User login
A Pain in the Bone
A 71‐year‐old man presented to a hospital with a one week history of fatigue, polyuria, and polydipsia. He also reported pain in his back, hips, and ribs, in addition to frequent falls, intermittent confusion, constipation, and a weight loss of 10 pounds over the last 2 weeks. He denied cough, shortness of breath, chest pain, fever, night sweats, headache, and focal weakness.
Polyuria, which is often associated with polydipsia, can be arbitrarily defined as a urine output exceeding 3 L per day. After excluding osmotic diuresis due to uncontrolled diabetes mellitus, the 3 major causes of polyuria are primary polydipsia, central diabetes insipidus, and nephrogenic diabetes insipidus. Approximately 30% to 50% of cases of central diabetes insipidus are idiopathic; however, primary or secondary brain tumors or infiltrative diseases involving the hypothalamic‐pituitary region need to be considered in this 71‐year‐old man. The most common causes of nephrogenic diabetes insipidus in adults are chronic lithium ingestion, hypokalemia, and hypercalcemia. The patient describes symptoms that can result from severe hypercalcemia, including fatigue, confusion, constipation, polyuria, and polydipsia.
The patient's past medical history included long‐standing, insulin‐requiring type 2 diabetes with associated complications including coronary artery disease, transient ischemic attacks, proliferative retinopathy, peripheral diabetic neuropathy, and nephropathy. Seven years prior to presentation, he received a cadaveric renal transplant that was complicated by BK virus (polyomavirus) nephropathy and secondary hyperparathyroidism. Three years after his transplant surgery, he developed squamous cell carcinoma of the skin, which was treated with local surgical resection. Two years after that, he developed stage I laryngeal cancer of the glottis and received laser surgery, and since then he had been considered disease‐free. He also had a history of hypertension, hypercholesterolemia, osteoporosis, and depression. His medications included aspirin, amlodipine, metoprolol succinate, valsartan, furosemide, simvastatin, insulin, prednisone, sirolimus, and sulfamethoxazole/trimethoprim. He was a married psychiatrist. He denied tobacco use and reported occasional alcohol use.
The prolonged immunosuppressive therapy that is required following organ transplantation carries a markedly increased risk of the subsequent development of malignant tumors, including cancers of the lips and skin, lymphoproliferative disorders, and bronchogenic carcinoma. Primary brain lymphoma resulting in central diabetes insipidus would be unlikely in the absence of headache or focal weakness. An increased risk of lung cancer occurs in recipients of heart and lung transplants, and to a much lesser degree, recipients of kidney transplants. However, metastatic lung cancer is less likely in the absence of respiratory symptoms and smoking history (present in approximately 90% of all lung cancers). Nephrogenic diabetes insipidus, in its mild form, is relatively common in elderly patients with acute or chronic renal insufficiency because of a reduction in maximum urinary concentrating ability. On the other hand, this alone does not explain his remaining symptoms. The instinctive diagnosis in this case is tertiary hyperparathyroidism due to progression of untreated secondary hyperparathyroidism. This causes hypercalcemia, nephrogenic diabetes insipidus, and significant bone pain related to renal osteodystrophy.
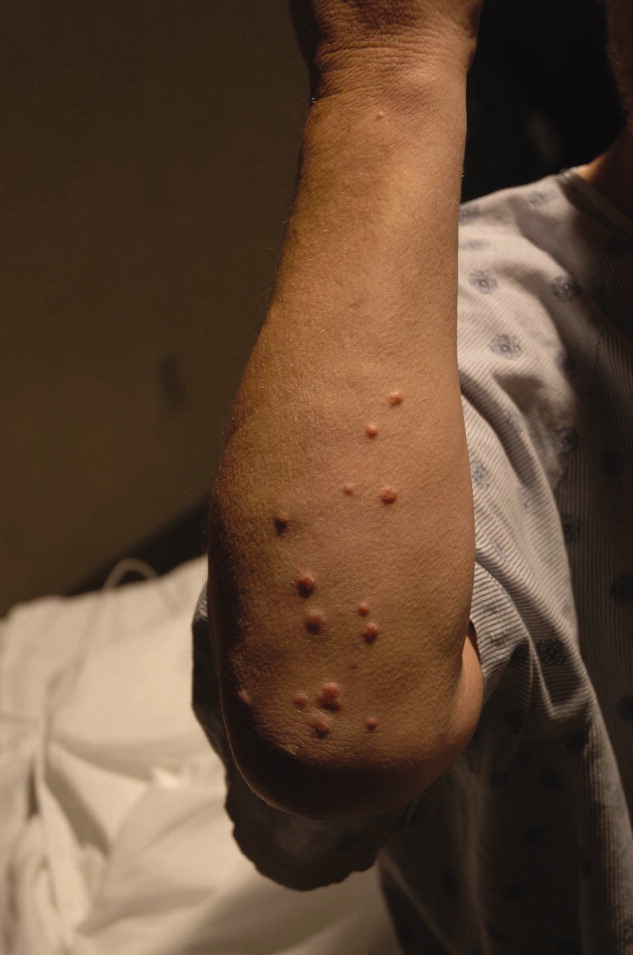
On physical exam, the patient appeared chronically ill, but was in no acute distress. He weighed 197.6 pounds and his height was 70.5 inches. He was afebrile with a blood pressure of 146/82 mm Hg, a heart rate of 76 beats per minute, a respiratory rate of 12 breaths per minute, and an oxygen saturation of 97% while breathing room air. He had no generalized lymphadenopathy. Thyroid examination was unremarkable. Examination of the lungs, heart, abdomen, and lower extremities was normal. The rectal examination revealed no masses or prostate nodules; a test for fecal occult blood was negative. He had loss of sensation to light touch and vibration in the feet with absent Achilles deep tendon reflexes. He had a poorly healing surgical wound on his forehead at the site of his prior skin cancer, but no rash or other lesions. There was no joint swelling or erythema. There were tender points over the cervical, thoracic, and lumbar spine; on multiple ribs; and on the pelvic rims.
Perhaps of greatest importance is the lack of lymphadenopathy, organomegaly, or other findings suggestive of diffuse lymphoproliferative disease. His multifocal bone tenderness is concerning for renal osteodystrophy, multiple myeloma, or primary or metastatic bone disease. Cancers in men that metastasize to the bone usually originate from the prostate, lung, kidney, or thyroid gland. In any case, his physical examination did not reveal an enlarged, asymmetric, or nodular prostate or thyroid gland. I recommend a chest film to rule out primary lung malignancy and a basic laboratory evaluation to narrow down the differential diagnosis.
A complete blood count showed a normocytic anemia with a hemoglobin of 8.7 g/dL and a hematocrit of 25%. Other laboratory tests revealed the following values: sodium, 139 mmol/L; potassium, 4.1 mmol/L; blood urea nitrogen, 70 mg/dL; creatinine, 3.5 mg/dL (most recent value 2 months ago was 1.9 mg/dL); total calcium, 13.2 mg/dL (normal range, 8.5‐10.5 mg/dL); phosphate, 5.3 mg/dL; magnesium, 2.5 mg/dL; total bilirubin, 0.5 mg/dL; alkaline phosphatase, 130 U/L; aspartate aminotransferase, 28 U/L; alanine aminotransferase, 19 U/L; albumin, 3.5 g/dL; and lactate dehydrogenase (LDH), 1258 IU/L (normal range, 105‐333 IU/L). A chest radiograph was normal.
The most important laboratory findings are severe hypercalcemia, acute on chronic renal failure, and anemia. Hypercalcemia most commonly results from malignancy or hyperparathyroidism. Less frequently, hypercalcemia may result from sarcoidosis, vitamin D intoxication, or hyperthyroidism. The degree of hypercalcemia is useful diagnostically as hyperparathyroidism commonly results in mild hypercalcemia (serum calcium concentration often below 11 mg/dL). Values above 13 mg/dL are unusual in hyperparathyroidism and are most often due to malignancy. Malignancy is often evident clinically by the time it causes hypercalcemia, and patients with hypercalcemia of malignancy are more often symptomatic than those with hyperparathyroidism. Additionally, localized bone pain and weight loss do not result from hypercalcemia itself and their presence also raises concern for malignancy.
Nonmelanoma skin cancer is the most common cancer occurring after transplantation but does not cause hypercalcemia. Squamous cancers of the head and neck can rarely cause hypercalcemia due to secretion of parathyroid hormone‐related peptide; however, his early‐stage laryngeal cancer and the expected high likelihood of cure argue against this possibility. Osteolytic metastases account for approximately 20% of cases of hypercalcemia of malignancy (Table 1). Prostate cancer rarely results in hypercalcemia since bone metastases are predominantly osteoblastic, whereas metastatic non‐small‐cell lung cancer, thyroid cancer, and kidney cancer more commonly cause hypercalcemia due to osteolytic bone lesions. The total alkaline phosphatase has been traditionally used to assess the osteoblastic component of bone remodeling. Its normal level tends to predict a negative bone scan and supports the likelihood of lytic lesions. Posttransplantation lymphoproliferative disorders, which include a wide range of syndromes, can rarely result in hypercalcemia. I am also worried about the possibility of multiple myeloma as he has the classic triad of hypercalcemia, bone pain, and subacute kidney injury.
|
| Osteolytic metastases |
| Breast cancer |
| Multiple myeloma |
| Lymphoma |
| Leukemia |
| Humoral hypercalcemia (PTH‐related protein) |
| Squamous cell carcinomas |
| Renal carcinomas |
| Bladder carcinoma |
| Breast cancer |
| Ovarian carcinoma |
| Leukemia |
| Lymphoma |
| 1,25‐Dihydroxyvitamin D secretion |
| Lymphoma |
| Ovarian dysgerminomas |
| Ectopic PTH secretion (rare) |
| Ovarian carcinoma |
| Lung carcinomas |
| Neuroectodermal tumor |
| Thyroid papillary carcinoma |
| Rhabdomyosarcoma |
| Pancreatic cancer |
The first purpose of the laboratory evaluation is to differentiate parathyroid hormone (PTH)‐mediated hypercalcemia (primary and tertiary hyperparathyroidism) from non‐PTH‐mediated hypercalcemia (primarily malignancy, hyperthyroidism, vitamin D intoxication, and granulomatous disease). The production of vitamin D metabolites, PTH‐related protein, or hypercalcemia from osteolysis in these latter cases results in suppressed PTH levels.
In severe elevations of calcium, the initial goals of treatment are directed toward fluid resuscitation with normal saline and, unless contraindicated, the immediate institution of bisphosphonate therapy. A loop diuretic such as furosemide is often used, but a recent review concluded that there is little evidence to support its use in this setting.
The patient was admitted and treated with intravenous saline and furosemide. Additional laboratory evaluation revealed normal levels of prostate‐specific antigen and thyroid‐stimulating hormone. PTH was 44 pg/mL (the most recent value was 906 pg/mL eight years ago; normal range, 15‐65 pg/mL) and beta‐2 microglobulin (B2M) was 8 mg/L (normal range, 0.8‐2.2 mg/L).
The normal PTH level makes tertiary hyperparathyroidism unlikely and points toward non‐PTH‐related hypercalcemia. An elevated B2M level may occur in patients with chronic graft rejection, renal tubular dysfunction, dialysis‐related amyloidosis, multiple myeloma, or lymphoma. LDH is often elevated in patients with multiple myeloma and lymphoma, but this is not a specific finding. The next laboratory test would be measurement of PTH‐related protein and vitamin D metabolites, as these tests can differentiate between the causes of non‐PTH‐mediated hypercalcemia.
Serum concentrations of the vitamin D metabolites, 25‐hydroxyvitamin D (calcidiol) and 1,25‐dihydroxyvitamin D (calcitriol), were low‐normal. PTH‐related protein was not detected.
The marked elevation of serum LDH and B2M, the relatively suppressed PTH level, combined with undetectable PTH‐related protein suggest multiple myeloma or lymphoma as the likely cause of the patient's clinical presentation. The combination of hypercalcemia and multifocal bone pain makes multiple myeloma the leading diagnosis as hypercalcemia is uncommon in patients with lymphoma, especially at the time of initial clinical presentation.
I would proceed with serum and urine protein electrophoresis (SPEP and UPEP, respectively) and a skeletal survey. If these tests do not confirm the diagnosis of multiple myeloma, I would order a noncontrast computed tomography (CT) of the chest and abdomen and a magnetic resonance imaging (MRI) of the spine. In addition, I would like to monitor his response to the intravenous saline and furosemide.
Forty‐eight hours after presentation, repeat serum calcium and creatinine levels were 11.3 mg/dL and 2.9 mg/dL, respectively. He received salmon calcitonin 4 U/kg every 12 hours. Pamidronate was avoided because of his kidney disease. His confusion resolved. He received intravenous morphine intermittently to alleviate his bone pain.
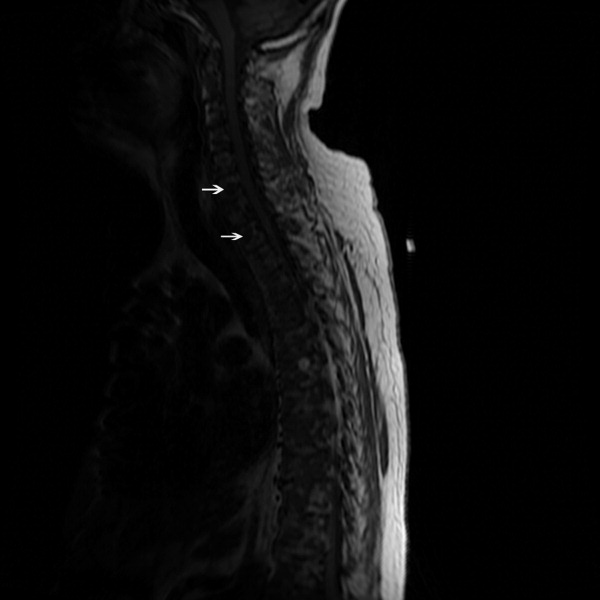
The SPEP revealed a monoclonal immunoglobulin G (IgG) lambda (light chain) spike representing roughly 3% (200 mg/dL) of total protein. His serum Ig levels were normal. The UPEP was negative for monoclonal immunoglobulin and Bence‐Jones protein. The skeletal survey revealed marked osteopenia, and the bone scan was normal. An MRI of the spine showed multiple round lesions in the cervical, thoracic, and lumbar spine (Figure 1). A CT of the chest showed similar bone lesions in the ribs and pelvis. A CT of the abdomen and chest did not suggest any primary malignancy nor did it show thoracic or abdominal lymphadenopathy.
The lack of lymphadenopathy, splenomegaly, or a visceral mass by CT imaging and physical examination, along with the normal PSA level, exclude most common forms of non‐Hodgkin lymphoma and bone metastasis from solid tumors. In multiple myeloma, cytokines secreted by plasma cells suppress osteoblast activity; therefore, while discrete lytic bone lesions are apparent on skeletal survey, the bone scan is typically normal. The absence of lytic lesions, normal serum immunoglobulin levels, and unremarkable UPEP make multiple myeloma or light‐chain deposition disease a less likely diagnosis.
Typically, primary lymphoma of the bone produces increased uptake with bone scanning. However, because primary lymphoma of the bone is one of the least common primary skeletal malignancies and varies widely in appearance on imaging, confident diagnosis based on imaging alone usually is not possible.
Posttransplantation lymphoproliferative disorder (PTLD) refers to a syndrome that ranges from a self‐limited form of lymphoproliferation to an aggressive disseminated disease. Although the patient is at risk for PTLD, isolated bone involvement has only rarely been reported.
Primary lymphoma of the bone and PTLD are my leading diagnoses in this patient. At this point, I recommend a bone marrow biopsy and biopsy of an easily accessible representative bone lesion with special staining for Epstein‐Barr virus (EBV) (EBV‐encoded RNA [EBER] and latent membrane protein 1 [LMP1]). I expect this test to provide a definitive diagnosis. As 95% of PTLD cases are induced by infection with EBV, information regarding pretransplantation EBV status of the patient and the donor, current EBV status of the patient, and type and intensity of immunosuppression at the time of transplantation would be very helpful to determine their likelihood.
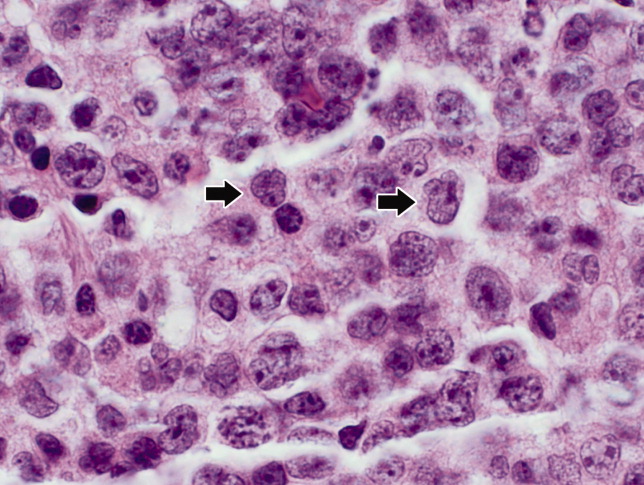
Seventy‐two hours after presentation, his serum calcium level normalized and most of his symptoms improved. Calcitonin was discontinued, and he was maintained on oral hydration. On hospital day number 5, he underwent CT‐guided bone biopsy of the L4 vertebral body, which showed large aggregates of atypical lymphoid cells (Figure 2). These cells were predominantly B‐cells interspersed with small reactive T‐cells. The cells did not express EBV LMP1 or EBER (Figure 3). On hospital day 7, he underwent a bone marrow biopsy, which revealed similar large atypical lymphoid cells that comprised the majority of marrow space (Figure 4). By immunohistochemistry, these cells brightly expressed the pan B cell marker, CD20, and coexpressed bcl‐2. EBER and LMP1 were also negative. A flow cytometry of the bone marrow demonstrated a lambda light chain restriction within the B lymphocytes.

The medical records indicated that the patient had positive pretransplantation EBV serologies. He received a regimen based on sirolimus, mycophenolate mofetil, and prednisone, and did not receive high doses of induction or maintenance immunosuppressive therapy.
The biopsy results establish a diagnosis of diffuse large B‐cell lymphoma of the bone. PTLD is unlikely given his positive pretransplantation EBV status, the late onset of his disease (6 years after transplantation), the isolated bone involvement, and the negative EBER and LMP1 tests.
The patient was discharged and was readmitted 1 week later for induction chemotherapy with etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone [EPOCH]Rituxan (rituximab). Over the next several months, he received 6 cycles of chemotherapy, his hypercalcemia resolved, and his back pain improved.
Commentary
Hypercalcemia is among the most common causes of nephrogenic diabetes insipidus in adults.1 A urinary concentrating defect usually becomes clinically apparent if the plasma calcium concentration is persistently above 11 mg/dL.1 This defect is generally reversible with correction of the hypercalcemia but may persist in patients in whom interstitial nephritis has induced permanent medullary damage. The mechanism by which the concentrating defect occurs is incompletely understood but may be related to impairments in sodium chloride reabsorption in the thick ascending limb and in the ability of antidiuretic hormone to increase water permeability in the collecting tubules.1
Although hypercalcemia in otherwise healthy outpatients is usually due to primary hyperparathyroidism, malignancy is more often responsible for hypercalcemia in hospitalized patients.2 While the signs and symptoms of hypercalcemia are similar regardless of the cause, several clinical features may help distinguish the etiology of hypercalcemia. For instance, the presence of tachycardia, warm skin, thinning of the hair, stare and lid lag, and widened pulse pressure points toward hypercalcemia related to hyperthyroidism. In addition, risk factors and comorbidities guide the diagnostic process. For example, low‐level hypercalcemia in an asymptomatic postmenopausal woman with a normal physical examination suggests primary hyperparathyroidism. In contrast, hypercalcemia in a transplant patient raises concern of malignancy including PTLDs.3, 4
PTLDs are uncommon causes of hypercalcemia but are among the most serious and potentially fatal complications of chronic immunosuppression in transplant recipients.5 They occur in 1.9% of patients after kidney transplantation. The lymphoproliferative disorders occurring after transplantation have different characteristics from those that occur in the general population. Non‐Hodgkin lymphoma accounts for 65% of lymphomas in the general population, compared to 93% in transplant recipients.5, 6 The pathogenesis of PTLD appears to be related to B cell proliferation induced by infection with EBV in the setting of chronic immunosuppression.6 Therefore, there is an increased frequency of PTLD among transplant recipients who are EBV seronegative at the time of operation. These patients, who have no preoperative immunity to EBV, usually acquire the infection from the donor. The level of immunosuppression (intensity and type) influences PTLD rates as well. The disease typically occurs within 12 months after transplantation and in two‐thirds of cases involves extranodal sites. Among these sites, the gastrointestinal tract is involved in about 26% of cases and central nervous system in about 27%. Isolated bone involvement is exceedingly rare.5, 6
Primary lymphoma of the bone is another rare cause of hypercalcemia and accounts for less than 5% of all primary bone tumors.7 The majority of cases are of the non‐Hodgkin's type, characterized as diffuse large B‐cell lymphomas, with peak occurrence in the sixth to seventh decades of life.8 The classic imaging findings of primary lymphoma of the bone are a solitary metadiaphyseal lesion with a layered periosteal reaction on plain radiographs, and corresponding surrounding soft‐tissue mass on MRI.9 Less commonly, primary lymphoma of the bone can be multifocal with diffuse osseous involvement and variable radiographic appearances, as in this case. Most series have reported that the long bones are affected most frequently (especially the femur), although a large series showed equal numbers of cases presenting in the long bones and the spine.712
In order to diagnose primary lymphoma of the bone, it is necessary to exclude nodal or disseminated disease by physical examination and imaging. As plain films are often normal, bone scan or MRI of clinically affected areas is necessary to establish disease extent.9 Distinguishing primary bone lymphomas (PLB) from other bone tumors is important because PLB has a better response to therapy and a better prognosis.10, 11
Randomized trials addressing treatment options for primary lymphoma of bone are not available. Historically, PLB was treated with radiotherapy alone with good local control. However, the rate of distant relapses was relatively high. Currently, chemotherapy with or without radiation therapy is preferred; 5‐year survival is approximately 70% after combined therapy.10, 11
In this case, symptomatic hypercalcemia, a history of transplantation, marked elevation of both LDH and B2M, and a normal PTH level all pointed toward the correct diagnosis of malignancy. Low or normal levels of vitamin D metabolites and PTH‐related protein occur in 20% of patients with hypercalcemia caused by malignancy.13, 14 Diffuse osteopenia on skeletal survey is a prominent feature of renal osteodystrophy or osteoporosis related to chronic corticosteroid use. However, in a patient with diffuse osteopenia and hypercalcemia, clinicians must consider multiple myeloma and other lymphoproliferative disorders; the absence of osteoblastic or osteolytic lesions and a normal alkaline phosphatase do not rule out these diagnoses. When the results of serum and urine protein electrophoresis exclude multiple myeloma, the next investigation should be a bone biopsy to exclude PLB, an uncommon cause of anemia, hypercalcemia, and osteopenic, painful bones.
Key Points for Hospitalists
-
Normal total alkaline phosphatase does not exclude primary or metastatic bone malignancy. While a normal level tends to predict a negative bone scan, further diagnostic tests are needed to exclude bone malignancy if high clinical suspicion exists.
-
The degree of hypercalcemia is useful diagnostically; values above 13 mg/dL are most often due to malignancy.
-
Hypercalcemia in transplant patients deserves special attention due to an increased risk of malignancy, including squamous cancers of the lips and skin, lymphoproliferative disorders, and bronchogenic carcinoma.
-
While rare, consider primary lymphoma of the bone in patients with hypercalcemia and bone pain, along with the more common diagnoses of multiple myeloma and metastatic bone disease.
The approach to clinical conundrums by an expert clinician is revealed through presentation of an actual patient's case in an approach typical of morning report. Similar to patient care, sequential pieces of information are provided to the clinician who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring the patient and the discussant.
- ,.Clinical Physiology of Acid‐Base and Electrolyte Disorders.5th ed.New York:McGraw‐Hill;2001:754–758.
- ,.Hypercalcemia: clinical manifestations, pathogenesis, diagnosis, and management. In: Favus MJ, ed.Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism.5th ed.Washington, DC:American Society for Bone and Mineral Research;2003:225–230.
- ,,, et al.Malignancy after renal transplantation: analysis of incidence and risk factors in 1700 patients followed during a 25‐year period.Transplant Proc.1997;29:831–833.
- ,.Malignancy‐associated hypercalcemia. In: DeGroot L, Jameson LJ, eds.Endocrinology.4th ed.Philadelphia, PA:Saunders;2001:1093–1100.
- ,.Diagnosis and management of posttransplant lymphoproliferative disorder in solid‐organ transplant recipients.Clin Infect Dis.2001;33(suppl 1):S38–S46.
- ,,, et al.Epstein‐Barr virus‐induced posttransplant lymphoproliferative disorders: ASTS/ASTP EBV‐PTLD Task Force and The Mayo Clinic Organized International Consensus Development Meeting.Transplantation.1999;68:1517–1525.
- ,,, et al.Primary bone lymphoma: a new and detailed characterization of 28 patients in a single‐institution study.Jpn J Clin Oncol.2007;37(3):216–223.
- ,,, et al.Diffuse large B‐cell lymphoma of bone. An analysis of differentiation‐associated antigens with clinical correlation.Am J Surg Pathol.2003;27:1269–1277.
- ,,,,,.Primary bone lymphoma: radiographic‐MR imaging correlation.Radiographics.2003;23:1371–1383.
- ,,, et al.Primary bone lymphoma in 24 patients treated between 1955 and 1999.Clin Orthop.2002;397:271–280.
- ,,, et al.A clinicopathological retrospective study of 131 patients with primary bone lymphoma: a population‐based study of successively treated cohorts from the British Columbia Cancer Agency.Ann Oncol.2007;18:129.
- ,,, et al.Malignant lymphoma of bone.Cancer.1986;58:2646–2655.
- .Hypercalcemia in malignant lymphoma and leukemia.Ann N Y Acad Sci.1974;230:240–246.
- .Incidence and prognostic significance of hypercalcemia in B‐cell non‐Hodgkin's lymphoma. [Letter]J Clin Pathol.2002;55:637–638.
A 71‐year‐old man presented to a hospital with a one week history of fatigue, polyuria, and polydipsia. He also reported pain in his back, hips, and ribs, in addition to frequent falls, intermittent confusion, constipation, and a weight loss of 10 pounds over the last 2 weeks. He denied cough, shortness of breath, chest pain, fever, night sweats, headache, and focal weakness.
Polyuria, which is often associated with polydipsia, can be arbitrarily defined as a urine output exceeding 3 L per day. After excluding osmotic diuresis due to uncontrolled diabetes mellitus, the 3 major causes of polyuria are primary polydipsia, central diabetes insipidus, and nephrogenic diabetes insipidus. Approximately 30% to 50% of cases of central diabetes insipidus are idiopathic; however, primary or secondary brain tumors or infiltrative diseases involving the hypothalamic‐pituitary region need to be considered in this 71‐year‐old man. The most common causes of nephrogenic diabetes insipidus in adults are chronic lithium ingestion, hypokalemia, and hypercalcemia. The patient describes symptoms that can result from severe hypercalcemia, including fatigue, confusion, constipation, polyuria, and polydipsia.
The patient's past medical history included long‐standing, insulin‐requiring type 2 diabetes with associated complications including coronary artery disease, transient ischemic attacks, proliferative retinopathy, peripheral diabetic neuropathy, and nephropathy. Seven years prior to presentation, he received a cadaveric renal transplant that was complicated by BK virus (polyomavirus) nephropathy and secondary hyperparathyroidism. Three years after his transplant surgery, he developed squamous cell carcinoma of the skin, which was treated with local surgical resection. Two years after that, he developed stage I laryngeal cancer of the glottis and received laser surgery, and since then he had been considered disease‐free. He also had a history of hypertension, hypercholesterolemia, osteoporosis, and depression. His medications included aspirin, amlodipine, metoprolol succinate, valsartan, furosemide, simvastatin, insulin, prednisone, sirolimus, and sulfamethoxazole/trimethoprim. He was a married psychiatrist. He denied tobacco use and reported occasional alcohol use.
The prolonged immunosuppressive therapy that is required following organ transplantation carries a markedly increased risk of the subsequent development of malignant tumors, including cancers of the lips and skin, lymphoproliferative disorders, and bronchogenic carcinoma. Primary brain lymphoma resulting in central diabetes insipidus would be unlikely in the absence of headache or focal weakness. An increased risk of lung cancer occurs in recipients of heart and lung transplants, and to a much lesser degree, recipients of kidney transplants. However, metastatic lung cancer is less likely in the absence of respiratory symptoms and smoking history (present in approximately 90% of all lung cancers). Nephrogenic diabetes insipidus, in its mild form, is relatively common in elderly patients with acute or chronic renal insufficiency because of a reduction in maximum urinary concentrating ability. On the other hand, this alone does not explain his remaining symptoms. The instinctive diagnosis in this case is tertiary hyperparathyroidism due to progression of untreated secondary hyperparathyroidism. This causes hypercalcemia, nephrogenic diabetes insipidus, and significant bone pain related to renal osteodystrophy.
On physical exam, the patient appeared chronically ill, but was in no acute distress. He weighed 197.6 pounds and his height was 70.5 inches. He was afebrile with a blood pressure of 146/82 mm Hg, a heart rate of 76 beats per minute, a respiratory rate of 12 breaths per minute, and an oxygen saturation of 97% while breathing room air. He had no generalized lymphadenopathy. Thyroid examination was unremarkable. Examination of the lungs, heart, abdomen, and lower extremities was normal. The rectal examination revealed no masses or prostate nodules; a test for fecal occult blood was negative. He had loss of sensation to light touch and vibration in the feet with absent Achilles deep tendon reflexes. He had a poorly healing surgical wound on his forehead at the site of his prior skin cancer, but no rash or other lesions. There was no joint swelling or erythema. There were tender points over the cervical, thoracic, and lumbar spine; on multiple ribs; and on the pelvic rims.
Perhaps of greatest importance is the lack of lymphadenopathy, organomegaly, or other findings suggestive of diffuse lymphoproliferative disease. His multifocal bone tenderness is concerning for renal osteodystrophy, multiple myeloma, or primary or metastatic bone disease. Cancers in men that metastasize to the bone usually originate from the prostate, lung, kidney, or thyroid gland. In any case, his physical examination did not reveal an enlarged, asymmetric, or nodular prostate or thyroid gland. I recommend a chest film to rule out primary lung malignancy and a basic laboratory evaluation to narrow down the differential diagnosis.
A complete blood count showed a normocytic anemia with a hemoglobin of 8.7 g/dL and a hematocrit of 25%. Other laboratory tests revealed the following values: sodium, 139 mmol/L; potassium, 4.1 mmol/L; blood urea nitrogen, 70 mg/dL; creatinine, 3.5 mg/dL (most recent value 2 months ago was 1.9 mg/dL); total calcium, 13.2 mg/dL (normal range, 8.5‐10.5 mg/dL); phosphate, 5.3 mg/dL; magnesium, 2.5 mg/dL; total bilirubin, 0.5 mg/dL; alkaline phosphatase, 130 U/L; aspartate aminotransferase, 28 U/L; alanine aminotransferase, 19 U/L; albumin, 3.5 g/dL; and lactate dehydrogenase (LDH), 1258 IU/L (normal range, 105‐333 IU/L). A chest radiograph was normal.
The most important laboratory findings are severe hypercalcemia, acute on chronic renal failure, and anemia. Hypercalcemia most commonly results from malignancy or hyperparathyroidism. Less frequently, hypercalcemia may result from sarcoidosis, vitamin D intoxication, or hyperthyroidism. The degree of hypercalcemia is useful diagnostically as hyperparathyroidism commonly results in mild hypercalcemia (serum calcium concentration often below 11 mg/dL). Values above 13 mg/dL are unusual in hyperparathyroidism and are most often due to malignancy. Malignancy is often evident clinically by the time it causes hypercalcemia, and patients with hypercalcemia of malignancy are more often symptomatic than those with hyperparathyroidism. Additionally, localized bone pain and weight loss do not result from hypercalcemia itself and their presence also raises concern for malignancy.
Nonmelanoma skin cancer is the most common cancer occurring after transplantation but does not cause hypercalcemia. Squamous cancers of the head and neck can rarely cause hypercalcemia due to secretion of parathyroid hormone‐related peptide; however, his early‐stage laryngeal cancer and the expected high likelihood of cure argue against this possibility. Osteolytic metastases account for approximately 20% of cases of hypercalcemia of malignancy (Table 1). Prostate cancer rarely results in hypercalcemia since bone metastases are predominantly osteoblastic, whereas metastatic non‐small‐cell lung cancer, thyroid cancer, and kidney cancer more commonly cause hypercalcemia due to osteolytic bone lesions. The total alkaline phosphatase has been traditionally used to assess the osteoblastic component of bone remodeling. Its normal level tends to predict a negative bone scan and supports the likelihood of lytic lesions. Posttransplantation lymphoproliferative disorders, which include a wide range of syndromes, can rarely result in hypercalcemia. I am also worried about the possibility of multiple myeloma as he has the classic triad of hypercalcemia, bone pain, and subacute kidney injury.
|
| Osteolytic metastases |
| Breast cancer |
| Multiple myeloma |
| Lymphoma |
| Leukemia |
| Humoral hypercalcemia (PTH‐related protein) |
| Squamous cell carcinomas |
| Renal carcinomas |
| Bladder carcinoma |
| Breast cancer |
| Ovarian carcinoma |
| Leukemia |
| Lymphoma |
| 1,25‐Dihydroxyvitamin D secretion |
| Lymphoma |
| Ovarian dysgerminomas |
| Ectopic PTH secretion (rare) |
| Ovarian carcinoma |
| Lung carcinomas |
| Neuroectodermal tumor |
| Thyroid papillary carcinoma |
| Rhabdomyosarcoma |
| Pancreatic cancer |
The first purpose of the laboratory evaluation is to differentiate parathyroid hormone (PTH)‐mediated hypercalcemia (primary and tertiary hyperparathyroidism) from non‐PTH‐mediated hypercalcemia (primarily malignancy, hyperthyroidism, vitamin D intoxication, and granulomatous disease). The production of vitamin D metabolites, PTH‐related protein, or hypercalcemia from osteolysis in these latter cases results in suppressed PTH levels.
In severe elevations of calcium, the initial goals of treatment are directed toward fluid resuscitation with normal saline and, unless contraindicated, the immediate institution of bisphosphonate therapy. A loop diuretic such as furosemide is often used, but a recent review concluded that there is little evidence to support its use in this setting.
The patient was admitted and treated with intravenous saline and furosemide. Additional laboratory evaluation revealed normal levels of prostate‐specific antigen and thyroid‐stimulating hormone. PTH was 44 pg/mL (the most recent value was 906 pg/mL eight years ago; normal range, 15‐65 pg/mL) and beta‐2 microglobulin (B2M) was 8 mg/L (normal range, 0.8‐2.2 mg/L).
The normal PTH level makes tertiary hyperparathyroidism unlikely and points toward non‐PTH‐related hypercalcemia. An elevated B2M level may occur in patients with chronic graft rejection, renal tubular dysfunction, dialysis‐related amyloidosis, multiple myeloma, or lymphoma. LDH is often elevated in patients with multiple myeloma and lymphoma, but this is not a specific finding. The next laboratory test would be measurement of PTH‐related protein and vitamin D metabolites, as these tests can differentiate between the causes of non‐PTH‐mediated hypercalcemia.
Serum concentrations of the vitamin D metabolites, 25‐hydroxyvitamin D (calcidiol) and 1,25‐dihydroxyvitamin D (calcitriol), were low‐normal. PTH‐related protein was not detected.
The marked elevation of serum LDH and B2M, the relatively suppressed PTH level, combined with undetectable PTH‐related protein suggest multiple myeloma or lymphoma as the likely cause of the patient's clinical presentation. The combination of hypercalcemia and multifocal bone pain makes multiple myeloma the leading diagnosis as hypercalcemia is uncommon in patients with lymphoma, especially at the time of initial clinical presentation.
I would proceed with serum and urine protein electrophoresis (SPEP and UPEP, respectively) and a skeletal survey. If these tests do not confirm the diagnosis of multiple myeloma, I would order a noncontrast computed tomography (CT) of the chest and abdomen and a magnetic resonance imaging (MRI) of the spine. In addition, I would like to monitor his response to the intravenous saline and furosemide.
Forty‐eight hours after presentation, repeat serum calcium and creatinine levels were 11.3 mg/dL and 2.9 mg/dL, respectively. He received salmon calcitonin 4 U/kg every 12 hours. Pamidronate was avoided because of his kidney disease. His confusion resolved. He received intravenous morphine intermittently to alleviate his bone pain.
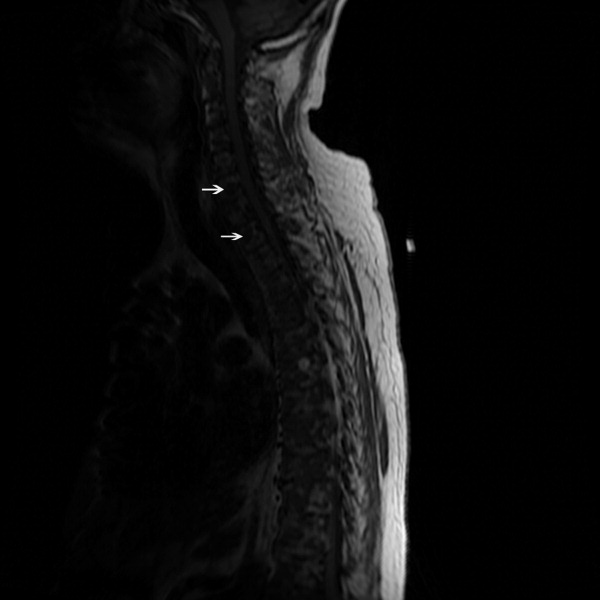
The SPEP revealed a monoclonal immunoglobulin G (IgG) lambda (light chain) spike representing roughly 3% (200 mg/dL) of total protein. His serum Ig levels were normal. The UPEP was negative for monoclonal immunoglobulin and Bence‐Jones protein. The skeletal survey revealed marked osteopenia, and the bone scan was normal. An MRI of the spine showed multiple round lesions in the cervical, thoracic, and lumbar spine (Figure 1). A CT of the chest showed similar bone lesions in the ribs and pelvis. A CT of the abdomen and chest did not suggest any primary malignancy nor did it show thoracic or abdominal lymphadenopathy.

