User login
Study identifies GI bleed risk factors in von Willebrand disease
Researchers have identified several risk factors associated with gastrointestinal (GI) bleeding in adult patients with von Willebrand disease (VWD), based on findings from a retrospective analysis.
“[We] evaluated prevalence and risk factors of GIB among individuals with and without VWD, using a large national database,” wrote Anastasia Tsagianni, MD, of the University of Pittsburgh, and colleagues. The findings were published in Thrombosis Research.
The researchers retrospectively reviewed discharge data from the National Inpatient Sample database. The team analyzed correlates of GI bleeding among patients with VWD and estimated prevalence rates using measures in the database. Risk factors for GI bleeding in VWD were correlated via multivariable logistic regression.
Between Jan. 1, 2009, and Dec. 31, 2014, there were 16,640 admissions with VWD and 618 were admitted with GI bleeding, the researchers reported. After analysis, the researchers found that the prevalence of GI bleeding was 3.7% and 1.49% in VWD and non-VWD patients, respectively – a 2.5-fold greater rate in VWD patients.
“Comorbidities associated with greater [GI bleeding] risk in individuals with VWD include past surgery, hypertension, hyperlipidemia, smoking, renal disease, hepatitis C, thrombocytopenia, or liver disease,” the researchers wrote.
In the multivariable analysis, the factors associated with GI bleeding were smoking status (odds ratio, 1.40), African American race (OR, 1.80), male gender (OR, 1.61), angiodysplasia (OR, 104.06), colonic diverticulitis (OR, 16.66), and hepatitis C (OR 2.17). These variables were similar to risk factors identified in the non-VWD group, the researchers noted.
“Although significant, age did not appear to be a strong risk factor for either group,” they wrote. “Steroids were not associated with increased risk for [GI bleeding] in either group.”
A key limitation of the study was the use of discharge diagnosis codes as an inclusion method, the researchers noted. As a result, misclassification bias could be present, they added.
The Pennsylvania Department of Health and the Health Resources and Services Administration funded the study. The authors reported having no conflicts of interest.
SOURCE: Tsagianni A et al. Thromb Res. 2019 Apr 17. doi: 10.1016/j.thromres.2019.04.017.
Researchers have identified several risk factors associated with gastrointestinal (GI) bleeding in adult patients with von Willebrand disease (VWD), based on findings from a retrospective analysis.
“[We] evaluated prevalence and risk factors of GIB among individuals with and without VWD, using a large national database,” wrote Anastasia Tsagianni, MD, of the University of Pittsburgh, and colleagues. The findings were published in Thrombosis Research.
The researchers retrospectively reviewed discharge data from the National Inpatient Sample database. The team analyzed correlates of GI bleeding among patients with VWD and estimated prevalence rates using measures in the database. Risk factors for GI bleeding in VWD were correlated via multivariable logistic regression.
Between Jan. 1, 2009, and Dec. 31, 2014, there were 16,640 admissions with VWD and 618 were admitted with GI bleeding, the researchers reported. After analysis, the researchers found that the prevalence of GI bleeding was 3.7% and 1.49% in VWD and non-VWD patients, respectively – a 2.5-fold greater rate in VWD patients.
“Comorbidities associated with greater [GI bleeding] risk in individuals with VWD include past surgery, hypertension, hyperlipidemia, smoking, renal disease, hepatitis C, thrombocytopenia, or liver disease,” the researchers wrote.
In the multivariable analysis, the factors associated with GI bleeding were smoking status (odds ratio, 1.40), African American race (OR, 1.80), male gender (OR, 1.61), angiodysplasia (OR, 104.06), colonic diverticulitis (OR, 16.66), and hepatitis C (OR 2.17). These variables were similar to risk factors identified in the non-VWD group, the researchers noted.
“Although significant, age did not appear to be a strong risk factor for either group,” they wrote. “Steroids were not associated with increased risk for [GI bleeding] in either group.”
A key limitation of the study was the use of discharge diagnosis codes as an inclusion method, the researchers noted. As a result, misclassification bias could be present, they added.
The Pennsylvania Department of Health and the Health Resources and Services Administration funded the study. The authors reported having no conflicts of interest.
SOURCE: Tsagianni A et al. Thromb Res. 2019 Apr 17. doi: 10.1016/j.thromres.2019.04.017.
Researchers have identified several risk factors associated with gastrointestinal (GI) bleeding in adult patients with von Willebrand disease (VWD), based on findings from a retrospective analysis.
“[We] evaluated prevalence and risk factors of GIB among individuals with and without VWD, using a large national database,” wrote Anastasia Tsagianni, MD, of the University of Pittsburgh, and colleagues. The findings were published in Thrombosis Research.
The researchers retrospectively reviewed discharge data from the National Inpatient Sample database. The team analyzed correlates of GI bleeding among patients with VWD and estimated prevalence rates using measures in the database. Risk factors for GI bleeding in VWD were correlated via multivariable logistic regression.
Between Jan. 1, 2009, and Dec. 31, 2014, there were 16,640 admissions with VWD and 618 were admitted with GI bleeding, the researchers reported. After analysis, the researchers found that the prevalence of GI bleeding was 3.7% and 1.49% in VWD and non-VWD patients, respectively – a 2.5-fold greater rate in VWD patients.
“Comorbidities associated with greater [GI bleeding] risk in individuals with VWD include past surgery, hypertension, hyperlipidemia, smoking, renal disease, hepatitis C, thrombocytopenia, or liver disease,” the researchers wrote.
In the multivariable analysis, the factors associated with GI bleeding were smoking status (odds ratio, 1.40), African American race (OR, 1.80), male gender (OR, 1.61), angiodysplasia (OR, 104.06), colonic diverticulitis (OR, 16.66), and hepatitis C (OR 2.17). These variables were similar to risk factors identified in the non-VWD group, the researchers noted.
“Although significant, age did not appear to be a strong risk factor for either group,” they wrote. “Steroids were not associated with increased risk for [GI bleeding] in either group.”
A key limitation of the study was the use of discharge diagnosis codes as an inclusion method, the researchers noted. As a result, misclassification bias could be present, they added.
The Pennsylvania Department of Health and the Health Resources and Services Administration funded the study. The authors reported having no conflicts of interest.
SOURCE: Tsagianni A et al. Thromb Res. 2019 Apr 17. doi: 10.1016/j.thromres.2019.04.017.
FROM THROMBOSIS RESEARCH
Key clinical point: Researchers identified several risk factors associated for gastrointestinal bleeding in adult patients with von Willebrand disease.
Major finding: In a multivariate analysis, the most significant factors associated with gastrointestinal bleeding in von Willebrand disease were smoking status, African American race, male gender, angiodysplasia, diverticulitis, and hepatitis C.
Study details: A retrospective analysis of 16,640 patients with the disease.
Disclosures: The Pennsylvania Department of Health and the Health Resources and Services Administration funded the study. The authors reported having no conflicts of interest.
Source: Tsagianni A et al. Thromb Res. 2019 Apr 17. doi: 10.1016/j.thromres.2019.04.017.
Insomnia meds get boxed warning from FDA
The Food and Drug Administration will now require that certain
Complex sleep behaviors have been seen with these medications in patients with and without a history of them, at low doses, and even after one dose of the medication. They’ve also been observed with and without concomitant use of alcohol or other CNS depressants.
Health care professionals should advise patients about these risks, even though they are rare. Patients should contact health care professionals if they either experience a complex sleep behavior while not fully awake on one of these medicines or have performed activities they don’t remember while taking the medicine.
More information about these risks and the safety warnings can be found in the FDA’s safety announcement. Other information is also available in a press announcement from the agency.
The Food and Drug Administration will now require that certain
Complex sleep behaviors have been seen with these medications in patients with and without a history of them, at low doses, and even after one dose of the medication. They’ve also been observed with and without concomitant use of alcohol or other CNS depressants.
Health care professionals should advise patients about these risks, even though they are rare. Patients should contact health care professionals if they either experience a complex sleep behavior while not fully awake on one of these medicines or have performed activities they don’t remember while taking the medicine.
More information about these risks and the safety warnings can be found in the FDA’s safety announcement. Other information is also available in a press announcement from the agency.
The Food and Drug Administration will now require that certain
Complex sleep behaviors have been seen with these medications in patients with and without a history of them, at low doses, and even after one dose of the medication. They’ve also been observed with and without concomitant use of alcohol or other CNS depressants.
Health care professionals should advise patients about these risks, even though they are rare. Patients should contact health care professionals if they either experience a complex sleep behavior while not fully awake on one of these medicines or have performed activities they don’t remember while taking the medicine.
More information about these risks and the safety warnings can be found in the FDA’s safety announcement. Other information is also available in a press announcement from the agency.
Time to embrace minimally invasive colorectal surgery?
LAS VEGAS – Two-thirds of colon resections in the United States are open procedures, but a colorectal surgeon told colleagues that evidence shows minimally invasive surgery deserves a wider place in his field.
Why? Because minimally invasive surgery – despite its limited utilization – is linked to multiple improved outcomes in colorectal surgery, said Matthew G. Mutch, MD, chief of colon and rectal surgery at Washington University, St. Louis, in a presentation at the Annual Minimally Invasive Surgery Symposium by Global Academy for Medical Education.
