User login
ROUNDTABLE: PART 1 OF 2: Using mesh to repair prolapse calls for more than a kit—it takes skill
MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.
SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.
VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.
MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
Mesh kits for repairing prolapse are proliferating like crazy, just as they did for midurethral sling procedures. But mesh augmentation of prolapse surgeries requires more than a prepackaged assortment of tools and materials. In this article, moderator Mickey M. Karram, MD, and a panel of nationally recognized urogynecologists and urologists describe the literature on mesh augmentation and discuss indications, contraindications, techniques, applicable cases, and the considerable training required.
In Part 2, which will appear in the February issue of OBG Management, the panel tackles the thorny topic of complications, including erosion, extrusion, foreshortening of the vagina, dyspareunia, and pain. Their discussion focuses on ways to avoid these problems, and methods for correcting them.
Do we have enough data?
DR. KARRAM: To start, let’s quickly review the peer-reviewed literature on the use of mesh augmentation during surgery for pelvic organ prolapse.
DR. WALTERS: Until recently, most data concerned open abdominal sacrocolpopexy (ASC) using polypropylene or Merseline mesh. There is significant clinical experience with this operation, and multiple cohort studies show long-term cure rates of 78% to 100% for apical prolapse.1
At least two randomized controlled trials have compared open ASC with sutured vaginal colpopexy procedures, and ASC is certainly equal to—perhaps better than—all transvaginal sutured repairs.2,3
With ASC, most recurrences affect the distal half of the vagina and involve one or more of the following:
- anterior or posterior vaginal wall prolapse (or both)
- stress urinary incontinence (SUI)
- distal rectocele.1,3
Mesh erosion occurs in 3.4% of cases and is usually easily managed.1 Other complications, including bowel injury, tend to be related to access, regardless of whether the operation is performed via laparotomy or laparoscopy.
Robotic sacrocolpopexy has become popular in recent years, and we will probably see data on this approach as we gain experience.
When it comes to vaginal mesh kits, the peer-reviewed literature is just beginning to expand, with many studies being presented at international meetings. For anterior and, possibly, apical vaginal prolapse, the cure rate after use of a mesh kit appears to be as high as, or higher than, the rate for sutured repairs.4 This high rate of anatomic cure is balanced somewhat by additional cost and complications involving mesh and the kits.
For posterior vaginal wall prolapse and rectocele, I firmly believe, based on our research and that of others, that sutured repairs are superior to graft-augmented surgery.5
DR. KARRAM: What are the indications and contraindications for mesh augmentation of prolapse repair ( FIGURES 1 and 2 )?
DR. LUCENTE: I believe mesh is indicated in any patient in need of surgical repair of pelvic organ prolapse who is seeking optimal durability and is willing to accept the known risks of the surgery.
The issue becomes more complex when it comes to contraindications. Absolute contraindications are fairly obvious; they include medically unstable patients and those who may have an inactive infectious process within the pelvis or even undiagnosed abnormal uterine bleeding.
At our center, because the potential for dyspareunia and pelvic discomfort is our biggest concern, we have developed a profile of the patient who is more likely to develop these complaints. The profile includes any patient who has a chronic pain disorder of any type, but especially chronic pelvic pain disorders such as endometriosis and vulvodynia. Other risk factors appear to be a history of pelvic surgery involving any permanent material, suture or mesh, and young age.
So if we have a patient in her late 30s who has undergone reconstructive surgery using permanent sutures and who has an element of chronic pelvic pain, we would counsel her strongly to consider surgical options other than the use of synthetic mesh.
DR. WALTERS: The main indications for mesh-augmented prolapse repair are recurrent posthysterectomy vaginal vault prolapse, for which I usually perform ASC, and recurrent cystocele or anteriorapical prolapse, for which I use one of the anterior mesh kits.
I still think sutured repairs—by that, I mean uterosacral ligament or sacrospinous colpopexy with sutured rectocele repair—work best for recurrent posterior wall and posteriorapical prolapse. I don’t use mesh augmentation for rectocele.
The main contraindication to mesh augmentation, as I see it, is a history of mesh complications. If I am repairing a mesh complication such as erosion or pain, I do not place another mesh.
Medical issues that might increase mesh complications, such as diabetes, steroid use, or severe vaginal atrophy, would, at the very least, make me consider carefully whether mesh augmentation is appropriate. The literature is not clear on this, so mesh could still be used if the surgeon thinks it is necessary.
DR. KARRAM: I haven’t found a definitive indication for mesh augmentation. We have used biologic meshes empirically, but I am not convinced that they really add long-term durability, regardless of whether they are used in the anterior or posterior vaginal segment.
Our published durability rate for traditional suture-type repairs is in the range of 85% at 5 years out.6 Even if I assumed that mesh would give me 100% 5-year durability, this rate would have to be at the expense of some erosion, pain, and other complications unique to mesh. I do not think that the potential improvement in durability is worth these potential complications.
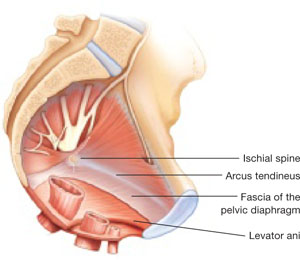
FIGURE 1 When the pelvic support system is intact, prolapse is rare
In the normal pelvis, organs are supported by a complex web of muscles, fascia, and connective tissue. 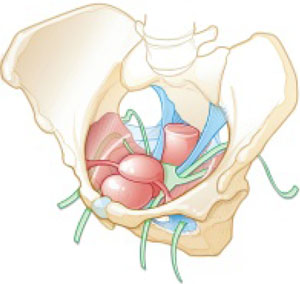
FIGURE 2 Mesh augmentation seeks to enhance the durability of repair
One type of mesh in final position. Mesh-augmented repair restores the vaginal apex and lends support to the walls of the vagina.
DR. KARRAM: If you are doing a lot of mesh repairs, you are obviously content with the results and feel that the few complications you are seeing are outweighed by the advantages mesh confers. How do you avoid extrusion and avert creation of a painful vagina?
DR. RAZ: Most of our cases are recurrent prolapse after failed vaginal or abdominal repair. I am indeed using a significant amount of soft polypropylene mesh for reconstructive procedures. As with the use of any other synthetic material, low-grade infection can develop after a few weeks or months. I use copious irrigation with antibiotic solution during reconstruction.
To avoid extrusion, I perform deep, rather than superficial, dissection of the vaginal wall to allow for better coverage of the mesh. For posterior mesh reconstruction, I cover the mesh with pararectal fascia to prevent erosion.
For mesh-augmented procedures, I cut the mesh myself in the operating room ( FIGURE 3 ). For a sling, I use a 10 cm × 1 cm soft polypropylene mesh. For a grade 3 or 4 cystocele, I use a trapezoid of soft polypropylene mesh with several points of fixation:
- at the sacrouterine ligament
- lateral to the obturator fascia
- distal to the bladder neck.
I always repair the vault at the same time.
For vault prolapse, I use a segment of soft polypropylene mesh in the shape of an apron with two arms (1 cm × 4 cm) and a central segment (4 cm × 7 cm). I support the vault using number 1-0 delayed absorbable suture and mesh. From outside the vaginal wall, in the posterolateral deep vaginal wall (inside the peritoneum), I incorporate the origin of the sacrouterine ligament and one arm of the mesh in the groove between the colon and levator ani, 15 cm from the introitus. I bring the suture 1 cm from the original entrance. A separate set of sutures brings the perirectal fascia together with the sacrouterine ligaments and perivesical fascia to close the peritoneal cavity. I tie the vault-suspension sutures, providing support to the cuff in a high posterior position (12 to 15 cm from the introitus).
In selected cases of significant recurrent rectocele, I use a rectangle of soft polypropylene mesh anchored to the origin of the sacrouterine ligament and distal to the perineal membrane. The mesh is covered by the pararectal fascia.
We have not seen vaginal, urethral, or bladder erosion in 1,800 cases of our distal urethral Prolene sling procedure using 10 cm × 1 cm soft mesh. In patients who have significant cystocele, vault prolapse, and recurrent rectocele, our vaginal erosion rate is 3%. We have never encountered rectal, bladder, or bowel perforation using our technique.
DR. LUCENTE: We often use mesh and are more than simply content with our results—we are extremely pleased, and so are our patients. Having said that, our techniques have definitely evolved over the past few years, as we’ve focused on how to decrease exposure and, more recently, optimize sexual function and vaginal comfort.
First, to avoid exposure, the most critical step is precise hydrodissection and distention of the true vesicovaginal space. This step can only be achieved through careful tactile guidance of the needle tip into the space, where it should remain while hydrodissection is performed. Always remember, sharp dissection “follows” hydrodissection. If you place the needle bevel within the vaginal wall, you will “split” the vaginal wall—as during standard colporrhaphy—which will lead to a high exposure rate.
Second, to avoid dyspareunia, it’s essential to pay close attention to POP-Q measurements, especially vaginal length, to ensure that the reconstruction restores the same length without foreshortening. This approach entails leaving the cervix in most patients who have a shorter vagina, and making sure that the mesh is secured above the ischial spine in younger, sexually active patients who have demonstrated a higher risk of postoperative deep, penetrating dyspareunia, compared with older, less sexually active patients.
Also paramount is to ensure that you have manually displaced the vagina inwardly as much as possible before deploying or setting the mesh. If you simply try to suture secure the mesh with the vagina incised open, without the ability to deploy the mesh with a closed, displaced vagina (to mimic deep penetration), it is difficult, if not impossible, to properly set the mesh for optimal comfort.
In the early days of midurethral pubovaginal slings using polypropylene, the adage was “looser is better than tighter.” This is even truer for transvaginal mesh.
DR. KARRAM: Dr. Walters, please describe your current surgical procedure of choice without mesh and explain why you haven’t adopted mesh for routine repairs.
DR. WALTERS: About 20% of my prolapse surgeries—usually for posthysterectomy or recurrent vaginal vault prolapse—involve ASC with placement of polypropylene mesh. I perform most of these cases through a Pfannenstiel incision, but I’ve also done them laparoscopically. Several of my partners perform ASC laparoscopically and robotically.
For the other 80% of my patients who have prolapse, I perform repairs transvaginally, usually using high bilateral uterosacralligament vaginal-vault suspension. We have learned to suture higher and slightly more medial on the uterosacral ligaments to attain greater vaginal depth and minimize ureteral obstruction. We use two or three sutures on each uterosacral ligament, usually a combination of permanent and delayed absorbable sutures.
I am also performing more sacrospinous ligament suspensions because this operation is being studied by the Pelvic Floor Disorders Network. Properly performed, it is an excellent surgery for apical prolapse. But, as with most of our surgeries for prolapse, recurrent anterior wall prolapse remains a problem.
Like you, Dr. Karram, we’ve studied our group’s anatomic and functional outcomes very carefully for more than 10 years and are mostly satisfied with our cure and complication rates. Although our anatomic outcomes with these surgeries are not always perfect, our reoperation rate for prolapse is only about 5%, with a high level of satisfaction in 88% to 92% of patients.
DR. RAZ: Unaugmented reconstruction fails in more than 30% of cases. Some patients who have significant prolapse and attenuated tissue think that this tissue will become healthier or stronger after reconstructive surgery, but that isn’t the case. In these situations, excision and plication make no clinical sense.
The problem is that we have yet to identify the ideal surrogate for poor-quality tissue. Most of us use polypropylene mesh in different variations. We need a better material that will be nonimmunogenic, well tolerated, and easily incorporated without erosion. Xenograft-like derivatives of dermis, or allografts such as cadaveric fascia, have failed over the long term because the body reabsorbs the graft without forming any new connective tissue.
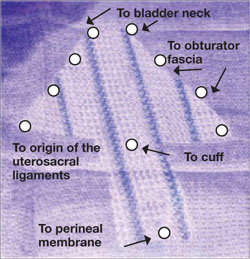
FIGURE 3 Mesh can be cut in the OR to custom-fit a patient
Hand-cut mesh and points of placement.
PHOTO: SHLOMO RAZ, MD
Is a kit a valuable aid?
DR. KARRAM: If a surgeon wants to augment a repair, what are the advantages of a packaged mesh kit, compared with simply cutting the mesh and performing surgery without a kit?
DR. WALTERS: The advantages of a packaged mesh kit are the convenience involved and the ability to consistently perform the same operation with the same product. That facilitates learning, teaching, and research. It also helps us understand the published literature a little better because “custom” prolapse repairs are operator-dependent and difficult to apply generally to a population of surgeons.
These advantages are most clearly apparent with midurethral sling mesh kits, which have almost revolutionized surgery for stress incontinence. I don’t believe mesh kits for prolapse are there yet, but they certainly have potential.
DR. RAZ: I’m opposed to the use of kits. They are industry-driven. One company has made $1 billion selling them. Imagine a patient who undergoes placement of a sling kit ($1,000), cystocele kit ($1,500), and posterior mesh kit ($1,500). How can our healthcare system sustain this burden, especially when there is no real evidence that a kit improves the operation, and given the incredible complication rate that we see?
Moreover, the kits contain a single-use needle and passer and a precut segment of polypropylene mesh. But every patient is different and requires a unique size or shape of mesh. I don’t believe that a surgeon who knows pelvic anatomy needs a kit to perform mesh-augmented reconstruction. We can buy the same segment of mesh for $200 to $400, cut it as needed, and perform the same operation advertised by industry.
For surgeons who prefer a kit, the tools that are included should be made reusable.
DR. LUCENTE: In my opinion, the primary advantage of a commercially available transvaginal mesh delivery system—notice, I avoided the word “kit,” because I think there are plenty of negative connotations associated with it—is the ability to deliver the mesh in a “tension”-free manner.
One alternative that many people pursue is cutting the mesh to size and using sutures to hold it in place while tissue ingrowth occurs. However, the hernia literature suggests that suturing mesh in place increases the risk of postoperative discomfort at the site of implantation. The true cause of the discomfort remains unclear, but it is thought to arise from nerve tethering or traction at the pre-committed points of attachment before the host tissue and mesh interface have adjusted or settled with tissue ingrowth.
All neuropathic complications of mesh implantation have been shown in the current hernia literature to be increased with the use of sutures.7 Also, as previously mentioned, it is extremely difficult to set or adjust the mesh with the vaginal incision remaining “open,” which is a downside to suture techniques.
What training is necessary to use a kit?
DR. KARRAM: Mesh kits are aggressively promoted by industry, with close to half a dozen different kits to be available soon. What is the minimum amount of training one should have before utilizing these kits?
DR. WALTERS: The surgeon should at least know how to perform traditional sutured prolapse repairs and SUI surgery and be able to perform cystoscopy. Ideally, the surgeon should undergo training on a cadaver with a skilled and experienced user of the mesh kit. The surgeon also should carefully review the risks and benefits of mesh kits with the patient and inform the patient that he or she is in the early learning curve of a particular surgery. The informed patient should have a right to refuse mesh-augmented prolapse surgery after the consent process.
DR. LUCENTE: I’m glad you asked this question. I strongly believe that surgical expertise and proficiency within gynecology need to be more effectively addressed by us all. We have a situation in our field in which techniques and technology are widening the gap between what is possible and what the surgeon is comfortable doing safely.
It’s incumbent on all of us, especially those who are in a leadership position as a chairperson or chief of a division, to work with our physician staff and faculty to optimize surgical skill and patient outcomes, including safety, with new technologies.
As for the minimal amount of training needed, that’s extremely variable. It depends on the current skill set of the physician and his or her ability to pick up the mechanics of the surgery as it is taught through a cadaver lab or preceptorship. It’s regrettable that some physicians lack the objectivity and insight to judge their own skill set. This, again, is the time for a chairperson or chief of a division to step up to the plate and ensure proper credentialing and demonstration of proficiency.
It is unrealistic to expect industry to decide who should or should not utilize this truly breakthrough technology. That is our responsibility as physicians.
DR. KARRAM: At a minimum, I think any surgeon utilizing a kit should have a firm understanding of pelvic floor anatomy and experience performing traditional repairs:
- intraperitoneal procedures such as Mc-Call culdoplasty and uterosacral suspension
- sacrospinous suspension
- retropubic procedures and anti-incontinence operations such as pubovaginal slings.
This three-dimensional understanding of the pelvic floor is mandatory if one is to assume that blind passage of trocars through potentially dangerous spaces is the wave of the future.
DR. RAZ: You need to be a pelvic surgeon, know your anatomy, and know how to manage complications if you are going to use one of these kits. You should stick to the surgery that works best in your hands. Industry cannot teach you to be a good pelvic surgeon; it takes lifelong experience.
DR. KARRAM: If you have a patient who is sexually inactive with pelvic organ prolapse, would you prefer a mesh repair or an obliterative procedure? And why?
DR. WALTERS: If the patient is sexually inactive—especially if she is older and definitely will not be in the future—it makes absolutely no sense to perform a mesh-augmented repair. A traditional, somewhat tight, sutured repair works fine in this setting and carries very low risk.
In fact, our group and others have found that, in carefully selected patients, partial colpectomy and colpocleisis procedures (without grafts) have among the highest cure and satisfaction rates of all surgeries we perform for prolapse; they also have relatively low risk.8 Recurrent prolapse after an obliterative procedure is rare; most of the dissatisfaction relates to postoperative voiding difficulties or persistent or de novo urinary incontinence.
DR. KARRAM: I also prefer an obliterative procedure. I see no reason to bring in the cost and potential for complications that mesh repair entails. An obliterative procedure should produce an anatomic success rate close to 100%, with minimal complications. It also can be performed quickly with minimal anesthesia and convalescence.
DR. LUCENTE: My response is based on a clinical study that my associate, Dr. Miles Murphy, has performed, comparing a transvaginal mesh procedure with a LaForte operation for severe pelvic organ prolapse.9 Both patient groups were well satisfied with the result, and success rates were comparable. However, the group that underwent the transvaginal mesh procedure had a shorter operative time.
As a result of these studies, we tend to prefer transvaginal mesh repair. Even though the woman may be sexually inactive, the procedure preserves vaginal function, and we all know that life has a way of being unpredictable. Her situation may change so that she once again desires sexual function.
However, for a very elderly woman—one in her late 80s or 90s—who has severe or extreme prolapse with a very large procidentia and vaginal length measuring, say, 13 cm beyond the introitus, I do prefer an obliterative procedure.
DR. RAZ: I agree. I would not offer a sexually inactive patient an obliterative procedure. You never know what the future will hold.
Mesh repair can be performed safely, provided the surgeon has good knowledge of anatomic landmarks and knows how to manage any potential complications that may arise.
1. Nygaard IE, McCreery R, Brubaker L, et al. Pelvic Floor Disorders Network. Abdominal sacrocolpopexy: a comprehensive review. Obstet Gynecol. 2004;104:805-823.
2. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421;discussion 1421-1422.
3. Maher CF, Qatawneh AM, Dwyer PL, Carey MP, Cornish A, Schluter PJ. Abdominal sacral colpopexy or vaginal sacrospinous colpopexy for vaginal vault prolapse: a prospective randomized study. Am J Obstet Gynecol. 2004;190:20-26.
4. Murphy M. Society of Gynecologic Surgeons Systematic Review Group. Clinical practice guidelines on vaginal graft use from the Society of Gynecologic Surgeons. Obstet Gynecol. 2008;112:1123-1130.
5. Paraiso MF, Barber MD, Muir TW, Walters MD. Rectocele repair: a randomized trial of three surgical procedures including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
6. Silva WA, Pauls RN, Segal JL, Rooney CM, Kleeman SD, Karram MM. Uterosacral ligament vault suspension: five-year outcomes. Obstet Gynecol 2006;108:255-263.
7. EU Hernia Trialists Collaboration. Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg. 2002;235:322-332.
8. Barber MD, Amundsen C, Paraiso MFR, Weidner A, Romero A, Walters MD. Quality of life after surgery for genital prolapse in elderly women: obliterative and reconstructive surgery. Int Urogynecol J. 2007;18:799-806.
9. Murphy M, van Raalte H, Mercurio E, Haff R, Wiseman B, Lucente VR. Incontinence-related quality of life and sexual function following the tension-free vaginal tape vs the “inside-out” tension-free vaginal tape obturator. Int Urogynecol J. 2008;19:481-487.
MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.
SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.
VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.
MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
Mesh kits for repairing prolapse are proliferating like crazy, just as they did for midurethral sling procedures. But mesh augmentation of prolapse surgeries requires more than a prepackaged assortment of tools and materials. In this article, moderator Mickey M. Karram, MD, and a panel of nationally recognized urogynecologists and urologists describe the literature on mesh augmentation and discuss indications, contraindications, techniques, applicable cases, and the considerable training required.
In Part 2, which will appear in the February issue of OBG Management, the panel tackles the thorny topic of complications, including erosion, extrusion, foreshortening of the vagina, dyspareunia, and pain. Their discussion focuses on ways to avoid these problems, and methods for correcting them.
Do we have enough data?
DR. KARRAM: To start, let’s quickly review the peer-reviewed literature on the use of mesh augmentation during surgery for pelvic organ prolapse.
DR. WALTERS: Until recently, most data concerned open abdominal sacrocolpopexy (ASC) using polypropylene or Merseline mesh. There is significant clinical experience with this operation, and multiple cohort studies show long-term cure rates of 78% to 100% for apical prolapse.1
At least two randomized controlled trials have compared open ASC with sutured vaginal colpopexy procedures, and ASC is certainly equal to—perhaps better than—all transvaginal sutured repairs.2,3
With ASC, most recurrences affect the distal half of the vagina and involve one or more of the following:
- anterior or posterior vaginal wall prolapse (or both)
- stress urinary incontinence (SUI)
- distal rectocele.1,3
Mesh erosion occurs in 3.4% of cases and is usually easily managed.1 Other complications, including bowel injury, tend to be related to access, regardless of whether the operation is performed via laparotomy or laparoscopy.
Robotic sacrocolpopexy has become popular in recent years, and we will probably see data on this approach as we gain experience.
When it comes to vaginal mesh kits, the peer-reviewed literature is just beginning to expand, with many studies being presented at international meetings. For anterior and, possibly, apical vaginal prolapse, the cure rate after use of a mesh kit appears to be as high as, or higher than, the rate for sutured repairs.4 This high rate of anatomic cure is balanced somewhat by additional cost and complications involving mesh and the kits.
For posterior vaginal wall prolapse and rectocele, I firmly believe, based on our research and that of others, that sutured repairs are superior to graft-augmented surgery.5
DR. KARRAM: What are the indications and contraindications for mesh augmentation of prolapse repair ( FIGURES 1 and 2 )?
DR. LUCENTE: I believe mesh is indicated in any patient in need of surgical repair of pelvic organ prolapse who is seeking optimal durability and is willing to accept the known risks of the surgery.
The issue becomes more complex when it comes to contraindications. Absolute contraindications are fairly obvious; they include medically unstable patients and those who may have an inactive infectious process within the pelvis or even undiagnosed abnormal uterine bleeding.
At our center, because the potential for dyspareunia and pelvic discomfort is our biggest concern, we have developed a profile of the patient who is more likely to develop these complaints. The profile includes any patient who has a chronic pain disorder of any type, but especially chronic pelvic pain disorders such as endometriosis and vulvodynia. Other risk factors appear to be a history of pelvic surgery involving any permanent material, suture or mesh, and young age.
So if we have a patient in her late 30s who has undergone reconstructive surgery using permanent sutures and who has an element of chronic pelvic pain, we would counsel her strongly to consider surgical options other than the use of synthetic mesh.
DR. WALTERS: The main indications for mesh-augmented prolapse repair are recurrent posthysterectomy vaginal vault prolapse, for which I usually perform ASC, and recurrent cystocele or anteriorapical prolapse, for which I use one of the anterior mesh kits.
I still think sutured repairs—by that, I mean uterosacral ligament or sacrospinous colpopexy with sutured rectocele repair—work best for recurrent posterior wall and posteriorapical prolapse. I don’t use mesh augmentation for rectocele.
The main contraindication to mesh augmentation, as I see it, is a history of mesh complications. If I am repairing a mesh complication such as erosion or pain, I do not place another mesh.
Medical issues that might increase mesh complications, such as diabetes, steroid use, or severe vaginal atrophy, would, at the very least, make me consider carefully whether mesh augmentation is appropriate. The literature is not clear on this, so mesh could still be used if the surgeon thinks it is necessary.
DR. KARRAM: I haven’t found a definitive indication for mesh augmentation. We have used biologic meshes empirically, but I am not convinced that they really add long-term durability, regardless of whether they are used in the anterior or posterior vaginal segment.
Our published durability rate for traditional suture-type repairs is in the range of 85% at 5 years out.6 Even if I assumed that mesh would give me 100% 5-year durability, this rate would have to be at the expense of some erosion, pain, and other complications unique to mesh. I do not think that the potential improvement in durability is worth these potential complications.

FIGURE 1 When the pelvic support system is intact, prolapse is rare
In the normal pelvis, organs are supported by a complex web of muscles, fascia, and connective tissue. 
FIGURE 2 Mesh augmentation seeks to enhance the durability of repair
One type of mesh in final position. Mesh-augmented repair restores the vaginal apex and lends support to the walls of the vagina.
DR. KARRAM: If you are doing a lot of mesh repairs, you are obviously content with the results and feel that the few complications you are seeing are outweighed by the advantages mesh confers. How do you avoid extrusion and avert creation of a painful vagina?
DR. RAZ: Most of our cases are recurrent prolapse after failed vaginal or abdominal repair. I am indeed using a significant amount of soft polypropylene mesh for reconstructive procedures. As with the use of any other synthetic material, low-grade infection can develop after a few weeks or months. I use copious irrigation with antibiotic solution during reconstruction.
To avoid extrusion, I perform deep, rather than superficial, dissection of the vaginal wall to allow for better coverage of the mesh. For posterior mesh reconstruction, I cover the mesh with pararectal fascia to prevent erosion.
For mesh-augmented procedures, I cut the mesh myself in the operating room ( FIGURE 3 ). For a sling, I use a 10 cm × 1 cm soft polypropylene mesh. For a grade 3 or 4 cystocele, I use a trapezoid of soft polypropylene mesh with several points of fixation:
- at the sacrouterine ligament
- lateral to the obturator fascia
- distal to the bladder neck.
I always repair the vault at the same time.
For vault prolapse, I use a segment of soft polypropylene mesh in the shape of an apron with two arms (1 cm × 4 cm) and a central segment (4 cm × 7 cm). I support the vault using number 1-0 delayed absorbable suture and mesh. From outside the vaginal wall, in the posterolateral deep vaginal wall (inside the peritoneum), I incorporate the origin of the sacrouterine ligament and one arm of the mesh in the groove between the colon and levator ani, 15 cm from the introitus. I bring the suture 1 cm from the original entrance. A separate set of sutures brings the perirectal fascia together with the sacrouterine ligaments and perivesical fascia to close the peritoneal cavity. I tie the vault-suspension sutures, providing support to the cuff in a high posterior position (12 to 15 cm from the introitus).
In selected cases of significant recurrent rectocele, I use a rectangle of soft polypropylene mesh anchored to the origin of the sacrouterine ligament and distal to the perineal membrane. The mesh is covered by the pararectal fascia.
We have not seen vaginal, urethral, or bladder erosion in 1,800 cases of our distal urethral Prolene sling procedure using 10 cm × 1 cm soft mesh. In patients who have significant cystocele, vault prolapse, and recurrent rectocele, our vaginal erosion rate is 3%. We have never encountered rectal, bladder, or bowel perforation using our technique.
DR. LUCENTE: We often use mesh and are more than simply content with our results—we are extremely pleased, and so are our patients. Having said that, our techniques have definitely evolved over the past few years, as we’ve focused on how to decrease exposure and, more recently, optimize sexual function and vaginal comfort.
First, to avoid exposure, the most critical step is precise hydrodissection and distention of the true vesicovaginal space. This step can only be achieved through careful tactile guidance of the needle tip into the space, where it should remain while hydrodissection is performed. Always remember, sharp dissection “follows” hydrodissection. If you place the needle bevel within the vaginal wall, you will “split” the vaginal wall—as during standard colporrhaphy—which will lead to a high exposure rate.
Second, to avoid dyspareunia, it’s essential to pay close attention to POP-Q measurements, especially vaginal length, to ensure that the reconstruction restores the same length without foreshortening. This approach entails leaving the cervix in most patients who have a shorter vagina, and making sure that the mesh is secured above the ischial spine in younger, sexually active patients who have demonstrated a higher risk of postoperative deep, penetrating dyspareunia, compared with older, less sexually active patients.
Also paramount is to ensure that you have manually displaced the vagina inwardly as much as possible before deploying or setting the mesh. If you simply try to suture secure the mesh with the vagina incised open, without the ability to deploy the mesh with a closed, displaced vagina (to mimic deep penetration), it is difficult, if not impossible, to properly set the mesh for optimal comfort.
In the early days of midurethral pubovaginal slings using polypropylene, the adage was “looser is better than tighter.” This is even truer for transvaginal mesh.
DR. KARRAM: Dr. Walters, please describe your current surgical procedure of choice without mesh and explain why you haven’t adopted mesh for routine repairs.
DR. WALTERS: About 20% of my prolapse surgeries—usually for posthysterectomy or recurrent vaginal vault prolapse—involve ASC with placement of polypropylene mesh. I perform most of these cases through a Pfannenstiel incision, but I’ve also done them laparoscopically. Several of my partners perform ASC laparoscopically and robotically.
For the other 80% of my patients who have prolapse, I perform repairs transvaginally, usually using high bilateral uterosacralligament vaginal-vault suspension. We have learned to suture higher and slightly more medial on the uterosacral ligaments to attain greater vaginal depth and minimize ureteral obstruction. We use two or three sutures on each uterosacral ligament, usually a combination of permanent and delayed absorbable sutures.
I am also performing more sacrospinous ligament suspensions because this operation is being studied by the Pelvic Floor Disorders Network. Properly performed, it is an excellent surgery for apical prolapse. But, as with most of our surgeries for prolapse, recurrent anterior wall prolapse remains a problem.
Like you, Dr. Karram, we’ve studied our group’s anatomic and functional outcomes very carefully for more than 10 years and are mostly satisfied with our cure and complication rates. Although our anatomic outcomes with these surgeries are not always perfect, our reoperation rate for prolapse is only about 5%, with a high level of satisfaction in 88% to 92% of patients.
DR. RAZ: Unaugmented reconstruction fails in more than 30% of cases. Some patients who have significant prolapse and attenuated tissue think that this tissue will become healthier or stronger after reconstructive surgery, but that isn’t the case. In these situations, excision and plication make no clinical sense.
The problem is that we have yet to identify the ideal surrogate for poor-quality tissue. Most of us use polypropylene mesh in different variations. We need a better material that will be nonimmunogenic, well tolerated, and easily incorporated without erosion. Xenograft-like derivatives of dermis, or allografts such as cadaveric fascia, have failed over the long term because the body reabsorbs the graft without forming any new connective tissue.

FIGURE 3 Mesh can be cut in the OR to custom-fit a patient
Hand-cut mesh and points of placement.
PHOTO: SHLOMO RAZ, MD
Is a kit a valuable aid?
DR. KARRAM: If a surgeon wants to augment a repair, what are the advantages of a packaged mesh kit, compared with simply cutting the mesh and performing surgery without a kit?
DR. WALTERS: The advantages of a packaged mesh kit are the convenience involved and the ability to consistently perform the same operation with the same product. That facilitates learning, teaching, and research. It also helps us understand the published literature a little better because “custom” prolapse repairs are operator-dependent and difficult to apply generally to a population of surgeons.
These advantages are most clearly apparent with midurethral sling mesh kits, which have almost revolutionized surgery for stress incontinence. I don’t believe mesh kits for prolapse are there yet, but they certainly have potential.
DR. RAZ: I’m opposed to the use of kits. They are industry-driven. One company has made $1 billion selling them. Imagine a patient who undergoes placement of a sling kit ($1,000), cystocele kit ($1,500), and posterior mesh kit ($1,500). How can our healthcare system sustain this burden, especially when there is no real evidence that a kit improves the operation, and given the incredible complication rate that we see?
Moreover, the kits contain a single-use needle and passer and a precut segment of polypropylene mesh. But every patient is different and requires a unique size or shape of mesh. I don’t believe that a surgeon who knows pelvic anatomy needs a kit to perform mesh-augmented reconstruction. We can buy the same segment of mesh for $200 to $400, cut it as needed, and perform the same operation advertised by industry.
For surgeons who prefer a kit, the tools that are included should be made reusable.
DR. LUCENTE: In my opinion, the primary advantage of a commercially available transvaginal mesh delivery system—notice, I avoided the word “kit,” because I think there are plenty of negative connotations associated with it—is the ability to deliver the mesh in a “tension”-free manner.
One alternative that many people pursue is cutting the mesh to size and using sutures to hold it in place while tissue ingrowth occurs. However, the hernia literature suggests that suturing mesh in place increases the risk of postoperative discomfort at the site of implantation. The true cause of the discomfort remains unclear, but it is thought to arise from nerve tethering or traction at the pre-committed points of attachment before the host tissue and mesh interface have adjusted or settled with tissue ingrowth.
All neuropathic complications of mesh implantation have been shown in the current hernia literature to be increased with the use of sutures.7 Also, as previously mentioned, it is extremely difficult to set or adjust the mesh with the vaginal incision remaining “open,” which is a downside to suture techniques.
What training is necessary to use a kit?
DR. KARRAM: Mesh kits are aggressively promoted by industry, with close to half a dozen different kits to be available soon. What is the minimum amount of training one should have before utilizing these kits?
DR. WALTERS: The surgeon should at least know how to perform traditional sutured prolapse repairs and SUI surgery and be able to perform cystoscopy. Ideally, the surgeon should undergo training on a cadaver with a skilled and experienced user of the mesh kit. The surgeon also should carefully review the risks and benefits of mesh kits with the patient and inform the patient that he or she is in the early learning curve of a particular surgery. The informed patient should have a right to refuse mesh-augmented prolapse surgery after the consent process.
DR. LUCENTE: I’m glad you asked this question. I strongly believe that surgical expertise and proficiency within gynecology need to be more effectively addressed by us all. We have a situation in our field in which techniques and technology are widening the gap between what is possible and what the surgeon is comfortable doing safely.
It’s incumbent on all of us, especially those who are in a leadership position as a chairperson or chief of a division, to work with our physician staff and faculty to optimize surgical skill and patient outcomes, including safety, with new technologies.
As for the minimal amount of training needed, that’s extremely variable. It depends on the current skill set of the physician and his or her ability to pick up the mechanics of the surgery as it is taught through a cadaver lab or preceptorship. It’s regrettable that some physicians lack the objectivity and insight to judge their own skill set. This, again, is the time for a chairperson or chief of a division to step up to the plate and ensure proper credentialing and demonstration of proficiency.
It is unrealistic to expect industry to decide who should or should not utilize this truly breakthrough technology. That is our responsibility as physicians.
DR. KARRAM: At a minimum, I think any surgeon utilizing a kit should have a firm understanding of pelvic floor anatomy and experience performing traditional repairs:
- intraperitoneal procedures such as Mc-Call culdoplasty and uterosacral suspension
- sacrospinous suspension
- retropubic procedures and anti-incontinence operations such as pubovaginal slings.
This three-dimensional understanding of the pelvic floor is mandatory if one is to assume that blind passage of trocars through potentially dangerous spaces is the wave of the future.
DR. RAZ: You need to be a pelvic surgeon, know your anatomy, and know how to manage complications if you are going to use one of these kits. You should stick to the surgery that works best in your hands. Industry cannot teach you to be a good pelvic surgeon; it takes lifelong experience.
DR. KARRAM: If you have a patient who is sexually inactive with pelvic organ prolapse, would you prefer a mesh repair or an obliterative procedure? And why?
DR. WALTERS: If the patient is sexually inactive—especially if she is older and definitely will not be in the future—it makes absolutely no sense to perform a mesh-augmented repair. A traditional, somewhat tight, sutured repair works fine in this setting and carries very low risk.
In fact, our group and others have found that, in carefully selected patients, partial colpectomy and colpocleisis procedures (without grafts) have among the highest cure and satisfaction rates of all surgeries we perform for prolapse; they also have relatively low risk.8 Recurrent prolapse after an obliterative procedure is rare; most of the dissatisfaction relates to postoperative voiding difficulties or persistent or de novo urinary incontinence.
DR. KARRAM: I also prefer an obliterative procedure. I see no reason to bring in the cost and potential for complications that mesh repair entails. An obliterative procedure should produce an anatomic success rate close to 100%, with minimal complications. It also can be performed quickly with minimal anesthesia and convalescence.
DR. LUCENTE: My response is based on a clinical study that my associate, Dr. Miles Murphy, has performed, comparing a transvaginal mesh procedure with a LaForte operation for severe pelvic organ prolapse.9 Both patient groups were well satisfied with the result, and success rates were comparable. However, the group that underwent the transvaginal mesh procedure had a shorter operative time.
As a result of these studies, we tend to prefer transvaginal mesh repair. Even though the woman may be sexually inactive, the procedure preserves vaginal function, and we all know that life has a way of being unpredictable. Her situation may change so that she once again desires sexual function.