The lack of lymphadenopathy, splenomegaly, or a visceral mass by CT imaging and physical examination, along with the normal PSA level, exclude most common forms of non‐Hodgkin lymphoma and bone metastasis from solid tumors. In multiple myeloma, cytokines secreted by plasma cells suppress osteoblast activity; therefore, while discrete lytic bone lesions are apparent on skeletal survey, the bone scan is typically normal. The absence of lytic lesions, normal serum immunoglobulin levels, and unremarkable UPEP make multiple myeloma or light‐chain deposition disease a less likely diagnosis.
Typically, primary lymphoma of the bone produces increased uptake with bone scanning. However, because primary lymphoma of the bone is one of the least common primary skeletal malignancies and varies widely in appearance on imaging, confident diagnosis based on imaging alone usually is not possible.
Posttransplantation lymphoproliferative disorder (PTLD) refers to a syndrome that ranges from a self‐limited form of lymphoproliferation to an aggressive disseminated disease. Although the patient is at risk for PTLD, isolated bone involvement has only rarely been reported.
Primary lymphoma of the bone and PTLD are my leading diagnoses in this patient. At this point, I recommend a bone marrow biopsy and biopsy of an easily accessible representative bone lesion with special staining for Epstein‐Barr virus (EBV) (EBV‐encoded RNA [EBER] and latent membrane protein 1 [LMP1]). I expect this test to provide a definitive diagnosis. As 95% of PTLD cases are induced by infection with EBV, information regarding pretransplantation EBV status of the patient and the donor, current EBV status of the patient, and type and intensity of immunosuppression at the time of transplantation would be very helpful to determine their likelihood.
Seventy‐two hours after presentation, his serum calcium level normalized and most of his symptoms improved. Calcitonin was discontinued, and he was maintained on oral hydration. On hospital day number 5, he underwent CT‐guided bone biopsy of the L4 vertebral body, which showed large aggregates of atypical lymphoid cells (Figure 2). These cells were predominantly B‐cells interspersed with small reactive T‐cells. The cells did not express EBV LMP1 or EBER (Figure 3). On hospital day 7, he underwent a bone marrow biopsy, which revealed similar large atypical lymphoid cells that comprised the majority of marrow space (Figure 4). By immunohistochemistry, these cells brightly expressed the pan B cell marker, CD20, and coexpressed bcl‐2. EBER and LMP1 were also negative. A flow cytometry of the bone marrow demonstrated a lambda light chain restriction within the B lymphocytes.



The medical records indicated that the patient had positive pretransplantation EBV serologies. He received a regimen based on sirolimus, mycophenolate mofetil, and prednisone, and did not receive high doses of induction or maintenance immunosuppressive therapy.
The biopsy results establish a diagnosis of diffuse large B‐cell lymphoma of the bone. PTLD is unlikely given his positive pretransplantation EBV status, the late onset of his disease (6 years after transplantation), the isolated bone involvement, and the negative EBER and LMP1 tests.
The patient was discharged and was readmitted 1 week later for induction chemotherapy with etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone [EPOCH]Rituxan (rituximab). Over the next several months, he received 6 cycles of chemotherapy, his hypercalcemia resolved, and his back pain improved.
Commentary
Hypercalcemia is among the most common causes of nephrogenic diabetes insipidus in adults.1 A urinary concentrating defect usually becomes clinically apparent if the plasma calcium concentration is persistently above 11 mg/dL.1 This defect is generally reversible with correction of the hypercalcemia but may persist in patients in whom interstitial nephritis has induced permanent medullary damage. The mechanism by which the concentrating defect occurs is incompletely understood but may be related to impairments in sodium chloride reabsorption in the thick ascending limb and in the ability of antidiuretic hormone to increase water permeability in the collecting tubules.1
Although hypercalcemia in otherwise healthy outpatients is usually due to primary hyperparathyroidism, malignancy is more often responsible for hypercalcemia in hospitalized patients.2 While the signs and symptoms of hypercalcemia are similar regardless of the cause, several clinical features may help distinguish the etiology of hypercalcemia. For instance, the presence of tachycardia, warm skin, thinning of the hair, stare and lid lag, and widened pulse pressure points toward hypercalcemia related to hyperthyroidism. In addition, risk factors and comorbidities guide the diagnostic process. For example, low‐level hypercalcemia in an asymptomatic postmenopausal woman with a normal physical examination suggests primary hyperparathyroidism. In contrast, hypercalcemia in a transplant patient raises concern of malignancy including PTLDs.3, 4
PTLDs are uncommon causes of hypercalcemia but are among the most serious and potentially fatal complications of chronic immunosuppression in transplant recipients.5 They occur in 1.9% of patients after kidney transplantation. The lymphoproliferative disorders occurring after transplantation have different characteristics from those that occur in the general population. Non‐Hodgkin lymphoma accounts for 65% of lymphomas in the general population, compared to 93% in transplant recipients.5, 6 The pathogenesis of PTLD appears to be related to B cell proliferation induced by infection with EBV in the setting of chronic immunosuppression.6 Therefore, there is an increased frequency of PTLD among transplant recipients who are EBV seronegative at the time of operation. These patients, who have no preoperative immunity to EBV, usually acquire the infection from the donor. The level of immunosuppression (intensity and type) influences PTLD rates as well. The disease typically occurs within 12 months after transplantation and in two‐thirds of cases involves extranodal sites. Among these sites, the gastrointestinal tract is involved in about 26% of cases and central nervous system in about 27%. Isolated bone involvement is exceedingly rare.5, 6
Primary lymphoma of the bone is another rare cause of hypercalcemia and accounts for less than 5% of all primary bone tumors.7 The majority of cases are of the non‐Hodgkin's type, characterized as diffuse large B‐cell lymphomas, with peak occurrence in the sixth to seventh decades of life.8 The classic imaging findings of primary lymphoma of the bone are a solitary metadiaphyseal lesion with a layered periosteal reaction on plain radiographs, and corresponding surrounding soft‐tissue mass on MRI.9 Less commonly, primary lymphoma of the bone can be multifocal with diffuse osseous involvement and variable radiographic appearances, as in this case. Most series have reported that the long bones are affected most frequently (especially the femur), although a large series showed equal numbers of cases presenting in the long bones and the spine.712
In order to diagnose primary lymphoma of the bone, it is necessary to exclude nodal or disseminated disease by physical examination and imaging. As plain films are often normal, bone scan or MRI of clinically affected areas is necessary to establish disease extent.9 Distinguishing primary bone lymphomas (PLB) from other bone tumors is important because PLB has a better response to therapy and a better prognosis.10, 11
Randomized trials addressing treatment options for primary lymphoma of bone are not available. Historically, PLB was treated with radiotherapy alone with good local control. However, the rate of distant relapses was relatively high. Currently, chemotherapy with or without radiation therapy is preferred; 5‐year survival is approximately 70% after combined therapy.10, 11
In this case, symptomatic hypercalcemia, a history of transplantation, marked elevation of both LDH and B2M, and a normal PTH level all pointed toward the correct diagnosis of malignancy. Low or normal levels of vitamin D metabolites and PTH‐related protein occur in 20% of patients with hypercalcemia caused by malignancy.13, 14 Diffuse osteopenia on skeletal survey is a prominent feature of renal osteodystrophy or osteoporosis related to chronic corticosteroid use. However, in a patient with diffuse osteopenia and hypercalcemia, clinicians must consider multiple myeloma and other lymphoproliferative disorders; the absence of osteoblastic or osteolytic lesions and a normal alkaline phosphatase do not rule out these diagnoses. When the results of serum and urine protein electrophoresis exclude multiple myeloma, the next investigation should be a bone biopsy to exclude PLB, an uncommon cause of anemia, hypercalcemia, and osteopenic, painful bones.
Key Points for Hospitalists
-
Normal total alkaline phosphatase does not exclude primary or metastatic bone malignancy. While a normal level tends to predict a negative bone scan, further diagnostic tests are needed to exclude bone malignancy if high clinical suspicion exists.
-
The degree of hypercalcemia is useful diagnostically; values above 13 mg/dL are most often due to malignancy.
-
Hypercalcemia in transplant patients deserves special attention due to an increased risk of malignancy, including squamous cancers of the lips and skin, lymphoproliferative disorders, and bronchogenic carcinoma.
-
While rare, consider primary lymphoma of the bone in patients with hypercalcemia and bone pain, along with the more common diagnoses of multiple myeloma and metastatic bone disease.
The approach to clinical conundrums by an expert clinician is revealed through presentation of an actual patient's case in an approach typical of morning report. Similar to patient care, sequential pieces of information are provided to the clinician who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring the patient and the discussant.
A 71‐year‐old man presented to a hospital with a one week history of fatigue, polyuria, and polydipsia. He also reported pain in his back, hips, and ribs, in addition to frequent falls, intermittent confusion, constipation, and a weight loss of 10 pounds over the last 2 weeks. He denied cough, shortness of breath, chest pain, fever, night sweats, headache, and focal weakness.
Polyuria, which is often associated with polydipsia, can be arbitrarily defined as a urine output exceeding 3 L per day. After excluding osmotic diuresis due to uncontrolled diabetes mellitus, the 3 major causes of polyuria are primary polydipsia, central diabetes insipidus, and nephrogenic diabetes insipidus. Approximately 30% to 50% of cases of central diabetes insipidus are idiopathic; however, primary or secondary brain tumors or infiltrative diseases involving the hypothalamic‐pituitary region need to be considered in this 71‐year‐old man. The most common causes of nephrogenic diabetes insipidus in adults are chronic lithium ingestion, hypokalemia, and hypercalcemia. The patient describes symptoms that can result from severe hypercalcemia, including fatigue, confusion, constipation, polyuria, and polydipsia.
The patient's past medical history included long‐standing, insulin‐requiring type 2 diabetes with associated complications including coronary artery disease, transient ischemic attacks, proliferative retinopathy, peripheral diabetic neuropathy, and nephropathy. Seven years prior to presentation, he received a cadaveric renal transplant that was complicated by BK virus (polyomavirus) nephropathy and secondary hyperparathyroidism. Three years after his transplant surgery, he developed squamous cell carcinoma of the skin, which was treated with local surgical resection. Two years after that, he developed stage I laryngeal cancer of the glottis and received laser surgery, and since then he had been considered disease‐free. He also had a history of hypertension, hypercholesterolemia, osteoporosis, and depression. His medications included aspirin, amlodipine, metoprolol succinate, valsartan, furosemide, simvastatin, insulin, prednisone, sirolimus, and sulfamethoxazole/trimethoprim. He was a married psychiatrist. He denied tobacco use and reported occasional alcohol use.
The prolonged immunosuppressive therapy that is required following organ transplantation carries a markedly increased risk of the subsequent development of malignant tumors, including cancers of the lips and skin, lymphoproliferative disorders, and bronchogenic carcinoma. Primary brain lymphoma resulting in central diabetes insipidus would be unlikely in the absence of headache or focal weakness. An increased risk of lung cancer occurs in recipients of heart and lung transplants, and to a much lesser degree, recipients of kidney transplants. However, metastatic lung cancer is less likely in the absence of respiratory symptoms and smoking history (present in approximately 90% of all lung cancers). Nephrogenic diabetes insipidus, in its mild form, is relatively common in elderly patients with acute or chronic renal insufficiency because of a reduction in maximum urinary concentrating ability. On the other hand, this alone does not explain his remaining symptoms. The instinctive diagnosis in this case is tertiary hyperparathyroidism due to progression of untreated secondary hyperparathyroidism. This causes hypercalcemia, nephrogenic diabetes insipidus, and significant bone pain related to renal osteodystrophy.
On physical exam, the patient appeared chronically ill, but was in no acute distress. He weighed 197.6 pounds and his height was 70.5 inches. He was afebrile with a blood pressure of 146/82 mm Hg, a heart rate of 76 beats per minute, a respiratory rate of 12 breaths per minute, and an oxygen saturation of 97% while breathing room air. He had no generalized lymphadenopathy. Thyroid examination was unremarkable. Examination of the lungs, heart, abdomen, and lower extremities was normal. The rectal examination revealed no masses or prostate nodules; a test for fecal occult blood was negative. He had loss of sensation to light touch and vibration in the feet with absent Achilles deep tendon reflexes. He had a poorly healing surgical wound on his forehead at the site of his prior skin cancer, but no rash or other lesions. There was no joint swelling or erythema. There were tender points over the cervical, thoracic, and lumbar spine; on multiple ribs; and on the pelvic rims.
Perhaps of greatest importance is the lack of lymphadenopathy, organomegaly, or other findings suggestive of diffuse lymphoproliferative disease. His multifocal bone tenderness is concerning for renal osteodystrophy, multiple myeloma, or primary or metastatic bone disease. Cancers in men that metastasize to the bone usually originate from the prostate, lung, kidney, or thyroid gland. In any case, his physical examination did not reveal an enlarged, asymmetric, or nodular prostate or thyroid gland. I recommend a chest film to rule out primary lung malignancy and a basic laboratory evaluation to narrow down the differential diagnosis.
A complete blood count showed a normocytic anemia with a hemoglobin of 8.7 g/dL and a hematocrit of 25%. Other laboratory tests revealed the following values: sodium, 139 mmol/L; potassium, 4.1 mmol/L; blood urea nitrogen, 70 mg/dL; creatinine, 3.5 mg/dL (most recent value 2 months ago was 1.9 mg/dL); total calcium, 13.2 mg/dL (normal range, 8.5‐10.5 mg/dL); phosphate, 5.3 mg/dL; magnesium, 2.5 mg/dL; total bilirubin, 0.5 mg/dL; alkaline phosphatase, 130 U/L; aspartate aminotransferase, 28 U/L; alanine aminotransferase, 19 U/L; albumin, 3.5 g/dL; and lactate dehydrogenase (LDH), 1258 IU/L (normal range, 105‐333 IU/L). A chest radiograph was normal.
The most important laboratory findings are severe hypercalcemia, acute on chronic renal failure, and anemia. Hypercalcemia most commonly results from malignancy or hyperparathyroidism. Less frequently, hypercalcemia may result from sarcoidosis, vitamin D intoxication, or hyperthyroidism. The degree of hypercalcemia is useful diagnostically as hyperparathyroidism commonly results in mild hypercalcemia (serum calcium concentration often below 11 mg/dL). Values above 13 mg/dL are unusual in hyperparathyroidism and are most often due to malignancy. Malignancy is often evident clinically by the time it causes hypercalcemia, and patients with hypercalcemia of malignancy are more often symptomatic than those with hyperparathyroidism. Additionally, localized bone pain and weight loss do not result from hypercalcemia itself and their presence also raises concern for malignancy.
Nonmelanoma skin cancer is the most common cancer occurring after transplantation but does not cause hypercalcemia. Squamous cancers of the head and neck can rarely cause hypercalcemia due to secretion of parathyroid hormone‐related peptide; however, his early‐stage laryngeal cancer and the expected high likelihood of cure argue against this possibility. Osteolytic metastases account for approximately 20% of cases of hypercalcemia of malignancy (Table 1). Prostate cancer rarely results in hypercalcemia since bone metastases are predominantly osteoblastic, whereas metastatic non‐small‐cell lung cancer, thyroid cancer, and kidney cancer more commonly cause hypercalcemia due to osteolytic bone lesions. The total alkaline phosphatase has been traditionally used to assess the osteoblastic component of bone remodeling. Its normal level tends to predict a negative bone scan and supports the likelihood of lytic lesions. Posttransplantation lymphoproliferative disorders, which include a wide range of syndromes, can rarely result in hypercalcemia. I am also worried about the possibility of multiple myeloma as he has the classic triad of hypercalcemia, bone pain, and subacute kidney injury.
|
| Osteolytic metastases |
| Breast cancer |
| Multiple myeloma |
| Lymphoma |
| Leukemia |
| Humoral hypercalcemia (PTH‐related protein) |
| Squamous cell carcinomas |
| Renal carcinomas |
| Bladder carcinoma |
| Breast cancer |
| Ovarian carcinoma |
| Leukemia |
| Lymphoma |
| 1,25‐Dihydroxyvitamin D secretion |
| Lymphoma |
| Ovarian dysgerminomas |
| Ectopic PTH secretion (rare) |
| Ovarian carcinoma |
| Lung carcinomas |
| Neuroectodermal tumor |
| Thyroid papillary carcinoma |
| Rhabdomyosarcoma |
| Pancreatic cancer |
The first purpose of the laboratory evaluation is to differentiate parathyroid hormone (PTH)‐mediated hypercalcemia (primary and tertiary hyperparathyroidism) from non‐PTH‐mediated hypercalcemia (primarily malignancy, hyperthyroidism, vitamin D intoxication, and granulomatous disease). The production of vitamin D metabolites, PTH‐related protein, or hypercalcemia from osteolysis in these latter cases results in suppressed PTH levels.
In severe elevations of calcium, the initial goals of treatment are directed toward fluid resuscitation with normal saline and, unless contraindicated, the immediate institution of bisphosphonate therapy. A loop diuretic such as furosemide is often used, but a recent review concluded that there is little evidence to support its use in this setting.
The patient was admitted and treated with intravenous saline and furosemide. Additional laboratory evaluation revealed normal levels of prostate‐specific antigen and thyroid‐stimulating hormone. PTH was 44 pg/mL (the most recent value was 906 pg/mL eight years ago; normal range, 15‐65 pg/mL) and beta‐2 microglobulin (B2M) was 8 mg/L (normal range, 0.8‐2.2 mg/L).
The normal PTH level makes tertiary hyperparathyroidism unlikely and points toward non‐PTH‐related hypercalcemia. An elevated B2M level may occur in patients with chronic graft rejection, renal tubular dysfunction, dialysis‐related amyloidosis, multiple myeloma, or lymphoma. LDH is often elevated in patients with multiple myeloma and lymphoma, but this is not a specific finding. The next laboratory test would be measurement of PTH‐related protein and vitamin D metabolites, as these tests can differentiate between the causes of non‐PTH‐mediated hypercalcemia.
Serum concentrations of the vitamin D metabolites, 25‐hydroxyvitamin D (calcidiol) and 1,25‐dihydroxyvitamin D (calcitriol), were low‐normal. PTH‐related protein was not detected.
The marked elevation of serum LDH and B2M, the relatively suppressed PTH level, combined with undetectable PTH‐related protein suggest multiple myeloma or lymphoma as the likely cause of the patient's clinical presentation. The combination of hypercalcemia and multifocal bone pain makes multiple myeloma the leading diagnosis as hypercalcemia is uncommon in patients with lymphoma, especially at the time of initial clinical presentation.
I would proceed with serum and urine protein electrophoresis (SPEP and UPEP, respectively) and a skeletal survey. If these tests do not confirm the diagnosis of multiple myeloma, I would order a noncontrast computed tomography (CT) of the chest and abdomen and a magnetic resonance imaging (MRI) of the spine. In addition, I would like to monitor his response to the intravenous saline and furosemide.
Forty‐eight hours after presentation, repeat serum calcium and creatinine levels were 11.3 mg/dL and 2.9 mg/dL, respectively. He received salmon calcitonin 4 U/kg every 12 hours. Pamidronate was avoided because of his kidney disease. His confusion resolved. He received intravenous morphine intermittently to alleviate his bone pain.
The SPEP revealed a monoclonal immunoglobulin G (IgG) lambda (light chain) spike representing roughly 3% (200 mg/dL) of total protein. His serum Ig levels were normal. The UPEP was negative for monoclonal immunoglobulin and Bence‐Jones protein. The skeletal survey revealed marked osteopenia, and the bone scan was normal. An MRI of the spine showed multiple round lesions in the cervical, thoracic, and lumbar spine (Figure 1). A CT of the chest showed similar bone lesions in the ribs and pelvis. A CT of the abdomen and chest did not suggest any primary malignancy nor did it show thoracic or abdominal lymphadenopathy.

The lack of lymphadenopathy, splenomegaly, or a visceral mass by CT imaging and physical examination, along with the normal PSA level, exclude most common forms of non‐Hodgkin lymphoma and bone metastasis from solid tumors. In multiple myeloma, cytokines secreted by plasma cells suppress osteoblast activity; therefore, while discrete lytic bone lesions are apparent on skeletal survey, the bone scan is typically normal. The absence of lytic lesions, normal serum immunoglobulin levels, and unremarkable UPEP make multiple myeloma or light‐chain deposition disease a less likely diagnosis.
Typically, primary lymphoma of the bone produces increased uptake with bone scanning. However, because primary lymphoma of the bone is one of the least common primary skeletal malignancies and varies widely in appearance on imaging, confident diagnosis based on imaging alone usually is not possible.
Posttransplantation lymphoproliferative disorder (PTLD) refers to a syndrome that ranges from a self‐limited form of lymphoproliferation to an aggressive disseminated disease. Although the patient is at risk for PTLD, isolated bone involvement has only rarely been reported.
Primary lymphoma of the bone and PTLD are my leading diagnoses in this patient. At this point, I recommend a bone marrow biopsy and biopsy of an easily accessible representative bone lesion with special staining for Epstein‐Barr virus (EBV) (EBV‐encoded RNA [EBER] and latent membrane protein 1 [LMP1]). I expect this test to provide a definitive diagnosis. As 95% of PTLD cases are induced by infection with EBV, information regarding pretransplantation EBV status of the patient and the donor, current EBV status of the patient, and type and intensity of immunosuppression at the time of transplantation would be very helpful to determine their likelihood.
Seventy‐two hours after presentation, his serum calcium level normalized and most of his symptoms improved. Calcitonin was discontinued, and he was maintained on oral hydration. On hospital day number 5, he underwent CT‐guided bone biopsy of the L4 vertebral body, which showed large aggregates of atypical lymphoid cells (Figure 2). These cells were predominantly B‐cells interspersed with small reactive T‐cells. The cells did not express EBV LMP1 or EBER (Figure 3). On hospital day 7, he underwent a bone marrow biopsy, which revealed similar large atypical lymphoid cells that comprised the majority of marrow space (Figure 4). By immunohistochemistry, these cells brightly expressed the pan B cell marker, CD20, and coexpressed bcl‐2. EBER and LMP1 were also negative. A flow cytometry of the bone marrow demonstrated a lambda light chain restriction within the B lymphocytes.