“Our goal should be to offer minimally invasive surgery to as many patients as possible by as many different methods as needed,” Dr. Mutch said. “If you’re willing to take this on and do this over a regular basis, you’ll get over that learning curve and expand the number of patients you can offer laparoscopy to.”
According to Dr. Mutch, benefits of minimally invasive colorectal surgery include:
- Improved short-term outcomes – length of stay and return of bowel function, and morbidity and mortality. A 2012 retrospective study of 85,712 colon resections that found laparoscopic resections, when feasible, “had better outcomes than open colectomy in the immediate perioperative period.” (Ann Surg. 2012 Sep;256[3]462-8).
- Improved long-term outcomes: faster recovery, fewer hernias, and fewer bowel obstructions.
- Lower overall costs.
- Fewer complications in the elderly.
When it comes to laparoscopic colorectal surgery, Dr. Mutch cautioned that the robotic technology has unclear benefit in rectal cancer, and the cost in colorectal cancer is unclear.
Another alternative is to perform laparoscopic colorectal surgery through alternative extraction sites such as the rectum, vagina, stomach, and even a stoma site or perineal wound. Both transanal and transvaginal extraction are feasible and safe, he said, adding that transvaginal procedures are best performed in conjunction with a hysterectomy. One benefit of these procedures is that they avoid abdominal wall trauma. However, he cautioned that colorectal surgery is unique because a cancerous specimen cannot be morcellated and must instead be removed whole.
Dr. Mutch also discussed laparoendoscopic resection of colon polyps. Benefits include shorter length of stay and faster recovery, he said, but complications can include perforation and bleeding. And, he said, there’s currently no code for the procedure.
Global Academy for Medical Education and this news organization are owned by the same parent company. Dr. Mutch has no relevant disclosures.
LAS VEGAS – Two-thirds of colon resections in the United States are open procedures, but a colorectal surgeon told colleagues that evidence shows minimally invasive surgery deserves a wider place in his field.
Why? Because minimally invasive surgery – despite its limited utilization – is linked to multiple improved outcomes in colorectal surgery, said Matthew G. Mutch, MD, chief of colon and rectal surgery at Washington University, St. Louis, in a presentation at the Annual Minimally Invasive Surgery Symposium by Global Academy for Medical Education.
“Our goal should be to offer minimally invasive surgery to as many patients as possible by as many different methods as needed,” Dr. Mutch said. “If you’re willing to take this on and do this over a regular basis, you’ll get over that learning curve and expand the number of patients you can offer laparoscopy to.”
According to Dr. Mutch, benefits of minimally invasive colorectal surgery include:
- Improved short-term outcomes – length of stay and return of bowel function, and morbidity and mortality. A 2012 retrospective study of 85,712 colon resections that found laparoscopic resections, when feasible, “had better outcomes than open colectomy in the immediate perioperative period.” (Ann Surg. 2012 Sep;256[3]462-8).
- Improved long-term outcomes: faster recovery, fewer hernias, and fewer bowel obstructions.
- Lower overall costs.
- Fewer complications in the elderly.
When it comes to laparoscopic colorectal surgery, Dr. Mutch cautioned that the robotic technology has unclear benefit in rectal cancer, and the cost in colorectal cancer is unclear.
Another alternative is to perform laparoscopic colorectal surgery through alternative extraction sites such as the rectum, vagina, stomach, and even a stoma site or perineal wound. Both transanal and transvaginal extraction are feasible and safe, he said, adding that transvaginal procedures are best performed in conjunction with a hysterectomy. One benefit of these procedures is that they avoid abdominal wall trauma. However, he cautioned that colorectal surgery is unique because a cancerous specimen cannot be morcellated and must instead be removed whole.
Dr. Mutch also discussed laparoendoscopic resection of colon polyps. Benefits include shorter length of stay and faster recovery, he said, but complications can include perforation and bleeding. And, he said, there’s currently no code for the procedure.
Global Academy for Medical Education and this news organization are owned by the same parent company. Dr. Mutch has no relevant disclosures.
LAS VEGAS – Two-thirds of colon resections in the United States are open procedures, but a colorectal surgeon told colleagues that evidence shows minimally invasive surgery deserves a wider place in his field.
Why? Because minimally invasive surgery – despite its limited utilization – is linked to multiple improved outcomes in colorectal surgery, said Matthew G. Mutch, MD, chief of colon and rectal surgery at Washington University, St. Louis, in a presentation at the Annual Minimally Invasive Surgery Symposium by Global Academy for Medical Education.
“Our goal should be to offer minimally invasive surgery to as many patients as possible by as many different methods as needed,” Dr. Mutch said. “If you’re willing to take this on and do this over a regular basis, you’ll get over that learning curve and expand the number of patients you can offer laparoscopy to.”
According to Dr. Mutch, benefits of minimally invasive colorectal surgery include:
- Improved short-term outcomes – length of stay and return of bowel function, and morbidity and mortality. A 2012 retrospective study of 85,712 colon resections that found laparoscopic resections, when feasible, “had better outcomes than open colectomy in the immediate perioperative period.” (Ann Surg. 2012 Sep;256[3]462-8).
- Improved long-term outcomes: faster recovery, fewer hernias, and fewer bowel obstructions.
- Lower overall costs.
- Fewer complications in the elderly.
When it comes to laparoscopic colorectal surgery, Dr. Mutch cautioned that the robotic technology has unclear benefit in rectal cancer, and the cost in colorectal cancer is unclear.
Another alternative is to perform laparoscopic colorectal surgery through alternative extraction sites such as the rectum, vagina, stomach, and even a stoma site or perineal wound. Both transanal and transvaginal extraction are feasible and safe, he said, adding that transvaginal procedures are best performed in conjunction with a hysterectomy. One benefit of these procedures is that they avoid abdominal wall trauma. However, he cautioned that colorectal surgery is unique because a cancerous specimen cannot be morcellated and must instead be removed whole.
Dr. Mutch also discussed laparoendoscopic resection of colon polyps. Benefits include shorter length of stay and faster recovery, he said, but complications can include perforation and bleeding. And, he said, there’s currently no code for the procedure.
Global Academy for Medical Education and this news organization are owned by the same parent company. Dr. Mutch has no relevant disclosures.
EXPERT ANALYSIS FROM MISS
Induction trough levels predicted ustekinumab response in Crohn’s disease
For patients with Crohn’s disease, therapeutic drug monitoring helped identify early primary nonresponders to induction with ustekinumab, according to researchers. The report is in Clinical Gastroenterology and Hepatology.
At week 8, median trough levels of ustekinumab were 6.0 mcg per mL (interquartile range, 3.1-8.0) among patients who achieved a primary response to induction at week 16, versus 1.3 mcg/mL (IQR, 0.9-5.6 ) among primary nonresponders (P = .03). An 8-week ustekinumab trough level cutoff of 2.0 mcg/mL distinguished week 16 responders from nonresponders with an area under the receiver operating curve (AUROC) of 0.75, wrote Ninon Soufflet of University Claude Bernard Lyon 1 in France, and associates. The researchers recommended “dedicated studies” to assess whether escalating the dose of ustekinumab can benefit patients with lower trough levels at week 8.
Few studies have explored biomarkers for response to ustekinumab induction therapy. Hence, the researchers assessed the relative utility of ustekinumab trough levels, C-reactive protein (CRP) levels, and fecal calprotectin levels for predicting early primary nonresponse. All 51 study participants had active luminal Crohn’s disease and received body weight–based intravenous infusions of ustekinumab at baseline, followed by subcutaneous injections of 90 mg. Primary nonresponders did not achieve steroid-free clinical and biochemical remission at week 16, defined as a Harvey-Bradshaw Index (HBI) of 4 points or less, a CRP level under 5 mg/L, and a fecal calprotectin level under 250 mcg/g. Week 16 was chosen to account for any delayed responders, the researchers noted.
A total of 32 patients (63%) achieved remission to ustekinumab induction therapy by week 16. An 8-week trough level of 2.0 mcg/mL was found to be optimal and distinguished primary nonresponders from responders with a sensitivity of 87%, a specificity of 66%, a positive predictive value of 82%, and a negative predictive value of 75%. In prior studies, optimal thresholds exceeded 3.3 mcg/mL for achieving remission and 4.5 mcg/mL at week 26 for achieving endoscopic response, the researchers noted. They said that this discrepancy might reflect different time points for evaluation, assays for measuring ustekinumab, patient populations, and a lack of endoscopic data in their study. “The relatively small sample size and the short period of follow-up evaluation [were] substantial limitations” they acknowledged.
In this study, levels of CRP did not change significantly between weeks 0 and 16 among either responders or nonresponders. In contrast, fecal calprotectin levels dropped rapidly and significantly over time only in responders. Median fecal calprotectin levels were 1,612 mcg/g of stools at week 0 versus 374 mcg/g at week 4 and 339 mcg/g at week 8. The finding “confirms the value of this biomarker, as previously shown in inflammatory bowel disease with anti–tumor necrosis factor,” the researchers wrote.
