User login
An algorithm for the treatment of chronic testicular pain
Order ultrasound of the scrotum and testes to evaluate chronic testicular pain, with color Doppler to identify areas of hypervascularity. C
Treat suspected epididymitis with empiric coverage for chlamydia with either a 10-day regimen of doxycycline (100 mg twice daily) or a single dose (1 g) of azithromycin; treat suspected gonorrhea with a single intramuscular injection (125 mg) of ceftriaxone. A
Do not treat small epididymal cysts that do not correlate with testicular pain; larger, painful cysts can be aspirated, injected with a sclerosing agent, or surgically excised. C
Consider surgical options only after medical and conservative therapies have failed to alleviate chronic testicular pain. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE 1 Vincent B, a 33-year-old executive, visits his family physician for an evaluation of chronic orchialgia. Although his testicular pain has waxed and waned for several years, it has recently worsened, making it increasingly difficult for him to exercise or to sit for extended periods of time. In fact, this visit was prompted by a lengthy meeting during which he developed a “dull ache” that did not let up until he left the meeting and walked around.
CASE 2 Jason H, a 42-year-old married father of 3 who had a vasectomy 2 years ago, has had progressively worsening testicular pain ever since. He also has occasional pain after ejaculation, but no known hematospermia. Recently, the pain has become so bad that it limits both his physical and sexual activities and is having a negative effect on his relationship with his wife. Jason is sexually monogamous, has no significant medical history, and takes no prescription medications.
These 2 cases are based on actual patients we have seen in our practices. If Vincent and Jason (not their real names) were your patients, how would you initiate a work-up for testicular pain? What treatments would you offer? And at what point would you consider a referral to a urologist?
Chronic orchialgia is a complex urogenital focal pain syndrome in which neurogenic inflammation is the principal mediator. This debilitating condition is associated with substantial anxiety and frustration, and is characterized by intermittent or constant unilateral or bilateral testicular pain, occurring for at least 3 months, that has a significant negative impact on activities of daily living and physical activity.1
A variety of procedural and surgical options may help to minimize or alleviate chronic orchialgia. But which approach is best? There are no evidence-based guidelines for the treatment of this condition, and no randomized controlled trials to demonstrate the superiority of 1 modality over another. All diagnostic and treatment recommendations are based on expert opinion derived from small cohort studies.
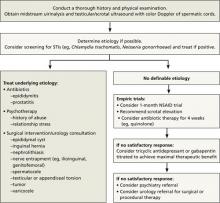
With that in mind, we conducted a systematic review of the literature evaluating medical and surgical therapies for chronic testicular pain—and developed an algorithm (FIGURE 1), along with the text and TABLE that follow, for family physicians (FPs) to use as a guide.
FIGURE 1
Chronic orchialgia: A diagnosis and treatment algorithm1,3,4,6,10
NSAID, nonsteroidal anti-inflammatory drug; STIs, sexually transmitted infections.
CASE 1 Vincent B
Over the last few years, Vincent has had similar episodes of bilateral testicular pain. He denies any history of direct trauma to the testicles, and he works out regularly by lifting weights and running. When the pain becomes unbearable, he takes acetaminophen or ibuprofen and takes a few days off from exercising, which provides modest—but temporary—relief.
Vincent reports that he has had about a dozen lifetime sexual partners and had chlamydia over a decade ago as a college student. He is currently engaged and sexually monogamous, and tested negative for Chlamydia trachomatis, Neisseria gonorrhoeae, hepatitis, syphilis, and human immunodeficiency virus (HIV) at his annual health maintenance examination last month. Shortly before that, Vincent was treated empirically for epididymitis with a 4-week course of ciprofloxacin, with no significant improvement in symptoms. He has no significant past medical history, denies depression, and takes no prescription medications.
Physical examination reveals mild to moderate diffuse tenderness to palpation throughout the scrotum, including both testicles and spermatic cords. There is no erythema of the scrotum. Nor are there any palpable scrotal masses, varicoceles, or hydroceles; testicular, scrotal, or penile lesions; inguinal masses; or lymph nodes. His urethral meatus is patent. The prostate is smooth, nonnodular, and non-tender. The remainder of the physical exam is unremarkable.
Determining a cause can be a challenge
There are numerous possible causes of testicular pain (TABLE), including an inguinal hernia, torsion of the testicle, trauma, and a history of chlamydia or gonorrhea, to name a few.
TABLE
Causes of acute and chronic orchialgia1,3,4
Acute
|
Chronic
|
Chronic testicular pain can also be psychogenic, often relating to a history of sexual abuse or relationship stress. One study examining comorbid psychological conditions in men with chronic orchialgia identified a somatization disorder in 56% of the patients, nongenital chronic pain syndromes in 50%, and major depression or chemical dependency in 27%.2 Overall, however, estimates suggest that in about 25% of patients with chronic orchialgia, no identifiable etiology is found. 1
Establish a baseline with a physical exam
Conduct a physical examination of the scrotum, testes, spermatic cords, penis, inguinal region, and prostate as a baseline measurement in a patient who presents with chronic orchialgia.3,4 An initial urinalysis should be performed to rule out infection or identify microscopic hematuria, which may prompt a more targeted work-up and therapeutic plan. Take a thorough medical and psychosocial/ sexual history, as well.
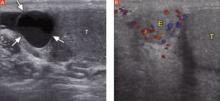
Order an ultrasound of the scrotum and testes, the accepted gold standard to highlight structural abnormalities of the testicles. The addition of color Doppler makes it possible to find areas of hypervascularity, an indication of inflammation in the testicle and epididymis (FIGURES 2A AND B).
FIGURE 2
Well-circumscribed extratesticular mass
In the image at left, ultrasound reveals an anechoic mass (arrows), representing either an epididymal cyst or spermatocele, superior to the testicle (T). A color Doppler image (right) reveals increased vascularity to the epididymis (E), as compared with the testicle.
Epididymal cysts are common findings on scrotal ultrasound; they are frequently incidental, but may relate to the patient’s pain, depending on the size of the cyst. Smaller cysts that do not correlate with pain do not require treatment. Larger, painful cysts can be treated with aspiration or injection with a sclerosing agent—or with surgical excision, which offers the highest potential cure rate.3,4 A computed tomography (CT) scan without contrast is the best way to find genitourinary system calculi, which could be the source of referred renal pain to the groin and scrotum. A contrast-enhanced CT is best to evaluate for solid renal masses.
Start with the most conservative treatment
In the absence of any findings that require surgical intervention, start conservatively.
Initiate a trial of nonsteroidal anti-inflammatory drugs (NSAIDs) for at least 1 month. Although this is the standard first-line treatment, NSAIDs have been shown to help only a small percentage of patients with chronic orchialgia, and only on a short-term basis.1,3,4
Recommend scrotal elevation with supportive undergarments to decrease venous congestion. Tell the patient, too, that modifying his seated posture to avoid scrotal pressure may alleviate pain and poses no discernible risk of worsening orchialgia.5
Treat suspected STIs. The Centers for Disease Control and Prevention report that in men 14 to 35 years of age, epididymitis is most commonly caused by chlamydia or gonorrhea.6 In males younger than 14 or older than 35, epididymitis is most commonly caused by urinary coliform pathogens, including Eschericia coli.
If epididymitis is suspected to be due to chlamydia or gonorrhea, treatment should include either doxycycline 100 mg orally twice daily for 10 days or a single dose of azithromycin 1 g orally (for chlamydia eradication) and a single dose of ceftriaxone 125 mg intramuscularly (for gonorrhea eradication).6,7 If coliform bacteria is suspected, order a standard dose of a quinolone (eg, ciprofloxacin or levofloxacin 500 mg/d) for 10 days.6 For refractory cases, treatment with a standard dose of a quinolone for 4 weeks is recommended.6
It is generally reasonable to treat most patients empirically for suspected epididymitis with antibiotics if no other identifiable etiology can be determined. Multiple antibiotic treatments should be avoided, however, in the absence of either an identifiable urogenital infection or ultrasound findings consistent with epididymitis (eg, congestion and enlargement). Antibiotics have not been shown to decrease the severity of chronic orchialgia and their use, unless clearly indicated, may lead to drug resistance.3
Consider a tricyclic antidepressant or gabapentin
Both tricyclic antidepressants (TCAs) and gabapentin have demonstrated benefit in the treatment of chronic pelvic and neuropathic pain.8,9 Doses should be titrated to achieve a maximal therapeutic benefit while avoiding anticholinergic and neurologic side effects.
A cohort study using a multidisciplinary team consisting of a psychologist, an anesthetist, a physiotherapist, and an occupational therapist found >50% symptomatic improvement in 62% of men with chronic orchialgia treated with gabapentin up to 1800 mg per day, and 67% of men treated with nortriptyline up to 150 mg per day.10
However, a subgroup of patients who reported postvasectomy testicular pain did not achieve a 50% symptomatic improvement rate with either TCA or gabapentin therapy.
CASE 1 Vincent B
The FP reassured Vincent that his physical examination was normal and recommended a 1-month trial of ibuprofen (600 mg every 6 hours), and regular use of supportive briefs. Since the patient had been treated with antibiotics in the past with no change in symptoms—and because he was thought to be at low risk for an STI—the physician did not prescribe another empiric trial of antibiotics. He did send the patient for an ultrasound evaluation of the scrotum and testes, which revealed only a 0.5 × 0.4 × 0.6-cm right epididymal cyst that was not palpable on examination.
The patient returned after 1 month, noting that his symptoms had neither improved nor worsened. The FP suggested that he stop taking the ibuprofen and begin a trial of gabapentin 100 mg daily, titrating up to 3 times daily for the first month, then to 300 mg 3 times daily in the second month.
When he returned 3 months later, Vincent reported that his symptoms had improved by about 50%. He has since been able to increase both the intensity and frequency of physical activity. Vincent is not interested in further increasing the dose of gabapentin and declined a referral to a urologist for consideration of procedural and surgical therapeutic options, but agreed to follow up as needed if his testicular pain worsened.
Postvasectomy pain is not unusual
Several years after a vasectomy, the diameter of a man’s ejaculatory ducts often doubles in size to counteract the increase in fluid pressure.11 The specific cause of long-term post-vasectomy pain syndrome, or congestive epididymitis, is unknown, but has been reported in 5% to 43% of men who have undergone this procedure.12-14 Sperm granulomas or spermatoceles represent the body’s effort to spare the testicle from damage secondary to increasing fluid pressure. While these granulomas are benign lesions, their presence may predispose a man to postvasectomy pain syndrome.15-17
CASE 2 Jason H
Two months before Jason’s visit to the FP, his testicular pain had become so excrutiating that he went to the ED seeking treatment. He was given an ultrasound with color Doppler and found to have postvasectomy surgical changes consistent with bilateral spermatoceles, but no evidence of epididymitis or a mass. Before leaving the ED, Jason received ceftriaxone (125 mg IM) as gonorrhea prophylaxis. He was discharged home with prophylactic antibiotics for chlamydia, as well as ibuprofen. He was advised to avoid strenuous physical activity and told to follow-up with his FP if his symptoms did not improve.
During several months of conservative medical therapy, including trials of NSAIDs, quinolone antibiotics, TCAs, and gabapentin, Jason did not experience any significant pain relief. He was frustrated by the dull, aching pain in his scrotum that continued to limit his physical and sexual activities.
Finally, the FP recommended a urologic consultation.
Consider these minimally invasive procedures
When conservative medical management fails, minimally invasive techniques are the next step. There are 2 commonly used procedures, both of which can be performed by a urologist in an outpatient setting.
Spermatic cord blocks with lidocaine and methylprednisolone have been shown to provide relief for weeks up to several months in small case studies, and may be repeated at intervals of several months if modest relief is achieved.18,19
Transrectal ultrasound-guided periprostatic anesthetic injections, another microinvasive option, offers minimal risk and may provide some short-term relief. However, data on long-term benefit and resolution of pain and disability are lacking.20
Consider surgery only after all else fails
If all medical and conservative therapies have been tried and the patient continues to have debilitating pain, surgical options should be considered. Because current surgical therapies are not always effective and are not reversible (and research on the various options is limited), it is important to initiate a detailed discussion with the patient. Such conversations should be held in consultation with a urologist.
Highlight risks and benefits and provide realistic expectations of short- and long-term postsurgical outcomes. It is also important to address psychological factors and social stressors that often contribute to chronic pelvic pain syndromes, which can improve long-term outcomes regardless of the chosen treatment. For this reason, a referral to a psychiatrist may be indicated.
Microsurgical denervation of the spermatic cord. Removal of the afferent nerve stimulus to the testicle is believed to result in the downregulation of the peripheral and central nervous systems, so the patient no longer has the perception of testicular pain. Several small trials have yielded favorable symptomatic pain relief scores in up to 71% of patients, with reported adverse outcomes including rare testicular atrophy—but no complaints of hypoesthesia or hyperesthesia of the scrotum, penile shaft, inguinal, or medial thigh skin.21,22 This treatment should be considered only in patients who have experienced a significant degree of temporary relief from spermatic cord injection.
Epididymectomy is recommended only when pain is localized to the epididymis, as this is a testicle-sparing procedure. Unilateral or bilateral epididymectomy is a viable option for the treatment of chronic orchialgia related to postvasectomy pain syndrome or chronic epididymitis. Reports highlighting symptomatic improvement based on small case series range from 43% to 74%, with the highest success rate found during a 5½-year follow-up.23-25 In 1 study, 90% of patients reported that they were satisfied with their choice to undergo the procedure.25
Vasectomy reversal (vasovasostomy) and inguinal or scrotal orchiectomy should be considered only after all other treatment modalities have failed. Vasovasostomy has the potential to restore fertility in up to 98% of cases,26 which may or may not be desirable. One study of men who experienced post-vasectomy pain syndrome and underwent microsurgical vasovasostomy found that after nearly 2½ years, 84% experienced complete pain resolution.27
The goal of orchiectomy is to relieve orchialgia by releasing the entrapped ipsilateral genitofemoral and/or ilioinguinal nerves. One study determined that 90% of men who underwent unilateral epididymectomy for chronic orchialgia required an orchiectomy to resolve pain.1 Another study found that 80% of patients continued to suffer both short- and long-term debilitating orchialgia postorchiectomy.28
CASE 2 Jason H
Jason saw a urologist, who initially offered him bilateral spermatic cord blocks. They provided Jason with moderate symptom relief on most days of the week and allowed him to increase his physical and sexual activities. Three months later, Jason went back to the urologist for evaluation because he felt that the effects of the spermatic cord blocks had worn off. In the next 6 months, he had 2 additional bilateral blocks.
