATIENT’S COURSE
Our patient’s medication list includes duloxetine, a serotonin-norepinephrine reuptake inhibitor related to drugs that have been associated with the development of microscopic colitis. We tapered the duloxetine, and her symptoms improved by 50%. Her symptoms were eventually controlled after an 8-week course of oral budesonide 9 mg and ongoing intermittent use of loperamide (Imodium).
- Swagerty DL, Walling AD, Klein RM. Lactose intolerance. Am Fam Physician 2002; 65:1845–1856.
- Thomas PD, Forbes A, Green J, et al. Guidelines for the investigation of chronic diarrhea, 2nd edition. Gut 2003; 52(suppl 5):1–5.
- Nelsen DA. Gluten-sensitive enteropathy (celiac disease): more common than you think. Am Fam Physician 2002; 66:2259–2266.
- Bardella MT, Trovato C, Cesana BM, Pagliari C, Gebbia C, Peracchi M. Serological markers for coeliac disease: is it time to change? Dig Liver Dis 2001; 33:426–431.
- Barta Z, Mekkel G, Csipo I, et al. Micropscopic colitis: a retrospective study of clinical presentation in 53 patients. World J Gastroenterol 2005; 11:1351–1355.
- Tremaine WJ. Diagnosing collagenous colitis: does it make a difference? Eur J Gastroenterol Hepatol 1999; 11:477–479.
- Bohr J, Tysk C, Yang P, Danielsson D, Järnerot G. Autoantibodies and immunoglobulins in collagenous colitis. Gut 1996; 39:77–81.
- Zins BJ, Tremaine WJ, Carpenter HA. Collagenous colitis: mucosal biopsies and association with fecal leukocytes. Mayo Clin Proc 1995; 70:430–433.
- Olsen M, Eriksson S, Bohr J, Järnerot G, Tysk C. Lymphocytic colitis: a retrospective clinical study of 199 Swedish patients. Gut 2004; 53:536–541.
- Pardi DS. Microscopic colitis: an update. Inflamm Bowel Dis 2004; 10:860–870.
- Fernandez-Banares F, Salas A, Esteve M, Espinos J, Forne M, Viver JM. Collagenous and lymphocytic colitis: evaluation of clinical and histological features, response to treatment, and long-term follow-up. Am J Gastroenterol 2003; 98:340–347.
- Aignet T, Neureiter D, Müller S, Küspert G, Belke J, Kirchner T. Extracellular matrix composition and gene expression in collagenous colitis. Gastroenterology 1997; 113:136–143.
- Parfitt JR, Driman DK. Pathological effects of drugs on the gastrointestinal tract: a review. Hum Pathol 2007; 38:527–536.
- Fernández-Bañares F, Esteve M, Espinós JC, et al. Drug consumption and the risk of microscopic colitis. Am J Gastroenterol 2007; 102:324–330.
- Thomson RD, Lestine LS, Bensen SP, et al. Lansoprazole-associated microscopic colitis: a case series. Am J Gastroenterol 2002; 97:2908–2913.
- Chande N, McDonald JWD, MacDonald JK. Cochrane Inflammatory Bowel Disease and Functional Bowel Disorders Group. Interventions for treating collagenous colitis. Cochrane Database Syst Rev 2007 Jan 24;(1):CD006096.
- Fine K, Lee E. Efficacy of open-label bismuth subsalicylate for the treatment of microscopic colitis. Gastroenterology 1998; 114:29–36.
- Munck LK, Kjeldsen J, Philipsen E, Fscher Hansen B. Incomplete remission with short-term prednisolone treatment in collagenous colitis: a randomized study. Scand J Gastroenterol 2003; 38:606–610.
- Madisch A, Miehlke S, Eichele E, et al. Boswellia serrata extract for the treatment of collagenous colitis: a randomized, double-blind, placebo-controlled, multicenter trial. Int J Colorectal Dis 2007; 22:1445–1451.
- Wildt S, Munck LK, Vinter-Jensen L, et al. Probiotic treatment of collagenous colitis: a randomized, double-blind, placebo-controlled trial with Lactobacillus acidophilus and Bifidobacterium animalis subsp. lactis. Inflamm Bowel Dis 2006; 12:395–401.
- Calabrese C, Fabbri A, Areni A, Zahlane D, Scialpi C, Di Febo G. Mesalazine with or without cholestyramine in the treatment of microscopic colitis: randomized controlled trial. J Gastroenterol Hepatol 2007; 22:809–814.
- Wall GC, Schirmer LL, Page MJ. Pharmacotherapy for microscopic colitis. Pharmacotherapy 2007; 27:425–433.
- Feyen B, Wall GC, Finnerty EP, DeWitt JE, Reyes RS. Meta-analysis: budesonide treatment for collagenous colitis. Aliment Pharmacol Ther 2004; 20:745–749.
- Madisch A, Heymer P, Voss C, et al. Oral budesonide therapy improves quality of life in patients with collagenous colitis. Int J Colorectal Dis 2005; 20:312–316.
- Craig CR, editor. Modern Pharmacology With Clinical Application. 6th edition. Philadelphia: Lippincott Williams and Wilkins, 2003:481.
- Limsui D, Pardi DS, Camilleri M, et al. Symptomatic overlap between irritable bowel syndrome and microscopic colitis. Inflamm Bowel Dis 2007; 13:175–181.
A 68-year-old white woman with irritable bowel syndrome has had worsening symptoms of right-sided abdominal pain, excessive bloating, and loose stools. Her bowel movements have increased from one a day to two or three a day. She has not noted any mucus or blood in the stool. She cannot identify any alleviating or aggravating factors, and the pain is not related to eating.
She consumes a normal diet, including meat and dairy. Over-the-counter antidiarrheal medications do not relieve the symptoms. She has had no fevers, chills, or night sweats, and she has not lost weight over the past year.
Her medical history includes breast cancer (in remission), alcohol abuse (in remission), and hypothyroidism, osteoporosis, and supraventricular tachycardia, all controlled with treatment as noted below. She has never undergone abdominal surgery.
A general review of systems is normal. Her current medications include oxybutynin (available as Ditropan, others), calcium polycarbophil (FiberCon, others), risedronate (Actonel), levothyroxine (Synthroid, others), simethicone (Maalox Anti-Gas, others), atenolol (Tenormin), trazodone (Desyrel), a calcium supplement, and aspirin. She began taking duloxetine (Cymbalta) 18 months ago, and the dose was increased from 60 mg to 90 mg 1 week before this visit.
She has never smoked, and she has abstained from alcohol for 10 years. She has no family history of colon cancer, celiac disease, or inflammatory bowel disease. She has not traveled outside the country in the past several years, and she notes no change in her source of drinking water.
On physical examination, she does not appear to be in acute distress. Her pulse is 64 and her blood pressure is 112/78 mm Hg. The cardiopulmonary examination is normal. Her abdomen is soft, symmetrical, nondistended, and nontender. Bowel sounds are normal. No abdominal masses, palpable organomegaly, or abdominal bruits are noted.
Results of basic laboratory tests, including thyroid-stimulating hormone (TSH), complete blood count, blood chemistries, renal function, and liver function, are normal. Colonoscopy shows normal mucosa as far as the cecum.
DIFFERENTIAL DIAGNOSIS
1. In addition to irritable bowel syndrome, which of these can explain her symptoms?
- Ulcerative colitis
- Celiac disease
- Microscopic colitis
- Hyperthyroidism
- Lactase deficiency
Ulcerative colitis typically presents with blood and mucus in the stool and gross abnormalities on colonoscopy, none of which is present in this patient.
Hyperthyroidism can be ruled out by the normal TSH level.
Lactase deficiency or lactose intolerance is unlikely because it is present in only 15% of people of northern European descent (compared with 80% of blacks and Hispanics and up to 100% of Native Americans and Asians).1 Furthermore, her pain is apparently not related to consuming dairy products.
The hydrogen breath test can aid in the diagnosis of lactase deficiency. This test relies on the breakdown of malabsorbed lactose by colonic flora. This is the most widely used test for this deficiency, but its high false-negative rate of 25% means that a negative result does not exclude the diagnosis and should not be relied on in working up a patient with chronic diarrhea.2 Simply noting whether symptoms develop after ingesting 50 g of lactose is clinically useful when lactase deficiency is suspected.
Based on the information so far, it is reasonable in this patient to evaluate for celiac disease and for microscopic colitis.
Celiac disease, also called gluten-sensitive enteropathy, has a varied presentation that includes nonspecific symptoms such as those in this patient. Classically, it causes diarrhea, but patients may present with a single nutrient deficiency and no diarrhea.
This patient lacks the elevated alkaline phosphatase or evidence of vitamin deficiencies characteristic of malabsorption in celiac disease (ie, vitamins A, B12, D, K, and folate)3. She also lacks evidence of malnutrition, such as iron deficiency anemia, weight loss, or low serum albumin. Finally, she does not have the dermatitis herpetiformis rash to suggest autoimmune gluten-sensitive enteropathy, nor does she have evidence of follicular hyperplasia or petechiae due to vitamin malabsorption.3
Because no single serologic test is ideal for diagnosing gluten-sensitive enteropathy, several tests are typically used: immunoglobulin A (IgA) antigliadin antibody, IgG antigliadin antibody, IgA antitransglutaminase antibody, and IgA antiendomysial antibody. IgA antitransglutaminase antibody is 92% to 98% sensitive and 91% to 100% specific for celiac disease. IgG antigliadin antibody is 92% to 97% sensitive and 99% specific. The positive predictive value of the IgA and IgG antigliadin antibody tests is less than 2% in the general population, whereas the positive predictive value for antiendomysial antibody and antitransglutaminase antibody are 15.7% and 21.8%, respectively.4 A positive serologic test for antiendomysial antibody is nearly 100% specific.
Our patient’s entire celiac antibody panel is negative, and thus celiac disease is unlikely.
Case continued: Features of microscopic colitis
In our patient, colonic biopsy reveals a mildly expanded lamina propria, intraepithelial lymphocytes, and a patchy but prominent thickening of the subepithelial collagen table. This set of features is consistent with collagenous colitis, a variant of microscopic colitis. Histologic signs on biopsy specimens are fairly specific for the disease.5
Chronic, intermittent, secretory diarrhea without bleeding is the hallmark of microscopic colitis. Associated symptoms may include abdominal pain, weight loss, and fatigue. If biopsies are not taken at the time of the initial evaluation, and the colonic pathology is overlooked, patients with collagenous colitis may be diagnosed with irritable bowel syndrome with diarrhea.6 The sedimentation rate is often elevated, and the antinuclear antibody test can be positive.7 Steatorrhea or protein-losing enteropathy can occur, and fecal leukocytes are present in more than 50% of patients.8
This patient fits well the demographics of the typical collagenous colitis patient: ie, a middle-aged woman in her 6th decade in otherwise good general health. The female-to-male ratio is 15:1 overall, although the relative frequency of collagenous colitis in women is greater than that of lymphocytic colitis.9 In a population-based study, the incidence of collagenous colitis was 5.1 per 100,000 per year, with a prevalence of 36 per 100,000; the incidence of lymphocytic colitis was 9.8 per 100,000 per year, with a prevalence of 64 per 100,000.10
Symptoms are typically vague and range from an annoyance to more than 20 non-bloody stools per day. The course of the disease also varies. Case series have reported a spontaneous remission rate of 15% to 20%,11 though flare-ups are common. Microscopic colitis is largely a benign disease. It does not increase a person’s risk of colon cancer.
CAUSES OF COLLAGENOUS COLITIS
2. What causes of collagenous colitis have been identified?
- Alcohol abuse
- Previous gastrointestinal surgery
- Drug-induced injury to colon
Neither alcohol use nor previous gastrointestinal surgery has been associated with the development of collagenous colitis.
Collagenous colitis has, however, been linked to several causes. Abnormal collagen metabolism has been demonstrated in patients as a result of increased expression of procollagen I and metalloproteinase inhibitor TIMP-1.12 Bacterial toxins and a bile-acid malabsorption defect in the terminal ileum and subsequent exposure of the colon to high concentrations of bile acids have also been linked to the development of collagenous colitis.
Many drugs have been linked to the development of collagenous colitis. Damage to the large intestine related to the use of non-steroidal anti-inflammatory drugs has been attributed to the blockage of prostaglandin synthesis.13 Simvastatin (Zocor), lansoprazole (Prilosec), and ticlopidine (Ticlid) have been linked to collagenous colitis; ticlopidine, flutamide (Eulexin), gold salts, lansoprazole, and sertraline (Zoloft) have been linked to the development of lymphocytic colitis.14 In one small series, patients developed colitis after switching from omeprazole (Prevacid) to lansoprazole. All patients had their symptoms and biopsy findings resolve within 1 week of stopping the drug.15
WHICH DRUG IS BEST?
3. Which drug is best for microscopic colitis, based on the current evidence?
- Bismuth (eg, Kaopectate, Pepto-Bismol)
- Sulfasalazine (Sulfazine)
- Budesonide (Entocort)
- Prednisolone
Studies have evaluated bismuth subsalicylate, Boswellia serrata extract, probiotics, prednisolone, budesonide, and other drugs for treating collagenous colitis.16
Bismuth trials have been small. In an open-label study of bismuth,17 symptoms improved in 11 of 12 patients.
Prednisolone recipients had a trend towards clinical response with treatment vs placebo, but it was not statistically significant, and there was incomplete remission of disease.18
Boswellia serrataextract19 and probiotics20 showed no clinical improvement.
Cholestyramine has been shown to be helpful when used in conjunction with an anti-inflammatory agent,21 and it may be helpful when used alone.
Aminosalicylate compounds have not been tested in prospective randomized trials, even though they are the cornerstone of treatment for ulcerative colitis. Retrospective trials have been equivocal.22
Budesonide currently has the best evidence of efficacy in collagenous colitis,23,24 and some evidence suggests it is also effective for other variants of microscopic colitis.
A total of 94 patients were enrolled in three placebo-controlled trials of budesonide at 9 mg daily or on a tapering schedule for 6 to 8 weeks. The pooled odds ratio for clinical response to treatment with budesonide was 12.32 (95% confidence interval 5.53–27.46), with a number needed to treat of 1.58. Significant histologic improvement with treatment was noted in all three trials.23
Quality of life has also been studied in patients with microscopic colitis who take budesonide. Symptoms, emotional functioning, and physical functioning are improved. Budesonide also improved stool consistency and significantly reduced the mean stool frequency compared with placebo.24
Compared with cortisol, budesonide has a 200 times greater affinity for the glucocorticoid receptor, and a 1,000 times greater topical anti-inflammatory potency. It is also well absorbed in the gastrointestinal tract but is substantially modified into very weak metabolites as a result of first-pass metabolism in the liver.25 This localized effect further supports the use of budesonide in patients with any form of microscopic colitis.
Although studies have shown budesonide to be effective, not every patient with a histologic diagnosis of microscopic colitis needs it. It is reasonable to try antidiarrheal agents, bismuth, or both as a first step because they are inexpensive and have few side effects. If budesonide is used, it should be given for 6 to 8 weeks, then stopped, and the patient should then be monitored for symptom recurrence. If a flare does occur, budesonide can be restarted and continued as maintenance therapy.
KEY CONSIDERATIONS
Microscopic colitis is diagnosed histologically, while irritable bowel syndrome is a clinical diagnosis. In population-based cohorts of histologically confirmed microscopic colitis, 50% to 70% met symptom-based Rome criteria for the diagnosis of irritable bowel syndrome. The clinical symptom-based criteria for irritable bowel syndrome are not specific enough to rule out the diagnosis of microscopic colitis. Therefore, patients with suspected diarrhea-predominant irritable bowel syndrome should undergo colonoscopy with biopsy to investigate microscopic colitis if symptoms are not well controlled by antidiarrheal therapy.26 The patient’s management may be very different depending on whether colonoscopy is done.
Management of microscopic colitis should include stopping any drugs associated with it. Simple antidiarrheal agents should be tried first to manage symptoms. If symptoms persist, patients can be treated with budesonide (Entocort EC) 9 mg by mouth daily for 8 weeks to induce remission, or 6 mg by mouth daily for 3 months as maintenance therapy.
OUR PATIENT’S COURSE
Our patient’s medication list includes duloxetine, a serotonin-norepinephrine reuptake inhibitor related to drugs that have been associated with the development of microscopic colitis. We tapered the duloxetine, and her symptoms improved by 50%. Her symptoms were eventually controlled after an 8-week course of oral budesonide 9 mg and ongoing intermittent use of loperamide (Imodium).
A 68-year-old white woman with irritable bowel syndrome has had worsening symptoms of right-sided abdominal pain, excessive bloating, and loose stools. Her bowel movements have increased from one a day to two or three a day. She has not noted any mucus or blood in the stool. She cannot identify any alleviating or aggravating factors, and the pain is not related to eating.
She consumes a normal diet, including meat and dairy. Over-the-counter antidiarrheal medications do not relieve the symptoms. She has had no fevers, chills, or night sweats, and she has not lost weight over the past year.
Her medical history includes breast cancer (in remission), alcohol abuse (in remission), and hypothyroidism, osteoporosis, and supraventricular tachycardia, all controlled with treatment as noted below. She has never undergone abdominal surgery.
A general review of systems is normal. Her current medications include oxybutynin (available as Ditropan, others), calcium polycarbophil (FiberCon, others), risedronate (Actonel), levothyroxine (Synthroid, others), simethicone (Maalox Anti-Gas, others), atenolol (Tenormin), trazodone (Desyrel), a calcium supplement, and aspirin. She began taking duloxetine (Cymbalta) 18 months ago, and the dose was increased from 60 mg to 90 mg 1 week before this visit.
She has never smoked, and she has abstained from alcohol for 10 years. She has no family history of colon cancer, celiac disease, or inflammatory bowel disease. She has not traveled outside the country in the past several years, and she notes no change in her source of drinking water.
On physical examination, she does not appear to be in acute distress. Her pulse is 64 and her blood pressure is 112/78 mm Hg. The cardiopulmonary examination is normal. Her abdomen is soft, symmetrical, nondistended, and nontender. Bowel sounds are normal. No abdominal masses, palpable organomegaly, or abdominal bruits are noted.
Results of basic laboratory tests, including thyroid-stimulating hormone (TSH), complete blood count, blood chemistries, renal function, and liver function, are normal. Colonoscopy shows normal mucosa as far as the cecum.
DIFFERENTIAL DIAGNOSIS
1. In addition to irritable bowel syndrome, which of these can explain her symptoms?
- Ulcerative colitis
- Celiac disease
- Microscopic colitis
- Hyperthyroidism
- Lactase deficiency
Ulcerative colitis typically presents with blood and mucus in the stool and gross abnormalities on colonoscopy, none of which is present in this patient.
Hyperthyroidism can be ruled out by the normal TSH level.
Lactase deficiency or lactose intolerance is unlikely because it is present in only 15% of people of northern European descent (compared with 80% of blacks and Hispanics and up to 100% of Native Americans and Asians).1 Furthermore, her pain is apparently not related to consuming dairy products.
The hydrogen breath test can aid in the diagnosis of lactase deficiency. This test relies on the breakdown of malabsorbed lactose by colonic flora. This is the most widely used test for this deficiency, but its high false-negative rate of 25% means that a negative result does not exclude the diagnosis and should not be relied on in working up a patient with chronic diarrhea.2 Simply noting whether symptoms develop after ingesting 50 g of lactose is clinically useful when lactase deficiency is suspected.
Based on the information so far, it is reasonable in this patient to evaluate for celiac disease and for microscopic colitis.
Celiac disease, also called gluten-sensitive enteropathy, has a varied presentation that includes nonspecific symptoms such as those in this patient. Classically, it causes diarrhea, but patients may present with a single nutrient deficiency and no diarrhea.
This patient lacks the elevated alkaline phosphatase or evidence of vitamin deficiencies characteristic of malabsorption in celiac disease (ie, vitamins A, B12, D, K, and folate)3. She also lacks evidence of malnutrition, such as iron deficiency anemia, weight loss, or low serum albumin. Finally, she does not have the dermatitis herpetiformis rash to suggest autoimmune gluten-sensitive enteropathy, nor does she have evidence of follicular hyperplasia or petechiae due to vitamin malabsorption.3
Because no single serologic test is ideal for diagnosing gluten-sensitive enteropathy, several tests are typically used: immunoglobulin A (IgA) antigliadin antibody, IgG antigliadin antibody, IgA antitransglutaminase antibody, and IgA antiendomysial antibody. IgA antitransglutaminase antibody is 92% to 98% sensitive and 91% to 100% specific for celiac disease. IgG antigliadin antibody is 92% to 97% sensitive and 99% specific. The positive predictive value of the IgA and IgG antigliadin antibody tests is less than 2% in the general population, whereas the positive predictive value for antiendomysial antibody and antitransglutaminase antibody are 15.7% and 21.8%, respectively.4 A positive serologic test for antiendomysial antibody is nearly 100% specific.
Our patient’s entire celiac antibody panel is negative, and thus celiac disease is unlikely.
Case continued: Features of microscopic colitis
In our patient, colonic biopsy reveals a mildly expanded lamina propria, intraepithelial lymphocytes, and a patchy but prominent thickening of the subepithelial collagen table. This set of features is consistent with collagenous colitis, a variant of microscopic colitis. Histologic signs on biopsy specimens are fairly specific for the disease.5
Chronic, intermittent, secretory diarrhea without bleeding is the hallmark of microscopic colitis. Associated symptoms may include abdominal pain, weight loss, and fatigue. If biopsies are not taken at the time of the initial evaluation, and the colonic pathology is overlooked, patients with collagenous colitis may be diagnosed with irritable bowel syndrome with diarrhea.6 The sedimentation rate is often elevated, and the antinuclear antibody test can be positive.7 Steatorrhea or protein-losing enteropathy can occur, and fecal leukocytes are present in more than 50% of patients.8
This patient fits well the demographics of the typical collagenous colitis patient: ie, a middle-aged woman in her 6th decade in otherwise good general health. The female-to-male ratio is 15:1 overall, although the relative frequency of collagenous colitis in women is greater than that of lymphocytic colitis.9 In a population-based study, the incidence of collagenous colitis was 5.1 per 100,000 per year, with a prevalence of 36 per 100,000; the incidence of lymphocytic colitis was 9.8 per 100,000 per year, with a prevalence of 64 per 100,000.10
Symptoms are typically vague and range from an annoyance to more than 20 non-bloody stools per day. The course of the disease also varies. Case series have reported a spontaneous remission rate of 15% to 20%,11 though flare-ups are common. Microscopic colitis is largely a benign disease. It does not increase a person’s risk of colon cancer.
CAUSES OF COLLAGENOUS COLITIS
2. What causes of collagenous colitis have been identified?
- Alcohol abuse
- Previous gastrointestinal surgery
- Drug-induced injury to colon
Neither alcohol use nor previous gastrointestinal surgery has been associated with the development of collagenous colitis.
Collagenous colitis has, however, been linked to several causes. Abnormal collagen metabolism has been demonstrated in patients as a result of increased expression of procollagen I and metalloproteinase inhibitor TIMP-1.12 Bacterial toxins and a bile-acid malabsorption defect in the terminal ileum and subsequent exposure of the colon to high concentrations of bile acids have also been linked to the development of collagenous colitis.
Many drugs have been linked to the development of collagenous colitis. Damage to the large intestine related to the use of non-steroidal anti-inflammatory drugs has been attributed to the blockage of prostaglandin synthesis.13 Simvastatin (Zocor), lansoprazole (Prilosec), and ticlopidine (Ticlid) have been linked to collagenous colitis; ticlopidine, flutamide (Eulexin), gold salts, lansoprazole, and sertraline (Zoloft) have been linked to the development of lymphocytic colitis.14 In one small series, patients developed colitis after switching from omeprazole (Prevacid) to lansoprazole. All patients had their symptoms and biopsy findings resolve within 1 week of stopping the drug.15
WHICH DRUG IS BEST?
3. Which drug is best for microscopic colitis, based on the current evidence?
- Bismuth (eg, Kaopectate, Pepto-Bismol)
- Sulfasalazine (Sulfazine)
- Budesonide (Entocort)
- Prednisolone
Studies have evaluated bismuth subsalicylate, Boswellia serrata extract, probiotics, prednisolone, budesonide, and other drugs for treating collagenous colitis.16
Bismuth trials have been small. In an open-label study of bismuth,17 symptoms improved in 11 of 12 patients.
Prednisolone recipients had a trend towards clinical response with treatment vs placebo, but it was not statistically significant, and there was incomplete remission of disease.18
Boswellia serrataextract19 and probiotics20 showed no clinical improvement.
Cholestyramine has been shown to be helpful when used in conjunction with an anti-inflammatory agent,21 and it may be helpful when used alone.
Aminosalicylate compounds have not been tested in prospective randomized trials, even though they are the cornerstone of treatment for ulcerative colitis. Retrospective trials have been equivocal.22
Budesonide currently has the best evidence of efficacy in collagenous colitis,23,24 and some evidence suggests it is also effective for other variants of microscopic colitis.
A total of 94 patients were enrolled in three placebo-controlled trials of budesonide at 9 mg daily or on a tapering schedule for 6 to 8 weeks. The pooled odds ratio for clinical response to treatment with budesonide was 12.32 (95% confidence interval 5.53–27.46), with a number needed to treat of 1.58. Significant histologic improvement with treatment was noted in all three trials.23
Quality of life has also been studied in patients with microscopic colitis who take budesonide. Symptoms, emotional functioning, and physical functioning are improved. Budesonide also improved stool consistency and significantly reduced the mean stool frequency compared with placebo.24
Compared with cortisol, budesonide has a 200 times greater affinity for the glucocorticoid receptor, and a 1,000 times greater topical anti-inflammatory potency. It is also well absorbed in the gastrointestinal tract but is substantially modified into very weak metabolites as a result of first-pass metabolism in the liver.25 This localized effect further supports the use of budesonide in patients with any form of microscopic colitis.
Although studies have shown budesonide to be effective, not every patient with a histologic diagnosis of microscopic colitis needs it. It is reasonable to try antidiarrheal agents, bismuth, or both as a first step because they are inexpensive and have few side effects. If budesonide is used, it should be given for 6 to 8 weeks, then stopped, and the patient should then be monitored for symptom recurrence. If a flare does occur, budesonide can be restarted and continued as maintenance therapy.
KEY CONSIDERATIONS
Microscopic colitis is diagnosed histologically, while irritable bowel syndrome is a clinical diagnosis. In population-based cohorts of histologically confirmed microscopic colitis, 50% to 70% met symptom-based Rome criteria for the diagnosis of irritable bowel syndrome. The clinical symptom-based criteria for irritable bowel syndrome are not specific enough to rule out the diagnosis of microscopic colitis. Therefore, patients with suspected diarrhea-predominant irritable bowel syndrome should undergo colonoscopy with biopsy to investigate microscopic colitis if symptoms are not well controlled by antidiarrheal therapy.26 The patient’s management may be very different depending on whether colonoscopy is done.
Management of microscopic colitis should include stopping any drugs associated with it. Simple antidiarrheal agents should be tried first to manage symptoms. If symptoms persist, patients can be treated with budesonide (Entocort EC) 9 mg by mouth daily for 8 weeks to induce remission, or 6 mg by mouth daily for 3 months as maintenance therapy.
OUR PATIENT’S COURSE
Our patient’s medication list includes duloxetine, a serotonin-norepinephrine reuptake inhibitor related to drugs that have been associated with the development of microscopic colitis. We tapered the duloxetine, and her symptoms improved by 50%. Her symptoms were eventually controlled after an 8-week course of oral budesonide 9 mg and ongoing intermittent use of loperamide (Imodium).
- Swagerty DL, Walling AD, Klein RM. Lactose intolerance. Am Fam Physician 2002; 65:1845–1856.
- Thomas PD, Forbes A, Green J, et al. Guidelines for the investigation of chronic diarrhea, 2nd edition. Gut 2003; 52(suppl 5):1–5.
- Nelsen DA. Gluten-sensitive enteropathy (celiac disease): more common than you think. Am Fam Physician 2002; 66:2259–2266.
- Bardella MT, Trovato C, Cesana BM, Pagliari C, Gebbia C, Peracchi M. Serological markers for coeliac disease: is it time to change? Dig Liver Dis 2001; 33:426–431.
- Barta Z, Mekkel G, Csipo I, et al. Micropscopic colitis: a retrospective study of clinical presentation in 53 patients. World J Gastroenterol 2005; 11:1351–1355.
- Tremaine WJ. Diagnosing collagenous colitis: does it make a difference? Eur J Gastroenterol Hepatol 1999; 11:477–479.
- Bohr J, Tysk C, Yang P, Danielsson D, Järnerot G. Autoantibodies and immunoglobulins in collagenous colitis. Gut 1996; 39:77–81.
- Zins BJ, Tremaine WJ, Carpenter HA. Collagenous colitis: mucosal biopsies and association with fecal leukocytes. Mayo Clin Proc 1995; 70:430–433.
- Olsen M, Eriksson S, Bohr J, Järnerot G, Tysk C. Lymphocytic colitis: a retrospective clinical study of 199 Swedish patients. Gut 2004; 53:536–541.
- Pardi DS. Microscopic colitis: an update. Inflamm Bowel Dis 2004; 10:860–870.
- Fernandez-Banares F, Salas A, Esteve M, Espinos J, Forne M, Viver JM. Collagenous and lymphocytic colitis: evaluation of clinical and histological features, response to treatment, and long-term follow-up. Am J Gastroenterol 2003; 98:340–347.
