User login
Rapidly Growing Cutaneous Nodules on the Scalp
The Diagnosis: B-Cell Acute Lymphoblastic Leukemia
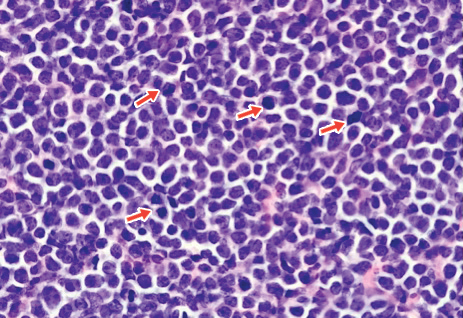
A 4-mm punch biopsy of one of the scalp lesions showed a diffuse infiltrate of intermediately sized cells with variably mature chromatin and irregular nuclear contours, consistent with a neoplastic process. Numerous mitotic figures were present, indicating a high proliferation rate (Figure 1). At that time there was no evidence of systemic involvement. A repeat biopsy with concurrent bone marrow biopsy was scheduled 10 days after the patient's initial presentation for further classification. Laboratory studies at that time revealed leukocytosis with elevated neutrophils and lymphocytes as well as a high absolute blast count.
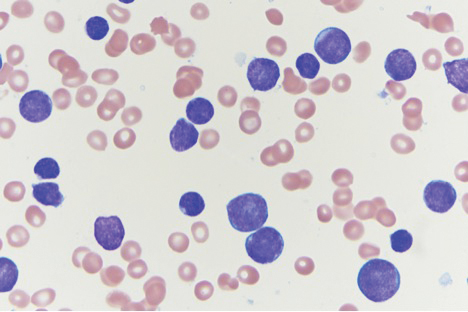
On immunohistochemical staining, the neoplastic cells were positive for CD45, which indicated the neoplasm was hematopoietic, as well as CD10 and the B-cell antigens PAX-5 and CD79a. The cells were negative for CD20, which also is a B-cell marker, but this marker is only expressed in approximately half of pediatric acute lymphoblastic leukemia (ALL) cases with B-cell precursor origin.1 Markers that typically are expressed in B-cell acute lymphoblastic leukemia (B-ALL)--CD34 and terminal deoxynucleotidyl transferase--were both negative. These results were somewhat contradictory, and the differential remained open to both B-ALL and mature B-cell lymphoma. A bone marrow biopsy showed approximately 65% blasts or leukemic cells (Figure 2). Flow cytometry showed the cells were positive for CD10, CD19, weak CD79a, and variable lambda surface antigen expression. The cells were negative for expression of CD20, CD34, terminal deoxynucleotidyl transferase, myeloid antigens, and CD3. Ultimately, the morphology and immunophenotype were most consistent with a diagnosis of B-ALL. Fluorescence in situ hybridization revealed mixed lineage leukemia, MLL, gene rearrangements.
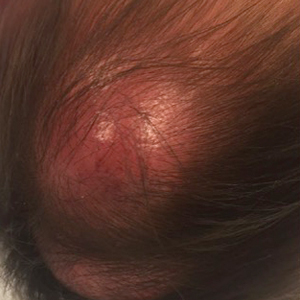
In general, when considering the differential diagnosis of superficial nodules, 5 elements are helpful to consider: the number of nodules (single vs multiple); the location; and the presence or absence of tenderness, pigmentation or erythema, and firmness.2 Our patient had multiple nodules on the scalp, which were erythematous to slightly purple and firm. The differential diagnosis can be categorized into malignant; infectious; and benign inflammatory, vascular, and fibrous tumors.
Potential oncologic processes include leukemia cutis, lymphoblastic leukemia/lymphoma, Langerhans cell histiocytosis, and rhabdomyosarcoma. Initial laboratory test results were reassuring. Infectious processes in the differential include deep fungal infections such as coccidioidomycosis and nontuberculous mycobacterial infections. Coccidioidomycosis was the most likely to cause skin lesions or masses in our patient; however, it was considered less likely because the patient's family had not traveled or been exposed to an endemic area.3
Benign tumors in the differential include deep hemangioma, which was deemed less likely in our patient because most hemangiomas reach 80% of their maximum size by 5 months of age.4 Another possible benign tumor is infantile myofibromatosis, which is rare but is the most common fibrous tumor of infancy.5
Early-onset childhood sarcoidosis also has been shown to produce multiple nontender firm nodules.2 This process was considered unlikely in our patient because not only is the disease relatively rare in the pediatric population, but most reported childhood cases have occurred in patients aged 13 to 15 years.6 Additionally, no uveitis or arthritis was observed in this case.
Ultimately, histopathology and bone marrow biopsy were necessary to determine the diagnosis of B-ALL. Although uncommon, cutaneous involvement can be an early sign of ALL in children.7 Thus, neoplastic etiologies should be considered in the workup of cutaneous nodules in children, especially when these nodules are hard, rapidly growing, ulcerated, fixed, and/or vascular.8 Once the diagnosis is established, initial workup of ALL in children should include complete blood cell count with manual differential, prothrombin time, partial thromboplastin time, electrolytes, uric acid, and renal and liver function tests. Often, baseline viral titers such as cytomegalovirus, Epstein-Barr virus, human immunodeficiency virus, hepatitis B virus, and varicella-zoster virus also are included. Patients are risk stratified to the appropriate level of treatment based on tumor immunophenotype, cytogenetic findings, patient age, white blood cell count at the time of diagnosis, and response to initial therapy. Treatment typically is comprised of a multidrug regimen divided into several phases--induction, consolidation, and maintenance--as well as therapy directed to the central nervous system. Treatment protocols usually take 2 to 3 years to complete.
Our patient was treated with 1 dose of intrathecal methotrexate before starting the Interfant-06 protocol with a 7-day methylprednisolone prophase. The patient's nodules shrank over time and were no longer present after 14 days of treatment.
- Dworzak MN, Schumich A, Printz D, et al. CD20 up-regulation in pediatric B-cell precursor acute lymphoblastic leukemia during induction treatment: setting the stage for anti-CD20 directed immunotherapy. Blood. 2008;112:3982-3988.
- Whelan JP, Zembowicz A. Case records of the Massachusetts General Hospital. case 19-2006. a 22-month-old boy with the rapid growth of subcutaneous nodules. N Engl J Med. 2006;354:2697-2704.
- Malo J, Luraschi-Monjagatta C, Wolk DM, et al. Update on the diagnosis of pulmonary coccidioidomycosis. Ann Am Thorac Soc. 2014;11:243-253.
- Chang LC, Haggstrom AN, Drolet BA, et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics. 2008;122:360-367.
- Schurr P, Moulsdale W. Infantile myofibroma. Adv Neonatal Care. 2008;8:13-20.
- Shetty AK, Gedalia A. Childhood sarcoidosis: a rare but fascinating disorder. Pediatr Rheumatol Online J. 2008;6:16.
- Millot F, Robert A, Bertrand Y, et al. Cutaneous involvement in children with acute lymphoblastic leukemia or lymphoblastic lymphoma. The Children's Leukemia Cooperative Group of the European Organization of Research and Treatment of Cancer (EORTC). Pediatrics. 1997;100:60-64.
- Fogelson S, Dohil M. Papular and nodular skin lesions in children. Semin Plast Surg. 2006;20:180-191.
The Diagnosis: B-Cell Acute Lymphoblastic Leukemia
A 4-mm punch biopsy of one of the scalp lesions showed a diffuse infiltrate of intermediately sized cells with variably mature chromatin and irregular nuclear contours, consistent with a neoplastic process. Numerous mitotic figures were present, indicating a high proliferation rate (Figure 1). At that time there was no evidence of systemic involvement. A repeat biopsy with concurrent bone marrow biopsy was scheduled 10 days after the patient's initial presentation for further classification. Laboratory studies at that time revealed leukocytosis with elevated neutrophils and lymphocytes as well as a high absolute blast count.

On immunohistochemical staining, the neoplastic cells were positive for CD45, which indicated the neoplasm was hematopoietic, as well as CD10 and the B-cell antigens PAX-5 and CD79a. The cells were negative for CD20, which also is a B-cell marker, but this marker is only expressed in approximately half of pediatric acute lymphoblastic leukemia (ALL) cases with B-cell precursor origin.1 Markers that typically are expressed in B-cell acute lymphoblastic leukemia (B-ALL)--CD34 and terminal deoxynucleotidyl transferase--were both negative. These results were somewhat contradictory, and the differential remained open to both B-ALL and mature B-cell lymphoma. A bone marrow biopsy showed approximately 65% blasts or leukemic cells (Figure 2). Flow cytometry showed the cells were positive for CD10, CD19, weak CD79a, and variable lambda surface antigen expression. The cells were negative for expression of CD20, CD34, terminal deoxynucleotidyl transferase, myeloid antigens, and CD3. Ultimately, the morphology and immunophenotype were most consistent with a diagnosis of B-ALL. Fluorescence in situ hybridization revealed mixed lineage leukemia, MLL, gene rearrangements.

In general, when considering the differential diagnosis of superficial nodules, 5 elements are helpful to consider: the number of nodules (single vs multiple); the location; and the presence or absence of tenderness, pigmentation or erythema, and firmness.2 Our patient had multiple nodules on the scalp, which were erythematous to slightly purple and firm. The differential diagnosis can be categorized into malignant; infectious; and benign inflammatory, vascular, and fibrous tumors.
Potential oncologic processes include leukemia cutis, lymphoblastic leukemia/lymphoma, Langerhans cell histiocytosis, and rhabdomyosarcoma. Initial laboratory test results were reassuring. Infectious processes in the differential include deep fungal infections such as coccidioidomycosis and nontuberculous mycobacterial infections. Coccidioidomycosis was the most likely to cause skin lesions or masses in our patient; however, it was considered less likely because the patient's family had not traveled or been exposed to an endemic area.3
Benign tumors in the differential include deep hemangioma, which was deemed less likely in our patient because most hemangiomas reach 80% of their maximum size by 5 months of age.4 Another possible benign tumor is infantile myofibromatosis, which is rare but is the most common fibrous tumor of infancy.5
Early-onset childhood sarcoidosis also has been shown to produce multiple nontender firm nodules.2 This process was considered unlikely in our patient because not only is the disease relatively rare in the pediatric population, but most reported childhood cases have occurred in patients aged 13 to 15 years.6 Additionally, no uveitis or arthritis was observed in this case.
Ultimately, histopathology and bone marrow biopsy were necessary to determine the diagnosis of B-ALL. Although uncommon, cutaneous involvement can be an early sign of ALL in children.7 Thus, neoplastic etiologies should be considered in the workup of cutaneous nodules in children, especially when these nodules are hard, rapidly growing, ulcerated, fixed, and/or vascular.8 Once the diagnosis is established, initial workup of ALL in children should include complete blood cell count with manual differential, prothrombin time, partial thromboplastin time, electrolytes, uric acid, and renal and liver function tests. Often, baseline viral titers such as cytomegalovirus, Epstein-Barr virus, human immunodeficiency virus, hepatitis B virus, and varicella-zoster virus also are included. Patients are risk stratified to the appropriate level of treatment based on tumor immunophenotype, cytogenetic findings, patient age, white blood cell count at the time of diagnosis, and response to initial therapy. Treatment typically is comprised of a multidrug regimen divided into several phases--induction, consolidation, and maintenance--as well as therapy directed to the central nervous system. Treatment protocols usually take 2 to 3 years to complete.