Nearly a year after a series of spermatic cord blocks, most of it spent in persistent discomfort, Jason returned to his FP with a request for narcotic pain medication. The FP tried to be supportive, but told Jason that chronic narcotic therapy was not an ideal choice—and referred him back to the urologist to discuss surgical options.
The urologist recommended a bilateral epididymectomy and the patient, who was desperate to obtain some pain relief and now regretted undergoing a vasectomy, agreed. Within the first few weeks after his surgery, he noticed a reduction in pain, and he slowly increased his physical activity. A year later, Jason reported only minimal testicular and scrotal discomfort that did not limit his physical or sexual activities—and he continues to be pleased with the outcome of his treatment.
CORRESPONDENCE Joel J. Heidelbaugh, MD, FAAFP, Ypsilanti Health Center, 200 Arnet, Suite 200, Ypsilanti, MI 48198; jheidel@umich.edu
1. Davis B, Noble MJ, Weigel JD, et al. Analysis and management of chronic testicular pain. J Urol. 1990;143:936-939.
2. Schover LR. Psychological factors in men with genital pain. Cleve Clin J Med. 1990;57:697-700.
3. Masarani M, Cox R. Th e aetiology, pathophysiology and management of chronic orchialgia. Br J Urol Int. 2003;91:435-437.
4. Granitsiotis P, Kirk D. Chronic testicular pain: an overview. Eur Urol. 2004;45:430-436.
5. Coogan CL. Painful scrotum. In: Myers JA, Millikan KW, Sacla-rides TJ, eds. Common Surgical Diseases. New York: Springer; 2008:293-295.
6. Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2006. MMWR Morb Mortal Wkly Rep. 2006;55(RR-11):1-94.
7. Newman LM, Moran JS, Workowski KA. Update on the management of gonorrhea in adults in the United States. Clin Infect Dis. 2007;44(suppl 3):S84-S101.
8. Wiffen PJ, McQuay HJ, Rees J, et al. Gabapentin for acute and chronic pain. Cochrane Database Syst Rev. 2005;(3):CD005452.-
9. American College of Obstetricians and Gynecologists. Chronic pelvic pain. ACOG Practice Bulletin No. 51. Obstet Gynecol. 2004;103:589-605.
10. Sinclair AM, Miller B, Lee LK. Chronic orchialgia: consider gabapentin or nortriptyline before considering surgery. Int J Urol. 2007;14:622-625.
11. Jarow JP, Budin RE, Dym M, et al. Quantitative pathologic changes in the human testis after vasectomy. N Engl J Med. 1985;313:1252-1256.
12. Choe J, Kirkemo A. Questionnaire-based outcomes study of nononcological post-vasectomy complications. J Urol. 1996;155:1284-1286.
13. McMahon A, Buckley J, Taylor A, et al. Chronic testicular pain following vasectomy. Br J Urol. 1992;69:188-191.
14. Ahmed I, Rasheed S, White C, et al. Th e incidence of post-vasectomy chronic testicular pain and the role of nerve stripping (denervation) of the spermatic cord in its management. Br J Urol. 1997;79:269-270.
15. Christiansen CG, Sandlow JI. Testicular pain following vasectomy: a review of postvasectomy pain syndrome. J Androl. 2003;24:293-298.
16. Shapiro EI, Silber SJ. Open-ended vasectomy, sperm granuloma, and postvasectomy orchialgia. Fertil Steril. 1979;32:546-550.
17. Taxy JB, Marshall FF, Erlickman RJ. Vasectomy: subclinical pathologic changes. Am J Surg Pathol. 1981;5:767-772.
18. Fuchs E. Cord block anesthesia for scrotal surgery. J Urol. 1982;128:718-719.
19. Issa M, Hsiao K, Bassel Y, et al. Spermatic cord anesthesia block for scrotal procedures in the outpatient clinic setting. J Urol. 2004;172:2358-2361.
20. Zorn B, Rauchenwald M, Steers WD. Periprostatic injection of local anesthesia for relief of chronic orchialgia. J Urol. 1994;151:411,A735.-
21. Levine LA, Matkov TG, Lubenow TR. Microsurgical denervation of the spermatic cord: a surgical alternative in the treatment of chronic orchialgia. J Urol. 1996;155:1005-1007.
22. Strom KH, Levine LA. Microsurgical denervation of the spermatic cord for chronic orchialgia: long-term results from a single center. J Urol. 2008;180:949-953.
23. Padmore DE, Norman RW, Millard OH. Analyses of indications for and outcomes of epdidymectomy. J Urol. 1996;156:95-96.
24. West AF, Leung HY, Powell PH. Epididymectomy is an effective treatment for scrotal pain after vasectomy. Br J Urol Int. 2000;85:1097-1099.
25. Siu W, Ohl DA, Schuster TG. Long-term follow-up after epididymectomy for chronic epidiymal pain. Urology. 2007;70:333-336.
26. Patel SR, Sigman M. Comparison of outcomes of vasovasostomy performed in the convoluted and straight vas deferens. J Urol. 2008;179:256-259.
27. Myers SA, Mershon CE, Fuchs EF. Vasectomy reversal for treatment of the post-vasectomy pain syndrome. J Urol. 1997;157:518-520.
28. Costabile RA, Hahn M, McLeod DG. Chronic orchialgia in the pain prone patient: the clinical perspective. J Urol. 1991;146:1571-1574.
Order ultrasound of the scrotum and testes to evaluate chronic testicular pain, with color Doppler to identify areas of hypervascularity. C
Treat suspected epididymitis with empiric coverage for chlamydia with either a 10-day regimen of doxycycline (100 mg twice daily) or a single dose (1 g) of azithromycin; treat suspected gonorrhea with a single intramuscular injection (125 mg) of ceftriaxone. A
Do not treat small epididymal cysts that do not correlate with testicular pain; larger, painful cysts can be aspirated, injected with a sclerosing agent, or surgically excised. C
Consider surgical options only after medical and conservative therapies have failed to alleviate chronic testicular pain. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE 1 Vincent B, a 33-year-old executive, visits his family physician for an evaluation of chronic orchialgia. Although his testicular pain has waxed and waned for several years, it has recently worsened, making it increasingly difficult for him to exercise or to sit for extended periods of time. In fact, this visit was prompted by a lengthy meeting during which he developed a “dull ache” that did not let up until he left the meeting and walked around.
CASE 2 Jason H, a 42-year-old married father of 3 who had a vasectomy 2 years ago, has had progressively worsening testicular pain ever since. He also has occasional pain after ejaculation, but no known hematospermia. Recently, the pain has become so bad that it limits both his physical and sexual activities and is having a negative effect on his relationship with his wife. Jason is sexually monogamous, has no significant medical history, and takes no prescription medications.
These 2 cases are based on actual patients we have seen in our practices. If Vincent and Jason (not their real names) were your patients, how would you initiate a work-up for testicular pain? What treatments would you offer? And at what point would you consider a referral to a urologist?
Chronic orchialgia is a complex urogenital focal pain syndrome in which neurogenic inflammation is the principal mediator. This debilitating condition is associated with substantial anxiety and frustration, and is characterized by intermittent or constant unilateral or bilateral testicular pain, occurring for at least 3 months, that has a significant negative impact on activities of daily living and physical activity.1
A variety of procedural and surgical options may help to minimize or alleviate chronic orchialgia. But which approach is best? There are no evidence-based guidelines for the treatment of this condition, and no randomized controlled trials to demonstrate the superiority of 1 modality over another. All diagnostic and treatment recommendations are based on expert opinion derived from small cohort studies.
With that in mind, we conducted a systematic review of the literature evaluating medical and surgical therapies for chronic testicular pain—and developed an algorithm (FIGURE 1), along with the text and TABLE that follow, for family physicians (FPs) to use as a guide.
FIGURE 1
Chronic orchialgia: A diagnosis and treatment algorithm1,3,4,6,10
NSAID, nonsteroidal anti-inflammatory drug; STIs, sexually transmitted infections.
CASE 1 Vincent B
Over the last few years, Vincent has had similar episodes of bilateral testicular pain. He denies any history of direct trauma to the testicles, and he works out regularly by lifting weights and running. When the pain becomes unbearable, he takes acetaminophen or ibuprofen and takes a few days off from exercising, which provides modest—but temporary—relief.
Vincent reports that he has had about a dozen lifetime sexual partners and had chlamydia over a decade ago as a college student. He is currently engaged and sexually monogamous, and tested negative for Chlamydia trachomatis, Neisseria gonorrhoeae, hepatitis, syphilis, and human immunodeficiency virus (HIV) at his annual health maintenance examination last month. Shortly before that, Vincent was treated empirically for epididymitis with a 4-week course of ciprofloxacin, with no significant improvement in symptoms. He has no significant past medical history, denies depression, and takes no prescription medications.
Physical examination reveals mild to moderate diffuse tenderness to palpation throughout the scrotum, including both testicles and spermatic cords. There is no erythema of the scrotum. Nor are there any palpable scrotal masses, varicoceles, or hydroceles; testicular, scrotal, or penile lesions; inguinal masses; or lymph nodes. His urethral meatus is patent. The prostate is smooth, nonnodular, and non-tender. The remainder of the physical exam is unremarkable.
Determining a cause can be a challenge
There are numerous possible causes of testicular pain (TABLE), including an inguinal hernia, torsion of the testicle, trauma, and a history of chlamydia or gonorrhea, to name a few.
TABLE
Causes of acute and chronic orchialgia1,3,4
Acute
|
Chronic
|
Chronic testicular pain can also be psychogenic, often relating to a history of sexual abuse or relationship stress. One study examining comorbid psychological conditions in men with chronic orchialgia identified a somatization disorder in 56% of the patients, nongenital chronic pain syndromes in 50%, and major depression or chemical dependency in 27%.2 Overall, however, estimates suggest that in about 25% of patients with chronic orchialgia, no identifiable etiology is found. 1
Establish a baseline with a physical exam
Conduct a physical examination of the scrotum, testes, spermatic cords, penis, inguinal region, and prostate as a baseline measurement in a patient who presents with chronic orchialgia.3,4 An initial urinalysis should be performed to rule out infection or identify microscopic hematuria, which may prompt a more targeted work-up and therapeutic plan. Take a thorough medical and psychosocial/ sexual history, as well.
Order an ultrasound of the scrotum and testes, the accepted gold standard to highlight structural abnormalities of the testicles. The addition of color Doppler makes it possible to find areas of hypervascularity, an indication of inflammation in the testicle and epididymis (FIGURES 2A AND B).
FIGURE 2
Well-circumscribed extratesticular mass
In the image at left, ultrasound reveals an anechoic mass (arrows), representing either an epididymal cyst or spermatocele, superior to the testicle (T). A color Doppler image (right) reveals increased vascularity to the epididymis (E), as compared with the testicle.
Epididymal cysts are common findings on scrotal ultrasound; they are frequently incidental, but may relate to the patient’s pain, depending on the size of the cyst. Smaller cysts that do not correlate with pain do not require treatment. Larger, painful cysts can be treated with aspiration or injection with a sclerosing agent—or with surgical excision, which offers the highest potential cure rate.3,4 A computed tomography (CT) scan without contrast is the best way to find genitourinary system calculi, which could be the source of referred renal pain to the groin and scrotum. A contrast-enhanced CT is best to evaluate for solid renal masses.
Start with the most conservative treatment
In the absence of any findings that require surgical intervention, start conservatively.
Initiate a trial of nonsteroidal anti-inflammatory drugs (NSAIDs) for at least 1 month. Although this is the standard first-line treatment, NSAIDs have been shown to help only a small percentage of patients with chronic orchialgia, and only on a short-term basis.1,3,4
Recommend scrotal elevation with supportive undergarments to decrease venous congestion. Tell the patient, too, that modifying his seated posture to avoid scrotal pressure may alleviate pain and poses no discernible risk of worsening orchialgia.5
Treat suspected STIs. The Centers for Disease Control and Prevention report that in men 14 to 35 years of age, epididymitis is most commonly caused by chlamydia or gonorrhea.6 In males younger than 14 or older than 35, epididymitis is most commonly caused by urinary coliform pathogens, including Eschericia coli.
If epididymitis is suspected to be due to chlamydia or gonorrhea, treatment should include either doxycycline 100 mg orally twice daily for 10 days or a single dose of azithromycin 1 g orally (for chlamydia eradication) and a single dose of ceftriaxone 125 mg intramuscularly (for gonorrhea eradication).6,7 If coliform bacteria is suspected, order a standard dose of a quinolone (eg, ciprofloxacin or levofloxacin 500 mg/d) for 10 days.6 For refractory cases, treatment with a standard dose of a quinolone for 4 weeks is recommended.6
It is generally reasonable to treat most patients empirically for suspected epididymitis with antibiotics if no other identifiable etiology can be determined. Multiple antibiotic treatments should be avoided, however, in the absence of either an identifiable urogenital infection or ultrasound findings consistent with epididymitis (eg, congestion and enlargement). Antibiotics have not been shown to decrease the severity of chronic orchialgia and their use, unless clearly indicated, may lead to drug resistance.3
Consider a tricyclic antidepressant or gabapentin
Both tricyclic antidepressants (TCAs) and gabapentin have demonstrated benefit in the treatment of chronic pelvic and neuropathic pain.8,9 Doses should be titrated to achieve a maximal therapeutic benefit while avoiding anticholinergic and neurologic side effects.
A cohort study using a multidisciplinary team consisting of a psychologist, an anesthetist, a physiotherapist, and an occupational therapist found >50% symptomatic improvement in 62% of men with chronic orchialgia treated with gabapentin up to 1800 mg per day, and 67% of men treated with nortriptyline up to 150 mg per day.10
However, a subgroup of patients who reported postvasectomy testicular pain did not achieve a 50% symptomatic improvement rate with either TCA or gabapentin therapy.
CASE 1 Vincent B
The FP reassured Vincent that his physical examination was normal and recommended a 1-month trial of ibuprofen (600 mg every 6 hours), and regular use of supportive briefs. Since the patient had been treated with antibiotics in the past with no change in symptoms—and because he was thought to be at low risk for an STI—the physician did not prescribe another empiric trial of antibiotics. He did send the patient for an ultrasound evaluation of the scrotum and testes, which revealed only a 0.5 × 0.4 × 0.6-cm right epididymal cyst that was not palpable on examination.
The patient returned after 1 month, noting that his symptoms had neither improved nor worsened. The FP suggested that he stop taking the ibuprofen and begin a trial of gabapentin 100 mg daily, titrating up to 3 times daily for the first month, then to 300 mg 3 times daily in the second month.