The medical records indicated that the patient had positive pretransplantation EBV serologies. He received a regimen based on sirolimus, mycophenolate mofetil, and prednisone, and did not receive high doses of induction or maintenance immunosuppressive therapy.
The biopsy results establish a diagnosis of diffuse large B‐cell lymphoma of the bone. PTLD is unlikely given his positive pretransplantation EBV status, the late onset of his disease (6 years after transplantation), the isolated bone involvement, and the negative EBER and LMP1 tests.
The patient was discharged and was readmitted 1 week later for induction chemotherapy with etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone [EPOCH]Rituxan (rituximab). Over the next several months, he received 6 cycles of chemotherapy, his hypercalcemia resolved, and his back pain improved.
Commentary
Hypercalcemia is among the most common causes of nephrogenic diabetes insipidus in adults.1 A urinary concentrating defect usually becomes clinically apparent if the plasma calcium concentration is persistently above 11 mg/dL.1 This defect is generally reversible with correction of the hypercalcemia but may persist in patients in whom interstitial nephritis has induced permanent medullary damage. The mechanism by which the concentrating defect occurs is incompletely understood but may be related to impairments in sodium chloride reabsorption in the thick ascending limb and in the ability of antidiuretic hormone to increase water permeability in the collecting tubules.1
Although hypercalcemia in otherwise healthy outpatients is usually due to primary hyperparathyroidism, malignancy is more often responsible for hypercalcemia in hospitalized patients.2 While the signs and symptoms of hypercalcemia are similar regardless of the cause, several clinical features may help distinguish the etiology of hypercalcemia. For instance, the presence of tachycardia, warm skin, thinning of the hair, stare and lid lag, and widened pulse pressure points toward hypercalcemia related to hyperthyroidism. In addition, risk factors and comorbidities guide the diagnostic process. For example, low‐level hypercalcemia in an asymptomatic postmenopausal woman with a normal physical examination suggests primary hyperparathyroidism. In contrast, hypercalcemia in a transplant patient raises concern of malignancy including PTLDs.3, 4
PTLDs are uncommon causes of hypercalcemia but are among the most serious and potentially fatal complications of chronic immunosuppression in transplant recipients.5 They occur in 1.9% of patients after kidney transplantation. The lymphoproliferative disorders occurring after transplantation have different characteristics from those that occur in the general population. Non‐Hodgkin lymphoma accounts for 65% of lymphomas in the general population, compared to 93% in transplant recipients.5, 6 The pathogenesis of PTLD appears to be related to B cell proliferation induced by infection with EBV in the setting of chronic immunosuppression.6 Therefore, there is an increased frequency of PTLD among transplant recipients who are EBV seronegative at the time of operation. These patients, who have no preoperative immunity to EBV, usually acquire the infection from the donor. The level of immunosuppression (intensity and type) influences PTLD rates as well. The disease typically occurs within 12 months after transplantation and in two‐thirds of cases involves extranodal sites. Among these sites, the gastrointestinal tract is involved in about 26% of cases and central nervous system in about 27%. Isolated bone involvement is exceedingly rare.5, 6
Primary lymphoma of the bone is another rare cause of hypercalcemia and accounts for less than 5% of all primary bone tumors.7 The majority of cases are of the non‐Hodgkin's type, characterized as diffuse large B‐cell lymphomas, with peak occurrence in the sixth to seventh decades of life.8 The classic imaging findings of primary lymphoma of the bone are a solitary metadiaphyseal lesion with a layered periosteal reaction on plain radiographs, and corresponding surrounding soft‐tissue mass on MRI.9 Less commonly, primary lymphoma of the bone can be multifocal with diffuse osseous involvement and variable radiographic appearances, as in this case. Most series have reported that the long bones are affected most frequently (especially the femur), although a large series showed equal numbers of cases presenting in the long bones and the spine.712
In order to diagnose primary lymphoma of the bone, it is necessary to exclude nodal or disseminated disease by physical examination and imaging. As plain films are often normal, bone scan or MRI of clinically affected areas is necessary to establish disease extent.9 Distinguishing primary bone lymphomas (PLB) from other bone tumors is important because PLB has a better response to therapy and a better prognosis.10, 11
Randomized trials addressing treatment options for primary lymphoma of bone are not available. Historically, PLB was treated with radiotherapy alone with good local control. However, the rate of distant relapses was relatively high. Currently, chemotherapy with or without radiation therapy is preferred; 5‐year survival is approximately 70% after combined therapy.10, 11
In this case, symptomatic hypercalcemia, a history of transplantation, marked elevation of both LDH and B2M, and a normal PTH level all pointed toward the correct diagnosis of malignancy. Low or normal levels of vitamin D metabolites and PTH‐related protein occur in 20% of patients with hypercalcemia caused by malignancy.13, 14 Diffuse osteopenia on skeletal survey is a prominent feature of renal osteodystrophy or osteoporosis related to chronic corticosteroid use. However, in a patient with diffuse osteopenia and hypercalcemia, clinicians must consider multiple myeloma and other lymphoproliferative disorders; the absence of osteoblastic or osteolytic lesions and a normal alkaline phosphatase do not rule out these diagnoses. When the results of serum and urine protein electrophoresis exclude multiple myeloma, the next investigation should be a bone biopsy to exclude PLB, an uncommon cause of anemia, hypercalcemia, and osteopenic, painful bones.
Key Points for Hospitalists
-
Normal total alkaline phosphatase does not exclude primary or metastatic bone malignancy. While a normal level tends to predict a negative bone scan, further diagnostic tests are needed to exclude bone malignancy if high clinical suspicion exists.
-
The degree of hypercalcemia is useful diagnostically; values above 13 mg/dL are most often due to malignancy.
-
Hypercalcemia in transplant patients deserves special attention due to an increased risk of malignancy, including squamous cancers of the lips and skin, lymphoproliferative disorders, and bronchogenic carcinoma.
-
While rare, consider primary lymphoma of the bone in patients with hypercalcemia and bone pain, along with the more common diagnoses of multiple myeloma and metastatic bone disease.
The approach to clinical conundrums by an expert clinician is revealed through presentation of an actual patient's case in an approach typical of morning report. Similar to patient care, sequential pieces of information are provided to the clinician who is unfamiliar with the case. The focus is on the thought processes of both the clinical team caring the patient and the discussant.
- ,.Clinical Physiology of Acid‐Base and Electrolyte Disorders.5th ed.New York:McGraw‐Hill;2001:754–758.
- ,.Hypercalcemia: clinical manifestations, pathogenesis, diagnosis, and management. In: Favus MJ, ed.Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism.5th ed.Washington, DC:American Society for Bone and Mineral Research;2003:225–230.
- ,,, et al.Malignancy after renal transplantation: analysis of incidence and risk factors in 1700 patients followed during a 25‐year period.Transplant Proc.1997;29:831–833.
- ,.Malignancy‐associated hypercalcemia. In: DeGroot L, Jameson LJ, eds.Endocrinology.4th ed.Philadelphia, PA:Saunders;2001:1093–1100.
- ,.Diagnosis and management of posttransplant lymphoproliferative disorder in solid‐organ transplant recipients.Clin Infect Dis.2001;33(suppl 1):S38–S46.
- ,,, et al.Epstein‐Barr virus‐induced posttransplant lymphoproliferative disorders: ASTS/ASTP EBV‐PTLD Task Force and The Mayo Clinic Organized International Consensus Development Meeting.Transplantation.1999;68:1517–1525.
- ,,, et al.Primary bone lymphoma: a new and detailed characterization of 28 patients in a single‐institution study.Jpn J Clin Oncol.2007;37(3):216–223.
- ,,, et al.Diffuse large B‐cell lymphoma of bone. An analysis of differentiation‐associated antigens with clinical correlation.Am J Surg Pathol.2003;27:1269–1277.
- ,,,,,.Primary bone lymphoma: radiographic‐MR imaging correlation.Radiographics.2003;23:1371–1383.
- ,,, et al.Primary bone lymphoma in 24 patients treated between 1955 and 1999.Clin Orthop.2002;397:271–280.
- ,,, et al.A clinicopathological retrospective study of 131 patients with primary bone lymphoma: a population‐based study of successively treated cohorts from the British Columbia Cancer Agency.Ann Oncol.2007;18:129.
- ,,, et al.Malignant lymphoma of bone.Cancer.1986;58:2646–2655.
- .Hypercalcemia in malignant lymphoma and leukemia.Ann N Y Acad Sci.1974;230:240–246.
- .Incidence and prognostic significance of hypercalcemia in B‐cell non‐Hodgkin's lymphoma. [Letter]J Clin Pathol.2002;55:637–638.
- ,.Clinical Physiology of Acid‐Base and Electrolyte Disorders.5th ed.New York:McGraw‐Hill;2001:754–758.
- ,.Hypercalcemia: clinical manifestations, pathogenesis, diagnosis, and management. In: Favus MJ, ed.Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism.5th ed.Washington, DC:American Society for Bone and Mineral Research;2003:225–230.
- ,,, et al.Malignancy after renal transplantation: analysis of incidence and risk factors in 1700 patients followed during a 25‐year period.Transplant Proc.1997;29:831–833.
- ,.Malignancy‐associated hypercalcemia. In: DeGroot L, Jameson LJ, eds.Endocrinology.4th ed.Philadelphia, PA:Saunders;2001:1093–1100.
- ,.Diagnosis and management of posttransplant lymphoproliferative disorder in solid‐organ transplant recipients.Clin Infect Dis.2001;33(suppl 1):S38–S46.
- ,,, et al.Epstein‐Barr virus‐induced posttransplant lymphoproliferative disorders: ASTS/ASTP EBV‐PTLD Task Force and The Mayo Clinic Organized International Consensus Development Meeting.Transplantation.1999;68:1517–1525.
- ,,, et al.Primary bone lymphoma: a new and detailed characterization of 28 patients in a single‐institution study.Jpn J Clin Oncol.2007;37(3):216–223.
- ,,, et al.Diffuse large B‐cell lymphoma of bone. An analysis of differentiation‐associated antigens with clinical correlation.Am J Surg Pathol.2003;27:1269–1277.
- ,,,,,.Primary bone lymphoma: radiographic‐MR imaging correlation.Radiographics.2003;23:1371–1383.
- ,,, et al.Primary bone lymphoma in 24 patients treated between 1955 and 1999.Clin Orthop.2002;397:271–280.
- ,,, et al.A clinicopathological retrospective study of 131 patients with primary bone lymphoma: a population‐based study of successively treated cohorts from the British Columbia Cancer Agency.Ann Oncol.2007;18:129.
- ,,, et al.Malignant lymphoma of bone.Cancer.1986;58:2646–2655.
- .Hypercalcemia in malignant lymphoma and leukemia.Ann N Y Acad Sci.1974;230:240–246.
- .Incidence and prognostic significance of hypercalcemia in B‐cell non‐Hodgkin's lymphoma. [Letter]J Clin Pathol.2002;55:637–638.
Small Bowel Obstruction by Gallstone Ileus
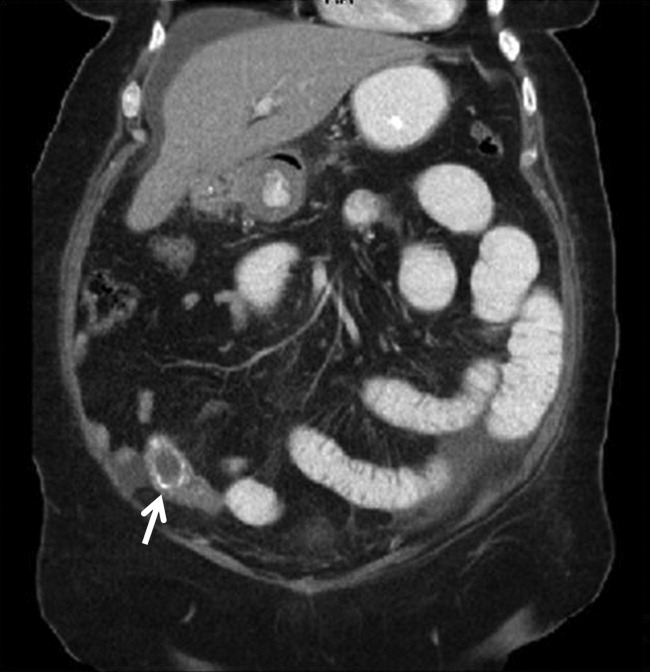
A 67‐year‐old female presented with nausea and nonspecific abdominal pain occurring intermittently for 2 weeks. Physical examination revealed no abdominal guarding or rigidity and was significant only for slightly hypoactive bowel sounds. Routine laboratory evaluation including liver panel was unremarkable. X‐ray of the abdomen showed air fluid levels, and a diagnosis of small bowel obstruction/emleus secondary to adhesions from a previous hysterectomy was established. Conservative management with nasogastric suction, intravenous (IV) fluids and nil‐by‐mouth was continued for 3 days but no clinical improvement was seen. Due to continued abdominal pain, an ultrasound of the abdomen was done, which revealed cholelithiasis and chronic cholecystitis. At this point the patient was transferred to our facility and a computerized axial tomography (CAT) scan of the abdomen was done, which revealed a gallstone in the ileum (Figure 1) and the diagnosis of gallstone ileus was made. Air in the biliary tract (pneumobilia) was noted, suggesting the presence of a fistula between the gallbladder and the gastrointestinal (GI) tract. The fistula itself could not be clearly visualized. The patient was immediately taken to surgery, where small bowel exploration was done. A large gallstone completely obstructing the terminal ileum was removed by enterotomy. The gallbladder was adherent to the stomach and a cholecystogastric fistula with a gallstone coming out on the gastric end was noted. This gallstone was removed and the cholecystogastric fistula was repaired. Cholecystectomy was done at the same time. The patient recovered without any complications and has been doing well.
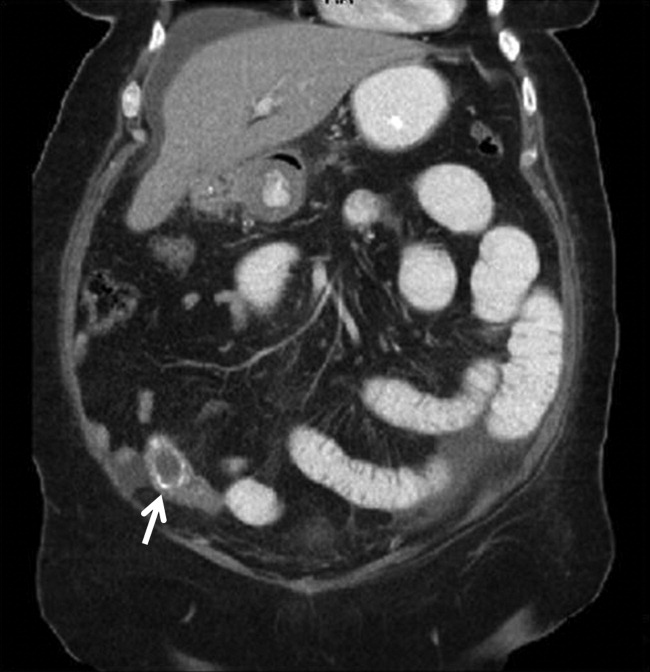
Discussion
Gallstone ileus accounts for approximately 1% to 2% cases of small bowel obstruction, most of which are in the elderly population. It is much more common in females as compared to males.1 Although the morbidity and mortality associated with gallstone ileus has reduced in comparison to the previous decade, probably due to the more effective usage of imaging techniques like ultrasound, CAT scan, magnetic resonance imaging (MRI), and endoscopy in the diagnostic evaluation of abdominal pain, the numbers still range between 12% to 25%.1, 2 Early diagnosis plays a major role in reducing the mortality in these patients. Therefore, it is important to consider gallstone ileus in the differential diagnosis of an elderly patient presenting with bowel obstruction.
Gallstone ileus is usually associated with a biliaryenteric fistula that allows the passage of a gallstone from the gallbladder into the bowel. This gallstone gets impacted in the gastrointestinal lumen and causes mechanical bowel obstruction. The term gallstone ileus is a misnomer as the gallstone causes actual obstruction rather than just ileus. Considering the high prevalence of cholelithiasis, it must be realized that formation of cholecystoenteric fistula is relatively rare (about 2% in patients with cholecystitis). It is proposed that pericholecystic inflammation after an episode of cholecystitis results in the formation of adhesions between the biliary and gastrointestinal tracts. The gallstone causes pressure necrosis of the biliary wall and then erodes through it to form a fistulous communication with the adherent enteric system. Among these fistulous communications, cholecystoduodenal fistulas are the most common (60%) while cholecystocolonic and cholecystogastric fistulas are also seen. After the biliary stone has eroded through the enteric wall, air within the intestinal tract now freely enters the biliary system, leading to an appearance of pneumobilia on imaging studies. Mirizzi syndrome, described as common hepatic duct obstruction caused by an extrinsic compression from an impacted stone in the cystic duct, is often associated with gallstone ileus.3
Once the gallstone enters the enteric tract through the fistula and traverses down the gastrointestinal tract, it causes intermittent abdominal pain, nausea, and vomiting. The symptoms may be spread over multiple days as the gallstone causes transient obstruction with its impaction and disimpaction. These obstructing gallstones that cause luminal obstruction are usually larger than 2 cm. Majority of them will traverse the duodenum, jejunum, and small intestine, and finally get lodged in the terminal ileum (60%), the narrowest part of the small intestine. Other sites where obstruction may occur include jejunum (16%), duodenum, stomach, and colon.1 Bouveret's syndrome is a variant of gallstone ileus wherein the gallstone impacts in the pylorus of the stomach or the duodenum, leading to gastric outlet obstruction.3
The diagnosis of gallstone ileus is not always straightforward and requires a high index of suspicion. It is classically described by the Rigler's triadpneumobilia, partial or complete bowel obstruction, and ectopic gallstone,4 although often all 3 signs are not elicited. Most patients need open enterolithotomy for relief of bowel obstruction. Cholecystectomy may or may not be performed. The literature is controversial regarding the best approach of surgical management. Two surgical approaches are equally accepted: (1) a 1‐stage approach, which includes enterolithotomy, cholecystectomy, and fistula repair at the same time; and (2) the other option is a 2‐stage approach in which enterolithotomy is performed first and biliary surgery is performed later, if indicated. The patient's age, comorbidities, and the associated surgical risks are often used to decide between the 2 surgical approaches.
Gallstone ileus should be considered in the differential for the etiology of small bowel obstruction, especially in an elderly female known to have cholelithiasis.
- ,.Gallstone ileus (a review of 1001 reported cases).Am Surg.1994;60:441–446.
- ,,, et al.Improving the outcome in gallstone ileus.Am J Surg.1986;151:572–576.
- ,,.The relationship of Mirizzi syndrome and cholecystoenteric fistula: validation of a modified classification.World J Surg.2008;32(10):2237–2243.
- ,,.Gallstone obstruction: pathogenesis and roentgen manifestations.JAMA.1941;117:1753–1759.
A 67‐year‐old female presented with nausea and nonspecific abdominal pain occurring intermittently for 2 weeks. Physical examination revealed no abdominal guarding or rigidity and was significant only for slightly hypoactive bowel sounds. Routine laboratory evaluation including liver panel was unremarkable. X‐ray of the abdomen showed air fluid levels, and a diagnosis of small bowel obstruction/emleus secondary to adhesions from a previous hysterectomy was established. Conservative management with nasogastric suction, intravenous (IV) fluids and nil‐by‐mouth was continued for 3 days but no clinical improvement was seen. Due to continued abdominal pain, an ultrasound of the abdomen was done, which revealed cholelithiasis and chronic cholecystitis. At this point the patient was transferred to our facility and a computerized axial tomography (CAT) scan of the abdomen was done, which revealed a gallstone in the ileum (Figure 1) and the diagnosis of gallstone ileus was made. Air in the biliary tract (pneumobilia) was noted, suggesting the presence of a fistula between the gallbladder and the gastrointestinal (GI) tract. The fistula itself could not be clearly visualized. The patient was immediately taken to surgery, where small bowel exploration was done. A large gallstone completely obstructing the terminal ileum was removed by enterotomy. The gallbladder was adherent to the stomach and a cholecystogastric fistula with a gallstone coming out on the gastric end was noted. This gallstone was removed and the cholecystogastric fistula was repaired. Cholecystectomy was done at the same time. The patient recovered without any complications and has been doing well.

Discussion
Gallstone ileus accounts for approximately 1% to 2% cases of small bowel obstruction, most of which are in the elderly population. It is much more common in females as compared to males.1 Although the morbidity and mortality associated with gallstone ileus has reduced in comparison to the previous decade, probably due to the more effective usage of imaging techniques like ultrasound, CAT scan, magnetic resonance imaging (MRI), and endoscopy in the diagnostic evaluation of abdominal pain, the numbers still range between 12% to 25%.1, 2 Early diagnosis plays a major role in reducing the mortality in these patients. Therefore, it is important to consider gallstone ileus in the differential diagnosis of an elderly patient presenting with bowel obstruction.
Gallstone ileus is usually associated with a biliaryenteric fistula that allows the passage of a gallstone from the gallbladder into the bowel. This gallstone gets impacted in the gastrointestinal lumen and causes mechanical bowel obstruction. The term gallstone ileus is a misnomer as the gallstone causes actual obstruction rather than just ileus. Considering the high prevalence of cholelithiasis, it must be realized that formation of cholecystoenteric fistula is relatively rare (about 2% in patients with cholecystitis). It is proposed that pericholecystic inflammation after an episode of cholecystitis results in the formation of adhesions between the biliary and gastrointestinal tracts. The gallstone causes pressure necrosis of the biliary wall and then erodes through it to form a fistulous communication with the adherent enteric system. Among these fistulous communications, cholecystoduodenal fistulas are the most common (60%) while cholecystocolonic and cholecystogastric fistulas are also seen. After the biliary stone has eroded through the enteric wall, air within the intestinal tract now freely enters the biliary system, leading to an appearance of pneumobilia on imaging studies. Mirizzi syndrome, described as common hepatic duct obstruction caused by an extrinsic compression from an impacted stone in the cystic duct, is often associated with gallstone ileus.3
Once the gallstone enters the enteric tract through the fistula and traverses down the gastrointestinal tract, it causes intermittent abdominal pain, nausea, and vomiting. The symptoms may be spread over multiple days as the gallstone causes transient obstruction with its impaction and disimpaction. These obstructing gallstones that cause luminal obstruction are usually larger than 2 cm. Majority of them will traverse the duodenum, jejunum, and small intestine, and finally get lodged in the terminal ileum (60%), the narrowest part of the small intestine. Other sites where obstruction may occur include jejunum (16%), duodenum, stomach, and colon.1 Bouveret's syndrome is a variant of gallstone ileus wherein the gallstone impacts in the pylorus of the stomach or the duodenum, leading to gastric outlet obstruction.3
The diagnosis of gallstone ileus is not always straightforward and requires a high index of suspicion. It is classically described by the Rigler's triadpneumobilia, partial or complete bowel obstruction, and ectopic gallstone,4 although often all 3 signs are not elicited. Most patients need open enterolithotomy for relief of bowel obstruction. Cholecystectomy may or may not be performed. The literature is controversial regarding the best approach of surgical management. Two surgical approaches are equally accepted: (1) a 1‐stage approach, which includes enterolithotomy, cholecystectomy, and fistula repair at the same time; and (2) the other option is a 2‐stage approach in which enterolithotomy is performed first and biliary surgery is performed later, if indicated. The patient's age, comorbidities, and the associated surgical risks are often used to decide between the 2 surgical approaches.
Gallstone ileus should be considered in the differential for the etiology of small bowel obstruction, especially in an elderly female known to have cholelithiasis.
A 67‐year‐old female presented with nausea and nonspecific abdominal pain occurring intermittently for 2 weeks. Physical examination revealed no abdominal guarding or rigidity and was significant only for slightly hypoactive bowel sounds. Routine laboratory evaluation including liver panel was unremarkable. X‐ray of the abdomen showed air fluid levels, and a diagnosis of small bowel obstruction/emleus secondary to adhesions from a previous hysterectomy was established. Conservative management with nasogastric suction, intravenous (IV) fluids and nil‐by‐mouth was continued for 3 days but no clinical improvement was seen. Due to continued abdominal pain, an ultrasound of the abdomen was done, which revealed cholelithiasis and chronic cholecystitis. At this point the patient was transferred to our facility and a computerized axial tomography (CAT) scan of the abdomen was done, which revealed a gallstone in the ileum (Figure 1) and the diagnosis of gallstone ileus was made. Air in the biliary tract (pneumobilia) was noted, suggesting the presence of a fistula between the gallbladder and the gastrointestinal (GI) tract. The fistula itself could not be clearly visualized. The patient was immediately taken to surgery, where small bowel exploration was done. A large gallstone completely obstructing the terminal ileum was removed by enterotomy. The gallbladder was adherent to the stomach and a cholecystogastric fistula with a gallstone coming out on the gastric end was noted. This gallstone was removed and the cholecystogastric fistula was repaired. Cholecystectomy was done at the same time. The patient recovered without any complications and has been doing well.