Our patient was treated with 1 dose of intrathecal methotrexate before starting the Interfant-06 protocol with a 7-day methylprednisolone prophase. The patient's nodules shrank over time and were no longer present after 14 days of treatment.
The Diagnosis: B-Cell Acute Lymphoblastic Leukemia
A 4-mm punch biopsy of one of the scalp lesions showed a diffuse infiltrate of intermediately sized cells with variably mature chromatin and irregular nuclear contours, consistent with a neoplastic process. Numerous mitotic figures were present, indicating a high proliferation rate (Figure 1). At that time there was no evidence of systemic involvement. A repeat biopsy with concurrent bone marrow biopsy was scheduled 10 days after the patient's initial presentation for further classification. Laboratory studies at that time revealed leukocytosis with elevated neutrophils and lymphocytes as well as a high absolute blast count.

On immunohistochemical staining, the neoplastic cells were positive for CD45, which indicated the neoplasm was hematopoietic, as well as CD10 and the B-cell antigens PAX-5 and CD79a. The cells were negative for CD20, which also is a B-cell marker, but this marker is only expressed in approximately half of pediatric acute lymphoblastic leukemia (ALL) cases with B-cell precursor origin.1 Markers that typically are expressed in B-cell acute lymphoblastic leukemia (B-ALL)--CD34 and terminal deoxynucleotidyl transferase--were both negative. These results were somewhat contradictory, and the differential remained open to both B-ALL and mature B-cell lymphoma. A bone marrow biopsy showed approximately 65% blasts or leukemic cells (Figure 2). Flow cytometry showed the cells were positive for CD10, CD19, weak CD79a, and variable lambda surface antigen expression. The cells were negative for expression of CD20, CD34, terminal deoxynucleotidyl transferase, myeloid antigens, and CD3. Ultimately, the morphology and immunophenotype were most consistent with a diagnosis of B-ALL. Fluorescence in situ hybridization revealed mixed lineage leukemia, MLL, gene rearrangements.

In general, when considering the differential diagnosis of superficial nodules, 5 elements are helpful to consider: the number of nodules (single vs multiple); the location; and the presence or absence of tenderness, pigmentation or erythema, and firmness.2 Our patient had multiple nodules on the scalp, which were erythematous to slightly purple and firm. The differential diagnosis can be categorized into malignant; infectious; and benign inflammatory, vascular, and fibrous tumors.
Potential oncologic processes include leukemia cutis, lymphoblastic leukemia/lymphoma, Langerhans cell histiocytosis, and rhabdomyosarcoma. Initial laboratory test results were reassuring. Infectious processes in the differential include deep fungal infections such as coccidioidomycosis and nontuberculous mycobacterial infections. Coccidioidomycosis was the most likely to cause skin lesions or masses in our patient; however, it was considered less likely because the patient's family had not traveled or been exposed to an endemic area.3
Benign tumors in the differential include deep hemangioma, which was deemed less likely in our patient because most hemangiomas reach 80% of their maximum size by 5 months of age.4 Another possible benign tumor is infantile myofibromatosis, which is rare but is the most common fibrous tumor of infancy.5
Early-onset childhood sarcoidosis also has been shown to produce multiple nontender firm nodules.2 This process was considered unlikely in our patient because not only is the disease relatively rare in the pediatric population, but most reported childhood cases have occurred in patients aged 13 to 15 years.6 Additionally, no uveitis or arthritis was observed in this case.
Ultimately, histopathology and bone marrow biopsy were necessary to determine the diagnosis of B-ALL. Although uncommon, cutaneous involvement can be an early sign of ALL in children.7 Thus, neoplastic etiologies should be considered in the workup of cutaneous nodules in children, especially when these nodules are hard, rapidly growing, ulcerated, fixed, and/or vascular.8 Once the diagnosis is established, initial workup of ALL in children should include complete blood cell count with manual differential, prothrombin time, partial thromboplastin time, electrolytes, uric acid, and renal and liver function tests. Often, baseline viral titers such as cytomegalovirus, Epstein-Barr virus, human immunodeficiency virus, hepatitis B virus, and varicella-zoster virus also are included. Patients are risk stratified to the appropriate level of treatment based on tumor immunophenotype, cytogenetic findings, patient age, white blood cell count at the time of diagnosis, and response to initial therapy. Treatment typically is comprised of a multidrug regimen divided into several phases--induction, consolidation, and maintenance--as well as therapy directed to the central nervous system. Treatment protocols usually take 2 to 3 years to complete.
Our patient was treated with 1 dose of intrathecal methotrexate before starting the Interfant-06 protocol with a 7-day methylprednisolone prophase. The patient's nodules shrank over time and were no longer present after 14 days of treatment.
- Dworzak MN, Schumich A, Printz D, et al. CD20 up-regulation in pediatric B-cell precursor acute lymphoblastic leukemia during induction treatment: setting the stage for anti-CD20 directed immunotherapy. Blood. 2008;112:3982-3988.
- Whelan JP, Zembowicz A. Case records of the Massachusetts General Hospital. case 19-2006. a 22-month-old boy with the rapid growth of subcutaneous nodules. N Engl J Med. 2006;354:2697-2704.
- Malo J, Luraschi-Monjagatta C, Wolk DM, et al. Update on the diagnosis of pulmonary coccidioidomycosis. Ann Am Thorac Soc. 2014;11:243-253.
- Chang LC, Haggstrom AN, Drolet BA, et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics. 2008;122:360-367.
- Schurr P, Moulsdale W. Infantile myofibroma. Adv Neonatal Care. 2008;8:13-20.
- Shetty AK, Gedalia A. Childhood sarcoidosis: a rare but fascinating disorder. Pediatr Rheumatol Online J. 2008;6:16.
- Millot F, Robert A, Bertrand Y, et al. Cutaneous involvement in children with acute lymphoblastic leukemia or lymphoblastic lymphoma. The Children's Leukemia Cooperative Group of the European Organization of Research and Treatment of Cancer (EORTC). Pediatrics. 1997;100:60-64.
- Fogelson S, Dohil M. Papular and nodular skin lesions in children. Semin Plast Surg. 2006;20:180-191.
- Dworzak MN, Schumich A, Printz D, et al. CD20 up-regulation in pediatric B-cell precursor acute lymphoblastic leukemia during induction treatment: setting the stage for anti-CD20 directed immunotherapy. Blood. 2008;112:3982-3988.
- Whelan JP, Zembowicz A. Case records of the Massachusetts General Hospital. case 19-2006. a 22-month-old boy with the rapid growth of subcutaneous nodules. N Engl J Med. 2006;354:2697-2704.
- Malo J, Luraschi-Monjagatta C, Wolk DM, et al. Update on the diagnosis of pulmonary coccidioidomycosis. Ann Am Thorac Soc. 2014;11:243-253.
- Chang LC, Haggstrom AN, Drolet BA, et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics. 2008;122:360-367.
- Schurr P, Moulsdale W. Infantile myofibroma. Adv Neonatal Care. 2008;8:13-20.
- Shetty AK, Gedalia A. Childhood sarcoidosis: a rare but fascinating disorder. Pediatr Rheumatol Online J. 2008;6:16.
- Millot F, Robert A, Bertrand Y, et al. Cutaneous involvement in children with acute lymphoblastic leukemia or lymphoblastic lymphoma. The Children's Leukemia Cooperative Group of the European Organization of Research and Treatment of Cancer (EORTC). Pediatrics. 1997;100:60-64.
- Fogelson S, Dohil M. Papular and nodular skin lesions in children. Semin Plast Surg. 2006;20:180-191.
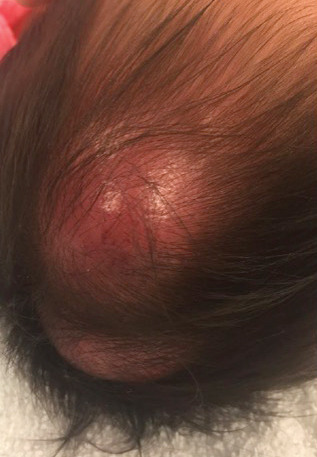
An 8-month-old infant girl presented with rapidly growing cutaneous nodules on the scalp of 1 month's duration. Her parents reported that she disliked lying flat but was otherwise growing and developing normally. Nondiagnostic ultrasonography of the head and brain had been performed as well as a skull radiograph, which found no evidence of lytic lesions. On physical examination, 3 erythematous to violaceous, subcutaneous, firm, fixed nodules were observed on the scalp. Notable cervical lymphadenopathy with several distinct, fixed, firm, subcutaneous nodules in the postauricular lymph chains also were noted. The patient had no pertinent medical history and was born via normal spontaneous vaginal delivery to healthy parents. The remainder of the physical examination and review of systems was negative.
Liraglutide indication expanded to include children
according to a news release from the agency. The approval comes after granting the application Priority Review, which means the FDA aimed to take action on it within 6 months instead of the usual 10 months seen with a Standard Review.
Liraglutide injection has been approved for treatment of adults with type 2 diabetes since 2010, but this is the first noninsulin treatment for pediatric patients since metformin received approval for this population in 2000.
“The expanded indication provides an additional treatment option at a time when an increasing number of children are being diagnosed with [type 2 diabetes],” said Lisa Yanoff, MD, acting director of the division of metabolism and endocrinology products in the agency’s Center for Drug Evaluation and Research.
The approval is based on efficacy and safety results from several placebo-controlled trials in adults, and one trial in 134 pediatric patients aged 10 years or older. In the latter trial, which ran for more than 26 weeks, hemoglobin A1c levels fell below 7% in approximately 64% of patients treated with liraglutide injection, compared with 37% of those who received placebo. Those results were seen regardless of whether patients concomitantly used insulin.
Although an increase in hypoglycemia risk has sometimes been seen in adult patients taking liraglutide injection with insulin or other drugs that raise insulin production, this heightened risk was seen in the pediatric patients regardless of whether they took other therapies for diabetes.
Liraglutide injection carries a Boxed Warning, the FDA’s strongest warning, for heightened risk of thyroid C-cell tumors. Therefore, the agency recommends against patients with history or family members with history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 using this treatment. Among its other warnings are those pertaining to patients with renal impairment or kidney failure, pancreatitis, and/or acute gallbladder disease.
The most common side effects are nausea, diarrhea, vomiting, decreased appetite, indigestion, and constipation. The full prescribing information can be found on the FDA website.
according to a news release from the agency. The approval comes after granting the application Priority Review, which means the FDA aimed to take action on it within 6 months instead of the usual 10 months seen with a Standard Review.
Liraglutide injection has been approved for treatment of adults with type 2 diabetes since 2010, but this is the first noninsulin treatment for pediatric patients since metformin received approval for this population in 2000.