When he returned 3 months later, Vincent reported that his symptoms had improved by about 50%. He has since been able to increase both the intensity and frequency of physical activity. Vincent is not interested in further increasing the dose of gabapentin and declined a referral to a urologist for consideration of procedural and surgical therapeutic options, but agreed to follow up as needed if his testicular pain worsened.
Postvasectomy pain is not unusual
Several years after a vasectomy, the diameter of a man’s ejaculatory ducts often doubles in size to counteract the increase in fluid pressure.11 The specific cause of long-term post-vasectomy pain syndrome, or congestive epididymitis, is unknown, but has been reported in 5% to 43% of men who have undergone this procedure.12-14 Sperm granulomas or spermatoceles represent the body’s effort to spare the testicle from damage secondary to increasing fluid pressure. While these granulomas are benign lesions, their presence may predispose a man to postvasectomy pain syndrome.15-17
CASE 2 Jason H
Two months before Jason’s visit to the FP, his testicular pain had become so excrutiating that he went to the ED seeking treatment. He was given an ultrasound with color Doppler and found to have postvasectomy surgical changes consistent with bilateral spermatoceles, but no evidence of epididymitis or a mass. Before leaving the ED, Jason received ceftriaxone (125 mg IM) as gonorrhea prophylaxis. He was discharged home with prophylactic antibiotics for chlamydia, as well as ibuprofen. He was advised to avoid strenuous physical activity and told to follow-up with his FP if his symptoms did not improve.
During several months of conservative medical therapy, including trials of NSAIDs, quinolone antibiotics, TCAs, and gabapentin, Jason did not experience any significant pain relief. He was frustrated by the dull, aching pain in his scrotum that continued to limit his physical and sexual activities.
Finally, the FP recommended a urologic consultation.
Consider these minimally invasive procedures
When conservative medical management fails, minimally invasive techniques are the next step. There are 2 commonly used procedures, both of which can be performed by a urologist in an outpatient setting.
Spermatic cord blocks with lidocaine and methylprednisolone have been shown to provide relief for weeks up to several months in small case studies, and may be repeated at intervals of several months if modest relief is achieved.18,19
Transrectal ultrasound-guided periprostatic anesthetic injections, another microinvasive option, offers minimal risk and may provide some short-term relief. However, data on long-term benefit and resolution of pain and disability are lacking.20
Consider surgery only after all else fails
If all medical and conservative therapies have been tried and the patient continues to have debilitating pain, surgical options should be considered. Because current surgical therapies are not always effective and are not reversible (and research on the various options is limited), it is important to initiate a detailed discussion with the patient. Such conversations should be held in consultation with a urologist.
Highlight risks and benefits and provide realistic expectations of short- and long-term postsurgical outcomes. It is also important to address psychological factors and social stressors that often contribute to chronic pelvic pain syndromes, which can improve long-term outcomes regardless of the chosen treatment. For this reason, a referral to a psychiatrist may be indicated.
Microsurgical denervation of the spermatic cord. Removal of the afferent nerve stimulus to the testicle is believed to result in the downregulation of the peripheral and central nervous systems, so the patient no longer has the perception of testicular pain. Several small trials have yielded favorable symptomatic pain relief scores in up to 71% of patients, with reported adverse outcomes including rare testicular atrophy—but no complaints of hypoesthesia or hyperesthesia of the scrotum, penile shaft, inguinal, or medial thigh skin.21,22 This treatment should be considered only in patients who have experienced a significant degree of temporary relief from spermatic cord injection.
Epididymectomy is recommended only when pain is localized to the epididymis, as this is a testicle-sparing procedure. Unilateral or bilateral epididymectomy is a viable option for the treatment of chronic orchialgia related to postvasectomy pain syndrome or chronic epididymitis. Reports highlighting symptomatic improvement based on small case series range from 43% to 74%, with the highest success rate found during a 5½-year follow-up.23-25 In 1 study, 90% of patients reported that they were satisfied with their choice to undergo the procedure.25
Vasectomy reversal (vasovasostomy) and inguinal or scrotal orchiectomy should be considered only after all other treatment modalities have failed. Vasovasostomy has the potential to restore fertility in up to 98% of cases,26 which may or may not be desirable. One study of men who experienced post-vasectomy pain syndrome and underwent microsurgical vasovasostomy found that after nearly 2½ years, 84% experienced complete pain resolution.27
The goal of orchiectomy is to relieve orchialgia by releasing the entrapped ipsilateral genitofemoral and/or ilioinguinal nerves. One study determined that 90% of men who underwent unilateral epididymectomy for chronic orchialgia required an orchiectomy to resolve pain.1 Another study found that 80% of patients continued to suffer both short- and long-term debilitating orchialgia postorchiectomy.28
CASE 2 Jason H
Jason saw a urologist, who initially offered him bilateral spermatic cord blocks. They provided Jason with moderate symptom relief on most days of the week and allowed him to increase his physical and sexual activities. Three months later, Jason went back to the urologist for evaluation because he felt that the effects of the spermatic cord blocks had worn off. In the next 6 months, he had 2 additional bilateral blocks.
Nearly a year after a series of spermatic cord blocks, most of it spent in persistent discomfort, Jason returned to his FP with a request for narcotic pain medication. The FP tried to be supportive, but told Jason that chronic narcotic therapy was not an ideal choice—and referred him back to the urologist to discuss surgical options.
The urologist recommended a bilateral epididymectomy and the patient, who was desperate to obtain some pain relief and now regretted undergoing a vasectomy, agreed. Within the first few weeks after his surgery, he noticed a reduction in pain, and he slowly increased his physical activity. A year later, Jason reported only minimal testicular and scrotal discomfort that did not limit his physical or sexual activities—and he continues to be pleased with the outcome of his treatment.
CORRESPONDENCE Joel J. Heidelbaugh, MD, FAAFP, Ypsilanti Health Center, 200 Arnet, Suite 200, Ypsilanti, MI 48198; jheidel@umich.edu
Order ultrasound of the scrotum and testes to evaluate chronic testicular pain, with color Doppler to identify areas of hypervascularity. C
Treat suspected epididymitis with empiric coverage for chlamydia with either a 10-day regimen of doxycycline (100 mg twice daily) or a single dose (1 g) of azithromycin; treat suspected gonorrhea with a single intramuscular injection (125 mg) of ceftriaxone. A
Do not treat small epididymal cysts that do not correlate with testicular pain; larger, painful cysts can be aspirated, injected with a sclerosing agent, or surgically excised. C
Consider surgical options only after medical and conservative therapies have failed to alleviate chronic testicular pain. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE 1 Vincent B, a 33-year-old executive, visits his family physician for an evaluation of chronic orchialgia. Although his testicular pain has waxed and waned for several years, it has recently worsened, making it increasingly difficult for him to exercise or to sit for extended periods of time. In fact, this visit was prompted by a lengthy meeting during which he developed a “dull ache” that did not let up until he left the meeting and walked around.
CASE 2 Jason H, a 42-year-old married father of 3 who had a vasectomy 2 years ago, has had progressively worsening testicular pain ever since. He also has occasional pain after ejaculation, but no known hematospermia. Recently, the pain has become so bad that it limits both his physical and sexual activities and is having a negative effect on his relationship with his wife. Jason is sexually monogamous, has no significant medical history, and takes no prescription medications.
These 2 cases are based on actual patients we have seen in our practices. If Vincent and Jason (not their real names) were your patients, how would you initiate a work-up for testicular pain? What treatments would you offer? And at what point would you consider a referral to a urologist?
Chronic orchialgia is a complex urogenital focal pain syndrome in which neurogenic inflammation is the principal mediator. This debilitating condition is associated with substantial anxiety and frustration, and is characterized by intermittent or constant unilateral or bilateral testicular pain, occurring for at least 3 months, that has a significant negative impact on activities of daily living and physical activity.1
A variety of procedural and surgical options may help to minimize or alleviate chronic orchialgia. But which approach is best? There are no evidence-based guidelines for the treatment of this condition, and no randomized controlled trials to demonstrate the superiority of 1 modality over another. All diagnostic and treatment recommendations are based on expert opinion derived from small cohort studies.
With that in mind, we conducted a systematic review of the literature evaluating medical and surgical therapies for chronic testicular pain—and developed an algorithm (FIGURE 1), along with the text and TABLE that follow, for family physicians (FPs) to use as a guide.
FIGURE 1
Chronic orchialgia: A diagnosis and treatment algorithm1,3,4,6,10
NSAID, nonsteroidal anti-inflammatory drug; STIs, sexually transmitted infections.
CASE 1 Vincent B
Over the last few years, Vincent has had similar episodes of bilateral testicular pain. He denies any history of direct trauma to the testicles, and he works out regularly by lifting weights and running. When the pain becomes unbearable, he takes acetaminophen or ibuprofen and takes a few days off from exercising, which provides modest—but temporary—relief.
Vincent reports that he has had about a dozen lifetime sexual partners and had chlamydia over a decade ago as a college student. He is currently engaged and sexually monogamous, and tested negative for Chlamydia trachomatis, Neisseria gonorrhoeae, hepatitis, syphilis, and human immunodeficiency virus (HIV) at his annual health maintenance examination last month. Shortly before that, Vincent was treated empirically for epididymitis with a 4-week course of ciprofloxacin, with no significant improvement in symptoms. He has no significant past medical history, denies depression, and takes no prescription medications.
Physical examination reveals mild to moderate diffuse tenderness to palpation throughout the scrotum, including both testicles and spermatic cords. There is no erythema of the scrotum. Nor are there any palpable scrotal masses, varicoceles, or hydroceles; testicular, scrotal, or penile lesions; inguinal masses; or lymph nodes. His urethral meatus is patent. The prostate is smooth, nonnodular, and non-tender. The remainder of the physical exam is unremarkable.
Determining a cause can be a challenge
There are numerous possible causes of testicular pain (TABLE), including an inguinal hernia, torsion of the testicle, trauma, and a history of chlamydia or gonorrhea, to name a few.
TABLE
Causes of acute and chronic orchialgia1,3,4
Acute
|
Chronic
|
Chronic testicular pain can also be psychogenic, often relating to a history of sexual abuse or relationship stress. One study examining comorbid psychological conditions in men with chronic orchialgia identified a somatization disorder in 56% of the patients, nongenital chronic pain syndromes in 50%, and major depression or chemical dependency in 27%.2 Overall, however, estimates suggest that in about 25% of patients with chronic orchialgia, no identifiable etiology is found. 1
Establish a baseline with a physical exam
Conduct a physical examination of the scrotum, testes, spermatic cords, penis, inguinal region, and prostate as a baseline measurement in a patient who presents with chronic orchialgia.3,4 An initial urinalysis should be performed to rule out infection or identify microscopic hematuria, which may prompt a more targeted work-up and therapeutic plan. Take a thorough medical and psychosocial/ sexual history, as well.
Order an ultrasound of the scrotum and testes, the accepted gold standard to highlight structural abnormalities of the testicles. The addition of color Doppler makes it possible to find areas of hypervascularity, an indication of inflammation in the testicle and epididymis (FIGURES 2A AND B).
FIGURE 2
Well-circumscribed extratesticular mass
In the image at left, ultrasound reveals an anechoic mass (arrows), representing either an epididymal cyst or spermatocele, superior to the testicle (T). A color Doppler image (right) reveals increased vascularity to the epididymis (E), as compared with the testicle.
Epididymal cysts are common findings on scrotal ultrasound; they are frequently incidental, but may relate to the patient’s pain, depending on the size of the cyst. Smaller cysts that do not correlate with pain do not require treatment. Larger, painful cysts can be treated with aspiration or injection with a sclerosing agent—or with surgical excision, which offers the highest potential cure rate.3,4 A computed tomography (CT) scan without contrast is the best way to find genitourinary system calculi, which could be the source of referred renal pain to the groin and scrotum. A contrast-enhanced CT is best to evaluate for solid renal masses.
Start with the most conservative treatment
In the absence of any findings that require surgical intervention, start conservatively.
Initiate a trial of nonsteroidal anti-inflammatory drugs (NSAIDs) for at least 1 month. Although this is the standard first-line treatment, NSAIDs have been shown to help only a small percentage of patients with chronic orchialgia, and only on a short-term basis.1,3,4
Recommend scrotal elevation with supportive undergarments to decrease venous congestion. Tell the patient, too, that modifying his seated posture to avoid scrotal pressure may alleviate pain and poses no discernible risk of worsening orchialgia.5
Treat suspected STIs. The Centers for Disease Control and Prevention report that in men 14 to 35 years of age, epididymitis is most commonly caused by chlamydia or gonorrhea.6 In males younger than 14 or older than 35, epididymitis is most commonly caused by urinary coliform pathogens, including Eschericia coli.
If epididymitis is suspected to be due to chlamydia or gonorrhea, treatment should include either doxycycline 100 mg orally twice daily for 10 days or a single dose of azithromycin 1 g orally (for chlamydia eradication) and a single dose of ceftriaxone 125 mg intramuscularly (for gonorrhea eradication).6,7 If coliform bacteria is suspected, order a standard dose of a quinolone (eg, ciprofloxacin or levofloxacin 500 mg/d) for 10 days.6 For refractory cases, treatment with a standard dose of a quinolone for 4 weeks is recommended.6
It is generally reasonable to treat most patients empirically for suspected epididymitis with antibiotics if no other identifiable etiology can be determined. Multiple antibiotic treatments should be avoided, however, in the absence of either an identifiable urogenital infection or ultrasound findings consistent with epididymitis (eg, congestion and enlargement). Antibiotics have not been shown to decrease the severity of chronic orchialgia and their use, unless clearly indicated, may lead to drug resistance.3
Consider a tricyclic antidepressant or gabapentin
Both tricyclic antidepressants (TCAs) and gabapentin have demonstrated benefit in the treatment of chronic pelvic and neuropathic pain.8,9 Doses should be titrated to achieve a maximal therapeutic benefit while avoiding anticholinergic and neurologic side effects.
A cohort study using a multidisciplinary team consisting of a psychologist, an anesthetist, a physiotherapist, and an occupational therapist found >50% symptomatic improvement in 62% of men with chronic orchialgia treated with gabapentin up to 1800 mg per day, and 67% of men treated with nortriptyline up to 150 mg per day.10
However, a subgroup of patients who reported postvasectomy testicular pain did not achieve a 50% symptomatic improvement rate with either TCA or gabapentin therapy.