Discussion
Gallstone ileus accounts for approximately 1% to 2% cases of small bowel obstruction, most of which are in the elderly population. It is much more common in females as compared to males.1 Although the morbidity and mortality associated with gallstone ileus has reduced in comparison to the previous decade, probably due to the more effective usage of imaging techniques like ultrasound, CAT scan, magnetic resonance imaging (MRI), and endoscopy in the diagnostic evaluation of abdominal pain, the numbers still range between 12% to 25%.1, 2 Early diagnosis plays a major role in reducing the mortality in these patients. Therefore, it is important to consider gallstone ileus in the differential diagnosis of an elderly patient presenting with bowel obstruction.
Gallstone ileus is usually associated with a biliaryenteric fistula that allows the passage of a gallstone from the gallbladder into the bowel. This gallstone gets impacted in the gastrointestinal lumen and causes mechanical bowel obstruction. The term gallstone ileus is a misnomer as the gallstone causes actual obstruction rather than just ileus. Considering the high prevalence of cholelithiasis, it must be realized that formation of cholecystoenteric fistula is relatively rare (about 2% in patients with cholecystitis). It is proposed that pericholecystic inflammation after an episode of cholecystitis results in the formation of adhesions between the biliary and gastrointestinal tracts. The gallstone causes pressure necrosis of the biliary wall and then erodes through it to form a fistulous communication with the adherent enteric system. Among these fistulous communications, cholecystoduodenal fistulas are the most common (60%) while cholecystocolonic and cholecystogastric fistulas are also seen. After the biliary stone has eroded through the enteric wall, air within the intestinal tract now freely enters the biliary system, leading to an appearance of pneumobilia on imaging studies. Mirizzi syndrome, described as common hepatic duct obstruction caused by an extrinsic compression from an impacted stone in the cystic duct, is often associated with gallstone ileus.3
Once the gallstone enters the enteric tract through the fistula and traverses down the gastrointestinal tract, it causes intermittent abdominal pain, nausea, and vomiting. The symptoms may be spread over multiple days as the gallstone causes transient obstruction with its impaction and disimpaction. These obstructing gallstones that cause luminal obstruction are usually larger than 2 cm. Majority of them will traverse the duodenum, jejunum, and small intestine, and finally get lodged in the terminal ileum (60%), the narrowest part of the small intestine. Other sites where obstruction may occur include jejunum (16%), duodenum, stomach, and colon.1 Bouveret's syndrome is a variant of gallstone ileus wherein the gallstone impacts in the pylorus of the stomach or the duodenum, leading to gastric outlet obstruction.3
The diagnosis of gallstone ileus is not always straightforward and requires a high index of suspicion. It is classically described by the Rigler's triadpneumobilia, partial or complete bowel obstruction, and ectopic gallstone,4 although often all 3 signs are not elicited. Most patients need open enterolithotomy for relief of bowel obstruction. Cholecystectomy may or may not be performed. The literature is controversial regarding the best approach of surgical management. Two surgical approaches are equally accepted: (1) a 1‐stage approach, which includes enterolithotomy, cholecystectomy, and fistula repair at the same time; and (2) the other option is a 2‐stage approach in which enterolithotomy is performed first and biliary surgery is performed later, if indicated. The patient's age, comorbidities, and the associated surgical risks are often used to decide between the 2 surgical approaches.
Gallstone ileus should be considered in the differential for the etiology of small bowel obstruction, especially in an elderly female known to have cholelithiasis.
- ,.Gallstone ileus (a review of 1001 reported cases).Am Surg.1994;60:441–446.
- ,,, et al.Improving the outcome in gallstone ileus.Am J Surg.1986;151:572–576.
- ,,.The relationship of Mirizzi syndrome and cholecystoenteric fistula: validation of a modified classification.World J Surg.2008;32(10):2237–2243.
- ,,.Gallstone obstruction: pathogenesis and roentgen manifestations.JAMA.1941;117:1753–1759.
- ,.Gallstone ileus (a review of 1001 reported cases).Am Surg.1994;60:441–446.
- ,,, et al.Improving the outcome in gallstone ileus.Am J Surg.1986;151:572–576.
- ,,.The relationship of Mirizzi syndrome and cholecystoenteric fistula: validation of a modified classification.World J Surg.2008;32(10):2237–2243.
- ,,.Gallstone obstruction: pathogenesis and roentgen manifestations.JAMA.1941;117:1753–1759.
Management of Ischemic Stroke: Part 2
Inpatient stroke management includes many elements of care, at least as important as the initial portion of the patient's stay, as reviewed in part 1 of this article. The extent of further diagnostic evaluation varies widely depending on apparent risk factors on presentation. Likewise, further therapy, both inpatient and secondary prevention is based on identification of stroke mechanism. Hospitalists are uniquely positioned to have a tremendous impact on both stroke care and the prevention of recurrent disease.
Case Presentation
A 76‐year‐old right‐handed male with a history of hyperlipidemia and myocardial infarction was found at 7 AM with right‐sided paralysis and poor responsiveness on the morning of admission. Upon arrival to the emergency department (ED), with symptoms of partial aphasia, right hemiplegia, and left gaze preference, there was a high suspicion for a left middle cerebral artery (MCA) stroke. Unfortunately, he was excluded from receiving intravenous (IV) tissue plasminogen activator (tPA) or any other acute interventions as the last time he was known to be neurologically intact was the prior evening, which is taken to be the time of onset. Antiplatelet therapy was continued, and the patient admitted for further workup.
Inpatient Care
When an acute ischemic stroke patient is admitted to the hospital, he or she should be placed on a standardized acute stroke protocol (also known as (a.k.a.) a care map, order set, clinical pathway)commonly created by a hospitalist/neurologist and a multidisciplinary team and admitted to a stroke unit. A stroke unit can take many forms, either as a physically separate unit in hospitals with sufficient volume or a floor where a lower volume of stroke patients are always admitted. Multidisciplinary care providers in the stroke unit have special training in stroke, and strong evidence from randomized trials shows that patients cared for in these units have significantly decreased mortality with improved functional outcomes.1 Essentials of the stroke protocol or order set include cardiac telemetry, maintaining euthermia and euglycemia, closely following blood pressure and neurologic status, actively avoiding complications, initiation of secondary prevention treatment, early involvement of rehabilitation services, and patient education.
Euthermia may be assisted by administering scheduled Tylenol to the patient for the first 48 hours, but is not strictly evidence‐based.2 Though euthermia and euglycemia have not been shown to improve outcomes in acute stroke, studies have shown that hyperthermia and hyperglycemia are associated with worsened outcomes for patients with acute strokes.35
Blood Pressure Management
Normally, cerebral vascular autoregulation leads to stable cerebral blood flow over a range of systemic blood pressures. In the setting of an acute stroke, the ability to autoregulate is diminished or absent in regions of and surrounding an acute ischemic stroke; as the area becomes ischemic, autoregulation opens the local vasculature maximally in an effort to drawn in as much blood as possible. Maximally dilated arterioles are perfused in direct correlation with systemic blood pressure, thus any drop in the systemic blood pressure leads to direct decreases in blood flow specifically in the area of ischemia; if there is a penumbra of marginally perfused tissue, such systemic blood pressure drops risk extending the area of fatal ischemia (increasing the size of the ischemic stroke).68 Thus in the acute period of an ischemic stroke, the American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for the Early Management of Adults With Ischemic Stroke (referred to herein as the Guidelines)10 suggest avoid treatment unless systolic blood pressures are >220 or diastolic pressures >105, and review the evidence to support this recommendation (p. 16711672). Those patients who receive tPA have a more stringent blood pressure threshold given their risk of intracranial hemorrhage; systolic blood pressures are accepted up to 180 prior to recommending treatment.
Higher‐quality Inpatient Stroke Care and Harmonized Performance Measures
Beginning in January 2008, a set of 10 performance measures (Table 1) for inpatient acute stroke care have been agreed upon (harmonized) by 3 major stakeholders including the Joint Commission, the ASA's Get with the GuidelinesStroke quality improvement program, and the Center for Disease Control and Prevention's (CDC's) Paul Coverdell Acute stroke registries. These performance measures were selected to help avoid complications (deep vein thrombosis [DVT], aspiration pneumonia), encourage appropriately aggressive care (tPA administration), optimize secondary prevention (antithrombotics, cholesterol lowering, smoking cessation, education), and facilitate functional recovery (early rehabilitation). All 10 measures are appropriate for consideration in every ischemic stroke patient, and 5 are appropriate for the hemorrhagic stroke types.
| Performance measure* | Definition* |
|---|---|
| |
| 1. DVT prophylaxis | Patients who are nonambulatory should start receiving DVT prophylaxis by end of hospital day 2 (can be either compression devices or any low‐dose heparin) |
| 2. Discharged on antithrombotic therapy | Antiplatelet agent(s) or warfarin anticoagulation |
| 3. Patients with atrial fibrillation receiving anticoagulation therapy | A proven approach to secondary prevention in such patients; practice at Harborview varies time of warfarin initiation based on infarct size with larger infarcts waiting up to 2 weeks before initiating warfarin (the best randomized trial showed no benefit for full‐dose low‐molecular‐weight heparin over aspirin in the first 2 weeks)50 |
| 4. Thrombolytic therapy administered | In ischemic stroke patients who arrive at the hospital within 120 minutes (2 hours) of time last known well, for whom IV tPA was initiated at this hospital within 180 minutes (3 hours) of time last known well, and who qualify under strict criteria |
| 5. Antithrombotic therapy by end of hospital day 2 | Usually just antiplatelet agents, a minimal standard of care for ischemic stroke patients; should be started as early as possible, usually in ER |
| 6. Discharged on statin medication | If LDL >100, or not measured or if on a statin drug prior to admission; to reduce risk of subsequent ischemic stroke |
| 7. Dysphagia screening | Prior to any PO food, fluids or medications; to reduce the chances of aspiration pneumonia |
| 8. Stroke education | Including for families if patient unable to participate, must include personal risk factors for stroke, warning signs for stroke, activation of emergency medical system, need for follow‐up after discharge, and medications prescribed |
| 9. Smoking cessation/advice/counseling | For any patient who has smoked in the last year |
| 10. Assessed for rehabilitation | Or received therapy services; to facilitate progress to an optimal function outcome |
Further Workup
After the ischemic stroke patient has had their computed tomography (CT) scan, possibly a computed tomography angiography (CTA), been admitted to the stroke unit, started on an antithrombotic medication, and had their blood pressure appropriately treated, attention then turns to defining the pathophysiology related to the stroke and starting an optimal regimen for secondary prevention. Imaging of the cerebral vasculature including both extracranial and intracranial large vessels is a vital first step in understanding the cause of ischemic stroke. There are multiple potential modalities (magnetic resonance angiography [MRA], CTA, and duplex/transcranial Doppler), the choice of which depends on local availability and expertise as well as the specific clinical situation. Magnetic resonance imaging (MRI) of the brain for all ischemic stroke patients is standard of care at most stroke centers; per the Guidelines, MRI is better at distinguishing acute, small cortical, small deep, and posterior fossa infarcts; at distinguishing acute from chronic ischemia; and at identifying subclinical satellite ischemic lesions that provide information on stroke mechanism (p. 1668). New techniques including magnetic resonance (MR) and CT perfusion scanning can show the ischemic region in the acute setting and may one day help select patients for specific therapies, but are not yet widely available nor have they been shown to alter outcomes.
An electrocardiogram is indicated for all stroke patients, as is admission to a cardiac telemetry bed for at least 24 hours to document any arrhythmias, the most common being atrial fibrillation (Guidelines, p. 1666, 1673). An echocardiographic study (ECHO) of the heart with bubble study should be performed in most cases (although which cases may specifically benefit is unclear) to identify a cardioembolic source for the stroke, such as low cardiac ejection fraction, atrial septal aneurysm, patent foramen ovale (PFO), or a cardiac thrombus. The bubble study increases the sensitivity of detecting a PFO, which could serve as a gateway for venous embolization to the cerebral arteries. Assuming a large PFO is discovered, other studies such as lower extremity Doppler may be warranted to investigate other potential sources of thrombi (ie, DVT).
Regarding laboratory testing, fasting lipids should be checked as hyperlipidemia is a common modifiable risk factor for ischemic stroke. The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial included ischemic stroke patients that had low‐density lipoprotein (LDL) cholesterol between 100 mg/dL and 190 mg/dL and randomized them to receive atorvastatin 80 mg/day vs. placebo. Results showed a 16% relative risk reduction in recurrent stroke; however, there was a small increased risk of intracranial hemorrhage.9 As shown in Table 1, use of a statin on discharge is now a national performance measure for ischemic stroke.
Dissection is a common cause of stroke in young patients without traditional risk factors. Other serologies, such as hypercoagulable studies, may be warranted in patients with no other risk factors for strokes, paradoxical embolus, or of young age (eg, 45 years and under). The arterial hypercoagulable panel consists of antiphospholipid antibody panel, homocysteine levels, lupus anticoagulant levels, and prothrombin time/partial thromboplastin time (PT/PTT). The venous hypercoagulable panel consists of the laboratory values checked, with the arterial hypercoagulable and activated protein C (APC) resistance, Factor VIII activity, Factor II DNA, Factor V DNA if the APC resistance is positive, antithrombin III activity, and activity of proteins C and S. If a patient is found to have a hypercoagulable state, long‐term therapy often involves careful consideration of the choice of antiplatelet therapy vs. anticoagulation with warfarin.10
Initiating Secondary Prevention
Upon admission, the clinician faces a variety of treatment choices for secondary stroke prevention. The proper choice depends on the results of the workup and the presumptive pathophysiology.
Noncardioembolic/Atherothrombotic/Lacunar
The Antithrombotic Trialists' Collaboration meta‐analysis found that patients with a prior stroke or transient ischemic attack (TIA) had a highly significant decrease in the rate of subsequent vascular events (over about 3 years) on antiplatelet therapy (17.8% vs. 21.4%, P 0.0001) and were unable to find a significant difference between low‐dose and high‐dose aspirin for secondary prevention.11 Thus, it is reasonable to place an acute stroke patient naive to antithrombotic therapy on 81 mg of aspirin or 325 mg for long‐term prevention (325 mg is specifically recommended in the acute setting). Several studies such as the WARSS and ESPRIT trials have shown antiplatelet agents to be at least as effective as anticoagulation in noncardioembolic ischemic strokes.12, 13 Guidelines from Europe, the American College of Chest Physicians, and the AHA/ASA all state it is acceptable to choose either aspirin monotherapy, aspirin/extended release dipyridamole combination therapy, or clopidogrel monotherapy as first‐line agents for long‐term secondary prevention in noncardioembolic ischemic stroke.1416 There is no clear evidence that patients who suffer an ischemic stroke while on aspirin will derive additional benefit from increasing the aspirin dose. The newer guidelines go on to recommend aspirin/extended release dipyridamole (ER‐DP) combination therapy or clopidogrel monotherapy over aspirin monotherapy, the former with a stronger level of recommendation based on the results of 2 randomized trials. These recommendations were all published without knowledge of the results of the Prevention Regimen For Effectively Avoiding Second Strokes (PRoFESS) study, which directly compared aspirin/extended release dipyridamole combination therapy to clopidogrel monotherapy for long‐term secondary prevention. The rate of first recurrent stroke was not significantly different between the 2 therapies (9.0% ER‐DP plus aspirin, 8.8% clopidogrel; hazard ratio [HR], 1.01; 95% confidence interval [CI], 0.921.11). Other outcomes also showed few differences, although there were more major hemorrhagic events in the ER‐DP plus aspirin group (4.1% vs. 3.6%; HR, 1.15; 95% CI, 1.001.32; P = 0.06).17
The ASA Stroke Prevention Guideline from 2006 states, with continued relevance, The selection of an antiplatelet agent should be individualized on the basis of patient risk factor profiles, tolerance, and other clinical characteristics.10 Of note, both the Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance (CHARISMA) and Management of ATherothrombosis with Clopidogrel in High‐risk patients with recent TIA or ischemic stroke (MATCH) trials found a significant increased risk for hemorrhage complications with long‐term use of the aspirin and clopidogrel combination,18, 19 and the 2008 update to the ASA Stroke prevention guidelines state that the addition of aspirin to clopidogrel increases the risk of hemorrhage. Combination therapy of aspirin and clopidogrel is not routinely recommended for ischemic stroke or TIA patients unless they have a specific indication for this therapy (i.e., coronary stent or acute coronary syndrome).15
Atrial Fibrillation
Though our case patient did not have atrial fibrillation, this condition does deserve mention. About 15% to 20% of ischemic stroke patients have atrial fibrillation. The overall risk for stroke in patients with atrial fibrillation is about 5% per year; however, patients who have a history of stroke increase their risk factors for subsequent strokes to about 12% per year. In most cases, anticoagulation has proven to be the superior agent for primary and secondary stroke prevention with warfarin reducing the risk by 67% compared to aspirin, which only reduces the risk of stroke by 20%. A meta‐analysis from 2002 showed that patients who had a prior stroke or TIA decrease their risk of subsequent strokes to 4%/year on oral anticoagulation therapy, resulting in an 8% absolute risk reduction. Patients on aspirin therapy only decrease their risk to 10%/year, or a 2% reduction in stroke events.20 Unless there is a strong contraindication (eg, bleeding diathesis, history of life threatening gastrointestinal [GI] bleeding, history of fall with subdural hematoma, etc.), virtually all ischemic stroke patients with atrial fibrillation should be anticoagulated for life. Anticoagulation in the setting of atrial fibrillation is seriously underutilized.21 The highest quality study on early anticoagulation for ischemic stroke associated with atrial fibrillation suggested that there was no benefit to starting anticoagulation earlier than 2 weeks after a stroke, and there may actually be a higher complication rate (compared to aspirin).22 Other cardiac indications for anticoagulation include left ventricular thrombus and mechanical valves.
Carotid Stenosis
Significant ipsilateral stenosis of the internal carotid artery in a patient with ischemic stroke is a strong indication for intervention, usually a standard carotid endarterectomy (CEA). Stenosis of 70% to 99% is the strongest indication for CEA, and may be of greatest benefit in men, those 75+ years of age, and if surgery is done 2 weeks after the most recent symptoms.23 In patients with minor stroke or TIA, recent recommendations and our practice is to admit to the hospital and perform endarterectomy as soon as possible (those with major stroke may have a greater risk of complications with early CEA).24 Stenting should only be considered instead of CEA if high risk (for surgical complications) criteria are present. These high risk criteria include patients having significant comorbidities and/or anatomic risk factors (ie, recurrent stenosis and/or previous radical neck dissection), and [who] would be poor candidates for CEA in the opinion of a surgeon.25 For stenoses of 50% to 69%, intervention is not as compelling, and decisions should be individualized based on patient characteristics; in this group, stenting should only be considered in the setting of a clinical trial or if an investigational device exemption (IDE) exists at your institution.26
Dissection of the Carotid or Vertebral Arteries
This is a common cause of stroke in younger adults. It should be suspected in patients without other clear causes of stroke and significant disease of the extracranial arteries. Diagnosis can usually be made with CTA or MRA, though it is suggested that the best modality may be T1‐fat‐saturated MRI images of the neck. Debate exists as to the best approach to treatment of dissections due to the absence of randomized trials. A recent comprehensive review suggested anticoagulation for 3 to 6 months followed by indefinite antiplatelet therapy for symptomatic dissections and antiplatelet therapy alone for asymptomatic dissections.27
PFO‐related Stroke
If the patient is found to have a PFO, its role in comparison to traditional risk factors must be weighed carefully. Epidemiological studies suggest that PFO may be most relevant in younger patients, those with cryptogenic stroke (no obvious cause and lack of traditional risk factors), those with higher risk associations including interatrial septal aneurysm, larger PFOs or history of previous cryptogenic stroke.28, 29 The best medical therapy for seemingly PFO‐related ischemic stroke is also unclear; a reasonable approach might be aspirin if neither high‐risk associations nor a hypercoagulable state is present, and warfarin if either are present. Transcatheter closure of PFO is approved by the U.S. Food and Drug Administration (FDA) only under an IDE for patients who have had a recurrent event on maximally tolerated medical treatment, and requires approval from the human research committee (internal review board [IRB]) at your hospital. It is not known if closure is superior or inferior to best medical therapy, and a practice parameter from the American Academy of Neurology strongly encourages appropriate patients to consider participation in ongoing randomized trials.28 Further information on these trials is available at:
Our patient underwent a CTA of the head and neck in the emergency room to see if he would be a candidate for other interventions; unfortunately, he did not meet the time criteria. CTA showed complete occlusion of the left internal carotid artery at the bifurcation with heterogeneous retrograde filling (Supporting Figure 1). Complete occlusion of the proximal third of the left M1 segment was also seen with relative oligemia in the left MCA distribution, though several small peripheral M3/M4 vessels were opacified in the territory indicating collateralization (Supporting Figure 2). A MRI showed a large area of diffusion‐weighted abnormality (Figure 1). Interestingly, the patient's transthoracic echocardiography (TTE), which did not show evidence of a PFO, did reveal a calcified thrombus in the left ventricle. Though no arrhythmias were captured on telemetry, this thrombus does serve as a potential source of cardioembolic emboli to the cerebral vasculature. It was felt that the most likely source of the patient's acute infarct was from artery‐to‐artery emboli from his internal carotid occlusion given the infarct location and the lack of infarction in other vascular distributions (as one might see from a cardiac embolic source). Therefore, his medical management consisted of an antiplatelet regimen for 2 weeks followed by a transition to warfarin alone 2 weeks after his acute infarct as secondary stroke prevention due to the cardiac thrombus. Given the complete occlusion of the internal carotid artery and M1 segment, there was concern that the penumbra might be at risk of infarction (supporting standard guidelines of permissive hypertension). By the end of his hospitalization, the patient had improved and was transferred to inpatient rehabilitation.
The guidelines for acute stroke management continue to rapidly evolve. Certainly, there are effective treatments for acute ischemic stroke, with variation based on the timing of patient arrival at the hospital, the underlying pathophysiology, and the treatment capabilities of the individual hospital. Secondary stroke prevention is extremely important and has been emphasized during inpatient admissions with the establishment of an appropriate medication regime, given that patients are more likely to stay on treatment that is initiated around the time of a diagnosis.29 Evidence strongly suggests that management of acute stroke is improved by an organized approach to care, including the expertise of a multidisciplinary team in a specialized stroke unit. Hospitals committed to high quality of care for acute stroke patients should strongly consider the Joint Commission certification process or an analogous local certification. Such certification demonstrates a hospital's commitment to providing high‐quality care, what every stroke patient wants and deserves.
- Organised inpatient (stroke unit) care for stroke.Stroke Unit Trialists' Collaboration.Cochrane Database Syst Rev.2000(2):CD000197.
- ,,, et al.Acetaminophen for altering body temperature in acute stroke: a randomized clinical trial.Stroke.2002;33(1):130–134.
- ,,, et al.Fever in acute stroke worsens prognosis. A prospective study.Stroke.1995;26(11):2040–2043.
- ,.Combating hyperthermia in acute stroke: a significant clinical concern.Stroke.1998;29(2):529–534.
- ,,, et al.Body temperature in acute stroke: relation to stroke severity, infarct size, mortality, and outcome.Lancet. 171996;347(8999):422–425.
- ,,.Thresholds in cerebral ischemia—the ischemic penumbra.Stroke.1981;12(6):723–725.
- .Ischaemic brain damage of cerebral perfusion failure type after treatment of severe hypertension.Br Med J. 271975;4(5999):739.
- ,,,,.Imaging of acute stroke.Lancet Neurol.2006;5(9):755–768.
- ,,, et al.High‐dose atorvastatin after stroke or transient ischemic attack.N Engl J Med.2006;355(6):549–559.
- ,,, et al.Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co‐sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline.Stroke.2006;37(2):577–617.
- Antithrombotic Trialists' Collaboration.Collaborative meta‐analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients.BMJ.2002;324(7329):71–86.
- ,,, et al.A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke.N Engl J Med.2001;345(20):1444–1451.
- .Warfarin or aspirin for recurrent ischemic stroke.N Engl J Med.2002;346(15):1169–1171.
- ,,, et al.Prevention. European Stroke Initiative.Cerebrovasc Dis.2004;17(suppl 2):15–29.
- ,,, et al.Update to the AHA/ASA recommendations for the prevention of stroke in patients with stroke and transient ischemic attack.Stroke.2008;39(5):1647–1652.
- ,,,,.Antithrombotic and thrombolytic therapy for ischemic stroke: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition).Chest.2008;133(suppl):630S–669S.
- ,,, et al.Aspirin and extended‐release dipyridamole versus clopidogrel for recurrent stroke.N Engl J Med.2008;359(12):1238–1251.
- ,,, et al.Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events.N Engl J Med.2006;354(16):1706–1717.
- ,,, et al.Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high‐risk patients (MATCH): randomised, double‐blind, placebo‐controlled trial.Lancet.2004;364(9431):331–337.
- ,,, et al.Oral anticoagulants vs aspirin in nonvalvular atrial fibrillation: an individual patient meta‐analysis.JAMA.2002;288(19):2441–2448.
- .Effective anticoagulation therapy: defining the gap between clinical studies and clinical practice.Am J Manag Care.2004;10(suppl):S297–S306; discussionS312–S297.
- ,,,.Low molecular‐weight heparin versus aspirin in patients with acute ischaemic stroke and atrial fibrillation: a double‐blind randomised study. HAEST Study Group. Heparin in Acute Embolic Stroke Trial.Lancet.2000;355(9211):1205–1210.
- ,,,,.Endarterectomy for symptomatic carotid stenosis in relation to clinical subgroups and timing of surgery.Lancet.2004;363(9413):915–924.
- ,,.The timing of carotid endarterectomy post stroke.Neurol Clin.2006;24(4):669–680.
- Centers for Medicare and Medicaid Services (CMS). Department of Health and Human Services (DHHS). CMS Manual System. Pub 100–03 Medicare National Coverage Determinations. Available at: http://www.cms.hhs.gov/Transmittals/Downloads/R64NCD.pdf. Accessed May2009.
- .Current status of carotid endarterectomy and stenting for symptomatic carotid stenosis.Cerebrovasc Dis.2007;24(suppl 1):116–125.
- ,,, et al.Antiplatelets versus anticoagulation in cervical artery dissection.Stroke.2007;38(9):2605–2611.
- ,,, et al.A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke.N Engl J Med2001;345(20):1444–1451.
- .Warfarin or aspirin for recurrent ischemic stroke.N Engl J Med2002;346(15):1169–1171.
- ,,, et al.Practice parameter: recurrent stroke with patent foramen ovale and atrial septal aneurysm: report of the Quality Standards Subcommittee of the American Academy of Neurology.Neurology.2004;62(7):1042–1050.
- ,,, et al.In‐hospital initiation of secondary stroke prevention therapies yields high rates of adherence at follow‐up.Stroke.2004;35(12):2879–2883.
Inpatient stroke management includes many elements of care, at least as important as the initial portion of the patient's stay, as reviewed in part 1 of this article. The extent of further diagnostic evaluation varies widely depending on apparent risk factors on presentation. Likewise, further therapy, both inpatient and secondary prevention is based on identification of stroke mechanism. Hospitalists are uniquely positioned to have a tremendous impact on both stroke care and the prevention of recurrent disease.
Case Presentation
A 76‐year‐old right‐handed male with a history of hyperlipidemia and myocardial infarction was found at 7 AM with right‐sided paralysis and poor responsiveness on the morning of admission. Upon arrival to the emergency department (ED), with symptoms of partial aphasia, right hemiplegia, and left gaze preference, there was a high suspicion for a left middle cerebral artery (MCA) stroke. Unfortunately, he was excluded from receiving intravenous (IV) tissue plasminogen activator (tPA) or any other acute interventions as the last time he was known to be neurologically intact was the prior evening, which is taken to be the time of onset. Antiplatelet therapy was continued, and the patient admitted for further workup.
Inpatient Care
When an acute ischemic stroke patient is admitted to the hospital, he or she should be placed on a standardized acute stroke protocol (also known as (a.k.a.) a care map, order set, clinical pathway)commonly created by a hospitalist/neurologist and a multidisciplinary team and admitted to a stroke unit. A stroke unit can take many forms, either as a physically separate unit in hospitals with sufficient volume or a floor where a lower volume of stroke patients are always admitted. Multidisciplinary care providers in the stroke unit have special training in stroke, and strong evidence from randomized trials shows that patients cared for in these units have significantly decreased mortality with improved functional outcomes.1 Essentials of the stroke protocol or order set include cardiac telemetry, maintaining euthermia and euglycemia, closely following blood pressure and neurologic status, actively avoiding complications, initiation of secondary prevention treatment, early involvement of rehabilitation services, and patient education.
Euthermia may be assisted by administering scheduled Tylenol to the patient for the first 48 hours, but is not strictly evidence‐based.2 Though euthermia and euglycemia have not been shown to improve outcomes in acute stroke, studies have shown that hyperthermia and hyperglycemia are associated with worsened outcomes for patients with acute strokes.35
Blood Pressure Management
Normally, cerebral vascular autoregulation leads to stable cerebral blood flow over a range of systemic blood pressures. In the setting of an acute stroke, the ability to autoregulate is diminished or absent in regions of and surrounding an acute ischemic stroke; as the area becomes ischemic, autoregulation opens the local vasculature maximally in an effort to drawn in as much blood as possible. Maximally dilated arterioles are perfused in direct correlation with systemic blood pressure, thus any drop in the systemic blood pressure leads to direct decreases in blood flow specifically in the area of ischemia; if there is a penumbra of marginally perfused tissue, such systemic blood pressure drops risk extending the area of fatal ischemia (increasing the size of the ischemic stroke).68 Thus in the acute period of an ischemic stroke, the American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for the Early Management of Adults With Ischemic Stroke (referred to herein as the Guidelines)10 suggest avoid treatment unless systolic blood pressures are >220 or diastolic pressures >105, and review the evidence to support this recommendation (p. 16711672). Those patients who receive tPA have a more stringent blood pressure threshold given their risk of intracranial hemorrhage; systolic blood pressures are accepted up to 180 prior to recommending treatment.
Higher‐quality Inpatient Stroke Care and Harmonized Performance Measures
Beginning in January 2008, a set of 10 performance measures (Table 1) for inpatient acute stroke care have been agreed upon (harmonized) by 3 major stakeholders including the Joint Commission, the ASA's Get with the GuidelinesStroke quality improvement program, and the Center for Disease Control and Prevention's (CDC's) Paul Coverdell Acute stroke registries. These performance measures were selected to help avoid complications (deep vein thrombosis [DVT], aspiration pneumonia), encourage appropriately aggressive care (tPA administration), optimize secondary prevention (antithrombotics, cholesterol lowering, smoking cessation, education), and facilitate functional recovery (early rehabilitation). All 10 measures are appropriate for consideration in every ischemic stroke patient, and 5 are appropriate for the hemorrhagic stroke types.
| Performance measure* | Definition* |
|---|---|
| |
| 1. DVT prophylaxis | Patients who are nonambulatory should start receiving DVT prophylaxis by end of hospital day 2 (can be either compression devices or any low‐dose heparin) |
| 2. Discharged on antithrombotic therapy | Antiplatelet agent(s) or warfarin anticoagulation |
| 3. Patients with atrial fibrillation receiving anticoagulation therapy | A proven approach to secondary prevention in such patients; practice at Harborview varies time of warfarin initiation based on infarct size with larger infarcts waiting up to 2 weeks before initiating warfarin (the best randomized trial showed no benefit for full‐dose low‐molecular‐weight heparin over aspirin in the first 2 weeks)50 |
| 4. Thrombolytic therapy administered | In ischemic stroke patients who arrive at the hospital within 120 minutes (2 hours) of time last known well, for whom IV tPA was initiated at this hospital within 180 minutes (3 hours) of time last known well, and who qualify under strict criteria |
| 5. Antithrombotic therapy by end of hospital day 2 | Usually just antiplatelet agents, a minimal standard of care for ischemic stroke patients; should be started as early as possible, usually in ER |
| 6. Discharged on statin medication | If LDL >100, or not measured or if on a statin drug prior to admission; to reduce risk of subsequent ischemic stroke |
| 7. Dysphagia screening | Prior to any PO food, fluids or medications; to reduce the chances of aspiration pneumonia |
| 8. Stroke education | Including for families if patient unable to participate, must include personal risk factors for stroke, warning signs for stroke, activation of emergency medical system, need for follow‐up after discharge, and medications prescribed |
| 9. Smoking cessation/advice/counseling | For any patient who has smoked in the last year |
| 10. Assessed for rehabilitation | Or received therapy services; to facilitate progress to an optimal function outcome |
Further Workup
After the ischemic stroke patient has had their computed tomography (CT) scan, possibly a computed tomography angiography (CTA), been admitted to the stroke unit, started on an antithrombotic medication, and had their blood pressure appropriately treated, attention then turns to defining the pathophysiology related to the stroke and starting an optimal regimen for secondary prevention. Imaging of the cerebral vasculature including both extracranial and intracranial large vessels is a vital first step in understanding the cause of ischemic stroke. There are multiple potential modalities (magnetic resonance angiography [MRA], CTA, and duplex/transcranial Doppler), the choice of which depends on local availability and expertise as well as the specific clinical situation. Magnetic resonance imaging (MRI) of the brain for all ischemic stroke patients is standard of care at most stroke centers; per the Guidelines, MRI is better at distinguishing acute, small cortical, small deep, and posterior fossa infarcts; at distinguishing acute from chronic ischemia; and at identifying subclinical satellite ischemic lesions that provide information on stroke mechanism (p. 1668). New techniques including magnetic resonance (MR) and CT perfusion scanning can show the ischemic region in the acute setting and may one day help select patients for specific therapies, but are not yet widely available nor have they been shown to alter outcomes.
An electrocardiogram is indicated for all stroke patients, as is admission to a cardiac telemetry bed for at least 24 hours to document any arrhythmias, the most common being atrial fibrillation (Guidelines, p. 1666, 1673). An echocardiographic study (ECHO) of the heart with bubble study should be performed in most cases (although which cases may specifically benefit is unclear) to identify a cardioembolic source for the stroke, such as low cardiac ejection fraction, atrial septal aneurysm, patent foramen ovale (PFO), or a cardiac thrombus. The bubble study increases the sensitivity of detecting a PFO, which could serve as a gateway for venous embolization to the cerebral arteries. Assuming a large PFO is discovered, other studies such as lower extremity Doppler may be warranted to investigate other potential sources of thrombi (ie, DVT).
Regarding laboratory testing, fasting lipids should be checked as hyperlipidemia is a common modifiable risk factor for ischemic stroke. The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial included ischemic stroke patients that had low‐density lipoprotein (LDL) cholesterol between 100 mg/dL and 190 mg/dL and randomized them to receive atorvastatin 80 mg/day vs. placebo. Results showed a 16% relative risk reduction in recurrent stroke; however, there was a small increased risk of intracranial hemorrhage.9 As shown in Table 1, use of a statin on discharge is now a national performance measure for ischemic stroke.
Dissection is a common cause of stroke in young patients without traditional risk factors. Other serologies, such as hypercoagulable studies, may be warranted in patients with no other risk factors for strokes, paradoxical embolus, or of young age (eg, 45 years and under). The arterial hypercoagulable panel consists of antiphospholipid antibody panel, homocysteine levels, lupus anticoagulant levels, and prothrombin time/partial thromboplastin time (PT/PTT). The venous hypercoagulable panel consists of the laboratory values checked, with the arterial hypercoagulable and activated protein C (APC) resistance, Factor VIII activity, Factor II DNA, Factor V DNA if the APC resistance is positive, antithrombin III activity, and activity of proteins C and S. If a patient is found to have a hypercoagulable state, long‐term therapy often involves careful consideration of the choice of antiplatelet therapy vs. anticoagulation with warfarin.10
Initiating Secondary Prevention
Upon admission, the clinician faces a variety of treatment choices for secondary stroke prevention. The proper choice depends on the results of the workup and the presumptive pathophysiology.
Noncardioembolic/Atherothrombotic/Lacunar
The Antithrombotic Trialists' Collaboration meta‐analysis found that patients with a prior stroke or transient ischemic attack (TIA) had a highly significant decrease in the rate of subsequent vascular events (over about 3 years) on antiplatelet therapy (17.8% vs. 21.4%, P 0.0001) and were unable to find a significant difference between low‐dose and high‐dose aspirin for secondary prevention.11 Thus, it is reasonable to place an acute stroke patient naive to antithrombotic therapy on 81 mg of aspirin or 325 mg for long‐term prevention (325 mg is specifically recommended in the acute setting). Several studies such as the WARSS and ESPRIT trials have shown antiplatelet agents to be at least as effective as anticoagulation in noncardioembolic ischemic strokes.12, 13 Guidelines from Europe, the American College of Chest Physicians, and the AHA/ASA all state it is acceptable to choose either aspirin monotherapy, aspirin/extended release dipyridamole combination therapy, or clopidogrel monotherapy as first‐line agents for long‐term secondary prevention in noncardioembolic ischemic stroke.1416 There is no clear evidence that patients who suffer an ischemic stroke while on aspirin will derive additional benefit from increasing the aspirin dose. The newer guidelines go on to recommend aspirin/extended release dipyridamole (ER‐DP) combination therapy or clopidogrel monotherapy over aspirin monotherapy, the former with a stronger level of recommendation based on the results of 2 randomized trials. These recommendations were all published without knowledge of the results of the Prevention Regimen For Effectively Avoiding Second Strokes (PRoFESS) study, which directly compared aspirin/extended release dipyridamole combination therapy to clopidogrel monotherapy for long‐term secondary prevention. The rate of first recurrent stroke was not significantly different between the 2 therapies (9.0% ER‐DP plus aspirin, 8.8% clopidogrel; hazard ratio [HR], 1.01; 95% confidence interval [CI], 0.921.11). Other outcomes also showed few differences, although there were more major hemorrhagic events in the ER‐DP plus aspirin group (4.1% vs. 3.6%; HR, 1.15; 95% CI, 1.001.32; P = 0.06).17
The ASA Stroke Prevention Guideline from 2006 states, with continued relevance, The selection of an antiplatelet agent should be individualized on the basis of patient risk factor profiles, tolerance, and other clinical characteristics.10 Of note, both the Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance (CHARISMA) and Management of ATherothrombosis with Clopidogrel in High‐risk patients with recent TIA or ischemic stroke (MATCH) trials found a significant increased risk for hemorrhage complications with long‐term use of the aspirin and clopidogrel combination,18, 19 and the 2008 update to the ASA Stroke prevention guidelines state that the addition of aspirin to clopidogrel increases the risk of hemorrhage. Combination therapy of aspirin and clopidogrel is not routinely recommended for ischemic stroke or TIA patients unless they have a specific indication for this therapy (i.e., coronary stent or acute coronary syndrome).15
Atrial Fibrillation
Though our case patient did not have atrial fibrillation, this condition does deserve mention. About 15% to 20% of ischemic stroke patients have atrial fibrillation. The overall risk for stroke in patients with atrial fibrillation is about 5% per year; however, patients who have a history of stroke increase their risk factors for subsequent strokes to about 12% per year. In most cases, anticoagulation has proven to be the superior agent for primary and secondary stroke prevention with warfarin reducing the risk by 67% compared to aspirin, which only reduces the risk of stroke by 20%. A meta‐analysis from 2002 showed that patients who had a prior stroke or TIA decrease their risk of subsequent strokes to 4%/year on oral anticoagulation therapy, resulting in an 8% absolute risk reduction. Patients on aspirin therapy only decrease their risk to 10%/year, or a 2% reduction in stroke events.20 Unless there is a strong contraindication (eg, bleeding diathesis, history of life threatening gastrointestinal [GI] bleeding, history of fall with subdural hematoma, etc.), virtually all ischemic stroke patients with atrial fibrillation should be anticoagulated for life. Anticoagulation in the setting of atrial fibrillation is seriously underutilized.21 The highest quality study on early anticoagulation for ischemic stroke associated with atrial fibrillation suggested that there was no benefit to starting anticoagulation earlier than 2 weeks after a stroke, and there may actually be a higher complication rate (compared to aspirin).22 Other cardiac indications for anticoagulation include left ventricular thrombus and mechanical valves.
Carotid Stenosis
Significant ipsilateral stenosis of the internal carotid artery in a patient with ischemic stroke is a strong indication for intervention, usually a standard carotid endarterectomy (CEA). Stenosis of 70% to 99% is the strongest indication for CEA, and may be of greatest benefit in men, those 75+ years of age, and if surgery is done 2 weeks after the most recent symptoms.23 In patients with minor stroke or TIA, recent recommendations and our practice is to admit to the hospital and perform endarterectomy as soon as possible (those with major stroke may have a greater risk of complications with early CEA).24 Stenting should only be considered instead of CEA if high risk (for surgical complications) criteria are present. These high risk criteria include patients having significant comorbidities and/or anatomic risk factors (ie, recurrent stenosis and/or previous radical neck dissection), and [who] would be poor candidates for CEA in the opinion of a surgeon.25 For stenoses of 50% to 69%, intervention is not as compelling, and decisions should be individualized based on patient characteristics; in this group, stenting should only be considered in the setting of a clinical trial or if an investigational device exemption (IDE) exists at your institution.26
Dissection of the Carotid or Vertebral Arteries
This is a common cause of stroke in younger adults. It should be suspected in patients without other clear causes of stroke and significant disease of the extracranial arteries. Diagnosis can usually be made with CTA or MRA, though it is suggested that the best modality may be T1‐fat‐saturated MRI images of the neck. Debate exists as to the best approach to treatment of dissections due to the absence of randomized trials. A recent comprehensive review suggested anticoagulation for 3 to 6 months followed by indefinite antiplatelet therapy for symptomatic dissections and antiplatelet therapy alone for asymptomatic dissections.27
PFO‐related Stroke
If the patient is found to have a PFO, its role in comparison to traditional risk factors must be weighed carefully. Epidemiological studies suggest that PFO may be most relevant in younger patients, those with cryptogenic stroke (no obvious cause and lack of traditional risk factors), those with higher risk associations including interatrial septal aneurysm, larger PFOs or history of previous cryptogenic stroke.28, 29 The best medical therapy for seemingly PFO‐related ischemic stroke is also unclear; a reasonable approach might be aspirin if neither high‐risk associations nor a hypercoagulable state is present, and warfarin if either are present. Transcatheter closure of PFO is approved by the U.S. Food and Drug Administration (FDA) only under an IDE for patients who have had a recurrent event on maximally tolerated medical treatment, and requires approval from the human research committee (internal review board [IRB]) at your hospital. It is not known if closure is superior or inferior to best medical therapy, and a practice parameter from the American Academy of Neurology strongly encourages appropriate patients to consider participation in ongoing randomized trials.28 Further information on these trials is available at:
Our patient underwent a CTA of the head and neck in the emergency room to see if he would be a candidate for other interventions; unfortunately, he did not meet the time criteria. CTA showed complete occlusion of the left internal carotid artery at the bifurcation with heterogeneous retrograde filling (Supporting Figure 1). Complete occlusion of the proximal third of the left M1 segment was also seen with relative oligemia in the left MCA distribution, though several small peripheral M3/M4 vessels were opacified in the territory indicating collateralization (Supporting Figure 2). A MRI showed a large area of diffusion‐weighted abnormality (Figure 1). Interestingly, the patient's transthoracic echocardiography (TTE), which did not show evidence of a PFO, did reveal a calcified thrombus in the left ventricle. Though no arrhythmias were captured on telemetry, this thrombus does serve as a potential source of cardioembolic emboli to the cerebral vasculature. It was felt that the most likely source of the patient's acute infarct was from artery‐to‐artery emboli from his internal carotid occlusion given the infarct location and the lack of infarction in other vascular distributions (as one might see from a cardiac embolic source). Therefore, his medical management consisted of an antiplatelet regimen for 2 weeks followed by a transition to warfarin alone 2 weeks after his acute infarct as secondary stroke prevention due to the cardiac thrombus. Given the complete occlusion of the internal carotid artery and M1 segment, there was concern that the penumbra might be at risk of infarction (supporting standard guidelines of permissive hypertension). By the end of his hospitalization, the patient had improved and was transferred to inpatient rehabilitation.