“The expanded indication provides an additional treatment option at a time when an increasing number of children are being diagnosed with [type 2 diabetes],” said Lisa Yanoff, MD, acting director of the division of metabolism and endocrinology products in the agency’s Center for Drug Evaluation and Research.
The approval is based on efficacy and safety results from several placebo-controlled trials in adults, and one trial in 134 pediatric patients aged 10 years or older. In the latter trial, which ran for more than 26 weeks, hemoglobin A1c levels fell below 7% in approximately 64% of patients treated with liraglutide injection, compared with 37% of those who received placebo. Those results were seen regardless of whether patients concomitantly used insulin.
Although an increase in hypoglycemia risk has sometimes been seen in adult patients taking liraglutide injection with insulin or other drugs that raise insulin production, this heightened risk was seen in the pediatric patients regardless of whether they took other therapies for diabetes.
Liraglutide injection carries a Boxed Warning, the FDA’s strongest warning, for heightened risk of thyroid C-cell tumors. Therefore, the agency recommends against patients with history or family members with history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 using this treatment. Among its other warnings are those pertaining to patients with renal impairment or kidney failure, pancreatitis, and/or acute gallbladder disease.
The most common side effects are nausea, diarrhea, vomiting, decreased appetite, indigestion, and constipation. The full prescribing information can be found on the FDA website.
according to a news release from the agency. The approval comes after granting the application Priority Review, which means the FDA aimed to take action on it within 6 months instead of the usual 10 months seen with a Standard Review.
Liraglutide injection has been approved for treatment of adults with type 2 diabetes since 2010, but this is the first noninsulin treatment for pediatric patients since metformin received approval for this population in 2000.
“The expanded indication provides an additional treatment option at a time when an increasing number of children are being diagnosed with [type 2 diabetes],” said Lisa Yanoff, MD, acting director of the division of metabolism and endocrinology products in the agency’s Center for Drug Evaluation and Research.
The approval is based on efficacy and safety results from several placebo-controlled trials in adults, and one trial in 134 pediatric patients aged 10 years or older. In the latter trial, which ran for more than 26 weeks, hemoglobin A1c levels fell below 7% in approximately 64% of patients treated with liraglutide injection, compared with 37% of those who received placebo. Those results were seen regardless of whether patients concomitantly used insulin.
Although an increase in hypoglycemia risk has sometimes been seen in adult patients taking liraglutide injection with insulin or other drugs that raise insulin production, this heightened risk was seen in the pediatric patients regardless of whether they took other therapies for diabetes.
Liraglutide injection carries a Boxed Warning, the FDA’s strongest warning, for heightened risk of thyroid C-cell tumors. Therefore, the agency recommends against patients with history or family members with history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 using this treatment. Among its other warnings are those pertaining to patients with renal impairment or kidney failure, pancreatitis, and/or acute gallbladder disease.
The most common side effects are nausea, diarrhea, vomiting, decreased appetite, indigestion, and constipation. The full prescribing information can be found on the FDA website.
These Hips Don’t Lie
ANSWER
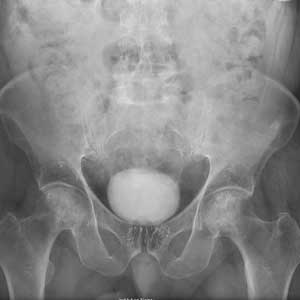
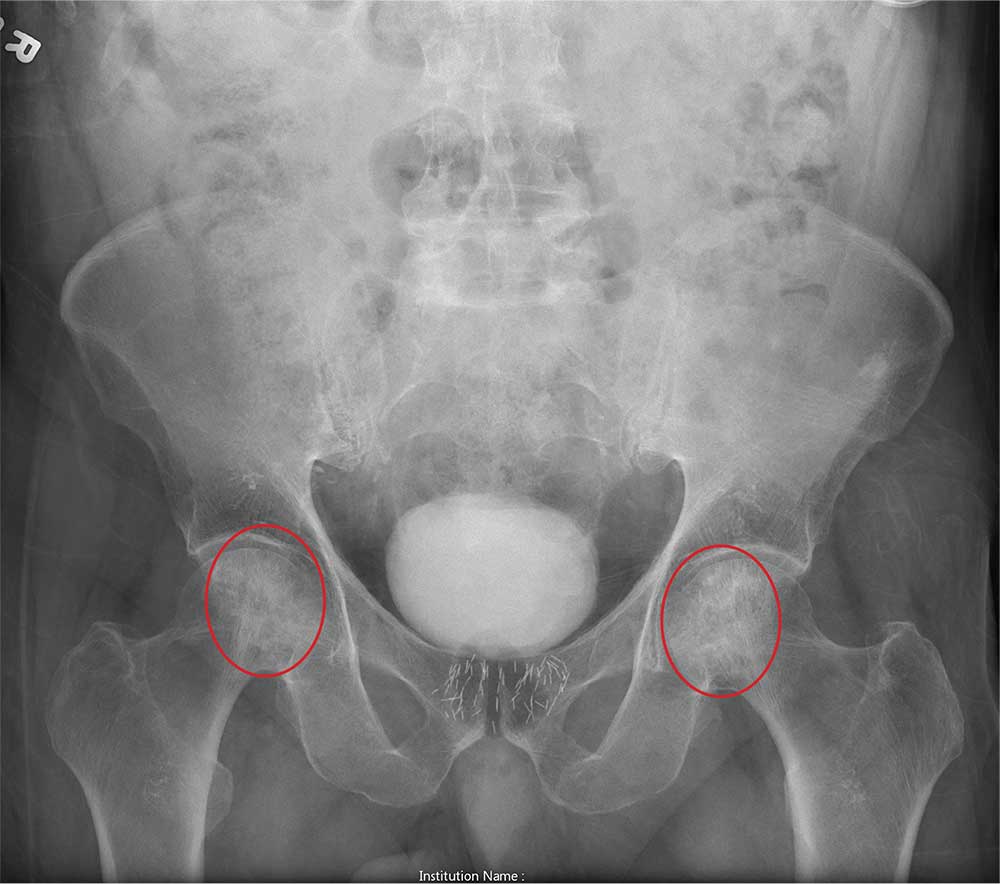
The radiograph shows no evidence of an acute fracture. Incidental findings include excreted contrast within the bladder and radiopaque markers from prostatic seed implants.
Fairly extensive sclerosis is noted within both femoral heads, which is suggestive of osteonecrosis (also known as avascular necrosis). Orthopedic consult was requested for further workup of this specific problem.

ANSWER
The radiograph shows no evidence of an acute fracture. Incidental findings include excreted contrast within the bladder and radiopaque markers from prostatic seed implants.
Fairly extensive sclerosis is noted within both femoral heads, which is suggestive of osteonecrosis (also known as avascular necrosis). Orthopedic consult was requested for further workup of this specific problem.

ANSWER
The radiograph shows no evidence of an acute fracture. Incidental findings include excreted contrast within the bladder and radiopaque markers from prostatic seed implants.
Fairly extensive sclerosis is noted within both femoral heads, which is suggestive of osteonecrosis (also known as avascular necrosis). Orthopedic consult was requested for further workup of this specific problem.
An 80-year-old man is transferred to your facility for evaluation of a lumbar compression fracture he sustained from a motor vehicle collision. The patient was a restrained driver in a vehicle that was broadsided at an unknown speed. His airbags deployed. In addition to mild back discomfort, he complains of severe right hip pain.
His medical history is significant for prostate cancer and coronary artery disease. Surgical history includes remote cardiac bypass surgery and recent revascularization with stents.
On examination, you note an elderly male who is awake and alert. His vital signs are stable. He is able to move all extremities
A portable pelvis radiograph is obtained (shown). What is your impression?
Study finds differences for HCC in women
SAN DIEGO – Hepatocellular carcinoma is the third leading cause of cancer-related death in the United States and its incidence is increasing worldwide. While it affects men much more frequently than women, approximately 4 to 1, the differences in risk factors between men and women have never been studied.
At the annual Digestive Disease Week, Meaghan Phipps, MD, of New York–Presbyterian Hospital, described in a video interview how she and her colleagues set up a retrospective study of these differences in 5,327 patients at five large academic centers around the country. She and her colleagues found that women tended to present later, and with less severe disease, which was more likely to be treated with resection than transplantation. Women had better overall survival. Women were significantly more likely to present without cirrhosis and with nonalcoholic fatty liver disease than were men. Dr. Phipps noted that they did not characterize the women in their study by menopausal status, and suggested that this would be an important thing to look at in a future prospective study because it has long been thought that estrogen confers some protection against hepatocellular carcinoma.
SAN DIEGO – Hepatocellular carcinoma is the third leading cause of cancer-related death in the United States and its incidence is increasing worldwide. While it affects men much more frequently than women, approximately 4 to 1, the differences in risk factors between men and women have never been studied.
At the annual Digestive Disease Week, Meaghan Phipps, MD, of New York–Presbyterian Hospital, described in a video interview how she and her colleagues set up a retrospective study of these differences in 5,327 patients at five large academic centers around the country. She and her colleagues found that women tended to present later, and with less severe disease, which was more likely to be treated with resection than transplantation. Women had better overall survival. Women were significantly more likely to present without cirrhosis and with nonalcoholic fatty liver disease than were men. Dr. Phipps noted that they did not characterize the women in their study by menopausal status, and suggested that this would be an important thing to look at in a future prospective study because it has long been thought that estrogen confers some protection against hepatocellular carcinoma.
SAN DIEGO – Hepatocellular carcinoma is the third leading cause of cancer-related death in the United States and its incidence is increasing worldwide. While it affects men much more frequently than women, approximately 4 to 1, the differences in risk factors between men and women have never been studied.
At the annual Digestive Disease Week, Meaghan Phipps, MD, of New York–Presbyterian Hospital, described in a video interview how she and her colleagues set up a retrospective study of these differences in 5,327 patients at five large academic centers around the country. She and her colleagues found that women tended to present later, and with less severe disease, which was more likely to be treated with resection than transplantation. Women had better overall survival. Women were significantly more likely to present without cirrhosis and with nonalcoholic fatty liver disease than were men. Dr. Phipps noted that they did not characterize the women in their study by menopausal status, and suggested that this would be an important thing to look at in a future prospective study because it has long been thought that estrogen confers some protection against hepatocellular carcinoma.
REPORTING FROM DDW 2019
CSF neurofilament light level could aid in diagnosis
according to an analysis published online ahead of print June 17 in JAMA Neurology. The biomarker has the potential to distinguish between frontotemporal dementia (FTD) and other dementia subtypes, as well as between Parkinson’s disease and atypical parkinsonian syndromes, said the investigators. It may be necessary to identify age- and sex-specific reference values for NfL, they added.
Neurologists have long understood CSF levels of NfL to be elevated in neurodegenerative conditions, but researchers previously had not compared these levels systematically among neurologic disorders. Similarly, the literature indicates a positive association between CSF NfL level and age in healthy controls, but this association has not been evaluated systematically in neurologic disorders. The resulting lack of clarity has impeded the use of NfL as a diagnostic biomarker.