CASE 1 Vincent B
The FP reassured Vincent that his physical examination was normal and recommended a 1-month trial of ibuprofen (600 mg every 6 hours), and regular use of supportive briefs. Since the patient had been treated with antibiotics in the past with no change in symptoms—and because he was thought to be at low risk for an STI—the physician did not prescribe another empiric trial of antibiotics. He did send the patient for an ultrasound evaluation of the scrotum and testes, which revealed only a 0.5 × 0.4 × 0.6-cm right epididymal cyst that was not palpable on examination.
The patient returned after 1 month, noting that his symptoms had neither improved nor worsened. The FP suggested that he stop taking the ibuprofen and begin a trial of gabapentin 100 mg daily, titrating up to 3 times daily for the first month, then to 300 mg 3 times daily in the second month.
When he returned 3 months later, Vincent reported that his symptoms had improved by about 50%. He has since been able to increase both the intensity and frequency of physical activity. Vincent is not interested in further increasing the dose of gabapentin and declined a referral to a urologist for consideration of procedural and surgical therapeutic options, but agreed to follow up as needed if his testicular pain worsened.
Postvasectomy pain is not unusual
Several years after a vasectomy, the diameter of a man’s ejaculatory ducts often doubles in size to counteract the increase in fluid pressure.11 The specific cause of long-term post-vasectomy pain syndrome, or congestive epididymitis, is unknown, but has been reported in 5% to 43% of men who have undergone this procedure.12-14 Sperm granulomas or spermatoceles represent the body’s effort to spare the testicle from damage secondary to increasing fluid pressure. While these granulomas are benign lesions, their presence may predispose a man to postvasectomy pain syndrome.15-17
CASE 2 Jason H
Two months before Jason’s visit to the FP, his testicular pain had become so excrutiating that he went to the ED seeking treatment. He was given an ultrasound with color Doppler and found to have postvasectomy surgical changes consistent with bilateral spermatoceles, but no evidence of epididymitis or a mass. Before leaving the ED, Jason received ceftriaxone (125 mg IM) as gonorrhea prophylaxis. He was discharged home with prophylactic antibiotics for chlamydia, as well as ibuprofen. He was advised to avoid strenuous physical activity and told to follow-up with his FP if his symptoms did not improve.
During several months of conservative medical therapy, including trials of NSAIDs, quinolone antibiotics, TCAs, and gabapentin, Jason did not experience any significant pain relief. He was frustrated by the dull, aching pain in his scrotum that continued to limit his physical and sexual activities.
Finally, the FP recommended a urologic consultation.
Consider these minimally invasive procedures
When conservative medical management fails, minimally invasive techniques are the next step. There are 2 commonly used procedures, both of which can be performed by a urologist in an outpatient setting.
Spermatic cord blocks with lidocaine and methylprednisolone have been shown to provide relief for weeks up to several months in small case studies, and may be repeated at intervals of several months if modest relief is achieved.18,19
Transrectal ultrasound-guided periprostatic anesthetic injections, another microinvasive option, offers minimal risk and may provide some short-term relief. However, data on long-term benefit and resolution of pain and disability are lacking.20
Consider surgery only after all else fails
If all medical and conservative therapies have been tried and the patient continues to have debilitating pain, surgical options should be considered. Because current surgical therapies are not always effective and are not reversible (and research on the various options is limited), it is important to initiate a detailed discussion with the patient. Such conversations should be held in consultation with a urologist.
Highlight risks and benefits and provide realistic expectations of short- and long-term postsurgical outcomes. It is also important to address psychological factors and social stressors that often contribute to chronic pelvic pain syndromes, which can improve long-term outcomes regardless of the chosen treatment. For this reason, a referral to a psychiatrist may be indicated.
Microsurgical denervation of the spermatic cord. Removal of the afferent nerve stimulus to the testicle is believed to result in the downregulation of the peripheral and central nervous systems, so the patient no longer has the perception of testicular pain. Several small trials have yielded favorable symptomatic pain relief scores in up to 71% of patients, with reported adverse outcomes including rare testicular atrophy—but no complaints of hypoesthesia or hyperesthesia of the scrotum, penile shaft, inguinal, or medial thigh skin.21,22 This treatment should be considered only in patients who have experienced a significant degree of temporary relief from spermatic cord injection.
Epididymectomy is recommended only when pain is localized to the epididymis, as this is a testicle-sparing procedure. Unilateral or bilateral epididymectomy is a viable option for the treatment of chronic orchialgia related to postvasectomy pain syndrome or chronic epididymitis. Reports highlighting symptomatic improvement based on small case series range from 43% to 74%, with the highest success rate found during a 5½-year follow-up.23-25 In 1 study, 90% of patients reported that they were satisfied with their choice to undergo the procedure.25
Vasectomy reversal (vasovasostomy) and inguinal or scrotal orchiectomy should be considered only after all other treatment modalities have failed. Vasovasostomy has the potential to restore fertility in up to 98% of cases,26 which may or may not be desirable. One study of men who experienced post-vasectomy pain syndrome and underwent microsurgical vasovasostomy found that after nearly 2½ years, 84% experienced complete pain resolution.27
The goal of orchiectomy is to relieve orchialgia by releasing the entrapped ipsilateral genitofemoral and/or ilioinguinal nerves. One study determined that 90% of men who underwent unilateral epididymectomy for chronic orchialgia required an orchiectomy to resolve pain.1 Another study found that 80% of patients continued to suffer both short- and long-term debilitating orchialgia postorchiectomy.28
CASE 2 Jason H
Jason saw a urologist, who initially offered him bilateral spermatic cord blocks. They provided Jason with moderate symptom relief on most days of the week and allowed him to increase his physical and sexual activities. Three months later, Jason went back to the urologist for evaluation because he felt that the effects of the spermatic cord blocks had worn off. In the next 6 months, he had 2 additional bilateral blocks.
Nearly a year after a series of spermatic cord blocks, most of it spent in persistent discomfort, Jason returned to his FP with a request for narcotic pain medication. The FP tried to be supportive, but told Jason that chronic narcotic therapy was not an ideal choice—and referred him back to the urologist to discuss surgical options.
The urologist recommended a bilateral epididymectomy and the patient, who was desperate to obtain some pain relief and now regretted undergoing a vasectomy, agreed. Within the first few weeks after his surgery, he noticed a reduction in pain, and he slowly increased his physical activity. A year later, Jason reported only minimal testicular and scrotal discomfort that did not limit his physical or sexual activities—and he continues to be pleased with the outcome of his treatment.
CORRESPONDENCE Joel J. Heidelbaugh, MD, FAAFP, Ypsilanti Health Center, 200 Arnet, Suite 200, Ypsilanti, MI 48198; jheidel@umich.edu
1. Davis B, Noble MJ, Weigel JD, et al. Analysis and management of chronic testicular pain. J Urol. 1990;143:936-939.
2. Schover LR. Psychological factors in men with genital pain. Cleve Clin J Med. 1990;57:697-700.
3. Masarani M, Cox R. Th e aetiology, pathophysiology and management of chronic orchialgia. Br J Urol Int. 2003;91:435-437.
4. Granitsiotis P, Kirk D. Chronic testicular pain: an overview. Eur Urol. 2004;45:430-436.
5. Coogan CL. Painful scrotum. In: Myers JA, Millikan KW, Sacla-rides TJ, eds. Common Surgical Diseases. New York: Springer; 2008:293-295.
6. Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2006. MMWR Morb Mortal Wkly Rep. 2006;55(RR-11):1-94.
7. Newman LM, Moran JS, Workowski KA. Update on the management of gonorrhea in adults in the United States. Clin Infect Dis. 2007;44(suppl 3):S84-S101.
8. Wiffen PJ, McQuay HJ, Rees J, et al. Gabapentin for acute and chronic pain. Cochrane Database Syst Rev. 2005;(3):CD005452.-
9. American College of Obstetricians and Gynecologists. Chronic pelvic pain. ACOG Practice Bulletin No. 51. Obstet Gynecol. 2004;103:589-605.
10. Sinclair AM, Miller B, Lee LK. Chronic orchialgia: consider gabapentin or nortriptyline before considering surgery. Int J Urol. 2007;14:622-625.
11. Jarow JP, Budin RE, Dym M, et al. Quantitative pathologic changes in the human testis after vasectomy. N Engl J Med. 1985;313:1252-1256.
12. Choe J, Kirkemo A. Questionnaire-based outcomes study of nononcological post-vasectomy complications. J Urol. 1996;155:1284-1286.
13. McMahon A, Buckley J, Taylor A, et al. Chronic testicular pain following vasectomy. Br J Urol. 1992;69:188-191.
14. Ahmed I, Rasheed S, White C, et al. Th e incidence of post-vasectomy chronic testicular pain and the role of nerve stripping (denervation) of the spermatic cord in its management. Br J Urol. 1997;79:269-270.
15. Christiansen CG, Sandlow JI. Testicular pain following vasectomy: a review of postvasectomy pain syndrome. J Androl. 2003;24:293-298.
16. Shapiro EI, Silber SJ. Open-ended vasectomy, sperm granuloma, and postvasectomy orchialgia. Fertil Steril. 1979;32:546-550.
17. Taxy JB, Marshall FF, Erlickman RJ. Vasectomy: subclinical pathologic changes. Am J Surg Pathol. 1981;5:767-772.
18. Fuchs E. Cord block anesthesia for scrotal surgery. J Urol. 1982;128:718-719.
19. Issa M, Hsiao K, Bassel Y, et al. Spermatic cord anesthesia block for scrotal procedures in the outpatient clinic setting. J Urol. 2004;172:2358-2361.
20. Zorn B, Rauchenwald M, Steers WD. Periprostatic injection of local anesthesia for relief of chronic orchialgia. J Urol. 1994;151:411,A735.-
21. Levine LA, Matkov TG, Lubenow TR. Microsurgical denervation of the spermatic cord: a surgical alternative in the treatment of chronic orchialgia. J Urol. 1996;155:1005-1007.
22. Strom KH, Levine LA. Microsurgical denervation of the spermatic cord for chronic orchialgia: long-term results from a single center. J Urol. 2008;180:949-953.
23. Padmore DE, Norman RW, Millard OH. Analyses of indications for and outcomes of epdidymectomy. J Urol. 1996;156:95-96.
24. West AF, Leung HY, Powell PH. Epididymectomy is an effective treatment for scrotal pain after vasectomy. Br J Urol Int. 2000;85:1097-1099.
25. Siu W, Ohl DA, Schuster TG. Long-term follow-up after epididymectomy for chronic epidiymal pain. Urology. 2007;70:333-336.
26. Patel SR, Sigman M. Comparison of outcomes of vasovasostomy performed in the convoluted and straight vas deferens. J Urol. 2008;179:256-259.
27. Myers SA, Mershon CE, Fuchs EF. Vasectomy reversal for treatment of the post-vasectomy pain syndrome. J Urol. 1997;157:518-520.
28. Costabile RA, Hahn M, McLeod DG. Chronic orchialgia in the pain prone patient: the clinical perspective. J Urol. 1991;146:1571-1574.
1. Davis B, Noble MJ, Weigel JD, et al. Analysis and management of chronic testicular pain. J Urol. 1990;143:936-939.
2. Schover LR. Psychological factors in men with genital pain. Cleve Clin J Med. 1990;57:697-700.
3. Masarani M, Cox R. Th e aetiology, pathophysiology and management of chronic orchialgia. Br J Urol Int. 2003;91:435-437.
4. Granitsiotis P, Kirk D. Chronic testicular pain: an overview. Eur Urol. 2004;45:430-436.
5. Coogan CL. Painful scrotum. In: Myers JA, Millikan KW, Sacla-rides TJ, eds. Common Surgical Diseases. New York: Springer; 2008:293-295.
6. Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2006. MMWR Morb Mortal Wkly Rep. 2006;55(RR-11):1-94.
7. Newman LM, Moran JS, Workowski KA. Update on the management of gonorrhea in adults in the United States. Clin Infect Dis. 2007;44(suppl 3):S84-S101.
8. Wiffen PJ, McQuay HJ, Rees J, et al. Gabapentin for acute and chronic pain. Cochrane Database Syst Rev. 2005;(3):CD005452.-
9. American College of Obstetricians and Gynecologists. Chronic pelvic pain. ACOG Practice Bulletin No. 51. Obstet Gynecol. 2004;103:589-605.
10. Sinclair AM, Miller B, Lee LK. Chronic orchialgia: consider gabapentin or nortriptyline before considering surgery. Int J Urol. 2007;14:622-625.
11. Jarow JP, Budin RE, Dym M, et al. Quantitative pathologic changes in the human testis after vasectomy. N Engl J Med. 1985;313:1252-1256.
12. Choe J, Kirkemo A. Questionnaire-based outcomes study of nononcological post-vasectomy complications. J Urol. 1996;155:1284-1286.
13. McMahon A, Buckley J, Taylor A, et al. Chronic testicular pain following vasectomy. Br J Urol. 1992;69:188-191.
14. Ahmed I, Rasheed S, White C, et al. Th e incidence of post-vasectomy chronic testicular pain and the role of nerve stripping (denervation) of the spermatic cord in its management. Br J Urol. 1997;79:269-270.
15. Christiansen CG, Sandlow JI. Testicular pain following vasectomy: a review of postvasectomy pain syndrome. J Androl. 2003;24:293-298.
16. Shapiro EI, Silber SJ. Open-ended vasectomy, sperm granuloma, and postvasectomy orchialgia. Fertil Steril. 1979;32:546-550.
17. Taxy JB, Marshall FF, Erlickman RJ. Vasectomy: subclinical pathologic changes. Am J Surg Pathol. 1981;5:767-772.
18. Fuchs E. Cord block anesthesia for scrotal surgery. J Urol. 1982;128:718-719.
19. Issa M, Hsiao K, Bassel Y, et al. Spermatic cord anesthesia block for scrotal procedures in the outpatient clinic setting. J Urol. 2004;172:2358-2361.
20. Zorn B, Rauchenwald M, Steers WD. Periprostatic injection of local anesthesia for relief of chronic orchialgia. J Urol. 1994;151:411,A735.-
21. Levine LA, Matkov TG, Lubenow TR. Microsurgical denervation of the spermatic cord: a surgical alternative in the treatment of chronic orchialgia. J Urol. 1996;155:1005-1007.
22. Strom KH, Levine LA. Microsurgical denervation of the spermatic cord for chronic orchialgia: long-term results from a single center. J Urol. 2008;180:949-953.
23. Padmore DE, Norman RW, Millard OH. Analyses of indications for and outcomes of epdidymectomy. J Urol. 1996;156:95-96.