The investigators did not acknowledge external funding sources. Dr. Soufflet reported having no conflicts of interest. The senior author and three coinvestigators disclosed ties to MSD, AbbVie, Tillots, and several other pharmaceutical companies.
Learn more about therapeutic drug monitoring in IBD by reviewing the AGA Institute guideline at http://www.gastrojournal.org/article/S0016-5085(17)35963-2/fulltext.
SOURCE: Soufflet N et al. Clin Gastroenterol Hepatol. 2019 Mar 6. doi: 10.1016/j.cgh.2019.02.042. https://www.cghjournal.org/article/S1542-3565(19)30248-4/abstract
For patients with Crohn’s disease, therapeutic drug monitoring helped identify early primary nonresponders to induction with ustekinumab, according to researchers. The report is in Clinical Gastroenterology and Hepatology.
At week 8, median trough levels of ustekinumab were 6.0 mcg per mL (interquartile range, 3.1-8.0) among patients who achieved a primary response to induction at week 16, versus 1.3 mcg/mL (IQR, 0.9-5.6 ) among primary nonresponders (P = .03). An 8-week ustekinumab trough level cutoff of 2.0 mcg/mL distinguished week 16 responders from nonresponders with an area under the receiver operating curve (AUROC) of 0.75, wrote Ninon Soufflet of University Claude Bernard Lyon 1 in France, and associates. The researchers recommended “dedicated studies” to assess whether escalating the dose of ustekinumab can benefit patients with lower trough levels at week 8.
Few studies have explored biomarkers for response to ustekinumab induction therapy. Hence, the researchers assessed the relative utility of ustekinumab trough levels, C-reactive protein (CRP) levels, and fecal calprotectin levels for predicting early primary nonresponse. All 51 study participants had active luminal Crohn’s disease and received body weight–based intravenous infusions of ustekinumab at baseline, followed by subcutaneous injections of 90 mg. Primary nonresponders did not achieve steroid-free clinical and biochemical remission at week 16, defined as a Harvey-Bradshaw Index (HBI) of 4 points or less, a CRP level under 5 mg/L, and a fecal calprotectin level under 250 mcg/g. Week 16 was chosen to account for any delayed responders, the researchers noted.
A total of 32 patients (63%) achieved remission to ustekinumab induction therapy by week 16. An 8-week trough level of 2.0 mcg/mL was found to be optimal and distinguished primary nonresponders from responders with a sensitivity of 87%, a specificity of 66%, a positive predictive value of 82%, and a negative predictive value of 75%. In prior studies, optimal thresholds exceeded 3.3 mcg/mL for achieving remission and 4.5 mcg/mL at week 26 for achieving endoscopic response, the researchers noted. They said that this discrepancy might reflect different time points for evaluation, assays for measuring ustekinumab, patient populations, and a lack of endoscopic data in their study. “The relatively small sample size and the short period of follow-up evaluation [were] substantial limitations” they acknowledged.
In this study, levels of CRP did not change significantly between weeks 0 and 16 among either responders or nonresponders. In contrast, fecal calprotectin levels dropped rapidly and significantly over time only in responders. Median fecal calprotectin levels were 1,612 mcg/g of stools at week 0 versus 374 mcg/g at week 4 and 339 mcg/g at week 8. The finding “confirms the value of this biomarker, as previously shown in inflammatory bowel disease with anti–tumor necrosis factor,” the researchers wrote.
The investigators did not acknowledge external funding sources. Dr. Soufflet reported having no conflicts of interest. The senior author and three coinvestigators disclosed ties to MSD, AbbVie, Tillots, and several other pharmaceutical companies.
Learn more about therapeutic drug monitoring in IBD by reviewing the AGA Institute guideline at http://www.gastrojournal.org/article/S0016-5085(17)35963-2/fulltext.
SOURCE: Soufflet N et al. Clin Gastroenterol Hepatol. 2019 Mar 6. doi: 10.1016/j.cgh.2019.02.042. https://www.cghjournal.org/article/S1542-3565(19)30248-4/abstract
For patients with Crohn’s disease, therapeutic drug monitoring helped identify early primary nonresponders to induction with ustekinumab, according to researchers. The report is in Clinical Gastroenterology and Hepatology.
At week 8, median trough levels of ustekinumab were 6.0 mcg per mL (interquartile range, 3.1-8.0) among patients who achieved a primary response to induction at week 16, versus 1.3 mcg/mL (IQR, 0.9-5.6 ) among primary nonresponders (P = .03). An 8-week ustekinumab trough level cutoff of 2.0 mcg/mL distinguished week 16 responders from nonresponders with an area under the receiver operating curve (AUROC) of 0.75, wrote Ninon Soufflet of University Claude Bernard Lyon 1 in France, and associates. The researchers recommended “dedicated studies” to assess whether escalating the dose of ustekinumab can benefit patients with lower trough levels at week 8.
Few studies have explored biomarkers for response to ustekinumab induction therapy. Hence, the researchers assessed the relative utility of ustekinumab trough levels, C-reactive protein (CRP) levels, and fecal calprotectin levels for predicting early primary nonresponse. All 51 study participants had active luminal Crohn’s disease and received body weight–based intravenous infusions of ustekinumab at baseline, followed by subcutaneous injections of 90 mg. Primary nonresponders did not achieve steroid-free clinical and biochemical remission at week 16, defined as a Harvey-Bradshaw Index (HBI) of 4 points or less, a CRP level under 5 mg/L, and a fecal calprotectin level under 250 mcg/g. Week 16 was chosen to account for any delayed responders, the researchers noted.
A total of 32 patients (63%) achieved remission to ustekinumab induction therapy by week 16. An 8-week trough level of 2.0 mcg/mL was found to be optimal and distinguished primary nonresponders from responders with a sensitivity of 87%, a specificity of 66%, a positive predictive value of 82%, and a negative predictive value of 75%. In prior studies, optimal thresholds exceeded 3.3 mcg/mL for achieving remission and 4.5 mcg/mL at week 26 for achieving endoscopic response, the researchers noted. They said that this discrepancy might reflect different time points for evaluation, assays for measuring ustekinumab, patient populations, and a lack of endoscopic data in their study. “The relatively small sample size and the short period of follow-up evaluation [were] substantial limitations” they acknowledged.
In this study, levels of CRP did not change significantly between weeks 0 and 16 among either responders or nonresponders. In contrast, fecal calprotectin levels dropped rapidly and significantly over time only in responders. Median fecal calprotectin levels were 1,612 mcg/g of stools at week 0 versus 374 mcg/g at week 4 and 339 mcg/g at week 8. The finding “confirms the value of this biomarker, as previously shown in inflammatory bowel disease with anti–tumor necrosis factor,” the researchers wrote.
The investigators did not acknowledge external funding sources. Dr. Soufflet reported having no conflicts of interest. The senior author and three coinvestigators disclosed ties to MSD, AbbVie, Tillots, and several other pharmaceutical companies.
Learn more about therapeutic drug monitoring in IBD by reviewing the AGA Institute guideline at http://www.gastrojournal.org/article/S0016-5085(17)35963-2/fulltext.
SOURCE: Soufflet N et al. Clin Gastroenterol Hepatol. 2019 Mar 6. doi: 10.1016/j.cgh.2019.02.042. https://www.cghjournal.org/article/S1542-3565(19)30248-4/abstract
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: For patients with Crohn’s disease, therapeutic drug monitoring helped identify early nonresponse to ustekinumab induction.
Major finding: At week 8, median trough ustekinumab levels were 6 mcg/mL in week 16 responders and 1.3 mcg/mL in week 16 nonresponders (P = .03). An ustekinumab trough level of 2.0 mcg/mL or higher distinguished responders from nonresponders with an area under the receiver operating curve of 0.75.
Study details: Prospective study of 51 patients with active luminal Crohn’s disease.
Disclosures: The researchers did not acknowledge external funding sources. Dr. Soufflet reported having no conflicts of interest. The senior author and three coinvestigators disclosed ties to MSD, AbbVie, Tillots, and several other pharmaceutical companies.
Source: Soufflet N et al. Clin Gastroenterol Hepatol. 2019 Mar 6. doi: 10.1016/j.cgh.2019.02.042.
Is elagolix safely effective at reducing heavy uterine bleeding in premenopausal women with uterine fibroids?
Uterine fibroids affect up to two-thirds of all women.1 The current medical treatment options for uterine fibroids include on- and off-label use of oral contraceptives, hormonal intrauterine devices, gonadotropin-releasing hormone (GnRH) receptor agonists, and progestins. Data on the oral GnRH antagonist elagolix, US Food and Drug Administration–approved to treat endometriosis, were presented at the 2019 Annual Clinical and Scientific Meeting of the American College of Obstetricians and Gynecologists.
Study details
In the Eularis I trial, a total of 412 women with heavy menstrual bleeding (>80 mL/cycle of menstrual blood loss) were included in the double-blind, randomized, placebo-controlled, 6-month, phase 3 study. Women were premenopausal, ranged in age from 18 to 51 years, and were assigned in a 1:1:2 ratio to placebo, elagolix 300 mg twice daily, or elagolix 300 mg twice daily in combination with 1 mg estradiol/0.5 mg norethindrone acetate once-daily add-back therapy. A total of 328 women completed treatment.