A meta-analysis of CSF samples
Claire Bridel, MD, PhD, of the department of clinical chemistry at the VU University Medical Centre in Amsterdam and colleagues conducted a systematic review and meta-analysis to compare CSF levels of NfL among diagnoses, assess the associations of age and sex with NfL, and evaluate the potential of NfL as a diagnostic biomarker. The investigators searched PubMed for studies published between Jan. 1, 2006, and Jan. 1, 2016, that reported CSF levels of NfL in neurologic or psychiatric conditions or in healthy controls. They included only studies that used the same commercially available immunoassay that has been used in most studies since 2006. The literature indicates that this enzyme-linked immunosorbent assay is sensitive and robust. Dr. Bridel and colleagues contacted study authors and requested their individual-level data.
The investigators sorted the most common neurologic conditions into three groups of similar disorders. The first group included inflammatory conditions of the CNS, such as multiple sclerosis, clinically isolated syndrome (CIS), and optic neuritis. The second group included dementia syndromes (such as Alzheimer’s disease, FTD, vascular dementia, and dementia with Lewy bodies) and amyotrophic lateral sclerosis (ALS). The third category included parkinsonian syndromes such as Parkinson’s disease, Parkinson’s disease dementia, multiple system atrophy (MSA), progressive supranuclear palsy (PSP), and corticobasal syndrome (CBS). The authors used generalized linear mixed-effects models to estimate the fixed effects of age, sex, and diagnosis on log-transformed NfL levels. They modeled cohort of origin as a random intercept.
NfL increased with age
Dr. Bridel and colleagues identified 153 relevant investigations, of which 44 met their inclusion criteria. The original investigators provided data sets for these studies, along with three previously unpublished data sets. The data sets included information from 10,059 participants (mean age, 59.7 years; 54.1% female). After excluding diagnostic categories with fewer than five observations per sex, Dr. Bridel and colleagues included data for 10,012 people in the analysis. In this population, the researchers identified 2,795 patients with inflammatory diseases of the CNS, 4,284 patients with dementia or predementia, 984 patients with parkinsonian disorders, and 1,332 healthy controls.
CSF level of NfL was elevated in most neurologic conditions, compared with healthy controls. The largest effect sizes were in cognitively impaired patients with HIV (21.36), patients with FTD/ALS (10.48), patients with ALS (7.58), and patients with Huntington’s disease (5.88).
In healthy controls, the level of NfL in CSF increased by 3.30% annually. The investigators also observed an association between age and CSF NfL level in people with subjective complaints, bipolar disorder, and most neurodegenerative conditions. They found no association, however, in patients with MS, HIV and cognitive impairment, and rapidly progressive neurodegenerative conditions (such as FTD, ALS, FTD/ALS, MSA, PSP, CBS, and Huntington’s disease). CSF level of NfL was 26.0% higher in men among healthy controls. This discrepancy also was observed in a minority of neurologic conditions, including MS, Alzheimer’s disease, vascular dementia, and Parkinson’s disease.
Mean CSF levels of NfL were similar between patients with inflammatory conditions of the CNS. Among dementias and related disorders, mean CNS level of NfL was significantly higher in FTD than in Alzheimer’s disease (2.08), vascular dementia (1.56), and dementia with Lewy bodies (2.50). Among parkinsonian syndromes, the mean CSF levels of NfL were higher in MSA, PSP, and CBS, compared with Parkinson’s disease.
Many factors influence NfL level in CSF
The association between CNS level of NfL with age among healthy controls “implies that age-specific reference values may be needed and that the diagnostic potential of CSF NfL may decrease with age,” said the researchers. The finding that CSF NfL level was higher in men in a minority of diagnoses has uncertain clinical significance, they added. Sex-specific reference values may be needed.
Dr. Bridel and colleagues found that age, sex, and cohort explained 46% of variation in CSF level of NfL, which suggests that many factors that determine this level have yet to be identified. Disease duration and disease severity could influence the CSF level of NfL, but the data sets that the investigators analyzed did not include this information.
Because CSF NfL level did not differ significantly between relapsing/remitting MS, secondary progressive MS, and primary progressive MS, this biomarker “may not differentiate acute inflammation-induced neuronal damage in the context of relapses from progressive neurodegeneration if the consequences of recent relapses or novel lesion formation are not considered,” said Dr. Bridel and colleagues. The findings do suggest, however, that CSF level of NfL can distinguish FTD from other dementias, as well as Parkinson’s disease from atypical parkinsonian syndromes. Furthermore, it is possible that the findings of this study can be translated to serum level of NfL, said the authors.
One of the study’s limitations was that diagnosis was based on clinical criteria, said Dr. Bridel and colleagues. In addition, the authors were unable to identify dementia of multifactorial origin, which might have reduced the differences in CSF NfL level distributions between dementia subtypes. Finally, the authors only analyzed studies that relied on a specific immunoassay for CSF NfL level.
The authors reported receiving funding from various pharmaceutical and biopharmaceutical companies, as well as from grants and research foundations. The funders did not influence the study design, data analysis, or interpretation, however.
SOURCE: Bridel C et al. JAMA Neurol. 2019 June 17. doi: 10.1001/jamaneurol.2019.1534.
according to an analysis published online ahead of print June 17 in JAMA Neurology. The biomarker has the potential to distinguish between frontotemporal dementia (FTD) and other dementia subtypes, as well as between Parkinson’s disease and atypical parkinsonian syndromes, said the investigators. It may be necessary to identify age- and sex-specific reference values for NfL, they added.
Neurologists have long understood CSF levels of NfL to be elevated in neurodegenerative conditions, but researchers previously had not compared these levels systematically among neurologic disorders. Similarly, the literature indicates a positive association between CSF NfL level and age in healthy controls, but this association has not been evaluated systematically in neurologic disorders. The resulting lack of clarity has impeded the use of NfL as a diagnostic biomarker.
A meta-analysis of CSF samples
Claire Bridel, MD, PhD, of the department of clinical chemistry at the VU University Medical Centre in Amsterdam and colleagues conducted a systematic review and meta-analysis to compare CSF levels of NfL among diagnoses, assess the associations of age and sex with NfL, and evaluate the potential of NfL as a diagnostic biomarker. The investigators searched PubMed for studies published between Jan. 1, 2006, and Jan. 1, 2016, that reported CSF levels of NfL in neurologic or psychiatric conditions or in healthy controls. They included only studies that used the same commercially available immunoassay that has been used in most studies since 2006. The literature indicates that this enzyme-linked immunosorbent assay is sensitive and robust. Dr. Bridel and colleagues contacted study authors and requested their individual-level data.
The investigators sorted the most common neurologic conditions into three groups of similar disorders. The first group included inflammatory conditions of the CNS, such as multiple sclerosis, clinically isolated syndrome (CIS), and optic neuritis. The second group included dementia syndromes (such as Alzheimer’s disease, FTD, vascular dementia, and dementia with Lewy bodies) and amyotrophic lateral sclerosis (ALS). The third category included parkinsonian syndromes such as Parkinson’s disease, Parkinson’s disease dementia, multiple system atrophy (MSA), progressive supranuclear palsy (PSP), and corticobasal syndrome (CBS). The authors used generalized linear mixed-effects models to estimate the fixed effects of age, sex, and diagnosis on log-transformed NfL levels. They modeled cohort of origin as a random intercept.
NfL increased with age
Dr. Bridel and colleagues identified 153 relevant investigations, of which 44 met their inclusion criteria. The original investigators provided data sets for these studies, along with three previously unpublished data sets. The data sets included information from 10,059 participants (mean age, 59.7 years; 54.1% female). After excluding diagnostic categories with fewer than five observations per sex, Dr. Bridel and colleagues included data for 10,012 people in the analysis. In this population, the researchers identified 2,795 patients with inflammatory diseases of the CNS, 4,284 patients with dementia or predementia, 984 patients with parkinsonian disorders, and 1,332 healthy controls.
CSF level of NfL was elevated in most neurologic conditions, compared with healthy controls. The largest effect sizes were in cognitively impaired patients with HIV (21.36), patients with FTD/ALS (10.48), patients with ALS (7.58), and patients with Huntington’s disease (5.88).
In healthy controls, the level of NfL in CSF increased by 3.30% annually. The investigators also observed an association between age and CSF NfL level in people with subjective complaints, bipolar disorder, and most neurodegenerative conditions. They found no association, however, in patients with MS, HIV and cognitive impairment, and rapidly progressive neurodegenerative conditions (such as FTD, ALS, FTD/ALS, MSA, PSP, CBS, and Huntington’s disease). CSF level of NfL was 26.0% higher in men among healthy controls. This discrepancy also was observed in a minority of neurologic conditions, including MS, Alzheimer’s disease, vascular dementia, and Parkinson’s disease.
Mean CSF levels of NfL were similar between patients with inflammatory conditions of the CNS. Among dementias and related disorders, mean CNS level of NfL was significantly higher in FTD than in Alzheimer’s disease (2.08), vascular dementia (1.56), and dementia with Lewy bodies (2.50). Among parkinsonian syndromes, the mean CSF levels of NfL were higher in MSA, PSP, and CBS, compared with Parkinson’s disease.
Many factors influence NfL level in CSF
The association between CNS level of NfL with age among healthy controls “implies that age-specific reference values may be needed and that the diagnostic potential of CSF NfL may decrease with age,” said the researchers. The finding that CSF NfL level was higher in men in a minority of diagnoses has uncertain clinical significance, they added. Sex-specific reference values may be needed.
Dr. Bridel and colleagues found that age, sex, and cohort explained 46% of variation in CSF level of NfL, which suggests that many factors that determine this level have yet to be identified. Disease duration and disease severity could influence the CSF level of NfL, but the data sets that the investigators analyzed did not include this information.
Because CSF NfL level did not differ significantly between relapsing/remitting MS, secondary progressive MS, and primary progressive MS, this biomarker “may not differentiate acute inflammation-induced neuronal damage in the context of relapses from progressive neurodegeneration if the consequences of recent relapses or novel lesion formation are not considered,” said Dr. Bridel and colleagues. The findings do suggest, however, that CSF level of NfL can distinguish FTD from other dementias, as well as Parkinson’s disease from atypical parkinsonian syndromes. Furthermore, it is possible that the findings of this study can be translated to serum level of NfL, said the authors.
One of the study’s limitations was that diagnosis was based on clinical criteria, said Dr. Bridel and colleagues. In addition, the authors were unable to identify dementia of multifactorial origin, which might have reduced the differences in CSF NfL level distributions between dementia subtypes. Finally, the authors only analyzed studies that relied on a specific immunoassay for CSF NfL level.
The authors reported receiving funding from various pharmaceutical and biopharmaceutical companies, as well as from grants and research foundations. The funders did not influence the study design, data analysis, or interpretation, however.
SOURCE: Bridel C et al. JAMA Neurol. 2019 June 17. doi: 10.1001/jamaneurol.2019.1534.
according to an analysis published online ahead of print June 17 in JAMA Neurology. The biomarker has the potential to distinguish between frontotemporal dementia (FTD) and other dementia subtypes, as well as between Parkinson’s disease and atypical parkinsonian syndromes, said the investigators. It may be necessary to identify age- and sex-specific reference values for NfL, they added.