24. West AF, Leung HY, Powell PH. Epididymectomy is an effective treatment for scrotal pain after vasectomy. Br J Urol Int. 2000;85:1097-1099.
25. Siu W, Ohl DA, Schuster TG. Long-term follow-up after epididymectomy for chronic epidiymal pain. Urology. 2007;70:333-336.
26. Patel SR, Sigman M. Comparison of outcomes of vasovasostomy performed in the convoluted and straight vas deferens. J Urol. 2008;179:256-259.
27. Myers SA, Mershon CE, Fuchs EF. Vasectomy reversal for treatment of the post-vasectomy pain syndrome. J Urol. 1997;157:518-520.
28. Costabile RA, Hahn M, McLeod DG. Chronic orchialgia in the pain prone patient: the clinical perspective. J Urol. 1991;146:1571-1574.
Managing a Child With Heart Palpitations
When children or adolescents present complaining of heart palpitations, first determine just what they are describing. “Heart palpitation” is a very vague term and could mean anything: Patients might say their “heart is racing,” they feel like they “skipped a heartbeat,” or it could just be a greater awareness of their heart beating.
A comprehensive history is important in determining the possibility of a significant arrhythmia. Every child walking in with a history of palpitation does not have a cardiac arrhythmia.
Significant arrhythmias are far fewer in children than adults. More common in children are noncardiac issues, such as heart palpitations associated with anxiety and panic attacks or minor arrhythmias such as premature atrial and ventricular beats.
Normal heart rhythm is sinus rhythm, and sinus tachycardia is not a problem most of the time. Sinus tachycardia has many possible etiologies. It can occur when a child or adolescent exercises, feels anxious, or is running a fever. Fortunately, sinus tachycardia is not something we normally worry about. Children are easily excited and may experience their heart racing. They may report that they feel short of breath during an episode, which may point to anxiety. In some cases, excessive caffeine intake may be responsible for these symptoms.
Ask about onset and duration of symptoms during history taking. For example, patients who report a rapid heartbeat that lasts a half-hour or longer are more of a concern than those who report a few minutes or seconds of symptoms where it feels like “their heart is popping out of their chest.” Also inquire about dizziness, which could indicate their blood pressure falls during an episode.
Pediatricians who feel comfortable addressing cardiac issues should try to differentiate benign sinus tachycardia from the more concerning forms: supraventricular tachycardia and ventricular tachycardia.
If parents report the child's heart was racing and it was 120 beats per minute (bpm), you can reassure them. If the heart rate is 120, 130, 140, 150, or even 160, it is generally nothing to worry about—it is bound to be sinus tachycardia. Even very fast sinus tachycardia may be normal given the circumstances. For example, a 16-year-old athlete at the peak of physical activity might have a heart rate closer to 190 or 200 bpm. That is fast, but it is appropriate for the age and level of activity.
More worrisome is supraventricular tachycardia (SVT), which has an entirely different mechanism in terms of electrophysiology of the heart, reentry being responsible for most. The electrical conduction system of the heart may have a bypass tract or a dual atrioventricular node which allows for reentry to occur. In general, the heart rate of 200 bpm or higher is likely to be SVT and warrants referral to a cardiac specialist.
Patients with heart rates falling in the in-between range (170-200 bpm) may have sinus tachycardia or SVT and should also have a work-up. Attempting to document the rhythm during episodes becomes important in these children.
SVT can occur in children of any age, including newborns and infants. It usually starts abruptly and unpredictably—out of the blue, the heart starts racing. The child can be completely inactive at the time. For a minority, exercise may be the trigger. The rapid heartbeat may be short or last for many hours, and then the episode stops as suddenly as it started. It is not life threatening unless the child is in incessant SVT for hours.
A routine ECG can sometimes help pediatricians with their differential diagnosis. Markers, such as a bypass tract, can show up on the ECG tracing. If a pre-excitement pattern is seen, further work-up may be needed, even in asymptomatic children incidentally discovered on routine testing for other reasons. Unequivocal diagnosis of SVT requires ECG documentation of the abnormal rhythm during an episode.
Because episodes of SVT tend to be unpredictable, trying to record the rhythm during the episode is not an easy exercise. One solution is to ask parents to record the child's pulse rate or heart rate during an episode, while the patient is complaining of symptoms. Going to the nearest emergency department increases the possibility of ECG identification of the SVT.
A pediatrician can also prescribe 24-hour Holter monitoring to try to catch an episode. If a patient is referred to a pediatric cardiologist, the patient is often monitored for a longer period of time in an attempt to catch the arrhythmia, such as 48-72 hours. Another helpful device is a loop recorder, which is worn constantly; it records and erases the ECG data after each half-hour. Parents are instructed to save the data after the episode by pushing a button. The data can then be transmitted to the monitoring company via telephone. The signal is turned into an ECG rhythm strip and sent to the physician for further analysis and necessary action.
The most worrisome arrhythmia in children is ventricular tachycardia (VT). It is a life-threatening arrhythmia. Fortunately, it is the most uncommon. Nonetheless, people who treat children have to be aware of conditions that are a setup for this kind of arrhythmia.
Hypertrophic cardiomyopathy is one such condition. It is a genetic condition leading to abnormality of the ventricular muscle. The abnormality produces a structural change with thickening of the heart muscle in an asymmetric fashion. The thickness may cause obstruction of the left ventricular outflow tract. In addition, such an individual is prone to VT. The presenting symptom for such individuals tends to be loss of consciousness or a fainting episode with the arrhythmia, generally occurring in the setting of vigorous physical activity. It can be a silent condition, fainting or collapse being the first symptom for some patients. Diagnosis is made by a combination of tests, including echocardiogram and ECG. Genetic testing is now available for the condition but does not help with practical management issues.
If a child is reporting other problems, such as constant fatigue and/or symptoms with minor daily activities, it suggests the possibility of myocarditis, usually related to viral infections. The heart of a child with such an infection becomes dilated and does not contract well. These patients are prone to VT. While some children with myocarditis recover, others may suffer permanent damage. The heart may remain dilated and prone to VT.
Another rare condition to include in your differential diagnosis is prolonged QT interval syndrome. The condition is genetic in nature and tends to run in families. The mechanism involves gates or ion channels in cell membranes that normally regulate the influx of sodium, potassium, and calcium. In a child with the syndrome, the gates remain open too long, thereby allowing too much sodium or calcium to enter the cells, and VT can ensue. ECG tracings and a history of syncope can be diagnostic. Genetic testing is available and helpful.
Ventricular tachycardia is very worrisome because it can be fatal within minutes. Therefore, VT is an emergency in most instances, although there are some benign forms that are well tolerated. The distinction can sometimes require an electrophysiology study. The placement of automatic external defibrillators in public places has enhanced the chances of survival in patients who experience rapid VT.
When children or adolescents present complaining of heart palpitations, first determine just what they are describing. “Heart palpitation” is a very vague term and could mean anything: Patients might say their “heart is racing,” they feel like they “skipped a heartbeat,” or it could just be a greater awareness of their heart beating.
A comprehensive history is important in determining the possibility of a significant arrhythmia. Every child walking in with a history of palpitation does not have a cardiac arrhythmia.
Significant arrhythmias are far fewer in children than adults. More common in children are noncardiac issues, such as heart palpitations associated with anxiety and panic attacks or minor arrhythmias such as premature atrial and ventricular beats.
Normal heart rhythm is sinus rhythm, and sinus tachycardia is not a problem most of the time. Sinus tachycardia has many possible etiologies. It can occur when a child or adolescent exercises, feels anxious, or is running a fever. Fortunately, sinus tachycardia is not something we normally worry about. Children are easily excited and may experience their heart racing. They may report that they feel short of breath during an episode, which may point to anxiety. In some cases, excessive caffeine intake may be responsible for these symptoms.
Ask about onset and duration of symptoms during history taking. For example, patients who report a rapid heartbeat that lasts a half-hour or longer are more of a concern than those who report a few minutes or seconds of symptoms where it feels like “their heart is popping out of their chest.” Also inquire about dizziness, which could indicate their blood pressure falls during an episode.
Pediatricians who feel comfortable addressing cardiac issues should try to differentiate benign sinus tachycardia from the more concerning forms: supraventricular tachycardia and ventricular tachycardia.
If parents report the child's heart was racing and it was 120 beats per minute (bpm), you can reassure them. If the heart rate is 120, 130, 140, 150, or even 160, it is generally nothing to worry about—it is bound to be sinus tachycardia. Even very fast sinus tachycardia may be normal given the circumstances. For example, a 16-year-old athlete at the peak of physical activity might have a heart rate closer to 190 or 200 bpm. That is fast, but it is appropriate for the age and level of activity.
More worrisome is supraventricular tachycardia (SVT), which has an entirely different mechanism in terms of electrophysiology of the heart, reentry being responsible for most. The electrical conduction system of the heart may have a bypass tract or a dual atrioventricular node which allows for reentry to occur. In general, the heart rate of 200 bpm or higher is likely to be SVT and warrants referral to a cardiac specialist.
Patients with heart rates falling in the in-between range (170-200 bpm) may have sinus tachycardia or SVT and should also have a work-up. Attempting to document the rhythm during episodes becomes important in these children.
SVT can occur in children of any age, including newborns and infants. It usually starts abruptly and unpredictably—out of the blue, the heart starts racing. The child can be completely inactive at the time. For a minority, exercise may be the trigger. The rapid heartbeat may be short or last for many hours, and then the episode stops as suddenly as it started. It is not life threatening unless the child is in incessant SVT for hours.
A routine ECG can sometimes help pediatricians with their differential diagnosis. Markers, such as a bypass tract, can show up on the ECG tracing. If a pre-excitement pattern is seen, further work-up may be needed, even in asymptomatic children incidentally discovered on routine testing for other reasons. Unequivocal diagnosis of SVT requires ECG documentation of the abnormal rhythm during an episode.
Because episodes of SVT tend to be unpredictable, trying to record the rhythm during the episode is not an easy exercise. One solution is to ask parents to record the child's pulse rate or heart rate during an episode, while the patient is complaining of symptoms. Going to the nearest emergency department increases the possibility of ECG identification of the SVT.
A pediatrician can also prescribe 24-hour Holter monitoring to try to catch an episode. If a patient is referred to a pediatric cardiologist, the patient is often monitored for a longer period of time in an attempt to catch the arrhythmia, such as 48-72 hours. Another helpful device is a loop recorder, which is worn constantly; it records and erases the ECG data after each half-hour. Parents are instructed to save the data after the episode by pushing a button. The data can then be transmitted to the monitoring company via telephone. The signal is turned into an ECG rhythm strip and sent to the physician for further analysis and necessary action.
The most worrisome arrhythmia in children is ventricular tachycardia (VT). It is a life-threatening arrhythmia. Fortunately, it is the most uncommon. Nonetheless, people who treat children have to be aware of conditions that are a setup for this kind of arrhythmia.
Hypertrophic cardiomyopathy is one such condition. It is a genetic condition leading to abnormality of the ventricular muscle. The abnormality produces a structural change with thickening of the heart muscle in an asymmetric fashion. The thickness may cause obstruction of the left ventricular outflow tract. In addition, such an individual is prone to VT. The presenting symptom for such individuals tends to be loss of consciousness or a fainting episode with the arrhythmia, generally occurring in the setting of vigorous physical activity. It can be a silent condition, fainting or collapse being the first symptom for some patients. Diagnosis is made by a combination of tests, including echocardiogram and ECG. Genetic testing is now available for the condition but does not help with practical management issues.
If a child is reporting other problems, such as constant fatigue and/or symptoms with minor daily activities, it suggests the possibility of myocarditis, usually related to viral infections. The heart of a child with such an infection becomes dilated and does not contract well. These patients are prone to VT. While some children with myocarditis recover, others may suffer permanent damage. The heart may remain dilated and prone to VT.
Another rare condition to include in your differential diagnosis is prolonged QT interval syndrome. The condition is genetic in nature and tends to run in families. The mechanism involves gates or ion channels in cell membranes that normally regulate the influx of sodium, potassium, and calcium. In a child with the syndrome, the gates remain open too long, thereby allowing too much sodium or calcium to enter the cells, and VT can ensue. ECG tracings and a history of syncope can be diagnostic. Genetic testing is available and helpful.
Ventricular tachycardia is very worrisome because it can be fatal within minutes. Therefore, VT is an emergency in most instances, although there are some benign forms that are well tolerated. The distinction can sometimes require an electrophysiology study. The placement of automatic external defibrillators in public places has enhanced the chances of survival in patients who experience rapid VT.
When children or adolescents present complaining of heart palpitations, first determine just what they are describing. “Heart palpitation” is a very vague term and could mean anything: Patients might say their “heart is racing,” they feel like they “skipped a heartbeat,” or it could just be a greater awareness of their heart beating.
A comprehensive history is important in determining the possibility of a significant arrhythmia. Every child walking in with a history of palpitation does not have a cardiac arrhythmia.
Significant arrhythmias are far fewer in children than adults. More common in children are noncardiac issues, such as heart palpitations associated with anxiety and panic attacks or minor arrhythmias such as premature atrial and ventricular beats.
Normal heart rhythm is sinus rhythm, and sinus tachycardia is not a problem most of the time. Sinus tachycardia has many possible etiologies. It can occur when a child or adolescent exercises, feels anxious, or is running a fever. Fortunately, sinus tachycardia is not something we normally worry about. Children are easily excited and may experience their heart racing. They may report that they feel short of breath during an episode, which may point to anxiety. In some cases, excessive caffeine intake may be responsible for these symptoms.
Ask about onset and duration of symptoms during history taking. For example, patients who report a rapid heartbeat that lasts a half-hour or longer are more of a concern than those who report a few minutes or seconds of symptoms where it feels like “their heart is popping out of their chest.” Also inquire about dizziness, which could indicate their blood pressure falls during an episode.
Pediatricians who feel comfortable addressing cardiac issues should try to differentiate benign sinus tachycardia from the more concerning forms: supraventricular tachycardia and ventricular tachycardia.
If parents report the child's heart was racing and it was 120 beats per minute (bpm), you can reassure them. If the heart rate is 120, 130, 140, 150, or even 160, it is generally nothing to worry about—it is bound to be sinus tachycardia. Even very fast sinus tachycardia may be normal given the circumstances. For example, a 16-year-old athlete at the peak of physical activity might have a heart rate closer to 190 or 200 bpm. That is fast, but it is appropriate for the age and level of activity.