- Aignet T, Neureiter D, Müller S, Küspert G, Belke J, Kirchner T. Extracellular matrix composition and gene expression in collagenous colitis. Gastroenterology 1997; 113:136–143.
- Parfitt JR, Driman DK. Pathological effects of drugs on the gastrointestinal tract: a review. Hum Pathol 2007; 38:527–536.
- Fernández-Bañares F, Esteve M, Espinós JC, et al. Drug consumption and the risk of microscopic colitis. Am J Gastroenterol 2007; 102:324–330.
- Thomson RD, Lestine LS, Bensen SP, et al. Lansoprazole-associated microscopic colitis: a case series. Am J Gastroenterol 2002; 97:2908–2913.
- Chande N, McDonald JWD, MacDonald JK. Cochrane Inflammatory Bowel Disease and Functional Bowel Disorders Group. Interventions for treating collagenous colitis. Cochrane Database Syst Rev 2007 Jan 24;(1):CD006096.
- Fine K, Lee E. Efficacy of open-label bismuth subsalicylate for the treatment of microscopic colitis. Gastroenterology 1998; 114:29–36.
- Munck LK, Kjeldsen J, Philipsen E, Fscher Hansen B. Incomplete remission with short-term prednisolone treatment in collagenous colitis: a randomized study. Scand J Gastroenterol 2003; 38:606–610.
- Madisch A, Miehlke S, Eichele E, et al. Boswellia serrata extract for the treatment of collagenous colitis: a randomized, double-blind, placebo-controlled, multicenter trial. Int J Colorectal Dis 2007; 22:1445–1451.
- Wildt S, Munck LK, Vinter-Jensen L, et al. Probiotic treatment of collagenous colitis: a randomized, double-blind, placebo-controlled trial with Lactobacillus acidophilus and Bifidobacterium animalis subsp. lactis. Inflamm Bowel Dis 2006; 12:395–401.
- Calabrese C, Fabbri A, Areni A, Zahlane D, Scialpi C, Di Febo G. Mesalazine with or without cholestyramine in the treatment of microscopic colitis: randomized controlled trial. J Gastroenterol Hepatol 2007; 22:809–814.
- Wall GC, Schirmer LL, Page MJ. Pharmacotherapy for microscopic colitis. Pharmacotherapy 2007; 27:425–433.
- Feyen B, Wall GC, Finnerty EP, DeWitt JE, Reyes RS. Meta-analysis: budesonide treatment for collagenous colitis. Aliment Pharmacol Ther 2004; 20:745–749.
- Madisch A, Heymer P, Voss C, et al. Oral budesonide therapy improves quality of life in patients with collagenous colitis. Int J Colorectal Dis 2005; 20:312–316.
- Craig CR, editor. Modern Pharmacology With Clinical Application. 6th edition. Philadelphia: Lippincott Williams and Wilkins, 2003:481.
- Limsui D, Pardi DS, Camilleri M, et al. Symptomatic overlap between irritable bowel syndrome and microscopic colitis. Inflamm Bowel Dis 2007; 13:175–181.
- Swagerty DL, Walling AD, Klein RM. Lactose intolerance. Am Fam Physician 2002; 65:1845–1856.
- Thomas PD, Forbes A, Green J, et al. Guidelines for the investigation of chronic diarrhea, 2nd edition. Gut 2003; 52(suppl 5):1–5.
- Nelsen DA. Gluten-sensitive enteropathy (celiac disease): more common than you think. Am Fam Physician 2002; 66:2259–2266.
- Bardella MT, Trovato C, Cesana BM, Pagliari C, Gebbia C, Peracchi M. Serological markers for coeliac disease: is it time to change? Dig Liver Dis 2001; 33:426–431.
- Barta Z, Mekkel G, Csipo I, et al. Micropscopic colitis: a retrospective study of clinical presentation in 53 patients. World J Gastroenterol 2005; 11:1351–1355.
- Tremaine WJ. Diagnosing collagenous colitis: does it make a difference? Eur J Gastroenterol Hepatol 1999; 11:477–479.
- Bohr J, Tysk C, Yang P, Danielsson D, Järnerot G. Autoantibodies and immunoglobulins in collagenous colitis. Gut 1996; 39:77–81.
- Zins BJ, Tremaine WJ, Carpenter HA. Collagenous colitis: mucosal biopsies and association with fecal leukocytes. Mayo Clin Proc 1995; 70:430–433.
- Olsen M, Eriksson S, Bohr J, Järnerot G, Tysk C. Lymphocytic colitis: a retrospective clinical study of 199 Swedish patients. Gut 2004; 53:536–541.
- Pardi DS. Microscopic colitis: an update. Inflamm Bowel Dis 2004; 10:860–870.
- Fernandez-Banares F, Salas A, Esteve M, Espinos J, Forne M, Viver JM. Collagenous and lymphocytic colitis: evaluation of clinical and histological features, response to treatment, and long-term follow-up. Am J Gastroenterol 2003; 98:340–347.
- Aignet T, Neureiter D, Müller S, Küspert G, Belke J, Kirchner T. Extracellular matrix composition and gene expression in collagenous colitis. Gastroenterology 1997; 113:136–143.
- Parfitt JR, Driman DK. Pathological effects of drugs on the gastrointestinal tract: a review. Hum Pathol 2007; 38:527–536.
- Fernández-Bañares F, Esteve M, Espinós JC, et al. Drug consumption and the risk of microscopic colitis. Am J Gastroenterol 2007; 102:324–330.
- Thomson RD, Lestine LS, Bensen SP, et al. Lansoprazole-associated microscopic colitis: a case series. Am J Gastroenterol 2002; 97:2908–2913.
- Chande N, McDonald JWD, MacDonald JK. Cochrane Inflammatory Bowel Disease and Functional Bowel Disorders Group. Interventions for treating collagenous colitis. Cochrane Database Syst Rev 2007 Jan 24;(1):CD006096.
- Fine K, Lee E. Efficacy of open-label bismuth subsalicylate for the treatment of microscopic colitis. Gastroenterology 1998; 114:29–36.
- Munck LK, Kjeldsen J, Philipsen E, Fscher Hansen B. Incomplete remission with short-term prednisolone treatment in collagenous colitis: a randomized study. Scand J Gastroenterol 2003; 38:606–610.
- Madisch A, Miehlke S, Eichele E, et al. Boswellia serrata extract for the treatment of collagenous colitis: a randomized, double-blind, placebo-controlled, multicenter trial. Int J Colorectal Dis 2007; 22:1445–1451.
- Wildt S, Munck LK, Vinter-Jensen L, et al. Probiotic treatment of collagenous colitis: a randomized, double-blind, placebo-controlled trial with Lactobacillus acidophilus and Bifidobacterium animalis subsp. lactis. Inflamm Bowel Dis 2006; 12:395–401.
- Calabrese C, Fabbri A, Areni A, Zahlane D, Scialpi C, Di Febo G. Mesalazine with or without cholestyramine in the treatment of microscopic colitis: randomized controlled trial. J Gastroenterol Hepatol 2007; 22:809–814.
- Wall GC, Schirmer LL, Page MJ. Pharmacotherapy for microscopic colitis. Pharmacotherapy 2007; 27:425–433.
- Feyen B, Wall GC, Finnerty EP, DeWitt JE, Reyes RS. Meta-analysis: budesonide treatment for collagenous colitis. Aliment Pharmacol Ther 2004; 20:745–749.
- Madisch A, Heymer P, Voss C, et al. Oral budesonide therapy improves quality of life in patients with collagenous colitis. Int J Colorectal Dis 2005; 20:312–316.
- Craig CR, editor. Modern Pharmacology With Clinical Application. 6th edition. Philadelphia: Lippincott Williams and Wilkins, 2003:481.
- Limsui D, Pardi DS, Camilleri M, et al. Symptomatic overlap between irritable bowel syndrome and microscopic colitis. Inflamm Bowel Dis 2007; 13:175–181.
A medical center is not a hospital
Editor’s note: We are interested in your thoughts on this article. See the Editor-in-Chief’s comments.
I use to be a hospital guy. I was only a few days into my third-year medicine clerkship in medical school nearly three decades ago when I fell in love with the hospital and knew I was going to be an internist. The hospital wasn’t called a medical center back then. It was a fascinating and magical place, where internists were fired in the furnace of rounds, night call, and morning report. I loved the association with the great case, the flush of excitement that accompanied the difficult diagnosis, the hard-earned annual promotion through the hierarchy of trainees seeking the rarefied air of the attending physicians. We bonded as fellow house officers more tightly than with friends outside the hospital. We prowled the wards, intensive care units, emergency room, and laboratories and never slept. The hospital was the most exclusive of clubs, and our training granted us lifelong membership.
A humming beehive of academic activity, the hospital was also a web of powerful social relationships. Everybody knew everybody, from the hospital CEO to the night security officer. The nurses called you by your first name and worked with you for weeks at a time, fostering mutual respect and sometimes even affection. In those days, nurses actually nursed their patients, spoon-feeding them broth with their medications, washing them in bed and bathroom, holding their hands and heads. Patients came to the hospital to be diagnosed and treated until they recovered from whatever illness had felled them. They stayed long enough so that you knew them and their families as well as you knew your own.
I have been a general internist and clinician-educator for 23 years, working in two university hospitals and one community hospital. That’s more than seven generations of house staff with whom I’ve toiled and learned. Somewhere along the way, I became increasingly aware that teaching clinical medicine to students, interns, and residents was getting harder and harder. The patients were sicker and stayed only 3.2 days in the hospital. What we were teaching wasn’t how to diagnose and treat diseases, but how to manage only their most serious complications—the respiratory distress from pneumonia, the ketosis of uncontrolled diabetes, the septic shock from infections. The wards became intensive care units, and the critical care units the province of “intensivists” who were more adept than we were at taming all the machinery and technology. We struggled to keep up with the unending deluge of arcane demands from the accreditation organizations watchdogging our teaching efforts. We pretended that we somehow distinguished teaching rounds from working rounds, and documented the silliness in computer files. Medical education slowly slipped from being a calling to folks like me, finally succumbing to bureaucratic lunacy. The pace of teaching and caring for acutely ill patients became intolerable. Rounds went from the bedside to the classroom to the cell phone. The house staff were getting cheated out of the whole point of residency—the miracle of turning medical students into attending physicians in a little over a thousand days.
Worse, though, was the ebbing of the lifeblood of the hospital. Now the medical center, riddled with “centers of excellence” instead of departments, answered only to administrators who cared nothing about medical education, except for the Medicare dollars they would lose if they cut the training programs. They spent enormous amounts of money marketing the centers of excellence, and they cut everything else to manipulate the bottom line.
The biggest casualty, of course, was the nursing staff. Underpaid, depleted of leadership and morale, they simply disappeared. They were replaced by agency nurses who worked their shifts and didn’t know the doctors or the patients. The complex bedside care of increasingly sick, old, and vulnerable patients was delegated to people with high school equivalency degrees. Nurses now cared for their patients by managing their own support staff, and spent much of their time entering useless information in the computer. The doctor-nurse collaboration I grew up with as a trainee and young attending didn’t exist any-more, and patients suffered as a result.
In 2000, the Institute of Medicine informed the public and the medical community that being a patient in an American hospital was dangerous.1 We were told that at least 44,000 and perhaps as many as 98,000 patients die annually in US hospitals as the result of preventable medical mishaps, more deaths than are attributable yearly to motor vehicle accidents, breast cancer, or AIDS.1 Although there has been an emerging body of literature pertaining to this epidemic, not much has changed, at least not in my hospital. We remain absurdly complacent about rising iatrogenic infection rates, knowing all too well that we are allowing immunocompromised patients to die unnecessarily in our intensive care units. There are alcohol-based hand-washing gels everywhere, but no police or policy with teeth in it to enforce handwashing. We lurch toward physician computer order entry, clinging to the false belief that software programs will prevent adverse drug reactions and delivery of the wrong dangerous drug to the wrong patient. We understaff our pharmacies so that they can’t get the medications to the patients on time or alert us to our own prescribing errors. We burn out our nurses despite years of loyal service. And worst of all, we capitulate to the for-profit insurance industry that informs us they won’t pay for day 4 of Mr. Jones’ hospitalization because he has failed to meet some arbitrary criteria in their manual.
I stepped down as chairman of my department 3 years ago because I couldn’t stand it any longer. I couldn’t stand the management retreats in which we obsessed about “customer service” while the waiting time in the emergency department ballooned to 12 hours because there were “no beds.” There were plenty of beds, but no nurses to staff them. I was marginalized when I protested the budget cycles bleeding out support of medical education in favor of the annual purchase of new scanners and surgical gizmos. I couldn’t get anybody fired up about patient safety.
Retreating to the privacy of clinical medicine, I realized the other day that my real job is not to diagnose, treat, and teach about diseases anymore. My real job is to do everything in my power to keep my patients out of the medical center. I walk the halls now and don’t recognize the institution I grew up in and came to love. Everywhere I look, I see not magic and promise, but dirt and danger.
I’m not a hospital guy anymore.
- Kohn LT, Corrigan JM, Donaldson MS, editors. To Err is Human: Building a Safer Health System. Washington, DC: National Academy Press, 2000.
Editor’s note: We are interested in your thoughts on this article. See the Editor-in-Chief’s comments.
I use to be a hospital guy. I was only a few days into my third-year medicine clerkship in medical school nearly three decades ago when I fell in love with the hospital and knew I was going to be an internist. The hospital wasn’t called a medical center back then. It was a fascinating and magical place, where internists were fired in the furnace of rounds, night call, and morning report. I loved the association with the great case, the flush of excitement that accompanied the difficult diagnosis, the hard-earned annual promotion through the hierarchy of trainees seeking the rarefied air of the attending physicians. We bonded as fellow house officers more tightly than with friends outside the hospital. We prowled the wards, intensive care units, emergency room, and laboratories and never slept. The hospital was the most exclusive of clubs, and our training granted us lifelong membership.
A humming beehive of academic activity, the hospital was also a web of powerful social relationships. Everybody knew everybody, from the hospital CEO to the night security officer. The nurses called you by your first name and worked with you for weeks at a time, fostering mutual respect and sometimes even affection. In those days, nurses actually nursed their patients, spoon-feeding them broth with their medications, washing them in bed and bathroom, holding their hands and heads. Patients came to the hospital to be diagnosed and treated until they recovered from whatever illness had felled them. They stayed long enough so that you knew them and their families as well as you knew your own.
I have been a general internist and clinician-educator for 23 years, working in two university hospitals and one community hospital. That’s more than seven generations of house staff with whom I’ve toiled and learned. Somewhere along the way, I became increasingly aware that teaching clinical medicine to students, interns, and residents was getting harder and harder. The patients were sicker and stayed only 3.2 days in the hospital. What we were teaching wasn’t how to diagnose and treat diseases, but how to manage only their most serious complications—the respiratory distress from pneumonia, the ketosis of uncontrolled diabetes, the septic shock from infections. The wards became intensive care units, and the critical care units the province of “intensivists” who were more adept than we were at taming all the machinery and technology. We struggled to keep up with the unending deluge of arcane demands from the accreditation organizations watchdogging our teaching efforts. We pretended that we somehow distinguished teaching rounds from working rounds, and documented the silliness in computer files. Medical education slowly slipped from being a calling to folks like me, finally succumbing to bureaucratic lunacy. The pace of teaching and caring for acutely ill patients became intolerable. Rounds went from the bedside to the classroom to the cell phone. The house staff were getting cheated out of the whole point of residency—the miracle of turning medical students into attending physicians in a little over a thousand days.
Worse, though, was the ebbing of the lifeblood of the hospital. Now the medical center, riddled with “centers of excellence” instead of departments, answered only to administrators who cared nothing about medical education, except for the Medicare dollars they would lose if they cut the training programs. They spent enormous amounts of money marketing the centers of excellence, and they cut everything else to manipulate the bottom line.
The biggest casualty, of course, was the nursing staff. Underpaid, depleted of leadership and morale, they simply disappeared. They were replaced by agency nurses who worked their shifts and didn’t know the doctors or the patients. The complex bedside care of increasingly sick, old, and vulnerable patients was delegated to people with high school equivalency degrees. Nurses now cared for their patients by managing their own support staff, and spent much of their time entering useless information in the computer. The doctor-nurse collaboration I grew up with as a trainee and young attending didn’t exist any-more, and patients suffered as a result.
In 2000, the Institute of Medicine informed the public and the medical community that being a patient in an American hospital was dangerous.1 We were told that at least 44,000 and perhaps as many as 98,000 patients die annually in US hospitals as the result of preventable medical mishaps, more deaths than are attributable yearly to motor vehicle accidents, breast cancer, or AIDS.1 Although there has been an emerging body of literature pertaining to this epidemic, not much has changed, at least not in my hospital. We remain absurdly complacent about rising iatrogenic infection rates, knowing all too well that we are allowing immunocompromised patients to die unnecessarily in our intensive care units. There are alcohol-based hand-washing gels everywhere, but no police or policy with teeth in it to enforce handwashing. We lurch toward physician computer order entry, clinging to the false belief that software programs will prevent adverse drug reactions and delivery of the wrong dangerous drug to the wrong patient. We understaff our pharmacies so that they can’t get the medications to the patients on time or alert us to our own prescribing errors. We burn out our nurses despite years of loyal service. And worst of all, we capitulate to the for-profit insurance industry that informs us they won’t pay for day 4 of Mr. Jones’ hospitalization because he has failed to meet some arbitrary criteria in their manual.
I stepped down as chairman of my department 3 years ago because I couldn’t stand it any longer. I couldn’t stand the management retreats in which we obsessed about “customer service” while the waiting time in the emergency department ballooned to 12 hours because there were “no beds.” There were plenty of beds, but no nurses to staff them. I was marginalized when I protested the budget cycles bleeding out support of medical education in favor of the annual purchase of new scanners and surgical gizmos. I couldn’t get anybody fired up about patient safety.
Retreating to the privacy of clinical medicine, I realized the other day that my real job is not to diagnose, treat, and teach about diseases anymore. My real job is to do everything in my power to keep my patients out of the medical center. I walk the halls now and don’t recognize the institution I grew up in and came to love. Everywhere I look, I see not magic and promise, but dirt and danger.
I’m not a hospital guy anymore.
Editor’s note: We are interested in your thoughts on this article. See the Editor-in-Chief’s comments.
I use to be a hospital guy. I was only a few days into my third-year medicine clerkship in medical school nearly three decades ago when I fell in love with the hospital and knew I was going to be an internist. The hospital wasn’t called a medical center back then. It was a fascinating and magical place, where internists were fired in the furnace of rounds, night call, and morning report. I loved the association with the great case, the flush of excitement that accompanied the difficult diagnosis, the hard-earned annual promotion through the hierarchy of trainees seeking the rarefied air of the attending physicians. We bonded as fellow house officers more tightly than with friends outside the hospital. We prowled the wards, intensive care units, emergency room, and laboratories and never slept. The hospital was the most exclusive of clubs, and our training granted us lifelong membership.
A humming beehive of academic activity, the hospital was also a web of powerful social relationships. Everybody knew everybody, from the hospital CEO to the night security officer. The nurses called you by your first name and worked with you for weeks at a time, fostering mutual respect and sometimes even affection. In those days, nurses actually nursed their patients, spoon-feeding them broth with their medications, washing them in bed and bathroom, holding their hands and heads. Patients came to the hospital to be diagnosed and treated until they recovered from whatever illness had felled them. They stayed long enough so that you knew them and their families as well as you knew your own.
I have been a general internist and clinician-educator for 23 years, working in two university hospitals and one community hospital. That’s more than seven generations of house staff with whom I’ve toiled and learned. Somewhere along the way, I became increasingly aware that teaching clinical medicine to students, interns, and residents was getting harder and harder. The patients were sicker and stayed only 3.2 days in the hospital. What we were teaching wasn’t how to diagnose and treat diseases, but how to manage only their most serious complications—the respiratory distress from pneumonia, the ketosis of uncontrolled diabetes, the septic shock from infections. The wards became intensive care units, and the critical care units the province of “intensivists” who were more adept than we were at taming all the machinery and technology. We struggled to keep up with the unending deluge of arcane demands from the accreditation organizations watchdogging our teaching efforts. We pretended that we somehow distinguished teaching rounds from working rounds, and documented the silliness in computer files. Medical education slowly slipped from being a calling to folks like me, finally succumbing to bureaucratic lunacy. The pace of teaching and caring for acutely ill patients became intolerable. Rounds went from the bedside to the classroom to the cell phone. The house staff were getting cheated out of the whole point of residency—the miracle of turning medical students into attending physicians in a little over a thousand days.
Worse, though, was the ebbing of the lifeblood of the hospital. Now the medical center, riddled with “centers of excellence” instead of departments, answered only to administrators who cared nothing about medical education, except for the Medicare dollars they would lose if they cut the training programs. They spent enormous amounts of money marketing the centers of excellence, and they cut everything else to manipulate the bottom line.
The biggest casualty, of course, was the nursing staff. Underpaid, depleted of leadership and morale, they simply disappeared. They were replaced by agency nurses who worked their shifts and didn’t know the doctors or the patients. The complex bedside care of increasingly sick, old, and vulnerable patients was delegated to people with high school equivalency degrees. Nurses now cared for their patients by managing their own support staff, and spent much of their time entering useless information in the computer. The doctor-nurse collaboration I grew up with as a trainee and young attending didn’t exist any-more, and patients suffered as a result.
In 2000, the Institute of Medicine informed the public and the medical community that being a patient in an American hospital was dangerous.1 We were told that at least 44,000 and perhaps as many as 98,000 patients die annually in US hospitals as the result of preventable medical mishaps, more deaths than are attributable yearly to motor vehicle accidents, breast cancer, or AIDS.1 Although there has been an emerging body of literature pertaining to this epidemic, not much has changed, at least not in my hospital. We remain absurdly complacent about rising iatrogenic infection rates, knowing all too well that we are allowing immunocompromised patients to die unnecessarily in our intensive care units. There are alcohol-based hand-washing gels everywhere, but no police or policy with teeth in it to enforce handwashing. We lurch toward physician computer order entry, clinging to the false belief that software programs will prevent adverse drug reactions and delivery of the wrong dangerous drug to the wrong patient. We understaff our pharmacies so that they can’t get the medications to the patients on time or alert us to our own prescribing errors. We burn out our nurses despite years of loyal service. And worst of all, we capitulate to the for-profit insurance industry that informs us they won’t pay for day 4 of Mr. Jones’ hospitalization because he has failed to meet some arbitrary criteria in their manual.
I stepped down as chairman of my department 3 years ago because I couldn’t stand it any longer. I couldn’t stand the management retreats in which we obsessed about “customer service” while the waiting time in the emergency department ballooned to 12 hours because there were “no beds.” There were plenty of beds, but no nurses to staff them. I was marginalized when I protested the budget cycles bleeding out support of medical education in favor of the annual purchase of new scanners and surgical gizmos. I couldn’t get anybody fired up about patient safety.
Retreating to the privacy of clinical medicine, I realized the other day that my real job is not to diagnose, treat, and teach about diseases anymore. My real job is to do everything in my power to keep my patients out of the medical center. I walk the halls now and don’t recognize the institution I grew up in and came to love. Everywhere I look, I see not magic and promise, but dirt and danger.
I’m not a hospital guy anymore.
- Kohn LT, Corrigan JM, Donaldson MS, editors. To Err is Human: Building a Safer Health System. Washington, DC: National Academy Press, 2000.
- Kohn LT, Corrigan JM, Donaldson MS, editors. To Err is Human: Building a Safer Health System. Washington, DC: National Academy Press, 2000.
Autism Gene Mutations May Have Shared Mechanism
Seemingly diverse autism mutations may share the same underlying mechanism, according to Eric M. Morrow, MD, PhD, Instructor in Psychiatry at Harvard Medical School in Boston, and colleagues.
“The regulation of expression of some autism candidate genes by neuronal membrane depolarization suggests the appealing hypothesis that neural activity–dependent regulation of synapse development may be a mechanism common to several autism mutations,” stated Dr. Morrow and colleagues in the July 11 Science. “Therefore, disruption of activity-regulated synaptic development may be one mechanism common to at least a subset of seemingly heterogeneous autism-associated mutations.”
The researchers studied 88 large families—in which both parents shared recent ancestors—to find inherited causes of autism spectrum disorders. The families, who came from Jordan, Saudi Arabia, Kuwait, Oman, Pakistan, Qatar, Turkey, and the United Arab Emirates, were selected to emphasize the role of inherited genetic mutations.
The investigators gathered data with use of homozygosity mapping and compared the DNA of family members with and without autism to identify recessive mutations. Most individuals exhibited different genetic causes with little overlap between families, but a few exceptions were observed.
“Although the large size of linked loci precluded systemic gene sequencing in most cases, we were surprised to see that several consanguineous pedigrees showed large, rare, inherited homozygous deletions within linked regions, some of which are very likely causative mutations,” Dr. Morrow and colleagues said. Specifically, such deletions linked to autism were found in five families, or 6.4% of the study sample. Family members with one remaining functional copy of their genome segments did not have autism, while those with both copies missing did have autism.
The investigators found six gene disruptions that contributed to autism spectrum disorders. The largest of these gene disruptions involved genes essential for learning in the brain, as they are regulated either directly or indirectly by neuronal activity triggered by experience. Not all the genes were deleted—the remaining genes were simply turned off, leaving room for therapies to possibly turn these genes back on.
“Early brain development is driven largely by intrinsic patterns of gene expression that do not depend on experience-driven synaptic activity,” the investigators stated. “In contrast, postnatal brain development requires input from the environment that triggers the release of neurotransmitter and promotes critical aspects of synaptic maturation.... The connection between experience-dependent neural activity and gene expression in the postnatal period forms the basis of learning and memory, and autism symptoms typically emerge during these later stages of development.”
The researchers’ findings also pointed to potential genetic similarities, such as inherited recessive causes, between autism and other neurologic disorders. Among families in the present study who had one member with autism, there was a relatively equivalent male/female ratio compared with other families with autism, indicating that parents who shared a common recent ancestor was a determining factor. In families with more than one member with autism, the male/female ratio was even more balanced.
“The accumulating number of distinct, individually rare genetic causes in autism suggests that the genetic architecture of autism resembles that of mental retardation and epilepsy, with many syndromes, each individually rare, as well as other cases potentially reflecting complex interactions between inherited changes,” Dr. Morrow and colleagues elaborated. “The relatively reduced male/female ratio of affected children and the reduced rate of linked de novo copy number variants in the consanguineous sample [of the present study] ... both suggest that consanguineous pedigrees with autism are enriched for autosomal recessive causes similar to other congenital neurological disorders in consanguineous populations.”
The research team stated that although their findings support recent studies that suggest autism is highly heterogeneous genetically, homozygosity mapping appears to be an effective way to find underlying shared mechanisms. Understanding these genetic underpinnings could eventually help direct various therapies for the different clinical manifestations of autism.
“Our finding that deletions of genes regulated by neuronal activity or regions potentially involved in regulation of gene expression in autism suggests that defects in activity-dependent gene expression may be a cause of cognitive deficits in patients with autism,” the researchers concluded. In addition, their “data implicating noncoding elements in patients with shared ancestry, as well as the heterozygous nonsense changes in patients without shared ancestry, suggest that loss of proper regulation of gene dosage may be an important genetic mechanism in autism.”
Suggested Reading
Morrow EM, Yoo SY, Flavell SW, et al. Identifying autism loci and genes by tracing recent shared ancestry. Science. 2008;321(5886):218-223.
Sutcliffe JS. Genetics: insights into the pathogenesis of autism. Science. 2008;321(5886):208-209.
Seemingly diverse autism mutations may share the same underlying mechanism, according to Eric M. Morrow, MD, PhD, Instructor in Psychiatry at Harvard Medical School in Boston, and colleagues.
“The regulation of expression of some autism candidate genes by neuronal membrane depolarization suggests the appealing hypothesis that neural activity–dependent regulation of synapse development may be a mechanism common to several autism mutations,” stated Dr. Morrow and colleagues in the July 11 Science. “Therefore, disruption of activity-regulated synaptic development may be one mechanism common to at least a subset of seemingly heterogeneous autism-associated mutations.”
The researchers studied 88 large families—in which both parents shared recent ancestors—to find inherited causes of autism spectrum disorders. The families, who came from Jordan, Saudi Arabia, Kuwait, Oman, Pakistan, Qatar, Turkey, and the United Arab Emirates, were selected to emphasize the role of inherited genetic mutations.
The investigators gathered data with use of homozygosity mapping and compared the DNA of family members with and without autism to identify recessive mutations. Most individuals exhibited different genetic causes with little overlap between families, but a few exceptions were observed.
“Although the large size of linked loci precluded systemic gene sequencing in most cases, we were surprised to see that several consanguineous pedigrees showed large, rare, inherited homozygous deletions within linked regions, some of which are very likely causative mutations,” Dr. Morrow and colleagues said. Specifically, such deletions linked to autism were found in five families, or 6.4% of the study sample. Family members with one remaining functional copy of their genome segments did not have autism, while those with both copies missing did have autism.
The investigators found six gene disruptions that contributed to autism spectrum disorders. The largest of these gene disruptions involved genes essential for learning in the brain, as they are regulated either directly or indirectly by neuronal activity triggered by experience. Not all the genes were deleted—the remaining genes were simply turned off, leaving room for therapies to possibly turn these genes back on.
“Early brain development is driven largely by intrinsic patterns of gene expression that do not depend on experience-driven synaptic activity,” the investigators stated. “In contrast, postnatal brain development requires input from the environment that triggers the release of neurotransmitter and promotes critical aspects of synaptic maturation.... The connection between experience-dependent neural activity and gene expression in the postnatal period forms the basis of learning and memory, and autism symptoms typically emerge during these later stages of development.”