At 6 months, 84.1%, 68.5%, and 8.7% of women taking elagolix alone, elagolix plus estradiol/norethindrone acetate, and placebo experienced a bleeding reduction that was 50% or greater from baseline.
Half of women who were treated with elagolix plus hormonal add-back therapy experienced adverse events, compared with 80% of women who took elagolix alone and 38% of women taking placebo. At study conclusion, the change from baseline for lumbar spine bone mass density was not significantly different from placebo for elagolix plus hormonal add-back therapy, but it was significantly different for elagolix alone compared with placebo.2
EULARIS II
A second study of safety and efficacy, which also was a double-blind, randomized, placebo-controlled, 6-month, phase 3 study, found that 76% of women randomly assigned to elagolix plus estradiol/norethindrone acetate experienced a bleeding reduction that was 50% or greater from baseline.3
Both EULARIS studies were funded by AbbVie, Inc.
- Al-Hendy A, Myers ER, Stewart E. Uterine fibroids: burden and unmet medical need. Semin Reprod Med . 2017;35:473-480.
- Elagolix reduced heavy menstrual bleeding with uterine fibroids: primary, 6-month, phase 3 results [press release]. New York, NY: W2O group; May 1, 2019.
- AbbVie announces positive topline results from phase 3 extension study evaluating investigational elagolix in women with uterine fibroids [press release]. New York, NY: PRNewswire; August 22, 2018.
Uterine fibroids affect up to two-thirds of all women.1 The current medical treatment options for uterine fibroids include on- and off-label use of oral contraceptives, hormonal intrauterine devices, gonadotropin-releasing hormone (GnRH) receptor agonists, and progestins. Data on the oral GnRH antagonist elagolix, US Food and Drug Administration–approved to treat endometriosis, were presented at the 2019 Annual Clinical and Scientific Meeting of the American College of Obstetricians and Gynecologists.
Study details
In the Eularis I trial, a total of 412 women with heavy menstrual bleeding (>80 mL/cycle of menstrual blood loss) were included in the double-blind, randomized, placebo-controlled, 6-month, phase 3 study. Women were premenopausal, ranged in age from 18 to 51 years, and were assigned in a 1:1:2 ratio to placebo, elagolix 300 mg twice daily, or elagolix 300 mg twice daily in combination with 1 mg estradiol/0.5 mg norethindrone acetate once-daily add-back therapy. A total of 328 women completed treatment.
At 6 months, 84.1%, 68.5%, and 8.7% of women taking elagolix alone, elagolix plus estradiol/norethindrone acetate, and placebo experienced a bleeding reduction that was 50% or greater from baseline.
Half of women who were treated with elagolix plus hormonal add-back therapy experienced adverse events, compared with 80% of women who took elagolix alone and 38% of women taking placebo. At study conclusion, the change from baseline for lumbar spine bone mass density was not significantly different from placebo for elagolix plus hormonal add-back therapy, but it was significantly different for elagolix alone compared with placebo.2
EULARIS II
A second study of safety and efficacy, which also was a double-blind, randomized, placebo-controlled, 6-month, phase 3 study, found that 76% of women randomly assigned to elagolix plus estradiol/norethindrone acetate experienced a bleeding reduction that was 50% or greater from baseline.3
Both EULARIS studies were funded by AbbVie, Inc.
Uterine fibroids affect up to two-thirds of all women.1 The current medical treatment options for uterine fibroids include on- and off-label use of oral contraceptives, hormonal intrauterine devices, gonadotropin-releasing hormone (GnRH) receptor agonists, and progestins. Data on the oral GnRH antagonist elagolix, US Food and Drug Administration–approved to treat endometriosis, were presented at the 2019 Annual Clinical and Scientific Meeting of the American College of Obstetricians and Gynecologists.
Study details
In the Eularis I trial, a total of 412 women with heavy menstrual bleeding (>80 mL/cycle of menstrual blood loss) were included in the double-blind, randomized, placebo-controlled, 6-month, phase 3 study. Women were premenopausal, ranged in age from 18 to 51 years, and were assigned in a 1:1:2 ratio to placebo, elagolix 300 mg twice daily, or elagolix 300 mg twice daily in combination with 1 mg estradiol/0.5 mg norethindrone acetate once-daily add-back therapy. A total of 328 women completed treatment.
At 6 months, 84.1%, 68.5%, and 8.7% of women taking elagolix alone, elagolix plus estradiol/norethindrone acetate, and placebo experienced a bleeding reduction that was 50% or greater from baseline.
Half of women who were treated with elagolix plus hormonal add-back therapy experienced adverse events, compared with 80% of women who took elagolix alone and 38% of women taking placebo. At study conclusion, the change from baseline for lumbar spine bone mass density was not significantly different from placebo for elagolix plus hormonal add-back therapy, but it was significantly different for elagolix alone compared with placebo.2
EULARIS II
A second study of safety and efficacy, which also was a double-blind, randomized, placebo-controlled, 6-month, phase 3 study, found that 76% of women randomly assigned to elagolix plus estradiol/norethindrone acetate experienced a bleeding reduction that was 50% or greater from baseline.3
Both EULARIS studies were funded by AbbVie, Inc.
- Al-Hendy A, Myers ER, Stewart E. Uterine fibroids: burden and unmet medical need. Semin Reprod Med . 2017;35:473-480.
- Elagolix reduced heavy menstrual bleeding with uterine fibroids: primary, 6-month, phase 3 results [press release]. New York, NY: W2O group; May 1, 2019.
- AbbVie announces positive topline results from phase 3 extension study evaluating investigational elagolix in women with uterine fibroids [press release]. New York, NY: PRNewswire; August 22, 2018.
- Al-Hendy A, Myers ER, Stewart E. Uterine fibroids: burden and unmet medical need. Semin Reprod Med . 2017;35:473-480.
- Elagolix reduced heavy menstrual bleeding with uterine fibroids: primary, 6-month, phase 3 results [press release]. New York, NY: W2O group; May 1, 2019.
- AbbVie announces positive topline results from phase 3 extension study evaluating investigational elagolix in women with uterine fibroids [press release]. New York, NY: PRNewswire; August 22, 2018.
Neoadjuvant removal of DCIS helps identify HER2+ nonsurgical trial candidates
Breast cancer patients with HER2-positive disease are more likely to be nonsurgical candidates for clinical trials after neoadjuvant systemic therapy if they have eradicated both invasive and ductal carcinoma in situ (DCIS) disease, according to research presented in a recent webcast from the annual meeting of the American Society of Breast Surgeons.
While there is a high rate of pathologic complete response (pCR) in HER2-postive breast cancer after neoadjuvant systemic therapy, it is difficult to determine which patients have achieved pCR because standard imaging generates a high rate of false negatives, noted Susie Sun, MD, from the University of Texas MD Anderson Cancer Center, Houston.
“Although radiological imaging such as mammograms, ultrasounds, and MRIs have been shown to be unreliable in identifying patients with pCR, we have previously determined that patients with clinically significant residual disease can be accurately identified using a combination of multimodality imaging and image-guided, vacuum-assisted biopsy to the tumor bed,” Dr. Sun said in her presentation.
In the Multicenter Trial for Eliminating Breast Cancer Surgery in Exceptional Responders With Neoadjuvant Systemic Therapy, Dr. Sun and colleagues enrolled 280 patients with T1-T2, N0-N1 HER2-positive breast cancer who had undergone HER2-targeted therapy, followed by surgical resection and axillary surgery. The researchers studied both the effects of neoadjuvant therapy patients with pCR and the clinicopathologic characteristics of residual disease to determine how patients with pCR differed from those with residual disease.
After neoadjuvant systemic therapy, 55.4% of pCR invasive cancer was eradicated in patients, 37.5% of both pCR invasive and DCIS cancer was eradicated in patients, and 17.9% of patients had eradication of only residual DCIS. Compared with patients where DCIS was not identified at initial biopsy, DCIS identification was associated with a higher likelihood of residual disease (69% vs. 57%; P = .04). The researchers found patients having hormone receptor–positive/HER2-positive disease was associated with a higher rate of predictive residual disease (26.6%), compared with patients who had hormone receptor–negative/HER2-positive disease (49.2%; odds ratio, 2.7; 95% confidence interval, P less than .0001).
“For the currently occurring trial, evaluating the safety of eliminating surgery for patients who are exceptional responders to neoadjuvant systemic therapy, eradication of both the invasive and DCIS components are necessary because DCIS may serve as a nidus for carcinoma in the future,” said Dr. Sun.
The researchers also studied the effectiveness of multimodality imaging on identifying pathologic response. The multimodality imaging consisted of a mammogram and ultrasound for all patients, and approximately 13% of patients had MRI in addition to mammogram and ultrasound. The multimodality imaging response after neoadjuvant systemic therapy had a sensitivity of 97.1% and a negative predictive value of 70.6% for detecting residual disease in the breast and lymph nodes.