The guidelines for acute stroke management continue to rapidly evolve. Certainly, there are effective treatments for acute ischemic stroke, with variation based on the timing of patient arrival at the hospital, the underlying pathophysiology, and the treatment capabilities of the individual hospital. Secondary stroke prevention is extremely important and has been emphasized during inpatient admissions with the establishment of an appropriate medication regime, given that patients are more likely to stay on treatment that is initiated around the time of a diagnosis.29 Evidence strongly suggests that management of acute stroke is improved by an organized approach to care, including the expertise of a multidisciplinary team in a specialized stroke unit. Hospitals committed to high quality of care for acute stroke patients should strongly consider the Joint Commission certification process or an analogous local certification. Such certification demonstrates a hospital's commitment to providing high‐quality care, what every stroke patient wants and deserves.
Inpatient stroke management includes many elements of care, at least as important as the initial portion of the patient's stay, as reviewed in part 1 of this article. The extent of further diagnostic evaluation varies widely depending on apparent risk factors on presentation. Likewise, further therapy, both inpatient and secondary prevention is based on identification of stroke mechanism. Hospitalists are uniquely positioned to have a tremendous impact on both stroke care and the prevention of recurrent disease.
Case Presentation
A 76‐year‐old right‐handed male with a history of hyperlipidemia and myocardial infarction was found at 7 AM with right‐sided paralysis and poor responsiveness on the morning of admission. Upon arrival to the emergency department (ED), with symptoms of partial aphasia, right hemiplegia, and left gaze preference, there was a high suspicion for a left middle cerebral artery (MCA) stroke. Unfortunately, he was excluded from receiving intravenous (IV) tissue plasminogen activator (tPA) or any other acute interventions as the last time he was known to be neurologically intact was the prior evening, which is taken to be the time of onset. Antiplatelet therapy was continued, and the patient admitted for further workup.
Inpatient Care
When an acute ischemic stroke patient is admitted to the hospital, he or she should be placed on a standardized acute stroke protocol (also known as (a.k.a.) a care map, order set, clinical pathway)commonly created by a hospitalist/neurologist and a multidisciplinary team and admitted to a stroke unit. A stroke unit can take many forms, either as a physically separate unit in hospitals with sufficient volume or a floor where a lower volume of stroke patients are always admitted. Multidisciplinary care providers in the stroke unit have special training in stroke, and strong evidence from randomized trials shows that patients cared for in these units have significantly decreased mortality with improved functional outcomes.1 Essentials of the stroke protocol or order set include cardiac telemetry, maintaining euthermia and euglycemia, closely following blood pressure and neurologic status, actively avoiding complications, initiation of secondary prevention treatment, early involvement of rehabilitation services, and patient education.
Euthermia may be assisted by administering scheduled Tylenol to the patient for the first 48 hours, but is not strictly evidence‐based.2 Though euthermia and euglycemia have not been shown to improve outcomes in acute stroke, studies have shown that hyperthermia and hyperglycemia are associated with worsened outcomes for patients with acute strokes.35
Blood Pressure Management
Normally, cerebral vascular autoregulation leads to stable cerebral blood flow over a range of systemic blood pressures. In the setting of an acute stroke, the ability to autoregulate is diminished or absent in regions of and surrounding an acute ischemic stroke; as the area becomes ischemic, autoregulation opens the local vasculature maximally in an effort to drawn in as much blood as possible. Maximally dilated arterioles are perfused in direct correlation with systemic blood pressure, thus any drop in the systemic blood pressure leads to direct decreases in blood flow specifically in the area of ischemia; if there is a penumbra of marginally perfused tissue, such systemic blood pressure drops risk extending the area of fatal ischemia (increasing the size of the ischemic stroke).68 Thus in the acute period of an ischemic stroke, the American Heart Association (AHA)/American Stroke Association (ASA) Guidelines for the Early Management of Adults With Ischemic Stroke (referred to herein as the Guidelines)10 suggest avoid treatment unless systolic blood pressures are >220 or diastolic pressures >105, and review the evidence to support this recommendation (p. 16711672). Those patients who receive tPA have a more stringent blood pressure threshold given their risk of intracranial hemorrhage; systolic blood pressures are accepted up to 180 prior to recommending treatment.
Higher‐quality Inpatient Stroke Care and Harmonized Performance Measures
Beginning in January 2008, a set of 10 performance measures (Table 1) for inpatient acute stroke care have been agreed upon (harmonized) by 3 major stakeholders including the Joint Commission, the ASA's Get with the GuidelinesStroke quality improvement program, and the Center for Disease Control and Prevention's (CDC's) Paul Coverdell Acute stroke registries. These performance measures were selected to help avoid complications (deep vein thrombosis [DVT], aspiration pneumonia), encourage appropriately aggressive care (tPA administration), optimize secondary prevention (antithrombotics, cholesterol lowering, smoking cessation, education), and facilitate functional recovery (early rehabilitation). All 10 measures are appropriate for consideration in every ischemic stroke patient, and 5 are appropriate for the hemorrhagic stroke types.
| Performance measure* | Definition* |
|---|---|
| |
| 1. DVT prophylaxis | Patients who are nonambulatory should start receiving DVT prophylaxis by end of hospital day 2 (can be either compression devices or any low‐dose heparin) |
| 2. Discharged on antithrombotic therapy | Antiplatelet agent(s) or warfarin anticoagulation |
| 3. Patients with atrial fibrillation receiving anticoagulation therapy | A proven approach to secondary prevention in such patients; practice at Harborview varies time of warfarin initiation based on infarct size with larger infarcts waiting up to 2 weeks before initiating warfarin (the best randomized trial showed no benefit for full‐dose low‐molecular‐weight heparin over aspirin in the first 2 weeks)50 |
| 4. Thrombolytic therapy administered | In ischemic stroke patients who arrive at the hospital within 120 minutes (2 hours) of time last known well, for whom IV tPA was initiated at this hospital within 180 minutes (3 hours) of time last known well, and who qualify under strict criteria |
| 5. Antithrombotic therapy by end of hospital day 2 | Usually just antiplatelet agents, a minimal standard of care for ischemic stroke patients; should be started as early as possible, usually in ER |
| 6. Discharged on statin medication | If LDL >100, or not measured or if on a statin drug prior to admission; to reduce risk of subsequent ischemic stroke |
| 7. Dysphagia screening | Prior to any PO food, fluids or medications; to reduce the chances of aspiration pneumonia |
| 8. Stroke education | Including for families if patient unable to participate, must include personal risk factors for stroke, warning signs for stroke, activation of emergency medical system, need for follow‐up after discharge, and medications prescribed |
| 9. Smoking cessation/advice/counseling | For any patient who has smoked in the last year |
| 10. Assessed for rehabilitation | Or received therapy services; to facilitate progress to an optimal function outcome |
Further Workup
After the ischemic stroke patient has had their computed tomography (CT) scan, possibly a computed tomography angiography (CTA), been admitted to the stroke unit, started on an antithrombotic medication, and had their blood pressure appropriately treated, attention then turns to defining the pathophysiology related to the stroke and starting an optimal regimen for secondary prevention. Imaging of the cerebral vasculature including both extracranial and intracranial large vessels is a vital first step in understanding the cause of ischemic stroke. There are multiple potential modalities (magnetic resonance angiography [MRA], CTA, and duplex/transcranial Doppler), the choice of which depends on local availability and expertise as well as the specific clinical situation. Magnetic resonance imaging (MRI) of the brain for all ischemic stroke patients is standard of care at most stroke centers; per the Guidelines, MRI is better at distinguishing acute, small cortical, small deep, and posterior fossa infarcts; at distinguishing acute from chronic ischemia; and at identifying subclinical satellite ischemic lesions that provide information on stroke mechanism (p. 1668). New techniques including magnetic resonance (MR) and CT perfusion scanning can show the ischemic region in the acute setting and may one day help select patients for specific therapies, but are not yet widely available nor have they been shown to alter outcomes.
An electrocardiogram is indicated for all stroke patients, as is admission to a cardiac telemetry bed for at least 24 hours to document any arrhythmias, the most common being atrial fibrillation (Guidelines, p. 1666, 1673). An echocardiographic study (ECHO) of the heart with bubble study should be performed in most cases (although which cases may specifically benefit is unclear) to identify a cardioembolic source for the stroke, such as low cardiac ejection fraction, atrial septal aneurysm, patent foramen ovale (PFO), or a cardiac thrombus. The bubble study increases the sensitivity of detecting a PFO, which could serve as a gateway for venous embolization to the cerebral arteries. Assuming a large PFO is discovered, other studies such as lower extremity Doppler may be warranted to investigate other potential sources of thrombi (ie, DVT).
Regarding laboratory testing, fasting lipids should be checked as hyperlipidemia is a common modifiable risk factor for ischemic stroke. The Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial included ischemic stroke patients that had low‐density lipoprotein (LDL) cholesterol between 100 mg/dL and 190 mg/dL and randomized them to receive atorvastatin 80 mg/day vs. placebo. Results showed a 16% relative risk reduction in recurrent stroke; however, there was a small increased risk of intracranial hemorrhage.9 As shown in Table 1, use of a statin on discharge is now a national performance measure for ischemic stroke.
Dissection is a common cause of stroke in young patients without traditional risk factors. Other serologies, such as hypercoagulable studies, may be warranted in patients with no other risk factors for strokes, paradoxical embolus, or of young age (eg, 45 years and under). The arterial hypercoagulable panel consists of antiphospholipid antibody panel, homocysteine levels, lupus anticoagulant levels, and prothrombin time/partial thromboplastin time (PT/PTT). The venous hypercoagulable panel consists of the laboratory values checked, with the arterial hypercoagulable and activated protein C (APC) resistance, Factor VIII activity, Factor II DNA, Factor V DNA if the APC resistance is positive, antithrombin III activity, and activity of proteins C and S. If a patient is found to have a hypercoagulable state, long‐term therapy often involves careful consideration of the choice of antiplatelet therapy vs. anticoagulation with warfarin.10
Initiating Secondary Prevention
Upon admission, the clinician faces a variety of treatment choices for secondary stroke prevention. The proper choice depends on the results of the workup and the presumptive pathophysiology.
Noncardioembolic/Atherothrombotic/Lacunar
The Antithrombotic Trialists' Collaboration meta‐analysis found that patients with a prior stroke or transient ischemic attack (TIA) had a highly significant decrease in the rate of subsequent vascular events (over about 3 years) on antiplatelet therapy (17.8% vs. 21.4%, P 0.0001) and were unable to find a significant difference between low‐dose and high‐dose aspirin for secondary prevention.11 Thus, it is reasonable to place an acute stroke patient naive to antithrombotic therapy on 81 mg of aspirin or 325 mg for long‐term prevention (325 mg is specifically recommended in the acute setting). Several studies such as the WARSS and ESPRIT trials have shown antiplatelet agents to be at least as effective as anticoagulation in noncardioembolic ischemic strokes.12, 13 Guidelines from Europe, the American College of Chest Physicians, and the AHA/ASA all state it is acceptable to choose either aspirin monotherapy, aspirin/extended release dipyridamole combination therapy, or clopidogrel monotherapy as first‐line agents for long‐term secondary prevention in noncardioembolic ischemic stroke.1416 There is no clear evidence that patients who suffer an ischemic stroke while on aspirin will derive additional benefit from increasing the aspirin dose. The newer guidelines go on to recommend aspirin/extended release dipyridamole (ER‐DP) combination therapy or clopidogrel monotherapy over aspirin monotherapy, the former with a stronger level of recommendation based on the results of 2 randomized trials. These recommendations were all published without knowledge of the results of the Prevention Regimen For Effectively Avoiding Second Strokes (PRoFESS) study, which directly compared aspirin/extended release dipyridamole combination therapy to clopidogrel monotherapy for long‐term secondary prevention. The rate of first recurrent stroke was not significantly different between the 2 therapies (9.0% ER‐DP plus aspirin, 8.8% clopidogrel; hazard ratio [HR], 1.01; 95% confidence interval [CI], 0.921.11). Other outcomes also showed few differences, although there were more major hemorrhagic events in the ER‐DP plus aspirin group (4.1% vs. 3.6%; HR, 1.15; 95% CI, 1.001.32; P = 0.06).17
The ASA Stroke Prevention Guideline from 2006 states, with continued relevance, The selection of an antiplatelet agent should be individualized on the basis of patient risk factor profiles, tolerance, and other clinical characteristics.10 Of note, both the Clopidogrel for High Atherothrombotic Risk and Ischemic Stabilization, Management and Avoidance (CHARISMA) and Management of ATherothrombosis with Clopidogrel in High‐risk patients with recent TIA or ischemic stroke (MATCH) trials found a significant increased risk for hemorrhage complications with long‐term use of the aspirin and clopidogrel combination,18, 19 and the 2008 update to the ASA Stroke prevention guidelines state that the addition of aspirin to clopidogrel increases the risk of hemorrhage. Combination therapy of aspirin and clopidogrel is not routinely recommended for ischemic stroke or TIA patients unless they have a specific indication for this therapy (i.e., coronary stent or acute coronary syndrome).15
Atrial Fibrillation
Though our case patient did not have atrial fibrillation, this condition does deserve mention. About 15% to 20% of ischemic stroke patients have atrial fibrillation. The overall risk for stroke in patients with atrial fibrillation is about 5% per year; however, patients who have a history of stroke increase their risk factors for subsequent strokes to about 12% per year. In most cases, anticoagulation has proven to be the superior agent for primary and secondary stroke prevention with warfarin reducing the risk by 67% compared to aspirin, which only reduces the risk of stroke by 20%. A meta‐analysis from 2002 showed that patients who had a prior stroke or TIA decrease their risk of subsequent strokes to 4%/year on oral anticoagulation therapy, resulting in an 8% absolute risk reduction. Patients on aspirin therapy only decrease their risk to 10%/year, or a 2% reduction in stroke events.20 Unless there is a strong contraindication (eg, bleeding diathesis, history of life threatening gastrointestinal [GI] bleeding, history of fall with subdural hematoma, etc.), virtually all ischemic stroke patients with atrial fibrillation should be anticoagulated for life. Anticoagulation in the setting of atrial fibrillation is seriously underutilized.21 The highest quality study on early anticoagulation for ischemic stroke associated with atrial fibrillation suggested that there was no benefit to starting anticoagulation earlier than 2 weeks after a stroke, and there may actually be a higher complication rate (compared to aspirin).22 Other cardiac indications for anticoagulation include left ventricular thrombus and mechanical valves.
Carotid Stenosis
Significant ipsilateral stenosis of the internal carotid artery in a patient with ischemic stroke is a strong indication for intervention, usually a standard carotid endarterectomy (CEA). Stenosis of 70% to 99% is the strongest indication for CEA, and may be of greatest benefit in men, those 75+ years of age, and if surgery is done 2 weeks after the most recent symptoms.23 In patients with minor stroke or TIA, recent recommendations and our practice is to admit to the hospital and perform endarterectomy as soon as possible (those with major stroke may have a greater risk of complications with early CEA).24 Stenting should only be considered instead of CEA if high risk (for surgical complications) criteria are present. These high risk criteria include patients having significant comorbidities and/or anatomic risk factors (ie, recurrent stenosis and/or previous radical neck dissection), and [who] would be poor candidates for CEA in the opinion of a surgeon.25 For stenoses of 50% to 69%, intervention is not as compelling, and decisions should be individualized based on patient characteristics; in this group, stenting should only be considered in the setting of a clinical trial or if an investigational device exemption (IDE) exists at your institution.26
Dissection of the Carotid or Vertebral Arteries
This is a common cause of stroke in younger adults. It should be suspected in patients without other clear causes of stroke and significant disease of the extracranial arteries. Diagnosis can usually be made with CTA or MRA, though it is suggested that the best modality may be T1‐fat‐saturated MRI images of the neck. Debate exists as to the best approach to treatment of dissections due to the absence of randomized trials. A recent comprehensive review suggested anticoagulation for 3 to 6 months followed by indefinite antiplatelet therapy for symptomatic dissections and antiplatelet therapy alone for asymptomatic dissections.27
PFO‐related Stroke
If the patient is found to have a PFO, its role in comparison to traditional risk factors must be weighed carefully. Epidemiological studies suggest that PFO may be most relevant in younger patients, those with cryptogenic stroke (no obvious cause and lack of traditional risk factors), those with higher risk associations including interatrial septal aneurysm, larger PFOs or history of previous cryptogenic stroke.28, 29 The best medical therapy for seemingly PFO‐related ischemic stroke is also unclear; a reasonable approach might be aspirin if neither high‐risk associations nor a hypercoagulable state is present, and warfarin if either are present. Transcatheter closure of PFO is approved by the U.S. Food and Drug Administration (FDA) only under an IDE for patients who have had a recurrent event on maximally tolerated medical treatment, and requires approval from the human research committee (internal review board [IRB]) at your hospital. It is not known if closure is superior or inferior to best medical therapy, and a practice parameter from the American Academy of Neurology strongly encourages appropriate patients to consider participation in ongoing randomized trials.28 Further information on these trials is available at:
Our patient underwent a CTA of the head and neck in the emergency room to see if he would be a candidate for other interventions; unfortunately, he did not meet the time criteria. CTA showed complete occlusion of the left internal carotid artery at the bifurcation with heterogeneous retrograde filling (Supporting Figure 1). Complete occlusion of the proximal third of the left M1 segment was also seen with relative oligemia in the left MCA distribution, though several small peripheral M3/M4 vessels were opacified in the territory indicating collateralization (Supporting Figure 2). A MRI showed a large area of diffusion‐weighted abnormality (Figure 1). Interestingly, the patient's transthoracic echocardiography (TTE), which did not show evidence of a PFO, did reveal a calcified thrombus in the left ventricle. Though no arrhythmias were captured on telemetry, this thrombus does serve as a potential source of cardioembolic emboli to the cerebral vasculature. It was felt that the most likely source of the patient's acute infarct was from artery‐to‐artery emboli from his internal carotid occlusion given the infarct location and the lack of infarction in other vascular distributions (as one might see from a cardiac embolic source). Therefore, his medical management consisted of an antiplatelet regimen for 2 weeks followed by a transition to warfarin alone 2 weeks after his acute infarct as secondary stroke prevention due to the cardiac thrombus. Given the complete occlusion of the internal carotid artery and M1 segment, there was concern that the penumbra might be at risk of infarction (supporting standard guidelines of permissive hypertension). By the end of his hospitalization, the patient had improved and was transferred to inpatient rehabilitation.