More worrisome is supraventricular tachycardia (SVT), which has an entirely different mechanism in terms of electrophysiology of the heart, reentry being responsible for most. The electrical conduction system of the heart may have a bypass tract or a dual atrioventricular node which allows for reentry to occur. In general, the heart rate of 200 bpm or higher is likely to be SVT and warrants referral to a cardiac specialist.
Patients with heart rates falling in the in-between range (170-200 bpm) may have sinus tachycardia or SVT and should also have a work-up. Attempting to document the rhythm during episodes becomes important in these children.
SVT can occur in children of any age, including newborns and infants. It usually starts abruptly and unpredictably—out of the blue, the heart starts racing. The child can be completely inactive at the time. For a minority, exercise may be the trigger. The rapid heartbeat may be short or last for many hours, and then the episode stops as suddenly as it started. It is not life threatening unless the child is in incessant SVT for hours.
A routine ECG can sometimes help pediatricians with their differential diagnosis. Markers, such as a bypass tract, can show up on the ECG tracing. If a pre-excitement pattern is seen, further work-up may be needed, even in asymptomatic children incidentally discovered on routine testing for other reasons. Unequivocal diagnosis of SVT requires ECG documentation of the abnormal rhythm during an episode.
Because episodes of SVT tend to be unpredictable, trying to record the rhythm during the episode is not an easy exercise. One solution is to ask parents to record the child's pulse rate or heart rate during an episode, while the patient is complaining of symptoms. Going to the nearest emergency department increases the possibility of ECG identification of the SVT.
A pediatrician can also prescribe 24-hour Holter monitoring to try to catch an episode. If a patient is referred to a pediatric cardiologist, the patient is often monitored for a longer period of time in an attempt to catch the arrhythmia, such as 48-72 hours. Another helpful device is a loop recorder, which is worn constantly; it records and erases the ECG data after each half-hour. Parents are instructed to save the data after the episode by pushing a button. The data can then be transmitted to the monitoring company via telephone. The signal is turned into an ECG rhythm strip and sent to the physician for further analysis and necessary action.
The most worrisome arrhythmia in children is ventricular tachycardia (VT). It is a life-threatening arrhythmia. Fortunately, it is the most uncommon. Nonetheless, people who treat children have to be aware of conditions that are a setup for this kind of arrhythmia.
Hypertrophic cardiomyopathy is one such condition. It is a genetic condition leading to abnormality of the ventricular muscle. The abnormality produces a structural change with thickening of the heart muscle in an asymmetric fashion. The thickness may cause obstruction of the left ventricular outflow tract. In addition, such an individual is prone to VT. The presenting symptom for such individuals tends to be loss of consciousness or a fainting episode with the arrhythmia, generally occurring in the setting of vigorous physical activity. It can be a silent condition, fainting or collapse being the first symptom for some patients. Diagnosis is made by a combination of tests, including echocardiogram and ECG. Genetic testing is now available for the condition but does not help with practical management issues.
If a child is reporting other problems, such as constant fatigue and/or symptoms with minor daily activities, it suggests the possibility of myocarditis, usually related to viral infections. The heart of a child with such an infection becomes dilated and does not contract well. These patients are prone to VT. While some children with myocarditis recover, others may suffer permanent damage. The heart may remain dilated and prone to VT.
Another rare condition to include in your differential diagnosis is prolonged QT interval syndrome. The condition is genetic in nature and tends to run in families. The mechanism involves gates or ion channels in cell membranes that normally regulate the influx of sodium, potassium, and calcium. In a child with the syndrome, the gates remain open too long, thereby allowing too much sodium or calcium to enter the cells, and VT can ensue. ECG tracings and a history of syncope can be diagnostic. Genetic testing is available and helpful.
Ventricular tachycardia is very worrisome because it can be fatal within minutes. Therefore, VT is an emergency in most instances, although there are some benign forms that are well tolerated. The distinction can sometimes require an electrophysiology study. The placement of automatic external defibrillators in public places has enhanced the chances of survival in patients who experience rapid VT.
Checklists Not Enough, Checklist Doctor Says
Peter Pronovost, MD, PhD, an intensivist and patient-safety researcher at Johns Hopkins University School of Medicine in Baltimore, has been widely profiled as the “checklist doctor” for his celebrated five-step checklist to reduce the incidence of central-line infections. But he objects to the label.
Just handing doctors and nurses a piece of paper is not likely to improve patient safety without two other essential steps: “We must also measure the results and give clinicians feedback, and we must change the culture so that they work collaboratively together,” he explains.
Dr. Pronovost shares his personal story as a safety expert who borrowed from aviation in developing his first checklist in 2001 in a Johns Hopkins surgical ICU in his new book, Safe Patients, Smart Hospitals, co-authored with Eric Vohr (New York: Hudson Street Press, 2010).
Dr. Pronovost says he still encounters resistance to the checklist in many U.S. hospitals. “Nobody debates that we should be doing the things on the checklist,” he says. “The evidence is strong. The barrier is culture or medical hierarchy. In what other industry would there be an accepted standard that failure to comply with it kills, in this case, 30,000 people per year, and yet we’re not comfortable having one worker question another about compliance with it?”
Hospitalists have a huge role in hospital quality and safety, he adds.
“I envision that they could take almost any practice guideline that’s out there and convert it into a checklist,” he says, emphasizing that hospitalists should appoint an interdisciplinary team to work on the project and make the checklists specific to one time and place. It also is important for hospitals to support hospitalists with dedicated time to work on such projects. “But in return, the hospitalists have to commit measuring safety performance and producing positive results,” he says.
Peter Pronovost, MD, PhD, an intensivist and patient-safety researcher at Johns Hopkins University School of Medicine in Baltimore, has been widely profiled as the “checklist doctor” for his celebrated five-step checklist to reduce the incidence of central-line infections. But he objects to the label.
Just handing doctors and nurses a piece of paper is not likely to improve patient safety without two other essential steps: “We must also measure the results and give clinicians feedback, and we must change the culture so that they work collaboratively together,” he explains.
Dr. Pronovost shares his personal story as a safety expert who borrowed from aviation in developing his first checklist in 2001 in a Johns Hopkins surgical ICU in his new book, Safe Patients, Smart Hospitals, co-authored with Eric Vohr (New York: Hudson Street Press, 2010).
Dr. Pronovost says he still encounters resistance to the checklist in many U.S. hospitals. “Nobody debates that we should be doing the things on the checklist,” he says. “The evidence is strong. The barrier is culture or medical hierarchy. In what other industry would there be an accepted standard that failure to comply with it kills, in this case, 30,000 people per year, and yet we’re not comfortable having one worker question another about compliance with it?”
Hospitalists have a huge role in hospital quality and safety, he adds.
“I envision that they could take almost any practice guideline that’s out there and convert it into a checklist,” he says, emphasizing that hospitalists should appoint an interdisciplinary team to work on the project and make the checklists specific to one time and place. It also is important for hospitals to support hospitalists with dedicated time to work on such projects. “But in return, the hospitalists have to commit measuring safety performance and producing positive results,” he says.
Peter Pronovost, MD, PhD, an intensivist and patient-safety researcher at Johns Hopkins University School of Medicine in Baltimore, has been widely profiled as the “checklist doctor” for his celebrated five-step checklist to reduce the incidence of central-line infections. But he objects to the label.
Just handing doctors and nurses a piece of paper is not likely to improve patient safety without two other essential steps: “We must also measure the results and give clinicians feedback, and we must change the culture so that they work collaboratively together,” he explains.
Dr. Pronovost shares his personal story as a safety expert who borrowed from aviation in developing his first checklist in 2001 in a Johns Hopkins surgical ICU in his new book, Safe Patients, Smart Hospitals, co-authored with Eric Vohr (New York: Hudson Street Press, 2010).
Dr. Pronovost says he still encounters resistance to the checklist in many U.S. hospitals. “Nobody debates that we should be doing the things on the checklist,” he says. “The evidence is strong. The barrier is culture or medical hierarchy. In what other industry would there be an accepted standard that failure to comply with it kills, in this case, 30,000 people per year, and yet we’re not comfortable having one worker question another about compliance with it?”
Hospitalists have a huge role in hospital quality and safety, he adds.
“I envision that they could take almost any practice guideline that’s out there and convert it into a checklist,” he says, emphasizing that hospitalists should appoint an interdisciplinary team to work on the project and make the checklists specific to one time and place. It also is important for hospitals to support hospitalists with dedicated time to work on such projects. “But in return, the hospitalists have to commit measuring safety performance and producing positive results,” he says.
Two-Pronged Approach
An author of a recent study that found improved results for depressed alcohol-dependent patients when they were treated for both diagnoses says hospitalists are positioned to help push increased usage of the dual-medication approach.
“This could change the way people treat these patients,” says first author Helen Pettinati, PhD, professor of psychiatry and director of the Addiction Treatment & Medication Development Division at the University of Pennsylvania School of Medicine’s Treatment Research Center in Philadelphia.
The study in the American Journal of Psychiatry tracked 170 patients, with some treated with 14 weeks of sertraline (200 mg/day, n=40), naltrexone (100 mg/day, n=49), both drugs (n=42), or double placebo (n=39). All groups received weekly counseling as well. Patients treated with both medications produced a higher alcohol abstinence rate (53.7%) and demonstrated a longer delay before relapse to heavy drinking (median delay=98 days) than the naltrexone (abstinence rate: 21.3%; delay=29 days), sertraline (abstinence rate: 27.5%; delay=23 days), and placebo (abstinence rate: 23.1%; delay=26 days) groups. In addition, patients treated with both medications “reported fewer serious adverse events, and tended to not be depressed by the end of treatment,” the authors wrote.
Pettinati says it is uncommon for hospitalists and other physicians to consider treating hospitalized patients with medication for alcohol dependence. She hopes hospitalists see this study as a spotlight on that approach. In past years, Pettinati suggests, medication for alcohol dependence might have been viewed as unnecessary because patient lengths-of-stay (LOS) were longer. Today, HM leaders are pushing for ever-shorter LOS.
“Now when a person comes in,” Pettinati says, “you have to make an immediate decision what kind of medication you want to treat this person with.”
An author of a recent study that found improved results for depressed alcohol-dependent patients when they were treated for both diagnoses says hospitalists are positioned to help push increased usage of the dual-medication approach.
“This could change the way people treat these patients,” says first author Helen Pettinati, PhD, professor of psychiatry and director of the Addiction Treatment & Medication Development Division at the University of Pennsylvania School of Medicine’s Treatment Research Center in Philadelphia.
The study in the American Journal of Psychiatry tracked 170 patients, with some treated with 14 weeks of sertraline (200 mg/day, n=40), naltrexone (100 mg/day, n=49), both drugs (n=42), or double placebo (n=39). All groups received weekly counseling as well. Patients treated with both medications produced a higher alcohol abstinence rate (53.7%) and demonstrated a longer delay before relapse to heavy drinking (median delay=98 days) than the naltrexone (abstinence rate: 21.3%; delay=29 days), sertraline (abstinence rate: 27.5%; delay=23 days), and placebo (abstinence rate: 23.1%; delay=26 days) groups. In addition, patients treated with both medications “reported fewer serious adverse events, and tended to not be depressed by the end of treatment,” the authors wrote.
Pettinati says it is uncommon for hospitalists and other physicians to consider treating hospitalized patients with medication for alcohol dependence. She hopes hospitalists see this study as a spotlight on that approach. In past years, Pettinati suggests, medication for alcohol dependence might have been viewed as unnecessary because patient lengths-of-stay (LOS) were longer. Today, HM leaders are pushing for ever-shorter LOS.
“Now when a person comes in,” Pettinati says, “you have to make an immediate decision what kind of medication you want to treat this person with.”
An author of a recent study that found improved results for depressed alcohol-dependent patients when they were treated for both diagnoses says hospitalists are positioned to help push increased usage of the dual-medication approach.
“This could change the way people treat these patients,” says first author Helen Pettinati, PhD, professor of psychiatry and director of the Addiction Treatment & Medication Development Division at the University of Pennsylvania School of Medicine’s Treatment Research Center in Philadelphia.
The study in the American Journal of Psychiatry tracked 170 patients, with some treated with 14 weeks of sertraline (200 mg/day, n=40), naltrexone (100 mg/day, n=49), both drugs (n=42), or double placebo (n=39). All groups received weekly counseling as well. Patients treated with both medications produced a higher alcohol abstinence rate (53.7%) and demonstrated a longer delay before relapse to heavy drinking (median delay=98 days) than the naltrexone (abstinence rate: 21.3%; delay=29 days), sertraline (abstinence rate: 27.5%; delay=23 days), and placebo (abstinence rate: 23.1%; delay=26 days) groups. In addition, patients treated with both medications “reported fewer serious adverse events, and tended to not be depressed by the end of treatment,” the authors wrote.
Pettinati says it is uncommon for hospitalists and other physicians to consider treating hospitalized patients with medication for alcohol dependence. She hopes hospitalists see this study as a spotlight on that approach. In past years, Pettinati suggests, medication for alcohol dependence might have been viewed as unnecessary because patient lengths-of-stay (LOS) were longer. Today, HM leaders are pushing for ever-shorter LOS.
“Now when a person comes in,” Pettinati says, “you have to make an immediate decision what kind of medication you want to treat this person with.”
New Cost-Control Strategy
A recent study that associates lower 30-day readmission rates for heart failure patients who receive followups within one week might be a jumpstart to new incentives for fewer readmissions, says the report’s author.
“In a way, [hospitalists] are central to all this,” says Adrian F. Hernandez, MD, MHS, an assistant professor at Duke University School of Medicine and a cardiologist at Duke University Medical Center in Durham, N.C. “During the hospital stay, they are quarterbacking that patient’s care. They have a central responsibility to make sure that patient discharge is seamless.”
The study tracked 30,136 patients who were seen at 225 hospitals from January 2003 to December 2006. It reported that in the first 30 days after discharge, 6,428 patients (21.3 percent) were readmitted (JAMA. 2010;303(17):1716-1722).
At the hospital level, the median rate of early followup was 38.3 percent. According to the study, patients whose index admission was in a hospital in the lowest quartile of early followup had a 23.3% 30-day readmission rate. The rates of 30-day readmission were 20.5% among patients in the second quartile, 20.5% among patients in the third quartile, and 20.9% among patients in the fourth quartile.
Dr. Hernandez says the next step is for hospitals and their staffs to commit to more streamlined transitional-care techniques that include immediate followup with patients, be it via teleconferencing with doctors or phone calls with nonphysician providers (NPPs) or clinical pharmacists. He adds that incentivizing doctors to reduce readmissions is a logical next step to improving the discharge process.