The researchers’ findings also pointed to potential genetic similarities, such as inherited recessive causes, between autism and other neurologic disorders. Among families in the present study who had one member with autism, there was a relatively equivalent male/female ratio compared with other families with autism, indicating that parents who shared a common recent ancestor was a determining factor. In families with more than one member with autism, the male/female ratio was even more balanced.
“The accumulating number of distinct, individually rare genetic causes in autism suggests that the genetic architecture of autism resembles that of mental retardation and epilepsy, with many syndromes, each individually rare, as well as other cases potentially reflecting complex interactions between inherited changes,” Dr. Morrow and colleagues elaborated. “The relatively reduced male/female ratio of affected children and the reduced rate of linked de novo copy number variants in the consanguineous sample [of the present study] ... both suggest that consanguineous pedigrees with autism are enriched for autosomal recessive causes similar to other congenital neurological disorders in consanguineous populations.”
The research team stated that although their findings support recent studies that suggest autism is highly heterogeneous genetically, homozygosity mapping appears to be an effective way to find underlying shared mechanisms. Understanding these genetic underpinnings could eventually help direct various therapies for the different clinical manifestations of autism.
“Our finding that deletions of genes regulated by neuronal activity or regions potentially involved in regulation of gene expression in autism suggests that defects in activity-dependent gene expression may be a cause of cognitive deficits in patients with autism,” the researchers concluded. In addition, their “data implicating noncoding elements in patients with shared ancestry, as well as the heterozygous nonsense changes in patients without shared ancestry, suggest that loss of proper regulation of gene dosage may be an important genetic mechanism in autism.”
Seemingly diverse autism mutations may share the same underlying mechanism, according to Eric M. Morrow, MD, PhD, Instructor in Psychiatry at Harvard Medical School in Boston, and colleagues.
“The regulation of expression of some autism candidate genes by neuronal membrane depolarization suggests the appealing hypothesis that neural activity–dependent regulation of synapse development may be a mechanism common to several autism mutations,” stated Dr. Morrow and colleagues in the July 11 Science. “Therefore, disruption of activity-regulated synaptic development may be one mechanism common to at least a subset of seemingly heterogeneous autism-associated mutations.”
The researchers studied 88 large families—in which both parents shared recent ancestors—to find inherited causes of autism spectrum disorders. The families, who came from Jordan, Saudi Arabia, Kuwait, Oman, Pakistan, Qatar, Turkey, and the United Arab Emirates, were selected to emphasize the role of inherited genetic mutations.
The investigators gathered data with use of homozygosity mapping and compared the DNA of family members with and without autism to identify recessive mutations. Most individuals exhibited different genetic causes with little overlap between families, but a few exceptions were observed.
“Although the large size of linked loci precluded systemic gene sequencing in most cases, we were surprised to see that several consanguineous pedigrees showed large, rare, inherited homozygous deletions within linked regions, some of which are very likely causative mutations,” Dr. Morrow and colleagues said. Specifically, such deletions linked to autism were found in five families, or 6.4% of the study sample. Family members with one remaining functional copy of their genome segments did not have autism, while those with both copies missing did have autism.
The investigators found six gene disruptions that contributed to autism spectrum disorders. The largest of these gene disruptions involved genes essential for learning in the brain, as they are regulated either directly or indirectly by neuronal activity triggered by experience. Not all the genes were deleted—the remaining genes were simply turned off, leaving room for therapies to possibly turn these genes back on.
“Early brain development is driven largely by intrinsic patterns of gene expression that do not depend on experience-driven synaptic activity,” the investigators stated. “In contrast, postnatal brain development requires input from the environment that triggers the release of neurotransmitter and promotes critical aspects of synaptic maturation.... The connection between experience-dependent neural activity and gene expression in the postnatal period forms the basis of learning and memory, and autism symptoms typically emerge during these later stages of development.”
The researchers’ findings also pointed to potential genetic similarities, such as inherited recessive causes, between autism and other neurologic disorders. Among families in the present study who had one member with autism, there was a relatively equivalent male/female ratio compared with other families with autism, indicating that parents who shared a common recent ancestor was a determining factor. In families with more than one member with autism, the male/female ratio was even more balanced.
“The accumulating number of distinct, individually rare genetic causes in autism suggests that the genetic architecture of autism resembles that of mental retardation and epilepsy, with many syndromes, each individually rare, as well as other cases potentially reflecting complex interactions between inherited changes,” Dr. Morrow and colleagues elaborated. “The relatively reduced male/female ratio of affected children and the reduced rate of linked de novo copy number variants in the consanguineous sample [of the present study] ... both suggest that consanguineous pedigrees with autism are enriched for autosomal recessive causes similar to other congenital neurological disorders in consanguineous populations.”
The research team stated that although their findings support recent studies that suggest autism is highly heterogeneous genetically, homozygosity mapping appears to be an effective way to find underlying shared mechanisms. Understanding these genetic underpinnings could eventually help direct various therapies for the different clinical manifestations of autism.
“Our finding that deletions of genes regulated by neuronal activity or regions potentially involved in regulation of gene expression in autism suggests that defects in activity-dependent gene expression may be a cause of cognitive deficits in patients with autism,” the researchers concluded. In addition, their “data implicating noncoding elements in patients with shared ancestry, as well as the heterozygous nonsense changes in patients without shared ancestry, suggest that loss of proper regulation of gene dosage may be an important genetic mechanism in autism.”
Suggested Reading
Morrow EM, Yoo SY, Flavell SW, et al. Identifying autism loci and genes by tracing recent shared ancestry. Science. 2008;321(5886):218-223.
Sutcliffe JS. Genetics: insights into the pathogenesis of autism. Science. 2008;321(5886):208-209.
Suggested Reading
Morrow EM, Yoo SY, Flavell SW, et al. Identifying autism loci and genes by tracing recent shared ancestry. Science. 2008;321(5886):218-223.
Sutcliffe JS. Genetics: insights into the pathogenesis of autism. Science. 2008;321(5886):208-209.
May 2008 Instant Poll Results
FAILED HOME BIRTH, NOW IN THE ED
You are at the hospital, caring for your patients in labor, when a 32-year-old G3P2 with two prior cesarean section deliveries is brought to the emergency department in labor after a failed home birth.
“Will you assume care for this woman?” the nursing administrator asks you. Quickly! What would you do?
7% Refuse to accept responsibility for a high-risk patient whom you’ve never seen
29% Assume her care and recommend cesarean section
46% Assume her care and recommend cesarean section—plus, later, report the responsible midwife to the department of public health and her credentialing organization
18% Agree to assume her care as long as the hospital’s attorney and risk management team indemnify you
FAILED HOME BIRTH, NOW IN THE ED
You are at the hospital, caring for your patients in labor, when a 32-year-old G3P2 with two prior cesarean section deliveries is brought to the emergency department in labor after a failed home birth.
“Will you assume care for this woman?” the nursing administrator asks you. Quickly! What would you do?

7% Refuse to accept responsibility for a high-risk patient whom you’ve never seen
29% Assume her care and recommend cesarean section
46% Assume her care and recommend cesarean section—plus, later, report the responsible midwife to the department of public health and her credentialing organization
18% Agree to assume her care as long as the hospital’s attorney and risk management team indemnify you
FAILED HOME BIRTH, NOW IN THE ED
You are at the hospital, caring for your patients in labor, when a 32-year-old G3P2 with two prior cesarean section deliveries is brought to the emergency department in labor after a failed home birth.
“Will you assume care for this woman?” the nursing administrator asks you. Quickly! What would you do?

7% Refuse to accept responsibility for a high-risk patient whom you’ve never seen
29% Assume her care and recommend cesarean section
46% Assume her care and recommend cesarean section—plus, later, report the responsible midwife to the department of public health and her credentialing organization
18% Agree to assume her care as long as the hospital’s attorney and risk management team indemnify you
Come October 1, a multitude of ICD-9 code additions and revisions arrive
The author reports no financial relationships relevant to this article.
OBs get codes for unremarkable sonograms ordered on the basis of suspicion. For gyn practice, options expand for abnormal Pap smear results. Here are the details.
Revisions and additions to the International Classification of Diseases, Clinical Modification (ICD-9-CM) for 2009, which take effect on October 1, 2008, bring especially good news to obstetricians who are testing for “conditions not found,” evaluating or treating twin-to-twin transfusion syndrome, dealing with the aftermath of maternal surgery, and providing the correct diagnostic code match for screening tests.
Gyn practitioners, don’t feel slighted: Many new codes take effect on that October day, covering abnormal Pap smear results, prophylactic drug treatment, breast conditions, and taking a patient’s personal history.
Remember: 1) October 1 is the key date here—when all the new and revised codes described in this article (and others not reviewed here) are added to the national code set, and 2) as in past years, there will be no grace period!
New and revised OB codes
FOR “CONDITIONS NOT FOUND”
How many times have you ordered a sonogram for a suspected problem with a pregnancy, only to have the scan reveal that all is normal? You then had to use either 1) a screening code for the condition or 2) an unspecified code because you could not assign a code that gave a condition to the patient that she did not have.
With addition of a new category of codes (V89), this obstetrical problem will be solved.
V89.01 Suspected problem with amniotic cavity and membrane not found
V89.02 Suspected placenta not found
V89.03 Suspected fetal anomaly not found
V89.04 Suspected problem with fetal growth not found
V89.05 Suspected cervical shortening not found
V89.09 Other suspected maternal and fetal condition not found
CERVICAL SHORTENING
Women undergo cervical shortening normally as their body prepares for labor, of course, but, on occasion, cervical shortening can indicate impending premature birth. Until now, you might have reflected this condition with 654.5x (Cervical incompetence complicating pregnancy), 654.6x (Other congenital or acquired abnormality of cervix), or 644.1x (Other threatened labor). Starting October 1, however, you’ll have a more precise code available to report this condition: 649.7x (Cervical shortening).
HIGH-RISK PREGNANCY
The V23 category of codes, which represent supervision of high-risk pregnancy, becomes more specific with two additions: V23.85 (Pregnancy resulting from assisted reproductive technology) and V23.86 (Pregnancy with history of in utero procedure during previous pregnancy).
ANTENATAL SCREENING
How to select the right code to report a screening test has been less than clear. Were you performing it to screen for malformation of a fetus? Some other reason? Three new antenatal codes and revision of an existing code (V28.3) clarify the distinction.
V28.3 Encounter for routine screening for malformation using ultrasonics
V28.81 Encounter for fetal anatomic survey
V28.82 Encounter for screening for risk of preterm labor
V28.89 Other specified antenatal screening
ICD-9-CM now directs that the latter code, V28.89, be reported for screening as part of chorionic villus sampling, nuchal translucency testing, genomic screening, and proteomic screening.
COMPLICATIONS OF PREGNANCY AND IN UTERO PROCEDURES
At last, you have a specific code for fetal conjoined twins (678.1x) and one for such fetal hematologic conditions as fetal anemia, thrombocytopenia, and twin-to-twin transfusion syndrome (678.0x).
In addition, complications from an in utero procedure will have two new codes: 679.0x (Maternal complications from in utero procedure) and 679.1x (Fetal complications from in utero procedure).
Gynecologic code changes and additions
ABNORMAL RESULTS OF A PAP SMEAR
You already know to look at the 795 series for ICD-9 codes to support various abnormal Pap smear results; after October 1, you’ll have a lot of new options.
Key developments:
- The risk of dysplasia and carcinoma is the same for the anus as it is for the cervix, so physicians can take anal cytologic smears.
- The cervix and the anus both have transformation zones where mucosa turns from squamous to columnar, so parallel codes have been created for anal smears.
The new codes are listed below.
CERVIX
795.07 Satisfactory cervical smear but lacking transformation zone
VAGINA AND VULVA
795.10 Abnormal Papanicolaou smear of vagina
795.11 Papanicolaou smear of vagina with atypical squamous cells of undetermined significance (ASC-US)
795.12 Papanicolaou smear of vagina with atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)
795.13 Papanicolaou smear of vagina with low grade squamous intraepithelial lesion (LGSIL)
795.14 Papanicolaou smear of vagina with high grade squamous intraepithelial lesion (HGSIL)
795.15 Vaginal high risk papillomavirus (HPV) DNA test positive
795.16 Papanicolaou smear of vagina with cytologic evidence of malignancy
795.18 Unsatisfactory cytology smear
795.19 Other abnormal smear of vagina and vaginal HPV
ANUS
796.70 Abnormal glandular Papanicolaou smear of anus
796.71 Papanicolaou smear of anus with atypical squamous cells of undetermined significance (ASC-US)
796.72 Papanicolaou smear of anus with atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)
796.73 Papanicolaou smear of anus with low grade squamous intraepithelial lesion (LGSIL)
796.74 Papanicolaou smear of anus with high grade squamous intraepithelial lesion (HGSIL)
796.75 Anal high risk human papillomavirus (HPV) DNA test positive
796.76 Papanicolaou smear of anus with cytologic evidence of malignancy
796.77 Satisfactory anal smear but lacking transformation zone
796.78 Unsatisfactory anal cytology smear
796.79 Other abnormal Papanicolaou smear of anus and anal HPV
There is also a new code, 569.44 (Dysplasia of anus), to report anal dysplasia. In the past, this condition was reported using 569.49 (Other specified disorders of rectum and anus).
ACQUIRED ABSENCE CODES
Until now, only V45.77 (Acquired absence of genital organs) could be used to report this patient status. As of October 1, you’ll have to be more specific about what is absent, using any of the following three new codes. You might find these codes helpful in supporting the performance of screening Pap smears:
V88.01 Acquired absence of both cervix and uterus
V88.02 Acquired absence of uterus with remaining cervical stump
V88.03 Acquired absence of cervix with remaining uterus
These new codes can be reported in conjunction with V67.01 (Follow-up vaginal Pap smear) and V76.47 (Special screening for malignant neoplasm of vagina).
URINARY PROBLEMS
Use these three new codes to report various presentations of hematuria:
599.70 Hematuria, unspecified
599.71 Gross hematuria
599.72 Microscopic hematuria
Note: The old code for hematuria (599.7) did not require a fifth digit; after October 1, using that old code will trigger a denial of your claim.
In addition, you have two new codes with which to report urinary symptoms:
788.91 Functional urinary incontinence
788.99 Other symptoms involving urinary symptoms
VULVODYNIA AND VULVAR VESTIBULITIS
A single code (625.8) has been available to describe vulvodynia, and it was grouped into a general category that covered symptoms. This condition has been given three new codes.
625.70 Vulvodynia, unspecified
625.71 Vulvar vestibulitis
625.79 Other vulvodynia
BREAST DISORDERS
New codes for breast conditions are about to take effect. These include ptosis (611.81), hypoplasia (611.82), and other disorders of the breast, such as capsular contracture of a breast implant (611.89).
For surgeons who handle follow-up after breast surgery, two new codes describe problems with the reconstructed breast: 612.0 (Deformity of reconstructed breast) and 612.1 (Disproportion of reconstructed breast).
WOUND DISRUPTION
Under current ICD-9-CM guidelines, you must specify “external wound” or “internal wound” to code correctly for dehiscence. On October 1, you have the option to report an unspecified code, 998.30 (Disruption of wound, unspecified) if the record does not specify the type of wound.
PROPHYLACTIC USE OF AGENTS AFFECTING ESTROGEN RECEPTORS AND ESTROGEN LEVELS
ICD-9-CM created a V code to capture data on the many women who receive tamoxifen and raloxifene after treatment of breast cancer. This code has been expanded to include V codes for different classes of drugs used for this type of therapy:
V07.51 Prophylactic use of selective estrogen receptor modulators (SERMs)
V07.52 Prophylactic use of aromatase inhibitors
V07.59 Prophylactic use of agents affecting estrogen receptors and estrogen levels
From a guideline perspective, you can use the cancer code with one of these codes throughout the course of treatment, including during routine chemotherapy and radiation therapy. Long-term use of a drug that falls under the V07.5x category doesn’t require continued use of the cancer code, however.
You can provide additional information on your patient by reporting her estrogen receptor-positive status (V86.0), personal or family history of breast cancer (V10.3/V16.3), genetic susceptibility to cancer (V84.01–V84.09), and postmenopausal status (V49.81).
TAKING A PERSONAL HISTORY
This year, 11 codes make their debut to allow you to report a patient’s personal history. Use them for encounters in which the personal history has a direct impact on the patient’s complaints or status.
V13.51 Personal history of pathologic fracture
V13.52 Personal history of stress fracture
V13.59 Personal history of other musculoskeletal disorders
V15.51 Personal history of traumatic fracture
V15.59 Personal history of other injury
V15.21 Personal history of undergoing in utero procedure during pregnancy
V15.22 Personal history of undergoing in utero procedure while a fetus
V15.29 Personal history of surgery to other organs
V87.41 Personal history of antineoplastic chemotherapy
V87.42 Personal history of monoclonal drug therapy
V87.49 Personal history of other drug therapy
The author reports no financial relationships relevant to this article.
OBs get codes for unremarkable sonograms ordered on the basis of suspicion. For gyn practice, options expand for abnormal Pap smear results. Here are the details.
Revisions and additions to the International Classification of Diseases, Clinical Modification (ICD-9-CM) for 2009, which take effect on October 1, 2008, bring especially good news to obstetricians who are testing for “conditions not found,” evaluating or treating twin-to-twin transfusion syndrome, dealing with the aftermath of maternal surgery, and providing the correct diagnostic code match for screening tests.
Gyn practitioners, don’t feel slighted: Many new codes take effect on that October day, covering abnormal Pap smear results, prophylactic drug treatment, breast conditions, and taking a patient’s personal history.
Remember: 1) October 1 is the key date here—when all the new and revised codes described in this article (and others not reviewed here) are added to the national code set, and 2) as in past years, there will be no grace period!
New and revised OB codes
FOR “CONDITIONS NOT FOUND”
How many times have you ordered a sonogram for a suspected problem with a pregnancy, only to have the scan reveal that all is normal? You then had to use either 1) a screening code for the condition or 2) an unspecified code because you could not assign a code that gave a condition to the patient that she did not have.
With addition of a new category of codes (V89), this obstetrical problem will be solved.
V89.01 Suspected problem with amniotic cavity and membrane not found
V89.02 Suspected placenta not found
V89.03 Suspected fetal anomaly not found
V89.04 Suspected problem with fetal growth not found
V89.05 Suspected cervical shortening not found
V89.09 Other suspected maternal and fetal condition not found
CERVICAL SHORTENING
Women undergo cervical shortening normally as their body prepares for labor, of course, but, on occasion, cervical shortening can indicate impending premature birth. Until now, you might have reflected this condition with 654.5x (Cervical incompetence complicating pregnancy), 654.6x (Other congenital or acquired abnormality of cervix), or 644.1x (Other threatened labor). Starting October 1, however, you’ll have a more precise code available to report this condition: 649.7x (Cervical shortening).
HIGH-RISK PREGNANCY
The V23 category of codes, which represent supervision of high-risk pregnancy, becomes more specific with two additions: V23.85 (Pregnancy resulting from assisted reproductive technology) and V23.86 (Pregnancy with history of in utero procedure during previous pregnancy).
ANTENATAL SCREENING
How to select the right code to report a screening test has been less than clear. Were you performing it to screen for malformation of a fetus? Some other reason? Three new antenatal codes and revision of an existing code (V28.3) clarify the distinction.
V28.3 Encounter for routine screening for malformation using ultrasonics
V28.81 Encounter for fetal anatomic survey
V28.82 Encounter for screening for risk of preterm labor
V28.89 Other specified antenatal screening
ICD-9-CM now directs that the latter code, V28.89, be reported for screening as part of chorionic villus sampling, nuchal translucency testing, genomic screening, and proteomic screening.
COMPLICATIONS OF PREGNANCY AND IN UTERO PROCEDURES
At last, you have a specific code for fetal conjoined twins (678.1x) and one for such fetal hematologic conditions as fetal anemia, thrombocytopenia, and twin-to-twin transfusion syndrome (678.0x).
In addition, complications from an in utero procedure will have two new codes: 679.0x (Maternal complications from in utero procedure) and 679.1x (Fetal complications from in utero procedure).
Gynecologic code changes and additions
ABNORMAL RESULTS OF A PAP SMEAR
You already know to look at the 795 series for ICD-9 codes to support various abnormal Pap smear results; after October 1, you’ll have a lot of new options.
Key developments:
- The risk of dysplasia and carcinoma is the same for the anus as it is for the cervix, so physicians can take anal cytologic smears.
- The cervix and the anus both have transformation zones where mucosa turns from squamous to columnar, so parallel codes have been created for anal smears.
The new codes are listed below.
CERVIX
795.07 Satisfactory cervical smear but lacking transformation zone
VAGINA AND VULVA
795.10 Abnormal Papanicolaou smear of vagina
795.11 Papanicolaou smear of vagina with atypical squamous cells of undetermined significance (ASC-US)
795.12 Papanicolaou smear of vagina with atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)
795.13 Papanicolaou smear of vagina with low grade squamous intraepithelial lesion (LGSIL)
795.14 Papanicolaou smear of vagina with high grade squamous intraepithelial lesion (HGSIL)
795.15 Vaginal high risk papillomavirus (HPV) DNA test positive
795.16 Papanicolaou smear of vagina with cytologic evidence of malignancy
795.18 Unsatisfactory cytology smear
795.19 Other abnormal smear of vagina and vaginal HPV
ANUS
796.70 Abnormal glandular Papanicolaou smear of anus
796.71 Papanicolaou smear of anus with atypical squamous cells of undetermined significance (ASC-US)
796.72 Papanicolaou smear of anus with atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)
796.73 Papanicolaou smear of anus with low grade squamous intraepithelial lesion (LGSIL)
796.74 Papanicolaou smear of anus with high grade squamous intraepithelial lesion (HGSIL)
796.75 Anal high risk human papillomavirus (HPV) DNA test positive
796.76 Papanicolaou smear of anus with cytologic evidence of malignancy
796.77 Satisfactory anal smear but lacking transformation zone
796.78 Unsatisfactory anal cytology smear
796.79 Other abnormal Papanicolaou smear of anus and anal HPV
There is also a new code, 569.44 (Dysplasia of anus), to report anal dysplasia. In the past, this condition was reported using 569.49 (Other specified disorders of rectum and anus).
ACQUIRED ABSENCE CODES
Until now, only V45.77 (Acquired absence of genital organs) could be used to report this patient status. As of October 1, you’ll have to be more specific about what is absent, using any of the following three new codes. You might find these codes helpful in supporting the performance of screening Pap smears:
V88.01 Acquired absence of both cervix and uterus
V88.02 Acquired absence of uterus with remaining cervical stump
V88.03 Acquired absence of cervix with remaining uterus
These new codes can be reported in conjunction with V67.01 (Follow-up vaginal Pap smear) and V76.47 (Special screening for malignant neoplasm of vagina).
URINARY PROBLEMS
Use these three new codes to report various presentations of hematuria:
599.70 Hematuria, unspecified
599.71 Gross hematuria
599.72 Microscopic hematuria
Note: The old code for hematuria (599.7) did not require a fifth digit; after October 1, using that old code will trigger a denial of your claim.
In addition, you have two new codes with which to report urinary symptoms:
788.91 Functional urinary incontinence
788.99 Other symptoms involving urinary symptoms
VULVODYNIA AND VULVAR VESTIBULITIS
A single code (625.8) has been available to describe vulvodynia, and it was grouped into a general category that covered symptoms. This condition has been given three new codes.
625.70 Vulvodynia, unspecified
625.71 Vulvar vestibulitis
625.79 Other vulvodynia
BREAST DISORDERS
New codes for breast conditions are about to take effect. These include ptosis (611.81), hypoplasia (611.82), and other disorders of the breast, such as capsular contracture of a breast implant (611.89).
For surgeons who handle follow-up after breast surgery, two new codes describe problems with the reconstructed breast: 612.0 (Deformity of reconstructed breast) and 612.1 (Disproportion of reconstructed breast).
WOUND DISRUPTION
Under current ICD-9-CM guidelines, you must specify “external wound” or “internal wound” to code correctly for dehiscence. On October 1, you have the option to report an unspecified code, 998.30 (Disruption of wound, unspecified) if the record does not specify the type of wound.
PROPHYLACTIC USE OF AGENTS AFFECTING ESTROGEN RECEPTORS AND ESTROGEN LEVELS
ICD-9-CM created a V code to capture data on the many women who receive tamoxifen and raloxifene after treatment of breast cancer. This code has been expanded to include V codes for different classes of drugs used for this type of therapy:
V07.51 Prophylactic use of selective estrogen receptor modulators (SERMs)
V07.52 Prophylactic use of aromatase inhibitors
V07.59 Prophylactic use of agents affecting estrogen receptors and estrogen levels
From a guideline perspective, you can use the cancer code with one of these codes throughout the course of treatment, including during routine chemotherapy and radiation therapy. Long-term use of a drug that falls under the V07.5x category doesn’t require continued use of the cancer code, however.
You can provide additional information on your patient by reporting her estrogen receptor-positive status (V86.0), personal or family history of breast cancer (V10.3/V16.3), genetic susceptibility to cancer (V84.01–V84.09), and postmenopausal status (V49.81).
TAKING A PERSONAL HISTORY
This year, 11 codes make their debut to allow you to report a patient’s personal history. Use them for encounters in which the personal history has a direct impact on the patient’s complaints or status.
V13.51 Personal history of pathologic fracture
V13.52 Personal history of stress fracture
V13.59 Personal history of other musculoskeletal disorders
V15.51 Personal history of traumatic fracture
V15.59 Personal history of other injury
V15.21 Personal history of undergoing in utero procedure during pregnancy
V15.22 Personal history of undergoing in utero procedure while a fetus
V15.29 Personal history of surgery to other organs
V87.41 Personal history of antineoplastic chemotherapy
V87.42 Personal history of monoclonal drug therapy
V87.49 Personal history of other drug therapy
The author reports no financial relationships relevant to this article.
OBs get codes for unremarkable sonograms ordered on the basis of suspicion. For gyn practice, options expand for abnormal Pap smear results. Here are the details.
Revisions and additions to the International Classification of Diseases, Clinical Modification (ICD-9-CM) for 2009, which take effect on October 1, 2008, bring especially good news to obstetricians who are testing for “conditions not found,” evaluating or treating twin-to-twin transfusion syndrome, dealing with the aftermath of maternal surgery, and providing the correct diagnostic code match for screening tests.
Gyn practitioners, don’t feel slighted: Many new codes take effect on that October day, covering abnormal Pap smear results, prophylactic drug treatment, breast conditions, and taking a patient’s personal history.
Remember: 1) October 1 is the key date here—when all the new and revised codes described in this article (and others not reviewed here) are added to the national code set, and 2) as in past years, there will be no grace period!
New and revised OB codes
FOR “CONDITIONS NOT FOUND”
How many times have you ordered a sonogram for a suspected problem with a pregnancy, only to have the scan reveal that all is normal? You then had to use either 1) a screening code for the condition or 2) an unspecified code because you could not assign a code that gave a condition to the patient that she did not have.
With addition of a new category of codes (V89), this obstetrical problem will be solved.
V89.01 Suspected problem with amniotic cavity and membrane not found
V89.02 Suspected placenta not found
V89.03 Suspected fetal anomaly not found
V89.04 Suspected problem with fetal growth not found
V89.05 Suspected cervical shortening not found
V89.09 Other suspected maternal and fetal condition not found
CERVICAL SHORTENING
Women undergo cervical shortening normally as their body prepares for labor, of course, but, on occasion, cervical shortening can indicate impending premature birth. Until now, you might have reflected this condition with 654.5x (Cervical incompetence complicating pregnancy), 654.6x (Other congenital or acquired abnormality of cervix), or 644.1x (Other threatened labor). Starting October 1, however, you’ll have a more precise code available to report this condition: 649.7x (Cervical shortening).
HIGH-RISK PREGNANCY
The V23 category of codes, which represent supervision of high-risk pregnancy, becomes more specific with two additions: V23.85 (Pregnancy resulting from assisted reproductive technology) and V23.86 (Pregnancy with history of in utero procedure during previous pregnancy).
ANTENATAL SCREENING
How to select the right code to report a screening test has been less than clear. Were you performing it to screen for malformation of a fetus? Some other reason? Three new antenatal codes and revision of an existing code (V28.3) clarify the distinction.
V28.3 Encounter for routine screening for malformation using ultrasonics
V28.81 Encounter for fetal anatomic survey
V28.82 Encounter for screening for risk of preterm labor
V28.89 Other specified antenatal screening
ICD-9-CM now directs that the latter code, V28.89, be reported for screening as part of chorionic villus sampling, nuchal translucency testing, genomic screening, and proteomic screening.
COMPLICATIONS OF PREGNANCY AND IN UTERO PROCEDURES
At last, you have a specific code for fetal conjoined twins (678.1x) and one for such fetal hematologic conditions as fetal anemia, thrombocytopenia, and twin-to-twin transfusion syndrome (678.0x).
In addition, complications from an in utero procedure will have two new codes: 679.0x (Maternal complications from in utero procedure) and 679.1x (Fetal complications from in utero procedure).
Gynecologic code changes and additions
ABNORMAL RESULTS OF A PAP SMEAR
You already know to look at the 795 series for ICD-9 codes to support various abnormal Pap smear results; after October 1, you’ll have a lot of new options.