The guidelines for acute stroke management continue to rapidly evolve. Certainly, there are effective treatments for acute ischemic stroke, with variation based on the timing of patient arrival at the hospital, the underlying pathophysiology, and the treatment capabilities of the individual hospital. Secondary stroke prevention is extremely important and has been emphasized during inpatient admissions with the establishment of an appropriate medication regime, given that patients are more likely to stay on treatment that is initiated around the time of a diagnosis.29 Evidence strongly suggests that management of acute stroke is improved by an organized approach to care, including the expertise of a multidisciplinary team in a specialized stroke unit. Hospitals committed to high quality of care for acute stroke patients should strongly consider the Joint Commission certification process or an analogous local certification. Such certification demonstrates a hospital's commitment to providing high‐quality care, what every stroke patient wants and deserves.
- Organised inpatient (stroke unit) care for stroke.Stroke Unit Trialists' Collaboration.Cochrane Database Syst Rev.2000(2):CD000197.
- ,,, et al.Acetaminophen for altering body temperature in acute stroke: a randomized clinical trial.Stroke.2002;33(1):130–134.
- ,,, et al.Fever in acute stroke worsens prognosis. A prospective study.Stroke.1995;26(11):2040–2043.
- ,.Combating hyperthermia in acute stroke: a significant clinical concern.Stroke.1998;29(2):529–534.
- ,,, et al.Body temperature in acute stroke: relation to stroke severity, infarct size, mortality, and outcome.Lancet. 171996;347(8999):422–425.
- ,,.Thresholds in cerebral ischemia—the ischemic penumbra.Stroke.1981;12(6):723–725.
- .Ischaemic brain damage of cerebral perfusion failure type after treatment of severe hypertension.Br Med J. 271975;4(5999):739.
- ,,,,.Imaging of acute stroke.Lancet Neurol.2006;5(9):755–768.
- ,,, et al.High‐dose atorvastatin after stroke or transient ischemic attack.N Engl J Med.2006;355(6):549–559.
- ,,, et al.Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co‐sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline.Stroke.2006;37(2):577–617.
- Antithrombotic Trialists' Collaboration.Collaborative meta‐analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients.BMJ.2002;324(7329):71–86.
- ,,, et al.A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke.N Engl J Med.2001;345(20):1444–1451.
- .Warfarin or aspirin for recurrent ischemic stroke.N Engl J Med.2002;346(15):1169–1171.
- ,,, et al.Prevention. European Stroke Initiative.Cerebrovasc Dis.2004;17(suppl 2):15–29.
- ,,, et al.Update to the AHA/ASA recommendations for the prevention of stroke in patients with stroke and transient ischemic attack.Stroke.2008;39(5):1647–1652.
- ,,,,.Antithrombotic and thrombolytic therapy for ischemic stroke: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition).Chest.2008;133(suppl):630S–669S.
- ,,, et al.Aspirin and extended‐release dipyridamole versus clopidogrel for recurrent stroke.N Engl J Med.2008;359(12):1238–1251.
- ,,, et al.Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events.N Engl J Med.2006;354(16):1706–1717.
- ,,, et al.Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high‐risk patients (MATCH): randomised, double‐blind, placebo‐controlled trial.Lancet.2004;364(9431):331–337.
- ,,, et al.Oral anticoagulants vs aspirin in nonvalvular atrial fibrillation: an individual patient meta‐analysis.JAMA.2002;288(19):2441–2448.
- .Effective anticoagulation therapy: defining the gap between clinical studies and clinical practice.Am J Manag Care.2004;10(suppl):S297–S306; discussionS312–S297.
- ,,,.Low molecular‐weight heparin versus aspirin in patients with acute ischaemic stroke and atrial fibrillation: a double‐blind randomised study. HAEST Study Group. Heparin in Acute Embolic Stroke Trial.Lancet.2000;355(9211):1205–1210.
- ,,,,.Endarterectomy for symptomatic carotid stenosis in relation to clinical subgroups and timing of surgery.Lancet.2004;363(9413):915–924.
- ,,.The timing of carotid endarterectomy post stroke.Neurol Clin.2006;24(4):669–680.
- Centers for Medicare and Medicaid Services (CMS). Department of Health and Human Services (DHHS). CMS Manual System. Pub 100–03 Medicare National Coverage Determinations. Available at: http://www.cms.hhs.gov/Transmittals/Downloads/R64NCD.pdf. Accessed May2009.
- .Current status of carotid endarterectomy and stenting for symptomatic carotid stenosis.Cerebrovasc Dis.2007;24(suppl 1):116–125.
- ,,, et al.Antiplatelets versus anticoagulation in cervical artery dissection.Stroke.2007;38(9):2605–2611.
- ,,, et al.A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke.N Engl J Med2001;345(20):1444–1451.
- .Warfarin or aspirin for recurrent ischemic stroke.N Engl J Med2002;346(15):1169–1171.
- ,,, et al.Practice parameter: recurrent stroke with patent foramen ovale and atrial septal aneurysm: report of the Quality Standards Subcommittee of the American Academy of Neurology.Neurology.2004;62(7):1042–1050.
- ,,, et al.In‐hospital initiation of secondary stroke prevention therapies yields high rates of adherence at follow‐up.Stroke.2004;35(12):2879–2883.
- Organised inpatient (stroke unit) care for stroke.Stroke Unit Trialists' Collaboration.Cochrane Database Syst Rev.2000(2):CD000197.
- ,,, et al.Acetaminophen for altering body temperature in acute stroke: a randomized clinical trial.Stroke.2002;33(1):130–134.
- ,,, et al.Fever in acute stroke worsens prognosis. A prospective study.Stroke.1995;26(11):2040–2043.
- ,.Combating hyperthermia in acute stroke: a significant clinical concern.Stroke.1998;29(2):529–534.
- ,,, et al.Body temperature in acute stroke: relation to stroke severity, infarct size, mortality, and outcome.Lancet. 171996;347(8999):422–425.
- ,,.Thresholds in cerebral ischemia—the ischemic penumbra.Stroke.1981;12(6):723–725.
- .Ischaemic brain damage of cerebral perfusion failure type after treatment of severe hypertension.Br Med J. 271975;4(5999):739.
- ,,,,.Imaging of acute stroke.Lancet Neurol.2006;5(9):755–768.
- ,,, et al.High‐dose atorvastatin after stroke or transient ischemic attack.N Engl J Med.2006;355(6):549–559.
- ,,, et al.Guidelines for prevention of stroke in patients with ischemic stroke or transient ischemic attack: a statement for healthcare professionals from the American Heart Association/American Stroke Association Council on Stroke: co‐sponsored by the Council on Cardiovascular Radiology and Intervention: the American Academy of Neurology affirms the value of this guideline.Stroke.2006;37(2):577–617.
- Antithrombotic Trialists' Collaboration.Collaborative meta‐analysis of randomised trials of antiplatelet therapy for prevention of death, myocardial infarction, and stroke in high risk patients.BMJ.2002;324(7329):71–86.
- ,,, et al.A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke.N Engl J Med.2001;345(20):1444–1451.
- .Warfarin or aspirin for recurrent ischemic stroke.N Engl J Med.2002;346(15):1169–1171.
- ,,, et al.Prevention. European Stroke Initiative.Cerebrovasc Dis.2004;17(suppl 2):15–29.
- ,,, et al.Update to the AHA/ASA recommendations for the prevention of stroke in patients with stroke and transient ischemic attack.Stroke.2008;39(5):1647–1652.
- ,,,,.Antithrombotic and thrombolytic therapy for ischemic stroke: American College of Chest Physicians Evidence‐Based Clinical Practice Guidelines (8th edition).Chest.2008;133(suppl):630S–669S.
- ,,, et al.Aspirin and extended‐release dipyridamole versus clopidogrel for recurrent stroke.N Engl J Med.2008;359(12):1238–1251.
- ,,, et al.Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events.N Engl J Med.2006;354(16):1706–1717.
- ,,, et al.Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high‐risk patients (MATCH): randomised, double‐blind, placebo‐controlled trial.Lancet.2004;364(9431):331–337.
- ,,, et al.Oral anticoagulants vs aspirin in nonvalvular atrial fibrillation: an individual patient meta‐analysis.JAMA.2002;288(19):2441–2448.
- .Effective anticoagulation therapy: defining the gap between clinical studies and clinical practice.Am J Manag Care.2004;10(suppl):S297–S306; discussionS312–S297.
- ,,,.Low molecular‐weight heparin versus aspirin in patients with acute ischaemic stroke and atrial fibrillation: a double‐blind randomised study. HAEST Study Group. Heparin in Acute Embolic Stroke Trial.Lancet.2000;355(9211):1205–1210.
- ,,,,.Endarterectomy for symptomatic carotid stenosis in relation to clinical subgroups and timing of surgery.Lancet.2004;363(9413):915–924.
- ,,.The timing of carotid endarterectomy post stroke.Neurol Clin.2006;24(4):669–680.
- Centers for Medicare and Medicaid Services (CMS). Department of Health and Human Services (DHHS). CMS Manual System. Pub 100–03 Medicare National Coverage Determinations. Available at: http://www.cms.hhs.gov/Transmittals/Downloads/R64NCD.pdf. Accessed May2009.
- .Current status of carotid endarterectomy and stenting for symptomatic carotid stenosis.Cerebrovasc Dis.2007;24(suppl 1):116–125.
- ,,, et al.Antiplatelets versus anticoagulation in cervical artery dissection.Stroke.2007;38(9):2605–2611.
- ,,, et al.A comparison of warfarin and aspirin for the prevention of recurrent ischemic stroke.N Engl J Med2001;345(20):1444–1451.
- .Warfarin or aspirin for recurrent ischemic stroke.N Engl J Med2002;346(15):1169–1171.
- ,,, et al.Practice parameter: recurrent stroke with patent foramen ovale and atrial septal aneurysm: report of the Quality Standards Subcommittee of the American Academy of Neurology.Neurology.2004;62(7):1042–1050.
- ,,, et al.In‐hospital initiation of secondary stroke prevention therapies yields high rates of adherence at follow‐up.Stroke.2004;35(12):2879–2883.
Gastric Involvement in NSG
Necrotizing sarcoid granulomatosis (NSG) is an immune system disorder characterized by necrotizing granulomas, as opposed to noncaseating granulomas in classical sarcoidosis. Over the past 3 decades there have been over 120 reported cases of NSG with pulmonary and extrapulmonary involvement.1 We present a patient found to have histological evidence of necrotizing granuloma in her gastric antrum, and we believe this is the first Case Report of NSG involving the stomach.
Case Report
A 21‐year‐old African‐American female first presented to an outside hospital with fever, epigastric pain, shortness of breath, and headache. Two days later she complained of nonbloody, nonbilious vomiting, and was found to have leukocytosis (17,770 cells/m3), elevated lipase (224 U/L), elevated C‐reactive protein (14.7 mg/L), and an inflamed pancreas on computed tomography (CT). She was treated conservatively for pancreatitis and started on ampicillin/sulbactam. After 2 contrast CT scans on consecutive days, she developed acute renal failure (creatinine 2.0 mg/dL compared to baseline of 1.0 mg/dL), and was transferred to our hospital for further evaluation and management.
Upon transfer, the patient's temperature was 37.0C, pulse was 102 beats/minute, blood pressure was 141/84 mm Hg, and oxygen saturation was 94%. On examination, she was tender to palpation in her epigastrium and right upper quadrant, but the remainder of the physical exam was unremarkable. She was started on moxifloxacin and managed with intravenous (IV) fluid hydration and pain control. Within 3 days, CT showed resolving pancreatitis, magnetic resonance cholangiopancreatography (MRCP) was negative, and her creatinine began normalizing (1.3 mg/dL). Nonetheless, she continued to complain of abdominal pain, shortness of breath, and intermittent low‐grade fevers. She then also developed bilateral panuveitis requiring high‐dose steroid eye drops.
Chest x‐ray showed subtle bilateral nodular and bronchiolitic infiltrates with no evidence of enlarged hilar nodes, and subsequent bronchoscopy showed no abnormalities. Additional workup included negative blood and urine cultures, purified protein derivative (PPD), and Clostridium difficile assay; as well as negative human immunodeficiency virus (HIV), cryptococcus, Helicobacter pylori, Borellia burgdorferi, syphilis (fluorescent treponemal antibody), aspergillus, histoplasma, and rheumatological serologies. Her white blood count (20,900 cells/m3), C‐reactive protein (6.6 mg/L), and erythrocyte sedimentation rate (100 mm/hour) remained elevated.
The patient continued to complain of epigastric pain. Repeat abdominal CT scan showed large retroperitoneal and mesenteric lymph nodes, and esophagogastroduodenoscopy (EGD) showed gastritis with an antral nodule (Figure 1). Biopsy of the nodule revealed a necrotizing granuloma with mixed cellular infiltrate. Biopsy stains were negative for bacteria, borellia, treponemes, acid‐fast bacilli, and fungi. The patient was diagnosed with necrotizing sarcoid granulomatosis and started on an oral prednisone taper. She responded to steroid treatment with prompt resolution of her uveitis, shortness of breath, abdominal pain, and fevers. She was discharged following treatment, has continued to do well, and is seen regularly at the sarcoid clinic for follow‐up.
Discussion
NSG was first characterized as a distinct variation from sarcoidosis by Liebow2 in 1973, and was noted to have 3 characteristic differences: (1) histological evidence of sarcoid‐appearing granuloma and necrosis, (2) pulmonary nodules without hilar lymphadenopathy on imaging, and (3) a clinically benign course. Over the past 3 decades, there have been more than 120 cases of reported NSG involving the lungs, gastrointestinal tract, kidney, skin, and central nervous system.1 Since Liebow's2 description, newly reported NSG cases have generally been found to be consistent with the aforementioned criteria, although hilar lymphadenopathy may be particularly more common than previously thought. One review noted a range of 8% to 79% prevalence of hilar lymphadenopathy in reported NSG series.3 Therefore, while hilar lymphadenopathy still currently appears to be less common in NSG than in typical sarcoidosis, its presence should not rule out the diagnosis.
Our patient's history is consistent with Liebow's2 criteria since she had histological evidence of necrotizing granuloma and pulmonary involvement without hilar lymphadenopathy, and responded promptly to steroid treatment. To our knowledge, this is the first case of NSG reported in the stomach.
While less than 1% of sarcoid patients are reported to have gastrointestinal involvement, all of these cases have either been noncaseating granulomas (classical sarcoidosis) or were found outside the stomach.1, 4, 5 Most of the data regarding sarcoid symptomatology and treatment are derived from reports on classical sarcoidosis. In classical sarcoidosis, there is gastric antral involvement in approximately 10% of patients with systemic disease.4 These patients may present with nausea, vomiting, and weight loss, and are often effectively treated with a single dose of prednisone 30 to 40 mg followed by a maintenance dose of 10 to 15 mg daily over 6 months.5 Less data are available regarding necrotizing sarcoid presentation and treatment, especially with regard to gastrointestinal involvement.
We hope to raise awareness regarding: (1) the variation in noncaseating versus necrotizing sarcoid‐type disorders, (2) the benefit of steroid treatment once infectious etiologies are ruled out, and (3) the potential for further extrapulmonary involvement in previously unreported organ systems.
- ,,.Necrotizing sarcoid granulomatosis mimicking an intracranial neoplasm: clinicopathologic features and review of the literature.Mod Pathol.2000;13(8)909–913.
- .Pulmonary angiitis and granulomatosis.Am J Respir Dis.1973;108:1–18.
- ,,,.Pulmonary angiitis and granulomatosis: radiologic‐pathologic correlation.Radiographics.1998;18(3):687–710.
- ,,, et al.Multiple antral ulcers in gastric sarcoid.J Clin Gastroenterol.1997;24(2):97–99.
- ,,.Gastric sarcoidosis: a case report and review of the literature.South Med J.2007;100(3):301–303.
Necrotizing sarcoid granulomatosis (NSG) is an immune system disorder characterized by necrotizing granulomas, as opposed to noncaseating granulomas in classical sarcoidosis. Over the past 3 decades there have been over 120 reported cases of NSG with pulmonary and extrapulmonary involvement.1 We present a patient found to have histological evidence of necrotizing granuloma in her gastric antrum, and we believe this is the first Case Report of NSG involving the stomach.
Case Report
A 21‐year‐old African‐American female first presented to an outside hospital with fever, epigastric pain, shortness of breath, and headache. Two days later she complained of nonbloody, nonbilious vomiting, and was found to have leukocytosis (17,770 cells/m3), elevated lipase (224 U/L), elevated C‐reactive protein (14.7 mg/L), and an inflamed pancreas on computed tomography (CT). She was treated conservatively for pancreatitis and started on ampicillin/sulbactam. After 2 contrast CT scans on consecutive days, she developed acute renal failure (creatinine 2.0 mg/dL compared to baseline of 1.0 mg/dL), and was transferred to our hospital for further evaluation and management.
Upon transfer, the patient's temperature was 37.0C, pulse was 102 beats/minute, blood pressure was 141/84 mm Hg, and oxygen saturation was 94%. On examination, she was tender to palpation in her epigastrium and right upper quadrant, but the remainder of the physical exam was unremarkable. She was started on moxifloxacin and managed with intravenous (IV) fluid hydration and pain control. Within 3 days, CT showed resolving pancreatitis, magnetic resonance cholangiopancreatography (MRCP) was negative, and her creatinine began normalizing (1.3 mg/dL). Nonetheless, she continued to complain of abdominal pain, shortness of breath, and intermittent low‐grade fevers. She then also developed bilateral panuveitis requiring high‐dose steroid eye drops.
Chest x‐ray showed subtle bilateral nodular and bronchiolitic infiltrates with no evidence of enlarged hilar nodes, and subsequent bronchoscopy showed no abnormalities. Additional workup included negative blood and urine cultures, purified protein derivative (PPD), and Clostridium difficile assay; as well as negative human immunodeficiency virus (HIV), cryptococcus, Helicobacter pylori, Borellia burgdorferi, syphilis (fluorescent treponemal antibody), aspergillus, histoplasma, and rheumatological serologies. Her white blood count (20,900 cells/m3), C‐reactive protein (6.6 mg/L), and erythrocyte sedimentation rate (100 mm/hour) remained elevated.
The patient continued to complain of epigastric pain. Repeat abdominal CT scan showed large retroperitoneal and mesenteric lymph nodes, and esophagogastroduodenoscopy (EGD) showed gastritis with an antral nodule (Figure 1). Biopsy of the nodule revealed a necrotizing granuloma with mixed cellular infiltrate. Biopsy stains were negative for bacteria, borellia, treponemes, acid‐fast bacilli, and fungi. The patient was diagnosed with necrotizing sarcoid granulomatosis and started on an oral prednisone taper. She responded to steroid treatment with prompt resolution of her uveitis, shortness of breath, abdominal pain, and fevers. She was discharged following treatment, has continued to do well, and is seen regularly at the sarcoid clinic for follow‐up.

Discussion
NSG was first characterized as a distinct variation from sarcoidosis by Liebow2 in 1973, and was noted to have 3 characteristic differences: (1) histological evidence of sarcoid‐appearing granuloma and necrosis, (2) pulmonary nodules without hilar lymphadenopathy on imaging, and (3) a clinically benign course. Over the past 3 decades, there have been more than 120 cases of reported NSG involving the lungs, gastrointestinal tract, kidney, skin, and central nervous system.1 Since Liebow's2 description, newly reported NSG cases have generally been found to be consistent with the aforementioned criteria, although hilar lymphadenopathy may be particularly more common than previously thought. One review noted a range of 8% to 79% prevalence of hilar lymphadenopathy in reported NSG series.3 Therefore, while hilar lymphadenopathy still currently appears to be less common in NSG than in typical sarcoidosis, its presence should not rule out the diagnosis.
Our patient's history is consistent with Liebow's2 criteria since she had histological evidence of necrotizing granuloma and pulmonary involvement without hilar lymphadenopathy, and responded promptly to steroid treatment. To our knowledge, this is the first case of NSG reported in the stomach.
While less than 1% of sarcoid patients are reported to have gastrointestinal involvement, all of these cases have either been noncaseating granulomas (classical sarcoidosis) or were found outside the stomach.1, 4, 5 Most of the data regarding sarcoid symptomatology and treatment are derived from reports on classical sarcoidosis. In classical sarcoidosis, there is gastric antral involvement in approximately 10% of patients with systemic disease.4 These patients may present with nausea, vomiting, and weight loss, and are often effectively treated with a single dose of prednisone 30 to 40 mg followed by a maintenance dose of 10 to 15 mg daily over 6 months.5 Less data are available regarding necrotizing sarcoid presentation and treatment, especially with regard to gastrointestinal involvement.
We hope to raise awareness regarding: (1) the variation in noncaseating versus necrotizing sarcoid‐type disorders, (2) the benefit of steroid treatment once infectious etiologies are ruled out, and (3) the potential for further extrapulmonary involvement in previously unreported organ systems.
Necrotizing sarcoid granulomatosis (NSG) is an immune system disorder characterized by necrotizing granulomas, as opposed to noncaseating granulomas in classical sarcoidosis. Over the past 3 decades there have been over 120 reported cases of NSG with pulmonary and extrapulmonary involvement.1 We present a patient found to have histological evidence of necrotizing granuloma in her gastric antrum, and we believe this is the first Case Report of NSG involving the stomach.
Case Report
A 21‐year‐old African‐American female first presented to an outside hospital with fever, epigastric pain, shortness of breath, and headache. Two days later she complained of nonbloody, nonbilious vomiting, and was found to have leukocytosis (17,770 cells/m3), elevated lipase (224 U/L), elevated C‐reactive protein (14.7 mg/L), and an inflamed pancreas on computed tomography (CT). She was treated conservatively for pancreatitis and started on ampicillin/sulbactam. After 2 contrast CT scans on consecutive days, she developed acute renal failure (creatinine 2.0 mg/dL compared to baseline of 1.0 mg/dL), and was transferred to our hospital for further evaluation and management.
Upon transfer, the patient's temperature was 37.0C, pulse was 102 beats/minute, blood pressure was 141/84 mm Hg, and oxygen saturation was 94%. On examination, she was tender to palpation in her epigastrium and right upper quadrant, but the remainder of the physical exam was unremarkable. She was started on moxifloxacin and managed with intravenous (IV) fluid hydration and pain control. Within 3 days, CT showed resolving pancreatitis, magnetic resonance cholangiopancreatography (MRCP) was negative, and her creatinine began normalizing (1.3 mg/dL). Nonetheless, she continued to complain of abdominal pain, shortness of breath, and intermittent low‐grade fevers. She then also developed bilateral panuveitis requiring high‐dose steroid eye drops.
Chest x‐ray showed subtle bilateral nodular and bronchiolitic infiltrates with no evidence of enlarged hilar nodes, and subsequent bronchoscopy showed no abnormalities. Additional workup included negative blood and urine cultures, purified protein derivative (PPD), and Clostridium difficile assay; as well as negative human immunodeficiency virus (HIV), cryptococcus, Helicobacter pylori, Borellia burgdorferi, syphilis (fluorescent treponemal antibody), aspergillus, histoplasma, and rheumatological serologies. Her white blood count (20,900 cells/m3), C‐reactive protein (6.6 mg/L), and erythrocyte sedimentation rate (100 mm/hour) remained elevated.
The patient continued to complain of epigastric pain. Repeat abdominal CT scan showed large retroperitoneal and mesenteric lymph nodes, and esophagogastroduodenoscopy (EGD) showed gastritis with an antral nodule (Figure 1). Biopsy of the nodule revealed a necrotizing granuloma with mixed cellular infiltrate. Biopsy stains were negative for bacteria, borellia, treponemes, acid‐fast bacilli, and fungi. The patient was diagnosed with necrotizing sarcoid granulomatosis and started on an oral prednisone taper. She responded to steroid treatment with prompt resolution of her uveitis, shortness of breath, abdominal pain, and fevers. She was discharged following treatment, has continued to do well, and is seen regularly at the sarcoid clinic for follow‐up.