“Now that 30-day readmissions are publicly reported and hospitals are being held accountable for that, they need to invest in systems that will enhance that transitional period,” Dr. Hernandez says.
A recent study that associates lower 30-day readmission rates for heart failure patients who receive followups within one week might be a jumpstart to new incentives for fewer readmissions, says the report’s author.
“In a way, [hospitalists] are central to all this,” says Adrian F. Hernandez, MD, MHS, an assistant professor at Duke University School of Medicine and a cardiologist at Duke University Medical Center in Durham, N.C. “During the hospital stay, they are quarterbacking that patient’s care. They have a central responsibility to make sure that patient discharge is seamless.”
The study tracked 30,136 patients who were seen at 225 hospitals from January 2003 to December 2006. It reported that in the first 30 days after discharge, 6,428 patients (21.3 percent) were readmitted (JAMA. 2010;303(17):1716-1722).
At the hospital level, the median rate of early followup was 38.3 percent. According to the study, patients whose index admission was in a hospital in the lowest quartile of early followup had a 23.3% 30-day readmission rate. The rates of 30-day readmission were 20.5% among patients in the second quartile, 20.5% among patients in the third quartile, and 20.9% among patients in the fourth quartile.
Dr. Hernandez says the next step is for hospitals and their staffs to commit to more streamlined transitional-care techniques that include immediate followup with patients, be it via teleconferencing with doctors or phone calls with nonphysician providers (NPPs) or clinical pharmacists. He adds that incentivizing doctors to reduce readmissions is a logical next step to improving the discharge process.
“Now that 30-day readmissions are publicly reported and hospitals are being held accountable for that, they need to invest in systems that will enhance that transitional period,” Dr. Hernandez says.
A recent study that associates lower 30-day readmission rates for heart failure patients who receive followups within one week might be a jumpstart to new incentives for fewer readmissions, says the report’s author.
“In a way, [hospitalists] are central to all this,” says Adrian F. Hernandez, MD, MHS, an assistant professor at Duke University School of Medicine and a cardiologist at Duke University Medical Center in Durham, N.C. “During the hospital stay, they are quarterbacking that patient’s care. They have a central responsibility to make sure that patient discharge is seamless.”
The study tracked 30,136 patients who were seen at 225 hospitals from January 2003 to December 2006. It reported that in the first 30 days after discharge, 6,428 patients (21.3 percent) were readmitted (JAMA. 2010;303(17):1716-1722).
At the hospital level, the median rate of early followup was 38.3 percent. According to the study, patients whose index admission was in a hospital in the lowest quartile of early followup had a 23.3% 30-day readmission rate. The rates of 30-day readmission were 20.5% among patients in the second quartile, 20.5% among patients in the third quartile, and 20.9% among patients in the fourth quartile.
Dr. Hernandez says the next step is for hospitals and their staffs to commit to more streamlined transitional-care techniques that include immediate followup with patients, be it via teleconferencing with doctors or phone calls with nonphysician providers (NPPs) or clinical pharmacists. He adds that incentivizing doctors to reduce readmissions is a logical next step to improving the discharge process.
“Now that 30-day readmissions are publicly reported and hospitals are being held accountable for that, they need to invest in systems that will enhance that transitional period,” Dr. Hernandez says.
In the Literature: Research You Need to Know
Clinical question: Do rapid-response teams (RRTs) reduce the rates of cardiopulmonary arrest and hospital mortality?
Background: RRTs are charged with prompt evaluation and treatment of inpatients with clinical deterioration to prevent cardiopulmonary arrest and its attendant mortality. Though hundreds of hospitals have implemented these teams as part of their quality-improvement (QI) initiatives, previous studies on RRTs have reported mixed results on the clinically meaningful outcome of hospital mortality.
Study design: Meta-analysis and systematic review.
Setting: Randomized, controlled trials and prospective studies on RRTs from multiple databases, including PubMed, EMBASE, and CINAHL.
Synopsis: Eighteen studies (13 adult and five pediatric) with a total sample size of nearly 1.3 million admissions were analyzed. Of these, 15 reported on the primary outcome of in-hospital mortality; 16 reported on the secondary outcome of cardiopulmonary arrest. All of the studies exhibited extensive heterogeneity of outcomes, but six studies were deemed to be of high quality.
In adults, RRT implementation reduced non-ICU cardiopulmonary arrest by 33.8%, without an effect on hospital mortality (pooled RR, 0.96; 95% CI, 0.84-1.09). Interestingly, the inclusion of recent evidence neutralized positive results from initial studies in the overall pooled analysis.
In children, apart from reduction in arrest (37.7%), a weak association with lower mortality rates (pooled RR, 0.79; 95% CI, 0.63-0.98) was noticed. This did not hold on sensitivity analysis, but that could be explained by their higher likelihood to survive cardiac arrest than adults.
The discordance between the primary and secondary outcomes could be due to pre-arrest transfer to ICU or establishment of DNR status by RRT, hence excluding them from mortality analysis.
Bottom line: Although RRTs reduce rates of cardiopulmonary arrest outside the ICU, no consistent evidence shows RRTs improve survival to discharge.
Citation: Chan PS, Jain R, Nallmothu BK, Berg RA, Sasson C. Rapid response teams: a systematic review and meta-analysis. Arch Intern Med. 2010;170(1):18-26.
Reviewed for TH eWire by Rubin Bahuva, MD, Chadi Alraies, MD, Anuradha Ramaswamy, MD, Sudhir Manda, MD, Maria Giselle Velez, MD, and Mital Patel, MD, Department of Hospital Medicine, Cleveland Clinic
For more physician reviews of HM-related research, visit our website.
Clinical question: Do rapid-response teams (RRTs) reduce the rates of cardiopulmonary arrest and hospital mortality?
Background: RRTs are charged with prompt evaluation and treatment of inpatients with clinical deterioration to prevent cardiopulmonary arrest and its attendant mortality. Though hundreds of hospitals have implemented these teams as part of their quality-improvement (QI) initiatives, previous studies on RRTs have reported mixed results on the clinically meaningful outcome of hospital mortality.
Study design: Meta-analysis and systematic review.
Setting: Randomized, controlled trials and prospective studies on RRTs from multiple databases, including PubMed, EMBASE, and CINAHL.
Synopsis: Eighteen studies (13 adult and five pediatric) with a total sample size of nearly 1.3 million admissions were analyzed. Of these, 15 reported on the primary outcome of in-hospital mortality; 16 reported on the secondary outcome of cardiopulmonary arrest. All of the studies exhibited extensive heterogeneity of outcomes, but six studies were deemed to be of high quality.
In adults, RRT implementation reduced non-ICU cardiopulmonary arrest by 33.8%, without an effect on hospital mortality (pooled RR, 0.96; 95% CI, 0.84-1.09). Interestingly, the inclusion of recent evidence neutralized positive results from initial studies in the overall pooled analysis.
In children, apart from reduction in arrest (37.7%), a weak association with lower mortality rates (pooled RR, 0.79; 95% CI, 0.63-0.98) was noticed. This did not hold on sensitivity analysis, but that could be explained by their higher likelihood to survive cardiac arrest than adults.
The discordance between the primary and secondary outcomes could be due to pre-arrest transfer to ICU or establishment of DNR status by RRT, hence excluding them from mortality analysis.
Bottom line: Although RRTs reduce rates of cardiopulmonary arrest outside the ICU, no consistent evidence shows RRTs improve survival to discharge.
Citation: Chan PS, Jain R, Nallmothu BK, Berg RA, Sasson C. Rapid response teams: a systematic review and meta-analysis. Arch Intern Med. 2010;170(1):18-26.
Reviewed for TH eWire by Rubin Bahuva, MD, Chadi Alraies, MD, Anuradha Ramaswamy, MD, Sudhir Manda, MD, Maria Giselle Velez, MD, and Mital Patel, MD, Department of Hospital Medicine, Cleveland Clinic
For more physician reviews of HM-related research, visit our website.
Clinical question: Do rapid-response teams (RRTs) reduce the rates of cardiopulmonary arrest and hospital mortality?
Background: RRTs are charged with prompt evaluation and treatment of inpatients with clinical deterioration to prevent cardiopulmonary arrest and its attendant mortality. Though hundreds of hospitals have implemented these teams as part of their quality-improvement (QI) initiatives, previous studies on RRTs have reported mixed results on the clinically meaningful outcome of hospital mortality.
Study design: Meta-analysis and systematic review.
Setting: Randomized, controlled trials and prospective studies on RRTs from multiple databases, including PubMed, EMBASE, and CINAHL.
Synopsis: Eighteen studies (13 adult and five pediatric) with a total sample size of nearly 1.3 million admissions were analyzed. Of these, 15 reported on the primary outcome of in-hospital mortality; 16 reported on the secondary outcome of cardiopulmonary arrest. All of the studies exhibited extensive heterogeneity of outcomes, but six studies were deemed to be of high quality.
In adults, RRT implementation reduced non-ICU cardiopulmonary arrest by 33.8%, without an effect on hospital mortality (pooled RR, 0.96; 95% CI, 0.84-1.09). Interestingly, the inclusion of recent evidence neutralized positive results from initial studies in the overall pooled analysis.
In children, apart from reduction in arrest (37.7%), a weak association with lower mortality rates (pooled RR, 0.79; 95% CI, 0.63-0.98) was noticed. This did not hold on sensitivity analysis, but that could be explained by their higher likelihood to survive cardiac arrest than adults.
The discordance between the primary and secondary outcomes could be due to pre-arrest transfer to ICU or establishment of DNR status by RRT, hence excluding them from mortality analysis.
Bottom line: Although RRTs reduce rates of cardiopulmonary arrest outside the ICU, no consistent evidence shows RRTs improve survival to discharge.
Citation: Chan PS, Jain R, Nallmothu BK, Berg RA, Sasson C. Rapid response teams: a systematic review and meta-analysis. Arch Intern Med. 2010;170(1):18-26.
Reviewed for TH eWire by Rubin Bahuva, MD, Chadi Alraies, MD, Anuradha Ramaswamy, MD, Sudhir Manda, MD, Maria Giselle Velez, MD, and Mital Patel, MD, Department of Hospital Medicine, Cleveland Clinic
For more physician reviews of HM-related research, visit our website.
Resident Refunds
The IRS has sided with medical residents and their employers who for years have argued that they should have always been eligible for the “student exemption”—but don’t count on any money just yet.
By mid-June, the IRS expects to have contacted hospitals, universities, and individual residents who filed Social Security and Medicare payroll tax refund claims as of April 1, 2005. The date is significant because it is when the IRS ruled that employees who work 40 hours or more at a school, college, or university are eligible for student exemptions.
The IRS’ administrative decision in early March affects taxes paid before 2005.
The IRS has only taken the first step and says instructions on how to further process claims will be forthcoming. For now, the federal agency says, “employers and individuals with pending claims do not need to take any action at this time.”
Still, Joseph Ming-Wah Li, MD, SFHM, assistant professor of medicine at Harvard Medical School and director of the hospital medicine division at Beth Israel Deaconess Medical Center in Boston, urges hospitalists to pay attention to the refund; over three years of residency, it could amount to several thousand dollars per physician.
“The government ruling recently was that residents should be treated more like students instead of employees,” says Dr. Li, SHM's president-elect.
Hospitals, medical schools, and residents themselves have been filing so-called “FICA refund claims” since the 1990s. A series of legal challenges led to opposing interpretations of tax codes, leading the IRS to suspend all claims until a ruling was made.
And while pending claims will now be processed, it is too late for new claims to be filed. However, residents who did not file individual claims in what the IRS calls a “timely fashion” should check with their residency institution to determine if a claim was filed on their behalf.
Read "Dr. Hospitalist's" take on this topic in this month's issue of The Hospitalist.
For more details, visit the IRS website.
The IRS has sided with medical residents and their employers who for years have argued that they should have always been eligible for the “student exemption”—but don’t count on any money just yet.
By mid-June, the IRS expects to have contacted hospitals, universities, and individual residents who filed Social Security and Medicare payroll tax refund claims as of April 1, 2005. The date is significant because it is when the IRS ruled that employees who work 40 hours or more at a school, college, or university are eligible for student exemptions.
The IRS’ administrative decision in early March affects taxes paid before 2005.
The IRS has only taken the first step and says instructions on how to further process claims will be forthcoming. For now, the federal agency says, “employers and individuals with pending claims do not need to take any action at this time.”
Still, Joseph Ming-Wah Li, MD, SFHM, assistant professor of medicine at Harvard Medical School and director of the hospital medicine division at Beth Israel Deaconess Medical Center in Boston, urges hospitalists to pay attention to the refund; over three years of residency, it could amount to several thousand dollars per physician.
“The government ruling recently was that residents should be treated more like students instead of employees,” says Dr. Li, SHM's president-elect.
Hospitals, medical schools, and residents themselves have been filing so-called “FICA refund claims” since the 1990s. A series of legal challenges led to opposing interpretations of tax codes, leading the IRS to suspend all claims until a ruling was made.
And while pending claims will now be processed, it is too late for new claims to be filed. However, residents who did not file individual claims in what the IRS calls a “timely fashion” should check with their residency institution to determine if a claim was filed on their behalf.
Read "Dr. Hospitalist's" take on this topic in this month's issue of The Hospitalist.
For more details, visit the IRS website.
The IRS has sided with medical residents and their employers who for years have argued that they should have always been eligible for the “student exemption”—but don’t count on any money just yet.
By mid-June, the IRS expects to have contacted hospitals, universities, and individual residents who filed Social Security and Medicare payroll tax refund claims as of April 1, 2005. The date is significant because it is when the IRS ruled that employees who work 40 hours or more at a school, college, or university are eligible for student exemptions.
The IRS’ administrative decision in early March affects taxes paid before 2005.
The IRS has only taken the first step and says instructions on how to further process claims will be forthcoming. For now, the federal agency says, “employers and individuals with pending claims do not need to take any action at this time.”
Still, Joseph Ming-Wah Li, MD, SFHM, assistant professor of medicine at Harvard Medical School and director of the hospital medicine division at Beth Israel Deaconess Medical Center in Boston, urges hospitalists to pay attention to the refund; over three years of residency, it could amount to several thousand dollars per physician.
“The government ruling recently was that residents should be treated more like students instead of employees,” says Dr. Li, SHM's president-elect.
Hospitals, medical schools, and residents themselves have been filing so-called “FICA refund claims” since the 1990s. A series of legal challenges led to opposing interpretations of tax codes, leading the IRS to suspend all claims until a ruling was made.
And while pending claims will now be processed, it is too late for new claims to be filed. However, residents who did not file individual claims in what the IRS calls a “timely fashion” should check with their residency institution to determine if a claim was filed on their behalf.