However, for a very elderly woman—one in her late 80s or 90s—who has severe or extreme prolapse with a very large procidentia and vaginal length measuring, say, 13 cm beyond the introitus, I do prefer an obliterative procedure.
DR. RAZ: I agree. I would not offer a sexually inactive patient an obliterative procedure. You never know what the future will hold.
Mesh repair can be performed safely, provided the surgeon has good knowledge of anatomic landmarks and knows how to manage any potential complications that may arise.
MICKEY M. KARRAM, MD, moderator, is Director of Urogynecology at Good Samaritan Hospital and Voluntary Professor of ObGyn at the University of Cincinnati School of Medicine in Cincinnati, Ohio.
SHLOMO RAZ, MD, is Professor of Urology and Chief of Pelvic Medicine and Reconstructive Urology at UCLA School of Medicine in Los Angeles.
VINCENT LUCENTE, MD, MBA, is Founder and Director of the Institute for Female Pelvic Medicine and Reconstructive Surgery in Allentown, Pa, and Clinical Professor of ObGyn at Temple University School of Medicine in Philadelphia.
MARK D. WALTERS, MD, is Professor and Vice Chair of Gynecology, Section of Urogynecology and Reconstructive Pelvic Surgery, Department of Obstetrics and Gynecology, at the Cleveland Clinic in Cleveland, Ohio.
Mesh kits for repairing prolapse are proliferating like crazy, just as they did for midurethral sling procedures. But mesh augmentation of prolapse surgeries requires more than a prepackaged assortment of tools and materials. In this article, moderator Mickey M. Karram, MD, and a panel of nationally recognized urogynecologists and urologists describe the literature on mesh augmentation and discuss indications, contraindications, techniques, applicable cases, and the considerable training required.
In Part 2, which will appear in the February issue of OBG Management, the panel tackles the thorny topic of complications, including erosion, extrusion, foreshortening of the vagina, dyspareunia, and pain. Their discussion focuses on ways to avoid these problems, and methods for correcting them.
Do we have enough data?
DR. KARRAM: To start, let’s quickly review the peer-reviewed literature on the use of mesh augmentation during surgery for pelvic organ prolapse.
DR. WALTERS: Until recently, most data concerned open abdominal sacrocolpopexy (ASC) using polypropylene or Merseline mesh. There is significant clinical experience with this operation, and multiple cohort studies show long-term cure rates of 78% to 100% for apical prolapse.1
At least two randomized controlled trials have compared open ASC with sutured vaginal colpopexy procedures, and ASC is certainly equal to—perhaps better than—all transvaginal sutured repairs.2,3
With ASC, most recurrences affect the distal half of the vagina and involve one or more of the following:
- anterior or posterior vaginal wall prolapse (or both)
- stress urinary incontinence (SUI)
- distal rectocele.1,3
Mesh erosion occurs in 3.4% of cases and is usually easily managed.1 Other complications, including bowel injury, tend to be related to access, regardless of whether the operation is performed via laparotomy or laparoscopy.
Robotic sacrocolpopexy has become popular in recent years, and we will probably see data on this approach as we gain experience.
When it comes to vaginal mesh kits, the peer-reviewed literature is just beginning to expand, with many studies being presented at international meetings. For anterior and, possibly, apical vaginal prolapse, the cure rate after use of a mesh kit appears to be as high as, or higher than, the rate for sutured repairs.4 This high rate of anatomic cure is balanced somewhat by additional cost and complications involving mesh and the kits.
For posterior vaginal wall prolapse and rectocele, I firmly believe, based on our research and that of others, that sutured repairs are superior to graft-augmented surgery.5
DR. KARRAM: What are the indications and contraindications for mesh augmentation of prolapse repair ( FIGURES 1 and 2 )?
DR. LUCENTE: I believe mesh is indicated in any patient in need of surgical repair of pelvic organ prolapse who is seeking optimal durability and is willing to accept the known risks of the surgery.
The issue becomes more complex when it comes to contraindications. Absolute contraindications are fairly obvious; they include medically unstable patients and those who may have an inactive infectious process within the pelvis or even undiagnosed abnormal uterine bleeding.
At our center, because the potential for dyspareunia and pelvic discomfort is our biggest concern, we have developed a profile of the patient who is more likely to develop these complaints. The profile includes any patient who has a chronic pain disorder of any type, but especially chronic pelvic pain disorders such as endometriosis and vulvodynia. Other risk factors appear to be a history of pelvic surgery involving any permanent material, suture or mesh, and young age.
So if we have a patient in her late 30s who has undergone reconstructive surgery using permanent sutures and who has an element of chronic pelvic pain, we would counsel her strongly to consider surgical options other than the use of synthetic mesh.
DR. WALTERS: The main indications for mesh-augmented prolapse repair are recurrent posthysterectomy vaginal vault prolapse, for which I usually perform ASC, and recurrent cystocele or anteriorapical prolapse, for which I use one of the anterior mesh kits.
I still think sutured repairs—by that, I mean uterosacral ligament or sacrospinous colpopexy with sutured rectocele repair—work best for recurrent posterior wall and posteriorapical prolapse. I don’t use mesh augmentation for rectocele.
The main contraindication to mesh augmentation, as I see it, is a history of mesh complications. If I am repairing a mesh complication such as erosion or pain, I do not place another mesh.
Medical issues that might increase mesh complications, such as diabetes, steroid use, or severe vaginal atrophy, would, at the very least, make me consider carefully whether mesh augmentation is appropriate. The literature is not clear on this, so mesh could still be used if the surgeon thinks it is necessary.
DR. KARRAM: I haven’t found a definitive indication for mesh augmentation. We have used biologic meshes empirically, but I am not convinced that they really add long-term durability, regardless of whether they are used in the anterior or posterior vaginal segment.
Our published durability rate for traditional suture-type repairs is in the range of 85% at 5 years out.6 Even if I assumed that mesh would give me 100% 5-year durability, this rate would have to be at the expense of some erosion, pain, and other complications unique to mesh. I do not think that the potential improvement in durability is worth these potential complications.

FIGURE 1 When the pelvic support system is intact, prolapse is rare
In the normal pelvis, organs are supported by a complex web of muscles, fascia, and connective tissue. 
FIGURE 2 Mesh augmentation seeks to enhance the durability of repair
One type of mesh in final position. Mesh-augmented repair restores the vaginal apex and lends support to the walls of the vagina.
DR. KARRAM: If you are doing a lot of mesh repairs, you are obviously content with the results and feel that the few complications you are seeing are outweighed by the advantages mesh confers. How do you avoid extrusion and avert creation of a painful vagina?
DR. RAZ: Most of our cases are recurrent prolapse after failed vaginal or abdominal repair. I am indeed using a significant amount of soft polypropylene mesh for reconstructive procedures. As with the use of any other synthetic material, low-grade infection can develop after a few weeks or months. I use copious irrigation with antibiotic solution during reconstruction.
To avoid extrusion, I perform deep, rather than superficial, dissection of the vaginal wall to allow for better coverage of the mesh. For posterior mesh reconstruction, I cover the mesh with pararectal fascia to prevent erosion.
For mesh-augmented procedures, I cut the mesh myself in the operating room ( FIGURE 3 ). For a sling, I use a 10 cm × 1 cm soft polypropylene mesh. For a grade 3 or 4 cystocele, I use a trapezoid of soft polypropylene mesh with several points of fixation:
- at the sacrouterine ligament
- lateral to the obturator fascia
- distal to the bladder neck.
I always repair the vault at the same time.
For vault prolapse, I use a segment of soft polypropylene mesh in the shape of an apron with two arms (1 cm × 4 cm) and a central segment (4 cm × 7 cm). I support the vault using number 1-0 delayed absorbable suture and mesh. From outside the vaginal wall, in the posterolateral deep vaginal wall (inside the peritoneum), I incorporate the origin of the sacrouterine ligament and one arm of the mesh in the groove between the colon and levator ani, 15 cm from the introitus. I bring the suture 1 cm from the original entrance. A separate set of sutures brings the perirectal fascia together with the sacrouterine ligaments and perivesical fascia to close the peritoneal cavity. I tie the vault-suspension sutures, providing support to the cuff in a high posterior position (12 to 15 cm from the introitus).
In selected cases of significant recurrent rectocele, I use a rectangle of soft polypropylene mesh anchored to the origin of the sacrouterine ligament and distal to the perineal membrane. The mesh is covered by the pararectal fascia.
We have not seen vaginal, urethral, or bladder erosion in 1,800 cases of our distal urethral Prolene sling procedure using 10 cm × 1 cm soft mesh. In patients who have significant cystocele, vault prolapse, and recurrent rectocele, our vaginal erosion rate is 3%. We have never encountered rectal, bladder, or bowel perforation using our technique.
DR. LUCENTE: We often use mesh and are more than simply content with our results—we are extremely pleased, and so are our patients. Having said that, our techniques have definitely evolved over the past few years, as we’ve focused on how to decrease exposure and, more recently, optimize sexual function and vaginal comfort.
First, to avoid exposure, the most critical step is precise hydrodissection and distention of the true vesicovaginal space. This step can only be achieved through careful tactile guidance of the needle tip into the space, where it should remain while hydrodissection is performed. Always remember, sharp dissection “follows” hydrodissection. If you place the needle bevel within the vaginal wall, you will “split” the vaginal wall—as during standard colporrhaphy—which will lead to a high exposure rate.
Second, to avoid dyspareunia, it’s essential to pay close attention to POP-Q measurements, especially vaginal length, to ensure that the reconstruction restores the same length without foreshortening. This approach entails leaving the cervix in most patients who have a shorter vagina, and making sure that the mesh is secured above the ischial spine in younger, sexually active patients who have demonstrated a higher risk of postoperative deep, penetrating dyspareunia, compared with older, less sexually active patients.
Also paramount is to ensure that you have manually displaced the vagina inwardly as much as possible before deploying or setting the mesh. If you simply try to suture secure the mesh with the vagina incised open, without the ability to deploy the mesh with a closed, displaced vagina (to mimic deep penetration), it is difficult, if not impossible, to properly set the mesh for optimal comfort.
In the early days of midurethral pubovaginal slings using polypropylene, the adage was “looser is better than tighter.” This is even truer for transvaginal mesh.
DR. KARRAM: Dr. Walters, please describe your current surgical procedure of choice without mesh and explain why you haven’t adopted mesh for routine repairs.
DR. WALTERS: About 20% of my prolapse surgeries—usually for posthysterectomy or recurrent vaginal vault prolapse—involve ASC with placement of polypropylene mesh. I perform most of these cases through a Pfannenstiel incision, but I’ve also done them laparoscopically. Several of my partners perform ASC laparoscopically and robotically.
For the other 80% of my patients who have prolapse, I perform repairs transvaginally, usually using high bilateral uterosacralligament vaginal-vault suspension. We have learned to suture higher and slightly more medial on the uterosacral ligaments to attain greater vaginal depth and minimize ureteral obstruction. We use two or three sutures on each uterosacral ligament, usually a combination of permanent and delayed absorbable sutures.
I am also performing more sacrospinous ligament suspensions because this operation is being studied by the Pelvic Floor Disorders Network. Properly performed, it is an excellent surgery for apical prolapse. But, as with most of our surgeries for prolapse, recurrent anterior wall prolapse remains a problem.
Like you, Dr. Karram, we’ve studied our group’s anatomic and functional outcomes very carefully for more than 10 years and are mostly satisfied with our cure and complication rates. Although our anatomic outcomes with these surgeries are not always perfect, our reoperation rate for prolapse is only about 5%, with a high level of satisfaction in 88% to 92% of patients.
DR. RAZ: Unaugmented reconstruction fails in more than 30% of cases. Some patients who have significant prolapse and attenuated tissue think that this tissue will become healthier or stronger after reconstructive surgery, but that isn’t the case. In these situations, excision and plication make no clinical sense.
The problem is that we have yet to identify the ideal surrogate for poor-quality tissue. Most of us use polypropylene mesh in different variations. We need a better material that will be nonimmunogenic, well tolerated, and easily incorporated without erosion. Xenograft-like derivatives of dermis, or allografts such as cadaveric fascia, have failed over the long term because the body reabsorbs the graft without forming any new connective tissue.

FIGURE 3 Mesh can be cut in the OR to custom-fit a patient
Hand-cut mesh and points of placement.
PHOTO: SHLOMO RAZ, MD
Is a kit a valuable aid?
DR. KARRAM: If a surgeon wants to augment a repair, what are the advantages of a packaged mesh kit, compared with simply cutting the mesh and performing surgery without a kit?
DR. WALTERS: The advantages of a packaged mesh kit are the convenience involved and the ability to consistently perform the same operation with the same product. That facilitates learning, teaching, and research. It also helps us understand the published literature a little better because “custom” prolapse repairs are operator-dependent and difficult to apply generally to a population of surgeons.
These advantages are most clearly apparent with midurethral sling mesh kits, which have almost revolutionized surgery for stress incontinence. I don’t believe mesh kits for prolapse are there yet, but they certainly have potential.
DR. RAZ: I’m opposed to the use of kits. They are industry-driven. One company has made $1 billion selling them. Imagine a patient who undergoes placement of a sling kit ($1,000), cystocele kit ($1,500), and posterior mesh kit ($1,500). How can our healthcare system sustain this burden, especially when there is no real evidence that a kit improves the operation, and given the incredible complication rate that we see?
Moreover, the kits contain a single-use needle and passer and a precut segment of polypropylene mesh. But every patient is different and requires a unique size or shape of mesh. I don’t believe that a surgeon who knows pelvic anatomy needs a kit to perform mesh-augmented reconstruction. We can buy the same segment of mesh for $200 to $400, cut it as needed, and perform the same operation advertised by industry.
For surgeons who prefer a kit, the tools that are included should be made reusable.
DR. LUCENTE: In my opinion, the primary advantage of a commercially available transvaginal mesh delivery system—notice, I avoided the word “kit,” because I think there are plenty of negative connotations associated with it—is the ability to deliver the mesh in a “tension”-free manner.
One alternative that many people pursue is cutting the mesh to size and using sutures to hold it in place while tissue ingrowth occurs. However, the hernia literature suggests that suturing mesh in place increases the risk of postoperative discomfort at the site of implantation. The true cause of the discomfort remains unclear, but it is thought to arise from nerve tethering or traction at the pre-committed points of attachment before the host tissue and mesh interface have adjusted or settled with tissue ingrowth.
All neuropathic complications of mesh implantation have been shown in the current hernia literature to be increased with the use of sutures.7 Also, as previously mentioned, it is extremely difficult to set or adjust the mesh with the vaginal incision remaining “open,” which is a downside to suture techniques.
What training is necessary to use a kit?
DR. KARRAM: Mesh kits are aggressively promoted by industry, with close to half a dozen different kits to be available soon. What is the minimum amount of training one should have before utilizing these kits?
DR. WALTERS: The surgeon should at least know how to perform traditional sutured prolapse repairs and SUI surgery and be able to perform cystoscopy. Ideally, the surgeon should undergo training on a cadaver with a skilled and experienced user of the mesh kit. The surgeon also should carefully review the risks and benefits of mesh kits with the patient and inform the patient that he or she is in the early learning curve of a particular surgery. The informed patient should have a right to refuse mesh-augmented prolapse surgery after the consent process.
DR. LUCENTE: I’m glad you asked this question. I strongly believe that surgical expertise and proficiency within gynecology need to be more effectively addressed by us all. We have a situation in our field in which techniques and technology are widening the gap between what is possible and what the surgeon is comfortable doing safely.
It’s incumbent on all of us, especially those who are in a leadership position as a chairperson or chief of a division, to work with our physician staff and faculty to optimize surgical skill and patient outcomes, including safety, with new technologies.
As for the minimal amount of training needed, that’s extremely variable. It depends on the current skill set of the physician and his or her ability to pick up the mechanics of the surgery as it is taught through a cadaver lab or preceptorship. It’s regrettable that some physicians lack the objectivity and insight to judge their own skill set. This, again, is the time for a chairperson or chief of a division to step up to the plate and ensure proper credentialing and demonstration of proficiency.
It is unrealistic to expect industry to decide who should or should not utilize this truly breakthrough technology. That is our responsibility as physicians.
DR. KARRAM: At a minimum, I think any surgeon utilizing a kit should have a firm understanding of pelvic floor anatomy and experience performing traditional repairs:
- intraperitoneal procedures such as Mc-Call culdoplasty and uterosacral suspension
- sacrospinous suspension
- retropubic procedures and anti-incontinence operations such as pubovaginal slings.
This three-dimensional understanding of the pelvic floor is mandatory if one is to assume that blind passage of trocars through potentially dangerous spaces is the wave of the future.
DR. RAZ: You need to be a pelvic surgeon, know your anatomy, and know how to manage complications if you are going to use one of these kits. You should stick to the surgery that works best in your hands. Industry cannot teach you to be a good pelvic surgeon; it takes lifelong experience.
DR. KARRAM: If you have a patient who is sexually inactive with pelvic organ prolapse, would you prefer a mesh repair or an obliterative procedure? And why?
DR. WALTERS: If the patient is sexually inactive—especially if she is older and definitely will not be in the future—it makes absolutely no sense to perform a mesh-augmented repair. A traditional, somewhat tight, sutured repair works fine in this setting and carries very low risk.
In fact, our group and others have found that, in carefully selected patients, partial colpectomy and colpocleisis procedures (without grafts) have among the highest cure and satisfaction rates of all surgeries we perform for prolapse; they also have relatively low risk.8 Recurrent prolapse after an obliterative procedure is rare; most of the dissatisfaction relates to postoperative voiding difficulties or persistent or de novo urinary incontinence.
DR. KARRAM: I also prefer an obliterative procedure. I see no reason to bring in the cost and potential for complications that mesh repair entails. An obliterative procedure should produce an anatomic success rate close to 100%, with minimal complications. It also can be performed quickly with minimal anesthesia and convalescence.
DR. LUCENTE: My response is based on a clinical study that my associate, Dr. Miles Murphy, has performed, comparing a transvaginal mesh procedure with a LaForte operation for severe pelvic organ prolapse.9 Both patient groups were well satisfied with the result, and success rates were comparable. However, the group that underwent the transvaginal mesh procedure had a shorter operative time.
As a result of these studies, we tend to prefer transvaginal mesh repair. Even though the woman may be sexually inactive, the procedure preserves vaginal function, and we all know that life has a way of being unpredictable. Her situation may change so that she once again desires sexual function.
However, for a very elderly woman—one in her late 80s or 90s—who has severe or extreme prolapse with a very large procidentia and vaginal length measuring, say, 13 cm beyond the introitus, I do prefer an obliterative procedure.
DR. RAZ: I agree. I would not offer a sexually inactive patient an obliterative procedure. You never know what the future will hold.