Discussion
NSG was first characterized as a distinct variation from sarcoidosis by Liebow2 in 1973, and was noted to have 3 characteristic differences: (1) histological evidence of sarcoid‐appearing granuloma and necrosis, (2) pulmonary nodules without hilar lymphadenopathy on imaging, and (3) a clinically benign course. Over the past 3 decades, there have been more than 120 cases of reported NSG involving the lungs, gastrointestinal tract, kidney, skin, and central nervous system.1 Since Liebow's2 description, newly reported NSG cases have generally been found to be consistent with the aforementioned criteria, although hilar lymphadenopathy may be particularly more common than previously thought. One review noted a range of 8% to 79% prevalence of hilar lymphadenopathy in reported NSG series.3 Therefore, while hilar lymphadenopathy still currently appears to be less common in NSG than in typical sarcoidosis, its presence should not rule out the diagnosis.
Our patient's history is consistent with Liebow's2 criteria since she had histological evidence of necrotizing granuloma and pulmonary involvement without hilar lymphadenopathy, and responded promptly to steroid treatment. To our knowledge, this is the first case of NSG reported in the stomach.
While less than 1% of sarcoid patients are reported to have gastrointestinal involvement, all of these cases have either been noncaseating granulomas (classical sarcoidosis) or were found outside the stomach.1, 4, 5 Most of the data regarding sarcoid symptomatology and treatment are derived from reports on classical sarcoidosis. In classical sarcoidosis, there is gastric antral involvement in approximately 10% of patients with systemic disease.4 These patients may present with nausea, vomiting, and weight loss, and are often effectively treated with a single dose of prednisone 30 to 40 mg followed by a maintenance dose of 10 to 15 mg daily over 6 months.5 Less data are available regarding necrotizing sarcoid presentation and treatment, especially with regard to gastrointestinal involvement.
We hope to raise awareness regarding: (1) the variation in noncaseating versus necrotizing sarcoid‐type disorders, (2) the benefit of steroid treatment once infectious etiologies are ruled out, and (3) the potential for further extrapulmonary involvement in previously unreported organ systems.
- ,,.Necrotizing sarcoid granulomatosis mimicking an intracranial neoplasm: clinicopathologic features and review of the literature.Mod Pathol.2000;13(8)909–913.
- .Pulmonary angiitis and granulomatosis.Am J Respir Dis.1973;108:1–18.
- ,,,.Pulmonary angiitis and granulomatosis: radiologic‐pathologic correlation.Radiographics.1998;18(3):687–710.
- ,,, et al.Multiple antral ulcers in gastric sarcoid.J Clin Gastroenterol.1997;24(2):97–99.
- ,,.Gastric sarcoidosis: a case report and review of the literature.South Med J.2007;100(3):301–303.
- ,,.Necrotizing sarcoid granulomatosis mimicking an intracranial neoplasm: clinicopathologic features and review of the literature.Mod Pathol.2000;13(8)909–913.
- .Pulmonary angiitis and granulomatosis.Am J Respir Dis.1973;108:1–18.
- ,,,.Pulmonary angiitis and granulomatosis: radiologic‐pathologic correlation.Radiographics.1998;18(3):687–710.
- ,,, et al.Multiple antral ulcers in gastric sarcoid.J Clin Gastroenterol.1997;24(2):97–99.
- ,,.Gastric sarcoidosis: a case report and review of the literature.South Med J.2007;100(3):301–303.
Nonphysicians in Hospital Medicine
Ford and Britting's1 editorial in this month's Journal of Hospital Medicine raises important questions concerning the use of nonphysician providers in hospital medicine. They focus primarily on the use of mid‐level providers (MLPs), namely physician‐assistants (PAs) and nurse practitioners (NPs), as a potential solution to the current physician workforce shortages in our field. While we acknowledge the challenges of meeting workforce needs, we also believe that much is unknown about the optimal use of MLPs on inpatient general medicine services and it is premature to tout MLPs as the solution to hospital medicine staffing problems. This is especially true in those hospitals where hospitalists care for complex, general medical patients with a wide variety of medical conditions, a trend that is especially common in academic medical centers.2
This article discusses the current literature, our own experiences with MLPs, and suggests some future initiatives that might help better integrate MLPs into hospital medicine.
The Literature on MLPs in Inpatient Venues
The existing literature on the use of MLPs in inpatient venues is quite limited, and a recent review, while suggesting that the existing literature does describe benefits of MLPs in the inpatient setting, also states that the overall quality of the evidence is quite poor and that many studies suffer from significant limitations, including small populations, limited patient mixes, use of selected settings, and short durations of outcome assessment.3
Ford and Britting,1 in their article, cite several studies46 as evidence that a MLP model of care either improved outcomes or provided cost benefits. Each of these studies has important limitations that are worth examining.
The study by Myers et al.4 described the use of MLPs in a chest pain unit. NPs partnered with hospitalists to care for a low‐acuity chest pain population. In addition, 5 NPs only staffed the unit during daytime weekday hours. Off‐hour and weekend staffing was accomplished through the use of resident physicians. Notably, the work suggests the service only admitted 113 low‐risk patients over 10 months. The service was staffed by 3 full‐time equivalent (FTE) NPs in addition to involving hospitalists during the day. It is not surprising, given the extremely low volume of patients coupled with a daytime‐only focus, that this service showed efficiency gains. In addition, given the service was only staffed by NPs 40 hours a week and by resident physicians on nights and weekends, the true cost of such an intervention needs to take into account the full cost of 24/7 coverage. In addition, the model of using residents to cover nonteaching patients is no longer permitted by the current Accreditation Council for Graduate Medical Education (ACGME) Internal Medicine Residency Requirements7 and thus implementation of a model such as this in 2009 would require alternative means of nighttime coverage.
The study by Nishimura et al.,5 also describing the use of MLPs in cardiovascular care, has important caveats that make full assessment of the model impossible. The model describes the implementation of a care team consisting of an attending, a fellow, and MLPs to replace a traditional teaching team of an attending, senior resident, and 2 interns. The study states that the model resulted in a lower length of stay (LOS) and lower costs per case. Importantly, the new MLP‐based team only admitted during the hours of 7 AM to 2 PM. The study does not fully describe the number of MLPs required nor does it fully describe the role of cardiovascular fellows in the model. The study does state that the cost savings offset the cost of the MLPs but it is not clear if this cost analysis took into account the cost of the fellow's daytime involvement or if it measured attending time required before and after the implementation of the new model. In addition, this model presumes the availability of other services to admit patients during afternoon and nighttime hours and so may not be generalizable to other settings.
The final study by Cowan et al.6 describes the addition of a NP, a hospitalist medical director, and daily multidisciplinary rounds to a traditional teaching service model. Importantly, the NP was not involved in the admission process nor were they the primary providers for day‐to‐day medical care but rather they focused on implementation of care protocols, multidisciplinary coordination of care and discharge planning, and postdischarge follow‐up. In addition, the NP worked only weekdays for about 40 hours a week. It is not surprising that adding multiple additional resources to existing care models might provide benefits but this does not address any issues in terms of the workforce since the care in this model required a higher total input of providers than the usual care model being studied. Cost savings from such a model may make it cost‐effective but it does not represent a workforce solution.
There have been other studies examining the use of MLPs in the inpatient setting in internal medicine. Some of these studies have suggested that MLP‐based models result in equivalent outcomes and efficiency810 to traditional teaching or nonteaching physician‐only models. There are 2 important caveats, however, that must be considered. The total resources required for such models may be quite high, especially taking into account the costs of 24/7 coverage and physician backup of the MLPs, and most importantly there is almost no literature that robustly examines ultimate clinical outcomes in these models. We do note that a recent study11 did show a lower inpatient mortality rate over a 2‐year period of time after substituting a PA‐hospitalist model for a traditional academic medicine residency model in a community hospital. Importantly, however, the new model also added 24/7 hospitalist physicians and night and weekend intensivists that were not present in the prior residency‐based model. Thus, the lower mortality rate could be attributed to the addition of hospitalists or the more robust in‐house physician coverage during off‐hours rather than the use of MLPs.
Notably, while the evidence base in internal medicine is not robust, many studies have described successful use of MLPs in non‐internal medicine inpatient settings.1214 The reasons for this success is debatable, but it may be that MLPs are more successful in settings where the care is either more protocol‐driven or where there is less diagnostic and therapeutic complexity.
Recent Experiences with MLPs in Academic Hospital Medicine
Given the paucity of data, it is clear that further research is needed on the role of MLPs in hospital medicine. While waiting for such evidence to appear, it may be worthwhile to reflect on the recent experience of 3 major medical centers. A recent article described 5 hospitalist models at major academic medical centers across the country. Two of the institutions described at the time (University of Michigan Health System, Ann Arbor, MI; and Brigham and Women's Hospital, Boston, MA) utilized MLPs as a major element of their staffing of nonresident hospitalist services while another (University of California, San Francisco [UCSF] Medical Center at Mt. Zion, San Francisco, CA) had previously used MLPs as part of its model but phased them out about 1 year prior to publication of the article.2 The model used by the Brigham and Women's Hospital was later described in more detail in a subsequent publication.8 Recently 1 of these institutions (Michigan) has chosen to phase out MLPs. At Michigan, a 4‐year experience with PAs on a general‐medicine focused hospitalist service eventually led to the conclusion that continued use of PAs was not cost‐effective. Significant barriers to success included a steep learning curve and the significant time required before PAs developed sufficient autonomy and efficiency in caring for a highly complex heterogeneous patient population. In the Michigan experience, PAs took up to 2 years to attain a significant level of autonomy and efficiency and even then some PAs still required a significant amount of physician oversight. Similar concerns at UCSF Mt. Zion led to the elimination of their MLP program as well. At Brigham and Women's, the MLP service continues but has required additional hospitalist staffing due to difficulties recruiting qualified MLPs with appropriate inpatient experience. In all cases, the models were challenged by high costs and the difficulty of developing MLPs to attain the level of autonomy and efficiency needed to justify their continued use. A key point is that in each institution, MLPs continue to play an important role in some specialty inpatient areas such as Hematology/Oncology and Bone Marrow Transplant, which is where MLPs have traditionally found their niche in inpatient Internal Medicine. These focus shops allow MLPs to develop a niche and expertise in a specialized area, where they may become more autonomous and efficient than house staff. Thus these settings may be more appropriate for MLPs than a heterogeneous general medicine inpatient setting.
Reviewing the Financial Case
In their article, Ford and Britting1 cite potential financial advantages for the use of MLPs in hospital medicine by comparing the relative salaries of MLPs to Hospitalists. What was missing in their analysis was the relative productivity of the 2 types of providers. We do have some limited data from the Society of Hospital Medicine (SHM) annual survey that looks at MLPs in hospital medicine but, again, the number of respondents for most data elements is less than 70, making generalizability difficult. Nonetheless, the data suggest that MLPs in hospital medicine average about 60% to 75% of the productivity of a physician when measured by encounters, although there is wide variability depending on the employment model (academic vs. multispecialty group).15 Importantly, the existing data do not provide any measure of how much physician input is provided to these MLPs but we suspect that in most models there is some physician time and input. If we presume that the MLPs bill independently and collect 85% of the physician fee schedule for a Medicare population, then collections would be about 50% to 65% of a typical physician. Given that median total compensation including benefits from the SHM survey was $120,000 for MLPs and $216,000 for physiciansabout a 55% ratiothis would argue for potential financial neutrality when substituting MLPs for physicians in a 2:1 ratio but only if we presume they require no physician supervision, which in our own experience is not likely in a general medicine population. In an alternative model, in which the physician sees every patient with the MLP and the physician bills, one would need to see roughly 50% more patients to achieve a financially neutral situation. In our experience at our own institutions, this level of increased productivity was not achievable. It is important to note that our figures are median compensation and benefit cost figures and local markets vary widely. We know that in major east and west coast cities MLPs may command far higher salaries while early career hospitalist physicians may be paid somewhat less than the reported medians. Recent market changes have significantly pressured MLP salaries,15, 16 further impacting the financial equation and perhaps tilting it farther against a financial benefit for MLPs. Furthermore, night coverage for MLP services should always be considered in a financial analysis and is not captured in this simple analysis.
Next Steps
Given the current shortage of physicians, we imagine that many hospitalist groups will consider the use of MLPs as a solution to the current workforce issues. However, data on how best to utilize MLPs and the true impact on both the cost and quality of such models is lacking. In addition to urging increased publication and dissemination of existing experiences with NP and PAs, we strongly suggest that groups considering starting a MLP model do so in a way which would facilitate robust analysis and comparison of the model with alternatives. We also suggest that SHM consider the following: modifying its biennial survey to better capture the nuances of MLP productivity (such as assessing the amount of physician input and supervision required); targeting MLPs so as to increase the number of respondents; and doing an additional survey to capture demographics and basic data on existing MLP models given the lack of published literature.
In addition to gathering more data on effective models, a critical gap that we have identified is the development of models for the training and development of MLPs interested in hospital medicine. It would be a mistake to believe that MLPs could function in a manner similar to residency‐trained physicians if they do not undergo similar training. NP/PA programs generally do not have a significant inpatient internal medicine focus and so newly minted graduates often lack the skills needed to succeed in hospital medicine.17 Some hospitalist programs train their MLPs on the job, but many programs cannot afford the amount of time and effort required to do this on their own. There are a small number of advanced training options for MLPs in hospital medicine18 but it is not likely such models will proliferate given the inherent opportunity costs that exist for extended training in the current competitive job market for MLPs. Instead we think that very motivated hospital medicine groups may develop training relationships with PA and NP schools in an effort to train their own. In addition, national initiatives such as the Hospital Medicine Boot Camp for NPs and PAs, which is cosponsored by SHM, the American Association of Physician Assistants (AAPA), and the American Academy of Nurse Practitioners (AANP),19 can help fill the educational needs for MLPs who are already in practice.
Conclusions
While some literature exists that suggests that MLPs can successfully be used in the inpatient internal medicine setting, it is important to note that the evidence is quite limited and cannot be generalized across all care settings and patient populations. There is an urgent need to gather more data and share our collective experiences to better inform our decision‐making before we state that MLPs are the solution to workforce shortages in hospital medicine. In addition, existing data and experience suggest that MLPs may not be a cost‐effective workforce solution for complex general medical patients who require significant physician input. We believe that redesigning the clinical training of MLPs to focus on inpatient skills may hold promise and encourage interested parties to consider developing partnerships with MLP training programs and hospital medicine groups, as a way to build a more robust and successful hospital medicine MLP workforce.
- ,.Nonphysician providers in the hospitalist model: a prescription for change and a warning about unintended side effects.J Hosp Med.2010;5:99–102.
- ,,,,.Non‐housestaff medicine services in academic medical centers: models and challenges.J Hosp Med.2008;3:247–255.
- ,,.Nurse practitioners and physician assistants in the intensive care unit: an evidence‐based review.Crit Care Med.2008;36:2888–2897.
- ,,.Improving resource utilization in a teaching hospital: development of a nonteaching service for chest pain admissions.Acad Med.2006;81:432–435.
- ,,,,,.A nonresident cardiovascular inpatient service improves residents' experiences in an academic medical center: a new model to meet the challenges of the new millennium.Acad Med.2004;79;426–431.
- .The effect of a multidisciplinary hospitalist/physician and advance practice nurse collaboration on hospital care.J Nurs Adm.2006;36:79–85.
- Accreditation Council for Graduate Medical Education. ACGME Program Requirements for Residency Education in Internal Medicine. Available at: http://www.acgme.org/acWebsite/downloads/RRC_progReq/140_internal_ medicine_07012009.pdf. Accessed July2009.
- ,,, et al.Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes.J Hosp Med.2008;3:361–368.
- ,,.Resource use by physician assistant services versus teaching services.JAAPA.2002;15:33–38.
- ,,,, et al.Outcomes‐based trial of an inpatient nurse practitioner service for general medical patients.J Eval Clin Pract.2001;7:21–33.
- ,.Replacing an academic internal medicine residency program with a physician assistant‐hospitalist model: a comparative analysis study.Am J Med Qual.2009;2:132–139.
- ,,,,.Integrating midlevel practitioners into a teaching service.Am J Surg.2006;1:119–124.
- ,,, et al.Physician extenders impact trauma systems.J Trauma.2005;58(5):917–920.
- ,.Physician assistants in cardiothoracic surgery: a 30‐year experience in a university center.Ann Thorac Surg.2006;1:195–199.
- 2007–2008 Society of Hospital Medicine Bi‐Annual Survey: the Authoritative Source on the State of the Hospital Medicine Movement.Philadelphia:Society of Hospital Medicine;2008.
- American Association of Physician Assistants. Physician Assistant Income. Available at: http://www.aapa.org/images/stories/iu08incchange. pdf. Accessed July2009.
- Accreditation Review Commission on Education for the Physician Assistant. Accreditation Standards for Physician Assistant Education, 3rd ed. Available at: http://www.arcpa.org/Standards/3rdeditionwithPDchangesandregionals4.24.08a.pdf. Accessed July2009.
- Association of Postgraduate PA Programs. Postgraduate PA Program Listing by State. Available at: http://www.appap.org/index1.html. Accessed July2009.
- American Association of Physician Assistants. Adult Hospitalist Physician Assistant and Nurse Practitioner Boot Camp. Available at: http://www. aapa.org/component/content/article/23‐‐general‐/673‐adult‐hospitalist‐physician‐assistant‐and‐nurse‐practitioner‐boot‐camp. Accessed July2009.
Ford and Britting's1 editorial in this month's Journal of Hospital Medicine raises important questions concerning the use of nonphysician providers in hospital medicine. They focus primarily on the use of mid‐level providers (MLPs), namely physician‐assistants (PAs) and nurse practitioners (NPs), as a potential solution to the current physician workforce shortages in our field. While we acknowledge the challenges of meeting workforce needs, we also believe that much is unknown about the optimal use of MLPs on inpatient general medicine services and it is premature to tout MLPs as the solution to hospital medicine staffing problems. This is especially true in those hospitals where hospitalists care for complex, general medical patients with a wide variety of medical conditions, a trend that is especially common in academic medical centers.2
This article discusses the current literature, our own experiences with MLPs, and suggests some future initiatives that might help better integrate MLPs into hospital medicine.
The Literature on MLPs in Inpatient Venues
The existing literature on the use of MLPs in inpatient venues is quite limited, and a recent review, while suggesting that the existing literature does describe benefits of MLPs in the inpatient setting, also states that the overall quality of the evidence is quite poor and that many studies suffer from significant limitations, including small populations, limited patient mixes, use of selected settings, and short durations of outcome assessment.3
Ford and Britting,1 in their article, cite several studies46 as evidence that a MLP model of care either improved outcomes or provided cost benefits. Each of these studies has important limitations that are worth examining.
The study by Myers et al.4 described the use of MLPs in a chest pain unit. NPs partnered with hospitalists to care for a low‐acuity chest pain population. In addition, 5 NPs only staffed the unit during daytime weekday hours. Off‐hour and weekend staffing was accomplished through the use of resident physicians. Notably, the work suggests the service only admitted 113 low‐risk patients over 10 months. The service was staffed by 3 full‐time equivalent (FTE) NPs in addition to involving hospitalists during the day. It is not surprising, given the extremely low volume of patients coupled with a daytime‐only focus, that this service showed efficiency gains. In addition, given the service was only staffed by NPs 40 hours a week and by resident physicians on nights and weekends, the true cost of such an intervention needs to take into account the full cost of 24/7 coverage. In addition, the model of using residents to cover nonteaching patients is no longer permitted by the current Accreditation Council for Graduate Medical Education (ACGME) Internal Medicine Residency Requirements7 and thus implementation of a model such as this in 2009 would require alternative means of nighttime coverage.
The study by Nishimura et al.,5 also describing the use of MLPs in cardiovascular care, has important caveats that make full assessment of the model impossible. The model describes the implementation of a care team consisting of an attending, a fellow, and MLPs to replace a traditional teaching team of an attending, senior resident, and 2 interns. The study states that the model resulted in a lower length of stay (LOS) and lower costs per case. Importantly, the new MLP‐based team only admitted during the hours of 7 AM to 2 PM. The study does not fully describe the number of MLPs required nor does it fully describe the role of cardiovascular fellows in the model. The study does state that the cost savings offset the cost of the MLPs but it is not clear if this cost analysis took into account the cost of the fellow's daytime involvement or if it measured attending time required before and after the implementation of the new model. In addition, this model presumes the availability of other services to admit patients during afternoon and nighttime hours and so may not be generalizable to other settings.
The final study by Cowan et al.6 describes the addition of a NP, a hospitalist medical director, and daily multidisciplinary rounds to a traditional teaching service model. Importantly, the NP was not involved in the admission process nor were they the primary providers for day‐to‐day medical care but rather they focused on implementation of care protocols, multidisciplinary coordination of care and discharge planning, and postdischarge follow‐up. In addition, the NP worked only weekdays for about 40 hours a week. It is not surprising that adding multiple additional resources to existing care models might provide benefits but this does not address any issues in terms of the workforce since the care in this model required a higher total input of providers than the usual care model being studied. Cost savings from such a model may make it cost‐effective but it does not represent a workforce solution.
There have been other studies examining the use of MLPs in the inpatient setting in internal medicine. Some of these studies have suggested that MLP‐based models result in equivalent outcomes and efficiency810 to traditional teaching or nonteaching physician‐only models. There are 2 important caveats, however, that must be considered. The total resources required for such models may be quite high, especially taking into account the costs of 24/7 coverage and physician backup of the MLPs, and most importantly there is almost no literature that robustly examines ultimate clinical outcomes in these models. We do note that a recent study11 did show a lower inpatient mortality rate over a 2‐year period of time after substituting a PA‐hospitalist model for a traditional academic medicine residency model in a community hospital. Importantly, however, the new model also added 24/7 hospitalist physicians and night and weekend intensivists that were not present in the prior residency‐based model. Thus, the lower mortality rate could be attributed to the addition of hospitalists or the more robust in‐house physician coverage during off‐hours rather than the use of MLPs.
Notably, while the evidence base in internal medicine is not robust, many studies have described successful use of MLPs in non‐internal medicine inpatient settings.1214 The reasons for this success is debatable, but it may be that MLPs are more successful in settings where the care is either more protocol‐driven or where there is less diagnostic and therapeutic complexity.
Recent Experiences with MLPs in Academic Hospital Medicine
Given the paucity of data, it is clear that further research is needed on the role of MLPs in hospital medicine. While waiting for such evidence to appear, it may be worthwhile to reflect on the recent experience of 3 major medical centers. A recent article described 5 hospitalist models at major academic medical centers across the country. Two of the institutions described at the time (University of Michigan Health System, Ann Arbor, MI; and Brigham and Women's Hospital, Boston, MA) utilized MLPs as a major element of their staffing of nonresident hospitalist services while another (University of California, San Francisco [UCSF] Medical Center at Mt. Zion, San Francisco, CA) had previously used MLPs as part of its model but phased them out about 1 year prior to publication of the article.2 The model used by the Brigham and Women's Hospital was later described in more detail in a subsequent publication.8 Recently 1 of these institutions (Michigan) has chosen to phase out MLPs. At Michigan, a 4‐year experience with PAs on a general‐medicine focused hospitalist service eventually led to the conclusion that continued use of PAs was not cost‐effective. Significant barriers to success included a steep learning curve and the significant time required before PAs developed sufficient autonomy and efficiency in caring for a highly complex heterogeneous patient population. In the Michigan experience, PAs took up to 2 years to attain a significant level of autonomy and efficiency and even then some PAs still required a significant amount of physician oversight. Similar concerns at UCSF Mt. Zion led to the elimination of their MLP program as well. At Brigham and Women's, the MLP service continues but has required additional hospitalist staffing due to difficulties recruiting qualified MLPs with appropriate inpatient experience. In all cases, the models were challenged by high costs and the difficulty of developing MLPs to attain the level of autonomy and efficiency needed to justify their continued use. A key point is that in each institution, MLPs continue to play an important role in some specialty inpatient areas such as Hematology/Oncology and Bone Marrow Transplant, which is where MLPs have traditionally found their niche in inpatient Internal Medicine. These focus shops allow MLPs to develop a niche and expertise in a specialized area, where they may become more autonomous and efficient than house staff. Thus these settings may be more appropriate for MLPs than a heterogeneous general medicine inpatient setting.
Reviewing the Financial Case
In their article, Ford and Britting1 cite potential financial advantages for the use of MLPs in hospital medicine by comparing the relative salaries of MLPs to Hospitalists. What was missing in their analysis was the relative productivity of the 2 types of providers. We do have some limited data from the Society of Hospital Medicine (SHM) annual survey that looks at MLPs in hospital medicine but, again, the number of respondents for most data elements is less than 70, making generalizability difficult. Nonetheless, the data suggest that MLPs in hospital medicine average about 60% to 75% of the productivity of a physician when measured by encounters, although there is wide variability depending on the employment model (academic vs. multispecialty group).15 Importantly, the existing data do not provide any measure of how much physician input is provided to these MLPs but we suspect that in most models there is some physician time and input. If we presume that the MLPs bill independently and collect 85% of the physician fee schedule for a Medicare population, then collections would be about 50% to 65% of a typical physician. Given that median total compensation including benefits from the SHM survey was $120,000 for MLPs and $216,000 for physiciansabout a 55% ratiothis would argue for potential financial neutrality when substituting MLPs for physicians in a 2:1 ratio but only if we presume they require no physician supervision, which in our own experience is not likely in a general medicine population. In an alternative model, in which the physician sees every patient with the MLP and the physician bills, one would need to see roughly 50% more patients to achieve a financially neutral situation. In our experience at our own institutions, this level of increased productivity was not achievable. It is important to note that our figures are median compensation and benefit cost figures and local markets vary widely. We know that in major east and west coast cities MLPs may command far higher salaries while early career hospitalist physicians may be paid somewhat less than the reported medians. Recent market changes have significantly pressured MLP salaries,15, 16 further impacting the financial equation and perhaps tilting it farther against a financial benefit for MLPs. Furthermore, night coverage for MLP services should always be considered in a financial analysis and is not captured in this simple analysis.
Next Steps
Given the current shortage of physicians, we imagine that many hospitalist groups will consider the use of MLPs as a solution to the current workforce issues. However, data on how best to utilize MLPs and the true impact on both the cost and quality of such models is lacking. In addition to urging increased publication and dissemination of existing experiences with NP and PAs, we strongly suggest that groups considering starting a MLP model do so in a way which would facilitate robust analysis and comparison of the model with alternatives. We also suggest that SHM consider the following: modifying its biennial survey to better capture the nuances of MLP productivity (such as assessing the amount of physician input and supervision required); targeting MLPs so as to increase the number of respondents; and doing an additional survey to capture demographics and basic data on existing MLP models given the lack of published literature.
In addition to gathering more data on effective models, a critical gap that we have identified is the development of models for the training and development of MLPs interested in hospital medicine. It would be a mistake to believe that MLPs could function in a manner similar to residency‐trained physicians if they do not undergo similar training. NP/PA programs generally do not have a significant inpatient internal medicine focus and so newly minted graduates often lack the skills needed to succeed in hospital medicine.17 Some hospitalist programs train their MLPs on the job, but many programs cannot afford the amount of time and effort required to do this on their own. There are a small number of advanced training options for MLPs in hospital medicine18 but it is not likely such models will proliferate given the inherent opportunity costs that exist for extended training in the current competitive job market for MLPs. Instead we think that very motivated hospital medicine groups may develop training relationships with PA and NP schools in an effort to train their own. In addition, national initiatives such as the Hospital Medicine Boot Camp for NPs and PAs, which is cosponsored by SHM, the American Association of Physician Assistants (AAPA), and the American Academy of Nurse Practitioners (AANP),19 can help fill the educational needs for MLPs who are already in practice.
Conclusions
While some literature exists that suggests that MLPs can successfully be used in the inpatient internal medicine setting, it is important to note that the evidence is quite limited and cannot be generalized across all care settings and patient populations. There is an urgent need to gather more data and share our collective experiences to better inform our decision‐making before we state that MLPs are the solution to workforce shortages in hospital medicine. In addition, existing data and experience suggest that MLPs may not be a cost‐effective workforce solution for complex general medical patients who require significant physician input. We believe that redesigning the clinical training of MLPs to focus on inpatient skills may hold promise and encourage interested parties to consider developing partnerships with MLP training programs and hospital medicine groups, as a way to build a more robust and successful hospital medicine MLP workforce.
Ford and Britting's1 editorial in this month's Journal of Hospital Medicine raises important questions concerning the use of nonphysician providers in hospital medicine. They focus primarily on the use of mid‐level providers (MLPs), namely physician‐assistants (PAs) and nurse practitioners (NPs), as a potential solution to the current physician workforce shortages in our field. While we acknowledge the challenges of meeting workforce needs, we also believe that much is unknown about the optimal use of MLPs on inpatient general medicine services and it is premature to tout MLPs as the solution to hospital medicine staffing problems. This is especially true in those hospitals where hospitalists care for complex, general medical patients with a wide variety of medical conditions, a trend that is especially common in academic medical centers.2
This article discusses the current literature, our own experiences with MLPs, and suggests some future initiatives that might help better integrate MLPs into hospital medicine.
The Literature on MLPs in Inpatient Venues
The existing literature on the use of MLPs in inpatient venues is quite limited, and a recent review, while suggesting that the existing literature does describe benefits of MLPs in the inpatient setting, also states that the overall quality of the evidence is quite poor and that many studies suffer from significant limitations, including small populations, limited patient mixes, use of selected settings, and short durations of outcome assessment.3
Ford and Britting,1 in their article, cite several studies46 as evidence that a MLP model of care either improved outcomes or provided cost benefits. Each of these studies has important limitations that are worth examining.
The study by Myers et al.4 described the use of MLPs in a chest pain unit. NPs partnered with hospitalists to care for a low‐acuity chest pain population. In addition, 5 NPs only staffed the unit during daytime weekday hours. Off‐hour and weekend staffing was accomplished through the use of resident physicians. Notably, the work suggests the service only admitted 113 low‐risk patients over 10 months. The service was staffed by 3 full‐time equivalent (FTE) NPs in addition to involving hospitalists during the day. It is not surprising, given the extremely low volume of patients coupled with a daytime‐only focus, that this service showed efficiency gains. In addition, given the service was only staffed by NPs 40 hours a week and by resident physicians on nights and weekends, the true cost of such an intervention needs to take into account the full cost of 24/7 coverage. In addition, the model of using residents to cover nonteaching patients is no longer permitted by the current Accreditation Council for Graduate Medical Education (ACGME) Internal Medicine Residency Requirements7 and thus implementation of a model such as this in 2009 would require alternative means of nighttime coverage.
The study by Nishimura et al.,5 also describing the use of MLPs in cardiovascular care, has important caveats that make full assessment of the model impossible. The model describes the implementation of a care team consisting of an attending, a fellow, and MLPs to replace a traditional teaching team of an attending, senior resident, and 2 interns. The study states that the model resulted in a lower length of stay (LOS) and lower costs per case. Importantly, the new MLP‐based team only admitted during the hours of 7 AM to 2 PM. The study does not fully describe the number of MLPs required nor does it fully describe the role of cardiovascular fellows in the model. The study does state that the cost savings offset the cost of the MLPs but it is not clear if this cost analysis took into account the cost of the fellow's daytime involvement or if it measured attending time required before and after the implementation of the new model. In addition, this model presumes the availability of other services to admit patients during afternoon and nighttime hours and so may not be generalizable to other settings.
The final study by Cowan et al.6 describes the addition of a NP, a hospitalist medical director, and daily multidisciplinary rounds to a traditional teaching service model. Importantly, the NP was not involved in the admission process nor were they the primary providers for day‐to‐day medical care but rather they focused on implementation of care protocols, multidisciplinary coordination of care and discharge planning, and postdischarge follow‐up. In addition, the NP worked only weekdays for about 40 hours a week. It is not surprising that adding multiple additional resources to existing care models might provide benefits but this does not address any issues in terms of the workforce since the care in this model required a higher total input of providers than the usual care model being studied. Cost savings from such a model may make it cost‐effective but it does not represent a workforce solution.
There have been other studies examining the use of MLPs in the inpatient setting in internal medicine. Some of these studies have suggested that MLP‐based models result in equivalent outcomes and efficiency810 to traditional teaching or nonteaching physician‐only models. There are 2 important caveats, however, that must be considered. The total resources required for such models may be quite high, especially taking into account the costs of 24/7 coverage and physician backup of the MLPs, and most importantly there is almost no literature that robustly examines ultimate clinical outcomes in these models. We do note that a recent study11 did show a lower inpatient mortality rate over a 2‐year period of time after substituting a PA‐hospitalist model for a traditional academic medicine residency model in a community hospital. Importantly, however, the new model also added 24/7 hospitalist physicians and night and weekend intensivists that were not present in the prior residency‐based model. Thus, the lower mortality rate could be attributed to the addition of hospitalists or the more robust in‐house physician coverage during off‐hours rather than the use of MLPs.
Notably, while the evidence base in internal medicine is not robust, many studies have described successful use of MLPs in non‐internal medicine inpatient settings.1214 The reasons for this success is debatable, but it may be that MLPs are more successful in settings where the care is either more protocol‐driven or where there is less diagnostic and therapeutic complexity.
Recent Experiences with MLPs in Academic Hospital Medicine
Given the paucity of data, it is clear that further research is needed on the role of MLPs in hospital medicine. While waiting for such evidence to appear, it may be worthwhile to reflect on the recent experience of 3 major medical centers. A recent article described 5 hospitalist models at major academic medical centers across the country. Two of the institutions described at the time (University of Michigan Health System, Ann Arbor, MI; and Brigham and Women's Hospital, Boston, MA) utilized MLPs as a major element of their staffing of nonresident hospitalist services while another (University of California, San Francisco [UCSF] Medical Center at Mt. Zion, San Francisco, CA) had previously used MLPs as part of its model but phased them out about 1 year prior to publication of the article.2 The model used by the Brigham and Women's Hospital was later described in more detail in a subsequent publication.8 Recently 1 of these institutions (Michigan) has chosen to phase out MLPs. At Michigan, a 4‐year experience with PAs on a general‐medicine focused hospitalist service eventually led to the conclusion that continued use of PAs was not cost‐effective. Significant barriers to success included a steep learning curve and the significant time required before PAs developed sufficient autonomy and efficiency in caring for a highly complex heterogeneous patient population. In the Michigan experience, PAs took up to 2 years to attain a significant level of autonomy and efficiency and even then some PAs still required a significant amount of physician oversight. Similar concerns at UCSF Mt. Zion led to the elimination of their MLP program as well. At Brigham and Women's, the MLP service continues but has required additional hospitalist staffing due to difficulties recruiting qualified MLPs with appropriate inpatient experience. In all cases, the models were challenged by high costs and the difficulty of developing MLPs to attain the level of autonomy and efficiency needed to justify their continued use. A key point is that in each institution, MLPs continue to play an important role in some specialty inpatient areas such as Hematology/Oncology and Bone Marrow Transplant, which is where MLPs have traditionally found their niche in inpatient Internal Medicine. These focus shops allow MLPs to develop a niche and expertise in a specialized area, where they may become more autonomous and efficient than house staff. Thus these settings may be more appropriate for MLPs than a heterogeneous general medicine inpatient setting.
Reviewing the Financial Case
In their article, Ford and Britting1 cite potential financial advantages for the use of MLPs in hospital medicine by comparing the relative salaries of MLPs to Hospitalists. What was missing in their analysis was the relative productivity of the 2 types of providers. We do have some limited data from the Society of Hospital Medicine (SHM) annual survey that looks at MLPs in hospital medicine but, again, the number of respondents for most data elements is less than 70, making generalizability difficult. Nonetheless, the data suggest that MLPs in hospital medicine average about 60% to 75% of the productivity of a physician when measured by encounters, although there is wide variability depending on the employment model (academic vs. multispecialty group).15 Importantly, the existing data do not provide any measure of how much physician input is provided to these MLPs but we suspect that in most models there is some physician time and input. If we presume that the MLPs bill independently and collect 85% of the physician fee schedule for a Medicare population, then collections would be about 50% to 65% of a typical physician. Given that median total compensation including benefits from the SHM survey was $120,000 for MLPs and $216,000 for physiciansabout a 55% ratiothis would argue for potential financial neutrality when substituting MLPs for physicians in a 2:1 ratio but only if we presume they require no physician supervision, which in our own experience is not likely in a general medicine population. In an alternative model, in which the physician sees every patient with the MLP and the physician bills, one would need to see roughly 50% more patients to achieve a financially neutral situation. In our experience at our own institutions, this level of increased productivity was not achievable. It is important to note that our figures are median compensation and benefit cost figures and local markets vary widely. We know that in major east and west coast cities MLPs may command far higher salaries while early career hospitalist physicians may be paid somewhat less than the reported medians. Recent market changes have significantly pressured MLP salaries,15, 16 further impacting the financial equation and perhaps tilting it farther against a financial benefit for MLPs. Furthermore, night coverage for MLP services should always be considered in a financial analysis and is not captured in this simple analysis.
Next Steps
Given the current shortage of physicians, we imagine that many hospitalist groups will consider the use of MLPs as a solution to the current workforce issues. However, data on how best to utilize MLPs and the true impact on both the cost and quality of such models is lacking. In addition to urging increased publication and dissemination of existing experiences with NP and PAs, we strongly suggest that groups considering starting a MLP model do so in a way which would facilitate robust analysis and comparison of the model with alternatives. We also suggest that SHM consider the following: modifying its biennial survey to better capture the nuances of MLP productivity (such as assessing the amount of physician input and supervision required); targeting MLPs so as to increase the number of respondents; and doing an additional survey to capture demographics and basic data on existing MLP models given the lack of published literature.
In addition to gathering more data on effective models, a critical gap that we have identified is the development of models for the training and development of MLPs interested in hospital medicine. It would be a mistake to believe that MLPs could function in a manner similar to residency‐trained physicians if they do not undergo similar training. NP/PA programs generally do not have a significant inpatient internal medicine focus and so newly minted graduates often lack the skills needed to succeed in hospital medicine.17 Some hospitalist programs train their MLPs on the job, but many programs cannot afford the amount of time and effort required to do this on their own. There are a small number of advanced training options for MLPs in hospital medicine18 but it is not likely such models will proliferate given the inherent opportunity costs that exist for extended training in the current competitive job market for MLPs. Instead we think that very motivated hospital medicine groups may develop training relationships with PA and NP schools in an effort to train their own. In addition, national initiatives such as the Hospital Medicine Boot Camp for NPs and PAs, which is cosponsored by SHM, the American Association of Physician Assistants (AAPA), and the American Academy of Nurse Practitioners (AANP),19 can help fill the educational needs for MLPs who are already in practice.
Conclusions
While some literature exists that suggests that MLPs can successfully be used in the inpatient internal medicine setting, it is important to note that the evidence is quite limited and cannot be generalized across all care settings and patient populations. There is an urgent need to gather more data and share our collective experiences to better inform our decision‐making before we state that MLPs are the solution to workforce shortages in hospital medicine. In addition, existing data and experience suggest that MLPs may not be a cost‐effective workforce solution for complex general medical patients who require significant physician input. We believe that redesigning the clinical training of MLPs to focus on inpatient skills may hold promise and encourage interested parties to consider developing partnerships with MLP training programs and hospital medicine groups, as a way to build a more robust and successful hospital medicine MLP workforce.
- ,.Nonphysician providers in the hospitalist model: a prescription for change and a warning about unintended side effects.J Hosp Med.2010;5:99–102.
- ,,,,.Non‐housestaff medicine services in academic medical centers: models and challenges.J Hosp Med.2008;3:247–255.
- ,,.Nurse practitioners and physician assistants in the intensive care unit: an evidence‐based review.Crit Care Med.2008;36:2888–2897.
- ,,.Improving resource utilization in a teaching hospital: development of a nonteaching service for chest pain admissions.Acad Med.2006;81:432–435.
- ,,,,,.A nonresident cardiovascular inpatient service improves residents' experiences in an academic medical center: a new model to meet the challenges of the new millennium.Acad Med.2004;79;426–431.
- .The effect of a multidisciplinary hospitalist/physician and advance practice nurse collaboration on hospital care.J Nurs Adm.2006;36:79–85.
- Accreditation Council for Graduate Medical Education. ACGME Program Requirements for Residency Education in Internal Medicine. Available at: http://www.acgme.org/acWebsite/downloads/RRC_progReq/140_internal_ medicine_07012009.pdf. Accessed July2009.
- ,,, et al.Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes.J Hosp Med.2008;3:361–368.
- ,,.Resource use by physician assistant services versus teaching services.JAAPA.2002;15:33–38.
- ,,,, et al.Outcomes‐based trial of an inpatient nurse practitioner service for general medical patients.J Eval Clin Pract.2001;7:21–33.
- ,.Replacing an academic internal medicine residency program with a physician assistant‐hospitalist model: a comparative analysis study.Am J Med Qual.2009;2:132–139.
- ,,,,.Integrating midlevel practitioners into a teaching service.Am J Surg.2006;1:119–124.
- ,,, et al.Physician extenders impact trauma systems.J Trauma.2005;58(5):917–920.
- ,.Physician assistants in cardiothoracic surgery: a 30‐year experience in a university center.Ann Thorac Surg.2006;1:195–199.
- 2007–2008 Society of Hospital Medicine Bi‐Annual Survey: the Authoritative Source on the State of the Hospital Medicine Movement.Philadelphia:Society of Hospital Medicine;2008.
- American Association of Physician Assistants. Physician Assistant Income. Available at: http://www.aapa.org/images/stories/iu08incchange. pdf. Accessed July2009.
- Accreditation Review Commission on Education for the Physician Assistant. Accreditation Standards for Physician Assistant Education, 3rd ed. Available at: http://www.arcpa.org/Standards/3rdeditionwithPDchangesandregionals4.24.08a.pdf. Accessed July2009.
- Association of Postgraduate PA Programs. Postgraduate PA Program Listing by State. Available at: http://www.appap.org/index1.html. Accessed July2009.
- American Association of Physician Assistants. Adult Hospitalist Physician Assistant and Nurse Practitioner Boot Camp. Available at: http://www. aapa.org/component/content/article/23‐‐general‐/673‐adult‐hospitalist‐physician‐assistant‐and‐nurse‐practitioner‐boot‐camp. Accessed July2009.
- ,.Nonphysician providers in the hospitalist model: a prescription for change and a warning about unintended side effects.J Hosp Med.2010;5:99–102.
- ,,,,.Non‐housestaff medicine services in academic medical centers: models and challenges.J Hosp Med.2008;3:247–255.
- ,,.Nurse practitioners and physician assistants in the intensive care unit: an evidence‐based review.Crit Care Med.2008;36:2888–2897.
- ,,.Improving resource utilization in a teaching hospital: development of a nonteaching service for chest pain admissions.Acad Med.2006;81:432–435.
- ,,,,,.A nonresident cardiovascular inpatient service improves residents' experiences in an academic medical center: a new model to meet the challenges of the new millennium.Acad Med.2004;79;426–431.
- .The effect of a multidisciplinary hospitalist/physician and advance practice nurse collaboration on hospital care.J Nurs Adm.2006;36:79–85.
- Accreditation Council for Graduate Medical Education. ACGME Program Requirements for Residency Education in Internal Medicine. Available at: http://www.acgme.org/acWebsite/downloads/RRC_progReq/140_internal_ medicine_07012009.pdf. Accessed July2009.
- ,,, et al.Implementation of a physician assistant/hospitalist service in an academic medical center: impact on efficiency and patient outcomes.J Hosp Med.2008;3:361–368.
- ,,.Resource use by physician assistant services versus teaching services.JAAPA.2002;15:33–38.
- ,,,, et al.Outcomes‐based trial of an inpatient nurse practitioner service for general medical patients.J Eval Clin Pract.2001;7:21–33.
- ,.Replacing an academic internal medicine residency program with a physician assistant‐hospitalist model: a comparative analysis study.Am J Med Qual.2009;2:132–139.
- ,,,,.Integrating midlevel practitioners into a teaching service.Am J Surg.2006;1:119–124.
- ,,, et al.Physician extenders impact trauma systems.J Trauma.2005;58(5):917–920.
- ,.Physician assistants in cardiothoracic surgery: a 30‐year experience in a university center.Ann Thorac Surg.2006;1:195–199.
- 2007–2008 Society of Hospital Medicine Bi‐Annual Survey: the Authoritative Source on the State of the Hospital Medicine Movement.Philadelphia:Society of Hospital Medicine;2008.
- American Association of Physician Assistants. Physician Assistant Income. Available at: http://www.aapa.org/images/stories/iu08incchange. pdf. Accessed July2009.
- Accreditation Review Commission on Education for the Physician Assistant. Accreditation Standards for Physician Assistant Education, 3rd ed. Available at: http://www.arcpa.org/Standards/3rdeditionwithPDchangesandregionals4.24.08a.pdf. Accessed July2009.
- Association of Postgraduate PA Programs. Postgraduate PA Program Listing by State. Available at: http://www.appap.org/index1.html. Accessed July2009.
- American Association of Physician Assistants. Adult Hospitalist Physician Assistant and Nurse Practitioner Boot Camp. Available at: http://www. aapa.org/component/content/article/23‐‐general‐/673‐adult‐hospitalist‐physician‐assistant‐and‐nurse‐practitioner‐boot‐camp. Accessed July2009.
Acute Pancreatitis with Eruptive Xanthomas
A 54‐year‐old man presented with a 1‐day history of epigastric abdominal pain. He also described 1 month of a nonpruritic but tender rash, on his right elbow, abdomen, buttocks, posterior thighs, and knees. He was obese with a past medical history remarkable for uncontrolled type 2 diabetes mellitus and hemoglobin A1C of 12.7. His abdomen was tender to palpation in the epigastric area with no rebound or guarding. Skin examination demonstrated multiple yellow waxy papules on the extensor surfaces of his arms, abdomen, thighs, knees, and buttocks, consistent with eruptive xanthomas (Figure 1). Laboratory values were significant for lipase at 852 g/L (normal, 8‐78 g/L) and triglycerides at 6200 mg/dL. An abdominal computed tomography (CT) scan without contrast demonstrated extensive inflammatory changes surrounding the head of the pancreas, consistent with acute pancreatitis (Figure 2). The diagnosis of acute pancreatitis secondary to hypertriglyceridemia was made. Dietary and pharmacologic interventions helped decrease the triglyceride level and his rash and abdominal pain were improved at outpatient follow‐up 2 weeks later.
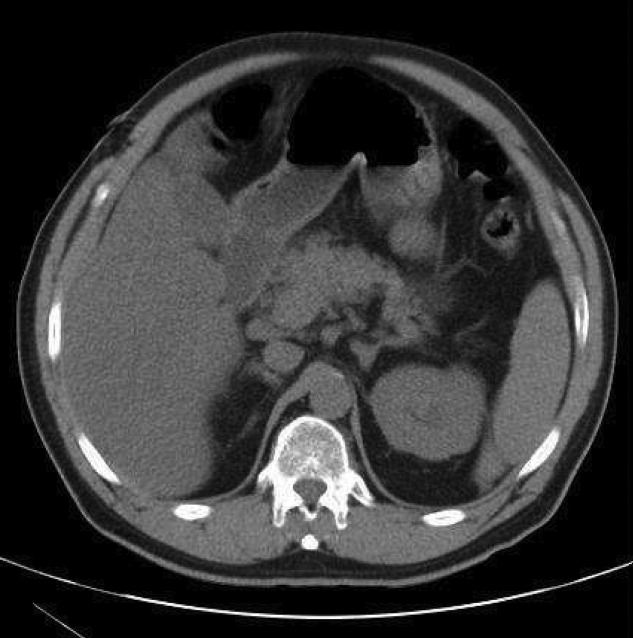
Hypertriglyceridemia increases the risk of acute pancreatitis.1 Eruptive xanthomas can be associated with primary and secondary hypertriglyceridemia, particularly in the setting of poorly controlled diabetes.2 The risk of acute pancreatitis and eruptive xanthomas increases when the level of serum triglyceride reaches the thousands.
Rapid recognition of eruptive xanthomas can assist in identifying the etiology of acute pancreatitis.
- .Hyperlipidemic pancreatitis. [Review].Gastroenterol Clin North Am.1990;19(4):783–791.
- .Xanthomas and hyperlipidemias.J Am Acad Dermatol.1985;13(1):1–30.
A 54‐year‐old man presented with a 1‐day history of epigastric abdominal pain. He also described 1 month of a nonpruritic but tender rash, on his right elbow, abdomen, buttocks, posterior thighs, and knees. He was obese with a past medical history remarkable for uncontrolled type 2 diabetes mellitus and hemoglobin A1C of 12.7. His abdomen was tender to palpation in the epigastric area with no rebound or guarding. Skin examination demonstrated multiple yellow waxy papules on the extensor surfaces of his arms, abdomen, thighs, knees, and buttocks, consistent with eruptive xanthomas (Figure 1). Laboratory values were significant for lipase at 852 g/L (normal, 8‐78 g/L) and triglycerides at 6200 mg/dL. An abdominal computed tomography (CT) scan without contrast demonstrated extensive inflammatory changes surrounding the head of the pancreas, consistent with acute pancreatitis (Figure 2). The diagnosis of acute pancreatitis secondary to hypertriglyceridemia was made. Dietary and pharmacologic interventions helped decrease the triglyceride level and his rash and abdominal pain were improved at outpatient follow‐up 2 weeks later.