Key developments:
- The risk of dysplasia and carcinoma is the same for the anus as it is for the cervix, so physicians can take anal cytologic smears.
- The cervix and the anus both have transformation zones where mucosa turns from squamous to columnar, so parallel codes have been created for anal smears.
The new codes are listed below.
CERVIX
795.07 Satisfactory cervical smear but lacking transformation zone
VAGINA AND VULVA
795.10 Abnormal Papanicolaou smear of vagina
795.11 Papanicolaou smear of vagina with atypical squamous cells of undetermined significance (ASC-US)
795.12 Papanicolaou smear of vagina with atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)
795.13 Papanicolaou smear of vagina with low grade squamous intraepithelial lesion (LGSIL)
795.14 Papanicolaou smear of vagina with high grade squamous intraepithelial lesion (HGSIL)
795.15 Vaginal high risk papillomavirus (HPV) DNA test positive
795.16 Papanicolaou smear of vagina with cytologic evidence of malignancy
795.18 Unsatisfactory cytology smear
795.19 Other abnormal smear of vagina and vaginal HPV
ANUS
796.70 Abnormal glandular Papanicolaou smear of anus
796.71 Papanicolaou smear of anus with atypical squamous cells of undetermined significance (ASC-US)
796.72 Papanicolaou smear of anus with atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)
796.73 Papanicolaou smear of anus with low grade squamous intraepithelial lesion (LGSIL)
796.74 Papanicolaou smear of anus with high grade squamous intraepithelial lesion (HGSIL)
796.75 Anal high risk human papillomavirus (HPV) DNA test positive
796.76 Papanicolaou smear of anus with cytologic evidence of malignancy
796.77 Satisfactory anal smear but lacking transformation zone
796.78 Unsatisfactory anal cytology smear
796.79 Other abnormal Papanicolaou smear of anus and anal HPV
There is also a new code, 569.44 (Dysplasia of anus), to report anal dysplasia. In the past, this condition was reported using 569.49 (Other specified disorders of rectum and anus).
ACQUIRED ABSENCE CODES
Until now, only V45.77 (Acquired absence of genital organs) could be used to report this patient status. As of October 1, you’ll have to be more specific about what is absent, using any of the following three new codes. You might find these codes helpful in supporting the performance of screening Pap smears:
V88.01 Acquired absence of both cervix and uterus
V88.02 Acquired absence of uterus with remaining cervical stump
V88.03 Acquired absence of cervix with remaining uterus
These new codes can be reported in conjunction with V67.01 (Follow-up vaginal Pap smear) and V76.47 (Special screening for malignant neoplasm of vagina).
URINARY PROBLEMS
Use these three new codes to report various presentations of hematuria:
599.70 Hematuria, unspecified
599.71 Gross hematuria
599.72 Microscopic hematuria
Note: The old code for hematuria (599.7) did not require a fifth digit; after October 1, using that old code will trigger a denial of your claim.
In addition, you have two new codes with which to report urinary symptoms:
788.91 Functional urinary incontinence
788.99 Other symptoms involving urinary symptoms
VULVODYNIA AND VULVAR VESTIBULITIS
A single code (625.8) has been available to describe vulvodynia, and it was grouped into a general category that covered symptoms. This condition has been given three new codes.
625.70 Vulvodynia, unspecified
625.71 Vulvar vestibulitis
625.79 Other vulvodynia
BREAST DISORDERS
New codes for breast conditions are about to take effect. These include ptosis (611.81), hypoplasia (611.82), and other disorders of the breast, such as capsular contracture of a breast implant (611.89).
For surgeons who handle follow-up after breast surgery, two new codes describe problems with the reconstructed breast: 612.0 (Deformity of reconstructed breast) and 612.1 (Disproportion of reconstructed breast).
WOUND DISRUPTION
Under current ICD-9-CM guidelines, you must specify “external wound” or “internal wound” to code correctly for dehiscence. On October 1, you have the option to report an unspecified code, 998.30 (Disruption of wound, unspecified) if the record does not specify the type of wound.
PROPHYLACTIC USE OF AGENTS AFFECTING ESTROGEN RECEPTORS AND ESTROGEN LEVELS
ICD-9-CM created a V code to capture data on the many women who receive tamoxifen and raloxifene after treatment of breast cancer. This code has been expanded to include V codes for different classes of drugs used for this type of therapy:
V07.51 Prophylactic use of selective estrogen receptor modulators (SERMs)
V07.52 Prophylactic use of aromatase inhibitors
V07.59 Prophylactic use of agents affecting estrogen receptors and estrogen levels
From a guideline perspective, you can use the cancer code with one of these codes throughout the course of treatment, including during routine chemotherapy and radiation therapy. Long-term use of a drug that falls under the V07.5x category doesn’t require continued use of the cancer code, however.
You can provide additional information on your patient by reporting her estrogen receptor-positive status (V86.0), personal or family history of breast cancer (V10.3/V16.3), genetic susceptibility to cancer (V84.01–V84.09), and postmenopausal status (V49.81).
TAKING A PERSONAL HISTORY
This year, 11 codes make their debut to allow you to report a patient’s personal history. Use them for encounters in which the personal history has a direct impact on the patient’s complaints or status.
V13.51 Personal history of pathologic fracture
V13.52 Personal history of stress fracture
V13.59 Personal history of other musculoskeletal disorders
V15.51 Personal history of traumatic fracture
V15.59 Personal history of other injury
V15.21 Personal history of undergoing in utero procedure during pregnancy
V15.22 Personal history of undergoing in utero procedure while a fetus
V15.29 Personal history of surgery to other organs
V87.41 Personal history of antineoplastic chemotherapy
V87.42 Personal history of monoclonal drug therapy
V87.49 Personal history of other drug therapy
Help smokers quit: Tell them their “lung age”
ILLUSTRATIVE CASE
A 48-year-old man comes to your office for a routine physical. He has a 30 pack-year smoking history. When you talk to him about smoking cessation, he tells you he’s tried to stop more than once, but he can’t seem to stay motivated. You find no evidence of chronic lung disease and do not perform spirometry screening. (The US Preventive Services Task Force does not recommend spirometry for asymptomatic patients.) But could spirometry have therapeutic value in this case?
Smoking is the leading modifiable risk factor for mortality in the United States,2 and smoking cessation is the most effective intervention. Nortriptyline, bupropion, nicotine replacement agents, and varenicline are effective pharmacological treatments.3 Adding counseling to medication significantly improves quit rates (estimated odds ratio [OR]=1.4; 95% confidence interval [CI], 1.2-1.6).3 Nonetheless, physicians’ efforts to help patients stop smoking frequently fail.
But another option has caught—and held—the attention of researchers.
The promise of biomarkers
It has long been suspected that presenting smokers with evidence of tobacco’s harmful effect on their bodies—biomarkers—might encourage them to stop. Biomarkers that have been tested in randomized controlled trials (RCTs) include spirometry, exhaled carbon monoxide measurement, ultrasonography of carotid and femoral arteries, and genetic susceptibility to lung cancer, as well as combinations of these markers. But the results of most biomarker studies have been disappointing. A 2005 Cochrane Database review found insufficient evidence of the effectiveness of these markers in boosting quit rates.4
Lung age, a biomarker that’s easily understood
Lung age, a clever presentation of spirometry results, had not been tested in an RCT prior to the study we summarize below. Defined in 1985, lung age refers to the average age of a nonsmoker with a forced expiratory volume at 1 second (FEV1) equal to that of the person being tested ( FIGURE 1 ). The primary purpose was to make spirometry results easier for patients to understand, but researchers also envisioned it as a way to demonstrate the premature lung damage suffered as a consequence of smoking.5
FIGURE 1
Translating FEV1 into lung age1
STUDY SUMMARY: Graphic display more effective than FEV1 results
This study was a well-done, multicenter RCT evaluating the effect on tobacco quit rates of informing adult smokers of their lung age.1 Smokers ages 35 and older from 5 general practices in England were invited to participate. The authors excluded patients using oxygen and those with a history of tuberculosis, lung cancer, asbestosis, bronchiectasis, silicosis, or pneumonectomy. The study included 561 participants with an average of 33 pack-years of smoking, who underwent spirometry before being divided into an intervention or a control group. The researchers used standardized instruments to confirm the baseline comparability of the 2 groups.
Subjects in both groups were given information about local smoking cessation clinics and strongly encouraged to quit. All were told that their lung function would be retested in 12 months.
The controls received letters with their spirometry results presented as FEV1. In contrast, participants in the intervention group received the results in the form of a computer-generated graphic display of lung age ( FIGURE 2 ), which was further explained by a health care worker. They also received a letter within 1 month containing the same data. Participants were evaluated for smoking cessation at 12 months, and those who reported quitting received confirmatory carbon monoxide breath testing and salivary cotinine testing. Eleven percent of the subjects were lost to follow-up.
FIGURE 2
Lung age helps spirometry pack a bigger punch
Drawing a vertical line from the patient’s age (on the horizontal axis) to reach the solid curve representing the lung function of the “susceptible smoker” and extending the line horizontally to reach the curve with the broken lines representing “never smokers” graphically shows the patient’s lung age and the accelerated decline in lung function associated with smoking. The patient shown here is a 52-year-old smoker with FEV1 equivalent to a 75-year-old nonsmoker.
Source: Parkes G et al. BMJ. 2008;336:598-600. Reproduced with permission from the BMJ Publishing Group.
Quit rates higher when patients know lung age
At 1 year, verified quit rates were 13.6% in the intervention group and 6.4% in the control group (a difference of 7.2%, 95% CI, 2.2%-12.1%; P=.005). This means that for every 14 smokers who are told their lung age and shown a graphic display of this biomarker, 1 additional smoker will quit after 1 year.
Contrary to what might be expected, the investigators found that quitting did not depend on the degree of lung damage. Patients with both normal and abnormal lung age quit smoking at similar rates.
WHAT’S NEW: Lung age resonates more than spirometry alone
This is the first RCT demonstrating that informing smokers of their lung age can help them quit, and the first well-designed study to demonstrate improved cessation rates using a physiological biomarker. The research also suggests that successful quitting may have less to do with spirometry results—the level of severity of lung damage it shows—than with the way the results are presented. Giving patients information about their lung function in an easily understandable format, the authors observe, appears to result in higher quit rates.
CAVEATS: Young smokers weren’t studied
The study did not test to see if this intervention would work in younger adults, as only those 35 years of age and older were enrolled. This is a single study, and it is possible that the findings cannot be generalized to other groups or are due to unmeasured confounding factors. However, the intervention is unlikely to cause any significant harm, so we see no risks associated with it other than the cost of spirometry.
CHALLENGES TO IMPLEMENTATION: Time and expense of spirometry
We suspect the biggest challenges to implementing this recommendation in clinical practice are the expense of obtaining a spirometer ( TABLE ), staff training for those practices without one, and the time needed for the intervention. The average time to perform spirometry on study participants was 30 minutes; a health care worker spent, on average, another 15 minutes reviewing results with each member of the intervention group.
Another challenge: Not all spirometers calculate lung age or can create a graphic similar to FIGURE 2 . However, any FEV1 measurement, whether it is generated by formal pulmonary function testing or by an inexpensive hand-held meter, can easily be converted to lung age using the formula shown in FIGURE 1 . If desired, the same elements—the patient’s age, height, and gender as well as FEV1—could also be used to create a computer-generated graphic display.
TABLE
Spirometry: equipment costs
| The initial cost of a spirometer varies widely, depending on the sophistication of the equipment and the available options and features. Additional costs—for disposable mouthpieces, line filters, nose clips, and hoses, for example—are low. A sampling of reasonably priced models well suited for office use is shown below. All of these models meet American Thoracic Society criteria for spirometry, and all calculate lung age. | ||
|---|---|---|
| SPIROMETER MANUFACTURER/MODEL | PRICE | SUPPLIER |
| Futuremed Discovery-2 | $2,125 | medsupplier.com |
| Micro Medical MicroLoop | $1,780 | Miami-med.com |
| Micro Medical SpiroUSB | $1,580 | Miami-med.com |
| NDD EasyOne Frontline | $1,000 | medsupplier.com |
| SDI Diagnostics Spirolab II | $2,600 | med-electronics.com |
This study was selected and evaluated using FPIN’s Priority Updates from the Research Literature (PURL) Surveillance System methodology. The criteria and findings leading to the selection of this study as a PURL can be accessed at www.jfponline.com/purls.
PURL METHODOLOGY
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Parkes G, Greenhalgh T, Griffin M, Dent R. Effect on smoking quit rate of telling patients their lung age: the Step2quit randomised controlled trial. BMJ. 2008;336:598-600.
2. Mokdad AH, Marks JS, Stroup DF, Gerberding JL. Actual causes of death in the United States, 2000. JAMA. 2004;291:1238-1245.
3. Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update. Clinical practice guideline. Rockville, MD: US Department of Health and Human Services, Public Health Service; May 2008. Available at: http://www.surgeongeneral.gov/tobacco/treating_tobacco_use08.pdf. Accessed July 6, 2008.
4. Bize R, Burnand B, Mueller Y, Cornuz J. Biomedical risk assessment as an aid for smoking cessation. Cochrane Database Syst Rev. 2005;(4):CD004705.-
5. Morris JF, Temple W. Spirometric “lung age” estimation for motivating smoking cessation. Prev Med. 1985;14:655-662.
ILLUSTRATIVE CASE
A 48-year-old man comes to your office for a routine physical. He has a 30 pack-year smoking history. When you talk to him about smoking cessation, he tells you he’s tried to stop more than once, but he can’t seem to stay motivated. You find no evidence of chronic lung disease and do not perform spirometry screening. (The US Preventive Services Task Force does not recommend spirometry for asymptomatic patients.) But could spirometry have therapeutic value in this case?
Smoking is the leading modifiable risk factor for mortality in the United States,2 and smoking cessation is the most effective intervention. Nortriptyline, bupropion, nicotine replacement agents, and varenicline are effective pharmacological treatments.3 Adding counseling to medication significantly improves quit rates (estimated odds ratio [OR]=1.4; 95% confidence interval [CI], 1.2-1.6).3 Nonetheless, physicians’ efforts to help patients stop smoking frequently fail.
But another option has caught—and held—the attention of researchers.
The promise of biomarkers
It has long been suspected that presenting smokers with evidence of tobacco’s harmful effect on their bodies—biomarkers—might encourage them to stop. Biomarkers that have been tested in randomized controlled trials (RCTs) include spirometry, exhaled carbon monoxide measurement, ultrasonography of carotid and femoral arteries, and genetic susceptibility to lung cancer, as well as combinations of these markers. But the results of most biomarker studies have been disappointing. A 2005 Cochrane Database review found insufficient evidence of the effectiveness of these markers in boosting quit rates.4
Lung age, a biomarker that’s easily understood
Lung age, a clever presentation of spirometry results, had not been tested in an RCT prior to the study we summarize below. Defined in 1985, lung age refers to the average age of a nonsmoker with a forced expiratory volume at 1 second (FEV1) equal to that of the person being tested ( FIGURE 1 ). The primary purpose was to make spirometry results easier for patients to understand, but researchers also envisioned it as a way to demonstrate the premature lung damage suffered as a consequence of smoking.5
FIGURE 1
Translating FEV1 into lung age1
STUDY SUMMARY: Graphic display more effective than FEV1 results
This study was a well-done, multicenter RCT evaluating the effect on tobacco quit rates of informing adult smokers of their lung age.1 Smokers ages 35 and older from 5 general practices in England were invited to participate. The authors excluded patients using oxygen and those with a history of tuberculosis, lung cancer, asbestosis, bronchiectasis, silicosis, or pneumonectomy. The study included 561 participants with an average of 33 pack-years of smoking, who underwent spirometry before being divided into an intervention or a control group. The researchers used standardized instruments to confirm the baseline comparability of the 2 groups.
Subjects in both groups were given information about local smoking cessation clinics and strongly encouraged to quit. All were told that their lung function would be retested in 12 months.
The controls received letters with their spirometry results presented as FEV1. In contrast, participants in the intervention group received the results in the form of a computer-generated graphic display of lung age ( FIGURE 2 ), which was further explained by a health care worker. They also received a letter within 1 month containing the same data. Participants were evaluated for smoking cessation at 12 months, and those who reported quitting received confirmatory carbon monoxide breath testing and salivary cotinine testing. Eleven percent of the subjects were lost to follow-up.
FIGURE 2
Lung age helps spirometry pack a bigger punch
Drawing a vertical line from the patient’s age (on the horizontal axis) to reach the solid curve representing the lung function of the “susceptible smoker” and extending the line horizontally to reach the curve with the broken lines representing “never smokers” graphically shows the patient’s lung age and the accelerated decline in lung function associated with smoking. The patient shown here is a 52-year-old smoker with FEV1 equivalent to a 75-year-old nonsmoker.
Source: Parkes G et al. BMJ. 2008;336:598-600. Reproduced with permission from the BMJ Publishing Group.
Quit rates higher when patients know lung age
At 1 year, verified quit rates were 13.6% in the intervention group and 6.4% in the control group (a difference of 7.2%, 95% CI, 2.2%-12.1%; P=.005). This means that for every 14 smokers who are told their lung age and shown a graphic display of this biomarker, 1 additional smoker will quit after 1 year.
Contrary to what might be expected, the investigators found that quitting did not depend on the degree of lung damage. Patients with both normal and abnormal lung age quit smoking at similar rates.
WHAT’S NEW: Lung age resonates more than spirometry alone
This is the first RCT demonstrating that informing smokers of their lung age can help them quit, and the first well-designed study to demonstrate improved cessation rates using a physiological biomarker. The research also suggests that successful quitting may have less to do with spirometry results—the level of severity of lung damage it shows—than with the way the results are presented. Giving patients information about their lung function in an easily understandable format, the authors observe, appears to result in higher quit rates.
CAVEATS: Young smokers weren’t studied
The study did not test to see if this intervention would work in younger adults, as only those 35 years of age and older were enrolled. This is a single study, and it is possible that the findings cannot be generalized to other groups or are due to unmeasured confounding factors. However, the intervention is unlikely to cause any significant harm, so we see no risks associated with it other than the cost of spirometry.
CHALLENGES TO IMPLEMENTATION: Time and expense of spirometry
We suspect the biggest challenges to implementing this recommendation in clinical practice are the expense of obtaining a spirometer ( TABLE ), staff training for those practices without one, and the time needed for the intervention. The average time to perform spirometry on study participants was 30 minutes; a health care worker spent, on average, another 15 minutes reviewing results with each member of the intervention group.
Another challenge: Not all spirometers calculate lung age or can create a graphic similar to FIGURE 2 . However, any FEV1 measurement, whether it is generated by formal pulmonary function testing or by an inexpensive hand-held meter, can easily be converted to lung age using the formula shown in FIGURE 1 . If desired, the same elements—the patient’s age, height, and gender as well as FEV1—could also be used to create a computer-generated graphic display.
TABLE
Spirometry: equipment costs
| The initial cost of a spirometer varies widely, depending on the sophistication of the equipment and the available options and features. Additional costs—for disposable mouthpieces, line filters, nose clips, and hoses, for example—are low. A sampling of reasonably priced models well suited for office use is shown below. All of these models meet American Thoracic Society criteria for spirometry, and all calculate lung age. | ||
|---|---|---|
| SPIROMETER MANUFACTURER/MODEL | PRICE | SUPPLIER |
| Futuremed Discovery-2 | $2,125 | medsupplier.com |
| Micro Medical MicroLoop | $1,780 | Miami-med.com |
| Micro Medical SpiroUSB | $1,580 | Miami-med.com |
| NDD EasyOne Frontline | $1,000 | medsupplier.com |
| SDI Diagnostics Spirolab II | $2,600 | med-electronics.com |
This study was selected and evaluated using FPIN’s Priority Updates from the Research Literature (PURL) Surveillance System methodology. The criteria and findings leading to the selection of this study as a PURL can be accessed at www.jfponline.com/purls.
PURL METHODOLOGY
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 48-year-old man comes to your office for a routine physical. He has a 30 pack-year smoking history. When you talk to him about smoking cessation, he tells you he’s tried to stop more than once, but he can’t seem to stay motivated. You find no evidence of chronic lung disease and do not perform spirometry screening. (The US Preventive Services Task Force does not recommend spirometry for asymptomatic patients.) But could spirometry have therapeutic value in this case?
Smoking is the leading modifiable risk factor for mortality in the United States,2 and smoking cessation is the most effective intervention. Nortriptyline, bupropion, nicotine replacement agents, and varenicline are effective pharmacological treatments.3 Adding counseling to medication significantly improves quit rates (estimated odds ratio [OR]=1.4; 95% confidence interval [CI], 1.2-1.6).3 Nonetheless, physicians’ efforts to help patients stop smoking frequently fail.
But another option has caught—and held—the attention of researchers.
The promise of biomarkers
It has long been suspected that presenting smokers with evidence of tobacco’s harmful effect on their bodies—biomarkers—might encourage them to stop. Biomarkers that have been tested in randomized controlled trials (RCTs) include spirometry, exhaled carbon monoxide measurement, ultrasonography of carotid and femoral arteries, and genetic susceptibility to lung cancer, as well as combinations of these markers. But the results of most biomarker studies have been disappointing. A 2005 Cochrane Database review found insufficient evidence of the effectiveness of these markers in boosting quit rates.4
Lung age, a biomarker that’s easily understood
Lung age, a clever presentation of spirometry results, had not been tested in an RCT prior to the study we summarize below. Defined in 1985, lung age refers to the average age of a nonsmoker with a forced expiratory volume at 1 second (FEV1) equal to that of the person being tested ( FIGURE 1 ). The primary purpose was to make spirometry results easier for patients to understand, but researchers also envisioned it as a way to demonstrate the premature lung damage suffered as a consequence of smoking.5
FIGURE 1
Translating FEV1 into lung age1
STUDY SUMMARY: Graphic display more effective than FEV1 results
This study was a well-done, multicenter RCT evaluating the effect on tobacco quit rates of informing adult smokers of their lung age.1 Smokers ages 35 and older from 5 general practices in England were invited to participate. The authors excluded patients using oxygen and those with a history of tuberculosis, lung cancer, asbestosis, bronchiectasis, silicosis, or pneumonectomy. The study included 561 participants with an average of 33 pack-years of smoking, who underwent spirometry before being divided into an intervention or a control group. The researchers used standardized instruments to confirm the baseline comparability of the 2 groups.
Subjects in both groups were given information about local smoking cessation clinics and strongly encouraged to quit. All were told that their lung function would be retested in 12 months.
The controls received letters with their spirometry results presented as FEV1. In contrast, participants in the intervention group received the results in the form of a computer-generated graphic display of lung age ( FIGURE 2 ), which was further explained by a health care worker. They also received a letter within 1 month containing the same data. Participants were evaluated for smoking cessation at 12 months, and those who reported quitting received confirmatory carbon monoxide breath testing and salivary cotinine testing. Eleven percent of the subjects were lost to follow-up.
FIGURE 2
Lung age helps spirometry pack a bigger punch
Drawing a vertical line from the patient’s age (on the horizontal axis) to reach the solid curve representing the lung function of the “susceptible smoker” and extending the line horizontally to reach the curve with the broken lines representing “never smokers” graphically shows the patient’s lung age and the accelerated decline in lung function associated with smoking. The patient shown here is a 52-year-old smoker with FEV1 equivalent to a 75-year-old nonsmoker.
Source: Parkes G et al. BMJ. 2008;336:598-600. Reproduced with permission from the BMJ Publishing Group.
Quit rates higher when patients know lung age
At 1 year, verified quit rates were 13.6% in the intervention group and 6.4% in the control group (a difference of 7.2%, 95% CI, 2.2%-12.1%; P=.005). This means that for every 14 smokers who are told their lung age and shown a graphic display of this biomarker, 1 additional smoker will quit after 1 year.
Contrary to what might be expected, the investigators found that quitting did not depend on the degree of lung damage. Patients with both normal and abnormal lung age quit smoking at similar rates.
WHAT’S NEW: Lung age resonates more than spirometry alone
This is the first RCT demonstrating that informing smokers of their lung age can help them quit, and the first well-designed study to demonstrate improved cessation rates using a physiological biomarker. The research also suggests that successful quitting may have less to do with spirometry results—the level of severity of lung damage it shows—than with the way the results are presented. Giving patients information about their lung function in an easily understandable format, the authors observe, appears to result in higher quit rates.
CAVEATS: Young smokers weren’t studied
The study did not test to see if this intervention would work in younger adults, as only those 35 years of age and older were enrolled. This is a single study, and it is possible that the findings cannot be generalized to other groups or are due to unmeasured confounding factors. However, the intervention is unlikely to cause any significant harm, so we see no risks associated with it other than the cost of spirometry.
CHALLENGES TO IMPLEMENTATION: Time and expense of spirometry
We suspect the biggest challenges to implementing this recommendation in clinical practice are the expense of obtaining a spirometer ( TABLE ), staff training for those practices without one, and the time needed for the intervention. The average time to perform spirometry on study participants was 30 minutes; a health care worker spent, on average, another 15 minutes reviewing results with each member of the intervention group.
Another challenge: Not all spirometers calculate lung age or can create a graphic similar to FIGURE 2 . However, any FEV1 measurement, whether it is generated by formal pulmonary function testing or by an inexpensive hand-held meter, can easily be converted to lung age using the formula shown in FIGURE 1 . If desired, the same elements—the patient’s age, height, and gender as well as FEV1—could also be used to create a computer-generated graphic display.
TABLE
Spirometry: equipment costs
| The initial cost of a spirometer varies widely, depending on the sophistication of the equipment and the available options and features. Additional costs—for disposable mouthpieces, line filters, nose clips, and hoses, for example—are low. A sampling of reasonably priced models well suited for office use is shown below. All of these models meet American Thoracic Society criteria for spirometry, and all calculate lung age. | ||
|---|---|---|
| SPIROMETER MANUFACTURER/MODEL | PRICE | SUPPLIER |
| Futuremed Discovery-2 | $2,125 | medsupplier.com |
| Micro Medical MicroLoop | $1,780 | Miami-med.com |
| Micro Medical SpiroUSB | $1,580 | Miami-med.com |
| NDD EasyOne Frontline | $1,000 | medsupplier.com |
| SDI Diagnostics Spirolab II | $2,600 | med-electronics.com |
This study was selected and evaluated using FPIN’s Priority Updates from the Research Literature (PURL) Surveillance System methodology. The criteria and findings leading to the selection of this study as a PURL can be accessed at www.jfponline.com/purls.
PURL METHODOLOGY
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Parkes G, Greenhalgh T, Griffin M, Dent R. Effect on smoking quit rate of telling patients their lung age: the Step2quit randomised controlled trial. BMJ. 2008;336:598-600.
2. Mokdad AH, Marks JS, Stroup DF, Gerberding JL. Actual causes of death in the United States, 2000. JAMA. 2004;291:1238-1245.
3. Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update. Clinical practice guideline. Rockville, MD: US Department of Health and Human Services, Public Health Service; May 2008. Available at: http://www.surgeongeneral.gov/tobacco/treating_tobacco_use08.pdf. Accessed July 6, 2008.
4. Bize R, Burnand B, Mueller Y, Cornuz J. Biomedical risk assessment as an aid for smoking cessation. Cochrane Database Syst Rev. 2005;(4):CD004705.-
5. Morris JF, Temple W. Spirometric “lung age” estimation for motivating smoking cessation. Prev Med. 1985;14:655-662.
1. Parkes G, Greenhalgh T, Griffin M, Dent R. Effect on smoking quit rate of telling patients their lung age: the Step2quit randomised controlled trial. BMJ. 2008;336:598-600.
2. Mokdad AH, Marks JS, Stroup DF, Gerberding JL. Actual causes of death in the United States, 2000. JAMA. 2004;291:1238-1245.
3. Fiore MC, Jaén CR, Baker TB, et al. Treating Tobacco Use and Dependence: 2008 Update. Clinical practice guideline. Rockville, MD: US Department of Health and Human Services, Public Health Service; May 2008. Available at: http://www.surgeongeneral.gov/tobacco/treating_tobacco_use08.pdf. Accessed July 6, 2008.
4. Bize R, Burnand B, Mueller Y, Cornuz J. Biomedical risk assessment as an aid for smoking cessation. Cochrane Database Syst Rev. 2005;(4):CD004705.-
5. Morris JF, Temple W. Spirometric “lung age” estimation for motivating smoking cessation. Prev Med. 1985;14:655-662.
Copyright © 2008 The Family Physicians Inquiries Network.
All rights reserved.
Help patients gain better asthma control
- Assess asthma severity before initiating treatment; monitor asthma control to guide adjustments in therapy using measures of impairment (B) and risk (C) (National Heart, Lung, and Blood Institute [NHLBI] and National Asthma Education and Prevention Program [NAEPP] third expert panel report [EPR-3]).
- Base treatment decisions on recommendations specific to each age group (0-4 years, 5-11 years, and ≥12 years) (A).
- Use spirometry in patients ≥5 years of age to diagnose asthma, classify severity, and assess control (C).
- Provide each patient with a written asthma action plan with instructions for daily disease management, as well as identification of, and response to, worsening symptoms (B).
EPR-3 evidence categories:
- Randomized, controlled trials (RCTs), rich body of data
- RCTs, limited body of data
- Nonrandomized trials and observational studies
- Panel consensus judgment
JJ, a 4-year-old boy, was taken to an urgent care clinic 3 times last winter for “recurrent bronchitis” and given a 7-day course of prednisone and antibiotics at each visit. His mother reports that “his colds always seem to go to his chest” and his skin is always dry. She was given a nebulizer and albuterol for use when JJ begins wheezing, which often happens when he has a cold, plays vigorously, or visits a friend who has cats.