Mesh repair can be performed safely, provided the surgeon has good knowledge of anatomic landmarks and knows how to manage any potential complications that may arise.
1. Nygaard IE, McCreery R, Brubaker L, et al. Pelvic Floor Disorders Network. Abdominal sacrocolpopexy: a comprehensive review. Obstet Gynecol. 2004;104:805-823.
2. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421;discussion 1421-1422.
3. Maher CF, Qatawneh AM, Dwyer PL, Carey MP, Cornish A, Schluter PJ. Abdominal sacral colpopexy or vaginal sacrospinous colpopexy for vaginal vault prolapse: a prospective randomized study. Am J Obstet Gynecol. 2004;190:20-26.
4. Murphy M. Society of Gynecologic Surgeons Systematic Review Group. Clinical practice guidelines on vaginal graft use from the Society of Gynecologic Surgeons. Obstet Gynecol. 2008;112:1123-1130.
5. Paraiso MF, Barber MD, Muir TW, Walters MD. Rectocele repair: a randomized trial of three surgical procedures including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
6. Silva WA, Pauls RN, Segal JL, Rooney CM, Kleeman SD, Karram MM. Uterosacral ligament vault suspension: five-year outcomes. Obstet Gynecol 2006;108:255-263.
7. EU Hernia Trialists Collaboration. Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg. 2002;235:322-332.
8. Barber MD, Amundsen C, Paraiso MFR, Weidner A, Romero A, Walters MD. Quality of life after surgery for genital prolapse in elderly women: obliterative and reconstructive surgery. Int Urogynecol J. 2007;18:799-806.
9. Murphy M, van Raalte H, Mercurio E, Haff R, Wiseman B, Lucente VR. Incontinence-related quality of life and sexual function following the tension-free vaginal tape vs the “inside-out” tension-free vaginal tape obturator. Int Urogynecol J. 2008;19:481-487.
1. Nygaard IE, McCreery R, Brubaker L, et al. Pelvic Floor Disorders Network. Abdominal sacrocolpopexy: a comprehensive review. Obstet Gynecol. 2004;104:805-823.
2. Benson JT, Lucente V, McClellan E. Vaginal versus abdominal reconstructive surgery for the treatment of pelvic support defects: a prospective randomized study with long-term outcome evaluation. Am J Obstet Gynecol. 1996;175:1418-1421;discussion 1421-1422.
3. Maher CF, Qatawneh AM, Dwyer PL, Carey MP, Cornish A, Schluter PJ. Abdominal sacral colpopexy or vaginal sacrospinous colpopexy for vaginal vault prolapse: a prospective randomized study. Am J Obstet Gynecol. 2004;190:20-26.
4. Murphy M. Society of Gynecologic Surgeons Systematic Review Group. Clinical practice guidelines on vaginal graft use from the Society of Gynecologic Surgeons. Obstet Gynecol. 2008;112:1123-1130.
5. Paraiso MF, Barber MD, Muir TW, Walters MD. Rectocele repair: a randomized trial of three surgical procedures including graft augmentation. Am J Obstet Gynecol. 2006;195:1762-1771.
6. Silva WA, Pauls RN, Segal JL, Rooney CM, Kleeman SD, Karram MM. Uterosacral ligament vault suspension: five-year outcomes. Obstet Gynecol 2006;108:255-263.
7. EU Hernia Trialists Collaboration. Repair of groin hernia with synthetic mesh: meta-analysis of randomized controlled trials. Ann Surg. 2002;235:322-332.
8. Barber MD, Amundsen C, Paraiso MFR, Weidner A, Romero A, Walters MD. Quality of life after surgery for genital prolapse in elderly women: obliterative and reconstructive surgery. Int Urogynecol J. 2007;18:799-806.
9. Murphy M, van Raalte H, Mercurio E, Haff R, Wiseman B, Lucente VR. Incontinence-related quality of life and sexual function following the tension-free vaginal tape vs the “inside-out” tension-free vaginal tape obturator. Int Urogynecol J. 2008;19:481-487.
A new year, a new CPT: Will these changes rattle your practice?
Take note, ObGyns: A number of changes in Current Procedural Terminology (CPT) 2009—those changes took effect January 1—are going to modify the way you bill and will have an impact on your reimbursement. Most of these changes are minor, although renumbering of infusion codes will require changes to the encounter form. And I have good and bad news for urogynecologists who perform vaginal paravaginal repairs and sling procedures for stress urinary incontinence. Read on for details!
Mesh for vaginal paravaginal defect repair—
code error corrected
Code 57267 is an add-on code that describes the insertion of mesh, or other prosthesis, through a vaginal approach when native tissues have been determined to be weak and inadequate for repair—especially in patients who have undergone a previous attempt at repair. As an add-on code, it can be billed only in addition to other, specific procedures.
Before January 1, code 57267 could only be reported with an anterior or posterior colporrhaphy, or both, or with a rectocele repair without colporrhaphy.
When performing a vaginal approach paravaginal defect repair, however, the same weakened tissues also require use of the mesh, yet code 57825 (paravaginal defect repair [including repair of cystocele, if performed]) was not included as one of the allowed codes. This error is rectified in 2009.
You must still be aware that reporting the 57267 add-on code requires that you establish medical necessity for its use. Documentation of weakened, attenuated, or incompetent pubocervical tissue in the case of a paravaginal repair (International Classification of Diseases Clinical Modification [ICD-9-CM] code 618.81) or rectovaginal tissue for rectocele/enterocele repair (618.82) continues to be important when reporting the add-on mesh code.
A reminder about anesthesia
Until January 1, codes 57400 (dilation of vagina), 57410 (pelvic examination), and 57415 (removal of impacted vaginal foreign body) read “under anesthesia.” In a move to standardize terminology, these codes will be revised to add the wording “other than local.” The revision clarifies that 1) all surgical codes include administration of a local anesthetic and 2) codes designated with “under anesthesia” refer to regional blocks and general anesthesia.
of 2 urogynecology procedures for UI!
Although not a CPT change, it’s worth noting that physicians who perform 1) sling operations for correcting stress urinary incontinence or 2) subsequent revisions because of problems with fascia or synthetic mesh need to be aware that the physician work relative value for these procedures has been decreased in 2009 by the Centers for Medicare and Medicaid Services (CMS). Why the drop? According to CMS, results of surveys by the American Urogynecologic Society and the American Urological Association indicate that the procedures are not as difficult to perform as once considered.
The two affected codes are:
| 57288 | Sling operation for stress incontinence (e.g., fascia or synthetic) |
| 57287 | Removal or revision of sling for stress urinary incontinence (e.g., fascia or synthetic) |
The change will result in a decline in payment for these procedures by Medicare and some non-Medicare payers, and will be felt harder with sling procedures than with revisions. Why? The work relative value units (RVUs) decreased for 57287, but that decrease was offset by an increase in practice expense relative value—which resulted in total RVUs increasing for this code in 2009, from 18.31 to 18.53.
Code 57288, on the other hand, has been tagged with a decrease in both the physician work and practice expense RVUs. Total RVUs for this code, therefore, have dropped from 21.59 to 19.62. In Medicare dollars, that equates to about $118 less for the same procedure when one applies the 2009 Medicare conversion factor of $36.07.
New human papillomavirus vaccine, new code
A new code, 90650, has been added to report the newer bivalent human papillomavirus (HPV) vaccine, which contains an adjuvant formulation and is intended to protect against infection by high-risk HPV types 16 and 18. The existing HPV vaccine code, 90649, targets those high-risk types of HPV and two low-risk types (6 and 11).
Coverage recommendations for the new vaccine match those of the existing, quadrivalent vaccine, but not all payers are covering the HPV vaccine based on those recommendations. The new vaccine offers a less costly alternative for patients whose health-care insurance does not cover the vaccine or who are uninsured.
Wholesale reorganization of injection and infusion codes
Codes 90760–90779 (covering therapeutic, prophylactic, and diagnostic injections and infusions) are deleted in 2009 and renumbered, with the same descriptors, to 96360–96379. This was done to organize all infusions and injections together. The biggest change for you and every other ObGyn? You must revise the practice’s encounter form to reflect the requirement that intramuscular and subcutaneous injections are now coded 96372 instead of 90772.
Modifier -21 and prolonged E/M services
Now deleted is Modifier -21 (prolonged evaluation and management [E/M] service). This modifier represented acknowledgment that a continuous face-to-face E/M service could exceed the maximum time allowed by the highest level of E/M service for the type being billed.
In other words, before January 1, 2009, if a patient’s condition was such that you documented an established or new patient visit (99215 or 99205) but in fact spent more time with her than the 45 or 60 minutes that typically accompanies these codes, you added modifier -21 in the hope of receiving higher reimbursement. Now the modifier is deleted because there is already a mechanism in place to report such prolonged service.
Add-on codes 99354–99357 are used to report face-to-face outpatient and inpatient prolonged E/M services. Guidelines for these codes mandate cumulative time rather than continuous time, and using the add-on codes is contingent on the additional time spent being 30 or more minutes above the typical time allotted for the basic E/M service that you are billing.
Here’s a case that exemplifies how coding works in these circumstances:
CASE
You evaluate a patient for severe uterine bleeding, and report a level-4 visit (99214), which has a typical time of 25 minutes. At the same visit, you determine that endometrial biopsy is required, and you perform it during the visit. But the patient faints during the procedure—and you spend an additional 35 minutes (cumulative time) with her before you send her home.
Because the typical time of 25 minutes was exceeded by at least 30 minutes, you should report 99354 (prolonged physician service in the office or other outpatient setting requiring direct [face-to-face] patient contact beyond the usual service; first hour [list separately in addition to code for office or other outpatient Evaluation and Management service]) in addition to 99214.
Guidelines for correct use of these codes are also being revised to emphasize that only outpatient prolonged services codes are intended to be used to report total duration of face-to-face time; on the other hand, inpatient codes are intended to report the total duration of the time spent (whether continuous or noncontinuous) by the physician on the unit actively involved in caring for the patient.
Take note, ObGyns: A number of changes in Current Procedural Terminology (CPT) 2009—those changes took effect January 1—are going to modify the way you bill and will have an impact on your reimbursement. Most of these changes are minor, although renumbering of infusion codes will require changes to the encounter form. And I have good and bad news for urogynecologists who perform vaginal paravaginal repairs and sling procedures for stress urinary incontinence. Read on for details!
Mesh for vaginal paravaginal defect repair—
code error corrected
Code 57267 is an add-on code that describes the insertion of mesh, or other prosthesis, through a vaginal approach when native tissues have been determined to be weak and inadequate for repair—especially in patients who have undergone a previous attempt at repair. As an add-on code, it can be billed only in addition to other, specific procedures.
Before January 1, code 57267 could only be reported with an anterior or posterior colporrhaphy, or both, or with a rectocele repair without colporrhaphy.
When performing a vaginal approach paravaginal defect repair, however, the same weakened tissues also require use of the mesh, yet code 57825 (paravaginal defect repair [including repair of cystocele, if performed]) was not included as one of the allowed codes. This error is rectified in 2009.
You must still be aware that reporting the 57267 add-on code requires that you establish medical necessity for its use. Documentation of weakened, attenuated, or incompetent pubocervical tissue in the case of a paravaginal repair (International Classification of Diseases Clinical Modification [ICD-9-CM] code 618.81) or rectovaginal tissue for rectocele/enterocele repair (618.82) continues to be important when reporting the add-on mesh code.
A reminder about anesthesia
Until January 1, codes 57400 (dilation of vagina), 57410 (pelvic examination), and 57415 (removal of impacted vaginal foreign body) read “under anesthesia.” In a move to standardize terminology, these codes will be revised to add the wording “other than local.” The revision clarifies that 1) all surgical codes include administration of a local anesthetic and 2) codes designated with “under anesthesia” refer to regional blocks and general anesthesia.
of 2 urogynecology procedures for UI!
Although not a CPT change, it’s worth noting that physicians who perform 1) sling operations for correcting stress urinary incontinence or 2) subsequent revisions because of problems with fascia or synthetic mesh need to be aware that the physician work relative value for these procedures has been decreased in 2009 by the Centers for Medicare and Medicaid Services (CMS). Why the drop? According to CMS, results of surveys by the American Urogynecologic Society and the American Urological Association indicate that the procedures are not as difficult to perform as once considered.
The two affected codes are:
| 57288 | Sling operation for stress incontinence (e.g., fascia or synthetic) |
| 57287 | Removal or revision of sling for stress urinary incontinence (e.g., fascia or synthetic) |
The change will result in a decline in payment for these procedures by Medicare and some non-Medicare payers, and will be felt harder with sling procedures than with revisions. Why? The work relative value units (RVUs) decreased for 57287, but that decrease was offset by an increase in practice expense relative value—which resulted in total RVUs increasing for this code in 2009, from 18.31 to 18.53.
Code 57288, on the other hand, has been tagged with a decrease in both the physician work and practice expense RVUs. Total RVUs for this code, therefore, have dropped from 21.59 to 19.62. In Medicare dollars, that equates to about $118 less for the same procedure when one applies the 2009 Medicare conversion factor of $36.07.
New human papillomavirus vaccine, new code
A new code, 90650, has been added to report the newer bivalent human papillomavirus (HPV) vaccine, which contains an adjuvant formulation and is intended to protect against infection by high-risk HPV types 16 and 18. The existing HPV vaccine code, 90649, targets those high-risk types of HPV and two low-risk types (6 and 11).
Coverage recommendations for the new vaccine match those of the existing, quadrivalent vaccine, but not all payers are covering the HPV vaccine based on those recommendations. The new vaccine offers a less costly alternative for patients whose health-care insurance does not cover the vaccine or who are uninsured.
Wholesale reorganization of injection and infusion codes
Codes 90760–90779 (covering therapeutic, prophylactic, and diagnostic injections and infusions) are deleted in 2009 and renumbered, with the same descriptors, to 96360–96379. This was done to organize all infusions and injections together. The biggest change for you and every other ObGyn? You must revise the practice’s encounter form to reflect the requirement that intramuscular and subcutaneous injections are now coded 96372 instead of 90772.
Modifier -21 and prolonged E/M services
Now deleted is Modifier -21 (prolonged evaluation and management [E/M] service). This modifier represented acknowledgment that a continuous face-to-face E/M service could exceed the maximum time allowed by the highest level of E/M service for the type being billed.
In other words, before January 1, 2009, if a patient’s condition was such that you documented an established or new patient visit (99215 or 99205) but in fact spent more time with her than the 45 or 60 minutes that typically accompanies these codes, you added modifier -21 in the hope of receiving higher reimbursement. Now the modifier is deleted because there is already a mechanism in place to report such prolonged service.
Add-on codes 99354–99357 are used to report face-to-face outpatient and inpatient prolonged E/M services. Guidelines for these codes mandate cumulative time rather than continuous time, and using the add-on codes is contingent on the additional time spent being 30 or more minutes above the typical time allotted for the basic E/M service that you are billing.
Here’s a case that exemplifies how coding works in these circumstances:
CASE
You evaluate a patient for severe uterine bleeding, and report a level-4 visit (99214), which has a typical time of 25 minutes. At the same visit, you determine that endometrial biopsy is required, and you perform it during the visit. But the patient faints during the procedure—and you spend an additional 35 minutes (cumulative time) with her before you send her home.
Because the typical time of 25 minutes was exceeded by at least 30 minutes, you should report 99354 (prolonged physician service in the office or other outpatient setting requiring direct [face-to-face] patient contact beyond the usual service; first hour [list separately in addition to code for office or other outpatient Evaluation and Management service]) in addition to 99214.
Guidelines for correct use of these codes are also being revised to emphasize that only outpatient prolonged services codes are intended to be used to report total duration of face-to-face time; on the other hand, inpatient codes are intended to report the total duration of the time spent (whether continuous or noncontinuous) by the physician on the unit actively involved in caring for the patient.
Take note, ObGyns: A number of changes in Current Procedural Terminology (CPT) 2009—those changes took effect January 1—are going to modify the way you bill and will have an impact on your reimbursement. Most of these changes are minor, although renumbering of infusion codes will require changes to the encounter form. And I have good and bad news for urogynecologists who perform vaginal paravaginal repairs and sling procedures for stress urinary incontinence. Read on for details!
Mesh for vaginal paravaginal defect repair—
code error corrected
Code 57267 is an add-on code that describes the insertion of mesh, or other prosthesis, through a vaginal approach when native tissues have been determined to be weak and inadequate for repair—especially in patients who have undergone a previous attempt at repair. As an add-on code, it can be billed only in addition to other, specific procedures.
Before January 1, code 57267 could only be reported with an anterior or posterior colporrhaphy, or both, or with a rectocele repair without colporrhaphy.
When performing a vaginal approach paravaginal defect repair, however, the same weakened tissues also require use of the mesh, yet code 57825 (paravaginal defect repair [including repair of cystocele, if performed]) was not included as one of the allowed codes. This error is rectified in 2009.
You must still be aware that reporting the 57267 add-on code requires that you establish medical necessity for its use. Documentation of weakened, attenuated, or incompetent pubocervical tissue in the case of a paravaginal repair (International Classification of Diseases Clinical Modification [ICD-9-CM] code 618.81) or rectovaginal tissue for rectocele/enterocele repair (618.82) continues to be important when reporting the add-on mesh code.
A reminder about anesthesia
Until January 1, codes 57400 (dilation of vagina), 57410 (pelvic examination), and 57415 (removal of impacted vaginal foreign body) read “under anesthesia.” In a move to standardize terminology, these codes will be revised to add the wording “other than local.” The revision clarifies that 1) all surgical codes include administration of a local anesthetic and 2) codes designated with “under anesthesia” refer to regional blocks and general anesthesia.
of 2 urogynecology procedures for UI!
Although not a CPT change, it’s worth noting that physicians who perform 1) sling operations for correcting stress urinary incontinence or 2) subsequent revisions because of problems with fascia or synthetic mesh need to be aware that the physician work relative value for these procedures has been decreased in 2009 by the Centers for Medicare and Medicaid Services (CMS). Why the drop? According to CMS, results of surveys by the American Urogynecologic Society and the American Urological Association indicate that the procedures are not as difficult to perform as once considered.
The two affected codes are:
| 57288 | Sling operation for stress incontinence (e.g., fascia or synthetic) |
| 57287 | Removal or revision of sling for stress urinary incontinence (e.g., fascia or synthetic) |
The change will result in a decline in payment for these procedures by Medicare and some non-Medicare payers, and will be felt harder with sling procedures than with revisions. Why? The work relative value units (RVUs) decreased for 57287, but that decrease was offset by an increase in practice expense relative value—which resulted in total RVUs increasing for this code in 2009, from 18.31 to 18.53.