Hypertriglyceridemia increases the risk of acute pancreatitis.1 Eruptive xanthomas can be associated with primary and secondary hypertriglyceridemia, particularly in the setting of poorly controlled diabetes.2 The risk of acute pancreatitis and eruptive xanthomas increases when the level of serum triglyceride reaches the thousands.
Rapid recognition of eruptive xanthomas can assist in identifying the etiology of acute pancreatitis.
A 54‐year‐old man presented with a 1‐day history of epigastric abdominal pain. He also described 1 month of a nonpruritic but tender rash, on his right elbow, abdomen, buttocks, posterior thighs, and knees. He was obese with a past medical history remarkable for uncontrolled type 2 diabetes mellitus and hemoglobin A1C of 12.7. His abdomen was tender to palpation in the epigastric area with no rebound or guarding. Skin examination demonstrated multiple yellow waxy papules on the extensor surfaces of his arms, abdomen, thighs, knees, and buttocks, consistent with eruptive xanthomas (Figure 1). Laboratory values were significant for lipase at 852 g/L (normal, 8‐78 g/L) and triglycerides at 6200 mg/dL. An abdominal computed tomography (CT) scan without contrast demonstrated extensive inflammatory changes surrounding the head of the pancreas, consistent with acute pancreatitis (Figure 2). The diagnosis of acute pancreatitis secondary to hypertriglyceridemia was made. Dietary and pharmacologic interventions helped decrease the triglyceride level and his rash and abdominal pain were improved at outpatient follow‐up 2 weeks later.


Hypertriglyceridemia increases the risk of acute pancreatitis.1 Eruptive xanthomas can be associated with primary and secondary hypertriglyceridemia, particularly in the setting of poorly controlled diabetes.2 The risk of acute pancreatitis and eruptive xanthomas increases when the level of serum triglyceride reaches the thousands.
Rapid recognition of eruptive xanthomas can assist in identifying the etiology of acute pancreatitis.
- .Hyperlipidemic pancreatitis. [Review].Gastroenterol Clin North Am.1990;19(4):783–791.
- .Xanthomas and hyperlipidemias.J Am Acad Dermatol.1985;13(1):1–30.
- .Hyperlipidemic pancreatitis. [Review].Gastroenterol Clin North Am.1990;19(4):783–791.
- .Xanthomas and hyperlipidemias.J Am Acad Dermatol.1985;13(1):1–30.
HM Often Lends a Critical-Care Hand
Hospitalists serving as attending physicians were overwhelmingly found in ICUs in hospitals where intensivists were used mostly as elective consultations, according to a study to be published in this month’s Journal of Hospital Medicine.
The report, "Characteristics of Intensive Care Units in Michigan: Not an Open and Closed Case" (JHM. 2010;5:4-9), is a cross-sectional survey of 115 ICUs in 72 Michigan hospitals. The way critical care is delivered is an issue many hospitalists pay mind to as they often are called upon to compensate for workforce shortages even though few are board-certified in critical care. The study reported that 25% of sites used a closed model of intensive care, and that those units were larger than sites with open ICUs (P<0.05). Hospitalists serving as attendings were "strongly associated with an open ICU" (odds ratio 95%; confidence interval=12.2).
"Because our study shows that hospitalists are already providing intensive care in many areas of Michigan … the best strategy [for hospitalists] would be to work within that context,” says lead researcher Robert C. Hyzy, MD, FCCM, associate professor in the Department of Internal Medicine and director of the Critical Care Medicine Unit at the University of Michigan in Ann Arbor.
Dr. Hyzy says hospitalists will "continue to play an important role" in ICU care as long as the intensivist shortage continues, even though many don’t have the extensive training to do so. Telemedicine has been used in recent years to help address the ICU physician shortage, although the debate continues as to its effectiveness on morbidity and mortality rates. In the meantime, Dr. Hyzy reports, only 20% of the sites in his study reported all attending physicians were board-certified in critical care; 60 sites had less than 50% board-certified attending physicians.
“Hospitalists can play a role in acknowledging the limitations of individuals who are not properly trained in critical care to advocate for transfer to larger centers under explicit clinical circumstances,” Dr. Hyzy says.
As a member of the Society of Critical Care Medicine’s (SCCM) fellowship program, Dr. Hyzy also urges any hospitalists who deliver critical-care services to investigate the SCCM’s certification track and to take advantage of any training opportunities available to them.
Hospitalists serving as attending physicians were overwhelmingly found in ICUs in hospitals where intensivists were used mostly as elective consultations, according to a study to be published in this month’s Journal of Hospital Medicine.
The report, "Characteristics of Intensive Care Units in Michigan: Not an Open and Closed Case" (JHM. 2010;5:4-9), is a cross-sectional survey of 115 ICUs in 72 Michigan hospitals. The way critical care is delivered is an issue many hospitalists pay mind to as they often are called upon to compensate for workforce shortages even though few are board-certified in critical care. The study reported that 25% of sites used a closed model of intensive care, and that those units were larger than sites with open ICUs (P<0.05). Hospitalists serving as attendings were "strongly associated with an open ICU" (odds ratio 95%; confidence interval=12.2).
"Because our study shows that hospitalists are already providing intensive care in many areas of Michigan … the best strategy [for hospitalists] would be to work within that context,” says lead researcher Robert C. Hyzy, MD, FCCM, associate professor in the Department of Internal Medicine and director of the Critical Care Medicine Unit at the University of Michigan in Ann Arbor.
Dr. Hyzy says hospitalists will "continue to play an important role" in ICU care as long as the intensivist shortage continues, even though many don’t have the extensive training to do so. Telemedicine has been used in recent years to help address the ICU physician shortage, although the debate continues as to its effectiveness on morbidity and mortality rates. In the meantime, Dr. Hyzy reports, only 20% of the sites in his study reported all attending physicians were board-certified in critical care; 60 sites had less than 50% board-certified attending physicians.
“Hospitalists can play a role in acknowledging the limitations of individuals who are not properly trained in critical care to advocate for transfer to larger centers under explicit clinical circumstances,” Dr. Hyzy says.
As a member of the Society of Critical Care Medicine’s (SCCM) fellowship program, Dr. Hyzy also urges any hospitalists who deliver critical-care services to investigate the SCCM’s certification track and to take advantage of any training opportunities available to them.
Hospitalists serving as attending physicians were overwhelmingly found in ICUs in hospitals where intensivists were used mostly as elective consultations, according to a study to be published in this month’s Journal of Hospital Medicine.
The report, "Characteristics of Intensive Care Units in Michigan: Not an Open and Closed Case" (JHM. 2010;5:4-9), is a cross-sectional survey of 115 ICUs in 72 Michigan hospitals. The way critical care is delivered is an issue many hospitalists pay mind to as they often are called upon to compensate for workforce shortages even though few are board-certified in critical care. The study reported that 25% of sites used a closed model of intensive care, and that those units were larger than sites with open ICUs (P<0.05). Hospitalists serving as attendings were "strongly associated with an open ICU" (odds ratio 95%; confidence interval=12.2).
"Because our study shows that hospitalists are already providing intensive care in many areas of Michigan … the best strategy [for hospitalists] would be to work within that context,” says lead researcher Robert C. Hyzy, MD, FCCM, associate professor in the Department of Internal Medicine and director of the Critical Care Medicine Unit at the University of Michigan in Ann Arbor.
Dr. Hyzy says hospitalists will "continue to play an important role" in ICU care as long as the intensivist shortage continues, even though many don’t have the extensive training to do so. Telemedicine has been used in recent years to help address the ICU physician shortage, although the debate continues as to its effectiveness on morbidity and mortality rates. In the meantime, Dr. Hyzy reports, only 20% of the sites in his study reported all attending physicians were board-certified in critical care; 60 sites had less than 50% board-certified attending physicians.
“Hospitalists can play a role in acknowledging the limitations of individuals who are not properly trained in critical care to advocate for transfer to larger centers under explicit clinical circumstances,” Dr. Hyzy says.
As a member of the Society of Critical Care Medicine’s (SCCM) fellowship program, Dr. Hyzy also urges any hospitalists who deliver critical-care services to investigate the SCCM’s certification track and to take advantage of any training opportunities available to them.
In the Literature: Research You Need to Know
Clinical question: Are aldosterone antagonists appropriately used in heart failure patients?
Background: Aldosterone antagonists are Class 1 treatment for patients with heart failure (HF) and low left ventricular ejection fraction (LVEF), according to the latest American College of Cardiology/American Heart Association (ACC/AHA) Chronic HF Guidelines. However, acceptance of the guidelines has been slow, possibly because of contraindications and adverse effects in some patients.
Study design: Observational cohort.
Setting: Inpatients at 241 hospitals participating in an HF registry.
Synopsis: Eligible inpatients from a QI registry who were discharged home from January 2005 to December 2007 were analyzed for appropriate and inappropriate aldosterone antagonist use.
Among the 12,565 patients who met the criteria for aldosterone antagonist therapy, only 4,087 (32.5%) received the therapy at discharge. In contrast, of the 8,610 total patients with HF discharged on aldosterone antagonists, 269 (3.1%) met at least one criterion for inappropriate use, and 640 (7.4%) met at least one criteria for potentially inappropriate use.
Use of an aldosterone antagonist was more likely in certain patient groups, including those who were younger, African-American, had lower systolic blood pressure, had no history of renal insufficiency, and those with implantable cardioverter-defibrillators. The authors noted aldosterone use increased significantly during the study period.
The study was limited by the possibility that aldosterone antagonists were used for non-HF conditions and the fact that it was retrospective.
Bottom line: Aldosterone antagonists are used in less than one-third of eligible HF patients.
Citation: Albert NM, Yancy CW, Liang L, et al. Use of aldosterone antagonists in heart failure. JAMA. 2009;302(15):1658-1665.
Reviewed for TH eWire by Jill Goldenberg, MD, Alan Briones, MD, Dennis Chang, MD, Brian Markoff, MD, FHM, Erin Rule, MD, Andrew Dunn, MD, FACP, FHM, Division of General Internal Medicine, Mount Sinai School of Medicine, New York City
For more HM-related literature reviews, visit our Web site.
Clinical question: Are aldosterone antagonists appropriately used in heart failure patients?
Background: Aldosterone antagonists are Class 1 treatment for patients with heart failure (HF) and low left ventricular ejection fraction (LVEF), according to the latest American College of Cardiology/American Heart Association (ACC/AHA) Chronic HF Guidelines. However, acceptance of the guidelines has been slow, possibly because of contraindications and adverse effects in some patients.
Study design: Observational cohort.
Setting: Inpatients at 241 hospitals participating in an HF registry.
Synopsis: Eligible inpatients from a QI registry who were discharged home from January 2005 to December 2007 were analyzed for appropriate and inappropriate aldosterone antagonist use.
Among the 12,565 patients who met the criteria for aldosterone antagonist therapy, only 4,087 (32.5%) received the therapy at discharge. In contrast, of the 8,610 total patients with HF discharged on aldosterone antagonists, 269 (3.1%) met at least one criterion for inappropriate use, and 640 (7.4%) met at least one criteria for potentially inappropriate use.
Use of an aldosterone antagonist was more likely in certain patient groups, including those who were younger, African-American, had lower systolic blood pressure, had no history of renal insufficiency, and those with implantable cardioverter-defibrillators. The authors noted aldosterone use increased significantly during the study period.
The study was limited by the possibility that aldosterone antagonists were used for non-HF conditions and the fact that it was retrospective.
Bottom line: Aldosterone antagonists are used in less than one-third of eligible HF patients.
Citation: Albert NM, Yancy CW, Liang L, et al. Use of aldosterone antagonists in heart failure. JAMA. 2009;302(15):1658-1665.
Reviewed for TH eWire by Jill Goldenberg, MD, Alan Briones, MD, Dennis Chang, MD, Brian Markoff, MD, FHM, Erin Rule, MD, Andrew Dunn, MD, FACP, FHM, Division of General Internal Medicine, Mount Sinai School of Medicine, New York City
For more HM-related literature reviews, visit our Web site.
Clinical question: Are aldosterone antagonists appropriately used in heart failure patients?
Background: Aldosterone antagonists are Class 1 treatment for patients with heart failure (HF) and low left ventricular ejection fraction (LVEF), according to the latest American College of Cardiology/American Heart Association (ACC/AHA) Chronic HF Guidelines. However, acceptance of the guidelines has been slow, possibly because of contraindications and adverse effects in some patients.
Study design: Observational cohort.
Setting: Inpatients at 241 hospitals participating in an HF registry.
Synopsis: Eligible inpatients from a QI registry who were discharged home from January 2005 to December 2007 were analyzed for appropriate and inappropriate aldosterone antagonist use.
Among the 12,565 patients who met the criteria for aldosterone antagonist therapy, only 4,087 (32.5%) received the therapy at discharge. In contrast, of the 8,610 total patients with HF discharged on aldosterone antagonists, 269 (3.1%) met at least one criterion for inappropriate use, and 640 (7.4%) met at least one criteria for potentially inappropriate use.
Use of an aldosterone antagonist was more likely in certain patient groups, including those who were younger, African-American, had lower systolic blood pressure, had no history of renal insufficiency, and those with implantable cardioverter-defibrillators. The authors noted aldosterone use increased significantly during the study period.
The study was limited by the possibility that aldosterone antagonists were used for non-HF conditions and the fact that it was retrospective.
Bottom line: Aldosterone antagonists are used in less than one-third of eligible HF patients.
Citation: Albert NM, Yancy CW, Liang L, et al. Use of aldosterone antagonists in heart failure. JAMA. 2009;302(15):1658-1665.
Reviewed for TH eWire by Jill Goldenberg, MD, Alan Briones, MD, Dennis Chang, MD, Brian Markoff, MD, FHM, Erin Rule, MD, Andrew Dunn, MD, FACP, FHM, Division of General Internal Medicine, Mount Sinai School of Medicine, New York City
For more HM-related literature reviews, visit our Web site.
Case 1: Management Decisions in an Adult Comorbid Patient With Type 2 Diabetes Having Primary HyperlipidemiaCase 2: Colesevelam Hydrochloride for Management of a Patient With Type 2 Diabetes Mellitus and Hyperlipidemia
Case 2: Colesevelam Hydrochloride for Management of a Patient With Type 2 Diabetes Mellitus and Hyperlipidemia
A Case Studies Compendium supplement to Internal Medicine News. This supplement was sponsored by Daiichi Sankyo, Inc.
•Case 1 Topics
•Case 2 Topics
•Faculty/Faculty Disclosures

To view the supplement, click the image above.
Case 1 Topics
• Background
• Current Visit
• Laboratory Results
• Clinical Discussion
• Endocrinologist Consultation
• New Treatment Regimen With Add-On Therapy
• Conclusions
Case 2 Topics
• Background
• Current Visit: Exam Findings
• Current Treatment Regimen
• Health History
• Laboratory Results
• Clinical Discussion
• Cardiologist Visit
• Three Months After Visiting the Cardiologist
• Add-On Therapy With Welchol for Patients With T2DM and CHD
• Treatment Goals for Alice
• Conclusions
Faculty/Faculty Disclosures
Yehuda Handelsman, MD, FACP, FACE
Medical Director
Metabolic Institute of America
Chair and Program Director
7th World Congress on InsulinResistance
Chair, International Committeefor Insulin Resistance
18372 Clark Street, Suite 212
Tarzana, CA 91356
E-mail:yhandelsman@pacbell.net
Web Site:www.TheMetabolicCenter.com
Dr Handelsman is a consultant for Bristol-Myers Squibb
Company, Daiichi Sankyo, Inc., GlaxoSmithKline, Medtronic, Merck, Tethys,
and Xoma; he has received clinical research grant funding from Daiichi Sankyo, Inc., GlaxoSmithKline, Novo Nordisk, and Takeda; and he ison the speakers bureau for AstraZeneca, Bristol-Myers Squibb, Daiichi Sankyo, Inc., GlaxoSmithKline, Merck, and Novartis. He also serves on the advisory board for CLINICAL ENDOCRINOLOGY NEWS.
Peter H. Jones, MD, FACP
Co-Director, Lipid Metabolism
and Atherosclerosis Clinic
Medical Director, Weight
Management Center
The Methodist Hospital
Associate Professor of Medicine
Section of Atherosclerosis andLipid Research
Baylor College of Medicine
Houston, TX 77030
E-mail: jones@bcm.tmc.edu
Dr Jones has disclosed that he has received support in the form of consulting agreements from Abbott Laboratories, AstraZeneca Pharmaceuticals LP, Daiichi Sankyo, Inc., and Merck.
Copyright © 2010 Elsevier Inc.
A Case Studies Compendium supplement to Internal Medicine News. This supplement was sponsored by Daiichi Sankyo, Inc.
•Case 1 Topics
•Case 2 Topics
•Faculty/Faculty Disclosures

To view the supplement, click the image above.
Case 1 Topics
• Background
• Current Visit
• Laboratory Results
• Clinical Discussion
• Endocrinologist Consultation
• New Treatment Regimen With Add-On Therapy
• Conclusions
Case 2 Topics
• Background
• Current Visit: Exam Findings
• Current Treatment Regimen
• Health History
• Laboratory Results
• Clinical Discussion
• Cardiologist Visit
• Three Months After Visiting the Cardiologist
• Add-On Therapy With Welchol for Patients With T2DM and CHD
• Treatment Goals for Alice
• Conclusions
Faculty/Faculty Disclosures
Yehuda Handelsman, MD, FACP, FACE
Medical Director
Metabolic Institute of America
Chair and Program Director
7th World Congress on InsulinResistance
Chair, International Committeefor Insulin Resistance
18372 Clark Street, Suite 212
Tarzana, CA 91356
E-mail:yhandelsman@pacbell.net
Web Site:www.TheMetabolicCenter.com
Dr Handelsman is a consultant for Bristol-Myers Squibb
Company, Daiichi Sankyo, Inc., GlaxoSmithKline, Medtronic, Merck, Tethys,
and Xoma; he has received clinical research grant funding from Daiichi Sankyo, Inc., GlaxoSmithKline, Novo Nordisk, and Takeda; and he ison the speakers bureau for AstraZeneca, Bristol-Myers Squibb, Daiichi Sankyo, Inc., GlaxoSmithKline, Merck, and Novartis. He also serves on the advisory board for CLINICAL ENDOCRINOLOGY NEWS.
Peter H. Jones, MD, FACP
Co-Director, Lipid Metabolism
and Atherosclerosis Clinic
Medical Director, Weight
Management Center
The Methodist Hospital
Associate Professor of Medicine
Section of Atherosclerosis andLipid Research
Baylor College of Medicine
Houston, TX 77030
E-mail: jones@bcm.tmc.edu
Dr Jones has disclosed that he has received support in the form of consulting agreements from Abbott Laboratories, AstraZeneca Pharmaceuticals LP, Daiichi Sankyo, Inc., and Merck.
Copyright © 2010 Elsevier Inc.
A Case Studies Compendium supplement to Internal Medicine News. This supplement was sponsored by Daiichi Sankyo, Inc.
•Case 1 Topics
•Case 2 Topics
•Faculty/Faculty Disclosures

To view the supplement, click the image above.
Case 1 Topics
• Background
• Current Visit
• Laboratory Results
• Clinical Discussion
• Endocrinologist Consultation
• New Treatment Regimen With Add-On Therapy
• Conclusions
Case 2 Topics
• Background
• Current Visit: Exam Findings
• Current Treatment Regimen
• Health History
• Laboratory Results
• Clinical Discussion
• Cardiologist Visit
• Three Months After Visiting the Cardiologist
• Add-On Therapy With Welchol for Patients With T2DM and CHD
• Treatment Goals for Alice
• Conclusions
Faculty/Faculty Disclosures
Yehuda Handelsman, MD, FACP, FACE
Medical Director
Metabolic Institute of America
Chair and Program Director
7th World Congress on InsulinResistance
Chair, International Committeefor Insulin Resistance
18372 Clark Street, Suite 212
Tarzana, CA 91356
E-mail:yhandelsman@pacbell.net
Web Site:www.TheMetabolicCenter.com
Dr Handelsman is a consultant for Bristol-Myers Squibb
Company, Daiichi Sankyo, Inc., GlaxoSmithKline, Medtronic, Merck, Tethys,
and Xoma; he has received clinical research grant funding from Daiichi Sankyo, Inc., GlaxoSmithKline, Novo Nordisk, and Takeda; and he ison the speakers bureau for AstraZeneca, Bristol-Myers Squibb, Daiichi Sankyo, Inc., GlaxoSmithKline, Merck, and Novartis. He also serves on the advisory board for CLINICAL ENDOCRINOLOGY NEWS.
Peter H. Jones, MD, FACP
Co-Director, Lipid Metabolism
and Atherosclerosis Clinic
Medical Director, Weight
Management Center
The Methodist Hospital
Associate Professor of Medicine
Section of Atherosclerosis andLipid Research
Baylor College of Medicine
Houston, TX 77030
E-mail: jones@bcm.tmc.edu
Dr Jones has disclosed that he has received support in the form of consulting agreements from Abbott Laboratories, AstraZeneca Pharmaceuticals LP, Daiichi Sankyo, Inc., and Merck.
Copyright © 2010 Elsevier Inc.
Case 2: Colesevelam Hydrochloride for Management of a Patient With Type 2 Diabetes Mellitus and Hyperlipidemia
Case 2: Colesevelam Hydrochloride for Management of a Patient With Type 2 Diabetes Mellitus and Hyperlipidemia
An Approach to the Management of Type 2 Diabetes Mellitus in Patients Receiving Add-On Therapy With Colesevelam HCl
A Journal Scan supplement to Family Practice News
A Journal Scan supplement to Family Practice News
A Journal Scan supplement to Family Practice News