JJ is one of approximately 6.7 million children—and 22.9 million US residents—who have asthma.1 To help guide the care of patients like JJ, the National Heart, Lung, and Blood Institute (NHLBI) and National Asthma Education and Prevention Program (NAEPP) released the third expert panel report (EPR-3) in 2007. Available at http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm, the EPR-3 provides the most comprehensive evidence-based guidance for the diagnosis and management of asthma to date.2
The guidelines were an invaluable resource for JJ’s family physician, who referred to them to categorize the severity of JJ’s asthma as “mild persistent.” In initiating treatment, JJ’s physician relied on specific recommendations for children 0 to 4 years of age to prescribe low-dose inhaled corticosteroids (ICS). Without the new guidelines, which underscore the safety of controller medication for young children, JJ’s physician would likely have been reluctant to place a 4-year-old on ICS.
This review highlights the EPR-3’s key recommendations to encourage widespread implementation by family physicians.
The EPR-3: What’s changed
The 2007 guidelines:
Recommend assessing asthma severity before starting treatment and assessing asthma control to guide adjustments in treatment.
Address both severity and control in terms of impairment and risk.
Feature 3 age breakdowns (0-4 years, 5-11 years, and ≥12 years) and a 6-step approach to asthma management.
Make it easier to individualize and adjust treatment.
What’s changed?
There’s a new paradigm
The 2007 update to guidelines released in 1997 and 2002 reflects a paradigm shift in the overall approach to asthma management. The change in focus addresses the highly variable nature of asthma2 and the recognition that asthma severity and asthma control are distinct concepts serving different functions in clinical practice.
Severity and control in 2 domains. Asthma severity—a measure of the intrinsic intensity of the disease process—is ideally assessed before initiating treatment. In contrast, asthma control is monitored over time to guide adjustments to therapy. The guidelines call for assessing severity and control within the domains of:
- impairment, based on asthma symptoms (identified by patient or caregiver recall of the past 2-4 weeks), quality of life, and functional limitations; and
- risk, of asthma exacerbations, progressive decline in pulmonary function (or reduced lung growth in children), or adverse events. Predictors of increased risk for exacerbations or death include persistent and/or severe airflow obstruction; at least 2 visits to the emergency department or hospitalizations for asthma within the past year; and a history of intubation or admission to intensive care, especially within the past 5 years.
The specific criteria for determining asthma severity and assessing asthma control are detailed in FIGURES 1 AND 2, respectively. Because treatment affects impairment and risk differently, this dual assessment helps ensure that therapeutic interventions minimize all manifestations of asthma as much as possible.
More steps and age-specific interventions. The EPR-3’s stepwise approach to asthma therapy has gone from 4 steps to 6, and the recommended treatments, as well as the levels of severity and criteria for assessing control that guide them, are now divided into 3 age groups: 0 to 4 years, 5 to 11 years, and ≥12 years (FIGURE 3). The previous guidelines, issued in 2002, divided treatment recommendations into 2 age groups: ≤5 years and >5 years. The EPR-3’s expansion makes it easier for physicians to initiate, individualize, and adjust treatment.
FIGURE 1
Classifying asthma severity and initiating therapy in children, adolescents, and adults
EIB, exercise-induced bronchospasm; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; ICS, inhaled corticosteroids; NA, not applicable; OCS, oral corticosteroids; SABA, short-acting β2-adrenergic agonist.
*Normal FEV1/FVC values are defined according to age: 8–9 years (85%), 20–39 years (80%), 40–59 years (75%), 60–80 years (70%).
†For treatment purposes, children with at least 2 exacerbations (eg, requiring urgent, unscheduled care; hospitalization; or intensive care unit admission) or adolescents/adults with at least 2 exacerbations requiring OCS in the past year may be considered the same as patients who have persistent asthma, even in the absence of impairment levels consistent with persistent asthma.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
FIGURE 2
Assessing asthma control and adjusting therapy
ACQ, Asthma Control Questionnaire; ACT, Asthma Control Test; ATAQ, Asthma Therapy Assessment Questionnaire; EIB, exercise-induced bronchospasm; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; N/A, not applicable; OCS, oral corticosteroids; SABA, short-acting β2-adrenergic agonist.
*ACQ values of 0.76 to 1.4 are indeterminate regarding well-controlled asthma.
†For treatment purposes, children with at least 2 exacerbations (eg, requiring urgent, unscheduled care; hospitalization; or intensive care unit admission) or adolescents/adults with at least 2 exacerbations requiring OCS in the past year may be considered the same as patients who have asthma that is not well controlled, even in the absence of impairment levels consistent with that classification.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
FIGURE 3
Stepwise approach for managing asthma
EIB, exercise-induced bronchospasm; ICS, inhaled corticosteroid; LABA, long-acting β2-adrenergic agonist; LTRA, leukotriene receptor antagonist; OCS, oral corticosteroid; PRN, as needed; SABA, short-acting β2-adrenergic agonist.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
Putting guidelines into practice begins with the history
A detailed medical history and a physical examination focusing on the upper respiratory tract, chest, and skin are needed to arrive at an asthma diagnosis. JJ’s physician asked his mother to describe recent symptoms and inquired about comorbid conditions that can aggravate asthma. He also identified viral respiratory infections, environmental causes, and activity as precipitating factors.
In considering an asthma diagnosis, try to determine the presence of episodic symptoms of airflow obstruction or bronchial hyperresponsiveness, as well as airflow obstruction that is at least partly reversible (an increase in forced expiratory volume in 1 second [FEV1] of >200 mL and ≥12% from baseline or an increase of ≥10% of predicted FEV1), and to exclude alternative diagnoses.
EPR-3 emphasizes spirometry
Recognizing that patients’ perception of airflow obstruction is highly variable and that pulmonary function measures do not always correlate directly with symptoms,3,4 the EPR-3 recommends spirometry for patients ≥5 years of age, both before and after bronchodilation. In addition to helping to confirm an asthma diagnosis, spirometry is the preferred measure of pulmonary function in classifying severity, because peak expiratory flow (PEF) testing has not proven reliable.5,6
Objective measurement of pulmonary function is difficult to obtain in children <5 years of age. If diagnosis remains uncertain for patients in this age group, a therapeutic trial of medication is recommended. In JJ’s case, however, 3 courses of oral corticosteroids (OCS) in less than 6 months were indicative of persistent asthma.
Spirometry is often underutilized. For patients ≥5 years of age, spirometry is a vital tool, but often underutilized in family practice. A recent study by Yawn and colleagues found that family physicians made changes in the management of approximately half of the asthma patients who underwent spirometry.7 (Information about spirometry training is available through the National Institute for Occupational Safety and Health at http://www.cdc.gov/niosh.) Referral to a specialist is recommended if the physician has difficulty making a differential diagnosis or is unable to perform spirometry on a patient who presents with atypical signs and symptoms of asthma.
What is the patient’s level of severity?
In patients who are not yet receiving long-term controller therapy, severity level is based on an assessment of impairment and risk (FIGURE 1). For patients who are already receiving treatment, severity is determined by the minimum pharmacologic therapy needed to maintain asthma control.
The severity classification—intermittent asthma or persistent asthma that is mild, moderate, or severe—is determined by the most severe category in which any feature occurs. (In children, FEV1/FVC [forced vital capacity] ratio has been shown to be a more sensitive determinant of severity than FEV1,4 which may be more useful in predicting exacerbations.8)
Asthma management: Preferred and alternative Tx
The recommended stepwise interventions include both preferred therapies (evidence-based) and alternative treatments (listed alphabetically in FIGURE 3 because there is insufficient evidence to rank them). The additional steps and age categories support the goal of using the least possible medication needed to maintain good control and minimize the potential for adverse events.
In initiating treatment, select the step that corresponds to the level of severity in the bottom row of FIGURE 1; to adjust medications, determine the patient’s level of asthma control and follow the corresponding guidance in the bottom row of FIGURE 2.
Inhaled corticosteroids remain the bedrock of therapy
ICS, the most potent and consistently effective long-term controller therapy, remain the foundation of therapy for patients of all ages who have persistent asthma. (Evidence: A).
Several of the age-based recommendations follow, with a focus on preferred treatments:
Children 0 to 4 years of age
- The guidelines recommend low-dose ICS at Step 2 (Evidence: A) and medium-dose ICS at Step 3 (Evidence: D), as inhaled corticosteroids have been shown to reduce impairment and risk in this age group.9-16 The potential risk is generally limited to a small reduction in growth velocity during the first year of treatment, and offset by the benefits of therapy.15,16
- Add a long-acting β2-adrenergic agonist (LABA) or montelukast to medium-dose ICS therapy at Step 4 rather than increasing the ICS dose (Evidence: D) to avoid the risk of side effects associated with high-dose ICS. Montelukast has demonstrated efficacy in children 2 to 5 years of age with persistent asthma.17
- Recommendations for preferred therapy at Steps 5 (high-dose ICS + LABA or montelukast) and 6 (Step 5 therapy + OCS) are based on expert panel judgment (Evidence: D). When severe persistent asthma warrants Step 6 therapy, start with a 2-week course of the lowest possible dose of OCS to confirm reversibility.
- In this age group, a therapeutic trial with close monitoring is recommended for patients whose asthma is not well controlled. If there is no response in 4 to 6 weeks, consider alternative therapies or diagnoses (Evidence: D).
Children 5 to 11 years of age
- For Step 3 therapy, the guidelines recommend either low-dose ICS plus a LABA, leukotriene receptor antagonist (LTRA), or theophylline; or medium-dose ICS (Evidence: B). Treatment decisions at Step 3 depend on whether impairment or risk is the chief concern, as well as on safety considerations.
- For Steps 4 and 5, ICS (medium dose for Step 5 and high dose for Step 6) plus a LABA is preferred, based on studies of patients ≥12 years of age (Evidence: B). Step 6 builds on Step 5, adding an OCS to the LABA/ICS combination (Evidence: D).
- If theophylline is prescribed—a viable option if cost and adherence to inhaled medications are key concerns—serum levels must be closely monitored because of the risk of toxicity.
- Closely monitor and be prepared to identify and respond to anaphylaxis in a child at Step 2, 3, or 4 who is receiving allergen immunotherapy.
Adolescents ≥12 years of age and adults
- There are 2 preferred Step 3 treatments: Low-dose ICS plus a LABA, or medium-dose ICS. The combination therapy has shown greater improvement in impairment24,25 and risk24-26 compared with the higher dose of ICS.
- Preferred treatments at Steps 4, 5, and 6 are the same as those for children ages 5 to 11 years, with one exception: Subcutaneous anti-IgE therapy (omalizumab) may be added to the regimen at Steps 5 and 6 for adolescents and adults with severe persistent allergic asthma to reduce the risk of exacerbations.27
Weigh the benefits and risks of therapy
Safety is a key consideration for all asthma patients. Carefully weigh the benefits and risks of therapy, including the rare but potential risk of life-threatening or fatal exacerbations with daily LABA therapy28 and systemic effects with higher doses of ICS.23 Patients who begin receiving oral corticosteroids require close monitoring, regardless of age.
Regular reassessment and monitoring are critical
Schedule visits at 2- to 6-week intervals for those who are starting therapy or require a step up to achieve or regain asthma control. After control is achieved, reassess at least every 1 to 6 months. Measures of asthma control are the same as those used to assess severity, with the addition of validated multidimensional questionnaires (eg, Asthma Control Test [ACT])29 to gauge impairment.
JJ’s physician scheduled a follow-up visit in 4 weeks, at which time he did a reassessment based on a physical exam and symptom recall. Finding JJ’s asthma to be well controlled, the physician asked the boy’s mother to bring him back to the office in 2 months, or earlier if symptoms recurred.
TABLE W1
Asthma education resources
| Allergy & Asthma Network Mothers of Asthmatics 2751 Prosperity Avenue, Suite 150 Fairfax, VA 22030 www.breatherville.org (800) 878-4403 or (703) 641-9595 | Asthma and Allergy Foundation of America 1233 20th Street, NW, Suite 402 Washington, DC 20036 www.aafa.org (800) 727-8462 |
| American Academy of Allergy, Asthma, and Immunology 555 East Wells Street, Suite 1100 Milwaukee, WI 53202-3823 www.aaaai.org (414) 272-6071 | Centers for Disease Control and Prevention 1600 Clifton Road Atlanta, GA 30333 www.cdc.gov (800) 311-3435 |
| American Association for Respiratory Care 9125 North macArthur boulevard, Suite 100 Irving, TX 75063 www.aarc.org (972) 243-2272 | Food Allergy & Anaphylaxis Network 11781 lee Jackson Highway, Suite 160 Fairfax, VA 22033 www.foodallergy.org (800) 929-4040 |
| American College of Allergy, Asthma, and Immunology 85 West Algonquin road, Suite 550 Arlington Heights, IL 60005 www.acaai.org (800) 842-7777 or (847) 427-1200 | National Heart, Lung, and Blood Institute Information Center P.O. Box 30105 Bethesda, MD 20824-0105 www.nhlbi.nih.gov (301) 592-8573 |
| American Lung Association 61 Broadway New York, NY 10006 www.lungusa.org (800) 586-4872 | National Jewish Medical and Research Center (Lung Line) 1400 Jackson Street Denver, CO 80206 www.njc.org (800) 222-lUNG |
| Association of Asthma Educators 1215 Anthony Avenue Columbia, SC 29201 www.asthmaeducators.org (888) 988-7747 | US Environmental Protection Agency National Center for Environmental Publications P.O. Box 42419 Cincinnati, OH 45242-0419 www.airnow.gov (800) 490-9198 |
Does your patient require a step down or step up?
A step down is recommended for patients whose asthma is well controlled for 3 months or more. Reduce the dose of ICS gradually, about 25% to 50% every 3 months, because deterioration in asthma control is highly variable. Review adherence and medication administration technique with patients whose asthma is not well controlled, and consider a step up in treatment. If an alternative treatment is used but does not result in an adequate response, it should be discontinued and the preferred treatment used before stepping up. Refer patients to an asthma specialist if their asthma does not respond to these adjustments.
Partner with patients for optimal care
The EPR-3 recommends the integration of patient education into all aspects of asthma care. To forge an active partnership, identify and address concerns about the condition and its treatment and involve the patient and family in developing treatment goals and making treatment decisions. If the patient is old enough, encourage self-monitoring and management.
The EPR-3 recommends that physicians give every patient a written asthma action plan that addresses individual symptoms and/or PEF measurements and includes instructions for self-management. Daily PEF monitoring can be useful in identifying early changes in the disease state and evaluating response to changes in therapy. It is ideal for those who have moderate to severe persistent asthma, difficulty recognizing signs of exacerbations, or a history of severe exacerbations.
Correspondence
Stuart W. Stoloff, MD, Clinical Professor, Department of Family and Community Medicine, University of Nevada–Reno, 1200 Mountain Street, Suite 220, Carson City, NV 89703; drstoloff@sbcglobal.net.
1. National Center for Health Statistics. Fast stats A to Z. Available at: www.cdc.gov/nchs/fastats/asthma.htm. Accessed August 1, 2008.
2. National Heart, Lung, and Blood Institute (NHLBI). National Asthma Education and Prevention Program Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma. Full Report 2007. Bethesda, MD: NHLBI; August 2007. NIH publication no. 07-4051. Available at: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm. Accessed July 17, 2008.
3. Stout JW, Visness CM, Enright P, et al. Classification of asthma severity in children: the contribution of pulmonary function testing. Arch Pediatr Adolesc Med. 2006;160:844-850.
4. Bacharier LB, Strunk RC, Mauger D, et al. Classifying asthma severity in children: mismatch between symptoms, medication use, and lung function. Am J Respir Crit Care Med. 2004;170:426-432.
5. Eid N, Yandell B, Howell L, Eddy M, Sheikh S. Can children with asthma? Pediatrics. 2000;105:354-358.
6. Llewellin P, Sawyer G, Lewis S, et al. The relationship between FEV1 and PEF in the assessment of the severity of airways obstruction. Respirology. 2002;7:333-337.
7. Yawn BP, Enright PL, Lemanske RF, Jr, et al. Spirometry can be done in family physicians’ offices and alters clinical decisions in management of asthma and COPD. Chest. 2007;132:1162-1168.
8. Fuhlbrigge AL, Kitch BT, Paltiel AD, et al. FEV1 is associated with risk of asthma attacks in a pediatric population. J Allergy Clin Immunol. 2001;107:61-67.
9. Roorda RJ, Mezei G, Bisgaard H, Maden C. Response of preschool children with asthma symptoms to fluticasone propionate. J Allergy Clin Immunol. 2001;108:540-546.
10. Baker JW, Mellon M, Wald J, Welch M, Cruz-Rivera M, Walton-Bowen K. A multiple-dosing, placebo-controlled study of budesonide inhalation suspension given once or twice daily for treatment of persistent asthma in young children and infants. Pediatrics. 1999;103:414-421.
11. Kemp JP, Skoner DP, Szefler SJ, Walton-Bowen K, Cruz-Rivera M, Smith JA. Once-daily budesonide inhalation suspension for the treatment of persistent asthma in infants and young children. Ann Allergy Asthma Immunol. 1999;83:231-239.
12. Shapiro G, Mendelson L, Kraemer MJ, Cruz-Rivera M, Walton-Bowen K, Smith JA. Efficacy and safety of budesonide inhalation suspension (Pulmicort Respules) in young children with inhaled steroid-dependent, persistent asthma. J Allergy Clin Immunol. 1998;102:789-796.
13. Bisgaard H, Gillies J, Groenewald M, Maden C. The effect of inhaled fluticasone propionate in the treatment of young asthmatic children: a dose comparison study. Am J Respir Crit Care Med. 1999;160:126-131.
14. Szefler SJ, Eigen H. Budesonide inhalation suspension: a nebulized corticosteroid for persistent asthma. J Allergy Clin Immunol. 2002;109:730-742.
15. Guilbert TW, Morgan WJ, Zeiger RS, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med. 2006;354:1985-1997.
16. Bisgaard H, Allen D, Milanowski J, Kalev I, Willits L, Davies P. Twelve-month safety and efficacy of inhaled fluticasone propionate in children aged 1 to 3 years with recurrent wheezing. Pediatrics. 2004;113:e87-e94.
17. Knorr B, Franchi LM, Bisgaard H, et al. Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years. Pediatrics 2001;108:e48.-
18. Russell G, Williams DA, Weller P, Price JF. Salmeterol xinafoate in children on high dose inhaled steroids. Ann Allergy Asthma Immunol. 1995;75:423-428.
19. Zimmerman B, D’Urzo A, Bérubé D. Efficacy and safety of formoterol Turbuhaler when added to inhaled corticosteroid treatment in children with asthma. Pediatr Pulmonol. 2004;37:122-127.
20. Simons FE, Villa JR, Lee BW, et al. Montelukast added to budesonide in children with persistent asthma: a randomized, double-blind, crossover study. J Pediatr. 2001;138:694-698.
21. Shapiro G, Bronsky EA, LaForce CF, et al. Dose-related efficacy of budesonide administered via a dry powder inhaler in the treatment of children with moderate to severe persistent asthma. J Pediatr. 1998;132:976-982.
22. Pauwels RA, Lofdahl C-G, Postma DS, et al. for the Formoterol and Corticosteroids Establishing Therapy (FACET) International Study Group. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med. 1997;337:1405-1411.
23. Tattersfield AE, Harrison TW, Hubbard RB, Mortimer K. Safety of inhaled corticosteroids. Proc Am Thorac Soc. 2004;1:171-175.
24. Bateman ED, Boushey HA, Bousquet J, et al. For the GOAL Investigators Group. Can guideline-defined asthma control be achieved? The Gaining Optimal Asthma ControL study. Am J Respir Crit Care Med. 2004;170:836-844.
25. O’Byrne PM, Barnes PJ, Rodriguez-Roisin R, et al. Low dose inhaled budesonide and formoterol in mild persistent asthma: the OPTIMA randomized trial. Am J Respir Crit Care Med. 2001;164:1392-1397.
26. Masoli M, Weatherall M, Holt S, Beasley R. Moderate dose inhaled corticosteroids plus salmeterol versus higher doses of inhaled corticosteroids in symptomatic asthma. Thorax. 2005;60:730-734.
27. Bousquet J, Wenzel S, Holgate S, Lumry W, Freeman P, Fox H. Predicting response to omalizumab, an anti-IgE antibody, in patients with allergic asthma. Chest. 2004;125:1378-1386.
28. Nelson HS, Weiss ST, Bleecker ER, Yancey SW, Dorinsky PM. For the SMART Study Group. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest. 2006;129:15-26.
29. Nathan RA, Sorkness CA, Kosinski M, et al. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol. 2004;113:59-65.
- Assess asthma severity before initiating treatment; monitor asthma control to guide adjustments in therapy using measures of impairment (B) and risk (C) (National Heart, Lung, and Blood Institute [NHLBI] and National Asthma Education and Prevention Program [NAEPP] third expert panel report [EPR-3]).
- Base treatment decisions on recommendations specific to each age group (0-4 years, 5-11 years, and ≥12 years) (A).
- Use spirometry in patients ≥5 years of age to diagnose asthma, classify severity, and assess control (C).
- Provide each patient with a written asthma action plan with instructions for daily disease management, as well as identification of, and response to, worsening symptoms (B).
EPR-3 evidence categories:
- Randomized, controlled trials (RCTs), rich body of data
- RCTs, limited body of data
- Nonrandomized trials and observational studies
- Panel consensus judgment
JJ, a 4-year-old boy, was taken to an urgent care clinic 3 times last winter for “recurrent bronchitis” and given a 7-day course of prednisone and antibiotics at each visit. His mother reports that “his colds always seem to go to his chest” and his skin is always dry. She was given a nebulizer and albuterol for use when JJ begins wheezing, which often happens when he has a cold, plays vigorously, or visits a friend who has cats.
JJ is one of approximately 6.7 million children—and 22.9 million US residents—who have asthma.1 To help guide the care of patients like JJ, the National Heart, Lung, and Blood Institute (NHLBI) and National Asthma Education and Prevention Program (NAEPP) released the third expert panel report (EPR-3) in 2007. Available at http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm, the EPR-3 provides the most comprehensive evidence-based guidance for the diagnosis and management of asthma to date.2
The guidelines were an invaluable resource for JJ’s family physician, who referred to them to categorize the severity of JJ’s asthma as “mild persistent.” In initiating treatment, JJ’s physician relied on specific recommendations for children 0 to 4 years of age to prescribe low-dose inhaled corticosteroids (ICS). Without the new guidelines, which underscore the safety of controller medication for young children, JJ’s physician would likely have been reluctant to place a 4-year-old on ICS.
This review highlights the EPR-3’s key recommendations to encourage widespread implementation by family physicians.
The EPR-3: What’s changed
The 2007 guidelines:
Recommend assessing asthma severity before starting treatment and assessing asthma control to guide adjustments in treatment.
Address both severity and control in terms of impairment and risk.
Feature 3 age breakdowns (0-4 years, 5-11 years, and ≥12 years) and a 6-step approach to asthma management.
Make it easier to individualize and adjust treatment.
What’s changed?
There’s a new paradigm
The 2007 update to guidelines released in 1997 and 2002 reflects a paradigm shift in the overall approach to asthma management. The change in focus addresses the highly variable nature of asthma2 and the recognition that asthma severity and asthma control are distinct concepts serving different functions in clinical practice.
Severity and control in 2 domains. Asthma severity—a measure of the intrinsic intensity of the disease process—is ideally assessed before initiating treatment. In contrast, asthma control is monitored over time to guide adjustments to therapy. The guidelines call for assessing severity and control within the domains of:
- impairment, based on asthma symptoms (identified by patient or caregiver recall of the past 2-4 weeks), quality of life, and functional limitations; and
- risk, of asthma exacerbations, progressive decline in pulmonary function (or reduced lung growth in children), or adverse events. Predictors of increased risk for exacerbations or death include persistent and/or severe airflow obstruction; at least 2 visits to the emergency department or hospitalizations for asthma within the past year; and a history of intubation or admission to intensive care, especially within the past 5 years.
The specific criteria for determining asthma severity and assessing asthma control are detailed in FIGURES 1 AND 2, respectively. Because treatment affects impairment and risk differently, this dual assessment helps ensure that therapeutic interventions minimize all manifestations of asthma as much as possible.
More steps and age-specific interventions. The EPR-3’s stepwise approach to asthma therapy has gone from 4 steps to 6, and the recommended treatments, as well as the levels of severity and criteria for assessing control that guide them, are now divided into 3 age groups: 0 to 4 years, 5 to 11 years, and ≥12 years (FIGURE 3). The previous guidelines, issued in 2002, divided treatment recommendations into 2 age groups: ≤5 years and >5 years. The EPR-3’s expansion makes it easier for physicians to initiate, individualize, and adjust treatment.
FIGURE 1
Classifying asthma severity and initiating therapy in children, adolescents, and adults
EIB, exercise-induced bronchospasm; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; ICS, inhaled corticosteroids; NA, not applicable; OCS, oral corticosteroids; SABA, short-acting β2-adrenergic agonist.
*Normal FEV1/FVC values are defined according to age: 8–9 years (85%), 20–39 years (80%), 40–59 years (75%), 60–80 years (70%).
†For treatment purposes, children with at least 2 exacerbations (eg, requiring urgent, unscheduled care; hospitalization; or intensive care unit admission) or adolescents/adults with at least 2 exacerbations requiring OCS in the past year may be considered the same as patients who have persistent asthma, even in the absence of impairment levels consistent with persistent asthma.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
FIGURE 2
Assessing asthma control and adjusting therapy
ACQ, Asthma Control Questionnaire; ACT, Asthma Control Test; ATAQ, Asthma Therapy Assessment Questionnaire; EIB, exercise-induced bronchospasm; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; N/A, not applicable; OCS, oral corticosteroids; SABA, short-acting β2-adrenergic agonist.
*ACQ values of 0.76 to 1.4 are indeterminate regarding well-controlled asthma.
†For treatment purposes, children with at least 2 exacerbations (eg, requiring urgent, unscheduled care; hospitalization; or intensive care unit admission) or adolescents/adults with at least 2 exacerbations requiring OCS in the past year may be considered the same as patients who have asthma that is not well controlled, even in the absence of impairment levels consistent with that classification.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
FIGURE 3
Stepwise approach for managing asthma
EIB, exercise-induced bronchospasm; ICS, inhaled corticosteroid; LABA, long-acting β2-adrenergic agonist; LTRA, leukotriene receptor antagonist; OCS, oral corticosteroid; PRN, as needed; SABA, short-acting β2-adrenergic agonist.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
Putting guidelines into practice begins with the history
A detailed medical history and a physical examination focusing on the upper respiratory tract, chest, and skin are needed to arrive at an asthma diagnosis. JJ’s physician asked his mother to describe recent symptoms and inquired about comorbid conditions that can aggravate asthma. He also identified viral respiratory infections, environmental causes, and activity as precipitating factors.
In considering an asthma diagnosis, try to determine the presence of episodic symptoms of airflow obstruction or bronchial hyperresponsiveness, as well as airflow obstruction that is at least partly reversible (an increase in forced expiratory volume in 1 second [FEV1] of >200 mL and ≥12% from baseline or an increase of ≥10% of predicted FEV1), and to exclude alternative diagnoses.
EPR-3 emphasizes spirometry
Recognizing that patients’ perception of airflow obstruction is highly variable and that pulmonary function measures do not always correlate directly with symptoms,3,4 the EPR-3 recommends spirometry for patients ≥5 years of age, both before and after bronchodilation. In addition to helping to confirm an asthma diagnosis, spirometry is the preferred measure of pulmonary function in classifying severity, because peak expiratory flow (PEF) testing has not proven reliable.5,6
Objective measurement of pulmonary function is difficult to obtain in children <5 years of age. If diagnosis remains uncertain for patients in this age group, a therapeutic trial of medication is recommended. In JJ’s case, however, 3 courses of oral corticosteroids (OCS) in less than 6 months were indicative of persistent asthma.
Spirometry is often underutilized. For patients ≥5 years of age, spirometry is a vital tool, but often underutilized in family practice. A recent study by Yawn and colleagues found that family physicians made changes in the management of approximately half of the asthma patients who underwent spirometry.7 (Information about spirometry training is available through the National Institute for Occupational Safety and Health at http://www.cdc.gov/niosh.) Referral to a specialist is recommended if the physician has difficulty making a differential diagnosis or is unable to perform spirometry on a patient who presents with atypical signs and symptoms of asthma.
What is the patient’s level of severity?
In patients who are not yet receiving long-term controller therapy, severity level is based on an assessment of impairment and risk (FIGURE 1). For patients who are already receiving treatment, severity is determined by the minimum pharmacologic therapy needed to maintain asthma control.
The severity classification—intermittent asthma or persistent asthma that is mild, moderate, or severe—is determined by the most severe category in which any feature occurs. (In children, FEV1/FVC [forced vital capacity] ratio has been shown to be a more sensitive determinant of severity than FEV1,4 which may be more useful in predicting exacerbations.8)
Asthma management: Preferred and alternative Tx
The recommended stepwise interventions include both preferred therapies (evidence-based) and alternative treatments (listed alphabetically in FIGURE 3 because there is insufficient evidence to rank them). The additional steps and age categories support the goal of using the least possible medication needed to maintain good control and minimize the potential for adverse events.
In initiating treatment, select the step that corresponds to the level of severity in the bottom row of FIGURE 1; to adjust medications, determine the patient’s level of asthma control and follow the corresponding guidance in the bottom row of FIGURE 2.