Code 57288, on the other hand, has been tagged with a decrease in both the physician work and practice expense RVUs. Total RVUs for this code, therefore, have dropped from 21.59 to 19.62. In Medicare dollars, that equates to about $118 less for the same procedure when one applies the 2009 Medicare conversion factor of $36.07.
New human papillomavirus vaccine, new code
A new code, 90650, has been added to report the newer bivalent human papillomavirus (HPV) vaccine, which contains an adjuvant formulation and is intended to protect against infection by high-risk HPV types 16 and 18. The existing HPV vaccine code, 90649, targets those high-risk types of HPV and two low-risk types (6 and 11).
Coverage recommendations for the new vaccine match those of the existing, quadrivalent vaccine, but not all payers are covering the HPV vaccine based on those recommendations. The new vaccine offers a less costly alternative for patients whose health-care insurance does not cover the vaccine or who are uninsured.
Wholesale reorganization of injection and infusion codes
Codes 90760–90779 (covering therapeutic, prophylactic, and diagnostic injections and infusions) are deleted in 2009 and renumbered, with the same descriptors, to 96360–96379. This was done to organize all infusions and injections together. The biggest change for you and every other ObGyn? You must revise the practice’s encounter form to reflect the requirement that intramuscular and subcutaneous injections are now coded 96372 instead of 90772.
Modifier -21 and prolonged E/M services
Now deleted is Modifier -21 (prolonged evaluation and management [E/M] service). This modifier represented acknowledgment that a continuous face-to-face E/M service could exceed the maximum time allowed by the highest level of E/M service for the type being billed.
In other words, before January 1, 2009, if a patient’s condition was such that you documented an established or new patient visit (99215 or 99205) but in fact spent more time with her than the 45 or 60 minutes that typically accompanies these codes, you added modifier -21 in the hope of receiving higher reimbursement. Now the modifier is deleted because there is already a mechanism in place to report such prolonged service.
Add-on codes 99354–99357 are used to report face-to-face outpatient and inpatient prolonged E/M services. Guidelines for these codes mandate cumulative time rather than continuous time, and using the add-on codes is contingent on the additional time spent being 30 or more minutes above the typical time allotted for the basic E/M service that you are billing.
Here’s a case that exemplifies how coding works in these circumstances:
CASE
You evaluate a patient for severe uterine bleeding, and report a level-4 visit (99214), which has a typical time of 25 minutes. At the same visit, you determine that endometrial biopsy is required, and you perform it during the visit. But the patient faints during the procedure—and you spend an additional 35 minutes (cumulative time) with her before you send her home.
Because the typical time of 25 minutes was exceeded by at least 30 minutes, you should report 99354 (prolonged physician service in the office or other outpatient setting requiring direct [face-to-face] patient contact beyond the usual service; first hour [list separately in addition to code for office or other outpatient Evaluation and Management service]) in addition to 99214.
Guidelines for correct use of these codes are also being revised to emphasize that only outpatient prolonged services codes are intended to be used to report total duration of face-to-face time; on the other hand, inpatient codes are intended to report the total duration of the time spent (whether continuous or noncontinuous) by the physician on the unit actively involved in caring for the patient.
Man treated for asthma dies of undiagnosed heart disease...Failure to confirm Echo result leads to cardiac arrest...more...
Man treated for asthma dies of undiagnosed heart disease
A MONTH AFTER HE BEGAN RECEIVING ASTHMA TREATMENT from his physician, a 50-year-old man suffered a heart attack and died. An autopsy revealed idiopathic dilated cardiomyopathy.
PLAINTIFF’S CLAIM: The doctor negligently failed to examine the patient for heart disease; the patient was in congestive heart failure during treatment.
DOCTOR’S DEFENSE: The physician claimed that he twice recommended that the patient see a cardiologist. The plaintiff countered that the doctor didn’t make a referral, despite chart notes to that effect.
VERDICT: California defense verdict.
COMMENT: Clear documentation of the history, physical, and differential diagnostic thinking helps fend off unwarranted lawsuits.
Failure to confirm Echo result leads to cardiac arrest
SUDDEN ONSET OF CHEST PAIN radiating to the back, which had started during rest, brought a 49-year-old woman to the hospital. The patient also complained of pain radiating to her left jaw and ear, which became worse when she inhaled or moved. She had no shortness of breath, palpitations, diaphoresis, or history of trauma. She did have a history of gastroesophageal reflux disease (GERD), but said that the pain didn’t resemble the pain of GERD. While in the triage area, she vomited.
Two electrocardiograms (EKGs) done in the emergency room showed sinus bradycardia and nonspecific T-wave abnormalities. A chest radiograph was reported as normal, but with a note of borderline heart enlargement and a tortuous aorta. A gastrointestinal (GI) cocktail of Nitropaste and Toradol didn’t relieve the pain, nor did Ativan. No workup for aortic dissection was done.
After consultation with a doctor covering for the patient’s primary care physician, the patient was hospitalized with orders for laboratory studies, a chest radiograph, and an EKG the next morning. The EKG again showed abnormalities, including a nonspecific T-wave abnormality, as did the chest radiograph (moderate cardiomegaly, tortuous aorta, mild prominence of the pulmonary vasculature without evidence of congestive failure, and small left pleural effusion or slight blunting of the left lateral costophrenic angle). But the radiograph wasn’t compared to the one taken the night before. A GI consult—by which time the patient’s hematocrit had dropped from 32 to 26—attributed the pain to GERD and recommended outpatient esophagogastroduodenoscopy.
The results of a routine echocardiogram—faxed to the patient’s floor the same day—were worrisome: a dilated aortic root and ascending aorta accompanied by at least moderately severe aortic insufficiency and normal ventricular function.
The patient’s primary care physician saw the patient and discharged her that evening. Fewer than 2 hours later, the patient suffered a cardiac arrest at home and couldn’t be resuscitated after transport to the hospital. An autopsy found the cause of death to be cardiac tamponade resulting from dissection of an aortic aneurysm.
PLAINTIFF’S CLAIM: The patient shouldn’t have been discharged without clarification of the echocardiogram results.
DOCTOR’S DEFENSE: The primary care physician’s understanding was that the cardiologist had ruled out heart-related problems, including aortic dissection, and that the patient had been diagnosed with a stomach illness, which would be followed on an outpatient basis. Even if a diagnosis of aortic dissection had been made, the outcome would have been the same.
VERDICT: $560,000 Massachusetts settlement.
COMMENT: Inadequate follow-up of testing—in this case, an inpatient echocardiogram—can have catastrophic results. Before discharge, each inpatient test should be reviewed and adjudicated, and a clear plan for follow-up delineated.
Cancer missed in patient with rectal bleeding
A 44-YEAR-OLD MAN went to his family physician, an internist, with complaints that included rectal bleeding. The physician performed a flexible sigmoidoscopy, which found hemorrhoids that weren’t inflamed or bleeding. A hemoccult test at a physical exam before the sigmoidoscopy was positive for bleeding.
A year later, the patient returned to the doctor complaining of blood in his underwear almost every other day. The doctor noted a “slightly inflamed hemorrhoid” on anoscopy, but no bleeding from the hemorrhoid; he didn’t test for occult bleeding.
Early the next year, the patient saw the physician for a complaint of blood in the stool and changes in bowel habits. A hemoccult test was positive, and the doctor diagnosed irritable bowel syndrome. The patient returned 6 months later with the same complaints and, he said, requested referral to a gastroenterologist. The doctor again attributed the complaints to irritable bowel syndrome.
Early the following year, the patient went to another internist because his insurance changed. This internist immediately diagnosed stage-3 rectal cancer. The patient underwent radiation, chemotherapy, and 2 surgeries, one to remove part of his rectum and a second to reverse an ileostomy done during the first operation. The patient was left impotent, with permanent, variable bowel dysfunction.
PLAINTIFF’S CLAIM: The diagnosis of hemorrhoids wasn’t reasonable; the patient should have been referred to a gastroenterologist or for colorectal cancer surgery. Early detection and diagnosis would have resulted in removal of a polyp or early cancer, which could have been done during a colonoscopy or by transanal excision.
DOCTOR’S DEFENSE: The patient’s doctor denied that the patient had requested a referral to a gastroenterologist and maintained that he believed the flexible sigmoidoscopy had ruled out a serious cause of bleeding.
VERDICT: $1 million Virginia verdict.
COMMENT: When a patient has persistent rectal bleeding without a clear cause, no matter what the patient’s age, further evaluation or referral is prudent.
Man treated for asthma dies of undiagnosed heart disease
A MONTH AFTER HE BEGAN RECEIVING ASTHMA TREATMENT from his physician, a 50-year-old man suffered a heart attack and died. An autopsy revealed idiopathic dilated cardiomyopathy.
PLAINTIFF’S CLAIM: The doctor negligently failed to examine the patient for heart disease; the patient was in congestive heart failure during treatment.
DOCTOR’S DEFENSE: The physician claimed that he twice recommended that the patient see a cardiologist. The plaintiff countered that the doctor didn’t make a referral, despite chart notes to that effect.
VERDICT: California defense verdict.
COMMENT: Clear documentation of the history, physical, and differential diagnostic thinking helps fend off unwarranted lawsuits.
Failure to confirm Echo result leads to cardiac arrest
SUDDEN ONSET OF CHEST PAIN radiating to the back, which had started during rest, brought a 49-year-old woman to the hospital. The patient also complained of pain radiating to her left jaw and ear, which became worse when she inhaled or moved. She had no shortness of breath, palpitations, diaphoresis, or history of trauma. She did have a history of gastroesophageal reflux disease (GERD), but said that the pain didn’t resemble the pain of GERD. While in the triage area, she vomited.
Two electrocardiograms (EKGs) done in the emergency room showed sinus bradycardia and nonspecific T-wave abnormalities. A chest radiograph was reported as normal, but with a note of borderline heart enlargement and a tortuous aorta. A gastrointestinal (GI) cocktail of Nitropaste and Toradol didn’t relieve the pain, nor did Ativan. No workup for aortic dissection was done.
After consultation with a doctor covering for the patient’s primary care physician, the patient was hospitalized with orders for laboratory studies, a chest radiograph, and an EKG the next morning. The EKG again showed abnormalities, including a nonspecific T-wave abnormality, as did the chest radiograph (moderate cardiomegaly, tortuous aorta, mild prominence of the pulmonary vasculature without evidence of congestive failure, and small left pleural effusion or slight blunting of the left lateral costophrenic angle). But the radiograph wasn’t compared to the one taken the night before. A GI consult—by which time the patient’s hematocrit had dropped from 32 to 26—attributed the pain to GERD and recommended outpatient esophagogastroduodenoscopy.
The results of a routine echocardiogram—faxed to the patient’s floor the same day—were worrisome: a dilated aortic root and ascending aorta accompanied by at least moderately severe aortic insufficiency and normal ventricular function.
The patient’s primary care physician saw the patient and discharged her that evening. Fewer than 2 hours later, the patient suffered a cardiac arrest at home and couldn’t be resuscitated after transport to the hospital. An autopsy found the cause of death to be cardiac tamponade resulting from dissection of an aortic aneurysm.
PLAINTIFF’S CLAIM: The patient shouldn’t have been discharged without clarification of the echocardiogram results.
DOCTOR’S DEFENSE: The primary care physician’s understanding was that the cardiologist had ruled out heart-related problems, including aortic dissection, and that the patient had been diagnosed with a stomach illness, which would be followed on an outpatient basis. Even if a diagnosis of aortic dissection had been made, the outcome would have been the same.
VERDICT: $560,000 Massachusetts settlement.
COMMENT: Inadequate follow-up of testing—in this case, an inpatient echocardiogram—can have catastrophic results. Before discharge, each inpatient test should be reviewed and adjudicated, and a clear plan for follow-up delineated.
Cancer missed in patient with rectal bleeding
A 44-YEAR-OLD MAN went to his family physician, an internist, with complaints that included rectal bleeding. The physician performed a flexible sigmoidoscopy, which found hemorrhoids that weren’t inflamed or bleeding. A hemoccult test at a physical exam before the sigmoidoscopy was positive for bleeding.
A year later, the patient returned to the doctor complaining of blood in his underwear almost every other day. The doctor noted a “slightly inflamed hemorrhoid” on anoscopy, but no bleeding from the hemorrhoid; he didn’t test for occult bleeding.
Early the next year, the patient saw the physician for a complaint of blood in the stool and changes in bowel habits. A hemoccult test was positive, and the doctor diagnosed irritable bowel syndrome. The patient returned 6 months later with the same complaints and, he said, requested referral to a gastroenterologist. The doctor again attributed the complaints to irritable bowel syndrome.
Early the following year, the patient went to another internist because his insurance changed. This internist immediately diagnosed stage-3 rectal cancer. The patient underwent radiation, chemotherapy, and 2 surgeries, one to remove part of his rectum and a second to reverse an ileostomy done during the first operation. The patient was left impotent, with permanent, variable bowel dysfunction.
PLAINTIFF’S CLAIM: The diagnosis of hemorrhoids wasn’t reasonable; the patient should have been referred to a gastroenterologist or for colorectal cancer surgery. Early detection and diagnosis would have resulted in removal of a polyp or early cancer, which could have been done during a colonoscopy or by transanal excision.
DOCTOR’S DEFENSE: The patient’s doctor denied that the patient had requested a referral to a gastroenterologist and maintained that he believed the flexible sigmoidoscopy had ruled out a serious cause of bleeding.
VERDICT: $1 million Virginia verdict.
COMMENT: When a patient has persistent rectal bleeding without a clear cause, no matter what the patient’s age, further evaluation or referral is prudent.
Man treated for asthma dies of undiagnosed heart disease
A MONTH AFTER HE BEGAN RECEIVING ASTHMA TREATMENT from his physician, a 50-year-old man suffered a heart attack and died. An autopsy revealed idiopathic dilated cardiomyopathy.
PLAINTIFF’S CLAIM: The doctor negligently failed to examine the patient for heart disease; the patient was in congestive heart failure during treatment.
DOCTOR’S DEFENSE: The physician claimed that he twice recommended that the patient see a cardiologist. The plaintiff countered that the doctor didn’t make a referral, despite chart notes to that effect.
VERDICT: California defense verdict.
COMMENT: Clear documentation of the history, physical, and differential diagnostic thinking helps fend off unwarranted lawsuits.
Failure to confirm Echo result leads to cardiac arrest
SUDDEN ONSET OF CHEST PAIN radiating to the back, which had started during rest, brought a 49-year-old woman to the hospital. The patient also complained of pain radiating to her left jaw and ear, which became worse when she inhaled or moved. She had no shortness of breath, palpitations, diaphoresis, or history of trauma. She did have a history of gastroesophageal reflux disease (GERD), but said that the pain didn’t resemble the pain of GERD. While in the triage area, she vomited.
Two electrocardiograms (EKGs) done in the emergency room showed sinus bradycardia and nonspecific T-wave abnormalities. A chest radiograph was reported as normal, but with a note of borderline heart enlargement and a tortuous aorta. A gastrointestinal (GI) cocktail of Nitropaste and Toradol didn’t relieve the pain, nor did Ativan. No workup for aortic dissection was done.
After consultation with a doctor covering for the patient’s primary care physician, the patient was hospitalized with orders for laboratory studies, a chest radiograph, and an EKG the next morning. The EKG again showed abnormalities, including a nonspecific T-wave abnormality, as did the chest radiograph (moderate cardiomegaly, tortuous aorta, mild prominence of the pulmonary vasculature without evidence of congestive failure, and small left pleural effusion or slight blunting of the left lateral costophrenic angle). But the radiograph wasn’t compared to the one taken the night before. A GI consult—by which time the patient’s hematocrit had dropped from 32 to 26—attributed the pain to GERD and recommended outpatient esophagogastroduodenoscopy.
The results of a routine echocardiogram—faxed to the patient’s floor the same day—were worrisome: a dilated aortic root and ascending aorta accompanied by at least moderately severe aortic insufficiency and normal ventricular function.
The patient’s primary care physician saw the patient and discharged her that evening. Fewer than 2 hours later, the patient suffered a cardiac arrest at home and couldn’t be resuscitated after transport to the hospital. An autopsy found the cause of death to be cardiac tamponade resulting from dissection of an aortic aneurysm.
PLAINTIFF’S CLAIM: The patient shouldn’t have been discharged without clarification of the echocardiogram results.
DOCTOR’S DEFENSE: The primary care physician’s understanding was that the cardiologist had ruled out heart-related problems, including aortic dissection, and that the patient had been diagnosed with a stomach illness, which would be followed on an outpatient basis. Even if a diagnosis of aortic dissection had been made, the outcome would have been the same.
VERDICT: $560,000 Massachusetts settlement.
COMMENT: Inadequate follow-up of testing—in this case, an inpatient echocardiogram—can have catastrophic results. Before discharge, each inpatient test should be reviewed and adjudicated, and a clear plan for follow-up delineated.
Cancer missed in patient with rectal bleeding
A 44-YEAR-OLD MAN went to his family physician, an internist, with complaints that included rectal bleeding. The physician performed a flexible sigmoidoscopy, which found hemorrhoids that weren’t inflamed or bleeding. A hemoccult test at a physical exam before the sigmoidoscopy was positive for bleeding.
A year later, the patient returned to the doctor complaining of blood in his underwear almost every other day. The doctor noted a “slightly inflamed hemorrhoid” on anoscopy, but no bleeding from the hemorrhoid; he didn’t test for occult bleeding.
Early the next year, the patient saw the physician for a complaint of blood in the stool and changes in bowel habits. A hemoccult test was positive, and the doctor diagnosed irritable bowel syndrome. The patient returned 6 months later with the same complaints and, he said, requested referral to a gastroenterologist. The doctor again attributed the complaints to irritable bowel syndrome.
Early the following year, the patient went to another internist because his insurance changed. This internist immediately diagnosed stage-3 rectal cancer. The patient underwent radiation, chemotherapy, and 2 surgeries, one to remove part of his rectum and a second to reverse an ileostomy done during the first operation. The patient was left impotent, with permanent, variable bowel dysfunction.
PLAINTIFF’S CLAIM: The diagnosis of hemorrhoids wasn’t reasonable; the patient should have been referred to a gastroenterologist or for colorectal cancer surgery. Early detection and diagnosis would have resulted in removal of a polyp or early cancer, which could have been done during a colonoscopy or by transanal excision.
DOCTOR’S DEFENSE: The patient’s doctor denied that the patient had requested a referral to a gastroenterologist and maintained that he believed the flexible sigmoidoscopy had ruled out a serious cause of bleeding.
VERDICT: $1 million Virginia verdict.
COMMENT: When a patient has persistent rectal bleeding without a clear cause, no matter what the patient’s age, further evaluation or referral is prudent.