“[O]ur study found that multimodality imaging was not reliable in assessing for pathologic response within the breast or lymph nodes,” said Dr. Sun. “Therefore, imaging alone cannot be used to select patients for no surgery. This requires patients who have image-guided percutaneous biopsy to safely select patients for inclusion and elimination of surgery trial.”
In a discussion session, Dr. Sun clarified the combination of multimodality imaging and image-guided percutaneous biopsy was used to select HER2-positive patients for a clinical trial, and is not standard of practice to determine pCR at the University of Texas MD Anderson Cancer Center.
Dr. Sun reported no relevant financial disclosures.
Breast cancer patients with HER2-positive disease are more likely to be nonsurgical candidates for clinical trials after neoadjuvant systemic therapy if they have eradicated both invasive and ductal carcinoma in situ (DCIS) disease, according to research presented in a recent webcast from the annual meeting of the American Society of Breast Surgeons.
While there is a high rate of pathologic complete response (pCR) in HER2-postive breast cancer after neoadjuvant systemic therapy, it is difficult to determine which patients have achieved pCR because standard imaging generates a high rate of false negatives, noted Susie Sun, MD, from the University of Texas MD Anderson Cancer Center, Houston.
“Although radiological imaging such as mammograms, ultrasounds, and MRIs have been shown to be unreliable in identifying patients with pCR, we have previously determined that patients with clinically significant residual disease can be accurately identified using a combination of multimodality imaging and image-guided, vacuum-assisted biopsy to the tumor bed,” Dr. Sun said in her presentation.
In the Multicenter Trial for Eliminating Breast Cancer Surgery in Exceptional Responders With Neoadjuvant Systemic Therapy, Dr. Sun and colleagues enrolled 280 patients with T1-T2, N0-N1 HER2-positive breast cancer who had undergone HER2-targeted therapy, followed by surgical resection and axillary surgery. The researchers studied both the effects of neoadjuvant therapy patients with pCR and the clinicopathologic characteristics of residual disease to determine how patients with pCR differed from those with residual disease.
After neoadjuvant systemic therapy, 55.4% of pCR invasive cancer was eradicated in patients, 37.5% of both pCR invasive and DCIS cancer was eradicated in patients, and 17.9% of patients had eradication of only residual DCIS. Compared with patients where DCIS was not identified at initial biopsy, DCIS identification was associated with a higher likelihood of residual disease (69% vs. 57%; P = .04). The researchers found patients having hormone receptor–positive/HER2-positive disease was associated with a higher rate of predictive residual disease (26.6%), compared with patients who had hormone receptor–negative/HER2-positive disease (49.2%; odds ratio, 2.7; 95% confidence interval, P less than .0001).
“For the currently occurring trial, evaluating the safety of eliminating surgery for patients who are exceptional responders to neoadjuvant systemic therapy, eradication of both the invasive and DCIS components are necessary because DCIS may serve as a nidus for carcinoma in the future,” said Dr. Sun.
The researchers also studied the effectiveness of multimodality imaging on identifying pathologic response. The multimodality imaging consisted of a mammogram and ultrasound for all patients, and approximately 13% of patients had MRI in addition to mammogram and ultrasound. The multimodality imaging response after neoadjuvant systemic therapy had a sensitivity of 97.1% and a negative predictive value of 70.6% for detecting residual disease in the breast and lymph nodes.
“[O]ur study found that multimodality imaging was not reliable in assessing for pathologic response within the breast or lymph nodes,” said Dr. Sun. “Therefore, imaging alone cannot be used to select patients for no surgery. This requires patients who have image-guided percutaneous biopsy to safely select patients for inclusion and elimination of surgery trial.”
In a discussion session, Dr. Sun clarified the combination of multimodality imaging and image-guided percutaneous biopsy was used to select HER2-positive patients for a clinical trial, and is not standard of practice to determine pCR at the University of Texas MD Anderson Cancer Center.
Dr. Sun reported no relevant financial disclosures.
Breast cancer patients with HER2-positive disease are more likely to be nonsurgical candidates for clinical trials after neoadjuvant systemic therapy if they have eradicated both invasive and ductal carcinoma in situ (DCIS) disease, according to research presented in a recent webcast from the annual meeting of the American Society of Breast Surgeons.
While there is a high rate of pathologic complete response (pCR) in HER2-postive breast cancer after neoadjuvant systemic therapy, it is difficult to determine which patients have achieved pCR because standard imaging generates a high rate of false negatives, noted Susie Sun, MD, from the University of Texas MD Anderson Cancer Center, Houston.
“Although radiological imaging such as mammograms, ultrasounds, and MRIs have been shown to be unreliable in identifying patients with pCR, we have previously determined that patients with clinically significant residual disease can be accurately identified using a combination of multimodality imaging and image-guided, vacuum-assisted biopsy to the tumor bed,” Dr. Sun said in her presentation.
In the Multicenter Trial for Eliminating Breast Cancer Surgery in Exceptional Responders With Neoadjuvant Systemic Therapy, Dr. Sun and colleagues enrolled 280 patients with T1-T2, N0-N1 HER2-positive breast cancer who had undergone HER2-targeted therapy, followed by surgical resection and axillary surgery. The researchers studied both the effects of neoadjuvant therapy patients with pCR and the clinicopathologic characteristics of residual disease to determine how patients with pCR differed from those with residual disease.
After neoadjuvant systemic therapy, 55.4% of pCR invasive cancer was eradicated in patients, 37.5% of both pCR invasive and DCIS cancer was eradicated in patients, and 17.9% of patients had eradication of only residual DCIS. Compared with patients where DCIS was not identified at initial biopsy, DCIS identification was associated with a higher likelihood of residual disease (69% vs. 57%; P = .04). The researchers found patients having hormone receptor–positive/HER2-positive disease was associated with a higher rate of predictive residual disease (26.6%), compared with patients who had hormone receptor–negative/HER2-positive disease (49.2%; odds ratio, 2.7; 95% confidence interval, P less than .0001).
“For the currently occurring trial, evaluating the safety of eliminating surgery for patients who are exceptional responders to neoadjuvant systemic therapy, eradication of both the invasive and DCIS components are necessary because DCIS may serve as a nidus for carcinoma in the future,” said Dr. Sun.
The researchers also studied the effectiveness of multimodality imaging on identifying pathologic response. The multimodality imaging consisted of a mammogram and ultrasound for all patients, and approximately 13% of patients had MRI in addition to mammogram and ultrasound. The multimodality imaging response after neoadjuvant systemic therapy had a sensitivity of 97.1% and a negative predictive value of 70.6% for detecting residual disease in the breast and lymph nodes.
“[O]ur study found that multimodality imaging was not reliable in assessing for pathologic response within the breast or lymph nodes,” said Dr. Sun. “Therefore, imaging alone cannot be used to select patients for no surgery. This requires patients who have image-guided percutaneous biopsy to safely select patients for inclusion and elimination of surgery trial.”
In a discussion session, Dr. Sun clarified the combination of multimodality imaging and image-guided percutaneous biopsy was used to select HER2-positive patients for a clinical trial, and is not standard of practice to determine pCR at the University of Texas MD Anderson Cancer Center.
Dr. Sun reported no relevant financial disclosures.
FROM ASBS 2019
Racial, economic disparities found in buprenorphine prescriptions
Buprenorphine for opioid use disorder is much less likely to be prescribed to patients who are black or who do not have health insurance, an analysis of two national surveys shows.
Researchers analyzed data from the National Ambulatory Medical Care Survey and the National Hospital Ambulatory Medical Care Survey from 2004 to 2015, including 13.4 million visits in which buprenorphine was prescribed. The analysis was published as a research letter in JAMA Psychiatry.
From 2012 to 2015, the number of ambulatory visits involving buprenorphine rose from 0.04% to 0.36%. Black patients were 77% less likely to receive a prescription for buprenorphine at their visit – even after adjustment for payment method, sex, and age – while the number of prescription received by white patients was considerably higher than for patients of any other ethnicity, wrote Pooja A. Lagisetty, MD, and coauthors.
, and the age group with the highest incidence of buprenorphine prescriptions was 30-50 years.
Self-pay and private health insurance were the most common payment methods, but the number of self-paying patients receiving buprenorphine prescriptions dramatically increased from 585,568 in 2004-2007 to 5.3 million in 2012-2015.
“This finding in nationally representative data builds on a previous study that reported buprenorphine treatment disparities on the basis of race/ethnicity and income in New York City,” said Dr. Lagisetty of the department of medicine at the University of Michigan, Ann Arbor, and coauthors.
However, they acknowledged that it was unclear whether the treatment disparity might in fact reflect a difference in the prevalence of opioid use disorder across ethnicities.
Commenting on the differences in payment methods, the authors noted that, despite the enactment of mental health parity legislation and the expansion of Medicaid, the proportion of self-pay visits remained relatively unchanged across the study period.
“A recent study demonstrated that half of the physicians prescribing buprenorphine in Ohio accepted cash alone, and our findings suggest that this practice may be widespread and may be associated with additional financial barriers for low-income populations,” the researchers wrote. “With rising rates of opioid overdoses, it is imperative that policy and research efforts specifically address racial/ethnic and economic differences in treatment access and engagement.”