Inhaled corticosteroids remain the bedrock of therapy
ICS, the most potent and consistently effective long-term controller therapy, remain the foundation of therapy for patients of all ages who have persistent asthma. (Evidence: A).
Several of the age-based recommendations follow, with a focus on preferred treatments:
Children 0 to 4 years of age
- The guidelines recommend low-dose ICS at Step 2 (Evidence: A) and medium-dose ICS at Step 3 (Evidence: D), as inhaled corticosteroids have been shown to reduce impairment and risk in this age group.9-16 The potential risk is generally limited to a small reduction in growth velocity during the first year of treatment, and offset by the benefits of therapy.15,16
- Add a long-acting β2-adrenergic agonist (LABA) or montelukast to medium-dose ICS therapy at Step 4 rather than increasing the ICS dose (Evidence: D) to avoid the risk of side effects associated with high-dose ICS. Montelukast has demonstrated efficacy in children 2 to 5 years of age with persistent asthma.17
- Recommendations for preferred therapy at Steps 5 (high-dose ICS + LABA or montelukast) and 6 (Step 5 therapy + OCS) are based on expert panel judgment (Evidence: D). When severe persistent asthma warrants Step 6 therapy, start with a 2-week course of the lowest possible dose of OCS to confirm reversibility.
- In this age group, a therapeutic trial with close monitoring is recommended for patients whose asthma is not well controlled. If there is no response in 4 to 6 weeks, consider alternative therapies or diagnoses (Evidence: D).
Children 5 to 11 years of age
- For Step 3 therapy, the guidelines recommend either low-dose ICS plus a LABA, leukotriene receptor antagonist (LTRA), or theophylline; or medium-dose ICS (Evidence: B). Treatment decisions at Step 3 depend on whether impairment or risk is the chief concern, as well as on safety considerations.
- For Steps 4 and 5, ICS (medium dose for Step 5 and high dose for Step 6) plus a LABA is preferred, based on studies of patients ≥12 years of age (Evidence: B). Step 6 builds on Step 5, adding an OCS to the LABA/ICS combination (Evidence: D).
- If theophylline is prescribed—a viable option if cost and adherence to inhaled medications are key concerns—serum levels must be closely monitored because of the risk of toxicity.
- Closely monitor and be prepared to identify and respond to anaphylaxis in a child at Step 2, 3, or 4 who is receiving allergen immunotherapy.
Adolescents ≥12 years of age and adults
- There are 2 preferred Step 3 treatments: Low-dose ICS plus a LABA, or medium-dose ICS. The combination therapy has shown greater improvement in impairment24,25 and risk24-26 compared with the higher dose of ICS.
- Preferred treatments at Steps 4, 5, and 6 are the same as those for children ages 5 to 11 years, with one exception: Subcutaneous anti-IgE therapy (omalizumab) may be added to the regimen at Steps 5 and 6 for adolescents and adults with severe persistent allergic asthma to reduce the risk of exacerbations.27
Weigh the benefits and risks of therapy
Safety is a key consideration for all asthma patients. Carefully weigh the benefits and risks of therapy, including the rare but potential risk of life-threatening or fatal exacerbations with daily LABA therapy28 and systemic effects with higher doses of ICS.23 Patients who begin receiving oral corticosteroids require close monitoring, regardless of age.
Regular reassessment and monitoring are critical
Schedule visits at 2- to 6-week intervals for those who are starting therapy or require a step up to achieve or regain asthma control. After control is achieved, reassess at least every 1 to 6 months. Measures of asthma control are the same as those used to assess severity, with the addition of validated multidimensional questionnaires (eg, Asthma Control Test [ACT])29 to gauge impairment.
JJ’s physician scheduled a follow-up visit in 4 weeks, at which time he did a reassessment based on a physical exam and symptom recall. Finding JJ’s asthma to be well controlled, the physician asked the boy’s mother to bring him back to the office in 2 months, or earlier if symptoms recurred.
TABLE W1
Asthma education resources
| Allergy & Asthma Network Mothers of Asthmatics 2751 Prosperity Avenue, Suite 150 Fairfax, VA 22030 www.breatherville.org (800) 878-4403 or (703) 641-9595 | Asthma and Allergy Foundation of America 1233 20th Street, NW, Suite 402 Washington, DC 20036 www.aafa.org (800) 727-8462 |
| American Academy of Allergy, Asthma, and Immunology 555 East Wells Street, Suite 1100 Milwaukee, WI 53202-3823 www.aaaai.org (414) 272-6071 | Centers for Disease Control and Prevention 1600 Clifton Road Atlanta, GA 30333 www.cdc.gov (800) 311-3435 |
| American Association for Respiratory Care 9125 North macArthur boulevard, Suite 100 Irving, TX 75063 www.aarc.org (972) 243-2272 | Food Allergy & Anaphylaxis Network 11781 lee Jackson Highway, Suite 160 Fairfax, VA 22033 www.foodallergy.org (800) 929-4040 |
| American College of Allergy, Asthma, and Immunology 85 West Algonquin road, Suite 550 Arlington Heights, IL 60005 www.acaai.org (800) 842-7777 or (847) 427-1200 | National Heart, Lung, and Blood Institute Information Center P.O. Box 30105 Bethesda, MD 20824-0105 www.nhlbi.nih.gov (301) 592-8573 |
| American Lung Association 61 Broadway New York, NY 10006 www.lungusa.org (800) 586-4872 | National Jewish Medical and Research Center (Lung Line) 1400 Jackson Street Denver, CO 80206 www.njc.org (800) 222-lUNG |
| Association of Asthma Educators 1215 Anthony Avenue Columbia, SC 29201 www.asthmaeducators.org (888) 988-7747 | US Environmental Protection Agency National Center for Environmental Publications P.O. Box 42419 Cincinnati, OH 45242-0419 www.airnow.gov (800) 490-9198 |
Does your patient require a step down or step up?
A step down is recommended for patients whose asthma is well controlled for 3 months or more. Reduce the dose of ICS gradually, about 25% to 50% every 3 months, because deterioration in asthma control is highly variable. Review adherence and medication administration technique with patients whose asthma is not well controlled, and consider a step up in treatment. If an alternative treatment is used but does not result in an adequate response, it should be discontinued and the preferred treatment used before stepping up. Refer patients to an asthma specialist if their asthma does not respond to these adjustments.
Partner with patients for optimal care
The EPR-3 recommends the integration of patient education into all aspects of asthma care. To forge an active partnership, identify and address concerns about the condition and its treatment and involve the patient and family in developing treatment goals and making treatment decisions. If the patient is old enough, encourage self-monitoring and management.
The EPR-3 recommends that physicians give every patient a written asthma action plan that addresses individual symptoms and/or PEF measurements and includes instructions for self-management. Daily PEF monitoring can be useful in identifying early changes in the disease state and evaluating response to changes in therapy. It is ideal for those who have moderate to severe persistent asthma, difficulty recognizing signs of exacerbations, or a history of severe exacerbations.
Correspondence
Stuart W. Stoloff, MD, Clinical Professor, Department of Family and Community Medicine, University of Nevada–Reno, 1200 Mountain Street, Suite 220, Carson City, NV 89703; drstoloff@sbcglobal.net.
- Assess asthma severity before initiating treatment; monitor asthma control to guide adjustments in therapy using measures of impairment (B) and risk (C) (National Heart, Lung, and Blood Institute [NHLBI] and National Asthma Education and Prevention Program [NAEPP] third expert panel report [EPR-3]).
- Base treatment decisions on recommendations specific to each age group (0-4 years, 5-11 years, and ≥12 years) (A).
- Use spirometry in patients ≥5 years of age to diagnose asthma, classify severity, and assess control (C).
- Provide each patient with a written asthma action plan with instructions for daily disease management, as well as identification of, and response to, worsening symptoms (B).
EPR-3 evidence categories:
- Randomized, controlled trials (RCTs), rich body of data
- RCTs, limited body of data
- Nonrandomized trials and observational studies
- Panel consensus judgment
JJ, a 4-year-old boy, was taken to an urgent care clinic 3 times last winter for “recurrent bronchitis” and given a 7-day course of prednisone and antibiotics at each visit. His mother reports that “his colds always seem to go to his chest” and his skin is always dry. She was given a nebulizer and albuterol for use when JJ begins wheezing, which often happens when he has a cold, plays vigorously, or visits a friend who has cats.
JJ is one of approximately 6.7 million children—and 22.9 million US residents—who have asthma.1 To help guide the care of patients like JJ, the National Heart, Lung, and Blood Institute (NHLBI) and National Asthma Education and Prevention Program (NAEPP) released the third expert panel report (EPR-3) in 2007. Available at http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm, the EPR-3 provides the most comprehensive evidence-based guidance for the diagnosis and management of asthma to date.2
The guidelines were an invaluable resource for JJ’s family physician, who referred to them to categorize the severity of JJ’s asthma as “mild persistent.” In initiating treatment, JJ’s physician relied on specific recommendations for children 0 to 4 years of age to prescribe low-dose inhaled corticosteroids (ICS). Without the new guidelines, which underscore the safety of controller medication for young children, JJ’s physician would likely have been reluctant to place a 4-year-old on ICS.
This review highlights the EPR-3’s key recommendations to encourage widespread implementation by family physicians.
The EPR-3: What’s changed
The 2007 guidelines:
Recommend assessing asthma severity before starting treatment and assessing asthma control to guide adjustments in treatment.
Address both severity and control in terms of impairment and risk.
Feature 3 age breakdowns (0-4 years, 5-11 years, and ≥12 years) and a 6-step approach to asthma management.
Make it easier to individualize and adjust treatment.
What’s changed?
There’s a new paradigm
The 2007 update to guidelines released in 1997 and 2002 reflects a paradigm shift in the overall approach to asthma management. The change in focus addresses the highly variable nature of asthma2 and the recognition that asthma severity and asthma control are distinct concepts serving different functions in clinical practice.
Severity and control in 2 domains. Asthma severity—a measure of the intrinsic intensity of the disease process—is ideally assessed before initiating treatment. In contrast, asthma control is monitored over time to guide adjustments to therapy. The guidelines call for assessing severity and control within the domains of:
- impairment, based on asthma symptoms (identified by patient or caregiver recall of the past 2-4 weeks), quality of life, and functional limitations; and
- risk, of asthma exacerbations, progressive decline in pulmonary function (or reduced lung growth in children), or adverse events. Predictors of increased risk for exacerbations or death include persistent and/or severe airflow obstruction; at least 2 visits to the emergency department or hospitalizations for asthma within the past year; and a history of intubation or admission to intensive care, especially within the past 5 years.
The specific criteria for determining asthma severity and assessing asthma control are detailed in FIGURES 1 AND 2, respectively. Because treatment affects impairment and risk differently, this dual assessment helps ensure that therapeutic interventions minimize all manifestations of asthma as much as possible.
More steps and age-specific interventions. The EPR-3’s stepwise approach to asthma therapy has gone from 4 steps to 6, and the recommended treatments, as well as the levels of severity and criteria for assessing control that guide them, are now divided into 3 age groups: 0 to 4 years, 5 to 11 years, and ≥12 years (FIGURE 3). The previous guidelines, issued in 2002, divided treatment recommendations into 2 age groups: ≤5 years and >5 years. The EPR-3’s expansion makes it easier for physicians to initiate, individualize, and adjust treatment.
FIGURE 1
Classifying asthma severity and initiating therapy in children, adolescents, and adults
EIB, exercise-induced bronchospasm; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; ICS, inhaled corticosteroids; NA, not applicable; OCS, oral corticosteroids; SABA, short-acting β2-adrenergic agonist.
*Normal FEV1/FVC values are defined according to age: 8–9 years (85%), 20–39 years (80%), 40–59 years (75%), 60–80 years (70%).
†For treatment purposes, children with at least 2 exacerbations (eg, requiring urgent, unscheduled care; hospitalization; or intensive care unit admission) or adolescents/adults with at least 2 exacerbations requiring OCS in the past year may be considered the same as patients who have persistent asthma, even in the absence of impairment levels consistent with persistent asthma.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
FIGURE 2
Assessing asthma control and adjusting therapy
ACQ, Asthma Control Questionnaire; ACT, Asthma Control Test; ATAQ, Asthma Therapy Assessment Questionnaire; EIB, exercise-induced bronchospasm; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; N/A, not applicable; OCS, oral corticosteroids; SABA, short-acting β2-adrenergic agonist.
*ACQ values of 0.76 to 1.4 are indeterminate regarding well-controlled asthma.
†For treatment purposes, children with at least 2 exacerbations (eg, requiring urgent, unscheduled care; hospitalization; or intensive care unit admission) or adolescents/adults with at least 2 exacerbations requiring OCS in the past year may be considered the same as patients who have asthma that is not well controlled, even in the absence of impairment levels consistent with that classification.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
FIGURE 3
Stepwise approach for managing asthma
EIB, exercise-induced bronchospasm; ICS, inhaled corticosteroid; LABA, long-acting β2-adrenergic agonist; LTRA, leukotriene receptor antagonist; OCS, oral corticosteroid; PRN, as needed; SABA, short-acting β2-adrenergic agonist.
Adapted from: National Heart, Lung, and Blood Institute (NHLBI).2
Putting guidelines into practice begins with the history
A detailed medical history and a physical examination focusing on the upper respiratory tract, chest, and skin are needed to arrive at an asthma diagnosis. JJ’s physician asked his mother to describe recent symptoms and inquired about comorbid conditions that can aggravate asthma. He also identified viral respiratory infections, environmental causes, and activity as precipitating factors.
In considering an asthma diagnosis, try to determine the presence of episodic symptoms of airflow obstruction or bronchial hyperresponsiveness, as well as airflow obstruction that is at least partly reversible (an increase in forced expiratory volume in 1 second [FEV1] of >200 mL and ≥12% from baseline or an increase of ≥10% of predicted FEV1), and to exclude alternative diagnoses.
EPR-3 emphasizes spirometry
Recognizing that patients’ perception of airflow obstruction is highly variable and that pulmonary function measures do not always correlate directly with symptoms,3,4 the EPR-3 recommends spirometry for patients ≥5 years of age, both before and after bronchodilation. In addition to helping to confirm an asthma diagnosis, spirometry is the preferred measure of pulmonary function in classifying severity, because peak expiratory flow (PEF) testing has not proven reliable.5,6
Objective measurement of pulmonary function is difficult to obtain in children <5 years of age. If diagnosis remains uncertain for patients in this age group, a therapeutic trial of medication is recommended. In JJ’s case, however, 3 courses of oral corticosteroids (OCS) in less than 6 months were indicative of persistent asthma.
Spirometry is often underutilized. For patients ≥5 years of age, spirometry is a vital tool, but often underutilized in family practice. A recent study by Yawn and colleagues found that family physicians made changes in the management of approximately half of the asthma patients who underwent spirometry.7 (Information about spirometry training is available through the National Institute for Occupational Safety and Health at http://www.cdc.gov/niosh.) Referral to a specialist is recommended if the physician has difficulty making a differential diagnosis or is unable to perform spirometry on a patient who presents with atypical signs and symptoms of asthma.
What is the patient’s level of severity?
In patients who are not yet receiving long-term controller therapy, severity level is based on an assessment of impairment and risk (FIGURE 1). For patients who are already receiving treatment, severity is determined by the minimum pharmacologic therapy needed to maintain asthma control.
The severity classification—intermittent asthma or persistent asthma that is mild, moderate, or severe—is determined by the most severe category in which any feature occurs. (In children, FEV1/FVC [forced vital capacity] ratio has been shown to be a more sensitive determinant of severity than FEV1,4 which may be more useful in predicting exacerbations.8)
Asthma management: Preferred and alternative Tx
The recommended stepwise interventions include both preferred therapies (evidence-based) and alternative treatments (listed alphabetically in FIGURE 3 because there is insufficient evidence to rank them). The additional steps and age categories support the goal of using the least possible medication needed to maintain good control and minimize the potential for adverse events.
In initiating treatment, select the step that corresponds to the level of severity in the bottom row of FIGURE 1; to adjust medications, determine the patient’s level of asthma control and follow the corresponding guidance in the bottom row of FIGURE 2.
Inhaled corticosteroids remain the bedrock of therapy
ICS, the most potent and consistently effective long-term controller therapy, remain the foundation of therapy for patients of all ages who have persistent asthma. (Evidence: A).
Several of the age-based recommendations follow, with a focus on preferred treatments:
Children 0 to 4 years of age
- The guidelines recommend low-dose ICS at Step 2 (Evidence: A) and medium-dose ICS at Step 3 (Evidence: D), as inhaled corticosteroids have been shown to reduce impairment and risk in this age group.9-16 The potential risk is generally limited to a small reduction in growth velocity during the first year of treatment, and offset by the benefits of therapy.15,16
- Add a long-acting β2-adrenergic agonist (LABA) or montelukast to medium-dose ICS therapy at Step 4 rather than increasing the ICS dose (Evidence: D) to avoid the risk of side effects associated with high-dose ICS. Montelukast has demonstrated efficacy in children 2 to 5 years of age with persistent asthma.17
- Recommendations for preferred therapy at Steps 5 (high-dose ICS + LABA or montelukast) and 6 (Step 5 therapy + OCS) are based on expert panel judgment (Evidence: D). When severe persistent asthma warrants Step 6 therapy, start with a 2-week course of the lowest possible dose of OCS to confirm reversibility.
- In this age group, a therapeutic trial with close monitoring is recommended for patients whose asthma is not well controlled. If there is no response in 4 to 6 weeks, consider alternative therapies or diagnoses (Evidence: D).
Children 5 to 11 years of age
- For Step 3 therapy, the guidelines recommend either low-dose ICS plus a LABA, leukotriene receptor antagonist (LTRA), or theophylline; or medium-dose ICS (Evidence: B). Treatment decisions at Step 3 depend on whether impairment or risk is the chief concern, as well as on safety considerations.
- For Steps 4 and 5, ICS (medium dose for Step 5 and high dose for Step 6) plus a LABA is preferred, based on studies of patients ≥12 years of age (Evidence: B). Step 6 builds on Step 5, adding an OCS to the LABA/ICS combination (Evidence: D).
- If theophylline is prescribed—a viable option if cost and adherence to inhaled medications are key concerns—serum levels must be closely monitored because of the risk of toxicity.
- Closely monitor and be prepared to identify and respond to anaphylaxis in a child at Step 2, 3, or 4 who is receiving allergen immunotherapy.
Adolescents ≥12 years of age and adults
- There are 2 preferred Step 3 treatments: Low-dose ICS plus a LABA, or medium-dose ICS. The combination therapy has shown greater improvement in impairment24,25 and risk24-26 compared with the higher dose of ICS.
- Preferred treatments at Steps 4, 5, and 6 are the same as those for children ages 5 to 11 years, with one exception: Subcutaneous anti-IgE therapy (omalizumab) may be added to the regimen at Steps 5 and 6 for adolescents and adults with severe persistent allergic asthma to reduce the risk of exacerbations.27
Weigh the benefits and risks of therapy
Safety is a key consideration for all asthma patients. Carefully weigh the benefits and risks of therapy, including the rare but potential risk of life-threatening or fatal exacerbations with daily LABA therapy28 and systemic effects with higher doses of ICS.23 Patients who begin receiving oral corticosteroids require close monitoring, regardless of age.
Regular reassessment and monitoring are critical
Schedule visits at 2- to 6-week intervals for those who are starting therapy or require a step up to achieve or regain asthma control. After control is achieved, reassess at least every 1 to 6 months. Measures of asthma control are the same as those used to assess severity, with the addition of validated multidimensional questionnaires (eg, Asthma Control Test [ACT])29 to gauge impairment.
JJ’s physician scheduled a follow-up visit in 4 weeks, at which time he did a reassessment based on a physical exam and symptom recall. Finding JJ’s asthma to be well controlled, the physician asked the boy’s mother to bring him back to the office in 2 months, or earlier if symptoms recurred.
TABLE W1
Asthma education resources
| Allergy & Asthma Network Mothers of Asthmatics 2751 Prosperity Avenue, Suite 150 Fairfax, VA 22030 www.breatherville.org (800) 878-4403 or (703) 641-9595 | Asthma and Allergy Foundation of America 1233 20th Street, NW, Suite 402 Washington, DC 20036 www.aafa.org (800) 727-8462 |
| American Academy of Allergy, Asthma, and Immunology 555 East Wells Street, Suite 1100 Milwaukee, WI 53202-3823 www.aaaai.org (414) 272-6071 | Centers for Disease Control and Prevention 1600 Clifton Road Atlanta, GA 30333 www.cdc.gov (800) 311-3435 |
| American Association for Respiratory Care 9125 North macArthur boulevard, Suite 100 Irving, TX 75063 www.aarc.org (972) 243-2272 | Food Allergy & Anaphylaxis Network 11781 lee Jackson Highway, Suite 160 Fairfax, VA 22033 www.foodallergy.org (800) 929-4040 |
| American College of Allergy, Asthma, and Immunology 85 West Algonquin road, Suite 550 Arlington Heights, IL 60005 www.acaai.org (800) 842-7777 or (847) 427-1200 | National Heart, Lung, and Blood Institute Information Center P.O. Box 30105 Bethesda, MD 20824-0105 www.nhlbi.nih.gov (301) 592-8573 |
| American Lung Association 61 Broadway New York, NY 10006 www.lungusa.org (800) 586-4872 | National Jewish Medical and Research Center (Lung Line) 1400 Jackson Street Denver, CO 80206 www.njc.org (800) 222-lUNG |
| Association of Asthma Educators 1215 Anthony Avenue Columbia, SC 29201 www.asthmaeducators.org (888) 988-7747 | US Environmental Protection Agency National Center for Environmental Publications P.O. Box 42419 Cincinnati, OH 45242-0419 www.airnow.gov (800) 490-9198 |
Does your patient require a step down or step up?
A step down is recommended for patients whose asthma is well controlled for 3 months or more. Reduce the dose of ICS gradually, about 25% to 50% every 3 months, because deterioration in asthma control is highly variable. Review adherence and medication administration technique with patients whose asthma is not well controlled, and consider a step up in treatment. If an alternative treatment is used but does not result in an adequate response, it should be discontinued and the preferred treatment used before stepping up. Refer patients to an asthma specialist if their asthma does not respond to these adjustments.
Partner with patients for optimal care
The EPR-3 recommends the integration of patient education into all aspects of asthma care. To forge an active partnership, identify and address concerns about the condition and its treatment and involve the patient and family in developing treatment goals and making treatment decisions. If the patient is old enough, encourage self-monitoring and management.
The EPR-3 recommends that physicians give every patient a written asthma action plan that addresses individual symptoms and/or PEF measurements and includes instructions for self-management. Daily PEF monitoring can be useful in identifying early changes in the disease state and evaluating response to changes in therapy. It is ideal for those who have moderate to severe persistent asthma, difficulty recognizing signs of exacerbations, or a history of severe exacerbations.
Correspondence
Stuart W. Stoloff, MD, Clinical Professor, Department of Family and Community Medicine, University of Nevada–Reno, 1200 Mountain Street, Suite 220, Carson City, NV 89703; drstoloff@sbcglobal.net.
1. National Center for Health Statistics. Fast stats A to Z. Available at: www.cdc.gov/nchs/fastats/asthma.htm. Accessed August 1, 2008.
2. National Heart, Lung, and Blood Institute (NHLBI). National Asthma Education and Prevention Program Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma. Full Report 2007. Bethesda, MD: NHLBI; August 2007. NIH publication no. 07-4051. Available at: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm. Accessed July 17, 2008.
3. Stout JW, Visness CM, Enright P, et al. Classification of asthma severity in children: the contribution of pulmonary function testing. Arch Pediatr Adolesc Med. 2006;160:844-850.
4. Bacharier LB, Strunk RC, Mauger D, et al. Classifying asthma severity in children: mismatch between symptoms, medication use, and lung function. Am J Respir Crit Care Med. 2004;170:426-432.
5. Eid N, Yandell B, Howell L, Eddy M, Sheikh S. Can children with asthma? Pediatrics. 2000;105:354-358.
6. Llewellin P, Sawyer G, Lewis S, et al. The relationship between FEV1 and PEF in the assessment of the severity of airways obstruction. Respirology. 2002;7:333-337.
7. Yawn BP, Enright PL, Lemanske RF, Jr, et al. Spirometry can be done in family physicians’ offices and alters clinical decisions in management of asthma and COPD. Chest. 2007;132:1162-1168.
8. Fuhlbrigge AL, Kitch BT, Paltiel AD, et al. FEV1 is associated with risk of asthma attacks in a pediatric population. J Allergy Clin Immunol. 2001;107:61-67.
9. Roorda RJ, Mezei G, Bisgaard H, Maden C. Response of preschool children with asthma symptoms to fluticasone propionate. J Allergy Clin Immunol. 2001;108:540-546.
10. Baker JW, Mellon M, Wald J, Welch M, Cruz-Rivera M, Walton-Bowen K. A multiple-dosing, placebo-controlled study of budesonide inhalation suspension given once or twice daily for treatment of persistent asthma in young children and infants. Pediatrics. 1999;103:414-421.
11. Kemp JP, Skoner DP, Szefler SJ, Walton-Bowen K, Cruz-Rivera M, Smith JA. Once-daily budesonide inhalation suspension for the treatment of persistent asthma in infants and young children. Ann Allergy Asthma Immunol. 1999;83:231-239.
12. Shapiro G, Mendelson L, Kraemer MJ, Cruz-Rivera M, Walton-Bowen K, Smith JA. Efficacy and safety of budesonide inhalation suspension (Pulmicort Respules) in young children with inhaled steroid-dependent, persistent asthma. J Allergy Clin Immunol. 1998;102:789-796.
13. Bisgaard H, Gillies J, Groenewald M, Maden C. The effect of inhaled fluticasone propionate in the treatment of young asthmatic children: a dose comparison study. Am J Respir Crit Care Med. 1999;160:126-131.
14. Szefler SJ, Eigen H. Budesonide inhalation suspension: a nebulized corticosteroid for persistent asthma. J Allergy Clin Immunol. 2002;109:730-742.
15. Guilbert TW, Morgan WJ, Zeiger RS, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med. 2006;354:1985-1997.
16. Bisgaard H, Allen D, Milanowski J, Kalev I, Willits L, Davies P. Twelve-month safety and efficacy of inhaled fluticasone propionate in children aged 1 to 3 years with recurrent wheezing. Pediatrics. 2004;113:e87-e94.
17. Knorr B, Franchi LM, Bisgaard H, et al. Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years. Pediatrics 2001;108:e48.-
18. Russell G, Williams DA, Weller P, Price JF. Salmeterol xinafoate in children on high dose inhaled steroids. Ann Allergy Asthma Immunol. 1995;75:423-428.
19. Zimmerman B, D’Urzo A, Bérubé D. Efficacy and safety of formoterol Turbuhaler when added to inhaled corticosteroid treatment in children with asthma. Pediatr Pulmonol. 2004;37:122-127.
20. Simons FE, Villa JR, Lee BW, et al. Montelukast added to budesonide in children with persistent asthma: a randomized, double-blind, crossover study. J Pediatr. 2001;138:694-698.
21. Shapiro G, Bronsky EA, LaForce CF, et al. Dose-related efficacy of budesonide administered via a dry powder inhaler in the treatment of children with moderate to severe persistent asthma. J Pediatr. 1998;132:976-982.
22. Pauwels RA, Lofdahl C-G, Postma DS, et al. for the Formoterol and Corticosteroids Establishing Therapy (FACET) International Study Group. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med. 1997;337:1405-1411.
23. Tattersfield AE, Harrison TW, Hubbard RB, Mortimer K. Safety of inhaled corticosteroids. Proc Am Thorac Soc. 2004;1:171-175.
24. Bateman ED, Boushey HA, Bousquet J, et al. For the GOAL Investigators Group. Can guideline-defined asthma control be achieved? The Gaining Optimal Asthma ControL study. Am J Respir Crit Care Med. 2004;170:836-844.
25. O’Byrne PM, Barnes PJ, Rodriguez-Roisin R, et al. Low dose inhaled budesonide and formoterol in mild persistent asthma: the OPTIMA randomized trial. Am J Respir Crit Care Med. 2001;164:1392-1397.
26. Masoli M, Weatherall M, Holt S, Beasley R. Moderate dose inhaled corticosteroids plus salmeterol versus higher doses of inhaled corticosteroids in symptomatic asthma. Thorax. 2005;60:730-734.
27. Bousquet J, Wenzel S, Holgate S, Lumry W, Freeman P, Fox H. Predicting response to omalizumab, an anti-IgE antibody, in patients with allergic asthma. Chest. 2004;125:1378-1386.
28. Nelson HS, Weiss ST, Bleecker ER, Yancey SW, Dorinsky PM. For the SMART Study Group. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest. 2006;129:15-26.
29. Nathan RA, Sorkness CA, Kosinski M, et al. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol. 2004;113:59-65.
1. National Center for Health Statistics. Fast stats A to Z. Available at: www.cdc.gov/nchs/fastats/asthma.htm. Accessed August 1, 2008.
2. National Heart, Lung, and Blood Institute (NHLBI). National Asthma Education and Prevention Program Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma. Full Report 2007. Bethesda, MD: NHLBI; August 2007. NIH publication no. 07-4051. Available at: http://www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm. Accessed July 17, 2008.
3. Stout JW, Visness CM, Enright P, et al. Classification of asthma severity in children: the contribution of pulmonary function testing. Arch Pediatr Adolesc Med. 2006;160:844-850.