ACE inhibitors and ARBs: One or the other—not both—for high-risk patients
Avoid prescribing an angiotensin-converting enzyme (ACE) inhibitor and an angiotensin receptor blocker (ARB) for patients at high risk of vascular events or renal dysfunction. The combination does not reduce poor outcomes, and leads to more adverse drug-related events than an ACE inhibitor or ARB alone.1
Strength of recommendation
B: 1 large, high-quality randomized controlled trial (RCT).
The ONTARGET investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547-1559.
ILLUSTRATIVE CASE
A 56-year-old patient with well-controlled type 2 diabetes and hypertension comes to see you for routine follow up. His blood pressure is controlled with lisinopril 40 mg/d. But his albumin-to-creatinine ratio is 75 mg/g, and your records reveal that his albuminuria is getting progressively worse.
You’re aware of the potential benefits of a dual angiotensin blockade, and are considering adding an angiotensin receptor blocker (ARB) to your patient’s medication regimen. You wonder whether the combination of an angiotensin-converting enzyme (ACE) inhibitor and an ARB will slow the decline of renal function. You also wonder whether the combination will reduce your patient’s cardiovascular risk.
ACE inhibitors are known to reduce cardiovascular morbidity and mortality, as well as proteinuria in patients with vascular disease or diabetes, whether or not they have heart failure.2 But few studies have compared the effects of ACE inhibitors and ARBs in high-risk patients without heart failure. Nor has there been a definitive study of the effects of an ACE inhibitor–ARB combination on proteinuria and cardiovascular risk.
Are 2 drugs better than 1?
In a recent meta-analysis, researchers reported that combination therapy had a beneficial effect on proteinuria.3 But that observation was based on a small number of patients (N=309 from 10 studies), short follow up, and a lack of data on key clinical end points such as decline of the glomerular filtration rate (GFR) and the onset of dialysis.
Other evidence comes from a study of 199 patients with diabetes and microalbuminuria, in which the ACE inhibitor-ARB combination reduced proteinuria more than either agent alone.4 And in a study of 336 patients with nondiabetic nephropathy, the 2-drug combination slowed the decline in renal function more than monotherapy.5
Small studies raise hopes. These preliminary findings, along with the theoretical benefits of dual angiotensin blockade, suggested that the benefits of taking both agents together could be significant. A large, well-done randomized controlled trial (RCT) was needed to determine the following: (1) whether an ARB is as effective as an ACE inhibitor in reducing morbidity and mortality in high-risk patients who don’t have heart failure, and (2) whether the ACE inhibitor–ARB combination is better than monotherapy for patients at high risk.
The ONTARGET study:
- established that telmisartan, an ARB, is not inferior to ramipril, an ACE inhibitor, in reducing cardiovascular and renal events in high-risk patients without heart failure.
- found that either drug alone is more effective than combination therapy for this patient population.
- cast fresh doubt on the assumption that proteinuria is an accurate surrogate marker for progressive renal dysfunction.
STUDY SUMMARY: Vascular outcomes same for ACE inhibitors, ARBs
The ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET), a multi-year study of thousands of patients, addressed both of those questions. The researchers compared the effects of both telmisartan (Micardis, an ARB) alone and a telmisartan + ramipril (Altace, an ACE inhibitor) combination with the effects of the ACE inhibitor alone in patients ≥55 years of age with established atherosclerotic vascular disease or diabetes with end-organ damage.1 Exclusion criteria included major renal artery stenosis, uncorrected volume or sodium depletion, a serum creatinine concentration of ≥3 mg/dL, and uncontrolled hypertension (>160 mm Hg systolic or >100 mm Hg diastolic).
After a 3-week run-in period to eliminate those who were unable to tolerate either medication or were nonadherent, a total of 25,620 patients remained. They were randomly assigned to take ramipril 10 mg/d, telmisartan 80 mg/d, or both the ACE inhibitor and the ARB. The researchers followed the patients for a median of 56 months.
The primary composite outcome was death from cardiovascular causes, myocardial infarction, stroke, or hospitalization for heart failure;1 the main renal outcome was a composite of first dialysis, doubling of serum creatinine, or death.6
The percentage of patients with the primary outcome was the same in all 3 groups (~16.5%). This finding was somewhat surprising because the blood pressure of patients in the combination therapy group was 2 to 3 mm Hg lower overall (both systolic and diastolic) than the blood pressure of patients on monotherapy—a difference that in other studies has been associated with an estimated 4% to 5% reduction in risk.1,2 Patients in the combination group had more hypotensive symptoms compared with those in the ramipril group (4.8% vs 1.7%, number needed to harm [NNH]=32, P<.001).
Renal dysfunction was highest in dual therapy group
Patients in the combination therapy group had higher rates of renal dysfunction than either the ramipril group (13.5% vs 10.2%, NNH=30, P<.001) or the telmisartan group (10.6%), despite a decrease in proteinuria among those on dual therapy. Patients taking the 2-drug combination also had higher rates of hyperkalemia.
While telmisartan proved to be equal to ramipril in reducing vascular events in high-risk patients, patients taking the ACE inhibitor experienced more cough (NNH=32, P<.001) and angioedema (NNH=500, P=.01). In both monotherapy groups, the rates of adverse drug reactions were probably lower than what we typically see in clinical practice because after the run-in period, only patients who were better able to tolerate both medications remained.
WHAT’S NEW: Combination causes renal impairment
This study established that telmisartan, an ARB, is not inferior to ramipril, an ACE inhibitor, in reducing cardiovascular and renal events in patients without heart failure. In addition, as the largest RCT to explore the effects of a dual blockade of the renin-angiotensin system with an ACE inhibitor and an ARB, it casts fresh doubt on the assumption that proteinuria is an accurate surrogate marker for progressive renal dysfunction. The reduction in proteinuria seen in patients in the combination therapy group came at a cost of increased renal impairment.
CAVEATS: Findings do not apply to heart failure patients
More than 11% of potential subjects were excluded from this study during the run-in period. This suggests that physicians in practice are likely to find a significant number of patients who are unable to tolerate (or fail to adhere to) monotherapy with ACE inhibitors or ARBs.
At baseline, only a small subgroup—13%—had overt diabetic nephropathy, the hallmark for a substantial continuous decline of GFR. However, 38% of the study group had diabetes, and almost 30% of these diabetes patients had microalbuminuria. Subgroup analysis found results consistent with the overall group, and the large sample size reduces the likelihood that these findings were due to low power. The overall rate of dialysis and doubling of serum creatinine was low, but still statistically significant, due to the large size of this study.
In determining treatment for high-risk patients with vascular disease or diabetes, it is important to keep the study population in mind. Studies of patients with poorly controlled congestive heart failure (CHF) have shown potential benefits from an ACE inhibitor–ARB combination.7 The ONTARGET trial specifically excluded individuals with CHF, and its findings—and recommendations to avoid combination therapy—should not be applied to heart failure patients.
CHALLENGES TO IMPLEMENTATION: Best microalbuminuria Tx remains elusive
Although albuminuria has been considered an early sign of the onset of diabetic nephropathy, the ONTARGET study demonstrated that combination therapy may cause further reduction in albuminuria but still adversely affect renal function. Thus, this study raises important questions about the best treatment for patients with diabetes who have microalbuminuria and are already on either an ACE inhibitor or an ARB. We wonder, too, whether we should continue to test for microalbuminuria in patients who are taking one of these agents, given the lack of guidance regarding further treatment.
Acknowledgements
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
PURLs methodology
This study was selected and evaluated using FPIN’s Priority Updates from the Research Literature (PURL) Surveillance System methodology. The criteria and findings leading to the selection of this study as a PURL can be accessed at www.jfponline.com/purls.
Click here to view PURL METHODOLOGY
1. The ONTARGET Investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547-1559.
2. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145-153.
3. Jennings DL, Kalus JS, Coleman CI, et al. Combination therapy with an ACE inhibitor and an angiotensin receptor blocker for diabetic nephropathy: a meta-analysis. Diabet Med. 2007;24:486-493.
4. Mogensen CE, Neldam S, Tikkanen I, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ. 2000;321:1440-1444.
5. Nakao N, Yoshimura A, Morita H, et al. Combination treatment of angiotensin-II receptor blocker and angiotensin-converting-enzyme inhibitor in non-diabetic renal disease (COOPERATE): a randomised controlled trial. Lancet. 2003;361:117-124.
6. Mann JF, Schmieder RE, McQueen M, et al. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet. 2008;372:547-553.
7. Cohn JN, Tognoni G. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345:1667-1675.
Avoid prescribing an angiotensin-converting enzyme (ACE) inhibitor and an angiotensin receptor blocker (ARB) for patients at high risk of vascular events or renal dysfunction. The combination does not reduce poor outcomes, and leads to more adverse drug-related events than an ACE inhibitor or ARB alone.1
Strength of recommendation
B: 1 large, high-quality randomized controlled trial (RCT).
The ONTARGET investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547-1559.
ILLUSTRATIVE CASE
A 56-year-old patient with well-controlled type 2 diabetes and hypertension comes to see you for routine follow up. His blood pressure is controlled with lisinopril 40 mg/d. But his albumin-to-creatinine ratio is 75 mg/g, and your records reveal that his albuminuria is getting progressively worse.
You’re aware of the potential benefits of a dual angiotensin blockade, and are considering adding an angiotensin receptor blocker (ARB) to your patient’s medication regimen. You wonder whether the combination of an angiotensin-converting enzyme (ACE) inhibitor and an ARB will slow the decline of renal function. You also wonder whether the combination will reduce your patient’s cardiovascular risk.
ACE inhibitors are known to reduce cardiovascular morbidity and mortality, as well as proteinuria in patients with vascular disease or diabetes, whether or not they have heart failure.2 But few studies have compared the effects of ACE inhibitors and ARBs in high-risk patients without heart failure. Nor has there been a definitive study of the effects of an ACE inhibitor–ARB combination on proteinuria and cardiovascular risk.
Are 2 drugs better than 1?
In a recent meta-analysis, researchers reported that combination therapy had a beneficial effect on proteinuria.3 But that observation was based on a small number of patients (N=309 from 10 studies), short follow up, and a lack of data on key clinical end points such as decline of the glomerular filtration rate (GFR) and the onset of dialysis.
Other evidence comes from a study of 199 patients with diabetes and microalbuminuria, in which the ACE inhibitor-ARB combination reduced proteinuria more than either agent alone.4 And in a study of 336 patients with nondiabetic nephropathy, the 2-drug combination slowed the decline in renal function more than monotherapy.5
Small studies raise hopes. These preliminary findings, along with the theoretical benefits of dual angiotensin blockade, suggested that the benefits of taking both agents together could be significant. A large, well-done randomized controlled trial (RCT) was needed to determine the following: (1) whether an ARB is as effective as an ACE inhibitor in reducing morbidity and mortality in high-risk patients who don’t have heart failure, and (2) whether the ACE inhibitor–ARB combination is better than monotherapy for patients at high risk.
The ONTARGET study:
- established that telmisartan, an ARB, is not inferior to ramipril, an ACE inhibitor, in reducing cardiovascular and renal events in high-risk patients without heart failure.
- found that either drug alone is more effective than combination therapy for this patient population.
- cast fresh doubt on the assumption that proteinuria is an accurate surrogate marker for progressive renal dysfunction.
STUDY SUMMARY: Vascular outcomes same for ACE inhibitors, ARBs
The ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET), a multi-year study of thousands of patients, addressed both of those questions. The researchers compared the effects of both telmisartan (Micardis, an ARB) alone and a telmisartan + ramipril (Altace, an ACE inhibitor) combination with the effects of the ACE inhibitor alone in patients ≥55 years of age with established atherosclerotic vascular disease or diabetes with end-organ damage.1 Exclusion criteria included major renal artery stenosis, uncorrected volume or sodium depletion, a serum creatinine concentration of ≥3 mg/dL, and uncontrolled hypertension (>160 mm Hg systolic or >100 mm Hg diastolic).
After a 3-week run-in period to eliminate those who were unable to tolerate either medication or were nonadherent, a total of 25,620 patients remained. They were randomly assigned to take ramipril 10 mg/d, telmisartan 80 mg/d, or both the ACE inhibitor and the ARB. The researchers followed the patients for a median of 56 months.
The primary composite outcome was death from cardiovascular causes, myocardial infarction, stroke, or hospitalization for heart failure;1 the main renal outcome was a composite of first dialysis, doubling of serum creatinine, or death.6
The percentage of patients with the primary outcome was the same in all 3 groups (~16.5%). This finding was somewhat surprising because the blood pressure of patients in the combination therapy group was 2 to 3 mm Hg lower overall (both systolic and diastolic) than the blood pressure of patients on monotherapy—a difference that in other studies has been associated with an estimated 4% to 5% reduction in risk.1,2 Patients in the combination group had more hypotensive symptoms compared with those in the ramipril group (4.8% vs 1.7%, number needed to harm [NNH]=32, P<.001).
Renal dysfunction was highest in dual therapy group
Patients in the combination therapy group had higher rates of renal dysfunction than either the ramipril group (13.5% vs 10.2%, NNH=30, P<.001) or the telmisartan group (10.6%), despite a decrease in proteinuria among those on dual therapy. Patients taking the 2-drug combination also had higher rates of hyperkalemia.
While telmisartan proved to be equal to ramipril in reducing vascular events in high-risk patients, patients taking the ACE inhibitor experienced more cough (NNH=32, P<.001) and angioedema (NNH=500, P=.01). In both monotherapy groups, the rates of adverse drug reactions were probably lower than what we typically see in clinical practice because after the run-in period, only patients who were better able to tolerate both medications remained.
WHAT’S NEW: Combination causes renal impairment
This study established that telmisartan, an ARB, is not inferior to ramipril, an ACE inhibitor, in reducing cardiovascular and renal events in patients without heart failure. In addition, as the largest RCT to explore the effects of a dual blockade of the renin-angiotensin system with an ACE inhibitor and an ARB, it casts fresh doubt on the assumption that proteinuria is an accurate surrogate marker for progressive renal dysfunction. The reduction in proteinuria seen in patients in the combination therapy group came at a cost of increased renal impairment.
CAVEATS: Findings do not apply to heart failure patients
More than 11% of potential subjects were excluded from this study during the run-in period. This suggests that physicians in practice are likely to find a significant number of patients who are unable to tolerate (or fail to adhere to) monotherapy with ACE inhibitors or ARBs.
At baseline, only a small subgroup—13%—had overt diabetic nephropathy, the hallmark for a substantial continuous decline of GFR. However, 38% of the study group had diabetes, and almost 30% of these diabetes patients had microalbuminuria. Subgroup analysis found results consistent with the overall group, and the large sample size reduces the likelihood that these findings were due to low power. The overall rate of dialysis and doubling of serum creatinine was low, but still statistically significant, due to the large size of this study.
In determining treatment for high-risk patients with vascular disease or diabetes, it is important to keep the study population in mind. Studies of patients with poorly controlled congestive heart failure (CHF) have shown potential benefits from an ACE inhibitor–ARB combination.7 The ONTARGET trial specifically excluded individuals with CHF, and its findings—and recommendations to avoid combination therapy—should not be applied to heart failure patients.
CHALLENGES TO IMPLEMENTATION: Best microalbuminuria Tx remains elusive
Although albuminuria has been considered an early sign of the onset of diabetic nephropathy, the ONTARGET study demonstrated that combination therapy may cause further reduction in albuminuria but still adversely affect renal function. Thus, this study raises important questions about the best treatment for patients with diabetes who have microalbuminuria and are already on either an ACE inhibitor or an ARB. We wonder, too, whether we should continue to test for microalbuminuria in patients who are taking one of these agents, given the lack of guidance regarding further treatment.
Acknowledgements
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
PURLs methodology
This study was selected and evaluated using FPIN’s Priority Updates from the Research Literature (PURL) Surveillance System methodology. The criteria and findings leading to the selection of this study as a PURL can be accessed at www.jfponline.com/purls.
Click here to view PURL METHODOLOGY
Avoid prescribing an angiotensin-converting enzyme (ACE) inhibitor and an angiotensin receptor blocker (ARB) for patients at high risk of vascular events or renal dysfunction. The combination does not reduce poor outcomes, and leads to more adverse drug-related events than an ACE inhibitor or ARB alone.1
Strength of recommendation
B: 1 large, high-quality randomized controlled trial (RCT).
The ONTARGET investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547-1559.
ILLUSTRATIVE CASE
A 56-year-old patient with well-controlled type 2 diabetes and hypertension comes to see you for routine follow up. His blood pressure is controlled with lisinopril 40 mg/d. But his albumin-to-creatinine ratio is 75 mg/g, and your records reveal that his albuminuria is getting progressively worse.
You’re aware of the potential benefits of a dual angiotensin blockade, and are considering adding an angiotensin receptor blocker (ARB) to your patient’s medication regimen. You wonder whether the combination of an angiotensin-converting enzyme (ACE) inhibitor and an ARB will slow the decline of renal function. You also wonder whether the combination will reduce your patient’s cardiovascular risk.
ACE inhibitors are known to reduce cardiovascular morbidity and mortality, as well as proteinuria in patients with vascular disease or diabetes, whether or not they have heart failure.2 But few studies have compared the effects of ACE inhibitors and ARBs in high-risk patients without heart failure. Nor has there been a definitive study of the effects of an ACE inhibitor–ARB combination on proteinuria and cardiovascular risk.
Are 2 drugs better than 1?
In a recent meta-analysis, researchers reported that combination therapy had a beneficial effect on proteinuria.3 But that observation was based on a small number of patients (N=309 from 10 studies), short follow up, and a lack of data on key clinical end points such as decline of the glomerular filtration rate (GFR) and the onset of dialysis.
Other evidence comes from a study of 199 patients with diabetes and microalbuminuria, in which the ACE inhibitor-ARB combination reduced proteinuria more than either agent alone.4 And in a study of 336 patients with nondiabetic nephropathy, the 2-drug combination slowed the decline in renal function more than monotherapy.5
Small studies raise hopes. These preliminary findings, along with the theoretical benefits of dual angiotensin blockade, suggested that the benefits of taking both agents together could be significant. A large, well-done randomized controlled trial (RCT) was needed to determine the following: (1) whether an ARB is as effective as an ACE inhibitor in reducing morbidity and mortality in high-risk patients who don’t have heart failure, and (2) whether the ACE inhibitor–ARB combination is better than monotherapy for patients at high risk.
The ONTARGET study:
- established that telmisartan, an ARB, is not inferior to ramipril, an ACE inhibitor, in reducing cardiovascular and renal events in high-risk patients without heart failure.
- found that either drug alone is more effective than combination therapy for this patient population.
- cast fresh doubt on the assumption that proteinuria is an accurate surrogate marker for progressive renal dysfunction.