Neurologists have long understood CSF levels of NfL to be elevated in neurodegenerative conditions, but researchers previously had not compared these levels systematically among neurologic disorders. Similarly, the literature indicates a positive association between CSF NfL level and age in healthy controls, but this association has not been evaluated systematically in neurologic disorders. The resulting lack of clarity has impeded the use of NfL as a diagnostic biomarker.
A meta-analysis of CSF samples
Claire Bridel, MD, PhD, of the department of clinical chemistry at the VU University Medical Centre in Amsterdam and colleagues conducted a systematic review and meta-analysis to compare CSF levels of NfL among diagnoses, assess the associations of age and sex with NfL, and evaluate the potential of NfL as a diagnostic biomarker. The investigators searched PubMed for studies published between Jan. 1, 2006, and Jan. 1, 2016, that reported CSF levels of NfL in neurologic or psychiatric conditions or in healthy controls. They included only studies that used the same commercially available immunoassay that has been used in most studies since 2006. The literature indicates that this enzyme-linked immunosorbent assay is sensitive and robust. Dr. Bridel and colleagues contacted study authors and requested their individual-level data.
The investigators sorted the most common neurologic conditions into three groups of similar disorders. The first group included inflammatory conditions of the CNS, such as multiple sclerosis, clinically isolated syndrome (CIS), and optic neuritis. The second group included dementia syndromes (such as Alzheimer’s disease, FTD, vascular dementia, and dementia with Lewy bodies) and amyotrophic lateral sclerosis (ALS). The third category included parkinsonian syndromes such as Parkinson’s disease, Parkinson’s disease dementia, multiple system atrophy (MSA), progressive supranuclear palsy (PSP), and corticobasal syndrome (CBS). The authors used generalized linear mixed-effects models to estimate the fixed effects of age, sex, and diagnosis on log-transformed NfL levels. They modeled cohort of origin as a random intercept.
NfL increased with age
Dr. Bridel and colleagues identified 153 relevant investigations, of which 44 met their inclusion criteria. The original investigators provided data sets for these studies, along with three previously unpublished data sets. The data sets included information from 10,059 participants (mean age, 59.7 years; 54.1% female). After excluding diagnostic categories with fewer than five observations per sex, Dr. Bridel and colleagues included data for 10,012 people in the analysis. In this population, the researchers identified 2,795 patients with inflammatory diseases of the CNS, 4,284 patients with dementia or predementia, 984 patients with parkinsonian disorders, and 1,332 healthy controls.
CSF level of NfL was elevated in most neurologic conditions, compared with healthy controls. The largest effect sizes were in cognitively impaired patients with HIV (21.36), patients with FTD/ALS (10.48), patients with ALS (7.58), and patients with Huntington’s disease (5.88).
In healthy controls, the level of NfL in CSF increased by 3.30% annually. The investigators also observed an association between age and CSF NfL level in people with subjective complaints, bipolar disorder, and most neurodegenerative conditions. They found no association, however, in patients with MS, HIV and cognitive impairment, and rapidly progressive neurodegenerative conditions (such as FTD, ALS, FTD/ALS, MSA, PSP, CBS, and Huntington’s disease). CSF level of NfL was 26.0% higher in men among healthy controls. This discrepancy also was observed in a minority of neurologic conditions, including MS, Alzheimer’s disease, vascular dementia, and Parkinson’s disease.
Mean CSF levels of NfL were similar between patients with inflammatory conditions of the CNS. Among dementias and related disorders, mean CNS level of NfL was significantly higher in FTD than in Alzheimer’s disease (2.08), vascular dementia (1.56), and dementia with Lewy bodies (2.50). Among parkinsonian syndromes, the mean CSF levels of NfL were higher in MSA, PSP, and CBS, compared with Parkinson’s disease.
Many factors influence NfL level in CSF
The association between CNS level of NfL with age among healthy controls “implies that age-specific reference values may be needed and that the diagnostic potential of CSF NfL may decrease with age,” said the researchers. The finding that CSF NfL level was higher in men in a minority of diagnoses has uncertain clinical significance, they added. Sex-specific reference values may be needed.
Dr. Bridel and colleagues found that age, sex, and cohort explained 46% of variation in CSF level of NfL, which suggests that many factors that determine this level have yet to be identified. Disease duration and disease severity could influence the CSF level of NfL, but the data sets that the investigators analyzed did not include this information.
Because CSF NfL level did not differ significantly between relapsing/remitting MS, secondary progressive MS, and primary progressive MS, this biomarker “may not differentiate acute inflammation-induced neuronal damage in the context of relapses from progressive neurodegeneration if the consequences of recent relapses or novel lesion formation are not considered,” said Dr. Bridel and colleagues. The findings do suggest, however, that CSF level of NfL can distinguish FTD from other dementias, as well as Parkinson’s disease from atypical parkinsonian syndromes. Furthermore, it is possible that the findings of this study can be translated to serum level of NfL, said the authors.
One of the study’s limitations was that diagnosis was based on clinical criteria, said Dr. Bridel and colleagues. In addition, the authors were unable to identify dementia of multifactorial origin, which might have reduced the differences in CSF NfL level distributions between dementia subtypes. Finally, the authors only analyzed studies that relied on a specific immunoassay for CSF NfL level.
The authors reported receiving funding from various pharmaceutical and biopharmaceutical companies, as well as from grants and research foundations. The funders did not influence the study design, data analysis, or interpretation, however.
SOURCE: Bridel C et al. JAMA Neurol. 2019 June 17. doi: 10.1001/jamaneurol.2019.1534.
FROM JAMA NEUROLOGY
FDA approves pembrolizumab for advanced SCLC
The Food and Drug Administration has granted accelerated approval to pembrolizumab for patients with metastatic small cell lung cancer (SCLC) with disease progression on or after platinum-based chemotherapy and at least one other prior line of therapy.
Approval was based on an overall response rate of 19% among 83 patients with SCLC who had disease progression on or after two or more prior lines of therapy enrolled in two nonrandomized trials, according to the FDA.
SCLC cohorts in KEYNOTE-028 and KEYNOTE-158 received either pembrolizumab 200 mg intravenously every 3 weeks (n = 64) or 10 mg/kg intravenously every 2 weeks (n = 19). Treatment continued until documented disease progression, unacceptable toxicity, or for a maximum of 24 months.
The ORR was 19% (95% confidence interval, 11%-29%), while the complete response rate was 2%. Responses were durable for 6 months or longer in 94% of the 16 responding patients.
Common adverse reactions included fatigue, decreased appetite, cough, nausea, and constipation. The most frequent serious adverse reactions were pneumonia and pleural effusion.
The recommended dosage for SCLC treatment is 200 mg, administered as an intravenous infusion over 30 minutes every 3 weeks until disease progression, unacceptable toxicity, or up to 24 months in patients without disease progression, the FDA said.
Pembrolizumab is marketed as Keytruda by Merck.
The Food and Drug Administration has granted accelerated approval to pembrolizumab for patients with metastatic small cell lung cancer (SCLC) with disease progression on or after platinum-based chemotherapy and at least one other prior line of therapy.
Approval was based on an overall response rate of 19% among 83 patients with SCLC who had disease progression on or after two or more prior lines of therapy enrolled in two nonrandomized trials, according to the FDA.
SCLC cohorts in KEYNOTE-028 and KEYNOTE-158 received either pembrolizumab 200 mg intravenously every 3 weeks (n = 64) or 10 mg/kg intravenously every 2 weeks (n = 19). Treatment continued until documented disease progression, unacceptable toxicity, or for a maximum of 24 months.
The ORR was 19% (95% confidence interval, 11%-29%), while the complete response rate was 2%. Responses were durable for 6 months or longer in 94% of the 16 responding patients.
Common adverse reactions included fatigue, decreased appetite, cough, nausea, and constipation. The most frequent serious adverse reactions were pneumonia and pleural effusion.
The recommended dosage for SCLC treatment is 200 mg, administered as an intravenous infusion over 30 minutes every 3 weeks until disease progression, unacceptable toxicity, or up to 24 months in patients without disease progression, the FDA said.
Pembrolizumab is marketed as Keytruda by Merck.
The Food and Drug Administration has granted accelerated approval to pembrolizumab for patients with metastatic small cell lung cancer (SCLC) with disease progression on or after platinum-based chemotherapy and at least one other prior line of therapy.
Approval was based on an overall response rate of 19% among 83 patients with SCLC who had disease progression on or after two or more prior lines of therapy enrolled in two nonrandomized trials, according to the FDA.
SCLC cohorts in KEYNOTE-028 and KEYNOTE-158 received either pembrolizumab 200 mg intravenously every 3 weeks (n = 64) or 10 mg/kg intravenously every 2 weeks (n = 19). Treatment continued until documented disease progression, unacceptable toxicity, or for a maximum of 24 months.
The ORR was 19% (95% confidence interval, 11%-29%), while the complete response rate was 2%. Responses were durable for 6 months or longer in 94% of the 16 responding patients.
Common adverse reactions included fatigue, decreased appetite, cough, nausea, and constipation. The most frequent serious adverse reactions were pneumonia and pleural effusion.
The recommended dosage for SCLC treatment is 200 mg, administered as an intravenous infusion over 30 minutes every 3 weeks until disease progression, unacceptable toxicity, or up to 24 months in patients without disease progression, the FDA said.
Pembrolizumab is marketed as Keytruda by Merck.
Gepant Safety & Lack of Liver Toxicity: Highlights from AAN 2019
The gepants and monoclonal antibodies (mAbs) against calcitonin gene-related peptide (CGRP) and its receptor appear to be effective, tolerable and safe, according to several posters recently presented at the 2019 American Academy of Neurology (AAN) Annual Meeting in Philadelphia. Information was presented on the 3 gepants being studied for US Food and Drug Administration (FDA) approval: 1 for acute care of migraine, 1 for prevention and 1 for both (though the presented data for rimegepant only covers acute care).
All drugs cause a small degree of adverse events (AEs), somewhat more than placebo. Based on the presented data, it seems that those associated with 3 times higher than normal elevation of liver enzymes, were usually not found to be the cause of that elevation. At no time was bilirubin elevated. This shows that all of these gepants appear to be effective and safe, despite the fact that some were found to have cause liver toxicity many years ago.
The first gepant study to be published was on olcegepant in 2004, in the New England Journal of Medicine. Professor Jes Olesen, MD, was the lead author of the study, which detailed the efficacy and safety of this small molecule CGRP receptor antagonist. Olcegepant was in an intravenous formulation and the plan was to convert it to a tablet, which never happened. Another company then produced telcagepant as a tablet and it was shown to be safe and effective in 2 large, multicenter, double-blind trials. Before receiving FDA approval for the acute care of migraine, it was studied on a daily basis for migraine prevention. It was found to cause some liver toxicity, so development was stopped. At that time several other gepants in development were placed on the shelf, partially for the fear of liver toxicity. The FDA is unlikely to approve a drug with significant liver toxicity, which can cause a range of symptoms including jaundice, itching, abdominal pain, fatigue, loss of appetite, nausea, vomiting, rash, and weight loss.