Read "Dr. Hospitalist's" take on this topic in this month's issue of The Hospitalist.
For more details, visit the IRS website.
In the Literature: Research You Need to Know
Clinical question: Does remote ICU monitoring improve mortality and length of stay?
Background: A shortage of intensivists has led to increased use of remote ICU monitoring or telemedicine technology to allow intensivists to remotely and simultaneously care for patients in multiple ICUs. Data evaluating this practice have been limited.
Study design: Pre- and postintervention observational study.
Setting: Open and closed medical-surgical ICUs in community, urban, and tertiary-care teaching U.S. hospitals.
Synopsis: This observational study in six ICUs aimed to assess the association of a telemedicine intervention with clinical outcomes. The intervention consisted of a remote office with real-time audiovisual monitoring, vital signs, early warning signals, and other electronic data. Comparing preintervention (n=2,034) and postintervention (n=2,108) groups, there were no differences in mortality, LOS, or complications.
Overall, the general limitation of the study was that integration of the tele-ICU and actual ICUs was limited. Physicians for nearly two-thirds of the patients chose “minimal delegation” to the tele-ICU physician. Tele-ICU involvement was particularly limited in “closed” units, which were already staffed by on-site intensivists. Furthermore, despite access to various real-time data, critical elements of the record such as physician order entry and progress notes were not shared in real time; notes, for example, required daily faxing.
While it is unfortunate that the study could not evaluate the full potential of the adjunctive tele-ICU, it illustrates the real-world obstacles of integrating such technology into clinical practice. In future studies, a standardized telemedicine approach might facilitate evaluation efforts.
Bottom line: While this study demonstrated no benefit of telemedicine, study limitations preclude conclusions. Further studies are needed.
Citation: Thomas EJ, Lucke JF, Wueste L, Weavind L, Patel B. Association of telemedicine for remote monitoring of intensive care patients with mortality, complications, and length of stay. JAMA. 2009;302(24):2671-2678.
Dr. Kim is a hospitalist at Brigham and Women's Hospital in Boston, and an instructor at Harvard Medical School.
For more reviews of HM-related research, visit our website.
Clinical question: Does remote ICU monitoring improve mortality and length of stay?
Background: A shortage of intensivists has led to increased use of remote ICU monitoring or telemedicine technology to allow intensivists to remotely and simultaneously care for patients in multiple ICUs. Data evaluating this practice have been limited.
Study design: Pre- and postintervention observational study.
Setting: Open and closed medical-surgical ICUs in community, urban, and tertiary-care teaching U.S. hospitals.
Synopsis: This observational study in six ICUs aimed to assess the association of a telemedicine intervention with clinical outcomes. The intervention consisted of a remote office with real-time audiovisual monitoring, vital signs, early warning signals, and other electronic data. Comparing preintervention (n=2,034) and postintervention (n=2,108) groups, there were no differences in mortality, LOS, or complications.
Overall, the general limitation of the study was that integration of the tele-ICU and actual ICUs was limited. Physicians for nearly two-thirds of the patients chose “minimal delegation” to the tele-ICU physician. Tele-ICU involvement was particularly limited in “closed” units, which were already staffed by on-site intensivists. Furthermore, despite access to various real-time data, critical elements of the record such as physician order entry and progress notes were not shared in real time; notes, for example, required daily faxing.
While it is unfortunate that the study could not evaluate the full potential of the adjunctive tele-ICU, it illustrates the real-world obstacles of integrating such technology into clinical practice. In future studies, a standardized telemedicine approach might facilitate evaluation efforts.
Bottom line: While this study demonstrated no benefit of telemedicine, study limitations preclude conclusions. Further studies are needed.
Citation: Thomas EJ, Lucke JF, Wueste L, Weavind L, Patel B. Association of telemedicine for remote monitoring of intensive care patients with mortality, complications, and length of stay. JAMA. 2009;302(24):2671-2678.
Dr. Kim is a hospitalist at Brigham and Women's Hospital in Boston, and an instructor at Harvard Medical School.
For more reviews of HM-related research, visit our website.
Clinical question: Does remote ICU monitoring improve mortality and length of stay?
Background: A shortage of intensivists has led to increased use of remote ICU monitoring or telemedicine technology to allow intensivists to remotely and simultaneously care for patients in multiple ICUs. Data evaluating this practice have been limited.
Study design: Pre- and postintervention observational study.
Setting: Open and closed medical-surgical ICUs in community, urban, and tertiary-care teaching U.S. hospitals.
Synopsis: This observational study in six ICUs aimed to assess the association of a telemedicine intervention with clinical outcomes. The intervention consisted of a remote office with real-time audiovisual monitoring, vital signs, early warning signals, and other electronic data. Comparing preintervention (n=2,034) and postintervention (n=2,108) groups, there were no differences in mortality, LOS, or complications.
Overall, the general limitation of the study was that integration of the tele-ICU and actual ICUs was limited. Physicians for nearly two-thirds of the patients chose “minimal delegation” to the tele-ICU physician. Tele-ICU involvement was particularly limited in “closed” units, which were already staffed by on-site intensivists. Furthermore, despite access to various real-time data, critical elements of the record such as physician order entry and progress notes were not shared in real time; notes, for example, required daily faxing.
While it is unfortunate that the study could not evaluate the full potential of the adjunctive tele-ICU, it illustrates the real-world obstacles of integrating such technology into clinical practice. In future studies, a standardized telemedicine approach might facilitate evaluation efforts.
Bottom line: While this study demonstrated no benefit of telemedicine, study limitations preclude conclusions. Further studies are needed.
Citation: Thomas EJ, Lucke JF, Wueste L, Weavind L, Patel B. Association of telemedicine for remote monitoring of intensive care patients with mortality, complications, and length of stay. JAMA. 2009;302(24):2671-2678.
Dr. Kim is a hospitalist at Brigham and Women's Hospital in Boston, and an instructor at Harvard Medical School.
For more reviews of HM-related research, visit our website.
Next Stop on Cost-Cutting Train: Readmission Reductions
When and how the national focus on reducing hospital readmissions will hit hospitals’ bottom lines is not clear, but it’s more a matter of when, not if, says Eric Coleman, MD, MPH, AGSF, FACP, director of the Care Transitions Program at the University of Colorado Denver.
Reducing readmissions “jumps off the page as an area where we could see enormous savings in national health expenditures,” Dr. Coleman told participants in an SHM webinar last month. The challenge, he said, is to align incentives with quality and safety for a moving target that also happens to be highly politicized. “We’re generally pretty good at identifying who’s at risk of readmission, but it’s harder to say who’s at modifiable risk,” he explained.
Evidence shows that hospitalists already reduce costs through improved length of stay. “Can hospitalists demonstrate the ability to reduce readmission rates as well?” Dr. Coleman asked.
Bundling payment for hospital stays with various post-hospital providers is a major focus of national efforts to reduce healthcare costs. Bundling gives providers on the healthcare continuum strong motivation to work together, Dr. Coleman said. The government won’t tell providers how to divide bundled payments, but Dr. Coleman predicts that consulting firms offering ideas for divvying up the money will emerge.
The Medicare Payment Advisory Commission (MEDPAC) has signaled its interest in changing payment incentives by reducing reimbursement for readmissions as well as several provisions that directly address readmissions in the healthcare reform package signed by President Obama in March. These include:
- A national pilot program on payment bundling;
- A hospital readmissions reduction program with financial penalties starting in October 2012 for select conditions; and
- A QI program to help hospitals with high severity-adjusted readmission rates.
When and how the national focus on reducing hospital readmissions will hit hospitals’ bottom lines is not clear, but it’s more a matter of when, not if, says Eric Coleman, MD, MPH, AGSF, FACP, director of the Care Transitions Program at the University of Colorado Denver.
Reducing readmissions “jumps off the page as an area where we could see enormous savings in national health expenditures,” Dr. Coleman told participants in an SHM webinar last month. The challenge, he said, is to align incentives with quality and safety for a moving target that also happens to be highly politicized. “We’re generally pretty good at identifying who’s at risk of readmission, but it’s harder to say who’s at modifiable risk,” he explained.
Evidence shows that hospitalists already reduce costs through improved length of stay. “Can hospitalists demonstrate the ability to reduce readmission rates as well?” Dr. Coleman asked.
Bundling payment for hospital stays with various post-hospital providers is a major focus of national efforts to reduce healthcare costs. Bundling gives providers on the healthcare continuum strong motivation to work together, Dr. Coleman said. The government won’t tell providers how to divide bundled payments, but Dr. Coleman predicts that consulting firms offering ideas for divvying up the money will emerge.
The Medicare Payment Advisory Commission (MEDPAC) has signaled its interest in changing payment incentives by reducing reimbursement for readmissions as well as several provisions that directly address readmissions in the healthcare reform package signed by President Obama in March. These include:
- A national pilot program on payment bundling;
- A hospital readmissions reduction program with financial penalties starting in October 2012 for select conditions; and
- A QI program to help hospitals with high severity-adjusted readmission rates.
When and how the national focus on reducing hospital readmissions will hit hospitals’ bottom lines is not clear, but it’s more a matter of when, not if, says Eric Coleman, MD, MPH, AGSF, FACP, director of the Care Transitions Program at the University of Colorado Denver.
Reducing readmissions “jumps off the page as an area where we could see enormous savings in national health expenditures,” Dr. Coleman told participants in an SHM webinar last month. The challenge, he said, is to align incentives with quality and safety for a moving target that also happens to be highly politicized. “We’re generally pretty good at identifying who’s at risk of readmission, but it’s harder to say who’s at modifiable risk,” he explained.
Evidence shows that hospitalists already reduce costs through improved length of stay. “Can hospitalists demonstrate the ability to reduce readmission rates as well?” Dr. Coleman asked.
Bundling payment for hospital stays with various post-hospital providers is a major focus of national efforts to reduce healthcare costs. Bundling gives providers on the healthcare continuum strong motivation to work together, Dr. Coleman said. The government won’t tell providers how to divide bundled payments, but Dr. Coleman predicts that consulting firms offering ideas for divvying up the money will emerge.
The Medicare Payment Advisory Commission (MEDPAC) has signaled its interest in changing payment incentives by reducing reimbursement for readmissions as well as several provisions that directly address readmissions in the healthcare reform package signed by President Obama in March. These include:
- A national pilot program on payment bundling;
- A hospital readmissions reduction program with financial penalties starting in October 2012 for select conditions; and
- A QI program to help hospitals with high severity-adjusted readmission rates.
Obama Confirms Partners’ Visitation, Decision Rights
HM leaders should pay close attention to the landmark decision by President Obama to extend visitation and critical-care decision rights to the partners of gay and lesbian patients, according to one hospitalist.
“The hospitalists would be the gatekeepers, in some ways, to make those calls,” says Heather Whelan, MD, an assistant professor and medical director at Mount Zion Medical Service, Division of Hospital Medicine, at the University of California at San Francisco. “I think hospitalists, group managers, and hospitals themselves are going to build this into policies and practices.”
Via a memo written in mid-April, the president mandated that all hospitals receiving federal funding must extend visitation rights to gay partners and “respect the patient’s choices about who may make critical heath-care decisions for them,” according to the Washington Post. Gay activists cheered the decisions; many conservative groups decried it.
An SHM spokesperson says the society “always expects that its hospitalists and their policies know and comply with federal, state and local regulations.”
Dr. Whelan notes that many hospitals in more progressive areas of the country—including San Francisco and New York City—already extend the rights that Obama has now codified. However, she says she has heard from physicians working elsewhere that the issue can crop up on who can be involved in critical-care meetings and decisions. The issue can be particularly sensitive in cases in which the patient is noncommunicative due to a stroke or other acute condition.
“This will standardize care,” Dr. Whelan says, adding, “It almost doesn’t matter how frequently it comes up. It’s a high-stakes event. One or two instances of it coming up are too many.”
HM leaders should pay close attention to the landmark decision by President Obama to extend visitation and critical-care decision rights to the partners of gay and lesbian patients, according to one hospitalist.
“The hospitalists would be the gatekeepers, in some ways, to make those calls,” says Heather Whelan, MD, an assistant professor and medical director at Mount Zion Medical Service, Division of Hospital Medicine, at the University of California at San Francisco. “I think hospitalists, group managers, and hospitals themselves are going to build this into policies and practices.”
Via a memo written in mid-April, the president mandated that all hospitals receiving federal funding must extend visitation rights to gay partners and “respect the patient’s choices about who may make critical heath-care decisions for them,” according to the Washington Post. Gay activists cheered the decisions; many conservative groups decried it.
An SHM spokesperson says the society “always expects that its hospitalists and their policies know and comply with federal, state and local regulations.”
Dr. Whelan notes that many hospitals in more progressive areas of the country—including San Francisco and New York City—already extend the rights that Obama has now codified. However, she says she has heard from physicians working elsewhere that the issue can crop up on who can be involved in critical-care meetings and decisions. The issue can be particularly sensitive in cases in which the patient is noncommunicative due to a stroke or other acute condition.
“This will standardize care,” Dr. Whelan says, adding, “It almost doesn’t matter how frequently it comes up. It’s a high-stakes event. One or two instances of it coming up are too many.”
HM leaders should pay close attention to the landmark decision by President Obama to extend visitation and critical-care decision rights to the partners of gay and lesbian patients, according to one hospitalist.
“The hospitalists would be the gatekeepers, in some ways, to make those calls,” says Heather Whelan, MD, an assistant professor and medical director at Mount Zion Medical Service, Division of Hospital Medicine, at the University of California at San Francisco. “I think hospitalists, group managers, and hospitals themselves are going to build this into policies and practices.”
Via a memo written in mid-April, the president mandated that all hospitals receiving federal funding must extend visitation rights to gay partners and “respect the patient’s choices about who may make critical heath-care decisions for them,” according to the Washington Post. Gay activists cheered the decisions; many conservative groups decried it.
An SHM spokesperson says the society “always expects that its hospitalists and their policies know and comply with federal, state and local regulations.”
Dr. Whelan notes that many hospitals in more progressive areas of the country—including San Francisco and New York City—already extend the rights that Obama has now codified. However, she says she has heard from physicians working elsewhere that the issue can crop up on who can be involved in critical-care meetings and decisions. The issue can be particularly sensitive in cases in which the patient is noncommunicative due to a stroke or other acute condition.
“This will standardize care,” Dr. Whelan says, adding, “It almost doesn’t matter how frequently it comes up. It’s a high-stakes event. One or two instances of it coming up are too many.”