STUDY SUMMARY: Vascular outcomes same for ACE inhibitors, ARBs
The ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET), a multi-year study of thousands of patients, addressed both of those questions. The researchers compared the effects of both telmisartan (Micardis, an ARB) alone and a telmisartan + ramipril (Altace, an ACE inhibitor) combination with the effects of the ACE inhibitor alone in patients ≥55 years of age with established atherosclerotic vascular disease or diabetes with end-organ damage.1 Exclusion criteria included major renal artery stenosis, uncorrected volume or sodium depletion, a serum creatinine concentration of ≥3 mg/dL, and uncontrolled hypertension (>160 mm Hg systolic or >100 mm Hg diastolic).
After a 3-week run-in period to eliminate those who were unable to tolerate either medication or were nonadherent, a total of 25,620 patients remained. They were randomly assigned to take ramipril 10 mg/d, telmisartan 80 mg/d, or both the ACE inhibitor and the ARB. The researchers followed the patients for a median of 56 months.
The primary composite outcome was death from cardiovascular causes, myocardial infarction, stroke, or hospitalization for heart failure;1 the main renal outcome was a composite of first dialysis, doubling of serum creatinine, or death.6
The percentage of patients with the primary outcome was the same in all 3 groups (~16.5%). This finding was somewhat surprising because the blood pressure of patients in the combination therapy group was 2 to 3 mm Hg lower overall (both systolic and diastolic) than the blood pressure of patients on monotherapy—a difference that in other studies has been associated with an estimated 4% to 5% reduction in risk.1,2 Patients in the combination group had more hypotensive symptoms compared with those in the ramipril group (4.8% vs 1.7%, number needed to harm [NNH]=32, P<.001).
Renal dysfunction was highest in dual therapy group
Patients in the combination therapy group had higher rates of renal dysfunction than either the ramipril group (13.5% vs 10.2%, NNH=30, P<.001) or the telmisartan group (10.6%), despite a decrease in proteinuria among those on dual therapy. Patients taking the 2-drug combination also had higher rates of hyperkalemia.
While telmisartan proved to be equal to ramipril in reducing vascular events in high-risk patients, patients taking the ACE inhibitor experienced more cough (NNH=32, P<.001) and angioedema (NNH=500, P=.01). In both monotherapy groups, the rates of adverse drug reactions were probably lower than what we typically see in clinical practice because after the run-in period, only patients who were better able to tolerate both medications remained.
WHAT’S NEW: Combination causes renal impairment
This study established that telmisartan, an ARB, is not inferior to ramipril, an ACE inhibitor, in reducing cardiovascular and renal events in patients without heart failure. In addition, as the largest RCT to explore the effects of a dual blockade of the renin-angiotensin system with an ACE inhibitor and an ARB, it casts fresh doubt on the assumption that proteinuria is an accurate surrogate marker for progressive renal dysfunction. The reduction in proteinuria seen in patients in the combination therapy group came at a cost of increased renal impairment.
CAVEATS: Findings do not apply to heart failure patients
More than 11% of potential subjects were excluded from this study during the run-in period. This suggests that physicians in practice are likely to find a significant number of patients who are unable to tolerate (or fail to adhere to) monotherapy with ACE inhibitors or ARBs.
At baseline, only a small subgroup—13%—had overt diabetic nephropathy, the hallmark for a substantial continuous decline of GFR. However, 38% of the study group had diabetes, and almost 30% of these diabetes patients had microalbuminuria. Subgroup analysis found results consistent with the overall group, and the large sample size reduces the likelihood that these findings were due to low power. The overall rate of dialysis and doubling of serum creatinine was low, but still statistically significant, due to the large size of this study.
In determining treatment for high-risk patients with vascular disease or diabetes, it is important to keep the study population in mind. Studies of patients with poorly controlled congestive heart failure (CHF) have shown potential benefits from an ACE inhibitor–ARB combination.7 The ONTARGET trial specifically excluded individuals with CHF, and its findings—and recommendations to avoid combination therapy—should not be applied to heart failure patients.
CHALLENGES TO IMPLEMENTATION: Best microalbuminuria Tx remains elusive
Although albuminuria has been considered an early sign of the onset of diabetic nephropathy, the ONTARGET study demonstrated that combination therapy may cause further reduction in albuminuria but still adversely affect renal function. Thus, this study raises important questions about the best treatment for patients with diabetes who have microalbuminuria and are already on either an ACE inhibitor or an ARB. We wonder, too, whether we should continue to test for microalbuminuria in patients who are taking one of these agents, given the lack of guidance regarding further treatment.
Acknowledgements
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
PURLs methodology
This study was selected and evaluated using FPIN’s Priority Updates from the Research Literature (PURL) Surveillance System methodology. The criteria and findings leading to the selection of this study as a PURL can be accessed at www.jfponline.com/purls.
Click here to view PURL METHODOLOGY
1. The ONTARGET Investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547-1559.
2. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145-153.
3. Jennings DL, Kalus JS, Coleman CI, et al. Combination therapy with an ACE inhibitor and an angiotensin receptor blocker for diabetic nephropathy: a meta-analysis. Diabet Med. 2007;24:486-493.
4. Mogensen CE, Neldam S, Tikkanen I, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ. 2000;321:1440-1444.
5. Nakao N, Yoshimura A, Morita H, et al. Combination treatment of angiotensin-II receptor blocker and angiotensin-converting-enzyme inhibitor in non-diabetic renal disease (COOPERATE): a randomised controlled trial. Lancet. 2003;361:117-124.
6. Mann JF, Schmieder RE, McQueen M, et al. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet. 2008;372:547-553.
7. Cohn JN, Tognoni G. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345:1667-1675.
1. The ONTARGET Investigators. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547-1559.
2. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145-153.
3. Jennings DL, Kalus JS, Coleman CI, et al. Combination therapy with an ACE inhibitor and an angiotensin receptor blocker for diabetic nephropathy: a meta-analysis. Diabet Med. 2007;24:486-493.
4. Mogensen CE, Neldam S, Tikkanen I, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ. 2000;321:1440-1444.
5. Nakao N, Yoshimura A, Morita H, et al. Combination treatment of angiotensin-II receptor blocker and angiotensin-converting-enzyme inhibitor in non-diabetic renal disease (COOPERATE): a randomised controlled trial. Lancet. 2003;361:117-124.
6. Mann JF, Schmieder RE, McQueen M, et al. Renal outcomes with telmisartan, ramipril, or both, in people at high vascular risk (the ONTARGET study): a multicentre, randomised, double-blind, controlled trial. Lancet. 2008;372:547-553.
7. Cohn JN, Tognoni G. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345:1667-1675.
Copyright © 2009 The Family Physicians Inquiries Network.
All rights reserved.
A plea for help with another coding conundrum
In “10 billing & coding tips to boost your reimbursement” (November 2008), the codes for the flu and pneumococcal vaccines and a preventive medicine visit were incorrect. The correct codes are:
- 99397 (preventive medicine, established patient age ≥65)
- 90658 (flu vaccine, ≥3 years dosage)
- 90732 (pneumococcal vaccine, adult dosage).
As a further clarification, the article should have noted that the preventive medicine code would be used only if the patient received a physical examination.
Please do a follow-up to your article on billing and coding—specifically, on 99215. I realize many physicians are trying to work up from 99213 to 99214. But we have a mature practice with many complex patients and need help working up to 99215.
Here’s an example: A patient comes in with an asthmatic exacerbation due to sinusitis, and it’s likely a level 4. But the original purpose of the visit was a lipid panel review and follow-up for abnormal liver enzymes—another level 4. The patient also wants to discuss his colonoscopy report, which shows the presence of polyps.
Yes, I know I can make such patients schedule 3 separate appointments. But they’ll get mad, their boss will fire them, and it’s inefficient besides. I also know I can spend nearly 40 minutes with them and code for “counseling.” But suppose I can treat the asthma and infection in 6 minutes, discuss and alter the lipid Rx in 4 minutes, and review the colonoscopy results in 2. Now I have a 12-minute visit that I’m stuck billing at the rate of a 99214, which is “typically 25 minutes.”
I know I’m supposed to feel guilty, but I’m not going to do all that “free” work just because I can do it quickly and efficiently. So how do I go from 99214 to 99215 for such complex cases? I’m not going to call other physicians, chat for 40 minutes, or order unneeded tests just so I can “code up,” but I want to be paid for more than the asthma exacerbation because I’m doing more than that.
Andrew Johnstone, MD, Indianapolis
In “10 billing & coding tips to boost your reimbursement” (November 2008), the codes for the flu and pneumococcal vaccines and a preventive medicine visit were incorrect. The correct codes are:
- 99397 (preventive medicine, established patient age ≥65)
- 90658 (flu vaccine, ≥3 years dosage)
- 90732 (pneumococcal vaccine, adult dosage).
As a further clarification, the article should have noted that the preventive medicine code would be used only if the patient received a physical examination.
Please do a follow-up to your article on billing and coding—specifically, on 99215. I realize many physicians are trying to work up from 99213 to 99214. But we have a mature practice with many complex patients and need help working up to 99215.
Here’s an example: A patient comes in with an asthmatic exacerbation due to sinusitis, and it’s likely a level 4. But the original purpose of the visit was a lipid panel review and follow-up for abnormal liver enzymes—another level 4. The patient also wants to discuss his colonoscopy report, which shows the presence of polyps.
Yes, I know I can make such patients schedule 3 separate appointments. But they’ll get mad, their boss will fire them, and it’s inefficient besides. I also know I can spend nearly 40 minutes with them and code for “counseling.” But suppose I can treat the asthma and infection in 6 minutes, discuss and alter the lipid Rx in 4 minutes, and review the colonoscopy results in 2. Now I have a 12-minute visit that I’m stuck billing at the rate of a 99214, which is “typically 25 minutes.”
I know I’m supposed to feel guilty, but I’m not going to do all that “free” work just because I can do it quickly and efficiently. So how do I go from 99214 to 99215 for such complex cases? I’m not going to call other physicians, chat for 40 minutes, or order unneeded tests just so I can “code up,” but I want to be paid for more than the asthma exacerbation because I’m doing more than that.
Andrew Johnstone, MD, Indianapolis
In “10 billing & coding tips to boost your reimbursement” (November 2008), the codes for the flu and pneumococcal vaccines and a preventive medicine visit were incorrect. The correct codes are:
- 99397 (preventive medicine, established patient age ≥65)
- 90658 (flu vaccine, ≥3 years dosage)
- 90732 (pneumococcal vaccine, adult dosage).
As a further clarification, the article should have noted that the preventive medicine code would be used only if the patient received a physical examination.
Please do a follow-up to your article on billing and coding—specifically, on 99215. I realize many physicians are trying to work up from 99213 to 99214. But we have a mature practice with many complex patients and need help working up to 99215.
Here’s an example: A patient comes in with an asthmatic exacerbation due to sinusitis, and it’s likely a level 4. But the original purpose of the visit was a lipid panel review and follow-up for abnormal liver enzymes—another level 4. The patient also wants to discuss his colonoscopy report, which shows the presence of polyps.
Yes, I know I can make such patients schedule 3 separate appointments. But they’ll get mad, their boss will fire them, and it’s inefficient besides. I also know I can spend nearly 40 minutes with them and code for “counseling.” But suppose I can treat the asthma and infection in 6 minutes, discuss and alter the lipid Rx in 4 minutes, and review the colonoscopy results in 2. Now I have a 12-minute visit that I’m stuck billing at the rate of a 99214, which is “typically 25 minutes.”
I know I’m supposed to feel guilty, but I’m not going to do all that “free” work just because I can do it quickly and efficiently. So how do I go from 99214 to 99215 for such complex cases? I’m not going to call other physicians, chat for 40 minutes, or order unneeded tests just so I can “code up,” but I want to be paid for more than the asthma exacerbation because I’m doing more than that.
Andrew Johnstone, MD, Indianapolis
No Coughing Matter
Jaxon Hernandes, MD, a hospitalist with Apogee Physicians at Clara Maass Medical Center in Belleville, N.J., believes he and his colleagues are positioned perfectly to help properly diagnose asthma, a timely opinion given new Canadian research suggesting the bronchial condition routinely is over-diagnosed.
The study in the Canadian Medical Association Journal (2008;179(11):1121-1131) found up to 30% of adults diagnosed with asthma had no evidence of the condition. It included 496 people from eight Canadian cities who reported a diagnosis of asthma from their physician. The researchers' goal was to determine whether obese people were more likely to be misdiagnosed with asthma, but researchers found the issue was just as prevalent in people of normal weight.
Henderson notes hospitalists rarely make initial diagnoses when a patient is first encountered in the hospital, but once they are admitted, a hospitalist can order peak-flow-rate and spirometric tests. Clinical guidelines recommend using a spirometer to objectively measure long volume and airway flow.
"The hospitalist is in a position where he can get the pulmonologist to do what he needs to do," Dr. Hernandes says. "He can force a diagnosis being made."
Dr. Hernandes adds hospitalists have an onus to order the tests because doctors "may be under-diagnosing the primary issue with a patient or over-diagnosing and psychologically scarring them."
Jaxon Hernandes, MD, a hospitalist with Apogee Physicians at Clara Maass Medical Center in Belleville, N.J., believes he and his colleagues are positioned perfectly to help properly diagnose asthma, a timely opinion given new Canadian research suggesting the bronchial condition routinely is over-diagnosed.
The study in the Canadian Medical Association Journal (2008;179(11):1121-1131) found up to 30% of adults diagnosed with asthma had no evidence of the condition. It included 496 people from eight Canadian cities who reported a diagnosis of asthma from their physician. The researchers' goal was to determine whether obese people were more likely to be misdiagnosed with asthma, but researchers found the issue was just as prevalent in people of normal weight.
Henderson notes hospitalists rarely make initial diagnoses when a patient is first encountered in the hospital, but once they are admitted, a hospitalist can order peak-flow-rate and spirometric tests. Clinical guidelines recommend using a spirometer to objectively measure long volume and airway flow.
"The hospitalist is in a position where he can get the pulmonologist to do what he needs to do," Dr. Hernandes says. "He can force a diagnosis being made."
Dr. Hernandes adds hospitalists have an onus to order the tests because doctors "may be under-diagnosing the primary issue with a patient or over-diagnosing and psychologically scarring them."
Jaxon Hernandes, MD, a hospitalist with Apogee Physicians at Clara Maass Medical Center in Belleville, N.J., believes he and his colleagues are positioned perfectly to help properly diagnose asthma, a timely opinion given new Canadian research suggesting the bronchial condition routinely is over-diagnosed.
The study in the Canadian Medical Association Journal (2008;179(11):1121-1131) found up to 30% of adults diagnosed with asthma had no evidence of the condition. It included 496 people from eight Canadian cities who reported a diagnosis of asthma from their physician. The researchers' goal was to determine whether obese people were more likely to be misdiagnosed with asthma, but researchers found the issue was just as prevalent in people of normal weight.
Henderson notes hospitalists rarely make initial diagnoses when a patient is first encountered in the hospital, but once they are admitted, a hospitalist can order peak-flow-rate and spirometric tests. Clinical guidelines recommend using a spirometer to objectively measure long volume and airway flow.
"The hospitalist is in a position where he can get the pulmonologist to do what he needs to do," Dr. Hernandes says. "He can force a diagnosis being made."
Dr. Hernandes adds hospitalists have an onus to order the tests because doctors "may be under-diagnosing the primary issue with a patient or over-diagnosing and psychologically scarring them."
All Aboard the HM Train
A small Illinois hospital has joined the ranks of facilities—including its neighbors—in adding a hospital medicine (HM) program. The Genesis Medical Center, Illini Campus, in Silvas, Ill., began providing full-time hospitalist coverage on Dec. 1.
As the influx of hospitalists allowed local primary care physicians to stop making hospital visits, the Illini Campus rushed to fill the gap with its own HM program. "We were the last acute care hospital to implement a hospitalist program," says Chuck Bruhn, CEO of Illini Campus. "It had become a medical community issue."
Illini Campus, located near the Quad Cities on the western Illinois-eastern Iowa border, is a 149-bed facility with an average daily census of 50 to 55 patients. Its sister facility, the Genesis Medical Center in Davenport, Iowa, has had a successful hospital medicine program since 2005. Genesis' agreement with Cogent Healthcare, Inc., recently expanded to manage the program at Illini Campus, with round-the-clock coverage, including one full-time hospitalist.
Just two weeks after implementation, "the hospitalist program is growing much more rapidly than we had anticipated," Bruhn says. "They’re already covering a census of 14 patients a day. We're already talking about adding a physician extender."
Bruhn is pleased with the way the fledgling program has taken root. "We see it as a definite improvement, not only to quality and continuity of care, but to expediency of care. And the hospitalists provide additional support; they provide education to our clinical staff."
A small Illinois hospital has joined the ranks of facilities—including its neighbors—in adding a hospital medicine (HM) program. The Genesis Medical Center, Illini Campus, in Silvas, Ill., began providing full-time hospitalist coverage on Dec. 1.
As the influx of hospitalists allowed local primary care physicians to stop making hospital visits, the Illini Campus rushed to fill the gap with its own HM program. "We were the last acute care hospital to implement a hospitalist program," says Chuck Bruhn, CEO of Illini Campus. "It had become a medical community issue."
Illini Campus, located near the Quad Cities on the western Illinois-eastern Iowa border, is a 149-bed facility with an average daily census of 50 to 55 patients. Its sister facility, the Genesis Medical Center in Davenport, Iowa, has had a successful hospital medicine program since 2005. Genesis' agreement with Cogent Healthcare, Inc., recently expanded to manage the program at Illini Campus, with round-the-clock coverage, including one full-time hospitalist.
Just two weeks after implementation, "the hospitalist program is growing much more rapidly than we had anticipated," Bruhn says. "They’re already covering a census of 14 patients a day. We're already talking about adding a physician extender."
Bruhn is pleased with the way the fledgling program has taken root. "We see it as a definite improvement, not only to quality and continuity of care, but to expediency of care. And the hospitalists provide additional support; they provide education to our clinical staff."
A small Illinois hospital has joined the ranks of facilities—including its neighbors—in adding a hospital medicine (HM) program. The Genesis Medical Center, Illini Campus, in Silvas, Ill., began providing full-time hospitalist coverage on Dec. 1.
As the influx of hospitalists allowed local primary care physicians to stop making hospital visits, the Illini Campus rushed to fill the gap with its own HM program. "We were the last acute care hospital to implement a hospitalist program," says Chuck Bruhn, CEO of Illini Campus. "It had become a medical community issue."
Illini Campus, located near the Quad Cities on the western Illinois-eastern Iowa border, is a 149-bed facility with an average daily census of 50 to 55 patients. Its sister facility, the Genesis Medical Center in Davenport, Iowa, has had a successful hospital medicine program since 2005. Genesis' agreement with Cogent Healthcare, Inc., recently expanded to manage the program at Illini Campus, with round-the-clock coverage, including one full-time hospitalist.