In the next few years we will have 4 mAbs for the prevention of migraine, and 3 gepants if all studies are positive. Below are the key takeaways from the presented posters on ubrogepant and atogepant, as information that is currently available on rimegepant.
Key Takeaways:
- Ubrogepant – Ailani J, Hutchinson S, Lipton R, et al.
- Intermittent use of ubrogepant for the acute treatment of migraine over 1 year was well-tolerated with no identified safety concerns. Throughout the 1-year, Phase III study of 1254 participants, 22,454 migraine attacks were treated with 31,968 doses of ubrogepant.
- Twenty cases of ALT/AST ≥3x ULN were reported and adjudicated by an independent panel of liver experts blinded to treatment.
- Of the 20 cases, 17 (4 usual care, 3 ubrogepant 50-mg, and 10 ubrogepant 100-mg) were determined to be unlikely related based on plausible alternative etiology/confounding factors.
- Just 2 cases (both ubrogepant 50-mg) were described as possibly related to study medication and 1 case (ubrogepant 100-mg) was adjudicated as probably related; however, confounding factors were noted.
- All cases were asymptomatic with no concurrent bilirubin elevation. ALT/AST elevations resolved in those who continued dosing.
- Atogepant – Goadsby PJ, Dodick DW, Trugman JM, et al.
- In a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial of adults with a history of migraine, with or without aura, atogepant was well tolerated with no treatment-related serious AEs.
- Of the 834 randomized subjects, 825 were evaluated in the safety population. Treatment-emergent AEs were reported by 480 subjects (58.2%), and for 170 (20.6%), the AEs were considered treatment-related. Seven subjects (0.8%) reported serious AEs, but none were determined as treatment related.
- There were 10 cases of treatment-emergent ALT/AST elevations >3x the upper limit of normal, and this was balanced across the treatment dosage groups (10 mg QD, 30 mg QD, 30 mg BID, 60 mg QD, and 60 mg BID).
- Rimegepant – See Biogen press release for more information
- In December 2019, Biohaven announced initial positive results from an ongoing long-term, open-label safety study for rimegepant.
- The interim results included hepatic safety and tolerability data of rimegepant 75 mg in study participants based on a review of adverse events and regularly scheduled liver function tests.
- A panel of external independent liver experts provided a consensus based on the Drug-Induced Liver Injury Network (DILIN) causality assessment, determining that there were no liver cases probably related to the study drug and that there were no Hy’s Law cases identified.
- The panel also concluded that there were no liver safety signals detected and that, compared to placebo arms of other migraine treatments, there was a very low incidence of overall elevations of liver abnormalities.
The gepants and monoclonal antibodies (mAbs) against calcitonin gene-related peptide (CGRP) and its receptor appear to be effective, tolerable and safe, according to several posters recently presented at the 2019 American Academy of Neurology (AAN) Annual Meeting in Philadelphia. Information was presented on the 3 gepants being studied for US Food and Drug Administration (FDA) approval: 1 for acute care of migraine, 1 for prevention and 1 for both (though the presented data for rimegepant only covers acute care).
All drugs cause a small degree of adverse events (AEs), somewhat more than placebo. Based on the presented data, it seems that those associated with 3 times higher than normal elevation of liver enzymes, were usually not found to be the cause of that elevation. At no time was bilirubin elevated. This shows that all of these gepants appear to be effective and safe, despite the fact that some were found to have cause liver toxicity many years ago.
The first gepant study to be published was on olcegepant in 2004, in the New England Journal of Medicine. Professor Jes Olesen, MD, was the lead author of the study, which detailed the efficacy and safety of this small molecule CGRP receptor antagonist. Olcegepant was in an intravenous formulation and the plan was to convert it to a tablet, which never happened. Another company then produced telcagepant as a tablet and it was shown to be safe and effective in 2 large, multicenter, double-blind trials. Before receiving FDA approval for the acute care of migraine, it was studied on a daily basis for migraine prevention. It was found to cause some liver toxicity, so development was stopped. At that time several other gepants in development were placed on the shelf, partially for the fear of liver toxicity. The FDA is unlikely to approve a drug with significant liver toxicity, which can cause a range of symptoms including jaundice, itching, abdominal pain, fatigue, loss of appetite, nausea, vomiting, rash, and weight loss.
In the next few years we will have 4 mAbs for the prevention of migraine, and 3 gepants if all studies are positive. Below are the key takeaways from the presented posters on ubrogepant and atogepant, as information that is currently available on rimegepant.
Key Takeaways:
- Ubrogepant – Ailani J, Hutchinson S, Lipton R, et al.
- Intermittent use of ubrogepant for the acute treatment of migraine over 1 year was well-tolerated with no identified safety concerns. Throughout the 1-year, Phase III study of 1254 participants, 22,454 migraine attacks were treated with 31,968 doses of ubrogepant.
- Twenty cases of ALT/AST ≥3x ULN were reported and adjudicated by an independent panel of liver experts blinded to treatment.
- Of the 20 cases, 17 (4 usual care, 3 ubrogepant 50-mg, and 10 ubrogepant 100-mg) were determined to be unlikely related based on plausible alternative etiology/confounding factors.
- Just 2 cases (both ubrogepant 50-mg) were described as possibly related to study medication and 1 case (ubrogepant 100-mg) was adjudicated as probably related; however, confounding factors were noted.
- All cases were asymptomatic with no concurrent bilirubin elevation. ALT/AST elevations resolved in those who continued dosing.
- Atogepant – Goadsby PJ, Dodick DW, Trugman JM, et al.
- In a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial of adults with a history of migraine, with or without aura, atogepant was well tolerated with no treatment-related serious AEs.
- Of the 834 randomized subjects, 825 were evaluated in the safety population. Treatment-emergent AEs were reported by 480 subjects (58.2%), and for 170 (20.6%), the AEs were considered treatment-related. Seven subjects (0.8%) reported serious AEs, but none were determined as treatment related.
- There were 10 cases of treatment-emergent ALT/AST elevations >3x the upper limit of normal, and this was balanced across the treatment dosage groups (10 mg QD, 30 mg QD, 30 mg BID, 60 mg QD, and 60 mg BID).
- Rimegepant – See Biogen press release for more information
- In December 2019, Biohaven announced initial positive results from an ongoing long-term, open-label safety study for rimegepant.
- The interim results included hepatic safety and tolerability data of rimegepant 75 mg in study participants based on a review of adverse events and regularly scheduled liver function tests.
- A panel of external independent liver experts provided a consensus based on the Drug-Induced Liver Injury Network (DILIN) causality assessment, determining that there were no liver cases probably related to the study drug and that there were no Hy’s Law cases identified.
- The panel also concluded that there were no liver safety signals detected and that, compared to placebo arms of other migraine treatments, there was a very low incidence of overall elevations of liver abnormalities.
The gepants and monoclonal antibodies (mAbs) against calcitonin gene-related peptide (CGRP) and its receptor appear to be effective, tolerable and safe, according to several posters recently presented at the 2019 American Academy of Neurology (AAN) Annual Meeting in Philadelphia. Information was presented on the 3 gepants being studied for US Food and Drug Administration (FDA) approval: 1 for acute care of migraine, 1 for prevention and 1 for both (though the presented data for rimegepant only covers acute care).
All drugs cause a small degree of adverse events (AEs), somewhat more than placebo. Based on the presented data, it seems that those associated with 3 times higher than normal elevation of liver enzymes, were usually not found to be the cause of that elevation. At no time was bilirubin elevated. This shows that all of these gepants appear to be effective and safe, despite the fact that some were found to have cause liver toxicity many years ago.
The first gepant study to be published was on olcegepant in 2004, in the New England Journal of Medicine. Professor Jes Olesen, MD, was the lead author of the study, which detailed the efficacy and safety of this small molecule CGRP receptor antagonist. Olcegepant was in an intravenous formulation and the plan was to convert it to a tablet, which never happened. Another company then produced telcagepant as a tablet and it was shown to be safe and effective in 2 large, multicenter, double-blind trials. Before receiving FDA approval for the acute care of migraine, it was studied on a daily basis for migraine prevention. It was found to cause some liver toxicity, so development was stopped. At that time several other gepants in development were placed on the shelf, partially for the fear of liver toxicity. The FDA is unlikely to approve a drug with significant liver toxicity, which can cause a range of symptoms including jaundice, itching, abdominal pain, fatigue, loss of appetite, nausea, vomiting, rash, and weight loss.
In the next few years we will have 4 mAbs for the prevention of migraine, and 3 gepants if all studies are positive. Below are the key takeaways from the presented posters on ubrogepant and atogepant, as information that is currently available on rimegepant.
Key Takeaways:
- Ubrogepant – Ailani J, Hutchinson S, Lipton R, et al.
- Intermittent use of ubrogepant for the acute treatment of migraine over 1 year was well-tolerated with no identified safety concerns. Throughout the 1-year, Phase III study of 1254 participants, 22,454 migraine attacks were treated with 31,968 doses of ubrogepant.
- Twenty cases of ALT/AST ≥3x ULN were reported and adjudicated by an independent panel of liver experts blinded to treatment.
- Of the 20 cases, 17 (4 usual care, 3 ubrogepant 50-mg, and 10 ubrogepant 100-mg) were determined to be unlikely related based on plausible alternative etiology/confounding factors.
- Just 2 cases (both ubrogepant 50-mg) were described as possibly related to study medication and 1 case (ubrogepant 100-mg) was adjudicated as probably related; however, confounding factors were noted.
- All cases were asymptomatic with no concurrent bilirubin elevation. ALT/AST elevations resolved in those who continued dosing.
- Atogepant – Goadsby PJ, Dodick DW, Trugman JM, et al.
- In a multicenter, randomized, double-blind, placebo-controlled, parallel-group trial of adults with a history of migraine, with or without aura, atogepant was well tolerated with no treatment-related serious AEs.
- Of the 834 randomized subjects, 825 were evaluated in the safety population. Treatment-emergent AEs were reported by 480 subjects (58.2%), and for 170 (20.6%), the AEs were considered treatment-related. Seven subjects (0.8%) reported serious AEs, but none were determined as treatment related.
- There were 10 cases of treatment-emergent ALT/AST elevations >3x the upper limit of normal, and this was balanced across the treatment dosage groups (10 mg QD, 30 mg QD, 30 mg BID, 60 mg QD, and 60 mg BID).
- Rimegepant – See Biogen press release for more information
- In December 2019, Biohaven announced initial positive results from an ongoing long-term, open-label safety study for rimegepant.
- The interim results included hepatic safety and tolerability data of rimegepant 75 mg in study participants based on a review of adverse events and regularly scheduled liver function tests.
- A panel of external independent liver experts provided a consensus based on the Drug-Induced Liver Injury Network (DILIN) causality assessment, determining that there were no liver cases probably related to the study drug and that there were no Hy’s Law cases identified.
- The panel also concluded that there were no liver safety signals detected and that, compared to placebo arms of other migraine treatments, there was a very low incidence of overall elevations of liver abnormalities.