No conflicts of interest were declared.
SOURCE: Lagisetty P et al. JAMA Psychiatry. 2019 May 8. doi: 10.1001/jamapsychiatry.2019.0876.
Buprenorphine for opioid use disorder is much less likely to be prescribed to patients who are black or who do not have health insurance, an analysis of two national surveys shows.
Researchers analyzed data from the National Ambulatory Medical Care Survey and the National Hospital Ambulatory Medical Care Survey from 2004 to 2015, including 13.4 million visits in which buprenorphine was prescribed. The analysis was published as a research letter in JAMA Psychiatry.
From 2012 to 2015, the number of ambulatory visits involving buprenorphine rose from 0.04% to 0.36%. Black patients were 77% less likely to receive a prescription for buprenorphine at their visit – even after adjustment for payment method, sex, and age – while the number of prescription received by white patients was considerably higher than for patients of any other ethnicity, wrote Pooja A. Lagisetty, MD, and coauthors.
, and the age group with the highest incidence of buprenorphine prescriptions was 30-50 years.
Self-pay and private health insurance were the most common payment methods, but the number of self-paying patients receiving buprenorphine prescriptions dramatically increased from 585,568 in 2004-2007 to 5.3 million in 2012-2015.
“This finding in nationally representative data builds on a previous study that reported buprenorphine treatment disparities on the basis of race/ethnicity and income in New York City,” said Dr. Lagisetty of the department of medicine at the University of Michigan, Ann Arbor, and coauthors.
However, they acknowledged that it was unclear whether the treatment disparity might in fact reflect a difference in the prevalence of opioid use disorder across ethnicities.
Commenting on the differences in payment methods, the authors noted that, despite the enactment of mental health parity legislation and the expansion of Medicaid, the proportion of self-pay visits remained relatively unchanged across the study period.
“A recent study demonstrated that half of the physicians prescribing buprenorphine in Ohio accepted cash alone, and our findings suggest that this practice may be widespread and may be associated with additional financial barriers for low-income populations,” the researchers wrote. “With rising rates of opioid overdoses, it is imperative that policy and research efforts specifically address racial/ethnic and economic differences in treatment access and engagement.”
No conflicts of interest were declared.
SOURCE: Lagisetty P et al. JAMA Psychiatry. 2019 May 8. doi: 10.1001/jamapsychiatry.2019.0876.
Buprenorphine for opioid use disorder is much less likely to be prescribed to patients who are black or who do not have health insurance, an analysis of two national surveys shows.
Researchers analyzed data from the National Ambulatory Medical Care Survey and the National Hospital Ambulatory Medical Care Survey from 2004 to 2015, including 13.4 million visits in which buprenorphine was prescribed. The analysis was published as a research letter in JAMA Psychiatry.
From 2012 to 2015, the number of ambulatory visits involving buprenorphine rose from 0.04% to 0.36%. Black patients were 77% less likely to receive a prescription for buprenorphine at their visit – even after adjustment for payment method, sex, and age – while the number of prescription received by white patients was considerably higher than for patients of any other ethnicity, wrote Pooja A. Lagisetty, MD, and coauthors.
, and the age group with the highest incidence of buprenorphine prescriptions was 30-50 years.
Self-pay and private health insurance were the most common payment methods, but the number of self-paying patients receiving buprenorphine prescriptions dramatically increased from 585,568 in 2004-2007 to 5.3 million in 2012-2015.
“This finding in nationally representative data builds on a previous study that reported buprenorphine treatment disparities on the basis of race/ethnicity and income in New York City,” said Dr. Lagisetty of the department of medicine at the University of Michigan, Ann Arbor, and coauthors.
However, they acknowledged that it was unclear whether the treatment disparity might in fact reflect a difference in the prevalence of opioid use disorder across ethnicities.
Commenting on the differences in payment methods, the authors noted that, despite the enactment of mental health parity legislation and the expansion of Medicaid, the proportion of self-pay visits remained relatively unchanged across the study period.
“A recent study demonstrated that half of the physicians prescribing buprenorphine in Ohio accepted cash alone, and our findings suggest that this practice may be widespread and may be associated with additional financial barriers for low-income populations,” the researchers wrote. “With rising rates of opioid overdoses, it is imperative that policy and research efforts specifically address racial/ethnic and economic differences in treatment access and engagement.”
No conflicts of interest were declared.
SOURCE: Lagisetty P et al. JAMA Psychiatry. 2019 May 8. doi: 10.1001/jamapsychiatry.2019.0876.
FROM JAMA PSYCHIATRY
Researchers propose new risk groups for NK-AML
NEWPORT BEACH, CALIF. – New research suggests patients with normal karyotype acute myeloid leukemia (NK-AML) can be divided into four risk groups associated with overall survival.
Investigators used machine learning algorithms to study the association between mutations and overall survival in 1,352 patients with NK-AML. The analysis revealed combinations of mutations that could be used to classify NK-AML patients into favorable, intermediate-1, intermediate-2, and unfavorable risk groups.
For example, patients who had NPM1 mutations but wild-type FLT3-ITD and DNMT3A, had a median overall survival of 99.1 months and could be classified as favorable risk. Conversely, patients who had NPM1, FLT3-ITD, and DNMT3A mutations, had a median overall survival of 13.4 months and could be classified as unfavorable risk.
Aziz Nazha, MD, of the Cleveland Clinic, and his colleagues conducted this research and presented the findings at the Acute Leukemia Forum of Hemedicus.
The investigators looked at genomic and clinical data from 1,352 patients with NK-AML. The patients were a median age of 55 years and had a median white blood cell count of 21.3 x 109/L, a median hemoglobin of 9.1 g/dL, and a median platelet count of 61 x 109/L. More than half of patients (57.3%) were male.
The patients were screened for 35 genes that are commonly mutated in AML and other myeloid malignancies. The investigators used machine learning algorithms, including random survival forest and recommender system algorithms, to study the association between mutations and overall survival in an “unbiased” way.
Dr. Nazha said there were a median of three mutations per patient sample, and “there are some competing interests between those mutations to impact the prognosis of the patient.”
The investigators used the mutations and their associations with overall survival to classify patients into the risk groups outlined in the table below.
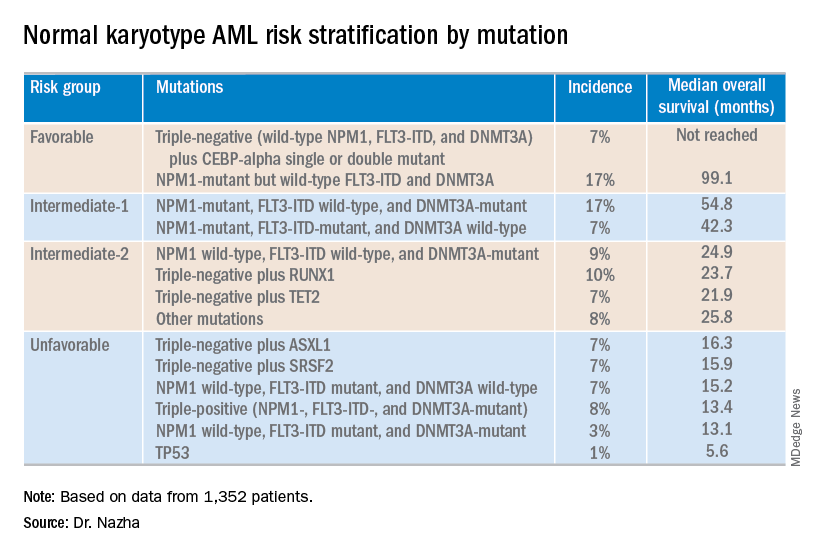
These findings can improve the risk stratification of NK-AML and may aid physicians in making treatment decisions, according to Dr. Nazha and his colleagues. To move this work forward, the investigators are attempting to develop a personalized model that can make predictions specific to an individual patient based on that patient’s mutation information.
Dr. Nazha reported having no financial disclosures relevant to this research. Other investigators reported relationships with the Munich Leukemia Laboratory.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. – New research suggests patients with normal karyotype acute myeloid leukemia (NK-AML) can be divided into four risk groups associated with overall survival.
Investigators used machine learning algorithms to study the association between mutations and overall survival in 1,352 patients with NK-AML. The analysis revealed combinations of mutations that could be used to classify NK-AML patients into favorable, intermediate-1, intermediate-2, and unfavorable risk groups.
For example, patients who had NPM1 mutations but wild-type FLT3-ITD and DNMT3A, had a median overall survival of 99.1 months and could be classified as favorable risk. Conversely, patients who had NPM1, FLT3-ITD, and DNMT3A mutations, had a median overall survival of 13.4 months and could be classified as unfavorable risk.
Aziz Nazha, MD, of the Cleveland Clinic, and his colleagues conducted this research and presented the findings at the Acute Leukemia Forum of Hemedicus.
The investigators looked at genomic and clinical data from 1,352 patients with NK-AML. The patients were a median age of 55 years and had a median white blood cell count of 21.3 x 109/L, a median hemoglobin of 9.1 g/dL, and a median platelet count of 61 x 109/L. More than half of patients (57.3%) were male.