Just two weeks after implementation, "the hospitalist program is growing much more rapidly than we had anticipated," Bruhn says. "They’re already covering a census of 14 patients a day. We're already talking about adding a physician extender."
Bruhn is pleased with the way the fledgling program has taken root. "We see it as a definite improvement, not only to quality and continuity of care, but to expediency of care. And the hospitalists provide additional support; they provide education to our clinical staff."
NFL Star’s Injury Spotlights Reporting Requirements
The recent suspension of a New York City doctor who failed to report a gunshot wound suffered by a football star is an opportunity for hospitalists to revisit their own reporting requirements, the president of SHM's NYC chapter says.
Josyann Abisaab, MD, of New York-Presbyterian Hospital/Weill Cornell Medical Center, was suspended after treating New York Giants wide receiver Plaxico Burress on Nov. 29. Less than a year after catching the winning touchdown pass in the Giants' Super Bowl victory, Burress, who told police he accidentally shot himself in the thigh at a NYC nightclub, has been suspended by the league and charged with criminal possession of a gun.
"I was not aware that something like this needed a report to the police," says Bradley Flansbaum, DO, MPH, chief of hospitalist services at Lenox Hill Hospital in Manhattan and president of SHM’s NYC chapter. "It opened up space in my brain. If I were confronted with this, when would I know when to and when not to call the police?"
Complicating matters is the fact hospitalists may have to report issues to more than just law enforcement; depending on diagnoses and patient histories, doctors may have to notify state and federal health agencies or social service departments. Rules vary by state, so Dr. Flansbaum says hospitalists would do well to brush up on their requirements and liabilities.
"I may not know the rules," Dr. Flansbaum said, "but I certainly would speak to the right people here and ask them: 'What are my obligations? How do I protect myself and the patient?' " He recommends hospitalists verify local requirements with their hospital administration.
The recent suspension of a New York City doctor who failed to report a gunshot wound suffered by a football star is an opportunity for hospitalists to revisit their own reporting requirements, the president of SHM's NYC chapter says.
Josyann Abisaab, MD, of New York-Presbyterian Hospital/Weill Cornell Medical Center, was suspended after treating New York Giants wide receiver Plaxico Burress on Nov. 29. Less than a year after catching the winning touchdown pass in the Giants' Super Bowl victory, Burress, who told police he accidentally shot himself in the thigh at a NYC nightclub, has been suspended by the league and charged with criminal possession of a gun.
"I was not aware that something like this needed a report to the police," says Bradley Flansbaum, DO, MPH, chief of hospitalist services at Lenox Hill Hospital in Manhattan and president of SHM’s NYC chapter. "It opened up space in my brain. If I were confronted with this, when would I know when to and when not to call the police?"
Complicating matters is the fact hospitalists may have to report issues to more than just law enforcement; depending on diagnoses and patient histories, doctors may have to notify state and federal health agencies or social service departments. Rules vary by state, so Dr. Flansbaum says hospitalists would do well to brush up on their requirements and liabilities.
"I may not know the rules," Dr. Flansbaum said, "but I certainly would speak to the right people here and ask them: 'What are my obligations? How do I protect myself and the patient?' " He recommends hospitalists verify local requirements with their hospital administration.
The recent suspension of a New York City doctor who failed to report a gunshot wound suffered by a football star is an opportunity for hospitalists to revisit their own reporting requirements, the president of SHM's NYC chapter says.
Josyann Abisaab, MD, of New York-Presbyterian Hospital/Weill Cornell Medical Center, was suspended after treating New York Giants wide receiver Plaxico Burress on Nov. 29. Less than a year after catching the winning touchdown pass in the Giants' Super Bowl victory, Burress, who told police he accidentally shot himself in the thigh at a NYC nightclub, has been suspended by the league and charged with criminal possession of a gun.
"I was not aware that something like this needed a report to the police," says Bradley Flansbaum, DO, MPH, chief of hospitalist services at Lenox Hill Hospital in Manhattan and president of SHM’s NYC chapter. "It opened up space in my brain. If I were confronted with this, when would I know when to and when not to call the police?"
Complicating matters is the fact hospitalists may have to report issues to more than just law enforcement; depending on diagnoses and patient histories, doctors may have to notify state and federal health agencies or social service departments. Rules vary by state, so Dr. Flansbaum says hospitalists would do well to brush up on their requirements and liabilities.
"I may not know the rules," Dr. Flansbaum said, "but I certainly would speak to the right people here and ask them: 'What are my obligations? How do I protect myself and the patient?' " He recommends hospitalists verify local requirements with their hospital administration.
Research Roundup
Question: Is sodium bicarbonate superior to sodium chloride in preventing contrast-induced nephropathy in patients with chronic kidney disease (CKD) undergoing coronary angiography?
Background: Sodium bicarbonate has been suggested as a possible strategy to prevent contrast-induced nephropathy—a condition that can lead to prolonged hospitalization, increased healthcare costs, and substantial morbidity and mortality.
Study design: Randomized, controlled, single-blind study.
Setting: Kaiser Permanente Medical Center, Los Angeles.
Synopsis: Data were collected over 12 months by selecting 353 patients with stable CKD undergoing coronary angiography who were 18 or older and had an estimated glomerular filtration rate (GFR) of 60mL/min/1.73m2 or less and one or more of diabetes mellitus, congestive heart failure, hypertension, or age older than 75 years. Patients were randomized to received sodium chloride (n=178) and sodium bicarbonate (n=175) at the same rate and duration.
There was no statistical difference in the primary endpoint (p=0.82), which was a 25% or greater decrease in the GFR on days 1 through 4 after contrast exposure.
Study results were limited by several factors; most importantly, it was not a double-blinded study and was performed at a single center. Also, the sodium content of the two fluids varied; normal saline carried 154mEq and sodium bicarbonate 130mEq of sodium, respectively.
Bottom line: Hydration with sodium bicarbonate is not superior to sodium chloride in preventing contrast-induced nephropathy in patients with moderate to severe CKD undergoing coronary angiography.
Citation: JAMA. 2008;300(9):1038-1046
—Reviewed for the e-wire by Elbert Chun, MD, John Vazquez, MD, Larry Beer, MD, Maged Doss, MD, Vana Bollineni, MD, Mohammed S. Singapuri, MD, Dan Dressler, MD, MsCR, Emory University Hospital, Atlanta
Question: Is sodium bicarbonate superior to sodium chloride in preventing contrast-induced nephropathy in patients with chronic kidney disease (CKD) undergoing coronary angiography?
Background: Sodium bicarbonate has been suggested as a possible strategy to prevent contrast-induced nephropathy—a condition that can lead to prolonged hospitalization, increased healthcare costs, and substantial morbidity and mortality.
Study design: Randomized, controlled, single-blind study.
Setting: Kaiser Permanente Medical Center, Los Angeles.
Synopsis: Data were collected over 12 months by selecting 353 patients with stable CKD undergoing coronary angiography who were 18 or older and had an estimated glomerular filtration rate (GFR) of 60mL/min/1.73m2 or less and one or more of diabetes mellitus, congestive heart failure, hypertension, or age older than 75 years. Patients were randomized to received sodium chloride (n=178) and sodium bicarbonate (n=175) at the same rate and duration.
There was no statistical difference in the primary endpoint (p=0.82), which was a 25% or greater decrease in the GFR on days 1 through 4 after contrast exposure.
Study results were limited by several factors; most importantly, it was not a double-blinded study and was performed at a single center. Also, the sodium content of the two fluids varied; normal saline carried 154mEq and sodium bicarbonate 130mEq of sodium, respectively.
Bottom line: Hydration with sodium bicarbonate is not superior to sodium chloride in preventing contrast-induced nephropathy in patients with moderate to severe CKD undergoing coronary angiography.
Citation: JAMA. 2008;300(9):1038-1046
—Reviewed for the e-wire by Elbert Chun, MD, John Vazquez, MD, Larry Beer, MD, Maged Doss, MD, Vana Bollineni, MD, Mohammed S. Singapuri, MD, Dan Dressler, MD, MsCR, Emory University Hospital, Atlanta
Question: Is sodium bicarbonate superior to sodium chloride in preventing contrast-induced nephropathy in patients with chronic kidney disease (CKD) undergoing coronary angiography?
Background: Sodium bicarbonate has been suggested as a possible strategy to prevent contrast-induced nephropathy—a condition that can lead to prolonged hospitalization, increased healthcare costs, and substantial morbidity and mortality.
Study design: Randomized, controlled, single-blind study.
Setting: Kaiser Permanente Medical Center, Los Angeles.
Synopsis: Data were collected over 12 months by selecting 353 patients with stable CKD undergoing coronary angiography who were 18 or older and had an estimated glomerular filtration rate (GFR) of 60mL/min/1.73m2 or less and one or more of diabetes mellitus, congestive heart failure, hypertension, or age older than 75 years. Patients were randomized to received sodium chloride (n=178) and sodium bicarbonate (n=175) at the same rate and duration.
There was no statistical difference in the primary endpoint (p=0.82), which was a 25% or greater decrease in the GFR on days 1 through 4 after contrast exposure.
Study results were limited by several factors; most importantly, it was not a double-blinded study and was performed at a single center. Also, the sodium content of the two fluids varied; normal saline carried 154mEq and sodium bicarbonate 130mEq of sodium, respectively.
Bottom line: Hydration with sodium bicarbonate is not superior to sodium chloride in preventing contrast-induced nephropathy in patients with moderate to severe CKD undergoing coronary angiography.
Citation: JAMA. 2008;300(9):1038-1046
—Reviewed for the e-wire by Elbert Chun, MD, John Vazquez, MD, Larry Beer, MD, Maged Doss, MD, Vana Bollineni, MD, Mohammed S. Singapuri, MD, Dan Dressler, MD, MsCR, Emory University Hospital, Atlanta
Study shows imatinib response is durable and improves with time

SAN FRANCISCO—The longest duration study of imatinib treatment for patients with Ph+ CML shows 86% of patients are still alive 7 years after beginning therapy.
The International Randomized Interferon versus STI571 (IRIS) study revealed only 1 early chronic-phase patient progressed to a more advanced phase between years 6 and 7, Stephen O’Brien, MD, PhD, of Newcastle University in the UK, said at the 50th Annual Meeting of the American Society of Hematology.
IRIS is an open-label, phase 3 clinical trial enrolling 1106 newly diagnosed patients with chronic phase Ph+ CML in 177 centers across 16 countries. One group of 553 patients received imatinib 400 mg per day. Another group of 553 patients received a target dose of interferon (IFN) of 5 MIU/m2/day in combination with cytarabine at 20 mg/m2/day for 10 days each month.
Because of tolerability issues, lack of response, or loss of response, 65% of patients in the IFN/cytarabine arm crossed over to the imatinib arm. Only 3% of patients in the imatinib arm crossed over to the IFN/cytarabine arm.
A low rate of progression has been reported every year since this trial began in 2001. Seven percent of patients treated with imatinib progressed to advanced phases of CML after 7 years. Of the 456 patients (82%) who achieved a complete cytogenetic response, 17% lost their response and 3% progressed to advanced phases.
“After 1 year of treatment, there is a small risk of progression,” Dr O’Brien said. “If patients achieve and maintain a complete cytogenetic response after 3 years, they are fairly safe.”
Treatment with imatinib in the IRIS study was well tolerated, he said. No new serious adverse events occurred between the sixth and seventh year of treatment.
The results from the IRIS study also reveal that, by year 6, 85% to 90% of patients still taking imatinib achieved a major molecular response. This key milestone indicates a reduction in the abnormal protein responsible for the uncontrolled production of abnormal white blood cells and may be a sensitive predictor of long-term progression-free survival.
“There was a steady improvement in major molecular responses between 4 and 7 years of treatment,” said Timothy Hughes, MD, of the Institute of Medical and Veterinary Science in Adelaide, Australia. “By 7 years, the vast majority of patients who achieved a complete cytogenetic response also achieved a major molecular response.”
A major molecular response at any time point represents a “safe haven” for patients, Dr Hughes said. Both molecular and cytogenetic evaluations should be used to guide treatment decisions until a complete cytogenetic response is achieved, followed by measurements of molecular assessments.
“In this, the seventh year of the IRIS study, CML patients treated with imatinib continue to demonstrate impressive long-term survival,” Dr O’Brien said. “Imatinib 400 mg daily is confirmed as the standard of care for the initial therapy of chronic-phase CML.”
Imatinib, the first therapy to inhibit the activity of Bcr-Abl, revolutionized the treatment of Ph+ CML, Dr O’Brien said. Prior to imatinib, about 50% of patients with Ph+ CML progressed from the initial phase to more advanced stages after 3 to 5 years. Once patients reached the final blast crisis phase, survival was generally 3 to 6 months. ![]()

SAN FRANCISCO—The longest duration study of imatinib treatment for patients with Ph+ CML shows 86% of patients are still alive 7 years after beginning therapy.
The International Randomized Interferon versus STI571 (IRIS) study revealed only 1 early chronic-phase patient progressed to a more advanced phase between years 6 and 7, Stephen O’Brien, MD, PhD, of Newcastle University in the UK, said at the 50th Annual Meeting of the American Society of Hematology.
IRIS is an open-label, phase 3 clinical trial enrolling 1106 newly diagnosed patients with chronic phase Ph+ CML in 177 centers across 16 countries. One group of 553 patients received imatinib 400 mg per day. Another group of 553 patients received a target dose of interferon (IFN) of 5 MIU/m2/day in combination with cytarabine at 20 mg/m2/day for 10 days each month.
Because of tolerability issues, lack of response, or loss of response, 65% of patients in the IFN/cytarabine arm crossed over to the imatinib arm. Only 3% of patients in the imatinib arm crossed over to the IFN/cytarabine arm.
A low rate of progression has been reported every year since this trial began in 2001. Seven percent of patients treated with imatinib progressed to advanced phases of CML after 7 years. Of the 456 patients (82%) who achieved a complete cytogenetic response, 17% lost their response and 3% progressed to advanced phases.
“After 1 year of treatment, there is a small risk of progression,” Dr O’Brien said. “If patients achieve and maintain a complete cytogenetic response after 3 years, they are fairly safe.”
Treatment with imatinib in the IRIS study was well tolerated, he said. No new serious adverse events occurred between the sixth and seventh year of treatment.
The results from the IRIS study also reveal that, by year 6, 85% to 90% of patients still taking imatinib achieved a major molecular response. This key milestone indicates a reduction in the abnormal protein responsible for the uncontrolled production of abnormal white blood cells and may be a sensitive predictor of long-term progression-free survival.
“There was a steady improvement in major molecular responses between 4 and 7 years of treatment,” said Timothy Hughes, MD, of the Institute of Medical and Veterinary Science in Adelaide, Australia. “By 7 years, the vast majority of patients who achieved a complete cytogenetic response also achieved a major molecular response.”
A major molecular response at any time point represents a “safe haven” for patients, Dr Hughes said. Both molecular and cytogenetic evaluations should be used to guide treatment decisions until a complete cytogenetic response is achieved, followed by measurements of molecular assessments.
“In this, the seventh year of the IRIS study, CML patients treated with imatinib continue to demonstrate impressive long-term survival,” Dr O’Brien said. “Imatinib 400 mg daily is confirmed as the standard of care for the initial therapy of chronic-phase CML.”
Imatinib, the first therapy to inhibit the activity of Bcr-Abl, revolutionized the treatment of Ph+ CML, Dr O’Brien said. Prior to imatinib, about 50% of patients with Ph+ CML progressed from the initial phase to more advanced stages after 3 to 5 years. Once patients reached the final blast crisis phase, survival was generally 3 to 6 months. ![]()

SAN FRANCISCO—The longest duration study of imatinib treatment for patients with Ph+ CML shows 86% of patients are still alive 7 years after beginning therapy.
The International Randomized Interferon versus STI571 (IRIS) study revealed only 1 early chronic-phase patient progressed to a more advanced phase between years 6 and 7, Stephen O’Brien, MD, PhD, of Newcastle University in the UK, said at the 50th Annual Meeting of the American Society of Hematology.
IRIS is an open-label, phase 3 clinical trial enrolling 1106 newly diagnosed patients with chronic phase Ph+ CML in 177 centers across 16 countries. One group of 553 patients received imatinib 400 mg per day. Another group of 553 patients received a target dose of interferon (IFN) of 5 MIU/m2/day in combination with cytarabine at 20 mg/m2/day for 10 days each month.
Because of tolerability issues, lack of response, or loss of response, 65% of patients in the IFN/cytarabine arm crossed over to the imatinib arm. Only 3% of patients in the imatinib arm crossed over to the IFN/cytarabine arm.
A low rate of progression has been reported every year since this trial began in 2001. Seven percent of patients treated with imatinib progressed to advanced phases of CML after 7 years. Of the 456 patients (82%) who achieved a complete cytogenetic response, 17% lost their response and 3% progressed to advanced phases.
“After 1 year of treatment, there is a small risk of progression,” Dr O’Brien said. “If patients achieve and maintain a complete cytogenetic response after 3 years, they are fairly safe.”
Treatment with imatinib in the IRIS study was well tolerated, he said. No new serious adverse events occurred between the sixth and seventh year of treatment.
The results from the IRIS study also reveal that, by year 6, 85% to 90% of patients still taking imatinib achieved a major molecular response. This key milestone indicates a reduction in the abnormal protein responsible for the uncontrolled production of abnormal white blood cells and may be a sensitive predictor of long-term progression-free survival.
“There was a steady improvement in major molecular responses between 4 and 7 years of treatment,” said Timothy Hughes, MD, of the Institute of Medical and Veterinary Science in Adelaide, Australia. “By 7 years, the vast majority of patients who achieved a complete cytogenetic response also achieved a major molecular response.”
A major molecular response at any time point represents a “safe haven” for patients, Dr Hughes said. Both molecular and cytogenetic evaluations should be used to guide treatment decisions until a complete cytogenetic response is achieved, followed by measurements of molecular assessments.
“In this, the seventh year of the IRIS study, CML patients treated with imatinib continue to demonstrate impressive long-term survival,” Dr O’Brien said. “Imatinib 400 mg daily is confirmed as the standard of care for the initial therapy of chronic-phase CML.”
Imatinib, the first therapy to inhibit the activity of Bcr-Abl, revolutionized the treatment of Ph+ CML, Dr O’Brien said. Prior to imatinib, about 50% of patients with Ph+ CML progressed from the initial phase to more advanced stages after 3 to 5 years. Once patients reached the final blast crisis phase, survival was generally 3 to 6 months. ![]()