4. Bacharier LB, Strunk RC, Mauger D, et al. Classifying asthma severity in children: mismatch between symptoms, medication use, and lung function. Am J Respir Crit Care Med. 2004;170:426-432.
5. Eid N, Yandell B, Howell L, Eddy M, Sheikh S. Can children with asthma? Pediatrics. 2000;105:354-358.
6. Llewellin P, Sawyer G, Lewis S, et al. The relationship between FEV1 and PEF in the assessment of the severity of airways obstruction. Respirology. 2002;7:333-337.
7. Yawn BP, Enright PL, Lemanske RF, Jr, et al. Spirometry can be done in family physicians’ offices and alters clinical decisions in management of asthma and COPD. Chest. 2007;132:1162-1168.
8. Fuhlbrigge AL, Kitch BT, Paltiel AD, et al. FEV1 is associated with risk of asthma attacks in a pediatric population. J Allergy Clin Immunol. 2001;107:61-67.
9. Roorda RJ, Mezei G, Bisgaard H, Maden C. Response of preschool children with asthma symptoms to fluticasone propionate. J Allergy Clin Immunol. 2001;108:540-546.
10. Baker JW, Mellon M, Wald J, Welch M, Cruz-Rivera M, Walton-Bowen K. A multiple-dosing, placebo-controlled study of budesonide inhalation suspension given once or twice daily for treatment of persistent asthma in young children and infants. Pediatrics. 1999;103:414-421.
11. Kemp JP, Skoner DP, Szefler SJ, Walton-Bowen K, Cruz-Rivera M, Smith JA. Once-daily budesonide inhalation suspension for the treatment of persistent asthma in infants and young children. Ann Allergy Asthma Immunol. 1999;83:231-239.
12. Shapiro G, Mendelson L, Kraemer MJ, Cruz-Rivera M, Walton-Bowen K, Smith JA. Efficacy and safety of budesonide inhalation suspension (Pulmicort Respules) in young children with inhaled steroid-dependent, persistent asthma. J Allergy Clin Immunol. 1998;102:789-796.
13. Bisgaard H, Gillies J, Groenewald M, Maden C. The effect of inhaled fluticasone propionate in the treatment of young asthmatic children: a dose comparison study. Am J Respir Crit Care Med. 1999;160:126-131.
14. Szefler SJ, Eigen H. Budesonide inhalation suspension: a nebulized corticosteroid for persistent asthma. J Allergy Clin Immunol. 2002;109:730-742.
15. Guilbert TW, Morgan WJ, Zeiger RS, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med. 2006;354:1985-1997.
16. Bisgaard H, Allen D, Milanowski J, Kalev I, Willits L, Davies P. Twelve-month safety and efficacy of inhaled fluticasone propionate in children aged 1 to 3 years with recurrent wheezing. Pediatrics. 2004;113:e87-e94.
17. Knorr B, Franchi LM, Bisgaard H, et al. Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years. Pediatrics 2001;108:e48.-
18. Russell G, Williams DA, Weller P, Price JF. Salmeterol xinafoate in children on high dose inhaled steroids. Ann Allergy Asthma Immunol. 1995;75:423-428.
19. Zimmerman B, D’Urzo A, Bérubé D. Efficacy and safety of formoterol Turbuhaler when added to inhaled corticosteroid treatment in children with asthma. Pediatr Pulmonol. 2004;37:122-127.
20. Simons FE, Villa JR, Lee BW, et al. Montelukast added to budesonide in children with persistent asthma: a randomized, double-blind, crossover study. J Pediatr. 2001;138:694-698.
21. Shapiro G, Bronsky EA, LaForce CF, et al. Dose-related efficacy of budesonide administered via a dry powder inhaler in the treatment of children with moderate to severe persistent asthma. J Pediatr. 1998;132:976-982.
22. Pauwels RA, Lofdahl C-G, Postma DS, et al. for the Formoterol and Corticosteroids Establishing Therapy (FACET) International Study Group. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med. 1997;337:1405-1411.
23. Tattersfield AE, Harrison TW, Hubbard RB, Mortimer K. Safety of inhaled corticosteroids. Proc Am Thorac Soc. 2004;1:171-175.
24. Bateman ED, Boushey HA, Bousquet J, et al. For the GOAL Investigators Group. Can guideline-defined asthma control be achieved? The Gaining Optimal Asthma ControL study. Am J Respir Crit Care Med. 2004;170:836-844.
25. O’Byrne PM, Barnes PJ, Rodriguez-Roisin R, et al. Low dose inhaled budesonide and formoterol in mild persistent asthma: the OPTIMA randomized trial. Am J Respir Crit Care Med. 2001;164:1392-1397.
26. Masoli M, Weatherall M, Holt S, Beasley R. Moderate dose inhaled corticosteroids plus salmeterol versus higher doses of inhaled corticosteroids in symptomatic asthma. Thorax. 2005;60:730-734.
27. Bousquet J, Wenzel S, Holgate S, Lumry W, Freeman P, Fox H. Predicting response to omalizumab, an anti-IgE antibody, in patients with allergic asthma. Chest. 2004;125:1378-1386.
28. Nelson HS, Weiss ST, Bleecker ER, Yancey SW, Dorinsky PM. For the SMART Study Group. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest. 2006;129:15-26.
29. Nathan RA, Sorkness CA, Kosinski M, et al. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol. 2004;113:59-65.
Primary care’s eroding earnings: Is Congress concerned?
- Write your senator and congressional representative about the need for Medicare payment reform that addresses the primary care/specialist payment gap. Let them know, too, if you are no longer able to accept Medicare patients due to reduced payments.
Purpose: Despite increasing data demonstrating the positive impact primary care has on quality of care and costs, our specialty faces uncertainty. Its popularity among medical students is declining, and the income gap is growing between primary care and other specialties. Congress has the power to intervene in this impending crisis. If we want to influence lawmakers’ actions, we need to know how they are thinking about these issues.
Methods: Using a set of questions covering several physician payment topics, we interviewed 14 congressional staff aides (5 aides on Medicare-oversight committees, 9 general staff aides) and one representative from each of 3 governmental agencies: the Medicare Payment Advisory Commission, Congressional Budget Office, and Government Accountability Office.
Results: Interviewees revealed that issues in primary care are not high on the congressional agenda, and that Medicare’s Sustainable Growth Rate (SGR) is the physician-payment issue on the minds of congressional staff members.
Conclusion: Attempts to solve primary care’s reimbursement difficulties should be tied to SGR reform.
The viability of primary care in the United States is in question, attributable in large part to declining provider payments in the face of rising medical school debt and fee-for-service pressures to increase patient volume.1-3 Congress—which has authority over Medicare and its price-setting function for provider reimbursement overall—is seemingly unaware of the problems facing primary care, including barriers to payment reform. The future of our specialty may hinge on our ability to persuade Congress that these problems are dire. A growing body of evidence supports the essential and integrative function primary care plays in health systems, and its positive impact on quality of care and costs.4-6
The confused order of things now
Advantages of primary care are proven. Regions with higher ratios of primary care physicians relative to specialists have lower rates of hospitalizations, lower Medicare costs, and higher quality of care.7,8 People with a primary care physician are more satisfied with their care and more likely to receive preventive services and better chronic disease management.9-11 Most countries that have built their health care systems on a strong foundation of primary care demonstrate better health outcomes, fewer health care disparities, and lower costs.4,6 Thus the waning of primary care presents risks to both personal and population health.
Still, society undervalues primary care. Despite evidence of the benefits just cited, the income disparity between primary care physicians and specialists continues to grow, discouraging medical students from entering primary care careers.12 The Medical Group Management Association shows that between 2000 and 2004, the median income for a family physician increased 7.5% to $156,000; for invasive cardiologists, 16.9% to $428,000; and for diagnostic radiologists, 36.2% to $407,000. Adjusted for inflation, primary care income fell 10% from 1995 to 2004.13
No wonder students shy away from primary care. Though there is little public sympathy for the financial woes of primary care doctors, lower incomes are contributing to a drying of the primary care pipeline.14,15 The number of US medical school graduates choosing family medicine residencies dropped by 50% between 1997 and 2005.16 From 1998 to 2004, the number of internal medicine residents choosing careers in primary care plummeted from 54% to 25%.17,18 This waning interest in primary care coincides, unfortunately, with the aging of the US baby boomers and an increasing prevalence of chronic disease.
How Congress could help fix the disparity
Medicare reimbursement has 2 components that Congress could amend to narrow the payment gap and help open the primary care pipeline: the Sustainable Growth Rate (SGR) and the Resource-Based Relative Value Scale (RBRVS) process.
The SGR formula sets a target for Medicare physician expenditures each year. Recently, physician expenditure growth has exceeded the target and, by law, the difference is subtracted from the fees paid to all physicians. According to the Medicare Payment Advisory Commission (MedPAC), much of the excess spending has come from rapidly increasing volumes of procedures used by specialists.19 The SGR system therefore disproportionately penalizes primary care physicians because payments to all physicians are cut regardless of which specialties are responsible for excess spending.
RBRVS is the system of relative values applied to every procedure and office visit. The Relative Value Units (RVUs) for each procedure or office visit are multiplied by a conversion factor determined by the SGR formula. RVUs are largely governed by the Relative Value Scale Update Committee (RUC), which advises the Centers for Medicare and Medicaid Services (CMS) on revisions to physician reimbursement.
The RUC reviews the relative value scale at least every 5 years. Though primary care physicians provide about half of Medicare physician visits, they represent just 15% of the RUC’s voting members.12
The committee’s reevaluation process tends to raise some RVUs without sufficiently deflating others.20 The resulting overall inflation of fees forces CMS to reduce payments equally to all physicians, meaning primary care is again disproportionately penalized. Moreover, both Medicare and private insurance companies follow the RUC’s recommendations.
Influencing Congress: Where to begin? As Congress escalates its deliberations on Medicare physician spending, we investigated how key legislators perceive issues in primary care and physician payment.
Methods
To better understand perspectives of congressional committees with jurisdiction over health care spending, we conducted semistructured key informant interviews in March 2007 with 14 health staff aides to members of Congress who have jurisdiction over Medicare. Interviews were done face to face and lasted 30 to 60 minutes.
The congressional committees with jurisdiction over Medicare physician payment are Senate Finance, House Ways and Means, and House Energy and Commerce. Each committee has 1 majority and 1 minority staffer specializing in Medicare part B, which includes physician payment. Of these 6 specialized staffers, 5 agreed to participate in semistructured interviews. Other staffers were contacted by using a purposeful sampling technique known as “snowballing” or chain-referral, whereby participants with whom contact has been made refer the researcher to other potential interviewees. This process yielded another 9 interviewees to total 14.
The aides identified several other information sources, and we interviewed 1 staff member each from 3 of these sources: MedPAC, the Congressional Budget Office (CBO), and the Government Accountability Office (GAO).
Interviews covered several topics, including views on the state of primary care and physician payment (TABLE). Three researchers separately reviewed the interview notes to identify and compile themes.
TABLE
6 Questions we asked the congressional staffers
| 1. What are your views on the current state of primary care in the United States? |
| 2. When considering legislation to improve health care in the United States, how—if at all—does primary care factor into your vision? |
| 3. If there is legislative movement to change the Sustainable Growth Rate and Resource-Based Relative Value Scale systems in the next year, what should the goal be? |
| 4. What is your sense of other health legislative assistants’ understanding of primary care? |
| 5. Who are you hearing from on issues of primary care? Who are you not hearing from? |
| 6. What are the best sources to learn about these issues? |
Results
Of the 14 congressional staffers, 8 were Republican and 6 were Democrat; 5 were committee staff and 9 were general staff. Committee representation was fairly even among staffers: Senate Finance (4), House Ways and Means (5), and House Energy and Commerce (5). Range of experience on Capitol Hill was 3 months to 9 years.
Some staffers are empathetic, others unaware. Most respondents expressed concern about the decreasing number of students entering primary care careers and the potential impact on patient access to care. One staffer acknowledged, “the way our reimbursement system works, primary care is not an option for students…reimbursement is so low…the number of primary care physicians is going down relative to other specialties.”
Another participant added that most staffers “recognize a role for primary care. It’s also tough because of how strong the specialty community is.” One staffer advised, “The Alliance of Specialty Medicine goes along with the AMA, trying to represent a coordinated front…I don’t see this much coordination around primary care.”
A few staffers did not understand the definition of primary care or did not know which physician groups represent primary care.
Legislation to improve US health care—and primary care. Participants varied in their input on this subject. One staffer stated that primary care is “important but rarely singled out…usually the goal is broader reform so [primary care] is still a goal, but unstated.”
Some committee staff described the need to incentivize greater use of primary care and increase coordination of care. A few proposed reevaluating RBRVS to help primary care, and they spontaneously raised the Medical Home concept as a way to encourage growth of primary care. The Medical Home involves pairing each Medicare beneficiary with a patient-centered practice that meets certain criteria including continuity with a personal physician, care coordination, quality assurance, increased access, and specific payment.21 A pilot project in North Carolina that incorporates the Medical Home is saving the state about $162 million annually.22,23 One staffer championed primary care, but pointed out that a critical barrier preventing Congress from investing in it is the CBO, which is not convinced that primary care can save money over the long term.
The SGR dominates discussions on physician payment
All respondents had a functional understanding of the SGR and desired reform, but few understood how the SGR contributes to the payment gap. Many staffers would like to do away with the SGR, but CBO estimates show that this would be cost-prohibitive.24
A few staffers believed that SGR reform may not happen until 2009, after the next president takes office. Some participants also predicted that SGR reform will not happen until more physicians refuse to see Medicare patients. To date, MedPAC has reported each year that there is no Medicare access crisis. Staffers from rural districts, however, affirmed that constituents are having difficulty finding primary care doctors who take Medicare.
Staffers uniformly agreed that nobody has the answer to fix the SGR. Several staffers commented on the complexity of the problem, pointing out that MedPAC’s March 2007 SGR report did not achieve a consensus on how to restructure the rate. Many participants were disappointed with the MedPAC report and want solutions to fix physician payment that are more directed and “convincing.”
Some expressed a need for “hands-on models and demonstration projects.” Although these staffers have heard of models that would split the SGR by specialty or geography, they remain skeptical about such proposals without evidence of efficacy. Staffers were also wary of splitting the SGR by specialty, believing it would cause infighting among physicians.
Staffers know far less about RBRVS than they do about the SGR. One staffer admitted, “I won’t pay attention until something is at a crisis point or we have a hearing or a vote.” A few staffers asserted that there should be a more rigorous RUC review to examine what services are over- and undervalued.
Government agencies are not asked to address primary care. At the time of interview (March 2007), staff from MedPAC, GAO, and CBO said that Congress had not asked them to study issues in primary care. One CBO analyst asserted that “nobody’s been able to demonstrate significant changes in volume or outcome [as a result of investing in primary care]…we need empirical data.” The analyst also mentioned CMS demonstration projects as a way to gather data. According to a Capitol Hill veteran, the CBO believes that even if primary care extends a person’s life, this may not necessarily save money.
Discussion
Although most of the interviewed congressional staffers recognize the payment gap and understand that the number of physicians entering primary care is decreasing, Congress has not taken action to address these issues. Several factors explain this.
SGR is the 800-pound gorilla. When discussing physician payment, congressional staffers appear far more concerned with reforming the SGR than addressing problems in primary care. This perception is supported by the fact that Congress has asked MedPAC and CBO to investigate the SGR, but has not asked them to examine issues in primary care. For Congress, the dilemma is to hold down physician spending while keeping physicians in the Medicare market. Staffers are dissatisfied with SGR reform proposals from MedPAC and are eager to learn about new possible solutions.
No one perceives a crisis in access to Medicare providers. According to annual MedPAC reports, the number of primary care doctors accepting Medicare patients is sufficient. Staff for members of Congress from rural areas, however, contend that some constituents cannot find a primary care provider who accepts Medicare.
Congress is not convinced that primary care saves money. Although some staffers believe that primary care can reduce costs, the CBO argues that this is not necessarily true. It is indeed difficult to prove cost savings from investing in preventive services because there is greater upfront cost, and extending people’s lives could incur higher future costs. Research, however, shows that primary care-oriented systems reduce preventable hospitalizations, which decreases costs.4,5,7,8 It seems that either the existing evidence is insufficient to convince the CBO or the evidence has not been communicated effectively.
Strategic leverage moving forward
The time is ripe for SGR reform because most staffers conveyed a desire for solutions. Because the SGR appears to take priority over primary care issues, it must be dealt with first. It is possible, however, for policy makers to address the SGR and RBRVS reforms while simultaneously investing in primary care. The SGR and RBRVS reforms could hold specialties accountable for their own volume growth and protect specialties with minimal volume growth.
The Medical Home is a concept gaining recognition among congressional staff and could involve restructured physician payment. In its Tax Relief and Health Care Act of 2006, Congress mandated a 3-year Medical Home demonstration to be conducted across multiple demographic communities in up to 8 states. The concept encompasses “large or small medical practices where a physician provides comprehensive and coordinated patient centered medical care and acts as the ‘personal physician’ to the patient.”25 (The Medical Home is also a focus of The Patient-Centered Primary Care Collaborative [http://www.pcpcc.net/], a coalition of medical societies, employers, insurers, consumer groups, and others that is exploring the concept as a way to contain health care costs and also achieve fair remuneration for physicians.)
The demonstration must be carefully crafted to test the concept fairly. Even before the demonstration begins, Congress could ask the CBO and GAO to investigate existing evidence of primary care’s cost-effectiveness. Support from the CBO is essential for Congress to invest in primary care.
Other experiments are underway. As of this publication, several major insurers are beginning regional experiments in raising fees for primary care visits in an effort to avoid greater costs down the road.23
Access issue needs further study. Our interviews revealed that while MedPAC asserts there is no primary care access issue, staffers from rural districts disagree. In fact, had Congress not over-ridden President Bush’s recent veto of a Medicare bill to increase physicians’ fees, doctors in urban areas would also have stopped accepting new Medicare patients.26 Additional physician workforce studies are necessary to fully understand the current primary care physician supply. Also useful would be studies by Medicaid and Medicare that investigate thresholds at which physicians stop seeing patients with low-paying coverage.
Advocacy is needed, too. Congressional staffers appear to understand some of the difficulties in primary care, but give priority to broader SGR reform. Further research and advocacy on the value of primary care and payment reform solutions will be necessary to establish primary care as a means to cost-effective, high-quality care in the United States.
Acknowledgment
Part of the content in this article was presented as a poster at the North American Primary Care Research Group Conference in Vancouver, British Columbia, October 2007.
Correspondence
Brian Yoshio Laing, MD, San Francisco General Hospital, 995 Potrero Avenue, Building 80, Ward 83, San Francisco, CA 94110; yoshi.laing@ucsf.edu.
1. Bodenheimer T. Primary care–will it survive? Ann Intern Med. 2007;146:301-306.
2. Phillips RL. Primary care in the United States: problems and possibilities. BMJ. 2006;332:151.-
3. American College of Physicians. The impending collapse of primary care medicine and its implications for the state of the nation’s health care. January 30, 2006. Available at: http://www.txpeds.org/u/documents/statehc06_1.pdf. Accessed April 10, 2007.
4. Ferrer RL, Hambidge SJ, Maly RC. The essential role of generalists in health care systems. Ann Intern Med. 2005;142:691-699.
5. Macinko J, Starfield B, Shi L. Quantifying the health benefits of primary care physician supply in the United States. Int J Health Serv. 2007;37:111-126.
6. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83:457-502.
7. Parchman ML, Culler S. Primary care physicians and avoidable hospitalizations. J Fam Pract. 1994;39:123-128.
8. Baicker K, Chandra A. Medicare spending, the physician workforce, and beneficiaries’ quality of care. Health Affairs [Web Exclusive]. April 7, 2004;W4-184-197. Available at: http://content.healthaffairs.org/cgi/content/abstract/hlthaff.w4.184. Accessed July 30, 2008.
9. Bindman AB, Grumbach K, Osmond D, et al. Primary care and receipt of preventive services. J Gen Intern Med. 1996;11:269-276.
10. Safran DG, Taira GA, Rogers WH, et al. Linking primary care performance to outcomes of care. J Fam Pract. 1998;47:213-220.
11. Stewart AL, Grumbach K, Osmond DH, et al. Primary care and patient perceptions of access to care. J Fam Pract. 1997;44:177-185.
12. Bodeheimer T, Berenson RA, Rudolf P. The primary care-specialty income gap: why it matters. Ann Intern Med. 2007;146:301-306.
13. Tu HT, Ginsburg PB. Losing ground: physician income, 1995-2003. Tracking Rep. June 2006;15:1-8.
14. Rosenblatt RA, Andrilla HA. The impact of US medical students’ debt on their choice of primary care careers. Acad Med. 2005;80:815-819.
15. Newton DA, Grayson MS, Thompson LF. The variable influence of lifestyle and income on medical students’ career specialty choices. Acad Med. 2005;80:809-814.
16. Pugno PA, Schmittling GT, Fetter GT, et al. Results of the 2005 national resident matching program: family medicine. Fam Med. 2005;37:555-564.
17. Garibaldi RA, Popkave C, Bylsma W. Career plans for trainees in internal medicine residency programs. Acad Med. 2005;80:507-512.
18. West CP, Popkave C, Schultz HJ, et al. Changes in career decisions of internal medicine residents during training. Ann Intern Med. 2006;145:774-779.
19. Medicare Payment Advisory Commission. Report to the Congress: Medicare Payment Policy. March 2006. Available at: http://www.medpac.gov/publications/congressional_reports/Mar06_EntireReport.pdf. Accessed April 25, 2007.
20. Ginsburg PB, Berenson RA. Revising Medicare’s physician fee schedule–much activity, little change. N Engl J Med. 2007;356:1201-1203.
21. American College of Physicians. The Advanced Medical Home: A Patient-Centered, Physician-Guided Model of Health Care. 2006. Available at: http://www.acponline.org/advocacy/where_we_stand/policy/adv_med.pdf. Accessed May 13, 2007.
22. Steiner BD, Denham AC, Ashkin E, et al. Community care of North Carolina: improving care through community health networks. Ann Fam Med. 2008;in press.
23. Freudenheim M. Trying to save by increasing doctors’ fees. The New York Times. July 21, 2008. Available at: http://www.nytimes.com/2008/07/21/business/21medhome.html?_r=1&scp=1&sq=Trying%20to%20save%20by%20increasing%20doctors%20fees&st=cse&oref=slogin. Accessed August 1, 2008.
24. Orzang P. CBO. “Medicare’s Payments to Physicians: Option for Changing the Sustainable Growth Rate,” testimony before the Committee on Finance United States Senate. March 1, 2007. Available at: http://www.senate.gov/~finance/hearings/testimony/2007test/030107potest.pdf. Accessed May 13, 2007.
25. American Medical Association. RUC Medicare Medical Home Demonstration project recommendations. Available at: http://www.ama-assn.org/ama/pub/category/18528.html. Accessed August 1, 2008.
26. Pear R. Doctors press Senate to undo Medicare cuts. The New York Times. July 7, 2008. Available at: http://www.nytimes.com/2008/07/07/health/policy/07medicare.html?scp=1&sq=Doctors%20press%20senate%20to%20undo%20Medicare%20cuts&st=cse. Accessed August 1, 2008.
- Write your senator and congressional representative about the need for Medicare payment reform that addresses the primary care/specialist payment gap. Let them know, too, if you are no longer able to accept Medicare patients due to reduced payments.
Purpose: Despite increasing data demonstrating the positive impact primary care has on quality of care and costs, our specialty faces uncertainty. Its popularity among medical students is declining, and the income gap is growing between primary care and other specialties. Congress has the power to intervene in this impending crisis. If we want to influence lawmakers’ actions, we need to know how they are thinking about these issues.
Methods: Using a set of questions covering several physician payment topics, we interviewed 14 congressional staff aides (5 aides on Medicare-oversight committees, 9 general staff aides) and one representative from each of 3 governmental agencies: the Medicare Payment Advisory Commission, Congressional Budget Office, and Government Accountability Office.
Results: Interviewees revealed that issues in primary care are not high on the congressional agenda, and that Medicare’s Sustainable Growth Rate (SGR) is the physician-payment issue on the minds of congressional staff members.
Conclusion: Attempts to solve primary care’s reimbursement difficulties should be tied to SGR reform.
The viability of primary care in the United States is in question, attributable in large part to declining provider payments in the face of rising medical school debt and fee-for-service pressures to increase patient volume.1-3 Congress—which has authority over Medicare and its price-setting function for provider reimbursement overall—is seemingly unaware of the problems facing primary care, including barriers to payment reform. The future of our specialty may hinge on our ability to persuade Congress that these problems are dire. A growing body of evidence supports the essential and integrative function primary care plays in health systems, and its positive impact on quality of care and costs.4-6
The confused order of things now
Advantages of primary care are proven. Regions with higher ratios of primary care physicians relative to specialists have lower rates of hospitalizations, lower Medicare costs, and higher quality of care.7,8 People with a primary care physician are more satisfied with their care and more likely to receive preventive services and better chronic disease management.9-11 Most countries that have built their health care systems on a strong foundation of primary care demonstrate better health outcomes, fewer health care disparities, and lower costs.4,6 Thus the waning of primary care presents risks to both personal and population health.
Still, society undervalues primary care. Despite evidence of the benefits just cited, the income disparity between primary care physicians and specialists continues to grow, discouraging medical students from entering primary care careers.12 The Medical Group Management Association shows that between 2000 and 2004, the median income for a family physician increased 7.5% to $156,000; for invasive cardiologists, 16.9% to $428,000; and for diagnostic radiologists, 36.2% to $407,000. Adjusted for inflation, primary care income fell 10% from 1995 to 2004.13
No wonder students shy away from primary care. Though there is little public sympathy for the financial woes of primary care doctors, lower incomes are contributing to a drying of the primary care pipeline.14,15 The number of US medical school graduates choosing family medicine residencies dropped by 50% between 1997 and 2005.16 From 1998 to 2004, the number of internal medicine residents choosing careers in primary care plummeted from 54% to 25%.17,18 This waning interest in primary care coincides, unfortunately, with the aging of the US baby boomers and an increasing prevalence of chronic disease.
How Congress could help fix the disparity
Medicare reimbursement has 2 components that Congress could amend to narrow the payment gap and help open the primary care pipeline: the Sustainable Growth Rate (SGR) and the Resource-Based Relative Value Scale (RBRVS) process.
The SGR formula sets a target for Medicare physician expenditures each year. Recently, physician expenditure growth has exceeded the target and, by law, the difference is subtracted from the fees paid to all physicians. According to the Medicare Payment Advisory Commission (MedPAC), much of the excess spending has come from rapidly increasing volumes of procedures used by specialists.19 The SGR system therefore disproportionately penalizes primary care physicians because payments to all physicians are cut regardless of which specialties are responsible for excess spending.
RBRVS is the system of relative values applied to every procedure and office visit. The Relative Value Units (RVUs) for each procedure or office visit are multiplied by a conversion factor determined by the SGR formula. RVUs are largely governed by the Relative Value Scale Update Committee (RUC), which advises the Centers for Medicare and Medicaid Services (CMS) on revisions to physician reimbursement.
The RUC reviews the relative value scale at least every 5 years. Though primary care physicians provide about half of Medicare physician visits, they represent just 15% of the RUC’s voting members.12
The committee’s reevaluation process tends to raise some RVUs without sufficiently deflating others.20 The resulting overall inflation of fees forces CMS to reduce payments equally to all physicians, meaning primary care is again disproportionately penalized. Moreover, both Medicare and private insurance companies follow the RUC’s recommendations.
Influencing Congress: Where to begin? As Congress escalates its deliberations on Medicare physician spending, we investigated how key legislators perceive issues in primary care and physician payment.
Methods
To better understand perspectives of congressional committees with jurisdiction over health care spending, we conducted semistructured key informant interviews in March 2007 with 14 health staff aides to members of Congress who have jurisdiction over Medicare. Interviews were done face to face and lasted 30 to 60 minutes.
The congressional committees with jurisdiction over Medicare physician payment are Senate Finance, House Ways and Means, and House Energy and Commerce. Each committee has 1 majority and 1 minority staffer specializing in Medicare part B, which includes physician payment. Of these 6 specialized staffers, 5 agreed to participate in semistructured interviews. Other staffers were contacted by using a purposeful sampling technique known as “snowballing” or chain-referral, whereby participants with whom contact has been made refer the researcher to other potential interviewees. This process yielded another 9 interviewees to total 14.
The aides identified several other information sources, and we interviewed 1 staff member each from 3 of these sources: MedPAC, the Congressional Budget Office (CBO), and the Government Accountability Office (GAO).
Interviews covered several topics, including views on the state of primary care and physician payment (TABLE). Three researchers separately reviewed the interview notes to identify and compile themes.
TABLE
6 Questions we asked the congressional staffers
| 1. What are your views on the current state of primary care in the United States? |
| 2. When considering legislation to improve health care in the United States, how—if at all—does primary care factor into your vision? |
| 3. If there is legislative movement to change the Sustainable Growth Rate and Resource-Based Relative Value Scale systems in the next year, what should the goal be? |
| 4. What is your sense of other health legislative assistants’ understanding of primary care? |
| 5. Who are you hearing from on issues of primary care? Who are you not hearing from? |
| 6. What are the best sources to learn about these issues? |
Results
Of the 14 congressional staffers, 8 were Republican and 6 were Democrat; 5 were committee staff and 9 were general staff. Committee representation was fairly even among staffers: Senate Finance (4), House Ways and Means (5), and House Energy and Commerce (5). Range of experience on Capitol Hill was 3 months to 9 years.