The patients were screened for 35 genes that are commonly mutated in AML and other myeloid malignancies. The investigators used machine learning algorithms, including random survival forest and recommender system algorithms, to study the association between mutations and overall survival in an “unbiased” way.
Dr. Nazha said there were a median of three mutations per patient sample, and “there are some competing interests between those mutations to impact the prognosis of the patient.”
The investigators used the mutations and their associations with overall survival to classify patients into the risk groups outlined in the table below.

These findings can improve the risk stratification of NK-AML and may aid physicians in making treatment decisions, according to Dr. Nazha and his colleagues. To move this work forward, the investigators are attempting to develop a personalized model that can make predictions specific to an individual patient based on that patient’s mutation information.
Dr. Nazha reported having no financial disclosures relevant to this research. Other investigators reported relationships with the Munich Leukemia Laboratory.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
NEWPORT BEACH, CALIF. – New research suggests patients with normal karyotype acute myeloid leukemia (NK-AML) can be divided into four risk groups associated with overall survival.
Investigators used machine learning algorithms to study the association between mutations and overall survival in 1,352 patients with NK-AML. The analysis revealed combinations of mutations that could be used to classify NK-AML patients into favorable, intermediate-1, intermediate-2, and unfavorable risk groups.
For example, patients who had NPM1 mutations but wild-type FLT3-ITD and DNMT3A, had a median overall survival of 99.1 months and could be classified as favorable risk. Conversely, patients who had NPM1, FLT3-ITD, and DNMT3A mutations, had a median overall survival of 13.4 months and could be classified as unfavorable risk.
Aziz Nazha, MD, of the Cleveland Clinic, and his colleagues conducted this research and presented the findings at the Acute Leukemia Forum of Hemedicus.
The investigators looked at genomic and clinical data from 1,352 patients with NK-AML. The patients were a median age of 55 years and had a median white blood cell count of 21.3 x 109/L, a median hemoglobin of 9.1 g/dL, and a median platelet count of 61 x 109/L. More than half of patients (57.3%) were male.
The patients were screened for 35 genes that are commonly mutated in AML and other myeloid malignancies. The investigators used machine learning algorithms, including random survival forest and recommender system algorithms, to study the association between mutations and overall survival in an “unbiased” way.
Dr. Nazha said there were a median of three mutations per patient sample, and “there are some competing interests between those mutations to impact the prognosis of the patient.”
The investigators used the mutations and their associations with overall survival to classify patients into the risk groups outlined in the table below.

These findings can improve the risk stratification of NK-AML and may aid physicians in making treatment decisions, according to Dr. Nazha and his colleagues. To move this work forward, the investigators are attempting to develop a personalized model that can make predictions specific to an individual patient based on that patient’s mutation information.
Dr. Nazha reported having no financial disclosures relevant to this research. Other investigators reported relationships with the Munich Leukemia Laboratory.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
REPORTING FROM ALF 2019
Ruzurgi approved for Lambert-Eaton myasthenic syndrome in patients under age 17
Amifampridine (Ruzurgi) has been approved for the treatment of Lambert-Eaton myasthenic syndrome (LEMS), a rare autoimmune neuromuscular disorder, in patients aged 6 to less than 17 years, according to a statement from the Food and Drug Administration.
The approval is the first for a LEMS treatment specifically for pediatric patients.
“This approval will provide a much-needed treatment option for pediatric patients with LEMS who have significant weakness and fatigue that can often cause great difficulties with daily activities,” Billy Dunn, MD, director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement.
The prevalence of LEMS in pediatric patients is not known, but the overall prevalence of LEMS is estimated to be three per million individuals worldwide, according to the FDA press release.
Use of amifampridine in patients 6 to less than 17 years of age is supported by pharmacokinetic data in adult patients, pharmacokinetic modeling and simulation to identify the dosing regimen in pediatric patients, and safety data from pediatric patients 6 to less than 17 years of age.
A randomized, double-blind, placebo-controlled withdrawal study enrolled 32 adult patients who had taken amifampridine for at least 3 months. The study compared patients continuing on amifampridine with patients switched to placebo. Effectiveness was measured by the degree of change in a test that assessed the time it took the patient to rise from a chair, walk three meters, and return to the chair for three consecutive laps without pause. The patients who continued on amifampridine experienced less impairment compared with those switched to placebo. Effectiveness was also measured with a self-assessment scale for LEMS-related weakness. The scores indicated greater perceived weakening in the patients switched to placebo.
The most common side effects among amifampridine users were paresthesia, abdominal pain, indigestion, dizziness, and nausea. Side effects reported in pediatric patients were similar to those seen in adult patients. Seizures have been observed in patients without a history of seizures. Signs of hypersensitivity reactions include rash, hives, itching, fever, swelling, or trouble breathing.
The FDA granted this application Priority Review and Fast Track designations. Amifampridine also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted the approval of amifampridine (Ruzurgi) to Jacobus Pharmaceutical Company.
Amifampridine (Ruzurgi) has been approved for the treatment of Lambert-Eaton myasthenic syndrome (LEMS), a rare autoimmune neuromuscular disorder, in patients aged 6 to less than 17 years, according to a statement from the Food and Drug Administration.
The approval is the first for a LEMS treatment specifically for pediatric patients.
“This approval will provide a much-needed treatment option for pediatric patients with LEMS who have significant weakness and fatigue that can often cause great difficulties with daily activities,” Billy Dunn, MD, director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement.
The prevalence of LEMS in pediatric patients is not known, but the overall prevalence of LEMS is estimated to be three per million individuals worldwide, according to the FDA press release.
Use of amifampridine in patients 6 to less than 17 years of age is supported by pharmacokinetic data in adult patients, pharmacokinetic modeling and simulation to identify the dosing regimen in pediatric patients, and safety data from pediatric patients 6 to less than 17 years of age.
A randomized, double-blind, placebo-controlled withdrawal study enrolled 32 adult patients who had taken amifampridine for at least 3 months. The study compared patients continuing on amifampridine with patients switched to placebo. Effectiveness was measured by the degree of change in a test that assessed the time it took the patient to rise from a chair, walk three meters, and return to the chair for three consecutive laps without pause. The patients who continued on amifampridine experienced less impairment compared with those switched to placebo. Effectiveness was also measured with a self-assessment scale for LEMS-related weakness. The scores indicated greater perceived weakening in the patients switched to placebo.
The most common side effects among amifampridine users were paresthesia, abdominal pain, indigestion, dizziness, and nausea. Side effects reported in pediatric patients were similar to those seen in adult patients. Seizures have been observed in patients without a history of seizures. Signs of hypersensitivity reactions include rash, hives, itching, fever, swelling, or trouble breathing.
The FDA granted this application Priority Review and Fast Track designations. Amifampridine also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted the approval of amifampridine (Ruzurgi) to Jacobus Pharmaceutical Company.
Amifampridine (Ruzurgi) has been approved for the treatment of Lambert-Eaton myasthenic syndrome (LEMS), a rare autoimmune neuromuscular disorder, in patients aged 6 to less than 17 years, according to a statement from the Food and Drug Administration.
The approval is the first for a LEMS treatment specifically for pediatric patients.
“This approval will provide a much-needed treatment option for pediatric patients with LEMS who have significant weakness and fatigue that can often cause great difficulties with daily activities,” Billy Dunn, MD, director of the Division of Neurology Products in the FDA’s Center for Drug Evaluation and Research, said in the statement.
The prevalence of LEMS in pediatric patients is not known, but the overall prevalence of LEMS is estimated to be three per million individuals worldwide, according to the FDA press release.
Use of amifampridine in patients 6 to less than 17 years of age is supported by pharmacokinetic data in adult patients, pharmacokinetic modeling and simulation to identify the dosing regimen in pediatric patients, and safety data from pediatric patients 6 to less than 17 years of age.
A randomized, double-blind, placebo-controlled withdrawal study enrolled 32 adult patients who had taken amifampridine for at least 3 months. The study compared patients continuing on amifampridine with patients switched to placebo. Effectiveness was measured by the degree of change in a test that assessed the time it took the patient to rise from a chair, walk three meters, and return to the chair for three consecutive laps without pause. The patients who continued on amifampridine experienced less impairment compared with those switched to placebo. Effectiveness was also measured with a self-assessment scale for LEMS-related weakness. The scores indicated greater perceived weakening in the patients switched to placebo.
The most common side effects among amifampridine users were paresthesia, abdominal pain, indigestion, dizziness, and nausea. Side effects reported in pediatric patients were similar to those seen in adult patients. Seizures have been observed in patients without a history of seizures. Signs of hypersensitivity reactions include rash, hives, itching, fever, swelling, or trouble breathing.
The FDA granted this application Priority Review and Fast Track designations. Amifampridine also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases.
The FDA granted the approval of amifampridine (Ruzurgi) to Jacobus Pharmaceutical Company.
Oropharynx cancer burden is shifting to older men
The burden of the U.S. oropharynx cancer epidemic is shifting toward older adults, finds a population-based cohort study spanning 23 years. As a result, the disease will likely become one predominantly affecting elderly white men over the next decade. In addition, projections suggest that the annual number of new cases will likely rise exponentially.