Patients with mood disorders may have altered microbiome
Discuss dietary interventions, such as probiotics, as ‘supplemental therapeutic options’
CRYSTAL CITY, VA. – Individuals with mood disorders might have an altered microbiome, but more information is needed to understand how the microorganisms that make up the microbiome affect patients’ health, an expert said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
“An increased understanding of the neurobiology of the microbiome is required so that the benefit that these microorganisms serve to human health can be fully harnessed,” said Emily G. Severance, PhD, assistant professor of pediatrics at John Hopkins University, Baltimore.
Diseases that involve the microbiome include those with a single identifiable infectious agent that produces persistent inflammation, central nervous system diseases with mucosal surface involvement, and diseases with “variable response to antibiotic and anti-inflammatory agents.”
“It’s becoming clear that [the microbiome is] integral for the modulation of the central nervous system,” which occurs through neurotransmitter production, Dr. Severance said at the meeting presented by Global Academy for Medical Education.
“We have an extensive enteric nervous system that has the very same receptors that the brain does,” she said. “If you have those receptors activated in the gut or [are] having the neurotransmitters produced in the gut, and if there’s a way for those neurotransmitters to reach the brain, that’s a very powerful mechanism to illustrate the gut-brain axis.”
In addition to neuropsychiatric diseases, the microbiome also can be involved in inflammatory gastrointestinal, systemic rheumatoid and autoimmune, chronic inflammatory lung, and periodontal diseases, as well as immune-mediated skin disorders. Mood disorders in particular have evidence for dysbiosis in low-level inflammation and leaky gut pathology, which is present in patients with depression, Dr. Severance said. “All these data suggest that We can do that because gut bacteria are easily accessed and can be altered through probiotics, prebiotics, diet, and fecal transplant, and in patients, Lactobacillus and Bifidobacterium combinations may improve mood, reduce anxiety, and enhance cognitive function.”
In addition, epidemiological studies show that antibiotic exposure can be a risk factor for developing mood disorders. One recent study found that anti-infective agents, particularly antibiotics, increased the risk of schizophrenia (hazard rate ratio, 2.05; 95% confidence interval, 1.77-2.38) and affective disorders (HRR, 2.59; 95% CI, 2.31-2.89), which the researchers attributed to brain inflammation, the microbiome, and environmental factors (Acta Psychiatr Scand. 2016 Nov 21. doi: 10.1111/acps.12671). In mice, other researchers found that those that received a fecal transplant with a “depression microbiota” showed symptoms of major depressive disorder, compared with mice that received a “healthy microbiota.” Those results suggest that change in microbiota can induce mood disorders (Mol Psychiatry. 2016 Apr 12. doi: 10.1038/mp.2016.44).
The evidence for probiotics is mixed, primarily because the study population in trials are so heterogeneous, but there is evidence for its efficacy in patients with mood disorders, Dr. Severance said. Probiotics have been shown to prevent rehospitalization for patients in mania. For example, one study showed reduced rehospitalization in patients with mania (8 of 33 patients) who received probiotics, compared with placebo (24 of 33 patients). Also, probiotic use was associated with fewer days of rehospitalization (Bipolar Disord. 2018 Apr 25. doi: 10. 1111/bdi.12652).
Meanwhile, a pilot study analyzing patients with irritable bowel syndrome and mild to moderate anxiety and/or depression found use of B. longum in this population reduced depression scores, but not anxiety or irritable bowel syndrome symptoms, compared with placebo (Gastroenterology. 2017 May 5. doi: 10.1053/j.gastro.2017.05.003).
Probiotic efficacy can be variable for patients with mood disorders, but the intervention is a “relatively low-risk, potentially high reward” option for these patients, Dr. Severance said. “Clinicians should inquire about patient GI conditions and overall GI health. Dietary interventions and the use of probiotics and their limitations should be discussed as supplemental therapeutic options.”
Dr. Severance reported no relevant financial disclosures.
Global Academy and this news organization are owned by the same parent company.
Discuss dietary interventions, such as probiotics, as ‘supplemental therapeutic options’
Discuss dietary interventions, such as probiotics, as ‘supplemental therapeutic options’
CRYSTAL CITY, VA. – Individuals with mood disorders might have an altered microbiome, but more information is needed to understand how the microorganisms that make up the microbiome affect patients’ health, an expert said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
“An increased understanding of the neurobiology of the microbiome is required so that the benefit that these microorganisms serve to human health can be fully harnessed,” said Emily G. Severance, PhD, assistant professor of pediatrics at John Hopkins University, Baltimore.
Diseases that involve the microbiome include those with a single identifiable infectious agent that produces persistent inflammation, central nervous system diseases with mucosal surface involvement, and diseases with “variable response to antibiotic and anti-inflammatory agents.”
“It’s becoming clear that [the microbiome is] integral for the modulation of the central nervous system,” which occurs through neurotransmitter production, Dr. Severance said at the meeting presented by Global Academy for Medical Education.
“We have an extensive enteric nervous system that has the very same receptors that the brain does,” she said. “If you have those receptors activated in the gut or [are] having the neurotransmitters produced in the gut, and if there’s a way for those neurotransmitters to reach the brain, that’s a very powerful mechanism to illustrate the gut-brain axis.”
In addition to neuropsychiatric diseases, the microbiome also can be involved in inflammatory gastrointestinal, systemic rheumatoid and autoimmune, chronic inflammatory lung, and periodontal diseases, as well as immune-mediated skin disorders. Mood disorders in particular have evidence for dysbiosis in low-level inflammation and leaky gut pathology, which is present in patients with depression, Dr. Severance said. “All these data suggest that We can do that because gut bacteria are easily accessed and can be altered through probiotics, prebiotics, diet, and fecal transplant, and in patients, Lactobacillus and Bifidobacterium combinations may improve mood, reduce anxiety, and enhance cognitive function.”
In addition, epidemiological studies show that antibiotic exposure can be a risk factor for developing mood disorders. One recent study found that anti-infective agents, particularly antibiotics, increased the risk of schizophrenia (hazard rate ratio, 2.05; 95% confidence interval, 1.77-2.38) and affective disorders (HRR, 2.59; 95% CI, 2.31-2.89), which the researchers attributed to brain inflammation, the microbiome, and environmental factors (Acta Psychiatr Scand. 2016 Nov 21. doi: 10.1111/acps.12671). In mice, other researchers found that those that received a fecal transplant with a “depression microbiota” showed symptoms of major depressive disorder, compared with mice that received a “healthy microbiota.” Those results suggest that change in microbiota can induce mood disorders (Mol Psychiatry. 2016 Apr 12. doi: 10.1038/mp.2016.44).
The evidence for probiotics is mixed, primarily because the study population in trials are so heterogeneous, but there is evidence for its efficacy in patients with mood disorders, Dr. Severance said. Probiotics have been shown to prevent rehospitalization for patients in mania. For example, one study showed reduced rehospitalization in patients with mania (8 of 33 patients) who received probiotics, compared with placebo (24 of 33 patients). Also, probiotic use was associated with fewer days of rehospitalization (Bipolar Disord. 2018 Apr 25. doi: 10. 1111/bdi.12652).
Meanwhile, a pilot study analyzing patients with irritable bowel syndrome and mild to moderate anxiety and/or depression found use of B. longum in this population reduced depression scores, but not anxiety or irritable bowel syndrome symptoms, compared with placebo (Gastroenterology. 2017 May 5. doi: 10.1053/j.gastro.2017.05.003).
Probiotic efficacy can be variable for patients with mood disorders, but the intervention is a “relatively low-risk, potentially high reward” option for these patients, Dr. Severance said. “Clinicians should inquire about patient GI conditions and overall GI health. Dietary interventions and the use of probiotics and their limitations should be discussed as supplemental therapeutic options.”
Dr. Severance reported no relevant financial disclosures.
Global Academy and this news organization are owned by the same parent company.
CRYSTAL CITY, VA. – Individuals with mood disorders might have an altered microbiome, but more information is needed to understand how the microorganisms that make up the microbiome affect patients’ health, an expert said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
“An increased understanding of the neurobiology of the microbiome is required so that the benefit that these microorganisms serve to human health can be fully harnessed,” said Emily G. Severance, PhD, assistant professor of pediatrics at John Hopkins University, Baltimore.
Diseases that involve the microbiome include those with a single identifiable infectious agent that produces persistent inflammation, central nervous system diseases with mucosal surface involvement, and diseases with “variable response to antibiotic and anti-inflammatory agents.”
“It’s becoming clear that [the microbiome is] integral for the modulation of the central nervous system,” which occurs through neurotransmitter production, Dr. Severance said at the meeting presented by Global Academy for Medical Education.
“We have an extensive enteric nervous system that has the very same receptors that the brain does,” she said. “If you have those receptors activated in the gut or [are] having the neurotransmitters produced in the gut, and if there’s a way for those neurotransmitters to reach the brain, that’s a very powerful mechanism to illustrate the gut-brain axis.”
In addition to neuropsychiatric diseases, the microbiome also can be involved in inflammatory gastrointestinal, systemic rheumatoid and autoimmune, chronic inflammatory lung, and periodontal diseases, as well as immune-mediated skin disorders. Mood disorders in particular have evidence for dysbiosis in low-level inflammation and leaky gut pathology, which is present in patients with depression, Dr. Severance said. “All these data suggest that We can do that because gut bacteria are easily accessed and can be altered through probiotics, prebiotics, diet, and fecal transplant, and in patients, Lactobacillus and Bifidobacterium combinations may improve mood, reduce anxiety, and enhance cognitive function.”
In addition, epidemiological studies show that antibiotic exposure can be a risk factor for developing mood disorders. One recent study found that anti-infective agents, particularly antibiotics, increased the risk of schizophrenia (hazard rate ratio, 2.05; 95% confidence interval, 1.77-2.38) and affective disorders (HRR, 2.59; 95% CI, 2.31-2.89), which the researchers attributed to brain inflammation, the microbiome, and environmental factors (Acta Psychiatr Scand. 2016 Nov 21. doi: 10.1111/acps.12671). In mice, other researchers found that those that received a fecal transplant with a “depression microbiota” showed symptoms of major depressive disorder, compared with mice that received a “healthy microbiota.” Those results suggest that change in microbiota can induce mood disorders (Mol Psychiatry. 2016 Apr 12. doi: 10.1038/mp.2016.44).
The evidence for probiotics is mixed, primarily because the study population in trials are so heterogeneous, but there is evidence for its efficacy in patients with mood disorders, Dr. Severance said. Probiotics have been shown to prevent rehospitalization for patients in mania. For example, one study showed reduced rehospitalization in patients with mania (8 of 33 patients) who received probiotics, compared with placebo (24 of 33 patients). Also, probiotic use was associated with fewer days of rehospitalization (Bipolar Disord. 2018 Apr 25. doi: 10. 1111/bdi.12652).
Meanwhile, a pilot study analyzing patients with irritable bowel syndrome and mild to moderate anxiety and/or depression found use of B. longum in this population reduced depression scores, but not anxiety or irritable bowel syndrome symptoms, compared with placebo (Gastroenterology. 2017 May 5. doi: 10.1053/j.gastro.2017.05.003).