Some staffers are empathetic, others unaware. Most respondents expressed concern about the decreasing number of students entering primary care careers and the potential impact on patient access to care. One staffer acknowledged, “the way our reimbursement system works, primary care is not an option for students…reimbursement is so low…the number of primary care physicians is going down relative to other specialties.”
Another participant added that most staffers “recognize a role for primary care. It’s also tough because of how strong the specialty community is.” One staffer advised, “The Alliance of Specialty Medicine goes along with the AMA, trying to represent a coordinated front…I don’t see this much coordination around primary care.”
A few staffers did not understand the definition of primary care or did not know which physician groups represent primary care.
Legislation to improve US health care—and primary care. Participants varied in their input on this subject. One staffer stated that primary care is “important but rarely singled out…usually the goal is broader reform so [primary care] is still a goal, but unstated.”
Some committee staff described the need to incentivize greater use of primary care and increase coordination of care. A few proposed reevaluating RBRVS to help primary care, and they spontaneously raised the Medical Home concept as a way to encourage growth of primary care. The Medical Home involves pairing each Medicare beneficiary with a patient-centered practice that meets certain criteria including continuity with a personal physician, care coordination, quality assurance, increased access, and specific payment.21 A pilot project in North Carolina that incorporates the Medical Home is saving the state about $162 million annually.22,23 One staffer championed primary care, but pointed out that a critical barrier preventing Congress from investing in it is the CBO, which is not convinced that primary care can save money over the long term.
The SGR dominates discussions on physician payment
All respondents had a functional understanding of the SGR and desired reform, but few understood how the SGR contributes to the payment gap. Many staffers would like to do away with the SGR, but CBO estimates show that this would be cost-prohibitive.24
A few staffers believed that SGR reform may not happen until 2009, after the next president takes office. Some participants also predicted that SGR reform will not happen until more physicians refuse to see Medicare patients. To date, MedPAC has reported each year that there is no Medicare access crisis. Staffers from rural districts, however, affirmed that constituents are having difficulty finding primary care doctors who take Medicare.
Staffers uniformly agreed that nobody has the answer to fix the SGR. Several staffers commented on the complexity of the problem, pointing out that MedPAC’s March 2007 SGR report did not achieve a consensus on how to restructure the rate. Many participants were disappointed with the MedPAC report and want solutions to fix physician payment that are more directed and “convincing.”
Some expressed a need for “hands-on models and demonstration projects.” Although these staffers have heard of models that would split the SGR by specialty or geography, they remain skeptical about such proposals without evidence of efficacy. Staffers were also wary of splitting the SGR by specialty, believing it would cause infighting among physicians.
Staffers know far less about RBRVS than they do about the SGR. One staffer admitted, “I won’t pay attention until something is at a crisis point or we have a hearing or a vote.” A few staffers asserted that there should be a more rigorous RUC review to examine what services are over- and undervalued.
Government agencies are not asked to address primary care. At the time of interview (March 2007), staff from MedPAC, GAO, and CBO said that Congress had not asked them to study issues in primary care. One CBO analyst asserted that “nobody’s been able to demonstrate significant changes in volume or outcome [as a result of investing in primary care]…we need empirical data.” The analyst also mentioned CMS demonstration projects as a way to gather data. According to a Capitol Hill veteran, the CBO believes that even if primary care extends a person’s life, this may not necessarily save money.
Discussion
Although most of the interviewed congressional staffers recognize the payment gap and understand that the number of physicians entering primary care is decreasing, Congress has not taken action to address these issues. Several factors explain this.
SGR is the 800-pound gorilla. When discussing physician payment, congressional staffers appear far more concerned with reforming the SGR than addressing problems in primary care. This perception is supported by the fact that Congress has asked MedPAC and CBO to investigate the SGR, but has not asked them to examine issues in primary care. For Congress, the dilemma is to hold down physician spending while keeping physicians in the Medicare market. Staffers are dissatisfied with SGR reform proposals from MedPAC and are eager to learn about new possible solutions.
No one perceives a crisis in access to Medicare providers. According to annual MedPAC reports, the number of primary care doctors accepting Medicare patients is sufficient. Staff for members of Congress from rural areas, however, contend that some constituents cannot find a primary care provider who accepts Medicare.
Congress is not convinced that primary care saves money. Although some staffers believe that primary care can reduce costs, the CBO argues that this is not necessarily true. It is indeed difficult to prove cost savings from investing in preventive services because there is greater upfront cost, and extending people’s lives could incur higher future costs. Research, however, shows that primary care-oriented systems reduce preventable hospitalizations, which decreases costs.4,5,7,8 It seems that either the existing evidence is insufficient to convince the CBO or the evidence has not been communicated effectively.
Strategic leverage moving forward
The time is ripe for SGR reform because most staffers conveyed a desire for solutions. Because the SGR appears to take priority over primary care issues, it must be dealt with first. It is possible, however, for policy makers to address the SGR and RBRVS reforms while simultaneously investing in primary care. The SGR and RBRVS reforms could hold specialties accountable for their own volume growth and protect specialties with minimal volume growth.
The Medical Home is a concept gaining recognition among congressional staff and could involve restructured physician payment. In its Tax Relief and Health Care Act of 2006, Congress mandated a 3-year Medical Home demonstration to be conducted across multiple demographic communities in up to 8 states. The concept encompasses “large or small medical practices where a physician provides comprehensive and coordinated patient centered medical care and acts as the ‘personal physician’ to the patient.”25 (The Medical Home is also a focus of The Patient-Centered Primary Care Collaborative [http://www.pcpcc.net/], a coalition of medical societies, employers, insurers, consumer groups, and others that is exploring the concept as a way to contain health care costs and also achieve fair remuneration for physicians.)
The demonstration must be carefully crafted to test the concept fairly. Even before the demonstration begins, Congress could ask the CBO and GAO to investigate existing evidence of primary care’s cost-effectiveness. Support from the CBO is essential for Congress to invest in primary care.
Other experiments are underway. As of this publication, several major insurers are beginning regional experiments in raising fees for primary care visits in an effort to avoid greater costs down the road.23
Access issue needs further study. Our interviews revealed that while MedPAC asserts there is no primary care access issue, staffers from rural districts disagree. In fact, had Congress not over-ridden President Bush’s recent veto of a Medicare bill to increase physicians’ fees, doctors in urban areas would also have stopped accepting new Medicare patients.26 Additional physician workforce studies are necessary to fully understand the current primary care physician supply. Also useful would be studies by Medicaid and Medicare that investigate thresholds at which physicians stop seeing patients with low-paying coverage.
Advocacy is needed, too. Congressional staffers appear to understand some of the difficulties in primary care, but give priority to broader SGR reform. Further research and advocacy on the value of primary care and payment reform solutions will be necessary to establish primary care as a means to cost-effective, high-quality care in the United States.
Acknowledgment
Part of the content in this article was presented as a poster at the North American Primary Care Research Group Conference in Vancouver, British Columbia, October 2007.
Correspondence
Brian Yoshio Laing, MD, San Francisco General Hospital, 995 Potrero Avenue, Building 80, Ward 83, San Francisco, CA 94110; yoshi.laing@ucsf.edu.
- Write your senator and congressional representative about the need for Medicare payment reform that addresses the primary care/specialist payment gap. Let them know, too, if you are no longer able to accept Medicare patients due to reduced payments.
Purpose: Despite increasing data demonstrating the positive impact primary care has on quality of care and costs, our specialty faces uncertainty. Its popularity among medical students is declining, and the income gap is growing between primary care and other specialties. Congress has the power to intervene in this impending crisis. If we want to influence lawmakers’ actions, we need to know how they are thinking about these issues.
Methods: Using a set of questions covering several physician payment topics, we interviewed 14 congressional staff aides (5 aides on Medicare-oversight committees, 9 general staff aides) and one representative from each of 3 governmental agencies: the Medicare Payment Advisory Commission, Congressional Budget Office, and Government Accountability Office.
Results: Interviewees revealed that issues in primary care are not high on the congressional agenda, and that Medicare’s Sustainable Growth Rate (SGR) is the physician-payment issue on the minds of congressional staff members.
Conclusion: Attempts to solve primary care’s reimbursement difficulties should be tied to SGR reform.
The viability of primary care in the United States is in question, attributable in large part to declining provider payments in the face of rising medical school debt and fee-for-service pressures to increase patient volume.1-3 Congress—which has authority over Medicare and its price-setting function for provider reimbursement overall—is seemingly unaware of the problems facing primary care, including barriers to payment reform. The future of our specialty may hinge on our ability to persuade Congress that these problems are dire. A growing body of evidence supports the essential and integrative function primary care plays in health systems, and its positive impact on quality of care and costs.4-6
The confused order of things now
Advantages of primary care are proven. Regions with higher ratios of primary care physicians relative to specialists have lower rates of hospitalizations, lower Medicare costs, and higher quality of care.7,8 People with a primary care physician are more satisfied with their care and more likely to receive preventive services and better chronic disease management.9-11 Most countries that have built their health care systems on a strong foundation of primary care demonstrate better health outcomes, fewer health care disparities, and lower costs.4,6 Thus the waning of primary care presents risks to both personal and population health.
Still, society undervalues primary care. Despite evidence of the benefits just cited, the income disparity between primary care physicians and specialists continues to grow, discouraging medical students from entering primary care careers.12 The Medical Group Management Association shows that between 2000 and 2004, the median income for a family physician increased 7.5% to $156,000; for invasive cardiologists, 16.9% to $428,000; and for diagnostic radiologists, 36.2% to $407,000. Adjusted for inflation, primary care income fell 10% from 1995 to 2004.13
No wonder students shy away from primary care. Though there is little public sympathy for the financial woes of primary care doctors, lower incomes are contributing to a drying of the primary care pipeline.14,15 The number of US medical school graduates choosing family medicine residencies dropped by 50% between 1997 and 2005.16 From 1998 to 2004, the number of internal medicine residents choosing careers in primary care plummeted from 54% to 25%.17,18 This waning interest in primary care coincides, unfortunately, with the aging of the US baby boomers and an increasing prevalence of chronic disease.
How Congress could help fix the disparity
Medicare reimbursement has 2 components that Congress could amend to narrow the payment gap and help open the primary care pipeline: the Sustainable Growth Rate (SGR) and the Resource-Based Relative Value Scale (RBRVS) process.
The SGR formula sets a target for Medicare physician expenditures each year. Recently, physician expenditure growth has exceeded the target and, by law, the difference is subtracted from the fees paid to all physicians. According to the Medicare Payment Advisory Commission (MedPAC), much of the excess spending has come from rapidly increasing volumes of procedures used by specialists.19 The SGR system therefore disproportionately penalizes primary care physicians because payments to all physicians are cut regardless of which specialties are responsible for excess spending.
RBRVS is the system of relative values applied to every procedure and office visit. The Relative Value Units (RVUs) for each procedure or office visit are multiplied by a conversion factor determined by the SGR formula. RVUs are largely governed by the Relative Value Scale Update Committee (RUC), which advises the Centers for Medicare and Medicaid Services (CMS) on revisions to physician reimbursement.
The RUC reviews the relative value scale at least every 5 years. Though primary care physicians provide about half of Medicare physician visits, they represent just 15% of the RUC’s voting members.12
The committee’s reevaluation process tends to raise some RVUs without sufficiently deflating others.20 The resulting overall inflation of fees forces CMS to reduce payments equally to all physicians, meaning primary care is again disproportionately penalized. Moreover, both Medicare and private insurance companies follow the RUC’s recommendations.
Influencing Congress: Where to begin? As Congress escalates its deliberations on Medicare physician spending, we investigated how key legislators perceive issues in primary care and physician payment.
Methods
To better understand perspectives of congressional committees with jurisdiction over health care spending, we conducted semistructured key informant interviews in March 2007 with 14 health staff aides to members of Congress who have jurisdiction over Medicare. Interviews were done face to face and lasted 30 to 60 minutes.
The congressional committees with jurisdiction over Medicare physician payment are Senate Finance, House Ways and Means, and House Energy and Commerce. Each committee has 1 majority and 1 minority staffer specializing in Medicare part B, which includes physician payment. Of these 6 specialized staffers, 5 agreed to participate in semistructured interviews. Other staffers were contacted by using a purposeful sampling technique known as “snowballing” or chain-referral, whereby participants with whom contact has been made refer the researcher to other potential interviewees. This process yielded another 9 interviewees to total 14.
The aides identified several other information sources, and we interviewed 1 staff member each from 3 of these sources: MedPAC, the Congressional Budget Office (CBO), and the Government Accountability Office (GAO).
Interviews covered several topics, including views on the state of primary care and physician payment (TABLE). Three researchers separately reviewed the interview notes to identify and compile themes.
TABLE
6 Questions we asked the congressional staffers
| 1. What are your views on the current state of primary care in the United States? |
| 2. When considering legislation to improve health care in the United States, how—if at all—does primary care factor into your vision? |
| 3. If there is legislative movement to change the Sustainable Growth Rate and Resource-Based Relative Value Scale systems in the next year, what should the goal be? |
| 4. What is your sense of other health legislative assistants’ understanding of primary care? |
| 5. Who are you hearing from on issues of primary care? Who are you not hearing from? |
| 6. What are the best sources to learn about these issues? |
Results
Of the 14 congressional staffers, 8 were Republican and 6 were Democrat; 5 were committee staff and 9 were general staff. Committee representation was fairly even among staffers: Senate Finance (4), House Ways and Means (5), and House Energy and Commerce (5). Range of experience on Capitol Hill was 3 months to 9 years.
Some staffers are empathetic, others unaware. Most respondents expressed concern about the decreasing number of students entering primary care careers and the potential impact on patient access to care. One staffer acknowledged, “the way our reimbursement system works, primary care is not an option for students…reimbursement is so low…the number of primary care physicians is going down relative to other specialties.”
Another participant added that most staffers “recognize a role for primary care. It’s also tough because of how strong the specialty community is.” One staffer advised, “The Alliance of Specialty Medicine goes along with the AMA, trying to represent a coordinated front…I don’t see this much coordination around primary care.”
A few staffers did not understand the definition of primary care or did not know which physician groups represent primary care.
Legislation to improve US health care—and primary care. Participants varied in their input on this subject. One staffer stated that primary care is “important but rarely singled out…usually the goal is broader reform so [primary care] is still a goal, but unstated.”
Some committee staff described the need to incentivize greater use of primary care and increase coordination of care. A few proposed reevaluating RBRVS to help primary care, and they spontaneously raised the Medical Home concept as a way to encourage growth of primary care. The Medical Home involves pairing each Medicare beneficiary with a patient-centered practice that meets certain criteria including continuity with a personal physician, care coordination, quality assurance, increased access, and specific payment.21 A pilot project in North Carolina that incorporates the Medical Home is saving the state about $162 million annually.22,23 One staffer championed primary care, but pointed out that a critical barrier preventing Congress from investing in it is the CBO, which is not convinced that primary care can save money over the long term.
The SGR dominates discussions on physician payment
All respondents had a functional understanding of the SGR and desired reform, but few understood how the SGR contributes to the payment gap. Many staffers would like to do away with the SGR, but CBO estimates show that this would be cost-prohibitive.24
A few staffers believed that SGR reform may not happen until 2009, after the next president takes office. Some participants also predicted that SGR reform will not happen until more physicians refuse to see Medicare patients. To date, MedPAC has reported each year that there is no Medicare access crisis. Staffers from rural districts, however, affirmed that constituents are having difficulty finding primary care doctors who take Medicare.
Staffers uniformly agreed that nobody has the answer to fix the SGR. Several staffers commented on the complexity of the problem, pointing out that MedPAC’s March 2007 SGR report did not achieve a consensus on how to restructure the rate. Many participants were disappointed with the MedPAC report and want solutions to fix physician payment that are more directed and “convincing.”
Some expressed a need for “hands-on models and demonstration projects.” Although these staffers have heard of models that would split the SGR by specialty or geography, they remain skeptical about such proposals without evidence of efficacy. Staffers were also wary of splitting the SGR by specialty, believing it would cause infighting among physicians.
Staffers know far less about RBRVS than they do about the SGR. One staffer admitted, “I won’t pay attention until something is at a crisis point or we have a hearing or a vote.” A few staffers asserted that there should be a more rigorous RUC review to examine what services are over- and undervalued.
Government agencies are not asked to address primary care. At the time of interview (March 2007), staff from MedPAC, GAO, and CBO said that Congress had not asked them to study issues in primary care. One CBO analyst asserted that “nobody’s been able to demonstrate significant changes in volume or outcome [as a result of investing in primary care]…we need empirical data.” The analyst also mentioned CMS demonstration projects as a way to gather data. According to a Capitol Hill veteran, the CBO believes that even if primary care extends a person’s life, this may not necessarily save money.
Discussion
Although most of the interviewed congressional staffers recognize the payment gap and understand that the number of physicians entering primary care is decreasing, Congress has not taken action to address these issues. Several factors explain this.
SGR is the 800-pound gorilla. When discussing physician payment, congressional staffers appear far more concerned with reforming the SGR than addressing problems in primary care. This perception is supported by the fact that Congress has asked MedPAC and CBO to investigate the SGR, but has not asked them to examine issues in primary care. For Congress, the dilemma is to hold down physician spending while keeping physicians in the Medicare market. Staffers are dissatisfied with SGR reform proposals from MedPAC and are eager to learn about new possible solutions.
No one perceives a crisis in access to Medicare providers. According to annual MedPAC reports, the number of primary care doctors accepting Medicare patients is sufficient. Staff for members of Congress from rural areas, however, contend that some constituents cannot find a primary care provider who accepts Medicare.
Congress is not convinced that primary care saves money. Although some staffers believe that primary care can reduce costs, the CBO argues that this is not necessarily true. It is indeed difficult to prove cost savings from investing in preventive services because there is greater upfront cost, and extending people’s lives could incur higher future costs. Research, however, shows that primary care-oriented systems reduce preventable hospitalizations, which decreases costs.4,5,7,8 It seems that either the existing evidence is insufficient to convince the CBO or the evidence has not been communicated effectively.
Strategic leverage moving forward
The time is ripe for SGR reform because most staffers conveyed a desire for solutions. Because the SGR appears to take priority over primary care issues, it must be dealt with first. It is possible, however, for policy makers to address the SGR and RBRVS reforms while simultaneously investing in primary care. The SGR and RBRVS reforms could hold specialties accountable for their own volume growth and protect specialties with minimal volume growth.
The Medical Home is a concept gaining recognition among congressional staff and could involve restructured physician payment. In its Tax Relief and Health Care Act of 2006, Congress mandated a 3-year Medical Home demonstration to be conducted across multiple demographic communities in up to 8 states. The concept encompasses “large or small medical practices where a physician provides comprehensive and coordinated patient centered medical care and acts as the ‘personal physician’ to the patient.”25 (The Medical Home is also a focus of The Patient-Centered Primary Care Collaborative [http://www.pcpcc.net/], a coalition of medical societies, employers, insurers, consumer groups, and others that is exploring the concept as a way to contain health care costs and also achieve fair remuneration for physicians.)
The demonstration must be carefully crafted to test the concept fairly. Even before the demonstration begins, Congress could ask the CBO and GAO to investigate existing evidence of primary care’s cost-effectiveness. Support from the CBO is essential for Congress to invest in primary care.
Other experiments are underway. As of this publication, several major insurers are beginning regional experiments in raising fees for primary care visits in an effort to avoid greater costs down the road.23
Access issue needs further study. Our interviews revealed that while MedPAC asserts there is no primary care access issue, staffers from rural districts disagree. In fact, had Congress not over-ridden President Bush’s recent veto of a Medicare bill to increase physicians’ fees, doctors in urban areas would also have stopped accepting new Medicare patients.26 Additional physician workforce studies are necessary to fully understand the current primary care physician supply. Also useful would be studies by Medicaid and Medicare that investigate thresholds at which physicians stop seeing patients with low-paying coverage.
Advocacy is needed, too. Congressional staffers appear to understand some of the difficulties in primary care, but give priority to broader SGR reform. Further research and advocacy on the value of primary care and payment reform solutions will be necessary to establish primary care as a means to cost-effective, high-quality care in the United States.
Acknowledgment
Part of the content in this article was presented as a poster at the North American Primary Care Research Group Conference in Vancouver, British Columbia, October 2007.
Correspondence
Brian Yoshio Laing, MD, San Francisco General Hospital, 995 Potrero Avenue, Building 80, Ward 83, San Francisco, CA 94110; yoshi.laing@ucsf.edu.
1. Bodenheimer T. Primary care–will it survive? Ann Intern Med. 2007;146:301-306.
2. Phillips RL. Primary care in the United States: problems and possibilities. BMJ. 2006;332:151.-
3. American College of Physicians. The impending collapse of primary care medicine and its implications for the state of the nation’s health care. January 30, 2006. Available at: http://www.txpeds.org/u/documents/statehc06_1.pdf. Accessed April 10, 2007.
4. Ferrer RL, Hambidge SJ, Maly RC. The essential role of generalists in health care systems. Ann Intern Med. 2005;142:691-699.
5. Macinko J, Starfield B, Shi L. Quantifying the health benefits of primary care physician supply in the United States. Int J Health Serv. 2007;37:111-126.
6. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83:457-502.
7. Parchman ML, Culler S. Primary care physicians and avoidable hospitalizations. J Fam Pract. 1994;39:123-128.
8. Baicker K, Chandra A. Medicare spending, the physician workforce, and beneficiaries’ quality of care. Health Affairs [Web Exclusive]. April 7, 2004;W4-184-197. Available at: http://content.healthaffairs.org/cgi/content/abstract/hlthaff.w4.184. Accessed July 30, 2008.
9. Bindman AB, Grumbach K, Osmond D, et al. Primary care and receipt of preventive services. J Gen Intern Med. 1996;11:269-276.
10. Safran DG, Taira GA, Rogers WH, et al. Linking primary care performance to outcomes of care. J Fam Pract. 1998;47:213-220.
11. Stewart AL, Grumbach K, Osmond DH, et al. Primary care and patient perceptions of access to care. J Fam Pract. 1997;44:177-185.
12. Bodeheimer T, Berenson RA, Rudolf P. The primary care-specialty income gap: why it matters. Ann Intern Med. 2007;146:301-306.
13. Tu HT, Ginsburg PB. Losing ground: physician income, 1995-2003. Tracking Rep. June 2006;15:1-8.
14. Rosenblatt RA, Andrilla HA. The impact of US medical students’ debt on their choice of primary care careers. Acad Med. 2005;80:815-819.
15. Newton DA, Grayson MS, Thompson LF. The variable influence of lifestyle and income on medical students’ career specialty choices. Acad Med. 2005;80:809-814.
16. Pugno PA, Schmittling GT, Fetter GT, et al. Results of the 2005 national resident matching program: family medicine. Fam Med. 2005;37:555-564.
17. Garibaldi RA, Popkave C, Bylsma W. Career plans for trainees in internal medicine residency programs. Acad Med. 2005;80:507-512.
18. West CP, Popkave C, Schultz HJ, et al. Changes in career decisions of internal medicine residents during training. Ann Intern Med. 2006;145:774-779.
19. Medicare Payment Advisory Commission. Report to the Congress: Medicare Payment Policy. March 2006. Available at: http://www.medpac.gov/publications/congressional_reports/Mar06_EntireReport.pdf. Accessed April 25, 2007.
20. Ginsburg PB, Berenson RA. Revising Medicare’s physician fee schedule–much activity, little change. N Engl J Med. 2007;356:1201-1203.
21. American College of Physicians. The Advanced Medical Home: A Patient-Centered, Physician-Guided Model of Health Care. 2006. Available at: http://www.acponline.org/advocacy/where_we_stand/policy/adv_med.pdf. Accessed May 13, 2007.
22. Steiner BD, Denham AC, Ashkin E, et al. Community care of North Carolina: improving care through community health networks. Ann Fam Med. 2008;in press.
23. Freudenheim M. Trying to save by increasing doctors’ fees. The New York Times. July 21, 2008. Available at: http://www.nytimes.com/2008/07/21/business/21medhome.html?_r=1&scp=1&sq=Trying%20to%20save%20by%20increasing%20doctors%20fees&st=cse&oref=slogin. Accessed August 1, 2008.
24. Orzang P. CBO. “Medicare’s Payments to Physicians: Option for Changing the Sustainable Growth Rate,” testimony before the Committee on Finance United States Senate. March 1, 2007. Available at: http://www.senate.gov/~finance/hearings/testimony/2007test/030107potest.pdf. Accessed May 13, 2007.
25. American Medical Association. RUC Medicare Medical Home Demonstration project recommendations. Available at: http://www.ama-assn.org/ama/pub/category/18528.html. Accessed August 1, 2008.
26. Pear R. Doctors press Senate to undo Medicare cuts. The New York Times. July 7, 2008. Available at: http://www.nytimes.com/2008/07/07/health/policy/07medicare.html?scp=1&sq=Doctors%20press%20senate%20to%20undo%20Medicare%20cuts&st=cse. Accessed August 1, 2008.
1. Bodenheimer T. Primary care–will it survive? Ann Intern Med. 2007;146:301-306.
2. Phillips RL. Primary care in the United States: problems and possibilities. BMJ. 2006;332:151.-
3. American College of Physicians. The impending collapse of primary care medicine and its implications for the state of the nation’s health care. January 30, 2006. Available at: http://www.txpeds.org/u/documents/statehc06_1.pdf. Accessed April 10, 2007.
4. Ferrer RL, Hambidge SJ, Maly RC. The essential role of generalists in health care systems. Ann Intern Med. 2005;142:691-699.
5. Macinko J, Starfield B, Shi L. Quantifying the health benefits of primary care physician supply in the United States. Int J Health Serv. 2007;37:111-126.
6. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83:457-502.
7. Parchman ML, Culler S. Primary care physicians and avoidable hospitalizations. J Fam Pract. 1994;39:123-128.
8. Baicker K, Chandra A. Medicare spending, the physician workforce, and beneficiaries’ quality of care. Health Affairs [Web Exclusive]. April 7, 2004;W4-184-197. Available at: http://content.healthaffairs.org/cgi/content/abstract/hlthaff.w4.184. Accessed July 30, 2008.
9. Bindman AB, Grumbach K, Osmond D, et al. Primary care and receipt of preventive services. J Gen Intern Med. 1996;11:269-276.
10. Safran DG, Taira GA, Rogers WH, et al. Linking primary care performance to outcomes of care. J Fam Pract. 1998;47:213-220.
11. Stewart AL, Grumbach K, Osmond DH, et al. Primary care and patient perceptions of access to care. J Fam Pract. 1997;44:177-185.
12. Bodeheimer T, Berenson RA, Rudolf P. The primary care-specialty income gap: why it matters. Ann Intern Med. 2007;146:301-306.
13. Tu HT, Ginsburg PB. Losing ground: physician income, 1995-2003. Tracking Rep. June 2006;15:1-8.
14. Rosenblatt RA, Andrilla HA. The impact of US medical students’ debt on their choice of primary care careers. Acad Med. 2005;80:815-819.
15. Newton DA, Grayson MS, Thompson LF. The variable influence of lifestyle and income on medical students’ career specialty choices. Acad Med. 2005;80:809-814.
16. Pugno PA, Schmittling GT, Fetter GT, et al. Results of the 2005 national resident matching program: family medicine. Fam Med. 2005;37:555-564.
17. Garibaldi RA, Popkave C, Bylsma W. Career plans for trainees in internal medicine residency programs. Acad Med. 2005;80:507-512.
18. West CP, Popkave C, Schultz HJ, et al. Changes in career decisions of internal medicine residents during training. Ann Intern Med. 2006;145:774-779.
19. Medicare Payment Advisory Commission. Report to the Congress: Medicare Payment Policy. March 2006. Available at: http://www.medpac.gov/publications/congressional_reports/Mar06_EntireReport.pdf. Accessed April 25, 2007.
20. Ginsburg PB, Berenson RA. Revising Medicare’s physician fee schedule–much activity, little change. N Engl J Med. 2007;356:1201-1203.
21. American College of Physicians. The Advanced Medical Home: A Patient-Centered, Physician-Guided Model of Health Care. 2006. Available at: http://www.acponline.org/advocacy/where_we_stand/policy/adv_med.pdf. Accessed May 13, 2007.
22. Steiner BD, Denham AC, Ashkin E, et al. Community care of North Carolina: improving care through community health networks. Ann Fam Med. 2008;in press.
23. Freudenheim M. Trying to save by increasing doctors’ fees. The New York Times. July 21, 2008. Available at: http://www.nytimes.com/2008/07/21/business/21medhome.html?_r=1&scp=1&sq=Trying%20to%20save%20by%20increasing%20doctors%20fees&st=cse&oref=slogin. Accessed August 1, 2008.
24. Orzang P. CBO. “Medicare’s Payments to Physicians: Option for Changing the Sustainable Growth Rate,” testimony before the Committee on Finance United States Senate. March 1, 2007. Available at: http://www.senate.gov/~finance/hearings/testimony/2007test/030107potest.pdf. Accessed May 13, 2007.
25. American Medical Association. RUC Medicare Medical Home Demonstration project recommendations. Available at: http://www.ama-assn.org/ama/pub/category/18528.html. Accessed August 1, 2008.
26. Pear R. Doctors press Senate to undo Medicare cuts. The New York Times. July 7, 2008. Available at: http://www.nytimes.com/2008/07/07/health/policy/07medicare.html?scp=1&sq=Doctors%20press%20senate%20to%20undo%20Medicare%20cuts&st=cse. Accessed August 1, 2008.