Historically, the incidence of oropharynx cancer rose rapidly among white men aged younger than 60 years because of human papillomavirus (HPV) infections (J Clin Oncol. 2011;29:4294-301). But given factors such as aging, possible changes in behavior, and vaccination, the current trajectory is unknown.
Investigators led by Joseph E. Tota, PhD, of the division of cancer epidemiology and genetics at the National Cancer Institute analyzed Surveillance, Epidemiology, and End Results registry data for 1992-2015 to ascertain whether the increases in oropharynx cancer have continued into recent birth cohorts and to forecast future burden across subgroups.
Results published in the Journal of Clinical Oncology showed that, among white men, oropharynx cancer incidence accelerated among those born during 1927-1939 (by 3.5% per 2-year birth cohort) and during 1939-1955 (by 5.3% per 2-year birth cohort), whereas the pace of increase moderated among those born during 1955-1969 (by 1.7% per 2-year birth cohort).
Given these trends, the investigators forecast that incidence will increase sharply between 2016 and 2029 among older white men aged 65-74 years (from 40.7 to 71.2 per 100,000) and 75-84 years (from 25.7 to 50.1 per 100,000), increase moderately among white men aged 55-64 years (from 40.3 to 52.0 per 100,000), and remain stable among white men aged 45-54 years (at roughly 18 per 100,000).
Taking population growth into account, Dr. Tota and colleagues project a 52% increase in annual number of cases in the United States (from 20,124 to 30,629 between 2016 and 2029), mainly driven by a 127% increase among older adults aged at least 65 years (from 7,976 to 18,072) and a 54% increase among white men (from 14,453 to 22,241). As of 2029, white men older than 65 years will account for approximately 44% of all cases.
“Our results suggest an ebbing of the oropharynx cancer epidemic in younger individuals, exaggeration of the epidemic in older individuals, and a continued exponential increase in the annual number of oropharynx cancers over the next decade,” Dr. Tota and coinvestigators summarized.
The findings have important implications regarding treatment of older patients with oropharynx cancer, they noted. “It is likely that the biology of HPV-positive tumors is similar in younger versus older patients; nonetheless, older patients have poorer survival outcomes because of competing comorbidities, treatment-associated acute and chronic toxicity with chemoradiation, or an inability to receive maximally effective therapies. … Thus, older patients with oropharynx cancer may have different risks and benefits when receiving deintensified regimens than younger patients. The emergence of immunotherapies, whose efficacy may be more age invariant than cytotoxic chemotherapies, could provide a promising treatment avenue for older patients with oropharynx cancer.”
Dr. Tota reported receiving travel, accommodations, and expenses from Merck. The study was supported by the Intramural Research Program of the National Institutes of Health/NCI.
SOURCE: Tota JE et al. J Clin Oncol. 2019 Apr 26. doi: 10.1200/JCO.19.00370.
The burden of the U.S. oropharynx cancer epidemic is shifting toward older adults, finds a population-based cohort study spanning 23 years. As a result, the disease will likely become one predominantly affecting elderly white men over the next decade. In addition, projections suggest that the annual number of new cases will likely rise exponentially.
Historically, the incidence of oropharynx cancer rose rapidly among white men aged younger than 60 years because of human papillomavirus (HPV) infections (J Clin Oncol. 2011;29:4294-301). But given factors such as aging, possible changes in behavior, and vaccination, the current trajectory is unknown.
Investigators led by Joseph E. Tota, PhD, of the division of cancer epidemiology and genetics at the National Cancer Institute analyzed Surveillance, Epidemiology, and End Results registry data for 1992-2015 to ascertain whether the increases in oropharynx cancer have continued into recent birth cohorts and to forecast future burden across subgroups.
Results published in the Journal of Clinical Oncology showed that, among white men, oropharynx cancer incidence accelerated among those born during 1927-1939 (by 3.5% per 2-year birth cohort) and during 1939-1955 (by 5.3% per 2-year birth cohort), whereas the pace of increase moderated among those born during 1955-1969 (by 1.7% per 2-year birth cohort).
Given these trends, the investigators forecast that incidence will increase sharply between 2016 and 2029 among older white men aged 65-74 years (from 40.7 to 71.2 per 100,000) and 75-84 years (from 25.7 to 50.1 per 100,000), increase moderately among white men aged 55-64 years (from 40.3 to 52.0 per 100,000), and remain stable among white men aged 45-54 years (at roughly 18 per 100,000).
Taking population growth into account, Dr. Tota and colleagues project a 52% increase in annual number of cases in the United States (from 20,124 to 30,629 between 2016 and 2029), mainly driven by a 127% increase among older adults aged at least 65 years (from 7,976 to 18,072) and a 54% increase among white men (from 14,453 to 22,241). As of 2029, white men older than 65 years will account for approximately 44% of all cases.
“Our results suggest an ebbing of the oropharynx cancer epidemic in younger individuals, exaggeration of the epidemic in older individuals, and a continued exponential increase in the annual number of oropharynx cancers over the next decade,” Dr. Tota and coinvestigators summarized.
The findings have important implications regarding treatment of older patients with oropharynx cancer, they noted. “It is likely that the biology of HPV-positive tumors is similar in younger versus older patients; nonetheless, older patients have poorer survival outcomes because of competing comorbidities, treatment-associated acute and chronic toxicity with chemoradiation, or an inability to receive maximally effective therapies. … Thus, older patients with oropharynx cancer may have different risks and benefits when receiving deintensified regimens than younger patients. The emergence of immunotherapies, whose efficacy may be more age invariant than cytotoxic chemotherapies, could provide a promising treatment avenue for older patients with oropharynx cancer.”
Dr. Tota reported receiving travel, accommodations, and expenses from Merck. The study was supported by the Intramural Research Program of the National Institutes of Health/NCI.
SOURCE: Tota JE et al. J Clin Oncol. 2019 Apr 26. doi: 10.1200/JCO.19.00370.
The burden of the U.S. oropharynx cancer epidemic is shifting toward older adults, finds a population-based cohort study spanning 23 years. As a result, the disease will likely become one predominantly affecting elderly white men over the next decade. In addition, projections suggest that the annual number of new cases will likely rise exponentially.
Historically, the incidence of oropharynx cancer rose rapidly among white men aged younger than 60 years because of human papillomavirus (HPV) infections (J Clin Oncol. 2011;29:4294-301). But given factors such as aging, possible changes in behavior, and vaccination, the current trajectory is unknown.
Investigators led by Joseph E. Tota, PhD, of the division of cancer epidemiology and genetics at the National Cancer Institute analyzed Surveillance, Epidemiology, and End Results registry data for 1992-2015 to ascertain whether the increases in oropharynx cancer have continued into recent birth cohorts and to forecast future burden across subgroups.
Results published in the Journal of Clinical Oncology showed that, among white men, oropharynx cancer incidence accelerated among those born during 1927-1939 (by 3.5% per 2-year birth cohort) and during 1939-1955 (by 5.3% per 2-year birth cohort), whereas the pace of increase moderated among those born during 1955-1969 (by 1.7% per 2-year birth cohort).
Given these trends, the investigators forecast that incidence will increase sharply between 2016 and 2029 among older white men aged 65-74 years (from 40.7 to 71.2 per 100,000) and 75-84 years (from 25.7 to 50.1 per 100,000), increase moderately among white men aged 55-64 years (from 40.3 to 52.0 per 100,000), and remain stable among white men aged 45-54 years (at roughly 18 per 100,000).
Taking population growth into account, Dr. Tota and colleagues project a 52% increase in annual number of cases in the United States (from 20,124 to 30,629 between 2016 and 2029), mainly driven by a 127% increase among older adults aged at least 65 years (from 7,976 to 18,072) and a 54% increase among white men (from 14,453 to 22,241). As of 2029, white men older than 65 years will account for approximately 44% of all cases.
“Our results suggest an ebbing of the oropharynx cancer epidemic in younger individuals, exaggeration of the epidemic in older individuals, and a continued exponential increase in the annual number of oropharynx cancers over the next decade,” Dr. Tota and coinvestigators summarized.
The findings have important implications regarding treatment of older patients with oropharynx cancer, they noted. “It is likely that the biology of HPV-positive tumors is similar in younger versus older patients; nonetheless, older patients have poorer survival outcomes because of competing comorbidities, treatment-associated acute and chronic toxicity with chemoradiation, or an inability to receive maximally effective therapies. … Thus, older patients with oropharynx cancer may have different risks and benefits when receiving deintensified regimens than younger patients. The emergence of immunotherapies, whose efficacy may be more age invariant than cytotoxic chemotherapies, could provide a promising treatment avenue for older patients with oropharynx cancer.”
Dr. Tota reported receiving travel, accommodations, and expenses from Merck. The study was supported by the Intramural Research Program of the National Institutes of Health/NCI.
SOURCE: Tota JE et al. J Clin Oncol. 2019 Apr 26. doi: 10.1200/JCO.19.00370.
FROM THE JOURNAL OF CLINICAL ONCOLOGY