Probiotic efficacy can be variable for patients with mood disorders, but the intervention is a “relatively low-risk, potentially high reward” option for these patients, Dr. Severance said. “Clinicians should inquire about patient GI conditions and overall GI health. Dietary interventions and the use of probiotics and their limitations should be discussed as supplemental therapeutic options.”
Dr. Severance reported no relevant financial disclosures.
Global Academy and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM FOCUS ON NEUROPSYCHIATRY 2019
Durable transfusion independence in MDS with imetelstat
AMSTERDAM – For patients with low-risk myelodysplastic syndrome (MDS) for whom erythropoietin therapy has failed, the novel telomerase inhibitor imetelstat may provide long-lasting independence from transfusion, investigators reported.
Among 38 patients with low-risk MDS who had relapsed or were refractory to treatment with an erythropoiesis stimulating agent (ESA) who received imetelstat, 16 (42%) were free from the need for transfusion for at least 8 weeks, with one patient being transfusion free for up to 141 weeks, reported Pierre Fenaux, MD, of Hôpital Saint-Louis in Paris.
Patients with a generally worse prognosis “tended to respond better to imetelstat in terms of transfusion independence, which suggests that the drug is promising for higher-risk MDS,” he said at a briefing prior to his presentation of the data at the annual congress of the European Hematology Association.
Imetelstat is a first-in-class telomerase inhibitor targeting cells with short telomere lengths and active telomerase, the enzyme that maintains telomere length. Higher telomerase activity and shorter telomeres are predictive of shorter overall survival in patients with MDS, Dr. Fenaux explained.
He and colleagues enrolled 38 patients, median age 71.5 years, with low-risk MDS, with an International Prognostic Scoring System (IPSS) score of low or intermediate-1, whose disease was relapsed or refractory to ESA or to erythropoietin at a dose of more than 500 mU/mL. Of this group, 24 patients had IPSS low disease, 14 had intermediate-1 disease.
The median transfusion burden was 8 units per 8 weeks (range 4-14). The majority of patients (34, or 89%) had received prior ESAs.
The patients were transfusion dependent, defined as the need for 4 or more units of red blood cells within 8 weeks over the 16 weeks prior to study entry.
No patients had the 5q deletion, and no patients had received either a hypomethylating agent or lenolidamide (Revlimid), neither of which are approved for this indication in Europe.
The patients received imetelstat 7.5 mg/kg intravenously every 4 weeks.
As noted earlier, 16 patients (42%) achieved the primary endpoint of 8-week transfusion independence, with a median duration of 85.9 weeks (range 8-141 weeks).
Eleven patients (29%) had transfusion independence lasting at least 24 weeks – a secondary endpoint – and 26 (68%) met International Working Group 2006 criteria for a HI-E (erythroid) response, with 12 of these patients having an increase in hemoglobin of 1.5 g/dL or greater lasting for at least 8 weeks, and all 26 having a reduction in transfusions of 4 or more units over 8 weeks.
There was evidence to suggest a disease-modifying effect of imetelstat, with five patients achieving a complete response (CR), and five having a marrow CR.
The most frequent adverse events were manageable and reversible grade 3 or greater cytopenias, but there were no new safety signals seen. Two patients were hospitalized for febrile neutropenia, but there were no treatment-related deaths.
Based on these results, investigators are planning a phase 3 study comparing imetelstat with placebo in a 2:1 ratio. The trial is scheduled to begin in the late summer or fall of 2019.
When asked if imetelstat might have off-target effects by inhibiting telomerase in other cells, Dr. Fenaux replied that the mechanism of action is unclear, and that its potential effects on erythropoiesis are still unknown.
Briefing moderator Anton Hagenbeek, MD, of Amsterdam University Medical Center, commented on the drug’s potential for treating MDS, and asked whether investigators are considering combining it with other therapies for MDS.
“I think the first step will be to study it in high-risk MDS as a single agent before combining it, including with hypomethylating agents, et cetera,” Dr. Fenaux replied.
SOURCE: Fenaux P et al. EHA 2019, Abstract S837.
AMSTERDAM – For patients with low-risk myelodysplastic syndrome (MDS) for whom erythropoietin therapy has failed, the novel telomerase inhibitor imetelstat may provide long-lasting independence from transfusion, investigators reported.
Among 38 patients with low-risk MDS who had relapsed or were refractory to treatment with an erythropoiesis stimulating agent (ESA) who received imetelstat, 16 (42%) were free from the need for transfusion for at least 8 weeks, with one patient being transfusion free for up to 141 weeks, reported Pierre Fenaux, MD, of Hôpital Saint-Louis in Paris.
Patients with a generally worse prognosis “tended to respond better to imetelstat in terms of transfusion independence, which suggests that the drug is promising for higher-risk MDS,” he said at a briefing prior to his presentation of the data at the annual congress of the European Hematology Association.
Imetelstat is a first-in-class telomerase inhibitor targeting cells with short telomere lengths and active telomerase, the enzyme that maintains telomere length. Higher telomerase activity and shorter telomeres are predictive of shorter overall survival in patients with MDS, Dr. Fenaux explained.
He and colleagues enrolled 38 patients, median age 71.5 years, with low-risk MDS, with an International Prognostic Scoring System (IPSS) score of low or intermediate-1, whose disease was relapsed or refractory to ESA or to erythropoietin at a dose of more than 500 mU/mL. Of this group, 24 patients had IPSS low disease, 14 had intermediate-1 disease.
The median transfusion burden was 8 units per 8 weeks (range 4-14). The majority of patients (34, or 89%) had received prior ESAs.
The patients were transfusion dependent, defined as the need for 4 or more units of red blood cells within 8 weeks over the 16 weeks prior to study entry.
No patients had the 5q deletion, and no patients had received either a hypomethylating agent or lenolidamide (Revlimid), neither of which are approved for this indication in Europe.
The patients received imetelstat 7.5 mg/kg intravenously every 4 weeks.
As noted earlier, 16 patients (42%) achieved the primary endpoint of 8-week transfusion independence, with a median duration of 85.9 weeks (range 8-141 weeks).
Eleven patients (29%) had transfusion independence lasting at least 24 weeks – a secondary endpoint – and 26 (68%) met International Working Group 2006 criteria for a HI-E (erythroid) response, with 12 of these patients having an increase in hemoglobin of 1.5 g/dL or greater lasting for at least 8 weeks, and all 26 having a reduction in transfusions of 4 or more units over 8 weeks.
There was evidence to suggest a disease-modifying effect of imetelstat, with five patients achieving a complete response (CR), and five having a marrow CR.
The most frequent adverse events were manageable and reversible grade 3 or greater cytopenias, but there were no new safety signals seen. Two patients were hospitalized for febrile neutropenia, but there were no treatment-related deaths.
Based on these results, investigators are planning a phase 3 study comparing imetelstat with placebo in a 2:1 ratio. The trial is scheduled to begin in the late summer or fall of 2019.
When asked if imetelstat might have off-target effects by inhibiting telomerase in other cells, Dr. Fenaux replied that the mechanism of action is unclear, and that its potential effects on erythropoiesis are still unknown.
Briefing moderator Anton Hagenbeek, MD, of Amsterdam University Medical Center, commented on the drug’s potential for treating MDS, and asked whether investigators are considering combining it with other therapies for MDS.
“I think the first step will be to study it in high-risk MDS as a single agent before combining it, including with hypomethylating agents, et cetera,” Dr. Fenaux replied.
SOURCE: Fenaux P et al. EHA 2019, Abstract S837.
AMSTERDAM – For patients with low-risk myelodysplastic syndrome (MDS) for whom erythropoietin therapy has failed, the novel telomerase inhibitor imetelstat may provide long-lasting independence from transfusion, investigators reported.
Among 38 patients with low-risk MDS who had relapsed or were refractory to treatment with an erythropoiesis stimulating agent (ESA) who received imetelstat, 16 (42%) were free from the need for transfusion for at least 8 weeks, with one patient being transfusion free for up to 141 weeks, reported Pierre Fenaux, MD, of Hôpital Saint-Louis in Paris.
Patients with a generally worse prognosis “tended to respond better to imetelstat in terms of transfusion independence, which suggests that the drug is promising for higher-risk MDS,” he said at a briefing prior to his presentation of the data at the annual congress of the European Hematology Association.
Imetelstat is a first-in-class telomerase inhibitor targeting cells with short telomere lengths and active telomerase, the enzyme that maintains telomere length. Higher telomerase activity and shorter telomeres are predictive of shorter overall survival in patients with MDS, Dr. Fenaux explained.
He and colleagues enrolled 38 patients, median age 71.5 years, with low-risk MDS, with an International Prognostic Scoring System (IPSS) score of low or intermediate-1, whose disease was relapsed or refractory to ESA or to erythropoietin at a dose of more than 500 mU/mL. Of this group, 24 patients had IPSS low disease, 14 had intermediate-1 disease.
The median transfusion burden was 8 units per 8 weeks (range 4-14). The majority of patients (34, or 89%) had received prior ESAs.
The patients were transfusion dependent, defined as the need for 4 or more units of red blood cells within 8 weeks over the 16 weeks prior to study entry.
No patients had the 5q deletion, and no patients had received either a hypomethylating agent or lenolidamide (Revlimid), neither of which are approved for this indication in Europe.
The patients received imetelstat 7.5 mg/kg intravenously every 4 weeks.
As noted earlier, 16 patients (42%) achieved the primary endpoint of 8-week transfusion independence, with a median duration of 85.9 weeks (range 8-141 weeks).
Eleven patients (29%) had transfusion independence lasting at least 24 weeks – a secondary endpoint – and 26 (68%) met International Working Group 2006 criteria for a HI-E (erythroid) response, with 12 of these patients having an increase in hemoglobin of 1.5 g/dL or greater lasting for at least 8 weeks, and all 26 having a reduction in transfusions of 4 or more units over 8 weeks.
There was evidence to suggest a disease-modifying effect of imetelstat, with five patients achieving a complete response (CR), and five having a marrow CR.
The most frequent adverse events were manageable and reversible grade 3 or greater cytopenias, but there were no new safety signals seen. Two patients were hospitalized for febrile neutropenia, but there were no treatment-related deaths.
Based on these results, investigators are planning a phase 3 study comparing imetelstat with placebo in a 2:1 ratio. The trial is scheduled to begin in the late summer or fall of 2019.
When asked if imetelstat might have off-target effects by inhibiting telomerase in other cells, Dr. Fenaux replied that the mechanism of action is unclear, and that its potential effects on erythropoiesis are still unknown.
Briefing moderator Anton Hagenbeek, MD, of Amsterdam University Medical Center, commented on the drug’s potential for treating MDS, and asked whether investigators are considering combining it with other therapies for MDS.
“I think the first step will be to study it in high-risk MDS as a single agent before combining it, including with hypomethylating agents, et cetera,” Dr. Fenaux replied.
SOURCE: Fenaux P et al. EHA 2019, Abstract S837.
REPORTING FROM EHA CONGRESS